User login
The Official Newspaper of the American Association for Thoracic Surgery
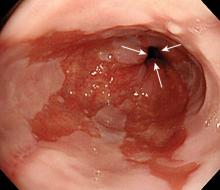
Alter Surveillance for Barrett's Esophagus?
The incidence of esophageal adenocarcinoma among patients with Barrett’s esophagus was only 1.2 cases per 1,000 person-years in a study of the entire population of Denmark reported in the New England Journal of Medicine.
That rate is four to five times lower than rates reported previously, said Dr. Frederik Hvid-Jensen of the department of surgical gastroenterology at Aarhus (Denmark) University and his associates.
"Our study provides solid evidence that esophageal adenocarcinoma will develop in very few patients with Barrett’s esophagus. Together with another recent study, as well as studies of cost-effectiveness and patients’ quality of life, the results of our study suggest that the risk of esophageal adenocarcinoma among patients with Barrett’s esophagus is so minor that in the absence of dysplasia, routine surveillance of such patients is of doubtful value," the investigators said.
The relevance of such surveillance programs has been questioned before because they have never been shown to improve survival and because an estimated 95% of patients with a new diagnosis of esophageal adenocarcinoma do not have a previous diagnosis of Barrett’s esophagus, they noted.
Dr. Hvid-Jensen and his colleagues used data from Denmark’s nationwide pathology and cancer registries to calculate the incidence of adenocarcinoma among patients with Barrett’s esophagus and compare it with the expected incidence in the general population of 5.4 million people.
A total of 11,028 patients underwent endoscopic biopsy and received a diagnosis of Barrett’s esophagus during 1992-2009. The median age at baseline was 63 years, and patients were followed for a median of 5.2 years.
During that time, 197 of these patients with Barrett’s esophagus developed new esophageal adenocarcinomas, which comprised 7.6% of all the 2,602 incident esophageal adenocarcinomas diagnosed in the general Danish population during 1992-2009.
After excluding cancer cases that developed in the first year after a diagnosis of Barrett’s esophagus, the incidence of esophageal adenocarcinoma among patients with Barrett’s esophagus was found to be 1.2 cases per 1,000 person-years, the investigators said (N. Engl. J. Med. 2011;365:1375-83).
The annual risk of developing the malignancy was only 0.12%, or one case of adenocarcinoma per 860 patient-years.
In contrast, there were four reviews of the literature published in the past decade that pooled the results of numerous small studies conducted throughout the United States and Europe. These studies calculated the esophageal adenocarcinoma incidence as ranging from 5.2 to 7.0 cases per 1,000 person-years. In addition, two previous registry studies calculated similar incidences of 4.0 and 5.0 cases of esophogeal cardinoma per 1,000 person-years.
Current surveillance guidelines are based upon these earlier studies, which appear to have overstated the risks, Dr. Hvid-Jensen and his associates stated.
Their population-based, nationwide study is one of the largest studies of the issue; it included patients of all ages and both sexes and had almost no loss to follow-up. Because of Denmark’s universal health care plan, this study also had no referral bias or diagnostic bias.
"The generalizability of our results is therefore high," they noted.
Moreover, a recent population-based study in Northern Ireland found remarkably similar results: an incidence of 1.3 cases of esophageal adenocarcinoma per 1,000 patient-years among people with Barrett’s esophagus.
And another recent study "in which Markov models were used to evaluate available data on the incidence of adenocarcinoma supports our findings ... [and suggests] that surveillance is not beneficial," the researchers added.
This study was supported by the University of Aarhus Clinical Institute. No financial conflicts of interest were reported.☐
The "elegant" epidemiologic study by Dr. Hvid-Jensen and associates clearly shows that the relative risk of esophageal adenocarcinoma for a person with Barrett’s esophagus, as compared with the general population, is 11.3, "a substantial drop from the increase by a factor of 30 or 40 estimated in early reports," said Dr. Peter J. Kahrilas.
"As our knowledge of the biologic characteristics of Barrett’s esophagus has matured, the significance of the lesion has dwindled. In fact, patients with Barrett’s esophagus have the same life expectancy as does the general population, and esophageal cancer proves to be an uncommon cause of death in patients with Barrett’s esophagus regardless of surveillance," he noted.
Dr. Kahrilas is with the department of medicine at Northwestern University, Chicago. He reported ties to numerous industry sources. These remarks were taken from his editorial accompanying Dr. Hvid-Jensen’s report (N. Engl. J. Med. 2011;365:1437-8).
The "elegant" epidemiologic study by Dr. Hvid-Jensen and associates clearly shows that the relative risk of esophageal adenocarcinoma for a person with Barrett’s esophagus, as compared with the general population, is 11.3, "a substantial drop from the increase by a factor of 30 or 40 estimated in early reports," said Dr. Peter J. Kahrilas.
"As our knowledge of the biologic characteristics of Barrett’s esophagus has matured, the significance of the lesion has dwindled. In fact, patients with Barrett’s esophagus have the same life expectancy as does the general population, and esophageal cancer proves to be an uncommon cause of death in patients with Barrett’s esophagus regardless of surveillance," he noted.
Dr. Kahrilas is with the department of medicine at Northwestern University, Chicago. He reported ties to numerous industry sources. These remarks were taken from his editorial accompanying Dr. Hvid-Jensen’s report (N. Engl. J. Med. 2011;365:1437-8).
The "elegant" epidemiologic study by Dr. Hvid-Jensen and associates clearly shows that the relative risk of esophageal adenocarcinoma for a person with Barrett’s esophagus, as compared with the general population, is 11.3, "a substantial drop from the increase by a factor of 30 or 40 estimated in early reports," said Dr. Peter J. Kahrilas.
"As our knowledge of the biologic characteristics of Barrett’s esophagus has matured, the significance of the lesion has dwindled. In fact, patients with Barrett’s esophagus have the same life expectancy as does the general population, and esophageal cancer proves to be an uncommon cause of death in patients with Barrett’s esophagus regardless of surveillance," he noted.
Dr. Kahrilas is with the department of medicine at Northwestern University, Chicago. He reported ties to numerous industry sources. These remarks were taken from his editorial accompanying Dr. Hvid-Jensen’s report (N. Engl. J. Med. 2011;365:1437-8).
The incidence of esophageal adenocarcinoma among patients with Barrett’s esophagus was only 1.2 cases per 1,000 person-years in a study of the entire population of Denmark reported in the New England Journal of Medicine.
That rate is four to five times lower than rates reported previously, said Dr. Frederik Hvid-Jensen of the department of surgical gastroenterology at Aarhus (Denmark) University and his associates.
"Our study provides solid evidence that esophageal adenocarcinoma will develop in very few patients with Barrett’s esophagus. Together with another recent study, as well as studies of cost-effectiveness and patients’ quality of life, the results of our study suggest that the risk of esophageal adenocarcinoma among patients with Barrett’s esophagus is so minor that in the absence of dysplasia, routine surveillance of such patients is of doubtful value," the investigators said.
The relevance of such surveillance programs has been questioned before because they have never been shown to improve survival and because an estimated 95% of patients with a new diagnosis of esophageal adenocarcinoma do not have a previous diagnosis of Barrett’s esophagus, they noted.
Dr. Hvid-Jensen and his colleagues used data from Denmark’s nationwide pathology and cancer registries to calculate the incidence of adenocarcinoma among patients with Barrett’s esophagus and compare it with the expected incidence in the general population of 5.4 million people.
A total of 11,028 patients underwent endoscopic biopsy and received a diagnosis of Barrett’s esophagus during 1992-2009. The median age at baseline was 63 years, and patients were followed for a median of 5.2 years.
During that time, 197 of these patients with Barrett’s esophagus developed new esophageal adenocarcinomas, which comprised 7.6% of all the 2,602 incident esophageal adenocarcinomas diagnosed in the general Danish population during 1992-2009.
After excluding cancer cases that developed in the first year after a diagnosis of Barrett’s esophagus, the incidence of esophageal adenocarcinoma among patients with Barrett’s esophagus was found to be 1.2 cases per 1,000 person-years, the investigators said (N. Engl. J. Med. 2011;365:1375-83).
The annual risk of developing the malignancy was only 0.12%, or one case of adenocarcinoma per 860 patient-years.
In contrast, there were four reviews of the literature published in the past decade that pooled the results of numerous small studies conducted throughout the United States and Europe. These studies calculated the esophageal adenocarcinoma incidence as ranging from 5.2 to 7.0 cases per 1,000 person-years. In addition, two previous registry studies calculated similar incidences of 4.0 and 5.0 cases of esophogeal cardinoma per 1,000 person-years.
Current surveillance guidelines are based upon these earlier studies, which appear to have overstated the risks, Dr. Hvid-Jensen and his associates stated.
Their population-based, nationwide study is one of the largest studies of the issue; it included patients of all ages and both sexes and had almost no loss to follow-up. Because of Denmark’s universal health care plan, this study also had no referral bias or diagnostic bias.
"The generalizability of our results is therefore high," they noted.
Moreover, a recent population-based study in Northern Ireland found remarkably similar results: an incidence of 1.3 cases of esophageal adenocarcinoma per 1,000 patient-years among people with Barrett’s esophagus.
And another recent study "in which Markov models were used to evaluate available data on the incidence of adenocarcinoma supports our findings ... [and suggests] that surveillance is not beneficial," the researchers added.
This study was supported by the University of Aarhus Clinical Institute. No financial conflicts of interest were reported.☐
The incidence of esophageal adenocarcinoma among patients with Barrett’s esophagus was only 1.2 cases per 1,000 person-years in a study of the entire population of Denmark reported in the New England Journal of Medicine.
That rate is four to five times lower than rates reported previously, said Dr. Frederik Hvid-Jensen of the department of surgical gastroenterology at Aarhus (Denmark) University and his associates.
"Our study provides solid evidence that esophageal adenocarcinoma will develop in very few patients with Barrett’s esophagus. Together with another recent study, as well as studies of cost-effectiveness and patients’ quality of life, the results of our study suggest that the risk of esophageal adenocarcinoma among patients with Barrett’s esophagus is so minor that in the absence of dysplasia, routine surveillance of such patients is of doubtful value," the investigators said.
The relevance of such surveillance programs has been questioned before because they have never been shown to improve survival and because an estimated 95% of patients with a new diagnosis of esophageal adenocarcinoma do not have a previous diagnosis of Barrett’s esophagus, they noted.
Dr. Hvid-Jensen and his colleagues used data from Denmark’s nationwide pathology and cancer registries to calculate the incidence of adenocarcinoma among patients with Barrett’s esophagus and compare it with the expected incidence in the general population of 5.4 million people.
A total of 11,028 patients underwent endoscopic biopsy and received a diagnosis of Barrett’s esophagus during 1992-2009. The median age at baseline was 63 years, and patients were followed for a median of 5.2 years.
During that time, 197 of these patients with Barrett’s esophagus developed new esophageal adenocarcinomas, which comprised 7.6% of all the 2,602 incident esophageal adenocarcinomas diagnosed in the general Danish population during 1992-2009.
After excluding cancer cases that developed in the first year after a diagnosis of Barrett’s esophagus, the incidence of esophageal adenocarcinoma among patients with Barrett’s esophagus was found to be 1.2 cases per 1,000 person-years, the investigators said (N. Engl. J. Med. 2011;365:1375-83).
The annual risk of developing the malignancy was only 0.12%, or one case of adenocarcinoma per 860 patient-years.
In contrast, there were four reviews of the literature published in the past decade that pooled the results of numerous small studies conducted throughout the United States and Europe. These studies calculated the esophageal adenocarcinoma incidence as ranging from 5.2 to 7.0 cases per 1,000 person-years. In addition, two previous registry studies calculated similar incidences of 4.0 and 5.0 cases of esophogeal cardinoma per 1,000 person-years.
Current surveillance guidelines are based upon these earlier studies, which appear to have overstated the risks, Dr. Hvid-Jensen and his associates stated.
Their population-based, nationwide study is one of the largest studies of the issue; it included patients of all ages and both sexes and had almost no loss to follow-up. Because of Denmark’s universal health care plan, this study also had no referral bias or diagnostic bias.
"The generalizability of our results is therefore high," they noted.
Moreover, a recent population-based study in Northern Ireland found remarkably similar results: an incidence of 1.3 cases of esophageal adenocarcinoma per 1,000 patient-years among people with Barrett’s esophagus.
And another recent study "in which Markov models were used to evaluate available data on the incidence of adenocarcinoma supports our findings ... [and suggests] that surveillance is not beneficial," the researchers added.
This study was supported by the University of Aarhus Clinical Institute. No financial conflicts of interest were reported.☐
Major Finding: The incidence of esophageal adenocarcinoma among all patients in Denmark with Barrett’s esophagus was 1.2 cases per 1,000 person-years, which is four to five times lower than estimated in previous, smaller studies.
Data Source: An epidemiologic cohort study of esophageal adenocarcinoma among the 5.4 million residents of Denmark, including 11,028 with Barrett’s esophagus followed for a median of 5.2 years.
Disclosures: This study was supported by the University of Aarhus Clinical Institute. No financial conflicts of interest were reported.
New PCI, CABG Guidelines Support The Heart Team
The Heart Team is now officially the home team for selecting the best revascularization approach for patients with challenging coronary artery disease.
Two expert panels organized by the American College of Cardiology and American Heart Association each simultaneously released a revised set of guidelines – for Percutaneous Coronary Intervention (PCI) and for Coronary Artery Bypass Grafting (CABG). Both documents firmly recommended that physicians rely on Heart Teams to determine the best way to revascularize each patient who presents with either unprotected left main or "complex" coronary artery disease. The new revisions were also notable for the congruence of their recommendations, down to identical tables in both documents, and the collaboration between the two guideline-writing committees in coming up with their core revascularization sections (J. Am. Coll. Cardiol. 2011;58:doi:10.1016/j.jacc.2011.08.007; J. Am. Coll. Cardiol. 2011;58:doi:10.1016/j.jacc.2011.08.009).
"The 2011 guideline includes an unprecedented degree of collaboration [among cardiologists and cardiothoracic surgeons] in generating revascularization recommendations for patients with CAD [coronary artery disease]," said Dr. Glenn N. Levine, professor of medicine and director of the cardiac care unit at Baylor College of Medicine in Houston, and chairman of the PCI guidelines panel, in a written statement.
"It’s a breakthrough, the complete concordance of the revascularization sections" of the PCI and CABG guidelines, said Dr. Peter K. Smith, professor of surgery and chief of cardiovascular and thoracic surgery at Duke University in Durham, N.C., and vice-chairman of the CABG panel. The revascularization recommendations contained in both documents "were made with complete unanimity between the two groups," he said in an interview.
The new PCI guidelines also received endorsement from the Society for Cardiovascular Angiography and Interventions, while the new CABG recommendations carried imprimaturs from the Society of Thoracic Surgeons (STS), the American Association for Thoracic Surgery, and the Society of Cardiovascular Anesthesiologists.
The concept of relying on a Heart Team, a collaboration between at least one cardiologist and cardiac surgeon, to determine the best management strategy for a patient with coronary disease who could be managed by either an endovascular or surgical approach, first came to prominence in the mid-2000s during the SYNTAX (Synergy Between Percutaneous Coronary Intervention with Taxus and Cardiac Surgery) trial, the most recent large study to compare the safety and efficacy of PCI and CABG (N. Engl. J. Med. 2009;360:961-72). More recently, the PARTNER (Placement of Aortic Transcatheter Valves) trial for assessing the safety and efficacy of transcatheter aortic valve replacement, served as another high-profile setting for Heart Teams (N. Engl. J. Med. 2011;364:2187-98). The new guidelines for both PCI and CABG, which each gave the Heart Team approach a class 1 recommendation for managing patients with unprotected left main or complex CAD, represent the first time the Heart Team strategy received official endorsement from a health-policy setting group.
"The Heart Team concept evolved from these randomized trials, where patients could get either treatment. If that was how the trials led to their results, they are best replicated by using the same design," said Dr. Smith. When the guidelines refer to "complex" CAD, they mean triple vessel disease, as well as patients with two-vessel CAD that involves the proximal left anterior descending coronary artery, he noted. In such patients, as well as those with unprotected left main disease, the goal of revascularization is reduced mortality. Both sets of guidelines suggest assessing CAD complexity by calculating each patient’s SYNTAX score, a formula for quantifying CAD complexity originally developed for the SYNTAX trial. A score of 23 or higher defines more complex CAD, according to the new guidelines.
Results from "SYNTAX and other trials showed that [patients can] do as well with PCI or CABG for their longevity benefit," As a consequence, it is important for a cardiologist and surgeon to determine the suitability of each of these patients for the two options, Dr. Smith said.
The guidelines suggest physicians assess patients’ risk for surgery by quantifying their cardiac health and comorbidity severity by calculation of a STS score, as well as taking into account any other comorbidities not included in the STS score. "When the surgical risk is low, CABG is preferred even when PCI is possible, but if the surgery risk is high then patients should undergo PCI," he said. "Cardiac surgeons need to refer some patients with left main disease to PCI" because their clinical status makes them poor surgical candidates. "This is a big change [for cardiac surgeons], compared with 5 years ago," Dr. Smith said. "But only about 25% of left main patients fall into this category," where PCI is the better option.
While a Heart Team is important for deciding the best treatment for patients with life-threatening CAD, this subgroup probably represents about a quarter of all patients who need revascularization treatment, Dr. Smith said. The remaining three-quarters need revascularization for symptom relief, and while CABG also works well for this purpose, most patients in this category would also benefit from PCI, which may be the preferred choice, he added.
This new approach will probably not have a big impact on the volume of U.S. PCIs performed, predicted Dr. James C. Blankenship, vice-chairman of the PCI guidelines committee and an interventional cardiologist practicing in Danville, Pa. "On the complex end, I doubt that many PCIs today are done in super complex patients. On the low end, there has been a big shift in recognizing coronary lesions that need PCI," using tools that identify ischemia-causing lesions such as intravascular ultrasound and measuring fractional flow reserve. "Most interventionalists have shifted to this paradigm. For any who haven’t, the guidelines emphasize the importance of this approach," Dr. Blankenship said in an interview.
The call for Heart Teams by the new guidelines raises the issue whether enough U.S. teams exist to handle the volume of appropriate patients. Dr. Smith said there are, although they may not have a formal Heart Team designation.
"I think surgeons and cardiologists collaborate on this more than people think. They may not even realize they are doing it. To a large extent today it is not a formal process, but cardiologists and surgeons have multiple encounters with each other over their patients and they develop a sense of where to go, and when it is a close call [on how to manage a patient] they get together," Dr. Smith said
"It may not be a Heart Team as defined in the SYNTAX study. It can be any cardiac surgeon, and any interventional cardiologist," said Dr. Blankenship.
But, Dr. Smith noted, "the average cardiologist doesn’t calculate a SYNTAX score. We hope [the new guidelines] will lead to a resetting of the thought process."
And there are undoubtedly cardiologists today who do not consult with surgeons as often as they should, said Dr. Blankenship. "For many cardiologists it’s routine, but some cardiologists may be more aggressive about using PCI and less aggressive about getting surgical input." The new guidelines "set it forth as standard, and codify it by making the SYNTAX score a surrogate for disease complexity."
Dr. Levine said that he had no disclosures. Dr. Smith said that he has been a consultant to Eli Lilly and Baxter BioSurgery. Dr. Blankenship said that he has received research support from Abiomed, AstraZeneca, Boston Scientific, Conor Medsystems, Kai Pharmaceuticals, and Schering-Plough.☐
The Heart Team is now officially the home team for selecting the best revascularization approach for patients with challenging coronary artery disease.
Two expert panels organized by the American College of Cardiology and American Heart Association each simultaneously released a revised set of guidelines – for Percutaneous Coronary Intervention (PCI) and for Coronary Artery Bypass Grafting (CABG). Both documents firmly recommended that physicians rely on Heart Teams to determine the best way to revascularize each patient who presents with either unprotected left main or "complex" coronary artery disease. The new revisions were also notable for the congruence of their recommendations, down to identical tables in both documents, and the collaboration between the two guideline-writing committees in coming up with their core revascularization sections (J. Am. Coll. Cardiol. 2011;58:doi:10.1016/j.jacc.2011.08.007; J. Am. Coll. Cardiol. 2011;58:doi:10.1016/j.jacc.2011.08.009).
"The 2011 guideline includes an unprecedented degree of collaboration [among cardiologists and cardiothoracic surgeons] in generating revascularization recommendations for patients with CAD [coronary artery disease]," said Dr. Glenn N. Levine, professor of medicine and director of the cardiac care unit at Baylor College of Medicine in Houston, and chairman of the PCI guidelines panel, in a written statement.
"It’s a breakthrough, the complete concordance of the revascularization sections" of the PCI and CABG guidelines, said Dr. Peter K. Smith, professor of surgery and chief of cardiovascular and thoracic surgery at Duke University in Durham, N.C., and vice-chairman of the CABG panel. The revascularization recommendations contained in both documents "were made with complete unanimity between the two groups," he said in an interview.
The new PCI guidelines also received endorsement from the Society for Cardiovascular Angiography and Interventions, while the new CABG recommendations carried imprimaturs from the Society of Thoracic Surgeons (STS), the American Association for Thoracic Surgery, and the Society of Cardiovascular Anesthesiologists.
The concept of relying on a Heart Team, a collaboration between at least one cardiologist and cardiac surgeon, to determine the best management strategy for a patient with coronary disease who could be managed by either an endovascular or surgical approach, first came to prominence in the mid-2000s during the SYNTAX (Synergy Between Percutaneous Coronary Intervention with Taxus and Cardiac Surgery) trial, the most recent large study to compare the safety and efficacy of PCI and CABG (N. Engl. J. Med. 2009;360:961-72). More recently, the PARTNER (Placement of Aortic Transcatheter Valves) trial for assessing the safety and efficacy of transcatheter aortic valve replacement, served as another high-profile setting for Heart Teams (N. Engl. J. Med. 2011;364:2187-98). The new guidelines for both PCI and CABG, which each gave the Heart Team approach a class 1 recommendation for managing patients with unprotected left main or complex CAD, represent the first time the Heart Team strategy received official endorsement from a health-policy setting group.
"The Heart Team concept evolved from these randomized trials, where patients could get either treatment. If that was how the trials led to their results, they are best replicated by using the same design," said Dr. Smith. When the guidelines refer to "complex" CAD, they mean triple vessel disease, as well as patients with two-vessel CAD that involves the proximal left anterior descending coronary artery, he noted. In such patients, as well as those with unprotected left main disease, the goal of revascularization is reduced mortality. Both sets of guidelines suggest assessing CAD complexity by calculating each patient’s SYNTAX score, a formula for quantifying CAD complexity originally developed for the SYNTAX trial. A score of 23 or higher defines more complex CAD, according to the new guidelines.
Results from "SYNTAX and other trials showed that [patients can] do as well with PCI or CABG for their longevity benefit," As a consequence, it is important for a cardiologist and surgeon to determine the suitability of each of these patients for the two options, Dr. Smith said.
The guidelines suggest physicians assess patients’ risk for surgery by quantifying their cardiac health and comorbidity severity by calculation of a STS score, as well as taking into account any other comorbidities not included in the STS score. "When the surgical risk is low, CABG is preferred even when PCI is possible, but if the surgery risk is high then patients should undergo PCI," he said. "Cardiac surgeons need to refer some patients with left main disease to PCI" because their clinical status makes them poor surgical candidates. "This is a big change [for cardiac surgeons], compared with 5 years ago," Dr. Smith said. "But only about 25% of left main patients fall into this category," where PCI is the better option.
While a Heart Team is important for deciding the best treatment for patients with life-threatening CAD, this subgroup probably represents about a quarter of all patients who need revascularization treatment, Dr. Smith said. The remaining three-quarters need revascularization for symptom relief, and while CABG also works well for this purpose, most patients in this category would also benefit from PCI, which may be the preferred choice, he added.
This new approach will probably not have a big impact on the volume of U.S. PCIs performed, predicted Dr. James C. Blankenship, vice-chairman of the PCI guidelines committee and an interventional cardiologist practicing in Danville, Pa. "On the complex end, I doubt that many PCIs today are done in super complex patients. On the low end, there has been a big shift in recognizing coronary lesions that need PCI," using tools that identify ischemia-causing lesions such as intravascular ultrasound and measuring fractional flow reserve. "Most interventionalists have shifted to this paradigm. For any who haven’t, the guidelines emphasize the importance of this approach," Dr. Blankenship said in an interview.
The call for Heart Teams by the new guidelines raises the issue whether enough U.S. teams exist to handle the volume of appropriate patients. Dr. Smith said there are, although they may not have a formal Heart Team designation.
"I think surgeons and cardiologists collaborate on this more than people think. They may not even realize they are doing it. To a large extent today it is not a formal process, but cardiologists and surgeons have multiple encounters with each other over their patients and they develop a sense of where to go, and when it is a close call [on how to manage a patient] they get together," Dr. Smith said
"It may not be a Heart Team as defined in the SYNTAX study. It can be any cardiac surgeon, and any interventional cardiologist," said Dr. Blankenship.
But, Dr. Smith noted, "the average cardiologist doesn’t calculate a SYNTAX score. We hope [the new guidelines] will lead to a resetting of the thought process."
And there are undoubtedly cardiologists today who do not consult with surgeons as often as they should, said Dr. Blankenship. "For many cardiologists it’s routine, but some cardiologists may be more aggressive about using PCI and less aggressive about getting surgical input." The new guidelines "set it forth as standard, and codify it by making the SYNTAX score a surrogate for disease complexity."
Dr. Levine said that he had no disclosures. Dr. Smith said that he has been a consultant to Eli Lilly and Baxter BioSurgery. Dr. Blankenship said that he has received research support from Abiomed, AstraZeneca, Boston Scientific, Conor Medsystems, Kai Pharmaceuticals, and Schering-Plough.☐
The Heart Team is now officially the home team for selecting the best revascularization approach for patients with challenging coronary artery disease.
Two expert panels organized by the American College of Cardiology and American Heart Association each simultaneously released a revised set of guidelines – for Percutaneous Coronary Intervention (PCI) and for Coronary Artery Bypass Grafting (CABG). Both documents firmly recommended that physicians rely on Heart Teams to determine the best way to revascularize each patient who presents with either unprotected left main or "complex" coronary artery disease. The new revisions were also notable for the congruence of their recommendations, down to identical tables in both documents, and the collaboration between the two guideline-writing committees in coming up with their core revascularization sections (J. Am. Coll. Cardiol. 2011;58:doi:10.1016/j.jacc.2011.08.007; J. Am. Coll. Cardiol. 2011;58:doi:10.1016/j.jacc.2011.08.009).
"The 2011 guideline includes an unprecedented degree of collaboration [among cardiologists and cardiothoracic surgeons] in generating revascularization recommendations for patients with CAD [coronary artery disease]," said Dr. Glenn N. Levine, professor of medicine and director of the cardiac care unit at Baylor College of Medicine in Houston, and chairman of the PCI guidelines panel, in a written statement.
"It’s a breakthrough, the complete concordance of the revascularization sections" of the PCI and CABG guidelines, said Dr. Peter K. Smith, professor of surgery and chief of cardiovascular and thoracic surgery at Duke University in Durham, N.C., and vice-chairman of the CABG panel. The revascularization recommendations contained in both documents "were made with complete unanimity between the two groups," he said in an interview.
The new PCI guidelines also received endorsement from the Society for Cardiovascular Angiography and Interventions, while the new CABG recommendations carried imprimaturs from the Society of Thoracic Surgeons (STS), the American Association for Thoracic Surgery, and the Society of Cardiovascular Anesthesiologists.
The concept of relying on a Heart Team, a collaboration between at least one cardiologist and cardiac surgeon, to determine the best management strategy for a patient with coronary disease who could be managed by either an endovascular or surgical approach, first came to prominence in the mid-2000s during the SYNTAX (Synergy Between Percutaneous Coronary Intervention with Taxus and Cardiac Surgery) trial, the most recent large study to compare the safety and efficacy of PCI and CABG (N. Engl. J. Med. 2009;360:961-72). More recently, the PARTNER (Placement of Aortic Transcatheter Valves) trial for assessing the safety and efficacy of transcatheter aortic valve replacement, served as another high-profile setting for Heart Teams (N. Engl. J. Med. 2011;364:2187-98). The new guidelines for both PCI and CABG, which each gave the Heart Team approach a class 1 recommendation for managing patients with unprotected left main or complex CAD, represent the first time the Heart Team strategy received official endorsement from a health-policy setting group.
"The Heart Team concept evolved from these randomized trials, where patients could get either treatment. If that was how the trials led to their results, they are best replicated by using the same design," said Dr. Smith. When the guidelines refer to "complex" CAD, they mean triple vessel disease, as well as patients with two-vessel CAD that involves the proximal left anterior descending coronary artery, he noted. In such patients, as well as those with unprotected left main disease, the goal of revascularization is reduced mortality. Both sets of guidelines suggest assessing CAD complexity by calculating each patient’s SYNTAX score, a formula for quantifying CAD complexity originally developed for the SYNTAX trial. A score of 23 or higher defines more complex CAD, according to the new guidelines.
Results from "SYNTAX and other trials showed that [patients can] do as well with PCI or CABG for their longevity benefit," As a consequence, it is important for a cardiologist and surgeon to determine the suitability of each of these patients for the two options, Dr. Smith said.
The guidelines suggest physicians assess patients’ risk for surgery by quantifying their cardiac health and comorbidity severity by calculation of a STS score, as well as taking into account any other comorbidities not included in the STS score. "When the surgical risk is low, CABG is preferred even when PCI is possible, but if the surgery risk is high then patients should undergo PCI," he said. "Cardiac surgeons need to refer some patients with left main disease to PCI" because their clinical status makes them poor surgical candidates. "This is a big change [for cardiac surgeons], compared with 5 years ago," Dr. Smith said. "But only about 25% of left main patients fall into this category," where PCI is the better option.
While a Heart Team is important for deciding the best treatment for patients with life-threatening CAD, this subgroup probably represents about a quarter of all patients who need revascularization treatment, Dr. Smith said. The remaining three-quarters need revascularization for symptom relief, and while CABG also works well for this purpose, most patients in this category would also benefit from PCI, which may be the preferred choice, he added.
This new approach will probably not have a big impact on the volume of U.S. PCIs performed, predicted Dr. James C. Blankenship, vice-chairman of the PCI guidelines committee and an interventional cardiologist practicing in Danville, Pa. "On the complex end, I doubt that many PCIs today are done in super complex patients. On the low end, there has been a big shift in recognizing coronary lesions that need PCI," using tools that identify ischemia-causing lesions such as intravascular ultrasound and measuring fractional flow reserve. "Most interventionalists have shifted to this paradigm. For any who haven’t, the guidelines emphasize the importance of this approach," Dr. Blankenship said in an interview.
The call for Heart Teams by the new guidelines raises the issue whether enough U.S. teams exist to handle the volume of appropriate patients. Dr. Smith said there are, although they may not have a formal Heart Team designation.
"I think surgeons and cardiologists collaborate on this more than people think. They may not even realize they are doing it. To a large extent today it is not a formal process, but cardiologists and surgeons have multiple encounters with each other over their patients and they develop a sense of where to go, and when it is a close call [on how to manage a patient] they get together," Dr. Smith said
"It may not be a Heart Team as defined in the SYNTAX study. It can be any cardiac surgeon, and any interventional cardiologist," said Dr. Blankenship.
But, Dr. Smith noted, "the average cardiologist doesn’t calculate a SYNTAX score. We hope [the new guidelines] will lead to a resetting of the thought process."
And there are undoubtedly cardiologists today who do not consult with surgeons as often as they should, said Dr. Blankenship. "For many cardiologists it’s routine, but some cardiologists may be more aggressive about using PCI and less aggressive about getting surgical input." The new guidelines "set it forth as standard, and codify it by making the SYNTAX score a surrogate for disease complexity."
Dr. Levine said that he had no disclosures. Dr. Smith said that he has been a consultant to Eli Lilly and Baxter BioSurgery. Dr. Blankenship said that he has received research support from Abiomed, AstraZeneca, Boston Scientific, Conor Medsystems, Kai Pharmaceuticals, and Schering-Plough.☐
New PCI, CABG Guidelines Support The Heart Team
The Heart Team is now officially the home team for selecting the best revascularization approach for patients with challenging coronary artery disease.
Two expert panels organized by the American College of Cardiology and American Heart Association each simultaneously released a revised set of guidelines – for Percutaneous Coronary Intervention (PCI) and for Coronary Artery Bypass Grafting (CABG). Both documents firmly recommended that physicians rely on Heart Teams to determine the best way to revascularize each patient who presents with either unprotected left main or "complex" coronary artery disease. The new revisions were also notable for the congruence of their recommendations, down to identical tables in both documents, and the collaboration between the two guideline-writing committees in coming up with their core revascularization sections (J. Am. Coll. Cardiol. 2011;58:doi:10.1016/j.jacc.2011.08.007; J. Am. Coll. Cardiol. 2011;58:doi:10.1016/j.jacc.2011.08.009).
"The 2011 guideline includes an unprecedented degree of collaboration [among cardiologists and cardiothoracic surgeons] in generating revascularization recommendations for patients with CAD [coronary artery disease]," said Dr. Glenn N. Levine, professor of medicine and director of the cardiac care unit at Baylor College of Medicine in Houston, and chairman of the PCI guidelines panel, in a written statement.
"It’s a breakthrough, the complete concordance of the revascularization sections" of the PCI and CABG guidelines, said Dr. Peter K. Smith, professor of surgery and chief of cardiovascular and thoracic surgery at Duke University in Durham, N.C., and vice-chairman of the CABG panel. The revascularization recommendations contained in both documents "were made with complete unanimity between the two groups," he said in an interview.
The new PCI guidelines also received endorsement from the Society for Cardiovascular Angiography and Interventions, while the new CABG recommendations carried imprimaturs from the Society of Thoracic Surgeons (STS), the American Association for Thoracic Surgery, and the Society of Cardiovascular Anesthesiologists.
The concept of relying on a Heart Team, a collaboration between at least one cardiologist and cardiac surgeon, to determine the best management strategy for a patient with coronary disease who could be managed by either an endovascular or surgical approach, first came to prominence in the mid-2000s during the SYNTAX (Synergy Between Percutaneous Coronary Intervention with Taxus and Cardiac Surgery) trial, the most recent large study to compare the safety and efficacy of PCI and CABG (N. Engl. J. Med. 2009;360:961-72). More recently, the PARTNER (Placement of Aortic Transcatheter Valves) trial for assessing the safety and efficacy of transcatheter aortic valve replacement, served as another high-profile setting for Heart Teams (N. Engl. J. Med. 2011;364:2187-98). The new guidelines for both PCI and CABG, which each gave the Heart Team approach a class 1 recommendation for managing patients with unprotected left main or complex CAD, represent the first time the Heart Team strategy received official endorsement from a health-policy setting group.
"The Heart Team concept evolved from these randomized trials, where patients could get either treatment. If that was how the trials led to their results, they are best replicated by using the same design," said Dr. Smith. When the guidelines refer to "complex" CAD, they mean triple vessel disease, as well as patients with two-vessel CAD that involves the proximal left anterior descending coronary artery, he noted. In such patients, as well as those with unprotected left main disease, the goal of revascularization is reduced mortality. Both sets of guidelines suggest assessing CAD complexity by calculating each patient’s SYNTAX score, a formula for quantifying CAD complexity originally developed for the SYNTAX trial. A score of 23 or higher defines more complex CAD, according to the new guidelines.
Results from "SYNTAX and other trials showed that [patients can] do as well with PCI or CABG for their longevity benefit," As a consequence, it is important for a cardiologist and surgeon to determine the suitability of each of these patients for the two options, Dr. Smith said.
The guidelines suggest physicians assess patients’ risk for surgery by quantifying their cardiac health and comorbidity severity by calculation of a STS score, as well as taking into account any other comorbidities not included in the STS score. "When the surgical risk is low, CABG is preferred even when PCI is possible, but if the surgery risk is high then patients should undergo PCI," he said. "Cardiac surgeons need to refer some patients with left main disease to PCI" because their clinical status makes them poor surgical candidates. "This is a big change [for cardiac surgeons], compared with 5 years ago," Dr. Smith said. "But only about 25% of left main patients fall into this category," where PCI is the better option.
While a Heart Team is important for deciding the best treatment for patients with life-threatening CAD, this subgroup probably represents about a quarter of all patients who need revascularization treatment, Dr. Smith said. The remaining three-quarters need revascularization for symptom relief, and while CABG also works well for this purpose, most patients in this category would also benefit from PCI, which may be the preferred choice, he added.
This new approach will probably not have a big impact on the volume of U.S. PCIs performed, predicted Dr. James C. Blankenship, vice-chairman of the PCI guidelines committee and an interventional cardiologist practicing in Danville, Pa. "On the complex end, I doubt that many PCIs today are done in super complex patients. On the low end, there has been a big shift in recognizing coronary lesions that need PCI," using tools that identify ischemia-causing lesions such as intravascular ultrasound and measuring fractional flow reserve. "Most interventionalists have shifted to this paradigm. For any who haven’t, the guidelines emphasize the importance of this approach," Dr. Blankenship said in an interview.
The call for Heart Teams by the new guidelines raises the issue whether enough U.S. teams exist to handle the volume of appropriate patients. Dr. Smith said there are, although they may not have a formal Heart Team designation.
"I think surgeons and cardiologists collaborate on this more than people think. They may not even realize they are doing it. To a large extent today it is not a formal process, but cardiologists and surgeons have multiple encounters with each other over their patients and they develop a sense of where to go, and when it is a close call [on how to manage a patient] they get together," Dr. Smith said
"It may not be a Heart Team as defined in the SYNTAX study. It can be any cardiac surgeon, and any interventional cardiologist," said Dr. Blankenship.
But, Dr. Smith noted, "the average cardiologist doesn’t calculate a SYNTAX score. We hope [the new guidelines] will lead to a resetting of the thought process."
And there are undoubtedly cardiologists today who do not consult with surgeons as often as they should, said Dr. Blankenship. "For many cardiologists it’s routine, but some cardiologists may be more aggressive about using PCI and less aggressive about getting surgical input." The new guidelines "set it forth as standard, and codify it by making the SYNTAX score a surrogate for disease complexity."
Dr. Levine said that he had no disclosures. Dr. Smith said that he has been a consultant to Eli Lilly and Baxter BioSurgery. Dr. Blankenship said that he has received research support from Abiomed, AstraZeneca, Boston Scientific, Conor Medsystems, Kai Pharmaceuticals, and Schering-Plough.☐
The Heart Team is now officially the home team for selecting the best revascularization approach for patients with challenging coronary artery disease.
Two expert panels organized by the American College of Cardiology and American Heart Association each simultaneously released a revised set of guidelines – for Percutaneous Coronary Intervention (PCI) and for Coronary Artery Bypass Grafting (CABG). Both documents firmly recommended that physicians rely on Heart Teams to determine the best way to revascularize each patient who presents with either unprotected left main or "complex" coronary artery disease. The new revisions were also notable for the congruence of their recommendations, down to identical tables in both documents, and the collaboration between the two guideline-writing committees in coming up with their core revascularization sections (J. Am. Coll. Cardiol. 2011;58:doi:10.1016/j.jacc.2011.08.007; J. Am. Coll. Cardiol. 2011;58:doi:10.1016/j.jacc.2011.08.009).
"The 2011 guideline includes an unprecedented degree of collaboration [among cardiologists and cardiothoracic surgeons] in generating revascularization recommendations for patients with CAD [coronary artery disease]," said Dr. Glenn N. Levine, professor of medicine and director of the cardiac care unit at Baylor College of Medicine in Houston, and chairman of the PCI guidelines panel, in a written statement.
"It’s a breakthrough, the complete concordance of the revascularization sections" of the PCI and CABG guidelines, said Dr. Peter K. Smith, professor of surgery and chief of cardiovascular and thoracic surgery at Duke University in Durham, N.C., and vice-chairman of the CABG panel. The revascularization recommendations contained in both documents "were made with complete unanimity between the two groups," he said in an interview.
The new PCI guidelines also received endorsement from the Society for Cardiovascular Angiography and Interventions, while the new CABG recommendations carried imprimaturs from the Society of Thoracic Surgeons (STS), the American Association for Thoracic Surgery, and the Society of Cardiovascular Anesthesiologists.
The concept of relying on a Heart Team, a collaboration between at least one cardiologist and cardiac surgeon, to determine the best management strategy for a patient with coronary disease who could be managed by either an endovascular or surgical approach, first came to prominence in the mid-2000s during the SYNTAX (Synergy Between Percutaneous Coronary Intervention with Taxus and Cardiac Surgery) trial, the most recent large study to compare the safety and efficacy of PCI and CABG (N. Engl. J. Med. 2009;360:961-72). More recently, the PARTNER (Placement of Aortic Transcatheter Valves) trial for assessing the safety and efficacy of transcatheter aortic valve replacement, served as another high-profile setting for Heart Teams (N. Engl. J. Med. 2011;364:2187-98). The new guidelines for both PCI and CABG, which each gave the Heart Team approach a class 1 recommendation for managing patients with unprotected left main or complex CAD, represent the first time the Heart Team strategy received official endorsement from a health-policy setting group.
"The Heart Team concept evolved from these randomized trials, where patients could get either treatment. If that was how the trials led to their results, they are best replicated by using the same design," said Dr. Smith. When the guidelines refer to "complex" CAD, they mean triple vessel disease, as well as patients with two-vessel CAD that involves the proximal left anterior descending coronary artery, he noted. In such patients, as well as those with unprotected left main disease, the goal of revascularization is reduced mortality. Both sets of guidelines suggest assessing CAD complexity by calculating each patient’s SYNTAX score, a formula for quantifying CAD complexity originally developed for the SYNTAX trial. A score of 23 or higher defines more complex CAD, according to the new guidelines.
Results from "SYNTAX and other trials showed that [patients can] do as well with PCI or CABG for their longevity benefit," As a consequence, it is important for a cardiologist and surgeon to determine the suitability of each of these patients for the two options, Dr. Smith said.
The guidelines suggest physicians assess patients’ risk for surgery by quantifying their cardiac health and comorbidity severity by calculation of a STS score, as well as taking into account any other comorbidities not included in the STS score. "When the surgical risk is low, CABG is preferred even when PCI is possible, but if the surgery risk is high then patients should undergo PCI," he said. "Cardiac surgeons need to refer some patients with left main disease to PCI" because their clinical status makes them poor surgical candidates. "This is a big change [for cardiac surgeons], compared with 5 years ago," Dr. Smith said. "But only about 25% of left main patients fall into this category," where PCI is the better option.
While a Heart Team is important for deciding the best treatment for patients with life-threatening CAD, this subgroup probably represents about a quarter of all patients who need revascularization treatment, Dr. Smith said. The remaining three-quarters need revascularization for symptom relief, and while CABG also works well for this purpose, most patients in this category would also benefit from PCI, which may be the preferred choice, he added.
This new approach will probably not have a big impact on the volume of U.S. PCIs performed, predicted Dr. James C. Blankenship, vice-chairman of the PCI guidelines committee and an interventional cardiologist practicing in Danville, Pa. "On the complex end, I doubt that many PCIs today are done in super complex patients. On the low end, there has been a big shift in recognizing coronary lesions that need PCI," using tools that identify ischemia-causing lesions such as intravascular ultrasound and measuring fractional flow reserve. "Most interventionalists have shifted to this paradigm. For any who haven’t, the guidelines emphasize the importance of this approach," Dr. Blankenship said in an interview.
The call for Heart Teams by the new guidelines raises the issue whether enough U.S. teams exist to handle the volume of appropriate patients. Dr. Smith said there are, although they may not have a formal Heart Team designation.
"I think surgeons and cardiologists collaborate on this more than people think. They may not even realize they are doing it. To a large extent today it is not a formal process, but cardiologists and surgeons have multiple encounters with each other over their patients and they develop a sense of where to go, and when it is a close call [on how to manage a patient] they get together," Dr. Smith said
"It may not be a Heart Team as defined in the SYNTAX study. It can be any cardiac surgeon, and any interventional cardiologist," said Dr. Blankenship.
But, Dr. Smith noted, "the average cardiologist doesn’t calculate a SYNTAX score. We hope [the new guidelines] will lead to a resetting of the thought process."
And there are undoubtedly cardiologists today who do not consult with surgeons as often as they should, said Dr. Blankenship. "For many cardiologists it’s routine, but some cardiologists may be more aggressive about using PCI and less aggressive about getting surgical input." The new guidelines "set it forth as standard, and codify it by making the SYNTAX score a surrogate for disease complexity."
Dr. Levine said that he had no disclosures. Dr. Smith said that he has been a consultant to Eli Lilly and Baxter BioSurgery. Dr. Blankenship said that he has received research support from Abiomed, AstraZeneca, Boston Scientific, Conor Medsystems, Kai Pharmaceuticals, and Schering-Plough.☐
The Heart Team is now officially the home team for selecting the best revascularization approach for patients with challenging coronary artery disease.
Two expert panels organized by the American College of Cardiology and American Heart Association each simultaneously released a revised set of guidelines – for Percutaneous Coronary Intervention (PCI) and for Coronary Artery Bypass Grafting (CABG). Both documents firmly recommended that physicians rely on Heart Teams to determine the best way to revascularize each patient who presents with either unprotected left main or "complex" coronary artery disease. The new revisions were also notable for the congruence of their recommendations, down to identical tables in both documents, and the collaboration between the two guideline-writing committees in coming up with their core revascularization sections (J. Am. Coll. Cardiol. 2011;58:doi:10.1016/j.jacc.2011.08.007; J. Am. Coll. Cardiol. 2011;58:doi:10.1016/j.jacc.2011.08.009).
"The 2011 guideline includes an unprecedented degree of collaboration [among cardiologists and cardiothoracic surgeons] in generating revascularization recommendations for patients with CAD [coronary artery disease]," said Dr. Glenn N. Levine, professor of medicine and director of the cardiac care unit at Baylor College of Medicine in Houston, and chairman of the PCI guidelines panel, in a written statement.
"It’s a breakthrough, the complete concordance of the revascularization sections" of the PCI and CABG guidelines, said Dr. Peter K. Smith, professor of surgery and chief of cardiovascular and thoracic surgery at Duke University in Durham, N.C., and vice-chairman of the CABG panel. The revascularization recommendations contained in both documents "were made with complete unanimity between the two groups," he said in an interview.
The new PCI guidelines also received endorsement from the Society for Cardiovascular Angiography and Interventions, while the new CABG recommendations carried imprimaturs from the Society of Thoracic Surgeons (STS), the American Association for Thoracic Surgery, and the Society of Cardiovascular Anesthesiologists.
The concept of relying on a Heart Team, a collaboration between at least one cardiologist and cardiac surgeon, to determine the best management strategy for a patient with coronary disease who could be managed by either an endovascular or surgical approach, first came to prominence in the mid-2000s during the SYNTAX (Synergy Between Percutaneous Coronary Intervention with Taxus and Cardiac Surgery) trial, the most recent large study to compare the safety and efficacy of PCI and CABG (N. Engl. J. Med. 2009;360:961-72). More recently, the PARTNER (Placement of Aortic Transcatheter Valves) trial for assessing the safety and efficacy of transcatheter aortic valve replacement, served as another high-profile setting for Heart Teams (N. Engl. J. Med. 2011;364:2187-98). The new guidelines for both PCI and CABG, which each gave the Heart Team approach a class 1 recommendation for managing patients with unprotected left main or complex CAD, represent the first time the Heart Team strategy received official endorsement from a health-policy setting group.
"The Heart Team concept evolved from these randomized trials, where patients could get either treatment. If that was how the trials led to their results, they are best replicated by using the same design," said Dr. Smith. When the guidelines refer to "complex" CAD, they mean triple vessel disease, as well as patients with two-vessel CAD that involves the proximal left anterior descending coronary artery, he noted. In such patients, as well as those with unprotected left main disease, the goal of revascularization is reduced mortality. Both sets of guidelines suggest assessing CAD complexity by calculating each patient’s SYNTAX score, a formula for quantifying CAD complexity originally developed for the SYNTAX trial. A score of 23 or higher defines more complex CAD, according to the new guidelines.
Results from "SYNTAX and other trials showed that [patients can] do as well with PCI or CABG for their longevity benefit," As a consequence, it is important for a cardiologist and surgeon to determine the suitability of each of these patients for the two options, Dr. Smith said.
The guidelines suggest physicians assess patients’ risk for surgery by quantifying their cardiac health and comorbidity severity by calculation of a STS score, as well as taking into account any other comorbidities not included in the STS score. "When the surgical risk is low, CABG is preferred even when PCI is possible, but if the surgery risk is high then patients should undergo PCI," he said. "Cardiac surgeons need to refer some patients with left main disease to PCI" because their clinical status makes them poor surgical candidates. "This is a big change [for cardiac surgeons], compared with 5 years ago," Dr. Smith said. "But only about 25% of left main patients fall into this category," where PCI is the better option.
While a Heart Team is important for deciding the best treatment for patients with life-threatening CAD, this subgroup probably represents about a quarter of all patients who need revascularization treatment, Dr. Smith said. The remaining three-quarters need revascularization for symptom relief, and while CABG also works well for this purpose, most patients in this category would also benefit from PCI, which may be the preferred choice, he added.
This new approach will probably not have a big impact on the volume of U.S. PCIs performed, predicted Dr. James C. Blankenship, vice-chairman of the PCI guidelines committee and an interventional cardiologist practicing in Danville, Pa. "On the complex end, I doubt that many PCIs today are done in super complex patients. On the low end, there has been a big shift in recognizing coronary lesions that need PCI," using tools that identify ischemia-causing lesions such as intravascular ultrasound and measuring fractional flow reserve. "Most interventionalists have shifted to this paradigm. For any who haven’t, the guidelines emphasize the importance of this approach," Dr. Blankenship said in an interview.
The call for Heart Teams by the new guidelines raises the issue whether enough U.S. teams exist to handle the volume of appropriate patients. Dr. Smith said there are, although they may not have a formal Heart Team designation.
"I think surgeons and cardiologists collaborate on this more than people think. They may not even realize they are doing it. To a large extent today it is not a formal process, but cardiologists and surgeons have multiple encounters with each other over their patients and they develop a sense of where to go, and when it is a close call [on how to manage a patient] they get together," Dr. Smith said
"It may not be a Heart Team as defined in the SYNTAX study. It can be any cardiac surgeon, and any interventional cardiologist," said Dr. Blankenship.
But, Dr. Smith noted, "the average cardiologist doesn’t calculate a SYNTAX score. We hope [the new guidelines] will lead to a resetting of the thought process."
And there are undoubtedly cardiologists today who do not consult with surgeons as often as they should, said Dr. Blankenship. "For many cardiologists it’s routine, but some cardiologists may be more aggressive about using PCI and less aggressive about getting surgical input." The new guidelines "set it forth as standard, and codify it by making the SYNTAX score a surrogate for disease complexity."
Dr. Levine said that he had no disclosures. Dr. Smith said that he has been a consultant to Eli Lilly and Baxter BioSurgery. Dr. Blankenship said that he has received research support from Abiomed, AstraZeneca, Boston Scientific, Conor Medsystems, Kai Pharmaceuticals, and Schering-Plough.☐
New PCI, CABG Guidelines Support The Heart Team
The Heart Team is now officially the home team for selecting the best revascularization approach for patients with challenging coronary artery disease.
Two expert panels organized by the American College of Cardiology and American Heart Association each simultaneously released a revised set of guidelines – for Percutaneous Coronary Intervention (PCI) and for Coronary Artery Bypass Grafting (CABG). Both documents firmly recommended that physicians rely on Heart Teams to determine the best way to revascularize each patient who presents with either unprotected left main or "complex" coronary artery disease. The new revisions were also notable for the congruence of their recommendations, down to identical tables in both documents, and the collaboration between the two guideline-writing committees in coming up with their core revascularization sections (J. Am. Coll. Cardiol. 2011;58:doi:10.1016/j.jacc.2011.08.007; J. Am. Coll. Cardiol. 2011;58:doi:10.1016/j.jacc.2011.08.009).
"The 2011 guideline includes an unprecedented degree of collaboration [among cardiologists and cardiothoracic surgeons] in generating revascularization recommendations for patients with CAD [coronary artery disease]," said Dr. Glenn N. Levine, professor of medicine and director of the cardiac care unit at Baylor College of Medicine in Houston, and chairman of the PCI guidelines panel, in a written statement.
"It’s a breakthrough, the complete concordance of the revascularization sections" of the PCI and CABG guidelines, said Dr. Peter K. Smith, professor of surgery and chief of cardiovascular and thoracic surgery at Duke University in Durham, N.C., and vice-chairman of the CABG panel. The revascularization recommendations contained in both documents "were made with complete unanimity between the two groups," he said in an interview.
The new PCI guidelines also received endorsement from the Society for Cardiovascular Angiography and Interventions, while the new CABG recommendations carried imprimaturs from the Society of Thoracic Surgeons (STS), the American Association for Thoracic Surgery, and the Society of Cardiovascular Anesthesiologists.
The concept of relying on a Heart Team, a collaboration between at least one cardiologist and cardiac surgeon, to determine the best management strategy for a patient with coronary disease who could be managed by either an endovascular or surgical approach, first came to prominence in the mid-2000s during the SYNTAX (Synergy Between Percutaneous Coronary Intervention with Taxus and Cardiac Surgery) trial, the most recent large study to compare the safety and efficacy of PCI and CABG (N. Engl. J. Med. 2009;360:961-72). More recently, the PARTNER (Placement of Aortic Transcatheter Valves) trial for assessing the safety and efficacy of transcatheter aortic valve replacement, served as another high-profile setting for Heart Teams (N. Engl. J. Med. 2011;364:2187-98). The new guidelines for both PCI and CABG, which each gave the Heart Team approach a class 1 recommendation for managing patients with unprotected left main or complex CAD, represent the first time the Heart Team strategy received official endorsement from a health-policy setting group.
"The Heart Team concept evolved from these randomized trials, where patients could get either treatment. If that was how the trials led to their results, they are best replicated by using the same design," said Dr. Smith. When the guidelines refer to "complex" CAD, they mean triple vessel disease, as well as patients with two-vessel CAD that involves the proximal left anterior descending coronary artery, he noted. In such patients, as well as those with unprotected left main disease, the goal of revascularization is reduced mortality. Both sets of guidelines suggest assessing CAD complexity by calculating each patient’s SYNTAX score, a formula for quantifying CAD complexity originally developed for the SYNTAX trial. A score of 23 or higher defines more complex CAD, according to the new guidelines.
Results from "SYNTAX and other trials showed that [patients can] do as well with PCI or CABG for their longevity benefit," As a consequence, it is important for a cardiologist and surgeon to determine the suitability of each of these patients for the two options, Dr. Smith said.
The guidelines suggest physicians assess patients’ risk for surgery by quantifying their cardiac health and comorbidity severity by calculation of a STS score, as well as taking into account any other comorbidities not included in the STS score. "When the surgical risk is low, CABG is preferred even when PCI is possible, but if the surgery risk is high then patients should undergo PCI," he said. "Cardiac surgeons need to refer some patients with left main disease to PCI" because their clinical status makes them poor surgical candidates. "This is a big change [for cardiac surgeons], compared with 5 years ago," Dr. Smith said. "But only about 25% of left main patients fall into this category," where PCI is the better option.
While a Heart Team is important for deciding the best treatment for patients with life-threatening CAD, this subgroup probably represents about a quarter of all patients who need revascularization treatment, Dr. Smith said. The remaining three-quarters need revascularization for symptom relief, and while CABG also works well for this purpose, most patients in this category would also benefit from PCI, which may be the preferred choice, he added.
This new approach will probably not have a big impact on the volume of U.S. PCIs performed, predicted Dr. James C. Blankenship, vice-chairman of the PCI guidelines committee and an interventional cardiologist practicing in Danville, Pa. "On the complex end, I doubt that many PCIs today are done in super complex patients. On the low end, there has been a big shift in recognizing coronary lesions that need PCI," using tools that identify ischemia-causing lesions such as intravascular ultrasound and measuring fractional flow reserve. "Most interventionalists have shifted to this paradigm. For any who haven’t, the guidelines emphasize the importance of this approach," Dr. Blankenship said in an interview.
The call for Heart Teams by the new guidelines raises the issue whether enough U.S. teams exist to handle the volume of appropriate patients. Dr. Smith said there are, although they may not have a formal Heart Team designation.
"I think surgeons and cardiologists collaborate on this more than people think. They may not even realize they are doing it. To a large extent today it is not a formal process, but cardiologists and surgeons have multiple encounters with each other over their patients and they develop a sense of where to go, and when it is a close call [on how to manage a patient] they get together," Dr. Smith said
"It may not be a Heart Team as defined in the SYNTAX study. It can be any cardiac surgeon, and any interventional cardiologist," said Dr. Blankenship.
But, Dr. Smith noted, "the average cardiologist doesn’t calculate a SYNTAX score. We hope [the new guidelines] will lead to a resetting of the thought process."
And there are undoubtedly cardiologists today who do not consult with surgeons as often as they should, said Dr. Blankenship. "For many cardiologists it’s routine, but some cardiologists may be more aggressive about using PCI and less aggressive about getting surgical input." The new guidelines "set it forth as standard, and codify it by making the SYNTAX score a surrogate for disease complexity."
Dr. Levine said that he had no disclosures. Dr. Smith said that he has been a consultant to Eli Lilly and Baxter BioSurgery. Dr. Blankenship said that he has received research support from Abiomed, AstraZeneca, Boston Scientific, Conor Medsystems, Kai Pharmaceuticals, and Schering-Plough.
The Heart Team is now officially the home team for selecting the best revascularization approach for patients with challenging coronary artery disease.
Two expert panels organized by the American College of Cardiology and American Heart Association each simultaneously released a revised set of guidelines – for Percutaneous Coronary Intervention (PCI) and for Coronary Artery Bypass Grafting (CABG). Both documents firmly recommended that physicians rely on Heart Teams to determine the best way to revascularize each patient who presents with either unprotected left main or "complex" coronary artery disease. The new revisions were also notable for the congruence of their recommendations, down to identical tables in both documents, and the collaboration between the two guideline-writing committees in coming up with their core revascularization sections (J. Am. Coll. Cardiol. 2011;58:doi:10.1016/j.jacc.2011.08.007; J. Am. Coll. Cardiol. 2011;58:doi:10.1016/j.jacc.2011.08.009).
"The 2011 guideline includes an unprecedented degree of collaboration [among cardiologists and cardiothoracic surgeons] in generating revascularization recommendations for patients with CAD [coronary artery disease]," said Dr. Glenn N. Levine, professor of medicine and director of the cardiac care unit at Baylor College of Medicine in Houston, and chairman of the PCI guidelines panel, in a written statement.
"It’s a breakthrough, the complete concordance of the revascularization sections" of the PCI and CABG guidelines, said Dr. Peter K. Smith, professor of surgery and chief of cardiovascular and thoracic surgery at Duke University in Durham, N.C., and vice-chairman of the CABG panel. The revascularization recommendations contained in both documents "were made with complete unanimity between the two groups," he said in an interview.
The new PCI guidelines also received endorsement from the Society for Cardiovascular Angiography and Interventions, while the new CABG recommendations carried imprimaturs from the Society of Thoracic Surgeons (STS), the American Association for Thoracic Surgery, and the Society of Cardiovascular Anesthesiologists.
The concept of relying on a Heart Team, a collaboration between at least one cardiologist and cardiac surgeon, to determine the best management strategy for a patient with coronary disease who could be managed by either an endovascular or surgical approach, first came to prominence in the mid-2000s during the SYNTAX (Synergy Between Percutaneous Coronary Intervention with Taxus and Cardiac Surgery) trial, the most recent large study to compare the safety and efficacy of PCI and CABG (N. Engl. J. Med. 2009;360:961-72). More recently, the PARTNER (Placement of Aortic Transcatheter Valves) trial for assessing the safety and efficacy of transcatheter aortic valve replacement, served as another high-profile setting for Heart Teams (N. Engl. J. Med. 2011;364:2187-98). The new guidelines for both PCI and CABG, which each gave the Heart Team approach a class 1 recommendation for managing patients with unprotected left main or complex CAD, represent the first time the Heart Team strategy received official endorsement from a health-policy setting group.
"The Heart Team concept evolved from these randomized trials, where patients could get either treatment. If that was how the trials led to their results, they are best replicated by using the same design," said Dr. Smith. When the guidelines refer to "complex" CAD, they mean triple vessel disease, as well as patients with two-vessel CAD that involves the proximal left anterior descending coronary artery, he noted. In such patients, as well as those with unprotected left main disease, the goal of revascularization is reduced mortality. Both sets of guidelines suggest assessing CAD complexity by calculating each patient’s SYNTAX score, a formula for quantifying CAD complexity originally developed for the SYNTAX trial. A score of 23 or higher defines more complex CAD, according to the new guidelines.
Results from "SYNTAX and other trials showed that [patients can] do as well with PCI or CABG for their longevity benefit," As a consequence, it is important for a cardiologist and surgeon to determine the suitability of each of these patients for the two options, Dr. Smith said.
The guidelines suggest physicians assess patients’ risk for surgery by quantifying their cardiac health and comorbidity severity by calculation of a STS score, as well as taking into account any other comorbidities not included in the STS score. "When the surgical risk is low, CABG is preferred even when PCI is possible, but if the surgery risk is high then patients should undergo PCI," he said. "Cardiac surgeons need to refer some patients with left main disease to PCI" because their clinical status makes them poor surgical candidates. "This is a big change [for cardiac surgeons], compared with 5 years ago," Dr. Smith said. "But only about 25% of left main patients fall into this category," where PCI is the better option.
While a Heart Team is important for deciding the best treatment for patients with life-threatening CAD, this subgroup probably represents about a quarter of all patients who need revascularization treatment, Dr. Smith said. The remaining three-quarters need revascularization for symptom relief, and while CABG also works well for this purpose, most patients in this category would also benefit from PCI, which may be the preferred choice, he added.
This new approach will probably not have a big impact on the volume of U.S. PCIs performed, predicted Dr. James C. Blankenship, vice-chairman of the PCI guidelines committee and an interventional cardiologist practicing in Danville, Pa. "On the complex end, I doubt that many PCIs today are done in super complex patients. On the low end, there has been a big shift in recognizing coronary lesions that need PCI," using tools that identify ischemia-causing lesions such as intravascular ultrasound and measuring fractional flow reserve. "Most interventionalists have shifted to this paradigm. For any who haven’t, the guidelines emphasize the importance of this approach," Dr. Blankenship said in an interview.
The call for Heart Teams by the new guidelines raises the issue whether enough U.S. teams exist to handle the volume of appropriate patients. Dr. Smith said there are, although they may not have a formal Heart Team designation.
"I think surgeons and cardiologists collaborate on this more than people think. They may not even realize they are doing it. To a large extent today it is not a formal process, but cardiologists and surgeons have multiple encounters with each other over their patients and they develop a sense of where to go, and when it is a close call [on how to manage a patient] they get together," Dr. Smith said
"It may not be a Heart Team as defined in the SYNTAX study. It can be any cardiac surgeon, and any interventional cardiologist," said Dr. Blankenship.
But, Dr. Smith noted, "the average cardiologist doesn’t calculate a SYNTAX score. We hope [the new guidelines] will lead to a resetting of the thought process."
And there are undoubtedly cardiologists today who do not consult with surgeons as often as they should, said Dr. Blankenship. "For many cardiologists it’s routine, but some cardiologists may be more aggressive about using PCI and less aggressive about getting surgical input." The new guidelines "set it forth as standard, and codify it by making the SYNTAX score a surrogate for disease complexity."
Dr. Levine said that he had no disclosures. Dr. Smith said that he has been a consultant to Eli Lilly and Baxter BioSurgery. Dr. Blankenship said that he has received research support from Abiomed, AstraZeneca, Boston Scientific, Conor Medsystems, Kai Pharmaceuticals, and Schering-Plough.
The Heart Team is now officially the home team for selecting the best revascularization approach for patients with challenging coronary artery disease.
Two expert panels organized by the American College of Cardiology and American Heart Association each simultaneously released a revised set of guidelines – for Percutaneous Coronary Intervention (PCI) and for Coronary Artery Bypass Grafting (CABG). Both documents firmly recommended that physicians rely on Heart Teams to determine the best way to revascularize each patient who presents with either unprotected left main or "complex" coronary artery disease. The new revisions were also notable for the congruence of their recommendations, down to identical tables in both documents, and the collaboration between the two guideline-writing committees in coming up with their core revascularization sections (J. Am. Coll. Cardiol. 2011;58:doi:10.1016/j.jacc.2011.08.007; J. Am. Coll. Cardiol. 2011;58:doi:10.1016/j.jacc.2011.08.009).
"The 2011 guideline includes an unprecedented degree of collaboration [among cardiologists and cardiothoracic surgeons] in generating revascularization recommendations for patients with CAD [coronary artery disease]," said Dr. Glenn N. Levine, professor of medicine and director of the cardiac care unit at Baylor College of Medicine in Houston, and chairman of the PCI guidelines panel, in a written statement.
"It’s a breakthrough, the complete concordance of the revascularization sections" of the PCI and CABG guidelines, said Dr. Peter K. Smith, professor of surgery and chief of cardiovascular and thoracic surgery at Duke University in Durham, N.C., and vice-chairman of the CABG panel. The revascularization recommendations contained in both documents "were made with complete unanimity between the two groups," he said in an interview.
The new PCI guidelines also received endorsement from the Society for Cardiovascular Angiography and Interventions, while the new CABG recommendations carried imprimaturs from the Society of Thoracic Surgeons (STS), the American Association for Thoracic Surgery, and the Society of Cardiovascular Anesthesiologists.
The concept of relying on a Heart Team, a collaboration between at least one cardiologist and cardiac surgeon, to determine the best management strategy for a patient with coronary disease who could be managed by either an endovascular or surgical approach, first came to prominence in the mid-2000s during the SYNTAX (Synergy Between Percutaneous Coronary Intervention with Taxus and Cardiac Surgery) trial, the most recent large study to compare the safety and efficacy of PCI and CABG (N. Engl. J. Med. 2009;360:961-72). More recently, the PARTNER (Placement of Aortic Transcatheter Valves) trial for assessing the safety and efficacy of transcatheter aortic valve replacement, served as another high-profile setting for Heart Teams (N. Engl. J. Med. 2011;364:2187-98). The new guidelines for both PCI and CABG, which each gave the Heart Team approach a class 1 recommendation for managing patients with unprotected left main or complex CAD, represent the first time the Heart Team strategy received official endorsement from a health-policy setting group.
"The Heart Team concept evolved from these randomized trials, where patients could get either treatment. If that was how the trials led to their results, they are best replicated by using the same design," said Dr. Smith. When the guidelines refer to "complex" CAD, they mean triple vessel disease, as well as patients with two-vessel CAD that involves the proximal left anterior descending coronary artery, he noted. In such patients, as well as those with unprotected left main disease, the goal of revascularization is reduced mortality. Both sets of guidelines suggest assessing CAD complexity by calculating each patient’s SYNTAX score, a formula for quantifying CAD complexity originally developed for the SYNTAX trial. A score of 23 or higher defines more complex CAD, according to the new guidelines.
Results from "SYNTAX and other trials showed that [patients can] do as well with PCI or CABG for their longevity benefit," As a consequence, it is important for a cardiologist and surgeon to determine the suitability of each of these patients for the two options, Dr. Smith said.
The guidelines suggest physicians assess patients’ risk for surgery by quantifying their cardiac health and comorbidity severity by calculation of a STS score, as well as taking into account any other comorbidities not included in the STS score. "When the surgical risk is low, CABG is preferred even when PCI is possible, but if the surgery risk is high then patients should undergo PCI," he said. "Cardiac surgeons need to refer some patients with left main disease to PCI" because their clinical status makes them poor surgical candidates. "This is a big change [for cardiac surgeons], compared with 5 years ago," Dr. Smith said. "But only about 25% of left main patients fall into this category," where PCI is the better option.
While a Heart Team is important for deciding the best treatment for patients with life-threatening CAD, this subgroup probably represents about a quarter of all patients who need revascularization treatment, Dr. Smith said. The remaining three-quarters need revascularization for symptom relief, and while CABG also works well for this purpose, most patients in this category would also benefit from PCI, which may be the preferred choice, he added.
This new approach will probably not have a big impact on the volume of U.S. PCIs performed, predicted Dr. James C. Blankenship, vice-chairman of the PCI guidelines committee and an interventional cardiologist practicing in Danville, Pa. "On the complex end, I doubt that many PCIs today are done in super complex patients. On the low end, there has been a big shift in recognizing coronary lesions that need PCI," using tools that identify ischemia-causing lesions such as intravascular ultrasound and measuring fractional flow reserve. "Most interventionalists have shifted to this paradigm. For any who haven’t, the guidelines emphasize the importance of this approach," Dr. Blankenship said in an interview.
The call for Heart Teams by the new guidelines raises the issue whether enough U.S. teams exist to handle the volume of appropriate patients. Dr. Smith said there are, although they may not have a formal Heart Team designation.
"I think surgeons and cardiologists collaborate on this more than people think. They may not even realize they are doing it. To a large extent today it is not a formal process, but cardiologists and surgeons have multiple encounters with each other over their patients and they develop a sense of where to go, and when it is a close call [on how to manage a patient] they get together," Dr. Smith said
"It may not be a Heart Team as defined in the SYNTAX study. It can be any cardiac surgeon, and any interventional cardiologist," said Dr. Blankenship.
But, Dr. Smith noted, "the average cardiologist doesn’t calculate a SYNTAX score. We hope [the new guidelines] will lead to a resetting of the thought process."
And there are undoubtedly cardiologists today who do not consult with surgeons as often as they should, said Dr. Blankenship. "For many cardiologists it’s routine, but some cardiologists may be more aggressive about using PCI and less aggressive about getting surgical input." The new guidelines "set it forth as standard, and codify it by making the SYNTAX score a surrogate for disease complexity."
Dr. Levine said that he had no disclosures. Dr. Smith said that he has been a consultant to Eli Lilly and Baxter BioSurgery. Dr. Blankenship said that he has received research support from Abiomed, AstraZeneca, Boston Scientific, Conor Medsystems, Kai Pharmaceuticals, and Schering-Plough.
The Super Committee and the Budget
The future of federal spending on health care, medical research, and countless other programs will be determined by the actions this fall of a new Congressional committee. The Joint Select Committee on Deficit Reduction, also known as the "Super Committee," was created by the Budget Control Act of 2011 as part of the August 2011 compromise to raise the federal government’s borrowing limit (the "debt ceiling") and reduce the federal deficit. The Super Committee is charged with producing a plan by Nov. 23, 2011 to reduce the federal deficit by at least $1.2 trillion over 10 years. If the Super Committee does not produce a plan that achieves this much in savings, or if the committee’s plan is not passed by both houses of Congress by Dec. 23, 2011, automatic spending cuts (implemented through a process called "sequestration") will take effect over fiscal years (FY) 2013 through 2021. The Budget Control Act also set new limits on discretionary spending, including spending on medical research for FY 2012 and 2013.
Meet the Super Committee
The Super Committee is composed of 12 members of Congress, 3 from each party from each house. The members were chosen by the leaders of each party in each house. The committee roster is:
Rep. Jeb Hensarling (R-Tex.), Co-Chair
Sen. Patty Murray (D-Wash.), Co-Chair
Sen. Max Baucus (D-Mont.)
Rep. Xavier Becerra (D-Calif.)
Rep. Dave Camp (R-Mich.)
Rep. Jim Clyburn (D-S.C.)
Sen. John Kerry (D-Mass.)
Sen. Jon Kyl (R-Ariz.)
Sen. Rob Portman (R-Ohio)
Sen. Pat Toomey (R-Pa.)
Rep. Fred Upton (R-Mich.)
Rep. Chris Van Hollen (D-Md.)
The Super Committee began meeting on Sep. 8, 2011, and as of Oct. 13, it held two public hearings and many closed door sessions to discussion options for spending cuts. Interest groups from across the country, representing nearly every issue in the federal budget, are trying to influence the Super Committee’s decisions and help it set priorities.
Potential Automatic Spending Cuts For FY 2013-2021
With an even number of members from each party, the Super Committee might not be able to reach an agreement, particularly when there are sharp partisan divisions about whether deficit reduction should include increased revenues as well as spending cuts. If the Super Committee succeeds at producing a bill, the legislation must be approved by both houses of Congress. No amendments or filibusters (prolonged debate in the Senate to avoid a vote) of the bill would be permitted. If Congress does not pass legislation to achieve the required level of deficit reduction by Dec. 23, 2011, automatic spending cuts will take effect in FY 2013-2012 through the sequestration process. Half of the automatic cuts would come from defense spending, and the other half would come from the rest of the federal budget, excluding Social Security and other retirement programs, Medicaid, and certain safety-net programs for people with low-incomes. Cuts to Medicare would be limited to 2% of payments to provider and Medicare Advantage plans; beneficiary costs would not increase. The cuts would be applied evenly across Medicare, but physician payments would be reduced by more than 2% if the physician update formula is not repealed.
Healthcare Options on the Table
One of the difficult issues the Super Committee faces is how to achieve reductions in Medicare spending while also preventing another payment cut under the physician fee schedule. Repealing the current physician payment update formula, which has produced payment reductions of 20%-30% before being reversed by Congress, is estimated to cost $300 billion over ten years. The Medicare Payment Advisory Commission (MedPAC) developed a proposal to replace the current update formula with a new approach that would freeze payments to primary care physicians and reduce payments to other physicians. Additional savings could come from other parts of the Medicare program. For example, payments to hospitals for bad debt and graduate medical education could be reduced, payments to physicians for imaging services could be cut, and beneficiary cost-sharing could be required for laboratory and home health services. Any or all of these options could be on the table for the Super Committee to consider. Medicare is a large part of the health spending at issue, but all other federal spending on health issues also is up for debate by the Super Committee. As the committee looks for potential deficit reduction options, it may consider spending on medical research, Medicaid, and other health-related expenses, as well.
Opportunities for Medical Research Spending in FY 2012 and Beyond
In addition to creating the Super Committee, the Budget Control Act set limits on discretionary spending – federal spending excluding Social Security, Medicare, and Medicaid and the wars in Iraq and Afghanistan. The spending cap for 2012 ($1.043 trillion) is $7 billion less than the total approved discretionary spending for FY 2011 ($1.050 trillion), but $24 billion more than the House of Representatives approved in its budget for FY 2012. The spending cap for FY 2013 is only $4 billion more than the FY 2012 cap, but beginning in 2014, the annual cap will increase by $20 billion to $25 billion per year. These increases in the spending cap could allow for increases in appropriations for the National Institutes of Health and other federal research programs.
What Happens Next?
The Super Committee is being watched closely for signs that it might be able to reach an agreement, as the President, other members of Congress, and interest groups prepare proposals for its consideration. Sequestration may be the most likely outcome of this process. The only certainty is that we should know by Nov. 23 whether the Super Committee will produce a plan, and we will know by Dec. 23 whether Congress will pass that plan.
Elizabeth Halpern, J.D., writes on medico-legal issues for the AATS and is an attorney at Hogan Lovells.
The future of federal spending on health care, medical research, and countless other programs will be determined by the actions this fall of a new Congressional committee. The Joint Select Committee on Deficit Reduction, also known as the "Super Committee," was created by the Budget Control Act of 2011 as part of the August 2011 compromise to raise the federal government’s borrowing limit (the "debt ceiling") and reduce the federal deficit. The Super Committee is charged with producing a plan by Nov. 23, 2011 to reduce the federal deficit by at least $1.2 trillion over 10 years. If the Super Committee does not produce a plan that achieves this much in savings, or if the committee’s plan is not passed by both houses of Congress by Dec. 23, 2011, automatic spending cuts (implemented through a process called "sequestration") will take effect over fiscal years (FY) 2013 through 2021. The Budget Control Act also set new limits on discretionary spending, including spending on medical research for FY 2012 and 2013.
Meet the Super Committee
The Super Committee is composed of 12 members of Congress, 3 from each party from each house. The members were chosen by the leaders of each party in each house. The committee roster is:
Rep. Jeb Hensarling (R-Tex.), Co-Chair
Sen. Patty Murray (D-Wash.), Co-Chair
Sen. Max Baucus (D-Mont.)
Rep. Xavier Becerra (D-Calif.)
Rep. Dave Camp (R-Mich.)
Rep. Jim Clyburn (D-S.C.)
Sen. John Kerry (D-Mass.)
Sen. Jon Kyl (R-Ariz.)
Sen. Rob Portman (R-Ohio)
Sen. Pat Toomey (R-Pa.)
Rep. Fred Upton (R-Mich.)
Rep. Chris Van Hollen (D-Md.)
The Super Committee began meeting on Sep. 8, 2011, and as of Oct. 13, it held two public hearings and many closed door sessions to discussion options for spending cuts. Interest groups from across the country, representing nearly every issue in the federal budget, are trying to influence the Super Committee’s decisions and help it set priorities.
Potential Automatic Spending Cuts For FY 2013-2021
With an even number of members from each party, the Super Committee might not be able to reach an agreement, particularly when there are sharp partisan divisions about whether deficit reduction should include increased revenues as well as spending cuts. If the Super Committee succeeds at producing a bill, the legislation must be approved by both houses of Congress. No amendments or filibusters (prolonged debate in the Senate to avoid a vote) of the bill would be permitted. If Congress does not pass legislation to achieve the required level of deficit reduction by Dec. 23, 2011, automatic spending cuts will take effect in FY 2013-2012 through the sequestration process. Half of the automatic cuts would come from defense spending, and the other half would come from the rest of the federal budget, excluding Social Security and other retirement programs, Medicaid, and certain safety-net programs for people with low-incomes. Cuts to Medicare would be limited to 2% of payments to provider and Medicare Advantage plans; beneficiary costs would not increase. The cuts would be applied evenly across Medicare, but physician payments would be reduced by more than 2% if the physician update formula is not repealed.
Healthcare Options on the Table
One of the difficult issues the Super Committee faces is how to achieve reductions in Medicare spending while also preventing another payment cut under the physician fee schedule. Repealing the current physician payment update formula, which has produced payment reductions of 20%-30% before being reversed by Congress, is estimated to cost $300 billion over ten years. The Medicare Payment Advisory Commission (MedPAC) developed a proposal to replace the current update formula with a new approach that would freeze payments to primary care physicians and reduce payments to other physicians. Additional savings could come from other parts of the Medicare program. For example, payments to hospitals for bad debt and graduate medical education could be reduced, payments to physicians for imaging services could be cut, and beneficiary cost-sharing could be required for laboratory and home health services. Any or all of these options could be on the table for the Super Committee to consider. Medicare is a large part of the health spending at issue, but all other federal spending on health issues also is up for debate by the Super Committee. As the committee looks for potential deficit reduction options, it may consider spending on medical research, Medicaid, and other health-related expenses, as well.
Opportunities for Medical Research Spending in FY 2012 and Beyond
In addition to creating the Super Committee, the Budget Control Act set limits on discretionary spending – federal spending excluding Social Security, Medicare, and Medicaid and the wars in Iraq and Afghanistan. The spending cap for 2012 ($1.043 trillion) is $7 billion less than the total approved discretionary spending for FY 2011 ($1.050 trillion), but $24 billion more than the House of Representatives approved in its budget for FY 2012. The spending cap for FY 2013 is only $4 billion more than the FY 2012 cap, but beginning in 2014, the annual cap will increase by $20 billion to $25 billion per year. These increases in the spending cap could allow for increases in appropriations for the National Institutes of Health and other federal research programs.
What Happens Next?
The Super Committee is being watched closely for signs that it might be able to reach an agreement, as the President, other members of Congress, and interest groups prepare proposals for its consideration. Sequestration may be the most likely outcome of this process. The only certainty is that we should know by Nov. 23 whether the Super Committee will produce a plan, and we will know by Dec. 23 whether Congress will pass that plan.
Elizabeth Halpern, J.D., writes on medico-legal issues for the AATS and is an attorney at Hogan Lovells.
The future of federal spending on health care, medical research, and countless other programs will be determined by the actions this fall of a new Congressional committee. The Joint Select Committee on Deficit Reduction, also known as the "Super Committee," was created by the Budget Control Act of 2011 as part of the August 2011 compromise to raise the federal government’s borrowing limit (the "debt ceiling") and reduce the federal deficit. The Super Committee is charged with producing a plan by Nov. 23, 2011 to reduce the federal deficit by at least $1.2 trillion over 10 years. If the Super Committee does not produce a plan that achieves this much in savings, or if the committee’s plan is not passed by both houses of Congress by Dec. 23, 2011, automatic spending cuts (implemented through a process called "sequestration") will take effect over fiscal years (FY) 2013 through 2021. The Budget Control Act also set new limits on discretionary spending, including spending on medical research for FY 2012 and 2013.
Meet the Super Committee
The Super Committee is composed of 12 members of Congress, 3 from each party from each house. The members were chosen by the leaders of each party in each house. The committee roster is:
Rep. Jeb Hensarling (R-Tex.), Co-Chair
Sen. Patty Murray (D-Wash.), Co-Chair
Sen. Max Baucus (D-Mont.)
Rep. Xavier Becerra (D-Calif.)
Rep. Dave Camp (R-Mich.)
Rep. Jim Clyburn (D-S.C.)
Sen. John Kerry (D-Mass.)
Sen. Jon Kyl (R-Ariz.)
Sen. Rob Portman (R-Ohio)
Sen. Pat Toomey (R-Pa.)
Rep. Fred Upton (R-Mich.)
Rep. Chris Van Hollen (D-Md.)
The Super Committee began meeting on Sep. 8, 2011, and as of Oct. 13, it held two public hearings and many closed door sessions to discussion options for spending cuts. Interest groups from across the country, representing nearly every issue in the federal budget, are trying to influence the Super Committee’s decisions and help it set priorities.
Potential Automatic Spending Cuts For FY 2013-2021
With an even number of members from each party, the Super Committee might not be able to reach an agreement, particularly when there are sharp partisan divisions about whether deficit reduction should include increased revenues as well as spending cuts. If the Super Committee succeeds at producing a bill, the legislation must be approved by both houses of Congress. No amendments or filibusters (prolonged debate in the Senate to avoid a vote) of the bill would be permitted. If Congress does not pass legislation to achieve the required level of deficit reduction by Dec. 23, 2011, automatic spending cuts will take effect in FY 2013-2012 through the sequestration process. Half of the automatic cuts would come from defense spending, and the other half would come from the rest of the federal budget, excluding Social Security and other retirement programs, Medicaid, and certain safety-net programs for people with low-incomes. Cuts to Medicare would be limited to 2% of payments to provider and Medicare Advantage plans; beneficiary costs would not increase. The cuts would be applied evenly across Medicare, but physician payments would be reduced by more than 2% if the physician update formula is not repealed.
Healthcare Options on the Table
One of the difficult issues the Super Committee faces is how to achieve reductions in Medicare spending while also preventing another payment cut under the physician fee schedule. Repealing the current physician payment update formula, which has produced payment reductions of 20%-30% before being reversed by Congress, is estimated to cost $300 billion over ten years. The Medicare Payment Advisory Commission (MedPAC) developed a proposal to replace the current update formula with a new approach that would freeze payments to primary care physicians and reduce payments to other physicians. Additional savings could come from other parts of the Medicare program. For example, payments to hospitals for bad debt and graduate medical education could be reduced, payments to physicians for imaging services could be cut, and beneficiary cost-sharing could be required for laboratory and home health services. Any or all of these options could be on the table for the Super Committee to consider. Medicare is a large part of the health spending at issue, but all other federal spending on health issues also is up for debate by the Super Committee. As the committee looks for potential deficit reduction options, it may consider spending on medical research, Medicaid, and other health-related expenses, as well.
Opportunities for Medical Research Spending in FY 2012 and Beyond
In addition to creating the Super Committee, the Budget Control Act set limits on discretionary spending – federal spending excluding Social Security, Medicare, and Medicaid and the wars in Iraq and Afghanistan. The spending cap for 2012 ($1.043 trillion) is $7 billion less than the total approved discretionary spending for FY 2011 ($1.050 trillion), but $24 billion more than the House of Representatives approved in its budget for FY 2012. The spending cap for FY 2013 is only $4 billion more than the FY 2012 cap, but beginning in 2014, the annual cap will increase by $20 billion to $25 billion per year. These increases in the spending cap could allow for increases in appropriations for the National Institutes of Health and other federal research programs.
What Happens Next?
The Super Committee is being watched closely for signs that it might be able to reach an agreement, as the President, other members of Congress, and interest groups prepare proposals for its consideration. Sequestration may be the most likely outcome of this process. The only certainty is that we should know by Nov. 23 whether the Super Committee will produce a plan, and we will know by Dec. 23 whether Congress will pass that plan.
Elizabeth Halpern, J.D., writes on medico-legal issues for the AATS and is an attorney at Hogan Lovells.
Blood Conservation Program Successful
COLORADO SPRINGS – Implementation of a comprehensive blood conservation algorithm in a community hospital cardiac surgery program led to a 41% reduction in total blood product usage with no adverse impact on safety, a study has shown.
The blood conservation strategy included lowering the postoperative hemoglobin threshold for transfusion to less than 7 g/dL, utilizing a miniature low prime perfusion circuit in patients on cardiopulmonary bypass, intraoperative point-of-care testing to avoid inappropriate RBC and component transfusion, and routine blood withdrawal and storage before bypass and transfusion after protamine administration, Dr. Steve Xydas explained at the annual meeting of the Western Thoracic Surgical Association.
Blood transfusions in patients undergoing cardiac surgery use 15%-20% of the nation’s blood supply.
At present the indications for transfusion aren’t standardized, and there is wide disparity in transfusion rates among cardiac surgery patients, noted Dr. Xydas of Morristown (N.J.) Medical Center (formerly Morristown Memorial Hospital).
For these reasons, he and the other three cardiac surgeons at the hospital decided to push for implementation of a comprehensive blood conservation program.
They prospectively collected data on 481 patients who underwent cardiac surgery during the 6 months prior to introduction of the program. Then, following a 3-month introductory program implementation period, they collected data for 6 months on the 557 patients whose surgery was performed under the new blood transfusion strategy.
Fifty-seven percent of the 1,038 patients underwent isolated coronary artery bypass grafting, 25% had isolated valve surgery, and 18% had both.
Total blood product usage (defined as the sum of blood, platelets, cryoprecipitate or fresh frozen plasma units) dropped from an average of 5.8 U per patient during the baseline period to 3.5 U per patient following introduction of the program.
Overall 30-day mortality was 1.3%, with no significant difference between the two groups. Nor were there differences in any major morbidity end points, including stroke, reoperation for bleeding, new-onset atrial fibrillation, acute MI, renal failure, or sternal wound infection. Length of stay was similar in the two groups as well.
Discussant Dr. James M. Maxwell of the International Heart Institute of Montana in Missoula observed that so much evidence has accumulated regarding the multiple harmful effects of transfusion that the current thrust is a search for meaningful evidence to support transfusion in the absence of life-threatening hemorrhage.
Because of transfusion’s harmful effects, he’d have expected to see significantly better outcomes in the group operated on under the blood conservation strategy. Dr. Maxwell attributed the lack of a significant difference in outcome between the two groups to the excellent surgical results the Morristown group obtained even before introducing the strategy.
In order to overcome the natural tendency to slide back into old habits, Dr. Xydas said he and his surgical colleagues make a point of sharing their updated results on a quarterly basis with staff cardiologists, pulmonologists, intensivists, and nurses.
Were there any barriers to implementation of the blood conservation program? he was asked. Dr. Xydas replied that this was a cardiac surgeon–led project; he and his surgical colleagues laid the groundwork by leading grand rounds for nonsurgeons in order to convince them the program was a good idea. There was occasional early resistance based on a physician’s anecdotal experience – for example, having encountered a single case of ischemic optic neuritis several decades earlier – but such reservations were easily overcome by presenting hard data on the harmful effects of transfusion, he said.
Dr. Xydas said he had no relevant financial disclosures.
COLORADO SPRINGS – Implementation of a comprehensive blood conservation algorithm in a community hospital cardiac surgery program led to a 41% reduction in total blood product usage with no adverse impact on safety, a study has shown.
The blood conservation strategy included lowering the postoperative hemoglobin threshold for transfusion to less than 7 g/dL, utilizing a miniature low prime perfusion circuit in patients on cardiopulmonary bypass, intraoperative point-of-care testing to avoid inappropriate RBC and component transfusion, and routine blood withdrawal and storage before bypass and transfusion after protamine administration, Dr. Steve Xydas explained at the annual meeting of the Western Thoracic Surgical Association.
Blood transfusions in patients undergoing cardiac surgery use 15%-20% of the nation’s blood supply.
At present the indications for transfusion aren’t standardized, and there is wide disparity in transfusion rates among cardiac surgery patients, noted Dr. Xydas of Morristown (N.J.) Medical Center (formerly Morristown Memorial Hospital).
For these reasons, he and the other three cardiac surgeons at the hospital decided to push for implementation of a comprehensive blood conservation program.
They prospectively collected data on 481 patients who underwent cardiac surgery during the 6 months prior to introduction of the program. Then, following a 3-month introductory program implementation period, they collected data for 6 months on the 557 patients whose surgery was performed under the new blood transfusion strategy.
Fifty-seven percent of the 1,038 patients underwent isolated coronary artery bypass grafting, 25% had isolated valve surgery, and 18% had both.
Total blood product usage (defined as the sum of blood, platelets, cryoprecipitate or fresh frozen plasma units) dropped from an average of 5.8 U per patient during the baseline period to 3.5 U per patient following introduction of the program.
Overall 30-day mortality was 1.3%, with no significant difference between the two groups. Nor were there differences in any major morbidity end points, including stroke, reoperation for bleeding, new-onset atrial fibrillation, acute MI, renal failure, or sternal wound infection. Length of stay was similar in the two groups as well.
Discussant Dr. James M. Maxwell of the International Heart Institute of Montana in Missoula observed that so much evidence has accumulated regarding the multiple harmful effects of transfusion that the current thrust is a search for meaningful evidence to support transfusion in the absence of life-threatening hemorrhage.
Because of transfusion’s harmful effects, he’d have expected to see significantly better outcomes in the group operated on under the blood conservation strategy. Dr. Maxwell attributed the lack of a significant difference in outcome between the two groups to the excellent surgical results the Morristown group obtained even before introducing the strategy.
In order to overcome the natural tendency to slide back into old habits, Dr. Xydas said he and his surgical colleagues make a point of sharing their updated results on a quarterly basis with staff cardiologists, pulmonologists, intensivists, and nurses.
Were there any barriers to implementation of the blood conservation program? he was asked. Dr. Xydas replied that this was a cardiac surgeon–led project; he and his surgical colleagues laid the groundwork by leading grand rounds for nonsurgeons in order to convince them the program was a good idea. There was occasional early resistance based on a physician’s anecdotal experience – for example, having encountered a single case of ischemic optic neuritis several decades earlier – but such reservations were easily overcome by presenting hard data on the harmful effects of transfusion, he said.
Dr. Xydas said he had no relevant financial disclosures.
COLORADO SPRINGS – Implementation of a comprehensive blood conservation algorithm in a community hospital cardiac surgery program led to a 41% reduction in total blood product usage with no adverse impact on safety, a study has shown.
The blood conservation strategy included lowering the postoperative hemoglobin threshold for transfusion to less than 7 g/dL, utilizing a miniature low prime perfusion circuit in patients on cardiopulmonary bypass, intraoperative point-of-care testing to avoid inappropriate RBC and component transfusion, and routine blood withdrawal and storage before bypass and transfusion after protamine administration, Dr. Steve Xydas explained at the annual meeting of the Western Thoracic Surgical Association.
Blood transfusions in patients undergoing cardiac surgery use 15%-20% of the nation’s blood supply.
At present the indications for transfusion aren’t standardized, and there is wide disparity in transfusion rates among cardiac surgery patients, noted Dr. Xydas of Morristown (N.J.) Medical Center (formerly Morristown Memorial Hospital).
For these reasons, he and the other three cardiac surgeons at the hospital decided to push for implementation of a comprehensive blood conservation program.
They prospectively collected data on 481 patients who underwent cardiac surgery during the 6 months prior to introduction of the program. Then, following a 3-month introductory program implementation period, they collected data for 6 months on the 557 patients whose surgery was performed under the new blood transfusion strategy.
Fifty-seven percent of the 1,038 patients underwent isolated coronary artery bypass grafting, 25% had isolated valve surgery, and 18% had both.
Total blood product usage (defined as the sum of blood, platelets, cryoprecipitate or fresh frozen plasma units) dropped from an average of 5.8 U per patient during the baseline period to 3.5 U per patient following introduction of the program.
Overall 30-day mortality was 1.3%, with no significant difference between the two groups. Nor were there differences in any major morbidity end points, including stroke, reoperation for bleeding, new-onset atrial fibrillation, acute MI, renal failure, or sternal wound infection. Length of stay was similar in the two groups as well.
Discussant Dr. James M. Maxwell of the International Heart Institute of Montana in Missoula observed that so much evidence has accumulated regarding the multiple harmful effects of transfusion that the current thrust is a search for meaningful evidence to support transfusion in the absence of life-threatening hemorrhage.
Because of transfusion’s harmful effects, he’d have expected to see significantly better outcomes in the group operated on under the blood conservation strategy. Dr. Maxwell attributed the lack of a significant difference in outcome between the two groups to the excellent surgical results the Morristown group obtained even before introducing the strategy.
In order to overcome the natural tendency to slide back into old habits, Dr. Xydas said he and his surgical colleagues make a point of sharing their updated results on a quarterly basis with staff cardiologists, pulmonologists, intensivists, and nurses.
Were there any barriers to implementation of the blood conservation program? he was asked. Dr. Xydas replied that this was a cardiac surgeon–led project; he and his surgical colleagues laid the groundwork by leading grand rounds for nonsurgeons in order to convince them the program was a good idea. There was occasional early resistance based on a physician’s anecdotal experience – for example, having encountered a single case of ischemic optic neuritis several decades earlier – but such reservations were easily overcome by presenting hard data on the harmful effects of transfusion, he said.
Dr. Xydas said he had no relevant financial disclosures.
Major Finding: Use of a blood conservation algorithm reduced total blood product usage from an average of 5.8 U per patient at baseline to 3.5 U per patient, without compromising safety.
Data Source: Prospective study of 1,038 patients undergoing isolated coronary artery bypass grafting.
Disclosures: Dr. Xydas said he had no relevant financial disclosures.
CABG Beats PCI Regardless of SYNTAX Score
PARIS – Coronary artery bypass grafting was found to surpass percutaneous coronary intervention in a "real-world" registry of patients with three-vessel coronary disease, largely confirming the findings of the SYNTAX trial in patients with high SYNTAX scores and in those with low scores.
However, because the new data – obtained at 26 Japanese centers during 2005-2007 and involving nearly 3,000 patients – came from a nonrandomized registry, it may have been flawed by selection biases that skewed which patients underwent bypass surgery and which ones had percutaneous revascularization.
Coronary artery bypass grafting (CABG) "remains the standard treatment option for patients with triple-vessel disease, even when their SYNTAX scores are high," Dr. Hiroki Shiomi said at the annual congress of the European society of Cardiology.
"Use of PCI [percutaneous coronary intervention] in patients with high SYNTAX scores should be seriously discouraged unless their operative risk is prohibitively high," said Dr. Shiomi, a cardiologist at Kyoto University Hospital, Japan.
The registry results "suggested that the clinical outcome of PCI is not comparable with CABG even in patients with low syntax scores."
But the study’s findings also suggested that a selection bias occurred that even the risk-adjusted analysis used by Dr. Shiomi and his associates failed to adequately control, commented Dr. Uwe Zeymer, an interventional cardiologist and professor at the Institute for Myocardial Infarction Research in Ludwigshafen, Germany.
A major clue that selection bias came into play was that all-cause death during the 3-year follow-up of the study was 62% higher among the patients who underwent PCI, compared with those who underwent CABG, a statistically significant difference; in contrast, the rate of cardiac death was not significantly different between the CABG and PCI patients.
This discrepancy "says there was selection bias. Physicians had to decide what to do with patients who had a lot of comorbidities," and most of those patients probably underwent PCI, Dr. Zeymer said in an interview.
"If the patients [undergoing PCI and CABG] were the same clinically, you’d expect that with PCI you would at least have the same result" for all-cause mortality. "There seems to have been a selection bias toward using PCI in patients with more comorbidities.
"What we can say is that in this real-world situation, cardiac mortality was the same" with both revascularization methods, "which is reassuring for the use of PCI," he added.
CREDO-Kyoto (Coronary Revascularization Demonstrating Outcome Study in Kyoto) enrolled more than 13,000 patients who underwent coronary revascularization at any of 26 Japanese centers during 2005-2007, a period when all PCIs used a drug-eluting coronary stent.
The current analysis focused on 2,981 patients with triple-vessel disease and no left main coronary disease or acute MI, and included 1,825 who were treated with CABG and 1,156 treated by PCI. The average age of the PCI patients was 70 years; the CABG patients averaged 68 years old.
After 1 year of follow-up, the combined rate of death, MI, or stroke was 23% higher in the patients treated with PCI, compared with those treated with CABG, a statistically significant difference in an unadjusted analysis for the study’s primary end point.
After adjustment for baseline differences between the CABG and PCI patients, the rate of this combined end point was 47% higher among the PCI patients, also a statistically significant difference.
Analysis of several secondary end points showed a mixed pattern of differences between the two treatment groups in the adjusted analyses.
A statistically significant difference in favor of fewer events with CABG occurred in the end point of all-cause death, which was found to be 62% higher with PCI, as well as in the end point of MI, which was 2.39-fold higher in the PCI group.
The protection against MI by CABG, compared with PCI, was "particularly remarkable," Dr. Shiomi said.
But the end points of cardiac death and stroke showed no statistically significant differences between the two treatment groups in either the unadjusted or adjusted analyses.
Dr. Shiomi said that baseline SYNTAX scores were available for 94% of the patients. The mean score was 24 in the PCI patients and 30 for the CABG patients.
For the primary end point of death, MI, or stroke, treatment with PCI linked with a statistically significant 59% higher rate of the combined, primary end point, compared with CABG, in the adjusted analysis of patients with the highest SYNTAX scores (33 or greater); among patients with the lowest scores (22 or less), the adjusted analysis showed a significant, 66% higher rate of the combined outcome end point with PCI, compared with CABG.
For patients with SYNTAX scores of 23-32, the two treatment strategies led to similar adverse event rates in the two treatment groups.
Dr. Shiomi said he had no disclosures. Dr. Zeymer said that the Institute of MI Research in Germany, where he works, has received research grant support from multiple cardiac-device companies.
Cardiologists devised the SYNTAX score (EuroIntervention 2005;1:219-27) as a tool for quantifying the complexity of coronary anatomy in patients enrolled in the landmark SYNTAX trial (N. Engl. J. Med. 2009;360:961-72).
The SYNTAX investigators recently published the 3-year outcomes results from SYNTAX (Eur. Heart J. 2011;32:2125-34). Those results showed that patients treated with CABG had significantly fewer major adverse coronary and cerebrovascular events (MACCE) than did those who underwent PCI among patients with a high SYNTAX score (greater than 33), including the analysis with all patients, in patients with triple-vessel disease, and in patients with left main coronary disease. Among patients with intermediate SYNTAX scores (23-32), the MACCE rate was significantly lower with CABG than with PCI among all patients and in the subgroup with triple-vessel disease; in those with left main coronary disease, the MACCE rate was similar among patients treated with CABG and those treated with PCI. In patients with low SYNTAX scores (22 or less), the MACCE rates were similar among all CABG and PCI patients, as well as those in the subgroups who had either triple-vessel disease or left main coronary disease.
Given this background, it is a big surprise to learn that the Japanese study found a statistically significant advantage for CABG in the 3-year incidence of death, MI, and stroke among patients with a low SYNTAX score who were treated with CABG, compared with those who underwent PCI. This finding needs further examination in future randomized studies.
The CREDO-Kyoto study differed from SYNTAX by being nonrandomized. The current analysis focused exclusively on patients with triple-vessel coronary disease and excluded those with left main coronary disease.
The new Japanese results confirmed the key findings from the SYNTAX study in a large, real-world population. The new results show the usefulness of the SYNTAX score when it is used in clinical practice; however, in this study, the score calculation and the analysis of its relevance to outcomes occurred retrospectively. The ability of the SYNTAX score to help assign patients to optimal treatment with either CABG or PCI needs testing prospectively.
Frans Van de Werf, M.D., is professor and chairman of the department of cardiovascular medicine at University Hospitals Leuven (Belgium). He said that he had no relevant disclosures. This is adapted from the comments he made as the discussant for the study.
Cardiologists devised the SYNTAX score (EuroIntervention 2005;1:219-27) as a tool for quantifying the complexity of coronary anatomy in patients enrolled in the landmark SYNTAX trial (N. Engl. J. Med. 2009;360:961-72).
The SYNTAX investigators recently published the 3-year outcomes results from SYNTAX (Eur. Heart J. 2011;32:2125-34). Those results showed that patients treated with CABG had significantly fewer major adverse coronary and cerebrovascular events (MACCE) than did those who underwent PCI among patients with a high SYNTAX score (greater than 33), including the analysis with all patients, in patients with triple-vessel disease, and in patients with left main coronary disease. Among patients with intermediate SYNTAX scores (23-32), the MACCE rate was significantly lower with CABG than with PCI among all patients and in the subgroup with triple-vessel disease; in those with left main coronary disease, the MACCE rate was similar among patients treated with CABG and those treated with PCI. In patients with low SYNTAX scores (22 or less), the MACCE rates were similar among all CABG and PCI patients, as well as those in the subgroups who had either triple-vessel disease or left main coronary disease.
Given this background, it is a big surprise to learn that the Japanese study found a statistically significant advantage for CABG in the 3-year incidence of death, MI, and stroke among patients with a low SYNTAX score who were treated with CABG, compared with those who underwent PCI. This finding needs further examination in future randomized studies.
The CREDO-Kyoto study differed from SYNTAX by being nonrandomized. The current analysis focused exclusively on patients with triple-vessel coronary disease and excluded those with left main coronary disease.
The new Japanese results confirmed the key findings from the SYNTAX study in a large, real-world population. The new results show the usefulness of the SYNTAX score when it is used in clinical practice; however, in this study, the score calculation and the analysis of its relevance to outcomes occurred retrospectively. The ability of the SYNTAX score to help assign patients to optimal treatment with either CABG or PCI needs testing prospectively.
Frans Van de Werf, M.D., is professor and chairman of the department of cardiovascular medicine at University Hospitals Leuven (Belgium). He said that he had no relevant disclosures. This is adapted from the comments he made as the discussant for the study.
Cardiologists devised the SYNTAX score (EuroIntervention 2005;1:219-27) as a tool for quantifying the complexity of coronary anatomy in patients enrolled in the landmark SYNTAX trial (N. Engl. J. Med. 2009;360:961-72).
The SYNTAX investigators recently published the 3-year outcomes results from SYNTAX (Eur. Heart J. 2011;32:2125-34). Those results showed that patients treated with CABG had significantly fewer major adverse coronary and cerebrovascular events (MACCE) than did those who underwent PCI among patients with a high SYNTAX score (greater than 33), including the analysis with all patients, in patients with triple-vessel disease, and in patients with left main coronary disease. Among patients with intermediate SYNTAX scores (23-32), the MACCE rate was significantly lower with CABG than with PCI among all patients and in the subgroup with triple-vessel disease; in those with left main coronary disease, the MACCE rate was similar among patients treated with CABG and those treated with PCI. In patients with low SYNTAX scores (22 or less), the MACCE rates were similar among all CABG and PCI patients, as well as those in the subgroups who had either triple-vessel disease or left main coronary disease.
Given this background, it is a big surprise to learn that the Japanese study found a statistically significant advantage for CABG in the 3-year incidence of death, MI, and stroke among patients with a low SYNTAX score who were treated with CABG, compared with those who underwent PCI. This finding needs further examination in future randomized studies.
The CREDO-Kyoto study differed from SYNTAX by being nonrandomized. The current analysis focused exclusively on patients with triple-vessel coronary disease and excluded those with left main coronary disease.
The new Japanese results confirmed the key findings from the SYNTAX study in a large, real-world population. The new results show the usefulness of the SYNTAX score when it is used in clinical practice; however, in this study, the score calculation and the analysis of its relevance to outcomes occurred retrospectively. The ability of the SYNTAX score to help assign patients to optimal treatment with either CABG or PCI needs testing prospectively.
Frans Van de Werf, M.D., is professor and chairman of the department of cardiovascular medicine at University Hospitals Leuven (Belgium). He said that he had no relevant disclosures. This is adapted from the comments he made as the discussant for the study.
PARIS – Coronary artery bypass grafting was found to surpass percutaneous coronary intervention in a "real-world" registry of patients with three-vessel coronary disease, largely confirming the findings of the SYNTAX trial in patients with high SYNTAX scores and in those with low scores.
However, because the new data – obtained at 26 Japanese centers during 2005-2007 and involving nearly 3,000 patients – came from a nonrandomized registry, it may have been flawed by selection biases that skewed which patients underwent bypass surgery and which ones had percutaneous revascularization.
Coronary artery bypass grafting (CABG) "remains the standard treatment option for patients with triple-vessel disease, even when their SYNTAX scores are high," Dr. Hiroki Shiomi said at the annual congress of the European society of Cardiology.
"Use of PCI [percutaneous coronary intervention] in patients with high SYNTAX scores should be seriously discouraged unless their operative risk is prohibitively high," said Dr. Shiomi, a cardiologist at Kyoto University Hospital, Japan.
The registry results "suggested that the clinical outcome of PCI is not comparable with CABG even in patients with low syntax scores."
But the study’s findings also suggested that a selection bias occurred that even the risk-adjusted analysis used by Dr. Shiomi and his associates failed to adequately control, commented Dr. Uwe Zeymer, an interventional cardiologist and professor at the Institute for Myocardial Infarction Research in Ludwigshafen, Germany.
A major clue that selection bias came into play was that all-cause death during the 3-year follow-up of the study was 62% higher among the patients who underwent PCI, compared with those who underwent CABG, a statistically significant difference; in contrast, the rate of cardiac death was not significantly different between the CABG and PCI patients.
This discrepancy "says there was selection bias. Physicians had to decide what to do with patients who had a lot of comorbidities," and most of those patients probably underwent PCI, Dr. Zeymer said in an interview.
"If the patients [undergoing PCI and CABG] were the same clinically, you’d expect that with PCI you would at least have the same result" for all-cause mortality. "There seems to have been a selection bias toward using PCI in patients with more comorbidities.
"What we can say is that in this real-world situation, cardiac mortality was the same" with both revascularization methods, "which is reassuring for the use of PCI," he added.
CREDO-Kyoto (Coronary Revascularization Demonstrating Outcome Study in Kyoto) enrolled more than 13,000 patients who underwent coronary revascularization at any of 26 Japanese centers during 2005-2007, a period when all PCIs used a drug-eluting coronary stent.
The current analysis focused on 2,981 patients with triple-vessel disease and no left main coronary disease or acute MI, and included 1,825 who were treated with CABG and 1,156 treated by PCI. The average age of the PCI patients was 70 years; the CABG patients averaged 68 years old.
After 1 year of follow-up, the combined rate of death, MI, or stroke was 23% higher in the patients treated with PCI, compared with those treated with CABG, a statistically significant difference in an unadjusted analysis for the study’s primary end point.
After adjustment for baseline differences between the CABG and PCI patients, the rate of this combined end point was 47% higher among the PCI patients, also a statistically significant difference.
Analysis of several secondary end points showed a mixed pattern of differences between the two treatment groups in the adjusted analyses.
A statistically significant difference in favor of fewer events with CABG occurred in the end point of all-cause death, which was found to be 62% higher with PCI, as well as in the end point of MI, which was 2.39-fold higher in the PCI group.
The protection against MI by CABG, compared with PCI, was "particularly remarkable," Dr. Shiomi said.
But the end points of cardiac death and stroke showed no statistically significant differences between the two treatment groups in either the unadjusted or adjusted analyses.
Dr. Shiomi said that baseline SYNTAX scores were available for 94% of the patients. The mean score was 24 in the PCI patients and 30 for the CABG patients.
For the primary end point of death, MI, or stroke, treatment with PCI linked with a statistically significant 59% higher rate of the combined, primary end point, compared with CABG, in the adjusted analysis of patients with the highest SYNTAX scores (33 or greater); among patients with the lowest scores (22 or less), the adjusted analysis showed a significant, 66% higher rate of the combined outcome end point with PCI, compared with CABG.
For patients with SYNTAX scores of 23-32, the two treatment strategies led to similar adverse event rates in the two treatment groups.
Dr. Shiomi said he had no disclosures. Dr. Zeymer said that the Institute of MI Research in Germany, where he works, has received research grant support from multiple cardiac-device companies.
PARIS – Coronary artery bypass grafting was found to surpass percutaneous coronary intervention in a "real-world" registry of patients with three-vessel coronary disease, largely confirming the findings of the SYNTAX trial in patients with high SYNTAX scores and in those with low scores.
However, because the new data – obtained at 26 Japanese centers during 2005-2007 and involving nearly 3,000 patients – came from a nonrandomized registry, it may have been flawed by selection biases that skewed which patients underwent bypass surgery and which ones had percutaneous revascularization.
Coronary artery bypass grafting (CABG) "remains the standard treatment option for patients with triple-vessel disease, even when their SYNTAX scores are high," Dr. Hiroki Shiomi said at the annual congress of the European society of Cardiology.
"Use of PCI [percutaneous coronary intervention] in patients with high SYNTAX scores should be seriously discouraged unless their operative risk is prohibitively high," said Dr. Shiomi, a cardiologist at Kyoto University Hospital, Japan.
The registry results "suggested that the clinical outcome of PCI is not comparable with CABG even in patients with low syntax scores."
But the study’s findings also suggested that a selection bias occurred that even the risk-adjusted analysis used by Dr. Shiomi and his associates failed to adequately control, commented Dr. Uwe Zeymer, an interventional cardiologist and professor at the Institute for Myocardial Infarction Research in Ludwigshafen, Germany.
A major clue that selection bias came into play was that all-cause death during the 3-year follow-up of the study was 62% higher among the patients who underwent PCI, compared with those who underwent CABG, a statistically significant difference; in contrast, the rate of cardiac death was not significantly different between the CABG and PCI patients.
This discrepancy "says there was selection bias. Physicians had to decide what to do with patients who had a lot of comorbidities," and most of those patients probably underwent PCI, Dr. Zeymer said in an interview.
"If the patients [undergoing PCI and CABG] were the same clinically, you’d expect that with PCI you would at least have the same result" for all-cause mortality. "There seems to have been a selection bias toward using PCI in patients with more comorbidities.
"What we can say is that in this real-world situation, cardiac mortality was the same" with both revascularization methods, "which is reassuring for the use of PCI," he added.
CREDO-Kyoto (Coronary Revascularization Demonstrating Outcome Study in Kyoto) enrolled more than 13,000 patients who underwent coronary revascularization at any of 26 Japanese centers during 2005-2007, a period when all PCIs used a drug-eluting coronary stent.
The current analysis focused on 2,981 patients with triple-vessel disease and no left main coronary disease or acute MI, and included 1,825 who were treated with CABG and 1,156 treated by PCI. The average age of the PCI patients was 70 years; the CABG patients averaged 68 years old.
After 1 year of follow-up, the combined rate of death, MI, or stroke was 23% higher in the patients treated with PCI, compared with those treated with CABG, a statistically significant difference in an unadjusted analysis for the study’s primary end point.
After adjustment for baseline differences between the CABG and PCI patients, the rate of this combined end point was 47% higher among the PCI patients, also a statistically significant difference.
Analysis of several secondary end points showed a mixed pattern of differences between the two treatment groups in the adjusted analyses.
A statistically significant difference in favor of fewer events with CABG occurred in the end point of all-cause death, which was found to be 62% higher with PCI, as well as in the end point of MI, which was 2.39-fold higher in the PCI group.
The protection against MI by CABG, compared with PCI, was "particularly remarkable," Dr. Shiomi said.
But the end points of cardiac death and stroke showed no statistically significant differences between the two treatment groups in either the unadjusted or adjusted analyses.
Dr. Shiomi said that baseline SYNTAX scores were available for 94% of the patients. The mean score was 24 in the PCI patients and 30 for the CABG patients.
For the primary end point of death, MI, or stroke, treatment with PCI linked with a statistically significant 59% higher rate of the combined, primary end point, compared with CABG, in the adjusted analysis of patients with the highest SYNTAX scores (33 or greater); among patients with the lowest scores (22 or less), the adjusted analysis showed a significant, 66% higher rate of the combined outcome end point with PCI, compared with CABG.
For patients with SYNTAX scores of 23-32, the two treatment strategies led to similar adverse event rates in the two treatment groups.
Dr. Shiomi said he had no disclosures. Dr. Zeymer said that the Institute of MI Research in Germany, where he works, has received research grant support from multiple cardiac-device companies.
Hospital Readmission Rates Stagnant
Many hospitals may be unprepared for a new Medicare requirement to lower readmissions, and could face resulting financial penalties, according to a new report from the Dartmouth Atlas Project.
Over a 5-year period, hospitals made little progress in reducing readmissions among Medicare beneficiaries 65 years and older. The Dartmouth Atlas researchers found that surgical 30-day readmission rates were 12.7% in both 2004 and 2009, and medical 30-day readmission rates rose from 15.9% in 2004 to 16.1% in 2009. Trends were similar for specific conditions. For example, rates were relatively unchanged for congestive heart failure (20.9% vs. 21.2%) and pneumonia (15.1% vs. 15.3%). However, U.S. hospitals showed some improvement in acute myocardial infarctions, reducing 30-day readmissions from 19.4% in 2004 to 18.5% in 2009.
"For a long-standing and well-recognized problem, not much progress has been made," Dr. David C. Goodman, the study’s lead author and director of the Center for Health Policy Research at the Dartmouth Institute for Health Policy and Clinical Practice, said during a press conference to release the findings.
The researchers analyzed data for fee-for-service Medicare beneficiaries aged 65 years and older who lived in 306 Dartmouth Atlas hospital referral regions and had both Part A and Part B Medicare coverage.
The Affordable Care Act calls on the Centers for Medicare and Medicaid Services to start measuring 30-day hospital readmission rates and to penalize poor performers. In October 2012, hospitals with high readmission rates will face penalties of 1% of their total Medicare billings, increasing to 2% the following year.
Part of the solution to reducing hospital readmissions is good discharge planning, Dr. Goodman said. "This sounds simple but oftendoesn’t happen."
That planning should include having the care team in the hospital develop a care plan and communicate that plan to the patient and their family. It also means ensuring that the patient has all the necessary prescriptions, understands what medications to take and when, and can get their prescriptions. And health care providers in the hospital should also help patients set up follow-up appointments with their primary care physician, Dr. Goodman said.
But aside from discharge planning, there are also "hidden" factors such as how local patterns of hospital use affect readmission rates. Dr. Goodman and his colleagues found that communities and health care systems with higher underlying admission rates also tended to have higher rates of hospital readmission.
The researchers reported no financial conflicts.
Many hospitals may be unprepared for a new Medicare requirement to lower readmissions, and could face resulting financial penalties, according to a new report from the Dartmouth Atlas Project.
Over a 5-year period, hospitals made little progress in reducing readmissions among Medicare beneficiaries 65 years and older. The Dartmouth Atlas researchers found that surgical 30-day readmission rates were 12.7% in both 2004 and 2009, and medical 30-day readmission rates rose from 15.9% in 2004 to 16.1% in 2009. Trends were similar for specific conditions. For example, rates were relatively unchanged for congestive heart failure (20.9% vs. 21.2%) and pneumonia (15.1% vs. 15.3%). However, U.S. hospitals showed some improvement in acute myocardial infarctions, reducing 30-day readmissions from 19.4% in 2004 to 18.5% in 2009.
"For a long-standing and well-recognized problem, not much progress has been made," Dr. David C. Goodman, the study’s lead author and director of the Center for Health Policy Research at the Dartmouth Institute for Health Policy and Clinical Practice, said during a press conference to release the findings.
The researchers analyzed data for fee-for-service Medicare beneficiaries aged 65 years and older who lived in 306 Dartmouth Atlas hospital referral regions and had both Part A and Part B Medicare coverage.
The Affordable Care Act calls on the Centers for Medicare and Medicaid Services to start measuring 30-day hospital readmission rates and to penalize poor performers. In October 2012, hospitals with high readmission rates will face penalties of 1% of their total Medicare billings, increasing to 2% the following year.
Part of the solution to reducing hospital readmissions is good discharge planning, Dr. Goodman said. "This sounds simple but oftendoesn’t happen."
That planning should include having the care team in the hospital develop a care plan and communicate that plan to the patient and their family. It also means ensuring that the patient has all the necessary prescriptions, understands what medications to take and when, and can get their prescriptions. And health care providers in the hospital should also help patients set up follow-up appointments with their primary care physician, Dr. Goodman said.
But aside from discharge planning, there are also "hidden" factors such as how local patterns of hospital use affect readmission rates. Dr. Goodman and his colleagues found that communities and health care systems with higher underlying admission rates also tended to have higher rates of hospital readmission.
The researchers reported no financial conflicts.
Many hospitals may be unprepared for a new Medicare requirement to lower readmissions, and could face resulting financial penalties, according to a new report from the Dartmouth Atlas Project.
Over a 5-year period, hospitals made little progress in reducing readmissions among Medicare beneficiaries 65 years and older. The Dartmouth Atlas researchers found that surgical 30-day readmission rates were 12.7% in both 2004 and 2009, and medical 30-day readmission rates rose from 15.9% in 2004 to 16.1% in 2009. Trends were similar for specific conditions. For example, rates were relatively unchanged for congestive heart failure (20.9% vs. 21.2%) and pneumonia (15.1% vs. 15.3%). However, U.S. hospitals showed some improvement in acute myocardial infarctions, reducing 30-day readmissions from 19.4% in 2004 to 18.5% in 2009.
"For a long-standing and well-recognized problem, not much progress has been made," Dr. David C. Goodman, the study’s lead author and director of the Center for Health Policy Research at the Dartmouth Institute for Health Policy and Clinical Practice, said during a press conference to release the findings.
The researchers analyzed data for fee-for-service Medicare beneficiaries aged 65 years and older who lived in 306 Dartmouth Atlas hospital referral regions and had both Part A and Part B Medicare coverage.
The Affordable Care Act calls on the Centers for Medicare and Medicaid Services to start measuring 30-day hospital readmission rates and to penalize poor performers. In October 2012, hospitals with high readmission rates will face penalties of 1% of their total Medicare billings, increasing to 2% the following year.
Part of the solution to reducing hospital readmissions is good discharge planning, Dr. Goodman said. "This sounds simple but oftendoesn’t happen."
That planning should include having the care team in the hospital develop a care plan and communicate that plan to the patient and their family. It also means ensuring that the patient has all the necessary prescriptions, understands what medications to take and when, and can get their prescriptions. And health care providers in the hospital should also help patients set up follow-up appointments with their primary care physician, Dr. Goodman said.
But aside from discharge planning, there are also "hidden" factors such as how local patterns of hospital use affect readmission rates. Dr. Goodman and his colleagues found that communities and health care systems with higher underlying admission rates also tended to have higher rates of hospital readmission.
The researchers reported no financial conflicts.
Major Finding: Medicare beneficiaries aged 65 years and older had a medical 30-day readmission rate of 16.1% in 2009, up slightly from 15.9% in 2004.
Data Source: Medicare fee-for-service hospital claims for discharges between July 1, 2003-June 20, 2004 and July 1, 2008-June 30, 2009.
Disclosures: The Dartmouth Atlas Project receives most of its funding from the Robert Wood Johnson Foundation, the National Institute on Aging, the California Healthcare Foundation, the United Healthcare Foundation, and the WellPoint Foundation. The researchers reported no financial conflicts.
Lower Radiation Dose Improves Lung Cancer Survival
MIAMI BEACH – Less turned out to be better in a large clinical trial comparing radiation doses in patients treated with radiation and chemotherapy for stage III non–small cell lung cancer, investigators reported here.
The median overall survival rate at 1 year was 81% for patients treated with standard-dose (60 Gy) radiation, compared with 70.4% for those who received the high dose (74 Gy), according to preliminary findings from the radiation-dose arm of the ongoing phase III Radiation Therapy Oncology Group (RTOG) 0617 trial. The respective median survival rates were 21.7 months and 20.7 months (P = .02).
A planned interim analysis from the trial showed that the radiation comparison had crossed the prespecified boundary for futility, and the high-dose arm was stopped in June 2011, reported Dr. Jeffrey Bradley from Washington University in St. Louis at the annual meeting of the American Society for Radiation Oncology.
"I think this changes practice: If [cancer centers] weren’t using 60 Gray before, perhaps they should go back to using 60 Gray, because it does not appear that a higher dose is better," Dr. Bradley commented at the annual meeting of the American Society of Radiation Oncology (ASTRO).
Dr. Tim R. Williams, from the Lynn Cancer Institute at Boca Raton (Fla.) Regional Hospital, the immediate-past chairman of ASTRO, noted that his center has used high-dose radiation in stage III non–small cell lung cancer (NSCLC) patients for about 5 years. Although practice patterns vary, it’s likely that many treatment centers currently use the higher dose, he said.
In RTOG 0617, a total of 500 patients with stage IIIA/IIIB NSCLC were scheduled for randomization to one of four arms in a 2 x 2 factorial design with each patient assigned to receive either 74 Gy or 60 Gy radiation with or without cetuximab (Erbitux), on a background chemotherapy regimen of weekly paclitaxel (45 mg/m2) and carboplatin (titrated to an area-under-the-curve of 2).
The radiation was delivered in 2-Gy fractions over 30-37 fractions.
The analysis was performed on 426 patients who had been enrolled in the study before June 17, 2011.
Seeking to understand why the higher radiation dose was not better – the investigators had originally hypothesized that 74 Gy would result in a 7-month improvement in overall survival vs. 64 Gy – they performed univariate analyses, and found that significant predictors for better outcomes included continuous therapy, nonsquamous histology, and, female gender. In multivariate analysis, radiation dose (60 Gy vs. 74 Gy) was associated with a hazard ratio for overall survival of 1.48 (P = .038), nonsquamous histology versus squamous was associated with an HR of 1.52 (P = .025), and gross or internal tumor volume had a small but significant HR of 1.002 (P = .011).
Dr. Benjamin Movsas, chair of radiation oncology at the Henry Ford Health System in Detroit, the invited discussant, said that "as of 2011, level I evidence demonstrates no role for dose escalation in stage III non–small cell lung cancer."
He noted that although there were small differences between the radiation dose groups in terms of tumor histology, gross tumor volume, and other factors, they were not large enough to explain the differences in outcomes.
Citing the advice of his late father, also a physician, Dr. Movsas reminded the audience that "More is not always better."
The trial is continuing, but with patients assigned to 60 Gy radiation only, with the goal of evaluating the secondary study end point of overall survival of patients with or without cetuximab added to concurrent chemoradiotherapy.
The RTOG 0617 trial is supported by grants from the U.S. National Cancer Institute, with additional support from Bristol-Myers Squibb and ImClone.
Dr. Bradley and Dr. Williams had no disclosures. Dr. Movsas disclosed departmental research support from Varian and Philips. He also has served as a chair of an RTOG committee, but was not involved in the 0617 study.
MIAMI BEACH – Less turned out to be better in a large clinical trial comparing radiation doses in patients treated with radiation and chemotherapy for stage III non–small cell lung cancer, investigators reported here.
The median overall survival rate at 1 year was 81% for patients treated with standard-dose (60 Gy) radiation, compared with 70.4% for those who received the high dose (74 Gy), according to preliminary findings from the radiation-dose arm of the ongoing phase III Radiation Therapy Oncology Group (RTOG) 0617 trial. The respective median survival rates were 21.7 months and 20.7 months (P = .02).
A planned interim analysis from the trial showed that the radiation comparison had crossed the prespecified boundary for futility, and the high-dose arm was stopped in June 2011, reported Dr. Jeffrey Bradley from Washington University in St. Louis at the annual meeting of the American Society for Radiation Oncology.
"I think this changes practice: If [cancer centers] weren’t using 60 Gray before, perhaps they should go back to using 60 Gray, because it does not appear that a higher dose is better," Dr. Bradley commented at the annual meeting of the American Society of Radiation Oncology (ASTRO).
Dr. Tim R. Williams, from the Lynn Cancer Institute at Boca Raton (Fla.) Regional Hospital, the immediate-past chairman of ASTRO, noted that his center has used high-dose radiation in stage III non–small cell lung cancer (NSCLC) patients for about 5 years. Although practice patterns vary, it’s likely that many treatment centers currently use the higher dose, he said.
In RTOG 0617, a total of 500 patients with stage IIIA/IIIB NSCLC were scheduled for randomization to one of four arms in a 2 x 2 factorial design with each patient assigned to receive either 74 Gy or 60 Gy radiation with or without cetuximab (Erbitux), on a background chemotherapy regimen of weekly paclitaxel (45 mg/m2) and carboplatin (titrated to an area-under-the-curve of 2).
The radiation was delivered in 2-Gy fractions over 30-37 fractions.
The analysis was performed on 426 patients who had been enrolled in the study before June 17, 2011.
Seeking to understand why the higher radiation dose was not better – the investigators had originally hypothesized that 74 Gy would result in a 7-month improvement in overall survival vs. 64 Gy – they performed univariate analyses, and found that significant predictors for better outcomes included continuous therapy, nonsquamous histology, and, female gender. In multivariate analysis, radiation dose (60 Gy vs. 74 Gy) was associated with a hazard ratio for overall survival of 1.48 (P = .038), nonsquamous histology versus squamous was associated with an HR of 1.52 (P = .025), and gross or internal tumor volume had a small but significant HR of 1.002 (P = .011).
Dr. Benjamin Movsas, chair of radiation oncology at the Henry Ford Health System in Detroit, the invited discussant, said that "as of 2011, level I evidence demonstrates no role for dose escalation in stage III non–small cell lung cancer."
He noted that although there were small differences between the radiation dose groups in terms of tumor histology, gross tumor volume, and other factors, they were not large enough to explain the differences in outcomes.
Citing the advice of his late father, also a physician, Dr. Movsas reminded the audience that "More is not always better."
The trial is continuing, but with patients assigned to 60 Gy radiation only, with the goal of evaluating the secondary study end point of overall survival of patients with or without cetuximab added to concurrent chemoradiotherapy.
The RTOG 0617 trial is supported by grants from the U.S. National Cancer Institute, with additional support from Bristol-Myers Squibb and ImClone.
Dr. Bradley and Dr. Williams had no disclosures. Dr. Movsas disclosed departmental research support from Varian and Philips. He also has served as a chair of an RTOG committee, but was not involved in the 0617 study.
MIAMI BEACH – Less turned out to be better in a large clinical trial comparing radiation doses in patients treated with radiation and chemotherapy for stage III non–small cell lung cancer, investigators reported here.
The median overall survival rate at 1 year was 81% for patients treated with standard-dose (60 Gy) radiation, compared with 70.4% for those who received the high dose (74 Gy), according to preliminary findings from the radiation-dose arm of the ongoing phase III Radiation Therapy Oncology Group (RTOG) 0617 trial. The respective median survival rates were 21.7 months and 20.7 months (P = .02).
A planned interim analysis from the trial showed that the radiation comparison had crossed the prespecified boundary for futility, and the high-dose arm was stopped in June 2011, reported Dr. Jeffrey Bradley from Washington University in St. Louis at the annual meeting of the American Society for Radiation Oncology.
"I think this changes practice: If [cancer centers] weren’t using 60 Gray before, perhaps they should go back to using 60 Gray, because it does not appear that a higher dose is better," Dr. Bradley commented at the annual meeting of the American Society of Radiation Oncology (ASTRO).
Dr. Tim R. Williams, from the Lynn Cancer Institute at Boca Raton (Fla.) Regional Hospital, the immediate-past chairman of ASTRO, noted that his center has used high-dose radiation in stage III non–small cell lung cancer (NSCLC) patients for about 5 years. Although practice patterns vary, it’s likely that many treatment centers currently use the higher dose, he said.
In RTOG 0617, a total of 500 patients with stage IIIA/IIIB NSCLC were scheduled for randomization to one of four arms in a 2 x 2 factorial design with each patient assigned to receive either 74 Gy or 60 Gy radiation with or without cetuximab (Erbitux), on a background chemotherapy regimen of weekly paclitaxel (45 mg/m2) and carboplatin (titrated to an area-under-the-curve of 2).
The radiation was delivered in 2-Gy fractions over 30-37 fractions.
The analysis was performed on 426 patients who had been enrolled in the study before June 17, 2011.
Seeking to understand why the higher radiation dose was not better – the investigators had originally hypothesized that 74 Gy would result in a 7-month improvement in overall survival vs. 64 Gy – they performed univariate analyses, and found that significant predictors for better outcomes included continuous therapy, nonsquamous histology, and, female gender. In multivariate analysis, radiation dose (60 Gy vs. 74 Gy) was associated with a hazard ratio for overall survival of 1.48 (P = .038), nonsquamous histology versus squamous was associated with an HR of 1.52 (P = .025), and gross or internal tumor volume had a small but significant HR of 1.002 (P = .011).
Dr. Benjamin Movsas, chair of radiation oncology at the Henry Ford Health System in Detroit, the invited discussant, said that "as of 2011, level I evidence demonstrates no role for dose escalation in stage III non–small cell lung cancer."
He noted that although there were small differences between the radiation dose groups in terms of tumor histology, gross tumor volume, and other factors, they were not large enough to explain the differences in outcomes.
Citing the advice of his late father, also a physician, Dr. Movsas reminded the audience that "More is not always better."
The trial is continuing, but with patients assigned to 60 Gy radiation only, with the goal of evaluating the secondary study end point of overall survival of patients with or without cetuximab added to concurrent chemoradiotherapy.
The RTOG 0617 trial is supported by grants from the U.S. National Cancer Institute, with additional support from Bristol-Myers Squibb and ImClone.
Dr. Bradley and Dr. Williams had no disclosures. Dr. Movsas disclosed departmental research support from Varian and Philips. He also has served as a chair of an RTOG committee, but was not involved in the 0617 study.
Major Finding: Median overall survival among patients with stage III NSCLC treated with chemotherapy was 20.7 months with high-dose radiation (74 Gy), vs. 21.7 months with standard-dose radiation (60 Gy; P =.02).
Data Source: 426 patients enrolled in the randomized controlled RTOG 0617 trial.
Disclosures: Dr. Bradley and Dr. Williams had no disclosures. Dr. Movsas disclosed departmental research support from Varian and Philips. He also has served as a chair of an RTOG committee, but was not involved in the 0617 study.
Reflux Surgery Redos Safe, Outcomes Not Always Optimal
Reoperation for failed antireflux surgery can be performed safely in experienced centers, but outcomes are not as good as with primary operations, according to the results of two new analyses.
Dr. Nicholas R.A. Symons of Imperial College, London, and his associates performed a systematic literature review of 20 studies comprising 930 operations in 922 patients. "We can conclude that laparoscopic revision antireflux surgery, when performed in units with an interest in this type of surgery, is feasible and safe but subject to somewhat greater risk of conversion, higher morbidity, longer hospital stay, and poorer outcomes than primary laparoscopic fundoplication," the researchers said (Am. J. Surg. 2011;202:336-43).
Similarly, based on their retrospective study of 275 patients, Dr. Omar Awais of the University of Pittsburgh and his associates said, "Redo antireflux surgery can be performed safely in experienced centers, and as expected, the outcomes after redo antireflux surgery are not as good as [those of] first-time procedures. The operative approach depends on the underlying cause of failure" (Ann. Thorac. Surg. 2011;92:1083-90).
Between 2.8% and 4.4% of patients who undergo laparoscopic fundoplication at a specialist center will require late reoperation for persistent or recurrent symptoms, and there may be an increased revision rate after primary laparoscopic antireflux surgery compared with an initial open approach. Revision of failed antireflux surgery increasingly is being performed laparoscopically, but data about this approach are limited, Dr. Symons and his associates noted.
Their analysis included 19 case-control series and one prospective case-control study. Patients ranged in age from 13 to 83 years, and 57% were female. Of the eight articles in which the mean time between initial fundoplication and revision was given, the mean interval was 45.5 months (range 2-360 months). Of 12 studies noting the number of previous fundoplications the patients had undergone, 47 procedures (6.9%) were second reoperations and 9 (1.3%) were third reoperations.
Of the 18 studies documenting the type of initial surgery, 62% were laparoscopic, 35% were performed via laparotomy, 3% via thoracotomy, and 0.2% using video-assisted thoracoscopic surgery. Nissen fundoplication and Toupet partial fundoplication were the most common initial and revision procedures. Reflux/heartburn, experienced by 61% of patients, and dysphagia in 31% were the most common indications for laparoscopic reoperation, the investigators reported. Surgical duration, reported in 13 of the case series had an overall mean of 166 minutes for 721 patients.
The overall rate of conversion from laparoscopic reoperation to open surgery – reported in all the studies – was 7.2%, while the rate for patients who had more than one previous fundoplication was 19%. The most common reason for conversion, reported in 16 studies, was adhesions from previous surgery. The overall 7.2% conversion rate is higher than that noted for primary surgery (3.7%), "but is not excessive given the increased complexity of revision surgery," they commented.
Postoperative complications, reported from 18 studies, occurred in 14%, ranging from 0% to 44%. There were two deaths, both occurring in the same study. Pneumothorax was the most common complication, in 2% (14/810). Mean length of stay, reported in 18 studies, ranged from 1.2 to 6 days. Satisfactory to excellent results were reported for 84% of the operations; 5% of patients had a further antireflux procedure.
While revision laparoscopic fundoplication does not confer excessive morbidity, evidence for the efficacy of this procedure "is far less convincing, mainly owing to the mixture of reporting time points, inconsistency of end point definitions, and methods of assessment between studies. There appears to be a larger proportion of patients undergoing a re-revision surgery than after primary fundoplication," Dr. Symons commented.
The University of Pittsburgh study included patients who underwent minimally invasive reoperative surgery after failed fundoplication from 1996 to 2008. The 275 patients had a median age of 52 years (range 17-88 years), and 11.3% had had more than one prior antireflux surgery. As with the systematic review, the most common presenting symptoms were heartburn (64%) and dysphagia (49.5%). The median time from the prior operation to the redo operation was 36 months.
Transmediastinal migration of the wrap or a recurrent hiatal hernia (64%) were the most common causes of failure of the prior antireflux operation. Esophageal shortening was noted in 43% of patients, and a defect in the crural repair was identified in 4.4%. The most common procedure during reoperation was a Nissen fundoplication with or without a Collis gastroplasty. Nearly all of the redo procedures (93%) were done with a minimally invasive approach. There were eight conversions to open surgery due to extensive adhesions or a recognized intraoperative perforation, Dr. Awais and his associates reported.
Major complications included postoperative leaks in 3.3%; atrial fibrillation in 2%; and bleeding, pulmonary embolism, and Clostridium difficile colitis, each in less than 1%. Reexploration was required in 1.4%, for complications related to leak or bleeding. There was no perioperative mortality. Length of stay ranged from 1 to 75 days (median 3 days).
During follow-up of up to 14.5 years (median 3.3 years), 11.3% had a failure of the redo operation, requiring surgical intervention. An esophagectomy was required in four patients. The estimated probability of freedom from failure was 95% at 1 year, 93% at 2 years, and 84% at 5 years. Age and partial fundoplication were significantly associated with failure of the redo operation. and there was a trend for multiple redo operations to be associated with failure, Dr. Awais and his associates said.
Dysphagia decreased significantly after the redo procedure in 135 patients, with dysphagia scores declining from 2.7 to 1.4. Scores on the GERD-Health Related Quality of Life questionnaire, available in 186 patients, were excellent in 52% and satisfactory in 33%.
Both Dr. Symons and Dr. Awais stated that they had no disclosures.
Reoperation for failed antireflux surgery can be performed safely in experienced centers, but outcomes are not as good as with primary operations, according to the results of two new analyses.
Dr. Nicholas R.A. Symons of Imperial College, London, and his associates performed a systematic literature review of 20 studies comprising 930 operations in 922 patients. "We can conclude that laparoscopic revision antireflux surgery, when performed in units with an interest in this type of surgery, is feasible and safe but subject to somewhat greater risk of conversion, higher morbidity, longer hospital stay, and poorer outcomes than primary laparoscopic fundoplication," the researchers said (Am. J. Surg. 2011;202:336-43).
Similarly, based on their retrospective study of 275 patients, Dr. Omar Awais of the University of Pittsburgh and his associates said, "Redo antireflux surgery can be performed safely in experienced centers, and as expected, the outcomes after redo antireflux surgery are not as good as [those of] first-time procedures. The operative approach depends on the underlying cause of failure" (Ann. Thorac. Surg. 2011;92:1083-90).
Between 2.8% and 4.4% of patients who undergo laparoscopic fundoplication at a specialist center will require late reoperation for persistent or recurrent symptoms, and there may be an increased revision rate after primary laparoscopic antireflux surgery compared with an initial open approach. Revision of failed antireflux surgery increasingly is being performed laparoscopically, but data about this approach are limited, Dr. Symons and his associates noted.
Their analysis included 19 case-control series and one prospective case-control study. Patients ranged in age from 13 to 83 years, and 57% were female. Of the eight articles in which the mean time between initial fundoplication and revision was given, the mean interval was 45.5 months (range 2-360 months). Of 12 studies noting the number of previous fundoplications the patients had undergone, 47 procedures (6.9%) were second reoperations and 9 (1.3%) were third reoperations.
Of the 18 studies documenting the type of initial surgery, 62% were laparoscopic, 35% were performed via laparotomy, 3% via thoracotomy, and 0.2% using video-assisted thoracoscopic surgery. Nissen fundoplication and Toupet partial fundoplication were the most common initial and revision procedures. Reflux/heartburn, experienced by 61% of patients, and dysphagia in 31% were the most common indications for laparoscopic reoperation, the investigators reported. Surgical duration, reported in 13 of the case series had an overall mean of 166 minutes for 721 patients.
The overall rate of conversion from laparoscopic reoperation to open surgery – reported in all the studies – was 7.2%, while the rate for patients who had more than one previous fundoplication was 19%. The most common reason for conversion, reported in 16 studies, was adhesions from previous surgery. The overall 7.2% conversion rate is higher than that noted for primary surgery (3.7%), "but is not excessive given the increased complexity of revision surgery," they commented.
Postoperative complications, reported from 18 studies, occurred in 14%, ranging from 0% to 44%. There were two deaths, both occurring in the same study. Pneumothorax was the most common complication, in 2% (14/810). Mean length of stay, reported in 18 studies, ranged from 1.2 to 6 days. Satisfactory to excellent results were reported for 84% of the operations; 5% of patients had a further antireflux procedure.
While revision laparoscopic fundoplication does not confer excessive morbidity, evidence for the efficacy of this procedure "is far less convincing, mainly owing to the mixture of reporting time points, inconsistency of end point definitions, and methods of assessment between studies. There appears to be a larger proportion of patients undergoing a re-revision surgery than after primary fundoplication," Dr. Symons commented.
The University of Pittsburgh study included patients who underwent minimally invasive reoperative surgery after failed fundoplication from 1996 to 2008. The 275 patients had a median age of 52 years (range 17-88 years), and 11.3% had had more than one prior antireflux surgery. As with the systematic review, the most common presenting symptoms were heartburn (64%) and dysphagia (49.5%). The median time from the prior operation to the redo operation was 36 months.
Transmediastinal migration of the wrap or a recurrent hiatal hernia (64%) were the most common causes of failure of the prior antireflux operation. Esophageal shortening was noted in 43% of patients, and a defect in the crural repair was identified in 4.4%. The most common procedure during reoperation was a Nissen fundoplication with or without a Collis gastroplasty. Nearly all of the redo procedures (93%) were done with a minimally invasive approach. There were eight conversions to open surgery due to extensive adhesions or a recognized intraoperative perforation, Dr. Awais and his associates reported.
Major complications included postoperative leaks in 3.3%; atrial fibrillation in 2%; and bleeding, pulmonary embolism, and Clostridium difficile colitis, each in less than 1%. Reexploration was required in 1.4%, for complications related to leak or bleeding. There was no perioperative mortality. Length of stay ranged from 1 to 75 days (median 3 days).
During follow-up of up to 14.5 years (median 3.3 years), 11.3% had a failure of the redo operation, requiring surgical intervention. An esophagectomy was required in four patients. The estimated probability of freedom from failure was 95% at 1 year, 93% at 2 years, and 84% at 5 years. Age and partial fundoplication were significantly associated with failure of the redo operation. and there was a trend for multiple redo operations to be associated with failure, Dr. Awais and his associates said.
Dysphagia decreased significantly after the redo procedure in 135 patients, with dysphagia scores declining from 2.7 to 1.4. Scores on the GERD-Health Related Quality of Life questionnaire, available in 186 patients, were excellent in 52% and satisfactory in 33%.
Both Dr. Symons and Dr. Awais stated that they had no disclosures.
Reoperation for failed antireflux surgery can be performed safely in experienced centers, but outcomes are not as good as with primary operations, according to the results of two new analyses.
Dr. Nicholas R.A. Symons of Imperial College, London, and his associates performed a systematic literature review of 20 studies comprising 930 operations in 922 patients. "We can conclude that laparoscopic revision antireflux surgery, when performed in units with an interest in this type of surgery, is feasible and safe but subject to somewhat greater risk of conversion, higher morbidity, longer hospital stay, and poorer outcomes than primary laparoscopic fundoplication," the researchers said (Am. J. Surg. 2011;202:336-43).
Similarly, based on their retrospective study of 275 patients, Dr. Omar Awais of the University of Pittsburgh and his associates said, "Redo antireflux surgery can be performed safely in experienced centers, and as expected, the outcomes after redo antireflux surgery are not as good as [those of] first-time procedures. The operative approach depends on the underlying cause of failure" (Ann. Thorac. Surg. 2011;92:1083-90).
Between 2.8% and 4.4% of patients who undergo laparoscopic fundoplication at a specialist center will require late reoperation for persistent or recurrent symptoms, and there may be an increased revision rate after primary laparoscopic antireflux surgery compared with an initial open approach. Revision of failed antireflux surgery increasingly is being performed laparoscopically, but data about this approach are limited, Dr. Symons and his associates noted.
Their analysis included 19 case-control series and one prospective case-control study. Patients ranged in age from 13 to 83 years, and 57% were female. Of the eight articles in which the mean time between initial fundoplication and revision was given, the mean interval was 45.5 months (range 2-360 months). Of 12 studies noting the number of previous fundoplications the patients had undergone, 47 procedures (6.9%) were second reoperations and 9 (1.3%) were third reoperations.
Of the 18 studies documenting the type of initial surgery, 62% were laparoscopic, 35% were performed via laparotomy, 3% via thoracotomy, and 0.2% using video-assisted thoracoscopic surgery. Nissen fundoplication and Toupet partial fundoplication were the most common initial and revision procedures. Reflux/heartburn, experienced by 61% of patients, and dysphagia in 31% were the most common indications for laparoscopic reoperation, the investigators reported. Surgical duration, reported in 13 of the case series had an overall mean of 166 minutes for 721 patients.
The overall rate of conversion from laparoscopic reoperation to open surgery – reported in all the studies – was 7.2%, while the rate for patients who had more than one previous fundoplication was 19%. The most common reason for conversion, reported in 16 studies, was adhesions from previous surgery. The overall 7.2% conversion rate is higher than that noted for primary surgery (3.7%), "but is not excessive given the increased complexity of revision surgery," they commented.
Postoperative complications, reported from 18 studies, occurred in 14%, ranging from 0% to 44%. There were two deaths, both occurring in the same study. Pneumothorax was the most common complication, in 2% (14/810). Mean length of stay, reported in 18 studies, ranged from 1.2 to 6 days. Satisfactory to excellent results were reported for 84% of the operations; 5% of patients had a further antireflux procedure.
While revision laparoscopic fundoplication does not confer excessive morbidity, evidence for the efficacy of this procedure "is far less convincing, mainly owing to the mixture of reporting time points, inconsistency of end point definitions, and methods of assessment between studies. There appears to be a larger proportion of patients undergoing a re-revision surgery than after primary fundoplication," Dr. Symons commented.
The University of Pittsburgh study included patients who underwent minimally invasive reoperative surgery after failed fundoplication from 1996 to 2008. The 275 patients had a median age of 52 years (range 17-88 years), and 11.3% had had more than one prior antireflux surgery. As with the systematic review, the most common presenting symptoms were heartburn (64%) and dysphagia (49.5%). The median time from the prior operation to the redo operation was 36 months.
Transmediastinal migration of the wrap or a recurrent hiatal hernia (64%) were the most common causes of failure of the prior antireflux operation. Esophageal shortening was noted in 43% of patients, and a defect in the crural repair was identified in 4.4%. The most common procedure during reoperation was a Nissen fundoplication with or without a Collis gastroplasty. Nearly all of the redo procedures (93%) were done with a minimally invasive approach. There were eight conversions to open surgery due to extensive adhesions or a recognized intraoperative perforation, Dr. Awais and his associates reported.
Major complications included postoperative leaks in 3.3%; atrial fibrillation in 2%; and bleeding, pulmonary embolism, and Clostridium difficile colitis, each in less than 1%. Reexploration was required in 1.4%, for complications related to leak or bleeding. There was no perioperative mortality. Length of stay ranged from 1 to 75 days (median 3 days).
During follow-up of up to 14.5 years (median 3.3 years), 11.3% had a failure of the redo operation, requiring surgical intervention. An esophagectomy was required in four patients. The estimated probability of freedom from failure was 95% at 1 year, 93% at 2 years, and 84% at 5 years. Age and partial fundoplication were significantly associated with failure of the redo operation. and there was a trend for multiple redo operations to be associated with failure, Dr. Awais and his associates said.
Dysphagia decreased significantly after the redo procedure in 135 patients, with dysphagia scores declining from 2.7 to 1.4. Scores on the GERD-Health Related Quality of Life questionnaire, available in 186 patients, were excellent in 52% and satisfactory in 33%.
Both Dr. Symons and Dr. Awais stated that they had no disclosures.