User login
The Official Newspaper of the American Association for Thoracic Surgery
Slowdown in health spending not due to reform
WASHINGTON – The nation’s health bill is slated to continue to grow slowly through the end of this year, but that trend is not attributable to the Affordable Care Act. By 2022, the ACA is expected to cut the number of uninsured by 30 million, but also add about 0.1% to the nation’s health tab each year, adding up to an additional $621 billion over the next decade, according to an annual estimate of trends in health spending by analysts from the Centers for Medicare and Medicaid Services in Health Affairs (doi:10.1377/hlthaff.2013.0721).
In 2012 spending overall grew only 3.9%, to $2.8 trillion. Spending is likely to stay under 4% in 2013, in line with the historically low rate of growth seen over the past few years.
The decrease in health spending cannot be attributed to the Affordable Care Act, which became law in 2010, they said.
"In our projections, we have incorporated some modest savings regarding delivery system reforms; however, at this time it’s a little too early to tell how substantial those savings will be in the longer term," Gigi Cuckler, an economist in the CMS Office of the Actuary, told reporters.
The most recent downturn in spending is driven by bigger trends. What has been seen over the past 50 years is that when the economy shrinks, so does health spending, said Ms. Cuckler. When the economy recovers, health care spending grows.
"Until we see evidence that relationship has been broken, it’s very difficult for us to conclude that something structural has occurred," said Stephen Heffler, also of the Office of the Actuary at the CMS.
Medicare spending grew by 4.6% in 2012, down from 6.2% growth in 2011. Factors included lower spending on skilled nursing facilities; lower spending on prescriptions drugs such as Plavix (clopidogrel), which went off-patent; and lower payments to some providers mandated by the ACA.
This year, Medicare spending will grow even more slowly, in part from a continued slowdown in spending on hospitals and physicians. The automatic 2% spending from sequestration will also play a role.
Medicaid spending grew just over 2% in 2012, in part because the federal government reduced its matching rate and states employed cost-containment efforts. Medicaid is expected to grow by almost 5% in 2013, partly because primary care physicians were being paid at the higher rate.
In 2014, when millions are expected to gain coverage through the ACA, health spending is expected to grow by 6% – almost 2% faster than expected if the law were not in effect. The main spending drivers in 2014 will be growth in Medicaid and growth in private health insurance.
There is a projected 7% increase in spending on physician services in 2014 – compared to just under 5% in 2012 and around 4% in 2013. That 7% figure would drop to a little under 5% if Congress allows the scheduled 25% cut in physician payments – mandated by the SGR – in January.
WASHINGTON – The nation’s health bill is slated to continue to grow slowly through the end of this year, but that trend is not attributable to the Affordable Care Act. By 2022, the ACA is expected to cut the number of uninsured by 30 million, but also add about 0.1% to the nation’s health tab each year, adding up to an additional $621 billion over the next decade, according to an annual estimate of trends in health spending by analysts from the Centers for Medicare and Medicaid Services in Health Affairs (doi:10.1377/hlthaff.2013.0721).
In 2012 spending overall grew only 3.9%, to $2.8 trillion. Spending is likely to stay under 4% in 2013, in line with the historically low rate of growth seen over the past few years.
The decrease in health spending cannot be attributed to the Affordable Care Act, which became law in 2010, they said.
"In our projections, we have incorporated some modest savings regarding delivery system reforms; however, at this time it’s a little too early to tell how substantial those savings will be in the longer term," Gigi Cuckler, an economist in the CMS Office of the Actuary, told reporters.
The most recent downturn in spending is driven by bigger trends. What has been seen over the past 50 years is that when the economy shrinks, so does health spending, said Ms. Cuckler. When the economy recovers, health care spending grows.
"Until we see evidence that relationship has been broken, it’s very difficult for us to conclude that something structural has occurred," said Stephen Heffler, also of the Office of the Actuary at the CMS.
Medicare spending grew by 4.6% in 2012, down from 6.2% growth in 2011. Factors included lower spending on skilled nursing facilities; lower spending on prescriptions drugs such as Plavix (clopidogrel), which went off-patent; and lower payments to some providers mandated by the ACA.
This year, Medicare spending will grow even more slowly, in part from a continued slowdown in spending on hospitals and physicians. The automatic 2% spending from sequestration will also play a role.
Medicaid spending grew just over 2% in 2012, in part because the federal government reduced its matching rate and states employed cost-containment efforts. Medicaid is expected to grow by almost 5% in 2013, partly because primary care physicians were being paid at the higher rate.
In 2014, when millions are expected to gain coverage through the ACA, health spending is expected to grow by 6% – almost 2% faster than expected if the law were not in effect. The main spending drivers in 2014 will be growth in Medicaid and growth in private health insurance.
There is a projected 7% increase in spending on physician services in 2014 – compared to just under 5% in 2012 and around 4% in 2013. That 7% figure would drop to a little under 5% if Congress allows the scheduled 25% cut in physician payments – mandated by the SGR – in January.
WASHINGTON – The nation’s health bill is slated to continue to grow slowly through the end of this year, but that trend is not attributable to the Affordable Care Act. By 2022, the ACA is expected to cut the number of uninsured by 30 million, but also add about 0.1% to the nation’s health tab each year, adding up to an additional $621 billion over the next decade, according to an annual estimate of trends in health spending by analysts from the Centers for Medicare and Medicaid Services in Health Affairs (doi:10.1377/hlthaff.2013.0721).
In 2012 spending overall grew only 3.9%, to $2.8 trillion. Spending is likely to stay under 4% in 2013, in line with the historically low rate of growth seen over the past few years.
The decrease in health spending cannot be attributed to the Affordable Care Act, which became law in 2010, they said.
"In our projections, we have incorporated some modest savings regarding delivery system reforms; however, at this time it’s a little too early to tell how substantial those savings will be in the longer term," Gigi Cuckler, an economist in the CMS Office of the Actuary, told reporters.
The most recent downturn in spending is driven by bigger trends. What has been seen over the past 50 years is that when the economy shrinks, so does health spending, said Ms. Cuckler. When the economy recovers, health care spending grows.
"Until we see evidence that relationship has been broken, it’s very difficult for us to conclude that something structural has occurred," said Stephen Heffler, also of the Office of the Actuary at the CMS.
Medicare spending grew by 4.6% in 2012, down from 6.2% growth in 2011. Factors included lower spending on skilled nursing facilities; lower spending on prescriptions drugs such as Plavix (clopidogrel), which went off-patent; and lower payments to some providers mandated by the ACA.
This year, Medicare spending will grow even more slowly, in part from a continued slowdown in spending on hospitals and physicians. The automatic 2% spending from sequestration will also play a role.
Medicaid spending grew just over 2% in 2012, in part because the federal government reduced its matching rate and states employed cost-containment efforts. Medicaid is expected to grow by almost 5% in 2013, partly because primary care physicians were being paid at the higher rate.
In 2014, when millions are expected to gain coverage through the ACA, health spending is expected to grow by 6% – almost 2% faster than expected if the law were not in effect. The main spending drivers in 2014 will be growth in Medicaid and growth in private health insurance.
There is a projected 7% increase in spending on physician services in 2014 – compared to just under 5% in 2012 and around 4% in 2013. That 7% figure would drop to a little under 5% if Congress allows the scheduled 25% cut in physician payments – mandated by the SGR – in January.
Apps track industry payments
Two new smartphone apps aim to help log drug, device, and diagnostic manufacturer payments to doctors and health care providers, as called for by the Affordable Care Act.
To promote transparency in relationships between providers and industry, the ACA requires that manufacturers track and report payments for consulting, honoraria, and more.
Originally known as the Sunshine Act, the effort is now called the Open Payments Program by the Centers for Medicare and Medicaid Services (CMS).
While physicians are not required to inventory anything of value they receive from manufacturers, CMS and many medical professional societies advise that they do so.
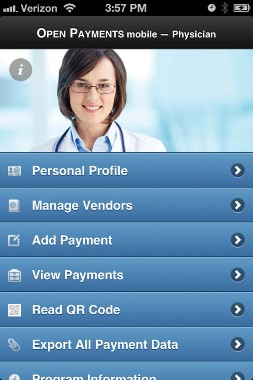
The app for physicians – Open Payments for Physicians – is designed to help doctors keep tabs on all their transactions in real time. Users can manually enter all the information regarding a particular transaction, for example, the receipt of a grant payment or a gift that’s worth more than $10.
The app is free and can be downloaded from the iTunes App Store or from Google Play.
CMS also created an app for industry representatives to use (Open Payments for Industry).
Industry users and physician users can exchange information with their apps. By using a built-in QR (quick response) code reader, the manufacturer can transfer a record of a transaction to the physician for review, according to the agency.
In a blog post, CMS Program Integrity Director Dr. Peter Budetti said the agency’s "foray into mobile technology is about providing user-friendly tools for doctors, manufacturers, and others in the health care industry to use in working with us to implement the law in a smart way."
The idea is that physicians can use the records contained in the app to compare what’s reported by manufacturers to CMS. There is a 45-day lag between when the data are reported to CMS and posted publicly. Physicians have that window to challenge the reports before they are posted on the Open Payments website. Corrections can be made later, but the erroneous data will likely stay public for awhile.
The first year of the program will be a little bit more forgiving. Data collected beginning Aug. 1 won’t be publicly reported until September 2014.
The apps can’t be used to directly transfer data to CMS, said the agency, which added that although it developed the apps, it will not "validate the accuracy of data stored in the apps, nor will it be responsible for protecting data stored in the apps."
Two new smartphone apps aim to help log drug, device, and diagnostic manufacturer payments to doctors and health care providers, as called for by the Affordable Care Act.
To promote transparency in relationships between providers and industry, the ACA requires that manufacturers track and report payments for consulting, honoraria, and more.
Originally known as the Sunshine Act, the effort is now called the Open Payments Program by the Centers for Medicare and Medicaid Services (CMS).
While physicians are not required to inventory anything of value they receive from manufacturers, CMS and many medical professional societies advise that they do so.
The app for physicians – Open Payments for Physicians – is designed to help doctors keep tabs on all their transactions in real time. Users can manually enter all the information regarding a particular transaction, for example, the receipt of a grant payment or a gift that’s worth more than $10.
The app is free and can be downloaded from the iTunes App Store or from Google Play.
CMS also created an app for industry representatives to use (Open Payments for Industry).
Industry users and physician users can exchange information with their apps. By using a built-in QR (quick response) code reader, the manufacturer can transfer a record of a transaction to the physician for review, according to the agency.
In a blog post, CMS Program Integrity Director Dr. Peter Budetti said the agency’s "foray into mobile technology is about providing user-friendly tools for doctors, manufacturers, and others in the health care industry to use in working with us to implement the law in a smart way."
The idea is that physicians can use the records contained in the app to compare what’s reported by manufacturers to CMS. There is a 45-day lag between when the data are reported to CMS and posted publicly. Physicians have that window to challenge the reports before they are posted on the Open Payments website. Corrections can be made later, but the erroneous data will likely stay public for awhile.
The first year of the program will be a little bit more forgiving. Data collected beginning Aug. 1 won’t be publicly reported until September 2014.
The apps can’t be used to directly transfer data to CMS, said the agency, which added that although it developed the apps, it will not "validate the accuracy of data stored in the apps, nor will it be responsible for protecting data stored in the apps."
Two new smartphone apps aim to help log drug, device, and diagnostic manufacturer payments to doctors and health care providers, as called for by the Affordable Care Act.
To promote transparency in relationships between providers and industry, the ACA requires that manufacturers track and report payments for consulting, honoraria, and more.
Originally known as the Sunshine Act, the effort is now called the Open Payments Program by the Centers for Medicare and Medicaid Services (CMS).
While physicians are not required to inventory anything of value they receive from manufacturers, CMS and many medical professional societies advise that they do so.
The app for physicians – Open Payments for Physicians – is designed to help doctors keep tabs on all their transactions in real time. Users can manually enter all the information regarding a particular transaction, for example, the receipt of a grant payment or a gift that’s worth more than $10.
The app is free and can be downloaded from the iTunes App Store or from Google Play.
CMS also created an app for industry representatives to use (Open Payments for Industry).
Industry users and physician users can exchange information with their apps. By using a built-in QR (quick response) code reader, the manufacturer can transfer a record of a transaction to the physician for review, according to the agency.
In a blog post, CMS Program Integrity Director Dr. Peter Budetti said the agency’s "foray into mobile technology is about providing user-friendly tools for doctors, manufacturers, and others in the health care industry to use in working with us to implement the law in a smart way."
The idea is that physicians can use the records contained in the app to compare what’s reported by manufacturers to CMS. There is a 45-day lag between when the data are reported to CMS and posted publicly. Physicians have that window to challenge the reports before they are posted on the Open Payments website. Corrections can be made later, but the erroneous data will likely stay public for awhile.
The first year of the program will be a little bit more forgiving. Data collected beginning Aug. 1 won’t be publicly reported until September 2014.
The apps can’t be used to directly transfer data to CMS, said the agency, which added that although it developed the apps, it will not "validate the accuracy of data stored in the apps, nor will it be responsible for protecting data stored in the apps."
SBRT in marginally operable NSCLC
ATLANTA – For older patients with marginally operable stage 1 non–small cell lung cancer, stereotactic body radiation therapy is significantly more cost effective than surgery.
For patients with clearly operable non–small cell lung cancer (NSCLS) tumors, however, lobectomy is the most cost-effective option, reported Dr. Anand Shah, a radiation oncology resident at Columbia University Medical Center in New York.
The findings, based on cost-effectiveness modeling, were robust over a wide range of assumptions, including various scenarios about treatment efficacies, toxicities, costs, and health state utilities.
"The rationale behind our study was that the traditional treatment for clearly operable patients with stage 1 lung cancer is lobectomy, whereas wedge resection and SBRT [stereotactic body radiation therapy] serve as alternatives in marginally operable patients.
Given an aging population and an increased prevalence of screening, it is likely more people will be diagnosed with stage 1 lung cancer, and thus we felt it was critical to compare the cost-effectiveness of these treatments," he said at the annual meeting of the American Society for Radiation Oncology.
The researchers created a Markov model in which hypothetical patient cohorts transition from one discrete, mutually exclusive health state to another at fixed time increments and at defined probabilities.
For a cohort with marginally operable disease, they compared SBRT with wedge resection, and for a cohort with clearly operable disease, they compared SBRT with lobectomy. Patients in the model were older than age 65
The model assumes that in both cohorts, SBRT will be similarly efficacious, but with higher toxicity for marginally operable patients, who are more likely to experience treatment-related morbidities.
The authors considered both open and less-invasive visually assisted surgical procedures for patients undergoing lobectomy and wedge resection. They considered costs from a Medicare perspective using 2012 dollars.
For the base case, SBRT for the marginally operable cohort cost a mean of $42,084, and the mean quality-adjusted life-year (QALY) gain was 8.03 years. In contrast, wedge resection cost a mean of $51,487, for a QALY gain of 7.93 years. In statistical parlance, SBRT for this cohort was the less costly and most effective strategy.
For clearly operable patients, however, SBRT was less costly than surgery. The mean cost was $40,107 vs. $49,083, but with less efficacy at 8.21 QALY compared with 8.89 for lobectomy. The investigators calculated an incremental cost-effectiveness ratio favoring lobectomy in this cohort, at a cost of $13,200 per QALY gained.
"We conducted a number of sensitivity analyses in which we varied the cost, efficacy, utility, and toxicity data, and in the marginally operable cohort SBRT was nearly always the dominant and thus cost-effective strategy. For patients who were considered clearly operable, lobectomy was the cost-effective treatment in nearly every sensitivity analysis," Dr. Shah said.
Dr. James B. Yu, the invited discussant, said that given current data, the findings of the study generally support current practice.
"However, even if you don’t agree that lobectomy is more cost effective for the clearly operable patient, at the very least this study will illuminate what we disagree about and where better data and clearer goals are needed," he said.
Dr. Yu is a therapeutic radiologist and cancer outcomes researcher at Yale School of Medicine in New Haven, Conn.
The funding source for the study was not disclosed. Dr. Shah and Dr. Yu reported having no relevant financial disclosures.
ATLANTA – For older patients with marginally operable stage 1 non–small cell lung cancer, stereotactic body radiation therapy is significantly more cost effective than surgery.
For patients with clearly operable non–small cell lung cancer (NSCLS) tumors, however, lobectomy is the most cost-effective option, reported Dr. Anand Shah, a radiation oncology resident at Columbia University Medical Center in New York.
The findings, based on cost-effectiveness modeling, were robust over a wide range of assumptions, including various scenarios about treatment efficacies, toxicities, costs, and health state utilities.
"The rationale behind our study was that the traditional treatment for clearly operable patients with stage 1 lung cancer is lobectomy, whereas wedge resection and SBRT [stereotactic body radiation therapy] serve as alternatives in marginally operable patients.
Given an aging population and an increased prevalence of screening, it is likely more people will be diagnosed with stage 1 lung cancer, and thus we felt it was critical to compare the cost-effectiveness of these treatments," he said at the annual meeting of the American Society for Radiation Oncology.
The researchers created a Markov model in which hypothetical patient cohorts transition from one discrete, mutually exclusive health state to another at fixed time increments and at defined probabilities.
For a cohort with marginally operable disease, they compared SBRT with wedge resection, and for a cohort with clearly operable disease, they compared SBRT with lobectomy. Patients in the model were older than age 65
The model assumes that in both cohorts, SBRT will be similarly efficacious, but with higher toxicity for marginally operable patients, who are more likely to experience treatment-related morbidities.
The authors considered both open and less-invasive visually assisted surgical procedures for patients undergoing lobectomy and wedge resection. They considered costs from a Medicare perspective using 2012 dollars.
For the base case, SBRT for the marginally operable cohort cost a mean of $42,084, and the mean quality-adjusted life-year (QALY) gain was 8.03 years. In contrast, wedge resection cost a mean of $51,487, for a QALY gain of 7.93 years. In statistical parlance, SBRT for this cohort was the less costly and most effective strategy.
For clearly operable patients, however, SBRT was less costly than surgery. The mean cost was $40,107 vs. $49,083, but with less efficacy at 8.21 QALY compared with 8.89 for lobectomy. The investigators calculated an incremental cost-effectiveness ratio favoring lobectomy in this cohort, at a cost of $13,200 per QALY gained.
"We conducted a number of sensitivity analyses in which we varied the cost, efficacy, utility, and toxicity data, and in the marginally operable cohort SBRT was nearly always the dominant and thus cost-effective strategy. For patients who were considered clearly operable, lobectomy was the cost-effective treatment in nearly every sensitivity analysis," Dr. Shah said.
Dr. James B. Yu, the invited discussant, said that given current data, the findings of the study generally support current practice.
"However, even if you don’t agree that lobectomy is more cost effective for the clearly operable patient, at the very least this study will illuminate what we disagree about and where better data and clearer goals are needed," he said.
Dr. Yu is a therapeutic radiologist and cancer outcomes researcher at Yale School of Medicine in New Haven, Conn.
The funding source for the study was not disclosed. Dr. Shah and Dr. Yu reported having no relevant financial disclosures.
ATLANTA – For older patients with marginally operable stage 1 non–small cell lung cancer, stereotactic body radiation therapy is significantly more cost effective than surgery.
For patients with clearly operable non–small cell lung cancer (NSCLS) tumors, however, lobectomy is the most cost-effective option, reported Dr. Anand Shah, a radiation oncology resident at Columbia University Medical Center in New York.
The findings, based on cost-effectiveness modeling, were robust over a wide range of assumptions, including various scenarios about treatment efficacies, toxicities, costs, and health state utilities.
"The rationale behind our study was that the traditional treatment for clearly operable patients with stage 1 lung cancer is lobectomy, whereas wedge resection and SBRT [stereotactic body radiation therapy] serve as alternatives in marginally operable patients.
Given an aging population and an increased prevalence of screening, it is likely more people will be diagnosed with stage 1 lung cancer, and thus we felt it was critical to compare the cost-effectiveness of these treatments," he said at the annual meeting of the American Society for Radiation Oncology.
The researchers created a Markov model in which hypothetical patient cohorts transition from one discrete, mutually exclusive health state to another at fixed time increments and at defined probabilities.
For a cohort with marginally operable disease, they compared SBRT with wedge resection, and for a cohort with clearly operable disease, they compared SBRT with lobectomy. Patients in the model were older than age 65
The model assumes that in both cohorts, SBRT will be similarly efficacious, but with higher toxicity for marginally operable patients, who are more likely to experience treatment-related morbidities.
The authors considered both open and less-invasive visually assisted surgical procedures for patients undergoing lobectomy and wedge resection. They considered costs from a Medicare perspective using 2012 dollars.
For the base case, SBRT for the marginally operable cohort cost a mean of $42,084, and the mean quality-adjusted life-year (QALY) gain was 8.03 years. In contrast, wedge resection cost a mean of $51,487, for a QALY gain of 7.93 years. In statistical parlance, SBRT for this cohort was the less costly and most effective strategy.
For clearly operable patients, however, SBRT was less costly than surgery. The mean cost was $40,107 vs. $49,083, but with less efficacy at 8.21 QALY compared with 8.89 for lobectomy. The investigators calculated an incremental cost-effectiveness ratio favoring lobectomy in this cohort, at a cost of $13,200 per QALY gained.
"We conducted a number of sensitivity analyses in which we varied the cost, efficacy, utility, and toxicity data, and in the marginally operable cohort SBRT was nearly always the dominant and thus cost-effective strategy. For patients who were considered clearly operable, lobectomy was the cost-effective treatment in nearly every sensitivity analysis," Dr. Shah said.
Dr. James B. Yu, the invited discussant, said that given current data, the findings of the study generally support current practice.
"However, even if you don’t agree that lobectomy is more cost effective for the clearly operable patient, at the very least this study will illuminate what we disagree about and where better data and clearer goals are needed," he said.
Dr. Yu is a therapeutic radiologist and cancer outcomes researcher at Yale School of Medicine in New Haven, Conn.
The funding source for the study was not disclosed. Dr. Shah and Dr. Yu reported having no relevant financial disclosures.
Losartan effective in Marfan syndrome
AMSTERDAM – Daily losartan significantly slowed the aortic root dilatation rate in adults with Marfan syndrome in a 3-year randomized clinical trial.
"I think we can be positive about this treatment. We can now recommend losartan in clinical practice," Dr. Maarten Groenink said at the annual congress of the European Society of Cardiology.
The COMPARE (Cozaar in Marfan Patients Reduces Aortic Enlargement) trial included 218 patients at all four university Marfan centers in the Netherlands. Patients were randomized to oral losartan at a target dose of 100 mg/day or no losartan in addition to standard-of-care treatment with beta-blockers. Roughly half of the patients in the losartan group were unable to tolerate the full dose of losartan in addition to a beta-blocker; those patients were maintained on losartan at 50 mg/day. Aortic root diameter was measured by MRI at enrollment and after 3 years of prospective follow-up. The aortic dilatation rate was significantly lower in the losartan group than in controls both in the patients with a native root and in those who had undergone aortic root replacement surgery, reported Dr. Groenink, a cardiologist at the Academic Medical Center, Amsterdam.
There were no aortic dissections in the losartan group and two in the control arm. Elective aortic replacement surgery was performed in a similar number of patients in both groups.
Blood pressure was lower in the losartan group, yet blood pressure didn’t correlate with the aortic dilatation rate. Dr. Groenink speculated that losartan’s chief mechanism of benefit in Marfan syndrome is its ability to curb overexpression of transforming growth factor-beta, which weakens the structure of the media layer of the aortic wall. He said it’s unknown whether losartan’s benefits are specific to that drug or are a class effect.
Ongoing clinical trials are evaluating losartan in children and adolescents with Marfan syndrome, he said, adding that there is a solid rationale for beginning treatment as early in life as possible. "I believe the adverse effects on the aortic wall in Marfan syndrome are caused by the fibrillin defect but also by wear and tear due to cyclic stress by the beating heart. So you can hypothesize that the earlier you start treatment, the better," he explained.
The prevalence of Marfan syndrome has been estimated at 1 in 5,000, but Dr. Groenink suspects the syndrome may actually be more common than that.
Simultaneous with Dr. Groenink’s presentation at the ESC, the COMPARE results were published online (Eur. Heart J. 2013 [doi:10.1093/eurheartj/eht334]).
Dr. Groenink reported having no disclosures.

|
| Dr. John Gordon Harold |
COMPARE is a very important study whose results are going to mean a paradigm shift for the management of Marfan syndrome.
It is intriguing to consider that the benefits of losartan might possibly also extend to patients with thoracic aortic disease in general, a worthy topic for future investigation.
Dr. John Gordon Harold is with Cedars-Sinai Heart Institute, Los Angeles, and president of the American College of Cardiology. He had no relevant financial disclosures.

|
| Dr. John Gordon Harold |
COMPARE is a very important study whose results are going to mean a paradigm shift for the management of Marfan syndrome.
It is intriguing to consider that the benefits of losartan might possibly also extend to patients with thoracic aortic disease in general, a worthy topic for future investigation.
Dr. John Gordon Harold is with Cedars-Sinai Heart Institute, Los Angeles, and president of the American College of Cardiology. He had no relevant financial disclosures.

|
| Dr. John Gordon Harold |
COMPARE is a very important study whose results are going to mean a paradigm shift for the management of Marfan syndrome.
It is intriguing to consider that the benefits of losartan might possibly also extend to patients with thoracic aortic disease in general, a worthy topic for future investigation.
Dr. John Gordon Harold is with Cedars-Sinai Heart Institute, Los Angeles, and president of the American College of Cardiology. He had no relevant financial disclosures.
AMSTERDAM – Daily losartan significantly slowed the aortic root dilatation rate in adults with Marfan syndrome in a 3-year randomized clinical trial.
"I think we can be positive about this treatment. We can now recommend losartan in clinical practice," Dr. Maarten Groenink said at the annual congress of the European Society of Cardiology.
The COMPARE (Cozaar in Marfan Patients Reduces Aortic Enlargement) trial included 218 patients at all four university Marfan centers in the Netherlands. Patients were randomized to oral losartan at a target dose of 100 mg/day or no losartan in addition to standard-of-care treatment with beta-blockers. Roughly half of the patients in the losartan group were unable to tolerate the full dose of losartan in addition to a beta-blocker; those patients were maintained on losartan at 50 mg/day. Aortic root diameter was measured by MRI at enrollment and after 3 years of prospective follow-up. The aortic dilatation rate was significantly lower in the losartan group than in controls both in the patients with a native root and in those who had undergone aortic root replacement surgery, reported Dr. Groenink, a cardiologist at the Academic Medical Center, Amsterdam.
There were no aortic dissections in the losartan group and two in the control arm. Elective aortic replacement surgery was performed in a similar number of patients in both groups.
Blood pressure was lower in the losartan group, yet blood pressure didn’t correlate with the aortic dilatation rate. Dr. Groenink speculated that losartan’s chief mechanism of benefit in Marfan syndrome is its ability to curb overexpression of transforming growth factor-beta, which weakens the structure of the media layer of the aortic wall. He said it’s unknown whether losartan’s benefits are specific to that drug or are a class effect.
Ongoing clinical trials are evaluating losartan in children and adolescents with Marfan syndrome, he said, adding that there is a solid rationale for beginning treatment as early in life as possible. "I believe the adverse effects on the aortic wall in Marfan syndrome are caused by the fibrillin defect but also by wear and tear due to cyclic stress by the beating heart. So you can hypothesize that the earlier you start treatment, the better," he explained.
The prevalence of Marfan syndrome has been estimated at 1 in 5,000, but Dr. Groenink suspects the syndrome may actually be more common than that.
Simultaneous with Dr. Groenink’s presentation at the ESC, the COMPARE results were published online (Eur. Heart J. 2013 [doi:10.1093/eurheartj/eht334]).
Dr. Groenink reported having no disclosures.
AMSTERDAM – Daily losartan significantly slowed the aortic root dilatation rate in adults with Marfan syndrome in a 3-year randomized clinical trial.
"I think we can be positive about this treatment. We can now recommend losartan in clinical practice," Dr. Maarten Groenink said at the annual congress of the European Society of Cardiology.
The COMPARE (Cozaar in Marfan Patients Reduces Aortic Enlargement) trial included 218 patients at all four university Marfan centers in the Netherlands. Patients were randomized to oral losartan at a target dose of 100 mg/day or no losartan in addition to standard-of-care treatment with beta-blockers. Roughly half of the patients in the losartan group were unable to tolerate the full dose of losartan in addition to a beta-blocker; those patients were maintained on losartan at 50 mg/day. Aortic root diameter was measured by MRI at enrollment and after 3 years of prospective follow-up. The aortic dilatation rate was significantly lower in the losartan group than in controls both in the patients with a native root and in those who had undergone aortic root replacement surgery, reported Dr. Groenink, a cardiologist at the Academic Medical Center, Amsterdam.
There were no aortic dissections in the losartan group and two in the control arm. Elective aortic replacement surgery was performed in a similar number of patients in both groups.
Blood pressure was lower in the losartan group, yet blood pressure didn’t correlate with the aortic dilatation rate. Dr. Groenink speculated that losartan’s chief mechanism of benefit in Marfan syndrome is its ability to curb overexpression of transforming growth factor-beta, which weakens the structure of the media layer of the aortic wall. He said it’s unknown whether losartan’s benefits are specific to that drug or are a class effect.
Ongoing clinical trials are evaluating losartan in children and adolescents with Marfan syndrome, he said, adding that there is a solid rationale for beginning treatment as early in life as possible. "I believe the adverse effects on the aortic wall in Marfan syndrome are caused by the fibrillin defect but also by wear and tear due to cyclic stress by the beating heart. So you can hypothesize that the earlier you start treatment, the better," he explained.
The prevalence of Marfan syndrome has been estimated at 1 in 5,000, but Dr. Groenink suspects the syndrome may actually be more common than that.
Simultaneous with Dr. Groenink’s presentation at the ESC, the COMPARE results were published online (Eur. Heart J. 2013 [doi:10.1093/eurheartj/eht334]).
Dr. Groenink reported having no disclosures.
Major finding: The rate of aortic root enlargement during 3 years of prospective follow-up was 0.77 mm in losartan-treated patients with Marfan syndrome, significantly less than the 1.35 mm in patients on standard-of-care treatment with no losartan.
Data source: The COMPARE trial was a randomized, prospective, open-label multicenter study in which 218 patients with Marfan syndrome were randomized to losartan at a target dose of 100 mg or to no losartan and followed for 3 years with the aortic root dilatation rate as measured by MRI the primary endpoint.
Disclosures: The COMPARE trial was supported by the Dutch Heart Association. Dr. Groenink reported having no financial conflicts.
Spouses surveyed report influence on CTS applicant decision making
A resident’s ability to balance work and personal life is dependent on multiple factors, with spousal or partner approval likely comprising one of the most important, according to a report published in the Journal of Surgical Education. The report details the results of an electronic survey sent to thoracic surgery spouses via contact with all thoracic surgery residents over 2 years at two training programs and all current thoracic surgery residents in 2010.
A total of 66 surveys were completed and returned (a response rate of 19%), with 86% of the respondents being women, and 82% being married for a mean of 4.3 years; 59% had children, and 64% were planning on having more children within 3 years.
Nearly 90%of the spouses responding reported that they want to travel to more of the interviews, and that they had some to complete influence on which training program to attend, according to Mr. Michael Bohl and Dr. Rishindra M. Reddy of the Section of Thoracic Surgery at the University of Michigan, Ann Arbor (J. Surg. Education 2013;70:640-6).
Nearly 80% of the respondents also reported that they wanted more information on salary and on housing, as well as access to faculty spouses. The top 3 factors in chosing a program they reported were: quality of fellowship, geographic location, and proximity to family.
Knowledge of the extent of influence and the desires of spouses with regard to CTStraining positions might allow targeting these needs at a point early enough in the process to help capture general surgery residents interested in but not committed to CTS residency, the researchers suggested.
"The results show numerous demographic and characteristic trends which, if further validated by definitive studies, would be applicable to all post-surgery residency training programs and may help CTS programs to be more competitive in attracting applicants and their families.
The authors had no relevant disclosures.
A resident’s ability to balance work and personal life is dependent on multiple factors, with spousal or partner approval likely comprising one of the most important, according to a report published in the Journal of Surgical Education. The report details the results of an electronic survey sent to thoracic surgery spouses via contact with all thoracic surgery residents over 2 years at two training programs and all current thoracic surgery residents in 2010.
A total of 66 surveys were completed and returned (a response rate of 19%), with 86% of the respondents being women, and 82% being married for a mean of 4.3 years; 59% had children, and 64% were planning on having more children within 3 years.
Nearly 90%of the spouses responding reported that they want to travel to more of the interviews, and that they had some to complete influence on which training program to attend, according to Mr. Michael Bohl and Dr. Rishindra M. Reddy of the Section of Thoracic Surgery at the University of Michigan, Ann Arbor (J. Surg. Education 2013;70:640-6).
Nearly 80% of the respondents also reported that they wanted more information on salary and on housing, as well as access to faculty spouses. The top 3 factors in chosing a program they reported were: quality of fellowship, geographic location, and proximity to family.
Knowledge of the extent of influence and the desires of spouses with regard to CTStraining positions might allow targeting these needs at a point early enough in the process to help capture general surgery residents interested in but not committed to CTS residency, the researchers suggested.
"The results show numerous demographic and characteristic trends which, if further validated by definitive studies, would be applicable to all post-surgery residency training programs and may help CTS programs to be more competitive in attracting applicants and their families.
The authors had no relevant disclosures.
A resident’s ability to balance work and personal life is dependent on multiple factors, with spousal or partner approval likely comprising one of the most important, according to a report published in the Journal of Surgical Education. The report details the results of an electronic survey sent to thoracic surgery spouses via contact with all thoracic surgery residents over 2 years at two training programs and all current thoracic surgery residents in 2010.
A total of 66 surveys were completed and returned (a response rate of 19%), with 86% of the respondents being women, and 82% being married for a mean of 4.3 years; 59% had children, and 64% were planning on having more children within 3 years.
Nearly 90%of the spouses responding reported that they want to travel to more of the interviews, and that they had some to complete influence on which training program to attend, according to Mr. Michael Bohl and Dr. Rishindra M. Reddy of the Section of Thoracic Surgery at the University of Michigan, Ann Arbor (J. Surg. Education 2013;70:640-6).
Nearly 80% of the respondents also reported that they wanted more information on salary and on housing, as well as access to faculty spouses. The top 3 factors in chosing a program they reported were: quality of fellowship, geographic location, and proximity to family.
Knowledge of the extent of influence and the desires of spouses with regard to CTStraining positions might allow targeting these needs at a point early enough in the process to help capture general surgery residents interested in but not committed to CTS residency, the researchers suggested.
"The results show numerous demographic and characteristic trends which, if further validated by definitive studies, would be applicable to all post-surgery residency training programs and may help CTS programs to be more competitive in attracting applicants and their families.
The authors had no relevant disclosures.
Nix new anticoagulants with mechanical valves
AMSTERDAM -- The complete failure of the oral direct thrombin inhibitor dabigatran in the RE-ALIGN trial spells big trouble for efforts to use the novel oral factor Xa inhibitors for anticoagulation in patients with a mechanical heart valve, RE-ALIGN steering committee cochair Dr. Frans Van de Werf cautioned at the annual congress of the European Society of Cardiology.
"It would not be wise for a cardiologist to prescribe these agents for a patient with a mechanical heart valve," declared Dr. Van de Werf, professor and chairman of the department of cardiovascular medicine at the Catholic University at Leuven (Belgium).
Dabigatran (Pradaxa) is approved as a more effective and convenient alternative to warfarin for prevention of stroke and other systemic embolism in patients with nonvalvular atrial fibrillation. RE-ALIGN (Randomized, Phase II Study to Evaluate the Safety and Pharmacokinetics of Oral Dabigatran Etexilate in Patients After Heart Valve Replacement) was an effort to expand the drug's indications to include prevention of thromboembolic events in patients with mechanical heart valves.
The study was halted prematurely late last year when an interim analysis showed that dabigatran was both less safe and less protective than standard warfarin therapy. Shortly afterward, the Food and Drug Administration and the European regulatory agency warned that dabigatran is contraindicated in patients with mechanical heart valves. Dr. Van de Werf's report in Amsterdam, however, was the first presentation of the actual data.
RE-ALIGN was stopped after 252 patients with mechanical heart valves had been randomized 2:1 to dabigatran or warfarin. Dabigatran dosing was adjusted on the basis of creatinine clearance to achieve a trough plasma level of at least 50 ng/mL.
The study halt came in response to an interim analysis showing a 5% stroke rate in the dabigatran group, compared with 0% with warfarin. This was accompanied by a 4% rate of major bleeding with dabigatran, compared with 2% with warfarin, and a 27% rate of any bleeding with dabigatran versus 12% with warfarin. All dabigatran-treated patients with major bleeding had pericardial bleeding and were in the subgroup of participants who started on the direct thrombin inhibitor within 7 days post surgery.
Other unwelcome outcomes in the interim analysis included a 2% MI rate and 3% rate of valve thrombosis without symptoms in the dabigatran group, versus no such events in the warfarin arm.
"Overall, this study was clearly negative: more thrombotic events and more bleeding complications in spite of adjusting the dose of dabigatran based upon renal function," Dr. Van de Werf observed.
Simultaneous with his presentation of the RE-ALIGN findings at the ESC congress in Amsterdam, the results were published online (N. Engl. J. Med. 2013 Sept. 1 [doi:10.1056/NEJMoa1300615]).
In an accompanying editorial, Dr. Elaine M. Hylek of Boston University said the most likely explanation for the disappointing results in RE-ALIGN was that blood levels of dabigatran were inadequate (N. Engl. J. Med. 2013 Sept. 1 [doi:10.1056/NEJMe1310399]).
Dr. Van de Werf, however, rejected that as unlikely. Given that there were excesses of both thromboembolic and bleeding events, other dosing regimens would surely have sent rates of one type of complication or the other even higher, he argued.
Instead, he and his coinvestigators believe that the most likely explanation for the negative results lies in the differences in the mechanisms of action of dabigatran and warfarin. Dabigatran acts near the tail end of the coagulation cascade, on thrombin. In patients with a mechanical heart valve, thrombin is generated by exposure of blood to the artificial surface of the valve leaflets, activating what is known as the contact pathway of coagulation. Thrombin also is generated by the release of tissue factor from tissues injured during the valve surgery. Warfarin acts at both the contact pathway and tissue injury levels; dabigatran doesn't address either.
The novel oral factor Xa inhibitors don't act at the contact pathway and tissue injury levels, either, which is why Dr. Van de Werf cautioned against their off-label use in patients with a mechanical valve.
Discussant Dr. Alec Vahanian said that he "fully agrees" with Dr. Van de Werf's interpretation that the negative results most likely stemmed from dabigatran's absence of efficacy on the contact pathway.
"It sounds from this study that there is no future for dabigatran with this indication. We should not give this agent to a patient with a mechanical prosthesis, and personally I would extend that further and say we should not give this agent to patients with a bioprosthesis or other high-risk patients with severe valve disease. We don't have the evidence for that yet," said Dr. Vahanian, head of cardiology at Bichat Hospital, Paris.
Patients with a mechanical heart valve constitute a challenging population at extreme thromboembolic risk, but Dr. Vahanian offered a comforting prediction: "We are nearing the end of the era of mechanical prosthetic valves," he declared. "Patients are getting older, bioprostheses are getting better, and valve repair techniques are improving."
Dr. Harry R. Buller took issue with Dr. Van de Werf's blanket prediction that the factor Xa inhibitors will prove similarly unsafe and ineffective in patients with mechanical valves, as was dabigatran.
"We still need to find that out. One molecule of factor Xa generates about 1,000 thrombin molecules, so inhibiting coagulation at the level of Xa might actually be effective. What you see with dabigatran is inhibition at the very last stages of the whole coagulation cascade, when there are millions and millions of molecules of thrombin. That's quite difficult. If you do it higher up in the cascade it might work," argued Dr. Buller, chairman of the department of vascular medicine at the Academic Medical Center, Amsterdam.
"Would you take that risk?" challenged Dr. Van de Werf.
"I know there are people taking that risk. I love studies; I would study it," Dr. Buller replied.
Session cochair Dr. Keith A.A. Fox of the University of Edinburgh posed a provocative question to Dr. Van de Werf: "Would you extrapolate these RE-ALIGN data to a patient in atrial fibrillation with a bioprosthetic valve?"
"It's a difficult question," Dr. Van de Werf replied. "In principle, you could give one of the novel anticoagulants approved for atrial fibrillation, including dabigatran, because it is assumed there is no indication for oral anticoagulation for the bioprosthetic valve. I'm not saying you necessarily should do that. If the patient is doing well on warfarin, I don't see the need to switch to one of the new anticoagulants."
RE-ALIGN was supported by Boehringer Ingelheim. Dr. Van de Werf reported receiving research grants and speakers' fees from the company. Dr. Buller, Dr. Fox, and Dr. Vahanian reported no relevant financial interests.
AMSTERDAM -- The complete failure of the oral direct thrombin inhibitor dabigatran in the RE-ALIGN trial spells big trouble for efforts to use the novel oral factor Xa inhibitors for anticoagulation in patients with a mechanical heart valve, RE-ALIGN steering committee cochair Dr. Frans Van de Werf cautioned at the annual congress of the European Society of Cardiology.
"It would not be wise for a cardiologist to prescribe these agents for a patient with a mechanical heart valve," declared Dr. Van de Werf, professor and chairman of the department of cardiovascular medicine at the Catholic University at Leuven (Belgium).
Dabigatran (Pradaxa) is approved as a more effective and convenient alternative to warfarin for prevention of stroke and other systemic embolism in patients with nonvalvular atrial fibrillation. RE-ALIGN (Randomized, Phase II Study to Evaluate the Safety and Pharmacokinetics of Oral Dabigatran Etexilate in Patients After Heart Valve Replacement) was an effort to expand the drug's indications to include prevention of thromboembolic events in patients with mechanical heart valves.
The study was halted prematurely late last year when an interim analysis showed that dabigatran was both less safe and less protective than standard warfarin therapy. Shortly afterward, the Food and Drug Administration and the European regulatory agency warned that dabigatran is contraindicated in patients with mechanical heart valves. Dr. Van de Werf's report in Amsterdam, however, was the first presentation of the actual data.
RE-ALIGN was stopped after 252 patients with mechanical heart valves had been randomized 2:1 to dabigatran or warfarin. Dabigatran dosing was adjusted on the basis of creatinine clearance to achieve a trough plasma level of at least 50 ng/mL.
The study halt came in response to an interim analysis showing a 5% stroke rate in the dabigatran group, compared with 0% with warfarin. This was accompanied by a 4% rate of major bleeding with dabigatran, compared with 2% with warfarin, and a 27% rate of any bleeding with dabigatran versus 12% with warfarin. All dabigatran-treated patients with major bleeding had pericardial bleeding and were in the subgroup of participants who started on the direct thrombin inhibitor within 7 days post surgery.
Other unwelcome outcomes in the interim analysis included a 2% MI rate and 3% rate of valve thrombosis without symptoms in the dabigatran group, versus no such events in the warfarin arm.
"Overall, this study was clearly negative: more thrombotic events and more bleeding complications in spite of adjusting the dose of dabigatran based upon renal function," Dr. Van de Werf observed.
Simultaneous with his presentation of the RE-ALIGN findings at the ESC congress in Amsterdam, the results were published online (N. Engl. J. Med. 2013 Sept. 1 [doi:10.1056/NEJMoa1300615]).
In an accompanying editorial, Dr. Elaine M. Hylek of Boston University said the most likely explanation for the disappointing results in RE-ALIGN was that blood levels of dabigatran were inadequate (N. Engl. J. Med. 2013 Sept. 1 [doi:10.1056/NEJMe1310399]).
Dr. Van de Werf, however, rejected that as unlikely. Given that there were excesses of both thromboembolic and bleeding events, other dosing regimens would surely have sent rates of one type of complication or the other even higher, he argued.
Instead, he and his coinvestigators believe that the most likely explanation for the negative results lies in the differences in the mechanisms of action of dabigatran and warfarin. Dabigatran acts near the tail end of the coagulation cascade, on thrombin. In patients with a mechanical heart valve, thrombin is generated by exposure of blood to the artificial surface of the valve leaflets, activating what is known as the contact pathway of coagulation. Thrombin also is generated by the release of tissue factor from tissues injured during the valve surgery. Warfarin acts at both the contact pathway and tissue injury levels; dabigatran doesn't address either.
The novel oral factor Xa inhibitors don't act at the contact pathway and tissue injury levels, either, which is why Dr. Van de Werf cautioned against their off-label use in patients with a mechanical valve.
Discussant Dr. Alec Vahanian said that he "fully agrees" with Dr. Van de Werf's interpretation that the negative results most likely stemmed from dabigatran's absence of efficacy on the contact pathway.
"It sounds from this study that there is no future for dabigatran with this indication. We should not give this agent to a patient with a mechanical prosthesis, and personally I would extend that further and say we should not give this agent to patients with a bioprosthesis or other high-risk patients with severe valve disease. We don't have the evidence for that yet," said Dr. Vahanian, head of cardiology at Bichat Hospital, Paris.
Patients with a mechanical heart valve constitute a challenging population at extreme thromboembolic risk, but Dr. Vahanian offered a comforting prediction: "We are nearing the end of the era of mechanical prosthetic valves," he declared. "Patients are getting older, bioprostheses are getting better, and valve repair techniques are improving."
Dr. Harry R. Buller took issue with Dr. Van de Werf's blanket prediction that the factor Xa inhibitors will prove similarly unsafe and ineffective in patients with mechanical valves, as was dabigatran.
"We still need to find that out. One molecule of factor Xa generates about 1,000 thrombin molecules, so inhibiting coagulation at the level of Xa might actually be effective. What you see with dabigatran is inhibition at the very last stages of the whole coagulation cascade, when there are millions and millions of molecules of thrombin. That's quite difficult. If you do it higher up in the cascade it might work," argued Dr. Buller, chairman of the department of vascular medicine at the Academic Medical Center, Amsterdam.
"Would you take that risk?" challenged Dr. Van de Werf.
"I know there are people taking that risk. I love studies; I would study it," Dr. Buller replied.
Session cochair Dr. Keith A.A. Fox of the University of Edinburgh posed a provocative question to Dr. Van de Werf: "Would you extrapolate these RE-ALIGN data to a patient in atrial fibrillation with a bioprosthetic valve?"
"It's a difficult question," Dr. Van de Werf replied. "In principle, you could give one of the novel anticoagulants approved for atrial fibrillation, including dabigatran, because it is assumed there is no indication for oral anticoagulation for the bioprosthetic valve. I'm not saying you necessarily should do that. If the patient is doing well on warfarin, I don't see the need to switch to one of the new anticoagulants."
RE-ALIGN was supported by Boehringer Ingelheim. Dr. Van de Werf reported receiving research grants and speakers' fees from the company. Dr. Buller, Dr. Fox, and Dr. Vahanian reported no relevant financial interests.
AMSTERDAM -- The complete failure of the oral direct thrombin inhibitor dabigatran in the RE-ALIGN trial spells big trouble for efforts to use the novel oral factor Xa inhibitors for anticoagulation in patients with a mechanical heart valve, RE-ALIGN steering committee cochair Dr. Frans Van de Werf cautioned at the annual congress of the European Society of Cardiology.
"It would not be wise for a cardiologist to prescribe these agents for a patient with a mechanical heart valve," declared Dr. Van de Werf, professor and chairman of the department of cardiovascular medicine at the Catholic University at Leuven (Belgium).
Dabigatran (Pradaxa) is approved as a more effective and convenient alternative to warfarin for prevention of stroke and other systemic embolism in patients with nonvalvular atrial fibrillation. RE-ALIGN (Randomized, Phase II Study to Evaluate the Safety and Pharmacokinetics of Oral Dabigatran Etexilate in Patients After Heart Valve Replacement) was an effort to expand the drug's indications to include prevention of thromboembolic events in patients with mechanical heart valves.
The study was halted prematurely late last year when an interim analysis showed that dabigatran was both less safe and less protective than standard warfarin therapy. Shortly afterward, the Food and Drug Administration and the European regulatory agency warned that dabigatran is contraindicated in patients with mechanical heart valves. Dr. Van de Werf's report in Amsterdam, however, was the first presentation of the actual data.
RE-ALIGN was stopped after 252 patients with mechanical heart valves had been randomized 2:1 to dabigatran or warfarin. Dabigatran dosing was adjusted on the basis of creatinine clearance to achieve a trough plasma level of at least 50 ng/mL.
The study halt came in response to an interim analysis showing a 5% stroke rate in the dabigatran group, compared with 0% with warfarin. This was accompanied by a 4% rate of major bleeding with dabigatran, compared with 2% with warfarin, and a 27% rate of any bleeding with dabigatran versus 12% with warfarin. All dabigatran-treated patients with major bleeding had pericardial bleeding and were in the subgroup of participants who started on the direct thrombin inhibitor within 7 days post surgery.
Other unwelcome outcomes in the interim analysis included a 2% MI rate and 3% rate of valve thrombosis without symptoms in the dabigatran group, versus no such events in the warfarin arm.
"Overall, this study was clearly negative: more thrombotic events and more bleeding complications in spite of adjusting the dose of dabigatran based upon renal function," Dr. Van de Werf observed.
Simultaneous with his presentation of the RE-ALIGN findings at the ESC congress in Amsterdam, the results were published online (N. Engl. J. Med. 2013 Sept. 1 [doi:10.1056/NEJMoa1300615]).
In an accompanying editorial, Dr. Elaine M. Hylek of Boston University said the most likely explanation for the disappointing results in RE-ALIGN was that blood levels of dabigatran were inadequate (N. Engl. J. Med. 2013 Sept. 1 [doi:10.1056/NEJMe1310399]).
Dr. Van de Werf, however, rejected that as unlikely. Given that there were excesses of both thromboembolic and bleeding events, other dosing regimens would surely have sent rates of one type of complication or the other even higher, he argued.
Instead, he and his coinvestigators believe that the most likely explanation for the negative results lies in the differences in the mechanisms of action of dabigatran and warfarin. Dabigatran acts near the tail end of the coagulation cascade, on thrombin. In patients with a mechanical heart valve, thrombin is generated by exposure of blood to the artificial surface of the valve leaflets, activating what is known as the contact pathway of coagulation. Thrombin also is generated by the release of tissue factor from tissues injured during the valve surgery. Warfarin acts at both the contact pathway and tissue injury levels; dabigatran doesn't address either.
The novel oral factor Xa inhibitors don't act at the contact pathway and tissue injury levels, either, which is why Dr. Van de Werf cautioned against their off-label use in patients with a mechanical valve.
Discussant Dr. Alec Vahanian said that he "fully agrees" with Dr. Van de Werf's interpretation that the negative results most likely stemmed from dabigatran's absence of efficacy on the contact pathway.
"It sounds from this study that there is no future for dabigatran with this indication. We should not give this agent to a patient with a mechanical prosthesis, and personally I would extend that further and say we should not give this agent to patients with a bioprosthesis or other high-risk patients with severe valve disease. We don't have the evidence for that yet," said Dr. Vahanian, head of cardiology at Bichat Hospital, Paris.
Patients with a mechanical heart valve constitute a challenging population at extreme thromboembolic risk, but Dr. Vahanian offered a comforting prediction: "We are nearing the end of the era of mechanical prosthetic valves," he declared. "Patients are getting older, bioprostheses are getting better, and valve repair techniques are improving."
Dr. Harry R. Buller took issue with Dr. Van de Werf's blanket prediction that the factor Xa inhibitors will prove similarly unsafe and ineffective in patients with mechanical valves, as was dabigatran.
"We still need to find that out. One molecule of factor Xa generates about 1,000 thrombin molecules, so inhibiting coagulation at the level of Xa might actually be effective. What you see with dabigatran is inhibition at the very last stages of the whole coagulation cascade, when there are millions and millions of molecules of thrombin. That's quite difficult. If you do it higher up in the cascade it might work," argued Dr. Buller, chairman of the department of vascular medicine at the Academic Medical Center, Amsterdam.
"Would you take that risk?" challenged Dr. Van de Werf.
"I know there are people taking that risk. I love studies; I would study it," Dr. Buller replied.
Session cochair Dr. Keith A.A. Fox of the University of Edinburgh posed a provocative question to Dr. Van de Werf: "Would you extrapolate these RE-ALIGN data to a patient in atrial fibrillation with a bioprosthetic valve?"
"It's a difficult question," Dr. Van de Werf replied. "In principle, you could give one of the novel anticoagulants approved for atrial fibrillation, including dabigatran, because it is assumed there is no indication for oral anticoagulation for the bioprosthetic valve. I'm not saying you necessarily should do that. If the patient is doing well on warfarin, I don't see the need to switch to one of the new anticoagulants."
RE-ALIGN was supported by Boehringer Ingelheim. Dr. Van de Werf reported receiving research grants and speakers' fees from the company. Dr. Buller, Dr. Fox, and Dr. Vahanian reported no relevant financial interests.
Docs: Major responsibility for cost control not ours
When it comes to reducing health costs, physicians believe burden of responsibility lies primarily with plaintiffs attorneys, followed by insurers, hospitals, drug and device makers, patients, and, lastly, themselves. Those conclusions are based on 2,438 responses from some 3,900 physicians randomly surveyed in 2012. Dr. Jon C. Tilburt of the Mayo Clinic, Rochester, Minn., and his colleagues reported their findings.
When asked whether individual physicians should have a major responsibility in reducing health costs, 36% of respondents said yes. Sixty percent said that trial lawyers bore the major burden, with health insurers coming in a close second.
More than half said that drug and device companies, hospitals and health systems, and patients also should have major responsibility for cost containment. A total of 44% said the government had that responsibility (JAMA 2013;310:380-8).
Physicians also were asked about their enthusiasm for various cost-control strategies and to examine their own role in cost containment by assessing their knowledge of prices of procedures and tests and their desire to personally curb costs in their practice. The authors asked about and analyzed potential barriers to physicians becoming more cost conscious, as well.
Doctors were very enthusiastic about improving the quality and efficiency of care, primarily through promoting continuity of care and going after fraud and abuse. Expanding access to preventive care was also warmly received. Physicians were also enthusiastic about limiting access to expensive treatments that had shown little benefit, using cost-effectiveness data to choose a therapy, and promoting head-to-head trials of competing therapies.
Just over half of respondents said that cutting pay for the highest-paid specialists should be embraced.
Eliminating fee for service altogether was rejected by 70% of respondents. Ninety percent said that they weren’t enthusiastic about letting the Medicare Sustainable Growth Rate cuts take effect. Two-thirds said that bundled pay and penalties for readmissions – both cost-control keystones advanced by the Obama administration – were not attractive.
Not surprisingly, increasing use of electronic health records also got a strong negative response, with 29% saying they were "not enthusiastic."
When it came to their own practice, 76% said they were aware of the costs of treatments or tests they recommended, and 84% said that cost is important whether a patient pays out of pocket or not.
When it comes to individual physicians’ responsibility for reducing health costs, the responses were very mixed. The survey participants largely agreed that "trying to contain costs is the responsibility of every physician" (85%) and that physicians should take a more prominent role in eliminating unnecessary tests (89%). But by almost the same percentages, physicians also said that they should be devoted to their individual patients, even if a test or therapy was expensive, and that they should not deny services to their patients because someone else might need it more.
"This apparent inconsistency may reflect inherent tensions in professional roles to serve patients individually and society as a whole," Dr. Tilburt and colleagues wrote.
Finally, physicians overwhelmingly said that fear of malpractice had substantially decreased their enjoyment of practicing medicine. The authors rated that fear as a barrier to cost-conscious practice.
They also found that 43% of physicians admitted they ordered more tests when they did not know the patient as well. Half said that being more cost conscious was the right thing to do, but large numbers said that it might not make a difference or could make things worse. A total of 40% said it would not limit unreasonable patient demands, and 28% said it could erode patients’ trust.
Dr. Tilburt and his colleagues pointed out that the findings should be viewed with caution in part because it could not fully reflect the opinions of all American physicians. Further, opinions could be in flux, given how much things are changing.
The study was funded by the Greenwall Foundation and the Mayo Clinic. The authors reported having no financial conflicts.
If there were ever an "all-hands-on-deck" moment in the history of health care, that moment is now. The findings of this study suggest that physician do not yet have the mentality this historical moment demands. Indeed, this survey suggests that in the face of this new and uncertain moment in the reform of the health care system, physicians are lapsing into the well-known, cautious, instinctual approaches humans adopt whenever confronted by uncertainty: Blame others and persevere with "business as usual."
Physicians have moved beyond denying that health care costs are a problem. Yet, they are not quite willing to accept physicians’ primary responsibility and take action. The study findings suggest that physicians are ambivalent; they reject transformative solutions, such as eliminating fee-for-service or bundled payments, which address the seriousness of the cost problem.
This study by Tilburt et al. indicates that the medical profession is not there yet – that many physicians would prefer to sit on the sidelines while other actors in the health care system do the real work of reform.
This could marginalize and demote physicians.
Dr. Ezekiel J. Emanuel is an ethicist at the University of Pennsylvania, Philadelphia. He reported no related conflicts. These remarks were taken from an editorial accompanying Dr. Tilburt’s study (JAMA 2013;310:374-5).
If there were ever an "all-hands-on-deck" moment in the history of health care, that moment is now. The findings of this study suggest that physician do not yet have the mentality this historical moment demands. Indeed, this survey suggests that in the face of this new and uncertain moment in the reform of the health care system, physicians are lapsing into the well-known, cautious, instinctual approaches humans adopt whenever confronted by uncertainty: Blame others and persevere with "business as usual."
Physicians have moved beyond denying that health care costs are a problem. Yet, they are not quite willing to accept physicians’ primary responsibility and take action. The study findings suggest that physicians are ambivalent; they reject transformative solutions, such as eliminating fee-for-service or bundled payments, which address the seriousness of the cost problem.
This study by Tilburt et al. indicates that the medical profession is not there yet – that many physicians would prefer to sit on the sidelines while other actors in the health care system do the real work of reform.
This could marginalize and demote physicians.
Dr. Ezekiel J. Emanuel is an ethicist at the University of Pennsylvania, Philadelphia. He reported no related conflicts. These remarks were taken from an editorial accompanying Dr. Tilburt’s study (JAMA 2013;310:374-5).
If there were ever an "all-hands-on-deck" moment in the history of health care, that moment is now. The findings of this study suggest that physician do not yet have the mentality this historical moment demands. Indeed, this survey suggests that in the face of this new and uncertain moment in the reform of the health care system, physicians are lapsing into the well-known, cautious, instinctual approaches humans adopt whenever confronted by uncertainty: Blame others and persevere with "business as usual."
Physicians have moved beyond denying that health care costs are a problem. Yet, they are not quite willing to accept physicians’ primary responsibility and take action. The study findings suggest that physicians are ambivalent; they reject transformative solutions, such as eliminating fee-for-service or bundled payments, which address the seriousness of the cost problem.
This study by Tilburt et al. indicates that the medical profession is not there yet – that many physicians would prefer to sit on the sidelines while other actors in the health care system do the real work of reform.
This could marginalize and demote physicians.
Dr. Ezekiel J. Emanuel is an ethicist at the University of Pennsylvania, Philadelphia. He reported no related conflicts. These remarks were taken from an editorial accompanying Dr. Tilburt’s study (JAMA 2013;310:374-5).
When it comes to reducing health costs, physicians believe burden of responsibility lies primarily with plaintiffs attorneys, followed by insurers, hospitals, drug and device makers, patients, and, lastly, themselves. Those conclusions are based on 2,438 responses from some 3,900 physicians randomly surveyed in 2012. Dr. Jon C. Tilburt of the Mayo Clinic, Rochester, Minn., and his colleagues reported their findings.
When asked whether individual physicians should have a major responsibility in reducing health costs, 36% of respondents said yes. Sixty percent said that trial lawyers bore the major burden, with health insurers coming in a close second.
More than half said that drug and device companies, hospitals and health systems, and patients also should have major responsibility for cost containment. A total of 44% said the government had that responsibility (JAMA 2013;310:380-8).
Physicians also were asked about their enthusiasm for various cost-control strategies and to examine their own role in cost containment by assessing their knowledge of prices of procedures and tests and their desire to personally curb costs in their practice. The authors asked about and analyzed potential barriers to physicians becoming more cost conscious, as well.
Doctors were very enthusiastic about improving the quality and efficiency of care, primarily through promoting continuity of care and going after fraud and abuse. Expanding access to preventive care was also warmly received. Physicians were also enthusiastic about limiting access to expensive treatments that had shown little benefit, using cost-effectiveness data to choose a therapy, and promoting head-to-head trials of competing therapies.
Just over half of respondents said that cutting pay for the highest-paid specialists should be embraced.
Eliminating fee for service altogether was rejected by 70% of respondents. Ninety percent said that they weren’t enthusiastic about letting the Medicare Sustainable Growth Rate cuts take effect. Two-thirds said that bundled pay and penalties for readmissions – both cost-control keystones advanced by the Obama administration – were not attractive.
Not surprisingly, increasing use of electronic health records also got a strong negative response, with 29% saying they were "not enthusiastic."
When it came to their own practice, 76% said they were aware of the costs of treatments or tests they recommended, and 84% said that cost is important whether a patient pays out of pocket or not.
When it comes to individual physicians’ responsibility for reducing health costs, the responses were very mixed. The survey participants largely agreed that "trying to contain costs is the responsibility of every physician" (85%) and that physicians should take a more prominent role in eliminating unnecessary tests (89%). But by almost the same percentages, physicians also said that they should be devoted to their individual patients, even if a test or therapy was expensive, and that they should not deny services to their patients because someone else might need it more.
"This apparent inconsistency may reflect inherent tensions in professional roles to serve patients individually and society as a whole," Dr. Tilburt and colleagues wrote.
Finally, physicians overwhelmingly said that fear of malpractice had substantially decreased their enjoyment of practicing medicine. The authors rated that fear as a barrier to cost-conscious practice.
They also found that 43% of physicians admitted they ordered more tests when they did not know the patient as well. Half said that being more cost conscious was the right thing to do, but large numbers said that it might not make a difference or could make things worse. A total of 40% said it would not limit unreasonable patient demands, and 28% said it could erode patients’ trust.
Dr. Tilburt and his colleagues pointed out that the findings should be viewed with caution in part because it could not fully reflect the opinions of all American physicians. Further, opinions could be in flux, given how much things are changing.
The study was funded by the Greenwall Foundation and the Mayo Clinic. The authors reported having no financial conflicts.
When it comes to reducing health costs, physicians believe burden of responsibility lies primarily with plaintiffs attorneys, followed by insurers, hospitals, drug and device makers, patients, and, lastly, themselves. Those conclusions are based on 2,438 responses from some 3,900 physicians randomly surveyed in 2012. Dr. Jon C. Tilburt of the Mayo Clinic, Rochester, Minn., and his colleagues reported their findings.
When asked whether individual physicians should have a major responsibility in reducing health costs, 36% of respondents said yes. Sixty percent said that trial lawyers bore the major burden, with health insurers coming in a close second.
More than half said that drug and device companies, hospitals and health systems, and patients also should have major responsibility for cost containment. A total of 44% said the government had that responsibility (JAMA 2013;310:380-8).
Physicians also were asked about their enthusiasm for various cost-control strategies and to examine their own role in cost containment by assessing their knowledge of prices of procedures and tests and their desire to personally curb costs in their practice. The authors asked about and analyzed potential barriers to physicians becoming more cost conscious, as well.
Doctors were very enthusiastic about improving the quality and efficiency of care, primarily through promoting continuity of care and going after fraud and abuse. Expanding access to preventive care was also warmly received. Physicians were also enthusiastic about limiting access to expensive treatments that had shown little benefit, using cost-effectiveness data to choose a therapy, and promoting head-to-head trials of competing therapies.
Just over half of respondents said that cutting pay for the highest-paid specialists should be embraced.
Eliminating fee for service altogether was rejected by 70% of respondents. Ninety percent said that they weren’t enthusiastic about letting the Medicare Sustainable Growth Rate cuts take effect. Two-thirds said that bundled pay and penalties for readmissions – both cost-control keystones advanced by the Obama administration – were not attractive.
Not surprisingly, increasing use of electronic health records also got a strong negative response, with 29% saying they were "not enthusiastic."
When it came to their own practice, 76% said they were aware of the costs of treatments or tests they recommended, and 84% said that cost is important whether a patient pays out of pocket or not.
When it comes to individual physicians’ responsibility for reducing health costs, the responses were very mixed. The survey participants largely agreed that "trying to contain costs is the responsibility of every physician" (85%) and that physicians should take a more prominent role in eliminating unnecessary tests (89%). But by almost the same percentages, physicians also said that they should be devoted to their individual patients, even if a test or therapy was expensive, and that they should not deny services to their patients because someone else might need it more.
"This apparent inconsistency may reflect inherent tensions in professional roles to serve patients individually and society as a whole," Dr. Tilburt and colleagues wrote.
Finally, physicians overwhelmingly said that fear of malpractice had substantially decreased their enjoyment of practicing medicine. The authors rated that fear as a barrier to cost-conscious practice.
They also found that 43% of physicians admitted they ordered more tests when they did not know the patient as well. Half said that being more cost conscious was the right thing to do, but large numbers said that it might not make a difference or could make things worse. A total of 40% said it would not limit unreasonable patient demands, and 28% said it could erode patients’ trust.
Dr. Tilburt and his colleagues pointed out that the findings should be viewed with caution in part because it could not fully reflect the opinions of all American physicians. Further, opinions could be in flux, given how much things are changing.
The study was funded by the Greenwall Foundation and the Mayo Clinic. The authors reported having no financial conflicts.
Medicare ups readmission penalties, changes rules
Beginning in October 2013, Medicare will double the penalties for preventable hospital readmissions and will change the rules for determining when to admit patients or to place them in observation status.
Under the fiscal year 2014 Hospital Inpatient Prospective Payment System rule, Medicare is raising the maximum penalties for preventable, unplanned hospital readmissions from 1% of base operating payments to 2%, starting in October 2013. Details of the payment hike, which is mandated by the Affordable Care Act, were announced Aug. 2 and were published Aug. 19 in the Federal Register.
The changes impact the Hospital Readmissions Reduction program, which was originally launched in October 2012.
That program penalizes hospitals with excess 30-day Medicare readmissions for acute myocardial infarction, heart failure, and pneumonia.
The new payment rule exempts more types of planned readmissions from the program. It also expands the program to include total hip and knee arthroplasty and acute chronic obstructive pulmonary disease, starting in October 2014.
In response to concerns from hospitals and patients, the hospital inpatient payment rule also provides greater guidance about which patients are appropriate for admission to the hospital and thus covered under Part A, and which patients should be considered for observation status, which is covered under Medicare Part B.
The criteria now will be based on the amount of time the physician expects the patient to spend as an inpatient.
Starting in October 2013, Medicare contractors will assume that a hospital stay is eligible for Part A payment if the physician expects the patient to stay as an inpatient in the hospital for at least 2 midnights. The inpatient stay is not triggered until the physician formally admits the patient.
However, officials at the Centers for Medicare and Medicaid Services said physicians may consider the patient’s time in observation, the emergency department, the operating room, and other in-hospital treatment areas when deciding if it is appropriate to expect the patient to stay for at least 2 midnights.
"This rule helps improve hospital care and establishes clearer guidance to hospitals for when we will consider inpatient care to be appropriate so the system works better for patients and providers," CMS Administrator Marilyn Tavenner said in a statement.
The CMS estimates that the policy change will increase spending by approximately $220 million because of increases in inpatient admissions, so the agency is reducing hospital payments accordingly.
Medicare’s changed admission policy is already drawing some critics.
The new criteria will do nothing to protect hospitals from "burdensome" audits and appeals, and it will require physicians to have a "sixth sense and predict the future treatment needs for patients," warned Blair Childs, senior vice president of public affairs for Premier, an alliance of hospitals and other health facilities.
"Moreover, these changes add insult to injury, imposing an associated 0.2% payment reduction to offset what CMS believes will be an increased inpatient volume," Mr. Childs said in a statement.
"We expect that this will result in even more confusion around what constitutes an appropriate inpatient hospital admission, all while cutting payments for following CMS’s rules."
Beginning in October 2013, Medicare will double the penalties for preventable hospital readmissions and will change the rules for determining when to admit patients or to place them in observation status.
Under the fiscal year 2014 Hospital Inpatient Prospective Payment System rule, Medicare is raising the maximum penalties for preventable, unplanned hospital readmissions from 1% of base operating payments to 2%, starting in October 2013. Details of the payment hike, which is mandated by the Affordable Care Act, were announced Aug. 2 and were published Aug. 19 in the Federal Register.
The changes impact the Hospital Readmissions Reduction program, which was originally launched in October 2012.
That program penalizes hospitals with excess 30-day Medicare readmissions for acute myocardial infarction, heart failure, and pneumonia.
The new payment rule exempts more types of planned readmissions from the program. It also expands the program to include total hip and knee arthroplasty and acute chronic obstructive pulmonary disease, starting in October 2014.
In response to concerns from hospitals and patients, the hospital inpatient payment rule also provides greater guidance about which patients are appropriate for admission to the hospital and thus covered under Part A, and which patients should be considered for observation status, which is covered under Medicare Part B.
The criteria now will be based on the amount of time the physician expects the patient to spend as an inpatient.
Starting in October 2013, Medicare contractors will assume that a hospital stay is eligible for Part A payment if the physician expects the patient to stay as an inpatient in the hospital for at least 2 midnights. The inpatient stay is not triggered until the physician formally admits the patient.
However, officials at the Centers for Medicare and Medicaid Services said physicians may consider the patient’s time in observation, the emergency department, the operating room, and other in-hospital treatment areas when deciding if it is appropriate to expect the patient to stay for at least 2 midnights.
"This rule helps improve hospital care and establishes clearer guidance to hospitals for when we will consider inpatient care to be appropriate so the system works better for patients and providers," CMS Administrator Marilyn Tavenner said in a statement.
The CMS estimates that the policy change will increase spending by approximately $220 million because of increases in inpatient admissions, so the agency is reducing hospital payments accordingly.
Medicare’s changed admission policy is already drawing some critics.
The new criteria will do nothing to protect hospitals from "burdensome" audits and appeals, and it will require physicians to have a "sixth sense and predict the future treatment needs for patients," warned Blair Childs, senior vice president of public affairs for Premier, an alliance of hospitals and other health facilities.
"Moreover, these changes add insult to injury, imposing an associated 0.2% payment reduction to offset what CMS believes will be an increased inpatient volume," Mr. Childs said in a statement.
"We expect that this will result in even more confusion around what constitutes an appropriate inpatient hospital admission, all while cutting payments for following CMS’s rules."
Beginning in October 2013, Medicare will double the penalties for preventable hospital readmissions and will change the rules for determining when to admit patients or to place them in observation status.
Under the fiscal year 2014 Hospital Inpatient Prospective Payment System rule, Medicare is raising the maximum penalties for preventable, unplanned hospital readmissions from 1% of base operating payments to 2%, starting in October 2013. Details of the payment hike, which is mandated by the Affordable Care Act, were announced Aug. 2 and were published Aug. 19 in the Federal Register.
The changes impact the Hospital Readmissions Reduction program, which was originally launched in October 2012.
That program penalizes hospitals with excess 30-day Medicare readmissions for acute myocardial infarction, heart failure, and pneumonia.
The new payment rule exempts more types of planned readmissions from the program. It also expands the program to include total hip and knee arthroplasty and acute chronic obstructive pulmonary disease, starting in October 2014.
In response to concerns from hospitals and patients, the hospital inpatient payment rule also provides greater guidance about which patients are appropriate for admission to the hospital and thus covered under Part A, and which patients should be considered for observation status, which is covered under Medicare Part B.
The criteria now will be based on the amount of time the physician expects the patient to spend as an inpatient.
Starting in October 2013, Medicare contractors will assume that a hospital stay is eligible for Part A payment if the physician expects the patient to stay as an inpatient in the hospital for at least 2 midnights. The inpatient stay is not triggered until the physician formally admits the patient.
However, officials at the Centers for Medicare and Medicaid Services said physicians may consider the patient’s time in observation, the emergency department, the operating room, and other in-hospital treatment areas when deciding if it is appropriate to expect the patient to stay for at least 2 midnights.
"This rule helps improve hospital care and establishes clearer guidance to hospitals for when we will consider inpatient care to be appropriate so the system works better for patients and providers," CMS Administrator Marilyn Tavenner said in a statement.
The CMS estimates that the policy change will increase spending by approximately $220 million because of increases in inpatient admissions, so the agency is reducing hospital payments accordingly.
Medicare’s changed admission policy is already drawing some critics.
The new criteria will do nothing to protect hospitals from "burdensome" audits and appeals, and it will require physicians to have a "sixth sense and predict the future treatment needs for patients," warned Blair Childs, senior vice president of public affairs for Premier, an alliance of hospitals and other health facilities.
"Moreover, these changes add insult to injury, imposing an associated 0.2% payment reduction to offset what CMS believes will be an increased inpatient volume," Mr. Childs said in a statement.
"We expect that this will result in even more confusion around what constitutes an appropriate inpatient hospital admission, all while cutting payments for following CMS’s rules."
Mitral replacement may grow with infant
NEW YORK – Physicians at Boston Children’s Hospital replaced the mitral valves of eight infants with irreparable mitral valve disease with a valve that offers the opportunity of sequential expansion as the child grows, according to Dr. Sitaram M. Emani. The results were presented at the 2013 Mitral Valve Conclave earlier this year.
"The Melody valve retains its competence if you expand it before putting it in. We asked whether the valve retains the ability to maintain competence even if expansion is performed after implantation as the patient grows," said Dr. Emani, a pediatric cardiac surgeon at Boston Children’s Hospital.
According to Dr. Emani, the current options for infants with damaged mitral valves that are beyond repair are replacement with mechanical or bioprosthetic valves or the Ross mitral procedure. Perhaps the main disadvantage of these options is the lack of a prosthetic valve small enough for an infant, one that is less than 12 mm in diameter. Another problem is the possibility of stenosis developing as the child grows, since the diameters of the prosthetics are fixed. Other drawbacks are that supra-annular fixation is generally associated with poor outcomes and that annular fixation limits the ability to upsize at reoperation.
The Melody valve is an externally stented bovine jugular vein graft that was designed for transcatheter pulmonary valve replacement. In this study, the valve was inserted surgically. The valve maintains competence over a range of sizes up to 22 mm. Although this valve is not approved for use for mitral valve replacement, the hope of using such a prosthetic is that it can be enlarged in the catheterization laboratory as the child grows.
Dr. Emani did a retrospective study of his experience with the Melody valve for mitral valve replacement in eight infants less than 12 months of age. The median age at implantation was 6 months (range, 1-9 months). Four infants had an atrioventricular canal (AVC) defect and four had congenital mitral valve stenosis. Most of the children had two prior operations for mitral valve repair. The longest follow-up to date has been 2 years.
At a median follow-up of 8 months, regurgitation on the echocardiogram was considered to be mild or less in all patients. The median gradient was 3 mm Hg (range, 2-7 mm Hg) on the immediate postoperative echocardiogram. Three patients developed a mild paravalvular leak; one of these patients had undergone aggressive stent resection, a modification Dr. Emani does not recommend. One patient developed left ventricular outflow tract obstruction (LVOTO), which Dr. Emani attributed to the lack of distal stent fixation in this patient. Another patient with an AVC defect developed complete heart block.
One patient who died 3 days postoperatively had heterotaxy, severe mitral regurgitation, and prior ventricular failure on extracorporeal membrane oxygenation support. That patient had undergone valve implantation as a last resort.
Three patients underwent sequential expansion about 6 months after implantation. After valve expansion, the median balloon size was 12 mm, ranging from 12 to 16 mm. None of the patients developed worsening valvular function and all had relief of obstruction. Transcatheter intervention was used to correct a paravalvular leak in one patient and to treat a left ventricular outflow tract problem in another. None of the patients developed endocarditis or a strut fracture, "although I worry about strut fracture if aggressive stent resection and manipulation is performed," he said at the meeting, which was sponsored by the AATS.
Dr. Emani offered some procedural tips. First, the Melody valve must be optimized for surgical implantation in infants. The length of the valve must be reduced by trimming it to reduce the chance of LVOTO or pulmonary vein obstruction. He recommends sizing the valves by echocardiogram and fixating the distal stent to the inferior free wall of the ventricle.
He reported that friction of the stent against the annulus prevents leakage. Early on he used a pericardial cuff to anchor to the annulus, particularly in patients who had undergone failed AVC repair. He tries to preserve at least part of the anterior leaflet to facilitate suture placement and create a "stand-off" from the LVOTO.
Dr. Emani also advised limiting intraoperative dilation to no more than 1 mm greater than the measured annulus. "Try not to overdilate at implantation to avoid heart block, LVOTO, and coronary compression. The nice thing is you don’t have to decide then and there what size you want. You can go back to the cath lab and, under direct visualization with the coronary view, you can dilate it under more controlled circumstances.
"The hope is that we will be able to dilate these valves as the patients grow into adolescence. If we can dilate them up to 22 mm, hopefully we will decrease the number of repeat replacements, delay the time to reoperation, and perhaps modify our thresholds for tolerating significant disease after unsuccessful repairs."
Dr. Emani reported no disclosures.
NEW YORK – Physicians at Boston Children’s Hospital replaced the mitral valves of eight infants with irreparable mitral valve disease with a valve that offers the opportunity of sequential expansion as the child grows, according to Dr. Sitaram M. Emani. The results were presented at the 2013 Mitral Valve Conclave earlier this year.
"The Melody valve retains its competence if you expand it before putting it in. We asked whether the valve retains the ability to maintain competence even if expansion is performed after implantation as the patient grows," said Dr. Emani, a pediatric cardiac surgeon at Boston Children’s Hospital.
According to Dr. Emani, the current options for infants with damaged mitral valves that are beyond repair are replacement with mechanical or bioprosthetic valves or the Ross mitral procedure. Perhaps the main disadvantage of these options is the lack of a prosthetic valve small enough for an infant, one that is less than 12 mm in diameter. Another problem is the possibility of stenosis developing as the child grows, since the diameters of the prosthetics are fixed. Other drawbacks are that supra-annular fixation is generally associated with poor outcomes and that annular fixation limits the ability to upsize at reoperation.
The Melody valve is an externally stented bovine jugular vein graft that was designed for transcatheter pulmonary valve replacement. In this study, the valve was inserted surgically. The valve maintains competence over a range of sizes up to 22 mm. Although this valve is not approved for use for mitral valve replacement, the hope of using such a prosthetic is that it can be enlarged in the catheterization laboratory as the child grows.
Dr. Emani did a retrospective study of his experience with the Melody valve for mitral valve replacement in eight infants less than 12 months of age. The median age at implantation was 6 months (range, 1-9 months). Four infants had an atrioventricular canal (AVC) defect and four had congenital mitral valve stenosis. Most of the children had two prior operations for mitral valve repair. The longest follow-up to date has been 2 years.
At a median follow-up of 8 months, regurgitation on the echocardiogram was considered to be mild or less in all patients. The median gradient was 3 mm Hg (range, 2-7 mm Hg) on the immediate postoperative echocardiogram. Three patients developed a mild paravalvular leak; one of these patients had undergone aggressive stent resection, a modification Dr. Emani does not recommend. One patient developed left ventricular outflow tract obstruction (LVOTO), which Dr. Emani attributed to the lack of distal stent fixation in this patient. Another patient with an AVC defect developed complete heart block.
One patient who died 3 days postoperatively had heterotaxy, severe mitral regurgitation, and prior ventricular failure on extracorporeal membrane oxygenation support. That patient had undergone valve implantation as a last resort.
Three patients underwent sequential expansion about 6 months after implantation. After valve expansion, the median balloon size was 12 mm, ranging from 12 to 16 mm. None of the patients developed worsening valvular function and all had relief of obstruction. Transcatheter intervention was used to correct a paravalvular leak in one patient and to treat a left ventricular outflow tract problem in another. None of the patients developed endocarditis or a strut fracture, "although I worry about strut fracture if aggressive stent resection and manipulation is performed," he said at the meeting, which was sponsored by the AATS.
Dr. Emani offered some procedural tips. First, the Melody valve must be optimized for surgical implantation in infants. The length of the valve must be reduced by trimming it to reduce the chance of LVOTO or pulmonary vein obstruction. He recommends sizing the valves by echocardiogram and fixating the distal stent to the inferior free wall of the ventricle.
He reported that friction of the stent against the annulus prevents leakage. Early on he used a pericardial cuff to anchor to the annulus, particularly in patients who had undergone failed AVC repair. He tries to preserve at least part of the anterior leaflet to facilitate suture placement and create a "stand-off" from the LVOTO.
Dr. Emani also advised limiting intraoperative dilation to no more than 1 mm greater than the measured annulus. "Try not to overdilate at implantation to avoid heart block, LVOTO, and coronary compression. The nice thing is you don’t have to decide then and there what size you want. You can go back to the cath lab and, under direct visualization with the coronary view, you can dilate it under more controlled circumstances.
"The hope is that we will be able to dilate these valves as the patients grow into adolescence. If we can dilate them up to 22 mm, hopefully we will decrease the number of repeat replacements, delay the time to reoperation, and perhaps modify our thresholds for tolerating significant disease after unsuccessful repairs."
Dr. Emani reported no disclosures.
NEW YORK – Physicians at Boston Children’s Hospital replaced the mitral valves of eight infants with irreparable mitral valve disease with a valve that offers the opportunity of sequential expansion as the child grows, according to Dr. Sitaram M. Emani. The results were presented at the 2013 Mitral Valve Conclave earlier this year.
"The Melody valve retains its competence if you expand it before putting it in. We asked whether the valve retains the ability to maintain competence even if expansion is performed after implantation as the patient grows," said Dr. Emani, a pediatric cardiac surgeon at Boston Children’s Hospital.
According to Dr. Emani, the current options for infants with damaged mitral valves that are beyond repair are replacement with mechanical or bioprosthetic valves or the Ross mitral procedure. Perhaps the main disadvantage of these options is the lack of a prosthetic valve small enough for an infant, one that is less than 12 mm in diameter. Another problem is the possibility of stenosis developing as the child grows, since the diameters of the prosthetics are fixed. Other drawbacks are that supra-annular fixation is generally associated with poor outcomes and that annular fixation limits the ability to upsize at reoperation.
The Melody valve is an externally stented bovine jugular vein graft that was designed for transcatheter pulmonary valve replacement. In this study, the valve was inserted surgically. The valve maintains competence over a range of sizes up to 22 mm. Although this valve is not approved for use for mitral valve replacement, the hope of using such a prosthetic is that it can be enlarged in the catheterization laboratory as the child grows.
Dr. Emani did a retrospective study of his experience with the Melody valve for mitral valve replacement in eight infants less than 12 months of age. The median age at implantation was 6 months (range, 1-9 months). Four infants had an atrioventricular canal (AVC) defect and four had congenital mitral valve stenosis. Most of the children had two prior operations for mitral valve repair. The longest follow-up to date has been 2 years.
At a median follow-up of 8 months, regurgitation on the echocardiogram was considered to be mild or less in all patients. The median gradient was 3 mm Hg (range, 2-7 mm Hg) on the immediate postoperative echocardiogram. Three patients developed a mild paravalvular leak; one of these patients had undergone aggressive stent resection, a modification Dr. Emani does not recommend. One patient developed left ventricular outflow tract obstruction (LVOTO), which Dr. Emani attributed to the lack of distal stent fixation in this patient. Another patient with an AVC defect developed complete heart block.
One patient who died 3 days postoperatively had heterotaxy, severe mitral regurgitation, and prior ventricular failure on extracorporeal membrane oxygenation support. That patient had undergone valve implantation as a last resort.
Three patients underwent sequential expansion about 6 months after implantation. After valve expansion, the median balloon size was 12 mm, ranging from 12 to 16 mm. None of the patients developed worsening valvular function and all had relief of obstruction. Transcatheter intervention was used to correct a paravalvular leak in one patient and to treat a left ventricular outflow tract problem in another. None of the patients developed endocarditis or a strut fracture, "although I worry about strut fracture if aggressive stent resection and manipulation is performed," he said at the meeting, which was sponsored by the AATS.
Dr. Emani offered some procedural tips. First, the Melody valve must be optimized for surgical implantation in infants. The length of the valve must be reduced by trimming it to reduce the chance of LVOTO or pulmonary vein obstruction. He recommends sizing the valves by echocardiogram and fixating the distal stent to the inferior free wall of the ventricle.
He reported that friction of the stent against the annulus prevents leakage. Early on he used a pericardial cuff to anchor to the annulus, particularly in patients who had undergone failed AVC repair. He tries to preserve at least part of the anterior leaflet to facilitate suture placement and create a "stand-off" from the LVOTO.
Dr. Emani also advised limiting intraoperative dilation to no more than 1 mm greater than the measured annulus. "Try not to overdilate at implantation to avoid heart block, LVOTO, and coronary compression. The nice thing is you don’t have to decide then and there what size you want. You can go back to the cath lab and, under direct visualization with the coronary view, you can dilate it under more controlled circumstances.
"The hope is that we will be able to dilate these valves as the patients grow into adolescence. If we can dilate them up to 22 mm, hopefully we will decrease the number of repeat replacements, delay the time to reoperation, and perhaps modify our thresholds for tolerating significant disease after unsuccessful repairs."
Dr. Emani reported no disclosures.
USPSTF supports CT screening for lung cancer
Low-dose computed tomography reduces lung cancer mortality and all-cause mortality when used as a screening tool in asymptomatic adults at high risk for the disease, according to the results of a systematic review conducted for the U.S. Preventive Services Task Force.
In 2004, the USPSTF deemed the evidence insufficient for recommending for or against low-dose computed tomography (LDCT) for lung cancer screening in asymptomatic individuals, but the current review suggests screening has a definite benefit for most patients, Dr. Linda L. Humphrey, of Oregon Health and Science University and the Portland Veterans Affairs Medical Center, and her colleagues reported.
A draft recommendation based on the findings, published online in the Annals of Internal Medicine (2013 July 29 [doi: 10.7326/0003-4819-159-6-201309170-00690]), is available on the USPSTF website for comment.
The researchers reviewed the literature published between 2000 and May 2013 and identified four trials that reported findings on the efficacy of LDCT screening in patients with smoking exposure for both intervention and control groups. Three small trials showed varying degrees of benefit with screening, but were underpowered; one large trial – the National Lung Screening Trial (NLST) – showed a significant 20% reduction in lung cancer mortality among those screened, as well as a 6.7% reduction in all-cause mortality.
The randomized multicenter NLST compared annual LDCT scans with annual single-view posterior-anterior chest radiographs for 3 years in more than 53,000 current or former smokers aged 55-74 years with at least a 30–pack-year history of smoking (N. Engl. J. Med. 2013;368:1980-91). One cancer death was prevented for every 320 patients who completed one screening, and one death from any cause was prevented for every 219 patients screened in that study; the trial was stopped early after 6.5 years of follow-up based on the findings.
The benefits of LDCT for lung cancer screening in this population outweighed the risks, they noted.
Harms associated with LDCT, according to findings from 7 trials and 13 cohort studies that reported on such outcomes, included radiation exposure, overdiagnosis, and a high rate of false-positive findings that were resolved by further imaging in most cases. False negatives were reported in six studies, and the rates ranged from 0% to 20%, but none of the studies evaluated the harm of false reassurance, the investigators noted. Screening benefits must be weighed against these potential harms.
Lung cancer is the leading cause of cancer-related deaths, accounting for nearly 27%. Furthermore, about 85% of U.S. lung cancer cases are attributable to smoking, and since about 20% of Americans currently smoke – and many more are former smokers who remain at increased risk because of their smoking history – "lung cancer will remain a major public health problem in this country for decades," the investigators wrote.
The studies included in this review were conducted in patients at high risk for lung cancer based on current or former smoking. However, patients at an increased risk for lung cancer, including older adults and those with a family history of lung cancer, chronic obstructive pulmonary disease, pulmonary fibrosis, and certain environmental and occupational exposures, may also benefit from LDCT screening.
"Future research to identify methods for focusing LDCT screening on persons at highest risk for disease, to improve discrimination between benign and malignant pulmonary nodules, and to find early indicators of aggressive disease is warranted," the investigators noted.
"If LDCT screening becomes routine, the risk for harms should be measured and methods to limit them identified."
The review was funded by grants from the Agency for Healthcare Research and Quality and the Portland Veterans Affairs Medical Center.
Low-dose computed tomography reduces lung cancer mortality and all-cause mortality when used as a screening tool in asymptomatic adults at high risk for the disease, according to the results of a systematic review conducted for the U.S. Preventive Services Task Force.
In 2004, the USPSTF deemed the evidence insufficient for recommending for or against low-dose computed tomography (LDCT) for lung cancer screening in asymptomatic individuals, but the current review suggests screening has a definite benefit for most patients, Dr. Linda L. Humphrey, of Oregon Health and Science University and the Portland Veterans Affairs Medical Center, and her colleagues reported.
A draft recommendation based on the findings, published online in the Annals of Internal Medicine (2013 July 29 [doi: 10.7326/0003-4819-159-6-201309170-00690]), is available on the USPSTF website for comment.
The researchers reviewed the literature published between 2000 and May 2013 and identified four trials that reported findings on the efficacy of LDCT screening in patients with smoking exposure for both intervention and control groups. Three small trials showed varying degrees of benefit with screening, but were underpowered; one large trial – the National Lung Screening Trial (NLST) – showed a significant 20% reduction in lung cancer mortality among those screened, as well as a 6.7% reduction in all-cause mortality.
The randomized multicenter NLST compared annual LDCT scans with annual single-view posterior-anterior chest radiographs for 3 years in more than 53,000 current or former smokers aged 55-74 years with at least a 30–pack-year history of smoking (N. Engl. J. Med. 2013;368:1980-91). One cancer death was prevented for every 320 patients who completed one screening, and one death from any cause was prevented for every 219 patients screened in that study; the trial was stopped early after 6.5 years of follow-up based on the findings.
The benefits of LDCT for lung cancer screening in this population outweighed the risks, they noted.
Harms associated with LDCT, according to findings from 7 trials and 13 cohort studies that reported on such outcomes, included radiation exposure, overdiagnosis, and a high rate of false-positive findings that were resolved by further imaging in most cases. False negatives were reported in six studies, and the rates ranged from 0% to 20%, but none of the studies evaluated the harm of false reassurance, the investigators noted. Screening benefits must be weighed against these potential harms.
Lung cancer is the leading cause of cancer-related deaths, accounting for nearly 27%. Furthermore, about 85% of U.S. lung cancer cases are attributable to smoking, and since about 20% of Americans currently smoke – and many more are former smokers who remain at increased risk because of their smoking history – "lung cancer will remain a major public health problem in this country for decades," the investigators wrote.
The studies included in this review were conducted in patients at high risk for lung cancer based on current or former smoking. However, patients at an increased risk for lung cancer, including older adults and those with a family history of lung cancer, chronic obstructive pulmonary disease, pulmonary fibrosis, and certain environmental and occupational exposures, may also benefit from LDCT screening.
"Future research to identify methods for focusing LDCT screening on persons at highest risk for disease, to improve discrimination between benign and malignant pulmonary nodules, and to find early indicators of aggressive disease is warranted," the investigators noted.
"If LDCT screening becomes routine, the risk for harms should be measured and methods to limit them identified."
The review was funded by grants from the Agency for Healthcare Research and Quality and the Portland Veterans Affairs Medical Center.
Low-dose computed tomography reduces lung cancer mortality and all-cause mortality when used as a screening tool in asymptomatic adults at high risk for the disease, according to the results of a systematic review conducted for the U.S. Preventive Services Task Force.
In 2004, the USPSTF deemed the evidence insufficient for recommending for or against low-dose computed tomography (LDCT) for lung cancer screening in asymptomatic individuals, but the current review suggests screening has a definite benefit for most patients, Dr. Linda L. Humphrey, of Oregon Health and Science University and the Portland Veterans Affairs Medical Center, and her colleagues reported.
A draft recommendation based on the findings, published online in the Annals of Internal Medicine (2013 July 29 [doi: 10.7326/0003-4819-159-6-201309170-00690]), is available on the USPSTF website for comment.
The researchers reviewed the literature published between 2000 and May 2013 and identified four trials that reported findings on the efficacy of LDCT screening in patients with smoking exposure for both intervention and control groups. Three small trials showed varying degrees of benefit with screening, but were underpowered; one large trial – the National Lung Screening Trial (NLST) – showed a significant 20% reduction in lung cancer mortality among those screened, as well as a 6.7% reduction in all-cause mortality.
The randomized multicenter NLST compared annual LDCT scans with annual single-view posterior-anterior chest radiographs for 3 years in more than 53,000 current or former smokers aged 55-74 years with at least a 30–pack-year history of smoking (N. Engl. J. Med. 2013;368:1980-91). One cancer death was prevented for every 320 patients who completed one screening, and one death from any cause was prevented for every 219 patients screened in that study; the trial was stopped early after 6.5 years of follow-up based on the findings.
The benefits of LDCT for lung cancer screening in this population outweighed the risks, they noted.
Harms associated with LDCT, according to findings from 7 trials and 13 cohort studies that reported on such outcomes, included radiation exposure, overdiagnosis, and a high rate of false-positive findings that were resolved by further imaging in most cases. False negatives were reported in six studies, and the rates ranged from 0% to 20%, but none of the studies evaluated the harm of false reassurance, the investigators noted. Screening benefits must be weighed against these potential harms.
Lung cancer is the leading cause of cancer-related deaths, accounting for nearly 27%. Furthermore, about 85% of U.S. lung cancer cases are attributable to smoking, and since about 20% of Americans currently smoke – and many more are former smokers who remain at increased risk because of their smoking history – "lung cancer will remain a major public health problem in this country for decades," the investigators wrote.
The studies included in this review were conducted in patients at high risk for lung cancer based on current or former smoking. However, patients at an increased risk for lung cancer, including older adults and those with a family history of lung cancer, chronic obstructive pulmonary disease, pulmonary fibrosis, and certain environmental and occupational exposures, may also benefit from LDCT screening.
"Future research to identify methods for focusing LDCT screening on persons at highest risk for disease, to improve discrimination between benign and malignant pulmonary nodules, and to find early indicators of aggressive disease is warranted," the investigators noted.
"If LDCT screening becomes routine, the risk for harms should be measured and methods to limit them identified."
The review was funded by grants from the Agency for Healthcare Research and Quality and the Portland Veterans Affairs Medical Center.