User login
The Official Newspaper of the American Association for Thoracic Surgery
New tools for stroke prediction in atrial fibrillation
SNOWMASS, COLO. – High-sensitivity troponin T and brain natriuretic peptide levels are better predictors of stroke risk in patients with atrial fibrillation than the CHA2DS2-VASc score that will replace the CHADS2 score in the forthcoming revised American College of Cardiology/American Heart Association guidelines.
Recent evidence indicates the biomarkers may be novel tools for improved stroke prediction in atrial fibrillation (AF), with prognostic value above and beyond that provided by the CHA2DS2-VASc scores.
These findings raise important unanswered questions about the relationship between AF and stroke. Conventional wisdom has held that left atrial thrombus is the cause of most strokes in patients with AF. But it’s not that simple, Dr. Bernard J. Gersh asserted at the Annual Cardiovascular Conference at Snowmass.
"What are we measuring with these biomarkers? This is what we really don’t understand. What has high-sensitivity troponin T got to do with left atrial thrombus?" asked Dr. Gersh, professor of medicine at the Mayo Clinic in Rochester, Minn.
It seems increasingly clear that it’s not just the atrial arrhythmia that’s important in stroke risk, it’s also the company AF keeps. In a substantial but still uncertain proportion of patients, AF is a marker of vascular disease burden expressed through atrial and vascular endothelial dysfunction, vascular inflammation, left atrial dilatation and fibrosis, and a hypercoagulable state, the cardiologist continued.
He was a coinvestigator on a couple of recent groundbreaking studies that show the prognostic power of biomarkers in predicting both stroke risk and cardiac death in AF patients.
In one report, the investigators looked at baseline high-sensitivity troponin T (hsTnT) levels, clinical risk factors for stroke, and CHA2DS2-VASc scores in 12,892 patients with AF who were randomized to apixaban or warfarin in the prospective, double-blind ARISTOTLE (Apixaban for the Prevention of Stroke in Subjects With Atrial Fibrillation) trial (N. Engl. J. Med. 2011;365:981-92). During a median 1.9 years of follow-up, patients in the highest quartile for baseline hsTnT had roughly a twofold greater risk of stroke or systemic embolism than did those in the lowest quartile.
Moreover, patients with a low-risk CHA2DS2-VASc score of 0 or 1 but in the top quartile for hsTnT, with a level in excess of 13 ng/L, had a very substantial stroke rate of 2.7% per year despite anticoagulation with apixaban (Eliquis) or warfarin. The relationship was even stronger for cardiac death, where subjects with a low-risk CHA2DS2-VASc score who were in the top quartile for hsTnT had a 6% annual risk. A higher baseline hsTnT was also independently associated with sharply increased risk of major bleeding in a multivariate regression analysis (J. Am. Coll. Cardiol. 2014;63:52-61).
In another recently published study, he and his international coworkers showed in ARISTOTLE participants with baseline N-terminal pro-B-type natriuretic peptide (NT-proBNP) levels that this biomarker also improved stroke prediction in AF, providing added value to CHA2DS2-VASc scores. Subjects in the top quartile for baseline NT-proBNP had an adjusted 2.35-fold greater risk of stroke or systemic embolism than those in the lowest quartile, irrespective of CHA2DS2-VASc score. They also had a 2.5-fold greater risk of cardiac death (J. Am. Coll. Cardiol. 2013;61:2274-84).
A study Dr. Gersh highlighted as "extremely interesting" involved the use of brain natriuretic peptide (BNP) as a marker to rule out delayed AF in stroke patients. The study, led by investigators at the University Hospital Center of Nice (France) and known as TARGET-AF, included 300 consecutive acute stroke patients with no history of AF and no AF on their baseline ECG. During a median 6.8 days of in-hospital Holter monitoring, 17% of the stroke patients developed newly diagnosed AF.
The strongest predictor of delayed AF was baseline plasma BNP. It outperformed the CHA2DS2-VASc score and all the other parameters examined, including anterior circulation location of the stroke, P-wave initial force, gender, National Institutes of Health Stroke Scale score, age, left atrial dilatation, and Score for the Targeting of AF (STAF) score. A BNP level greater than 131 pg/mL had a 98.1% sensitivity, 71.4% specificity, and 99.4% negative predictive value for delayed AF.
"Our data indicate that a BNP level of 131 pg/mL or less might rule out delayed AF in stroke survivors and could be included in algorithms for AF detection," the French investigators concluded (J. Stroke Cerebrovasc. Dis. 2013;22:e103-10).
There is plenty of direct evidence from transesophageal echocardiography studies and other sources that a substantial proportion of thromboemboli are directly the result of AF. However, indirect evidence points to additional causal factors. For example, there is a high incidence of thromboembolic events in AF patients without left atrial appendage thrombus. Plus, in natural history studies patients with AF without additional risk factors have a low incidence of stroke. And CHADS2 and CHA2DS2-VASc scores predict vascular events but don’t correlate with left atrial appendage thrombus, Dr. Gersh noted.
He said the CHA2DS2-VASc score is clearly an improvement over CHADS2, and its adoption in the forthcoming ACC/AHA guidelines is to be welcomed. The CHA2DS2-VASc score increases the number of patients considered at significant risk of stroke and therefore warranting anticoagulation. For example, in a large Danish registry of nearly 48,000 AF patients with a CHADS2 score of 0-1 not on anticoagulation, patients with a CHADS2 score of 1 but a CHA2DS2-VASc score of 2 had twice the stroke risk of patients with a CHA2DS2-VASc of 1 (Thromb. Haemost. 2012;107:1172-9).
That being said, neither risk score is all that impressive. The C-statistic, a measure of a test’s predictive power, is 0.56 for CHADS2 and it was 0.62 for CHA2DS2-VASc in the ARISTOTLE analysis. To put those figures in perspective, a coin toss has a C-statistic of 0.50.
"The individual predictive values are not good. We use CHADS2 and CHA2DS2-VASc in practice and in the guidelines, but we should not pretend they are highly predictive. We need new risk stratification schemes," according to Dr. Gersh.
He reported serving as an adviser to Boston Scientific and St. Jude Medical.
SNOWMASS, COLO. – High-sensitivity troponin T and brain natriuretic peptide levels are better predictors of stroke risk in patients with atrial fibrillation than the CHA2DS2-VASc score that will replace the CHADS2 score in the forthcoming revised American College of Cardiology/American Heart Association guidelines.
Recent evidence indicates the biomarkers may be novel tools for improved stroke prediction in atrial fibrillation (AF), with prognostic value above and beyond that provided by the CHA2DS2-VASc scores.
These findings raise important unanswered questions about the relationship between AF and stroke. Conventional wisdom has held that left atrial thrombus is the cause of most strokes in patients with AF. But it’s not that simple, Dr. Bernard J. Gersh asserted at the Annual Cardiovascular Conference at Snowmass.
"What are we measuring with these biomarkers? This is what we really don’t understand. What has high-sensitivity troponin T got to do with left atrial thrombus?" asked Dr. Gersh, professor of medicine at the Mayo Clinic in Rochester, Minn.
It seems increasingly clear that it’s not just the atrial arrhythmia that’s important in stroke risk, it’s also the company AF keeps. In a substantial but still uncertain proportion of patients, AF is a marker of vascular disease burden expressed through atrial and vascular endothelial dysfunction, vascular inflammation, left atrial dilatation and fibrosis, and a hypercoagulable state, the cardiologist continued.
He was a coinvestigator on a couple of recent groundbreaking studies that show the prognostic power of biomarkers in predicting both stroke risk and cardiac death in AF patients.
In one report, the investigators looked at baseline high-sensitivity troponin T (hsTnT) levels, clinical risk factors for stroke, and CHA2DS2-VASc scores in 12,892 patients with AF who were randomized to apixaban or warfarin in the prospective, double-blind ARISTOTLE (Apixaban for the Prevention of Stroke in Subjects With Atrial Fibrillation) trial (N. Engl. J. Med. 2011;365:981-92). During a median 1.9 years of follow-up, patients in the highest quartile for baseline hsTnT had roughly a twofold greater risk of stroke or systemic embolism than did those in the lowest quartile.
Moreover, patients with a low-risk CHA2DS2-VASc score of 0 or 1 but in the top quartile for hsTnT, with a level in excess of 13 ng/L, had a very substantial stroke rate of 2.7% per year despite anticoagulation with apixaban (Eliquis) or warfarin. The relationship was even stronger for cardiac death, where subjects with a low-risk CHA2DS2-VASc score who were in the top quartile for hsTnT had a 6% annual risk. A higher baseline hsTnT was also independently associated with sharply increased risk of major bleeding in a multivariate regression analysis (J. Am. Coll. Cardiol. 2014;63:52-61).
In another recently published study, he and his international coworkers showed in ARISTOTLE participants with baseline N-terminal pro-B-type natriuretic peptide (NT-proBNP) levels that this biomarker also improved stroke prediction in AF, providing added value to CHA2DS2-VASc scores. Subjects in the top quartile for baseline NT-proBNP had an adjusted 2.35-fold greater risk of stroke or systemic embolism than those in the lowest quartile, irrespective of CHA2DS2-VASc score. They also had a 2.5-fold greater risk of cardiac death (J. Am. Coll. Cardiol. 2013;61:2274-84).
A study Dr. Gersh highlighted as "extremely interesting" involved the use of brain natriuretic peptide (BNP) as a marker to rule out delayed AF in stroke patients. The study, led by investigators at the University Hospital Center of Nice (France) and known as TARGET-AF, included 300 consecutive acute stroke patients with no history of AF and no AF on their baseline ECG. During a median 6.8 days of in-hospital Holter monitoring, 17% of the stroke patients developed newly diagnosed AF.
The strongest predictor of delayed AF was baseline plasma BNP. It outperformed the CHA2DS2-VASc score and all the other parameters examined, including anterior circulation location of the stroke, P-wave initial force, gender, National Institutes of Health Stroke Scale score, age, left atrial dilatation, and Score for the Targeting of AF (STAF) score. A BNP level greater than 131 pg/mL had a 98.1% sensitivity, 71.4% specificity, and 99.4% negative predictive value for delayed AF.
"Our data indicate that a BNP level of 131 pg/mL or less might rule out delayed AF in stroke survivors and could be included in algorithms for AF detection," the French investigators concluded (J. Stroke Cerebrovasc. Dis. 2013;22:e103-10).
There is plenty of direct evidence from transesophageal echocardiography studies and other sources that a substantial proportion of thromboemboli are directly the result of AF. However, indirect evidence points to additional causal factors. For example, there is a high incidence of thromboembolic events in AF patients without left atrial appendage thrombus. Plus, in natural history studies patients with AF without additional risk factors have a low incidence of stroke. And CHADS2 and CHA2DS2-VASc scores predict vascular events but don’t correlate with left atrial appendage thrombus, Dr. Gersh noted.
He said the CHA2DS2-VASc score is clearly an improvement over CHADS2, and its adoption in the forthcoming ACC/AHA guidelines is to be welcomed. The CHA2DS2-VASc score increases the number of patients considered at significant risk of stroke and therefore warranting anticoagulation. For example, in a large Danish registry of nearly 48,000 AF patients with a CHADS2 score of 0-1 not on anticoagulation, patients with a CHADS2 score of 1 but a CHA2DS2-VASc score of 2 had twice the stroke risk of patients with a CHA2DS2-VASc of 1 (Thromb. Haemost. 2012;107:1172-9).
That being said, neither risk score is all that impressive. The C-statistic, a measure of a test’s predictive power, is 0.56 for CHADS2 and it was 0.62 for CHA2DS2-VASc in the ARISTOTLE analysis. To put those figures in perspective, a coin toss has a C-statistic of 0.50.
"The individual predictive values are not good. We use CHADS2 and CHA2DS2-VASc in practice and in the guidelines, but we should not pretend they are highly predictive. We need new risk stratification schemes," according to Dr. Gersh.
He reported serving as an adviser to Boston Scientific and St. Jude Medical.
SNOWMASS, COLO. – High-sensitivity troponin T and brain natriuretic peptide levels are better predictors of stroke risk in patients with atrial fibrillation than the CHA2DS2-VASc score that will replace the CHADS2 score in the forthcoming revised American College of Cardiology/American Heart Association guidelines.
Recent evidence indicates the biomarkers may be novel tools for improved stroke prediction in atrial fibrillation (AF), with prognostic value above and beyond that provided by the CHA2DS2-VASc scores.
These findings raise important unanswered questions about the relationship between AF and stroke. Conventional wisdom has held that left atrial thrombus is the cause of most strokes in patients with AF. But it’s not that simple, Dr. Bernard J. Gersh asserted at the Annual Cardiovascular Conference at Snowmass.
"What are we measuring with these biomarkers? This is what we really don’t understand. What has high-sensitivity troponin T got to do with left atrial thrombus?" asked Dr. Gersh, professor of medicine at the Mayo Clinic in Rochester, Minn.
It seems increasingly clear that it’s not just the atrial arrhythmia that’s important in stroke risk, it’s also the company AF keeps. In a substantial but still uncertain proportion of patients, AF is a marker of vascular disease burden expressed through atrial and vascular endothelial dysfunction, vascular inflammation, left atrial dilatation and fibrosis, and a hypercoagulable state, the cardiologist continued.
He was a coinvestigator on a couple of recent groundbreaking studies that show the prognostic power of biomarkers in predicting both stroke risk and cardiac death in AF patients.
In one report, the investigators looked at baseline high-sensitivity troponin T (hsTnT) levels, clinical risk factors for stroke, and CHA2DS2-VASc scores in 12,892 patients with AF who were randomized to apixaban or warfarin in the prospective, double-blind ARISTOTLE (Apixaban for the Prevention of Stroke in Subjects With Atrial Fibrillation) trial (N. Engl. J. Med. 2011;365:981-92). During a median 1.9 years of follow-up, patients in the highest quartile for baseline hsTnT had roughly a twofold greater risk of stroke or systemic embolism than did those in the lowest quartile.
Moreover, patients with a low-risk CHA2DS2-VASc score of 0 or 1 but in the top quartile for hsTnT, with a level in excess of 13 ng/L, had a very substantial stroke rate of 2.7% per year despite anticoagulation with apixaban (Eliquis) or warfarin. The relationship was even stronger for cardiac death, where subjects with a low-risk CHA2DS2-VASc score who were in the top quartile for hsTnT had a 6% annual risk. A higher baseline hsTnT was also independently associated with sharply increased risk of major bleeding in a multivariate regression analysis (J. Am. Coll. Cardiol. 2014;63:52-61).
In another recently published study, he and his international coworkers showed in ARISTOTLE participants with baseline N-terminal pro-B-type natriuretic peptide (NT-proBNP) levels that this biomarker also improved stroke prediction in AF, providing added value to CHA2DS2-VASc scores. Subjects in the top quartile for baseline NT-proBNP had an adjusted 2.35-fold greater risk of stroke or systemic embolism than those in the lowest quartile, irrespective of CHA2DS2-VASc score. They also had a 2.5-fold greater risk of cardiac death (J. Am. Coll. Cardiol. 2013;61:2274-84).
A study Dr. Gersh highlighted as "extremely interesting" involved the use of brain natriuretic peptide (BNP) as a marker to rule out delayed AF in stroke patients. The study, led by investigators at the University Hospital Center of Nice (France) and known as TARGET-AF, included 300 consecutive acute stroke patients with no history of AF and no AF on their baseline ECG. During a median 6.8 days of in-hospital Holter monitoring, 17% of the stroke patients developed newly diagnosed AF.
The strongest predictor of delayed AF was baseline plasma BNP. It outperformed the CHA2DS2-VASc score and all the other parameters examined, including anterior circulation location of the stroke, P-wave initial force, gender, National Institutes of Health Stroke Scale score, age, left atrial dilatation, and Score for the Targeting of AF (STAF) score. A BNP level greater than 131 pg/mL had a 98.1% sensitivity, 71.4% specificity, and 99.4% negative predictive value for delayed AF.
"Our data indicate that a BNP level of 131 pg/mL or less might rule out delayed AF in stroke survivors and could be included in algorithms for AF detection," the French investigators concluded (J. Stroke Cerebrovasc. Dis. 2013;22:e103-10).
There is plenty of direct evidence from transesophageal echocardiography studies and other sources that a substantial proportion of thromboemboli are directly the result of AF. However, indirect evidence points to additional causal factors. For example, there is a high incidence of thromboembolic events in AF patients without left atrial appendage thrombus. Plus, in natural history studies patients with AF without additional risk factors have a low incidence of stroke. And CHADS2 and CHA2DS2-VASc scores predict vascular events but don’t correlate with left atrial appendage thrombus, Dr. Gersh noted.
He said the CHA2DS2-VASc score is clearly an improvement over CHADS2, and its adoption in the forthcoming ACC/AHA guidelines is to be welcomed. The CHA2DS2-VASc score increases the number of patients considered at significant risk of stroke and therefore warranting anticoagulation. For example, in a large Danish registry of nearly 48,000 AF patients with a CHADS2 score of 0-1 not on anticoagulation, patients with a CHADS2 score of 1 but a CHA2DS2-VASc score of 2 had twice the stroke risk of patients with a CHA2DS2-VASc of 1 (Thromb. Haemost. 2012;107:1172-9).
That being said, neither risk score is all that impressive. The C-statistic, a measure of a test’s predictive power, is 0.56 for CHADS2 and it was 0.62 for CHA2DS2-VASc in the ARISTOTLE analysis. To put those figures in perspective, a coin toss has a C-statistic of 0.50.
"The individual predictive values are not good. We use CHADS2 and CHA2DS2-VASc in practice and in the guidelines, but we should not pretend they are highly predictive. We need new risk stratification schemes," according to Dr. Gersh.
He reported serving as an adviser to Boston Scientific and St. Jude Medical.
EXPERT ANALYSIS FROM THE CARDIOVASCULAR CONFERENCE AT SNOWMASS
Endoscopic resection for adenocarcinoma
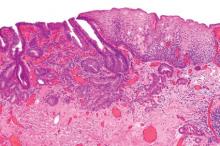
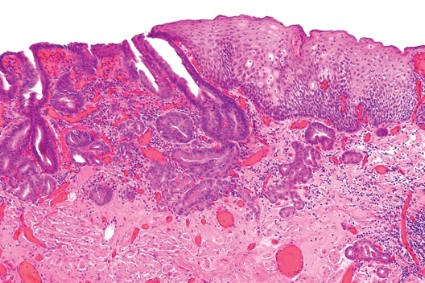
Endoscopic resection for mucosal esophageal adenocarcinoma is safe and highly effective, and should be the new standard of care. That's according to Dr. Oliver Pech, whose study in the March issue of Gastroenterology showed a complete remission rate of 93.8% over nearly 5 years of follow-up (doi: 10.1053/j.gasro.2013.11.006).
Dr. Pech of the University of Regensburg (Germany) and his colleagues looked at 1,000 consecutive patients (mean age, 69 years; 861 men) with mucosal adenocarcinoma of the esophagus, referred to a single center from October 1996 to September 2010.
All patients had mucosal Barrett's carcinoma; lesions judged resectable were subjected to diagnostic endoscopic resection for staging, even when the macroscopic appearance suggested submucosal disease. Patients with low-grade dysplasia, high-grade dysplasia, and submucosal or more advanced cancer (T1 or greater) were excluded.
In total, 481 patients had short-segment Barrett's esophagus, and the remainder had long-segment Barrett's. The majority (n = 493) had intraepithelial adenocarcinoma, according to staging by endoscopic resection, while 240 patients had adenocarcinoma invading the tunica propria, 124 had invasion of the first layer of the muscularis mucosae, and the remaining 143 had disease of the second layer of the muscularis mucosae. En bloc resection was performed in 508 patients; piecemeal resection in the rest.
Complete remission, defined as an R0 resection plus one normal surveillance endoscopy, was achieved in 963 (96.3%) of 1,000 patients in the study.
Among these, recurrence of neoplasia (high-grade dysplasia or adenocarcinoma) was detected in 14.5% of the patients (140 out of the 963) after a median 26.5 months; 115 were successfully retreated with additional endoscopic resection. That translated to a long-term complete remission rate of 93.8% (mean, 56.6 months) and a 5-year survival rate of 91.5%.
Looking at safety, Dr. Pech reported that 15 patients experienced major complications, including bleeding with a corresponding drop in hemoglobin of at least 2 g/dL (in 14 cases) and perforation (in 1). He added that the relatively minor complication of stenosis requiring dilation occurred in 13 cases, all of which were managed endoscopically. Finally, in an analysis of which patients were more likely to have successful endoscopic treatment, they determined that long-segment Barrett's as well as poorly differentiated mucosal adenocarcinoma had a significantly higher risk for failure (P less than .0001 for both).
The authors conceded that referral bias cannot be excluded in this cohort. Additionally, over the long course of the study, best practices for Barrett's esophagus and high-grade dysplasia have evolved considerably.
The authors had no disclosures.
Endoscopic resection for mucosal esophageal adenocarcinoma is safe and highly effective, and should be the new standard of care. That's according to Dr. Oliver Pech, whose study in the March issue of Gastroenterology showed a complete remission rate of 93.8% over nearly 5 years of follow-up (doi: 10.1053/j.gasro.2013.11.006).
Dr. Pech of the University of Regensburg (Germany) and his colleagues looked at 1,000 consecutive patients (mean age, 69 years; 861 men) with mucosal adenocarcinoma of the esophagus, referred to a single center from October 1996 to September 2010.
All patients had mucosal Barrett's carcinoma; lesions judged resectable were subjected to diagnostic endoscopic resection for staging, even when the macroscopic appearance suggested submucosal disease. Patients with low-grade dysplasia, high-grade dysplasia, and submucosal or more advanced cancer (T1 or greater) were excluded.
In total, 481 patients had short-segment Barrett's esophagus, and the remainder had long-segment Barrett's. The majority (n = 493) had intraepithelial adenocarcinoma, according to staging by endoscopic resection, while 240 patients had adenocarcinoma invading the tunica propria, 124 had invasion of the first layer of the muscularis mucosae, and the remaining 143 had disease of the second layer of the muscularis mucosae. En bloc resection was performed in 508 patients; piecemeal resection in the rest.
Complete remission, defined as an R0 resection plus one normal surveillance endoscopy, was achieved in 963 (96.3%) of 1,000 patients in the study.
Among these, recurrence of neoplasia (high-grade dysplasia or adenocarcinoma) was detected in 14.5% of the patients (140 out of the 963) after a median 26.5 months; 115 were successfully retreated with additional endoscopic resection. That translated to a long-term complete remission rate of 93.8% (mean, 56.6 months) and a 5-year survival rate of 91.5%.
Looking at safety, Dr. Pech reported that 15 patients experienced major complications, including bleeding with a corresponding drop in hemoglobin of at least 2 g/dL (in 14 cases) and perforation (in 1). He added that the relatively minor complication of stenosis requiring dilation occurred in 13 cases, all of which were managed endoscopically. Finally, in an analysis of which patients were more likely to have successful endoscopic treatment, they determined that long-segment Barrett's as well as poorly differentiated mucosal adenocarcinoma had a significantly higher risk for failure (P less than .0001 for both).
The authors conceded that referral bias cannot be excluded in this cohort. Additionally, over the long course of the study, best practices for Barrett's esophagus and high-grade dysplasia have evolved considerably.
The authors had no disclosures.
Endoscopic resection for mucosal esophageal adenocarcinoma is safe and highly effective, and should be the new standard of care. That's according to Dr. Oliver Pech, whose study in the March issue of Gastroenterology showed a complete remission rate of 93.8% over nearly 5 years of follow-up (doi: 10.1053/j.gasro.2013.11.006).
Dr. Pech of the University of Regensburg (Germany) and his colleagues looked at 1,000 consecutive patients (mean age, 69 years; 861 men) with mucosal adenocarcinoma of the esophagus, referred to a single center from October 1996 to September 2010.
All patients had mucosal Barrett's carcinoma; lesions judged resectable were subjected to diagnostic endoscopic resection for staging, even when the macroscopic appearance suggested submucosal disease. Patients with low-grade dysplasia, high-grade dysplasia, and submucosal or more advanced cancer (T1 or greater) were excluded.
In total, 481 patients had short-segment Barrett's esophagus, and the remainder had long-segment Barrett's. The majority (n = 493) had intraepithelial adenocarcinoma, according to staging by endoscopic resection, while 240 patients had adenocarcinoma invading the tunica propria, 124 had invasion of the first layer of the muscularis mucosae, and the remaining 143 had disease of the second layer of the muscularis mucosae. En bloc resection was performed in 508 patients; piecemeal resection in the rest.
Complete remission, defined as an R0 resection plus one normal surveillance endoscopy, was achieved in 963 (96.3%) of 1,000 patients in the study.
Among these, recurrence of neoplasia (high-grade dysplasia or adenocarcinoma) was detected in 14.5% of the patients (140 out of the 963) after a median 26.5 months; 115 were successfully retreated with additional endoscopic resection. That translated to a long-term complete remission rate of 93.8% (mean, 56.6 months) and a 5-year survival rate of 91.5%.
Looking at safety, Dr. Pech reported that 15 patients experienced major complications, including bleeding with a corresponding drop in hemoglobin of at least 2 g/dL (in 14 cases) and perforation (in 1). He added that the relatively minor complication of stenosis requiring dilation occurred in 13 cases, all of which were managed endoscopically. Finally, in an analysis of which patients were more likely to have successful endoscopic treatment, they determined that long-segment Barrett's as well as poorly differentiated mucosal adenocarcinoma had a significantly higher risk for failure (P less than .0001 for both).
The authors conceded that referral bias cannot be excluded in this cohort. Additionally, over the long course of the study, best practices for Barrett's esophagus and high-grade dysplasia have evolved considerably.
The authors had no disclosures.
Major finding: Endoscopic resection of esophageal adenocarcinoma resulted in a long-term complete remission rate of 93.8%.
Data source: Data from 1,000 consecutive patients with mucosal adenocarcinoma of the esophagus.
Disclosures: The authors stated that they had no conflicts of interest to disclose. They disclosed no funding.
Anti-PD-L1 therapy shows promise in early NSCLC trials
SAN DIEGO – Results from studies conducted to date indicate that anti-PD-L1 therapy is well tolerated in patients with non–small cell lung cancer, with rapid response rates.
Responses are "not only very rapid, they’re also very durable," Dr. Leora Horn said at the Joint Conference on the Molecular Origins of Lung Cancer sponsored by the American Association for Cancer Research and the International Association for the Study of Lung Cancer.
"We’re seeing continued responses even when treatment is discontinued. The response rates appear to be somewhat higher in tumors that express PD-L1, and we see no pneumonitis or treatment-related grade 5 adverse events to date."
The four anti-PD-L1 agents that have been investigated or are currently being investigated in patients with non–small cell lung cancer (NSCLC) are BMS-936559, MPDL3280A, MEDI-4736, and MSB0010718C, said Dr. Horn, clinical director of the thoracic oncology research program at Vanderbilt University, Nashville, Tenn.
BMS-936559, developed by Bristol-Myers Squibb, was part of a phase IA dose study at 0.3, 1, 3, and 10 mg/kg in patients with multiple tumor types including NSCLC. Of the 207 patients enrolled, 75 had NSCLC (N. Engl. J. Med 2012;366:2455-65).
More than half of all patients (61%) had treatment-related adverse events, with fatigue the most common (16%), followed by infusion reaction (10%) and diarrhea (9%). Grade 3 and 4 adverse events occurred in 9% of patients. "One of the themes with anti-PD-L1 therapy is that we’re not seeing grade 3-5 pneumonitis in these like we have seen with anti-PD-1 agents," said Dr. Horn. "This may have to do with different expression of PD-1 and PD-2 within the body and the targets of PD-1 compared to anti-PD-L1 antibodies."
The response rate among NSCLC patients treated in the trial reached about 10%, and responses were seen in squamous and nonsquamous NSCLC. "This agent is not being further developed in NSCLC patients," Dr. Horn said. "Nivolumab, a PD-1- blocking antibody, has really taken over as far as development for Bristol-Myers Squibb."
The next agent she discussed, MPDL3280A, is being developed by Roche and Genentech. A phase I trial presented at the 2013 European Society for Medical Oncology (ESMO) Congress examined different doses in 85 patients with NSCLC ranging from 10 to 20 mg/kg once every week for just under 1 year. Key eligibility criteria were measurable disease per Response Evaluation Criteria in Solid Tumors v1.1 and Eastern Cooperative Oncology Group Performance Status 0 or 1. Dr. Horn said that the majority of adverse events were grade 1-2 and did not require intervention. "There was also no maximum tolerated dose or dose-limiting toxicities, and no grade 3-5 pneumonitis was observed," she said.
In a study of MPDL3280A presented by Dr. Horn and her associates at the 2013 World Conference on Lung Cancer, the clinical impact was assessed in 53 patients with NSCLC. Patients first dosed at 1-20 mg/kg by Oct. 1, 2012, with data cutoff on April 30, 2013. The response rate was 83% among patients who had an immunohistochemistry (IHC) score of 3, 46% among those who were IHC 2 and 3, and 31% among those who were IHC 1, 2, and 3. "In all-comers the response rate was 23%," Dr. Horn said. "The responses in these patients are very rapid. We see them by their first or second CT [computed tomography] scan. The responses are also very durable regardless of their IHC status and in patients who are PD-L1 negative. They’ve also been responding even after treatment has been discontinued."
She and her associates examined the response rate to MPDL3280A based on smoking status and mutational status. The response rate was higher in patients who were current or former smokers compared with never smokers (26% vs. 10%, respectively). By molecular status, the response rate in EGFR [epidermal growth factor receptor] wild-type patients was 26%, compared with 17% in EGFR mutant patients. In addition, there was a 30% response rate in patients who were KRAS wild-type and a 10% response rate in KRAS mutant patients, although the numbers are very small, she noted.
Dr. Horn said there are three separate, ongoing studies looking at MPDL3280A in NSCLC patients: one in patients with PD-L1-positive NSCLC, one in combination with bevacizumab and/or chemotherapy, and one randomized phase III trial comparing the agent with docetaxel in patients after platinum failure.
Data on the next agent Dr. Horn discussed, MEDI-4736 from MedImmune, are limited. A study was presented at the 2013 ESMO Congress and included 11 patients who received doses of 0.1, 0.3, and 1.0 mg/kg. The most common adverse events were diarrhea, vomiting, and dizziness (18% each), and no grade 3 or 4 adverse events or pneumonitis have been reported to date.
Pharmacokinetic studies of MEDI-4736 indicate a dose-dependent increase in target engagement, consistent with binding of the agent to PD-L1. Initial clinical data on eight patients demonstrated response at all different dose levels, ranging from 42% to 80%. "This involves small subsets of patients, so hopefully we’ll see more data on this agent in the next couple of years," Dr. Horn said.
The final anti-PD-L1 in clinical development is MSB0010718C from EMD Serono, but no data are available yet. A three-dose escalation study up to 10 mg/kg is underway. "Hopefully, we’ll see some data on this later this year," Dr. Horn said.
Dr. Horn disclosed that she is a speaker for Bristol-Myers Squibb, Theradex, and other companies.
The PD-L1/PD-1 ligand complex is a natural suppressive pathway used by cells to inhibit IL-2 production and T-cell proliferation so that inflammation is kept under control. However, some remarkably clever cancers including renal cell, ovarian, and non–small cell lung cancer exploit this pathway by up-regulating PD-L1 to evade and hide from the host’s immune system by suppressing inflammation. Four different monoclonal antibodies, such as MPDL3280A, that block PD-L1 have been developed. By blocking this anti-inflammatory pathway, these agents expose the cancer to the host’s activated immune system – the activated "killer" (cytotoxic) T cells.
| Dr. Lary Robinson |
Early results in a number of phase I clinical trials of anti-PD-L1 agents have shown remarkable and exciting responses using these minimally toxic agents in patients with highly chemoresistant stage IV lung cancer. Somewhat higher responses rates are seen with these agents, and the results are rapid and durable even when treatment is discontinued. This novel immunotherapy approach to systemic treatment of lung cancer is regarded by thoracic oncologists as a potential breakthrough in treatment, and it may soon become the preferred first-line, well-tolerated therapy for this very large group of metastatic lung cancer patients who express high levels of PD-L1, as well as PD-L1–negative tumors. Numerous trials with these agents are ongoing in order to ascertain the most appropriate dosages, potential use with other chemotherapy drugs, and most suitable patient population.
Dr. Lary A. Robinson is professor of thoracic surgery and interdisciplinary oncology at the University of South Florida, Tampa.
The PD-L1/PD-1 ligand complex is a natural suppressive pathway used by cells to inhibit IL-2 production and T-cell proliferation so that inflammation is kept under control. However, some remarkably clever cancers including renal cell, ovarian, and non–small cell lung cancer exploit this pathway by up-regulating PD-L1 to evade and hide from the host’s immune system by suppressing inflammation. Four different monoclonal antibodies, such as MPDL3280A, that block PD-L1 have been developed. By blocking this anti-inflammatory pathway, these agents expose the cancer to the host’s activated immune system – the activated "killer" (cytotoxic) T cells.
| Dr. Lary Robinson |
Early results in a number of phase I clinical trials of anti-PD-L1 agents have shown remarkable and exciting responses using these minimally toxic agents in patients with highly chemoresistant stage IV lung cancer. Somewhat higher responses rates are seen with these agents, and the results are rapid and durable even when treatment is discontinued. This novel immunotherapy approach to systemic treatment of lung cancer is regarded by thoracic oncologists as a potential breakthrough in treatment, and it may soon become the preferred first-line, well-tolerated therapy for this very large group of metastatic lung cancer patients who express high levels of PD-L1, as well as PD-L1–negative tumors. Numerous trials with these agents are ongoing in order to ascertain the most appropriate dosages, potential use with other chemotherapy drugs, and most suitable patient population.
Dr. Lary A. Robinson is professor of thoracic surgery and interdisciplinary oncology at the University of South Florida, Tampa.
The PD-L1/PD-1 ligand complex is a natural suppressive pathway used by cells to inhibit IL-2 production and T-cell proliferation so that inflammation is kept under control. However, some remarkably clever cancers including renal cell, ovarian, and non–small cell lung cancer exploit this pathway by up-regulating PD-L1 to evade and hide from the host’s immune system by suppressing inflammation. Four different monoclonal antibodies, such as MPDL3280A, that block PD-L1 have been developed. By blocking this anti-inflammatory pathway, these agents expose the cancer to the host’s activated immune system – the activated "killer" (cytotoxic) T cells.
| Dr. Lary Robinson |
Early results in a number of phase I clinical trials of anti-PD-L1 agents have shown remarkable and exciting responses using these minimally toxic agents in patients with highly chemoresistant stage IV lung cancer. Somewhat higher responses rates are seen with these agents, and the results are rapid and durable even when treatment is discontinued. This novel immunotherapy approach to systemic treatment of lung cancer is regarded by thoracic oncologists as a potential breakthrough in treatment, and it may soon become the preferred first-line, well-tolerated therapy for this very large group of metastatic lung cancer patients who express high levels of PD-L1, as well as PD-L1–negative tumors. Numerous trials with these agents are ongoing in order to ascertain the most appropriate dosages, potential use with other chemotherapy drugs, and most suitable patient population.
Dr. Lary A. Robinson is professor of thoracic surgery and interdisciplinary oncology at the University of South Florida, Tampa.
SAN DIEGO – Results from studies conducted to date indicate that anti-PD-L1 therapy is well tolerated in patients with non–small cell lung cancer, with rapid response rates.
Responses are "not only very rapid, they’re also very durable," Dr. Leora Horn said at the Joint Conference on the Molecular Origins of Lung Cancer sponsored by the American Association for Cancer Research and the International Association for the Study of Lung Cancer.
"We’re seeing continued responses even when treatment is discontinued. The response rates appear to be somewhat higher in tumors that express PD-L1, and we see no pneumonitis or treatment-related grade 5 adverse events to date."
The four anti-PD-L1 agents that have been investigated or are currently being investigated in patients with non–small cell lung cancer (NSCLC) are BMS-936559, MPDL3280A, MEDI-4736, and MSB0010718C, said Dr. Horn, clinical director of the thoracic oncology research program at Vanderbilt University, Nashville, Tenn.
BMS-936559, developed by Bristol-Myers Squibb, was part of a phase IA dose study at 0.3, 1, 3, and 10 mg/kg in patients with multiple tumor types including NSCLC. Of the 207 patients enrolled, 75 had NSCLC (N. Engl. J. Med 2012;366:2455-65).
More than half of all patients (61%) had treatment-related adverse events, with fatigue the most common (16%), followed by infusion reaction (10%) and diarrhea (9%). Grade 3 and 4 adverse events occurred in 9% of patients. "One of the themes with anti-PD-L1 therapy is that we’re not seeing grade 3-5 pneumonitis in these like we have seen with anti-PD-1 agents," said Dr. Horn. "This may have to do with different expression of PD-1 and PD-2 within the body and the targets of PD-1 compared to anti-PD-L1 antibodies."
The response rate among NSCLC patients treated in the trial reached about 10%, and responses were seen in squamous and nonsquamous NSCLC. "This agent is not being further developed in NSCLC patients," Dr. Horn said. "Nivolumab, a PD-1- blocking antibody, has really taken over as far as development for Bristol-Myers Squibb."
The next agent she discussed, MPDL3280A, is being developed by Roche and Genentech. A phase I trial presented at the 2013 European Society for Medical Oncology (ESMO) Congress examined different doses in 85 patients with NSCLC ranging from 10 to 20 mg/kg once every week for just under 1 year. Key eligibility criteria were measurable disease per Response Evaluation Criteria in Solid Tumors v1.1 and Eastern Cooperative Oncology Group Performance Status 0 or 1. Dr. Horn said that the majority of adverse events were grade 1-2 and did not require intervention. "There was also no maximum tolerated dose or dose-limiting toxicities, and no grade 3-5 pneumonitis was observed," she said.
In a study of MPDL3280A presented by Dr. Horn and her associates at the 2013 World Conference on Lung Cancer, the clinical impact was assessed in 53 patients with NSCLC. Patients first dosed at 1-20 mg/kg by Oct. 1, 2012, with data cutoff on April 30, 2013. The response rate was 83% among patients who had an immunohistochemistry (IHC) score of 3, 46% among those who were IHC 2 and 3, and 31% among those who were IHC 1, 2, and 3. "In all-comers the response rate was 23%," Dr. Horn said. "The responses in these patients are very rapid. We see them by their first or second CT [computed tomography] scan. The responses are also very durable regardless of their IHC status and in patients who are PD-L1 negative. They’ve also been responding even after treatment has been discontinued."
She and her associates examined the response rate to MPDL3280A based on smoking status and mutational status. The response rate was higher in patients who were current or former smokers compared with never smokers (26% vs. 10%, respectively). By molecular status, the response rate in EGFR [epidermal growth factor receptor] wild-type patients was 26%, compared with 17% in EGFR mutant patients. In addition, there was a 30% response rate in patients who were KRAS wild-type and a 10% response rate in KRAS mutant patients, although the numbers are very small, she noted.
Dr. Horn said there are three separate, ongoing studies looking at MPDL3280A in NSCLC patients: one in patients with PD-L1-positive NSCLC, one in combination with bevacizumab and/or chemotherapy, and one randomized phase III trial comparing the agent with docetaxel in patients after platinum failure.
Data on the next agent Dr. Horn discussed, MEDI-4736 from MedImmune, are limited. A study was presented at the 2013 ESMO Congress and included 11 patients who received doses of 0.1, 0.3, and 1.0 mg/kg. The most common adverse events were diarrhea, vomiting, and dizziness (18% each), and no grade 3 or 4 adverse events or pneumonitis have been reported to date.
Pharmacokinetic studies of MEDI-4736 indicate a dose-dependent increase in target engagement, consistent with binding of the agent to PD-L1. Initial clinical data on eight patients demonstrated response at all different dose levels, ranging from 42% to 80%. "This involves small subsets of patients, so hopefully we’ll see more data on this agent in the next couple of years," Dr. Horn said.
The final anti-PD-L1 in clinical development is MSB0010718C from EMD Serono, but no data are available yet. A three-dose escalation study up to 10 mg/kg is underway. "Hopefully, we’ll see some data on this later this year," Dr. Horn said.
Dr. Horn disclosed that she is a speaker for Bristol-Myers Squibb, Theradex, and other companies.
SAN DIEGO – Results from studies conducted to date indicate that anti-PD-L1 therapy is well tolerated in patients with non–small cell lung cancer, with rapid response rates.
Responses are "not only very rapid, they’re also very durable," Dr. Leora Horn said at the Joint Conference on the Molecular Origins of Lung Cancer sponsored by the American Association for Cancer Research and the International Association for the Study of Lung Cancer.
"We’re seeing continued responses even when treatment is discontinued. The response rates appear to be somewhat higher in tumors that express PD-L1, and we see no pneumonitis or treatment-related grade 5 adverse events to date."
The four anti-PD-L1 agents that have been investigated or are currently being investigated in patients with non–small cell lung cancer (NSCLC) are BMS-936559, MPDL3280A, MEDI-4736, and MSB0010718C, said Dr. Horn, clinical director of the thoracic oncology research program at Vanderbilt University, Nashville, Tenn.
BMS-936559, developed by Bristol-Myers Squibb, was part of a phase IA dose study at 0.3, 1, 3, and 10 mg/kg in patients with multiple tumor types including NSCLC. Of the 207 patients enrolled, 75 had NSCLC (N. Engl. J. Med 2012;366:2455-65).
More than half of all patients (61%) had treatment-related adverse events, with fatigue the most common (16%), followed by infusion reaction (10%) and diarrhea (9%). Grade 3 and 4 adverse events occurred in 9% of patients. "One of the themes with anti-PD-L1 therapy is that we’re not seeing grade 3-5 pneumonitis in these like we have seen with anti-PD-1 agents," said Dr. Horn. "This may have to do with different expression of PD-1 and PD-2 within the body and the targets of PD-1 compared to anti-PD-L1 antibodies."
The response rate among NSCLC patients treated in the trial reached about 10%, and responses were seen in squamous and nonsquamous NSCLC. "This agent is not being further developed in NSCLC patients," Dr. Horn said. "Nivolumab, a PD-1- blocking antibody, has really taken over as far as development for Bristol-Myers Squibb."
The next agent she discussed, MPDL3280A, is being developed by Roche and Genentech. A phase I trial presented at the 2013 European Society for Medical Oncology (ESMO) Congress examined different doses in 85 patients with NSCLC ranging from 10 to 20 mg/kg once every week for just under 1 year. Key eligibility criteria were measurable disease per Response Evaluation Criteria in Solid Tumors v1.1 and Eastern Cooperative Oncology Group Performance Status 0 or 1. Dr. Horn said that the majority of adverse events were grade 1-2 and did not require intervention. "There was also no maximum tolerated dose or dose-limiting toxicities, and no grade 3-5 pneumonitis was observed," she said.
In a study of MPDL3280A presented by Dr. Horn and her associates at the 2013 World Conference on Lung Cancer, the clinical impact was assessed in 53 patients with NSCLC. Patients first dosed at 1-20 mg/kg by Oct. 1, 2012, with data cutoff on April 30, 2013. The response rate was 83% among patients who had an immunohistochemistry (IHC) score of 3, 46% among those who were IHC 2 and 3, and 31% among those who were IHC 1, 2, and 3. "In all-comers the response rate was 23%," Dr. Horn said. "The responses in these patients are very rapid. We see them by their first or second CT [computed tomography] scan. The responses are also very durable regardless of their IHC status and in patients who are PD-L1 negative. They’ve also been responding even after treatment has been discontinued."
She and her associates examined the response rate to MPDL3280A based on smoking status and mutational status. The response rate was higher in patients who were current or former smokers compared with never smokers (26% vs. 10%, respectively). By molecular status, the response rate in EGFR [epidermal growth factor receptor] wild-type patients was 26%, compared with 17% in EGFR mutant patients. In addition, there was a 30% response rate in patients who were KRAS wild-type and a 10% response rate in KRAS mutant patients, although the numbers are very small, she noted.
Dr. Horn said there are three separate, ongoing studies looking at MPDL3280A in NSCLC patients: one in patients with PD-L1-positive NSCLC, one in combination with bevacizumab and/or chemotherapy, and one randomized phase III trial comparing the agent with docetaxel in patients after platinum failure.
Data on the next agent Dr. Horn discussed, MEDI-4736 from MedImmune, are limited. A study was presented at the 2013 ESMO Congress and included 11 patients who received doses of 0.1, 0.3, and 1.0 mg/kg. The most common adverse events were diarrhea, vomiting, and dizziness (18% each), and no grade 3 or 4 adverse events or pneumonitis have been reported to date.
Pharmacokinetic studies of MEDI-4736 indicate a dose-dependent increase in target engagement, consistent with binding of the agent to PD-L1. Initial clinical data on eight patients demonstrated response at all different dose levels, ranging from 42% to 80%. "This involves small subsets of patients, so hopefully we’ll see more data on this agent in the next couple of years," Dr. Horn said.
The final anti-PD-L1 in clinical development is MSB0010718C from EMD Serono, but no data are available yet. A three-dose escalation study up to 10 mg/kg is underway. "Hopefully, we’ll see some data on this later this year," Dr. Horn said.
Dr. Horn disclosed that she is a speaker for Bristol-Myers Squibb, Theradex, and other companies.
Genotyping adds little to optimized warfarin dosing
DALLAS - Genotyping to guide the starting dosage of warfarin treatment showed no added value above tailoring treatment with a panel of clinical features in a randomized, controlled U.S. trial of more than 1,000 patients.
The resounding null result from adding genotype information should spell the end of this practice, said Dr. Stephen E. Kimmel, lead investigator for the study, and professor and director of cardiovascular epidemiology at the University of Pennsylvania in Philadelphia.
"Based on what we've seen, I don't believe there is sufficient evidence to add genetic information on top of the available clinical algorithms," Dr. Kimmel said at the American Heart Association scientific sessions. "I don't think we'll see another genetics trial in warfarin treatment. I think this is it."
Good outcomes in the comparison group largely accounted for the failure of genotyping to significantly improve the percentage of time that patients' international normalized ratios (INRs) were in the target range of 2.0-3.0. The amount of time in the therapeutic INR range (PTTR) averaged 45% in patients in the comparison arm during the first 4 weeks of warfarin treatment.
"When the comparison group does so well, it's more difficult for genotyping to have an effect," said Dr. Elaine M. Hylek, professor of medicine and an anticoagulant specialist at Boston University.
That limitation, coupled with the shifting anticoagulant landscape, knock genotyping out of the picture, she agreed. "It will be difficult funding [another study of genotyping] with all the anticoagulant alternatives that are now out there" for preventing thrombosis in patients with atrial fibrillation or a recent venous thromboembolism.
The Clarification of Optimal Anticoagulant through Genetics (COAG) trial enrolled 1,015 patients initiating warfarin therapy at 18 U.S. centers during September 2009 to April 2013. The study randomized patients to two different ways to calculate their warfarin dosage during the first 5 days of treatment.
Half had their dosage calculated by a formula that took into account seven clinical and demographic factors, including age, race, smoking status, and body surface area. The others had their dosage calculated with the same formula and factors plus added information on the patient's genotype for two genes that affect warfarin activity, CYP2C9 and VKORC1. Patients' average age was 58 years; just over a quarter were African American.
During the first 4 weeks on treatment, the average PTTR was 45% in both arms of the study, the trial's primary endpoint. Adding genotyping information led to a bigger improvement in the PTTR among the non-African American patients compared with those who were African American, but did not produce a significantly increased PTTR in the non-African American subgroup.
Dr. Munir Pirmohamed reported results from a similar study that enrolled 455 patients starting warfarin therapy at any of five centers in the United Kingdom and Sweden.
The EU-Pharmacogenetics of Anticoagulant Therapy (EU-PACT) Warfarin study mainly differed from the COAG study by the background method used to calculate a starting warfarin dosage over the first 3 days of treatment.
In EU-PACT, the warfarin dosage of all patients was adjusted for age but not for other factors. In the intervention arm, the starting dosages were adjusted for the status of the same two genes, CYP2C9 and VKORC1.
During the first 12 weeks after starting warfarin, the cumulative average PTTR was 67% in the patients whose dose was adjusted by genotype and 60% in patients who were not genotyped, a statistically significant difference for this study's primary endpoint, reported Dr. Pirmohamed, professor and head of molecular and clinical pharmacology at the University of Liverpool, England.
Taken together, the findings of the two studies highlight that patients should not start warfarin treatment on a fixed dosage, said Dr. Patrick T. Ellinor, a cardiologist and arrhythmia specialist at Massachusetts General Hospital, Boston, and designated discussant for both reports at the meeting.
The findings support use of a clinical algorithm that takes into account several clinical factors, he added.
Concurrently with the meeting, the reports were published online for COAG (N. Engl. J. Med. 2013 [doi: 10.1056/NEJMoa1310669]) and for EU-PACT (N. Engl. J. Med. 2013 [doi: 10.1056/NEJMoa1311386]).
Warfarin on top despite competition
Warfarin remains the most widely used anticoagulant in the United States. Among patients with atrial fibrillation taking anticoagulation treatment, 72% used warfarin during the third quarter of 2013, data from a large registry show.
The new anticoagulants on the U.S. market - dabigatran (Pradaxa), apixaban (Eliquis), and rivaroxaban (Xarelto) - are gaining ground and widely acknowledged to be better, safer, and easier to manage. But warfarin clings to the market largely because of familiarity and low price, according to several experts.
"There is no doubt that the new anticoagulants look better compared with warfarin, but the clinical fact is that it's what you know versus what you don't know, and warfarin has been used for 60 years," Dr. Kimmel said at the press conference.
"The new drugs perform better than warfarin does; they get patients to where they need to be more quickly, but warfarin has been around a long time. We know its interactions and risks, there is the ability to reverse its effect, and the cost to patients is a real issue. If the new anticoagulants were the same price as warfarin then I think you'd see a lot more patients get a new drug," Dr. Ellinor said in an interview.
Data on current U.S. uptake of the new oral anticoagulants came from the 148,320 unique patients with atrial fibrillation included during June to September of 2013 in the PINNACLE Registry database run by the American College of Cardiology. Among these patients, about 83,000 (56%) were on some anticoagulant treatment, and within this subgroup, 72% were on warfarin, 27% on a new anticoagulant, and the remainder on different treatment, said Dr. John Gordon Harold, ACC president and a cardiologist at Cedars-Sinai Heart Institute in Los Angeles.
"In my own practice the new anticoagulants are being used with increasing frequency, mainly driven by direct-to-consumer advertising," Dr. Harold said in an interview. "We have patients who are completely stable on warfarin. (They) come in because of a consumer ad and they ask if they should switch drugs. When patients are stable I don't encourage them to switch, but we have a shared decision making conversation and go over the pros and cons, the cost, and the outcomes data. A lot of patients prefer to pay the difference" and switch to a new anticoagulant.
Dr. Harold said he also recommends that patients switch off warfarin if they have problems with compliance and variability in their international normalized ratio (INR).
"If you can keep a patient on warfarin in their INR target range 80% or more of the time then I wouldn't change, but most patients on warfarin have a very hard time maintaining an INR of 2-3," said Dr. Mark S. Link, professor and codirector of the cardiac arrhythmia center at Tufts Medical Center, Boston. But he said cost is a major factor keeping many patients on warfarin.
The new anticoagulants "are better than warfarin, but we are often forced by insurers to start with the cheaper drug," Dr. Link said during the news conference.
The COAG and EU-PACT studies did not receive any direct commercial sponsorship. Dr. Kimmel has been a consultant to Pfizer and Janssen. Dr. Hylek has been a consultant or adviser to Bayer, Boehringer Ingelheim, Bristol-Myers Squibb, Daiichi Sankyo, Johnson & Johnson, and Pfizer. Dr. Pirmohamed, Dr. Ellinor, Dr. Harold, and Dr. Link had no relevant disclosures.
On Twitter @mitchelzoler
DALLAS - Genotyping to guide the starting dosage of warfarin treatment showed no added value above tailoring treatment with a panel of clinical features in a randomized, controlled U.S. trial of more than 1,000 patients.
The resounding null result from adding genotype information should spell the end of this practice, said Dr. Stephen E. Kimmel, lead investigator for the study, and professor and director of cardiovascular epidemiology at the University of Pennsylvania in Philadelphia.
"Based on what we've seen, I don't believe there is sufficient evidence to add genetic information on top of the available clinical algorithms," Dr. Kimmel said at the American Heart Association scientific sessions. "I don't think we'll see another genetics trial in warfarin treatment. I think this is it."
Good outcomes in the comparison group largely accounted for the failure of genotyping to significantly improve the percentage of time that patients' international normalized ratios (INRs) were in the target range of 2.0-3.0. The amount of time in the therapeutic INR range (PTTR) averaged 45% in patients in the comparison arm during the first 4 weeks of warfarin treatment.
"When the comparison group does so well, it's more difficult for genotyping to have an effect," said Dr. Elaine M. Hylek, professor of medicine and an anticoagulant specialist at Boston University.
That limitation, coupled with the shifting anticoagulant landscape, knock genotyping out of the picture, she agreed. "It will be difficult funding [another study of genotyping] with all the anticoagulant alternatives that are now out there" for preventing thrombosis in patients with atrial fibrillation or a recent venous thromboembolism.
The Clarification of Optimal Anticoagulant through Genetics (COAG) trial enrolled 1,015 patients initiating warfarin therapy at 18 U.S. centers during September 2009 to April 2013. The study randomized patients to two different ways to calculate their warfarin dosage during the first 5 days of treatment.
Half had their dosage calculated by a formula that took into account seven clinical and demographic factors, including age, race, smoking status, and body surface area. The others had their dosage calculated with the same formula and factors plus added information on the patient's genotype for two genes that affect warfarin activity, CYP2C9 and VKORC1. Patients' average age was 58 years; just over a quarter were African American.
During the first 4 weeks on treatment, the average PTTR was 45% in both arms of the study, the trial's primary endpoint. Adding genotyping information led to a bigger improvement in the PTTR among the non-African American patients compared with those who were African American, but did not produce a significantly increased PTTR in the non-African American subgroup.
Dr. Munir Pirmohamed reported results from a similar study that enrolled 455 patients starting warfarin therapy at any of five centers in the United Kingdom and Sweden.
The EU-Pharmacogenetics of Anticoagulant Therapy (EU-PACT) Warfarin study mainly differed from the COAG study by the background method used to calculate a starting warfarin dosage over the first 3 days of treatment.
In EU-PACT, the warfarin dosage of all patients was adjusted for age but not for other factors. In the intervention arm, the starting dosages were adjusted for the status of the same two genes, CYP2C9 and VKORC1.
During the first 12 weeks after starting warfarin, the cumulative average PTTR was 67% in the patients whose dose was adjusted by genotype and 60% in patients who were not genotyped, a statistically significant difference for this study's primary endpoint, reported Dr. Pirmohamed, professor and head of molecular and clinical pharmacology at the University of Liverpool, England.
Taken together, the findings of the two studies highlight that patients should not start warfarin treatment on a fixed dosage, said Dr. Patrick T. Ellinor, a cardiologist and arrhythmia specialist at Massachusetts General Hospital, Boston, and designated discussant for both reports at the meeting.
The findings support use of a clinical algorithm that takes into account several clinical factors, he added.
Concurrently with the meeting, the reports were published online for COAG (N. Engl. J. Med. 2013 [doi: 10.1056/NEJMoa1310669]) and for EU-PACT (N. Engl. J. Med. 2013 [doi: 10.1056/NEJMoa1311386]).
Warfarin on top despite competition
Warfarin remains the most widely used anticoagulant in the United States. Among patients with atrial fibrillation taking anticoagulation treatment, 72% used warfarin during the third quarter of 2013, data from a large registry show.
The new anticoagulants on the U.S. market - dabigatran (Pradaxa), apixaban (Eliquis), and rivaroxaban (Xarelto) - are gaining ground and widely acknowledged to be better, safer, and easier to manage. But warfarin clings to the market largely because of familiarity and low price, according to several experts.
"There is no doubt that the new anticoagulants look better compared with warfarin, but the clinical fact is that it's what you know versus what you don't know, and warfarin has been used for 60 years," Dr. Kimmel said at the press conference.
"The new drugs perform better than warfarin does; they get patients to where they need to be more quickly, but warfarin has been around a long time. We know its interactions and risks, there is the ability to reverse its effect, and the cost to patients is a real issue. If the new anticoagulants were the same price as warfarin then I think you'd see a lot more patients get a new drug," Dr. Ellinor said in an interview.
Data on current U.S. uptake of the new oral anticoagulants came from the 148,320 unique patients with atrial fibrillation included during June to September of 2013 in the PINNACLE Registry database run by the American College of Cardiology. Among these patients, about 83,000 (56%) were on some anticoagulant treatment, and within this subgroup, 72% were on warfarin, 27% on a new anticoagulant, and the remainder on different treatment, said Dr. John Gordon Harold, ACC president and a cardiologist at Cedars-Sinai Heart Institute in Los Angeles.
"In my own practice the new anticoagulants are being used with increasing frequency, mainly driven by direct-to-consumer advertising," Dr. Harold said in an interview. "We have patients who are completely stable on warfarin. (They) come in because of a consumer ad and they ask if they should switch drugs. When patients are stable I don't encourage them to switch, but we have a shared decision making conversation and go over the pros and cons, the cost, and the outcomes data. A lot of patients prefer to pay the difference" and switch to a new anticoagulant.
Dr. Harold said he also recommends that patients switch off warfarin if they have problems with compliance and variability in their international normalized ratio (INR).
"If you can keep a patient on warfarin in their INR target range 80% or more of the time then I wouldn't change, but most patients on warfarin have a very hard time maintaining an INR of 2-3," said Dr. Mark S. Link, professor and codirector of the cardiac arrhythmia center at Tufts Medical Center, Boston. But he said cost is a major factor keeping many patients on warfarin.
The new anticoagulants "are better than warfarin, but we are often forced by insurers to start with the cheaper drug," Dr. Link said during the news conference.
The COAG and EU-PACT studies did not receive any direct commercial sponsorship. Dr. Kimmel has been a consultant to Pfizer and Janssen. Dr. Hylek has been a consultant or adviser to Bayer, Boehringer Ingelheim, Bristol-Myers Squibb, Daiichi Sankyo, Johnson & Johnson, and Pfizer. Dr. Pirmohamed, Dr. Ellinor, Dr. Harold, and Dr. Link had no relevant disclosures.
On Twitter @mitchelzoler
DALLAS - Genotyping to guide the starting dosage of warfarin treatment showed no added value above tailoring treatment with a panel of clinical features in a randomized, controlled U.S. trial of more than 1,000 patients.
The resounding null result from adding genotype information should spell the end of this practice, said Dr. Stephen E. Kimmel, lead investigator for the study, and professor and director of cardiovascular epidemiology at the University of Pennsylvania in Philadelphia.
"Based on what we've seen, I don't believe there is sufficient evidence to add genetic information on top of the available clinical algorithms," Dr. Kimmel said at the American Heart Association scientific sessions. "I don't think we'll see another genetics trial in warfarin treatment. I think this is it."
Good outcomes in the comparison group largely accounted for the failure of genotyping to significantly improve the percentage of time that patients' international normalized ratios (INRs) were in the target range of 2.0-3.0. The amount of time in the therapeutic INR range (PTTR) averaged 45% in patients in the comparison arm during the first 4 weeks of warfarin treatment.
"When the comparison group does so well, it's more difficult for genotyping to have an effect," said Dr. Elaine M. Hylek, professor of medicine and an anticoagulant specialist at Boston University.
That limitation, coupled with the shifting anticoagulant landscape, knock genotyping out of the picture, she agreed. "It will be difficult funding [another study of genotyping] with all the anticoagulant alternatives that are now out there" for preventing thrombosis in patients with atrial fibrillation or a recent venous thromboembolism.
The Clarification of Optimal Anticoagulant through Genetics (COAG) trial enrolled 1,015 patients initiating warfarin therapy at 18 U.S. centers during September 2009 to April 2013. The study randomized patients to two different ways to calculate their warfarin dosage during the first 5 days of treatment.
Half had their dosage calculated by a formula that took into account seven clinical and demographic factors, including age, race, smoking status, and body surface area. The others had their dosage calculated with the same formula and factors plus added information on the patient's genotype for two genes that affect warfarin activity, CYP2C9 and VKORC1. Patients' average age was 58 years; just over a quarter were African American.
During the first 4 weeks on treatment, the average PTTR was 45% in both arms of the study, the trial's primary endpoint. Adding genotyping information led to a bigger improvement in the PTTR among the non-African American patients compared with those who were African American, but did not produce a significantly increased PTTR in the non-African American subgroup.
Dr. Munir Pirmohamed reported results from a similar study that enrolled 455 patients starting warfarin therapy at any of five centers in the United Kingdom and Sweden.
The EU-Pharmacogenetics of Anticoagulant Therapy (EU-PACT) Warfarin study mainly differed from the COAG study by the background method used to calculate a starting warfarin dosage over the first 3 days of treatment.
In EU-PACT, the warfarin dosage of all patients was adjusted for age but not for other factors. In the intervention arm, the starting dosages were adjusted for the status of the same two genes, CYP2C9 and VKORC1.
During the first 12 weeks after starting warfarin, the cumulative average PTTR was 67% in the patients whose dose was adjusted by genotype and 60% in patients who were not genotyped, a statistically significant difference for this study's primary endpoint, reported Dr. Pirmohamed, professor and head of molecular and clinical pharmacology at the University of Liverpool, England.
Taken together, the findings of the two studies highlight that patients should not start warfarin treatment on a fixed dosage, said Dr. Patrick T. Ellinor, a cardiologist and arrhythmia specialist at Massachusetts General Hospital, Boston, and designated discussant for both reports at the meeting.
The findings support use of a clinical algorithm that takes into account several clinical factors, he added.
Concurrently with the meeting, the reports were published online for COAG (N. Engl. J. Med. 2013 [doi: 10.1056/NEJMoa1310669]) and for EU-PACT (N. Engl. J. Med. 2013 [doi: 10.1056/NEJMoa1311386]).
Warfarin on top despite competition
Warfarin remains the most widely used anticoagulant in the United States. Among patients with atrial fibrillation taking anticoagulation treatment, 72% used warfarin during the third quarter of 2013, data from a large registry show.
The new anticoagulants on the U.S. market - dabigatran (Pradaxa), apixaban (Eliquis), and rivaroxaban (Xarelto) - are gaining ground and widely acknowledged to be better, safer, and easier to manage. But warfarin clings to the market largely because of familiarity and low price, according to several experts.
"There is no doubt that the new anticoagulants look better compared with warfarin, but the clinical fact is that it's what you know versus what you don't know, and warfarin has been used for 60 years," Dr. Kimmel said at the press conference.
"The new drugs perform better than warfarin does; they get patients to where they need to be more quickly, but warfarin has been around a long time. We know its interactions and risks, there is the ability to reverse its effect, and the cost to patients is a real issue. If the new anticoagulants were the same price as warfarin then I think you'd see a lot more patients get a new drug," Dr. Ellinor said in an interview.
Data on current U.S. uptake of the new oral anticoagulants came from the 148,320 unique patients with atrial fibrillation included during June to September of 2013 in the PINNACLE Registry database run by the American College of Cardiology. Among these patients, about 83,000 (56%) were on some anticoagulant treatment, and within this subgroup, 72% were on warfarin, 27% on a new anticoagulant, and the remainder on different treatment, said Dr. John Gordon Harold, ACC president and a cardiologist at Cedars-Sinai Heart Institute in Los Angeles.
"In my own practice the new anticoagulants are being used with increasing frequency, mainly driven by direct-to-consumer advertising," Dr. Harold said in an interview. "We have patients who are completely stable on warfarin. (They) come in because of a consumer ad and they ask if they should switch drugs. When patients are stable I don't encourage them to switch, but we have a shared decision making conversation and go over the pros and cons, the cost, and the outcomes data. A lot of patients prefer to pay the difference" and switch to a new anticoagulant.
Dr. Harold said he also recommends that patients switch off warfarin if they have problems with compliance and variability in their international normalized ratio (INR).
"If you can keep a patient on warfarin in their INR target range 80% or more of the time then I wouldn't change, but most patients on warfarin have a very hard time maintaining an INR of 2-3," said Dr. Mark S. Link, professor and codirector of the cardiac arrhythmia center at Tufts Medical Center, Boston. But he said cost is a major factor keeping many patients on warfarin.
The new anticoagulants "are better than warfarin, but we are often forced by insurers to start with the cheaper drug," Dr. Link said during the news conference.
The COAG and EU-PACT studies did not receive any direct commercial sponsorship. Dr. Kimmel has been a consultant to Pfizer and Janssen. Dr. Hylek has been a consultant or adviser to Bayer, Boehringer Ingelheim, Bristol-Myers Squibb, Daiichi Sankyo, Johnson & Johnson, and Pfizer. Dr. Pirmohamed, Dr. Ellinor, Dr. Harold, and Dr. Link had no relevant disclosures.
On Twitter @mitchelzoler
Major finding: Patients starting warfarin with dosing based on a formula that took into account seven clinical and demographic factors averaged 45% of the time in therapeutic range regardless of whether the starting dosage was adjusted based on genotype results.
Data source: COAG, a randomized trial with 1,015 patients starting warfarin therapy at 18 U.S. centers.
Disclosures: The COAG and EU-PACT studies did not receive any direct commercial sponsorship. Dr. Kimmel has been a consultant to Pfizer and Janssen. Dr. Hylek has been a consultant or adviser to Bayer, Boehringer Ingelheim, Bristol-Myers Squibb, Daiichi Sankyo, Johnson & Johnson, and Pfizer. Dr. Pirmohamed and Dr. Ellinor had no disclosures.
On the Go Education: Mobile software in cardiothoracic training
In nearly every facet of our lives, our mobile devices have taken over. Managing our calendars, organizing our contacts, and planning our driving directions -- our devices have become invaluable and ubiquitously present. While the ease of use of smartphones and tablets puts the power of portable computing in the hands of everyone, mobile software seems to be particularly appreciated by young professionals, who seek the convenience of on-the-go functionality and feel comfortable with computing in the palms of their hands. Throughout the world of education and a breadth of academic fields, advanced software programs have gained momentum, recognized for their ability to provide up-to-date, on-the-ground information.
In recent years, there has been an explosion of new software programs applicable to the field of cardiothoracic surgery, and these applications have been well received by modern trainees.
"Mobile apps are incredibly convenient because they provide a means of accessing information while on the go," states Jonathan Spicer, a thoracic trainee at M.D. Anderson Cancer Center in Houston.
He continues, "Having the capacity to look up helpful information from my phone while in the operating room, on the ward, or in transit is particularly helpful."
In this article, we aim to highlight some of the more exciting and innovative mobile software programs available today for those interested in expanding their knowledge in cardiothoracic surgery or looking for an easy-to-access resource.
iBronch (Edward Bender), $0.99: iBronch is one of the many outstanding thoracic surgical apps developed by Ed Bender. This program aims to guide learners through the basic anatomy of the trachea and bronchial tree, with correlation of simultaneous images from a fiberoptic bronchoscope and along an anatomic airway diagram. Branches of the pulmonary tree are labeled on the schematic and the bronchoscopy images. This app is particularly useful for those trainees gaining comfort with bronchoscopic procedures; however, its utility may be less significant for more advanced learners. Regardless, this is a great program, quite helpful for the intended audience.
Thoracic Lymph Node Map (RADIOLOGiQ, LLC), Free: This app provides a color-coded lymph node map, associated with computed tomography images and adapted from the International Association for the Study of Lung Cancer (IASLC) lung cancer project. This program provides excellent illustrations of the anatomic definitions for each of the intrathoracic lymph node stations. This is helpful both in examining imaging studies of actual patients and in the operating room.
CT Journals (Edward Bender), Free: This software program serves as a scholarly journal aggregator for the field of cardiothoracic surgery. The app displays feeds for journals of interest, with inclusion of those relevant periodicals with the most readership and highest impact factors. Not only can one access the articles while online, abstracts can be saved for future use offline. This is a great resource, but users should be aware that access to the full articles is available only for those who have active accounts providing them access to the specific journals.
CTSNetWiki (Edward Bender), Free: Cardiothoracic Surgery Notes is an online review developed and maintained by residents in thoracic surgical training. This resource is a tremendous repository of information, compiling graphics, text, and other multimedia content on a breadth of topics. This app allows general review of a wide variety of cardiothoracic surgical problems and is appropriate for both the novice learner and as a review for those who are further along in their training.
SESATS IX (Edward Bender), Free: Perhaps the most valuable mobile software application out there, the Self Education Self Assessment in Thoracic Surgery (SESATS) IX application contains actual questions from previous versions of the SESATS. The mobile app even includes the associated images, videos, and CT scans that correspond with the questions. Although the program does not contain the latest version of SESATS, the utility of this app cannot be overestimated. This program is enormously helpful for self-testing, on-the-go topic-specific learning, and exam preparation. This is a real gem of a find, and it comes with a strong recommendation to all trainees for its download and use.
TSRA Primer of Cardiothoracic Surgery (Thoracic Surgery Residents' Association), $4.99: Produced by CT residents for CT residents, this is probably the most useful resource for the intern, junior resident, or new fellow who needs to brush up on the basics before rounds, in between consults, and before assisting in the OR. It's not comprehensive, but it is full of clinical pearls covering all the major divisions of cardiothoracic surgery. Many cardiac residents, especially younger integrated residents, have been waiting for a straightforward, practical tutorial like this for years. Beautiful and often interactive illustrations and videos really make this iBook memorable.
NCCN Guidelines (TIP Medical Communications), Free: Available for Android and iOS, this compendium of NCCN guidelines for 56 cancers and cancer-related topics is indispensible for trainees. The utility of having up-to-date, in-depth guidelines for diagnosis and staging of all commonly encountered malignancies cannot be overstated. Additional topics ranging from management of cancer-related emesis to lung cancer screening guidelines polish it off. A must have for anyone who treats cancer, not just thoracic surgeons.
CathSource (ECGSource, LLC), $3.99: Available for Android and iOS, CathSource is a mobile app that aims to teach cardiovascular medicine fellows about coronary anatomy, angiogram projections, and catheter-based hemodynamic measurements. Luckily, CT trainees stand to benefit from the app as well. It seems to be most helpful for learning coronary anatomy on the different projections, but it also has exhaustive hemodynamic formulae and tracings for more detailed review. The app has over 30 videos of normal and abnormal findings.
EchoSource (ECGSource, LLC), $4.99:It's the same idea as above, but -- you guessed it -- for echocardiography. Both are good tools, especially for residents who teach. Both apps take simple, conceptual drawings to start and expand them with real imaging. Residents who have spent a good deal of time in learning cath and echo may find these apps less useful.
Pocket Heart (PocketAnatomy), $9.99: This is an interactive, 3D heart model with a fairly detailed presentation of cardiac anatomy and added features such as pinning quizzes and case studies. While cardiothoracic residents ought to have the anatomy down, the app can be used to teach patients and families about various anatomical aspects of cardiac disease. The graphics leave a little to be desired, but anyone who teaches medical students or patients frequently will enjoy having this easy-to-understand tool handy.
This list of cardiothoracic-specific mobile applications is by no means exhaustive. Each physician's needs will be different, and the options are countless. Countless risk calculators, mnemonic databases, formula compendiums, and pharmacologic formularies clutter the app store. Note-taking suites such as OneNote and Evernote (personal favorite of both of the authors) can help turn the most hare-brained resident into a paragon of organizational excellence. Journal citation managers such as EndNote, Dropbox, Mendeley, and Yep can help organize and manage that virtual pile of unread but probably important journal articles that keeps building up in your inbox. Even the humble iBooks app can be used to read and mark up pdf files on the fly, all while syncing with your library on your home computer. Many hospital EMRs have mobile platforms with various levels of functionality for tablets and smartphones.
What is obvious is that mobile computing technology is rapidly changing medicine and surgery in many ways. Although each one of us strives to be a complete physician, utterly self-reliant and assured of one's clinical knowledge, we all must learn the basics first. Whether at the bus stop, in a resident lounge, or in the operating room, these mobile technologies help us to learn more efficiently while on the go.
If there are any gems we have forgotten to highlight, please send an e-mail to Thoracic Surgery News and we will try to present them in the future. We hope that the residents reading this column can find a new app they didn't know they needed, one that will energize them and push their learning to a new height. Just don't forget to look up once in a while.
Dr. Antonoff is a 2nd-year, Thoracic-track trainee at Washington University in St Louis. Dr. Zeigler is a 3rd-year, integrated Cardiothoracic Surgery trainee at Stanford (Calif.) University. They reported no relevant financial conflicts.
In nearly every facet of our lives, our mobile devices have taken over. Managing our calendars, organizing our contacts, and planning our driving directions -- our devices have become invaluable and ubiquitously present. While the ease of use of smartphones and tablets puts the power of portable computing in the hands of everyone, mobile software seems to be particularly appreciated by young professionals, who seek the convenience of on-the-go functionality and feel comfortable with computing in the palms of their hands. Throughout the world of education and a breadth of academic fields, advanced software programs have gained momentum, recognized for their ability to provide up-to-date, on-the-ground information.
In recent years, there has been an explosion of new software programs applicable to the field of cardiothoracic surgery, and these applications have been well received by modern trainees.
"Mobile apps are incredibly convenient because they provide a means of accessing information while on the go," states Jonathan Spicer, a thoracic trainee at M.D. Anderson Cancer Center in Houston.
He continues, "Having the capacity to look up helpful information from my phone while in the operating room, on the ward, or in transit is particularly helpful."
In this article, we aim to highlight some of the more exciting and innovative mobile software programs available today for those interested in expanding their knowledge in cardiothoracic surgery or looking for an easy-to-access resource.
iBronch (Edward Bender), $0.99: iBronch is one of the many outstanding thoracic surgical apps developed by Ed Bender. This program aims to guide learners through the basic anatomy of the trachea and bronchial tree, with correlation of simultaneous images from a fiberoptic bronchoscope and along an anatomic airway diagram. Branches of the pulmonary tree are labeled on the schematic and the bronchoscopy images. This app is particularly useful for those trainees gaining comfort with bronchoscopic procedures; however, its utility may be less significant for more advanced learners. Regardless, this is a great program, quite helpful for the intended audience.
Thoracic Lymph Node Map (RADIOLOGiQ, LLC), Free: This app provides a color-coded lymph node map, associated with computed tomography images and adapted from the International Association for the Study of Lung Cancer (IASLC) lung cancer project. This program provides excellent illustrations of the anatomic definitions for each of the intrathoracic lymph node stations. This is helpful both in examining imaging studies of actual patients and in the operating room.
CT Journals (Edward Bender), Free: This software program serves as a scholarly journal aggregator for the field of cardiothoracic surgery. The app displays feeds for journals of interest, with inclusion of those relevant periodicals with the most readership and highest impact factors. Not only can one access the articles while online, abstracts can be saved for future use offline. This is a great resource, but users should be aware that access to the full articles is available only for those who have active accounts providing them access to the specific journals.
CTSNetWiki (Edward Bender), Free: Cardiothoracic Surgery Notes is an online review developed and maintained by residents in thoracic surgical training. This resource is a tremendous repository of information, compiling graphics, text, and other multimedia content on a breadth of topics. This app allows general review of a wide variety of cardiothoracic surgical problems and is appropriate for both the novice learner and as a review for those who are further along in their training.
SESATS IX (Edward Bender), Free: Perhaps the most valuable mobile software application out there, the Self Education Self Assessment in Thoracic Surgery (SESATS) IX application contains actual questions from previous versions of the SESATS. The mobile app even includes the associated images, videos, and CT scans that correspond with the questions. Although the program does not contain the latest version of SESATS, the utility of this app cannot be overestimated. This program is enormously helpful for self-testing, on-the-go topic-specific learning, and exam preparation. This is a real gem of a find, and it comes with a strong recommendation to all trainees for its download and use.
TSRA Primer of Cardiothoracic Surgery (Thoracic Surgery Residents' Association), $4.99: Produced by CT residents for CT residents, this is probably the most useful resource for the intern, junior resident, or new fellow who needs to brush up on the basics before rounds, in between consults, and before assisting in the OR. It's not comprehensive, but it is full of clinical pearls covering all the major divisions of cardiothoracic surgery. Many cardiac residents, especially younger integrated residents, have been waiting for a straightforward, practical tutorial like this for years. Beautiful and often interactive illustrations and videos really make this iBook memorable.
NCCN Guidelines (TIP Medical Communications), Free: Available for Android and iOS, this compendium of NCCN guidelines for 56 cancers and cancer-related topics is indispensible for trainees. The utility of having up-to-date, in-depth guidelines for diagnosis and staging of all commonly encountered malignancies cannot be overstated. Additional topics ranging from management of cancer-related emesis to lung cancer screening guidelines polish it off. A must have for anyone who treats cancer, not just thoracic surgeons.
CathSource (ECGSource, LLC), $3.99: Available for Android and iOS, CathSource is a mobile app that aims to teach cardiovascular medicine fellows about coronary anatomy, angiogram projections, and catheter-based hemodynamic measurements. Luckily, CT trainees stand to benefit from the app as well. It seems to be most helpful for learning coronary anatomy on the different projections, but it also has exhaustive hemodynamic formulae and tracings for more detailed review. The app has over 30 videos of normal and abnormal findings.
EchoSource (ECGSource, LLC), $4.99:It's the same idea as above, but -- you guessed it -- for echocardiography. Both are good tools, especially for residents who teach. Both apps take simple, conceptual drawings to start and expand them with real imaging. Residents who have spent a good deal of time in learning cath and echo may find these apps less useful.
Pocket Heart (PocketAnatomy), $9.99: This is an interactive, 3D heart model with a fairly detailed presentation of cardiac anatomy and added features such as pinning quizzes and case studies. While cardiothoracic residents ought to have the anatomy down, the app can be used to teach patients and families about various anatomical aspects of cardiac disease. The graphics leave a little to be desired, but anyone who teaches medical students or patients frequently will enjoy having this easy-to-understand tool handy.
This list of cardiothoracic-specific mobile applications is by no means exhaustive. Each physician's needs will be different, and the options are countless. Countless risk calculators, mnemonic databases, formula compendiums, and pharmacologic formularies clutter the app store. Note-taking suites such as OneNote and Evernote (personal favorite of both of the authors) can help turn the most hare-brained resident into a paragon of organizational excellence. Journal citation managers such as EndNote, Dropbox, Mendeley, and Yep can help organize and manage that virtual pile of unread but probably important journal articles that keeps building up in your inbox. Even the humble iBooks app can be used to read and mark up pdf files on the fly, all while syncing with your library on your home computer. Many hospital EMRs have mobile platforms with various levels of functionality for tablets and smartphones.
What is obvious is that mobile computing technology is rapidly changing medicine and surgery in many ways. Although each one of us strives to be a complete physician, utterly self-reliant and assured of one's clinical knowledge, we all must learn the basics first. Whether at the bus stop, in a resident lounge, or in the operating room, these mobile technologies help us to learn more efficiently while on the go.
If there are any gems we have forgotten to highlight, please send an e-mail to Thoracic Surgery News and we will try to present them in the future. We hope that the residents reading this column can find a new app they didn't know they needed, one that will energize them and push their learning to a new height. Just don't forget to look up once in a while.
Dr. Antonoff is a 2nd-year, Thoracic-track trainee at Washington University in St Louis. Dr. Zeigler is a 3rd-year, integrated Cardiothoracic Surgery trainee at Stanford (Calif.) University. They reported no relevant financial conflicts.
In nearly every facet of our lives, our mobile devices have taken over. Managing our calendars, organizing our contacts, and planning our driving directions -- our devices have become invaluable and ubiquitously present. While the ease of use of smartphones and tablets puts the power of portable computing in the hands of everyone, mobile software seems to be particularly appreciated by young professionals, who seek the convenience of on-the-go functionality and feel comfortable with computing in the palms of their hands. Throughout the world of education and a breadth of academic fields, advanced software programs have gained momentum, recognized for their ability to provide up-to-date, on-the-ground information.
In recent years, there has been an explosion of new software programs applicable to the field of cardiothoracic surgery, and these applications have been well received by modern trainees.
"Mobile apps are incredibly convenient because they provide a means of accessing information while on the go," states Jonathan Spicer, a thoracic trainee at M.D. Anderson Cancer Center in Houston.
He continues, "Having the capacity to look up helpful information from my phone while in the operating room, on the ward, or in transit is particularly helpful."
In this article, we aim to highlight some of the more exciting and innovative mobile software programs available today for those interested in expanding their knowledge in cardiothoracic surgery or looking for an easy-to-access resource.
iBronch (Edward Bender), $0.99: iBronch is one of the many outstanding thoracic surgical apps developed by Ed Bender. This program aims to guide learners through the basic anatomy of the trachea and bronchial tree, with correlation of simultaneous images from a fiberoptic bronchoscope and along an anatomic airway diagram. Branches of the pulmonary tree are labeled on the schematic and the bronchoscopy images. This app is particularly useful for those trainees gaining comfort with bronchoscopic procedures; however, its utility may be less significant for more advanced learners. Regardless, this is a great program, quite helpful for the intended audience.
Thoracic Lymph Node Map (RADIOLOGiQ, LLC), Free: This app provides a color-coded lymph node map, associated with computed tomography images and adapted from the International Association for the Study of Lung Cancer (IASLC) lung cancer project. This program provides excellent illustrations of the anatomic definitions for each of the intrathoracic lymph node stations. This is helpful both in examining imaging studies of actual patients and in the operating room.
CT Journals (Edward Bender), Free: This software program serves as a scholarly journal aggregator for the field of cardiothoracic surgery. The app displays feeds for journals of interest, with inclusion of those relevant periodicals with the most readership and highest impact factors. Not only can one access the articles while online, abstracts can be saved for future use offline. This is a great resource, but users should be aware that access to the full articles is available only for those who have active accounts providing them access to the specific journals.
CTSNetWiki (Edward Bender), Free: Cardiothoracic Surgery Notes is an online review developed and maintained by residents in thoracic surgical training. This resource is a tremendous repository of information, compiling graphics, text, and other multimedia content on a breadth of topics. This app allows general review of a wide variety of cardiothoracic surgical problems and is appropriate for both the novice learner and as a review for those who are further along in their training.
SESATS IX (Edward Bender), Free: Perhaps the most valuable mobile software application out there, the Self Education Self Assessment in Thoracic Surgery (SESATS) IX application contains actual questions from previous versions of the SESATS. The mobile app even includes the associated images, videos, and CT scans that correspond with the questions. Although the program does not contain the latest version of SESATS, the utility of this app cannot be overestimated. This program is enormously helpful for self-testing, on-the-go topic-specific learning, and exam preparation. This is a real gem of a find, and it comes with a strong recommendation to all trainees for its download and use.
TSRA Primer of Cardiothoracic Surgery (Thoracic Surgery Residents' Association), $4.99: Produced by CT residents for CT residents, this is probably the most useful resource for the intern, junior resident, or new fellow who needs to brush up on the basics before rounds, in between consults, and before assisting in the OR. It's not comprehensive, but it is full of clinical pearls covering all the major divisions of cardiothoracic surgery. Many cardiac residents, especially younger integrated residents, have been waiting for a straightforward, practical tutorial like this for years. Beautiful and often interactive illustrations and videos really make this iBook memorable.
NCCN Guidelines (TIP Medical Communications), Free: Available for Android and iOS, this compendium of NCCN guidelines for 56 cancers and cancer-related topics is indispensible for trainees. The utility of having up-to-date, in-depth guidelines for diagnosis and staging of all commonly encountered malignancies cannot be overstated. Additional topics ranging from management of cancer-related emesis to lung cancer screening guidelines polish it off. A must have for anyone who treats cancer, not just thoracic surgeons.
CathSource (ECGSource, LLC), $3.99: Available for Android and iOS, CathSource is a mobile app that aims to teach cardiovascular medicine fellows about coronary anatomy, angiogram projections, and catheter-based hemodynamic measurements. Luckily, CT trainees stand to benefit from the app as well. It seems to be most helpful for learning coronary anatomy on the different projections, but it also has exhaustive hemodynamic formulae and tracings for more detailed review. The app has over 30 videos of normal and abnormal findings.
EchoSource (ECGSource, LLC), $4.99:It's the same idea as above, but -- you guessed it -- for echocardiography. Both are good tools, especially for residents who teach. Both apps take simple, conceptual drawings to start and expand them with real imaging. Residents who have spent a good deal of time in learning cath and echo may find these apps less useful.
Pocket Heart (PocketAnatomy), $9.99: This is an interactive, 3D heart model with a fairly detailed presentation of cardiac anatomy and added features such as pinning quizzes and case studies. While cardiothoracic residents ought to have the anatomy down, the app can be used to teach patients and families about various anatomical aspects of cardiac disease. The graphics leave a little to be desired, but anyone who teaches medical students or patients frequently will enjoy having this easy-to-understand tool handy.
This list of cardiothoracic-specific mobile applications is by no means exhaustive. Each physician's needs will be different, and the options are countless. Countless risk calculators, mnemonic databases, formula compendiums, and pharmacologic formularies clutter the app store. Note-taking suites such as OneNote and Evernote (personal favorite of both of the authors) can help turn the most hare-brained resident into a paragon of organizational excellence. Journal citation managers such as EndNote, Dropbox, Mendeley, and Yep can help organize and manage that virtual pile of unread but probably important journal articles that keeps building up in your inbox. Even the humble iBooks app can be used to read and mark up pdf files on the fly, all while syncing with your library on your home computer. Many hospital EMRs have mobile platforms with various levels of functionality for tablets and smartphones.
What is obvious is that mobile computing technology is rapidly changing medicine and surgery in many ways. Although each one of us strives to be a complete physician, utterly self-reliant and assured of one's clinical knowledge, we all must learn the basics first. Whether at the bus stop, in a resident lounge, or in the operating room, these mobile technologies help us to learn more efficiently while on the go.
If there are any gems we have forgotten to highlight, please send an e-mail to Thoracic Surgery News and we will try to present them in the future. We hope that the residents reading this column can find a new app they didn't know they needed, one that will energize them and push their learning to a new height. Just don't forget to look up once in a while.
Dr. Antonoff is a 2nd-year, Thoracic-track trainee at Washington University in St Louis. Dr. Zeigler is a 3rd-year, integrated Cardiothoracic Surgery trainee at Stanford (Calif.) University. They reported no relevant financial conflicts.
Social media liability
Question: Which of the following is incorrect?
A. Medical malpractice lawsuits arising out of social media interactions are still uncommon.
B. Comments shared by an ex-employee with friends on Facebook may breach doctor-patient confidentiality, with liability imputed to the doctor-employer.
C. Using the same platform, a doctor must promptly rebut disparaging comments on Yelp in order to protect his or her reputation.
D. An employment contract should cover matters concerning confidentiality and privacy.
E. Staff should use office computers only for work-related activities.
Answer: C. Physicians’ widespread use of social media sites such as Facebook, LinkedIn, and Twitter has spawned novel issues of professional liability. Use of such media, augmented by ubiquitous mobile devices such as smartphones and tablets, typically involves physician-to-physician and physician-to-patient communications but may also be personal in nature. Many patients have approached their doctors to "friend" them on Facebook. About a third of all doctors are said to have received such requests, and about a quarter have accepted. Other doctors are regular or occasional bloggers, offering views both medical and nonmedical.
While embracing the immense value of social media, the physician must remain mindful of the legal and ethical risks that such networking poses. State medical boards are facing increasing complaints of online professional breach, and civil lawsuits can be expected to mount.
Allegations of medical malpractice can arise if there is a showing that negligent conduct has caused an injury of some kind. Though currently uncommon, one can expect such lawsuits to proliferate. To be sure, there will be arguments about whether there exists a doctor-patient relationship from which a duty of care arises (Internal Medicine News, "Liability in the Internet Age," April 15, 2011, p. 74), but liability can come about in unexpected ways.
In a recent Massachusetts case, a pediatrician faced a malpractice suit that alleged a failure to diagnose diabetes and diabetic ketoacidosis. An offer to settle followed quickly once it was realized that the plaintiff’s attorney had discovered the defendant’s publicly blogged details about his deposition and trial preparation (American Medical News, "Internet won’t protect your secret identity," Aug. 13, 2007) Lesson: Use the blogosphere to educate, not vent; and never presume to successfully hide behind the veil of anonymity.
There are other legal issues. For example, some state employment laws forbid navigating the Internet in search of an applicant’s medical or criminal history, as such searches are permissible only after a tentative job offer has been made.
Another legal issue involves staff who use office computers or mobile devices for personal activities. This should be pointedly forbidden, as any negligence may be imputed to the doctor under the doctrine of vicarious liability. Current or former staff may unwittingly or even intentionally disclose confidential details of patients. So, as a risk-management strategy, employment contracts should address all of these matters proactively.
In addition to civil suits by an aggrieved patient and/or family, the doctor may face civil and criminal sanctions under the federal Health Insurance Portability and Accountability Act (HIPAA) and other statutes. All professional liability carriers are keen to assist their insured members in formulating office policies and procedures that govern privacy, confidentiality, and disclosure, and practitioners should take advantage of this service.
The ethics surrounding social media typically center on privacy;, for example, should a liver transplant physician use social media to ferret out a patient’s recent drinking habits?
Where there is professional misconduct arising out of Internet postings, a state medical board may launch an investigation. A preliminary study indicates that the most common violations are inappropriate patient communication of a sexual nature, Internet prescribing for unknown individuals, and online misrepresentation of credentials (JAMA 2012;307:1141-2).
In a recent illustrative article, the same authors posed 10 hypothetical scenarios to determine the need for disciplinary action (Ann. Int. Med. 2013;158:124-30). The evaluators deemed 4 of the 10 definitely worthy of investigation: misleading information about clinical outcomes, using images without consent, misrepresenting credentials, and inappropriately contacting patients. Other vignettes thought to be probably reprehensible were the depiction of alcohol intoxication, violating patient confidentiality, and using discriminatory speech. There was even concern raised regarding derogatory speech toward patients, showing alcohol use without intoxication, and providing clinical narratives without violating confidentiality.
Errant behavior may be observed early in one’s training. Most medical schools have identified instances of unprofessional student online postings such as breaching patient confidentiality, using profane or discriminatory language, depiction of intoxication, and sexually suggestive material (JAMA 2009;302:1309-15).
It may be impossible to separate personal from professional use of social media, so it has been suggested that ethical guidelines be framed in terms of appropriateness rather than boundaries (JAMA 2013;310:581-2).
Physicians must remain mindful that their online postings are searchable and permanent, notwithstanding the facade of anonymity. Venting of frustration or work stress is rarely justified in the public domain of the Internet.
One doctor, reportedly with some 3,000 followers, gained recent notoriety – and criticism – with his sarcasm, profanity, and patient-bashing through his tweets. Another was fined $500 and lost hospital privileges for posting information traceable to a specific person, despite not divulging the patient’s identity (American Medical News, "Anonymous posts: Liberating or unprofessional?" July 11, 2011).
Recognizing the growing prevalence of doctors’ participation on social media, a growing number of professional organizations – including the American Medical Association, the American College of Physicians, and the Mayo Clinic, among others – have offered guidelines in this area. Most relevantly, the Federation of State Medical Boards, a national nonprofit organization representing the 70 medical and osteopathic boards of the United States and its territories, has published a reader-friendly report entitled, "Model Policy Guidelines for the Appropriate Use of Social Media and Social Networking in Medical Practice."
Then there is the patient who posts negative comments about his or her doctor, say, on Yelp. Occasionally, these comments are derogatory, even defamatory. Such online attacks are difficult to counter, but engaging in an online war is more likely to be aggravating than salutary and adds unwanted publicity.
The preferred way is to attempt to identify the source and to request that the material be removed from the website, either by the poster or the domain host. If a simple request fails, an attorney’s letter, a subpoena, or a judge’s restraining order may be warranted. Occasionally, a defamation suit, even if time consuming and expensive, may prove necessary – and successful.
Dr. Tan is professor emeritus of medicine and former adjunct professor of law at the University of Hawaii. This article is meant to be educational and does not constitute medical, ethical, or legal advice. It is adapted from the author’s book, "Medical Malpractice: Understanding the Law, Managing the Risk" (2006).
Question: Which of the following is incorrect?
A. Medical malpractice lawsuits arising out of social media interactions are still uncommon.
B. Comments shared by an ex-employee with friends on Facebook may breach doctor-patient confidentiality, with liability imputed to the doctor-employer.
C. Using the same platform, a doctor must promptly rebut disparaging comments on Yelp in order to protect his or her reputation.
D. An employment contract should cover matters concerning confidentiality and privacy.
E. Staff should use office computers only for work-related activities.
Answer: C. Physicians’ widespread use of social media sites such as Facebook, LinkedIn, and Twitter has spawned novel issues of professional liability. Use of such media, augmented by ubiquitous mobile devices such as smartphones and tablets, typically involves physician-to-physician and physician-to-patient communications but may also be personal in nature. Many patients have approached their doctors to "friend" them on Facebook. About a third of all doctors are said to have received such requests, and about a quarter have accepted. Other doctors are regular or occasional bloggers, offering views both medical and nonmedical.
While embracing the immense value of social media, the physician must remain mindful of the legal and ethical risks that such networking poses. State medical boards are facing increasing complaints of online professional breach, and civil lawsuits can be expected to mount.
Allegations of medical malpractice can arise if there is a showing that negligent conduct has caused an injury of some kind. Though currently uncommon, one can expect such lawsuits to proliferate. To be sure, there will be arguments about whether there exists a doctor-patient relationship from which a duty of care arises (Internal Medicine News, "Liability in the Internet Age," April 15, 2011, p. 74), but liability can come about in unexpected ways.
In a recent Massachusetts case, a pediatrician faced a malpractice suit that alleged a failure to diagnose diabetes and diabetic ketoacidosis. An offer to settle followed quickly once it was realized that the plaintiff’s attorney had discovered the defendant’s publicly blogged details about his deposition and trial preparation (American Medical News, "Internet won’t protect your secret identity," Aug. 13, 2007) Lesson: Use the blogosphere to educate, not vent; and never presume to successfully hide behind the veil of anonymity.
There are other legal issues. For example, some state employment laws forbid navigating the Internet in search of an applicant’s medical or criminal history, as such searches are permissible only after a tentative job offer has been made.
Another legal issue involves staff who use office computers or mobile devices for personal activities. This should be pointedly forbidden, as any negligence may be imputed to the doctor under the doctrine of vicarious liability. Current or former staff may unwittingly or even intentionally disclose confidential details of patients. So, as a risk-management strategy, employment contracts should address all of these matters proactively.
In addition to civil suits by an aggrieved patient and/or family, the doctor may face civil and criminal sanctions under the federal Health Insurance Portability and Accountability Act (HIPAA) and other statutes. All professional liability carriers are keen to assist their insured members in formulating office policies and procedures that govern privacy, confidentiality, and disclosure, and practitioners should take advantage of this service.
The ethics surrounding social media typically center on privacy;, for example, should a liver transplant physician use social media to ferret out a patient’s recent drinking habits?
Where there is professional misconduct arising out of Internet postings, a state medical board may launch an investigation. A preliminary study indicates that the most common violations are inappropriate patient communication of a sexual nature, Internet prescribing for unknown individuals, and online misrepresentation of credentials (JAMA 2012;307:1141-2).
In a recent illustrative article, the same authors posed 10 hypothetical scenarios to determine the need for disciplinary action (Ann. Int. Med. 2013;158:124-30). The evaluators deemed 4 of the 10 definitely worthy of investigation: misleading information about clinical outcomes, using images without consent, misrepresenting credentials, and inappropriately contacting patients. Other vignettes thought to be probably reprehensible were the depiction of alcohol intoxication, violating patient confidentiality, and using discriminatory speech. There was even concern raised regarding derogatory speech toward patients, showing alcohol use without intoxication, and providing clinical narratives without violating confidentiality.
Errant behavior may be observed early in one’s training. Most medical schools have identified instances of unprofessional student online postings such as breaching patient confidentiality, using profane or discriminatory language, depiction of intoxication, and sexually suggestive material (JAMA 2009;302:1309-15).
It may be impossible to separate personal from professional use of social media, so it has been suggested that ethical guidelines be framed in terms of appropriateness rather than boundaries (JAMA 2013;310:581-2).
Physicians must remain mindful that their online postings are searchable and permanent, notwithstanding the facade of anonymity. Venting of frustration or work stress is rarely justified in the public domain of the Internet.
One doctor, reportedly with some 3,000 followers, gained recent notoriety – and criticism – with his sarcasm, profanity, and patient-bashing through his tweets. Another was fined $500 and lost hospital privileges for posting information traceable to a specific person, despite not divulging the patient’s identity (American Medical News, "Anonymous posts: Liberating or unprofessional?" July 11, 2011).
Recognizing the growing prevalence of doctors’ participation on social media, a growing number of professional organizations – including the American Medical Association, the American College of Physicians, and the Mayo Clinic, among others – have offered guidelines in this area. Most relevantly, the Federation of State Medical Boards, a national nonprofit organization representing the 70 medical and osteopathic boards of the United States and its territories, has published a reader-friendly report entitled, "Model Policy Guidelines for the Appropriate Use of Social Media and Social Networking in Medical Practice."
Then there is the patient who posts negative comments about his or her doctor, say, on Yelp. Occasionally, these comments are derogatory, even defamatory. Such online attacks are difficult to counter, but engaging in an online war is more likely to be aggravating than salutary and adds unwanted publicity.
The preferred way is to attempt to identify the source and to request that the material be removed from the website, either by the poster or the domain host. If a simple request fails, an attorney’s letter, a subpoena, or a judge’s restraining order may be warranted. Occasionally, a defamation suit, even if time consuming and expensive, may prove necessary – and successful.
Dr. Tan is professor emeritus of medicine and former adjunct professor of law at the University of Hawaii. This article is meant to be educational and does not constitute medical, ethical, or legal advice. It is adapted from the author’s book, "Medical Malpractice: Understanding the Law, Managing the Risk" (2006).
Question: Which of the following is incorrect?
A. Medical malpractice lawsuits arising out of social media interactions are still uncommon.
B. Comments shared by an ex-employee with friends on Facebook may breach doctor-patient confidentiality, with liability imputed to the doctor-employer.
C. Using the same platform, a doctor must promptly rebut disparaging comments on Yelp in order to protect his or her reputation.
D. An employment contract should cover matters concerning confidentiality and privacy.
E. Staff should use office computers only for work-related activities.
Answer: C. Physicians’ widespread use of social media sites such as Facebook, LinkedIn, and Twitter has spawned novel issues of professional liability. Use of such media, augmented by ubiquitous mobile devices such as smartphones and tablets, typically involves physician-to-physician and physician-to-patient communications but may also be personal in nature. Many patients have approached their doctors to "friend" them on Facebook. About a third of all doctors are said to have received such requests, and about a quarter have accepted. Other doctors are regular or occasional bloggers, offering views both medical and nonmedical.
While embracing the immense value of social media, the physician must remain mindful of the legal and ethical risks that such networking poses. State medical boards are facing increasing complaints of online professional breach, and civil lawsuits can be expected to mount.
Allegations of medical malpractice can arise if there is a showing that negligent conduct has caused an injury of some kind. Though currently uncommon, one can expect such lawsuits to proliferate. To be sure, there will be arguments about whether there exists a doctor-patient relationship from which a duty of care arises (Internal Medicine News, "Liability in the Internet Age," April 15, 2011, p. 74), but liability can come about in unexpected ways.
In a recent Massachusetts case, a pediatrician faced a malpractice suit that alleged a failure to diagnose diabetes and diabetic ketoacidosis. An offer to settle followed quickly once it was realized that the plaintiff’s attorney had discovered the defendant’s publicly blogged details about his deposition and trial preparation (American Medical News, "Internet won’t protect your secret identity," Aug. 13, 2007) Lesson: Use the blogosphere to educate, not vent; and never presume to successfully hide behind the veil of anonymity.
There are other legal issues. For example, some state employment laws forbid navigating the Internet in search of an applicant’s medical or criminal history, as such searches are permissible only after a tentative job offer has been made.
Another legal issue involves staff who use office computers or mobile devices for personal activities. This should be pointedly forbidden, as any negligence may be imputed to the doctor under the doctrine of vicarious liability. Current or former staff may unwittingly or even intentionally disclose confidential details of patients. So, as a risk-management strategy, employment contracts should address all of these matters proactively.
In addition to civil suits by an aggrieved patient and/or family, the doctor may face civil and criminal sanctions under the federal Health Insurance Portability and Accountability Act (HIPAA) and other statutes. All professional liability carriers are keen to assist their insured members in formulating office policies and procedures that govern privacy, confidentiality, and disclosure, and practitioners should take advantage of this service.
The ethics surrounding social media typically center on privacy;, for example, should a liver transplant physician use social media to ferret out a patient’s recent drinking habits?
Where there is professional misconduct arising out of Internet postings, a state medical board may launch an investigation. A preliminary study indicates that the most common violations are inappropriate patient communication of a sexual nature, Internet prescribing for unknown individuals, and online misrepresentation of credentials (JAMA 2012;307:1141-2).
In a recent illustrative article, the same authors posed 10 hypothetical scenarios to determine the need for disciplinary action (Ann. Int. Med. 2013;158:124-30). The evaluators deemed 4 of the 10 definitely worthy of investigation: misleading information about clinical outcomes, using images without consent, misrepresenting credentials, and inappropriately contacting patients. Other vignettes thought to be probably reprehensible were the depiction of alcohol intoxication, violating patient confidentiality, and using discriminatory speech. There was even concern raised regarding derogatory speech toward patients, showing alcohol use without intoxication, and providing clinical narratives without violating confidentiality.
Errant behavior may be observed early in one’s training. Most medical schools have identified instances of unprofessional student online postings such as breaching patient confidentiality, using profane or discriminatory language, depiction of intoxication, and sexually suggestive material (JAMA 2009;302:1309-15).
It may be impossible to separate personal from professional use of social media, so it has been suggested that ethical guidelines be framed in terms of appropriateness rather than boundaries (JAMA 2013;310:581-2).
Physicians must remain mindful that their online postings are searchable and permanent, notwithstanding the facade of anonymity. Venting of frustration or work stress is rarely justified in the public domain of the Internet.
One doctor, reportedly with some 3,000 followers, gained recent notoriety – and criticism – with his sarcasm, profanity, and patient-bashing through his tweets. Another was fined $500 and lost hospital privileges for posting information traceable to a specific person, despite not divulging the patient’s identity (American Medical News, "Anonymous posts: Liberating or unprofessional?" July 11, 2011).
Recognizing the growing prevalence of doctors’ participation on social media, a growing number of professional organizations – including the American Medical Association, the American College of Physicians, and the Mayo Clinic, among others – have offered guidelines in this area. Most relevantly, the Federation of State Medical Boards, a national nonprofit organization representing the 70 medical and osteopathic boards of the United States and its territories, has published a reader-friendly report entitled, "Model Policy Guidelines for the Appropriate Use of Social Media and Social Networking in Medical Practice."
Then there is the patient who posts negative comments about his or her doctor, say, on Yelp. Occasionally, these comments are derogatory, even defamatory. Such online attacks are difficult to counter, but engaging in an online war is more likely to be aggravating than salutary and adds unwanted publicity.
The preferred way is to attempt to identify the source and to request that the material be removed from the website, either by the poster or the domain host. If a simple request fails, an attorney’s letter, a subpoena, or a judge’s restraining order may be warranted. Occasionally, a defamation suit, even if time consuming and expensive, may prove necessary – and successful.
Dr. Tan is professor emeritus of medicine and former adjunct professor of law at the University of Hawaii. This article is meant to be educational and does not constitute medical, ethical, or legal advice. It is adapted from the author’s book, "Medical Malpractice: Understanding the Law, Managing the Risk" (2006).
CT lung screen plagued by 18% overdiagnosis
An estimated 18% of the early lung cancers detected by low-dose CT screening in the National Lung Screening Trial were likely indolent and probably represent overdiagnosis, according to a report published in JAMA Internal Medicine.
The NLST found "an encouraging" 20% relative reduction in lung cancer-specific mortality among high-risk patients who were screened using low-dose CT, compared with chest radiography.
"These findings were met with enthusiasm, but before a widespread public health screening program is implemented, risks of screening also need to be considered," said Dr. Edward F. Patz Jr. of the department of radiology, Duke University Medical Center, Durham, N.C., and his associates in the NLST.
The chief risk in this case is overdiagnosis: identifying an early-stage lesion in an asymptomatic patient that would not progress or affect that patient’s long-term health.
It is likely that some of the tumors detected on low-dose CT were just such indolent cancers, and that those patients unnecessarily underwent invasive diagnostic procedures, surgical resection, and multiple follow-up studies.
To estimate how many of the detected cancers in the NLST were indolent and thus overdiagnosed, Dr. Patz and his colleagues used statistical probability methods to analyze extended follow-up data from the study.
The NLST involved 53,452 men and women aged 55-74 years who were enrolled during 2002-2004 and who had at least a 30-pack-year history of cigarette smoking.
The patients were randomly assigned to undergo lung cancer screening using either three annual low-dose CT exams or three annual single-view chest radiographs.
Mean follow-up was approximately 6 years. "At the end of the entire trial, there were 1,089 total lung cancer cases in the low-dose CT arm (649 detected by low-dose CT screening) and 969 cases in the [radiology] arm, for an excess of 120 cases. This gives [an] excess cancer rate of 18.5%," the investigators said (JAMA Intern. Med. 2013 Dec. 9 [doi: 10.1001/jamainternmed.2013.12738]).
"The data from this study suggest that ... 18% of persons in the low-dose CT arm with screen-detected lung cancer and 22% of those in the low-dose CT arm with screen-detected NSCLC [non-small cell lung cancer] may be cases of overdiagnosis," Dr. Patz and his associates said.
"In other words, if these individuals had not entered the NLST, they would not have received a lung cancer diagnosis or treatment, at least for the next 5 years."
In the future, the study authors noted, "once there are better biomarkers and imaging techniques to predict which individuals with a diagnosis of lung cancer will have more or less aggressive disease, treatment options can be optimized, and a mass screening program can become more valuable."
The National Institutes of Health supported the NSLT. No financial conflicts of interest were reported.
An estimated 18% of the early lung cancers detected by low-dose CT screening in the National Lung Screening Trial were likely indolent and probably represent overdiagnosis, according to a report published in JAMA Internal Medicine.
The NLST found "an encouraging" 20% relative reduction in lung cancer-specific mortality among high-risk patients who were screened using low-dose CT, compared with chest radiography.
"These findings were met with enthusiasm, but before a widespread public health screening program is implemented, risks of screening also need to be considered," said Dr. Edward F. Patz Jr. of the department of radiology, Duke University Medical Center, Durham, N.C., and his associates in the NLST.
The chief risk in this case is overdiagnosis: identifying an early-stage lesion in an asymptomatic patient that would not progress or affect that patient’s long-term health.
It is likely that some of the tumors detected on low-dose CT were just such indolent cancers, and that those patients unnecessarily underwent invasive diagnostic procedures, surgical resection, and multiple follow-up studies.
To estimate how many of the detected cancers in the NLST were indolent and thus overdiagnosed, Dr. Patz and his colleagues used statistical probability methods to analyze extended follow-up data from the study.
The NLST involved 53,452 men and women aged 55-74 years who were enrolled during 2002-2004 and who had at least a 30-pack-year history of cigarette smoking.
The patients were randomly assigned to undergo lung cancer screening using either three annual low-dose CT exams or three annual single-view chest radiographs.
Mean follow-up was approximately 6 years. "At the end of the entire trial, there were 1,089 total lung cancer cases in the low-dose CT arm (649 detected by low-dose CT screening) and 969 cases in the [radiology] arm, for an excess of 120 cases. This gives [an] excess cancer rate of 18.5%," the investigators said (JAMA Intern. Med. 2013 Dec. 9 [doi: 10.1001/jamainternmed.2013.12738]).
"The data from this study suggest that ... 18% of persons in the low-dose CT arm with screen-detected lung cancer and 22% of those in the low-dose CT arm with screen-detected NSCLC [non-small cell lung cancer] may be cases of overdiagnosis," Dr. Patz and his associates said.
"In other words, if these individuals had not entered the NLST, they would not have received a lung cancer diagnosis or treatment, at least for the next 5 years."
In the future, the study authors noted, "once there are better biomarkers and imaging techniques to predict which individuals with a diagnosis of lung cancer will have more or less aggressive disease, treatment options can be optimized, and a mass screening program can become more valuable."
The National Institutes of Health supported the NSLT. No financial conflicts of interest were reported.
An estimated 18% of the early lung cancers detected by low-dose CT screening in the National Lung Screening Trial were likely indolent and probably represent overdiagnosis, according to a report published in JAMA Internal Medicine.
The NLST found "an encouraging" 20% relative reduction in lung cancer-specific mortality among high-risk patients who were screened using low-dose CT, compared with chest radiography.
"These findings were met with enthusiasm, but before a widespread public health screening program is implemented, risks of screening also need to be considered," said Dr. Edward F. Patz Jr. of the department of radiology, Duke University Medical Center, Durham, N.C., and his associates in the NLST.
The chief risk in this case is overdiagnosis: identifying an early-stage lesion in an asymptomatic patient that would not progress or affect that patient’s long-term health.
It is likely that some of the tumors detected on low-dose CT were just such indolent cancers, and that those patients unnecessarily underwent invasive diagnostic procedures, surgical resection, and multiple follow-up studies.
To estimate how many of the detected cancers in the NLST were indolent and thus overdiagnosed, Dr. Patz and his colleagues used statistical probability methods to analyze extended follow-up data from the study.
The NLST involved 53,452 men and women aged 55-74 years who were enrolled during 2002-2004 and who had at least a 30-pack-year history of cigarette smoking.
The patients were randomly assigned to undergo lung cancer screening using either three annual low-dose CT exams or three annual single-view chest radiographs.
Mean follow-up was approximately 6 years. "At the end of the entire trial, there were 1,089 total lung cancer cases in the low-dose CT arm (649 detected by low-dose CT screening) and 969 cases in the [radiology] arm, for an excess of 120 cases. This gives [an] excess cancer rate of 18.5%," the investigators said (JAMA Intern. Med. 2013 Dec. 9 [doi: 10.1001/jamainternmed.2013.12738]).
"The data from this study suggest that ... 18% of persons in the low-dose CT arm with screen-detected lung cancer and 22% of those in the low-dose CT arm with screen-detected NSCLC [non-small cell lung cancer] may be cases of overdiagnosis," Dr. Patz and his associates said.
"In other words, if these individuals had not entered the NLST, they would not have received a lung cancer diagnosis or treatment, at least for the next 5 years."
In the future, the study authors noted, "once there are better biomarkers and imaging techniques to predict which individuals with a diagnosis of lung cancer will have more or less aggressive disease, treatment options can be optimized, and a mass screening program can become more valuable."
The National Institutes of Health supported the NSLT. No financial conflicts of interest were reported.
Postop atrial fib onset occurs at two distinct times
DALLAS -- New-onset postoperative atrial fibrillation following cardiac surgery occurs in two distinct phases, with different risk factors for each.
That's the key finding in an analysis of the recent experience at the University of Alabama, Birmingham, where new-onset postoperative AF occurred in 27% of 1,583 consecutive patients who underwent coronary artery bypass graft and/or valve surgery.
The first peak in the onset of postoperative AF occurred in the first 3 hours after surgery. This was followed by a sharp decline in incidence over the next 24 hours. The second peak happened at postoperative day 2, followed by a more gradual tail off, Dr. Spencer J. Melby reported at the American Heart Association scientific sessions.
Ninety-six percent of all cases of postoperative AF in this retrospective chart review began within the first 7 days after surgery. Of the 423 patients who developed this common and vexing condition, 16% did so within the first 24 hours, 48% had their onset at 24-72 hours, and 36% did so after 72 hours, according to Dr. Melby, a cardiothoracic surgeon at the university.
In a multivariate analysis, the most potent risk factor for onset of postoperative AF during the first phase was mitral valve repair or replacement, which was associated with a 2.5-fold increased risk. The other significant risk factors for early-onset AF were older age -- with 70-year-olds having a 1.6-fold greater risk than 50-year-olds -- and longer ischemic time, with patients whose cross-clamp time was 70 minutes having a 1.3-fold greater risk compared with those who had a cross-clamp time of 35 minutes.
Advanced age was also a risk factor for late-phase onset of AF, but it was a stronger risk factor there than for early onset AF, with 70-year-olds being at threefold greater risk than 50-year-olds -- nearly twice as great a risk as for early-onset postoperative AF. The other significant risk factors for late-phase postoperative AF in a multivariate analysis were white race, with a 1.9-fold increased risk, and heavier body weight, with patients weighing 100 kg being at 1.6-fold greater risk than those tipping the scales at 70 kg.
"We believe that these different risk factors at each time phase are indicative of change over time in the mechanisms that drive postoperative atrial fibrillation," the surgeon said.
The first peak may be the result of tissue trauma, which is typically greater with more complex operations having longer cross-clamp times. And mitral valve procedures often entail contact with the left atrium.
"We?ve found that in patients with onset in the second peak there?s a polymorphonuclear leukocyte riot going on in the pericardial space after surgery. Inflammation is high, oxidative stress is high. Troponin levels are extremely high in the pericardial fluid, so there are problems in the heart muscle itself. The body is trying to heal and may be overdoing it as reflected by the inflammation in that space," Dr. Melby explained.
Dr. Brendan M. Everett, session cochair, noted that postoperative day 2 is when a host of process-of-care issues potentially relevant to postoperative AF come into play.
"Postop day 2 is when pain control begins to slip a bit. Patients are mobilize; chest tubes are pulled. A lot of factors are going on that are very important overall to getting a patient up and out of the hospital but are serious stimuli -- adrenergic and otherwise -- that may force a patient to go into atrial fibrillation," observed Dr. Everett, director of the general cardiology inpatient service at Brigham and Women's Hospital, Boston.
Audience members praised Dr. Melby's study as having "tremendous implications" for clinical practice, especially with regard to patient management guidelines. Dr. Melby noted that postoperative AF is a costly matter: It increases hospital length of stay by 1-2 days at a cost of roughly $10,000 per episode. Nationally, postoperative AF costs $4 billion per year.
Dr. Melby reported having no financial conflicts regarding his study.
DALLAS -- New-onset postoperative atrial fibrillation following cardiac surgery occurs in two distinct phases, with different risk factors for each.
That's the key finding in an analysis of the recent experience at the University of Alabama, Birmingham, where new-onset postoperative AF occurred in 27% of 1,583 consecutive patients who underwent coronary artery bypass graft and/or valve surgery.
The first peak in the onset of postoperative AF occurred in the first 3 hours after surgery. This was followed by a sharp decline in incidence over the next 24 hours. The second peak happened at postoperative day 2, followed by a more gradual tail off, Dr. Spencer J. Melby reported at the American Heart Association scientific sessions.
Ninety-six percent of all cases of postoperative AF in this retrospective chart review began within the first 7 days after surgery. Of the 423 patients who developed this common and vexing condition, 16% did so within the first 24 hours, 48% had their onset at 24-72 hours, and 36% did so after 72 hours, according to Dr. Melby, a cardiothoracic surgeon at the university.
In a multivariate analysis, the most potent risk factor for onset of postoperative AF during the first phase was mitral valve repair or replacement, which was associated with a 2.5-fold increased risk. The other significant risk factors for early-onset AF were older age -- with 70-year-olds having a 1.6-fold greater risk than 50-year-olds -- and longer ischemic time, with patients whose cross-clamp time was 70 minutes having a 1.3-fold greater risk compared with those who had a cross-clamp time of 35 minutes.
Advanced age was also a risk factor for late-phase onset of AF, but it was a stronger risk factor there than for early onset AF, with 70-year-olds being at threefold greater risk than 50-year-olds -- nearly twice as great a risk as for early-onset postoperative AF. The other significant risk factors for late-phase postoperative AF in a multivariate analysis were white race, with a 1.9-fold increased risk, and heavier body weight, with patients weighing 100 kg being at 1.6-fold greater risk than those tipping the scales at 70 kg.
"We believe that these different risk factors at each time phase are indicative of change over time in the mechanisms that drive postoperative atrial fibrillation," the surgeon said.
The first peak may be the result of tissue trauma, which is typically greater with more complex operations having longer cross-clamp times. And mitral valve procedures often entail contact with the left atrium.
"We?ve found that in patients with onset in the second peak there?s a polymorphonuclear leukocyte riot going on in the pericardial space after surgery. Inflammation is high, oxidative stress is high. Troponin levels are extremely high in the pericardial fluid, so there are problems in the heart muscle itself. The body is trying to heal and may be overdoing it as reflected by the inflammation in that space," Dr. Melby explained.
Dr. Brendan M. Everett, session cochair, noted that postoperative day 2 is when a host of process-of-care issues potentially relevant to postoperative AF come into play.
"Postop day 2 is when pain control begins to slip a bit. Patients are mobilize; chest tubes are pulled. A lot of factors are going on that are very important overall to getting a patient up and out of the hospital but are serious stimuli -- adrenergic and otherwise -- that may force a patient to go into atrial fibrillation," observed Dr. Everett, director of the general cardiology inpatient service at Brigham and Women's Hospital, Boston.
Audience members praised Dr. Melby's study as having "tremendous implications" for clinical practice, especially with regard to patient management guidelines. Dr. Melby noted that postoperative AF is a costly matter: It increases hospital length of stay by 1-2 days at a cost of roughly $10,000 per episode. Nationally, postoperative AF costs $4 billion per year.
Dr. Melby reported having no financial conflicts regarding his study.
DALLAS -- New-onset postoperative atrial fibrillation following cardiac surgery occurs in two distinct phases, with different risk factors for each.
That's the key finding in an analysis of the recent experience at the University of Alabama, Birmingham, where new-onset postoperative AF occurred in 27% of 1,583 consecutive patients who underwent coronary artery bypass graft and/or valve surgery.
The first peak in the onset of postoperative AF occurred in the first 3 hours after surgery. This was followed by a sharp decline in incidence over the next 24 hours. The second peak happened at postoperative day 2, followed by a more gradual tail off, Dr. Spencer J. Melby reported at the American Heart Association scientific sessions.
Ninety-six percent of all cases of postoperative AF in this retrospective chart review began within the first 7 days after surgery. Of the 423 patients who developed this common and vexing condition, 16% did so within the first 24 hours, 48% had their onset at 24-72 hours, and 36% did so after 72 hours, according to Dr. Melby, a cardiothoracic surgeon at the university.
In a multivariate analysis, the most potent risk factor for onset of postoperative AF during the first phase was mitral valve repair or replacement, which was associated with a 2.5-fold increased risk. The other significant risk factors for early-onset AF were older age -- with 70-year-olds having a 1.6-fold greater risk than 50-year-olds -- and longer ischemic time, with patients whose cross-clamp time was 70 minutes having a 1.3-fold greater risk compared with those who had a cross-clamp time of 35 minutes.
Advanced age was also a risk factor for late-phase onset of AF, but it was a stronger risk factor there than for early onset AF, with 70-year-olds being at threefold greater risk than 50-year-olds -- nearly twice as great a risk as for early-onset postoperative AF. The other significant risk factors for late-phase postoperative AF in a multivariate analysis were white race, with a 1.9-fold increased risk, and heavier body weight, with patients weighing 100 kg being at 1.6-fold greater risk than those tipping the scales at 70 kg.
"We believe that these different risk factors at each time phase are indicative of change over time in the mechanisms that drive postoperative atrial fibrillation," the surgeon said.
The first peak may be the result of tissue trauma, which is typically greater with more complex operations having longer cross-clamp times. And mitral valve procedures often entail contact with the left atrium.
"We?ve found that in patients with onset in the second peak there?s a polymorphonuclear leukocyte riot going on in the pericardial space after surgery. Inflammation is high, oxidative stress is high. Troponin levels are extremely high in the pericardial fluid, so there are problems in the heart muscle itself. The body is trying to heal and may be overdoing it as reflected by the inflammation in that space," Dr. Melby explained.
Dr. Brendan M. Everett, session cochair, noted that postoperative day 2 is when a host of process-of-care issues potentially relevant to postoperative AF come into play.
"Postop day 2 is when pain control begins to slip a bit. Patients are mobilize; chest tubes are pulled. A lot of factors are going on that are very important overall to getting a patient up and out of the hospital but are serious stimuli -- adrenergic and otherwise -- that may force a patient to go into atrial fibrillation," observed Dr. Everett, director of the general cardiology inpatient service at Brigham and Women's Hospital, Boston.
Audience members praised Dr. Melby's study as having "tremendous implications" for clinical practice, especially with regard to patient management guidelines. Dr. Melby noted that postoperative AF is a costly matter: It increases hospital length of stay by 1-2 days at a cost of roughly $10,000 per episode. Nationally, postoperative AF costs $4 billion per year.
Dr. Melby reported having no financial conflicts regarding his study.
2014 budget: Less $ for ACA, IPAB
A $1 trillion spending bill that funds the government through Sept. 30 restores money for some health programs but delivers a blow to the Affordable Care Act.
Even so, President Obama is expected to sign it.
The House approved the Consolidated Appropriations Act for FY 2014 (H.R. 3547) on Jan. 15 by a vote of 359-67, and the Senate approved it a day later 72-26.
Republican members of the House Appropriations Committee added language to the bill that puts a hold on any new funding for the ACA in fiscal 2014 and takes $1 billion out of the law’s Prevention and Public Health Fund. The fund is hotly contested and has been labeled a "slush fund," by opponents.
The House Appropriations panel also succeeded in cutting $10 million in funding for the Independent Payment Advisory Board (IPAB). The IPAB was due to make its first recommendations by mid-January; however, the Obama administration has yet to appoint any members to the Board.
The spending bill continues to ban the use federal funds for needle exchanges; for research that creates or uses embryos; and for abortion, except in the case of rape, incest, or endangerment of the life of the mother.
The bill includes a $3.7 billion budget for the Centers for Medicare and Medicaid Services – almost $200 million less than it received in fiscal year 2013 but equal to what it would receive under sequestration. Some $305 million is earmarked for the timely processing and payment of benefits.
The bill increases funding for other federal health-related agencies. The National Institutes of Health budget was increased $1 billion, which should allow it to begin 385 clinical trials, according to the Senate Appropriations Committee.
There is new funding for the Brain Research Through Advancing of Innovative Neurotechnologies (BRAIN) Initiative, and funding for an initiative to study prevention and treatments for Alzheimer’s disease.
Mental health programs at various labor, health, and education agencies will receive $1.13 billion, an increase of $213 million, according to the Senate panel. Those programs include violence prevention and grants to schools to help train teachers and to help build a mental health workforce.
The Senate Committee estimates that new training will help add 4,375 social workers, psychologists, therapists and other mental health professionals to the behavioral health workforce. The Substance Abuse and Mental Health Services Administration received a $144 million increase in its budget, bringing it to $3.6 billion.
Funding for the Centers for Disease Control and Prevention was increased $567 million to $6.9 billion. That budget includes $30 million to support the Advanced Molecular Detection initiative, which helps the agency detect and stop infectious disease outbreaks and $160 million for the Preventive Health & Health Services Block Grant.
The legislation also includes $3.6 billion to improve the quantity and quality of health care services in medically underserved areas and populations. As part of that, $350 million is appropriated to create more than 450 new community health centers and expand services at existing ones.
A $1 trillion spending bill that funds the government through Sept. 30 restores money for some health programs but delivers a blow to the Affordable Care Act.
Even so, President Obama is expected to sign it.
The House approved the Consolidated Appropriations Act for FY 2014 (H.R. 3547) on Jan. 15 by a vote of 359-67, and the Senate approved it a day later 72-26.
Republican members of the House Appropriations Committee added language to the bill that puts a hold on any new funding for the ACA in fiscal 2014 and takes $1 billion out of the law’s Prevention and Public Health Fund. The fund is hotly contested and has been labeled a "slush fund," by opponents.
The House Appropriations panel also succeeded in cutting $10 million in funding for the Independent Payment Advisory Board (IPAB). The IPAB was due to make its first recommendations by mid-January; however, the Obama administration has yet to appoint any members to the Board.
The spending bill continues to ban the use federal funds for needle exchanges; for research that creates or uses embryos; and for abortion, except in the case of rape, incest, or endangerment of the life of the mother.
The bill includes a $3.7 billion budget for the Centers for Medicare and Medicaid Services – almost $200 million less than it received in fiscal year 2013 but equal to what it would receive under sequestration. Some $305 million is earmarked for the timely processing and payment of benefits.
The bill increases funding for other federal health-related agencies. The National Institutes of Health budget was increased $1 billion, which should allow it to begin 385 clinical trials, according to the Senate Appropriations Committee.
There is new funding for the Brain Research Through Advancing of Innovative Neurotechnologies (BRAIN) Initiative, and funding for an initiative to study prevention and treatments for Alzheimer’s disease.
Mental health programs at various labor, health, and education agencies will receive $1.13 billion, an increase of $213 million, according to the Senate panel. Those programs include violence prevention and grants to schools to help train teachers and to help build a mental health workforce.
The Senate Committee estimates that new training will help add 4,375 social workers, psychologists, therapists and other mental health professionals to the behavioral health workforce. The Substance Abuse and Mental Health Services Administration received a $144 million increase in its budget, bringing it to $3.6 billion.
Funding for the Centers for Disease Control and Prevention was increased $567 million to $6.9 billion. That budget includes $30 million to support the Advanced Molecular Detection initiative, which helps the agency detect and stop infectious disease outbreaks and $160 million for the Preventive Health & Health Services Block Grant.
The legislation also includes $3.6 billion to improve the quantity and quality of health care services in medically underserved areas and populations. As part of that, $350 million is appropriated to create more than 450 new community health centers and expand services at existing ones.
A $1 trillion spending bill that funds the government through Sept. 30 restores money for some health programs but delivers a blow to the Affordable Care Act.
Even so, President Obama is expected to sign it.
The House approved the Consolidated Appropriations Act for FY 2014 (H.R. 3547) on Jan. 15 by a vote of 359-67, and the Senate approved it a day later 72-26.
Republican members of the House Appropriations Committee added language to the bill that puts a hold on any new funding for the ACA in fiscal 2014 and takes $1 billion out of the law’s Prevention and Public Health Fund. The fund is hotly contested and has been labeled a "slush fund," by opponents.
The House Appropriations panel also succeeded in cutting $10 million in funding for the Independent Payment Advisory Board (IPAB). The IPAB was due to make its first recommendations by mid-January; however, the Obama administration has yet to appoint any members to the Board.
The spending bill continues to ban the use federal funds for needle exchanges; for research that creates or uses embryos; and for abortion, except in the case of rape, incest, or endangerment of the life of the mother.
The bill includes a $3.7 billion budget for the Centers for Medicare and Medicaid Services – almost $200 million less than it received in fiscal year 2013 but equal to what it would receive under sequestration. Some $305 million is earmarked for the timely processing and payment of benefits.
The bill increases funding for other federal health-related agencies. The National Institutes of Health budget was increased $1 billion, which should allow it to begin 385 clinical trials, according to the Senate Appropriations Committee.
There is new funding for the Brain Research Through Advancing of Innovative Neurotechnologies (BRAIN) Initiative, and funding for an initiative to study prevention and treatments for Alzheimer’s disease.
Mental health programs at various labor, health, and education agencies will receive $1.13 billion, an increase of $213 million, according to the Senate panel. Those programs include violence prevention and grants to schools to help train teachers and to help build a mental health workforce.
The Senate Committee estimates that new training will help add 4,375 social workers, psychologists, therapists and other mental health professionals to the behavioral health workforce. The Substance Abuse and Mental Health Services Administration received a $144 million increase in its budget, bringing it to $3.6 billion.
Funding for the Centers for Disease Control and Prevention was increased $567 million to $6.9 billion. That budget includes $30 million to support the Advanced Molecular Detection initiative, which helps the agency detect and stop infectious disease outbreaks and $160 million for the Preventive Health & Health Services Block Grant.
The legislation also includes $3.6 billion to improve the quantity and quality of health care services in medically underserved areas and populations. As part of that, $350 million is appropriated to create more than 450 new community health centers and expand services at existing ones.
Esophagectomy treatment response nodes provide prognostic information
ORLANDO – Tumor response nodes obtained from patients who undergo esophagectomy for early-stage adenocarcinoma provide valuable prognostic information, according to Dr. Dylan Nieman.
Such nodes – lymph nodes with evidence of neoadjuvant treatment effect without residual cancer cells – may mark the prior spread of tumor, and should be counted as positive, Dr. Nieman of the University of Rochester (N.Y.) said at the annual meeting of the Society of Thoracic Surgeons.
The current practice of ignoring these nodes likely results in systematic pathologic understaging, he explained.
In 90 patients who underwent esophagectomy after neoadjuvant therapy for esophageal adenocarcinoma, the median number of nodes found per resection specimen was 18. A total of 100 tumor response nodes without viable malignant cells were identified in 38 (42%) of the patients.
The majority of the patients with treatment response nodes had only one node detected, Dr. Nieman said.
The median survival for the entire cohort was 55.6 months, and the 5-year survival was 35%. The median survival of patients with evidence of treatment response nodes was poorer, but not significantly different from those without treatment response nodes (45.9 vs. 55.6 months), he noted.
"However, for a subset of 62 patients classified as having limited or no nodal involvement – that is, AJCC N0 or N1 – the presence of treatment response nodes was associated with significantly poorer survival. This effect remained when adjusting for patient age and [American Joint Committee on Cancer] stage (hazard ratio, 2.7)," he said.
In a subset of 46 patients with pathologic AJCC stage 2B or less, the presence of treatment response nodes was still associated with poorer survival, even after adjustment for age and AJCC stage.
"To look at this a different way, if tumor response nodes were to be counted as positive, the nodal status of 29 of these patients would be upstaged. This includes 18 of 39 patients, or 46%, who were classified as node negative, and 8 of 23 patients, or 34%, who were classified as N1 by AJCC pathological assessment," he said.
The recategorization of those 29 patients resulted in better survival for the entire group, but particularly for those in the lowest-stage group, he noted.
When investigators modeled stage-adjusted survival, the counting of tumor response nodes as positive offered a better model fit, compared with following the current practice of ignoring tumor response nodes, he explained.
Patients included in the study were identified from a prospectively collected clinical database of esophagectomy patients, and were treated with neoadjuvant therapy for esophageal adenocarcinoma between 2006 and 2011. Most (82 of 90) were men. The patients had a mean age of 62 years, and were followed for a median of 27 months. Forty patients received preoperative chemotherapy, and 50 received preoperative chemoradiation.
In all cases, pathologic resection margins were negative.
On pathologic review, the majority had T3 tumors. More than 40% were staged as node negative, and more than two-thirds were staged with N0 or N1 disease.
"Prior to neoadjuvant therapy, all of these patients were clinically staged stage 2 or stage 3, but at the time of resection after neoadjuvant therapy, by AJCC staging, 24% of the patients were stage 0 or 1, 27% were stage 2, and almost half were stage 3," he said.
The findings are notable, because the current AJCC pathologic staging for esophageal adenocarcinoma is derived from the experience of patients undergoing esophagectomy alone. This approach has unclear relevance in patients who receive multimodality therapy, which has supplanted primary surgery as the standard of care for locoregionally advanced esophageal adenocarcinoma, he said.
"Future efforts at revising the staging system for esophageal adenocarcinoma should consider treatment response lymph nodes. ... We currently have pathological staging models that are of limited usefulness for our neoadjuvantly treated population. Perhaps consideration of these nodes can help improve that," he concluded.
Dr. Nieman reported having no financial disclosures.
ORLANDO – Tumor response nodes obtained from patients who undergo esophagectomy for early-stage adenocarcinoma provide valuable prognostic information, according to Dr. Dylan Nieman.
Such nodes – lymph nodes with evidence of neoadjuvant treatment effect without residual cancer cells – may mark the prior spread of tumor, and should be counted as positive, Dr. Nieman of the University of Rochester (N.Y.) said at the annual meeting of the Society of Thoracic Surgeons.
The current practice of ignoring these nodes likely results in systematic pathologic understaging, he explained.
In 90 patients who underwent esophagectomy after neoadjuvant therapy for esophageal adenocarcinoma, the median number of nodes found per resection specimen was 18. A total of 100 tumor response nodes without viable malignant cells were identified in 38 (42%) of the patients.
The majority of the patients with treatment response nodes had only one node detected, Dr. Nieman said.
The median survival for the entire cohort was 55.6 months, and the 5-year survival was 35%. The median survival of patients with evidence of treatment response nodes was poorer, but not significantly different from those without treatment response nodes (45.9 vs. 55.6 months), he noted.
"However, for a subset of 62 patients classified as having limited or no nodal involvement – that is, AJCC N0 or N1 – the presence of treatment response nodes was associated with significantly poorer survival. This effect remained when adjusting for patient age and [American Joint Committee on Cancer] stage (hazard ratio, 2.7)," he said.
In a subset of 46 patients with pathologic AJCC stage 2B or less, the presence of treatment response nodes was still associated with poorer survival, even after adjustment for age and AJCC stage.
"To look at this a different way, if tumor response nodes were to be counted as positive, the nodal status of 29 of these patients would be upstaged. This includes 18 of 39 patients, or 46%, who were classified as node negative, and 8 of 23 patients, or 34%, who were classified as N1 by AJCC pathological assessment," he said.
The recategorization of those 29 patients resulted in better survival for the entire group, but particularly for those in the lowest-stage group, he noted.
When investigators modeled stage-adjusted survival, the counting of tumor response nodes as positive offered a better model fit, compared with following the current practice of ignoring tumor response nodes, he explained.
Patients included in the study were identified from a prospectively collected clinical database of esophagectomy patients, and were treated with neoadjuvant therapy for esophageal adenocarcinoma between 2006 and 2011. Most (82 of 90) were men. The patients had a mean age of 62 years, and were followed for a median of 27 months. Forty patients received preoperative chemotherapy, and 50 received preoperative chemoradiation.
In all cases, pathologic resection margins were negative.
On pathologic review, the majority had T3 tumors. More than 40% were staged as node negative, and more than two-thirds were staged with N0 or N1 disease.
"Prior to neoadjuvant therapy, all of these patients were clinically staged stage 2 or stage 3, but at the time of resection after neoadjuvant therapy, by AJCC staging, 24% of the patients were stage 0 or 1, 27% were stage 2, and almost half were stage 3," he said.
The findings are notable, because the current AJCC pathologic staging for esophageal adenocarcinoma is derived from the experience of patients undergoing esophagectomy alone. This approach has unclear relevance in patients who receive multimodality therapy, which has supplanted primary surgery as the standard of care for locoregionally advanced esophageal adenocarcinoma, he said.
"Future efforts at revising the staging system for esophageal adenocarcinoma should consider treatment response lymph nodes. ... We currently have pathological staging models that are of limited usefulness for our neoadjuvantly treated population. Perhaps consideration of these nodes can help improve that," he concluded.
Dr. Nieman reported having no financial disclosures.
ORLANDO – Tumor response nodes obtained from patients who undergo esophagectomy for early-stage adenocarcinoma provide valuable prognostic information, according to Dr. Dylan Nieman.
Such nodes – lymph nodes with evidence of neoadjuvant treatment effect without residual cancer cells – may mark the prior spread of tumor, and should be counted as positive, Dr. Nieman of the University of Rochester (N.Y.) said at the annual meeting of the Society of Thoracic Surgeons.
The current practice of ignoring these nodes likely results in systematic pathologic understaging, he explained.
In 90 patients who underwent esophagectomy after neoadjuvant therapy for esophageal adenocarcinoma, the median number of nodes found per resection specimen was 18. A total of 100 tumor response nodes without viable malignant cells were identified in 38 (42%) of the patients.
The majority of the patients with treatment response nodes had only one node detected, Dr. Nieman said.
The median survival for the entire cohort was 55.6 months, and the 5-year survival was 35%. The median survival of patients with evidence of treatment response nodes was poorer, but not significantly different from those without treatment response nodes (45.9 vs. 55.6 months), he noted.
"However, for a subset of 62 patients classified as having limited or no nodal involvement – that is, AJCC N0 or N1 – the presence of treatment response nodes was associated with significantly poorer survival. This effect remained when adjusting for patient age and [American Joint Committee on Cancer] stage (hazard ratio, 2.7)," he said.
In a subset of 46 patients with pathologic AJCC stage 2B or less, the presence of treatment response nodes was still associated with poorer survival, even after adjustment for age and AJCC stage.
"To look at this a different way, if tumor response nodes were to be counted as positive, the nodal status of 29 of these patients would be upstaged. This includes 18 of 39 patients, or 46%, who were classified as node negative, and 8 of 23 patients, or 34%, who were classified as N1 by AJCC pathological assessment," he said.
The recategorization of those 29 patients resulted in better survival for the entire group, but particularly for those in the lowest-stage group, he noted.
When investigators modeled stage-adjusted survival, the counting of tumor response nodes as positive offered a better model fit, compared with following the current practice of ignoring tumor response nodes, he explained.
Patients included in the study were identified from a prospectively collected clinical database of esophagectomy patients, and were treated with neoadjuvant therapy for esophageal adenocarcinoma between 2006 and 2011. Most (82 of 90) were men. The patients had a mean age of 62 years, and were followed for a median of 27 months. Forty patients received preoperative chemotherapy, and 50 received preoperative chemoradiation.
In all cases, pathologic resection margins were negative.
On pathologic review, the majority had T3 tumors. More than 40% were staged as node negative, and more than two-thirds were staged with N0 or N1 disease.
"Prior to neoadjuvant therapy, all of these patients were clinically staged stage 2 or stage 3, but at the time of resection after neoadjuvant therapy, by AJCC staging, 24% of the patients were stage 0 or 1, 27% were stage 2, and almost half were stage 3," he said.
The findings are notable, because the current AJCC pathologic staging for esophageal adenocarcinoma is derived from the experience of patients undergoing esophagectomy alone. This approach has unclear relevance in patients who receive multimodality therapy, which has supplanted primary surgery as the standard of care for locoregionally advanced esophageal adenocarcinoma, he said.
"Future efforts at revising the staging system for esophageal adenocarcinoma should consider treatment response lymph nodes. ... We currently have pathological staging models that are of limited usefulness for our neoadjuvantly treated population. Perhaps consideration of these nodes can help improve that," he concluded.
Dr. Nieman reported having no financial disclosures.
AT THE STS ANNUAL MEETING
Major finding: In 62 patients classified as having limited or no nodal involvement, the presence of treatment response nodes was associated with significantly poorer survival (adjusted hazard ratio, 2.7).
Data source: A review of 90 prospectively collected cases.
Disclosures: Dr. Nieman reported having no financial disclosures.