User login
The Official Newspaper of the American Association for Thoracic Surgery
Inpatient safety efforts yield mixed results
Adverse events decreased over the last decade for Medicare inpatients with acute myocardial infarction or heart failure but barely changed for those with pneumonia or conditions requiring surgery, according to an analysis of a Medicare database.
The improvements likely translated to 81,000 fewer adverse events for patients with acute MI (AMI) and heart failure (HF) from 2010 to 2011 alone, according to the study published Jan. 22 in the New England Journal of Medicine.
"Although this suggests that national efforts focused on patient safety have made some inroads, the lack of reductions across the board is disappointing," Yun Wang, Ph.D., of the Harvard School of Public Health, Boston, and his colleagues wrote.
The researchers examined whether hospitalized patients are any better off in light of the current focus on patient safety, including the launch of initiatives such as the American College of Surgeons’ National Surgical Quality Improvement Program and the federal government’s Surgical Infection Prevention Project.
They used three composite outcomes measures: the rate of occurrence for adverse events for which patients were at risk (for instance, only patients receiving warfarin were at risk for warfarin-related events); the proportion of patients with one or more adverse events; and the number of adverse events per 1,000 hospitalizations. They analyzed data on 61,523 patients who were discharged from 4,372 hospitals; the data were extracted from the Medicare Patient Safety Monitoring System database (N. Engl. J. Med. 2014:370;341-51).
The 61,523 patients included 11,399 with AMI, 15,374 with HF, 18,269 with pneumonia, and 16,481 with conditions requiring surgery. Postsurgical patients largely were being treated for joint replacement procedures and other osteoarthritis-related conditions, femur fracture, colon cancer, post-AMI procedures, or other forms of chronic ischemic heart disease.
From 2005-2006 to 2010-2011, AMI and HF patients saw a 1.3 percentage point decline in the rate of adverse events, from 5% to 3.7%. The proportion who had one or more such events decreased from 26% to 19%. The number of adverse events per 1,000 hospitalizations declined from 402 to 262 for AMI patients and from 235 to 167 for HF patients.
Infection-related and drug-related adverse events declined significantly in heart attack and HF patients. There was also a substantial improvement in postprocedure events in HF patients.
Postsurgical patients experienced slight increases in all three outcomes measures, in particular, increases in infection-related and postprocedural events such as venous thromboembolism, and cardiac and catheter-related events. The number of events per 1,000 hospitalizations for pneumonia patients increased insignificantly from 216 to 223. For postsurgical patients, the number of events increased insignificantly from 352/1,000 to 368/1,000.
Patients who had adverse events had significantly longer hospital stays and were at higher risk for death. As the number of adverse events increased, so did the risk of death.
The authors noted that declines in events for AMI and HF patients might be a reflection of the numerous efforts and initiatives to improve care in those two conditions.
But they also found that concerted efforts to improve safety did not necessarily work. There was an increase in pressure ulcers in postsurgical patients, and no decline in ventilator-associated pneumonia in most patients, even though there have been initiatives focused on those conditions.
"Our finding of an increased adverse-event rate among surgical patients indicates a continuing challenge and identifies an important target for patient-safety initiatives," the researchers said.
The study was supported by the Agency for Healthcare Research and Quality as well as academic and federal grants. Several researchers were associated with Qualidigm, a consultancy that administers the Medicare database. No other relevant conflicts of interest were disclosed.
On Twitter @aliciaault
Adverse events decreased over the last decade for Medicare inpatients with acute myocardial infarction or heart failure but barely changed for those with pneumonia or conditions requiring surgery, according to an analysis of a Medicare database.
The improvements likely translated to 81,000 fewer adverse events for patients with acute MI (AMI) and heart failure (HF) from 2010 to 2011 alone, according to the study published Jan. 22 in the New England Journal of Medicine.
"Although this suggests that national efforts focused on patient safety have made some inroads, the lack of reductions across the board is disappointing," Yun Wang, Ph.D., of the Harvard School of Public Health, Boston, and his colleagues wrote.
The researchers examined whether hospitalized patients are any better off in light of the current focus on patient safety, including the launch of initiatives such as the American College of Surgeons’ National Surgical Quality Improvement Program and the federal government’s Surgical Infection Prevention Project.
They used three composite outcomes measures: the rate of occurrence for adverse events for which patients were at risk (for instance, only patients receiving warfarin were at risk for warfarin-related events); the proportion of patients with one or more adverse events; and the number of adverse events per 1,000 hospitalizations. They analyzed data on 61,523 patients who were discharged from 4,372 hospitals; the data were extracted from the Medicare Patient Safety Monitoring System database (N. Engl. J. Med. 2014:370;341-51).
The 61,523 patients included 11,399 with AMI, 15,374 with HF, 18,269 with pneumonia, and 16,481 with conditions requiring surgery. Postsurgical patients largely were being treated for joint replacement procedures and other osteoarthritis-related conditions, femur fracture, colon cancer, post-AMI procedures, or other forms of chronic ischemic heart disease.
From 2005-2006 to 2010-2011, AMI and HF patients saw a 1.3 percentage point decline in the rate of adverse events, from 5% to 3.7%. The proportion who had one or more such events decreased from 26% to 19%. The number of adverse events per 1,000 hospitalizations declined from 402 to 262 for AMI patients and from 235 to 167 for HF patients.
Infection-related and drug-related adverse events declined significantly in heart attack and HF patients. There was also a substantial improvement in postprocedure events in HF patients.
Postsurgical patients experienced slight increases in all three outcomes measures, in particular, increases in infection-related and postprocedural events such as venous thromboembolism, and cardiac and catheter-related events. The number of events per 1,000 hospitalizations for pneumonia patients increased insignificantly from 216 to 223. For postsurgical patients, the number of events increased insignificantly from 352/1,000 to 368/1,000.
Patients who had adverse events had significantly longer hospital stays and were at higher risk for death. As the number of adverse events increased, so did the risk of death.
The authors noted that declines in events for AMI and HF patients might be a reflection of the numerous efforts and initiatives to improve care in those two conditions.
But they also found that concerted efforts to improve safety did not necessarily work. There was an increase in pressure ulcers in postsurgical patients, and no decline in ventilator-associated pneumonia in most patients, even though there have been initiatives focused on those conditions.
"Our finding of an increased adverse-event rate among surgical patients indicates a continuing challenge and identifies an important target for patient-safety initiatives," the researchers said.
The study was supported by the Agency for Healthcare Research and Quality as well as academic and federal grants. Several researchers were associated with Qualidigm, a consultancy that administers the Medicare database. No other relevant conflicts of interest were disclosed.
On Twitter @aliciaault
Adverse events decreased over the last decade for Medicare inpatients with acute myocardial infarction or heart failure but barely changed for those with pneumonia or conditions requiring surgery, according to an analysis of a Medicare database.
The improvements likely translated to 81,000 fewer adverse events for patients with acute MI (AMI) and heart failure (HF) from 2010 to 2011 alone, according to the study published Jan. 22 in the New England Journal of Medicine.
"Although this suggests that national efforts focused on patient safety have made some inroads, the lack of reductions across the board is disappointing," Yun Wang, Ph.D., of the Harvard School of Public Health, Boston, and his colleagues wrote.
The researchers examined whether hospitalized patients are any better off in light of the current focus on patient safety, including the launch of initiatives such as the American College of Surgeons’ National Surgical Quality Improvement Program and the federal government’s Surgical Infection Prevention Project.
They used three composite outcomes measures: the rate of occurrence for adverse events for which patients were at risk (for instance, only patients receiving warfarin were at risk for warfarin-related events); the proportion of patients with one or more adverse events; and the number of adverse events per 1,000 hospitalizations. They analyzed data on 61,523 patients who were discharged from 4,372 hospitals; the data were extracted from the Medicare Patient Safety Monitoring System database (N. Engl. J. Med. 2014:370;341-51).
The 61,523 patients included 11,399 with AMI, 15,374 with HF, 18,269 with pneumonia, and 16,481 with conditions requiring surgery. Postsurgical patients largely were being treated for joint replacement procedures and other osteoarthritis-related conditions, femur fracture, colon cancer, post-AMI procedures, or other forms of chronic ischemic heart disease.
From 2005-2006 to 2010-2011, AMI and HF patients saw a 1.3 percentage point decline in the rate of adverse events, from 5% to 3.7%. The proportion who had one or more such events decreased from 26% to 19%. The number of adverse events per 1,000 hospitalizations declined from 402 to 262 for AMI patients and from 235 to 167 for HF patients.
Infection-related and drug-related adverse events declined significantly in heart attack and HF patients. There was also a substantial improvement in postprocedure events in HF patients.
Postsurgical patients experienced slight increases in all three outcomes measures, in particular, increases in infection-related and postprocedural events such as venous thromboembolism, and cardiac and catheter-related events. The number of events per 1,000 hospitalizations for pneumonia patients increased insignificantly from 216 to 223. For postsurgical patients, the number of events increased insignificantly from 352/1,000 to 368/1,000.
Patients who had adverse events had significantly longer hospital stays and were at higher risk for death. As the number of adverse events increased, so did the risk of death.
The authors noted that declines in events for AMI and HF patients might be a reflection of the numerous efforts and initiatives to improve care in those two conditions.
But they also found that concerted efforts to improve safety did not necessarily work. There was an increase in pressure ulcers in postsurgical patients, and no decline in ventilator-associated pneumonia in most patients, even though there have been initiatives focused on those conditions.
"Our finding of an increased adverse-event rate among surgical patients indicates a continuing challenge and identifies an important target for patient-safety initiatives," the researchers said.
The study was supported by the Agency for Healthcare Research and Quality as well as academic and federal grants. Several researchers were associated with Qualidigm, a consultancy that administers the Medicare database. No other relevant conflicts of interest were disclosed.
On Twitter @aliciaault
The role of immunotherapy in NSCLC to expand
SAN DIEGO – Expect an expanded role of immunotherapy in patients with non–small cell lung cancer, Dr. Roy S. Herbst predicted at the Joint Conference on the Molecular Origins of Lung Cancer sponsored by the American Association for Cancer Research and the International Association for the Study of Lung Cancer.
Dr. Herbst, who is a professor of medical oncology at Yale University, New Haven, Conn., characterized immunotherapy as "probably the most exciting new and specific therapies we have for NSCLC. The extent in its response is impressive, and this is a therapy that has memory. The adaptability of immunotherapy is important as well."
He advised researchers and clinicians to consider using immunotherapy that includes CTLA-4 antibodies, PD-1 antibodies, and PD-L1 antibodies alone or together in patients with earlier stages of lung disease. Clinical studies of immunotherapy in NSCLC patients suggest that some patients don’t get better with immunotherapy, "but a lot of patients do," said Dr. Herbst.
"We want to figure out who those patients are. When we see activity like this, we think, can we bring this therapy to earlier disease? These agents might have a role in maintenance therapy and adjuvant/neoadjuvant therapy. Of course, we worry about side effects such as pneumonitis, which occurs rarely, but we still hope these agents will have a benefit in the adjuvant setting. The biology speaks to that. But what about using these agents as maintenance therapy? I think that needs to be explored."
Using immunotherapy as frontline treatment in patients with stage IV lung cancer is also feasible, he said. "I’d feel much better about it if we had a marker, but we should think about some single-agent trials," he said.
"Other possibilities in stage IV disease include using immunotherapy with chemotherapy and with tyrosine kinase inhibitors."
Immunotherapy-related adverse events are "not overwhelming, but they’re different than what we see with chemotherapy," Dr. Herbst continued. "For example, some of the endocrine events are not something we often see. We are working on ways to manage this."
Use of biomarkers and immune monitoring can also help clinicians gauge the efficacy of immunotherapy in their NSCLC patients.
Dr. Herbst and his associates at Yale Cancer Center follow these patients with biopsies at baseline, during therapy, and at the end of therapy, "because after their therapy at 1 year or more, you wonder: Is this active tumor? Or is this necrotic tissue?" he said. "We now have ways to figure out who is responding and why they’re responding."
Another trend being seen in the future of immunotherapy involves combining with other agents that address key mechanisms in positive and negative regulation of the immune system.
Dr. Herbst explained that the biological goal of combinations with a checkpoint inhibitor include the ability to induce antigen-specific T cells, provide more antigen-presenting cells (APCs), activation/modulation of APCs, drive T-cell expansion to expand the pool of antigen-specific cells, and remove other regulatory checkpoints/suppressive factors for T-cell activation/expansion in periphery.
"The current challenge is to identify the critical deficiencies in individual patients," he said.
"We have to continue to investigate the biologic significance of all potential ligand-receptor interactions in the tumor microenvironment."
Dr. Herbst disclosed that he is on the scientific advisory boards of Biothera, Diatech, Kolltan, N of 1, Novarx, and Quintiles. He also has done consulting for Ariad, Astellas, and other companies.
SAN DIEGO – Expect an expanded role of immunotherapy in patients with non–small cell lung cancer, Dr. Roy S. Herbst predicted at the Joint Conference on the Molecular Origins of Lung Cancer sponsored by the American Association for Cancer Research and the International Association for the Study of Lung Cancer.
Dr. Herbst, who is a professor of medical oncology at Yale University, New Haven, Conn., characterized immunotherapy as "probably the most exciting new and specific therapies we have for NSCLC. The extent in its response is impressive, and this is a therapy that has memory. The adaptability of immunotherapy is important as well."
He advised researchers and clinicians to consider using immunotherapy that includes CTLA-4 antibodies, PD-1 antibodies, and PD-L1 antibodies alone or together in patients with earlier stages of lung disease. Clinical studies of immunotherapy in NSCLC patients suggest that some patients don’t get better with immunotherapy, "but a lot of patients do," said Dr. Herbst.
"We want to figure out who those patients are. When we see activity like this, we think, can we bring this therapy to earlier disease? These agents might have a role in maintenance therapy and adjuvant/neoadjuvant therapy. Of course, we worry about side effects such as pneumonitis, which occurs rarely, but we still hope these agents will have a benefit in the adjuvant setting. The biology speaks to that. But what about using these agents as maintenance therapy? I think that needs to be explored."
Using immunotherapy as frontline treatment in patients with stage IV lung cancer is also feasible, he said. "I’d feel much better about it if we had a marker, but we should think about some single-agent trials," he said.
"Other possibilities in stage IV disease include using immunotherapy with chemotherapy and with tyrosine kinase inhibitors."
Immunotherapy-related adverse events are "not overwhelming, but they’re different than what we see with chemotherapy," Dr. Herbst continued. "For example, some of the endocrine events are not something we often see. We are working on ways to manage this."
Use of biomarkers and immune monitoring can also help clinicians gauge the efficacy of immunotherapy in their NSCLC patients.
Dr. Herbst and his associates at Yale Cancer Center follow these patients with biopsies at baseline, during therapy, and at the end of therapy, "because after their therapy at 1 year or more, you wonder: Is this active tumor? Or is this necrotic tissue?" he said. "We now have ways to figure out who is responding and why they’re responding."
Another trend being seen in the future of immunotherapy involves combining with other agents that address key mechanisms in positive and negative regulation of the immune system.
Dr. Herbst explained that the biological goal of combinations with a checkpoint inhibitor include the ability to induce antigen-specific T cells, provide more antigen-presenting cells (APCs), activation/modulation of APCs, drive T-cell expansion to expand the pool of antigen-specific cells, and remove other regulatory checkpoints/suppressive factors for T-cell activation/expansion in periphery.
"The current challenge is to identify the critical deficiencies in individual patients," he said.
"We have to continue to investigate the biologic significance of all potential ligand-receptor interactions in the tumor microenvironment."
Dr. Herbst disclosed that he is on the scientific advisory boards of Biothera, Diatech, Kolltan, N of 1, Novarx, and Quintiles. He also has done consulting for Ariad, Astellas, and other companies.
SAN DIEGO – Expect an expanded role of immunotherapy in patients with non–small cell lung cancer, Dr. Roy S. Herbst predicted at the Joint Conference on the Molecular Origins of Lung Cancer sponsored by the American Association for Cancer Research and the International Association for the Study of Lung Cancer.
Dr. Herbst, who is a professor of medical oncology at Yale University, New Haven, Conn., characterized immunotherapy as "probably the most exciting new and specific therapies we have for NSCLC. The extent in its response is impressive, and this is a therapy that has memory. The adaptability of immunotherapy is important as well."
He advised researchers and clinicians to consider using immunotherapy that includes CTLA-4 antibodies, PD-1 antibodies, and PD-L1 antibodies alone or together in patients with earlier stages of lung disease. Clinical studies of immunotherapy in NSCLC patients suggest that some patients don’t get better with immunotherapy, "but a lot of patients do," said Dr. Herbst.
"We want to figure out who those patients are. When we see activity like this, we think, can we bring this therapy to earlier disease? These agents might have a role in maintenance therapy and adjuvant/neoadjuvant therapy. Of course, we worry about side effects such as pneumonitis, which occurs rarely, but we still hope these agents will have a benefit in the adjuvant setting. The biology speaks to that. But what about using these agents as maintenance therapy? I think that needs to be explored."
Using immunotherapy as frontline treatment in patients with stage IV lung cancer is also feasible, he said. "I’d feel much better about it if we had a marker, but we should think about some single-agent trials," he said.
"Other possibilities in stage IV disease include using immunotherapy with chemotherapy and with tyrosine kinase inhibitors."
Immunotherapy-related adverse events are "not overwhelming, but they’re different than what we see with chemotherapy," Dr. Herbst continued. "For example, some of the endocrine events are not something we often see. We are working on ways to manage this."
Use of biomarkers and immune monitoring can also help clinicians gauge the efficacy of immunotherapy in their NSCLC patients.
Dr. Herbst and his associates at Yale Cancer Center follow these patients with biopsies at baseline, during therapy, and at the end of therapy, "because after their therapy at 1 year or more, you wonder: Is this active tumor? Or is this necrotic tissue?" he said. "We now have ways to figure out who is responding and why they’re responding."
Another trend being seen in the future of immunotherapy involves combining with other agents that address key mechanisms in positive and negative regulation of the immune system.
Dr. Herbst explained that the biological goal of combinations with a checkpoint inhibitor include the ability to induce antigen-specific T cells, provide more antigen-presenting cells (APCs), activation/modulation of APCs, drive T-cell expansion to expand the pool of antigen-specific cells, and remove other regulatory checkpoints/suppressive factors for T-cell activation/expansion in periphery.
"The current challenge is to identify the critical deficiencies in individual patients," he said.
"We have to continue to investigate the biologic significance of all potential ligand-receptor interactions in the tumor microenvironment."
Dr. Herbst disclosed that he is on the scientific advisory boards of Biothera, Diatech, Kolltan, N of 1, Novarx, and Quintiles. He also has done consulting for Ariad, Astellas, and other companies.
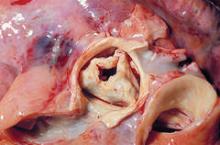
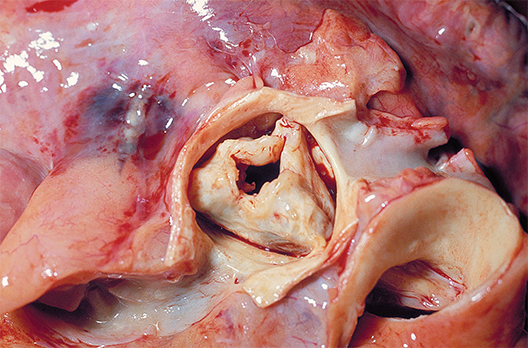
New valve guideline promotes early surgery
The updated practice guideline for managing adults with valvular heart disease has a new, "modular" format to facilitate clinicians' access to "concise, relevant bytes of information at the point of care, when clinical knowledge is needed most," according to reports published online simultaneously March 3 in Circulation and the Journal of the American College of Cardiology.
The guideline, compiled by a committee of cardiologists, interventionalists, surgeons, and anesthesiologists under the aegis of the American Heart Association and the American College of Cardiology, was last updated in 2008.
"Some recommendations from the earlier valvular heart disease guideline have been updated as warranted by new evidence or a better understanding of earlier evidence, whereas others that were inaccurate, irrelevant, or overlapping were deleted or modified," said writing committee cochairs Dr. Rick A. Nishimura of the division of cardiovascular diseases, Mayo Clinic, Rochester, Minn.; and Dr. Catherine M. Otto, director of the University of Washington Medical Center's Heart Valve Clinic, Seattle.
The narrative text of the guideline is limited, and instead it uses decision pathway diagrams and numerous summary tables of current evidence and recommendations. These include links to relevant references. It is hoped that clinicians can more easily use the new guideline as a quick reference. This format also will enable individual sections to be updated or amended as new evidence comes to light. The PDF of the guideline is available for free.
"This novel approach to evidence-based guideline development will revolutionize the clinical impact of guideline recommendations, ensuring they are always current and allowing seamless integration with electronic medical record systems," Dr. Otto said in a press statement accompanying the reports.
The guideline now includes gradations of disease severity, to help clinicians determine the optimal timing of intervention. Whether or not intervention is indicated depends on five factors: the presence or absence of symptoms, the severity of valvular heart disease, the response of the left and/or right ventricle to the volume or pressure overload caused by the valvular disease, the effect on the pulmonary or systemic circulation, and any change in heart rhythm.
Disease severity ranges from stage A, "at risk," which denotes patients who have risk factors for developing valvular heart disease; through stage B, "progressive," which indicates patients who are asymptomatic but have mildly to moderately severe disease; through stage C, "asymptomatic severe," which includes patients with severe yet still asymptomatic valvular disease in which the left or right ventricle remains compensated or in which the left or right ventricle has decompensated; to stage D, "symptomatic severe," which indicates patients whose severe valvular disease has produced symptoms.
"In patients with stenotic lesions, there is an additional category of 'very severe' stenosis based on studies of the natural history showing that prognosis becomes poorer as the severity of stenosis increases," the guideline states.
Information is provided for assessing the various disease states associated with the aortic, mitral, and tricuspid valves, and addresses the issues of valve repair, replacement, and the use of prosthetic valves.
Compared with the previous guideline, the new one suggests surgical intervention at an earlier stage for certain patients. "Due to more knowledge regarding the natural history of untreated patients with severe valvular heart disease and better outcomes from surgery, we've lowered the threshold for operation to include more patients with asymptomatic severe disease. Now, select patients with severe asymptomatic aortic stenosis and severe asymptomatic mitral regurgitation can be considered for intervention, depending on certain other factors such as operative mortality and … the ability to achieve a durable valve repair," Dr. Nishimura said in the press statement.
The new guideline also proposes a new approach to risk assessment, to be applied to all patients for whom intervention is being considered. Previous risk scoring systems were "useful but limited"; the new approach takes into consideration "procedure-specific impediments, major organ system compromise, comorbidities, patient frailty, and the Society of Thoracic Surgeons predicted risk of mortality model."
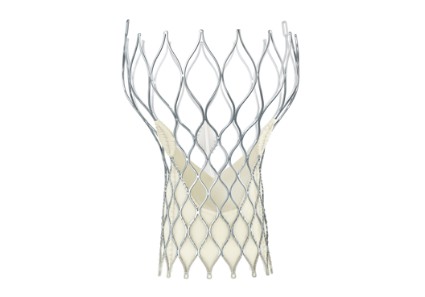
For the first time, the guideline discusses transcatheter aortic valve replacement and other catheter-based treatments, new technologies that have improved patient care but also have complicated risk assessment. Separate recommendations are now offered regarding the choice and the timing of these interventions.
In addition to the AHA and the ACC, this guideline was developed in collaboration with the American Association for Thoracic Surgery, American Society for Echocardiography, Society for Cardiovascular Angiography and Interventions, Society of Cardiovascular Anesthesiologists, and Society of Thoracic Surgeons.
The complete 2014 Guideline for the Management of Patients With Valvular Heart Disease is available from the American College of Cardiology and the American Heart Association.
Dr. Nishimura and Dr. Otto reported no financial conflicts of interest; their associates on the ACC/AHA Task Force's writing committee reported ties to Edwards Scientific, Medtronic, and St. Jude Medical.
The updated practice guideline for managing adults with valvular heart disease has a new, "modular" format to facilitate clinicians' access to "concise, relevant bytes of information at the point of care, when clinical knowledge is needed most," according to reports published online simultaneously March 3 in Circulation and the Journal of the American College of Cardiology.
The guideline, compiled by a committee of cardiologists, interventionalists, surgeons, and anesthesiologists under the aegis of the American Heart Association and the American College of Cardiology, was last updated in 2008.
"Some recommendations from the earlier valvular heart disease guideline have been updated as warranted by new evidence or a better understanding of earlier evidence, whereas others that were inaccurate, irrelevant, or overlapping were deleted or modified," said writing committee cochairs Dr. Rick A. Nishimura of the division of cardiovascular diseases, Mayo Clinic, Rochester, Minn.; and Dr. Catherine M. Otto, director of the University of Washington Medical Center's Heart Valve Clinic, Seattle.
The narrative text of the guideline is limited, and instead it uses decision pathway diagrams and numerous summary tables of current evidence and recommendations. These include links to relevant references. It is hoped that clinicians can more easily use the new guideline as a quick reference. This format also will enable individual sections to be updated or amended as new evidence comes to light. The PDF of the guideline is available for free.
"This novel approach to evidence-based guideline development will revolutionize the clinical impact of guideline recommendations, ensuring they are always current and allowing seamless integration with electronic medical record systems," Dr. Otto said in a press statement accompanying the reports.
The guideline now includes gradations of disease severity, to help clinicians determine the optimal timing of intervention. Whether or not intervention is indicated depends on five factors: the presence or absence of symptoms, the severity of valvular heart disease, the response of the left and/or right ventricle to the volume or pressure overload caused by the valvular disease, the effect on the pulmonary or systemic circulation, and any change in heart rhythm.
Disease severity ranges from stage A, "at risk," which denotes patients who have risk factors for developing valvular heart disease; through stage B, "progressive," which indicates patients who are asymptomatic but have mildly to moderately severe disease; through stage C, "asymptomatic severe," which includes patients with severe yet still asymptomatic valvular disease in which the left or right ventricle remains compensated or in which the left or right ventricle has decompensated; to stage D, "symptomatic severe," which indicates patients whose severe valvular disease has produced symptoms.
"In patients with stenotic lesions, there is an additional category of 'very severe' stenosis based on studies of the natural history showing that prognosis becomes poorer as the severity of stenosis increases," the guideline states.
Information is provided for assessing the various disease states associated with the aortic, mitral, and tricuspid valves, and addresses the issues of valve repair, replacement, and the use of prosthetic valves.
Compared with the previous guideline, the new one suggests surgical intervention at an earlier stage for certain patients. "Due to more knowledge regarding the natural history of untreated patients with severe valvular heart disease and better outcomes from surgery, we've lowered the threshold for operation to include more patients with asymptomatic severe disease. Now, select patients with severe asymptomatic aortic stenosis and severe asymptomatic mitral regurgitation can be considered for intervention, depending on certain other factors such as operative mortality and … the ability to achieve a durable valve repair," Dr. Nishimura said in the press statement.
The new guideline also proposes a new approach to risk assessment, to be applied to all patients for whom intervention is being considered. Previous risk scoring systems were "useful but limited"; the new approach takes into consideration "procedure-specific impediments, major organ system compromise, comorbidities, patient frailty, and the Society of Thoracic Surgeons predicted risk of mortality model."
For the first time, the guideline discusses transcatheter aortic valve replacement and other catheter-based treatments, new technologies that have improved patient care but also have complicated risk assessment. Separate recommendations are now offered regarding the choice and the timing of these interventions.
In addition to the AHA and the ACC, this guideline was developed in collaboration with the American Association for Thoracic Surgery, American Society for Echocardiography, Society for Cardiovascular Angiography and Interventions, Society of Cardiovascular Anesthesiologists, and Society of Thoracic Surgeons.
The complete 2014 Guideline for the Management of Patients With Valvular Heart Disease is available from the American College of Cardiology and the American Heart Association.
Dr. Nishimura and Dr. Otto reported no financial conflicts of interest; their associates on the ACC/AHA Task Force's writing committee reported ties to Edwards Scientific, Medtronic, and St. Jude Medical.
The updated practice guideline for managing adults with valvular heart disease has a new, "modular" format to facilitate clinicians' access to "concise, relevant bytes of information at the point of care, when clinical knowledge is needed most," according to reports published online simultaneously March 3 in Circulation and the Journal of the American College of Cardiology.
The guideline, compiled by a committee of cardiologists, interventionalists, surgeons, and anesthesiologists under the aegis of the American Heart Association and the American College of Cardiology, was last updated in 2008.
"Some recommendations from the earlier valvular heart disease guideline have been updated as warranted by new evidence or a better understanding of earlier evidence, whereas others that were inaccurate, irrelevant, or overlapping were deleted or modified," said writing committee cochairs Dr. Rick A. Nishimura of the division of cardiovascular diseases, Mayo Clinic, Rochester, Minn.; and Dr. Catherine M. Otto, director of the University of Washington Medical Center's Heart Valve Clinic, Seattle.
The narrative text of the guideline is limited, and instead it uses decision pathway diagrams and numerous summary tables of current evidence and recommendations. These include links to relevant references. It is hoped that clinicians can more easily use the new guideline as a quick reference. This format also will enable individual sections to be updated or amended as new evidence comes to light. The PDF of the guideline is available for free.
"This novel approach to evidence-based guideline development will revolutionize the clinical impact of guideline recommendations, ensuring they are always current and allowing seamless integration with electronic medical record systems," Dr. Otto said in a press statement accompanying the reports.
The guideline now includes gradations of disease severity, to help clinicians determine the optimal timing of intervention. Whether or not intervention is indicated depends on five factors: the presence or absence of symptoms, the severity of valvular heart disease, the response of the left and/or right ventricle to the volume or pressure overload caused by the valvular disease, the effect on the pulmonary or systemic circulation, and any change in heart rhythm.
Disease severity ranges from stage A, "at risk," which denotes patients who have risk factors for developing valvular heart disease; through stage B, "progressive," which indicates patients who are asymptomatic but have mildly to moderately severe disease; through stage C, "asymptomatic severe," which includes patients with severe yet still asymptomatic valvular disease in which the left or right ventricle remains compensated or in which the left or right ventricle has decompensated; to stage D, "symptomatic severe," which indicates patients whose severe valvular disease has produced symptoms.
"In patients with stenotic lesions, there is an additional category of 'very severe' stenosis based on studies of the natural history showing that prognosis becomes poorer as the severity of stenosis increases," the guideline states.
Information is provided for assessing the various disease states associated with the aortic, mitral, and tricuspid valves, and addresses the issues of valve repair, replacement, and the use of prosthetic valves.
Compared with the previous guideline, the new one suggests surgical intervention at an earlier stage for certain patients. "Due to more knowledge regarding the natural history of untreated patients with severe valvular heart disease and better outcomes from surgery, we've lowered the threshold for operation to include more patients with asymptomatic severe disease. Now, select patients with severe asymptomatic aortic stenosis and severe asymptomatic mitral regurgitation can be considered for intervention, depending on certain other factors such as operative mortality and … the ability to achieve a durable valve repair," Dr. Nishimura said in the press statement.
The new guideline also proposes a new approach to risk assessment, to be applied to all patients for whom intervention is being considered. Previous risk scoring systems were "useful but limited"; the new approach takes into consideration "procedure-specific impediments, major organ system compromise, comorbidities, patient frailty, and the Society of Thoracic Surgeons predicted risk of mortality model."
For the first time, the guideline discusses transcatheter aortic valve replacement and other catheter-based treatments, new technologies that have improved patient care but also have complicated risk assessment. Separate recommendations are now offered regarding the choice and the timing of these interventions.
In addition to the AHA and the ACC, this guideline was developed in collaboration with the American Association for Thoracic Surgery, American Society for Echocardiography, Society for Cardiovascular Angiography and Interventions, Society of Cardiovascular Anesthesiologists, and Society of Thoracic Surgeons.
The complete 2014 Guideline for the Management of Patients With Valvular Heart Disease is available from the American College of Cardiology and the American Heart Association.
Dr. Nishimura and Dr. Otto reported no financial conflicts of interest; their associates on the ACC/AHA Task Force's writing committee reported ties to Edwards Scientific, Medtronic, and St. Jude Medical.
Physician Compare site adds quality scores
Physician Compare, Medicare's online portal for checking on physician qualifications, has added quality scores for physicians in some group practices and Accountable Care Organizations. The site displays how a group of physicians performed on five quality indicators related to management of diabetes and heart disease. The website will display the ratings using stars, as well as the percentage score.
"This is an important first step in publicly reporting quality measures on Physician Compare," Dr. Patrick Conway, chief medical officer and deputy administrator for innovation and quality at the Centers for Medicare & Medicaid Services, said in a statement. "Offering a strong set of meaningful quality measures on the site will ultimately help consumers make decisions and it will encourage quality improvement among the clinician community, who shares the CMS's strong commitment to the best possible patient care."
The Physician Compare site, which was created under the Affordable Care Act, already displays the specialties offered by physicians, their board certification, and hospital affiliations. It also displays whether physicians participate in Medicare quality programs such as the Physician Quality Reporting System, the Electronic Prescribing Incentive Program, and the Electronic Health Records Incentive Program.
The information comes primarily from the Provider, Enrollment, Chain, and Ownership System and is checked against Medicare claims data. The site includes information on physicians enrolled in the Medicare program only.
This year, the CMS is posting quality data from 66 group practices and 141 ACOs. The data are reported at the group practice and ACO level. The quality measures include:
- Controlling hemoglobin A1c in patients with diabetes.
- Controlling blood pressure in patients with diabetes.
- Prescribing aspirin to patients with diabetes and heart disease.
- Reporting the number of diabetic patients who do not use tobacco.
- Prescribing medicine to improve the pumping action of the heart in patients who have both heart disease and certain other conditions.
Physician Compare, Medicare's online portal for checking on physician qualifications, has added quality scores for physicians in some group practices and Accountable Care Organizations. The site displays how a group of physicians performed on five quality indicators related to management of diabetes and heart disease. The website will display the ratings using stars, as well as the percentage score.
"This is an important first step in publicly reporting quality measures on Physician Compare," Dr. Patrick Conway, chief medical officer and deputy administrator for innovation and quality at the Centers for Medicare & Medicaid Services, said in a statement. "Offering a strong set of meaningful quality measures on the site will ultimately help consumers make decisions and it will encourage quality improvement among the clinician community, who shares the CMS's strong commitment to the best possible patient care."
The Physician Compare site, which was created under the Affordable Care Act, already displays the specialties offered by physicians, their board certification, and hospital affiliations. It also displays whether physicians participate in Medicare quality programs such as the Physician Quality Reporting System, the Electronic Prescribing Incentive Program, and the Electronic Health Records Incentive Program.
The information comes primarily from the Provider, Enrollment, Chain, and Ownership System and is checked against Medicare claims data. The site includes information on physicians enrolled in the Medicare program only.
This year, the CMS is posting quality data from 66 group practices and 141 ACOs. The data are reported at the group practice and ACO level. The quality measures include:
- Controlling hemoglobin A1c in patients with diabetes.
- Controlling blood pressure in patients with diabetes.
- Prescribing aspirin to patients with diabetes and heart disease.
- Reporting the number of diabetic patients who do not use tobacco.
- Prescribing medicine to improve the pumping action of the heart in patients who have both heart disease and certain other conditions.
Physician Compare, Medicare's online portal for checking on physician qualifications, has added quality scores for physicians in some group practices and Accountable Care Organizations. The site displays how a group of physicians performed on five quality indicators related to management of diabetes and heart disease. The website will display the ratings using stars, as well as the percentage score.
"This is an important first step in publicly reporting quality measures on Physician Compare," Dr. Patrick Conway, chief medical officer and deputy administrator for innovation and quality at the Centers for Medicare & Medicaid Services, said in a statement. "Offering a strong set of meaningful quality measures on the site will ultimately help consumers make decisions and it will encourage quality improvement among the clinician community, who shares the CMS's strong commitment to the best possible patient care."
The Physician Compare site, which was created under the Affordable Care Act, already displays the specialties offered by physicians, their board certification, and hospital affiliations. It also displays whether physicians participate in Medicare quality programs such as the Physician Quality Reporting System, the Electronic Prescribing Incentive Program, and the Electronic Health Records Incentive Program.
The information comes primarily from the Provider, Enrollment, Chain, and Ownership System and is checked against Medicare claims data. The site includes information on physicians enrolled in the Medicare program only.
This year, the CMS is posting quality data from 66 group practices and 141 ACOs. The data are reported at the group practice and ACO level. The quality measures include:
- Controlling hemoglobin A1c in patients with diabetes.
- Controlling blood pressure in patients with diabetes.
- Prescribing aspirin to patients with diabetes and heart disease.
- Reporting the number of diabetic patients who do not use tobacco.
- Prescribing medicine to improve the pumping action of the heart in patients who have both heart disease and certain other conditions.
New HAI reduction targets proposed
Federal officials are proposing new targets for reducing health care–associated infections, including a 75% reduction in invasive methicillin-resistant Staphylococcus aureus infections by 2020.
The targets are available at www.health.gov/hai/pdfs/HAI-Targets.pdf and were produced by a committee of experts in the prevention of HAIs, who were charged with updating the federal government’s National Action Plan to Prevent Health Care-Associated Infections. The 5-year goals outlined in that plan, originally released in 2009, expired last year.
The current proposal would make HAI reduction in U.S. hospitals more aggressive in some areas, while dropping five Surgical Care Improvement Project measures from the target list. The process measures, which aim to prevent surgical site infections, are now widely accepted as standards of practice and have adherence rates greater than 95%, according to HHS.
Some of the new targets will be based on 2015 baseline data, which are not yet available.
The seven proposed targets for 2020 are:
- Reduce central line–associated bloodstream infections by 50% from the 2015 baseline. Infections from mucosal barrier injury will be excluded from the calculation.
- Reduce catheter-associated urinary tract infections by 25% from the 2015 baseline.
- Reduce invasive health care–associated MRSA infections by 75% from the 2007 to 2008 baseline (27.08 infections per 100,000 persons).
- Reduce facility-onset MRSA by 50% from the 2015 baseline.
- Reduce facility-onset Clostridium difficile infections by 30% from the 2015 baseline.
- Reduce the rate of C. difficile hospitalizations by 30% from the 2015 baseline.
- Reduce surgical site infection admission and readmission by 30% from the 2015 baseline.
Hospitals and health care providers are seeing mixed results in reducing HAIs so far, according to data from HHS. As of October 2012, there have been steady improvements in reducing central line–associated bloodstream infections, health-care–associated invasive MRSA infections, and surgical site infections. But progress on reducing hospitalizations with C. difficile infection has leveled off, and providers are also not on track to meet targets for catheter-associated urinary tract infections, according to HHS.
mschneider@frontlinemedcom.com
On Twitter @maryellenny
Federal officials are proposing new targets for reducing health care–associated infections, including a 75% reduction in invasive methicillin-resistant Staphylococcus aureus infections by 2020.
The targets are available at www.health.gov/hai/pdfs/HAI-Targets.pdf and were produced by a committee of experts in the prevention of HAIs, who were charged with updating the federal government’s National Action Plan to Prevent Health Care-Associated Infections. The 5-year goals outlined in that plan, originally released in 2009, expired last year.
The current proposal would make HAI reduction in U.S. hospitals more aggressive in some areas, while dropping five Surgical Care Improvement Project measures from the target list. The process measures, which aim to prevent surgical site infections, are now widely accepted as standards of practice and have adherence rates greater than 95%, according to HHS.
Some of the new targets will be based on 2015 baseline data, which are not yet available.
The seven proposed targets for 2020 are:
- Reduce central line–associated bloodstream infections by 50% from the 2015 baseline. Infections from mucosal barrier injury will be excluded from the calculation.
- Reduce catheter-associated urinary tract infections by 25% from the 2015 baseline.
- Reduce invasive health care–associated MRSA infections by 75% from the 2007 to 2008 baseline (27.08 infections per 100,000 persons).
- Reduce facility-onset MRSA by 50% from the 2015 baseline.
- Reduce facility-onset Clostridium difficile infections by 30% from the 2015 baseline.
- Reduce the rate of C. difficile hospitalizations by 30% from the 2015 baseline.
- Reduce surgical site infection admission and readmission by 30% from the 2015 baseline.
Hospitals and health care providers are seeing mixed results in reducing HAIs so far, according to data from HHS. As of October 2012, there have been steady improvements in reducing central line–associated bloodstream infections, health-care–associated invasive MRSA infections, and surgical site infections. But progress on reducing hospitalizations with C. difficile infection has leveled off, and providers are also not on track to meet targets for catheter-associated urinary tract infections, according to HHS.
mschneider@frontlinemedcom.com
On Twitter @maryellenny
Federal officials are proposing new targets for reducing health care–associated infections, including a 75% reduction in invasive methicillin-resistant Staphylococcus aureus infections by 2020.
The targets are available at www.health.gov/hai/pdfs/HAI-Targets.pdf and were produced by a committee of experts in the prevention of HAIs, who were charged with updating the federal government’s National Action Plan to Prevent Health Care-Associated Infections. The 5-year goals outlined in that plan, originally released in 2009, expired last year.
The current proposal would make HAI reduction in U.S. hospitals more aggressive in some areas, while dropping five Surgical Care Improvement Project measures from the target list. The process measures, which aim to prevent surgical site infections, are now widely accepted as standards of practice and have adherence rates greater than 95%, according to HHS.
Some of the new targets will be based on 2015 baseline data, which are not yet available.
The seven proposed targets for 2020 are:
- Reduce central line–associated bloodstream infections by 50% from the 2015 baseline. Infections from mucosal barrier injury will be excluded from the calculation.
- Reduce catheter-associated urinary tract infections by 25% from the 2015 baseline.
- Reduce invasive health care–associated MRSA infections by 75% from the 2007 to 2008 baseline (27.08 infections per 100,000 persons).
- Reduce facility-onset MRSA by 50% from the 2015 baseline.
- Reduce facility-onset Clostridium difficile infections by 30% from the 2015 baseline.
- Reduce the rate of C. difficile hospitalizations by 30% from the 2015 baseline.
- Reduce surgical site infection admission and readmission by 30% from the 2015 baseline.
Hospitals and health care providers are seeing mixed results in reducing HAIs so far, according to data from HHS. As of October 2012, there have been steady improvements in reducing central line–associated bloodstream infections, health-care–associated invasive MRSA infections, and surgical site infections. But progress on reducing hospitalizations with C. difficile infection has leveled off, and providers are also not on track to meet targets for catheter-associated urinary tract infections, according to HHS.
mschneider@frontlinemedcom.com
On Twitter @maryellenny
Senate passes 1-year SGR patch; delays ICD-10 until 2015
In a last-minute move, Congress averted a scheduled 24% cut to Medicare physician fees slated to start on April 1, while simultaneously delaying the move to the ICD-10 coding sets.
On March 31, the Senate voted 64 to 35 to approve H.R. 4302, a bill that replaces the scheduled cut called for by the Medicare Sustainable Growth Rate formula with a 0.5% fee increase through the end of 2014 and a pay freeze from Jan. 1, 2015 through March 15, 2015. The bill also bars the Health and Human Services (HHS) department from implementing ICD-10 until Oct. 1, 2015.
The House passed the same legislation on March 27. It now heads to the White House for President Obama’s signature.
At first glance, the bill looks like good news for physicians, who will get relief from both a hefty Medicare fee cut and a costly new regulatory requirement. But many physician organizations, led by the American Medical Association, have campaigned against the temporary patch, saying that it essentially stops the work to permanently repeal the SGR.
"It appears that an unprecedented, bipartisan agreement on Medicare reform is on the verge of being cast aside because elected leaders are unwilling to make tough choices to strengthen programs serving 50 million Americans," the AMA and more than 50 other medical societies wrote to congressional leaders before the House vote.
Several physician groups had endorsed a bipartisan legislative package that would have eliminated the SGR and replaced it with a combination of small fee increases and delivery system reforms. The hurdle to passing the legislation was finding a way to pay for it that would appeal to both Democrats and Republicans.
Recently, the House passed H.R. 4015, which would have funded the permanent SGR repeal package with a 5-year delay of the Affordable Care Act’s individual insurance mandate. But that bill is unlikely to be considered in the Democratic-controlled Senate. And an attempt by Sen. Ron Wyden (D.-Ore.) to move the permanent SGR repeal forward and pay for it using the savings from the end of the wars in Iraq and Afghanistan failed to gain enough votes in the Senate.
Before the March 31 vote, Sen. Wyden chided fellow lawmakers for continuing to pass temporary fixes to the SGR problem rather than replacing it and moving to a payment system that incentivizes quality improvement. "It can’t be ducked much longer," he said.
But Sen. Orrin Hatch (R.-Utah), who worked on the bipartisan bill to replace the SGR, said he objects to the funding mechanism that Sen. Wyden proposed. He said the 12-month SGR patch will give lawmakers more time to agree on how to pay for a permanent fix. "I’m not going to make the perfect the enemy of the good," he said on the Senate floor.
mschneider@frontlinemedcom.com
On Twitter @maryellenny
In a last-minute move, Congress averted a scheduled 24% cut to Medicare physician fees slated to start on April 1, while simultaneously delaying the move to the ICD-10 coding sets.
On March 31, the Senate voted 64 to 35 to approve H.R. 4302, a bill that replaces the scheduled cut called for by the Medicare Sustainable Growth Rate formula with a 0.5% fee increase through the end of 2014 and a pay freeze from Jan. 1, 2015 through March 15, 2015. The bill also bars the Health and Human Services (HHS) department from implementing ICD-10 until Oct. 1, 2015.
The House passed the same legislation on March 27. It now heads to the White House for President Obama’s signature.
At first glance, the bill looks like good news for physicians, who will get relief from both a hefty Medicare fee cut and a costly new regulatory requirement. But many physician organizations, led by the American Medical Association, have campaigned against the temporary patch, saying that it essentially stops the work to permanently repeal the SGR.
"It appears that an unprecedented, bipartisan agreement on Medicare reform is on the verge of being cast aside because elected leaders are unwilling to make tough choices to strengthen programs serving 50 million Americans," the AMA and more than 50 other medical societies wrote to congressional leaders before the House vote.
Several physician groups had endorsed a bipartisan legislative package that would have eliminated the SGR and replaced it with a combination of small fee increases and delivery system reforms. The hurdle to passing the legislation was finding a way to pay for it that would appeal to both Democrats and Republicans.
Recently, the House passed H.R. 4015, which would have funded the permanent SGR repeal package with a 5-year delay of the Affordable Care Act’s individual insurance mandate. But that bill is unlikely to be considered in the Democratic-controlled Senate. And an attempt by Sen. Ron Wyden (D.-Ore.) to move the permanent SGR repeal forward and pay for it using the savings from the end of the wars in Iraq and Afghanistan failed to gain enough votes in the Senate.
Before the March 31 vote, Sen. Wyden chided fellow lawmakers for continuing to pass temporary fixes to the SGR problem rather than replacing it and moving to a payment system that incentivizes quality improvement. "It can’t be ducked much longer," he said.
But Sen. Orrin Hatch (R.-Utah), who worked on the bipartisan bill to replace the SGR, said he objects to the funding mechanism that Sen. Wyden proposed. He said the 12-month SGR patch will give lawmakers more time to agree on how to pay for a permanent fix. "I’m not going to make the perfect the enemy of the good," he said on the Senate floor.
mschneider@frontlinemedcom.com
On Twitter @maryellenny
In a last-minute move, Congress averted a scheduled 24% cut to Medicare physician fees slated to start on April 1, while simultaneously delaying the move to the ICD-10 coding sets.
On March 31, the Senate voted 64 to 35 to approve H.R. 4302, a bill that replaces the scheduled cut called for by the Medicare Sustainable Growth Rate formula with a 0.5% fee increase through the end of 2014 and a pay freeze from Jan. 1, 2015 through March 15, 2015. The bill also bars the Health and Human Services (HHS) department from implementing ICD-10 until Oct. 1, 2015.
The House passed the same legislation on March 27. It now heads to the White House for President Obama’s signature.
At first glance, the bill looks like good news for physicians, who will get relief from both a hefty Medicare fee cut and a costly new regulatory requirement. But many physician organizations, led by the American Medical Association, have campaigned against the temporary patch, saying that it essentially stops the work to permanently repeal the SGR.
"It appears that an unprecedented, bipartisan agreement on Medicare reform is on the verge of being cast aside because elected leaders are unwilling to make tough choices to strengthen programs serving 50 million Americans," the AMA and more than 50 other medical societies wrote to congressional leaders before the House vote.
Several physician groups had endorsed a bipartisan legislative package that would have eliminated the SGR and replaced it with a combination of small fee increases and delivery system reforms. The hurdle to passing the legislation was finding a way to pay for it that would appeal to both Democrats and Republicans.
Recently, the House passed H.R. 4015, which would have funded the permanent SGR repeal package with a 5-year delay of the Affordable Care Act’s individual insurance mandate. But that bill is unlikely to be considered in the Democratic-controlled Senate. And an attempt by Sen. Ron Wyden (D.-Ore.) to move the permanent SGR repeal forward and pay for it using the savings from the end of the wars in Iraq and Afghanistan failed to gain enough votes in the Senate.
Before the March 31 vote, Sen. Wyden chided fellow lawmakers for continuing to pass temporary fixes to the SGR problem rather than replacing it and moving to a payment system that incentivizes quality improvement. "It can’t be ducked much longer," he said.
But Sen. Orrin Hatch (R.-Utah), who worked on the bipartisan bill to replace the SGR, said he objects to the funding mechanism that Sen. Wyden proposed. He said the 12-month SGR patch will give lawmakers more time to agree on how to pay for a permanent fix. "I’m not going to make the perfect the enemy of the good," he said on the Senate floor.
mschneider@frontlinemedcom.com
On Twitter @maryellenny
STS Resident Symposium 2014
The 2014 Residents’ Symposium at the STS 50th Annual Meeting in Orlando was heavily attended and greatly appreciated by current thoracic surgical trainees.
This outstanding forum boasted several experienced and informative speakers, with the overall goals of helping residents to find the right job and transition to practice.
Moderated by Dr. Sidhu Gangadharan, Dr. Sandra Starnes, and Dr. Ara A. Vaporciyan, this session addressed several key foci, specifically aiming to help residents plan a successful job search, negotiate important elements of a contract, plan for ways to bring new technologies into a practice, recognize the legal issues of billing and coding, and identify the important aspects of early career development. This phenomenal collection of talks featured a number of key speakers, who provided tips and recommendations received with great interest by the resident participants.
The symposium was kicked off by Dr. Danny Chu of the University of Pittsburgh Medical Center, who spoke on how to find a job position. He shared his personal experiences and gave attendees an overview of the actual job-search process. He provided several tips for finding the best job. He stated that since, "good positions are almost never advertised until they are filled, it helps to have a big network." He explained that, "in such a small community, there are literally 3 degrees of separation, and everyone knows everyone. In addition to being a good surgeon, being a nice and affable person will get you a good position." His top tips for finding a job are to "Work hard, be nice, and be social." Dr. Chu concluded his presentation by explaining that there’s no magic formula for finding a job, but that it’s very much like dating. He warns that money is not everything, details are all negotiable, and one should be cautious in trusting others. In the end, he believes that "nice guys will win," and tells the group despite much anxiety, it will work out in the end.
This was followed by a talk from Dr. Christine Lau of the University of Virginia, Charlottesville, on interviewing and self-marketing. Dr. Lau provided her insights on how to best represent yourself in the interview process, with a number of great points about putting your best foot forward. She emphasized the importance of being prepared, and she strongly advised that, prior to going on an interview, you do your homework. "Know what the position entails," explained Dr. Lau, "and have a well-thought-out plan regarding what you uniquely bring to it."
She advised speaking to a number of people in advance in order to elucidate the specific needs of the group. And, while Dr. Lau encourages you to have a clear explanation as to how you might meet the expectations of the job, she offered a reminder of the importance of being honest. "Yes, you want to explain how you fit into the spot; however, if it’s not a good fit, don’t try to be something that you’re not. You won’t be happy in the long run, and neither will they." Dr. Lau also offered some advice regarding the concept of specialization.
"Everybody wants to have a niche. But you don’t need to force yourself to differentiate early on." She continued, "If your interests and skills are broad at this point, that’s not a bad thing. You can always morph later."
Dr. Faiz Bhora of Columbia University, N.Y., wrapped up the first session with a discussion of contract negotiations – helping attendees prioritize what’s worth fighting for, how to fight for it, and how to avoid going so far as to negotiate oneself out of a job.
After the opportunity to partake in both small and large group sessions and a short break, the symposium resumed, with the second half focusing on the transition from trainee to attending surgeon.
Dr. Edward Chen of Emory University in Atlanta provided suggestions for adding new technologies to one’s practice and the effective team interactions required to do so. He listed several key strategies that he feels are critical to a successful transition. Communication, communication, and communication were at the top of his list. He advised that it’s a two-way street and that ample meetings may be necessary regarding one’s needs and expectations. He warned not to make any assumptions and to be flexible about adapting to the system that is in place. He also stressed the importance of a willingness to educate the staff about specific technology – in the ICU, in the OR, and on the ward. Dr. Chen also spoke about professional behavior and team building once one is out in the real world.
His advice to trainees entering practice entails the following three recommendations: "1) Make patient care your absolute top priority. 2) Treat everyone around you with utmost respect and dignity. Care about and find value in all team members, even those who are not the strongest members. Always have your team member’s back and never openly criticize anyone. And 3) embrace your new institution and new team members." These suggestions are useful for trainees entering their first job; however, they are also incredibly practical advice for anybody working on a medical team, regardless of the environment or their role on the team.
Dr. Francis C. Nichols, from the Mayo Clinic, in Rochester, Minn., gave a great update on the new ICD-10, explaining key differences between previous coding systems and the new one.
He provided attendees with a helpful understanding of the importance of appropriate coding and billing strategies, and offered tips for accurately optimizing one’s ability to bill for provided services.
Finally, this wonderful series of brief lectures was finished off with a top-notch presentation from Dr. Elizabeth A. David of the University of California, Davis, who spoke about the various means of developing your career in the early years. She provided insight and details about developing reasonable, achievable 5- and 10-year plans; finding mentors (both within and outside of one’s institution); and how not to fail as a junior attending. The most important thing that you should do as a junior attending, according to Dr. David, is to make sure that your patients do well. She recommends, "Check everything, be vigilant, make sure your outcomes are yours, and be available to your patients and your referring physicians."
When asked what she wishes she’d known before venturing into practice, Dr. David states, "I wish I had known how hard I was going to work as a junior faculty member. People had told me that the first year would be a hard year, but it never really sunk in until I was experiencing it." She continues, "my first year was definitely a challenging year, but like all things you work hard for – it was very rewarding!"
Likewise, the symposium was incredibly rewarding. States Dr. Michelle Ellis, a finishing fellow at the University of Michigan, Ann Arbor, "I enjoyed the symposium very much. It was timely and relevant. I would definitely recommend it to other residents and fellows. I especially liked the break-out sessions which allowed for detailed discussion."
Dr. Antonoff is a second-year, thoracic-track trainee at Washington University in St Louis.
The 2014 Residents’ Symposium at the STS 50th Annual Meeting in Orlando was heavily attended and greatly appreciated by current thoracic surgical trainees.
This outstanding forum boasted several experienced and informative speakers, with the overall goals of helping residents to find the right job and transition to practice.
Moderated by Dr. Sidhu Gangadharan, Dr. Sandra Starnes, and Dr. Ara A. Vaporciyan, this session addressed several key foci, specifically aiming to help residents plan a successful job search, negotiate important elements of a contract, plan for ways to bring new technologies into a practice, recognize the legal issues of billing and coding, and identify the important aspects of early career development. This phenomenal collection of talks featured a number of key speakers, who provided tips and recommendations received with great interest by the resident participants.
The symposium was kicked off by Dr. Danny Chu of the University of Pittsburgh Medical Center, who spoke on how to find a job position. He shared his personal experiences and gave attendees an overview of the actual job-search process. He provided several tips for finding the best job. He stated that since, "good positions are almost never advertised until they are filled, it helps to have a big network." He explained that, "in such a small community, there are literally 3 degrees of separation, and everyone knows everyone. In addition to being a good surgeon, being a nice and affable person will get you a good position." His top tips for finding a job are to "Work hard, be nice, and be social." Dr. Chu concluded his presentation by explaining that there’s no magic formula for finding a job, but that it’s very much like dating. He warns that money is not everything, details are all negotiable, and one should be cautious in trusting others. In the end, he believes that "nice guys will win," and tells the group despite much anxiety, it will work out in the end.
This was followed by a talk from Dr. Christine Lau of the University of Virginia, Charlottesville, on interviewing and self-marketing. Dr. Lau provided her insights on how to best represent yourself in the interview process, with a number of great points about putting your best foot forward. She emphasized the importance of being prepared, and she strongly advised that, prior to going on an interview, you do your homework. "Know what the position entails," explained Dr. Lau, "and have a well-thought-out plan regarding what you uniquely bring to it."
She advised speaking to a number of people in advance in order to elucidate the specific needs of the group. And, while Dr. Lau encourages you to have a clear explanation as to how you might meet the expectations of the job, she offered a reminder of the importance of being honest. "Yes, you want to explain how you fit into the spot; however, if it’s not a good fit, don’t try to be something that you’re not. You won’t be happy in the long run, and neither will they." Dr. Lau also offered some advice regarding the concept of specialization.
"Everybody wants to have a niche. But you don’t need to force yourself to differentiate early on." She continued, "If your interests and skills are broad at this point, that’s not a bad thing. You can always morph later."
Dr. Faiz Bhora of Columbia University, N.Y., wrapped up the first session with a discussion of contract negotiations – helping attendees prioritize what’s worth fighting for, how to fight for it, and how to avoid going so far as to negotiate oneself out of a job.
After the opportunity to partake in both small and large group sessions and a short break, the symposium resumed, with the second half focusing on the transition from trainee to attending surgeon.
Dr. Edward Chen of Emory University in Atlanta provided suggestions for adding new technologies to one’s practice and the effective team interactions required to do so. He listed several key strategies that he feels are critical to a successful transition. Communication, communication, and communication were at the top of his list. He advised that it’s a two-way street and that ample meetings may be necessary regarding one’s needs and expectations. He warned not to make any assumptions and to be flexible about adapting to the system that is in place. He also stressed the importance of a willingness to educate the staff about specific technology – in the ICU, in the OR, and on the ward. Dr. Chen also spoke about professional behavior and team building once one is out in the real world.
His advice to trainees entering practice entails the following three recommendations: "1) Make patient care your absolute top priority. 2) Treat everyone around you with utmost respect and dignity. Care about and find value in all team members, even those who are not the strongest members. Always have your team member’s back and never openly criticize anyone. And 3) embrace your new institution and new team members." These suggestions are useful for trainees entering their first job; however, they are also incredibly practical advice for anybody working on a medical team, regardless of the environment or their role on the team.
Dr. Francis C. Nichols, from the Mayo Clinic, in Rochester, Minn., gave a great update on the new ICD-10, explaining key differences between previous coding systems and the new one.
He provided attendees with a helpful understanding of the importance of appropriate coding and billing strategies, and offered tips for accurately optimizing one’s ability to bill for provided services.
Finally, this wonderful series of brief lectures was finished off with a top-notch presentation from Dr. Elizabeth A. David of the University of California, Davis, who spoke about the various means of developing your career in the early years. She provided insight and details about developing reasonable, achievable 5- and 10-year plans; finding mentors (both within and outside of one’s institution); and how not to fail as a junior attending. The most important thing that you should do as a junior attending, according to Dr. David, is to make sure that your patients do well. She recommends, "Check everything, be vigilant, make sure your outcomes are yours, and be available to your patients and your referring physicians."
When asked what she wishes she’d known before venturing into practice, Dr. David states, "I wish I had known how hard I was going to work as a junior faculty member. People had told me that the first year would be a hard year, but it never really sunk in until I was experiencing it." She continues, "my first year was definitely a challenging year, but like all things you work hard for – it was very rewarding!"
Likewise, the symposium was incredibly rewarding. States Dr. Michelle Ellis, a finishing fellow at the University of Michigan, Ann Arbor, "I enjoyed the symposium very much. It was timely and relevant. I would definitely recommend it to other residents and fellows. I especially liked the break-out sessions which allowed for detailed discussion."
Dr. Antonoff is a second-year, thoracic-track trainee at Washington University in St Louis.
The 2014 Residents’ Symposium at the STS 50th Annual Meeting in Orlando was heavily attended and greatly appreciated by current thoracic surgical trainees.
This outstanding forum boasted several experienced and informative speakers, with the overall goals of helping residents to find the right job and transition to practice.
Moderated by Dr. Sidhu Gangadharan, Dr. Sandra Starnes, and Dr. Ara A. Vaporciyan, this session addressed several key foci, specifically aiming to help residents plan a successful job search, negotiate important elements of a contract, plan for ways to bring new technologies into a practice, recognize the legal issues of billing and coding, and identify the important aspects of early career development. This phenomenal collection of talks featured a number of key speakers, who provided tips and recommendations received with great interest by the resident participants.
The symposium was kicked off by Dr. Danny Chu of the University of Pittsburgh Medical Center, who spoke on how to find a job position. He shared his personal experiences and gave attendees an overview of the actual job-search process. He provided several tips for finding the best job. He stated that since, "good positions are almost never advertised until they are filled, it helps to have a big network." He explained that, "in such a small community, there are literally 3 degrees of separation, and everyone knows everyone. In addition to being a good surgeon, being a nice and affable person will get you a good position." His top tips for finding a job are to "Work hard, be nice, and be social." Dr. Chu concluded his presentation by explaining that there’s no magic formula for finding a job, but that it’s very much like dating. He warns that money is not everything, details are all negotiable, and one should be cautious in trusting others. In the end, he believes that "nice guys will win," and tells the group despite much anxiety, it will work out in the end.
This was followed by a talk from Dr. Christine Lau of the University of Virginia, Charlottesville, on interviewing and self-marketing. Dr. Lau provided her insights on how to best represent yourself in the interview process, with a number of great points about putting your best foot forward. She emphasized the importance of being prepared, and she strongly advised that, prior to going on an interview, you do your homework. "Know what the position entails," explained Dr. Lau, "and have a well-thought-out plan regarding what you uniquely bring to it."
She advised speaking to a number of people in advance in order to elucidate the specific needs of the group. And, while Dr. Lau encourages you to have a clear explanation as to how you might meet the expectations of the job, she offered a reminder of the importance of being honest. "Yes, you want to explain how you fit into the spot; however, if it’s not a good fit, don’t try to be something that you’re not. You won’t be happy in the long run, and neither will they." Dr. Lau also offered some advice regarding the concept of specialization.
"Everybody wants to have a niche. But you don’t need to force yourself to differentiate early on." She continued, "If your interests and skills are broad at this point, that’s not a bad thing. You can always morph later."
Dr. Faiz Bhora of Columbia University, N.Y., wrapped up the first session with a discussion of contract negotiations – helping attendees prioritize what’s worth fighting for, how to fight for it, and how to avoid going so far as to negotiate oneself out of a job.
After the opportunity to partake in both small and large group sessions and a short break, the symposium resumed, with the second half focusing on the transition from trainee to attending surgeon.
Dr. Edward Chen of Emory University in Atlanta provided suggestions for adding new technologies to one’s practice and the effective team interactions required to do so. He listed several key strategies that he feels are critical to a successful transition. Communication, communication, and communication were at the top of his list. He advised that it’s a two-way street and that ample meetings may be necessary regarding one’s needs and expectations. He warned not to make any assumptions and to be flexible about adapting to the system that is in place. He also stressed the importance of a willingness to educate the staff about specific technology – in the ICU, in the OR, and on the ward. Dr. Chen also spoke about professional behavior and team building once one is out in the real world.
His advice to trainees entering practice entails the following three recommendations: "1) Make patient care your absolute top priority. 2) Treat everyone around you with utmost respect and dignity. Care about and find value in all team members, even those who are not the strongest members. Always have your team member’s back and never openly criticize anyone. And 3) embrace your new institution and new team members." These suggestions are useful for trainees entering their first job; however, they are also incredibly practical advice for anybody working on a medical team, regardless of the environment or their role on the team.
Dr. Francis C. Nichols, from the Mayo Clinic, in Rochester, Minn., gave a great update on the new ICD-10, explaining key differences between previous coding systems and the new one.
He provided attendees with a helpful understanding of the importance of appropriate coding and billing strategies, and offered tips for accurately optimizing one’s ability to bill for provided services.
Finally, this wonderful series of brief lectures was finished off with a top-notch presentation from Dr. Elizabeth A. David of the University of California, Davis, who spoke about the various means of developing your career in the early years. She provided insight and details about developing reasonable, achievable 5- and 10-year plans; finding mentors (both within and outside of one’s institution); and how not to fail as a junior attending. The most important thing that you should do as a junior attending, according to Dr. David, is to make sure that your patients do well. She recommends, "Check everything, be vigilant, make sure your outcomes are yours, and be available to your patients and your referring physicians."
When asked what she wishes she’d known before venturing into practice, Dr. David states, "I wish I had known how hard I was going to work as a junior faculty member. People had told me that the first year would be a hard year, but it never really sunk in until I was experiencing it." She continues, "my first year was definitely a challenging year, but like all things you work hard for – it was very rewarding!"
Likewise, the symposium was incredibly rewarding. States Dr. Michelle Ellis, a finishing fellow at the University of Michigan, Ann Arbor, "I enjoyed the symposium very much. It was timely and relevant. I would definitely recommend it to other residents and fellows. I especially liked the break-out sessions which allowed for detailed discussion."
Dr. Antonoff is a second-year, thoracic-track trainee at Washington University in St Louis.
Testing now is critical to ICD-10 readiness
Still not prepared for the switch over to ICD-10? Experts say there’s still time to catch up before the Oct. 1 compliance date.
In an ideal world, physicians, their coders, and office staff would have already fully assessed the cost of transitioning to ICD-10, would have undergone training about how to improve clinical documentation and appropriate use of the new diagnosis codes, and would have internally tested their upgraded software. That would leave more than half a year to complete external testing with health plans to ensure that they get paid in October.
The reality? Few are on track to meet those milestones on time.
"Everybody is behind, not just practices," said Robert Tennant, senior policy adviser at the Medical Group Management Association (MGMA). For a practice to be ready for ICD-10, the "key trading partners have to be ready and our surveys are indicating that they are not," he said.
Those partners include practice management software vendors, electronic health record (EHR) vendors, clearinghouses, and private and public payers.
So does that mean another ICD-10 delay is likely? The Centers for Medicare and Medicaid Services says no.
Part of the reason may be that health plans, which are investing money in upgrading their own systems, are pushing the agency to move forward on time. They are signaling to the government that they don’t want another delay, Mr. Tennant said.
The MGMA is advising its members to prepare for an Oct. 1 launch of ICD-10. But Mr. Tennant said he’s not entirely convinced that the CMS will stick to the date if much of the health care industry is unprepared to make the switch.
"Claims have to be paid," he said. "The system cannot grind to a halt because practices that aren’t paid can’t see patients."
Start with training
Assuming that most physician offices have assessed which parts of their practice management and EHR systems need to be upgraded, and have determined the cost for upgrades and training, it’s time to get familiar with the new diagnosis coding system.
Coders and billers will need the most training on the new coding methodology, but doctors still need to get familiar with the level of documentation that’s need for their most frequently used codes. In general, the new system calls for great specificity, though it’s not an absolute rule.
For instance, the classification for asthma has changed from ICD-9 to ICD-10. Physicians will need to provide more specific documentation about the severity level (moderately persistent, severely persistent, etc.) for their coders to select the appropriate ICD-10 code, according to Kathryn DeVault, a coding expert at the American Health Information Management Association (AHIMA).
"The good news is that as different as ICD-10 and ICD-9 are, they are similar," Ms. DeVault said. "If there’s a familiarity or comfort level working with ICD-9, it’s a natural transition to ICD-10."
While there are some significant changes, especially related to orthopedic codes, for many subspecialties the differences will be minimal, she said.
Ms. DeVault recommended having the practice manager or lead coder identify the top 20 diagnosis codes for every physician in the practice and build some education around those frequently used codes.
She cautioned doctors not to skimp on the time and money needed to thoroughly train themselves and their staffs. "The key here is to do it right and do it right the first time," she said.
Check the books
Physicians and their staff also need to evaluate their current cash flow and revenue cycle, including the age of account balances, billing lag time, and other issues that may result in delayed or denied claims, said Asia Blunt, practice management strategist at the American Academy of Family Physicians. Correct those problems now, she said, then reevaluate between April and August.
Experts at the AAFP are recommending that physicians put aside a cash reserve, if possible, to cover expenses during the first 3 months of the transition in case large numbers of claims are denied. (See below for more tips on planning for the worst.)
Testing with vendors, payers
Internal and external testing is also key. Before Oct. 1, practices should have completed end-to-end testing of their upgraded systems, ensuring that everything works smoothly from the time they code a claim to when they receive payment from the health insurer.
Practices can begin internal testing as soon as they have upgraded their software. But external testing will depend on when clearinghouses and health plans are ready.
The CMS will provide the first testing opportunity March 3-7. The agency will hold a national ICD-10 testing week allowing practices and clearinghouses to submit claims using the new coding system. Practices will receive an acknowledgement that the claims were either accepted or rejected by the system. Practices must register in advance through their local Medicare Administrative Contractor (MAC) website to test. Find your local MAC here.
But this type of front-end testing is only a first step, Mr. Tennant said. Front-end testing determines whether the claim contains an ICD-10 code and if it is in the right place and the right format. But practices will need to conduct further testing with payers to determine if the code they used is appropriate and whether they will get paid.
For instance, when submitting a claim for a sprained ankle under ICD-10, the coder might specify right or left ankle, or leave it as unspecified. Depending on the health insurer’s policy around the code, the insurer could pay the claim, reject it, or hold or "pend" it while seeking additional information. The complicating factor, Mr. Tennant said, is that each health insurer has different coding policies and those policies have yet to be released for ICD-10.
"It’s very frustrating for everybody," Mr. Tennant said.
To minimize the impact, Mr. Tennant recommended identifying the payers responsible for the majority of your claims. Keep in contact with them about the release of their payment policies and testing schedules, he said.
"Be aggressive in your outreach to those plans," he said.
Contingency plans
Just in case a worst-case scenario develops, Mr. Tennant offered the advice on ICD-10 contingency plans:
• Research back-up options for practice management systems and clearinghouses. If the vendors aren’t providing a clear answer on when they will be ready to offer upgrades and testing, start researching alternatives. Ask colleagues if they have vendors that are prepared for the transition.
• Don’t rely on one coder. Train more than one staff member on how to use the new coding system. That way, if the chief coder leaves 3 weeks before the compliance date, someone else can step in.
• Limit vacations around the Oct. 1 compliance date. This is not a time to operate short staffed.
• Don’t wait around for health plans to start ICD-10 testing. Start with context testing. Take a subset of high-dollar, high-volume ICD-9 claims that have already been paid by the health plan and practice coding them in ICD-10. Similarly, begin to code claims in parallel in both ICD-9 and ICD-10 and move them through your internal workflow. In both of these testing approaches, check if the documentation provided is sufficient to identify the best ICD-10 code. If not, it’s time for more training.
• Ensure you have enough cash to operate in case claims are rejected or delayed. Setting aside cash reserves is a good move. Consider postponing major capital investments for a few months before and after Oct. 1. Obtaining a line of credit to cover a few months of operating expenses is another option.
• Submit as many of claims as possible with ICD-9 codes before Oct. 1.
Free ICD-10 resources
• ICD-10 guide with checklists and timelines (CMS).
• Sample letter to gauge vendors’ ICD-10 readiness (AHIMA).
• Cost calculator and ICD-10 timeline (AAFP).
• Twelve step transition plan, white papers, and practice tool (AMA)
mschneider@frontlinemedcom.com
On Twitter @maryellenny
Still not prepared for the switch over to ICD-10? Experts say there’s still time to catch up before the Oct. 1 compliance date.
In an ideal world, physicians, their coders, and office staff would have already fully assessed the cost of transitioning to ICD-10, would have undergone training about how to improve clinical documentation and appropriate use of the new diagnosis codes, and would have internally tested their upgraded software. That would leave more than half a year to complete external testing with health plans to ensure that they get paid in October.
The reality? Few are on track to meet those milestones on time.
"Everybody is behind, not just practices," said Robert Tennant, senior policy adviser at the Medical Group Management Association (MGMA). For a practice to be ready for ICD-10, the "key trading partners have to be ready and our surveys are indicating that they are not," he said.
Those partners include practice management software vendors, electronic health record (EHR) vendors, clearinghouses, and private and public payers.
So does that mean another ICD-10 delay is likely? The Centers for Medicare and Medicaid Services says no.
Part of the reason may be that health plans, which are investing money in upgrading their own systems, are pushing the agency to move forward on time. They are signaling to the government that they don’t want another delay, Mr. Tennant said.
The MGMA is advising its members to prepare for an Oct. 1 launch of ICD-10. But Mr. Tennant said he’s not entirely convinced that the CMS will stick to the date if much of the health care industry is unprepared to make the switch.
"Claims have to be paid," he said. "The system cannot grind to a halt because practices that aren’t paid can’t see patients."
Start with training
Assuming that most physician offices have assessed which parts of their practice management and EHR systems need to be upgraded, and have determined the cost for upgrades and training, it’s time to get familiar with the new diagnosis coding system.
Coders and billers will need the most training on the new coding methodology, but doctors still need to get familiar with the level of documentation that’s need for their most frequently used codes. In general, the new system calls for great specificity, though it’s not an absolute rule.
For instance, the classification for asthma has changed from ICD-9 to ICD-10. Physicians will need to provide more specific documentation about the severity level (moderately persistent, severely persistent, etc.) for their coders to select the appropriate ICD-10 code, according to Kathryn DeVault, a coding expert at the American Health Information Management Association (AHIMA).
"The good news is that as different as ICD-10 and ICD-9 are, they are similar," Ms. DeVault said. "If there’s a familiarity or comfort level working with ICD-9, it’s a natural transition to ICD-10."
While there are some significant changes, especially related to orthopedic codes, for many subspecialties the differences will be minimal, she said.
Ms. DeVault recommended having the practice manager or lead coder identify the top 20 diagnosis codes for every physician in the practice and build some education around those frequently used codes.
She cautioned doctors not to skimp on the time and money needed to thoroughly train themselves and their staffs. "The key here is to do it right and do it right the first time," she said.
Check the books
Physicians and their staff also need to evaluate their current cash flow and revenue cycle, including the age of account balances, billing lag time, and other issues that may result in delayed or denied claims, said Asia Blunt, practice management strategist at the American Academy of Family Physicians. Correct those problems now, she said, then reevaluate between April and August.
Experts at the AAFP are recommending that physicians put aside a cash reserve, if possible, to cover expenses during the first 3 months of the transition in case large numbers of claims are denied. (See below for more tips on planning for the worst.)
Testing with vendors, payers
Internal and external testing is also key. Before Oct. 1, practices should have completed end-to-end testing of their upgraded systems, ensuring that everything works smoothly from the time they code a claim to when they receive payment from the health insurer.
Practices can begin internal testing as soon as they have upgraded their software. But external testing will depend on when clearinghouses and health plans are ready.
The CMS will provide the first testing opportunity March 3-7. The agency will hold a national ICD-10 testing week allowing practices and clearinghouses to submit claims using the new coding system. Practices will receive an acknowledgement that the claims were either accepted or rejected by the system. Practices must register in advance through their local Medicare Administrative Contractor (MAC) website to test. Find your local MAC here.
But this type of front-end testing is only a first step, Mr. Tennant said. Front-end testing determines whether the claim contains an ICD-10 code and if it is in the right place and the right format. But practices will need to conduct further testing with payers to determine if the code they used is appropriate and whether they will get paid.
For instance, when submitting a claim for a sprained ankle under ICD-10, the coder might specify right or left ankle, or leave it as unspecified. Depending on the health insurer’s policy around the code, the insurer could pay the claim, reject it, or hold or "pend" it while seeking additional information. The complicating factor, Mr. Tennant said, is that each health insurer has different coding policies and those policies have yet to be released for ICD-10.
"It’s very frustrating for everybody," Mr. Tennant said.
To minimize the impact, Mr. Tennant recommended identifying the payers responsible for the majority of your claims. Keep in contact with them about the release of their payment policies and testing schedules, he said.
"Be aggressive in your outreach to those plans," he said.
Contingency plans
Just in case a worst-case scenario develops, Mr. Tennant offered the advice on ICD-10 contingency plans:
• Research back-up options for practice management systems and clearinghouses. If the vendors aren’t providing a clear answer on when they will be ready to offer upgrades and testing, start researching alternatives. Ask colleagues if they have vendors that are prepared for the transition.
• Don’t rely on one coder. Train more than one staff member on how to use the new coding system. That way, if the chief coder leaves 3 weeks before the compliance date, someone else can step in.
• Limit vacations around the Oct. 1 compliance date. This is not a time to operate short staffed.
• Don’t wait around for health plans to start ICD-10 testing. Start with context testing. Take a subset of high-dollar, high-volume ICD-9 claims that have already been paid by the health plan and practice coding them in ICD-10. Similarly, begin to code claims in parallel in both ICD-9 and ICD-10 and move them through your internal workflow. In both of these testing approaches, check if the documentation provided is sufficient to identify the best ICD-10 code. If not, it’s time for more training.
• Ensure you have enough cash to operate in case claims are rejected or delayed. Setting aside cash reserves is a good move. Consider postponing major capital investments for a few months before and after Oct. 1. Obtaining a line of credit to cover a few months of operating expenses is another option.
• Submit as many of claims as possible with ICD-9 codes before Oct. 1.
Free ICD-10 resources
• ICD-10 guide with checklists and timelines (CMS).
• Sample letter to gauge vendors’ ICD-10 readiness (AHIMA).
• Cost calculator and ICD-10 timeline (AAFP).
• Twelve step transition plan, white papers, and practice tool (AMA)
mschneider@frontlinemedcom.com
On Twitter @maryellenny
Still not prepared for the switch over to ICD-10? Experts say there’s still time to catch up before the Oct. 1 compliance date.
In an ideal world, physicians, their coders, and office staff would have already fully assessed the cost of transitioning to ICD-10, would have undergone training about how to improve clinical documentation and appropriate use of the new diagnosis codes, and would have internally tested their upgraded software. That would leave more than half a year to complete external testing with health plans to ensure that they get paid in October.
The reality? Few are on track to meet those milestones on time.
"Everybody is behind, not just practices," said Robert Tennant, senior policy adviser at the Medical Group Management Association (MGMA). For a practice to be ready for ICD-10, the "key trading partners have to be ready and our surveys are indicating that they are not," he said.
Those partners include practice management software vendors, electronic health record (EHR) vendors, clearinghouses, and private and public payers.
So does that mean another ICD-10 delay is likely? The Centers for Medicare and Medicaid Services says no.
Part of the reason may be that health plans, which are investing money in upgrading their own systems, are pushing the agency to move forward on time. They are signaling to the government that they don’t want another delay, Mr. Tennant said.
The MGMA is advising its members to prepare for an Oct. 1 launch of ICD-10. But Mr. Tennant said he’s not entirely convinced that the CMS will stick to the date if much of the health care industry is unprepared to make the switch.
"Claims have to be paid," he said. "The system cannot grind to a halt because practices that aren’t paid can’t see patients."
Start with training
Assuming that most physician offices have assessed which parts of their practice management and EHR systems need to be upgraded, and have determined the cost for upgrades and training, it’s time to get familiar with the new diagnosis coding system.
Coders and billers will need the most training on the new coding methodology, but doctors still need to get familiar with the level of documentation that’s need for their most frequently used codes. In general, the new system calls for great specificity, though it’s not an absolute rule.
For instance, the classification for asthma has changed from ICD-9 to ICD-10. Physicians will need to provide more specific documentation about the severity level (moderately persistent, severely persistent, etc.) for their coders to select the appropriate ICD-10 code, according to Kathryn DeVault, a coding expert at the American Health Information Management Association (AHIMA).
"The good news is that as different as ICD-10 and ICD-9 are, they are similar," Ms. DeVault said. "If there’s a familiarity or comfort level working with ICD-9, it’s a natural transition to ICD-10."
While there are some significant changes, especially related to orthopedic codes, for many subspecialties the differences will be minimal, she said.
Ms. DeVault recommended having the practice manager or lead coder identify the top 20 diagnosis codes for every physician in the practice and build some education around those frequently used codes.
She cautioned doctors not to skimp on the time and money needed to thoroughly train themselves and their staffs. "The key here is to do it right and do it right the first time," she said.
Check the books
Physicians and their staff also need to evaluate their current cash flow and revenue cycle, including the age of account balances, billing lag time, and other issues that may result in delayed or denied claims, said Asia Blunt, practice management strategist at the American Academy of Family Physicians. Correct those problems now, she said, then reevaluate between April and August.
Experts at the AAFP are recommending that physicians put aside a cash reserve, if possible, to cover expenses during the first 3 months of the transition in case large numbers of claims are denied. (See below for more tips on planning for the worst.)
Testing with vendors, payers
Internal and external testing is also key. Before Oct. 1, practices should have completed end-to-end testing of their upgraded systems, ensuring that everything works smoothly from the time they code a claim to when they receive payment from the health insurer.
Practices can begin internal testing as soon as they have upgraded their software. But external testing will depend on when clearinghouses and health plans are ready.
The CMS will provide the first testing opportunity March 3-7. The agency will hold a national ICD-10 testing week allowing practices and clearinghouses to submit claims using the new coding system. Practices will receive an acknowledgement that the claims were either accepted or rejected by the system. Practices must register in advance through their local Medicare Administrative Contractor (MAC) website to test. Find your local MAC here.
But this type of front-end testing is only a first step, Mr. Tennant said. Front-end testing determines whether the claim contains an ICD-10 code and if it is in the right place and the right format. But practices will need to conduct further testing with payers to determine if the code they used is appropriate and whether they will get paid.
For instance, when submitting a claim for a sprained ankle under ICD-10, the coder might specify right or left ankle, or leave it as unspecified. Depending on the health insurer’s policy around the code, the insurer could pay the claim, reject it, or hold or "pend" it while seeking additional information. The complicating factor, Mr. Tennant said, is that each health insurer has different coding policies and those policies have yet to be released for ICD-10.
"It’s very frustrating for everybody," Mr. Tennant said.
To minimize the impact, Mr. Tennant recommended identifying the payers responsible for the majority of your claims. Keep in contact with them about the release of their payment policies and testing schedules, he said.
"Be aggressive in your outreach to those plans," he said.
Contingency plans
Just in case a worst-case scenario develops, Mr. Tennant offered the advice on ICD-10 contingency plans:
• Research back-up options for practice management systems and clearinghouses. If the vendors aren’t providing a clear answer on when they will be ready to offer upgrades and testing, start researching alternatives. Ask colleagues if they have vendors that are prepared for the transition.
• Don’t rely on one coder. Train more than one staff member on how to use the new coding system. That way, if the chief coder leaves 3 weeks before the compliance date, someone else can step in.
• Limit vacations around the Oct. 1 compliance date. This is not a time to operate short staffed.
• Don’t wait around for health plans to start ICD-10 testing. Start with context testing. Take a subset of high-dollar, high-volume ICD-9 claims that have already been paid by the health plan and practice coding them in ICD-10. Similarly, begin to code claims in parallel in both ICD-9 and ICD-10 and move them through your internal workflow. In both of these testing approaches, check if the documentation provided is sufficient to identify the best ICD-10 code. If not, it’s time for more training.
• Ensure you have enough cash to operate in case claims are rejected or delayed. Setting aside cash reserves is a good move. Consider postponing major capital investments for a few months before and after Oct. 1. Obtaining a line of credit to cover a few months of operating expenses is another option.
• Submit as many of claims as possible with ICD-9 codes before Oct. 1.
Free ICD-10 resources
• ICD-10 guide with checklists and timelines (CMS).
• Sample letter to gauge vendors’ ICD-10 readiness (AHIMA).
• Cost calculator and ICD-10 timeline (AAFP).
• Twelve step transition plan, white papers, and practice tool (AMA)
mschneider@frontlinemedcom.com
On Twitter @maryellenny
Preoperative organ dysfunction worsens SAVR outcomes
SNOWMASS, COLO. – The presence of preoperative dysfunction in more than any one of four key organ systems profoundly reduces survival in patients undergoing surgical aortic valve replacement, a study showed.
"If you have two or more dysfunctional organ systems, you really need to think about what you’re doing for this patient. At 5 years, only about 40% of these patients are alive. It makes a lot of sense to me to say that if you have a patient with severe COPD [chronic obstructive pulmonary disease] and renal dysfunction, that patient should probably never get a surgical valve," Dr. Vinod H. Thourani said at the Annual Cardiovascular Conference at Snowmass.
In a retrospective analysis of a registry with prospectively entered data, 29% of 1,759 patients who underwent surgical aortic valve replacement (SAVR) with or without coronary artery bypass grafting at Emory University during 2002-2010 had preoperative dysfunction of one or more of four organ systems under scrutiny. Eighty-five patients had severe COPD, as defined by a forced expiratory volume in 1 second (FEV1) that was less than 50% of predicted, 140 had chronic renal failure, 149 had a prior stroke, and 241 had heart failure with a left ventricular ejection less than 35%.
Patients with chronic renal failure had far and away the worst 30-day and long-term outcomes. Half were dead within 3 years. The 7-year survival rate was just 11.7%.
The second-worst outcomes were seen in patients with severe COPD preoperatively. Their 7-year survival rate was 30.8%.
"Anyone with an FEV1 below about 40% becomes a higher-risk surgical candidate; think instead of TAVR [transcatheter aortic valve replacement],"advised Dr. Thourani of the division of cardiothoracic surgery at Emory University, Atlanta.
In contrast, outcomes in patients with either heart failure or prior stroke "were not that bad," he said, pointing to 7-year survival rates of 55.9% and 48.6%, respectively.
Ninety-five patients (5.4%) in this recently published study (Ann. Thorac. Surg. 2013;95:838-45) had more than one dysfunctional organ system prior to SAVR. Median survival in patients without dysfunction in any of the four organ systems was 8.2 years and counting. With one dysfunctional organ, it was still good at 7.2 years. However, with two dysfunctional organ systems, the median survival dropped precipitously to 4.1 years. With three dysfunctional organ systems, it was 5.9 years.
Dr. Thourin serves as a consultant to Edwards Lifesciences, Sorin, and St. Jude Medical.
SNOWMASS, COLO. – The presence of preoperative dysfunction in more than any one of four key organ systems profoundly reduces survival in patients undergoing surgical aortic valve replacement, a study showed.
"If you have two or more dysfunctional organ systems, you really need to think about what you’re doing for this patient. At 5 years, only about 40% of these patients are alive. It makes a lot of sense to me to say that if you have a patient with severe COPD [chronic obstructive pulmonary disease] and renal dysfunction, that patient should probably never get a surgical valve," Dr. Vinod H. Thourani said at the Annual Cardiovascular Conference at Snowmass.
In a retrospective analysis of a registry with prospectively entered data, 29% of 1,759 patients who underwent surgical aortic valve replacement (SAVR) with or without coronary artery bypass grafting at Emory University during 2002-2010 had preoperative dysfunction of one or more of four organ systems under scrutiny. Eighty-five patients had severe COPD, as defined by a forced expiratory volume in 1 second (FEV1) that was less than 50% of predicted, 140 had chronic renal failure, 149 had a prior stroke, and 241 had heart failure with a left ventricular ejection less than 35%.
Patients with chronic renal failure had far and away the worst 30-day and long-term outcomes. Half were dead within 3 years. The 7-year survival rate was just 11.7%.
The second-worst outcomes were seen in patients with severe COPD preoperatively. Their 7-year survival rate was 30.8%.
"Anyone with an FEV1 below about 40% becomes a higher-risk surgical candidate; think instead of TAVR [transcatheter aortic valve replacement],"advised Dr. Thourani of the division of cardiothoracic surgery at Emory University, Atlanta.
In contrast, outcomes in patients with either heart failure or prior stroke "were not that bad," he said, pointing to 7-year survival rates of 55.9% and 48.6%, respectively.
Ninety-five patients (5.4%) in this recently published study (Ann. Thorac. Surg. 2013;95:838-45) had more than one dysfunctional organ system prior to SAVR. Median survival in patients without dysfunction in any of the four organ systems was 8.2 years and counting. With one dysfunctional organ, it was still good at 7.2 years. However, with two dysfunctional organ systems, the median survival dropped precipitously to 4.1 years. With three dysfunctional organ systems, it was 5.9 years.
Dr. Thourin serves as a consultant to Edwards Lifesciences, Sorin, and St. Jude Medical.
SNOWMASS, COLO. – The presence of preoperative dysfunction in more than any one of four key organ systems profoundly reduces survival in patients undergoing surgical aortic valve replacement, a study showed.
"If you have two or more dysfunctional organ systems, you really need to think about what you’re doing for this patient. At 5 years, only about 40% of these patients are alive. It makes a lot of sense to me to say that if you have a patient with severe COPD [chronic obstructive pulmonary disease] and renal dysfunction, that patient should probably never get a surgical valve," Dr. Vinod H. Thourani said at the Annual Cardiovascular Conference at Snowmass.
In a retrospective analysis of a registry with prospectively entered data, 29% of 1,759 patients who underwent surgical aortic valve replacement (SAVR) with or without coronary artery bypass grafting at Emory University during 2002-2010 had preoperative dysfunction of one or more of four organ systems under scrutiny. Eighty-five patients had severe COPD, as defined by a forced expiratory volume in 1 second (FEV1) that was less than 50% of predicted, 140 had chronic renal failure, 149 had a prior stroke, and 241 had heart failure with a left ventricular ejection less than 35%.
Patients with chronic renal failure had far and away the worst 30-day and long-term outcomes. Half were dead within 3 years. The 7-year survival rate was just 11.7%.
The second-worst outcomes were seen in patients with severe COPD preoperatively. Their 7-year survival rate was 30.8%.
"Anyone with an FEV1 below about 40% becomes a higher-risk surgical candidate; think instead of TAVR [transcatheter aortic valve replacement],"advised Dr. Thourani of the division of cardiothoracic surgery at Emory University, Atlanta.
In contrast, outcomes in patients with either heart failure or prior stroke "were not that bad," he said, pointing to 7-year survival rates of 55.9% and 48.6%, respectively.
Ninety-five patients (5.4%) in this recently published study (Ann. Thorac. Surg. 2013;95:838-45) had more than one dysfunctional organ system prior to SAVR. Median survival in patients without dysfunction in any of the four organ systems was 8.2 years and counting. With one dysfunctional organ, it was still good at 7.2 years. However, with two dysfunctional organ systems, the median survival dropped precipitously to 4.1 years. With three dysfunctional organ systems, it was 5.9 years.
Dr. Thourin serves as a consultant to Edwards Lifesciences, Sorin, and St. Jude Medical.
EXPERT ANALYSIS FROM THE CARDIOVASCULAR CONFERENCE AT SNOWMASS
CoreValve holds size advantage for U.S. TAVR
When the Food and Drug Administration in January granted marketing approval to a second transcatheter aortic valve replacement system for inoperable patients with aortic stenosis, the CoreValve marketed by Medtronic, the new valve conceded a greater than 2-year head start to the first system on the U.S. market, Sapien marketed by Edwards.
But cardiologists see that 2-year edge in familiarity eclipsed for at least some patients by two major advantages that CoreValve currently holds over Sapien: delivery via a significantly thinner sheath, and the option of larger-diameter valves that allow replacement in patients with a wider aortic annulus.
The CoreValve delivery sheath is 18 French, compared with a 22F or 24F size for the Sapien transcatheter aortic valve replacement (TAVR) with U.S. approval, and the Sapien valves come in diameters of 23 and 26 mm, compared with options of 23, 26, 29, and 31 mm for the CoreValve.
"CoreValve is the device of choice for patients with smaller vessel sizes. Sapien has been a wonderful device to use, and we have so much experience with it, but the smaller CoreValve size will allow many more patients to be done with a transfemoral approach," said Dr. Peter C. Block, a professor of medicine at Emory University in Atlanta and an interventional cardiologist who performs TAVR.
"More patients will qualify for TAVR and will be treated transfemorally with the larger valve diameters and smaller sheath size," agreed Dr. Mauricio G. Cohen, director of cardiac catheterization at the University of Miami and TAVR interventionalist. Another potential positive of having CoreValve on the U.S. market is that competition between the two options will likely drive down the cost of a TAVR system, which until now has run more than $30,000, Dr. Cohen said in an interview.
CoreValve received FDA approval less than 3 months after researchers first reported data from the company’s U.S. Pivotal Trial Extreme Risk Iliofemoral Study last October at the TCT (Transcatheter Cardiovascular Therapeutics) annual meeting. In that study, 471 inoperable aortic stenosis patients had a 26% 1-year rate of death or major stroke, substantially surpassing the 43% rate that the study set up as the target for superiority, reported Dr. Jeffrey J. Popma, lead investigator on the study.
Dr. Popma warned against comparing CoreValve’s efficacy and safety performance in the trial and the Sapien system’s performance in its pivotal trial in inoperable patients, the PARTNER cohort B trial (N. Engl. J. Med. 2010;363:1597-607). "It’s very difficult to make cross-trial comparison," he said in an interview, a limitation also noted by Dr. Cohen and Dr. Block. But Dr. Popma highlighted the 2.4% 30-day stroke rate in the pivotal trial, and a 1.8% 30-day stroke rate seen with CoreValve in inoperable patients in a continued access program at the trial’s study sites. He also highlighted the 11% rate of moderate paravalvular aortic regurgitation after 30 days that dropped to a 4% rate after 1 year.
Perhaps the biggest downside of CoreValve’s performance in the pivotal trial was that 22% of patients required a permanent pacemaker implant within the first 30 days, increasing to 27% of patients with 1-year follow-up. Increased risk for a pacemaker is an inherent downside of CoreValve because of its longer size compared with the Sapien valve and how the CoreValve sits in the aortic annulus. The CoreValve is designed for supravalvular placement and anchoring in the left ventricular outflow tract near the left bundle branch that can result in mechanical irritation and arrhythmia with the need for pacing, explained Dr. Popma, professor of medicine at Harvard University and an interventional cardiologist at Beth Israel Deaconess Medical Center, Boston.
"I think our pacemaker rate was very acceptable. I don’t think it will ever be as low as with Sapien, but it’s a worthwhile trade-off because the CoreValve functions well and results in a low rate of paravalvular regurgitation," he said.
Dr. Popma also stressed that 1-year mortality was no greater among the patients who required a pacemaker implant in the pivotal trial. A subgroup analysis of results from the trial to try to identify which patients had the greatest risk for needing a pacemaker after a CoreValve implant has not yet finished, he said. It’s possible that certain patients with preexisting conduction abnormalities, such as a right bundle branch block coupled with a left anterior fascicular block, have the greatest vulnerability.
Patients for whom the Sapien system remains ideal are those with a narrow sinus of Valsalva, because the longer CoreValve frame crosses the sinus and may compromise coronary blood flow in patients with a narrow sinus, Dr. Popma said.
The choice between CoreValve and Sapien systems will grow even more complicated for U.S. cardiologists and surgeons when the Sapien XT valve system receives FDA marketing approval, likely later this year. The Sapien XT delivery sheath matches the 18F size of CoreValve and will also come in a 29-mm size, blunting two of CoreValves main advantages.
Medtronic also faces charges of patent infringement by its CoreValve in a court case initiated by Edwards. In mid-January, a jury in a U.S. District Court assessed a penalty of $394 million against Medtronic. Edwards is also seeking a court-ordered halt to U.S. marketing of CoreValve. But Medtronic is appealing the jury verdict and continues to fight the injunction, and a company spokesperson said in an interview that the legal maneuverings will likely take at least another year to fully resolve. In the meantime, Medtronic began U.S. distribution of the CoreValve on Jan. 17.
Dr. Block said that his institution received a research grant to participate in Sapien trials. Dr. Cohen said that he has been a consultant to Medtronic and Edwards. Dr. Popma said that his institution received research support from Medtronic and that he has been a consultant to and received research support from Boston Scientific.
When the Food and Drug Administration in January granted marketing approval to a second transcatheter aortic valve replacement system for inoperable patients with aortic stenosis, the CoreValve marketed by Medtronic, the new valve conceded a greater than 2-year head start to the first system on the U.S. market, Sapien marketed by Edwards.
But cardiologists see that 2-year edge in familiarity eclipsed for at least some patients by two major advantages that CoreValve currently holds over Sapien: delivery via a significantly thinner sheath, and the option of larger-diameter valves that allow replacement in patients with a wider aortic annulus.
The CoreValve delivery sheath is 18 French, compared with a 22F or 24F size for the Sapien transcatheter aortic valve replacement (TAVR) with U.S. approval, and the Sapien valves come in diameters of 23 and 26 mm, compared with options of 23, 26, 29, and 31 mm for the CoreValve.
"CoreValve is the device of choice for patients with smaller vessel sizes. Sapien has been a wonderful device to use, and we have so much experience with it, but the smaller CoreValve size will allow many more patients to be done with a transfemoral approach," said Dr. Peter C. Block, a professor of medicine at Emory University in Atlanta and an interventional cardiologist who performs TAVR.
"More patients will qualify for TAVR and will be treated transfemorally with the larger valve diameters and smaller sheath size," agreed Dr. Mauricio G. Cohen, director of cardiac catheterization at the University of Miami and TAVR interventionalist. Another potential positive of having CoreValve on the U.S. market is that competition between the two options will likely drive down the cost of a TAVR system, which until now has run more than $30,000, Dr. Cohen said in an interview.
CoreValve received FDA approval less than 3 months after researchers first reported data from the company’s U.S. Pivotal Trial Extreme Risk Iliofemoral Study last October at the TCT (Transcatheter Cardiovascular Therapeutics) annual meeting. In that study, 471 inoperable aortic stenosis patients had a 26% 1-year rate of death or major stroke, substantially surpassing the 43% rate that the study set up as the target for superiority, reported Dr. Jeffrey J. Popma, lead investigator on the study.
Dr. Popma warned against comparing CoreValve’s efficacy and safety performance in the trial and the Sapien system’s performance in its pivotal trial in inoperable patients, the PARTNER cohort B trial (N. Engl. J. Med. 2010;363:1597-607). "It’s very difficult to make cross-trial comparison," he said in an interview, a limitation also noted by Dr. Cohen and Dr. Block. But Dr. Popma highlighted the 2.4% 30-day stroke rate in the pivotal trial, and a 1.8% 30-day stroke rate seen with CoreValve in inoperable patients in a continued access program at the trial’s study sites. He also highlighted the 11% rate of moderate paravalvular aortic regurgitation after 30 days that dropped to a 4% rate after 1 year.
Perhaps the biggest downside of CoreValve’s performance in the pivotal trial was that 22% of patients required a permanent pacemaker implant within the first 30 days, increasing to 27% of patients with 1-year follow-up. Increased risk for a pacemaker is an inherent downside of CoreValve because of its longer size compared with the Sapien valve and how the CoreValve sits in the aortic annulus. The CoreValve is designed for supravalvular placement and anchoring in the left ventricular outflow tract near the left bundle branch that can result in mechanical irritation and arrhythmia with the need for pacing, explained Dr. Popma, professor of medicine at Harvard University and an interventional cardiologist at Beth Israel Deaconess Medical Center, Boston.
"I think our pacemaker rate was very acceptable. I don’t think it will ever be as low as with Sapien, but it’s a worthwhile trade-off because the CoreValve functions well and results in a low rate of paravalvular regurgitation," he said.
Dr. Popma also stressed that 1-year mortality was no greater among the patients who required a pacemaker implant in the pivotal trial. A subgroup analysis of results from the trial to try to identify which patients had the greatest risk for needing a pacemaker after a CoreValve implant has not yet finished, he said. It’s possible that certain patients with preexisting conduction abnormalities, such as a right bundle branch block coupled with a left anterior fascicular block, have the greatest vulnerability.
Patients for whom the Sapien system remains ideal are those with a narrow sinus of Valsalva, because the longer CoreValve frame crosses the sinus and may compromise coronary blood flow in patients with a narrow sinus, Dr. Popma said.
The choice between CoreValve and Sapien systems will grow even more complicated for U.S. cardiologists and surgeons when the Sapien XT valve system receives FDA marketing approval, likely later this year. The Sapien XT delivery sheath matches the 18F size of CoreValve and will also come in a 29-mm size, blunting two of CoreValves main advantages.
Medtronic also faces charges of patent infringement by its CoreValve in a court case initiated by Edwards. In mid-January, a jury in a U.S. District Court assessed a penalty of $394 million against Medtronic. Edwards is also seeking a court-ordered halt to U.S. marketing of CoreValve. But Medtronic is appealing the jury verdict and continues to fight the injunction, and a company spokesperson said in an interview that the legal maneuverings will likely take at least another year to fully resolve. In the meantime, Medtronic began U.S. distribution of the CoreValve on Jan. 17.
Dr. Block said that his institution received a research grant to participate in Sapien trials. Dr. Cohen said that he has been a consultant to Medtronic and Edwards. Dr. Popma said that his institution received research support from Medtronic and that he has been a consultant to and received research support from Boston Scientific.
When the Food and Drug Administration in January granted marketing approval to a second transcatheter aortic valve replacement system for inoperable patients with aortic stenosis, the CoreValve marketed by Medtronic, the new valve conceded a greater than 2-year head start to the first system on the U.S. market, Sapien marketed by Edwards.
But cardiologists see that 2-year edge in familiarity eclipsed for at least some patients by two major advantages that CoreValve currently holds over Sapien: delivery via a significantly thinner sheath, and the option of larger-diameter valves that allow replacement in patients with a wider aortic annulus.
The CoreValve delivery sheath is 18 French, compared with a 22F or 24F size for the Sapien transcatheter aortic valve replacement (TAVR) with U.S. approval, and the Sapien valves come in diameters of 23 and 26 mm, compared with options of 23, 26, 29, and 31 mm for the CoreValve.
"CoreValve is the device of choice for patients with smaller vessel sizes. Sapien has been a wonderful device to use, and we have so much experience with it, but the smaller CoreValve size will allow many more patients to be done with a transfemoral approach," said Dr. Peter C. Block, a professor of medicine at Emory University in Atlanta and an interventional cardiologist who performs TAVR.
"More patients will qualify for TAVR and will be treated transfemorally with the larger valve diameters and smaller sheath size," agreed Dr. Mauricio G. Cohen, director of cardiac catheterization at the University of Miami and TAVR interventionalist. Another potential positive of having CoreValve on the U.S. market is that competition between the two options will likely drive down the cost of a TAVR system, which until now has run more than $30,000, Dr. Cohen said in an interview.
CoreValve received FDA approval less than 3 months after researchers first reported data from the company’s U.S. Pivotal Trial Extreme Risk Iliofemoral Study last October at the TCT (Transcatheter Cardiovascular Therapeutics) annual meeting. In that study, 471 inoperable aortic stenosis patients had a 26% 1-year rate of death or major stroke, substantially surpassing the 43% rate that the study set up as the target for superiority, reported Dr. Jeffrey J. Popma, lead investigator on the study.
Dr. Popma warned against comparing CoreValve’s efficacy and safety performance in the trial and the Sapien system’s performance in its pivotal trial in inoperable patients, the PARTNER cohort B trial (N. Engl. J. Med. 2010;363:1597-607). "It’s very difficult to make cross-trial comparison," he said in an interview, a limitation also noted by Dr. Cohen and Dr. Block. But Dr. Popma highlighted the 2.4% 30-day stroke rate in the pivotal trial, and a 1.8% 30-day stroke rate seen with CoreValve in inoperable patients in a continued access program at the trial’s study sites. He also highlighted the 11% rate of moderate paravalvular aortic regurgitation after 30 days that dropped to a 4% rate after 1 year.
Perhaps the biggest downside of CoreValve’s performance in the pivotal trial was that 22% of patients required a permanent pacemaker implant within the first 30 days, increasing to 27% of patients with 1-year follow-up. Increased risk for a pacemaker is an inherent downside of CoreValve because of its longer size compared with the Sapien valve and how the CoreValve sits in the aortic annulus. The CoreValve is designed for supravalvular placement and anchoring in the left ventricular outflow tract near the left bundle branch that can result in mechanical irritation and arrhythmia with the need for pacing, explained Dr. Popma, professor of medicine at Harvard University and an interventional cardiologist at Beth Israel Deaconess Medical Center, Boston.
"I think our pacemaker rate was very acceptable. I don’t think it will ever be as low as with Sapien, but it’s a worthwhile trade-off because the CoreValve functions well and results in a low rate of paravalvular regurgitation," he said.
Dr. Popma also stressed that 1-year mortality was no greater among the patients who required a pacemaker implant in the pivotal trial. A subgroup analysis of results from the trial to try to identify which patients had the greatest risk for needing a pacemaker after a CoreValve implant has not yet finished, he said. It’s possible that certain patients with preexisting conduction abnormalities, such as a right bundle branch block coupled with a left anterior fascicular block, have the greatest vulnerability.
Patients for whom the Sapien system remains ideal are those with a narrow sinus of Valsalva, because the longer CoreValve frame crosses the sinus and may compromise coronary blood flow in patients with a narrow sinus, Dr. Popma said.
The choice between CoreValve and Sapien systems will grow even more complicated for U.S. cardiologists and surgeons when the Sapien XT valve system receives FDA marketing approval, likely later this year. The Sapien XT delivery sheath matches the 18F size of CoreValve and will also come in a 29-mm size, blunting two of CoreValves main advantages.
Medtronic also faces charges of patent infringement by its CoreValve in a court case initiated by Edwards. In mid-January, a jury in a U.S. District Court assessed a penalty of $394 million against Medtronic. Edwards is also seeking a court-ordered halt to U.S. marketing of CoreValve. But Medtronic is appealing the jury verdict and continues to fight the injunction, and a company spokesperson said in an interview that the legal maneuverings will likely take at least another year to fully resolve. In the meantime, Medtronic began U.S. distribution of the CoreValve on Jan. 17.
Dr. Block said that his institution received a research grant to participate in Sapien trials. Dr. Cohen said that he has been a consultant to Medtronic and Edwards. Dr. Popma said that his institution received research support from Medtronic and that he has been a consultant to and received research support from Boston Scientific.