User login
Psoriasis linked to increased heart failure risk
AMSTERDAM – Psoriasis proved to be independently associated with an increased risk for new-onset heart failure in the first nationwide study to look at a possible relationship.
As word of the newly identified psoriasis/heart failure link spreads, it’s likely cardiologists will receive a growing number of referrals from dermatologists and primary care physicians for evaluation of possible heart failure in psoriasis patients who develop shortness of breath or other symptoms suggestive of ventricular dysfunction, Dr. Usman Khalid said at the annual congress of the European Society of Cardiology.
"Our results underlie the importance of considering the psoriatic population as a high-risk patient group in terms of cardiovascular risk. We encourage early screening for cardiovascular risk factors in psoriasis patients," he said.
Psoriasis is known to be associated with an increased cardiovascular event rate, presumably due to shared systemic inflammatory pathways, but the association between the dermatologic disease and heart failure, specifically, hadn’t been looked at in depth prior to Dr. Khalid’s presentation of a Danish study involving all 5.85 million Danish adults, who were followed from 1997 through 2009.
Among the Danish population without known heart failure at baseline were 57,049 individuals with mild psoriasis – identified by their use of prescription topical agents – and 11,638 others with severe psoriasis. The incidence of new-onset heart failure during follow-up was 2.27 cases per 1,000 person-years in subjects without psoriasis and a significantly greater 4.02 per 1,000 person-years in those with mild psoriasis and 4.50 per 1,000 person-years in individuals with severe psoriasis, reported Dr. Khalid of the University of Copenhagen.
In a multivariate regression analysis adjusted for demographic and socioeconomic factors, comorbid conditions, and use of cardiovascular medications, the likelihood of developing new-onset heart failure during follow-up was 64% greater among individuals with mild psoriasis and 85% greater in those with severe psoriasis than in psoriasis-free subjects.
Because of limitations in the registry database, it is not possible to determine the proportion of new-onset heart failure among psoriasis patients that involved systolic as opposed to diastolic dysfunction, or ischemic versus nonischemic etiology, Dr. Khalid said in response to audience questions.
This study was supported by a research grant from Leo Pharma. Dr. Khalid reported having no financial conflicts of interest.
AMSTERDAM – Psoriasis proved to be independently associated with an increased risk for new-onset heart failure in the first nationwide study to look at a possible relationship.
As word of the newly identified psoriasis/heart failure link spreads, it’s likely cardiologists will receive a growing number of referrals from dermatologists and primary care physicians for evaluation of possible heart failure in psoriasis patients who develop shortness of breath or other symptoms suggestive of ventricular dysfunction, Dr. Usman Khalid said at the annual congress of the European Society of Cardiology.
"Our results underlie the importance of considering the psoriatic population as a high-risk patient group in terms of cardiovascular risk. We encourage early screening for cardiovascular risk factors in psoriasis patients," he said.
Psoriasis is known to be associated with an increased cardiovascular event rate, presumably due to shared systemic inflammatory pathways, but the association between the dermatologic disease and heart failure, specifically, hadn’t been looked at in depth prior to Dr. Khalid’s presentation of a Danish study involving all 5.85 million Danish adults, who were followed from 1997 through 2009.
Among the Danish population without known heart failure at baseline were 57,049 individuals with mild psoriasis – identified by their use of prescription topical agents – and 11,638 others with severe psoriasis. The incidence of new-onset heart failure during follow-up was 2.27 cases per 1,000 person-years in subjects without psoriasis and a significantly greater 4.02 per 1,000 person-years in those with mild psoriasis and 4.50 per 1,000 person-years in individuals with severe psoriasis, reported Dr. Khalid of the University of Copenhagen.
In a multivariate regression analysis adjusted for demographic and socioeconomic factors, comorbid conditions, and use of cardiovascular medications, the likelihood of developing new-onset heart failure during follow-up was 64% greater among individuals with mild psoriasis and 85% greater in those with severe psoriasis than in psoriasis-free subjects.
Because of limitations in the registry database, it is not possible to determine the proportion of new-onset heart failure among psoriasis patients that involved systolic as opposed to diastolic dysfunction, or ischemic versus nonischemic etiology, Dr. Khalid said in response to audience questions.
This study was supported by a research grant from Leo Pharma. Dr. Khalid reported having no financial conflicts of interest.
AMSTERDAM – Psoriasis proved to be independently associated with an increased risk for new-onset heart failure in the first nationwide study to look at a possible relationship.
As word of the newly identified psoriasis/heart failure link spreads, it’s likely cardiologists will receive a growing number of referrals from dermatologists and primary care physicians for evaluation of possible heart failure in psoriasis patients who develop shortness of breath or other symptoms suggestive of ventricular dysfunction, Dr. Usman Khalid said at the annual congress of the European Society of Cardiology.
"Our results underlie the importance of considering the psoriatic population as a high-risk patient group in terms of cardiovascular risk. We encourage early screening for cardiovascular risk factors in psoriasis patients," he said.
Psoriasis is known to be associated with an increased cardiovascular event rate, presumably due to shared systemic inflammatory pathways, but the association between the dermatologic disease and heart failure, specifically, hadn’t been looked at in depth prior to Dr. Khalid’s presentation of a Danish study involving all 5.85 million Danish adults, who were followed from 1997 through 2009.
Among the Danish population without known heart failure at baseline were 57,049 individuals with mild psoriasis – identified by their use of prescription topical agents – and 11,638 others with severe psoriasis. The incidence of new-onset heart failure during follow-up was 2.27 cases per 1,000 person-years in subjects without psoriasis and a significantly greater 4.02 per 1,000 person-years in those with mild psoriasis and 4.50 per 1,000 person-years in individuals with severe psoriasis, reported Dr. Khalid of the University of Copenhagen.
In a multivariate regression analysis adjusted for demographic and socioeconomic factors, comorbid conditions, and use of cardiovascular medications, the likelihood of developing new-onset heart failure during follow-up was 64% greater among individuals with mild psoriasis and 85% greater in those with severe psoriasis than in psoriasis-free subjects.
Because of limitations in the registry database, it is not possible to determine the proportion of new-onset heart failure among psoriasis patients that involved systolic as opposed to diastolic dysfunction, or ischemic versus nonischemic etiology, Dr. Khalid said in response to audience questions.
This study was supported by a research grant from Leo Pharma. Dr. Khalid reported having no financial conflicts of interest.
AT THE ESC CONGRESS 2013
Major finding: Danish patients with mild psoriasis developed new-onset heart failure at the rate of 4.02 cases per 1,000 person-years of follow-up, while those with severe psoriasis did so at a pace of 4.50 cases per 1,000 person-years, both significantly higher rates than the 2.27 per 1,000 person-years in the general psoriasis-free population.
Data source: This was a Danish nationwide registry study that included all 5.85 million Danish adults. They were followed during 1997-2009 for development of heart failure.
Disclosures: The study was supported by a research grant from Leo Pharma. The presenter reported having no financial conflicts of interest.
CRT appears deadly in short-QRS patients
AMSTERDAM – Patients with severe heart failure and a narrow QRS duration who received cardiac resynchronization therapy had more than twice the rate of cardiovascular deaths as patients who did not undergo active CRT treatment in a multinational randomized study with 809 patients.
The study’s enrollment criteria focused on patients with a QRS duration of less than 130 msec, and in this group active CRT produced no benefit, compared with the CRT function turned off in control patients for the study’s primary efficacy endpoint of death from any cause or first hospitalization for worsening heart failure.
In addition, during an average 19 months’ follow-up, patients in the CRT-on group had an all-cause death rate nearly five percentage points above the controls, an 80% relatively increased hazard, and a cardiovascular death rate also five percentage points above the controls, a greater than two-fold relative hazard, Dr. Johannes Holzmeister said at the annual congress of the European Society of Cardiology.
Both increased hazard rates were statistically significant and were a "surprising, unexpected finding," said Dr. Holzmeister, a cardiologist at the University of Zurich.
"This is the final nail in the coffin for CRT in patients with only slightly-prolonged QRS," commented Dr. Douglas P. Zipes, a professor and electrophysiologist at Indiana University in Indianapolis.
Existing recommendations from the American College of Cardiology and American Heart Association (J. Am. Coll. Cardiol. 2013 [doi:10.1016/j.jacc2013.05.019]) restrict unequivocal endorsement of CRT to heart failure patients with a QRS duration of at least 150 msec and say that CRT can be useful for patients with New York Heart Association class II, III, or IV heart failure, left bundle branch block (LBBB), and a QRS duration of 120-149 msec. Recommendations from the European Society of Cardiology are more permissive, calling CRT recommended for patients in sinus rhythm with a QRS duration of at least 120 msec, LBBB morphology on their QRS wave, and an ejection fraction of at least 35% who are expected to remain in good functional status for more than 1 year (Eur. Heart J. 2012;33:1787-847).
The ESC recommendations also say that CRT should be considered in patients with a QRS duration of at least 150 msec, irrespective of their QRS morphology.
It seems like many patients with shorter QRS durations have been receiving CRT treatment. During his report at the meeting, Dr. Holzmeister cited data from a 2008-2009 European-wide survey of CRT patients by the European Heart Rhythm Association, which showed that, 4-5 years ago, 9% of patients in Europe who received a CRT device had a QRS duration of less than 120 msec and another 10% of the CRT recipients had durations of 120-129 msec (Eur. Heart J. 2009;30:2450-60).
Substantial numbers of patients continue to receive CRT treatment with a QRS duration of 120-129 msec, a treatment that the data indicate should be a "last resort," said Dr. John G.F. Cleland, professor of medicine and a heart failure specialist at the University of Hull (England).
Although existing society recommendations sanction CRT for selected heart failure patients with a QRS duration of 120-149 msec, many experts have become convinced that the field needs a new set of QRS criteria.
"I think the cutoff for CRT implantation should be 150 msec," said Dr. Gordon F. Tomaselli, professor of cardiology and an electrophysiologist at Johns Hopkins University in Baltimore. "Patients with a QRS duration of less than 150 msec, particularly without left bundle branch block, should probably not get CRTs unless some special circumstances are present," he said in an interview.
"This reinforces the conclusion that we should not use CRT devices in heart failure patients with a QRS duration of less than 130 msec," commented Dr. Stefan D. Anker, professor and cardiologist at Charitè Hospital in Berlin and designated discussant for Dr. Holzmeister’s talk at the meeting. Dr. Anker called CRT use in patients with QRS durations of 130-149 msec uncertain and in need of more evidence for efficacy and safety from trials. "What is certain is that in patients with a QRS of 150 msec or greater there is a strong mortality reduction with CRT," he said.
The Echocardiography Guided Cardiac Resynchronization Therapy (EchoCRT) study randomized 809 patients with a QRS duration of less than 130 msec and left ventricular ejection fraction of 35% or less, and also required that patients meet criteria for having left-ventricular dyssynchrony detected by echocardiography. All patients received a CRT device, which was turned on in 404 patients. The study began in August 2008, and enrollment stopped on March 13, 2013, because of futility and a potential for harm.
At that time, cardiovascular deaths had occurred in 9% of patients with their CRT turned on and in 4% of those with the device turned off. Concurrent with Dr. Holzmeister’s report at the meeting, an article with the results was published online (N. Engl. J. Med. 2013 [doi:10.1056/NEJMoa1306687]).
The EchoCRT trial was sponsored by Biotronik and GE Healthcare. Dr. Holzmeister said that he has been a consultant to Biotronik. Dr. Zipes said that he has been a consultant to Health System Networks and consults for and is a co-owner of Insight Telehealth. Dr. Anker said that he has received fees and honoraria from St. Jude, Abbott Vascular, and several other drug and device companies. Dr. Tomaselli said that he had no disclosures.
On Twitter @mitchelzoler
The results from EchoCRT fit well with the results of a meta-analysis that my associates and I recently ran on data from 3,782 heart failure patients treated with CRT in five trials sponsored by Medtronic. The results showed that QRS duration was a powerful predictor of the effects of CRT on morbidity and mortality.
The meta-analysis results confirmed the benefit of CRT in patients with mild, moderate, or severe heart failure symptoms, in sinus rhythm, and with a QRS duration of at least 140 msec, and in these patients CRT is standard of care. The results also showed that the benefits from CRT diminish as the QRS duration goes below 140 msec. Patients with a QRS duration of 130-139 msec are in a gray zone. If they need a defibrillator, then making it a CRT device makes sense, but if no device implant is planned then continued attempts at medical treatment are probably better than going to CRT.

|
|
I would avoid CRT in patients with a QRS of less than 130 msec, and now the EchoCRT results show evidence of harm in these patients. A lot of patients with a QRS duration of 120-129 msec have been receiving CRT devices when the chances of benefit are small. This might be a treatment of last resort, but only when you have exhausted all the other treatment alternatives.
The meta-analysis also showed that QRS duration was the only independent predictor of CRT outcomes (Eur. Heart J. 2013 [doi:10.1093/eurheartj/eht290]). QRS morphology – whether or not there is a left bundle branch block – was not a significant factor relative to QRS duration. The EchoCRT results also showed that echocardiography used to diagnose left-ventricular dyssynchrony failed to identify a subgroup of patients with a narrow QRS duration who benefited from CRT.
Dr. John G.F. Cleland is professor of medicine at the University of Hull (England). He has been a consultant to Biotronik, St. Jude, and Medtronic. He made these comments in an interview.
The results from EchoCRT fit well with the results of a meta-analysis that my associates and I recently ran on data from 3,782 heart failure patients treated with CRT in five trials sponsored by Medtronic. The results showed that QRS duration was a powerful predictor of the effects of CRT on morbidity and mortality.
The meta-analysis results confirmed the benefit of CRT in patients with mild, moderate, or severe heart failure symptoms, in sinus rhythm, and with a QRS duration of at least 140 msec, and in these patients CRT is standard of care. The results also showed that the benefits from CRT diminish as the QRS duration goes below 140 msec. Patients with a QRS duration of 130-139 msec are in a gray zone. If they need a defibrillator, then making it a CRT device makes sense, but if no device implant is planned then continued attempts at medical treatment are probably better than going to CRT.

|
|
I would avoid CRT in patients with a QRS of less than 130 msec, and now the EchoCRT results show evidence of harm in these patients. A lot of patients with a QRS duration of 120-129 msec have been receiving CRT devices when the chances of benefit are small. This might be a treatment of last resort, but only when you have exhausted all the other treatment alternatives.
The meta-analysis also showed that QRS duration was the only independent predictor of CRT outcomes (Eur. Heart J. 2013 [doi:10.1093/eurheartj/eht290]). QRS morphology – whether or not there is a left bundle branch block – was not a significant factor relative to QRS duration. The EchoCRT results also showed that echocardiography used to diagnose left-ventricular dyssynchrony failed to identify a subgroup of patients with a narrow QRS duration who benefited from CRT.
Dr. John G.F. Cleland is professor of medicine at the University of Hull (England). He has been a consultant to Biotronik, St. Jude, and Medtronic. He made these comments in an interview.
The results from EchoCRT fit well with the results of a meta-analysis that my associates and I recently ran on data from 3,782 heart failure patients treated with CRT in five trials sponsored by Medtronic. The results showed that QRS duration was a powerful predictor of the effects of CRT on morbidity and mortality.
The meta-analysis results confirmed the benefit of CRT in patients with mild, moderate, or severe heart failure symptoms, in sinus rhythm, and with a QRS duration of at least 140 msec, and in these patients CRT is standard of care. The results also showed that the benefits from CRT diminish as the QRS duration goes below 140 msec. Patients with a QRS duration of 130-139 msec are in a gray zone. If they need a defibrillator, then making it a CRT device makes sense, but if no device implant is planned then continued attempts at medical treatment are probably better than going to CRT.

|
|
I would avoid CRT in patients with a QRS of less than 130 msec, and now the EchoCRT results show evidence of harm in these patients. A lot of patients with a QRS duration of 120-129 msec have been receiving CRT devices when the chances of benefit are small. This might be a treatment of last resort, but only when you have exhausted all the other treatment alternatives.
The meta-analysis also showed that QRS duration was the only independent predictor of CRT outcomes (Eur. Heart J. 2013 [doi:10.1093/eurheartj/eht290]). QRS morphology – whether or not there is a left bundle branch block – was not a significant factor relative to QRS duration. The EchoCRT results also showed that echocardiography used to diagnose left-ventricular dyssynchrony failed to identify a subgroup of patients with a narrow QRS duration who benefited from CRT.
Dr. John G.F. Cleland is professor of medicine at the University of Hull (England). He has been a consultant to Biotronik, St. Jude, and Medtronic. He made these comments in an interview.
AMSTERDAM – Patients with severe heart failure and a narrow QRS duration who received cardiac resynchronization therapy had more than twice the rate of cardiovascular deaths as patients who did not undergo active CRT treatment in a multinational randomized study with 809 patients.
The study’s enrollment criteria focused on patients with a QRS duration of less than 130 msec, and in this group active CRT produced no benefit, compared with the CRT function turned off in control patients for the study’s primary efficacy endpoint of death from any cause or first hospitalization for worsening heart failure.
In addition, during an average 19 months’ follow-up, patients in the CRT-on group had an all-cause death rate nearly five percentage points above the controls, an 80% relatively increased hazard, and a cardiovascular death rate also five percentage points above the controls, a greater than two-fold relative hazard, Dr. Johannes Holzmeister said at the annual congress of the European Society of Cardiology.
Both increased hazard rates were statistically significant and were a "surprising, unexpected finding," said Dr. Holzmeister, a cardiologist at the University of Zurich.
"This is the final nail in the coffin for CRT in patients with only slightly-prolonged QRS," commented Dr. Douglas P. Zipes, a professor and electrophysiologist at Indiana University in Indianapolis.
Existing recommendations from the American College of Cardiology and American Heart Association (J. Am. Coll. Cardiol. 2013 [doi:10.1016/j.jacc2013.05.019]) restrict unequivocal endorsement of CRT to heart failure patients with a QRS duration of at least 150 msec and say that CRT can be useful for patients with New York Heart Association class II, III, or IV heart failure, left bundle branch block (LBBB), and a QRS duration of 120-149 msec. Recommendations from the European Society of Cardiology are more permissive, calling CRT recommended for patients in sinus rhythm with a QRS duration of at least 120 msec, LBBB morphology on their QRS wave, and an ejection fraction of at least 35% who are expected to remain in good functional status for more than 1 year (Eur. Heart J. 2012;33:1787-847).
The ESC recommendations also say that CRT should be considered in patients with a QRS duration of at least 150 msec, irrespective of their QRS morphology.
It seems like many patients with shorter QRS durations have been receiving CRT treatment. During his report at the meeting, Dr. Holzmeister cited data from a 2008-2009 European-wide survey of CRT patients by the European Heart Rhythm Association, which showed that, 4-5 years ago, 9% of patients in Europe who received a CRT device had a QRS duration of less than 120 msec and another 10% of the CRT recipients had durations of 120-129 msec (Eur. Heart J. 2009;30:2450-60).
Substantial numbers of patients continue to receive CRT treatment with a QRS duration of 120-129 msec, a treatment that the data indicate should be a "last resort," said Dr. John G.F. Cleland, professor of medicine and a heart failure specialist at the University of Hull (England).
Although existing society recommendations sanction CRT for selected heart failure patients with a QRS duration of 120-149 msec, many experts have become convinced that the field needs a new set of QRS criteria.
"I think the cutoff for CRT implantation should be 150 msec," said Dr. Gordon F. Tomaselli, professor of cardiology and an electrophysiologist at Johns Hopkins University in Baltimore. "Patients with a QRS duration of less than 150 msec, particularly without left bundle branch block, should probably not get CRTs unless some special circumstances are present," he said in an interview.
"This reinforces the conclusion that we should not use CRT devices in heart failure patients with a QRS duration of less than 130 msec," commented Dr. Stefan D. Anker, professor and cardiologist at Charitè Hospital in Berlin and designated discussant for Dr. Holzmeister’s talk at the meeting. Dr. Anker called CRT use in patients with QRS durations of 130-149 msec uncertain and in need of more evidence for efficacy and safety from trials. "What is certain is that in patients with a QRS of 150 msec or greater there is a strong mortality reduction with CRT," he said.
The Echocardiography Guided Cardiac Resynchronization Therapy (EchoCRT) study randomized 809 patients with a QRS duration of less than 130 msec and left ventricular ejection fraction of 35% or less, and also required that patients meet criteria for having left-ventricular dyssynchrony detected by echocardiography. All patients received a CRT device, which was turned on in 404 patients. The study began in August 2008, and enrollment stopped on March 13, 2013, because of futility and a potential for harm.
At that time, cardiovascular deaths had occurred in 9% of patients with their CRT turned on and in 4% of those with the device turned off. Concurrent with Dr. Holzmeister’s report at the meeting, an article with the results was published online (N. Engl. J. Med. 2013 [doi:10.1056/NEJMoa1306687]).
The EchoCRT trial was sponsored by Biotronik and GE Healthcare. Dr. Holzmeister said that he has been a consultant to Biotronik. Dr. Zipes said that he has been a consultant to Health System Networks and consults for and is a co-owner of Insight Telehealth. Dr. Anker said that he has received fees and honoraria from St. Jude, Abbott Vascular, and several other drug and device companies. Dr. Tomaselli said that he had no disclosures.
On Twitter @mitchelzoler
AMSTERDAM – Patients with severe heart failure and a narrow QRS duration who received cardiac resynchronization therapy had more than twice the rate of cardiovascular deaths as patients who did not undergo active CRT treatment in a multinational randomized study with 809 patients.
The study’s enrollment criteria focused on patients with a QRS duration of less than 130 msec, and in this group active CRT produced no benefit, compared with the CRT function turned off in control patients for the study’s primary efficacy endpoint of death from any cause or first hospitalization for worsening heart failure.
In addition, during an average 19 months’ follow-up, patients in the CRT-on group had an all-cause death rate nearly five percentage points above the controls, an 80% relatively increased hazard, and a cardiovascular death rate also five percentage points above the controls, a greater than two-fold relative hazard, Dr. Johannes Holzmeister said at the annual congress of the European Society of Cardiology.
Both increased hazard rates were statistically significant and were a "surprising, unexpected finding," said Dr. Holzmeister, a cardiologist at the University of Zurich.
"This is the final nail in the coffin for CRT in patients with only slightly-prolonged QRS," commented Dr. Douglas P. Zipes, a professor and electrophysiologist at Indiana University in Indianapolis.
Existing recommendations from the American College of Cardiology and American Heart Association (J. Am. Coll. Cardiol. 2013 [doi:10.1016/j.jacc2013.05.019]) restrict unequivocal endorsement of CRT to heart failure patients with a QRS duration of at least 150 msec and say that CRT can be useful for patients with New York Heart Association class II, III, or IV heart failure, left bundle branch block (LBBB), and a QRS duration of 120-149 msec. Recommendations from the European Society of Cardiology are more permissive, calling CRT recommended for patients in sinus rhythm with a QRS duration of at least 120 msec, LBBB morphology on their QRS wave, and an ejection fraction of at least 35% who are expected to remain in good functional status for more than 1 year (Eur. Heart J. 2012;33:1787-847).
The ESC recommendations also say that CRT should be considered in patients with a QRS duration of at least 150 msec, irrespective of their QRS morphology.
It seems like many patients with shorter QRS durations have been receiving CRT treatment. During his report at the meeting, Dr. Holzmeister cited data from a 2008-2009 European-wide survey of CRT patients by the European Heart Rhythm Association, which showed that, 4-5 years ago, 9% of patients in Europe who received a CRT device had a QRS duration of less than 120 msec and another 10% of the CRT recipients had durations of 120-129 msec (Eur. Heart J. 2009;30:2450-60).
Substantial numbers of patients continue to receive CRT treatment with a QRS duration of 120-129 msec, a treatment that the data indicate should be a "last resort," said Dr. John G.F. Cleland, professor of medicine and a heart failure specialist at the University of Hull (England).
Although existing society recommendations sanction CRT for selected heart failure patients with a QRS duration of 120-149 msec, many experts have become convinced that the field needs a new set of QRS criteria.
"I think the cutoff for CRT implantation should be 150 msec," said Dr. Gordon F. Tomaselli, professor of cardiology and an electrophysiologist at Johns Hopkins University in Baltimore. "Patients with a QRS duration of less than 150 msec, particularly without left bundle branch block, should probably not get CRTs unless some special circumstances are present," he said in an interview.
"This reinforces the conclusion that we should not use CRT devices in heart failure patients with a QRS duration of less than 130 msec," commented Dr. Stefan D. Anker, professor and cardiologist at Charitè Hospital in Berlin and designated discussant for Dr. Holzmeister’s talk at the meeting. Dr. Anker called CRT use in patients with QRS durations of 130-149 msec uncertain and in need of more evidence for efficacy and safety from trials. "What is certain is that in patients with a QRS of 150 msec or greater there is a strong mortality reduction with CRT," he said.
The Echocardiography Guided Cardiac Resynchronization Therapy (EchoCRT) study randomized 809 patients with a QRS duration of less than 130 msec and left ventricular ejection fraction of 35% or less, and also required that patients meet criteria for having left-ventricular dyssynchrony detected by echocardiography. All patients received a CRT device, which was turned on in 404 patients. The study began in August 2008, and enrollment stopped on March 13, 2013, because of futility and a potential for harm.
At that time, cardiovascular deaths had occurred in 9% of patients with their CRT turned on and in 4% of those with the device turned off. Concurrent with Dr. Holzmeister’s report at the meeting, an article with the results was published online (N. Engl. J. Med. 2013 [doi:10.1056/NEJMoa1306687]).
The EchoCRT trial was sponsored by Biotronik and GE Healthcare. Dr. Holzmeister said that he has been a consultant to Biotronik. Dr. Zipes said that he has been a consultant to Health System Networks and consults for and is a co-owner of Insight Telehealth. Dr. Anker said that he has received fees and honoraria from St. Jude, Abbott Vascular, and several other drug and device companies. Dr. Tomaselli said that he had no disclosures.
On Twitter @mitchelzoler
AT THE ESC CONGRESS 2013
Major finding: Cardiovascular death occurred in 9% of patients on active cardiac resynchronization therapy and in 4% of control patients.
Data source: Data came from the EchoCRT trial, a multicenter, randomized trial with 809 patients with NYHA class III or IV heart failure and a QRS duration of less than 130 msec.
Disclosures: EchoCRT was sponsored by Biotronik and GE Healthcare. Dr. Holzmeister has been a consultant to Biotronik. Dr. Zipes has been a consultant to Health System Networks and consults for and is a co-owner of Insight Telehealth. Dr. Anker has received fees and honoraria from St. Jude, Abbott Vascular, and several other drug and device companies. Dr. Tomaselli said that he had no disclosures. Dr. Cleland said that he has been a consultant to Biotronik, Medtronic, and St. Jude.
New heart failure inotrope could be ‘Holy Grail’
AMSTERDAM – The novel intravenous inotropic agent omecamtiv mecarbil achieved greater reduction in dyspnea scores and less likelihood of worsening heart failure at the highest dose studied, compared with placebo, in patients with acute decompensated heart failure in a phase II, dose-ranging study.
The drug also demonstrated a strong dose-dependent increase in systolic ejection time, no evidence of a proarrhythmic effect, and a greater reduction in heart rate with a larger increase in blood pressure than in placebo-treated controls, Dr. John R. Teerlink reported at the annual congress of the European Society of Cardiology.
In the overall population on omecamtiv mecarbil, however, which included the patients in the low- and intermediate-dose arms of this dose-ranging study, the dyspnea response rate wasn’t better than with placebo. Hence, the drug’s future is uncertain.
In a press release following Dr. Teerlink’s presentation, Amgen, cosponsor of the phase II ATOMIC-AHF (Acute Treatment With Omecamtiv Mecarbil to Increase Contractility in Acute Heart Failure) trial, said that no decision has been made as to whether to move omecamtiv mecarbil into phase III clinical trials. Dr. Teerlink thinks the drug has earned a shot.
"I personally believe this drug has continued to demonstrate its promise all along the way. It could replace milrinone and dobutamine in the hospital to improve cardiac performance," predicted Dr. Teerlink, professor of medicine at the University of California, San Francisco, and director of the heart failure program at the San Francisco Veterans Affairs Medical Center.
Omecamtiv mecarbil is a novel selective cardiac myosin activator. It increases the entry rate of myosin into the force-producing state with actin. It boosts cardiac output through prolongation of the duration of systole and increased stroke volume, with no change in myocardial contractility and no increases in myocardial oxygen demand or myocyte calcium.
"It’s essentially producing more hands pulling on the rope," the cardiologist explained.
Discussant Theresa A. McDonagh said omecamtiv mecarbil has drawn great interest within the heart failure field because current inotropes are recognized as "very inadequate."
"It is truly a new inotrope, a drug that acts on the sarcomere itself. It does seem to be free of the nemesis of other inotropes: increased heart rate, greater myocardial oxygen consumption, arrhythmia production, and increased mortality in heart failure. It appears to be the Holy Grail, and may be capable of replacing dobutamine and milrinone," said Dr. McDonagh, professor of heart failure at King’s College London.
"I think this [ATOMIC-AHF trial] is a very promising start for omecamtiv mecarbil in acute heart failure," she added.
ATOMIC-AHF included 613 patients with a left ventricular ejection fraction of 40% or less who were hospitalized for acute heart failure with dyspnea at rest or with minimal exertion despite intravenous diuretic therapy. The study was conducted at 106 sites on three continents. Participants were randomized to 48 hours of intravenous placebo or omecamtiv mecarbil in three different ascending dose regimens.
The primary efficacy endpoint, dyspnea at 6, 24, and 48 hours across the three doses, compared with the pooled placebo group, was not met.
However, patients in the highest-dose omecamtiv mecarbil study arm had a 51% rate of significant improvement in dyspnea on a self-rated 7-point Likert scale at 48 hours, a significantly better response than the 37% rate in matched placebo-treated controls (P = .03). This comparison was a prespecified secondary endpoint in the study.
In addition, patients in the middle- and high-dose omecamtiv mecarbil arms had roughly a 45% reduction in the 7-day incidence of worsening heart failure, compared with the 17% rate in controls.
Not only was there no evidence of a proarrhythmic effect with omecamtiv mecarbil, the drug was associated with a 50% reduction in the incidence of supraventricular tachyarrhythmias, compared with placebo, he continued.
A modest increase in troponin I levels was noted in the omecamtiv mecarbil–treated group, but it was unrelated to plasma drug concentrations, which Dr. Teerlink found reassuring. Nevertheless, Dr. McDonagh said the small increases in troponin "should herald some caution amid the enthusiasm here."
The drug’s mechanism of action, which results in greater systolic ejection time, always raises the concern that it might also shorten diastole and compromise coronary perfusion. However, the observed reduction in heart rate makes that seem less likely, she added.
The ATOMIC-AHF study population was narrowly defined and of a profile commonly seen in clinical practice. They had an estimated glomerular filtration rate of about 50 mL/min, and they presented with dyspnea but didn’t have an acute coronary syndrome.
"This study population is an area of great unmet need in heart failure, with mortality rates of about 10% during their hospitalization and 30% at 1 year following discharge," according to Dr. McDonagh.
Nevertheless, it will be important in phase III testing to see how the drug performs in a broader group of heart failure patients, including those with lower estimated glomerular filtration rates, she added.
In an interview, Dr. Teerlink said that before a decision is reached regarding advancement of omecamtiv mecarbil to phase III studies, investigators and the study sponsors first want to see the data from an ongoing phase II dose-finding study of an oral formulation of the drug in chronic heart failure patients. This study, COSMIC-HF (Chronic Oral Study of Myosin Activation to Increase Contractility in Heart Failure), is still enrolling patients.
"We’ll get the combined data from ATOMIC-AHF and COSMIC-HF and then decide what phase III should look like. There are a lot of options here: IV [intravenous] alone, IV transitioning to chronic oral therapy, or even oral alone," he explained.
"One of the greatest challenges of the ATOMIC-AHF trial has been managing expectations," according to Dr. Teerlink. "Omecamtiv mecarbil is probably one of the most interesting new chemical entities in cardiovascular medicine now, and consequently expectations are very high in the scientific and lay communities."
The studies are sponsored by Amgen and Cytokinetics. Dr. Teerlink has received research grants and/or consulting fees from those companies as well as a handful of others. Dr. McDonagh reported no financial conflicts.
AMSTERDAM – The novel intravenous inotropic agent omecamtiv mecarbil achieved greater reduction in dyspnea scores and less likelihood of worsening heart failure at the highest dose studied, compared with placebo, in patients with acute decompensated heart failure in a phase II, dose-ranging study.
The drug also demonstrated a strong dose-dependent increase in systolic ejection time, no evidence of a proarrhythmic effect, and a greater reduction in heart rate with a larger increase in blood pressure than in placebo-treated controls, Dr. John R. Teerlink reported at the annual congress of the European Society of Cardiology.
In the overall population on omecamtiv mecarbil, however, which included the patients in the low- and intermediate-dose arms of this dose-ranging study, the dyspnea response rate wasn’t better than with placebo. Hence, the drug’s future is uncertain.
In a press release following Dr. Teerlink’s presentation, Amgen, cosponsor of the phase II ATOMIC-AHF (Acute Treatment With Omecamtiv Mecarbil to Increase Contractility in Acute Heart Failure) trial, said that no decision has been made as to whether to move omecamtiv mecarbil into phase III clinical trials. Dr. Teerlink thinks the drug has earned a shot.
"I personally believe this drug has continued to demonstrate its promise all along the way. It could replace milrinone and dobutamine in the hospital to improve cardiac performance," predicted Dr. Teerlink, professor of medicine at the University of California, San Francisco, and director of the heart failure program at the San Francisco Veterans Affairs Medical Center.
Omecamtiv mecarbil is a novel selective cardiac myosin activator. It increases the entry rate of myosin into the force-producing state with actin. It boosts cardiac output through prolongation of the duration of systole and increased stroke volume, with no change in myocardial contractility and no increases in myocardial oxygen demand or myocyte calcium.
"It’s essentially producing more hands pulling on the rope," the cardiologist explained.
Discussant Theresa A. McDonagh said omecamtiv mecarbil has drawn great interest within the heart failure field because current inotropes are recognized as "very inadequate."
"It is truly a new inotrope, a drug that acts on the sarcomere itself. It does seem to be free of the nemesis of other inotropes: increased heart rate, greater myocardial oxygen consumption, arrhythmia production, and increased mortality in heart failure. It appears to be the Holy Grail, and may be capable of replacing dobutamine and milrinone," said Dr. McDonagh, professor of heart failure at King’s College London.
"I think this [ATOMIC-AHF trial] is a very promising start for omecamtiv mecarbil in acute heart failure," she added.
ATOMIC-AHF included 613 patients with a left ventricular ejection fraction of 40% or less who were hospitalized for acute heart failure with dyspnea at rest or with minimal exertion despite intravenous diuretic therapy. The study was conducted at 106 sites on three continents. Participants were randomized to 48 hours of intravenous placebo or omecamtiv mecarbil in three different ascending dose regimens.
The primary efficacy endpoint, dyspnea at 6, 24, and 48 hours across the three doses, compared with the pooled placebo group, was not met.
However, patients in the highest-dose omecamtiv mecarbil study arm had a 51% rate of significant improvement in dyspnea on a self-rated 7-point Likert scale at 48 hours, a significantly better response than the 37% rate in matched placebo-treated controls (P = .03). This comparison was a prespecified secondary endpoint in the study.
In addition, patients in the middle- and high-dose omecamtiv mecarbil arms had roughly a 45% reduction in the 7-day incidence of worsening heart failure, compared with the 17% rate in controls.
Not only was there no evidence of a proarrhythmic effect with omecamtiv mecarbil, the drug was associated with a 50% reduction in the incidence of supraventricular tachyarrhythmias, compared with placebo, he continued.
A modest increase in troponin I levels was noted in the omecamtiv mecarbil–treated group, but it was unrelated to plasma drug concentrations, which Dr. Teerlink found reassuring. Nevertheless, Dr. McDonagh said the small increases in troponin "should herald some caution amid the enthusiasm here."
The drug’s mechanism of action, which results in greater systolic ejection time, always raises the concern that it might also shorten diastole and compromise coronary perfusion. However, the observed reduction in heart rate makes that seem less likely, she added.
The ATOMIC-AHF study population was narrowly defined and of a profile commonly seen in clinical practice. They had an estimated glomerular filtration rate of about 50 mL/min, and they presented with dyspnea but didn’t have an acute coronary syndrome.
"This study population is an area of great unmet need in heart failure, with mortality rates of about 10% during their hospitalization and 30% at 1 year following discharge," according to Dr. McDonagh.
Nevertheless, it will be important in phase III testing to see how the drug performs in a broader group of heart failure patients, including those with lower estimated glomerular filtration rates, she added.
In an interview, Dr. Teerlink said that before a decision is reached regarding advancement of omecamtiv mecarbil to phase III studies, investigators and the study sponsors first want to see the data from an ongoing phase II dose-finding study of an oral formulation of the drug in chronic heart failure patients. This study, COSMIC-HF (Chronic Oral Study of Myosin Activation to Increase Contractility in Heart Failure), is still enrolling patients.
"We’ll get the combined data from ATOMIC-AHF and COSMIC-HF and then decide what phase III should look like. There are a lot of options here: IV [intravenous] alone, IV transitioning to chronic oral therapy, or even oral alone," he explained.
"One of the greatest challenges of the ATOMIC-AHF trial has been managing expectations," according to Dr. Teerlink. "Omecamtiv mecarbil is probably one of the most interesting new chemical entities in cardiovascular medicine now, and consequently expectations are very high in the scientific and lay communities."
The studies are sponsored by Amgen and Cytokinetics. Dr. Teerlink has received research grants and/or consulting fees from those companies as well as a handful of others. Dr. McDonagh reported no financial conflicts.
AMSTERDAM – The novel intravenous inotropic agent omecamtiv mecarbil achieved greater reduction in dyspnea scores and less likelihood of worsening heart failure at the highest dose studied, compared with placebo, in patients with acute decompensated heart failure in a phase II, dose-ranging study.
The drug also demonstrated a strong dose-dependent increase in systolic ejection time, no evidence of a proarrhythmic effect, and a greater reduction in heart rate with a larger increase in blood pressure than in placebo-treated controls, Dr. John R. Teerlink reported at the annual congress of the European Society of Cardiology.
In the overall population on omecamtiv mecarbil, however, which included the patients in the low- and intermediate-dose arms of this dose-ranging study, the dyspnea response rate wasn’t better than with placebo. Hence, the drug’s future is uncertain.
In a press release following Dr. Teerlink’s presentation, Amgen, cosponsor of the phase II ATOMIC-AHF (Acute Treatment With Omecamtiv Mecarbil to Increase Contractility in Acute Heart Failure) trial, said that no decision has been made as to whether to move omecamtiv mecarbil into phase III clinical trials. Dr. Teerlink thinks the drug has earned a shot.
"I personally believe this drug has continued to demonstrate its promise all along the way. It could replace milrinone and dobutamine in the hospital to improve cardiac performance," predicted Dr. Teerlink, professor of medicine at the University of California, San Francisco, and director of the heart failure program at the San Francisco Veterans Affairs Medical Center.
Omecamtiv mecarbil is a novel selective cardiac myosin activator. It increases the entry rate of myosin into the force-producing state with actin. It boosts cardiac output through prolongation of the duration of systole and increased stroke volume, with no change in myocardial contractility and no increases in myocardial oxygen demand or myocyte calcium.
"It’s essentially producing more hands pulling on the rope," the cardiologist explained.
Discussant Theresa A. McDonagh said omecamtiv mecarbil has drawn great interest within the heart failure field because current inotropes are recognized as "very inadequate."
"It is truly a new inotrope, a drug that acts on the sarcomere itself. It does seem to be free of the nemesis of other inotropes: increased heart rate, greater myocardial oxygen consumption, arrhythmia production, and increased mortality in heart failure. It appears to be the Holy Grail, and may be capable of replacing dobutamine and milrinone," said Dr. McDonagh, professor of heart failure at King’s College London.
"I think this [ATOMIC-AHF trial] is a very promising start for omecamtiv mecarbil in acute heart failure," she added.
ATOMIC-AHF included 613 patients with a left ventricular ejection fraction of 40% or less who were hospitalized for acute heart failure with dyspnea at rest or with minimal exertion despite intravenous diuretic therapy. The study was conducted at 106 sites on three continents. Participants were randomized to 48 hours of intravenous placebo or omecamtiv mecarbil in three different ascending dose regimens.
The primary efficacy endpoint, dyspnea at 6, 24, and 48 hours across the three doses, compared with the pooled placebo group, was not met.
However, patients in the highest-dose omecamtiv mecarbil study arm had a 51% rate of significant improvement in dyspnea on a self-rated 7-point Likert scale at 48 hours, a significantly better response than the 37% rate in matched placebo-treated controls (P = .03). This comparison was a prespecified secondary endpoint in the study.
In addition, patients in the middle- and high-dose omecamtiv mecarbil arms had roughly a 45% reduction in the 7-day incidence of worsening heart failure, compared with the 17% rate in controls.
Not only was there no evidence of a proarrhythmic effect with omecamtiv mecarbil, the drug was associated with a 50% reduction in the incidence of supraventricular tachyarrhythmias, compared with placebo, he continued.
A modest increase in troponin I levels was noted in the omecamtiv mecarbil–treated group, but it was unrelated to plasma drug concentrations, which Dr. Teerlink found reassuring. Nevertheless, Dr. McDonagh said the small increases in troponin "should herald some caution amid the enthusiasm here."
The drug’s mechanism of action, which results in greater systolic ejection time, always raises the concern that it might also shorten diastole and compromise coronary perfusion. However, the observed reduction in heart rate makes that seem less likely, she added.
The ATOMIC-AHF study population was narrowly defined and of a profile commonly seen in clinical practice. They had an estimated glomerular filtration rate of about 50 mL/min, and they presented with dyspnea but didn’t have an acute coronary syndrome.
"This study population is an area of great unmet need in heart failure, with mortality rates of about 10% during their hospitalization and 30% at 1 year following discharge," according to Dr. McDonagh.
Nevertheless, it will be important in phase III testing to see how the drug performs in a broader group of heart failure patients, including those with lower estimated glomerular filtration rates, she added.
In an interview, Dr. Teerlink said that before a decision is reached regarding advancement of omecamtiv mecarbil to phase III studies, investigators and the study sponsors first want to see the data from an ongoing phase II dose-finding study of an oral formulation of the drug in chronic heart failure patients. This study, COSMIC-HF (Chronic Oral Study of Myosin Activation to Increase Contractility in Heart Failure), is still enrolling patients.
"We’ll get the combined data from ATOMIC-AHF and COSMIC-HF and then decide what phase III should look like. There are a lot of options here: IV [intravenous] alone, IV transitioning to chronic oral therapy, or even oral alone," he explained.
"One of the greatest challenges of the ATOMIC-AHF trial has been managing expectations," according to Dr. Teerlink. "Omecamtiv mecarbil is probably one of the most interesting new chemical entities in cardiovascular medicine now, and consequently expectations are very high in the scientific and lay communities."
The studies are sponsored by Amgen and Cytokinetics. Dr. Teerlink has received research grants and/or consulting fees from those companies as well as a handful of others. Dr. McDonagh reported no financial conflicts.
AT THE ESC CONGRESS 2013
Major finding: Fifty-one percent of patients with acute heart failure assigned to the high-dose arm in a study of intravenous omecamtiv mecarbil showed significant improvement in dyspnea scores at 48 hours, compared with 37% of placebo-treated controls.
Data source: The ATOMIC-AHF trial was a 613-patient, phase II, randomized, placebo-controlled, double-blind, dose-finding study.
Disclosures: The study was sponsored by Amgen and Cytokinetics. The presenter has received research grants and consultant fees from the companies.
Five steps to fewer heart failure hospitalizations
ESTES PARK, COLO. – Introducing five evidence-based interventions in patients with heart failure with reduced ejection fraction would dramatically cut admissions for heart failure, according to Dr. JoAnn Lindenfeld, vice president of the Heart Failure Society of America.
Here are the five interventions: recognizing when to switch from furosemide to another oral loop diuretic with far better bioavailability; up-titrating beta-blocker therapy to the maximum recommended dose as quickly as possible; adding a low-dose aldosterone antagonist to the treatment regimen; prescribing digoxin in symptomatic patients with a low ejection fraction; and identifying and treating iron deficiency, Dr. Lindenfeld said at a conference on internal medicine sponsored by the University of Colorado.
• Loop diuretics: Furosemide, everybody’s favorite low-cost loop diuretic, turns out to have an enormously variable oral bioavailability, ranging from 10% to 90% from patient to patient. It also varies substantially from day to day within the same individual. In contrast, torsemide (Demadex) and bumetanide (Bumex) have a consistently high oral bioavailability of roughly 90%. They are useful alternatives in poorly compensated heart failure patients.
"When your patient says they’re not diuresing and you’re pretty sure they’re taking their drugs, or if they’ve had more than one recent admission for heart failure and they’re having trouble with congestion and fluid retention, think about switching to bumetanide or torsemide," said Dr. Lindenfeld, professor of medicine and medical director of the heart transplant program at the university.
"In my own practice, when I have a patient admitted for acute decompensated heart failure with congestion and I don’t find another reversible cause, I will usually switch them," noted Dr. Lindenfeld, who also is codirector of the university’s Center for Women’s Health Research.
In a classic study, 234 patients hospitalized for acute decompensated heart failure were randomized at discharge to torsemide or furosemide in equivalent doses. The torsemide group subsequently had a 52% lower rate of heart failure hospitalization (Am. J. Med. 2001;111:513-21).
Bumetanide is now a pretty inexpensive drug, Dr. Lindenfeld noted. In making the switch, remember that 40 mg of furosemide is equivalent to 1 mg of bumetanide or 20 mg of torsemide.
• Beta-blocker up-titration: Beta-blocker and angiotensin-converting enzyme* (ACE) inhibitor therapy both have a class IA recommendation in heart failure. But what’s the best way to juggle the timing of dual dose increases?
"None of the guidelines says how to manage up-titration, but I strongly believe that once you have somebody on a reasonable dose of an ACE inhibitor – say, 5 mg of lisinopril or the equivalent – then you should go to the beta-blocker and up-titrate it to its maximum," she said. "Then later, come back to the ACE inhibitor and get the patient on the maximum dose of that."
The rationale for this approach is based on a comparison of the outcomes of the landmark beta-blocker trials versus ATLAS, a 3,104-patient trial conducted in the pre-beta-blocker era in which patients were randomized to low-dose lisinopril at 2.5-5 mg/day or high-dose therapy at 32.5-35 mg/day to determine which was better. After 4 years of follow-up, the high-dose group showed a 24% reduction in the risk of heart failure hospitalizations, but no significant advantage in terms of all-cause mortality (Circulation 1999;100:2312-8).
Contrast those results with the outcomes of the major clinical trials for carvedilol, metoprolol, and bisoprolol, each of which featured up-titration to the target dose within 8 weeks whenever possible. All three studies were halted within less than a year because of a roughly 35% reduction in mortality, compared with placebo. And that mortality benefit became apparent at 3 months.
"These are huge reductions in mortality," Dr. Lindenfeld noted. "You don’t want to have a patient come back every 4 weeks to up-titrate their ACE inhibitor for 5 months and miss the opportunity to get the patient on an effective dose of a beta-blocker, when the lifesaving benefit begins so early."
The recommended maximum doses in heart failure patients are carvedilol (Coreg) at 25 mg twice daily, or 50 mg twice daily for patients weighing more than 85 kg; 200 mg/day for extended-release metoprolol (Toprol XL); and 10 mg/day for bisoprolol (Zebeta). The three beta-blockers are similar in their efficacy for treating heart failure, Dr. Lindenfeld said. However, bisoprolol has the fewest pulmonary effects and is thus the best choice in patients with chronic obstructive pulmonary disease (COPD), even though it lacks a specific Food and Drug Administration (FDA)-approved indication for heart failure, she continued.
• Aldosterone antagonists: In terms of mortality benefit, the randomized trial data show that the aldosterone antagonists are nearly as good as beta-blockers. Yet they remain widely underutilized in the United States, according to Dr. Lindenfeld.
Indeed, three major randomized trials showed roughly a 25% reduction in total mortality, compared with placebo, in patients on standard background therapy including a beta-blocker and ACE inhibitor, along with a 20% decrease in risk of sudden cardiac death. The doses used were spironolactone at 12.5-25 mg/day or eplerenone (Inspra) at 25-50 mg/day.
An intriguing retrospective analysis conducted in close to 7,000 patients with heart failure following an acute myocardial infarction concluded that getting the aldosterone antagonist onboard early in that situation is key. Patients who started on the drug less than 7 days post MI had a 29% reduction in total mortality and a 47% decrease in sudden cardiac death, compared with those started on day 7 or later (Eur. J. Heart Fail. 2009;11:1099-1105). That benefit is believed to be the result of early left ventricular remodeling.
A definitive European prospective, randomized trial looking at the impact of starting an aldosterone antagonist within 7 days after acute MI is due to be presented later this year. The inside word is the results are highly favorable, she noted.
Hyperkalemia is a legitimate concern when prescribing an aldosterone antagonist. These agents should be avoided in a patient who has a creatinine level above 2.5 mg/dL or an estimated glomerular filtration rate below 30 mL/min per 1.73 m2, or if other potassium-sparing drugs are onboard. Potassium levels should be checked after the first 3-7 days of therapy, again at 1 month, and then every 3 months, as well as anytime a patient becomes dehydrated.
• Digoxin: In the classic digoxin trial involving close to 7,000 patients, heart failure hospitalization was a prospectively defined endpoint. In patients with a left ventricular ejection fraction of 25%-45%, hospital admission for heart failure was reduced by 26% in patients assigned to digoxin. In those whose ejection fraction was less than 25%, the reduction in hospitalization was 39%.
"So don’t forget that digoxin is still a good drug in patients with low ejection fraction or who have substantial symptoms," Dr. Lindenfeld said. "If we had a drug approved today that didn’t change mortality but reduced hospital admissions by 39%, we’d all be giving it – and we’d be paying a lot for it."
• Role of iron deficiency in heart failure: A new European study is illuminating on this score: Among a cohort of 1,506 patients with chronic heart failure, fully 50% were determined to have iron deficiency, as defined by a ferritin level less than 100 mcg/L, or a ferritin level of 100-299 mcg/L with a transferring saturation lower than 20%. In a multivariate regression analysis, iron deficiency was a strong independent predictor for mortality, associated with a 42% increased risk (Am. Heart J. 2013;165:575-82).
"I think if you restricted the study to hospitalized heart failure patients, the iron deficiency rate would be even higher. It’s just appalling how many people we send home iron deficient without iron replacement therapy," Dr. Lindenfeld asserted.
She noted that in the European FAIR-HF trial involving 459 hospitalized iron-deficient heart failure patients randomized at discharge to intravenous iron corrective and maintenance therapy or to a matching placebo, the iron replacement group demonstrated significant improvement in quality of life and exercise capacity. The benefits were seen regardless of whether a patient’s baseline hemoglobin was high or low.
In addition, the rate of the combined endpoint of first hospitalization for worsening heart failure or death was 7.5% in the iron recipients, compared with 13.9% in placebo-treated controls – a difference that didn’t achieve statistical significance because the study was underpowered to evaluated that endpoint (N. Engl. J. Med. 2009;36:2436-48).
"Iron replacement is a distinct advantage for these patients, so you should be looking for iron deficiency. You probably don’t need to use IV iron, but if your patient is in the hospital anyway, IV iron is pretty benign and will get him iron-repleted almost immediately," Dr. Lindenfeld continued.
Before sending iron-deficient patients home on oral iron, make sure they can absorb it. Many older individuals can’t. Indeed, among patients hospitalized at the University of Colorado heart failure service, only 13% can actually absorb oral iron, she explained.
A simple way to tell is to draw a serum iron level, give the patient an iron tablet, and check the serum iron level again in 1-3 hours. It should roughly double, Dr. Lindenfeld explained.
Dr. Lindenfeld reported serving as a consultant for Medtronic, St. Jude, and other companies.
*Correction, 8/12/2013: An earlier version of this story misstated the meaning of ACE.
The five-point checklist as noted represents a reliable strategy for ensuring we apply evidence to our clinical practice. I would emphasize the following points:
•Pharmacodynamic issues such bioavailability of loop diuretics are often not well known, and the use of Bumex (bumetanide) can have a meaningful impact on those refractory to furosemide.

|
| Dr. Hiren Shah |
•Up-titration to maximum beta-blockade prior to doing the same with ACE inhibitors is strongly supported by current evidence but not always done in clinical practice.
•Early use of aldosterone antagonists post MI is critical, but in those with chronic HF, it should still be restricted to patients with New York Heart Association (NYHA) class II HF and left ventricular ejection fraction (LVEF) less than or equal to 30% or class III to IV HF and EF less than or equal to 35%.
In addition, aldosterone antagonist therapy should be continued following hospital discharge only in patients who can be carefully monitored for hyperkalemia. Similarly, one should be aware of the occurrence of adverse drug reactions when using digoxin, owing to its narrow therapeutic index requiring serum level monitoring.
•Finally, the most overlooked aspect of HF management on this list is the importance of correcting iron-deficiency anemia, which occurs mainly through chronic renal insufficiency in HF patients.
We also now know that the anemia itself can worsen cardiac function, both because it causes cardiac stress through tachycardia and increased stroke volume, and because it can cause a reduced renal blood flow and fluid retention, adding further stress to the heart. Therefore, it is critical to correct the iron deficiency to break the vicious circle wherein CHF causes anemia, and the anemia causes more CHF, and both damage the kidneys, worsening the anemia and the CHF further.
We should all aim for the preferred strategy of intravenous iron for our hospitalized patients and then convert to oral iron, noting the absorption issues that should be evaluated prior to discharge. Of course, any new iron-deficiency anemia should still be worked up considering other causes such as GI losses.
Dr. Hiren Shah is an assistant professor of medicine in the Feinberg School of Medicine, Northwestern University, Chicago, and a medical director of the Medicine and Cardiac Telemetry Hospitalist Unit at Northwestern Memorial Hospital in Chicago.
The five-point checklist as noted represents a reliable strategy for ensuring we apply evidence to our clinical practice. I would emphasize the following points:
•Pharmacodynamic issues such bioavailability of loop diuretics are often not well known, and the use of Bumex (bumetanide) can have a meaningful impact on those refractory to furosemide.

|
| Dr. Hiren Shah |
•Up-titration to maximum beta-blockade prior to doing the same with ACE inhibitors is strongly supported by current evidence but not always done in clinical practice.
•Early use of aldosterone antagonists post MI is critical, but in those with chronic HF, it should still be restricted to patients with New York Heart Association (NYHA) class II HF and left ventricular ejection fraction (LVEF) less than or equal to 30% or class III to IV HF and EF less than or equal to 35%.
In addition, aldosterone antagonist therapy should be continued following hospital discharge only in patients who can be carefully monitored for hyperkalemia. Similarly, one should be aware of the occurrence of adverse drug reactions when using digoxin, owing to its narrow therapeutic index requiring serum level monitoring.
•Finally, the most overlooked aspect of HF management on this list is the importance of correcting iron-deficiency anemia, which occurs mainly through chronic renal insufficiency in HF patients.
We also now know that the anemia itself can worsen cardiac function, both because it causes cardiac stress through tachycardia and increased stroke volume, and because it can cause a reduced renal blood flow and fluid retention, adding further stress to the heart. Therefore, it is critical to correct the iron deficiency to break the vicious circle wherein CHF causes anemia, and the anemia causes more CHF, and both damage the kidneys, worsening the anemia and the CHF further.
We should all aim for the preferred strategy of intravenous iron for our hospitalized patients and then convert to oral iron, noting the absorption issues that should be evaluated prior to discharge. Of course, any new iron-deficiency anemia should still be worked up considering other causes such as GI losses.
Dr. Hiren Shah is an assistant professor of medicine in the Feinberg School of Medicine, Northwestern University, Chicago, and a medical director of the Medicine and Cardiac Telemetry Hospitalist Unit at Northwestern Memorial Hospital in Chicago.
The five-point checklist as noted represents a reliable strategy for ensuring we apply evidence to our clinical practice. I would emphasize the following points:
•Pharmacodynamic issues such bioavailability of loop diuretics are often not well known, and the use of Bumex (bumetanide) can have a meaningful impact on those refractory to furosemide.

|
| Dr. Hiren Shah |
•Up-titration to maximum beta-blockade prior to doing the same with ACE inhibitors is strongly supported by current evidence but not always done in clinical practice.
•Early use of aldosterone antagonists post MI is critical, but in those with chronic HF, it should still be restricted to patients with New York Heart Association (NYHA) class II HF and left ventricular ejection fraction (LVEF) less than or equal to 30% or class III to IV HF and EF less than or equal to 35%.
In addition, aldosterone antagonist therapy should be continued following hospital discharge only in patients who can be carefully monitored for hyperkalemia. Similarly, one should be aware of the occurrence of adverse drug reactions when using digoxin, owing to its narrow therapeutic index requiring serum level monitoring.
•Finally, the most overlooked aspect of HF management on this list is the importance of correcting iron-deficiency anemia, which occurs mainly through chronic renal insufficiency in HF patients.
We also now know that the anemia itself can worsen cardiac function, both because it causes cardiac stress through tachycardia and increased stroke volume, and because it can cause a reduced renal blood flow and fluid retention, adding further stress to the heart. Therefore, it is critical to correct the iron deficiency to break the vicious circle wherein CHF causes anemia, and the anemia causes more CHF, and both damage the kidneys, worsening the anemia and the CHF further.
We should all aim for the preferred strategy of intravenous iron for our hospitalized patients and then convert to oral iron, noting the absorption issues that should be evaluated prior to discharge. Of course, any new iron-deficiency anemia should still be worked up considering other causes such as GI losses.
Dr. Hiren Shah is an assistant professor of medicine in the Feinberg School of Medicine, Northwestern University, Chicago, and a medical director of the Medicine and Cardiac Telemetry Hospitalist Unit at Northwestern Memorial Hospital in Chicago.
ESTES PARK, COLO. – Introducing five evidence-based interventions in patients with heart failure with reduced ejection fraction would dramatically cut admissions for heart failure, according to Dr. JoAnn Lindenfeld, vice president of the Heart Failure Society of America.
Here are the five interventions: recognizing when to switch from furosemide to another oral loop diuretic with far better bioavailability; up-titrating beta-blocker therapy to the maximum recommended dose as quickly as possible; adding a low-dose aldosterone antagonist to the treatment regimen; prescribing digoxin in symptomatic patients with a low ejection fraction; and identifying and treating iron deficiency, Dr. Lindenfeld said at a conference on internal medicine sponsored by the University of Colorado.
• Loop diuretics: Furosemide, everybody’s favorite low-cost loop diuretic, turns out to have an enormously variable oral bioavailability, ranging from 10% to 90% from patient to patient. It also varies substantially from day to day within the same individual. In contrast, torsemide (Demadex) and bumetanide (Bumex) have a consistently high oral bioavailability of roughly 90%. They are useful alternatives in poorly compensated heart failure patients.
"When your patient says they’re not diuresing and you’re pretty sure they’re taking their drugs, or if they’ve had more than one recent admission for heart failure and they’re having trouble with congestion and fluid retention, think about switching to bumetanide or torsemide," said Dr. Lindenfeld, professor of medicine and medical director of the heart transplant program at the university.
"In my own practice, when I have a patient admitted for acute decompensated heart failure with congestion and I don’t find another reversible cause, I will usually switch them," noted Dr. Lindenfeld, who also is codirector of the university’s Center for Women’s Health Research.
In a classic study, 234 patients hospitalized for acute decompensated heart failure were randomized at discharge to torsemide or furosemide in equivalent doses. The torsemide group subsequently had a 52% lower rate of heart failure hospitalization (Am. J. Med. 2001;111:513-21).
Bumetanide is now a pretty inexpensive drug, Dr. Lindenfeld noted. In making the switch, remember that 40 mg of furosemide is equivalent to 1 mg of bumetanide or 20 mg of torsemide.
• Beta-blocker up-titration: Beta-blocker and angiotensin-converting enzyme* (ACE) inhibitor therapy both have a class IA recommendation in heart failure. But what’s the best way to juggle the timing of dual dose increases?
"None of the guidelines says how to manage up-titration, but I strongly believe that once you have somebody on a reasonable dose of an ACE inhibitor – say, 5 mg of lisinopril or the equivalent – then you should go to the beta-blocker and up-titrate it to its maximum," she said. "Then later, come back to the ACE inhibitor and get the patient on the maximum dose of that."
The rationale for this approach is based on a comparison of the outcomes of the landmark beta-blocker trials versus ATLAS, a 3,104-patient trial conducted in the pre-beta-blocker era in which patients were randomized to low-dose lisinopril at 2.5-5 mg/day or high-dose therapy at 32.5-35 mg/day to determine which was better. After 4 years of follow-up, the high-dose group showed a 24% reduction in the risk of heart failure hospitalizations, but no significant advantage in terms of all-cause mortality (Circulation 1999;100:2312-8).
Contrast those results with the outcomes of the major clinical trials for carvedilol, metoprolol, and bisoprolol, each of which featured up-titration to the target dose within 8 weeks whenever possible. All three studies were halted within less than a year because of a roughly 35% reduction in mortality, compared with placebo. And that mortality benefit became apparent at 3 months.
"These are huge reductions in mortality," Dr. Lindenfeld noted. "You don’t want to have a patient come back every 4 weeks to up-titrate their ACE inhibitor for 5 months and miss the opportunity to get the patient on an effective dose of a beta-blocker, when the lifesaving benefit begins so early."
The recommended maximum doses in heart failure patients are carvedilol (Coreg) at 25 mg twice daily, or 50 mg twice daily for patients weighing more than 85 kg; 200 mg/day for extended-release metoprolol (Toprol XL); and 10 mg/day for bisoprolol (Zebeta). The three beta-blockers are similar in their efficacy for treating heart failure, Dr. Lindenfeld said. However, bisoprolol has the fewest pulmonary effects and is thus the best choice in patients with chronic obstructive pulmonary disease (COPD), even though it lacks a specific Food and Drug Administration (FDA)-approved indication for heart failure, she continued.
• Aldosterone antagonists: In terms of mortality benefit, the randomized trial data show that the aldosterone antagonists are nearly as good as beta-blockers. Yet they remain widely underutilized in the United States, according to Dr. Lindenfeld.
Indeed, three major randomized trials showed roughly a 25% reduction in total mortality, compared with placebo, in patients on standard background therapy including a beta-blocker and ACE inhibitor, along with a 20% decrease in risk of sudden cardiac death. The doses used were spironolactone at 12.5-25 mg/day or eplerenone (Inspra) at 25-50 mg/day.
An intriguing retrospective analysis conducted in close to 7,000 patients with heart failure following an acute myocardial infarction concluded that getting the aldosterone antagonist onboard early in that situation is key. Patients who started on the drug less than 7 days post MI had a 29% reduction in total mortality and a 47% decrease in sudden cardiac death, compared with those started on day 7 or later (Eur. J. Heart Fail. 2009;11:1099-1105). That benefit is believed to be the result of early left ventricular remodeling.
A definitive European prospective, randomized trial looking at the impact of starting an aldosterone antagonist within 7 days after acute MI is due to be presented later this year. The inside word is the results are highly favorable, she noted.
Hyperkalemia is a legitimate concern when prescribing an aldosterone antagonist. These agents should be avoided in a patient who has a creatinine level above 2.5 mg/dL or an estimated glomerular filtration rate below 30 mL/min per 1.73 m2, or if other potassium-sparing drugs are onboard. Potassium levels should be checked after the first 3-7 days of therapy, again at 1 month, and then every 3 months, as well as anytime a patient becomes dehydrated.
• Digoxin: In the classic digoxin trial involving close to 7,000 patients, heart failure hospitalization was a prospectively defined endpoint. In patients with a left ventricular ejection fraction of 25%-45%, hospital admission for heart failure was reduced by 26% in patients assigned to digoxin. In those whose ejection fraction was less than 25%, the reduction in hospitalization was 39%.
"So don’t forget that digoxin is still a good drug in patients with low ejection fraction or who have substantial symptoms," Dr. Lindenfeld said. "If we had a drug approved today that didn’t change mortality but reduced hospital admissions by 39%, we’d all be giving it – and we’d be paying a lot for it."
• Role of iron deficiency in heart failure: A new European study is illuminating on this score: Among a cohort of 1,506 patients with chronic heart failure, fully 50% were determined to have iron deficiency, as defined by a ferritin level less than 100 mcg/L, or a ferritin level of 100-299 mcg/L with a transferring saturation lower than 20%. In a multivariate regression analysis, iron deficiency was a strong independent predictor for mortality, associated with a 42% increased risk (Am. Heart J. 2013;165:575-82).
"I think if you restricted the study to hospitalized heart failure patients, the iron deficiency rate would be even higher. It’s just appalling how many people we send home iron deficient without iron replacement therapy," Dr. Lindenfeld asserted.
She noted that in the European FAIR-HF trial involving 459 hospitalized iron-deficient heart failure patients randomized at discharge to intravenous iron corrective and maintenance therapy or to a matching placebo, the iron replacement group demonstrated significant improvement in quality of life and exercise capacity. The benefits were seen regardless of whether a patient’s baseline hemoglobin was high or low.
In addition, the rate of the combined endpoint of first hospitalization for worsening heart failure or death was 7.5% in the iron recipients, compared with 13.9% in placebo-treated controls – a difference that didn’t achieve statistical significance because the study was underpowered to evaluated that endpoint (N. Engl. J. Med. 2009;36:2436-48).
"Iron replacement is a distinct advantage for these patients, so you should be looking for iron deficiency. You probably don’t need to use IV iron, but if your patient is in the hospital anyway, IV iron is pretty benign and will get him iron-repleted almost immediately," Dr. Lindenfeld continued.
Before sending iron-deficient patients home on oral iron, make sure they can absorb it. Many older individuals can’t. Indeed, among patients hospitalized at the University of Colorado heart failure service, only 13% can actually absorb oral iron, she explained.
A simple way to tell is to draw a serum iron level, give the patient an iron tablet, and check the serum iron level again in 1-3 hours. It should roughly double, Dr. Lindenfeld explained.
Dr. Lindenfeld reported serving as a consultant for Medtronic, St. Jude, and other companies.
*Correction, 8/12/2013: An earlier version of this story misstated the meaning of ACE.
ESTES PARK, COLO. – Introducing five evidence-based interventions in patients with heart failure with reduced ejection fraction would dramatically cut admissions for heart failure, according to Dr. JoAnn Lindenfeld, vice president of the Heart Failure Society of America.
Here are the five interventions: recognizing when to switch from furosemide to another oral loop diuretic with far better bioavailability; up-titrating beta-blocker therapy to the maximum recommended dose as quickly as possible; adding a low-dose aldosterone antagonist to the treatment regimen; prescribing digoxin in symptomatic patients with a low ejection fraction; and identifying and treating iron deficiency, Dr. Lindenfeld said at a conference on internal medicine sponsored by the University of Colorado.
• Loop diuretics: Furosemide, everybody’s favorite low-cost loop diuretic, turns out to have an enormously variable oral bioavailability, ranging from 10% to 90% from patient to patient. It also varies substantially from day to day within the same individual. In contrast, torsemide (Demadex) and bumetanide (Bumex) have a consistently high oral bioavailability of roughly 90%. They are useful alternatives in poorly compensated heart failure patients.
"When your patient says they’re not diuresing and you’re pretty sure they’re taking their drugs, or if they’ve had more than one recent admission for heart failure and they’re having trouble with congestion and fluid retention, think about switching to bumetanide or torsemide," said Dr. Lindenfeld, professor of medicine and medical director of the heart transplant program at the university.
"In my own practice, when I have a patient admitted for acute decompensated heart failure with congestion and I don’t find another reversible cause, I will usually switch them," noted Dr. Lindenfeld, who also is codirector of the university’s Center for Women’s Health Research.
In a classic study, 234 patients hospitalized for acute decompensated heart failure were randomized at discharge to torsemide or furosemide in equivalent doses. The torsemide group subsequently had a 52% lower rate of heart failure hospitalization (Am. J. Med. 2001;111:513-21).
Bumetanide is now a pretty inexpensive drug, Dr. Lindenfeld noted. In making the switch, remember that 40 mg of furosemide is equivalent to 1 mg of bumetanide or 20 mg of torsemide.
• Beta-blocker up-titration: Beta-blocker and angiotensin-converting enzyme* (ACE) inhibitor therapy both have a class IA recommendation in heart failure. But what’s the best way to juggle the timing of dual dose increases?
"None of the guidelines says how to manage up-titration, but I strongly believe that once you have somebody on a reasonable dose of an ACE inhibitor – say, 5 mg of lisinopril or the equivalent – then you should go to the beta-blocker and up-titrate it to its maximum," she said. "Then later, come back to the ACE inhibitor and get the patient on the maximum dose of that."
The rationale for this approach is based on a comparison of the outcomes of the landmark beta-blocker trials versus ATLAS, a 3,104-patient trial conducted in the pre-beta-blocker era in which patients were randomized to low-dose lisinopril at 2.5-5 mg/day or high-dose therapy at 32.5-35 mg/day to determine which was better. After 4 years of follow-up, the high-dose group showed a 24% reduction in the risk of heart failure hospitalizations, but no significant advantage in terms of all-cause mortality (Circulation 1999;100:2312-8).
Contrast those results with the outcomes of the major clinical trials for carvedilol, metoprolol, and bisoprolol, each of which featured up-titration to the target dose within 8 weeks whenever possible. All three studies were halted within less than a year because of a roughly 35% reduction in mortality, compared with placebo. And that mortality benefit became apparent at 3 months.
"These are huge reductions in mortality," Dr. Lindenfeld noted. "You don’t want to have a patient come back every 4 weeks to up-titrate their ACE inhibitor for 5 months and miss the opportunity to get the patient on an effective dose of a beta-blocker, when the lifesaving benefit begins so early."
The recommended maximum doses in heart failure patients are carvedilol (Coreg) at 25 mg twice daily, or 50 mg twice daily for patients weighing more than 85 kg; 200 mg/day for extended-release metoprolol (Toprol XL); and 10 mg/day for bisoprolol (Zebeta). The three beta-blockers are similar in their efficacy for treating heart failure, Dr. Lindenfeld said. However, bisoprolol has the fewest pulmonary effects and is thus the best choice in patients with chronic obstructive pulmonary disease (COPD), even though it lacks a specific Food and Drug Administration (FDA)-approved indication for heart failure, she continued.
• Aldosterone antagonists: In terms of mortality benefit, the randomized trial data show that the aldosterone antagonists are nearly as good as beta-blockers. Yet they remain widely underutilized in the United States, according to Dr. Lindenfeld.
Indeed, three major randomized trials showed roughly a 25% reduction in total mortality, compared with placebo, in patients on standard background therapy including a beta-blocker and ACE inhibitor, along with a 20% decrease in risk of sudden cardiac death. The doses used were spironolactone at 12.5-25 mg/day or eplerenone (Inspra) at 25-50 mg/day.
An intriguing retrospective analysis conducted in close to 7,000 patients with heart failure following an acute myocardial infarction concluded that getting the aldosterone antagonist onboard early in that situation is key. Patients who started on the drug less than 7 days post MI had a 29% reduction in total mortality and a 47% decrease in sudden cardiac death, compared with those started on day 7 or later (Eur. J. Heart Fail. 2009;11:1099-1105). That benefit is believed to be the result of early left ventricular remodeling.
A definitive European prospective, randomized trial looking at the impact of starting an aldosterone antagonist within 7 days after acute MI is due to be presented later this year. The inside word is the results are highly favorable, she noted.
Hyperkalemia is a legitimate concern when prescribing an aldosterone antagonist. These agents should be avoided in a patient who has a creatinine level above 2.5 mg/dL or an estimated glomerular filtration rate below 30 mL/min per 1.73 m2, or if other potassium-sparing drugs are onboard. Potassium levels should be checked after the first 3-7 days of therapy, again at 1 month, and then every 3 months, as well as anytime a patient becomes dehydrated.
• Digoxin: In the classic digoxin trial involving close to 7,000 patients, heart failure hospitalization was a prospectively defined endpoint. In patients with a left ventricular ejection fraction of 25%-45%, hospital admission for heart failure was reduced by 26% in patients assigned to digoxin. In those whose ejection fraction was less than 25%, the reduction in hospitalization was 39%.
"So don’t forget that digoxin is still a good drug in patients with low ejection fraction or who have substantial symptoms," Dr. Lindenfeld said. "If we had a drug approved today that didn’t change mortality but reduced hospital admissions by 39%, we’d all be giving it – and we’d be paying a lot for it."
• Role of iron deficiency in heart failure: A new European study is illuminating on this score: Among a cohort of 1,506 patients with chronic heart failure, fully 50% were determined to have iron deficiency, as defined by a ferritin level less than 100 mcg/L, or a ferritin level of 100-299 mcg/L with a transferring saturation lower than 20%. In a multivariate regression analysis, iron deficiency was a strong independent predictor for mortality, associated with a 42% increased risk (Am. Heart J. 2013;165:575-82).
"I think if you restricted the study to hospitalized heart failure patients, the iron deficiency rate would be even higher. It’s just appalling how many people we send home iron deficient without iron replacement therapy," Dr. Lindenfeld asserted.
She noted that in the European FAIR-HF trial involving 459 hospitalized iron-deficient heart failure patients randomized at discharge to intravenous iron corrective and maintenance therapy or to a matching placebo, the iron replacement group demonstrated significant improvement in quality of life and exercise capacity. The benefits were seen regardless of whether a patient’s baseline hemoglobin was high or low.
In addition, the rate of the combined endpoint of first hospitalization for worsening heart failure or death was 7.5% in the iron recipients, compared with 13.9% in placebo-treated controls – a difference that didn’t achieve statistical significance because the study was underpowered to evaluated that endpoint (N. Engl. J. Med. 2009;36:2436-48).
"Iron replacement is a distinct advantage for these patients, so you should be looking for iron deficiency. You probably don’t need to use IV iron, but if your patient is in the hospital anyway, IV iron is pretty benign and will get him iron-repleted almost immediately," Dr. Lindenfeld continued.
Before sending iron-deficient patients home on oral iron, make sure they can absorb it. Many older individuals can’t. Indeed, among patients hospitalized at the University of Colorado heart failure service, only 13% can actually absorb oral iron, she explained.
A simple way to tell is to draw a serum iron level, give the patient an iron tablet, and check the serum iron level again in 1-3 hours. It should roughly double, Dr. Lindenfeld explained.
Dr. Lindenfeld reported serving as a consultant for Medtronic, St. Jude, and other companies.
*Correction, 8/12/2013: An earlier version of this story misstated the meaning of ACE.
EXPERT ANALYSIS FROM THE ANNUAL INTERNAL MEDICINE PROGRAM
Guidelines issued on radiation-induced heart disease
Cancer patients undergoing radiation therapy need to have baseline studies of cardiac function and routine screening for heart disease, according to recommendations from the European Society of Cardiology and the American Society of Echocardiography published July 16 in the European Heart Journal–Cardiovascular Imaging.
The groups recommend baseline preradiation echocardiography along with a cardiac exam as well as screening for risk factors. An annual cardiac history and physical should be performed to check for new-onset heart problems.
Within 10 years of treatment, 10%-30% of patients who undergo radiation therapy develop radiation-induced heart diseases (RIHD), including chronic pericarditis, myocardial fibrosis, coronary artery disease, aortic calcification, and valve regurgitation or stenosis. The hope of screening is to catch early RIHD, but screening is not currently routine.
"We wrote the expert consensus to raise the alarm that the risks of radiation-induced heart disease should not be ignored. The prevalence ... is increasing because the rate of cancer survival has improved," said Dr. Patrizio Lancellotti, who is a professor of cardiology at the University Hospital of Liège, Belgium, and led the recommendations task force.
Radiotherapy is given in more targeted form and at lower doses than it once was, but "patients are still at increased risk of RIHD, particularly when the heart is in the radiation field. This applies to patients treated for lymphoma, breast cancer, and esophageal cancer. Patients who receive radiotherapy for neck cancer are also at risk because lesions can develop on the carotid artery and increase the risk of stroke," Dr. Lancellotti said in a statement.
Using targeted radiation and alternate radiation fields, with avoidance and shielding of the heart, remain "the most important interventions to prevent" cardiac complications, the authors noted.
The task force advises that high-risk patients without evidence of heart disease on history and physical should have screening echocardiography every 5 years and noninvasive stress testing every 5-10 years; low-risk patients should have screening echocardiography every 10 years. If heart disorders are detected, routine monitoring should include echocardiography, cardiac magnetic resonance imaging, or carotid ultrasound as appropriate.
High-risk patients include those who received radiotherapy at younger ages; those who have cardiovascular risk factors or preexisting heart disease; and those who receive high-dose radiation (greater than 30 Gy), concomitant chemotherapy, radiation without shielding, or anterior or left chest radiation (Eur. Heart J. Cardiovasc. Imaging 2013;14:721-40).
The recommendations are based on an extensive literature review and analysis by Dr. Lancellotti and other specialists.
The authors reported no financial conflicts or outside funding for their work.
Cancer patients undergoing radiation therapy need to have baseline studies of cardiac function and routine screening for heart disease, according to recommendations from the European Society of Cardiology and the American Society of Echocardiography published July 16 in the European Heart Journal–Cardiovascular Imaging.
The groups recommend baseline preradiation echocardiography along with a cardiac exam as well as screening for risk factors. An annual cardiac history and physical should be performed to check for new-onset heart problems.
Within 10 years of treatment, 10%-30% of patients who undergo radiation therapy develop radiation-induced heart diseases (RIHD), including chronic pericarditis, myocardial fibrosis, coronary artery disease, aortic calcification, and valve regurgitation or stenosis. The hope of screening is to catch early RIHD, but screening is not currently routine.
"We wrote the expert consensus to raise the alarm that the risks of radiation-induced heart disease should not be ignored. The prevalence ... is increasing because the rate of cancer survival has improved," said Dr. Patrizio Lancellotti, who is a professor of cardiology at the University Hospital of Liège, Belgium, and led the recommendations task force.
Radiotherapy is given in more targeted form and at lower doses than it once was, but "patients are still at increased risk of RIHD, particularly when the heart is in the radiation field. This applies to patients treated for lymphoma, breast cancer, and esophageal cancer. Patients who receive radiotherapy for neck cancer are also at risk because lesions can develop on the carotid artery and increase the risk of stroke," Dr. Lancellotti said in a statement.
Using targeted radiation and alternate radiation fields, with avoidance and shielding of the heart, remain "the most important interventions to prevent" cardiac complications, the authors noted.
The task force advises that high-risk patients without evidence of heart disease on history and physical should have screening echocardiography every 5 years and noninvasive stress testing every 5-10 years; low-risk patients should have screening echocardiography every 10 years. If heart disorders are detected, routine monitoring should include echocardiography, cardiac magnetic resonance imaging, or carotid ultrasound as appropriate.
High-risk patients include those who received radiotherapy at younger ages; those who have cardiovascular risk factors or preexisting heart disease; and those who receive high-dose radiation (greater than 30 Gy), concomitant chemotherapy, radiation without shielding, or anterior or left chest radiation (Eur. Heart J. Cardiovasc. Imaging 2013;14:721-40).
The recommendations are based on an extensive literature review and analysis by Dr. Lancellotti and other specialists.
The authors reported no financial conflicts or outside funding for their work.
Cancer patients undergoing radiation therapy need to have baseline studies of cardiac function and routine screening for heart disease, according to recommendations from the European Society of Cardiology and the American Society of Echocardiography published July 16 in the European Heart Journal–Cardiovascular Imaging.
The groups recommend baseline preradiation echocardiography along with a cardiac exam as well as screening for risk factors. An annual cardiac history and physical should be performed to check for new-onset heart problems.
Within 10 years of treatment, 10%-30% of patients who undergo radiation therapy develop radiation-induced heart diseases (RIHD), including chronic pericarditis, myocardial fibrosis, coronary artery disease, aortic calcification, and valve regurgitation or stenosis. The hope of screening is to catch early RIHD, but screening is not currently routine.
"We wrote the expert consensus to raise the alarm that the risks of radiation-induced heart disease should not be ignored. The prevalence ... is increasing because the rate of cancer survival has improved," said Dr. Patrizio Lancellotti, who is a professor of cardiology at the University Hospital of Liège, Belgium, and led the recommendations task force.
Radiotherapy is given in more targeted form and at lower doses than it once was, but "patients are still at increased risk of RIHD, particularly when the heart is in the radiation field. This applies to patients treated for lymphoma, breast cancer, and esophageal cancer. Patients who receive radiotherapy for neck cancer are also at risk because lesions can develop on the carotid artery and increase the risk of stroke," Dr. Lancellotti said in a statement.
Using targeted radiation and alternate radiation fields, with avoidance and shielding of the heart, remain "the most important interventions to prevent" cardiac complications, the authors noted.
The task force advises that high-risk patients without evidence of heart disease on history and physical should have screening echocardiography every 5 years and noninvasive stress testing every 5-10 years; low-risk patients should have screening echocardiography every 10 years. If heart disorders are detected, routine monitoring should include echocardiography, cardiac magnetic resonance imaging, or carotid ultrasound as appropriate.
High-risk patients include those who received radiotherapy at younger ages; those who have cardiovascular risk factors or preexisting heart disease; and those who receive high-dose radiation (greater than 30 Gy), concomitant chemotherapy, radiation without shielding, or anterior or left chest radiation (Eur. Heart J. Cardiovasc. Imaging 2013;14:721-40).
The recommendations are based on an extensive literature review and analysis by Dr. Lancellotti and other specialists.
The authors reported no financial conflicts or outside funding for their work.
FROM THE EUROPEAN HEART JOURNAL – CARDIOVASCULAR IMAGING
Novel drug improved walk distance in pulmonary hypertension patients
Riociguat, an agent from a new class of compounds known as soluble guanylate cyclase stimulators, was found effective and safe for treating two different types of pulmonary hypertension in separate industry-sponsored phase III clinical trials reported online July 25 in the New England Journal of Medicine.
Compared with placebo, daily oral riociguat significantly improved exercise capacity as measured by 6-minute walk distance, and also improved pulmonary vascular resistance and World Health Organization (WHO) functional class. The results are from one international study involving 261 patients who had chronic inoperable thromboembolic pulmonary hypertension and another study involving 443 patients who had pulmonary arterial hypertension.
The magnitude of the improvement was greater than that reported for existing medications such as endothelin-receptor antagonists and prostanoids, both groups of researchers said.
The pathogenesis of pulmonary hypertension involves impairment of both nitric oxide synthesis and signaling through the nitric oxide–soluble guanylate cyclase–cyclic guanosine monophosphate pathway. Riociguat directly stimulates soluble guanylate cyclase independently of nitric oxide and also increases the enzyme’s sensitivity to nitric oxide. The drug also raises levels of cyclic guanosine monophosphate, which induces vasorelaxation and has additional antiproliferative and antifibrotic effects.
The CHEST-1 trial
In the CHEST-1 (Chronic Thromboembolic Pulmonary Hypertension Soluble Guanylate Cyclase-Stimulator) trial, funded by Bayer HealthCare, the drug was assessed in adults with chronic thromboembolic pulmonary hypertension who were either ineligible for the standard surgical treatment (pulmonary endarterectomy) or whose condition persisted or recurred after they underwent the surgery. It is estimated that 63% of patients with this disorder are ineligible for endarterectomy, which is the only potentially curative treatment available, and that thromboembolic pulmonary hypertension persists or recurs in 35% of patients who do have the procedure. So an alternative approach is clearly needed, said Dr. Hossein-Ardeschir Ghofrani of University Hospital Giessen and Marburg, Germany, and his associates.
The CHEST-1 study subjects, who were treated and followed at 89 medical centers in 26 countries, were randomly assigned to receive either riociguat (173 patients) or matching placebo (88 patients) for 16 weeks.
The primary endpoint was change in 6-minute walk distance. In the intention-to-treat analysis, this increased by a mean of 39 m in patients taking riociguat, compared with a decrease of 6 m in those taking placebo. The benefit was similar in a per-protocol analysis, the investigators reported (N. Engl. J. Med. 2013 July 25 [doi: 10.1056/NEJMoa1209657]).
Riociguat improved 6-minute walk distance by 54 m in the patients who were ineligible for surgery and by 26 m in those who had persistent or recurrent disease after surgery. The drug’s beneficial effect on exercise capacity was consistent across several subgroups of patients.
In addition, the active drug significantly reduced pulmonary vascular resistance; improved other hemodynamic factors, including mean pulmonary artery pressure and cardiac output; improved WHO functional class, which is known to correlate with survival; and decreased levels of N-terminal pro-brain natriuretic peptide (NT-proBNP).
In exploratory analyses, riociguat also improved scores on the Borg dyspnea index when compared with placebo, and nominally improved scores on one measure of quality of life but not on a disease-specific QOL tool.
Drug-related serious adverse effects included three cases of syncope and one case each of gastritis, acute renal failure, and hypotension with riociguat, and one case each of syncope and trauma with placebo. Three percent of the riociguat group and 2% of the placebo group dropped out of the study owing to adverse events, and another 2% of each group dropped out because of serious adverse events that were not considered to be related to the study drug.
There were two deaths in the riociguat group and three in the placebo group.
A total of 237 of these study subjects elected to enroll in an extended study of the long-term safety and efficacy of riociguat (the CHEST-2 clinical trial). An exploratory analysis of data from the first 12 weeks of that study showed further increases of 51-63 m in the 6-minute walk distance, Dr. Ghofrani and his associates said.
The PATENT-1 trial
The other phase III clinical trial reported in the New England Journal of Medicine was the PATENT-1 (Pulmonary Arterial Hypertension Soluble Guanylate Cyclase-Stimulator) study. This trial also was funded by Bayer HealthCare and headed by Dr. Ghofrani.
In it, 443 patients who had symptomatic pulmonary arterial hypertension were treated and followed up at 124 medical centers in 30 countries. These subjects had idiopathic or familial disease, or pulmonary arterial hypertension associated with connective-tissue disease, congenital heart disease, portal hypertension with liver cirrhosis, anorexigen use, or amphetamine use.
A total of 44% of patients were already receiving an endothelin-receptor antagonist (usually bosentan), and 6% were taking prostanoids (usually inhaled iloprost) for the disorder, and the other half of the study population was not receiving any other treatments. Concomitant therapy with oral anticoagulants, diuretics, and supplemental oxygen was permitted during the study.
These study subjects were randomly assigned to receive high-dose riociguat capped at 2.5 mg three times daily (254 patients), low-dose riociguat capped at 1.5 mg three times daily (63 patients), or placebo (126 patients) for 12 weeks. The analysis of data from the low-dose group was considered exploratory and was reported separately.
The primary endpoint – change from baseline in 6-minute walk distance – increased by a mean of 30 m in the high-dose group but decreased by a mean of 6 m in the placebo group in the intention-to-treat analysis. The results were similar in the per-protocol analysis, the investigators reported (N. Engl. J. Med. 2013 July 25 [doi: 10.1056/NEJMoa1209655]).
This treatment benefit was consistent across several subgroups of patients. In addition, the active drug significantly decreased pulmonary vascular resistance, improved mean pulmonary artery pressure and cardiac output, lengthened the interval to clinical worsening, improved NT-proBNP levels, improved WHO functional class, and improved scores on the Borg dyspnea scale.
In exploratory analyses, riociguat did not improve scores on one measure of QOL but did so on a disease-specific QOL tool.
Drug-related serious adverse events occurred in 1.4 % of patients receiving high-dose riociguat and 1% of those receiving placebo. A total of 3% of patients in the riociguat group and 7% of those in the placebo group discontinued the study drug because of adverse events.
Serious or drug-related adverse events in both groups included increased hepatic enzyme levels, acute renal failure, syncope, esophageal pain and swelling, supraventricular tachycardia, hypotension, generalized edema, hypoxemia, dyspnea, and worsening of pulmonary hypertension. There were two deaths in the riociguat group and three in the placebo group, none of which were considered to be related to the study drug.
A total of 396 of the subjects in the PATENT-1 study elected to enroll in an extended study of the long-term safety and efficacy of riociguat (the PATENT-2 clinical trial). An exploratory analysis of data from the first 12 weeks of that study showed a further mean increase of 53 m in 6-minute walk distance with high-dose riociguat.
CHEST-1 and PATENT-1 were funded by Bayer HealthCare. Dr. Ghofrani reported ties to Actelion, Bayer, and other companies. His associates reported ties to numerous industry sources.
Riociguat "is poised for examination by the Food and Drug Administration as a therapy for pulmonary hypertension and, if approved, has the potential to generate substantial revenue" for the sponsor of these two clinical trials, said Dr. Stephen L. Archer.
Both CHEST-1 and PATENT-1 were sponsored by Bayer HealthCare, which also provided the statistician and editorial assistance for both trials. "In light of the financial stakes, both real and apparent investigator autonomy remain key to ensuring the delivery of new drugs for pulmonary hypertension for patients," he noted.
Although riociguat yielded only "modest" gains in exercise capacity, it looks promising and may prove to be the first effective oral therapy for inoperable Group 4 pulmonary hypertension, Dr. Archer added.
However, he noted limitations in both studies. The CHEST-1 trial was limited by its "failure to examine the effects of the study drug on the right ventricle. The 6-minute walk distance is as reflective of right ventricular or skeletal-muscle function as it is of reduction in pulmonary vascular resistance, the authors’ presumed mechanism of benefit."
PATENT-1 was limited by its modest effect size, he added. "This is particularly important, since 50% of the patients were receiving no other treatment for pulmonary arterial hypertension, and the rate of response to treatment among such patients is usually higher than the rate among patients who are receiving concomitant treatment for pulmonary arterial hypertension," Dr. Archer said.
Dr. Archer is in the department of medicine at Queen’s University, Kingston, Ont. He reported no financial conflicts of interest. These remarks were taken from his editorial accompanying Dr. Ghofrani’s reports (N. Engl. J. Med. 2013 July 25 [doi: 10.1056/NEJMe1306684]).
Riociguat "is poised for examination by the Food and Drug Administration as a therapy for pulmonary hypertension and, if approved, has the potential to generate substantial revenue" for the sponsor of these two clinical trials, said Dr. Stephen L. Archer.
Both CHEST-1 and PATENT-1 were sponsored by Bayer HealthCare, which also provided the statistician and editorial assistance for both trials. "In light of the financial stakes, both real and apparent investigator autonomy remain key to ensuring the delivery of new drugs for pulmonary hypertension for patients," he noted.
Although riociguat yielded only "modest" gains in exercise capacity, it looks promising and may prove to be the first effective oral therapy for inoperable Group 4 pulmonary hypertension, Dr. Archer added.
However, he noted limitations in both studies. The CHEST-1 trial was limited by its "failure to examine the effects of the study drug on the right ventricle. The 6-minute walk distance is as reflective of right ventricular or skeletal-muscle function as it is of reduction in pulmonary vascular resistance, the authors’ presumed mechanism of benefit."
PATENT-1 was limited by its modest effect size, he added. "This is particularly important, since 50% of the patients were receiving no other treatment for pulmonary arterial hypertension, and the rate of response to treatment among such patients is usually higher than the rate among patients who are receiving concomitant treatment for pulmonary arterial hypertension," Dr. Archer said.
Dr. Archer is in the department of medicine at Queen’s University, Kingston, Ont. He reported no financial conflicts of interest. These remarks were taken from his editorial accompanying Dr. Ghofrani’s reports (N. Engl. J. Med. 2013 July 25 [doi: 10.1056/NEJMe1306684]).
Riociguat "is poised for examination by the Food and Drug Administration as a therapy for pulmonary hypertension and, if approved, has the potential to generate substantial revenue" for the sponsor of these two clinical trials, said Dr. Stephen L. Archer.
Both CHEST-1 and PATENT-1 were sponsored by Bayer HealthCare, which also provided the statistician and editorial assistance for both trials. "In light of the financial stakes, both real and apparent investigator autonomy remain key to ensuring the delivery of new drugs for pulmonary hypertension for patients," he noted.
Although riociguat yielded only "modest" gains in exercise capacity, it looks promising and may prove to be the first effective oral therapy for inoperable Group 4 pulmonary hypertension, Dr. Archer added.
However, he noted limitations in both studies. The CHEST-1 trial was limited by its "failure to examine the effects of the study drug on the right ventricle. The 6-minute walk distance is as reflective of right ventricular or skeletal-muscle function as it is of reduction in pulmonary vascular resistance, the authors’ presumed mechanism of benefit."
PATENT-1 was limited by its modest effect size, he added. "This is particularly important, since 50% of the patients were receiving no other treatment for pulmonary arterial hypertension, and the rate of response to treatment among such patients is usually higher than the rate among patients who are receiving concomitant treatment for pulmonary arterial hypertension," Dr. Archer said.
Dr. Archer is in the department of medicine at Queen’s University, Kingston, Ont. He reported no financial conflicts of interest. These remarks were taken from his editorial accompanying Dr. Ghofrani’s reports (N. Engl. J. Med. 2013 July 25 [doi: 10.1056/NEJMe1306684]).
Riociguat, an agent from a new class of compounds known as soluble guanylate cyclase stimulators, was found effective and safe for treating two different types of pulmonary hypertension in separate industry-sponsored phase III clinical trials reported online July 25 in the New England Journal of Medicine.
Compared with placebo, daily oral riociguat significantly improved exercise capacity as measured by 6-minute walk distance, and also improved pulmonary vascular resistance and World Health Organization (WHO) functional class. The results are from one international study involving 261 patients who had chronic inoperable thromboembolic pulmonary hypertension and another study involving 443 patients who had pulmonary arterial hypertension.
The magnitude of the improvement was greater than that reported for existing medications such as endothelin-receptor antagonists and prostanoids, both groups of researchers said.
The pathogenesis of pulmonary hypertension involves impairment of both nitric oxide synthesis and signaling through the nitric oxide–soluble guanylate cyclase–cyclic guanosine monophosphate pathway. Riociguat directly stimulates soluble guanylate cyclase independently of nitric oxide and also increases the enzyme’s sensitivity to nitric oxide. The drug also raises levels of cyclic guanosine monophosphate, which induces vasorelaxation and has additional antiproliferative and antifibrotic effects.
The CHEST-1 trial
In the CHEST-1 (Chronic Thromboembolic Pulmonary Hypertension Soluble Guanylate Cyclase-Stimulator) trial, funded by Bayer HealthCare, the drug was assessed in adults with chronic thromboembolic pulmonary hypertension who were either ineligible for the standard surgical treatment (pulmonary endarterectomy) or whose condition persisted or recurred after they underwent the surgery. It is estimated that 63% of patients with this disorder are ineligible for endarterectomy, which is the only potentially curative treatment available, and that thromboembolic pulmonary hypertension persists or recurs in 35% of patients who do have the procedure. So an alternative approach is clearly needed, said Dr. Hossein-Ardeschir Ghofrani of University Hospital Giessen and Marburg, Germany, and his associates.
The CHEST-1 study subjects, who were treated and followed at 89 medical centers in 26 countries, were randomly assigned to receive either riociguat (173 patients) or matching placebo (88 patients) for 16 weeks.
The primary endpoint was change in 6-minute walk distance. In the intention-to-treat analysis, this increased by a mean of 39 m in patients taking riociguat, compared with a decrease of 6 m in those taking placebo. The benefit was similar in a per-protocol analysis, the investigators reported (N. Engl. J. Med. 2013 July 25 [doi: 10.1056/NEJMoa1209657]).
Riociguat improved 6-minute walk distance by 54 m in the patients who were ineligible for surgery and by 26 m in those who had persistent or recurrent disease after surgery. The drug’s beneficial effect on exercise capacity was consistent across several subgroups of patients.
In addition, the active drug significantly reduced pulmonary vascular resistance; improved other hemodynamic factors, including mean pulmonary artery pressure and cardiac output; improved WHO functional class, which is known to correlate with survival; and decreased levels of N-terminal pro-brain natriuretic peptide (NT-proBNP).
In exploratory analyses, riociguat also improved scores on the Borg dyspnea index when compared with placebo, and nominally improved scores on one measure of quality of life but not on a disease-specific QOL tool.
Drug-related serious adverse effects included three cases of syncope and one case each of gastritis, acute renal failure, and hypotension with riociguat, and one case each of syncope and trauma with placebo. Three percent of the riociguat group and 2% of the placebo group dropped out of the study owing to adverse events, and another 2% of each group dropped out because of serious adverse events that were not considered to be related to the study drug.
There were two deaths in the riociguat group and three in the placebo group.
A total of 237 of these study subjects elected to enroll in an extended study of the long-term safety and efficacy of riociguat (the CHEST-2 clinical trial). An exploratory analysis of data from the first 12 weeks of that study showed further increases of 51-63 m in the 6-minute walk distance, Dr. Ghofrani and his associates said.
The PATENT-1 trial
The other phase III clinical trial reported in the New England Journal of Medicine was the PATENT-1 (Pulmonary Arterial Hypertension Soluble Guanylate Cyclase-Stimulator) study. This trial also was funded by Bayer HealthCare and headed by Dr. Ghofrani.
In it, 443 patients who had symptomatic pulmonary arterial hypertension were treated and followed up at 124 medical centers in 30 countries. These subjects had idiopathic or familial disease, or pulmonary arterial hypertension associated with connective-tissue disease, congenital heart disease, portal hypertension with liver cirrhosis, anorexigen use, or amphetamine use.
A total of 44% of patients were already receiving an endothelin-receptor antagonist (usually bosentan), and 6% were taking prostanoids (usually inhaled iloprost) for the disorder, and the other half of the study population was not receiving any other treatments. Concomitant therapy with oral anticoagulants, diuretics, and supplemental oxygen was permitted during the study.
These study subjects were randomly assigned to receive high-dose riociguat capped at 2.5 mg three times daily (254 patients), low-dose riociguat capped at 1.5 mg three times daily (63 patients), or placebo (126 patients) for 12 weeks. The analysis of data from the low-dose group was considered exploratory and was reported separately.
The primary endpoint – change from baseline in 6-minute walk distance – increased by a mean of 30 m in the high-dose group but decreased by a mean of 6 m in the placebo group in the intention-to-treat analysis. The results were similar in the per-protocol analysis, the investigators reported (N. Engl. J. Med. 2013 July 25 [doi: 10.1056/NEJMoa1209655]).
This treatment benefit was consistent across several subgroups of patients. In addition, the active drug significantly decreased pulmonary vascular resistance, improved mean pulmonary artery pressure and cardiac output, lengthened the interval to clinical worsening, improved NT-proBNP levels, improved WHO functional class, and improved scores on the Borg dyspnea scale.
In exploratory analyses, riociguat did not improve scores on one measure of QOL but did so on a disease-specific QOL tool.
Drug-related serious adverse events occurred in 1.4 % of patients receiving high-dose riociguat and 1% of those receiving placebo. A total of 3% of patients in the riociguat group and 7% of those in the placebo group discontinued the study drug because of adverse events.
Serious or drug-related adverse events in both groups included increased hepatic enzyme levels, acute renal failure, syncope, esophageal pain and swelling, supraventricular tachycardia, hypotension, generalized edema, hypoxemia, dyspnea, and worsening of pulmonary hypertension. There were two deaths in the riociguat group and three in the placebo group, none of which were considered to be related to the study drug.
A total of 396 of the subjects in the PATENT-1 study elected to enroll in an extended study of the long-term safety and efficacy of riociguat (the PATENT-2 clinical trial). An exploratory analysis of data from the first 12 weeks of that study showed a further mean increase of 53 m in 6-minute walk distance with high-dose riociguat.
CHEST-1 and PATENT-1 were funded by Bayer HealthCare. Dr. Ghofrani reported ties to Actelion, Bayer, and other companies. His associates reported ties to numerous industry sources.
Riociguat, an agent from a new class of compounds known as soluble guanylate cyclase stimulators, was found effective and safe for treating two different types of pulmonary hypertension in separate industry-sponsored phase III clinical trials reported online July 25 in the New England Journal of Medicine.
Compared with placebo, daily oral riociguat significantly improved exercise capacity as measured by 6-minute walk distance, and also improved pulmonary vascular resistance and World Health Organization (WHO) functional class. The results are from one international study involving 261 patients who had chronic inoperable thromboembolic pulmonary hypertension and another study involving 443 patients who had pulmonary arterial hypertension.
The magnitude of the improvement was greater than that reported for existing medications such as endothelin-receptor antagonists and prostanoids, both groups of researchers said.
The pathogenesis of pulmonary hypertension involves impairment of both nitric oxide synthesis and signaling through the nitric oxide–soluble guanylate cyclase–cyclic guanosine monophosphate pathway. Riociguat directly stimulates soluble guanylate cyclase independently of nitric oxide and also increases the enzyme’s sensitivity to nitric oxide. The drug also raises levels of cyclic guanosine monophosphate, which induces vasorelaxation and has additional antiproliferative and antifibrotic effects.
The CHEST-1 trial
In the CHEST-1 (Chronic Thromboembolic Pulmonary Hypertension Soluble Guanylate Cyclase-Stimulator) trial, funded by Bayer HealthCare, the drug was assessed in adults with chronic thromboembolic pulmonary hypertension who were either ineligible for the standard surgical treatment (pulmonary endarterectomy) or whose condition persisted or recurred after they underwent the surgery. It is estimated that 63% of patients with this disorder are ineligible for endarterectomy, which is the only potentially curative treatment available, and that thromboembolic pulmonary hypertension persists or recurs in 35% of patients who do have the procedure. So an alternative approach is clearly needed, said Dr. Hossein-Ardeschir Ghofrani of University Hospital Giessen and Marburg, Germany, and his associates.
The CHEST-1 study subjects, who were treated and followed at 89 medical centers in 26 countries, were randomly assigned to receive either riociguat (173 patients) or matching placebo (88 patients) for 16 weeks.
The primary endpoint was change in 6-minute walk distance. In the intention-to-treat analysis, this increased by a mean of 39 m in patients taking riociguat, compared with a decrease of 6 m in those taking placebo. The benefit was similar in a per-protocol analysis, the investigators reported (N. Engl. J. Med. 2013 July 25 [doi: 10.1056/NEJMoa1209657]).
Riociguat improved 6-minute walk distance by 54 m in the patients who were ineligible for surgery and by 26 m in those who had persistent or recurrent disease after surgery. The drug’s beneficial effect on exercise capacity was consistent across several subgroups of patients.
In addition, the active drug significantly reduced pulmonary vascular resistance; improved other hemodynamic factors, including mean pulmonary artery pressure and cardiac output; improved WHO functional class, which is known to correlate with survival; and decreased levels of N-terminal pro-brain natriuretic peptide (NT-proBNP).
In exploratory analyses, riociguat also improved scores on the Borg dyspnea index when compared with placebo, and nominally improved scores on one measure of quality of life but not on a disease-specific QOL tool.
Drug-related serious adverse effects included three cases of syncope and one case each of gastritis, acute renal failure, and hypotension with riociguat, and one case each of syncope and trauma with placebo. Three percent of the riociguat group and 2% of the placebo group dropped out of the study owing to adverse events, and another 2% of each group dropped out because of serious adverse events that were not considered to be related to the study drug.
There were two deaths in the riociguat group and three in the placebo group.
A total of 237 of these study subjects elected to enroll in an extended study of the long-term safety and efficacy of riociguat (the CHEST-2 clinical trial). An exploratory analysis of data from the first 12 weeks of that study showed further increases of 51-63 m in the 6-minute walk distance, Dr. Ghofrani and his associates said.
The PATENT-1 trial
The other phase III clinical trial reported in the New England Journal of Medicine was the PATENT-1 (Pulmonary Arterial Hypertension Soluble Guanylate Cyclase-Stimulator) study. This trial also was funded by Bayer HealthCare and headed by Dr. Ghofrani.
In it, 443 patients who had symptomatic pulmonary arterial hypertension were treated and followed up at 124 medical centers in 30 countries. These subjects had idiopathic or familial disease, or pulmonary arterial hypertension associated with connective-tissue disease, congenital heart disease, portal hypertension with liver cirrhosis, anorexigen use, or amphetamine use.
A total of 44% of patients were already receiving an endothelin-receptor antagonist (usually bosentan), and 6% were taking prostanoids (usually inhaled iloprost) for the disorder, and the other half of the study population was not receiving any other treatments. Concomitant therapy with oral anticoagulants, diuretics, and supplemental oxygen was permitted during the study.
These study subjects were randomly assigned to receive high-dose riociguat capped at 2.5 mg three times daily (254 patients), low-dose riociguat capped at 1.5 mg three times daily (63 patients), or placebo (126 patients) for 12 weeks. The analysis of data from the low-dose group was considered exploratory and was reported separately.
The primary endpoint – change from baseline in 6-minute walk distance – increased by a mean of 30 m in the high-dose group but decreased by a mean of 6 m in the placebo group in the intention-to-treat analysis. The results were similar in the per-protocol analysis, the investigators reported (N. Engl. J. Med. 2013 July 25 [doi: 10.1056/NEJMoa1209655]).
This treatment benefit was consistent across several subgroups of patients. In addition, the active drug significantly decreased pulmonary vascular resistance, improved mean pulmonary artery pressure and cardiac output, lengthened the interval to clinical worsening, improved NT-proBNP levels, improved WHO functional class, and improved scores on the Borg dyspnea scale.
In exploratory analyses, riociguat did not improve scores on one measure of QOL but did so on a disease-specific QOL tool.
Drug-related serious adverse events occurred in 1.4 % of patients receiving high-dose riociguat and 1% of those receiving placebo. A total of 3% of patients in the riociguat group and 7% of those in the placebo group discontinued the study drug because of adverse events.
Serious or drug-related adverse events in both groups included increased hepatic enzyme levels, acute renal failure, syncope, esophageal pain and swelling, supraventricular tachycardia, hypotension, generalized edema, hypoxemia, dyspnea, and worsening of pulmonary hypertension. There were two deaths in the riociguat group and three in the placebo group, none of which were considered to be related to the study drug.
A total of 396 of the subjects in the PATENT-1 study elected to enroll in an extended study of the long-term safety and efficacy of riociguat (the PATENT-2 clinical trial). An exploratory analysis of data from the first 12 weeks of that study showed a further mean increase of 53 m in 6-minute walk distance with high-dose riociguat.
CHEST-1 and PATENT-1 were funded by Bayer HealthCare. Dr. Ghofrani reported ties to Actelion, Bayer, and other companies. His associates reported ties to numerous industry sources.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major finding: Riociguat significantly improved 6-minute walk distance by a mean of 39 m in patients with thromboembolic pulmonary hypertension and by a mean of 30 m in patients with pulmonary arterial hypertension.
Data source: Two separate, international, phase III, randomized controlled trials assessing the safety and efficacy of riociguat for thromboembolic pulmonary hypertension (261 patients) and pulmonary arterial hypertension (443 patients).
Disclosures: CHEST-1 and PATENT-1 were funded by Bayer HealthCare. Dr. Ghofrani reported ties to Actelion, Bayer, and other companies. His associates reported ties to numerous industry sources.
Copeptin helped differential diagnosis of hyponatremia
SAN FRANCISCO – Measuring plasma copeptin levels may provide an accurate and easier way to identify the cause of severe hyponatremia, compared with measuring arginine vasopressin levels, preliminary data on 175 patients suggested.
A plasma copeptin level greater than 70 pmol/L correlated with a diagnosis of hypovolemic or diuretic-induced hyponatremia requiring saline infusion with 91% specificity. A copeptin/urinary sodium ratio greater than 1.84 identified patients who clearly needed saline infusion with a specificity of 90%, Dr. Nicole Nigro reported at the annual meeting of the Endocrine Society.
"The differential diagnosis of hyponatremia is not easy to make" and often relies on detecting adequate or inadequate plasma levels of arginine vasopressin (AVP), she said in an interview. Blood samples for AVP measurements must be kept on ice, making handling and transportation to the lab more difficult. AVP mostly attaches to platelets, it clears rapidly, and AVP levels are known to be very unstable.
Copeptin, on the other hand, is very stable in plasma, easy to measure, and released by the body in an equimolar ratio with AVP, said Dr. Nigro of University Hospital, Basel, Switzerland.
She presented data on the first 175 patients in an ongoing study that has enrolled 290 of an expected 300 consecutive patients who present with severe hypo-osmolar hyponatremia at the emergency departments of two Swiss tertiary referral centers. All patients had a sodium level below 125 mmol/L. Three experts who were blinded to patients’ copeptin levels made the final diagnoses based on a clinical algorithm, a chart review, and response to therapy.
The median plasma copeptin level was 2.8 pmol/L in 17 patients diagnosed with primary polydipsia, 13.2 pmol/L in 45 patients with diuretic-induced hyponatremia, 13 pmol/L in 56 patients with syndrome of inappropriate antidiuretic hormone, 28 pmol/L in 25 patients with hypervolemic hyponatremia, and 55 pmol/L in 32 patients with hypovolemic hyponatremia, Dr. Nigro reported in an oral presentation and a featured poster at the meeting.
The 77 patients who required saline infusion had significantly higher copeptin levels (27 pmol/L), than patients who did not need saline (12 pmol/L).
Dr. Nigro’s hospital routinely measures copeptin levels but is not yet using them to guide the differential diagnosis of hyponatremia. If the current study produces clear findings when it’s finished, the investigators next may conduct a study that uses copeptin levels to guide treatment of patients with severe hyponatremia.
Ultimately, "copeptin may help us to guide the therapy and management of these patients with severe hyponatremia," she suggested.
Some of Dr. Nigro’s associates in the study have been speakers for B.R.A.H.M.S./Thermo Fisher Scientific. She reported having no other relevant financial disclosures.
On Twitter @sherryboschert
SAN FRANCISCO – Measuring plasma copeptin levels may provide an accurate and easier way to identify the cause of severe hyponatremia, compared with measuring arginine vasopressin levels, preliminary data on 175 patients suggested.
A plasma copeptin level greater than 70 pmol/L correlated with a diagnosis of hypovolemic or diuretic-induced hyponatremia requiring saline infusion with 91% specificity. A copeptin/urinary sodium ratio greater than 1.84 identified patients who clearly needed saline infusion with a specificity of 90%, Dr. Nicole Nigro reported at the annual meeting of the Endocrine Society.
"The differential diagnosis of hyponatremia is not easy to make" and often relies on detecting adequate or inadequate plasma levels of arginine vasopressin (AVP), she said in an interview. Blood samples for AVP measurements must be kept on ice, making handling and transportation to the lab more difficult. AVP mostly attaches to platelets, it clears rapidly, and AVP levels are known to be very unstable.
Copeptin, on the other hand, is very stable in plasma, easy to measure, and released by the body in an equimolar ratio with AVP, said Dr. Nigro of University Hospital, Basel, Switzerland.
She presented data on the first 175 patients in an ongoing study that has enrolled 290 of an expected 300 consecutive patients who present with severe hypo-osmolar hyponatremia at the emergency departments of two Swiss tertiary referral centers. All patients had a sodium level below 125 mmol/L. Three experts who were blinded to patients’ copeptin levels made the final diagnoses based on a clinical algorithm, a chart review, and response to therapy.
The median plasma copeptin level was 2.8 pmol/L in 17 patients diagnosed with primary polydipsia, 13.2 pmol/L in 45 patients with diuretic-induced hyponatremia, 13 pmol/L in 56 patients with syndrome of inappropriate antidiuretic hormone, 28 pmol/L in 25 patients with hypervolemic hyponatremia, and 55 pmol/L in 32 patients with hypovolemic hyponatremia, Dr. Nigro reported in an oral presentation and a featured poster at the meeting.
The 77 patients who required saline infusion had significantly higher copeptin levels (27 pmol/L), than patients who did not need saline (12 pmol/L).
Dr. Nigro’s hospital routinely measures copeptin levels but is not yet using them to guide the differential diagnosis of hyponatremia. If the current study produces clear findings when it’s finished, the investigators next may conduct a study that uses copeptin levels to guide treatment of patients with severe hyponatremia.
Ultimately, "copeptin may help us to guide the therapy and management of these patients with severe hyponatremia," she suggested.
Some of Dr. Nigro’s associates in the study have been speakers for B.R.A.H.M.S./Thermo Fisher Scientific. She reported having no other relevant financial disclosures.
On Twitter @sherryboschert
SAN FRANCISCO – Measuring plasma copeptin levels may provide an accurate and easier way to identify the cause of severe hyponatremia, compared with measuring arginine vasopressin levels, preliminary data on 175 patients suggested.
A plasma copeptin level greater than 70 pmol/L correlated with a diagnosis of hypovolemic or diuretic-induced hyponatremia requiring saline infusion with 91% specificity. A copeptin/urinary sodium ratio greater than 1.84 identified patients who clearly needed saline infusion with a specificity of 90%, Dr. Nicole Nigro reported at the annual meeting of the Endocrine Society.
"The differential diagnosis of hyponatremia is not easy to make" and often relies on detecting adequate or inadequate plasma levels of arginine vasopressin (AVP), she said in an interview. Blood samples for AVP measurements must be kept on ice, making handling and transportation to the lab more difficult. AVP mostly attaches to platelets, it clears rapidly, and AVP levels are known to be very unstable.
Copeptin, on the other hand, is very stable in plasma, easy to measure, and released by the body in an equimolar ratio with AVP, said Dr. Nigro of University Hospital, Basel, Switzerland.
She presented data on the first 175 patients in an ongoing study that has enrolled 290 of an expected 300 consecutive patients who present with severe hypo-osmolar hyponatremia at the emergency departments of two Swiss tertiary referral centers. All patients had a sodium level below 125 mmol/L. Three experts who were blinded to patients’ copeptin levels made the final diagnoses based on a clinical algorithm, a chart review, and response to therapy.
The median plasma copeptin level was 2.8 pmol/L in 17 patients diagnosed with primary polydipsia, 13.2 pmol/L in 45 patients with diuretic-induced hyponatremia, 13 pmol/L in 56 patients with syndrome of inappropriate antidiuretic hormone, 28 pmol/L in 25 patients with hypervolemic hyponatremia, and 55 pmol/L in 32 patients with hypovolemic hyponatremia, Dr. Nigro reported in an oral presentation and a featured poster at the meeting.
The 77 patients who required saline infusion had significantly higher copeptin levels (27 pmol/L), than patients who did not need saline (12 pmol/L).
Dr. Nigro’s hospital routinely measures copeptin levels but is not yet using them to guide the differential diagnosis of hyponatremia. If the current study produces clear findings when it’s finished, the investigators next may conduct a study that uses copeptin levels to guide treatment of patients with severe hyponatremia.
Ultimately, "copeptin may help us to guide the therapy and management of these patients with severe hyponatremia," she suggested.
Some of Dr. Nigro’s associates in the study have been speakers for B.R.A.H.M.S./Thermo Fisher Scientific. She reported having no other relevant financial disclosures.
On Twitter @sherryboschert
AT ENDO 2013
Major finding: A plasma copeptin level greater than 70 pmol/L in patients presenting with severe hypo-osmolar hyponatremia correlated with a diagnosis of hypovolemic or diuretic-induced hyponatremia requiring saline infusion with 91% specificity.
Data source: A prospective, multicenter observational study of 175 consecutive patients presenting to emergency departments at two Swiss tertiary referral centers.
Disclosures: Some of Dr. Nigro’s associates in the study have been speakers for B.R.A.H.M.S./Thermo Fisher Scientific. She reported having no other relevant financial disclosures.
Cardiac rehabilitation benefits elderly heart failure patients
ROME – A multiweek program of cardiac rehabilitation is as beneficial in elderly patients with chronic heart failure as it is in younger heart failure patients, according to a review of 243 patients at one Belgium center.
"Although they have lower exercise capacity at baseline, older patients have at least as much benefit from an exercise program as younger patients with chronic heart failure," Ms. Sofie Pardaens* reported in a poster at the annual meeting of the European Association for Cardiovascular Prevention and Rehabilitation.
A second analysis by Ms. Pardaens, a researcher at Ghent (Belgium) University, and her associates, reported in a separate poster, showed that a prolonged cardiac rehabilitation program was as effective in patients recently discharged from a heart failure hospitalization as it was in patients following cardiac surgery or after an acute coronary syndrome (ACS) event.
Their assessment of cardiac rehabilitation relative to a patient’s age included 243 patients who participated in a rehabilitation program at the University of Ghent, who had chronic heart failure, and who had an amino-terminal pro-B-type natriuretic peptide value of at least 400 pg/mL, a level very suggestive of heart failure (Circulation 2011;123:2015-9). The group included 43 patients (18%) who were at least 75 years old (average, 78 years) and 68 patients younger than 60 (average, 51 years), with the remaining 132 patients evenly distributed across the range of 60-74 years old.
All participants had just been hospitalized, for an ACS event, cardiac surgery, or heart failure.
The hospital-based rehabilitation program combined aerobic and strength training, and was designed to bring a patient’s heart rate to his anaerobic threshold during each session. Sessions occurred two or three times a week, and the full program included 45 sessions over a period of 4-5 months. The patients studied averaged 34 sessions each; patients aged 75 or older averaged 32 sessions each, while those younger than 60 averaged 35 sessions each.
The researchers measured peak exercise capacity using cardiopulmonary exercise testing at baseline and at the end of the rehabilitation session sequence, and found that the 16% average level of improvement among patients at least 75 years old closely matched the average 19% improvement among the patients younger than 60, and the 17% average improvement among everyone else, Ms. Pardaens and her associates reported. All age groups also showed similar improvements in their average ventilatory equivalence ratio, as well as their average 6-minute walk distance; however, the 29% average increased distance among patients younger than 60 years significantly exceeded the 19% average increase among those aged 75 or older.
The group’s second analysis focused on the 371 patients who underwent cardiac rehabilitation at the University of Ghent during January 2010 through May 2012 from among the 1,253 patients hospitalized during this period for an acute coronary syndrome event, cardiac surgery, or heart failure. In this pool of more than 1,000 patients who were potentially eligible to participate, only 30% actually enrolled in the rehabilitation program. The cardiac rehabilitation program again involved two to three sessions per week, with a goal for patients to complete 45 sessions within 5 months.
The sign-up rate for rehabilitation lagged even more among the 428 patients from the larger group whose index hospitalization had been for heart failure, with 37 of the acute heart failure patients (9%) actually engaging in rehabilitation. Rehabilitation participation was highest, a 56% rate, among the 358 patients who had been hospitalized for cardiac surgery, with a 28% uptake rate among 467 patients who had an ACS event.
Despite the low, 9% uptake of cardiac rehabilitation in heart failure patients, their benefit from participation closely tracked the benefit seen in surgery and ACS patients. Improvement in peak exercise capacity over baseline at the end of rehabilitation averaged 19% in the heart failure patients, 17% in the ACS patients, and 24% in the surgery patients, differences that were not statistically significant, reported Ms. Pardaens. All three subgroups also had similar average improvements in their 6-minute walk distance, which rose by an average of 21% in the heart failure patients and by averages of 27% and 28% in the other two subgroups.
Based on the efficacy but low usage of cardiac rehabilitation, future research should examine ways to boost its use by heart failure patients, concluded Ms. Pardaens.
Ms. Pardaens and her associates said they had no relevant financial disclosures.
On Twitter @mitchelzoler
*Correction 6/28/13: An earlier version of this article incorrectly reported researcher Sofie Pardaens' title. She is currently studying for her PhD in the department of internal medicine at Ghent University.
Cardiac rehabilitation is undoubtedly an essential component of the contemporary treatment of patients with coronary disease and heart failure.
Exercise training has the potential to act as a catalyst for promoting other aspects of rehabilitation, including risk factor modification through therapeutic lifestyle changes and optimization of psychosocial support. Similarly, among patients who are elderly, such outcome measures may include the achievement of functional independence, the prevention of premature disability, and a reduction in the need for custodial care.
Despite limited data, older patients have shown improvement in their exercise tolerance comparable to that of younger patients participating in equivalent exercise programs. In addition, the safety of exercise within cardiac rehabilitation programs is well accepted and established.
Dr. Jun Chiong is associate professor of medicine at Loma Linda (Calif.) University Medical Center. He is on the on the advisory board of CHEST Physician.
Cardiac rehabilitation is undoubtedly an essential component of the contemporary treatment of patients with coronary disease and heart failure.
Exercise training has the potential to act as a catalyst for promoting other aspects of rehabilitation, including risk factor modification through therapeutic lifestyle changes and optimization of psychosocial support. Similarly, among patients who are elderly, such outcome measures may include the achievement of functional independence, the prevention of premature disability, and a reduction in the need for custodial care.
Despite limited data, older patients have shown improvement in their exercise tolerance comparable to that of younger patients participating in equivalent exercise programs. In addition, the safety of exercise within cardiac rehabilitation programs is well accepted and established.
Dr. Jun Chiong is associate professor of medicine at Loma Linda (Calif.) University Medical Center. He is on the on the advisory board of CHEST Physician.
Cardiac rehabilitation is undoubtedly an essential component of the contemporary treatment of patients with coronary disease and heart failure.
Exercise training has the potential to act as a catalyst for promoting other aspects of rehabilitation, including risk factor modification through therapeutic lifestyle changes and optimization of psychosocial support. Similarly, among patients who are elderly, such outcome measures may include the achievement of functional independence, the prevention of premature disability, and a reduction in the need for custodial care.
Despite limited data, older patients have shown improvement in their exercise tolerance comparable to that of younger patients participating in equivalent exercise programs. In addition, the safety of exercise within cardiac rehabilitation programs is well accepted and established.
Dr. Jun Chiong is associate professor of medicine at Loma Linda (Calif.) University Medical Center. He is on the on the advisory board of CHEST Physician.
ROME – A multiweek program of cardiac rehabilitation is as beneficial in elderly patients with chronic heart failure as it is in younger heart failure patients, according to a review of 243 patients at one Belgium center.
"Although they have lower exercise capacity at baseline, older patients have at least as much benefit from an exercise program as younger patients with chronic heart failure," Ms. Sofie Pardaens* reported in a poster at the annual meeting of the European Association for Cardiovascular Prevention and Rehabilitation.
A second analysis by Ms. Pardaens, a researcher at Ghent (Belgium) University, and her associates, reported in a separate poster, showed that a prolonged cardiac rehabilitation program was as effective in patients recently discharged from a heart failure hospitalization as it was in patients following cardiac surgery or after an acute coronary syndrome (ACS) event.
Their assessment of cardiac rehabilitation relative to a patient’s age included 243 patients who participated in a rehabilitation program at the University of Ghent, who had chronic heart failure, and who had an amino-terminal pro-B-type natriuretic peptide value of at least 400 pg/mL, a level very suggestive of heart failure (Circulation 2011;123:2015-9). The group included 43 patients (18%) who were at least 75 years old (average, 78 years) and 68 patients younger than 60 (average, 51 years), with the remaining 132 patients evenly distributed across the range of 60-74 years old.
All participants had just been hospitalized, for an ACS event, cardiac surgery, or heart failure.
The hospital-based rehabilitation program combined aerobic and strength training, and was designed to bring a patient’s heart rate to his anaerobic threshold during each session. Sessions occurred two or three times a week, and the full program included 45 sessions over a period of 4-5 months. The patients studied averaged 34 sessions each; patients aged 75 or older averaged 32 sessions each, while those younger than 60 averaged 35 sessions each.
The researchers measured peak exercise capacity using cardiopulmonary exercise testing at baseline and at the end of the rehabilitation session sequence, and found that the 16% average level of improvement among patients at least 75 years old closely matched the average 19% improvement among the patients younger than 60, and the 17% average improvement among everyone else, Ms. Pardaens and her associates reported. All age groups also showed similar improvements in their average ventilatory equivalence ratio, as well as their average 6-minute walk distance; however, the 29% average increased distance among patients younger than 60 years significantly exceeded the 19% average increase among those aged 75 or older.
The group’s second analysis focused on the 371 patients who underwent cardiac rehabilitation at the University of Ghent during January 2010 through May 2012 from among the 1,253 patients hospitalized during this period for an acute coronary syndrome event, cardiac surgery, or heart failure. In this pool of more than 1,000 patients who were potentially eligible to participate, only 30% actually enrolled in the rehabilitation program. The cardiac rehabilitation program again involved two to three sessions per week, with a goal for patients to complete 45 sessions within 5 months.
The sign-up rate for rehabilitation lagged even more among the 428 patients from the larger group whose index hospitalization had been for heart failure, with 37 of the acute heart failure patients (9%) actually engaging in rehabilitation. Rehabilitation participation was highest, a 56% rate, among the 358 patients who had been hospitalized for cardiac surgery, with a 28% uptake rate among 467 patients who had an ACS event.
Despite the low, 9% uptake of cardiac rehabilitation in heart failure patients, their benefit from participation closely tracked the benefit seen in surgery and ACS patients. Improvement in peak exercise capacity over baseline at the end of rehabilitation averaged 19% in the heart failure patients, 17% in the ACS patients, and 24% in the surgery patients, differences that were not statistically significant, reported Ms. Pardaens. All three subgroups also had similar average improvements in their 6-minute walk distance, which rose by an average of 21% in the heart failure patients and by averages of 27% and 28% in the other two subgroups.
Based on the efficacy but low usage of cardiac rehabilitation, future research should examine ways to boost its use by heart failure patients, concluded Ms. Pardaens.
Ms. Pardaens and her associates said they had no relevant financial disclosures.
On Twitter @mitchelzoler
*Correction 6/28/13: An earlier version of this article incorrectly reported researcher Sofie Pardaens' title. She is currently studying for her PhD in the department of internal medicine at Ghent University.
ROME – A multiweek program of cardiac rehabilitation is as beneficial in elderly patients with chronic heart failure as it is in younger heart failure patients, according to a review of 243 patients at one Belgium center.
"Although they have lower exercise capacity at baseline, older patients have at least as much benefit from an exercise program as younger patients with chronic heart failure," Ms. Sofie Pardaens* reported in a poster at the annual meeting of the European Association for Cardiovascular Prevention and Rehabilitation.
A second analysis by Ms. Pardaens, a researcher at Ghent (Belgium) University, and her associates, reported in a separate poster, showed that a prolonged cardiac rehabilitation program was as effective in patients recently discharged from a heart failure hospitalization as it was in patients following cardiac surgery or after an acute coronary syndrome (ACS) event.
Their assessment of cardiac rehabilitation relative to a patient’s age included 243 patients who participated in a rehabilitation program at the University of Ghent, who had chronic heart failure, and who had an amino-terminal pro-B-type natriuretic peptide value of at least 400 pg/mL, a level very suggestive of heart failure (Circulation 2011;123:2015-9). The group included 43 patients (18%) who were at least 75 years old (average, 78 years) and 68 patients younger than 60 (average, 51 years), with the remaining 132 patients evenly distributed across the range of 60-74 years old.
All participants had just been hospitalized, for an ACS event, cardiac surgery, or heart failure.
The hospital-based rehabilitation program combined aerobic and strength training, and was designed to bring a patient’s heart rate to his anaerobic threshold during each session. Sessions occurred two or three times a week, and the full program included 45 sessions over a period of 4-5 months. The patients studied averaged 34 sessions each; patients aged 75 or older averaged 32 sessions each, while those younger than 60 averaged 35 sessions each.
The researchers measured peak exercise capacity using cardiopulmonary exercise testing at baseline and at the end of the rehabilitation session sequence, and found that the 16% average level of improvement among patients at least 75 years old closely matched the average 19% improvement among the patients younger than 60, and the 17% average improvement among everyone else, Ms. Pardaens and her associates reported. All age groups also showed similar improvements in their average ventilatory equivalence ratio, as well as their average 6-minute walk distance; however, the 29% average increased distance among patients younger than 60 years significantly exceeded the 19% average increase among those aged 75 or older.
The group’s second analysis focused on the 371 patients who underwent cardiac rehabilitation at the University of Ghent during January 2010 through May 2012 from among the 1,253 patients hospitalized during this period for an acute coronary syndrome event, cardiac surgery, or heart failure. In this pool of more than 1,000 patients who were potentially eligible to participate, only 30% actually enrolled in the rehabilitation program. The cardiac rehabilitation program again involved two to three sessions per week, with a goal for patients to complete 45 sessions within 5 months.
The sign-up rate for rehabilitation lagged even more among the 428 patients from the larger group whose index hospitalization had been for heart failure, with 37 of the acute heart failure patients (9%) actually engaging in rehabilitation. Rehabilitation participation was highest, a 56% rate, among the 358 patients who had been hospitalized for cardiac surgery, with a 28% uptake rate among 467 patients who had an ACS event.
Despite the low, 9% uptake of cardiac rehabilitation in heart failure patients, their benefit from participation closely tracked the benefit seen in surgery and ACS patients. Improvement in peak exercise capacity over baseline at the end of rehabilitation averaged 19% in the heart failure patients, 17% in the ACS patients, and 24% in the surgery patients, differences that were not statistically significant, reported Ms. Pardaens. All three subgroups also had similar average improvements in their 6-minute walk distance, which rose by an average of 21% in the heart failure patients and by averages of 27% and 28% in the other two subgroups.
Based on the efficacy but low usage of cardiac rehabilitation, future research should examine ways to boost its use by heart failure patients, concluded Ms. Pardaens.
Ms. Pardaens and her associates said they had no relevant financial disclosures.
On Twitter @mitchelzoler
*Correction 6/28/13: An earlier version of this article incorrectly reported researcher Sofie Pardaens' title. She is currently studying for her PhD in the department of internal medicine at Ghent University.
AT EUROPREVENT 2013
Major finding: Cardiac rehabilitation boosted exercise capacity by an average 16% in elderly patients and an average 19% in younger patients.
Data source: Data came from a review of 243 patients with chronic heart failure who participated in cardiac rehabilitation at one Belgium center.
Disclosures: Dr. Pardaens and her associates said they had no relevant financial disclosures.
Hospital revenue generated by cardiologists continues to drop
Average annual hospital revenue generated by invasive cardiologists dropped 3.2% from 2010 to 2013, while the revenue generated by noninvasive cardiologists dropped 6.6%, according to a survey by physician job placement firm Merritt Hawkins.
"In the last several years, some services provided by cardiologists have been considered reimbursement ‘outliers’ by Medicare and have been targeted for significant" cuts, the survey authors noted, adding that "revenues generated by invasive cardiologists for hospitals can be expected to decline in coming years."
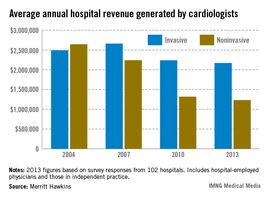
Average revenue of $2.17 million was reported for invasive cardiologists and just under $1.23 million for noninvasive cardiologists in 2013. That compares with almost $1.42 million for all specialists and $1.57 for all primary care physicians. The specialists’ average is the lowest Merritt Hawkins has reported since it started the survey in 2002 and is lower than the primary care average for the first time ever, the report noted.
The average net revenue generated for all physicians was $1.45 million – a drop of 6.2% from 2010 and, again, the lowest average since 2002, Merritt Hawkins said in its analysis.
The survey was sent to 5,500 hospitals in January 2013 and completed by 102 hospital chief financial officers.
Average annual hospital revenue generated by invasive cardiologists dropped 3.2% from 2010 to 2013, while the revenue generated by noninvasive cardiologists dropped 6.6%, according to a survey by physician job placement firm Merritt Hawkins.
"In the last several years, some services provided by cardiologists have been considered reimbursement ‘outliers’ by Medicare and have been targeted for significant" cuts, the survey authors noted, adding that "revenues generated by invasive cardiologists for hospitals can be expected to decline in coming years."

Average revenue of $2.17 million was reported for invasive cardiologists and just under $1.23 million for noninvasive cardiologists in 2013. That compares with almost $1.42 million for all specialists and $1.57 for all primary care physicians. The specialists’ average is the lowest Merritt Hawkins has reported since it started the survey in 2002 and is lower than the primary care average for the first time ever, the report noted.
The average net revenue generated for all physicians was $1.45 million – a drop of 6.2% from 2010 and, again, the lowest average since 2002, Merritt Hawkins said in its analysis.
The survey was sent to 5,500 hospitals in January 2013 and completed by 102 hospital chief financial officers.
Average annual hospital revenue generated by invasive cardiologists dropped 3.2% from 2010 to 2013, while the revenue generated by noninvasive cardiologists dropped 6.6%, according to a survey by physician job placement firm Merritt Hawkins.
"In the last several years, some services provided by cardiologists have been considered reimbursement ‘outliers’ by Medicare and have been targeted for significant" cuts, the survey authors noted, adding that "revenues generated by invasive cardiologists for hospitals can be expected to decline in coming years."

Average revenue of $2.17 million was reported for invasive cardiologists and just under $1.23 million for noninvasive cardiologists in 2013. That compares with almost $1.42 million for all specialists and $1.57 for all primary care physicians. The specialists’ average is the lowest Merritt Hawkins has reported since it started the survey in 2002 and is lower than the primary care average for the first time ever, the report noted.
The average net revenue generated for all physicians was $1.45 million – a drop of 6.2% from 2010 and, again, the lowest average since 2002, Merritt Hawkins said in its analysis.
The survey was sent to 5,500 hospitals in January 2013 and completed by 102 hospital chief financial officers.
Shock-Less trial improves physicians' ICD programming
DENVER – The rate of adherence to evidence-based implantable cardioverter-defibrillator programming strategies known to reduce the rate of unnecessary shocks climbed significantly in a large, prospective study in which physicians received detailed reports on their own performance and how it stacked up to that of others.
The Shock-Less study was a real-world international study involving 4,131 ICD recipients and their physicians at 118 sites. The improved adherence to evidence-based programming achieved through the use of the individualized, multipage therapy programming reports (TPRs) translated into a highly significant 27% reduction in the risk of all-cause ICD shocks during follow-up, Dr. Marc T. Silver reported at the annual meeting of the Heart Rhythm Society.
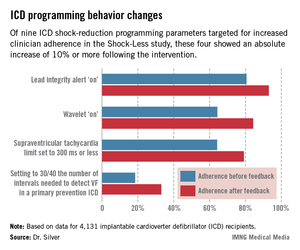
This is welcome news for patients. It means less shock-related morbidity and potentially less mortality, said Dr. Silver of WakeMed in Raleigh, N.C.
The study included 2,693 patients who received their Medtronic primary or secondary prevention ICD before a center received its first Shock-Less report and 1,438 implanted after the report. A total of 265 all-cause shocks occurred in the "before" group, 116 in the "after" cohort.
In a multivariate logistic regression analysis adjusted for factors known to affect shock rates, including patient age, smoking status, New York Heart Association functional class, and atrial fibrillation, patients in the "after" group had a 27% reduction in the relative risk of both appropriate and inappropriate shocks (P = .002).
The TPRs provided ICD centers and their individual physicians with detailed feedback on rates of adherence to nine evidence-based programming settings that help reduce shocks. Most ICDs don’t arrive from the manufacturer with these settings in place. Some of the changes in programming were quite impressive (see graphic), including a near-doubling of the rate of primary prevention ICDs programmed to 30/40 as the number of intervals to detect ventricular fibrillation; this rate improved from 18.5% to 33.1%.
Dr. Silver offered personal testimony as to the power of the TPRs as a behavior-modification tool.
"Having received TPRs myself, it is a character-building experience. If you can get hold of information like this on your own practices, I guarantee you will leave a little smaller afterwards, like I did. Bigger in some way, smaller in others," he said.
Yet there remains a clear opportunity for further improvement in physician performance, Dr. Silver added.
"Achieving numbers in the 33% range for adherence to 30/40 [the number of intervals needed to detect ventricular fibrillation] is not what I think many of us would hope for one day," he observed.
Along those lines, audience member Dr. Thomas F. Deering of the Piedmont Heart Institute, Atlanta, commented that while the changes in physician behavior achieved through the Shock-Less project were significant, they were not sweeping in magnitude. Given the negative clinical consequences that result from lack of adherence to evidence-based programming, isn’t it time to request that the device industry change the default settings on their ICDs in accord with the evidence-based guidelines?, he asked.
"As someone who’s become very interested in physician behavior," Dr. Silver replied, "I regret to say that changing nominal settings on the devices may be the best way to move our profession forward. I say that with some degree of regret, but I think that’s the truth."
Dr. Silver reported serving as a consultant to Medtronic, which sponsored the Shock-Less study.
DENVER – The rate of adherence to evidence-based implantable cardioverter-defibrillator programming strategies known to reduce the rate of unnecessary shocks climbed significantly in a large, prospective study in which physicians received detailed reports on their own performance and how it stacked up to that of others.
The Shock-Less study was a real-world international study involving 4,131 ICD recipients and their physicians at 118 sites. The improved adherence to evidence-based programming achieved through the use of the individualized, multipage therapy programming reports (TPRs) translated into a highly significant 27% reduction in the risk of all-cause ICD shocks during follow-up, Dr. Marc T. Silver reported at the annual meeting of the Heart Rhythm Society.

This is welcome news for patients. It means less shock-related morbidity and potentially less mortality, said Dr. Silver of WakeMed in Raleigh, N.C.
The study included 2,693 patients who received their Medtronic primary or secondary prevention ICD before a center received its first Shock-Less report and 1,438 implanted after the report. A total of 265 all-cause shocks occurred in the "before" group, 116 in the "after" cohort.
In a multivariate logistic regression analysis adjusted for factors known to affect shock rates, including patient age, smoking status, New York Heart Association functional class, and atrial fibrillation, patients in the "after" group had a 27% reduction in the relative risk of both appropriate and inappropriate shocks (P = .002).
The TPRs provided ICD centers and their individual physicians with detailed feedback on rates of adherence to nine evidence-based programming settings that help reduce shocks. Most ICDs don’t arrive from the manufacturer with these settings in place. Some of the changes in programming were quite impressive (see graphic), including a near-doubling of the rate of primary prevention ICDs programmed to 30/40 as the number of intervals to detect ventricular fibrillation; this rate improved from 18.5% to 33.1%.
Dr. Silver offered personal testimony as to the power of the TPRs as a behavior-modification tool.
"Having received TPRs myself, it is a character-building experience. If you can get hold of information like this on your own practices, I guarantee you will leave a little smaller afterwards, like I did. Bigger in some way, smaller in others," he said.
Yet there remains a clear opportunity for further improvement in physician performance, Dr. Silver added.
"Achieving numbers in the 33% range for adherence to 30/40 [the number of intervals needed to detect ventricular fibrillation] is not what I think many of us would hope for one day," he observed.
Along those lines, audience member Dr. Thomas F. Deering of the Piedmont Heart Institute, Atlanta, commented that while the changes in physician behavior achieved through the Shock-Less project were significant, they were not sweeping in magnitude. Given the negative clinical consequences that result from lack of adherence to evidence-based programming, isn’t it time to request that the device industry change the default settings on their ICDs in accord with the evidence-based guidelines?, he asked.
"As someone who’s become very interested in physician behavior," Dr. Silver replied, "I regret to say that changing nominal settings on the devices may be the best way to move our profession forward. I say that with some degree of regret, but I think that’s the truth."
Dr. Silver reported serving as a consultant to Medtronic, which sponsored the Shock-Less study.
DENVER – The rate of adherence to evidence-based implantable cardioverter-defibrillator programming strategies known to reduce the rate of unnecessary shocks climbed significantly in a large, prospective study in which physicians received detailed reports on their own performance and how it stacked up to that of others.
The Shock-Less study was a real-world international study involving 4,131 ICD recipients and their physicians at 118 sites. The improved adherence to evidence-based programming achieved through the use of the individualized, multipage therapy programming reports (TPRs) translated into a highly significant 27% reduction in the risk of all-cause ICD shocks during follow-up, Dr. Marc T. Silver reported at the annual meeting of the Heart Rhythm Society.

This is welcome news for patients. It means less shock-related morbidity and potentially less mortality, said Dr. Silver of WakeMed in Raleigh, N.C.
The study included 2,693 patients who received their Medtronic primary or secondary prevention ICD before a center received its first Shock-Less report and 1,438 implanted after the report. A total of 265 all-cause shocks occurred in the "before" group, 116 in the "after" cohort.
In a multivariate logistic regression analysis adjusted for factors known to affect shock rates, including patient age, smoking status, New York Heart Association functional class, and atrial fibrillation, patients in the "after" group had a 27% reduction in the relative risk of both appropriate and inappropriate shocks (P = .002).
The TPRs provided ICD centers and their individual physicians with detailed feedback on rates of adherence to nine evidence-based programming settings that help reduce shocks. Most ICDs don’t arrive from the manufacturer with these settings in place. Some of the changes in programming were quite impressive (see graphic), including a near-doubling of the rate of primary prevention ICDs programmed to 30/40 as the number of intervals to detect ventricular fibrillation; this rate improved from 18.5% to 33.1%.
Dr. Silver offered personal testimony as to the power of the TPRs as a behavior-modification tool.
"Having received TPRs myself, it is a character-building experience. If you can get hold of information like this on your own practices, I guarantee you will leave a little smaller afterwards, like I did. Bigger in some way, smaller in others," he said.
Yet there remains a clear opportunity for further improvement in physician performance, Dr. Silver added.
"Achieving numbers in the 33% range for adherence to 30/40 [the number of intervals needed to detect ventricular fibrillation] is not what I think many of us would hope for one day," he observed.
Along those lines, audience member Dr. Thomas F. Deering of the Piedmont Heart Institute, Atlanta, commented that while the changes in physician behavior achieved through the Shock-Less project were significant, they were not sweeping in magnitude. Given the negative clinical consequences that result from lack of adherence to evidence-based programming, isn’t it time to request that the device industry change the default settings on their ICDs in accord with the evidence-based guidelines?, he asked.
"As someone who’s become very interested in physician behavior," Dr. Silver replied, "I regret to say that changing nominal settings on the devices may be the best way to move our profession forward. I say that with some degree of regret, but I think that’s the truth."
Dr. Silver reported serving as a consultant to Medtronic, which sponsored the Shock-Less study.
AT HEART RHYTHM 2013
Major Finding: A 27% reduction in shocks from ICDs ensued after physicians received detailed, structured reports on their rates of adherence to evidence-based shock-reduction programming strategies.
Data Source: Shock-Less is an international prospective cohort study involving 4,131 ICD recipients and their physicians at 118 centers.
Disclosures: Dr. Silver reported serving as a consultant to Medtronic, which sponsored the Shock-Less study.