User login
Getting a Leg up on the Diagnosis
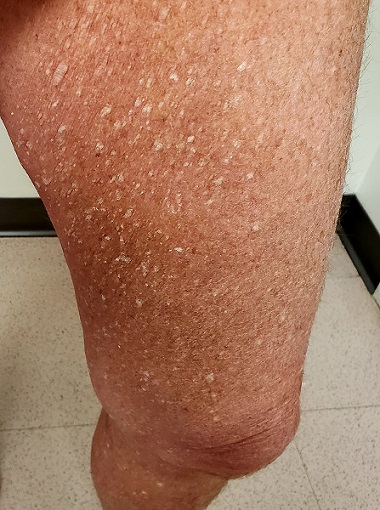
Three years ago, lesions began appearing on this now 68-year-old woman’s legs. They have grown in size and number, and their roughness disturbs the patient. She has been told the lesions are related to aging, but she has never seen anything like them on her friends or family—and she is worried about what they might mean for her health.
Her primary care provider diagnosed warts and performed cryotherapy on several of the lesions. However, the pain was intolerable and the treatment ineffective. To add insult to injury, each treated spot blistered and took more than a month to heal, leaving behind a pinkish brown blemish.
In all other respects, the patient’s health is excellent.
EXAMINATION
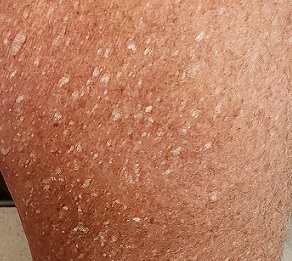
Both legs, from the upper thighs to the tops of the feet, are covered with thousands of uniformly distributed, tiny, keratotic, rough, dry papules. All the lesions are essentially identical: white, with no associated signs of inflammation. The patient’s skin is quite dry in general. Neither her palms nor soles are affected.
What’s the diagnosis?
DISCUSSION
The most common problem seen in dermatology offices worldwide is seborrheic keratosis (SK), a totally benign epidermal excrescence that appears to be related to aging and heredity. Most patients are in their 50s when they first notice an SK, and with a bit of luck, they will only see a few in their lifetime. But some patients develop hundreds of SKs, many of which become quite large (3-5 cm) and unsightly. In certain circumstances, SKs can herald the arrival of an occult carcinoma (the Leser-Trelat sign).
This patient has what some consider a variant of SK, called stucco keratosis. These lesions manifest almost exclusively on the lower legs and feet—perhaps due to the relative lack of sebaceous glands in those areas—and most often on men older than 60. Distressing as they are, stucco keratoses have no pathologic implications.
Grossly and histologically, stucco keratoses are different from ordinary SKs. Each stucco keratosis lesion is essentially identical to the others, with a spiculated surface, white color, and average diameter of 2 to 3 mm. Histologically, they demonstrate a thickened epidermis with focal exophytic upward projections that resemble church spires. The lesions do not extend into the dermis.
Treatment of stucco keratoses is, at best, tedious, painful, and futile. The modalities used are cryotherapy or electrodessication with curettage. For a degree of comfort, use of a loofah after bathing will remove or smooth down a few lesions, but this process must be followed by application of a heavy emollient. Alas, regrowth is a certainty.
TAKE-HOME LEARNING POINTS
- Stucco keratosis is considered a variant of seborrheic keratosis, although they differ in several significant ways.
- The lesions of stucco keratosis are fairly uniform in appearance: white, rough, spiculated, epidermal papules measuring 2 to 3 mm.
- Stucco keratoses affect about 10% of the population (men more often than women) and have no racial predilection or pathologic implications.
- The lesions are found almost exclusively on the legs, from the knees down to and including the dorsa of the feet.
- Treatment is far from satisfactory, for multiple reasons, including resultant pain and scarring.
Three years ago, lesions began appearing on this now 68-year-old woman’s legs. They have grown in size and number, and their roughness disturbs the patient. She has been told the lesions are related to aging, but she has never seen anything like them on her friends or family—and she is worried about what they might mean for her health.
Her primary care provider diagnosed warts and performed cryotherapy on several of the lesions. However, the pain was intolerable and the treatment ineffective. To add insult to injury, each treated spot blistered and took more than a month to heal, leaving behind a pinkish brown blemish.
In all other respects, the patient’s health is excellent.
EXAMINATION
Both legs, from the upper thighs to the tops of the feet, are covered with thousands of uniformly distributed, tiny, keratotic, rough, dry papules. All the lesions are essentially identical: white, with no associated signs of inflammation. The patient’s skin is quite dry in general. Neither her palms nor soles are affected.

What’s the diagnosis?
DISCUSSION
The most common problem seen in dermatology offices worldwide is seborrheic keratosis (SK), a totally benign epidermal excrescence that appears to be related to aging and heredity. Most patients are in their 50s when they first notice an SK, and with a bit of luck, they will only see a few in their lifetime. But some patients develop hundreds of SKs, many of which become quite large (3-5 cm) and unsightly. In certain circumstances, SKs can herald the arrival of an occult carcinoma (the Leser-Trelat sign).
This patient has what some consider a variant of SK, called stucco keratosis. These lesions manifest almost exclusively on the lower legs and feet—perhaps due to the relative lack of sebaceous glands in those areas—and most often on men older than 60. Distressing as they are, stucco keratoses have no pathologic implications.
Grossly and histologically, stucco keratoses are different from ordinary SKs. Each stucco keratosis lesion is essentially identical to the others, with a spiculated surface, white color, and average diameter of 2 to 3 mm. Histologically, they demonstrate a thickened epidermis with focal exophytic upward projections that resemble church spires. The lesions do not extend into the dermis.
Treatment of stucco keratoses is, at best, tedious, painful, and futile. The modalities used are cryotherapy or electrodessication with curettage. For a degree of comfort, use of a loofah after bathing will remove or smooth down a few lesions, but this process must be followed by application of a heavy emollient. Alas, regrowth is a certainty.
TAKE-HOME LEARNING POINTS
- Stucco keratosis is considered a variant of seborrheic keratosis, although they differ in several significant ways.
- The lesions of stucco keratosis are fairly uniform in appearance: white, rough, spiculated, epidermal papules measuring 2 to 3 mm.
- Stucco keratoses affect about 10% of the population (men more often than women) and have no racial predilection or pathologic implications.
- The lesions are found almost exclusively on the legs, from the knees down to and including the dorsa of the feet.
- Treatment is far from satisfactory, for multiple reasons, including resultant pain and scarring.
Three years ago, lesions began appearing on this now 68-year-old woman’s legs. They have grown in size and number, and their roughness disturbs the patient. She has been told the lesions are related to aging, but she has never seen anything like them on her friends or family—and she is worried about what they might mean for her health.
Her primary care provider diagnosed warts and performed cryotherapy on several of the lesions. However, the pain was intolerable and the treatment ineffective. To add insult to injury, each treated spot blistered and took more than a month to heal, leaving behind a pinkish brown blemish.
In all other respects, the patient’s health is excellent.
EXAMINATION
Both legs, from the upper thighs to the tops of the feet, are covered with thousands of uniformly distributed, tiny, keratotic, rough, dry papules. All the lesions are essentially identical: white, with no associated signs of inflammation. The patient’s skin is quite dry in general. Neither her palms nor soles are affected.

What’s the diagnosis?
DISCUSSION
The most common problem seen in dermatology offices worldwide is seborrheic keratosis (SK), a totally benign epidermal excrescence that appears to be related to aging and heredity. Most patients are in their 50s when they first notice an SK, and with a bit of luck, they will only see a few in their lifetime. But some patients develop hundreds of SKs, many of which become quite large (3-5 cm) and unsightly. In certain circumstances, SKs can herald the arrival of an occult carcinoma (the Leser-Trelat sign).
This patient has what some consider a variant of SK, called stucco keratosis. These lesions manifest almost exclusively on the lower legs and feet—perhaps due to the relative lack of sebaceous glands in those areas—and most often on men older than 60. Distressing as they are, stucco keratoses have no pathologic implications.
Grossly and histologically, stucco keratoses are different from ordinary SKs. Each stucco keratosis lesion is essentially identical to the others, with a spiculated surface, white color, and average diameter of 2 to 3 mm. Histologically, they demonstrate a thickened epidermis with focal exophytic upward projections that resemble church spires. The lesions do not extend into the dermis.
Treatment of stucco keratoses is, at best, tedious, painful, and futile. The modalities used are cryotherapy or electrodessication with curettage. For a degree of comfort, use of a loofah after bathing will remove or smooth down a few lesions, but this process must be followed by application of a heavy emollient. Alas, regrowth is a certainty.
TAKE-HOME LEARNING POINTS
- Stucco keratosis is considered a variant of seborrheic keratosis, although they differ in several significant ways.
- The lesions of stucco keratosis are fairly uniform in appearance: white, rough, spiculated, epidermal papules measuring 2 to 3 mm.
- Stucco keratoses affect about 10% of the population (men more often than women) and have no racial predilection or pathologic implications.
- The lesions are found almost exclusively on the legs, from the knees down to and including the dorsa of the feet.
- Treatment is far from satisfactory, for multiple reasons, including resultant pain and scarring.
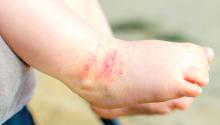
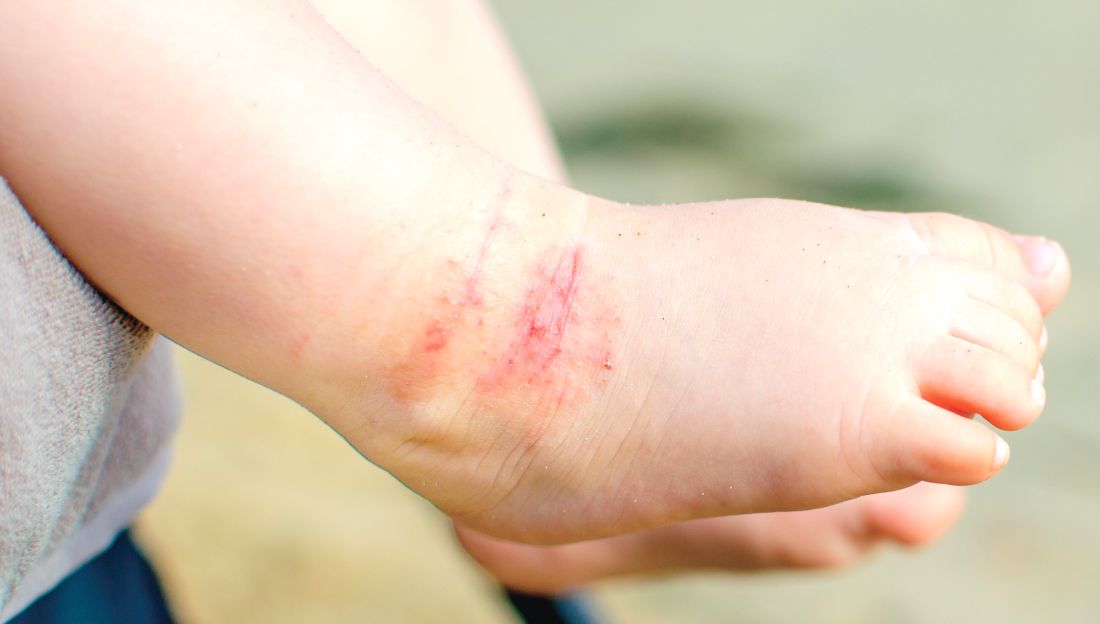
Desquamating pustular rash
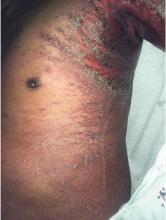
A 66-year-old man presented with burning pain and erythema over the left axilla, and pustules that had ruptured and crusted over. The rash also involved the right axilla, trunk, abdomen, and face.
He said the symptoms had developed 3 days after starting to use ciprofloxacin eye drops for eye redness and purulent discharge that had been diagnosed as bacterial conjunctivitis. He was taking no other new medications. He was afebrile.
The ciprofloxacin drops were stopped. The skin lesions were treated with emollients, topical steroids, and topical mupirocin. Improvement was noted 3 days into the hospitalization, as the lesions started to crust over and dry up and no new lesions were forming. The conjunctivitis improved with topical bacitracin ointment and prednisolone drops.
ACUTE GENERALIZED EXANTHEMATOUS PUSTULOSIS
The differential diagnosis of drug-related acute generalized exanthematous pustulosis includes Stevens-Johnson syndrome, pustular psoriasis, folliculitis, and varicella infection. The characteristic features of the rash and lesions and the temporal relationship between the start of ciprofloxacin eye drops and the development of symptoms, combined with rapid resolution of symptoms within days after discontinuing the drops, accompanied by skin biopsy study showing diffuse spongiosis with scattered eosinophils and a subcorneal pustule, confirmed the diagnosis of AGEP.
Key features of AGEP include numerous small, sterile, nonfollicular pustules on an erythematous background with associated fever and sometimes neutrophilia and eosinophilia.1 It usually begins on the face or in the intertriginous areas and then spreads to the trunk and lower limbs with rare mucosal involvement.1 It can be associated with viral infections, but most reported cases are related to drug reactions.2
Our patient’s case was unusual because AGEP triggered by topical medications is rarely reported, especially with ophthalmic medications.3 Drugs most commonly implicated are antibiotics including penicillins, sulfonamides, and quinolones, but other drugs such as terbinafine, diltiazem, and hydroxychloroquine have also been associated.2
AGEP may present with extensive skin desquamation, as in our patient, sometimes with bullae formation and skin sloughing manifesting as AGEP with overlapping toxic epidermal necrolysis.4
Diagnosis entails a careful review of medications, attention to lesion morphology, compatible disease course, and a high index of suspicion. Treatment is supportive and consists of stopping the offending agent, wound care, and antipyretics. Evidence for the use of steroids is weak.5
TAKE-HOME POINTS
AGEP should be considered in sudden-onset pustular desquamating erythematous rash related to use of a new medication. It is important to be aware that topical and ophthalmic medications are possible triggers. A thorough medication review should be done. Antibiotics are the most commonly implicated medications. An alternative medication should be tried. Treatment is supportive, as the condition is usually self-limiting once the offending medication is discontinued. Rarely, extensive desquamation and bullae formation may occur, which may be a manifestation of overlap features with toxic epidermal necrolysis.
- Speeckaert MM, Speeckaert R, Lambert J, Brochez L. Acute generalized exanthematous pustulosis: an overview of the clinical, immunological and diagnostic concepts. Eur J Dermatol 2010; 20(4):425–433. doi:10.1684/ejd.2010.0932
- Sidoroff A, Dunant A, Viboud C, et al. Risk factors for acute generalized exanthematous pustulosis (AGEP)—results of a multinational case–control study (EuroSCAR). Br J Dermatol 2007; 157(5):989–996. doi:10.1111/j.1365-2133.2007.08156.x
- Beltran C, Vergier B, Doutre MS, Beylot C, Beylot-Barry M. Acute generalized exanthematous pustulosis induced by topical application of Algipan. Ann Dermatol Venereol 2009; 136(10):709–712. French. doi:10.1016/j.annder.2008.10.042
- Peermohamed S, Haber RM. Acute generalized exanthematous pustulosis simulating toxic epidermal necrolysis: a case report and review of the literature. Arch Dermatol 2011; 147(6):697–701. doi:10.1001/archdermatol.2011.147
- Feldmeyer L, Heidemeyer K, Yawalkar N. Acute generalized exanthematous pustulosis: pathogenesis, genetic background, clinical variants and therapy. Int J Mol Sci 2016; 17(8). pii:E1214. doi:10.3390/ijms17081214
A 66-year-old man presented with burning pain and erythema over the left axilla, and pustules that had ruptured and crusted over. The rash also involved the right axilla, trunk, abdomen, and face.
He said the symptoms had developed 3 days after starting to use ciprofloxacin eye drops for eye redness and purulent discharge that had been diagnosed as bacterial conjunctivitis. He was taking no other new medications. He was afebrile.
The ciprofloxacin drops were stopped. The skin lesions were treated with emollients, topical steroids, and topical mupirocin. Improvement was noted 3 days into the hospitalization, as the lesions started to crust over and dry up and no new lesions were forming. The conjunctivitis improved with topical bacitracin ointment and prednisolone drops.
ACUTE GENERALIZED EXANTHEMATOUS PUSTULOSIS
The differential diagnosis of drug-related acute generalized exanthematous pustulosis includes Stevens-Johnson syndrome, pustular psoriasis, folliculitis, and varicella infection. The characteristic features of the rash and lesions and the temporal relationship between the start of ciprofloxacin eye drops and the development of symptoms, combined with rapid resolution of symptoms within days after discontinuing the drops, accompanied by skin biopsy study showing diffuse spongiosis with scattered eosinophils and a subcorneal pustule, confirmed the diagnosis of AGEP.
Key features of AGEP include numerous small, sterile, nonfollicular pustules on an erythematous background with associated fever and sometimes neutrophilia and eosinophilia.1 It usually begins on the face or in the intertriginous areas and then spreads to the trunk and lower limbs with rare mucosal involvement.1 It can be associated with viral infections, but most reported cases are related to drug reactions.2
Our patient’s case was unusual because AGEP triggered by topical medications is rarely reported, especially with ophthalmic medications.3 Drugs most commonly implicated are antibiotics including penicillins, sulfonamides, and quinolones, but other drugs such as terbinafine, diltiazem, and hydroxychloroquine have also been associated.2
AGEP may present with extensive skin desquamation, as in our patient, sometimes with bullae formation and skin sloughing manifesting as AGEP with overlapping toxic epidermal necrolysis.4
Diagnosis entails a careful review of medications, attention to lesion morphology, compatible disease course, and a high index of suspicion. Treatment is supportive and consists of stopping the offending agent, wound care, and antipyretics. Evidence for the use of steroids is weak.5
TAKE-HOME POINTS
AGEP should be considered in sudden-onset pustular desquamating erythematous rash related to use of a new medication. It is important to be aware that topical and ophthalmic medications are possible triggers. A thorough medication review should be done. Antibiotics are the most commonly implicated medications. An alternative medication should be tried. Treatment is supportive, as the condition is usually self-limiting once the offending medication is discontinued. Rarely, extensive desquamation and bullae formation may occur, which may be a manifestation of overlap features with toxic epidermal necrolysis.
A 66-year-old man presented with burning pain and erythema over the left axilla, and pustules that had ruptured and crusted over. The rash also involved the right axilla, trunk, abdomen, and face.
He said the symptoms had developed 3 days after starting to use ciprofloxacin eye drops for eye redness and purulent discharge that had been diagnosed as bacterial conjunctivitis. He was taking no other new medications. He was afebrile.
The ciprofloxacin drops were stopped. The skin lesions were treated with emollients, topical steroids, and topical mupirocin. Improvement was noted 3 days into the hospitalization, as the lesions started to crust over and dry up and no new lesions were forming. The conjunctivitis improved with topical bacitracin ointment and prednisolone drops.
ACUTE GENERALIZED EXANTHEMATOUS PUSTULOSIS
The differential diagnosis of drug-related acute generalized exanthematous pustulosis includes Stevens-Johnson syndrome, pustular psoriasis, folliculitis, and varicella infection. The characteristic features of the rash and lesions and the temporal relationship between the start of ciprofloxacin eye drops and the development of symptoms, combined with rapid resolution of symptoms within days after discontinuing the drops, accompanied by skin biopsy study showing diffuse spongiosis with scattered eosinophils and a subcorneal pustule, confirmed the diagnosis of AGEP.
Key features of AGEP include numerous small, sterile, nonfollicular pustules on an erythematous background with associated fever and sometimes neutrophilia and eosinophilia.1 It usually begins on the face or in the intertriginous areas and then spreads to the trunk and lower limbs with rare mucosal involvement.1 It can be associated with viral infections, but most reported cases are related to drug reactions.2
Our patient’s case was unusual because AGEP triggered by topical medications is rarely reported, especially with ophthalmic medications.3 Drugs most commonly implicated are antibiotics including penicillins, sulfonamides, and quinolones, but other drugs such as terbinafine, diltiazem, and hydroxychloroquine have also been associated.2
AGEP may present with extensive skin desquamation, as in our patient, sometimes with bullae formation and skin sloughing manifesting as AGEP with overlapping toxic epidermal necrolysis.4
Diagnosis entails a careful review of medications, attention to lesion morphology, compatible disease course, and a high index of suspicion. Treatment is supportive and consists of stopping the offending agent, wound care, and antipyretics. Evidence for the use of steroids is weak.5
TAKE-HOME POINTS
AGEP should be considered in sudden-onset pustular desquamating erythematous rash related to use of a new medication. It is important to be aware that topical and ophthalmic medications are possible triggers. A thorough medication review should be done. Antibiotics are the most commonly implicated medications. An alternative medication should be tried. Treatment is supportive, as the condition is usually self-limiting once the offending medication is discontinued. Rarely, extensive desquamation and bullae formation may occur, which may be a manifestation of overlap features with toxic epidermal necrolysis.
- Speeckaert MM, Speeckaert R, Lambert J, Brochez L. Acute generalized exanthematous pustulosis: an overview of the clinical, immunological and diagnostic concepts. Eur J Dermatol 2010; 20(4):425–433. doi:10.1684/ejd.2010.0932
- Sidoroff A, Dunant A, Viboud C, et al. Risk factors for acute generalized exanthematous pustulosis (AGEP)—results of a multinational case–control study (EuroSCAR). Br J Dermatol 2007; 157(5):989–996. doi:10.1111/j.1365-2133.2007.08156.x
- Beltran C, Vergier B, Doutre MS, Beylot C, Beylot-Barry M. Acute generalized exanthematous pustulosis induced by topical application of Algipan. Ann Dermatol Venereol 2009; 136(10):709–712. French. doi:10.1016/j.annder.2008.10.042
- Peermohamed S, Haber RM. Acute generalized exanthematous pustulosis simulating toxic epidermal necrolysis: a case report and review of the literature. Arch Dermatol 2011; 147(6):697–701. doi:10.1001/archdermatol.2011.147
- Feldmeyer L, Heidemeyer K, Yawalkar N. Acute generalized exanthematous pustulosis: pathogenesis, genetic background, clinical variants and therapy. Int J Mol Sci 2016; 17(8). pii:E1214. doi:10.3390/ijms17081214
- Speeckaert MM, Speeckaert R, Lambert J, Brochez L. Acute generalized exanthematous pustulosis: an overview of the clinical, immunological and diagnostic concepts. Eur J Dermatol 2010; 20(4):425–433. doi:10.1684/ejd.2010.0932
- Sidoroff A, Dunant A, Viboud C, et al. Risk factors for acute generalized exanthematous pustulosis (AGEP)—results of a multinational case–control study (EuroSCAR). Br J Dermatol 2007; 157(5):989–996. doi:10.1111/j.1365-2133.2007.08156.x
- Beltran C, Vergier B, Doutre MS, Beylot C, Beylot-Barry M. Acute generalized exanthematous pustulosis induced by topical application of Algipan. Ann Dermatol Venereol 2009; 136(10):709–712. French. doi:10.1016/j.annder.2008.10.042
- Peermohamed S, Haber RM. Acute generalized exanthematous pustulosis simulating toxic epidermal necrolysis: a case report and review of the literature. Arch Dermatol 2011; 147(6):697–701. doi:10.1001/archdermatol.2011.147
- Feldmeyer L, Heidemeyer K, Yawalkar N. Acute generalized exanthematous pustulosis: pathogenesis, genetic background, clinical variants and therapy. Int J Mol Sci 2016; 17(8). pii:E1214. doi:10.3390/ijms17081214
Certolizumab safety profile varies widely across indications
MADRID – , Andrew Blauvelt, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
He presented a comprehensive analysis of safety data from all 49 clinical trials of the tumor necrosis factor inhibitor for its approved indications. The data set included 11,317 patients who received certolizumab for a collective 21,695 person-years in 27 trials in rheumatoid arthritis patients, 5 in psoriasis, 15 for Crohn’s disease, and one trial each for axial spondyloarthritis and psoriatic arthritis.
“It’s not real-world data, but it is a large group of patients [studied] over many years,” noted Dr. Blauvelt, a dermatologist and president of the Oregon Medical Research Center, Portland.
As a renowned authority on psoriasis, he was part of a multidisciplinary expert panel commissioned by UCB to analyze serious adverse events in the complete clinical trials experience involving the company’s tumor necrosis factor inhibitor certolizumab (Cimzia). The panel included experts from rheumatology, gastroenterology, epidemiology, and other disciplines.
The key takeaway: “When you think about the serious side effects of the drug, you have to think about what the indication is, whether the patients are on systemic corticosteroids, and whether they’re heavy or not,” Dr. Blauvelt said.
Take, for example, the risk of serious infections requiring treatment with intravenous antibiotics. The incidence rates ranged from a low of 1.5 per 100 patient-years in psoriasis patients on certolizumab to a high of 5.97 in those with Crohn’s disease, with rates of 3.44 cases per 100 patient-years among rheumatoid arthritis patients and 1.64-1.67 in those with psoriatic arthritis and ankylosing spondylitis, respectively. Patients with Crohn’s disease were 2.22-fold more likely than were those with rheumatoid arthritis to experience a serious infection during their clinical trial experience on certolizumab. In contrast, psoriasis patients had a 52% relative risk reduction and those with psoriatic arthritis were 31% less likely to develop a serious infection compared with those with rheumatoid arthritis.
The explanation for these highly variable serious infection rates lies in part on the huge differences in the concurrent use of systemic corticosteroids with certolizumab across indications. A mere 3.3% of psoriasis patients were also on steroids, compared with 46.2% of rheumatoid arthritis patients, 50.8% of those with ankylosing spondylitis, and about 25% of the Crohn’s disease and psoriatic arthritis patients, he noted.
Advanced age was independently associated with increased risk of serious infections. Patients aged 65 or older were 1.68-fold more likely to experience this event than were those under age 45. And patients whose disease duration was 10 years or more at baseline had a 1.36-fold increased serious infection risk compared with those who had less than a 1-year-long disease history, independent of which disease they had.
The prevalence of baseline obesity varied by indication. The mean body mass index was 30.1 kg/m2 in the psoriasis patients, 29.8 kg/m2 in those with psoriatic arthritis, lowest at 24 kg/m2 in Crohn’s disease patients, and a bit over 27 kg/m2 in those with rheumatoid arthritis or ankylosing spondylitis.
Obesity alone was not an independent risk factor for serious infection in certolizumab-treated patients; however, the combination of a BMI of 30 kg/m2 or more plus systemic corticosteroid use was associated with a greater risk than with steroids alone.
Based upon a multivariate regression analysis adjusted for age, sex, indication, disease duration, use of methotrexate, and prior use of other TNF inhibitors, the investigators calculated that in patients with Crohn’s disease 16.6% of serious infections in patients on certolizumab were attributable to systemic corticosteroid use.
Risks of major adverse cardiovascular events and cancer on certolizumab
The risk of major adverse cardiovascular events (MACE) while on certolizumab ranged from a high of 0.62 MACE events per 100 patient-years in the rheumatoid arthritis population to a low of 0.1 per 100 patient-years in patients treated for Crohn’s disease or ankylosing spondylitis. Psoriasis and psoriatic arthritis patients had MACE rates of 0.27 and 0.54, respectively.
Obesity was independently associated with increased risk of an acute MI and other MACEs. So was advanced age. No surprises there. The investigators calculated that 16.7% of MACEs in patients on certolizumab were attributable to obesity and another 20.9% were attributable to use of systemic corticosteroids.
The incidence rate for all malignancies, including nonmelanoma skin cancer, ranged from a low of 0.46 cases per 100 patient-years in the psoriatic arthritis cohort on certolizumab to a high of 0.93 in those with rheumatoid arthritis, with rates of 0.68, 0.73, and 0.51 in patients with psoriasis, Crohn’s disease, and ankylosing spondylitis, respectively.
Neither systemic corticosteroids, obesity, disease duration, or prior exposure to a TNF inhibitor was linked to increased risk of cancer in patients on certolizumab. The standout risk factor was age: Patients who were 65 or older at baseline were 11.4-fold more likely to develop cancer during participation in their clinical trial than were those younger than 45. Those who were 45 to 65 years old were 4.3-fold more likely to be diagnosed with a malignancy than were those younger than age 45.
Of note, concomitant use of methotrexate was associated with a statistically significant 28% reduction in malignancy risk.
Dr. Blauvelt reported serving as a consultant to and receiving research funding from UCB, the study sponsor, as well as more than two dozen other pharmaceutical companies.
SOURCE: Blauvelt A. EADV Congress, Abstract FC04.06.
MADRID – , Andrew Blauvelt, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
He presented a comprehensive analysis of safety data from all 49 clinical trials of the tumor necrosis factor inhibitor for its approved indications. The data set included 11,317 patients who received certolizumab for a collective 21,695 person-years in 27 trials in rheumatoid arthritis patients, 5 in psoriasis, 15 for Crohn’s disease, and one trial each for axial spondyloarthritis and psoriatic arthritis.
“It’s not real-world data, but it is a large group of patients [studied] over many years,” noted Dr. Blauvelt, a dermatologist and president of the Oregon Medical Research Center, Portland.
As a renowned authority on psoriasis, he was part of a multidisciplinary expert panel commissioned by UCB to analyze serious adverse events in the complete clinical trials experience involving the company’s tumor necrosis factor inhibitor certolizumab (Cimzia). The panel included experts from rheumatology, gastroenterology, epidemiology, and other disciplines.
The key takeaway: “When you think about the serious side effects of the drug, you have to think about what the indication is, whether the patients are on systemic corticosteroids, and whether they’re heavy or not,” Dr. Blauvelt said.
Take, for example, the risk of serious infections requiring treatment with intravenous antibiotics. The incidence rates ranged from a low of 1.5 per 100 patient-years in psoriasis patients on certolizumab to a high of 5.97 in those with Crohn’s disease, with rates of 3.44 cases per 100 patient-years among rheumatoid arthritis patients and 1.64-1.67 in those with psoriatic arthritis and ankylosing spondylitis, respectively. Patients with Crohn’s disease were 2.22-fold more likely than were those with rheumatoid arthritis to experience a serious infection during their clinical trial experience on certolizumab. In contrast, psoriasis patients had a 52% relative risk reduction and those with psoriatic arthritis were 31% less likely to develop a serious infection compared with those with rheumatoid arthritis.
The explanation for these highly variable serious infection rates lies in part on the huge differences in the concurrent use of systemic corticosteroids with certolizumab across indications. A mere 3.3% of psoriasis patients were also on steroids, compared with 46.2% of rheumatoid arthritis patients, 50.8% of those with ankylosing spondylitis, and about 25% of the Crohn’s disease and psoriatic arthritis patients, he noted.
Advanced age was independently associated with increased risk of serious infections. Patients aged 65 or older were 1.68-fold more likely to experience this event than were those under age 45. And patients whose disease duration was 10 years or more at baseline had a 1.36-fold increased serious infection risk compared with those who had less than a 1-year-long disease history, independent of which disease they had.
The prevalence of baseline obesity varied by indication. The mean body mass index was 30.1 kg/m2 in the psoriasis patients, 29.8 kg/m2 in those with psoriatic arthritis, lowest at 24 kg/m2 in Crohn’s disease patients, and a bit over 27 kg/m2 in those with rheumatoid arthritis or ankylosing spondylitis.
Obesity alone was not an independent risk factor for serious infection in certolizumab-treated patients; however, the combination of a BMI of 30 kg/m2 or more plus systemic corticosteroid use was associated with a greater risk than with steroids alone.
Based upon a multivariate regression analysis adjusted for age, sex, indication, disease duration, use of methotrexate, and prior use of other TNF inhibitors, the investigators calculated that in patients with Crohn’s disease 16.6% of serious infections in patients on certolizumab were attributable to systemic corticosteroid use.
Risks of major adverse cardiovascular events and cancer on certolizumab
The risk of major adverse cardiovascular events (MACE) while on certolizumab ranged from a high of 0.62 MACE events per 100 patient-years in the rheumatoid arthritis population to a low of 0.1 per 100 patient-years in patients treated for Crohn’s disease or ankylosing spondylitis. Psoriasis and psoriatic arthritis patients had MACE rates of 0.27 and 0.54, respectively.
Obesity was independently associated with increased risk of an acute MI and other MACEs. So was advanced age. No surprises there. The investigators calculated that 16.7% of MACEs in patients on certolizumab were attributable to obesity and another 20.9% were attributable to use of systemic corticosteroids.
The incidence rate for all malignancies, including nonmelanoma skin cancer, ranged from a low of 0.46 cases per 100 patient-years in the psoriatic arthritis cohort on certolizumab to a high of 0.93 in those with rheumatoid arthritis, with rates of 0.68, 0.73, and 0.51 in patients with psoriasis, Crohn’s disease, and ankylosing spondylitis, respectively.
Neither systemic corticosteroids, obesity, disease duration, or prior exposure to a TNF inhibitor was linked to increased risk of cancer in patients on certolizumab. The standout risk factor was age: Patients who were 65 or older at baseline were 11.4-fold more likely to develop cancer during participation in their clinical trial than were those younger than 45. Those who were 45 to 65 years old were 4.3-fold more likely to be diagnosed with a malignancy than were those younger than age 45.
Of note, concomitant use of methotrexate was associated with a statistically significant 28% reduction in malignancy risk.
Dr. Blauvelt reported serving as a consultant to and receiving research funding from UCB, the study sponsor, as well as more than two dozen other pharmaceutical companies.
SOURCE: Blauvelt A. EADV Congress, Abstract FC04.06.
MADRID – , Andrew Blauvelt, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
He presented a comprehensive analysis of safety data from all 49 clinical trials of the tumor necrosis factor inhibitor for its approved indications. The data set included 11,317 patients who received certolizumab for a collective 21,695 person-years in 27 trials in rheumatoid arthritis patients, 5 in psoriasis, 15 for Crohn’s disease, and one trial each for axial spondyloarthritis and psoriatic arthritis.
“It’s not real-world data, but it is a large group of patients [studied] over many years,” noted Dr. Blauvelt, a dermatologist and president of the Oregon Medical Research Center, Portland.
As a renowned authority on psoriasis, he was part of a multidisciplinary expert panel commissioned by UCB to analyze serious adverse events in the complete clinical trials experience involving the company’s tumor necrosis factor inhibitor certolizumab (Cimzia). The panel included experts from rheumatology, gastroenterology, epidemiology, and other disciplines.
The key takeaway: “When you think about the serious side effects of the drug, you have to think about what the indication is, whether the patients are on systemic corticosteroids, and whether they’re heavy or not,” Dr. Blauvelt said.
Take, for example, the risk of serious infections requiring treatment with intravenous antibiotics. The incidence rates ranged from a low of 1.5 per 100 patient-years in psoriasis patients on certolizumab to a high of 5.97 in those with Crohn’s disease, with rates of 3.44 cases per 100 patient-years among rheumatoid arthritis patients and 1.64-1.67 in those with psoriatic arthritis and ankylosing spondylitis, respectively. Patients with Crohn’s disease were 2.22-fold more likely than were those with rheumatoid arthritis to experience a serious infection during their clinical trial experience on certolizumab. In contrast, psoriasis patients had a 52% relative risk reduction and those with psoriatic arthritis were 31% less likely to develop a serious infection compared with those with rheumatoid arthritis.
The explanation for these highly variable serious infection rates lies in part on the huge differences in the concurrent use of systemic corticosteroids with certolizumab across indications. A mere 3.3% of psoriasis patients were also on steroids, compared with 46.2% of rheumatoid arthritis patients, 50.8% of those with ankylosing spondylitis, and about 25% of the Crohn’s disease and psoriatic arthritis patients, he noted.
Advanced age was independently associated with increased risk of serious infections. Patients aged 65 or older were 1.68-fold more likely to experience this event than were those under age 45. And patients whose disease duration was 10 years or more at baseline had a 1.36-fold increased serious infection risk compared with those who had less than a 1-year-long disease history, independent of which disease they had.
The prevalence of baseline obesity varied by indication. The mean body mass index was 30.1 kg/m2 in the psoriasis patients, 29.8 kg/m2 in those with psoriatic arthritis, lowest at 24 kg/m2 in Crohn’s disease patients, and a bit over 27 kg/m2 in those with rheumatoid arthritis or ankylosing spondylitis.
Obesity alone was not an independent risk factor for serious infection in certolizumab-treated patients; however, the combination of a BMI of 30 kg/m2 or more plus systemic corticosteroid use was associated with a greater risk than with steroids alone.
Based upon a multivariate regression analysis adjusted for age, sex, indication, disease duration, use of methotrexate, and prior use of other TNF inhibitors, the investigators calculated that in patients with Crohn’s disease 16.6% of serious infections in patients on certolizumab were attributable to systemic corticosteroid use.
Risks of major adverse cardiovascular events and cancer on certolizumab
The risk of major adverse cardiovascular events (MACE) while on certolizumab ranged from a high of 0.62 MACE events per 100 patient-years in the rheumatoid arthritis population to a low of 0.1 per 100 patient-years in patients treated for Crohn’s disease or ankylosing spondylitis. Psoriasis and psoriatic arthritis patients had MACE rates of 0.27 and 0.54, respectively.
Obesity was independently associated with increased risk of an acute MI and other MACEs. So was advanced age. No surprises there. The investigators calculated that 16.7% of MACEs in patients on certolizumab were attributable to obesity and another 20.9% were attributable to use of systemic corticosteroids.
The incidence rate for all malignancies, including nonmelanoma skin cancer, ranged from a low of 0.46 cases per 100 patient-years in the psoriatic arthritis cohort on certolizumab to a high of 0.93 in those with rheumatoid arthritis, with rates of 0.68, 0.73, and 0.51 in patients with psoriasis, Crohn’s disease, and ankylosing spondylitis, respectively.
Neither systemic corticosteroids, obesity, disease duration, or prior exposure to a TNF inhibitor was linked to increased risk of cancer in patients on certolizumab. The standout risk factor was age: Patients who were 65 or older at baseline were 11.4-fold more likely to develop cancer during participation in their clinical trial than were those younger than 45. Those who were 45 to 65 years old were 4.3-fold more likely to be diagnosed with a malignancy than were those younger than age 45.
Of note, concomitant use of methotrexate was associated with a statistically significant 28% reduction in malignancy risk.
Dr. Blauvelt reported serving as a consultant to and receiving research funding from UCB, the study sponsor, as well as more than two dozen other pharmaceutical companies.
SOURCE: Blauvelt A. EADV Congress, Abstract FC04.06.
REPORTING FROM THE EADV CONGRESS
Combo elicits lasting responses in metastatic melanoma
NATIONAL HARBOR, MD. – The combination of bempegaldesleukin and nivolumab produced durable responses in a phase 1/2 trial of patients with previously untreated metastatic melanoma.
The overall response rate was 53%, and most responders were still in response at a median follow-up of about 19 months. The median progression-free survival was not reached, and the combination was considered well tolerated.
Adi Diab, MD, of the University of Texas MD Anderson Cancer Center, Houston, presented these results from the PIVOT-02 study at the annual meeting of the Society for Immunotherapy of Cancer.
Dr. Diab explained that bempegaldesleukin (bempeg) is a CD122-preferential interleukin-2 pathway agonist, and earlier results from the PIVOT-02 trial showed that adding bempeg to nivolumab can convert baseline tumors from programmed death–ligand 1 (PD-L1) negative to PD-L1 positive (SITC 2018, Abstract O4).
Dr. Diab presented updated results from PIVOT-02 (NCT02983045) in 41 patients with metastatic melanoma who received bempeg plus nivolumab as first-line treatment. The patients had a median age of 63 years (range, 22-80 years) at baseline, and 58.5% were male. Most patients (58.5%) were PD-L1 positive, although PD-L1 status was unknown in 7.3% of patients.
Patients received bempeg at 0.006 mg/kg and nivolumab at 360 mg every 3 weeks. They received a median of nine cycles (range, 1-34), and the median follow-up was 18.6 months.
Efficacy
In the 38 patients who were evaluable for efficacy, the overall response rate was 53% (n = 20), and the complete response rate was 34% (n = 13). The median time to response was 2.0 months, and the median time to complete response was 7.9 months.
Dr. Diab noted that responses were seen regardless of PD-L1 expression at baseline. The response rate was 39% among PD-L1-negative patients, 64% among PD-L1-positive patients, and 33% among patients whose PD-L1 status was unknown.
Dr. Diab also pointed out that responses were durable and deepened over time. The median duration of response was not reached, and 17 of the 20 responders had ongoing responses at last follow-up. The median progression-free survival has not been reached.
Safety
“This combination is safe and tolerable, there’s no overlapping immune-related adverse events, and the most common side effects are grade 1/2 flu-like symptoms,” Dr. Diab said.
The most common grade 1/2 treatment-related adverse events (AEs) were flu-like symptoms (80.5%), rash (70.7%), fatigue (65.9%), pruritus (48.8%), nausea (46.3%), arthralgia (43.9%), decreased appetite (36.6%), and myalgia (36.6%).
Dr. Diab noted that cytokine-related AEs (flu-like symptoms, rash, and pruritus) were easily managed with NSAIDs; decreased with subsequent cycles of treatment; and did not necessitate dose delays, reductions, or discontinuations.
Grade 3/4 treatment-related AEs included two cases of acute kidney injury, two cases of atrial fibrillation, one case of dizziness, one case of dyspnea, one case of hypoxia, one case of hyperglycemia, and one case of hypernatremia.
Five patients discontinued treatment because of related AEs, including cerebrovascular accident, peripheral edema, blood creatinine increase, malaise, and pharyngitis. There were no treatment-related deaths.
Dr. Diab said these results were used to support the recent breakthrough therapy designation granted to bempeg in combination with nivolumab. The results have also prompted a phase 3 trial in which researchers are comparing the combination with nivolumab alone (NCT03635983).
The phase 1/2 trial is sponsored by Nektar Therapeutics in collaboration with Bristol-Myers Squibb. Dr. Diab reported relationships with Nektar, Celgene, CureVac, Idera, and Pfizer.
SOURCE: Diab A et al. SITC 2019, Abstract O35.
NATIONAL HARBOR, MD. – The combination of bempegaldesleukin and nivolumab produced durable responses in a phase 1/2 trial of patients with previously untreated metastatic melanoma.
The overall response rate was 53%, and most responders were still in response at a median follow-up of about 19 months. The median progression-free survival was not reached, and the combination was considered well tolerated.
Adi Diab, MD, of the University of Texas MD Anderson Cancer Center, Houston, presented these results from the PIVOT-02 study at the annual meeting of the Society for Immunotherapy of Cancer.
Dr. Diab explained that bempegaldesleukin (bempeg) is a CD122-preferential interleukin-2 pathway agonist, and earlier results from the PIVOT-02 trial showed that adding bempeg to nivolumab can convert baseline tumors from programmed death–ligand 1 (PD-L1) negative to PD-L1 positive (SITC 2018, Abstract O4).
Dr. Diab presented updated results from PIVOT-02 (NCT02983045) in 41 patients with metastatic melanoma who received bempeg plus nivolumab as first-line treatment. The patients had a median age of 63 years (range, 22-80 years) at baseline, and 58.5% were male. Most patients (58.5%) were PD-L1 positive, although PD-L1 status was unknown in 7.3% of patients.
Patients received bempeg at 0.006 mg/kg and nivolumab at 360 mg every 3 weeks. They received a median of nine cycles (range, 1-34), and the median follow-up was 18.6 months.
Efficacy
In the 38 patients who were evaluable for efficacy, the overall response rate was 53% (n = 20), and the complete response rate was 34% (n = 13). The median time to response was 2.0 months, and the median time to complete response was 7.9 months.
Dr. Diab noted that responses were seen regardless of PD-L1 expression at baseline. The response rate was 39% among PD-L1-negative patients, 64% among PD-L1-positive patients, and 33% among patients whose PD-L1 status was unknown.
Dr. Diab also pointed out that responses were durable and deepened over time. The median duration of response was not reached, and 17 of the 20 responders had ongoing responses at last follow-up. The median progression-free survival has not been reached.
Safety
“This combination is safe and tolerable, there’s no overlapping immune-related adverse events, and the most common side effects are grade 1/2 flu-like symptoms,” Dr. Diab said.
The most common grade 1/2 treatment-related adverse events (AEs) were flu-like symptoms (80.5%), rash (70.7%), fatigue (65.9%), pruritus (48.8%), nausea (46.3%), arthralgia (43.9%), decreased appetite (36.6%), and myalgia (36.6%).
Dr. Diab noted that cytokine-related AEs (flu-like symptoms, rash, and pruritus) were easily managed with NSAIDs; decreased with subsequent cycles of treatment; and did not necessitate dose delays, reductions, or discontinuations.
Grade 3/4 treatment-related AEs included two cases of acute kidney injury, two cases of atrial fibrillation, one case of dizziness, one case of dyspnea, one case of hypoxia, one case of hyperglycemia, and one case of hypernatremia.
Five patients discontinued treatment because of related AEs, including cerebrovascular accident, peripheral edema, blood creatinine increase, malaise, and pharyngitis. There were no treatment-related deaths.
Dr. Diab said these results were used to support the recent breakthrough therapy designation granted to bempeg in combination with nivolumab. The results have also prompted a phase 3 trial in which researchers are comparing the combination with nivolumab alone (NCT03635983).
The phase 1/2 trial is sponsored by Nektar Therapeutics in collaboration with Bristol-Myers Squibb. Dr. Diab reported relationships with Nektar, Celgene, CureVac, Idera, and Pfizer.
SOURCE: Diab A et al. SITC 2019, Abstract O35.
NATIONAL HARBOR, MD. – The combination of bempegaldesleukin and nivolumab produced durable responses in a phase 1/2 trial of patients with previously untreated metastatic melanoma.
The overall response rate was 53%, and most responders were still in response at a median follow-up of about 19 months. The median progression-free survival was not reached, and the combination was considered well tolerated.
Adi Diab, MD, of the University of Texas MD Anderson Cancer Center, Houston, presented these results from the PIVOT-02 study at the annual meeting of the Society for Immunotherapy of Cancer.
Dr. Diab explained that bempegaldesleukin (bempeg) is a CD122-preferential interleukin-2 pathway agonist, and earlier results from the PIVOT-02 trial showed that adding bempeg to nivolumab can convert baseline tumors from programmed death–ligand 1 (PD-L1) negative to PD-L1 positive (SITC 2018, Abstract O4).
Dr. Diab presented updated results from PIVOT-02 (NCT02983045) in 41 patients with metastatic melanoma who received bempeg plus nivolumab as first-line treatment. The patients had a median age of 63 years (range, 22-80 years) at baseline, and 58.5% were male. Most patients (58.5%) were PD-L1 positive, although PD-L1 status was unknown in 7.3% of patients.
Patients received bempeg at 0.006 mg/kg and nivolumab at 360 mg every 3 weeks. They received a median of nine cycles (range, 1-34), and the median follow-up was 18.6 months.
Efficacy
In the 38 patients who were evaluable for efficacy, the overall response rate was 53% (n = 20), and the complete response rate was 34% (n = 13). The median time to response was 2.0 months, and the median time to complete response was 7.9 months.
Dr. Diab noted that responses were seen regardless of PD-L1 expression at baseline. The response rate was 39% among PD-L1-negative patients, 64% among PD-L1-positive patients, and 33% among patients whose PD-L1 status was unknown.
Dr. Diab also pointed out that responses were durable and deepened over time. The median duration of response was not reached, and 17 of the 20 responders had ongoing responses at last follow-up. The median progression-free survival has not been reached.
Safety
“This combination is safe and tolerable, there’s no overlapping immune-related adverse events, and the most common side effects are grade 1/2 flu-like symptoms,” Dr. Diab said.
The most common grade 1/2 treatment-related adverse events (AEs) were flu-like symptoms (80.5%), rash (70.7%), fatigue (65.9%), pruritus (48.8%), nausea (46.3%), arthralgia (43.9%), decreased appetite (36.6%), and myalgia (36.6%).
Dr. Diab noted that cytokine-related AEs (flu-like symptoms, rash, and pruritus) were easily managed with NSAIDs; decreased with subsequent cycles of treatment; and did not necessitate dose delays, reductions, or discontinuations.
Grade 3/4 treatment-related AEs included two cases of acute kidney injury, two cases of atrial fibrillation, one case of dizziness, one case of dyspnea, one case of hypoxia, one case of hyperglycemia, and one case of hypernatremia.
Five patients discontinued treatment because of related AEs, including cerebrovascular accident, peripheral edema, blood creatinine increase, malaise, and pharyngitis. There were no treatment-related deaths.
Dr. Diab said these results were used to support the recent breakthrough therapy designation granted to bempeg in combination with nivolumab. The results have also prompted a phase 3 trial in which researchers are comparing the combination with nivolumab alone (NCT03635983).
The phase 1/2 trial is sponsored by Nektar Therapeutics in collaboration with Bristol-Myers Squibb. Dr. Diab reported relationships with Nektar, Celgene, CureVac, Idera, and Pfizer.
SOURCE: Diab A et al. SITC 2019, Abstract O35.
REPORTING FROM SITC 2019
Omalizumab proves effective for severe pediatric atopic dermatitis
A new study has found that omalizumab (Xolair) reduced severity and improved quality of life in pediatric patients with severe atopic dermatitis.
“Future work with an even larger sample size, a longer duration, and higher-affinity versions of omalizumab would clarify the precise role of anti-IgE therapy and its ideal target population,” wrote Susan Chan, MD, of Guy’s and St. Thomas’ NHS Foundation Trust in London and her coauthors. The study was published in JAMA Pediatrics.
To determine the benefits of omalizumab in reducing immunoglobulin E levels and thereby treating severe childhood eczema, the researchers launched the Atopic Dermatitis Anti-IgE Pediatric Trial (ADAPT). This randomized clinical trial recruited 62 patients between the ages of 4 and 19 years with severe eczema, which was defined as a score over 40 on the objective Scoring Atopic Dermatitis (SCORAD) index. They received 24 weeks of treatment with either omalizumab (n = 30) or placebo (n = 32) followed by 24 weeks of follow-up. Participants had a mean age of 10.3 years.
After 24 weeks, the adjusted mean difference in objective SCORAD index between the two groups was –6.9 (95% confidence interval, –12.2 to –1.5; P = .01) and significantly favored omalizumab therapy. The adjusted mean difference for the Eczema Area and Severity Index (–6.7; 95% CI, –13.2 to –0.1) also favored omalizumab. In regard to quality of life, after 24 weeks the Children’s Dermatology Life Quality Index/Dermatology Life Quality Index favored the omalizumab group with an adjusted mean difference of –3.5 (95% CI, –6.4 to –0.5).
In an accompanying editorial, Ann Chen Wu, MD, of Harvard Medical School in Boston noted that the results of the study from Chan et al. were promising but “more questions need to be answered before the drug can be used to treat atopic dermatitis in clinical practice” (JAMA Pediatr. 2019 Nov. 25. doi: 10.1001/jamapediatrics.2019.4509).
Her initial concern was price; she acknowledged that “omalizumab is a costly intervention” but said atopic dermatitis is also costly, raising the question as to whether the high costs of both justify treatment. In addition, omalizumab as treatment can come with both benefits and harms. Severe atopic dermatitis can decrease quality of life and, though omalizumab appears to be safe, there are adverse effects and logistical burdens to overcome.
More than anything, she recognized the need to prioritize, wondering what level of atopic dermatitis patients would truly benefit from this level of treatment. “Is using a $100,000-per-year medication for an itchy condition an overtreatment,” she asked, “or a lifesaver?”
The study was funded by the National Institute for Health Research Efficacy and Mechanism Evaluation Programme and Guy’s and St. Thomas’ Charity. The authors had numerous financial disclosures, including receiving grants from the NIHR EME Programme and Guy’s and St. Thomas’ Charity along with active and placebo drugs from Novartis for use in the study. Dr. Wu reported receiving a grant from GlaxoSmithKline.
SOURCE: Chan S et al. JAMA Pediatr. 2019 Nov 25. doi: 10.1001/jamapediatrics.2019.4476.
A new study has found that omalizumab (Xolair) reduced severity and improved quality of life in pediatric patients with severe atopic dermatitis.
“Future work with an even larger sample size, a longer duration, and higher-affinity versions of omalizumab would clarify the precise role of anti-IgE therapy and its ideal target population,” wrote Susan Chan, MD, of Guy’s and St. Thomas’ NHS Foundation Trust in London and her coauthors. The study was published in JAMA Pediatrics.
To determine the benefits of omalizumab in reducing immunoglobulin E levels and thereby treating severe childhood eczema, the researchers launched the Atopic Dermatitis Anti-IgE Pediatric Trial (ADAPT). This randomized clinical trial recruited 62 patients between the ages of 4 and 19 years with severe eczema, which was defined as a score over 40 on the objective Scoring Atopic Dermatitis (SCORAD) index. They received 24 weeks of treatment with either omalizumab (n = 30) or placebo (n = 32) followed by 24 weeks of follow-up. Participants had a mean age of 10.3 years.
After 24 weeks, the adjusted mean difference in objective SCORAD index between the two groups was –6.9 (95% confidence interval, –12.2 to –1.5; P = .01) and significantly favored omalizumab therapy. The adjusted mean difference for the Eczema Area and Severity Index (–6.7; 95% CI, –13.2 to –0.1) also favored omalizumab. In regard to quality of life, after 24 weeks the Children’s Dermatology Life Quality Index/Dermatology Life Quality Index favored the omalizumab group with an adjusted mean difference of –3.5 (95% CI, –6.4 to –0.5).
In an accompanying editorial, Ann Chen Wu, MD, of Harvard Medical School in Boston noted that the results of the study from Chan et al. were promising but “more questions need to be answered before the drug can be used to treat atopic dermatitis in clinical practice” (JAMA Pediatr. 2019 Nov. 25. doi: 10.1001/jamapediatrics.2019.4509).
Her initial concern was price; she acknowledged that “omalizumab is a costly intervention” but said atopic dermatitis is also costly, raising the question as to whether the high costs of both justify treatment. In addition, omalizumab as treatment can come with both benefits and harms. Severe atopic dermatitis can decrease quality of life and, though omalizumab appears to be safe, there are adverse effects and logistical burdens to overcome.
More than anything, she recognized the need to prioritize, wondering what level of atopic dermatitis patients would truly benefit from this level of treatment. “Is using a $100,000-per-year medication for an itchy condition an overtreatment,” she asked, “or a lifesaver?”
The study was funded by the National Institute for Health Research Efficacy and Mechanism Evaluation Programme and Guy’s and St. Thomas’ Charity. The authors had numerous financial disclosures, including receiving grants from the NIHR EME Programme and Guy’s and St. Thomas’ Charity along with active and placebo drugs from Novartis for use in the study. Dr. Wu reported receiving a grant from GlaxoSmithKline.
SOURCE: Chan S et al. JAMA Pediatr. 2019 Nov 25. doi: 10.1001/jamapediatrics.2019.4476.
A new study has found that omalizumab (Xolair) reduced severity and improved quality of life in pediatric patients with severe atopic dermatitis.
“Future work with an even larger sample size, a longer duration, and higher-affinity versions of omalizumab would clarify the precise role of anti-IgE therapy and its ideal target population,” wrote Susan Chan, MD, of Guy’s and St. Thomas’ NHS Foundation Trust in London and her coauthors. The study was published in JAMA Pediatrics.
To determine the benefits of omalizumab in reducing immunoglobulin E levels and thereby treating severe childhood eczema, the researchers launched the Atopic Dermatitis Anti-IgE Pediatric Trial (ADAPT). This randomized clinical trial recruited 62 patients between the ages of 4 and 19 years with severe eczema, which was defined as a score over 40 on the objective Scoring Atopic Dermatitis (SCORAD) index. They received 24 weeks of treatment with either omalizumab (n = 30) or placebo (n = 32) followed by 24 weeks of follow-up. Participants had a mean age of 10.3 years.
After 24 weeks, the adjusted mean difference in objective SCORAD index between the two groups was –6.9 (95% confidence interval, –12.2 to –1.5; P = .01) and significantly favored omalizumab therapy. The adjusted mean difference for the Eczema Area and Severity Index (–6.7; 95% CI, –13.2 to –0.1) also favored omalizumab. In regard to quality of life, after 24 weeks the Children’s Dermatology Life Quality Index/Dermatology Life Quality Index favored the omalizumab group with an adjusted mean difference of –3.5 (95% CI, –6.4 to –0.5).
In an accompanying editorial, Ann Chen Wu, MD, of Harvard Medical School in Boston noted that the results of the study from Chan et al. were promising but “more questions need to be answered before the drug can be used to treat atopic dermatitis in clinical practice” (JAMA Pediatr. 2019 Nov. 25. doi: 10.1001/jamapediatrics.2019.4509).
Her initial concern was price; she acknowledged that “omalizumab is a costly intervention” but said atopic dermatitis is also costly, raising the question as to whether the high costs of both justify treatment. In addition, omalizumab as treatment can come with both benefits and harms. Severe atopic dermatitis can decrease quality of life and, though omalizumab appears to be safe, there are adverse effects and logistical burdens to overcome.
More than anything, she recognized the need to prioritize, wondering what level of atopic dermatitis patients would truly benefit from this level of treatment. “Is using a $100,000-per-year medication for an itchy condition an overtreatment,” she asked, “or a lifesaver?”
The study was funded by the National Institute for Health Research Efficacy and Mechanism Evaluation Programme and Guy’s and St. Thomas’ Charity. The authors had numerous financial disclosures, including receiving grants from the NIHR EME Programme and Guy’s and St. Thomas’ Charity along with active and placebo drugs from Novartis for use in the study. Dr. Wu reported receiving a grant from GlaxoSmithKline.
SOURCE: Chan S et al. JAMA Pediatr. 2019 Nov 25. doi: 10.1001/jamapediatrics.2019.4476.
FROM JAMA PEDIATRICS
Naturopaths emphasize role of diet in atopic dermatitis
, based on data from a small survey of the two.
Data from previous studies show that more than half of patients with AD have used complementary and alternative medicine in addition to allopathic care, but providers may be unaware of each other’s treatment approaches and confuse patients, wrote Julie Dhossche, MD, of Oregon Health & Science University, Portland, and her colleagues.
In a study published in Pediatric Dermatology, the researchers assessed results of an 11-question, free-text survey of 30 allopathic providers and 21 naturopathic providers about AD. The survey included questions on patient education and evaluation, skin care, and treatment.
Overall, both allopathic and naturopathic providers recommended skin care protocols involving moisturization and “soak and seal” bathing. However, allopathic providers were more likely to prescribe topical corticosteroids for mild to moderate disease (100% vs. 19%), followed by phototherapy and systemic treatments in more severe cases. Naturopathic providers were more likely than allopathic providers to choose topical botanicals, oils, or probiotics (52% vs. 0%) for mild to moderate disease, as well stress relief and acupuncture. Naturopathic providers favored topical corticosteroids and referrals to dermatologists for second- or third-line treatment.
Of note, 85% of naturopathic providers said they thought diet had a probable or definite role in AD, compared with 3% of allopathic providers.
In addition, naturopathic providers differed in their response to an optional question on the use of additional education about food and diet. A total of 11 of 19 naturopathic providers (58%) recommended dietary changes, including “remove potential food allergens/reduce sugar” and “emphasize anti-inflammatory diet,” the researchers said.
“Confusion regarding the role of food in AD management is a common source of frustration for patients, and perhaps a consensus statement from both fields regarding the role of food allergy in AD management could be aspired toward in the name of reducing patient confusion,” they wrote.
The study findings were limited by several factors, including the small sample size and self-selection bias, as well as the subjective nature of an open-ended survey, the researchers noted. However, the results provide evidence of differences in treatment approaches between allopathic and naturopathic providers and suggest that “respectful collaboration between allopathic and naturopathic providers will help practitioners find common ground, decrease patient confusion, and improve patient outcomes,” they concluded.
The researchers had no financial conflicts to disclose.
SOURCE: Dhossche J et al. Ped Dermatol. 2019 Nov 19. doi: 10.1111/pde.14036.
, based on data from a small survey of the two.
Data from previous studies show that more than half of patients with AD have used complementary and alternative medicine in addition to allopathic care, but providers may be unaware of each other’s treatment approaches and confuse patients, wrote Julie Dhossche, MD, of Oregon Health & Science University, Portland, and her colleagues.
In a study published in Pediatric Dermatology, the researchers assessed results of an 11-question, free-text survey of 30 allopathic providers and 21 naturopathic providers about AD. The survey included questions on patient education and evaluation, skin care, and treatment.
Overall, both allopathic and naturopathic providers recommended skin care protocols involving moisturization and “soak and seal” bathing. However, allopathic providers were more likely to prescribe topical corticosteroids for mild to moderate disease (100% vs. 19%), followed by phototherapy and systemic treatments in more severe cases. Naturopathic providers were more likely than allopathic providers to choose topical botanicals, oils, or probiotics (52% vs. 0%) for mild to moderate disease, as well stress relief and acupuncture. Naturopathic providers favored topical corticosteroids and referrals to dermatologists for second- or third-line treatment.
Of note, 85% of naturopathic providers said they thought diet had a probable or definite role in AD, compared with 3% of allopathic providers.
In addition, naturopathic providers differed in their response to an optional question on the use of additional education about food and diet. A total of 11 of 19 naturopathic providers (58%) recommended dietary changes, including “remove potential food allergens/reduce sugar” and “emphasize anti-inflammatory diet,” the researchers said.
“Confusion regarding the role of food in AD management is a common source of frustration for patients, and perhaps a consensus statement from both fields regarding the role of food allergy in AD management could be aspired toward in the name of reducing patient confusion,” they wrote.
The study findings were limited by several factors, including the small sample size and self-selection bias, as well as the subjective nature of an open-ended survey, the researchers noted. However, the results provide evidence of differences in treatment approaches between allopathic and naturopathic providers and suggest that “respectful collaboration between allopathic and naturopathic providers will help practitioners find common ground, decrease patient confusion, and improve patient outcomes,” they concluded.
The researchers had no financial conflicts to disclose.
SOURCE: Dhossche J et al. Ped Dermatol. 2019 Nov 19. doi: 10.1111/pde.14036.
, based on data from a small survey of the two.
Data from previous studies show that more than half of patients with AD have used complementary and alternative medicine in addition to allopathic care, but providers may be unaware of each other’s treatment approaches and confuse patients, wrote Julie Dhossche, MD, of Oregon Health & Science University, Portland, and her colleagues.
In a study published in Pediatric Dermatology, the researchers assessed results of an 11-question, free-text survey of 30 allopathic providers and 21 naturopathic providers about AD. The survey included questions on patient education and evaluation, skin care, and treatment.
Overall, both allopathic and naturopathic providers recommended skin care protocols involving moisturization and “soak and seal” bathing. However, allopathic providers were more likely to prescribe topical corticosteroids for mild to moderate disease (100% vs. 19%), followed by phototherapy and systemic treatments in more severe cases. Naturopathic providers were more likely than allopathic providers to choose topical botanicals, oils, or probiotics (52% vs. 0%) for mild to moderate disease, as well stress relief and acupuncture. Naturopathic providers favored topical corticosteroids and referrals to dermatologists for second- or third-line treatment.
Of note, 85% of naturopathic providers said they thought diet had a probable or definite role in AD, compared with 3% of allopathic providers.
In addition, naturopathic providers differed in their response to an optional question on the use of additional education about food and diet. A total of 11 of 19 naturopathic providers (58%) recommended dietary changes, including “remove potential food allergens/reduce sugar” and “emphasize anti-inflammatory diet,” the researchers said.
“Confusion regarding the role of food in AD management is a common source of frustration for patients, and perhaps a consensus statement from both fields regarding the role of food allergy in AD management could be aspired toward in the name of reducing patient confusion,” they wrote.
The study findings were limited by several factors, including the small sample size and self-selection bias, as well as the subjective nature of an open-ended survey, the researchers noted. However, the results provide evidence of differences in treatment approaches between allopathic and naturopathic providers and suggest that “respectful collaboration between allopathic and naturopathic providers will help practitioners find common ground, decrease patient confusion, and improve patient outcomes,” they concluded.
The researchers had no financial conflicts to disclose.
SOURCE: Dhossche J et al. Ped Dermatol. 2019 Nov 19. doi: 10.1111/pde.14036.
FROM PEDIATRIC DERMATOLOGY
Atopic dermatitis acts differently in certain populations
LAS VEGAS – Eczema is eczema is eczema, right? Maybe not. “Atopic dermatitis might not be one disease,” a dermatologist told colleagues, and treatments may need to be adjusted to reflect the age and ethnicity of patients.
More research is needed, Kenneth B. Gordon, MD, chair and professor of dermatology at the Medical College of Wisconsin, Milwaukee, said during a presentation at Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar. “We’re probably just on the tip of the iceberg of understanding the physiology of atopic dermatitis. Hopefully, it will lead to the therapeutic advances we’ve seen in psoriasis.”
As Dr. Gordon explained, there’s a , he said, “and our medicines aren’t well understood.”
As for the disease itself, he said, “you might hear a renowned [expert] say, ‘This is how it works,’ and another say, ‘This is absolutely not how it works.’ ” One camp focused on the skin barrier, he said, while another camp highlighted inflammation in AD.
“Both the barrier and inflammation are important,” he said. “There are multiple cell types and cytokines that are important, but we don’t know yet the relative importance of them all. You have this cytokine soup, and we’re still trying to figure out the driving forces.”
What is clear, Dr. Gordon said, is that AD acts differently in certain patient populations. It’s not the same in pediatric versus adult patients, he said, and it’s not the same in white versus black versus Asian patients. Research, for example, suggests that Th2, Th22, and Th17 pathways appear to be important in pediatric AD, but not Th1, he said. In contrast, the Th1 pathway plays a role in white adults – but not in black adults
Different cytokines appear to play different roles in these populations, he said. “One of the key things moving forward is going to be figuring out which patients you apply these medications to,” he noted.
Dr. Gordon has multiple disclosures including honoraria or research support from Abbvie, Lilly, Novartis, Pfizer, UCB, and others. SDEF and this news organization are owned by the same parent company.
LAS VEGAS – Eczema is eczema is eczema, right? Maybe not. “Atopic dermatitis might not be one disease,” a dermatologist told colleagues, and treatments may need to be adjusted to reflect the age and ethnicity of patients.
More research is needed, Kenneth B. Gordon, MD, chair and professor of dermatology at the Medical College of Wisconsin, Milwaukee, said during a presentation at Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar. “We’re probably just on the tip of the iceberg of understanding the physiology of atopic dermatitis. Hopefully, it will lead to the therapeutic advances we’ve seen in psoriasis.”
As Dr. Gordon explained, there’s a , he said, “and our medicines aren’t well understood.”
As for the disease itself, he said, “you might hear a renowned [expert] say, ‘This is how it works,’ and another say, ‘This is absolutely not how it works.’ ” One camp focused on the skin barrier, he said, while another camp highlighted inflammation in AD.
“Both the barrier and inflammation are important,” he said. “There are multiple cell types and cytokines that are important, but we don’t know yet the relative importance of them all. You have this cytokine soup, and we’re still trying to figure out the driving forces.”
What is clear, Dr. Gordon said, is that AD acts differently in certain patient populations. It’s not the same in pediatric versus adult patients, he said, and it’s not the same in white versus black versus Asian patients. Research, for example, suggests that Th2, Th22, and Th17 pathways appear to be important in pediatric AD, but not Th1, he said. In contrast, the Th1 pathway plays a role in white adults – but not in black adults
Different cytokines appear to play different roles in these populations, he said. “One of the key things moving forward is going to be figuring out which patients you apply these medications to,” he noted.
Dr. Gordon has multiple disclosures including honoraria or research support from Abbvie, Lilly, Novartis, Pfizer, UCB, and others. SDEF and this news organization are owned by the same parent company.
LAS VEGAS – Eczema is eczema is eczema, right? Maybe not. “Atopic dermatitis might not be one disease,” a dermatologist told colleagues, and treatments may need to be adjusted to reflect the age and ethnicity of patients.
More research is needed, Kenneth B. Gordon, MD, chair and professor of dermatology at the Medical College of Wisconsin, Milwaukee, said during a presentation at Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar. “We’re probably just on the tip of the iceberg of understanding the physiology of atopic dermatitis. Hopefully, it will lead to the therapeutic advances we’ve seen in psoriasis.”
As Dr. Gordon explained, there’s a , he said, “and our medicines aren’t well understood.”
As for the disease itself, he said, “you might hear a renowned [expert] say, ‘This is how it works,’ and another say, ‘This is absolutely not how it works.’ ” One camp focused on the skin barrier, he said, while another camp highlighted inflammation in AD.
“Both the barrier and inflammation are important,” he said. “There are multiple cell types and cytokines that are important, but we don’t know yet the relative importance of them all. You have this cytokine soup, and we’re still trying to figure out the driving forces.”
What is clear, Dr. Gordon said, is that AD acts differently in certain patient populations. It’s not the same in pediatric versus adult patients, he said, and it’s not the same in white versus black versus Asian patients. Research, for example, suggests that Th2, Th22, and Th17 pathways appear to be important in pediatric AD, but not Th1, he said. In contrast, the Th1 pathway plays a role in white adults – but not in black adults
Different cytokines appear to play different roles in these populations, he said. “One of the key things moving forward is going to be figuring out which patients you apply these medications to,” he noted.
Dr. Gordon has multiple disclosures including honoraria or research support from Abbvie, Lilly, Novartis, Pfizer, UCB, and others. SDEF and this news organization are owned by the same parent company.
REPORTING FROM SDEF LAS VEGAS DERMATOLOGY SEMINAR
Oral baricitinib performs well in phase 3 for atopic dermatitis
MADRID – Adding the oral Janus kinase (JAK) inhibitor baricitinib to standard atopic dermatitis therapy with low- and midpotency topical corticosteroids markedly improved disease severity and key patient-reported outcomes, compared with topical corticosteroids alone, in the phase 3, randomized, double-blind BREEZE-AD7 trial, Kristian Reich, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
in the phase 3 BREEZE-AD1 and -AD2 trials. But BREEZE-AD7 further advances the field because it’s the first phase 3 study testing the efficacy of a JAK inhibitor in combination with low- and midpotency topical steroids.
“I think this study is important because it looks into the situation that’s more like what happens in the real world, which is, as with dupilumab and other drugs, we use the systemic agent in combination with topical therapies and, in particular, with topical corticosteroids,” commented Dr. Reich, professor of dermatology at University Medical Center, Hamburg, and medical director at SCIderm, a scientific research company.
“This is what I think we can expect from existing and upcoming systemic therapies in atopic dermatitis: We will use them in combination with topical corticosteroids, and hopefully this will allow patients to dramatically reduce the concomitant use of topical corticosteroids, as shown here in BREEZE-AD7,” he added.
BREEZE-AD7 was a 16-week study that included 329 adults with moderate or severe atopic dermatitis who were randomized to low- and midpotency topical corticosteroids plus either baricitinib at 2 mg once daily, baricitinib at 4 mg once daily, or placebo. The group’s mean baseline Eczema Area and Severity Index (EASI) score was 29. Overall, 45% of participants had a baseline Investigator’s Global Assessment (IGA) of disease severity of 4 on a 0-4 scale.
The primary endpoint was achievement of an IGA of 0 or 1, meaning clear or almost clear, along with at least a 2-point IGA improvement from baseline at week 16. This was accomplished in 30.6% of those on 4 mg/day of baricitinib, 23.9% of patients in the 2-mg group, and 14.7% of controls.
The 4-mg dose of baricitinib was statistically superior to placebo; the 2-mg dose was not. However, Dr. Reich indicated he was untroubled by this because the primary endpoint was set at a high bar, and both doses of baricitinib proved to be significantly better than topical steroids plus placebo in terms of EASI 75 response rates, as well as reductions in itch, skin pain, and sleep problems, which aren’t captured in EASI scores (see graphic).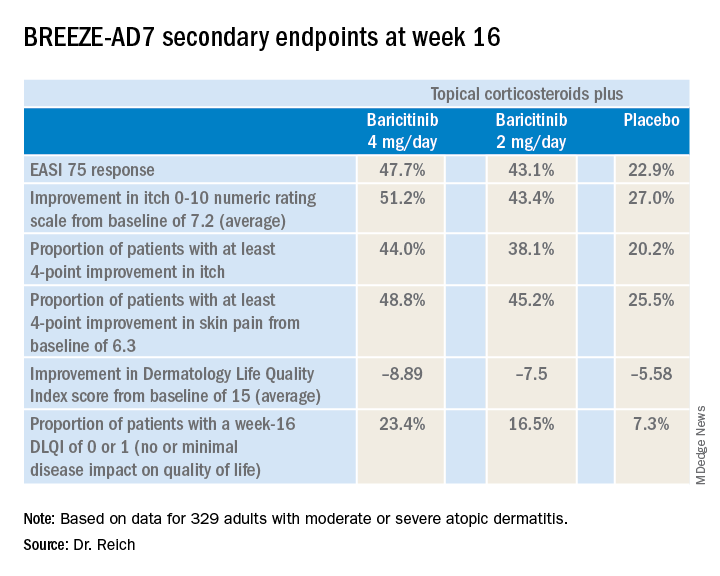
“One of my big learnings from this year’s EADV is that we have to rethink the dimensions of atopic dermatitis. I think we have underestimated the relevance of important symptoms such as itch, the impact atopic dermatitis has on pain, and the effect it has on sleeping problems,” the dermatologist said. “My feeling is that baricitinib is strongest in reducing itch, improving sleep, and reducing pain, but it also has good effects on the clinical signs of atopic dermatitis.”
The baricitinib-treated patients’ rapidity of improvement in the various endpoints was particularly impressive. Both doses of the JAK 1/2 inhibitor showed significant separation from the control group in the first week, and the majority of improvement occurred by week 4.
A key finding was that patients on baricitinib at 2 mg/day and 4 mg/day used a mean total of 162 g and 137 g of midpotency topical steroids, respectively, during the 16 weeks, compared with 225 g in the control group. The higher-dose baricitinib group was topical corticosteroid-free on 33% of study days, compared with 25% of days for the baricitinib 2 mg patients and 17% of days for controls.
In terms of safety, there was a case of pulmonary embolism in the higher-dose baricitinib group and an opportunistic toxoplasmosis eye infection in the control population. The frequency of oral herpes and herpes simplex virus infections was 2.8% in controls, 4.6% in the baricitinib 2-mg group, and 6.3% in the 4-mg group. There was also a signal of a dose-dependent increased risk of new-onset acne, with rates of 0.9% in controls and patients on baricitinib 2 mg, climbing to 3.6% with baricitinib 4 mg.
“In phase 2 results with upadacitinib [another oral JAK inhibitor], we saw that more than 10% of patients in the highest-dose group developed what was classified as acne. I cannot explain this, but it’s something we will monitor in the future,” Dr. Reich promised.
A fuller picture of baricitinib’s safety profile in the setting of atopic dermatitis clearly requires larger and longer-term studies, he added.
Baricitinib at the 2 mg daily dose is already marketed as Olumiant for the treatment of rheumatoid arthritis, with labeling that includes a boxed warning about serious infections, malignancy, and thrombosis. The Food and Drug Administration did not approve the 4-mg dose after determining that its higher safety hazard outweighed the efficacy advantage over the lower dose.
The BREEZE-AD7 study was sponsored by Eli Lilly. Dr. Reich reported serving as an adviser to, paid speaker for, and recipient of research grants from that pharmaceutical company and more than two dozen others.
MADRID – Adding the oral Janus kinase (JAK) inhibitor baricitinib to standard atopic dermatitis therapy with low- and midpotency topical corticosteroids markedly improved disease severity and key patient-reported outcomes, compared with topical corticosteroids alone, in the phase 3, randomized, double-blind BREEZE-AD7 trial, Kristian Reich, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
in the phase 3 BREEZE-AD1 and -AD2 trials. But BREEZE-AD7 further advances the field because it’s the first phase 3 study testing the efficacy of a JAK inhibitor in combination with low- and midpotency topical steroids.
“I think this study is important because it looks into the situation that’s more like what happens in the real world, which is, as with dupilumab and other drugs, we use the systemic agent in combination with topical therapies and, in particular, with topical corticosteroids,” commented Dr. Reich, professor of dermatology at University Medical Center, Hamburg, and medical director at SCIderm, a scientific research company.
“This is what I think we can expect from existing and upcoming systemic therapies in atopic dermatitis: We will use them in combination with topical corticosteroids, and hopefully this will allow patients to dramatically reduce the concomitant use of topical corticosteroids, as shown here in BREEZE-AD7,” he added.
BREEZE-AD7 was a 16-week study that included 329 adults with moderate or severe atopic dermatitis who were randomized to low- and midpotency topical corticosteroids plus either baricitinib at 2 mg once daily, baricitinib at 4 mg once daily, or placebo. The group’s mean baseline Eczema Area and Severity Index (EASI) score was 29. Overall, 45% of participants had a baseline Investigator’s Global Assessment (IGA) of disease severity of 4 on a 0-4 scale.
The primary endpoint was achievement of an IGA of 0 or 1, meaning clear or almost clear, along with at least a 2-point IGA improvement from baseline at week 16. This was accomplished in 30.6% of those on 4 mg/day of baricitinib, 23.9% of patients in the 2-mg group, and 14.7% of controls.
The 4-mg dose of baricitinib was statistically superior to placebo; the 2-mg dose was not. However, Dr. Reich indicated he was untroubled by this because the primary endpoint was set at a high bar, and both doses of baricitinib proved to be significantly better than topical steroids plus placebo in terms of EASI 75 response rates, as well as reductions in itch, skin pain, and sleep problems, which aren’t captured in EASI scores (see graphic).
“One of my big learnings from this year’s EADV is that we have to rethink the dimensions of atopic dermatitis. I think we have underestimated the relevance of important symptoms such as itch, the impact atopic dermatitis has on pain, and the effect it has on sleeping problems,” the dermatologist said. “My feeling is that baricitinib is strongest in reducing itch, improving sleep, and reducing pain, but it also has good effects on the clinical signs of atopic dermatitis.”
The baricitinib-treated patients’ rapidity of improvement in the various endpoints was particularly impressive. Both doses of the JAK 1/2 inhibitor showed significant separation from the control group in the first week, and the majority of improvement occurred by week 4.
A key finding was that patients on baricitinib at 2 mg/day and 4 mg/day used a mean total of 162 g and 137 g of midpotency topical steroids, respectively, during the 16 weeks, compared with 225 g in the control group. The higher-dose baricitinib group was topical corticosteroid-free on 33% of study days, compared with 25% of days for the baricitinib 2 mg patients and 17% of days for controls.
In terms of safety, there was a case of pulmonary embolism in the higher-dose baricitinib group and an opportunistic toxoplasmosis eye infection in the control population. The frequency of oral herpes and herpes simplex virus infections was 2.8% in controls, 4.6% in the baricitinib 2-mg group, and 6.3% in the 4-mg group. There was also a signal of a dose-dependent increased risk of new-onset acne, with rates of 0.9% in controls and patients on baricitinib 2 mg, climbing to 3.6% with baricitinib 4 mg.
“In phase 2 results with upadacitinib [another oral JAK inhibitor], we saw that more than 10% of patients in the highest-dose group developed what was classified as acne. I cannot explain this, but it’s something we will monitor in the future,” Dr. Reich promised.
A fuller picture of baricitinib’s safety profile in the setting of atopic dermatitis clearly requires larger and longer-term studies, he added.
Baricitinib at the 2 mg daily dose is already marketed as Olumiant for the treatment of rheumatoid arthritis, with labeling that includes a boxed warning about serious infections, malignancy, and thrombosis. The Food and Drug Administration did not approve the 4-mg dose after determining that its higher safety hazard outweighed the efficacy advantage over the lower dose.
The BREEZE-AD7 study was sponsored by Eli Lilly. Dr. Reich reported serving as an adviser to, paid speaker for, and recipient of research grants from that pharmaceutical company and more than two dozen others.
MADRID – Adding the oral Janus kinase (JAK) inhibitor baricitinib to standard atopic dermatitis therapy with low- and midpotency topical corticosteroids markedly improved disease severity and key patient-reported outcomes, compared with topical corticosteroids alone, in the phase 3, randomized, double-blind BREEZE-AD7 trial, Kristian Reich, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
in the phase 3 BREEZE-AD1 and -AD2 trials. But BREEZE-AD7 further advances the field because it’s the first phase 3 study testing the efficacy of a JAK inhibitor in combination with low- and midpotency topical steroids.
“I think this study is important because it looks into the situation that’s more like what happens in the real world, which is, as with dupilumab and other drugs, we use the systemic agent in combination with topical therapies and, in particular, with topical corticosteroids,” commented Dr. Reich, professor of dermatology at University Medical Center, Hamburg, and medical director at SCIderm, a scientific research company.
“This is what I think we can expect from existing and upcoming systemic therapies in atopic dermatitis: We will use them in combination with topical corticosteroids, and hopefully this will allow patients to dramatically reduce the concomitant use of topical corticosteroids, as shown here in BREEZE-AD7,” he added.
BREEZE-AD7 was a 16-week study that included 329 adults with moderate or severe atopic dermatitis who were randomized to low- and midpotency topical corticosteroids plus either baricitinib at 2 mg once daily, baricitinib at 4 mg once daily, or placebo. The group’s mean baseline Eczema Area and Severity Index (EASI) score was 29. Overall, 45% of participants had a baseline Investigator’s Global Assessment (IGA) of disease severity of 4 on a 0-4 scale.
The primary endpoint was achievement of an IGA of 0 or 1, meaning clear or almost clear, along with at least a 2-point IGA improvement from baseline at week 16. This was accomplished in 30.6% of those on 4 mg/day of baricitinib, 23.9% of patients in the 2-mg group, and 14.7% of controls.
The 4-mg dose of baricitinib was statistically superior to placebo; the 2-mg dose was not. However, Dr. Reich indicated he was untroubled by this because the primary endpoint was set at a high bar, and both doses of baricitinib proved to be significantly better than topical steroids plus placebo in terms of EASI 75 response rates, as well as reductions in itch, skin pain, and sleep problems, which aren’t captured in EASI scores (see graphic).
“One of my big learnings from this year’s EADV is that we have to rethink the dimensions of atopic dermatitis. I think we have underestimated the relevance of important symptoms such as itch, the impact atopic dermatitis has on pain, and the effect it has on sleeping problems,” the dermatologist said. “My feeling is that baricitinib is strongest in reducing itch, improving sleep, and reducing pain, but it also has good effects on the clinical signs of atopic dermatitis.”
The baricitinib-treated patients’ rapidity of improvement in the various endpoints was particularly impressive. Both doses of the JAK 1/2 inhibitor showed significant separation from the control group in the first week, and the majority of improvement occurred by week 4.
A key finding was that patients on baricitinib at 2 mg/day and 4 mg/day used a mean total of 162 g and 137 g of midpotency topical steroids, respectively, during the 16 weeks, compared with 225 g in the control group. The higher-dose baricitinib group was topical corticosteroid-free on 33% of study days, compared with 25% of days for the baricitinib 2 mg patients and 17% of days for controls.
In terms of safety, there was a case of pulmonary embolism in the higher-dose baricitinib group and an opportunistic toxoplasmosis eye infection in the control population. The frequency of oral herpes and herpes simplex virus infections was 2.8% in controls, 4.6% in the baricitinib 2-mg group, and 6.3% in the 4-mg group. There was also a signal of a dose-dependent increased risk of new-onset acne, with rates of 0.9% in controls and patients on baricitinib 2 mg, climbing to 3.6% with baricitinib 4 mg.
“In phase 2 results with upadacitinib [another oral JAK inhibitor], we saw that more than 10% of patients in the highest-dose group developed what was classified as acne. I cannot explain this, but it’s something we will monitor in the future,” Dr. Reich promised.
A fuller picture of baricitinib’s safety profile in the setting of atopic dermatitis clearly requires larger and longer-term studies, he added.
Baricitinib at the 2 mg daily dose is already marketed as Olumiant for the treatment of rheumatoid arthritis, with labeling that includes a boxed warning about serious infections, malignancy, and thrombosis. The Food and Drug Administration did not approve the 4-mg dose after determining that its higher safety hazard outweighed the efficacy advantage over the lower dose.
The BREEZE-AD7 study was sponsored by Eli Lilly. Dr. Reich reported serving as an adviser to, paid speaker for, and recipient of research grants from that pharmaceutical company and more than two dozen others.
REPORTING FROM EADV CONGRESS
Key clinical point: The Janus kinase 1/2 inhibitor baricitinib shows promise as a novel oral treatment for moderate or severe atopic dermatitis.
Major finding: Among atopic dermatitis patients on concomitant topical corticosteroids, a 75% improvement on Eczema Area and Severity Index at 16 weeks was achieved in 48% of those on baricitinib at 4 mg/day, 43% with baricitinib at 2 mg/day, and 23% on placebo.
Study details: BREEZE-AD7 was a phase 3, multicenter, 16-week, double-blind, three-arm study including 329 adults with moderate or severe atopic dermatitis.
Disclosures: The BREEZE-AD7 study was sponsored by Eli Lilly. The presenter reported serving as an adviser to, paid speaker for, and/or recipient of research grants from that pharmaceutical company and more than two dozen others.
Source: Reich K. EADV Congress, late breaker.
Early onset of atopic dermatitis linked to poorer control
according to a study published in the Journal of the American Academy of Dermatology.
Atopic dermatitis most commonly arises in infancy but also can emerge in later childhood and even adolescence, leading to a distinction between early- and late-onset disease, wrote Joy Wan, MD, of the University of Pennsylvania, Philadelphia, and coauthors.
“Early-onset, mid-onset, and late-onset AD appear to differ in the presence of active disease over time; however, whether these groups also differ in terms of the severity of AD is unknown,” they wrote.
In this observational cohort study, 8,015 individuals with childhood-onset atopic dermatitis – 53% of whom were female – were assessed twice-yearly for up to 10 years. Nearly three-quarters (72%) of the group had early-onset atopic dermatitis – defined as onset before 2 years of age – while 19% had mid-onset disease (3-7 years) and 9% had late-onset disease (8-17 years).
The study found that older age of onset was associated with better control, such that for each additional year of age at the onset of disease, there was a 7% reduction in the odds of poorer control of disease. Those who had mid-onset disease had a 29% lower odds of poorer control compared with those with early-onset, while those with late-onset disease had a 49% lower odds of poorer control.
The likelihood of atopic dermatitis persisting beyond childhood also appeared to be linked to the age of onset. Those with mid-onset disease had a 55% lower odds of persistent atopic dermatitis, compared with those with early-onset disease, while those with late-onset disease had an 81% lower odds.
“In all 3 groups, the proportion of subjects reporting persistent AD generally declined with older age, and the differences among the 3 onset age groups were most pronounced from early adolescence onward,” the authors wrote.
They noted that there was considerable research currently focused on identifying distinct atopic dermatitis phenotypes and endotypes, and their evidence on the different disease course for early-, mid-, and late-onset disease supported this idea of disease subtypes.
“However, additional research is needed to understand whether and how early-, mid-, and late-onset AD differ molecularly or immunologically, and whether they respond differentially to treatment,” they wrote. They also suggested that the timing of onset could help identify patients who were at greater risk of persistent or poorly controlled disease, and who benefits from more intensive monitoring or treatment.
The study was partly supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases and the Dermatology Foundation. Three authors declared funding, consultancies, or advisory board positions with the pharmaceutical sector. No other conflicts of interest were declared.
SOURCE: Wan J et al. J Am Acad Dermatol. 2019 Dec;81(6):1292-9.
according to a study published in the Journal of the American Academy of Dermatology.
Atopic dermatitis most commonly arises in infancy but also can emerge in later childhood and even adolescence, leading to a distinction between early- and late-onset disease, wrote Joy Wan, MD, of the University of Pennsylvania, Philadelphia, and coauthors.
“Early-onset, mid-onset, and late-onset AD appear to differ in the presence of active disease over time; however, whether these groups also differ in terms of the severity of AD is unknown,” they wrote.
In this observational cohort study, 8,015 individuals with childhood-onset atopic dermatitis – 53% of whom were female – were assessed twice-yearly for up to 10 years. Nearly three-quarters (72%) of the group had early-onset atopic dermatitis – defined as onset before 2 years of age – while 19% had mid-onset disease (3-7 years) and 9% had late-onset disease (8-17 years).
The study found that older age of onset was associated with better control, such that for each additional year of age at the onset of disease, there was a 7% reduction in the odds of poorer control of disease. Those who had mid-onset disease had a 29% lower odds of poorer control compared with those with early-onset, while those with late-onset disease had a 49% lower odds of poorer control.
The likelihood of atopic dermatitis persisting beyond childhood also appeared to be linked to the age of onset. Those with mid-onset disease had a 55% lower odds of persistent atopic dermatitis, compared with those with early-onset disease, while those with late-onset disease had an 81% lower odds.
“In all 3 groups, the proportion of subjects reporting persistent AD generally declined with older age, and the differences among the 3 onset age groups were most pronounced from early adolescence onward,” the authors wrote.
They noted that there was considerable research currently focused on identifying distinct atopic dermatitis phenotypes and endotypes, and their evidence on the different disease course for early-, mid-, and late-onset disease supported this idea of disease subtypes.
“However, additional research is needed to understand whether and how early-, mid-, and late-onset AD differ molecularly or immunologically, and whether they respond differentially to treatment,” they wrote. They also suggested that the timing of onset could help identify patients who were at greater risk of persistent or poorly controlled disease, and who benefits from more intensive monitoring or treatment.
The study was partly supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases and the Dermatology Foundation. Three authors declared funding, consultancies, or advisory board positions with the pharmaceutical sector. No other conflicts of interest were declared.
SOURCE: Wan J et al. J Am Acad Dermatol. 2019 Dec;81(6):1292-9.
according to a study published in the Journal of the American Academy of Dermatology.
Atopic dermatitis most commonly arises in infancy but also can emerge in later childhood and even adolescence, leading to a distinction between early- and late-onset disease, wrote Joy Wan, MD, of the University of Pennsylvania, Philadelphia, and coauthors.
“Early-onset, mid-onset, and late-onset AD appear to differ in the presence of active disease over time; however, whether these groups also differ in terms of the severity of AD is unknown,” they wrote.
In this observational cohort study, 8,015 individuals with childhood-onset atopic dermatitis – 53% of whom were female – were assessed twice-yearly for up to 10 years. Nearly three-quarters (72%) of the group had early-onset atopic dermatitis – defined as onset before 2 years of age – while 19% had mid-onset disease (3-7 years) and 9% had late-onset disease (8-17 years).
The study found that older age of onset was associated with better control, such that for each additional year of age at the onset of disease, there was a 7% reduction in the odds of poorer control of disease. Those who had mid-onset disease had a 29% lower odds of poorer control compared with those with early-onset, while those with late-onset disease had a 49% lower odds of poorer control.
The likelihood of atopic dermatitis persisting beyond childhood also appeared to be linked to the age of onset. Those with mid-onset disease had a 55% lower odds of persistent atopic dermatitis, compared with those with early-onset disease, while those with late-onset disease had an 81% lower odds.
“In all 3 groups, the proportion of subjects reporting persistent AD generally declined with older age, and the differences among the 3 onset age groups were most pronounced from early adolescence onward,” the authors wrote.
They noted that there was considerable research currently focused on identifying distinct atopic dermatitis phenotypes and endotypes, and their evidence on the different disease course for early-, mid-, and late-onset disease supported this idea of disease subtypes.
“However, additional research is needed to understand whether and how early-, mid-, and late-onset AD differ molecularly or immunologically, and whether they respond differentially to treatment,” they wrote. They also suggested that the timing of onset could help identify patients who were at greater risk of persistent or poorly controlled disease, and who benefits from more intensive monitoring or treatment.
The study was partly supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases and the Dermatology Foundation. Three authors declared funding, consultancies, or advisory board positions with the pharmaceutical sector. No other conflicts of interest were declared.
SOURCE: Wan J et al. J Am Acad Dermatol. 2019 Dec;81(6):1292-9.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Skin barrier dysfunction mutations vary by race, disease persistence in children with AD
Among children with atopic dermatitis, genetic variants associated with skin barrier dysfunction vary significantly by race and by their influence on disease persistence, according to authors of a cohort study.
In the study, which was based on data from a pediatric eczema registry, The investigators remarked on “profound” differences by race in the study, which used a high-throughput sequencing method to identify FLG LoF variants, some of which were common in white children but not so frequently seen in black children.
Conversely, some variants common in black children were completely absent in the white children, according to the investigators, led by David J. Margolis, MD, PhD, professor of dermatology at the University of Pennsylvania, Philadelphia. The study was published in JAMA Dermatology.
These findings imply that any genetic tests developed for AD should be “inclusive,” they wrote, and shouldn’t simply rely on the most common variants associated with patients of European ancestry, namely p.R501*, c.2282del4[p.S761fs], p.S3247*, and p.R2447*.
“Relying on the classic 4 FLG LoF variants would result in approximately 8% of white children and 64% of black children with an FLG LoF variant being improperly classified,” Dr. Margolis and coinvestigators wrote.
Their comprehensive analysis of FLG LoF variants was based on a U.S. cohort of 741 children with mild to moderate AD in the Pediatric Eczema Elective Registry (PEER), enrolled from 2005 to 2017. The mean age of onset of AD among the children was almost 2 years. Using massively parallel sequencing, the investigators identified a total of 23 FLG LoF variants in 177 children, or 23.9% of the overall cohort.
White children had a higher frequency of FLG LoF variants, according to the investigators. The prevalence of variants was 31.5% in white and 15.3% in black participants, translating into an odds ratio of 2.44 for carrying any variant in a white versus black child (95% confidence interval, 1.76-3.39).
In previous studies, FLG LoF variants are seen in 25%-30% of people with AD who have European and Asian ancestry; by contrast, they are “uncommonly” exhibited in individuals of African ancestry, the investigators wrote.
Persistent AD was more likely among children with FLG LoF variants, with an odds ratio of 0.67 (95% CI, 0.56-0.80), according to Dr. Margolis and coauthors. However, the black children in this cohort had more persistent disease, compared with white children, regardless of whether they had FLG LoF variants or not.
Exon 3 FLG LoF are known to be the most common variants linked to skin barrier dysfunction, the investigators noted.
“However, all FLG LoF variants might not confer an increased risk of AD, and further, they may not all have the same effect on the persistence of AD over time,” they added in a discussion of their results.
The study was supported by a grant from the National Institute of Arthritis and Musculoskeletal and Skin Diseases. The PEER cohort is funded by Valeant Pharmaceuticals. Dr. Margolis reported receiving research funding as the principal investigator via the trustees of the University of Pennsylvania, and receiving funding from the National Institutes of Health and Valeant; disclosures not related to the study included consulting activities primarily as a member of a data monitoring or scientific advisory boards for several pharmaceutical companies.
SOURCE: Margolis DJ et al. JAMA Dermatol. 2019 Jul 31. doi: 10.1011/jamadermatol.2019.1946.
Among children with atopic dermatitis, genetic variants associated with skin barrier dysfunction vary significantly by race and by their influence on disease persistence, according to authors of a cohort study.
In the study, which was based on data from a pediatric eczema registry, The investigators remarked on “profound” differences by race in the study, which used a high-throughput sequencing method to identify FLG LoF variants, some of which were common in white children but not so frequently seen in black children.
Conversely, some variants common in black children were completely absent in the white children, according to the investigators, led by David J. Margolis, MD, PhD, professor of dermatology at the University of Pennsylvania, Philadelphia. The study was published in JAMA Dermatology.
These findings imply that any genetic tests developed for AD should be “inclusive,” they wrote, and shouldn’t simply rely on the most common variants associated with patients of European ancestry, namely p.R501*, c.2282del4[p.S761fs], p.S3247*, and p.R2447*.
“Relying on the classic 4 FLG LoF variants would result in approximately 8% of white children and 64% of black children with an FLG LoF variant being improperly classified,” Dr. Margolis and coinvestigators wrote.
Their comprehensive analysis of FLG LoF variants was based on a U.S. cohort of 741 children with mild to moderate AD in the Pediatric Eczema Elective Registry (PEER), enrolled from 2005 to 2017. The mean age of onset of AD among the children was almost 2 years. Using massively parallel sequencing, the investigators identified a total of 23 FLG LoF variants in 177 children, or 23.9% of the overall cohort.
White children had a higher frequency of FLG LoF variants, according to the investigators. The prevalence of variants was 31.5% in white and 15.3% in black participants, translating into an odds ratio of 2.44 for carrying any variant in a white versus black child (95% confidence interval, 1.76-3.39).
In previous studies, FLG LoF variants are seen in 25%-30% of people with AD who have European and Asian ancestry; by contrast, they are “uncommonly” exhibited in individuals of African ancestry, the investigators wrote.
Persistent AD was more likely among children with FLG LoF variants, with an odds ratio of 0.67 (95% CI, 0.56-0.80), according to Dr. Margolis and coauthors. However, the black children in this cohort had more persistent disease, compared with white children, regardless of whether they had FLG LoF variants or not.
Exon 3 FLG LoF are known to be the most common variants linked to skin barrier dysfunction, the investigators noted.
“However, all FLG LoF variants might not confer an increased risk of AD, and further, they may not all have the same effect on the persistence of AD over time,” they added in a discussion of their results.
The study was supported by a grant from the National Institute of Arthritis and Musculoskeletal and Skin Diseases. The PEER cohort is funded by Valeant Pharmaceuticals. Dr. Margolis reported receiving research funding as the principal investigator via the trustees of the University of Pennsylvania, and receiving funding from the National Institutes of Health and Valeant; disclosures not related to the study included consulting activities primarily as a member of a data monitoring or scientific advisory boards for several pharmaceutical companies.
SOURCE: Margolis DJ et al. JAMA Dermatol. 2019 Jul 31. doi: 10.1011/jamadermatol.2019.1946.
Among children with atopic dermatitis, genetic variants associated with skin barrier dysfunction vary significantly by race and by their influence on disease persistence, according to authors of a cohort study.
In the study, which was based on data from a pediatric eczema registry, The investigators remarked on “profound” differences by race in the study, which used a high-throughput sequencing method to identify FLG LoF variants, some of which were common in white children but not so frequently seen in black children.
Conversely, some variants common in black children were completely absent in the white children, according to the investigators, led by David J. Margolis, MD, PhD, professor of dermatology at the University of Pennsylvania, Philadelphia. The study was published in JAMA Dermatology.
These findings imply that any genetic tests developed for AD should be “inclusive,” they wrote, and shouldn’t simply rely on the most common variants associated with patients of European ancestry, namely p.R501*, c.2282del4[p.S761fs], p.S3247*, and p.R2447*.
“Relying on the classic 4 FLG LoF variants would result in approximately 8% of white children and 64% of black children with an FLG LoF variant being improperly classified,” Dr. Margolis and coinvestigators wrote.
Their comprehensive analysis of FLG LoF variants was based on a U.S. cohort of 741 children with mild to moderate AD in the Pediatric Eczema Elective Registry (PEER), enrolled from 2005 to 2017. The mean age of onset of AD among the children was almost 2 years. Using massively parallel sequencing, the investigators identified a total of 23 FLG LoF variants in 177 children, or 23.9% of the overall cohort.
White children had a higher frequency of FLG LoF variants, according to the investigators. The prevalence of variants was 31.5% in white and 15.3% in black participants, translating into an odds ratio of 2.44 for carrying any variant in a white versus black child (95% confidence interval, 1.76-3.39).
In previous studies, FLG LoF variants are seen in 25%-30% of people with AD who have European and Asian ancestry; by contrast, they are “uncommonly” exhibited in individuals of African ancestry, the investigators wrote.
Persistent AD was more likely among children with FLG LoF variants, with an odds ratio of 0.67 (95% CI, 0.56-0.80), according to Dr. Margolis and coauthors. However, the black children in this cohort had more persistent disease, compared with white children, regardless of whether they had FLG LoF variants or not.
Exon 3 FLG LoF are known to be the most common variants linked to skin barrier dysfunction, the investigators noted.
“However, all FLG LoF variants might not confer an increased risk of AD, and further, they may not all have the same effect on the persistence of AD over time,” they added in a discussion of their results.
The study was supported by a grant from the National Institute of Arthritis and Musculoskeletal and Skin Diseases. The PEER cohort is funded by Valeant Pharmaceuticals. Dr. Margolis reported receiving research funding as the principal investigator via the trustees of the University of Pennsylvania, and receiving funding from the National Institutes of Health and Valeant; disclosures not related to the study included consulting activities primarily as a member of a data monitoring or scientific advisory boards for several pharmaceutical companies.
SOURCE: Margolis DJ et al. JAMA Dermatol. 2019 Jul 31. doi: 10.1011/jamadermatol.2019.1946.
FROM JAMA DERMATOLOGY