User login
'An Organization in Turmoil': Ken Kizer on the Challenges Facing the VA
'An Organization in Turmoil': Ken Kizer on the Challenges Facing the VA
Kenneth W. Kizer, MD, MPH, knows a thing or two about transition at the US Department of Veterans Affairs (VA). He served as VA Under Secretary of Health from 1994 to 1999, stepping in during an era of crisis with a mandate for transformation.
Kizer, a Distinguished Professor Emeritus at the University of California, Davis School of Medicine, is among the top thinkers about the VA and its future. He recently spoke with Federal Practitioner about community care, the electronic health record transition, and other challenges facing the Veterans Health Administration (VHA).
At stake, Kizer explained, is an invaluable service for veterans—and much more. “VA is the largest provider of training for... multiple types of health professionals that people use every day,” he said. “There’s also the research, the direct care provided to veterans, and the contingency support the VA provides, which was very well demonstrated during the COVID pandemic. These are things that benefit all Americans, not just veterans.”
When you look at the VA, what do you see?
I see an organization in turmoil, a great health care system struggling with multiple major challenges simultaneously. The VHA is becoming a very large health insurance program without the necessary infrastructure, and costs are rising rapidly. And it is trying to roll out a new EHR and implement new third-party administrator contracts while suffering from significant staffing reductions and very depressed morale.
There are a host of other high-visibility and high-impact issues, including a major reorganization. There’s been a paucity of details about exactly what is going to change, who is going to be doing what, and how the changes will affect staffing and workflow.
How will the loss of 35,000 health care positions affect veterans' care?
If you don’t have enough people, then you’re not going to be able to provide the care that is needed. Years ago, I led a project assessing the Roseburg VA Medical Center in Central Oregon. Among other things, there were a lot of problems with cardiology care. The biggest complaint the cardiologists had, and why the hospital couldn’t keep cardiologists on staff, was that there weren’t enough support staff to do the electrocardiograms. The cardiologists had to do the electrocardiograms themselves, which meant they weren’t doing other things they should be doing. You can amplify that example in a hundred different ways in VA today. If physicians don’t have adequate support, they get frustrated and disenfranchised. And they leave.
One of the fallacies I’ve heard mentioned in some congressional hearings is that it’s mainly a matter of lower pay in the VA. Pay may be an issue somewhere on the list of recruiting challenges, but more important issues higher up are things like the support clinicians receive, the work environment, whether they feel valued, and how easy it is for them to do their work. Case in point: If you put in a new EHR that doesn’t work as well as the existing one, then some doctors are going to leave.
Is VA being pushed toward privatization?
At some point it becomes a self-fulfilling prophecy. If you don’t have the staff to provide the services, then you refer more veterans to the community, and you get in a downward spiral. Patients are going to the community, you lose more staff, you continue to be unable to provide services, and more care goes to the community.
A part of this equation that hasn’t been given adequate attention is VA’s teaching mission. If care is increasingly going to the community, those patients won’t be available for the trainees in teaching programs. That in turn impacts the pipeline of clinicians who will be available to serve the population at large. The negative effects will be seen far beyond the VA.
Why have you expressed concern over VA care fragmentation?1
Greater than 80% of VA ICU [intensive care unit] care is now being provided in community hospitals. When patients are discharged from those hospitals, they often continue getting follow-up care in the community because VA doesn’t have good mechanisms to reconnect those patients back to VA care.2
[Other researchers] found that the majority of emergency department care for enrolled veterans in New York State was being paid for by entities other than the VA, most commonly Medicare but also Medicaid and private insurance. Where follow-up care occurred often depended on who paid for the emergency department visit, not necessarily what was best for the patient.3
The core problem is that the VA has very little insight into what’s happening when its enrollees get care that is paid for by another payer. VA doesn’t know when their patients are in a private hospital emergency department, so they can’t reach out in real time, and they can’t reconnect with them afterward.
That is very different than for commercial health plans. They know when one of their enrollees is admitted to an out-of-network hospital, and when they are discharged, and they follow up immediately. VA doesn’t have the infrastructure in place to do that.
Why did the VA spend $44 billion on Medicare Advantage double-payments from 2018 to 2021?4
That number is much larger now—$87 billion from 2019 to 2023. Here’s the problem: When VA enrollees are also enrolled in a Medicare Advantage plan, the Medicare plan gets paid to provide the care for those veterans. But when those enrollees come to the VA, the VA provides and pays for the care but cannot bill Medicare for the costs. So the federal government ends up paying twice for care of the same person.
In a paper I coauthored last December we showed that in 2023 alone VA spent $23 billion for care of veterans enrolled in Medicare Advantage plan. Those duplicative payments accounted for almost 20% of VA's entire medical care budget.5
How can fragmented care be reduced?
Two things really stand out. First, real-time health insurance data sharing across payers is foundational. VA has to know when its patients get care by non-VA providers if it is going to coordinate and provide follow-up care. As a first step, VA and the Centers for Medicare & Medicaid Services need to create a data sharing platform for veterans dually enrolled in VA and Medicare or Medicaid.
This is not a new idea. I tried to do it when I was Under Secretary for Health in the late 1990s, but it never happened for various political reasons. Others have tried since. Maybe now, given how much money is at stake, it will finally get done.
Second, the VA needs to implement rigorous case management for high utilizers. The costs are not evenly distributed across enrollees. Approximately 10% of community care users account for almost 90% of community care expenditures. Common sense says you should intensely manage the care of those high-need patients who account for so much of the costs, try to avoid out-of-network ICU and emergency department care as much as possible, and build relationships with other providers so there are clear mechanisms to reconnect those patients back to VA care after an acute episode is treated outside the VA health system.
Is community care itself the problem?
No. Community care is a good thing for many veterans. It has increased access and made it easier for enrolled veterans to get care in some situations. The problem is that the VA hasn’t built in the mechanisms and processes to share information, manage complex patients, provide follow-up care, or oversee quality in community care.
Historically, VA has been an integrated delivery system that provided the overwhelming majority of care within its own facilities. However, over the last decade it has become a hybrid purchaser-provider system. It has become a very large purchaser of non-VA care, going from about $7 billion to $50 billion in community care spending over the past decade. But the VA hasn’t built the infrastructure—information exchange, case management, utilization review, quality oversight—that a hybrid purchaser-provider system needs to be a prudent purchaser.
What is your perspective on VHA's EHR transition?
The many problems with the rollout of the Oracle/Cerner EHR have been well-documented by the Inspector General, frontline clinicians, and others. The problems have been so bad that implementation has been halted a couple times. They’re now moving forward again, but it remains to be seen whether the problems truly have been fixed.
Still unaddressed is the more fundamental question of whether VistA could have been upgraded and modernized at far less cost and disruption of care. No thorough, deliberative analysis of that was ever done. And some of the ostensible problems with upgrading VistA in years past are no longer an issue.
Given the challenges VA faces, are you optimistic about its future?
While there definitely are problems, they are all solvable. Every challenge the VA is facing can be addressed. The question is when and how, and whether the VA is going to be given a fair chance to work through its challenges.
As for those who look to the private sector and think that’s the solution: They haven’t looked closely enough. The private sector is also struggling with staffing and financing issues, many of the same issues VA is dealing with, just in a somewhat different way. The problems in the private sector will be an increasing challenge for community care going forward.
Overall, my life experience is that dark times are always followed by daylight, so I am confident there are brighter days ahead for VA.
1. Kizer KW. Curbing the growing fragmentation of veterans’ health care. JAMA Health Forum. 2025;6:e254148. doi:10.1001/jamahealthforum.2025.4148
2. Hahn Z, Naiditch H, Talisa V, et al. Intensive care unit admissions purchased or delivered by veterans in the VA health care system. JAMA Health Forum. 2025;6:e255605. doi:10.1001/jamahealthforum.2025.5605
3. Vashi AA, Urech T, Wu S, Asch S. Fragmented financing in emergency department use among US veterans. JAMA Health Forum. 2025;6:e255635. doi:10.1001/jamahealthforum.2025.5635
4. Maremont M, Weaver C, McGinty T. Insurers collected billions from medicare for veterans who cost them almost nothing. The Wall Street Journal. December 2, 2024. Accessed March 17, 2026. https://www.wsj.com/health/healthcare /veterans-medicare-insurers-collect-billions-bfd47d27
5. Trivedi AN, Jiang L, Meyers DJ, et al. Spending by the Veterans Affairs health care system for Medicare Advantage Enrollees. JAMA Health Forum. 2025;6:e255653. doi:10.1001/jamahealthforum.2025.5653
Kenneth W. Kizer, MD, MPH, knows a thing or two about transition at the US Department of Veterans Affairs (VA). He served as VA Under Secretary of Health from 1994 to 1999, stepping in during an era of crisis with a mandate for transformation.
Kizer, a Distinguished Professor Emeritus at the University of California, Davis School of Medicine, is among the top thinkers about the VA and its future. He recently spoke with Federal Practitioner about community care, the electronic health record transition, and other challenges facing the Veterans Health Administration (VHA).
At stake, Kizer explained, is an invaluable service for veterans—and much more. “VA is the largest provider of training for... multiple types of health professionals that people use every day,” he said. “There’s also the research, the direct care provided to veterans, and the contingency support the VA provides, which was very well demonstrated during the COVID pandemic. These are things that benefit all Americans, not just veterans.”
When you look at the VA, what do you see?
I see an organization in turmoil, a great health care system struggling with multiple major challenges simultaneously. The VHA is becoming a very large health insurance program without the necessary infrastructure, and costs are rising rapidly. And it is trying to roll out a new EHR and implement new third-party administrator contracts while suffering from significant staffing reductions and very depressed morale.
There are a host of other high-visibility and high-impact issues, including a major reorganization. There’s been a paucity of details about exactly what is going to change, who is going to be doing what, and how the changes will affect staffing and workflow.
How will the loss of 35,000 health care positions affect veterans' care?
If you don’t have enough people, then you’re not going to be able to provide the care that is needed. Years ago, I led a project assessing the Roseburg VA Medical Center in Central Oregon. Among other things, there were a lot of problems with cardiology care. The biggest complaint the cardiologists had, and why the hospital couldn’t keep cardiologists on staff, was that there weren’t enough support staff to do the electrocardiograms. The cardiologists had to do the electrocardiograms themselves, which meant they weren’t doing other things they should be doing. You can amplify that example in a hundred different ways in VA today. If physicians don’t have adequate support, they get frustrated and disenfranchised. And they leave.
One of the fallacies I’ve heard mentioned in some congressional hearings is that it’s mainly a matter of lower pay in the VA. Pay may be an issue somewhere on the list of recruiting challenges, but more important issues higher up are things like the support clinicians receive, the work environment, whether they feel valued, and how easy it is for them to do their work. Case in point: If you put in a new EHR that doesn’t work as well as the existing one, then some doctors are going to leave.
Is VA being pushed toward privatization?
At some point it becomes a self-fulfilling prophecy. If you don’t have the staff to provide the services, then you refer more veterans to the community, and you get in a downward spiral. Patients are going to the community, you lose more staff, you continue to be unable to provide services, and more care goes to the community.
A part of this equation that hasn’t been given adequate attention is VA’s teaching mission. If care is increasingly going to the community, those patients won’t be available for the trainees in teaching programs. That in turn impacts the pipeline of clinicians who will be available to serve the population at large. The negative effects will be seen far beyond the VA.
Why have you expressed concern over VA care fragmentation?1
Greater than 80% of VA ICU [intensive care unit] care is now being provided in community hospitals. When patients are discharged from those hospitals, they often continue getting follow-up care in the community because VA doesn’t have good mechanisms to reconnect those patients back to VA care.2
[Other researchers] found that the majority of emergency department care for enrolled veterans in New York State was being paid for by entities other than the VA, most commonly Medicare but also Medicaid and private insurance. Where follow-up care occurred often depended on who paid for the emergency department visit, not necessarily what was best for the patient.3
The core problem is that the VA has very little insight into what’s happening when its enrollees get care that is paid for by another payer. VA doesn’t know when their patients are in a private hospital emergency department, so they can’t reach out in real time, and they can’t reconnect with them afterward.
That is very different than for commercial health plans. They know when one of their enrollees is admitted to an out-of-network hospital, and when they are discharged, and they follow up immediately. VA doesn’t have the infrastructure in place to do that.
Why did the VA spend $44 billion on Medicare Advantage double-payments from 2018 to 2021?4
That number is much larger now—$87 billion from 2019 to 2023. Here’s the problem: When VA enrollees are also enrolled in a Medicare Advantage plan, the Medicare plan gets paid to provide the care for those veterans. But when those enrollees come to the VA, the VA provides and pays for the care but cannot bill Medicare for the costs. So the federal government ends up paying twice for care of the same person.
In a paper I coauthored last December we showed that in 2023 alone VA spent $23 billion for care of veterans enrolled in Medicare Advantage plan. Those duplicative payments accounted for almost 20% of VA's entire medical care budget.5
How can fragmented care be reduced?
Two things really stand out. First, real-time health insurance data sharing across payers is foundational. VA has to know when its patients get care by non-VA providers if it is going to coordinate and provide follow-up care. As a first step, VA and the Centers for Medicare & Medicaid Services need to create a data sharing platform for veterans dually enrolled in VA and Medicare or Medicaid.
This is not a new idea. I tried to do it when I was Under Secretary for Health in the late 1990s, but it never happened for various political reasons. Others have tried since. Maybe now, given how much money is at stake, it will finally get done.
Second, the VA needs to implement rigorous case management for high utilizers. The costs are not evenly distributed across enrollees. Approximately 10% of community care users account for almost 90% of community care expenditures. Common sense says you should intensely manage the care of those high-need patients who account for so much of the costs, try to avoid out-of-network ICU and emergency department care as much as possible, and build relationships with other providers so there are clear mechanisms to reconnect those patients back to VA care after an acute episode is treated outside the VA health system.
Is community care itself the problem?
No. Community care is a good thing for many veterans. It has increased access and made it easier for enrolled veterans to get care in some situations. The problem is that the VA hasn’t built in the mechanisms and processes to share information, manage complex patients, provide follow-up care, or oversee quality in community care.
Historically, VA has been an integrated delivery system that provided the overwhelming majority of care within its own facilities. However, over the last decade it has become a hybrid purchaser-provider system. It has become a very large purchaser of non-VA care, going from about $7 billion to $50 billion in community care spending over the past decade. But the VA hasn’t built the infrastructure—information exchange, case management, utilization review, quality oversight—that a hybrid purchaser-provider system needs to be a prudent purchaser.
What is your perspective on VHA's EHR transition?
The many problems with the rollout of the Oracle/Cerner EHR have been well-documented by the Inspector General, frontline clinicians, and others. The problems have been so bad that implementation has been halted a couple times. They’re now moving forward again, but it remains to be seen whether the problems truly have been fixed.
Still unaddressed is the more fundamental question of whether VistA could have been upgraded and modernized at far less cost and disruption of care. No thorough, deliberative analysis of that was ever done. And some of the ostensible problems with upgrading VistA in years past are no longer an issue.
Given the challenges VA faces, are you optimistic about its future?
While there definitely are problems, they are all solvable. Every challenge the VA is facing can be addressed. The question is when and how, and whether the VA is going to be given a fair chance to work through its challenges.
As for those who look to the private sector and think that’s the solution: They haven’t looked closely enough. The private sector is also struggling with staffing and financing issues, many of the same issues VA is dealing with, just in a somewhat different way. The problems in the private sector will be an increasing challenge for community care going forward.
Overall, my life experience is that dark times are always followed by daylight, so I am confident there are brighter days ahead for VA.
Kenneth W. Kizer, MD, MPH, knows a thing or two about transition at the US Department of Veterans Affairs (VA). He served as VA Under Secretary of Health from 1994 to 1999, stepping in during an era of crisis with a mandate for transformation.
Kizer, a Distinguished Professor Emeritus at the University of California, Davis School of Medicine, is among the top thinkers about the VA and its future. He recently spoke with Federal Practitioner about community care, the electronic health record transition, and other challenges facing the Veterans Health Administration (VHA).
At stake, Kizer explained, is an invaluable service for veterans—and much more. “VA is the largest provider of training for... multiple types of health professionals that people use every day,” he said. “There’s also the research, the direct care provided to veterans, and the contingency support the VA provides, which was very well demonstrated during the COVID pandemic. These are things that benefit all Americans, not just veterans.”
When you look at the VA, what do you see?
I see an organization in turmoil, a great health care system struggling with multiple major challenges simultaneously. The VHA is becoming a very large health insurance program without the necessary infrastructure, and costs are rising rapidly. And it is trying to roll out a new EHR and implement new third-party administrator contracts while suffering from significant staffing reductions and very depressed morale.
There are a host of other high-visibility and high-impact issues, including a major reorganization. There’s been a paucity of details about exactly what is going to change, who is going to be doing what, and how the changes will affect staffing and workflow.
How will the loss of 35,000 health care positions affect veterans' care?
If you don’t have enough people, then you’re not going to be able to provide the care that is needed. Years ago, I led a project assessing the Roseburg VA Medical Center in Central Oregon. Among other things, there were a lot of problems with cardiology care. The biggest complaint the cardiologists had, and why the hospital couldn’t keep cardiologists on staff, was that there weren’t enough support staff to do the electrocardiograms. The cardiologists had to do the electrocardiograms themselves, which meant they weren’t doing other things they should be doing. You can amplify that example in a hundred different ways in VA today. If physicians don’t have adequate support, they get frustrated and disenfranchised. And they leave.
One of the fallacies I’ve heard mentioned in some congressional hearings is that it’s mainly a matter of lower pay in the VA. Pay may be an issue somewhere on the list of recruiting challenges, but more important issues higher up are things like the support clinicians receive, the work environment, whether they feel valued, and how easy it is for them to do their work. Case in point: If you put in a new EHR that doesn’t work as well as the existing one, then some doctors are going to leave.
Is VA being pushed toward privatization?
At some point it becomes a self-fulfilling prophecy. If you don’t have the staff to provide the services, then you refer more veterans to the community, and you get in a downward spiral. Patients are going to the community, you lose more staff, you continue to be unable to provide services, and more care goes to the community.
A part of this equation that hasn’t been given adequate attention is VA’s teaching mission. If care is increasingly going to the community, those patients won’t be available for the trainees in teaching programs. That in turn impacts the pipeline of clinicians who will be available to serve the population at large. The negative effects will be seen far beyond the VA.
Why have you expressed concern over VA care fragmentation?1
Greater than 80% of VA ICU [intensive care unit] care is now being provided in community hospitals. When patients are discharged from those hospitals, they often continue getting follow-up care in the community because VA doesn’t have good mechanisms to reconnect those patients back to VA care.2
[Other researchers] found that the majority of emergency department care for enrolled veterans in New York State was being paid for by entities other than the VA, most commonly Medicare but also Medicaid and private insurance. Where follow-up care occurred often depended on who paid for the emergency department visit, not necessarily what was best for the patient.3
The core problem is that the VA has very little insight into what’s happening when its enrollees get care that is paid for by another payer. VA doesn’t know when their patients are in a private hospital emergency department, so they can’t reach out in real time, and they can’t reconnect with them afterward.
That is very different than for commercial health plans. They know when one of their enrollees is admitted to an out-of-network hospital, and when they are discharged, and they follow up immediately. VA doesn’t have the infrastructure in place to do that.
Why did the VA spend $44 billion on Medicare Advantage double-payments from 2018 to 2021?4
That number is much larger now—$87 billion from 2019 to 2023. Here’s the problem: When VA enrollees are also enrolled in a Medicare Advantage plan, the Medicare plan gets paid to provide the care for those veterans. But when those enrollees come to the VA, the VA provides and pays for the care but cannot bill Medicare for the costs. So the federal government ends up paying twice for care of the same person.
In a paper I coauthored last December we showed that in 2023 alone VA spent $23 billion for care of veterans enrolled in Medicare Advantage plan. Those duplicative payments accounted for almost 20% of VA's entire medical care budget.5
How can fragmented care be reduced?
Two things really stand out. First, real-time health insurance data sharing across payers is foundational. VA has to know when its patients get care by non-VA providers if it is going to coordinate and provide follow-up care. As a first step, VA and the Centers for Medicare & Medicaid Services need to create a data sharing platform for veterans dually enrolled in VA and Medicare or Medicaid.
This is not a new idea. I tried to do it when I was Under Secretary for Health in the late 1990s, but it never happened for various political reasons. Others have tried since. Maybe now, given how much money is at stake, it will finally get done.
Second, the VA needs to implement rigorous case management for high utilizers. The costs are not evenly distributed across enrollees. Approximately 10% of community care users account for almost 90% of community care expenditures. Common sense says you should intensely manage the care of those high-need patients who account for so much of the costs, try to avoid out-of-network ICU and emergency department care as much as possible, and build relationships with other providers so there are clear mechanisms to reconnect those patients back to VA care after an acute episode is treated outside the VA health system.
Is community care itself the problem?
No. Community care is a good thing for many veterans. It has increased access and made it easier for enrolled veterans to get care in some situations. The problem is that the VA hasn’t built in the mechanisms and processes to share information, manage complex patients, provide follow-up care, or oversee quality in community care.
Historically, VA has been an integrated delivery system that provided the overwhelming majority of care within its own facilities. However, over the last decade it has become a hybrid purchaser-provider system. It has become a very large purchaser of non-VA care, going from about $7 billion to $50 billion in community care spending over the past decade. But the VA hasn’t built the infrastructure—information exchange, case management, utilization review, quality oversight—that a hybrid purchaser-provider system needs to be a prudent purchaser.
What is your perspective on VHA's EHR transition?
The many problems with the rollout of the Oracle/Cerner EHR have been well-documented by the Inspector General, frontline clinicians, and others. The problems have been so bad that implementation has been halted a couple times. They’re now moving forward again, but it remains to be seen whether the problems truly have been fixed.
Still unaddressed is the more fundamental question of whether VistA could have been upgraded and modernized at far less cost and disruption of care. No thorough, deliberative analysis of that was ever done. And some of the ostensible problems with upgrading VistA in years past are no longer an issue.
Given the challenges VA faces, are you optimistic about its future?
While there definitely are problems, they are all solvable. Every challenge the VA is facing can be addressed. The question is when and how, and whether the VA is going to be given a fair chance to work through its challenges.
As for those who look to the private sector and think that’s the solution: They haven’t looked closely enough. The private sector is also struggling with staffing and financing issues, many of the same issues VA is dealing with, just in a somewhat different way. The problems in the private sector will be an increasing challenge for community care going forward.
Overall, my life experience is that dark times are always followed by daylight, so I am confident there are brighter days ahead for VA.
1. Kizer KW. Curbing the growing fragmentation of veterans’ health care. JAMA Health Forum. 2025;6:e254148. doi:10.1001/jamahealthforum.2025.4148
2. Hahn Z, Naiditch H, Talisa V, et al. Intensive care unit admissions purchased or delivered by veterans in the VA health care system. JAMA Health Forum. 2025;6:e255605. doi:10.1001/jamahealthforum.2025.5605
3. Vashi AA, Urech T, Wu S, Asch S. Fragmented financing in emergency department use among US veterans. JAMA Health Forum. 2025;6:e255635. doi:10.1001/jamahealthforum.2025.5635
4. Maremont M, Weaver C, McGinty T. Insurers collected billions from medicare for veterans who cost them almost nothing. The Wall Street Journal. December 2, 2024. Accessed March 17, 2026. https://www.wsj.com/health/healthcare /veterans-medicare-insurers-collect-billions-bfd47d27
5. Trivedi AN, Jiang L, Meyers DJ, et al. Spending by the Veterans Affairs health care system for Medicare Advantage Enrollees. JAMA Health Forum. 2025;6:e255653. doi:10.1001/jamahealthforum.2025.5653
1. Kizer KW. Curbing the growing fragmentation of veterans’ health care. JAMA Health Forum. 2025;6:e254148. doi:10.1001/jamahealthforum.2025.4148
2. Hahn Z, Naiditch H, Talisa V, et al. Intensive care unit admissions purchased or delivered by veterans in the VA health care system. JAMA Health Forum. 2025;6:e255605. doi:10.1001/jamahealthforum.2025.5605
3. Vashi AA, Urech T, Wu S, Asch S. Fragmented financing in emergency department use among US veterans. JAMA Health Forum. 2025;6:e255635. doi:10.1001/jamahealthforum.2025.5635
4. Maremont M, Weaver C, McGinty T. Insurers collected billions from medicare for veterans who cost them almost nothing. The Wall Street Journal. December 2, 2024. Accessed March 17, 2026. https://www.wsj.com/health/healthcare /veterans-medicare-insurers-collect-billions-bfd47d27
5. Trivedi AN, Jiang L, Meyers DJ, et al. Spending by the Veterans Affairs health care system for Medicare Advantage Enrollees. JAMA Health Forum. 2025;6:e255653. doi:10.1001/jamahealthforum.2025.5653
'An Organization in Turmoil': Ken Kizer on the Challenges Facing the VA
'An Organization in Turmoil': Ken Kizer on the Challenges Facing the VA
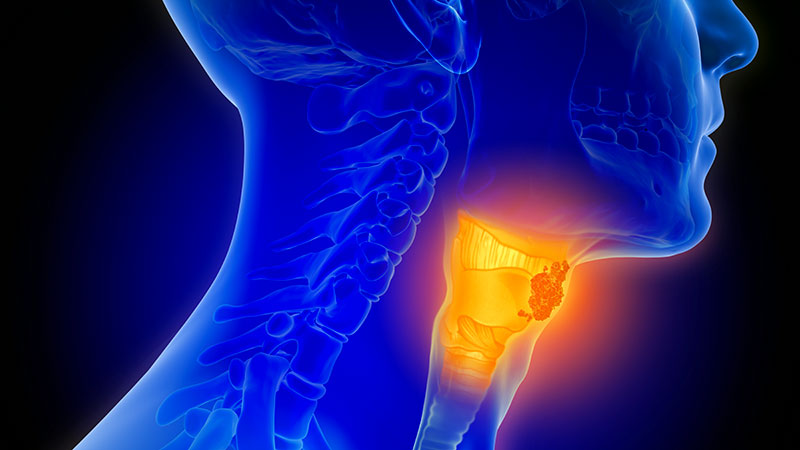
Team-Based Care is Crucial for Head-and-Neck Cancer Cases
Team-Based Care is Crucial for Head-and-Neck Cancer Cases
PHOENIX – A 70-year-old Vietnam veteran with oropharyngeal cancer presented challenges beyond his disease.
He couldn’t afford transportation for daily radiation treatments and had lost > 10% of his body weight due to pain and eating difficulties, recalled radiation oncologist Vinita Takiar, MD, PhD, in a presentation at the annual meeting of the Association of VA Hematology/Oncology.
To make matters more difficult, his wife held medical power of attorney despite his apparent competence to make decisions, said Takiar, who formerly worked with the US Department of Veterans Affairs (VA) Cincinnati Healthcare System and is now chair of radiation oncology at Penn State University.
All these factors would likely have derailed his treatment if not for a coordinated team intervention, Takiar said. Fortunately, the clinic launched a multifaceted effort involving representatives from the social work, dentistry, ethics, nutrition, and chaplaincy departments.
When surgery became impossible because the patient couldn’t lie on the operating table for adequate tumor exposure, she said, the existing team framework enabled a seamless and rapid transition to radiation with concurrent chemotherapy.
The patient completed treatment with an excellent response, offering a lesson in the importance of multidisciplinary care in head-and-neck cancers, she said.
In fact, when it comes to these forms of cancer, coordinated care “is probably more impactful than any treatment that we’re going to come up with,” she said. “The data show that when we do multidisciplinary care and we do it well, it actually improves the patient experience and outcomes.”
As Takiar noted, teamwork matters in many ways. It leads to better logistics and can address disparities, reduce financial burden and stigma, and even increase clinical trial involvement.
She pointed to studies linking teamwork to better outcomes, support for patients, and overall survival.
Takiar highlighted different parts of teams headed by radiation oncologists who act as “a node to improve multimodal care delivery.”
Speech and swallowing specialists, for example, are helpful in head-and-neck cancer because “there’s an impact on speech, swallowing, and appearance. Our patients don’t want to go out to dinner with friends because they can’t do it.”
Dentists and prosthodontists are key team members too: “I have dentists who have my cell phone number. They just call me: ‘Can I do this extraction? Was this in your radiation field? What was the dose?’”
Other team members include ear, nose, and throat specialists, palliative and supportive care specialists, medical oncologists, nurses, pathologists, transportation workers, and service connection specialists. She noted that previous military experience can affect radiation therapy. For example, the physical restraints required during treatment present particular challenges for veterans who’ve had wartime trauma. These patients may require therapy adjustments.
What’s next on the horizon? Takiar highlighted precision oncology and molecular profiling, artificial intelligence in care decisions and in radiation planning, telemedicine and virtual tumor boards, and expanded survivorship programs.
As for now, she urged colleagues to not be afraid to chat with radiation oncologists. “Please talk to us. We prioritize open communication and shared decision-making with the entire team,” she said. “If you see something and think your radiation oncologist should know about it, you think it was caused by the radiation, you should reach out to us.”
Takiar reported no disclosures.
PHOENIX – A 70-year-old Vietnam veteran with oropharyngeal cancer presented challenges beyond his disease.
He couldn’t afford transportation for daily radiation treatments and had lost > 10% of his body weight due to pain and eating difficulties, recalled radiation oncologist Vinita Takiar, MD, PhD, in a presentation at the annual meeting of the Association of VA Hematology/Oncology.
To make matters more difficult, his wife held medical power of attorney despite his apparent competence to make decisions, said Takiar, who formerly worked with the US Department of Veterans Affairs (VA) Cincinnati Healthcare System and is now chair of radiation oncology at Penn State University.
All these factors would likely have derailed his treatment if not for a coordinated team intervention, Takiar said. Fortunately, the clinic launched a multifaceted effort involving representatives from the social work, dentistry, ethics, nutrition, and chaplaincy departments.
When surgery became impossible because the patient couldn’t lie on the operating table for adequate tumor exposure, she said, the existing team framework enabled a seamless and rapid transition to radiation with concurrent chemotherapy.
The patient completed treatment with an excellent response, offering a lesson in the importance of multidisciplinary care in head-and-neck cancers, she said.
In fact, when it comes to these forms of cancer, coordinated care “is probably more impactful than any treatment that we’re going to come up with,” she said. “The data show that when we do multidisciplinary care and we do it well, it actually improves the patient experience and outcomes.”
As Takiar noted, teamwork matters in many ways. It leads to better logistics and can address disparities, reduce financial burden and stigma, and even increase clinical trial involvement.
She pointed to studies linking teamwork to better outcomes, support for patients, and overall survival.
Takiar highlighted different parts of teams headed by radiation oncologists who act as “a node to improve multimodal care delivery.”
Speech and swallowing specialists, for example, are helpful in head-and-neck cancer because “there’s an impact on speech, swallowing, and appearance. Our patients don’t want to go out to dinner with friends because they can’t do it.”
Dentists and prosthodontists are key team members too: “I have dentists who have my cell phone number. They just call me: ‘Can I do this extraction? Was this in your radiation field? What was the dose?’”
Other team members include ear, nose, and throat specialists, palliative and supportive care specialists, medical oncologists, nurses, pathologists, transportation workers, and service connection specialists. She noted that previous military experience can affect radiation therapy. For example, the physical restraints required during treatment present particular challenges for veterans who’ve had wartime trauma. These patients may require therapy adjustments.
What’s next on the horizon? Takiar highlighted precision oncology and molecular profiling, artificial intelligence in care decisions and in radiation planning, telemedicine and virtual tumor boards, and expanded survivorship programs.
As for now, she urged colleagues to not be afraid to chat with radiation oncologists. “Please talk to us. We prioritize open communication and shared decision-making with the entire team,” she said. “If you see something and think your radiation oncologist should know about it, you think it was caused by the radiation, you should reach out to us.”
Takiar reported no disclosures.
PHOENIX – A 70-year-old Vietnam veteran with oropharyngeal cancer presented challenges beyond his disease.
He couldn’t afford transportation for daily radiation treatments and had lost > 10% of his body weight due to pain and eating difficulties, recalled radiation oncologist Vinita Takiar, MD, PhD, in a presentation at the annual meeting of the Association of VA Hematology/Oncology.
To make matters more difficult, his wife held medical power of attorney despite his apparent competence to make decisions, said Takiar, who formerly worked with the US Department of Veterans Affairs (VA) Cincinnati Healthcare System and is now chair of radiation oncology at Penn State University.
All these factors would likely have derailed his treatment if not for a coordinated team intervention, Takiar said. Fortunately, the clinic launched a multifaceted effort involving representatives from the social work, dentistry, ethics, nutrition, and chaplaincy departments.
When surgery became impossible because the patient couldn’t lie on the operating table for adequate tumor exposure, she said, the existing team framework enabled a seamless and rapid transition to radiation with concurrent chemotherapy.
The patient completed treatment with an excellent response, offering a lesson in the importance of multidisciplinary care in head-and-neck cancers, she said.
In fact, when it comes to these forms of cancer, coordinated care “is probably more impactful than any treatment that we’re going to come up with,” she said. “The data show that when we do multidisciplinary care and we do it well, it actually improves the patient experience and outcomes.”
As Takiar noted, teamwork matters in many ways. It leads to better logistics and can address disparities, reduce financial burden and stigma, and even increase clinical trial involvement.
She pointed to studies linking teamwork to better outcomes, support for patients, and overall survival.
Takiar highlighted different parts of teams headed by radiation oncologists who act as “a node to improve multimodal care delivery.”
Speech and swallowing specialists, for example, are helpful in head-and-neck cancer because “there’s an impact on speech, swallowing, and appearance. Our patients don’t want to go out to dinner with friends because they can’t do it.”
Dentists and prosthodontists are key team members too: “I have dentists who have my cell phone number. They just call me: ‘Can I do this extraction? Was this in your radiation field? What was the dose?’”
Other team members include ear, nose, and throat specialists, palliative and supportive care specialists, medical oncologists, nurses, pathologists, transportation workers, and service connection specialists. She noted that previous military experience can affect radiation therapy. For example, the physical restraints required during treatment present particular challenges for veterans who’ve had wartime trauma. These patients may require therapy adjustments.
What’s next on the horizon? Takiar highlighted precision oncology and molecular profiling, artificial intelligence in care decisions and in radiation planning, telemedicine and virtual tumor boards, and expanded survivorship programs.
As for now, she urged colleagues to not be afraid to chat with radiation oncologists. “Please talk to us. We prioritize open communication and shared decision-making with the entire team,” she said. “If you see something and think your radiation oncologist should know about it, you think it was caused by the radiation, you should reach out to us.”
Takiar reported no disclosures.
Team-Based Care is Crucial for Head-and-Neck Cancer Cases
Team-Based Care is Crucial for Head-and-Neck Cancer Cases
'Distress is the Norm': How Oncologists Can Open the Door to Patient Mental Health
'Distress is the Norm': How Oncologists Can Open the Door to Patient Mental Health
For patients with cancer, the determining factor in whether they pursue mental health services is often whether their oncologist explicitly says it is a good idea, a psychologist said during the July Association of VA Hematology and Oncology (AVAHO) seminar in Long Beach, California, on treating veterans with renal cell carcinoma (RCC).
Kysa Christie, PhD, of the West Los Angeles Veterans Affairs Medical Center, presented findings from a 2018 study in which researchers asked Swiss patients with cancer whether their oncologist discussed their emotional health with them.
In terms of boosting intake, it did not matter if oncologists acknowledged distress or pointed out that psychosocial services existed. Instead, a direct recommendation made a difference, increasing the likelihood of using the services over a 4-month period after initial assessment (odds ratio, 6.27).
“What it took was, ‘I really recommend this. This is something that I would want you to try,’” Christie said.
Oncologists are crucial links between patients and mental health services, Christie said: “If people don’t ask about [distress], you’re not going to see it, but it’s there. Distress is the norm, right? It is not a weakness. It is something that we expect to see.”
Christie noted that an estimated 20% of cancer patients have major depressive disorder, and 35% to 40% have a diagnosable psychiatric condition. RCC shows disproportionately high rates of mental strain. According to Christie, research suggests that about three-fourths of the population report elevated levels of distress as evidenced by patients who scored ≥ 5 on the NCCN Distress Thermometer. Patients with cancer have an estimated 20% higher risk of suicide, especially during the first 12 months after diagnosis and at end of life, she added.
“Early during a diagnosis phase, where you’re having a lot of tests being done, you know something is happening. But you don’t know what,” Christie said. “It could be very serious. That’s just a lot of stress to hold and not know how to plan for.”
After diagnosis, routine could set in and lower distress, she said. Then terminal illness may spike it back up again. Does mental health treatment work in patients with cancer?
“There’s a really strong body of evidence-based treatments for depression, anxiety, adjustment disorders, and coping with different cancers,” Christie said. But it is a step too far to expect patients to ask for help while they are juggling appointments, tests, infusions, and more. “It’s a big ask, right? It’s setting people up for failure.”
To help, Christie said she is embedded with a medical oncology team and routinely talks with the staff about which patients may need help. “One thing I like to do is try to have brief visits with veterans and introduce myself when they come to clinic. I treat it like an opt-out rather than an opt-in program: I’ll just pop into the exam room. They don’t have to ask to see me.”
Christie focuses on open-ended questions and talks about resources ranging from support groups and brief appointments to extensive individual therapy.
Another approach is a strategy known as the “warm handoff,” when an oncologist directly introduces a patient to a mental health professional. “It’s a transfer of care in front of the veteran: It’s much more time-efficient than putting in a referral.”
Christie explained how this can work. A clinician will ask her to meet with a patient during an appointment, perhaps in a couple minutes.
“Then I pop into the room, and the oncologist says, ‘Thanks for joining us. This is Mr. Jones. He has been experiencing feelings of anxiety and sadness, and we’d appreciate your help in exploring some options that might help.’ I turn to the patient and ask, ‘What more would you add?’ Then I either take Mr. Jones back to my office or stay in clinic, and we’re off to the races.”
Christie reported no disclosures.
For patients with cancer, the determining factor in whether they pursue mental health services is often whether their oncologist explicitly says it is a good idea, a psychologist said during the July Association of VA Hematology and Oncology (AVAHO) seminar in Long Beach, California, on treating veterans with renal cell carcinoma (RCC).
Kysa Christie, PhD, of the West Los Angeles Veterans Affairs Medical Center, presented findings from a 2018 study in which researchers asked Swiss patients with cancer whether their oncologist discussed their emotional health with them.
In terms of boosting intake, it did not matter if oncologists acknowledged distress or pointed out that psychosocial services existed. Instead, a direct recommendation made a difference, increasing the likelihood of using the services over a 4-month period after initial assessment (odds ratio, 6.27).
“What it took was, ‘I really recommend this. This is something that I would want you to try,’” Christie said.
Oncologists are crucial links between patients and mental health services, Christie said: “If people don’t ask about [distress], you’re not going to see it, but it’s there. Distress is the norm, right? It is not a weakness. It is something that we expect to see.”
Christie noted that an estimated 20% of cancer patients have major depressive disorder, and 35% to 40% have a diagnosable psychiatric condition. RCC shows disproportionately high rates of mental strain. According to Christie, research suggests that about three-fourths of the population report elevated levels of distress as evidenced by patients who scored ≥ 5 on the NCCN Distress Thermometer. Patients with cancer have an estimated 20% higher risk of suicide, especially during the first 12 months after diagnosis and at end of life, she added.
“Early during a diagnosis phase, where you’re having a lot of tests being done, you know something is happening. But you don’t know what,” Christie said. “It could be very serious. That’s just a lot of stress to hold and not know how to plan for.”
After diagnosis, routine could set in and lower distress, she said. Then terminal illness may spike it back up again. Does mental health treatment work in patients with cancer?
“There’s a really strong body of evidence-based treatments for depression, anxiety, adjustment disorders, and coping with different cancers,” Christie said. But it is a step too far to expect patients to ask for help while they are juggling appointments, tests, infusions, and more. “It’s a big ask, right? It’s setting people up for failure.”
To help, Christie said she is embedded with a medical oncology team and routinely talks with the staff about which patients may need help. “One thing I like to do is try to have brief visits with veterans and introduce myself when they come to clinic. I treat it like an opt-out rather than an opt-in program: I’ll just pop into the exam room. They don’t have to ask to see me.”
Christie focuses on open-ended questions and talks about resources ranging from support groups and brief appointments to extensive individual therapy.
Another approach is a strategy known as the “warm handoff,” when an oncologist directly introduces a patient to a mental health professional. “It’s a transfer of care in front of the veteran: It’s much more time-efficient than putting in a referral.”
Christie explained how this can work. A clinician will ask her to meet with a patient during an appointment, perhaps in a couple minutes.
“Then I pop into the room, and the oncologist says, ‘Thanks for joining us. This is Mr. Jones. He has been experiencing feelings of anxiety and sadness, and we’d appreciate your help in exploring some options that might help.’ I turn to the patient and ask, ‘What more would you add?’ Then I either take Mr. Jones back to my office or stay in clinic, and we’re off to the races.”
Christie reported no disclosures.
For patients with cancer, the determining factor in whether they pursue mental health services is often whether their oncologist explicitly says it is a good idea, a psychologist said during the July Association of VA Hematology and Oncology (AVAHO) seminar in Long Beach, California, on treating veterans with renal cell carcinoma (RCC).
Kysa Christie, PhD, of the West Los Angeles Veterans Affairs Medical Center, presented findings from a 2018 study in which researchers asked Swiss patients with cancer whether their oncologist discussed their emotional health with them.
In terms of boosting intake, it did not matter if oncologists acknowledged distress or pointed out that psychosocial services existed. Instead, a direct recommendation made a difference, increasing the likelihood of using the services over a 4-month period after initial assessment (odds ratio, 6.27).
“What it took was, ‘I really recommend this. This is something that I would want you to try,’” Christie said.
Oncologists are crucial links between patients and mental health services, Christie said: “If people don’t ask about [distress], you’re not going to see it, but it’s there. Distress is the norm, right? It is not a weakness. It is something that we expect to see.”
Christie noted that an estimated 20% of cancer patients have major depressive disorder, and 35% to 40% have a diagnosable psychiatric condition. RCC shows disproportionately high rates of mental strain. According to Christie, research suggests that about three-fourths of the population report elevated levels of distress as evidenced by patients who scored ≥ 5 on the NCCN Distress Thermometer. Patients with cancer have an estimated 20% higher risk of suicide, especially during the first 12 months after diagnosis and at end of life, she added.
“Early during a diagnosis phase, where you’re having a lot of tests being done, you know something is happening. But you don’t know what,” Christie said. “It could be very serious. That’s just a lot of stress to hold and not know how to plan for.”
After diagnosis, routine could set in and lower distress, she said. Then terminal illness may spike it back up again. Does mental health treatment work in patients with cancer?
“There’s a really strong body of evidence-based treatments for depression, anxiety, adjustment disorders, and coping with different cancers,” Christie said. But it is a step too far to expect patients to ask for help while they are juggling appointments, tests, infusions, and more. “It’s a big ask, right? It’s setting people up for failure.”
To help, Christie said she is embedded with a medical oncology team and routinely talks with the staff about which patients may need help. “One thing I like to do is try to have brief visits with veterans and introduce myself when they come to clinic. I treat it like an opt-out rather than an opt-in program: I’ll just pop into the exam room. They don’t have to ask to see me.”
Christie focuses on open-ended questions and talks about resources ranging from support groups and brief appointments to extensive individual therapy.
Another approach is a strategy known as the “warm handoff,” when an oncologist directly introduces a patient to a mental health professional. “It’s a transfer of care in front of the veteran: It’s much more time-efficient than putting in a referral.”
Christie explained how this can work. A clinician will ask her to meet with a patient during an appointment, perhaps in a couple minutes.
“Then I pop into the room, and the oncologist says, ‘Thanks for joining us. This is Mr. Jones. He has been experiencing feelings of anxiety and sadness, and we’d appreciate your help in exploring some options that might help.’ I turn to the patient and ask, ‘What more would you add?’ Then I either take Mr. Jones back to my office or stay in clinic, and we’re off to the races.”
Christie reported no disclosures.
'Distress is the Norm': How Oncologists Can Open the Door to Patient Mental Health
'Distress is the Norm': How Oncologists Can Open the Door to Patient Mental Health
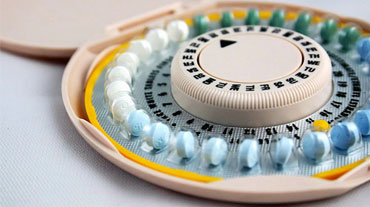
Contraceptive Care Clinic Focuses on Military Readiness
SAN DIEGO — Not surprisingly, the contraception clinic at Madigan Army Medical Center near Tacoma, Wash., is popular among female soldiers seeking to avoid pregnancy. However, about half of the patients drop by for other reasons, the military pharmacist who runs the program told colleagues here at the Joint Federal Pharmacy Seminar.
“They come to suppress menstruation, to get help with pain, to get help with PCOS [polycystic ovary syndrome] symptoms. They're coming for a wide range of indications that we use contraception to treat,” said Sarah Abel, PharmD, a clinical pharmacist.
Regardless of the reason, Abel emphasized that contraceptives can significantly impact the ability of female soldiers to do their jobs. “If you have heavy periods and can't make it in work, or you have endometriosis and requiring a lot of doctor's appointments, or you're deployed and you get pregnant, these are all situations where contraceptive care matters,” she said. Rates of unintended pregnancy are higher in servicewomen than in the general population.
Abel, who opened the medical center’s contraceptive clinic about 10 years ago, stressed that it’s crucial to military readiness considering that the percentage of women in the American military is approaching 20%.
Thanks to a 2022 edict, military hospitals and clinics are required to offer walk-in contraceptive services with same-day access, no requirements for appointments or referrals. An announcement about the mandate noted that these contraceptive services, such as preventing unplanned pregnancy and decreasing menstrual periods, “support the overall well-being of the force and optimize personal warrior readiness.”
As Abel noted, 29 states and Washington D.C. allow pharmacists to prescribe contraception to outpatients, although the requirements vary. “Can we start practicing at the top of our license and start prescribing in the outpatient setting? Absolutely we should,” she said. “Pharmacists have a very unique opportunity to be a part of this.”
Abel also shared that setting up a contraceptive program requires patience and education. “I cannot tell you how many women have come to me who don't know the different names of their body parts, women who've had two babies that don't understand how their body works. So, I constantly find myself taking extra time to do general sexual education,” she said.
There are many lessons to impart to patients about sexual health. For example, birth control drugs and devices do not prevent transmission of sexually transmitted infections (STIs). “So I have bowls of condoms literally everywhere because condoms are the only thing that protects against STIs,” Abel said.
In terms of devices, “we have diaphragms available and cervical caps,” she said. “The Caya diaphragm is a TRICARE-covered benefit. It’s a small purple diaphragm, one size fits most. We can prescribe it, and it is good for 2 years. Unfortunately, spermicide, which you have to use with these things, is not a TRICARE-covered benefit.”
Hormonal contraceptives are also available, with Abel recommending the continuous monophasic type for most women. “Please don't tell women they have to have their periods. They don't,” she said. “What I'm trying to do is give a woman some stability in her hormones. She can know and expect what she's going to feel like. She's not going to wake up and say, ‘Oh God, today's the day. I'm going to be like this for a week.’”
Patches are another option, and a flurry of patients have been asking about them because of recent TikTok videos promoting their use. “We have the Xulane patch, our bread and butter. They wear it on their shoulder, their hip, their butt, or their back. They leave it in place for a week at a time. And every week, they will change that patch. I usually have to walk patients through a whole month to help them understand how that works.”
Another option, the NuvaRing, is notable because it’s linked to low amounts of breakthrough bleeding Abel noted. An extended form is now available that doesn’t need to be removed during menstrual periods.
Medroxyprogesterone injections, which are linked to bone loss, and subdermal implants, which may be less effective in women over 130% of their ideal weight are also available, she said.
Finally, IUDs are an option, although when they fail, they’re linked to ectopic pregnancies.
Abel has no disclosures.
SAN DIEGO — Not surprisingly, the contraception clinic at Madigan Army Medical Center near Tacoma, Wash., is popular among female soldiers seeking to avoid pregnancy. However, about half of the patients drop by for other reasons, the military pharmacist who runs the program told colleagues here at the Joint Federal Pharmacy Seminar.
“They come to suppress menstruation, to get help with pain, to get help with PCOS [polycystic ovary syndrome] symptoms. They're coming for a wide range of indications that we use contraception to treat,” said Sarah Abel, PharmD, a clinical pharmacist.
Regardless of the reason, Abel emphasized that contraceptives can significantly impact the ability of female soldiers to do their jobs. “If you have heavy periods and can't make it in work, or you have endometriosis and requiring a lot of doctor's appointments, or you're deployed and you get pregnant, these are all situations where contraceptive care matters,” she said. Rates of unintended pregnancy are higher in servicewomen than in the general population.
Abel, who opened the medical center’s contraceptive clinic about 10 years ago, stressed that it’s crucial to military readiness considering that the percentage of women in the American military is approaching 20%.
Thanks to a 2022 edict, military hospitals and clinics are required to offer walk-in contraceptive services with same-day access, no requirements for appointments or referrals. An announcement about the mandate noted that these contraceptive services, such as preventing unplanned pregnancy and decreasing menstrual periods, “support the overall well-being of the force and optimize personal warrior readiness.”
As Abel noted, 29 states and Washington D.C. allow pharmacists to prescribe contraception to outpatients, although the requirements vary. “Can we start practicing at the top of our license and start prescribing in the outpatient setting? Absolutely we should,” she said. “Pharmacists have a very unique opportunity to be a part of this.”
Abel also shared that setting up a contraceptive program requires patience and education. “I cannot tell you how many women have come to me who don't know the different names of their body parts, women who've had two babies that don't understand how their body works. So, I constantly find myself taking extra time to do general sexual education,” she said.
There are many lessons to impart to patients about sexual health. For example, birth control drugs and devices do not prevent transmission of sexually transmitted infections (STIs). “So I have bowls of condoms literally everywhere because condoms are the only thing that protects against STIs,” Abel said.
In terms of devices, “we have diaphragms available and cervical caps,” she said. “The Caya diaphragm is a TRICARE-covered benefit. It’s a small purple diaphragm, one size fits most. We can prescribe it, and it is good for 2 years. Unfortunately, spermicide, which you have to use with these things, is not a TRICARE-covered benefit.”
Hormonal contraceptives are also available, with Abel recommending the continuous monophasic type for most women. “Please don't tell women they have to have their periods. They don't,” she said. “What I'm trying to do is give a woman some stability in her hormones. She can know and expect what she's going to feel like. She's not going to wake up and say, ‘Oh God, today's the day. I'm going to be like this for a week.’”
Patches are another option, and a flurry of patients have been asking about them because of recent TikTok videos promoting their use. “We have the Xulane patch, our bread and butter. They wear it on their shoulder, their hip, their butt, or their back. They leave it in place for a week at a time. And every week, they will change that patch. I usually have to walk patients through a whole month to help them understand how that works.”
Another option, the NuvaRing, is notable because it’s linked to low amounts of breakthrough bleeding Abel noted. An extended form is now available that doesn’t need to be removed during menstrual periods.
Medroxyprogesterone injections, which are linked to bone loss, and subdermal implants, which may be less effective in women over 130% of their ideal weight are also available, she said.
Finally, IUDs are an option, although when they fail, they’re linked to ectopic pregnancies.
Abel has no disclosures.
SAN DIEGO — Not surprisingly, the contraception clinic at Madigan Army Medical Center near Tacoma, Wash., is popular among female soldiers seeking to avoid pregnancy. However, about half of the patients drop by for other reasons, the military pharmacist who runs the program told colleagues here at the Joint Federal Pharmacy Seminar.
“They come to suppress menstruation, to get help with pain, to get help with PCOS [polycystic ovary syndrome] symptoms. They're coming for a wide range of indications that we use contraception to treat,” said Sarah Abel, PharmD, a clinical pharmacist.
Regardless of the reason, Abel emphasized that contraceptives can significantly impact the ability of female soldiers to do their jobs. “If you have heavy periods and can't make it in work, or you have endometriosis and requiring a lot of doctor's appointments, or you're deployed and you get pregnant, these are all situations where contraceptive care matters,” she said. Rates of unintended pregnancy are higher in servicewomen than in the general population.
Abel, who opened the medical center’s contraceptive clinic about 10 years ago, stressed that it’s crucial to military readiness considering that the percentage of women in the American military is approaching 20%.
Thanks to a 2022 edict, military hospitals and clinics are required to offer walk-in contraceptive services with same-day access, no requirements for appointments or referrals. An announcement about the mandate noted that these contraceptive services, such as preventing unplanned pregnancy and decreasing menstrual periods, “support the overall well-being of the force and optimize personal warrior readiness.”
As Abel noted, 29 states and Washington D.C. allow pharmacists to prescribe contraception to outpatients, although the requirements vary. “Can we start practicing at the top of our license and start prescribing in the outpatient setting? Absolutely we should,” she said. “Pharmacists have a very unique opportunity to be a part of this.”
Abel also shared that setting up a contraceptive program requires patience and education. “I cannot tell you how many women have come to me who don't know the different names of their body parts, women who've had two babies that don't understand how their body works. So, I constantly find myself taking extra time to do general sexual education,” she said.
There are many lessons to impart to patients about sexual health. For example, birth control drugs and devices do not prevent transmission of sexually transmitted infections (STIs). “So I have bowls of condoms literally everywhere because condoms are the only thing that protects against STIs,” Abel said.
In terms of devices, “we have diaphragms available and cervical caps,” she said. “The Caya diaphragm is a TRICARE-covered benefit. It’s a small purple diaphragm, one size fits most. We can prescribe it, and it is good for 2 years. Unfortunately, spermicide, which you have to use with these things, is not a TRICARE-covered benefit.”
Hormonal contraceptives are also available, with Abel recommending the continuous monophasic type for most women. “Please don't tell women they have to have their periods. They don't,” she said. “What I'm trying to do is give a woman some stability in her hormones. She can know and expect what she's going to feel like. She's not going to wake up and say, ‘Oh God, today's the day. I'm going to be like this for a week.’”
Patches are another option, and a flurry of patients have been asking about them because of recent TikTok videos promoting their use. “We have the Xulane patch, our bread and butter. They wear it on their shoulder, their hip, their butt, or their back. They leave it in place for a week at a time. And every week, they will change that patch. I usually have to walk patients through a whole month to help them understand how that works.”
Another option, the NuvaRing, is notable because it’s linked to low amounts of breakthrough bleeding Abel noted. An extended form is now available that doesn’t need to be removed during menstrual periods.
Medroxyprogesterone injections, which are linked to bone loss, and subdermal implants, which may be less effective in women over 130% of their ideal weight are also available, she said.
Finally, IUDs are an option, although when they fail, they’re linked to ectopic pregnancies.
Abel has no disclosures.
Rising Cancer Rates Among Young People Spur New Fertility Preservation Options
Rising Cancer Rates Among Young People Spur New Fertility Preservation Options
ATLANTA —Jacqueline Lee, MD, a reproductive endocrinologist at Emory School of Medicine, frequently treats patients with cancer. Recently, she treated 4 women in their 30s with histories of colon cancer, acute lymphoblastic leukemia, lymphoma, and breast cancer. A young man in his 20s sought her care, to discuss his case of lymphoma.
All these patients sought guidance from Lee because they want to protect their ability to have children. At the annual meeting of the Association of VA Hematology/Oncology, Lee explained that plenty of patients are finding themselves in similar straits due in part to recent trends.
Cancer rates in the US have been rising among people aged 15 to 39 years, who now account for 4.2% of all cancer cases. An estimated 84,100 people in this age group are expected to be diagnosed with cancer this year. Meanwhile, women are having children later in life-birth rates are up among those aged 25 to 49 years-making it more likely that they have histories of cancer.
Although it's difficult to predict how cancer will affect fertility, Lee emphasized that many chemotherapy medications, including cisplatin and carboplatin, are cytotoxic. "It's hard to always predict what someone's arc of care is going to be," she said, "so I really have a low threshold for recommending fertility preservation in patients who have a strong desire to have future childbearing."
For women with cancer, egg preservation isn't the only strategy. Clinicians can also try to protect ovarian tissue from pelvic radiation through surgical reposition of the ovaries, Lee noted. In addition goserelin, a hormone-suppressing therapy, may protect the ovaries from chemotherapy, though its effectiveness in boosting pregnancy rates is still unclear.
"When I mentioned this option, it's usually for patients who can't preserve fertility via egg or embryo preservation, or we don't have the luxury of that kind of time," Lee said. "I say that if helps at all, it might help you resume menses after treatment. But infertility is still very common."
For some patients, freezing eggs is an easy decision. "They don't have a reproductive partner they're ready to make embryos with, so we proceed with egg preservation. It's no longer considered experimental and comes with lower upfront costs since the costs of actually making embryos are deferred until the future."
In addition, she said, freezing eggs also avoids the touchy topic of disposing of embryos. Lee cautions patients that retrieving eggs is a 2-week process that requires any initiation of cancer care to be delayed. However, the retrieval process can be adjusted in patients with special needs due to the type of cancer they have.
For prepubertal girls with cancer, ovarian tissue can be removed and frozen as a fertility preservation option. However, this is not considered standard of care. "We don't do it," she said. "We refer out if needed. Hopefully we'll develop a program in the future."
As for the 5 patients that Lee mentioned, with details changed to protect their privacy, their outcomes were as follows:
- The woman with colon cancer, who had undergone a hemicolectomy, chose to defer fertility preservation.
- The woman with acute lymphoblastic leukemia, who was taking depo-Lupron, had undetectable anti-Müllerian hormone (AMH) levels. Lee discussed the possibility of IVF with a donor egg.
- The woman with breast cancer, who was newly diagnosed, deferred fertility preservation.
- The man with lymphoma (Hodgkin's), who was awaiting chemotherapy, had his sperm frozen.
- The woman with lymphoma (new diagnosis) had 27 eggs frozen.
Lee had no disclosures to report.
ATLANTA —Jacqueline Lee, MD, a reproductive endocrinologist at Emory School of Medicine, frequently treats patients with cancer. Recently, she treated 4 women in their 30s with histories of colon cancer, acute lymphoblastic leukemia, lymphoma, and breast cancer. A young man in his 20s sought her care, to discuss his case of lymphoma.
All these patients sought guidance from Lee because they want to protect their ability to have children. At the annual meeting of the Association of VA Hematology/Oncology, Lee explained that plenty of patients are finding themselves in similar straits due in part to recent trends.
Cancer rates in the US have been rising among people aged 15 to 39 years, who now account for 4.2% of all cancer cases. An estimated 84,100 people in this age group are expected to be diagnosed with cancer this year. Meanwhile, women are having children later in life-birth rates are up among those aged 25 to 49 years-making it more likely that they have histories of cancer.
Although it's difficult to predict how cancer will affect fertility, Lee emphasized that many chemotherapy medications, including cisplatin and carboplatin, are cytotoxic. "It's hard to always predict what someone's arc of care is going to be," she said, "so I really have a low threshold for recommending fertility preservation in patients who have a strong desire to have future childbearing."
For women with cancer, egg preservation isn't the only strategy. Clinicians can also try to protect ovarian tissue from pelvic radiation through surgical reposition of the ovaries, Lee noted. In addition goserelin, a hormone-suppressing therapy, may protect the ovaries from chemotherapy, though its effectiveness in boosting pregnancy rates is still unclear.
"When I mentioned this option, it's usually for patients who can't preserve fertility via egg or embryo preservation, or we don't have the luxury of that kind of time," Lee said. "I say that if helps at all, it might help you resume menses after treatment. But infertility is still very common."
For some patients, freezing eggs is an easy decision. "They don't have a reproductive partner they're ready to make embryos with, so we proceed with egg preservation. It's no longer considered experimental and comes with lower upfront costs since the costs of actually making embryos are deferred until the future."
In addition, she said, freezing eggs also avoids the touchy topic of disposing of embryos. Lee cautions patients that retrieving eggs is a 2-week process that requires any initiation of cancer care to be delayed. However, the retrieval process can be adjusted in patients with special needs due to the type of cancer they have.
For prepubertal girls with cancer, ovarian tissue can be removed and frozen as a fertility preservation option. However, this is not considered standard of care. "We don't do it," she said. "We refer out if needed. Hopefully we'll develop a program in the future."
As for the 5 patients that Lee mentioned, with details changed to protect their privacy, their outcomes were as follows:
- The woman with colon cancer, who had undergone a hemicolectomy, chose to defer fertility preservation.
- The woman with acute lymphoblastic leukemia, who was taking depo-Lupron, had undetectable anti-Müllerian hormone (AMH) levels. Lee discussed the possibility of IVF with a donor egg.
- The woman with breast cancer, who was newly diagnosed, deferred fertility preservation.
- The man with lymphoma (Hodgkin's), who was awaiting chemotherapy, had his sperm frozen.
- The woman with lymphoma (new diagnosis) had 27 eggs frozen.
Lee had no disclosures to report.
ATLANTA —Jacqueline Lee, MD, a reproductive endocrinologist at Emory School of Medicine, frequently treats patients with cancer. Recently, she treated 4 women in their 30s with histories of colon cancer, acute lymphoblastic leukemia, lymphoma, and breast cancer. A young man in his 20s sought her care, to discuss his case of lymphoma.
All these patients sought guidance from Lee because they want to protect their ability to have children. At the annual meeting of the Association of VA Hematology/Oncology, Lee explained that plenty of patients are finding themselves in similar straits due in part to recent trends.
Cancer rates in the US have been rising among people aged 15 to 39 years, who now account for 4.2% of all cancer cases. An estimated 84,100 people in this age group are expected to be diagnosed with cancer this year. Meanwhile, women are having children later in life-birth rates are up among those aged 25 to 49 years-making it more likely that they have histories of cancer.
Although it's difficult to predict how cancer will affect fertility, Lee emphasized that many chemotherapy medications, including cisplatin and carboplatin, are cytotoxic. "It's hard to always predict what someone's arc of care is going to be," she said, "so I really have a low threshold for recommending fertility preservation in patients who have a strong desire to have future childbearing."
For women with cancer, egg preservation isn't the only strategy. Clinicians can also try to protect ovarian tissue from pelvic radiation through surgical reposition of the ovaries, Lee noted. In addition goserelin, a hormone-suppressing therapy, may protect the ovaries from chemotherapy, though its effectiveness in boosting pregnancy rates is still unclear.
"When I mentioned this option, it's usually for patients who can't preserve fertility via egg or embryo preservation, or we don't have the luxury of that kind of time," Lee said. "I say that if helps at all, it might help you resume menses after treatment. But infertility is still very common."
For some patients, freezing eggs is an easy decision. "They don't have a reproductive partner they're ready to make embryos with, so we proceed with egg preservation. It's no longer considered experimental and comes with lower upfront costs since the costs of actually making embryos are deferred until the future."
In addition, she said, freezing eggs also avoids the touchy topic of disposing of embryos. Lee cautions patients that retrieving eggs is a 2-week process that requires any initiation of cancer care to be delayed. However, the retrieval process can be adjusted in patients with special needs due to the type of cancer they have.
For prepubertal girls with cancer, ovarian tissue can be removed and frozen as a fertility preservation option. However, this is not considered standard of care. "We don't do it," she said. "We refer out if needed. Hopefully we'll develop a program in the future."
As for the 5 patients that Lee mentioned, with details changed to protect their privacy, their outcomes were as follows:
- The woman with colon cancer, who had undergone a hemicolectomy, chose to defer fertility preservation.
- The woman with acute lymphoblastic leukemia, who was taking depo-Lupron, had undetectable anti-Müllerian hormone (AMH) levels. Lee discussed the possibility of IVF with a donor egg.
- The woman with breast cancer, who was newly diagnosed, deferred fertility preservation.
- The man with lymphoma (Hodgkin's), who was awaiting chemotherapy, had his sperm frozen.
- The woman with lymphoma (new diagnosis) had 27 eggs frozen.
Lee had no disclosures to report.
Rising Cancer Rates Among Young People Spur New Fertility Preservation Options
Rising Cancer Rates Among Young People Spur New Fertility Preservation Options
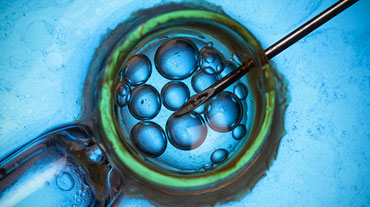
AI Scribes or VHA Docs: Which Created Better Clinical Notes?
Artificial intelligence (AI) scribes produced lower-quality documentation of clinical notes than human clinicians, and especially struggled in settings with background noise or clinicians wearing masks, a new Veterans Health Administration (VHA) study finds.
In 5 simulated clinical cases, notes written by various AI programs scored lower than reports produced by humans on the modified Physician Documentation Quality Instrument (PDQI-9), a measurement of note quality scale, reported Ashok Reddy, MD, MSc, of the University of Washington and Veterans Affairs Puget Sound Health Care System, Seattle, et al in the April issue of Annals of Internal Medicine.
AI scribes scored lower compared with humans across all domains, including accuracy, thoroughness, and usefulness. There was an especially large gap in scores on the 50-point PDQI-9 in an acute low back pain case (human, 43.8 points; AI, 20.3 points; difference, 23.5 points).
“For clinicians, AI scribes should be regarded as tools for generating draft documentation that requires review and editing, rather than as a substitute for clinician-authored notes,” the authors wrote. “Although ambient AI scribes hold promise for reducing clinician burden, rigorous and ongoing evaluation of their quality is essential to ensure that these tools enhance rather than compromise the quality of clinical care.”
AI Scribe Use is Widespread
Taylor N. Anderson, MD, a clinical informatics fellow at Oregon Health & Science University, Portland, is familiar with the study findings and noted that the use of AI scribes in medicine has grown rapidly. All major health organizations are either using it or facing “enormous pressure” from clinicians to do so, she told Federal Practitioner.
Previous research has linked the use of AI scribes for clinical notes to less electronic health record usage and documentation time for clinicians, leading to more time for patient visits. Still, the quality of clinical notes written by AI is “quite variable across vendors,” Anderson said.
Anderson led a 2025 study that examined 5 AI scribe platforms and found an average of 3.0 errors per case with “potential for moderate-to-severe harm.”
For the new study on the simulated cases, part of a VHA-sponsored “technology sprint” via Challenge.gov, researchers developed audio descriptions of 5 clinical cases reflecting common patient encounters in primary care: acute low back pain, chest pain, a new diagnosis of diabetes, a pharmacy consultation, and a follow-up with a nurse case manager for heart failure.
Two cases included non-English accents, 1 included background noise, and 1 featured speech through a medical mask. All the “patients” were played by what the authors described as “trained standardized patient actors.”
For each case, 3 humans and 11 AI scribe programs produced clinical notes. The clinical notes were then evaluated by 6 raters.
Researchers found that AI scribe-generated notes scored worse than human-generated notes across all 10 domains of the modified PDQI-9 (accuracy, thoroughness, usefulness, organization, comprehensiveness, succinctness, synthesization, internal consistency, and freedom from hallucination and bias).
There were especially large gaps between the AI and human notes in the domains of thoroughness, organization, and usefulness. Even wider gaps were observed for the encounters with noise and mask usage.
“These findings highlight that although ambient AI scribes can generate complete notes, the overall quality remains broadly below that of human-authored documentation,” the authors wrote.
No Comparison Between AI Scribes
The researchers noted that “given contractual limitations, we cannot interpret the results for specific vendors.” They also noted that the study did not use professional scribes, who may produce even higher-quality results, and the humans were not producing notes in a real-world clinical environment.
Anderson, the clinical informatics fellow, pointed out that the study does not examine the common scenario in which a clinician edits notes produced by an AI scribe. In fact, she said, there is no current research on this, failing to examine “the postediting note that would actually go into the chart.”
In an accompanying commentary, collaborative scientist Aaron Tierney, PhD, and Kristine Lee, MD, an associate executive director, both with the Permanente Medical Group, California, called for future research to focus on “real-world performance, promote the development of documentation policies that prioritize patient care over billing requirements, and systematically incorporate patient perspectives into assessments of quality.”
Why AI Misses the Mark
In an interview with Federal Practitioner, AI researcher Maxim Topaz, PhD, RN, MA, an associate professor of Nursing and Data Science at Columbia University School of Nursing, New York City, who is familiar with the study but did not participate in it, praised the research.
He pointed out that AI has trouble accurately representing clinical encounters because they “tend to fill gaps with plausible-sounding language, which can mask omissions and make errors harder to catch.” Also, “ambient scribes can only document what is verbalized aloud. Physical exam findings the clinician notices but does not narrate, nonverbal cues, and patient-initiated concerns that drift past in conversation are systematically underrepresented.”
Moving forward, Topaz advised clinicians to “treat AI-generated notes as a first draft, not a finished product. Read them carefully, especially for omissions, which the current evidence suggests are by far the most common error type and which are harder to spot than fabrications because the surrounding note still reads coherently.”
Two study authors disclosed employment by the US Department of Veterans Affairs. Other authors had no disclosures. The commentary authors have no disclosures. Anderson has no disclosures. Topaz discloses
Artificial intelligence (AI) scribes produced lower-quality documentation of clinical notes than human clinicians, and especially struggled in settings with background noise or clinicians wearing masks, a new Veterans Health Administration (VHA) study finds.
In 5 simulated clinical cases, notes written by various AI programs scored lower than reports produced by humans on the modified Physician Documentation Quality Instrument (PDQI-9), a measurement of note quality scale, reported Ashok Reddy, MD, MSc, of the University of Washington and Veterans Affairs Puget Sound Health Care System, Seattle, et al in the April issue of Annals of Internal Medicine.
AI scribes scored lower compared with humans across all domains, including accuracy, thoroughness, and usefulness. There was an especially large gap in scores on the 50-point PDQI-9 in an acute low back pain case (human, 43.8 points; AI, 20.3 points; difference, 23.5 points).
“For clinicians, AI scribes should be regarded as tools for generating draft documentation that requires review and editing, rather than as a substitute for clinician-authored notes,” the authors wrote. “Although ambient AI scribes hold promise for reducing clinician burden, rigorous and ongoing evaluation of their quality is essential to ensure that these tools enhance rather than compromise the quality of clinical care.”
AI Scribe Use is Widespread
Taylor N. Anderson, MD, a clinical informatics fellow at Oregon Health & Science University, Portland, is familiar with the study findings and noted that the use of AI scribes in medicine has grown rapidly. All major health organizations are either using it or facing “enormous pressure” from clinicians to do so, she told Federal Practitioner.
Previous research has linked the use of AI scribes for clinical notes to less electronic health record usage and documentation time for clinicians, leading to more time for patient visits. Still, the quality of clinical notes written by AI is “quite variable across vendors,” Anderson said.
Anderson led a 2025 study that examined 5 AI scribe platforms and found an average of 3.0 errors per case with “potential for moderate-to-severe harm.”
For the new study on the simulated cases, part of a VHA-sponsored “technology sprint” via Challenge.gov, researchers developed audio descriptions of 5 clinical cases reflecting common patient encounters in primary care: acute low back pain, chest pain, a new diagnosis of diabetes, a pharmacy consultation, and a follow-up with a nurse case manager for heart failure.
Two cases included non-English accents, 1 included background noise, and 1 featured speech through a medical mask. All the “patients” were played by what the authors described as “trained standardized patient actors.”
For each case, 3 humans and 11 AI scribe programs produced clinical notes. The clinical notes were then evaluated by 6 raters.
Researchers found that AI scribe-generated notes scored worse than human-generated notes across all 10 domains of the modified PDQI-9 (accuracy, thoroughness, usefulness, organization, comprehensiveness, succinctness, synthesization, internal consistency, and freedom from hallucination and bias).
There were especially large gaps between the AI and human notes in the domains of thoroughness, organization, and usefulness. Even wider gaps were observed for the encounters with noise and mask usage.
“These findings highlight that although ambient AI scribes can generate complete notes, the overall quality remains broadly below that of human-authored documentation,” the authors wrote.
No Comparison Between AI Scribes
The researchers noted that “given contractual limitations, we cannot interpret the results for specific vendors.” They also noted that the study did not use professional scribes, who may produce even higher-quality results, and the humans were not producing notes in a real-world clinical environment.
Anderson, the clinical informatics fellow, pointed out that the study does not examine the common scenario in which a clinician edits notes produced by an AI scribe. In fact, she said, there is no current research on this, failing to examine “the postediting note that would actually go into the chart.”
In an accompanying commentary, collaborative scientist Aaron Tierney, PhD, and Kristine Lee, MD, an associate executive director, both with the Permanente Medical Group, California, called for future research to focus on “real-world performance, promote the development of documentation policies that prioritize patient care over billing requirements, and systematically incorporate patient perspectives into assessments of quality.”
Why AI Misses the Mark
In an interview with Federal Practitioner, AI researcher Maxim Topaz, PhD, RN, MA, an associate professor of Nursing and Data Science at Columbia University School of Nursing, New York City, who is familiar with the study but did not participate in it, praised the research.
He pointed out that AI has trouble accurately representing clinical encounters because they “tend to fill gaps with plausible-sounding language, which can mask omissions and make errors harder to catch.” Also, “ambient scribes can only document what is verbalized aloud. Physical exam findings the clinician notices but does not narrate, nonverbal cues, and patient-initiated concerns that drift past in conversation are systematically underrepresented.”
Moving forward, Topaz advised clinicians to “treat AI-generated notes as a first draft, not a finished product. Read them carefully, especially for omissions, which the current evidence suggests are by far the most common error type and which are harder to spot than fabrications because the surrounding note still reads coherently.”
Two study authors disclosed employment by the US Department of Veterans Affairs. Other authors had no disclosures. The commentary authors have no disclosures. Anderson has no disclosures. Topaz discloses
Artificial intelligence (AI) scribes produced lower-quality documentation of clinical notes than human clinicians, and especially struggled in settings with background noise or clinicians wearing masks, a new Veterans Health Administration (VHA) study finds.
In 5 simulated clinical cases, notes written by various AI programs scored lower than reports produced by humans on the modified Physician Documentation Quality Instrument (PDQI-9), a measurement of note quality scale, reported Ashok Reddy, MD, MSc, of the University of Washington and Veterans Affairs Puget Sound Health Care System, Seattle, et al in the April issue of Annals of Internal Medicine.
AI scribes scored lower compared with humans across all domains, including accuracy, thoroughness, and usefulness. There was an especially large gap in scores on the 50-point PDQI-9 in an acute low back pain case (human, 43.8 points; AI, 20.3 points; difference, 23.5 points).
“For clinicians, AI scribes should be regarded as tools for generating draft documentation that requires review and editing, rather than as a substitute for clinician-authored notes,” the authors wrote. “Although ambient AI scribes hold promise for reducing clinician burden, rigorous and ongoing evaluation of their quality is essential to ensure that these tools enhance rather than compromise the quality of clinical care.”
AI Scribe Use is Widespread
Taylor N. Anderson, MD, a clinical informatics fellow at Oregon Health & Science University, Portland, is familiar with the study findings and noted that the use of AI scribes in medicine has grown rapidly. All major health organizations are either using it or facing “enormous pressure” from clinicians to do so, she told Federal Practitioner.
Previous research has linked the use of AI scribes for clinical notes to less electronic health record usage and documentation time for clinicians, leading to more time for patient visits. Still, the quality of clinical notes written by AI is “quite variable across vendors,” Anderson said.
Anderson led a 2025 study that examined 5 AI scribe platforms and found an average of 3.0 errors per case with “potential for moderate-to-severe harm.”
For the new study on the simulated cases, part of a VHA-sponsored “technology sprint” via Challenge.gov, researchers developed audio descriptions of 5 clinical cases reflecting common patient encounters in primary care: acute low back pain, chest pain, a new diagnosis of diabetes, a pharmacy consultation, and a follow-up with a nurse case manager for heart failure.
Two cases included non-English accents, 1 included background noise, and 1 featured speech through a medical mask. All the “patients” were played by what the authors described as “trained standardized patient actors.”
For each case, 3 humans and 11 AI scribe programs produced clinical notes. The clinical notes were then evaluated by 6 raters.
Researchers found that AI scribe-generated notes scored worse than human-generated notes across all 10 domains of the modified PDQI-9 (accuracy, thoroughness, usefulness, organization, comprehensiveness, succinctness, synthesization, internal consistency, and freedom from hallucination and bias).
There were especially large gaps between the AI and human notes in the domains of thoroughness, organization, and usefulness. Even wider gaps were observed for the encounters with noise and mask usage.
“These findings highlight that although ambient AI scribes can generate complete notes, the overall quality remains broadly below that of human-authored documentation,” the authors wrote.
No Comparison Between AI Scribes
The researchers noted that “given contractual limitations, we cannot interpret the results for specific vendors.” They also noted that the study did not use professional scribes, who may produce even higher-quality results, and the humans were not producing notes in a real-world clinical environment.
Anderson, the clinical informatics fellow, pointed out that the study does not examine the common scenario in which a clinician edits notes produced by an AI scribe. In fact, she said, there is no current research on this, failing to examine “the postediting note that would actually go into the chart.”
In an accompanying commentary, collaborative scientist Aaron Tierney, PhD, and Kristine Lee, MD, an associate executive director, both with the Permanente Medical Group, California, called for future research to focus on “real-world performance, promote the development of documentation policies that prioritize patient care over billing requirements, and systematically incorporate patient perspectives into assessments of quality.”
Why AI Misses the Mark
In an interview with Federal Practitioner, AI researcher Maxim Topaz, PhD, RN, MA, an associate professor of Nursing and Data Science at Columbia University School of Nursing, New York City, who is familiar with the study but did not participate in it, praised the research.
He pointed out that AI has trouble accurately representing clinical encounters because they “tend to fill gaps with plausible-sounding language, which can mask omissions and make errors harder to catch.” Also, “ambient scribes can only document what is verbalized aloud. Physical exam findings the clinician notices but does not narrate, nonverbal cues, and patient-initiated concerns that drift past in conversation are systematically underrepresented.”
Moving forward, Topaz advised clinicians to “treat AI-generated notes as a first draft, not a finished product. Read them carefully, especially for omissions, which the current evidence suggests are by far the most common error type and which are harder to spot than fabrications because the surrounding note still reads coherently.”
Two study authors disclosed employment by the US Department of Veterans Affairs. Other authors had no disclosures. The commentary authors have no disclosures. Anderson has no disclosures. Topaz discloses
VA Lags on Cardiac Rehabilitation
Cardiac rehabilitation (CR) remains dramatically underused among US veterans, as < 11% of eligible patients attend a single session and usage appears to be declining over time, a recently published retrospective cohort study reported.
CR use is much lower among eligible patients across the US Department of Veterans Affairs (VA) compared with Medicare (10.4% vs. 28%, respectively), reported researchers at Veterans Affairs Connecticut Healthcare System and Yale School of Medicine, in JACC: Advances.
The overall CR rate in the VA was lower than the 13.2% reported in a 2018 study. And while there was no significant difference in use between men and women, veterans from the poorest neighborhoods were less likely to take advantage of CR compared with veterans from the wealthiest neighborhoods (adjusted odds ratio, 0.82; P < .001).
“As providers, the time to act is now,” Merilyn Varghese, MD, MSc, said in an interview with Federal Practitioner. “We need to urgently get more of our veterans to cardiac rehab.”
As Varghese explained, “CR is a preventive intervention that has been shown to improve quality of life and reduce mortality and hospitalizations for patients with specific cardiac conditions.”
Patients may be eligible if they have experienced myocardial infarction (MI), percutaneous coronary intervention (PCI), coronary artery bypass surgery (CABG), heart transplant, valve surgery, stable angina, or stable heart failure.
“CR combines multiple aspects of cardiac care such as exercise training, medication management, and behavioral assessments,” Varghese said. “For example, patients who have had a heart attack may have challenges in getting back to an exercise routine, managing new medications, and adjusting to life after such an event. CR can help bridge the gap between hospital to home.” In-person CR typically includes 3 sessions per week for 12 weeks.
In 2024, a systematic review and meta-analysis reported that CR reduces all-cause mortality (relative risk, 0.74): “These results support the utilization of CR as a critical element in the management of further secondary prevention of CVDs (cardiovascular diseases).”
Examining VA Data
Researchers conducted the 2026 study “to better understand the current landscape of CR among veterans, particularly among women veterans who comprise a significant part of the veteran population but have previously been underrepresented in research,” Varghese said.
“Women veterans also share a different burden of cardiovascular risk factors, so understanding CR participation among both women and men veterans was of particular interest.”
The study tracked 82,496 VA-enrolled veterans eligible for CR from 2021-2023 (3.6% women). Average age of participants were 64.0 years among women and 71.5 years among men. Among women, 58.3% were White, and 31.8% were Black, and 2.24% were Asian. Among men, 71.9% were White, 18.8% were Black, and 2.3% were Asian.
The rates of CR participation were low among both men (10.4%) and women (10.2%). Older people and Black patients were less likely to take part in CR than younger people and White patients, according to the study. Those who underwent CABG and PCI were more likely to participate in CR compared with those who had heart attacks only.
As for the gap in use between the wealthiest and poorest neighborhoods, Varghese said: “Area deprivation may compound some of the other barriers to CR access, including transportation difficulties, work responsibilities, and out-of-pocket costs.”
How can CR uptake be improved? “A key first step is understanding who can be referred, and second, to spend time discussing the importance of attending with veterans,” Varghese said. “Studies have shown that provider engagement and championing of CR are important positive facilitators that encourage CR participation.
“The VA has been at the forefront of innovation with the home-based CR program that offers veterans a way to attend CR remotely,” she added. “Expanding such novel methods of CR delivery is likely part of the solution to expand CR access.”
Outside Perspective: Make Referrals the Default
Justin Bachmann, MD, MPH, staff physician and research scientist at VA Tennessee Valley Healthcare System, told Federal Practitioner that CR is an American College of Cardiology/American Heart Association Class I recommended secondary prevention therapy following MI, PCI, and CABG “with strong evidence for reduced cardiovascular mortality and improved function and quality of life.”
Still, CR “has been persistently underused for decades as travel, cost, scheduling, and uneven geographic capacity create real logistical barriers,” said Bachmann, who serves as the medical director of a VA Office of Rural Health home-based CR program.
Bachmann praised the study methodology and offered this advice to colleagues: “Embed CR referral in the post-MI, post-PCI, and post-CABG order sets so that referral is the default. Scale home-based CR well beyond the roughly 40 sites where it is currently available, and track facility-level referral and enrollment rates as quality measures.”
Preventive cardiology specialist Randal J. Thomas, MD, professor of Medicine at the Mayo Clinic in Rochester, Minn., echoed the importance of physician referral to Federal Practitioner.
“Patients can’t actually participate [directly] in most programs. They must have a physician referral,” he said. “The physician referral and the strength of referral is key. If a physician says, ‘You can go there if you want, but it’s not that important,’ the patients aren’t going to go.”
Outside Perspective: VA Deserves Blame
“The VA lags far behind most medical systems,” according to Quinn R. Pack, MD, associate professor of medicine at the University of Massachusetts Chan Medical School-Baystate. “Some of this is probably the patient population—more mental health problems, more smoking, more disease. But I’d squarely put most of this on the VA health system. They haven’t created the systems of care that make attending cardiac rehabilitation easy, reliable, and consistent.”
He noted that that automatic referral combined with a bedside visit by a liaison such as a representative of a CR program can double or triple enrollment.
“When physicians and nurses really encourage patients to go [to CR], these words are powerful,” Pack said. “When a patient enrolls in cardiac rehabilitation, we help them form new habits of exercise.”
No study fundings are reported. The Varghese discloses a relationship with the Veterans Health Administration. Other study authors had no disclosures. Bachmann disclosed a relationship with the VA. Pack and Thomas have no disclosures.
Cardiac rehabilitation (CR) remains dramatically underused among US veterans, as < 11% of eligible patients attend a single session and usage appears to be declining over time, a recently published retrospective cohort study reported.
CR use is much lower among eligible patients across the US Department of Veterans Affairs (VA) compared with Medicare (10.4% vs. 28%, respectively), reported researchers at Veterans Affairs Connecticut Healthcare System and Yale School of Medicine, in JACC: Advances.
The overall CR rate in the VA was lower than the 13.2% reported in a 2018 study. And while there was no significant difference in use between men and women, veterans from the poorest neighborhoods were less likely to take advantage of CR compared with veterans from the wealthiest neighborhoods (adjusted odds ratio, 0.82; P < .001).
“As providers, the time to act is now,” Merilyn Varghese, MD, MSc, said in an interview with Federal Practitioner. “We need to urgently get more of our veterans to cardiac rehab.”
As Varghese explained, “CR is a preventive intervention that has been shown to improve quality of life and reduce mortality and hospitalizations for patients with specific cardiac conditions.”
Patients may be eligible if they have experienced myocardial infarction (MI), percutaneous coronary intervention (PCI), coronary artery bypass surgery (CABG), heart transplant, valve surgery, stable angina, or stable heart failure.
“CR combines multiple aspects of cardiac care such as exercise training, medication management, and behavioral assessments,” Varghese said. “For example, patients who have had a heart attack may have challenges in getting back to an exercise routine, managing new medications, and adjusting to life after such an event. CR can help bridge the gap between hospital to home.” In-person CR typically includes 3 sessions per week for 12 weeks.
In 2024, a systematic review and meta-analysis reported that CR reduces all-cause mortality (relative risk, 0.74): “These results support the utilization of CR as a critical element in the management of further secondary prevention of CVDs (cardiovascular diseases).”
Examining VA Data
Researchers conducted the 2026 study “to better understand the current landscape of CR among veterans, particularly among women veterans who comprise a significant part of the veteran population but have previously been underrepresented in research,” Varghese said.
“Women veterans also share a different burden of cardiovascular risk factors, so understanding CR participation among both women and men veterans was of particular interest.”
The study tracked 82,496 VA-enrolled veterans eligible for CR from 2021-2023 (3.6% women). Average age of participants were 64.0 years among women and 71.5 years among men. Among women, 58.3% were White, and 31.8% were Black, and 2.24% were Asian. Among men, 71.9% were White, 18.8% were Black, and 2.3% were Asian.
The rates of CR participation were low among both men (10.4%) and women (10.2%). Older people and Black patients were less likely to take part in CR than younger people and White patients, according to the study. Those who underwent CABG and PCI were more likely to participate in CR compared with those who had heart attacks only.
As for the gap in use between the wealthiest and poorest neighborhoods, Varghese said: “Area deprivation may compound some of the other barriers to CR access, including transportation difficulties, work responsibilities, and out-of-pocket costs.”
How can CR uptake be improved? “A key first step is understanding who can be referred, and second, to spend time discussing the importance of attending with veterans,” Varghese said. “Studies have shown that provider engagement and championing of CR are important positive facilitators that encourage CR participation.
“The VA has been at the forefront of innovation with the home-based CR program that offers veterans a way to attend CR remotely,” she added. “Expanding such novel methods of CR delivery is likely part of the solution to expand CR access.”
Outside Perspective: Make Referrals the Default
Justin Bachmann, MD, MPH, staff physician and research scientist at VA Tennessee Valley Healthcare System, told Federal Practitioner that CR is an American College of Cardiology/American Heart Association Class I recommended secondary prevention therapy following MI, PCI, and CABG “with strong evidence for reduced cardiovascular mortality and improved function and quality of life.”
Still, CR “has been persistently underused for decades as travel, cost, scheduling, and uneven geographic capacity create real logistical barriers,” said Bachmann, who serves as the medical director of a VA Office of Rural Health home-based CR program.
Bachmann praised the study methodology and offered this advice to colleagues: “Embed CR referral in the post-MI, post-PCI, and post-CABG order sets so that referral is the default. Scale home-based CR well beyond the roughly 40 sites where it is currently available, and track facility-level referral and enrollment rates as quality measures.”
Preventive cardiology specialist Randal J. Thomas, MD, professor of Medicine at the Mayo Clinic in Rochester, Minn., echoed the importance of physician referral to Federal Practitioner.
“Patients can’t actually participate [directly] in most programs. They must have a physician referral,” he said. “The physician referral and the strength of referral is key. If a physician says, ‘You can go there if you want, but it’s not that important,’ the patients aren’t going to go.”
Outside Perspective: VA Deserves Blame
“The VA lags far behind most medical systems,” according to Quinn R. Pack, MD, associate professor of medicine at the University of Massachusetts Chan Medical School-Baystate. “Some of this is probably the patient population—more mental health problems, more smoking, more disease. But I’d squarely put most of this on the VA health system. They haven’t created the systems of care that make attending cardiac rehabilitation easy, reliable, and consistent.”
He noted that that automatic referral combined with a bedside visit by a liaison such as a representative of a CR program can double or triple enrollment.
“When physicians and nurses really encourage patients to go [to CR], these words are powerful,” Pack said. “When a patient enrolls in cardiac rehabilitation, we help them form new habits of exercise.”
No study fundings are reported. The Varghese discloses a relationship with the Veterans Health Administration. Other study authors had no disclosures. Bachmann disclosed a relationship with the VA. Pack and Thomas have no disclosures.
Cardiac rehabilitation (CR) remains dramatically underused among US veterans, as < 11% of eligible patients attend a single session and usage appears to be declining over time, a recently published retrospective cohort study reported.
CR use is much lower among eligible patients across the US Department of Veterans Affairs (VA) compared with Medicare (10.4% vs. 28%, respectively), reported researchers at Veterans Affairs Connecticut Healthcare System and Yale School of Medicine, in JACC: Advances.
The overall CR rate in the VA was lower than the 13.2% reported in a 2018 study. And while there was no significant difference in use between men and women, veterans from the poorest neighborhoods were less likely to take advantage of CR compared with veterans from the wealthiest neighborhoods (adjusted odds ratio, 0.82; P < .001).
“As providers, the time to act is now,” Merilyn Varghese, MD, MSc, said in an interview with Federal Practitioner. “We need to urgently get more of our veterans to cardiac rehab.”
As Varghese explained, “CR is a preventive intervention that has been shown to improve quality of life and reduce mortality and hospitalizations for patients with specific cardiac conditions.”
Patients may be eligible if they have experienced myocardial infarction (MI), percutaneous coronary intervention (PCI), coronary artery bypass surgery (CABG), heart transplant, valve surgery, stable angina, or stable heart failure.
“CR combines multiple aspects of cardiac care such as exercise training, medication management, and behavioral assessments,” Varghese said. “For example, patients who have had a heart attack may have challenges in getting back to an exercise routine, managing new medications, and adjusting to life after such an event. CR can help bridge the gap between hospital to home.” In-person CR typically includes 3 sessions per week for 12 weeks.
In 2024, a systematic review and meta-analysis reported that CR reduces all-cause mortality (relative risk, 0.74): “These results support the utilization of CR as a critical element in the management of further secondary prevention of CVDs (cardiovascular diseases).”
Examining VA Data
Researchers conducted the 2026 study “to better understand the current landscape of CR among veterans, particularly among women veterans who comprise a significant part of the veteran population but have previously been underrepresented in research,” Varghese said.
“Women veterans also share a different burden of cardiovascular risk factors, so understanding CR participation among both women and men veterans was of particular interest.”
The study tracked 82,496 VA-enrolled veterans eligible for CR from 2021-2023 (3.6% women). Average age of participants were 64.0 years among women and 71.5 years among men. Among women, 58.3% were White, and 31.8% were Black, and 2.24% were Asian. Among men, 71.9% were White, 18.8% were Black, and 2.3% were Asian.
The rates of CR participation were low among both men (10.4%) and women (10.2%). Older people and Black patients were less likely to take part in CR than younger people and White patients, according to the study. Those who underwent CABG and PCI were more likely to participate in CR compared with those who had heart attacks only.
As for the gap in use between the wealthiest and poorest neighborhoods, Varghese said: “Area deprivation may compound some of the other barriers to CR access, including transportation difficulties, work responsibilities, and out-of-pocket costs.”
How can CR uptake be improved? “A key first step is understanding who can be referred, and second, to spend time discussing the importance of attending with veterans,” Varghese said. “Studies have shown that provider engagement and championing of CR are important positive facilitators that encourage CR participation.
“The VA has been at the forefront of innovation with the home-based CR program that offers veterans a way to attend CR remotely,” she added. “Expanding such novel methods of CR delivery is likely part of the solution to expand CR access.”
Outside Perspective: Make Referrals the Default
Justin Bachmann, MD, MPH, staff physician and research scientist at VA Tennessee Valley Healthcare System, told Federal Practitioner that CR is an American College of Cardiology/American Heart Association Class I recommended secondary prevention therapy following MI, PCI, and CABG “with strong evidence for reduced cardiovascular mortality and improved function and quality of life.”
Still, CR “has been persistently underused for decades as travel, cost, scheduling, and uneven geographic capacity create real logistical barriers,” said Bachmann, who serves as the medical director of a VA Office of Rural Health home-based CR program.
Bachmann praised the study methodology and offered this advice to colleagues: “Embed CR referral in the post-MI, post-PCI, and post-CABG order sets so that referral is the default. Scale home-based CR well beyond the roughly 40 sites where it is currently available, and track facility-level referral and enrollment rates as quality measures.”
Preventive cardiology specialist Randal J. Thomas, MD, professor of Medicine at the Mayo Clinic in Rochester, Minn., echoed the importance of physician referral to Federal Practitioner.
“Patients can’t actually participate [directly] in most programs. They must have a physician referral,” he said. “The physician referral and the strength of referral is key. If a physician says, ‘You can go there if you want, but it’s not that important,’ the patients aren’t going to go.”
Outside Perspective: VA Deserves Blame
“The VA lags far behind most medical systems,” according to Quinn R. Pack, MD, associate professor of medicine at the University of Massachusetts Chan Medical School-Baystate. “Some of this is probably the patient population—more mental health problems, more smoking, more disease. But I’d squarely put most of this on the VA health system. They haven’t created the systems of care that make attending cardiac rehabilitation easy, reliable, and consistent.”
He noted that that automatic referral combined with a bedside visit by a liaison such as a representative of a CR program can double or triple enrollment.
“When physicians and nurses really encourage patients to go [to CR], these words are powerful,” Pack said. “When a patient enrolls in cardiac rehabilitation, we help them form new habits of exercise.”
No study fundings are reported. The Varghese discloses a relationship with the Veterans Health Administration. Other study authors had no disclosures. Bachmann disclosed a relationship with the VA. Pack and Thomas have no disclosures.
High VA Telehealth Use Linked to Reduced Vaccination Rates
Quality measures for primary care in the US Department of Veterans Affairs (VA) remained stable when telehealth was mixed with in-person visits, but influenza vaccination fell among patients who relied on online visits the most, a retrospective cohort study finds.
Analysis of the medical records for 744,599 veterans from federal fiscal years 2022 and 2023 revealed that patients aged 19-65 years who relied on telehealth for at least half of their primary care visits were less likely to receive an influenza vaccine (37.9%) compared with those seen only in person (50.0%, P < .001). The study was lead by researchers at VA Puget Sound and published in JAMA Network Open.
There was also an influenza vaccination gap in patients aged ≥ 66 years: 62.8% in patients who received some care via telehealth telehealth vs 71.5% seen only in person, respectively (P < .001).
“Our study showed that primary care quality at the VA is quite high,” Jonathan Staloff, MD, MSc, a family medicine physician with VA Puget Sound told Federal Practitioner. “And we found that for almost all quality measures, having a low proportion of care via telehealth was associated with the same quality as in-person care.”
As Staloff explained, “telehealth in primary care, as well as in general, has emerged as an additional means of preserving access to care for veterans. Evidence suggests that veterans have a high degree of satisfaction with telehealth but it’s mixed as it relates to quality outcome differences between those who receive any via telehealth vs none.”
For the study, Staloff said, “we wanted to see if there was a dose-response relationship between telehealth utilization and care quality and if certain hybrid models could help optimize quality of care. To our knowledge, this study was the first national evaluation to investigate primary care telehealth and care quality in this way.”
Reassuring Findings About Low Telehealth Use
For the study, researchers tracked a national sample of patient data from the Veterans Health Administration (VHA) Support Service Center Capital Assets Databases, Primary Care Management Module, and VHA Corporate Data Warehouse (mean age, 65 years; 86% male; 63% White, 22% Black, 10% Hispanic).
The study defined categories of primary-care telehealth use as no telehealth, low telehealth (> 0.0% to < 28.6%), intermediate telehealth (28.6% to < 50.0%), and high telehealth (> 50.0%).
Highest Telehealth Use Raises Red Flags
The differences in influenza vaccine rates between the no-telehealth and high-telehealth groups held up in an adjusted analysis.
The study found small but statistically significant worsening of several quality measures in the high-telehealth use vs no-telehealth use cohorts: hypertension control, statin therapy and adherence, and annual screening for depression, alcohol use, and tobacco use.
The study cites limitations such as reliance on patients with ≥ 3 or more evaluation-and- management visits and lack of information about influenza vaccines delivered outside the VA.
In a statement, VA Telehealth Services said it is “encouraged” the study demonstrates “equivalence in many clinical measures among veterans using telehealth. This study reinforces the potential of telehealth to provide high-quality health care to veterans.”
The organization added that it’s “committed to better understanding potential gaps highlighted in this study,” and “it is critical that research databases capture care rendered outside VA … and whether care was offered during a telehealth visit.”
Batching In-Person Services May Be Helpful
As for messages from the study for clinicians, Staloff said there are some preventive care measures that may be more difficult to deliver through telehealth.
“Clinicians should consider batching these in-person services for patients that have a high reliance on telehealth when they have an opportunity to see these patients in-person,” Staloff said. “Health systems may need new workflows to optimize hybrid care, particularly for those that receive most of their care via telehealth.”
Outside Perspective: ‘Access is Not the Same as Quality’
After reviewing the study findings, Ilana Graetz, PhD, a professor who studies health policy at the Emory University Rollins School of Public Health, praised the research design and said the results overall are “more reassuring than alarming.” However, she did caution that there could potentially be ways these patients differ that could not be categorized by the data.
“Patients with higher telehealth use may differ from those with lower telehealth use in important ways not fully captured in the data — barriers to in-person care, the complexity of the visit, patient preferences, or care received outside the system,” Graetz said.
As for the influenza vaccine, Graetz said patients need to be physically present: “Patients seen mostly by telehealth will have fewer opportunities to receive any preventive care that can only be delivered in person.”
Graetz said the study is timely given ongoing debates over COVID-19 pandemic-era telehealth flexibilities.
“The findings suggest that telehealth can function well as part of a hybrid primary care model,” she said, “but health systems still need to ensure that preventive services, chronic disease management, and follow-up care are not lost in the shift to virtual care.”
VHA Primary Care Analytics Team supported the study with funding from the VHA Office of Primary Care. Staloff has no disclosures. One coauthor disclosed a relationship with the US Department of Veterans Affairs.
Graetz disclosed relationships the Donaghue Foundation, Pfizer, PRIME Education, and the National Institutes of Health.
Quality measures for primary care in the US Department of Veterans Affairs (VA) remained stable when telehealth was mixed with in-person visits, but influenza vaccination fell among patients who relied on online visits the most, a retrospective cohort study finds.
Analysis of the medical records for 744,599 veterans from federal fiscal years 2022 and 2023 revealed that patients aged 19-65 years who relied on telehealth for at least half of their primary care visits were less likely to receive an influenza vaccine (37.9%) compared with those seen only in person (50.0%, P < .001). The study was lead by researchers at VA Puget Sound and published in JAMA Network Open.
There was also an influenza vaccination gap in patients aged ≥ 66 years: 62.8% in patients who received some care via telehealth telehealth vs 71.5% seen only in person, respectively (P < .001).
“Our study showed that primary care quality at the VA is quite high,” Jonathan Staloff, MD, MSc, a family medicine physician with VA Puget Sound told Federal Practitioner. “And we found that for almost all quality measures, having a low proportion of care via telehealth was associated with the same quality as in-person care.”
As Staloff explained, “telehealth in primary care, as well as in general, has emerged as an additional means of preserving access to care for veterans. Evidence suggests that veterans have a high degree of satisfaction with telehealth but it’s mixed as it relates to quality outcome differences between those who receive any via telehealth vs none.”
For the study, Staloff said, “we wanted to see if there was a dose-response relationship between telehealth utilization and care quality and if certain hybrid models could help optimize quality of care. To our knowledge, this study was the first national evaluation to investigate primary care telehealth and care quality in this way.”
Reassuring Findings About Low Telehealth Use
For the study, researchers tracked a national sample of patient data from the Veterans Health Administration (VHA) Support Service Center Capital Assets Databases, Primary Care Management Module, and VHA Corporate Data Warehouse (mean age, 65 years; 86% male; 63% White, 22% Black, 10% Hispanic).
The study defined categories of primary-care telehealth use as no telehealth, low telehealth (> 0.0% to < 28.6%), intermediate telehealth (28.6% to < 50.0%), and high telehealth (> 50.0%).
Highest Telehealth Use Raises Red Flags
The differences in influenza vaccine rates between the no-telehealth and high-telehealth groups held up in an adjusted analysis.
The study found small but statistically significant worsening of several quality measures in the high-telehealth use vs no-telehealth use cohorts: hypertension control, statin therapy and adherence, and annual screening for depression, alcohol use, and tobacco use.
The study cites limitations such as reliance on patients with ≥ 3 or more evaluation-and- management visits and lack of information about influenza vaccines delivered outside the VA.
In a statement, VA Telehealth Services said it is “encouraged” the study demonstrates “equivalence in many clinical measures among veterans using telehealth. This study reinforces the potential of telehealth to provide high-quality health care to veterans.”
The organization added that it’s “committed to better understanding potential gaps highlighted in this study,” and “it is critical that research databases capture care rendered outside VA … and whether care was offered during a telehealth visit.”
Batching In-Person Services May Be Helpful
As for messages from the study for clinicians, Staloff said there are some preventive care measures that may be more difficult to deliver through telehealth.
“Clinicians should consider batching these in-person services for patients that have a high reliance on telehealth when they have an opportunity to see these patients in-person,” Staloff said. “Health systems may need new workflows to optimize hybrid care, particularly for those that receive most of their care via telehealth.”
Outside Perspective: ‘Access is Not the Same as Quality’
After reviewing the study findings, Ilana Graetz, PhD, a professor who studies health policy at the Emory University Rollins School of Public Health, praised the research design and said the results overall are “more reassuring than alarming.” However, she did caution that there could potentially be ways these patients differ that could not be categorized by the data.
“Patients with higher telehealth use may differ from those with lower telehealth use in important ways not fully captured in the data — barriers to in-person care, the complexity of the visit, patient preferences, or care received outside the system,” Graetz said.
As for the influenza vaccine, Graetz said patients need to be physically present: “Patients seen mostly by telehealth will have fewer opportunities to receive any preventive care that can only be delivered in person.”
Graetz said the study is timely given ongoing debates over COVID-19 pandemic-era telehealth flexibilities.
“The findings suggest that telehealth can function well as part of a hybrid primary care model,” she said, “but health systems still need to ensure that preventive services, chronic disease management, and follow-up care are not lost in the shift to virtual care.”
VHA Primary Care Analytics Team supported the study with funding from the VHA Office of Primary Care. Staloff has no disclosures. One coauthor disclosed a relationship with the US Department of Veterans Affairs.
Graetz disclosed relationships the Donaghue Foundation, Pfizer, PRIME Education, and the National Institutes of Health.
Quality measures for primary care in the US Department of Veterans Affairs (VA) remained stable when telehealth was mixed with in-person visits, but influenza vaccination fell among patients who relied on online visits the most, a retrospective cohort study finds.
Analysis of the medical records for 744,599 veterans from federal fiscal years 2022 and 2023 revealed that patients aged 19-65 years who relied on telehealth for at least half of their primary care visits were less likely to receive an influenza vaccine (37.9%) compared with those seen only in person (50.0%, P < .001). The study was lead by researchers at VA Puget Sound and published in JAMA Network Open.
There was also an influenza vaccination gap in patients aged ≥ 66 years: 62.8% in patients who received some care via telehealth telehealth vs 71.5% seen only in person, respectively (P < .001).
“Our study showed that primary care quality at the VA is quite high,” Jonathan Staloff, MD, MSc, a family medicine physician with VA Puget Sound told Federal Practitioner. “And we found that for almost all quality measures, having a low proportion of care via telehealth was associated with the same quality as in-person care.”
As Staloff explained, “telehealth in primary care, as well as in general, has emerged as an additional means of preserving access to care for veterans. Evidence suggests that veterans have a high degree of satisfaction with telehealth but it’s mixed as it relates to quality outcome differences between those who receive any via telehealth vs none.”
For the study, Staloff said, “we wanted to see if there was a dose-response relationship between telehealth utilization and care quality and if certain hybrid models could help optimize quality of care. To our knowledge, this study was the first national evaluation to investigate primary care telehealth and care quality in this way.”
Reassuring Findings About Low Telehealth Use
For the study, researchers tracked a national sample of patient data from the Veterans Health Administration (VHA) Support Service Center Capital Assets Databases, Primary Care Management Module, and VHA Corporate Data Warehouse (mean age, 65 years; 86% male; 63% White, 22% Black, 10% Hispanic).
The study defined categories of primary-care telehealth use as no telehealth, low telehealth (> 0.0% to < 28.6%), intermediate telehealth (28.6% to < 50.0%), and high telehealth (> 50.0%).
Highest Telehealth Use Raises Red Flags
The differences in influenza vaccine rates between the no-telehealth and high-telehealth groups held up in an adjusted analysis.
The study found small but statistically significant worsening of several quality measures in the high-telehealth use vs no-telehealth use cohorts: hypertension control, statin therapy and adherence, and annual screening for depression, alcohol use, and tobacco use.
The study cites limitations such as reliance on patients with ≥ 3 or more evaluation-and- management visits and lack of information about influenza vaccines delivered outside the VA.
In a statement, VA Telehealth Services said it is “encouraged” the study demonstrates “equivalence in many clinical measures among veterans using telehealth. This study reinforces the potential of telehealth to provide high-quality health care to veterans.”
The organization added that it’s “committed to better understanding potential gaps highlighted in this study,” and “it is critical that research databases capture care rendered outside VA … and whether care was offered during a telehealth visit.”
Batching In-Person Services May Be Helpful
As for messages from the study for clinicians, Staloff said there are some preventive care measures that may be more difficult to deliver through telehealth.
“Clinicians should consider batching these in-person services for patients that have a high reliance on telehealth when they have an opportunity to see these patients in-person,” Staloff said. “Health systems may need new workflows to optimize hybrid care, particularly for those that receive most of their care via telehealth.”
Outside Perspective: ‘Access is Not the Same as Quality’
After reviewing the study findings, Ilana Graetz, PhD, a professor who studies health policy at the Emory University Rollins School of Public Health, praised the research design and said the results overall are “more reassuring than alarming.” However, she did caution that there could potentially be ways these patients differ that could not be categorized by the data.
“Patients with higher telehealth use may differ from those with lower telehealth use in important ways not fully captured in the data — barriers to in-person care, the complexity of the visit, patient preferences, or care received outside the system,” Graetz said.
As for the influenza vaccine, Graetz said patients need to be physically present: “Patients seen mostly by telehealth will have fewer opportunities to receive any preventive care that can only be delivered in person.”
Graetz said the study is timely given ongoing debates over COVID-19 pandemic-era telehealth flexibilities.
“The findings suggest that telehealth can function well as part of a hybrid primary care model,” she said, “but health systems still need to ensure that preventive services, chronic disease management, and follow-up care are not lost in the shift to virtual care.”
VHA Primary Care Analytics Team supported the study with funding from the VHA Office of Primary Care. Staloff has no disclosures. One coauthor disclosed a relationship with the US Department of Veterans Affairs.
Graetz disclosed relationships the Donaghue Foundation, Pfizer, PRIME Education, and the National Institutes of Health.
Evolving and Future Treatments for Follicular Lymphoma
Evolving and Future Treatments for Follicular Lymphoma
SAN FRANCISCO – Treatment for follicular lymphoma (FL) continues to evolve, according to a US Department of Veterans Affairs (VA) hematologist-oncologist, as second-line regimens emerge but the withdrawal of a recently approved agent complicates the picture.
“The future for our understanding and treatment of follicular lymphoma remains bright,” said Gerald Hsu, MD, PhD, of the University of California at San Francisco and the San Francisco VA Health Care System, during a presentation at the March Association of VA Hematology/Oncology (AVAHO) regional meeting on lymphoma.
By the Numbers
About 16,500 people in the US are diagnosed with FL each year. The median age of diagnosis is 64 years, and the 5-year survival rate from 2015-2021 was 89.0%, according to the National Cancer Institute.
FL is slow-growing and indolent, Hsu said.
“[That] means that we tend to see patients who are older when they are diagnosed,” he added. “They tend to live a long time, and they’re not usually curable.”
A better understanding of the biology of FL has allowed for the development of new markers and ways of measuring residual disease, Hsu said. Additional insight may allow clinicians to identify which patients could benefit most from specific therapies.
Frontline Options
Hsu highlighted the VA Oncology Clinical Pathway for FL, which offers step-by-step guidance regarding therapy and was updated in March 2026. “It walks you through the pathway, but it’s not something that you are beholden to,” he said.
If the patient has classic FL grades 1-3A, is not at risk of transformation to aggressive lymphoma, is not in stage 1 or continuous stage lymphoma, and is indicated for therapy, the guideline recommends lenalidomide plus rituximab (R2 or R-Len) or rituximab-bendamustine (R-Benda).
“There’s a lot of data to support R-Benda,” Hsu said, pointing to a pair of studies with large numbers of patients with FL. The 2013 StiL trial tracked > 500 patients with indolent or mantle cell lymphoma (46% high risk). Those on R-Benda displayed better progression-free survival (PFS) than those taking the combination rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP; hazard ratio [HR], 0.61).
In a 5-year update of the BRIGHT trial, which enrolled > 500 patients with indolent or mantle cell lymphoma (9% high risk), the R-Benda group had better PFS than patients on R-CHOP or rituximab plus cyclophosphamide, vincristine, and prednisone (HR, 0.61).
R2 is a somewhat newer regimen, Hsu said. Data from the 2018 RELEVANCE study (> 1000 patients) found R2 to be nonsuperior to 3 rituximab-plus-chemotherapy regimens but with a lower rate of grade 3-4 neutropenia (32% vs 50% for the other regimens).
However, R2 is “not an FDA-approved regimen in the frontline because it did not demonstrate superiority” over other treatments, Hsu said.
Selecting the Right Therapy
Which therapy is best? It’s a bit of a wash, Hsu said.
He noted that R2, R-Benda, and another therapy that’s not yet in the VA pathway (R-CHOP) appear to be noninferior to one another, although R-Benda has the edge. R2 is better regarding neutropenia risk, although it lacks FDA approval.
“I think about these 3 regimens as appropriate and good,” Hsu said. “It’s nice having 3 wonderful regimens.”
Hsu highlighted the importance of complete remission (CR) as a goal. He pointed to a 2022 analysis of > 5,200 patients that showed progression within 24 months greatly boosted the risk of death vs no-progression (HR, 3.03). Progression within 24 months also lowered estimated 5-year overall survival to 71%.
“Timing really does matter,” Hsu said. “We often worry about transformation to diffuse large B-cell in patients who relapse, particularly this early.”
Second-Line Therapy Options
Two regimens have recently achieved National Comprehensive Cancer Network Category 1 preferred status in the second-line setting, Hsu said, although neither appears in the VA pathway.
One is tafasitamab plus R2, which was shown to extend median PFS to 22.4 months vs 13.9 months for R2 alone in the 2026 inMIND study (HR, 0.43), but without an overall survival benefit.
The other therapy is epcoritamab plus R2: Data from the 2026 EPCORE FL-1 study showed an overall response rate (ORR) of 95% for the combination vs 79% for R2 alone and an estimated 16-month PFS of 85.5% for the combination vs 40.2% for R2.
Hsu cautioned about the adverse event profile for community infusion centers. The combination carried higher rates of grade ≥ 3 infections (33% vs 16%) and neutropenia (69% vs 42%) compared with R2 alone. However, grade ≥ 3 cytokine release syndrome was absent.
“Stay alert to higher risk for infections and neutropenia here,” Hsu said.
Beyond Second Line: Biospecifics and CAR-T
The biospecifics mosunetuzumab and epcoritamab are now FDA-approved for patients who have relapsed ≥ 2 times. Mosunetuzumab showed ORR of 78% and CR rate of 60% in a 2025 study, while epcoritamab monotherapy showed ORR of 82% and CR of 63% in a 2024 study.
Mosunetuzumab had a 2.2% rate of cytokine release syndrome and a 4.4% rate of immune effector cell-associated neurotoxicity syndrome; epcoritamab had 0% rates of both.
“Think of these 2 options as getting you to the same place, potentially, but maybe with slightly different rates of toxicity,” Hsu said.
Meanwhile, CAR-T therapy has shown “impressive results for the right patient,” Hsu said.
Tazemetostat Withdrawn
Hsu noted that tazemetostat, an EZH2 inhibitor that was FDA-approved for relapsed/refractory FL with EZH2 mutations and patients with FL and no satisfactory alternative options, was withdrawn from the market by Eisai in March 2026. The cause of withdrawal was increased rates of secondary hematologic malignancies.
Meanwhile, patients enrolled in the ongoing SYMPHONY-1 trial will be switched to R2.
The withdrawal was “unfortunate,” Hsu said, “but the concept is important. Identifying new targets for therapy and developing those is how we make progress.”
SAN FRANCISCO – Treatment for follicular lymphoma (FL) continues to evolve, according to a US Department of Veterans Affairs (VA) hematologist-oncologist, as second-line regimens emerge but the withdrawal of a recently approved agent complicates the picture.
“The future for our understanding and treatment of follicular lymphoma remains bright,” said Gerald Hsu, MD, PhD, of the University of California at San Francisco and the San Francisco VA Health Care System, during a presentation at the March Association of VA Hematology/Oncology (AVAHO) regional meeting on lymphoma.
By the Numbers
About 16,500 people in the US are diagnosed with FL each year. The median age of diagnosis is 64 years, and the 5-year survival rate from 2015-2021 was 89.0%, according to the National Cancer Institute.
FL is slow-growing and indolent, Hsu said.
“[That] means that we tend to see patients who are older when they are diagnosed,” he added. “They tend to live a long time, and they’re not usually curable.”
A better understanding of the biology of FL has allowed for the development of new markers and ways of measuring residual disease, Hsu said. Additional insight may allow clinicians to identify which patients could benefit most from specific therapies.
Frontline Options
Hsu highlighted the VA Oncology Clinical Pathway for FL, which offers step-by-step guidance regarding therapy and was updated in March 2026. “It walks you through the pathway, but it’s not something that you are beholden to,” he said.
If the patient has classic FL grades 1-3A, is not at risk of transformation to aggressive lymphoma, is not in stage 1 or continuous stage lymphoma, and is indicated for therapy, the guideline recommends lenalidomide plus rituximab (R2 or R-Len) or rituximab-bendamustine (R-Benda).
“There’s a lot of data to support R-Benda,” Hsu said, pointing to a pair of studies with large numbers of patients with FL. The 2013 StiL trial tracked > 500 patients with indolent or mantle cell lymphoma (46% high risk). Those on R-Benda displayed better progression-free survival (PFS) than those taking the combination rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP; hazard ratio [HR], 0.61).
In a 5-year update of the BRIGHT trial, which enrolled > 500 patients with indolent or mantle cell lymphoma (9% high risk), the R-Benda group had better PFS than patients on R-CHOP or rituximab plus cyclophosphamide, vincristine, and prednisone (HR, 0.61).
R2 is a somewhat newer regimen, Hsu said. Data from the 2018 RELEVANCE study (> 1000 patients) found R2 to be nonsuperior to 3 rituximab-plus-chemotherapy regimens but with a lower rate of grade 3-4 neutropenia (32% vs 50% for the other regimens).
However, R2 is “not an FDA-approved regimen in the frontline because it did not demonstrate superiority” over other treatments, Hsu said.
Selecting the Right Therapy
Which therapy is best? It’s a bit of a wash, Hsu said.
He noted that R2, R-Benda, and another therapy that’s not yet in the VA pathway (R-CHOP) appear to be noninferior to one another, although R-Benda has the edge. R2 is better regarding neutropenia risk, although it lacks FDA approval.
“I think about these 3 regimens as appropriate and good,” Hsu said. “It’s nice having 3 wonderful regimens.”
Hsu highlighted the importance of complete remission (CR) as a goal. He pointed to a 2022 analysis of > 5,200 patients that showed progression within 24 months greatly boosted the risk of death vs no-progression (HR, 3.03). Progression within 24 months also lowered estimated 5-year overall survival to 71%.
“Timing really does matter,” Hsu said. “We often worry about transformation to diffuse large B-cell in patients who relapse, particularly this early.”
Second-Line Therapy Options
Two regimens have recently achieved National Comprehensive Cancer Network Category 1 preferred status in the second-line setting, Hsu said, although neither appears in the VA pathway.
One is tafasitamab plus R2, which was shown to extend median PFS to 22.4 months vs 13.9 months for R2 alone in the 2026 inMIND study (HR, 0.43), but without an overall survival benefit.
The other therapy is epcoritamab plus R2: Data from the 2026 EPCORE FL-1 study showed an overall response rate (ORR) of 95% for the combination vs 79% for R2 alone and an estimated 16-month PFS of 85.5% for the combination vs 40.2% for R2.
Hsu cautioned about the adverse event profile for community infusion centers. The combination carried higher rates of grade ≥ 3 infections (33% vs 16%) and neutropenia (69% vs 42%) compared with R2 alone. However, grade ≥ 3 cytokine release syndrome was absent.
“Stay alert to higher risk for infections and neutropenia here,” Hsu said.
Beyond Second Line: Biospecifics and CAR-T
The biospecifics mosunetuzumab and epcoritamab are now FDA-approved for patients who have relapsed ≥ 2 times. Mosunetuzumab showed ORR of 78% and CR rate of 60% in a 2025 study, while epcoritamab monotherapy showed ORR of 82% and CR of 63% in a 2024 study.
Mosunetuzumab had a 2.2% rate of cytokine release syndrome and a 4.4% rate of immune effector cell-associated neurotoxicity syndrome; epcoritamab had 0% rates of both.
“Think of these 2 options as getting you to the same place, potentially, but maybe with slightly different rates of toxicity,” Hsu said.
Meanwhile, CAR-T therapy has shown “impressive results for the right patient,” Hsu said.
Tazemetostat Withdrawn
Hsu noted that tazemetostat, an EZH2 inhibitor that was FDA-approved for relapsed/refractory FL with EZH2 mutations and patients with FL and no satisfactory alternative options, was withdrawn from the market by Eisai in March 2026. The cause of withdrawal was increased rates of secondary hematologic malignancies.
Meanwhile, patients enrolled in the ongoing SYMPHONY-1 trial will be switched to R2.
The withdrawal was “unfortunate,” Hsu said, “but the concept is important. Identifying new targets for therapy and developing those is how we make progress.”
SAN FRANCISCO – Treatment for follicular lymphoma (FL) continues to evolve, according to a US Department of Veterans Affairs (VA) hematologist-oncologist, as second-line regimens emerge but the withdrawal of a recently approved agent complicates the picture.
“The future for our understanding and treatment of follicular lymphoma remains bright,” said Gerald Hsu, MD, PhD, of the University of California at San Francisco and the San Francisco VA Health Care System, during a presentation at the March Association of VA Hematology/Oncology (AVAHO) regional meeting on lymphoma.
By the Numbers
About 16,500 people in the US are diagnosed with FL each year. The median age of diagnosis is 64 years, and the 5-year survival rate from 2015-2021 was 89.0%, according to the National Cancer Institute.
FL is slow-growing and indolent, Hsu said.
“[That] means that we tend to see patients who are older when they are diagnosed,” he added. “They tend to live a long time, and they’re not usually curable.”
A better understanding of the biology of FL has allowed for the development of new markers and ways of measuring residual disease, Hsu said. Additional insight may allow clinicians to identify which patients could benefit most from specific therapies.
Frontline Options
Hsu highlighted the VA Oncology Clinical Pathway for FL, which offers step-by-step guidance regarding therapy and was updated in March 2026. “It walks you through the pathway, but it’s not something that you are beholden to,” he said.
If the patient has classic FL grades 1-3A, is not at risk of transformation to aggressive lymphoma, is not in stage 1 or continuous stage lymphoma, and is indicated for therapy, the guideline recommends lenalidomide plus rituximab (R2 or R-Len) or rituximab-bendamustine (R-Benda).
“There’s a lot of data to support R-Benda,” Hsu said, pointing to a pair of studies with large numbers of patients with FL. The 2013 StiL trial tracked > 500 patients with indolent or mantle cell lymphoma (46% high risk). Those on R-Benda displayed better progression-free survival (PFS) than those taking the combination rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP; hazard ratio [HR], 0.61).
In a 5-year update of the BRIGHT trial, which enrolled > 500 patients with indolent or mantle cell lymphoma (9% high risk), the R-Benda group had better PFS than patients on R-CHOP or rituximab plus cyclophosphamide, vincristine, and prednisone (HR, 0.61).
R2 is a somewhat newer regimen, Hsu said. Data from the 2018 RELEVANCE study (> 1000 patients) found R2 to be nonsuperior to 3 rituximab-plus-chemotherapy regimens but with a lower rate of grade 3-4 neutropenia (32% vs 50% for the other regimens).
However, R2 is “not an FDA-approved regimen in the frontline because it did not demonstrate superiority” over other treatments, Hsu said.
Selecting the Right Therapy
Which therapy is best? It’s a bit of a wash, Hsu said.
He noted that R2, R-Benda, and another therapy that’s not yet in the VA pathway (R-CHOP) appear to be noninferior to one another, although R-Benda has the edge. R2 is better regarding neutropenia risk, although it lacks FDA approval.
“I think about these 3 regimens as appropriate and good,” Hsu said. “It’s nice having 3 wonderful regimens.”
Hsu highlighted the importance of complete remission (CR) as a goal. He pointed to a 2022 analysis of > 5,200 patients that showed progression within 24 months greatly boosted the risk of death vs no-progression (HR, 3.03). Progression within 24 months also lowered estimated 5-year overall survival to 71%.
“Timing really does matter,” Hsu said. “We often worry about transformation to diffuse large B-cell in patients who relapse, particularly this early.”
Second-Line Therapy Options
Two regimens have recently achieved National Comprehensive Cancer Network Category 1 preferred status in the second-line setting, Hsu said, although neither appears in the VA pathway.
One is tafasitamab plus R2, which was shown to extend median PFS to 22.4 months vs 13.9 months for R2 alone in the 2026 inMIND study (HR, 0.43), but without an overall survival benefit.
The other therapy is epcoritamab plus R2: Data from the 2026 EPCORE FL-1 study showed an overall response rate (ORR) of 95% for the combination vs 79% for R2 alone and an estimated 16-month PFS of 85.5% for the combination vs 40.2% for R2.
Hsu cautioned about the adverse event profile for community infusion centers. The combination carried higher rates of grade ≥ 3 infections (33% vs 16%) and neutropenia (69% vs 42%) compared with R2 alone. However, grade ≥ 3 cytokine release syndrome was absent.
“Stay alert to higher risk for infections and neutropenia here,” Hsu said.
Beyond Second Line: Biospecifics and CAR-T
The biospecifics mosunetuzumab and epcoritamab are now FDA-approved for patients who have relapsed ≥ 2 times. Mosunetuzumab showed ORR of 78% and CR rate of 60% in a 2025 study, while epcoritamab monotherapy showed ORR of 82% and CR of 63% in a 2024 study.
Mosunetuzumab had a 2.2% rate of cytokine release syndrome and a 4.4% rate of immune effector cell-associated neurotoxicity syndrome; epcoritamab had 0% rates of both.
“Think of these 2 options as getting you to the same place, potentially, but maybe with slightly different rates of toxicity,” Hsu said.
Meanwhile, CAR-T therapy has shown “impressive results for the right patient,” Hsu said.
Tazemetostat Withdrawn
Hsu noted that tazemetostat, an EZH2 inhibitor that was FDA-approved for relapsed/refractory FL with EZH2 mutations and patients with FL and no satisfactory alternative options, was withdrawn from the market by Eisai in March 2026. The cause of withdrawal was increased rates of secondary hematologic malignancies.
Meanwhile, patients enrolled in the ongoing SYMPHONY-1 trial will be switched to R2.
The withdrawal was “unfortunate,” Hsu said, “but the concept is important. Identifying new targets for therapy and developing those is how we make progress.”
Evolving and Future Treatments for Follicular Lymphoma
Evolving and Future Treatments for Follicular Lymphoma
Male Vets Less Likely to Undergo Intimate Partner Violence Screening
Male veterans are less likely than their female counterparts to be referred for follow-up questions when initial screening suggests they may be at risk of intimate partner violence (IPV), a recent large cross-sectional study finds.
Among 67,379 patients from 131 US Department of Veterans Affairs (VA) medical centers who screened positive for risk of IPV from October 2022 through September 2023, 17.7% failed to receive a mandated secondary screen to determine whether they were in danger of lethal violence, reported Galina A. Portnoy, PhD, of VA Connecticut Healthcare System and Yale School of Medicine, et al in JAMA Network Open. The rate was higher for men with initial positive screens than women (19.3% vs 12.1%, respectively, adjusted odds ratio [AOR], 1.42, P < .001).
Overall, women who underwent secondary screening were more likely to be considered in lethal danger from IPV than men (27.9% vs 13.3%, respectively, AOR 2.29, P < .001).
“While women face higher lethality risk, men’s IPV experiences are often overlooked, underscoring the need for consistent and reliable screening practices to identify all high-risk patients and connect them to life-saving services,” Portnoy told Federal Practitioner.
“IPV is one of the strongest predictors of homicide with risk escalating over time and especially high during periods of separation.”
“IPV among men is often underreported, unrecognized, and inadequately addressed in clinical settings,” Portnoy noted. “Men who experience IPV often face barriers to reporting—stigma, shame, and concerns about not being taken seriously.”
The VA has implemented annual screening of IPV in women of reproductive age using a modified version of the 5-question Hurt, Insult, Threaten, Scream (HITS) tool. HITS asks how often a woman’s partner had screamed, cursed, insulted, or talked down to them; threatened to harm or physically hurt them, or forced or pressured them to “have sexual contact against your will, or when you were unable to say no” in the last year.
If a patient answers yes to any of these questions, clinicians should follow up with a secondary lethality screen with 3 questions:
Has the IPV behavior increased in frequency/severity in the past 6 months?
Has your partner ever choked or strangled you? and
Do you believe your partner may kill you?
The test is considered positive if a patient answers yes to any question.
The study focused on 67,379 patients out of 1,265,115 at the VA who scored positive on HITS (mean age, 52.3 years; 23% women; 62.9% White; 8.2% Hispanic/Latino). More than two-thirds (69.0%) had a service-connected disability rating > 50%.
Portnoy said there are several possible reasons for the gender disparity in misclassification such as time constraints, discomfort, limited resources, and lack of training. Clinician bias can be a factor, too, “with IPV still widely seen as primarily a women’s issue.”
“We don’t know whether IPV screening tools work the same for men as they do for women,” Portnoy added. “The HITS tool was developed and validated using samples of women who experienced IPV, and research is needed to test whether it performs as effectively in men.”
Bethany L. Backes, PhD, associate professor and lead, Violence Against Women Faculty Cluster, University of Central Florida, Orlando, is familiar with the study findings and said in an interview that discomfort among clinicians is a significant factor in preventing follow-up IPV screening.
“When you’re asking about this and someone says ‘yes,’ how do you respond? You just go to the next thing, the next question: ‘How many drinks have you had in the last week?’” Backes told Federal Practitioner. “We’ve talked about creating some scripts for our student health clinicians on campus about how to talk to someone when they disclose, how to then engage or provide resources.”
This is especially important because “it’s hard for people to admit that they’re experiencing this, and then when they do and it’s brushed over, they’re less likely to tell someone again,” Backes added.
C. Nadine Wathen, PhD, a professor who studies IPV at Western University in London, is also familiar with the study findings, but critiqued the HITS, calling it a “terrible name.” The tool, she said, asks about very different behaviors–being screamed or cursed, for example, and forced sexual contact,” she explained to Federal Practitioner.
“If you’re a physician and you’re asking a man, ‘Does she scream or curse at you?’ and he says ‘Yeah, she screams all the time,’ a provider might say, ‘I’m not actually thinking that he’s experiencing intimate partner violence,” Wathen said. “He might be experiencing a bad relationship.’”
That could be true, Wathen said. Couples may scream and throw things at each other, and “you probably could benefit with some couples counseling on how to have a better relationship and manage stress and anger in your relationship. But that is different than ‘intimate partner terrorism,’ where there‘s a pattern of control.”
Wathen prefers a screening tool she helped develop called the Composite Abuse Scale, which she considers more sensitive and specific than HITS. It differentiates the types of abuse that people experience, and “it also recognizes that men in relationships with other men can experience those forms of intimate terrorism, and women can also be the perpetrator of those forms.”
Recognizing that VA clinicians may not have a choice of screening tool, Wathen suggested they follow up the question about screaming and cursing question this query: “Does that make you afraid?”
The study was funded by US Department of Veterans Affairs Quality Enhancement Research Initiative and the Veterans Health Administration’s Care Management and Social Work Service via the Intimate Partner Violence Center for Implementation, Research, and Evaluation.
Portnoy has no disclosures. One author discloses relationships with the National Council on Family Relations and Military Family Research Institute. Backes and Wathen have no disclosures.
Male veterans are less likely than their female counterparts to be referred for follow-up questions when initial screening suggests they may be at risk of intimate partner violence (IPV), a recent large cross-sectional study finds.
Among 67,379 patients from 131 US Department of Veterans Affairs (VA) medical centers who screened positive for risk of IPV from October 2022 through September 2023, 17.7% failed to receive a mandated secondary screen to determine whether they were in danger of lethal violence, reported Galina A. Portnoy, PhD, of VA Connecticut Healthcare System and Yale School of Medicine, et al in JAMA Network Open. The rate was higher for men with initial positive screens than women (19.3% vs 12.1%, respectively, adjusted odds ratio [AOR], 1.42, P < .001).
Overall, women who underwent secondary screening were more likely to be considered in lethal danger from IPV than men (27.9% vs 13.3%, respectively, AOR 2.29, P < .001).
“While women face higher lethality risk, men’s IPV experiences are often overlooked, underscoring the need for consistent and reliable screening practices to identify all high-risk patients and connect them to life-saving services,” Portnoy told Federal Practitioner.
“IPV is one of the strongest predictors of homicide with risk escalating over time and especially high during periods of separation.”
“IPV among men is often underreported, unrecognized, and inadequately addressed in clinical settings,” Portnoy noted. “Men who experience IPV often face barriers to reporting—stigma, shame, and concerns about not being taken seriously.”
The VA has implemented annual screening of IPV in women of reproductive age using a modified version of the 5-question Hurt, Insult, Threaten, Scream (HITS) tool. HITS asks how often a woman’s partner had screamed, cursed, insulted, or talked down to them; threatened to harm or physically hurt them, or forced or pressured them to “have sexual contact against your will, or when you were unable to say no” in the last year.
If a patient answers yes to any of these questions, clinicians should follow up with a secondary lethality screen with 3 questions:
Has the IPV behavior increased in frequency/severity in the past 6 months?
Has your partner ever choked or strangled you? and
Do you believe your partner may kill you?
The test is considered positive if a patient answers yes to any question.
The study focused on 67,379 patients out of 1,265,115 at the VA who scored positive on HITS (mean age, 52.3 years; 23% women; 62.9% White; 8.2% Hispanic/Latino). More than two-thirds (69.0%) had a service-connected disability rating > 50%.
Portnoy said there are several possible reasons for the gender disparity in misclassification such as time constraints, discomfort, limited resources, and lack of training. Clinician bias can be a factor, too, “with IPV still widely seen as primarily a women’s issue.”
“We don’t know whether IPV screening tools work the same for men as they do for women,” Portnoy added. “The HITS tool was developed and validated using samples of women who experienced IPV, and research is needed to test whether it performs as effectively in men.”
Bethany L. Backes, PhD, associate professor and lead, Violence Against Women Faculty Cluster, University of Central Florida, Orlando, is familiar with the study findings and said in an interview that discomfort among clinicians is a significant factor in preventing follow-up IPV screening.
“When you’re asking about this and someone says ‘yes,’ how do you respond? You just go to the next thing, the next question: ‘How many drinks have you had in the last week?’” Backes told Federal Practitioner. “We’ve talked about creating some scripts for our student health clinicians on campus about how to talk to someone when they disclose, how to then engage or provide resources.”
This is especially important because “it’s hard for people to admit that they’re experiencing this, and then when they do and it’s brushed over, they’re less likely to tell someone again,” Backes added.
C. Nadine Wathen, PhD, a professor who studies IPV at Western University in London, is also familiar with the study findings, but critiqued the HITS, calling it a “terrible name.” The tool, she said, asks about very different behaviors–being screamed or cursed, for example, and forced sexual contact,” she explained to Federal Practitioner.
“If you’re a physician and you’re asking a man, ‘Does she scream or curse at you?’ and he says ‘Yeah, she screams all the time,’ a provider might say, ‘I’m not actually thinking that he’s experiencing intimate partner violence,” Wathen said. “He might be experiencing a bad relationship.’”
That could be true, Wathen said. Couples may scream and throw things at each other, and “you probably could benefit with some couples counseling on how to have a better relationship and manage stress and anger in your relationship. But that is different than ‘intimate partner terrorism,’ where there‘s a pattern of control.”
Wathen prefers a screening tool she helped develop called the Composite Abuse Scale, which she considers more sensitive and specific than HITS. It differentiates the types of abuse that people experience, and “it also recognizes that men in relationships with other men can experience those forms of intimate terrorism, and women can also be the perpetrator of those forms.”
Recognizing that VA clinicians may not have a choice of screening tool, Wathen suggested they follow up the question about screaming and cursing question this query: “Does that make you afraid?”
The study was funded by US Department of Veterans Affairs Quality Enhancement Research Initiative and the Veterans Health Administration’s Care Management and Social Work Service via the Intimate Partner Violence Center for Implementation, Research, and Evaluation.
Portnoy has no disclosures. One author discloses relationships with the National Council on Family Relations and Military Family Research Institute. Backes and Wathen have no disclosures.
Male veterans are less likely than their female counterparts to be referred for follow-up questions when initial screening suggests they may be at risk of intimate partner violence (IPV), a recent large cross-sectional study finds.
Among 67,379 patients from 131 US Department of Veterans Affairs (VA) medical centers who screened positive for risk of IPV from October 2022 through September 2023, 17.7% failed to receive a mandated secondary screen to determine whether they were in danger of lethal violence, reported Galina A. Portnoy, PhD, of VA Connecticut Healthcare System and Yale School of Medicine, et al in JAMA Network Open. The rate was higher for men with initial positive screens than women (19.3% vs 12.1%, respectively, adjusted odds ratio [AOR], 1.42, P < .001).
Overall, women who underwent secondary screening were more likely to be considered in lethal danger from IPV than men (27.9% vs 13.3%, respectively, AOR 2.29, P < .001).
“While women face higher lethality risk, men’s IPV experiences are often overlooked, underscoring the need for consistent and reliable screening practices to identify all high-risk patients and connect them to life-saving services,” Portnoy told Federal Practitioner.
“IPV is one of the strongest predictors of homicide with risk escalating over time and especially high during periods of separation.”
“IPV among men is often underreported, unrecognized, and inadequately addressed in clinical settings,” Portnoy noted. “Men who experience IPV often face barriers to reporting—stigma, shame, and concerns about not being taken seriously.”
The VA has implemented annual screening of IPV in women of reproductive age using a modified version of the 5-question Hurt, Insult, Threaten, Scream (HITS) tool. HITS asks how often a woman’s partner had screamed, cursed, insulted, or talked down to them; threatened to harm or physically hurt them, or forced or pressured them to “have sexual contact against your will, or when you were unable to say no” in the last year.
If a patient answers yes to any of these questions, clinicians should follow up with a secondary lethality screen with 3 questions:
Has the IPV behavior increased in frequency/severity in the past 6 months?
Has your partner ever choked or strangled you? and
Do you believe your partner may kill you?
The test is considered positive if a patient answers yes to any question.
The study focused on 67,379 patients out of 1,265,115 at the VA who scored positive on HITS (mean age, 52.3 years; 23% women; 62.9% White; 8.2% Hispanic/Latino). More than two-thirds (69.0%) had a service-connected disability rating > 50%.
Portnoy said there are several possible reasons for the gender disparity in misclassification such as time constraints, discomfort, limited resources, and lack of training. Clinician bias can be a factor, too, “with IPV still widely seen as primarily a women’s issue.”
“We don’t know whether IPV screening tools work the same for men as they do for women,” Portnoy added. “The HITS tool was developed and validated using samples of women who experienced IPV, and research is needed to test whether it performs as effectively in men.”
Bethany L. Backes, PhD, associate professor and lead, Violence Against Women Faculty Cluster, University of Central Florida, Orlando, is familiar with the study findings and said in an interview that discomfort among clinicians is a significant factor in preventing follow-up IPV screening.
“When you’re asking about this and someone says ‘yes,’ how do you respond? You just go to the next thing, the next question: ‘How many drinks have you had in the last week?’” Backes told Federal Practitioner. “We’ve talked about creating some scripts for our student health clinicians on campus about how to talk to someone when they disclose, how to then engage or provide resources.”
This is especially important because “it’s hard for people to admit that they’re experiencing this, and then when they do and it’s brushed over, they’re less likely to tell someone again,” Backes added.
C. Nadine Wathen, PhD, a professor who studies IPV at Western University in London, is also familiar with the study findings, but critiqued the HITS, calling it a “terrible name.” The tool, she said, asks about very different behaviors–being screamed or cursed, for example, and forced sexual contact,” she explained to Federal Practitioner.
“If you’re a physician and you’re asking a man, ‘Does she scream or curse at you?’ and he says ‘Yeah, she screams all the time,’ a provider might say, ‘I’m not actually thinking that he’s experiencing intimate partner violence,” Wathen said. “He might be experiencing a bad relationship.’”
That could be true, Wathen said. Couples may scream and throw things at each other, and “you probably could benefit with some couples counseling on how to have a better relationship and manage stress and anger in your relationship. But that is different than ‘intimate partner terrorism,’ where there‘s a pattern of control.”
Wathen prefers a screening tool she helped develop called the Composite Abuse Scale, which she considers more sensitive and specific than HITS. It differentiates the types of abuse that people experience, and “it also recognizes that men in relationships with other men can experience those forms of intimate terrorism, and women can also be the perpetrator of those forms.”
Recognizing that VA clinicians may not have a choice of screening tool, Wathen suggested they follow up the question about screaming and cursing question this query: “Does that make you afraid?”
The study was funded by US Department of Veterans Affairs Quality Enhancement Research Initiative and the Veterans Health Administration’s Care Management and Social Work Service via the Intimate Partner Violence Center for Implementation, Research, and Evaluation.
Portnoy has no disclosures. One author discloses relationships with the National Council on Family Relations and Military Family Research Institute. Backes and Wathen have no disclosures.