User login
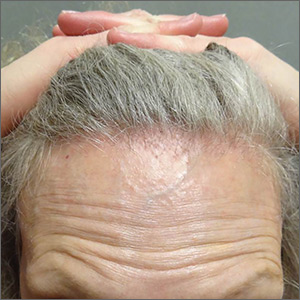
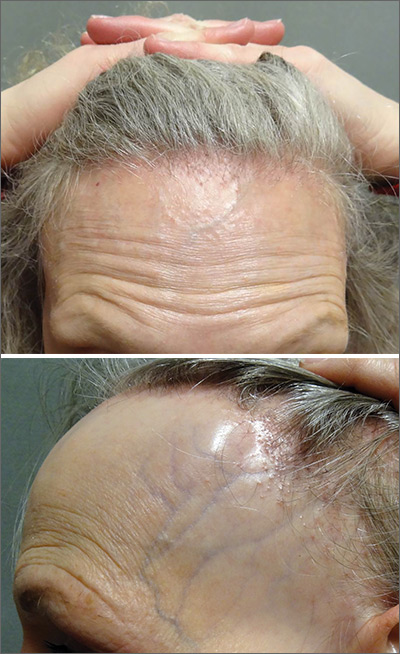
Symmetric hair loss across frontal and temporal scalp
The FP referred the patient to a dermatology clinic that specialized in hair loss. Based on the clinical findings, physicians at the clinic suspected that this was a case of frontal fibrosing alopecia (FFA), a primary lymphocytic cicatricial (scarring) alopecia. A dermatopathologist confirmed the diagnosis via histologic review.
FFA is characterized by symmetric band-like hair loss with evidence of scarring in the frontal and temporal regions of the scalp. (The extent of hair loss can be assessed by retracting the patient’s hair and having the patient raise his or her eyebrows and wrinkle the forehead in a surprised look.) FFA is accompanied by eyebrow loss in 73% to 95% of patients. Mild to severe perifollicular (and possibly more generalized) erythema and scale are usually present. In addition, erythematous or skin-colored papules may appear on the face, and marked exaggeration of the temporal veins is a common finding.
Most patients with FFA (83%) are postmenopausal women and nearly all (98.6%) have Fitzpatrick skin type 1 or 2 (white skin that burns easily and doesn’t readily tan). Other pertinent findings include the absence of oral lesions, nail changes, or other skin diseases.
A punch biopsy taken from the leading edge of the hair loss confirms the diagnosis of FFA and, ideally, should be reviewed by a dermatopathologist. Histologic examination will reveal a lichenoid lymphocytic infiltrate (predominantly around the hair follicle where the follicular stem cells reside), resulting in fibrosis and scarring. In addition to confirming the diagnosis with histologic examination, the following conditions must be ruled out in the differential: alopecia areata, female pattern hair loss, discoid lupus erythematosus, central centrifugal cicatricial alopecia, and traction alopecia.
Early detection is key. In general, physicians should initiate treatment as soon as possible to prevent disease progression and reduce permanent scarring and hair loss. Intralesional steroids such as triamcinolone acetonide (5-10 mg/cc), as well as high-potency topical steroids, are generally helpful to stabilize the disease. There is also some evidence of benefit from oral dutasteride or finasteride at variable doses. Immunosuppressants such as hydroxychloroquine also may be used as second-line treatments, although the benefit-to-risk ratio needs to be taken into consideration.
This patient was started on a regimen of topical high-potency steroids (clobetasol foam, 0.05%), with targeted, intralesional injection of steroids (10 mg/cc of triamcinolone acetonide) to areas with the most inflammation. She also was advised to use ketoconazole 2% shampoo while showering. These interventions decreased the patient’s symptoms dramatically. Her scalp erythema and scale improved, but the hair did not regrow. One year later, her hairline was clinically stable with no evidence of disease progression. She continued to see a dermatologist annually.
This case was adapted from: Power DV, Disse M, Hordinsky M. Progressive hair loss. J Fam Pract. 2017;66:521-523.

The FP referred the patient to a dermatology clinic that specialized in hair loss. Based on the clinical findings, physicians at the clinic suspected that this was a case of frontal fibrosing alopecia (FFA), a primary lymphocytic cicatricial (scarring) alopecia. A dermatopathologist confirmed the diagnosis via histologic review.
FFA is characterized by symmetric band-like hair loss with evidence of scarring in the frontal and temporal regions of the scalp. (The extent of hair loss can be assessed by retracting the patient’s hair and having the patient raise his or her eyebrows and wrinkle the forehead in a surprised look.) FFA is accompanied by eyebrow loss in 73% to 95% of patients. Mild to severe perifollicular (and possibly more generalized) erythema and scale are usually present. In addition, erythematous or skin-colored papules may appear on the face, and marked exaggeration of the temporal veins is a common finding.
Most patients with FFA (83%) are postmenopausal women and nearly all (98.6%) have Fitzpatrick skin type 1 or 2 (white skin that burns easily and doesn’t readily tan). Other pertinent findings include the absence of oral lesions, nail changes, or other skin diseases.
A punch biopsy taken from the leading edge of the hair loss confirms the diagnosis of FFA and, ideally, should be reviewed by a dermatopathologist. Histologic examination will reveal a lichenoid lymphocytic infiltrate (predominantly around the hair follicle where the follicular stem cells reside), resulting in fibrosis and scarring. In addition to confirming the diagnosis with histologic examination, the following conditions must be ruled out in the differential: alopecia areata, female pattern hair loss, discoid lupus erythematosus, central centrifugal cicatricial alopecia, and traction alopecia.
Early detection is key. In general, physicians should initiate treatment as soon as possible to prevent disease progression and reduce permanent scarring and hair loss. Intralesional steroids such as triamcinolone acetonide (5-10 mg/cc), as well as high-potency topical steroids, are generally helpful to stabilize the disease. There is also some evidence of benefit from oral dutasteride or finasteride at variable doses. Immunosuppressants such as hydroxychloroquine also may be used as second-line treatments, although the benefit-to-risk ratio needs to be taken into consideration.
This patient was started on a regimen of topical high-potency steroids (clobetasol foam, 0.05%), with targeted, intralesional injection of steroids (10 mg/cc of triamcinolone acetonide) to areas with the most inflammation. She also was advised to use ketoconazole 2% shampoo while showering. These interventions decreased the patient’s symptoms dramatically. Her scalp erythema and scale improved, but the hair did not regrow. One year later, her hairline was clinically stable with no evidence of disease progression. She continued to see a dermatologist annually.
This case was adapted from: Power DV, Disse M, Hordinsky M. Progressive hair loss. J Fam Pract. 2017;66:521-523.

The FP referred the patient to a dermatology clinic that specialized in hair loss. Based on the clinical findings, physicians at the clinic suspected that this was a case of frontal fibrosing alopecia (FFA), a primary lymphocytic cicatricial (scarring) alopecia. A dermatopathologist confirmed the diagnosis via histologic review.
FFA is characterized by symmetric band-like hair loss with evidence of scarring in the frontal and temporal regions of the scalp. (The extent of hair loss can be assessed by retracting the patient’s hair and having the patient raise his or her eyebrows and wrinkle the forehead in a surprised look.) FFA is accompanied by eyebrow loss in 73% to 95% of patients. Mild to severe perifollicular (and possibly more generalized) erythema and scale are usually present. In addition, erythematous or skin-colored papules may appear on the face, and marked exaggeration of the temporal veins is a common finding.
Most patients with FFA (83%) are postmenopausal women and nearly all (98.6%) have Fitzpatrick skin type 1 or 2 (white skin that burns easily and doesn’t readily tan). Other pertinent findings include the absence of oral lesions, nail changes, or other skin diseases.
A punch biopsy taken from the leading edge of the hair loss confirms the diagnosis of FFA and, ideally, should be reviewed by a dermatopathologist. Histologic examination will reveal a lichenoid lymphocytic infiltrate (predominantly around the hair follicle where the follicular stem cells reside), resulting in fibrosis and scarring. In addition to confirming the diagnosis with histologic examination, the following conditions must be ruled out in the differential: alopecia areata, female pattern hair loss, discoid lupus erythematosus, central centrifugal cicatricial alopecia, and traction alopecia.
Early detection is key. In general, physicians should initiate treatment as soon as possible to prevent disease progression and reduce permanent scarring and hair loss. Intralesional steroids such as triamcinolone acetonide (5-10 mg/cc), as well as high-potency topical steroids, are generally helpful to stabilize the disease. There is also some evidence of benefit from oral dutasteride or finasteride at variable doses. Immunosuppressants such as hydroxychloroquine also may be used as second-line treatments, although the benefit-to-risk ratio needs to be taken into consideration.
This patient was started on a regimen of topical high-potency steroids (clobetasol foam, 0.05%), with targeted, intralesional injection of steroids (10 mg/cc of triamcinolone acetonide) to areas with the most inflammation. She also was advised to use ketoconazole 2% shampoo while showering. These interventions decreased the patient’s symptoms dramatically. Her scalp erythema and scale improved, but the hair did not regrow. One year later, her hairline was clinically stable with no evidence of disease progression. She continued to see a dermatologist annually.
This case was adapted from: Power DV, Disse M, Hordinsky M. Progressive hair loss. J Fam Pract. 2017;66:521-523.
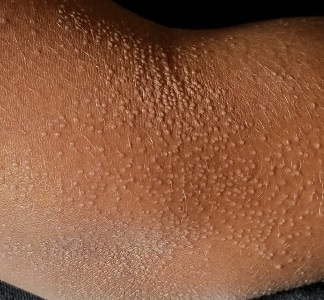
Keeping Lesions at Arm’s Length
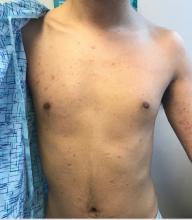
A 14-year-old boy presents to dermatology for evaluation of an asymptomatic “rash” present on his arms since age 6. The condition has caught the attention of family members and teachers over the years, particularly in regard to possible contagion.
The patient is otherwise reasonably healthy, although he has asthma and seasonal allergies.
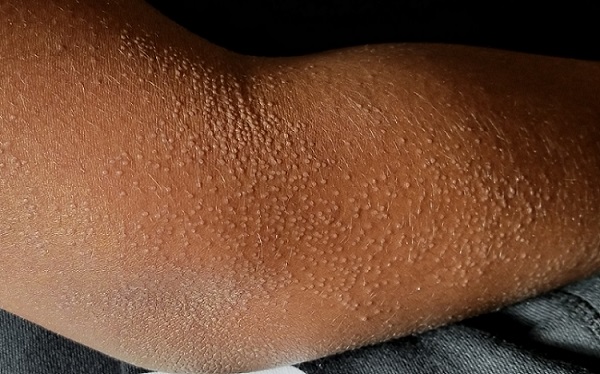
EXAMINATION
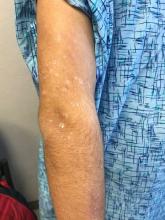
The "rash" consists of uniformly distributed and sized planar papules. Although they are tiny, averaging only 1 mm wide, they are prominent enough to be noticeable and palpable. They appear slightly lighter than the surrounding skin. Distribution is from the lower deltoid to mid-dorsal forearm, affecting both arms identically. The volar aspects and triceps of both arms are totally spared.
The patient has type IV skin.
What’s the diagnosis?
DISCUSSION
This case is an almost perfect representation of lichen nitidus (LN), in terms of morphology, distribution, and configuration. Close examination of individual lesions revealed that the papules were somewhat planar (ie, flat-topped), giving their surfaces a reflective appearance that the eye interprets as white (particularly contrasted with darker skin).
LN can occur in anyone, but it is most often seen in those with darker skin. It is also frequently seen in children, many of whom are atopic, with dry, sensitive skin that is prone to eczema.
In terms of distribution, LN typically affects the extensor triceps, elbow, and forearms bilaterally. With its flat-topped and shiny appearance, LN is sometimes called "mini-lichen planus"—a condition that can demonstrate similar features. Fortunately, LN is seldom itchy and shares none of the distinct histologic characteristics of lichen planus.
LN is quite unusual, if not rare. It is also idiopathic and nearly always resolves on its own—although this can take months to years.
Emollients help to make the affected skin smoother and less visible. Class 4 steroid creams (eg, triamcinolone 0.05%) can help with itching.
TAKE-HOME LEARNING POINTS
- Lichen nitidus (LN) is a rare idiopathic skin condition manifesting with patches of tiny planar papules; it typically affects the elbow and dorsal forearm.
- LN has no pathologic implications and is asymptomatic and self-limited.
- The lesions of LN have a “lichenoid” appearance—ie, a shiny, flat-topped look similar to that seen with lichen planus.
- Fortunately, LN rarely requires treatment, aside from relief of mild itching.
A 14-year-old boy presents to dermatology for evaluation of an asymptomatic “rash” present on his arms since age 6. The condition has caught the attention of family members and teachers over the years, particularly in regard to possible contagion.
The patient is otherwise reasonably healthy, although he has asthma and seasonal allergies.

EXAMINATION
The "rash" consists of uniformly distributed and sized planar papules. Although they are tiny, averaging only 1 mm wide, they are prominent enough to be noticeable and palpable. They appear slightly lighter than the surrounding skin. Distribution is from the lower deltoid to mid-dorsal forearm, affecting both arms identically. The volar aspects and triceps of both arms are totally spared.
The patient has type IV skin.
What’s the diagnosis?
DISCUSSION
This case is an almost perfect representation of lichen nitidus (LN), in terms of morphology, distribution, and configuration. Close examination of individual lesions revealed that the papules were somewhat planar (ie, flat-topped), giving their surfaces a reflective appearance that the eye interprets as white (particularly contrasted with darker skin).
LN can occur in anyone, but it is most often seen in those with darker skin. It is also frequently seen in children, many of whom are atopic, with dry, sensitive skin that is prone to eczema.
In terms of distribution, LN typically affects the extensor triceps, elbow, and forearms bilaterally. With its flat-topped and shiny appearance, LN is sometimes called "mini-lichen planus"—a condition that can demonstrate similar features. Fortunately, LN is seldom itchy and shares none of the distinct histologic characteristics of lichen planus.
LN is quite unusual, if not rare. It is also idiopathic and nearly always resolves on its own—although this can take months to years.
Emollients help to make the affected skin smoother and less visible. Class 4 steroid creams (eg, triamcinolone 0.05%) can help with itching.
TAKE-HOME LEARNING POINTS
- Lichen nitidus (LN) is a rare idiopathic skin condition manifesting with patches of tiny planar papules; it typically affects the elbow and dorsal forearm.
- LN has no pathologic implications and is asymptomatic and self-limited.
- The lesions of LN have a “lichenoid” appearance—ie, a shiny, flat-topped look similar to that seen with lichen planus.
- Fortunately, LN rarely requires treatment, aside from relief of mild itching.
A 14-year-old boy presents to dermatology for evaluation of an asymptomatic “rash” present on his arms since age 6. The condition has caught the attention of family members and teachers over the years, particularly in regard to possible contagion.
The patient is otherwise reasonably healthy, although he has asthma and seasonal allergies.

EXAMINATION
The "rash" consists of uniformly distributed and sized planar papules. Although they are tiny, averaging only 1 mm wide, they are prominent enough to be noticeable and palpable. They appear slightly lighter than the surrounding skin. Distribution is from the lower deltoid to mid-dorsal forearm, affecting both arms identically. The volar aspects and triceps of both arms are totally spared.
The patient has type IV skin.
What’s the diagnosis?
DISCUSSION
This case is an almost perfect representation of lichen nitidus (LN), in terms of morphology, distribution, and configuration. Close examination of individual lesions revealed that the papules were somewhat planar (ie, flat-topped), giving their surfaces a reflective appearance that the eye interprets as white (particularly contrasted with darker skin).
LN can occur in anyone, but it is most often seen in those with darker skin. It is also frequently seen in children, many of whom are atopic, with dry, sensitive skin that is prone to eczema.
In terms of distribution, LN typically affects the extensor triceps, elbow, and forearms bilaterally. With its flat-topped and shiny appearance, LN is sometimes called "mini-lichen planus"—a condition that can demonstrate similar features. Fortunately, LN is seldom itchy and shares none of the distinct histologic characteristics of lichen planus.
LN is quite unusual, if not rare. It is also idiopathic and nearly always resolves on its own—although this can take months to years.
Emollients help to make the affected skin smoother and less visible. Class 4 steroid creams (eg, triamcinolone 0.05%) can help with itching.
TAKE-HOME LEARNING POINTS
- Lichen nitidus (LN) is a rare idiopathic skin condition manifesting with patches of tiny planar papules; it typically affects the elbow and dorsal forearm.
- LN has no pathologic implications and is asymptomatic and self-limited.
- The lesions of LN have a “lichenoid” appearance—ie, a shiny, flat-topped look similar to that seen with lichen planus.
- Fortunately, LN rarely requires treatment, aside from relief of mild itching.
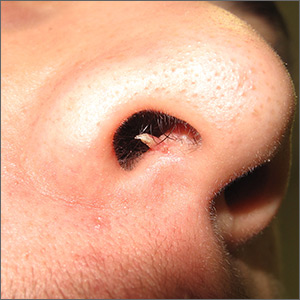
Firm and tender growth in right nostril
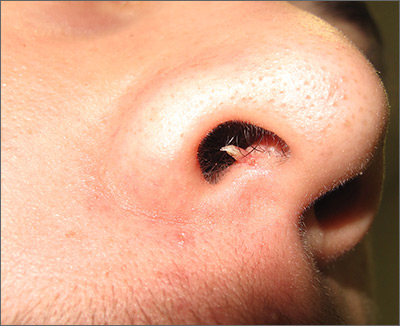
The clinical features were consistent with a filiform wart, which is caused by human papillomavirus and common on the face. Filiform warts may occur on mucosal surfaces, including the nasal mucosa, lips, or eyelids. Most are benign and resolve within 2 years without treatment, but others can be symptomatic. Larger filiform warts may develop the clinical features of a cutaneous horn and mimic a squamous cell carcinoma.
Patients often want the wart removed for functional or cosmetic reasons. Although, there are many treatments available for warts, none have success rates that exceed about 70%. The most common options for treatment include topical salicylic acid in a liquid or plaster, cryotherapy, intralesional immunotherapy with candida antigen, excision, and topical acid.
The location of this patient’s wart limited the treatment options to cryotherapy or snip excision and cautery. The patient opted for cryotherapy. In this process, a hemostat or heavy gauge tweezer is dipped in liquid nitrogen and allowed to cool. Then, without anesthesia, the clinician pinches the lesion gently with the instrument and holds it until the freeze horizon extends to the base. This is then repeated. Each cycle may take 10 and 15 seconds. A benefit of this technique (which is also useful for skin tags and lesions close to the eye) is the ability to avoid overspray of liquid nitrogen and thus, minimize collateral tissue damage. It is also quick and bloodless.
The FP performed cryotherapy on the wart and advised the patient to expect the lesion to become more inflamed over the next 2 to 3 days and then peel off within 1 to 2 weeks. The FP also instructed the patient to return in a week or 2 so that the FP could evaluate whether additional treatment would be necessary. In general, non-genital warts require 2 to 3 individual treatments to clear, but a smaller and pedunculated wart like the one seen in this case tend to clear more easily.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained).

The clinical features were consistent with a filiform wart, which is caused by human papillomavirus and common on the face. Filiform warts may occur on mucosal surfaces, including the nasal mucosa, lips, or eyelids. Most are benign and resolve within 2 years without treatment, but others can be symptomatic. Larger filiform warts may develop the clinical features of a cutaneous horn and mimic a squamous cell carcinoma.
Patients often want the wart removed for functional or cosmetic reasons. Although, there are many treatments available for warts, none have success rates that exceed about 70%. The most common options for treatment include topical salicylic acid in a liquid or plaster, cryotherapy, intralesional immunotherapy with candida antigen, excision, and topical acid.
The location of this patient’s wart limited the treatment options to cryotherapy or snip excision and cautery. The patient opted for cryotherapy. In this process, a hemostat or heavy gauge tweezer is dipped in liquid nitrogen and allowed to cool. Then, without anesthesia, the clinician pinches the lesion gently with the instrument and holds it until the freeze horizon extends to the base. This is then repeated. Each cycle may take 10 and 15 seconds. A benefit of this technique (which is also useful for skin tags and lesions close to the eye) is the ability to avoid overspray of liquid nitrogen and thus, minimize collateral tissue damage. It is also quick and bloodless.
The FP performed cryotherapy on the wart and advised the patient to expect the lesion to become more inflamed over the next 2 to 3 days and then peel off within 1 to 2 weeks. The FP also instructed the patient to return in a week or 2 so that the FP could evaluate whether additional treatment would be necessary. In general, non-genital warts require 2 to 3 individual treatments to clear, but a smaller and pedunculated wart like the one seen in this case tend to clear more easily.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained).

The clinical features were consistent with a filiform wart, which is caused by human papillomavirus and common on the face. Filiform warts may occur on mucosal surfaces, including the nasal mucosa, lips, or eyelids. Most are benign and resolve within 2 years without treatment, but others can be symptomatic. Larger filiform warts may develop the clinical features of a cutaneous horn and mimic a squamous cell carcinoma.
Patients often want the wart removed for functional or cosmetic reasons. Although, there are many treatments available for warts, none have success rates that exceed about 70%. The most common options for treatment include topical salicylic acid in a liquid or plaster, cryotherapy, intralesional immunotherapy with candida antigen, excision, and topical acid.
The location of this patient’s wart limited the treatment options to cryotherapy or snip excision and cautery. The patient opted for cryotherapy. In this process, a hemostat or heavy gauge tweezer is dipped in liquid nitrogen and allowed to cool. Then, without anesthesia, the clinician pinches the lesion gently with the instrument and holds it until the freeze horizon extends to the base. This is then repeated. Each cycle may take 10 and 15 seconds. A benefit of this technique (which is also useful for skin tags and lesions close to the eye) is the ability to avoid overspray of liquid nitrogen and thus, minimize collateral tissue damage. It is also quick and bloodless.
The FP performed cryotherapy on the wart and advised the patient to expect the lesion to become more inflamed over the next 2 to 3 days and then peel off within 1 to 2 weeks. The FP also instructed the patient to return in a week or 2 so that the FP could evaluate whether additional treatment would be necessary. In general, non-genital warts require 2 to 3 individual treatments to clear, but a smaller and pedunculated wart like the one seen in this case tend to clear more easily.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained).
Melanoma incidence drops in younger age groups
, according to results of a population-based registry study of 988,103 cases of invasive melanoma.
These data are observational, “and thus cannot conclusively determine the cause of this statistically and clinically significant decrease,” wrote Kelly G. Paulson, MD, PhD, of the Fred Hutchinson Cancer Research Center, Seattle, and colleagues. However, they added, “a likely explanation for the reduced melanoma incidence in adolescents and young adults is success at increased UV exposure protection. These data provide an impetus to further improve multimodal efforts aimed at reducing the burden of melanoma and encourage ongoing UV exposure protection efforts throughout the lifetime of individuals.”
Public health measures to promote sun-protective behaviors including sunscreen use, protective clothing, and seeking shade were initiated in the United States in the late 1990s and early 2000s, but the public health impact remains unknown, they noted in the study, published in JAMA Dermatology.
For the study, they reviewed data from the National Program of Cancer Registries – Surveillance Epidemiology and End Results combined database for the years 2001-2015. Overall, the incidence of invasive melanoma among people of all ages in the United States increased from 50,272 cases in 2001 to 83,362 in 2015. However, in 2015 only 67 cases were reported in children younger than 10 years, 251 in adolescents aged 10-19 years, and 1,973 in young adults (aged 20-29 years).
Between 2006 and 2015, the annual percentage change in melanoma incidence decreased by 4.4% for male adolescents, 5.4% for female adolescents, 3.7% for male young adults, and 3.6% for female young adults; these changes were statistically significant. The trends in incidence was similar when the population was limited to non-Hispanic whites, considered a high-risk group for melanoma.
By contrast, melanoma incidence increased by an annual percentage change of 1.8% for both men and women aged 40 years and older during the same period of time. Young adult women had a greater incidence of melanoma compared with young adult men (about twofold greater), but older men had a greater incidence of melanoma compared with older women, the researchers said.
The findings were limited by a lack of data about potential confounders, such as skin pigmentation, UV light exposure, sunburn history, sunscreen use, sun avoidance, protective clothing, and tanning bed use; and the absence of information kept the researchers from estimating an association between increased sun-protective behaviors and decreased incidence of melanoma.
“However, this change in behavior remains a plausible explanation for decreased melanoma rates in adolescent and young adult populations,” and the data support continued strategies to promote UV protection throughout life, they said.
The study was supported in part by the National Institutes of Health, the Fred Hutchinson Cancer Research Center Integrated Immunotherapy Research Core, and a Society for Immunotherapy of Cancer–Merck fellowship. Dr. Paulson disclosed grants from the Society for Immunotherapy of Cancer–Merck, bluebird biosciences, EMD Serono; she also disclosed an issued and licensed patent for a Merkel cell carcinoma T cell receptor.
SOURCE: Paulson KG et al. JAMA Dermatol. 2019. Nov 13. doi: 10.1001/jamadermatol.2019.3353.
, according to results of a population-based registry study of 988,103 cases of invasive melanoma.
These data are observational, “and thus cannot conclusively determine the cause of this statistically and clinically significant decrease,” wrote Kelly G. Paulson, MD, PhD, of the Fred Hutchinson Cancer Research Center, Seattle, and colleagues. However, they added, “a likely explanation for the reduced melanoma incidence in adolescents and young adults is success at increased UV exposure protection. These data provide an impetus to further improve multimodal efforts aimed at reducing the burden of melanoma and encourage ongoing UV exposure protection efforts throughout the lifetime of individuals.”
Public health measures to promote sun-protective behaviors including sunscreen use, protective clothing, and seeking shade were initiated in the United States in the late 1990s and early 2000s, but the public health impact remains unknown, they noted in the study, published in JAMA Dermatology.
For the study, they reviewed data from the National Program of Cancer Registries – Surveillance Epidemiology and End Results combined database for the years 2001-2015. Overall, the incidence of invasive melanoma among people of all ages in the United States increased from 50,272 cases in 2001 to 83,362 in 2015. However, in 2015 only 67 cases were reported in children younger than 10 years, 251 in adolescents aged 10-19 years, and 1,973 in young adults (aged 20-29 years).
Between 2006 and 2015, the annual percentage change in melanoma incidence decreased by 4.4% for male adolescents, 5.4% for female adolescents, 3.7% for male young adults, and 3.6% for female young adults; these changes were statistically significant. The trends in incidence was similar when the population was limited to non-Hispanic whites, considered a high-risk group for melanoma.
By contrast, melanoma incidence increased by an annual percentage change of 1.8% for both men and women aged 40 years and older during the same period of time. Young adult women had a greater incidence of melanoma compared with young adult men (about twofold greater), but older men had a greater incidence of melanoma compared with older women, the researchers said.
The findings were limited by a lack of data about potential confounders, such as skin pigmentation, UV light exposure, sunburn history, sunscreen use, sun avoidance, protective clothing, and tanning bed use; and the absence of information kept the researchers from estimating an association between increased sun-protective behaviors and decreased incidence of melanoma.
“However, this change in behavior remains a plausible explanation for decreased melanoma rates in adolescent and young adult populations,” and the data support continued strategies to promote UV protection throughout life, they said.
The study was supported in part by the National Institutes of Health, the Fred Hutchinson Cancer Research Center Integrated Immunotherapy Research Core, and a Society for Immunotherapy of Cancer–Merck fellowship. Dr. Paulson disclosed grants from the Society for Immunotherapy of Cancer–Merck, bluebird biosciences, EMD Serono; she also disclosed an issued and licensed patent for a Merkel cell carcinoma T cell receptor.
SOURCE: Paulson KG et al. JAMA Dermatol. 2019. Nov 13. doi: 10.1001/jamadermatol.2019.3353.
, according to results of a population-based registry study of 988,103 cases of invasive melanoma.
These data are observational, “and thus cannot conclusively determine the cause of this statistically and clinically significant decrease,” wrote Kelly G. Paulson, MD, PhD, of the Fred Hutchinson Cancer Research Center, Seattle, and colleagues. However, they added, “a likely explanation for the reduced melanoma incidence in adolescents and young adults is success at increased UV exposure protection. These data provide an impetus to further improve multimodal efforts aimed at reducing the burden of melanoma and encourage ongoing UV exposure protection efforts throughout the lifetime of individuals.”
Public health measures to promote sun-protective behaviors including sunscreen use, protective clothing, and seeking shade were initiated in the United States in the late 1990s and early 2000s, but the public health impact remains unknown, they noted in the study, published in JAMA Dermatology.
For the study, they reviewed data from the National Program of Cancer Registries – Surveillance Epidemiology and End Results combined database for the years 2001-2015. Overall, the incidence of invasive melanoma among people of all ages in the United States increased from 50,272 cases in 2001 to 83,362 in 2015. However, in 2015 only 67 cases were reported in children younger than 10 years, 251 in adolescents aged 10-19 years, and 1,973 in young adults (aged 20-29 years).
Between 2006 and 2015, the annual percentage change in melanoma incidence decreased by 4.4% for male adolescents, 5.4% for female adolescents, 3.7% for male young adults, and 3.6% for female young adults; these changes were statistically significant. The trends in incidence was similar when the population was limited to non-Hispanic whites, considered a high-risk group for melanoma.
By contrast, melanoma incidence increased by an annual percentage change of 1.8% for both men and women aged 40 years and older during the same period of time. Young adult women had a greater incidence of melanoma compared with young adult men (about twofold greater), but older men had a greater incidence of melanoma compared with older women, the researchers said.
The findings were limited by a lack of data about potential confounders, such as skin pigmentation, UV light exposure, sunburn history, sunscreen use, sun avoidance, protective clothing, and tanning bed use; and the absence of information kept the researchers from estimating an association between increased sun-protective behaviors and decreased incidence of melanoma.
“However, this change in behavior remains a plausible explanation for decreased melanoma rates in adolescent and young adult populations,” and the data support continued strategies to promote UV protection throughout life, they said.
The study was supported in part by the National Institutes of Health, the Fred Hutchinson Cancer Research Center Integrated Immunotherapy Research Core, and a Society for Immunotherapy of Cancer–Merck fellowship. Dr. Paulson disclosed grants from the Society for Immunotherapy of Cancer–Merck, bluebird biosciences, EMD Serono; she also disclosed an issued and licensed patent for a Merkel cell carcinoma T cell receptor.
SOURCE: Paulson KG et al. JAMA Dermatol. 2019. Nov 13. doi: 10.1001/jamadermatol.2019.3353.
FROM JAMA DERMATOLOGY
Apremilast for Behçet’s oral ulcers: Benefits maintained at 64 weeks
MADRID – of the long-term extension phase of the pivotal RELIEF trial, Alfred Mahr, MD, PhD, reported at the annual congress of the European Academy of Dermatology and Venereology.
“We now have strong evidence that apremilast is an effective and safe therapy to treat oral ulcers in patients with Behçet’s syndrome. I think this is a major advance in the field,” declared Dr. Mahr, a rheumatologist at St. Gallen (Switzerland) Cantonal Hospital.
Based largely upon the results of the 12-week, double-blind portion of the phase 3 RELIEF trial, the Food and Drug Administration approved apremilast (Otezla) for the treatment of oral ulcers in patients with Behçet’s disease in the summer of 2019.
The safety profile of the oral phosphodiesterase-4 inhibitor was as seen in other studies, including in patients with psoriatic arthritis, an FDA-approved indication for the drug since 2014. The main side effects in the long-term extension of RELIEF were diarrhea and nausea, typically mild or moderate in nature and roughly twice as frequent as in placebo-treated controls in the double-blind study phase.
“At the end of the day, at week 64, only 12% of patients treated with apremilast during the entire 64 weeks discontinued the drug due to a treatment-emergent adverse event, which I believe is a good indicator of the safety of this medication,” the rheumatologist said. “The overall feeling is that the benefit-to-risk ratio is very good and it’s a safe drug to prescribe.”
At the close of the initial 12-week, double-blind phase of RELIEF, 178 of the original 207 participants elected to enter the long-term extension, either staying on apremilast at 30 mg twice a day for an additional 52 weeks or switching to that regimen from placebo.
The focus of the long-term extension was on disease activity and quality of life outcomes. The results in patients who had switched from placebo to apremilast after 12 weeks proved to be reassuringly similar to outcomes in patients on the drug for the full duration. For example, the mean improvement on the patient-reported Behçet’s Syndrome Activity Scale was 18.6 points after 12 weeks of double-blind apremilast, 16.9 points after 64 weeks of continuous apremilast, and 16.8 points with 12 weeks of placebo followed by 52 weeks of active therapy.
After 12 weeks of double-blind apremilast, patients averaged a 3.4-point improvement on the Behçet’s Disease Quality of Life measure. After 64 weeks on the drug, the improvement over baseline was 3.6 points, while in the switch group it was 3.4 points. Similarly, on all three components of the SF-36 quality of life metric, the continuous apremilast group showed maintenance of effect from week 12 to week 64, while the placebo-to-apremilast group caught up. The same was true with regards to the Behçet’s Disease Current Activity Index, which encompasses measures of both the patient’s and clinician’s perception of disease activity.
At the outset of the RELIEF trial, participants averaged four oral ulcers. At week 64, the continuous apremilast group averaged 1.4 and the switch group 0.8, a nonsignificant difference.
Asked if apremilast had a favorable impact upon other manifestations of Behçet’s disease besides the oral ulcers, Dr. Mahr replied, “This is a very good question. People often wonder about it. We do, too. But this trial was not designed to capture less common manifestations of Behçet’s syndrome, such as genital ulcers. There have been some analyses done, but the number of patients who had genital ulcers at 12 weeks were very few. The same was true for eye manifestations. There was sort of a signal that it works, but we can’t prove it in a placebo-controlled trial.”
Dr. Mahr reported receiving research funding from and serving as a consultant to Celgene, the study sponsor.
MADRID – of the long-term extension phase of the pivotal RELIEF trial, Alfred Mahr, MD, PhD, reported at the annual congress of the European Academy of Dermatology and Venereology.
“We now have strong evidence that apremilast is an effective and safe therapy to treat oral ulcers in patients with Behçet’s syndrome. I think this is a major advance in the field,” declared Dr. Mahr, a rheumatologist at St. Gallen (Switzerland) Cantonal Hospital.
Based largely upon the results of the 12-week, double-blind portion of the phase 3 RELIEF trial, the Food and Drug Administration approved apremilast (Otezla) for the treatment of oral ulcers in patients with Behçet’s disease in the summer of 2019.
The safety profile of the oral phosphodiesterase-4 inhibitor was as seen in other studies, including in patients with psoriatic arthritis, an FDA-approved indication for the drug since 2014. The main side effects in the long-term extension of RELIEF were diarrhea and nausea, typically mild or moderate in nature and roughly twice as frequent as in placebo-treated controls in the double-blind study phase.
“At the end of the day, at week 64, only 12% of patients treated with apremilast during the entire 64 weeks discontinued the drug due to a treatment-emergent adverse event, which I believe is a good indicator of the safety of this medication,” the rheumatologist said. “The overall feeling is that the benefit-to-risk ratio is very good and it’s a safe drug to prescribe.”
At the close of the initial 12-week, double-blind phase of RELIEF, 178 of the original 207 participants elected to enter the long-term extension, either staying on apremilast at 30 mg twice a day for an additional 52 weeks or switching to that regimen from placebo.
The focus of the long-term extension was on disease activity and quality of life outcomes. The results in patients who had switched from placebo to apremilast after 12 weeks proved to be reassuringly similar to outcomes in patients on the drug for the full duration. For example, the mean improvement on the patient-reported Behçet’s Syndrome Activity Scale was 18.6 points after 12 weeks of double-blind apremilast, 16.9 points after 64 weeks of continuous apremilast, and 16.8 points with 12 weeks of placebo followed by 52 weeks of active therapy.
After 12 weeks of double-blind apremilast, patients averaged a 3.4-point improvement on the Behçet’s Disease Quality of Life measure. After 64 weeks on the drug, the improvement over baseline was 3.6 points, while in the switch group it was 3.4 points. Similarly, on all three components of the SF-36 quality of life metric, the continuous apremilast group showed maintenance of effect from week 12 to week 64, while the placebo-to-apremilast group caught up. The same was true with regards to the Behçet’s Disease Current Activity Index, which encompasses measures of both the patient’s and clinician’s perception of disease activity.
At the outset of the RELIEF trial, participants averaged four oral ulcers. At week 64, the continuous apremilast group averaged 1.4 and the switch group 0.8, a nonsignificant difference.
Asked if apremilast had a favorable impact upon other manifestations of Behçet’s disease besides the oral ulcers, Dr. Mahr replied, “This is a very good question. People often wonder about it. We do, too. But this trial was not designed to capture less common manifestations of Behçet’s syndrome, such as genital ulcers. There have been some analyses done, but the number of patients who had genital ulcers at 12 weeks were very few. The same was true for eye manifestations. There was sort of a signal that it works, but we can’t prove it in a placebo-controlled trial.”
Dr. Mahr reported receiving research funding from and serving as a consultant to Celgene, the study sponsor.
MADRID – of the long-term extension phase of the pivotal RELIEF trial, Alfred Mahr, MD, PhD, reported at the annual congress of the European Academy of Dermatology and Venereology.
“We now have strong evidence that apremilast is an effective and safe therapy to treat oral ulcers in patients with Behçet’s syndrome. I think this is a major advance in the field,” declared Dr. Mahr, a rheumatologist at St. Gallen (Switzerland) Cantonal Hospital.
Based largely upon the results of the 12-week, double-blind portion of the phase 3 RELIEF trial, the Food and Drug Administration approved apremilast (Otezla) for the treatment of oral ulcers in patients with Behçet’s disease in the summer of 2019.
The safety profile of the oral phosphodiesterase-4 inhibitor was as seen in other studies, including in patients with psoriatic arthritis, an FDA-approved indication for the drug since 2014. The main side effects in the long-term extension of RELIEF were diarrhea and nausea, typically mild or moderate in nature and roughly twice as frequent as in placebo-treated controls in the double-blind study phase.
“At the end of the day, at week 64, only 12% of patients treated with apremilast during the entire 64 weeks discontinued the drug due to a treatment-emergent adverse event, which I believe is a good indicator of the safety of this medication,” the rheumatologist said. “The overall feeling is that the benefit-to-risk ratio is very good and it’s a safe drug to prescribe.”
At the close of the initial 12-week, double-blind phase of RELIEF, 178 of the original 207 participants elected to enter the long-term extension, either staying on apremilast at 30 mg twice a day for an additional 52 weeks or switching to that regimen from placebo.
The focus of the long-term extension was on disease activity and quality of life outcomes. The results in patients who had switched from placebo to apremilast after 12 weeks proved to be reassuringly similar to outcomes in patients on the drug for the full duration. For example, the mean improvement on the patient-reported Behçet’s Syndrome Activity Scale was 18.6 points after 12 weeks of double-blind apremilast, 16.9 points after 64 weeks of continuous apremilast, and 16.8 points with 12 weeks of placebo followed by 52 weeks of active therapy.
After 12 weeks of double-blind apremilast, patients averaged a 3.4-point improvement on the Behçet’s Disease Quality of Life measure. After 64 weeks on the drug, the improvement over baseline was 3.6 points, while in the switch group it was 3.4 points. Similarly, on all three components of the SF-36 quality of life metric, the continuous apremilast group showed maintenance of effect from week 12 to week 64, while the placebo-to-apremilast group caught up. The same was true with regards to the Behçet’s Disease Current Activity Index, which encompasses measures of both the patient’s and clinician’s perception of disease activity.
At the outset of the RELIEF trial, participants averaged four oral ulcers. At week 64, the continuous apremilast group averaged 1.4 and the switch group 0.8, a nonsignificant difference.
Asked if apremilast had a favorable impact upon other manifestations of Behçet’s disease besides the oral ulcers, Dr. Mahr replied, “This is a very good question. People often wonder about it. We do, too. But this trial was not designed to capture less common manifestations of Behçet’s syndrome, such as genital ulcers. There have been some analyses done, but the number of patients who had genital ulcers at 12 weeks were very few. The same was true for eye manifestations. There was sort of a signal that it works, but we can’t prove it in a placebo-controlled trial.”
Dr. Mahr reported receiving research funding from and serving as a consultant to Celgene, the study sponsor.
REPORTING FROM EADV 2019
FDA announces approval of fifth adalimumab biosimilar, Abrilada
The Food and Drug Administration has cleared adalimumab-afzb (Abrilada) as the fifth approved Humira biosimilar and the 25th approved biosimilar drug overall, the agency said in a Nov. 15 announcement.
According to a press release from Pfizer, approval for Abrilada was based on review of a comprehensive data package demonstrating biosimilarity of the drug to the reference product. This included data from a clinical comparative study, which found no clinically meaningful difference between Abrilada and the reference in terms of efficacy, safety, and immunogenicity in patients with moderate to severe rheumatoid arthritis (RA). In addition to RA, Abrilada is indicated for juvenile idiopathic arthritis, psoriatic arthritis, ankylosing spondylitis, adult Crohn’s disease, ulcerative colitis, and plaque psoriasis.
Common adverse events in adalimumab clinical trials included infection, injection-site reactions, headache, and rash.
Pfizer said that it “is working to make Abrilada available to U.S. patients as soon as feasible based on the terms of our agreement with AbbVie [the manufacturer of Humira]. Our current plans are to launch in 2023.”
The Food and Drug Administration has cleared adalimumab-afzb (Abrilada) as the fifth approved Humira biosimilar and the 25th approved biosimilar drug overall, the agency said in a Nov. 15 announcement.
According to a press release from Pfizer, approval for Abrilada was based on review of a comprehensive data package demonstrating biosimilarity of the drug to the reference product. This included data from a clinical comparative study, which found no clinically meaningful difference between Abrilada and the reference in terms of efficacy, safety, and immunogenicity in patients with moderate to severe rheumatoid arthritis (RA). In addition to RA, Abrilada is indicated for juvenile idiopathic arthritis, psoriatic arthritis, ankylosing spondylitis, adult Crohn’s disease, ulcerative colitis, and plaque psoriasis.
Common adverse events in adalimumab clinical trials included infection, injection-site reactions, headache, and rash.
Pfizer said that it “is working to make Abrilada available to U.S. patients as soon as feasible based on the terms of our agreement with AbbVie [the manufacturer of Humira]. Our current plans are to launch in 2023.”
The Food and Drug Administration has cleared adalimumab-afzb (Abrilada) as the fifth approved Humira biosimilar and the 25th approved biosimilar drug overall, the agency said in a Nov. 15 announcement.
According to a press release from Pfizer, approval for Abrilada was based on review of a comprehensive data package demonstrating biosimilarity of the drug to the reference product. This included data from a clinical comparative study, which found no clinically meaningful difference between Abrilada and the reference in terms of efficacy, safety, and immunogenicity in patients with moderate to severe rheumatoid arthritis (RA). In addition to RA, Abrilada is indicated for juvenile idiopathic arthritis, psoriatic arthritis, ankylosing spondylitis, adult Crohn’s disease, ulcerative colitis, and plaque psoriasis.
Common adverse events in adalimumab clinical trials included infection, injection-site reactions, headache, and rash.
Pfizer said that it “is working to make Abrilada available to U.S. patients as soon as feasible based on the terms of our agreement with AbbVie [the manufacturer of Humira]. Our current plans are to launch in 2023.”
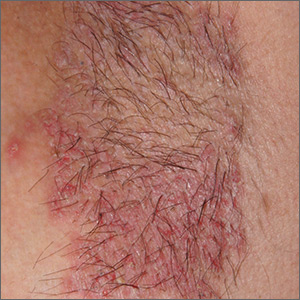
Recurring rash on neck and axilla
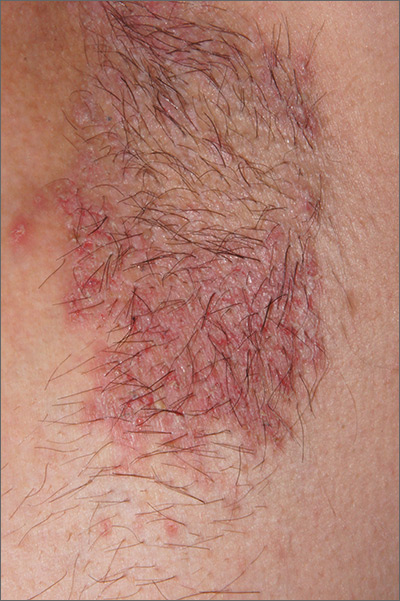
The FP initially treated the area with topical ketoconazole cream, which stung, but partially improved the patient’s symptoms. Because the rash persisted, the FP performed a punch biopsy, which showed widespread epidermal acantholysis or separation of epidermal cells. This is a hallmark of pemphigus vulgaris and benign familial pemphigus (Hailey-Hailey disease).
Hailey-Hailey disease is an uncommon autosomal dominant inherited blistering disorder that affects connecting proteins in the epidermis, which is why histology overlaps with pemphigus. Symptoms may not present until the second or third decade of life and often occur in flexural or high friction areas, including the axilla and inguinal folds. Any skin injury may trigger a flare, including sunburn, infections, heavy sweating, or friction from clothes. Bacterial overgrowth or colonization can cause a bad odor and social isolation in severe cases.
The differential diagnosis of axillary skin disorders is broad and includes irritant contact dermatitis, contact dermatitis, seborrheic dermatitis, hidradenitis suppurativa, candida intertrigo, and psoriasis. Clinical clues that favor Hailey-Hailey disease include fragile vesicles or pustules at the periphery and small focal erosions. The family history is helpful but not always known.
Some patients require topical therapy sequentially, in combination, or personalized through trial and error that addresses the inflammation, bacterial overgrowth, and fungal disease. Patients also may require long-term doxycycline therapy to suppress flares. Most patients will benefit from at least prn use of topical steroids for inflammation. Addressing sweating with topical aluminum chloride or botulinum injections can be beneficial. Low dose naltrexone, as well as afamelanotide, has shown promise in a few small case series.
The patient in this case improved with topical triamcinolone 0.1% ointment bid and systemic doxycycline 100 mg bid for 2 weeks. However, he continued to require occasional rounds of oral doxycycline with flares.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained).

The FP initially treated the area with topical ketoconazole cream, which stung, but partially improved the patient’s symptoms. Because the rash persisted, the FP performed a punch biopsy, which showed widespread epidermal acantholysis or separation of epidermal cells. This is a hallmark of pemphigus vulgaris and benign familial pemphigus (Hailey-Hailey disease).
Hailey-Hailey disease is an uncommon autosomal dominant inherited blistering disorder that affects connecting proteins in the epidermis, which is why histology overlaps with pemphigus. Symptoms may not present until the second or third decade of life and often occur in flexural or high friction areas, including the axilla and inguinal folds. Any skin injury may trigger a flare, including sunburn, infections, heavy sweating, or friction from clothes. Bacterial overgrowth or colonization can cause a bad odor and social isolation in severe cases.
The differential diagnosis of axillary skin disorders is broad and includes irritant contact dermatitis, contact dermatitis, seborrheic dermatitis, hidradenitis suppurativa, candida intertrigo, and psoriasis. Clinical clues that favor Hailey-Hailey disease include fragile vesicles or pustules at the periphery and small focal erosions. The family history is helpful but not always known.
Some patients require topical therapy sequentially, in combination, or personalized through trial and error that addresses the inflammation, bacterial overgrowth, and fungal disease. Patients also may require long-term doxycycline therapy to suppress flares. Most patients will benefit from at least prn use of topical steroids for inflammation. Addressing sweating with topical aluminum chloride or botulinum injections can be beneficial. Low dose naltrexone, as well as afamelanotide, has shown promise in a few small case series.
The patient in this case improved with topical triamcinolone 0.1% ointment bid and systemic doxycycline 100 mg bid for 2 weeks. However, he continued to require occasional rounds of oral doxycycline with flares.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained).

The FP initially treated the area with topical ketoconazole cream, which stung, but partially improved the patient’s symptoms. Because the rash persisted, the FP performed a punch biopsy, which showed widespread epidermal acantholysis or separation of epidermal cells. This is a hallmark of pemphigus vulgaris and benign familial pemphigus (Hailey-Hailey disease).
Hailey-Hailey disease is an uncommon autosomal dominant inherited blistering disorder that affects connecting proteins in the epidermis, which is why histology overlaps with pemphigus. Symptoms may not present until the second or third decade of life and often occur in flexural or high friction areas, including the axilla and inguinal folds. Any skin injury may trigger a flare, including sunburn, infections, heavy sweating, or friction from clothes. Bacterial overgrowth or colonization can cause a bad odor and social isolation in severe cases.
The differential diagnosis of axillary skin disorders is broad and includes irritant contact dermatitis, contact dermatitis, seborrheic dermatitis, hidradenitis suppurativa, candida intertrigo, and psoriasis. Clinical clues that favor Hailey-Hailey disease include fragile vesicles or pustules at the periphery and small focal erosions. The family history is helpful but not always known.
Some patients require topical therapy sequentially, in combination, or personalized through trial and error that addresses the inflammation, bacterial overgrowth, and fungal disease. Patients also may require long-term doxycycline therapy to suppress flares. Most patients will benefit from at least prn use of topical steroids for inflammation. Addressing sweating with topical aluminum chloride or botulinum injections can be beneficial. Low dose naltrexone, as well as afamelanotide, has shown promise in a few small case series.
The patient in this case improved with topical triamcinolone 0.1% ointment bid and systemic doxycycline 100 mg bid for 2 weeks. However, he continued to require occasional rounds of oral doxycycline with flares.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained).
Pink scaly rash on torso and extremities
Tumor necrosis factor–alpha (TNF-alpha) inhibitors are therapeutic agents used to treat a variety of inflammatory conditions such as rheumatoid arthritis and inflammatory bowel disease, as well as psoriasis of the skin (PSO) and psoriatic arthritis. In a 2017 systematic review, there were 216 reported cases of new-onset TNF-alpha inhibitor–induced psoriasis, with an estimated rate of 1 per 1,000. The cases thus far have had a wide range of presentations, the most common being plaque psoriasis, scalp psoriasis, as well as palmoplantar pustular psoriasis.1
A retrospective chart review study at Mayo clinic published in 2017 evaluated children younger than 19 years seen in 2003-2015 who developed new-onset or recurrent PSO with a history of inflammatory bowel disease being treated with anti-TNF-alpha therapy. The review showed variable latency in the development of PSO in these patients, although it typically occurred during inflammatory bowel disease remission.2 It is unclear whether there is an association between a personal or family history of psoriasis and development of these lesions.
TNF-alpha, interleukin (IL)–17) and interferon-alpha (IFN-alpha) are main cytokines that contribute to the development of psoriasis. The mechanism of action for paradoxical PSO/psoriasis in patients treated with anti-TNF is not clearly understood; however, many hypotheses are based on an imbalance between TNF-alpha and interferon-alpha – more specifically, an increased production of interferon-alpha. TNF-alpha inhibits the activity of plasmacytoid dendritic cells which are key producers of IFN-alpha. Because of this blockade, there is unopposed IFN-alpha production. Interferon-alpha allows for the expression of chemokines such as CXCR3, which favor T cells homing to the skin. IFN-alpha also stimulates and activates T cells to produce TNF-alpha and IL-17, which in turn sustains inflammatory mechanisms and allows for the development of psoriatic lesions.3
There are no universal management guidelines. Most of these patients’ treatment plans mirror standard psoriasis therapies while the main question remains the decision to continue the same anti-TNF therapy, change anti-TNF agents, or entirely switch classes of biologic or other systemic therapy. This decision in management requires several considerations: treatability of TNF-alpha inhibitor-induced psoriasis, the severity of background disease (i.e., rheumatoid arthritis, inflammatory bowel disease, other systemic condition), and whether the underlying disease is well controlled on current therapy, as well as the consideration of possible loss in efficacy if a drug is discontinued and then restarted at a later date.4
A reasonable initial approach in patients with well-controlled underlying disease and mild skin eruption is to continue anti-TNF therapy and manage skin topically with topical corticosteroids and/or phototherapy. In patients that either do not have well-controlled underlying disease or moderate skin involvement, changing to an alternative anti-TNF or other agent may be reasonable, and requires coordinated care with involved specialists. In the 2017 pediatric review mentioned previously, nearly half of the patients required a change in their initial anti-TNF-alpha agent despite conventional skin-directed therapies, and one-third of patients discontinued all anti-TNF-alpha therapy because of PSO.2
The psoriasiform papulosquamous features of this case along with the history suggests the diagnosis. Pityriasis rosea would be highly atypical on the feet and with the duration of findings. Lichen planus and atopic dermatitis morphology are inconsistent with this eruption, and coxsackie viral infection would have a shorter course.
Dr. Tracy is a research fellow in pediatric dermatology at Rady Children’s Hospital–San Diego and the University of California, San Diego. Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children’s Hospital–San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. Email them at pdnews@mdedge.com.
References
1. J Am Acad Dermatol. 2017 Feb;76(2):334-41.
2. Pediatr Dermatol. 2017 May;34(3):253-60.
3. RMD Open. 2016 Jul 15;2(2):e000239.
4. J Psoriasis Psoriatic Arthritis. 2019 Apr;4(2):70-80.
Tumor necrosis factor–alpha (TNF-alpha) inhibitors are therapeutic agents used to treat a variety of inflammatory conditions such as rheumatoid arthritis and inflammatory bowel disease, as well as psoriasis of the skin (PSO) and psoriatic arthritis. In a 2017 systematic review, there were 216 reported cases of new-onset TNF-alpha inhibitor–induced psoriasis, with an estimated rate of 1 per 1,000. The cases thus far have had a wide range of presentations, the most common being plaque psoriasis, scalp psoriasis, as well as palmoplantar pustular psoriasis.1
A retrospective chart review study at Mayo clinic published in 2017 evaluated children younger than 19 years seen in 2003-2015 who developed new-onset or recurrent PSO with a history of inflammatory bowel disease being treated with anti-TNF-alpha therapy. The review showed variable latency in the development of PSO in these patients, although it typically occurred during inflammatory bowel disease remission.2 It is unclear whether there is an association between a personal or family history of psoriasis and development of these lesions.
TNF-alpha, interleukin (IL)–17) and interferon-alpha (IFN-alpha) are main cytokines that contribute to the development of psoriasis. The mechanism of action for paradoxical PSO/psoriasis in patients treated with anti-TNF is not clearly understood; however, many hypotheses are based on an imbalance between TNF-alpha and interferon-alpha – more specifically, an increased production of interferon-alpha. TNF-alpha inhibits the activity of plasmacytoid dendritic cells which are key producers of IFN-alpha. Because of this blockade, there is unopposed IFN-alpha production. Interferon-alpha allows for the expression of chemokines such as CXCR3, which favor T cells homing to the skin. IFN-alpha also stimulates and activates T cells to produce TNF-alpha and IL-17, which in turn sustains inflammatory mechanisms and allows for the development of psoriatic lesions.3
There are no universal management guidelines. Most of these patients’ treatment plans mirror standard psoriasis therapies while the main question remains the decision to continue the same anti-TNF therapy, change anti-TNF agents, or entirely switch classes of biologic or other systemic therapy. This decision in management requires several considerations: treatability of TNF-alpha inhibitor-induced psoriasis, the severity of background disease (i.e., rheumatoid arthritis, inflammatory bowel disease, other systemic condition), and whether the underlying disease is well controlled on current therapy, as well as the consideration of possible loss in efficacy if a drug is discontinued and then restarted at a later date.4
A reasonable initial approach in patients with well-controlled underlying disease and mild skin eruption is to continue anti-TNF therapy and manage skin topically with topical corticosteroids and/or phototherapy. In patients that either do not have well-controlled underlying disease or moderate skin involvement, changing to an alternative anti-TNF or other agent may be reasonable, and requires coordinated care with involved specialists. In the 2017 pediatric review mentioned previously, nearly half of the patients required a change in their initial anti-TNF-alpha agent despite conventional skin-directed therapies, and one-third of patients discontinued all anti-TNF-alpha therapy because of PSO.2
The psoriasiform papulosquamous features of this case along with the history suggests the diagnosis. Pityriasis rosea would be highly atypical on the feet and with the duration of findings. Lichen planus and atopic dermatitis morphology are inconsistent with this eruption, and coxsackie viral infection would have a shorter course.
Dr. Tracy is a research fellow in pediatric dermatology at Rady Children’s Hospital–San Diego and the University of California, San Diego. Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children’s Hospital–San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. Email them at pdnews@mdedge.com.
References
1. J Am Acad Dermatol. 2017 Feb;76(2):334-41.
2. Pediatr Dermatol. 2017 May;34(3):253-60.
3. RMD Open. 2016 Jul 15;2(2):e000239.
4. J Psoriasis Psoriatic Arthritis. 2019 Apr;4(2):70-80.
Tumor necrosis factor–alpha (TNF-alpha) inhibitors are therapeutic agents used to treat a variety of inflammatory conditions such as rheumatoid arthritis and inflammatory bowel disease, as well as psoriasis of the skin (PSO) and psoriatic arthritis. In a 2017 systematic review, there were 216 reported cases of new-onset TNF-alpha inhibitor–induced psoriasis, with an estimated rate of 1 per 1,000. The cases thus far have had a wide range of presentations, the most common being plaque psoriasis, scalp psoriasis, as well as palmoplantar pustular psoriasis.1
A retrospective chart review study at Mayo clinic published in 2017 evaluated children younger than 19 years seen in 2003-2015 who developed new-onset or recurrent PSO with a history of inflammatory bowel disease being treated with anti-TNF-alpha therapy. The review showed variable latency in the development of PSO in these patients, although it typically occurred during inflammatory bowel disease remission.2 It is unclear whether there is an association between a personal or family history of psoriasis and development of these lesions.
TNF-alpha, interleukin (IL)–17) and interferon-alpha (IFN-alpha) are main cytokines that contribute to the development of psoriasis. The mechanism of action for paradoxical PSO/psoriasis in patients treated with anti-TNF is not clearly understood; however, many hypotheses are based on an imbalance between TNF-alpha and interferon-alpha – more specifically, an increased production of interferon-alpha. TNF-alpha inhibits the activity of plasmacytoid dendritic cells which are key producers of IFN-alpha. Because of this blockade, there is unopposed IFN-alpha production. Interferon-alpha allows for the expression of chemokines such as CXCR3, which favor T cells homing to the skin. IFN-alpha also stimulates and activates T cells to produce TNF-alpha and IL-17, which in turn sustains inflammatory mechanisms and allows for the development of psoriatic lesions.3
There are no universal management guidelines. Most of these patients’ treatment plans mirror standard psoriasis therapies while the main question remains the decision to continue the same anti-TNF therapy, change anti-TNF agents, or entirely switch classes of biologic or other systemic therapy. This decision in management requires several considerations: treatability of TNF-alpha inhibitor-induced psoriasis, the severity of background disease (i.e., rheumatoid arthritis, inflammatory bowel disease, other systemic condition), and whether the underlying disease is well controlled on current therapy, as well as the consideration of possible loss in efficacy if a drug is discontinued and then restarted at a later date.4
A reasonable initial approach in patients with well-controlled underlying disease and mild skin eruption is to continue anti-TNF therapy and manage skin topically with topical corticosteroids and/or phototherapy. In patients that either do not have well-controlled underlying disease or moderate skin involvement, changing to an alternative anti-TNF or other agent may be reasonable, and requires coordinated care with involved specialists. In the 2017 pediatric review mentioned previously, nearly half of the patients required a change in their initial anti-TNF-alpha agent despite conventional skin-directed therapies, and one-third of patients discontinued all anti-TNF-alpha therapy because of PSO.2
The psoriasiform papulosquamous features of this case along with the history suggests the diagnosis. Pityriasis rosea would be highly atypical on the feet and with the duration of findings. Lichen planus and atopic dermatitis morphology are inconsistent with this eruption, and coxsackie viral infection would have a shorter course.
Dr. Tracy is a research fellow in pediatric dermatology at Rady Children’s Hospital–San Diego and the University of California, San Diego. Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children’s Hospital–San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. Email them at pdnews@mdedge.com.
References
1. J Am Acad Dermatol. 2017 Feb;76(2):334-41.
2. Pediatr Dermatol. 2017 May;34(3):253-60.
3. RMD Open. 2016 Jul 15;2(2):e000239.
4. J Psoriasis Psoriatic Arthritis. 2019 Apr;4(2):70-80.
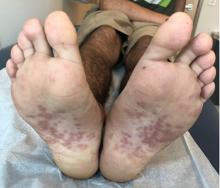
A 17-year-old male with a history of Crohn's disease, well controlled on infliximab, is seen for evaluation of a pink scaly rash on the torso, upper extremities, and lower extremities. The rash began 5 months previously and has been mostly asymptomatic. The patient denies pruritus or pain at the affected areas. There is no fever or drainage from any of the sites. The patient has not undergone any treatments. He does not have a personal or family history of chronic skin conditions.
On physical exam, he is noted to have numerous pink papules and plaques with overlying scale on his trunk, as well as the dorsal aspects of bilateral hands and the plantar surfaces of bilateral feet. His skin is otherwise unremarkable.
Expert shares tips for TNF-alpha inhibitor use in special populations
LAS VEGAS – Francisco A. Kerdel, BSc, MBBS, said at the Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar.
Dr. Kerdel, professor and vice chair of the department of dermatology at Florida International University, Miami, noted that, while tumor necrosis factor (TNF)–alpha inhibitors are category B drugs, inadequate data exist regarding lactation and exposure throughout pregnancy. “Rates of malformations and spontaneous abortions with therapy are similar to those in the general population, higher concentrations of infliximab and adalimumab have been found in infant and cord blood, compared with certolizumab pegol,” an anti-TNF biologic, he said.
In a prospective, postmarketing, multicenter pharmacokinetic study, researchers found a lack of placental transfer of certolizumab pegol during pregnancy (Ann Rheum Dis. 2018;77:228-33). Specifically, certolizumab levels were below the lower limit of quantification (less than 0.032 mcg/mL) in 13 of 14 infant samples at birth and in all infant samples at weeks 4 and 8. Only one infant had a minimal certolizumab level at birth (infant/mother ratio of 0.0009). No antibodies were detected at any time point during the study. Safety data in mothers were in line with the known safety profile of certolizumab and pregnancy profile of these underlying diseases. Adverse events experienced by the infants did not show any patterns or clusters of events suggesting a specific safety signal in children.
In a separate postmarketing pharmacokinetic study, investigators evaluated the transfer of certolizumab into breast milk (Ann Rheum Dis. 2017;76:1890-6). They found that the average daily infant dose of certolizumab was minimal. Specifically, the highest concentration of certolizumab in breast milk (0.0758 mcg/mL) was less than 1% of the expected mean plasma trough concentration of a therapeutic dose.
How do TNF-alpha inhibitors fare in the pediatric population? In a retrospective study of 390 children with psoriasis treated at 20 centers in the United States, Canada, and Europe, researchers evaluated the safety of systemic agents (JAMA Dermatol. 2017;153[11]:1147-57). Most (69%) were prescribed methotrexate, followed by biologics, acitretin, cyclosporine, and fumaric acid. Drug discontinuation (because of adverse events), which is sometimes used as an efficacy parameter, occurred in 12% of those who were on methotrexate, compared with 3% of those on biologics, 67% of those on acitretin, and 68% of those on fumaric acid.
At the other end of the age spectrum, biologic therapy is generally effective and well tolerated in elderly patients. “Sometimes, they may be more effective than other traditional drugs,” Dr. Kerdel said. “We’re a little bit concerned about immunosenescence, which can increase the risk for severe infections and malignancies. And, 90% of elderly patients with psoriasis may have comorbidities that need to be taken into account when treating psoriasis.”
Other factors come into play when choosing the right anti-TNF agent, including weight. While clinical trials show efficacy across weight groups, infliximab has weight-based dosing, “which may make it a better choice,” Dr. Kerdel said. “Patients taking etanercept may need a biweekly dose.”
Treatment flexibility also comes into play. For example, stopping therapy because of an infection or surgery may be problematic in drugs with a long half-life. Then there’s the issue of patient preference. “Some people don’t want to be injected frequently,” he said. “Some people don’t want to be injected at all and may require a simpler dosing regimen.”
Optimizing anti-TNF-alpha treatment starts with recognizing that there is a loss of response over time, Dr. Kerdel said, “or there may not be a response at all.” Contributing factors may include immunogenicity, suboptimal dosing, and poor patient adherence. In order to optimize treatment, clinicians can try switching agents or combination therapy, and explore continuous versus intermittent dosing.
“We really don’t have good data on the best protocol for switching treatment after failure of an anti-TNF-alpha agent,” he added. In cases of primary and secondary treatment failure, there is no consensus or guidelines on which second-line agent to use, nor good data on which measures to use.
No evidence-based guidelines are available for screening and monitoring patients receiving biologic therapy for psoriasis, either. “Evidence is strongest [grade B] for tuberculosis screening in patients treated with biologic agents,” Dr. Kerdel said. “Among known hepatitis B virus carriers, consider monitoring liver function tests and viral load [grade C]. High-grade evidence is lacking to support other routine testing. Physicians should use clinical judgment when screening and monitoring patients.”
He concluded his presentation by noting that there are a number of biosimilar agents available or in the pipeline for infliximab, adalimumab, and etanercept. This raises a number of questions for current and future consideration. For one, “will biosimilars show the same long-term efficacy and safety as the innovator products?” he asked. “Real-world, postmarketing, and registry data are needed. Will biosimilar agents offer significant cost benefits? Will biosimilar labeling be adequately transparent? Will we find biomarkers to help us target biologic agents to specific patients and subtypes of psoriasis?”
Dr. Kerdel reported that he is a member of the speaker’s bureau for AbbVie, Amgen, Celgene, Janssen, Novartis, Lilly, Leo, Ortho, and Novartis. He has also received grant/research support from AbbVie, Amgen, AstraZeneca, Celgene, Janssen, Leo, Lilly, Menlo Therapeutics, Novartis, Pfizer, and XBiotech.
SDEF and this news organization are owned by the same parent company.
LAS VEGAS – Francisco A. Kerdel, BSc, MBBS, said at the Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar.
Dr. Kerdel, professor and vice chair of the department of dermatology at Florida International University, Miami, noted that, while tumor necrosis factor (TNF)–alpha inhibitors are category B drugs, inadequate data exist regarding lactation and exposure throughout pregnancy. “Rates of malformations and spontaneous abortions with therapy are similar to those in the general population, higher concentrations of infliximab and adalimumab have been found in infant and cord blood, compared with certolizumab pegol,” an anti-TNF biologic, he said.
In a prospective, postmarketing, multicenter pharmacokinetic study, researchers found a lack of placental transfer of certolizumab pegol during pregnancy (Ann Rheum Dis. 2018;77:228-33). Specifically, certolizumab levels were below the lower limit of quantification (less than 0.032 mcg/mL) in 13 of 14 infant samples at birth and in all infant samples at weeks 4 and 8. Only one infant had a minimal certolizumab level at birth (infant/mother ratio of 0.0009). No antibodies were detected at any time point during the study. Safety data in mothers were in line with the known safety profile of certolizumab and pregnancy profile of these underlying diseases. Adverse events experienced by the infants did not show any patterns or clusters of events suggesting a specific safety signal in children.
In a separate postmarketing pharmacokinetic study, investigators evaluated the transfer of certolizumab into breast milk (Ann Rheum Dis. 2017;76:1890-6). They found that the average daily infant dose of certolizumab was minimal. Specifically, the highest concentration of certolizumab in breast milk (0.0758 mcg/mL) was less than 1% of the expected mean plasma trough concentration of a therapeutic dose.
How do TNF-alpha inhibitors fare in the pediatric population? In a retrospective study of 390 children with psoriasis treated at 20 centers in the United States, Canada, and Europe, researchers evaluated the safety of systemic agents (JAMA Dermatol. 2017;153[11]:1147-57). Most (69%) were prescribed methotrexate, followed by biologics, acitretin, cyclosporine, and fumaric acid. Drug discontinuation (because of adverse events), which is sometimes used as an efficacy parameter, occurred in 12% of those who were on methotrexate, compared with 3% of those on biologics, 67% of those on acitretin, and 68% of those on fumaric acid.
At the other end of the age spectrum, biologic therapy is generally effective and well tolerated in elderly patients. “Sometimes, they may be more effective than other traditional drugs,” Dr. Kerdel said. “We’re a little bit concerned about immunosenescence, which can increase the risk for severe infections and malignancies. And, 90% of elderly patients with psoriasis may have comorbidities that need to be taken into account when treating psoriasis.”
Other factors come into play when choosing the right anti-TNF agent, including weight. While clinical trials show efficacy across weight groups, infliximab has weight-based dosing, “which may make it a better choice,” Dr. Kerdel said. “Patients taking etanercept may need a biweekly dose.”
Treatment flexibility also comes into play. For example, stopping therapy because of an infection or surgery may be problematic in drugs with a long half-life. Then there’s the issue of patient preference. “Some people don’t want to be injected frequently,” he said. “Some people don’t want to be injected at all and may require a simpler dosing regimen.”
Optimizing anti-TNF-alpha treatment starts with recognizing that there is a loss of response over time, Dr. Kerdel said, “or there may not be a response at all.” Contributing factors may include immunogenicity, suboptimal dosing, and poor patient adherence. In order to optimize treatment, clinicians can try switching agents or combination therapy, and explore continuous versus intermittent dosing.
“We really don’t have good data on the best protocol for switching treatment after failure of an anti-TNF-alpha agent,” he added. In cases of primary and secondary treatment failure, there is no consensus or guidelines on which second-line agent to use, nor good data on which measures to use.
No evidence-based guidelines are available for screening and monitoring patients receiving biologic therapy for psoriasis, either. “Evidence is strongest [grade B] for tuberculosis screening in patients treated with biologic agents,” Dr. Kerdel said. “Among known hepatitis B virus carriers, consider monitoring liver function tests and viral load [grade C]. High-grade evidence is lacking to support other routine testing. Physicians should use clinical judgment when screening and monitoring patients.”
He concluded his presentation by noting that there are a number of biosimilar agents available or in the pipeline for infliximab, adalimumab, and etanercept. This raises a number of questions for current and future consideration. For one, “will biosimilars show the same long-term efficacy and safety as the innovator products?” he asked. “Real-world, postmarketing, and registry data are needed. Will biosimilar agents offer significant cost benefits? Will biosimilar labeling be adequately transparent? Will we find biomarkers to help us target biologic agents to specific patients and subtypes of psoriasis?”
Dr. Kerdel reported that he is a member of the speaker’s bureau for AbbVie, Amgen, Celgene, Janssen, Novartis, Lilly, Leo, Ortho, and Novartis. He has also received grant/research support from AbbVie, Amgen, AstraZeneca, Celgene, Janssen, Leo, Lilly, Menlo Therapeutics, Novartis, Pfizer, and XBiotech.
SDEF and this news organization are owned by the same parent company.
LAS VEGAS – Francisco A. Kerdel, BSc, MBBS, said at the Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar.
Dr. Kerdel, professor and vice chair of the department of dermatology at Florida International University, Miami, noted that, while tumor necrosis factor (TNF)–alpha inhibitors are category B drugs, inadequate data exist regarding lactation and exposure throughout pregnancy. “Rates of malformations and spontaneous abortions with therapy are similar to those in the general population, higher concentrations of infliximab and adalimumab have been found in infant and cord blood, compared with certolizumab pegol,” an anti-TNF biologic, he said.
In a prospective, postmarketing, multicenter pharmacokinetic study, researchers found a lack of placental transfer of certolizumab pegol during pregnancy (Ann Rheum Dis. 2018;77:228-33). Specifically, certolizumab levels were below the lower limit of quantification (less than 0.032 mcg/mL) in 13 of 14 infant samples at birth and in all infant samples at weeks 4 and 8. Only one infant had a minimal certolizumab level at birth (infant/mother ratio of 0.0009). No antibodies were detected at any time point during the study. Safety data in mothers were in line with the known safety profile of certolizumab and pregnancy profile of these underlying diseases. Adverse events experienced by the infants did not show any patterns or clusters of events suggesting a specific safety signal in children.
In a separate postmarketing pharmacokinetic study, investigators evaluated the transfer of certolizumab into breast milk (Ann Rheum Dis. 2017;76:1890-6). They found that the average daily infant dose of certolizumab was minimal. Specifically, the highest concentration of certolizumab in breast milk (0.0758 mcg/mL) was less than 1% of the expected mean plasma trough concentration of a therapeutic dose.
How do TNF-alpha inhibitors fare in the pediatric population? In a retrospective study of 390 children with psoriasis treated at 20 centers in the United States, Canada, and Europe, researchers evaluated the safety of systemic agents (JAMA Dermatol. 2017;153[11]:1147-57). Most (69%) were prescribed methotrexate, followed by biologics, acitretin, cyclosporine, and fumaric acid. Drug discontinuation (because of adverse events), which is sometimes used as an efficacy parameter, occurred in 12% of those who were on methotrexate, compared with 3% of those on biologics, 67% of those on acitretin, and 68% of those on fumaric acid.
At the other end of the age spectrum, biologic therapy is generally effective and well tolerated in elderly patients. “Sometimes, they may be more effective than other traditional drugs,” Dr. Kerdel said. “We’re a little bit concerned about immunosenescence, which can increase the risk for severe infections and malignancies. And, 90% of elderly patients with psoriasis may have comorbidities that need to be taken into account when treating psoriasis.”
Other factors come into play when choosing the right anti-TNF agent, including weight. While clinical trials show efficacy across weight groups, infliximab has weight-based dosing, “which may make it a better choice,” Dr. Kerdel said. “Patients taking etanercept may need a biweekly dose.”
Treatment flexibility also comes into play. For example, stopping therapy because of an infection or surgery may be problematic in drugs with a long half-life. Then there’s the issue of patient preference. “Some people don’t want to be injected frequently,” he said. “Some people don’t want to be injected at all and may require a simpler dosing regimen.”
Optimizing anti-TNF-alpha treatment starts with recognizing that there is a loss of response over time, Dr. Kerdel said, “or there may not be a response at all.” Contributing factors may include immunogenicity, suboptimal dosing, and poor patient adherence. In order to optimize treatment, clinicians can try switching agents or combination therapy, and explore continuous versus intermittent dosing.
“We really don’t have good data on the best protocol for switching treatment after failure of an anti-TNF-alpha agent,” he added. In cases of primary and secondary treatment failure, there is no consensus or guidelines on which second-line agent to use, nor good data on which measures to use.
No evidence-based guidelines are available for screening and monitoring patients receiving biologic therapy for psoriasis, either. “Evidence is strongest [grade B] for tuberculosis screening in patients treated with biologic agents,” Dr. Kerdel said. “Among known hepatitis B virus carriers, consider monitoring liver function tests and viral load [grade C]. High-grade evidence is lacking to support other routine testing. Physicians should use clinical judgment when screening and monitoring patients.”
He concluded his presentation by noting that there are a number of biosimilar agents available or in the pipeline for infliximab, adalimumab, and etanercept. This raises a number of questions for current and future consideration. For one, “will biosimilars show the same long-term efficacy and safety as the innovator products?” he asked. “Real-world, postmarketing, and registry data are needed. Will biosimilar agents offer significant cost benefits? Will biosimilar labeling be adequately transparent? Will we find biomarkers to help us target biologic agents to specific patients and subtypes of psoriasis?”
Dr. Kerdel reported that he is a member of the speaker’s bureau for AbbVie, Amgen, Celgene, Janssen, Novartis, Lilly, Leo, Ortho, and Novartis. He has also received grant/research support from AbbVie, Amgen, AstraZeneca, Celgene, Janssen, Leo, Lilly, Menlo Therapeutics, Novartis, Pfizer, and XBiotech.
SDEF and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM THE SDEF LAS VEGAS DERMATOLOGY SEMINAR
Guselkumab improves psoriatic arthritis regardless of prior TNFi use
ATLANTA – Guselkumab improved outcomes in psoriatic arthritis patients regardless of past treatment with tumor necrosis factor inhibitors in the phase 3 DISCOVER-1 trial.
The anti-interleukin-23p19 monoclonal antibody is approved in the United States for the treatment of moderate to severe plaque psoriasis (PsO).
Benefits in psoriatic arthritis (PsA) were seen in both biologic-naive and tumor necrosis factor inhibitor (TNFi)–treated patients and occurred with both 4- and 8-week dosing regimens, Atul Deodhar, MD, reported during a plenary session at the annual meeting of the American College of Rheumatology.
For example, the primary endpoint of ACR 20 response at 24 weeks was achieved in 58.6% and 52.8% of patients randomized to receive 100 mg of guselkumab delivered subcutaneously either at baseline and every 4 weeks or at baseline, week 4, and then every 8 weeks, respectively, compared with 22.2% of those randomized to receive placebo, said Dr. Deodhar, professor of medicine at Oregon Health & Science University, Portland.
Greater proportions of patients in the guselkumab groups achieved ACR 20 response at week 16; ACR 50 response at weeks 16 and 24; ACR 70 response at week 24; Psoriasis Area and Severity Index 75, 90, and 100 responses at week 24; and minimal disease activity response at week 24, he said, adding that improvements were also seen with guselkumab versus placebo for the controlled major secondary endpoints of change from baseline in Health Assessment Questionnaire–Disability Index score, Short Form 36 Health Survey score, and investigator global assessment (IGA) of PsO response.
The response rates with guselkumab versus placebo were seen regardless of prior TNFi use, he said.
The study included 381 patients with active PsA, defined as three or more swollen joints, three or more tender joints, and C-reactive protein of 0.3 mg/dL or greater despite standard therapies. About 30% were exposed to up to two TNFi therapies and 10% were nonresponders or inadequate responders to those therapies.
Concomitant use of select nonbiologic disease-modifying antirheumatic drugs, oral corticosteroids, and NSAIDs was allowed, and patients with less than 5% improvement in tender plus swollen joints at week 16 could initiate or increase the dose of the permitted medications while continuing study treatment, Dr. Deodhar said.
The mean body surface area with PsO involvement was 13.4%; 42.5% of patients had an IGA of 3-4 for skin involvement. Mean swollen and tender joint counts were 9.8 and 19.3, respectively, indicating a population with moderate to severe disease, he added.
Serious adverse events, serious infections, and death occurred in 2.4%, 0.5%, and 0.3% of patients, respectively.
“Both guselkumab regimens were safe and well tolerated through week 24,” Dr. Deodhar said, noting that the safety profile was consistent with that established in the treatment of PsO and described in the label.
DISCOVER-1 was funded by Janssen Research & Development. Dr. Deodhar reported relationships (advisory board activity, consulting, and/or research grant funding) with several pharmaceutical companies including Janssen. Several coauthors are employees of Janssen.
SOURCE: Deodhar A et al. Arthritis Rheumatol. 2019;71(suppl 10), Abstract 807.
ATLANTA – Guselkumab improved outcomes in psoriatic arthritis patients regardless of past treatment with tumor necrosis factor inhibitors in the phase 3 DISCOVER-1 trial.
The anti-interleukin-23p19 monoclonal antibody is approved in the United States for the treatment of moderate to severe plaque psoriasis (PsO).
Benefits in psoriatic arthritis (PsA) were seen in both biologic-naive and tumor necrosis factor inhibitor (TNFi)–treated patients and occurred with both 4- and 8-week dosing regimens, Atul Deodhar, MD, reported during a plenary session at the annual meeting of the American College of Rheumatology.
For example, the primary endpoint of ACR 20 response at 24 weeks was achieved in 58.6% and 52.8% of patients randomized to receive 100 mg of guselkumab delivered subcutaneously either at baseline and every 4 weeks or at baseline, week 4, and then every 8 weeks, respectively, compared with 22.2% of those randomized to receive placebo, said Dr. Deodhar, professor of medicine at Oregon Health & Science University, Portland.
Greater proportions of patients in the guselkumab groups achieved ACR 20 response at week 16; ACR 50 response at weeks 16 and 24; ACR 70 response at week 24; Psoriasis Area and Severity Index 75, 90, and 100 responses at week 24; and minimal disease activity response at week 24, he said, adding that improvements were also seen with guselkumab versus placebo for the controlled major secondary endpoints of change from baseline in Health Assessment Questionnaire–Disability Index score, Short Form 36 Health Survey score, and investigator global assessment (IGA) of PsO response.
The response rates with guselkumab versus placebo were seen regardless of prior TNFi use, he said.
The study included 381 patients with active PsA, defined as three or more swollen joints, three or more tender joints, and C-reactive protein of 0.3 mg/dL or greater despite standard therapies. About 30% were exposed to up to two TNFi therapies and 10% were nonresponders or inadequate responders to those therapies.
Concomitant use of select nonbiologic disease-modifying antirheumatic drugs, oral corticosteroids, and NSAIDs was allowed, and patients with less than 5% improvement in tender plus swollen joints at week 16 could initiate or increase the dose of the permitted medications while continuing study treatment, Dr. Deodhar said.
The mean body surface area with PsO involvement was 13.4%; 42.5% of patients had an IGA of 3-4 for skin involvement. Mean swollen and tender joint counts were 9.8 and 19.3, respectively, indicating a population with moderate to severe disease, he added.
Serious adverse events, serious infections, and death occurred in 2.4%, 0.5%, and 0.3% of patients, respectively.
“Both guselkumab regimens were safe and well tolerated through week 24,” Dr. Deodhar said, noting that the safety profile was consistent with that established in the treatment of PsO and described in the label.
DISCOVER-1 was funded by Janssen Research & Development. Dr. Deodhar reported relationships (advisory board activity, consulting, and/or research grant funding) with several pharmaceutical companies including Janssen. Several coauthors are employees of Janssen.
SOURCE: Deodhar A et al. Arthritis Rheumatol. 2019;71(suppl 10), Abstract 807.
ATLANTA – Guselkumab improved outcomes in psoriatic arthritis patients regardless of past treatment with tumor necrosis factor inhibitors in the phase 3 DISCOVER-1 trial.
The anti-interleukin-23p19 monoclonal antibody is approved in the United States for the treatment of moderate to severe plaque psoriasis (PsO).
Benefits in psoriatic arthritis (PsA) were seen in both biologic-naive and tumor necrosis factor inhibitor (TNFi)–treated patients and occurred with both 4- and 8-week dosing regimens, Atul Deodhar, MD, reported during a plenary session at the annual meeting of the American College of Rheumatology.
For example, the primary endpoint of ACR 20 response at 24 weeks was achieved in 58.6% and 52.8% of patients randomized to receive 100 mg of guselkumab delivered subcutaneously either at baseline and every 4 weeks or at baseline, week 4, and then every 8 weeks, respectively, compared with 22.2% of those randomized to receive placebo, said Dr. Deodhar, professor of medicine at Oregon Health & Science University, Portland.
Greater proportions of patients in the guselkumab groups achieved ACR 20 response at week 16; ACR 50 response at weeks 16 and 24; ACR 70 response at week 24; Psoriasis Area and Severity Index 75, 90, and 100 responses at week 24; and minimal disease activity response at week 24, he said, adding that improvements were also seen with guselkumab versus placebo for the controlled major secondary endpoints of change from baseline in Health Assessment Questionnaire–Disability Index score, Short Form 36 Health Survey score, and investigator global assessment (IGA) of PsO response.
The response rates with guselkumab versus placebo were seen regardless of prior TNFi use, he said.
The study included 381 patients with active PsA, defined as three or more swollen joints, three or more tender joints, and C-reactive protein of 0.3 mg/dL or greater despite standard therapies. About 30% were exposed to up to two TNFi therapies and 10% were nonresponders or inadequate responders to those therapies.
Concomitant use of select nonbiologic disease-modifying antirheumatic drugs, oral corticosteroids, and NSAIDs was allowed, and patients with less than 5% improvement in tender plus swollen joints at week 16 could initiate or increase the dose of the permitted medications while continuing study treatment, Dr. Deodhar said.
The mean body surface area with PsO involvement was 13.4%; 42.5% of patients had an IGA of 3-4 for skin involvement. Mean swollen and tender joint counts were 9.8 and 19.3, respectively, indicating a population with moderate to severe disease, he added.
Serious adverse events, serious infections, and death occurred in 2.4%, 0.5%, and 0.3% of patients, respectively.
“Both guselkumab regimens were safe and well tolerated through week 24,” Dr. Deodhar said, noting that the safety profile was consistent with that established in the treatment of PsO and described in the label.
DISCOVER-1 was funded by Janssen Research & Development. Dr. Deodhar reported relationships (advisory board activity, consulting, and/or research grant funding) with several pharmaceutical companies including Janssen. Several coauthors are employees of Janssen.
SOURCE: Deodhar A et al. Arthritis Rheumatol. 2019;71(suppl 10), Abstract 807.