User login
Fast, aggressive eczema treatment linked to fewer food allergies by age 2
Researchers in Japan report that
For their research published in the Journal of Allergy and Clinical Immunology: In Practice, Yumiko Miyaji, MD, PhD, of Japan’s National Center for Child Health and Development in Tokyo and colleagues looked at 3 years’ worth of records for 177 infants younger than 1 year of age seen at a hospital allergy center for eczema. Of these infants, 89 were treated with betamethasone valerate within 4 months of disease onset, and 88 were treated after 4 months of onset. Most (142) were followed-up at 22-24 months, when all were in complete remission or near remission from eczema.
At follow-up, clinicians collected information about anaphylactic reactions to food, administered specific food challenges, and tested serum immunoglobin E levels for food allergens. Dr. Miyaji and colleagues found a significant difference in the prevalence of allergies between the early-treated and late-treated groups to chicken egg, cow’s milk, wheat, peanuts, soy, or fish (25% vs. 46%, respectively; P equal to .013). For individual food allergies, only chicken egg was associated with a statistically significant difference in prevalence (15% vs 36%, P equal to .006).
“Our present study may be the first to demonstrate that early aggressive topical corticosteroid treatment to shorten the duration of eczema in infants was significantly associated with a decrease in later development of [food allergies],” Dr. Miyaji and colleagues wrote in their analysis.
The investigators acknowledged as limitations of their study some between-group differences at baseline, with characteristics such as Staphylococcus aureus infections and some inflammatory biomarkers higher in the early treatment group.
The Japan Agency for Medical Research and Development supported the study, and the investigators disclosed no conflicts of interest related to their findings.
SOURCE: Miyaji Y et al. J Allergy Clin Immunol Pract. 2019. doi: 10.1016/j.jaip.2019.11.036
Researchers in Japan report that
For their research published in the Journal of Allergy and Clinical Immunology: In Practice, Yumiko Miyaji, MD, PhD, of Japan’s National Center for Child Health and Development in Tokyo and colleagues looked at 3 years’ worth of records for 177 infants younger than 1 year of age seen at a hospital allergy center for eczema. Of these infants, 89 were treated with betamethasone valerate within 4 months of disease onset, and 88 were treated after 4 months of onset. Most (142) were followed-up at 22-24 months, when all were in complete remission or near remission from eczema.
At follow-up, clinicians collected information about anaphylactic reactions to food, administered specific food challenges, and tested serum immunoglobin E levels for food allergens. Dr. Miyaji and colleagues found a significant difference in the prevalence of allergies between the early-treated and late-treated groups to chicken egg, cow’s milk, wheat, peanuts, soy, or fish (25% vs. 46%, respectively; P equal to .013). For individual food allergies, only chicken egg was associated with a statistically significant difference in prevalence (15% vs 36%, P equal to .006).
“Our present study may be the first to demonstrate that early aggressive topical corticosteroid treatment to shorten the duration of eczema in infants was significantly associated with a decrease in later development of [food allergies],” Dr. Miyaji and colleagues wrote in their analysis.
The investigators acknowledged as limitations of their study some between-group differences at baseline, with characteristics such as Staphylococcus aureus infections and some inflammatory biomarkers higher in the early treatment group.
The Japan Agency for Medical Research and Development supported the study, and the investigators disclosed no conflicts of interest related to their findings.
SOURCE: Miyaji Y et al. J Allergy Clin Immunol Pract. 2019. doi: 10.1016/j.jaip.2019.11.036
Researchers in Japan report that
For their research published in the Journal of Allergy and Clinical Immunology: In Practice, Yumiko Miyaji, MD, PhD, of Japan’s National Center for Child Health and Development in Tokyo and colleagues looked at 3 years’ worth of records for 177 infants younger than 1 year of age seen at a hospital allergy center for eczema. Of these infants, 89 were treated with betamethasone valerate within 4 months of disease onset, and 88 were treated after 4 months of onset. Most (142) were followed-up at 22-24 months, when all were in complete remission or near remission from eczema.
At follow-up, clinicians collected information about anaphylactic reactions to food, administered specific food challenges, and tested serum immunoglobin E levels for food allergens. Dr. Miyaji and colleagues found a significant difference in the prevalence of allergies between the early-treated and late-treated groups to chicken egg, cow’s milk, wheat, peanuts, soy, or fish (25% vs. 46%, respectively; P equal to .013). For individual food allergies, only chicken egg was associated with a statistically significant difference in prevalence (15% vs 36%, P equal to .006).
“Our present study may be the first to demonstrate that early aggressive topical corticosteroid treatment to shorten the duration of eczema in infants was significantly associated with a decrease in later development of [food allergies],” Dr. Miyaji and colleagues wrote in their analysis.
The investigators acknowledged as limitations of their study some between-group differences at baseline, with characteristics such as Staphylococcus aureus infections and some inflammatory biomarkers higher in the early treatment group.
The Japan Agency for Medical Research and Development supported the study, and the investigators disclosed no conflicts of interest related to their findings.
SOURCE: Miyaji Y et al. J Allergy Clin Immunol Pract. 2019. doi: 10.1016/j.jaip.2019.11.036
FROM THE JOURNAL OF ALLERGY AND CLINICAL IMMUNOLOGY: IN PRACTICE
Atopic dermatitis in egg-, milk-allergic kids may up anaphylaxis risk
compared with allergic patients without atopic dermatitis, based on retrospective data from 347 individuals.
Atopic dermatitis has been associated with increased risk of food allergies, but the association and predictive factors of skin reactions to certain foods remain unclear, wrote Bryce C. Hoffman, MD, of National Jewish Health, Denver, and colleagues.
In a letter published in the Annals of Allergy, Asthma & Immunology, the researchers identified children aged 0-18 years with peanut, cow’s milk, and/or egg allergies with or without atopic dermatitis (AD) using an institutional research database and conducted a retrospective study of medical records.
Overall, children with egg and milk allergies plus AD had significantly higher rates of anaphylaxis than allergic children without AD (47% vs. 11% for egg, 50% vs. 19% for milk). Anaphylaxis rates were similar in children with peanut allergies with or without AD (27% vs. 23%).
“This finding may suggest that skin barrier dysfunction plays a role in the severity of [food allergy]. However, this is not universal to all food antigens, and other mechanisms are likely important in the association of anaphylaxis with a particular food,” the researchers noted.
Rates of tolerance for both baked egg and baked milk were similar between AD and non-AD patients (83% vs. 61% for milk; 82% vs. 67% for egg). In addition, levels of total IgE were increased in children with egg and milk allergies plus AD, compared with children without AD. However, children with peanut allergies plus AD had decreased total IgE, compared with children with peanut allergies but no AD. This “may support a link between Th2 polarization and [food allergy] severity, ” Dr. Hoffman and associates wrote.
The findings were limited by several factors, including the retrospective study design, exclusion of many patients, and lack of data on the amount of food that triggered anaphylactic reactions, the researchers noted.
Nonetheless, the results suggest that children with atopic dermatitis and allergies to eggs and milk are at increased risk and that clinicians should counsel these patients and families about the potential for more-severe reactions to oral food challenges, Dr. Hoffman and associates concluded.
The study was supported by National Jewish Health and the Edelstein Family Chair of Pediatric Allergy and Immunology. The researchers had no financial conflicts to disclose.
SOURCE: Hoffman BC et al. Ann Allergy Asthma Immunol. 2019 Sep 11. doi: 10.1016/j.anai.2019.09.008.
compared with allergic patients without atopic dermatitis, based on retrospective data from 347 individuals.
Atopic dermatitis has been associated with increased risk of food allergies, but the association and predictive factors of skin reactions to certain foods remain unclear, wrote Bryce C. Hoffman, MD, of National Jewish Health, Denver, and colleagues.
In a letter published in the Annals of Allergy, Asthma & Immunology, the researchers identified children aged 0-18 years with peanut, cow’s milk, and/or egg allergies with or without atopic dermatitis (AD) using an institutional research database and conducted a retrospective study of medical records.
Overall, children with egg and milk allergies plus AD had significantly higher rates of anaphylaxis than allergic children without AD (47% vs. 11% for egg, 50% vs. 19% for milk). Anaphylaxis rates were similar in children with peanut allergies with or without AD (27% vs. 23%).
“This finding may suggest that skin barrier dysfunction plays a role in the severity of [food allergy]. However, this is not universal to all food antigens, and other mechanisms are likely important in the association of anaphylaxis with a particular food,” the researchers noted.
Rates of tolerance for both baked egg and baked milk were similar between AD and non-AD patients (83% vs. 61% for milk; 82% vs. 67% for egg). In addition, levels of total IgE were increased in children with egg and milk allergies plus AD, compared with children without AD. However, children with peanut allergies plus AD had decreased total IgE, compared with children with peanut allergies but no AD. This “may support a link between Th2 polarization and [food allergy] severity, ” Dr. Hoffman and associates wrote.
The findings were limited by several factors, including the retrospective study design, exclusion of many patients, and lack of data on the amount of food that triggered anaphylactic reactions, the researchers noted.
Nonetheless, the results suggest that children with atopic dermatitis and allergies to eggs and milk are at increased risk and that clinicians should counsel these patients and families about the potential for more-severe reactions to oral food challenges, Dr. Hoffman and associates concluded.
The study was supported by National Jewish Health and the Edelstein Family Chair of Pediatric Allergy and Immunology. The researchers had no financial conflicts to disclose.
SOURCE: Hoffman BC et al. Ann Allergy Asthma Immunol. 2019 Sep 11. doi: 10.1016/j.anai.2019.09.008.
compared with allergic patients without atopic dermatitis, based on retrospective data from 347 individuals.
Atopic dermatitis has been associated with increased risk of food allergies, but the association and predictive factors of skin reactions to certain foods remain unclear, wrote Bryce C. Hoffman, MD, of National Jewish Health, Denver, and colleagues.
In a letter published in the Annals of Allergy, Asthma & Immunology, the researchers identified children aged 0-18 years with peanut, cow’s milk, and/or egg allergies with or without atopic dermatitis (AD) using an institutional research database and conducted a retrospective study of medical records.
Overall, children with egg and milk allergies plus AD had significantly higher rates of anaphylaxis than allergic children without AD (47% vs. 11% for egg, 50% vs. 19% for milk). Anaphylaxis rates were similar in children with peanut allergies with or without AD (27% vs. 23%).
“This finding may suggest that skin barrier dysfunction plays a role in the severity of [food allergy]. However, this is not universal to all food antigens, and other mechanisms are likely important in the association of anaphylaxis with a particular food,” the researchers noted.
Rates of tolerance for both baked egg and baked milk were similar between AD and non-AD patients (83% vs. 61% for milk; 82% vs. 67% for egg). In addition, levels of total IgE were increased in children with egg and milk allergies plus AD, compared with children without AD. However, children with peanut allergies plus AD had decreased total IgE, compared with children with peanut allergies but no AD. This “may support a link between Th2 polarization and [food allergy] severity, ” Dr. Hoffman and associates wrote.
The findings were limited by several factors, including the retrospective study design, exclusion of many patients, and lack of data on the amount of food that triggered anaphylactic reactions, the researchers noted.
Nonetheless, the results suggest that children with atopic dermatitis and allergies to eggs and milk are at increased risk and that clinicians should counsel these patients and families about the potential for more-severe reactions to oral food challenges, Dr. Hoffman and associates concluded.
The study was supported by National Jewish Health and the Edelstein Family Chair of Pediatric Allergy and Immunology. The researchers had no financial conflicts to disclose.
SOURCE: Hoffman BC et al. Ann Allergy Asthma Immunol. 2019 Sep 11. doi: 10.1016/j.anai.2019.09.008.
FROM THE ANNALS OF ALLERGY, ASTHMA & IMMUNOLOGY
Frequent soaks ease pediatric atopic dermatitis
A regimen of twice-daily baths followed by occlusive moisturizer improved atopic dermatitis in children with moderate to severe disease more effectively than did a twice-weekly protocol, based on data from 42 children.
Guidelines for bathing frequency for children with atopic dermatitis are inconsistent and often confusing for parents, according to Ivan D. Cardona, MD, of Maine Medical Research Institute, Portland, and colleagues.
In a study published in the Journal of Allergy and Clinical Immunology: In Practice, the researchers randomized 42 children aged 6 months to 11 years with moderate to severe atopic dermatitis to a routine of twice-weekly “soak and seal” (SS) procedures consisting of soaking baths for 10 minutes or less, followed by an occlusive emollient, or to twice-daily SS with baths of 15-20 minutes followed by emollient. The groups were treated for 2 weeks, then switched protocols. The study included a total of four clinic visits over 5 weeks. All patients also received standard of care low-potency topical corticosteroids and moisturizer.
Overall, the frequent bathing (“wet method”) led to a decrease of 21.2 on the SCORing Atopic Dermatitis Index (SCORAD) compared with the less frequent bathing (“dry method”). Improvements in SCORAD (the primary outcome) correlated with a secondary outcome of improved scores on the parent-rated Atopic Dermatitis Quickscore.
The findings were limited by several factors including the small sample size, large rate of attrition prior to randomization among initially screened children, lack of data on environmental factors such as water temperature and quality, and the lack of a washout period between the treatment protocols, the researchers noted. They acknowledged that “twice-daily SS bathing in the real world can be time consuming, making adherence difficult for families.”
However, the results suggest that the frequent bathing protocol was safe and effective at improving symptoms of atopic dermatitis, and may reduce steroid use, they concluded.
The researchers had no financial conflicts to disclose.
SOURCE: Cardona ID et al. J Allergy Clin Immunol Pract. 2019 Nov 13. doi: 10.1016/j.jaip.2019.10.042.
A regimen of twice-daily baths followed by occlusive moisturizer improved atopic dermatitis in children with moderate to severe disease more effectively than did a twice-weekly protocol, based on data from 42 children.
Guidelines for bathing frequency for children with atopic dermatitis are inconsistent and often confusing for parents, according to Ivan D. Cardona, MD, of Maine Medical Research Institute, Portland, and colleagues.
In a study published in the Journal of Allergy and Clinical Immunology: In Practice, the researchers randomized 42 children aged 6 months to 11 years with moderate to severe atopic dermatitis to a routine of twice-weekly “soak and seal” (SS) procedures consisting of soaking baths for 10 minutes or less, followed by an occlusive emollient, or to twice-daily SS with baths of 15-20 minutes followed by emollient. The groups were treated for 2 weeks, then switched protocols. The study included a total of four clinic visits over 5 weeks. All patients also received standard of care low-potency topical corticosteroids and moisturizer.
Overall, the frequent bathing (“wet method”) led to a decrease of 21.2 on the SCORing Atopic Dermatitis Index (SCORAD) compared with the less frequent bathing (“dry method”). Improvements in SCORAD (the primary outcome) correlated with a secondary outcome of improved scores on the parent-rated Atopic Dermatitis Quickscore.
The findings were limited by several factors including the small sample size, large rate of attrition prior to randomization among initially screened children, lack of data on environmental factors such as water temperature and quality, and the lack of a washout period between the treatment protocols, the researchers noted. They acknowledged that “twice-daily SS bathing in the real world can be time consuming, making adherence difficult for families.”
However, the results suggest that the frequent bathing protocol was safe and effective at improving symptoms of atopic dermatitis, and may reduce steroid use, they concluded.
The researchers had no financial conflicts to disclose.
SOURCE: Cardona ID et al. J Allergy Clin Immunol Pract. 2019 Nov 13. doi: 10.1016/j.jaip.2019.10.042.
A regimen of twice-daily baths followed by occlusive moisturizer improved atopic dermatitis in children with moderate to severe disease more effectively than did a twice-weekly protocol, based on data from 42 children.
Guidelines for bathing frequency for children with atopic dermatitis are inconsistent and often confusing for parents, according to Ivan D. Cardona, MD, of Maine Medical Research Institute, Portland, and colleagues.
In a study published in the Journal of Allergy and Clinical Immunology: In Practice, the researchers randomized 42 children aged 6 months to 11 years with moderate to severe atopic dermatitis to a routine of twice-weekly “soak and seal” (SS) procedures consisting of soaking baths for 10 minutes or less, followed by an occlusive emollient, or to twice-daily SS with baths of 15-20 minutes followed by emollient. The groups were treated for 2 weeks, then switched protocols. The study included a total of four clinic visits over 5 weeks. All patients also received standard of care low-potency topical corticosteroids and moisturizer.
Overall, the frequent bathing (“wet method”) led to a decrease of 21.2 on the SCORing Atopic Dermatitis Index (SCORAD) compared with the less frequent bathing (“dry method”). Improvements in SCORAD (the primary outcome) correlated with a secondary outcome of improved scores on the parent-rated Atopic Dermatitis Quickscore.
The findings were limited by several factors including the small sample size, large rate of attrition prior to randomization among initially screened children, lack of data on environmental factors such as water temperature and quality, and the lack of a washout period between the treatment protocols, the researchers noted. They acknowledged that “twice-daily SS bathing in the real world can be time consuming, making adherence difficult for families.”
However, the results suggest that the frequent bathing protocol was safe and effective at improving symptoms of atopic dermatitis, and may reduce steroid use, they concluded.
The researchers had no financial conflicts to disclose.
SOURCE: Cardona ID et al. J Allergy Clin Immunol Pract. 2019 Nov 13. doi: 10.1016/j.jaip.2019.10.042.
FROM THE JOURNAL OF ALLERGY AND CLINICAL IMMUNOLOGY: IN PRACTICE
Evidence builds for effects of obesity, low physical activity on development of psoriatic arthritis
A new Norwegian study has identified a high body mass index and lower levels of physical activity as modifiable risk factors for developing psoriatic arthritis (PsA).
“Our study adds to the growing evidence that the risk of PsA is modifiable and highlights the importance of preventive work against obesity as well as encouraging physical activity in order to reduce the incidence of PsA,” wrote Ruth S. Thomsen, MD, of the Norwegian University of Science and Technology, and coauthors. The study was published in Arthritis Care & Research.
To determine the impact that adiposity and body fat distribution have on developing PsA, the researchers analyzed data from 36,626 women and men who participated in two surveys from the longitudinal, population-based Norwegian HUNT Study. All participants did not have diagnosed PsA at baseline in 1995-1997. Variables used in statistical analysis included calculated baseline BMI, waist circumference, waist-hip ratio, and level of physical activity.
Between baseline and follow-up in 2012, 185 new cases of PsA were reported. One standard deviation increase in BMI (4.2 kg/m2 for women, 3.5 kg/m2 for men) and waist circumstance (10.8 cm for women, 8.6 cm for men) was associated with adjusted hazard ratios of 1.40 (95% confidence interval, 1.24-1.58) and 1.48 (95% CI, 1.31-1.68), accordingly. Obese individuals – defined as BMI of 30 kg/m2 or higher – had an adjusted HR of 2.46 (95% CI, 1.65-3.68) when compared with individuals at normal weight.
Compared to individuals with BMI less than 25 kg/m2 and a high level of physical activity, individuals with BMI at 25 kg/m2 or higher and any lower level of physical activity had an adjusted HR of 2.06 (95% CI, 1.18-3.58). In addition, individuals with a high waist circumference – defined as 81 cm or more in women and 95 cm or more in men – and low physical activity had an adjusted HR of 2.22 (95% CI, 1.37-3.58) in comparison to those with a high waist circumference and high physical activity (adjusted HR, 1.84; 95% CI, 0.97-3.47). Physical activity level was considered low with less than 3 hours of light physical activity and no hard physical activity per week and high with any amount of light activity plus 1 or more hours of hard physical activity.
The authors acknowledged the study’s potential limitations, including the requirement for participants to complete the final two HUNT study surveys and the use of stricter criteria than usual in validating PsA diagnoses.
The study was partially funded by a grant Dr. Thomsen received from the Norwegian Extra Foundation for Health and Rehabilitation. The authors reported no conflicts of interest.
SOURCE: Thomsen RS et al. Arthritis Care Res. 2019 Dec 7. doi: 10.1002/acr.24121.
A new Norwegian study has identified a high body mass index and lower levels of physical activity as modifiable risk factors for developing psoriatic arthritis (PsA).
“Our study adds to the growing evidence that the risk of PsA is modifiable and highlights the importance of preventive work against obesity as well as encouraging physical activity in order to reduce the incidence of PsA,” wrote Ruth S. Thomsen, MD, of the Norwegian University of Science and Technology, and coauthors. The study was published in Arthritis Care & Research.
To determine the impact that adiposity and body fat distribution have on developing PsA, the researchers analyzed data from 36,626 women and men who participated in two surveys from the longitudinal, population-based Norwegian HUNT Study. All participants did not have diagnosed PsA at baseline in 1995-1997. Variables used in statistical analysis included calculated baseline BMI, waist circumference, waist-hip ratio, and level of physical activity.
Between baseline and follow-up in 2012, 185 new cases of PsA were reported. One standard deviation increase in BMI (4.2 kg/m2 for women, 3.5 kg/m2 for men) and waist circumstance (10.8 cm for women, 8.6 cm for men) was associated with adjusted hazard ratios of 1.40 (95% confidence interval, 1.24-1.58) and 1.48 (95% CI, 1.31-1.68), accordingly. Obese individuals – defined as BMI of 30 kg/m2 or higher – had an adjusted HR of 2.46 (95% CI, 1.65-3.68) when compared with individuals at normal weight.
Compared to individuals with BMI less than 25 kg/m2 and a high level of physical activity, individuals with BMI at 25 kg/m2 or higher and any lower level of physical activity had an adjusted HR of 2.06 (95% CI, 1.18-3.58). In addition, individuals with a high waist circumference – defined as 81 cm or more in women and 95 cm or more in men – and low physical activity had an adjusted HR of 2.22 (95% CI, 1.37-3.58) in comparison to those with a high waist circumference and high physical activity (adjusted HR, 1.84; 95% CI, 0.97-3.47). Physical activity level was considered low with less than 3 hours of light physical activity and no hard physical activity per week and high with any amount of light activity plus 1 or more hours of hard physical activity.
The authors acknowledged the study’s potential limitations, including the requirement for participants to complete the final two HUNT study surveys and the use of stricter criteria than usual in validating PsA diagnoses.
The study was partially funded by a grant Dr. Thomsen received from the Norwegian Extra Foundation for Health and Rehabilitation. The authors reported no conflicts of interest.
SOURCE: Thomsen RS et al. Arthritis Care Res. 2019 Dec 7. doi: 10.1002/acr.24121.
A new Norwegian study has identified a high body mass index and lower levels of physical activity as modifiable risk factors for developing psoriatic arthritis (PsA).
“Our study adds to the growing evidence that the risk of PsA is modifiable and highlights the importance of preventive work against obesity as well as encouraging physical activity in order to reduce the incidence of PsA,” wrote Ruth S. Thomsen, MD, of the Norwegian University of Science and Technology, and coauthors. The study was published in Arthritis Care & Research.
To determine the impact that adiposity and body fat distribution have on developing PsA, the researchers analyzed data from 36,626 women and men who participated in two surveys from the longitudinal, population-based Norwegian HUNT Study. All participants did not have diagnosed PsA at baseline in 1995-1997. Variables used in statistical analysis included calculated baseline BMI, waist circumference, waist-hip ratio, and level of physical activity.
Between baseline and follow-up in 2012, 185 new cases of PsA were reported. One standard deviation increase in BMI (4.2 kg/m2 for women, 3.5 kg/m2 for men) and waist circumstance (10.8 cm for women, 8.6 cm for men) was associated with adjusted hazard ratios of 1.40 (95% confidence interval, 1.24-1.58) and 1.48 (95% CI, 1.31-1.68), accordingly. Obese individuals – defined as BMI of 30 kg/m2 or higher – had an adjusted HR of 2.46 (95% CI, 1.65-3.68) when compared with individuals at normal weight.
Compared to individuals with BMI less than 25 kg/m2 and a high level of physical activity, individuals with BMI at 25 kg/m2 or higher and any lower level of physical activity had an adjusted HR of 2.06 (95% CI, 1.18-3.58). In addition, individuals with a high waist circumference – defined as 81 cm or more in women and 95 cm or more in men – and low physical activity had an adjusted HR of 2.22 (95% CI, 1.37-3.58) in comparison to those with a high waist circumference and high physical activity (adjusted HR, 1.84; 95% CI, 0.97-3.47). Physical activity level was considered low with less than 3 hours of light physical activity and no hard physical activity per week and high with any amount of light activity plus 1 or more hours of hard physical activity.
The authors acknowledged the study’s potential limitations, including the requirement for participants to complete the final two HUNT study surveys and the use of stricter criteria than usual in validating PsA diagnoses.
The study was partially funded by a grant Dr. Thomsen received from the Norwegian Extra Foundation for Health and Rehabilitation. The authors reported no conflicts of interest.
SOURCE: Thomsen RS et al. Arthritis Care Res. 2019 Dec 7. doi: 10.1002/acr.24121.
FROM ARTHRITIS CARE & RESEARCH
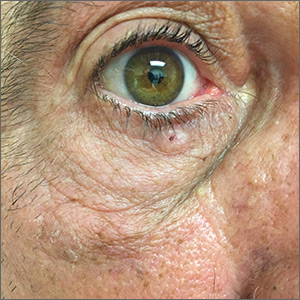
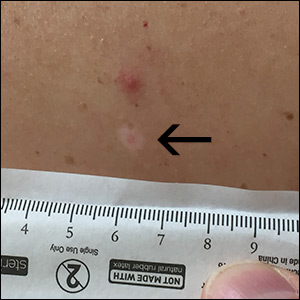
Pigmented lesion on face
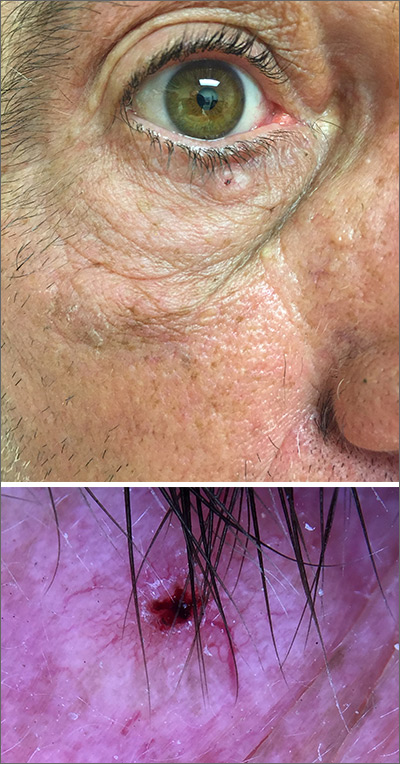
While the lesion’s proximity to the eyelashes and lid margin made dermoscopy difficult, the physician was able to use a dermatoscope to view the lesion and recognize it as nodular basal cell carcinoma (BCC). (If dermoscopy had not been an option, a hand magnifier or otoscope could have been used to help with magnification and diagnosis.)
Nodular BCCs usually present with a raised pearly border, a central ulceration, and telangiectasias. In this case, the central erosion was much more obvious with dermoscopy. Also visible were abnormal telangiectasias around the central erosion; they were especially dilated and tortuous (referred to as an arborizing pattern) at the 4:00 position. The diagnosis was confirmed by a small tangential shave biopsy of the inferior aspect of the lesion.
BCCs are referred for Mohs micrographic surgery (MMS) when they are any of the following: in high-risk locations such as the T-zone of the face (eyes, nose, and mouth); > 2 cm in diameter; a recurrence of a previous BCC; or a high-risk type including infiltrating, morpheaform, or basosquamous (based on pathology). Lower risk nodular BCCs are usually treated with excision or electrodesiccation and curettage.
In this case, the BCC was in a high-risk location and required MMS. The challenge was that the lesion was so close to the lid margin that resection of the cancer and subsequent repair could lead to ectropion/poor lid closure. The Mohs surgeon resected the lesion in 3 stages. The oculoplastic surgeon then closed the defect via a multilayered repair.
Images and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.

While the lesion’s proximity to the eyelashes and lid margin made dermoscopy difficult, the physician was able to use a dermatoscope to view the lesion and recognize it as nodular basal cell carcinoma (BCC). (If dermoscopy had not been an option, a hand magnifier or otoscope could have been used to help with magnification and diagnosis.)
Nodular BCCs usually present with a raised pearly border, a central ulceration, and telangiectasias. In this case, the central erosion was much more obvious with dermoscopy. Also visible were abnormal telangiectasias around the central erosion; they were especially dilated and tortuous (referred to as an arborizing pattern) at the 4:00 position. The diagnosis was confirmed by a small tangential shave biopsy of the inferior aspect of the lesion.
BCCs are referred for Mohs micrographic surgery (MMS) when they are any of the following: in high-risk locations such as the T-zone of the face (eyes, nose, and mouth); > 2 cm in diameter; a recurrence of a previous BCC; or a high-risk type including infiltrating, morpheaform, or basosquamous (based on pathology). Lower risk nodular BCCs are usually treated with excision or electrodesiccation and curettage.
In this case, the BCC was in a high-risk location and required MMS. The challenge was that the lesion was so close to the lid margin that resection of the cancer and subsequent repair could lead to ectropion/poor lid closure. The Mohs surgeon resected the lesion in 3 stages. The oculoplastic surgeon then closed the defect via a multilayered repair.
Images and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.

While the lesion’s proximity to the eyelashes and lid margin made dermoscopy difficult, the physician was able to use a dermatoscope to view the lesion and recognize it as nodular basal cell carcinoma (BCC). (If dermoscopy had not been an option, a hand magnifier or otoscope could have been used to help with magnification and diagnosis.)
Nodular BCCs usually present with a raised pearly border, a central ulceration, and telangiectasias. In this case, the central erosion was much more obvious with dermoscopy. Also visible were abnormal telangiectasias around the central erosion; they were especially dilated and tortuous (referred to as an arborizing pattern) at the 4:00 position. The diagnosis was confirmed by a small tangential shave biopsy of the inferior aspect of the lesion.
BCCs are referred for Mohs micrographic surgery (MMS) when they are any of the following: in high-risk locations such as the T-zone of the face (eyes, nose, and mouth); > 2 cm in diameter; a recurrence of a previous BCC; or a high-risk type including infiltrating, morpheaform, or basosquamous (based on pathology). Lower risk nodular BCCs are usually treated with excision or electrodesiccation and curettage.
In this case, the BCC was in a high-risk location and required MMS. The challenge was that the lesion was so close to the lid margin that resection of the cancer and subsequent repair could lead to ectropion/poor lid closure. The Mohs surgeon resected the lesion in 3 stages. The oculoplastic surgeon then closed the defect via a multilayered repair.
Images and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
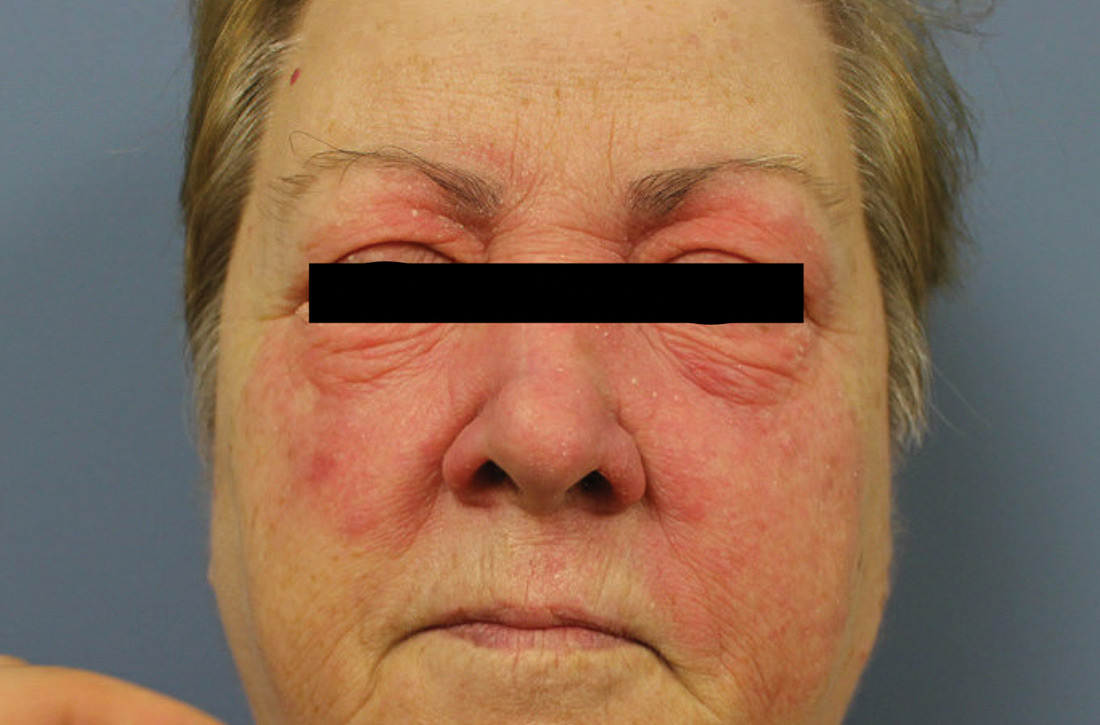
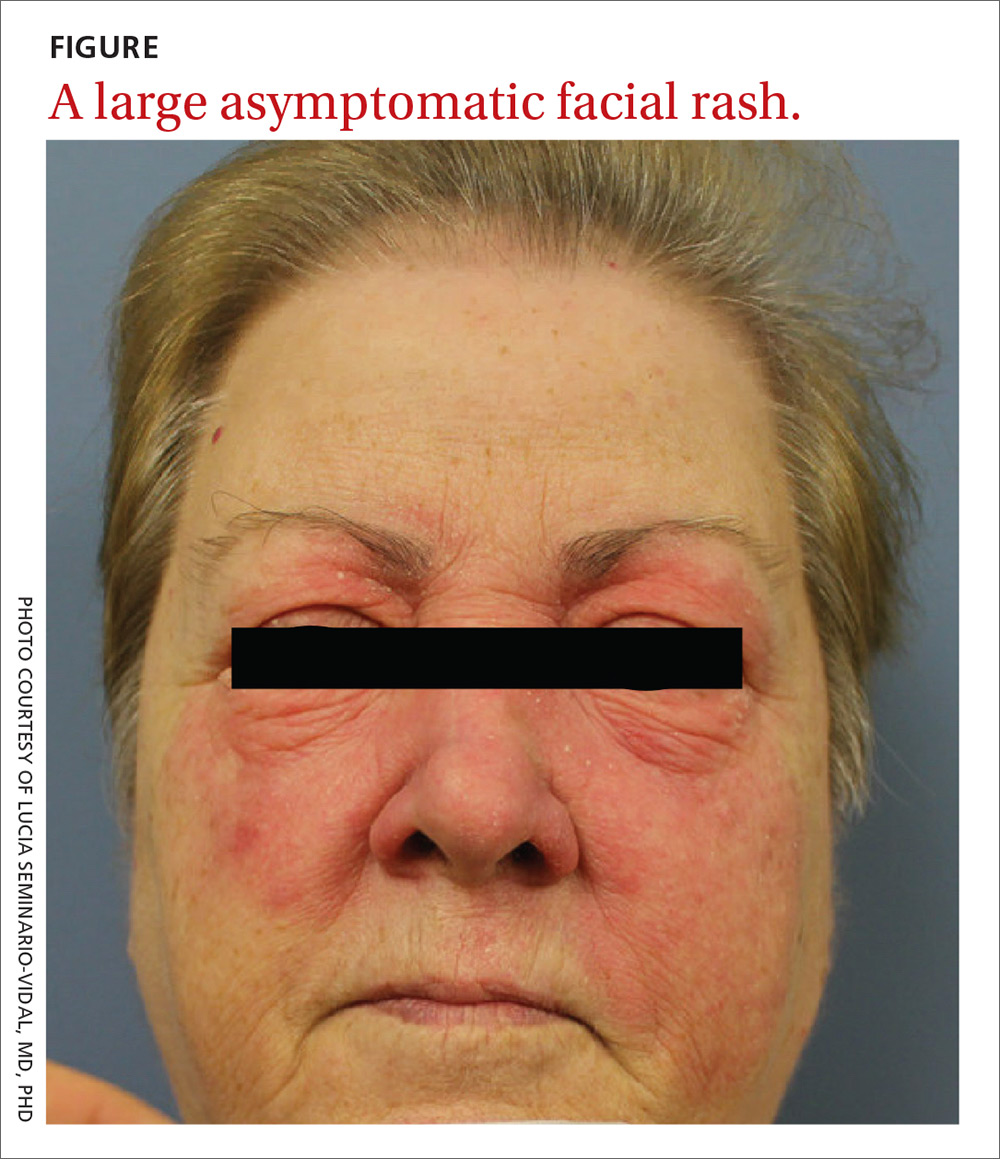
An erythematous facial rash
A 59-year-old woman presented to our clinic with a large asymptomatic facial rash that had developed several months earlier. The rash had been slowly growing but did not change day to day. Her past medical history was significant for hypertension, hyperlipidemia, and cutaneous lymphoma, which was localized to her arms. She denied the use of any new products, including hair or facial products, nail polish, or any new medications.
Initially, she was presumed (by an outside provider) to have rosacea, and she received treatment with doxycycline 100 mg/d for 2 months. However, the rash did not improve.
Physical examination revealed a large erythematous rash involving her cheeks, nose, and periocular area with no other significant findings (FIGURE).
A biopsy of her right cheek was performed.
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Mycosis fungoides
Following the biopsy of her right cheek, a histopathologic analysis demonstrated an atypical lymphocytic infiltrate positive for CD3 and CD4. These histopathologic features led to a diagnosis of recurrent mycosis fungoides (MF), a type of cutaneous lymphoma. (Our patient’s cutaneous lymphoma had been in remission for a year following local radiotherapy.)
MF is the most common type of cutaneous lymphoma, with an incidence of 6.4 to 9.6 cases per million people in the United States.1 There are also 2 rare subtypes of MF: the psoriasiform and palmoplantar forms. Psoriasiform MF presents with psoriasis-like plaques, while palmoplantar MF initially presents on the palms and soles.
Patients with classic MF typically present with patches and plaques—with the late evolution of tumors—on non–sun-exposed areas.1 Our patient’s clinical presentation was atypical because the rash manifested on a sun-exposed area of her body.
MF and other cutaneous lymphomas should always be part of the differential diagnosis for an unexplained persistent rash, especially in a patient with a history of MF. The development of lymphomas is thought to be a stepwise process through which chronic antigenic stimulation results in an accumulation of genetic mutations that then cause cells to undergo clonal expansion and, ultimately, malignant transformation. Genetic, environmental, and immunologic factors that contribute to the disease pathogenesis have been identified.2
Once clinical features point toward MF, the diagnosis can be further differentiated from other benign inflammatory mimics with a biopsy demonstrating cerebriform lymphocytes homing toward the epidermis, monoclonal expansion of T cells, and defective apoptosis.3
Continue to: Differential includes rosacea and seborrheic dermatitis
Differential includes rosacea and seborrheic dermatitis
The diagnosis of MF can be difficult as it often imitates other benign inflammatory conditions.
Rosacea manifests as an erythematous facial rash but usually spares the nasolabial folds and eyelids. There are several forms, including ocular (featuring swollen and irritated conjunctiva), erythematotelangiectatic (with visible blood vessels), and papulopustular (with acneic lesions). Over time, the skin may develop a thickened, bumpy texture, referred to as phymatous rosacea.4 A history of acute worsening with exposure to certain hot or spicy foods, alcohol, or ultraviolet light suggests a diagnosis of rosacea.
Seborrheic dermatitis classically presents as yellow scaling on a mildly erythematous base and often involves nasolabial folds and eyebrows. Seborrheic dermatitis can be associated with human immunodeficiency virus, Parkinson’s disease, and other chronic medical conditions.
Allergic contact dermatitis can look identical to MF, but in our case, there was no new allergen in the history. A thorough history regarding new medications, creams, and household supplies is integral to differentiating this diagnosis.
Misdiagnosis can lead to advanced-stage disease
This case of persistent facial erythema, originally treated as rosacea, highlights the importance of having a low threshold of suspicion of MF, especially in a patient with a prior history of MF. A recent study by Kelati et al3 indicated that certain subtypes of MF are easily misdiagnosed and treated as psoriasis or eczema respectively for an average of 10.5 years.3 These years of misdiagnosis are significantly correlated with the development of advanced-stage MF, which is more difficult to treat.3
Continue to: Treatment with topical desonide and mechlorethamine
Treatment with topical desonide and mechlorethamine
There are multiple treatment options for MF, depending on the stage, starting with topical therapies and advancing to systemic therapies in more advanced stages. Topical treatments include steroids, nitrogen mustard, and retinoids.5 Our patient was referred to a multidisciplinary lymphoma clinic, where topical treatment was initiated with desonide cream .05% and mechlorethamine gel .016%. Our patient experienced a 50% improvement in skin involvement at 3 months.
As MF progresses to more advanced stages, treatment often combines skin-directed therapies with systemic immunomodulators, biologics, radiation, and total skin electron beam therapy.6 TSEBT is a low-dose full-body radiation treatment that targets the skin surface and therefore effectively treats cutaneous lymphoma. Although TSEBT is usually well tolerated, there have been documented acute and chronic adverse effects, including dermatitis, alopecia, peripheral edema, cutaneous malignancies, and infertility in men.7
While the use of topical desonide and mechlorethamine was initially favored over radiation due to eyelid involvement, our patient developed new patches on her legs 11 months after her initial visit. When biopsies indicated MF with large cell transformation, she received 1 course of low-dose TSEBT (12 Gy), with complete response noted at the 2 month follow-up.
CORRESPONDENCE
Lucia Seminario-Vidal, MD, PhD, Department of Dermatology and Cutaneous Surgery, 13330 USF Laurel Drive, Tampa, FL 33612; luciasem@usf.edu
1. Jawed S, Myskowski P, Horwitz S, et al. Primary cutaneous T-cell lymphoma (mycosis fungoides and Sézary syndrome). Part I. Diagnosis: clinical and histopathologic features and new molecular and biologic markers. J Am Acad Dermatol. 2014;70:205.e1-e16.
2. Wohl Y, Tur E. Environmental risk factors for mycosis fungoides. Curr Probl Dermatol. 2007;35:52-64.
3. Kelati A, Gallouj S, Tahiri L, et al. Defining the mimics and clinico-histological diagnosis criteria for mycosis fungoides to minimize misdiagnosis. Int J Womens Dermatol. 2017;3:100-106.
4. Two AM, Wu W, Gallo RL, et al. Rosacea. part I. Introduction, categorization, histology, pathogenesis, and risk factors. J AM Acad Dermatol. 2015;72:749-758.
5. Lessin SR, Duvic M, Guitart J, et al. Topical chemotherapy in cutaneous T-cell lymphoma positive results of a randomized, controlled, multicenter trial testing the efficacy and safety of a novel mechlorethamine, 0.02%, gel in mycosis fungoides. JAMA Dermatol. 2013;149:25-32.
6. Jawed S, Myskowski P, Horwitz S, et al. Continuing medical education: Primary cutaneous T-cell lymphoma (mycosis fungoides and Sézary syndrome): part II. Prognosis, management, and future directions. J Am Acad Dermatol. 2014;70:223.e1-e17.
7. De Moraes FY, Carvalho Hde A, Hanna SA, et al. Literature review of clinical results of total skin electron irradiation (TSEBT) of mycosis fungoides in adults. Rep Pract Oncol Radiother. 2014;19:92-98.
A 59-year-old woman presented to our clinic with a large asymptomatic facial rash that had developed several months earlier. The rash had been slowly growing but did not change day to day. Her past medical history was significant for hypertension, hyperlipidemia, and cutaneous lymphoma, which was localized to her arms. She denied the use of any new products, including hair or facial products, nail polish, or any new medications.
Initially, she was presumed (by an outside provider) to have rosacea, and she received treatment with doxycycline 100 mg/d for 2 months. However, the rash did not improve.
Physical examination revealed a large erythematous rash involving her cheeks, nose, and periocular area with no other significant findings (FIGURE).

A biopsy of her right cheek was performed.
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Mycosis fungoides
Following the biopsy of her right cheek, a histopathologic analysis demonstrated an atypical lymphocytic infiltrate positive for CD3 and CD4. These histopathologic features led to a diagnosis of recurrent mycosis fungoides (MF), a type of cutaneous lymphoma. (Our patient’s cutaneous lymphoma had been in remission for a year following local radiotherapy.)
MF is the most common type of cutaneous lymphoma, with an incidence of 6.4 to 9.6 cases per million people in the United States.1 There are also 2 rare subtypes of MF: the psoriasiform and palmoplantar forms. Psoriasiform MF presents with psoriasis-like plaques, while palmoplantar MF initially presents on the palms and soles.
Patients with classic MF typically present with patches and plaques—with the late evolution of tumors—on non–sun-exposed areas.1 Our patient’s clinical presentation was atypical because the rash manifested on a sun-exposed area of her body.
MF and other cutaneous lymphomas should always be part of the differential diagnosis for an unexplained persistent rash, especially in a patient with a history of MF. The development of lymphomas is thought to be a stepwise process through which chronic antigenic stimulation results in an accumulation of genetic mutations that then cause cells to undergo clonal expansion and, ultimately, malignant transformation. Genetic, environmental, and immunologic factors that contribute to the disease pathogenesis have been identified.2
Once clinical features point toward MF, the diagnosis can be further differentiated from other benign inflammatory mimics with a biopsy demonstrating cerebriform lymphocytes homing toward the epidermis, monoclonal expansion of T cells, and defective apoptosis.3
Continue to: Differential includes rosacea and seborrheic dermatitis
Differential includes rosacea and seborrheic dermatitis
The diagnosis of MF can be difficult as it often imitates other benign inflammatory conditions.
Rosacea manifests as an erythematous facial rash but usually spares the nasolabial folds and eyelids. There are several forms, including ocular (featuring swollen and irritated conjunctiva), erythematotelangiectatic (with visible blood vessels), and papulopustular (with acneic lesions). Over time, the skin may develop a thickened, bumpy texture, referred to as phymatous rosacea.4 A history of acute worsening with exposure to certain hot or spicy foods, alcohol, or ultraviolet light suggests a diagnosis of rosacea.
Seborrheic dermatitis classically presents as yellow scaling on a mildly erythematous base and often involves nasolabial folds and eyebrows. Seborrheic dermatitis can be associated with human immunodeficiency virus, Parkinson’s disease, and other chronic medical conditions.
Allergic contact dermatitis can look identical to MF, but in our case, there was no new allergen in the history. A thorough history regarding new medications, creams, and household supplies is integral to differentiating this diagnosis.
Misdiagnosis can lead to advanced-stage disease
This case of persistent facial erythema, originally treated as rosacea, highlights the importance of having a low threshold of suspicion of MF, especially in a patient with a prior history of MF. A recent study by Kelati et al3 indicated that certain subtypes of MF are easily misdiagnosed and treated as psoriasis or eczema respectively for an average of 10.5 years.3 These years of misdiagnosis are significantly correlated with the development of advanced-stage MF, which is more difficult to treat.3
Continue to: Treatment with topical desonide and mechlorethamine
Treatment with topical desonide and mechlorethamine
There are multiple treatment options for MF, depending on the stage, starting with topical therapies and advancing to systemic therapies in more advanced stages. Topical treatments include steroids, nitrogen mustard, and retinoids.5 Our patient was referred to a multidisciplinary lymphoma clinic, where topical treatment was initiated with desonide cream .05% and mechlorethamine gel .016%. Our patient experienced a 50% improvement in skin involvement at 3 months.
As MF progresses to more advanced stages, treatment often combines skin-directed therapies with systemic immunomodulators, biologics, radiation, and total skin electron beam therapy.6 TSEBT is a low-dose full-body radiation treatment that targets the skin surface and therefore effectively treats cutaneous lymphoma. Although TSEBT is usually well tolerated, there have been documented acute and chronic adverse effects, including dermatitis, alopecia, peripheral edema, cutaneous malignancies, and infertility in men.7
While the use of topical desonide and mechlorethamine was initially favored over radiation due to eyelid involvement, our patient developed new patches on her legs 11 months after her initial visit. When biopsies indicated MF with large cell transformation, she received 1 course of low-dose TSEBT (12 Gy), with complete response noted at the 2 month follow-up.
CORRESPONDENCE
Lucia Seminario-Vidal, MD, PhD, Department of Dermatology and Cutaneous Surgery, 13330 USF Laurel Drive, Tampa, FL 33612; luciasem@usf.edu
A 59-year-old woman presented to our clinic with a large asymptomatic facial rash that had developed several months earlier. The rash had been slowly growing but did not change day to day. Her past medical history was significant for hypertension, hyperlipidemia, and cutaneous lymphoma, which was localized to her arms. She denied the use of any new products, including hair or facial products, nail polish, or any new medications.
Initially, she was presumed (by an outside provider) to have rosacea, and she received treatment with doxycycline 100 mg/d for 2 months. However, the rash did not improve.
Physical examination revealed a large erythematous rash involving her cheeks, nose, and periocular area with no other significant findings (FIGURE).

A biopsy of her right cheek was performed.
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Mycosis fungoides
Following the biopsy of her right cheek, a histopathologic analysis demonstrated an atypical lymphocytic infiltrate positive for CD3 and CD4. These histopathologic features led to a diagnosis of recurrent mycosis fungoides (MF), a type of cutaneous lymphoma. (Our patient’s cutaneous lymphoma had been in remission for a year following local radiotherapy.)
MF is the most common type of cutaneous lymphoma, with an incidence of 6.4 to 9.6 cases per million people in the United States.1 There are also 2 rare subtypes of MF: the psoriasiform and palmoplantar forms. Psoriasiform MF presents with psoriasis-like plaques, while palmoplantar MF initially presents on the palms and soles.
Patients with classic MF typically present with patches and plaques—with the late evolution of tumors—on non–sun-exposed areas.1 Our patient’s clinical presentation was atypical because the rash manifested on a sun-exposed area of her body.
MF and other cutaneous lymphomas should always be part of the differential diagnosis for an unexplained persistent rash, especially in a patient with a history of MF. The development of lymphomas is thought to be a stepwise process through which chronic antigenic stimulation results in an accumulation of genetic mutations that then cause cells to undergo clonal expansion and, ultimately, malignant transformation. Genetic, environmental, and immunologic factors that contribute to the disease pathogenesis have been identified.2
Once clinical features point toward MF, the diagnosis can be further differentiated from other benign inflammatory mimics with a biopsy demonstrating cerebriform lymphocytes homing toward the epidermis, monoclonal expansion of T cells, and defective apoptosis.3
Continue to: Differential includes rosacea and seborrheic dermatitis
Differential includes rosacea and seborrheic dermatitis
The diagnosis of MF can be difficult as it often imitates other benign inflammatory conditions.
Rosacea manifests as an erythematous facial rash but usually spares the nasolabial folds and eyelids. There are several forms, including ocular (featuring swollen and irritated conjunctiva), erythematotelangiectatic (with visible blood vessels), and papulopustular (with acneic lesions). Over time, the skin may develop a thickened, bumpy texture, referred to as phymatous rosacea.4 A history of acute worsening with exposure to certain hot or spicy foods, alcohol, or ultraviolet light suggests a diagnosis of rosacea.
Seborrheic dermatitis classically presents as yellow scaling on a mildly erythematous base and often involves nasolabial folds and eyebrows. Seborrheic dermatitis can be associated with human immunodeficiency virus, Parkinson’s disease, and other chronic medical conditions.
Allergic contact dermatitis can look identical to MF, but in our case, there was no new allergen in the history. A thorough history regarding new medications, creams, and household supplies is integral to differentiating this diagnosis.
Misdiagnosis can lead to advanced-stage disease
This case of persistent facial erythema, originally treated as rosacea, highlights the importance of having a low threshold of suspicion of MF, especially in a patient with a prior history of MF. A recent study by Kelati et al3 indicated that certain subtypes of MF are easily misdiagnosed and treated as psoriasis or eczema respectively for an average of 10.5 years.3 These years of misdiagnosis are significantly correlated with the development of advanced-stage MF, which is more difficult to treat.3
Continue to: Treatment with topical desonide and mechlorethamine
Treatment with topical desonide and mechlorethamine
There are multiple treatment options for MF, depending on the stage, starting with topical therapies and advancing to systemic therapies in more advanced stages. Topical treatments include steroids, nitrogen mustard, and retinoids.5 Our patient was referred to a multidisciplinary lymphoma clinic, where topical treatment was initiated with desonide cream .05% and mechlorethamine gel .016%. Our patient experienced a 50% improvement in skin involvement at 3 months.
As MF progresses to more advanced stages, treatment often combines skin-directed therapies with systemic immunomodulators, biologics, radiation, and total skin electron beam therapy.6 TSEBT is a low-dose full-body radiation treatment that targets the skin surface and therefore effectively treats cutaneous lymphoma. Although TSEBT is usually well tolerated, there have been documented acute and chronic adverse effects, including dermatitis, alopecia, peripheral edema, cutaneous malignancies, and infertility in men.7
While the use of topical desonide and mechlorethamine was initially favored over radiation due to eyelid involvement, our patient developed new patches on her legs 11 months after her initial visit. When biopsies indicated MF with large cell transformation, she received 1 course of low-dose TSEBT (12 Gy), with complete response noted at the 2 month follow-up.
CORRESPONDENCE
Lucia Seminario-Vidal, MD, PhD, Department of Dermatology and Cutaneous Surgery, 13330 USF Laurel Drive, Tampa, FL 33612; luciasem@usf.edu
1. Jawed S, Myskowski P, Horwitz S, et al. Primary cutaneous T-cell lymphoma (mycosis fungoides and Sézary syndrome). Part I. Diagnosis: clinical and histopathologic features and new molecular and biologic markers. J Am Acad Dermatol. 2014;70:205.e1-e16.
2. Wohl Y, Tur E. Environmental risk factors for mycosis fungoides. Curr Probl Dermatol. 2007;35:52-64.
3. Kelati A, Gallouj S, Tahiri L, et al. Defining the mimics and clinico-histological diagnosis criteria for mycosis fungoides to minimize misdiagnosis. Int J Womens Dermatol. 2017;3:100-106.
4. Two AM, Wu W, Gallo RL, et al. Rosacea. part I. Introduction, categorization, histology, pathogenesis, and risk factors. J AM Acad Dermatol. 2015;72:749-758.
5. Lessin SR, Duvic M, Guitart J, et al. Topical chemotherapy in cutaneous T-cell lymphoma positive results of a randomized, controlled, multicenter trial testing the efficacy and safety of a novel mechlorethamine, 0.02%, gel in mycosis fungoides. JAMA Dermatol. 2013;149:25-32.
6. Jawed S, Myskowski P, Horwitz S, et al. Continuing medical education: Primary cutaneous T-cell lymphoma (mycosis fungoides and Sézary syndrome): part II. Prognosis, management, and future directions. J Am Acad Dermatol. 2014;70:223.e1-e17.
7. De Moraes FY, Carvalho Hde A, Hanna SA, et al. Literature review of clinical results of total skin electron irradiation (TSEBT) of mycosis fungoides in adults. Rep Pract Oncol Radiother. 2014;19:92-98.
1. Jawed S, Myskowski P, Horwitz S, et al. Primary cutaneous T-cell lymphoma (mycosis fungoides and Sézary syndrome). Part I. Diagnosis: clinical and histopathologic features and new molecular and biologic markers. J Am Acad Dermatol. 2014;70:205.e1-e16.
2. Wohl Y, Tur E. Environmental risk factors for mycosis fungoides. Curr Probl Dermatol. 2007;35:52-64.
3. Kelati A, Gallouj S, Tahiri L, et al. Defining the mimics and clinico-histological diagnosis criteria for mycosis fungoides to minimize misdiagnosis. Int J Womens Dermatol. 2017;3:100-106.
4. Two AM, Wu W, Gallo RL, et al. Rosacea. part I. Introduction, categorization, histology, pathogenesis, and risk factors. J AM Acad Dermatol. 2015;72:749-758.
5. Lessin SR, Duvic M, Guitart J, et al. Topical chemotherapy in cutaneous T-cell lymphoma positive results of a randomized, controlled, multicenter trial testing the efficacy and safety of a novel mechlorethamine, 0.02%, gel in mycosis fungoides. JAMA Dermatol. 2013;149:25-32.
6. Jawed S, Myskowski P, Horwitz S, et al. Continuing medical education: Primary cutaneous T-cell lymphoma (mycosis fungoides and Sézary syndrome): part II. Prognosis, management, and future directions. J Am Acad Dermatol. 2014;70:223.e1-e17.
7. De Moraes FY, Carvalho Hde A, Hanna SA, et al. Literature review of clinical results of total skin electron irradiation (TSEBT) of mycosis fungoides in adults. Rep Pract Oncol Radiother. 2014;19:92-98.
FDA approves infliximab-axxq for numerous indications
The Food and Drug Administration has approved the biosimilar infliximab-axxq (Avsola) for various indications, making it the fourth biosimilar of infliximab (Remicade) to be cleared for marketing by the agency.
The tumor necrosis factor inhibitor is indicated for patients with Crohn’s disease or ulcerative colitis who are aged 6 years and older, RA in combination with methotrexate, ankylosing spondylitis, psoriatic arthritis, and plaque psoriasis. The approval is based on numerous trials. The most common adverse reactions are infections, infusion-related reactions, headache, and abdominal pain.
Full prescribing information can be found on the FDA website, as can more information about biosimilars.
The Food and Drug Administration has approved the biosimilar infliximab-axxq (Avsola) for various indications, making it the fourth biosimilar of infliximab (Remicade) to be cleared for marketing by the agency.
The tumor necrosis factor inhibitor is indicated for patients with Crohn’s disease or ulcerative colitis who are aged 6 years and older, RA in combination with methotrexate, ankylosing spondylitis, psoriatic arthritis, and plaque psoriasis. The approval is based on numerous trials. The most common adverse reactions are infections, infusion-related reactions, headache, and abdominal pain.
Full prescribing information can be found on the FDA website, as can more information about biosimilars.
The Food and Drug Administration has approved the biosimilar infliximab-axxq (Avsola) for various indications, making it the fourth biosimilar of infliximab (Remicade) to be cleared for marketing by the agency.
The tumor necrosis factor inhibitor is indicated for patients with Crohn’s disease or ulcerative colitis who are aged 6 years and older, RA in combination with methotrexate, ankylosing spondylitis, psoriatic arthritis, and plaque psoriasis. The approval is based on numerous trials. The most common adverse reactions are infections, infusion-related reactions, headache, and abdominal pain.
Full prescribing information can be found on the FDA website, as can more information about biosimilars.
Recurrent Angiotensin-Converting Enzyme Inhibitor-Induced Angioedema Refractory to Fresh Frozen Plasma
Angioedema induced by angiotensin-converting enzyme inhibitors (ACEIs) is present in from 0.1% to 0.7% of treated patients and more often involves the head, neck, face, lips, tongue, and larynx.1 ACEI-induced angioedema results from inhibition of angiotensin-converting enzyme (ACE), which results in reduced degradation and resultant accumulation of bradykinin, a potent inflammatory mediator.2
The treatment of choice is discontinuing all ACEIs; however, the patient may be at increased risk of a subsequent angioedema attack for many weeks.3 Antihistamines (H1 and H2 receptor blockade), epinephrine, and glucocorticoids are effective in allergic/histaminergic angioedema but are usually ineffective for hereditary angioedema or ACEI angioedema and are not recommended for acute therapy.4 Kallikrein-bradykinin pathway targeted therapies are now approved by the Food and Drug Administration (FDA) for hereditary angioedema attacks and have been studied for ACEI-induced angioedema. Ecallantide and icatibant inhibit conversion of precursors to bradykinin. Multiple randomized trials of ecallantide have not shown any advantage over traditional therapies.5 On the other hand, icatibant has shown resolution of angioedema in several case reports and in a randomized trial.6 Icatibant for ACEI-induced angioedema continues to be off-label because the data are conflicting.
Case Presentation
A 67-year-old man presented with a medical history of arterial hypertension (diagnosed 17 years previously), hypercholesterolemia, type 2 diabetes mellitus, alcohol dependence, and obesity. His outpatient medications included simvastatin, aripiprazole, losartan/hydrochlorothiazide, and amlodipine. He was voluntarily admitted for inpatient detoxification. After evaluation by the internist, medication reconciliation was done, and the therapy was adjusted according to medication availability. He reported having no drug allergies, and the losartan was changed for lisinopril. About 24 hours after the first dose of lisinopril, the patient developed swelling of the lips. Antihistamine and IV steroids were administered, and the ACEI was discontinued. His baseline vital signs were temperature 98° F, heart rate 83 beats per minute, respiratory rate 19 breaths per minute, blood pressure 150/94, and oxygen saturation 98% by pulse oximeter.
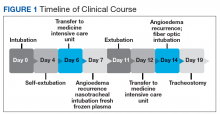
During the night shift the patient’s symptoms worsened, developing difficulty swallowing and shortness of breath. He was transferred to the medicine intensive care unit (MICU), intubated, and placed on mechanical ventilation to protect his airway. Laryngoscopic examination was notable for edematous tongue, uvula, and larynx. Also, the patient had mild stridor. His laboratory test results showed normal levels of complement, tryptase, and C1 esterase. On the fourth day after admission to MICU (Figure 1), the patient extubated himself. At that time, he did not present stridor or respiratory distress and remained at the MICU for 24 hours for close monitoring.
Thirty-six hours after self-extubation the patient developed stridor and shortness of breath at the general medicine ward. In view of his clinical presentation of recurrent ACEI-induced angioedema, the Anesthesiology Service was consulted. Direct visualization of the airways showed edema of the epiglottis and vocal cords, requiring nasotracheal intubation. Two units of fresh frozen plasma (FFP) were administered. Complete resolution of angioedema took at least 72 hours even after the administration of FFP. As part of the ventilator-associated pneumonia prevention bundle, the patient continued with daily spontaneous breathing trials. On the fourth day, he was he was extubated after a cuff-leak test was positive and his rapid shallow breathing index was adequate.
The cuff-leak test is usually done to predict postextubation stridor. It consists of deflating the endotracheal tube cuff to verify if gas can pass around the tube. Absence of cuff leak is suggestive of airway edema, a risk factor for postextubation stridor and failure of extubation. For example, if the patient has an endotracheal tube that is too large in relation to the patient’s airway, the leak test can result in a false negative. In this case, fiber optic visualization of the airway can confirm the endotracheal tube occluding all the airway even with the cuff deflated and without evidence of swelling of the vocal cords. The rapid shallow breathing index is a ratio of respiratory rate over tidal volume in liters and is used to predict successful extubation. Values < 105 have a high sensitivity for successful extubation.
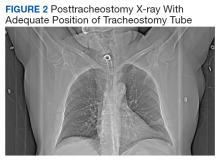
The patient remained under observation for 24 hours in the MICU and then was transferred to the general medicine ward. Unfortunately, 36 hours after, the patient had a new episode of angioedema requiring endotracheal intubation and placement on mechanical ventilation. This was his third episode of angioedema; he had a difficult airway classified as a Cormack-Lehane grade 3, requiring intubation with fiber-optic laryngoscope. In view of the recurrent events, a tracheostomy was done several days later. Figure 2 shows posttracheostomy X-ray with adequate position of the tracheostomy tube.
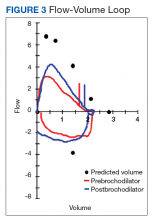
The patient was transferred to the Respiratory Care Unit and weaned off mechanical ventilation. He completed an intensive physical rehabilitation program and was discharged home. On discharge, he was followed by the Otorhinolaryngology Service and was decannulated about 5 months after. After tracheostomy decannulation, he developed asymptomatic stridor. A neck computer tomography scan revealed soft tissue thickening at the anterior and lateral aspects of the proximal tracheal likely representing granulation tissue/scarring. The findings were consistent with proximal tracheal stenosis sequelae of tracheostomy and intubation. In Figure 3, the upper portion of the curve represents the expiratory limb of the forced vital capacity and the lower portion represents inspiration. The flow-volume loop graph showed flattening of the inspiratory limb. There was a plateau in the inspiratory limb, suggestive of limitation of inspiratory flow as seen in variable extrathoracic lesions, such as glotticstricture, tumors, and vocal cord paralysis.7 The findings on the flow-volume loop were consistent with the subglottic stenosis identified by laryngoscopic examination. The patient was reluctant to undergo further interventions.
Discussion
The standard therapy for ACEI-inducedangioedema continues to be airway management and discontinuation of medication. However, life-threatening progression of symptoms have led to the use of off-label therapies, including FFP and bradykinin receptor antagonists, such as icatibant, which has been approved by the FDA for the treatment of hereditary angioedema. Icatibant is expensive and most hospitals do not have access to it. When considering the bradykinin pathway for therapy, FFP is commonly used. The cases described in the literature that have reported success with the use of FFP have used up to 2 units. There is no reported benefit of its use beyond 2 units. The initial randomized trials of icatibant for ACEI angioedema showed decreased time of resolution of angioedema.6 However, repeated trials showed conflicting results. At Veterans Affairs Caribbean Healthcare System, this medication was not available, and we decided to use FFP to improve the patient’s symptoms.
The administration of 2 units of FFP has been documented on case reports as a method to decrease the time of resolution of angioedema and the risk of recurrence. The mechanism of action thought to be involved includes the degradation of bradykinin by the enzyme ACE into inactive peptides and by supplying C1 inhibitor.8 No randomized clinical trial has investigated the use of FFP for the treatment of ACEI-induced angioedema. However, a retrospective cohort study report compared patients who presented with acute (nonhereditary) angioedema and airway compromise and received FFP with patients who were not treated with FFP.9 The study suggested a shorter ICU stay in the group treated with FFP, but the findings did not present statistical outcomes.
Nevertheless, our patient had recurrent ACEI-induced angioedema refractory to FFP. In addition to ACE or kininase II, FFP contains high-molecular weight-kininogen and kallikrein, the substrates that form bradykinin, which explained the mechanism of worsening angioedema.10 No randomized trials have investigated the use of FFP for the treatment of bradykinin-induced angioedema nor the appropriate dose.
Conclusion
In view of the emerging case reports of the effectiveness of FFP, this case of refractory angioedema raises concern for its true effectiveness and other possible factors involved in the mechanism of recurrence. Probably it would be unwise to conduct randomized studies in clinical situations such as the ones outlined. A collection of case series where FFP administration was done may be a more reasonable source of conclusions to be analyzed by a panel of experts.
1. Sánchez-Borges M, González-Aveledo LA. Angiotensin-converting enzyme inhibitors and angioedema. Allergy Asthma Immunol Res. 2010;2(3):195-198.
2. Kaplan AP. Angioedema. World Allergy Organ J. 2008;1(6):103-113.
3. Moellman JJ, Bernstein JA, Lindsell C, et al; American College of Allergy, Asthma & Immunology (ACAAI); Society for Academic Emergency Medicine (SAEM). A consensus parameter for the evaluation and management of angioedema in the emergency department. Acad Emerg Med. 2014;21(4):469-484.
4. LoVerde D, Files DC, Krishnaswamy G. Angioedema. Crit Care Med. 2017;45(4):725-735.
5. van den Elzen M, Go MFLC, Knulst AC, Blankestijn MA, van Os-Medendorp H, Otten HG. Efficacy of treatment of non-hereditary angioedema. Clinic Rev Allerg Immunol. 2018;54(3):412-431.
6. Bas M, Greve J, Stelter S, et al. A randomized trial of icatibant in ace-inhibitor–induced angioedema. N Engl J Med. 2015;372(5):418-425.
7. Diaz J, Casal J, Rodriguez W. Flow-volume loops: clinical correlation. PR Health Sci J. 2008;27(2):181-182.
8. Stewart M, McGlone R. Fresh frozen plasma in the treatment of ACE inhibitor-induced angioedema. BMJ Case Rep. 2012;2012:pii:bcr2012006849.
9. Saeb A, Hagglund KH, Cigolle CT. Using fresh frozen plasma for acute airway angioedema to prevent intubation in the emergency department: a retrospective cohort study. Emerg Med Int. 2016;2016:6091510.
10. Brown T, Gonzalez J, Monteleone C. Angiotensin-converting enzyme inhibitor-induced angioedema: a review of the literature. J Clin Hypertens (Greenwich). 2017;19(12):1377-1382.
Angioedema induced by angiotensin-converting enzyme inhibitors (ACEIs) is present in from 0.1% to 0.7% of treated patients and more often involves the head, neck, face, lips, tongue, and larynx.1 ACEI-induced angioedema results from inhibition of angiotensin-converting enzyme (ACE), which results in reduced degradation and resultant accumulation of bradykinin, a potent inflammatory mediator.2
The treatment of choice is discontinuing all ACEIs; however, the patient may be at increased risk of a subsequent angioedema attack for many weeks.3 Antihistamines (H1 and H2 receptor blockade), epinephrine, and glucocorticoids are effective in allergic/histaminergic angioedema but are usually ineffective for hereditary angioedema or ACEI angioedema and are not recommended for acute therapy.4 Kallikrein-bradykinin pathway targeted therapies are now approved by the Food and Drug Administration (FDA) for hereditary angioedema attacks and have been studied for ACEI-induced angioedema. Ecallantide and icatibant inhibit conversion of precursors to bradykinin. Multiple randomized trials of ecallantide have not shown any advantage over traditional therapies.5 On the other hand, icatibant has shown resolution of angioedema in several case reports and in a randomized trial.6 Icatibant for ACEI-induced angioedema continues to be off-label because the data are conflicting.
Case Presentation
A 67-year-old man presented with a medical history of arterial hypertension (diagnosed 17 years previously), hypercholesterolemia, type 2 diabetes mellitus, alcohol dependence, and obesity. His outpatient medications included simvastatin, aripiprazole, losartan/hydrochlorothiazide, and amlodipine. He was voluntarily admitted for inpatient detoxification. After evaluation by the internist, medication reconciliation was done, and the therapy was adjusted according to medication availability. He reported having no drug allergies, and the losartan was changed for lisinopril. About 24 hours after the first dose of lisinopril, the patient developed swelling of the lips. Antihistamine and IV steroids were administered, and the ACEI was discontinued. His baseline vital signs were temperature 98° F, heart rate 83 beats per minute, respiratory rate 19 breaths per minute, blood pressure 150/94, and oxygen saturation 98% by pulse oximeter.
During the night shift the patient’s symptoms worsened, developing difficulty swallowing and shortness of breath. He was transferred to the medicine intensive care unit (MICU), intubated, and placed on mechanical ventilation to protect his airway. Laryngoscopic examination was notable for edematous tongue, uvula, and larynx. Also, the patient had mild stridor. His laboratory test results showed normal levels of complement, tryptase, and C1 esterase. On the fourth day after admission to MICU (Figure 1), the patient extubated himself. At that time, he did not present stridor or respiratory distress and remained at the MICU for 24 hours for close monitoring.
Thirty-six hours after self-extubation the patient developed stridor and shortness of breath at the general medicine ward. In view of his clinical presentation of recurrent ACEI-induced angioedema, the Anesthesiology Service was consulted. Direct visualization of the airways showed edema of the epiglottis and vocal cords, requiring nasotracheal intubation. Two units of fresh frozen plasma (FFP) were administered. Complete resolution of angioedema took at least 72 hours even after the administration of FFP. As part of the ventilator-associated pneumonia prevention bundle, the patient continued with daily spontaneous breathing trials. On the fourth day, he was he was extubated after a cuff-leak test was positive and his rapid shallow breathing index was adequate.
The cuff-leak test is usually done to predict postextubation stridor. It consists of deflating the endotracheal tube cuff to verify if gas can pass around the tube. Absence of cuff leak is suggestive of airway edema, a risk factor for postextubation stridor and failure of extubation. For example, if the patient has an endotracheal tube that is too large in relation to the patient’s airway, the leak test can result in a false negative. In this case, fiber optic visualization of the airway can confirm the endotracheal tube occluding all the airway even with the cuff deflated and without evidence of swelling of the vocal cords. The rapid shallow breathing index is a ratio of respiratory rate over tidal volume in liters and is used to predict successful extubation. Values < 105 have a high sensitivity for successful extubation.
The patient remained under observation for 24 hours in the MICU and then was transferred to the general medicine ward. Unfortunately, 36 hours after, the patient had a new episode of angioedema requiring endotracheal intubation and placement on mechanical ventilation. This was his third episode of angioedema; he had a difficult airway classified as a Cormack-Lehane grade 3, requiring intubation with fiber-optic laryngoscope. In view of the recurrent events, a tracheostomy was done several days later. Figure 2 shows posttracheostomy X-ray with adequate position of the tracheostomy tube.
The patient was transferred to the Respiratory Care Unit and weaned off mechanical ventilation. He completed an intensive physical rehabilitation program and was discharged home. On discharge, he was followed by the Otorhinolaryngology Service and was decannulated about 5 months after. After tracheostomy decannulation, he developed asymptomatic stridor. A neck computer tomography scan revealed soft tissue thickening at the anterior and lateral aspects of the proximal tracheal likely representing granulation tissue/scarring. The findings were consistent with proximal tracheal stenosis sequelae of tracheostomy and intubation. In Figure 3, the upper portion of the curve represents the expiratory limb of the forced vital capacity and the lower portion represents inspiration. The flow-volume loop graph showed flattening of the inspiratory limb. There was a plateau in the inspiratory limb, suggestive of limitation of inspiratory flow as seen in variable extrathoracic lesions, such as glotticstricture, tumors, and vocal cord paralysis.7 The findings on the flow-volume loop were consistent with the subglottic stenosis identified by laryngoscopic examination. The patient was reluctant to undergo further interventions.
Discussion
The standard therapy for ACEI-inducedangioedema continues to be airway management and discontinuation of medication. However, life-threatening progression of symptoms have led to the use of off-label therapies, including FFP and bradykinin receptor antagonists, such as icatibant, which has been approved by the FDA for the treatment of hereditary angioedema. Icatibant is expensive and most hospitals do not have access to it. When considering the bradykinin pathway for therapy, FFP is commonly used. The cases described in the literature that have reported success with the use of FFP have used up to 2 units. There is no reported benefit of its use beyond 2 units. The initial randomized trials of icatibant for ACEI angioedema showed decreased time of resolution of angioedema.6 However, repeated trials showed conflicting results. At Veterans Affairs Caribbean Healthcare System, this medication was not available, and we decided to use FFP to improve the patient’s symptoms.
The administration of 2 units of FFP has been documented on case reports as a method to decrease the time of resolution of angioedema and the risk of recurrence. The mechanism of action thought to be involved includes the degradation of bradykinin by the enzyme ACE into inactive peptides and by supplying C1 inhibitor.8 No randomized clinical trial has investigated the use of FFP for the treatment of ACEI-induced angioedema. However, a retrospective cohort study report compared patients who presented with acute (nonhereditary) angioedema and airway compromise and received FFP with patients who were not treated with FFP.9 The study suggested a shorter ICU stay in the group treated with FFP, but the findings did not present statistical outcomes.
Nevertheless, our patient had recurrent ACEI-induced angioedema refractory to FFP. In addition to ACE or kininase II, FFP contains high-molecular weight-kininogen and kallikrein, the substrates that form bradykinin, which explained the mechanism of worsening angioedema.10 No randomized trials have investigated the use of FFP for the treatment of bradykinin-induced angioedema nor the appropriate dose.
Conclusion
In view of the emerging case reports of the effectiveness of FFP, this case of refractory angioedema raises concern for its true effectiveness and other possible factors involved in the mechanism of recurrence. Probably it would be unwise to conduct randomized studies in clinical situations such as the ones outlined. A collection of case series where FFP administration was done may be a more reasonable source of conclusions to be analyzed by a panel of experts.
Angioedema induced by angiotensin-converting enzyme inhibitors (ACEIs) is present in from 0.1% to 0.7% of treated patients and more often involves the head, neck, face, lips, tongue, and larynx.1 ACEI-induced angioedema results from inhibition of angiotensin-converting enzyme (ACE), which results in reduced degradation and resultant accumulation of bradykinin, a potent inflammatory mediator.2
The treatment of choice is discontinuing all ACEIs; however, the patient may be at increased risk of a subsequent angioedema attack for many weeks.3 Antihistamines (H1 and H2 receptor blockade), epinephrine, and glucocorticoids are effective in allergic/histaminergic angioedema but are usually ineffective for hereditary angioedema or ACEI angioedema and are not recommended for acute therapy.4 Kallikrein-bradykinin pathway targeted therapies are now approved by the Food and Drug Administration (FDA) for hereditary angioedema attacks and have been studied for ACEI-induced angioedema. Ecallantide and icatibant inhibit conversion of precursors to bradykinin. Multiple randomized trials of ecallantide have not shown any advantage over traditional therapies.5 On the other hand, icatibant has shown resolution of angioedema in several case reports and in a randomized trial.6 Icatibant for ACEI-induced angioedema continues to be off-label because the data are conflicting.
Case Presentation
A 67-year-old man presented with a medical history of arterial hypertension (diagnosed 17 years previously), hypercholesterolemia, type 2 diabetes mellitus, alcohol dependence, and obesity. His outpatient medications included simvastatin, aripiprazole, losartan/hydrochlorothiazide, and amlodipine. He was voluntarily admitted for inpatient detoxification. After evaluation by the internist, medication reconciliation was done, and the therapy was adjusted according to medication availability. He reported having no drug allergies, and the losartan was changed for lisinopril. About 24 hours after the first dose of lisinopril, the patient developed swelling of the lips. Antihistamine and IV steroids were administered, and the ACEI was discontinued. His baseline vital signs were temperature 98° F, heart rate 83 beats per minute, respiratory rate 19 breaths per minute, blood pressure 150/94, and oxygen saturation 98% by pulse oximeter.
During the night shift the patient’s symptoms worsened, developing difficulty swallowing and shortness of breath. He was transferred to the medicine intensive care unit (MICU), intubated, and placed on mechanical ventilation to protect his airway. Laryngoscopic examination was notable for edematous tongue, uvula, and larynx. Also, the patient had mild stridor. His laboratory test results showed normal levels of complement, tryptase, and C1 esterase. On the fourth day after admission to MICU (Figure 1), the patient extubated himself. At that time, he did not present stridor or respiratory distress and remained at the MICU for 24 hours for close monitoring.
Thirty-six hours after self-extubation the patient developed stridor and shortness of breath at the general medicine ward. In view of his clinical presentation of recurrent ACEI-induced angioedema, the Anesthesiology Service was consulted. Direct visualization of the airways showed edema of the epiglottis and vocal cords, requiring nasotracheal intubation. Two units of fresh frozen plasma (FFP) were administered. Complete resolution of angioedema took at least 72 hours even after the administration of FFP. As part of the ventilator-associated pneumonia prevention bundle, the patient continued with daily spontaneous breathing trials. On the fourth day, he was he was extubated after a cuff-leak test was positive and his rapid shallow breathing index was adequate.
The cuff-leak test is usually done to predict postextubation stridor. It consists of deflating the endotracheal tube cuff to verify if gas can pass around the tube. Absence of cuff leak is suggestive of airway edema, a risk factor for postextubation stridor and failure of extubation. For example, if the patient has an endotracheal tube that is too large in relation to the patient’s airway, the leak test can result in a false negative. In this case, fiber optic visualization of the airway can confirm the endotracheal tube occluding all the airway even with the cuff deflated and without evidence of swelling of the vocal cords. The rapid shallow breathing index is a ratio of respiratory rate over tidal volume in liters and is used to predict successful extubation. Values < 105 have a high sensitivity for successful extubation.
The patient remained under observation for 24 hours in the MICU and then was transferred to the general medicine ward. Unfortunately, 36 hours after, the patient had a new episode of angioedema requiring endotracheal intubation and placement on mechanical ventilation. This was his third episode of angioedema; he had a difficult airway classified as a Cormack-Lehane grade 3, requiring intubation with fiber-optic laryngoscope. In view of the recurrent events, a tracheostomy was done several days later. Figure 2 shows posttracheostomy X-ray with adequate position of the tracheostomy tube.
The patient was transferred to the Respiratory Care Unit and weaned off mechanical ventilation. He completed an intensive physical rehabilitation program and was discharged home. On discharge, he was followed by the Otorhinolaryngology Service and was decannulated about 5 months after. After tracheostomy decannulation, he developed asymptomatic stridor. A neck computer tomography scan revealed soft tissue thickening at the anterior and lateral aspects of the proximal tracheal likely representing granulation tissue/scarring. The findings were consistent with proximal tracheal stenosis sequelae of tracheostomy and intubation. In Figure 3, the upper portion of the curve represents the expiratory limb of the forced vital capacity and the lower portion represents inspiration. The flow-volume loop graph showed flattening of the inspiratory limb. There was a plateau in the inspiratory limb, suggestive of limitation of inspiratory flow as seen in variable extrathoracic lesions, such as glotticstricture, tumors, and vocal cord paralysis.7 The findings on the flow-volume loop were consistent with the subglottic stenosis identified by laryngoscopic examination. The patient was reluctant to undergo further interventions.
Discussion
The standard therapy for ACEI-inducedangioedema continues to be airway management and discontinuation of medication. However, life-threatening progression of symptoms have led to the use of off-label therapies, including FFP and bradykinin receptor antagonists, such as icatibant, which has been approved by the FDA for the treatment of hereditary angioedema. Icatibant is expensive and most hospitals do not have access to it. When considering the bradykinin pathway for therapy, FFP is commonly used. The cases described in the literature that have reported success with the use of FFP have used up to 2 units. There is no reported benefit of its use beyond 2 units. The initial randomized trials of icatibant for ACEI angioedema showed decreased time of resolution of angioedema.6 However, repeated trials showed conflicting results. At Veterans Affairs Caribbean Healthcare System, this medication was not available, and we decided to use FFP to improve the patient’s symptoms.
The administration of 2 units of FFP has been documented on case reports as a method to decrease the time of resolution of angioedema and the risk of recurrence. The mechanism of action thought to be involved includes the degradation of bradykinin by the enzyme ACE into inactive peptides and by supplying C1 inhibitor.8 No randomized clinical trial has investigated the use of FFP for the treatment of ACEI-induced angioedema. However, a retrospective cohort study report compared patients who presented with acute (nonhereditary) angioedema and airway compromise and received FFP with patients who were not treated with FFP.9 The study suggested a shorter ICU stay in the group treated with FFP, but the findings did not present statistical outcomes.
Nevertheless, our patient had recurrent ACEI-induced angioedema refractory to FFP. In addition to ACE or kininase II, FFP contains high-molecular weight-kininogen and kallikrein, the substrates that form bradykinin, which explained the mechanism of worsening angioedema.10 No randomized trials have investigated the use of FFP for the treatment of bradykinin-induced angioedema nor the appropriate dose.
Conclusion
In view of the emerging case reports of the effectiveness of FFP, this case of refractory angioedema raises concern for its true effectiveness and other possible factors involved in the mechanism of recurrence. Probably it would be unwise to conduct randomized studies in clinical situations such as the ones outlined. A collection of case series where FFP administration was done may be a more reasonable source of conclusions to be analyzed by a panel of experts.
1. Sánchez-Borges M, González-Aveledo LA. Angiotensin-converting enzyme inhibitors and angioedema. Allergy Asthma Immunol Res. 2010;2(3):195-198.
2. Kaplan AP. Angioedema. World Allergy Organ J. 2008;1(6):103-113.
3. Moellman JJ, Bernstein JA, Lindsell C, et al; American College of Allergy, Asthma & Immunology (ACAAI); Society for Academic Emergency Medicine (SAEM). A consensus parameter for the evaluation and management of angioedema in the emergency department. Acad Emerg Med. 2014;21(4):469-484.
4. LoVerde D, Files DC, Krishnaswamy G. Angioedema. Crit Care Med. 2017;45(4):725-735.
5. van den Elzen M, Go MFLC, Knulst AC, Blankestijn MA, van Os-Medendorp H, Otten HG. Efficacy of treatment of non-hereditary angioedema. Clinic Rev Allerg Immunol. 2018;54(3):412-431.
6. Bas M, Greve J, Stelter S, et al. A randomized trial of icatibant in ace-inhibitor–induced angioedema. N Engl J Med. 2015;372(5):418-425.
7. Diaz J, Casal J, Rodriguez W. Flow-volume loops: clinical correlation. PR Health Sci J. 2008;27(2):181-182.
8. Stewart M, McGlone R. Fresh frozen plasma in the treatment of ACE inhibitor-induced angioedema. BMJ Case Rep. 2012;2012:pii:bcr2012006849.
9. Saeb A, Hagglund KH, Cigolle CT. Using fresh frozen plasma for acute airway angioedema to prevent intubation in the emergency department: a retrospective cohort study. Emerg Med Int. 2016;2016:6091510.
10. Brown T, Gonzalez J, Monteleone C. Angiotensin-converting enzyme inhibitor-induced angioedema: a review of the literature. J Clin Hypertens (Greenwich). 2017;19(12):1377-1382.
1. Sánchez-Borges M, González-Aveledo LA. Angiotensin-converting enzyme inhibitors and angioedema. Allergy Asthma Immunol Res. 2010;2(3):195-198.
2. Kaplan AP. Angioedema. World Allergy Organ J. 2008;1(6):103-113.
3. Moellman JJ, Bernstein JA, Lindsell C, et al; American College of Allergy, Asthma & Immunology (ACAAI); Society for Academic Emergency Medicine (SAEM). A consensus parameter for the evaluation and management of angioedema in the emergency department. Acad Emerg Med. 2014;21(4):469-484.
4. LoVerde D, Files DC, Krishnaswamy G. Angioedema. Crit Care Med. 2017;45(4):725-735.
5. van den Elzen M, Go MFLC, Knulst AC, Blankestijn MA, van Os-Medendorp H, Otten HG. Efficacy of treatment of non-hereditary angioedema. Clinic Rev Allerg Immunol. 2018;54(3):412-431.
6. Bas M, Greve J, Stelter S, et al. A randomized trial of icatibant in ace-inhibitor–induced angioedema. N Engl J Med. 2015;372(5):418-425.
7. Diaz J, Casal J, Rodriguez W. Flow-volume loops: clinical correlation. PR Health Sci J. 2008;27(2):181-182.
8. Stewart M, McGlone R. Fresh frozen plasma in the treatment of ACE inhibitor-induced angioedema. BMJ Case Rep. 2012;2012:pii:bcr2012006849.
9. Saeb A, Hagglund KH, Cigolle CT. Using fresh frozen plasma for acute airway angioedema to prevent intubation in the emergency department: a retrospective cohort study. Emerg Med Int. 2016;2016:6091510.
10. Brown T, Gonzalez J, Monteleone C. Angiotensin-converting enzyme inhibitor-induced angioedema: a review of the literature. J Clin Hypertens (Greenwich). 2017;19(12):1377-1382.
New opioid recommendations: Pain from most dermatologic procedures should be managed with acetaminophen, ibuprofen
has recommended.
Rotation flaps, interpolation flaps, wedge resections, cartilage alar-batten grafts, and Mustarde flaps were among the 20 procedures that can be managed with up to 10 oral oxycodone 5-mg equivalents, according to the panel. Only the Abbe procedure might warrant dispensing up to 15 oxycodone 5-mg pills, Justin McLawhorn, MD, and colleagues wrote in the Journal of the American Academy of Dermatology. The recommended amount of opioids are in addition to nonopioid analgesics, the guidelines point out.
All the other procedures can – and should – be managed with a combination of acetaminophen and ibuprofen, either alone or in an alternating dose pattern, said Dr. McLawhorn, of the department of dermatology at the University of Oklahoma Health Sciences Center, Oklahoma City, and coauthors.
But limited opioid prescribing is an important part of healing for patients who undergo the most invasive procedures, they wrote. “The management of complications, including adequate pain control, should be tailored to each patient on a case-by-case basis. Moreover, any pain management plan should not strictly adhere to any single guideline, but rather should be formed with consideration of the expected pain from the procedure and/or closure and consider the patient’s expectations for pain control.”
The time is ripe for dermatologists to make a stand in combating the opioid crisis, according to a group email response to questions from Dr. McLawhorn, Thomas Stasko, MD, professor and chair of dermatology at the University of Oklahoma, Oklahoma City, and Lindsey Collins, MD, also of the University of Oklahoma.
“The opioid crisis has reached epidemic proportions. More than 70,000 Americans have died from an opioid overdose in 2017,” they wrote. “Moreover, recent data suggest that nearly 6% of postsurgical, opioid-naive patients become long-term users of opioids. The lack of specific evidence-based recommendations likely contributes to a wide variety in prescribing patterns and a steady supply of unused opioids. Countering the opioid crisis necessitates a restructuring of the opioid prescribing practices that addresses pain in a procedure-specific manner. These recommendations are one tool in the dermatologists’ arsenal that can be used as a reference to help guide opioid management and prevent excessive opioid prescriptions at discharge following dermatologic interventions.”
Unfortunately, they added, dermatologists have inadvertently fueled the opioid abuse fire.
“It is difficult to quantify which providers are responsible for the onslaught of opioids into our communities,” the authors wrote in the email interview. “However, we can deduce, based on recent opioid prescribing patterns, that dermatologists provide approximately 500,000 unused opioid pills to their communities on an annual basis. This is the result of a wide variation in practice patterns and narratives that have been previously circulated in an attempt to mitigate the providers’ perception of the addictive nature of opioid analgesics. Our hope is that by addressing pain in a procedure-specific manner, we can help to limit the excessive number of unused opioid pills that are provided by dermatologists and ultimately decrease the rate of opioid-related complications, including addiction and death.”
Still, patients need and deserve effective pain management after a procedure. In the guidelines, the investigators wrote that a “one-size-fits-all” approach “does not account for the mechanism of pain, the invasiveness of the procedure, or the anatomic structures that are manipulated. As a result, current guidelines cannot accurately predict the quantity of opioids that are necessary to manage postoperative pain.”
The panel brought together experts in general dermatology, dermatologic surgery, cosmetics, and phlebology to develop a consensus on opioid prescribing guidelines for 87 of the most common procedures. Everyone on the panel was a member of the American College of Mohs Surgery, American Academy of Dermatology, or the American Vein and Lymphatic Society. The panel conducted a literature review to determine which procedures might require opioids and which would not. At least 75% of the panel had to agree on a reasonable but effective opioid amount; they were then polled as to whether they might employ that recommendation in their own clinical practice.
The recommendations are aimed at patients who experienced no peri- or postoperative complications.
The panel agreed that acetaminophen and ibuprofen – alone, in combination, or with opioids – were reasonable choices for all the 87 procedures. In such instances, acetaminophen 1 g can be staggered with ibuprofen 400 mg every 4 or 8 hours.
“I think providers will encounter a mixed bag of preconceived notions regarding patients’ expectations for pain control,” Dr. McLawhorn and coauthors wrote in the interview. “The important point for providers to make is to emphasize the noninferiority of acetaminophen and/or ibuprofen in controlling acute pain for patients who are not dependent on opioids for the management of chronic pain. Our experience in caring for many surgical patients has shown that patients are usually receptive to the use of nonopioid analgesics as many are familiar with their addictive potential because of the uptick in the publicity of the opioid-related complications.”
In cases where opioids might be appropriate, the panel unanimously agreed that dose limits be imposed. For 15 of the 87 procedures, the panel recommend a maximum prescription of 10 oxycodone 5-mg equivalents. Only one other – the Abbe flap – might warrant more, with a maximum of 15 oxycodone 5-mg pills at discharge.
Sometimes called a “lip switch,” the Abbe flap is reconstruction for full-thickness lip defects. It is a composite flap that moves skin, muscle, mucosa, and blood supply from the lower lip to reconstruct a defect of the upper lip. This reconstruction attempts to respect the native anatomic landmarks of the lip and allow for a better functional outcome.
“Because of the extensive nature of the repair and the anatomic territories that are manipulated, including the suturing of the lower lip to the upper lip with delayed separation, adequate pain control may require opioid analgesics in the immediate postoperative period,” the team wrote in the interview.
The panel could not agree on pain management strategies for five other procedures: Karapandzic flaps, en bloc nail excisions, facial resurfacing with deep chemical peels, and small- or large-volume liposuction. This was partly because of a lack of personal experience. Only 8 of the 40 panelists performed Karapandzic flaps. The maximum number of 5-mg oxycodone tablets any panelist prescribed for Karapandzic flaps and en bloc nail excisions was 20.
Facial resurfacing was likewise an uncommon procedure for the panel, with just 11 members performing this using deep chemical peels. However, five of those panelists said that opioids were routinely needed for postoperative pain with a maximum of 15 oxycodone 5-mg equivalents. And just four panelists performed liposuction, for which they used a maximum of 15 oxycodone 5-mg equivalents.
“However,” they wrote in the guidelines, “these providers noted that the location where the procedure is performed strongly influences the need for opioid pain management, with small-volume removal in the neck, arms, or flanks being unlikely to require opioids for adequate pain control, whereas large-volume removal in the thighs, knees, and hips may routinely require opioids.”
Addressing patient expectations is a very important part of pain management, the panel noted. “Patients will invariably experience postoperative pain after cutaneous surgeries or other interventions, often peaking within 4 hours after surgery. Wound tension, size and type of repair, anatomical location/nerve innervation, and patient pain tolerance are all factors that contribute to postoperative discomfort and should be considered when developing a postoperative pain management plan.”
Ultimately, according to Dr. McLawhorn and coauthors, the decision to use opioids at discharge for postoperative pain control should be an individual one based on patients’ comorbidities and expectations.
“Admittedly, many of the procedures listed within the recommendations may result in a rather large or complex defect that requires an equally large or complex repair,” they wrote in the interview. “However, proper education of the patient and provider regarding the risks of addiction with the use of opioids even short term should be discussed as part of every preoperative consultation. Furthermore, the patient and the provider must discuss their expectations for postoperative pain interventions for adequate pain control.”
SOURCE: McLawhorn J et al. J Am Acad Dermatol. 2019 Nov 12. doi: 10.1016/j.jaad.2019.09.080.
has recommended.
Rotation flaps, interpolation flaps, wedge resections, cartilage alar-batten grafts, and Mustarde flaps were among the 20 procedures that can be managed with up to 10 oral oxycodone 5-mg equivalents, according to the panel. Only the Abbe procedure might warrant dispensing up to 15 oxycodone 5-mg pills, Justin McLawhorn, MD, and colleagues wrote in the Journal of the American Academy of Dermatology. The recommended amount of opioids are in addition to nonopioid analgesics, the guidelines point out.
All the other procedures can – and should – be managed with a combination of acetaminophen and ibuprofen, either alone or in an alternating dose pattern, said Dr. McLawhorn, of the department of dermatology at the University of Oklahoma Health Sciences Center, Oklahoma City, and coauthors.
But limited opioid prescribing is an important part of healing for patients who undergo the most invasive procedures, they wrote. “The management of complications, including adequate pain control, should be tailored to each patient on a case-by-case basis. Moreover, any pain management plan should not strictly adhere to any single guideline, but rather should be formed with consideration of the expected pain from the procedure and/or closure and consider the patient’s expectations for pain control.”
The time is ripe for dermatologists to make a stand in combating the opioid crisis, according to a group email response to questions from Dr. McLawhorn, Thomas Stasko, MD, professor and chair of dermatology at the University of Oklahoma, Oklahoma City, and Lindsey Collins, MD, also of the University of Oklahoma.
“The opioid crisis has reached epidemic proportions. More than 70,000 Americans have died from an opioid overdose in 2017,” they wrote. “Moreover, recent data suggest that nearly 6% of postsurgical, opioid-naive patients become long-term users of opioids. The lack of specific evidence-based recommendations likely contributes to a wide variety in prescribing patterns and a steady supply of unused opioids. Countering the opioid crisis necessitates a restructuring of the opioid prescribing practices that addresses pain in a procedure-specific manner. These recommendations are one tool in the dermatologists’ arsenal that can be used as a reference to help guide opioid management and prevent excessive opioid prescriptions at discharge following dermatologic interventions.”
Unfortunately, they added, dermatologists have inadvertently fueled the opioid abuse fire.
“It is difficult to quantify which providers are responsible for the onslaught of opioids into our communities,” the authors wrote in the email interview. “However, we can deduce, based on recent opioid prescribing patterns, that dermatologists provide approximately 500,000 unused opioid pills to their communities on an annual basis. This is the result of a wide variation in practice patterns and narratives that have been previously circulated in an attempt to mitigate the providers’ perception of the addictive nature of opioid analgesics. Our hope is that by addressing pain in a procedure-specific manner, we can help to limit the excessive number of unused opioid pills that are provided by dermatologists and ultimately decrease the rate of opioid-related complications, including addiction and death.”
Still, patients need and deserve effective pain management after a procedure. In the guidelines, the investigators wrote that a “one-size-fits-all” approach “does not account for the mechanism of pain, the invasiveness of the procedure, or the anatomic structures that are manipulated. As a result, current guidelines cannot accurately predict the quantity of opioids that are necessary to manage postoperative pain.”
The panel brought together experts in general dermatology, dermatologic surgery, cosmetics, and phlebology to develop a consensus on opioid prescribing guidelines for 87 of the most common procedures. Everyone on the panel was a member of the American College of Mohs Surgery, American Academy of Dermatology, or the American Vein and Lymphatic Society. The panel conducted a literature review to determine which procedures might require opioids and which would not. At least 75% of the panel had to agree on a reasonable but effective opioid amount; they were then polled as to whether they might employ that recommendation in their own clinical practice.
The recommendations are aimed at patients who experienced no peri- or postoperative complications.
The panel agreed that acetaminophen and ibuprofen – alone, in combination, or with opioids – were reasonable choices for all the 87 procedures. In such instances, acetaminophen 1 g can be staggered with ibuprofen 400 mg every 4 or 8 hours.
“I think providers will encounter a mixed bag of preconceived notions regarding patients’ expectations for pain control,” Dr. McLawhorn and coauthors wrote in the interview. “The important point for providers to make is to emphasize the noninferiority of acetaminophen and/or ibuprofen in controlling acute pain for patients who are not dependent on opioids for the management of chronic pain. Our experience in caring for many surgical patients has shown that patients are usually receptive to the use of nonopioid analgesics as many are familiar with their addictive potential because of the uptick in the publicity of the opioid-related complications.”
In cases where opioids might be appropriate, the panel unanimously agreed that dose limits be imposed. For 15 of the 87 procedures, the panel recommend a maximum prescription of 10 oxycodone 5-mg equivalents. Only one other – the Abbe flap – might warrant more, with a maximum of 15 oxycodone 5-mg pills at discharge.
Sometimes called a “lip switch,” the Abbe flap is reconstruction for full-thickness lip defects. It is a composite flap that moves skin, muscle, mucosa, and blood supply from the lower lip to reconstruct a defect of the upper lip. This reconstruction attempts to respect the native anatomic landmarks of the lip and allow for a better functional outcome.
“Because of the extensive nature of the repair and the anatomic territories that are manipulated, including the suturing of the lower lip to the upper lip with delayed separation, adequate pain control may require opioid analgesics in the immediate postoperative period,” the team wrote in the interview.
The panel could not agree on pain management strategies for five other procedures: Karapandzic flaps, en bloc nail excisions, facial resurfacing with deep chemical peels, and small- or large-volume liposuction. This was partly because of a lack of personal experience. Only 8 of the 40 panelists performed Karapandzic flaps. The maximum number of 5-mg oxycodone tablets any panelist prescribed for Karapandzic flaps and en bloc nail excisions was 20.
Facial resurfacing was likewise an uncommon procedure for the panel, with just 11 members performing this using deep chemical peels. However, five of those panelists said that opioids were routinely needed for postoperative pain with a maximum of 15 oxycodone 5-mg equivalents. And just four panelists performed liposuction, for which they used a maximum of 15 oxycodone 5-mg equivalents.
“However,” they wrote in the guidelines, “these providers noted that the location where the procedure is performed strongly influences the need for opioid pain management, with small-volume removal in the neck, arms, or flanks being unlikely to require opioids for adequate pain control, whereas large-volume removal in the thighs, knees, and hips may routinely require opioids.”
Addressing patient expectations is a very important part of pain management, the panel noted. “Patients will invariably experience postoperative pain after cutaneous surgeries or other interventions, often peaking within 4 hours after surgery. Wound tension, size and type of repair, anatomical location/nerve innervation, and patient pain tolerance are all factors that contribute to postoperative discomfort and should be considered when developing a postoperative pain management plan.”
Ultimately, according to Dr. McLawhorn and coauthors, the decision to use opioids at discharge for postoperative pain control should be an individual one based on patients’ comorbidities and expectations.
“Admittedly, many of the procedures listed within the recommendations may result in a rather large or complex defect that requires an equally large or complex repair,” they wrote in the interview. “However, proper education of the patient and provider regarding the risks of addiction with the use of opioids even short term should be discussed as part of every preoperative consultation. Furthermore, the patient and the provider must discuss their expectations for postoperative pain interventions for adequate pain control.”
SOURCE: McLawhorn J et al. J Am Acad Dermatol. 2019 Nov 12. doi: 10.1016/j.jaad.2019.09.080.
has recommended.
Rotation flaps, interpolation flaps, wedge resections, cartilage alar-batten grafts, and Mustarde flaps were among the 20 procedures that can be managed with up to 10 oral oxycodone 5-mg equivalents, according to the panel. Only the Abbe procedure might warrant dispensing up to 15 oxycodone 5-mg pills, Justin McLawhorn, MD, and colleagues wrote in the Journal of the American Academy of Dermatology. The recommended amount of opioids are in addition to nonopioid analgesics, the guidelines point out.
All the other procedures can – and should – be managed with a combination of acetaminophen and ibuprofen, either alone or in an alternating dose pattern, said Dr. McLawhorn, of the department of dermatology at the University of Oklahoma Health Sciences Center, Oklahoma City, and coauthors.
But limited opioid prescribing is an important part of healing for patients who undergo the most invasive procedures, they wrote. “The management of complications, including adequate pain control, should be tailored to each patient on a case-by-case basis. Moreover, any pain management plan should not strictly adhere to any single guideline, but rather should be formed with consideration of the expected pain from the procedure and/or closure and consider the patient’s expectations for pain control.”
The time is ripe for dermatologists to make a stand in combating the opioid crisis, according to a group email response to questions from Dr. McLawhorn, Thomas Stasko, MD, professor and chair of dermatology at the University of Oklahoma, Oklahoma City, and Lindsey Collins, MD, also of the University of Oklahoma.
“The opioid crisis has reached epidemic proportions. More than 70,000 Americans have died from an opioid overdose in 2017,” they wrote. “Moreover, recent data suggest that nearly 6% of postsurgical, opioid-naive patients become long-term users of opioids. The lack of specific evidence-based recommendations likely contributes to a wide variety in prescribing patterns and a steady supply of unused opioids. Countering the opioid crisis necessitates a restructuring of the opioid prescribing practices that addresses pain in a procedure-specific manner. These recommendations are one tool in the dermatologists’ arsenal that can be used as a reference to help guide opioid management and prevent excessive opioid prescriptions at discharge following dermatologic interventions.”
Unfortunately, they added, dermatologists have inadvertently fueled the opioid abuse fire.
“It is difficult to quantify which providers are responsible for the onslaught of opioids into our communities,” the authors wrote in the email interview. “However, we can deduce, based on recent opioid prescribing patterns, that dermatologists provide approximately 500,000 unused opioid pills to their communities on an annual basis. This is the result of a wide variation in practice patterns and narratives that have been previously circulated in an attempt to mitigate the providers’ perception of the addictive nature of opioid analgesics. Our hope is that by addressing pain in a procedure-specific manner, we can help to limit the excessive number of unused opioid pills that are provided by dermatologists and ultimately decrease the rate of opioid-related complications, including addiction and death.”
Still, patients need and deserve effective pain management after a procedure. In the guidelines, the investigators wrote that a “one-size-fits-all” approach “does not account for the mechanism of pain, the invasiveness of the procedure, or the anatomic structures that are manipulated. As a result, current guidelines cannot accurately predict the quantity of opioids that are necessary to manage postoperative pain.”
The panel brought together experts in general dermatology, dermatologic surgery, cosmetics, and phlebology to develop a consensus on opioid prescribing guidelines for 87 of the most common procedures. Everyone on the panel was a member of the American College of Mohs Surgery, American Academy of Dermatology, or the American Vein and Lymphatic Society. The panel conducted a literature review to determine which procedures might require opioids and which would not. At least 75% of the panel had to agree on a reasonable but effective opioid amount; they were then polled as to whether they might employ that recommendation in their own clinical practice.
The recommendations are aimed at patients who experienced no peri- or postoperative complications.
The panel agreed that acetaminophen and ibuprofen – alone, in combination, or with opioids – were reasonable choices for all the 87 procedures. In such instances, acetaminophen 1 g can be staggered with ibuprofen 400 mg every 4 or 8 hours.
“I think providers will encounter a mixed bag of preconceived notions regarding patients’ expectations for pain control,” Dr. McLawhorn and coauthors wrote in the interview. “The important point for providers to make is to emphasize the noninferiority of acetaminophen and/or ibuprofen in controlling acute pain for patients who are not dependent on opioids for the management of chronic pain. Our experience in caring for many surgical patients has shown that patients are usually receptive to the use of nonopioid analgesics as many are familiar with their addictive potential because of the uptick in the publicity of the opioid-related complications.”
In cases where opioids might be appropriate, the panel unanimously agreed that dose limits be imposed. For 15 of the 87 procedures, the panel recommend a maximum prescription of 10 oxycodone 5-mg equivalents. Only one other – the Abbe flap – might warrant more, with a maximum of 15 oxycodone 5-mg pills at discharge.
Sometimes called a “lip switch,” the Abbe flap is reconstruction for full-thickness lip defects. It is a composite flap that moves skin, muscle, mucosa, and blood supply from the lower lip to reconstruct a defect of the upper lip. This reconstruction attempts to respect the native anatomic landmarks of the lip and allow for a better functional outcome.
“Because of the extensive nature of the repair and the anatomic territories that are manipulated, including the suturing of the lower lip to the upper lip with delayed separation, adequate pain control may require opioid analgesics in the immediate postoperative period,” the team wrote in the interview.
The panel could not agree on pain management strategies for five other procedures: Karapandzic flaps, en bloc nail excisions, facial resurfacing with deep chemical peels, and small- or large-volume liposuction. This was partly because of a lack of personal experience. Only 8 of the 40 panelists performed Karapandzic flaps. The maximum number of 5-mg oxycodone tablets any panelist prescribed for Karapandzic flaps and en bloc nail excisions was 20.
Facial resurfacing was likewise an uncommon procedure for the panel, with just 11 members performing this using deep chemical peels. However, five of those panelists said that opioids were routinely needed for postoperative pain with a maximum of 15 oxycodone 5-mg equivalents. And just four panelists performed liposuction, for which they used a maximum of 15 oxycodone 5-mg equivalents.
“However,” they wrote in the guidelines, “these providers noted that the location where the procedure is performed strongly influences the need for opioid pain management, with small-volume removal in the neck, arms, or flanks being unlikely to require opioids for adequate pain control, whereas large-volume removal in the thighs, knees, and hips may routinely require opioids.”
Addressing patient expectations is a very important part of pain management, the panel noted. “Patients will invariably experience postoperative pain after cutaneous surgeries or other interventions, often peaking within 4 hours after surgery. Wound tension, size and type of repair, anatomical location/nerve innervation, and patient pain tolerance are all factors that contribute to postoperative discomfort and should be considered when developing a postoperative pain management plan.”
Ultimately, according to Dr. McLawhorn and coauthors, the decision to use opioids at discharge for postoperative pain control should be an individual one based on patients’ comorbidities and expectations.
“Admittedly, many of the procedures listed within the recommendations may result in a rather large or complex defect that requires an equally large or complex repair,” they wrote in the interview. “However, proper education of the patient and provider regarding the risks of addiction with the use of opioids even short term should be discussed as part of every preoperative consultation. Furthermore, the patient and the provider must discuss their expectations for postoperative pain interventions for adequate pain control.”
SOURCE: McLawhorn J et al. J Am Acad Dermatol. 2019 Nov 12. doi: 10.1016/j.jaad.2019.09.080.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Mole on back
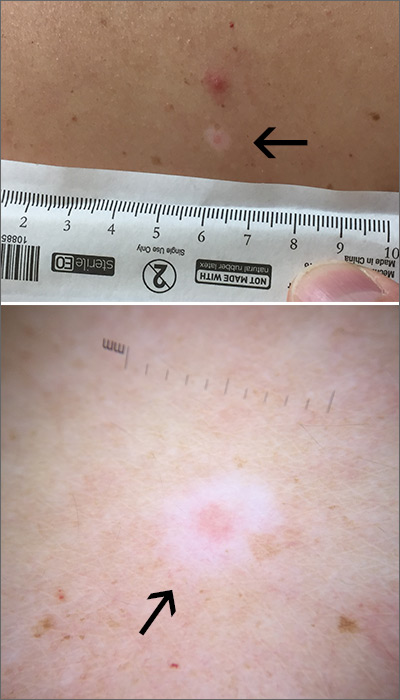
The FP used a dermatoscope to get a better look at the lesion and recognized this as a halo nevus, which is characterized by a central nevus with a halo of depigmentation around it (arrow) and sometimes depigmentation within the nevus itself.
The halo is caused when, occasionally, the body develops an immune reaction to a nevus and its melanocytes. As cytotoxic T cells target those melanocytes, there is loss of pigment to the tissue surrounding the nevus.1
The appearance of the central nevus, rather than the hypopigmentation, determines management strategies. A globular or homogeneous pattern seen on dermoscopy is typically indicative of a benign lesion.2 An atypical pigment network or other melanoma specific structures should raise your suspicions for melanoma or atypical nevus and prompt a deep shave excision sent for pathology to rule out melanoma. A nevus without suspicious features, other than the surrounding hypopigmentation, can be managed conservatively with self-monitoring and re-evaluation.
In this case, the lesion displayed a homogeneous pattern, and the FP advised the patient to self-monitor.
Images and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque, NM.
1. Bayer-Garner IB, Ivan D, Schwartz MR, et al. The immunopathology of regression in benign lichenoid keratosis, keratoacanthoma and halo nevus. Clin Med Res. 2004;2:89-97.
2. Porto AC, Blumetti TP, de Paula Ramos Castro R, et al. Recurrent halo nevus: dermoscopy and confocal microscopy features. JAAD Case Rep. 2017;3:256-258.

The FP used a dermatoscope to get a better look at the lesion and recognized this as a halo nevus, which is characterized by a central nevus with a halo of depigmentation around it (arrow) and sometimes depigmentation within the nevus itself.
The halo is caused when, occasionally, the body develops an immune reaction to a nevus and its melanocytes. As cytotoxic T cells target those melanocytes, there is loss of pigment to the tissue surrounding the nevus.1
The appearance of the central nevus, rather than the hypopigmentation, determines management strategies. A globular or homogeneous pattern seen on dermoscopy is typically indicative of a benign lesion.2 An atypical pigment network or other melanoma specific structures should raise your suspicions for melanoma or atypical nevus and prompt a deep shave excision sent for pathology to rule out melanoma. A nevus without suspicious features, other than the surrounding hypopigmentation, can be managed conservatively with self-monitoring and re-evaluation.
In this case, the lesion displayed a homogeneous pattern, and the FP advised the patient to self-monitor.
Images and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque, NM.

The FP used a dermatoscope to get a better look at the lesion and recognized this as a halo nevus, which is characterized by a central nevus with a halo of depigmentation around it (arrow) and sometimes depigmentation within the nevus itself.
The halo is caused when, occasionally, the body develops an immune reaction to a nevus and its melanocytes. As cytotoxic T cells target those melanocytes, there is loss of pigment to the tissue surrounding the nevus.1
The appearance of the central nevus, rather than the hypopigmentation, determines management strategies. A globular or homogeneous pattern seen on dermoscopy is typically indicative of a benign lesion.2 An atypical pigment network or other melanoma specific structures should raise your suspicions for melanoma or atypical nevus and prompt a deep shave excision sent for pathology to rule out melanoma. A nevus without suspicious features, other than the surrounding hypopigmentation, can be managed conservatively with self-monitoring and re-evaluation.
In this case, the lesion displayed a homogeneous pattern, and the FP advised the patient to self-monitor.
Images and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque, NM.
1. Bayer-Garner IB, Ivan D, Schwartz MR, et al. The immunopathology of regression in benign lichenoid keratosis, keratoacanthoma and halo nevus. Clin Med Res. 2004;2:89-97.
2. Porto AC, Blumetti TP, de Paula Ramos Castro R, et al. Recurrent halo nevus: dermoscopy and confocal microscopy features. JAAD Case Rep. 2017;3:256-258.
1. Bayer-Garner IB, Ivan D, Schwartz MR, et al. The immunopathology of regression in benign lichenoid keratosis, keratoacanthoma and halo nevus. Clin Med Res. 2004;2:89-97.
2. Porto AC, Blumetti TP, de Paula Ramos Castro R, et al. Recurrent halo nevus: dermoscopy and confocal microscopy features. JAAD Case Rep. 2017;3:256-258.