User login
Adult atopic dermatitis brings increased osteoporosis risk
MADRID – – even if they’ve never taken systemic corticosteroids, according to a large observational Danish national registry study.
A key study finding was that these elevated risks were concentrated in the patients who used potent or superpotent topical corticosteroids. Adult AD patients who used mild- or moderate-potency topical steroids were not at significantly increased risk. Neither were patients on topical calcineurin inhibitors, Jacob P. Thyssen, MD, PhD, reported at a meeting of the European Task Force on Atopic Dermatitis held in conjunction with the annual congress of the European Academy of Dermatology and Venereology.
“The absolute risk is low, but it’s real,” commented Dr. Thyssen, professor of dermatology at the University of Copenhagen.
His advice to colleagues: “Dermatologists should consider alternative treatments in the chronic excessive users of topical corticosteroids, or use them in combination with prophylactic treatment to preserve bone homeostasis in such patients.”
He presented the results of a retrospective case-control study of 10,636 Danish adults with AD and 87,989 matched controls. At baseline in this study, which featured a maximum of 20 years of follow-up starting in 1997, participants had no history of osteoporosis.
Dr. Thyssen expressed the absolute risk of being diagnosed with osteoporosis in the study as follows: If 10,000 adult AD patients were followed for 1 year, on average 23.5 of them would be diagnosed with osteoporosis, a rate more than double the 10.3 per 10,000 in the general population. Moreover, on average, 42.6 out of 10,000 adult AD patients would incur a major osteoporotic fracture during a year of follow-up, compared with 32.3 individuals in the general population.
In the subgroup of patients who never used systemic corticosteroids, the risk of being diagnosed with osteoporosis was 12.8 per 10,000 per year, significantly higher than the 7.4 per 10,000 rate in the general population. Similarly, the 1-year rate of major osteoporotic fractures was 33.1 per 10,000 among the AD group and 29.6 in matched controls.
In a Cox regression analysis adjusted for age, sex, socioeconomic status, body mass index, asthma, and the use of a variety of medications thought to potentially have a negative effect upon bone metabolism, the risk of osteoporosis in the entire group of 10,636 adult AD patients was 51% greater than in matched controls, and their risk of major osteoporotic fractures was 18% greater. In the subgroup of AD patients who never used systemic steroids, the risks of osteoporosis and major osteoporotic fractures were 82% and 14% greater than in controls. The medications adjusted for in the regression analysis included proton pump inhibitors, thiazide diuretics, H2 receptor blockers, statins, cyclosporine, hormone therapy, contraceptives, and psychotropic medications.
Scoring Atopic Dermatitis (SCORAD) ratings were available on roughly 4,000 of the adult AD patients. In an analysis of this large subgroup, disease severity as reflected in SCORAD scores did not explain the increased osteoporosis and fracture risks. However, the use of potent or superpotent topical corticosteroids did. Patients who used potent topical steroids had a statistically significant 16% increased risk of being diagnosed with osteoporosis than nonusers, as well as a 7% increased risk of major osteoporotic fractures. Patients who applied superpotent topical steroids had 42% and 18% increased risks of those two adverse outcomes.
In contrast, neither the use of topical calcineurin inhibitors nor mild- or mid-potency topical steroids was associated with increased risk of bone events in a Cox regression analysis adjusted for potential confounders.
A relationship between the use of high-potency topical corticosteroids and adverse bone events is biologically plausible, according to Dr. Thyssen. He and his coinvestigators have previously documented a 100%-400% increased rate of chemical penetration through atopic skin, which is notoriously barrier damaged.
“We find it very likely that, if you put topical steroids on atopic skin in high amounts and for a very long time, you may have systemic effects,” he said.
A great many adult AD patients do exactly that. When Dr. Thyssen and coworkers analyzed Danish national prescription drug registry data for their patient cohort, they found that roughly one-third of the elderly subgroup had filled prescriptions totaling greater than 2 kg of mometasone or other similar-potency steroids over the previous 10 years.
“So we know that a significant proportion of our atopic dermatitis patients are really high users of topical corticosteroids,” the dermatologist noted.
Dr. Thyssen’s national osteoporosis and fracture study was funded with a government research grant. He reported serving as an advisor to and/or recipient of research grants from AbbVie, Pfizer, Leo Pharma, Eli Lilly, Regeneron, Sanofi Genzyme, and Union Therapeutics.
MADRID – – even if they’ve never taken systemic corticosteroids, according to a large observational Danish national registry study.
A key study finding was that these elevated risks were concentrated in the patients who used potent or superpotent topical corticosteroids. Adult AD patients who used mild- or moderate-potency topical steroids were not at significantly increased risk. Neither were patients on topical calcineurin inhibitors, Jacob P. Thyssen, MD, PhD, reported at a meeting of the European Task Force on Atopic Dermatitis held in conjunction with the annual congress of the European Academy of Dermatology and Venereology.
“The absolute risk is low, but it’s real,” commented Dr. Thyssen, professor of dermatology at the University of Copenhagen.
His advice to colleagues: “Dermatologists should consider alternative treatments in the chronic excessive users of topical corticosteroids, or use them in combination with prophylactic treatment to preserve bone homeostasis in such patients.”
He presented the results of a retrospective case-control study of 10,636 Danish adults with AD and 87,989 matched controls. At baseline in this study, which featured a maximum of 20 years of follow-up starting in 1997, participants had no history of osteoporosis.
Dr. Thyssen expressed the absolute risk of being diagnosed with osteoporosis in the study as follows: If 10,000 adult AD patients were followed for 1 year, on average 23.5 of them would be diagnosed with osteoporosis, a rate more than double the 10.3 per 10,000 in the general population. Moreover, on average, 42.6 out of 10,000 adult AD patients would incur a major osteoporotic fracture during a year of follow-up, compared with 32.3 individuals in the general population.
In the subgroup of patients who never used systemic corticosteroids, the risk of being diagnosed with osteoporosis was 12.8 per 10,000 per year, significantly higher than the 7.4 per 10,000 rate in the general population. Similarly, the 1-year rate of major osteoporotic fractures was 33.1 per 10,000 among the AD group and 29.6 in matched controls.
In a Cox regression analysis adjusted for age, sex, socioeconomic status, body mass index, asthma, and the use of a variety of medications thought to potentially have a negative effect upon bone metabolism, the risk of osteoporosis in the entire group of 10,636 adult AD patients was 51% greater than in matched controls, and their risk of major osteoporotic fractures was 18% greater. In the subgroup of AD patients who never used systemic steroids, the risks of osteoporosis and major osteoporotic fractures were 82% and 14% greater than in controls. The medications adjusted for in the regression analysis included proton pump inhibitors, thiazide diuretics, H2 receptor blockers, statins, cyclosporine, hormone therapy, contraceptives, and psychotropic medications.
Scoring Atopic Dermatitis (SCORAD) ratings were available on roughly 4,000 of the adult AD patients. In an analysis of this large subgroup, disease severity as reflected in SCORAD scores did not explain the increased osteoporosis and fracture risks. However, the use of potent or superpotent topical corticosteroids did. Patients who used potent topical steroids had a statistically significant 16% increased risk of being diagnosed with osteoporosis than nonusers, as well as a 7% increased risk of major osteoporotic fractures. Patients who applied superpotent topical steroids had 42% and 18% increased risks of those two adverse outcomes.
In contrast, neither the use of topical calcineurin inhibitors nor mild- or mid-potency topical steroids was associated with increased risk of bone events in a Cox regression analysis adjusted for potential confounders.
A relationship between the use of high-potency topical corticosteroids and adverse bone events is biologically plausible, according to Dr. Thyssen. He and his coinvestigators have previously documented a 100%-400% increased rate of chemical penetration through atopic skin, which is notoriously barrier damaged.
“We find it very likely that, if you put topical steroids on atopic skin in high amounts and for a very long time, you may have systemic effects,” he said.
A great many adult AD patients do exactly that. When Dr. Thyssen and coworkers analyzed Danish national prescription drug registry data for their patient cohort, they found that roughly one-third of the elderly subgroup had filled prescriptions totaling greater than 2 kg of mometasone or other similar-potency steroids over the previous 10 years.
“So we know that a significant proportion of our atopic dermatitis patients are really high users of topical corticosteroids,” the dermatologist noted.
Dr. Thyssen’s national osteoporosis and fracture study was funded with a government research grant. He reported serving as an advisor to and/or recipient of research grants from AbbVie, Pfizer, Leo Pharma, Eli Lilly, Regeneron, Sanofi Genzyme, and Union Therapeutics.
MADRID – – even if they’ve never taken systemic corticosteroids, according to a large observational Danish national registry study.
A key study finding was that these elevated risks were concentrated in the patients who used potent or superpotent topical corticosteroids. Adult AD patients who used mild- or moderate-potency topical steroids were not at significantly increased risk. Neither were patients on topical calcineurin inhibitors, Jacob P. Thyssen, MD, PhD, reported at a meeting of the European Task Force on Atopic Dermatitis held in conjunction with the annual congress of the European Academy of Dermatology and Venereology.
“The absolute risk is low, but it’s real,” commented Dr. Thyssen, professor of dermatology at the University of Copenhagen.
His advice to colleagues: “Dermatologists should consider alternative treatments in the chronic excessive users of topical corticosteroids, or use them in combination with prophylactic treatment to preserve bone homeostasis in such patients.”
He presented the results of a retrospective case-control study of 10,636 Danish adults with AD and 87,989 matched controls. At baseline in this study, which featured a maximum of 20 years of follow-up starting in 1997, participants had no history of osteoporosis.
Dr. Thyssen expressed the absolute risk of being diagnosed with osteoporosis in the study as follows: If 10,000 adult AD patients were followed for 1 year, on average 23.5 of them would be diagnosed with osteoporosis, a rate more than double the 10.3 per 10,000 in the general population. Moreover, on average, 42.6 out of 10,000 adult AD patients would incur a major osteoporotic fracture during a year of follow-up, compared with 32.3 individuals in the general population.
In the subgroup of patients who never used systemic corticosteroids, the risk of being diagnosed with osteoporosis was 12.8 per 10,000 per year, significantly higher than the 7.4 per 10,000 rate in the general population. Similarly, the 1-year rate of major osteoporotic fractures was 33.1 per 10,000 among the AD group and 29.6 in matched controls.
In a Cox regression analysis adjusted for age, sex, socioeconomic status, body mass index, asthma, and the use of a variety of medications thought to potentially have a negative effect upon bone metabolism, the risk of osteoporosis in the entire group of 10,636 adult AD patients was 51% greater than in matched controls, and their risk of major osteoporotic fractures was 18% greater. In the subgroup of AD patients who never used systemic steroids, the risks of osteoporosis and major osteoporotic fractures were 82% and 14% greater than in controls. The medications adjusted for in the regression analysis included proton pump inhibitors, thiazide diuretics, H2 receptor blockers, statins, cyclosporine, hormone therapy, contraceptives, and psychotropic medications.
Scoring Atopic Dermatitis (SCORAD) ratings were available on roughly 4,000 of the adult AD patients. In an analysis of this large subgroup, disease severity as reflected in SCORAD scores did not explain the increased osteoporosis and fracture risks. However, the use of potent or superpotent topical corticosteroids did. Patients who used potent topical steroids had a statistically significant 16% increased risk of being diagnosed with osteoporosis than nonusers, as well as a 7% increased risk of major osteoporotic fractures. Patients who applied superpotent topical steroids had 42% and 18% increased risks of those two adverse outcomes.
In contrast, neither the use of topical calcineurin inhibitors nor mild- or mid-potency topical steroids was associated with increased risk of bone events in a Cox regression analysis adjusted for potential confounders.
A relationship between the use of high-potency topical corticosteroids and adverse bone events is biologically plausible, according to Dr. Thyssen. He and his coinvestigators have previously documented a 100%-400% increased rate of chemical penetration through atopic skin, which is notoriously barrier damaged.
“We find it very likely that, if you put topical steroids on atopic skin in high amounts and for a very long time, you may have systemic effects,” he said.
A great many adult AD patients do exactly that. When Dr. Thyssen and coworkers analyzed Danish national prescription drug registry data for their patient cohort, they found that roughly one-third of the elderly subgroup had filled prescriptions totaling greater than 2 kg of mometasone or other similar-potency steroids over the previous 10 years.
“So we know that a significant proportion of our atopic dermatitis patients are really high users of topical corticosteroids,” the dermatologist noted.
Dr. Thyssen’s national osteoporosis and fracture study was funded with a government research grant. He reported serving as an advisor to and/or recipient of research grants from AbbVie, Pfizer, Leo Pharma, Eli Lilly, Regeneron, Sanofi Genzyme, and Union Therapeutics.
REPORTING FROM EADV 2019
Which children are at greatest risk for atopic dermatitis?
MADRID – A parental history of asthma or allergic rhinitis significantly increases the risk that a child will develop atopic dermatitis, and that risk doubles if a parent has a history of atopic dermatitis rather than another atopic disease, Nina H. Ravn reported at a meeting of the European Task Force on Atopic Dermatitis held in conjunction with the annual congress of the European Academy of Dermatology and Venereology.
She presented a comprehensive meta-analysis of 149 published studies addressing the risk of developing atopic dermatitis according to parental history of atopic disease. The studies included more than 656,000 participants. The picture that emerged from the meta-analysis was one of a stepwise increase in the risk of pediatric atopic dermatitis according to the type and number of parental atopic diseases present.
“This is something that hopefully can be useful when you talk with parents or parents-to-be with atopic diseases and they want to know how their disease might affect their child,” explained Ms. Ravn of the University of Copenhagen.
It’s also information that clinicians will find helpful in appropriately targeting primary prevention interventions if and when methods of proven efficacy become available. That’s a likely prospect, as this is now an extremely active field of research, she noted.
The meta-analysis showed that a parental history of atopic dermatitis was associated with a 3.3-fold greater risk of atopic dermatitis in the offspring than in families without a parental history of atopy. A parental history of asthma was associated with a 1.56-fold increased risk, while allergic rhinitis in a parent was linked to a 1.68-fold increased risk.
“It does matter what type of atopic disease the parents have,” she observed. “Those with a parental history of asthma or allergic rhinitis can be considered as being at more of an intermediate risk level, while those with a parental history of atopic dermatitis are a particularly high risk group.”
Of note, the risk of pediatric atopic dermatitis was the same regardless of whether the father or mother was the one with a history of atopic disease. If one parent had a history of an atopic disease, the pediatric risk was increased 1.3-fold compared to when the parental history was negative. If both parents had a history of atopic illness, the risk jumped to 2.08-fold. And if one parent had a history of more than one form of atopic disease, the pediatric risk of atopic dermatitis was increased 2.32-fold.
“An interesting result that was new to me what that fathers’ and mothers’ contribution to risk is equal,” said session cochair Andreas Wollenberg, MD, professor of dermatology at Ludwig Maximilian University of Munich. “For the past 2 decades we were always taught that the mother would have a greater impact on that risk.”
“I was also surprised by our findings,” Ms. Ravn replied. “But when we pooled all the data there really was no difference, nor in any of our subanalyses.”
She reported having no financial conflicts regarding her study.
SOURCE: Ravn NH. THE EADV CONGRESS.
MADRID – A parental history of asthma or allergic rhinitis significantly increases the risk that a child will develop atopic dermatitis, and that risk doubles if a parent has a history of atopic dermatitis rather than another atopic disease, Nina H. Ravn reported at a meeting of the European Task Force on Atopic Dermatitis held in conjunction with the annual congress of the European Academy of Dermatology and Venereology.
She presented a comprehensive meta-analysis of 149 published studies addressing the risk of developing atopic dermatitis according to parental history of atopic disease. The studies included more than 656,000 participants. The picture that emerged from the meta-analysis was one of a stepwise increase in the risk of pediatric atopic dermatitis according to the type and number of parental atopic diseases present.
“This is something that hopefully can be useful when you talk with parents or parents-to-be with atopic diseases and they want to know how their disease might affect their child,” explained Ms. Ravn of the University of Copenhagen.
It’s also information that clinicians will find helpful in appropriately targeting primary prevention interventions if and when methods of proven efficacy become available. That’s a likely prospect, as this is now an extremely active field of research, she noted.
The meta-analysis showed that a parental history of atopic dermatitis was associated with a 3.3-fold greater risk of atopic dermatitis in the offspring than in families without a parental history of atopy. A parental history of asthma was associated with a 1.56-fold increased risk, while allergic rhinitis in a parent was linked to a 1.68-fold increased risk.
“It does matter what type of atopic disease the parents have,” she observed. “Those with a parental history of asthma or allergic rhinitis can be considered as being at more of an intermediate risk level, while those with a parental history of atopic dermatitis are a particularly high risk group.”
Of note, the risk of pediatric atopic dermatitis was the same regardless of whether the father or mother was the one with a history of atopic disease. If one parent had a history of an atopic disease, the pediatric risk was increased 1.3-fold compared to when the parental history was negative. If both parents had a history of atopic illness, the risk jumped to 2.08-fold. And if one parent had a history of more than one form of atopic disease, the pediatric risk of atopic dermatitis was increased 2.32-fold.
“An interesting result that was new to me what that fathers’ and mothers’ contribution to risk is equal,” said session cochair Andreas Wollenberg, MD, professor of dermatology at Ludwig Maximilian University of Munich. “For the past 2 decades we were always taught that the mother would have a greater impact on that risk.”
“I was also surprised by our findings,” Ms. Ravn replied. “But when we pooled all the data there really was no difference, nor in any of our subanalyses.”
She reported having no financial conflicts regarding her study.
SOURCE: Ravn NH. THE EADV CONGRESS.
MADRID – A parental history of asthma or allergic rhinitis significantly increases the risk that a child will develop atopic dermatitis, and that risk doubles if a parent has a history of atopic dermatitis rather than another atopic disease, Nina H. Ravn reported at a meeting of the European Task Force on Atopic Dermatitis held in conjunction with the annual congress of the European Academy of Dermatology and Venereology.
She presented a comprehensive meta-analysis of 149 published studies addressing the risk of developing atopic dermatitis according to parental history of atopic disease. The studies included more than 656,000 participants. The picture that emerged from the meta-analysis was one of a stepwise increase in the risk of pediatric atopic dermatitis according to the type and number of parental atopic diseases present.
“This is something that hopefully can be useful when you talk with parents or parents-to-be with atopic diseases and they want to know how their disease might affect their child,” explained Ms. Ravn of the University of Copenhagen.
It’s also information that clinicians will find helpful in appropriately targeting primary prevention interventions if and when methods of proven efficacy become available. That’s a likely prospect, as this is now an extremely active field of research, she noted.
The meta-analysis showed that a parental history of atopic dermatitis was associated with a 3.3-fold greater risk of atopic dermatitis in the offspring than in families without a parental history of atopy. A parental history of asthma was associated with a 1.56-fold increased risk, while allergic rhinitis in a parent was linked to a 1.68-fold increased risk.
“It does matter what type of atopic disease the parents have,” she observed. “Those with a parental history of asthma or allergic rhinitis can be considered as being at more of an intermediate risk level, while those with a parental history of atopic dermatitis are a particularly high risk group.”
Of note, the risk of pediatric atopic dermatitis was the same regardless of whether the father or mother was the one with a history of atopic disease. If one parent had a history of an atopic disease, the pediatric risk was increased 1.3-fold compared to when the parental history was negative. If both parents had a history of atopic illness, the risk jumped to 2.08-fold. And if one parent had a history of more than one form of atopic disease, the pediatric risk of atopic dermatitis was increased 2.32-fold.
“An interesting result that was new to me what that fathers’ and mothers’ contribution to risk is equal,” said session cochair Andreas Wollenberg, MD, professor of dermatology at Ludwig Maximilian University of Munich. “For the past 2 decades we were always taught that the mother would have a greater impact on that risk.”
“I was also surprised by our findings,” Ms. Ravn replied. “But when we pooled all the data there really was no difference, nor in any of our subanalyses.”
She reported having no financial conflicts regarding her study.
SOURCE: Ravn NH. THE EADV CONGRESS.
REPORTING FROM The EADV CONGRESS
Key clinical point: Pediatric atopic dermatitis risk varies according to type of parental history of atopic disease.
Major finding: A parental history of atopic dermatitis is associated with a 3.3-fold increased risk of atopic dermatitis in the child, twice the risk associated with parental asthma or allergic rhinitis.
Study details: This was a systematic review and meta-analysis of 149 published studies with 656,711 participants.
Disclosures: The presenter reported having no financial conflicts regarding the study, conducted free of commercial support.
Source: Ravn NH. The EADV Congress.
Identifying bacterial infections in setting of atopic dermatitis
While of infections in AD and features common to both can make it difficult to make a clinical diagnosis of infections.
Addressing this issue, the International Eczema Council Skin Infection Group reviewed the most current evidence on the clinical features of bacterial infections and the interaction between host and bacterial factors that affect severity and morbidity in people with atopic dermatitis (AD). Recurrent skin infections, especially from Staphylococcus aureus and occasionally from beta-hemolytic streptococci, are more common in people with AD than those who do not have AD for a variety of reasons, Helen Alexander, MD, from the unit for population-based dermatology research at St. John’s Institute of Dermatology, Guy’s and St Thomas’ NHS Foundation Trust and King’s College London, and associates wrote in the review article published in the British Journal of Dermatology.
“The reduced skin barrier, cutaneous innate and adaptive immune abnormalities and trauma from scratching all contribute to the increased risk of skin infection,” they wrote. “However, the wide variability in clinical presentation of bacterial infection in AD and the inherent features of AD – cutaneous erythema and warmth, oozing associated with edema, and regional lymphadenopathy – overlap with those of infection, making clinical diagnosis challenging.”
The clinical appearance of AD may also mask signs of the bacterial infection, they added, and providers cannot rely on positive skin swab culture from the possibly infected area since S. aureus is so common in AD. An added challenge can occur in patients of different ethnicities, in whom both AD and bacterial infection may manifest differently. For example, perifollicular accentuation often occurs with AD in dark-skinned patients with violet-colored, often muted erythema.
An estimated 70% of lesional and 39% of nonlesional AD skin is colonized with S. aureus, the authors noted, but it’s not clear how best to recognize and manage asymptomatic S. aureus colonization. Among the factors that can increase susceptibility to S. aureus colonization and infection are impaired skin barriers, type 2 inflammation and lower levels of microbial diversity in the skin microbiome.
Specific clinical features of S. aureus in patients with AD include “weeping, honey-colored crusts and pustules, both interfollicular and follicular based,” and the pustules, though not common, can involve pain and itching. By comparison, signs of beta-hemolytic streptococcal infection may include “well-defined, bright red erythema, thick-walled pustules and heavy crusting,” the authors wrote.
Fever and lymphadenopathy may occur in severe cases, as well as abscesses, particularly with methicillin-resistant S. aureus (MRSA) infection. It’s unclear whether MRSA occurs more often in people with AD since its incidence varies so widely geographically, they noted.
In the differential diagnosis, providers should consider the possibility that a patient has a concomitant viral or fungal infection. Eczema herpeticum from herpes simplex virus is a common viral infection with risk factors that include “moderate to severe AD, filaggrin loss-of-function mutation, a history of S. aureus skin infection, greater allergen sensitization and type 2 immunity,” the authors wrote.
The yeast Malassezia is implicated in inflammation in patients with dermatitis that affects the head, neck, upper chest, back, and other areas high in sebaceous glands. Some patients have greater sensitivity to Malassezia, and “cross-reactivity between Malassezia-specific IgE and Candida albicans” has occurred as well, they wrote. Current evidence favors benefit from antifungal drugs, though not conclusively.
“Although we have some understanding of how S. aureus colonizes the skin and causes inflammation in AD, many questions related to this complex relationship remain unanswered,” the authors concluded. They added that better understanding the mechanisms of S. aureus and downstream host immune mediators of inflammatory pathways involving S. aureus could potentially lead to new therapeutic targets for infection in AD patients.
The statement was funded in part by the senior author’s fellowships from the National Institutes of Health Research, and the International Eczema Council received sponsorship from AbbVie, Amgen, Asana Biosciences, Celgene, Chugai, Dermavant, Dermira, Eli Lilly, Galderma, Incyte, LEO Pharma, Kyowa Kirin, Novartis, Pierre Fabre, Pfizer, Sanofi Genzyme, Regeneron Pharmaceuticals, Sienna and Valeant. Of the 16 authors, 13 disclosed financial ties to a wide range of pharmaceutical companies, including those listed above.
SOURCE: Alexander H et al. Br J Dermatol. 2019 Nov 1. doi: 10.1111/bjd.1864319.
While of infections in AD and features common to both can make it difficult to make a clinical diagnosis of infections.
Addressing this issue, the International Eczema Council Skin Infection Group reviewed the most current evidence on the clinical features of bacterial infections and the interaction between host and bacterial factors that affect severity and morbidity in people with atopic dermatitis (AD). Recurrent skin infections, especially from Staphylococcus aureus and occasionally from beta-hemolytic streptococci, are more common in people with AD than those who do not have AD for a variety of reasons, Helen Alexander, MD, from the unit for population-based dermatology research at St. John’s Institute of Dermatology, Guy’s and St Thomas’ NHS Foundation Trust and King’s College London, and associates wrote in the review article published in the British Journal of Dermatology.
“The reduced skin barrier, cutaneous innate and adaptive immune abnormalities and trauma from scratching all contribute to the increased risk of skin infection,” they wrote. “However, the wide variability in clinical presentation of bacterial infection in AD and the inherent features of AD – cutaneous erythema and warmth, oozing associated with edema, and regional lymphadenopathy – overlap with those of infection, making clinical diagnosis challenging.”
The clinical appearance of AD may also mask signs of the bacterial infection, they added, and providers cannot rely on positive skin swab culture from the possibly infected area since S. aureus is so common in AD. An added challenge can occur in patients of different ethnicities, in whom both AD and bacterial infection may manifest differently. For example, perifollicular accentuation often occurs with AD in dark-skinned patients with violet-colored, often muted erythema.
An estimated 70% of lesional and 39% of nonlesional AD skin is colonized with S. aureus, the authors noted, but it’s not clear how best to recognize and manage asymptomatic S. aureus colonization. Among the factors that can increase susceptibility to S. aureus colonization and infection are impaired skin barriers, type 2 inflammation and lower levels of microbial diversity in the skin microbiome.
Specific clinical features of S. aureus in patients with AD include “weeping, honey-colored crusts and pustules, both interfollicular and follicular based,” and the pustules, though not common, can involve pain and itching. By comparison, signs of beta-hemolytic streptococcal infection may include “well-defined, bright red erythema, thick-walled pustules and heavy crusting,” the authors wrote.
Fever and lymphadenopathy may occur in severe cases, as well as abscesses, particularly with methicillin-resistant S. aureus (MRSA) infection. It’s unclear whether MRSA occurs more often in people with AD since its incidence varies so widely geographically, they noted.
In the differential diagnosis, providers should consider the possibility that a patient has a concomitant viral or fungal infection. Eczema herpeticum from herpes simplex virus is a common viral infection with risk factors that include “moderate to severe AD, filaggrin loss-of-function mutation, a history of S. aureus skin infection, greater allergen sensitization and type 2 immunity,” the authors wrote.
The yeast Malassezia is implicated in inflammation in patients with dermatitis that affects the head, neck, upper chest, back, and other areas high in sebaceous glands. Some patients have greater sensitivity to Malassezia, and “cross-reactivity between Malassezia-specific IgE and Candida albicans” has occurred as well, they wrote. Current evidence favors benefit from antifungal drugs, though not conclusively.
“Although we have some understanding of how S. aureus colonizes the skin and causes inflammation in AD, many questions related to this complex relationship remain unanswered,” the authors concluded. They added that better understanding the mechanisms of S. aureus and downstream host immune mediators of inflammatory pathways involving S. aureus could potentially lead to new therapeutic targets for infection in AD patients.
The statement was funded in part by the senior author’s fellowships from the National Institutes of Health Research, and the International Eczema Council received sponsorship from AbbVie, Amgen, Asana Biosciences, Celgene, Chugai, Dermavant, Dermira, Eli Lilly, Galderma, Incyte, LEO Pharma, Kyowa Kirin, Novartis, Pierre Fabre, Pfizer, Sanofi Genzyme, Regeneron Pharmaceuticals, Sienna and Valeant. Of the 16 authors, 13 disclosed financial ties to a wide range of pharmaceutical companies, including those listed above.
SOURCE: Alexander H et al. Br J Dermatol. 2019 Nov 1. doi: 10.1111/bjd.1864319.
While of infections in AD and features common to both can make it difficult to make a clinical diagnosis of infections.
Addressing this issue, the International Eczema Council Skin Infection Group reviewed the most current evidence on the clinical features of bacterial infections and the interaction between host and bacterial factors that affect severity and morbidity in people with atopic dermatitis (AD). Recurrent skin infections, especially from Staphylococcus aureus and occasionally from beta-hemolytic streptococci, are more common in people with AD than those who do not have AD for a variety of reasons, Helen Alexander, MD, from the unit for population-based dermatology research at St. John’s Institute of Dermatology, Guy’s and St Thomas’ NHS Foundation Trust and King’s College London, and associates wrote in the review article published in the British Journal of Dermatology.
“The reduced skin barrier, cutaneous innate and adaptive immune abnormalities and trauma from scratching all contribute to the increased risk of skin infection,” they wrote. “However, the wide variability in clinical presentation of bacterial infection in AD and the inherent features of AD – cutaneous erythema and warmth, oozing associated with edema, and regional lymphadenopathy – overlap with those of infection, making clinical diagnosis challenging.”
The clinical appearance of AD may also mask signs of the bacterial infection, they added, and providers cannot rely on positive skin swab culture from the possibly infected area since S. aureus is so common in AD. An added challenge can occur in patients of different ethnicities, in whom both AD and bacterial infection may manifest differently. For example, perifollicular accentuation often occurs with AD in dark-skinned patients with violet-colored, often muted erythema.
An estimated 70% of lesional and 39% of nonlesional AD skin is colonized with S. aureus, the authors noted, but it’s not clear how best to recognize and manage asymptomatic S. aureus colonization. Among the factors that can increase susceptibility to S. aureus colonization and infection are impaired skin barriers, type 2 inflammation and lower levels of microbial diversity in the skin microbiome.
Specific clinical features of S. aureus in patients with AD include “weeping, honey-colored crusts and pustules, both interfollicular and follicular based,” and the pustules, though not common, can involve pain and itching. By comparison, signs of beta-hemolytic streptococcal infection may include “well-defined, bright red erythema, thick-walled pustules and heavy crusting,” the authors wrote.
Fever and lymphadenopathy may occur in severe cases, as well as abscesses, particularly with methicillin-resistant S. aureus (MRSA) infection. It’s unclear whether MRSA occurs more often in people with AD since its incidence varies so widely geographically, they noted.
In the differential diagnosis, providers should consider the possibility that a patient has a concomitant viral or fungal infection. Eczema herpeticum from herpes simplex virus is a common viral infection with risk factors that include “moderate to severe AD, filaggrin loss-of-function mutation, a history of S. aureus skin infection, greater allergen sensitization and type 2 immunity,” the authors wrote.
The yeast Malassezia is implicated in inflammation in patients with dermatitis that affects the head, neck, upper chest, back, and other areas high in sebaceous glands. Some patients have greater sensitivity to Malassezia, and “cross-reactivity between Malassezia-specific IgE and Candida albicans” has occurred as well, they wrote. Current evidence favors benefit from antifungal drugs, though not conclusively.
“Although we have some understanding of how S. aureus colonizes the skin and causes inflammation in AD, many questions related to this complex relationship remain unanswered,” the authors concluded. They added that better understanding the mechanisms of S. aureus and downstream host immune mediators of inflammatory pathways involving S. aureus could potentially lead to new therapeutic targets for infection in AD patients.
The statement was funded in part by the senior author’s fellowships from the National Institutes of Health Research, and the International Eczema Council received sponsorship from AbbVie, Amgen, Asana Biosciences, Celgene, Chugai, Dermavant, Dermira, Eli Lilly, Galderma, Incyte, LEO Pharma, Kyowa Kirin, Novartis, Pierre Fabre, Pfizer, Sanofi Genzyme, Regeneron Pharmaceuticals, Sienna and Valeant. Of the 16 authors, 13 disclosed financial ties to a wide range of pharmaceutical companies, including those listed above.
SOURCE: Alexander H et al. Br J Dermatol. 2019 Nov 1. doi: 10.1111/bjd.1864319.
FROM THE BRITISH JOURNAL OF DERMATOLOGY
Fast, aggressive eczema treatment linked to fewer food allergies by age 2
Researchers in Japan report that
For their research published in the Journal of Allergy and Clinical Immunology: In Practice, Yumiko Miyaji, MD, PhD, of Japan’s National Center for Child Health and Development in Tokyo and colleagues looked at 3 years’ worth of records for 177 infants younger than 1 year of age seen at a hospital allergy center for eczema. Of these infants, 89 were treated with betamethasone valerate within 4 months of disease onset, and 88 were treated after 4 months of onset. Most (142) were followed-up at 22-24 months, when all were in complete remission or near remission from eczema.
At follow-up, clinicians collected information about anaphylactic reactions to food, administered specific food challenges, and tested serum immunoglobin E levels for food allergens. Dr. Miyaji and colleagues found a significant difference in the prevalence of allergies between the early-treated and late-treated groups to chicken egg, cow’s milk, wheat, peanuts, soy, or fish (25% vs. 46%, respectively; P equal to .013). For individual food allergies, only chicken egg was associated with a statistically significant difference in prevalence (15% vs 36%, P equal to .006).
“Our present study may be the first to demonstrate that early aggressive topical corticosteroid treatment to shorten the duration of eczema in infants was significantly associated with a decrease in later development of [food allergies],” Dr. Miyaji and colleagues wrote in their analysis.
The investigators acknowledged as limitations of their study some between-group differences at baseline, with characteristics such as Staphylococcus aureus infections and some inflammatory biomarkers higher in the early treatment group.
The Japan Agency for Medical Research and Development supported the study, and the investigators disclosed no conflicts of interest related to their findings.
SOURCE: Miyaji Y et al. J Allergy Clin Immunol Pract. 2019. doi: 10.1016/j.jaip.2019.11.036
Researchers in Japan report that
For their research published in the Journal of Allergy and Clinical Immunology: In Practice, Yumiko Miyaji, MD, PhD, of Japan’s National Center for Child Health and Development in Tokyo and colleagues looked at 3 years’ worth of records for 177 infants younger than 1 year of age seen at a hospital allergy center for eczema. Of these infants, 89 were treated with betamethasone valerate within 4 months of disease onset, and 88 were treated after 4 months of onset. Most (142) were followed-up at 22-24 months, when all were in complete remission or near remission from eczema.
At follow-up, clinicians collected information about anaphylactic reactions to food, administered specific food challenges, and tested serum immunoglobin E levels for food allergens. Dr. Miyaji and colleagues found a significant difference in the prevalence of allergies between the early-treated and late-treated groups to chicken egg, cow’s milk, wheat, peanuts, soy, or fish (25% vs. 46%, respectively; P equal to .013). For individual food allergies, only chicken egg was associated with a statistically significant difference in prevalence (15% vs 36%, P equal to .006).
“Our present study may be the first to demonstrate that early aggressive topical corticosteroid treatment to shorten the duration of eczema in infants was significantly associated with a decrease in later development of [food allergies],” Dr. Miyaji and colleagues wrote in their analysis.
The investigators acknowledged as limitations of their study some between-group differences at baseline, with characteristics such as Staphylococcus aureus infections and some inflammatory biomarkers higher in the early treatment group.
The Japan Agency for Medical Research and Development supported the study, and the investigators disclosed no conflicts of interest related to their findings.
SOURCE: Miyaji Y et al. J Allergy Clin Immunol Pract. 2019. doi: 10.1016/j.jaip.2019.11.036
Researchers in Japan report that
For their research published in the Journal of Allergy and Clinical Immunology: In Practice, Yumiko Miyaji, MD, PhD, of Japan’s National Center for Child Health and Development in Tokyo and colleagues looked at 3 years’ worth of records for 177 infants younger than 1 year of age seen at a hospital allergy center for eczema. Of these infants, 89 were treated with betamethasone valerate within 4 months of disease onset, and 88 were treated after 4 months of onset. Most (142) were followed-up at 22-24 months, when all were in complete remission or near remission from eczema.
At follow-up, clinicians collected information about anaphylactic reactions to food, administered specific food challenges, and tested serum immunoglobin E levels for food allergens. Dr. Miyaji and colleagues found a significant difference in the prevalence of allergies between the early-treated and late-treated groups to chicken egg, cow’s milk, wheat, peanuts, soy, or fish (25% vs. 46%, respectively; P equal to .013). For individual food allergies, only chicken egg was associated with a statistically significant difference in prevalence (15% vs 36%, P equal to .006).
“Our present study may be the first to demonstrate that early aggressive topical corticosteroid treatment to shorten the duration of eczema in infants was significantly associated with a decrease in later development of [food allergies],” Dr. Miyaji and colleagues wrote in their analysis.
The investigators acknowledged as limitations of their study some between-group differences at baseline, with characteristics such as Staphylococcus aureus infections and some inflammatory biomarkers higher in the early treatment group.
The Japan Agency for Medical Research and Development supported the study, and the investigators disclosed no conflicts of interest related to their findings.
SOURCE: Miyaji Y et al. J Allergy Clin Immunol Pract. 2019. doi: 10.1016/j.jaip.2019.11.036
FROM THE JOURNAL OF ALLERGY AND CLINICAL IMMUNOLOGY: IN PRACTICE
Atopic dermatitis in egg-, milk-allergic kids may up anaphylaxis risk
compared with allergic patients without atopic dermatitis, based on retrospective data from 347 individuals.
Atopic dermatitis has been associated with increased risk of food allergies, but the association and predictive factors of skin reactions to certain foods remain unclear, wrote Bryce C. Hoffman, MD, of National Jewish Health, Denver, and colleagues.
In a letter published in the Annals of Allergy, Asthma & Immunology, the researchers identified children aged 0-18 years with peanut, cow’s milk, and/or egg allergies with or without atopic dermatitis (AD) using an institutional research database and conducted a retrospective study of medical records.
Overall, children with egg and milk allergies plus AD had significantly higher rates of anaphylaxis than allergic children without AD (47% vs. 11% for egg, 50% vs. 19% for milk). Anaphylaxis rates were similar in children with peanut allergies with or without AD (27% vs. 23%).
“This finding may suggest that skin barrier dysfunction plays a role in the severity of [food allergy]. However, this is not universal to all food antigens, and other mechanisms are likely important in the association of anaphylaxis with a particular food,” the researchers noted.
Rates of tolerance for both baked egg and baked milk were similar between AD and non-AD patients (83% vs. 61% for milk; 82% vs. 67% for egg). In addition, levels of total IgE were increased in children with egg and milk allergies plus AD, compared with children without AD. However, children with peanut allergies plus AD had decreased total IgE, compared with children with peanut allergies but no AD. This “may support a link between Th2 polarization and [food allergy] severity, ” Dr. Hoffman and associates wrote.
The findings were limited by several factors, including the retrospective study design, exclusion of many patients, and lack of data on the amount of food that triggered anaphylactic reactions, the researchers noted.
Nonetheless, the results suggest that children with atopic dermatitis and allergies to eggs and milk are at increased risk and that clinicians should counsel these patients and families about the potential for more-severe reactions to oral food challenges, Dr. Hoffman and associates concluded.
The study was supported by National Jewish Health and the Edelstein Family Chair of Pediatric Allergy and Immunology. The researchers had no financial conflicts to disclose.
SOURCE: Hoffman BC et al. Ann Allergy Asthma Immunol. 2019 Sep 11. doi: 10.1016/j.anai.2019.09.008.
compared with allergic patients without atopic dermatitis, based on retrospective data from 347 individuals.
Atopic dermatitis has been associated with increased risk of food allergies, but the association and predictive factors of skin reactions to certain foods remain unclear, wrote Bryce C. Hoffman, MD, of National Jewish Health, Denver, and colleagues.
In a letter published in the Annals of Allergy, Asthma & Immunology, the researchers identified children aged 0-18 years with peanut, cow’s milk, and/or egg allergies with or without atopic dermatitis (AD) using an institutional research database and conducted a retrospective study of medical records.
Overall, children with egg and milk allergies plus AD had significantly higher rates of anaphylaxis than allergic children without AD (47% vs. 11% for egg, 50% vs. 19% for milk). Anaphylaxis rates were similar in children with peanut allergies with or without AD (27% vs. 23%).
“This finding may suggest that skin barrier dysfunction plays a role in the severity of [food allergy]. However, this is not universal to all food antigens, and other mechanisms are likely important in the association of anaphylaxis with a particular food,” the researchers noted.
Rates of tolerance for both baked egg and baked milk were similar between AD and non-AD patients (83% vs. 61% for milk; 82% vs. 67% for egg). In addition, levels of total IgE were increased in children with egg and milk allergies plus AD, compared with children without AD. However, children with peanut allergies plus AD had decreased total IgE, compared with children with peanut allergies but no AD. This “may support a link between Th2 polarization and [food allergy] severity, ” Dr. Hoffman and associates wrote.
The findings were limited by several factors, including the retrospective study design, exclusion of many patients, and lack of data on the amount of food that triggered anaphylactic reactions, the researchers noted.
Nonetheless, the results suggest that children with atopic dermatitis and allergies to eggs and milk are at increased risk and that clinicians should counsel these patients and families about the potential for more-severe reactions to oral food challenges, Dr. Hoffman and associates concluded.
The study was supported by National Jewish Health and the Edelstein Family Chair of Pediatric Allergy and Immunology. The researchers had no financial conflicts to disclose.
SOURCE: Hoffman BC et al. Ann Allergy Asthma Immunol. 2019 Sep 11. doi: 10.1016/j.anai.2019.09.008.
compared with allergic patients without atopic dermatitis, based on retrospective data from 347 individuals.
Atopic dermatitis has been associated with increased risk of food allergies, but the association and predictive factors of skin reactions to certain foods remain unclear, wrote Bryce C. Hoffman, MD, of National Jewish Health, Denver, and colleagues.
In a letter published in the Annals of Allergy, Asthma & Immunology, the researchers identified children aged 0-18 years with peanut, cow’s milk, and/or egg allergies with or without atopic dermatitis (AD) using an institutional research database and conducted a retrospective study of medical records.
Overall, children with egg and milk allergies plus AD had significantly higher rates of anaphylaxis than allergic children without AD (47% vs. 11% for egg, 50% vs. 19% for milk). Anaphylaxis rates were similar in children with peanut allergies with or without AD (27% vs. 23%).
“This finding may suggest that skin barrier dysfunction plays a role in the severity of [food allergy]. However, this is not universal to all food antigens, and other mechanisms are likely important in the association of anaphylaxis with a particular food,” the researchers noted.
Rates of tolerance for both baked egg and baked milk were similar between AD and non-AD patients (83% vs. 61% for milk; 82% vs. 67% for egg). In addition, levels of total IgE were increased in children with egg and milk allergies plus AD, compared with children without AD. However, children with peanut allergies plus AD had decreased total IgE, compared with children with peanut allergies but no AD. This “may support a link between Th2 polarization and [food allergy] severity, ” Dr. Hoffman and associates wrote.
The findings were limited by several factors, including the retrospective study design, exclusion of many patients, and lack of data on the amount of food that triggered anaphylactic reactions, the researchers noted.
Nonetheless, the results suggest that children with atopic dermatitis and allergies to eggs and milk are at increased risk and that clinicians should counsel these patients and families about the potential for more-severe reactions to oral food challenges, Dr. Hoffman and associates concluded.
The study was supported by National Jewish Health and the Edelstein Family Chair of Pediatric Allergy and Immunology. The researchers had no financial conflicts to disclose.
SOURCE: Hoffman BC et al. Ann Allergy Asthma Immunol. 2019 Sep 11. doi: 10.1016/j.anai.2019.09.008.
FROM THE ANNALS OF ALLERGY, ASTHMA & IMMUNOLOGY
Frequent soaks ease pediatric atopic dermatitis
A regimen of twice-daily baths followed by occlusive moisturizer improved atopic dermatitis in children with moderate to severe disease more effectively than did a twice-weekly protocol, based on data from 42 children.
Guidelines for bathing frequency for children with atopic dermatitis are inconsistent and often confusing for parents, according to Ivan D. Cardona, MD, of Maine Medical Research Institute, Portland, and colleagues.
In a study published in the Journal of Allergy and Clinical Immunology: In Practice, the researchers randomized 42 children aged 6 months to 11 years with moderate to severe atopic dermatitis to a routine of twice-weekly “soak and seal” (SS) procedures consisting of soaking baths for 10 minutes or less, followed by an occlusive emollient, or to twice-daily SS with baths of 15-20 minutes followed by emollient. The groups were treated for 2 weeks, then switched protocols. The study included a total of four clinic visits over 5 weeks. All patients also received standard of care low-potency topical corticosteroids and moisturizer.
Overall, the frequent bathing (“wet method”) led to a decrease of 21.2 on the SCORing Atopic Dermatitis Index (SCORAD) compared with the less frequent bathing (“dry method”). Improvements in SCORAD (the primary outcome) correlated with a secondary outcome of improved scores on the parent-rated Atopic Dermatitis Quickscore.
The findings were limited by several factors including the small sample size, large rate of attrition prior to randomization among initially screened children, lack of data on environmental factors such as water temperature and quality, and the lack of a washout period between the treatment protocols, the researchers noted. They acknowledged that “twice-daily SS bathing in the real world can be time consuming, making adherence difficult for families.”
However, the results suggest that the frequent bathing protocol was safe and effective at improving symptoms of atopic dermatitis, and may reduce steroid use, they concluded.
The researchers had no financial conflicts to disclose.
SOURCE: Cardona ID et al. J Allergy Clin Immunol Pract. 2019 Nov 13. doi: 10.1016/j.jaip.2019.10.042.
A regimen of twice-daily baths followed by occlusive moisturizer improved atopic dermatitis in children with moderate to severe disease more effectively than did a twice-weekly protocol, based on data from 42 children.
Guidelines for bathing frequency for children with atopic dermatitis are inconsistent and often confusing for parents, according to Ivan D. Cardona, MD, of Maine Medical Research Institute, Portland, and colleagues.
In a study published in the Journal of Allergy and Clinical Immunology: In Practice, the researchers randomized 42 children aged 6 months to 11 years with moderate to severe atopic dermatitis to a routine of twice-weekly “soak and seal” (SS) procedures consisting of soaking baths for 10 minutes or less, followed by an occlusive emollient, or to twice-daily SS with baths of 15-20 minutes followed by emollient. The groups were treated for 2 weeks, then switched protocols. The study included a total of four clinic visits over 5 weeks. All patients also received standard of care low-potency topical corticosteroids and moisturizer.
Overall, the frequent bathing (“wet method”) led to a decrease of 21.2 on the SCORing Atopic Dermatitis Index (SCORAD) compared with the less frequent bathing (“dry method”). Improvements in SCORAD (the primary outcome) correlated with a secondary outcome of improved scores on the parent-rated Atopic Dermatitis Quickscore.
The findings were limited by several factors including the small sample size, large rate of attrition prior to randomization among initially screened children, lack of data on environmental factors such as water temperature and quality, and the lack of a washout period between the treatment protocols, the researchers noted. They acknowledged that “twice-daily SS bathing in the real world can be time consuming, making adherence difficult for families.”
However, the results suggest that the frequent bathing protocol was safe and effective at improving symptoms of atopic dermatitis, and may reduce steroid use, they concluded.
The researchers had no financial conflicts to disclose.
SOURCE: Cardona ID et al. J Allergy Clin Immunol Pract. 2019 Nov 13. doi: 10.1016/j.jaip.2019.10.042.
A regimen of twice-daily baths followed by occlusive moisturizer improved atopic dermatitis in children with moderate to severe disease more effectively than did a twice-weekly protocol, based on data from 42 children.
Guidelines for bathing frequency for children with atopic dermatitis are inconsistent and often confusing for parents, according to Ivan D. Cardona, MD, of Maine Medical Research Institute, Portland, and colleagues.
In a study published in the Journal of Allergy and Clinical Immunology: In Practice, the researchers randomized 42 children aged 6 months to 11 years with moderate to severe atopic dermatitis to a routine of twice-weekly “soak and seal” (SS) procedures consisting of soaking baths for 10 minutes or less, followed by an occlusive emollient, or to twice-daily SS with baths of 15-20 minutes followed by emollient. The groups were treated for 2 weeks, then switched protocols. The study included a total of four clinic visits over 5 weeks. All patients also received standard of care low-potency topical corticosteroids and moisturizer.
Overall, the frequent bathing (“wet method”) led to a decrease of 21.2 on the SCORing Atopic Dermatitis Index (SCORAD) compared with the less frequent bathing (“dry method”). Improvements in SCORAD (the primary outcome) correlated with a secondary outcome of improved scores on the parent-rated Atopic Dermatitis Quickscore.
The findings were limited by several factors including the small sample size, large rate of attrition prior to randomization among initially screened children, lack of data on environmental factors such as water temperature and quality, and the lack of a washout period between the treatment protocols, the researchers noted. They acknowledged that “twice-daily SS bathing in the real world can be time consuming, making adherence difficult for families.”
However, the results suggest that the frequent bathing protocol was safe and effective at improving symptoms of atopic dermatitis, and may reduce steroid use, they concluded.
The researchers had no financial conflicts to disclose.
SOURCE: Cardona ID et al. J Allergy Clin Immunol Pract. 2019 Nov 13. doi: 10.1016/j.jaip.2019.10.042.
FROM THE JOURNAL OF ALLERGY AND CLINICAL IMMUNOLOGY: IN PRACTICE
Omalizumab proves effective for severe pediatric atopic dermatitis
A new study has found that omalizumab (Xolair) reduced severity and improved quality of life in pediatric patients with severe atopic dermatitis.
“Future work with an even larger sample size, a longer duration, and higher-affinity versions of omalizumab would clarify the precise role of anti-IgE therapy and its ideal target population,” wrote Susan Chan, MD, of Guy’s and St. Thomas’ NHS Foundation Trust in London and her coauthors. The study was published in JAMA Pediatrics.
To determine the benefits of omalizumab in reducing immunoglobulin E levels and thereby treating severe childhood eczema, the researchers launched the Atopic Dermatitis Anti-IgE Pediatric Trial (ADAPT). This randomized clinical trial recruited 62 patients between the ages of 4 and 19 years with severe eczema, which was defined as a score over 40 on the objective Scoring Atopic Dermatitis (SCORAD) index. They received 24 weeks of treatment with either omalizumab (n = 30) or placebo (n = 32) followed by 24 weeks of follow-up. Participants had a mean age of 10.3 years.
After 24 weeks, the adjusted mean difference in objective SCORAD index between the two groups was –6.9 (95% confidence interval, –12.2 to –1.5; P = .01) and significantly favored omalizumab therapy. The adjusted mean difference for the Eczema Area and Severity Index (–6.7; 95% CI, –13.2 to –0.1) also favored omalizumab. In regard to quality of life, after 24 weeks the Children’s Dermatology Life Quality Index/Dermatology Life Quality Index favored the omalizumab group with an adjusted mean difference of –3.5 (95% CI, –6.4 to –0.5).
In an accompanying editorial, Ann Chen Wu, MD, of Harvard Medical School in Boston noted that the results of the study from Chan et al. were promising but “more questions need to be answered before the drug can be used to treat atopic dermatitis in clinical practice” (JAMA Pediatr. 2019 Nov. 25. doi: 10.1001/jamapediatrics.2019.4509).
Her initial concern was price; she acknowledged that “omalizumab is a costly intervention” but said atopic dermatitis is also costly, raising the question as to whether the high costs of both justify treatment. In addition, omalizumab as treatment can come with both benefits and harms. Severe atopic dermatitis can decrease quality of life and, though omalizumab appears to be safe, there are adverse effects and logistical burdens to overcome.
More than anything, she recognized the need to prioritize, wondering what level of atopic dermatitis patients would truly benefit from this level of treatment. “Is using a $100,000-per-year medication for an itchy condition an overtreatment,” she asked, “or a lifesaver?”
The study was funded by the National Institute for Health Research Efficacy and Mechanism Evaluation Programme and Guy’s and St. Thomas’ Charity. The authors had numerous financial disclosures, including receiving grants from the NIHR EME Programme and Guy’s and St. Thomas’ Charity along with active and placebo drugs from Novartis for use in the study. Dr. Wu reported receiving a grant from GlaxoSmithKline.
SOURCE: Chan S et al. JAMA Pediatr. 2019 Nov 25. doi: 10.1001/jamapediatrics.2019.4476.
A new study has found that omalizumab (Xolair) reduced severity and improved quality of life in pediatric patients with severe atopic dermatitis.
“Future work with an even larger sample size, a longer duration, and higher-affinity versions of omalizumab would clarify the precise role of anti-IgE therapy and its ideal target population,” wrote Susan Chan, MD, of Guy’s and St. Thomas’ NHS Foundation Trust in London and her coauthors. The study was published in JAMA Pediatrics.
To determine the benefits of omalizumab in reducing immunoglobulin E levels and thereby treating severe childhood eczema, the researchers launched the Atopic Dermatitis Anti-IgE Pediatric Trial (ADAPT). This randomized clinical trial recruited 62 patients between the ages of 4 and 19 years with severe eczema, which was defined as a score over 40 on the objective Scoring Atopic Dermatitis (SCORAD) index. They received 24 weeks of treatment with either omalizumab (n = 30) or placebo (n = 32) followed by 24 weeks of follow-up. Participants had a mean age of 10.3 years.
After 24 weeks, the adjusted mean difference in objective SCORAD index between the two groups was –6.9 (95% confidence interval, –12.2 to –1.5; P = .01) and significantly favored omalizumab therapy. The adjusted mean difference for the Eczema Area and Severity Index (–6.7; 95% CI, –13.2 to –0.1) also favored omalizumab. In regard to quality of life, after 24 weeks the Children’s Dermatology Life Quality Index/Dermatology Life Quality Index favored the omalizumab group with an adjusted mean difference of –3.5 (95% CI, –6.4 to –0.5).
In an accompanying editorial, Ann Chen Wu, MD, of Harvard Medical School in Boston noted that the results of the study from Chan et al. were promising but “more questions need to be answered before the drug can be used to treat atopic dermatitis in clinical practice” (JAMA Pediatr. 2019 Nov. 25. doi: 10.1001/jamapediatrics.2019.4509).
Her initial concern was price; she acknowledged that “omalizumab is a costly intervention” but said atopic dermatitis is also costly, raising the question as to whether the high costs of both justify treatment. In addition, omalizumab as treatment can come with both benefits and harms. Severe atopic dermatitis can decrease quality of life and, though omalizumab appears to be safe, there are adverse effects and logistical burdens to overcome.
More than anything, she recognized the need to prioritize, wondering what level of atopic dermatitis patients would truly benefit from this level of treatment. “Is using a $100,000-per-year medication for an itchy condition an overtreatment,” she asked, “or a lifesaver?”
The study was funded by the National Institute for Health Research Efficacy and Mechanism Evaluation Programme and Guy’s and St. Thomas’ Charity. The authors had numerous financial disclosures, including receiving grants from the NIHR EME Programme and Guy’s and St. Thomas’ Charity along with active and placebo drugs from Novartis for use in the study. Dr. Wu reported receiving a grant from GlaxoSmithKline.
SOURCE: Chan S et al. JAMA Pediatr. 2019 Nov 25. doi: 10.1001/jamapediatrics.2019.4476.
A new study has found that omalizumab (Xolair) reduced severity and improved quality of life in pediatric patients with severe atopic dermatitis.
“Future work with an even larger sample size, a longer duration, and higher-affinity versions of omalizumab would clarify the precise role of anti-IgE therapy and its ideal target population,” wrote Susan Chan, MD, of Guy’s and St. Thomas’ NHS Foundation Trust in London and her coauthors. The study was published in JAMA Pediatrics.
To determine the benefits of omalizumab in reducing immunoglobulin E levels and thereby treating severe childhood eczema, the researchers launched the Atopic Dermatitis Anti-IgE Pediatric Trial (ADAPT). This randomized clinical trial recruited 62 patients between the ages of 4 and 19 years with severe eczema, which was defined as a score over 40 on the objective Scoring Atopic Dermatitis (SCORAD) index. They received 24 weeks of treatment with either omalizumab (n = 30) or placebo (n = 32) followed by 24 weeks of follow-up. Participants had a mean age of 10.3 years.
After 24 weeks, the adjusted mean difference in objective SCORAD index between the two groups was –6.9 (95% confidence interval, –12.2 to –1.5; P = .01) and significantly favored omalizumab therapy. The adjusted mean difference for the Eczema Area and Severity Index (–6.7; 95% CI, –13.2 to –0.1) also favored omalizumab. In regard to quality of life, after 24 weeks the Children’s Dermatology Life Quality Index/Dermatology Life Quality Index favored the omalizumab group with an adjusted mean difference of –3.5 (95% CI, –6.4 to –0.5).
In an accompanying editorial, Ann Chen Wu, MD, of Harvard Medical School in Boston noted that the results of the study from Chan et al. were promising but “more questions need to be answered before the drug can be used to treat atopic dermatitis in clinical practice” (JAMA Pediatr. 2019 Nov. 25. doi: 10.1001/jamapediatrics.2019.4509).
Her initial concern was price; she acknowledged that “omalizumab is a costly intervention” but said atopic dermatitis is also costly, raising the question as to whether the high costs of both justify treatment. In addition, omalizumab as treatment can come with both benefits and harms. Severe atopic dermatitis can decrease quality of life and, though omalizumab appears to be safe, there are adverse effects and logistical burdens to overcome.
More than anything, she recognized the need to prioritize, wondering what level of atopic dermatitis patients would truly benefit from this level of treatment. “Is using a $100,000-per-year medication for an itchy condition an overtreatment,” she asked, “or a lifesaver?”
The study was funded by the National Institute for Health Research Efficacy and Mechanism Evaluation Programme and Guy’s and St. Thomas’ Charity. The authors had numerous financial disclosures, including receiving grants from the NIHR EME Programme and Guy’s and St. Thomas’ Charity along with active and placebo drugs from Novartis for use in the study. Dr. Wu reported receiving a grant from GlaxoSmithKline.
SOURCE: Chan S et al. JAMA Pediatr. 2019 Nov 25. doi: 10.1001/jamapediatrics.2019.4476.
FROM JAMA PEDIATRICS
Naturopaths emphasize role of diet in atopic dermatitis
, based on data from a small survey of the two.
Data from previous studies show that more than half of patients with AD have used complementary and alternative medicine in addition to allopathic care, but providers may be unaware of each other’s treatment approaches and confuse patients, wrote Julie Dhossche, MD, of Oregon Health & Science University, Portland, and her colleagues.
In a study published in Pediatric Dermatology, the researchers assessed results of an 11-question, free-text survey of 30 allopathic providers and 21 naturopathic providers about AD. The survey included questions on patient education and evaluation, skin care, and treatment.
Overall, both allopathic and naturopathic providers recommended skin care protocols involving moisturization and “soak and seal” bathing. However, allopathic providers were more likely to prescribe topical corticosteroids for mild to moderate disease (100% vs. 19%), followed by phototherapy and systemic treatments in more severe cases. Naturopathic providers were more likely than allopathic providers to choose topical botanicals, oils, or probiotics (52% vs. 0%) for mild to moderate disease, as well stress relief and acupuncture. Naturopathic providers favored topical corticosteroids and referrals to dermatologists for second- or third-line treatment.
Of note, 85% of naturopathic providers said they thought diet had a probable or definite role in AD, compared with 3% of allopathic providers.
In addition, naturopathic providers differed in their response to an optional question on the use of additional education about food and diet. A total of 11 of 19 naturopathic providers (58%) recommended dietary changes, including “remove potential food allergens/reduce sugar” and “emphasize anti-inflammatory diet,” the researchers said.
“Confusion regarding the role of food in AD management is a common source of frustration for patients, and perhaps a consensus statement from both fields regarding the role of food allergy in AD management could be aspired toward in the name of reducing patient confusion,” they wrote.
The study findings were limited by several factors, including the small sample size and self-selection bias, as well as the subjective nature of an open-ended survey, the researchers noted. However, the results provide evidence of differences in treatment approaches between allopathic and naturopathic providers and suggest that “respectful collaboration between allopathic and naturopathic providers will help practitioners find common ground, decrease patient confusion, and improve patient outcomes,” they concluded.
The researchers had no financial conflicts to disclose.
SOURCE: Dhossche J et al. Ped Dermatol. 2019 Nov 19. doi: 10.1111/pde.14036.
, based on data from a small survey of the two.
Data from previous studies show that more than half of patients with AD have used complementary and alternative medicine in addition to allopathic care, but providers may be unaware of each other’s treatment approaches and confuse patients, wrote Julie Dhossche, MD, of Oregon Health & Science University, Portland, and her colleagues.
In a study published in Pediatric Dermatology, the researchers assessed results of an 11-question, free-text survey of 30 allopathic providers and 21 naturopathic providers about AD. The survey included questions on patient education and evaluation, skin care, and treatment.
Overall, both allopathic and naturopathic providers recommended skin care protocols involving moisturization and “soak and seal” bathing. However, allopathic providers were more likely to prescribe topical corticosteroids for mild to moderate disease (100% vs. 19%), followed by phototherapy and systemic treatments in more severe cases. Naturopathic providers were more likely than allopathic providers to choose topical botanicals, oils, or probiotics (52% vs. 0%) for mild to moderate disease, as well stress relief and acupuncture. Naturopathic providers favored topical corticosteroids and referrals to dermatologists for second- or third-line treatment.
Of note, 85% of naturopathic providers said they thought diet had a probable or definite role in AD, compared with 3% of allopathic providers.
In addition, naturopathic providers differed in their response to an optional question on the use of additional education about food and diet. A total of 11 of 19 naturopathic providers (58%) recommended dietary changes, including “remove potential food allergens/reduce sugar” and “emphasize anti-inflammatory diet,” the researchers said.
“Confusion regarding the role of food in AD management is a common source of frustration for patients, and perhaps a consensus statement from both fields regarding the role of food allergy in AD management could be aspired toward in the name of reducing patient confusion,” they wrote.
The study findings were limited by several factors, including the small sample size and self-selection bias, as well as the subjective nature of an open-ended survey, the researchers noted. However, the results provide evidence of differences in treatment approaches between allopathic and naturopathic providers and suggest that “respectful collaboration between allopathic and naturopathic providers will help practitioners find common ground, decrease patient confusion, and improve patient outcomes,” they concluded.
The researchers had no financial conflicts to disclose.
SOURCE: Dhossche J et al. Ped Dermatol. 2019 Nov 19. doi: 10.1111/pde.14036.
, based on data from a small survey of the two.
Data from previous studies show that more than half of patients with AD have used complementary and alternative medicine in addition to allopathic care, but providers may be unaware of each other’s treatment approaches and confuse patients, wrote Julie Dhossche, MD, of Oregon Health & Science University, Portland, and her colleagues.
In a study published in Pediatric Dermatology, the researchers assessed results of an 11-question, free-text survey of 30 allopathic providers and 21 naturopathic providers about AD. The survey included questions on patient education and evaluation, skin care, and treatment.
Overall, both allopathic and naturopathic providers recommended skin care protocols involving moisturization and “soak and seal” bathing. However, allopathic providers were more likely to prescribe topical corticosteroids for mild to moderate disease (100% vs. 19%), followed by phototherapy and systemic treatments in more severe cases. Naturopathic providers were more likely than allopathic providers to choose topical botanicals, oils, or probiotics (52% vs. 0%) for mild to moderate disease, as well stress relief and acupuncture. Naturopathic providers favored topical corticosteroids and referrals to dermatologists for second- or third-line treatment.
Of note, 85% of naturopathic providers said they thought diet had a probable or definite role in AD, compared with 3% of allopathic providers.
In addition, naturopathic providers differed in their response to an optional question on the use of additional education about food and diet. A total of 11 of 19 naturopathic providers (58%) recommended dietary changes, including “remove potential food allergens/reduce sugar” and “emphasize anti-inflammatory diet,” the researchers said.
“Confusion regarding the role of food in AD management is a common source of frustration for patients, and perhaps a consensus statement from both fields regarding the role of food allergy in AD management could be aspired toward in the name of reducing patient confusion,” they wrote.
The study findings were limited by several factors, including the small sample size and self-selection bias, as well as the subjective nature of an open-ended survey, the researchers noted. However, the results provide evidence of differences in treatment approaches between allopathic and naturopathic providers and suggest that “respectful collaboration between allopathic and naturopathic providers will help practitioners find common ground, decrease patient confusion, and improve patient outcomes,” they concluded.
The researchers had no financial conflicts to disclose.
SOURCE: Dhossche J et al. Ped Dermatol. 2019 Nov 19. doi: 10.1111/pde.14036.
FROM PEDIATRIC DERMATOLOGY
Atopic dermatitis acts differently in certain populations
LAS VEGAS – Eczema is eczema is eczema, right? Maybe not. “Atopic dermatitis might not be one disease,” a dermatologist told colleagues, and treatments may need to be adjusted to reflect the age and ethnicity of patients.
More research is needed, Kenneth B. Gordon, MD, chair and professor of dermatology at the Medical College of Wisconsin, Milwaukee, said during a presentation at Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar. “We’re probably just on the tip of the iceberg of understanding the physiology of atopic dermatitis. Hopefully, it will lead to the therapeutic advances we’ve seen in psoriasis.”
As Dr. Gordon explained, there’s a , he said, “and our medicines aren’t well understood.”
As for the disease itself, he said, “you might hear a renowned [expert] say, ‘This is how it works,’ and another say, ‘This is absolutely not how it works.’ ” One camp focused on the skin barrier, he said, while another camp highlighted inflammation in AD.
“Both the barrier and inflammation are important,” he said. “There are multiple cell types and cytokines that are important, but we don’t know yet the relative importance of them all. You have this cytokine soup, and we’re still trying to figure out the driving forces.”
What is clear, Dr. Gordon said, is that AD acts differently in certain patient populations. It’s not the same in pediatric versus adult patients, he said, and it’s not the same in white versus black versus Asian patients. Research, for example, suggests that Th2, Th22, and Th17 pathways appear to be important in pediatric AD, but not Th1, he said. In contrast, the Th1 pathway plays a role in white adults – but not in black adults
Different cytokines appear to play different roles in these populations, he said. “One of the key things moving forward is going to be figuring out which patients you apply these medications to,” he noted.
Dr. Gordon has multiple disclosures including honoraria or research support from Abbvie, Lilly, Novartis, Pfizer, UCB, and others. SDEF and this news organization are owned by the same parent company.
LAS VEGAS – Eczema is eczema is eczema, right? Maybe not. “Atopic dermatitis might not be one disease,” a dermatologist told colleagues, and treatments may need to be adjusted to reflect the age and ethnicity of patients.
More research is needed, Kenneth B. Gordon, MD, chair and professor of dermatology at the Medical College of Wisconsin, Milwaukee, said during a presentation at Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar. “We’re probably just on the tip of the iceberg of understanding the physiology of atopic dermatitis. Hopefully, it will lead to the therapeutic advances we’ve seen in psoriasis.”
As Dr. Gordon explained, there’s a , he said, “and our medicines aren’t well understood.”
As for the disease itself, he said, “you might hear a renowned [expert] say, ‘This is how it works,’ and another say, ‘This is absolutely not how it works.’ ” One camp focused on the skin barrier, he said, while another camp highlighted inflammation in AD.
“Both the barrier and inflammation are important,” he said. “There are multiple cell types and cytokines that are important, but we don’t know yet the relative importance of them all. You have this cytokine soup, and we’re still trying to figure out the driving forces.”
What is clear, Dr. Gordon said, is that AD acts differently in certain patient populations. It’s not the same in pediatric versus adult patients, he said, and it’s not the same in white versus black versus Asian patients. Research, for example, suggests that Th2, Th22, and Th17 pathways appear to be important in pediatric AD, but not Th1, he said. In contrast, the Th1 pathway plays a role in white adults – but not in black adults
Different cytokines appear to play different roles in these populations, he said. “One of the key things moving forward is going to be figuring out which patients you apply these medications to,” he noted.
Dr. Gordon has multiple disclosures including honoraria or research support from Abbvie, Lilly, Novartis, Pfizer, UCB, and others. SDEF and this news organization are owned by the same parent company.
LAS VEGAS – Eczema is eczema is eczema, right? Maybe not. “Atopic dermatitis might not be one disease,” a dermatologist told colleagues, and treatments may need to be adjusted to reflect the age and ethnicity of patients.
More research is needed, Kenneth B. Gordon, MD, chair and professor of dermatology at the Medical College of Wisconsin, Milwaukee, said during a presentation at Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar. “We’re probably just on the tip of the iceberg of understanding the physiology of atopic dermatitis. Hopefully, it will lead to the therapeutic advances we’ve seen in psoriasis.”
As Dr. Gordon explained, there’s a , he said, “and our medicines aren’t well understood.”
As for the disease itself, he said, “you might hear a renowned [expert] say, ‘This is how it works,’ and another say, ‘This is absolutely not how it works.’ ” One camp focused on the skin barrier, he said, while another camp highlighted inflammation in AD.
“Both the barrier and inflammation are important,” he said. “There are multiple cell types and cytokines that are important, but we don’t know yet the relative importance of them all. You have this cytokine soup, and we’re still trying to figure out the driving forces.”
What is clear, Dr. Gordon said, is that AD acts differently in certain patient populations. It’s not the same in pediatric versus adult patients, he said, and it’s not the same in white versus black versus Asian patients. Research, for example, suggests that Th2, Th22, and Th17 pathways appear to be important in pediatric AD, but not Th1, he said. In contrast, the Th1 pathway plays a role in white adults – but not in black adults
Different cytokines appear to play different roles in these populations, he said. “One of the key things moving forward is going to be figuring out which patients you apply these medications to,” he noted.
Dr. Gordon has multiple disclosures including honoraria or research support from Abbvie, Lilly, Novartis, Pfizer, UCB, and others. SDEF and this news organization are owned by the same parent company.
REPORTING FROM SDEF LAS VEGAS DERMATOLOGY SEMINAR
Oral baricitinib performs well in phase 3 for atopic dermatitis
MADRID – Adding the oral Janus kinase (JAK) inhibitor baricitinib to standard atopic dermatitis therapy with low- and midpotency topical corticosteroids markedly improved disease severity and key patient-reported outcomes, compared with topical corticosteroids alone, in the phase 3, randomized, double-blind BREEZE-AD7 trial, Kristian Reich, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
in the phase 3 BREEZE-AD1 and -AD2 trials. But BREEZE-AD7 further advances the field because it’s the first phase 3 study testing the efficacy of a JAK inhibitor in combination with low- and midpotency topical steroids.
“I think this study is important because it looks into the situation that’s more like what happens in the real world, which is, as with dupilumab and other drugs, we use the systemic agent in combination with topical therapies and, in particular, with topical corticosteroids,” commented Dr. Reich, professor of dermatology at University Medical Center, Hamburg, and medical director at SCIderm, a scientific research company.
“This is what I think we can expect from existing and upcoming systemic therapies in atopic dermatitis: We will use them in combination with topical corticosteroids, and hopefully this will allow patients to dramatically reduce the concomitant use of topical corticosteroids, as shown here in BREEZE-AD7,” he added.
BREEZE-AD7 was a 16-week study that included 329 adults with moderate or severe atopic dermatitis who were randomized to low- and midpotency topical corticosteroids plus either baricitinib at 2 mg once daily, baricitinib at 4 mg once daily, or placebo. The group’s mean baseline Eczema Area and Severity Index (EASI) score was 29. Overall, 45% of participants had a baseline Investigator’s Global Assessment (IGA) of disease severity of 4 on a 0-4 scale.
The primary endpoint was achievement of an IGA of 0 or 1, meaning clear or almost clear, along with at least a 2-point IGA improvement from baseline at week 16. This was accomplished in 30.6% of those on 4 mg/day of baricitinib, 23.9% of patients in the 2-mg group, and 14.7% of controls.
The 4-mg dose of baricitinib was statistically superior to placebo; the 2-mg dose was not. However, Dr. Reich indicated he was untroubled by this because the primary endpoint was set at a high bar, and both doses of baricitinib proved to be significantly better than topical steroids plus placebo in terms of EASI 75 response rates, as well as reductions in itch, skin pain, and sleep problems, which aren’t captured in EASI scores (see graphic).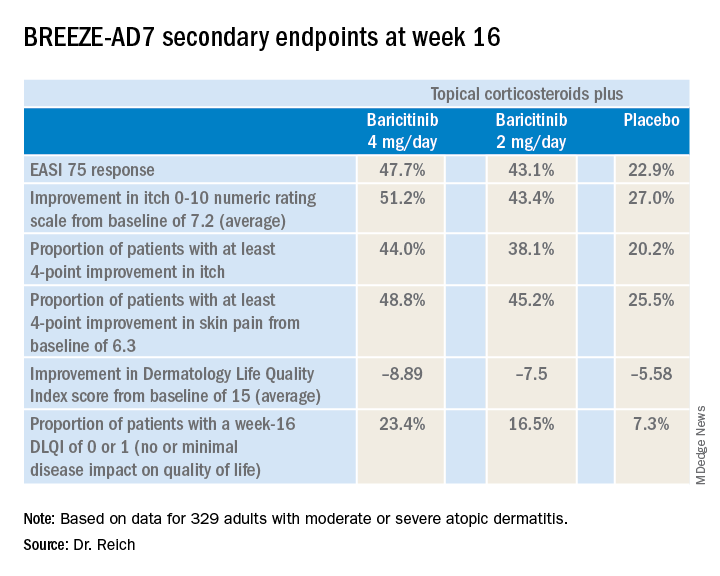
“One of my big learnings from this year’s EADV is that we have to rethink the dimensions of atopic dermatitis. I think we have underestimated the relevance of important symptoms such as itch, the impact atopic dermatitis has on pain, and the effect it has on sleeping problems,” the dermatologist said. “My feeling is that baricitinib is strongest in reducing itch, improving sleep, and reducing pain, but it also has good effects on the clinical signs of atopic dermatitis.”
The baricitinib-treated patients’ rapidity of improvement in the various endpoints was particularly impressive. Both doses of the JAK 1/2 inhibitor showed significant separation from the control group in the first week, and the majority of improvement occurred by week 4.
A key finding was that patients on baricitinib at 2 mg/day and 4 mg/day used a mean total of 162 g and 137 g of midpotency topical steroids, respectively, during the 16 weeks, compared with 225 g in the control group. The higher-dose baricitinib group was topical corticosteroid-free on 33% of study days, compared with 25% of days for the baricitinib 2 mg patients and 17% of days for controls.
In terms of safety, there was a case of pulmonary embolism in the higher-dose baricitinib group and an opportunistic toxoplasmosis eye infection in the control population. The frequency of oral herpes and herpes simplex virus infections was 2.8% in controls, 4.6% in the baricitinib 2-mg group, and 6.3% in the 4-mg group. There was also a signal of a dose-dependent increased risk of new-onset acne, with rates of 0.9% in controls and patients on baricitinib 2 mg, climbing to 3.6% with baricitinib 4 mg.
“In phase 2 results with upadacitinib [another oral JAK inhibitor], we saw that more than 10% of patients in the highest-dose group developed what was classified as acne. I cannot explain this, but it’s something we will monitor in the future,” Dr. Reich promised.
A fuller picture of baricitinib’s safety profile in the setting of atopic dermatitis clearly requires larger and longer-term studies, he added.
Baricitinib at the 2 mg daily dose is already marketed as Olumiant for the treatment of rheumatoid arthritis, with labeling that includes a boxed warning about serious infections, malignancy, and thrombosis. The Food and Drug Administration did not approve the 4-mg dose after determining that its higher safety hazard outweighed the efficacy advantage over the lower dose.
The BREEZE-AD7 study was sponsored by Eli Lilly. Dr. Reich reported serving as an adviser to, paid speaker for, and recipient of research grants from that pharmaceutical company and more than two dozen others.
MADRID – Adding the oral Janus kinase (JAK) inhibitor baricitinib to standard atopic dermatitis therapy with low- and midpotency topical corticosteroids markedly improved disease severity and key patient-reported outcomes, compared with topical corticosteroids alone, in the phase 3, randomized, double-blind BREEZE-AD7 trial, Kristian Reich, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
in the phase 3 BREEZE-AD1 and -AD2 trials. But BREEZE-AD7 further advances the field because it’s the first phase 3 study testing the efficacy of a JAK inhibitor in combination with low- and midpotency topical steroids.
“I think this study is important because it looks into the situation that’s more like what happens in the real world, which is, as with dupilumab and other drugs, we use the systemic agent in combination with topical therapies and, in particular, with topical corticosteroids,” commented Dr. Reich, professor of dermatology at University Medical Center, Hamburg, and medical director at SCIderm, a scientific research company.
“This is what I think we can expect from existing and upcoming systemic therapies in atopic dermatitis: We will use them in combination with topical corticosteroids, and hopefully this will allow patients to dramatically reduce the concomitant use of topical corticosteroids, as shown here in BREEZE-AD7,” he added.
BREEZE-AD7 was a 16-week study that included 329 adults with moderate or severe atopic dermatitis who were randomized to low- and midpotency topical corticosteroids plus either baricitinib at 2 mg once daily, baricitinib at 4 mg once daily, or placebo. The group’s mean baseline Eczema Area and Severity Index (EASI) score was 29. Overall, 45% of participants had a baseline Investigator’s Global Assessment (IGA) of disease severity of 4 on a 0-4 scale.
The primary endpoint was achievement of an IGA of 0 or 1, meaning clear or almost clear, along with at least a 2-point IGA improvement from baseline at week 16. This was accomplished in 30.6% of those on 4 mg/day of baricitinib, 23.9% of patients in the 2-mg group, and 14.7% of controls.
The 4-mg dose of baricitinib was statistically superior to placebo; the 2-mg dose was not. However, Dr. Reich indicated he was untroubled by this because the primary endpoint was set at a high bar, and both doses of baricitinib proved to be significantly better than topical steroids plus placebo in terms of EASI 75 response rates, as well as reductions in itch, skin pain, and sleep problems, which aren’t captured in EASI scores (see graphic).
“One of my big learnings from this year’s EADV is that we have to rethink the dimensions of atopic dermatitis. I think we have underestimated the relevance of important symptoms such as itch, the impact atopic dermatitis has on pain, and the effect it has on sleeping problems,” the dermatologist said. “My feeling is that baricitinib is strongest in reducing itch, improving sleep, and reducing pain, but it also has good effects on the clinical signs of atopic dermatitis.”
The baricitinib-treated patients’ rapidity of improvement in the various endpoints was particularly impressive. Both doses of the JAK 1/2 inhibitor showed significant separation from the control group in the first week, and the majority of improvement occurred by week 4.
A key finding was that patients on baricitinib at 2 mg/day and 4 mg/day used a mean total of 162 g and 137 g of midpotency topical steroids, respectively, during the 16 weeks, compared with 225 g in the control group. The higher-dose baricitinib group was topical corticosteroid-free on 33% of study days, compared with 25% of days for the baricitinib 2 mg patients and 17% of days for controls.
In terms of safety, there was a case of pulmonary embolism in the higher-dose baricitinib group and an opportunistic toxoplasmosis eye infection in the control population. The frequency of oral herpes and herpes simplex virus infections was 2.8% in controls, 4.6% in the baricitinib 2-mg group, and 6.3% in the 4-mg group. There was also a signal of a dose-dependent increased risk of new-onset acne, with rates of 0.9% in controls and patients on baricitinib 2 mg, climbing to 3.6% with baricitinib 4 mg.
“In phase 2 results with upadacitinib [another oral JAK inhibitor], we saw that more than 10% of patients in the highest-dose group developed what was classified as acne. I cannot explain this, but it’s something we will monitor in the future,” Dr. Reich promised.
A fuller picture of baricitinib’s safety profile in the setting of atopic dermatitis clearly requires larger and longer-term studies, he added.
Baricitinib at the 2 mg daily dose is already marketed as Olumiant for the treatment of rheumatoid arthritis, with labeling that includes a boxed warning about serious infections, malignancy, and thrombosis. The Food and Drug Administration did not approve the 4-mg dose after determining that its higher safety hazard outweighed the efficacy advantage over the lower dose.
The BREEZE-AD7 study was sponsored by Eli Lilly. Dr. Reich reported serving as an adviser to, paid speaker for, and recipient of research grants from that pharmaceutical company and more than two dozen others.
MADRID – Adding the oral Janus kinase (JAK) inhibitor baricitinib to standard atopic dermatitis therapy with low- and midpotency topical corticosteroids markedly improved disease severity and key patient-reported outcomes, compared with topical corticosteroids alone, in the phase 3, randomized, double-blind BREEZE-AD7 trial, Kristian Reich, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
in the phase 3 BREEZE-AD1 and -AD2 trials. But BREEZE-AD7 further advances the field because it’s the first phase 3 study testing the efficacy of a JAK inhibitor in combination with low- and midpotency topical steroids.
“I think this study is important because it looks into the situation that’s more like what happens in the real world, which is, as with dupilumab and other drugs, we use the systemic agent in combination with topical therapies and, in particular, with topical corticosteroids,” commented Dr. Reich, professor of dermatology at University Medical Center, Hamburg, and medical director at SCIderm, a scientific research company.
“This is what I think we can expect from existing and upcoming systemic therapies in atopic dermatitis: We will use them in combination with topical corticosteroids, and hopefully this will allow patients to dramatically reduce the concomitant use of topical corticosteroids, as shown here in BREEZE-AD7,” he added.
BREEZE-AD7 was a 16-week study that included 329 adults with moderate or severe atopic dermatitis who were randomized to low- and midpotency topical corticosteroids plus either baricitinib at 2 mg once daily, baricitinib at 4 mg once daily, or placebo. The group’s mean baseline Eczema Area and Severity Index (EASI) score was 29. Overall, 45% of participants had a baseline Investigator’s Global Assessment (IGA) of disease severity of 4 on a 0-4 scale.
The primary endpoint was achievement of an IGA of 0 or 1, meaning clear or almost clear, along with at least a 2-point IGA improvement from baseline at week 16. This was accomplished in 30.6% of those on 4 mg/day of baricitinib, 23.9% of patients in the 2-mg group, and 14.7% of controls.
The 4-mg dose of baricitinib was statistically superior to placebo; the 2-mg dose was not. However, Dr. Reich indicated he was untroubled by this because the primary endpoint was set at a high bar, and both doses of baricitinib proved to be significantly better than topical steroids plus placebo in terms of EASI 75 response rates, as well as reductions in itch, skin pain, and sleep problems, which aren’t captured in EASI scores (see graphic).
“One of my big learnings from this year’s EADV is that we have to rethink the dimensions of atopic dermatitis. I think we have underestimated the relevance of important symptoms such as itch, the impact atopic dermatitis has on pain, and the effect it has on sleeping problems,” the dermatologist said. “My feeling is that baricitinib is strongest in reducing itch, improving sleep, and reducing pain, but it also has good effects on the clinical signs of atopic dermatitis.”
The baricitinib-treated patients’ rapidity of improvement in the various endpoints was particularly impressive. Both doses of the JAK 1/2 inhibitor showed significant separation from the control group in the first week, and the majority of improvement occurred by week 4.
A key finding was that patients on baricitinib at 2 mg/day and 4 mg/day used a mean total of 162 g and 137 g of midpotency topical steroids, respectively, during the 16 weeks, compared with 225 g in the control group. The higher-dose baricitinib group was topical corticosteroid-free on 33% of study days, compared with 25% of days for the baricitinib 2 mg patients and 17% of days for controls.
In terms of safety, there was a case of pulmonary embolism in the higher-dose baricitinib group and an opportunistic toxoplasmosis eye infection in the control population. The frequency of oral herpes and herpes simplex virus infections was 2.8% in controls, 4.6% in the baricitinib 2-mg group, and 6.3% in the 4-mg group. There was also a signal of a dose-dependent increased risk of new-onset acne, with rates of 0.9% in controls and patients on baricitinib 2 mg, climbing to 3.6% with baricitinib 4 mg.
“In phase 2 results with upadacitinib [another oral JAK inhibitor], we saw that more than 10% of patients in the highest-dose group developed what was classified as acne. I cannot explain this, but it’s something we will monitor in the future,” Dr. Reich promised.
A fuller picture of baricitinib’s safety profile in the setting of atopic dermatitis clearly requires larger and longer-term studies, he added.
Baricitinib at the 2 mg daily dose is already marketed as Olumiant for the treatment of rheumatoid arthritis, with labeling that includes a boxed warning about serious infections, malignancy, and thrombosis. The Food and Drug Administration did not approve the 4-mg dose after determining that its higher safety hazard outweighed the efficacy advantage over the lower dose.
The BREEZE-AD7 study was sponsored by Eli Lilly. Dr. Reich reported serving as an adviser to, paid speaker for, and recipient of research grants from that pharmaceutical company and more than two dozen others.
REPORTING FROM EADV CONGRESS
Key clinical point: The Janus kinase 1/2 inhibitor baricitinib shows promise as a novel oral treatment for moderate or severe atopic dermatitis.
Major finding: Among atopic dermatitis patients on concomitant topical corticosteroids, a 75% improvement on Eczema Area and Severity Index at 16 weeks was achieved in 48% of those on baricitinib at 4 mg/day, 43% with baricitinib at 2 mg/day, and 23% on placebo.
Study details: BREEZE-AD7 was a phase 3, multicenter, 16-week, double-blind, three-arm study including 329 adults with moderate or severe atopic dermatitis.
Disclosures: The BREEZE-AD7 study was sponsored by Eli Lilly. The presenter reported serving as an adviser to, paid speaker for, and/or recipient of research grants from that pharmaceutical company and more than two dozen others.
Source: Reich K. EADV Congress, late breaker.