User login
Dupilumab approval sought for AD under age 12
LAHAINA, HAWAII – Reassuring evidence of the long-term effectiveness and safety of dupilumab in adolescents with moderate to severe atopic dermatitis comes from a phase 3 open-label extension study of the first teenagers in the world to have received the monoclonal antibody, Lawrence F. Eichenfield, MD, reported at the SDEF Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
Dupilumab (Dupixent), a monoclonal antibody directed against interleukins-4 and -13 initially approved in adults, received an expanded indication from the Food and Drug Administration in March 2019 for treatment of 12- to 17-year-olds with moderate to severe atopic dermatitis (AD) on the strength of a pivotal 251-patient, phase 3 randomized trial of 16 weeks’ duration (JAMA Dermatol. 2019 Nov 6. doi: 10.1001/jamadermatol.2019.3336). But since AD is a chronic disease, it was important to learn how dupilumab performs well beyond the 16-week mark in adolescents, observed Dr. Eichenfield, professor of dermatology and pediatrics at the University of California, San Diego, and chief of pediatric and adolescent dermatology at Rady Children’s Hospital.
In addition to highlighting some of the emerging fine points of dupilumab therapy in adolescents, Dr. Eichenfield discussed the clinical implications of a potential further expanded indication for treatment of 6- to 12-year-olds, an event he considers likely in the coming months. He also described early data from an ongoing dupilumab clinical trials program in the 2- to 5-year-olds.
Long-term dupilumab in teens
Dr. Eichenfield was a coauthor of the recently published phase 3 international long-term extension study. The 40 participants experienced a mean 85% decrease from baseline at 52 weeks in EASI (Eczema Area and Severity Index) scores on 2 mg/kg per week dosing and an 84% reduction on 4 mg/kg per week dosing. This represented a substantial further improvement from week 2, when the EASI reductions were 34% and 51%, respectively.
The mean trough serum dupilumab concentrations over the course of the year were markedly lower in the 2 mg/kg group: 74 mg/L, as compared to 161 mg/L with dosing at 4 mg/kg per week (Br J Dermatol. 2020 Jan;182[1]:85-96).
“It’ll be interesting to see how this works out over time,” the dermatologist commented. “The issue of dose by weight becomes important as we start to treat younger patients because the pharmacokinetics are very different at 4 and 2 mg/kg, and it may have an impact on efficacy.”
The extension study also established the safety and effectiveness of utilizing dupilumab in combination with standard topical corticosteroid therapy, which wasn’t allowed in the pivotal 16-week trial.
Some have commented that dupilumab may be less effective in adolescents than in adults. They point to the 24% rate of an Investigator Global Assessment (IGA) of 0 or 1 – that is, clear or almost clear – at week 16 in the pivotal adolescent trial, a substantially lower rate than in the adult trials. However, Dr. Eichenfield noted that the adolescent study population was heavily skewed to the severe end of the disease spectrum, the placebo response rate was very low, and the absolute placebo-subtracted benefit turned out to be quite similar to what was seen in the adult trials. Moreover, he added, in a post hoc analysis of the pivotal trial data which utilized a different measure of clinically meaningful response – a composite of either a 50% reduction in EASI score, a 3-point or greater improvement on a 10-point pruritus scale, or at least a 6-point improvement from baseline on the Children’s Dermatology Quality Life Index – that outcome was achieved by 74% of adolescents who didn’t achieve clear or almost clear.
What’s next for dupilumab in pediatric AD
Approval of dupilumab in children under aged 12 years is eagerly awaited, Dr. Eichenfield said. The Food and Drug Administration is now analyzing as-yet unreleased data from completed clinical trials of dupilumab in 6- to 12-year-olds with moderate to severe AD with an eye toward a possible further expanded indication. The side effect profile appears to be the same as in 12- to 18-year-olds.
“I assume it will be approved,” Dr. Eichenfield said. “We don’t know what’s going to happen in 6- to 12-year-olds in terms of the ultimate dosing recommendations that will be put out, but be aware that the pharmacokinetics vary by weight over time.”
Early data in children aged 2-5 years with severe AD from the phase 2, open-label, single ascending dose Liberty AD PRESCHOOL study showed that weight-based dosing in that age group made a big difference in terms of pharmacokinetics. In terms of efficacy, the mean reduction in EASI scores 4 weeks after a single dose of dupilumab was 27% with 3 mg/kg and 49% with 6 mg/kg.
Avoidance of live vaccines while on dupilumab becomes more of a consideration in the under-12 population. The second dose of varicella is supposed to be administered at 4 to 6 years of age, as is the second dose of MMR. The nasal influenza vaccine is a live virus vaccine, as is the yellow fever vaccine.
“We don’t know if live vaccines are dangerous for someone on dupilumab, it’s just that it’s listed that you shouldn’t use them and they haven’t been studied,” Dr. Eichenfield observed.
He reported receiving research grants from or serving as a consultant to several dozen pharmaceutical companies.
The SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
LAHAINA, HAWAII – Reassuring evidence of the long-term effectiveness and safety of dupilumab in adolescents with moderate to severe atopic dermatitis comes from a phase 3 open-label extension study of the first teenagers in the world to have received the monoclonal antibody, Lawrence F. Eichenfield, MD, reported at the SDEF Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
Dupilumab (Dupixent), a monoclonal antibody directed against interleukins-4 and -13 initially approved in adults, received an expanded indication from the Food and Drug Administration in March 2019 for treatment of 12- to 17-year-olds with moderate to severe atopic dermatitis (AD) on the strength of a pivotal 251-patient, phase 3 randomized trial of 16 weeks’ duration (JAMA Dermatol. 2019 Nov 6. doi: 10.1001/jamadermatol.2019.3336). But since AD is a chronic disease, it was important to learn how dupilumab performs well beyond the 16-week mark in adolescents, observed Dr. Eichenfield, professor of dermatology and pediatrics at the University of California, San Diego, and chief of pediatric and adolescent dermatology at Rady Children’s Hospital.
In addition to highlighting some of the emerging fine points of dupilumab therapy in adolescents, Dr. Eichenfield discussed the clinical implications of a potential further expanded indication for treatment of 6- to 12-year-olds, an event he considers likely in the coming months. He also described early data from an ongoing dupilumab clinical trials program in the 2- to 5-year-olds.
Long-term dupilumab in teens
Dr. Eichenfield was a coauthor of the recently published phase 3 international long-term extension study. The 40 participants experienced a mean 85% decrease from baseline at 52 weeks in EASI (Eczema Area and Severity Index) scores on 2 mg/kg per week dosing and an 84% reduction on 4 mg/kg per week dosing. This represented a substantial further improvement from week 2, when the EASI reductions were 34% and 51%, respectively.
The mean trough serum dupilumab concentrations over the course of the year were markedly lower in the 2 mg/kg group: 74 mg/L, as compared to 161 mg/L with dosing at 4 mg/kg per week (Br J Dermatol. 2020 Jan;182[1]:85-96).
“It’ll be interesting to see how this works out over time,” the dermatologist commented. “The issue of dose by weight becomes important as we start to treat younger patients because the pharmacokinetics are very different at 4 and 2 mg/kg, and it may have an impact on efficacy.”
The extension study also established the safety and effectiveness of utilizing dupilumab in combination with standard topical corticosteroid therapy, which wasn’t allowed in the pivotal 16-week trial.
Some have commented that dupilumab may be less effective in adolescents than in adults. They point to the 24% rate of an Investigator Global Assessment (IGA) of 0 or 1 – that is, clear or almost clear – at week 16 in the pivotal adolescent trial, a substantially lower rate than in the adult trials. However, Dr. Eichenfield noted that the adolescent study population was heavily skewed to the severe end of the disease spectrum, the placebo response rate was very low, and the absolute placebo-subtracted benefit turned out to be quite similar to what was seen in the adult trials. Moreover, he added, in a post hoc analysis of the pivotal trial data which utilized a different measure of clinically meaningful response – a composite of either a 50% reduction in EASI score, a 3-point or greater improvement on a 10-point pruritus scale, or at least a 6-point improvement from baseline on the Children’s Dermatology Quality Life Index – that outcome was achieved by 74% of adolescents who didn’t achieve clear or almost clear.
What’s next for dupilumab in pediatric AD
Approval of dupilumab in children under aged 12 years is eagerly awaited, Dr. Eichenfield said. The Food and Drug Administration is now analyzing as-yet unreleased data from completed clinical trials of dupilumab in 6- to 12-year-olds with moderate to severe AD with an eye toward a possible further expanded indication. The side effect profile appears to be the same as in 12- to 18-year-olds.
“I assume it will be approved,” Dr. Eichenfield said. “We don’t know what’s going to happen in 6- to 12-year-olds in terms of the ultimate dosing recommendations that will be put out, but be aware that the pharmacokinetics vary by weight over time.”
Early data in children aged 2-5 years with severe AD from the phase 2, open-label, single ascending dose Liberty AD PRESCHOOL study showed that weight-based dosing in that age group made a big difference in terms of pharmacokinetics. In terms of efficacy, the mean reduction in EASI scores 4 weeks after a single dose of dupilumab was 27% with 3 mg/kg and 49% with 6 mg/kg.
Avoidance of live vaccines while on dupilumab becomes more of a consideration in the under-12 population. The second dose of varicella is supposed to be administered at 4 to 6 years of age, as is the second dose of MMR. The nasal influenza vaccine is a live virus vaccine, as is the yellow fever vaccine.
“We don’t know if live vaccines are dangerous for someone on dupilumab, it’s just that it’s listed that you shouldn’t use them and they haven’t been studied,” Dr. Eichenfield observed.
He reported receiving research grants from or serving as a consultant to several dozen pharmaceutical companies.
The SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
LAHAINA, HAWAII – Reassuring evidence of the long-term effectiveness and safety of dupilumab in adolescents with moderate to severe atopic dermatitis comes from a phase 3 open-label extension study of the first teenagers in the world to have received the monoclonal antibody, Lawrence F. Eichenfield, MD, reported at the SDEF Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
Dupilumab (Dupixent), a monoclonal antibody directed against interleukins-4 and -13 initially approved in adults, received an expanded indication from the Food and Drug Administration in March 2019 for treatment of 12- to 17-year-olds with moderate to severe atopic dermatitis (AD) on the strength of a pivotal 251-patient, phase 3 randomized trial of 16 weeks’ duration (JAMA Dermatol. 2019 Nov 6. doi: 10.1001/jamadermatol.2019.3336). But since AD is a chronic disease, it was important to learn how dupilumab performs well beyond the 16-week mark in adolescents, observed Dr. Eichenfield, professor of dermatology and pediatrics at the University of California, San Diego, and chief of pediatric and adolescent dermatology at Rady Children’s Hospital.
In addition to highlighting some of the emerging fine points of dupilumab therapy in adolescents, Dr. Eichenfield discussed the clinical implications of a potential further expanded indication for treatment of 6- to 12-year-olds, an event he considers likely in the coming months. He also described early data from an ongoing dupilumab clinical trials program in the 2- to 5-year-olds.
Long-term dupilumab in teens
Dr. Eichenfield was a coauthor of the recently published phase 3 international long-term extension study. The 40 participants experienced a mean 85% decrease from baseline at 52 weeks in EASI (Eczema Area and Severity Index) scores on 2 mg/kg per week dosing and an 84% reduction on 4 mg/kg per week dosing. This represented a substantial further improvement from week 2, when the EASI reductions were 34% and 51%, respectively.
The mean trough serum dupilumab concentrations over the course of the year were markedly lower in the 2 mg/kg group: 74 mg/L, as compared to 161 mg/L with dosing at 4 mg/kg per week (Br J Dermatol. 2020 Jan;182[1]:85-96).
“It’ll be interesting to see how this works out over time,” the dermatologist commented. “The issue of dose by weight becomes important as we start to treat younger patients because the pharmacokinetics are very different at 4 and 2 mg/kg, and it may have an impact on efficacy.”
The extension study also established the safety and effectiveness of utilizing dupilumab in combination with standard topical corticosteroid therapy, which wasn’t allowed in the pivotal 16-week trial.
Some have commented that dupilumab may be less effective in adolescents than in adults. They point to the 24% rate of an Investigator Global Assessment (IGA) of 0 or 1 – that is, clear or almost clear – at week 16 in the pivotal adolescent trial, a substantially lower rate than in the adult trials. However, Dr. Eichenfield noted that the adolescent study population was heavily skewed to the severe end of the disease spectrum, the placebo response rate was very low, and the absolute placebo-subtracted benefit turned out to be quite similar to what was seen in the adult trials. Moreover, he added, in a post hoc analysis of the pivotal trial data which utilized a different measure of clinically meaningful response – a composite of either a 50% reduction in EASI score, a 3-point or greater improvement on a 10-point pruritus scale, or at least a 6-point improvement from baseline on the Children’s Dermatology Quality Life Index – that outcome was achieved by 74% of adolescents who didn’t achieve clear or almost clear.
What’s next for dupilumab in pediatric AD
Approval of dupilumab in children under aged 12 years is eagerly awaited, Dr. Eichenfield said. The Food and Drug Administration is now analyzing as-yet unreleased data from completed clinical trials of dupilumab in 6- to 12-year-olds with moderate to severe AD with an eye toward a possible further expanded indication. The side effect profile appears to be the same as in 12- to 18-year-olds.
“I assume it will be approved,” Dr. Eichenfield said. “We don’t know what’s going to happen in 6- to 12-year-olds in terms of the ultimate dosing recommendations that will be put out, but be aware that the pharmacokinetics vary by weight over time.”
Early data in children aged 2-5 years with severe AD from the phase 2, open-label, single ascending dose Liberty AD PRESCHOOL study showed that weight-based dosing in that age group made a big difference in terms of pharmacokinetics. In terms of efficacy, the mean reduction in EASI scores 4 weeks after a single dose of dupilumab was 27% with 3 mg/kg and 49% with 6 mg/kg.
Avoidance of live vaccines while on dupilumab becomes more of a consideration in the under-12 population. The second dose of varicella is supposed to be administered at 4 to 6 years of age, as is the second dose of MMR. The nasal influenza vaccine is a live virus vaccine, as is the yellow fever vaccine.
“We don’t know if live vaccines are dangerous for someone on dupilumab, it’s just that it’s listed that you shouldn’t use them and they haven’t been studied,” Dr. Eichenfield observed.
He reported receiving research grants from or serving as a consultant to several dozen pharmaceutical companies.
The SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
REPORTING FROM SDEF HAWAII DERMATOLOGY SEMINAR
A 7-month-old male presents with perioral rash and fever
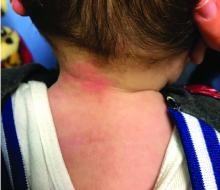
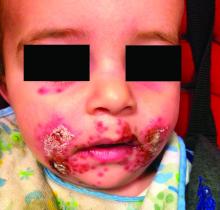
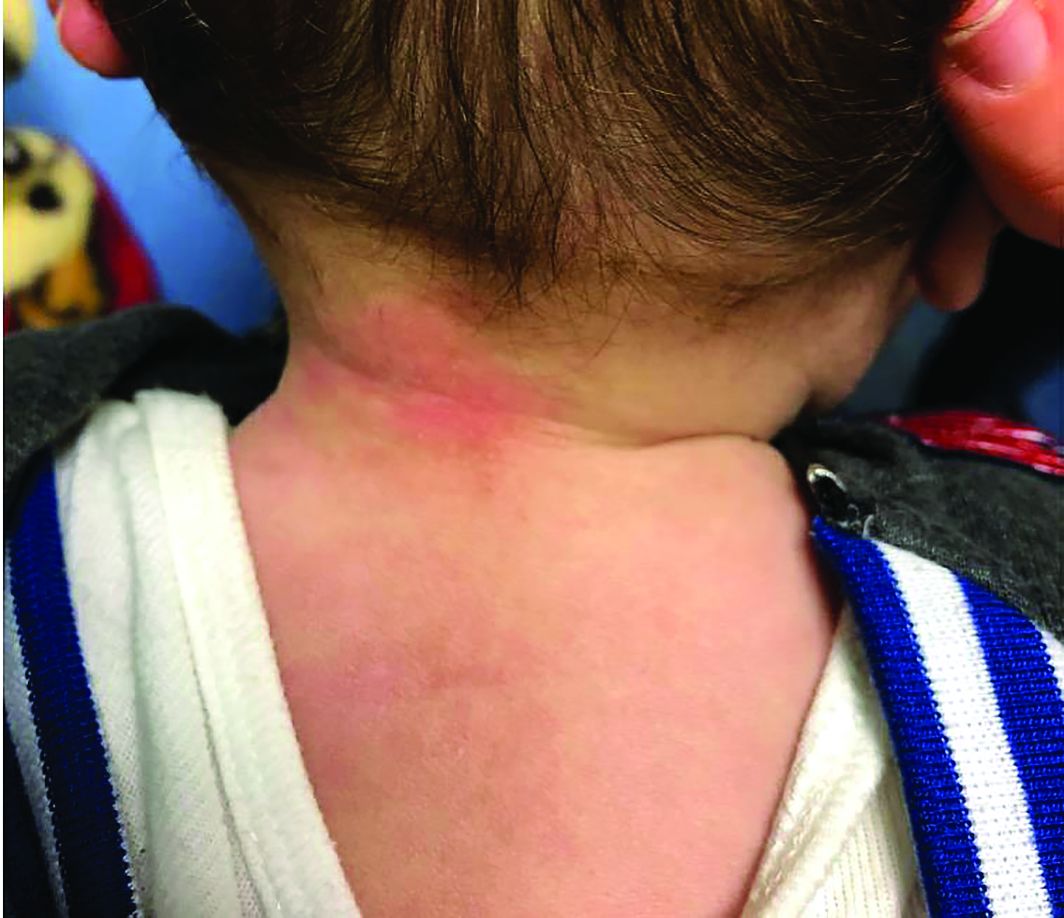
Patients with atopic dermatitis are at risk for developing the herpes simplex virus (HSV)–related skin complication “eczema herpeticum,” also known as Kaposi’s varicelliform eruption. Eczema herpeticum is characterized by cutaneous pain and vesicular skin lesions, most commonly secondary to infection with HSV-1. The condition may affect individuals with atopic dermatitis or other inflammatory skin disorders. Eczema herpeticum develops when the virus infects large areas of skin, rather than being confined to a small area as in the common cold sore. Eczema herpeticum often appears on the face and neck, although it can appear anywhere on the body. In some cases, the rash may be difficult to distinguish from a patient’s baseline eczema if the latter is poorly controlled. Skin symptoms of eczema herpeticum include clusters of small blisters that are itchy and painful; vesicles that appear red, purple, or black; purulent blisters; or crusting. Classically, the morphology of vesicles or crusted lesions shows a “cluster of grapes” appearance. Eczema herpeticum may present with a high fever, chills, and swollen lymph glands.
While a clinical diagnosis based on the history, physical findings, and morphologic appearance of the rash is reasonable, testing may confirm the diagnosis. The most sensitive and specific tests are polymerase chain reaction sequencing for HSV, direct fluorescent antibody stain, and/or viral culture, while Tzanck smear may show characteristic histologic changes. Treatment is with oral antiviral therapy and treatment of the eczema.
Hand, foot, and mouth disease (HFMD) is a common viral illness usually affecting infants and children. The infection often involves the hands, feet, mouth, and sometimes, the genitals and buttocks. The viral exanthem is most commonly caused by the coxsackievirus, of the enterovirus family. Coxsackievirus A16 and enterovirus A71 are the serotypes that are most commonly implicated as the causative agents. HFMD initially presents with a low-grade fever, reduced appetite, and general malaise. About 1-2 days later, the child may develop painful mouth sores with an exanthem that involves the dorsum of the hands, soles of the feet, buttocks, legs, and arms. The exanthem consists of vesicles surrounded by a thin halo of erythema, eventually rupturing and forming superficial ulcers with a gray-yellow base and erythematous rim. The exanthem is itchy, and can be macular, papular, or vesicular. The lesions are nonpruritic, and typically not painful. The diagnosis of HFMD usually is made clinically, although a physician can swab the mouth or get a stool sample for polymerase chain reaction, which will show the virus; treatment is supportive. In children with atopic dermatitis, lesions also can tend to concentrate in areas previously or currently affected by the dermatitis, similar to eczema herpeticum, and the terms eczema coxsackium or atypical HFMD are applicable. In young adults, the disease may present with erythematous papulovesicular lesions on the face, oral mucosa, extensor surfaces of the upper and lower extremities, and palms and soles; confluent, hemorrhagic, and crusted lesions also can be seen on the extremities. Systemic symptoms usually subside in a few days; the skin lesions resolve without scarring in days to weeks.
Secondary bacterial infection is not uncommon in eczema herpeticum patients, reflecting common Staphylococcus aureus infection in atopic dermatitis patients. Streptococcus also may be seen as a concurrent infection. Treatment of secondary bacterial infection may be considered based on clinic context and culture.
Impetiginized eczema also is in the differential diagnosis of eczema herpeticum. S. aureus and Streptococci are the most important causative organisms. Lesions can manifest as a single red papule or macule that quickly becomes vesicular or eroded. Subsequently, the content dries, forming honey-colored crusts. Impetigo may resolve spontaneously, although in the context of infected eczema both topical anti-inflammatory agents (e.g. topical corticosteroids) along with systemic antibiotics may be a reasonable treatment option. Although our patient had honey-colored crusting, the wound culture showed normal bacterial flora.
Primary varicella infection causes acute fever and rash, with an initial exanthem of disseminated pruritic erythematous macules that progress beyond the papular stage, forming clear, fluid-filled vesicles (like dewdrops on a rose petal). In children, the rash presents on the stomach, back, and face, and then spreads to other parts of the body. Blisters also can arise inside the mouth.
In this patient, perioral HSV PCR 1 was positive, and wound culture showed normal oral flora with no organisms or white blood cells seen. The patient responded well to oral acyclovir, and treatment of his underlying atopic dermatitis with low-potency topical corticosteroids.
Dr. Bhatti is a research fellow in pediatric dermatology at Rady Children’s Hospital and the University of California, San Diego. Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children’s Hospital–San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. Neither of the physicians had relevant financial disclosures. Email them at pdnews@mdedge.com.
Sources
Can Fam Physician. 2012 Dec;58(12):1358-61.
William L Weston, MD., William Howe, MD. UpToDate. Treatment of atopic dermatitis (eczema).
Christine Johnson, MD, Anna Wald, MD, MPH. UpToDate. Epidemiology, clinical manifestations, and diagnosis of herpes simplex virus type 1 infection.
Robert Sidbury, MD, MPH. UpToDate. Atypical exanthems in children.
National Eczema Association. Eczema herpeticum.
Centers for Disease Control and Prevention. Symptoms and diagnosis of hand, foot, and mouth disease (HFMD).
Patients with atopic dermatitis are at risk for developing the herpes simplex virus (HSV)–related skin complication “eczema herpeticum,” also known as Kaposi’s varicelliform eruption. Eczema herpeticum is characterized by cutaneous pain and vesicular skin lesions, most commonly secondary to infection with HSV-1. The condition may affect individuals with atopic dermatitis or other inflammatory skin disorders. Eczema herpeticum develops when the virus infects large areas of skin, rather than being confined to a small area as in the common cold sore. Eczema herpeticum often appears on the face and neck, although it can appear anywhere on the body. In some cases, the rash may be difficult to distinguish from a patient’s baseline eczema if the latter is poorly controlled. Skin symptoms of eczema herpeticum include clusters of small blisters that are itchy and painful; vesicles that appear red, purple, or black; purulent blisters; or crusting. Classically, the morphology of vesicles or crusted lesions shows a “cluster of grapes” appearance. Eczema herpeticum may present with a high fever, chills, and swollen lymph glands.
While a clinical diagnosis based on the history, physical findings, and morphologic appearance of the rash is reasonable, testing may confirm the diagnosis. The most sensitive and specific tests are polymerase chain reaction sequencing for HSV, direct fluorescent antibody stain, and/or viral culture, while Tzanck smear may show characteristic histologic changes. Treatment is with oral antiviral therapy and treatment of the eczema.
Hand, foot, and mouth disease (HFMD) is a common viral illness usually affecting infants and children. The infection often involves the hands, feet, mouth, and sometimes, the genitals and buttocks. The viral exanthem is most commonly caused by the coxsackievirus, of the enterovirus family. Coxsackievirus A16 and enterovirus A71 are the serotypes that are most commonly implicated as the causative agents. HFMD initially presents with a low-grade fever, reduced appetite, and general malaise. About 1-2 days later, the child may develop painful mouth sores with an exanthem that involves the dorsum of the hands, soles of the feet, buttocks, legs, and arms. The exanthem consists of vesicles surrounded by a thin halo of erythema, eventually rupturing and forming superficial ulcers with a gray-yellow base and erythematous rim. The exanthem is itchy, and can be macular, papular, or vesicular. The lesions are nonpruritic, and typically not painful. The diagnosis of HFMD usually is made clinically, although a physician can swab the mouth or get a stool sample for polymerase chain reaction, which will show the virus; treatment is supportive. In children with atopic dermatitis, lesions also can tend to concentrate in areas previously or currently affected by the dermatitis, similar to eczema herpeticum, and the terms eczema coxsackium or atypical HFMD are applicable. In young adults, the disease may present with erythematous papulovesicular lesions on the face, oral mucosa, extensor surfaces of the upper and lower extremities, and palms and soles; confluent, hemorrhagic, and crusted lesions also can be seen on the extremities. Systemic symptoms usually subside in a few days; the skin lesions resolve without scarring in days to weeks.
Secondary bacterial infection is not uncommon in eczema herpeticum patients, reflecting common Staphylococcus aureus infection in atopic dermatitis patients. Streptococcus also may be seen as a concurrent infection. Treatment of secondary bacterial infection may be considered based on clinic context and culture.
Impetiginized eczema also is in the differential diagnosis of eczema herpeticum. S. aureus and Streptococci are the most important causative organisms. Lesions can manifest as a single red papule or macule that quickly becomes vesicular or eroded. Subsequently, the content dries, forming honey-colored crusts. Impetigo may resolve spontaneously, although in the context of infected eczema both topical anti-inflammatory agents (e.g. topical corticosteroids) along with systemic antibiotics may be a reasonable treatment option. Although our patient had honey-colored crusting, the wound culture showed normal bacterial flora.
Primary varicella infection causes acute fever and rash, with an initial exanthem of disseminated pruritic erythematous macules that progress beyond the papular stage, forming clear, fluid-filled vesicles (like dewdrops on a rose petal). In children, the rash presents on the stomach, back, and face, and then spreads to other parts of the body. Blisters also can arise inside the mouth.
In this patient, perioral HSV PCR 1 was positive, and wound culture showed normal oral flora with no organisms or white blood cells seen. The patient responded well to oral acyclovir, and treatment of his underlying atopic dermatitis with low-potency topical corticosteroids.
Dr. Bhatti is a research fellow in pediatric dermatology at Rady Children’s Hospital and the University of California, San Diego. Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children’s Hospital–San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. Neither of the physicians had relevant financial disclosures. Email them at pdnews@mdedge.com.
Sources
Can Fam Physician. 2012 Dec;58(12):1358-61.
William L Weston, MD., William Howe, MD. UpToDate. Treatment of atopic dermatitis (eczema).
Christine Johnson, MD, Anna Wald, MD, MPH. UpToDate. Epidemiology, clinical manifestations, and diagnosis of herpes simplex virus type 1 infection.
Robert Sidbury, MD, MPH. UpToDate. Atypical exanthems in children.
National Eczema Association. Eczema herpeticum.
Centers for Disease Control and Prevention. Symptoms and diagnosis of hand, foot, and mouth disease (HFMD).
Patients with atopic dermatitis are at risk for developing the herpes simplex virus (HSV)–related skin complication “eczema herpeticum,” also known as Kaposi’s varicelliform eruption. Eczema herpeticum is characterized by cutaneous pain and vesicular skin lesions, most commonly secondary to infection with HSV-1. The condition may affect individuals with atopic dermatitis or other inflammatory skin disorders. Eczema herpeticum develops when the virus infects large areas of skin, rather than being confined to a small area as in the common cold sore. Eczema herpeticum often appears on the face and neck, although it can appear anywhere on the body. In some cases, the rash may be difficult to distinguish from a patient’s baseline eczema if the latter is poorly controlled. Skin symptoms of eczema herpeticum include clusters of small blisters that are itchy and painful; vesicles that appear red, purple, or black; purulent blisters; or crusting. Classically, the morphology of vesicles or crusted lesions shows a “cluster of grapes” appearance. Eczema herpeticum may present with a high fever, chills, and swollen lymph glands.
While a clinical diagnosis based on the history, physical findings, and morphologic appearance of the rash is reasonable, testing may confirm the diagnosis. The most sensitive and specific tests are polymerase chain reaction sequencing for HSV, direct fluorescent antibody stain, and/or viral culture, while Tzanck smear may show characteristic histologic changes. Treatment is with oral antiviral therapy and treatment of the eczema.
Hand, foot, and mouth disease (HFMD) is a common viral illness usually affecting infants and children. The infection often involves the hands, feet, mouth, and sometimes, the genitals and buttocks. The viral exanthem is most commonly caused by the coxsackievirus, of the enterovirus family. Coxsackievirus A16 and enterovirus A71 are the serotypes that are most commonly implicated as the causative agents. HFMD initially presents with a low-grade fever, reduced appetite, and general malaise. About 1-2 days later, the child may develop painful mouth sores with an exanthem that involves the dorsum of the hands, soles of the feet, buttocks, legs, and arms. The exanthem consists of vesicles surrounded by a thin halo of erythema, eventually rupturing and forming superficial ulcers with a gray-yellow base and erythematous rim. The exanthem is itchy, and can be macular, papular, or vesicular. The lesions are nonpruritic, and typically not painful. The diagnosis of HFMD usually is made clinically, although a physician can swab the mouth or get a stool sample for polymerase chain reaction, which will show the virus; treatment is supportive. In children with atopic dermatitis, lesions also can tend to concentrate in areas previously or currently affected by the dermatitis, similar to eczema herpeticum, and the terms eczema coxsackium or atypical HFMD are applicable. In young adults, the disease may present with erythematous papulovesicular lesions on the face, oral mucosa, extensor surfaces of the upper and lower extremities, and palms and soles; confluent, hemorrhagic, and crusted lesions also can be seen on the extremities. Systemic symptoms usually subside in a few days; the skin lesions resolve without scarring in days to weeks.
Secondary bacterial infection is not uncommon in eczema herpeticum patients, reflecting common Staphylococcus aureus infection in atopic dermatitis patients. Streptococcus also may be seen as a concurrent infection. Treatment of secondary bacterial infection may be considered based on clinic context and culture.
Impetiginized eczema also is in the differential diagnosis of eczema herpeticum. S. aureus and Streptococci are the most important causative organisms. Lesions can manifest as a single red papule or macule that quickly becomes vesicular or eroded. Subsequently, the content dries, forming honey-colored crusts. Impetigo may resolve spontaneously, although in the context of infected eczema both topical anti-inflammatory agents (e.g. topical corticosteroids) along with systemic antibiotics may be a reasonable treatment option. Although our patient had honey-colored crusting, the wound culture showed normal bacterial flora.
Primary varicella infection causes acute fever and rash, with an initial exanthem of disseminated pruritic erythematous macules that progress beyond the papular stage, forming clear, fluid-filled vesicles (like dewdrops on a rose petal). In children, the rash presents on the stomach, back, and face, and then spreads to other parts of the body. Blisters also can arise inside the mouth.
In this patient, perioral HSV PCR 1 was positive, and wound culture showed normal oral flora with no organisms or white blood cells seen. The patient responded well to oral acyclovir, and treatment of his underlying atopic dermatitis with low-potency topical corticosteroids.
Dr. Bhatti is a research fellow in pediatric dermatology at Rady Children’s Hospital and the University of California, San Diego. Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children’s Hospital–San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. Neither of the physicians had relevant financial disclosures. Email them at pdnews@mdedge.com.
Sources
Can Fam Physician. 2012 Dec;58(12):1358-61.
William L Weston, MD., William Howe, MD. UpToDate. Treatment of atopic dermatitis (eczema).
Christine Johnson, MD, Anna Wald, MD, MPH. UpToDate. Epidemiology, clinical manifestations, and diagnosis of herpes simplex virus type 1 infection.
Robert Sidbury, MD, MPH. UpToDate. Atypical exanthems in children.
National Eczema Association. Eczema herpeticum.
Centers for Disease Control and Prevention. Symptoms and diagnosis of hand, foot, and mouth disease (HFMD).
Emollients didn’t prevent atopic dermatitis in high-risk infants
The use of including those at high risk, in two new clinical trials.
The BEEP (Barrier Enhancement for Eczema Prevention) study compared the rates of AD among infants identified as at risk of AD because of family history who had daily applications of emollients (Diprobase cream or Doublebase gel) for the first year of life, compared with a standard skin care group. PreventADALL (Preventing Atopic Dermatitis and Allergies in Children) is a randomized, primary-prevention study conducted in Norway and Sweden that randomized infants into one of four groups: controls whose parents followed regular skin care advice and nutrition guidelines; those who received skin emollients (the addition of emulsified oil to their bath and application of facial cream on at least 4 days a week from age 2 weeks to 8 months); those who received early complementary feeding of peanut, cow’s milk, wheat, and egg introduced between aged 12 and 16 weeks; and a group that combined both the emollient and diet interventions.
Neither of the studies, published in the Lancet, found statistically significant differences in AD rates between the intervention and control groups.
The results put a damper on hopes raised by previous studies that included two small pilot studies, which found that daily use of leave-on emollients in infants considered at high risk of AD prevented the development of AD (J Allergy Clin Immunol 2014 Oct;134:824-30.e6; J Allergy Clin Immunol Oct 2014;134:818-23).
“It was maybe a little bit overly hopeful to think that we could just moisturize and prevent such a complex disorder,” Robert Sidbury, MD, chief of dermatology at Seattle Children’s Hospital, said in an interview. He emphasized that the studies only addressed emollients as a preventative, and that “there’s no question that emollients are still critical for the therapy of eczema.”
Bruce Brod, MD, clinical professor of dermatology at the University of Pennsylvania, Philadelphia, suggested that homogeneous patient populations or insufficient numbers might explain the negative findings. PreventADALL drew patients from Norway and Sweden, while BEEP recruited from the United Kingdom. “They’re important studies, but I think they still lend themselves to further studies with different patient populations and larger groups of patients,” Dr. Brod said in an interview.
BEEP was headed by Joanne Chalmers, PhD, and Hywel Williams, DSc, of the Centre of Evidence-Based Dermatology at the University of Nottingham (England). Håvard Ove Skjerven, PhD, and Karin C Lødrup Carlsen, PhD, of Oslo University Hospital led the PreventADALL study.
The BEEP study randomized 1,394 newborns at 16 sites in the United Kingdom to daily emollient treatment with standard skin care, or standard skin care alone. At one year, compliance was 74% in the intervention group. At age 2, 23% of the intervention group had AD, compared with 25% of controls (hazard ratio, 0.95; P =.61). Skin infections were also higher in the treatment arm (mean, 0.23 per year vs. 0.15 per year; adjusted incidence ratio, 1.55; 95% confidence interval, 1.15-2.09).
“Our study does not support the use of emollients for preventing eczema in high-risk infants, a finding supported by PreventADALL, another large trial using a skin barrier enhancing intervention,” they concluded. Their data “relate only to prevention of eczema and do not directly challenge the practice of using emollients as first-line treatment for eczema.”
In the PreventADALL study, 2,397 newborn infants born between 2015 and 2017 were randomized to one of the four groups. Use of facial cream and emollients during bathing began at 2 weeks, and early complementary feeding of peanut, cow’s milk, wheat, and egg at 3-4 months. The frequency of AD at aged 12 months in the control group was 8%, compared with 11% in the skin-intervention group, 9% in the food-intervention group, and 5% in the combined-intervention group.
These differences were not statistically significant, and “the primary hypothesis that either skin intervention or food intervention reduced atopic dermatitis were not confirmed,” the authors wrote. Parental atopy did not influence the effects of the interventions. Their results were in line with the BEEP results, and the authors “cannot recommend these interventions as primary prevention strategies.”
The researchers will continue to follow children until age 3 years to evaluate the food allergy rates, if the combined-treatment group experiences a long-term benefit. Adherence to the protocol was poor, with 44% compliance with the facial cream application and 27% compliance with bathing emollients; 32% fully adhered to the diet protocols.
The studies were funded by the National Institute for Health Research Health Technology Assessment (BEEP); and a range of public and private funders (PreventADALL). One author of the PreventADALL study disclosed receiving honoraria for presentations from several pharmaceutical companies, and one author received honoraria for presentations from Thermo Fisher Scientific; the rest had no disclosures. Dr. Sidbury has been an investigator for Regeneron. Dr. Brod had no relevant financial disclosures.
SOURCES: Chalmers JR et al. Lancet. 2020 Feb 19. doi: 10.1016/S0140-6736(19)32984-8; Skjerven HO et al. Lancet. 2020 Feb 19. doi: 10.1016/S0140-6736(19)32983-6.
The “null findings” of these two studies were “unexpected,” Kirsten P. Perrett, MBBS, Phd, and Rachel L. Peters, PhD, of the department of population allergy at Murdoch Children’s Research Institute, Parkville, Australia, wrote in an accompanying editorial. They noted that emollients are used regularly in the management of atopic dermatitis, where they help maintain the skin barrier and reduce the need for anti-inflammatory therapies.
These two large prevention studies were “prompted” by the results of small, proof-of-concept pilot studies, which “provided strong efficacy signals for the hypothesis that daily emollient use could prevent atopic dermatitis,” they wrote. But the two studies “found no evidence that daily emollient use in either a population-based or high-risk cohort of infants during the first year of life could delay, suppress, or prevent atopic dermatitis.” The lower incidence of atopic dermatitis among those in the dietary and emollient combination, compared with controls (5% vs. 8%) in PreventADALL, could be a chance finding.
The large, randomized Prevention of Eczema by a Barrier Lipid Equilibrium Strategy (PEBBLES) trial is ongoing to confirm results from a small study suggesting the efficacy of a ceramide-dominant emollient. But the PreventADALL study showed low compliance, suggesting that this intervention, if effective, a twice-daily emollient regimen may be tough to implement. “At this stage, emollients should not be recommended for the primary prevention of atopic dermatitis in infants,” they concluded.
Dr. Perrett and Dr. Peters declared no competing interests. Their comments appeared in the Lancet (2020 Feb 19. doi: 10.1016/S0140-6736[19]33174-5).
The “null findings” of these two studies were “unexpected,” Kirsten P. Perrett, MBBS, Phd, and Rachel L. Peters, PhD, of the department of population allergy at Murdoch Children’s Research Institute, Parkville, Australia, wrote in an accompanying editorial. They noted that emollients are used regularly in the management of atopic dermatitis, where they help maintain the skin barrier and reduce the need for anti-inflammatory therapies.
These two large prevention studies were “prompted” by the results of small, proof-of-concept pilot studies, which “provided strong efficacy signals for the hypothesis that daily emollient use could prevent atopic dermatitis,” they wrote. But the two studies “found no evidence that daily emollient use in either a population-based or high-risk cohort of infants during the first year of life could delay, suppress, or prevent atopic dermatitis.” The lower incidence of atopic dermatitis among those in the dietary and emollient combination, compared with controls (5% vs. 8%) in PreventADALL, could be a chance finding.
The large, randomized Prevention of Eczema by a Barrier Lipid Equilibrium Strategy (PEBBLES) trial is ongoing to confirm results from a small study suggesting the efficacy of a ceramide-dominant emollient. But the PreventADALL study showed low compliance, suggesting that this intervention, if effective, a twice-daily emollient regimen may be tough to implement. “At this stage, emollients should not be recommended for the primary prevention of atopic dermatitis in infants,” they concluded.
Dr. Perrett and Dr. Peters declared no competing interests. Their comments appeared in the Lancet (2020 Feb 19. doi: 10.1016/S0140-6736[19]33174-5).
The “null findings” of these two studies were “unexpected,” Kirsten P. Perrett, MBBS, Phd, and Rachel L. Peters, PhD, of the department of population allergy at Murdoch Children’s Research Institute, Parkville, Australia, wrote in an accompanying editorial. They noted that emollients are used regularly in the management of atopic dermatitis, where they help maintain the skin barrier and reduce the need for anti-inflammatory therapies.
These two large prevention studies were “prompted” by the results of small, proof-of-concept pilot studies, which “provided strong efficacy signals for the hypothesis that daily emollient use could prevent atopic dermatitis,” they wrote. But the two studies “found no evidence that daily emollient use in either a population-based or high-risk cohort of infants during the first year of life could delay, suppress, or prevent atopic dermatitis.” The lower incidence of atopic dermatitis among those in the dietary and emollient combination, compared with controls (5% vs. 8%) in PreventADALL, could be a chance finding.
The large, randomized Prevention of Eczema by a Barrier Lipid Equilibrium Strategy (PEBBLES) trial is ongoing to confirm results from a small study suggesting the efficacy of a ceramide-dominant emollient. But the PreventADALL study showed low compliance, suggesting that this intervention, if effective, a twice-daily emollient regimen may be tough to implement. “At this stage, emollients should not be recommended for the primary prevention of atopic dermatitis in infants,” they concluded.
Dr. Perrett and Dr. Peters declared no competing interests. Their comments appeared in the Lancet (2020 Feb 19. doi: 10.1016/S0140-6736[19]33174-5).
The use of including those at high risk, in two new clinical trials.
The BEEP (Barrier Enhancement for Eczema Prevention) study compared the rates of AD among infants identified as at risk of AD because of family history who had daily applications of emollients (Diprobase cream or Doublebase gel) for the first year of life, compared with a standard skin care group. PreventADALL (Preventing Atopic Dermatitis and Allergies in Children) is a randomized, primary-prevention study conducted in Norway and Sweden that randomized infants into one of four groups: controls whose parents followed regular skin care advice and nutrition guidelines; those who received skin emollients (the addition of emulsified oil to their bath and application of facial cream on at least 4 days a week from age 2 weeks to 8 months); those who received early complementary feeding of peanut, cow’s milk, wheat, and egg introduced between aged 12 and 16 weeks; and a group that combined both the emollient and diet interventions.
Neither of the studies, published in the Lancet, found statistically significant differences in AD rates between the intervention and control groups.
The results put a damper on hopes raised by previous studies that included two small pilot studies, which found that daily use of leave-on emollients in infants considered at high risk of AD prevented the development of AD (J Allergy Clin Immunol 2014 Oct;134:824-30.e6; J Allergy Clin Immunol Oct 2014;134:818-23).
“It was maybe a little bit overly hopeful to think that we could just moisturize and prevent such a complex disorder,” Robert Sidbury, MD, chief of dermatology at Seattle Children’s Hospital, said in an interview. He emphasized that the studies only addressed emollients as a preventative, and that “there’s no question that emollients are still critical for the therapy of eczema.”
Bruce Brod, MD, clinical professor of dermatology at the University of Pennsylvania, Philadelphia, suggested that homogeneous patient populations or insufficient numbers might explain the negative findings. PreventADALL drew patients from Norway and Sweden, while BEEP recruited from the United Kingdom. “They’re important studies, but I think they still lend themselves to further studies with different patient populations and larger groups of patients,” Dr. Brod said in an interview.
BEEP was headed by Joanne Chalmers, PhD, and Hywel Williams, DSc, of the Centre of Evidence-Based Dermatology at the University of Nottingham (England). Håvard Ove Skjerven, PhD, and Karin C Lødrup Carlsen, PhD, of Oslo University Hospital led the PreventADALL study.
The BEEP study randomized 1,394 newborns at 16 sites in the United Kingdom to daily emollient treatment with standard skin care, or standard skin care alone. At one year, compliance was 74% in the intervention group. At age 2, 23% of the intervention group had AD, compared with 25% of controls (hazard ratio, 0.95; P =.61). Skin infections were also higher in the treatment arm (mean, 0.23 per year vs. 0.15 per year; adjusted incidence ratio, 1.55; 95% confidence interval, 1.15-2.09).
“Our study does not support the use of emollients for preventing eczema in high-risk infants, a finding supported by PreventADALL, another large trial using a skin barrier enhancing intervention,” they concluded. Their data “relate only to prevention of eczema and do not directly challenge the practice of using emollients as first-line treatment for eczema.”
In the PreventADALL study, 2,397 newborn infants born between 2015 and 2017 were randomized to one of the four groups. Use of facial cream and emollients during bathing began at 2 weeks, and early complementary feeding of peanut, cow’s milk, wheat, and egg at 3-4 months. The frequency of AD at aged 12 months in the control group was 8%, compared with 11% in the skin-intervention group, 9% in the food-intervention group, and 5% in the combined-intervention group.
These differences were not statistically significant, and “the primary hypothesis that either skin intervention or food intervention reduced atopic dermatitis were not confirmed,” the authors wrote. Parental atopy did not influence the effects of the interventions. Their results were in line with the BEEP results, and the authors “cannot recommend these interventions as primary prevention strategies.”
The researchers will continue to follow children until age 3 years to evaluate the food allergy rates, if the combined-treatment group experiences a long-term benefit. Adherence to the protocol was poor, with 44% compliance with the facial cream application and 27% compliance with bathing emollients; 32% fully adhered to the diet protocols.
The studies were funded by the National Institute for Health Research Health Technology Assessment (BEEP); and a range of public and private funders (PreventADALL). One author of the PreventADALL study disclosed receiving honoraria for presentations from several pharmaceutical companies, and one author received honoraria for presentations from Thermo Fisher Scientific; the rest had no disclosures. Dr. Sidbury has been an investigator for Regeneron. Dr. Brod had no relevant financial disclosures.
SOURCES: Chalmers JR et al. Lancet. 2020 Feb 19. doi: 10.1016/S0140-6736(19)32984-8; Skjerven HO et al. Lancet. 2020 Feb 19. doi: 10.1016/S0140-6736(19)32983-6.
The use of including those at high risk, in two new clinical trials.
The BEEP (Barrier Enhancement for Eczema Prevention) study compared the rates of AD among infants identified as at risk of AD because of family history who had daily applications of emollients (Diprobase cream or Doublebase gel) for the first year of life, compared with a standard skin care group. PreventADALL (Preventing Atopic Dermatitis and Allergies in Children) is a randomized, primary-prevention study conducted in Norway and Sweden that randomized infants into one of four groups: controls whose parents followed regular skin care advice and nutrition guidelines; those who received skin emollients (the addition of emulsified oil to their bath and application of facial cream on at least 4 days a week from age 2 weeks to 8 months); those who received early complementary feeding of peanut, cow’s milk, wheat, and egg introduced between aged 12 and 16 weeks; and a group that combined both the emollient and diet interventions.
Neither of the studies, published in the Lancet, found statistically significant differences in AD rates between the intervention and control groups.
The results put a damper on hopes raised by previous studies that included two small pilot studies, which found that daily use of leave-on emollients in infants considered at high risk of AD prevented the development of AD (J Allergy Clin Immunol 2014 Oct;134:824-30.e6; J Allergy Clin Immunol Oct 2014;134:818-23).
“It was maybe a little bit overly hopeful to think that we could just moisturize and prevent such a complex disorder,” Robert Sidbury, MD, chief of dermatology at Seattle Children’s Hospital, said in an interview. He emphasized that the studies only addressed emollients as a preventative, and that “there’s no question that emollients are still critical for the therapy of eczema.”
Bruce Brod, MD, clinical professor of dermatology at the University of Pennsylvania, Philadelphia, suggested that homogeneous patient populations or insufficient numbers might explain the negative findings. PreventADALL drew patients from Norway and Sweden, while BEEP recruited from the United Kingdom. “They’re important studies, but I think they still lend themselves to further studies with different patient populations and larger groups of patients,” Dr. Brod said in an interview.
BEEP was headed by Joanne Chalmers, PhD, and Hywel Williams, DSc, of the Centre of Evidence-Based Dermatology at the University of Nottingham (England). Håvard Ove Skjerven, PhD, and Karin C Lødrup Carlsen, PhD, of Oslo University Hospital led the PreventADALL study.
The BEEP study randomized 1,394 newborns at 16 sites in the United Kingdom to daily emollient treatment with standard skin care, or standard skin care alone. At one year, compliance was 74% in the intervention group. At age 2, 23% of the intervention group had AD, compared with 25% of controls (hazard ratio, 0.95; P =.61). Skin infections were also higher in the treatment arm (mean, 0.23 per year vs. 0.15 per year; adjusted incidence ratio, 1.55; 95% confidence interval, 1.15-2.09).
“Our study does not support the use of emollients for preventing eczema in high-risk infants, a finding supported by PreventADALL, another large trial using a skin barrier enhancing intervention,” they concluded. Their data “relate only to prevention of eczema and do not directly challenge the practice of using emollients as first-line treatment for eczema.”
In the PreventADALL study, 2,397 newborn infants born between 2015 and 2017 were randomized to one of the four groups. Use of facial cream and emollients during bathing began at 2 weeks, and early complementary feeding of peanut, cow’s milk, wheat, and egg at 3-4 months. The frequency of AD at aged 12 months in the control group was 8%, compared with 11% in the skin-intervention group, 9% in the food-intervention group, and 5% in the combined-intervention group.
These differences were not statistically significant, and “the primary hypothesis that either skin intervention or food intervention reduced atopic dermatitis were not confirmed,” the authors wrote. Parental atopy did not influence the effects of the interventions. Their results were in line with the BEEP results, and the authors “cannot recommend these interventions as primary prevention strategies.”
The researchers will continue to follow children until age 3 years to evaluate the food allergy rates, if the combined-treatment group experiences a long-term benefit. Adherence to the protocol was poor, with 44% compliance with the facial cream application and 27% compliance with bathing emollients; 32% fully adhered to the diet protocols.
The studies were funded by the National Institute for Health Research Health Technology Assessment (BEEP); and a range of public and private funders (PreventADALL). One author of the PreventADALL study disclosed receiving honoraria for presentations from several pharmaceutical companies, and one author received honoraria for presentations from Thermo Fisher Scientific; the rest had no disclosures. Dr. Sidbury has been an investigator for Regeneron. Dr. Brod had no relevant financial disclosures.
SOURCES: Chalmers JR et al. Lancet. 2020 Feb 19. doi: 10.1016/S0140-6736(19)32984-8; Skjerven HO et al. Lancet. 2020 Feb 19. doi: 10.1016/S0140-6736(19)32983-6.
FROM THE LANCET
Dupilumab for severe AD: Expert advocates continuous treatment
LAHAINA, HAWAII – rather than treatment on an as-needed basis, Andrew Blauvelt, MD, advised at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
“I view atopic dermatitis as a chronic disease requiring chronic treatment. So be very careful about stopping. We know that if you start and stop biologics you’re going to be far more prone to develop antidrug antibodies resulting in drug resistance than with continual dosing,” said Dr. Blauvelt, a dermatologist and clinical trialist who is president of the Oregon Medical Research Center, Portland.
He said dupilumab (Dupixent) seldom induces disease remission as defined by clear skin while off all drugs for at least 1 year, although he has a few patients who seem to be exceptions. Yet clearly dupilumab doesn’t change an individual’s predisposing genetics or environmental allergen exposure pattern, so it’s best to think of it as a treatment for the long haul.
Dr. Blauvelt considers dupilumab far and away the best medication for treatment of adults and teenagers whose atopic dermatitis (AD) is uncontrolled with topical therapy. Payers often balk at authorizing dupilumab unless a patient has first undergone an unsuccessful trial of cyclosporine or methotrexate, which are far less expensive. But that’s not what the expert consensus guidelines recommend (Ann Allergy Asthma Immunol. 2018 Jan;120[1]:10-22.e2).
“The guidelines don’t suggest that failure on methotrexate or cyclosporine should be a prerequisite for dupilumab. So if you’re having problems with an insurance company and you really want to use dupilumab, you can point to this paper and say, ‘Look, the experts do not recommend step therapy, we can go directly to dupilumab.’ And the dupilumab label says simply that failure of topical therapy is required before being allowed to use dupilumab. So both the label and the experts say you don’t have to go through a bunch of steps in order to get to what I consider the very best drug for our patients,” he explained.
Both cyclosporine and methotrexate are far more broadly immunosuppressive and hence less safe than dupilumab. Both require laboratory monitoring. In contrast, blood work isn’t required in patients on dupilumab; in fact, Dr. Blauvelt considers it an unwise use of resources. Nor is tuberculosis testing advised prior to starting dupilumab.
When he can’t get authorization for dupilumab, Dr. Blauvelt’s go-to drug is methotrexate at 15-25 mg/week. It’s not as effective as cyclosporine for rapid clearing, but it’s safer for long-term use.
“Methotrexate is the devil we know – we know how to use it, and we know how to monitor for it,” he commented, adding that he reserves cyclosporine for a maximum of a month or 2 of acute crisis management, or as a bridge in getting patients off of systemic corticosteroids.
Set realistic efficacy expectations
Dermatologists who prescribe the newest biologics for psoriasis are accustomed to routinely seeing PASI 90 responses and even complete disease clearing. However, AD is a more challenging disease. In the landmark dupilumab phase 3 randomized trials, roughly two-thirds of patients achieved an Eczema Area and Severity Index (EASI) 75 response, with a mean 80% improvement in EASI symptom scores over baseline. Roughly 20% of dupilumab-treated adults with AD achieve disease clearance, and a similar percentage become almost clear. The improvements are durable in long-term follow-up studies.
“Dupilumab doesn’t get a lot of people to zero. They’re not going to be completely clearing their eczema. So they shouldn’t be freaking out if they still have eczema. What they can expect is diminution of the disease to much lower levels,” Dr. Blauvelt said.
The marked improvement in quality of life that occurs with dupilumab therapy isn’t adequately captured by EASI scores. “In my experience, more than 80%-90% of patients are happy on this drug,” Dr. Blauvelt said.
Conference codirector Linda Stein Gold, MD, agreed, commenting that she has found dupilumab to be “absolutely life altering” for her patients with severe AD.
“They know they still have AD, but now they can go whole days without thinking about it,” said Dr. Stein Gold, director of dermatology research and head of the division of dermatology at the Henry Ford Health System in Detroit.
Dr. Blauvelt noted that most of his patients on dupilumab remain on topical therapy, typically with triamcinolone on the body and hydrocortisone on the face. What he terms “miniflares” in patients on dupilumab are not at all unusual, but they’re readily manageable.
“Flares that used to last for weeks now last for a day or 2, maybe 3, and then it’s back to normal in patients on dupilumab,” Dr. Blauvelt said.
Safety
Dupilumab is a targeted inhibitor of interleukins-4 and -13, cytokines involved in allergy-mediated inflammation and the control of parasitic infections, but which have no bearing on control of bacterial or viral infections or malignancies. Indeed, the randomized trials have demonstrated that the incidence of skin infections is actually lower with dupilumab than with placebo.
“You’re improving the skin barrier so much that they’re not going to be getting staph or herpes simplex,” he explained.
The main side effect consists of dupilumab-associated eye issues. These occur in up to 20% of treated patients and encompass a spectrum ranging from dry eye to nonallergic conjunctivitis, inflammation of the eyelid, and keratitis. The mechanism is unknown. The condition is not infectious and doesn’t affect vision. Intriguingly, it doesn’t occur in patients with asthma, a disease for which dupilumab is also approved.
“Ask about eye issues at every office visit,” the dermatologist urged.
He sends all of his AD patients with dupilumab-associated eye issues to a single trusted local ophthalmologist and lets him manage the condition, which is generally mild to moderate. Eye issues have resulted in discontinuation of dupilumab in only 2 of the roughly 150 AD patients Dr. Blauvelt has placed on the biologic. The ophthalmologist generally relies upon lubricating eye drops and a couple of weeks of steroid eye drops or, in some cases, topical cyclosporine 0.05% ophthalmic emulsion, followed by episodic use of the steroid eye drops on an as-needed basis.
Residual facial disease in AD patients on dupilumab can be caused by a variety of causes, including breakthrough AD, rosacea, allergic contact dermatitis, steroid withdrawal, or photosensitivity, with Demodex thought to play a role in some cases.
Dr. Blauvelt reported serving as a scientific adviser to and paid clinical trial investigator for several dozen pharmaceutical companies. SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
LAHAINA, HAWAII – rather than treatment on an as-needed basis, Andrew Blauvelt, MD, advised at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
“I view atopic dermatitis as a chronic disease requiring chronic treatment. So be very careful about stopping. We know that if you start and stop biologics you’re going to be far more prone to develop antidrug antibodies resulting in drug resistance than with continual dosing,” said Dr. Blauvelt, a dermatologist and clinical trialist who is president of the Oregon Medical Research Center, Portland.
He said dupilumab (Dupixent) seldom induces disease remission as defined by clear skin while off all drugs for at least 1 year, although he has a few patients who seem to be exceptions. Yet clearly dupilumab doesn’t change an individual’s predisposing genetics or environmental allergen exposure pattern, so it’s best to think of it as a treatment for the long haul.
Dr. Blauvelt considers dupilumab far and away the best medication for treatment of adults and teenagers whose atopic dermatitis (AD) is uncontrolled with topical therapy. Payers often balk at authorizing dupilumab unless a patient has first undergone an unsuccessful trial of cyclosporine or methotrexate, which are far less expensive. But that’s not what the expert consensus guidelines recommend (Ann Allergy Asthma Immunol. 2018 Jan;120[1]:10-22.e2).
“The guidelines don’t suggest that failure on methotrexate or cyclosporine should be a prerequisite for dupilumab. So if you’re having problems with an insurance company and you really want to use dupilumab, you can point to this paper and say, ‘Look, the experts do not recommend step therapy, we can go directly to dupilumab.’ And the dupilumab label says simply that failure of topical therapy is required before being allowed to use dupilumab. So both the label and the experts say you don’t have to go through a bunch of steps in order to get to what I consider the very best drug for our patients,” he explained.
Both cyclosporine and methotrexate are far more broadly immunosuppressive and hence less safe than dupilumab. Both require laboratory monitoring. In contrast, blood work isn’t required in patients on dupilumab; in fact, Dr. Blauvelt considers it an unwise use of resources. Nor is tuberculosis testing advised prior to starting dupilumab.
When he can’t get authorization for dupilumab, Dr. Blauvelt’s go-to drug is methotrexate at 15-25 mg/week. It’s not as effective as cyclosporine for rapid clearing, but it’s safer for long-term use.
“Methotrexate is the devil we know – we know how to use it, and we know how to monitor for it,” he commented, adding that he reserves cyclosporine for a maximum of a month or 2 of acute crisis management, or as a bridge in getting patients off of systemic corticosteroids.
Set realistic efficacy expectations
Dermatologists who prescribe the newest biologics for psoriasis are accustomed to routinely seeing PASI 90 responses and even complete disease clearing. However, AD is a more challenging disease. In the landmark dupilumab phase 3 randomized trials, roughly two-thirds of patients achieved an Eczema Area and Severity Index (EASI) 75 response, with a mean 80% improvement in EASI symptom scores over baseline. Roughly 20% of dupilumab-treated adults with AD achieve disease clearance, and a similar percentage become almost clear. The improvements are durable in long-term follow-up studies.
“Dupilumab doesn’t get a lot of people to zero. They’re not going to be completely clearing their eczema. So they shouldn’t be freaking out if they still have eczema. What they can expect is diminution of the disease to much lower levels,” Dr. Blauvelt said.
The marked improvement in quality of life that occurs with dupilumab therapy isn’t adequately captured by EASI scores. “In my experience, more than 80%-90% of patients are happy on this drug,” Dr. Blauvelt said.
Conference codirector Linda Stein Gold, MD, agreed, commenting that she has found dupilumab to be “absolutely life altering” for her patients with severe AD.
“They know they still have AD, but now they can go whole days without thinking about it,” said Dr. Stein Gold, director of dermatology research and head of the division of dermatology at the Henry Ford Health System in Detroit.
Dr. Blauvelt noted that most of his patients on dupilumab remain on topical therapy, typically with triamcinolone on the body and hydrocortisone on the face. What he terms “miniflares” in patients on dupilumab are not at all unusual, but they’re readily manageable.
“Flares that used to last for weeks now last for a day or 2, maybe 3, and then it’s back to normal in patients on dupilumab,” Dr. Blauvelt said.
Safety
Dupilumab is a targeted inhibitor of interleukins-4 and -13, cytokines involved in allergy-mediated inflammation and the control of parasitic infections, but which have no bearing on control of bacterial or viral infections or malignancies. Indeed, the randomized trials have demonstrated that the incidence of skin infections is actually lower with dupilumab than with placebo.
“You’re improving the skin barrier so much that they’re not going to be getting staph or herpes simplex,” he explained.
The main side effect consists of dupilumab-associated eye issues. These occur in up to 20% of treated patients and encompass a spectrum ranging from dry eye to nonallergic conjunctivitis, inflammation of the eyelid, and keratitis. The mechanism is unknown. The condition is not infectious and doesn’t affect vision. Intriguingly, it doesn’t occur in patients with asthma, a disease for which dupilumab is also approved.
“Ask about eye issues at every office visit,” the dermatologist urged.
He sends all of his AD patients with dupilumab-associated eye issues to a single trusted local ophthalmologist and lets him manage the condition, which is generally mild to moderate. Eye issues have resulted in discontinuation of dupilumab in only 2 of the roughly 150 AD patients Dr. Blauvelt has placed on the biologic. The ophthalmologist generally relies upon lubricating eye drops and a couple of weeks of steroid eye drops or, in some cases, topical cyclosporine 0.05% ophthalmic emulsion, followed by episodic use of the steroid eye drops on an as-needed basis.
Residual facial disease in AD patients on dupilumab can be caused by a variety of causes, including breakthrough AD, rosacea, allergic contact dermatitis, steroid withdrawal, or photosensitivity, with Demodex thought to play a role in some cases.
Dr. Blauvelt reported serving as a scientific adviser to and paid clinical trial investigator for several dozen pharmaceutical companies. SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
LAHAINA, HAWAII – rather than treatment on an as-needed basis, Andrew Blauvelt, MD, advised at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
“I view atopic dermatitis as a chronic disease requiring chronic treatment. So be very careful about stopping. We know that if you start and stop biologics you’re going to be far more prone to develop antidrug antibodies resulting in drug resistance than with continual dosing,” said Dr. Blauvelt, a dermatologist and clinical trialist who is president of the Oregon Medical Research Center, Portland.
He said dupilumab (Dupixent) seldom induces disease remission as defined by clear skin while off all drugs for at least 1 year, although he has a few patients who seem to be exceptions. Yet clearly dupilumab doesn’t change an individual’s predisposing genetics or environmental allergen exposure pattern, so it’s best to think of it as a treatment for the long haul.
Dr. Blauvelt considers dupilumab far and away the best medication for treatment of adults and teenagers whose atopic dermatitis (AD) is uncontrolled with topical therapy. Payers often balk at authorizing dupilumab unless a patient has first undergone an unsuccessful trial of cyclosporine or methotrexate, which are far less expensive. But that’s not what the expert consensus guidelines recommend (Ann Allergy Asthma Immunol. 2018 Jan;120[1]:10-22.e2).
“The guidelines don’t suggest that failure on methotrexate or cyclosporine should be a prerequisite for dupilumab. So if you’re having problems with an insurance company and you really want to use dupilumab, you can point to this paper and say, ‘Look, the experts do not recommend step therapy, we can go directly to dupilumab.’ And the dupilumab label says simply that failure of topical therapy is required before being allowed to use dupilumab. So both the label and the experts say you don’t have to go through a bunch of steps in order to get to what I consider the very best drug for our patients,” he explained.
Both cyclosporine and methotrexate are far more broadly immunosuppressive and hence less safe than dupilumab. Both require laboratory monitoring. In contrast, blood work isn’t required in patients on dupilumab; in fact, Dr. Blauvelt considers it an unwise use of resources. Nor is tuberculosis testing advised prior to starting dupilumab.
When he can’t get authorization for dupilumab, Dr. Blauvelt’s go-to drug is methotrexate at 15-25 mg/week. It’s not as effective as cyclosporine for rapid clearing, but it’s safer for long-term use.
“Methotrexate is the devil we know – we know how to use it, and we know how to monitor for it,” he commented, adding that he reserves cyclosporine for a maximum of a month or 2 of acute crisis management, or as a bridge in getting patients off of systemic corticosteroids.
Set realistic efficacy expectations
Dermatologists who prescribe the newest biologics for psoriasis are accustomed to routinely seeing PASI 90 responses and even complete disease clearing. However, AD is a more challenging disease. In the landmark dupilumab phase 3 randomized trials, roughly two-thirds of patients achieved an Eczema Area and Severity Index (EASI) 75 response, with a mean 80% improvement in EASI symptom scores over baseline. Roughly 20% of dupilumab-treated adults with AD achieve disease clearance, and a similar percentage become almost clear. The improvements are durable in long-term follow-up studies.
“Dupilumab doesn’t get a lot of people to zero. They’re not going to be completely clearing their eczema. So they shouldn’t be freaking out if they still have eczema. What they can expect is diminution of the disease to much lower levels,” Dr. Blauvelt said.
The marked improvement in quality of life that occurs with dupilumab therapy isn’t adequately captured by EASI scores. “In my experience, more than 80%-90% of patients are happy on this drug,” Dr. Blauvelt said.
Conference codirector Linda Stein Gold, MD, agreed, commenting that she has found dupilumab to be “absolutely life altering” for her patients with severe AD.
“They know they still have AD, but now they can go whole days without thinking about it,” said Dr. Stein Gold, director of dermatology research and head of the division of dermatology at the Henry Ford Health System in Detroit.
Dr. Blauvelt noted that most of his patients on dupilumab remain on topical therapy, typically with triamcinolone on the body and hydrocortisone on the face. What he terms “miniflares” in patients on dupilumab are not at all unusual, but they’re readily manageable.
“Flares that used to last for weeks now last for a day or 2, maybe 3, and then it’s back to normal in patients on dupilumab,” Dr. Blauvelt said.
Safety
Dupilumab is a targeted inhibitor of interleukins-4 and -13, cytokines involved in allergy-mediated inflammation and the control of parasitic infections, but which have no bearing on control of bacterial or viral infections or malignancies. Indeed, the randomized trials have demonstrated that the incidence of skin infections is actually lower with dupilumab than with placebo.
“You’re improving the skin barrier so much that they’re not going to be getting staph or herpes simplex,” he explained.
The main side effect consists of dupilumab-associated eye issues. These occur in up to 20% of treated patients and encompass a spectrum ranging from dry eye to nonallergic conjunctivitis, inflammation of the eyelid, and keratitis. The mechanism is unknown. The condition is not infectious and doesn’t affect vision. Intriguingly, it doesn’t occur in patients with asthma, a disease for which dupilumab is also approved.
“Ask about eye issues at every office visit,” the dermatologist urged.
He sends all of his AD patients with dupilumab-associated eye issues to a single trusted local ophthalmologist and lets him manage the condition, which is generally mild to moderate. Eye issues have resulted in discontinuation of dupilumab in only 2 of the roughly 150 AD patients Dr. Blauvelt has placed on the biologic. The ophthalmologist generally relies upon lubricating eye drops and a couple of weeks of steroid eye drops or, in some cases, topical cyclosporine 0.05% ophthalmic emulsion, followed by episodic use of the steroid eye drops on an as-needed basis.
Residual facial disease in AD patients on dupilumab can be caused by a variety of causes, including breakthrough AD, rosacea, allergic contact dermatitis, steroid withdrawal, or photosensitivity, with Demodex thought to play a role in some cases.
Dr. Blauvelt reported serving as a scientific adviser to and paid clinical trial investigator for several dozen pharmaceutical companies. SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
EXPERT OPINION FROM SDEF HAWAII DERMATOLOGY SEMINAR
Consider allergic contact dermatitis in children with AD with disease flares, new rash
ORLANDO – Do you have ? Consider patch testing to assess whether they have allergic contact dermatitis.
“Of the patients who are sent to me by local pediatric dermatologists, 50% of them are positive” for allergens, said Jonathan H. Zippin, MD, PhD, director of the contact, occupational, and photodermatitis service at Cornell University, New York.
Speaking at the ODAC Dermatology, Aesthetic, and Surgical Conference, Dr. Zippin noted the prevalence of allergen sensitization is between 13% and 25% among children who are asymptomatic, while the prevalence of sensitization to at least one allergen among children with suspected allergic contact dermatitis (ACD) is between 25% and 96%. In 2014, a study from the National American Contact Dermatitis Group (NACDG) showed that of 883 children who were patch tested, 56.7% had at least one relevant positive patch test (RPPT) result.
“The take-home message here is that pediatric contact dermatitis is common, much more common than a lot of people realize,” Dr. Zippin said.
He described three common scenarios to keep in mind: a worsening rash, a new rash, and failure of a rash to improve after the patient avoids all of his or her positive allergens.
When a rash worsens, patch testing is likely to offer answers. In an analysis of 1,142 patients with suspected ACD aged 18 years or younger (mean age, 10.5 years; 64% female) in the Pediatric Contact Dermatitis Registry study database, 65% had at least one positive patch test, and 48% had at least 1 RPPT (Dermatitis 2016; 27[5] 293-302).
But not all patch testing is the same: The study also found that 24% of the RPPT cases would have been missed if assessed with the T.R.U.E. TEST compared with extended patch testing. If a T.R.U.E. TEST fails to explain generalized atopic dermatitis, the patient should be sent for more comprehensive testing where available, Dr. Zippin advised.
Pediatric patients also have unique allergens clinicians should consider. In the same study, children had a number of allergens similar to those of adults as reported in previous studies, such as nickel, cobalt, and neomycin. However, propylene glycol and cocamidopropyl betaine were allergens identified as unique to the pediatric population.
Another study looking at the same group of patients found that compared with children who did not have AD, children with AD had 7.4 times higher odds of having an RPPT to cocamidopropyl betaine, 7.6 times higher odds of having an RPPT to parthenolide, 5.3 times higher odds of having an RPPT to tixocortol pivalate, 4.2 times higher odds of having an RPPT to wool alcohols, and 4 times higher odds of having an RPPT to lanolin (JAMA Dermatology 2017;153[8]:765-70).
All of these are components of topical medicaments used to treat AD, “either components of emollients that we recommend, or components of steroids that we recommend,” Dr. Zippin pointed out.
One of these allergens could be the culprit when a child develops a new rash but there are no new apparent changes in products, exposures, and activities. Lanolin, also called wool grease, is used in many skin care products, for example. Dr. Zippin described the case of a 6-year-old girl with a history of AD, who presented with a new rash on her scalp and behind her ears, not explained by any obvious changes to products, exposures, or activities. Subsequent patch testing determined that the rash was caused by baby shampoo, which contained cocamidopropyl betaine, which is used in hypoallergenic products. The rash resolved after a different shampoo was used.
“Sometimes, we really have to be thinking when the rash is getting worse, is there something they’re being exposed to that might be an allergen?” Dr. Zippin said.
In patients who have avoided all their positive allergens but a rash has not improved, clinicians should consider systemic contact dermatitis (SCD). Patients can develop SCD through different types of exposures, including transepidermal, transmucosal, oral, intravenous, subcutaneous, intramuscular, inhalation, and implantation routes.
SCD also has a variety of presentations, including pompholyx/dyshidrosis/vesicular dermatitis, maculopapular eruption, chronic pruritus, exfoliative erythroderma/toxiderma, chronic urticaria, erythema multiforme and vasculitis, hyperkeratotic papules of the elbows, acute generalized exanthematous pustulosis, and pruritus ani, according to Dr. Zippin.
SCD should be considered when a patient has a positive patch test to an allergen that is known to cause SCD, and does not clear after avoiding cutaneous exposure to the allergen, Dr. Zippin advised.
Patients will most often develop SCD from plants and herbs, Dr. Zippin noted. Chrysanthemums and chamomile tea are common culprits for compositae allergy and can trigger SCD; other causes are Anacardiaceae, Balsam of Peru, and propolis. Metals (nickel, cobalt, gold, and chromium), medications (aminoglycosides, corticosteroids, and ethylenediamine), and other sources (formaldehyde, propylene glycol in frozen foods, gallates, and methylisothiazolinone) can cause SCD as well.
Methylisothiazolinone in particular is a very common sensitizer, Dr. Zippin said. “If you have a patient who is positive to this, it’s almost always the cause of their problem.”
Balsam of Peru is in a number of different foods, and patients who need to follow a diet free of Balsam of Peru should avoid a long list of foods including citrus; bakery goods; Danish pastry; candy; gum; spices such as cinnamon, cloves, vanilla, curry, allspice, anise, and ginger; spicy condiments such as ketchup, chili sauce, barbecue sauce; chili, pizza, and foods with red sauces; tomatoes; pickles; alcohol (wine, beer, gin, vermouth); tea (perfumed or flavored); tobacco; chocolate and ice cream; and soft drinks (cola or spiced soft drinks).
Patients starting a nickel-free diet should avoid soy, peanuts and other nuts, legumes, chocolate, cocoa, oats, fish, and whole wheat flours. Any elimination diet should last for 3 months but should at least be tried for 3-4 weeks, with gradual reintroduction of foods suspected as triggers once per week. Any type I allergies that are discovered or suspected can be referred to an allergist for allergen challenge and desensitization therapy.
For more information, Dr. Zippin recommended the American Contact Dermatitis Society website for more information.
Dr. Zippin reported that he is the founder and holds stock options at CEP Biotech; is on the medical advisory board and receives stock options from YouV Labs., is a paid consultant and performs industry-sponsored research for Pfizer, receives stock options from Regeneron, and is on the medical advisory board for Hoth Therapeutics Inc. He is on the board of directors for the American Contact Dermatitis Society.
ORLANDO – Do you have ? Consider patch testing to assess whether they have allergic contact dermatitis.
“Of the patients who are sent to me by local pediatric dermatologists, 50% of them are positive” for allergens, said Jonathan H. Zippin, MD, PhD, director of the contact, occupational, and photodermatitis service at Cornell University, New York.
Speaking at the ODAC Dermatology, Aesthetic, and Surgical Conference, Dr. Zippin noted the prevalence of allergen sensitization is between 13% and 25% among children who are asymptomatic, while the prevalence of sensitization to at least one allergen among children with suspected allergic contact dermatitis (ACD) is between 25% and 96%. In 2014, a study from the National American Contact Dermatitis Group (NACDG) showed that of 883 children who were patch tested, 56.7% had at least one relevant positive patch test (RPPT) result.
“The take-home message here is that pediatric contact dermatitis is common, much more common than a lot of people realize,” Dr. Zippin said.
He described three common scenarios to keep in mind: a worsening rash, a new rash, and failure of a rash to improve after the patient avoids all of his or her positive allergens.
When a rash worsens, patch testing is likely to offer answers. In an analysis of 1,142 patients with suspected ACD aged 18 years or younger (mean age, 10.5 years; 64% female) in the Pediatric Contact Dermatitis Registry study database, 65% had at least one positive patch test, and 48% had at least 1 RPPT (Dermatitis 2016; 27[5] 293-302).
But not all patch testing is the same: The study also found that 24% of the RPPT cases would have been missed if assessed with the T.R.U.E. TEST compared with extended patch testing. If a T.R.U.E. TEST fails to explain generalized atopic dermatitis, the patient should be sent for more comprehensive testing where available, Dr. Zippin advised.
Pediatric patients also have unique allergens clinicians should consider. In the same study, children had a number of allergens similar to those of adults as reported in previous studies, such as nickel, cobalt, and neomycin. However, propylene glycol and cocamidopropyl betaine were allergens identified as unique to the pediatric population.
Another study looking at the same group of patients found that compared with children who did not have AD, children with AD had 7.4 times higher odds of having an RPPT to cocamidopropyl betaine, 7.6 times higher odds of having an RPPT to parthenolide, 5.3 times higher odds of having an RPPT to tixocortol pivalate, 4.2 times higher odds of having an RPPT to wool alcohols, and 4 times higher odds of having an RPPT to lanolin (JAMA Dermatology 2017;153[8]:765-70).
All of these are components of topical medicaments used to treat AD, “either components of emollients that we recommend, or components of steroids that we recommend,” Dr. Zippin pointed out.
One of these allergens could be the culprit when a child develops a new rash but there are no new apparent changes in products, exposures, and activities. Lanolin, also called wool grease, is used in many skin care products, for example. Dr. Zippin described the case of a 6-year-old girl with a history of AD, who presented with a new rash on her scalp and behind her ears, not explained by any obvious changes to products, exposures, or activities. Subsequent patch testing determined that the rash was caused by baby shampoo, which contained cocamidopropyl betaine, which is used in hypoallergenic products. The rash resolved after a different shampoo was used.
“Sometimes, we really have to be thinking when the rash is getting worse, is there something they’re being exposed to that might be an allergen?” Dr. Zippin said.
In patients who have avoided all their positive allergens but a rash has not improved, clinicians should consider systemic contact dermatitis (SCD). Patients can develop SCD through different types of exposures, including transepidermal, transmucosal, oral, intravenous, subcutaneous, intramuscular, inhalation, and implantation routes.
SCD also has a variety of presentations, including pompholyx/dyshidrosis/vesicular dermatitis, maculopapular eruption, chronic pruritus, exfoliative erythroderma/toxiderma, chronic urticaria, erythema multiforme and vasculitis, hyperkeratotic papules of the elbows, acute generalized exanthematous pustulosis, and pruritus ani, according to Dr. Zippin.
SCD should be considered when a patient has a positive patch test to an allergen that is known to cause SCD, and does not clear after avoiding cutaneous exposure to the allergen, Dr. Zippin advised.
Patients will most often develop SCD from plants and herbs, Dr. Zippin noted. Chrysanthemums and chamomile tea are common culprits for compositae allergy and can trigger SCD; other causes are Anacardiaceae, Balsam of Peru, and propolis. Metals (nickel, cobalt, gold, and chromium), medications (aminoglycosides, corticosteroids, and ethylenediamine), and other sources (formaldehyde, propylene glycol in frozen foods, gallates, and methylisothiazolinone) can cause SCD as well.
Methylisothiazolinone in particular is a very common sensitizer, Dr. Zippin said. “If you have a patient who is positive to this, it’s almost always the cause of their problem.”
Balsam of Peru is in a number of different foods, and patients who need to follow a diet free of Balsam of Peru should avoid a long list of foods including citrus; bakery goods; Danish pastry; candy; gum; spices such as cinnamon, cloves, vanilla, curry, allspice, anise, and ginger; spicy condiments such as ketchup, chili sauce, barbecue sauce; chili, pizza, and foods with red sauces; tomatoes; pickles; alcohol (wine, beer, gin, vermouth); tea (perfumed or flavored); tobacco; chocolate and ice cream; and soft drinks (cola or spiced soft drinks).
Patients starting a nickel-free diet should avoid soy, peanuts and other nuts, legumes, chocolate, cocoa, oats, fish, and whole wheat flours. Any elimination diet should last for 3 months but should at least be tried for 3-4 weeks, with gradual reintroduction of foods suspected as triggers once per week. Any type I allergies that are discovered or suspected can be referred to an allergist for allergen challenge and desensitization therapy.
For more information, Dr. Zippin recommended the American Contact Dermatitis Society website for more information.
Dr. Zippin reported that he is the founder and holds stock options at CEP Biotech; is on the medical advisory board and receives stock options from YouV Labs., is a paid consultant and performs industry-sponsored research for Pfizer, receives stock options from Regeneron, and is on the medical advisory board for Hoth Therapeutics Inc. He is on the board of directors for the American Contact Dermatitis Society.
ORLANDO – Do you have ? Consider patch testing to assess whether they have allergic contact dermatitis.
“Of the patients who are sent to me by local pediatric dermatologists, 50% of them are positive” for allergens, said Jonathan H. Zippin, MD, PhD, director of the contact, occupational, and photodermatitis service at Cornell University, New York.
Speaking at the ODAC Dermatology, Aesthetic, and Surgical Conference, Dr. Zippin noted the prevalence of allergen sensitization is between 13% and 25% among children who are asymptomatic, while the prevalence of sensitization to at least one allergen among children with suspected allergic contact dermatitis (ACD) is between 25% and 96%. In 2014, a study from the National American Contact Dermatitis Group (NACDG) showed that of 883 children who were patch tested, 56.7% had at least one relevant positive patch test (RPPT) result.
“The take-home message here is that pediatric contact dermatitis is common, much more common than a lot of people realize,” Dr. Zippin said.
He described three common scenarios to keep in mind: a worsening rash, a new rash, and failure of a rash to improve after the patient avoids all of his or her positive allergens.
When a rash worsens, patch testing is likely to offer answers. In an analysis of 1,142 patients with suspected ACD aged 18 years or younger (mean age, 10.5 years; 64% female) in the Pediatric Contact Dermatitis Registry study database, 65% had at least one positive patch test, and 48% had at least 1 RPPT (Dermatitis 2016; 27[5] 293-302).
But not all patch testing is the same: The study also found that 24% of the RPPT cases would have been missed if assessed with the T.R.U.E. TEST compared with extended patch testing. If a T.R.U.E. TEST fails to explain generalized atopic dermatitis, the patient should be sent for more comprehensive testing where available, Dr. Zippin advised.
Pediatric patients also have unique allergens clinicians should consider. In the same study, children had a number of allergens similar to those of adults as reported in previous studies, such as nickel, cobalt, and neomycin. However, propylene glycol and cocamidopropyl betaine were allergens identified as unique to the pediatric population.
Another study looking at the same group of patients found that compared with children who did not have AD, children with AD had 7.4 times higher odds of having an RPPT to cocamidopropyl betaine, 7.6 times higher odds of having an RPPT to parthenolide, 5.3 times higher odds of having an RPPT to tixocortol pivalate, 4.2 times higher odds of having an RPPT to wool alcohols, and 4 times higher odds of having an RPPT to lanolin (JAMA Dermatology 2017;153[8]:765-70).
All of these are components of topical medicaments used to treat AD, “either components of emollients that we recommend, or components of steroids that we recommend,” Dr. Zippin pointed out.
One of these allergens could be the culprit when a child develops a new rash but there are no new apparent changes in products, exposures, and activities. Lanolin, also called wool grease, is used in many skin care products, for example. Dr. Zippin described the case of a 6-year-old girl with a history of AD, who presented with a new rash on her scalp and behind her ears, not explained by any obvious changes to products, exposures, or activities. Subsequent patch testing determined that the rash was caused by baby shampoo, which contained cocamidopropyl betaine, which is used in hypoallergenic products. The rash resolved after a different shampoo was used.
“Sometimes, we really have to be thinking when the rash is getting worse, is there something they’re being exposed to that might be an allergen?” Dr. Zippin said.
In patients who have avoided all their positive allergens but a rash has not improved, clinicians should consider systemic contact dermatitis (SCD). Patients can develop SCD through different types of exposures, including transepidermal, transmucosal, oral, intravenous, subcutaneous, intramuscular, inhalation, and implantation routes.
SCD also has a variety of presentations, including pompholyx/dyshidrosis/vesicular dermatitis, maculopapular eruption, chronic pruritus, exfoliative erythroderma/toxiderma, chronic urticaria, erythema multiforme and vasculitis, hyperkeratotic papules of the elbows, acute generalized exanthematous pustulosis, and pruritus ani, according to Dr. Zippin.
SCD should be considered when a patient has a positive patch test to an allergen that is known to cause SCD, and does not clear after avoiding cutaneous exposure to the allergen, Dr. Zippin advised.
Patients will most often develop SCD from plants and herbs, Dr. Zippin noted. Chrysanthemums and chamomile tea are common culprits for compositae allergy and can trigger SCD; other causes are Anacardiaceae, Balsam of Peru, and propolis. Metals (nickel, cobalt, gold, and chromium), medications (aminoglycosides, corticosteroids, and ethylenediamine), and other sources (formaldehyde, propylene glycol in frozen foods, gallates, and methylisothiazolinone) can cause SCD as well.
Methylisothiazolinone in particular is a very common sensitizer, Dr. Zippin said. “If you have a patient who is positive to this, it’s almost always the cause of their problem.”
Balsam of Peru is in a number of different foods, and patients who need to follow a diet free of Balsam of Peru should avoid a long list of foods including citrus; bakery goods; Danish pastry; candy; gum; spices such as cinnamon, cloves, vanilla, curry, allspice, anise, and ginger; spicy condiments such as ketchup, chili sauce, barbecue sauce; chili, pizza, and foods with red sauces; tomatoes; pickles; alcohol (wine, beer, gin, vermouth); tea (perfumed or flavored); tobacco; chocolate and ice cream; and soft drinks (cola or spiced soft drinks).
Patients starting a nickel-free diet should avoid soy, peanuts and other nuts, legumes, chocolate, cocoa, oats, fish, and whole wheat flours. Any elimination diet should last for 3 months but should at least be tried for 3-4 weeks, with gradual reintroduction of foods suspected as triggers once per week. Any type I allergies that are discovered or suspected can be referred to an allergist for allergen challenge and desensitization therapy.
For more information, Dr. Zippin recommended the American Contact Dermatitis Society website for more information.
Dr. Zippin reported that he is the founder and holds stock options at CEP Biotech; is on the medical advisory board and receives stock options from YouV Labs., is a paid consultant and performs industry-sponsored research for Pfizer, receives stock options from Regeneron, and is on the medical advisory board for Hoth Therapeutics Inc. He is on the board of directors for the American Contact Dermatitis Society.
EXPERT ANALYSIS FROM ODAC 2020
Effect of In-Office Samples on Dermatologists’ Prescribing Habits: A Retrospective Review
Over the years, there has been growing concern about the relationship between physicians and pharmaceutical companies. Many studies have demonstrated that pharmaceutical interactions and incentives can influence physicians’ prescribing habits.1-3 As a result, many academic centers have adopted policies that attempt to limit the pharmaceutical industry’s influence on faculty and in-training physicians. Although these policies can vary greatly, they generally limit access of pharmaceutical representatives to providers and restrict pharmaceutical samples.4,5 This policy shift has even been reported in private practice.6
At the heart of the matter is the question: What really influences physicians to write a prescription for a particular medication? Is it cost, efficacy, or representatives pushing a product? Prior studies illustrate that generic medications are equivalent to their brand-name counterparts. In fact, current regulations require no more than 5% to 7% difference in bioequivalence.7-9 Although most generic medications are bioequivalent, it may not be universal.10
Garrison and Levin11 distributed a survey to US-based prescribers in family practice, psychiatry, and internal medicine and found that prescribers deemed patient response and success as the highest priority when determining which drugs to prescribe. In contrast, drug representatives and free samples only slightly contributed.11 Considering the minimum duration for efficacy of a medication such as an antidepressant vs a topical steroid, this pattern may differ with samples in dermatologic settings. Interestingly, another survey concluded that samples were associated with “sticky” prescribing habits, noting that physicians would prescribe a brand-name medication after using a sample, despite increased cost to the patient.12 Further, it has been suggested that recipients of free samples may experience increased costs in the long run, which contrasts a stated goal of affordability to patients.12,13
Physician interaction with pharmaceutical companies begins as early as medical school,14 with physicians reporting interactions as often as 4 times each month.14-18 Interactions can include meetings with pharmaceutical representatives, sponsored meals, gifts, continuing medical education sponsorship, funding for travel, pharmaceutical representative speakers, research funding, and drug samples.3
A 2014 study reported that prescribing habits are influenced by the free drug samples provided by nongeneric pharmaceutical companies.19 Nationally, the number of brand-name and branded generic medications constitute 79% of prescriptions, yet together they only comprise 17% of medications prescribed at an academic medical clinic that does not provide samples. The number of medications with samples being prescribed by dermatologists increased by 15% over 9 years, which may correlate with the wider availability of medication samples, more specifically an increase in branded generic samples.19 This potential interaction is the reason why institutions question the current influence of pharmaceutical companies. Samples may appear convenient, allowing a patient to test the medication prior to committing; however, with brand-name samples being provided to the physician, he/she may become more inclined to prescribe the branded medication.12,15,19-22 Because brand-name medications are more expensive than generic medications, this practice can increase the cost of health care.13 One study found that over 1 year, the overuse of nongeneric medications led to a loss of potential savings throughout 49 states, equating to $229 million just through Medicaid; interestingly, it was noted that in some states, a maximum reimbursement is set by Medicaid, regardless of whether the generic or branded medication is dispensed. The authors also noted variability in the potential savings by state, which may be a function of the state-by-state maximum reimbursements for certain medications.23 Another study on oral combination medications estimated Medicare spending on branded drugs relative to the cost if generic combinations had been purchased instead. This study examined branded medications for which the active components were available as over-the-counter (OTC), generic, or same-class generic, and the authors estimated that $925 million could have been saved in 2016 by purchasing a generic substitute.24 The overuse of nongeneric medications when generic alternatives are available becomes an issue that not only financially impacts patients but all taxpayers. However, this pattern may differ if limited only to dermatologic medications, which was not the focus of the prior studies.
To limit conflicts of interest in interactions with the pharmaceutical, medical device, and biotechnology industries, the University of South Florida (USF) Morsani College of Medicine (COM)(Tampa, Florida) implemented its own set of regulations that eliminated in-office pharmaceutical samples, in addition to other restrictions. This study aimed to investigate if there was a change in the prescribing habits of academic dermatologists after their medical school implemented these new policies.
We hypothesized that the number of brand-name drugs prescribed by physicians in the Department of Dermatology & Cutaneous Surgery would change following USF Morsani COM pharmaceutical policy changes. We sought to determine how physician prescribing practices within the Department of Dermatology & Cutaneous Surgery changed following USF Morsani COM pharmaceutical policy changes.
Methods
Data Collection
A retrospective review of medical records was conducted to investigate the effect of the USF Morsani COM pharmaceutical policy changes on physician prescribing practices within the Department of Dermatology & Cutaneous Surgery. Medical records of patients seen for common dermatology diagnoses before (January 1, 2010, to May 30, 2010) and after (August 1, 2011, to December 31, 2011) the pharmaceutical policy changes were reviewed, and all medications prescribed were recorded. Data were collected from medical records within the USF Health electronic medical record system and included visits with each of the department’s 3 attending dermatologists. The diagnoses included in the study—acne vulgaris, atopic dermatitis, onychomycosis, psoriasis, and rosacea—were chosen because in-office samples were available. Prescribing data from the first 100 consecutive medical records were collected from each time period, and a medical record was included only if it contained at least 1 of the following diagnoses: acne vulgaris, atopic dermatitis, onychomycosis, psoriasis, or rosacea. The assessment and plan of each progress note were reviewed, and the exact medication name and associated diagnosis were recorded for each prescription. Subsequently, each medication was reviewed and placed in 1 of 3 categories: brand name, generic, and OTC. The total number of prescriptions for each diagnosis (per visit/note); the specific number of brand, generic, and OTC medications prescribed (per visit/note); and the percentage of brand, generic, and OTC medications prescribed (per visit/note and per diagnosis in total) were calculated. To ensure only intended medications were included, each medication recorded in the medical record note was cross-referenced with the prescribed medication in the electronic medical record. The primary objective of this study was to capture the prescribing physician’s intent as proxied by the pattern of prescription. Thus, changes made in prescriptions after the initial plan—whether insurance related or otherwise—were not relevant to this investigation.
The data were collected to compare the percentage of brand vs generic or OTC prescriptions per diagnosis to see if there was a difference in the prescribing habits before and after the pharmaceutical policy changes. Of note, several other pieces of data were collected from each medical record, including age, race, class of insurance (ie, Medicare, Medicaid, private health maintenance organization, private preferred provider organization), subtype diagnoses, and whether the prescription was new or a refill. The information gathered from the written record on the assessment and plan was verified using prescriptions ordered in the Allscripts electronic record, and any difference was noted. No identifying information that could be used to easily identify study participants was recorded.
Differences in prescribing habits across diagnoses before and after the policy changes were ascertained using a Fisher exact test and were further assessed using a mixed effects ordinal logistic regression model that accounted for within-provider clustering and baseline patient characteristics. An ordinal model was chosen to recognize differences in average cost among brand-name, generic, and OTC medications.
Results
In total, 200 medical records were collected. For the period analyzed before the policy change, 252 brand-name medications were prescribed compared to 231 prescribed for the period analyzed after the policy changes. There was insufficient evidence of an overall difference in brand-name medications prescribed before and after the policy changes (P=.145; Fisher exact test)(Table 1). There also was insufficient evidence of an overall difference in generic prescriptions, which totaled 153 before and 134 after the policy changes (P=.872; Fisher exact test)(Table 2). Over-the-counter prescriptions totaled 49 before and 69 after the policy changes. There was insufficient evidence of an overall difference before and after the policy changes for OTC medications (P=.192; Fisher exact test)(Table 3).
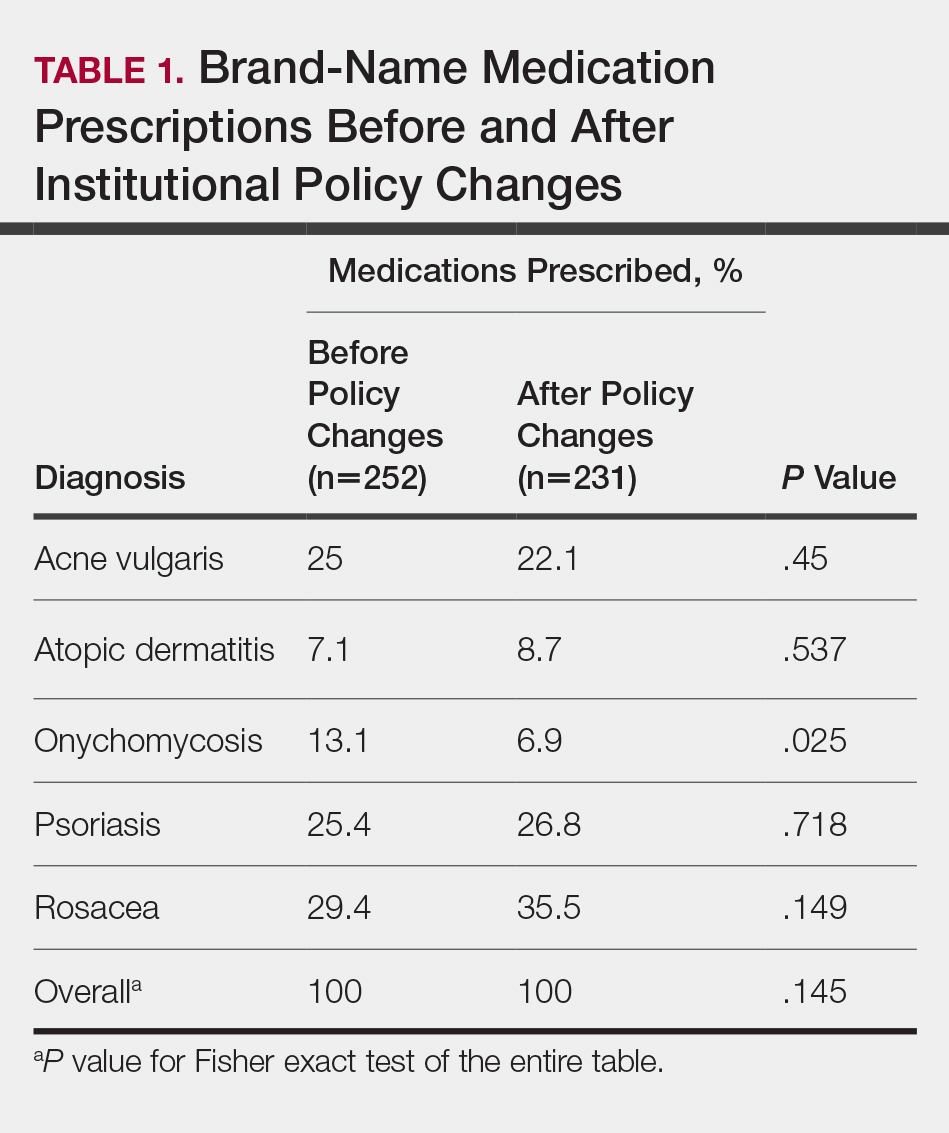

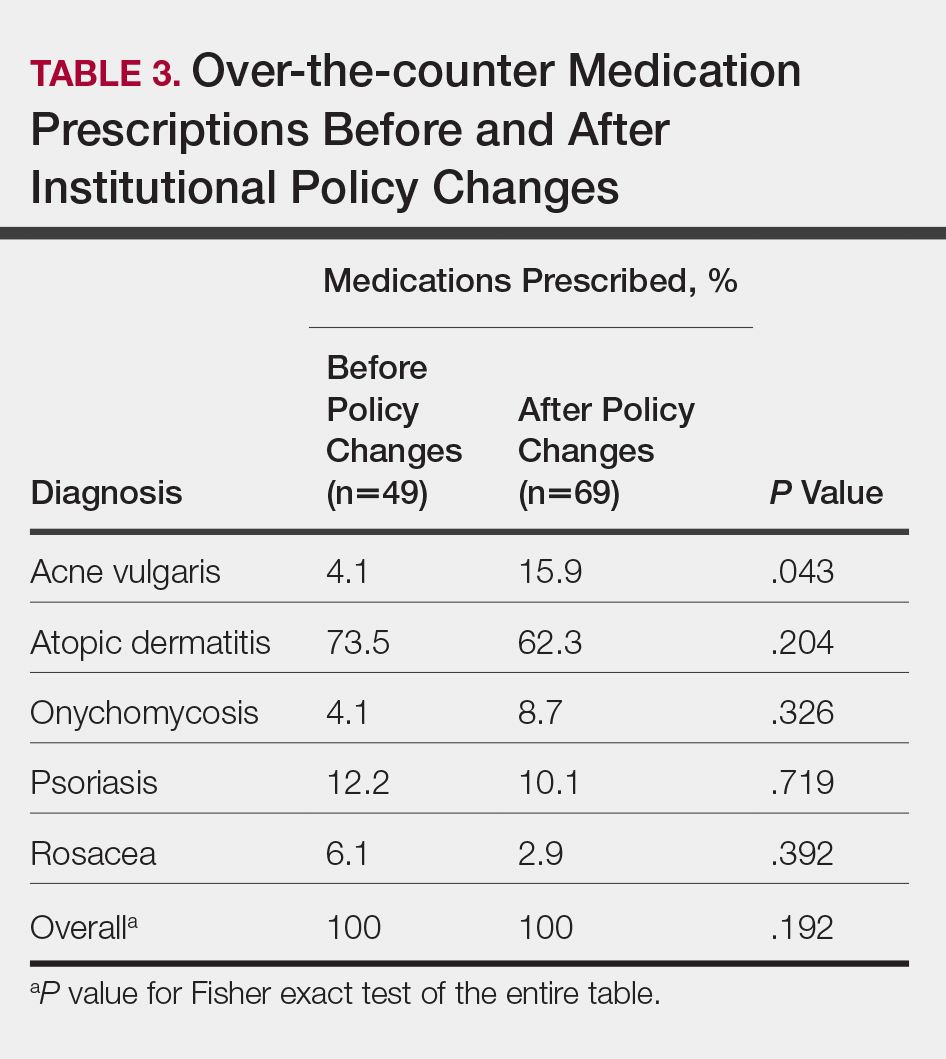
Comment
Although some medical institutions are diligently working to limit the potential influence pharmaceutical companies have on physician prescribing habits,4,5,25 the effect on physician prescribing habits is only now being established.15 Prior studies12,19,21 have found evidence that medication samples may lead to overuse of brand-name medications, but these findings do not hold true for the USF dermatologists included in this study, perhaps due to the difference in pharmaceutical company interactions or physicians maintaining prior prescription habits that were unrelated to the policy. Although this study focused on policy changes for in-office samples, prior studies either included other forms of interaction21 or did not include samples.22
Pharmaceutical samples allow patients to try a medication before committing to a long-term course of treatment with a particular medication, which has utility for physicians and patients. Although brand-name prescriptions may cost more, a trial period may assist the patient in deciding whether the medication is worth purchasing. Furthermore, physicians may feel more comfortable prescribing a medication once the individual patient has demonstrated a benefit from the sample, which may be particularly true in a specialty such as dermatology in which many branded topical medications contain a different vehicle than generic formulations, resulting in notable variations in active medication delivery and efficacy. Given the higher cost of branded topical medications, proving efficacy in patients through samples can provide a useful tool to the physician to determine the need for a branded formulation.
The benefits described are subjective but should not be disregarded. Although Hurley et al19 found that the number of brand-name medications prescribed increases as more samples are given out, our study demonstrated that after eliminating medication samples, there was no significant difference in the percentage of brand-name medications prescribed compared to generic and OTC medications.
Physician education concerning the price of each brand-name medication prescribed in office may be one method of reducing the amount of such prescriptions. Physicians generally are uninformed of the cost of the medications being prescribed26 and may not recognize the financial burden one medication may have compared to its alternative. However, educating physicians will empower them to make the conscious decision to prefer or not prefer a brand-name medication. With some generic medications shown to have a difference in bioequivalence compared to their brand-name counterparts, a physician may find more success prescribing the brand-name medications, regardless of pharmaceutical company influence, which is an alternative solution to policy changes that eliminate samples entirely. Although this study found insufficient evidence that removing samples decreases brand-name medication prescriptions, it is imperative that solutions are established to reduce the country’s increasing burden of medical costs.
Possible shortfalls of this study include the short period of time between which prepolicy data and postpolicy data were collected. It is possible that providers did not have enough time to adjust their prescribing habits or that providers would not have changed a prescribing pattern or preference simply because of a policy change. Future studies could allow a time period greater than 2 years to compare prepolicy and postpolicy prescribing habits, or a future study might make comparisons of prescriber patterns at different institutions that have different policies. Another possible shortfall is that providers and patients were limited to those at the Department of Dermatology & Cutaneous Surgery at the USF Morsani COM. Although this study has found insufficient evidence of a difference in prescribing habits, it may be beneficial to conduct a larger study that encompasses multiple academic institutions with similar policy changes. Most importantly, this study only investigated the influence of in-office pharmaceutical samples on prescribing patterns. This study did not look at the many other ways in which providers may be influenced by pharmaceutical companies, which likely is a significant confounding variable in this study. Continued additional studies that specifically examine other methods through which providers may be influenced would be helpful in further examining the many ways in which physician prescription habits are influenced.
Conclusion
Changes in pharmaceutical policy in 2011 at USF Morsani COM specifically banned in-office samples. The totality of evidence in this study shows modest observational evidence of a change in the postpolicy odds relative to prepolicy odds, but the data also are compatible with no change between prescribing habits before and after the policy changes. Further study is needed to fully understand this relationship.
- Sondergaard J, Vach K, Kragstrup J, et al. Impact of pharmaceutical representative visits on GPs’ drug preferences. Fam Pract. 2009;26:204-209.
- Jelinek GA, Neate SL. The influence of the pharmaceutical industry in medicine. J Law Med. 2009;17:216-223.
- Wazana A. Physicians and the pharmaceutical industry: is a gift ever just a gift? JAMA. 2000;283:373-380.
- Coleman DL. Establishing policies for the relationship between industry and clinicians: lessons learned from two academic health centers. Acad Med. 2008;83:882-887.
- Coleman DL, Kazdin AE, Miller LA, et al. Guidelines for interactions between clinical faculty and the pharmaceutical industry: one medical school’s approach. Acad Med. 2006;81:154-160.
- Evans D, Hartung DM, Beasley D, et al. Breaking up is hard to do: lessons learned from a pharma-free practice transformation. J Am Board Fam Med. 2013;26:332-338.
- Davit BM, Nwakama PE, Buehler GJ, et al. Comparing generic and innovator drugs: a review of 12 years of bioequivalence data from the United States Food and Drug Administration. Ann Pharmacother. 2009;43:1583-1597.
- Kesselheim AS, Misono AS, Lee JL, et al. Clinical equivalence of generic and brand-name drugs used in cardiovascular disease: a systematic review and meta-analysis. JAMA. 2008;300:2514-2526.
- McCormack J, Chmelicek JT. Generic versus brand name: the other drug war. Can Fam Physician. 2014;60:911.
- Borgheini G. The bioequivalence and therapeutic efficacy of generic versus brand-name psychoactive drugs. Clin Ther. 2003;25:1578-1592.
- Garrison GD, Levin GM. Factors affecting prescribing of the newer antidepressants. Ann Pharmacother. 2000;34:10-14.
- Rafique S, Sarwar W, Rashid A, et al. Influence of free drug samples on prescribing by physicians: a cross sectional survey. J Pak Med Assoc. 2017;67:465-467.
- Alexander GC, Zhang J, Basu A. Characteristics of patients receiving pharmaceutical samples and association between sample receipt and out-of-pocket prescription costs. Med Care. 2008;46:394-402.
- Hodges B. Interactions with the pharmaceutical industry: experiences and attitudes of psychiatry residents, interns and clerks. CMAJ. 1995;153:553-559.
- Brotzman GL, Mark DH. The effect on resident attitudes of regulatory policies regarding pharmaceutical representative activities. J Gen Intern Med. 1993;8:130-134.
- Keim SM, Sanders AB, Witzke DB, et al. Beliefs and practices of emergency medicine faculty and residents regarding professional interactions with the biomedical industry. Ann Emerg Med. 1993;22:1576-1581.
- Thomson AN, Craig BJ, Barham PM. Attitudes of general practitioners in New Zealand to pharmaceutical representatives. Br J Gen Pract. 1994;44:220-223.
- Ziegler MG, Lew P, Singer BC. The accuracy of drug information from pharmaceutical sales representatives. JAMA. 1995;273:1296-1298.
- Hurley MP, Stafford RS, Lane AT. Characterizing the relationship between free drug samples and prescription patterns for acne vulgaris and rosacea. JAMA Dermatol. 2014;150:487-493.
- Lexchin J. Interactions between physicians and the pharmaceutical industry: what does the literature say? CMAJ. 1993;149:1401-1407.
- Lieb K, Scheurich A. Contact between doctors and the pharmaceutical industry, their perceptions, and the effects on prescribing habits. PLoS One. 2014;9:e110130.
- Spurling GK, Mansfield PR, Montgomery BD, et al. Information from pharmaceutical companies and the quality, quantity, and cost of physicians’ prescribing: a systematic review. PLoS Med. 2010;7:e1000352.
- Fischer MA, Avorn J. Economic consequences of underuse of generic drugs: evidence from Medicaid and implications for prescription drug benefit plans. Health Serv Res. 2003;38:1051-1064.
- Sacks CA, Lee CC, Kesselheim AS, et al. Medicare spending on brand-name combination medications vs their generic constituents. JAMA. 2018;320:650-656.
- Brennan TA, Rothman DJ, Blank L, et al. Health industry practices that create conflicts of interest: a policy proposal for academic medical centers. JAMA. 2006;295:429-433.
- Allan GM, Lexchin J, Wiebe N. Physician awareness of drug cost: a systematic review. PLoS Med. 2007;4:e283.
Over the years, there has been growing concern about the relationship between physicians and pharmaceutical companies. Many studies have demonstrated that pharmaceutical interactions and incentives can influence physicians’ prescribing habits.1-3 As a result, many academic centers have adopted policies that attempt to limit the pharmaceutical industry’s influence on faculty and in-training physicians. Although these policies can vary greatly, they generally limit access of pharmaceutical representatives to providers and restrict pharmaceutical samples.4,5 This policy shift has even been reported in private practice.6
At the heart of the matter is the question: What really influences physicians to write a prescription for a particular medication? Is it cost, efficacy, or representatives pushing a product? Prior studies illustrate that generic medications are equivalent to their brand-name counterparts. In fact, current regulations require no more than 5% to 7% difference in bioequivalence.7-9 Although most generic medications are bioequivalent, it may not be universal.10
Garrison and Levin11 distributed a survey to US-based prescribers in family practice, psychiatry, and internal medicine and found that prescribers deemed patient response and success as the highest priority when determining which drugs to prescribe. In contrast, drug representatives and free samples only slightly contributed.11 Considering the minimum duration for efficacy of a medication such as an antidepressant vs a topical steroid, this pattern may differ with samples in dermatologic settings. Interestingly, another survey concluded that samples were associated with “sticky” prescribing habits, noting that physicians would prescribe a brand-name medication after using a sample, despite increased cost to the patient.12 Further, it has been suggested that recipients of free samples may experience increased costs in the long run, which contrasts a stated goal of affordability to patients.12,13
Physician interaction with pharmaceutical companies begins as early as medical school,14 with physicians reporting interactions as often as 4 times each month.14-18 Interactions can include meetings with pharmaceutical representatives, sponsored meals, gifts, continuing medical education sponsorship, funding for travel, pharmaceutical representative speakers, research funding, and drug samples.3
A 2014 study reported that prescribing habits are influenced by the free drug samples provided by nongeneric pharmaceutical companies.19 Nationally, the number of brand-name and branded generic medications constitute 79% of prescriptions, yet together they only comprise 17% of medications prescribed at an academic medical clinic that does not provide samples. The number of medications with samples being prescribed by dermatologists increased by 15% over 9 years, which may correlate with the wider availability of medication samples, more specifically an increase in branded generic samples.19 This potential interaction is the reason why institutions question the current influence of pharmaceutical companies. Samples may appear convenient, allowing a patient to test the medication prior to committing; however, with brand-name samples being provided to the physician, he/she may become more inclined to prescribe the branded medication.12,15,19-22 Because brand-name medications are more expensive than generic medications, this practice can increase the cost of health care.13 One study found that over 1 year, the overuse of nongeneric medications led to a loss of potential savings throughout 49 states, equating to $229 million just through Medicaid; interestingly, it was noted that in some states, a maximum reimbursement is set by Medicaid, regardless of whether the generic or branded medication is dispensed. The authors also noted variability in the potential savings by state, which may be a function of the state-by-state maximum reimbursements for certain medications.23 Another study on oral combination medications estimated Medicare spending on branded drugs relative to the cost if generic combinations had been purchased instead. This study examined branded medications for which the active components were available as over-the-counter (OTC), generic, or same-class generic, and the authors estimated that $925 million could have been saved in 2016 by purchasing a generic substitute.24 The overuse of nongeneric medications when generic alternatives are available becomes an issue that not only financially impacts patients but all taxpayers. However, this pattern may differ if limited only to dermatologic medications, which was not the focus of the prior studies.
To limit conflicts of interest in interactions with the pharmaceutical, medical device, and biotechnology industries, the University of South Florida (USF) Morsani College of Medicine (COM)(Tampa, Florida) implemented its own set of regulations that eliminated in-office pharmaceutical samples, in addition to other restrictions. This study aimed to investigate if there was a change in the prescribing habits of academic dermatologists after their medical school implemented these new policies.
We hypothesized that the number of brand-name drugs prescribed by physicians in the Department of Dermatology & Cutaneous Surgery would change following USF Morsani COM pharmaceutical policy changes. We sought to determine how physician prescribing practices within the Department of Dermatology & Cutaneous Surgery changed following USF Morsani COM pharmaceutical policy changes.
Methods
Data Collection
A retrospective review of medical records was conducted to investigate the effect of the USF Morsani COM pharmaceutical policy changes on physician prescribing practices within the Department of Dermatology & Cutaneous Surgery. Medical records of patients seen for common dermatology diagnoses before (January 1, 2010, to May 30, 2010) and after (August 1, 2011, to December 31, 2011) the pharmaceutical policy changes were reviewed, and all medications prescribed were recorded. Data were collected from medical records within the USF Health electronic medical record system and included visits with each of the department’s 3 attending dermatologists. The diagnoses included in the study—acne vulgaris, atopic dermatitis, onychomycosis, psoriasis, and rosacea—were chosen because in-office samples were available. Prescribing data from the first 100 consecutive medical records were collected from each time period, and a medical record was included only if it contained at least 1 of the following diagnoses: acne vulgaris, atopic dermatitis, onychomycosis, psoriasis, or rosacea. The assessment and plan of each progress note were reviewed, and the exact medication name and associated diagnosis were recorded for each prescription. Subsequently, each medication was reviewed and placed in 1 of 3 categories: brand name, generic, and OTC. The total number of prescriptions for each diagnosis (per visit/note); the specific number of brand, generic, and OTC medications prescribed (per visit/note); and the percentage of brand, generic, and OTC medications prescribed (per visit/note and per diagnosis in total) were calculated. To ensure only intended medications were included, each medication recorded in the medical record note was cross-referenced with the prescribed medication in the electronic medical record. The primary objective of this study was to capture the prescribing physician’s intent as proxied by the pattern of prescription. Thus, changes made in prescriptions after the initial plan—whether insurance related or otherwise—were not relevant to this investigation.
The data were collected to compare the percentage of brand vs generic or OTC prescriptions per diagnosis to see if there was a difference in the prescribing habits before and after the pharmaceutical policy changes. Of note, several other pieces of data were collected from each medical record, including age, race, class of insurance (ie, Medicare, Medicaid, private health maintenance organization, private preferred provider organization), subtype diagnoses, and whether the prescription was new or a refill. The information gathered from the written record on the assessment and plan was verified using prescriptions ordered in the Allscripts electronic record, and any difference was noted. No identifying information that could be used to easily identify study participants was recorded.
Differences in prescribing habits across diagnoses before and after the policy changes were ascertained using a Fisher exact test and were further assessed using a mixed effects ordinal logistic regression model that accounted for within-provider clustering and baseline patient characteristics. An ordinal model was chosen to recognize differences in average cost among brand-name, generic, and OTC medications.
Results
In total, 200 medical records were collected. For the period analyzed before the policy change, 252 brand-name medications were prescribed compared to 231 prescribed for the period analyzed after the policy changes. There was insufficient evidence of an overall difference in brand-name medications prescribed before and after the policy changes (P=.145; Fisher exact test)(Table 1). There also was insufficient evidence of an overall difference in generic prescriptions, which totaled 153 before and 134 after the policy changes (P=.872; Fisher exact test)(Table 2). Over-the-counter prescriptions totaled 49 before and 69 after the policy changes. There was insufficient evidence of an overall difference before and after the policy changes for OTC medications (P=.192; Fisher exact test)(Table 3).




Comment
Although some medical institutions are diligently working to limit the potential influence pharmaceutical companies have on physician prescribing habits,4,5,25 the effect on physician prescribing habits is only now being established.15 Prior studies12,19,21 have found evidence that medication samples may lead to overuse of brand-name medications, but these findings do not hold true for the USF dermatologists included in this study, perhaps due to the difference in pharmaceutical company interactions or physicians maintaining prior prescription habits that were unrelated to the policy. Although this study focused on policy changes for in-office samples, prior studies either included other forms of interaction21 or did not include samples.22
Pharmaceutical samples allow patients to try a medication before committing to a long-term course of treatment with a particular medication, which has utility for physicians and patients. Although brand-name prescriptions may cost more, a trial period may assist the patient in deciding whether the medication is worth purchasing. Furthermore, physicians may feel more comfortable prescribing a medication once the individual patient has demonstrated a benefit from the sample, which may be particularly true in a specialty such as dermatology in which many branded topical medications contain a different vehicle than generic formulations, resulting in notable variations in active medication delivery and efficacy. Given the higher cost of branded topical medications, proving efficacy in patients through samples can provide a useful tool to the physician to determine the need for a branded formulation.
The benefits described are subjective but should not be disregarded. Although Hurley et al19 found that the number of brand-name medications prescribed increases as more samples are given out, our study demonstrated that after eliminating medication samples, there was no significant difference in the percentage of brand-name medications prescribed compared to generic and OTC medications.
Physician education concerning the price of each brand-name medication prescribed in office may be one method of reducing the amount of such prescriptions. Physicians generally are uninformed of the cost of the medications being prescribed26 and may not recognize the financial burden one medication may have compared to its alternative. However, educating physicians will empower them to make the conscious decision to prefer or not prefer a brand-name medication. With some generic medications shown to have a difference in bioequivalence compared to their brand-name counterparts, a physician may find more success prescribing the brand-name medications, regardless of pharmaceutical company influence, which is an alternative solution to policy changes that eliminate samples entirely. Although this study found insufficient evidence that removing samples decreases brand-name medication prescriptions, it is imperative that solutions are established to reduce the country’s increasing burden of medical costs.
Possible shortfalls of this study include the short period of time between which prepolicy data and postpolicy data were collected. It is possible that providers did not have enough time to adjust their prescribing habits or that providers would not have changed a prescribing pattern or preference simply because of a policy change. Future studies could allow a time period greater than 2 years to compare prepolicy and postpolicy prescribing habits, or a future study might make comparisons of prescriber patterns at different institutions that have different policies. Another possible shortfall is that providers and patients were limited to those at the Department of Dermatology & Cutaneous Surgery at the USF Morsani COM. Although this study has found insufficient evidence of a difference in prescribing habits, it may be beneficial to conduct a larger study that encompasses multiple academic institutions with similar policy changes. Most importantly, this study only investigated the influence of in-office pharmaceutical samples on prescribing patterns. This study did not look at the many other ways in which providers may be influenced by pharmaceutical companies, which likely is a significant confounding variable in this study. Continued additional studies that specifically examine other methods through which providers may be influenced would be helpful in further examining the many ways in which physician prescription habits are influenced.
Conclusion
Changes in pharmaceutical policy in 2011 at USF Morsani COM specifically banned in-office samples. The totality of evidence in this study shows modest observational evidence of a change in the postpolicy odds relative to prepolicy odds, but the data also are compatible with no change between prescribing habits before and after the policy changes. Further study is needed to fully understand this relationship.
Over the years, there has been growing concern about the relationship between physicians and pharmaceutical companies. Many studies have demonstrated that pharmaceutical interactions and incentives can influence physicians’ prescribing habits.1-3 As a result, many academic centers have adopted policies that attempt to limit the pharmaceutical industry’s influence on faculty and in-training physicians. Although these policies can vary greatly, they generally limit access of pharmaceutical representatives to providers and restrict pharmaceutical samples.4,5 This policy shift has even been reported in private practice.6
At the heart of the matter is the question: What really influences physicians to write a prescription for a particular medication? Is it cost, efficacy, or representatives pushing a product? Prior studies illustrate that generic medications are equivalent to their brand-name counterparts. In fact, current regulations require no more than 5% to 7% difference in bioequivalence.7-9 Although most generic medications are bioequivalent, it may not be universal.10
Garrison and Levin11 distributed a survey to US-based prescribers in family practice, psychiatry, and internal medicine and found that prescribers deemed patient response and success as the highest priority when determining which drugs to prescribe. In contrast, drug representatives and free samples only slightly contributed.11 Considering the minimum duration for efficacy of a medication such as an antidepressant vs a topical steroid, this pattern may differ with samples in dermatologic settings. Interestingly, another survey concluded that samples were associated with “sticky” prescribing habits, noting that physicians would prescribe a brand-name medication after using a sample, despite increased cost to the patient.12 Further, it has been suggested that recipients of free samples may experience increased costs in the long run, which contrasts a stated goal of affordability to patients.12,13
Physician interaction with pharmaceutical companies begins as early as medical school,14 with physicians reporting interactions as often as 4 times each month.14-18 Interactions can include meetings with pharmaceutical representatives, sponsored meals, gifts, continuing medical education sponsorship, funding for travel, pharmaceutical representative speakers, research funding, and drug samples.3
A 2014 study reported that prescribing habits are influenced by the free drug samples provided by nongeneric pharmaceutical companies.19 Nationally, the number of brand-name and branded generic medications constitute 79% of prescriptions, yet together they only comprise 17% of medications prescribed at an academic medical clinic that does not provide samples. The number of medications with samples being prescribed by dermatologists increased by 15% over 9 years, which may correlate with the wider availability of medication samples, more specifically an increase in branded generic samples.19 This potential interaction is the reason why institutions question the current influence of pharmaceutical companies. Samples may appear convenient, allowing a patient to test the medication prior to committing; however, with brand-name samples being provided to the physician, he/she may become more inclined to prescribe the branded medication.12,15,19-22 Because brand-name medications are more expensive than generic medications, this practice can increase the cost of health care.13 One study found that over 1 year, the overuse of nongeneric medications led to a loss of potential savings throughout 49 states, equating to $229 million just through Medicaid; interestingly, it was noted that in some states, a maximum reimbursement is set by Medicaid, regardless of whether the generic or branded medication is dispensed. The authors also noted variability in the potential savings by state, which may be a function of the state-by-state maximum reimbursements for certain medications.23 Another study on oral combination medications estimated Medicare spending on branded drugs relative to the cost if generic combinations had been purchased instead. This study examined branded medications for which the active components were available as over-the-counter (OTC), generic, or same-class generic, and the authors estimated that $925 million could have been saved in 2016 by purchasing a generic substitute.24 The overuse of nongeneric medications when generic alternatives are available becomes an issue that not only financially impacts patients but all taxpayers. However, this pattern may differ if limited only to dermatologic medications, which was not the focus of the prior studies.
To limit conflicts of interest in interactions with the pharmaceutical, medical device, and biotechnology industries, the University of South Florida (USF) Morsani College of Medicine (COM)(Tampa, Florida) implemented its own set of regulations that eliminated in-office pharmaceutical samples, in addition to other restrictions. This study aimed to investigate if there was a change in the prescribing habits of academic dermatologists after their medical school implemented these new policies.
We hypothesized that the number of brand-name drugs prescribed by physicians in the Department of Dermatology & Cutaneous Surgery would change following USF Morsani COM pharmaceutical policy changes. We sought to determine how physician prescribing practices within the Department of Dermatology & Cutaneous Surgery changed following USF Morsani COM pharmaceutical policy changes.
Methods
Data Collection
A retrospective review of medical records was conducted to investigate the effect of the USF Morsani COM pharmaceutical policy changes on physician prescribing practices within the Department of Dermatology & Cutaneous Surgery. Medical records of patients seen for common dermatology diagnoses before (January 1, 2010, to May 30, 2010) and after (August 1, 2011, to December 31, 2011) the pharmaceutical policy changes were reviewed, and all medications prescribed were recorded. Data were collected from medical records within the USF Health electronic medical record system and included visits with each of the department’s 3 attending dermatologists. The diagnoses included in the study—acne vulgaris, atopic dermatitis, onychomycosis, psoriasis, and rosacea—were chosen because in-office samples were available. Prescribing data from the first 100 consecutive medical records were collected from each time period, and a medical record was included only if it contained at least 1 of the following diagnoses: acne vulgaris, atopic dermatitis, onychomycosis, psoriasis, or rosacea. The assessment and plan of each progress note were reviewed, and the exact medication name and associated diagnosis were recorded for each prescription. Subsequently, each medication was reviewed and placed in 1 of 3 categories: brand name, generic, and OTC. The total number of prescriptions for each diagnosis (per visit/note); the specific number of brand, generic, and OTC medications prescribed (per visit/note); and the percentage of brand, generic, and OTC medications prescribed (per visit/note and per diagnosis in total) were calculated. To ensure only intended medications were included, each medication recorded in the medical record note was cross-referenced with the prescribed medication in the electronic medical record. The primary objective of this study was to capture the prescribing physician’s intent as proxied by the pattern of prescription. Thus, changes made in prescriptions after the initial plan—whether insurance related or otherwise—were not relevant to this investigation.
The data were collected to compare the percentage of brand vs generic or OTC prescriptions per diagnosis to see if there was a difference in the prescribing habits before and after the pharmaceutical policy changes. Of note, several other pieces of data were collected from each medical record, including age, race, class of insurance (ie, Medicare, Medicaid, private health maintenance organization, private preferred provider organization), subtype diagnoses, and whether the prescription was new or a refill. The information gathered from the written record on the assessment and plan was verified using prescriptions ordered in the Allscripts electronic record, and any difference was noted. No identifying information that could be used to easily identify study participants was recorded.
Differences in prescribing habits across diagnoses before and after the policy changes were ascertained using a Fisher exact test and were further assessed using a mixed effects ordinal logistic regression model that accounted for within-provider clustering and baseline patient characteristics. An ordinal model was chosen to recognize differences in average cost among brand-name, generic, and OTC medications.
Results
In total, 200 medical records were collected. For the period analyzed before the policy change, 252 brand-name medications were prescribed compared to 231 prescribed for the period analyzed after the policy changes. There was insufficient evidence of an overall difference in brand-name medications prescribed before and after the policy changes (P=.145; Fisher exact test)(Table 1). There also was insufficient evidence of an overall difference in generic prescriptions, which totaled 153 before and 134 after the policy changes (P=.872; Fisher exact test)(Table 2). Over-the-counter prescriptions totaled 49 before and 69 after the policy changes. There was insufficient evidence of an overall difference before and after the policy changes for OTC medications (P=.192; Fisher exact test)(Table 3).




Comment
Although some medical institutions are diligently working to limit the potential influence pharmaceutical companies have on physician prescribing habits,4,5,25 the effect on physician prescribing habits is only now being established.15 Prior studies12,19,21 have found evidence that medication samples may lead to overuse of brand-name medications, but these findings do not hold true for the USF dermatologists included in this study, perhaps due to the difference in pharmaceutical company interactions or physicians maintaining prior prescription habits that were unrelated to the policy. Although this study focused on policy changes for in-office samples, prior studies either included other forms of interaction21 or did not include samples.22
Pharmaceutical samples allow patients to try a medication before committing to a long-term course of treatment with a particular medication, which has utility for physicians and patients. Although brand-name prescriptions may cost more, a trial period may assist the patient in deciding whether the medication is worth purchasing. Furthermore, physicians may feel more comfortable prescribing a medication once the individual patient has demonstrated a benefit from the sample, which may be particularly true in a specialty such as dermatology in which many branded topical medications contain a different vehicle than generic formulations, resulting in notable variations in active medication delivery and efficacy. Given the higher cost of branded topical medications, proving efficacy in patients through samples can provide a useful tool to the physician to determine the need for a branded formulation.
The benefits described are subjective but should not be disregarded. Although Hurley et al19 found that the number of brand-name medications prescribed increases as more samples are given out, our study demonstrated that after eliminating medication samples, there was no significant difference in the percentage of brand-name medications prescribed compared to generic and OTC medications.
Physician education concerning the price of each brand-name medication prescribed in office may be one method of reducing the amount of such prescriptions. Physicians generally are uninformed of the cost of the medications being prescribed26 and may not recognize the financial burden one medication may have compared to its alternative. However, educating physicians will empower them to make the conscious decision to prefer or not prefer a brand-name medication. With some generic medications shown to have a difference in bioequivalence compared to their brand-name counterparts, a physician may find more success prescribing the brand-name medications, regardless of pharmaceutical company influence, which is an alternative solution to policy changes that eliminate samples entirely. Although this study found insufficient evidence that removing samples decreases brand-name medication prescriptions, it is imperative that solutions are established to reduce the country’s increasing burden of medical costs.
Possible shortfalls of this study include the short period of time between which prepolicy data and postpolicy data were collected. It is possible that providers did not have enough time to adjust their prescribing habits or that providers would not have changed a prescribing pattern or preference simply because of a policy change. Future studies could allow a time period greater than 2 years to compare prepolicy and postpolicy prescribing habits, or a future study might make comparisons of prescriber patterns at different institutions that have different policies. Another possible shortfall is that providers and patients were limited to those at the Department of Dermatology & Cutaneous Surgery at the USF Morsani COM. Although this study has found insufficient evidence of a difference in prescribing habits, it may be beneficial to conduct a larger study that encompasses multiple academic institutions with similar policy changes. Most importantly, this study only investigated the influence of in-office pharmaceutical samples on prescribing patterns. This study did not look at the many other ways in which providers may be influenced by pharmaceutical companies, which likely is a significant confounding variable in this study. Continued additional studies that specifically examine other methods through which providers may be influenced would be helpful in further examining the many ways in which physician prescription habits are influenced.
Conclusion
Changes in pharmaceutical policy in 2011 at USF Morsani COM specifically banned in-office samples. The totality of evidence in this study shows modest observational evidence of a change in the postpolicy odds relative to prepolicy odds, but the data also are compatible with no change between prescribing habits before and after the policy changes. Further study is needed to fully understand this relationship.
- Sondergaard J, Vach K, Kragstrup J, et al. Impact of pharmaceutical representative visits on GPs’ drug preferences. Fam Pract. 2009;26:204-209.
- Jelinek GA, Neate SL. The influence of the pharmaceutical industry in medicine. J Law Med. 2009;17:216-223.
- Wazana A. Physicians and the pharmaceutical industry: is a gift ever just a gift? JAMA. 2000;283:373-380.
- Coleman DL. Establishing policies for the relationship between industry and clinicians: lessons learned from two academic health centers. Acad Med. 2008;83:882-887.
- Coleman DL, Kazdin AE, Miller LA, et al. Guidelines for interactions between clinical faculty and the pharmaceutical industry: one medical school’s approach. Acad Med. 2006;81:154-160.
- Evans D, Hartung DM, Beasley D, et al. Breaking up is hard to do: lessons learned from a pharma-free practice transformation. J Am Board Fam Med. 2013;26:332-338.
- Davit BM, Nwakama PE, Buehler GJ, et al. Comparing generic and innovator drugs: a review of 12 years of bioequivalence data from the United States Food and Drug Administration. Ann Pharmacother. 2009;43:1583-1597.
- Kesselheim AS, Misono AS, Lee JL, et al. Clinical equivalence of generic and brand-name drugs used in cardiovascular disease: a systematic review and meta-analysis. JAMA. 2008;300:2514-2526.
- McCormack J, Chmelicek JT. Generic versus brand name: the other drug war. Can Fam Physician. 2014;60:911.
- Borgheini G. The bioequivalence and therapeutic efficacy of generic versus brand-name psychoactive drugs. Clin Ther. 2003;25:1578-1592.
- Garrison GD, Levin GM. Factors affecting prescribing of the newer antidepressants. Ann Pharmacother. 2000;34:10-14.
- Rafique S, Sarwar W, Rashid A, et al. Influence of free drug samples on prescribing by physicians: a cross sectional survey. J Pak Med Assoc. 2017;67:465-467.
- Alexander GC, Zhang J, Basu A. Characteristics of patients receiving pharmaceutical samples and association between sample receipt and out-of-pocket prescription costs. Med Care. 2008;46:394-402.
- Hodges B. Interactions with the pharmaceutical industry: experiences and attitudes of psychiatry residents, interns and clerks. CMAJ. 1995;153:553-559.
- Brotzman GL, Mark DH. The effect on resident attitudes of regulatory policies regarding pharmaceutical representative activities. J Gen Intern Med. 1993;8:130-134.
- Keim SM, Sanders AB, Witzke DB, et al. Beliefs and practices of emergency medicine faculty and residents regarding professional interactions with the biomedical industry. Ann Emerg Med. 1993;22:1576-1581.
- Thomson AN, Craig BJ, Barham PM. Attitudes of general practitioners in New Zealand to pharmaceutical representatives. Br J Gen Pract. 1994;44:220-223.
- Ziegler MG, Lew P, Singer BC. The accuracy of drug information from pharmaceutical sales representatives. JAMA. 1995;273:1296-1298.
- Hurley MP, Stafford RS, Lane AT. Characterizing the relationship between free drug samples and prescription patterns for acne vulgaris and rosacea. JAMA Dermatol. 2014;150:487-493.
- Lexchin J. Interactions between physicians and the pharmaceutical industry: what does the literature say? CMAJ. 1993;149:1401-1407.
- Lieb K, Scheurich A. Contact between doctors and the pharmaceutical industry, their perceptions, and the effects on prescribing habits. PLoS One. 2014;9:e110130.
- Spurling GK, Mansfield PR, Montgomery BD, et al. Information from pharmaceutical companies and the quality, quantity, and cost of physicians’ prescribing: a systematic review. PLoS Med. 2010;7:e1000352.
- Fischer MA, Avorn J. Economic consequences of underuse of generic drugs: evidence from Medicaid and implications for prescription drug benefit plans. Health Serv Res. 2003;38:1051-1064.
- Sacks CA, Lee CC, Kesselheim AS, et al. Medicare spending on brand-name combination medications vs their generic constituents. JAMA. 2018;320:650-656.
- Brennan TA, Rothman DJ, Blank L, et al. Health industry practices that create conflicts of interest: a policy proposal for academic medical centers. JAMA. 2006;295:429-433.
- Allan GM, Lexchin J, Wiebe N. Physician awareness of drug cost: a systematic review. PLoS Med. 2007;4:e283.
- Sondergaard J, Vach K, Kragstrup J, et al. Impact of pharmaceutical representative visits on GPs’ drug preferences. Fam Pract. 2009;26:204-209.
- Jelinek GA, Neate SL. The influence of the pharmaceutical industry in medicine. J Law Med. 2009;17:216-223.
- Wazana A. Physicians and the pharmaceutical industry: is a gift ever just a gift? JAMA. 2000;283:373-380.
- Coleman DL. Establishing policies for the relationship between industry and clinicians: lessons learned from two academic health centers. Acad Med. 2008;83:882-887.
- Coleman DL, Kazdin AE, Miller LA, et al. Guidelines for interactions between clinical faculty and the pharmaceutical industry: one medical school’s approach. Acad Med. 2006;81:154-160.
- Evans D, Hartung DM, Beasley D, et al. Breaking up is hard to do: lessons learned from a pharma-free practice transformation. J Am Board Fam Med. 2013;26:332-338.
- Davit BM, Nwakama PE, Buehler GJ, et al. Comparing generic and innovator drugs: a review of 12 years of bioequivalence data from the United States Food and Drug Administration. Ann Pharmacother. 2009;43:1583-1597.
- Kesselheim AS, Misono AS, Lee JL, et al. Clinical equivalence of generic and brand-name drugs used in cardiovascular disease: a systematic review and meta-analysis. JAMA. 2008;300:2514-2526.
- McCormack J, Chmelicek JT. Generic versus brand name: the other drug war. Can Fam Physician. 2014;60:911.
- Borgheini G. The bioequivalence and therapeutic efficacy of generic versus brand-name psychoactive drugs. Clin Ther. 2003;25:1578-1592.
- Garrison GD, Levin GM. Factors affecting prescribing of the newer antidepressants. Ann Pharmacother. 2000;34:10-14.
- Rafique S, Sarwar W, Rashid A, et al. Influence of free drug samples on prescribing by physicians: a cross sectional survey. J Pak Med Assoc. 2017;67:465-467.
- Alexander GC, Zhang J, Basu A. Characteristics of patients receiving pharmaceutical samples and association between sample receipt and out-of-pocket prescription costs. Med Care. 2008;46:394-402.
- Hodges B. Interactions with the pharmaceutical industry: experiences and attitudes of psychiatry residents, interns and clerks. CMAJ. 1995;153:553-559.
- Brotzman GL, Mark DH. The effect on resident attitudes of regulatory policies regarding pharmaceutical representative activities. J Gen Intern Med. 1993;8:130-134.
- Keim SM, Sanders AB, Witzke DB, et al. Beliefs and practices of emergency medicine faculty and residents regarding professional interactions with the biomedical industry. Ann Emerg Med. 1993;22:1576-1581.
- Thomson AN, Craig BJ, Barham PM. Attitudes of general practitioners in New Zealand to pharmaceutical representatives. Br J Gen Pract. 1994;44:220-223.
- Ziegler MG, Lew P, Singer BC. The accuracy of drug information from pharmaceutical sales representatives. JAMA. 1995;273:1296-1298.
- Hurley MP, Stafford RS, Lane AT. Characterizing the relationship between free drug samples and prescription patterns for acne vulgaris and rosacea. JAMA Dermatol. 2014;150:487-493.
- Lexchin J. Interactions between physicians and the pharmaceutical industry: what does the literature say? CMAJ. 1993;149:1401-1407.
- Lieb K, Scheurich A. Contact between doctors and the pharmaceutical industry, their perceptions, and the effects on prescribing habits. PLoS One. 2014;9:e110130.
- Spurling GK, Mansfield PR, Montgomery BD, et al. Information from pharmaceutical companies and the quality, quantity, and cost of physicians’ prescribing: a systematic review. PLoS Med. 2010;7:e1000352.
- Fischer MA, Avorn J. Economic consequences of underuse of generic drugs: evidence from Medicaid and implications for prescription drug benefit plans. Health Serv Res. 2003;38:1051-1064.
- Sacks CA, Lee CC, Kesselheim AS, et al. Medicare spending on brand-name combination medications vs their generic constituents. JAMA. 2018;320:650-656.
- Brennan TA, Rothman DJ, Blank L, et al. Health industry practices that create conflicts of interest: a policy proposal for academic medical centers. JAMA. 2006;295:429-433.
- Allan GM, Lexchin J, Wiebe N. Physician awareness of drug cost: a systematic review. PLoS Med. 2007;4:e283.
Practice Points
- There has been growing concern that pharmaceutical interactions and incentives can influence physicians’ prescribing habits.
- Many academic centers have adopted policies that attempt to limit the pharmaceutical industry’s influence on faculty and in-training physicians.
- This study aimed to investigate if there was a change in the prescribing habits of academic dermatologists after the medical school implemented new policies that banned in-office samples.
Systemic therapy options for pediatric skin diseases are improving
ORLANDO – Because Food and Drug Administration–approved treatment options for medications. However, this scenario is changing, A. Yasmine Kirkorian, MD, said at the ODAC Dermatology, Aesthetic & Surgical Conference.
“I really would like to emphasize that children with severe disease need to be treated,” added Dr. Kirkorian, a pediatric dermatologist at George Washington University, Washington, and Children’s National Health System, where she is interim chief of the division of dermatology.
Current on-label systemic therapies for pediatric skin disease include etanercept for psoriasis (4 years and older), ustekinumab for psoriasis (12 years and older), adalimumab for hidradenitis suppurativa (12 years and older), and omalizumab for chronic idiopathic urticaria (12 years and older). A new addition to the list is dupilumab, which was approved for children and adolescents with atopic dermatitis (AD) aged 12 years and older in 2019, she noted.
Dupilumab is currently being studied in children aged 6 months to 12 years, and other clinical trials are evaluating more options for pediatric patients with AD, alopecia areata, and psoriasis. They include a clinical trial of the oral Janus kinase 3 (JAK3) inhibitor PF-06651600 in patients aged 12 years and older with alopecia areata. Six biologic therapies are being evaluated for psoriasis in patients beginning at 6 years: ixekizumab, secukinumab, ustekinumab, guselkumab, brodalumab, and apremilast.
Some systemic therapies are off-label “but used all the time” for dermatologic diseases in pediatrics, Dr. Kirkorian noted. One example is methotrexate, which is approved by the FDA for acute lymphoblastic leukemia, meningeal leukemia, and juvenile idiopathic arthritis down to infancy. Having existing efficacy and safety data for a medication in a pediatric population, even for a different disease, can be helpful when counseling parents of children with severe dermatologic disease. “If you have something, even in an older population of children, it can be reassuring, or you can use evidence from other diseases,” she said.
While methotrexate is a cheap option and approved by the FDA for other pediatric indications down to infancy, the cons of using it to treat AD in pediatric patients are numerous. Treatment requires a number of blood draws for lab testing, which can be discouraging for younger patients, and the reported adverse effect profile may be concerning to some parents, while “in practice doesn’t really occur,” she said. Methotrexate is a teratogen so is not appropriate for teenagers who are sexually active and not using contraception.
The “biggest problem,” though, is the issue of whether methotrexate is effective, since it doesn’t always work for AD, Dr. Kirkorian said. “Even at the highest doses, I often feel that we fail the atopic children,” as opposed to using it to treat psoriasis, “where you know I’m going to get you on something that works.”
In contrast, cyclosporine is FDA approved down to infancy, and works quickly as a bridge to other therapy, and is not expensive, Dr. Kirkorian said. Cons include the need for blood draws, blood pressure checks, drug interactions, and adverse effects, she noted, adding that she tries to use cyclosporine as a bridge to on-label and off-label dupilumab.
Even with FDA approval for dupilumab down to age 12 years, she said it can be difficult to get insurance approval for the on-label treatment for patients in this age group with AD, before they first fail other therapies (even with off-label systemic drugs). For patients under age 12 years, getting approval is even more challenging and requires rigorous documentation of what therapies the child has failed, and how it has affected their quality of life, she said.
“If you send in a letter to the insurance company without an IGA [Investigator Global Assessment] or SCORAD, you’re going to get rejected,” Dr. Kirkorian said. In addition to those two measures, she provides “everything else,” including the impact of the disease on quality of life of patients, and school, she said, adding, “Did they miss school, did they get hospitalized for infections? And do they have comorbid diseases that might help you get approval?”
In pediatric patients with psoriasis, common issues are more likely to be about how insurance dictates step therapy. She has often found that young children may stop responding to etanercept after a few years, which can justify a switch to ustekinumab or a new treatment in a clinical trial, she said. Adolescents with psoriasis can receive ustekinumab, which is approved for psoriasis in patients aged 12-17 years, she said, noting that the infrequent ustekinumab dosing schedule is often beneficial in this population.
When all other approved options fail for young patients with psoriasis, justifying off-label use isn’t always easy. “You just have to make a justification based on the literature, even though it’s off label,” citing available safety information for other diseases, and “demonstrate over and over the impact on quality of life,” which works “most of the time,” Dr. Kirkorian said.
She reported having no conflicts of interest.
ORLANDO – Because Food and Drug Administration–approved treatment options for medications. However, this scenario is changing, A. Yasmine Kirkorian, MD, said at the ODAC Dermatology, Aesthetic & Surgical Conference.
“I really would like to emphasize that children with severe disease need to be treated,” added Dr. Kirkorian, a pediatric dermatologist at George Washington University, Washington, and Children’s National Health System, where she is interim chief of the division of dermatology.
Current on-label systemic therapies for pediatric skin disease include etanercept for psoriasis (4 years and older), ustekinumab for psoriasis (12 years and older), adalimumab for hidradenitis suppurativa (12 years and older), and omalizumab for chronic idiopathic urticaria (12 years and older). A new addition to the list is dupilumab, which was approved for children and adolescents with atopic dermatitis (AD) aged 12 years and older in 2019, she noted.
Dupilumab is currently being studied in children aged 6 months to 12 years, and other clinical trials are evaluating more options for pediatric patients with AD, alopecia areata, and psoriasis. They include a clinical trial of the oral Janus kinase 3 (JAK3) inhibitor PF-06651600 in patients aged 12 years and older with alopecia areata. Six biologic therapies are being evaluated for psoriasis in patients beginning at 6 years: ixekizumab, secukinumab, ustekinumab, guselkumab, brodalumab, and apremilast.
Some systemic therapies are off-label “but used all the time” for dermatologic diseases in pediatrics, Dr. Kirkorian noted. One example is methotrexate, which is approved by the FDA for acute lymphoblastic leukemia, meningeal leukemia, and juvenile idiopathic arthritis down to infancy. Having existing efficacy and safety data for a medication in a pediatric population, even for a different disease, can be helpful when counseling parents of children with severe dermatologic disease. “If you have something, even in an older population of children, it can be reassuring, or you can use evidence from other diseases,” she said.
While methotrexate is a cheap option and approved by the FDA for other pediatric indications down to infancy, the cons of using it to treat AD in pediatric patients are numerous. Treatment requires a number of blood draws for lab testing, which can be discouraging for younger patients, and the reported adverse effect profile may be concerning to some parents, while “in practice doesn’t really occur,” she said. Methotrexate is a teratogen so is not appropriate for teenagers who are sexually active and not using contraception.
The “biggest problem,” though, is the issue of whether methotrexate is effective, since it doesn’t always work for AD, Dr. Kirkorian said. “Even at the highest doses, I often feel that we fail the atopic children,” as opposed to using it to treat psoriasis, “where you know I’m going to get you on something that works.”
In contrast, cyclosporine is FDA approved down to infancy, and works quickly as a bridge to other therapy, and is not expensive, Dr. Kirkorian said. Cons include the need for blood draws, blood pressure checks, drug interactions, and adverse effects, she noted, adding that she tries to use cyclosporine as a bridge to on-label and off-label dupilumab.
Even with FDA approval for dupilumab down to age 12 years, she said it can be difficult to get insurance approval for the on-label treatment for patients in this age group with AD, before they first fail other therapies (even with off-label systemic drugs). For patients under age 12 years, getting approval is even more challenging and requires rigorous documentation of what therapies the child has failed, and how it has affected their quality of life, she said.
“If you send in a letter to the insurance company without an IGA [Investigator Global Assessment] or SCORAD, you’re going to get rejected,” Dr. Kirkorian said. In addition to those two measures, she provides “everything else,” including the impact of the disease on quality of life of patients, and school, she said, adding, “Did they miss school, did they get hospitalized for infections? And do they have comorbid diseases that might help you get approval?”
In pediatric patients with psoriasis, common issues are more likely to be about how insurance dictates step therapy. She has often found that young children may stop responding to etanercept after a few years, which can justify a switch to ustekinumab or a new treatment in a clinical trial, she said. Adolescents with psoriasis can receive ustekinumab, which is approved for psoriasis in patients aged 12-17 years, she said, noting that the infrequent ustekinumab dosing schedule is often beneficial in this population.
When all other approved options fail for young patients with psoriasis, justifying off-label use isn’t always easy. “You just have to make a justification based on the literature, even though it’s off label,” citing available safety information for other diseases, and “demonstrate over and over the impact on quality of life,” which works “most of the time,” Dr. Kirkorian said.
She reported having no conflicts of interest.
ORLANDO – Because Food and Drug Administration–approved treatment options for medications. However, this scenario is changing, A. Yasmine Kirkorian, MD, said at the ODAC Dermatology, Aesthetic & Surgical Conference.
“I really would like to emphasize that children with severe disease need to be treated,” added Dr. Kirkorian, a pediatric dermatologist at George Washington University, Washington, and Children’s National Health System, where she is interim chief of the division of dermatology.
Current on-label systemic therapies for pediatric skin disease include etanercept for psoriasis (4 years and older), ustekinumab for psoriasis (12 years and older), adalimumab for hidradenitis suppurativa (12 years and older), and omalizumab for chronic idiopathic urticaria (12 years and older). A new addition to the list is dupilumab, which was approved for children and adolescents with atopic dermatitis (AD) aged 12 years and older in 2019, she noted.
Dupilumab is currently being studied in children aged 6 months to 12 years, and other clinical trials are evaluating more options for pediatric patients with AD, alopecia areata, and psoriasis. They include a clinical trial of the oral Janus kinase 3 (JAK3) inhibitor PF-06651600 in patients aged 12 years and older with alopecia areata. Six biologic therapies are being evaluated for psoriasis in patients beginning at 6 years: ixekizumab, secukinumab, ustekinumab, guselkumab, brodalumab, and apremilast.
Some systemic therapies are off-label “but used all the time” for dermatologic diseases in pediatrics, Dr. Kirkorian noted. One example is methotrexate, which is approved by the FDA for acute lymphoblastic leukemia, meningeal leukemia, and juvenile idiopathic arthritis down to infancy. Having existing efficacy and safety data for a medication in a pediatric population, even for a different disease, can be helpful when counseling parents of children with severe dermatologic disease. “If you have something, even in an older population of children, it can be reassuring, or you can use evidence from other diseases,” she said.
While methotrexate is a cheap option and approved by the FDA for other pediatric indications down to infancy, the cons of using it to treat AD in pediatric patients are numerous. Treatment requires a number of blood draws for lab testing, which can be discouraging for younger patients, and the reported adverse effect profile may be concerning to some parents, while “in practice doesn’t really occur,” she said. Methotrexate is a teratogen so is not appropriate for teenagers who are sexually active and not using contraception.
The “biggest problem,” though, is the issue of whether methotrexate is effective, since it doesn’t always work for AD, Dr. Kirkorian said. “Even at the highest doses, I often feel that we fail the atopic children,” as opposed to using it to treat psoriasis, “where you know I’m going to get you on something that works.”
In contrast, cyclosporine is FDA approved down to infancy, and works quickly as a bridge to other therapy, and is not expensive, Dr. Kirkorian said. Cons include the need for blood draws, blood pressure checks, drug interactions, and adverse effects, she noted, adding that she tries to use cyclosporine as a bridge to on-label and off-label dupilumab.
Even with FDA approval for dupilumab down to age 12 years, she said it can be difficult to get insurance approval for the on-label treatment for patients in this age group with AD, before they first fail other therapies (even with off-label systemic drugs). For patients under age 12 years, getting approval is even more challenging and requires rigorous documentation of what therapies the child has failed, and how it has affected their quality of life, she said.
“If you send in a letter to the insurance company without an IGA [Investigator Global Assessment] or SCORAD, you’re going to get rejected,” Dr. Kirkorian said. In addition to those two measures, she provides “everything else,” including the impact of the disease on quality of life of patients, and school, she said, adding, “Did they miss school, did they get hospitalized for infections? And do they have comorbid diseases that might help you get approval?”
In pediatric patients with psoriasis, common issues are more likely to be about how insurance dictates step therapy. She has often found that young children may stop responding to etanercept after a few years, which can justify a switch to ustekinumab or a new treatment in a clinical trial, she said. Adolescents with psoriasis can receive ustekinumab, which is approved for psoriasis in patients aged 12-17 years, she said, noting that the infrequent ustekinumab dosing schedule is often beneficial in this population.
When all other approved options fail for young patients with psoriasis, justifying off-label use isn’t always easy. “You just have to make a justification based on the literature, even though it’s off label,” citing available safety information for other diseases, and “demonstrate over and over the impact on quality of life,” which works “most of the time,” Dr. Kirkorian said.
She reported having no conflicts of interest.
EXPERT ANALYSIS FROM ODAC 2020
Experts in Europe issue guidance on atopic dermatitis in pregnancy
MADRID – European atopic dermatitis experts have issued formal guidance on a seriously neglected topic: treatment of the disease during pregnancy, breastfeeding, and in men planning to father children.
The impetus for the project was clear: “,” Christian Vestergaard, MD, PhD, declared at a meeting of the European Task Force on Atopic Dermatitis held in conjunction with the annual congress of the European Academy of Dermatology and Venereology.
He presented highlights of the task force’s position paper on the topic, for which he served as first author. The group’s recommendations are based on expert opinion, since randomized clinical trial literature in this area is nonexistent because of ethical concerns. But the task force, comprising a who’s who in European dermatology, drew on a wealth of collective clinical experience in this area.
“We have all of Europe involved in doing this position statement. It’s meant as what we think is proper treatment and what we can say about the different drugs,” explained Dr. Vestergaard, a dermatologist at the University of Aarhus (Denmark).
Most nonobstetricians are intimidated by atopic dermatitis (AD) in pregnancy, and are concerned about the potential for treatment-related harm to the fetus. As a consequence, they are reluctant to recommend anything beyond weak class I topical corticosteroids and emollients. That’s clearly insufficient in light of the vast scope of need, he asserted. After all, AD affects 15%-20% of all children and persists or reappears in adulthood in one out of five of them. Half of those adults are women, many of whom will at some point wish to become pregnant. And many men with AD will eventually want to father children.
A key message from the task force is that untreated AD in pregnancy potentially places the mother and fetus at risk of serious complications, including Staphylococcus aureus infection and eczema herpeticum.
“If you take one thing away from our position paper, it’s that you can use class II or III topical corticosteroids in pregnant women as first-line therapy,” Dr. Vestergaard said.
This stance contradicts a longstanding widely held concern that topical steroids in pregnancy might increase the risk of facial cleft in the offspring, a worry that has been convincingly debunked in a Cochrane systematic review of 14 studies including more than 1.6 million pregnancies. The report concluded there was no association between topical corticosteroids of any potency with preterm delivery, birth defects, or low Apgar scores (Cochrane Database Syst Rev. 2015 Oct 26. doi: 10.1002/14651858.CD007346.pub3).
The task force recommends that if class II or III topical corticosteroid use in pregnancy exceeds 200 g/month, it’s worth considering add-on UV therapy, with narrow band UVB-311 nm as the regimen of choice; it can be used liberally. UV therapy with psoralens is not advised because of a theoretical risk of mutagenicity.
Product labeling for the topical calcineurin inhibitors declares that the agents should not be used during pregnancy. However, the European task force position paper takes issue with that and declares that topical tacrolimus (Protopic) can be considered an off-label first-line therapy in pregnant women with an insufficient response to liberal use of emollients. The same holds true for breastfeeding patients with AD. Just as when topical corticosteroids are used in the nipple area, topical tacrolimus should be applied after nursing, and the nipple area should be gently cleaned before nursing.
The rationale behind recommending topical tacrolimus as a first-line treatment is that systemic absorption of the drug is trivial. Plus, observational studies of oral tacrolimus in pregnant women who have received a solid organ transplant have shown no increase in congenital malformations.
The task force recommends against the use of topical pimecrolimus (Elidel) or crisaborole (Eucrisa) in pregnancy or lactation due to lack of clinical experience in these settings, Dr. Vestergaard continued.
The task force position is that chlorhexidine and other topical antiseptics – with the notable exception of triclosan – can be used in pregnancy to prevent recurrent skin infections. Aminoglycosides should be avoided, but topical fusidic acid is a reasonable antibiotic for treatment of small areas of clinically infected atopic dermatitis in pregnancy.
Systemic therapies
If disease control is insufficient with topical therapy, it’s appropriate to engage in shared decision-making with the patient regarding systemic treatment. She needs to understand up front that the worldwide overall background stillbirth rate in the general population is about 3%, and that severe congenital malformations are present in up to 6% of all live births.
“You need to inform them that they can have systemic therapy and give birth to a child with congenital defects which have nothing to do with the medication,” noted Dr. Vestergaard.
That said, the task force recommends cyclosporine as the off-label, first-line systemic therapy in pregnancy and lactation when long-term treatment is required. This guidance is based largely upon reassuring evidence in solid organ transplant recipients.
The recommended second-line therapy is systemic corticosteroids, but it’s a qualified recommendation. Dr. Vestergaard and colleagues find that systemic corticosteroid therapy is only rarely needed in pregnant AD patients, and the task force recommendation is to limit the use to less than 2-3 weeks and no more than 0.5 mg/kg per day of prednisone. Dexamethasone is not recommended.
Azathioprine should not be started in pregnancy, according to the task force, but when no other options are available, it may be continued in women already on the drug, albeit at half of the prepregnancy dose.
Dupilumab (Dupixent) is to be avoided in pregnant women with AD until more clinical experience becomes available.
Treatment of prospective fathers with AD
The European task force recommends that topical therapies can be prescribed in prospective fathers without any special concerns. The same is true for systemic corticosteroids. Methotrexate should be halted 3 months before planned pregnancy, as is the case for mycophenolate mofetil (CellCept). Azathioprine is recommended when other options have failed. Cyclosporine is deemed a reasonable option in the treatment of men with severe AD at the time of conception if other treatments have failed; of note, neither the Food and Drug Administration nor the European regulatory agency have issued contraindications for the use of the drug in men who wish to become fathers.
Mycophenolate mofetil carries a theoretical risk of teratogenicity. The European task force recommends that men should use condoms while on the drug and for at least 90 days afterward.
Unplanned pregnancy in women on systemic therapy
The recommended course of action is to immediately stop systemic therapy, intensify appropriate topical therapy in anticipation of worsening AD, and refer the patient to an obstetrician and a teratology information center for an individualized risk assessment. Methotrexate and mycophenolate mofetil are known teratogens.
The full 16-page task force position paper was published shortly before EADV 2019 (J Eur Acad Dermatol Venereol. 2019 Sep;33[9]:1644-59).
The report was developed without commercial sponsorship. Dr. Vestergaard indicated he has received research grants from and/or serves as a consultant to eight pharmaceutical companies.
Jenny Murase, MD, is with the department of dermatology, University of California, San Francisco, and is the director of medical consultative dermatology at the Palo Alto Foundation Medical Group, Mountain View, Calif. She has served on advisory boards for Dermira, Sanofi, and UCB; performed dermatologic consulting for UpToDate and Ferndale, and given nonbranded lectures for disease state management awareness for Regeneron and UCB.
Jenny Murase, MD, is with the department of dermatology, University of California, San Francisco, and is the director of medical consultative dermatology at the Palo Alto Foundation Medical Group, Mountain View, Calif. She has served on advisory boards for Dermira, Sanofi, and UCB; performed dermatologic consulting for UpToDate and Ferndale, and given nonbranded lectures for disease state management awareness for Regeneron and UCB.
Jenny Murase, MD, is with the department of dermatology, University of California, San Francisco, and is the director of medical consultative dermatology at the Palo Alto Foundation Medical Group, Mountain View, Calif. She has served on advisory boards for Dermira, Sanofi, and UCB; performed dermatologic consulting for UpToDate and Ferndale, and given nonbranded lectures for disease state management awareness for Regeneron and UCB.
MADRID – European atopic dermatitis experts have issued formal guidance on a seriously neglected topic: treatment of the disease during pregnancy, breastfeeding, and in men planning to father children.
The impetus for the project was clear: “,” Christian Vestergaard, MD, PhD, declared at a meeting of the European Task Force on Atopic Dermatitis held in conjunction with the annual congress of the European Academy of Dermatology and Venereology.
He presented highlights of the task force’s position paper on the topic, for which he served as first author. The group’s recommendations are based on expert opinion, since randomized clinical trial literature in this area is nonexistent because of ethical concerns. But the task force, comprising a who’s who in European dermatology, drew on a wealth of collective clinical experience in this area.
“We have all of Europe involved in doing this position statement. It’s meant as what we think is proper treatment and what we can say about the different drugs,” explained Dr. Vestergaard, a dermatologist at the University of Aarhus (Denmark).
Most nonobstetricians are intimidated by atopic dermatitis (AD) in pregnancy, and are concerned about the potential for treatment-related harm to the fetus. As a consequence, they are reluctant to recommend anything beyond weak class I topical corticosteroids and emollients. That’s clearly insufficient in light of the vast scope of need, he asserted. After all, AD affects 15%-20% of all children and persists or reappears in adulthood in one out of five of them. Half of those adults are women, many of whom will at some point wish to become pregnant. And many men with AD will eventually want to father children.
A key message from the task force is that untreated AD in pregnancy potentially places the mother and fetus at risk of serious complications, including Staphylococcus aureus infection and eczema herpeticum.
“If you take one thing away from our position paper, it’s that you can use class II or III topical corticosteroids in pregnant women as first-line therapy,” Dr. Vestergaard said.
This stance contradicts a longstanding widely held concern that topical steroids in pregnancy might increase the risk of facial cleft in the offspring, a worry that has been convincingly debunked in a Cochrane systematic review of 14 studies including more than 1.6 million pregnancies. The report concluded there was no association between topical corticosteroids of any potency with preterm delivery, birth defects, or low Apgar scores (Cochrane Database Syst Rev. 2015 Oct 26. doi: 10.1002/14651858.CD007346.pub3).
The task force recommends that if class II or III topical corticosteroid use in pregnancy exceeds 200 g/month, it’s worth considering add-on UV therapy, with narrow band UVB-311 nm as the regimen of choice; it can be used liberally. UV therapy with psoralens is not advised because of a theoretical risk of mutagenicity.
Product labeling for the topical calcineurin inhibitors declares that the agents should not be used during pregnancy. However, the European task force position paper takes issue with that and declares that topical tacrolimus (Protopic) can be considered an off-label first-line therapy in pregnant women with an insufficient response to liberal use of emollients. The same holds true for breastfeeding patients with AD. Just as when topical corticosteroids are used in the nipple area, topical tacrolimus should be applied after nursing, and the nipple area should be gently cleaned before nursing.
The rationale behind recommending topical tacrolimus as a first-line treatment is that systemic absorption of the drug is trivial. Plus, observational studies of oral tacrolimus in pregnant women who have received a solid organ transplant have shown no increase in congenital malformations.
The task force recommends against the use of topical pimecrolimus (Elidel) or crisaborole (Eucrisa) in pregnancy or lactation due to lack of clinical experience in these settings, Dr. Vestergaard continued.
The task force position is that chlorhexidine and other topical antiseptics – with the notable exception of triclosan – can be used in pregnancy to prevent recurrent skin infections. Aminoglycosides should be avoided, but topical fusidic acid is a reasonable antibiotic for treatment of small areas of clinically infected atopic dermatitis in pregnancy.
Systemic therapies
If disease control is insufficient with topical therapy, it’s appropriate to engage in shared decision-making with the patient regarding systemic treatment. She needs to understand up front that the worldwide overall background stillbirth rate in the general population is about 3%, and that severe congenital malformations are present in up to 6% of all live births.
“You need to inform them that they can have systemic therapy and give birth to a child with congenital defects which have nothing to do with the medication,” noted Dr. Vestergaard.
That said, the task force recommends cyclosporine as the off-label, first-line systemic therapy in pregnancy and lactation when long-term treatment is required. This guidance is based largely upon reassuring evidence in solid organ transplant recipients.
The recommended second-line therapy is systemic corticosteroids, but it’s a qualified recommendation. Dr. Vestergaard and colleagues find that systemic corticosteroid therapy is only rarely needed in pregnant AD patients, and the task force recommendation is to limit the use to less than 2-3 weeks and no more than 0.5 mg/kg per day of prednisone. Dexamethasone is not recommended.
Azathioprine should not be started in pregnancy, according to the task force, but when no other options are available, it may be continued in women already on the drug, albeit at half of the prepregnancy dose.
Dupilumab (Dupixent) is to be avoided in pregnant women with AD until more clinical experience becomes available.
Treatment of prospective fathers with AD
The European task force recommends that topical therapies can be prescribed in prospective fathers without any special concerns. The same is true for systemic corticosteroids. Methotrexate should be halted 3 months before planned pregnancy, as is the case for mycophenolate mofetil (CellCept). Azathioprine is recommended when other options have failed. Cyclosporine is deemed a reasonable option in the treatment of men with severe AD at the time of conception if other treatments have failed; of note, neither the Food and Drug Administration nor the European regulatory agency have issued contraindications for the use of the drug in men who wish to become fathers.
Mycophenolate mofetil carries a theoretical risk of teratogenicity. The European task force recommends that men should use condoms while on the drug and for at least 90 days afterward.
Unplanned pregnancy in women on systemic therapy
The recommended course of action is to immediately stop systemic therapy, intensify appropriate topical therapy in anticipation of worsening AD, and refer the patient to an obstetrician and a teratology information center for an individualized risk assessment. Methotrexate and mycophenolate mofetil are known teratogens.
The full 16-page task force position paper was published shortly before EADV 2019 (J Eur Acad Dermatol Venereol. 2019 Sep;33[9]:1644-59).
The report was developed without commercial sponsorship. Dr. Vestergaard indicated he has received research grants from and/or serves as a consultant to eight pharmaceutical companies.
MADRID – European atopic dermatitis experts have issued formal guidance on a seriously neglected topic: treatment of the disease during pregnancy, breastfeeding, and in men planning to father children.
The impetus for the project was clear: “,” Christian Vestergaard, MD, PhD, declared at a meeting of the European Task Force on Atopic Dermatitis held in conjunction with the annual congress of the European Academy of Dermatology and Venereology.
He presented highlights of the task force’s position paper on the topic, for which he served as first author. The group’s recommendations are based on expert opinion, since randomized clinical trial literature in this area is nonexistent because of ethical concerns. But the task force, comprising a who’s who in European dermatology, drew on a wealth of collective clinical experience in this area.
“We have all of Europe involved in doing this position statement. It’s meant as what we think is proper treatment and what we can say about the different drugs,” explained Dr. Vestergaard, a dermatologist at the University of Aarhus (Denmark).
Most nonobstetricians are intimidated by atopic dermatitis (AD) in pregnancy, and are concerned about the potential for treatment-related harm to the fetus. As a consequence, they are reluctant to recommend anything beyond weak class I topical corticosteroids and emollients. That’s clearly insufficient in light of the vast scope of need, he asserted. After all, AD affects 15%-20% of all children and persists or reappears in adulthood in one out of five of them. Half of those adults are women, many of whom will at some point wish to become pregnant. And many men with AD will eventually want to father children.
A key message from the task force is that untreated AD in pregnancy potentially places the mother and fetus at risk of serious complications, including Staphylococcus aureus infection and eczema herpeticum.
“If you take one thing away from our position paper, it’s that you can use class II or III topical corticosteroids in pregnant women as first-line therapy,” Dr. Vestergaard said.
This stance contradicts a longstanding widely held concern that topical steroids in pregnancy might increase the risk of facial cleft in the offspring, a worry that has been convincingly debunked in a Cochrane systematic review of 14 studies including more than 1.6 million pregnancies. The report concluded there was no association between topical corticosteroids of any potency with preterm delivery, birth defects, or low Apgar scores (Cochrane Database Syst Rev. 2015 Oct 26. doi: 10.1002/14651858.CD007346.pub3).
The task force recommends that if class II or III topical corticosteroid use in pregnancy exceeds 200 g/month, it’s worth considering add-on UV therapy, with narrow band UVB-311 nm as the regimen of choice; it can be used liberally. UV therapy with psoralens is not advised because of a theoretical risk of mutagenicity.
Product labeling for the topical calcineurin inhibitors declares that the agents should not be used during pregnancy. However, the European task force position paper takes issue with that and declares that topical tacrolimus (Protopic) can be considered an off-label first-line therapy in pregnant women with an insufficient response to liberal use of emollients. The same holds true for breastfeeding patients with AD. Just as when topical corticosteroids are used in the nipple area, topical tacrolimus should be applied after nursing, and the nipple area should be gently cleaned before nursing.
The rationale behind recommending topical tacrolimus as a first-line treatment is that systemic absorption of the drug is trivial. Plus, observational studies of oral tacrolimus in pregnant women who have received a solid organ transplant have shown no increase in congenital malformations.
The task force recommends against the use of topical pimecrolimus (Elidel) or crisaborole (Eucrisa) in pregnancy or lactation due to lack of clinical experience in these settings, Dr. Vestergaard continued.
The task force position is that chlorhexidine and other topical antiseptics – with the notable exception of triclosan – can be used in pregnancy to prevent recurrent skin infections. Aminoglycosides should be avoided, but topical fusidic acid is a reasonable antibiotic for treatment of small areas of clinically infected atopic dermatitis in pregnancy.
Systemic therapies
If disease control is insufficient with topical therapy, it’s appropriate to engage in shared decision-making with the patient regarding systemic treatment. She needs to understand up front that the worldwide overall background stillbirth rate in the general population is about 3%, and that severe congenital malformations are present in up to 6% of all live births.
“You need to inform them that they can have systemic therapy and give birth to a child with congenital defects which have nothing to do with the medication,” noted Dr. Vestergaard.
That said, the task force recommends cyclosporine as the off-label, first-line systemic therapy in pregnancy and lactation when long-term treatment is required. This guidance is based largely upon reassuring evidence in solid organ transplant recipients.
The recommended second-line therapy is systemic corticosteroids, but it’s a qualified recommendation. Dr. Vestergaard and colleagues find that systemic corticosteroid therapy is only rarely needed in pregnant AD patients, and the task force recommendation is to limit the use to less than 2-3 weeks and no more than 0.5 mg/kg per day of prednisone. Dexamethasone is not recommended.
Azathioprine should not be started in pregnancy, according to the task force, but when no other options are available, it may be continued in women already on the drug, albeit at half of the prepregnancy dose.
Dupilumab (Dupixent) is to be avoided in pregnant women with AD until more clinical experience becomes available.
Treatment of prospective fathers with AD
The European task force recommends that topical therapies can be prescribed in prospective fathers without any special concerns. The same is true for systemic corticosteroids. Methotrexate should be halted 3 months before planned pregnancy, as is the case for mycophenolate mofetil (CellCept). Azathioprine is recommended when other options have failed. Cyclosporine is deemed a reasonable option in the treatment of men with severe AD at the time of conception if other treatments have failed; of note, neither the Food and Drug Administration nor the European regulatory agency have issued contraindications for the use of the drug in men who wish to become fathers.
Mycophenolate mofetil carries a theoretical risk of teratogenicity. The European task force recommends that men should use condoms while on the drug and for at least 90 days afterward.
Unplanned pregnancy in women on systemic therapy
The recommended course of action is to immediately stop systemic therapy, intensify appropriate topical therapy in anticipation of worsening AD, and refer the patient to an obstetrician and a teratology information center for an individualized risk assessment. Methotrexate and mycophenolate mofetil are known teratogens.
The full 16-page task force position paper was published shortly before EADV 2019 (J Eur Acad Dermatol Venereol. 2019 Sep;33[9]:1644-59).
The report was developed without commercial sponsorship. Dr. Vestergaard indicated he has received research grants from and/or serves as a consultant to eight pharmaceutical companies.
REPORTING FROM THE EADV CONGRESS
Dupilumab-induced head and neck erythema described in atopic dermatitis patients
MADRID – A growing recognition that from their background skin disease emerged as a hot topic of discussion at a meeting of the European Task Force on Atopic Dermatitis held in conjunction with the annual congress of the European Academy of Dermatology and Venereology.
“During treatment with dupilumab, we saw something that is really different from the classic eczema that patients experienced prior to dupilumab, with no or minimal scaling, itch, or burning sensation. We do not believe this is a delayed effect of dupilumab on that specific region. We think this is a dupilumab-induced entity that we’re looking at. You should take home, in my opinion, that this is a common side effect that’s underreported in daily practice at this moment, and it’s not reported in clinical trials at all,” said Linde de Wijs, MD, of the department of dermatology, Erasmus University Medical Center in Rotterdam, the Netherlands.
She presented a detailed case series of seven affected patients which included histologic examination of lesional skin biopsies. The biopsies were characterized by a perivascular lymphohistiocytic infiltrate, an increase in ectatic capillaries in the papillary dermis, and a notable dearth of spongiosis, eosinophils, and neutrophils. Four patients had bulbous elongated rete ridges evocative of a psoriasiform dermatitis. The overall histologic picture was suggestive of a drug-induced skin reaction.
A striking finding was that, even though AD patients typically place high importance on achieving total clearing of disease on the head and neck, these seven closely studied patients nonetheless rated their treatment satisfaction as 9 out of a possible 10 points. Dr. de Wijs interpreted this as testimony to dupilumab’s potent efficacy and comparatively acceptable safety profile, especially the apparent side effect’s absence of scaling and itch.
“Remember, these are patients with really severe atopic dermatitis who’ve been treated with a lot of immunosuppressants prior to dupilumab,” she said.
Once the investigators began to suspect the existence of a novel dupilumab-induced skin reaction, they conducted a retrospective chart review of more than 150 patients treated with dupilumab (Dupixent) and determined that roughly 30% had developed this distinctive sharply demarcated patchy erythema on the head and neck characterized by absence of itch. The sequence involved clearance of the AD in response to dupilumab, followed by gradual development of the head and neck erythema 10-39 weeks after the start of treatment.
The erythema proved treatment refractory. Dr. de Wijs and her colleagues tried topical corticosteroids, including potent ones, as well as topical tacrolimus, antifungals, antibiotics, emollients, oral steroids, and antihistamines, to no avail. Patch testing to investigate allergic contact dermatitis as a possible etiology was unremarkable.
She hypothesized that, since dupilumab blocks the key signaling pathways for Th2 T-cell differentiation by targeting the interleukin-4 receptor alpha, it’s possible that the biologic promotes a shift towards activation of the Th17 pathway, which might explain the observed histologic findings.
The fact that this erythema wasn’t reported in the major randomized clinical trials of dupilumab underscores the enormous value of clinical practice registries, she said.
“We are not the only ones observing this phenomenon,” noted Dr. de Wijs, citing recently published reports by other investigators (J Am Acad Dermatol. 2020 Jan;82[1]:230-2; JAMA Dermatol. 2019 Jul 1;155[7]:850-2).
Indeed, her talk was immediately followed by a presentation by Sebastien Barbarot, MD, PhD, who reported on a French national retrospective study of head and neck dermatitis arising in patients on dupilumab that was conducted by the French Atopic Dermatitis Network using the organization’s GREAT database. Among 1,000 adult patients with AD treated with the biologic at 29 French centers, 10 developed a de novo head and neck dermatitis, and 32 others experienced more than 50% worsening of eczema signs on the head and neck from baseline beginning about 2 months after starting on dupilumab.
This 4.2% incidence is probably an underestimate, since dermatologists weren’t aware of the phenomenon and didn’t specifically ask patients about it, observed Dr. Barbarot, a dermatologist at the University of Nantes (France).
Among the key findings: No differences in clinical characteristics were found between the de novo and exacerbation groups, nearly half of affected patients had concomitant conjunctivitis, and seven patients discontinued dupilumab because of an intolerable burning sensation on the head/neck.
“I think this condition is quite different from rosacea,” Dr. Barbarot emphasized.
French dermatologists generally turned to topical corticosteroids or topical tacrolimus to treat the face and neck dermatitis, with mixed results; 22 of the 42 patients showed improvement and 8 worsened.
Marjolein de Bruin-Weller, MD, PhD, a dermatologist at Utrecht (the Netherlands) University and head of the Dutch National Eczema Expertise Center, said she and her colleagues have also encountered this dupilumab-related head and neck erythema and are convinced that a subset of affected patients have Malassezia-induced dermatitis with neutrophils present on lesional biopsies. “It responds very well to treatment. I think it’s very important to try itraconazole because sometimes it works,” she said.
Dr. de Wijs replied that she and her coworkers tried 2 weeks of itraconazole in several patients, with no effect. And none of their seven biopsied patients had an increase in neutrophils.
“It might be a very heterogenous polyform entity that we’re now observing,” she commented. Dr. de Bruin-Weller concurred.
Dr. Barbarot said he’d be interested in a formal study of antifungal therapy in patients with dupilumab-related head and neck dermatitis. Mechanistically, it seems plausible that dupilumab-induced activation of the TH17 pathway might lead to proliferation of Malassezia fungus.
Dr. de Wijs and Dr. Barbarot reported having no financial conflicts regarding their respective studies, which were conducted free of commercial sponsorship.
MADRID – A growing recognition that from their background skin disease emerged as a hot topic of discussion at a meeting of the European Task Force on Atopic Dermatitis held in conjunction with the annual congress of the European Academy of Dermatology and Venereology.
“During treatment with dupilumab, we saw something that is really different from the classic eczema that patients experienced prior to dupilumab, with no or minimal scaling, itch, or burning sensation. We do not believe this is a delayed effect of dupilumab on that specific region. We think this is a dupilumab-induced entity that we’re looking at. You should take home, in my opinion, that this is a common side effect that’s underreported in daily practice at this moment, and it’s not reported in clinical trials at all,” said Linde de Wijs, MD, of the department of dermatology, Erasmus University Medical Center in Rotterdam, the Netherlands.
She presented a detailed case series of seven affected patients which included histologic examination of lesional skin biopsies. The biopsies were characterized by a perivascular lymphohistiocytic infiltrate, an increase in ectatic capillaries in the papillary dermis, and a notable dearth of spongiosis, eosinophils, and neutrophils. Four patients had bulbous elongated rete ridges evocative of a psoriasiform dermatitis. The overall histologic picture was suggestive of a drug-induced skin reaction.
A striking finding was that, even though AD patients typically place high importance on achieving total clearing of disease on the head and neck, these seven closely studied patients nonetheless rated their treatment satisfaction as 9 out of a possible 10 points. Dr. de Wijs interpreted this as testimony to dupilumab’s potent efficacy and comparatively acceptable safety profile, especially the apparent side effect’s absence of scaling and itch.
“Remember, these are patients with really severe atopic dermatitis who’ve been treated with a lot of immunosuppressants prior to dupilumab,” she said.
Once the investigators began to suspect the existence of a novel dupilumab-induced skin reaction, they conducted a retrospective chart review of more than 150 patients treated with dupilumab (Dupixent) and determined that roughly 30% had developed this distinctive sharply demarcated patchy erythema on the head and neck characterized by absence of itch. The sequence involved clearance of the AD in response to dupilumab, followed by gradual development of the head and neck erythema 10-39 weeks after the start of treatment.
The erythema proved treatment refractory. Dr. de Wijs and her colleagues tried topical corticosteroids, including potent ones, as well as topical tacrolimus, antifungals, antibiotics, emollients, oral steroids, and antihistamines, to no avail. Patch testing to investigate allergic contact dermatitis as a possible etiology was unremarkable.
She hypothesized that, since dupilumab blocks the key signaling pathways for Th2 T-cell differentiation by targeting the interleukin-4 receptor alpha, it’s possible that the biologic promotes a shift towards activation of the Th17 pathway, which might explain the observed histologic findings.
The fact that this erythema wasn’t reported in the major randomized clinical trials of dupilumab underscores the enormous value of clinical practice registries, she said.
“We are not the only ones observing this phenomenon,” noted Dr. de Wijs, citing recently published reports by other investigators (J Am Acad Dermatol. 2020 Jan;82[1]:230-2; JAMA Dermatol. 2019 Jul 1;155[7]:850-2).
Indeed, her talk was immediately followed by a presentation by Sebastien Barbarot, MD, PhD, who reported on a French national retrospective study of head and neck dermatitis arising in patients on dupilumab that was conducted by the French Atopic Dermatitis Network using the organization’s GREAT database. Among 1,000 adult patients with AD treated with the biologic at 29 French centers, 10 developed a de novo head and neck dermatitis, and 32 others experienced more than 50% worsening of eczema signs on the head and neck from baseline beginning about 2 months after starting on dupilumab.
This 4.2% incidence is probably an underestimate, since dermatologists weren’t aware of the phenomenon and didn’t specifically ask patients about it, observed Dr. Barbarot, a dermatologist at the University of Nantes (France).
Among the key findings: No differences in clinical characteristics were found between the de novo and exacerbation groups, nearly half of affected patients had concomitant conjunctivitis, and seven patients discontinued dupilumab because of an intolerable burning sensation on the head/neck.
“I think this condition is quite different from rosacea,” Dr. Barbarot emphasized.
French dermatologists generally turned to topical corticosteroids or topical tacrolimus to treat the face and neck dermatitis, with mixed results; 22 of the 42 patients showed improvement and 8 worsened.
Marjolein de Bruin-Weller, MD, PhD, a dermatologist at Utrecht (the Netherlands) University and head of the Dutch National Eczema Expertise Center, said she and her colleagues have also encountered this dupilumab-related head and neck erythema and are convinced that a subset of affected patients have Malassezia-induced dermatitis with neutrophils present on lesional biopsies. “It responds very well to treatment. I think it’s very important to try itraconazole because sometimes it works,” she said.
Dr. de Wijs replied that she and her coworkers tried 2 weeks of itraconazole in several patients, with no effect. And none of their seven biopsied patients had an increase in neutrophils.
“It might be a very heterogenous polyform entity that we’re now observing,” she commented. Dr. de Bruin-Weller concurred.
Dr. Barbarot said he’d be interested in a formal study of antifungal therapy in patients with dupilumab-related head and neck dermatitis. Mechanistically, it seems plausible that dupilumab-induced activation of the TH17 pathway might lead to proliferation of Malassezia fungus.
Dr. de Wijs and Dr. Barbarot reported having no financial conflicts regarding their respective studies, which were conducted free of commercial sponsorship.
MADRID – A growing recognition that from their background skin disease emerged as a hot topic of discussion at a meeting of the European Task Force on Atopic Dermatitis held in conjunction with the annual congress of the European Academy of Dermatology and Venereology.
“During treatment with dupilumab, we saw something that is really different from the classic eczema that patients experienced prior to dupilumab, with no or minimal scaling, itch, or burning sensation. We do not believe this is a delayed effect of dupilumab on that specific region. We think this is a dupilumab-induced entity that we’re looking at. You should take home, in my opinion, that this is a common side effect that’s underreported in daily practice at this moment, and it’s not reported in clinical trials at all,” said Linde de Wijs, MD, of the department of dermatology, Erasmus University Medical Center in Rotterdam, the Netherlands.
She presented a detailed case series of seven affected patients which included histologic examination of lesional skin biopsies. The biopsies were characterized by a perivascular lymphohistiocytic infiltrate, an increase in ectatic capillaries in the papillary dermis, and a notable dearth of spongiosis, eosinophils, and neutrophils. Four patients had bulbous elongated rete ridges evocative of a psoriasiform dermatitis. The overall histologic picture was suggestive of a drug-induced skin reaction.
A striking finding was that, even though AD patients typically place high importance on achieving total clearing of disease on the head and neck, these seven closely studied patients nonetheless rated their treatment satisfaction as 9 out of a possible 10 points. Dr. de Wijs interpreted this as testimony to dupilumab’s potent efficacy and comparatively acceptable safety profile, especially the apparent side effect’s absence of scaling and itch.
“Remember, these are patients with really severe atopic dermatitis who’ve been treated with a lot of immunosuppressants prior to dupilumab,” she said.
Once the investigators began to suspect the existence of a novel dupilumab-induced skin reaction, they conducted a retrospective chart review of more than 150 patients treated with dupilumab (Dupixent) and determined that roughly 30% had developed this distinctive sharply demarcated patchy erythema on the head and neck characterized by absence of itch. The sequence involved clearance of the AD in response to dupilumab, followed by gradual development of the head and neck erythema 10-39 weeks after the start of treatment.
The erythema proved treatment refractory. Dr. de Wijs and her colleagues tried topical corticosteroids, including potent ones, as well as topical tacrolimus, antifungals, antibiotics, emollients, oral steroids, and antihistamines, to no avail. Patch testing to investigate allergic contact dermatitis as a possible etiology was unremarkable.
She hypothesized that, since dupilumab blocks the key signaling pathways for Th2 T-cell differentiation by targeting the interleukin-4 receptor alpha, it’s possible that the biologic promotes a shift towards activation of the Th17 pathway, which might explain the observed histologic findings.
The fact that this erythema wasn’t reported in the major randomized clinical trials of dupilumab underscores the enormous value of clinical practice registries, she said.
“We are not the only ones observing this phenomenon,” noted Dr. de Wijs, citing recently published reports by other investigators (J Am Acad Dermatol. 2020 Jan;82[1]:230-2; JAMA Dermatol. 2019 Jul 1;155[7]:850-2).
Indeed, her talk was immediately followed by a presentation by Sebastien Barbarot, MD, PhD, who reported on a French national retrospective study of head and neck dermatitis arising in patients on dupilumab that was conducted by the French Atopic Dermatitis Network using the organization’s GREAT database. Among 1,000 adult patients with AD treated with the biologic at 29 French centers, 10 developed a de novo head and neck dermatitis, and 32 others experienced more than 50% worsening of eczema signs on the head and neck from baseline beginning about 2 months after starting on dupilumab.
This 4.2% incidence is probably an underestimate, since dermatologists weren’t aware of the phenomenon and didn’t specifically ask patients about it, observed Dr. Barbarot, a dermatologist at the University of Nantes (France).
Among the key findings: No differences in clinical characteristics were found between the de novo and exacerbation groups, nearly half of affected patients had concomitant conjunctivitis, and seven patients discontinued dupilumab because of an intolerable burning sensation on the head/neck.
“I think this condition is quite different from rosacea,” Dr. Barbarot emphasized.
French dermatologists generally turned to topical corticosteroids or topical tacrolimus to treat the face and neck dermatitis, with mixed results; 22 of the 42 patients showed improvement and 8 worsened.
Marjolein de Bruin-Weller, MD, PhD, a dermatologist at Utrecht (the Netherlands) University and head of the Dutch National Eczema Expertise Center, said she and her colleagues have also encountered this dupilumab-related head and neck erythema and are convinced that a subset of affected patients have Malassezia-induced dermatitis with neutrophils present on lesional biopsies. “It responds very well to treatment. I think it’s very important to try itraconazole because sometimes it works,” she said.
Dr. de Wijs replied that she and her coworkers tried 2 weeks of itraconazole in several patients, with no effect. And none of their seven biopsied patients had an increase in neutrophils.
“It might be a very heterogenous polyform entity that we’re now observing,” she commented. Dr. de Bruin-Weller concurred.
Dr. Barbarot said he’d be interested in a formal study of antifungal therapy in patients with dupilumab-related head and neck dermatitis. Mechanistically, it seems plausible that dupilumab-induced activation of the TH17 pathway might lead to proliferation of Malassezia fungus.
Dr. de Wijs and Dr. Barbarot reported having no financial conflicts regarding their respective studies, which were conducted free of commercial sponsorship.
REPORTING FROM THE EADV CONGRESS
Efficacy and safety of lowering dupilumab frequency for AD
Patients with moderate to severe atopic dermatitis who responded well to the approved dupilumab regimen of 300 mg every 2 weeks in pivotal phase 3 monotherapy trials were more likely to have a continued response over the longer term if they maintained this regimen rather than switching to longer dosing intervals or discontinuing the medication.
This finding comes from a 36-week, randomized, double-blind, placebo-controlled trial that enrolled 422 – SOLO 1 and SOLO 2.
The new international study – SOLO-CONTINUE – randomized these patients to continue the original regimen (weekly or every 2 weeks), to receive 300 mg of the biologic medication every 4 or 8 weeks, or to receive placebo.
Patients who continued the original regimen had the most consistent maintenance of treatment effect, while patients on longer dosage intervals or placebo had a dose-dependent reduction in response and no safety advantage. The incidence of treatment-emergent antidrug antibody was lowest with dupilumab weekly or every 2 weeks, and slightly higher with less-frequent dosing intervals, reported Margitta Worm, MD, of the Charité Universitätsmedizin Berlin, and coinvestigators.
“Because administration every 4 weeks or every 8 weeks did not provide an additional safety advantage and was numerically outperformed by administration weekly or every 2 weeks, we believe that it is prudent to adhere to the approved every 2 weeks regimen for adults and avoid less frequent treatment regimens (every 4 weeks or every 8 weeks) for long-term maintenance of efficacy,” they wrote in JAMA Dermatology.
Treatment success in the original SOLO trials was defined as having achieved an Investigator’s Global Assessment score of 0-1, or 75% improvement in Eczema Area and Severity Index Scores (EASI-75). As primary endpoints, SOLO-CONTINUE looked at the mean percentage change in EASI score over the course of the trial, and the percentage of patients who maintained EASI-75 at week 36.
Patients in the SOLO-CONTINUE trial who were randomized to receive dupilumab weekly or every 2 weeks had a mean percent change in EASI score of –0.06%. In contrast, patients assigned to the placebo group had a 21.7% decrease, and those taking the medication at 4- and 8-week intervals had mean changes of –3.84% and –6.84%, respectively. Post hoc analyses showed no apparent difference between dupilumab weekly and every 2 weeks in the maintenance of clinical response, the investigators reported.
Among patients with EASI-75 response at baseline, significantly more patients maintained this response at week 36 than patients receiving placebo, and there was again an apparent dose-dependent response. The percentage with EASI-75 at week 36 was 71.6% with the weekly or every-2-weeks regimen, 58.3% with the 4-week regimen, 54.9% with the 8-week regimen, and 30.4% in the placebo group.
Continuing treatment with 300 mg weekly or every 2 weeks resulted in greater maintenance of response across multiple other clinical endpoints and patient-reported outcomes as well (such as pruritus, atopic dermatitis symptoms, sleep, pain or discomfort, quality of life, and symptoms of anxiety and depression).
The more-frequent regimens also conferred no greater risk than less-frequent administration, and there were no new safety signals over the 36-week trial. Treatment-emergent adverse events (the most common were headache, nasopharyngitis, injection-site reactions, and herpes simplex virus infection) occurred in 70.7% of patients in the weekly or every-2-weeks group, 73.6% in the 4-week group, 75% in the 8-week group, and 81.7% in the placebo group.
Unlike earlier studies, the incidence of conjunctivitis was low (less than 6%) across all treatment groups, possibly because patients in the SOLO-CONTINUE trial were all high-level responders who tend to have conjunctivitis less frequently, the authors wrote.
Patients receiving less-frequent doses of dupilumab, particularly every 8 weeks, had greater rates of skin infections, flares, and rescue medication use than patients receiving doses weekly or every 2 weeks, the investigators reported. Treatment-emergent antidrug antibody incidence was slightly higher with less-frequent doses (11.7% and 6% in the 8-week and 4-week groups, respectively, compared with 4.3% and 1.2% in the every-2-weeks and weekly groups), which indicates a “higher incidence of immunogenicity with less-frequent dosage intervals” and is “consistent with other biologics,” they wrote.
Dupilumab is a human monoclonal antibody against the interleukin-4 receptor alpha that inhibits signaling of IL-4 and IL-13. The study was conducted at 185 sites in North America, Europe, Asia, and Japan. Patients had a mean age of 38.2 years; 53.8% were male.
While the trial suggests that the approved regimen of 300 mg every 2 weeks is best for long-term treatment, “therapeutic decisions are often influenced by cost-benefit considerations, in which case practitioners and other stakeholders involved in these decisions should carefully balance potential savings against suboptimal efficacy and long-term risks associated with discontinuous treatment regimens,” the investigators wrote.
The SOLO-CONTINUE trial was funded by Sanofi and Regeneron, the companies that market dupilumab. Dr. Worm reported receiving honoraria for consulting and lecture activity from Regeneron and Sanofi during and outside the conduct of the study, among other disclosures. The other authors had multiple disclosures related to these and multiple other pharmaceutical companies, or were employees of Sanofi or Regeneron.
SOURCE: Worm M et al. JAMA Dermatol. 2019 Dec 26. doi: 10.1001/jamadermatol.2019.3617.
The desire to decrease or stop a therapy such as dupilumab may be motivated by cost, current and potential adverse effects, and individual needs. Because atopic dermatitis is a waxing-and-waning disease with a predilection for cycles of escalation, there is some thought a priori that reduced treatment schedules or discontinued use of a drug may be possible in a state of low disease activity.
The investigators of the SOLO-CONTINUE trial found, however, that continuous treatment with the dosage used in the original SOLO trials (300 mg weekly or every 2 weeks) resulted in a better maintenance of response than a less-frequent dosage and was significantly better than placebo for all endpoints. The less-frequent dosage regimens (every 4 weeks and every 8 weeks), on the other hand, produced some dose-dependent reduction in efficacy.
The development of antidrug antibodies was found in approximately 11% of individuals who received placebo or dupilumab every 8 weeks, 6% of the monthly treatment group, and only 1% in the weekly group, suggesting that less-frequent administration results in higher immunogenicity. However, most of the antidrug antibody levels were low and did not seem to have any clinical effect, making this finding of uncertain relevance to patient care.
The study is valuable because, as more patients are exposed to the drug, more will want or need to reduce the dosage or stop use over time – and although it seems optimal to continue an every-2-weeks treatment regimen, this may not always be feasible. As we integrate new therapies and learn more about atopic dermatitis, it is important that we understand the options and implications around decreasing the dosage of dupilumab. This newly concluded trial is helpful in this regard.
Peter A. Lio, MD, is with the department of dermatology at Northwestern University, Chicago, and the Chicago Integrative Eczema Center. He reported receiving grants and personal fees from Regeneron, Sanofi Genzyme, and other companies, as well as other disclosures. His comments appear in JAMA Dermatology (2019 Dec 26. doi: 10.1001/jamadermatol.2019.3331).
The desire to decrease or stop a therapy such as dupilumab may be motivated by cost, current and potential adverse effects, and individual needs. Because atopic dermatitis is a waxing-and-waning disease with a predilection for cycles of escalation, there is some thought a priori that reduced treatment schedules or discontinued use of a drug may be possible in a state of low disease activity.
The investigators of the SOLO-CONTINUE trial found, however, that continuous treatment with the dosage used in the original SOLO trials (300 mg weekly or every 2 weeks) resulted in a better maintenance of response than a less-frequent dosage and was significantly better than placebo for all endpoints. The less-frequent dosage regimens (every 4 weeks and every 8 weeks), on the other hand, produced some dose-dependent reduction in efficacy.
The development of antidrug antibodies was found in approximately 11% of individuals who received placebo or dupilumab every 8 weeks, 6% of the monthly treatment group, and only 1% in the weekly group, suggesting that less-frequent administration results in higher immunogenicity. However, most of the antidrug antibody levels were low and did not seem to have any clinical effect, making this finding of uncertain relevance to patient care.
The study is valuable because, as more patients are exposed to the drug, more will want or need to reduce the dosage or stop use over time – and although it seems optimal to continue an every-2-weeks treatment regimen, this may not always be feasible. As we integrate new therapies and learn more about atopic dermatitis, it is important that we understand the options and implications around decreasing the dosage of dupilumab. This newly concluded trial is helpful in this regard.
Peter A. Lio, MD, is with the department of dermatology at Northwestern University, Chicago, and the Chicago Integrative Eczema Center. He reported receiving grants and personal fees from Regeneron, Sanofi Genzyme, and other companies, as well as other disclosures. His comments appear in JAMA Dermatology (2019 Dec 26. doi: 10.1001/jamadermatol.2019.3331).
The desire to decrease or stop a therapy such as dupilumab may be motivated by cost, current and potential adverse effects, and individual needs. Because atopic dermatitis is a waxing-and-waning disease with a predilection for cycles of escalation, there is some thought a priori that reduced treatment schedules or discontinued use of a drug may be possible in a state of low disease activity.
The investigators of the SOLO-CONTINUE trial found, however, that continuous treatment with the dosage used in the original SOLO trials (300 mg weekly or every 2 weeks) resulted in a better maintenance of response than a less-frequent dosage and was significantly better than placebo for all endpoints. The less-frequent dosage regimens (every 4 weeks and every 8 weeks), on the other hand, produced some dose-dependent reduction in efficacy.
The development of antidrug antibodies was found in approximately 11% of individuals who received placebo or dupilumab every 8 weeks, 6% of the monthly treatment group, and only 1% in the weekly group, suggesting that less-frequent administration results in higher immunogenicity. However, most of the antidrug antibody levels were low and did not seem to have any clinical effect, making this finding of uncertain relevance to patient care.
The study is valuable because, as more patients are exposed to the drug, more will want or need to reduce the dosage or stop use over time – and although it seems optimal to continue an every-2-weeks treatment regimen, this may not always be feasible. As we integrate new therapies and learn more about atopic dermatitis, it is important that we understand the options and implications around decreasing the dosage of dupilumab. This newly concluded trial is helpful in this regard.
Peter A. Lio, MD, is with the department of dermatology at Northwestern University, Chicago, and the Chicago Integrative Eczema Center. He reported receiving grants and personal fees from Regeneron, Sanofi Genzyme, and other companies, as well as other disclosures. His comments appear in JAMA Dermatology (2019 Dec 26. doi: 10.1001/jamadermatol.2019.3331).
Patients with moderate to severe atopic dermatitis who responded well to the approved dupilumab regimen of 300 mg every 2 weeks in pivotal phase 3 monotherapy trials were more likely to have a continued response over the longer term if they maintained this regimen rather than switching to longer dosing intervals or discontinuing the medication.
This finding comes from a 36-week, randomized, double-blind, placebo-controlled trial that enrolled 422 – SOLO 1 and SOLO 2.
The new international study – SOLO-CONTINUE – randomized these patients to continue the original regimen (weekly or every 2 weeks), to receive 300 mg of the biologic medication every 4 or 8 weeks, or to receive placebo.
Patients who continued the original regimen had the most consistent maintenance of treatment effect, while patients on longer dosage intervals or placebo had a dose-dependent reduction in response and no safety advantage. The incidence of treatment-emergent antidrug antibody was lowest with dupilumab weekly or every 2 weeks, and slightly higher with less-frequent dosing intervals, reported Margitta Worm, MD, of the Charité Universitätsmedizin Berlin, and coinvestigators.
“Because administration every 4 weeks or every 8 weeks did not provide an additional safety advantage and was numerically outperformed by administration weekly or every 2 weeks, we believe that it is prudent to adhere to the approved every 2 weeks regimen for adults and avoid less frequent treatment regimens (every 4 weeks or every 8 weeks) for long-term maintenance of efficacy,” they wrote in JAMA Dermatology.
Treatment success in the original SOLO trials was defined as having achieved an Investigator’s Global Assessment score of 0-1, or 75% improvement in Eczema Area and Severity Index Scores (EASI-75). As primary endpoints, SOLO-CONTINUE looked at the mean percentage change in EASI score over the course of the trial, and the percentage of patients who maintained EASI-75 at week 36.
Patients in the SOLO-CONTINUE trial who were randomized to receive dupilumab weekly or every 2 weeks had a mean percent change in EASI score of –0.06%. In contrast, patients assigned to the placebo group had a 21.7% decrease, and those taking the medication at 4- and 8-week intervals had mean changes of –3.84% and –6.84%, respectively. Post hoc analyses showed no apparent difference between dupilumab weekly and every 2 weeks in the maintenance of clinical response, the investigators reported.
Among patients with EASI-75 response at baseline, significantly more patients maintained this response at week 36 than patients receiving placebo, and there was again an apparent dose-dependent response. The percentage with EASI-75 at week 36 was 71.6% with the weekly or every-2-weeks regimen, 58.3% with the 4-week regimen, 54.9% with the 8-week regimen, and 30.4% in the placebo group.
Continuing treatment with 300 mg weekly or every 2 weeks resulted in greater maintenance of response across multiple other clinical endpoints and patient-reported outcomes as well (such as pruritus, atopic dermatitis symptoms, sleep, pain or discomfort, quality of life, and symptoms of anxiety and depression).
The more-frequent regimens also conferred no greater risk than less-frequent administration, and there were no new safety signals over the 36-week trial. Treatment-emergent adverse events (the most common were headache, nasopharyngitis, injection-site reactions, and herpes simplex virus infection) occurred in 70.7% of patients in the weekly or every-2-weeks group, 73.6% in the 4-week group, 75% in the 8-week group, and 81.7% in the placebo group.
Unlike earlier studies, the incidence of conjunctivitis was low (less than 6%) across all treatment groups, possibly because patients in the SOLO-CONTINUE trial were all high-level responders who tend to have conjunctivitis less frequently, the authors wrote.
Patients receiving less-frequent doses of dupilumab, particularly every 8 weeks, had greater rates of skin infections, flares, and rescue medication use than patients receiving doses weekly or every 2 weeks, the investigators reported. Treatment-emergent antidrug antibody incidence was slightly higher with less-frequent doses (11.7% and 6% in the 8-week and 4-week groups, respectively, compared with 4.3% and 1.2% in the every-2-weeks and weekly groups), which indicates a “higher incidence of immunogenicity with less-frequent dosage intervals” and is “consistent with other biologics,” they wrote.
Dupilumab is a human monoclonal antibody against the interleukin-4 receptor alpha that inhibits signaling of IL-4 and IL-13. The study was conducted at 185 sites in North America, Europe, Asia, and Japan. Patients had a mean age of 38.2 years; 53.8% were male.
While the trial suggests that the approved regimen of 300 mg every 2 weeks is best for long-term treatment, “therapeutic decisions are often influenced by cost-benefit considerations, in which case practitioners and other stakeholders involved in these decisions should carefully balance potential savings against suboptimal efficacy and long-term risks associated with discontinuous treatment regimens,” the investigators wrote.
The SOLO-CONTINUE trial was funded by Sanofi and Regeneron, the companies that market dupilumab. Dr. Worm reported receiving honoraria for consulting and lecture activity from Regeneron and Sanofi during and outside the conduct of the study, among other disclosures. The other authors had multiple disclosures related to these and multiple other pharmaceutical companies, or were employees of Sanofi or Regeneron.
SOURCE: Worm M et al. JAMA Dermatol. 2019 Dec 26. doi: 10.1001/jamadermatol.2019.3617.
Patients with moderate to severe atopic dermatitis who responded well to the approved dupilumab regimen of 300 mg every 2 weeks in pivotal phase 3 monotherapy trials were more likely to have a continued response over the longer term if they maintained this regimen rather than switching to longer dosing intervals or discontinuing the medication.
This finding comes from a 36-week, randomized, double-blind, placebo-controlled trial that enrolled 422 – SOLO 1 and SOLO 2.
The new international study – SOLO-CONTINUE – randomized these patients to continue the original regimen (weekly or every 2 weeks), to receive 300 mg of the biologic medication every 4 or 8 weeks, or to receive placebo.
Patients who continued the original regimen had the most consistent maintenance of treatment effect, while patients on longer dosage intervals or placebo had a dose-dependent reduction in response and no safety advantage. The incidence of treatment-emergent antidrug antibody was lowest with dupilumab weekly or every 2 weeks, and slightly higher with less-frequent dosing intervals, reported Margitta Worm, MD, of the Charité Universitätsmedizin Berlin, and coinvestigators.
“Because administration every 4 weeks or every 8 weeks did not provide an additional safety advantage and was numerically outperformed by administration weekly or every 2 weeks, we believe that it is prudent to adhere to the approved every 2 weeks regimen for adults and avoid less frequent treatment regimens (every 4 weeks or every 8 weeks) for long-term maintenance of efficacy,” they wrote in JAMA Dermatology.
Treatment success in the original SOLO trials was defined as having achieved an Investigator’s Global Assessment score of 0-1, or 75% improvement in Eczema Area and Severity Index Scores (EASI-75). As primary endpoints, SOLO-CONTINUE looked at the mean percentage change in EASI score over the course of the trial, and the percentage of patients who maintained EASI-75 at week 36.
Patients in the SOLO-CONTINUE trial who were randomized to receive dupilumab weekly or every 2 weeks had a mean percent change in EASI score of –0.06%. In contrast, patients assigned to the placebo group had a 21.7% decrease, and those taking the medication at 4- and 8-week intervals had mean changes of –3.84% and –6.84%, respectively. Post hoc analyses showed no apparent difference between dupilumab weekly and every 2 weeks in the maintenance of clinical response, the investigators reported.
Among patients with EASI-75 response at baseline, significantly more patients maintained this response at week 36 than patients receiving placebo, and there was again an apparent dose-dependent response. The percentage with EASI-75 at week 36 was 71.6% with the weekly or every-2-weeks regimen, 58.3% with the 4-week regimen, 54.9% with the 8-week regimen, and 30.4% in the placebo group.
Continuing treatment with 300 mg weekly or every 2 weeks resulted in greater maintenance of response across multiple other clinical endpoints and patient-reported outcomes as well (such as pruritus, atopic dermatitis symptoms, sleep, pain or discomfort, quality of life, and symptoms of anxiety and depression).
The more-frequent regimens also conferred no greater risk than less-frequent administration, and there were no new safety signals over the 36-week trial. Treatment-emergent adverse events (the most common were headache, nasopharyngitis, injection-site reactions, and herpes simplex virus infection) occurred in 70.7% of patients in the weekly or every-2-weeks group, 73.6% in the 4-week group, 75% in the 8-week group, and 81.7% in the placebo group.
Unlike earlier studies, the incidence of conjunctivitis was low (less than 6%) across all treatment groups, possibly because patients in the SOLO-CONTINUE trial were all high-level responders who tend to have conjunctivitis less frequently, the authors wrote.
Patients receiving less-frequent doses of dupilumab, particularly every 8 weeks, had greater rates of skin infections, flares, and rescue medication use than patients receiving doses weekly or every 2 weeks, the investigators reported. Treatment-emergent antidrug antibody incidence was slightly higher with less-frequent doses (11.7% and 6% in the 8-week and 4-week groups, respectively, compared with 4.3% and 1.2% in the every-2-weeks and weekly groups), which indicates a “higher incidence of immunogenicity with less-frequent dosage intervals” and is “consistent with other biologics,” they wrote.
Dupilumab is a human monoclonal antibody against the interleukin-4 receptor alpha that inhibits signaling of IL-4 and IL-13. The study was conducted at 185 sites in North America, Europe, Asia, and Japan. Patients had a mean age of 38.2 years; 53.8% were male.
While the trial suggests that the approved regimen of 300 mg every 2 weeks is best for long-term treatment, “therapeutic decisions are often influenced by cost-benefit considerations, in which case practitioners and other stakeholders involved in these decisions should carefully balance potential savings against suboptimal efficacy and long-term risks associated with discontinuous treatment regimens,” the investigators wrote.
The SOLO-CONTINUE trial was funded by Sanofi and Regeneron, the companies that market dupilumab. Dr. Worm reported receiving honoraria for consulting and lecture activity from Regeneron and Sanofi during and outside the conduct of the study, among other disclosures. The other authors had multiple disclosures related to these and multiple other pharmaceutical companies, or were employees of Sanofi or Regeneron.
SOURCE: Worm M et al. JAMA Dermatol. 2019 Dec 26. doi: 10.1001/jamadermatol.2019.3617.
FROM JAMA DERMATOLOGY