User login
Documentation for Mohs Surgery
In 2013, the Centers for Medicare and Medicaid Services (CMS) issued a guidance to reduce reimbursement issues for Mohs micrographic surgery (MMS).1 One crucial question that remains is when and if these documentation guidelines will be formally implemented. The guidelines outlined by the CMS currently are regarded as suggestions until Medicare contractors adopt them into the local coverage determinations (LCDs).
Key Documentation Guidelines
To reduce MMS reimbursement issues, documentation in the patient’s medical record should support the medical necessity of the procedure and reflect the number and anatomic locations of specimens taken and the reason for the procedure should be clearly communicated. The specific tumor type also should be approved for treatment with MMS in the respective LCD.
Nonphysician providers are not authorized by Medicare to perform MMS. To ensure proper coding, both surgery and pathology must be performed by a single physician and should be supported by documentation in the patient’s medical record (eg, relevant chart notes should be made under the provider’s signature). These documentation guidelines are not new but are included in the CMS guidance to reiterate their importance in reducing MMS reimbursement issues.
Per customary clinical practice, the CMS guidance specifies that MMS documentation should include gross description of the tissue removed, including the location, number, and size of the lesions, as well as how many specimens were removed for each stage. However, the guidance diverges from routine MMS documentation requirements in its emphasis on providing a histologic description of the tissue removed. The guidance suggests that the depth of tumor invasion, pathologic pattern, cell morphology (which is not typically specified for skin cancers), and, if present, the existence of perineural invasion or scar tissue should be documented. If these features are constant across stages, they only need to be noted for the first stage.
Adapting Guidelines for Clinical Practice
The CMS guidance may create some conundrums for physicians regarding MMS documentation; for instance, if a tumor is cleared in one stage, as is often the case, no tumor will be seen on glass slides prepared to assess tissue margins during the procedure and therefore documentation of characteristics like depth and pattern will be impossible. Similarly, cell morphology is not a feature that usually is relevant for most squamous and basal cell carcinomas, although it may be useful in certain unusual instances, such as in cases of rare tumors with particular histologic features that may influence management and/or prognosis. When in doubt regarding the appropriate documentation method for MMS, the surgeon should use his or her best judgment based on clinical experience rather than simply following guidelines that may not be applicable.
Final Thoughts
The CMS guidance serves as a reminder of the documentation requirements for MMS and extends current practice by suggesting a detailed microscopic description of the removed tissue. The American Academy of Dermatology has developed a guide to help Mohs surgeons provide the necessary documentation without creating cumbersome chart notes.2 Mohs surgeons should consult the most recent version of the LCD that applies to their geographic area to determine if the new documentation guidelines have been adopted.
- Centers for Medicare and Medicaid Services. Guidance to Reduce Mohs Surgery Reimbursement Issues. Bethesda, MD: Centers for Medicare and Medicaid Services, US Department of Health and Human Services; 2013. MLN Matters SE1318.
- Position statement on documentation of frozen section specimens during Mohs micrographic surgery. American Academy of Dermatology Web site. https://www.aad.org/forms/policies/Uploads/PS/PS%20-%20Documentation%20of%20Frozen%20Section%20Specimens%20during%20Mohs%20Micrographic%20Surgery.pdf. Accessed November 30, 2015.
In 2013, the Centers for Medicare and Medicaid Services (CMS) issued a guidance to reduce reimbursement issues for Mohs micrographic surgery (MMS).1 One crucial question that remains is when and if these documentation guidelines will be formally implemented. The guidelines outlined by the CMS currently are regarded as suggestions until Medicare contractors adopt them into the local coverage determinations (LCDs).
Key Documentation Guidelines
To reduce MMS reimbursement issues, documentation in the patient’s medical record should support the medical necessity of the procedure and reflect the number and anatomic locations of specimens taken and the reason for the procedure should be clearly communicated. The specific tumor type also should be approved for treatment with MMS in the respective LCD.
Nonphysician providers are not authorized by Medicare to perform MMS. To ensure proper coding, both surgery and pathology must be performed by a single physician and should be supported by documentation in the patient’s medical record (eg, relevant chart notes should be made under the provider’s signature). These documentation guidelines are not new but are included in the CMS guidance to reiterate their importance in reducing MMS reimbursement issues.
Per customary clinical practice, the CMS guidance specifies that MMS documentation should include gross description of the tissue removed, including the location, number, and size of the lesions, as well as how many specimens were removed for each stage. However, the guidance diverges from routine MMS documentation requirements in its emphasis on providing a histologic description of the tissue removed. The guidance suggests that the depth of tumor invasion, pathologic pattern, cell morphology (which is not typically specified for skin cancers), and, if present, the existence of perineural invasion or scar tissue should be documented. If these features are constant across stages, they only need to be noted for the first stage.
Adapting Guidelines for Clinical Practice
The CMS guidance may create some conundrums for physicians regarding MMS documentation; for instance, if a tumor is cleared in one stage, as is often the case, no tumor will be seen on glass slides prepared to assess tissue margins during the procedure and therefore documentation of characteristics like depth and pattern will be impossible. Similarly, cell morphology is not a feature that usually is relevant for most squamous and basal cell carcinomas, although it may be useful in certain unusual instances, such as in cases of rare tumors with particular histologic features that may influence management and/or prognosis. When in doubt regarding the appropriate documentation method for MMS, the surgeon should use his or her best judgment based on clinical experience rather than simply following guidelines that may not be applicable.
Final Thoughts
The CMS guidance serves as a reminder of the documentation requirements for MMS and extends current practice by suggesting a detailed microscopic description of the removed tissue. The American Academy of Dermatology has developed a guide to help Mohs surgeons provide the necessary documentation without creating cumbersome chart notes.2 Mohs surgeons should consult the most recent version of the LCD that applies to their geographic area to determine if the new documentation guidelines have been adopted.
In 2013, the Centers for Medicare and Medicaid Services (CMS) issued a guidance to reduce reimbursement issues for Mohs micrographic surgery (MMS).1 One crucial question that remains is when and if these documentation guidelines will be formally implemented. The guidelines outlined by the CMS currently are regarded as suggestions until Medicare contractors adopt them into the local coverage determinations (LCDs).
Key Documentation Guidelines
To reduce MMS reimbursement issues, documentation in the patient’s medical record should support the medical necessity of the procedure and reflect the number and anatomic locations of specimens taken and the reason for the procedure should be clearly communicated. The specific tumor type also should be approved for treatment with MMS in the respective LCD.
Nonphysician providers are not authorized by Medicare to perform MMS. To ensure proper coding, both surgery and pathology must be performed by a single physician and should be supported by documentation in the patient’s medical record (eg, relevant chart notes should be made under the provider’s signature). These documentation guidelines are not new but are included in the CMS guidance to reiterate their importance in reducing MMS reimbursement issues.
Per customary clinical practice, the CMS guidance specifies that MMS documentation should include gross description of the tissue removed, including the location, number, and size of the lesions, as well as how many specimens were removed for each stage. However, the guidance diverges from routine MMS documentation requirements in its emphasis on providing a histologic description of the tissue removed. The guidance suggests that the depth of tumor invasion, pathologic pattern, cell morphology (which is not typically specified for skin cancers), and, if present, the existence of perineural invasion or scar tissue should be documented. If these features are constant across stages, they only need to be noted for the first stage.
Adapting Guidelines for Clinical Practice
The CMS guidance may create some conundrums for physicians regarding MMS documentation; for instance, if a tumor is cleared in one stage, as is often the case, no tumor will be seen on glass slides prepared to assess tissue margins during the procedure and therefore documentation of characteristics like depth and pattern will be impossible. Similarly, cell morphology is not a feature that usually is relevant for most squamous and basal cell carcinomas, although it may be useful in certain unusual instances, such as in cases of rare tumors with particular histologic features that may influence management and/or prognosis. When in doubt regarding the appropriate documentation method for MMS, the surgeon should use his or her best judgment based on clinical experience rather than simply following guidelines that may not be applicable.
Final Thoughts
The CMS guidance serves as a reminder of the documentation requirements for MMS and extends current practice by suggesting a detailed microscopic description of the removed tissue. The American Academy of Dermatology has developed a guide to help Mohs surgeons provide the necessary documentation without creating cumbersome chart notes.2 Mohs surgeons should consult the most recent version of the LCD that applies to their geographic area to determine if the new documentation guidelines have been adopted.
- Centers for Medicare and Medicaid Services. Guidance to Reduce Mohs Surgery Reimbursement Issues. Bethesda, MD: Centers for Medicare and Medicaid Services, US Department of Health and Human Services; 2013. MLN Matters SE1318.
- Position statement on documentation of frozen section specimens during Mohs micrographic surgery. American Academy of Dermatology Web site. https://www.aad.org/forms/policies/Uploads/PS/PS%20-%20Documentation%20of%20Frozen%20Section%20Specimens%20during%20Mohs%20Micrographic%20Surgery.pdf. Accessed November 30, 2015.
- Centers for Medicare and Medicaid Services. Guidance to Reduce Mohs Surgery Reimbursement Issues. Bethesda, MD: Centers for Medicare and Medicaid Services, US Department of Health and Human Services; 2013. MLN Matters SE1318.
- Position statement on documentation of frozen section specimens during Mohs micrographic surgery. American Academy of Dermatology Web site. https://www.aad.org/forms/policies/Uploads/PS/PS%20-%20Documentation%20of%20Frozen%20Section%20Specimens%20during%20Mohs%20Micrographic%20Surgery.pdf. Accessed November 30, 2015.
Melia azedarach
Native to China, India, and Iran, Melia azedarach – also known as Chinaberry (Ku-lian), Pride of China, Indian lilac, Persian lilac, and white cedar – is a large nondeciduous tree now cultivated throughout the world.1-3 The leaves and fruits of this member of the Meliaceae family have been found to exhibit anthelmintic and antifeedant activity toward insects.4 The root bark or dried stem bark of the plant is used in traditional Chinese medicine to treat dermatoses and to expel and repel worms.5,6M. azedarach has also been used in Ayurveda3; it is known as Bakayn in India and Pakistan, where it has been used in traditional medicine to treat leprosy, scrofula, nausea, vomiting, thirst, and eruptive skin conditions.3,7
Traditional indications
Cropley and Hasegawa noted in 2007 that M. azedarach was used in the 19th century in the United States primarily as an anthelmintic, but also to treat tinea capitis and to eliminate lice.Further, they noted that the plant, which was widely cultivated in the South, was used, according to the literature of the 1800s, as a diuretic, as well as to promote hair growth, and treat scalp eruptions, fever, and malaria.8
A 2006 ethnopharmacologic study by Saikia et al. of medicinal plants used in Assam (Northeast India) to treat skin disorders and for cosmetic purposes revealed that M. azedarach was one of the primary plants used for medicinal purposes, including 18 skin conditions such as acne, burns, carbuncles, abscesses, measles, pediculosis, cellulitis, and prickly heat.9
Four years later, in an ethnopharmacologic study of medicinal plants used to treat cutaneous disorders and in folk cosmetics, Abbasi et al. interviewed residents in 30 remote tribal communities in the Northwest Frontier Province of Pakistan, finding that M. azedarach was one of 15 plant species identified as curative for numerous skin diseases, including boils, acne, and scabies.10
Biologic activity
Various constituents of M. azedarach have been associated with a wide range of biologic activity. The antifungal activity of the plant has been attributed to its hydroxycoumarin content; anti-inflammatory activity to its monoterpenes; bacteriostatic properties to its beta-carboline alkaloids; antiviral effects to its meliacin and meliacarpin; and insecticidal and antifeedant activity to its limonoids, which belong to the tetranortriterpenoid class of compounds.7,11 Limonoids also are reputed to confer antibacterial, antiviral, and antifungal activities.12 Recent research suggests the potential of M. azedarach to offer a correspondingly broad impact on various conditions.
Anticancer potential
Zhou et al. isolated new ring C-seco limonoids from Brazilian M. azedarach in 2004 and evaluated their cytotoxic activity, finding that two limonoids significantly inhibited HeLa S3 cancer cells.4,11
In 2009, He et al. extracted water-soluble polysaccharides from the fruit of M. azedarach and assessed their cytotoxic activity against four human cell lines in vitro. They found that the major polysaccharide component exhibited potent cytotoxic activity in the BGC-823 cell line.3 Also that year, Wu et al. isolated three new and multiple known steroids from the leaves of Chinese M. azedarach, finding that two of the newly identified steroids – (20S)-5,24(28)-ergostadiene-3-beta,7-alpha,16-beta,20-tetrol and (20S)-5-ergostene-3-beta,7-alpha,16-beta,20-tetrol – and one of the known ones exerted significant cytotoxic impact in three human cancer cell lines (A-549, H460, U251).1
Two years later, Wu et al. isolated two new triterpenoids (21,24-cycloeupha-7-ene-3-beta,16beta,21-alpha,25-tetrol and 3-beta-acetoxy-12-beta-hydroxy-eupha-7,24-dien-21,16-beta-olide) and two new steroids (29-hydroperoxy-stigmasta-7,24(28) xi-dien-3-beta-ol and 24-xi-hydroperoxy-24-vinyl-lathosterol) along with several known related substances from M. azedarach bark. Five of the known compounds demonstrated cytotoxic activity against three human cancer cell lines (A-549, H460, HGC27).6
More recently, Yuan et al. isolated two limonoids, one triterpenoid, one steroid, and one sesquiterpenoid, as well as nine known limonoids from the bark of M. azedarach. In addition, the investigators observed that seven of the limonoids exhibited significant inhibitory properties against five human tumor cell lines (HL-60, SMM-C7721, A-549, MCF-7, and SW480).5
Efficacy against head lice
In 2007, Carpinella et al. investigated in vitro the pediculicidal and ovicidal activity of an M. azedarach extract against Pediculus humanus capitis, noting the increasing resistance of head lice to synthetic drugs. A combination of 20% ripe fruit extract with 10% ripe fruit oil was found to be the most effective in eradicating adult lice (96.5%). The investigators also prepared a formulation of extract and oil at 10% combined with an emulsifier and preservatives that yielded 92.3% mortality of adult lice and complete inhibition of nymph emergence. They concluded that the use of M. azedarach formulations may be a viable and natural way to control head lice.2M. azedarach also has been the key active ingredient in an antimicrobial cream and a preparation to treat tinea pedis.7
Topical cream
Saleem et al. reported in 2008 on an herbal cream preparation using a standardized extract of M. azedarach flowers that displayed strong antibacterial activity against cellulitis, pustules, and pyogenic infections, and other conditions in clinical trials in children under the age of 10 years in four different villages in Pakistan. Subjects with bacterial infections anywhere on the body were divided into three groups, one treated with M. azedarach cream, one with placebo (negative control), and one with neomycin (positive control). Each treatment was applied twice daily for 15 days. The investigators observed that the healing effects of M. azedarach were similar to those rendered by neomycin.7
Conclusion
Not only has M. azedarach been used in several traditional medical systems, but its uses have consistently included cutaneous indications. Its most convincing modern application may be for head lice, but recent evidence and traditional uses suggest that this versatile plant may indeed warrant a more prominent role in the current dermatologic armamentarium. Future research is needed, and should provide some interesting answers.
References
1. Steroids. 2009 Sep;74(9):761-5.
2. J Am Acad Dermatol. 2007 Feb;56(2):250-6.
3. Fitoterapia. 2009 Oct;80(7):399-403.
4. Chem Pharm Bull (Tokyo). 2005 Oct;53(10):1362-5.
5. Planta Med. 2013 Jan;79(2):163-8.
6. Planta Med. 2011 Jun;77(9):922-8.
7. Phytomedicine. 2008 Apr;15(4):231-6.
8. J Am Acad Dermatol. 2007 Aug;57(2):366-7.
9. J Ethnopharmacol. 2006 Jun 30;106(2):149-57.
10. J Ethnopharmacol. 2010 Mar 24;128(2):322-35.
11. J Nat Prod. 2004 Sep;67(9):1544-7.
12. Chem Biodivers. 2010 Apr;7(4):839-59.
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in the Design District in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote the textbook, “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002), and a book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). She has contributed to the Cosmeceutical Critique column in Dermatology News since January 2001. Her latest book, “Cosmeceuticals and Cosmetic Ingredients,” was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Evolus, Galderma, GlaxoSmithKline, Kythera Biopharmaceuticals, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Topix Pharmaceuticals, and Unilever.
Native to China, India, and Iran, Melia azedarach – also known as Chinaberry (Ku-lian), Pride of China, Indian lilac, Persian lilac, and white cedar – is a large nondeciduous tree now cultivated throughout the world.1-3 The leaves and fruits of this member of the Meliaceae family have been found to exhibit anthelmintic and antifeedant activity toward insects.4 The root bark or dried stem bark of the plant is used in traditional Chinese medicine to treat dermatoses and to expel and repel worms.5,6M. azedarach has also been used in Ayurveda3; it is known as Bakayn in India and Pakistan, where it has been used in traditional medicine to treat leprosy, scrofula, nausea, vomiting, thirst, and eruptive skin conditions.3,7
Traditional indications
Cropley and Hasegawa noted in 2007 that M. azedarach was used in the 19th century in the United States primarily as an anthelmintic, but also to treat tinea capitis and to eliminate lice.Further, they noted that the plant, which was widely cultivated in the South, was used, according to the literature of the 1800s, as a diuretic, as well as to promote hair growth, and treat scalp eruptions, fever, and malaria.8
A 2006 ethnopharmacologic study by Saikia et al. of medicinal plants used in Assam (Northeast India) to treat skin disorders and for cosmetic purposes revealed that M. azedarach was one of the primary plants used for medicinal purposes, including 18 skin conditions such as acne, burns, carbuncles, abscesses, measles, pediculosis, cellulitis, and prickly heat.9
Four years later, in an ethnopharmacologic study of medicinal plants used to treat cutaneous disorders and in folk cosmetics, Abbasi et al. interviewed residents in 30 remote tribal communities in the Northwest Frontier Province of Pakistan, finding that M. azedarach was one of 15 plant species identified as curative for numerous skin diseases, including boils, acne, and scabies.10
Biologic activity
Various constituents of M. azedarach have been associated with a wide range of biologic activity. The antifungal activity of the plant has been attributed to its hydroxycoumarin content; anti-inflammatory activity to its monoterpenes; bacteriostatic properties to its beta-carboline alkaloids; antiviral effects to its meliacin and meliacarpin; and insecticidal and antifeedant activity to its limonoids, which belong to the tetranortriterpenoid class of compounds.7,11 Limonoids also are reputed to confer antibacterial, antiviral, and antifungal activities.12 Recent research suggests the potential of M. azedarach to offer a correspondingly broad impact on various conditions.
Anticancer potential
Zhou et al. isolated new ring C-seco limonoids from Brazilian M. azedarach in 2004 and evaluated their cytotoxic activity, finding that two limonoids significantly inhibited HeLa S3 cancer cells.4,11
In 2009, He et al. extracted water-soluble polysaccharides from the fruit of M. azedarach and assessed their cytotoxic activity against four human cell lines in vitro. They found that the major polysaccharide component exhibited potent cytotoxic activity in the BGC-823 cell line.3 Also that year, Wu et al. isolated three new and multiple known steroids from the leaves of Chinese M. azedarach, finding that two of the newly identified steroids – (20S)-5,24(28)-ergostadiene-3-beta,7-alpha,16-beta,20-tetrol and (20S)-5-ergostene-3-beta,7-alpha,16-beta,20-tetrol – and one of the known ones exerted significant cytotoxic impact in three human cancer cell lines (A-549, H460, U251).1
Two years later, Wu et al. isolated two new triterpenoids (21,24-cycloeupha-7-ene-3-beta,16beta,21-alpha,25-tetrol and 3-beta-acetoxy-12-beta-hydroxy-eupha-7,24-dien-21,16-beta-olide) and two new steroids (29-hydroperoxy-stigmasta-7,24(28) xi-dien-3-beta-ol and 24-xi-hydroperoxy-24-vinyl-lathosterol) along with several known related substances from M. azedarach bark. Five of the known compounds demonstrated cytotoxic activity against three human cancer cell lines (A-549, H460, HGC27).6
More recently, Yuan et al. isolated two limonoids, one triterpenoid, one steroid, and one sesquiterpenoid, as well as nine known limonoids from the bark of M. azedarach. In addition, the investigators observed that seven of the limonoids exhibited significant inhibitory properties against five human tumor cell lines (HL-60, SMM-C7721, A-549, MCF-7, and SW480).5
Efficacy against head lice
In 2007, Carpinella et al. investigated in vitro the pediculicidal and ovicidal activity of an M. azedarach extract against Pediculus humanus capitis, noting the increasing resistance of head lice to synthetic drugs. A combination of 20% ripe fruit extract with 10% ripe fruit oil was found to be the most effective in eradicating adult lice (96.5%). The investigators also prepared a formulation of extract and oil at 10% combined with an emulsifier and preservatives that yielded 92.3% mortality of adult lice and complete inhibition of nymph emergence. They concluded that the use of M. azedarach formulations may be a viable and natural way to control head lice.2M. azedarach also has been the key active ingredient in an antimicrobial cream and a preparation to treat tinea pedis.7
Topical cream
Saleem et al. reported in 2008 on an herbal cream preparation using a standardized extract of M. azedarach flowers that displayed strong antibacterial activity against cellulitis, pustules, and pyogenic infections, and other conditions in clinical trials in children under the age of 10 years in four different villages in Pakistan. Subjects with bacterial infections anywhere on the body were divided into three groups, one treated with M. azedarach cream, one with placebo (negative control), and one with neomycin (positive control). Each treatment was applied twice daily for 15 days. The investigators observed that the healing effects of M. azedarach were similar to those rendered by neomycin.7
Conclusion
Not only has M. azedarach been used in several traditional medical systems, but its uses have consistently included cutaneous indications. Its most convincing modern application may be for head lice, but recent evidence and traditional uses suggest that this versatile plant may indeed warrant a more prominent role in the current dermatologic armamentarium. Future research is needed, and should provide some interesting answers.
References
1. Steroids. 2009 Sep;74(9):761-5.
2. J Am Acad Dermatol. 2007 Feb;56(2):250-6.
3. Fitoterapia. 2009 Oct;80(7):399-403.
4. Chem Pharm Bull (Tokyo). 2005 Oct;53(10):1362-5.
5. Planta Med. 2013 Jan;79(2):163-8.
6. Planta Med. 2011 Jun;77(9):922-8.
7. Phytomedicine. 2008 Apr;15(4):231-6.
8. J Am Acad Dermatol. 2007 Aug;57(2):366-7.
9. J Ethnopharmacol. 2006 Jun 30;106(2):149-57.
10. J Ethnopharmacol. 2010 Mar 24;128(2):322-35.
11. J Nat Prod. 2004 Sep;67(9):1544-7.
12. Chem Biodivers. 2010 Apr;7(4):839-59.
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in the Design District in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote the textbook, “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002), and a book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). She has contributed to the Cosmeceutical Critique column in Dermatology News since January 2001. Her latest book, “Cosmeceuticals and Cosmetic Ingredients,” was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Evolus, Galderma, GlaxoSmithKline, Kythera Biopharmaceuticals, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Topix Pharmaceuticals, and Unilever.
Native to China, India, and Iran, Melia azedarach – also known as Chinaberry (Ku-lian), Pride of China, Indian lilac, Persian lilac, and white cedar – is a large nondeciduous tree now cultivated throughout the world.1-3 The leaves and fruits of this member of the Meliaceae family have been found to exhibit anthelmintic and antifeedant activity toward insects.4 The root bark or dried stem bark of the plant is used in traditional Chinese medicine to treat dermatoses and to expel and repel worms.5,6M. azedarach has also been used in Ayurveda3; it is known as Bakayn in India and Pakistan, where it has been used in traditional medicine to treat leprosy, scrofula, nausea, vomiting, thirst, and eruptive skin conditions.3,7
Traditional indications
Cropley and Hasegawa noted in 2007 that M. azedarach was used in the 19th century in the United States primarily as an anthelmintic, but also to treat tinea capitis and to eliminate lice.Further, they noted that the plant, which was widely cultivated in the South, was used, according to the literature of the 1800s, as a diuretic, as well as to promote hair growth, and treat scalp eruptions, fever, and malaria.8
A 2006 ethnopharmacologic study by Saikia et al. of medicinal plants used in Assam (Northeast India) to treat skin disorders and for cosmetic purposes revealed that M. azedarach was one of the primary plants used for medicinal purposes, including 18 skin conditions such as acne, burns, carbuncles, abscesses, measles, pediculosis, cellulitis, and prickly heat.9
Four years later, in an ethnopharmacologic study of medicinal plants used to treat cutaneous disorders and in folk cosmetics, Abbasi et al. interviewed residents in 30 remote tribal communities in the Northwest Frontier Province of Pakistan, finding that M. azedarach was one of 15 plant species identified as curative for numerous skin diseases, including boils, acne, and scabies.10
Biologic activity
Various constituents of M. azedarach have been associated with a wide range of biologic activity. The antifungal activity of the plant has been attributed to its hydroxycoumarin content; anti-inflammatory activity to its monoterpenes; bacteriostatic properties to its beta-carboline alkaloids; antiviral effects to its meliacin and meliacarpin; and insecticidal and antifeedant activity to its limonoids, which belong to the tetranortriterpenoid class of compounds.7,11 Limonoids also are reputed to confer antibacterial, antiviral, and antifungal activities.12 Recent research suggests the potential of M. azedarach to offer a correspondingly broad impact on various conditions.
Anticancer potential
Zhou et al. isolated new ring C-seco limonoids from Brazilian M. azedarach in 2004 and evaluated their cytotoxic activity, finding that two limonoids significantly inhibited HeLa S3 cancer cells.4,11
In 2009, He et al. extracted water-soluble polysaccharides from the fruit of M. azedarach and assessed their cytotoxic activity against four human cell lines in vitro. They found that the major polysaccharide component exhibited potent cytotoxic activity in the BGC-823 cell line.3 Also that year, Wu et al. isolated three new and multiple known steroids from the leaves of Chinese M. azedarach, finding that two of the newly identified steroids – (20S)-5,24(28)-ergostadiene-3-beta,7-alpha,16-beta,20-tetrol and (20S)-5-ergostene-3-beta,7-alpha,16-beta,20-tetrol – and one of the known ones exerted significant cytotoxic impact in three human cancer cell lines (A-549, H460, U251).1
Two years later, Wu et al. isolated two new triterpenoids (21,24-cycloeupha-7-ene-3-beta,16beta,21-alpha,25-tetrol and 3-beta-acetoxy-12-beta-hydroxy-eupha-7,24-dien-21,16-beta-olide) and two new steroids (29-hydroperoxy-stigmasta-7,24(28) xi-dien-3-beta-ol and 24-xi-hydroperoxy-24-vinyl-lathosterol) along with several known related substances from M. azedarach bark. Five of the known compounds demonstrated cytotoxic activity against three human cancer cell lines (A-549, H460, HGC27).6
More recently, Yuan et al. isolated two limonoids, one triterpenoid, one steroid, and one sesquiterpenoid, as well as nine known limonoids from the bark of M. azedarach. In addition, the investigators observed that seven of the limonoids exhibited significant inhibitory properties against five human tumor cell lines (HL-60, SMM-C7721, A-549, MCF-7, and SW480).5
Efficacy against head lice
In 2007, Carpinella et al. investigated in vitro the pediculicidal and ovicidal activity of an M. azedarach extract against Pediculus humanus capitis, noting the increasing resistance of head lice to synthetic drugs. A combination of 20% ripe fruit extract with 10% ripe fruit oil was found to be the most effective in eradicating adult lice (96.5%). The investigators also prepared a formulation of extract and oil at 10% combined with an emulsifier and preservatives that yielded 92.3% mortality of adult lice and complete inhibition of nymph emergence. They concluded that the use of M. azedarach formulations may be a viable and natural way to control head lice.2M. azedarach also has been the key active ingredient in an antimicrobial cream and a preparation to treat tinea pedis.7
Topical cream
Saleem et al. reported in 2008 on an herbal cream preparation using a standardized extract of M. azedarach flowers that displayed strong antibacterial activity against cellulitis, pustules, and pyogenic infections, and other conditions in clinical trials in children under the age of 10 years in four different villages in Pakistan. Subjects with bacterial infections anywhere on the body were divided into three groups, one treated with M. azedarach cream, one with placebo (negative control), and one with neomycin (positive control). Each treatment was applied twice daily for 15 days. The investigators observed that the healing effects of M. azedarach were similar to those rendered by neomycin.7
Conclusion
Not only has M. azedarach been used in several traditional medical systems, but its uses have consistently included cutaneous indications. Its most convincing modern application may be for head lice, but recent evidence and traditional uses suggest that this versatile plant may indeed warrant a more prominent role in the current dermatologic armamentarium. Future research is needed, and should provide some interesting answers.
References
1. Steroids. 2009 Sep;74(9):761-5.
2. J Am Acad Dermatol. 2007 Feb;56(2):250-6.
3. Fitoterapia. 2009 Oct;80(7):399-403.
4. Chem Pharm Bull (Tokyo). 2005 Oct;53(10):1362-5.
5. Planta Med. 2013 Jan;79(2):163-8.
6. Planta Med. 2011 Jun;77(9):922-8.
7. Phytomedicine. 2008 Apr;15(4):231-6.
8. J Am Acad Dermatol. 2007 Aug;57(2):366-7.
9. J Ethnopharmacol. 2006 Jun 30;106(2):149-57.
10. J Ethnopharmacol. 2010 Mar 24;128(2):322-35.
11. J Nat Prod. 2004 Sep;67(9):1544-7.
12. Chem Biodivers. 2010 Apr;7(4):839-59.
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in the Design District in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote the textbook, “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002), and a book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). She has contributed to the Cosmeceutical Critique column in Dermatology News since January 2001. Her latest book, “Cosmeceuticals and Cosmetic Ingredients,” was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Evolus, Galderma, GlaxoSmithKline, Kythera Biopharmaceuticals, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Topix Pharmaceuticals, and Unilever.
Cosmetic Corner: Dermatologists Weigh in on Products for Babies
To improve patient care and outcomes, leading dermatologists offered their recommendations on top products for babies. Consideration must be given to:
- CeraVe Hydrating Cleanser
Valeant Pharmaceuticals North America LLC
Recommended by Gary Goldenberg, MD, New York, New York
- Cetaphil Baby products
Galderma Laboratories, LP
Recommended by Gary Goldenberg, MD, New York, New York
- Cetaphil Gentle Skin Cleanser and Moisturizing Lotion
Galderma Laboratories, LP
“This is my favorite combination for babies. In my opinion, it is the best choice and what I used for my own children.”—Wm. Phillip Werschler, MD, Seattle, Washington
- Everyday/Year-Round Broad Spectrum SPF 30+ Sunscreen
California Baby
“It contains only titanium dioxide as the UV blocker and has a mild ingredient profile.”—Anthony M. Rossi, MD, New York, New York
- Johnson’s Baby products
Johnson & Johnson Consumer Companies, Inc
“Johnson & Johnson’s baby care products (No More Tears formula) are an excellent choice.”—Wm. Phillip Werschler, Seattle, Washington
- Vanicream products
Pharmaceutical Specialties, Inc
Recommended by Gary Goldenberg, MD, New York, New York
Cutis invites readers to send us their recommendations. Cosmetic fillers, antiperspirants, shampoos, and conditioners will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
To improve patient care and outcomes, leading dermatologists offered their recommendations on top products for babies. Consideration must be given to:
- CeraVe Hydrating Cleanser
Valeant Pharmaceuticals North America LLC
Recommended by Gary Goldenberg, MD, New York, New York
- Cetaphil Baby products
Galderma Laboratories, LP
Recommended by Gary Goldenberg, MD, New York, New York
- Cetaphil Gentle Skin Cleanser and Moisturizing Lotion
Galderma Laboratories, LP
“This is my favorite combination for babies. In my opinion, it is the best choice and what I used for my own children.”—Wm. Phillip Werschler, MD, Seattle, Washington
- Everyday/Year-Round Broad Spectrum SPF 30+ Sunscreen
California Baby
“It contains only titanium dioxide as the UV blocker and has a mild ingredient profile.”—Anthony M. Rossi, MD, New York, New York
- Johnson’s Baby products
Johnson & Johnson Consumer Companies, Inc
“Johnson & Johnson’s baby care products (No More Tears formula) are an excellent choice.”—Wm. Phillip Werschler, Seattle, Washington
- Vanicream products
Pharmaceutical Specialties, Inc
Recommended by Gary Goldenberg, MD, New York, New York
Cutis invites readers to send us their recommendations. Cosmetic fillers, antiperspirants, shampoos, and conditioners will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
To improve patient care and outcomes, leading dermatologists offered their recommendations on top products for babies. Consideration must be given to:
- CeraVe Hydrating Cleanser
Valeant Pharmaceuticals North America LLC
Recommended by Gary Goldenberg, MD, New York, New York
- Cetaphil Baby products
Galderma Laboratories, LP
Recommended by Gary Goldenberg, MD, New York, New York
- Cetaphil Gentle Skin Cleanser and Moisturizing Lotion
Galderma Laboratories, LP
“This is my favorite combination for babies. In my opinion, it is the best choice and what I used for my own children.”—Wm. Phillip Werschler, MD, Seattle, Washington
- Everyday/Year-Round Broad Spectrum SPF 30+ Sunscreen
California Baby
“It contains only titanium dioxide as the UV blocker and has a mild ingredient profile.”—Anthony M. Rossi, MD, New York, New York
- Johnson’s Baby products
Johnson & Johnson Consumer Companies, Inc
“Johnson & Johnson’s baby care products (No More Tears formula) are an excellent choice.”—Wm. Phillip Werschler, Seattle, Washington
- Vanicream products
Pharmaceutical Specialties, Inc
Recommended by Gary Goldenberg, MD, New York, New York
Cutis invites readers to send us their recommendations. Cosmetic fillers, antiperspirants, shampoos, and conditioners will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
Décolletage Rejuvenation With Cosmetic Injectables and Beyond

As more patients undergo facial rejuvenation procedures for a more youthful look, there is a growing demand for rejuvenation of the décolletage (neck and chest) to achieve a more natural and seamless transition from the skin of the face to the chest. The same modalities that are used on the face to treat skin rhytides, texture, and discoloration have been used successfully in the décolletage area.
Vanaman and Fabi (Plast Reconstr Surg. 2015;136[suppl 5]:276S-281S) recently reviewed the chest anatomy and discussed the safe and effective use of cosmetic injectables alone or in combination with other modalities to address rhytides of the décolletage. The relatively low density of skin pilosebaceous units on the chest allows for slower healing and thus makes the area more vulnerable to scarring with the use of more invasive resurfacing modalities (eg, deeper chemical peels, ablative lasers). The use of cosmetic injectables offers a safer treatment option of chest rhytides. Furthermore, proper candidate selection excludes patients with known sensitivity to cosmetic injectables or their components, history of keloid or hypertrophic scar formation, and active inflammation in the treatment area.
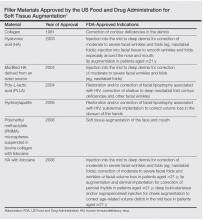
Poly-L-lactic acid (PLLA) is a biodegradable, biocompatible, semipermanent, synthetic soft tissue biostimulator that promotes neocollagenesis by fibroblasts over time (3–6 months). The manufacturer’s recommendation for PLLA reconstitution is at least 2 hours prior to injection with sterile water of no less than 5 mL dilution. Vanaman and Fabi reported usually diluting the day prior to injection with 16 mL total volume. This technique showed the greatest improvement in chest rhytides with no adverse events reported in a retrospective analysis. Poly-L-lactic acid should be injected in a retrograde linear fashion in the plane of the subcutaneous fat, with injection boundaries on the suprasternal notch superiorly, the midclavicular line laterally, and the fourth rib inferolaterally for rejuvenation of the décolletage.
Nodule formation is a well-known complication of PLLA injection, although pain, bruising, edema, pruritus, and hematomas are more commonly seen. The risk of nodule formation can be decreased using several techniques, including avoiding overcorrection and excessive use of product in each individual session, avoiding intradermal injection, diluting to more than 5 mL with reconstitution at least overnight, massaging the area posttreatment (in office by the clinician and at home by the patient), and scheduling treatment sessions at least 4 weeks apart. Usually, 3 to 4 treatments are required and the results can last 2 years or longer without touch-ups.
Nonanimal stabilized hyaluronic acid (NASHA) fillers also can be used to correct chest rhytides; however, using NASHA fillers requires more syringes and results typically last only 6 to 8 months, making it more cost effective to use 2 to 3 vials of PLLA. Moreover, in Vanaman and Fabi’s experience, PLLA is associated with fewer nodules, possibly due to the depth of injection of PLLA into the subcutaneous fat versus injection into the deep dermis with NASHA fillers. Vanaman and Fabi currently are investigating the use of calcium hydroxylapatite fillers alone or in combination with an energy-based modality (microfocused ultrasound) with visualization in the treatment of rhytides in the décolletage.
What’s the Issue?
The availability of many modalities to keep facial skin looking fresh and rejuvenated has led to an increased demand for products and procedures to rejuvenate the décolletage. It is important for dermatologists to acknowledge the more delicate nature of the décolletage versus the face. Less invasive modalities such as cosmetic injectables can be employed in a safe and effective manner to correct rhytides of the chest with proper techniques, products, and patient selection. For a more natural transition from the skin of the face to the décolletage, it also may be necessary to adopt a multimodal approach by using botulinum toxin and fillers, as well as going beyond correction of rhytides to address skin texture and discoloration with chemical peels and lasers. Have you seen an increased demand for procedures to rejuvenate the décolletage in your practice?

As more patients undergo facial rejuvenation procedures for a more youthful look, there is a growing demand for rejuvenation of the décolletage (neck and chest) to achieve a more natural and seamless transition from the skin of the face to the chest. The same modalities that are used on the face to treat skin rhytides, texture, and discoloration have been used successfully in the décolletage area.
Vanaman and Fabi (Plast Reconstr Surg. 2015;136[suppl 5]:276S-281S) recently reviewed the chest anatomy and discussed the safe and effective use of cosmetic injectables alone or in combination with other modalities to address rhytides of the décolletage. The relatively low density of skin pilosebaceous units on the chest allows for slower healing and thus makes the area more vulnerable to scarring with the use of more invasive resurfacing modalities (eg, deeper chemical peels, ablative lasers). The use of cosmetic injectables offers a safer treatment option of chest rhytides. Furthermore, proper candidate selection excludes patients with known sensitivity to cosmetic injectables or their components, history of keloid or hypertrophic scar formation, and active inflammation in the treatment area.
Poly-L-lactic acid (PLLA) is a biodegradable, biocompatible, semipermanent, synthetic soft tissue biostimulator that promotes neocollagenesis by fibroblasts over time (3–6 months). The manufacturer’s recommendation for PLLA reconstitution is at least 2 hours prior to injection with sterile water of no less than 5 mL dilution. Vanaman and Fabi reported usually diluting the day prior to injection with 16 mL total volume. This technique showed the greatest improvement in chest rhytides with no adverse events reported in a retrospective analysis. Poly-L-lactic acid should be injected in a retrograde linear fashion in the plane of the subcutaneous fat, with injection boundaries on the suprasternal notch superiorly, the midclavicular line laterally, and the fourth rib inferolaterally for rejuvenation of the décolletage.
Nodule formation is a well-known complication of PLLA injection, although pain, bruising, edema, pruritus, and hematomas are more commonly seen. The risk of nodule formation can be decreased using several techniques, including avoiding overcorrection and excessive use of product in each individual session, avoiding intradermal injection, diluting to more than 5 mL with reconstitution at least overnight, massaging the area posttreatment (in office by the clinician and at home by the patient), and scheduling treatment sessions at least 4 weeks apart. Usually, 3 to 4 treatments are required and the results can last 2 years or longer without touch-ups.
Nonanimal stabilized hyaluronic acid (NASHA) fillers also can be used to correct chest rhytides; however, using NASHA fillers requires more syringes and results typically last only 6 to 8 months, making it more cost effective to use 2 to 3 vials of PLLA. Moreover, in Vanaman and Fabi’s experience, PLLA is associated with fewer nodules, possibly due to the depth of injection of PLLA into the subcutaneous fat versus injection into the deep dermis with NASHA fillers. Vanaman and Fabi currently are investigating the use of calcium hydroxylapatite fillers alone or in combination with an energy-based modality (microfocused ultrasound) with visualization in the treatment of rhytides in the décolletage.
What’s the Issue?
The availability of many modalities to keep facial skin looking fresh and rejuvenated has led to an increased demand for products and procedures to rejuvenate the décolletage. It is important for dermatologists to acknowledge the more delicate nature of the décolletage versus the face. Less invasive modalities such as cosmetic injectables can be employed in a safe and effective manner to correct rhytides of the chest with proper techniques, products, and patient selection. For a more natural transition from the skin of the face to the décolletage, it also may be necessary to adopt a multimodal approach by using botulinum toxin and fillers, as well as going beyond correction of rhytides to address skin texture and discoloration with chemical peels and lasers. Have you seen an increased demand for procedures to rejuvenate the décolletage in your practice?

As more patients undergo facial rejuvenation procedures for a more youthful look, there is a growing demand for rejuvenation of the décolletage (neck and chest) to achieve a more natural and seamless transition from the skin of the face to the chest. The same modalities that are used on the face to treat skin rhytides, texture, and discoloration have been used successfully in the décolletage area.
Vanaman and Fabi (Plast Reconstr Surg. 2015;136[suppl 5]:276S-281S) recently reviewed the chest anatomy and discussed the safe and effective use of cosmetic injectables alone or in combination with other modalities to address rhytides of the décolletage. The relatively low density of skin pilosebaceous units on the chest allows for slower healing and thus makes the area more vulnerable to scarring with the use of more invasive resurfacing modalities (eg, deeper chemical peels, ablative lasers). The use of cosmetic injectables offers a safer treatment option of chest rhytides. Furthermore, proper candidate selection excludes patients with known sensitivity to cosmetic injectables or their components, history of keloid or hypertrophic scar formation, and active inflammation in the treatment area.
Poly-L-lactic acid (PLLA) is a biodegradable, biocompatible, semipermanent, synthetic soft tissue biostimulator that promotes neocollagenesis by fibroblasts over time (3–6 months). The manufacturer’s recommendation for PLLA reconstitution is at least 2 hours prior to injection with sterile water of no less than 5 mL dilution. Vanaman and Fabi reported usually diluting the day prior to injection with 16 mL total volume. This technique showed the greatest improvement in chest rhytides with no adverse events reported in a retrospective analysis. Poly-L-lactic acid should be injected in a retrograde linear fashion in the plane of the subcutaneous fat, with injection boundaries on the suprasternal notch superiorly, the midclavicular line laterally, and the fourth rib inferolaterally for rejuvenation of the décolletage.
Nodule formation is a well-known complication of PLLA injection, although pain, bruising, edema, pruritus, and hematomas are more commonly seen. The risk of nodule formation can be decreased using several techniques, including avoiding overcorrection and excessive use of product in each individual session, avoiding intradermal injection, diluting to more than 5 mL with reconstitution at least overnight, massaging the area posttreatment (in office by the clinician and at home by the patient), and scheduling treatment sessions at least 4 weeks apart. Usually, 3 to 4 treatments are required and the results can last 2 years or longer without touch-ups.
Nonanimal stabilized hyaluronic acid (NASHA) fillers also can be used to correct chest rhytides; however, using NASHA fillers requires more syringes and results typically last only 6 to 8 months, making it more cost effective to use 2 to 3 vials of PLLA. Moreover, in Vanaman and Fabi’s experience, PLLA is associated with fewer nodules, possibly due to the depth of injection of PLLA into the subcutaneous fat versus injection into the deep dermis with NASHA fillers. Vanaman and Fabi currently are investigating the use of calcium hydroxylapatite fillers alone or in combination with an energy-based modality (microfocused ultrasound) with visualization in the treatment of rhytides in the décolletage.
What’s the Issue?
The availability of many modalities to keep facial skin looking fresh and rejuvenated has led to an increased demand for products and procedures to rejuvenate the décolletage. It is important for dermatologists to acknowledge the more delicate nature of the décolletage versus the face. Less invasive modalities such as cosmetic injectables can be employed in a safe and effective manner to correct rhytides of the chest with proper techniques, products, and patient selection. For a more natural transition from the skin of the face to the décolletage, it also may be necessary to adopt a multimodal approach by using botulinum toxin and fillers, as well as going beyond correction of rhytides to address skin texture and discoloration with chemical peels and lasers. Have you seen an increased demand for procedures to rejuvenate the décolletage in your practice?
Gel patch speeds laser tattoo removal
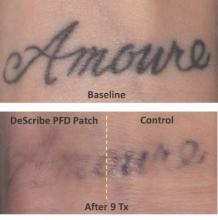
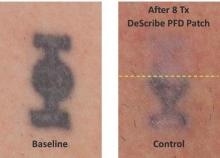
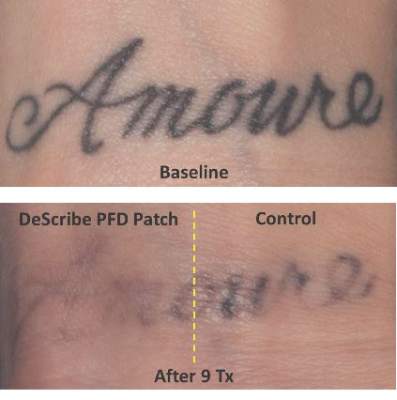
CHICAGO – Treating tattoos through a transparent perfluorodecalin-infused gel patch may cut down on the number of laser treatments needed to remove unwanted tattoos, results of a small pilot study suggest.
In 11 of 17 patients, tattoos treated through the perfluorodecalin (PFD) patch showed more rapid clearance of their tattoos compared with conventional treatment with the same 755-nm Q-switched alexandrite laser, Dr. Brian Biesman said at the annual meeting of the American Society for Dermatologic Surgery.
There were, however, a wide variety of responses, likely reflecting the heterogeneity of the tattoos themselves, ranging from no difference after many treatment sessions to more than 80% clearance after only two sessions, said Dr. Biesman, who is in private practice in Nashville, Tenn. Perfluorodecalin, a fluorocarbon that enhances oxygen delivery, reduces the epidermal whitening that typically limits Q-switched laser tattoo removal to a single pass. Use of the PFD patch in a prior study resolved whitening within an average of 5 seconds, allowing for three to four laser passes in less than 5 minutes (Lasers Surg Med. 2013 Feb;45[2]:76-80).
The DeScribe Transparent PFD Patch (ON Light Sciences) was approved in the United States in April 2015 for use as an accessory to laser tattoo removal procedures using a 755-nm Q-switched alexandrite laser in Fitzpatrick skin type I-III patients.
Anecdotal evidence presented by Dr. Biesman from his own patients suggests that the PFD patch may also speed tattoo clearance when used with different pulse durations and wavelengths, including 5-nanosecond (1,064 nm, 532 nm, and 650 nm) and picosecond (755 nm, 532 nm, and 1,064 nm) lasers.
“It does appear that this patch is efficacious with other Q-switched lasers, both nanosecond and picosecond,” he said, adding that this work is preliminary and under careful study.
The pilot study involved 17 patients with Fitzpatrick skin types I-III and black or blue tattoos treated with a conventional Q-switched alexandrite laser at the maximum tolerable fluence through the PFD patch on one side for three to four passes and with one pass through the air on the other side.
The patch allowed a factor of 1.5-times to 1.8-times higher fluence than control, Dr. Biesman said. In no case did the control side tattoo fade faster than the PFD patch side.
The study was sufficiently powered to qualitatively answer the question of whether the patch could enhance the clearance rate relative to control, but the quantitative rate could not be measured because of the small patient numbers, he noted.
Predictive factors were not identifiable because of the large number of variables, such as tattoo ink, tattoo location, and patient age.
There were no unanticipated adverse events with up to a year of follow-up. Three patients reported three areas of skin hypopigmentation that fully resolved, Dr. Biesman said.
The majority of patients in the pilot study, as well as in a 30-patient pivotal study, reported higher satisfaction with the patch treatment. This seemed to be from a combination of factors, including less discomfort during and after treatment, easier posttreatment recovery, and faster resolution, he said in an interview.
“In our practice, the patch has become the standard of care,” he added. “My nursing staff is highly protective of our patients and will not permit treatment of our patients without the patch. Based on our patients’ objective and subjective experience, I concur.”
Further studies to formally evaluate the performance of the patch in the treatment of benign pigmented lesions and with other Q-switched lasers, using a variety of wavelengths and pulse durations, are currently under consideration.
CHICAGO – Treating tattoos through a transparent perfluorodecalin-infused gel patch may cut down on the number of laser treatments needed to remove unwanted tattoos, results of a small pilot study suggest.
In 11 of 17 patients, tattoos treated through the perfluorodecalin (PFD) patch showed more rapid clearance of their tattoos compared with conventional treatment with the same 755-nm Q-switched alexandrite laser, Dr. Brian Biesman said at the annual meeting of the American Society for Dermatologic Surgery.
There were, however, a wide variety of responses, likely reflecting the heterogeneity of the tattoos themselves, ranging from no difference after many treatment sessions to more than 80% clearance after only two sessions, said Dr. Biesman, who is in private practice in Nashville, Tenn. Perfluorodecalin, a fluorocarbon that enhances oxygen delivery, reduces the epidermal whitening that typically limits Q-switched laser tattoo removal to a single pass. Use of the PFD patch in a prior study resolved whitening within an average of 5 seconds, allowing for three to four laser passes in less than 5 minutes (Lasers Surg Med. 2013 Feb;45[2]:76-80).
The DeScribe Transparent PFD Patch (ON Light Sciences) was approved in the United States in April 2015 for use as an accessory to laser tattoo removal procedures using a 755-nm Q-switched alexandrite laser in Fitzpatrick skin type I-III patients.
Anecdotal evidence presented by Dr. Biesman from his own patients suggests that the PFD patch may also speed tattoo clearance when used with different pulse durations and wavelengths, including 5-nanosecond (1,064 nm, 532 nm, and 650 nm) and picosecond (755 nm, 532 nm, and 1,064 nm) lasers.
“It does appear that this patch is efficacious with other Q-switched lasers, both nanosecond and picosecond,” he said, adding that this work is preliminary and under careful study.
The pilot study involved 17 patients with Fitzpatrick skin types I-III and black or blue tattoos treated with a conventional Q-switched alexandrite laser at the maximum tolerable fluence through the PFD patch on one side for three to four passes and with one pass through the air on the other side.
The patch allowed a factor of 1.5-times to 1.8-times higher fluence than control, Dr. Biesman said. In no case did the control side tattoo fade faster than the PFD patch side.
The study was sufficiently powered to qualitatively answer the question of whether the patch could enhance the clearance rate relative to control, but the quantitative rate could not be measured because of the small patient numbers, he noted.
Predictive factors were not identifiable because of the large number of variables, such as tattoo ink, tattoo location, and patient age.
There were no unanticipated adverse events with up to a year of follow-up. Three patients reported three areas of skin hypopigmentation that fully resolved, Dr. Biesman said.
The majority of patients in the pilot study, as well as in a 30-patient pivotal study, reported higher satisfaction with the patch treatment. This seemed to be from a combination of factors, including less discomfort during and after treatment, easier posttreatment recovery, and faster resolution, he said in an interview.
“In our practice, the patch has become the standard of care,” he added. “My nursing staff is highly protective of our patients and will not permit treatment of our patients without the patch. Based on our patients’ objective and subjective experience, I concur.”
Further studies to formally evaluate the performance of the patch in the treatment of benign pigmented lesions and with other Q-switched lasers, using a variety of wavelengths and pulse durations, are currently under consideration.
CHICAGO – Treating tattoos through a transparent perfluorodecalin-infused gel patch may cut down on the number of laser treatments needed to remove unwanted tattoos, results of a small pilot study suggest.
In 11 of 17 patients, tattoos treated through the perfluorodecalin (PFD) patch showed more rapid clearance of their tattoos compared with conventional treatment with the same 755-nm Q-switched alexandrite laser, Dr. Brian Biesman said at the annual meeting of the American Society for Dermatologic Surgery.
There were, however, a wide variety of responses, likely reflecting the heterogeneity of the tattoos themselves, ranging from no difference after many treatment sessions to more than 80% clearance after only two sessions, said Dr. Biesman, who is in private practice in Nashville, Tenn. Perfluorodecalin, a fluorocarbon that enhances oxygen delivery, reduces the epidermal whitening that typically limits Q-switched laser tattoo removal to a single pass. Use of the PFD patch in a prior study resolved whitening within an average of 5 seconds, allowing for three to four laser passes in less than 5 minutes (Lasers Surg Med. 2013 Feb;45[2]:76-80).
The DeScribe Transparent PFD Patch (ON Light Sciences) was approved in the United States in April 2015 for use as an accessory to laser tattoo removal procedures using a 755-nm Q-switched alexandrite laser in Fitzpatrick skin type I-III patients.
Anecdotal evidence presented by Dr. Biesman from his own patients suggests that the PFD patch may also speed tattoo clearance when used with different pulse durations and wavelengths, including 5-nanosecond (1,064 nm, 532 nm, and 650 nm) and picosecond (755 nm, 532 nm, and 1,064 nm) lasers.
“It does appear that this patch is efficacious with other Q-switched lasers, both nanosecond and picosecond,” he said, adding that this work is preliminary and under careful study.
The pilot study involved 17 patients with Fitzpatrick skin types I-III and black or blue tattoos treated with a conventional Q-switched alexandrite laser at the maximum tolerable fluence through the PFD patch on one side for three to four passes and with one pass through the air on the other side.
The patch allowed a factor of 1.5-times to 1.8-times higher fluence than control, Dr. Biesman said. In no case did the control side tattoo fade faster than the PFD patch side.
The study was sufficiently powered to qualitatively answer the question of whether the patch could enhance the clearance rate relative to control, but the quantitative rate could not be measured because of the small patient numbers, he noted.
Predictive factors were not identifiable because of the large number of variables, such as tattoo ink, tattoo location, and patient age.
There were no unanticipated adverse events with up to a year of follow-up. Three patients reported three areas of skin hypopigmentation that fully resolved, Dr. Biesman said.
The majority of patients in the pilot study, as well as in a 30-patient pivotal study, reported higher satisfaction with the patch treatment. This seemed to be from a combination of factors, including less discomfort during and after treatment, easier posttreatment recovery, and faster resolution, he said in an interview.
“In our practice, the patch has become the standard of care,” he added. “My nursing staff is highly protective of our patients and will not permit treatment of our patients without the patch. Based on our patients’ objective and subjective experience, I concur.”
Further studies to formally evaluate the performance of the patch in the treatment of benign pigmented lesions and with other Q-switched lasers, using a variety of wavelengths and pulse durations, are currently under consideration.
AT THE ASDS ANNUAL MEETING
Key clinical point: A transparent perfluorodecalin-infused patch allows for more rapid, multipass laser tattoo removal.
Major finding: Tattoo clearance was accelerated with the PFD patch in 11 of 17 patients.
Data source: A split-skin pilot study comparing results of laser treatments with and without the patch in 17 patients with black or blue tattoos.
Disclosures: Dr. Biesman reported being a shareholder of ON Light Sciences, the study sponsor and manufacturer of the PFD patch.
For skin rejuvenation, pick the right light-based technique
LAS VEGAS – Technologies to address the many skin changes associated with photoaging run the gamut from nonablative to ablative – and choosing the right technology for a given patient is a balancing act, according to Dr. Mathew Avram.
“Basically, it’s a trade-off between increased efficacy and increased side effects and downtime,” Dr. Avram said at the Skin Disease Education Foundation’s annual Las Vegas dermatology seminar.
Dr. Avram, director of the Massachusetts General Hospital dermatology, laser, and cosmetic center, Boston, said that traditional resurfacing remains the preferred method for repairing photodamaged skin. “One treatment provides reproducible, excellent results,” but at the cost of a significant amount of wound care and prolonged downtime. Potential adverse effects also include clear lines of demarcation with improper feathering, prolonged erythema, permanent hypopigmentation, infection, and scarring, he added.
For these reasons, the popularity of traditional resurfacing to treat photodamaged skin has declined over time as nonablative and intermediate techniques have arisen.
Intense pulsed light (IPL) is a nonlaser, nonablative technique that can produce overall skin rejuvenation, a “side effect” that patients often notice after they receive IPL for vascular lesions or hyperpigmentation. The benefits tend to be modest, even after several treatments, so this is not usually performed as a standalone technique for skin laxity or rhytides.
Nonablative fractional lasers act by creating thousands of microscopic wounds that are “columns of thermal coagulation,” leaving an intact stratum corneum, said Dr. Avram, who is also faculty director for laser and cosmetic training, Harvard Medical School, Boston.
The most common reason for treatment with a nonablative laser is mild to moderate wrinkles, though it can also be used for various pigmentation issues, as well as acne, surgical, and burn scars.
Although side effects are typically minor, the devices can be used off face, and there’s no worry about lines of demarcation; improvement is also more modest and will happen only with multiple treatments. “Fractional treatments give you fractional results,” Dr. Avram commented.
“You want to think about your treatment in terms of the pathology of what you’re treating,” he added, noting that fractional resurfacing can target both epidermal and dermal conditions. Examples of epidermal conditions that benefit from superficial treatment include dermatoheliosis, lentigines, and melasma.
Acne scars and deep wrinkles should receive deeper treatment in order to create thermal damage to the dermis proper, which, over a period of months, will trigger collagen remodeling and new collagen formation. Pulse energy will determine the depth and width of the zone of coagulation in the skin, so higher energy should be used with deeper skin pathology, Dr. Avram said.
Dermatologists also need to consider density of treatment – what percent of the skin is treated. “These devices go from about 5% to about 48% of skin coverage,” and increased density is really what increases the intensity of treatment, he noted. High-density treatment will result in more redness and swelling and has a greater potential for hyperpigmentation, but will not necessarily yield increased benefit in treatment outcomes, he added.
Pretreatment considerations should include assessing the patient’s expectations and availability for downtime post procedure. Treatment planning should also consider the patient’s skin type and probability for sun exposure. Patients should know they will see only partial improvement in wrinkles, scars, and pigmentation, and should expect some discomfort during the procedure and some side effects and healing time post procedure. Patients with a history of herpes labialis should receive valacyclovir 500 mg twice daily, beginning the day before treatment and continuing for 5-7 days, Dr. Avram said.
Cold-air cooling makes the treatment both safer and more comfortable, he pointed out. Topical compounded anesthesia including higher percentages of lidocaine and tetracaine can also be effective. Though local injected anesthesia can be effective in focal treatment of scars, the injection should be deep, and treatment should not begin immediately. This allows the lidocaine to disperse, minimizing the risk of ulceration from the thermal instability that a depot of lidocaine could cause, he said.
Fractional lasers can also be effective for treating background dermatoheliosis, as opposed to targeted treatment of individual lentigines. “Fractional is going to be great for clearing background damage,” Dr. Avram said.
A hybrid technique called ablative fractional photothermolysis creates tiny columns of ablation, while leaving the surrounding untreated tissue available as a reservoir for rapid healing. This technique will improve the more severe wrinkles that nonablative treatments don’t help, with less downtime than a fully ablative technique. Interestingly, patients have less postprocedure pain with this technique than with nonablative techniques, probably because the ablated channels offer a way for heat to escape the skin, he said
Dr. Avram disclosed that he is a paid consultant or has received honoraria from Invasix, Zeltiq Aesthetics, Galderma, and Kythera Biopharmaceuticals; that he is on the scientific advisory board of Cytrellis and Zeltiq; that he has intellectual property and royalty interests with Cytrellis; and that he has received research equipment from Cutera.
SDEF and this news organization are owned by the same parent company.
On Twitter @karioakes
LAS VEGAS – Technologies to address the many skin changes associated with photoaging run the gamut from nonablative to ablative – and choosing the right technology for a given patient is a balancing act, according to Dr. Mathew Avram.
“Basically, it’s a trade-off between increased efficacy and increased side effects and downtime,” Dr. Avram said at the Skin Disease Education Foundation’s annual Las Vegas dermatology seminar.
Dr. Avram, director of the Massachusetts General Hospital dermatology, laser, and cosmetic center, Boston, said that traditional resurfacing remains the preferred method for repairing photodamaged skin. “One treatment provides reproducible, excellent results,” but at the cost of a significant amount of wound care and prolonged downtime. Potential adverse effects also include clear lines of demarcation with improper feathering, prolonged erythema, permanent hypopigmentation, infection, and scarring, he added.
For these reasons, the popularity of traditional resurfacing to treat photodamaged skin has declined over time as nonablative and intermediate techniques have arisen.
Intense pulsed light (IPL) is a nonlaser, nonablative technique that can produce overall skin rejuvenation, a “side effect” that patients often notice after they receive IPL for vascular lesions or hyperpigmentation. The benefits tend to be modest, even after several treatments, so this is not usually performed as a standalone technique for skin laxity or rhytides.
Nonablative fractional lasers act by creating thousands of microscopic wounds that are “columns of thermal coagulation,” leaving an intact stratum corneum, said Dr. Avram, who is also faculty director for laser and cosmetic training, Harvard Medical School, Boston.
The most common reason for treatment with a nonablative laser is mild to moderate wrinkles, though it can also be used for various pigmentation issues, as well as acne, surgical, and burn scars.
Although side effects are typically minor, the devices can be used off face, and there’s no worry about lines of demarcation; improvement is also more modest and will happen only with multiple treatments. “Fractional treatments give you fractional results,” Dr. Avram commented.
“You want to think about your treatment in terms of the pathology of what you’re treating,” he added, noting that fractional resurfacing can target both epidermal and dermal conditions. Examples of epidermal conditions that benefit from superficial treatment include dermatoheliosis, lentigines, and melasma.
Acne scars and deep wrinkles should receive deeper treatment in order to create thermal damage to the dermis proper, which, over a period of months, will trigger collagen remodeling and new collagen formation. Pulse energy will determine the depth and width of the zone of coagulation in the skin, so higher energy should be used with deeper skin pathology, Dr. Avram said.
Dermatologists also need to consider density of treatment – what percent of the skin is treated. “These devices go from about 5% to about 48% of skin coverage,” and increased density is really what increases the intensity of treatment, he noted. High-density treatment will result in more redness and swelling and has a greater potential for hyperpigmentation, but will not necessarily yield increased benefit in treatment outcomes, he added.
Pretreatment considerations should include assessing the patient’s expectations and availability for downtime post procedure. Treatment planning should also consider the patient’s skin type and probability for sun exposure. Patients should know they will see only partial improvement in wrinkles, scars, and pigmentation, and should expect some discomfort during the procedure and some side effects and healing time post procedure. Patients with a history of herpes labialis should receive valacyclovir 500 mg twice daily, beginning the day before treatment and continuing for 5-7 days, Dr. Avram said.
Cold-air cooling makes the treatment both safer and more comfortable, he pointed out. Topical compounded anesthesia including higher percentages of lidocaine and tetracaine can also be effective. Though local injected anesthesia can be effective in focal treatment of scars, the injection should be deep, and treatment should not begin immediately. This allows the lidocaine to disperse, minimizing the risk of ulceration from the thermal instability that a depot of lidocaine could cause, he said.
Fractional lasers can also be effective for treating background dermatoheliosis, as opposed to targeted treatment of individual lentigines. “Fractional is going to be great for clearing background damage,” Dr. Avram said.
A hybrid technique called ablative fractional photothermolysis creates tiny columns of ablation, while leaving the surrounding untreated tissue available as a reservoir for rapid healing. This technique will improve the more severe wrinkles that nonablative treatments don’t help, with less downtime than a fully ablative technique. Interestingly, patients have less postprocedure pain with this technique than with nonablative techniques, probably because the ablated channels offer a way for heat to escape the skin, he said
Dr. Avram disclosed that he is a paid consultant or has received honoraria from Invasix, Zeltiq Aesthetics, Galderma, and Kythera Biopharmaceuticals; that he is on the scientific advisory board of Cytrellis and Zeltiq; that he has intellectual property and royalty interests with Cytrellis; and that he has received research equipment from Cutera.
SDEF and this news organization are owned by the same parent company.
On Twitter @karioakes
LAS VEGAS – Technologies to address the many skin changes associated with photoaging run the gamut from nonablative to ablative – and choosing the right technology for a given patient is a balancing act, according to Dr. Mathew Avram.
“Basically, it’s a trade-off between increased efficacy and increased side effects and downtime,” Dr. Avram said at the Skin Disease Education Foundation’s annual Las Vegas dermatology seminar.
Dr. Avram, director of the Massachusetts General Hospital dermatology, laser, and cosmetic center, Boston, said that traditional resurfacing remains the preferred method for repairing photodamaged skin. “One treatment provides reproducible, excellent results,” but at the cost of a significant amount of wound care and prolonged downtime. Potential adverse effects also include clear lines of demarcation with improper feathering, prolonged erythema, permanent hypopigmentation, infection, and scarring, he added.
For these reasons, the popularity of traditional resurfacing to treat photodamaged skin has declined over time as nonablative and intermediate techniques have arisen.
Intense pulsed light (IPL) is a nonlaser, nonablative technique that can produce overall skin rejuvenation, a “side effect” that patients often notice after they receive IPL for vascular lesions or hyperpigmentation. The benefits tend to be modest, even after several treatments, so this is not usually performed as a standalone technique for skin laxity or rhytides.
Nonablative fractional lasers act by creating thousands of microscopic wounds that are “columns of thermal coagulation,” leaving an intact stratum corneum, said Dr. Avram, who is also faculty director for laser and cosmetic training, Harvard Medical School, Boston.
The most common reason for treatment with a nonablative laser is mild to moderate wrinkles, though it can also be used for various pigmentation issues, as well as acne, surgical, and burn scars.
Although side effects are typically minor, the devices can be used off face, and there’s no worry about lines of demarcation; improvement is also more modest and will happen only with multiple treatments. “Fractional treatments give you fractional results,” Dr. Avram commented.
“You want to think about your treatment in terms of the pathology of what you’re treating,” he added, noting that fractional resurfacing can target both epidermal and dermal conditions. Examples of epidermal conditions that benefit from superficial treatment include dermatoheliosis, lentigines, and melasma.
Acne scars and deep wrinkles should receive deeper treatment in order to create thermal damage to the dermis proper, which, over a period of months, will trigger collagen remodeling and new collagen formation. Pulse energy will determine the depth and width of the zone of coagulation in the skin, so higher energy should be used with deeper skin pathology, Dr. Avram said.
Dermatologists also need to consider density of treatment – what percent of the skin is treated. “These devices go from about 5% to about 48% of skin coverage,” and increased density is really what increases the intensity of treatment, he noted. High-density treatment will result in more redness and swelling and has a greater potential for hyperpigmentation, but will not necessarily yield increased benefit in treatment outcomes, he added.
Pretreatment considerations should include assessing the patient’s expectations and availability for downtime post procedure. Treatment planning should also consider the patient’s skin type and probability for sun exposure. Patients should know they will see only partial improvement in wrinkles, scars, and pigmentation, and should expect some discomfort during the procedure and some side effects and healing time post procedure. Patients with a history of herpes labialis should receive valacyclovir 500 mg twice daily, beginning the day before treatment and continuing for 5-7 days, Dr. Avram said.
Cold-air cooling makes the treatment both safer and more comfortable, he pointed out. Topical compounded anesthesia including higher percentages of lidocaine and tetracaine can also be effective. Though local injected anesthesia can be effective in focal treatment of scars, the injection should be deep, and treatment should not begin immediately. This allows the lidocaine to disperse, minimizing the risk of ulceration from the thermal instability that a depot of lidocaine could cause, he said.
Fractional lasers can also be effective for treating background dermatoheliosis, as opposed to targeted treatment of individual lentigines. “Fractional is going to be great for clearing background damage,” Dr. Avram said.
A hybrid technique called ablative fractional photothermolysis creates tiny columns of ablation, while leaving the surrounding untreated tissue available as a reservoir for rapid healing. This technique will improve the more severe wrinkles that nonablative treatments don’t help, with less downtime than a fully ablative technique. Interestingly, patients have less postprocedure pain with this technique than with nonablative techniques, probably because the ablated channels offer a way for heat to escape the skin, he said
Dr. Avram disclosed that he is a paid consultant or has received honoraria from Invasix, Zeltiq Aesthetics, Galderma, and Kythera Biopharmaceuticals; that he is on the scientific advisory board of Cytrellis and Zeltiq; that he has intellectual property and royalty interests with Cytrellis; and that he has received research equipment from Cutera.
SDEF and this news organization are owned by the same parent company.
On Twitter @karioakes
EXPERT ANALYSIS FROM SDEF LAS VEGAS DERMATOLOGY SEMINAR
Dermatologists’ management of melanoma varies
CHICAGO – Significant variance exists in management of primary cutaneous melanoma, according to a national survey of 510 dermatologists.
Most dermatologists (36%) preferred a shave biopsy for lesions suspected of being melanoma, despite guidelines from the American Academy of Dermatology (AAD) and National Comprehensive Cancer Network (NCCN) guidelines that recommend narrow excision biopsy.
In all, 31% of dermatologists used a narrow local excision (less than 5 mm margin), 13% saucerization/scoop shave biopsy, 11%, punch biopsy, 3% wide local excision, and 7% other.
“The guidelines and academy are all very clear that one of the goals of the biopsy is to obtain tumor depth, so we were surprised that a significant number of providers use shave biopsy or other methods that may leave a risk of not getting the correct depth,” study co-author Dr. Aaron S. Farberg, a melanoma clinical research fellow at the National Society for Cutaneous Medicine in New York City, said in an interview.
Notably, dermatologists in academic and dermatology-based group practices were significantly less likely than those in multispecialty or solo practice to use narrow excision (23% vs. 42%; P < .001).
Although treatment for melanoma evolves continuously, the authors observed that dermatologists remain at the forefront of melanoma management and play a critical role in patient decision making.
“This study suggests that a knowledge gap may exist representing an educational opportunity to more effectively disseminate and implement recommended approaches,” Dr. Farberg and Dr. Darrell Rigel, of New York University School of Medicine, reported in a poster presentation at the annual meeting of the American Society for Dermatologic Surgery.
The survey also revealed that dermatologists are going beyond suggested surgical margins when excising melanoma.
For malignant melanoma in situ (MMIS), 62% used a 5 mm or less margin, 36% a 6 mm to 10 mm margin, and 2% a 1.1 cm to 1.9 cm margin. For these lesions, the AAD recommends a 0.5 cm-1 cm (5 mm-10 mm) margin and the NCCN a 0.5 cm margin, Drs. Farber and Rigel reported.
Academic dermatologists were significantly more likely than all other practice types to refer patients with MMIS out for excision (18% vs. 10%; P < .05).
For invasive melanoma less than 1 mm in depth, both the AAD and NCCN recommend a 1 cm margin (10 mm). In all, 61% of dermatologists reported using 6 mm to 10 mm margins, with 34% opting for 1.1 cm to 1.9 cm margins, 3% at least 2 cm margins, and 2% no more than 5 mm margins.
No significant difference was found across provider types for treatment of melanomas less than 1 mm in depth.
For invasive melanoma greater than 1 mm in depth, 54% of respondents used 1.1 cm to 1.9 cm margins, with most (67%) referring the patient to another provider. Both national guidelines recommend 1 cm to 2 cm margins for melanomas 1 mm to 2 mm in depth and 2 cm margins for melanoma greater than 2 mm in depth.
Academic dermatologists were significantly more likely than other dermatologists to treat these lesions rather than to refer the patient out (51% vs. 30%; P < .001).
“This is exciting new data that suggests that there still is a variance in early melanoma management,” Dr. Rigel, past president of AAD and ASDS, said in an interview. “The data suggest more studies need to be done to better access why this is occurring.”
Dr. Hensin Tsao, who served on the 2011 AAD guideline working group and is co-chairing the AAD’s pending guideline update, said in an interview that, “Dr. Rigel is well respected in the field and the project will undoubtedly be submitted for publication and subject to further review. It is worthwhile to wait on the final published results and conclusions.”
He agreed, however, with the authors’ suggestion that there is significant variation in practice. Regarding the finding that 36% of respondents use a shave biopsy for suspicious lesions, the AAD guidelines recommend that the entire lesion be removed with a 1 mm to 3 mm margin, which can be accomplished by an elliptical or punch excision with sutures or shave removal to a depth below the anticipated plane of the lesion, Dr. Tsao, of Massachusetts General Hospital in Boston, said.
“It is quite possible that some of the respondents to the questionnaire interpreted ‘shave biopsy’ as a full shave disk excision,” he said. “That said, intentional and routine partial sampling of suspected melanomas would be at odds with the guidelines.”
It is not inappropriate to remove a suspicious lesion, if small enough, with a punch biopsy, Dr. Tsao said, adding, “Perhaps again, the respondents failed to distinguish between partial punch biopsy and punch excision.”
On occasion, the AAD guidelines also make accommodations for an incisional biopsy “of the clinically or dermatoscopically most atypical portion of the lesion.” In this situation, a smaller incisional punch biopsy may be performed of the highly suspicious area.
“There are certainly areas that the investigators have identified which could represent potential knowledge gaps,” Dr. Tsao said. “For instance, 14% of the respondents used a 0.6 cm to 1.0 cm (6 mm-10 mm) margin for melanoma greater than 1 mm.”
Dermatologists’ surveillance of patients in the survey was somewhat less divergent. The most recommended follow-up interval for patients diagnosed within the last 5 years, was 6 months (49%), followed by a 3-month interval (25%), and other (24%). Follow-up was extended to yearly by 63% of dermatologists for patients diagnosed more than five years earlier, Dr. Farberg and Dr. Rigel reported.
Both the AAD and NCCN recommend follow-up every 3 to 12 months for those diagnosed with melanoma within the previous 5 years, while for those diagnosed more than 5 years prior, the AAD recommends follow-up every 3 to 12 months and the NCCN every 12 months.
“Adherence to evidence-based guidelines should lead to improved patient outcomes and quality of care. However, newer studies and emerging data may also justify deviations from existing guidelines, suggesting review of those guidelines may be indicated,” Dr. Farberg and Dr. Rigel concluded.
For example, a prospective series examining surgical margins in 1,072 patients with 1,120 malignant melanoma in situs showed that only 86% were successfully excised with a 6-mm margin, significantly less than the 98.9% clearance achieved with a 9-mm margin, Dr. Farberg told this publication.
That said, a 9-mm margin may not be appropriate for all lesions, such as those on the face, he observed.
The investigators said they hope the survey results will spur on revision of the guidelines, last updated in 2011.
Limitations of the survey include a lack of information on clinical factors such as patient history or anatomic site, the survey may have led to generalized answers, and access to care and reimbursement also may have impacted management, the authors noted.
The response rate to the survey was also low at 8%, with 6,177 practicing U.S. dermatologists surveyed. The demographics of the respondents, however, strongly reflected the full AAD membership, Dr. Farberg said. No significant geographical differences were observed.
CHICAGO – Significant variance exists in management of primary cutaneous melanoma, according to a national survey of 510 dermatologists.
Most dermatologists (36%) preferred a shave biopsy for lesions suspected of being melanoma, despite guidelines from the American Academy of Dermatology (AAD) and National Comprehensive Cancer Network (NCCN) guidelines that recommend narrow excision biopsy.
In all, 31% of dermatologists used a narrow local excision (less than 5 mm margin), 13% saucerization/scoop shave biopsy, 11%, punch biopsy, 3% wide local excision, and 7% other.
“The guidelines and academy are all very clear that one of the goals of the biopsy is to obtain tumor depth, so we were surprised that a significant number of providers use shave biopsy or other methods that may leave a risk of not getting the correct depth,” study co-author Dr. Aaron S. Farberg, a melanoma clinical research fellow at the National Society for Cutaneous Medicine in New York City, said in an interview.
Notably, dermatologists in academic and dermatology-based group practices were significantly less likely than those in multispecialty or solo practice to use narrow excision (23% vs. 42%; P < .001).
Although treatment for melanoma evolves continuously, the authors observed that dermatologists remain at the forefront of melanoma management and play a critical role in patient decision making.
“This study suggests that a knowledge gap may exist representing an educational opportunity to more effectively disseminate and implement recommended approaches,” Dr. Farberg and Dr. Darrell Rigel, of New York University School of Medicine, reported in a poster presentation at the annual meeting of the American Society for Dermatologic Surgery.
The survey also revealed that dermatologists are going beyond suggested surgical margins when excising melanoma.
For malignant melanoma in situ (MMIS), 62% used a 5 mm or less margin, 36% a 6 mm to 10 mm margin, and 2% a 1.1 cm to 1.9 cm margin. For these lesions, the AAD recommends a 0.5 cm-1 cm (5 mm-10 mm) margin and the NCCN a 0.5 cm margin, Drs. Farber and Rigel reported.
Academic dermatologists were significantly more likely than all other practice types to refer patients with MMIS out for excision (18% vs. 10%; P < .05).
For invasive melanoma less than 1 mm in depth, both the AAD and NCCN recommend a 1 cm margin (10 mm). In all, 61% of dermatologists reported using 6 mm to 10 mm margins, with 34% opting for 1.1 cm to 1.9 cm margins, 3% at least 2 cm margins, and 2% no more than 5 mm margins.
No significant difference was found across provider types for treatment of melanomas less than 1 mm in depth.
For invasive melanoma greater than 1 mm in depth, 54% of respondents used 1.1 cm to 1.9 cm margins, with most (67%) referring the patient to another provider. Both national guidelines recommend 1 cm to 2 cm margins for melanomas 1 mm to 2 mm in depth and 2 cm margins for melanoma greater than 2 mm in depth.
Academic dermatologists were significantly more likely than other dermatologists to treat these lesions rather than to refer the patient out (51% vs. 30%; P < .001).
“This is exciting new data that suggests that there still is a variance in early melanoma management,” Dr. Rigel, past president of AAD and ASDS, said in an interview. “The data suggest more studies need to be done to better access why this is occurring.”
Dr. Hensin Tsao, who served on the 2011 AAD guideline working group and is co-chairing the AAD’s pending guideline update, said in an interview that, “Dr. Rigel is well respected in the field and the project will undoubtedly be submitted for publication and subject to further review. It is worthwhile to wait on the final published results and conclusions.”
He agreed, however, with the authors’ suggestion that there is significant variation in practice. Regarding the finding that 36% of respondents use a shave biopsy for suspicious lesions, the AAD guidelines recommend that the entire lesion be removed with a 1 mm to 3 mm margin, which can be accomplished by an elliptical or punch excision with sutures or shave removal to a depth below the anticipated plane of the lesion, Dr. Tsao, of Massachusetts General Hospital in Boston, said.
“It is quite possible that some of the respondents to the questionnaire interpreted ‘shave biopsy’ as a full shave disk excision,” he said. “That said, intentional and routine partial sampling of suspected melanomas would be at odds with the guidelines.”
It is not inappropriate to remove a suspicious lesion, if small enough, with a punch biopsy, Dr. Tsao said, adding, “Perhaps again, the respondents failed to distinguish between partial punch biopsy and punch excision.”
On occasion, the AAD guidelines also make accommodations for an incisional biopsy “of the clinically or dermatoscopically most atypical portion of the lesion.” In this situation, a smaller incisional punch biopsy may be performed of the highly suspicious area.
“There are certainly areas that the investigators have identified which could represent potential knowledge gaps,” Dr. Tsao said. “For instance, 14% of the respondents used a 0.6 cm to 1.0 cm (6 mm-10 mm) margin for melanoma greater than 1 mm.”
Dermatologists’ surveillance of patients in the survey was somewhat less divergent. The most recommended follow-up interval for patients diagnosed within the last 5 years, was 6 months (49%), followed by a 3-month interval (25%), and other (24%). Follow-up was extended to yearly by 63% of dermatologists for patients diagnosed more than five years earlier, Dr. Farberg and Dr. Rigel reported.
Both the AAD and NCCN recommend follow-up every 3 to 12 months for those diagnosed with melanoma within the previous 5 years, while for those diagnosed more than 5 years prior, the AAD recommends follow-up every 3 to 12 months and the NCCN every 12 months.
“Adherence to evidence-based guidelines should lead to improved patient outcomes and quality of care. However, newer studies and emerging data may also justify deviations from existing guidelines, suggesting review of those guidelines may be indicated,” Dr. Farberg and Dr. Rigel concluded.
For example, a prospective series examining surgical margins in 1,072 patients with 1,120 malignant melanoma in situs showed that only 86% were successfully excised with a 6-mm margin, significantly less than the 98.9% clearance achieved with a 9-mm margin, Dr. Farberg told this publication.
That said, a 9-mm margin may not be appropriate for all lesions, such as those on the face, he observed.
The investigators said they hope the survey results will spur on revision of the guidelines, last updated in 2011.
Limitations of the survey include a lack of information on clinical factors such as patient history or anatomic site, the survey may have led to generalized answers, and access to care and reimbursement also may have impacted management, the authors noted.
The response rate to the survey was also low at 8%, with 6,177 practicing U.S. dermatologists surveyed. The demographics of the respondents, however, strongly reflected the full AAD membership, Dr. Farberg said. No significant geographical differences were observed.
CHICAGO – Significant variance exists in management of primary cutaneous melanoma, according to a national survey of 510 dermatologists.
Most dermatologists (36%) preferred a shave biopsy for lesions suspected of being melanoma, despite guidelines from the American Academy of Dermatology (AAD) and National Comprehensive Cancer Network (NCCN) guidelines that recommend narrow excision biopsy.
In all, 31% of dermatologists used a narrow local excision (less than 5 mm margin), 13% saucerization/scoop shave biopsy, 11%, punch biopsy, 3% wide local excision, and 7% other.
“The guidelines and academy are all very clear that one of the goals of the biopsy is to obtain tumor depth, so we were surprised that a significant number of providers use shave biopsy or other methods that may leave a risk of not getting the correct depth,” study co-author Dr. Aaron S. Farberg, a melanoma clinical research fellow at the National Society for Cutaneous Medicine in New York City, said in an interview.
Notably, dermatologists in academic and dermatology-based group practices were significantly less likely than those in multispecialty or solo practice to use narrow excision (23% vs. 42%; P < .001).
Although treatment for melanoma evolves continuously, the authors observed that dermatologists remain at the forefront of melanoma management and play a critical role in patient decision making.
“This study suggests that a knowledge gap may exist representing an educational opportunity to more effectively disseminate and implement recommended approaches,” Dr. Farberg and Dr. Darrell Rigel, of New York University School of Medicine, reported in a poster presentation at the annual meeting of the American Society for Dermatologic Surgery.
The survey also revealed that dermatologists are going beyond suggested surgical margins when excising melanoma.
For malignant melanoma in situ (MMIS), 62% used a 5 mm or less margin, 36% a 6 mm to 10 mm margin, and 2% a 1.1 cm to 1.9 cm margin. For these lesions, the AAD recommends a 0.5 cm-1 cm (5 mm-10 mm) margin and the NCCN a 0.5 cm margin, Drs. Farber and Rigel reported.
Academic dermatologists were significantly more likely than all other practice types to refer patients with MMIS out for excision (18% vs. 10%; P < .05).
For invasive melanoma less than 1 mm in depth, both the AAD and NCCN recommend a 1 cm margin (10 mm). In all, 61% of dermatologists reported using 6 mm to 10 mm margins, with 34% opting for 1.1 cm to 1.9 cm margins, 3% at least 2 cm margins, and 2% no more than 5 mm margins.
No significant difference was found across provider types for treatment of melanomas less than 1 mm in depth.
For invasive melanoma greater than 1 mm in depth, 54% of respondents used 1.1 cm to 1.9 cm margins, with most (67%) referring the patient to another provider. Both national guidelines recommend 1 cm to 2 cm margins for melanomas 1 mm to 2 mm in depth and 2 cm margins for melanoma greater than 2 mm in depth.
Academic dermatologists were significantly more likely than other dermatologists to treat these lesions rather than to refer the patient out (51% vs. 30%; P < .001).
“This is exciting new data that suggests that there still is a variance in early melanoma management,” Dr. Rigel, past president of AAD and ASDS, said in an interview. “The data suggest more studies need to be done to better access why this is occurring.”
Dr. Hensin Tsao, who served on the 2011 AAD guideline working group and is co-chairing the AAD’s pending guideline update, said in an interview that, “Dr. Rigel is well respected in the field and the project will undoubtedly be submitted for publication and subject to further review. It is worthwhile to wait on the final published results and conclusions.”
He agreed, however, with the authors’ suggestion that there is significant variation in practice. Regarding the finding that 36% of respondents use a shave biopsy for suspicious lesions, the AAD guidelines recommend that the entire lesion be removed with a 1 mm to 3 mm margin, which can be accomplished by an elliptical or punch excision with sutures or shave removal to a depth below the anticipated plane of the lesion, Dr. Tsao, of Massachusetts General Hospital in Boston, said.
“It is quite possible that some of the respondents to the questionnaire interpreted ‘shave biopsy’ as a full shave disk excision,” he said. “That said, intentional and routine partial sampling of suspected melanomas would be at odds with the guidelines.”
It is not inappropriate to remove a suspicious lesion, if small enough, with a punch biopsy, Dr. Tsao said, adding, “Perhaps again, the respondents failed to distinguish between partial punch biopsy and punch excision.”
On occasion, the AAD guidelines also make accommodations for an incisional biopsy “of the clinically or dermatoscopically most atypical portion of the lesion.” In this situation, a smaller incisional punch biopsy may be performed of the highly suspicious area.
“There are certainly areas that the investigators have identified which could represent potential knowledge gaps,” Dr. Tsao said. “For instance, 14% of the respondents used a 0.6 cm to 1.0 cm (6 mm-10 mm) margin for melanoma greater than 1 mm.”
Dermatologists’ surveillance of patients in the survey was somewhat less divergent. The most recommended follow-up interval for patients diagnosed within the last 5 years, was 6 months (49%), followed by a 3-month interval (25%), and other (24%). Follow-up was extended to yearly by 63% of dermatologists for patients diagnosed more than five years earlier, Dr. Farberg and Dr. Rigel reported.
Both the AAD and NCCN recommend follow-up every 3 to 12 months for those diagnosed with melanoma within the previous 5 years, while for those diagnosed more than 5 years prior, the AAD recommends follow-up every 3 to 12 months and the NCCN every 12 months.
“Adherence to evidence-based guidelines should lead to improved patient outcomes and quality of care. However, newer studies and emerging data may also justify deviations from existing guidelines, suggesting review of those guidelines may be indicated,” Dr. Farberg and Dr. Rigel concluded.
For example, a prospective series examining surgical margins in 1,072 patients with 1,120 malignant melanoma in situs showed that only 86% were successfully excised with a 6-mm margin, significantly less than the 98.9% clearance achieved with a 9-mm margin, Dr. Farberg told this publication.
That said, a 9-mm margin may not be appropriate for all lesions, such as those on the face, he observed.
The investigators said they hope the survey results will spur on revision of the guidelines, last updated in 2011.
Limitations of the survey include a lack of information on clinical factors such as patient history or anatomic site, the survey may have led to generalized answers, and access to care and reimbursement also may have impacted management, the authors noted.
The response rate to the survey was also low at 8%, with 6,177 practicing U.S. dermatologists surveyed. The demographics of the respondents, however, strongly reflected the full AAD membership, Dr. Farberg said. No significant geographical differences were observed.
AT THE ASDS ANNUAL MEETING
Key clinical point: Significant variance exists in dermatologists’ management of melanoma.
Major finding: Only 31% of dermatologists used narrow excision as recommended to biopsy suspicious lesions.
Data source: Survey of 510 U.S. practicing dermatologists.
Disclosures: The authors reported having no financial disclosures.
Survey illustrates opportunities for dermatologists to help manage tattoo risks, regrets
About a fifth of adults reported a history of an itchy tattoo, and most said they believed that dermatologists are knowledgeable about managing tattoo complications, according to a survey of 501 adults with one or more tattoos.
The survey, conducted by researchers at Tulane University, New Orleans, found that almost 23% had had pruritus at the tattoo site more than a month after being tattooed. In addition, 3.2% said they had a tattoo that had become infected; these people were significantly more likely to have more tattoos and to have undergone more tattooing sessions, compared with those who had not had an infected tattoo.
In addition, 3.8% said they had pain at the site of at least one tattoo after the 1-month mark, reported Dr. Walter Liszewski of Tulane University and his coauthors (Dermatol Surg. 2015 Nov;41[11]:1283-9).
Their 18-question survey was administered to 501 people aged 18 years and older with at least one tattoo in the French Quarter over a 3-day period in January 2015. They were from 38 states, Puerto Rico, and Washington, D.C.; almost 55% were women.
When it came to where to seek help managing these tattoo-related complications and misgivings, the dermatologist’s office ran second to the tattoo parlor. Of those surveyed, 93.2% of all participants believed tattoo artists were “very knowledgeable” or “knowledgeable” about the allergic and infectious tattoo complications, while 78.8% reported thinking the same about dermatologists – which suggests “that there is considerable trust in the ability of dermatologists to manage tattoo complications,” the authors pointed out.
Almost 70% thought primary care physicians and almost 40% thought pharmacists were similarly competent.
Almost a fifth of those surveyed said they had received at least one tattoo in a setting other than a tattoo parlor, such as in prison or at a party. And about a fifth said they had received one or more tattoos while inebriated.
The rates of tattoo regret were higher among those who were younger when they had received their first tattoo, ranging from 11.4% of those who were aged 21 years and older when they got their first tattoo to 35.1% of those who had their first tattoo before aged 18 years. Moreover, almost 43% of those who had their first tattoo before age 18 said they were would be interested in having at least one tattoo removed for free, compared with 19.1% and 14.4% of those who received their first tattoo between ages 18 and 20 years and at ages 21 and older, respectively.
“Given the incidence of pruritic tattoos, tattoo regret, unsafe tattooing practices, and desire to have tattoos removed among the study participants, dermatologists have an opportunity to provide medical and cosmetic care, as well as anticipatory guidance, for these patients,” the authors concluded.
On Twitter @whitneymcknight
About a fifth of adults reported a history of an itchy tattoo, and most said they believed that dermatologists are knowledgeable about managing tattoo complications, according to a survey of 501 adults with one or more tattoos.
The survey, conducted by researchers at Tulane University, New Orleans, found that almost 23% had had pruritus at the tattoo site more than a month after being tattooed. In addition, 3.2% said they had a tattoo that had become infected; these people were significantly more likely to have more tattoos and to have undergone more tattooing sessions, compared with those who had not had an infected tattoo.
In addition, 3.8% said they had pain at the site of at least one tattoo after the 1-month mark, reported Dr. Walter Liszewski of Tulane University and his coauthors (Dermatol Surg. 2015 Nov;41[11]:1283-9).
Their 18-question survey was administered to 501 people aged 18 years and older with at least one tattoo in the French Quarter over a 3-day period in January 2015. They were from 38 states, Puerto Rico, and Washington, D.C.; almost 55% were women.
When it came to where to seek help managing these tattoo-related complications and misgivings, the dermatologist’s office ran second to the tattoo parlor. Of those surveyed, 93.2% of all participants believed tattoo artists were “very knowledgeable” or “knowledgeable” about the allergic and infectious tattoo complications, while 78.8% reported thinking the same about dermatologists – which suggests “that there is considerable trust in the ability of dermatologists to manage tattoo complications,” the authors pointed out.
Almost 70% thought primary care physicians and almost 40% thought pharmacists were similarly competent.
Almost a fifth of those surveyed said they had received at least one tattoo in a setting other than a tattoo parlor, such as in prison or at a party. And about a fifth said they had received one or more tattoos while inebriated.
The rates of tattoo regret were higher among those who were younger when they had received their first tattoo, ranging from 11.4% of those who were aged 21 years and older when they got their first tattoo to 35.1% of those who had their first tattoo before aged 18 years. Moreover, almost 43% of those who had their first tattoo before age 18 said they were would be interested in having at least one tattoo removed for free, compared with 19.1% and 14.4% of those who received their first tattoo between ages 18 and 20 years and at ages 21 and older, respectively.
“Given the incidence of pruritic tattoos, tattoo regret, unsafe tattooing practices, and desire to have tattoos removed among the study participants, dermatologists have an opportunity to provide medical and cosmetic care, as well as anticipatory guidance, for these patients,” the authors concluded.
On Twitter @whitneymcknight
About a fifth of adults reported a history of an itchy tattoo, and most said they believed that dermatologists are knowledgeable about managing tattoo complications, according to a survey of 501 adults with one or more tattoos.
The survey, conducted by researchers at Tulane University, New Orleans, found that almost 23% had had pruritus at the tattoo site more than a month after being tattooed. In addition, 3.2% said they had a tattoo that had become infected; these people were significantly more likely to have more tattoos and to have undergone more tattooing sessions, compared with those who had not had an infected tattoo.
In addition, 3.8% said they had pain at the site of at least one tattoo after the 1-month mark, reported Dr. Walter Liszewski of Tulane University and his coauthors (Dermatol Surg. 2015 Nov;41[11]:1283-9).
Their 18-question survey was administered to 501 people aged 18 years and older with at least one tattoo in the French Quarter over a 3-day period in January 2015. They were from 38 states, Puerto Rico, and Washington, D.C.; almost 55% were women.
When it came to where to seek help managing these tattoo-related complications and misgivings, the dermatologist’s office ran second to the tattoo parlor. Of those surveyed, 93.2% of all participants believed tattoo artists were “very knowledgeable” or “knowledgeable” about the allergic and infectious tattoo complications, while 78.8% reported thinking the same about dermatologists – which suggests “that there is considerable trust in the ability of dermatologists to manage tattoo complications,” the authors pointed out.
Almost 70% thought primary care physicians and almost 40% thought pharmacists were similarly competent.
Almost a fifth of those surveyed said they had received at least one tattoo in a setting other than a tattoo parlor, such as in prison or at a party. And about a fifth said they had received one or more tattoos while inebriated.
The rates of tattoo regret were higher among those who were younger when they had received their first tattoo, ranging from 11.4% of those who were aged 21 years and older when they got their first tattoo to 35.1% of those who had their first tattoo before aged 18 years. Moreover, almost 43% of those who had their first tattoo before age 18 said they were would be interested in having at least one tattoo removed for free, compared with 19.1% and 14.4% of those who received their first tattoo between ages 18 and 20 years and at ages 21 and older, respectively.
“Given the incidence of pruritic tattoos, tattoo regret, unsafe tattooing practices, and desire to have tattoos removed among the study participants, dermatologists have an opportunity to provide medical and cosmetic care, as well as anticipatory guidance, for these patients,” the authors concluded.
On Twitter @whitneymcknight
FROM DERMATOLOGIC SURGERY
Key clinical point: Opportunities exist for dermatologists to provide tattoo-related medical and cosmetic care.
Major finding: About one-fifth of surveyed adults reported having had a pruritic tattoo at least 1 month after receiving the tattoo; most said they believed dermatologists could help manage tattoo complications.
Data source: A survey of 501 adults with at least one tattoo, conducted in New Orleans, which included questions about tattoo complications, regret, and practices.
Disclosures: The authors reported no relevant financial disclosures.
AK clearance rates maintained 1 year after thermally modulated PDT
Clearance rates for actinic keratoses (AKs) on the extremities were prolonged by adding thermal modulation to photodynamic therapy (PDT), a study has shown.
The single-site study of 17 adults with 10 or more AKs on their arms or legs evaluated thermally modulated PDT: 20% 5-aminolevulinic acid (ALA) applied to skin, occluded with plastic wrap and warmed with an electric heating pad to a median 41.2° C for one hour, then exposed to blue light. Treatment resulted in 90% clearance rates at 3 months and 12 months, reported Dr. Andrea Willey, a dermatologist in Sacramento, Calif., and her associates (Dermatol Surg. 2015 Nov;41[11]:1290-5).
They were inspired to conduct the study as a follow-on to a recently published pilot study of three patients, which found that clearance rates of AKs on the arm treated with thermally modulated 5-ALA blue light PDT persisted up to 14 months after treatment, when compared with the control arm, which was not heated.
In their study, there were 724 grade 1 and 2 AKs at baseline – a median of 15 per extremity; patients were followed up at 3, 6, 9, and 12 months. The total lesion count across the entire cohort was 70 at 3 months, 69 at 6 months, 64 at 9 months, and 72 at 1 year. No new AKs were seen in the treated areas during the 1-year follow-up.
There were 13 grade 3 hypertrophic AK lesions among all study participants. Although these lesions persisted, Dr. Willey and her coauthors noted that they did shrink initially after treatment.
Adverse events associated with the combination therapy were low, and included mild erythema and crusting up to 1 week post treatment, indicating the therapy was well tolerated, and patient satisfaction was high.
The results “suggest that mild skin warming may both improve efficacy and reduce variability of response to PDT in practice,” the authors wrote.
On Twitter @whitneymcknight
Clearance rates for actinic keratoses (AKs) on the extremities were prolonged by adding thermal modulation to photodynamic therapy (PDT), a study has shown.
The single-site study of 17 adults with 10 or more AKs on their arms or legs evaluated thermally modulated PDT: 20% 5-aminolevulinic acid (ALA) applied to skin, occluded with plastic wrap and warmed with an electric heating pad to a median 41.2° C for one hour, then exposed to blue light. Treatment resulted in 90% clearance rates at 3 months and 12 months, reported Dr. Andrea Willey, a dermatologist in Sacramento, Calif., and her associates (Dermatol Surg. 2015 Nov;41[11]:1290-5).
They were inspired to conduct the study as a follow-on to a recently published pilot study of three patients, which found that clearance rates of AKs on the arm treated with thermally modulated 5-ALA blue light PDT persisted up to 14 months after treatment, when compared with the control arm, which was not heated.
In their study, there were 724 grade 1 and 2 AKs at baseline – a median of 15 per extremity; patients were followed up at 3, 6, 9, and 12 months. The total lesion count across the entire cohort was 70 at 3 months, 69 at 6 months, 64 at 9 months, and 72 at 1 year. No new AKs were seen in the treated areas during the 1-year follow-up.
There were 13 grade 3 hypertrophic AK lesions among all study participants. Although these lesions persisted, Dr. Willey and her coauthors noted that they did shrink initially after treatment.
Adverse events associated with the combination therapy were low, and included mild erythema and crusting up to 1 week post treatment, indicating the therapy was well tolerated, and patient satisfaction was high.
The results “suggest that mild skin warming may both improve efficacy and reduce variability of response to PDT in practice,” the authors wrote.
On Twitter @whitneymcknight
Clearance rates for actinic keratoses (AKs) on the extremities were prolonged by adding thermal modulation to photodynamic therapy (PDT), a study has shown.
The single-site study of 17 adults with 10 or more AKs on their arms or legs evaluated thermally modulated PDT: 20% 5-aminolevulinic acid (ALA) applied to skin, occluded with plastic wrap and warmed with an electric heating pad to a median 41.2° C for one hour, then exposed to blue light. Treatment resulted in 90% clearance rates at 3 months and 12 months, reported Dr. Andrea Willey, a dermatologist in Sacramento, Calif., and her associates (Dermatol Surg. 2015 Nov;41[11]:1290-5).
They were inspired to conduct the study as a follow-on to a recently published pilot study of three patients, which found that clearance rates of AKs on the arm treated with thermally modulated 5-ALA blue light PDT persisted up to 14 months after treatment, when compared with the control arm, which was not heated.
In their study, there were 724 grade 1 and 2 AKs at baseline – a median of 15 per extremity; patients were followed up at 3, 6, 9, and 12 months. The total lesion count across the entire cohort was 70 at 3 months, 69 at 6 months, 64 at 9 months, and 72 at 1 year. No new AKs were seen in the treated areas during the 1-year follow-up.
There were 13 grade 3 hypertrophic AK lesions among all study participants. Although these lesions persisted, Dr. Willey and her coauthors noted that they did shrink initially after treatment.
Adverse events associated with the combination therapy were low, and included mild erythema and crusting up to 1 week post treatment, indicating the therapy was well tolerated, and patient satisfaction was high.
The results “suggest that mild skin warming may both improve efficacy and reduce variability of response to PDT in practice,” the authors wrote.
On Twitter @whitneymcknight
FROM DERMATOLOGIC SURGERY
Key clinical point: Mildly warming affected skin sites during photodynamic therapy can prolong the clearance rates for AKs on the extremities.
Major finding: AK clearance rates on the extremities remained at about 90% during the 1 year after treatment with thermally modulated PDT.
Data source: A single site, prospective 1-year study evaluating treatment with thermally modulated PDT for 17 adults with 10 or more AKs on their extremities.
Disclosures: Dr. Willey is a member of the scientific advisory board of DUSA Pharmaceuticals; she received a research grant, equipment loan, and study drug from the company. Her two coauthors had no relevant disclosures.
Fillers in Dermatology: From Past to Present
Fillers are products that are injected into soft tissue and are classified as either resorbable or nonresorbable (permanent). Several dermal and subcutaneous fillers for soft tissue augmentation currently are available. This article provides a brief review of the history of filler agents currently available for soft tissue augmentation. Cadaveric-derived fillers and implants will not be discussed, as these materials are expensive, are not all approved by the US Food and Drug Administration (FDA), and are more commonly used in burn victims than in cosmetic patients.
History of Fillers in Dermatology
The first known injectable agent was paraffin, but its use as a dermal filler was abandoned after complications including embolization, migration (ie, movement into surrounding tissue), and granuloma formation were reported.1 Silicone also was banned by the FDA for use as a soft tissue filler due to similar complications. Bovine collagen was the first agent to be approved by the FDA for cosmetic injection, and since then many filler agents have revolutionized a new era of cosmetic enhancement.1 Injectable soft tissue fillers now can be classified based on several characteristics, including site of placement (ie, dermal or subcutaneous), animal versus nonanimal derivation (eg, autologous, xenograft, semisynthetic, synthetic), and duration of effects (temporary [<6 months], long-lasting [6 months– 2 years], semipermanent [2–5 years], permanent [>5 years]).2
In the 19th century, early instances of soft tissue augmentation using dermal fillers included autologous fat harvested from the arms for correction of depressed facial defects and scars in a patient with tuberculous osteitis as well as injection of paraffin into the scrotum as a testicular prosthesis in a patient with advanced tuberculosis.1 Later, a technique using a syringe to transfer autologous fat from the extremities was used for facial soft tissue augmentation and contouring, and permanent facial soft tissue augmentation using liquid silicone also was performed. In 1981, purified bovine dermal collagen was first approved by the FDA as a xenogenic agent for dermal injection.3
In 2003, the FDA began to approve new fillers for temporary soft tissue augmentation.3 Approval of injectable purified human collagen derived from fibroblasts was followed by another class of new agents: hyaluronic acid fillers.1 A biodegradable non–animal based stabilized hyaluronic acid was followed by more agents derived from rooster combs. Further investigations and research have continued, and more long-lasting synthetic fillers have become available, including calcium hydroxylapatite and poly-L-lactic acid. A renewed interest in permanent agents and longer lasting products such as silicone oil and polymethyl methacrylate also has emerged.4
The Future of Fillers
While earlier filler materials were limited, those used today are composed of a wide range of substances including collagen, hyaluronic acid, calcium hydroxylapatite, poly-L-lactic acid, and synthetic or manmade polymers (Table). The FDA has approved approximately 21 filler products for dermatologic indications, each with unique properties, advantages, and disadvantages.3 For example, there are several fillers that not only provide soft tissue augmentation but also stimulate collagen production. However, complications can occur, often many years after the initial treatment. Side effects such as swelling, erythema, and nodules may occur and in rare instances foreign body granulomas may develop and may be difficult to eradicate.5 Except for autologous fat, all fillers are foreign bodies and therefore can cause foreign body granulomatous reactions ranging from common (eg, with paraffin) to rare (eg, with hyaluronic acid) in occurrence.6 The clinical presentation of these reactions is variable, ranging from single to multiple nodules at the injection site to diffuse, hard swelling of the face accompanied by reddening of the skin.
With the increasing desire for a youthful appearance among the aging population, the pharmaceutical industry has responded by increasing the number of available treatment options to meet the demands of cosmetic patients, one of the fastest growing subpopulations in the field of dermatology. Fillers also have provided new options for patients who are unable to afford plastic surgery or those who are poor surgical candidates. The ideal filler material is nonallergenic, noncarcinogenic, and nonteratogenic. It should be stable, affordable, malleable, reversible, and durable with results that can be reproduced and should cause minimal inflammation, migration, and detectable changes. The ideal filler also should have predictable and consistent results, feel natural, take little time to administer, require minimal preparation, cause no patient downtime, and have a low risk for complications. Ideal administration should be painless, user-friendly, and conducted in an outpatient setting with minimal recovery and easy storage. Although no single filler is ideal for all patients, indications, and situations, residents should be aware of the properties and characteristics that make each product unique in order to optimize treatment in all patients.
As demands for cosmetic procedures increase, it is important to incorporate knowledge of cosmetic procedures (eg, fillers for soft tissue augmentation) in resident education and training. Although cosmetic dermatology has been featured prominently in dermatology residency, surveys have shown that residents desire more training in this area.7 Although lectures on soft tissue augmentation are popular topics in dermatology, hands-on experience performing these procedures varies widely depending on different training programs. My institution offers several lectures on cosmetic dermatology, and residents are able to perform procedures for soft tissue augmentation as the first assistant or first surgeon during our cosmetic clinic sessions twice weekly.
Final Thoughts
There are a variety of fillers on the horizon to improve aging and volume loss and the science behind cosmetic injections is evolving. Regardless of the filler material chosen, optimal results are yielded by the combination of patient expectations, physician judgment based on clinical experience, and injection technique.
1. Kontis TC, Rivkin A. The history of injectable facial fillers. Facial Plast Surg. 2009;25:67-72.
2. Ashinoff R. Overview: soft tissue augmentation. Clin Plast Surg. 2000;27:479-487.
3. Soft tissue fillers approved by the Center for Devices and Radiological Health. US Food and Drug Administration Web site. http://www.fda.gov/MedicalDevices/ProductsandMedicalProcedures/CosmeticDevices/WrinkleFillers/ucm227749.htm. Updated July 27, 2015. Accessed November 16, 2015.
4. Rohrich RJ, Ghavami A, Crosby MA. The role of hyaluronic acid fillers (restylane) in facial cosmetic surgery: review and technical considerations. Plast Reconstr Surg. 2007;120(suppl 6):S41-S54.
5. Braun M, Braun S. Nodule formation following lip augmentation using porcine collagen-derived filler. J Drugs Dermatol. 2008;7:579-581.
6. Lee JM, Kim YJ. Foreign body granulomas after the use of dermal fillers: pathophysiology, clinical appearance, histologic features, and treatment. Arch Plast Surg. 2015;42:232-239.
7. Kirby JS, Adgerson CN, Anderson BE. A survey of dermatology resident education in cosmetic procedures. J Am Acad Dermatol 2013;68:e23-e28.
Fillers are products that are injected into soft tissue and are classified as either resorbable or nonresorbable (permanent). Several dermal and subcutaneous fillers for soft tissue augmentation currently are available. This article provides a brief review of the history of filler agents currently available for soft tissue augmentation. Cadaveric-derived fillers and implants will not be discussed, as these materials are expensive, are not all approved by the US Food and Drug Administration (FDA), and are more commonly used in burn victims than in cosmetic patients.
History of Fillers in Dermatology
The first known injectable agent was paraffin, but its use as a dermal filler was abandoned after complications including embolization, migration (ie, movement into surrounding tissue), and granuloma formation were reported.1 Silicone also was banned by the FDA for use as a soft tissue filler due to similar complications. Bovine collagen was the first agent to be approved by the FDA for cosmetic injection, and since then many filler agents have revolutionized a new era of cosmetic enhancement.1 Injectable soft tissue fillers now can be classified based on several characteristics, including site of placement (ie, dermal or subcutaneous), animal versus nonanimal derivation (eg, autologous, xenograft, semisynthetic, synthetic), and duration of effects (temporary [<6 months], long-lasting [6 months– 2 years], semipermanent [2–5 years], permanent [>5 years]).2
In the 19th century, early instances of soft tissue augmentation using dermal fillers included autologous fat harvested from the arms for correction of depressed facial defects and scars in a patient with tuberculous osteitis as well as injection of paraffin into the scrotum as a testicular prosthesis in a patient with advanced tuberculosis.1 Later, a technique using a syringe to transfer autologous fat from the extremities was used for facial soft tissue augmentation and contouring, and permanent facial soft tissue augmentation using liquid silicone also was performed. In 1981, purified bovine dermal collagen was first approved by the FDA as a xenogenic agent for dermal injection.3
In 2003, the FDA began to approve new fillers for temporary soft tissue augmentation.3 Approval of injectable purified human collagen derived from fibroblasts was followed by another class of new agents: hyaluronic acid fillers.1 A biodegradable non–animal based stabilized hyaluronic acid was followed by more agents derived from rooster combs. Further investigations and research have continued, and more long-lasting synthetic fillers have become available, including calcium hydroxylapatite and poly-L-lactic acid. A renewed interest in permanent agents and longer lasting products such as silicone oil and polymethyl methacrylate also has emerged.4
The Future of Fillers
While earlier filler materials were limited, those used today are composed of a wide range of substances including collagen, hyaluronic acid, calcium hydroxylapatite, poly-L-lactic acid, and synthetic or manmade polymers (Table). The FDA has approved approximately 21 filler products for dermatologic indications, each with unique properties, advantages, and disadvantages.3 For example, there are several fillers that not only provide soft tissue augmentation but also stimulate collagen production. However, complications can occur, often many years after the initial treatment. Side effects such as swelling, erythema, and nodules may occur and in rare instances foreign body granulomas may develop and may be difficult to eradicate.5 Except for autologous fat, all fillers are foreign bodies and therefore can cause foreign body granulomatous reactions ranging from common (eg, with paraffin) to rare (eg, with hyaluronic acid) in occurrence.6 The clinical presentation of these reactions is variable, ranging from single to multiple nodules at the injection site to diffuse, hard swelling of the face accompanied by reddening of the skin.
With the increasing desire for a youthful appearance among the aging population, the pharmaceutical industry has responded by increasing the number of available treatment options to meet the demands of cosmetic patients, one of the fastest growing subpopulations in the field of dermatology. Fillers also have provided new options for patients who are unable to afford plastic surgery or those who are poor surgical candidates. The ideal filler material is nonallergenic, noncarcinogenic, and nonteratogenic. It should be stable, affordable, malleable, reversible, and durable with results that can be reproduced and should cause minimal inflammation, migration, and detectable changes. The ideal filler also should have predictable and consistent results, feel natural, take little time to administer, require minimal preparation, cause no patient downtime, and have a low risk for complications. Ideal administration should be painless, user-friendly, and conducted in an outpatient setting with minimal recovery and easy storage. Although no single filler is ideal for all patients, indications, and situations, residents should be aware of the properties and characteristics that make each product unique in order to optimize treatment in all patients.
As demands for cosmetic procedures increase, it is important to incorporate knowledge of cosmetic procedures (eg, fillers for soft tissue augmentation) in resident education and training. Although cosmetic dermatology has been featured prominently in dermatology residency, surveys have shown that residents desire more training in this area.7 Although lectures on soft tissue augmentation are popular topics in dermatology, hands-on experience performing these procedures varies widely depending on different training programs. My institution offers several lectures on cosmetic dermatology, and residents are able to perform procedures for soft tissue augmentation as the first assistant or first surgeon during our cosmetic clinic sessions twice weekly.
Final Thoughts
There are a variety of fillers on the horizon to improve aging and volume loss and the science behind cosmetic injections is evolving. Regardless of the filler material chosen, optimal results are yielded by the combination of patient expectations, physician judgment based on clinical experience, and injection technique.
Fillers are products that are injected into soft tissue and are classified as either resorbable or nonresorbable (permanent). Several dermal and subcutaneous fillers for soft tissue augmentation currently are available. This article provides a brief review of the history of filler agents currently available for soft tissue augmentation. Cadaveric-derived fillers and implants will not be discussed, as these materials are expensive, are not all approved by the US Food and Drug Administration (FDA), and are more commonly used in burn victims than in cosmetic patients.
History of Fillers in Dermatology
The first known injectable agent was paraffin, but its use as a dermal filler was abandoned after complications including embolization, migration (ie, movement into surrounding tissue), and granuloma formation were reported.1 Silicone also was banned by the FDA for use as a soft tissue filler due to similar complications. Bovine collagen was the first agent to be approved by the FDA for cosmetic injection, and since then many filler agents have revolutionized a new era of cosmetic enhancement.1 Injectable soft tissue fillers now can be classified based on several characteristics, including site of placement (ie, dermal or subcutaneous), animal versus nonanimal derivation (eg, autologous, xenograft, semisynthetic, synthetic), and duration of effects (temporary [<6 months], long-lasting [6 months– 2 years], semipermanent [2–5 years], permanent [>5 years]).2
In the 19th century, early instances of soft tissue augmentation using dermal fillers included autologous fat harvested from the arms for correction of depressed facial defects and scars in a patient with tuberculous osteitis as well as injection of paraffin into the scrotum as a testicular prosthesis in a patient with advanced tuberculosis.1 Later, a technique using a syringe to transfer autologous fat from the extremities was used for facial soft tissue augmentation and contouring, and permanent facial soft tissue augmentation using liquid silicone also was performed. In 1981, purified bovine dermal collagen was first approved by the FDA as a xenogenic agent for dermal injection.3
In 2003, the FDA began to approve new fillers for temporary soft tissue augmentation.3 Approval of injectable purified human collagen derived from fibroblasts was followed by another class of new agents: hyaluronic acid fillers.1 A biodegradable non–animal based stabilized hyaluronic acid was followed by more agents derived from rooster combs. Further investigations and research have continued, and more long-lasting synthetic fillers have become available, including calcium hydroxylapatite and poly-L-lactic acid. A renewed interest in permanent agents and longer lasting products such as silicone oil and polymethyl methacrylate also has emerged.4
The Future of Fillers
While earlier filler materials were limited, those used today are composed of a wide range of substances including collagen, hyaluronic acid, calcium hydroxylapatite, poly-L-lactic acid, and synthetic or manmade polymers (Table). The FDA has approved approximately 21 filler products for dermatologic indications, each with unique properties, advantages, and disadvantages.3 For example, there are several fillers that not only provide soft tissue augmentation but also stimulate collagen production. However, complications can occur, often many years after the initial treatment. Side effects such as swelling, erythema, and nodules may occur and in rare instances foreign body granulomas may develop and may be difficult to eradicate.5 Except for autologous fat, all fillers are foreign bodies and therefore can cause foreign body granulomatous reactions ranging from common (eg, with paraffin) to rare (eg, with hyaluronic acid) in occurrence.6 The clinical presentation of these reactions is variable, ranging from single to multiple nodules at the injection site to diffuse, hard swelling of the face accompanied by reddening of the skin.
With the increasing desire for a youthful appearance among the aging population, the pharmaceutical industry has responded by increasing the number of available treatment options to meet the demands of cosmetic patients, one of the fastest growing subpopulations in the field of dermatology. Fillers also have provided new options for patients who are unable to afford plastic surgery or those who are poor surgical candidates. The ideal filler material is nonallergenic, noncarcinogenic, and nonteratogenic. It should be stable, affordable, malleable, reversible, and durable with results that can be reproduced and should cause minimal inflammation, migration, and detectable changes. The ideal filler also should have predictable and consistent results, feel natural, take little time to administer, require minimal preparation, cause no patient downtime, and have a low risk for complications. Ideal administration should be painless, user-friendly, and conducted in an outpatient setting with minimal recovery and easy storage. Although no single filler is ideal for all patients, indications, and situations, residents should be aware of the properties and characteristics that make each product unique in order to optimize treatment in all patients.
As demands for cosmetic procedures increase, it is important to incorporate knowledge of cosmetic procedures (eg, fillers for soft tissue augmentation) in resident education and training. Although cosmetic dermatology has been featured prominently in dermatology residency, surveys have shown that residents desire more training in this area.7 Although lectures on soft tissue augmentation are popular topics in dermatology, hands-on experience performing these procedures varies widely depending on different training programs. My institution offers several lectures on cosmetic dermatology, and residents are able to perform procedures for soft tissue augmentation as the first assistant or first surgeon during our cosmetic clinic sessions twice weekly.
Final Thoughts
There are a variety of fillers on the horizon to improve aging and volume loss and the science behind cosmetic injections is evolving. Regardless of the filler material chosen, optimal results are yielded by the combination of patient expectations, physician judgment based on clinical experience, and injection technique.
1. Kontis TC, Rivkin A. The history of injectable facial fillers. Facial Plast Surg. 2009;25:67-72.
2. Ashinoff R. Overview: soft tissue augmentation. Clin Plast Surg. 2000;27:479-487.
3. Soft tissue fillers approved by the Center for Devices and Radiological Health. US Food and Drug Administration Web site. http://www.fda.gov/MedicalDevices/ProductsandMedicalProcedures/CosmeticDevices/WrinkleFillers/ucm227749.htm. Updated July 27, 2015. Accessed November 16, 2015.
4. Rohrich RJ, Ghavami A, Crosby MA. The role of hyaluronic acid fillers (restylane) in facial cosmetic surgery: review and technical considerations. Plast Reconstr Surg. 2007;120(suppl 6):S41-S54.
5. Braun M, Braun S. Nodule formation following lip augmentation using porcine collagen-derived filler. J Drugs Dermatol. 2008;7:579-581.
6. Lee JM, Kim YJ. Foreign body granulomas after the use of dermal fillers: pathophysiology, clinical appearance, histologic features, and treatment. Arch Plast Surg. 2015;42:232-239.
7. Kirby JS, Adgerson CN, Anderson BE. A survey of dermatology resident education in cosmetic procedures. J Am Acad Dermatol 2013;68:e23-e28.
1. Kontis TC, Rivkin A. The history of injectable facial fillers. Facial Plast Surg. 2009;25:67-72.
2. Ashinoff R. Overview: soft tissue augmentation. Clin Plast Surg. 2000;27:479-487.
3. Soft tissue fillers approved by the Center for Devices and Radiological Health. US Food and Drug Administration Web site. http://www.fda.gov/MedicalDevices/ProductsandMedicalProcedures/CosmeticDevices/WrinkleFillers/ucm227749.htm. Updated July 27, 2015. Accessed November 16, 2015.
4. Rohrich RJ, Ghavami A, Crosby MA. The role of hyaluronic acid fillers (restylane) in facial cosmetic surgery: review and technical considerations. Plast Reconstr Surg. 2007;120(suppl 6):S41-S54.
5. Braun M, Braun S. Nodule formation following lip augmentation using porcine collagen-derived filler. J Drugs Dermatol. 2008;7:579-581.
6. Lee JM, Kim YJ. Foreign body granulomas after the use of dermal fillers: pathophysiology, clinical appearance, histologic features, and treatment. Arch Plast Surg. 2015;42:232-239.
7. Kirby JS, Adgerson CN, Anderson BE. A survey of dermatology resident education in cosmetic procedures. J Am Acad Dermatol 2013;68:e23-e28.