User login
Aggressive approach to photodamaged skin is safe, effective, in small study
BOSTON – A more aggressive approach with fractional radiofrequency treatment of photodamaged facial skin in a single treatment achieved the same results as multiple treatments, with no increase in adverse events, in a small study.
“All of these nonablative fractional devices that are out there [are] generally dependent on multiple treatments; they’re fractional by their very nature, so you have to treat people three, five [or more] times,” Dr. David J. Goldberg, a dermatologist in group practice in New York and New Jersey, said in an interview at the annual meeting of the American Society for Laser Medicine and Surgery.
In the study of 25 individuals with photodamaged skin, “we took a somewhat more aggressive approach, and wanted to see what would happen after just one treatment,” Dr. Goldberg explained, adding that historically, single-treatment approaches with conventional fractional radiofrequency (RF) treatments cover only about 10% of photodamaged skin.
All patients were treated in his group practice at an energy setting of 60 mJ/pin using the Fractora RF device, a 60-pin bipolar RF device manufactured by InMode. Dr. Goldberg and his coinvestigators collected data on skin texture (or roughness), photodamage, wrinkling, tone, luminosity, and overall quality at baseline and 30, 60, 90, and 180 days after treatment.
All patients had their entire face treated during the single session, with downtime due to erythema lasting 3-4 days in all cases. By the end of day 4, all subjects were satisfied enough with the results that they were able to resume normal daily routines. Furthermore, all subjects showed signs of improvement after undergoing one treatment, and there were no adverse reactions, Dr. Goldberg said.
After a single treatment, skin tone improvement was evident in 40% of patients and improvements in texture, photodamage, and wrinkling were evident in 50% of subjects. For most patients, these improvements were seen between 60 and 90 days after treatment. However, some patients saw improvements in skin luminosity, texture, tone, and overall quality as early as the 30-day follow-up, according to Dr. Goldberg. Skin roughness improved by a “statistically significant” margin at 60- and 90-day follow-up, compared with baseline, and continued to improve through the 180-day follow-up.
“Patients don’t want to come in five times for treatment,” he said. “You can treat them safely and effectively with just one treatment by being just a little bit more aggressive.”
Dr. Goldberg disclosed speaking, consulting, and honoraria relationships with Cutera, as well as consulting and honoraria relationships with Cutera, InMode, Endymed and Aerolase.
BOSTON – A more aggressive approach with fractional radiofrequency treatment of photodamaged facial skin in a single treatment achieved the same results as multiple treatments, with no increase in adverse events, in a small study.
“All of these nonablative fractional devices that are out there [are] generally dependent on multiple treatments; they’re fractional by their very nature, so you have to treat people three, five [or more] times,” Dr. David J. Goldberg, a dermatologist in group practice in New York and New Jersey, said in an interview at the annual meeting of the American Society for Laser Medicine and Surgery.
In the study of 25 individuals with photodamaged skin, “we took a somewhat more aggressive approach, and wanted to see what would happen after just one treatment,” Dr. Goldberg explained, adding that historically, single-treatment approaches with conventional fractional radiofrequency (RF) treatments cover only about 10% of photodamaged skin.
All patients were treated in his group practice at an energy setting of 60 mJ/pin using the Fractora RF device, a 60-pin bipolar RF device manufactured by InMode. Dr. Goldberg and his coinvestigators collected data on skin texture (or roughness), photodamage, wrinkling, tone, luminosity, and overall quality at baseline and 30, 60, 90, and 180 days after treatment.
All patients had their entire face treated during the single session, with downtime due to erythema lasting 3-4 days in all cases. By the end of day 4, all subjects were satisfied enough with the results that they were able to resume normal daily routines. Furthermore, all subjects showed signs of improvement after undergoing one treatment, and there were no adverse reactions, Dr. Goldberg said.
After a single treatment, skin tone improvement was evident in 40% of patients and improvements in texture, photodamage, and wrinkling were evident in 50% of subjects. For most patients, these improvements were seen between 60 and 90 days after treatment. However, some patients saw improvements in skin luminosity, texture, tone, and overall quality as early as the 30-day follow-up, according to Dr. Goldberg. Skin roughness improved by a “statistically significant” margin at 60- and 90-day follow-up, compared with baseline, and continued to improve through the 180-day follow-up.
“Patients don’t want to come in five times for treatment,” he said. “You can treat them safely and effectively with just one treatment by being just a little bit more aggressive.”
Dr. Goldberg disclosed speaking, consulting, and honoraria relationships with Cutera, as well as consulting and honoraria relationships with Cutera, InMode, Endymed and Aerolase.
BOSTON – A more aggressive approach with fractional radiofrequency treatment of photodamaged facial skin in a single treatment achieved the same results as multiple treatments, with no increase in adverse events, in a small study.
“All of these nonablative fractional devices that are out there [are] generally dependent on multiple treatments; they’re fractional by their very nature, so you have to treat people three, five [or more] times,” Dr. David J. Goldberg, a dermatologist in group practice in New York and New Jersey, said in an interview at the annual meeting of the American Society for Laser Medicine and Surgery.
In the study of 25 individuals with photodamaged skin, “we took a somewhat more aggressive approach, and wanted to see what would happen after just one treatment,” Dr. Goldberg explained, adding that historically, single-treatment approaches with conventional fractional radiofrequency (RF) treatments cover only about 10% of photodamaged skin.
All patients were treated in his group practice at an energy setting of 60 mJ/pin using the Fractora RF device, a 60-pin bipolar RF device manufactured by InMode. Dr. Goldberg and his coinvestigators collected data on skin texture (or roughness), photodamage, wrinkling, tone, luminosity, and overall quality at baseline and 30, 60, 90, and 180 days after treatment.
All patients had their entire face treated during the single session, with downtime due to erythema lasting 3-4 days in all cases. By the end of day 4, all subjects were satisfied enough with the results that they were able to resume normal daily routines. Furthermore, all subjects showed signs of improvement after undergoing one treatment, and there were no adverse reactions, Dr. Goldberg said.
After a single treatment, skin tone improvement was evident in 40% of patients and improvements in texture, photodamage, and wrinkling were evident in 50% of subjects. For most patients, these improvements were seen between 60 and 90 days after treatment. However, some patients saw improvements in skin luminosity, texture, tone, and overall quality as early as the 30-day follow-up, according to Dr. Goldberg. Skin roughness improved by a “statistically significant” margin at 60- and 90-day follow-up, compared with baseline, and continued to improve through the 180-day follow-up.
“Patients don’t want to come in five times for treatment,” he said. “You can treat them safely and effectively with just one treatment by being just a little bit more aggressive.”
Dr. Goldberg disclosed speaking, consulting, and honoraria relationships with Cutera, as well as consulting and honoraria relationships with Cutera, InMode, Endymed and Aerolase.
AT LASER 2016
Key clinical point: More aggressive high-energy fractional radiofrequency treatment is safe enough on photodamaged facial skin that patients can avoid coming back for multiple treatments.
Major finding: 50% of patients showed improvement in skin texture, photodamage, and wrinkling at 60-, 90-, and 180-day follow-ups, and 40% showed improvement in skin tone after only one treatment.
Data source: A single-center study of high-energy laser treatment (60 mJ/pin) to repair photodamaged facial skin in 25 individuals.
Disclosures: Dr. Goldberg disclosed speaking, consulting, and honoraria relationships with Cutera, as well as consulting and honoraria relationships with Cutera, InMode, Endymed, and Aerolase.
Smooth hair – an acne-causing epidemic
Do you ask your acne patients about which hair products they use? This common question has recently brought our attention to popular hair products that are causing an acne epidemic. Have we forgotten about “Pomade acne”? Well, it’s making a comeback. Originally described in ethnic women, new frizz-fighting hair products have resurged and so has pomade acne in all skin types and in both men and women.
Smoothing serums, heat styling sprays, leave-in products popularly known as “It’s-a-10,” “Biosilk,” “anti-frizz serums,” “heat-protectants,” “thermal setting sprays,” and “shine sprays,” contain silicone-derived ingredients and oils to control frizz, add shine, and detangle the hair. They work by smoothing the hair cuticle, and for women with difficult-to-manage hair, they have become an essential part of the daily beauty regimen.
Men are not in the clear either. Hair waxes and pomades used to style men’s hair contain greasy wax-based ingredients that also clog pores, trap bacteria, and cause inflammatory breakouts.
As a general rule in skin and body care, most products work well for what they are made to do, but when misused, they can cause mishaps. You wouldn’t moisturize your face with your hair serum would you? It seems obvious that this could cause some skin issues; however, most people will not think to correlate their acne breakouts with their hair products until we mention it. These products rub off on the face or on the pillow at night. In addition, the less we wash our hair, the more we are going to bed and getting the daytime products all over our pillowcases. Our faces are rolling around in oily, waxy, hair products all night.
Makeup is known to cause acne, and some of the makeups that are well known culprits contain the same ingredients as in hair products. Foundations, primers, and popular “BB” creams often contain cyclopentasiloxane and dimethicone. They serve a similar purpose: smoothing the skin and smoothing the hair. Both should be avoided in acne-prone patients.
Common culprits in hair products include PVP/DMAPA acrylates, cyclopentasiloxane, panthenol, dimethicone, silicone, Quaternium-70, oils, and petrolatum.
The only way to eliminate acne caused by hair products is to completely eliminate the hair product from the daily routine. However, if your patients can’t live without their hair products, here are some tips to share with them to reduce breakouts:
• Choose a hairstyle that keeps the hair away from the face, or wear hair up to avoid prolonged contact with the face, particularly while sleeping.
• Change pillowcase often (every day if possible), especially for side sleepers. Regardless of the fabric, pillowcases trap oil, dirt, and bacteria.
• Shower at night and sleep with clean hair and clean skin.
• Style hair before applying makeup. Wash hands thoroughly to remove all hair products before touching the skin.
• Cover the face prior to applying any hair sprays.
• Cover the hair at bedtime; however, tight head coverings can stimulate sweat and cause scalp breakouts.
As a general rule, any patient with difficult-to-control acne, recalcitrant acne, or acne in areas on the cheeks or hairline should eliminate these hair products in their daily routine or avoid skin contact with these products.
References
1. J Clin Aesthet Dermatol. 2010 Apr;3(4):24-38.
2. Arch Dermatol. 1972;106 (6):843-50.
3. J Am Acad Dermatol. 2003; 48:S127-33.
4. Arch Dermatol. 1970;101(5):580-584.
5. “Cosmetics in Dermatology,” Second Edition, by Zoe Diana Draelos (New York: Churchill Livingstone, 1995).
Dr. Wesley and Dr. Talakoub are cocontributors to this column. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Talakoub.
Do you ask your acne patients about which hair products they use? This common question has recently brought our attention to popular hair products that are causing an acne epidemic. Have we forgotten about “Pomade acne”? Well, it’s making a comeback. Originally described in ethnic women, new frizz-fighting hair products have resurged and so has pomade acne in all skin types and in both men and women.
Smoothing serums, heat styling sprays, leave-in products popularly known as “It’s-a-10,” “Biosilk,” “anti-frizz serums,” “heat-protectants,” “thermal setting sprays,” and “shine sprays,” contain silicone-derived ingredients and oils to control frizz, add shine, and detangle the hair. They work by smoothing the hair cuticle, and for women with difficult-to-manage hair, they have become an essential part of the daily beauty regimen.
Men are not in the clear either. Hair waxes and pomades used to style men’s hair contain greasy wax-based ingredients that also clog pores, trap bacteria, and cause inflammatory breakouts.
As a general rule in skin and body care, most products work well for what they are made to do, but when misused, they can cause mishaps. You wouldn’t moisturize your face with your hair serum would you? It seems obvious that this could cause some skin issues; however, most people will not think to correlate their acne breakouts with their hair products until we mention it. These products rub off on the face or on the pillow at night. In addition, the less we wash our hair, the more we are going to bed and getting the daytime products all over our pillowcases. Our faces are rolling around in oily, waxy, hair products all night.
Makeup is known to cause acne, and some of the makeups that are well known culprits contain the same ingredients as in hair products. Foundations, primers, and popular “BB” creams often contain cyclopentasiloxane and dimethicone. They serve a similar purpose: smoothing the skin and smoothing the hair. Both should be avoided in acne-prone patients.
Common culprits in hair products include PVP/DMAPA acrylates, cyclopentasiloxane, panthenol, dimethicone, silicone, Quaternium-70, oils, and petrolatum.
The only way to eliminate acne caused by hair products is to completely eliminate the hair product from the daily routine. However, if your patients can’t live without their hair products, here are some tips to share with them to reduce breakouts:
• Choose a hairstyle that keeps the hair away from the face, or wear hair up to avoid prolonged contact with the face, particularly while sleeping.
• Change pillowcase often (every day if possible), especially for side sleepers. Regardless of the fabric, pillowcases trap oil, dirt, and bacteria.
• Shower at night and sleep with clean hair and clean skin.
• Style hair before applying makeup. Wash hands thoroughly to remove all hair products before touching the skin.
• Cover the face prior to applying any hair sprays.
• Cover the hair at bedtime; however, tight head coverings can stimulate sweat and cause scalp breakouts.
As a general rule, any patient with difficult-to-control acne, recalcitrant acne, or acne in areas on the cheeks or hairline should eliminate these hair products in their daily routine or avoid skin contact with these products.
References
1. J Clin Aesthet Dermatol. 2010 Apr;3(4):24-38.
2. Arch Dermatol. 1972;106 (6):843-50.
3. J Am Acad Dermatol. 2003; 48:S127-33.
4. Arch Dermatol. 1970;101(5):580-584.
5. “Cosmetics in Dermatology,” Second Edition, by Zoe Diana Draelos (New York: Churchill Livingstone, 1995).
Dr. Wesley and Dr. Talakoub are cocontributors to this column. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Talakoub.
Do you ask your acne patients about which hair products they use? This common question has recently brought our attention to popular hair products that are causing an acne epidemic. Have we forgotten about “Pomade acne”? Well, it’s making a comeback. Originally described in ethnic women, new frizz-fighting hair products have resurged and so has pomade acne in all skin types and in both men and women.
Smoothing serums, heat styling sprays, leave-in products popularly known as “It’s-a-10,” “Biosilk,” “anti-frizz serums,” “heat-protectants,” “thermal setting sprays,” and “shine sprays,” contain silicone-derived ingredients and oils to control frizz, add shine, and detangle the hair. They work by smoothing the hair cuticle, and for women with difficult-to-manage hair, they have become an essential part of the daily beauty regimen.
Men are not in the clear either. Hair waxes and pomades used to style men’s hair contain greasy wax-based ingredients that also clog pores, trap bacteria, and cause inflammatory breakouts.
As a general rule in skin and body care, most products work well for what they are made to do, but when misused, they can cause mishaps. You wouldn’t moisturize your face with your hair serum would you? It seems obvious that this could cause some skin issues; however, most people will not think to correlate their acne breakouts with their hair products until we mention it. These products rub off on the face or on the pillow at night. In addition, the less we wash our hair, the more we are going to bed and getting the daytime products all over our pillowcases. Our faces are rolling around in oily, waxy, hair products all night.
Makeup is known to cause acne, and some of the makeups that are well known culprits contain the same ingredients as in hair products. Foundations, primers, and popular “BB” creams often contain cyclopentasiloxane and dimethicone. They serve a similar purpose: smoothing the skin and smoothing the hair. Both should be avoided in acne-prone patients.
Common culprits in hair products include PVP/DMAPA acrylates, cyclopentasiloxane, panthenol, dimethicone, silicone, Quaternium-70, oils, and petrolatum.
The only way to eliminate acne caused by hair products is to completely eliminate the hair product from the daily routine. However, if your patients can’t live without their hair products, here are some tips to share with them to reduce breakouts:
• Choose a hairstyle that keeps the hair away from the face, or wear hair up to avoid prolonged contact with the face, particularly while sleeping.
• Change pillowcase often (every day if possible), especially for side sleepers. Regardless of the fabric, pillowcases trap oil, dirt, and bacteria.
• Shower at night and sleep with clean hair and clean skin.
• Style hair before applying makeup. Wash hands thoroughly to remove all hair products before touching the skin.
• Cover the face prior to applying any hair sprays.
• Cover the hair at bedtime; however, tight head coverings can stimulate sweat and cause scalp breakouts.
As a general rule, any patient with difficult-to-control acne, recalcitrant acne, or acne in areas on the cheeks or hairline should eliminate these hair products in their daily routine or avoid skin contact with these products.
References
1. J Clin Aesthet Dermatol. 2010 Apr;3(4):24-38.
2. Arch Dermatol. 1972;106 (6):843-50.
3. J Am Acad Dermatol. 2003; 48:S127-33.
4. Arch Dermatol. 1970;101(5):580-584.
5. “Cosmetics in Dermatology,” Second Edition, by Zoe Diana Draelos (New York: Churchill Livingstone, 1995).
Dr. Wesley and Dr. Talakoub are cocontributors to this column. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Talakoub.
OnabotulinumtoxinA most popular cosmetic procedure in 2015
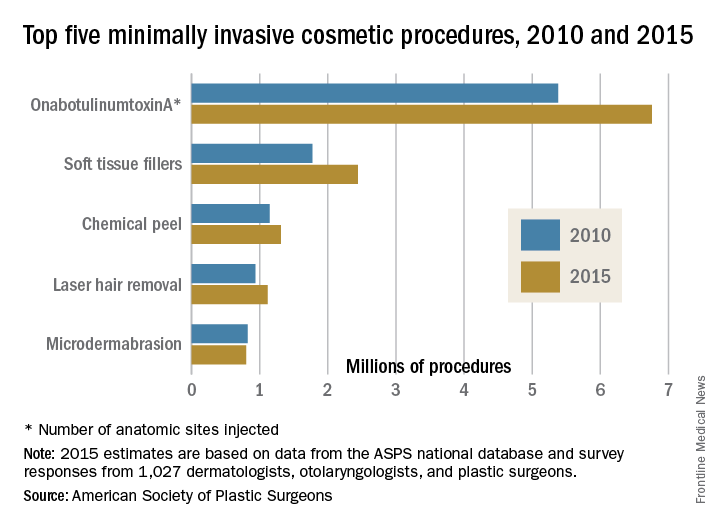
OnabotulinumtoxinA injection continued as the most popular minimally invasive cosmetic procedure in 2015, but its 1% increase in the number of procedures from the previous year was the smallest since 2009, when there was a 4% drop from the year before, according to the American Society of Plastic Surgeons.
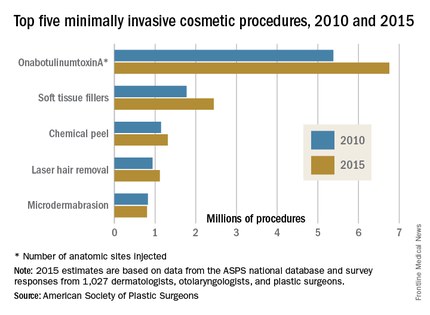
There was a total of 14.2 million minimally invasive cosmetic procedures performed in the United States in 2015, a 2% increase over 2014, the ASPS projected from a survey of 1,027 dermatologists, plastic surgeons, and otolaryngologists combined with its Tracking Operations and Outcomes for Plastic Surgeons database.
Of the 2015 total, an estimated 6.8 million were onabotulinumtoxinA procedures, with each injection of an anatomic site counting as a separate procedure. Soft tissue fillers were second in popularity among cosmetic patients, with over 2.4 million procedures performed, followed by chemical peel (1.3 million procedures), laser hair removal (1.1 million), and microdermabrasion (800,000), the ASPS reported.
Since 2010, soft tissue fillers have seen the most growth, increasing by 37.6%, followed by onabotulinumtoxinA (25.6%), laser hair removal (19.1%), and chemical peel (14.4%), according to ASPS data. The use of microdermabrasion dropped in that time period (–3%).
OnabotulinumtoxinA injection continued as the most popular minimally invasive cosmetic procedure in 2015, but its 1% increase in the number of procedures from the previous year was the smallest since 2009, when there was a 4% drop from the year before, according to the American Society of Plastic Surgeons.

There was a total of 14.2 million minimally invasive cosmetic procedures performed in the United States in 2015, a 2% increase over 2014, the ASPS projected from a survey of 1,027 dermatologists, plastic surgeons, and otolaryngologists combined with its Tracking Operations and Outcomes for Plastic Surgeons database.
Of the 2015 total, an estimated 6.8 million were onabotulinumtoxinA procedures, with each injection of an anatomic site counting as a separate procedure. Soft tissue fillers were second in popularity among cosmetic patients, with over 2.4 million procedures performed, followed by chemical peel (1.3 million procedures), laser hair removal (1.1 million), and microdermabrasion (800,000), the ASPS reported.
Since 2010, soft tissue fillers have seen the most growth, increasing by 37.6%, followed by onabotulinumtoxinA (25.6%), laser hair removal (19.1%), and chemical peel (14.4%), according to ASPS data. The use of microdermabrasion dropped in that time period (–3%).
OnabotulinumtoxinA injection continued as the most popular minimally invasive cosmetic procedure in 2015, but its 1% increase in the number of procedures from the previous year was the smallest since 2009, when there was a 4% drop from the year before, according to the American Society of Plastic Surgeons.

There was a total of 14.2 million minimally invasive cosmetic procedures performed in the United States in 2015, a 2% increase over 2014, the ASPS projected from a survey of 1,027 dermatologists, plastic surgeons, and otolaryngologists combined with its Tracking Operations and Outcomes for Plastic Surgeons database.
Of the 2015 total, an estimated 6.8 million were onabotulinumtoxinA procedures, with each injection of an anatomic site counting as a separate procedure. Soft tissue fillers were second in popularity among cosmetic patients, with over 2.4 million procedures performed, followed by chemical peel (1.3 million procedures), laser hair removal (1.1 million), and microdermabrasion (800,000), the ASPS reported.
Since 2010, soft tissue fillers have seen the most growth, increasing by 37.6%, followed by onabotulinumtoxinA (25.6%), laser hair removal (19.1%), and chemical peel (14.4%), according to ASPS data. The use of microdermabrasion dropped in that time period (–3%).
For BCC, consider curettage only
WAIKOLOA, HAWAII – Curettage alone for the treatment of basal cell carcinomas offers several distinct advantages over the widely accepted technique of curettage followed by electrodesiccation, Dr. David L. Swanson said at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
When he asked for an audience show of hands as to who treats BCCs using curettage without electrodesiccation, the packed auditorium full of dermatologists remained hands-down. But there is good evidence that curettage alone provides cure rates similar to published rates for curettage and electrodesiccation, along with improved cosmesis, faster healing, and fewer problems with scarring and hypopigmentation, according to Dr. Swanson, a dermatologist at Mayo Clinic Arizona, Scottsdale.
He cited a persuasive study by other dermatologists at Mayo Clinic Arizona (J Am Acad Dermatol. 2006 Jun;54[6]:1039-45).
“This article was transformative for my practice,” said Dr. Swanson, who wasn’t involved in the study.
The investigators reviewed the records of 302 biopsy-proven BCCs in 136 patients. All were amenable to curettage and electrodesiccation but were instead treated by curettage alone during 1993-1998. The BCCs lacked histopathologic high-risk features; most were superficial or nodular lesions. Nearly one-quarter of the BCCs, however, were located on high-risk anatomic sites.
The 5-year cure rate was 96%. The authors’ review of 10 published series of BCCs treated using curettage and electrodesiccation showed recurrence rates mostly in the 3%-7% range.
Among the advantages cited by the investigators of going with curettage alone were its simplicity, reduced equipment requirement, cost effectiveness, lack of electrical interference with implantable cardiac devices, the avoidance of potentially hazardous viral smoke plumes, and reduced rates of hypopigmentation and scarring.
This was a retrospective study. Its other significant limitation was that all 302 BCCs were treated by the same dermatologist: Dr. Mark J. Zalla, who Dr. Swanson considers to be a dermatologic surgeon of exceptional talent.
“I’m not Mark Zalla. Nobody else in this room is Mark Zalla, so we can’t necessarily extrapolate from this study what your outcomes are going to be. That said, I’ve adopted this technique, and most of my colleagues do not do electrodesiccation anymore. I take off several basal cell carcinomas per day with curettage, and it’s my sense that I don’t have higher recurrence rates and that it does heal better with less scarring,” Dr. Swanson said.
He reported having no financial conflicts of interest regarding his presentation. SDEF and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – Curettage alone for the treatment of basal cell carcinomas offers several distinct advantages over the widely accepted technique of curettage followed by electrodesiccation, Dr. David L. Swanson said at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
When he asked for an audience show of hands as to who treats BCCs using curettage without electrodesiccation, the packed auditorium full of dermatologists remained hands-down. But there is good evidence that curettage alone provides cure rates similar to published rates for curettage and electrodesiccation, along with improved cosmesis, faster healing, and fewer problems with scarring and hypopigmentation, according to Dr. Swanson, a dermatologist at Mayo Clinic Arizona, Scottsdale.
He cited a persuasive study by other dermatologists at Mayo Clinic Arizona (J Am Acad Dermatol. 2006 Jun;54[6]:1039-45).
“This article was transformative for my practice,” said Dr. Swanson, who wasn’t involved in the study.
The investigators reviewed the records of 302 biopsy-proven BCCs in 136 patients. All were amenable to curettage and electrodesiccation but were instead treated by curettage alone during 1993-1998. The BCCs lacked histopathologic high-risk features; most were superficial or nodular lesions. Nearly one-quarter of the BCCs, however, were located on high-risk anatomic sites.
The 5-year cure rate was 96%. The authors’ review of 10 published series of BCCs treated using curettage and electrodesiccation showed recurrence rates mostly in the 3%-7% range.
Among the advantages cited by the investigators of going with curettage alone were its simplicity, reduced equipment requirement, cost effectiveness, lack of electrical interference with implantable cardiac devices, the avoidance of potentially hazardous viral smoke plumes, and reduced rates of hypopigmentation and scarring.
This was a retrospective study. Its other significant limitation was that all 302 BCCs were treated by the same dermatologist: Dr. Mark J. Zalla, who Dr. Swanson considers to be a dermatologic surgeon of exceptional talent.
“I’m not Mark Zalla. Nobody else in this room is Mark Zalla, so we can’t necessarily extrapolate from this study what your outcomes are going to be. That said, I’ve adopted this technique, and most of my colleagues do not do electrodesiccation anymore. I take off several basal cell carcinomas per day with curettage, and it’s my sense that I don’t have higher recurrence rates and that it does heal better with less scarring,” Dr. Swanson said.
He reported having no financial conflicts of interest regarding his presentation. SDEF and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – Curettage alone for the treatment of basal cell carcinomas offers several distinct advantages over the widely accepted technique of curettage followed by electrodesiccation, Dr. David L. Swanson said at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
When he asked for an audience show of hands as to who treats BCCs using curettage without electrodesiccation, the packed auditorium full of dermatologists remained hands-down. But there is good evidence that curettage alone provides cure rates similar to published rates for curettage and electrodesiccation, along with improved cosmesis, faster healing, and fewer problems with scarring and hypopigmentation, according to Dr. Swanson, a dermatologist at Mayo Clinic Arizona, Scottsdale.
He cited a persuasive study by other dermatologists at Mayo Clinic Arizona (J Am Acad Dermatol. 2006 Jun;54[6]:1039-45).
“This article was transformative for my practice,” said Dr. Swanson, who wasn’t involved in the study.
The investigators reviewed the records of 302 biopsy-proven BCCs in 136 patients. All were amenable to curettage and electrodesiccation but were instead treated by curettage alone during 1993-1998. The BCCs lacked histopathologic high-risk features; most were superficial or nodular lesions. Nearly one-quarter of the BCCs, however, were located on high-risk anatomic sites.
The 5-year cure rate was 96%. The authors’ review of 10 published series of BCCs treated using curettage and electrodesiccation showed recurrence rates mostly in the 3%-7% range.
Among the advantages cited by the investigators of going with curettage alone were its simplicity, reduced equipment requirement, cost effectiveness, lack of electrical interference with implantable cardiac devices, the avoidance of potentially hazardous viral smoke plumes, and reduced rates of hypopigmentation and scarring.
This was a retrospective study. Its other significant limitation was that all 302 BCCs were treated by the same dermatologist: Dr. Mark J. Zalla, who Dr. Swanson considers to be a dermatologic surgeon of exceptional talent.
“I’m not Mark Zalla. Nobody else in this room is Mark Zalla, so we can’t necessarily extrapolate from this study what your outcomes are going to be. That said, I’ve adopted this technique, and most of my colleagues do not do electrodesiccation anymore. I take off several basal cell carcinomas per day with curettage, and it’s my sense that I don’t have higher recurrence rates and that it does heal better with less scarring,” Dr. Swanson said.
He reported having no financial conflicts of interest regarding his presentation. SDEF and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF HAWAII DERMATOLOGY SEMINAR
Merkel Cell Carcinoma: A Review
Merkel cells originally were described by German histopathologist Friedrich Sigmund Merkel in 1875. These unique tactile cells were described as epidermal, nondendritic, and nonkeratinizing. Merkel cells are thought to arise from the neural crest and are believed to be primary neural cells found within the basal layer of the epidermis.1,2 They likely function primarily as slowly adapting type I mechanoreceptors. Origin from the neural crest is controversial, as other investigators have suggested derivation from epidermal keratinocytes.1,2 Tumor cells have been linked to the amine precursor uptake and decarboxylation system.3 In 1972, Toker4 described several cases of trabecular or sweat gland carcinomas of the skin. Upon further investigation, the cells that comprised these tumors were found to have dense core granules on electron microscopy, typical of Merkel cells.1,2 Other terms such as neuroendocrine carcinoma of the skin, small cell carcinoma of the skin, and anaplastic carcinoma of the skin have been used to describe Merkel cell carcinoma (MCC),1 which was suggested by De Wolf-Peeters et al5 in 1980.
Despite being a rare malignancy, MCC follows an aggressive clinical course. Upon presentation, approximately 66% of patients have local disease, 27% have nodal involvement, and 7% have distant metastasis.1 Future treatments will likely center around the novel Merkel cell polyomavirus (MCPyV) and modification of immune responses toward tumor cells. Standardization continues to be lacking in both staging and treatment of this aggressive tumor.
Epidemiology of MCC

| ||
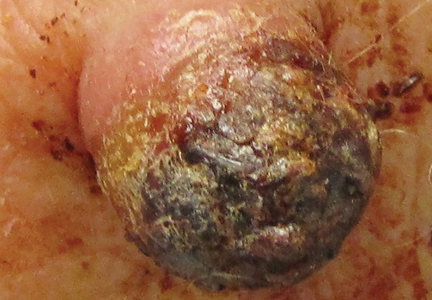
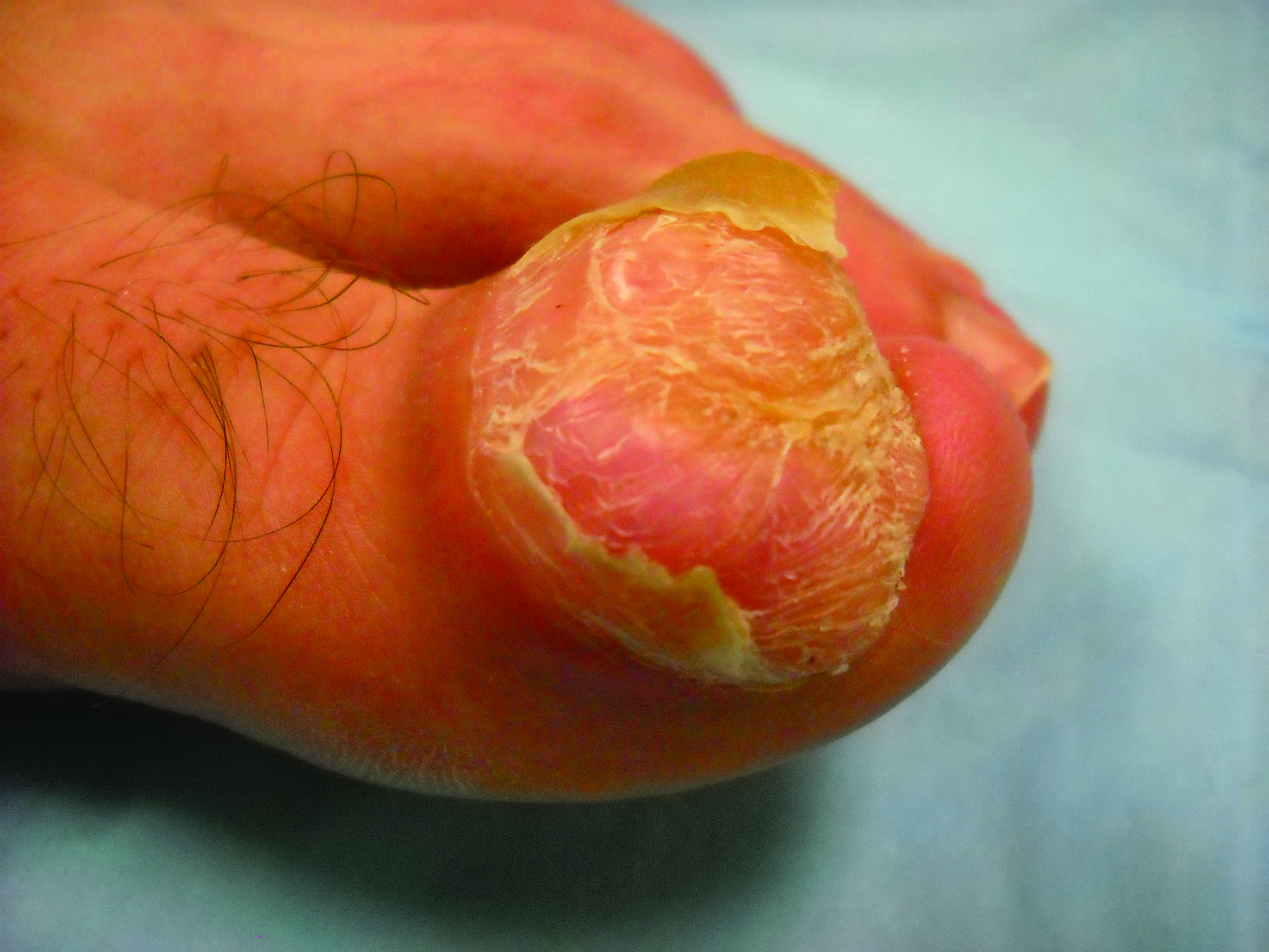
Figure 1. A 2.3×1.5×1.2-cm, hemorrhagic, crusted, | ||
| ||
| ||
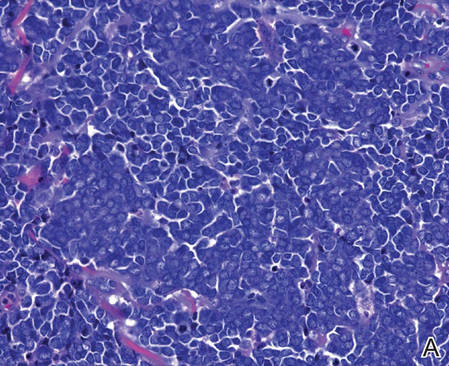
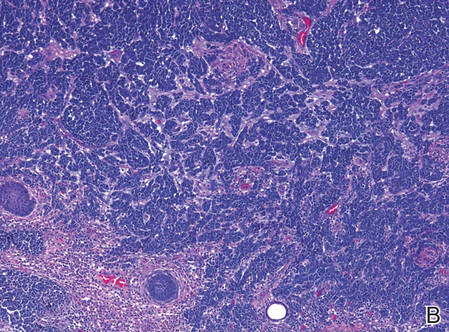
| Figure 2. Merkel cells are small- to medium-sized cells with round nuclei and scant cytoplasm. Granular or stippled chromatin can be seen (A)(H&E, original magnification ×40). Merkel cell carcinoma with trabecular pattern (B) (H&E, original magnification ×10). | ||
Between 1986 and 2006, the incidence of MCC grew substantially.1,2 Figures have been reported at 0.15 cases per 100,000 individuals to 0.6 cases per 100,000 individuals worldwide. In the United States, the age-adjusted incidence of MCC is estimated at 0.24 per 100,000 person-years, which is higher than the estimated 0.13 per 100,000 person-years found in Europe.3 The highest incidence worldwide has been noted in Western Australia, likely due to high levels of UV exposure.1 The incidence of MCC in psoriasis patients who are treated with oral methoxsalen (psoralen) and UVA photochemotherapy is 100 times greater than in the general population, further supporting the role of UV light in the development of MCC.1 White individuals have the highest incidence of MCC worldwide, with men being affected more frequently than women.1,3 The majority of patients with MCC are diagnosed at 70 years or older.1 Approximately 5% of reported MCC patients are diagnosed before 50 years of age.2 Immunosuppression and immunodeficiency likely play a role in the pathogenesis of MCC, and the incidence is increased in solid organ transplant recipients, most commonly renal transplant recipients,1 as well as individuals with chronic lymphocytic leukemia, human immunodeficiency virus infection, and AIDS.1,3 Patients with autoimmune diseases such as rheumatoid arthritis also are at increased risk for MCC.3 Individuals who are diagnosed with MCC are at an increased risk for development of other malignancies including nonmelanoma skin cancers, chronic lymphocytic leukemia, Hodgkin lymphoma, and non-Hodgkin lymphoma.3
Clinical Presentation of MCC
The clinical presentation of MCC can be variable. Most tumors present as firm, red to purple, nontender papules or nodules (Figure 1).1 Tumor size may range from 2 to 200 mm but is most commonly less than 20 mm.2 Growth can be rapid, and tumors are most commonly located on sun-exposed skin. The head and neck areas account for 48% of all MCC cases,1 with the eyelids being frequently involved.2 Merkel cell carcinoma also has been reported on the arms, legs, trunk, back, and buttocks.1 Non–sun-exposed areas are less commonly affected. Mucosal sites (eg, larynx, nasal cavity, pharynx, mouth) account for 5% of primary MCCs.1 Merkel cell carcinoma also has been reported to affect the vulva and penis. Subcutaneous primary MCC has presented without overlying epidermal changes.1 In a case series by Heath et al,6 14% (27/195) of MCC patients presented with nodal disease without any identifiable primary tumor, with the inguinal nodal chain being the most common for this presentation. It currently is not known whether these nodal tumors are primary tumors or metastatic disease with a regression of the primary tumor.1
|
Histopathology of MCC
| ||
| ||
| ||
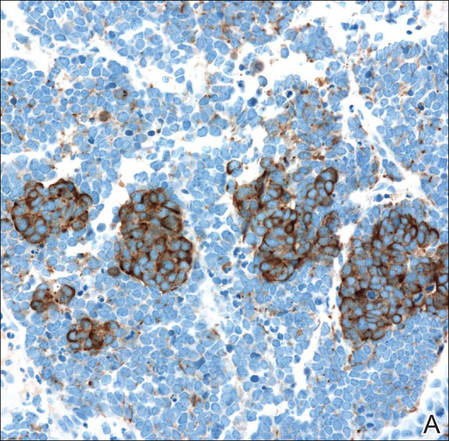
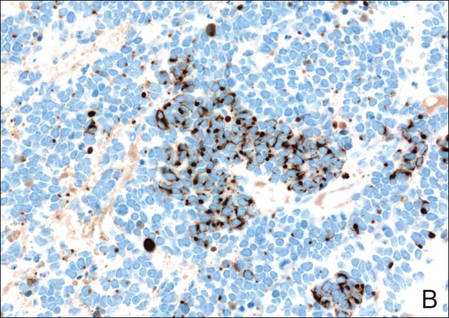
Figure 3. Positive chromogranin staining (A)(original | ||
Merkel cells are small- to medium-sized basophilic cells with round nuclei and scant cytoplasm. Granular or stippled chromatin can be seen on histopathology (Figure 2A).1 Some tumor cells have more vesicular chromatin, multiple small nucleoli, irregular contours, and more abundant cytoplasm. In some reports, irregular contours and abundant cytoplasm were associated with no detectible MCPyV infection.1,3 Merkel cell carcinomas have a primarily nodular architecture, and classification is based on growth pattern and cell size. Three histopathologic growth patterns have been described: nodular, infiltrative, and trabecular. The trabecular pattern is composed of interconnecting strands ofcells (Figure 2B). Tumors with solely intraepidermal involvement (MCC in situ) have been described but are exceedingly rare.1 Cell types are classified according to size, with the intermediate cell type being the most common. The small cell variant may be mistaken for a lymphocytic infiltrate due to the similar size and appearance of both types of cells.1,3
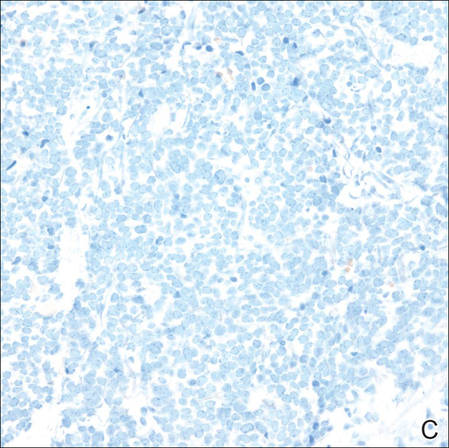
Merkel cell carcinomas can have histopathological overlap with lymphomas, small cell lung cancers, carcinoid tumors, primitive neuroectodermal tumors, neuroblastomas, small cell osteosarcomas, rhabdomyosarcomas, or Ewing sarcomas.1,3 Specifically, differentiation from small cell carcinoma of the lung is of utmost importance. Merkel cell carcinoma stains positively for cytokeratins 8, 18, 19, and 20. The neuroendocrine markers chromogranin (Figure 3A), synaptophysin, and neuron-specific enolase also may show positive staining. Cytokeratin 20, low-molecular-weight cytokeratins (CAM 5.2), and neurofilament immunostains have a high sensitivity for MCC and are the most frequently used.1 Cytokeratin 20 stains in the characteristic paranuclear dot–like pattern, which is a hallmark of MCC (Figure 3B). Cytokeratin 20 positivity in conjunction with negative staining for thyroid transcription factor 1 (Figure 3C) and cytokeratin 7 can aid in differentiation from small cell carcinoma of the lung.1,3
Pathogenesis of MCC
In 2008, Feng et al7 discovered a novel polyomavirus associated with the development of MCC. This novel polyomavirus, MCPyV, is found in approximately 80% of all cases of MCC. Seventeen members of the polyomavirus family have been identified, 9 of which have been found to infect humans, including BK virus, JC virus, WU, MCPyV, human polyomavirus 6, human polyomavirus 7, trichodysplasia spinulosa–associated polyomavirus, human polyomavirus 9, and Simian virus 40.1 Merkel cell polyomavirus infection is found in approximately 60% of the general population and exposure likely occurs early in life. The virus likely is transmitted through skin shedding and nasal secretions, though it also has been found in urine specimens.3 Currently, there is no evidence to suggest vertical viral transmission from mother to fetus.
Merkel cell polyomavirus is composed of early and late gene regions. The early gene region contains both large T antigen (LT) and small T antigen reading frames, which are necessary for viral replication.8 The late region is responsible for encoding viral proteins necessary for viral capsid assembly. Mutations found in viral protein 1 prevent formation of viral particles.9 Large T antigen is substantially overexpressed in MCC and is responsible for tumor suppression through retinoblastoma tumor suppressor protein. It also serves as a binding domain for both heat shock proteins and helicases.8,10 These domains allow the polyomaviruses to use host-cell machinery for viral genome replication while targeting tumor suppressor proteins.8 Upon viral integration into host DNA, viral replication ceases while oncogenic function persists.
The exact mechanism by which the MCPyV contributes to the development of MCC still has yet to be identified. Hypotheses suggest a combination of viral infection with external mutagens (eg, UV radiation). Experimental observations suggest viral contribution is likely due to the large percentage of MCCs that are positive for MCPyV, the identification of LT antigen expression and the role it plays in preserving cell cycle progression, and the role persistent LT antigen expression plays in continued growth of MCC cell lines in vitro.8 Two important cell line preservation mechanisms ensure continued tumor growth, including prevention of apoptosis triggered by DNA damage response mechanisms following UV damage and interaction with the retinoblastoma tumor suppressor protein allowing continued growth.8,11 Other important factors in tumor growth and survival may be the inhibition of apoptosis through the BCL2 (B-cell chronic lymphocytic leukemia/lymphoma 2) proto-oncogene and survivin (baculoviral inhibitor of apoptosis repeat-containing 5 [BIRC5]).12 Survivin has been found to play an important role in MCPyV-positive MCCs.12,13 It has been suggested that lymphangiogenesis in MCC likely is driven by vascular endothelial growth factor-C+CD68+CD163+ M2 macrophages.14 Another survival mechanism specific to polyomaviruses is their ability to interfere with the p53 tumor suppressor pathway.8 Loss of p53 expression by tumor cell nuclei has been associated with poor prognosis.15
Immune Response
Immune response as a role in tumor progression can be primarily centered on the concept of persistent antigen expression as a means of immune downregulation. Dunn et al16 suggested that cancer cells must interact through 3 consecutive phases with the host immune system (immunoediting hypothesis). In the elimination phase, the host immune system is able to recognize and destroy newly transformed cells through both the innate and adaptive immune systems. The second equilibrium phase allows the tumor to remain dormant and growth remains stagnant. Lastly, the tumor is allowed to evade the immune system through the escape phase.8
Host immune responses play an important role in both the progression and prognosis of MCC. High anti-MCPyV capsid antibody titers have been associated with better progression-free survival in some patients.8 Patients with high antibody titers (>10,000) likely have better progression-free survival than those with low antibody titers (<10,000).17 Antibody titers to the LT antigen may serve as a biomarker of MCC disease burden in the future. Rising LT antigen titers have been shown to correlate with disease progression and falling titers correlate with successful treatment.8 Tumoral infiltration of CD8+ T lymphocytes has been shown to be a predictor of survival compared to no intratumoral infiltration.6 Sihto et al18 suggested that this better prognosis from high intratumoral infiltration is not specific to MCPyV-positive MCC; however, it does highlight an important aspect of tumor evasion through the downregulation of cell surface expression of class I major histocompatibility complex antigens, which allows presentation of tumor intracellular peptides to CD8+ T lymphocytes.8 Upregulation of this specific immune response may play a role in the future treatment of MCC.
Staging and Prognosis
Due to the extremely aggressive nature of MCC, patients with local disease and tumors 2 cm or smaller in diameter have a 66% survival at 5 years.1,3 The 5-year survival rate for patients with local metastasis to regional lymph nodes ranges from 26% to 42%. Patients with distant metastasis have an 18% survival rate at 5 years.1,3 Data suggest that sentinel lymph node biopsy should be performed on all patients with MCC regardless of tumor size.1 There are no consensus guidelines to date regarding imaging for the staging of MCC patients. It is suggested that (18F)fluorodeoxyglucose positron emission tomography alone or in combination with computed tomography (CT) may be of value as a single whole-body diagnostic tool for accurate staging.10 It also has been suggested that (18F)fluorodeoxyglucose positron emission tomography and CT may offer more accurate staging than other screening modalities such as CT alone or magnetic resonance imaging.14,19
Treatment of MCC
Surgery remains the mainstay of treatment of MCC. Current National Comprehensive Cancer Network guidelines20 recommend 1- to 2-cm margins for wide local excision or treatment with Mohs micrographic surgery. Sentinel lymph node biopsy should be performed intraoperatively in patients undergoing wide local excision and preoperatively for patients undergoing Mohs micrographic surgery due to potential alterations in lymphatic drainage that may affect lymphoscintigraphy.1
Radiation may be used as primary or adjuvant therapy in patients with MCC. Radiation as primary therapy generally is reserved for patients who are not surgical candidates. It has been suggested that there was no difference in outcome in a small group of patients treated with radiation alone compared to patients who underwent surgery and radiation to the tumor bed.1 Current guidelines suggest a small group of patients may not require adjuvant therapy following adequate resection of some small tumors, and clinical observation may be appropriate.1,3 Chemotherapy may play a palliative role in patients with metastatic MCC. Merkel cell carcinoma has been shown to be chemosensitive but with a high recurrence rate.1 Because the immune system plays an important role in disease prognosis, having an intact immune system likely is paramount in the prevention of further disease progression.
Future Treatments of MCC
Future treatment of MCC may be focused on the viral etiology of most tumors and upregulation of the immune response, which may lead to the possibility of specifically interfering with virus-specific oncoproteins and stimulation of immune responses to virally infected tumor cells.8 The MCPyV large T antigen has been found to be overexpressed in some tumors and may serve as a specific target of therapy.10,21 Survivin, a key cell cycle protein encoded by LT antigen, may be an interesting target given its implication in other cancers.13 Other potential nonviral molecular target antigens include the oncoprotein H1P1 that interacts with c-KIT.8 Specific immunostimulatory cytokines that may be used to upregulate the immune response to tumoral cells may include IL-2, IL-12, IL-15, or IL-21. Therapeutic agents that may be studied in the future to target the immune exhaustion phenomenon associated with tumorigenesis include ipilimumab (cytotoxic T lymphocyte antigen 4 receptor-blocking agent) as well as programmed cell death 1 and programmed cell death 1 ligand 1 (PD-1/PD-L1).8 Neuroendocrine tumors including MCC tend to be highly vascular and express vascular endothelial growth factors and platelet-derived growth factors, which may be other potential therapeutic targets. It has been reported that approximately 95% of MCC patients have CD56+ tumors, and current clinical trials suggest a promising therapeutic response with the immunogen anti-CD56 monoclonal antibody.3
Conclusion
Merkel cell carcinoma is a rare aggressive neuroendocrine tumor that has been associated with a novel polyomavirus. Merkel cell carcinoma tends to affect elderly and immunocompromised patients as well as white individuals. Tumors are most often found in areas of high UV exposure and clinically on sun-exposed skin. Merkel cell polyomavirus is associated with approximately 80% of tumors, and tumorigenesis likely is caused by a number of sequential steps from viral integration into host DNA, mutagenic events, and specific immune responses. Currently there are no consensus guidelines for using imaging for staging of MCCs, but sentinel lymph node biopsy is recommended for all cases due to the aggressive nature of even smaller tumors. Surgery remains the mainstay of treatment, and radiation therapy may be used as a primary or adjuvant treatment. Chemotherapy usually is reserved for patients with metastatic disease purely for palliation. Future treatments of MCC likely will center on the viral etiology of MCC and upregulation of immune responses to virally infected tumor cells.
1. Han S, North J, Canavan T, et al. Merkel cell carcinoma. Hematol Oncol Clin N Am. 2012;26:1351-1374.
2. Goessling W, McKee P, Mayer R. Merkel cell carcinoma. J Clin Oncol. 2002;20:588-598.
3. Donepudi S, DeConti R, Samlowski W. Recent advances in the understanding of the genetics, etiology, and treatment of merkel cell carcinoma. Semin Oncol. 2012;39:163-172.
4. Toker C. Trabecular carcinoma of the skin. Arch Dermatol. 1972;105:107-110.
5. De Wolff-Peeters C, Marien K, Mebis J, et al. A cutaneous APUDoma or Merkel cell tumor? a morphologically recognizable tumor with a biological and histological malignant aspect in contrast with its clinical behavior. Cancer. 1980;46:810-816.
6. Heath M, Jaimes N, Lemos B, et al. Clinical characteristics of Merkel cell carcinoma at diagnosis in 195 patients: the AEIOU features. J Am Acad Dermatol. 2008;58:375-381.
7. Feng H, Shuda M, Chang Y, et al. Clonal integration of a polyomavirus in human Merkel cell carcinoma. Science. 2008;319:1096-1100.
8. Bhatia S, Afanasiev O, Nghiem P. Immunobiology of Merkel cell carcinoma: implications for immunotherapy of a polyomavirus-associated cancer. Curr Oncol Rep. 2011;13:488-497.
9. Amber K, McLeod M, Nouri K. The Merkel cell polyomavirus and its involvement in Merkel cell carcinoma. Dermatol Surg. 2013;39:232-238.
10. Erovic B, Al Habeeb A, Harris L, et al. Significant overexpression of the Merkel cell polyomavirus (MCPyV) large T antigen in Merkel cell carcinoma. Head Neck. 2013;35:184-189.
11. Demetriou S, Ona-Vu K, Sullivan E, et al. Defective DNA repair and cell cycle arrest in cells expressing Merkel cell polyomavirus T antigen. Int J Cancer. 2012;131:1818-1827.
12. Sahi H, Koljonen V, Kavola H, et al. Bcl-2 expression indicates better prognosis of Merkel cell carcinoma regardless of the presence of Merkel cell polyomavirus. Virchows Arch. 2012;461:553-559.
13. Arora R, Shuda M, Guastafierro A, et al. Survivin is a therapeutic target in Merkel cell carcinoma. Sci Transl Med. 2012;4:1-11.
14. Hawryluk E, O’Regan K, Sheehy N, et al. Positron emission tomography/computed tomography imaging in Merkel cell carcinoma: a study of 270 scans in 97 patients at the Dana-Farber/Brigham and Women’s Cancer Center. J Am Acad Dermatol. 2013;68:592-599.
15. Hall B, Pincus L, Yu S, et al. Immunohistochemical prognostication of Merkel cell carcinoma: p63 expression but not polyomavirus status correlates with outcome. J Cutan Pathol. 2012;39:911-917.
16. Dunn GP, Bruce AT, Ikeda H, et al. Cancer immunoediting: from immunosurveillance to tumor escape. Nat Immunol. 2002;3:991-998.
17. Touze A, Le Bidre E, Laude H, et al. High levels of antibodies against Merkel cell polyomavirus identify a subset of patients with Merkel cell carcinoma with better clinical outcome. J Clin Oncol. 2011;29:1612-1619.
18. Sihto H, Bohling T, Kavola H, et al. Tumor infiltrating immune cells and outcome of Merkel cell carcinoma: a population-based study. Clin Cancer Res. 2012;18:2872-2881.
19. Colgan M, Tarantola T, Weaver A, et al. The predictive value of imaging studies in evaluating regional lymph node involvement in Merkel cell carcinoma. J Am Acad Dermatol. 2012;67:1250-1256.
20. NCCN Clinical Practice Guidelines in Oncology. National Comprehensive Cancer Network website. http://www.nccn.org/professionals/physician_gls/f_guidelines.asp#site. Accessed March 22, 2016.
21. Angermeyer S, Hesbacher S, Becker J, et al. Merkel cell polyomavirus-positive Merkel cell carcinoma cells do not require expression of the viral small T antigen. J Invest Dermatol. 2013;133:1-6.
Merkel cells originally were described by German histopathologist Friedrich Sigmund Merkel in 1875. These unique tactile cells were described as epidermal, nondendritic, and nonkeratinizing. Merkel cells are thought to arise from the neural crest and are believed to be primary neural cells found within the basal layer of the epidermis.1,2 They likely function primarily as slowly adapting type I mechanoreceptors. Origin from the neural crest is controversial, as other investigators have suggested derivation from epidermal keratinocytes.1,2 Tumor cells have been linked to the amine precursor uptake and decarboxylation system.3 In 1972, Toker4 described several cases of trabecular or sweat gland carcinomas of the skin. Upon further investigation, the cells that comprised these tumors were found to have dense core granules on electron microscopy, typical of Merkel cells.1,2 Other terms such as neuroendocrine carcinoma of the skin, small cell carcinoma of the skin, and anaplastic carcinoma of the skin have been used to describe Merkel cell carcinoma (MCC),1 which was suggested by De Wolf-Peeters et al5 in 1980.
Despite being a rare malignancy, MCC follows an aggressive clinical course. Upon presentation, approximately 66% of patients have local disease, 27% have nodal involvement, and 7% have distant metastasis.1 Future treatments will likely center around the novel Merkel cell polyomavirus (MCPyV) and modification of immune responses toward tumor cells. Standardization continues to be lacking in both staging and treatment of this aggressive tumor.
Epidemiology of MCC

| ||
Figure 1. A 2.3×1.5×1.2-cm, hemorrhagic, crusted, | ||

| ||

| ||
| Figure 2. Merkel cells are small- to medium-sized cells with round nuclei and scant cytoplasm. Granular or stippled chromatin can be seen (A)(H&E, original magnification ×40). Merkel cell carcinoma with trabecular pattern (B) (H&E, original magnification ×10). | ||
Between 1986 and 2006, the incidence of MCC grew substantially.1,2 Figures have been reported at 0.15 cases per 100,000 individuals to 0.6 cases per 100,000 individuals worldwide. In the United States, the age-adjusted incidence of MCC is estimated at 0.24 per 100,000 person-years, which is higher than the estimated 0.13 per 100,000 person-years found in Europe.3 The highest incidence worldwide has been noted in Western Australia, likely due to high levels of UV exposure.1 The incidence of MCC in psoriasis patients who are treated with oral methoxsalen (psoralen) and UVA photochemotherapy is 100 times greater than in the general population, further supporting the role of UV light in the development of MCC.1 White individuals have the highest incidence of MCC worldwide, with men being affected more frequently than women.1,3 The majority of patients with MCC are diagnosed at 70 years or older.1 Approximately 5% of reported MCC patients are diagnosed before 50 years of age.2 Immunosuppression and immunodeficiency likely play a role in the pathogenesis of MCC, and the incidence is increased in solid organ transplant recipients, most commonly renal transplant recipients,1 as well as individuals with chronic lymphocytic leukemia, human immunodeficiency virus infection, and AIDS.1,3 Patients with autoimmune diseases such as rheumatoid arthritis also are at increased risk for MCC.3 Individuals who are diagnosed with MCC are at an increased risk for development of other malignancies including nonmelanoma skin cancers, chronic lymphocytic leukemia, Hodgkin lymphoma, and non-Hodgkin lymphoma.3
Clinical Presentation of MCC
The clinical presentation of MCC can be variable. Most tumors present as firm, red to purple, nontender papules or nodules (Figure 1).1 Tumor size may range from 2 to 200 mm but is most commonly less than 20 mm.2 Growth can be rapid, and tumors are most commonly located on sun-exposed skin. The head and neck areas account for 48% of all MCC cases,1 with the eyelids being frequently involved.2 Merkel cell carcinoma also has been reported on the arms, legs, trunk, back, and buttocks.1 Non–sun-exposed areas are less commonly affected. Mucosal sites (eg, larynx, nasal cavity, pharynx, mouth) account for 5% of primary MCCs.1 Merkel cell carcinoma also has been reported to affect the vulva and penis. Subcutaneous primary MCC has presented without overlying epidermal changes.1 In a case series by Heath et al,6 14% (27/195) of MCC patients presented with nodal disease without any identifiable primary tumor, with the inguinal nodal chain being the most common for this presentation. It currently is not known whether these nodal tumors are primary tumors or metastatic disease with a regression of the primary tumor.1
|
Histopathology of MCC

| ||

| ||

| ||
Figure 3. Positive chromogranin staining (A)(original | ||
Merkel cells are small- to medium-sized basophilic cells with round nuclei and scant cytoplasm. Granular or stippled chromatin can be seen on histopathology (Figure 2A).1 Some tumor cells have more vesicular chromatin, multiple small nucleoli, irregular contours, and more abundant cytoplasm. In some reports, irregular contours and abundant cytoplasm were associated with no detectible MCPyV infection.1,3 Merkel cell carcinomas have a primarily nodular architecture, and classification is based on growth pattern and cell size. Three histopathologic growth patterns have been described: nodular, infiltrative, and trabecular. The trabecular pattern is composed of interconnecting strands ofcells (Figure 2B). Tumors with solely intraepidermal involvement (MCC in situ) have been described but are exceedingly rare.1 Cell types are classified according to size, with the intermediate cell type being the most common. The small cell variant may be mistaken for a lymphocytic infiltrate due to the similar size and appearance of both types of cells.1,3
Merkel cell carcinomas can have histopathological overlap with lymphomas, small cell lung cancers, carcinoid tumors, primitive neuroectodermal tumors, neuroblastomas, small cell osteosarcomas, rhabdomyosarcomas, or Ewing sarcomas.1,3 Specifically, differentiation from small cell carcinoma of the lung is of utmost importance. Merkel cell carcinoma stains positively for cytokeratins 8, 18, 19, and 20. The neuroendocrine markers chromogranin (Figure 3A), synaptophysin, and neuron-specific enolase also may show positive staining. Cytokeratin 20, low-molecular-weight cytokeratins (CAM 5.2), and neurofilament immunostains have a high sensitivity for MCC and are the most frequently used.1 Cytokeratin 20 stains in the characteristic paranuclear dot–like pattern, which is a hallmark of MCC (Figure 3B). Cytokeratin 20 positivity in conjunction with negative staining for thyroid transcription factor 1 (Figure 3C) and cytokeratin 7 can aid in differentiation from small cell carcinoma of the lung.1,3
Pathogenesis of MCC
In 2008, Feng et al7 discovered a novel polyomavirus associated with the development of MCC. This novel polyomavirus, MCPyV, is found in approximately 80% of all cases of MCC. Seventeen members of the polyomavirus family have been identified, 9 of which have been found to infect humans, including BK virus, JC virus, WU, MCPyV, human polyomavirus 6, human polyomavirus 7, trichodysplasia spinulosa–associated polyomavirus, human polyomavirus 9, and Simian virus 40.1 Merkel cell polyomavirus infection is found in approximately 60% of the general population and exposure likely occurs early in life. The virus likely is transmitted through skin shedding and nasal secretions, though it also has been found in urine specimens.3 Currently, there is no evidence to suggest vertical viral transmission from mother to fetus.
Merkel cell polyomavirus is composed of early and late gene regions. The early gene region contains both large T antigen (LT) and small T antigen reading frames, which are necessary for viral replication.8 The late region is responsible for encoding viral proteins necessary for viral capsid assembly. Mutations found in viral protein 1 prevent formation of viral particles.9 Large T antigen is substantially overexpressed in MCC and is responsible for tumor suppression through retinoblastoma tumor suppressor protein. It also serves as a binding domain for both heat shock proteins and helicases.8,10 These domains allow the polyomaviruses to use host-cell machinery for viral genome replication while targeting tumor suppressor proteins.8 Upon viral integration into host DNA, viral replication ceases while oncogenic function persists.
The exact mechanism by which the MCPyV contributes to the development of MCC still has yet to be identified. Hypotheses suggest a combination of viral infection with external mutagens (eg, UV radiation). Experimental observations suggest viral contribution is likely due to the large percentage of MCCs that are positive for MCPyV, the identification of LT antigen expression and the role it plays in preserving cell cycle progression, and the role persistent LT antigen expression plays in continued growth of MCC cell lines in vitro.8 Two important cell line preservation mechanisms ensure continued tumor growth, including prevention of apoptosis triggered by DNA damage response mechanisms following UV damage and interaction with the retinoblastoma tumor suppressor protein allowing continued growth.8,11 Other important factors in tumor growth and survival may be the inhibition of apoptosis through the BCL2 (B-cell chronic lymphocytic leukemia/lymphoma 2) proto-oncogene and survivin (baculoviral inhibitor of apoptosis repeat-containing 5 [BIRC5]).12 Survivin has been found to play an important role in MCPyV-positive MCCs.12,13 It has been suggested that lymphangiogenesis in MCC likely is driven by vascular endothelial growth factor-C+CD68+CD163+ M2 macrophages.14 Another survival mechanism specific to polyomaviruses is their ability to interfere with the p53 tumor suppressor pathway.8 Loss of p53 expression by tumor cell nuclei has been associated with poor prognosis.15
Immune Response
Immune response as a role in tumor progression can be primarily centered on the concept of persistent antigen expression as a means of immune downregulation. Dunn et al16 suggested that cancer cells must interact through 3 consecutive phases with the host immune system (immunoediting hypothesis). In the elimination phase, the host immune system is able to recognize and destroy newly transformed cells through both the innate and adaptive immune systems. The second equilibrium phase allows the tumor to remain dormant and growth remains stagnant. Lastly, the tumor is allowed to evade the immune system through the escape phase.8
Host immune responses play an important role in both the progression and prognosis of MCC. High anti-MCPyV capsid antibody titers have been associated with better progression-free survival in some patients.8 Patients with high antibody titers (>10,000) likely have better progression-free survival than those with low antibody titers (<10,000).17 Antibody titers to the LT antigen may serve as a biomarker of MCC disease burden in the future. Rising LT antigen titers have been shown to correlate with disease progression and falling titers correlate with successful treatment.8 Tumoral infiltration of CD8+ T lymphocytes has been shown to be a predictor of survival compared to no intratumoral infiltration.6 Sihto et al18 suggested that this better prognosis from high intratumoral infiltration is not specific to MCPyV-positive MCC; however, it does highlight an important aspect of tumor evasion through the downregulation of cell surface expression of class I major histocompatibility complex antigens, which allows presentation of tumor intracellular peptides to CD8+ T lymphocytes.8 Upregulation of this specific immune response may play a role in the future treatment of MCC.
Staging and Prognosis
Due to the extremely aggressive nature of MCC, patients with local disease and tumors 2 cm or smaller in diameter have a 66% survival at 5 years.1,3 The 5-year survival rate for patients with local metastasis to regional lymph nodes ranges from 26% to 42%. Patients with distant metastasis have an 18% survival rate at 5 years.1,3 Data suggest that sentinel lymph node biopsy should be performed on all patients with MCC regardless of tumor size.1 There are no consensus guidelines to date regarding imaging for the staging of MCC patients. It is suggested that (18F)fluorodeoxyglucose positron emission tomography alone or in combination with computed tomography (CT) may be of value as a single whole-body diagnostic tool for accurate staging.10 It also has been suggested that (18F)fluorodeoxyglucose positron emission tomography and CT may offer more accurate staging than other screening modalities such as CT alone or magnetic resonance imaging.14,19
Treatment of MCC
Surgery remains the mainstay of treatment of MCC. Current National Comprehensive Cancer Network guidelines20 recommend 1- to 2-cm margins for wide local excision or treatment with Mohs micrographic surgery. Sentinel lymph node biopsy should be performed intraoperatively in patients undergoing wide local excision and preoperatively for patients undergoing Mohs micrographic surgery due to potential alterations in lymphatic drainage that may affect lymphoscintigraphy.1
Radiation may be used as primary or adjuvant therapy in patients with MCC. Radiation as primary therapy generally is reserved for patients who are not surgical candidates. It has been suggested that there was no difference in outcome in a small group of patients treated with radiation alone compared to patients who underwent surgery and radiation to the tumor bed.1 Current guidelines suggest a small group of patients may not require adjuvant therapy following adequate resection of some small tumors, and clinical observation may be appropriate.1,3 Chemotherapy may play a palliative role in patients with metastatic MCC. Merkel cell carcinoma has been shown to be chemosensitive but with a high recurrence rate.1 Because the immune system plays an important role in disease prognosis, having an intact immune system likely is paramount in the prevention of further disease progression.
Future Treatments of MCC
Future treatment of MCC may be focused on the viral etiology of most tumors and upregulation of the immune response, which may lead to the possibility of specifically interfering with virus-specific oncoproteins and stimulation of immune responses to virally infected tumor cells.8 The MCPyV large T antigen has been found to be overexpressed in some tumors and may serve as a specific target of therapy.10,21 Survivin, a key cell cycle protein encoded by LT antigen, may be an interesting target given its implication in other cancers.13 Other potential nonviral molecular target antigens include the oncoprotein H1P1 that interacts with c-KIT.8 Specific immunostimulatory cytokines that may be used to upregulate the immune response to tumoral cells may include IL-2, IL-12, IL-15, or IL-21. Therapeutic agents that may be studied in the future to target the immune exhaustion phenomenon associated with tumorigenesis include ipilimumab (cytotoxic T lymphocyte antigen 4 receptor-blocking agent) as well as programmed cell death 1 and programmed cell death 1 ligand 1 (PD-1/PD-L1).8 Neuroendocrine tumors including MCC tend to be highly vascular and express vascular endothelial growth factors and platelet-derived growth factors, which may be other potential therapeutic targets. It has been reported that approximately 95% of MCC patients have CD56+ tumors, and current clinical trials suggest a promising therapeutic response with the immunogen anti-CD56 monoclonal antibody.3
Conclusion
Merkel cell carcinoma is a rare aggressive neuroendocrine tumor that has been associated with a novel polyomavirus. Merkel cell carcinoma tends to affect elderly and immunocompromised patients as well as white individuals. Tumors are most often found in areas of high UV exposure and clinically on sun-exposed skin. Merkel cell polyomavirus is associated with approximately 80% of tumors, and tumorigenesis likely is caused by a number of sequential steps from viral integration into host DNA, mutagenic events, and specific immune responses. Currently there are no consensus guidelines for using imaging for staging of MCCs, but sentinel lymph node biopsy is recommended for all cases due to the aggressive nature of even smaller tumors. Surgery remains the mainstay of treatment, and radiation therapy may be used as a primary or adjuvant treatment. Chemotherapy usually is reserved for patients with metastatic disease purely for palliation. Future treatments of MCC likely will center on the viral etiology of MCC and upregulation of immune responses to virally infected tumor cells.
Merkel cells originally were described by German histopathologist Friedrich Sigmund Merkel in 1875. These unique tactile cells were described as epidermal, nondendritic, and nonkeratinizing. Merkel cells are thought to arise from the neural crest and are believed to be primary neural cells found within the basal layer of the epidermis.1,2 They likely function primarily as slowly adapting type I mechanoreceptors. Origin from the neural crest is controversial, as other investigators have suggested derivation from epidermal keratinocytes.1,2 Tumor cells have been linked to the amine precursor uptake and decarboxylation system.3 In 1972, Toker4 described several cases of trabecular or sweat gland carcinomas of the skin. Upon further investigation, the cells that comprised these tumors were found to have dense core granules on electron microscopy, typical of Merkel cells.1,2 Other terms such as neuroendocrine carcinoma of the skin, small cell carcinoma of the skin, and anaplastic carcinoma of the skin have been used to describe Merkel cell carcinoma (MCC),1 which was suggested by De Wolf-Peeters et al5 in 1980.
Despite being a rare malignancy, MCC follows an aggressive clinical course. Upon presentation, approximately 66% of patients have local disease, 27% have nodal involvement, and 7% have distant metastasis.1 Future treatments will likely center around the novel Merkel cell polyomavirus (MCPyV) and modification of immune responses toward tumor cells. Standardization continues to be lacking in both staging and treatment of this aggressive tumor.
Epidemiology of MCC

| ||
Figure 1. A 2.3×1.5×1.2-cm, hemorrhagic, crusted, | ||

| ||

| ||
| Figure 2. Merkel cells are small- to medium-sized cells with round nuclei and scant cytoplasm. Granular or stippled chromatin can be seen (A)(H&E, original magnification ×40). Merkel cell carcinoma with trabecular pattern (B) (H&E, original magnification ×10). | ||
Between 1986 and 2006, the incidence of MCC grew substantially.1,2 Figures have been reported at 0.15 cases per 100,000 individuals to 0.6 cases per 100,000 individuals worldwide. In the United States, the age-adjusted incidence of MCC is estimated at 0.24 per 100,000 person-years, which is higher than the estimated 0.13 per 100,000 person-years found in Europe.3 The highest incidence worldwide has been noted in Western Australia, likely due to high levels of UV exposure.1 The incidence of MCC in psoriasis patients who are treated with oral methoxsalen (psoralen) and UVA photochemotherapy is 100 times greater than in the general population, further supporting the role of UV light in the development of MCC.1 White individuals have the highest incidence of MCC worldwide, with men being affected more frequently than women.1,3 The majority of patients with MCC are diagnosed at 70 years or older.1 Approximately 5% of reported MCC patients are diagnosed before 50 years of age.2 Immunosuppression and immunodeficiency likely play a role in the pathogenesis of MCC, and the incidence is increased in solid organ transplant recipients, most commonly renal transplant recipients,1 as well as individuals with chronic lymphocytic leukemia, human immunodeficiency virus infection, and AIDS.1,3 Patients with autoimmune diseases such as rheumatoid arthritis also are at increased risk for MCC.3 Individuals who are diagnosed with MCC are at an increased risk for development of other malignancies including nonmelanoma skin cancers, chronic lymphocytic leukemia, Hodgkin lymphoma, and non-Hodgkin lymphoma.3
Clinical Presentation of MCC
The clinical presentation of MCC can be variable. Most tumors present as firm, red to purple, nontender papules or nodules (Figure 1).1 Tumor size may range from 2 to 200 mm but is most commonly less than 20 mm.2 Growth can be rapid, and tumors are most commonly located on sun-exposed skin. The head and neck areas account for 48% of all MCC cases,1 with the eyelids being frequently involved.2 Merkel cell carcinoma also has been reported on the arms, legs, trunk, back, and buttocks.1 Non–sun-exposed areas are less commonly affected. Mucosal sites (eg, larynx, nasal cavity, pharynx, mouth) account for 5% of primary MCCs.1 Merkel cell carcinoma also has been reported to affect the vulva and penis. Subcutaneous primary MCC has presented without overlying epidermal changes.1 In a case series by Heath et al,6 14% (27/195) of MCC patients presented with nodal disease without any identifiable primary tumor, with the inguinal nodal chain being the most common for this presentation. It currently is not known whether these nodal tumors are primary tumors or metastatic disease with a regression of the primary tumor.1
|
Histopathology of MCC

| ||

| ||

| ||
Figure 3. Positive chromogranin staining (A)(original | ||
Merkel cells are small- to medium-sized basophilic cells with round nuclei and scant cytoplasm. Granular or stippled chromatin can be seen on histopathology (Figure 2A).1 Some tumor cells have more vesicular chromatin, multiple small nucleoli, irregular contours, and more abundant cytoplasm. In some reports, irregular contours and abundant cytoplasm were associated with no detectible MCPyV infection.1,3 Merkel cell carcinomas have a primarily nodular architecture, and classification is based on growth pattern and cell size. Three histopathologic growth patterns have been described: nodular, infiltrative, and trabecular. The trabecular pattern is composed of interconnecting strands ofcells (Figure 2B). Tumors with solely intraepidermal involvement (MCC in situ) have been described but are exceedingly rare.1 Cell types are classified according to size, with the intermediate cell type being the most common. The small cell variant may be mistaken for a lymphocytic infiltrate due to the similar size and appearance of both types of cells.1,3
Merkel cell carcinomas can have histopathological overlap with lymphomas, small cell lung cancers, carcinoid tumors, primitive neuroectodermal tumors, neuroblastomas, small cell osteosarcomas, rhabdomyosarcomas, or Ewing sarcomas.1,3 Specifically, differentiation from small cell carcinoma of the lung is of utmost importance. Merkel cell carcinoma stains positively for cytokeratins 8, 18, 19, and 20. The neuroendocrine markers chromogranin (Figure 3A), synaptophysin, and neuron-specific enolase also may show positive staining. Cytokeratin 20, low-molecular-weight cytokeratins (CAM 5.2), and neurofilament immunostains have a high sensitivity for MCC and are the most frequently used.1 Cytokeratin 20 stains in the characteristic paranuclear dot–like pattern, which is a hallmark of MCC (Figure 3B). Cytokeratin 20 positivity in conjunction with negative staining for thyroid transcription factor 1 (Figure 3C) and cytokeratin 7 can aid in differentiation from small cell carcinoma of the lung.1,3
Pathogenesis of MCC
In 2008, Feng et al7 discovered a novel polyomavirus associated with the development of MCC. This novel polyomavirus, MCPyV, is found in approximately 80% of all cases of MCC. Seventeen members of the polyomavirus family have been identified, 9 of which have been found to infect humans, including BK virus, JC virus, WU, MCPyV, human polyomavirus 6, human polyomavirus 7, trichodysplasia spinulosa–associated polyomavirus, human polyomavirus 9, and Simian virus 40.1 Merkel cell polyomavirus infection is found in approximately 60% of the general population and exposure likely occurs early in life. The virus likely is transmitted through skin shedding and nasal secretions, though it also has been found in urine specimens.3 Currently, there is no evidence to suggest vertical viral transmission from mother to fetus.
Merkel cell polyomavirus is composed of early and late gene regions. The early gene region contains both large T antigen (LT) and small T antigen reading frames, which are necessary for viral replication.8 The late region is responsible for encoding viral proteins necessary for viral capsid assembly. Mutations found in viral protein 1 prevent formation of viral particles.9 Large T antigen is substantially overexpressed in MCC and is responsible for tumor suppression through retinoblastoma tumor suppressor protein. It also serves as a binding domain for both heat shock proteins and helicases.8,10 These domains allow the polyomaviruses to use host-cell machinery for viral genome replication while targeting tumor suppressor proteins.8 Upon viral integration into host DNA, viral replication ceases while oncogenic function persists.
The exact mechanism by which the MCPyV contributes to the development of MCC still has yet to be identified. Hypotheses suggest a combination of viral infection with external mutagens (eg, UV radiation). Experimental observations suggest viral contribution is likely due to the large percentage of MCCs that are positive for MCPyV, the identification of LT antigen expression and the role it plays in preserving cell cycle progression, and the role persistent LT antigen expression plays in continued growth of MCC cell lines in vitro.8 Two important cell line preservation mechanisms ensure continued tumor growth, including prevention of apoptosis triggered by DNA damage response mechanisms following UV damage and interaction with the retinoblastoma tumor suppressor protein allowing continued growth.8,11 Other important factors in tumor growth and survival may be the inhibition of apoptosis through the BCL2 (B-cell chronic lymphocytic leukemia/lymphoma 2) proto-oncogene and survivin (baculoviral inhibitor of apoptosis repeat-containing 5 [BIRC5]).12 Survivin has been found to play an important role in MCPyV-positive MCCs.12,13 It has been suggested that lymphangiogenesis in MCC likely is driven by vascular endothelial growth factor-C+CD68+CD163+ M2 macrophages.14 Another survival mechanism specific to polyomaviruses is their ability to interfere with the p53 tumor suppressor pathway.8 Loss of p53 expression by tumor cell nuclei has been associated with poor prognosis.15
Immune Response
Immune response as a role in tumor progression can be primarily centered on the concept of persistent antigen expression as a means of immune downregulation. Dunn et al16 suggested that cancer cells must interact through 3 consecutive phases with the host immune system (immunoediting hypothesis). In the elimination phase, the host immune system is able to recognize and destroy newly transformed cells through both the innate and adaptive immune systems. The second equilibrium phase allows the tumor to remain dormant and growth remains stagnant. Lastly, the tumor is allowed to evade the immune system through the escape phase.8
Host immune responses play an important role in both the progression and prognosis of MCC. High anti-MCPyV capsid antibody titers have been associated with better progression-free survival in some patients.8 Patients with high antibody titers (>10,000) likely have better progression-free survival than those with low antibody titers (<10,000).17 Antibody titers to the LT antigen may serve as a biomarker of MCC disease burden in the future. Rising LT antigen titers have been shown to correlate with disease progression and falling titers correlate with successful treatment.8 Tumoral infiltration of CD8+ T lymphocytes has been shown to be a predictor of survival compared to no intratumoral infiltration.6 Sihto et al18 suggested that this better prognosis from high intratumoral infiltration is not specific to MCPyV-positive MCC; however, it does highlight an important aspect of tumor evasion through the downregulation of cell surface expression of class I major histocompatibility complex antigens, which allows presentation of tumor intracellular peptides to CD8+ T lymphocytes.8 Upregulation of this specific immune response may play a role in the future treatment of MCC.
Staging and Prognosis
Due to the extremely aggressive nature of MCC, patients with local disease and tumors 2 cm or smaller in diameter have a 66% survival at 5 years.1,3 The 5-year survival rate for patients with local metastasis to regional lymph nodes ranges from 26% to 42%. Patients with distant metastasis have an 18% survival rate at 5 years.1,3 Data suggest that sentinel lymph node biopsy should be performed on all patients with MCC regardless of tumor size.1 There are no consensus guidelines to date regarding imaging for the staging of MCC patients. It is suggested that (18F)fluorodeoxyglucose positron emission tomography alone or in combination with computed tomography (CT) may be of value as a single whole-body diagnostic tool for accurate staging.10 It also has been suggested that (18F)fluorodeoxyglucose positron emission tomography and CT may offer more accurate staging than other screening modalities such as CT alone or magnetic resonance imaging.14,19
Treatment of MCC
Surgery remains the mainstay of treatment of MCC. Current National Comprehensive Cancer Network guidelines20 recommend 1- to 2-cm margins for wide local excision or treatment with Mohs micrographic surgery. Sentinel lymph node biopsy should be performed intraoperatively in patients undergoing wide local excision and preoperatively for patients undergoing Mohs micrographic surgery due to potential alterations in lymphatic drainage that may affect lymphoscintigraphy.1
Radiation may be used as primary or adjuvant therapy in patients with MCC. Radiation as primary therapy generally is reserved for patients who are not surgical candidates. It has been suggested that there was no difference in outcome in a small group of patients treated with radiation alone compared to patients who underwent surgery and radiation to the tumor bed.1 Current guidelines suggest a small group of patients may not require adjuvant therapy following adequate resection of some small tumors, and clinical observation may be appropriate.1,3 Chemotherapy may play a palliative role in patients with metastatic MCC. Merkel cell carcinoma has been shown to be chemosensitive but with a high recurrence rate.1 Because the immune system plays an important role in disease prognosis, having an intact immune system likely is paramount in the prevention of further disease progression.
Future Treatments of MCC
Future treatment of MCC may be focused on the viral etiology of most tumors and upregulation of the immune response, which may lead to the possibility of specifically interfering with virus-specific oncoproteins and stimulation of immune responses to virally infected tumor cells.8 The MCPyV large T antigen has been found to be overexpressed in some tumors and may serve as a specific target of therapy.10,21 Survivin, a key cell cycle protein encoded by LT antigen, may be an interesting target given its implication in other cancers.13 Other potential nonviral molecular target antigens include the oncoprotein H1P1 that interacts with c-KIT.8 Specific immunostimulatory cytokines that may be used to upregulate the immune response to tumoral cells may include IL-2, IL-12, IL-15, or IL-21. Therapeutic agents that may be studied in the future to target the immune exhaustion phenomenon associated with tumorigenesis include ipilimumab (cytotoxic T lymphocyte antigen 4 receptor-blocking agent) as well as programmed cell death 1 and programmed cell death 1 ligand 1 (PD-1/PD-L1).8 Neuroendocrine tumors including MCC tend to be highly vascular and express vascular endothelial growth factors and platelet-derived growth factors, which may be other potential therapeutic targets. It has been reported that approximately 95% of MCC patients have CD56+ tumors, and current clinical trials suggest a promising therapeutic response with the immunogen anti-CD56 monoclonal antibody.3
Conclusion
Merkel cell carcinoma is a rare aggressive neuroendocrine tumor that has been associated with a novel polyomavirus. Merkel cell carcinoma tends to affect elderly and immunocompromised patients as well as white individuals. Tumors are most often found in areas of high UV exposure and clinically on sun-exposed skin. Merkel cell polyomavirus is associated with approximately 80% of tumors, and tumorigenesis likely is caused by a number of sequential steps from viral integration into host DNA, mutagenic events, and specific immune responses. Currently there are no consensus guidelines for using imaging for staging of MCCs, but sentinel lymph node biopsy is recommended for all cases due to the aggressive nature of even smaller tumors. Surgery remains the mainstay of treatment, and radiation therapy may be used as a primary or adjuvant treatment. Chemotherapy usually is reserved for patients with metastatic disease purely for palliation. Future treatments of MCC likely will center on the viral etiology of MCC and upregulation of immune responses to virally infected tumor cells.
1. Han S, North J, Canavan T, et al. Merkel cell carcinoma. Hematol Oncol Clin N Am. 2012;26:1351-1374.
2. Goessling W, McKee P, Mayer R. Merkel cell carcinoma. J Clin Oncol. 2002;20:588-598.
3. Donepudi S, DeConti R, Samlowski W. Recent advances in the understanding of the genetics, etiology, and treatment of merkel cell carcinoma. Semin Oncol. 2012;39:163-172.
4. Toker C. Trabecular carcinoma of the skin. Arch Dermatol. 1972;105:107-110.
5. De Wolff-Peeters C, Marien K, Mebis J, et al. A cutaneous APUDoma or Merkel cell tumor? a morphologically recognizable tumor with a biological and histological malignant aspect in contrast with its clinical behavior. Cancer. 1980;46:810-816.
6. Heath M, Jaimes N, Lemos B, et al. Clinical characteristics of Merkel cell carcinoma at diagnosis in 195 patients: the AEIOU features. J Am Acad Dermatol. 2008;58:375-381.
7. Feng H, Shuda M, Chang Y, et al. Clonal integration of a polyomavirus in human Merkel cell carcinoma. Science. 2008;319:1096-1100.
8. Bhatia S, Afanasiev O, Nghiem P. Immunobiology of Merkel cell carcinoma: implications for immunotherapy of a polyomavirus-associated cancer. Curr Oncol Rep. 2011;13:488-497.
9. Amber K, McLeod M, Nouri K. The Merkel cell polyomavirus and its involvement in Merkel cell carcinoma. Dermatol Surg. 2013;39:232-238.
10. Erovic B, Al Habeeb A, Harris L, et al. Significant overexpression of the Merkel cell polyomavirus (MCPyV) large T antigen in Merkel cell carcinoma. Head Neck. 2013;35:184-189.
11. Demetriou S, Ona-Vu K, Sullivan E, et al. Defective DNA repair and cell cycle arrest in cells expressing Merkel cell polyomavirus T antigen. Int J Cancer. 2012;131:1818-1827.
12. Sahi H, Koljonen V, Kavola H, et al. Bcl-2 expression indicates better prognosis of Merkel cell carcinoma regardless of the presence of Merkel cell polyomavirus. Virchows Arch. 2012;461:553-559.
13. Arora R, Shuda M, Guastafierro A, et al. Survivin is a therapeutic target in Merkel cell carcinoma. Sci Transl Med. 2012;4:1-11.
14. Hawryluk E, O’Regan K, Sheehy N, et al. Positron emission tomography/computed tomography imaging in Merkel cell carcinoma: a study of 270 scans in 97 patients at the Dana-Farber/Brigham and Women’s Cancer Center. J Am Acad Dermatol. 2013;68:592-599.
15. Hall B, Pincus L, Yu S, et al. Immunohistochemical prognostication of Merkel cell carcinoma: p63 expression but not polyomavirus status correlates with outcome. J Cutan Pathol. 2012;39:911-917.
16. Dunn GP, Bruce AT, Ikeda H, et al. Cancer immunoediting: from immunosurveillance to tumor escape. Nat Immunol. 2002;3:991-998.
17. Touze A, Le Bidre E, Laude H, et al. High levels of antibodies against Merkel cell polyomavirus identify a subset of patients with Merkel cell carcinoma with better clinical outcome. J Clin Oncol. 2011;29:1612-1619.
18. Sihto H, Bohling T, Kavola H, et al. Tumor infiltrating immune cells and outcome of Merkel cell carcinoma: a population-based study. Clin Cancer Res. 2012;18:2872-2881.
19. Colgan M, Tarantola T, Weaver A, et al. The predictive value of imaging studies in evaluating regional lymph node involvement in Merkel cell carcinoma. J Am Acad Dermatol. 2012;67:1250-1256.
20. NCCN Clinical Practice Guidelines in Oncology. National Comprehensive Cancer Network website. http://www.nccn.org/professionals/physician_gls/f_guidelines.asp#site. Accessed March 22, 2016.
21. Angermeyer S, Hesbacher S, Becker J, et al. Merkel cell polyomavirus-positive Merkel cell carcinoma cells do not require expression of the viral small T antigen. J Invest Dermatol. 2013;133:1-6.
1. Han S, North J, Canavan T, et al. Merkel cell carcinoma. Hematol Oncol Clin N Am. 2012;26:1351-1374.
2. Goessling W, McKee P, Mayer R. Merkel cell carcinoma. J Clin Oncol. 2002;20:588-598.
3. Donepudi S, DeConti R, Samlowski W. Recent advances in the understanding of the genetics, etiology, and treatment of merkel cell carcinoma. Semin Oncol. 2012;39:163-172.
4. Toker C. Trabecular carcinoma of the skin. Arch Dermatol. 1972;105:107-110.
5. De Wolff-Peeters C, Marien K, Mebis J, et al. A cutaneous APUDoma or Merkel cell tumor? a morphologically recognizable tumor with a biological and histological malignant aspect in contrast with its clinical behavior. Cancer. 1980;46:810-816.
6. Heath M, Jaimes N, Lemos B, et al. Clinical characteristics of Merkel cell carcinoma at diagnosis in 195 patients: the AEIOU features. J Am Acad Dermatol. 2008;58:375-381.
7. Feng H, Shuda M, Chang Y, et al. Clonal integration of a polyomavirus in human Merkel cell carcinoma. Science. 2008;319:1096-1100.
8. Bhatia S, Afanasiev O, Nghiem P. Immunobiology of Merkel cell carcinoma: implications for immunotherapy of a polyomavirus-associated cancer. Curr Oncol Rep. 2011;13:488-497.
9. Amber K, McLeod M, Nouri K. The Merkel cell polyomavirus and its involvement in Merkel cell carcinoma. Dermatol Surg. 2013;39:232-238.
10. Erovic B, Al Habeeb A, Harris L, et al. Significant overexpression of the Merkel cell polyomavirus (MCPyV) large T antigen in Merkel cell carcinoma. Head Neck. 2013;35:184-189.
11. Demetriou S, Ona-Vu K, Sullivan E, et al. Defective DNA repair and cell cycle arrest in cells expressing Merkel cell polyomavirus T antigen. Int J Cancer. 2012;131:1818-1827.
12. Sahi H, Koljonen V, Kavola H, et al. Bcl-2 expression indicates better prognosis of Merkel cell carcinoma regardless of the presence of Merkel cell polyomavirus. Virchows Arch. 2012;461:553-559.
13. Arora R, Shuda M, Guastafierro A, et al. Survivin is a therapeutic target in Merkel cell carcinoma. Sci Transl Med. 2012;4:1-11.
14. Hawryluk E, O’Regan K, Sheehy N, et al. Positron emission tomography/computed tomography imaging in Merkel cell carcinoma: a study of 270 scans in 97 patients at the Dana-Farber/Brigham and Women’s Cancer Center. J Am Acad Dermatol. 2013;68:592-599.
15. Hall B, Pincus L, Yu S, et al. Immunohistochemical prognostication of Merkel cell carcinoma: p63 expression but not polyomavirus status correlates with outcome. J Cutan Pathol. 2012;39:911-917.
16. Dunn GP, Bruce AT, Ikeda H, et al. Cancer immunoediting: from immunosurveillance to tumor escape. Nat Immunol. 2002;3:991-998.
17. Touze A, Le Bidre E, Laude H, et al. High levels of antibodies against Merkel cell polyomavirus identify a subset of patients with Merkel cell carcinoma with better clinical outcome. J Clin Oncol. 2011;29:1612-1619.
18. Sihto H, Bohling T, Kavola H, et al. Tumor infiltrating immune cells and outcome of Merkel cell carcinoma: a population-based study. Clin Cancer Res. 2012;18:2872-2881.
19. Colgan M, Tarantola T, Weaver A, et al. The predictive value of imaging studies in evaluating regional lymph node involvement in Merkel cell carcinoma. J Am Acad Dermatol. 2012;67:1250-1256.
20. NCCN Clinical Practice Guidelines in Oncology. National Comprehensive Cancer Network website. http://www.nccn.org/professionals/physician_gls/f_guidelines.asp#site. Accessed March 22, 2016.
21. Angermeyer S, Hesbacher S, Becker J, et al. Merkel cell polyomavirus-positive Merkel cell carcinoma cells do not require expression of the viral small T antigen. J Invest Dermatol. 2013;133:1-6.
Practice Points
- Merkel cell carcinoma has been associated with a novel polyomavirus.
- Merkel cell carcinoma follows a very aggressive course and is most likely metastatic at diagnosis.
Toe Nodule Obliterating the Nail Bed
The Diagnosis: Superficial Acral Fibromyxoma
Superficial acral fibromyxoma (SAF) was first described in 2001 by Fetsch et al.1 Subsequently, the term digital fibromyxoma was proposed in 2012 by Hollmann et al2 to describe a distinctive, slow-growing, soft-tissue tumor with a predilection for the periungual or subungual regions of the fingers and toes. The benign growth typically presents as a painless or tender nodule in middle-aged adults with a slight male predominance (1.3:1 ratio).1,2 In a case series (N=124) described by Hollmann et al,2 9 of 25 patients (36%) who had imaging studies showed bone involvement by an erosive or lytic lesion. Reports of SAF with bone involvement also have been described in the radiologic and orthopedic surgery literature.3,4 Radiographically, the soft-tissue invasion of the bone is demonstrated by scalloping on plain radiographs (Figure 1).3
Histologically, SAFs are moderately cellular with spindled or stellate fibroblastlike cells within a myxoid or collagenous matrix (Figure 2).1 The vasculature is mildly accentuated and an increase in mast cells usually is observed. The nuclei have a low degree of atypia with few mitotic figures, and the stellate cells exhibit positive immunohistochemical staining for CD34 (Figure 3), epithelial membrane antigen, and CD99.1 Hollmann et al2 found that 66 of 95 tumors (69.5%) infiltrated the dermal collagen, 26 (27.4%) infiltrated fat, and 3 (3.2%) invaded bone. Of the 47 cases that were evaluated on follow-up, 10 tumors (21.3%) recurred locally (all near the nail unit of the fingers or toes) after a mean interval of 27 months. Although invasion of underlying tissues and recurrence of the tumor has been demonstrated, this growth is considered benign. The histologic differential diagnosis includes neurofibroma, myxoma, fibroma, low-grade fibromyxoid sarcoma, dermatofibroma, superficial angiomyxoma, and dermatofibrosarcoma protuberans.2
The primary treatment of SAF is local excision. The incidence of local recurrence found in the case series by Hollmann et al2 was directly linked to positive margins after the first excision (10/47 [21.3%] recurrent lesions had positive margins). To date, there are no known reports of metastatic disease in SAF.2 Our case manifested with a late recurrence of the tumor and bone involvement requiring surgical excision, which illustrates the role of adjuvant imaging and close follow-up following excision of any soft-tissue tumors of the fingers and toes that have been histologically confirmed as SAF, particularly those of the periungual region.
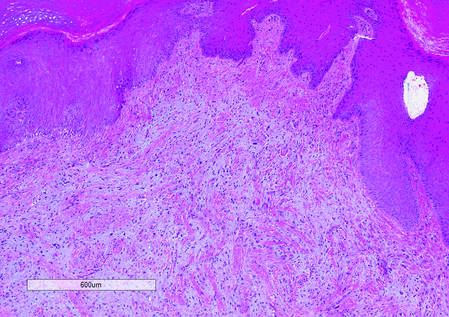
|
|
|
|
1. Fetsch JF, Laskin WB, Miettinen M. Superficial acral fibromyxoma (a clinicopathologic and immunohistochemical analysis of 37 cases of a distinctive soft tissue tumor with a predilection for the fingers and toes.) Hum Pathol. 2001;32:704-714.
2. Hollmann TJ, Bovée JV, Fletcher CD. Digital fibromyxoma (superficial acral fibromyxoma): a detailed characterization of 124 cases. Am J Surg Pathol. 2012;36:789-798.
3. Varikatt W, Soper J, Simmon G, et al. Superficial acral fibromyxoma: a report of two cases with radiological findings. Skeletal Radiol. 2008;37:499-503.
4. Oteo-Alvaro A, Meizoso T, Scarpellini A, et al. Superficial acral fibromyxoma of the toe, with erosion of the distal phalanx. a clinical report. Arch Orthop Trauma Surg. 2008;128:271-274.
The Diagnosis: Superficial Acral Fibromyxoma
Superficial acral fibromyxoma (SAF) was first described in 2001 by Fetsch et al.1 Subsequently, the term digital fibromyxoma was proposed in 2012 by Hollmann et al2 to describe a distinctive, slow-growing, soft-tissue tumor with a predilection for the periungual or subungual regions of the fingers and toes. The benign growth typically presents as a painless or tender nodule in middle-aged adults with a slight male predominance (1.3:1 ratio).1,2 In a case series (N=124) described by Hollmann et al,2 9 of 25 patients (36%) who had imaging studies showed bone involvement by an erosive or lytic lesion. Reports of SAF with bone involvement also have been described in the radiologic and orthopedic surgery literature.3,4 Radiographically, the soft-tissue invasion of the bone is demonstrated by scalloping on plain radiographs (Figure 1).3
Histologically, SAFs are moderately cellular with spindled or stellate fibroblastlike cells within a myxoid or collagenous matrix (Figure 2).1 The vasculature is mildly accentuated and an increase in mast cells usually is observed. The nuclei have a low degree of atypia with few mitotic figures, and the stellate cells exhibit positive immunohistochemical staining for CD34 (Figure 3), epithelial membrane antigen, and CD99.1 Hollmann et al2 found that 66 of 95 tumors (69.5%) infiltrated the dermal collagen, 26 (27.4%) infiltrated fat, and 3 (3.2%) invaded bone. Of the 47 cases that were evaluated on follow-up, 10 tumors (21.3%) recurred locally (all near the nail unit of the fingers or toes) after a mean interval of 27 months. Although invasion of underlying tissues and recurrence of the tumor has been demonstrated, this growth is considered benign. The histologic differential diagnosis includes neurofibroma, myxoma, fibroma, low-grade fibromyxoid sarcoma, dermatofibroma, superficial angiomyxoma, and dermatofibrosarcoma protuberans.2
The primary treatment of SAF is local excision. The incidence of local recurrence found in the case series by Hollmann et al2 was directly linked to positive margins after the first excision (10/47 [21.3%] recurrent lesions had positive margins). To date, there are no known reports of metastatic disease in SAF.2 Our case manifested with a late recurrence of the tumor and bone involvement requiring surgical excision, which illustrates the role of adjuvant imaging and close follow-up following excision of any soft-tissue tumors of the fingers and toes that have been histologically confirmed as SAF, particularly those of the periungual region.

|
|

|
|
The Diagnosis: Superficial Acral Fibromyxoma
Superficial acral fibromyxoma (SAF) was first described in 2001 by Fetsch et al.1 Subsequently, the term digital fibromyxoma was proposed in 2012 by Hollmann et al2 to describe a distinctive, slow-growing, soft-tissue tumor with a predilection for the periungual or subungual regions of the fingers and toes. The benign growth typically presents as a painless or tender nodule in middle-aged adults with a slight male predominance (1.3:1 ratio).1,2 In a case series (N=124) described by Hollmann et al,2 9 of 25 patients (36%) who had imaging studies showed bone involvement by an erosive or lytic lesion. Reports of SAF with bone involvement also have been described in the radiologic and orthopedic surgery literature.3,4 Radiographically, the soft-tissue invasion of the bone is demonstrated by scalloping on plain radiographs (Figure 1).3
Histologically, SAFs are moderately cellular with spindled or stellate fibroblastlike cells within a myxoid or collagenous matrix (Figure 2).1 The vasculature is mildly accentuated and an increase in mast cells usually is observed. The nuclei have a low degree of atypia with few mitotic figures, and the stellate cells exhibit positive immunohistochemical staining for CD34 (Figure 3), epithelial membrane antigen, and CD99.1 Hollmann et al2 found that 66 of 95 tumors (69.5%) infiltrated the dermal collagen, 26 (27.4%) infiltrated fat, and 3 (3.2%) invaded bone. Of the 47 cases that were evaluated on follow-up, 10 tumors (21.3%) recurred locally (all near the nail unit of the fingers or toes) after a mean interval of 27 months. Although invasion of underlying tissues and recurrence of the tumor has been demonstrated, this growth is considered benign. The histologic differential diagnosis includes neurofibroma, myxoma, fibroma, low-grade fibromyxoid sarcoma, dermatofibroma, superficial angiomyxoma, and dermatofibrosarcoma protuberans.2
The primary treatment of SAF is local excision. The incidence of local recurrence found in the case series by Hollmann et al2 was directly linked to positive margins after the first excision (10/47 [21.3%] recurrent lesions had positive margins). To date, there are no known reports of metastatic disease in SAF.2 Our case manifested with a late recurrence of the tumor and bone involvement requiring surgical excision, which illustrates the role of adjuvant imaging and close follow-up following excision of any soft-tissue tumors of the fingers and toes that have been histologically confirmed as SAF, particularly those of the periungual region.

|
|

|
|
1. Fetsch JF, Laskin WB, Miettinen M. Superficial acral fibromyxoma (a clinicopathologic and immunohistochemical analysis of 37 cases of a distinctive soft tissue tumor with a predilection for the fingers and toes.) Hum Pathol. 2001;32:704-714.
2. Hollmann TJ, Bovée JV, Fletcher CD. Digital fibromyxoma (superficial acral fibromyxoma): a detailed characterization of 124 cases. Am J Surg Pathol. 2012;36:789-798.
3. Varikatt W, Soper J, Simmon G, et al. Superficial acral fibromyxoma: a report of two cases with radiological findings. Skeletal Radiol. 2008;37:499-503.
4. Oteo-Alvaro A, Meizoso T, Scarpellini A, et al. Superficial acral fibromyxoma of the toe, with erosion of the distal phalanx. a clinical report. Arch Orthop Trauma Surg. 2008;128:271-274.
1. Fetsch JF, Laskin WB, Miettinen M. Superficial acral fibromyxoma (a clinicopathologic and immunohistochemical analysis of 37 cases of a distinctive soft tissue tumor with a predilection for the fingers and toes.) Hum Pathol. 2001;32:704-714.
2. Hollmann TJ, Bovée JV, Fletcher CD. Digital fibromyxoma (superficial acral fibromyxoma): a detailed characterization of 124 cases. Am J Surg Pathol. 2012;36:789-798.
3. Varikatt W, Soper J, Simmon G, et al. Superficial acral fibromyxoma: a report of two cases with radiological findings. Skeletal Radiol. 2008;37:499-503.
4. Oteo-Alvaro A, Meizoso T, Scarpellini A, et al. Superficial acral fibromyxoma of the toe, with erosion of the distal phalanx. a clinical report. Arch Orthop Trauma Surg. 2008;128:271-274.
A generally healthy 30-year-old man presented with a 3-cm exophytic, yellowish red, subungual nodule of the left great toe of 1 year’s duration that was obliterating the nail plate. Ten years prior, a similar nodule in the same location was removed via laser by a podiatrist. Medical records were not retrievable, but the patient reported that he was told the excised lesion was a benign tumor. Plain radiographs were performed at the current presentation and demonstrated an inferior cortical lucency of the distal phalanx as well as a lucency over the nail bed region with extension of calcification to the soft tissues. Magnetic resonance imaging showed a mass with a proximal to distal maximum dimension of 2.1 cm that involved the dorsal surface of the proximal phalanx. Magnetic resonance imaging also demonstrated bone erosion from the overlying mass. A 4-mm incisional punch biopsy was performed prior to surgical excision.
Marine ingredients and the skin
Just as we learned early in life that 70% of the human body is composed of water, water covers approximately the same percentage of the earth’s surface. While fishing and harvesting of algae have occurred throughout human history,1 it has only been since the 1970s that widespread scientific interest in the great biological and chemical diversity of the vast oceans of the world has led to investigations into medical and cosmetic applications of the rich life beneath the sea.2 During this period, the marine environment has been found to boast multiple organisms with unique metabolisms adapted for survival in challenging conditions, yielding secondary metabolites, some of which have become valuable in the pharmaceutical and cosmeceutical markets.3,4 Thus, the inclusion of bioactive substances from the sea in drugs and cosmetic products is primarily a recent phenomenon.1 In fact, marine ingredients in cosmetics are thought to confer various benefits to skin health, including antioxidant, anti-acne, anti-wrinkle, and anti-tyrosinase activity.
Chemistry and biologic activity
Several marine microbial natural products have been found to display antimicrobial, antitumor, and anti-inflammatory activity.2,5 And seaweed extracts (green, brown, and red algal compounds that include constituents such as phlorotannins, sulfated polysaccharides, and tyrosinase inhibitors) have been incorporated into cosmeceutical products, with a long history of traditional folk uses for various health – including skin – conditions.3,6,7 Kim and Li reviewed the beneficial health effects of marine fungi-derived terpenoids in 2012, reporting that hundreds of these compounds have been discovered in the last few decades, with many exhibiting anti-inflammatory, anticancer, antimicrobial, and antioxidant activity.8,9 Terpenoids, or isoprenoids, are a subclass of prenyllipids, which include prenylquinones, sterols, and terpenes, the largest class of natural substances.10
The terpenes are the largest group of biologically diverse marine compounds, and include the pseudopterosins, which are structurally discrete active metabolites of the Caribbean gorgonian soft coral Pseudopterogorgia elisabethae, which is native to the waters of the Caribbean Sea, Central Bahamas, Bermuda, the West Indies, and the Florida keys.11,12 The most common gorgonian corals are diterpenes.13 Twenty-six derivatives of the octocoral P. elisabethae (designated PsA-PsZ), also known as the sea whip, sea fan, or sea plume, have been isolated.11,12,14 Pseudopterosins were first isolated in 1986.14,15
Based on the identified biologic activities, particularly anti-inflammatory capacity, of pseudopterosins, researchers have investigated their potential for treatment of various conditions including asthma, cancer, contact dermatitis, dermatoheliosis, HIV, photodamage, psoriasis, and rheumatoid arthritis.1,11
After decades of extensive research of pseudopterosins, these tricyclic diterpene glycosides are thought to provide superior anti-inflammatory and analgesic properties, compared to standard anti-inflammatory treatments, without inducing adverse side effects; they also offer marked antimicrobial and wound-healing effects.3,11,14,16-19
Other marine diterpene glycosides include eleutherobins and fucosides, which also exhibit notable biologic activity.15 In particular, the anti-inflammatory and analgesic activities of pseudopterosins have been found to be concentration- and dose-dependently more potent than the standard-bearing indomethacin.11,14,17
Marine ingredients in topical formulations
The first product to include pseudopterosins was the skin formulation Resilience marketed by Estée Lauder over a decade ago.19,20 Natural marine ingredients have since been incorporated into a few more products, such as Imedeen, an oral skin care preparation that contains Marine Complex.21
In 2012, Rietveld et al. ascertained whether the Marine Complex from Imedeen could variously alter skin morphogenesis in female and male human skin equivalents. Cells were culled from female and male donors between the ages of 30 and 45 years for human skin equivalents that were cultured for 7 or 11 weeks with or without Marine Complex. The investigators found that the number of Ki67-positive epidermal cells was greatly augmented by Marine Complex in female human skin equivalents. The Marine Complex significantly spurred the level of secreted pro-collagen I and elevated the deposition of laminin 332 and collagen type VII in the dermis. Human skin equivalents treated with Marine Complex also exhibited more viable epidermal cell layers and a thicker dermal extracellular matrix, compared to controls, with these effects less salient in male human skin equivalents. The investigators concluded that supplementation with Marine Complex positively stimulated overall human skin equivalent tissue formation, with its effects on the basement membrane and dermal constituents suggestive of potential for use against human skin aging.21
Previously, Xhauflaire-Uhoda et al. evaluated the skin hydrating and firming dose-response effects of cosmetic preparations enriched in algae- and fish collagen–derived substances in randomized controlled double-blind medium-term (12 subjects aged 18-55 years) and short-term (3 subjects over the age of 50) trials. In the short term, serum formulations enriched in marine compounds manifested a superior moisturizing effect on the forearm compared with creams. In later stages, cream formulations were more active, especially after repeated applications. Investigators observed a sustained firming activity in association with both the lotion and cream during treatment, but such results did not persist after treatment was stopped.22
Product development
Technological advances, including sampling strategies, nanoscale nuclear magnetic resonance for structure determination, total chemical synthesis, fermentation, exploration of genomic and metagenomic resources, combinatorial biosynthesis, synthetic biology, and biotechnology represent important ways in which novel marine natural products are being developed, according to several authors.1,2,4
Conclusion
Marine ingredients are a relatively new and fascinating category of substances that can and are being harnessed for pharmaceutical, cosmeceutical, cosmetic, and nutritional uses. Beyond the challenges of obtaining sufficient raw materials and producing effective formulations, the continued viability of such resources may be threatened by human exploitation of the seas and climate change. That said, the oceans offer the greatest biodiversity on the planet and dermatologic preparations derived from such sources present intriguing possibilities, particularly the apparent anti-inflammatory activity of gorgonian and other terpenes. These compounds appear to have the potential to replace, or serve as desirable alternatives to, conventional therapies for inflammatory skin disorders.
References
1. Biotechnol Adv. 2011;29(5):468-82.
2. Mar Drugs. 2013;11(3):700-17.
3. Mar Drugs. 2014;12(2):1066-101.
4. Future Med Chem. 2011;3(12):1475-89.
5. Org Lett. 2000;2(4):507-10.
6. Mar Drugs. 2013;11(1):146-64.
7. J Cosmet Dermatol. 2014;13(1):56-67.
8. Adv Food Nutr Res. 2012;65:409-13.
9. Crit Rev Microbiol. 2011;37(3):245-9.
10. Nat Chem Biol. 2007;3(7):408-14.
11. J Drugs Dermatol. 2013;12(10):1177-9.
12. J Ind Microbiol Biotechnol. 2006;33(7):532-8.
13. Nat Prod Rep. 2009;26(5):681-710.
14. Proc Natl Acad Sci USA. 1986;83(17):6238-40.
15. Bioorg Med Chem. 2011;19(22):6702-19.
16. Arch Biochem Biophys. 2004;424(1):97-104.
17. Asia Pac J Clin Nutr. 2006;15(2):143-52.
18. J Nat Prod. 2004;67(10):1672-80.
19. Mar Drugs. 2004 May;2:73-82.
20. J Nat Prod. 2004;67(8):1216-38.
21. J Cosmet Dermatol. 2012;11(3):213-22.
22. Int J Cosmet Sci. 2008;30(2):131-8.
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in the Design District in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote the textbook, “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002), and a book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). She has contributed to the Cosmeceutical Critique column in Dermatology News since January 2001. Her latest book, “Cosmeceuticals and Cosmetic Ingredients,” was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Evolus, Galderma, GlaxoSmithKline, Kythera Biopharmaceuticals, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Topix Pharmaceuticals, and Unilever.
Just as we learned early in life that 70% of the human body is composed of water, water covers approximately the same percentage of the earth’s surface. While fishing and harvesting of algae have occurred throughout human history,1 it has only been since the 1970s that widespread scientific interest in the great biological and chemical diversity of the vast oceans of the world has led to investigations into medical and cosmetic applications of the rich life beneath the sea.2 During this period, the marine environment has been found to boast multiple organisms with unique metabolisms adapted for survival in challenging conditions, yielding secondary metabolites, some of which have become valuable in the pharmaceutical and cosmeceutical markets.3,4 Thus, the inclusion of bioactive substances from the sea in drugs and cosmetic products is primarily a recent phenomenon.1 In fact, marine ingredients in cosmetics are thought to confer various benefits to skin health, including antioxidant, anti-acne, anti-wrinkle, and anti-tyrosinase activity.
Chemistry and biologic activity
Several marine microbial natural products have been found to display antimicrobial, antitumor, and anti-inflammatory activity.2,5 And seaweed extracts (green, brown, and red algal compounds that include constituents such as phlorotannins, sulfated polysaccharides, and tyrosinase inhibitors) have been incorporated into cosmeceutical products, with a long history of traditional folk uses for various health – including skin – conditions.3,6,7 Kim and Li reviewed the beneficial health effects of marine fungi-derived terpenoids in 2012, reporting that hundreds of these compounds have been discovered in the last few decades, with many exhibiting anti-inflammatory, anticancer, antimicrobial, and antioxidant activity.8,9 Terpenoids, or isoprenoids, are a subclass of prenyllipids, which include prenylquinones, sterols, and terpenes, the largest class of natural substances.10
The terpenes are the largest group of biologically diverse marine compounds, and include the pseudopterosins, which are structurally discrete active metabolites of the Caribbean gorgonian soft coral Pseudopterogorgia elisabethae, which is native to the waters of the Caribbean Sea, Central Bahamas, Bermuda, the West Indies, and the Florida keys.11,12 The most common gorgonian corals are diterpenes.13 Twenty-six derivatives of the octocoral P. elisabethae (designated PsA-PsZ), also known as the sea whip, sea fan, or sea plume, have been isolated.11,12,14 Pseudopterosins were first isolated in 1986.14,15
Based on the identified biologic activities, particularly anti-inflammatory capacity, of pseudopterosins, researchers have investigated their potential for treatment of various conditions including asthma, cancer, contact dermatitis, dermatoheliosis, HIV, photodamage, psoriasis, and rheumatoid arthritis.1,11
After decades of extensive research of pseudopterosins, these tricyclic diterpene glycosides are thought to provide superior anti-inflammatory and analgesic properties, compared to standard anti-inflammatory treatments, without inducing adverse side effects; they also offer marked antimicrobial and wound-healing effects.3,11,14,16-19
Other marine diterpene glycosides include eleutherobins and fucosides, which also exhibit notable biologic activity.15 In particular, the anti-inflammatory and analgesic activities of pseudopterosins have been found to be concentration- and dose-dependently more potent than the standard-bearing indomethacin.11,14,17
Marine ingredients in topical formulations
The first product to include pseudopterosins was the skin formulation Resilience marketed by Estée Lauder over a decade ago.19,20 Natural marine ingredients have since been incorporated into a few more products, such as Imedeen, an oral skin care preparation that contains Marine Complex.21
In 2012, Rietveld et al. ascertained whether the Marine Complex from Imedeen could variously alter skin morphogenesis in female and male human skin equivalents. Cells were culled from female and male donors between the ages of 30 and 45 years for human skin equivalents that were cultured for 7 or 11 weeks with or without Marine Complex. The investigators found that the number of Ki67-positive epidermal cells was greatly augmented by Marine Complex in female human skin equivalents. The Marine Complex significantly spurred the level of secreted pro-collagen I and elevated the deposition of laminin 332 and collagen type VII in the dermis. Human skin equivalents treated with Marine Complex also exhibited more viable epidermal cell layers and a thicker dermal extracellular matrix, compared to controls, with these effects less salient in male human skin equivalents. The investigators concluded that supplementation with Marine Complex positively stimulated overall human skin equivalent tissue formation, with its effects on the basement membrane and dermal constituents suggestive of potential for use against human skin aging.21
Previously, Xhauflaire-Uhoda et al. evaluated the skin hydrating and firming dose-response effects of cosmetic preparations enriched in algae- and fish collagen–derived substances in randomized controlled double-blind medium-term (12 subjects aged 18-55 years) and short-term (3 subjects over the age of 50) trials. In the short term, serum formulations enriched in marine compounds manifested a superior moisturizing effect on the forearm compared with creams. In later stages, cream formulations were more active, especially after repeated applications. Investigators observed a sustained firming activity in association with both the lotion and cream during treatment, but such results did not persist after treatment was stopped.22
Product development
Technological advances, including sampling strategies, nanoscale nuclear magnetic resonance for structure determination, total chemical synthesis, fermentation, exploration of genomic and metagenomic resources, combinatorial biosynthesis, synthetic biology, and biotechnology represent important ways in which novel marine natural products are being developed, according to several authors.1,2,4
Conclusion
Marine ingredients are a relatively new and fascinating category of substances that can and are being harnessed for pharmaceutical, cosmeceutical, cosmetic, and nutritional uses. Beyond the challenges of obtaining sufficient raw materials and producing effective formulations, the continued viability of such resources may be threatened by human exploitation of the seas and climate change. That said, the oceans offer the greatest biodiversity on the planet and dermatologic preparations derived from such sources present intriguing possibilities, particularly the apparent anti-inflammatory activity of gorgonian and other terpenes. These compounds appear to have the potential to replace, or serve as desirable alternatives to, conventional therapies for inflammatory skin disorders.
References
1. Biotechnol Adv. 2011;29(5):468-82.
2. Mar Drugs. 2013;11(3):700-17.
3. Mar Drugs. 2014;12(2):1066-101.
4. Future Med Chem. 2011;3(12):1475-89.
5. Org Lett. 2000;2(4):507-10.
6. Mar Drugs. 2013;11(1):146-64.
7. J Cosmet Dermatol. 2014;13(1):56-67.
8. Adv Food Nutr Res. 2012;65:409-13.
9. Crit Rev Microbiol. 2011;37(3):245-9.
10. Nat Chem Biol. 2007;3(7):408-14.
11. J Drugs Dermatol. 2013;12(10):1177-9.
12. J Ind Microbiol Biotechnol. 2006;33(7):532-8.
13. Nat Prod Rep. 2009;26(5):681-710.
14. Proc Natl Acad Sci USA. 1986;83(17):6238-40.
15. Bioorg Med Chem. 2011;19(22):6702-19.
16. Arch Biochem Biophys. 2004;424(1):97-104.
17. Asia Pac J Clin Nutr. 2006;15(2):143-52.
18. J Nat Prod. 2004;67(10):1672-80.
19. Mar Drugs. 2004 May;2:73-82.
20. J Nat Prod. 2004;67(8):1216-38.
21. J Cosmet Dermatol. 2012;11(3):213-22.
22. Int J Cosmet Sci. 2008;30(2):131-8.
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in the Design District in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote the textbook, “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002), and a book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). She has contributed to the Cosmeceutical Critique column in Dermatology News since January 2001. Her latest book, “Cosmeceuticals and Cosmetic Ingredients,” was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Evolus, Galderma, GlaxoSmithKline, Kythera Biopharmaceuticals, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Topix Pharmaceuticals, and Unilever.
Just as we learned early in life that 70% of the human body is composed of water, water covers approximately the same percentage of the earth’s surface. While fishing and harvesting of algae have occurred throughout human history,1 it has only been since the 1970s that widespread scientific interest in the great biological and chemical diversity of the vast oceans of the world has led to investigations into medical and cosmetic applications of the rich life beneath the sea.2 During this period, the marine environment has been found to boast multiple organisms with unique metabolisms adapted for survival in challenging conditions, yielding secondary metabolites, some of which have become valuable in the pharmaceutical and cosmeceutical markets.3,4 Thus, the inclusion of bioactive substances from the sea in drugs and cosmetic products is primarily a recent phenomenon.1 In fact, marine ingredients in cosmetics are thought to confer various benefits to skin health, including antioxidant, anti-acne, anti-wrinkle, and anti-tyrosinase activity.
Chemistry and biologic activity
Several marine microbial natural products have been found to display antimicrobial, antitumor, and anti-inflammatory activity.2,5 And seaweed extracts (green, brown, and red algal compounds that include constituents such as phlorotannins, sulfated polysaccharides, and tyrosinase inhibitors) have been incorporated into cosmeceutical products, with a long history of traditional folk uses for various health – including skin – conditions.3,6,7 Kim and Li reviewed the beneficial health effects of marine fungi-derived terpenoids in 2012, reporting that hundreds of these compounds have been discovered in the last few decades, with many exhibiting anti-inflammatory, anticancer, antimicrobial, and antioxidant activity.8,9 Terpenoids, or isoprenoids, are a subclass of prenyllipids, which include prenylquinones, sterols, and terpenes, the largest class of natural substances.10
The terpenes are the largest group of biologically diverse marine compounds, and include the pseudopterosins, which are structurally discrete active metabolites of the Caribbean gorgonian soft coral Pseudopterogorgia elisabethae, which is native to the waters of the Caribbean Sea, Central Bahamas, Bermuda, the West Indies, and the Florida keys.11,12 The most common gorgonian corals are diterpenes.13 Twenty-six derivatives of the octocoral P. elisabethae (designated PsA-PsZ), also known as the sea whip, sea fan, or sea plume, have been isolated.11,12,14 Pseudopterosins were first isolated in 1986.14,15
Based on the identified biologic activities, particularly anti-inflammatory capacity, of pseudopterosins, researchers have investigated their potential for treatment of various conditions including asthma, cancer, contact dermatitis, dermatoheliosis, HIV, photodamage, psoriasis, and rheumatoid arthritis.1,11
After decades of extensive research of pseudopterosins, these tricyclic diterpene glycosides are thought to provide superior anti-inflammatory and analgesic properties, compared to standard anti-inflammatory treatments, without inducing adverse side effects; they also offer marked antimicrobial and wound-healing effects.3,11,14,16-19
Other marine diterpene glycosides include eleutherobins and fucosides, which also exhibit notable biologic activity.15 In particular, the anti-inflammatory and analgesic activities of pseudopterosins have been found to be concentration- and dose-dependently more potent than the standard-bearing indomethacin.11,14,17
Marine ingredients in topical formulations
The first product to include pseudopterosins was the skin formulation Resilience marketed by Estée Lauder over a decade ago.19,20 Natural marine ingredients have since been incorporated into a few more products, such as Imedeen, an oral skin care preparation that contains Marine Complex.21
In 2012, Rietveld et al. ascertained whether the Marine Complex from Imedeen could variously alter skin morphogenesis in female and male human skin equivalents. Cells were culled from female and male donors between the ages of 30 and 45 years for human skin equivalents that were cultured for 7 or 11 weeks with or without Marine Complex. The investigators found that the number of Ki67-positive epidermal cells was greatly augmented by Marine Complex in female human skin equivalents. The Marine Complex significantly spurred the level of secreted pro-collagen I and elevated the deposition of laminin 332 and collagen type VII in the dermis. Human skin equivalents treated with Marine Complex also exhibited more viable epidermal cell layers and a thicker dermal extracellular matrix, compared to controls, with these effects less salient in male human skin equivalents. The investigators concluded that supplementation with Marine Complex positively stimulated overall human skin equivalent tissue formation, with its effects on the basement membrane and dermal constituents suggestive of potential for use against human skin aging.21
Previously, Xhauflaire-Uhoda et al. evaluated the skin hydrating and firming dose-response effects of cosmetic preparations enriched in algae- and fish collagen–derived substances in randomized controlled double-blind medium-term (12 subjects aged 18-55 years) and short-term (3 subjects over the age of 50) trials. In the short term, serum formulations enriched in marine compounds manifested a superior moisturizing effect on the forearm compared with creams. In later stages, cream formulations were more active, especially after repeated applications. Investigators observed a sustained firming activity in association with both the lotion and cream during treatment, but such results did not persist after treatment was stopped.22
Product development
Technological advances, including sampling strategies, nanoscale nuclear magnetic resonance for structure determination, total chemical synthesis, fermentation, exploration of genomic and metagenomic resources, combinatorial biosynthesis, synthetic biology, and biotechnology represent important ways in which novel marine natural products are being developed, according to several authors.1,2,4
Conclusion
Marine ingredients are a relatively new and fascinating category of substances that can and are being harnessed for pharmaceutical, cosmeceutical, cosmetic, and nutritional uses. Beyond the challenges of obtaining sufficient raw materials and producing effective formulations, the continued viability of such resources may be threatened by human exploitation of the seas and climate change. That said, the oceans offer the greatest biodiversity on the planet and dermatologic preparations derived from such sources present intriguing possibilities, particularly the apparent anti-inflammatory activity of gorgonian and other terpenes. These compounds appear to have the potential to replace, or serve as desirable alternatives to, conventional therapies for inflammatory skin disorders.
References
1. Biotechnol Adv. 2011;29(5):468-82.
2. Mar Drugs. 2013;11(3):700-17.
3. Mar Drugs. 2014;12(2):1066-101.
4. Future Med Chem. 2011;3(12):1475-89.
5. Org Lett. 2000;2(4):507-10.
6. Mar Drugs. 2013;11(1):146-64.
7. J Cosmet Dermatol. 2014;13(1):56-67.
8. Adv Food Nutr Res. 2012;65:409-13.
9. Crit Rev Microbiol. 2011;37(3):245-9.
10. Nat Chem Biol. 2007;3(7):408-14.
11. J Drugs Dermatol. 2013;12(10):1177-9.
12. J Ind Microbiol Biotechnol. 2006;33(7):532-8.
13. Nat Prod Rep. 2009;26(5):681-710.
14. Proc Natl Acad Sci USA. 1986;83(17):6238-40.
15. Bioorg Med Chem. 2011;19(22):6702-19.
16. Arch Biochem Biophys. 2004;424(1):97-104.
17. Asia Pac J Clin Nutr. 2006;15(2):143-52.
18. J Nat Prod. 2004;67(10):1672-80.
19. Mar Drugs. 2004 May;2:73-82.
20. J Nat Prod. 2004;67(8):1216-38.
21. J Cosmet Dermatol. 2012;11(3):213-22.
22. Int J Cosmet Sci. 2008;30(2):131-8.
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in the Design District in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote the textbook, “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002), and a book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). She has contributed to the Cosmeceutical Critique column in Dermatology News since January 2001. Her latest book, “Cosmeceuticals and Cosmetic Ingredients,” was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Evolus, Galderma, GlaxoSmithKline, Kythera Biopharmaceuticals, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Topix Pharmaceuticals, and Unilever.
Platelet-Rich Plasma for Androgenetic Alopecia
Many male and female patients seek therapy for androgenetic alopecia (AGA) at some point. Results are rarely achieved with single-modality therapy. In fact, the treatment of AGA often requires a combination approach of systemic medications and supplements; topical therapy; and if indicated, some type of minimally invasive procedure or surgical option.
The use of platelet-rich plasma (PRP) to pretreat follicular unit hair grafts may increase hair growth and density. Although the method by which PRP helps promote hair growth remains a source of speculation, it is known that platelets contain many growth factors in the platelet α granules. When these granules become activated, they release many types of growth factors ranging from vascular endothelial growth factor, transforming growth factor ß, epidermal growth factor, platelet-derived growth factor, and insulinlike growth factor.
Injection or topical application of PRP has become a popular treatment for skin rejuvenation, alopecia, and autologous fat grafting; however, many of these treatments are being done without the studies to support them.
Alves and Grimalt (Dermatol Surg. 2016;42:491-497) conducted a prospective, split-scalp, placebo-controlled study evaluating the use of PRP injections in the treatment of AGA in 25 patients (12 men, 13 women) with a mean age of 39 years (age range, 21–62 years). Photographs were taken and 4 areas per split scalp were marked with red tattoo dots. The patients were randomized to receive PRP on 1 side and saline injections on the other side. Patients were blinded, but the physician performing the treatment was not blinded. Treatments were performed monthly for 3 sessions. Follow-up photographs and hair density counts were performed at 3 and 6 months after treatment.
Results showed that after 6 months there was a statistically significant (P<.05) increase in the total hair density and terminal hair counts on the PRP-treated side compared to baseline and to the placebo-treated side. When broken down by demographics, patients who did best were men 40 years or younger with AGA beginning at 25 years or older, a positive family history of AGA, and AGA duration of greater than 10 years.
What’s the issue?
The results of this study suggest that PRP has a mild to modest effect (approximately 10% increase) on increasing hair density in a specific patient population. This split-scalp, placebo-controlled study is one of the few of its kind in the field of PRP research and the authors should be applauded for the design of this study. However, it would be beneficial to have a 1-year follow-up to see if the results are maintained and to better help determine if maintenance injections would be needed to sustain the results.
The field of hair restoration and regeneration will continue to grow as newer technologies in hair stem cell injections come to the forefront as well. Rigorous studies will be needed to better help physicians and patients make the right treatment decisions.
Have patients been asking you about PRP for skin rejuvenation and hair restoration?
Many male and female patients seek therapy for androgenetic alopecia (AGA) at some point. Results are rarely achieved with single-modality therapy. In fact, the treatment of AGA often requires a combination approach of systemic medications and supplements; topical therapy; and if indicated, some type of minimally invasive procedure or surgical option.
The use of platelet-rich plasma (PRP) to pretreat follicular unit hair grafts may increase hair growth and density. Although the method by which PRP helps promote hair growth remains a source of speculation, it is known that platelets contain many growth factors in the platelet α granules. When these granules become activated, they release many types of growth factors ranging from vascular endothelial growth factor, transforming growth factor ß, epidermal growth factor, platelet-derived growth factor, and insulinlike growth factor.
Injection or topical application of PRP has become a popular treatment for skin rejuvenation, alopecia, and autologous fat grafting; however, many of these treatments are being done without the studies to support them.
Alves and Grimalt (Dermatol Surg. 2016;42:491-497) conducted a prospective, split-scalp, placebo-controlled study evaluating the use of PRP injections in the treatment of AGA in 25 patients (12 men, 13 women) with a mean age of 39 years (age range, 21–62 years). Photographs were taken and 4 areas per split scalp were marked with red tattoo dots. The patients were randomized to receive PRP on 1 side and saline injections on the other side. Patients were blinded, but the physician performing the treatment was not blinded. Treatments were performed monthly for 3 sessions. Follow-up photographs and hair density counts were performed at 3 and 6 months after treatment.
Results showed that after 6 months there was a statistically significant (P<.05) increase in the total hair density and terminal hair counts on the PRP-treated side compared to baseline and to the placebo-treated side. When broken down by demographics, patients who did best were men 40 years or younger with AGA beginning at 25 years or older, a positive family history of AGA, and AGA duration of greater than 10 years.
What’s the issue?
The results of this study suggest that PRP has a mild to modest effect (approximately 10% increase) on increasing hair density in a specific patient population. This split-scalp, placebo-controlled study is one of the few of its kind in the field of PRP research and the authors should be applauded for the design of this study. However, it would be beneficial to have a 1-year follow-up to see if the results are maintained and to better help determine if maintenance injections would be needed to sustain the results.
The field of hair restoration and regeneration will continue to grow as newer technologies in hair stem cell injections come to the forefront as well. Rigorous studies will be needed to better help physicians and patients make the right treatment decisions.
Have patients been asking you about PRP for skin rejuvenation and hair restoration?
Many male and female patients seek therapy for androgenetic alopecia (AGA) at some point. Results are rarely achieved with single-modality therapy. In fact, the treatment of AGA often requires a combination approach of systemic medications and supplements; topical therapy; and if indicated, some type of minimally invasive procedure or surgical option.
The use of platelet-rich plasma (PRP) to pretreat follicular unit hair grafts may increase hair growth and density. Although the method by which PRP helps promote hair growth remains a source of speculation, it is known that platelets contain many growth factors in the platelet α granules. When these granules become activated, they release many types of growth factors ranging from vascular endothelial growth factor, transforming growth factor ß, epidermal growth factor, platelet-derived growth factor, and insulinlike growth factor.
Injection or topical application of PRP has become a popular treatment for skin rejuvenation, alopecia, and autologous fat grafting; however, many of these treatments are being done without the studies to support them.
Alves and Grimalt (Dermatol Surg. 2016;42:491-497) conducted a prospective, split-scalp, placebo-controlled study evaluating the use of PRP injections in the treatment of AGA in 25 patients (12 men, 13 women) with a mean age of 39 years (age range, 21–62 years). Photographs were taken and 4 areas per split scalp were marked with red tattoo dots. The patients were randomized to receive PRP on 1 side and saline injections on the other side. Patients were blinded, but the physician performing the treatment was not blinded. Treatments were performed monthly for 3 sessions. Follow-up photographs and hair density counts were performed at 3 and 6 months after treatment.
Results showed that after 6 months there was a statistically significant (P<.05) increase in the total hair density and terminal hair counts on the PRP-treated side compared to baseline and to the placebo-treated side. When broken down by demographics, patients who did best were men 40 years or younger with AGA beginning at 25 years or older, a positive family history of AGA, and AGA duration of greater than 10 years.
What’s the issue?
The results of this study suggest that PRP has a mild to modest effect (approximately 10% increase) on increasing hair density in a specific patient population. This split-scalp, placebo-controlled study is one of the few of its kind in the field of PRP research and the authors should be applauded for the design of this study. However, it would be beneficial to have a 1-year follow-up to see if the results are maintained and to better help determine if maintenance injections would be needed to sustain the results.
The field of hair restoration and regeneration will continue to grow as newer technologies in hair stem cell injections come to the forefront as well. Rigorous studies will be needed to better help physicians and patients make the right treatment decisions.
Have patients been asking you about PRP for skin rejuvenation and hair restoration?
Cosmetic Corner: Dermatologists Weigh in on Scar Treatments
To improve patient care and outcomes, leading dermatologists offered their recommendations on scar treatments. Consideration must be given to:
- bioCorneum+
- Mederma Scar Cream Plus SPF 30
- Organic vitamin E oil
- ScarAway Silicone Scar Sheets
- Scar Recovery Gel with Centelline
Cutis invites readers to send us their recommendations. Body scrubs, OTC acne treatments, self-tanners, and cleansing devices will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
To improve patient care and outcomes, leading dermatologists offered their recommendations on scar treatments. Consideration must be given to:
- bioCorneum+
- Mederma Scar Cream Plus SPF 30
- Organic vitamin E oil
- ScarAway Silicone Scar Sheets
- Scar Recovery Gel with Centelline
Cutis invites readers to send us their recommendations. Body scrubs, OTC acne treatments, self-tanners, and cleansing devices will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
To improve patient care and outcomes, leading dermatologists offered their recommendations on scar treatments. Consideration must be given to:
- bioCorneum+
- Mederma Scar Cream Plus SPF 30
- Organic vitamin E oil
- ScarAway Silicone Scar Sheets
- Scar Recovery Gel with Centelline
Cutis invites readers to send us their recommendations. Body scrubs, OTC acne treatments, self-tanners, and cleansing devices will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
Experts say topical retinoid still a winner for facial rejuvenation
WAIKOLOA, HAWAII – Have topical retinoids fallen by the wayside for treatment of facial wrinkles in this era of cosmeceuticals, spa facilities, lasers, and injectables?
Not by a long shot, a panel of experts in aesthetic dermatology agreed at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
“I think there’s an ethical issue here. When my patients come in for an assessment, I feel bound to say, ‘Look, we can do all these fancy procedures, lasers, and so forth, but if you use Retin-A for 6 months you’re going to see a nice improvement, so maybe you should try that first.’ It’s quite common that I’ll do that, although I prefer Tazorac [tazarotene] for a number of reasons,” said Dr. Christopher B. Zachary, professor and chair of the department of dermatology at the University of California, Irvine.
That being said, he added a caveat: “I couldn’t use a topical because I just don’t have it in me to put something on my face every night, but there are people who want that. I prefer a quick fix, like a laser.”
Dr. Suzanne L. Kilmer, director of the Laser and Skin Surgery Center of Northern California, Sacramento, declared, “I tell everybody, ‘Number one, I want you to block the sun. I believe only in the zinc oxide sunscreens because I want to block all the way up to 400 nm.’ ” And number two, she says that tretinoin and tazarotene are by far the most studied, and that “ ‘there are lots of great data that it helps not only with acne and wrinkles, but dysplastic nevi, actinic keratoses – it’s definitely a normalizer for your skin. You’ll get great results. And if you’re using that you’re probably going to get better results with any treatments I do.’ ”
Dr. Michael S. Kaminer, a partner in SkinCare Physicians in Chestnut Hill, Ma., said one thing that hasn’t changed over the years is that quite a few patients – especially those who are fair-skinned – can’t tolerate a topical retinoid on the face. They find it too irritating.
“I think it’s a lot of people. They just have a hard time with it,” observed Dr. Kaminer, also of Yale University in New Haven, Conn.
Asked if patients can get results comparable to Retin-A using OTC topical cosmeceuticals, Dr. Brooke C. Sikora, a dermatologist who also practices at SkinCare Physicians, said it’s tough to say because the studies that have been done were cosmeceutical-industry sponsored.
“Some of the industry-sponsored studies claim and show evidence that with some of the retinol products they can get near-tretinoin results. But you have to take that with a grain of salt,” she advised.
All of the speakers reported receiving grant support from and serving as consultants to various pharmaceutical companies. SDEF and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – Have topical retinoids fallen by the wayside for treatment of facial wrinkles in this era of cosmeceuticals, spa facilities, lasers, and injectables?
Not by a long shot, a panel of experts in aesthetic dermatology agreed at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
“I think there’s an ethical issue here. When my patients come in for an assessment, I feel bound to say, ‘Look, we can do all these fancy procedures, lasers, and so forth, but if you use Retin-A for 6 months you’re going to see a nice improvement, so maybe you should try that first.’ It’s quite common that I’ll do that, although I prefer Tazorac [tazarotene] for a number of reasons,” said Dr. Christopher B. Zachary, professor and chair of the department of dermatology at the University of California, Irvine.
That being said, he added a caveat: “I couldn’t use a topical because I just don’t have it in me to put something on my face every night, but there are people who want that. I prefer a quick fix, like a laser.”
Dr. Suzanne L. Kilmer, director of the Laser and Skin Surgery Center of Northern California, Sacramento, declared, “I tell everybody, ‘Number one, I want you to block the sun. I believe only in the zinc oxide sunscreens because I want to block all the way up to 400 nm.’ ” And number two, she says that tretinoin and tazarotene are by far the most studied, and that “ ‘there are lots of great data that it helps not only with acne and wrinkles, but dysplastic nevi, actinic keratoses – it’s definitely a normalizer for your skin. You’ll get great results. And if you’re using that you’re probably going to get better results with any treatments I do.’ ”
Dr. Michael S. Kaminer, a partner in SkinCare Physicians in Chestnut Hill, Ma., said one thing that hasn’t changed over the years is that quite a few patients – especially those who are fair-skinned – can’t tolerate a topical retinoid on the face. They find it too irritating.
“I think it’s a lot of people. They just have a hard time with it,” observed Dr. Kaminer, also of Yale University in New Haven, Conn.
Asked if patients can get results comparable to Retin-A using OTC topical cosmeceuticals, Dr. Brooke C. Sikora, a dermatologist who also practices at SkinCare Physicians, said it’s tough to say because the studies that have been done were cosmeceutical-industry sponsored.
“Some of the industry-sponsored studies claim and show evidence that with some of the retinol products they can get near-tretinoin results. But you have to take that with a grain of salt,” she advised.
All of the speakers reported receiving grant support from and serving as consultants to various pharmaceutical companies. SDEF and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – Have topical retinoids fallen by the wayside for treatment of facial wrinkles in this era of cosmeceuticals, spa facilities, lasers, and injectables?
Not by a long shot, a panel of experts in aesthetic dermatology agreed at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
“I think there’s an ethical issue here. When my patients come in for an assessment, I feel bound to say, ‘Look, we can do all these fancy procedures, lasers, and so forth, but if you use Retin-A for 6 months you’re going to see a nice improvement, so maybe you should try that first.’ It’s quite common that I’ll do that, although I prefer Tazorac [tazarotene] for a number of reasons,” said Dr. Christopher B. Zachary, professor and chair of the department of dermatology at the University of California, Irvine.
That being said, he added a caveat: “I couldn’t use a topical because I just don’t have it in me to put something on my face every night, but there are people who want that. I prefer a quick fix, like a laser.”
Dr. Suzanne L. Kilmer, director of the Laser and Skin Surgery Center of Northern California, Sacramento, declared, “I tell everybody, ‘Number one, I want you to block the sun. I believe only in the zinc oxide sunscreens because I want to block all the way up to 400 nm.’ ” And number two, she says that tretinoin and tazarotene are by far the most studied, and that “ ‘there are lots of great data that it helps not only with acne and wrinkles, but dysplastic nevi, actinic keratoses – it’s definitely a normalizer for your skin. You’ll get great results. And if you’re using that you’re probably going to get better results with any treatments I do.’ ”
Dr. Michael S. Kaminer, a partner in SkinCare Physicians in Chestnut Hill, Ma., said one thing that hasn’t changed over the years is that quite a few patients – especially those who are fair-skinned – can’t tolerate a topical retinoid on the face. They find it too irritating.
“I think it’s a lot of people. They just have a hard time with it,” observed Dr. Kaminer, also of Yale University in New Haven, Conn.
Asked if patients can get results comparable to Retin-A using OTC topical cosmeceuticals, Dr. Brooke C. Sikora, a dermatologist who also practices at SkinCare Physicians, said it’s tough to say because the studies that have been done were cosmeceutical-industry sponsored.
“Some of the industry-sponsored studies claim and show evidence that with some of the retinol products they can get near-tretinoin results. But you have to take that with a grain of salt,” she advised.
All of the speakers reported receiving grant support from and serving as consultants to various pharmaceutical companies. SDEF and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF HAWAII DERMATOLOGY SEMINAR