User login
How to use cryolipolysis for reduction of double chin
WAIKOLOA, HAWAII – Patient interest is soaring in fat cell reduction via cryolipolysis using the noninvasive CoolSculpting technology, Dr. Suzanne L. Kilmer said at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
She was one of the developers of the “treatment to transformation” (T2T) approach to CoolSculpting, in which larger areas of fat deposits are safely treated per session than in the earliest days of the therapy’s availability.
“Treatment to transformation has been a home run. Our use of this approach has increased greatly in our practice because a lot of people don’t want to do liposuction, which has more risk and downtime. We’ve purchased a second device to accommodate patients and treat two areas at once, said Dr. Kilmer, director of the Laser and Skin Surgery Center of Northern California in Sacramento.
She was lead author of the pivotal trial which led to Food and Drug Administration clearance of Zeltiq Aesthetics’ CoolSculpting for treatment of submental fat. That’s the fifth body area for which FDA clearance has been obtained on the basis of solid evidence of safety and efficacy, the others being the inner thighs, outer thighs, flanks or love handles, and abdomen.
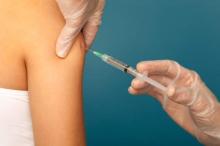
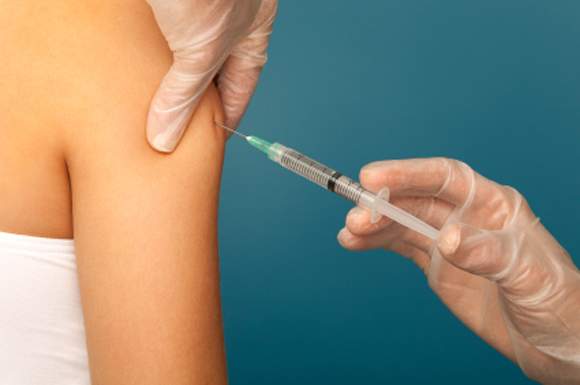
The pivotal trial included 60 patients who underwent CoolSculpting for a double chin, or submental fullness, using a prototype of the device known as the CoolMini submental applicator, a small-volume vacuum delivery cup. The patient’s double chin was placed in the applicator, then a 60-minute–long treatment cycle was delivered to that area at –10° C. An optional second treatment could be delivered 6 weeks later.
There were no procedure-related serious adverse events. Ultrasound assessment carried out 12 weeks after the final cryolipolysis treatment showed a mean 2-mm reduction in fat layer thickness. Eighty-three percent of patients declared themselves satisfied, 77% reported visible fat reduction, 77% reported that they felt their appearance had improved, 76% found the procedure comfortable, and 80% indicated they would recommend submental cryolipolysis to a friend (Lasers Surg Med. 2016 Jan;48[1]:3-13).
Dr. Kilmer noted that cryolipolysis has been cleared by the FDA for fat reduction since 2010. It works by inducing a delayed slow death of fat cells via apoptosis. There is no immediate effect and the noninvasive procedure involves no anesthetics. There is very little inflammation, discomfort, or downtime for recovery. The typical result is roughly a 20% loss in fat cells in treated areas.
She offered a couple of practical tips regarding cryolipolysis for reduction of submental fat. First, the entire targeted area of submental fat must be able to fit inside the applicator. And after successful cryolipolysis, any residual areas of fat cells will have to be targeted using injections of deoxycholic acid (Kybella), because a small pocket of submental fat won’t get sucked up into the CoolMini applicator. (Deoxycholic acid was approved by the FDA in 2015, for treating moderate to severe submental fat in adults.)
“Actually, we use both treatments together on most people. When they have a lot of fat we start out by killing it with cryolipolysis. Then when it gets smaller we trim it down with deoxycholic acid, often with a neuromodulator to deal with the platysmal bands,” Dr. Kilmer said.
She reported serving on medical advisory boards for Zeltiq and a handful of other dermatologic device companies and receiving research funding from half a dozen companies.
SDEF and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – Patient interest is soaring in fat cell reduction via cryolipolysis using the noninvasive CoolSculpting technology, Dr. Suzanne L. Kilmer said at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
She was one of the developers of the “treatment to transformation” (T2T) approach to CoolSculpting, in which larger areas of fat deposits are safely treated per session than in the earliest days of the therapy’s availability.
“Treatment to transformation has been a home run. Our use of this approach has increased greatly in our practice because a lot of people don’t want to do liposuction, which has more risk and downtime. We’ve purchased a second device to accommodate patients and treat two areas at once, said Dr. Kilmer, director of the Laser and Skin Surgery Center of Northern California in Sacramento.
She was lead author of the pivotal trial which led to Food and Drug Administration clearance of Zeltiq Aesthetics’ CoolSculpting for treatment of submental fat. That’s the fifth body area for which FDA clearance has been obtained on the basis of solid evidence of safety and efficacy, the others being the inner thighs, outer thighs, flanks or love handles, and abdomen.
The pivotal trial included 60 patients who underwent CoolSculpting for a double chin, or submental fullness, using a prototype of the device known as the CoolMini submental applicator, a small-volume vacuum delivery cup. The patient’s double chin was placed in the applicator, then a 60-minute–long treatment cycle was delivered to that area at –10° C. An optional second treatment could be delivered 6 weeks later.
There were no procedure-related serious adverse events. Ultrasound assessment carried out 12 weeks after the final cryolipolysis treatment showed a mean 2-mm reduction in fat layer thickness. Eighty-three percent of patients declared themselves satisfied, 77% reported visible fat reduction, 77% reported that they felt their appearance had improved, 76% found the procedure comfortable, and 80% indicated they would recommend submental cryolipolysis to a friend (Lasers Surg Med. 2016 Jan;48[1]:3-13).
Dr. Kilmer noted that cryolipolysis has been cleared by the FDA for fat reduction since 2010. It works by inducing a delayed slow death of fat cells via apoptosis. There is no immediate effect and the noninvasive procedure involves no anesthetics. There is very little inflammation, discomfort, or downtime for recovery. The typical result is roughly a 20% loss in fat cells in treated areas.
She offered a couple of practical tips regarding cryolipolysis for reduction of submental fat. First, the entire targeted area of submental fat must be able to fit inside the applicator. And after successful cryolipolysis, any residual areas of fat cells will have to be targeted using injections of deoxycholic acid (Kybella), because a small pocket of submental fat won’t get sucked up into the CoolMini applicator. (Deoxycholic acid was approved by the FDA in 2015, for treating moderate to severe submental fat in adults.)
“Actually, we use both treatments together on most people. When they have a lot of fat we start out by killing it with cryolipolysis. Then when it gets smaller we trim it down with deoxycholic acid, often with a neuromodulator to deal with the platysmal bands,” Dr. Kilmer said.
She reported serving on medical advisory boards for Zeltiq and a handful of other dermatologic device companies and receiving research funding from half a dozen companies.
SDEF and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – Patient interest is soaring in fat cell reduction via cryolipolysis using the noninvasive CoolSculpting technology, Dr. Suzanne L. Kilmer said at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
She was one of the developers of the “treatment to transformation” (T2T) approach to CoolSculpting, in which larger areas of fat deposits are safely treated per session than in the earliest days of the therapy’s availability.
“Treatment to transformation has been a home run. Our use of this approach has increased greatly in our practice because a lot of people don’t want to do liposuction, which has more risk and downtime. We’ve purchased a second device to accommodate patients and treat two areas at once, said Dr. Kilmer, director of the Laser and Skin Surgery Center of Northern California in Sacramento.
She was lead author of the pivotal trial which led to Food and Drug Administration clearance of Zeltiq Aesthetics’ CoolSculpting for treatment of submental fat. That’s the fifth body area for which FDA clearance has been obtained on the basis of solid evidence of safety and efficacy, the others being the inner thighs, outer thighs, flanks or love handles, and abdomen.
The pivotal trial included 60 patients who underwent CoolSculpting for a double chin, or submental fullness, using a prototype of the device known as the CoolMini submental applicator, a small-volume vacuum delivery cup. The patient’s double chin was placed in the applicator, then a 60-minute–long treatment cycle was delivered to that area at –10° C. An optional second treatment could be delivered 6 weeks later.
There were no procedure-related serious adverse events. Ultrasound assessment carried out 12 weeks after the final cryolipolysis treatment showed a mean 2-mm reduction in fat layer thickness. Eighty-three percent of patients declared themselves satisfied, 77% reported visible fat reduction, 77% reported that they felt their appearance had improved, 76% found the procedure comfortable, and 80% indicated they would recommend submental cryolipolysis to a friend (Lasers Surg Med. 2016 Jan;48[1]:3-13).
Dr. Kilmer noted that cryolipolysis has been cleared by the FDA for fat reduction since 2010. It works by inducing a delayed slow death of fat cells via apoptosis. There is no immediate effect and the noninvasive procedure involves no anesthetics. There is very little inflammation, discomfort, or downtime for recovery. The typical result is roughly a 20% loss in fat cells in treated areas.
She offered a couple of practical tips regarding cryolipolysis for reduction of submental fat. First, the entire targeted area of submental fat must be able to fit inside the applicator. And after successful cryolipolysis, any residual areas of fat cells will have to be targeted using injections of deoxycholic acid (Kybella), because a small pocket of submental fat won’t get sucked up into the CoolMini applicator. (Deoxycholic acid was approved by the FDA in 2015, for treating moderate to severe submental fat in adults.)
“Actually, we use both treatments together on most people. When they have a lot of fat we start out by killing it with cryolipolysis. Then when it gets smaller we trim it down with deoxycholic acid, often with a neuromodulator to deal with the platysmal bands,” Dr. Kilmer said.
She reported serving on medical advisory boards for Zeltiq and a handful of other dermatologic device companies and receiving research funding from half a dozen companies.
SDEF and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF HAWAII DERMATOLOGY SEMINAR
What’s Trending in OTC Antiaging Products: Report From the AAD Meeting
Patient interest in over-the-counter (OTC) antiaging products is increasing; however, the number of products to choose from can be overwhelming and patients may end up spending a lot of money on products that do not work. Dr. Anthony Rossi provides an overview of the latest trends in OTC antiaging products, including antioxidant-containing sunscreens, growth factors, and topical hyaluronic acid. Dr. Rossi notes that patients often call on dermatologists to offer product recommendations. Therefore, it is important for dermatologists to be familiar with product ingredients and counsel patients accordingly in order to maximize their effects on the skin.
Patient interest in over-the-counter (OTC) antiaging products is increasing; however, the number of products to choose from can be overwhelming and patients may end up spending a lot of money on products that do not work. Dr. Anthony Rossi provides an overview of the latest trends in OTC antiaging products, including antioxidant-containing sunscreens, growth factors, and topical hyaluronic acid. Dr. Rossi notes that patients often call on dermatologists to offer product recommendations. Therefore, it is important for dermatologists to be familiar with product ingredients and counsel patients accordingly in order to maximize their effects on the skin.
Patient interest in over-the-counter (OTC) antiaging products is increasing; however, the number of products to choose from can be overwhelming and patients may end up spending a lot of money on products that do not work. Dr. Anthony Rossi provides an overview of the latest trends in OTC antiaging products, including antioxidant-containing sunscreens, growth factors, and topical hyaluronic acid. Dr. Rossi notes that patients often call on dermatologists to offer product recommendations. Therefore, it is important for dermatologists to be familiar with product ingredients and counsel patients accordingly in order to maximize their effects on the skin.
Aesthetic Dermatology: Eyelash extensions
The obsession with longer, fuller, darker eyelashes has become a mainstay in our culture – initially with the ever growing options of mascaras and glue on eyelashes, and now with options that are longer lasting, including eyelash extensions (semipermanent eyelashes) and topical eyelash growth enhancers (such as bimatoprost).
Eyelash extensions are not the same as glue-on strip or individual lashes bought at the drug store or makeup counter that last 1-2 days. These are silk, mink, or poly nylon synthetic lashes that typically last for approximately four weeks, with refills often required at 2-4 week intervals as the natural eyelash sheds. They are adhered to the person’s natural eyelash via an adhesive bonding process that can take 1-2 hours for initial application. Generally, a single lash is applied to each natural lash.
When applied properly, neither the extension eyelash nor the glue should touch the eyelid. The bond is designed to last until the lashes naturally fall out, although the extensions may fall out faster if one uses oil-based eye makeup remover or rubs the eyes regularly, as oil weakens the bond between the glue and the lash. Eyelash extensions are waterproof and give the appearance of having mascara on without wearing it. In the United States, eyelash extension services can range from $100 to $500 for the initial application, with decreased cost for refills. Lash extensions are waterproof and popular for special occasions and vacations, and even more so now for every day.
Potential adverse effects of eyelash extensions include ocular hyperemia, keratoconjunctivitis, allergic blepharitis, and allergic contact dermatitis in the patient. Keratoconjunctivitis is thought to be due to formaldehyde contained in some of the glues used for application.1 Eyelash extensions have also been associated with occupational allergic contact dermatitis, allergic rhinitis, and occupational asthma in the practitioner applying the eyelash extensions, particularly with the cyanoacrylate-based glues.2,3
In a national survey of eyelash extensions and their health-related problems in Japan, 10% (205) of the respondents had experience with eyelash extensions. Of those women, 27% (55) experienced problems that included ocular hyperemia, pain, and itchy swollen eyelids.4 Conjunctival erosion from the eyelid fixing tape used during application and subconjunctival hemorrhage from compression during removal of the extensions has been also reported.1 Hair breakage and even traction alopecia may occur, especially in patients who accidentally or intentionally pull the extensions off.
If permanent eyelash damage occurs, eyelash transplantation may be required to replace the eyelash, as eyelash growth medications such as bimatoprost may not be effective if the follicle is missing or severely damaged. Eyelash transplants often grow long enough where they require trimming, especially if donor sites are taken from the scalp.5
Eyelash extensions offer a nice alternative to daily use of mascara, temporary glue-on eyelashes, and daily application of topical eyelash growth products. As this procedure has increased in number, the dermatologist may be consulted for recommendations and treatment of any potential adverse events associated with it.
References
1. Cornea. 2012 Feb;31(2):121-5.
2. Contact Dermatitis. 2012 Nov;67(5):307-8.
3. Occup Med (Lond). 2013 Jun;63(4):294-7.
4. Nihon Eiseigaku Zasshi. 2013;68(3):168-74.
5. Plast Reconstr Surg Glob Open. 2015 Apr 7;3(3):e324.
Dr. Wesley and Dr. Talakoub are co-contributors to this column. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Wesley.
The obsession with longer, fuller, darker eyelashes has become a mainstay in our culture – initially with the ever growing options of mascaras and glue on eyelashes, and now with options that are longer lasting, including eyelash extensions (semipermanent eyelashes) and topical eyelash growth enhancers (such as bimatoprost).
Eyelash extensions are not the same as glue-on strip or individual lashes bought at the drug store or makeup counter that last 1-2 days. These are silk, mink, or poly nylon synthetic lashes that typically last for approximately four weeks, with refills often required at 2-4 week intervals as the natural eyelash sheds. They are adhered to the person’s natural eyelash via an adhesive bonding process that can take 1-2 hours for initial application. Generally, a single lash is applied to each natural lash.
When applied properly, neither the extension eyelash nor the glue should touch the eyelid. The bond is designed to last until the lashes naturally fall out, although the extensions may fall out faster if one uses oil-based eye makeup remover or rubs the eyes regularly, as oil weakens the bond between the glue and the lash. Eyelash extensions are waterproof and give the appearance of having mascara on without wearing it. In the United States, eyelash extension services can range from $100 to $500 for the initial application, with decreased cost for refills. Lash extensions are waterproof and popular for special occasions and vacations, and even more so now for every day.
Potential adverse effects of eyelash extensions include ocular hyperemia, keratoconjunctivitis, allergic blepharitis, and allergic contact dermatitis in the patient. Keratoconjunctivitis is thought to be due to formaldehyde contained in some of the glues used for application.1 Eyelash extensions have also been associated with occupational allergic contact dermatitis, allergic rhinitis, and occupational asthma in the practitioner applying the eyelash extensions, particularly with the cyanoacrylate-based glues.2,3
In a national survey of eyelash extensions and their health-related problems in Japan, 10% (205) of the respondents had experience with eyelash extensions. Of those women, 27% (55) experienced problems that included ocular hyperemia, pain, and itchy swollen eyelids.4 Conjunctival erosion from the eyelid fixing tape used during application and subconjunctival hemorrhage from compression during removal of the extensions has been also reported.1 Hair breakage and even traction alopecia may occur, especially in patients who accidentally or intentionally pull the extensions off.
If permanent eyelash damage occurs, eyelash transplantation may be required to replace the eyelash, as eyelash growth medications such as bimatoprost may not be effective if the follicle is missing or severely damaged. Eyelash transplants often grow long enough where they require trimming, especially if donor sites are taken from the scalp.5
Eyelash extensions offer a nice alternative to daily use of mascara, temporary glue-on eyelashes, and daily application of topical eyelash growth products. As this procedure has increased in number, the dermatologist may be consulted for recommendations and treatment of any potential adverse events associated with it.
References
1. Cornea. 2012 Feb;31(2):121-5.
2. Contact Dermatitis. 2012 Nov;67(5):307-8.
3. Occup Med (Lond). 2013 Jun;63(4):294-7.
4. Nihon Eiseigaku Zasshi. 2013;68(3):168-74.
5. Plast Reconstr Surg Glob Open. 2015 Apr 7;3(3):e324.
Dr. Wesley and Dr. Talakoub are co-contributors to this column. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Wesley.
The obsession with longer, fuller, darker eyelashes has become a mainstay in our culture – initially with the ever growing options of mascaras and glue on eyelashes, and now with options that are longer lasting, including eyelash extensions (semipermanent eyelashes) and topical eyelash growth enhancers (such as bimatoprost).
Eyelash extensions are not the same as glue-on strip or individual lashes bought at the drug store or makeup counter that last 1-2 days. These are silk, mink, or poly nylon synthetic lashes that typically last for approximately four weeks, with refills often required at 2-4 week intervals as the natural eyelash sheds. They are adhered to the person’s natural eyelash via an adhesive bonding process that can take 1-2 hours for initial application. Generally, a single lash is applied to each natural lash.
When applied properly, neither the extension eyelash nor the glue should touch the eyelid. The bond is designed to last until the lashes naturally fall out, although the extensions may fall out faster if one uses oil-based eye makeup remover or rubs the eyes regularly, as oil weakens the bond between the glue and the lash. Eyelash extensions are waterproof and give the appearance of having mascara on without wearing it. In the United States, eyelash extension services can range from $100 to $500 for the initial application, with decreased cost for refills. Lash extensions are waterproof and popular for special occasions and vacations, and even more so now for every day.
Potential adverse effects of eyelash extensions include ocular hyperemia, keratoconjunctivitis, allergic blepharitis, and allergic contact dermatitis in the patient. Keratoconjunctivitis is thought to be due to formaldehyde contained in some of the glues used for application.1 Eyelash extensions have also been associated with occupational allergic contact dermatitis, allergic rhinitis, and occupational asthma in the practitioner applying the eyelash extensions, particularly with the cyanoacrylate-based glues.2,3
In a national survey of eyelash extensions and their health-related problems in Japan, 10% (205) of the respondents had experience with eyelash extensions. Of those women, 27% (55) experienced problems that included ocular hyperemia, pain, and itchy swollen eyelids.4 Conjunctival erosion from the eyelid fixing tape used during application and subconjunctival hemorrhage from compression during removal of the extensions has been also reported.1 Hair breakage and even traction alopecia may occur, especially in patients who accidentally or intentionally pull the extensions off.
If permanent eyelash damage occurs, eyelash transplantation may be required to replace the eyelash, as eyelash growth medications such as bimatoprost may not be effective if the follicle is missing or severely damaged. Eyelash transplants often grow long enough where they require trimming, especially if donor sites are taken from the scalp.5
Eyelash extensions offer a nice alternative to daily use of mascara, temporary glue-on eyelashes, and daily application of topical eyelash growth products. As this procedure has increased in number, the dermatologist may be consulted for recommendations and treatment of any potential adverse events associated with it.
References
1. Cornea. 2012 Feb;31(2):121-5.
2. Contact Dermatitis. 2012 Nov;67(5):307-8.
3. Occup Med (Lond). 2013 Jun;63(4):294-7.
4. Nihon Eiseigaku Zasshi. 2013;68(3):168-74.
5. Plast Reconstr Surg Glob Open. 2015 Apr 7;3(3):e324.
Dr. Wesley and Dr. Talakoub are co-contributors to this column. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Wesley.
Flu vaccination found safe in surgical patients
Immunizing surgical patients against seasonal influenza before they are discharged from the hospital appears safe and is a sound strategy for expanding vaccine coverage, especially among people at high risk, according to a report published online March 14 in Annals of Internal Medicine.
All health care contacts, including hospitalizations, are considered excellent opportunities for influenza vaccination, and current recommendations advise that eligible inpatients receive the immunization before discharge. However, surgical patients don’t often get the flu vaccine before they leave the hospital, likely because of concerns that potential adverse effects like fever and myalgia could be falsely attributed to surgical complications. This would lead to unnecessary patient evaluations and could interfere with postsurgical care, said Sara Y. Tartof, Ph.D., and her associates in the department of research and evaluation, Kaiser Permanente Southern California, Pasadena.
“Although this concern is understandable, few clinical data support it,” they noted.
“To provide clinical evidence that would either substantiate or refute” these concerns about perioperative flu vaccination, the investigators analyzed data in the electronic health records for 81,647 surgeries. All the study participants were deemed eligible for flu vaccination. They were socioeconomically and ethnically diverse, ranged in age from 6 months to 106 years, and underwent surgery at 14 hospitals during three consecutive flu seasons. Operations included general, cardiac, eye, dermatologic, ENT, neurologic, ob.gyn., oral/maxillofacial, orthopedic, plastic, podiatric, urologic, and vascular procedures.
Patients received a flu vaccine in 6,420 hospital stays for surgery – only 15% of 42,777 eligible hospitalizations – usually on the day of discharge. (The remaining 38,870 patients either had been vaccinated before hospital admission or were vaccinated more than a week after discharge and were not included in further analyses.)
Compared with eligible patients who didn’t receive a flu vaccine during hospitalization for surgery, those who did showed no increased risk for subsequent inpatient visits, ED visits, or clinical work-ups for infection. Patients who received the flu vaccine before discharge showed a minimally increased risk for outpatient visits during the week following hospitalization, but this was considered unlikely “to translate into substantial clinical impact,” especially when balanced against the benefit of immunization, Dr. Tartof and her associates said (Ann Intern Med. 2016 Mar 14. doi: 10.7326/M15-1667).
Giving the flu vaccine during a surgical hospitalization “is an opportunity to protect a high-risk population,” because surgery patients tend to be of an age, and to have comorbid conditions, that raise their risk for flu complications. In addition, previous research has reported that 39%-46% of adults hospitalized for influenza-related disease in a given year had been hospitalized during the preceding autumn, indicating that recent hospitalization also raises the risk for flu complications, the investigators said.
“Our data support the rationale for increasing vaccination rates among surgical inpatients,” they said.
This study was funded by the U.S. Centers for Disease Control and Prevention through the Vaccine Safety Datalink program. Dr. Tartof reported receiving grants from Merck outside of this work; two of her associates reported receiving grants from Novartis and GlaxoSmithKline outside of this work.
Immunizing surgical patients against seasonal influenza before they are discharged from the hospital appears safe and is a sound strategy for expanding vaccine coverage, especially among people at high risk, according to a report published online March 14 in Annals of Internal Medicine.
All health care contacts, including hospitalizations, are considered excellent opportunities for influenza vaccination, and current recommendations advise that eligible inpatients receive the immunization before discharge. However, surgical patients don’t often get the flu vaccine before they leave the hospital, likely because of concerns that potential adverse effects like fever and myalgia could be falsely attributed to surgical complications. This would lead to unnecessary patient evaluations and could interfere with postsurgical care, said Sara Y. Tartof, Ph.D., and her associates in the department of research and evaluation, Kaiser Permanente Southern California, Pasadena.
“Although this concern is understandable, few clinical data support it,” they noted.
“To provide clinical evidence that would either substantiate or refute” these concerns about perioperative flu vaccination, the investigators analyzed data in the electronic health records for 81,647 surgeries. All the study participants were deemed eligible for flu vaccination. They were socioeconomically and ethnically diverse, ranged in age from 6 months to 106 years, and underwent surgery at 14 hospitals during three consecutive flu seasons. Operations included general, cardiac, eye, dermatologic, ENT, neurologic, ob.gyn., oral/maxillofacial, orthopedic, plastic, podiatric, urologic, and vascular procedures.
Patients received a flu vaccine in 6,420 hospital stays for surgery – only 15% of 42,777 eligible hospitalizations – usually on the day of discharge. (The remaining 38,870 patients either had been vaccinated before hospital admission or were vaccinated more than a week after discharge and were not included in further analyses.)
Compared with eligible patients who didn’t receive a flu vaccine during hospitalization for surgery, those who did showed no increased risk for subsequent inpatient visits, ED visits, or clinical work-ups for infection. Patients who received the flu vaccine before discharge showed a minimally increased risk for outpatient visits during the week following hospitalization, but this was considered unlikely “to translate into substantial clinical impact,” especially when balanced against the benefit of immunization, Dr. Tartof and her associates said (Ann Intern Med. 2016 Mar 14. doi: 10.7326/M15-1667).
Giving the flu vaccine during a surgical hospitalization “is an opportunity to protect a high-risk population,” because surgery patients tend to be of an age, and to have comorbid conditions, that raise their risk for flu complications. In addition, previous research has reported that 39%-46% of adults hospitalized for influenza-related disease in a given year had been hospitalized during the preceding autumn, indicating that recent hospitalization also raises the risk for flu complications, the investigators said.
“Our data support the rationale for increasing vaccination rates among surgical inpatients,” they said.
This study was funded by the U.S. Centers for Disease Control and Prevention through the Vaccine Safety Datalink program. Dr. Tartof reported receiving grants from Merck outside of this work; two of her associates reported receiving grants from Novartis and GlaxoSmithKline outside of this work.
Immunizing surgical patients against seasonal influenza before they are discharged from the hospital appears safe and is a sound strategy for expanding vaccine coverage, especially among people at high risk, according to a report published online March 14 in Annals of Internal Medicine.
All health care contacts, including hospitalizations, are considered excellent opportunities for influenza vaccination, and current recommendations advise that eligible inpatients receive the immunization before discharge. However, surgical patients don’t often get the flu vaccine before they leave the hospital, likely because of concerns that potential adverse effects like fever and myalgia could be falsely attributed to surgical complications. This would lead to unnecessary patient evaluations and could interfere with postsurgical care, said Sara Y. Tartof, Ph.D., and her associates in the department of research and evaluation, Kaiser Permanente Southern California, Pasadena.
“Although this concern is understandable, few clinical data support it,” they noted.
“To provide clinical evidence that would either substantiate or refute” these concerns about perioperative flu vaccination, the investigators analyzed data in the electronic health records for 81,647 surgeries. All the study participants were deemed eligible for flu vaccination. They were socioeconomically and ethnically diverse, ranged in age from 6 months to 106 years, and underwent surgery at 14 hospitals during three consecutive flu seasons. Operations included general, cardiac, eye, dermatologic, ENT, neurologic, ob.gyn., oral/maxillofacial, orthopedic, plastic, podiatric, urologic, and vascular procedures.
Patients received a flu vaccine in 6,420 hospital stays for surgery – only 15% of 42,777 eligible hospitalizations – usually on the day of discharge. (The remaining 38,870 patients either had been vaccinated before hospital admission or were vaccinated more than a week after discharge and were not included in further analyses.)
Compared with eligible patients who didn’t receive a flu vaccine during hospitalization for surgery, those who did showed no increased risk for subsequent inpatient visits, ED visits, or clinical work-ups for infection. Patients who received the flu vaccine before discharge showed a minimally increased risk for outpatient visits during the week following hospitalization, but this was considered unlikely “to translate into substantial clinical impact,” especially when balanced against the benefit of immunization, Dr. Tartof and her associates said (Ann Intern Med. 2016 Mar 14. doi: 10.7326/M15-1667).
Giving the flu vaccine during a surgical hospitalization “is an opportunity to protect a high-risk population,” because surgery patients tend to be of an age, and to have comorbid conditions, that raise their risk for flu complications. In addition, previous research has reported that 39%-46% of adults hospitalized for influenza-related disease in a given year had been hospitalized during the preceding autumn, indicating that recent hospitalization also raises the risk for flu complications, the investigators said.
“Our data support the rationale for increasing vaccination rates among surgical inpatients,” they said.
This study was funded by the U.S. Centers for Disease Control and Prevention through the Vaccine Safety Datalink program. Dr. Tartof reported receiving grants from Merck outside of this work; two of her associates reported receiving grants from Novartis and GlaxoSmithKline outside of this work.
FROM ANNALS OF INTERNAL MEDICINE
Key clinical point: Immunizing surgical patients against seasonal influenza before they leave the hospital appears safe.
Major finding: Patients received a flu vaccine in only 6,420 hospital stays for surgery, comprising only 15% of the patient hospitalizations that were eligible.
Data source: A retrospective cohort study involving 81,647 surgeries at 14 California hospitals during three consecutive flu seasons.
Disclosures: This study was funded by the U.S. Centers for Disease Control and Prevention through the Vaccine Safety Datalink program. Dr. Tartof reported receiving grants from Merck outside of this work; two of her associates reported receiving grants from Novartis and GlaxoSmithKline outside of this work.
Identifying and Managing Abscess Formation Related to Soft-Tissue Fillers
Injectable soft-tissue fillers continue to be popular in the cosmetic arena. In the United States there are many fillers currently on the market and many more coming through the pipeline. A multitude of products are available outside the United States. As with any procedure, the more fillers we inject, the more complications we are bound to see.
Conrad et al (Modern Plastic Surgery. 2015;5:14-18) performed a retrospective analysis of patients treated over a 10-year period with soft-tissue injections (1559 patients) looking for cases complicated by abscess formation. Four patients were identified (0.3% of total patients). The authors discussed the 4 cases, the patients’ medical history and experience with other injectable agents, and the management of each complication.
Case 1 was a 52-year-old woman with systemic lupus erythematosus on a low-dose steroid who presented with an inflammatory response in the lower lip 7 days following injection with a hyaluronic acid (HA)–based gel filler in 2011. Her history was notable for prior HA filler in 2008 and polyacrylamide filler in 2009 and 2010. She was treated with 4 sessions of incision and drainage (I&D) and systemic clindamycin. Most of the cultures were negative, but one showed streptococci.
Case 2 was a 56-year-old woman treated in the nasolabial fold with HA in 2009. She developed inflammation shortly after and an abscess at the site a month later. She was treated with clindamycin both times, though cultures were negative. Furthermore, the abscess was treated with I&D and an intralesional steroid. She was a smoker and had been treated with a polymethyl methacrylate filler in 2002 and subsequently in 2013 with no issues.
Case 3 was a 39-year-old woman injected with an HA filler in the upper and lower lips in 2011. One month later she developed abscesses in both areas that were treated twice with I&D. Cultures were negative. She had a history of polyacrylamide injections of the nasolabial fold in 2009. The patient’s medical history was notable for scleroderma.
Case 4 was a 58-year-old woman injected with an HA filler in 2009 in the prejowl sulcus and nasolabial fold. She developed recurrent sterile abscesses in the areas 8 months after treatment that were managed by drainage of the areas and intralesional steroid injections over the ensuing 6 months. The scars were then excised, lasered 6 weeks later, and then filled in with expanded polytetrafluoroethylene implants, followed by 1 more session of laser resurfacing. She had a history of polymethyl methacrylate filler in 2002.
All patients eventually recovered. The authors stressed 3 important factors in managing dermal filler complications: (1) identifying the causative pathogen, (2) choosing the appropriate treatment of delayed-onset abscess formation, and (3) identifying the risk factors for patients at risk for abscess formation.
The issue of biofilms complicates the ability to identify the bacterial agent, yet biofilms are becoming recognized as the causative factors in what were previously thought of as sterile abscesses. The authors suggested using a peptide nucleic acid fluorescent in situ hybridization test to identify the biofilm bacteria. Conrad et el also discussed the development of slippery liquid-infused porous surfaces technology to coat the inside of syringes to help prevent biofilm formation.
The management of these patients is tricky because it is difficult to differentiate between a biofilm abscess and a hypersensitivity reaction. For this reason, the authors advocated using hyaluronidase versus intralesional steroids in the initial management to make the area more susceptible to antibiotics and to avoid promoting the growth of bacteria with the use of steroids. For patient risk factors, the authors focused on the fact that 2 of 4 patients had concomitant autoimmune disorders—scleroderma and systemic lupus erythematosus—that may have predisposed them to infection. Lastly, 2 patients had prior polyacrylamide injections and the authors also speculated if the positive charge of this filler attracted bacteria.
What’s the issue?
The use of fillers will continue to increase as there are more fillers with novel properties entering the market. As with new technology, only time will tell if we will see any particular type of reaction or risk for infection with them. The issue of biofilm bacterial contamination is real. It is recognized as one of the causes of capsular contraction following breast implant surgery. The etiology may not be from contamination during production but from contamination of the filler after injection due to any transient bacteremia that the patient may experience. A concern is that dental manipulation (eg, dental cleaning, filling of dental caries, periodontal surgery) during the 2- to 4-week postfiller period may “seed” bacteria into the area and cause the bacteria to settle and grow on the foreign substance. For patients who have semipermanent or permanent fillers such as polyacrylamide, polymethyl methacrylate beads, or poly-L-lactic acid, biofilm risk is greater and can occur months to years after the procedure. I have personally seen 2 cases of poly-L-lactic acid filler develop red, tender, sterile abscesses 1 year after placement in the tissue. Both cases responded to prolonged clarithromycin use (2 months). However, these cases highlight the fact that the fillers persist long after we place them, and any bacteremia, even mild, can cause an unsightly reaction.
Have you seen delayed soft-tissue filler reactions in your practice? Given this information, will you change the way you advise patients on dental procedures in the 2- to 4-week postfiller period?
Injectable soft-tissue fillers continue to be popular in the cosmetic arena. In the United States there are many fillers currently on the market and many more coming through the pipeline. A multitude of products are available outside the United States. As with any procedure, the more fillers we inject, the more complications we are bound to see.
Conrad et al (Modern Plastic Surgery. 2015;5:14-18) performed a retrospective analysis of patients treated over a 10-year period with soft-tissue injections (1559 patients) looking for cases complicated by abscess formation. Four patients were identified (0.3% of total patients). The authors discussed the 4 cases, the patients’ medical history and experience with other injectable agents, and the management of each complication.
Case 1 was a 52-year-old woman with systemic lupus erythematosus on a low-dose steroid who presented with an inflammatory response in the lower lip 7 days following injection with a hyaluronic acid (HA)–based gel filler in 2011. Her history was notable for prior HA filler in 2008 and polyacrylamide filler in 2009 and 2010. She was treated with 4 sessions of incision and drainage (I&D) and systemic clindamycin. Most of the cultures were negative, but one showed streptococci.
Case 2 was a 56-year-old woman treated in the nasolabial fold with HA in 2009. She developed inflammation shortly after and an abscess at the site a month later. She was treated with clindamycin both times, though cultures were negative. Furthermore, the abscess was treated with I&D and an intralesional steroid. She was a smoker and had been treated with a polymethyl methacrylate filler in 2002 and subsequently in 2013 with no issues.
Case 3 was a 39-year-old woman injected with an HA filler in the upper and lower lips in 2011. One month later she developed abscesses in both areas that were treated twice with I&D. Cultures were negative. She had a history of polyacrylamide injections of the nasolabial fold in 2009. The patient’s medical history was notable for scleroderma.
Case 4 was a 58-year-old woman injected with an HA filler in 2009 in the prejowl sulcus and nasolabial fold. She developed recurrent sterile abscesses in the areas 8 months after treatment that were managed by drainage of the areas and intralesional steroid injections over the ensuing 6 months. The scars were then excised, lasered 6 weeks later, and then filled in with expanded polytetrafluoroethylene implants, followed by 1 more session of laser resurfacing. She had a history of polymethyl methacrylate filler in 2002.
All patients eventually recovered. The authors stressed 3 important factors in managing dermal filler complications: (1) identifying the causative pathogen, (2) choosing the appropriate treatment of delayed-onset abscess formation, and (3) identifying the risk factors for patients at risk for abscess formation.
The issue of biofilms complicates the ability to identify the bacterial agent, yet biofilms are becoming recognized as the causative factors in what were previously thought of as sterile abscesses. The authors suggested using a peptide nucleic acid fluorescent in situ hybridization test to identify the biofilm bacteria. Conrad et el also discussed the development of slippery liquid-infused porous surfaces technology to coat the inside of syringes to help prevent biofilm formation.
The management of these patients is tricky because it is difficult to differentiate between a biofilm abscess and a hypersensitivity reaction. For this reason, the authors advocated using hyaluronidase versus intralesional steroids in the initial management to make the area more susceptible to antibiotics and to avoid promoting the growth of bacteria with the use of steroids. For patient risk factors, the authors focused on the fact that 2 of 4 patients had concomitant autoimmune disorders—scleroderma and systemic lupus erythematosus—that may have predisposed them to infection. Lastly, 2 patients had prior polyacrylamide injections and the authors also speculated if the positive charge of this filler attracted bacteria.
What’s the issue?
The use of fillers will continue to increase as there are more fillers with novel properties entering the market. As with new technology, only time will tell if we will see any particular type of reaction or risk for infection with them. The issue of biofilm bacterial contamination is real. It is recognized as one of the causes of capsular contraction following breast implant surgery. The etiology may not be from contamination during production but from contamination of the filler after injection due to any transient bacteremia that the patient may experience. A concern is that dental manipulation (eg, dental cleaning, filling of dental caries, periodontal surgery) during the 2- to 4-week postfiller period may “seed” bacteria into the area and cause the bacteria to settle and grow on the foreign substance. For patients who have semipermanent or permanent fillers such as polyacrylamide, polymethyl methacrylate beads, or poly-L-lactic acid, biofilm risk is greater and can occur months to years after the procedure. I have personally seen 2 cases of poly-L-lactic acid filler develop red, tender, sterile abscesses 1 year after placement in the tissue. Both cases responded to prolonged clarithromycin use (2 months). However, these cases highlight the fact that the fillers persist long after we place them, and any bacteremia, even mild, can cause an unsightly reaction.
Have you seen delayed soft-tissue filler reactions in your practice? Given this information, will you change the way you advise patients on dental procedures in the 2- to 4-week postfiller period?
Injectable soft-tissue fillers continue to be popular in the cosmetic arena. In the United States there are many fillers currently on the market and many more coming through the pipeline. A multitude of products are available outside the United States. As with any procedure, the more fillers we inject, the more complications we are bound to see.
Conrad et al (Modern Plastic Surgery. 2015;5:14-18) performed a retrospective analysis of patients treated over a 10-year period with soft-tissue injections (1559 patients) looking for cases complicated by abscess formation. Four patients were identified (0.3% of total patients). The authors discussed the 4 cases, the patients’ medical history and experience with other injectable agents, and the management of each complication.
Case 1 was a 52-year-old woman with systemic lupus erythematosus on a low-dose steroid who presented with an inflammatory response in the lower lip 7 days following injection with a hyaluronic acid (HA)–based gel filler in 2011. Her history was notable for prior HA filler in 2008 and polyacrylamide filler in 2009 and 2010. She was treated with 4 sessions of incision and drainage (I&D) and systemic clindamycin. Most of the cultures were negative, but one showed streptococci.
Case 2 was a 56-year-old woman treated in the nasolabial fold with HA in 2009. She developed inflammation shortly after and an abscess at the site a month later. She was treated with clindamycin both times, though cultures were negative. Furthermore, the abscess was treated with I&D and an intralesional steroid. She was a smoker and had been treated with a polymethyl methacrylate filler in 2002 and subsequently in 2013 with no issues.
Case 3 was a 39-year-old woman injected with an HA filler in the upper and lower lips in 2011. One month later she developed abscesses in both areas that were treated twice with I&D. Cultures were negative. She had a history of polyacrylamide injections of the nasolabial fold in 2009. The patient’s medical history was notable for scleroderma.
Case 4 was a 58-year-old woman injected with an HA filler in 2009 in the prejowl sulcus and nasolabial fold. She developed recurrent sterile abscesses in the areas 8 months after treatment that were managed by drainage of the areas and intralesional steroid injections over the ensuing 6 months. The scars were then excised, lasered 6 weeks later, and then filled in with expanded polytetrafluoroethylene implants, followed by 1 more session of laser resurfacing. She had a history of polymethyl methacrylate filler in 2002.
All patients eventually recovered. The authors stressed 3 important factors in managing dermal filler complications: (1) identifying the causative pathogen, (2) choosing the appropriate treatment of delayed-onset abscess formation, and (3) identifying the risk factors for patients at risk for abscess formation.
The issue of biofilms complicates the ability to identify the bacterial agent, yet biofilms are becoming recognized as the causative factors in what were previously thought of as sterile abscesses. The authors suggested using a peptide nucleic acid fluorescent in situ hybridization test to identify the biofilm bacteria. Conrad et el also discussed the development of slippery liquid-infused porous surfaces technology to coat the inside of syringes to help prevent biofilm formation.
The management of these patients is tricky because it is difficult to differentiate between a biofilm abscess and a hypersensitivity reaction. For this reason, the authors advocated using hyaluronidase versus intralesional steroids in the initial management to make the area more susceptible to antibiotics and to avoid promoting the growth of bacteria with the use of steroids. For patient risk factors, the authors focused on the fact that 2 of 4 patients had concomitant autoimmune disorders—scleroderma and systemic lupus erythematosus—that may have predisposed them to infection. Lastly, 2 patients had prior polyacrylamide injections and the authors also speculated if the positive charge of this filler attracted bacteria.
What’s the issue?
The use of fillers will continue to increase as there are more fillers with novel properties entering the market. As with new technology, only time will tell if we will see any particular type of reaction or risk for infection with them. The issue of biofilm bacterial contamination is real. It is recognized as one of the causes of capsular contraction following breast implant surgery. The etiology may not be from contamination during production but from contamination of the filler after injection due to any transient bacteremia that the patient may experience. A concern is that dental manipulation (eg, dental cleaning, filling of dental caries, periodontal surgery) during the 2- to 4-week postfiller period may “seed” bacteria into the area and cause the bacteria to settle and grow on the foreign substance. For patients who have semipermanent or permanent fillers such as polyacrylamide, polymethyl methacrylate beads, or poly-L-lactic acid, biofilm risk is greater and can occur months to years after the procedure. I have personally seen 2 cases of poly-L-lactic acid filler develop red, tender, sterile abscesses 1 year after placement in the tissue. Both cases responded to prolonged clarithromycin use (2 months). However, these cases highlight the fact that the fillers persist long after we place them, and any bacteremia, even mild, can cause an unsightly reaction.
Have you seen delayed soft-tissue filler reactions in your practice? Given this information, will you change the way you advise patients on dental procedures in the 2- to 4-week postfiller period?
Cosmetic Corner: Dermatologists Weigh in on Hand Creams
To improve patient care and outcomes, leading dermatologists offered their recommendations on hand creams. Consideration must be given to:
- CeraVe Therapeutic Hand Cream
- Maximum Body Repair
- Neutrogena Norwegian Formula Hand Cream
- O’Keeffe’s Working Hands
Cutis invites readers to send us their recommendations. Scar treatments, body scrubs, and OTC acne treatments will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
To improve patient care and outcomes, leading dermatologists offered their recommendations on hand creams. Consideration must be given to:
- CeraVe Therapeutic Hand Cream
- Maximum Body Repair
- Neutrogena Norwegian Formula Hand Cream
- O’Keeffe’s Working Hands
Cutis invites readers to send us their recommendations. Scar treatments, body scrubs, and OTC acne treatments will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
To improve patient care and outcomes, leading dermatologists offered their recommendations on hand creams. Consideration must be given to:
- CeraVe Therapeutic Hand Cream
- Maximum Body Repair
- Neutrogena Norwegian Formula Hand Cream
- O’Keeffe’s Working Hands
Cutis invites readers to send us their recommendations. Scar treatments, body scrubs, and OTC acne treatments will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
Clinical Pearl: The Squeeze Maneuver
Practice Gap
Warts may negatively impact a patient's quality of life, as they may cause not only discomfort and pain but also embarrassment and low self-esteem.1 Moreover, Ciconte et al1 demonstrated that study participants with warts on their feet were more likely to report physical discomfort than those with warts on their hands. Therefore, plantar warts should be diagnosed promptly to allow for proper treatment.
Warts may be identified by viewing the dilated capillaries that lie on their surface, which appear as small black dots to the naked eye.1 The formation of a plantar wart obliterates the normal plantar creases, thereby flattening the skin’s natural markings. However, a plantar wart may appear clinically similar to a callus and both lesions typically form in pressure point areas, warranting the use of a tool that aids in its diagnostic evaluation.1,2
Diagnostic Tools
Dermoscopy, a noninvasive tool that creates a microscopic visualization of lesions, is commonly used to distinguish dermatologic pathology if the clinical presentation overlaps with a similar lesion, such as a callus, corn, or plantar wart.1,3 However, there is another way of differentiating plantar warts from calluses using a simple 2-step clinical maneuver that we learned from Dr. Lewis Kaplan at the University of Miami.
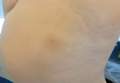
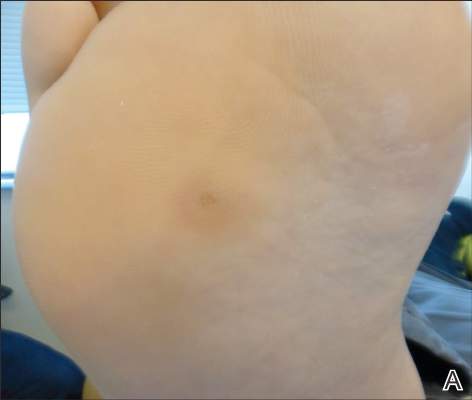
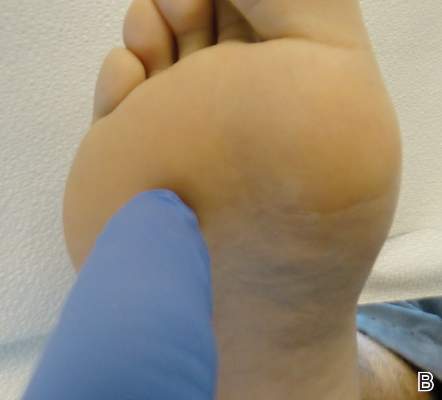
Using the thumb or index finger, apply pressure at a perpendicular angle to the lesion on the sole of the patient’s foot, which will not create substantial discomfort or pain in a patient who has a plantar wart (Figure) but will be painful in a patient who has a callus due to the underlying bony spur. The next step involves applying pressure to the left and right sides of the lesion by squeezing toward the center with the thumb and index finger at a 45° angle. This maneuver will create substantial discomfort and pain in patients with plantar warts, thus helping to confirm the diagnosis.
| 
|
| A plantar wart before (A) and after undergoing the squeeze maneuver (B). The patient denied feelings of discomfort or pain. | |
Practice Implications
Rarely, a plantar wart can progress to form a verrucous carcinoma if left untreated.2 Thus, it is important to diagnose and treat plantar warts to avoid pain and potential complications. The technique discussed here, which we are coining as the “squeeze maneuver,” allows for easy diagnosis and negates the need for an expensive diagnostic tool.To submit a clinical pearl, contact our Editorial Office.
- Ciconte A, Campbell J, Tabrizi S, et al. Warts are not merely blemishes on the skin: a study on the morbidity associated with having viral cutaneous warts. Australas J Dermatol. 2003;44:169-173.
- Cardoso J, Calonje E. Cutaneous manifestations of human papillomaviruses: a review. Acta Dermatovenerol. 2011;20:145-154.
- Bae J, Kang H, Kim H, et al. Differential diagnosis of plantar wart from corn, callus and healed wart with the aid of dermoscopy. Br J Dermatol. 2009;160:220-222.
Practice Gap
Warts may negatively impact a patient's quality of life, as they may cause not only discomfort and pain but also embarrassment and low self-esteem.1 Moreover, Ciconte et al1 demonstrated that study participants with warts on their feet were more likely to report physical discomfort than those with warts on their hands. Therefore, plantar warts should be diagnosed promptly to allow for proper treatment.
Warts may be identified by viewing the dilated capillaries that lie on their surface, which appear as small black dots to the naked eye.1 The formation of a plantar wart obliterates the normal plantar creases, thereby flattening the skin’s natural markings. However, a plantar wart may appear clinically similar to a callus and both lesions typically form in pressure point areas, warranting the use of a tool that aids in its diagnostic evaluation.1,2
Diagnostic Tools
Dermoscopy, a noninvasive tool that creates a microscopic visualization of lesions, is commonly used to distinguish dermatologic pathology if the clinical presentation overlaps with a similar lesion, such as a callus, corn, or plantar wart.1,3 However, there is another way of differentiating plantar warts from calluses using a simple 2-step clinical maneuver that we learned from Dr. Lewis Kaplan at the University of Miami.
Using the thumb or index finger, apply pressure at a perpendicular angle to the lesion on the sole of the patient’s foot, which will not create substantial discomfort or pain in a patient who has a plantar wart (Figure) but will be painful in a patient who has a callus due to the underlying bony spur. The next step involves applying pressure to the left and right sides of the lesion by squeezing toward the center with the thumb and index finger at a 45° angle. This maneuver will create substantial discomfort and pain in patients with plantar warts, thus helping to confirm the diagnosis.

| 
|
| A plantar wart before (A) and after undergoing the squeeze maneuver (B). The patient denied feelings of discomfort or pain. | |
Practice Implications
Rarely, a plantar wart can progress to form a verrucous carcinoma if left untreated.2 Thus, it is important to diagnose and treat plantar warts to avoid pain and potential complications. The technique discussed here, which we are coining as the “squeeze maneuver,” allows for easy diagnosis and negates the need for an expensive diagnostic tool.To submit a clinical pearl, contact our Editorial Office.
Practice Gap
Warts may negatively impact a patient's quality of life, as they may cause not only discomfort and pain but also embarrassment and low self-esteem.1 Moreover, Ciconte et al1 demonstrated that study participants with warts on their feet were more likely to report physical discomfort than those with warts on their hands. Therefore, plantar warts should be diagnosed promptly to allow for proper treatment.
Warts may be identified by viewing the dilated capillaries that lie on their surface, which appear as small black dots to the naked eye.1 The formation of a plantar wart obliterates the normal plantar creases, thereby flattening the skin’s natural markings. However, a plantar wart may appear clinically similar to a callus and both lesions typically form in pressure point areas, warranting the use of a tool that aids in its diagnostic evaluation.1,2
Diagnostic Tools
Dermoscopy, a noninvasive tool that creates a microscopic visualization of lesions, is commonly used to distinguish dermatologic pathology if the clinical presentation overlaps with a similar lesion, such as a callus, corn, or plantar wart.1,3 However, there is another way of differentiating plantar warts from calluses using a simple 2-step clinical maneuver that we learned from Dr. Lewis Kaplan at the University of Miami.
Using the thumb or index finger, apply pressure at a perpendicular angle to the lesion on the sole of the patient’s foot, which will not create substantial discomfort or pain in a patient who has a plantar wart (Figure) but will be painful in a patient who has a callus due to the underlying bony spur. The next step involves applying pressure to the left and right sides of the lesion by squeezing toward the center with the thumb and index finger at a 45° angle. This maneuver will create substantial discomfort and pain in patients with plantar warts, thus helping to confirm the diagnosis.

| 
|
| A plantar wart before (A) and after undergoing the squeeze maneuver (B). The patient denied feelings of discomfort or pain. | |
Practice Implications
Rarely, a plantar wart can progress to form a verrucous carcinoma if left untreated.2 Thus, it is important to diagnose and treat plantar warts to avoid pain and potential complications. The technique discussed here, which we are coining as the “squeeze maneuver,” allows for easy diagnosis and negates the need for an expensive diagnostic tool.To submit a clinical pearl, contact our Editorial Office.
- Ciconte A, Campbell J, Tabrizi S, et al. Warts are not merely blemishes on the skin: a study on the morbidity associated with having viral cutaneous warts. Australas J Dermatol. 2003;44:169-173.
- Cardoso J, Calonje E. Cutaneous manifestations of human papillomaviruses: a review. Acta Dermatovenerol. 2011;20:145-154.
- Bae J, Kang H, Kim H, et al. Differential diagnosis of plantar wart from corn, callus and healed wart with the aid of dermoscopy. Br J Dermatol. 2009;160:220-222.
- Ciconte A, Campbell J, Tabrizi S, et al. Warts are not merely blemishes on the skin: a study on the morbidity associated with having viral cutaneous warts. Australas J Dermatol. 2003;44:169-173.
- Cardoso J, Calonje E. Cutaneous manifestations of human papillomaviruses: a review. Acta Dermatovenerol. 2011;20:145-154.
- Bae J, Kang H, Kim H, et al. Differential diagnosis of plantar wart from corn, callus and healed wart with the aid of dermoscopy. Br J Dermatol. 2009;160:220-222.
Don’t Get Hung Up on Fishhooks: A Guide to Fishhook Removal
Fishing is one of the world’s most beloved activities, enjoyed as a sport or a leisure activity. However, a common injury from fishing is embedment of the fishhook in the cutaneous tissue. Barbed fishhooks are used for their effectiveness in maintaining the fish on the hook once it is caught, but when implanted in the hand of a fisherman or fisherwoman, barbs can pose problems for removal without exacerbating internal tissue injury. Nevertheless, dermatologists should not shy away from removal of barbed fishhooks, as there are several simple methods that can be easily utilized in the outpatient setting.
Case Report
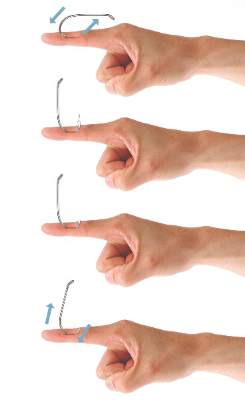
A 68-year-old man presented to an outpatient dermatology clinic after sustaining a barbed fishhook injury while fishing. The fishhook was firmly inserted into the ventral side of the third digit of the right hand (Figure 1).
Prior to presenting to dermatology, the patient went to 2 urgent care clinics the same day seeking treatment. He reported that practitioners at the first clinic were not able to remove the fishhook because they did not have pliers in stock. At the second clinic he was told the fishhook might be embedded in deeper tissues and was advised to go to the emergency department at the local hospital. When he arrived at the emergency department, a 6-hour wait time prompted him to see a local dermatologist instead.
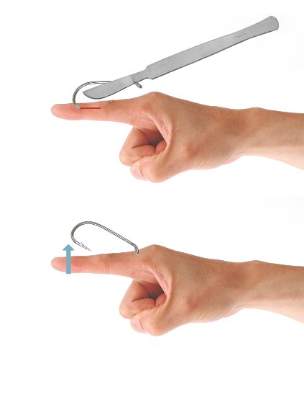
To remove the fishhook, the area was cleaned and prepared first; lidocaine 2% was administered for local anesthesia. An 18-gauge needle was then advanced through the puncture site parallel to the fishhook’s inner shaft on the same side as the barb, which could be successfully palpated using the tip of the 18-gauge needle. The tip of the needle was then used to cap the barb beneath the skin. This technique allowed for the hook to be easily extracted in a retrograde manner without causing further destruction to the surrounding tissue. The patient then was started on prophylaxis cephalexin 500 mg 3 times daily for 3 days.
Comment
The hand is the most common site of fishhook injury, followed closely by the head and eyes.1 Barbless fishhooks usually can be removed by pushing the hook in a retrograde manner along the path of insertion. This method is simple and rarely results in complications. However, there are no guidelines for removal of barbed fishhooks. Furthermore, removing a barbed fishhook in the same retrograde manner would result in extensive internal tissue destruction and increased complications. Due to the popularity of the sport of fishing, fishhook injuries, depending on geographical location, are not uncommon.2 For this reason, trauma and emergency practitioners have become well versed in safe methods for barbed fishhook removal. However, patients are not always able or willing to seek medical care in emergency departments and may opt to seek treatment in outpatient settings, such as in our case. As a result, dermatologists should familiarize themselves with safe and effective fishhook removal methods, as they are not time consuming and do not require complex equipment. Failure to treat the patient may lead to further patient discomfort and increased risk for complications. Additionally, many of the techniques for removal may be useful with other foreign bodies embedded in cutaneous tissue (eg, splinters).
There are a number of safe and effective techniques for removing barbed fishhooks from cutaneous tissue, including the advance-and-cut method, the cut-it-out technique, the string-pull method, and the needle cover technique.1-3 The method chosen to remove the fishhook is dependent on a variety of factors, such as anatomic location, tissue depth, and provider comfort.
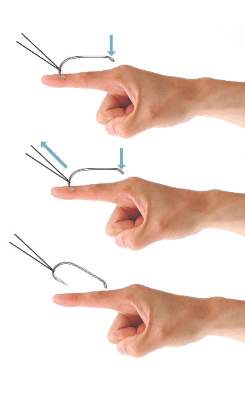
With the advance-and-cut method (Figure 2), the affected area is anesthetized and a small incision in the skin is created to expose the barb. The fishhook is then advanced through the incision, providing visibility of the barb and thus allowing the practitioner to cut the barbed tip without creating further damage to the surrounding tissue. The shaft of the fishhook can subsequently be removed in a retrograde fashion. The advantages of this technique include that it may be successfully used in all types of barbed fishhooks and it provides the practitioner with direct visibility of the barb, thus minimizing risk for neurovascular injury during removal.1 However, the primary disadvantage is that a second cutaneous wound is created in exposing the barb.
| 
| |
| Figure 2. The advance-and-cut method for fishhook removal. | Figure 3. The cut-it-out method for fishhook removal. |
|
The cut-it-out technique (Figure 3) is similar to the advance-and-cut method in that they both require anesthesia along with creating an incision. With this method, a scalpel is used to create a small linear incision originating at the fishhook entrance site and ending at the approximated location of the fishhook’s tip. The fishhook then is simply lifted superiorly in a retrograde fashion.
|
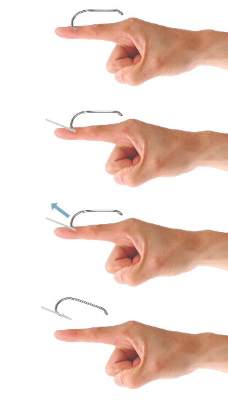
The string-pull method (Figure 4) has been credited to fishermen in South Australia and was first described by Cooke2 in 1961. This method is relatively painless, does not require anesthesia, and has a high success rate when properly administered. However, it does require rapid and confident motions (ie, without hesitation) by the practitioner and should not be performed on free-moving areas of the body (eg, earlobe).3 With this technique, a sturdy piece of suture (eg, 2/0 or 3/0 strength silk) is looped around the hook and is extended away from the practitioner at a 30° angle. The free end of the suture is then securely fastened around the index finger of the practitioner’s dominant hand. The index finger of the nondominant hand should apply a downward pressure to the hook shaft to disengage the barb from the tissue. Simultaneously and rather quickly and forcefully the practitioner must pull the dominant index finger with the string attached in a superior and lateral direction, as depicted by the long arrow in Figure 4. If successful, the barbed hook will pull out of the entrance site. The use of string in pulling the fishhook parallel to the site of injury is helpful for smaller fishhooks that may be difficult to grab with fingers alone. However, with larger fishhooks, the string may not be required so long as the practitioner is able to obtain a secure grasp on the fishhook shaft. The string-pull method becomes particularly useful when anesthesia is unavailable or when the barb of the hook is embedded too deeply for safe advancement through tissue to visualize and cut the barb.
|
Lastly, the needle cover technique (Figure 5) is another simple method that does not require the creation of a secondary wound. An 18-gauge needle is simply inserted parallel to the fishhook curvature into the site of entry. By using the needle to slide along the fishhook’s curve, the practitioner is able to follow its pathway while in the tissue. The tip of the 18-gauge needle is then used to cap or cover the barb, thus allowing the fishhook to be removed in a retrograde fashion from the wound. In an outpatient setting, this technique does not require the creation of additional tissue damage and practitioners who are inexperienced with fishhook removal may proceed through the motions more slowly and methodically than the string-pull method permits.
Wound care following fishhook removal should involve adequate flushing of the wound with normal saline along with the application of topical antibiotics and a simple dressing and adhesive bandage. Oral prophylactic antibiotics typically are not required for shallow cutaneous injuries unless the fishhook is dirty, the patient is immunocompromised, or the patient has a condition lending to poor wound healing (eg, diabetes mellitus, peripheral vascular disease).3 When deciding on antibiotics, it is important to note that fishhook injuries while saltwater fishing are associated with Vibrio infection, while injuries sustained during freshwater fishing are associated with gram-negative bacteria (eg, Pseudomonas and Aeromonas species).3 Lastly, it is essential to find out the immunization status of the patient, and tetanus immune globulin should be provided if necessary.
| 
| |
| Figure 4. The string-pull method for fishhook removal. | Figure 5. The needle cover technique for fishhook removal. |
Conclusion
Although guidelines for barbed fishhook removal are not available, outpatient physicians, including dermatologists, should not fear removal procedures. There are many safe and effective fishhook removal methods that are not time consuming and do not require complex equipment. Furthermore, familiarization with these same techniques may be useful for removal of other foreign bodies embedded in cutaneous tissue.
1. Khan HA, Kamal Y, Lone AU. Fish hook injury: removal by “push through and cut off” technique: a case report and brief literature review [published online March 24, 2014]. Trauma Mon. 2014;19:e17728.
2. Cooke T. How to remove fish-hooks with a bit of string. Med J Aust. 1961;48:815-816.
3. Thommasen HV, Thommasen A. The occasional removal of an embedded fish hook. Can J Rural Med. 2005;10:255-259.
Fishing is one of the world’s most beloved activities, enjoyed as a sport or a leisure activity. However, a common injury from fishing is embedment of the fishhook in the cutaneous tissue. Barbed fishhooks are used for their effectiveness in maintaining the fish on the hook once it is caught, but when implanted in the hand of a fisherman or fisherwoman, barbs can pose problems for removal without exacerbating internal tissue injury. Nevertheless, dermatologists should not shy away from removal of barbed fishhooks, as there are several simple methods that can be easily utilized in the outpatient setting.
Case Report
A 68-year-old man presented to an outpatient dermatology clinic after sustaining a barbed fishhook injury while fishing. The fishhook was firmly inserted into the ventral side of the third digit of the right hand (Figure 1).
Prior to presenting to dermatology, the patient went to 2 urgent care clinics the same day seeking treatment. He reported that practitioners at the first clinic were not able to remove the fishhook because they did not have pliers in stock. At the second clinic he was told the fishhook might be embedded in deeper tissues and was advised to go to the emergency department at the local hospital. When he arrived at the emergency department, a 6-hour wait time prompted him to see a local dermatologist instead.
To remove the fishhook, the area was cleaned and prepared first; lidocaine 2% was administered for local anesthesia. An 18-gauge needle was then advanced through the puncture site parallel to the fishhook’s inner shaft on the same side as the barb, which could be successfully palpated using the tip of the 18-gauge needle. The tip of the needle was then used to cap the barb beneath the skin. This technique allowed for the hook to be easily extracted in a retrograde manner without causing further destruction to the surrounding tissue. The patient then was started on prophylaxis cephalexin 500 mg 3 times daily for 3 days.
Comment
The hand is the most common site of fishhook injury, followed closely by the head and eyes.1 Barbless fishhooks usually can be removed by pushing the hook in a retrograde manner along the path of insertion. This method is simple and rarely results in complications. However, there are no guidelines for removal of barbed fishhooks. Furthermore, removing a barbed fishhook in the same retrograde manner would result in extensive internal tissue destruction and increased complications. Due to the popularity of the sport of fishing, fishhook injuries, depending on geographical location, are not uncommon.2 For this reason, trauma and emergency practitioners have become well versed in safe methods for barbed fishhook removal. However, patients are not always able or willing to seek medical care in emergency departments and may opt to seek treatment in outpatient settings, such as in our case. As a result, dermatologists should familiarize themselves with safe and effective fishhook removal methods, as they are not time consuming and do not require complex equipment. Failure to treat the patient may lead to further patient discomfort and increased risk for complications. Additionally, many of the techniques for removal may be useful with other foreign bodies embedded in cutaneous tissue (eg, splinters).
There are a number of safe and effective techniques for removing barbed fishhooks from cutaneous tissue, including the advance-and-cut method, the cut-it-out technique, the string-pull method, and the needle cover technique.1-3 The method chosen to remove the fishhook is dependent on a variety of factors, such as anatomic location, tissue depth, and provider comfort.
With the advance-and-cut method (Figure 2), the affected area is anesthetized and a small incision in the skin is created to expose the barb. The fishhook is then advanced through the incision, providing visibility of the barb and thus allowing the practitioner to cut the barbed tip without creating further damage to the surrounding tissue. The shaft of the fishhook can subsequently be removed in a retrograde fashion. The advantages of this technique include that it may be successfully used in all types of barbed fishhooks and it provides the practitioner with direct visibility of the barb, thus minimizing risk for neurovascular injury during removal.1 However, the primary disadvantage is that a second cutaneous wound is created in exposing the barb.

| 
| |
| Figure 2. The advance-and-cut method for fishhook removal. | Figure 3. The cut-it-out method for fishhook removal. |
|
The cut-it-out technique (Figure 3) is similar to the advance-and-cut method in that they both require anesthesia along with creating an incision. With this method, a scalpel is used to create a small linear incision originating at the fishhook entrance site and ending at the approximated location of the fishhook’s tip. The fishhook then is simply lifted superiorly in a retrograde fashion.
|
The string-pull method (Figure 4) has been credited to fishermen in South Australia and was first described by Cooke2 in 1961. This method is relatively painless, does not require anesthesia, and has a high success rate when properly administered. However, it does require rapid and confident motions (ie, without hesitation) by the practitioner and should not be performed on free-moving areas of the body (eg, earlobe).3 With this technique, a sturdy piece of suture (eg, 2/0 or 3/0 strength silk) is looped around the hook and is extended away from the practitioner at a 30° angle. The free end of the suture is then securely fastened around the index finger of the practitioner’s dominant hand. The index finger of the nondominant hand should apply a downward pressure to the hook shaft to disengage the barb from the tissue. Simultaneously and rather quickly and forcefully the practitioner must pull the dominant index finger with the string attached in a superior and lateral direction, as depicted by the long arrow in Figure 4. If successful, the barbed hook will pull out of the entrance site. The use of string in pulling the fishhook parallel to the site of injury is helpful for smaller fishhooks that may be difficult to grab with fingers alone. However, with larger fishhooks, the string may not be required so long as the practitioner is able to obtain a secure grasp on the fishhook shaft. The string-pull method becomes particularly useful when anesthesia is unavailable or when the barb of the hook is embedded too deeply for safe advancement through tissue to visualize and cut the barb.
|
Lastly, the needle cover technique (Figure 5) is another simple method that does not require the creation of a secondary wound. An 18-gauge needle is simply inserted parallel to the fishhook curvature into the site of entry. By using the needle to slide along the fishhook’s curve, the practitioner is able to follow its pathway while in the tissue. The tip of the 18-gauge needle is then used to cap or cover the barb, thus allowing the fishhook to be removed in a retrograde fashion from the wound. In an outpatient setting, this technique does not require the creation of additional tissue damage and practitioners who are inexperienced with fishhook removal may proceed through the motions more slowly and methodically than the string-pull method permits.
Wound care following fishhook removal should involve adequate flushing of the wound with normal saline along with the application of topical antibiotics and a simple dressing and adhesive bandage. Oral prophylactic antibiotics typically are not required for shallow cutaneous injuries unless the fishhook is dirty, the patient is immunocompromised, or the patient has a condition lending to poor wound healing (eg, diabetes mellitus, peripheral vascular disease).3 When deciding on antibiotics, it is important to note that fishhook injuries while saltwater fishing are associated with Vibrio infection, while injuries sustained during freshwater fishing are associated with gram-negative bacteria (eg, Pseudomonas and Aeromonas species).3 Lastly, it is essential to find out the immunization status of the patient, and tetanus immune globulin should be provided if necessary.

| 
| |
| Figure 4. The string-pull method for fishhook removal. | Figure 5. The needle cover technique for fishhook removal. |
Conclusion
Although guidelines for barbed fishhook removal are not available, outpatient physicians, including dermatologists, should not fear removal procedures. There are many safe and effective fishhook removal methods that are not time consuming and do not require complex equipment. Furthermore, familiarization with these same techniques may be useful for removal of other foreign bodies embedded in cutaneous tissue.
Fishing is one of the world’s most beloved activities, enjoyed as a sport or a leisure activity. However, a common injury from fishing is embedment of the fishhook in the cutaneous tissue. Barbed fishhooks are used for their effectiveness in maintaining the fish on the hook once it is caught, but when implanted in the hand of a fisherman or fisherwoman, barbs can pose problems for removal without exacerbating internal tissue injury. Nevertheless, dermatologists should not shy away from removal of barbed fishhooks, as there are several simple methods that can be easily utilized in the outpatient setting.
Case Report
A 68-year-old man presented to an outpatient dermatology clinic after sustaining a barbed fishhook injury while fishing. The fishhook was firmly inserted into the ventral side of the third digit of the right hand (Figure 1).
Prior to presenting to dermatology, the patient went to 2 urgent care clinics the same day seeking treatment. He reported that practitioners at the first clinic were not able to remove the fishhook because they did not have pliers in stock. At the second clinic he was told the fishhook might be embedded in deeper tissues and was advised to go to the emergency department at the local hospital. When he arrived at the emergency department, a 6-hour wait time prompted him to see a local dermatologist instead.
To remove the fishhook, the area was cleaned and prepared first; lidocaine 2% was administered for local anesthesia. An 18-gauge needle was then advanced through the puncture site parallel to the fishhook’s inner shaft on the same side as the barb, which could be successfully palpated using the tip of the 18-gauge needle. The tip of the needle was then used to cap the barb beneath the skin. This technique allowed for the hook to be easily extracted in a retrograde manner without causing further destruction to the surrounding tissue. The patient then was started on prophylaxis cephalexin 500 mg 3 times daily for 3 days.
Comment
The hand is the most common site of fishhook injury, followed closely by the head and eyes.1 Barbless fishhooks usually can be removed by pushing the hook in a retrograde manner along the path of insertion. This method is simple and rarely results in complications. However, there are no guidelines for removal of barbed fishhooks. Furthermore, removing a barbed fishhook in the same retrograde manner would result in extensive internal tissue destruction and increased complications. Due to the popularity of the sport of fishing, fishhook injuries, depending on geographical location, are not uncommon.2 For this reason, trauma and emergency practitioners have become well versed in safe methods for barbed fishhook removal. However, patients are not always able or willing to seek medical care in emergency departments and may opt to seek treatment in outpatient settings, such as in our case. As a result, dermatologists should familiarize themselves with safe and effective fishhook removal methods, as they are not time consuming and do not require complex equipment. Failure to treat the patient may lead to further patient discomfort and increased risk for complications. Additionally, many of the techniques for removal may be useful with other foreign bodies embedded in cutaneous tissue (eg, splinters).
There are a number of safe and effective techniques for removing barbed fishhooks from cutaneous tissue, including the advance-and-cut method, the cut-it-out technique, the string-pull method, and the needle cover technique.1-3 The method chosen to remove the fishhook is dependent on a variety of factors, such as anatomic location, tissue depth, and provider comfort.
With the advance-and-cut method (Figure 2), the affected area is anesthetized and a small incision in the skin is created to expose the barb. The fishhook is then advanced through the incision, providing visibility of the barb and thus allowing the practitioner to cut the barbed tip without creating further damage to the surrounding tissue. The shaft of the fishhook can subsequently be removed in a retrograde fashion. The advantages of this technique include that it may be successfully used in all types of barbed fishhooks and it provides the practitioner with direct visibility of the barb, thus minimizing risk for neurovascular injury during removal.1 However, the primary disadvantage is that a second cutaneous wound is created in exposing the barb.

| 
| |
| Figure 2. The advance-and-cut method for fishhook removal. | Figure 3. The cut-it-out method for fishhook removal. |
|
The cut-it-out technique (Figure 3) is similar to the advance-and-cut method in that they both require anesthesia along with creating an incision. With this method, a scalpel is used to create a small linear incision originating at the fishhook entrance site and ending at the approximated location of the fishhook’s tip. The fishhook then is simply lifted superiorly in a retrograde fashion.
|
The string-pull method (Figure 4) has been credited to fishermen in South Australia and was first described by Cooke2 in 1961. This method is relatively painless, does not require anesthesia, and has a high success rate when properly administered. However, it does require rapid and confident motions (ie, without hesitation) by the practitioner and should not be performed on free-moving areas of the body (eg, earlobe).3 With this technique, a sturdy piece of suture (eg, 2/0 or 3/0 strength silk) is looped around the hook and is extended away from the practitioner at a 30° angle. The free end of the suture is then securely fastened around the index finger of the practitioner’s dominant hand. The index finger of the nondominant hand should apply a downward pressure to the hook shaft to disengage the barb from the tissue. Simultaneously and rather quickly and forcefully the practitioner must pull the dominant index finger with the string attached in a superior and lateral direction, as depicted by the long arrow in Figure 4. If successful, the barbed hook will pull out of the entrance site. The use of string in pulling the fishhook parallel to the site of injury is helpful for smaller fishhooks that may be difficult to grab with fingers alone. However, with larger fishhooks, the string may not be required so long as the practitioner is able to obtain a secure grasp on the fishhook shaft. The string-pull method becomes particularly useful when anesthesia is unavailable or when the barb of the hook is embedded too deeply for safe advancement through tissue to visualize and cut the barb.
|
Lastly, the needle cover technique (Figure 5) is another simple method that does not require the creation of a secondary wound. An 18-gauge needle is simply inserted parallel to the fishhook curvature into the site of entry. By using the needle to slide along the fishhook’s curve, the practitioner is able to follow its pathway while in the tissue. The tip of the 18-gauge needle is then used to cap or cover the barb, thus allowing the fishhook to be removed in a retrograde fashion from the wound. In an outpatient setting, this technique does not require the creation of additional tissue damage and practitioners who are inexperienced with fishhook removal may proceed through the motions more slowly and methodically than the string-pull method permits.
Wound care following fishhook removal should involve adequate flushing of the wound with normal saline along with the application of topical antibiotics and a simple dressing and adhesive bandage. Oral prophylactic antibiotics typically are not required for shallow cutaneous injuries unless the fishhook is dirty, the patient is immunocompromised, or the patient has a condition lending to poor wound healing (eg, diabetes mellitus, peripheral vascular disease).3 When deciding on antibiotics, it is important to note that fishhook injuries while saltwater fishing are associated with Vibrio infection, while injuries sustained during freshwater fishing are associated with gram-negative bacteria (eg, Pseudomonas and Aeromonas species).3 Lastly, it is essential to find out the immunization status of the patient, and tetanus immune globulin should be provided if necessary.

| 
| |
| Figure 4. The string-pull method for fishhook removal. | Figure 5. The needle cover technique for fishhook removal. |
Conclusion
Although guidelines for barbed fishhook removal are not available, outpatient physicians, including dermatologists, should not fear removal procedures. There are many safe and effective fishhook removal methods that are not time consuming and do not require complex equipment. Furthermore, familiarization with these same techniques may be useful for removal of other foreign bodies embedded in cutaneous tissue.
1. Khan HA, Kamal Y, Lone AU. Fish hook injury: removal by “push through and cut off” technique: a case report and brief literature review [published online March 24, 2014]. Trauma Mon. 2014;19:e17728.
2. Cooke T. How to remove fish-hooks with a bit of string. Med J Aust. 1961;48:815-816.
3. Thommasen HV, Thommasen A. The occasional removal of an embedded fish hook. Can J Rural Med. 2005;10:255-259.
1. Khan HA, Kamal Y, Lone AU. Fish hook injury: removal by “push through and cut off” technique: a case report and brief literature review [published online March 24, 2014]. Trauma Mon. 2014;19:e17728.
2. Cooke T. How to remove fish-hooks with a bit of string. Med J Aust. 1961;48:815-816.
3. Thommasen HV, Thommasen A. The occasional removal of an embedded fish hook. Can J Rural Med. 2005;10:255-259.
Practice Points
- Barbed fishhooks should never be removed by pushing the hook in a retrograde manner along the path of insertion, as this method may result in extensive internal tissue destruction and increased complications.
- There are a number of safe and effective techniques for removing barbed fishhooks from cutaneous tissue that also may be applicable in removing other foreign bodies embedded in cutaneous tissue (eg, splinters).
Minimally invasive system smooths cellulite for up to 3 years
WASHINGTON – The key to treating cellulite may be as simple as cutting a tiny tether.
Vertical bundles of fibrous septae that run through subdermal fat and anchor the skin seem to cause the dimples and bulges that annoy up to 95% of women. Releasing those fibers with a minimally invasive instrument causes an immediate smoothing of the skin – an improvement that appears to last for at least 3 years, according to data presented at the annual meeting of the American Academy of Dermatology.
“At 3 years, the data are remarkably consistent,” said Dr. Michael Kaminer of Yale University, New Haven, Conn. “No one was unsatisfied with the result, and in fact, 93% of the patients were still satisfied or very satisfied. So I would say this meets the criteria of something that is reasonable to offer to our patients.
The Cellfina system can treat up to 30 dimples in a single hour-long session, according to Dr. Kaminer, who discussed the procedure in a video interview. The procedure costs anywhere from $2,500-$6,000 per session, depending on how many dimples are treated.
The device consists of a circular area that provides enough suction to elevate the skin over each dimple. One disposable click-in module contains a 22-gauge multibore needle that delivers local anesthetic. After the anesthetic is delivered, a second module clicks into place. This contains an 18-gauge probe tipped with a very small triangular blade. Inserted 5-6 mm below the skin, the probe can be pivoted back and forth, cutting the septae under the dimple. This releases their constricting effect on the skin, which immediately rebounds and appears smooth. The system does not remove any fat.
Cellfina’s earlier data earned it a 2-year Food and Drug Administration clearance for cellulite treatment. The data presented at the meeting confirm its effect at 3 years, Dr. Kaminer said, and form the basis of an application for a 3-year clearance.
The follow-up study comprised 45 women who had undergone the procedure and were reevaluated by several means: a blinded physician rating of before and after pictures, and a 1- 5 point patient self-evaluation scale, with a change of at least 1 point rated as “satisfactory.”
These women represented all six Fitzpatrick skin types. They were a mean of 40 years old at baseline and had a mean body mass index of 25 kg/m2.
Independent, blinded physicians were given before-and-after photos of all patients and asked to identify which photo showed change. All of the physicians correctly identified all of the “after” photos.
“This was not a subtle change they were seeing in the pictures,” Dr. Kaminer said. “It’s a very, very noticeable improvement that a physician who doesn’t treat cellulite would recognize as something having been done.”
At the 1-year follow-up, patients reported an average improvement of 2 points on the severity scale. This was largely maintained at 3 years; 42 patients (93%) remained satisfied. Of the entire group, 19 patients said they were very satisfied, 23 were “satisfied,” and the rest were neutral. No one was unsatisfied, he noted.
“What this means is that about half of patients had an amazing result and the rest had a very good result,” Dr. Kaminer said. He added that none of the women had requested any retreatment. “Most felt like the improvement they experienced was good enough not to need more.”
In fact, he said, the 3-year follow-up photos seem to show that the contours of the treated areas continued to improve. “It certainly looks that way. We have no idea why this would be,” he said but suggested that once women feel better about the appearance of their legs and rear, they may be more motivated to make such beneficial lifestyle changes as improved diet and regular exercise.
The system has mostly been used for buttock cellulite, but that’s expanding. Dr. Kaminer said. “We are treating a lot of outer thighs now, and we’re starting to move into anterior thighs and seeing excellent results.”
He added that the procedure is very safe and quite easy for patients to tolerate. Postprocedural pain is minimal and managed with nonprescription acetaminophen. Aftercare consists only of wearing a light compression garment – like a body shaper – for a week after the procedure. Adverse events included slight ecchymosis that cleared rapidly; one patient experienced scratching for pruritus.
Some women, however, will experience a residual firmness in treated areas. It’s not clear what this is, Dr. Kaminer said, but it recedes spontaneously over 6-12 months. “If it’s really bothering them, you can inject a little steroid into it. It can sometimes go away with massage.”
Informing patients about the possibility is key, he added. “If you tell them they might experience it, it’s really not a big deal to them.”
Dr. Kaminer received financial benefits during the initial development and deployment of Cellfina, but said he has no financial ties to the company now.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
WASHINGTON – The key to treating cellulite may be as simple as cutting a tiny tether.
Vertical bundles of fibrous septae that run through subdermal fat and anchor the skin seem to cause the dimples and bulges that annoy up to 95% of women. Releasing those fibers with a minimally invasive instrument causes an immediate smoothing of the skin – an improvement that appears to last for at least 3 years, according to data presented at the annual meeting of the American Academy of Dermatology.
“At 3 years, the data are remarkably consistent,” said Dr. Michael Kaminer of Yale University, New Haven, Conn. “No one was unsatisfied with the result, and in fact, 93% of the patients were still satisfied or very satisfied. So I would say this meets the criteria of something that is reasonable to offer to our patients.
The Cellfina system can treat up to 30 dimples in a single hour-long session, according to Dr. Kaminer, who discussed the procedure in a video interview. The procedure costs anywhere from $2,500-$6,000 per session, depending on how many dimples are treated.
The device consists of a circular area that provides enough suction to elevate the skin over each dimple. One disposable click-in module contains a 22-gauge multibore needle that delivers local anesthetic. After the anesthetic is delivered, a second module clicks into place. This contains an 18-gauge probe tipped with a very small triangular blade. Inserted 5-6 mm below the skin, the probe can be pivoted back and forth, cutting the septae under the dimple. This releases their constricting effect on the skin, which immediately rebounds and appears smooth. The system does not remove any fat.
Cellfina’s earlier data earned it a 2-year Food and Drug Administration clearance for cellulite treatment. The data presented at the meeting confirm its effect at 3 years, Dr. Kaminer said, and form the basis of an application for a 3-year clearance.
The follow-up study comprised 45 women who had undergone the procedure and were reevaluated by several means: a blinded physician rating of before and after pictures, and a 1- 5 point patient self-evaluation scale, with a change of at least 1 point rated as “satisfactory.”
These women represented all six Fitzpatrick skin types. They were a mean of 40 years old at baseline and had a mean body mass index of 25 kg/m2.
Independent, blinded physicians were given before-and-after photos of all patients and asked to identify which photo showed change. All of the physicians correctly identified all of the “after” photos.
“This was not a subtle change they were seeing in the pictures,” Dr. Kaminer said. “It’s a very, very noticeable improvement that a physician who doesn’t treat cellulite would recognize as something having been done.”
At the 1-year follow-up, patients reported an average improvement of 2 points on the severity scale. This was largely maintained at 3 years; 42 patients (93%) remained satisfied. Of the entire group, 19 patients said they were very satisfied, 23 were “satisfied,” and the rest were neutral. No one was unsatisfied, he noted.
“What this means is that about half of patients had an amazing result and the rest had a very good result,” Dr. Kaminer said. He added that none of the women had requested any retreatment. “Most felt like the improvement they experienced was good enough not to need more.”
In fact, he said, the 3-year follow-up photos seem to show that the contours of the treated areas continued to improve. “It certainly looks that way. We have no idea why this would be,” he said but suggested that once women feel better about the appearance of their legs and rear, they may be more motivated to make such beneficial lifestyle changes as improved diet and regular exercise.
The system has mostly been used for buttock cellulite, but that’s expanding. Dr. Kaminer said. “We are treating a lot of outer thighs now, and we’re starting to move into anterior thighs and seeing excellent results.”
He added that the procedure is very safe and quite easy for patients to tolerate. Postprocedural pain is minimal and managed with nonprescription acetaminophen. Aftercare consists only of wearing a light compression garment – like a body shaper – for a week after the procedure. Adverse events included slight ecchymosis that cleared rapidly; one patient experienced scratching for pruritus.
Some women, however, will experience a residual firmness in treated areas. It’s not clear what this is, Dr. Kaminer said, but it recedes spontaneously over 6-12 months. “If it’s really bothering them, you can inject a little steroid into it. It can sometimes go away with massage.”
Informing patients about the possibility is key, he added. “If you tell them they might experience it, it’s really not a big deal to them.”
Dr. Kaminer received financial benefits during the initial development and deployment of Cellfina, but said he has no financial ties to the company now.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
WASHINGTON – The key to treating cellulite may be as simple as cutting a tiny tether.
Vertical bundles of fibrous septae that run through subdermal fat and anchor the skin seem to cause the dimples and bulges that annoy up to 95% of women. Releasing those fibers with a minimally invasive instrument causes an immediate smoothing of the skin – an improvement that appears to last for at least 3 years, according to data presented at the annual meeting of the American Academy of Dermatology.
“At 3 years, the data are remarkably consistent,” said Dr. Michael Kaminer of Yale University, New Haven, Conn. “No one was unsatisfied with the result, and in fact, 93% of the patients were still satisfied or very satisfied. So I would say this meets the criteria of something that is reasonable to offer to our patients.
The Cellfina system can treat up to 30 dimples in a single hour-long session, according to Dr. Kaminer, who discussed the procedure in a video interview. The procedure costs anywhere from $2,500-$6,000 per session, depending on how many dimples are treated.
The device consists of a circular area that provides enough suction to elevate the skin over each dimple. One disposable click-in module contains a 22-gauge multibore needle that delivers local anesthetic. After the anesthetic is delivered, a second module clicks into place. This contains an 18-gauge probe tipped with a very small triangular blade. Inserted 5-6 mm below the skin, the probe can be pivoted back and forth, cutting the septae under the dimple. This releases their constricting effect on the skin, which immediately rebounds and appears smooth. The system does not remove any fat.
Cellfina’s earlier data earned it a 2-year Food and Drug Administration clearance for cellulite treatment. The data presented at the meeting confirm its effect at 3 years, Dr. Kaminer said, and form the basis of an application for a 3-year clearance.
The follow-up study comprised 45 women who had undergone the procedure and were reevaluated by several means: a blinded physician rating of before and after pictures, and a 1- 5 point patient self-evaluation scale, with a change of at least 1 point rated as “satisfactory.”
These women represented all six Fitzpatrick skin types. They were a mean of 40 years old at baseline and had a mean body mass index of 25 kg/m2.
Independent, blinded physicians were given before-and-after photos of all patients and asked to identify which photo showed change. All of the physicians correctly identified all of the “after” photos.
“This was not a subtle change they were seeing in the pictures,” Dr. Kaminer said. “It’s a very, very noticeable improvement that a physician who doesn’t treat cellulite would recognize as something having been done.”
At the 1-year follow-up, patients reported an average improvement of 2 points on the severity scale. This was largely maintained at 3 years; 42 patients (93%) remained satisfied. Of the entire group, 19 patients said they were very satisfied, 23 were “satisfied,” and the rest were neutral. No one was unsatisfied, he noted.
“What this means is that about half of patients had an amazing result and the rest had a very good result,” Dr. Kaminer said. He added that none of the women had requested any retreatment. “Most felt like the improvement they experienced was good enough not to need more.”
In fact, he said, the 3-year follow-up photos seem to show that the contours of the treated areas continued to improve. “It certainly looks that way. We have no idea why this would be,” he said but suggested that once women feel better about the appearance of their legs and rear, they may be more motivated to make such beneficial lifestyle changes as improved diet and regular exercise.
The system has mostly been used for buttock cellulite, but that’s expanding. Dr. Kaminer said. “We are treating a lot of outer thighs now, and we’re starting to move into anterior thighs and seeing excellent results.”
He added that the procedure is very safe and quite easy for patients to tolerate. Postprocedural pain is minimal and managed with nonprescription acetaminophen. Aftercare consists only of wearing a light compression garment – like a body shaper – for a week after the procedure. Adverse events included slight ecchymosis that cleared rapidly; one patient experienced scratching for pruritus.
Some women, however, will experience a residual firmness in treated areas. It’s not clear what this is, Dr. Kaminer said, but it recedes spontaneously over 6-12 months. “If it’s really bothering them, you can inject a little steroid into it. It can sometimes go away with massage.”
Informing patients about the possibility is key, he added. “If you tell them they might experience it, it’s really not a big deal to them.”
Dr. Kaminer received financial benefits during the initial development and deployment of Cellfina, but said he has no financial ties to the company now.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT AAD 2016
Key clinical point: The Cellfina cellulite treatment system appears to confer a lasting benefit.
Major finding: Three years after the procedure, 93% of women were either “satisfied” or “very satisfied” with their results.
Data source: The follow-up study comprised 45 women.
Disclosures: Dr. Kaminer had financial ties with the company during the product’s development and early deployment. He is no longer associated with it.
Therapies to Improve the Cosmetic Symptoms of Atopic Dermatitis
Atopic dermatitis (AD), more commonly referred to as eczema, is a chronic pruritic inflammatory skin disease that frequently affects both children and adults. Atopic dermatitis is most common in urban and developed countries, with a prevalence of approximately 11% in the United States.1 The pathophysiology of AD is complex and not fully understood, despite the increasing incidence of the disease.2 A myriad of factors, including genetics, defects in the innate and adaptive immune response, and skin barrier abnormalities all contribute to the pathogenesis.3,4 As a result of these abnormalities, patients with AD are more prone to damage from environmental irritants and allergens.
The diagnosis of AD is made clinically based on patient history and visual assessment of the skin.5 Atopic dermatitis follows a chronic and relapsing course characterized by severe pruritus and visible skin changes including xerosis, redness, blistering, oozing, crusting, scaling, thickening, and color change.6,7 Due to the genetic predisposition to make IgE antibodies in response to common environmental and food antigens, patients also may develop allergic rhinitis, asthma, and food-induced anaphylaxis.8,9 Patients also are susceptible to cutaneous viral, fungal, and bacterial infections, the most common of which is an infection with Staphylococcus aureus.10
Atopic dermatitis can have a substantial impact on quality of life, which has been revealed in studies linking chronic skin conditions to depression, impairment of self-esteem, and financial hardship.11 Because skin appearance impacts how a person is initially perceived by others, patients often report feeling self-conscious about their disease and experience teasing or bullying.12 To improve their physical appearance, patients may incur considerable medical expenses. According to 2 population-based studies comprising more than 60,000 adults aged 18 to 85 years, individuals with AD face substantial financial burdens and utilize the health care system more than those without the disease. On average, patients with AD spend $371 to $489 per year on costly out-of-pocket medical expenses and report more absences from work.13
Although there currently is no cure for AD, treatment is aimed at relieving its symptoms and preventing acute exacerbations as well as improving cosmetic appearance to enhance quality of life. Treatment must follow a stepwise approach, which focuses on hydrating the skin, repairing the dysfunctional epithelial barrier, and controlling inflammation. Thus, the standard of care focuses on avoiding skin irritants and triggers along with the use of moisturizers and topical corticosteroids (TCs). In patients with recurring severe disease, topical calcineurin inhibitors, phototherapy, and systemic agents also may be utilized.14
Avoiding Irritants and Triggers
Atopic dermatitis is worsened by skin contact with physical and chemical irritants. Exacerbating factors in AD include exposure to food allergens, dust, emotional stress, detergents, fragranced soaps, textiles, and ingredients in cosmetic products. Patients should be advised to use mild detergents and fragrance-free soaps and to avoid harsh materials such as wool. However, avoidance of specific ingredients in cosmetic products is not as straightforward because manufacturers are not required to disclose certain ingredients. In general, fragrances such as balsam of Peru and cinnamaldehyde, as well as preservatives such as parabens, isothiazolinones, and formaldehyde, should be avoided when selecting cosmetic products. Patients with AD should purchase fragrance-free products that are specifically formulated for sensitive skin. Additionally, patients should not apply makeup if their skin is irritated or oozing, as the flare may worsen.15
Moisturizers
Due to the impaired skin barrier function in patients with AD, regular application of fragrance-free moisturizers is essential to maintain hydration and to reduce xerosis. Various classes of moisturizers may be prescribed (eg, lotions, creams, gels, ointments) based on disease severity and patient preference. Light preparations such as lotions, creams, and gels have a high water content and generally are more appealing from a cosmetic standpoint because they do not create any residue on the skin. However, these options may require more frequent application because they are absorbed quickly. Heavy preparations such as ointments have longer-lasting effects due to their high oil content but tend to be less cosmetically appealing because of their greasiness.16
Although the amount and frequency of application of moisturizers has not been defined, liberal application several times daily is generally advised to minimize xerosis.17 Most physicians recommend applying moisturizer to the skin immediately after bathing to seal in moisture. Some patients prefer to use lotions and creams during the day because these products make the skin feel smooth and reserve the greasier ointments for nighttime application.
Topical Corticosteroids
Prescribed in conjunction with moisturizers, TCs are the mainstay of anti-inflammatory therapy in AD. Topical corticosteroids are classified into 7 groups based on potency, ranging from superpotent (class 1) to least potent (class 7). For acute AD flares, TCs should be applied daily for up to several weeks. Once the inflammation has resolved, it is recommended to apply TCs once to twice weekly to reduce the rate of relapse.18 Despite their effectiveness in the treatment of acute AD flares, TCs have a considerable side-effect profile. Potential adverse effects include skin atrophy, striae, telangiectasia, hypopigmentation, increased hair growth, steroid acne, growth retardation, and Cushing syndrome. Skin atrophy, which is the most common complication associated with TCs, results in shiny transparent skin, allowing for visualization of veins.19,20 Although many of these side effects will resolve after discontinuing the TCs, they are aesthetically displeasing during treatment, making it crucial for physicians to educate their patients on the proper usage of TCs to prevent negative outcomes.
Topical Calcineurin Inhibitors
Topical calcineurin inhibitors (TCIs) are a class of anti-inflammatories that are used to overcome the adverse effects of TCs. They are approved as alternatives to TCs in patients who have failed to respond to other topical treatments as well as those who have developed cutaneous atrophy from the use of TCs or have AD in sensitive areas such as the face, neck, and/or skin folds. Unlike TCs, TCIs do not cause atrophy, striae, or discoloration of the skin, which makes them more desirable from a cosmetic perspective. Their mechanism of action is distinct from TCs in that they inhibit calcineurin-dependent T-cell activation, thus preventing the transcription of inflammatory cytokines.21 Two TCIs are currently available: tacrolimus ointment 0.03% and 0.1% concentrations for moderate to severe AD and pimecrolimus cream 1% for mild to moderate AD.22 Twice-daily application of TCIs is recommended to decrease inflammation and pruritus associated with AD. Studies also have shown that intermittent use of TCIs 3 times weekly can aid in reducing relapses.23-25
The results from clinical trials demonstrate the rapid and continuous effects of both pimecrolimus and tacrolimus. In a controlled long-term study of adults, pimecrolimus provided significant relief of pruritus as soon as day 3 (P<.001).26,27 Pimecrolimus also provides long-term relief by preventing disease progression to flares, which was exemplified in a study (N=713) with no flares in 51% of pimecrolimus patients at 12 months versus 28% in the conventional treatment group (P<.001).28 Similarly, long-term studies of tacrolimus demonstrated an improvement of all symptoms of AD after 1 week of treatment. Maximal improvement was achieved with continued use of tacrolimus, and up to 1 year of tacrolimus use was found to be safe and effective.29,30 Thus, TCIs have been proven to be an effective choice in maintenance therapy for AD and have a good safety profile. The most common adverse effects of TCIs are local skin reactions, such as stinging and burning at the site of application. Rare cases of skin cancer and lymphoma have been reported; however, a causal relationship has yet to be established.31,32
Additional Therapies
Wet wrap therapy is effective for rapid control of flares and in controlling recalcitrant AD. Wet wraps function via several mechanisms; they provide a mechanical barrier against scratching, increase moisture and soften the skin, and enhance absorption of topical medications.33,34 The following method is employed when using wet wraps: an emollient or TC is applied to the area, a tubular bandage soaked in warm water is wrapped over the area, and dry bandages are used to form the outermost layer. Although wet wrap therapy is beneficial in treating AD, it is labor intensive and may require the expertise of a nurse. Thus, unlike other therapies, which patients can easily apply without interfering with their day, wet wraps must be applied at home or in a hospital setting.
Light therapy is another effective method of controlling AD. Although multiple forms of UV phototherapy are beneficial for symptom control in AD, there is no definitive recommendation regarding the specific type of light therapy due to a lack of comparative studies. Natural sunlight, narrowband UVB, broadband UVB, UVA, oral or topical psoralen plus UVA, as well as UVA and UVB can all be utilized in the treatment of AD. However, similar to natural sunlight, artificial light therapy can cause burning, blistering, hyperpigmentation, dark spots, and wrinkles. Because society places a large emphasis on maintaining a youthful appearance, patients may be hesitant to use a treatment that could potentially advance the skin’s aging process. Thus, it is important that this therapy is properly controlled to prevent further skin damage.35-37
When optimal topical regimens and phototherapy have failed to control AD, systemic immunomodulation therapies may be used. Currently, the most commonly used medications are cyclosporine 150 to 300 mg daily, methotrexate 7.5 to 25 mg weekly, mycophenolate mofetil 0.5 to 3 g daily, and azathioprine 1 to 3 mg/kg daily.38,39 Decisions regarding the specific class of drugs should be based on the patient’s AD status, comorbidities, and personal preference.
Conclusion
Atopic dermatitis is a common chronic condition that can occur at any age and cause substantial physical, psychological, social, and/or emotional stress for patients and their families. Although TCs have been the standard of treatment for many years, ongoing concerns regarding their safety have led to the use of TCIs, which overcome some of the drawbacks of steroid therapy. Phototherapy and systemic immunosuppressant therapy are reserved for patients who have not responded to optimal topical therapies. Although several therapeutic avenues exist for patients, there is a need for the development of more effective and safer drugs. Furthermore, cosmetic products created specifically for patients with AD would be beneficial, as patients often struggle to select products that do not cause more harm than good. Given the complexity of the pathogenesis of AD, further research must focus on defining the specific pathways involved in the disease and targeting these pathways with therapies.
1. Shaw TE, Currie GP, Koudelka CW, et al. Eczema prevalence in the United States: data from the 2003 National Survey of Children’s Health. J Invest Dermatol. 2011;131:67-73.
2. Deckers IA, McLean S, Linssen S, et al. Investigating international time trends in the incidence and prevalence of atopic eczema 1990-2010: a systematic review of epidemiological studies. PLoS One. 2012;7:e39803.
3. Boguniewicz M, Leung DY. Atopic dermatitis: a disease of altered skin barrier and immune dysregulation. Immunol Rev. 2011;242:233-246.
4. Peate I. Eczema: causes, symptoms and treatment in the community. Br J Community Nurs. 2011;16:324, 326-331.
5. Williams HC, Burney PG, Pembroke AC, et al. The U.K. Working Party’s diagnostic criteria for atopic dermatitis. III. independent hospital validation. Br J Dermatol. 1994;131:406-416.
6. Magin P, Adams J, Heading G, et al. Experiences of appearance-related teasing and bullying in skin diseases and their psychological sequelae: results of a qualitative study. Scand J Caring Sci. 2008;22:430-436.
7. Beattie P, Lewis-Jones M. A comparative study of impairment of quality of life in children with skin disease and children with other chronic childhood diseases. Br J Dermatol. 2006;155:145-151.
8. Spergel JM. From atopic dermatitis to asthma: the atopic march [published online January 22, 2010]. Ann Allergy Asthma Immunol. 2010;105:99-106; quiz 107-109, 117.
9. Leung DY. New insights into atopic dermatitis: role of skin barrier and immune dysregulation. Allergol Int. 2013;62:151-161.
10. Balma-Mena A, Lara-Corrales I, Zeller J, et al. Colonization with community-acquired methicillin-resistant Staphylococcus aureus in children with atopic dermatitis: a cross-sectional study. Int J Dermatol. 2011;50:682-688.
11. Strawser MS, Storch EA, Roberti JW. The Teasing Questionnaire-Revised: measurement of childhood teasing in adults. J Anxiety Disord. 2005;19:780-792.
12. Magin P, Adams J, Heading G, et al. Experiences of appearance-related teasing and bullying in skin diseases and their psychological sequelae: results of a qualitative study. Scand J Caring Sci. 2008;22:430-436.
13. Silverberg J. Health care utilization, patient costs, and access to care in US adults with eczema: a population-based study. JAMA Dermatol. 2015;151:743-752.
14. Ellis C, Luger T, Abeck D, et al. International Consensus Conference on Atopic Dermatitis II (ICCAD II): clinical update and current treatment strategies. Br J Dermatol. 2003;148(suppl 63):3-10.
15. Kim K. Influences of environmental chemicals on atopic dermatitis. Toxicol Res. 2015;31:89-96.
16. Ridd M, Redmond N, Hollinghurst S, et al. Choice of Moisturiser for Eczema Treatment (COMET): study protocol for a randomized controlled trial. Trials. 2015;16:304.
17. Hon KL, Ching GK, Leung TF, et al. Estimating emollient usage in patients with eczema. Clin Exp Dermatol. 2010;35:22-26.
18. Hanifin J, Gupta AK, Rajagopalan R. Intermittent dosing of fluticasone propionate cream for reducing the risk of relapse in atopic dermatitis patients. Br J Dermatol. 2002;147:528-537.
19. Hill CJ, Rostenberg A Jr. Adverse effects from topical steroids. Cutis. 1978;21:624-628.
20. Ruiz-Maldonado R, Zapata G, Lourdes T, et al. Cushing’s syndrome after topical application of corticosteroids. Am J Dis Child. 1982;136:274-275.
21. Grassberger M, Baumruker T, Enz A, et al. A novel anti-inflammatory drug, SDZ ASM 981, for the treatment of skin diseases: in vitro pharmacology. Br J Dermatol. 1999;141:264-273.
22. Eichenfield L, Wynnis T, Berger T. Guidelines of care for the management of atopic dermatitis: management and treatment of atopic dermatitis with topical therapies. J Am Acad Dermatol. 2014;71:116-132.
23. Reitamo S, Harper J, Bos JD, et al. 0.03% Tacrolimus ointment applied once or twice daily is more efficacious than 1% hydrocortisone acetate in children with moderate to severe atopic dermatitis: results of a randomized double-blind controlled trial. Br J Dermatol. 2004;150:554-562.
24. Ruer-Mulard M, Aberer W, Gunstone A, et al. Twice-daily versus once-daily applications of pimecrolimus cream 1% for the prevention of disease relapse in pediatric patients with atopic dermatitis. Pediatr Dermatol. 2009;26:551-558.
25. Breneman D, Fleischer AB Jr, Abramovits W, et al. Intermittent therapy for flare prevention and long-term disease control in stabilized atopic dermatitis: a randomized comparison of 3-times-weekly applications of tacrolimus ointment versus vehicle. J Am Acad Dermatol. 2008;58:990-999.
26. Meurer M, Fölster-Holst R, Wozel G, et al. Pimecrolimus cream 1% (Elidel) provides significant and rapid relief of pruritus and improves disease control and quality of life in atopic dermatitis in adults. J Invest Dermatol. 2002;119:350.
27. Meurer M, Fölster-Holst R, Wozel G, et al. Pimecrolimus cream in the long-term management of atopic dermatitis in adults: a six-month study. Dermatology. 2002;205:271-277.
28. Wahn U, Bos JD, Goodfield M, et al. Efficacy and safety of pimecrolimus cream in the long-term management of atopic dermatitis in children. Pediatrics. 2002;110(1, pt 1):e2.
29. Kang S, Lucky AW, Pariser D, et al. Long-term safety and efficacy of tacrolimus ointment for the treatment of atopic dermatitis in children. J Am Acad Dermatol. 2001;44(suppl 1):S58-S64.
30. Reitamo S, Wollenberg A, Schöpf E, et al. Safety and efficacy of 1 year of tacrolimus ointment monotherapy in adults with atopic dermatitis. the European Tacrolimus Ointment Study Group. Arch Dermatol. 2000;136:999-1006.
31. Frankel HC, Qureshi AA. Comparative effectiveness of topical calcineurin inhibitors in adult patients with atopic dermatitis. Am J Clin Dermatol. 2012;13:113-123.
32. Tennis P, Gelfand JM, Rothman KJ. Evaluation of cancer risk related to atopic dermatitis and use of topical calcineurin inhibitors. Br J Dermatol. 2011;165:465-473.
33. Dabade TS, Davis DM, Wetter DA, et al. Wet dressing therapy in conjunction with topical corticosteroids is effective for rapid control of severe pediatric atopic dermatitis: experience with 218 patients over 30 years at Mayo Clinic. J Am Acad Dermatol. 2012;67:100-106.
34. Devillers AC, Oranje AP. Efficacy and safety of ‘wet-wrap’ dressings as an intervention treatment in children with severe and/or refractory atopic dermatitis: a critical review of the literature. Br J Dermatol. 2006;154:579-585.
35. Meduri NB, Vandergriff T, Rasmussen H, et al. Phototherapy in the management of atopic dermatitis: a systematic review. Photodermatol Photoimmunol Photomed. 2007;23:106-112.
36. Clayton TH, Clark SM, Turner D, et al. The treatment of severe atopic dermatitis in childhood with narrowband ultraviolet B phototherapy. Clin Exp Dermatol. 2007;32:28-33.
37. Jekler J, Larko O. UVB phototherapy of atopic dermatitis. Br J Dermatol. 1988;119:697-705.
38. Roekevisch E, Spuls PI, Kuester D, et al. Efficacy and safety of systemic treatments for moderate-to-severe atopic dermatitis: a systematic review. J Allergy Clin Immunol. 2014;133:429-438.39. Hoare C, Li Wan Po A, Williams H. Systematic review of treatments for atopic eczema. Health Technol Assess. 2000;4:1-191.
39. Hoare C, Li Wan Po A, Williams H. Systematic review of treatments for atopic eczema. Health Technol Assess. 2000;4:1-191.
Atopic dermatitis (AD), more commonly referred to as eczema, is a chronic pruritic inflammatory skin disease that frequently affects both children and adults. Atopic dermatitis is most common in urban and developed countries, with a prevalence of approximately 11% in the United States.1 The pathophysiology of AD is complex and not fully understood, despite the increasing incidence of the disease.2 A myriad of factors, including genetics, defects in the innate and adaptive immune response, and skin barrier abnormalities all contribute to the pathogenesis.3,4 As a result of these abnormalities, patients with AD are more prone to damage from environmental irritants and allergens.
The diagnosis of AD is made clinically based on patient history and visual assessment of the skin.5 Atopic dermatitis follows a chronic and relapsing course characterized by severe pruritus and visible skin changes including xerosis, redness, blistering, oozing, crusting, scaling, thickening, and color change.6,7 Due to the genetic predisposition to make IgE antibodies in response to common environmental and food antigens, patients also may develop allergic rhinitis, asthma, and food-induced anaphylaxis.8,9 Patients also are susceptible to cutaneous viral, fungal, and bacterial infections, the most common of which is an infection with Staphylococcus aureus.10
Atopic dermatitis can have a substantial impact on quality of life, which has been revealed in studies linking chronic skin conditions to depression, impairment of self-esteem, and financial hardship.11 Because skin appearance impacts how a person is initially perceived by others, patients often report feeling self-conscious about their disease and experience teasing or bullying.12 To improve their physical appearance, patients may incur considerable medical expenses. According to 2 population-based studies comprising more than 60,000 adults aged 18 to 85 years, individuals with AD face substantial financial burdens and utilize the health care system more than those without the disease. On average, patients with AD spend $371 to $489 per year on costly out-of-pocket medical expenses and report more absences from work.13
Although there currently is no cure for AD, treatment is aimed at relieving its symptoms and preventing acute exacerbations as well as improving cosmetic appearance to enhance quality of life. Treatment must follow a stepwise approach, which focuses on hydrating the skin, repairing the dysfunctional epithelial barrier, and controlling inflammation. Thus, the standard of care focuses on avoiding skin irritants and triggers along with the use of moisturizers and topical corticosteroids (TCs). In patients with recurring severe disease, topical calcineurin inhibitors, phototherapy, and systemic agents also may be utilized.14
Avoiding Irritants and Triggers
Atopic dermatitis is worsened by skin contact with physical and chemical irritants. Exacerbating factors in AD include exposure to food allergens, dust, emotional stress, detergents, fragranced soaps, textiles, and ingredients in cosmetic products. Patients should be advised to use mild detergents and fragrance-free soaps and to avoid harsh materials such as wool. However, avoidance of specific ingredients in cosmetic products is not as straightforward because manufacturers are not required to disclose certain ingredients. In general, fragrances such as balsam of Peru and cinnamaldehyde, as well as preservatives such as parabens, isothiazolinones, and formaldehyde, should be avoided when selecting cosmetic products. Patients with AD should purchase fragrance-free products that are specifically formulated for sensitive skin. Additionally, patients should not apply makeup if their skin is irritated or oozing, as the flare may worsen.15
Moisturizers
Due to the impaired skin barrier function in patients with AD, regular application of fragrance-free moisturizers is essential to maintain hydration and to reduce xerosis. Various classes of moisturizers may be prescribed (eg, lotions, creams, gels, ointments) based on disease severity and patient preference. Light preparations such as lotions, creams, and gels have a high water content and generally are more appealing from a cosmetic standpoint because they do not create any residue on the skin. However, these options may require more frequent application because they are absorbed quickly. Heavy preparations such as ointments have longer-lasting effects due to their high oil content but tend to be less cosmetically appealing because of their greasiness.16
Although the amount and frequency of application of moisturizers has not been defined, liberal application several times daily is generally advised to minimize xerosis.17 Most physicians recommend applying moisturizer to the skin immediately after bathing to seal in moisture. Some patients prefer to use lotions and creams during the day because these products make the skin feel smooth and reserve the greasier ointments for nighttime application.
Topical Corticosteroids
Prescribed in conjunction with moisturizers, TCs are the mainstay of anti-inflammatory therapy in AD. Topical corticosteroids are classified into 7 groups based on potency, ranging from superpotent (class 1) to least potent (class 7). For acute AD flares, TCs should be applied daily for up to several weeks. Once the inflammation has resolved, it is recommended to apply TCs once to twice weekly to reduce the rate of relapse.18 Despite their effectiveness in the treatment of acute AD flares, TCs have a considerable side-effect profile. Potential adverse effects include skin atrophy, striae, telangiectasia, hypopigmentation, increased hair growth, steroid acne, growth retardation, and Cushing syndrome. Skin atrophy, which is the most common complication associated with TCs, results in shiny transparent skin, allowing for visualization of veins.19,20 Although many of these side effects will resolve after discontinuing the TCs, they are aesthetically displeasing during treatment, making it crucial for physicians to educate their patients on the proper usage of TCs to prevent negative outcomes.
Topical Calcineurin Inhibitors
Topical calcineurin inhibitors (TCIs) are a class of anti-inflammatories that are used to overcome the adverse effects of TCs. They are approved as alternatives to TCs in patients who have failed to respond to other topical treatments as well as those who have developed cutaneous atrophy from the use of TCs or have AD in sensitive areas such as the face, neck, and/or skin folds. Unlike TCs, TCIs do not cause atrophy, striae, or discoloration of the skin, which makes them more desirable from a cosmetic perspective. Their mechanism of action is distinct from TCs in that they inhibit calcineurin-dependent T-cell activation, thus preventing the transcription of inflammatory cytokines.21 Two TCIs are currently available: tacrolimus ointment 0.03% and 0.1% concentrations for moderate to severe AD and pimecrolimus cream 1% for mild to moderate AD.22 Twice-daily application of TCIs is recommended to decrease inflammation and pruritus associated with AD. Studies also have shown that intermittent use of TCIs 3 times weekly can aid in reducing relapses.23-25
The results from clinical trials demonstrate the rapid and continuous effects of both pimecrolimus and tacrolimus. In a controlled long-term study of adults, pimecrolimus provided significant relief of pruritus as soon as day 3 (P<.001).26,27 Pimecrolimus also provides long-term relief by preventing disease progression to flares, which was exemplified in a study (N=713) with no flares in 51% of pimecrolimus patients at 12 months versus 28% in the conventional treatment group (P<.001).28 Similarly, long-term studies of tacrolimus demonstrated an improvement of all symptoms of AD after 1 week of treatment. Maximal improvement was achieved with continued use of tacrolimus, and up to 1 year of tacrolimus use was found to be safe and effective.29,30 Thus, TCIs have been proven to be an effective choice in maintenance therapy for AD and have a good safety profile. The most common adverse effects of TCIs are local skin reactions, such as stinging and burning at the site of application. Rare cases of skin cancer and lymphoma have been reported; however, a causal relationship has yet to be established.31,32
Additional Therapies
Wet wrap therapy is effective for rapid control of flares and in controlling recalcitrant AD. Wet wraps function via several mechanisms; they provide a mechanical barrier against scratching, increase moisture and soften the skin, and enhance absorption of topical medications.33,34 The following method is employed when using wet wraps: an emollient or TC is applied to the area, a tubular bandage soaked in warm water is wrapped over the area, and dry bandages are used to form the outermost layer. Although wet wrap therapy is beneficial in treating AD, it is labor intensive and may require the expertise of a nurse. Thus, unlike other therapies, which patients can easily apply without interfering with their day, wet wraps must be applied at home or in a hospital setting.
Light therapy is another effective method of controlling AD. Although multiple forms of UV phototherapy are beneficial for symptom control in AD, there is no definitive recommendation regarding the specific type of light therapy due to a lack of comparative studies. Natural sunlight, narrowband UVB, broadband UVB, UVA, oral or topical psoralen plus UVA, as well as UVA and UVB can all be utilized in the treatment of AD. However, similar to natural sunlight, artificial light therapy can cause burning, blistering, hyperpigmentation, dark spots, and wrinkles. Because society places a large emphasis on maintaining a youthful appearance, patients may be hesitant to use a treatment that could potentially advance the skin’s aging process. Thus, it is important that this therapy is properly controlled to prevent further skin damage.35-37
When optimal topical regimens and phototherapy have failed to control AD, systemic immunomodulation therapies may be used. Currently, the most commonly used medications are cyclosporine 150 to 300 mg daily, methotrexate 7.5 to 25 mg weekly, mycophenolate mofetil 0.5 to 3 g daily, and azathioprine 1 to 3 mg/kg daily.38,39 Decisions regarding the specific class of drugs should be based on the patient’s AD status, comorbidities, and personal preference.
Conclusion
Atopic dermatitis is a common chronic condition that can occur at any age and cause substantial physical, psychological, social, and/or emotional stress for patients and their families. Although TCs have been the standard of treatment for many years, ongoing concerns regarding their safety have led to the use of TCIs, which overcome some of the drawbacks of steroid therapy. Phototherapy and systemic immunosuppressant therapy are reserved for patients who have not responded to optimal topical therapies. Although several therapeutic avenues exist for patients, there is a need for the development of more effective and safer drugs. Furthermore, cosmetic products created specifically for patients with AD would be beneficial, as patients often struggle to select products that do not cause more harm than good. Given the complexity of the pathogenesis of AD, further research must focus on defining the specific pathways involved in the disease and targeting these pathways with therapies.
Atopic dermatitis (AD), more commonly referred to as eczema, is a chronic pruritic inflammatory skin disease that frequently affects both children and adults. Atopic dermatitis is most common in urban and developed countries, with a prevalence of approximately 11% in the United States.1 The pathophysiology of AD is complex and not fully understood, despite the increasing incidence of the disease.2 A myriad of factors, including genetics, defects in the innate and adaptive immune response, and skin barrier abnormalities all contribute to the pathogenesis.3,4 As a result of these abnormalities, patients with AD are more prone to damage from environmental irritants and allergens.
The diagnosis of AD is made clinically based on patient history and visual assessment of the skin.5 Atopic dermatitis follows a chronic and relapsing course characterized by severe pruritus and visible skin changes including xerosis, redness, blistering, oozing, crusting, scaling, thickening, and color change.6,7 Due to the genetic predisposition to make IgE antibodies in response to common environmental and food antigens, patients also may develop allergic rhinitis, asthma, and food-induced anaphylaxis.8,9 Patients also are susceptible to cutaneous viral, fungal, and bacterial infections, the most common of which is an infection with Staphylococcus aureus.10
Atopic dermatitis can have a substantial impact on quality of life, which has been revealed in studies linking chronic skin conditions to depression, impairment of self-esteem, and financial hardship.11 Because skin appearance impacts how a person is initially perceived by others, patients often report feeling self-conscious about their disease and experience teasing or bullying.12 To improve their physical appearance, patients may incur considerable medical expenses. According to 2 population-based studies comprising more than 60,000 adults aged 18 to 85 years, individuals with AD face substantial financial burdens and utilize the health care system more than those without the disease. On average, patients with AD spend $371 to $489 per year on costly out-of-pocket medical expenses and report more absences from work.13
Although there currently is no cure for AD, treatment is aimed at relieving its symptoms and preventing acute exacerbations as well as improving cosmetic appearance to enhance quality of life. Treatment must follow a stepwise approach, which focuses on hydrating the skin, repairing the dysfunctional epithelial barrier, and controlling inflammation. Thus, the standard of care focuses on avoiding skin irritants and triggers along with the use of moisturizers and topical corticosteroids (TCs). In patients with recurring severe disease, topical calcineurin inhibitors, phototherapy, and systemic agents also may be utilized.14
Avoiding Irritants and Triggers
Atopic dermatitis is worsened by skin contact with physical and chemical irritants. Exacerbating factors in AD include exposure to food allergens, dust, emotional stress, detergents, fragranced soaps, textiles, and ingredients in cosmetic products. Patients should be advised to use mild detergents and fragrance-free soaps and to avoid harsh materials such as wool. However, avoidance of specific ingredients in cosmetic products is not as straightforward because manufacturers are not required to disclose certain ingredients. In general, fragrances such as balsam of Peru and cinnamaldehyde, as well as preservatives such as parabens, isothiazolinones, and formaldehyde, should be avoided when selecting cosmetic products. Patients with AD should purchase fragrance-free products that are specifically formulated for sensitive skin. Additionally, patients should not apply makeup if their skin is irritated or oozing, as the flare may worsen.15
Moisturizers
Due to the impaired skin barrier function in patients with AD, regular application of fragrance-free moisturizers is essential to maintain hydration and to reduce xerosis. Various classes of moisturizers may be prescribed (eg, lotions, creams, gels, ointments) based on disease severity and patient preference. Light preparations such as lotions, creams, and gels have a high water content and generally are more appealing from a cosmetic standpoint because they do not create any residue on the skin. However, these options may require more frequent application because they are absorbed quickly. Heavy preparations such as ointments have longer-lasting effects due to their high oil content but tend to be less cosmetically appealing because of their greasiness.16
Although the amount and frequency of application of moisturizers has not been defined, liberal application several times daily is generally advised to minimize xerosis.17 Most physicians recommend applying moisturizer to the skin immediately after bathing to seal in moisture. Some patients prefer to use lotions and creams during the day because these products make the skin feel smooth and reserve the greasier ointments for nighttime application.
Topical Corticosteroids
Prescribed in conjunction with moisturizers, TCs are the mainstay of anti-inflammatory therapy in AD. Topical corticosteroids are classified into 7 groups based on potency, ranging from superpotent (class 1) to least potent (class 7). For acute AD flares, TCs should be applied daily for up to several weeks. Once the inflammation has resolved, it is recommended to apply TCs once to twice weekly to reduce the rate of relapse.18 Despite their effectiveness in the treatment of acute AD flares, TCs have a considerable side-effect profile. Potential adverse effects include skin atrophy, striae, telangiectasia, hypopigmentation, increased hair growth, steroid acne, growth retardation, and Cushing syndrome. Skin atrophy, which is the most common complication associated with TCs, results in shiny transparent skin, allowing for visualization of veins.19,20 Although many of these side effects will resolve after discontinuing the TCs, they are aesthetically displeasing during treatment, making it crucial for physicians to educate their patients on the proper usage of TCs to prevent negative outcomes.
Topical Calcineurin Inhibitors
Topical calcineurin inhibitors (TCIs) are a class of anti-inflammatories that are used to overcome the adverse effects of TCs. They are approved as alternatives to TCs in patients who have failed to respond to other topical treatments as well as those who have developed cutaneous atrophy from the use of TCs or have AD in sensitive areas such as the face, neck, and/or skin folds. Unlike TCs, TCIs do not cause atrophy, striae, or discoloration of the skin, which makes them more desirable from a cosmetic perspective. Their mechanism of action is distinct from TCs in that they inhibit calcineurin-dependent T-cell activation, thus preventing the transcription of inflammatory cytokines.21 Two TCIs are currently available: tacrolimus ointment 0.03% and 0.1% concentrations for moderate to severe AD and pimecrolimus cream 1% for mild to moderate AD.22 Twice-daily application of TCIs is recommended to decrease inflammation and pruritus associated with AD. Studies also have shown that intermittent use of TCIs 3 times weekly can aid in reducing relapses.23-25
The results from clinical trials demonstrate the rapid and continuous effects of both pimecrolimus and tacrolimus. In a controlled long-term study of adults, pimecrolimus provided significant relief of pruritus as soon as day 3 (P<.001).26,27 Pimecrolimus also provides long-term relief by preventing disease progression to flares, which was exemplified in a study (N=713) with no flares in 51% of pimecrolimus patients at 12 months versus 28% in the conventional treatment group (P<.001).28 Similarly, long-term studies of tacrolimus demonstrated an improvement of all symptoms of AD after 1 week of treatment. Maximal improvement was achieved with continued use of tacrolimus, and up to 1 year of tacrolimus use was found to be safe and effective.29,30 Thus, TCIs have been proven to be an effective choice in maintenance therapy for AD and have a good safety profile. The most common adverse effects of TCIs are local skin reactions, such as stinging and burning at the site of application. Rare cases of skin cancer and lymphoma have been reported; however, a causal relationship has yet to be established.31,32
Additional Therapies
Wet wrap therapy is effective for rapid control of flares and in controlling recalcitrant AD. Wet wraps function via several mechanisms; they provide a mechanical barrier against scratching, increase moisture and soften the skin, and enhance absorption of topical medications.33,34 The following method is employed when using wet wraps: an emollient or TC is applied to the area, a tubular bandage soaked in warm water is wrapped over the area, and dry bandages are used to form the outermost layer. Although wet wrap therapy is beneficial in treating AD, it is labor intensive and may require the expertise of a nurse. Thus, unlike other therapies, which patients can easily apply without interfering with their day, wet wraps must be applied at home or in a hospital setting.
Light therapy is another effective method of controlling AD. Although multiple forms of UV phototherapy are beneficial for symptom control in AD, there is no definitive recommendation regarding the specific type of light therapy due to a lack of comparative studies. Natural sunlight, narrowband UVB, broadband UVB, UVA, oral or topical psoralen plus UVA, as well as UVA and UVB can all be utilized in the treatment of AD. However, similar to natural sunlight, artificial light therapy can cause burning, blistering, hyperpigmentation, dark spots, and wrinkles. Because society places a large emphasis on maintaining a youthful appearance, patients may be hesitant to use a treatment that could potentially advance the skin’s aging process. Thus, it is important that this therapy is properly controlled to prevent further skin damage.35-37
When optimal topical regimens and phototherapy have failed to control AD, systemic immunomodulation therapies may be used. Currently, the most commonly used medications are cyclosporine 150 to 300 mg daily, methotrexate 7.5 to 25 mg weekly, mycophenolate mofetil 0.5 to 3 g daily, and azathioprine 1 to 3 mg/kg daily.38,39 Decisions regarding the specific class of drugs should be based on the patient’s AD status, comorbidities, and personal preference.
Conclusion
Atopic dermatitis is a common chronic condition that can occur at any age and cause substantial physical, psychological, social, and/or emotional stress for patients and their families. Although TCs have been the standard of treatment for many years, ongoing concerns regarding their safety have led to the use of TCIs, which overcome some of the drawbacks of steroid therapy. Phototherapy and systemic immunosuppressant therapy are reserved for patients who have not responded to optimal topical therapies. Although several therapeutic avenues exist for patients, there is a need for the development of more effective and safer drugs. Furthermore, cosmetic products created specifically for patients with AD would be beneficial, as patients often struggle to select products that do not cause more harm than good. Given the complexity of the pathogenesis of AD, further research must focus on defining the specific pathways involved in the disease and targeting these pathways with therapies.
1. Shaw TE, Currie GP, Koudelka CW, et al. Eczema prevalence in the United States: data from the 2003 National Survey of Children’s Health. J Invest Dermatol. 2011;131:67-73.
2. Deckers IA, McLean S, Linssen S, et al. Investigating international time trends in the incidence and prevalence of atopic eczema 1990-2010: a systematic review of epidemiological studies. PLoS One. 2012;7:e39803.
3. Boguniewicz M, Leung DY. Atopic dermatitis: a disease of altered skin barrier and immune dysregulation. Immunol Rev. 2011;242:233-246.
4. Peate I. Eczema: causes, symptoms and treatment in the community. Br J Community Nurs. 2011;16:324, 326-331.
5. Williams HC, Burney PG, Pembroke AC, et al. The U.K. Working Party’s diagnostic criteria for atopic dermatitis. III. independent hospital validation. Br J Dermatol. 1994;131:406-416.
6. Magin P, Adams J, Heading G, et al. Experiences of appearance-related teasing and bullying in skin diseases and their psychological sequelae: results of a qualitative study. Scand J Caring Sci. 2008;22:430-436.
7. Beattie P, Lewis-Jones M. A comparative study of impairment of quality of life in children with skin disease and children with other chronic childhood diseases. Br J Dermatol. 2006;155:145-151.
8. Spergel JM. From atopic dermatitis to asthma: the atopic march [published online January 22, 2010]. Ann Allergy Asthma Immunol. 2010;105:99-106; quiz 107-109, 117.
9. Leung DY. New insights into atopic dermatitis: role of skin barrier and immune dysregulation. Allergol Int. 2013;62:151-161.
10. Balma-Mena A, Lara-Corrales I, Zeller J, et al. Colonization with community-acquired methicillin-resistant Staphylococcus aureus in children with atopic dermatitis: a cross-sectional study. Int J Dermatol. 2011;50:682-688.
11. Strawser MS, Storch EA, Roberti JW. The Teasing Questionnaire-Revised: measurement of childhood teasing in adults. J Anxiety Disord. 2005;19:780-792.
12. Magin P, Adams J, Heading G, et al. Experiences of appearance-related teasing and bullying in skin diseases and their psychological sequelae: results of a qualitative study. Scand J Caring Sci. 2008;22:430-436.
13. Silverberg J. Health care utilization, patient costs, and access to care in US adults with eczema: a population-based study. JAMA Dermatol. 2015;151:743-752.
14. Ellis C, Luger T, Abeck D, et al. International Consensus Conference on Atopic Dermatitis II (ICCAD II): clinical update and current treatment strategies. Br J Dermatol. 2003;148(suppl 63):3-10.
15. Kim K. Influences of environmental chemicals on atopic dermatitis. Toxicol Res. 2015;31:89-96.
16. Ridd M, Redmond N, Hollinghurst S, et al. Choice of Moisturiser for Eczema Treatment (COMET): study protocol for a randomized controlled trial. Trials. 2015;16:304.
17. Hon KL, Ching GK, Leung TF, et al. Estimating emollient usage in patients with eczema. Clin Exp Dermatol. 2010;35:22-26.
18. Hanifin J, Gupta AK, Rajagopalan R. Intermittent dosing of fluticasone propionate cream for reducing the risk of relapse in atopic dermatitis patients. Br J Dermatol. 2002;147:528-537.
19. Hill CJ, Rostenberg A Jr. Adverse effects from topical steroids. Cutis. 1978;21:624-628.
20. Ruiz-Maldonado R, Zapata G, Lourdes T, et al. Cushing’s syndrome after topical application of corticosteroids. Am J Dis Child. 1982;136:274-275.
21. Grassberger M, Baumruker T, Enz A, et al. A novel anti-inflammatory drug, SDZ ASM 981, for the treatment of skin diseases: in vitro pharmacology. Br J Dermatol. 1999;141:264-273.
22. Eichenfield L, Wynnis T, Berger T. Guidelines of care for the management of atopic dermatitis: management and treatment of atopic dermatitis with topical therapies. J Am Acad Dermatol. 2014;71:116-132.
23. Reitamo S, Harper J, Bos JD, et al. 0.03% Tacrolimus ointment applied once or twice daily is more efficacious than 1% hydrocortisone acetate in children with moderate to severe atopic dermatitis: results of a randomized double-blind controlled trial. Br J Dermatol. 2004;150:554-562.
24. Ruer-Mulard M, Aberer W, Gunstone A, et al. Twice-daily versus once-daily applications of pimecrolimus cream 1% for the prevention of disease relapse in pediatric patients with atopic dermatitis. Pediatr Dermatol. 2009;26:551-558.
25. Breneman D, Fleischer AB Jr, Abramovits W, et al. Intermittent therapy for flare prevention and long-term disease control in stabilized atopic dermatitis: a randomized comparison of 3-times-weekly applications of tacrolimus ointment versus vehicle. J Am Acad Dermatol. 2008;58:990-999.
26. Meurer M, Fölster-Holst R, Wozel G, et al. Pimecrolimus cream 1% (Elidel) provides significant and rapid relief of pruritus and improves disease control and quality of life in atopic dermatitis in adults. J Invest Dermatol. 2002;119:350.
27. Meurer M, Fölster-Holst R, Wozel G, et al. Pimecrolimus cream in the long-term management of atopic dermatitis in adults: a six-month study. Dermatology. 2002;205:271-277.
28. Wahn U, Bos JD, Goodfield M, et al. Efficacy and safety of pimecrolimus cream in the long-term management of atopic dermatitis in children. Pediatrics. 2002;110(1, pt 1):e2.
29. Kang S, Lucky AW, Pariser D, et al. Long-term safety and efficacy of tacrolimus ointment for the treatment of atopic dermatitis in children. J Am Acad Dermatol. 2001;44(suppl 1):S58-S64.
30. Reitamo S, Wollenberg A, Schöpf E, et al. Safety and efficacy of 1 year of tacrolimus ointment monotherapy in adults with atopic dermatitis. the European Tacrolimus Ointment Study Group. Arch Dermatol. 2000;136:999-1006.
31. Frankel HC, Qureshi AA. Comparative effectiveness of topical calcineurin inhibitors in adult patients with atopic dermatitis. Am J Clin Dermatol. 2012;13:113-123.
32. Tennis P, Gelfand JM, Rothman KJ. Evaluation of cancer risk related to atopic dermatitis and use of topical calcineurin inhibitors. Br J Dermatol. 2011;165:465-473.
33. Dabade TS, Davis DM, Wetter DA, et al. Wet dressing therapy in conjunction with topical corticosteroids is effective for rapid control of severe pediatric atopic dermatitis: experience with 218 patients over 30 years at Mayo Clinic. J Am Acad Dermatol. 2012;67:100-106.
34. Devillers AC, Oranje AP. Efficacy and safety of ‘wet-wrap’ dressings as an intervention treatment in children with severe and/or refractory atopic dermatitis: a critical review of the literature. Br J Dermatol. 2006;154:579-585.
35. Meduri NB, Vandergriff T, Rasmussen H, et al. Phototherapy in the management of atopic dermatitis: a systematic review. Photodermatol Photoimmunol Photomed. 2007;23:106-112.
36. Clayton TH, Clark SM, Turner D, et al. The treatment of severe atopic dermatitis in childhood with narrowband ultraviolet B phototherapy. Clin Exp Dermatol. 2007;32:28-33.
37. Jekler J, Larko O. UVB phototherapy of atopic dermatitis. Br J Dermatol. 1988;119:697-705.
38. Roekevisch E, Spuls PI, Kuester D, et al. Efficacy and safety of systemic treatments for moderate-to-severe atopic dermatitis: a systematic review. J Allergy Clin Immunol. 2014;133:429-438.39. Hoare C, Li Wan Po A, Williams H. Systematic review of treatments for atopic eczema. Health Technol Assess. 2000;4:1-191.
39. Hoare C, Li Wan Po A, Williams H. Systematic review of treatments for atopic eczema. Health Technol Assess. 2000;4:1-191.
1. Shaw TE, Currie GP, Koudelka CW, et al. Eczema prevalence in the United States: data from the 2003 National Survey of Children’s Health. J Invest Dermatol. 2011;131:67-73.
2. Deckers IA, McLean S, Linssen S, et al. Investigating international time trends in the incidence and prevalence of atopic eczema 1990-2010: a systematic review of epidemiological studies. PLoS One. 2012;7:e39803.
3. Boguniewicz M, Leung DY. Atopic dermatitis: a disease of altered skin barrier and immune dysregulation. Immunol Rev. 2011;242:233-246.
4. Peate I. Eczema: causes, symptoms and treatment in the community. Br J Community Nurs. 2011;16:324, 326-331.
5. Williams HC, Burney PG, Pembroke AC, et al. The U.K. Working Party’s diagnostic criteria for atopic dermatitis. III. independent hospital validation. Br J Dermatol. 1994;131:406-416.
6. Magin P, Adams J, Heading G, et al. Experiences of appearance-related teasing and bullying in skin diseases and their psychological sequelae: results of a qualitative study. Scand J Caring Sci. 2008;22:430-436.
7. Beattie P, Lewis-Jones M. A comparative study of impairment of quality of life in children with skin disease and children with other chronic childhood diseases. Br J Dermatol. 2006;155:145-151.
8. Spergel JM. From atopic dermatitis to asthma: the atopic march [published online January 22, 2010]. Ann Allergy Asthma Immunol. 2010;105:99-106; quiz 107-109, 117.
9. Leung DY. New insights into atopic dermatitis: role of skin barrier and immune dysregulation. Allergol Int. 2013;62:151-161.
10. Balma-Mena A, Lara-Corrales I, Zeller J, et al. Colonization with community-acquired methicillin-resistant Staphylococcus aureus in children with atopic dermatitis: a cross-sectional study. Int J Dermatol. 2011;50:682-688.
11. Strawser MS, Storch EA, Roberti JW. The Teasing Questionnaire-Revised: measurement of childhood teasing in adults. J Anxiety Disord. 2005;19:780-792.
12. Magin P, Adams J, Heading G, et al. Experiences of appearance-related teasing and bullying in skin diseases and their psychological sequelae: results of a qualitative study. Scand J Caring Sci. 2008;22:430-436.
13. Silverberg J. Health care utilization, patient costs, and access to care in US adults with eczema: a population-based study. JAMA Dermatol. 2015;151:743-752.
14. Ellis C, Luger T, Abeck D, et al. International Consensus Conference on Atopic Dermatitis II (ICCAD II): clinical update and current treatment strategies. Br J Dermatol. 2003;148(suppl 63):3-10.
15. Kim K. Influences of environmental chemicals on atopic dermatitis. Toxicol Res. 2015;31:89-96.
16. Ridd M, Redmond N, Hollinghurst S, et al. Choice of Moisturiser for Eczema Treatment (COMET): study protocol for a randomized controlled trial. Trials. 2015;16:304.
17. Hon KL, Ching GK, Leung TF, et al. Estimating emollient usage in patients with eczema. Clin Exp Dermatol. 2010;35:22-26.
18. Hanifin J, Gupta AK, Rajagopalan R. Intermittent dosing of fluticasone propionate cream for reducing the risk of relapse in atopic dermatitis patients. Br J Dermatol. 2002;147:528-537.
19. Hill CJ, Rostenberg A Jr. Adverse effects from topical steroids. Cutis. 1978;21:624-628.
20. Ruiz-Maldonado R, Zapata G, Lourdes T, et al. Cushing’s syndrome after topical application of corticosteroids. Am J Dis Child. 1982;136:274-275.
21. Grassberger M, Baumruker T, Enz A, et al. A novel anti-inflammatory drug, SDZ ASM 981, for the treatment of skin diseases: in vitro pharmacology. Br J Dermatol. 1999;141:264-273.
22. Eichenfield L, Wynnis T, Berger T. Guidelines of care for the management of atopic dermatitis: management and treatment of atopic dermatitis with topical therapies. J Am Acad Dermatol. 2014;71:116-132.
23. Reitamo S, Harper J, Bos JD, et al. 0.03% Tacrolimus ointment applied once or twice daily is more efficacious than 1% hydrocortisone acetate in children with moderate to severe atopic dermatitis: results of a randomized double-blind controlled trial. Br J Dermatol. 2004;150:554-562.
24. Ruer-Mulard M, Aberer W, Gunstone A, et al. Twice-daily versus once-daily applications of pimecrolimus cream 1% for the prevention of disease relapse in pediatric patients with atopic dermatitis. Pediatr Dermatol. 2009;26:551-558.
25. Breneman D, Fleischer AB Jr, Abramovits W, et al. Intermittent therapy for flare prevention and long-term disease control in stabilized atopic dermatitis: a randomized comparison of 3-times-weekly applications of tacrolimus ointment versus vehicle. J Am Acad Dermatol. 2008;58:990-999.
26. Meurer M, Fölster-Holst R, Wozel G, et al. Pimecrolimus cream 1% (Elidel) provides significant and rapid relief of pruritus and improves disease control and quality of life in atopic dermatitis in adults. J Invest Dermatol. 2002;119:350.
27. Meurer M, Fölster-Holst R, Wozel G, et al. Pimecrolimus cream in the long-term management of atopic dermatitis in adults: a six-month study. Dermatology. 2002;205:271-277.
28. Wahn U, Bos JD, Goodfield M, et al. Efficacy and safety of pimecrolimus cream in the long-term management of atopic dermatitis in children. Pediatrics. 2002;110(1, pt 1):e2.
29. Kang S, Lucky AW, Pariser D, et al. Long-term safety and efficacy of tacrolimus ointment for the treatment of atopic dermatitis in children. J Am Acad Dermatol. 2001;44(suppl 1):S58-S64.
30. Reitamo S, Wollenberg A, Schöpf E, et al. Safety and efficacy of 1 year of tacrolimus ointment monotherapy in adults with atopic dermatitis. the European Tacrolimus Ointment Study Group. Arch Dermatol. 2000;136:999-1006.
31. Frankel HC, Qureshi AA. Comparative effectiveness of topical calcineurin inhibitors in adult patients with atopic dermatitis. Am J Clin Dermatol. 2012;13:113-123.
32. Tennis P, Gelfand JM, Rothman KJ. Evaluation of cancer risk related to atopic dermatitis and use of topical calcineurin inhibitors. Br J Dermatol. 2011;165:465-473.
33. Dabade TS, Davis DM, Wetter DA, et al. Wet dressing therapy in conjunction with topical corticosteroids is effective for rapid control of severe pediatric atopic dermatitis: experience with 218 patients over 30 years at Mayo Clinic. J Am Acad Dermatol. 2012;67:100-106.
34. Devillers AC, Oranje AP. Efficacy and safety of ‘wet-wrap’ dressings as an intervention treatment in children with severe and/or refractory atopic dermatitis: a critical review of the literature. Br J Dermatol. 2006;154:579-585.
35. Meduri NB, Vandergriff T, Rasmussen H, et al. Phototherapy in the management of atopic dermatitis: a systematic review. Photodermatol Photoimmunol Photomed. 2007;23:106-112.
36. Clayton TH, Clark SM, Turner D, et al. The treatment of severe atopic dermatitis in childhood with narrowband ultraviolet B phototherapy. Clin Exp Dermatol. 2007;32:28-33.
37. Jekler J, Larko O. UVB phototherapy of atopic dermatitis. Br J Dermatol. 1988;119:697-705.
38. Roekevisch E, Spuls PI, Kuester D, et al. Efficacy and safety of systemic treatments for moderate-to-severe atopic dermatitis: a systematic review. J Allergy Clin Immunol. 2014;133:429-438.39. Hoare C, Li Wan Po A, Williams H. Systematic review of treatments for atopic eczema. Health Technol Assess. 2000;4:1-191.
39. Hoare C, Li Wan Po A, Williams H. Systematic review of treatments for atopic eczema. Health Technol Assess. 2000;4:1-191.
Practice Points
- Cosmetic symptoms of atopic dermatitis can have a serious impact on the patient’s quality of life.
- Avoidance of flares and prevention of triggers is an important aspect of care.
- Treatment options range from optimized skin care to topical prescription therapies to systemic medications.