User login
Factors Affecting Academic Leadership in Dermatology
Leadership is widely recognized as a key component in the role of a physician,1 which is especially true in dermatology, a specialty that faces severe challenges in the recruitment and retention of academic faculty.2 A study of the dermatology workforce found that academic institutions are more likely to be seeking to hire new faculty2 and that many dermatology residency programs often are looking to replace chairpersons (chairs) and/or chiefs.3 Although fewer dermatology residents are pursuing academic careers than careers in private practice, full-time faculty members also are leaving their academic posts. This shift is demonstrated by the younger mean age of academic dermatologists2 and the increased rate of departure from academia prior to pursuing more formalized leadership roles.4
It has been suggested that the number of full-time faculty and number of faculty publications positively influence graduates of dermatology residency programs to pursue academic careers; however, variables affecting the likelihood of graduates of dermatology residency programs becoming academic leaders later in their career have not been well studied.3 The purpose of this study is to determine the factors that influence the development of program chairs/chiefs and program directors (PDs) of dermatology residency programs.
Methods
Data were collected from all accredited dermatology residency programs in the United States as of December 31, 2008. Residency programs that were started after 2004 were excluded from the study, as it was thought that these programs may not have graduated a sufficient number of residents for assessment. Military residency programs also were excluded, as graduates from these programs often do not freely choose their careers after residency.
Primary end points were the number of chairs/chiefs and PDs who had graduated from each dermatology residency program. Variables included the number of years the program had been in existence, status of the program as a department or division, number of full-time faculty members, number of residents, National Institutes of Health funding received in 2008 (http://www.report.nih.gov/award/index.cfm), Dermatology Foundation (DF) funding received (http://www.dermatologyfoundation.org/rap/), number of publications from full-time faculty members in 2008 (http://www.ncbi.nlm.nih.gov/pubmed/), number of faculty lectures given at annual meetings of 5 societies in 2008 (American Academy of Dermatology, the Society for Investigative Dermatology, the American Society of Dermatopathology, the Society for Pediatric Dermatology, and American Society for Dermatologic Surgery), and the number of faculty members on the editorial boards of 6 major dermatology journals (Journal of the American Academy of Dermatology, Journal of Investigative Dermatology, Archives of Dermatology [currently known as JAMA Dermatology], Dermatologic Surgery, Pediatric Dermatology, and Journal of Cutaneous Pathology). Data regarding faculty and residents were obtained from program Web sites and inquiries from individual programs. The year 1974 was used as a cutoff for the number of years a program had been in existence. Years of existence of a program was controlled for in the analysis. The ratio of faculty to residents was calculated per year and categorized as 4 or more or less than 4 to minimize the effect of changing program size over the years. For faculty members who split time between 2 residency programs, each program was given credit for the duration of time spent at that program. Faculty members who hold a PhD only and those who completed their residencies in non-US dermatology residency programs were excluded from the outcome variables. To avoid duplicate faculty publications, collections for each residency program were created within PubMed (ie, if 2 authors from the same program coauthored an article, it was only counted once toward the total number of faculty publications from that program).
Because the data were skewed (ie, there were a large number of programs with 0 graduating chairs/chiefs and PDs), nonparametric analyses were utilized. Logistic regression was used to calculate the odds of producing chairs/chiefs or PDs (yes vs no). Multiple logistic regression helped to determine those variables that were most closely associated with odds of graduating a chair/chief or PD. Variables with a significance level of P<.10 were considered in the multiple logistic regression, and backward selection was used to determine a model. Multiple linear regression was used to determine correlation coefficients for each of the variables and the number of chairs/chiefs or PDs graduated, controlling for the estimated number of graduates from the program and number of years the program had been in existence. Analyses for graduating chairs/chiefs and PDs were conducted separately. The final significance level used was P<.05. Data were analyzed using SAS version 9.3. This study was approved by the institutional review board at Kaiser Permanente Southern California.
Results
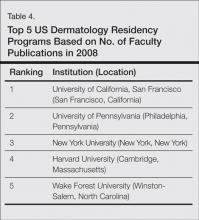
Data from 103 dermatology residency programs were included in the analysis. Of these programs, 47 had graduated at least 1 chair/chief and 55 had graduated at least 1 PD. Among the programs graduating any chairs/chiefs, they produced an average of 2.04 chairs/chiefs and 1.86 PDs. The 5 dermatology residency programs that graduated the highest total number of chairs/chiefs and PDs were Harvard University (Cambridge, Massachusetts), the University of Michigan (Ann Arbor, Michigan), New York University (New York, New York), Yale-New Haven Hospital (New Haven, Connecticut), and the University of Minnesota (Minneapolis, Minnesota).
Factors that had the highest effect on the odds of a program graduating a chair/chief included the ratio of faculty to residents per year, presence of DF funding in 2008, number of years program was in existence, number of residents, number of full-time faculty, and number of full-time faculty on editorial boards of 6 major dermatology journals (Table 1). When controlling for each of these variables in the final multivariable analysis, programs with 4 or more faculty per resident had 3.31 times the odds of producing a chair/chief (95% confidence interval [CI], 1.14-9.66; P=.028).
Factors that had the highest effect on the odds of a program graduating a PD included status as department versus division, ratio of faculty to residents per year, presence of DF funding in 2008, number of lectures given by full-time faculty members at annual society meetings, number of residents, number of years program was in existence, number of full-time faculty, and number of publications from full-time faculty members (Table 2). The most significant factor associated with graduating PDs after controlling for other variables was the number of publications from full-time faculty members. The odds increased by 3.2% for every 1 additional publication and 32% for every 10 additional publications (95% CI, 1.01-1.06; P=.026).
Multiple linear regression demonstrated a positive relationship between the number of graduating chairs/chiefs and total full-time faculty members (R2=0.26; P=.034) and ratio of full-time faculty to residents (R2=0.29; P<.001). Marginally significant correlations were seen between the number of PDs and ratio of full-time faculty to residents (R2=0.32; P=.05) as well as the number of publications from full-time faculty members (R2=0.32; P=.05).
Comment
The ratio of full-time faculty to residents increased a program’s odds of graduating a chair/chief. More faculty members may lead to more opportunities for mentorship of residents and young faculty. Mentors are widely perceived to be integral to the learning and development of residents, not only in dermatology5 but across all specialties.6 Mentors also have been noted to play a key role in bolstering and maintaining interest in academics,7 which is true not only with regard to recruiting new residents but for retaining young faculty members. In a study (N=109) that examined factors associated with residents’ loss of interest in academic careers, half of the participants reported a lack of effective mentors, role models, and professional guidance.8 Mentors provide teaching, supervision, and advice, especially with regard to research and career paths.9 A large number of faculty members provides more opportunities for direct mentorship and offers residents more exposure to research, specialty clinics, and academic philosophies, which may positively influence and even inspire academic pursuits and leadership.3
Although the solution to producing future chairs/chiefs and PDs may lie in faculty guidance, finding and retaining faculty members as mentors amidst a shortage of academic dermatologists presents an underlying issue.3 In addition to a lack of mentorship, residents cite bureaucracy, salary differentials, and location to explain a loss of interest in academic careers.8 Several programs have been developed to address the recruitment of dermatology residents for academic careers, including combined medical-dermatology programs, 2+2 programs (2 years of clinical residency plus 2 additional research years), clinical research fellowships,10 and the Society for Investigative Dermatology’s Dermatology Resident Retreat for Future Academicians (http://www.sidnet.org/fortraineesandresidents).11 Perhaps recruitment should even start at the medical student level. In light of the academic strength of the current pool of dermatology residency applicants,12 training programs should continue to screen for applicants with sincere interests in academia.13 Students with more research and publications may be more likely to pursue academic careers, in accordance with prior studies of dermatology trainees.3,14 Studies also have shown that graduates of foreign dermatology residencies15 and individuals who hold both MD and PhD degrees may be more likely to enter into academic careers.16
For creating future chairs/chiefs and PDs, retention of young faculty in academics is as important as recruiting residents.17 At the mid-career level, the decline of funds for research has generated pressure for academic physicians to see increasing numbers of patients, leaving insufficient time for the many duties that accompany academic posts,2 including teaching and publishing. Other reasons that faculty members leave their posts before 40 years of age include financial and family concerns18 as well as the desire for more autonomy.4 Formalized training is seen with the American Academy of Dermatology’s Academic Dermatology Leadership Program (https://www.aad.org/members/leadership-institute/mentoring/aad-mentoring-opportunities/academic-dermatology-leadership-program-mentee), which promotes advanced leadership training to recent graduates.5 Other methods include support of young faculty with mentorship; grant applications; and administration at the department, hospital, and government levels.17 Recruitment of faculty from private practice may represent another potential source of faculty who wish to pursue more scholarly endeavors.4 Teaching has been cited as a primary reason for faculty members to remain in academia,18 and thus time for teaching must be protected. Such a strategy is in accordance with our findings that amount of annual DF funding received, number of full-time faculty publications, number of faculty members on editorial boards of major dermatology journals, and number of lectures given by full-time faculty factors at annual society meetings are positively associated with the odds of producing chairs/chiefs or PDs. In particular, the number of full-time faculty publications is directly related to increased odds of graduates becoming PDs. Residents and young faculty members who take part in research and attend national conferences may find inspiration or develop a passion for academic leadership.
A limitation to this study is that the ratio of faculty to graduated residents for some programs likely has fluctuated over the last 35 years. This study assumed that certain programs remained generally small or large during the course of their existence, which was controlled by using the ratio between faculty and residents. Additionally, the number of years that a program has been in existence influences the likelihood of that program to graduate higher numbers of residents who become chairs/chiefs or PDs. As a result, we used multiple linear regression to control for the number of residents and number of years that a program had been in existence. Finally, while the relationship between academic leaders and research may be explained by the increased likelihood of faculty with more funding, publications, or lectures to be selected for leadership roles, this finding supports the notion that research can contribute to leadership. This analysis identifies modifiable factors among residency training programs to improve the odds of graduating future academic leaders.
Conclusion
As the present study shows, the ratio of faculty to residents and the number of full-time faculty publications are key to graduating academic leaders in dermatology. Retention of faculty as leaders in academic dermatology is as crucial to the field as recruitment of residents into academic dermatology. Mentorship should be highly encouraged through the creation of formal programs and should not end at the resident level. Emphasizing the intellectually stimulating aspects of academia and providing administrative resources may help decrease the burden of academic duties, allowing the pursuit of teaching and research and ultimately the resources to become candidates for leadership positions in academia.
1. Wood LD, Miller JJ, Marks JG Jr. The need for leadership: how can we better train the next generation of dermatologists? J Am Acad Dermatol. 2008;58:715-717.
2. Resneck JS Jr, Tierney EP, Kimball AB. Challenges facing academic dermatology: survey data on the faculty workforce. J Am Acad Dermatol. 2006;54:211-216.
3. Wu JJ, Ramirez CC, Alonso CA, et al. Dermatology residency program characteristics that correlate with graduates selecting an academic dermatology career. Arch Dermatol. 2006;142:845-850.
4. Loo DS, Liu CL, Geller AC, et al. Academic dermatology manpower: issues of recruitment and retention. Arch Dermatol. 2007;143:341-347.
5. Donovan JC. A survey of dermatology residency program directors’ views on mentorship. Dermatol Online J. 2009;15:1.
6. Sambunjak D, Straus SE, Marusi´c A. Mentoring in academic medicine: a systematic review. JAMA. 2006;296:1103-1115.
7. Rubenstein DS, Blauvelt A, Chen SC, et al. The future of academic dermatology in the United States: report on the resident retreat for future physician-scientists, June 15-17, 2001. J Am Acad Dermatol. 2002;47:300-303.
8. Reck SJ, Stratman EJ, Vogel C, et al. Assessment of residents’ loss of interest in academic careers and identification of correctable factors. Arch Dermatol. 2006;142:855-858.
9. Clark RA. Teacher, supervisor, adviser, or mentor? J Invest Dermatol. 2011;131:1779-1780.
10. Wu JJ. Current strategies to address the ongoing shortage of academic dermatologists. J Am Acad Dermatol. 2007;56:1065-1066.
11. Culton DA, Rubenstein DS, Diaz LA. The resident retreat for future academicians. J Invest Dermatol. 2010;130:1775-1777.
12. Wu JJ, Tyring SK. The academic strength of current dermatology residency applicants. Dermatol Online J. 2003;9:22.
13. Kia KF, Gielczyk RA, Ellis CN. Academia is the life for me, I’m sure. Arch Dermatol. 2006;142:911-913.
14. Miller CJ, Wood GC, Miller JJ, et al. Academics or private practice? the future of dermatologic surgery education. Dermatol Surg. 2006;32:70-75, discussion 76-78.
15. Wu JJ, Davis KF, Ramirez CC, et al. Graduates-of-foreign-dermatology residencies and military dermatology residencies and women in academic dermatology. Dermatol Online J. 2009;15:2.
16. Wu JJ, Davis KF, Ramirez CC, et al. MD/PhDs are more likely than MDs to choose a career in academic dermatology. Dermatol Online J. 2008;14:27.
17. Olerud JE. Academic workforce in dermatology. Arch Dermatol. 2007;143:409-410.
18. Turner E, Yoo J, Salter S, et al. Leadership workforce in academic dermatology. Arch Dermatol. 2007;143:948-949.
Leadership is widely recognized as a key component in the role of a physician,1 which is especially true in dermatology, a specialty that faces severe challenges in the recruitment and retention of academic faculty.2 A study of the dermatology workforce found that academic institutions are more likely to be seeking to hire new faculty2 and that many dermatology residency programs often are looking to replace chairpersons (chairs) and/or chiefs.3 Although fewer dermatology residents are pursuing academic careers than careers in private practice, full-time faculty members also are leaving their academic posts. This shift is demonstrated by the younger mean age of academic dermatologists2 and the increased rate of departure from academia prior to pursuing more formalized leadership roles.4
It has been suggested that the number of full-time faculty and number of faculty publications positively influence graduates of dermatology residency programs to pursue academic careers; however, variables affecting the likelihood of graduates of dermatology residency programs becoming academic leaders later in their career have not been well studied.3 The purpose of this study is to determine the factors that influence the development of program chairs/chiefs and program directors (PDs) of dermatology residency programs.
Methods
Data were collected from all accredited dermatology residency programs in the United States as of December 31, 2008. Residency programs that were started after 2004 were excluded from the study, as it was thought that these programs may not have graduated a sufficient number of residents for assessment. Military residency programs also were excluded, as graduates from these programs often do not freely choose their careers after residency.
Primary end points were the number of chairs/chiefs and PDs who had graduated from each dermatology residency program. Variables included the number of years the program had been in existence, status of the program as a department or division, number of full-time faculty members, number of residents, National Institutes of Health funding received in 2008 (http://www.report.nih.gov/award/index.cfm), Dermatology Foundation (DF) funding received (http://www.dermatologyfoundation.org/rap/), number of publications from full-time faculty members in 2008 (http://www.ncbi.nlm.nih.gov/pubmed/), number of faculty lectures given at annual meetings of 5 societies in 2008 (American Academy of Dermatology, the Society for Investigative Dermatology, the American Society of Dermatopathology, the Society for Pediatric Dermatology, and American Society for Dermatologic Surgery), and the number of faculty members on the editorial boards of 6 major dermatology journals (Journal of the American Academy of Dermatology, Journal of Investigative Dermatology, Archives of Dermatology [currently known as JAMA Dermatology], Dermatologic Surgery, Pediatric Dermatology, and Journal of Cutaneous Pathology). Data regarding faculty and residents were obtained from program Web sites and inquiries from individual programs. The year 1974 was used as a cutoff for the number of years a program had been in existence. Years of existence of a program was controlled for in the analysis. The ratio of faculty to residents was calculated per year and categorized as 4 or more or less than 4 to minimize the effect of changing program size over the years. For faculty members who split time between 2 residency programs, each program was given credit for the duration of time spent at that program. Faculty members who hold a PhD only and those who completed their residencies in non-US dermatology residency programs were excluded from the outcome variables. To avoid duplicate faculty publications, collections for each residency program were created within PubMed (ie, if 2 authors from the same program coauthored an article, it was only counted once toward the total number of faculty publications from that program).
Because the data were skewed (ie, there were a large number of programs with 0 graduating chairs/chiefs and PDs), nonparametric analyses were utilized. Logistic regression was used to calculate the odds of producing chairs/chiefs or PDs (yes vs no). Multiple logistic regression helped to determine those variables that were most closely associated with odds of graduating a chair/chief or PD. Variables with a significance level of P<.10 were considered in the multiple logistic regression, and backward selection was used to determine a model. Multiple linear regression was used to determine correlation coefficients for each of the variables and the number of chairs/chiefs or PDs graduated, controlling for the estimated number of graduates from the program and number of years the program had been in existence. Analyses for graduating chairs/chiefs and PDs were conducted separately. The final significance level used was P<.05. Data were analyzed using SAS version 9.3. This study was approved by the institutional review board at Kaiser Permanente Southern California.
Results
Data from 103 dermatology residency programs were included in the analysis. Of these programs, 47 had graduated at least 1 chair/chief and 55 had graduated at least 1 PD. Among the programs graduating any chairs/chiefs, they produced an average of 2.04 chairs/chiefs and 1.86 PDs. The 5 dermatology residency programs that graduated the highest total number of chairs/chiefs and PDs were Harvard University (Cambridge, Massachusetts), the University of Michigan (Ann Arbor, Michigan), New York University (New York, New York), Yale-New Haven Hospital (New Haven, Connecticut), and the University of Minnesota (Minneapolis, Minnesota).
Factors that had the highest effect on the odds of a program graduating a chair/chief included the ratio of faculty to residents per year, presence of DF funding in 2008, number of years program was in existence, number of residents, number of full-time faculty, and number of full-time faculty on editorial boards of 6 major dermatology journals (Table 1). When controlling for each of these variables in the final multivariable analysis, programs with 4 or more faculty per resident had 3.31 times the odds of producing a chair/chief (95% confidence interval [CI], 1.14-9.66; P=.028).
Factors that had the highest effect on the odds of a program graduating a PD included status as department versus division, ratio of faculty to residents per year, presence of DF funding in 2008, number of lectures given by full-time faculty members at annual society meetings, number of residents, number of years program was in existence, number of full-time faculty, and number of publications from full-time faculty members (Table 2). The most significant factor associated with graduating PDs after controlling for other variables was the number of publications from full-time faculty members. The odds increased by 3.2% for every 1 additional publication and 32% for every 10 additional publications (95% CI, 1.01-1.06; P=.026).
Multiple linear regression demonstrated a positive relationship between the number of graduating chairs/chiefs and total full-time faculty members (R2=0.26; P=.034) and ratio of full-time faculty to residents (R2=0.29; P<.001). Marginally significant correlations were seen between the number of PDs and ratio of full-time faculty to residents (R2=0.32; P=.05) as well as the number of publications from full-time faculty members (R2=0.32; P=.05).
Comment
The ratio of full-time faculty to residents increased a program’s odds of graduating a chair/chief. More faculty members may lead to more opportunities for mentorship of residents and young faculty. Mentors are widely perceived to be integral to the learning and development of residents, not only in dermatology5 but across all specialties.6 Mentors also have been noted to play a key role in bolstering and maintaining interest in academics,7 which is true not only with regard to recruiting new residents but for retaining young faculty members. In a study (N=109) that examined factors associated with residents’ loss of interest in academic careers, half of the participants reported a lack of effective mentors, role models, and professional guidance.8 Mentors provide teaching, supervision, and advice, especially with regard to research and career paths.9 A large number of faculty members provides more opportunities for direct mentorship and offers residents more exposure to research, specialty clinics, and academic philosophies, which may positively influence and even inspire academic pursuits and leadership.3
Although the solution to producing future chairs/chiefs and PDs may lie in faculty guidance, finding and retaining faculty members as mentors amidst a shortage of academic dermatologists presents an underlying issue.3 In addition to a lack of mentorship, residents cite bureaucracy, salary differentials, and location to explain a loss of interest in academic careers.8 Several programs have been developed to address the recruitment of dermatology residents for academic careers, including combined medical-dermatology programs, 2+2 programs (2 years of clinical residency plus 2 additional research years), clinical research fellowships,10 and the Society for Investigative Dermatology’s Dermatology Resident Retreat for Future Academicians (http://www.sidnet.org/fortraineesandresidents).11 Perhaps recruitment should even start at the medical student level. In light of the academic strength of the current pool of dermatology residency applicants,12 training programs should continue to screen for applicants with sincere interests in academia.13 Students with more research and publications may be more likely to pursue academic careers, in accordance with prior studies of dermatology trainees.3,14 Studies also have shown that graduates of foreign dermatology residencies15 and individuals who hold both MD and PhD degrees may be more likely to enter into academic careers.16
For creating future chairs/chiefs and PDs, retention of young faculty in academics is as important as recruiting residents.17 At the mid-career level, the decline of funds for research has generated pressure for academic physicians to see increasing numbers of patients, leaving insufficient time for the many duties that accompany academic posts,2 including teaching and publishing. Other reasons that faculty members leave their posts before 40 years of age include financial and family concerns18 as well as the desire for more autonomy.4 Formalized training is seen with the American Academy of Dermatology’s Academic Dermatology Leadership Program (https://www.aad.org/members/leadership-institute/mentoring/aad-mentoring-opportunities/academic-dermatology-leadership-program-mentee), which promotes advanced leadership training to recent graduates.5 Other methods include support of young faculty with mentorship; grant applications; and administration at the department, hospital, and government levels.17 Recruitment of faculty from private practice may represent another potential source of faculty who wish to pursue more scholarly endeavors.4 Teaching has been cited as a primary reason for faculty members to remain in academia,18 and thus time for teaching must be protected. Such a strategy is in accordance with our findings that amount of annual DF funding received, number of full-time faculty publications, number of faculty members on editorial boards of major dermatology journals, and number of lectures given by full-time faculty factors at annual society meetings are positively associated with the odds of producing chairs/chiefs or PDs. In particular, the number of full-time faculty publications is directly related to increased odds of graduates becoming PDs. Residents and young faculty members who take part in research and attend national conferences may find inspiration or develop a passion for academic leadership.
A limitation to this study is that the ratio of faculty to graduated residents for some programs likely has fluctuated over the last 35 years. This study assumed that certain programs remained generally small or large during the course of their existence, which was controlled by using the ratio between faculty and residents. Additionally, the number of years that a program has been in existence influences the likelihood of that program to graduate higher numbers of residents who become chairs/chiefs or PDs. As a result, we used multiple linear regression to control for the number of residents and number of years that a program had been in existence. Finally, while the relationship between academic leaders and research may be explained by the increased likelihood of faculty with more funding, publications, or lectures to be selected for leadership roles, this finding supports the notion that research can contribute to leadership. This analysis identifies modifiable factors among residency training programs to improve the odds of graduating future academic leaders.
Conclusion
As the present study shows, the ratio of faculty to residents and the number of full-time faculty publications are key to graduating academic leaders in dermatology. Retention of faculty as leaders in academic dermatology is as crucial to the field as recruitment of residents into academic dermatology. Mentorship should be highly encouraged through the creation of formal programs and should not end at the resident level. Emphasizing the intellectually stimulating aspects of academia and providing administrative resources may help decrease the burden of academic duties, allowing the pursuit of teaching and research and ultimately the resources to become candidates for leadership positions in academia.
Leadership is widely recognized as a key component in the role of a physician,1 which is especially true in dermatology, a specialty that faces severe challenges in the recruitment and retention of academic faculty.2 A study of the dermatology workforce found that academic institutions are more likely to be seeking to hire new faculty2 and that many dermatology residency programs often are looking to replace chairpersons (chairs) and/or chiefs.3 Although fewer dermatology residents are pursuing academic careers than careers in private practice, full-time faculty members also are leaving their academic posts. This shift is demonstrated by the younger mean age of academic dermatologists2 and the increased rate of departure from academia prior to pursuing more formalized leadership roles.4
It has been suggested that the number of full-time faculty and number of faculty publications positively influence graduates of dermatology residency programs to pursue academic careers; however, variables affecting the likelihood of graduates of dermatology residency programs becoming academic leaders later in their career have not been well studied.3 The purpose of this study is to determine the factors that influence the development of program chairs/chiefs and program directors (PDs) of dermatology residency programs.
Methods
Data were collected from all accredited dermatology residency programs in the United States as of December 31, 2008. Residency programs that were started after 2004 were excluded from the study, as it was thought that these programs may not have graduated a sufficient number of residents for assessment. Military residency programs also were excluded, as graduates from these programs often do not freely choose their careers after residency.
Primary end points were the number of chairs/chiefs and PDs who had graduated from each dermatology residency program. Variables included the number of years the program had been in existence, status of the program as a department or division, number of full-time faculty members, number of residents, National Institutes of Health funding received in 2008 (http://www.report.nih.gov/award/index.cfm), Dermatology Foundation (DF) funding received (http://www.dermatologyfoundation.org/rap/), number of publications from full-time faculty members in 2008 (http://www.ncbi.nlm.nih.gov/pubmed/), number of faculty lectures given at annual meetings of 5 societies in 2008 (American Academy of Dermatology, the Society for Investigative Dermatology, the American Society of Dermatopathology, the Society for Pediatric Dermatology, and American Society for Dermatologic Surgery), and the number of faculty members on the editorial boards of 6 major dermatology journals (Journal of the American Academy of Dermatology, Journal of Investigative Dermatology, Archives of Dermatology [currently known as JAMA Dermatology], Dermatologic Surgery, Pediatric Dermatology, and Journal of Cutaneous Pathology). Data regarding faculty and residents were obtained from program Web sites and inquiries from individual programs. The year 1974 was used as a cutoff for the number of years a program had been in existence. Years of existence of a program was controlled for in the analysis. The ratio of faculty to residents was calculated per year and categorized as 4 or more or less than 4 to minimize the effect of changing program size over the years. For faculty members who split time between 2 residency programs, each program was given credit for the duration of time spent at that program. Faculty members who hold a PhD only and those who completed their residencies in non-US dermatology residency programs were excluded from the outcome variables. To avoid duplicate faculty publications, collections for each residency program were created within PubMed (ie, if 2 authors from the same program coauthored an article, it was only counted once toward the total number of faculty publications from that program).
Because the data were skewed (ie, there were a large number of programs with 0 graduating chairs/chiefs and PDs), nonparametric analyses were utilized. Logistic regression was used to calculate the odds of producing chairs/chiefs or PDs (yes vs no). Multiple logistic regression helped to determine those variables that were most closely associated with odds of graduating a chair/chief or PD. Variables with a significance level of P<.10 were considered in the multiple logistic regression, and backward selection was used to determine a model. Multiple linear regression was used to determine correlation coefficients for each of the variables and the number of chairs/chiefs or PDs graduated, controlling for the estimated number of graduates from the program and number of years the program had been in existence. Analyses for graduating chairs/chiefs and PDs were conducted separately. The final significance level used was P<.05. Data were analyzed using SAS version 9.3. This study was approved by the institutional review board at Kaiser Permanente Southern California.
Results
Data from 103 dermatology residency programs were included in the analysis. Of these programs, 47 had graduated at least 1 chair/chief and 55 had graduated at least 1 PD. Among the programs graduating any chairs/chiefs, they produced an average of 2.04 chairs/chiefs and 1.86 PDs. The 5 dermatology residency programs that graduated the highest total number of chairs/chiefs and PDs were Harvard University (Cambridge, Massachusetts), the University of Michigan (Ann Arbor, Michigan), New York University (New York, New York), Yale-New Haven Hospital (New Haven, Connecticut), and the University of Minnesota (Minneapolis, Minnesota).
Factors that had the highest effect on the odds of a program graduating a chair/chief included the ratio of faculty to residents per year, presence of DF funding in 2008, number of years program was in existence, number of residents, number of full-time faculty, and number of full-time faculty on editorial boards of 6 major dermatology journals (Table 1). When controlling for each of these variables in the final multivariable analysis, programs with 4 or more faculty per resident had 3.31 times the odds of producing a chair/chief (95% confidence interval [CI], 1.14-9.66; P=.028).
Factors that had the highest effect on the odds of a program graduating a PD included status as department versus division, ratio of faculty to residents per year, presence of DF funding in 2008, number of lectures given by full-time faculty members at annual society meetings, number of residents, number of years program was in existence, number of full-time faculty, and number of publications from full-time faculty members (Table 2). The most significant factor associated with graduating PDs after controlling for other variables was the number of publications from full-time faculty members. The odds increased by 3.2% for every 1 additional publication and 32% for every 10 additional publications (95% CI, 1.01-1.06; P=.026).
Multiple linear regression demonstrated a positive relationship between the number of graduating chairs/chiefs and total full-time faculty members (R2=0.26; P=.034) and ratio of full-time faculty to residents (R2=0.29; P<.001). Marginally significant correlations were seen between the number of PDs and ratio of full-time faculty to residents (R2=0.32; P=.05) as well as the number of publications from full-time faculty members (R2=0.32; P=.05).
Comment
The ratio of full-time faculty to residents increased a program’s odds of graduating a chair/chief. More faculty members may lead to more opportunities for mentorship of residents and young faculty. Mentors are widely perceived to be integral to the learning and development of residents, not only in dermatology5 but across all specialties.6 Mentors also have been noted to play a key role in bolstering and maintaining interest in academics,7 which is true not only with regard to recruiting new residents but for retaining young faculty members. In a study (N=109) that examined factors associated with residents’ loss of interest in academic careers, half of the participants reported a lack of effective mentors, role models, and professional guidance.8 Mentors provide teaching, supervision, and advice, especially with regard to research and career paths.9 A large number of faculty members provides more opportunities for direct mentorship and offers residents more exposure to research, specialty clinics, and academic philosophies, which may positively influence and even inspire academic pursuits and leadership.3
Although the solution to producing future chairs/chiefs and PDs may lie in faculty guidance, finding and retaining faculty members as mentors amidst a shortage of academic dermatologists presents an underlying issue.3 In addition to a lack of mentorship, residents cite bureaucracy, salary differentials, and location to explain a loss of interest in academic careers.8 Several programs have been developed to address the recruitment of dermatology residents for academic careers, including combined medical-dermatology programs, 2+2 programs (2 years of clinical residency plus 2 additional research years), clinical research fellowships,10 and the Society for Investigative Dermatology’s Dermatology Resident Retreat for Future Academicians (http://www.sidnet.org/fortraineesandresidents).11 Perhaps recruitment should even start at the medical student level. In light of the academic strength of the current pool of dermatology residency applicants,12 training programs should continue to screen for applicants with sincere interests in academia.13 Students with more research and publications may be more likely to pursue academic careers, in accordance with prior studies of dermatology trainees.3,14 Studies also have shown that graduates of foreign dermatology residencies15 and individuals who hold both MD and PhD degrees may be more likely to enter into academic careers.16
For creating future chairs/chiefs and PDs, retention of young faculty in academics is as important as recruiting residents.17 At the mid-career level, the decline of funds for research has generated pressure for academic physicians to see increasing numbers of patients, leaving insufficient time for the many duties that accompany academic posts,2 including teaching and publishing. Other reasons that faculty members leave their posts before 40 years of age include financial and family concerns18 as well as the desire for more autonomy.4 Formalized training is seen with the American Academy of Dermatology’s Academic Dermatology Leadership Program (https://www.aad.org/members/leadership-institute/mentoring/aad-mentoring-opportunities/academic-dermatology-leadership-program-mentee), which promotes advanced leadership training to recent graduates.5 Other methods include support of young faculty with mentorship; grant applications; and administration at the department, hospital, and government levels.17 Recruitment of faculty from private practice may represent another potential source of faculty who wish to pursue more scholarly endeavors.4 Teaching has been cited as a primary reason for faculty members to remain in academia,18 and thus time for teaching must be protected. Such a strategy is in accordance with our findings that amount of annual DF funding received, number of full-time faculty publications, number of faculty members on editorial boards of major dermatology journals, and number of lectures given by full-time faculty factors at annual society meetings are positively associated with the odds of producing chairs/chiefs or PDs. In particular, the number of full-time faculty publications is directly related to increased odds of graduates becoming PDs. Residents and young faculty members who take part in research and attend national conferences may find inspiration or develop a passion for academic leadership.
A limitation to this study is that the ratio of faculty to graduated residents for some programs likely has fluctuated over the last 35 years. This study assumed that certain programs remained generally small or large during the course of their existence, which was controlled by using the ratio between faculty and residents. Additionally, the number of years that a program has been in existence influences the likelihood of that program to graduate higher numbers of residents who become chairs/chiefs or PDs. As a result, we used multiple linear regression to control for the number of residents and number of years that a program had been in existence. Finally, while the relationship between academic leaders and research may be explained by the increased likelihood of faculty with more funding, publications, or lectures to be selected for leadership roles, this finding supports the notion that research can contribute to leadership. This analysis identifies modifiable factors among residency training programs to improve the odds of graduating future academic leaders.
Conclusion
As the present study shows, the ratio of faculty to residents and the number of full-time faculty publications are key to graduating academic leaders in dermatology. Retention of faculty as leaders in academic dermatology is as crucial to the field as recruitment of residents into academic dermatology. Mentorship should be highly encouraged through the creation of formal programs and should not end at the resident level. Emphasizing the intellectually stimulating aspects of academia and providing administrative resources may help decrease the burden of academic duties, allowing the pursuit of teaching and research and ultimately the resources to become candidates for leadership positions in academia.
1. Wood LD, Miller JJ, Marks JG Jr. The need for leadership: how can we better train the next generation of dermatologists? J Am Acad Dermatol. 2008;58:715-717.
2. Resneck JS Jr, Tierney EP, Kimball AB. Challenges facing academic dermatology: survey data on the faculty workforce. J Am Acad Dermatol. 2006;54:211-216.
3. Wu JJ, Ramirez CC, Alonso CA, et al. Dermatology residency program characteristics that correlate with graduates selecting an academic dermatology career. Arch Dermatol. 2006;142:845-850.
4. Loo DS, Liu CL, Geller AC, et al. Academic dermatology manpower: issues of recruitment and retention. Arch Dermatol. 2007;143:341-347.
5. Donovan JC. A survey of dermatology residency program directors’ views on mentorship. Dermatol Online J. 2009;15:1.
6. Sambunjak D, Straus SE, Marusi´c A. Mentoring in academic medicine: a systematic review. JAMA. 2006;296:1103-1115.
7. Rubenstein DS, Blauvelt A, Chen SC, et al. The future of academic dermatology in the United States: report on the resident retreat for future physician-scientists, June 15-17, 2001. J Am Acad Dermatol. 2002;47:300-303.
8. Reck SJ, Stratman EJ, Vogel C, et al. Assessment of residents’ loss of interest in academic careers and identification of correctable factors. Arch Dermatol. 2006;142:855-858.
9. Clark RA. Teacher, supervisor, adviser, or mentor? J Invest Dermatol. 2011;131:1779-1780.
10. Wu JJ. Current strategies to address the ongoing shortage of academic dermatologists. J Am Acad Dermatol. 2007;56:1065-1066.
11. Culton DA, Rubenstein DS, Diaz LA. The resident retreat for future academicians. J Invest Dermatol. 2010;130:1775-1777.
12. Wu JJ, Tyring SK. The academic strength of current dermatology residency applicants. Dermatol Online J. 2003;9:22.
13. Kia KF, Gielczyk RA, Ellis CN. Academia is the life for me, I’m sure. Arch Dermatol. 2006;142:911-913.
14. Miller CJ, Wood GC, Miller JJ, et al. Academics or private practice? the future of dermatologic surgery education. Dermatol Surg. 2006;32:70-75, discussion 76-78.
15. Wu JJ, Davis KF, Ramirez CC, et al. Graduates-of-foreign-dermatology residencies and military dermatology residencies and women in academic dermatology. Dermatol Online J. 2009;15:2.
16. Wu JJ, Davis KF, Ramirez CC, et al. MD/PhDs are more likely than MDs to choose a career in academic dermatology. Dermatol Online J. 2008;14:27.
17. Olerud JE. Academic workforce in dermatology. Arch Dermatol. 2007;143:409-410.
18. Turner E, Yoo J, Salter S, et al. Leadership workforce in academic dermatology. Arch Dermatol. 2007;143:948-949.
1. Wood LD, Miller JJ, Marks JG Jr. The need for leadership: how can we better train the next generation of dermatologists? J Am Acad Dermatol. 2008;58:715-717.
2. Resneck JS Jr, Tierney EP, Kimball AB. Challenges facing academic dermatology: survey data on the faculty workforce. J Am Acad Dermatol. 2006;54:211-216.
3. Wu JJ, Ramirez CC, Alonso CA, et al. Dermatology residency program characteristics that correlate with graduates selecting an academic dermatology career. Arch Dermatol. 2006;142:845-850.
4. Loo DS, Liu CL, Geller AC, et al. Academic dermatology manpower: issues of recruitment and retention. Arch Dermatol. 2007;143:341-347.
5. Donovan JC. A survey of dermatology residency program directors’ views on mentorship. Dermatol Online J. 2009;15:1.
6. Sambunjak D, Straus SE, Marusi´c A. Mentoring in academic medicine: a systematic review. JAMA. 2006;296:1103-1115.
7. Rubenstein DS, Blauvelt A, Chen SC, et al. The future of academic dermatology in the United States: report on the resident retreat for future physician-scientists, June 15-17, 2001. J Am Acad Dermatol. 2002;47:300-303.
8. Reck SJ, Stratman EJ, Vogel C, et al. Assessment of residents’ loss of interest in academic careers and identification of correctable factors. Arch Dermatol. 2006;142:855-858.
9. Clark RA. Teacher, supervisor, adviser, or mentor? J Invest Dermatol. 2011;131:1779-1780.
10. Wu JJ. Current strategies to address the ongoing shortage of academic dermatologists. J Am Acad Dermatol. 2007;56:1065-1066.
11. Culton DA, Rubenstein DS, Diaz LA. The resident retreat for future academicians. J Invest Dermatol. 2010;130:1775-1777.
12. Wu JJ, Tyring SK. The academic strength of current dermatology residency applicants. Dermatol Online J. 2003;9:22.
13. Kia KF, Gielczyk RA, Ellis CN. Academia is the life for me, I’m sure. Arch Dermatol. 2006;142:911-913.
14. Miller CJ, Wood GC, Miller JJ, et al. Academics or private practice? the future of dermatologic surgery education. Dermatol Surg. 2006;32:70-75, discussion 76-78.
15. Wu JJ, Davis KF, Ramirez CC, et al. Graduates-of-foreign-dermatology residencies and military dermatology residencies and women in academic dermatology. Dermatol Online J. 2009;15:2.
16. Wu JJ, Davis KF, Ramirez CC, et al. MD/PhDs are more likely than MDs to choose a career in academic dermatology. Dermatol Online J. 2008;14:27.
17. Olerud JE. Academic workforce in dermatology. Arch Dermatol. 2007;143:409-410.
18. Turner E, Yoo J, Salter S, et al. Leadership workforce in academic dermatology. Arch Dermatol. 2007;143:948-949.
Practice Points
- Leadership in dermatology is key to the future of academics.
- Opportunity for mentorship and research are the most important residency program factors leading to the graduation of future chairs/chiefs and program directors.
- The retention of residents and young faculty in academics can be aided by research and scholarly activity.
Sunscreens Causing Cancer? The Facts
Skin cancer is the most common form of cancer in the United States and continues to rise in incidence and mortality each year.1 It is common knowledge that UV light plays a major role in the development of skin cancer.2,3 Studies have long demonstrated that using sunscreen on a daily basis can help prevent the development of skin cancer, premature aging, and exacerbation of photodermatoses.4-7 Although there are several photoprotective measures available, sunscreen remains the most popular and widely used among patients.8 Sunscreens that are on the market today contain either organic or inorganic UV filters or a combination of both based on their chemical composition and photoprotection mechanisms.9 Concerns about these ingredients causing cancer have created confusion among consumers. I will attempt to clarify these concerns by critically analyzing available evidence-based data on sunscreen use so that as dermatology residents we will be more knowledgeable about sunscreen safety topics and will be able to provide accurate and up-to-date information to our patients.
Organic UV Filters
Organic UV filters are classified as aromatic compounds that provide photoprotection by absorbing UV light.10 Aside from the photoallergic potential of organic UV filters, controversy has arisen in response to studies reporting their possible hormone disruptive effects.11-18 Although there are several US Food and Drug Administration (FDA)–approved organic UV filters in use today, one of the most commonly manufactured and controversial agents is oxybenzone.10 Claims regarding the estrogenic and antiandrogenic effects of oxybenzone have been investigated with results refuting the claims or concluding that more sensitive studies are needed to determine if these organic ingredients pose such risks.10,19,20 One study demonstrated that nearly 300 years of daily sunscreen application would be needed to reach similar exposure levels of oxybenzone used and described in prior animal studies.21 Additionally, most of the studied adverse effects of UV filters have been evaluated based on oral exposure rather than actual dermal application.11 Although these compounds are absorbed systemically, studies have reported that the amounts are insignificant and noncumulative in the body.10,22-24 Furthermore, the binding affinity of oxybenzone for estrogen receptors has been shown to be much weaker and near insignificant compared to estrogen and estradiol.24,25 Although numerous important studies examining systemic absorption have not shown a clinically significant disruption of hormonal homeostasis or acute toxicity in humans by organic UV filters, further studies are needed.
Inorganic UV Filters
Used as the main active ingredients in sunscreen for decades, titanium dioxide (TiO2) and zinc oxide (ZnO) compounds generally are more photostable and less photoallergic than their organic counterparts.10 In recent years, the safety of these long-used photoprotectors has been questioned because of the development of nanoparticle (<100 nm) formulas that are less opaque on application. Although this formula provides a thin, transparent, and cosmetically appealing medium, there is concern that the metal oxides penetrate the skin and cause local and systemic toxicities.26-28 Several recent scientific studies have shown no percutaneous permeation of these particles in normal adult human skin and reported no causal damage to mammalian cells.10,29-31 Although skin penetration of TiO2 and ZnO has been described as insignificant, focus has shifted to health risks associated with inhaling TiO2 through the use of spray or powder products following statements made by the International Agency for Research on Cancer in 2006.32 Several studies investigating increased health risks, specifically lung cancer, in factory workers who were subjected to TiO2 and ZnO inhalation concluded that exposure was unlikely to pose substantial health risks or subchronic toxicity.33,34 Despite a relatively strong safety profile, a major concern of using these metal oxides as UV filters has been potential free radical formation.35-39 For this reason, the Scientific Committee on Emerging and Newly Identified Health Risks extensively researched and delivered opinions on the use of TiO2 and ZnO in cosmetics, concluding that topical application of either compound does not result in toxicity or other adverse effects.30,40-42 Additionally, an effort has been made by manufacturers to encapsulate nanoparticles with magnesium and other materials to quench the reactive oxygen species along with the human body’s own antioxidant defense system.10 In summary, it appears that the current weight of scientific evidence suggests that percutaneous absorption and toxicity by UV filters in humans may be overestimated and that the use of nanoparticles in sunscreens poses no or negligible potential risks to human health.43,44
Concerns Beyond Organic and Inorganic UV Filters
Beyond these concerns with organic and inorganic UV filters, there are several other claims regarding sunscreen safety that have stirred up controversy, including the side-effect profile of retinyl palmitate, vitamin D deficiency, phototoxicity, environmental effects, futility of sun protection factor levels greater than 50, and increased health risks in children. Although some studies report mixed results, the majority of scientific investigations have addressed and refuted several of these claims, again confirming the relative safety of sunscreen use. It is beyond the scope of this article to further discuss these topics specifically. However, it is worth mentioning that consumer studies report that the actual use of sunscreens is 0.5 mg/cm2 or less compared to the ideal application of 2 mg/cm2, thereby confounding many of the claims made about sunscreen use, such as vitamin D deficiency.45 Sunscreens often contain a combination of several UV filters. To date, only a few existing studies have shown that mixtures of the photoprotective agents discussed might interact and exhibit toxic activity when combined, even when there is no observed adverse toxic effect when used individually in products.46-48
The current FDA ruling on sunscreen labeling does not require manufacturers to state if inorganic UV filters have been formulated into nanoparticles; however, manufacturers are now required to include a statement on all sunscreen labels warning consumers to avoid using sunscreen on damaged or broken skin49 in an effort to prevent the active ingredients from getting under the skin, potentially causing inflammation and/or health risks, because available data do not provide conclusive evidence on increased penetration of open skin.50 Additional information regarding the 2011 FDA sunscreen ruling can be found in a prior Cutis Resident Corner column.51
Final Thoughts
As health care providers, we should take advantage of opportunities to educate our patients about other sun safety practices, such as avoiding excessive sun exposure during peak hours (10 am to 2 pm), seeking shade, and wearing photoprotective clothing (eg, wide-brimmed hats, sunglasses).
The research is quite clear: Using broadband sunscreens that absorb and/or block UV radiation results in reduced damage to the skin’s DNA, a fact that should be considered when taking into account the risks and benefits of sunscreen use.2,3 Although sunscreen use is highly recommended in addition to the other sun protection methods, it is ultimately the patient’s choice. If a patient is still concerned about the active ingredients of UV filters, even given the high probability of safety, there are products available on the market that do not include organic filters or nanoparticles. Given the established benefits of UV protection, the use of sunscreens remain one of the most important photoprotective methods, and with increased usage by the public, continuous monitoring of the overall safety and benefit profile of future products is prudent.
1. Skin cancer statistics. Centers for Disease Control and Prevention Web site. http://www.cdc.gov/cancer/skin/statistics/index.htm. Updated September 2, 2014. Accessed December 30, 2014.
2. World Health Organization, International Agency for Research on Cancer. Solar and ultraviolet radiation. In: IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. Vol 55. Lyon, France: International Agency for Research on Cancer; 1992.
3. Green AC, Williams GM, Logan V, et al. Reduced melanoma after regular sunscreen use: randomized trial follow-up. J Clin Oncol. 2011;29:257-263.
4. Darlington S, Williams G, Neale R, et al. A randomized controlled trial to assess sunscreen application and beta carotene supplementation in the prevention of solar keratoses. Arch Dermatol. 2003;139:451-455.
5. Van der Pols JC, Williams GM, Pandeya N, et al. Prolonged prevention of squamous cell carcinoma of the skin by regular sunscreen use. Cancer Epidemiol Biomarkers Prev. 2006;15:2546-2548.
6. Hughes MC, Williams GM, Baker P, et al. Sunscreen and prevention of skin aging: a randomized trial. Ann Intern Med. 2013;158:781-790.
7. Bissonnette R, Nigen S, Bolduc C. Influence of the quantity of sunscreen applied on the ability to protect against ultraviolet-induced polymorphous light eruption. Photodermatol Photoimmunol Photomed. 2012;28:240-243.
8. Cancer trends progress report 2011/2012 update: sun protection. National Cancer Institute Web site. http://progressreport.cancer.gov/doc_detail.asp?pid¡1&did¡2009&chid¡91&coid¡911. Accessed December 30, 2014.
9. Sunscreen Drug Products for Over-the-counter Human Use, 21 CFR §352.10. http://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/cfrsearch.cfm?fr=352.10. Updated September 1, 2014. Accessed December 30, 2014.
10. Burnett ME, Wang SQ. Current sunscreen controversies: a critical review. Photodermatol Photoimmunol Photomed. 2011;27:58-67.
11. Krause M, Klit A, Blomberg Jensen M, et al. Sunscreens: are they beneficial for health? an overview of endocrine disrupting properties of UV-filters. Int J Androl. 2012;35:424-436.
12. Schlumpf M, Cotton B, Conscience M, et al. In vitro and in vivo estrogenicity of UV screens. Environ Health Perspect. 2001;109:239-244.
13. Schlumpf M, Schmid P, Durrer S, et al. Endocrine activity and developmental toxicity of cosmetic UV filters–an update. Toxicol. 2004;205:113-122.
14. Schlumpf M, Kypke K, Vökt C, et al. Endocrine active UV filters: developmental toxicity and exposure through breast milk. Chimia. 2008;62:345-351.
15. Nakagawa Y, Suzuki T. Metabolism of 2-hydroxy-4-methoxybenzophenone in isolated rat hepatocytes and xenoestrogenic effects of its metabolites on MCF-7 human breast cancer cells. Chem Biol Interact. 2002;139:115-128.
16. Ma R, Cotton B, Lichtensteiger W, et al. UV filters with antagonistic action at androgen receptors in the MDA-kb2 cell transcriptional-activation assay. Toxicol Sci. 2003;74:43-50.
17. Heneweer M, Muusse M, van den Berg M, et al. Additive estrogenic effects of mixtures of frequently used UV filters on pS2-gene transcription in MCF-7 cells. Toxicol Appl Pharmacol. 2005;208:170-177.
18. Knobler E, Almeida L, Ruzkowski AM, et al. Photoallergy to benzophenone. Arch Dermatol. 1989;125:801-804.
19. Draelos ZD. Are sunscreens safe? J Cosmet Dermatol. 2010;9:1-2.
20. Gilbert E, Pirot F, Bertholle V. Commonly used UV filter toxicity on biological functions: review of last decade studies. Int J of Cosmet Sci. 2013;35:208-219.
21. Wang SQ, Burnett ME, Lim HW. Safety of oxybenzone: putting numbers into perspective. Arch Dermatol. 2011;147:865-866.
22. Mancebo SE, Hu JY, Wang SQ. Sunscreens: a review of health benefits, regulations, and controversies. Dermatol Clin. 2014;32:427-438.
23. Jansen R, Osterwalder U, Wang SQ, et al. Photoprotection: part II. sunscreen: development, efficacy, and controversies. J Am Acad Dermatol. 2013;69:867.e1-867.e14.
24. Janjua NR, Mogensen B, Andersson AM, et al. Systemic absorption of the sunscreens benzo- phenone-3, octyl-methoxycinnamate, and 3-(4-methyl-benzy-lidene) camphor after whole-body topical application and reproductive hormone levels in humans. J Invest Dermatol. 2004;123:57-61.
25. Kadry AM, Chukwuemeka SO, Mohamed S, et al. Pharmacokinetics of benzophenone-3 after oral exposure in male rats. J Appl Toxicol. 1995;15:97-102.
26. Gulson B, McCall M, Korsch M, et al. Small amounts of zinc from zinc oxide particles in sunscreens applied outdoors are absorbed through human skin. Toxicol Sci. 2010;118:140-149.
27. Gulson B, Wong H, Korsch M, et al. Comparison of dermal absorption of zinc from different sunscreen formulations and differing UV exposure based on stable isotope tracing. Sci Total Environ. 2012:420:313-318.
28. Benech-Kieffer F, Meuling WJ, Leclerc C, et al. Percutaneous absorption of Mexoryl SX in human volunteers: comparison with in vitro data. Skin Pharmacol Appl Skin Physiol. 2003;16:343-355.
29. Nash JF. Human safety and efficacy of ultraviolet filters and sunscreen products. Dermatol Clin. 2006;24:35-51.
30. Nohynek GJ, Lademann J, Ribaud C, et al. Grey goo on the skin? nanotechnology, cosmetic and sunscreen safety. Crit Rev Toxicol. 2007;37:251-277.
31. Sadrieh N, Wokovich AM, Gopee NV, et al. Lack of significant dermal penetration of titanium dioxide from sunscreen formulations containing nano- and submicron-size TiO2 particles. Toxicol Sci. 2010;115:156-166.
32. International Agency for Research on Cancer. Carbon black, titanium dioxide, and talc. In: IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. Vol 93. Lyon, France: International Agency for Research on Cancer; 2006.
33. Liao CM, Chiang YH, Chio CP. Model-based assessment for human inhalation exposure risk to airborne nano/fine titanium dioxide particles. Sci Total Environ. 2008:15;407:165-177.
34. Adamcakova-Dodd A, Stebounova LV, Kim JS, et al. Toxicity assessment of zinc oxide nanoparticles using sub-acute and sub-chronic murine inhalation models. Part Fibre Toxicol. 2014;11:15.
35. Wamer WG, Yin JJ, Wei RR. Oxidative damage to nucleic acids photosensitized by titanium dioxide. Free Radic Biol Med. 1997;23:851-858.
36. Nakagawa Y, Wakuri S, Sakamoto K, et al. The photogenotoxicity of titanium dioxide particles. Mutat Res. 1997;394:125-132.
37. Dunford R, Salinaro A, Cai L, et al. Chemical oxidation and DNA damage catalysed by inorganic sunscreen ingredients. FEBS Lett. 1997;418:87-90, 99.
38. Hidaka H, Kobayashi H, Koike T, et al. DNA damage photoinduced by cosmetic pigments and sunscreen agents under solar exposure and artificial UV illumination. J Oleo Sci. 2006;55:249-261.
39. Dufour EK, Kumaravel T, Nohynek GJ, et al. Clastogenicity, photo-clastogenicity or pseudo-photo-clastogenicity: genotoxic effects of zinc oxide in the dark, in pre-irradiated or simultaneously irradiated Chinese hamster ovary cells [published online ahead of print June 21, 2006]. Mutat Res. 2006;607:215-224.
40. Opinion of the Scientific Committee on Cosmetic Products and Non-Food Products intended for Consumers concerning titanium dioxide. http://ec.europa.eu/health/archive/ph_risk/committees/sccp/documents/out135_en.pdf. Published October 24, 2000. Accessed December 30, 2014.
41. The Scientific Committee on Cosmetic Products and Non-Food Products intended for Consumers opinion concerning zinc oxide. http://ec.europa.eu/health/archive/ph_risk/committees/sccp/documents/out222_en.pdf. Published June 24-25, 2003. Accessed December 30, 2014.
42. Hackenberg S, Friehs G, Kessler M, et al. Nanosized titanium dioxide particles do not induce DNA damage in human peripheral blood lymphocytes. Environ Mol Mutagen. 2010;52:264-268.
43. Bach-Thomsen M, Wulf HC. Sunbather’s application of sunscreen is probably inadequate to obtain the sun protection factor assigned to the preparation. Photodermatol Photoimmunol Photomed. 1993:9;242-244.
44. Nohynek GJ, Antignac E, Re T, et al. Safety assessment of personal care products/cosmetics and their ingredients. Toxicol Appl Pharmacol. 2010:1;243:239-259.
45. Diffey BL. Sunscreens: use and misuse. In: Giacomoni PU, ed. Sun Protection in Man. Vol 3. Amsterdam, the Netherlands: Elsevier Science BV; 2001:521-534.
46. Heneweer M, Muusse M, Van den BM, et al. Additive estrogenic effects of mixtures of frequently used UV-filters on pS2-gene transcription in MCF-7 cells. Toxicol Appl Pharmacol. 2005;208:170-177.
47. Kunz PY, Galicia HF, Fent K. Comparison of in vitro and in vivo estrogenic activity of UV-filters in fish. Toxicol Sci. 2006;90:349-361.
48. Kortenkamp A, Faust M, Scholze M, et al. Low-level exposure to multiple chemicals: reason for human health concerns? Environ Health Perspect. 2007;115(suppl 1):106-114.
49. Labeling and effectiveness testing: sunscreen drug products for over-the-counter human use—small entity compliance guide. US Food and Drug Administration Web site. http://www.fda.gov/drugs/guidancecomplianceregulatoryinformation/guidances/ucm330694.htm. Published December 2012. Updated May 13, 2014. Accessed December 30, 2014.
50. Schafer-Korting M, Korting HC, Ponce-Poschl E. Liposomal tretinoin for uncomplicated acne vulgaris. Clin Investig. 1994;72:1086-1091.
51. Bronfenbrener R. Simplifying sun safety: a guide to the new FDA sunscreen monograph. Cutis. 2014;93:e17-e19.
Skin cancer is the most common form of cancer in the United States and continues to rise in incidence and mortality each year.1 It is common knowledge that UV light plays a major role in the development of skin cancer.2,3 Studies have long demonstrated that using sunscreen on a daily basis can help prevent the development of skin cancer, premature aging, and exacerbation of photodermatoses.4-7 Although there are several photoprotective measures available, sunscreen remains the most popular and widely used among patients.8 Sunscreens that are on the market today contain either organic or inorganic UV filters or a combination of both based on their chemical composition and photoprotection mechanisms.9 Concerns about these ingredients causing cancer have created confusion among consumers. I will attempt to clarify these concerns by critically analyzing available evidence-based data on sunscreen use so that as dermatology residents we will be more knowledgeable about sunscreen safety topics and will be able to provide accurate and up-to-date information to our patients.
Organic UV Filters
Organic UV filters are classified as aromatic compounds that provide photoprotection by absorbing UV light.10 Aside from the photoallergic potential of organic UV filters, controversy has arisen in response to studies reporting their possible hormone disruptive effects.11-18 Although there are several US Food and Drug Administration (FDA)–approved organic UV filters in use today, one of the most commonly manufactured and controversial agents is oxybenzone.10 Claims regarding the estrogenic and antiandrogenic effects of oxybenzone have been investigated with results refuting the claims or concluding that more sensitive studies are needed to determine if these organic ingredients pose such risks.10,19,20 One study demonstrated that nearly 300 years of daily sunscreen application would be needed to reach similar exposure levels of oxybenzone used and described in prior animal studies.21 Additionally, most of the studied adverse effects of UV filters have been evaluated based on oral exposure rather than actual dermal application.11 Although these compounds are absorbed systemically, studies have reported that the amounts are insignificant and noncumulative in the body.10,22-24 Furthermore, the binding affinity of oxybenzone for estrogen receptors has been shown to be much weaker and near insignificant compared to estrogen and estradiol.24,25 Although numerous important studies examining systemic absorption have not shown a clinically significant disruption of hormonal homeostasis or acute toxicity in humans by organic UV filters, further studies are needed.
Inorganic UV Filters
Used as the main active ingredients in sunscreen for decades, titanium dioxide (TiO2) and zinc oxide (ZnO) compounds generally are more photostable and less photoallergic than their organic counterparts.10 In recent years, the safety of these long-used photoprotectors has been questioned because of the development of nanoparticle (<100 nm) formulas that are less opaque on application. Although this formula provides a thin, transparent, and cosmetically appealing medium, there is concern that the metal oxides penetrate the skin and cause local and systemic toxicities.26-28 Several recent scientific studies have shown no percutaneous permeation of these particles in normal adult human skin and reported no causal damage to mammalian cells.10,29-31 Although skin penetration of TiO2 and ZnO has been described as insignificant, focus has shifted to health risks associated with inhaling TiO2 through the use of spray or powder products following statements made by the International Agency for Research on Cancer in 2006.32 Several studies investigating increased health risks, specifically lung cancer, in factory workers who were subjected to TiO2 and ZnO inhalation concluded that exposure was unlikely to pose substantial health risks or subchronic toxicity.33,34 Despite a relatively strong safety profile, a major concern of using these metal oxides as UV filters has been potential free radical formation.35-39 For this reason, the Scientific Committee on Emerging and Newly Identified Health Risks extensively researched and delivered opinions on the use of TiO2 and ZnO in cosmetics, concluding that topical application of either compound does not result in toxicity or other adverse effects.30,40-42 Additionally, an effort has been made by manufacturers to encapsulate nanoparticles with magnesium and other materials to quench the reactive oxygen species along with the human body’s own antioxidant defense system.10 In summary, it appears that the current weight of scientific evidence suggests that percutaneous absorption and toxicity by UV filters in humans may be overestimated and that the use of nanoparticles in sunscreens poses no or negligible potential risks to human health.43,44
Concerns Beyond Organic and Inorganic UV Filters
Beyond these concerns with organic and inorganic UV filters, there are several other claims regarding sunscreen safety that have stirred up controversy, including the side-effect profile of retinyl palmitate, vitamin D deficiency, phototoxicity, environmental effects, futility of sun protection factor levels greater than 50, and increased health risks in children. Although some studies report mixed results, the majority of scientific investigations have addressed and refuted several of these claims, again confirming the relative safety of sunscreen use. It is beyond the scope of this article to further discuss these topics specifically. However, it is worth mentioning that consumer studies report that the actual use of sunscreens is 0.5 mg/cm2 or less compared to the ideal application of 2 mg/cm2, thereby confounding many of the claims made about sunscreen use, such as vitamin D deficiency.45 Sunscreens often contain a combination of several UV filters. To date, only a few existing studies have shown that mixtures of the photoprotective agents discussed might interact and exhibit toxic activity when combined, even when there is no observed adverse toxic effect when used individually in products.46-48
The current FDA ruling on sunscreen labeling does not require manufacturers to state if inorganic UV filters have been formulated into nanoparticles; however, manufacturers are now required to include a statement on all sunscreen labels warning consumers to avoid using sunscreen on damaged or broken skin49 in an effort to prevent the active ingredients from getting under the skin, potentially causing inflammation and/or health risks, because available data do not provide conclusive evidence on increased penetration of open skin.50 Additional information regarding the 2011 FDA sunscreen ruling can be found in a prior Cutis Resident Corner column.51
Final Thoughts
As health care providers, we should take advantage of opportunities to educate our patients about other sun safety practices, such as avoiding excessive sun exposure during peak hours (10 am to 2 pm), seeking shade, and wearing photoprotective clothing (eg, wide-brimmed hats, sunglasses).
The research is quite clear: Using broadband sunscreens that absorb and/or block UV radiation results in reduced damage to the skin’s DNA, a fact that should be considered when taking into account the risks and benefits of sunscreen use.2,3 Although sunscreen use is highly recommended in addition to the other sun protection methods, it is ultimately the patient’s choice. If a patient is still concerned about the active ingredients of UV filters, even given the high probability of safety, there are products available on the market that do not include organic filters or nanoparticles. Given the established benefits of UV protection, the use of sunscreens remain one of the most important photoprotective methods, and with increased usage by the public, continuous monitoring of the overall safety and benefit profile of future products is prudent.
Skin cancer is the most common form of cancer in the United States and continues to rise in incidence and mortality each year.1 It is common knowledge that UV light plays a major role in the development of skin cancer.2,3 Studies have long demonstrated that using sunscreen on a daily basis can help prevent the development of skin cancer, premature aging, and exacerbation of photodermatoses.4-7 Although there are several photoprotective measures available, sunscreen remains the most popular and widely used among patients.8 Sunscreens that are on the market today contain either organic or inorganic UV filters or a combination of both based on their chemical composition and photoprotection mechanisms.9 Concerns about these ingredients causing cancer have created confusion among consumers. I will attempt to clarify these concerns by critically analyzing available evidence-based data on sunscreen use so that as dermatology residents we will be more knowledgeable about sunscreen safety topics and will be able to provide accurate and up-to-date information to our patients.
Organic UV Filters
Organic UV filters are classified as aromatic compounds that provide photoprotection by absorbing UV light.10 Aside from the photoallergic potential of organic UV filters, controversy has arisen in response to studies reporting their possible hormone disruptive effects.11-18 Although there are several US Food and Drug Administration (FDA)–approved organic UV filters in use today, one of the most commonly manufactured and controversial agents is oxybenzone.10 Claims regarding the estrogenic and antiandrogenic effects of oxybenzone have been investigated with results refuting the claims or concluding that more sensitive studies are needed to determine if these organic ingredients pose such risks.10,19,20 One study demonstrated that nearly 300 years of daily sunscreen application would be needed to reach similar exposure levels of oxybenzone used and described in prior animal studies.21 Additionally, most of the studied adverse effects of UV filters have been evaluated based on oral exposure rather than actual dermal application.11 Although these compounds are absorbed systemically, studies have reported that the amounts are insignificant and noncumulative in the body.10,22-24 Furthermore, the binding affinity of oxybenzone for estrogen receptors has been shown to be much weaker and near insignificant compared to estrogen and estradiol.24,25 Although numerous important studies examining systemic absorption have not shown a clinically significant disruption of hormonal homeostasis or acute toxicity in humans by organic UV filters, further studies are needed.
Inorganic UV Filters
Used as the main active ingredients in sunscreen for decades, titanium dioxide (TiO2) and zinc oxide (ZnO) compounds generally are more photostable and less photoallergic than their organic counterparts.10 In recent years, the safety of these long-used photoprotectors has been questioned because of the development of nanoparticle (<100 nm) formulas that are less opaque on application. Although this formula provides a thin, transparent, and cosmetically appealing medium, there is concern that the metal oxides penetrate the skin and cause local and systemic toxicities.26-28 Several recent scientific studies have shown no percutaneous permeation of these particles in normal adult human skin and reported no causal damage to mammalian cells.10,29-31 Although skin penetration of TiO2 and ZnO has been described as insignificant, focus has shifted to health risks associated with inhaling TiO2 through the use of spray or powder products following statements made by the International Agency for Research on Cancer in 2006.32 Several studies investigating increased health risks, specifically lung cancer, in factory workers who were subjected to TiO2 and ZnO inhalation concluded that exposure was unlikely to pose substantial health risks or subchronic toxicity.33,34 Despite a relatively strong safety profile, a major concern of using these metal oxides as UV filters has been potential free radical formation.35-39 For this reason, the Scientific Committee on Emerging and Newly Identified Health Risks extensively researched and delivered opinions on the use of TiO2 and ZnO in cosmetics, concluding that topical application of either compound does not result in toxicity or other adverse effects.30,40-42 Additionally, an effort has been made by manufacturers to encapsulate nanoparticles with magnesium and other materials to quench the reactive oxygen species along with the human body’s own antioxidant defense system.10 In summary, it appears that the current weight of scientific evidence suggests that percutaneous absorption and toxicity by UV filters in humans may be overestimated and that the use of nanoparticles in sunscreens poses no or negligible potential risks to human health.43,44
Concerns Beyond Organic and Inorganic UV Filters
Beyond these concerns with organic and inorganic UV filters, there are several other claims regarding sunscreen safety that have stirred up controversy, including the side-effect profile of retinyl palmitate, vitamin D deficiency, phototoxicity, environmental effects, futility of sun protection factor levels greater than 50, and increased health risks in children. Although some studies report mixed results, the majority of scientific investigations have addressed and refuted several of these claims, again confirming the relative safety of sunscreen use. It is beyond the scope of this article to further discuss these topics specifically. However, it is worth mentioning that consumer studies report that the actual use of sunscreens is 0.5 mg/cm2 or less compared to the ideal application of 2 mg/cm2, thereby confounding many of the claims made about sunscreen use, such as vitamin D deficiency.45 Sunscreens often contain a combination of several UV filters. To date, only a few existing studies have shown that mixtures of the photoprotective agents discussed might interact and exhibit toxic activity when combined, even when there is no observed adverse toxic effect when used individually in products.46-48
The current FDA ruling on sunscreen labeling does not require manufacturers to state if inorganic UV filters have been formulated into nanoparticles; however, manufacturers are now required to include a statement on all sunscreen labels warning consumers to avoid using sunscreen on damaged or broken skin49 in an effort to prevent the active ingredients from getting under the skin, potentially causing inflammation and/or health risks, because available data do not provide conclusive evidence on increased penetration of open skin.50 Additional information regarding the 2011 FDA sunscreen ruling can be found in a prior Cutis Resident Corner column.51
Final Thoughts
As health care providers, we should take advantage of opportunities to educate our patients about other sun safety practices, such as avoiding excessive sun exposure during peak hours (10 am to 2 pm), seeking shade, and wearing photoprotective clothing (eg, wide-brimmed hats, sunglasses).
The research is quite clear: Using broadband sunscreens that absorb and/or block UV radiation results in reduced damage to the skin’s DNA, a fact that should be considered when taking into account the risks and benefits of sunscreen use.2,3 Although sunscreen use is highly recommended in addition to the other sun protection methods, it is ultimately the patient’s choice. If a patient is still concerned about the active ingredients of UV filters, even given the high probability of safety, there are products available on the market that do not include organic filters or nanoparticles. Given the established benefits of UV protection, the use of sunscreens remain one of the most important photoprotective methods, and with increased usage by the public, continuous monitoring of the overall safety and benefit profile of future products is prudent.
1. Skin cancer statistics. Centers for Disease Control and Prevention Web site. http://www.cdc.gov/cancer/skin/statistics/index.htm. Updated September 2, 2014. Accessed December 30, 2014.
2. World Health Organization, International Agency for Research on Cancer. Solar and ultraviolet radiation. In: IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. Vol 55. Lyon, France: International Agency for Research on Cancer; 1992.
3. Green AC, Williams GM, Logan V, et al. Reduced melanoma after regular sunscreen use: randomized trial follow-up. J Clin Oncol. 2011;29:257-263.
4. Darlington S, Williams G, Neale R, et al. A randomized controlled trial to assess sunscreen application and beta carotene supplementation in the prevention of solar keratoses. Arch Dermatol. 2003;139:451-455.
5. Van der Pols JC, Williams GM, Pandeya N, et al. Prolonged prevention of squamous cell carcinoma of the skin by regular sunscreen use. Cancer Epidemiol Biomarkers Prev. 2006;15:2546-2548.
6. Hughes MC, Williams GM, Baker P, et al. Sunscreen and prevention of skin aging: a randomized trial. Ann Intern Med. 2013;158:781-790.
7. Bissonnette R, Nigen S, Bolduc C. Influence of the quantity of sunscreen applied on the ability to protect against ultraviolet-induced polymorphous light eruption. Photodermatol Photoimmunol Photomed. 2012;28:240-243.
8. Cancer trends progress report 2011/2012 update: sun protection. National Cancer Institute Web site. http://progressreport.cancer.gov/doc_detail.asp?pid¡1&did¡2009&chid¡91&coid¡911. Accessed December 30, 2014.
9. Sunscreen Drug Products for Over-the-counter Human Use, 21 CFR §352.10. http://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/cfrsearch.cfm?fr=352.10. Updated September 1, 2014. Accessed December 30, 2014.
10. Burnett ME, Wang SQ. Current sunscreen controversies: a critical review. Photodermatol Photoimmunol Photomed. 2011;27:58-67.
11. Krause M, Klit A, Blomberg Jensen M, et al. Sunscreens: are they beneficial for health? an overview of endocrine disrupting properties of UV-filters. Int J Androl. 2012;35:424-436.
12. Schlumpf M, Cotton B, Conscience M, et al. In vitro and in vivo estrogenicity of UV screens. Environ Health Perspect. 2001;109:239-244.
13. Schlumpf M, Schmid P, Durrer S, et al. Endocrine activity and developmental toxicity of cosmetic UV filters–an update. Toxicol. 2004;205:113-122.
14. Schlumpf M, Kypke K, Vökt C, et al. Endocrine active UV filters: developmental toxicity and exposure through breast milk. Chimia. 2008;62:345-351.
15. Nakagawa Y, Suzuki T. Metabolism of 2-hydroxy-4-methoxybenzophenone in isolated rat hepatocytes and xenoestrogenic effects of its metabolites on MCF-7 human breast cancer cells. Chem Biol Interact. 2002;139:115-128.
16. Ma R, Cotton B, Lichtensteiger W, et al. UV filters with antagonistic action at androgen receptors in the MDA-kb2 cell transcriptional-activation assay. Toxicol Sci. 2003;74:43-50.
17. Heneweer M, Muusse M, van den Berg M, et al. Additive estrogenic effects of mixtures of frequently used UV filters on pS2-gene transcription in MCF-7 cells. Toxicol Appl Pharmacol. 2005;208:170-177.
18. Knobler E, Almeida L, Ruzkowski AM, et al. Photoallergy to benzophenone. Arch Dermatol. 1989;125:801-804.
19. Draelos ZD. Are sunscreens safe? J Cosmet Dermatol. 2010;9:1-2.
20. Gilbert E, Pirot F, Bertholle V. Commonly used UV filter toxicity on biological functions: review of last decade studies. Int J of Cosmet Sci. 2013;35:208-219.
21. Wang SQ, Burnett ME, Lim HW. Safety of oxybenzone: putting numbers into perspective. Arch Dermatol. 2011;147:865-866.
22. Mancebo SE, Hu JY, Wang SQ. Sunscreens: a review of health benefits, regulations, and controversies. Dermatol Clin. 2014;32:427-438.
23. Jansen R, Osterwalder U, Wang SQ, et al. Photoprotection: part II. sunscreen: development, efficacy, and controversies. J Am Acad Dermatol. 2013;69:867.e1-867.e14.
24. Janjua NR, Mogensen B, Andersson AM, et al. Systemic absorption of the sunscreens benzo- phenone-3, octyl-methoxycinnamate, and 3-(4-methyl-benzy-lidene) camphor after whole-body topical application and reproductive hormone levels in humans. J Invest Dermatol. 2004;123:57-61.
25. Kadry AM, Chukwuemeka SO, Mohamed S, et al. Pharmacokinetics of benzophenone-3 after oral exposure in male rats. J Appl Toxicol. 1995;15:97-102.
26. Gulson B, McCall M, Korsch M, et al. Small amounts of zinc from zinc oxide particles in sunscreens applied outdoors are absorbed through human skin. Toxicol Sci. 2010;118:140-149.
27. Gulson B, Wong H, Korsch M, et al. Comparison of dermal absorption of zinc from different sunscreen formulations and differing UV exposure based on stable isotope tracing. Sci Total Environ. 2012:420:313-318.
28. Benech-Kieffer F, Meuling WJ, Leclerc C, et al. Percutaneous absorption of Mexoryl SX in human volunteers: comparison with in vitro data. Skin Pharmacol Appl Skin Physiol. 2003;16:343-355.
29. Nash JF. Human safety and efficacy of ultraviolet filters and sunscreen products. Dermatol Clin. 2006;24:35-51.
30. Nohynek GJ, Lademann J, Ribaud C, et al. Grey goo on the skin? nanotechnology, cosmetic and sunscreen safety. Crit Rev Toxicol. 2007;37:251-277.
31. Sadrieh N, Wokovich AM, Gopee NV, et al. Lack of significant dermal penetration of titanium dioxide from sunscreen formulations containing nano- and submicron-size TiO2 particles. Toxicol Sci. 2010;115:156-166.
32. International Agency for Research on Cancer. Carbon black, titanium dioxide, and talc. In: IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. Vol 93. Lyon, France: International Agency for Research on Cancer; 2006.
33. Liao CM, Chiang YH, Chio CP. Model-based assessment for human inhalation exposure risk to airborne nano/fine titanium dioxide particles. Sci Total Environ. 2008:15;407:165-177.
34. Adamcakova-Dodd A, Stebounova LV, Kim JS, et al. Toxicity assessment of zinc oxide nanoparticles using sub-acute and sub-chronic murine inhalation models. Part Fibre Toxicol. 2014;11:15.
35. Wamer WG, Yin JJ, Wei RR. Oxidative damage to nucleic acids photosensitized by titanium dioxide. Free Radic Biol Med. 1997;23:851-858.
36. Nakagawa Y, Wakuri S, Sakamoto K, et al. The photogenotoxicity of titanium dioxide particles. Mutat Res. 1997;394:125-132.
37. Dunford R, Salinaro A, Cai L, et al. Chemical oxidation and DNA damage catalysed by inorganic sunscreen ingredients. FEBS Lett. 1997;418:87-90, 99.
38. Hidaka H, Kobayashi H, Koike T, et al. DNA damage photoinduced by cosmetic pigments and sunscreen agents under solar exposure and artificial UV illumination. J Oleo Sci. 2006;55:249-261.
39. Dufour EK, Kumaravel T, Nohynek GJ, et al. Clastogenicity, photo-clastogenicity or pseudo-photo-clastogenicity: genotoxic effects of zinc oxide in the dark, in pre-irradiated or simultaneously irradiated Chinese hamster ovary cells [published online ahead of print June 21, 2006]. Mutat Res. 2006;607:215-224.
40. Opinion of the Scientific Committee on Cosmetic Products and Non-Food Products intended for Consumers concerning titanium dioxide. http://ec.europa.eu/health/archive/ph_risk/committees/sccp/documents/out135_en.pdf. Published October 24, 2000. Accessed December 30, 2014.
41. The Scientific Committee on Cosmetic Products and Non-Food Products intended for Consumers opinion concerning zinc oxide. http://ec.europa.eu/health/archive/ph_risk/committees/sccp/documents/out222_en.pdf. Published June 24-25, 2003. Accessed December 30, 2014.
42. Hackenberg S, Friehs G, Kessler M, et al. Nanosized titanium dioxide particles do not induce DNA damage in human peripheral blood lymphocytes. Environ Mol Mutagen. 2010;52:264-268.
43. Bach-Thomsen M, Wulf HC. Sunbather’s application of sunscreen is probably inadequate to obtain the sun protection factor assigned to the preparation. Photodermatol Photoimmunol Photomed. 1993:9;242-244.
44. Nohynek GJ, Antignac E, Re T, et al. Safety assessment of personal care products/cosmetics and their ingredients. Toxicol Appl Pharmacol. 2010:1;243:239-259.
45. Diffey BL. Sunscreens: use and misuse. In: Giacomoni PU, ed. Sun Protection in Man. Vol 3. Amsterdam, the Netherlands: Elsevier Science BV; 2001:521-534.
46. Heneweer M, Muusse M, Van den BM, et al. Additive estrogenic effects of mixtures of frequently used UV-filters on pS2-gene transcription in MCF-7 cells. Toxicol Appl Pharmacol. 2005;208:170-177.
47. Kunz PY, Galicia HF, Fent K. Comparison of in vitro and in vivo estrogenic activity of UV-filters in fish. Toxicol Sci. 2006;90:349-361.
48. Kortenkamp A, Faust M, Scholze M, et al. Low-level exposure to multiple chemicals: reason for human health concerns? Environ Health Perspect. 2007;115(suppl 1):106-114.
49. Labeling and effectiveness testing: sunscreen drug products for over-the-counter human use—small entity compliance guide. US Food and Drug Administration Web site. http://www.fda.gov/drugs/guidancecomplianceregulatoryinformation/guidances/ucm330694.htm. Published December 2012. Updated May 13, 2014. Accessed December 30, 2014.
50. Schafer-Korting M, Korting HC, Ponce-Poschl E. Liposomal tretinoin for uncomplicated acne vulgaris. Clin Investig. 1994;72:1086-1091.
51. Bronfenbrener R. Simplifying sun safety: a guide to the new FDA sunscreen monograph. Cutis. 2014;93:e17-e19.
1. Skin cancer statistics. Centers for Disease Control and Prevention Web site. http://www.cdc.gov/cancer/skin/statistics/index.htm. Updated September 2, 2014. Accessed December 30, 2014.
2. World Health Organization, International Agency for Research on Cancer. Solar and ultraviolet radiation. In: IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. Vol 55. Lyon, France: International Agency for Research on Cancer; 1992.
3. Green AC, Williams GM, Logan V, et al. Reduced melanoma after regular sunscreen use: randomized trial follow-up. J Clin Oncol. 2011;29:257-263.
4. Darlington S, Williams G, Neale R, et al. A randomized controlled trial to assess sunscreen application and beta carotene supplementation in the prevention of solar keratoses. Arch Dermatol. 2003;139:451-455.
5. Van der Pols JC, Williams GM, Pandeya N, et al. Prolonged prevention of squamous cell carcinoma of the skin by regular sunscreen use. Cancer Epidemiol Biomarkers Prev. 2006;15:2546-2548.
6. Hughes MC, Williams GM, Baker P, et al. Sunscreen and prevention of skin aging: a randomized trial. Ann Intern Med. 2013;158:781-790.
7. Bissonnette R, Nigen S, Bolduc C. Influence of the quantity of sunscreen applied on the ability to protect against ultraviolet-induced polymorphous light eruption. Photodermatol Photoimmunol Photomed. 2012;28:240-243.
8. Cancer trends progress report 2011/2012 update: sun protection. National Cancer Institute Web site. http://progressreport.cancer.gov/doc_detail.asp?pid¡1&did¡2009&chid¡91&coid¡911. Accessed December 30, 2014.
9. Sunscreen Drug Products for Over-the-counter Human Use, 21 CFR §352.10. http://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/cfrsearch.cfm?fr=352.10. Updated September 1, 2014. Accessed December 30, 2014.
10. Burnett ME, Wang SQ. Current sunscreen controversies: a critical review. Photodermatol Photoimmunol Photomed. 2011;27:58-67.
11. Krause M, Klit A, Blomberg Jensen M, et al. Sunscreens: are they beneficial for health? an overview of endocrine disrupting properties of UV-filters. Int J Androl. 2012;35:424-436.
12. Schlumpf M, Cotton B, Conscience M, et al. In vitro and in vivo estrogenicity of UV screens. Environ Health Perspect. 2001;109:239-244.
13. Schlumpf M, Schmid P, Durrer S, et al. Endocrine activity and developmental toxicity of cosmetic UV filters–an update. Toxicol. 2004;205:113-122.
14. Schlumpf M, Kypke K, Vökt C, et al. Endocrine active UV filters: developmental toxicity and exposure through breast milk. Chimia. 2008;62:345-351.
15. Nakagawa Y, Suzuki T. Metabolism of 2-hydroxy-4-methoxybenzophenone in isolated rat hepatocytes and xenoestrogenic effects of its metabolites on MCF-7 human breast cancer cells. Chem Biol Interact. 2002;139:115-128.
16. Ma R, Cotton B, Lichtensteiger W, et al. UV filters with antagonistic action at androgen receptors in the MDA-kb2 cell transcriptional-activation assay. Toxicol Sci. 2003;74:43-50.
17. Heneweer M, Muusse M, van den Berg M, et al. Additive estrogenic effects of mixtures of frequently used UV filters on pS2-gene transcription in MCF-7 cells. Toxicol Appl Pharmacol. 2005;208:170-177.
18. Knobler E, Almeida L, Ruzkowski AM, et al. Photoallergy to benzophenone. Arch Dermatol. 1989;125:801-804.
19. Draelos ZD. Are sunscreens safe? J Cosmet Dermatol. 2010;9:1-2.
20. Gilbert E, Pirot F, Bertholle V. Commonly used UV filter toxicity on biological functions: review of last decade studies. Int J of Cosmet Sci. 2013;35:208-219.
21. Wang SQ, Burnett ME, Lim HW. Safety of oxybenzone: putting numbers into perspective. Arch Dermatol. 2011;147:865-866.
22. Mancebo SE, Hu JY, Wang SQ. Sunscreens: a review of health benefits, regulations, and controversies. Dermatol Clin. 2014;32:427-438.
23. Jansen R, Osterwalder U, Wang SQ, et al. Photoprotection: part II. sunscreen: development, efficacy, and controversies. J Am Acad Dermatol. 2013;69:867.e1-867.e14.
24. Janjua NR, Mogensen B, Andersson AM, et al. Systemic absorption of the sunscreens benzo- phenone-3, octyl-methoxycinnamate, and 3-(4-methyl-benzy-lidene) camphor after whole-body topical application and reproductive hormone levels in humans. J Invest Dermatol. 2004;123:57-61.
25. Kadry AM, Chukwuemeka SO, Mohamed S, et al. Pharmacokinetics of benzophenone-3 after oral exposure in male rats. J Appl Toxicol. 1995;15:97-102.
26. Gulson B, McCall M, Korsch M, et al. Small amounts of zinc from zinc oxide particles in sunscreens applied outdoors are absorbed through human skin. Toxicol Sci. 2010;118:140-149.
27. Gulson B, Wong H, Korsch M, et al. Comparison of dermal absorption of zinc from different sunscreen formulations and differing UV exposure based on stable isotope tracing. Sci Total Environ. 2012:420:313-318.
28. Benech-Kieffer F, Meuling WJ, Leclerc C, et al. Percutaneous absorption of Mexoryl SX in human volunteers: comparison with in vitro data. Skin Pharmacol Appl Skin Physiol. 2003;16:343-355.
29. Nash JF. Human safety and efficacy of ultraviolet filters and sunscreen products. Dermatol Clin. 2006;24:35-51.
30. Nohynek GJ, Lademann J, Ribaud C, et al. Grey goo on the skin? nanotechnology, cosmetic and sunscreen safety. Crit Rev Toxicol. 2007;37:251-277.
31. Sadrieh N, Wokovich AM, Gopee NV, et al. Lack of significant dermal penetration of titanium dioxide from sunscreen formulations containing nano- and submicron-size TiO2 particles. Toxicol Sci. 2010;115:156-166.
32. International Agency for Research on Cancer. Carbon black, titanium dioxide, and talc. In: IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. Vol 93. Lyon, France: International Agency for Research on Cancer; 2006.
33. Liao CM, Chiang YH, Chio CP. Model-based assessment for human inhalation exposure risk to airborne nano/fine titanium dioxide particles. Sci Total Environ. 2008:15;407:165-177.
34. Adamcakova-Dodd A, Stebounova LV, Kim JS, et al. Toxicity assessment of zinc oxide nanoparticles using sub-acute and sub-chronic murine inhalation models. Part Fibre Toxicol. 2014;11:15.
35. Wamer WG, Yin JJ, Wei RR. Oxidative damage to nucleic acids photosensitized by titanium dioxide. Free Radic Biol Med. 1997;23:851-858.
36. Nakagawa Y, Wakuri S, Sakamoto K, et al. The photogenotoxicity of titanium dioxide particles. Mutat Res. 1997;394:125-132.
37. Dunford R, Salinaro A, Cai L, et al. Chemical oxidation and DNA damage catalysed by inorganic sunscreen ingredients. FEBS Lett. 1997;418:87-90, 99.
38. Hidaka H, Kobayashi H, Koike T, et al. DNA damage photoinduced by cosmetic pigments and sunscreen agents under solar exposure and artificial UV illumination. J Oleo Sci. 2006;55:249-261.
39. Dufour EK, Kumaravel T, Nohynek GJ, et al. Clastogenicity, photo-clastogenicity or pseudo-photo-clastogenicity: genotoxic effects of zinc oxide in the dark, in pre-irradiated or simultaneously irradiated Chinese hamster ovary cells [published online ahead of print June 21, 2006]. Mutat Res. 2006;607:215-224.
40. Opinion of the Scientific Committee on Cosmetic Products and Non-Food Products intended for Consumers concerning titanium dioxide. http://ec.europa.eu/health/archive/ph_risk/committees/sccp/documents/out135_en.pdf. Published October 24, 2000. Accessed December 30, 2014.
41. The Scientific Committee on Cosmetic Products and Non-Food Products intended for Consumers opinion concerning zinc oxide. http://ec.europa.eu/health/archive/ph_risk/committees/sccp/documents/out222_en.pdf. Published June 24-25, 2003. Accessed December 30, 2014.
42. Hackenberg S, Friehs G, Kessler M, et al. Nanosized titanium dioxide particles do not induce DNA damage in human peripheral blood lymphocytes. Environ Mol Mutagen. 2010;52:264-268.
43. Bach-Thomsen M, Wulf HC. Sunbather’s application of sunscreen is probably inadequate to obtain the sun protection factor assigned to the preparation. Photodermatol Photoimmunol Photomed. 1993:9;242-244.
44. Nohynek GJ, Antignac E, Re T, et al. Safety assessment of personal care products/cosmetics and their ingredients. Toxicol Appl Pharmacol. 2010:1;243:239-259.
45. Diffey BL. Sunscreens: use and misuse. In: Giacomoni PU, ed. Sun Protection in Man. Vol 3. Amsterdam, the Netherlands: Elsevier Science BV; 2001:521-534.
46. Heneweer M, Muusse M, Van den BM, et al. Additive estrogenic effects of mixtures of frequently used UV-filters on pS2-gene transcription in MCF-7 cells. Toxicol Appl Pharmacol. 2005;208:170-177.
47. Kunz PY, Galicia HF, Fent K. Comparison of in vitro and in vivo estrogenic activity of UV-filters in fish. Toxicol Sci. 2006;90:349-361.
48. Kortenkamp A, Faust M, Scholze M, et al. Low-level exposure to multiple chemicals: reason for human health concerns? Environ Health Perspect. 2007;115(suppl 1):106-114.
49. Labeling and effectiveness testing: sunscreen drug products for over-the-counter human use—small entity compliance guide. US Food and Drug Administration Web site. http://www.fda.gov/drugs/guidancecomplianceregulatoryinformation/guidances/ucm330694.htm. Published December 2012. Updated May 13, 2014. Accessed December 30, 2014.
50. Schafer-Korting M, Korting HC, Ponce-Poschl E. Liposomal tretinoin for uncomplicated acne vulgaris. Clin Investig. 1994;72:1086-1091.
51. Bronfenbrener R. Simplifying sun safety: a guide to the new FDA sunscreen monograph. Cutis. 2014;93:e17-e19.
Should the use of ‘endorse’ be endorsed in writing in psychiatry?
The word “endorse” often appears in the medical literature and is heard in oral presentations; psychiatrists use the term to mean that a person is reporting psychiatric symptoms or problems. However, such usage may be a stylistic catachresis—one that has the potential for misinterpretation or misunderstanding.
Finding ‘endorse’ in the psychiatric literature
We conducted a literature search to identify instances of “endorse” in scholarly articles published in psychiatric journals between January 1, 2012, and November 25, 2013. Table 11-14 shows examples of typical uses of “endorse” in recent publications.
Even when “endorse” is used as a synonym for “report” or “describe,” use of the word in that context can seem out of place. We could not find any rationale in the medical literature for using “endorse” as a synonym for “report” or “describe.”
The definition of “endorse” in Merriam-Webster15 and Oxford Dictionaries16 includes:
• inscribing or signing a legal document, check, or bill
• approving or recommending an idea, product, or candidate.
We believe that using “endorse” in a psychiatric context could create confusion among medical trainees and professionals who are familiar with the correct meanings of the word.
Survey: Some residents use ‘endorse’ in oral presentations
We asked residents in the Department of Psychiatry at Drexel University College of Medicine to respond to a questionnaire regarding their understanding of the use of “endorse.” Their responses are summarized in Table 2.
What is wrong with using ‘endorse’? Except when “endorse” describes a patient formally affixing her (his) signature to a document for the purpose of (1) certification or (2) giving or showing one’s support for a cause, we think that use of the word in psychiatry is not in keeping with its formal, accepted definition. Furthermore, residents’ responses to our survey suggest that there is the danger of causing confusion in using the word“endorse” when “report” or “describe” is meant.
For example, if a patient “endorses” antisocial behavior, is she stating that she feels justified in exhibiting such behavior? Do students who “endorse” drug use approve of drug use? Another example: Youth who “endorse” gang membership have merely confirmed that they belonged to a gang at some time.
The intended meaning of “endorse” in these examples is probably closer to “admit” or “acknowledge.” The patient replies “yes” when the physician asks if she uses drugs or has had behavior problems; she is not necessarily recommending or approving these behaviors.
Usage is shifting. In the past, “complain” was common medical parlance for a patient’s report of symptoms or other health-related problems. In fact, medical, surgical, and psychiatric evaluations still begin with a “chief complaint” section. It’s possible that, because “complaint” might suggest that the patient is whining, the word fell out of favor in the medical lexicon and was replaced in the scholarly literature by the construction “the patient reports….”
Avoid jargon. Employ accurate terminology
We propose that “endorse,” like “complain,” is a cant of psychiatrists. We recommend that, when describing a patient’s statement or report of symptoms or experiences, practitioners should avoid “endorse” and write or say “report,” “express,” “exhibit,” or similar words. Using accurate terminology and avoiding imprecise or misleading jargon is not only linguistically appropriate but also can help avoid misunderstanding and improve patient care.
Acknowledgment
Diana Winters, Academic Publishing Services, Drexel University College of Medicine, provided editorial assistance in preparing the manuscript of this article and offered comment on the use of medical jargon.
Disclosures
The authors report no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Strauss GP, Gold JM. A new perspective on anhedonia in schizophrenia. Am J Psychiatry. 2012;169(4):364-373.
2. Thomas JJ, Weigel TJ, Lawton RK, et al. Cognitive-behavioral treatment of body image disturbance in a congenitally blind patient with anorexia nervosa. Am J Psychiatry. 2012;169(1):16-20.
3. Purcell B, Heisel MJ, Speice J, et al. Family connectedness moderates the association between living alone and suicide ideation in a clinical sample of adults 50 years and older. Am J Geriatr Psychiatry. 2012;20(8):717-723.
4. Dakin EK, Areán P. Patient perspectives on the benefits of psychotherapy for late-life depression. Am J Geriatr Psychiatry. 2013;21(2):155-163.
5. Mezuk B, Lohman M, Dumenci, L, et al. Are depression and frailty overlapping syndromes in mid- and late-life? A latent variable analysis. Am J Geriatr Psychiatry. 2013;21(6):560-569.
6. Boindala NS, Tucker P, Trautman RP. “Culture and psychiatry”: a course for second-year psychiatry residents. Acad Psychiatry. 2013;37(1):46-50.
7. Henry A, Kisicki MD, Varley C. Efficacy and safety of antidepressant drug treatment in children and adolescents. Mol Psychiatry. 2012;17(12):1186-1193.
8. Rodriguez CI, Kegeles LS, Levinson A, et al. A randomized controlled crossover trial of ketamine in obsessive-compulsive disorder (abstract W7). Neuropsychopharmacology. 2012;38:S317.
9. Whelan R, Garavan H. Fractionating the impulsivity construct in adolescence. Neuropsychopharmacology. 2013;38(1):250-251.
10. Rück C, Larsson KJ, Mataix-Cols D. Predictors of medium and long-term outcome following capsulotomy for obsessive-compulsive disorder: one site may not fit all. Eur Neuropsychopharmacol. 2012;22(6):406-414.
11. Mausbach BT, Chattillion EA, Roepke SK, et al. A comparison of psychosocial outcomes in elderly Alzheimer caregivers and noncaregivers. Am J Geriatr Psychiatry. 2013;21(1):5-13.
12. Peavy GM, Salmon DP, Edland SD, et al. Neuropsychiatric features of frontal lobe dysfunction in autopsy-confirmed patients with lewy bodies and “pure” Alzheimer disease. Am J Geriatr Psychiatry. 2013;21(6):509-519.
13. Coid JW, Ullrich S, Keers R, et al. Gang membership, violence, and psychiatric morbidity. Am J Psychiatry. 2013;170(9):985-993.
14. Choi D, Tolova V, Socha E, et al. Substance use and attitudes on professional conduct among medical students: a single-institution study. Acad Psychiatry. 2013;37(3):191-195.
15. Endorse. Merriam-Webster. http://www.merriam-webster. com/dictionary/endorse. Accessed January 14, 2014.
16. Endorse. Oxford Dictionaries. http://www.oxforddictionaries. com/definition/english/endorse. Accessed January 14, 2014.
The word “endorse” often appears in the medical literature and is heard in oral presentations; psychiatrists use the term to mean that a person is reporting psychiatric symptoms or problems. However, such usage may be a stylistic catachresis—one that has the potential for misinterpretation or misunderstanding.
Finding ‘endorse’ in the psychiatric literature
We conducted a literature search to identify instances of “endorse” in scholarly articles published in psychiatric journals between January 1, 2012, and November 25, 2013. Table 11-14 shows examples of typical uses of “endorse” in recent publications.
Even when “endorse” is used as a synonym for “report” or “describe,” use of the word in that context can seem out of place. We could not find any rationale in the medical literature for using “endorse” as a synonym for “report” or “describe.”
The definition of “endorse” in Merriam-Webster15 and Oxford Dictionaries16 includes:
• inscribing or signing a legal document, check, or bill
• approving or recommending an idea, product, or candidate.
We believe that using “endorse” in a psychiatric context could create confusion among medical trainees and professionals who are familiar with the correct meanings of the word.
Survey: Some residents use ‘endorse’ in oral presentations
We asked residents in the Department of Psychiatry at Drexel University College of Medicine to respond to a questionnaire regarding their understanding of the use of “endorse.” Their responses are summarized in Table 2.
What is wrong with using ‘endorse’? Except when “endorse” describes a patient formally affixing her (his) signature to a document for the purpose of (1) certification or (2) giving or showing one’s support for a cause, we think that use of the word in psychiatry is not in keeping with its formal, accepted definition. Furthermore, residents’ responses to our survey suggest that there is the danger of causing confusion in using the word“endorse” when “report” or “describe” is meant.
For example, if a patient “endorses” antisocial behavior, is she stating that she feels justified in exhibiting such behavior? Do students who “endorse” drug use approve of drug use? Another example: Youth who “endorse” gang membership have merely confirmed that they belonged to a gang at some time.
The intended meaning of “endorse” in these examples is probably closer to “admit” or “acknowledge.” The patient replies “yes” when the physician asks if she uses drugs or has had behavior problems; she is not necessarily recommending or approving these behaviors.
Usage is shifting. In the past, “complain” was common medical parlance for a patient’s report of symptoms or other health-related problems. In fact, medical, surgical, and psychiatric evaluations still begin with a “chief complaint” section. It’s possible that, because “complaint” might suggest that the patient is whining, the word fell out of favor in the medical lexicon and was replaced in the scholarly literature by the construction “the patient reports….”
Avoid jargon. Employ accurate terminology
We propose that “endorse,” like “complain,” is a cant of psychiatrists. We recommend that, when describing a patient’s statement or report of symptoms or experiences, practitioners should avoid “endorse” and write or say “report,” “express,” “exhibit,” or similar words. Using accurate terminology and avoiding imprecise or misleading jargon is not only linguistically appropriate but also can help avoid misunderstanding and improve patient care.
Acknowledgment
Diana Winters, Academic Publishing Services, Drexel University College of Medicine, provided editorial assistance in preparing the manuscript of this article and offered comment on the use of medical jargon.
Disclosures
The authors report no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
The word “endorse” often appears in the medical literature and is heard in oral presentations; psychiatrists use the term to mean that a person is reporting psychiatric symptoms or problems. However, such usage may be a stylistic catachresis—one that has the potential for misinterpretation or misunderstanding.
Finding ‘endorse’ in the psychiatric literature
We conducted a literature search to identify instances of “endorse” in scholarly articles published in psychiatric journals between January 1, 2012, and November 25, 2013. Table 11-14 shows examples of typical uses of “endorse” in recent publications.
Even when “endorse” is used as a synonym for “report” or “describe,” use of the word in that context can seem out of place. We could not find any rationale in the medical literature for using “endorse” as a synonym for “report” or “describe.”
The definition of “endorse” in Merriam-Webster15 and Oxford Dictionaries16 includes:
• inscribing or signing a legal document, check, or bill
• approving or recommending an idea, product, or candidate.
We believe that using “endorse” in a psychiatric context could create confusion among medical trainees and professionals who are familiar with the correct meanings of the word.
Survey: Some residents use ‘endorse’ in oral presentations
We asked residents in the Department of Psychiatry at Drexel University College of Medicine to respond to a questionnaire regarding their understanding of the use of “endorse.” Their responses are summarized in Table 2.
What is wrong with using ‘endorse’? Except when “endorse” describes a patient formally affixing her (his) signature to a document for the purpose of (1) certification or (2) giving or showing one’s support for a cause, we think that use of the word in psychiatry is not in keeping with its formal, accepted definition. Furthermore, residents’ responses to our survey suggest that there is the danger of causing confusion in using the word“endorse” when “report” or “describe” is meant.
For example, if a patient “endorses” antisocial behavior, is she stating that she feels justified in exhibiting such behavior? Do students who “endorse” drug use approve of drug use? Another example: Youth who “endorse” gang membership have merely confirmed that they belonged to a gang at some time.
The intended meaning of “endorse” in these examples is probably closer to “admit” or “acknowledge.” The patient replies “yes” when the physician asks if she uses drugs or has had behavior problems; she is not necessarily recommending or approving these behaviors.
Usage is shifting. In the past, “complain” was common medical parlance for a patient’s report of symptoms or other health-related problems. In fact, medical, surgical, and psychiatric evaluations still begin with a “chief complaint” section. It’s possible that, because “complaint” might suggest that the patient is whining, the word fell out of favor in the medical lexicon and was replaced in the scholarly literature by the construction “the patient reports….”
Avoid jargon. Employ accurate terminology
We propose that “endorse,” like “complain,” is a cant of psychiatrists. We recommend that, when describing a patient’s statement or report of symptoms or experiences, practitioners should avoid “endorse” and write or say “report,” “express,” “exhibit,” or similar words. Using accurate terminology and avoiding imprecise or misleading jargon is not only linguistically appropriate but also can help avoid misunderstanding and improve patient care.
Acknowledgment
Diana Winters, Academic Publishing Services, Drexel University College of Medicine, provided editorial assistance in preparing the manuscript of this article and offered comment on the use of medical jargon.
Disclosures
The authors report no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Strauss GP, Gold JM. A new perspective on anhedonia in schizophrenia. Am J Psychiatry. 2012;169(4):364-373.
2. Thomas JJ, Weigel TJ, Lawton RK, et al. Cognitive-behavioral treatment of body image disturbance in a congenitally blind patient with anorexia nervosa. Am J Psychiatry. 2012;169(1):16-20.
3. Purcell B, Heisel MJ, Speice J, et al. Family connectedness moderates the association between living alone and suicide ideation in a clinical sample of adults 50 years and older. Am J Geriatr Psychiatry. 2012;20(8):717-723.
4. Dakin EK, Areán P. Patient perspectives on the benefits of psychotherapy for late-life depression. Am J Geriatr Psychiatry. 2013;21(2):155-163.
5. Mezuk B, Lohman M, Dumenci, L, et al. Are depression and frailty overlapping syndromes in mid- and late-life? A latent variable analysis. Am J Geriatr Psychiatry. 2013;21(6):560-569.
6. Boindala NS, Tucker P, Trautman RP. “Culture and psychiatry”: a course for second-year psychiatry residents. Acad Psychiatry. 2013;37(1):46-50.
7. Henry A, Kisicki MD, Varley C. Efficacy and safety of antidepressant drug treatment in children and adolescents. Mol Psychiatry. 2012;17(12):1186-1193.
8. Rodriguez CI, Kegeles LS, Levinson A, et al. A randomized controlled crossover trial of ketamine in obsessive-compulsive disorder (abstract W7). Neuropsychopharmacology. 2012;38:S317.
9. Whelan R, Garavan H. Fractionating the impulsivity construct in adolescence. Neuropsychopharmacology. 2013;38(1):250-251.
10. Rück C, Larsson KJ, Mataix-Cols D. Predictors of medium and long-term outcome following capsulotomy for obsessive-compulsive disorder: one site may not fit all. Eur Neuropsychopharmacol. 2012;22(6):406-414.
11. Mausbach BT, Chattillion EA, Roepke SK, et al. A comparison of psychosocial outcomes in elderly Alzheimer caregivers and noncaregivers. Am J Geriatr Psychiatry. 2013;21(1):5-13.
12. Peavy GM, Salmon DP, Edland SD, et al. Neuropsychiatric features of frontal lobe dysfunction in autopsy-confirmed patients with lewy bodies and “pure” Alzheimer disease. Am J Geriatr Psychiatry. 2013;21(6):509-519.
13. Coid JW, Ullrich S, Keers R, et al. Gang membership, violence, and psychiatric morbidity. Am J Psychiatry. 2013;170(9):985-993.
14. Choi D, Tolova V, Socha E, et al. Substance use and attitudes on professional conduct among medical students: a single-institution study. Acad Psychiatry. 2013;37(3):191-195.
15. Endorse. Merriam-Webster. http://www.merriam-webster. com/dictionary/endorse. Accessed January 14, 2014.
16. Endorse. Oxford Dictionaries. http://www.oxforddictionaries. com/definition/english/endorse. Accessed January 14, 2014.
1. Strauss GP, Gold JM. A new perspective on anhedonia in schizophrenia. Am J Psychiatry. 2012;169(4):364-373.
2. Thomas JJ, Weigel TJ, Lawton RK, et al. Cognitive-behavioral treatment of body image disturbance in a congenitally blind patient with anorexia nervosa. Am J Psychiatry. 2012;169(1):16-20.
3. Purcell B, Heisel MJ, Speice J, et al. Family connectedness moderates the association between living alone and suicide ideation in a clinical sample of adults 50 years and older. Am J Geriatr Psychiatry. 2012;20(8):717-723.
4. Dakin EK, Areán P. Patient perspectives on the benefits of psychotherapy for late-life depression. Am J Geriatr Psychiatry. 2013;21(2):155-163.
5. Mezuk B, Lohman M, Dumenci, L, et al. Are depression and frailty overlapping syndromes in mid- and late-life? A latent variable analysis. Am J Geriatr Psychiatry. 2013;21(6):560-569.
6. Boindala NS, Tucker P, Trautman RP. “Culture and psychiatry”: a course for second-year psychiatry residents. Acad Psychiatry. 2013;37(1):46-50.
7. Henry A, Kisicki MD, Varley C. Efficacy and safety of antidepressant drug treatment in children and adolescents. Mol Psychiatry. 2012;17(12):1186-1193.
8. Rodriguez CI, Kegeles LS, Levinson A, et al. A randomized controlled crossover trial of ketamine in obsessive-compulsive disorder (abstract W7). Neuropsychopharmacology. 2012;38:S317.
9. Whelan R, Garavan H. Fractionating the impulsivity construct in adolescence. Neuropsychopharmacology. 2013;38(1):250-251.
10. Rück C, Larsson KJ, Mataix-Cols D. Predictors of medium and long-term outcome following capsulotomy for obsessive-compulsive disorder: one site may not fit all. Eur Neuropsychopharmacol. 2012;22(6):406-414.
11. Mausbach BT, Chattillion EA, Roepke SK, et al. A comparison of psychosocial outcomes in elderly Alzheimer caregivers and noncaregivers. Am J Geriatr Psychiatry. 2013;21(1):5-13.
12. Peavy GM, Salmon DP, Edland SD, et al. Neuropsychiatric features of frontal lobe dysfunction in autopsy-confirmed patients with lewy bodies and “pure” Alzheimer disease. Am J Geriatr Psychiatry. 2013;21(6):509-519.
13. Coid JW, Ullrich S, Keers R, et al. Gang membership, violence, and psychiatric morbidity. Am J Psychiatry. 2013;170(9):985-993.
14. Choi D, Tolova V, Socha E, et al. Substance use and attitudes on professional conduct among medical students: a single-institution study. Acad Psychiatry. 2013;37(3):191-195.
15. Endorse. Merriam-Webster. http://www.merriam-webster. com/dictionary/endorse. Accessed January 14, 2014.
16. Endorse. Oxford Dictionaries. http://www.oxforddictionaries. com/definition/english/endorse. Accessed January 14, 2014.
The Epidemic of Tommy John Surgery: The Role of the Orthopedic Surgeon
Ulnar collateral ligament (UCL) reconstruction, commonly referred to as Tommy John surgery, is a well-described surgical treatment for elite athletes with a symptomatic, deficient UCL.1, 2 The procedure was first performed by the late Dr. Frank Jobe in 1974, described in 1986, and has undergone several modifications over the past 30 years.3 Different graft choices, tunnel positions, graft configurations, and tunnel fixation methods are just some of the alterations that have been made to the original Jobe technique.4-6 With time, the index procedure has become more refined, with predictable outcomes in Major League Baseball (MLB) pitchers as well as other elite overhead throwing athletes.2,7,8 However, though this surgery was originally described for elite athletes suffering from UCL deficiency, recent times have seen an increase of over 50% in the number of UCL reconstructions performed on high school–aged and younger athletes.9 Furthermore, in 2000, a total of 13 MLB pitchers underwent UCL reconstruction, while in 2012 this number increased nearly threefold to 32.2 This paradigm shift of performing UCL reconstructions more frequently and on younger athletes raises a very important question: what is the role of the orthopedic surgeon in this epidemic?
UCL reconstruction has become a reliable procedure for MLB pitchers and other overhead throwing athletes.7,10,11 Recent studies have reported that MLB pitchers who undergo UCL reconstruction return to pitch in the MLB 83% of the time, whereas only 3% fail to return to pitch in either MLB or the minor league.2 Furthermore, pitchers who undergo UCL reconstruction perform similarly after surgery as prior to their UCL reconstruction, with fewer innings pitched after surgery, but, more importantly, a lower earned run average (ERA) and walks plus hits per inning pitched (WHIP) after surgery. These last 2 statistics, known as sabermetrics, evaluate the pitcher’s effectiveness; the fact that these are improved after surgery is reassuring for pitchers who undergo this procedure. However, it must be recognized that these pitchers pitched fewer innings after surgery.
There has been a sharp increase in the number of MLB pitchers who have undergone UCL reconstruction in recent years, especially the past 3 seasons, in which over 60 pitchers have had Tommy John surgery.2 This increase, however, is not confined to MLB pitchers. High school–aged pitchers have also been part of this drastic rise in the number of UCL reconstructions performed throughout the country. Dr. James Andrews and colleagues noted a 50% increase from 1988-1994 to 1995-2003 in the proportion of high school–aged pitchers who underwent UCL reconstruction (while the absolute number increased from 7 to 77 in high school–aged players compared with 85 to 609 in adult athletes).9 Given the increase in MLB pitchers over the past few years, it is likely this number has also increased among adolescent pitchers.
This data again raises the question: what is the role of the orthopedic surgeon in this epidemic? There are many plausible responses, but in my opinion, there is one answer that surpasses the others. As a trained professional, surgeons are tasked with the responsibility of looking out for the best interest of their patients, even when this conflicts with the patient’s, or the patient’s parent’s or coach’s desires. This includes injury prevention, such as instituting pitch counts and developing products that allow coaches to determine when a pitcher may be at risk for injury from fatigue, as well as injury treatment.12 It is difficult for a patient to understand the gravity of surgery and the rehabilitation process, specifically a procedure as involved as UCL reconstruction, and especially if the patient is an adolescent who has their outlook clouded by the fact that they believe they will be the next MLB star pitcher. The reality is that the National Collegiate Athletic Association (NCAA)13 has released data that has demonstrated that only 6.8% of high school baseball players will play baseball in college. Furthermore, only 9.4% of college baseball players will reach the professional level. That equates to 0.5%, or 1 in 200 high school players who will eventually play professional baseball.13 However, the reverse of this is also true, that out of every 200 players, 1 will make it to the major leagues, and that 1 player could be the patient in question. Hence, the purpose of this data is to show parents and athletes that, while they do have a chance of playing professional, and certainly collegiate, baseball, that percentage must be weighed against the risks of surgery.
MLB pitchers who have an endless supply of rehabilitation facilities, trainers, etc, do not return to pitching competitively and consistently in the majors for more than 15 months after UCL reconstruction.2 The time commitment and rehabilitation required for these patients is staggering.14,15 Furthermore, parents of these children who are consenting for them also have a difficult time comprehending the workload they are signing their child up for. Some parents believe this surgery will help their child throw faster, longer, and more accurately—beliefs that numerous studies have shown to be flat-out inaccurate. In fact, pitchers tend to lose a slight amount of velocity and accuracy after UCL reconstruction.11,16 Ahmad and colleagues17 administered a questionnaire to 189 players, 15 coaches, and 31 parents about the indications, risks, benefits, etc, regarding UCL reconstruction to determine the public’s perception regarding this surgery. The results demonstrated that the public, including coaches, have a significantly skewed perception of exactly how serious this surgery is. The study showed that 28% of players and 20% of coaches believed the pitcher’s performance would be improved after surgery, and, more strikingly, 26% of collegiate athletes, 30% percent of coaches, 37% of parents, and 51% of high school athletes believed UCL reconstruction should be performed as a prophylactic procedure to enhance performance in an uninjured athlete.17
Henceforth, it becomes the surgeon’s responsibility to ensure that both the patient and the parents understand what the surgery and rehabilitation process entails, to keep the expectations of the patient and his or her family realistic, and to counsel these patients on alternative options with lower risks. As Ahmad and colleagues17 demonstrated, this is not an easy task given the public’s preconceived notions. Many patients, especially patients of the younger generation, seem to be willing to jump to surgery as the first option for treatment without having truly tried any nonoperative measures, because they believe surgery to be a quick, easy, and definitive answer. This is not always the case, and a trial of nonoperative treatment, including rest, ice, physical therapy, and possibly platelet-rich plasma (PRP), should be instituted for high school–aged players who present with UCL insufficiency prior to discussing surgery.18,19
Medial UCL reconstruction is a successful procedure for elite MLB athletes. However, UCL reconstruction is becoming a victim of its own success as younger and younger athletes who will likely never play at the major league level are undergoing this procedure at an alarming rate. This is an epidemic which must be addressed by surgeons, coaches, and parents alike to curb the beliefs that UCL reconstruction will make high school–aged pitchers more successful. This procedure should not be performed prophylactically on an athlete of any age, especially those in high school. Further studies on the effectiveness of both nonoperative rest and rehabilitation and of PRP on partial-thickness UCL tears are warranted. New technology in the form of a compression sleeve with imbedded sensors to track the biomechanics of a pitcher’s elbow has been released and will hopefully provide information to coaches about when pitchers’ elbows begin to fatigue based on several biomechanical parameters.12 The future of UCL reconstruction is still fluid, and with proper prevention strategies, nonoperative treatment, indications, and preoperative discussions, the Tommy John epidemic can be cured. ◾
1. Conway JE, Jobe FW, Glousman RE, Pink M. Medial instability of the elbow in throwing athletes. Treatment by repair or reconstruction of the ulnar collateral ligament. J Bone Joint Surg Am. 1992;74(1):67-83.
2. Erickson BJ, Gupta AK, Harris JD, et al. Rate of return to pitching and performance after Tommy John surgery in Major League Baseball pitchers. Am J Sports Med. 2014;42(3):536-543.
3. Jobe FW, Stark H, Lombardo SJ. Reconstruction of the ulnar collateral ligament in athletes. J Bone Joint Surg Am. 1986;68(8):1158-1163.
4. Jackson TJ, Adamson GJ, Peterson A, Patton J, McGarry MH, Lee TQ. Ulnar collateral ligament reconstruction using bisuspensory fixation: a biomechanical comparison with the docking technique. Am J Sports Med. 2013;41(5):1158-1164.
5. Dines JS, ElAttrache NS, Conway JE, Smith W, Ahmad CS. Clinical outcomes of the DANE TJ technique to treat ulnar collateral ligament insufficiency of the elbow. Am J Sports Med. 2007;35(12):2039-2044.
6. Andrews JR, Jost PW, Cain EL. The ulnar collateral ligament procedure revisited: the procedure we use. Sports Health. 2012;4(5):438-441.
7. Dines JS, Jones KJ, Kahlenberg C, Rosenbaum A, Osbahr DC, Altchek DW. Elbow ulnar collateral ligament reconstruction in javelin throwers at a minimum 2-year follow-up. Am J Sports Med. 2012;40(1):148-151.
8. Gibson BW, Webner D, Huffman GR, Sennett BJ. Ulnar collateral ligament reconstruction in major league baseball pitchers. Am J Sports Med. 2007;35(4):575-581.
9. Petty DH, Andrews JR, Fleisig GS, Cain EL. Ulnar collateral ligament reconstruction in high school baseball players: clinical results and injury risk factors. Am J Sports Med. 2004;32(5):1158-1164.
10. Osbahr DC, Cain EL Jr, Raines BT, Fortenbaugh D, Dugas JR, Andrews JR. Long-term outcomes after ulnar collateral ligament reconstruction in competitive baseball players: minimum 10-year follow-up. Am J Sports Med. 2014;42(6):1333-1342.
11. Jiang JJ, Leland JM. Analysis of pitching velocity in major league baseball players before and after ulnar collateral ligament reconstruction. Am J Sports Med. 2014;42(4):880-885.
12. Carroll W. The sleeve that could save baseball: exclusive look at new MLB technology. Bleacher Report. http://bleacherreport.com/articles/2097866-the-sleeve-that-could-save-baseball-exclusive-look-at-new-mlb-technology?utm_campaign=tsipad&utm_medium=referral&utm_source=teamstream. Published July 2, 2014. Accessed November 12, 2014.
13. National Collegiate Athletic Association. Estimated probability of competing in athletics beyond the high school interscholastic level. https://www.ncaa.org/sites/default/files/Probability-of-going-pro-methodology_Update2013.pdf. Updated September 24, 2013. Accessed November 12, 2014.
14. Wilk KE, Macrina LC, Cain EL, Dugas JR, Andrews JR. Rehabilitation of the overhead athlete’s elbow. Sports Health. 2012;4(5):404-414.
15. Wilk KE, Reinold MM, Andrews JR. Rehabilitation of the thrower’s elbow. Tech Hand Up Extrem Surg. 2003;7(4):197-216.
16. Makhni EC, Lee RW, Morrow ZS, Gualtieri AP, Gorroochurn P, Ahmad CS. Performance, return to competition, and reinjury after Tommy John surgery in Major League Baseball pitchers: a review of 147 cases. Am J Sports Med. 2014;42(6):1323-1332.
17. Ahmad CS, Grantham WJ, Greiwe RM. Public perceptions of Tommy John surgery. Phys Sportsmed. 2012;40(2):64-72.
18. Rettig AC, Sherrill C, Snead DS, Mendler JC, Mieling P. Nonoperative treatment of ulnar collateral ligament injuries in throwing athletes. Am J Sports Med. 2001;29(1):15-17.
19. Podesta L, Crow SA, Volkmer D, Bert T, Yocum LA. Treatment of partial ulnar collateral ligament tears in the elbow with platelet-rich plasma. Am J Sports Med. 2013;41(7):1689-1694.
Ulnar collateral ligament (UCL) reconstruction, commonly referred to as Tommy John surgery, is a well-described surgical treatment for elite athletes with a symptomatic, deficient UCL.1, 2 The procedure was first performed by the late Dr. Frank Jobe in 1974, described in 1986, and has undergone several modifications over the past 30 years.3 Different graft choices, tunnel positions, graft configurations, and tunnel fixation methods are just some of the alterations that have been made to the original Jobe technique.4-6 With time, the index procedure has become more refined, with predictable outcomes in Major League Baseball (MLB) pitchers as well as other elite overhead throwing athletes.2,7,8 However, though this surgery was originally described for elite athletes suffering from UCL deficiency, recent times have seen an increase of over 50% in the number of UCL reconstructions performed on high school–aged and younger athletes.9 Furthermore, in 2000, a total of 13 MLB pitchers underwent UCL reconstruction, while in 2012 this number increased nearly threefold to 32.2 This paradigm shift of performing UCL reconstructions more frequently and on younger athletes raises a very important question: what is the role of the orthopedic surgeon in this epidemic?
UCL reconstruction has become a reliable procedure for MLB pitchers and other overhead throwing athletes.7,10,11 Recent studies have reported that MLB pitchers who undergo UCL reconstruction return to pitch in the MLB 83% of the time, whereas only 3% fail to return to pitch in either MLB or the minor league.2 Furthermore, pitchers who undergo UCL reconstruction perform similarly after surgery as prior to their UCL reconstruction, with fewer innings pitched after surgery, but, more importantly, a lower earned run average (ERA) and walks plus hits per inning pitched (WHIP) after surgery. These last 2 statistics, known as sabermetrics, evaluate the pitcher’s effectiveness; the fact that these are improved after surgery is reassuring for pitchers who undergo this procedure. However, it must be recognized that these pitchers pitched fewer innings after surgery.
There has been a sharp increase in the number of MLB pitchers who have undergone UCL reconstruction in recent years, especially the past 3 seasons, in which over 60 pitchers have had Tommy John surgery.2 This increase, however, is not confined to MLB pitchers. High school–aged pitchers have also been part of this drastic rise in the number of UCL reconstructions performed throughout the country. Dr. James Andrews and colleagues noted a 50% increase from 1988-1994 to 1995-2003 in the proportion of high school–aged pitchers who underwent UCL reconstruction (while the absolute number increased from 7 to 77 in high school–aged players compared with 85 to 609 in adult athletes).9 Given the increase in MLB pitchers over the past few years, it is likely this number has also increased among adolescent pitchers.
This data again raises the question: what is the role of the orthopedic surgeon in this epidemic? There are many plausible responses, but in my opinion, there is one answer that surpasses the others. As a trained professional, surgeons are tasked with the responsibility of looking out for the best interest of their patients, even when this conflicts with the patient’s, or the patient’s parent’s or coach’s desires. This includes injury prevention, such as instituting pitch counts and developing products that allow coaches to determine when a pitcher may be at risk for injury from fatigue, as well as injury treatment.12 It is difficult for a patient to understand the gravity of surgery and the rehabilitation process, specifically a procedure as involved as UCL reconstruction, and especially if the patient is an adolescent who has their outlook clouded by the fact that they believe they will be the next MLB star pitcher. The reality is that the National Collegiate Athletic Association (NCAA)13 has released data that has demonstrated that only 6.8% of high school baseball players will play baseball in college. Furthermore, only 9.4% of college baseball players will reach the professional level. That equates to 0.5%, or 1 in 200 high school players who will eventually play professional baseball.13 However, the reverse of this is also true, that out of every 200 players, 1 will make it to the major leagues, and that 1 player could be the patient in question. Hence, the purpose of this data is to show parents and athletes that, while they do have a chance of playing professional, and certainly collegiate, baseball, that percentage must be weighed against the risks of surgery.
MLB pitchers who have an endless supply of rehabilitation facilities, trainers, etc, do not return to pitching competitively and consistently in the majors for more than 15 months after UCL reconstruction.2 The time commitment and rehabilitation required for these patients is staggering.14,15 Furthermore, parents of these children who are consenting for them also have a difficult time comprehending the workload they are signing their child up for. Some parents believe this surgery will help their child throw faster, longer, and more accurately—beliefs that numerous studies have shown to be flat-out inaccurate. In fact, pitchers tend to lose a slight amount of velocity and accuracy after UCL reconstruction.11,16 Ahmad and colleagues17 administered a questionnaire to 189 players, 15 coaches, and 31 parents about the indications, risks, benefits, etc, regarding UCL reconstruction to determine the public’s perception regarding this surgery. The results demonstrated that the public, including coaches, have a significantly skewed perception of exactly how serious this surgery is. The study showed that 28% of players and 20% of coaches believed the pitcher’s performance would be improved after surgery, and, more strikingly, 26% of collegiate athletes, 30% percent of coaches, 37% of parents, and 51% of high school athletes believed UCL reconstruction should be performed as a prophylactic procedure to enhance performance in an uninjured athlete.17
Henceforth, it becomes the surgeon’s responsibility to ensure that both the patient and the parents understand what the surgery and rehabilitation process entails, to keep the expectations of the patient and his or her family realistic, and to counsel these patients on alternative options with lower risks. As Ahmad and colleagues17 demonstrated, this is not an easy task given the public’s preconceived notions. Many patients, especially patients of the younger generation, seem to be willing to jump to surgery as the first option for treatment without having truly tried any nonoperative measures, because they believe surgery to be a quick, easy, and definitive answer. This is not always the case, and a trial of nonoperative treatment, including rest, ice, physical therapy, and possibly platelet-rich plasma (PRP), should be instituted for high school–aged players who present with UCL insufficiency prior to discussing surgery.18,19
Medial UCL reconstruction is a successful procedure for elite MLB athletes. However, UCL reconstruction is becoming a victim of its own success as younger and younger athletes who will likely never play at the major league level are undergoing this procedure at an alarming rate. This is an epidemic which must be addressed by surgeons, coaches, and parents alike to curb the beliefs that UCL reconstruction will make high school–aged pitchers more successful. This procedure should not be performed prophylactically on an athlete of any age, especially those in high school. Further studies on the effectiveness of both nonoperative rest and rehabilitation and of PRP on partial-thickness UCL tears are warranted. New technology in the form of a compression sleeve with imbedded sensors to track the biomechanics of a pitcher’s elbow has been released and will hopefully provide information to coaches about when pitchers’ elbows begin to fatigue based on several biomechanical parameters.12 The future of UCL reconstruction is still fluid, and with proper prevention strategies, nonoperative treatment, indications, and preoperative discussions, the Tommy John epidemic can be cured. ◾
Ulnar collateral ligament (UCL) reconstruction, commonly referred to as Tommy John surgery, is a well-described surgical treatment for elite athletes with a symptomatic, deficient UCL.1, 2 The procedure was first performed by the late Dr. Frank Jobe in 1974, described in 1986, and has undergone several modifications over the past 30 years.3 Different graft choices, tunnel positions, graft configurations, and tunnel fixation methods are just some of the alterations that have been made to the original Jobe technique.4-6 With time, the index procedure has become more refined, with predictable outcomes in Major League Baseball (MLB) pitchers as well as other elite overhead throwing athletes.2,7,8 However, though this surgery was originally described for elite athletes suffering from UCL deficiency, recent times have seen an increase of over 50% in the number of UCL reconstructions performed on high school–aged and younger athletes.9 Furthermore, in 2000, a total of 13 MLB pitchers underwent UCL reconstruction, while in 2012 this number increased nearly threefold to 32.2 This paradigm shift of performing UCL reconstructions more frequently and on younger athletes raises a very important question: what is the role of the orthopedic surgeon in this epidemic?
UCL reconstruction has become a reliable procedure for MLB pitchers and other overhead throwing athletes.7,10,11 Recent studies have reported that MLB pitchers who undergo UCL reconstruction return to pitch in the MLB 83% of the time, whereas only 3% fail to return to pitch in either MLB or the minor league.2 Furthermore, pitchers who undergo UCL reconstruction perform similarly after surgery as prior to their UCL reconstruction, with fewer innings pitched after surgery, but, more importantly, a lower earned run average (ERA) and walks plus hits per inning pitched (WHIP) after surgery. These last 2 statistics, known as sabermetrics, evaluate the pitcher’s effectiveness; the fact that these are improved after surgery is reassuring for pitchers who undergo this procedure. However, it must be recognized that these pitchers pitched fewer innings after surgery.
There has been a sharp increase in the number of MLB pitchers who have undergone UCL reconstruction in recent years, especially the past 3 seasons, in which over 60 pitchers have had Tommy John surgery.2 This increase, however, is not confined to MLB pitchers. High school–aged pitchers have also been part of this drastic rise in the number of UCL reconstructions performed throughout the country. Dr. James Andrews and colleagues noted a 50% increase from 1988-1994 to 1995-2003 in the proportion of high school–aged pitchers who underwent UCL reconstruction (while the absolute number increased from 7 to 77 in high school–aged players compared with 85 to 609 in adult athletes).9 Given the increase in MLB pitchers over the past few years, it is likely this number has also increased among adolescent pitchers.
This data again raises the question: what is the role of the orthopedic surgeon in this epidemic? There are many plausible responses, but in my opinion, there is one answer that surpasses the others. As a trained professional, surgeons are tasked with the responsibility of looking out for the best interest of their patients, even when this conflicts with the patient’s, or the patient’s parent’s or coach’s desires. This includes injury prevention, such as instituting pitch counts and developing products that allow coaches to determine when a pitcher may be at risk for injury from fatigue, as well as injury treatment.12 It is difficult for a patient to understand the gravity of surgery and the rehabilitation process, specifically a procedure as involved as UCL reconstruction, and especially if the patient is an adolescent who has their outlook clouded by the fact that they believe they will be the next MLB star pitcher. The reality is that the National Collegiate Athletic Association (NCAA)13 has released data that has demonstrated that only 6.8% of high school baseball players will play baseball in college. Furthermore, only 9.4% of college baseball players will reach the professional level. That equates to 0.5%, or 1 in 200 high school players who will eventually play professional baseball.13 However, the reverse of this is also true, that out of every 200 players, 1 will make it to the major leagues, and that 1 player could be the patient in question. Hence, the purpose of this data is to show parents and athletes that, while they do have a chance of playing professional, and certainly collegiate, baseball, that percentage must be weighed against the risks of surgery.
MLB pitchers who have an endless supply of rehabilitation facilities, trainers, etc, do not return to pitching competitively and consistently in the majors for more than 15 months after UCL reconstruction.2 The time commitment and rehabilitation required for these patients is staggering.14,15 Furthermore, parents of these children who are consenting for them also have a difficult time comprehending the workload they are signing their child up for. Some parents believe this surgery will help their child throw faster, longer, and more accurately—beliefs that numerous studies have shown to be flat-out inaccurate. In fact, pitchers tend to lose a slight amount of velocity and accuracy after UCL reconstruction.11,16 Ahmad and colleagues17 administered a questionnaire to 189 players, 15 coaches, and 31 parents about the indications, risks, benefits, etc, regarding UCL reconstruction to determine the public’s perception regarding this surgery. The results demonstrated that the public, including coaches, have a significantly skewed perception of exactly how serious this surgery is. The study showed that 28% of players and 20% of coaches believed the pitcher’s performance would be improved after surgery, and, more strikingly, 26% of collegiate athletes, 30% percent of coaches, 37% of parents, and 51% of high school athletes believed UCL reconstruction should be performed as a prophylactic procedure to enhance performance in an uninjured athlete.17
Henceforth, it becomes the surgeon’s responsibility to ensure that both the patient and the parents understand what the surgery and rehabilitation process entails, to keep the expectations of the patient and his or her family realistic, and to counsel these patients on alternative options with lower risks. As Ahmad and colleagues17 demonstrated, this is not an easy task given the public’s preconceived notions. Many patients, especially patients of the younger generation, seem to be willing to jump to surgery as the first option for treatment without having truly tried any nonoperative measures, because they believe surgery to be a quick, easy, and definitive answer. This is not always the case, and a trial of nonoperative treatment, including rest, ice, physical therapy, and possibly platelet-rich plasma (PRP), should be instituted for high school–aged players who present with UCL insufficiency prior to discussing surgery.18,19
Medial UCL reconstruction is a successful procedure for elite MLB athletes. However, UCL reconstruction is becoming a victim of its own success as younger and younger athletes who will likely never play at the major league level are undergoing this procedure at an alarming rate. This is an epidemic which must be addressed by surgeons, coaches, and parents alike to curb the beliefs that UCL reconstruction will make high school–aged pitchers more successful. This procedure should not be performed prophylactically on an athlete of any age, especially those in high school. Further studies on the effectiveness of both nonoperative rest and rehabilitation and of PRP on partial-thickness UCL tears are warranted. New technology in the form of a compression sleeve with imbedded sensors to track the biomechanics of a pitcher’s elbow has been released and will hopefully provide information to coaches about when pitchers’ elbows begin to fatigue based on several biomechanical parameters.12 The future of UCL reconstruction is still fluid, and with proper prevention strategies, nonoperative treatment, indications, and preoperative discussions, the Tommy John epidemic can be cured. ◾
1. Conway JE, Jobe FW, Glousman RE, Pink M. Medial instability of the elbow in throwing athletes. Treatment by repair or reconstruction of the ulnar collateral ligament. J Bone Joint Surg Am. 1992;74(1):67-83.
2. Erickson BJ, Gupta AK, Harris JD, et al. Rate of return to pitching and performance after Tommy John surgery in Major League Baseball pitchers. Am J Sports Med. 2014;42(3):536-543.
3. Jobe FW, Stark H, Lombardo SJ. Reconstruction of the ulnar collateral ligament in athletes. J Bone Joint Surg Am. 1986;68(8):1158-1163.
4. Jackson TJ, Adamson GJ, Peterson A, Patton J, McGarry MH, Lee TQ. Ulnar collateral ligament reconstruction using bisuspensory fixation: a biomechanical comparison with the docking technique. Am J Sports Med. 2013;41(5):1158-1164.
5. Dines JS, ElAttrache NS, Conway JE, Smith W, Ahmad CS. Clinical outcomes of the DANE TJ technique to treat ulnar collateral ligament insufficiency of the elbow. Am J Sports Med. 2007;35(12):2039-2044.
6. Andrews JR, Jost PW, Cain EL. The ulnar collateral ligament procedure revisited: the procedure we use. Sports Health. 2012;4(5):438-441.
7. Dines JS, Jones KJ, Kahlenberg C, Rosenbaum A, Osbahr DC, Altchek DW. Elbow ulnar collateral ligament reconstruction in javelin throwers at a minimum 2-year follow-up. Am J Sports Med. 2012;40(1):148-151.
8. Gibson BW, Webner D, Huffman GR, Sennett BJ. Ulnar collateral ligament reconstruction in major league baseball pitchers. Am J Sports Med. 2007;35(4):575-581.
9. Petty DH, Andrews JR, Fleisig GS, Cain EL. Ulnar collateral ligament reconstruction in high school baseball players: clinical results and injury risk factors. Am J Sports Med. 2004;32(5):1158-1164.
10. Osbahr DC, Cain EL Jr, Raines BT, Fortenbaugh D, Dugas JR, Andrews JR. Long-term outcomes after ulnar collateral ligament reconstruction in competitive baseball players: minimum 10-year follow-up. Am J Sports Med. 2014;42(6):1333-1342.
11. Jiang JJ, Leland JM. Analysis of pitching velocity in major league baseball players before and after ulnar collateral ligament reconstruction. Am J Sports Med. 2014;42(4):880-885.
12. Carroll W. The sleeve that could save baseball: exclusive look at new MLB technology. Bleacher Report. http://bleacherreport.com/articles/2097866-the-sleeve-that-could-save-baseball-exclusive-look-at-new-mlb-technology?utm_campaign=tsipad&utm_medium=referral&utm_source=teamstream. Published July 2, 2014. Accessed November 12, 2014.
13. National Collegiate Athletic Association. Estimated probability of competing in athletics beyond the high school interscholastic level. https://www.ncaa.org/sites/default/files/Probability-of-going-pro-methodology_Update2013.pdf. Updated September 24, 2013. Accessed November 12, 2014.
14. Wilk KE, Macrina LC, Cain EL, Dugas JR, Andrews JR. Rehabilitation of the overhead athlete’s elbow. Sports Health. 2012;4(5):404-414.
15. Wilk KE, Reinold MM, Andrews JR. Rehabilitation of the thrower’s elbow. Tech Hand Up Extrem Surg. 2003;7(4):197-216.
16. Makhni EC, Lee RW, Morrow ZS, Gualtieri AP, Gorroochurn P, Ahmad CS. Performance, return to competition, and reinjury after Tommy John surgery in Major League Baseball pitchers: a review of 147 cases. Am J Sports Med. 2014;42(6):1323-1332.
17. Ahmad CS, Grantham WJ, Greiwe RM. Public perceptions of Tommy John surgery. Phys Sportsmed. 2012;40(2):64-72.
18. Rettig AC, Sherrill C, Snead DS, Mendler JC, Mieling P. Nonoperative treatment of ulnar collateral ligament injuries in throwing athletes. Am J Sports Med. 2001;29(1):15-17.
19. Podesta L, Crow SA, Volkmer D, Bert T, Yocum LA. Treatment of partial ulnar collateral ligament tears in the elbow with platelet-rich plasma. Am J Sports Med. 2013;41(7):1689-1694.
1. Conway JE, Jobe FW, Glousman RE, Pink M. Medial instability of the elbow in throwing athletes. Treatment by repair or reconstruction of the ulnar collateral ligament. J Bone Joint Surg Am. 1992;74(1):67-83.
2. Erickson BJ, Gupta AK, Harris JD, et al. Rate of return to pitching and performance after Tommy John surgery in Major League Baseball pitchers. Am J Sports Med. 2014;42(3):536-543.
3. Jobe FW, Stark H, Lombardo SJ. Reconstruction of the ulnar collateral ligament in athletes. J Bone Joint Surg Am. 1986;68(8):1158-1163.
4. Jackson TJ, Adamson GJ, Peterson A, Patton J, McGarry MH, Lee TQ. Ulnar collateral ligament reconstruction using bisuspensory fixation: a biomechanical comparison with the docking technique. Am J Sports Med. 2013;41(5):1158-1164.
5. Dines JS, ElAttrache NS, Conway JE, Smith W, Ahmad CS. Clinical outcomes of the DANE TJ technique to treat ulnar collateral ligament insufficiency of the elbow. Am J Sports Med. 2007;35(12):2039-2044.
6. Andrews JR, Jost PW, Cain EL. The ulnar collateral ligament procedure revisited: the procedure we use. Sports Health. 2012;4(5):438-441.
7. Dines JS, Jones KJ, Kahlenberg C, Rosenbaum A, Osbahr DC, Altchek DW. Elbow ulnar collateral ligament reconstruction in javelin throwers at a minimum 2-year follow-up. Am J Sports Med. 2012;40(1):148-151.
8. Gibson BW, Webner D, Huffman GR, Sennett BJ. Ulnar collateral ligament reconstruction in major league baseball pitchers. Am J Sports Med. 2007;35(4):575-581.
9. Petty DH, Andrews JR, Fleisig GS, Cain EL. Ulnar collateral ligament reconstruction in high school baseball players: clinical results and injury risk factors. Am J Sports Med. 2004;32(5):1158-1164.
10. Osbahr DC, Cain EL Jr, Raines BT, Fortenbaugh D, Dugas JR, Andrews JR. Long-term outcomes after ulnar collateral ligament reconstruction in competitive baseball players: minimum 10-year follow-up. Am J Sports Med. 2014;42(6):1333-1342.
11. Jiang JJ, Leland JM. Analysis of pitching velocity in major league baseball players before and after ulnar collateral ligament reconstruction. Am J Sports Med. 2014;42(4):880-885.
12. Carroll W. The sleeve that could save baseball: exclusive look at new MLB technology. Bleacher Report. http://bleacherreport.com/articles/2097866-the-sleeve-that-could-save-baseball-exclusive-look-at-new-mlb-technology?utm_campaign=tsipad&utm_medium=referral&utm_source=teamstream. Published July 2, 2014. Accessed November 12, 2014.
13. National Collegiate Athletic Association. Estimated probability of competing in athletics beyond the high school interscholastic level. https://www.ncaa.org/sites/default/files/Probability-of-going-pro-methodology_Update2013.pdf. Updated September 24, 2013. Accessed November 12, 2014.
14. Wilk KE, Macrina LC, Cain EL, Dugas JR, Andrews JR. Rehabilitation of the overhead athlete’s elbow. Sports Health. 2012;4(5):404-414.
15. Wilk KE, Reinold MM, Andrews JR. Rehabilitation of the thrower’s elbow. Tech Hand Up Extrem Surg. 2003;7(4):197-216.
16. Makhni EC, Lee RW, Morrow ZS, Gualtieri AP, Gorroochurn P, Ahmad CS. Performance, return to competition, and reinjury after Tommy John surgery in Major League Baseball pitchers: a review of 147 cases. Am J Sports Med. 2014;42(6):1323-1332.
17. Ahmad CS, Grantham WJ, Greiwe RM. Public perceptions of Tommy John surgery. Phys Sportsmed. 2012;40(2):64-72.
18. Rettig AC, Sherrill C, Snead DS, Mendler JC, Mieling P. Nonoperative treatment of ulnar collateral ligament injuries in throwing athletes. Am J Sports Med. 2001;29(1):15-17.
19. Podesta L, Crow SA, Volkmer D, Bert T, Yocum LA. Treatment of partial ulnar collateral ligament tears in the elbow with platelet-rich plasma. Am J Sports Med. 2013;41(7):1689-1694.
Stains and Smears: Resident Guide to Bedside Diagnostic Testing
Dermatologists are fortunate to specialize in the organ that is most accessible to evaluation. Although we use the physical examination to formulate the initial differential diagnosis, at times we must rely on ancillary tests to narrow down the diagnosis. Various bedside testing modalities—potassium hydroxide (KOH) preparation, Tzanck smear, mineral oil preparation, and Gram stain—are most useful in diagnosing infectious causes of cutaneous disease. This guide serves as a useful reference for residents on how to perform these tests and which conditions they can help diagnose. Several of these procedures have no standard protocol for performing them; the literature is littered with various methodologies, fixatives, and stains, and as such, this article will attempt to describe a technique that is convenient and quick to perform with readily available materials while still offering high diagnostic utility.
KOH Preparation
A standard in the armamentarium of a dermatologist, the KOH preparation is invaluable to diagnose fungal and yeast infections. Although there are many available preparations including varying concentrations of KOH, dimethyl sulfoxide, and various inks, the procedure is similar for all of them.1 The first step involves collecting the specimen, which can be scale from an active border of suspected cutaneous dermatophyte or Malassezia infection, debris from suspected candidiasis, or hair shafts plucked from an area of alopecia of presumed tinea capitis. A no. 15 blade can be used to scrape the specimen onto a microscope slide, though a second microscope slide can be used in lieu of a blade in patients who will not remain still, and then a coverslip is placed. Two drops of the KOH solution of your choice are then placed on opposite ends of the coverslip, allowing capillary action to spread the stain evenly. A paper towel can be folded in half and pushed down on the surface of the coverslip to spread the stain and soak up any excess, and this pressure also can help the KOH solution digest the keratin in the specimen. Briefly heating the underside of the slide (below boiling point) will help digest the keratin; this step is not necessary when you are using a KOH preparation with dimethyl sulfoxide. Although many dermatologists view the slide almost immediately, ideally at least 5 minutes should pass before it is read. Particularly thick specimens may require additional digestion time, so setting them aside for later review may help visualize infectious agents. In a busy clinic where an immediate diagnosis may not be requisite and a prescription can be called in pending the result, waiting to review the slide may be feasible.
Tzanck Smear
The Tzanck smear is a useful cytopathologic test in the rapid diagnosis of herpetic lesions, though it cannot differentiate between herpes simplex virus type 1, herpes simplex virus type 2, and varicella-zoster virus. It also has shown utility for rapid diagnosis of protean other dermatologic conditions including autoimmune blistering disorders, cutaneous malignancies, and other infectious processes, though it has been superseded by histopathology in most cases.2 An ideal sample is collected by scraping the base of a fresh blister with a no. 15 blade or a second microscope slide. The scrapings then are smeared onto another microscope slide and allowed to air-dry briefly. Then, Wright-Giemsa stain is dispensed to cover the sample and allowed to sit for 15 minutes before being washed off with sterile water. After air-drying, the sample is examined for the presence of clumped multinucleated giant cells, a feature that confirms herpetic infection and allows rapid initiation of antiviral medication.3
Mineral Oil Preparation
A mineral oil preparation has utility in diagnosing ectoparasitic infestation. In the case of scabies, a positive microscopic examination is diagnostic and requires no further testing, allowing for rapid initiation of therapy. This technique also is useful in diagnosing rosacea related to Demodex, which requires a treatment algorithm that differs from the classic papulopustular rosacea which it mimics.4
Mineral oil preparations can be rapidly performed and interpreted. Several drops of mineral oil are placed onto a microscope slide and a no. 15 blade is dipped into this oil prior to scraping the sample lesion. For scabies, a burrow is scraped repeatedly with the blade, and the debris is collected in the mineral oil. Occasionally, the mite can be dermoscopically visualized as a jet plane or arrowhead at the leading edge of a burrow; scraping should be focused in the vicinity of the mite.5 A coverslip is applied to the microscope slide and examination for the mite, egg casings, and scybala can be performed with microscopy.6 For Demodex infestation, a facial pustule can be expressed or several eyelash hairs can be plucked and suspended in mineral oil. Examination of this specimen is identical to scabies.
Gram Stain
The Gram stain is invaluable in classifying bacteria, and a properly performed test can narrow the identification of a causative organism based on cellular morphology. Although it is more technically complex than other bedside diagnostic maneuvers, it can be rapidly performed once the sequence of stains is mastered. The collected sample is smeared onto a glass slide and then briefly passed over a flame several times to heat-fix the specimen. Caution should be taken to avoid direct or prolonged flame contact with the underside of the slide. After fixation, the staining can be performed. First, crystal violet is instilled onto the slide and remains on for 30 seconds before being rinsed off with sink water. Then, Gram iodine is used for 30 seconds, followed by another rinse in water. Next, pour the decolorizer solution over the slide until the runoff is clear, and then rinse in water. Finally, flood with safranin counterstain for 30 seconds and give the slide a final rinse. After air-drying, it is ready to be interpreted.7
Final Thoughts
Although the modern dermatologist has access to biopsies, cultures, and sophisticated diagnostic techniques, it is important to remember these useful bedside tests. The ability to rapidly pin a diagnosis is particularly useful on the consultative service where critically ill patients can benefit from identification of a causative pathogen sooner rather than later. Residents should master these stains in their training, as this knowledge may prove to be invaluable in their careers.
1. Trozak DJ, Tennenhouse DJ, Russell JJ. Dermatology Skills for Primary Care: An Illustrated Guide. Totowa, NJ: Humana Press; 2006.
2. Kelly B, Shimoni T. Reintroducing the Tzanck smear. Am J Clin Dermatol. 2009;10:141-152.
3. Singhi M, Gupta L. Tzanck smear: a useful diagnostic tool. Indian J Dermatol Venereol Leprol. 2005;71:295.
4. Elston DM. Demodex mites: facts and controversies. Clin Dermatol. 2010;28:502-504.
5. Dupuy A, Dehen L, Bourrat E, et al. Accuracy of standard dermoscopy for diagnosing scabies. J Am Acad Dermatol. 2007;56:53-62.
6. Bolognia J, Schaffer J, Duncan K, et al. Dermatology Essentials. St. Louis, MO: Saunders Elsevier; 2014.
7. Ruocco E, Baroni A, Donnarumma G, et al. Diagnostic procedures in dermatology. Clin Dermatol. 2011;29:548-556.
Dermatologists are fortunate to specialize in the organ that is most accessible to evaluation. Although we use the physical examination to formulate the initial differential diagnosis, at times we must rely on ancillary tests to narrow down the diagnosis. Various bedside testing modalities—potassium hydroxide (KOH) preparation, Tzanck smear, mineral oil preparation, and Gram stain—are most useful in diagnosing infectious causes of cutaneous disease. This guide serves as a useful reference for residents on how to perform these tests and which conditions they can help diagnose. Several of these procedures have no standard protocol for performing them; the literature is littered with various methodologies, fixatives, and stains, and as such, this article will attempt to describe a technique that is convenient and quick to perform with readily available materials while still offering high diagnostic utility.
KOH Preparation
A standard in the armamentarium of a dermatologist, the KOH preparation is invaluable to diagnose fungal and yeast infections. Although there are many available preparations including varying concentrations of KOH, dimethyl sulfoxide, and various inks, the procedure is similar for all of them.1 The first step involves collecting the specimen, which can be scale from an active border of suspected cutaneous dermatophyte or Malassezia infection, debris from suspected candidiasis, or hair shafts plucked from an area of alopecia of presumed tinea capitis. A no. 15 blade can be used to scrape the specimen onto a microscope slide, though a second microscope slide can be used in lieu of a blade in patients who will not remain still, and then a coverslip is placed. Two drops of the KOH solution of your choice are then placed on opposite ends of the coverslip, allowing capillary action to spread the stain evenly. A paper towel can be folded in half and pushed down on the surface of the coverslip to spread the stain and soak up any excess, and this pressure also can help the KOH solution digest the keratin in the specimen. Briefly heating the underside of the slide (below boiling point) will help digest the keratin; this step is not necessary when you are using a KOH preparation with dimethyl sulfoxide. Although many dermatologists view the slide almost immediately, ideally at least 5 minutes should pass before it is read. Particularly thick specimens may require additional digestion time, so setting them aside for later review may help visualize infectious agents. In a busy clinic where an immediate diagnosis may not be requisite and a prescription can be called in pending the result, waiting to review the slide may be feasible.
Tzanck Smear
The Tzanck smear is a useful cytopathologic test in the rapid diagnosis of herpetic lesions, though it cannot differentiate between herpes simplex virus type 1, herpes simplex virus type 2, and varicella-zoster virus. It also has shown utility for rapid diagnosis of protean other dermatologic conditions including autoimmune blistering disorders, cutaneous malignancies, and other infectious processes, though it has been superseded by histopathology in most cases.2 An ideal sample is collected by scraping the base of a fresh blister with a no. 15 blade or a second microscope slide. The scrapings then are smeared onto another microscope slide and allowed to air-dry briefly. Then, Wright-Giemsa stain is dispensed to cover the sample and allowed to sit for 15 minutes before being washed off with sterile water. After air-drying, the sample is examined for the presence of clumped multinucleated giant cells, a feature that confirms herpetic infection and allows rapid initiation of antiviral medication.3
Mineral Oil Preparation
A mineral oil preparation has utility in diagnosing ectoparasitic infestation. In the case of scabies, a positive microscopic examination is diagnostic and requires no further testing, allowing for rapid initiation of therapy. This technique also is useful in diagnosing rosacea related to Demodex, which requires a treatment algorithm that differs from the classic papulopustular rosacea which it mimics.4
Mineral oil preparations can be rapidly performed and interpreted. Several drops of mineral oil are placed onto a microscope slide and a no. 15 blade is dipped into this oil prior to scraping the sample lesion. For scabies, a burrow is scraped repeatedly with the blade, and the debris is collected in the mineral oil. Occasionally, the mite can be dermoscopically visualized as a jet plane or arrowhead at the leading edge of a burrow; scraping should be focused in the vicinity of the mite.5 A coverslip is applied to the microscope slide and examination for the mite, egg casings, and scybala can be performed with microscopy.6 For Demodex infestation, a facial pustule can be expressed or several eyelash hairs can be plucked and suspended in mineral oil. Examination of this specimen is identical to scabies.
Gram Stain
The Gram stain is invaluable in classifying bacteria, and a properly performed test can narrow the identification of a causative organism based on cellular morphology. Although it is more technically complex than other bedside diagnostic maneuvers, it can be rapidly performed once the sequence of stains is mastered. The collected sample is smeared onto a glass slide and then briefly passed over a flame several times to heat-fix the specimen. Caution should be taken to avoid direct or prolonged flame contact with the underside of the slide. After fixation, the staining can be performed. First, crystal violet is instilled onto the slide and remains on for 30 seconds before being rinsed off with sink water. Then, Gram iodine is used for 30 seconds, followed by another rinse in water. Next, pour the decolorizer solution over the slide until the runoff is clear, and then rinse in water. Finally, flood with safranin counterstain for 30 seconds and give the slide a final rinse. After air-drying, it is ready to be interpreted.7
Final Thoughts
Although the modern dermatologist has access to biopsies, cultures, and sophisticated diagnostic techniques, it is important to remember these useful bedside tests. The ability to rapidly pin a diagnosis is particularly useful on the consultative service where critically ill patients can benefit from identification of a causative pathogen sooner rather than later. Residents should master these stains in their training, as this knowledge may prove to be invaluable in their careers.
Dermatologists are fortunate to specialize in the organ that is most accessible to evaluation. Although we use the physical examination to formulate the initial differential diagnosis, at times we must rely on ancillary tests to narrow down the diagnosis. Various bedside testing modalities—potassium hydroxide (KOH) preparation, Tzanck smear, mineral oil preparation, and Gram stain—are most useful in diagnosing infectious causes of cutaneous disease. This guide serves as a useful reference for residents on how to perform these tests and which conditions they can help diagnose. Several of these procedures have no standard protocol for performing them; the literature is littered with various methodologies, fixatives, and stains, and as such, this article will attempt to describe a technique that is convenient and quick to perform with readily available materials while still offering high diagnostic utility.
KOH Preparation
A standard in the armamentarium of a dermatologist, the KOH preparation is invaluable to diagnose fungal and yeast infections. Although there are many available preparations including varying concentrations of KOH, dimethyl sulfoxide, and various inks, the procedure is similar for all of them.1 The first step involves collecting the specimen, which can be scale from an active border of suspected cutaneous dermatophyte or Malassezia infection, debris from suspected candidiasis, or hair shafts plucked from an area of alopecia of presumed tinea capitis. A no. 15 blade can be used to scrape the specimen onto a microscope slide, though a second microscope slide can be used in lieu of a blade in patients who will not remain still, and then a coverslip is placed. Two drops of the KOH solution of your choice are then placed on opposite ends of the coverslip, allowing capillary action to spread the stain evenly. A paper towel can be folded in half and pushed down on the surface of the coverslip to spread the stain and soak up any excess, and this pressure also can help the KOH solution digest the keratin in the specimen. Briefly heating the underside of the slide (below boiling point) will help digest the keratin; this step is not necessary when you are using a KOH preparation with dimethyl sulfoxide. Although many dermatologists view the slide almost immediately, ideally at least 5 minutes should pass before it is read. Particularly thick specimens may require additional digestion time, so setting them aside for later review may help visualize infectious agents. In a busy clinic where an immediate diagnosis may not be requisite and a prescription can be called in pending the result, waiting to review the slide may be feasible.
Tzanck Smear
The Tzanck smear is a useful cytopathologic test in the rapid diagnosis of herpetic lesions, though it cannot differentiate between herpes simplex virus type 1, herpes simplex virus type 2, and varicella-zoster virus. It also has shown utility for rapid diagnosis of protean other dermatologic conditions including autoimmune blistering disorders, cutaneous malignancies, and other infectious processes, though it has been superseded by histopathology in most cases.2 An ideal sample is collected by scraping the base of a fresh blister with a no. 15 blade or a second microscope slide. The scrapings then are smeared onto another microscope slide and allowed to air-dry briefly. Then, Wright-Giemsa stain is dispensed to cover the sample and allowed to sit for 15 minutes before being washed off with sterile water. After air-drying, the sample is examined for the presence of clumped multinucleated giant cells, a feature that confirms herpetic infection and allows rapid initiation of antiviral medication.3
Mineral Oil Preparation
A mineral oil preparation has utility in diagnosing ectoparasitic infestation. In the case of scabies, a positive microscopic examination is diagnostic and requires no further testing, allowing for rapid initiation of therapy. This technique also is useful in diagnosing rosacea related to Demodex, which requires a treatment algorithm that differs from the classic papulopustular rosacea which it mimics.4
Mineral oil preparations can be rapidly performed and interpreted. Several drops of mineral oil are placed onto a microscope slide and a no. 15 blade is dipped into this oil prior to scraping the sample lesion. For scabies, a burrow is scraped repeatedly with the blade, and the debris is collected in the mineral oil. Occasionally, the mite can be dermoscopically visualized as a jet plane or arrowhead at the leading edge of a burrow; scraping should be focused in the vicinity of the mite.5 A coverslip is applied to the microscope slide and examination for the mite, egg casings, and scybala can be performed with microscopy.6 For Demodex infestation, a facial pustule can be expressed or several eyelash hairs can be plucked and suspended in mineral oil. Examination of this specimen is identical to scabies.
Gram Stain
The Gram stain is invaluable in classifying bacteria, and a properly performed test can narrow the identification of a causative organism based on cellular morphology. Although it is more technically complex than other bedside diagnostic maneuvers, it can be rapidly performed once the sequence of stains is mastered. The collected sample is smeared onto a glass slide and then briefly passed over a flame several times to heat-fix the specimen. Caution should be taken to avoid direct or prolonged flame contact with the underside of the slide. After fixation, the staining can be performed. First, crystal violet is instilled onto the slide and remains on for 30 seconds before being rinsed off with sink water. Then, Gram iodine is used for 30 seconds, followed by another rinse in water. Next, pour the decolorizer solution over the slide until the runoff is clear, and then rinse in water. Finally, flood with safranin counterstain for 30 seconds and give the slide a final rinse. After air-drying, it is ready to be interpreted.7
Final Thoughts
Although the modern dermatologist has access to biopsies, cultures, and sophisticated diagnostic techniques, it is important to remember these useful bedside tests. The ability to rapidly pin a diagnosis is particularly useful on the consultative service where critically ill patients can benefit from identification of a causative pathogen sooner rather than later. Residents should master these stains in their training, as this knowledge may prove to be invaluable in their careers.
1. Trozak DJ, Tennenhouse DJ, Russell JJ. Dermatology Skills for Primary Care: An Illustrated Guide. Totowa, NJ: Humana Press; 2006.
2. Kelly B, Shimoni T. Reintroducing the Tzanck smear. Am J Clin Dermatol. 2009;10:141-152.
3. Singhi M, Gupta L. Tzanck smear: a useful diagnostic tool. Indian J Dermatol Venereol Leprol. 2005;71:295.
4. Elston DM. Demodex mites: facts and controversies. Clin Dermatol. 2010;28:502-504.
5. Dupuy A, Dehen L, Bourrat E, et al. Accuracy of standard dermoscopy for diagnosing scabies. J Am Acad Dermatol. 2007;56:53-62.
6. Bolognia J, Schaffer J, Duncan K, et al. Dermatology Essentials. St. Louis, MO: Saunders Elsevier; 2014.
7. Ruocco E, Baroni A, Donnarumma G, et al. Diagnostic procedures in dermatology. Clin Dermatol. 2011;29:548-556.
1. Trozak DJ, Tennenhouse DJ, Russell JJ. Dermatology Skills for Primary Care: An Illustrated Guide. Totowa, NJ: Humana Press; 2006.
2. Kelly B, Shimoni T. Reintroducing the Tzanck smear. Am J Clin Dermatol. 2009;10:141-152.
3. Singhi M, Gupta L. Tzanck smear: a useful diagnostic tool. Indian J Dermatol Venereol Leprol. 2005;71:295.
4. Elston DM. Demodex mites: facts and controversies. Clin Dermatol. 2010;28:502-504.
5. Dupuy A, Dehen L, Bourrat E, et al. Accuracy of standard dermoscopy for diagnosing scabies. J Am Acad Dermatol. 2007;56:53-62.
6. Bolognia J, Schaffer J, Duncan K, et al. Dermatology Essentials. St. Louis, MO: Saunders Elsevier; 2014.
7. Ruocco E, Baroni A, Donnarumma G, et al. Diagnostic procedures in dermatology. Clin Dermatol. 2011;29:548-556.
Applications of Lasers in Medical Dermatology
The use of lasers in dermatology has had a major impact on the treatment of many dermatologic conditions. In this column practical applications of lasers in medical dermatology will be discussed to give dermatology residents a broad overview of both established indications and the reasoning behind the usage of lasers in treating these skin conditions. The applications for lasers in aesthetic dermatology are numerous and are constantly being refined and developed; they have been discussed extensively in the literature. Given the vast variety of uses of lasers in dermatology today, a comprehensive review of this topic would likely span several volumes. This article will focus on recent evidence regarding the use of lasers in medical dermatology, specifically laser treatment of selected common dermatoses and cutaneous malignancies.
Laser Treatment of Skin Diseases
Many common dermatoses seen in the dermatologist’s office (eg, discoid lupus erythematosus [DLE], morphea, alopecia) already have an established therapeutic ladder, with most patients responding to either first- or second-line therapies; however, a number of patients present with refractory disease that can be difficult to treat due to either treatment resistance or other contraindications to therapy. With the advent and development of modern lasers, we are now able to target many of these conditions and provide a viable safe treatment option for these patients. Although many physicians may be familiar with the use of the excimer laser in the treatment of psoriasis,1 a long-standing and well-accepted treatment modality for this condition, many novel applications for different types of lasers have been developed.
First, it is important to consider what a laser is able to accomplish to modulate the skin. With ablative lasers such as the CO2 laser, it is possible to destroy superficial layers of the skin (ie, the epidermis). It would stand to reason that this approach would be ideal for treating epidermal processes such as viral warts; in fact, this modality has been used for this indication for more than 3 decades, with the earliest references coming from the podiatric and urologic literature.2,3 Despite conflicting reports of the risk for human papillomavirus aerosolization and subsequent contamination of the treatment area,4,5 CO2 laser therapy has been advocated as a nonsurgical approach to difficult-to-treat cases of viral warts.
On the other hand, the pulsed dye laser (PDL) can target blood vessels because the wavelength corresponds to the absorption spectrum of hemoglobin and penetrates to the level of the dermis, while the pulse duration can be set to be shorter than the thermal relaxation time of a small cutaneous blood vessel.6 In clinical practice, the PDL has been used for the treatment of vascular lesions including hemangiomas, nevus flammeus, and other vascular proliferations.7-9 However, the PDL also can be used to target the vessels in cutaneous inflammatory diseases that feature vascular dilation and/or perivascular inflammation as a prominent feature.
Discoid lupus erythematosus is a form of chronic cutaneous lupus erythematosus that may be difficult to treat, with recalcitrant lesions displaying continued inflammation leading to chronic scarring and dyspigmentation. A small study (N=12) presented the efficacy of the PDL in the treatment of DLE lesions, suggesting that it has good efficacy in treating recalcitrant lesions with significant reduction in the cutaneous lupus erythematosus disease area and severity index after 6 weeks of treatment and 6 weeks of follow-up (P<.0001) with decreased erythema and scaling.10 It is important to note, however, that scarring, dyspigmentation, and atrophy were not affected, which suggests that early intervention may be optimal to prevent development of these sequelae. More interestingly, a more recent study expounded on this idea and attempted to examine pathophysiologic mechanisms behind this observed improvement. Evaluation of biopsy specimens before and after treatment and immunohistochemistry revealed that PDL treatment of cutaneous DLE lesions led to a decrease in vascular endothelial proteins—intercellular adhesion molecule 1 and vascular cell adhesion molecule 1—with a coincident reduction in the dermal lymphocytic infiltrate in treated lesions.11 These results offer a somewhat satisfying view on the correlation between the theory and basic science of laser therapy and the subsequent clinical benefits afforded by laser treatment. A case series provided further evidence that PDL or intense pulsed light can ameliorate the cutaneous lesions of DLE in 16 patients in whom all other treatments had failed.12
Several other inflammatory dermatoses can be treated with PDL, though the evidence for most of these conditions is sporadic at best, consisting mostly of case reports and a few case series. Granuloma faciale is one such condition, with evidence of efficacy of the PDL dating back as far as 1999,13 though a more recent case series of 4 patients only showed response in 2 patients.14 Because granuloma faciale features vasculitis as a prominent feature in its pathology, targeting the blood vessels may be helpful, but it is important to remember that there is a complex interplay between multiple factors. For example, treatment with typical fluences used in dermatology can be proinflammatory, leading to tissue damage, necrosis, and posttreatment erythema. However, low-level laser therapy (LLLT) has been shown to downregulate proinflammatory mediators.15 Additionally, the presence of a large burden of inflammatory cells also may alter the effectiveness of the laser. Several case reports also the show effectiveness of both PDL and the CO2 laser in treating lesions of cutaneous sarcoidosis, especially lupus pernio.16-19 Of these 2 modalities, the use of the CO2 laser for effective remodeling of lupus pernio may be more intuitive; however, it is still important to note that the mechanism of action of several of these laser modalities is unclear with regard to the clinical benefit shown. Morphea and scleroderma also have been treated with laser therapy. It is essential to understand that in many cases, laser therapy may be targeted to treat the precise cutaneous manifestations of disease in each individual patient (eg, CO2 laser to treat disabling contractures and calcinosis cutis,20,21 PDL to treat telangiectases related to morphea22). Again, the most critical consideration is that the treatment modality should align with the cutaneous lesion being targeted.
A relatively recent development in the use of lasers has been LLLT, which refers to the use of lasers below levels where they would cause any thermal effects, thereby limiting tissue damage. Although the technology has existed for decades, there has been a recent flurry of reports extolling the many benefits of LLLT; however, the true physiologic effects of LLLT have yet to be determined, with many studies trying to elucidate its numerous effects on various signaling pathways, cell proliferation, and cellular respiration.23-26 Upon reviewing the literature, the list of cutaneous conditions that are being treated with LLLT is vast, spanning acne, vitiligo, wounds, burns, psoriasis, and alopecia, among others.15 It is important to consider that the definition of LLLT in the literature is rather broad with a wide range of wavelengths, fluences, and power densities. As such, the specific laser settings and protocols may vary considerably among different practitioners and therefore the treatment results also may vary. Nevertheless, many studies have hinted at promising results in the use of LLLT in conditions that may have previously been extremely difficult to treat (eg, alopecia). Earlier trials had demonstrated a faster resolution time in patients with alopecia areata when LLLT was added to a topical regimen27; however, the improvement was modest and lesions tended to improve with or without LLLT. Perhaps more compelling is the use of LLLT in treating androgenetic alopecia, a condition for which a satisfying facile treatment would truly carry great impact. Although physicians should be cautious of studies regarding LLLT and hair regrowth that are conducted by groups who may stand to benefit from producing such a device, the results are nonetheless notable, if only for the relative paucity of other therapeutic approaches toward this condition.28,29 A randomized, double-blind, controlled, multicenter trial showed significant improvements in median hair thickness and density with LLLT (P=.01 and P=.003, respectively), though global appearance did not change significantly.30
Laser Treatment of Skin Cancer
Lasers also have been used to treat cutaneous malignancies. Although they may be powerful in the treatment of these conditions, this treatment approach must be used with caution. As with any superficial treatment modality for skin cancer, it is difficult to ascertain if a lesion has been completely treated without any residual cancer cells, and therein lies the main caveat of laser treatment. With the use of a modality that causes a cutaneous response that may mask any underlying process, it is important to ensure that there is a reasonable degree of certainty that this treatment can effectively remove a cancerous lesion in its entirety while avoiding the theoretical risk that disturbing underlying vasculature and/or lymphatics may be modulating the ability of a cancer to metastasize. Thankfully, current evidence does not suggest that there are any downsides to laser treatment for malignancies. Clinically, we know that basal cell carcinomas (BCCs) often feature prominent vasculature, with telangiectases being used as a clinical marker to suggest the diagnosis of a BCC. Capitalizing on this aspect of the clinical lesion, PDL has been used to treat BCCs in 2 small studies with a response rate of approximately 75% for small BCCs in both studies.31,32 A recent randomized controlled trial showed significant superiority of PDL as compared to the control (P<.0001) in treatment of BCC, with nearly 80% (44/56) of cases showing histologically proven complete remission at 6-month follow-up.33 Thus, we have some promising data that suggest PDL may be a viable treatment option in BCC, especially in areas that are difficult to treat surgically.
Additionally, a newer treatment approach for BCC capitalizes on the ability of confocal microscopy to provide a feasible, bedside imaging modality to identify tumor margins. Confocal microscopy has been used as a road map to identify where and how to apply the laser treatment, thus allowing for a higher likelihood of complete destruction of the tumor, at least in theory.34 Although the concept of using confocal microscopy to guide laser treatment of skin cancer has been shown in smaller proof-of-concept case series, it remains to be seen if it is not only an efficacious approach that may be widely adopted but also whether it is pragmatic to do so, as the equipment and expertise involved in using confocal microscopy is not trivial.
Finally, lasers also have been used in the treatment of mycosis fungoides (MF), or cutaneous T-cell lymphoma. It has been suggested that this modality is an excellent treatment option as a skin-directed therapy for stage IA or IB MFs limited to the acral surfaces or MF palmaris et plantaris.35 The reasoning behind this approach was the effectiveness of narrowband UVB for early-stage MF, with an excimer laser operating at a similar wavelength (308 nm) and offering similar therapeutic benefits while limiting adverse effects to surrounding skin.36 More recently, the excimer laser was applied to a small population of 6 patients, with 3 achieving complete response, 1 with partial response, 1 with stable disease, and 1 with progressive disease. The authors were careful to point out that the excimer laser should not be thought of as a replacement for narrowband UVB in early-stage MF but rather as an adjunctive treatment of specific targeted lesional areas.36
Conclusion
Lasers are an important part of the dermatologist’s treatment arsenal. Although much attention has been focused on laser treatment for aesthetic indications, it is important not to overlook the fact that lasers also can be useful in the treatment of refractory skin diseases, as a first-line treatment in some conditions such as vascular lesions, or as an adjunctive treatment modality. There is a great deal of exciting research that may lead to new indications and a better understanding of how to best use these powerful tools, and the outlook is bright for the use of lasers in dermatology.
1. Bonis B, Kemeny L, Dobozy A, et al. 308 nm UVB excimer laser for psoriasis. Lancet. 1997;350:1522.
2. Fuselier HA Jr, McBurney EI, Brannan W, et al. Treatment of condylomata acuminata with carbon dioxide laser. Urology. 1980;15:265-266.
3. Mueller TJ, Carlson BA, Lindy MP. The use of the carbon dioxide surgical laser for the treatment of verrucae. J Am Podiatry Assoc. 1980;70:136-141.
4. Weyandt GH, Tollmann F, Kristen P, et al. Low risk of contamination with human papilloma virus during treatment of condylomata acuminata with multilayer argon plasma coagulation and CO2 laser ablation. Arch Dermatol Res. 2011;303:141-144.
5. Ferenczy A, Bergeron C, Richart RM. Human papillomavirus DNA in CO2 laser-generated plume of smoke and its consequences to the surgeon. Obstet Gynecol. 1990;75:114-118.
6. Anderson RR, Parrish JA. Microvasculature can be selectively damaged using dye lasers: a basic theory and experimental evidence in human skin. Lasers Surg Med. 1981:263-276.
7. Morelli JG, Tan OT, Garden J, et al. Tunable dye laser (577 nm) treatment of port wine stains. Lasers Surg Med. 1986;6:94-99.
8. Reyes BA, Geronemus R. Treatment of port-wine stains during childhood with the flashlamp-pumped pulsed dye laser. J Am Acad Dermatol. 1990;23:1142-1148.
9. Ashinoff R, Geronemus RG. Capillary hemangiomas and treatment with the flash lamp-pumped pulsed dye laser. Arch Dermatol. 1991;127:202-205.
10. Erceg A, Bovenschen HJ, van de Kerkhof PC, et al. Efficacy and safety of pulsed dye laser treatment for cutaneous discoid lupus erythematosus. J Am Acad Dermatol. 2009;60:626-632.
11. Diez MT, Boixeda P, Moreno C, et al. Histopathology and immunohistochemistry of cutaneous lupus erythematosus after pulsed dye laser treatment. Dermatol Surg. 2011;37:971-981.
12. Ekback MP, Troilius A. Laser therapy for refractory discoid lupus erythematosus when everything else has failed. J Cosmet Laser Ther. 2013;15:260-265.
13. Welsh JH, Schroeder TL, Levy ML. Granuloma faciale in a child successfully treated with the pulsed dye laser. J Am Acad Dermatol. 1999;41:351-353.
14. Cheung ST, Lanigan SW. Granuloma faciale treated with the pulsed-dye laser: a case series. Clin Exp Dermatol. 2005;30:373-375.
15. Avci P, Gupta A, Sadasivam M, et al. Low-level laser (light) therapy (LLLT) in skin: stimulating, healing, restoring. Semin Cutan Med Surg. 2013;32:41-52.
16. Roos S, Raulin C, Ockenfels HM, et al. Successful treatment of cutaneous sarcoidosis lesions with the flashlamp pumped pulsed dye laser: a case report. Dermatol Surg. 2009;35:1139-1140.
17. Cliff S, Felix RH, Singh L, et al. The successful treatment of lupus pernio with the flashlamp pulsed dye laser. J Cutan Laser Ther. 1999;1:49-52.
18. O’Donoghue NB, Barlow RJ. Laser remodelling of nodular nasal lupus pernio. Clin Exp Dermatol. 2006;31:27-29.
19. Young HS, Chalmers RJ, Griffiths CE, et al. CO2 laser vaporization for disfiguring lupus pernio. J Cosmet Laser Ther. 2002;4:87-90.
20. Kineston D, Kwan JM, Uebelhoer NS, et al. Use of a fractional ablative 10.6-mum carbon dioxide laser in the treatment of a morphea-related contracture. Arch Dermatol. 2011;147:1148-1150.
21. Chamberlain AJ, Walker NP. Successful palliation and significant remission of cutaneous calcinosis in CREST syndrome with carbon dioxide laser. Dermatol Surg. 2003;29:968-970.
22. Ciatti S, Varga J, Greenbaum SS. The 585 nm flashlamp-pumped pulsed dye laser for the treatment of telangiectases in patients with scleroderma. J Am Acad Dermatol. 1996;35:487-488.
23. Karu TI, Kolyakov SF. Exact action spectra for cellular responses relevant to phototherapy. Photomed Laser Surg. 2005;23:355-361.
24. Greco M, Guida G, Perlino E, et al. Increase in RNA and protein synthesis by mitochondria irradiated with helium-neon laser. Biochem Biophys Res Commun. 1989;163:1428-1434.
25. Karu TI, Pyatibrat LV, Kalendo GS. Photobiological modulation of cell attachment via cytochrome c oxidase. Photochem Photobiol Sci. 2004;3:211-216.
26. Wong-Riley MT, Liang HL, Eells JT, et al. Photobiomodulation directly benefits primary neurons functionally inactivated by toxins: role of cytochrome c oxidase. J Biol Chem. 2005;280:4761-4771.
27. Yamazaki M, Miura Y, Tsuboi R, et al. Linear polarized infrared irradiation using Super Lizer is an effective treatment for multiple-type alopecia areata. Int J Dermatol. 2003;42:738-740.
28. Leavitt M, Charles G, Heyman E, et al. HairMax LaserComb laser phototherapy device in the treatment of male androgenetic alopecia: a randomized, double-blind, sham device-controlled, multicentre trial. Clin Drug Investig. 2009;29:283-292.
29. Munck A, Gavazzoni MF, Trueb RM. Use of low-level laser therapy as monotherapy or concomitant therapy for male and female androgenetic alopecia. Int J Trichology. 2014;6:45-49.
30. Kim H, Choi JW, Kim JY, et al. Low-level light therapy for androgenetic alopecia: a 24-week, randomized, double-blind, sham device-controlled multicenter trial. Dermatol Surg. 2013;39:1177-1183.
31. Minars N, Blyumin-Karasik M. Treatment of basal cell carcinomas with pulsed dye laser: a case series [published online ahead of print December 13, 2012]. J Skin Cancer. 2012;2012:286480.
32. Jalian HR, Avram MM, Stankiewicz KJ, et al. Combined 585 nm pulsed-dye and 1,064 nm Nd:YAG lasers for the treatment of basal cell carcinoma. Lasers Surg Med. 2014;46:1-7.
33. Karsai S, Friedl H, Buhck H, et al. The role of the 595-nm pulsed dye laser in treating superficial basal cell carcinoma: outcome of a double-blind randomized placebo-controlled trial [published online ahead of print July 12, 2014]. Br J Dermatol. doi:10.1111/bjd.13266.
34. Chen CS, Sierra H, Cordova M, et al. Confocal microscopy-guided laser ablation for superficial and early nodular Basal cell carcinoma: a promising surgical alternative for superficial skin cancers. JAMA Dermatol. 2014;150:994-998.
35. Jin SP, Jeon YK, Cho KH, et al. Excimer laser therapy (308 nm) for mycosis fungoides palmaris et plantaris: a skin-directed and anatomically feasible treatment. Br J Dermatol. 2010;163:651-653.
36. Deaver D, Cauthen A, Cohen G, et al. Excimer laser in the treatment of mycosis fungoides. J Am Acad Dermatol. 2014;70:1058-1060.
The use of lasers in dermatology has had a major impact on the treatment of many dermatologic conditions. In this column practical applications of lasers in medical dermatology will be discussed to give dermatology residents a broad overview of both established indications and the reasoning behind the usage of lasers in treating these skin conditions. The applications for lasers in aesthetic dermatology are numerous and are constantly being refined and developed; they have been discussed extensively in the literature. Given the vast variety of uses of lasers in dermatology today, a comprehensive review of this topic would likely span several volumes. This article will focus on recent evidence regarding the use of lasers in medical dermatology, specifically laser treatment of selected common dermatoses and cutaneous malignancies.
Laser Treatment of Skin Diseases
Many common dermatoses seen in the dermatologist’s office (eg, discoid lupus erythematosus [DLE], morphea, alopecia) already have an established therapeutic ladder, with most patients responding to either first- or second-line therapies; however, a number of patients present with refractory disease that can be difficult to treat due to either treatment resistance or other contraindications to therapy. With the advent and development of modern lasers, we are now able to target many of these conditions and provide a viable safe treatment option for these patients. Although many physicians may be familiar with the use of the excimer laser in the treatment of psoriasis,1 a long-standing and well-accepted treatment modality for this condition, many novel applications for different types of lasers have been developed.
First, it is important to consider what a laser is able to accomplish to modulate the skin. With ablative lasers such as the CO2 laser, it is possible to destroy superficial layers of the skin (ie, the epidermis). It would stand to reason that this approach would be ideal for treating epidermal processes such as viral warts; in fact, this modality has been used for this indication for more than 3 decades, with the earliest references coming from the podiatric and urologic literature.2,3 Despite conflicting reports of the risk for human papillomavirus aerosolization and subsequent contamination of the treatment area,4,5 CO2 laser therapy has been advocated as a nonsurgical approach to difficult-to-treat cases of viral warts.
On the other hand, the pulsed dye laser (PDL) can target blood vessels because the wavelength corresponds to the absorption spectrum of hemoglobin and penetrates to the level of the dermis, while the pulse duration can be set to be shorter than the thermal relaxation time of a small cutaneous blood vessel.6 In clinical practice, the PDL has been used for the treatment of vascular lesions including hemangiomas, nevus flammeus, and other vascular proliferations.7-9 However, the PDL also can be used to target the vessels in cutaneous inflammatory diseases that feature vascular dilation and/or perivascular inflammation as a prominent feature.
Discoid lupus erythematosus is a form of chronic cutaneous lupus erythematosus that may be difficult to treat, with recalcitrant lesions displaying continued inflammation leading to chronic scarring and dyspigmentation. A small study (N=12) presented the efficacy of the PDL in the treatment of DLE lesions, suggesting that it has good efficacy in treating recalcitrant lesions with significant reduction in the cutaneous lupus erythematosus disease area and severity index after 6 weeks of treatment and 6 weeks of follow-up (P<.0001) with decreased erythema and scaling.10 It is important to note, however, that scarring, dyspigmentation, and atrophy were not affected, which suggests that early intervention may be optimal to prevent development of these sequelae. More interestingly, a more recent study expounded on this idea and attempted to examine pathophysiologic mechanisms behind this observed improvement. Evaluation of biopsy specimens before and after treatment and immunohistochemistry revealed that PDL treatment of cutaneous DLE lesions led to a decrease in vascular endothelial proteins—intercellular adhesion molecule 1 and vascular cell adhesion molecule 1—with a coincident reduction in the dermal lymphocytic infiltrate in treated lesions.11 These results offer a somewhat satisfying view on the correlation between the theory and basic science of laser therapy and the subsequent clinical benefits afforded by laser treatment. A case series provided further evidence that PDL or intense pulsed light can ameliorate the cutaneous lesions of DLE in 16 patients in whom all other treatments had failed.12
Several other inflammatory dermatoses can be treated with PDL, though the evidence for most of these conditions is sporadic at best, consisting mostly of case reports and a few case series. Granuloma faciale is one such condition, with evidence of efficacy of the PDL dating back as far as 1999,13 though a more recent case series of 4 patients only showed response in 2 patients.14 Because granuloma faciale features vasculitis as a prominent feature in its pathology, targeting the blood vessels may be helpful, but it is important to remember that there is a complex interplay between multiple factors. For example, treatment with typical fluences used in dermatology can be proinflammatory, leading to tissue damage, necrosis, and posttreatment erythema. However, low-level laser therapy (LLLT) has been shown to downregulate proinflammatory mediators.15 Additionally, the presence of a large burden of inflammatory cells also may alter the effectiveness of the laser. Several case reports also the show effectiveness of both PDL and the CO2 laser in treating lesions of cutaneous sarcoidosis, especially lupus pernio.16-19 Of these 2 modalities, the use of the CO2 laser for effective remodeling of lupus pernio may be more intuitive; however, it is still important to note that the mechanism of action of several of these laser modalities is unclear with regard to the clinical benefit shown. Morphea and scleroderma also have been treated with laser therapy. It is essential to understand that in many cases, laser therapy may be targeted to treat the precise cutaneous manifestations of disease in each individual patient (eg, CO2 laser to treat disabling contractures and calcinosis cutis,20,21 PDL to treat telangiectases related to morphea22). Again, the most critical consideration is that the treatment modality should align with the cutaneous lesion being targeted.
A relatively recent development in the use of lasers has been LLLT, which refers to the use of lasers below levels where they would cause any thermal effects, thereby limiting tissue damage. Although the technology has existed for decades, there has been a recent flurry of reports extolling the many benefits of LLLT; however, the true physiologic effects of LLLT have yet to be determined, with many studies trying to elucidate its numerous effects on various signaling pathways, cell proliferation, and cellular respiration.23-26 Upon reviewing the literature, the list of cutaneous conditions that are being treated with LLLT is vast, spanning acne, vitiligo, wounds, burns, psoriasis, and alopecia, among others.15 It is important to consider that the definition of LLLT in the literature is rather broad with a wide range of wavelengths, fluences, and power densities. As such, the specific laser settings and protocols may vary considerably among different practitioners and therefore the treatment results also may vary. Nevertheless, many studies have hinted at promising results in the use of LLLT in conditions that may have previously been extremely difficult to treat (eg, alopecia). Earlier trials had demonstrated a faster resolution time in patients with alopecia areata when LLLT was added to a topical regimen27; however, the improvement was modest and lesions tended to improve with or without LLLT. Perhaps more compelling is the use of LLLT in treating androgenetic alopecia, a condition for which a satisfying facile treatment would truly carry great impact. Although physicians should be cautious of studies regarding LLLT and hair regrowth that are conducted by groups who may stand to benefit from producing such a device, the results are nonetheless notable, if only for the relative paucity of other therapeutic approaches toward this condition.28,29 A randomized, double-blind, controlled, multicenter trial showed significant improvements in median hair thickness and density with LLLT (P=.01 and P=.003, respectively), though global appearance did not change significantly.30
Laser Treatment of Skin Cancer
Lasers also have been used to treat cutaneous malignancies. Although they may be powerful in the treatment of these conditions, this treatment approach must be used with caution. As with any superficial treatment modality for skin cancer, it is difficult to ascertain if a lesion has been completely treated without any residual cancer cells, and therein lies the main caveat of laser treatment. With the use of a modality that causes a cutaneous response that may mask any underlying process, it is important to ensure that there is a reasonable degree of certainty that this treatment can effectively remove a cancerous lesion in its entirety while avoiding the theoretical risk that disturbing underlying vasculature and/or lymphatics may be modulating the ability of a cancer to metastasize. Thankfully, current evidence does not suggest that there are any downsides to laser treatment for malignancies. Clinically, we know that basal cell carcinomas (BCCs) often feature prominent vasculature, with telangiectases being used as a clinical marker to suggest the diagnosis of a BCC. Capitalizing on this aspect of the clinical lesion, PDL has been used to treat BCCs in 2 small studies with a response rate of approximately 75% for small BCCs in both studies.31,32 A recent randomized controlled trial showed significant superiority of PDL as compared to the control (P<.0001) in treatment of BCC, with nearly 80% (44/56) of cases showing histologically proven complete remission at 6-month follow-up.33 Thus, we have some promising data that suggest PDL may be a viable treatment option in BCC, especially in areas that are difficult to treat surgically.
Additionally, a newer treatment approach for BCC capitalizes on the ability of confocal microscopy to provide a feasible, bedside imaging modality to identify tumor margins. Confocal microscopy has been used as a road map to identify where and how to apply the laser treatment, thus allowing for a higher likelihood of complete destruction of the tumor, at least in theory.34 Although the concept of using confocal microscopy to guide laser treatment of skin cancer has been shown in smaller proof-of-concept case series, it remains to be seen if it is not only an efficacious approach that may be widely adopted but also whether it is pragmatic to do so, as the equipment and expertise involved in using confocal microscopy is not trivial.
Finally, lasers also have been used in the treatment of mycosis fungoides (MF), or cutaneous T-cell lymphoma. It has been suggested that this modality is an excellent treatment option as a skin-directed therapy for stage IA or IB MFs limited to the acral surfaces or MF palmaris et plantaris.35 The reasoning behind this approach was the effectiveness of narrowband UVB for early-stage MF, with an excimer laser operating at a similar wavelength (308 nm) and offering similar therapeutic benefits while limiting adverse effects to surrounding skin.36 More recently, the excimer laser was applied to a small population of 6 patients, with 3 achieving complete response, 1 with partial response, 1 with stable disease, and 1 with progressive disease. The authors were careful to point out that the excimer laser should not be thought of as a replacement for narrowband UVB in early-stage MF but rather as an adjunctive treatment of specific targeted lesional areas.36
Conclusion
Lasers are an important part of the dermatologist’s treatment arsenal. Although much attention has been focused on laser treatment for aesthetic indications, it is important not to overlook the fact that lasers also can be useful in the treatment of refractory skin diseases, as a first-line treatment in some conditions such as vascular lesions, or as an adjunctive treatment modality. There is a great deal of exciting research that may lead to new indications and a better understanding of how to best use these powerful tools, and the outlook is bright for the use of lasers in dermatology.
The use of lasers in dermatology has had a major impact on the treatment of many dermatologic conditions. In this column practical applications of lasers in medical dermatology will be discussed to give dermatology residents a broad overview of both established indications and the reasoning behind the usage of lasers in treating these skin conditions. The applications for lasers in aesthetic dermatology are numerous and are constantly being refined and developed; they have been discussed extensively in the literature. Given the vast variety of uses of lasers in dermatology today, a comprehensive review of this topic would likely span several volumes. This article will focus on recent evidence regarding the use of lasers in medical dermatology, specifically laser treatment of selected common dermatoses and cutaneous malignancies.
Laser Treatment of Skin Diseases
Many common dermatoses seen in the dermatologist’s office (eg, discoid lupus erythematosus [DLE], morphea, alopecia) already have an established therapeutic ladder, with most patients responding to either first- or second-line therapies; however, a number of patients present with refractory disease that can be difficult to treat due to either treatment resistance or other contraindications to therapy. With the advent and development of modern lasers, we are now able to target many of these conditions and provide a viable safe treatment option for these patients. Although many physicians may be familiar with the use of the excimer laser in the treatment of psoriasis,1 a long-standing and well-accepted treatment modality for this condition, many novel applications for different types of lasers have been developed.
First, it is important to consider what a laser is able to accomplish to modulate the skin. With ablative lasers such as the CO2 laser, it is possible to destroy superficial layers of the skin (ie, the epidermis). It would stand to reason that this approach would be ideal for treating epidermal processes such as viral warts; in fact, this modality has been used for this indication for more than 3 decades, with the earliest references coming from the podiatric and urologic literature.2,3 Despite conflicting reports of the risk for human papillomavirus aerosolization and subsequent contamination of the treatment area,4,5 CO2 laser therapy has been advocated as a nonsurgical approach to difficult-to-treat cases of viral warts.
On the other hand, the pulsed dye laser (PDL) can target blood vessels because the wavelength corresponds to the absorption spectrum of hemoglobin and penetrates to the level of the dermis, while the pulse duration can be set to be shorter than the thermal relaxation time of a small cutaneous blood vessel.6 In clinical practice, the PDL has been used for the treatment of vascular lesions including hemangiomas, nevus flammeus, and other vascular proliferations.7-9 However, the PDL also can be used to target the vessels in cutaneous inflammatory diseases that feature vascular dilation and/or perivascular inflammation as a prominent feature.
Discoid lupus erythematosus is a form of chronic cutaneous lupus erythematosus that may be difficult to treat, with recalcitrant lesions displaying continued inflammation leading to chronic scarring and dyspigmentation. A small study (N=12) presented the efficacy of the PDL in the treatment of DLE lesions, suggesting that it has good efficacy in treating recalcitrant lesions with significant reduction in the cutaneous lupus erythematosus disease area and severity index after 6 weeks of treatment and 6 weeks of follow-up (P<.0001) with decreased erythema and scaling.10 It is important to note, however, that scarring, dyspigmentation, and atrophy were not affected, which suggests that early intervention may be optimal to prevent development of these sequelae. More interestingly, a more recent study expounded on this idea and attempted to examine pathophysiologic mechanisms behind this observed improvement. Evaluation of biopsy specimens before and after treatment and immunohistochemistry revealed that PDL treatment of cutaneous DLE lesions led to a decrease in vascular endothelial proteins—intercellular adhesion molecule 1 and vascular cell adhesion molecule 1—with a coincident reduction in the dermal lymphocytic infiltrate in treated lesions.11 These results offer a somewhat satisfying view on the correlation between the theory and basic science of laser therapy and the subsequent clinical benefits afforded by laser treatment. A case series provided further evidence that PDL or intense pulsed light can ameliorate the cutaneous lesions of DLE in 16 patients in whom all other treatments had failed.12
Several other inflammatory dermatoses can be treated with PDL, though the evidence for most of these conditions is sporadic at best, consisting mostly of case reports and a few case series. Granuloma faciale is one such condition, with evidence of efficacy of the PDL dating back as far as 1999,13 though a more recent case series of 4 patients only showed response in 2 patients.14 Because granuloma faciale features vasculitis as a prominent feature in its pathology, targeting the blood vessels may be helpful, but it is important to remember that there is a complex interplay between multiple factors. For example, treatment with typical fluences used in dermatology can be proinflammatory, leading to tissue damage, necrosis, and posttreatment erythema. However, low-level laser therapy (LLLT) has been shown to downregulate proinflammatory mediators.15 Additionally, the presence of a large burden of inflammatory cells also may alter the effectiveness of the laser. Several case reports also the show effectiveness of both PDL and the CO2 laser in treating lesions of cutaneous sarcoidosis, especially lupus pernio.16-19 Of these 2 modalities, the use of the CO2 laser for effective remodeling of lupus pernio may be more intuitive; however, it is still important to note that the mechanism of action of several of these laser modalities is unclear with regard to the clinical benefit shown. Morphea and scleroderma also have been treated with laser therapy. It is essential to understand that in many cases, laser therapy may be targeted to treat the precise cutaneous manifestations of disease in each individual patient (eg, CO2 laser to treat disabling contractures and calcinosis cutis,20,21 PDL to treat telangiectases related to morphea22). Again, the most critical consideration is that the treatment modality should align with the cutaneous lesion being targeted.
A relatively recent development in the use of lasers has been LLLT, which refers to the use of lasers below levels where they would cause any thermal effects, thereby limiting tissue damage. Although the technology has existed for decades, there has been a recent flurry of reports extolling the many benefits of LLLT; however, the true physiologic effects of LLLT have yet to be determined, with many studies trying to elucidate its numerous effects on various signaling pathways, cell proliferation, and cellular respiration.23-26 Upon reviewing the literature, the list of cutaneous conditions that are being treated with LLLT is vast, spanning acne, vitiligo, wounds, burns, psoriasis, and alopecia, among others.15 It is important to consider that the definition of LLLT in the literature is rather broad with a wide range of wavelengths, fluences, and power densities. As such, the specific laser settings and protocols may vary considerably among different practitioners and therefore the treatment results also may vary. Nevertheless, many studies have hinted at promising results in the use of LLLT in conditions that may have previously been extremely difficult to treat (eg, alopecia). Earlier trials had demonstrated a faster resolution time in patients with alopecia areata when LLLT was added to a topical regimen27; however, the improvement was modest and lesions tended to improve with or without LLLT. Perhaps more compelling is the use of LLLT in treating androgenetic alopecia, a condition for which a satisfying facile treatment would truly carry great impact. Although physicians should be cautious of studies regarding LLLT and hair regrowth that are conducted by groups who may stand to benefit from producing such a device, the results are nonetheless notable, if only for the relative paucity of other therapeutic approaches toward this condition.28,29 A randomized, double-blind, controlled, multicenter trial showed significant improvements in median hair thickness and density with LLLT (P=.01 and P=.003, respectively), though global appearance did not change significantly.30
Laser Treatment of Skin Cancer
Lasers also have been used to treat cutaneous malignancies. Although they may be powerful in the treatment of these conditions, this treatment approach must be used with caution. As with any superficial treatment modality for skin cancer, it is difficult to ascertain if a lesion has been completely treated without any residual cancer cells, and therein lies the main caveat of laser treatment. With the use of a modality that causes a cutaneous response that may mask any underlying process, it is important to ensure that there is a reasonable degree of certainty that this treatment can effectively remove a cancerous lesion in its entirety while avoiding the theoretical risk that disturbing underlying vasculature and/or lymphatics may be modulating the ability of a cancer to metastasize. Thankfully, current evidence does not suggest that there are any downsides to laser treatment for malignancies. Clinically, we know that basal cell carcinomas (BCCs) often feature prominent vasculature, with telangiectases being used as a clinical marker to suggest the diagnosis of a BCC. Capitalizing on this aspect of the clinical lesion, PDL has been used to treat BCCs in 2 small studies with a response rate of approximately 75% for small BCCs in both studies.31,32 A recent randomized controlled trial showed significant superiority of PDL as compared to the control (P<.0001) in treatment of BCC, with nearly 80% (44/56) of cases showing histologically proven complete remission at 6-month follow-up.33 Thus, we have some promising data that suggest PDL may be a viable treatment option in BCC, especially in areas that are difficult to treat surgically.
Additionally, a newer treatment approach for BCC capitalizes on the ability of confocal microscopy to provide a feasible, bedside imaging modality to identify tumor margins. Confocal microscopy has been used as a road map to identify where and how to apply the laser treatment, thus allowing for a higher likelihood of complete destruction of the tumor, at least in theory.34 Although the concept of using confocal microscopy to guide laser treatment of skin cancer has been shown in smaller proof-of-concept case series, it remains to be seen if it is not only an efficacious approach that may be widely adopted but also whether it is pragmatic to do so, as the equipment and expertise involved in using confocal microscopy is not trivial.
Finally, lasers also have been used in the treatment of mycosis fungoides (MF), or cutaneous T-cell lymphoma. It has been suggested that this modality is an excellent treatment option as a skin-directed therapy for stage IA or IB MFs limited to the acral surfaces or MF palmaris et plantaris.35 The reasoning behind this approach was the effectiveness of narrowband UVB for early-stage MF, with an excimer laser operating at a similar wavelength (308 nm) and offering similar therapeutic benefits while limiting adverse effects to surrounding skin.36 More recently, the excimer laser was applied to a small population of 6 patients, with 3 achieving complete response, 1 with partial response, 1 with stable disease, and 1 with progressive disease. The authors were careful to point out that the excimer laser should not be thought of as a replacement for narrowband UVB in early-stage MF but rather as an adjunctive treatment of specific targeted lesional areas.36
Conclusion
Lasers are an important part of the dermatologist’s treatment arsenal. Although much attention has been focused on laser treatment for aesthetic indications, it is important not to overlook the fact that lasers also can be useful in the treatment of refractory skin diseases, as a first-line treatment in some conditions such as vascular lesions, or as an adjunctive treatment modality. There is a great deal of exciting research that may lead to new indications and a better understanding of how to best use these powerful tools, and the outlook is bright for the use of lasers in dermatology.
1. Bonis B, Kemeny L, Dobozy A, et al. 308 nm UVB excimer laser for psoriasis. Lancet. 1997;350:1522.
2. Fuselier HA Jr, McBurney EI, Brannan W, et al. Treatment of condylomata acuminata with carbon dioxide laser. Urology. 1980;15:265-266.
3. Mueller TJ, Carlson BA, Lindy MP. The use of the carbon dioxide surgical laser for the treatment of verrucae. J Am Podiatry Assoc. 1980;70:136-141.
4. Weyandt GH, Tollmann F, Kristen P, et al. Low risk of contamination with human papilloma virus during treatment of condylomata acuminata with multilayer argon plasma coagulation and CO2 laser ablation. Arch Dermatol Res. 2011;303:141-144.
5. Ferenczy A, Bergeron C, Richart RM. Human papillomavirus DNA in CO2 laser-generated plume of smoke and its consequences to the surgeon. Obstet Gynecol. 1990;75:114-118.
6. Anderson RR, Parrish JA. Microvasculature can be selectively damaged using dye lasers: a basic theory and experimental evidence in human skin. Lasers Surg Med. 1981:263-276.
7. Morelli JG, Tan OT, Garden J, et al. Tunable dye laser (577 nm) treatment of port wine stains. Lasers Surg Med. 1986;6:94-99.
8. Reyes BA, Geronemus R. Treatment of port-wine stains during childhood with the flashlamp-pumped pulsed dye laser. J Am Acad Dermatol. 1990;23:1142-1148.
9. Ashinoff R, Geronemus RG. Capillary hemangiomas and treatment with the flash lamp-pumped pulsed dye laser. Arch Dermatol. 1991;127:202-205.
10. Erceg A, Bovenschen HJ, van de Kerkhof PC, et al. Efficacy and safety of pulsed dye laser treatment for cutaneous discoid lupus erythematosus. J Am Acad Dermatol. 2009;60:626-632.
11. Diez MT, Boixeda P, Moreno C, et al. Histopathology and immunohistochemistry of cutaneous lupus erythematosus after pulsed dye laser treatment. Dermatol Surg. 2011;37:971-981.
12. Ekback MP, Troilius A. Laser therapy for refractory discoid lupus erythematosus when everything else has failed. J Cosmet Laser Ther. 2013;15:260-265.
13. Welsh JH, Schroeder TL, Levy ML. Granuloma faciale in a child successfully treated with the pulsed dye laser. J Am Acad Dermatol. 1999;41:351-353.
14. Cheung ST, Lanigan SW. Granuloma faciale treated with the pulsed-dye laser: a case series. Clin Exp Dermatol. 2005;30:373-375.
15. Avci P, Gupta A, Sadasivam M, et al. Low-level laser (light) therapy (LLLT) in skin: stimulating, healing, restoring. Semin Cutan Med Surg. 2013;32:41-52.
16. Roos S, Raulin C, Ockenfels HM, et al. Successful treatment of cutaneous sarcoidosis lesions with the flashlamp pumped pulsed dye laser: a case report. Dermatol Surg. 2009;35:1139-1140.
17. Cliff S, Felix RH, Singh L, et al. The successful treatment of lupus pernio with the flashlamp pulsed dye laser. J Cutan Laser Ther. 1999;1:49-52.
18. O’Donoghue NB, Barlow RJ. Laser remodelling of nodular nasal lupus pernio. Clin Exp Dermatol. 2006;31:27-29.
19. Young HS, Chalmers RJ, Griffiths CE, et al. CO2 laser vaporization for disfiguring lupus pernio. J Cosmet Laser Ther. 2002;4:87-90.
20. Kineston D, Kwan JM, Uebelhoer NS, et al. Use of a fractional ablative 10.6-mum carbon dioxide laser in the treatment of a morphea-related contracture. Arch Dermatol. 2011;147:1148-1150.
21. Chamberlain AJ, Walker NP. Successful palliation and significant remission of cutaneous calcinosis in CREST syndrome with carbon dioxide laser. Dermatol Surg. 2003;29:968-970.
22. Ciatti S, Varga J, Greenbaum SS. The 585 nm flashlamp-pumped pulsed dye laser for the treatment of telangiectases in patients with scleroderma. J Am Acad Dermatol. 1996;35:487-488.
23. Karu TI, Kolyakov SF. Exact action spectra for cellular responses relevant to phototherapy. Photomed Laser Surg. 2005;23:355-361.
24. Greco M, Guida G, Perlino E, et al. Increase in RNA and protein synthesis by mitochondria irradiated with helium-neon laser. Biochem Biophys Res Commun. 1989;163:1428-1434.
25. Karu TI, Pyatibrat LV, Kalendo GS. Photobiological modulation of cell attachment via cytochrome c oxidase. Photochem Photobiol Sci. 2004;3:211-216.
26. Wong-Riley MT, Liang HL, Eells JT, et al. Photobiomodulation directly benefits primary neurons functionally inactivated by toxins: role of cytochrome c oxidase. J Biol Chem. 2005;280:4761-4771.
27. Yamazaki M, Miura Y, Tsuboi R, et al. Linear polarized infrared irradiation using Super Lizer is an effective treatment for multiple-type alopecia areata. Int J Dermatol. 2003;42:738-740.
28. Leavitt M, Charles G, Heyman E, et al. HairMax LaserComb laser phototherapy device in the treatment of male androgenetic alopecia: a randomized, double-blind, sham device-controlled, multicentre trial. Clin Drug Investig. 2009;29:283-292.
29. Munck A, Gavazzoni MF, Trueb RM. Use of low-level laser therapy as monotherapy or concomitant therapy for male and female androgenetic alopecia. Int J Trichology. 2014;6:45-49.
30. Kim H, Choi JW, Kim JY, et al. Low-level light therapy for androgenetic alopecia: a 24-week, randomized, double-blind, sham device-controlled multicenter trial. Dermatol Surg. 2013;39:1177-1183.
31. Minars N, Blyumin-Karasik M. Treatment of basal cell carcinomas with pulsed dye laser: a case series [published online ahead of print December 13, 2012]. J Skin Cancer. 2012;2012:286480.
32. Jalian HR, Avram MM, Stankiewicz KJ, et al. Combined 585 nm pulsed-dye and 1,064 nm Nd:YAG lasers for the treatment of basal cell carcinoma. Lasers Surg Med. 2014;46:1-7.
33. Karsai S, Friedl H, Buhck H, et al. The role of the 595-nm pulsed dye laser in treating superficial basal cell carcinoma: outcome of a double-blind randomized placebo-controlled trial [published online ahead of print July 12, 2014]. Br J Dermatol. doi:10.1111/bjd.13266.
34. Chen CS, Sierra H, Cordova M, et al. Confocal microscopy-guided laser ablation for superficial and early nodular Basal cell carcinoma: a promising surgical alternative for superficial skin cancers. JAMA Dermatol. 2014;150:994-998.
35. Jin SP, Jeon YK, Cho KH, et al. Excimer laser therapy (308 nm) for mycosis fungoides palmaris et plantaris: a skin-directed and anatomically feasible treatment. Br J Dermatol. 2010;163:651-653.
36. Deaver D, Cauthen A, Cohen G, et al. Excimer laser in the treatment of mycosis fungoides. J Am Acad Dermatol. 2014;70:1058-1060.
1. Bonis B, Kemeny L, Dobozy A, et al. 308 nm UVB excimer laser for psoriasis. Lancet. 1997;350:1522.
2. Fuselier HA Jr, McBurney EI, Brannan W, et al. Treatment of condylomata acuminata with carbon dioxide laser. Urology. 1980;15:265-266.
3. Mueller TJ, Carlson BA, Lindy MP. The use of the carbon dioxide surgical laser for the treatment of verrucae. J Am Podiatry Assoc. 1980;70:136-141.
4. Weyandt GH, Tollmann F, Kristen P, et al. Low risk of contamination with human papilloma virus during treatment of condylomata acuminata with multilayer argon plasma coagulation and CO2 laser ablation. Arch Dermatol Res. 2011;303:141-144.
5. Ferenczy A, Bergeron C, Richart RM. Human papillomavirus DNA in CO2 laser-generated plume of smoke and its consequences to the surgeon. Obstet Gynecol. 1990;75:114-118.
6. Anderson RR, Parrish JA. Microvasculature can be selectively damaged using dye lasers: a basic theory and experimental evidence in human skin. Lasers Surg Med. 1981:263-276.
7. Morelli JG, Tan OT, Garden J, et al. Tunable dye laser (577 nm) treatment of port wine stains. Lasers Surg Med. 1986;6:94-99.
8. Reyes BA, Geronemus R. Treatment of port-wine stains during childhood with the flashlamp-pumped pulsed dye laser. J Am Acad Dermatol. 1990;23:1142-1148.
9. Ashinoff R, Geronemus RG. Capillary hemangiomas and treatment with the flash lamp-pumped pulsed dye laser. Arch Dermatol. 1991;127:202-205.
10. Erceg A, Bovenschen HJ, van de Kerkhof PC, et al. Efficacy and safety of pulsed dye laser treatment for cutaneous discoid lupus erythematosus. J Am Acad Dermatol. 2009;60:626-632.
11. Diez MT, Boixeda P, Moreno C, et al. Histopathology and immunohistochemistry of cutaneous lupus erythematosus after pulsed dye laser treatment. Dermatol Surg. 2011;37:971-981.
12. Ekback MP, Troilius A. Laser therapy for refractory discoid lupus erythematosus when everything else has failed. J Cosmet Laser Ther. 2013;15:260-265.
13. Welsh JH, Schroeder TL, Levy ML. Granuloma faciale in a child successfully treated with the pulsed dye laser. J Am Acad Dermatol. 1999;41:351-353.
14. Cheung ST, Lanigan SW. Granuloma faciale treated with the pulsed-dye laser: a case series. Clin Exp Dermatol. 2005;30:373-375.
15. Avci P, Gupta A, Sadasivam M, et al. Low-level laser (light) therapy (LLLT) in skin: stimulating, healing, restoring. Semin Cutan Med Surg. 2013;32:41-52.
16. Roos S, Raulin C, Ockenfels HM, et al. Successful treatment of cutaneous sarcoidosis lesions with the flashlamp pumped pulsed dye laser: a case report. Dermatol Surg. 2009;35:1139-1140.
17. Cliff S, Felix RH, Singh L, et al. The successful treatment of lupus pernio with the flashlamp pulsed dye laser. J Cutan Laser Ther. 1999;1:49-52.
18. O’Donoghue NB, Barlow RJ. Laser remodelling of nodular nasal lupus pernio. Clin Exp Dermatol. 2006;31:27-29.
19. Young HS, Chalmers RJ, Griffiths CE, et al. CO2 laser vaporization for disfiguring lupus pernio. J Cosmet Laser Ther. 2002;4:87-90.
20. Kineston D, Kwan JM, Uebelhoer NS, et al. Use of a fractional ablative 10.6-mum carbon dioxide laser in the treatment of a morphea-related contracture. Arch Dermatol. 2011;147:1148-1150.
21. Chamberlain AJ, Walker NP. Successful palliation and significant remission of cutaneous calcinosis in CREST syndrome with carbon dioxide laser. Dermatol Surg. 2003;29:968-970.
22. Ciatti S, Varga J, Greenbaum SS. The 585 nm flashlamp-pumped pulsed dye laser for the treatment of telangiectases in patients with scleroderma. J Am Acad Dermatol. 1996;35:487-488.
23. Karu TI, Kolyakov SF. Exact action spectra for cellular responses relevant to phototherapy. Photomed Laser Surg. 2005;23:355-361.
24. Greco M, Guida G, Perlino E, et al. Increase in RNA and protein synthesis by mitochondria irradiated with helium-neon laser. Biochem Biophys Res Commun. 1989;163:1428-1434.
25. Karu TI, Pyatibrat LV, Kalendo GS. Photobiological modulation of cell attachment via cytochrome c oxidase. Photochem Photobiol Sci. 2004;3:211-216.
26. Wong-Riley MT, Liang HL, Eells JT, et al. Photobiomodulation directly benefits primary neurons functionally inactivated by toxins: role of cytochrome c oxidase. J Biol Chem. 2005;280:4761-4771.
27. Yamazaki M, Miura Y, Tsuboi R, et al. Linear polarized infrared irradiation using Super Lizer is an effective treatment for multiple-type alopecia areata. Int J Dermatol. 2003;42:738-740.
28. Leavitt M, Charles G, Heyman E, et al. HairMax LaserComb laser phototherapy device in the treatment of male androgenetic alopecia: a randomized, double-blind, sham device-controlled, multicentre trial. Clin Drug Investig. 2009;29:283-292.
29. Munck A, Gavazzoni MF, Trueb RM. Use of low-level laser therapy as monotherapy or concomitant therapy for male and female androgenetic alopecia. Int J Trichology. 2014;6:45-49.
30. Kim H, Choi JW, Kim JY, et al. Low-level light therapy for androgenetic alopecia: a 24-week, randomized, double-blind, sham device-controlled multicenter trial. Dermatol Surg. 2013;39:1177-1183.
31. Minars N, Blyumin-Karasik M. Treatment of basal cell carcinomas with pulsed dye laser: a case series [published online ahead of print December 13, 2012]. J Skin Cancer. 2012;2012:286480.
32. Jalian HR, Avram MM, Stankiewicz KJ, et al. Combined 585 nm pulsed-dye and 1,064 nm Nd:YAG lasers for the treatment of basal cell carcinoma. Lasers Surg Med. 2014;46:1-7.
33. Karsai S, Friedl H, Buhck H, et al. The role of the 595-nm pulsed dye laser in treating superficial basal cell carcinoma: outcome of a double-blind randomized placebo-controlled trial [published online ahead of print July 12, 2014]. Br J Dermatol. doi:10.1111/bjd.13266.
34. Chen CS, Sierra H, Cordova M, et al. Confocal microscopy-guided laser ablation for superficial and early nodular Basal cell carcinoma: a promising surgical alternative for superficial skin cancers. JAMA Dermatol. 2014;150:994-998.
35. Jin SP, Jeon YK, Cho KH, et al. Excimer laser therapy (308 nm) for mycosis fungoides palmaris et plantaris: a skin-directed and anatomically feasible treatment. Br J Dermatol. 2010;163:651-653.
36. Deaver D, Cauthen A, Cohen G, et al. Excimer laser in the treatment of mycosis fungoides. J Am Acad Dermatol. 2014;70:1058-1060.
Top Dermatology Residency Programs
For more information, access Dr. Wu's article from the October 2014 issue, "US Dermatology Residency Program Rankings."
For more information, access Dr. Wu's article from the October 2014 issue, "US Dermatology Residency Program Rankings."
For more information, access Dr. Wu's article from the October 2014 issue, "US Dermatology Residency Program Rankings."
Discharging your patient: A complex process
Let me begin with a story.
A few years ago, when I was hoping to get into a psychiatry residency program, I did a month-long rotation in the intensive care unit (ICU) of a local hospital. One of our patients was a long-term resident of a nearby assisted living facility, who was treated for exacerbation of a chronic medical illness. Eventually this patient was stabilized to the point at which he could be discharged.
When the ICU physician decided to discharge this patient, he told the team that the man would need to be “sent back to a nursing home.” The social worker, assuming that the physician wanted to place the patient in a skilled nursing facility, spent several hours trying to place the man in one of the local facilities. When the patient’s daughter arrived to visit her father and began asking questions about why he was being placed in a nursing home, staff immediately realized that the physician had simply meant for him to go back “home”—that is, to the facility from which he had come and where he had been living for several years.
Being the only person in the ICU who was a licensed nursing home administrator, with more than 10 years experience in a long-term care, I should have pointed out this miscommunication or, at least, should have raised the question to clarify the physician’s intent. At the time, however, I wasn’t comfortable expressing my concern because I was “just an FMG observer” trying to stay on the attending’s good side.
I made a commitment to myself, however, to always talk about patients’ long-term care options and discharge planning algorithm with medical students, fellow residents, and other medical professionals I meet in my work. The following is an expression of that commitment.
Why focus on discharge when care is still underway?
Discharge planning usually begins on the first day of hospitalization. Before we are ready to discharge any patient, we, the physicians, usually have had many conversations with members of the multidisciplinary team and, always, with the patient and his (her) guardian(s). Why do we do all of this? The answer is simple: Physicians make the ultimate decision about what kind of environment (clinical, social, etc.) the patient is safe to be discharged to; after that decision is made, everything else is the patient’s choice. Our decision should be based on, first, global assessment of functioning—the ability to perform activities of daily living (ADL) and instrumental (non-essential) ADL—and, second, essential input from members of multidisciplinary team.
Here is an example to illustrate this point: If we (the multidisciplinary team) believe that a patient who has lived alone and, up to this point, was able to handle his own affairs, will not be safe if he is discharged to his home (based on observation of his overall daily functioning) but he refuses to be institutionalized, we can evaluate his competency and initiate a motion to obtain a temporary guardianship.
If, on the other hand, we think that a patient needs to be placed in a skilled nursing facility and he, being fully aware of his condition, agrees with the decision of the multidisciplinary team, we cannot place him in a facility of our choosing (if it is against his will). Rather, we must give him options of facilities with similar services that meet his needs and let him or his guardian select the facility in which he’s to be placed.
How do we decide on the best course?
To choose what kind of environment a patient needs to be placed in after discharge, we can apply a simple algorithm (Box):
The patient does not need 24-hour protective oversight or needs some, but not 24-hour, care. Discharge him to a home-and community-based care setting—with arrangements for home health agency services or a home-modification program. The patient would either live independently or in a group home setting, depending on how much assistance he requires.
The patient does need 24-hour protective oversight and more than minimal assistance with ADL but doesn’t need 24-hour care, IV medication, etc.). In this case, the patient can be discharged to an assisted living or residential care facility, assuming that he is able to 1) find a so-called path to safety in an emergency (this why facilities are required to perform 1 fire drill per shift per month) and 2) afford rent, because Medicaid, Medicare, and many private insurance policies do not cover housing expenses (see “Keep financing in mind,” in the next section).
The patient is bed-bound or needs 24-hour treatment (eg, receives IV medication or needs total nursing care) or is not bed-bound but is unable to find a path to safety (eg, a person with dementia). This kind of patient must be placed in a skilled nursing facility
Keep financing in mind
The patient’s ability to pay, as well as having access to insurance or a financial assistance program, is a major contributing factor in discharge planning. All financial options need to be considered by the physicians leading the discharge planning team.
Neither Medicaid nor Medicare benefits are available to pay rent; these insurance programs pay for medical services only. Medicaid does provide some money for assistance with ADL in home- and community-based settings (such assistance is arranged through, and provided by, home health care agencies) and to Medicaid-eligible residents of an assisted living facility.
Medicaid covers 100% of a nursing home stay for an eligible resident. Medicare might cover the cost of skilled-nursing facility care if the placement falls under the criterion of an “episode of care.”
It is worth mentioning that some Veterans’ Administration money might be available to a veteran or his (her) surviving spouse for assistance with ADL in home- and community-based settings or if he (she) is institutionalized. Other local programs might provide eligible recipients with long-term care services; discharge social workers, as members of the multidisciplinary team, usually are resourceful at identifying such programs.
All in all, a complex project
Discharge planning is almost as important as the treatment given to the patient. It can be difficult to put all the pieces of the discharge plan together; sometimes, unclear disposition is the only reason a patient is kept in the hospital after being stabilized.
Above all, our ability to work with the multidisciplinary team and our knowledge of these simple steps will help us navigate our patients’ care plan successfully.
Disclosure
Dr. Graypel reports no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
Let me begin with a story.
A few years ago, when I was hoping to get into a psychiatry residency program, I did a month-long rotation in the intensive care unit (ICU) of a local hospital. One of our patients was a long-term resident of a nearby assisted living facility, who was treated for exacerbation of a chronic medical illness. Eventually this patient was stabilized to the point at which he could be discharged.
When the ICU physician decided to discharge this patient, he told the team that the man would need to be “sent back to a nursing home.” The social worker, assuming that the physician wanted to place the patient in a skilled nursing facility, spent several hours trying to place the man in one of the local facilities. When the patient’s daughter arrived to visit her father and began asking questions about why he was being placed in a nursing home, staff immediately realized that the physician had simply meant for him to go back “home”—that is, to the facility from which he had come and where he had been living for several years.
Being the only person in the ICU who was a licensed nursing home administrator, with more than 10 years experience in a long-term care, I should have pointed out this miscommunication or, at least, should have raised the question to clarify the physician’s intent. At the time, however, I wasn’t comfortable expressing my concern because I was “just an FMG observer” trying to stay on the attending’s good side.
I made a commitment to myself, however, to always talk about patients’ long-term care options and discharge planning algorithm with medical students, fellow residents, and other medical professionals I meet in my work. The following is an expression of that commitment.
Why focus on discharge when care is still underway?
Discharge planning usually begins on the first day of hospitalization. Before we are ready to discharge any patient, we, the physicians, usually have had many conversations with members of the multidisciplinary team and, always, with the patient and his (her) guardian(s). Why do we do all of this? The answer is simple: Physicians make the ultimate decision about what kind of environment (clinical, social, etc.) the patient is safe to be discharged to; after that decision is made, everything else is the patient’s choice. Our decision should be based on, first, global assessment of functioning—the ability to perform activities of daily living (ADL) and instrumental (non-essential) ADL—and, second, essential input from members of multidisciplinary team.
Here is an example to illustrate this point: If we (the multidisciplinary team) believe that a patient who has lived alone and, up to this point, was able to handle his own affairs, will not be safe if he is discharged to his home (based on observation of his overall daily functioning) but he refuses to be institutionalized, we can evaluate his competency and initiate a motion to obtain a temporary guardianship.
If, on the other hand, we think that a patient needs to be placed in a skilled nursing facility and he, being fully aware of his condition, agrees with the decision of the multidisciplinary team, we cannot place him in a facility of our choosing (if it is against his will). Rather, we must give him options of facilities with similar services that meet his needs and let him or his guardian select the facility in which he’s to be placed.
How do we decide on the best course?
To choose what kind of environment a patient needs to be placed in after discharge, we can apply a simple algorithm (Box):
The patient does not need 24-hour protective oversight or needs some, but not 24-hour, care. Discharge him to a home-and community-based care setting—with arrangements for home health agency services or a home-modification program. The patient would either live independently or in a group home setting, depending on how much assistance he requires.
The patient does need 24-hour protective oversight and more than minimal assistance with ADL but doesn’t need 24-hour care, IV medication, etc.). In this case, the patient can be discharged to an assisted living or residential care facility, assuming that he is able to 1) find a so-called path to safety in an emergency (this why facilities are required to perform 1 fire drill per shift per month) and 2) afford rent, because Medicaid, Medicare, and many private insurance policies do not cover housing expenses (see “Keep financing in mind,” in the next section).
The patient is bed-bound or needs 24-hour treatment (eg, receives IV medication or needs total nursing care) or is not bed-bound but is unable to find a path to safety (eg, a person with dementia). This kind of patient must be placed in a skilled nursing facility
Keep financing in mind
The patient’s ability to pay, as well as having access to insurance or a financial assistance program, is a major contributing factor in discharge planning. All financial options need to be considered by the physicians leading the discharge planning team.
Neither Medicaid nor Medicare benefits are available to pay rent; these insurance programs pay for medical services only. Medicaid does provide some money for assistance with ADL in home- and community-based settings (such assistance is arranged through, and provided by, home health care agencies) and to Medicaid-eligible residents of an assisted living facility.
Medicaid covers 100% of a nursing home stay for an eligible resident. Medicare might cover the cost of skilled-nursing facility care if the placement falls under the criterion of an “episode of care.”
It is worth mentioning that some Veterans’ Administration money might be available to a veteran or his (her) surviving spouse for assistance with ADL in home- and community-based settings or if he (she) is institutionalized. Other local programs might provide eligible recipients with long-term care services; discharge social workers, as members of the multidisciplinary team, usually are resourceful at identifying such programs.
All in all, a complex project
Discharge planning is almost as important as the treatment given to the patient. It can be difficult to put all the pieces of the discharge plan together; sometimes, unclear disposition is the only reason a patient is kept in the hospital after being stabilized.
Above all, our ability to work with the multidisciplinary team and our knowledge of these simple steps will help us navigate our patients’ care plan successfully.
Disclosure
Dr. Graypel reports no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
Let me begin with a story.
A few years ago, when I was hoping to get into a psychiatry residency program, I did a month-long rotation in the intensive care unit (ICU) of a local hospital. One of our patients was a long-term resident of a nearby assisted living facility, who was treated for exacerbation of a chronic medical illness. Eventually this patient was stabilized to the point at which he could be discharged.
When the ICU physician decided to discharge this patient, he told the team that the man would need to be “sent back to a nursing home.” The social worker, assuming that the physician wanted to place the patient in a skilled nursing facility, spent several hours trying to place the man in one of the local facilities. When the patient’s daughter arrived to visit her father and began asking questions about why he was being placed in a nursing home, staff immediately realized that the physician had simply meant for him to go back “home”—that is, to the facility from which he had come and where he had been living for several years.
Being the only person in the ICU who was a licensed nursing home administrator, with more than 10 years experience in a long-term care, I should have pointed out this miscommunication or, at least, should have raised the question to clarify the physician’s intent. At the time, however, I wasn’t comfortable expressing my concern because I was “just an FMG observer” trying to stay on the attending’s good side.
I made a commitment to myself, however, to always talk about patients’ long-term care options and discharge planning algorithm with medical students, fellow residents, and other medical professionals I meet in my work. The following is an expression of that commitment.
Why focus on discharge when care is still underway?
Discharge planning usually begins on the first day of hospitalization. Before we are ready to discharge any patient, we, the physicians, usually have had many conversations with members of the multidisciplinary team and, always, with the patient and his (her) guardian(s). Why do we do all of this? The answer is simple: Physicians make the ultimate decision about what kind of environment (clinical, social, etc.) the patient is safe to be discharged to; after that decision is made, everything else is the patient’s choice. Our decision should be based on, first, global assessment of functioning—the ability to perform activities of daily living (ADL) and instrumental (non-essential) ADL—and, second, essential input from members of multidisciplinary team.
Here is an example to illustrate this point: If we (the multidisciplinary team) believe that a patient who has lived alone and, up to this point, was able to handle his own affairs, will not be safe if he is discharged to his home (based on observation of his overall daily functioning) but he refuses to be institutionalized, we can evaluate his competency and initiate a motion to obtain a temporary guardianship.
If, on the other hand, we think that a patient needs to be placed in a skilled nursing facility and he, being fully aware of his condition, agrees with the decision of the multidisciplinary team, we cannot place him in a facility of our choosing (if it is against his will). Rather, we must give him options of facilities with similar services that meet his needs and let him or his guardian select the facility in which he’s to be placed.
How do we decide on the best course?
To choose what kind of environment a patient needs to be placed in after discharge, we can apply a simple algorithm (Box):
The patient does not need 24-hour protective oversight or needs some, but not 24-hour, care. Discharge him to a home-and community-based care setting—with arrangements for home health agency services or a home-modification program. The patient would either live independently or in a group home setting, depending on how much assistance he requires.
The patient does need 24-hour protective oversight and more than minimal assistance with ADL but doesn’t need 24-hour care, IV medication, etc.). In this case, the patient can be discharged to an assisted living or residential care facility, assuming that he is able to 1) find a so-called path to safety in an emergency (this why facilities are required to perform 1 fire drill per shift per month) and 2) afford rent, because Medicaid, Medicare, and many private insurance policies do not cover housing expenses (see “Keep financing in mind,” in the next section).
The patient is bed-bound or needs 24-hour treatment (eg, receives IV medication or needs total nursing care) or is not bed-bound but is unable to find a path to safety (eg, a person with dementia). This kind of patient must be placed in a skilled nursing facility
Keep financing in mind
The patient’s ability to pay, as well as having access to insurance or a financial assistance program, is a major contributing factor in discharge planning. All financial options need to be considered by the physicians leading the discharge planning team.
Neither Medicaid nor Medicare benefits are available to pay rent; these insurance programs pay for medical services only. Medicaid does provide some money for assistance with ADL in home- and community-based settings (such assistance is arranged through, and provided by, home health care agencies) and to Medicaid-eligible residents of an assisted living facility.
Medicaid covers 100% of a nursing home stay for an eligible resident. Medicare might cover the cost of skilled-nursing facility care if the placement falls under the criterion of an “episode of care.”
It is worth mentioning that some Veterans’ Administration money might be available to a veteran or his (her) surviving spouse for assistance with ADL in home- and community-based settings or if he (she) is institutionalized. Other local programs might provide eligible recipients with long-term care services; discharge social workers, as members of the multidisciplinary team, usually are resourceful at identifying such programs.
All in all, a complex project
Discharge planning is almost as important as the treatment given to the patient. It can be difficult to put all the pieces of the discharge plan together; sometimes, unclear disposition is the only reason a patient is kept in the hospital after being stabilized.
Above all, our ability to work with the multidisciplinary team and our knowledge of these simple steps will help us navigate our patients’ care plan successfully.
Disclosure
Dr. Graypel reports no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
Learning Dermatopathology in the Digital Age
As in the study of clinical dermatology, establishing a strong fund of knowledge regarding dermatopathology requires visual exposure to countless representative cases. In the not-so-distant past, textbooks relied on grayscale representations to illustrate these diagnoses, but residents today enjoy full-color images; however, textbooks lack the plasticity of digital media, which allow for more immersive interaction with the content. With technological advances in whole-slide imaging, teaching cases can be saved and shared, and rare diagnoses can be studied by individuals who are far removed from the original specimen.1 Even more exciting is that many of the applications (apps) that facilitate digital learning of dermatopathology are available free of charge. In this article, I will review some of the available apps, focusing on their usability, content, and utility as a learning resource for dermatologists at all stages of training. They are discussed in the order of their utility to students of dermatopathology. I have no financial ties to any of the products reviewed, and my recommendations reflect my opinions and observations after real-world use.
Winner: Clearpath
The Clearpath app (http://www.dermpathlab.com/clearpath/) is a fantastic representation of well-executed digital pathology software. Initially released for $50.00 in 2013, the app has since become free while maintaining a steady stream of updates and expanded content. The app is incredibly intuitive and easy to use, made possible by its modern user interface and versatile search function (Figure). For those just beginning to learn dermatopathology, the glossary contains well-written definitions as well as images, which have highlighting that can be toggled on and off to show an area of interest; for instance, if you cannot wrap your mind around the concept of a “grenz zone,” the app can highlight and focus your attention on the respective area in a related image. The app’s library contains more than 250 diagnoses; by clicking on a diagnosis, you are first shown several images displaying features of the pathology identified with highlighting. Then you can study a digital slide as if your tablet was a microscope stage, panning and zooming as you choose. When you are comfortable with the slides, the integrated quiz mode allows for board review with up to 25 answer choices per slide. Although Clearpath’s image-intensive program does require a wireless connection, it also offers the ability to download slides for offline review.
The app has few notable shortcomings related mostly to compatibility, as it is only available for download from the Apple App Store for iPad. Additionally, in comparison to other programs, there is a relative paucity of pathology images to look at, though new diagnoses frequently are added. Regardless, for those with iPads, it is the most refined introduction to a digital dermatopathology product, and a must-have download.
Runner-up: myDermPath
In my February 2014 column,2 I interviewed Dirk M. Elston, MD, and we briefly discussed the myDermPath app (http://mydermpath.com/), which had just recently been made available for free. The myDermPath app excels in the sheer volume of diagnoses it presents—more than 1000 in all—including more unusual pathologic entities. Physicians looking for images of barnyard pox or inflammatory myofibroblastic tumor, for example, do not need to go any further. The pathologic images presented are accompanied by coherent descriptions of clinical features and usually are supplemented with clinical photographs. Furthermore, the app includes a video primer on normal histology narrated by Dr. Elston, a step-by-step algorithm for arriving at a diagnosis, and detailed descriptions of immunofluorescence studies and stains. These additional features make myDermPath a more comprehensive application and a more useful reference source. Its universal compatibility on a range of digital devices makes access to myDermPath convenient for users on any platform (ie, iOS, Android, Web).
The app’s most notable limitation is that, at the time of this writing, it feels somewhat less polished, especially compared to the Clearpath app. This antiquated feel also is evident in the app’s apparent instability on my smartphone, as it frequently stops responding while I am navigating through the menus or looking at histology and often makes it cumbersome to use. This stability issue is not evident on the Web-based version. The app also does not fully support the larger screen sizes of some of the newer smartphones, and therefore the display includes wasted dead space. These faults aside, the volume of material presented and the app’s comprehensive content still make myDermPath a useful addition to your digital dermatopathology repertoire.
Honorable Mention: Derm In-Review
Derm In-Review (http://dermatologyinreview.com/Merz) is sponsored by Merz Pharma and is well known as a broad-reaching resource that reviews the entire breadth of our field for those preparing for in-service or the boards examination. To learn dermatopathology, there are 2 ways to access the digital images: through the Web-based interface and via the mobile app (compatible with iOS and Android). The slides are not categorized but rather are presented in a random order to facilitate quiz taking. The slide images are only photographs of individual features and are not meant to be manipulated as true digital slides; however, the images are good representations of diagnoses, and short descriptions help with learning histologic features. Currently, Aurora Diagnostics (Woodbury, New York) is funding the dermatopathology portion of Derm In-Review, and the online application has already seen a face-lift. With the addition of more content, an updated mobile app, and possibly digital slides, this app will become a more useful tool for learning dermatopathology. Access to Derm In-Review is free with registration on the Web site.
Honorable Mention: Dermpath University
Dermpath University (http://www.dermpathdiagnostics.com/university/digitaldermpath) is a Web-based educational resource of Dermpath Diagnostics that houses a large collection of digital slides. These slides are categorized and can be viewed as unknown cases or with the diagnoses revealed. The images are of high quality and the software is intuitive; however, aside from the diagnosis, slides are not labeled with histologic features or comments about them. The best way to think of this collection is to imagine it is a digital version of the organized slide boxes many residency programs have for teaching purposes. Access to Dermpath University is free with registration on the Web site. Dermpath University also is home to weekly live teledermatology sessions; the schedule can be found on the Web site.
Online Courses: DermpathMD and MDlive
Although structured differently than the other apps described in this article, DermpathMD (http://www.dermpathmd.com) and MDlive (http://www.mdlive.net/dermpath_sch.htm) offer free online dermatopathology courses that are also valuable resources. Rather than discrete apps or digital slides, the courses available from these sources are presented in a lecture-based format to provide overviews on specific topics in dermatopathology. DermpathMD has lectures available as PDFs to download, while MDlive has narrated presentations. Both of these resources are good supplements to a dermatopathology textbook and can be used to obtain a basic foothold on the subject matter before more detailed study.
Conclusion
Learning dermatopathology is no longer done exclusively behind a microscope. The resources presented here bring the experience of learning and reviewing histology slides to your fingertips, sharpening your ability to hone in on the correct features to make an accurate diagnosis. Studying from these digital resources is convenient, comprehensive, and generally free of charge. I hope that you enjoy experimenting with these programs to find a combination that suits your educational needs.
1. Pantanowitz L, Valenstein PN, Evans AJ, et al. Review of the current state of whole slide imaging in pathology. J Pathol Inform. 2011;2:36.
2. Bronfenbrener R. Learning from a leader: an interview with Dirk M. Elston, MD. Cutis. 2014;93:E7-E9.
As in the study of clinical dermatology, establishing a strong fund of knowledge regarding dermatopathology requires visual exposure to countless representative cases. In the not-so-distant past, textbooks relied on grayscale representations to illustrate these diagnoses, but residents today enjoy full-color images; however, textbooks lack the plasticity of digital media, which allow for more immersive interaction with the content. With technological advances in whole-slide imaging, teaching cases can be saved and shared, and rare diagnoses can be studied by individuals who are far removed from the original specimen.1 Even more exciting is that many of the applications (apps) that facilitate digital learning of dermatopathology are available free of charge. In this article, I will review some of the available apps, focusing on their usability, content, and utility as a learning resource for dermatologists at all stages of training. They are discussed in the order of their utility to students of dermatopathology. I have no financial ties to any of the products reviewed, and my recommendations reflect my opinions and observations after real-world use.
Winner: Clearpath
The Clearpath app (http://www.dermpathlab.com/clearpath/) is a fantastic representation of well-executed digital pathology software. Initially released for $50.00 in 2013, the app has since become free while maintaining a steady stream of updates and expanded content. The app is incredibly intuitive and easy to use, made possible by its modern user interface and versatile search function (Figure). For those just beginning to learn dermatopathology, the glossary contains well-written definitions as well as images, which have highlighting that can be toggled on and off to show an area of interest; for instance, if you cannot wrap your mind around the concept of a “grenz zone,” the app can highlight and focus your attention on the respective area in a related image. The app’s library contains more than 250 diagnoses; by clicking on a diagnosis, you are first shown several images displaying features of the pathology identified with highlighting. Then you can study a digital slide as if your tablet was a microscope stage, panning and zooming as you choose. When you are comfortable with the slides, the integrated quiz mode allows for board review with up to 25 answer choices per slide. Although Clearpath’s image-intensive program does require a wireless connection, it also offers the ability to download slides for offline review.
The app has few notable shortcomings related mostly to compatibility, as it is only available for download from the Apple App Store for iPad. Additionally, in comparison to other programs, there is a relative paucity of pathology images to look at, though new diagnoses frequently are added. Regardless, for those with iPads, it is the most refined introduction to a digital dermatopathology product, and a must-have download.
Runner-up: myDermPath
In my February 2014 column,2 I interviewed Dirk M. Elston, MD, and we briefly discussed the myDermPath app (http://mydermpath.com/), which had just recently been made available for free. The myDermPath app excels in the sheer volume of diagnoses it presents—more than 1000 in all—including more unusual pathologic entities. Physicians looking for images of barnyard pox or inflammatory myofibroblastic tumor, for example, do not need to go any further. The pathologic images presented are accompanied by coherent descriptions of clinical features and usually are supplemented with clinical photographs. Furthermore, the app includes a video primer on normal histology narrated by Dr. Elston, a step-by-step algorithm for arriving at a diagnosis, and detailed descriptions of immunofluorescence studies and stains. These additional features make myDermPath a more comprehensive application and a more useful reference source. Its universal compatibility on a range of digital devices makes access to myDermPath convenient for users on any platform (ie, iOS, Android, Web).
The app’s most notable limitation is that, at the time of this writing, it feels somewhat less polished, especially compared to the Clearpath app. This antiquated feel also is evident in the app’s apparent instability on my smartphone, as it frequently stops responding while I am navigating through the menus or looking at histology and often makes it cumbersome to use. This stability issue is not evident on the Web-based version. The app also does not fully support the larger screen sizes of some of the newer smartphones, and therefore the display includes wasted dead space. These faults aside, the volume of material presented and the app’s comprehensive content still make myDermPath a useful addition to your digital dermatopathology repertoire.
Honorable Mention: Derm In-Review
Derm In-Review (http://dermatologyinreview.com/Merz) is sponsored by Merz Pharma and is well known as a broad-reaching resource that reviews the entire breadth of our field for those preparing for in-service or the boards examination. To learn dermatopathology, there are 2 ways to access the digital images: through the Web-based interface and via the mobile app (compatible with iOS and Android). The slides are not categorized but rather are presented in a random order to facilitate quiz taking. The slide images are only photographs of individual features and are not meant to be manipulated as true digital slides; however, the images are good representations of diagnoses, and short descriptions help with learning histologic features. Currently, Aurora Diagnostics (Woodbury, New York) is funding the dermatopathology portion of Derm In-Review, and the online application has already seen a face-lift. With the addition of more content, an updated mobile app, and possibly digital slides, this app will become a more useful tool for learning dermatopathology. Access to Derm In-Review is free with registration on the Web site.
Honorable Mention: Dermpath University
Dermpath University (http://www.dermpathdiagnostics.com/university/digitaldermpath) is a Web-based educational resource of Dermpath Diagnostics that houses a large collection of digital slides. These slides are categorized and can be viewed as unknown cases or with the diagnoses revealed. The images are of high quality and the software is intuitive; however, aside from the diagnosis, slides are not labeled with histologic features or comments about them. The best way to think of this collection is to imagine it is a digital version of the organized slide boxes many residency programs have for teaching purposes. Access to Dermpath University is free with registration on the Web site. Dermpath University also is home to weekly live teledermatology sessions; the schedule can be found on the Web site.
Online Courses: DermpathMD and MDlive
Although structured differently than the other apps described in this article, DermpathMD (http://www.dermpathmd.com) and MDlive (http://www.mdlive.net/dermpath_sch.htm) offer free online dermatopathology courses that are also valuable resources. Rather than discrete apps or digital slides, the courses available from these sources are presented in a lecture-based format to provide overviews on specific topics in dermatopathology. DermpathMD has lectures available as PDFs to download, while MDlive has narrated presentations. Both of these resources are good supplements to a dermatopathology textbook and can be used to obtain a basic foothold on the subject matter before more detailed study.
Conclusion
Learning dermatopathology is no longer done exclusively behind a microscope. The resources presented here bring the experience of learning and reviewing histology slides to your fingertips, sharpening your ability to hone in on the correct features to make an accurate diagnosis. Studying from these digital resources is convenient, comprehensive, and generally free of charge. I hope that you enjoy experimenting with these programs to find a combination that suits your educational needs.
As in the study of clinical dermatology, establishing a strong fund of knowledge regarding dermatopathology requires visual exposure to countless representative cases. In the not-so-distant past, textbooks relied on grayscale representations to illustrate these diagnoses, but residents today enjoy full-color images; however, textbooks lack the plasticity of digital media, which allow for more immersive interaction with the content. With technological advances in whole-slide imaging, teaching cases can be saved and shared, and rare diagnoses can be studied by individuals who are far removed from the original specimen.1 Even more exciting is that many of the applications (apps) that facilitate digital learning of dermatopathology are available free of charge. In this article, I will review some of the available apps, focusing on their usability, content, and utility as a learning resource for dermatologists at all stages of training. They are discussed in the order of their utility to students of dermatopathology. I have no financial ties to any of the products reviewed, and my recommendations reflect my opinions and observations after real-world use.
Winner: Clearpath
The Clearpath app (http://www.dermpathlab.com/clearpath/) is a fantastic representation of well-executed digital pathology software. Initially released for $50.00 in 2013, the app has since become free while maintaining a steady stream of updates and expanded content. The app is incredibly intuitive and easy to use, made possible by its modern user interface and versatile search function (Figure). For those just beginning to learn dermatopathology, the glossary contains well-written definitions as well as images, which have highlighting that can be toggled on and off to show an area of interest; for instance, if you cannot wrap your mind around the concept of a “grenz zone,” the app can highlight and focus your attention on the respective area in a related image. The app’s library contains more than 250 diagnoses; by clicking on a diagnosis, you are first shown several images displaying features of the pathology identified with highlighting. Then you can study a digital slide as if your tablet was a microscope stage, panning and zooming as you choose. When you are comfortable with the slides, the integrated quiz mode allows for board review with up to 25 answer choices per slide. Although Clearpath’s image-intensive program does require a wireless connection, it also offers the ability to download slides for offline review.
The app has few notable shortcomings related mostly to compatibility, as it is only available for download from the Apple App Store for iPad. Additionally, in comparison to other programs, there is a relative paucity of pathology images to look at, though new diagnoses frequently are added. Regardless, for those with iPads, it is the most refined introduction to a digital dermatopathology product, and a must-have download.
Runner-up: myDermPath
In my February 2014 column,2 I interviewed Dirk M. Elston, MD, and we briefly discussed the myDermPath app (http://mydermpath.com/), which had just recently been made available for free. The myDermPath app excels in the sheer volume of diagnoses it presents—more than 1000 in all—including more unusual pathologic entities. Physicians looking for images of barnyard pox or inflammatory myofibroblastic tumor, for example, do not need to go any further. The pathologic images presented are accompanied by coherent descriptions of clinical features and usually are supplemented with clinical photographs. Furthermore, the app includes a video primer on normal histology narrated by Dr. Elston, a step-by-step algorithm for arriving at a diagnosis, and detailed descriptions of immunofluorescence studies and stains. These additional features make myDermPath a more comprehensive application and a more useful reference source. Its universal compatibility on a range of digital devices makes access to myDermPath convenient for users on any platform (ie, iOS, Android, Web).
The app’s most notable limitation is that, at the time of this writing, it feels somewhat less polished, especially compared to the Clearpath app. This antiquated feel also is evident in the app’s apparent instability on my smartphone, as it frequently stops responding while I am navigating through the menus or looking at histology and often makes it cumbersome to use. This stability issue is not evident on the Web-based version. The app also does not fully support the larger screen sizes of some of the newer smartphones, and therefore the display includes wasted dead space. These faults aside, the volume of material presented and the app’s comprehensive content still make myDermPath a useful addition to your digital dermatopathology repertoire.
Honorable Mention: Derm In-Review
Derm In-Review (http://dermatologyinreview.com/Merz) is sponsored by Merz Pharma and is well known as a broad-reaching resource that reviews the entire breadth of our field for those preparing for in-service or the boards examination. To learn dermatopathology, there are 2 ways to access the digital images: through the Web-based interface and via the mobile app (compatible with iOS and Android). The slides are not categorized but rather are presented in a random order to facilitate quiz taking. The slide images are only photographs of individual features and are not meant to be manipulated as true digital slides; however, the images are good representations of diagnoses, and short descriptions help with learning histologic features. Currently, Aurora Diagnostics (Woodbury, New York) is funding the dermatopathology portion of Derm In-Review, and the online application has already seen a face-lift. With the addition of more content, an updated mobile app, and possibly digital slides, this app will become a more useful tool for learning dermatopathology. Access to Derm In-Review is free with registration on the Web site.
Honorable Mention: Dermpath University
Dermpath University (http://www.dermpathdiagnostics.com/university/digitaldermpath) is a Web-based educational resource of Dermpath Diagnostics that houses a large collection of digital slides. These slides are categorized and can be viewed as unknown cases or with the diagnoses revealed. The images are of high quality and the software is intuitive; however, aside from the diagnosis, slides are not labeled with histologic features or comments about them. The best way to think of this collection is to imagine it is a digital version of the organized slide boxes many residency programs have for teaching purposes. Access to Dermpath University is free with registration on the Web site. Dermpath University also is home to weekly live teledermatology sessions; the schedule can be found on the Web site.
Online Courses: DermpathMD and MDlive
Although structured differently than the other apps described in this article, DermpathMD (http://www.dermpathmd.com) and MDlive (http://www.mdlive.net/dermpath_sch.htm) offer free online dermatopathology courses that are also valuable resources. Rather than discrete apps or digital slides, the courses available from these sources are presented in a lecture-based format to provide overviews on specific topics in dermatopathology. DermpathMD has lectures available as PDFs to download, while MDlive has narrated presentations. Both of these resources are good supplements to a dermatopathology textbook and can be used to obtain a basic foothold on the subject matter before more detailed study.
Conclusion
Learning dermatopathology is no longer done exclusively behind a microscope. The resources presented here bring the experience of learning and reviewing histology slides to your fingertips, sharpening your ability to hone in on the correct features to make an accurate diagnosis. Studying from these digital resources is convenient, comprehensive, and generally free of charge. I hope that you enjoy experimenting with these programs to find a combination that suits your educational needs.
1. Pantanowitz L, Valenstein PN, Evans AJ, et al. Review of the current state of whole slide imaging in pathology. J Pathol Inform. 2011;2:36.
2. Bronfenbrener R. Learning from a leader: an interview with Dirk M. Elston, MD. Cutis. 2014;93:E7-E9.
1. Pantanowitz L, Valenstein PN, Evans AJ, et al. Review of the current state of whole slide imaging in pathology. J Pathol Inform. 2011;2:36.
2. Bronfenbrener R. Learning from a leader: an interview with Dirk M. Elston, MD. Cutis. 2014;93:E7-E9.
US Dermatology Residency Program Rankings
Many hospital-based adult specialties are routinely ranked annually by US News & World Report. Its 2014-2015 rankings included cancer; cardiology and heart surgery; diabetes and endocrinology; ear, nose, and throat; gastroenterology and gastrointestinal surgery; geriatrics; gynecology; nephrology; neurology and neurosurgery; ophthalmology; orthopedics; pulmonology; psychiatry; rehabilitation; rheumatology; and urology.1 Only teaching hospitals that saw a high volume of patients were included in the rankings. Ophthalmology, psychiatry, rehabilitation, and rheumatology were ranked based on reputation among specialists; the others were ranked based on objective data (eg, hospital mortality rates, nursing ratios) in addition to a reputation score from a survey of physicians.1
Dermatology has never been included in the annual US News & World Report rankings. In the past, dermatology residency programs have been ranked solely according to the amount of annual funding received from the National Institutes of Health (NIH).2 Wu et al3 expanded the scope by creating an algorithm used to rank dermatology residency programs based on scholarly achievement. Included were a number of NIH grants as well as 4 other factors—publications in 2001-2004, Dermatology Foundation (DF) grants from 2001-2004, faculty lectures in 2004 delivered at national conferences, and number of full-time faculty members who were on the editorial boards of the top 3 US dermatology journals and the top 4 subspecialty journals—deemed important by the authors.3
The current study refines the prior algorithm by creating a weighted ranking algorithm and using criteria that the authors considered to be more meaningful than the original criteria used.3 Specifically, the current study considered the amount of NIH and DF funding received versus the number of grants received, and less importance was given to the number of faculty members on editorial boards and DF funding relative to other criteria. We used publicly available data from Web searches to conduct this study.
Methods
The overall ranking algorithm was designed based on the methodology used by the Institute of Higher Education, Shanghai Jiao Tong University, to rank universities in the annual Academic Ranking of World Universities, which is published annually and uses a weighted ranking algorithm that includes academic and research performance factors to evaluate universities worldwide.4
The names of all dermatology residency programs in the United States were obtained as of December 31, 2008, from FREIDA Online using the search term dermatology; the names of all full-time faculty members at these residency programs and number of residents also were obtained by searching the programs’ Web sites.
For another related study investigating the relationship between residency program characteristics and residents pursuing a career in dermatology, the following data were obtained: total number of full-time faculty members at the program; total number of residents; amount of NIH funding received in 2008 (http://report.nih.gov/award/index.cfm); amount of DF funding received in 2008 (http://dermatologyfoundation.org/pdf/pubs/DF_2008_Annual_Report.pdf.); number of publications by full-time faculty members in 2008 (http://www.ncbi.nlm.nih.gov/pubmed/); number of faculty lectures given at annual meetings of 5 societies in 2008 (American Academy of Dermatology, the Society for Investigative Dermatology, the American Society of Dermatopathology, the Society for Pediatric Dermatology, and the American Society for Dermatologic Surgery); the number of full-time faculty members who were on the editorial boards of 6 dermatology journals (Journal of Investigative Dermatology, Archives of Dermatology [currently known as JAMA Dermatology], Journal of the American Academy of Dermatology, Dermatologic Surgery, Journal of Cutaneous Pathology, and Pediatric Dermatology); and whether a program was housed within an institution’s department of dermatology or division of internal medicine.
The data were summed for all faculty members at a given program. To avoid duplicate faculty publications, collections for each residency program were created within PubMed (ie, if 2 authors from the same program coauthored an article, it was only counted once toward the total number of faculty publications from that program).
The dermatology residency programs that were excluded from this analysis included University of Texas at Austin, University of Texas Medical Branch, and the University of Connecticut, which were started in 2008, as well as Kaiser Permanente Southern California, which was started in 2010. The combined Boston University and Tufts University dermatology residency program was established prior to 2008 and therefore was counted as such in the analysis. The program at Harbor-UCLA Medical Center was not included in the analysis because the program was reestablished in 2004 and we could not assume that the prior program had similar attributes. Military residency programs also were excluded from the analysis, as residents are assigned to faculty positions upon graduation. The NIH residency program also was excluded because it is not a traditional 3-year residency program.
There were 5 factors that were deemed by the authors to be the most reflective of academic achievement among dermatology residency programs: number of faculty publications in 2008; amount of NIH funding received in 2008; number of faculty lectures given at 5 society meetings in 2008; amount of DF funding received in 2008; and the number of faculty members who were on the editorial boards of 6 dermatology journals in 2008. We wished to get a broad range of subspecialties of dermatology (eg, medical, surgical) for the journals. Further, the 6 journals selected were chosen because they had the highest impact factors at the time for general dermatology and dermatopathology.
Each residency program was assigned a score from 0 to 1 for each of these factors. The program with the highest number of faculty publications was assigned a score of 1 and the program with the lowest number of publications was assigned a score of 0. The programs in between were subsequently assigned scores from 0 to 1 based on the number of publications as a percentage of the number of publications from the program with the most publications.
A weighted ranking scheme was used to rank programs based on the relative importance of each factor. The authors decided that NIH funding, number of faculty publications, and number of lectures at society meetings were relatively more important than the other factors; thus these factors were given a weight of 1.0. The remaining factors—DF funding and number of faculty members on editorial boards of journals—were given a weight of 0.5. Values were totaled and programs were ranked based on the sum of these values.
Data were analyzed using SAS 9.2 software. This study was approved by the institutional review board at Kaiser Permanente Southern California.
Results
The overall ranking of the top 20 US dermatology residency programs in 2008 is presented in Table 1. The top 5 programs based on each of the 5 factors used in the weighted ranking algorithm are presented in Tables 2 through 6.
A separate analysis was performed to evaluate the relationship between the ranking factors and the size of the residency programs, as the data seemed to favor larger programs. Table 7 demonstrates that the amount of NIH and DF funding was positively correlated with the number of faculty members in a residency program. The remaining factors were not correlated with the number of faculty in the program.
Due to space considerations, analyses are based on data not shown in this manuscript. Data about the characteristics of each residency program are available from the authors.
Comment
There previously have been few attempts to rank US dermatology residency programs based on factors related to academic achievement. Individual faculty and programs have been ranked based on the number of publications in the literature.5-7 Dermatology institutions/organizations (eg, departments, hospitals, medical schools) have been ranked based on amount of NIH funding received and number of journal citations.8
Dermatology residency programs have been ranked based on amount of NIH funding received annually.2 Based on the prior Wu et al3 ranking algorithm for 2004 data, the institutions with the top 5 residency programs were the University of Pennsylvania, University of California, San Francisco; Yale University; New York University; and the University of Michigan.
The current study refined this ranking algorithm by including residency programs housed within the institution’s division of dermatology of a department of internal medicine, which were previously excluded from NIH funding data, as opposed to just the department of dermatology. The authors also did not count publications coauthored by faculty members at the same institution more than once. The annual amount of DF funding received was considered rather than number of grants, as this factor was thought to better reflect the scale of research being conducted. Additionally, relatively more weight was given to annual NIH funding, annual number of faculty publications, and number of faculty lectures at annual society meetings than to annual amount of DF funding and number of faculty on journal editorial boards. The University of Pennsylvania; the University of California, San Francisco; and Yale University were in the top 5 based on data from 2004 in the prior study3 and 2008 in the current study.
Distribution of grant funding and number of faculty publications in the literature often are used as a measure of scholarly achievement. Within the specialty of dermatology, the amount of NIH and DF funding received could be considered the most prestigious achievement for a residency program. Faculty members are encouraged to develop expertise in a specialized area of dermatology as well as to conduct research and publish articles in the area of their choosing. One of the most common ways to attain tenure and achieve academic recognition is by publishing articles in peer-reviewed journals with high impact factors. Other factors that demonstrate expertise and accomplishment in one’s field include giving lectures at national society meetings and sitting on editorial boards of prestigious journals.
The authors deemed these factors to be the most reflective of academic achievement in a dermatology residency program. It should come as no surprise that the top programs according to our algorithm generally are regarded as the most prestigious programs in the country.
The current study did have some limitations. For instance, residency programs were ranked solely based on academic achievements. Although academic achievement is an important aspect of the reputation of a dermatology residency program, it does not account for other important aspects of a program such as commitment to teaching and patient care. These aspects are difficult to measure and were not included in our ranking algorithm. For this reason, there are many excellent residency programs that may not be listed in this article as top programs but still provide outstanding clinical training and patient care. Our ranking algorithm is more indicative of a program’s commitment to research and scholarship and does not necessarily reflect how well a program trains its residents.
The factors included in our ranking algorithm also were somewhat arbitrary. The 5 factors that were chosen by the authors were considered to be most reflective of academic achievement and also will be easy to obtain in future years to replicate these rankings; however, there are other important factors that could have been used instead or in addition to the factors we chose. Some of the chosen factors were more important than others, so a decision was made to weight the factors. In-training examination scores from the American Board of Dermatology, boards passing rate, or percentage of residents who received fellowships or academic appointments were not used because this information is not publicly available. The current study also appeared to favor larger residency programs. Programs with fewer faculty members generally receive less research funding and have fewer publications, fewer faculty members on journal editorial boards, and fewer lecturers at national society meetings. This factor was not controlled for in the original analysis, as larger programs generally are thought to be more prestigious; therefore, this bias should be accounted for in the rankings.
Conclusion
Based on our weighted ranking algorithm, the top 5 dermatology residency programs in 2008 were University of California, San Francisco; Northwestern University; University of Pennsylvania; Yale University; and Stanford University.
Acknowledgements—We thank all of the program coordinators, full-time faculty members, program directors, chairs, and chiefs who were kind enough to field our questions if we had any missing data about their programs. We thank Mary H. Black, PhD, Pasadena, California, for her contribution to the analytical plan.
1. Best hospitals 2014-2015. US News & World Report. http://health.usnews.com/best-hospitals/rankings. Accessed September 18, 2014.
2. NIH awards for 2005: dermatology departments. ResidentPhysician.com Web site. http://www.resident physician.com/Dermatology_rankings.htm. Accessed September 18, 2014.
3. Wu JJ, Ramirez CC, Alonso CA, et al. Ranking the dermatology programs based on measurements of academic achievement. Dermatol Online J. 2007;13:3.
4. Liu NC, Cheng Y. Academic ranking of world universities—methodologies and problems. High Educ Eur. 2005;30:127-136.
5. Stern RS, Arndt KA. Top cited authors in dermatology: a citation study from 24 journals: 1982-1996. Arch Dermatol. 1999;135:299-302.
6. Stern RS, Arndt KA. Top-cited dermatology authors publishing in 5 “high-impact” general medical journals. Arch Dermatol. 2000;136:357-361.
7. Dermatology: high-impact US institutions. ScienceWatch Web site. http://archive.sciencewatch.com/dr/sci/10/apr25-10_2. Published April 25, 2010. Accessed September 18, 2014.
8. Dubin DB, Arndt KA. Organizational impact in the dermatologic literature. Arch Dermatol. 1996;132:1293-1294.
Many hospital-based adult specialties are routinely ranked annually by US News & World Report. Its 2014-2015 rankings included cancer; cardiology and heart surgery; diabetes and endocrinology; ear, nose, and throat; gastroenterology and gastrointestinal surgery; geriatrics; gynecology; nephrology; neurology and neurosurgery; ophthalmology; orthopedics; pulmonology; psychiatry; rehabilitation; rheumatology; and urology.1 Only teaching hospitals that saw a high volume of patients were included in the rankings. Ophthalmology, psychiatry, rehabilitation, and rheumatology were ranked based on reputation among specialists; the others were ranked based on objective data (eg, hospital mortality rates, nursing ratios) in addition to a reputation score from a survey of physicians.1
Dermatology has never been included in the annual US News & World Report rankings. In the past, dermatology residency programs have been ranked solely according to the amount of annual funding received from the National Institutes of Health (NIH).2 Wu et al3 expanded the scope by creating an algorithm used to rank dermatology residency programs based on scholarly achievement. Included were a number of NIH grants as well as 4 other factors—publications in 2001-2004, Dermatology Foundation (DF) grants from 2001-2004, faculty lectures in 2004 delivered at national conferences, and number of full-time faculty members who were on the editorial boards of the top 3 US dermatology journals and the top 4 subspecialty journals—deemed important by the authors.3
The current study refines the prior algorithm by creating a weighted ranking algorithm and using criteria that the authors considered to be more meaningful than the original criteria used.3 Specifically, the current study considered the amount of NIH and DF funding received versus the number of grants received, and less importance was given to the number of faculty members on editorial boards and DF funding relative to other criteria. We used publicly available data from Web searches to conduct this study.
Methods
The overall ranking algorithm was designed based on the methodology used by the Institute of Higher Education, Shanghai Jiao Tong University, to rank universities in the annual Academic Ranking of World Universities, which is published annually and uses a weighted ranking algorithm that includes academic and research performance factors to evaluate universities worldwide.4
The names of all dermatology residency programs in the United States were obtained as of December 31, 2008, from FREIDA Online using the search term dermatology; the names of all full-time faculty members at these residency programs and number of residents also were obtained by searching the programs’ Web sites.
For another related study investigating the relationship between residency program characteristics and residents pursuing a career in dermatology, the following data were obtained: total number of full-time faculty members at the program; total number of residents; amount of NIH funding received in 2008 (http://report.nih.gov/award/index.cfm); amount of DF funding received in 2008 (http://dermatologyfoundation.org/pdf/pubs/DF_2008_Annual_Report.pdf.); number of publications by full-time faculty members in 2008 (http://www.ncbi.nlm.nih.gov/pubmed/); number of faculty lectures given at annual meetings of 5 societies in 2008 (American Academy of Dermatology, the Society for Investigative Dermatology, the American Society of Dermatopathology, the Society for Pediatric Dermatology, and the American Society for Dermatologic Surgery); the number of full-time faculty members who were on the editorial boards of 6 dermatology journals (Journal of Investigative Dermatology, Archives of Dermatology [currently known as JAMA Dermatology], Journal of the American Academy of Dermatology, Dermatologic Surgery, Journal of Cutaneous Pathology, and Pediatric Dermatology); and whether a program was housed within an institution’s department of dermatology or division of internal medicine.
The data were summed for all faculty members at a given program. To avoid duplicate faculty publications, collections for each residency program were created within PubMed (ie, if 2 authors from the same program coauthored an article, it was only counted once toward the total number of faculty publications from that program).
The dermatology residency programs that were excluded from this analysis included University of Texas at Austin, University of Texas Medical Branch, and the University of Connecticut, which were started in 2008, as well as Kaiser Permanente Southern California, which was started in 2010. The combined Boston University and Tufts University dermatology residency program was established prior to 2008 and therefore was counted as such in the analysis. The program at Harbor-UCLA Medical Center was not included in the analysis because the program was reestablished in 2004 and we could not assume that the prior program had similar attributes. Military residency programs also were excluded from the analysis, as residents are assigned to faculty positions upon graduation. The NIH residency program also was excluded because it is not a traditional 3-year residency program.
There were 5 factors that were deemed by the authors to be the most reflective of academic achievement among dermatology residency programs: number of faculty publications in 2008; amount of NIH funding received in 2008; number of faculty lectures given at 5 society meetings in 2008; amount of DF funding received in 2008; and the number of faculty members who were on the editorial boards of 6 dermatology journals in 2008. We wished to get a broad range of subspecialties of dermatology (eg, medical, surgical) for the journals. Further, the 6 journals selected were chosen because they had the highest impact factors at the time for general dermatology and dermatopathology.
Each residency program was assigned a score from 0 to 1 for each of these factors. The program with the highest number of faculty publications was assigned a score of 1 and the program with the lowest number of publications was assigned a score of 0. The programs in between were subsequently assigned scores from 0 to 1 based on the number of publications as a percentage of the number of publications from the program with the most publications.
A weighted ranking scheme was used to rank programs based on the relative importance of each factor. The authors decided that NIH funding, number of faculty publications, and number of lectures at society meetings were relatively more important than the other factors; thus these factors were given a weight of 1.0. The remaining factors—DF funding and number of faculty members on editorial boards of journals—were given a weight of 0.5. Values were totaled and programs were ranked based on the sum of these values.
Data were analyzed using SAS 9.2 software. This study was approved by the institutional review board at Kaiser Permanente Southern California.
Results
The overall ranking of the top 20 US dermatology residency programs in 2008 is presented in Table 1. The top 5 programs based on each of the 5 factors used in the weighted ranking algorithm are presented in Tables 2 through 6.
A separate analysis was performed to evaluate the relationship between the ranking factors and the size of the residency programs, as the data seemed to favor larger programs. Table 7 demonstrates that the amount of NIH and DF funding was positively correlated with the number of faculty members in a residency program. The remaining factors were not correlated with the number of faculty in the program.
Due to space considerations, analyses are based on data not shown in this manuscript. Data about the characteristics of each residency program are available from the authors.
Comment
There previously have been few attempts to rank US dermatology residency programs based on factors related to academic achievement. Individual faculty and programs have been ranked based on the number of publications in the literature.5-7 Dermatology institutions/organizations (eg, departments, hospitals, medical schools) have been ranked based on amount of NIH funding received and number of journal citations.8
Dermatology residency programs have been ranked based on amount of NIH funding received annually.2 Based on the prior Wu et al3 ranking algorithm for 2004 data, the institutions with the top 5 residency programs were the University of Pennsylvania, University of California, San Francisco; Yale University; New York University; and the University of Michigan.
The current study refined this ranking algorithm by including residency programs housed within the institution’s division of dermatology of a department of internal medicine, which were previously excluded from NIH funding data, as opposed to just the department of dermatology. The authors also did not count publications coauthored by faculty members at the same institution more than once. The annual amount of DF funding received was considered rather than number of grants, as this factor was thought to better reflect the scale of research being conducted. Additionally, relatively more weight was given to annual NIH funding, annual number of faculty publications, and number of faculty lectures at annual society meetings than to annual amount of DF funding and number of faculty on journal editorial boards. The University of Pennsylvania; the University of California, San Francisco; and Yale University were in the top 5 based on data from 2004 in the prior study3 and 2008 in the current study.
Distribution of grant funding and number of faculty publications in the literature often are used as a measure of scholarly achievement. Within the specialty of dermatology, the amount of NIH and DF funding received could be considered the most prestigious achievement for a residency program. Faculty members are encouraged to develop expertise in a specialized area of dermatology as well as to conduct research and publish articles in the area of their choosing. One of the most common ways to attain tenure and achieve academic recognition is by publishing articles in peer-reviewed journals with high impact factors. Other factors that demonstrate expertise and accomplishment in one’s field include giving lectures at national society meetings and sitting on editorial boards of prestigious journals.
The authors deemed these factors to be the most reflective of academic achievement in a dermatology residency program. It should come as no surprise that the top programs according to our algorithm generally are regarded as the most prestigious programs in the country.
The current study did have some limitations. For instance, residency programs were ranked solely based on academic achievements. Although academic achievement is an important aspect of the reputation of a dermatology residency program, it does not account for other important aspects of a program such as commitment to teaching and patient care. These aspects are difficult to measure and were not included in our ranking algorithm. For this reason, there are many excellent residency programs that may not be listed in this article as top programs but still provide outstanding clinical training and patient care. Our ranking algorithm is more indicative of a program’s commitment to research and scholarship and does not necessarily reflect how well a program trains its residents.
The factors included in our ranking algorithm also were somewhat arbitrary. The 5 factors that were chosen by the authors were considered to be most reflective of academic achievement and also will be easy to obtain in future years to replicate these rankings; however, there are other important factors that could have been used instead or in addition to the factors we chose. Some of the chosen factors were more important than others, so a decision was made to weight the factors. In-training examination scores from the American Board of Dermatology, boards passing rate, or percentage of residents who received fellowships or academic appointments were not used because this information is not publicly available. The current study also appeared to favor larger residency programs. Programs with fewer faculty members generally receive less research funding and have fewer publications, fewer faculty members on journal editorial boards, and fewer lecturers at national society meetings. This factor was not controlled for in the original analysis, as larger programs generally are thought to be more prestigious; therefore, this bias should be accounted for in the rankings.
Conclusion
Based on our weighted ranking algorithm, the top 5 dermatology residency programs in 2008 were University of California, San Francisco; Northwestern University; University of Pennsylvania; Yale University; and Stanford University.
Acknowledgements—We thank all of the program coordinators, full-time faculty members, program directors, chairs, and chiefs who were kind enough to field our questions if we had any missing data about their programs. We thank Mary H. Black, PhD, Pasadena, California, for her contribution to the analytical plan.
Many hospital-based adult specialties are routinely ranked annually by US News & World Report. Its 2014-2015 rankings included cancer; cardiology and heart surgery; diabetes and endocrinology; ear, nose, and throat; gastroenterology and gastrointestinal surgery; geriatrics; gynecology; nephrology; neurology and neurosurgery; ophthalmology; orthopedics; pulmonology; psychiatry; rehabilitation; rheumatology; and urology.1 Only teaching hospitals that saw a high volume of patients were included in the rankings. Ophthalmology, psychiatry, rehabilitation, and rheumatology were ranked based on reputation among specialists; the others were ranked based on objective data (eg, hospital mortality rates, nursing ratios) in addition to a reputation score from a survey of physicians.1
Dermatology has never been included in the annual US News & World Report rankings. In the past, dermatology residency programs have been ranked solely according to the amount of annual funding received from the National Institutes of Health (NIH).2 Wu et al3 expanded the scope by creating an algorithm used to rank dermatology residency programs based on scholarly achievement. Included were a number of NIH grants as well as 4 other factors—publications in 2001-2004, Dermatology Foundation (DF) grants from 2001-2004, faculty lectures in 2004 delivered at national conferences, and number of full-time faculty members who were on the editorial boards of the top 3 US dermatology journals and the top 4 subspecialty journals—deemed important by the authors.3
The current study refines the prior algorithm by creating a weighted ranking algorithm and using criteria that the authors considered to be more meaningful than the original criteria used.3 Specifically, the current study considered the amount of NIH and DF funding received versus the number of grants received, and less importance was given to the number of faculty members on editorial boards and DF funding relative to other criteria. We used publicly available data from Web searches to conduct this study.
Methods
The overall ranking algorithm was designed based on the methodology used by the Institute of Higher Education, Shanghai Jiao Tong University, to rank universities in the annual Academic Ranking of World Universities, which is published annually and uses a weighted ranking algorithm that includes academic and research performance factors to evaluate universities worldwide.4
The names of all dermatology residency programs in the United States were obtained as of December 31, 2008, from FREIDA Online using the search term dermatology; the names of all full-time faculty members at these residency programs and number of residents also were obtained by searching the programs’ Web sites.
For another related study investigating the relationship between residency program characteristics and residents pursuing a career in dermatology, the following data were obtained: total number of full-time faculty members at the program; total number of residents; amount of NIH funding received in 2008 (http://report.nih.gov/award/index.cfm); amount of DF funding received in 2008 (http://dermatologyfoundation.org/pdf/pubs/DF_2008_Annual_Report.pdf.); number of publications by full-time faculty members in 2008 (http://www.ncbi.nlm.nih.gov/pubmed/); number of faculty lectures given at annual meetings of 5 societies in 2008 (American Academy of Dermatology, the Society for Investigative Dermatology, the American Society of Dermatopathology, the Society for Pediatric Dermatology, and the American Society for Dermatologic Surgery); the number of full-time faculty members who were on the editorial boards of 6 dermatology journals (Journal of Investigative Dermatology, Archives of Dermatology [currently known as JAMA Dermatology], Journal of the American Academy of Dermatology, Dermatologic Surgery, Journal of Cutaneous Pathology, and Pediatric Dermatology); and whether a program was housed within an institution’s department of dermatology or division of internal medicine.
The data were summed for all faculty members at a given program. To avoid duplicate faculty publications, collections for each residency program were created within PubMed (ie, if 2 authors from the same program coauthored an article, it was only counted once toward the total number of faculty publications from that program).
The dermatology residency programs that were excluded from this analysis included University of Texas at Austin, University of Texas Medical Branch, and the University of Connecticut, which were started in 2008, as well as Kaiser Permanente Southern California, which was started in 2010. The combined Boston University and Tufts University dermatology residency program was established prior to 2008 and therefore was counted as such in the analysis. The program at Harbor-UCLA Medical Center was not included in the analysis because the program was reestablished in 2004 and we could not assume that the prior program had similar attributes. Military residency programs also were excluded from the analysis, as residents are assigned to faculty positions upon graduation. The NIH residency program also was excluded because it is not a traditional 3-year residency program.
There were 5 factors that were deemed by the authors to be the most reflective of academic achievement among dermatology residency programs: number of faculty publications in 2008; amount of NIH funding received in 2008; number of faculty lectures given at 5 society meetings in 2008; amount of DF funding received in 2008; and the number of faculty members who were on the editorial boards of 6 dermatology journals in 2008. We wished to get a broad range of subspecialties of dermatology (eg, medical, surgical) for the journals. Further, the 6 journals selected were chosen because they had the highest impact factors at the time for general dermatology and dermatopathology.
Each residency program was assigned a score from 0 to 1 for each of these factors. The program with the highest number of faculty publications was assigned a score of 1 and the program with the lowest number of publications was assigned a score of 0. The programs in between were subsequently assigned scores from 0 to 1 based on the number of publications as a percentage of the number of publications from the program with the most publications.
A weighted ranking scheme was used to rank programs based on the relative importance of each factor. The authors decided that NIH funding, number of faculty publications, and number of lectures at society meetings were relatively more important than the other factors; thus these factors were given a weight of 1.0. The remaining factors—DF funding and number of faculty members on editorial boards of journals—were given a weight of 0.5. Values were totaled and programs were ranked based on the sum of these values.
Data were analyzed using SAS 9.2 software. This study was approved by the institutional review board at Kaiser Permanente Southern California.
Results
The overall ranking of the top 20 US dermatology residency programs in 2008 is presented in Table 1. The top 5 programs based on each of the 5 factors used in the weighted ranking algorithm are presented in Tables 2 through 6.
A separate analysis was performed to evaluate the relationship between the ranking factors and the size of the residency programs, as the data seemed to favor larger programs. Table 7 demonstrates that the amount of NIH and DF funding was positively correlated with the number of faculty members in a residency program. The remaining factors were not correlated with the number of faculty in the program.
Due to space considerations, analyses are based on data not shown in this manuscript. Data about the characteristics of each residency program are available from the authors.
Comment
There previously have been few attempts to rank US dermatology residency programs based on factors related to academic achievement. Individual faculty and programs have been ranked based on the number of publications in the literature.5-7 Dermatology institutions/organizations (eg, departments, hospitals, medical schools) have been ranked based on amount of NIH funding received and number of journal citations.8
Dermatology residency programs have been ranked based on amount of NIH funding received annually.2 Based on the prior Wu et al3 ranking algorithm for 2004 data, the institutions with the top 5 residency programs were the University of Pennsylvania, University of California, San Francisco; Yale University; New York University; and the University of Michigan.
The current study refined this ranking algorithm by including residency programs housed within the institution’s division of dermatology of a department of internal medicine, which were previously excluded from NIH funding data, as opposed to just the department of dermatology. The authors also did not count publications coauthored by faculty members at the same institution more than once. The annual amount of DF funding received was considered rather than number of grants, as this factor was thought to better reflect the scale of research being conducted. Additionally, relatively more weight was given to annual NIH funding, annual number of faculty publications, and number of faculty lectures at annual society meetings than to annual amount of DF funding and number of faculty on journal editorial boards. The University of Pennsylvania; the University of California, San Francisco; and Yale University were in the top 5 based on data from 2004 in the prior study3 and 2008 in the current study.
Distribution of grant funding and number of faculty publications in the literature often are used as a measure of scholarly achievement. Within the specialty of dermatology, the amount of NIH and DF funding received could be considered the most prestigious achievement for a residency program. Faculty members are encouraged to develop expertise in a specialized area of dermatology as well as to conduct research and publish articles in the area of their choosing. One of the most common ways to attain tenure and achieve academic recognition is by publishing articles in peer-reviewed journals with high impact factors. Other factors that demonstrate expertise and accomplishment in one’s field include giving lectures at national society meetings and sitting on editorial boards of prestigious journals.
The authors deemed these factors to be the most reflective of academic achievement in a dermatology residency program. It should come as no surprise that the top programs according to our algorithm generally are regarded as the most prestigious programs in the country.
The current study did have some limitations. For instance, residency programs were ranked solely based on academic achievements. Although academic achievement is an important aspect of the reputation of a dermatology residency program, it does not account for other important aspects of a program such as commitment to teaching and patient care. These aspects are difficult to measure and were not included in our ranking algorithm. For this reason, there are many excellent residency programs that may not be listed in this article as top programs but still provide outstanding clinical training and patient care. Our ranking algorithm is more indicative of a program’s commitment to research and scholarship and does not necessarily reflect how well a program trains its residents.
The factors included in our ranking algorithm also were somewhat arbitrary. The 5 factors that were chosen by the authors were considered to be most reflective of academic achievement and also will be easy to obtain in future years to replicate these rankings; however, there are other important factors that could have been used instead or in addition to the factors we chose. Some of the chosen factors were more important than others, so a decision was made to weight the factors. In-training examination scores from the American Board of Dermatology, boards passing rate, or percentage of residents who received fellowships or academic appointments were not used because this information is not publicly available. The current study also appeared to favor larger residency programs. Programs with fewer faculty members generally receive less research funding and have fewer publications, fewer faculty members on journal editorial boards, and fewer lecturers at national society meetings. This factor was not controlled for in the original analysis, as larger programs generally are thought to be more prestigious; therefore, this bias should be accounted for in the rankings.
Conclusion
Based on our weighted ranking algorithm, the top 5 dermatology residency programs in 2008 were University of California, San Francisco; Northwestern University; University of Pennsylvania; Yale University; and Stanford University.
Acknowledgements—We thank all of the program coordinators, full-time faculty members, program directors, chairs, and chiefs who were kind enough to field our questions if we had any missing data about their programs. We thank Mary H. Black, PhD, Pasadena, California, for her contribution to the analytical plan.
1. Best hospitals 2014-2015. US News & World Report. http://health.usnews.com/best-hospitals/rankings. Accessed September 18, 2014.
2. NIH awards for 2005: dermatology departments. ResidentPhysician.com Web site. http://www.resident physician.com/Dermatology_rankings.htm. Accessed September 18, 2014.
3. Wu JJ, Ramirez CC, Alonso CA, et al. Ranking the dermatology programs based on measurements of academic achievement. Dermatol Online J. 2007;13:3.
4. Liu NC, Cheng Y. Academic ranking of world universities—methodologies and problems. High Educ Eur. 2005;30:127-136.
5. Stern RS, Arndt KA. Top cited authors in dermatology: a citation study from 24 journals: 1982-1996. Arch Dermatol. 1999;135:299-302.
6. Stern RS, Arndt KA. Top-cited dermatology authors publishing in 5 “high-impact” general medical journals. Arch Dermatol. 2000;136:357-361.
7. Dermatology: high-impact US institutions. ScienceWatch Web site. http://archive.sciencewatch.com/dr/sci/10/apr25-10_2. Published April 25, 2010. Accessed September 18, 2014.
8. Dubin DB, Arndt KA. Organizational impact in the dermatologic literature. Arch Dermatol. 1996;132:1293-1294.
1. Best hospitals 2014-2015. US News & World Report. http://health.usnews.com/best-hospitals/rankings. Accessed September 18, 2014.
2. NIH awards for 2005: dermatology departments. ResidentPhysician.com Web site. http://www.resident physician.com/Dermatology_rankings.htm. Accessed September 18, 2014.
3. Wu JJ, Ramirez CC, Alonso CA, et al. Ranking the dermatology programs based on measurements of academic achievement. Dermatol Online J. 2007;13:3.
4. Liu NC, Cheng Y. Academic ranking of world universities—methodologies and problems. High Educ Eur. 2005;30:127-136.
5. Stern RS, Arndt KA. Top cited authors in dermatology: a citation study from 24 journals: 1982-1996. Arch Dermatol. 1999;135:299-302.
6. Stern RS, Arndt KA. Top-cited dermatology authors publishing in 5 “high-impact” general medical journals. Arch Dermatol. 2000;136:357-361.
7. Dermatology: high-impact US institutions. ScienceWatch Web site. http://archive.sciencewatch.com/dr/sci/10/apr25-10_2. Published April 25, 2010. Accessed September 18, 2014.
8. Dubin DB, Arndt KA. Organizational impact in the dermatologic literature. Arch Dermatol. 1996;132:1293-1294.
Practice Points
- Dermatology is not among the many hospital-based adult specialties that are routinely ranked annually by US News & World Report.
- US dermatology residency programs were ranked based on various academic factors, including annual amount of National Institutes of Health and Dermatology Foundation funding received; number of publications from full-time faculty members; number of faculty lectures given at 5 annual society meetings; and number of full-time faculty members who were on the editorial boards of 6 dermatology journals with the highest impact factors.