User login
International Educational Opportunities for Dermatology Residents
Little has been written about the various types of international educational opportunities in dermatology, which include medical student electives, mentorship and scholarship programs for residents, international residency programs and conferences, and clinical or research-based fellowships (Table). After residency, there are many opportunities for teaching and volunteering in developing countries and ways to get involved in resident education. Although it may be difficult to participate in exchange programs during residency given the demanding schedules and limited vacation and elective time, international exchange programs in dermatology have become more accessible to residents and dermatologists worldwide. This exchange of ideas and information within our field promotes the advancement of scientific and clinical dermatologic insights that may not be commonplace elsewhere.
Resident Opportunities in Europe
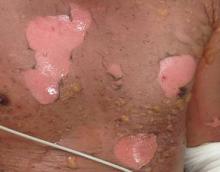
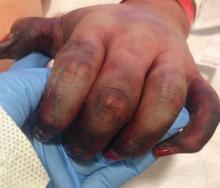
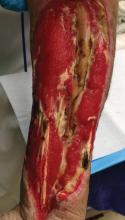
During my internship, I received the Imrich Sarkany Non-European Memorial Scholarship from the European Academy of Dermatology and Venereology, which facilitated my attendance at the organization’s 11th Spring Symposium in Belgrade, Serbia. I participated in the Department of Dermatology at the University of Belgrade, one of the oldest dermatologic programs in Europe, where I learned about Serbian culture, the country’s history as part of Yugoslavia, its health and medical education system, and its unique patient population that includes individuals from various ethnic groups. One of the highlights of my experience was learning about the Belgrade dermatovenereologic moulage collection (Figure), which was developed between 1925 and 1958 after the formation of University of Belgrade’s School of Medicine and the Institute of Dermatovenereology.1 In the early half of the 20th century when photographs were not yet established, the use of moulages as an artistic tool for medical education became very significant. The moulage collection in Belgrade is one of the most well known and is comprised of 350 pieces of which 280 are completely preserved while the rest are damaged.1 These moulages illustrate a wide variety of skin and venereal diseases that have been analyzed and contributed to the historical aspect of dermatologic education and medical conditions that are no longer prevalent thanks to modern medicine. Before World War I when these moulages were created, there was a high incidence of venereal diseases (eg, syphilis) and leprosy. Interestingly, most European dermatology residency programs incorporate the study of sexually transmitted infections and venereology as part of their training. In fact, many European dermatologists also use the term dermatovenereologist to describe their profession. Many of the moulages in the Belgrade collection were made by sculptors and painters, while others were made by physicians themselves, with great authenticity based on comparison of the original diagnosis to current diagnostic criteria.
I also had an opportunity to travel to Kuwait City, Kuwait, for the Kuwait Derma Update and Laser Conference when I was a research fellow at the University of Miami, Florida. In addition to learning opportunities in the form of seminars, workshops, and interactive sessions, each morning began with a visit to one of the government hospitals for a Grand Rounds discussion and presentation of difficult cases for management recommendations. The scientific program was led by Nawaf al-Mutairi, MD, the president of the conference, and involved diverse topics particularly on laser devices and modern therapies for psoriasis and vitiligo in ethnic skin, as well as how to address complications from these therapies. I first learned about platelet-rich plasma treatments and mesotherapy through a workshop at the conference, which became wildly popular in the Middle East and elsewhere during that time.
International Resident Opportunities Through the American Academy of Dermatology
The American Academy of Dermatology (AAD) is dedicated to international education opportunities. The Education and Volunteers Abroad Committee provides 4 grants per year to US dermatology residents who are in their second or third year of residency to participate in a rural health elective in Chinle, Arizona (https://www.aad.org/education/awards-grants-and-scholarships/native-american -health-service-resident-rotation-program). This 1- to 2-week elective takes place at the Native American Health Service where residents provide dermatologic care to the Navajo Nation population and work with primary health care providers to assist with diagnosis and management of dermatologic diseases.
The AAD also provides funding for 15 senior dermatology residents from the United States and Canada to participate in a 4- to 6-week elective in Gaborone, Botswana, which provides opportunities for residents to learn about the care of tropical and human immunodeficiency virus–related dermatologic conditions (https://www.aad.org/education/awards-grants-and-scholarships/resident-international-grant). These programs allow residents to develop image databases, perform teledermatology consultations, and practice in underserved areas with finite resources.
The AAD’s World Congress Fund Review Task Force also offers a limited number of scholarships to US and Canadian dermatology residents, fellows, or young dermatologists within 5 years of dermatology residency to attend the World Congress of Dermatology, the world’s oldest continuous international dermatology meeting, which takes place every 4 years (https://www.aad.org/education/awards-grants-and-scholarships/world-congress-of-dermatology). Grants also are offered for travel to dermatology meetings in Asia, Europe, and Latin America through mutual arrangements with several international dermatologic societies and the International Affairs Committee of the AAD (https://www.aad.org/education/awards-grants-and-scholarships/international-society-annual-meeting-travel-grant). These grants offer participants an opportunity to meet foreign colleagues and establish long-lasting professional relationships.
International Resident Opportunities Through the Women’s Dermatologic Society
The Women’s Dermatologic Society Mentorship Award Program helps to develop long-term mentoring relationships for residents and/or junior faculty that might not otherwise be possible due to distance or funding availability (http://www.womensderm.org/?page=MentorshipAward). As a recipient of this award in 2015, I was paired with Evangeline Handog, MD, current president of the International Society of Dermatology and the dermatology department chairperson at Asian Hospital and Medical Center, Muntinlupa City, Philippines. During my time in the Philippines, I was mostly at the Research Institute for Tropical Medicine, Manila. I also spent a day at the Asian Hospital and Medical Center, a state-of-the-art facility that is accredited by the Joint Commission International, and I attended a cosmetic dermatology workshop led by the Philippine Academy of Dermatologic Surgery Foundation, Inc. During this trip, I was surprised by the number of leprosy and human immunodeficiency virus cases that I encountered. Growing up in the United States for most of my life, I had never seen a case of leprosy. I also was very touched by the hospitality, generosity, and warmth of my mentor Dr. Handog, as well as the professors and dermatology residents that I encountered. The Women’s Dermatologic Society Mentorship Award Program exceeded my expectations, and I learned much more than I could by reading a textbook. I am very grateful for this experience and would do it again if I could.
Final Thoughts
Although it may be challenging to schedule international resident electives and opportunities while in residency training, international educational experiences allow for professional growth and development. An international medical experience can provide excellent opportunities for learning and service in remote or underserved areas in the world.
Acknowledgment
I would like to thank the European Academy of Dermatology and Venereology scholarship committee; Nawaf al-Mutairi, MD, and the Kuwaiti Ministry of Health; the Women’s Dermatologic Society; and the AAD for giving me the chance to participate in these wonderful international opportunities throughout my career.
Reference
1. Medenica L, Lalevic-Vasic B, Skiljevic DS. The Belgrade dermatovenereologic moulage collection: past and present. J Eur Acad Dermatol Venereol. 2008;22:937-942.
Little has been written about the various types of international educational opportunities in dermatology, which include medical student electives, mentorship and scholarship programs for residents, international residency programs and conferences, and clinical or research-based fellowships (Table). After residency, there are many opportunities for teaching and volunteering in developing countries and ways to get involved in resident education. Although it may be difficult to participate in exchange programs during residency given the demanding schedules and limited vacation and elective time, international exchange programs in dermatology have become more accessible to residents and dermatologists worldwide. This exchange of ideas and information within our field promotes the advancement of scientific and clinical dermatologic insights that may not be commonplace elsewhere.
Resident Opportunities in Europe
During my internship, I received the Imrich Sarkany Non-European Memorial Scholarship from the European Academy of Dermatology and Venereology, which facilitated my attendance at the organization’s 11th Spring Symposium in Belgrade, Serbia. I participated in the Department of Dermatology at the University of Belgrade, one of the oldest dermatologic programs in Europe, where I learned about Serbian culture, the country’s history as part of Yugoslavia, its health and medical education system, and its unique patient population that includes individuals from various ethnic groups. One of the highlights of my experience was learning about the Belgrade dermatovenereologic moulage collection (Figure), which was developed between 1925 and 1958 after the formation of University of Belgrade’s School of Medicine and the Institute of Dermatovenereology.1 In the early half of the 20th century when photographs were not yet established, the use of moulages as an artistic tool for medical education became very significant. The moulage collection in Belgrade is one of the most well known and is comprised of 350 pieces of which 280 are completely preserved while the rest are damaged.1 These moulages illustrate a wide variety of skin and venereal diseases that have been analyzed and contributed to the historical aspect of dermatologic education and medical conditions that are no longer prevalent thanks to modern medicine. Before World War I when these moulages were created, there was a high incidence of venereal diseases (eg, syphilis) and leprosy. Interestingly, most European dermatology residency programs incorporate the study of sexually transmitted infections and venereology as part of their training. In fact, many European dermatologists also use the term dermatovenereologist to describe their profession. Many of the moulages in the Belgrade collection were made by sculptors and painters, while others were made by physicians themselves, with great authenticity based on comparison of the original diagnosis to current diagnostic criteria.
I also had an opportunity to travel to Kuwait City, Kuwait, for the Kuwait Derma Update and Laser Conference when I was a research fellow at the University of Miami, Florida. In addition to learning opportunities in the form of seminars, workshops, and interactive sessions, each morning began with a visit to one of the government hospitals for a Grand Rounds discussion and presentation of difficult cases for management recommendations. The scientific program was led by Nawaf al-Mutairi, MD, the president of the conference, and involved diverse topics particularly on laser devices and modern therapies for psoriasis and vitiligo in ethnic skin, as well as how to address complications from these therapies. I first learned about platelet-rich plasma treatments and mesotherapy through a workshop at the conference, which became wildly popular in the Middle East and elsewhere during that time.
International Resident Opportunities Through the American Academy of Dermatology
The American Academy of Dermatology (AAD) is dedicated to international education opportunities. The Education and Volunteers Abroad Committee provides 4 grants per year to US dermatology residents who are in their second or third year of residency to participate in a rural health elective in Chinle, Arizona (https://www.aad.org/education/awards-grants-and-scholarships/native-american -health-service-resident-rotation-program). This 1- to 2-week elective takes place at the Native American Health Service where residents provide dermatologic care to the Navajo Nation population and work with primary health care providers to assist with diagnosis and management of dermatologic diseases.
The AAD also provides funding for 15 senior dermatology residents from the United States and Canada to participate in a 4- to 6-week elective in Gaborone, Botswana, which provides opportunities for residents to learn about the care of tropical and human immunodeficiency virus–related dermatologic conditions (https://www.aad.org/education/awards-grants-and-scholarships/resident-international-grant). These programs allow residents to develop image databases, perform teledermatology consultations, and practice in underserved areas with finite resources.
The AAD’s World Congress Fund Review Task Force also offers a limited number of scholarships to US and Canadian dermatology residents, fellows, or young dermatologists within 5 years of dermatology residency to attend the World Congress of Dermatology, the world’s oldest continuous international dermatology meeting, which takes place every 4 years (https://www.aad.org/education/awards-grants-and-scholarships/world-congress-of-dermatology). Grants also are offered for travel to dermatology meetings in Asia, Europe, and Latin America through mutual arrangements with several international dermatologic societies and the International Affairs Committee of the AAD (https://www.aad.org/education/awards-grants-and-scholarships/international-society-annual-meeting-travel-grant). These grants offer participants an opportunity to meet foreign colleagues and establish long-lasting professional relationships.
International Resident Opportunities Through the Women’s Dermatologic Society
The Women’s Dermatologic Society Mentorship Award Program helps to develop long-term mentoring relationships for residents and/or junior faculty that might not otherwise be possible due to distance or funding availability (http://www.womensderm.org/?page=MentorshipAward). As a recipient of this award in 2015, I was paired with Evangeline Handog, MD, current president of the International Society of Dermatology and the dermatology department chairperson at Asian Hospital and Medical Center, Muntinlupa City, Philippines. During my time in the Philippines, I was mostly at the Research Institute for Tropical Medicine, Manila. I also spent a day at the Asian Hospital and Medical Center, a state-of-the-art facility that is accredited by the Joint Commission International, and I attended a cosmetic dermatology workshop led by the Philippine Academy of Dermatologic Surgery Foundation, Inc. During this trip, I was surprised by the number of leprosy and human immunodeficiency virus cases that I encountered. Growing up in the United States for most of my life, I had never seen a case of leprosy. I also was very touched by the hospitality, generosity, and warmth of my mentor Dr. Handog, as well as the professors and dermatology residents that I encountered. The Women’s Dermatologic Society Mentorship Award Program exceeded my expectations, and I learned much more than I could by reading a textbook. I am very grateful for this experience and would do it again if I could.
Final Thoughts
Although it may be challenging to schedule international resident electives and opportunities while in residency training, international educational experiences allow for professional growth and development. An international medical experience can provide excellent opportunities for learning and service in remote or underserved areas in the world.
Acknowledgment
I would like to thank the European Academy of Dermatology and Venereology scholarship committee; Nawaf al-Mutairi, MD, and the Kuwaiti Ministry of Health; the Women’s Dermatologic Society; and the AAD for giving me the chance to participate in these wonderful international opportunities throughout my career.
Little has been written about the various types of international educational opportunities in dermatology, which include medical student electives, mentorship and scholarship programs for residents, international residency programs and conferences, and clinical or research-based fellowships (Table). After residency, there are many opportunities for teaching and volunteering in developing countries and ways to get involved in resident education. Although it may be difficult to participate in exchange programs during residency given the demanding schedules and limited vacation and elective time, international exchange programs in dermatology have become more accessible to residents and dermatologists worldwide. This exchange of ideas and information within our field promotes the advancement of scientific and clinical dermatologic insights that may not be commonplace elsewhere.
Resident Opportunities in Europe
During my internship, I received the Imrich Sarkany Non-European Memorial Scholarship from the European Academy of Dermatology and Venereology, which facilitated my attendance at the organization’s 11th Spring Symposium in Belgrade, Serbia. I participated in the Department of Dermatology at the University of Belgrade, one of the oldest dermatologic programs in Europe, where I learned about Serbian culture, the country’s history as part of Yugoslavia, its health and medical education system, and its unique patient population that includes individuals from various ethnic groups. One of the highlights of my experience was learning about the Belgrade dermatovenereologic moulage collection (Figure), which was developed between 1925 and 1958 after the formation of University of Belgrade’s School of Medicine and the Institute of Dermatovenereology.1 In the early half of the 20th century when photographs were not yet established, the use of moulages as an artistic tool for medical education became very significant. The moulage collection in Belgrade is one of the most well known and is comprised of 350 pieces of which 280 are completely preserved while the rest are damaged.1 These moulages illustrate a wide variety of skin and venereal diseases that have been analyzed and contributed to the historical aspect of dermatologic education and medical conditions that are no longer prevalent thanks to modern medicine. Before World War I when these moulages were created, there was a high incidence of venereal diseases (eg, syphilis) and leprosy. Interestingly, most European dermatology residency programs incorporate the study of sexually transmitted infections and venereology as part of their training. In fact, many European dermatologists also use the term dermatovenereologist to describe their profession. Many of the moulages in the Belgrade collection were made by sculptors and painters, while others were made by physicians themselves, with great authenticity based on comparison of the original diagnosis to current diagnostic criteria.
I also had an opportunity to travel to Kuwait City, Kuwait, for the Kuwait Derma Update and Laser Conference when I was a research fellow at the University of Miami, Florida. In addition to learning opportunities in the form of seminars, workshops, and interactive sessions, each morning began with a visit to one of the government hospitals for a Grand Rounds discussion and presentation of difficult cases for management recommendations. The scientific program was led by Nawaf al-Mutairi, MD, the president of the conference, and involved diverse topics particularly on laser devices and modern therapies for psoriasis and vitiligo in ethnic skin, as well as how to address complications from these therapies. I first learned about platelet-rich plasma treatments and mesotherapy through a workshop at the conference, which became wildly popular in the Middle East and elsewhere during that time.
International Resident Opportunities Through the American Academy of Dermatology
The American Academy of Dermatology (AAD) is dedicated to international education opportunities. The Education and Volunteers Abroad Committee provides 4 grants per year to US dermatology residents who are in their second or third year of residency to participate in a rural health elective in Chinle, Arizona (https://www.aad.org/education/awards-grants-and-scholarships/native-american -health-service-resident-rotation-program). This 1- to 2-week elective takes place at the Native American Health Service where residents provide dermatologic care to the Navajo Nation population and work with primary health care providers to assist with diagnosis and management of dermatologic diseases.
The AAD also provides funding for 15 senior dermatology residents from the United States and Canada to participate in a 4- to 6-week elective in Gaborone, Botswana, which provides opportunities for residents to learn about the care of tropical and human immunodeficiency virus–related dermatologic conditions (https://www.aad.org/education/awards-grants-and-scholarships/resident-international-grant). These programs allow residents to develop image databases, perform teledermatology consultations, and practice in underserved areas with finite resources.
The AAD’s World Congress Fund Review Task Force also offers a limited number of scholarships to US and Canadian dermatology residents, fellows, or young dermatologists within 5 years of dermatology residency to attend the World Congress of Dermatology, the world’s oldest continuous international dermatology meeting, which takes place every 4 years (https://www.aad.org/education/awards-grants-and-scholarships/world-congress-of-dermatology). Grants also are offered for travel to dermatology meetings in Asia, Europe, and Latin America through mutual arrangements with several international dermatologic societies and the International Affairs Committee of the AAD (https://www.aad.org/education/awards-grants-and-scholarships/international-society-annual-meeting-travel-grant). These grants offer participants an opportunity to meet foreign colleagues and establish long-lasting professional relationships.
International Resident Opportunities Through the Women’s Dermatologic Society
The Women’s Dermatologic Society Mentorship Award Program helps to develop long-term mentoring relationships for residents and/or junior faculty that might not otherwise be possible due to distance or funding availability (http://www.womensderm.org/?page=MentorshipAward). As a recipient of this award in 2015, I was paired with Evangeline Handog, MD, current president of the International Society of Dermatology and the dermatology department chairperson at Asian Hospital and Medical Center, Muntinlupa City, Philippines. During my time in the Philippines, I was mostly at the Research Institute for Tropical Medicine, Manila. I also spent a day at the Asian Hospital and Medical Center, a state-of-the-art facility that is accredited by the Joint Commission International, and I attended a cosmetic dermatology workshop led by the Philippine Academy of Dermatologic Surgery Foundation, Inc. During this trip, I was surprised by the number of leprosy and human immunodeficiency virus cases that I encountered. Growing up in the United States for most of my life, I had never seen a case of leprosy. I also was very touched by the hospitality, generosity, and warmth of my mentor Dr. Handog, as well as the professors and dermatology residents that I encountered. The Women’s Dermatologic Society Mentorship Award Program exceeded my expectations, and I learned much more than I could by reading a textbook. I am very grateful for this experience and would do it again if I could.
Final Thoughts
Although it may be challenging to schedule international resident electives and opportunities while in residency training, international educational experiences allow for professional growth and development. An international medical experience can provide excellent opportunities for learning and service in remote or underserved areas in the world.
Acknowledgment
I would like to thank the European Academy of Dermatology and Venereology scholarship committee; Nawaf al-Mutairi, MD, and the Kuwaiti Ministry of Health; the Women’s Dermatologic Society; and the AAD for giving me the chance to participate in these wonderful international opportunities throughout my career.
Reference
1. Medenica L, Lalevic-Vasic B, Skiljevic DS. The Belgrade dermatovenereologic moulage collection: past and present. J Eur Acad Dermatol Venereol. 2008;22:937-942.
Reference
1. Medenica L, Lalevic-Vasic B, Skiljevic DS. The Belgrade dermatovenereologic moulage collection: past and present. J Eur Acad Dermatol Venereol. 2008;22:937-942.
Orthopedic Residents: What Are We Worth?
The modern age of health care has driven a national interest in quality, health care economics, and proving value. A commonly used definition for quality is value/cost. Defining the value of orthopedic residents is difficult. With changes in the delivery of health care, the implementation of the Affordable Care Act, and an increasing federal deficit, defining the value of orthopedic residents has never been more important.1
Funding for graduate medical education (GME) has been a source of recent intense debate.2-8 From the inception of Medicare and Medicaid services, the value of residents has been recognized, and funding has been provided for resident and fellowship education. In 2012, public tax dollars provided more than $15 billion towards GME, with more than 90% coming from the Center of Medicare and Medicaid Services (CMS).4 This funding was initially established to:
- support the education of physicians
- provide well-trained physicians for future generations
- account for a disproportion of care provided to underfunded patients at teaching hospitals
- account for specialty services (eg, burn centers, trauma centers, emergency psychiatric services) that can be net revenue negative.
The significant cost of these programs, which are almost exclusively government-funded, has been the subject of cost-cutting discussions in Congress since the Balanced Budget Act of 1997 that froze GME funding.9 More recently, the National Commission on Fiscal Responsibility and Reform report authored by the Bowles-Simpson Commission proposed decreases in both direct medical education (DME) and indirect medical education (IME) payments that could total $6 billion by 2015 and $60 billion by 2020.4,7,8 The proposed cuts come on the heels of the Affordable Care Act and the projected significant increase in health care demand.1 It is important to note that private payers do not support GME despite receiving health care provided by residents and fellows.
Despite a track record of producing well-trained and skilled physicians at the end of GME training, several reports from both the public and private sectors have identified weaknesses in the GME system. These include a mismatch between the specialty composition of physician trainees and the population needs, geographic maldistribution of the physician workforce, and a lack of fiscal transparency of GME fund use by hospitals.2 A recent comprehensive report from the Institute of Medicine (IOM) entitled Graduate Medical Education that Meets the Nation’s Health Needs highlights the current issues surrounding GME funding.2 The report made note of several important problems with the current GME system, including:
- The revenue impact and cost savings associated with sponsoring residents are neither tracked nor reported, and they are rarely acknowledged in analyses of GME costs.
- In 1997, Congress capped the number of Medicare-supported physician training slots. Hospitals may add residents beyond the cap but cannot receive additional Medicare payments for those trainees. The cap is equal to each hospital’s number of residents in 1996—essentially freezing the geographic distribution of Medicare-supported residencies without regard for future changes in local or regional health workforce priorities or the geography and demography of the US population.9
- By distributing funds directly to teaching hospitals, the Medicare payment system discourages physician training outside the hospital, in clinical settings where most health care is delivered.
- Because Medicare GME funding is formula-driven, the payments are essentially guaranteed regardless of whether the funded trainees reflect local, national, or regional health needs.
- The system’s only mechanism for ensuring accountability is the requirement that residency programs be accredited. The system does not yield useful data on program outcomes and performance.
- Despite receiving government support for their residency or fellowship training, the graduate physician has no obligation to return this government investment through service.
Some of the IOM’s proposed changes to the system include:
- Updating the GME funding to account for inflation and make GME payments based on accountable performance.
- Phasing out the current GME payment system.
- Specifying funds for “transformational” programs that promote innovation and planning for the future.
- Analyzing and redistributing GME funds based on current population needs and performance metrics.
- Increasing fiscal transparency of the CMS payments and their use by hospitals.
- Establishing a GME center within CMS for ongoing oversight.
The American Orthopaedic Association recently held a forum on GME funding for resident education.10 At that forum, departmental leaders noted the difficulty in securing additional funded resident spots from hospitals and the difficulty in proving residents’ value to the hospital administration. Some in the forum suggested that, in the future, residents may need to pay for their residency like they pay for medical school.
There is very limited published data on the financial value added by orthopedic residents. A recent study examined the service provided by a single junior resident during 2 years of primary orthopedic call.11 They found that the potentially billable services provided by the resident totaled more than $79,000 per year. This only accounted for services delivered while on call every sixth night. This did not account for any surgical assisting or outpatient clinic support.11 This amount is nearly twice the amount provided in DME funds to the hospital for resident support.
Although the care and service that residents deliver is obvious to orthopedic attending physicians, we must “prove” our value through continued research and reporting of services provided by residents. If we do not demonstrate our value to the funders of GME, the government, and Congress, I worry that residents who follow behind us may have to fund their own training. An additional concern is that the current shortage of orthopedic surgeons may be worsened if GME funding is decreased.12 This shortage will be exacerbated by the aging population’s increased need for orthopedic care.13
As health care goes through dramatic changes, orthopedic residents and attending surgeons need to be engaged in the discussion so that we can help shape our future in a way that meets the needs of our patients and continues to allow orthopedic care to be delivered at a high level nationally.
1. Sommers BD, Swartz K, Epstein A. Policy makers should prepare for major uncertainties in Medicaid enrollment, costs, and needs for physicians under health reform. Health Aff (Millwood). 2011;30(11):2186-2193.
2. Eden J, Berwick D, Wilensky G, eds. Graduate Medical Education That Meets the Nation’s Health Needs. Washington, DC: National Academies Press; 2014.
3. Chandra A, Khullar D, Wilensky GR. The economics of graduate medical education. N Engl J Med. 2014;370(25):2357-2360.
4. Iglehart JK. The uncertain future of Medicare and graduate medical education. N Engl J Med. 2011;365(14):1340-1345.
5. Nuckols TK, Bhattacharya J, Wolman DM, Ulmer C, Escarce JJ. Cost implications of reduced work hours and workloads for resident physicians. N Engl J Med. 2009;360(21):2202-2215.
6. Robbins RA, Singarajah CU. IOM releases report on graduate medical education. Southwest J Pulm Crit Care. 2014;9(2):123-125. http://www.swjpcc.com/editorial/2014/8/12/iom-releases-report-on-graduate-medical-education.html. Published August 12, 2014. Accessed March 5, 2015.
7. Iglehart JK. Financing graduate medical education—mounting pressure for reform. N Engl J Med. 2012;366(17):1562-1563.
8. The National Commission on Fiscal Responsibility and Reform. The Moment of Truth: Report of the National Commission on Fiscal Responsibility and Reform. The National Commission on Fiscal Responsibility and Reform website. http://www.fiscalcommission.gov/sites/fiscalcommission.gov/files/documents/TheMomentofTruth12_1_2010.pdf. Published December 2010. Accessed March 5, 2015.
9. Balanced Budget Act of 1997, Pub L No. 105-33, 111 Stat 251.
10. Weinstein S. Departmental Leadship Forum III—Orthopaedic workforce needs: challenges in a changing enviroment. Final Program: 2014 Combined Meeting of The American Orthopaedic Association and the Canadian Orthopaedic Association. American Orthopaedic Association website. https://www.aoassn.org/media/410447/final-program-with-covers.pdf. Published June 2014. Accessed March 5, 2015.
11. Jackson JB, Huntington WP, Frick SL. Assessing the value of work done by an orthopedic resident during call. J Grad Med Educ. 2014;6(3):567-570.
12. Frick SL, Richards BS, Weinstein SL, Beaty JH, Wattenbarger JM. Workforce, work, and advocacy issues in pediatric orthopaedics. J Bone Joint Surg Am. 2010;92(17):e31.
13. Physician supply and demand through 2025: key findings. Association of American Medical Colleges website. https://www.aamc.org/download/153160/data/physician_shortages_to_worsen_without_increases_in_residency_tr.pdf. Published March 3, 2015. Accessed March 5, 2015.
The modern age of health care has driven a national interest in quality, health care economics, and proving value. A commonly used definition for quality is value/cost. Defining the value of orthopedic residents is difficult. With changes in the delivery of health care, the implementation of the Affordable Care Act, and an increasing federal deficit, defining the value of orthopedic residents has never been more important.1
Funding for graduate medical education (GME) has been a source of recent intense debate.2-8 From the inception of Medicare and Medicaid services, the value of residents has been recognized, and funding has been provided for resident and fellowship education. In 2012, public tax dollars provided more than $15 billion towards GME, with more than 90% coming from the Center of Medicare and Medicaid Services (CMS).4 This funding was initially established to:
- support the education of physicians
- provide well-trained physicians for future generations
- account for a disproportion of care provided to underfunded patients at teaching hospitals
- account for specialty services (eg, burn centers, trauma centers, emergency psychiatric services) that can be net revenue negative.
The significant cost of these programs, which are almost exclusively government-funded, has been the subject of cost-cutting discussions in Congress since the Balanced Budget Act of 1997 that froze GME funding.9 More recently, the National Commission on Fiscal Responsibility and Reform report authored by the Bowles-Simpson Commission proposed decreases in both direct medical education (DME) and indirect medical education (IME) payments that could total $6 billion by 2015 and $60 billion by 2020.4,7,8 The proposed cuts come on the heels of the Affordable Care Act and the projected significant increase in health care demand.1 It is important to note that private payers do not support GME despite receiving health care provided by residents and fellows.
Despite a track record of producing well-trained and skilled physicians at the end of GME training, several reports from both the public and private sectors have identified weaknesses in the GME system. These include a mismatch between the specialty composition of physician trainees and the population needs, geographic maldistribution of the physician workforce, and a lack of fiscal transparency of GME fund use by hospitals.2 A recent comprehensive report from the Institute of Medicine (IOM) entitled Graduate Medical Education that Meets the Nation’s Health Needs highlights the current issues surrounding GME funding.2 The report made note of several important problems with the current GME system, including:
- The revenue impact and cost savings associated with sponsoring residents are neither tracked nor reported, and they are rarely acknowledged in analyses of GME costs.
- In 1997, Congress capped the number of Medicare-supported physician training slots. Hospitals may add residents beyond the cap but cannot receive additional Medicare payments for those trainees. The cap is equal to each hospital’s number of residents in 1996—essentially freezing the geographic distribution of Medicare-supported residencies without regard for future changes in local or regional health workforce priorities or the geography and demography of the US population.9
- By distributing funds directly to teaching hospitals, the Medicare payment system discourages physician training outside the hospital, in clinical settings where most health care is delivered.
- Because Medicare GME funding is formula-driven, the payments are essentially guaranteed regardless of whether the funded trainees reflect local, national, or regional health needs.
- The system’s only mechanism for ensuring accountability is the requirement that residency programs be accredited. The system does not yield useful data on program outcomes and performance.
- Despite receiving government support for their residency or fellowship training, the graduate physician has no obligation to return this government investment through service.
Some of the IOM’s proposed changes to the system include:
- Updating the GME funding to account for inflation and make GME payments based on accountable performance.
- Phasing out the current GME payment system.
- Specifying funds for “transformational” programs that promote innovation and planning for the future.
- Analyzing and redistributing GME funds based on current population needs and performance metrics.
- Increasing fiscal transparency of the CMS payments and their use by hospitals.
- Establishing a GME center within CMS for ongoing oversight.
The American Orthopaedic Association recently held a forum on GME funding for resident education.10 At that forum, departmental leaders noted the difficulty in securing additional funded resident spots from hospitals and the difficulty in proving residents’ value to the hospital administration. Some in the forum suggested that, in the future, residents may need to pay for their residency like they pay for medical school.
There is very limited published data on the financial value added by orthopedic residents. A recent study examined the service provided by a single junior resident during 2 years of primary orthopedic call.11 They found that the potentially billable services provided by the resident totaled more than $79,000 per year. This only accounted for services delivered while on call every sixth night. This did not account for any surgical assisting or outpatient clinic support.11 This amount is nearly twice the amount provided in DME funds to the hospital for resident support.
Although the care and service that residents deliver is obvious to orthopedic attending physicians, we must “prove” our value through continued research and reporting of services provided by residents. If we do not demonstrate our value to the funders of GME, the government, and Congress, I worry that residents who follow behind us may have to fund their own training. An additional concern is that the current shortage of orthopedic surgeons may be worsened if GME funding is decreased.12 This shortage will be exacerbated by the aging population’s increased need for orthopedic care.13
As health care goes through dramatic changes, orthopedic residents and attending surgeons need to be engaged in the discussion so that we can help shape our future in a way that meets the needs of our patients and continues to allow orthopedic care to be delivered at a high level nationally.
The modern age of health care has driven a national interest in quality, health care economics, and proving value. A commonly used definition for quality is value/cost. Defining the value of orthopedic residents is difficult. With changes in the delivery of health care, the implementation of the Affordable Care Act, and an increasing federal deficit, defining the value of orthopedic residents has never been more important.1
Funding for graduate medical education (GME) has been a source of recent intense debate.2-8 From the inception of Medicare and Medicaid services, the value of residents has been recognized, and funding has been provided for resident and fellowship education. In 2012, public tax dollars provided more than $15 billion towards GME, with more than 90% coming from the Center of Medicare and Medicaid Services (CMS).4 This funding was initially established to:
- support the education of physicians
- provide well-trained physicians for future generations
- account for a disproportion of care provided to underfunded patients at teaching hospitals
- account for specialty services (eg, burn centers, trauma centers, emergency psychiatric services) that can be net revenue negative.
The significant cost of these programs, which are almost exclusively government-funded, has been the subject of cost-cutting discussions in Congress since the Balanced Budget Act of 1997 that froze GME funding.9 More recently, the National Commission on Fiscal Responsibility and Reform report authored by the Bowles-Simpson Commission proposed decreases in both direct medical education (DME) and indirect medical education (IME) payments that could total $6 billion by 2015 and $60 billion by 2020.4,7,8 The proposed cuts come on the heels of the Affordable Care Act and the projected significant increase in health care demand.1 It is important to note that private payers do not support GME despite receiving health care provided by residents and fellows.
Despite a track record of producing well-trained and skilled physicians at the end of GME training, several reports from both the public and private sectors have identified weaknesses in the GME system. These include a mismatch between the specialty composition of physician trainees and the population needs, geographic maldistribution of the physician workforce, and a lack of fiscal transparency of GME fund use by hospitals.2 A recent comprehensive report from the Institute of Medicine (IOM) entitled Graduate Medical Education that Meets the Nation’s Health Needs highlights the current issues surrounding GME funding.2 The report made note of several important problems with the current GME system, including:
- The revenue impact and cost savings associated with sponsoring residents are neither tracked nor reported, and they are rarely acknowledged in analyses of GME costs.
- In 1997, Congress capped the number of Medicare-supported physician training slots. Hospitals may add residents beyond the cap but cannot receive additional Medicare payments for those trainees. The cap is equal to each hospital’s number of residents in 1996—essentially freezing the geographic distribution of Medicare-supported residencies without regard for future changes in local or regional health workforce priorities or the geography and demography of the US population.9
- By distributing funds directly to teaching hospitals, the Medicare payment system discourages physician training outside the hospital, in clinical settings where most health care is delivered.
- Because Medicare GME funding is formula-driven, the payments are essentially guaranteed regardless of whether the funded trainees reflect local, national, or regional health needs.
- The system’s only mechanism for ensuring accountability is the requirement that residency programs be accredited. The system does not yield useful data on program outcomes and performance.
- Despite receiving government support for their residency or fellowship training, the graduate physician has no obligation to return this government investment through service.
Some of the IOM’s proposed changes to the system include:
- Updating the GME funding to account for inflation and make GME payments based on accountable performance.
- Phasing out the current GME payment system.
- Specifying funds for “transformational” programs that promote innovation and planning for the future.
- Analyzing and redistributing GME funds based on current population needs and performance metrics.
- Increasing fiscal transparency of the CMS payments and their use by hospitals.
- Establishing a GME center within CMS for ongoing oversight.
The American Orthopaedic Association recently held a forum on GME funding for resident education.10 At that forum, departmental leaders noted the difficulty in securing additional funded resident spots from hospitals and the difficulty in proving residents’ value to the hospital administration. Some in the forum suggested that, in the future, residents may need to pay for their residency like they pay for medical school.
There is very limited published data on the financial value added by orthopedic residents. A recent study examined the service provided by a single junior resident during 2 years of primary orthopedic call.11 They found that the potentially billable services provided by the resident totaled more than $79,000 per year. This only accounted for services delivered while on call every sixth night. This did not account for any surgical assisting or outpatient clinic support.11 This amount is nearly twice the amount provided in DME funds to the hospital for resident support.
Although the care and service that residents deliver is obvious to orthopedic attending physicians, we must “prove” our value through continued research and reporting of services provided by residents. If we do not demonstrate our value to the funders of GME, the government, and Congress, I worry that residents who follow behind us may have to fund their own training. An additional concern is that the current shortage of orthopedic surgeons may be worsened if GME funding is decreased.12 This shortage will be exacerbated by the aging population’s increased need for orthopedic care.13
As health care goes through dramatic changes, orthopedic residents and attending surgeons need to be engaged in the discussion so that we can help shape our future in a way that meets the needs of our patients and continues to allow orthopedic care to be delivered at a high level nationally.
1. Sommers BD, Swartz K, Epstein A. Policy makers should prepare for major uncertainties in Medicaid enrollment, costs, and needs for physicians under health reform. Health Aff (Millwood). 2011;30(11):2186-2193.
2. Eden J, Berwick D, Wilensky G, eds. Graduate Medical Education That Meets the Nation’s Health Needs. Washington, DC: National Academies Press; 2014.
3. Chandra A, Khullar D, Wilensky GR. The economics of graduate medical education. N Engl J Med. 2014;370(25):2357-2360.
4. Iglehart JK. The uncertain future of Medicare and graduate medical education. N Engl J Med. 2011;365(14):1340-1345.
5. Nuckols TK, Bhattacharya J, Wolman DM, Ulmer C, Escarce JJ. Cost implications of reduced work hours and workloads for resident physicians. N Engl J Med. 2009;360(21):2202-2215.
6. Robbins RA, Singarajah CU. IOM releases report on graduate medical education. Southwest J Pulm Crit Care. 2014;9(2):123-125. http://www.swjpcc.com/editorial/2014/8/12/iom-releases-report-on-graduate-medical-education.html. Published August 12, 2014. Accessed March 5, 2015.
7. Iglehart JK. Financing graduate medical education—mounting pressure for reform. N Engl J Med. 2012;366(17):1562-1563.
8. The National Commission on Fiscal Responsibility and Reform. The Moment of Truth: Report of the National Commission on Fiscal Responsibility and Reform. The National Commission on Fiscal Responsibility and Reform website. http://www.fiscalcommission.gov/sites/fiscalcommission.gov/files/documents/TheMomentofTruth12_1_2010.pdf. Published December 2010. Accessed March 5, 2015.
9. Balanced Budget Act of 1997, Pub L No. 105-33, 111 Stat 251.
10. Weinstein S. Departmental Leadship Forum III—Orthopaedic workforce needs: challenges in a changing enviroment. Final Program: 2014 Combined Meeting of The American Orthopaedic Association and the Canadian Orthopaedic Association. American Orthopaedic Association website. https://www.aoassn.org/media/410447/final-program-with-covers.pdf. Published June 2014. Accessed March 5, 2015.
11. Jackson JB, Huntington WP, Frick SL. Assessing the value of work done by an orthopedic resident during call. J Grad Med Educ. 2014;6(3):567-570.
12. Frick SL, Richards BS, Weinstein SL, Beaty JH, Wattenbarger JM. Workforce, work, and advocacy issues in pediatric orthopaedics. J Bone Joint Surg Am. 2010;92(17):e31.
13. Physician supply and demand through 2025: key findings. Association of American Medical Colleges website. https://www.aamc.org/download/153160/data/physician_shortages_to_worsen_without_increases_in_residency_tr.pdf. Published March 3, 2015. Accessed March 5, 2015.
1. Sommers BD, Swartz K, Epstein A. Policy makers should prepare for major uncertainties in Medicaid enrollment, costs, and needs for physicians under health reform. Health Aff (Millwood). 2011;30(11):2186-2193.
2. Eden J, Berwick D, Wilensky G, eds. Graduate Medical Education That Meets the Nation’s Health Needs. Washington, DC: National Academies Press; 2014.
3. Chandra A, Khullar D, Wilensky GR. The economics of graduate medical education. N Engl J Med. 2014;370(25):2357-2360.
4. Iglehart JK. The uncertain future of Medicare and graduate medical education. N Engl J Med. 2011;365(14):1340-1345.
5. Nuckols TK, Bhattacharya J, Wolman DM, Ulmer C, Escarce JJ. Cost implications of reduced work hours and workloads for resident physicians. N Engl J Med. 2009;360(21):2202-2215.
6. Robbins RA, Singarajah CU. IOM releases report on graduate medical education. Southwest J Pulm Crit Care. 2014;9(2):123-125. http://www.swjpcc.com/editorial/2014/8/12/iom-releases-report-on-graduate-medical-education.html. Published August 12, 2014. Accessed March 5, 2015.
7. Iglehart JK. Financing graduate medical education—mounting pressure for reform. N Engl J Med. 2012;366(17):1562-1563.
8. The National Commission on Fiscal Responsibility and Reform. The Moment of Truth: Report of the National Commission on Fiscal Responsibility and Reform. The National Commission on Fiscal Responsibility and Reform website. http://www.fiscalcommission.gov/sites/fiscalcommission.gov/files/documents/TheMomentofTruth12_1_2010.pdf. Published December 2010. Accessed March 5, 2015.
9. Balanced Budget Act of 1997, Pub L No. 105-33, 111 Stat 251.
10. Weinstein S. Departmental Leadship Forum III—Orthopaedic workforce needs: challenges in a changing enviroment. Final Program: 2014 Combined Meeting of The American Orthopaedic Association and the Canadian Orthopaedic Association. American Orthopaedic Association website. https://www.aoassn.org/media/410447/final-program-with-covers.pdf. Published June 2014. Accessed March 5, 2015.
11. Jackson JB, Huntington WP, Frick SL. Assessing the value of work done by an orthopedic resident during call. J Grad Med Educ. 2014;6(3):567-570.
12. Frick SL, Richards BS, Weinstein SL, Beaty JH, Wattenbarger JM. Workforce, work, and advocacy issues in pediatric orthopaedics. J Bone Joint Surg Am. 2010;92(17):e31.
13. Physician supply and demand through 2025: key findings. Association of American Medical Colleges website. https://www.aamc.org/download/153160/data/physician_shortages_to_worsen_without_increases_in_residency_tr.pdf. Published March 3, 2015. Accessed March 5, 2015.
Reducing the likelihood that a patient will be readmitted: A resident's perspective
Over the past year, as a part of a treatment team, I encountered many discharged patients who did not heed their physician’s instructions—be it rehabilitation advice, follow-up appointments, or adherence to a drug regimen. Consequently, these patients found themselves back in the inpatient unit a few days later. A cycle of admission−discharge−readmission began for them.
I have witnessed conflicting emotions on the part of the staff (nurses and residents) toward these patients. Some staff are empathetic to their needs; others see the recurrent admissions as a ruse to find food and shelter and get attention.
In this article, I explore several aspects of this behavioral pattern and possible reasons for it, and describe the staff’s reaction to a returning patient in one case.
CASE REPORT Depressed and heavily intoxicated
Mr. R, age 35, with a history of major depressive disorder and nonadherence to treatment, is brought to the emergency room (ER) by emergency medical services because he is heavily intoxicated (blood alcohol level, >200 mg/dL). Mr. R has had 4 admissions to the inpatient psychiatry unit in the past 6 months, including 2 in the past 30 days.
After a few hours’ stay in the ER, Mr. R’s blood alcohol level drops to <100 mg/dL. He is being prepared for discharge to follow-up with outpatient psychiatric services when he begins complaining of chest pain. A cardiac workup is negative; he is again prepared for discharge when he begins reporting suicidal ideation, with a plan to jump in front of traffic.
Mr. R is admitted to the inpatient psychiatry unit.
During Mr. R’s hospitalization, he admits that he lied about being suicidal because he recently lost his job and is homeless and in dire need of food and shelter. He stays in the inpatient unit for 6 days.
An unexpected ‘adverse reaction’
During the hospital stay, staff members, who initially were concerned about Mr. R’s condition, underwent a striking transformation in their attitude toward the patient once his suicidal ideation was exposed as a hoax: They became less receptive to his needs.
The staff’s experience with Mr. R also altered their approach to other patients, who were put under unnecessary scrutiny in response to heightened suspicion of feigned illness—a classic case of “once bitten, twice shy.” The staff felt betrayed by Mr. R’s false claim of being suicidal.
Furthermore, I noticed self-doubt creeping into the minds of the residents who had admitted Mr. R. Consequently, they advocated that he should be discharged patients of an acute care bed.
The attending physicians and other members of the staff remained compassionate toward the patient, however; instead of condemning him, they tried to understand the root cause of why he sought admission: Was it nonadherence with his medication regimen? Substance abuse? Social issues? These staff members were opposed to discharging Mr. R because they believed that forced discharge would encourage him to further manipulate the system—and he would be back in the ER.
CASE CONCLUDED
The medical team concludes that it is prudent to prepare a well-thought-out discharge plan for Mr. R. He is allowed to remain as an inpatient until his social issues are addressed; he is plugged into the rehabilitation program for his alcohol addiction, with a plan for close outpatient psychiatry follow-up.
One year later, Mr. R has not been admitted again.
How to tackle shortcomings of the system
Because of changing hospital policies, an acute shortage of psychiatry inpatient beds, and the reluctance of insurance companies to reimburse for an extended stay, these beds are often hurriedly evacuated and patients are discharged prematurely to make room for acutely ill patients.1 Such policies can lead to failure to reach a therapeutic medication dosage or establish an appropriate disposition plan. Patients might relapse and find their way back to the inpatient unit.
Even though this is a system—not a personal—shortcoming, these patients are viewed negatively and are unwelcome when they return to the hospital. Notably, longer hospital stays do not necessarily lead to better care or fewer readmissions. Patients who have a longer length of stay are, in fact, sicker and have inadequate community and social support.1,2
After a year’s experience as a psychiatry resident, I came to understand that, before discharging a patient from the inpatient unit, a resident should pose a few questions to himself (herself), including:
• What is the likelihood that the patient will adhere to his (her) medication regimen?
• Where is the patient going to get his medications? Will he (she) be able to pay for them?
• Does he have a substance use disorder?
• Have the patient’s personal circumstances changed since he was admitted? If so, how?
Finding answers to these questions and working on solutions can help minimize the readmission rate.
The post-discharge component of care has a significant role, too, including psycho-education of the patient and the family regarding:
• ongoing psychiatric disease
• potential side effects of medications
• post-discharge telephone calls
• timely follow-up (within 2 or 3 weeks)
• good communication with the outpatient provider, through telephone calls or a faxed discharge summary.2,3
I’ve learned that it isn’t uncommon for health care providers to give in to negative emotions and become frustrated. For residents and other members of the team alike, it is important to talk to one’s supervisor and colleagues about that frustration. It is the duty of every member of the treatment team to support each another and maintain a therapeutic posture on the unit.
At the end of every day, of course, what matters is the well-being of our patients.
Disclosure
Dr. Sharma reports no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Botha UA, Koen L, Joska JA, et al. The revolving door phenomenon in psychiatry: comparing low-frequency and high-frequency users in psychiatry inpatient services in a developing country. Soc Psychiatry Psychiatr Epidemiol. 2010;45(4):461-468.
2. Clary C, Dever A, Schweizer E. Psychiatric inpatient’s knowledge of medication at hospital discharge. Hosp Community Psychiatry. 1992;43(2):140-144.
3. Boyer CA, McAlpine DD, Pottick KJ, et al. Identifying risk factors and key strategies in linkage to outpatient psychiatric care. Am J Psychiatry. 2000;157(10):1592-1598.
Over the past year, as a part of a treatment team, I encountered many discharged patients who did not heed their physician’s instructions—be it rehabilitation advice, follow-up appointments, or adherence to a drug regimen. Consequently, these patients found themselves back in the inpatient unit a few days later. A cycle of admission−discharge−readmission began for them.
I have witnessed conflicting emotions on the part of the staff (nurses and residents) toward these patients. Some staff are empathetic to their needs; others see the recurrent admissions as a ruse to find food and shelter and get attention.
In this article, I explore several aspects of this behavioral pattern and possible reasons for it, and describe the staff’s reaction to a returning patient in one case.
CASE REPORT Depressed and heavily intoxicated
Mr. R, age 35, with a history of major depressive disorder and nonadherence to treatment, is brought to the emergency room (ER) by emergency medical services because he is heavily intoxicated (blood alcohol level, >200 mg/dL). Mr. R has had 4 admissions to the inpatient psychiatry unit in the past 6 months, including 2 in the past 30 days.
After a few hours’ stay in the ER, Mr. R’s blood alcohol level drops to <100 mg/dL. He is being prepared for discharge to follow-up with outpatient psychiatric services when he begins complaining of chest pain. A cardiac workup is negative; he is again prepared for discharge when he begins reporting suicidal ideation, with a plan to jump in front of traffic.
Mr. R is admitted to the inpatient psychiatry unit.
During Mr. R’s hospitalization, he admits that he lied about being suicidal because he recently lost his job and is homeless and in dire need of food and shelter. He stays in the inpatient unit for 6 days.
An unexpected ‘adverse reaction’
During the hospital stay, staff members, who initially were concerned about Mr. R’s condition, underwent a striking transformation in their attitude toward the patient once his suicidal ideation was exposed as a hoax: They became less receptive to his needs.
The staff’s experience with Mr. R also altered their approach to other patients, who were put under unnecessary scrutiny in response to heightened suspicion of feigned illness—a classic case of “once bitten, twice shy.” The staff felt betrayed by Mr. R’s false claim of being suicidal.
Furthermore, I noticed self-doubt creeping into the minds of the residents who had admitted Mr. R. Consequently, they advocated that he should be discharged patients of an acute care bed.
The attending physicians and other members of the staff remained compassionate toward the patient, however; instead of condemning him, they tried to understand the root cause of why he sought admission: Was it nonadherence with his medication regimen? Substance abuse? Social issues? These staff members were opposed to discharging Mr. R because they believed that forced discharge would encourage him to further manipulate the system—and he would be back in the ER.
CASE CONCLUDED
The medical team concludes that it is prudent to prepare a well-thought-out discharge plan for Mr. R. He is allowed to remain as an inpatient until his social issues are addressed; he is plugged into the rehabilitation program for his alcohol addiction, with a plan for close outpatient psychiatry follow-up.
One year later, Mr. R has not been admitted again.
How to tackle shortcomings of the system
Because of changing hospital policies, an acute shortage of psychiatry inpatient beds, and the reluctance of insurance companies to reimburse for an extended stay, these beds are often hurriedly evacuated and patients are discharged prematurely to make room for acutely ill patients.1 Such policies can lead to failure to reach a therapeutic medication dosage or establish an appropriate disposition plan. Patients might relapse and find their way back to the inpatient unit.
Even though this is a system—not a personal—shortcoming, these patients are viewed negatively and are unwelcome when they return to the hospital. Notably, longer hospital stays do not necessarily lead to better care or fewer readmissions. Patients who have a longer length of stay are, in fact, sicker and have inadequate community and social support.1,2
After a year’s experience as a psychiatry resident, I came to understand that, before discharging a patient from the inpatient unit, a resident should pose a few questions to himself (herself), including:
• What is the likelihood that the patient will adhere to his (her) medication regimen?
• Where is the patient going to get his medications? Will he (she) be able to pay for them?
• Does he have a substance use disorder?
• Have the patient’s personal circumstances changed since he was admitted? If so, how?
Finding answers to these questions and working on solutions can help minimize the readmission rate.
The post-discharge component of care has a significant role, too, including psycho-education of the patient and the family regarding:
• ongoing psychiatric disease
• potential side effects of medications
• post-discharge telephone calls
• timely follow-up (within 2 or 3 weeks)
• good communication with the outpatient provider, through telephone calls or a faxed discharge summary.2,3
I’ve learned that it isn’t uncommon for health care providers to give in to negative emotions and become frustrated. For residents and other members of the team alike, it is important to talk to one’s supervisor and colleagues about that frustration. It is the duty of every member of the treatment team to support each another and maintain a therapeutic posture on the unit.
At the end of every day, of course, what matters is the well-being of our patients.
Disclosure
Dr. Sharma reports no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
Over the past year, as a part of a treatment team, I encountered many discharged patients who did not heed their physician’s instructions—be it rehabilitation advice, follow-up appointments, or adherence to a drug regimen. Consequently, these patients found themselves back in the inpatient unit a few days later. A cycle of admission−discharge−readmission began for them.
I have witnessed conflicting emotions on the part of the staff (nurses and residents) toward these patients. Some staff are empathetic to their needs; others see the recurrent admissions as a ruse to find food and shelter and get attention.
In this article, I explore several aspects of this behavioral pattern and possible reasons for it, and describe the staff’s reaction to a returning patient in one case.
CASE REPORT Depressed and heavily intoxicated
Mr. R, age 35, with a history of major depressive disorder and nonadherence to treatment, is brought to the emergency room (ER) by emergency medical services because he is heavily intoxicated (blood alcohol level, >200 mg/dL). Mr. R has had 4 admissions to the inpatient psychiatry unit in the past 6 months, including 2 in the past 30 days.
After a few hours’ stay in the ER, Mr. R’s blood alcohol level drops to <100 mg/dL. He is being prepared for discharge to follow-up with outpatient psychiatric services when he begins complaining of chest pain. A cardiac workup is negative; he is again prepared for discharge when he begins reporting suicidal ideation, with a plan to jump in front of traffic.
Mr. R is admitted to the inpatient psychiatry unit.
During Mr. R’s hospitalization, he admits that he lied about being suicidal because he recently lost his job and is homeless and in dire need of food and shelter. He stays in the inpatient unit for 6 days.
An unexpected ‘adverse reaction’
During the hospital stay, staff members, who initially were concerned about Mr. R’s condition, underwent a striking transformation in their attitude toward the patient once his suicidal ideation was exposed as a hoax: They became less receptive to his needs.
The staff’s experience with Mr. R also altered their approach to other patients, who were put under unnecessary scrutiny in response to heightened suspicion of feigned illness—a classic case of “once bitten, twice shy.” The staff felt betrayed by Mr. R’s false claim of being suicidal.
Furthermore, I noticed self-doubt creeping into the minds of the residents who had admitted Mr. R. Consequently, they advocated that he should be discharged patients of an acute care bed.
The attending physicians and other members of the staff remained compassionate toward the patient, however; instead of condemning him, they tried to understand the root cause of why he sought admission: Was it nonadherence with his medication regimen? Substance abuse? Social issues? These staff members were opposed to discharging Mr. R because they believed that forced discharge would encourage him to further manipulate the system—and he would be back in the ER.
CASE CONCLUDED
The medical team concludes that it is prudent to prepare a well-thought-out discharge plan for Mr. R. He is allowed to remain as an inpatient until his social issues are addressed; he is plugged into the rehabilitation program for his alcohol addiction, with a plan for close outpatient psychiatry follow-up.
One year later, Mr. R has not been admitted again.
How to tackle shortcomings of the system
Because of changing hospital policies, an acute shortage of psychiatry inpatient beds, and the reluctance of insurance companies to reimburse for an extended stay, these beds are often hurriedly evacuated and patients are discharged prematurely to make room for acutely ill patients.1 Such policies can lead to failure to reach a therapeutic medication dosage or establish an appropriate disposition plan. Patients might relapse and find their way back to the inpatient unit.
Even though this is a system—not a personal—shortcoming, these patients are viewed negatively and are unwelcome when they return to the hospital. Notably, longer hospital stays do not necessarily lead to better care or fewer readmissions. Patients who have a longer length of stay are, in fact, sicker and have inadequate community and social support.1,2
After a year’s experience as a psychiatry resident, I came to understand that, before discharging a patient from the inpatient unit, a resident should pose a few questions to himself (herself), including:
• What is the likelihood that the patient will adhere to his (her) medication regimen?
• Where is the patient going to get his medications? Will he (she) be able to pay for them?
• Does he have a substance use disorder?
• Have the patient’s personal circumstances changed since he was admitted? If so, how?
Finding answers to these questions and working on solutions can help minimize the readmission rate.
The post-discharge component of care has a significant role, too, including psycho-education of the patient and the family regarding:
• ongoing psychiatric disease
• potential side effects of medications
• post-discharge telephone calls
• timely follow-up (within 2 or 3 weeks)
• good communication with the outpatient provider, through telephone calls or a faxed discharge summary.2,3
I’ve learned that it isn’t uncommon for health care providers to give in to negative emotions and become frustrated. For residents and other members of the team alike, it is important to talk to one’s supervisor and colleagues about that frustration. It is the duty of every member of the treatment team to support each another and maintain a therapeutic posture on the unit.
At the end of every day, of course, what matters is the well-being of our patients.
Disclosure
Dr. Sharma reports no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Botha UA, Koen L, Joska JA, et al. The revolving door phenomenon in psychiatry: comparing low-frequency and high-frequency users in psychiatry inpatient services in a developing country. Soc Psychiatry Psychiatr Epidemiol. 2010;45(4):461-468.
2. Clary C, Dever A, Schweizer E. Psychiatric inpatient’s knowledge of medication at hospital discharge. Hosp Community Psychiatry. 1992;43(2):140-144.
3. Boyer CA, McAlpine DD, Pottick KJ, et al. Identifying risk factors and key strategies in linkage to outpatient psychiatric care. Am J Psychiatry. 2000;157(10):1592-1598.
1. Botha UA, Koen L, Joska JA, et al. The revolving door phenomenon in psychiatry: comparing low-frequency and high-frequency users in psychiatry inpatient services in a developing country. Soc Psychiatry Psychiatr Epidemiol. 2010;45(4):461-468.
2. Clary C, Dever A, Schweizer E. Psychiatric inpatient’s knowledge of medication at hospital discharge. Hosp Community Psychiatry. 1992;43(2):140-144.
3. Boyer CA, McAlpine DD, Pottick KJ, et al. Identifying risk factors and key strategies in linkage to outpatient psychiatric care. Am J Psychiatry. 2000;157(10):1592-1598.
Yoga for Dermatologic Conditions
Regardless of its spiritual origins, yoga has become a popular way of reaching mind and body well-being with nearly 30 million people practicing regularly worldwide.1 Yoga, which is the combination of physical postures, controlled breathing, and meditation or mindfulness, has long been used in complementary and alternative medicine around the world and recently has gained popularity as a therapeutic practice, with nearly 14 million Americans reporting that yoga was recommended to them by a physician or therapist.2,3 Studies suggest that people who participate in even brief yoga programs may see improvements in anxiety, somatic stress and discomfort, health-related quality of life, and self-rated sleep quality, all benefits that can help medical conditions, especially those that are dermatologic in nature.4,5
Stress and Dermatologic Conditions
The interaction between the mind, skin, and body is well known. Research in psychoneuroimmunology, the interaction between psychological processes and the nervous and immune systems, has examined the role of neuropeptides, hormones, and neurotransmitters in psychodermatological disorders. The correlation between neuroimmunological pathways and skin inflammation is now well recognized, specifically the interactions between the brain and skin underlying many dermatological diseases (eg, acne, alopecia areata, various types of eczema and dermatitis, oral and genital herpes, hyperhidrosis, pruritus, psoriasis, rosacea, urticaria, warts, breaking or ridging of the nails).6-9
Two biological systems are known to be affected by the systemic stress response: (1) the hypothalamic-pituitary-adrenal axis, which regulates the release of adrenocorticotropin, ß-endorphin, and cortisol, and (2) the sympathoadrenal medullary system, which regulates the release of catecholamines (eg, epinephrine, norepinephrine).7 Cortisol and catecholamines have been shown to have potent effects on the immune system as well as the inflammatory response.9 Additionally, it has been shown that cutaneous sensory nerve terminals release neuropeptides, including calcitonin gene-related peptide and substance P, both of which have different effects on the local inflammatory response.10,11
Psychological stress is well known to trigger many dermatologic conditions, but it also may lead to abnormal skin barrier function.12 The mechanism in which skin barrier function is affected appears to involve a stress-induced increase of endogenous glucocorticoids, which may consequently disrupt skin barrier function and recovery rates, stratum corneum cohesion, and epidermal antimicrobial function.13,14
Atopic dermatitis, for example, is classified as a psychophysiological disorder. Although it is not caused by stress, atopic dermatitis has been described to be precipitated or exacerbated by stress in patients.15 In fact, it was found that stressful life events preceded the onset of itching in more than 70% of patients with atopic dermatitis,16 which is especially relevant, as there is no cure and patients often experience a lifelong struggle with the condition. Additionally, stress mediates the degranulation of mast cells via corticotropin-releasing hormone and neuropeptides, and the upregulation of mast cell corticotropin-releasing hormone receptors supporting its putative role in the pathogenesis of urticaria.9,17 Furthermore, the increase in cortisol also has been described in the exacerbation of acne during times of stress.18
Psychological factors affect the management of skin conditions in more than one-third of reported dermatology patients; therefore, it is important to consider these factors in the treatment of chronic dermatological conditions, especially when they are inquired by the patient.19,20
Yoga Benefits in the Literature
The therapeutic potential of yoga has been explored in a growing number of randomized controlled trials to date.21 A recently published bibliometric analysis provided a comprehensive review of the characteristics of the randomized yoga trials available in the literature.22 The review included 366 full-text articles, with the 2 earliest studies published in 1975 and nearly 90% published within the last decade. In addition to healthy patients, it was found these randomized controlled yoga trials most commonly enrolled patients with breast cancer, depression, asthma, and type 2 diabetes mellitus.22 Another study examined psychological (eg, self-rated stress and stress behavior, anger, exhaustion, quality of life) and physiological (eg, blood pressure, heart rate, urinary catecholamines, salivary cortisol) measurements obtained before and after a 10-session yoga program that participants completed over a 4-month period, with results showing significant improvements (P<.05) on almost all stress-related subjective and physiological variables. Results were comparable with cognitive behavioral therapy.23
Not only has it been shown that yoga helps patients on a psychological level, but a recent study reported that 90-minute sessions of mindfulness meditation and gentle Hatha yoga over an 8-week period led to observable benefits on a cellular level, as telomere length was maintained in distressed breast cancer survivors compared to decreases in telomere length in the control group with patients who solely participated in a stress management seminar.24 To date, there are no known studies examining the effects of yoga on patients with skin cancer. However, a few studies have specifically examined the effect of yoga in managing non–cancer-related dermatologic issues. Specifically, one small study of psoriasis patients found that those who listened to mindfulness meditation tapes while undergoing standard phototherapy (psoralen plus UVA) healed faster than those who underwent phototherapy treatment alone.25
Because some dermatologic problems have comorbidities and increased risk factors of other medical problems, such as psoriasis with psoriatic arthritis and metabolic diseases (eg, abdominal obesity, diabetes, nonalcoholic fatty liver disease, dyslipidemia, metabolic syndrome, chronic kidney disease), it is even more pertinent to recommend approaches for healthy mind and body well-being as a supplement to medical care.26
Final Thoughts
With accurate diagnosis by a dermatologist, appropriate conventional treatments can improve dermatologic problems. These treatments alone can reduce patients’ stress and improve skin, hair, and nail conditions; however, if it is clear that stress is interfering with a patient’s overall well-being and ability to cope with his/her dermatologic condition, concurrent stress management interventions may be warranted. In some instances, recommending yoga sessions, mindful meditation, or breathing exercises may help, while in others referral to a mental health professional may be necessary.
Beyond the direct physiological effects of stress, it also is worth mentioning that patients who deal with stress also tend to scratch, pick, or irritate their skin more and often lack the motivation to adhere to skin care regimens or treatments, again supporting the idea that our approach in managing these patients must be multifaceted. As dermatologists in training, residents should be cognizant of the potential psychological sequelae of some dermatologic problems and be aware of the possible use of supplemental interventions by our patients.
1. Dangerfield A. Yoga wars. BBC News. http://news.bbc.co.uk/1/hi/7844691.stm. Published January 23, 2009. Accessed March 25, 2015.
2. Yoga Journal releases 2012 yoga in America market study [press release]. San Francisco, CA: Yoga Journal; December 6, 2012.
3. De Michaelis E. A History of Modern Yoga: Patanjali and Western Esotericism. London, United Kingdom: A&C Black; 2005.
4. Telles S, Singh N, Yadav A, et al. Effect of yoga on different aspects of mental health. Indian J Physiol Pharmacol. 2012;56:245-254.
5. Rodriguez-Vallecillo E, Woodbury-Fariña MA. Dermatological manifestations of stress in normal and psychiatric populations. Psychiatr Clin North Am. 2014;37:625-651.
6. Stander S, Raap U, Weisshaar E, et al. Pathogenesis of pruritus. J Dtsch Dermatol Ges. 2011;9:456-463.
7. Arck PC, Slominski A, Theoharides TC, et al. Neuroimmunology of stress: skin takes center stage. J Invest Dermatol. 2006;126:1697-1704.
8. Recognizing the mind-skin connection. Harvard Health Publications Web site. http://www.health.harvard.edu/newsletter_article/Recognizing_the_mind-skin_connection. Published November 1, 2006. Accessed March 31, 2015.
9. Tausk F, Elenkov I, Moynihan J. Psychoneuroimmunology. Dermatol Ther. 2008;21:22-31.
10. Pavlovic S, Liezmann C, Blois SM, et al. Substance P is a key mediator of stress-induced protection from allergic sensitization via modified antigen presentation. J Immunol. 2011;186:848-855.
11. Toyoda M, Nakamura M, Makino T, et al. Nerve growth factor and substance P are useful plasma markers of disease activity in atopic dermatitis. Br J Dermatol. 2002;147:71-79.
12. Koo JYM, Lee CS. General approach to evaluating psychodermatological disorders. In: Koo JYM, Lee CS, eds. Psychocutaneous Medicine. New York, NY: Marcel Dekker; 2003:1-29.
13. Garg A, Chren MM, Sands LP, et al. Psychological stress perturbs epidermal permeability barrier homeostasis: implications for the pathogenesis of stress-associated skin disorders. Arch Dermatol. 2001;137:53-59.
14. Elias PM, Sun R, Eder AR, et al. Treating atopic dermatitis at the source: corrective barrier repair therapy based upon new pathogenic insights. Expert Rev Dermatol. 2013;8:27-36.
15. Morren MA, Przybilla B, Bamelis M, et al. Atopic dermatitis: triggering factors. J Am Acad Dermatol. 1994;31:467-473.
16. Faulstich ME, Williamson DA. An overview of atopic dermatitis: toward a bio-behavioural integration. J Psychosom Res. 1985;29:647-654.
17. Theoharides TC, Donelan JM, Papadopoulou N, et al. Mast cells as targets of corticotropin-releasing factor and related peptides. Trends Pharmacol Sci. 2004;25:563-568.
18. Suh DH, Kwon HH. What’s new in the physiopathology of acne [published online ahead of print Jan 24, 2015]? Br J Dermatol. doi:10.1111/bjd.13634.
19. Picardi A, Mazzotti E, Pasquini P. Prevalence and correlates of suicidal ideation among patients with skin disease. J Am Acad Dermatol. 2006;54:420-426.
20. Ponarovsky B, Amital D, Lazarov A, et al. Anxiety and depression in patients with allergic and non-allergic cutaneous disorders. Int J Dermatol. 2011;50:1217-1222.
21. Khalsa SB. Yoga as a therapeutic intervention: a bibliometric analysis of published research studies. Indian J Physiol Pharmacol. 2004;48:269-285.
22. Cramer H, Lauche R, Dobos G. Characteristics of randomized controlled trials of yoga: a bibliometric analysis. BMC Complement Altern Med. 2014;14:328.
23. Granath J, Ingvarsson S, von Thiele U, et al. Stress management: a randomized study of cognitive behavioural therapy and yoga. Cogn Behav Ther. 2006;35:3-10.
24. Carlson LE, Beattie TL, Giese-Davis J, et al. Mindfulness-based cancer recovery and supportive-expressive therapy maintain telomere length relative to controls in distressed breast cancer survivors. Cancer. 2015;121:476-484.
25. Kabat-Zinn J, Wheeler E, Light T, et al. Influence of a mindfulness meditation-based stress reduction intervention on rates of skin clearing in patients with moderate to severe psoriasis undergoing phototherapy (UVB) and photochemotherapy (PUVA). Psychosom Med. 1998;60:625-632.
26. Gisondi P, Galvan A, Idolazzi L, et al. Management of moderate to severe psoriasis in patients with metabolic comorbidities. Front Med (Lausanne). 2015;2:1.
Regardless of its spiritual origins, yoga has become a popular way of reaching mind and body well-being with nearly 30 million people practicing regularly worldwide.1 Yoga, which is the combination of physical postures, controlled breathing, and meditation or mindfulness, has long been used in complementary and alternative medicine around the world and recently has gained popularity as a therapeutic practice, with nearly 14 million Americans reporting that yoga was recommended to them by a physician or therapist.2,3 Studies suggest that people who participate in even brief yoga programs may see improvements in anxiety, somatic stress and discomfort, health-related quality of life, and self-rated sleep quality, all benefits that can help medical conditions, especially those that are dermatologic in nature.4,5
Stress and Dermatologic Conditions
The interaction between the mind, skin, and body is well known. Research in psychoneuroimmunology, the interaction between psychological processes and the nervous and immune systems, has examined the role of neuropeptides, hormones, and neurotransmitters in psychodermatological disorders. The correlation between neuroimmunological pathways and skin inflammation is now well recognized, specifically the interactions between the brain and skin underlying many dermatological diseases (eg, acne, alopecia areata, various types of eczema and dermatitis, oral and genital herpes, hyperhidrosis, pruritus, psoriasis, rosacea, urticaria, warts, breaking or ridging of the nails).6-9
Two biological systems are known to be affected by the systemic stress response: (1) the hypothalamic-pituitary-adrenal axis, which regulates the release of adrenocorticotropin, ß-endorphin, and cortisol, and (2) the sympathoadrenal medullary system, which regulates the release of catecholamines (eg, epinephrine, norepinephrine).7 Cortisol and catecholamines have been shown to have potent effects on the immune system as well as the inflammatory response.9 Additionally, it has been shown that cutaneous sensory nerve terminals release neuropeptides, including calcitonin gene-related peptide and substance P, both of which have different effects on the local inflammatory response.10,11
Psychological stress is well known to trigger many dermatologic conditions, but it also may lead to abnormal skin barrier function.12 The mechanism in which skin barrier function is affected appears to involve a stress-induced increase of endogenous glucocorticoids, which may consequently disrupt skin barrier function and recovery rates, stratum corneum cohesion, and epidermal antimicrobial function.13,14
Atopic dermatitis, for example, is classified as a psychophysiological disorder. Although it is not caused by stress, atopic dermatitis has been described to be precipitated or exacerbated by stress in patients.15 In fact, it was found that stressful life events preceded the onset of itching in more than 70% of patients with atopic dermatitis,16 which is especially relevant, as there is no cure and patients often experience a lifelong struggle with the condition. Additionally, stress mediates the degranulation of mast cells via corticotropin-releasing hormone and neuropeptides, and the upregulation of mast cell corticotropin-releasing hormone receptors supporting its putative role in the pathogenesis of urticaria.9,17 Furthermore, the increase in cortisol also has been described in the exacerbation of acne during times of stress.18
Psychological factors affect the management of skin conditions in more than one-third of reported dermatology patients; therefore, it is important to consider these factors in the treatment of chronic dermatological conditions, especially when they are inquired by the patient.19,20
Yoga Benefits in the Literature
The therapeutic potential of yoga has been explored in a growing number of randomized controlled trials to date.21 A recently published bibliometric analysis provided a comprehensive review of the characteristics of the randomized yoga trials available in the literature.22 The review included 366 full-text articles, with the 2 earliest studies published in 1975 and nearly 90% published within the last decade. In addition to healthy patients, it was found these randomized controlled yoga trials most commonly enrolled patients with breast cancer, depression, asthma, and type 2 diabetes mellitus.22 Another study examined psychological (eg, self-rated stress and stress behavior, anger, exhaustion, quality of life) and physiological (eg, blood pressure, heart rate, urinary catecholamines, salivary cortisol) measurements obtained before and after a 10-session yoga program that participants completed over a 4-month period, with results showing significant improvements (P<.05) on almost all stress-related subjective and physiological variables. Results were comparable with cognitive behavioral therapy.23
Not only has it been shown that yoga helps patients on a psychological level, but a recent study reported that 90-minute sessions of mindfulness meditation and gentle Hatha yoga over an 8-week period led to observable benefits on a cellular level, as telomere length was maintained in distressed breast cancer survivors compared to decreases in telomere length in the control group with patients who solely participated in a stress management seminar.24 To date, there are no known studies examining the effects of yoga on patients with skin cancer. However, a few studies have specifically examined the effect of yoga in managing non–cancer-related dermatologic issues. Specifically, one small study of psoriasis patients found that those who listened to mindfulness meditation tapes while undergoing standard phototherapy (psoralen plus UVA) healed faster than those who underwent phototherapy treatment alone.25
Because some dermatologic problems have comorbidities and increased risk factors of other medical problems, such as psoriasis with psoriatic arthritis and metabolic diseases (eg, abdominal obesity, diabetes, nonalcoholic fatty liver disease, dyslipidemia, metabolic syndrome, chronic kidney disease), it is even more pertinent to recommend approaches for healthy mind and body well-being as a supplement to medical care.26
Final Thoughts
With accurate diagnosis by a dermatologist, appropriate conventional treatments can improve dermatologic problems. These treatments alone can reduce patients’ stress and improve skin, hair, and nail conditions; however, if it is clear that stress is interfering with a patient’s overall well-being and ability to cope with his/her dermatologic condition, concurrent stress management interventions may be warranted. In some instances, recommending yoga sessions, mindful meditation, or breathing exercises may help, while in others referral to a mental health professional may be necessary.
Beyond the direct physiological effects of stress, it also is worth mentioning that patients who deal with stress also tend to scratch, pick, or irritate their skin more and often lack the motivation to adhere to skin care regimens or treatments, again supporting the idea that our approach in managing these patients must be multifaceted. As dermatologists in training, residents should be cognizant of the potential psychological sequelae of some dermatologic problems and be aware of the possible use of supplemental interventions by our patients.
Regardless of its spiritual origins, yoga has become a popular way of reaching mind and body well-being with nearly 30 million people practicing regularly worldwide.1 Yoga, which is the combination of physical postures, controlled breathing, and meditation or mindfulness, has long been used in complementary and alternative medicine around the world and recently has gained popularity as a therapeutic practice, with nearly 14 million Americans reporting that yoga was recommended to them by a physician or therapist.2,3 Studies suggest that people who participate in even brief yoga programs may see improvements in anxiety, somatic stress and discomfort, health-related quality of life, and self-rated sleep quality, all benefits that can help medical conditions, especially those that are dermatologic in nature.4,5
Stress and Dermatologic Conditions
The interaction between the mind, skin, and body is well known. Research in psychoneuroimmunology, the interaction between psychological processes and the nervous and immune systems, has examined the role of neuropeptides, hormones, and neurotransmitters in psychodermatological disorders. The correlation between neuroimmunological pathways and skin inflammation is now well recognized, specifically the interactions between the brain and skin underlying many dermatological diseases (eg, acne, alopecia areata, various types of eczema and dermatitis, oral and genital herpes, hyperhidrosis, pruritus, psoriasis, rosacea, urticaria, warts, breaking or ridging of the nails).6-9
Two biological systems are known to be affected by the systemic stress response: (1) the hypothalamic-pituitary-adrenal axis, which regulates the release of adrenocorticotropin, ß-endorphin, and cortisol, and (2) the sympathoadrenal medullary system, which regulates the release of catecholamines (eg, epinephrine, norepinephrine).7 Cortisol and catecholamines have been shown to have potent effects on the immune system as well as the inflammatory response.9 Additionally, it has been shown that cutaneous sensory nerve terminals release neuropeptides, including calcitonin gene-related peptide and substance P, both of which have different effects on the local inflammatory response.10,11
Psychological stress is well known to trigger many dermatologic conditions, but it also may lead to abnormal skin barrier function.12 The mechanism in which skin barrier function is affected appears to involve a stress-induced increase of endogenous glucocorticoids, which may consequently disrupt skin barrier function and recovery rates, stratum corneum cohesion, and epidermal antimicrobial function.13,14
Atopic dermatitis, for example, is classified as a psychophysiological disorder. Although it is not caused by stress, atopic dermatitis has been described to be precipitated or exacerbated by stress in patients.15 In fact, it was found that stressful life events preceded the onset of itching in more than 70% of patients with atopic dermatitis,16 which is especially relevant, as there is no cure and patients often experience a lifelong struggle with the condition. Additionally, stress mediates the degranulation of mast cells via corticotropin-releasing hormone and neuropeptides, and the upregulation of mast cell corticotropin-releasing hormone receptors supporting its putative role in the pathogenesis of urticaria.9,17 Furthermore, the increase in cortisol also has been described in the exacerbation of acne during times of stress.18
Psychological factors affect the management of skin conditions in more than one-third of reported dermatology patients; therefore, it is important to consider these factors in the treatment of chronic dermatological conditions, especially when they are inquired by the patient.19,20
Yoga Benefits in the Literature
The therapeutic potential of yoga has been explored in a growing number of randomized controlled trials to date.21 A recently published bibliometric analysis provided a comprehensive review of the characteristics of the randomized yoga trials available in the literature.22 The review included 366 full-text articles, with the 2 earliest studies published in 1975 and nearly 90% published within the last decade. In addition to healthy patients, it was found these randomized controlled yoga trials most commonly enrolled patients with breast cancer, depression, asthma, and type 2 diabetes mellitus.22 Another study examined psychological (eg, self-rated stress and stress behavior, anger, exhaustion, quality of life) and physiological (eg, blood pressure, heart rate, urinary catecholamines, salivary cortisol) measurements obtained before and after a 10-session yoga program that participants completed over a 4-month period, with results showing significant improvements (P<.05) on almost all stress-related subjective and physiological variables. Results were comparable with cognitive behavioral therapy.23
Not only has it been shown that yoga helps patients on a psychological level, but a recent study reported that 90-minute sessions of mindfulness meditation and gentle Hatha yoga over an 8-week period led to observable benefits on a cellular level, as telomere length was maintained in distressed breast cancer survivors compared to decreases in telomere length in the control group with patients who solely participated in a stress management seminar.24 To date, there are no known studies examining the effects of yoga on patients with skin cancer. However, a few studies have specifically examined the effect of yoga in managing non–cancer-related dermatologic issues. Specifically, one small study of psoriasis patients found that those who listened to mindfulness meditation tapes while undergoing standard phototherapy (psoralen plus UVA) healed faster than those who underwent phototherapy treatment alone.25
Because some dermatologic problems have comorbidities and increased risk factors of other medical problems, such as psoriasis with psoriatic arthritis and metabolic diseases (eg, abdominal obesity, diabetes, nonalcoholic fatty liver disease, dyslipidemia, metabolic syndrome, chronic kidney disease), it is even more pertinent to recommend approaches for healthy mind and body well-being as a supplement to medical care.26
Final Thoughts
With accurate diagnosis by a dermatologist, appropriate conventional treatments can improve dermatologic problems. These treatments alone can reduce patients’ stress and improve skin, hair, and nail conditions; however, if it is clear that stress is interfering with a patient’s overall well-being and ability to cope with his/her dermatologic condition, concurrent stress management interventions may be warranted. In some instances, recommending yoga sessions, mindful meditation, or breathing exercises may help, while in others referral to a mental health professional may be necessary.
Beyond the direct physiological effects of stress, it also is worth mentioning that patients who deal with stress also tend to scratch, pick, or irritate their skin more and often lack the motivation to adhere to skin care regimens or treatments, again supporting the idea that our approach in managing these patients must be multifaceted. As dermatologists in training, residents should be cognizant of the potential psychological sequelae of some dermatologic problems and be aware of the possible use of supplemental interventions by our patients.
1. Dangerfield A. Yoga wars. BBC News. http://news.bbc.co.uk/1/hi/7844691.stm. Published January 23, 2009. Accessed March 25, 2015.
2. Yoga Journal releases 2012 yoga in America market study [press release]. San Francisco, CA: Yoga Journal; December 6, 2012.
3. De Michaelis E. A History of Modern Yoga: Patanjali and Western Esotericism. London, United Kingdom: A&C Black; 2005.
4. Telles S, Singh N, Yadav A, et al. Effect of yoga on different aspects of mental health. Indian J Physiol Pharmacol. 2012;56:245-254.
5. Rodriguez-Vallecillo E, Woodbury-Fariña MA. Dermatological manifestations of stress in normal and psychiatric populations. Psychiatr Clin North Am. 2014;37:625-651.
6. Stander S, Raap U, Weisshaar E, et al. Pathogenesis of pruritus. J Dtsch Dermatol Ges. 2011;9:456-463.
7. Arck PC, Slominski A, Theoharides TC, et al. Neuroimmunology of stress: skin takes center stage. J Invest Dermatol. 2006;126:1697-1704.
8. Recognizing the mind-skin connection. Harvard Health Publications Web site. http://www.health.harvard.edu/newsletter_article/Recognizing_the_mind-skin_connection. Published November 1, 2006. Accessed March 31, 2015.
9. Tausk F, Elenkov I, Moynihan J. Psychoneuroimmunology. Dermatol Ther. 2008;21:22-31.
10. Pavlovic S, Liezmann C, Blois SM, et al. Substance P is a key mediator of stress-induced protection from allergic sensitization via modified antigen presentation. J Immunol. 2011;186:848-855.
11. Toyoda M, Nakamura M, Makino T, et al. Nerve growth factor and substance P are useful plasma markers of disease activity in atopic dermatitis. Br J Dermatol. 2002;147:71-79.
12. Koo JYM, Lee CS. General approach to evaluating psychodermatological disorders. In: Koo JYM, Lee CS, eds. Psychocutaneous Medicine. New York, NY: Marcel Dekker; 2003:1-29.
13. Garg A, Chren MM, Sands LP, et al. Psychological stress perturbs epidermal permeability barrier homeostasis: implications for the pathogenesis of stress-associated skin disorders. Arch Dermatol. 2001;137:53-59.
14. Elias PM, Sun R, Eder AR, et al. Treating atopic dermatitis at the source: corrective barrier repair therapy based upon new pathogenic insights. Expert Rev Dermatol. 2013;8:27-36.
15. Morren MA, Przybilla B, Bamelis M, et al. Atopic dermatitis: triggering factors. J Am Acad Dermatol. 1994;31:467-473.
16. Faulstich ME, Williamson DA. An overview of atopic dermatitis: toward a bio-behavioural integration. J Psychosom Res. 1985;29:647-654.
17. Theoharides TC, Donelan JM, Papadopoulou N, et al. Mast cells as targets of corticotropin-releasing factor and related peptides. Trends Pharmacol Sci. 2004;25:563-568.
18. Suh DH, Kwon HH. What’s new in the physiopathology of acne [published online ahead of print Jan 24, 2015]? Br J Dermatol. doi:10.1111/bjd.13634.
19. Picardi A, Mazzotti E, Pasquini P. Prevalence and correlates of suicidal ideation among patients with skin disease. J Am Acad Dermatol. 2006;54:420-426.
20. Ponarovsky B, Amital D, Lazarov A, et al. Anxiety and depression in patients with allergic and non-allergic cutaneous disorders. Int J Dermatol. 2011;50:1217-1222.
21. Khalsa SB. Yoga as a therapeutic intervention: a bibliometric analysis of published research studies. Indian J Physiol Pharmacol. 2004;48:269-285.
22. Cramer H, Lauche R, Dobos G. Characteristics of randomized controlled trials of yoga: a bibliometric analysis. BMC Complement Altern Med. 2014;14:328.
23. Granath J, Ingvarsson S, von Thiele U, et al. Stress management: a randomized study of cognitive behavioural therapy and yoga. Cogn Behav Ther. 2006;35:3-10.
24. Carlson LE, Beattie TL, Giese-Davis J, et al. Mindfulness-based cancer recovery and supportive-expressive therapy maintain telomere length relative to controls in distressed breast cancer survivors. Cancer. 2015;121:476-484.
25. Kabat-Zinn J, Wheeler E, Light T, et al. Influence of a mindfulness meditation-based stress reduction intervention on rates of skin clearing in patients with moderate to severe psoriasis undergoing phototherapy (UVB) and photochemotherapy (PUVA). Psychosom Med. 1998;60:625-632.
26. Gisondi P, Galvan A, Idolazzi L, et al. Management of moderate to severe psoriasis in patients with metabolic comorbidities. Front Med (Lausanne). 2015;2:1.
1. Dangerfield A. Yoga wars. BBC News. http://news.bbc.co.uk/1/hi/7844691.stm. Published January 23, 2009. Accessed March 25, 2015.
2. Yoga Journal releases 2012 yoga in America market study [press release]. San Francisco, CA: Yoga Journal; December 6, 2012.
3. De Michaelis E. A History of Modern Yoga: Patanjali and Western Esotericism. London, United Kingdom: A&C Black; 2005.
4. Telles S, Singh N, Yadav A, et al. Effect of yoga on different aspects of mental health. Indian J Physiol Pharmacol. 2012;56:245-254.
5. Rodriguez-Vallecillo E, Woodbury-Fariña MA. Dermatological manifestations of stress in normal and psychiatric populations. Psychiatr Clin North Am. 2014;37:625-651.
6. Stander S, Raap U, Weisshaar E, et al. Pathogenesis of pruritus. J Dtsch Dermatol Ges. 2011;9:456-463.
7. Arck PC, Slominski A, Theoharides TC, et al. Neuroimmunology of stress: skin takes center stage. J Invest Dermatol. 2006;126:1697-1704.
8. Recognizing the mind-skin connection. Harvard Health Publications Web site. http://www.health.harvard.edu/newsletter_article/Recognizing_the_mind-skin_connection. Published November 1, 2006. Accessed March 31, 2015.
9. Tausk F, Elenkov I, Moynihan J. Psychoneuroimmunology. Dermatol Ther. 2008;21:22-31.
10. Pavlovic S, Liezmann C, Blois SM, et al. Substance P is a key mediator of stress-induced protection from allergic sensitization via modified antigen presentation. J Immunol. 2011;186:848-855.
11. Toyoda M, Nakamura M, Makino T, et al. Nerve growth factor and substance P are useful plasma markers of disease activity in atopic dermatitis. Br J Dermatol. 2002;147:71-79.
12. Koo JYM, Lee CS. General approach to evaluating psychodermatological disorders. In: Koo JYM, Lee CS, eds. Psychocutaneous Medicine. New York, NY: Marcel Dekker; 2003:1-29.
13. Garg A, Chren MM, Sands LP, et al. Psychological stress perturbs epidermal permeability barrier homeostasis: implications for the pathogenesis of stress-associated skin disorders. Arch Dermatol. 2001;137:53-59.
14. Elias PM, Sun R, Eder AR, et al. Treating atopic dermatitis at the source: corrective barrier repair therapy based upon new pathogenic insights. Expert Rev Dermatol. 2013;8:27-36.
15. Morren MA, Przybilla B, Bamelis M, et al. Atopic dermatitis: triggering factors. J Am Acad Dermatol. 1994;31:467-473.
16. Faulstich ME, Williamson DA. An overview of atopic dermatitis: toward a bio-behavioural integration. J Psychosom Res. 1985;29:647-654.
17. Theoharides TC, Donelan JM, Papadopoulou N, et al. Mast cells as targets of corticotropin-releasing factor and related peptides. Trends Pharmacol Sci. 2004;25:563-568.
18. Suh DH, Kwon HH. What’s new in the physiopathology of acne [published online ahead of print Jan 24, 2015]? Br J Dermatol. doi:10.1111/bjd.13634.
19. Picardi A, Mazzotti E, Pasquini P. Prevalence and correlates of suicidal ideation among patients with skin disease. J Am Acad Dermatol. 2006;54:420-426.
20. Ponarovsky B, Amital D, Lazarov A, et al. Anxiety and depression in patients with allergic and non-allergic cutaneous disorders. Int J Dermatol. 2011;50:1217-1222.
21. Khalsa SB. Yoga as a therapeutic intervention: a bibliometric analysis of published research studies. Indian J Physiol Pharmacol. 2004;48:269-285.
22. Cramer H, Lauche R, Dobos G. Characteristics of randomized controlled trials of yoga: a bibliometric analysis. BMC Complement Altern Med. 2014;14:328.
23. Granath J, Ingvarsson S, von Thiele U, et al. Stress management: a randomized study of cognitive behavioural therapy and yoga. Cogn Behav Ther. 2006;35:3-10.
24. Carlson LE, Beattie TL, Giese-Davis J, et al. Mindfulness-based cancer recovery and supportive-expressive therapy maintain telomere length relative to controls in distressed breast cancer survivors. Cancer. 2015;121:476-484.
25. Kabat-Zinn J, Wheeler E, Light T, et al. Influence of a mindfulness meditation-based stress reduction intervention on rates of skin clearing in patients with moderate to severe psoriasis undergoing phototherapy (UVB) and photochemotherapy (PUVA). Psychosom Med. 1998;60:625-632.
26. Gisondi P, Galvan A, Idolazzi L, et al. Management of moderate to severe psoriasis in patients with metabolic comorbidities. Front Med (Lausanne). 2015;2:1.
Factors Affecting the Pursuit of Academic Careers Among Dermatology Residents
Dermatology has become one of the most competitive, if not the most competitive, medical specialties to enter. It attracts the brightest and most accomplished medical students who have excelled not only in the classroom and clinical setting but also in the research setting. Many successful applicants take a substantial amount of time off to pursue research and publish articles.
Despite the competitive nature of the specialty, it is well known that a marked shortage of academic dermatologists has existed for more than 30 years.1-3 In fact, the number of graduates from US dermatology residency programs who pursue academic careers has progressively declined.4 Nearly all dermatology residents have a strong academic background; however, many residents opt to pursue private practice instead of a career in academia.5-9 This trend has implications not only for future dermatology research but also for the teaching and training of future generations of dermatologists.8
To address this shortage, it is important to recruit dermatology residents who have a genuine interest in pursuing academic careers. Unfortunately, many residency applicants may overinflate their interest in academics to boost their chances of acceptance.4 Additionally, it has been shown that dermatology residents who were interested in academic careers at the time of application to the program often lost interest during residency.10
Because it can be difficult to determine a resident’s true interest in an academic career at the time of application and his/her initial interest may wean during residency, it may be more helpful to encourage dermatology residency programs to create environments that will produce residents who are more enthusiastic about and more likely to pursue careers in academia. A lack of mentorship has been shown to be associated with a loss of interest in academic careers during residency.10 If better mentorship opportunities were provided, then perhaps dermatology residents would be more likely to pursue careers in teaching and research.
A 2006 study by Wu et al5 demonstrated that various program characteristics were associated with the pursuit of academic careers among dermatology residents. The number of faculty members and the number of full-time faculty publications at a given residency program were most strongly correlated with the number of residents who pursued academic careers, which suggested that having a large faculty and encouraging dermatology residents to publish research during residency may motivate their pursuit of academic careers in dermatology.5 The current study was designed to replicate these data and respond to limitations in the original study.
Methods
Data were collected from all accredited dermatology residency programs in the United States as of December 31, 2008. The names of all full-time faculty members at these resident programs were obtained, and it was determined where each faculty member attended dermatology residency. The number of graduates who became full-time clinical or research faculty members and the number of graduates who became chairs or chiefs were counted. Residency programs excluded from these analyses included The University of Texas at Austin, University of Texas Medical Branch, and University of Connecticut, which commenced in 2008, as well as Kaiser Permanente Southern California, which commenced in 2010. Residency programs that were started after 2004 were excluded from the study, as it was thought that these programs may not have graduated a sufficient number of residents for assessment. Military residency programs also were excluded, as graduates from these programs often do not freely choose their careers after residency, and the National Institutes of Health (NIH) dermatology residency program was excluded because it is not a traditional 3-year residency program.
The primary end point was the ratio of full-time faculty members graduated to the total number of graduates from each dermatology residency program. Based on a prior study by Wu et al5 in 2006, it was believed that several program variables might affect pursuit of academic careers among dermatology residents, including total number of full-time faculty members, total number of residents, NIH funding received (in dollars) in 2008 (http://www.report.nih.gov/award/index.cfm), Dermatology Foundation (DF) funding received (in dollars) in 2008 (http://www.dermatology foundation.org/rap/), number of publications from full-time faculty members in 2008 (http://www.ncbi.nlm.nih.gov/pubmed/), number of full-time faculty lectures given at annual meetings of 5 societies in 2008 (American Academy of Dermatology, the Society for Investigative Dermatology, the American Society of Dermatopathology, the Society for Pediatric Dermatology, and American Society for Dermatologic Surgery), number of faculty members on the editorial boards of 6 major dermatology journals (Journal of the American Academy of Dermatology, Journal of Investigative Dermatology, Archives of Dermatology [currently known as JAMA Dermatology], Dermatologic Surgery, Pediatric Dermatology, and Journal of Cutaneous Pathology), and status as a department of dermatology or a division of internal medicine. The association between the ratio of number of full-time faculty members to number of residents for each residency program were determined for each of the outcome variables because they were believed to serve as an indicator of mentorship.
Data regarding faculty and residents were obtained from program Web sites and inquiries from individual programs. The year 1974 was used as a cutoff for the total number of graduates from each program. For faculty members who split time between 2 residency programs, each program was given credit for the duration of time spent at that program. If it was not clear how long the faculty member spent at each program, a credit of 1.5 years was given, which is half the duration of a dermatology residency. Faculty members who held a PhD only and those who completed their residencies in non-US dermatology residency programs were excluded from the outcome variables. To avoid duplicate faculty publications, collections for each residency program were created within PubMed (ie, if 2 authors from the same program coauthored an article, it was only counted once toward the total number of faculty publications from that program).
Descriptive exploratory statistical analysis in the form of a correlation matrix was completed to determine the most strongly positive and negative variables that were correlated with the ratio of graduating full-time faculty to estimated total graduates. Variables also were correlated with the secondary outcomes of ratio of graduating department chairs/chiefs to estimated total number of graduates and ratio of graduating program directors to estimated total number of graduates. Spearman rank correlation coefficients and P values were reported. Additionally, a 2-sample t test was performed to compare primary and secondary outcome variables between dermatology department versus division of dermatology under the department of internal medicine. Data were analyzed using SAS version 9.2. The institutional review board at Kaiser Permanente Southern California approved this study.
Results
Due to space considerations, analyses are based on data that are not published in this article. Data regarding the characteristics of each residency program are available from the authors.
Data from 103 dermatology residency programs were included in the analysis. Of these programs, 43% had received NIH funding in 2008 and 22% had received DF funding. Two-thirds of programs had at least 1 faculty member on the editorial boards of 6 major dermatology journals; 38% had at least 2 faculty members and 9% had at least 5 faculty members on editorial boards. One-third of programs had no faculty members on these editorial boards. Sixty-nine percent of programs had 1 or more lectures given by full-time faculty members at annual society meetings in 2008; 48% of these programs had 1 to 5 lectures, 17% had 6 to 10, and 5% had more than 10. Thirty-one percent had no faculty members lecture at these meetings. Ninety-six percent of programs had 1 or more publications from full-time faculty members in 2008; 54% of programs had 1 to 20 publications, 24% had 21 to 40 publications, 14% had 41 to 60 publications, 5% had 61 to 80 publications, and 3% had more than 81 publications. Four percent of programs had no publications. Seventy-seven percent of programs were classified as departments and 23% were classified as divisions.
Factors Correlated With Producing Full-time Faculty
The Spearman rank correlation coefficient and P value were reported for each variable (Table 1). All coefficients were positive, signifying a positive correlation. Values closer to 1 were indicative of stronger correlations. P<.05 indicated statistically significant correlations for all factors investigated. The most strongly correlated factor was the ratio of faculty to residents in 2008, followed by number of full-time faculty, number of full-time faculty publications, number of lectures from full-time faculty, and number of faculty on editorial boards. The amount of NIH and DF funding received as well as total number of residents in 2008 also were correlated.
A 2-sample t test was performed to compare the number of graduates pursuing careers in academia from departments versus divisions, but the results were not statistically significant (P=.92).
Ranking Individual Programs With the Highest Number of Graduates Pursuing Academic Careers
The top 5 dermatology residency programs with the highest ratio of full-time faculty members graduated to the estimated total number of graduates were Brown University (Providence, Rhode Island), Loyola University (Chicago, Illinois), UMDNJ-Robert Wood Johnson Medical School at Camden (Camden, New Jersey), Harvard University (Cambridge, Massachusetts), and the University of Vermont (Burlington, Vermont)(Table 2). If faculty members spent time at more than one residency program, they were given credit for time spent at each program. For this reason, not all numbers are integers.
Ranking Individual Programs With the Highest Number of Full-time Faculty Members Who Completed Residency at the Same Institution
The top 10 institutions with the highest percentage of full-time faculty members who completed their residency at the same institution are shown in Table 3. Most of the programs with higher percentages are small programs. Fourteen programs had no faculty members who completed residency at the same program (data not shown).
Comment
Although this study focused on US dermatology residency programs, a shortage of academic dermatologists has been noted worldwide.11-13 By determining residency program variables associated with the pursuit of academic careers, individual programs may be able to make changes that would encourage residents to pursue careers in academia following graduation.
The factor most strongly correlated with graduates pursuing a career in academics was the ratio of faculty to residents in 2008. Also highly correlated was total number of full-time faculty members. We hypothesize that programs with more faculty members may provide better mentorship for residents. A study by Reck et al10 demonstrated that a lack of mentorship was associated with residents’ loss of interest in academics. A survey of residency program directors demonstrated that mentorship played a role in career development and that it was important for residents to have mentors.14 Given the shortage of academic faculty, increasing the number of faculty may not be feasible for individual programs; however, assuming that more faculty members is a marker for mentorship, there are many ways that individual programs may improve mentorship. There are ample opportunities in clinics to demonstrate to residents the value of research to patient care.13 Residency programs also could establish mentorship programs, pairing residents with individual faculty members who share similar interests. Some programs currently have such mentorship programs but many do not.
Also strongly correlated with the number of graduates pursuing careers in academia was the number of full-time faculty publications. It is presumed that these programs also have published extensively in the past, which may have positively influenced the programs’ residents toward academics. This factor can be easily addressed among individual residency programs, and in fact many residency programs do encourage or require residents to publish during their residency. Exposure to the process of collecting data and writing manuscripts can bolster a resident’s interest in academics. In a prior study by Wu et al,5 publications were counted multiple times if multiple faculty members were authors. This limitation was addressed by only counting each manuscript once.
Other variables correlated with the number of graduates pursuing academic careers included number of lectures from full-time faculty members at annual society meetings, number of full-time faculty on editorial boards, and amount of NIH and DF funding received. All of these variables represent the importance of establishing an academic environment and promoting dermatology research. A 2009 study by Lim and Kimball,15 which also evaluated factors associated with pursuing a career in academics, demonstrated that the number of publications prior to residency and volunteerism were associated with an academic career choice. Residents with an MD/PhD were more likely to pursue a career in academics, which also was demonstrated by a similar study in 2008.16
Wu et al5 demonstrated similar results using data from 2001 to 2004; however, amount of NIH and DF funding received was found to be negatively correlated with graduates pursuing a career in academics in the original study. This finding was surprising but was not replicated in the current study. In the current study, the amount of funding (in dollars) rather than number of grants was analyzed because it was felt that the amount of funding received was a better reflection of the quantity of research being conducted. The prior study examined the relationship between the number of grants received and the number of graduates pursuing a career in academics. It is unclear why results varied in the 2 studies, but the most recent data are consistent with our hypothesis that increased research funding is associated with more residents pursuing careers in academia.
The current study also has some limitations. It is a retrospective observational study looking at data from 2008. Choosing data from another study period may have provided different results; however, it is reassuring that the data from the current study were very similar to a prior study looking at data from 2001 to 2004.5
It is assumed that residency program characteristics remained constant over time. It was also assumed that the number of residents in a program remained constant over time. These assumptions were necessary to draw conclusions about the data. It may be that some programs have changed substantially over time, which was not accounted for in the current study. To estimate the total number of graduates, it was assumed that faculty did not practice for more than 35 years, which may not have been true; if the faculty member practiced more than 35 years, it would have altered our estimates. It is also assumed that data on residency programs’ Web sites at time of data collection were updated and accurate. It is likely that data were not 100% accurate on all Web sites at the time of data collection. However, verification of accuracy of data would have been cumbersome, and it would have been difficult to get participation from all programs.
This study does not differentiate the total number of new faculty members who join a residency program and those who are retained for many years. Encouraging residents to pursue a career in academics may increase the number of academic faculty.17 However, efforts must also be placed on retention of faculty members because many of the newly graduated residents who enter academics ultimately leave.18,19
Conclusion
In conclusion, our data suggest that programs with more faculty members may encourage residents to enter careers in academia following graduation. Additionally, the number of publications increases the likelihood of residents pursuing academic careers. By providing mentorship and research opportunities to residents, perhaps residency programs can encourage their graduates to become academic dermatologists. A program’s overall academic environment, including faculty lectures at annual society meetings, faculty on editorial boards, and increased research funding are all associated with graduates pursuing careers in academics.
Ackowledgements—We thank all of the program coordinators, full-time faculty members, program directors, chairs, and chiefs who were kind enough to field our questions if we had any missing data about their programs. We would like to thank Mary H. Black, PhD (Los Angeles, California), for her contribution to the analytical plan.
APPENDIX
1. Wheeler CE Jr, Briggaman RA, Lynch PJ, et al. Shortage of full-time faculty in dermatology. Arch Dermatol. 1973;107:529-532.
2. Wheeler CE Jr, Briggaman RA, Caro I. Shortage of full-time faculty in dermatology. Arch Dermatol. 1977;113:297-301.
3. Resneck J. Too few or too many academic dermatologists? difficulties in assessing optimal workforce size. Arch Dermatol. 2001;137:1295-1301.
4. Kia KF, Gielczyk RA, Ellis CN. Academia is the life for me, I’m sure. Arch Dermatol. 2006;142:911-913.
5. Wu JJ, Ramirez CC, Alonso CA, et al. Dermatology residency program characteristics that correlate with graduates selecting an academic dermatology career. Arch Dermatol. 2006;142:845-850.
6. Wu JJ, Tyring SK. The academic strength of current dermatology residency applicants. Dermatol Online J. 2003;9:22.
7. Hinchman KF, Wu JJ. Decisions in choosing a career in academic dermatology. Cutis. 2008;82:368-371.
8. Resneck JS Jr, Tierney EP, Kimball AB. Challenges facing academic dermatology: survey data on the faculty workforce. J Am Acad Dermatol. 2006;54:211-216.
9. Rubenstein DS, Blauvelt A, Chen SC, et al. The future of academic dermatology in the United States: report on the resident retreat for future physician-scientists, June 15-17, 2001. J Am Acad Dermatol. 2002;47:300-303.
10. Reck SJ, Stratman EJ, Vogel C, et al. Assessment of residents’ loss of interest in academic careers and identification of correctable factors. Arch Dermatol. 2006;142:855-858.
11. Olerud JE. Academic workforce in dermatology. Arch Dermatol. 2007;143:409-410.
12. Singer N. More doctors turning to the business of beauty. New York Times. November 30, 2006:A1.
13. Dogra S. Fate of medical dermatology in the era of cosmetic dermatology and dermatosurgery. Indian J Dermatol Venereol Leprol. 2009;75:4-7.
14. Donovan JC. A survey of dermatology residency program directors’ views on mentorship. Dermatol Online J. 2009;15:1.
15. Lim JL, Kimball AB. Residency applications and identification of factors associated with residents’ ultimate career decisions. Arch Dermatol. 2009;145:943-944.
16. Wu JJ, Davis KF, Ramirez CC, et al. MD/PhDs are more likely than MDs to choose a career in academics. Dermatol Online J. 2008;14:27.
17. Wu JJ. Current strategies to address the ongoing shortage of academic dermatologists. J Am Acad Dermatol. 2007;56:1065-1066.
18. Loo DS, Liu CL, Geller AC, et al. Academic dermatology manpower. Arch Dermatol. 2007;143:341-347.
19. Turner E, Yoo J, Salter S, et al. Leadership workforce in academic dermatology. Arch Dermatol. 2007;143:948-949.
Dermatology has become one of the most competitive, if not the most competitive, medical specialties to enter. It attracts the brightest and most accomplished medical students who have excelled not only in the classroom and clinical setting but also in the research setting. Many successful applicants take a substantial amount of time off to pursue research and publish articles.
Despite the competitive nature of the specialty, it is well known that a marked shortage of academic dermatologists has existed for more than 30 years.1-3 In fact, the number of graduates from US dermatology residency programs who pursue academic careers has progressively declined.4 Nearly all dermatology residents have a strong academic background; however, many residents opt to pursue private practice instead of a career in academia.5-9 This trend has implications not only for future dermatology research but also for the teaching and training of future generations of dermatologists.8
To address this shortage, it is important to recruit dermatology residents who have a genuine interest in pursuing academic careers. Unfortunately, many residency applicants may overinflate their interest in academics to boost their chances of acceptance.4 Additionally, it has been shown that dermatology residents who were interested in academic careers at the time of application to the program often lost interest during residency.10
Because it can be difficult to determine a resident’s true interest in an academic career at the time of application and his/her initial interest may wean during residency, it may be more helpful to encourage dermatology residency programs to create environments that will produce residents who are more enthusiastic about and more likely to pursue careers in academia. A lack of mentorship has been shown to be associated with a loss of interest in academic careers during residency.10 If better mentorship opportunities were provided, then perhaps dermatology residents would be more likely to pursue careers in teaching and research.
A 2006 study by Wu et al5 demonstrated that various program characteristics were associated with the pursuit of academic careers among dermatology residents. The number of faculty members and the number of full-time faculty publications at a given residency program were most strongly correlated with the number of residents who pursued academic careers, which suggested that having a large faculty and encouraging dermatology residents to publish research during residency may motivate their pursuit of academic careers in dermatology.5 The current study was designed to replicate these data and respond to limitations in the original study.
Methods
Data were collected from all accredited dermatology residency programs in the United States as of December 31, 2008. The names of all full-time faculty members at these resident programs were obtained, and it was determined where each faculty member attended dermatology residency. The number of graduates who became full-time clinical or research faculty members and the number of graduates who became chairs or chiefs were counted. Residency programs excluded from these analyses included The University of Texas at Austin, University of Texas Medical Branch, and University of Connecticut, which commenced in 2008, as well as Kaiser Permanente Southern California, which commenced in 2010. Residency programs that were started after 2004 were excluded from the study, as it was thought that these programs may not have graduated a sufficient number of residents for assessment. Military residency programs also were excluded, as graduates from these programs often do not freely choose their careers after residency, and the National Institutes of Health (NIH) dermatology residency program was excluded because it is not a traditional 3-year residency program.
The primary end point was the ratio of full-time faculty members graduated to the total number of graduates from each dermatology residency program. Based on a prior study by Wu et al5 in 2006, it was believed that several program variables might affect pursuit of academic careers among dermatology residents, including total number of full-time faculty members, total number of residents, NIH funding received (in dollars) in 2008 (http://www.report.nih.gov/award/index.cfm), Dermatology Foundation (DF) funding received (in dollars) in 2008 (http://www.dermatology foundation.org/rap/), number of publications from full-time faculty members in 2008 (http://www.ncbi.nlm.nih.gov/pubmed/), number of full-time faculty lectures given at annual meetings of 5 societies in 2008 (American Academy of Dermatology, the Society for Investigative Dermatology, the American Society of Dermatopathology, the Society for Pediatric Dermatology, and American Society for Dermatologic Surgery), number of faculty members on the editorial boards of 6 major dermatology journals (Journal of the American Academy of Dermatology, Journal of Investigative Dermatology, Archives of Dermatology [currently known as JAMA Dermatology], Dermatologic Surgery, Pediatric Dermatology, and Journal of Cutaneous Pathology), and status as a department of dermatology or a division of internal medicine. The association between the ratio of number of full-time faculty members to number of residents for each residency program were determined for each of the outcome variables because they were believed to serve as an indicator of mentorship.
Data regarding faculty and residents were obtained from program Web sites and inquiries from individual programs. The year 1974 was used as a cutoff for the total number of graduates from each program. For faculty members who split time between 2 residency programs, each program was given credit for the duration of time spent at that program. If it was not clear how long the faculty member spent at each program, a credit of 1.5 years was given, which is half the duration of a dermatology residency. Faculty members who held a PhD only and those who completed their residencies in non-US dermatology residency programs were excluded from the outcome variables. To avoid duplicate faculty publications, collections for each residency program were created within PubMed (ie, if 2 authors from the same program coauthored an article, it was only counted once toward the total number of faculty publications from that program).
Descriptive exploratory statistical analysis in the form of a correlation matrix was completed to determine the most strongly positive and negative variables that were correlated with the ratio of graduating full-time faculty to estimated total graduates. Variables also were correlated with the secondary outcomes of ratio of graduating department chairs/chiefs to estimated total number of graduates and ratio of graduating program directors to estimated total number of graduates. Spearman rank correlation coefficients and P values were reported. Additionally, a 2-sample t test was performed to compare primary and secondary outcome variables between dermatology department versus division of dermatology under the department of internal medicine. Data were analyzed using SAS version 9.2. The institutional review board at Kaiser Permanente Southern California approved this study.
Results
Due to space considerations, analyses are based on data that are not published in this article. Data regarding the characteristics of each residency program are available from the authors.
Data from 103 dermatology residency programs were included in the analysis. Of these programs, 43% had received NIH funding in 2008 and 22% had received DF funding. Two-thirds of programs had at least 1 faculty member on the editorial boards of 6 major dermatology journals; 38% had at least 2 faculty members and 9% had at least 5 faculty members on editorial boards. One-third of programs had no faculty members on these editorial boards. Sixty-nine percent of programs had 1 or more lectures given by full-time faculty members at annual society meetings in 2008; 48% of these programs had 1 to 5 lectures, 17% had 6 to 10, and 5% had more than 10. Thirty-one percent had no faculty members lecture at these meetings. Ninety-six percent of programs had 1 or more publications from full-time faculty members in 2008; 54% of programs had 1 to 20 publications, 24% had 21 to 40 publications, 14% had 41 to 60 publications, 5% had 61 to 80 publications, and 3% had more than 81 publications. Four percent of programs had no publications. Seventy-seven percent of programs were classified as departments and 23% were classified as divisions.
Factors Correlated With Producing Full-time Faculty
The Spearman rank correlation coefficient and P value were reported for each variable (Table 1). All coefficients were positive, signifying a positive correlation. Values closer to 1 were indicative of stronger correlations. P<.05 indicated statistically significant correlations for all factors investigated. The most strongly correlated factor was the ratio of faculty to residents in 2008, followed by number of full-time faculty, number of full-time faculty publications, number of lectures from full-time faculty, and number of faculty on editorial boards. The amount of NIH and DF funding received as well as total number of residents in 2008 also were correlated.
A 2-sample t test was performed to compare the number of graduates pursuing careers in academia from departments versus divisions, but the results were not statistically significant (P=.92).
Ranking Individual Programs With the Highest Number of Graduates Pursuing Academic Careers
The top 5 dermatology residency programs with the highest ratio of full-time faculty members graduated to the estimated total number of graduates were Brown University (Providence, Rhode Island), Loyola University (Chicago, Illinois), UMDNJ-Robert Wood Johnson Medical School at Camden (Camden, New Jersey), Harvard University (Cambridge, Massachusetts), and the University of Vermont (Burlington, Vermont)(Table 2). If faculty members spent time at more than one residency program, they were given credit for time spent at each program. For this reason, not all numbers are integers.
Ranking Individual Programs With the Highest Number of Full-time Faculty Members Who Completed Residency at the Same Institution
The top 10 institutions with the highest percentage of full-time faculty members who completed their residency at the same institution are shown in Table 3. Most of the programs with higher percentages are small programs. Fourteen programs had no faculty members who completed residency at the same program (data not shown).
Comment
Although this study focused on US dermatology residency programs, a shortage of academic dermatologists has been noted worldwide.11-13 By determining residency program variables associated with the pursuit of academic careers, individual programs may be able to make changes that would encourage residents to pursue careers in academia following graduation.
The factor most strongly correlated with graduates pursuing a career in academics was the ratio of faculty to residents in 2008. Also highly correlated was total number of full-time faculty members. We hypothesize that programs with more faculty members may provide better mentorship for residents. A study by Reck et al10 demonstrated that a lack of mentorship was associated with residents’ loss of interest in academics. A survey of residency program directors demonstrated that mentorship played a role in career development and that it was important for residents to have mentors.14 Given the shortage of academic faculty, increasing the number of faculty may not be feasible for individual programs; however, assuming that more faculty members is a marker for mentorship, there are many ways that individual programs may improve mentorship. There are ample opportunities in clinics to demonstrate to residents the value of research to patient care.13 Residency programs also could establish mentorship programs, pairing residents with individual faculty members who share similar interests. Some programs currently have such mentorship programs but many do not.
Also strongly correlated with the number of graduates pursuing careers in academia was the number of full-time faculty publications. It is presumed that these programs also have published extensively in the past, which may have positively influenced the programs’ residents toward academics. This factor can be easily addressed among individual residency programs, and in fact many residency programs do encourage or require residents to publish during their residency. Exposure to the process of collecting data and writing manuscripts can bolster a resident’s interest in academics. In a prior study by Wu et al,5 publications were counted multiple times if multiple faculty members were authors. This limitation was addressed by only counting each manuscript once.
Other variables correlated with the number of graduates pursuing academic careers included number of lectures from full-time faculty members at annual society meetings, number of full-time faculty on editorial boards, and amount of NIH and DF funding received. All of these variables represent the importance of establishing an academic environment and promoting dermatology research. A 2009 study by Lim and Kimball,15 which also evaluated factors associated with pursuing a career in academics, demonstrated that the number of publications prior to residency and volunteerism were associated with an academic career choice. Residents with an MD/PhD were more likely to pursue a career in academics, which also was demonstrated by a similar study in 2008.16
Wu et al5 demonstrated similar results using data from 2001 to 2004; however, amount of NIH and DF funding received was found to be negatively correlated with graduates pursuing a career in academics in the original study. This finding was surprising but was not replicated in the current study. In the current study, the amount of funding (in dollars) rather than number of grants was analyzed because it was felt that the amount of funding received was a better reflection of the quantity of research being conducted. The prior study examined the relationship between the number of grants received and the number of graduates pursuing a career in academics. It is unclear why results varied in the 2 studies, but the most recent data are consistent with our hypothesis that increased research funding is associated with more residents pursuing careers in academia.
The current study also has some limitations. It is a retrospective observational study looking at data from 2008. Choosing data from another study period may have provided different results; however, it is reassuring that the data from the current study were very similar to a prior study looking at data from 2001 to 2004.5
It is assumed that residency program characteristics remained constant over time. It was also assumed that the number of residents in a program remained constant over time. These assumptions were necessary to draw conclusions about the data. It may be that some programs have changed substantially over time, which was not accounted for in the current study. To estimate the total number of graduates, it was assumed that faculty did not practice for more than 35 years, which may not have been true; if the faculty member practiced more than 35 years, it would have altered our estimates. It is also assumed that data on residency programs’ Web sites at time of data collection were updated and accurate. It is likely that data were not 100% accurate on all Web sites at the time of data collection. However, verification of accuracy of data would have been cumbersome, and it would have been difficult to get participation from all programs.
This study does not differentiate the total number of new faculty members who join a residency program and those who are retained for many years. Encouraging residents to pursue a career in academics may increase the number of academic faculty.17 However, efforts must also be placed on retention of faculty members because many of the newly graduated residents who enter academics ultimately leave.18,19
Conclusion
In conclusion, our data suggest that programs with more faculty members may encourage residents to enter careers in academia following graduation. Additionally, the number of publications increases the likelihood of residents pursuing academic careers. By providing mentorship and research opportunities to residents, perhaps residency programs can encourage their graduates to become academic dermatologists. A program’s overall academic environment, including faculty lectures at annual society meetings, faculty on editorial boards, and increased research funding are all associated with graduates pursuing careers in academics.
Ackowledgements—We thank all of the program coordinators, full-time faculty members, program directors, chairs, and chiefs who were kind enough to field our questions if we had any missing data about their programs. We would like to thank Mary H. Black, PhD (Los Angeles, California), for her contribution to the analytical plan.
APPENDIX
Dermatology has become one of the most competitive, if not the most competitive, medical specialties to enter. It attracts the brightest and most accomplished medical students who have excelled not only in the classroom and clinical setting but also in the research setting. Many successful applicants take a substantial amount of time off to pursue research and publish articles.
Despite the competitive nature of the specialty, it is well known that a marked shortage of academic dermatologists has existed for more than 30 years.1-3 In fact, the number of graduates from US dermatology residency programs who pursue academic careers has progressively declined.4 Nearly all dermatology residents have a strong academic background; however, many residents opt to pursue private practice instead of a career in academia.5-9 This trend has implications not only for future dermatology research but also for the teaching and training of future generations of dermatologists.8
To address this shortage, it is important to recruit dermatology residents who have a genuine interest in pursuing academic careers. Unfortunately, many residency applicants may overinflate their interest in academics to boost their chances of acceptance.4 Additionally, it has been shown that dermatology residents who were interested in academic careers at the time of application to the program often lost interest during residency.10
Because it can be difficult to determine a resident’s true interest in an academic career at the time of application and his/her initial interest may wean during residency, it may be more helpful to encourage dermatology residency programs to create environments that will produce residents who are more enthusiastic about and more likely to pursue careers in academia. A lack of mentorship has been shown to be associated with a loss of interest in academic careers during residency.10 If better mentorship opportunities were provided, then perhaps dermatology residents would be more likely to pursue careers in teaching and research.
A 2006 study by Wu et al5 demonstrated that various program characteristics were associated with the pursuit of academic careers among dermatology residents. The number of faculty members and the number of full-time faculty publications at a given residency program were most strongly correlated with the number of residents who pursued academic careers, which suggested that having a large faculty and encouraging dermatology residents to publish research during residency may motivate their pursuit of academic careers in dermatology.5 The current study was designed to replicate these data and respond to limitations in the original study.
Methods
Data were collected from all accredited dermatology residency programs in the United States as of December 31, 2008. The names of all full-time faculty members at these resident programs were obtained, and it was determined where each faculty member attended dermatology residency. The number of graduates who became full-time clinical or research faculty members and the number of graduates who became chairs or chiefs were counted. Residency programs excluded from these analyses included The University of Texas at Austin, University of Texas Medical Branch, and University of Connecticut, which commenced in 2008, as well as Kaiser Permanente Southern California, which commenced in 2010. Residency programs that were started after 2004 were excluded from the study, as it was thought that these programs may not have graduated a sufficient number of residents for assessment. Military residency programs also were excluded, as graduates from these programs often do not freely choose their careers after residency, and the National Institutes of Health (NIH) dermatology residency program was excluded because it is not a traditional 3-year residency program.
The primary end point was the ratio of full-time faculty members graduated to the total number of graduates from each dermatology residency program. Based on a prior study by Wu et al5 in 2006, it was believed that several program variables might affect pursuit of academic careers among dermatology residents, including total number of full-time faculty members, total number of residents, NIH funding received (in dollars) in 2008 (http://www.report.nih.gov/award/index.cfm), Dermatology Foundation (DF) funding received (in dollars) in 2008 (http://www.dermatology foundation.org/rap/), number of publications from full-time faculty members in 2008 (http://www.ncbi.nlm.nih.gov/pubmed/), number of full-time faculty lectures given at annual meetings of 5 societies in 2008 (American Academy of Dermatology, the Society for Investigative Dermatology, the American Society of Dermatopathology, the Society for Pediatric Dermatology, and American Society for Dermatologic Surgery), number of faculty members on the editorial boards of 6 major dermatology journals (Journal of the American Academy of Dermatology, Journal of Investigative Dermatology, Archives of Dermatology [currently known as JAMA Dermatology], Dermatologic Surgery, Pediatric Dermatology, and Journal of Cutaneous Pathology), and status as a department of dermatology or a division of internal medicine. The association between the ratio of number of full-time faculty members to number of residents for each residency program were determined for each of the outcome variables because they were believed to serve as an indicator of mentorship.
Data regarding faculty and residents were obtained from program Web sites and inquiries from individual programs. The year 1974 was used as a cutoff for the total number of graduates from each program. For faculty members who split time between 2 residency programs, each program was given credit for the duration of time spent at that program. If it was not clear how long the faculty member spent at each program, a credit of 1.5 years was given, which is half the duration of a dermatology residency. Faculty members who held a PhD only and those who completed their residencies in non-US dermatology residency programs were excluded from the outcome variables. To avoid duplicate faculty publications, collections for each residency program were created within PubMed (ie, if 2 authors from the same program coauthored an article, it was only counted once toward the total number of faculty publications from that program).
Descriptive exploratory statistical analysis in the form of a correlation matrix was completed to determine the most strongly positive and negative variables that were correlated with the ratio of graduating full-time faculty to estimated total graduates. Variables also were correlated with the secondary outcomes of ratio of graduating department chairs/chiefs to estimated total number of graduates and ratio of graduating program directors to estimated total number of graduates. Spearman rank correlation coefficients and P values were reported. Additionally, a 2-sample t test was performed to compare primary and secondary outcome variables between dermatology department versus division of dermatology under the department of internal medicine. Data were analyzed using SAS version 9.2. The institutional review board at Kaiser Permanente Southern California approved this study.
Results
Due to space considerations, analyses are based on data that are not published in this article. Data regarding the characteristics of each residency program are available from the authors.
Data from 103 dermatology residency programs were included in the analysis. Of these programs, 43% had received NIH funding in 2008 and 22% had received DF funding. Two-thirds of programs had at least 1 faculty member on the editorial boards of 6 major dermatology journals; 38% had at least 2 faculty members and 9% had at least 5 faculty members on editorial boards. One-third of programs had no faculty members on these editorial boards. Sixty-nine percent of programs had 1 or more lectures given by full-time faculty members at annual society meetings in 2008; 48% of these programs had 1 to 5 lectures, 17% had 6 to 10, and 5% had more than 10. Thirty-one percent had no faculty members lecture at these meetings. Ninety-six percent of programs had 1 or more publications from full-time faculty members in 2008; 54% of programs had 1 to 20 publications, 24% had 21 to 40 publications, 14% had 41 to 60 publications, 5% had 61 to 80 publications, and 3% had more than 81 publications. Four percent of programs had no publications. Seventy-seven percent of programs were classified as departments and 23% were classified as divisions.
Factors Correlated With Producing Full-time Faculty
The Spearman rank correlation coefficient and P value were reported for each variable (Table 1). All coefficients were positive, signifying a positive correlation. Values closer to 1 were indicative of stronger correlations. P<.05 indicated statistically significant correlations for all factors investigated. The most strongly correlated factor was the ratio of faculty to residents in 2008, followed by number of full-time faculty, number of full-time faculty publications, number of lectures from full-time faculty, and number of faculty on editorial boards. The amount of NIH and DF funding received as well as total number of residents in 2008 also were correlated.
A 2-sample t test was performed to compare the number of graduates pursuing careers in academia from departments versus divisions, but the results were not statistically significant (P=.92).
Ranking Individual Programs With the Highest Number of Graduates Pursuing Academic Careers
The top 5 dermatology residency programs with the highest ratio of full-time faculty members graduated to the estimated total number of graduates were Brown University (Providence, Rhode Island), Loyola University (Chicago, Illinois), UMDNJ-Robert Wood Johnson Medical School at Camden (Camden, New Jersey), Harvard University (Cambridge, Massachusetts), and the University of Vermont (Burlington, Vermont)(Table 2). If faculty members spent time at more than one residency program, they were given credit for time spent at each program. For this reason, not all numbers are integers.
Ranking Individual Programs With the Highest Number of Full-time Faculty Members Who Completed Residency at the Same Institution
The top 10 institutions with the highest percentage of full-time faculty members who completed their residency at the same institution are shown in Table 3. Most of the programs with higher percentages are small programs. Fourteen programs had no faculty members who completed residency at the same program (data not shown).
Comment
Although this study focused on US dermatology residency programs, a shortage of academic dermatologists has been noted worldwide.11-13 By determining residency program variables associated with the pursuit of academic careers, individual programs may be able to make changes that would encourage residents to pursue careers in academia following graduation.
The factor most strongly correlated with graduates pursuing a career in academics was the ratio of faculty to residents in 2008. Also highly correlated was total number of full-time faculty members. We hypothesize that programs with more faculty members may provide better mentorship for residents. A study by Reck et al10 demonstrated that a lack of mentorship was associated with residents’ loss of interest in academics. A survey of residency program directors demonstrated that mentorship played a role in career development and that it was important for residents to have mentors.14 Given the shortage of academic faculty, increasing the number of faculty may not be feasible for individual programs; however, assuming that more faculty members is a marker for mentorship, there are many ways that individual programs may improve mentorship. There are ample opportunities in clinics to demonstrate to residents the value of research to patient care.13 Residency programs also could establish mentorship programs, pairing residents with individual faculty members who share similar interests. Some programs currently have such mentorship programs but many do not.
Also strongly correlated with the number of graduates pursuing careers in academia was the number of full-time faculty publications. It is presumed that these programs also have published extensively in the past, which may have positively influenced the programs’ residents toward academics. This factor can be easily addressed among individual residency programs, and in fact many residency programs do encourage or require residents to publish during their residency. Exposure to the process of collecting data and writing manuscripts can bolster a resident’s interest in academics. In a prior study by Wu et al,5 publications were counted multiple times if multiple faculty members were authors. This limitation was addressed by only counting each manuscript once.
Other variables correlated with the number of graduates pursuing academic careers included number of lectures from full-time faculty members at annual society meetings, number of full-time faculty on editorial boards, and amount of NIH and DF funding received. All of these variables represent the importance of establishing an academic environment and promoting dermatology research. A 2009 study by Lim and Kimball,15 which also evaluated factors associated with pursuing a career in academics, demonstrated that the number of publications prior to residency and volunteerism were associated with an academic career choice. Residents with an MD/PhD were more likely to pursue a career in academics, which also was demonstrated by a similar study in 2008.16
Wu et al5 demonstrated similar results using data from 2001 to 2004; however, amount of NIH and DF funding received was found to be negatively correlated with graduates pursuing a career in academics in the original study. This finding was surprising but was not replicated in the current study. In the current study, the amount of funding (in dollars) rather than number of grants was analyzed because it was felt that the amount of funding received was a better reflection of the quantity of research being conducted. The prior study examined the relationship between the number of grants received and the number of graduates pursuing a career in academics. It is unclear why results varied in the 2 studies, but the most recent data are consistent with our hypothesis that increased research funding is associated with more residents pursuing careers in academia.
The current study also has some limitations. It is a retrospective observational study looking at data from 2008. Choosing data from another study period may have provided different results; however, it is reassuring that the data from the current study were very similar to a prior study looking at data from 2001 to 2004.5
It is assumed that residency program characteristics remained constant over time. It was also assumed that the number of residents in a program remained constant over time. These assumptions were necessary to draw conclusions about the data. It may be that some programs have changed substantially over time, which was not accounted for in the current study. To estimate the total number of graduates, it was assumed that faculty did not practice for more than 35 years, which may not have been true; if the faculty member practiced more than 35 years, it would have altered our estimates. It is also assumed that data on residency programs’ Web sites at time of data collection were updated and accurate. It is likely that data were not 100% accurate on all Web sites at the time of data collection. However, verification of accuracy of data would have been cumbersome, and it would have been difficult to get participation from all programs.
This study does not differentiate the total number of new faculty members who join a residency program and those who are retained for many years. Encouraging residents to pursue a career in academics may increase the number of academic faculty.17 However, efforts must also be placed on retention of faculty members because many of the newly graduated residents who enter academics ultimately leave.18,19
Conclusion
In conclusion, our data suggest that programs with more faculty members may encourage residents to enter careers in academia following graduation. Additionally, the number of publications increases the likelihood of residents pursuing academic careers. By providing mentorship and research opportunities to residents, perhaps residency programs can encourage their graduates to become academic dermatologists. A program’s overall academic environment, including faculty lectures at annual society meetings, faculty on editorial boards, and increased research funding are all associated with graduates pursuing careers in academics.
Ackowledgements—We thank all of the program coordinators, full-time faculty members, program directors, chairs, and chiefs who were kind enough to field our questions if we had any missing data about their programs. We would like to thank Mary H. Black, PhD (Los Angeles, California), for her contribution to the analytical plan.
APPENDIX
1. Wheeler CE Jr, Briggaman RA, Lynch PJ, et al. Shortage of full-time faculty in dermatology. Arch Dermatol. 1973;107:529-532.
2. Wheeler CE Jr, Briggaman RA, Caro I. Shortage of full-time faculty in dermatology. Arch Dermatol. 1977;113:297-301.
3. Resneck J. Too few or too many academic dermatologists? difficulties in assessing optimal workforce size. Arch Dermatol. 2001;137:1295-1301.
4. Kia KF, Gielczyk RA, Ellis CN. Academia is the life for me, I’m sure. Arch Dermatol. 2006;142:911-913.
5. Wu JJ, Ramirez CC, Alonso CA, et al. Dermatology residency program characteristics that correlate with graduates selecting an academic dermatology career. Arch Dermatol. 2006;142:845-850.
6. Wu JJ, Tyring SK. The academic strength of current dermatology residency applicants. Dermatol Online J. 2003;9:22.
7. Hinchman KF, Wu JJ. Decisions in choosing a career in academic dermatology. Cutis. 2008;82:368-371.
8. Resneck JS Jr, Tierney EP, Kimball AB. Challenges facing academic dermatology: survey data on the faculty workforce. J Am Acad Dermatol. 2006;54:211-216.
9. Rubenstein DS, Blauvelt A, Chen SC, et al. The future of academic dermatology in the United States: report on the resident retreat for future physician-scientists, June 15-17, 2001. J Am Acad Dermatol. 2002;47:300-303.
10. Reck SJ, Stratman EJ, Vogel C, et al. Assessment of residents’ loss of interest in academic careers and identification of correctable factors. Arch Dermatol. 2006;142:855-858.
11. Olerud JE. Academic workforce in dermatology. Arch Dermatol. 2007;143:409-410.
12. Singer N. More doctors turning to the business of beauty. New York Times. November 30, 2006:A1.
13. Dogra S. Fate of medical dermatology in the era of cosmetic dermatology and dermatosurgery. Indian J Dermatol Venereol Leprol. 2009;75:4-7.
14. Donovan JC. A survey of dermatology residency program directors’ views on mentorship. Dermatol Online J. 2009;15:1.
15. Lim JL, Kimball AB. Residency applications and identification of factors associated with residents’ ultimate career decisions. Arch Dermatol. 2009;145:943-944.
16. Wu JJ, Davis KF, Ramirez CC, et al. MD/PhDs are more likely than MDs to choose a career in academics. Dermatol Online J. 2008;14:27.
17. Wu JJ. Current strategies to address the ongoing shortage of academic dermatologists. J Am Acad Dermatol. 2007;56:1065-1066.
18. Loo DS, Liu CL, Geller AC, et al. Academic dermatology manpower. Arch Dermatol. 2007;143:341-347.
19. Turner E, Yoo J, Salter S, et al. Leadership workforce in academic dermatology. Arch Dermatol. 2007;143:948-949.
1. Wheeler CE Jr, Briggaman RA, Lynch PJ, et al. Shortage of full-time faculty in dermatology. Arch Dermatol. 1973;107:529-532.
2. Wheeler CE Jr, Briggaman RA, Caro I. Shortage of full-time faculty in dermatology. Arch Dermatol. 1977;113:297-301.
3. Resneck J. Too few or too many academic dermatologists? difficulties in assessing optimal workforce size. Arch Dermatol. 2001;137:1295-1301.
4. Kia KF, Gielczyk RA, Ellis CN. Academia is the life for me, I’m sure. Arch Dermatol. 2006;142:911-913.
5. Wu JJ, Ramirez CC, Alonso CA, et al. Dermatology residency program characteristics that correlate with graduates selecting an academic dermatology career. Arch Dermatol. 2006;142:845-850.
6. Wu JJ, Tyring SK. The academic strength of current dermatology residency applicants. Dermatol Online J. 2003;9:22.
7. Hinchman KF, Wu JJ. Decisions in choosing a career in academic dermatology. Cutis. 2008;82:368-371.
8. Resneck JS Jr, Tierney EP, Kimball AB. Challenges facing academic dermatology: survey data on the faculty workforce. J Am Acad Dermatol. 2006;54:211-216.
9. Rubenstein DS, Blauvelt A, Chen SC, et al. The future of academic dermatology in the United States: report on the resident retreat for future physician-scientists, June 15-17, 2001. J Am Acad Dermatol. 2002;47:300-303.
10. Reck SJ, Stratman EJ, Vogel C, et al. Assessment of residents’ loss of interest in academic careers and identification of correctable factors. Arch Dermatol. 2006;142:855-858.
11. Olerud JE. Academic workforce in dermatology. Arch Dermatol. 2007;143:409-410.
12. Singer N. More doctors turning to the business of beauty. New York Times. November 30, 2006:A1.
13. Dogra S. Fate of medical dermatology in the era of cosmetic dermatology and dermatosurgery. Indian J Dermatol Venereol Leprol. 2009;75:4-7.
14. Donovan JC. A survey of dermatology residency program directors’ views on mentorship. Dermatol Online J. 2009;15:1.
15. Lim JL, Kimball AB. Residency applications and identification of factors associated with residents’ ultimate career decisions. Arch Dermatol. 2009;145:943-944.
16. Wu JJ, Davis KF, Ramirez CC, et al. MD/PhDs are more likely than MDs to choose a career in academics. Dermatol Online J. 2008;14:27.
17. Wu JJ. Current strategies to address the ongoing shortage of academic dermatologists. J Am Acad Dermatol. 2007;56:1065-1066.
18. Loo DS, Liu CL, Geller AC, et al. Academic dermatology manpower. Arch Dermatol. 2007;143:341-347.
19. Turner E, Yoo J, Salter S, et al. Leadership workforce in academic dermatology. Arch Dermatol. 2007;143:948-949.
Practice Points
- Because of the shortage of academic dermatologists, dermatology residency programs may wish to create an environment that will produce residents who are more likely to pursue careers in academia.
- Dermatology residency programs with more faculty members may encourage residents to enter careers in academia following graduation.
‘Screen-time transferential interference’ in encounters with patients
I cannot recall the last time that I had a good look at the cashier who was scanning my grocery purchases. I could not tell you what color eyes he (or she?) had or how he styled his hair. This isn’t for lack of an effort to recall, or a manifestation of poor memory or absentmindedness. Rather, I think that the situation reflects a larger cultural shift that has gained momentum since the beginning of the new century: that is, the effect of a preponderance of so-called screen time in our lives.
In that mundane scene in the grocery store, screen time encompasses the impersonal and mechanical act of swiping my debit card, entering my PIN, and impatiently waiting for the receipt to print. All the while, I stand awkwardly, eyes downcast and fixed on the display of the card reader, ignoring the human being directly across from me.
Obsession with screens
Our engagement with screen time has grown to pandemic proportions, and television is no longer the main culprit. According to a Nielsen global consumer report,1 in 2010 in the United States, people spent an average of 5 hours a day in front of the “boob tube.” Even if we take that statistic with a grain of salt, it still represents only the most visible tip of the media iceberg. Smartphones, laptop and desktop monitors, portable gaming consoles, electronic tablets, PIN pad displays, video billboards, and any number of other LED and LCD screen surfaces have infiltrated the landscape.
Whereas most recent epidemiologic studies have addressed the deleterious effects of so-called sit time (sedentary activities with or without a screen) on physical health, I would like to address the deleterious effect of screen time on mental health and relational connectedness and the relevance of that screen time to psychiatric practice.
The ‘techno-bubble of private space’
Almond,2 in a humorous social commentary, “Connection Error,” conducted an impromptu experiment in which he attempted to connect spontaneously with strangers, especially those who had a smartphone, in Boston. His narrative navigates the gamut of human interaction, from tedious and boorish to comedic and absurd, noting that, conspicuously, “smartphone users have created a techno-bubble of private space” in which they are physically present but emotionally unavailable.
A chance encounter with a young professional led Almond to this conclusion:
It’s precisely the intrusive alienation of the “techno-bubble” that blunders into the modern patient-physician interaction in my clinical psychiatric practice in a busy outpatient clinic at a university medical center. Specifically, the ever-glowing, ever-distracting computer monitor sitting between me and my patient, with its promise of digital information at my fingertips, serves more to distance me from my patient than to connect us in a meaningful, human way. Just as I can’t recall the countenance of the grocery-store cashier, I miss the delicate, information-laden, minute-to-minute social interaction with the patient because it competes with the electronic intruder.
What’s at risk when a computer screen is in the room?
Transference in the psychotherapeutic encounter is an established tenet of psychoanalytic theory. In “Basic theory of psychoanalysis,”3 Waelder defines transference as “not simply the attribution to new objects of characteristics of old ones but the attempt to re-establish and relive, with whatever object will permit it, an infantile situation much longed for because it was once either greatly enjoyed or greatly missed.” This definition applies to the positive pole of transferential phenomena—and it is this position that is desired in a successful patient−physician encounter.
A patient’s warm and genial regard toward a provider secures trust, cooperation, and faith in the healing process. Establishment of positive transference toward the physician is essential to enhance the clinical encounter, regardless of what early object (caring mother, omnipotent father) is being projected onto the physician.
Attunement. Research into infant observation has revealed the critical role of caretaker responsiveness in the development of early infantile emotional regulation. Tronick et al4 demonstrated the importance of interactional reciprocity in the mother−child dyad.
In a series of experiments using the so-called still-face paradigm, Tronick et al4 saw that infants quickly fall into a state of despair and related negative affects when the mother assumes an unresponsive and detached still face. These episodes intentionally produce infant-mother emotional misattunement, which, although instantly damaging, can be successfully repaired through re-attunement by the mother. It is the primary caretaker’s ability to reconnect and repair that is paramount to the infant’s healthy psychological development.
This sentiment is echoed in Winnicott’s concept of the “good-enough” mother (or parent), formulated years earlier, in which failures in infant−caretaker attunement are inevitable and to be expected—as long as repair outcompetes deficiency.5
Divided attention: Patient or screen? Or both?
What we can understand by applying the ideas of transference and optimal attunement to the clinical encounter is how important uninterrupted face-to-face time with the patient is. Indeed, nonverbal communication from the patient, expressed though body language and facial articulation, is particularly salient to the practice of psychiatry. Information technology— especially the electronic health record—now encroaches on the time-honored central dyad of the patient-physician interaction by introducing a third entity into the traditional encounter.
Clinical misattunement, as understood through the still-face paradigm, increases in proportion to a provider’s need to divide his (her) attention between the patient and the computer screen. And, as the degree of misattunement increases, positive transference is more difficult to establish and maintain. The quality of the clinical encounter then deteriorates, undermining the care of the patient and reducing physician satisfaction.
Acknowledgment
Philip LeFevre, MD, Department of Neurology & Psychiatry, Saint Louis University, St. Louis, Missouri, provided inspiration and encouragement in the development of the manuscript.
Disclosure
Dr. Afanasevich reports no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
1. The Nielsen Company. How people watch: a global Nielsen consumer report. http://www.nielsen.com/content/dam/ corporate/mx/reports/2011/Lo-que-la-gente-ve.pdf. Published August 2010. Accessed March 17, 2015.
2. Almond S. Connection error. Spirit Magazine. April 2014:76-86.
3. Waelder R. Basic theory of psychoanalysis. New York, NY: International Universities Press; 1960.
4. Tronick E, Als H, Adamson L, et al. The infant’s response to entrapment between contradictory messages in face-to-face interaction. J Am Acad Child Psychiatry. 1978;17(1):1-13.
5. Winnicott DW. The child, the family, and the outside world. London, United Kingdom: Penguin Books; 1964.
I cannot recall the last time that I had a good look at the cashier who was scanning my grocery purchases. I could not tell you what color eyes he (or she?) had or how he styled his hair. This isn’t for lack of an effort to recall, or a manifestation of poor memory or absentmindedness. Rather, I think that the situation reflects a larger cultural shift that has gained momentum since the beginning of the new century: that is, the effect of a preponderance of so-called screen time in our lives.
In that mundane scene in the grocery store, screen time encompasses the impersonal and mechanical act of swiping my debit card, entering my PIN, and impatiently waiting for the receipt to print. All the while, I stand awkwardly, eyes downcast and fixed on the display of the card reader, ignoring the human being directly across from me.
Obsession with screens
Our engagement with screen time has grown to pandemic proportions, and television is no longer the main culprit. According to a Nielsen global consumer report,1 in 2010 in the United States, people spent an average of 5 hours a day in front of the “boob tube.” Even if we take that statistic with a grain of salt, it still represents only the most visible tip of the media iceberg. Smartphones, laptop and desktop monitors, portable gaming consoles, electronic tablets, PIN pad displays, video billboards, and any number of other LED and LCD screen surfaces have infiltrated the landscape.
Whereas most recent epidemiologic studies have addressed the deleterious effects of so-called sit time (sedentary activities with or without a screen) on physical health, I would like to address the deleterious effect of screen time on mental health and relational connectedness and the relevance of that screen time to psychiatric practice.
The ‘techno-bubble of private space’
Almond,2 in a humorous social commentary, “Connection Error,” conducted an impromptu experiment in which he attempted to connect spontaneously with strangers, especially those who had a smartphone, in Boston. His narrative navigates the gamut of human interaction, from tedious and boorish to comedic and absurd, noting that, conspicuously, “smartphone users have created a techno-bubble of private space” in which they are physically present but emotionally unavailable.
A chance encounter with a young professional led Almond to this conclusion:
It’s precisely the intrusive alienation of the “techno-bubble” that blunders into the modern patient-physician interaction in my clinical psychiatric practice in a busy outpatient clinic at a university medical center. Specifically, the ever-glowing, ever-distracting computer monitor sitting between me and my patient, with its promise of digital information at my fingertips, serves more to distance me from my patient than to connect us in a meaningful, human way. Just as I can’t recall the countenance of the grocery-store cashier, I miss the delicate, information-laden, minute-to-minute social interaction with the patient because it competes with the electronic intruder.
What’s at risk when a computer screen is in the room?
Transference in the psychotherapeutic encounter is an established tenet of psychoanalytic theory. In “Basic theory of psychoanalysis,”3 Waelder defines transference as “not simply the attribution to new objects of characteristics of old ones but the attempt to re-establish and relive, with whatever object will permit it, an infantile situation much longed for because it was once either greatly enjoyed or greatly missed.” This definition applies to the positive pole of transferential phenomena—and it is this position that is desired in a successful patient−physician encounter.
A patient’s warm and genial regard toward a provider secures trust, cooperation, and faith in the healing process. Establishment of positive transference toward the physician is essential to enhance the clinical encounter, regardless of what early object (caring mother, omnipotent father) is being projected onto the physician.
Attunement. Research into infant observation has revealed the critical role of caretaker responsiveness in the development of early infantile emotional regulation. Tronick et al4 demonstrated the importance of interactional reciprocity in the mother−child dyad.
In a series of experiments using the so-called still-face paradigm, Tronick et al4 saw that infants quickly fall into a state of despair and related negative affects when the mother assumes an unresponsive and detached still face. These episodes intentionally produce infant-mother emotional misattunement, which, although instantly damaging, can be successfully repaired through re-attunement by the mother. It is the primary caretaker’s ability to reconnect and repair that is paramount to the infant’s healthy psychological development.
This sentiment is echoed in Winnicott’s concept of the “good-enough” mother (or parent), formulated years earlier, in which failures in infant−caretaker attunement are inevitable and to be expected—as long as repair outcompetes deficiency.5
Divided attention: Patient or screen? Or both?
What we can understand by applying the ideas of transference and optimal attunement to the clinical encounter is how important uninterrupted face-to-face time with the patient is. Indeed, nonverbal communication from the patient, expressed though body language and facial articulation, is particularly salient to the practice of psychiatry. Information technology— especially the electronic health record—now encroaches on the time-honored central dyad of the patient-physician interaction by introducing a third entity into the traditional encounter.
Clinical misattunement, as understood through the still-face paradigm, increases in proportion to a provider’s need to divide his (her) attention between the patient and the computer screen. And, as the degree of misattunement increases, positive transference is more difficult to establish and maintain. The quality of the clinical encounter then deteriorates, undermining the care of the patient and reducing physician satisfaction.
Acknowledgment
Philip LeFevre, MD, Department of Neurology & Psychiatry, Saint Louis University, St. Louis, Missouri, provided inspiration and encouragement in the development of the manuscript.
Disclosure
Dr. Afanasevich reports no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
I cannot recall the last time that I had a good look at the cashier who was scanning my grocery purchases. I could not tell you what color eyes he (or she?) had or how he styled his hair. This isn’t for lack of an effort to recall, or a manifestation of poor memory or absentmindedness. Rather, I think that the situation reflects a larger cultural shift that has gained momentum since the beginning of the new century: that is, the effect of a preponderance of so-called screen time in our lives.
In that mundane scene in the grocery store, screen time encompasses the impersonal and mechanical act of swiping my debit card, entering my PIN, and impatiently waiting for the receipt to print. All the while, I stand awkwardly, eyes downcast and fixed on the display of the card reader, ignoring the human being directly across from me.
Obsession with screens
Our engagement with screen time has grown to pandemic proportions, and television is no longer the main culprit. According to a Nielsen global consumer report,1 in 2010 in the United States, people spent an average of 5 hours a day in front of the “boob tube.” Even if we take that statistic with a grain of salt, it still represents only the most visible tip of the media iceberg. Smartphones, laptop and desktop monitors, portable gaming consoles, electronic tablets, PIN pad displays, video billboards, and any number of other LED and LCD screen surfaces have infiltrated the landscape.
Whereas most recent epidemiologic studies have addressed the deleterious effects of so-called sit time (sedentary activities with or without a screen) on physical health, I would like to address the deleterious effect of screen time on mental health and relational connectedness and the relevance of that screen time to psychiatric practice.
The ‘techno-bubble of private space’
Almond,2 in a humorous social commentary, “Connection Error,” conducted an impromptu experiment in which he attempted to connect spontaneously with strangers, especially those who had a smartphone, in Boston. His narrative navigates the gamut of human interaction, from tedious and boorish to comedic and absurd, noting that, conspicuously, “smartphone users have created a techno-bubble of private space” in which they are physically present but emotionally unavailable.
A chance encounter with a young professional led Almond to this conclusion:
It’s precisely the intrusive alienation of the “techno-bubble” that blunders into the modern patient-physician interaction in my clinical psychiatric practice in a busy outpatient clinic at a university medical center. Specifically, the ever-glowing, ever-distracting computer monitor sitting between me and my patient, with its promise of digital information at my fingertips, serves more to distance me from my patient than to connect us in a meaningful, human way. Just as I can’t recall the countenance of the grocery-store cashier, I miss the delicate, information-laden, minute-to-minute social interaction with the patient because it competes with the electronic intruder.
What’s at risk when a computer screen is in the room?
Transference in the psychotherapeutic encounter is an established tenet of psychoanalytic theory. In “Basic theory of psychoanalysis,”3 Waelder defines transference as “not simply the attribution to new objects of characteristics of old ones but the attempt to re-establish and relive, with whatever object will permit it, an infantile situation much longed for because it was once either greatly enjoyed or greatly missed.” This definition applies to the positive pole of transferential phenomena—and it is this position that is desired in a successful patient−physician encounter.
A patient’s warm and genial regard toward a provider secures trust, cooperation, and faith in the healing process. Establishment of positive transference toward the physician is essential to enhance the clinical encounter, regardless of what early object (caring mother, omnipotent father) is being projected onto the physician.
Attunement. Research into infant observation has revealed the critical role of caretaker responsiveness in the development of early infantile emotional regulation. Tronick et al4 demonstrated the importance of interactional reciprocity in the mother−child dyad.
In a series of experiments using the so-called still-face paradigm, Tronick et al4 saw that infants quickly fall into a state of despair and related negative affects when the mother assumes an unresponsive and detached still face. These episodes intentionally produce infant-mother emotional misattunement, which, although instantly damaging, can be successfully repaired through re-attunement by the mother. It is the primary caretaker’s ability to reconnect and repair that is paramount to the infant’s healthy psychological development.
This sentiment is echoed in Winnicott’s concept of the “good-enough” mother (or parent), formulated years earlier, in which failures in infant−caretaker attunement are inevitable and to be expected—as long as repair outcompetes deficiency.5
Divided attention: Patient or screen? Or both?
What we can understand by applying the ideas of transference and optimal attunement to the clinical encounter is how important uninterrupted face-to-face time with the patient is. Indeed, nonverbal communication from the patient, expressed though body language and facial articulation, is particularly salient to the practice of psychiatry. Information technology— especially the electronic health record—now encroaches on the time-honored central dyad of the patient-physician interaction by introducing a third entity into the traditional encounter.
Clinical misattunement, as understood through the still-face paradigm, increases in proportion to a provider’s need to divide his (her) attention between the patient and the computer screen. And, as the degree of misattunement increases, positive transference is more difficult to establish and maintain. The quality of the clinical encounter then deteriorates, undermining the care of the patient and reducing physician satisfaction.
Acknowledgment
Philip LeFevre, MD, Department of Neurology & Psychiatry, Saint Louis University, St. Louis, Missouri, provided inspiration and encouragement in the development of the manuscript.
Disclosure
Dr. Afanasevich reports no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
1. The Nielsen Company. How people watch: a global Nielsen consumer report. http://www.nielsen.com/content/dam/ corporate/mx/reports/2011/Lo-que-la-gente-ve.pdf. Published August 2010. Accessed March 17, 2015.
2. Almond S. Connection error. Spirit Magazine. April 2014:76-86.
3. Waelder R. Basic theory of psychoanalysis. New York, NY: International Universities Press; 1960.
4. Tronick E, Als H, Adamson L, et al. The infant’s response to entrapment between contradictory messages in face-to-face interaction. J Am Acad Child Psychiatry. 1978;17(1):1-13.
5. Winnicott DW. The child, the family, and the outside world. London, United Kingdom: Penguin Books; 1964.
1. The Nielsen Company. How people watch: a global Nielsen consumer report. http://www.nielsen.com/content/dam/ corporate/mx/reports/2011/Lo-que-la-gente-ve.pdf. Published August 2010. Accessed March 17, 2015.
2. Almond S. Connection error. Spirit Magazine. April 2014:76-86.
3. Waelder R. Basic theory of psychoanalysis. New York, NY: International Universities Press; 1960.
4. Tronick E, Als H, Adamson L, et al. The infant’s response to entrapment between contradictory messages in face-to-face interaction. J Am Acad Child Psychiatry. 1978;17(1):1-13.
5. Winnicott DW. The child, the family, and the outside world. London, United Kingdom: Penguin Books; 1964.
Fellowships After Dermatology Residency: The Traditional and Beyond
Dermatology residents, such as myself, often wonder what we will do after graduation. There are many resources for finding job opportunities, and many of us have received solicitation e-mails from various headhunters and medical groups that are looking to hire. The American Academy of Dermatology (AAD) has a resource called the AAD Career Compass (http://www.healthecareers.com/aad), which is an exhaustive database of job listings for dermatologists. However, I could not locate a definitive resource containing information that might be useful for dermatology residents who are interested in subspecializing or pursuing fellowships.
Subspecialty training is typically pursued after successful completion of a dermatology residency training program. Fellowships are traditionally offered in dermatopathology, pediatric dermatology, micrographic surgery and dermatologic oncology (procedural dermatology), and cosmetic dermatologic surgery. Fellowships also are available in other subspecialties or for those pursuing an academic career. The goal of this article is to help dermatology residents learn more about traditional and nontraditional opportunities for graduate education and certification in various dermatologic subspecialties, with links to sources for more detailed information.
Traditional Fellowship Programs by Subspecialty
Dermatopathology
One- to 2-year dermatopathology fellowship programs are certified by both the American Board of Dermatology (ABD) and the American Board of Pathology and are available to graduates of either dermatology or pathology residency programs. These programs offer combined training in either anatomic pathology (for dermatologists) or clinical dermatology (for pathologists), along with dermatopathology; the majority of time is devoted to the latter. The Accreditation Council for Graduate Medical Education and the ABD have issued specific requirements for graduate medical education and subspecialty certification in dermatopathology.1,2 Fellowship matches are institution dependent, and the application process and match generally takes place during the second year of dermatology residency for those residents who want to start a fellowship program immediately following graduation. The American Society of Dermatopathology offers a dermatopathology fellowship program finder on its Web site.
Pediatric Dermatology
Fellowships in pediatric dermatology are typically 1- to 2-year programs that focus on dermatologic diseases in the pediatric population. Applicants are matched to these programs through the San Francisco Matching Program (SF Match) and the programs are ABD accredited.3,4 (There also are a number of non–ABD-approved training opportunities available.5) On completion of the training program, fellows may qualify for subspecialty board certification in pediatric dermatology. Applications are open starting in January, and the rank order list and match occur in August of the same year. As of 2012, there were 20 participating programs with 28 available positions, while the match included 22 applicants; of these applicants, 15 matched formally into pediatric dermatology fellowships.6
Micrographic Surgery and Dermatologic Oncology (Procedural Dermatology)
There are specific requirements issued by the Accreditation Council for Graduate Medical Education for dermatologic surgery fellowships,7 which are typically 1- to 2-year programs. Many fellowship programs also are accredited by the American College of Mohs Surgery. This subspecialty is not ABD accredited; therefore, there is no certification process upon completion of a fellowship program. The American College of Mohs Surgery sponsors the match process through SF Match. Applicant registration begins in July and the match occurs in December of the same year. As of 2013, there were 47 participating programs offering 55 positions. Of 77 applicants, 49 obtained fellowship positions formally through the match.8 The American Society for Dermatologic Surgery (ASDS) Web site provides the DermSurg Fellowship Finder, which includes information about independent fellowship programs.
Cosmetic Dermatologic Surgery
The ASDS has an accreditation program for fellowships in cosmetic dermatologic surgery,9 which are generally 1-year programs. Certification in this subspecialty is not ABD accredited. Fellowship opportunities can be found using the ASDS DermSurg Fellowship Finder.
Nontraditional Fellowship Programs
The following are fellowship programs that are in nontraditional subspecialties, are only available at certain institutions, and are not accredited. This list is not exhaustive of all available programs but are those that may be of interest to dermatology residents who are drawn to a particular dermatologic subspecialty or have an interest in academic dermatology. There is no formal match process and applications vary by institution.
Clinician Educator Fellowship
The clinician educator fellowship is available at the Department of Dermatology at the University of Pennsylvania (Philadelphia, Pennsylvania) and is intended to foster dermatologic clinician educators. More information can be found on the program’s Web site.
Cutaneous Oncology Fellowship
This 1- to 2-year fellowship program focuses on diagnosis and management of melanoma and nonmelanoma skin cancers as well as cutaneous lymphomas. Fellowships in cutaneous oncology are offered at the University of California, San Francisco (San Francisco, California), Brigham and Women’s Hospital (Boston, Massachusetts), the University of Pennsylvania (Philadelphia, Pennsylvania), Case Western Reserve University (Cleveland, Ohio), the University of Pittsburgh (Pittsburgh, Pennsylvania), and Stanford University Medical Center (Stanford, California).
Dermatology/Rheumatology
The dermatology/rheumatology fellowship offered by Brigham and Women’s Hospital is a 1-year program that focuses on the management of connective-tissue diseases in a multidisciplinary fashion with rheumatology.
Advanced Medical Dermatology/Complex Medical Dermatology
Several programs offer fellowships in medical dermatology under different titles but with a similar curriculum and goal: to foster dermatologists interested in careers as academic medical dermatologists or as future clinician scientists by means of specialized training and mentorship in complex medical and dermatological issues in the outpatient and inpatient settings. The 2-year program at New York University School of Medicine (New York, New York) also gives fellows the opportunity to earn a master of science in clinical investigation degree. The University of California, San Francisco, program offers protected time for career development.
Epidemiology Training Program
Fellows and residents in the University of Pennsylvania’s dermatology training program may elect to work with the Center for Clinical Epidemiology and Biostatistics and have the opportunity to earn a graduate degree (MSCE or PhD).
Contact Dermatitis and Patch Testing Fellowship
The Dermatology Department at the Cleveland Clinic (Cleveland, Ohio) offers a 1-year contact dermatitis and patch testing fellowship that includes clinical research.
Conclusion
Both traditional and nontraditional fellowship opportunities exist after dermatology residency. This guide serves as an overview of the training programs in dermatopathology, pediatric dermatology, micrographic surgery and dermatologic oncology (procedural dermatology), and cosmetic dermatologic surgery, as well as the fellowships offered at certain institutions for those interested in more specific subspecialties or academia.
1. Accreditation Council for Graduate Medical Education. ACGME program requirements for graduate medical education in dermatopathology. http://www.acgme.org/acgmeweb/Portals/0/PFAssets/ProgramRequirements/100_dermatopathology_2016_1-YR.pdf. Revised April 2014. Effective July 1, 2015. Accessed on February 26, 2015.
2. Subspecialty certification in dermatopathology. The American Board of Dermatology, Inc Web site. http://www.abderm.org/subspecialties/derm.html. Accessed February 26, 2015.
3. American Board of Dermatology (ABD) approved pediatric dermatology fellowship programs. The Society for Pediatric Dermatology Web site. http://pedsderm.net/training/fellowships/abd-approved-pediatric-dermatology-fellowship-programs/. Updated June 9, 2014. Accessed February 26, 2015.
4. Subspecialty certification in pediatric dermatology. The American Board of Dermatology, Inc Web site. http://www.abderm.org/subspecialties/pediatric.html. Accessed February 26, 2015.
5. Non-ABD pediatric dermatology fellowship programs. The Society for Pediatric Dermatology Web site. https://pedsderm.net/training/fellowships/non-abd-pediatric-dermatology-fellowship-programs/. Accessed February 26, 2015.
6. Pediatric dermatology match report. SF Match Web site. https://www.sfmatch.org/SpecialtyInsideAll.aspx?id=16&typ=1&name=Pediatric%20Dermatology#. Accessed March 4, 2015.
7. Accreditation Council for Graduate Medical Education. ACGME program requirements for graduate medical education in procedural dermatology. https://www.acgme.org/acgmeweb/Portals/0/PFAssets/ProgramRequirements/081_procedural_derm_1-YR_07012014.pdf. Effective July 2014. Accessed February 26, 2015.
8. Statistics: micrographic surgery & dermatologic oncology fellowship. SF Match Web site. https://www.sfmatch.org/SpecialtyInsideAll.aspx?id=10&typ=1&name=Micrographic%20Surgery%20and%20Dermatologic%20Oncology#. Accessed March 4, 2015.
9. ASDS cosmetic dermatologic surgery fellowship accreditation program. American Society for Dermatologic Surgery Web site. http://www.asds.net/cosmetic-accreditation/. Accessed February 26, 2015.
Dermatology residents, such as myself, often wonder what we will do after graduation. There are many resources for finding job opportunities, and many of us have received solicitation e-mails from various headhunters and medical groups that are looking to hire. The American Academy of Dermatology (AAD) has a resource called the AAD Career Compass (http://www.healthecareers.com/aad), which is an exhaustive database of job listings for dermatologists. However, I could not locate a definitive resource containing information that might be useful for dermatology residents who are interested in subspecializing or pursuing fellowships.
Subspecialty training is typically pursued after successful completion of a dermatology residency training program. Fellowships are traditionally offered in dermatopathology, pediatric dermatology, micrographic surgery and dermatologic oncology (procedural dermatology), and cosmetic dermatologic surgery. Fellowships also are available in other subspecialties or for those pursuing an academic career. The goal of this article is to help dermatology residents learn more about traditional and nontraditional opportunities for graduate education and certification in various dermatologic subspecialties, with links to sources for more detailed information.
Traditional Fellowship Programs by Subspecialty
Dermatopathology
One- to 2-year dermatopathology fellowship programs are certified by both the American Board of Dermatology (ABD) and the American Board of Pathology and are available to graduates of either dermatology or pathology residency programs. These programs offer combined training in either anatomic pathology (for dermatologists) or clinical dermatology (for pathologists), along with dermatopathology; the majority of time is devoted to the latter. The Accreditation Council for Graduate Medical Education and the ABD have issued specific requirements for graduate medical education and subspecialty certification in dermatopathology.1,2 Fellowship matches are institution dependent, and the application process and match generally takes place during the second year of dermatology residency for those residents who want to start a fellowship program immediately following graduation. The American Society of Dermatopathology offers a dermatopathology fellowship program finder on its Web site.
Pediatric Dermatology
Fellowships in pediatric dermatology are typically 1- to 2-year programs that focus on dermatologic diseases in the pediatric population. Applicants are matched to these programs through the San Francisco Matching Program (SF Match) and the programs are ABD accredited.3,4 (There also are a number of non–ABD-approved training opportunities available.5) On completion of the training program, fellows may qualify for subspecialty board certification in pediatric dermatology. Applications are open starting in January, and the rank order list and match occur in August of the same year. As of 2012, there were 20 participating programs with 28 available positions, while the match included 22 applicants; of these applicants, 15 matched formally into pediatric dermatology fellowships.6
Micrographic Surgery and Dermatologic Oncology (Procedural Dermatology)
There are specific requirements issued by the Accreditation Council for Graduate Medical Education for dermatologic surgery fellowships,7 which are typically 1- to 2-year programs. Many fellowship programs also are accredited by the American College of Mohs Surgery. This subspecialty is not ABD accredited; therefore, there is no certification process upon completion of a fellowship program. The American College of Mohs Surgery sponsors the match process through SF Match. Applicant registration begins in July and the match occurs in December of the same year. As of 2013, there were 47 participating programs offering 55 positions. Of 77 applicants, 49 obtained fellowship positions formally through the match.8 The American Society for Dermatologic Surgery (ASDS) Web site provides the DermSurg Fellowship Finder, which includes information about independent fellowship programs.
Cosmetic Dermatologic Surgery
The ASDS has an accreditation program for fellowships in cosmetic dermatologic surgery,9 which are generally 1-year programs. Certification in this subspecialty is not ABD accredited. Fellowship opportunities can be found using the ASDS DermSurg Fellowship Finder.
Nontraditional Fellowship Programs
The following are fellowship programs that are in nontraditional subspecialties, are only available at certain institutions, and are not accredited. This list is not exhaustive of all available programs but are those that may be of interest to dermatology residents who are drawn to a particular dermatologic subspecialty or have an interest in academic dermatology. There is no formal match process and applications vary by institution.
Clinician Educator Fellowship
The clinician educator fellowship is available at the Department of Dermatology at the University of Pennsylvania (Philadelphia, Pennsylvania) and is intended to foster dermatologic clinician educators. More information can be found on the program’s Web site.
Cutaneous Oncology Fellowship
This 1- to 2-year fellowship program focuses on diagnosis and management of melanoma and nonmelanoma skin cancers as well as cutaneous lymphomas. Fellowships in cutaneous oncology are offered at the University of California, San Francisco (San Francisco, California), Brigham and Women’s Hospital (Boston, Massachusetts), the University of Pennsylvania (Philadelphia, Pennsylvania), Case Western Reserve University (Cleveland, Ohio), the University of Pittsburgh (Pittsburgh, Pennsylvania), and Stanford University Medical Center (Stanford, California).
Dermatology/Rheumatology
The dermatology/rheumatology fellowship offered by Brigham and Women’s Hospital is a 1-year program that focuses on the management of connective-tissue diseases in a multidisciplinary fashion with rheumatology.
Advanced Medical Dermatology/Complex Medical Dermatology
Several programs offer fellowships in medical dermatology under different titles but with a similar curriculum and goal: to foster dermatologists interested in careers as academic medical dermatologists or as future clinician scientists by means of specialized training and mentorship in complex medical and dermatological issues in the outpatient and inpatient settings. The 2-year program at New York University School of Medicine (New York, New York) also gives fellows the opportunity to earn a master of science in clinical investigation degree. The University of California, San Francisco, program offers protected time for career development.
Epidemiology Training Program
Fellows and residents in the University of Pennsylvania’s dermatology training program may elect to work with the Center for Clinical Epidemiology and Biostatistics and have the opportunity to earn a graduate degree (MSCE or PhD).
Contact Dermatitis and Patch Testing Fellowship
The Dermatology Department at the Cleveland Clinic (Cleveland, Ohio) offers a 1-year contact dermatitis and patch testing fellowship that includes clinical research.
Conclusion
Both traditional and nontraditional fellowship opportunities exist after dermatology residency. This guide serves as an overview of the training programs in dermatopathology, pediatric dermatology, micrographic surgery and dermatologic oncology (procedural dermatology), and cosmetic dermatologic surgery, as well as the fellowships offered at certain institutions for those interested in more specific subspecialties or academia.
Dermatology residents, such as myself, often wonder what we will do after graduation. There are many resources for finding job opportunities, and many of us have received solicitation e-mails from various headhunters and medical groups that are looking to hire. The American Academy of Dermatology (AAD) has a resource called the AAD Career Compass (http://www.healthecareers.com/aad), which is an exhaustive database of job listings for dermatologists. However, I could not locate a definitive resource containing information that might be useful for dermatology residents who are interested in subspecializing or pursuing fellowships.
Subspecialty training is typically pursued after successful completion of a dermatology residency training program. Fellowships are traditionally offered in dermatopathology, pediatric dermatology, micrographic surgery and dermatologic oncology (procedural dermatology), and cosmetic dermatologic surgery. Fellowships also are available in other subspecialties or for those pursuing an academic career. The goal of this article is to help dermatology residents learn more about traditional and nontraditional opportunities for graduate education and certification in various dermatologic subspecialties, with links to sources for more detailed information.
Traditional Fellowship Programs by Subspecialty
Dermatopathology
One- to 2-year dermatopathology fellowship programs are certified by both the American Board of Dermatology (ABD) and the American Board of Pathology and are available to graduates of either dermatology or pathology residency programs. These programs offer combined training in either anatomic pathology (for dermatologists) or clinical dermatology (for pathologists), along with dermatopathology; the majority of time is devoted to the latter. The Accreditation Council for Graduate Medical Education and the ABD have issued specific requirements for graduate medical education and subspecialty certification in dermatopathology.1,2 Fellowship matches are institution dependent, and the application process and match generally takes place during the second year of dermatology residency for those residents who want to start a fellowship program immediately following graduation. The American Society of Dermatopathology offers a dermatopathology fellowship program finder on its Web site.
Pediatric Dermatology
Fellowships in pediatric dermatology are typically 1- to 2-year programs that focus on dermatologic diseases in the pediatric population. Applicants are matched to these programs through the San Francisco Matching Program (SF Match) and the programs are ABD accredited.3,4 (There also are a number of non–ABD-approved training opportunities available.5) On completion of the training program, fellows may qualify for subspecialty board certification in pediatric dermatology. Applications are open starting in January, and the rank order list and match occur in August of the same year. As of 2012, there were 20 participating programs with 28 available positions, while the match included 22 applicants; of these applicants, 15 matched formally into pediatric dermatology fellowships.6
Micrographic Surgery and Dermatologic Oncology (Procedural Dermatology)
There are specific requirements issued by the Accreditation Council for Graduate Medical Education for dermatologic surgery fellowships,7 which are typically 1- to 2-year programs. Many fellowship programs also are accredited by the American College of Mohs Surgery. This subspecialty is not ABD accredited; therefore, there is no certification process upon completion of a fellowship program. The American College of Mohs Surgery sponsors the match process through SF Match. Applicant registration begins in July and the match occurs in December of the same year. As of 2013, there were 47 participating programs offering 55 positions. Of 77 applicants, 49 obtained fellowship positions formally through the match.8 The American Society for Dermatologic Surgery (ASDS) Web site provides the DermSurg Fellowship Finder, which includes information about independent fellowship programs.
Cosmetic Dermatologic Surgery
The ASDS has an accreditation program for fellowships in cosmetic dermatologic surgery,9 which are generally 1-year programs. Certification in this subspecialty is not ABD accredited. Fellowship opportunities can be found using the ASDS DermSurg Fellowship Finder.
Nontraditional Fellowship Programs
The following are fellowship programs that are in nontraditional subspecialties, are only available at certain institutions, and are not accredited. This list is not exhaustive of all available programs but are those that may be of interest to dermatology residents who are drawn to a particular dermatologic subspecialty or have an interest in academic dermatology. There is no formal match process and applications vary by institution.
Clinician Educator Fellowship
The clinician educator fellowship is available at the Department of Dermatology at the University of Pennsylvania (Philadelphia, Pennsylvania) and is intended to foster dermatologic clinician educators. More information can be found on the program’s Web site.
Cutaneous Oncology Fellowship
This 1- to 2-year fellowship program focuses on diagnosis and management of melanoma and nonmelanoma skin cancers as well as cutaneous lymphomas. Fellowships in cutaneous oncology are offered at the University of California, San Francisco (San Francisco, California), Brigham and Women’s Hospital (Boston, Massachusetts), the University of Pennsylvania (Philadelphia, Pennsylvania), Case Western Reserve University (Cleveland, Ohio), the University of Pittsburgh (Pittsburgh, Pennsylvania), and Stanford University Medical Center (Stanford, California).
Dermatology/Rheumatology
The dermatology/rheumatology fellowship offered by Brigham and Women’s Hospital is a 1-year program that focuses on the management of connective-tissue diseases in a multidisciplinary fashion with rheumatology.
Advanced Medical Dermatology/Complex Medical Dermatology
Several programs offer fellowships in medical dermatology under different titles but with a similar curriculum and goal: to foster dermatologists interested in careers as academic medical dermatologists or as future clinician scientists by means of specialized training and mentorship in complex medical and dermatological issues in the outpatient and inpatient settings. The 2-year program at New York University School of Medicine (New York, New York) also gives fellows the opportunity to earn a master of science in clinical investigation degree. The University of California, San Francisco, program offers protected time for career development.
Epidemiology Training Program
Fellows and residents in the University of Pennsylvania’s dermatology training program may elect to work with the Center for Clinical Epidemiology and Biostatistics and have the opportunity to earn a graduate degree (MSCE or PhD).
Contact Dermatitis and Patch Testing Fellowship
The Dermatology Department at the Cleveland Clinic (Cleveland, Ohio) offers a 1-year contact dermatitis and patch testing fellowship that includes clinical research.
Conclusion
Both traditional and nontraditional fellowship opportunities exist after dermatology residency. This guide serves as an overview of the training programs in dermatopathology, pediatric dermatology, micrographic surgery and dermatologic oncology (procedural dermatology), and cosmetic dermatologic surgery, as well as the fellowships offered at certain institutions for those interested in more specific subspecialties or academia.
1. Accreditation Council for Graduate Medical Education. ACGME program requirements for graduate medical education in dermatopathology. http://www.acgme.org/acgmeweb/Portals/0/PFAssets/ProgramRequirements/100_dermatopathology_2016_1-YR.pdf. Revised April 2014. Effective July 1, 2015. Accessed on February 26, 2015.
2. Subspecialty certification in dermatopathology. The American Board of Dermatology, Inc Web site. http://www.abderm.org/subspecialties/derm.html. Accessed February 26, 2015.
3. American Board of Dermatology (ABD) approved pediatric dermatology fellowship programs. The Society for Pediatric Dermatology Web site. http://pedsderm.net/training/fellowships/abd-approved-pediatric-dermatology-fellowship-programs/. Updated June 9, 2014. Accessed February 26, 2015.
4. Subspecialty certification in pediatric dermatology. The American Board of Dermatology, Inc Web site. http://www.abderm.org/subspecialties/pediatric.html. Accessed February 26, 2015.
5. Non-ABD pediatric dermatology fellowship programs. The Society for Pediatric Dermatology Web site. https://pedsderm.net/training/fellowships/non-abd-pediatric-dermatology-fellowship-programs/. Accessed February 26, 2015.
6. Pediatric dermatology match report. SF Match Web site. https://www.sfmatch.org/SpecialtyInsideAll.aspx?id=16&typ=1&name=Pediatric%20Dermatology#. Accessed March 4, 2015.
7. Accreditation Council for Graduate Medical Education. ACGME program requirements for graduate medical education in procedural dermatology. https://www.acgme.org/acgmeweb/Portals/0/PFAssets/ProgramRequirements/081_procedural_derm_1-YR_07012014.pdf. Effective July 2014. Accessed February 26, 2015.
8. Statistics: micrographic surgery & dermatologic oncology fellowship. SF Match Web site. https://www.sfmatch.org/SpecialtyInsideAll.aspx?id=10&typ=1&name=Micrographic%20Surgery%20and%20Dermatologic%20Oncology#. Accessed March 4, 2015.
9. ASDS cosmetic dermatologic surgery fellowship accreditation program. American Society for Dermatologic Surgery Web site. http://www.asds.net/cosmetic-accreditation/. Accessed February 26, 2015.
1. Accreditation Council for Graduate Medical Education. ACGME program requirements for graduate medical education in dermatopathology. http://www.acgme.org/acgmeweb/Portals/0/PFAssets/ProgramRequirements/100_dermatopathology_2016_1-YR.pdf. Revised April 2014. Effective July 1, 2015. Accessed on February 26, 2015.
2. Subspecialty certification in dermatopathology. The American Board of Dermatology, Inc Web site. http://www.abderm.org/subspecialties/derm.html. Accessed February 26, 2015.
3. American Board of Dermatology (ABD) approved pediatric dermatology fellowship programs. The Society for Pediatric Dermatology Web site. http://pedsderm.net/training/fellowships/abd-approved-pediatric-dermatology-fellowship-programs/. Updated June 9, 2014. Accessed February 26, 2015.
4. Subspecialty certification in pediatric dermatology. The American Board of Dermatology, Inc Web site. http://www.abderm.org/subspecialties/pediatric.html. Accessed February 26, 2015.
5. Non-ABD pediatric dermatology fellowship programs. The Society for Pediatric Dermatology Web site. https://pedsderm.net/training/fellowships/non-abd-pediatric-dermatology-fellowship-programs/. Accessed February 26, 2015.
6. Pediatric dermatology match report. SF Match Web site. https://www.sfmatch.org/SpecialtyInsideAll.aspx?id=16&typ=1&name=Pediatric%20Dermatology#. Accessed March 4, 2015.
7. Accreditation Council for Graduate Medical Education. ACGME program requirements for graduate medical education in procedural dermatology. https://www.acgme.org/acgmeweb/Portals/0/PFAssets/ProgramRequirements/081_procedural_derm_1-YR_07012014.pdf. Effective July 2014. Accessed February 26, 2015.
8. Statistics: micrographic surgery & dermatologic oncology fellowship. SF Match Web site. https://www.sfmatch.org/SpecialtyInsideAll.aspx?id=10&typ=1&name=Micrographic%20Surgery%20and%20Dermatologic%20Oncology#. Accessed March 4, 2015.
9. ASDS cosmetic dermatologic surgery fellowship accreditation program. American Society for Dermatologic Surgery Web site. http://www.asds.net/cosmetic-accreditation/. Accessed February 26, 2015.
Nanotechnology: Why Should We Care?
The orthopedic community is increasingly deluged with advancements in the basic sciences. With each step, we must evaluate the necessity of new information and the relevance of these topics for clinical practice. Since the late 1990s, the promise of nanotechnology to effect significant changes in the medical field has been heralded. However, in this coming decade, we as a profession will see unprecedented advances in the movement of this technology “from the bench to the bedside.” Not unlike many other basic science advancements in our field, nanotechnology is poorly understood among clinicians and residents. As the use of biologics and drug delivery systems expands in orthopedics, nanoparticle-based devices will become more prevalent and have a momentous impact on the way we treat and diagnose orthopedic patients.
A nanoparticle is generally defined as a particle in which at least 1 dimension is between 1 to 100 nanometers and has material properties consistent with quantum mechanics.1 Nanomaterials can be composed of organic and inorganic chemical elements that enable basic chemical processes to create more complex systems. Individual nanoparticle units can be synthesized to form nanostructures, including nanotubes, nanoscaffolds, nanofibers, and even nanodiamonds.2-4 Nanoparticles at this scale display unique optical, chemical, and physical properties that can be manipulated to create specific end-use applications. Such uses may include glass fabrication, optical probes, television screens, drug delivery, gene delivery, and multiplex diagnostic assays.5-7 By crossing disciplines of physics, engineering, and medical sciences, we can create novel technology that includes nanomanufacturing, targeted drug delivery, nanorobotics in conjunction with artificial intelligence, and point-of-care diagnostics.7-9
The field of orthopedics has benefited from nanotechnologic advances, such as new therapeutics and implant-related technology. Nanotubes are hollow nanosized cylinders that are commonly created from titania, silica, or carbon-based substrates. They have garnered significant interest for their high tensile and shear strength, favorable microstructure for bony ingrowth, and their capacity to hold antibiotics or growth factors, such as bone morphogenic proteins (BMPs).10 The current local delivery limitations of BMPs via a collagen sponge have the potential to be maximized and better controlled with a nanotechnology-based approach. The size, internal structure, and shape of the nanoparticle can be manipulated to control the release of these growth factors, and certain nanoparticles can be dual-layered, allowing for release of multiple growth factors at once or in succession.11,12 A more powerful and targeted delivery system of these types of growth factors may result in improved or more robust outcomes, and further research is warranted.
It is possible that carbon-based nanotubes can be categorized as a biomedical implant secondary to their mechanical properties.13 Their strength and ability to be augmented with osteogenic materials has made them an attractive area of research as alternative implant surfaces and stand-alone implants. Nanotubes are capable of acting as a scaffold for antibiotic-loaded, carbon-based nanodiamonds for localized treatment of periprosthetic infection, and research has been directed toward controlled release of the nanodiamond-antibiotic construct from these scaffolds or hydrogels.4,14 Technologies like this may allow the clinician to treat periprosthetic infections locally and minimize the use of systemic antibiotics. The perfection of this type of delivery system may augment the role of antibiotic-laden cement and improve our treatment success rates, even in traditionally hard-to-treat organisms.
Nanoscaffolds and nanofibers are created from nanosized polymers and rendered into a 3-dimensional structure that can be loaded with biologic particles or acting as a scaffold/template for tissue or bone ingrowth. Nanofibers created using biodegradable substrates such as poly(lactic-co-glycolic acid) (PLGA) and chitosan have been extensively studied for their delayed-release properties and biocompatibility.15 These scaffolds are often soaked or loaded with chondrogenic, osteogenic, or antibacterial agents, and have been evaluated in both in vitro and in vivo studies with promising results.15,16 They have been an exciting area of research in tissue engineering, and have been accepted as an adjunct in tendon-repair treatments and local bone regeneration.3,17 As this technology is perfected, the potential to treat more effectively massive rotator cuff tears or tears with poor tissue integrity will dramatically improve and expand the indications for rotator cuff repair.
Augmentation of implant surfaces with nanomaterials that improve osseointegration, or that act as antimicrobial agents have also been a focus of research in hopes of decreasing the rates of aseptic failure and periprosthetic infection in arthroplasty procedures. Nanocrystalline surfaces made of hydroxyapatite and cobalt chromium have been evaluated for their enhanced osteoconductive properties, and may replace standard surfaces.18-20 Recent work evaluating nanoparticle-antibiotic constructs that have been covalently bound to implant surfaces for delayed release of antibiotics during the perioperative period has shown promise, and may allow a more targeted and localized treatment strategy for periprosthetic infection.21,22
Major limitations regarding successful clinical implementation of nanotechnology include both cost and regulatory processes. Currently, pharmaceutical companies estimate that, on average, successful clinical trials from phase 1 to completion for new drugs can cost hundreds of millions of dollars.23 Such high costs result partially from the laborious and capital-intensive process of conducting clinical trials that meet US Food and Drug Administration (FDA) requirements. These regulations would apply to both surface-coated implants and nanoparticle-based drug delivery systems. These types of implants would not be expedited into the market secondary to their drug delivery component and would likely require lengthy clinical studies. Implant companies may be reluctant to invest millions of dollars in multiple FDA trials when they have lucrative implants on the market.
Other limitations include the particles’ complex 3-dimensional structure, which can present challenges for mass production. Producing large quantities of nanoparticles at a consistent quality may be a major limitation to the more unique and target-based nanotherapies. Recent concerns with the toxicity profile of nanotechnology-based medicines have resulted in more intense scrutiny of the nanotechnology safety profile.24,25 Currently, nanoparticle technology is evaluated case by case with each technology requiring its own toxicology and safety profile testing if it is intended for human use. These tests can be cost-prohibitive and require extensive private and government capital for successful market entry. Despite these limitations, nanotechnology will impact the next generation of orthopedic surgeons. Current estimates project the nanomedicine market to be worth $177.6 billion by 2019.26
Advances in nanobased orthopedic technologies have expanded dramatically in the past decade, and we, as the treating physicians, must make educated decisions on how and when to use nanoparticle-based therapies and treatment options. Nanotechnology’s basic science is confusing and often burdensome, but contemporary review articles may be helpful in keeping the orthopedic resident and clinician current with advancements.10,27,28 The more we educate ourselves about evolving nanotechnologies, the less reluctance we will have when evaluating new diagnostic and therapeutic treatment modalities.
1. Hewakuruppu YL, Dombrovsky LA, Chen C, et al. Plasmonic “pump-probe” method to study semi-transparent nanofluids. Appl Opt. 2013;52(24):6041-6050.
2. Balasundaram G, Webster TJ. An overview of nano-polymers for orthopedic applications. Macromol Biosci. 2007;7(5):635-642.
3. Zhang Z, Hu J, Ma PX. Nanofiber-based delivery of bioactive agents and stem cells to bone sites. Adv Drug Deliv Rev. 2012;64(12):1129-1141.
4. Mochalin VN, Shenderova O, Ho D, Gogotsi Y. The properties and applications of nanodiamonds. Nat Nanotechnol. 2012;7(1):11-23.
5. Kneipp J, Kneipp H, Rice WL, Kneipp K. Optical probes for biological applications based on surface-enhanced Raman scattering from indocyanine green on gold nanoparticles. Anal Chem. 2005;77(8):2381-2385.
6. Wang L, O’Donoghue MB, Tan W. Nanoparticles for multiplex diagnostics and imaging. Nanomedicine (Lond). 2006;1(4):413-426.
7. Krebs MD, Salter E, Chen E, Sutter KA, Alsberg E. Calcium phosphate-DNA nanoparticle gene delivery from alginate hydrogels induces in vivo osteogenesis. J Biomed Mater Res A. 2010;92(3):1131-1138.
8. Myers FB, Lee LP. Innovations in optical microfluidic technologies for point-of-care diagnostics. Lab Chip. 2008;8(12):2015-2031.
9. Sacha GM, Varona P. Artificial intelligence in nanotechnology. Nanotechnology. 2013;24(45):452002.
10. Ganguly DY, Shahbazian R, Shokuhfar T. Recent advances in nanotubes for orthopedic implants. J Nanotech Smart Mater. 2014;1:1-10.
11. Srivastava S, Kotov NA. Composite Layer-by-Layer (LBL) assembly with inorganic nanoparticles and nanowires. Acc Chem Res. 2008;41(12):1831-1841.
12. Panda HS, Srivastava R, Bahadur D. Shape and size control of nano dispersed Mg/Al layered double hydroxide. J Nanosci Nanotechnol. 2008;8(8):4218-4223.
13. Wang X, Li Q, Xie J, et al. Fabrication of ultralong and electrically uniform single-walled carbon nanotubes on clean substrates. Nano Lett. 2009;9(9):3137-3141.
14. Zhu Y, Li J, Li W, et al. The biocompatibility of nanodiamonds and their application in drug delivery systems. Theranostics. 2012;2(3):302-312.
15. Wu L, Ding J. In vitro degradation of three-dimensional porous poly(D,L-lactide-co-glycolide) scaffolds for tissue engineering. Biomaterials. 2004;25(2):5821-5830.
16. Wu X, Rabkin-Aikawa E, Guleserian KJ, et al. Tissue-engineered microvessels on three-dimensional biodegradable scaffolds using human endothelial progenitor cells. Am J Physiol Heart Circ Physiol. 2004;287(2):H480-H487.
17. Xia W, Liu W, Cui L, et al. Tissue engineering of cartilage with the use of chitosan-gelatin complex scaffolds. J Biomed Mater Res B Appl Biomater. 2004;71(2):373-380.
18. Laurencin CT, Kumbar SG, Nukavarapu SP. Nanotechnology and orthopedics: a personal perspective. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2009;1(1):6-10.
19. Webster TJ, Ejiofor JU. Increased osteoblast adhesion on nanophase metals: Ti, Ti6Al4V, and CoCrMo. Biomaterials. 2004;25(19):4731-4739.
20. Webster TJ, Ergun C, Doremus RH, Siegel RW, Bizios R. Enhanced functions of osteoblasts on nanophase ceramics. Biomaterials. 2000;21(17):1803-1810.
21. Stewart S, Barr S, Engiles J, et al. Vancomycin-modified implant surface inhibits biofilm formation and supports bone-healing in an infected osteotomy model in sheep: a proof-of-concept study. J Bone Joint Surg Am. 2012;94(15):1406-1415.
22. Hickok NJ, Shapiro IM. Immobilized antibiotics to prevent orthopaedic implant infections. Adv Drug Deliv Rev. 2012;64(12):1165-1176.
23. DiMasi JA, Hansen RW, Grabowski HG. The price of innovation: new estimates of drug development costs. J Health Econ. 2003;22(2):151-185.
24. Vines T, Faunce T. Assessing the safety and cost-effectiveness of early nanodrugs. J Law Med. 2009;16(5):822-845.
25. Nel A, Xia T, Mädler L, Li N. Toxic potential of materials at the nanolevel. Science. 2006;311(5761):622-627.
26. Nanomedicine Market (Neurology, Cardiovascular, Anti-Inflammatory, Anti-Infective, and Oncology Applications): Global Industry Analysis, Size, Share, Growth, Trends and Forecast, 2013-2019. Transparency Market Research website. http://www.transparencymarketresearch.com/nanomedicine-market.html. Published August 1, 2014. Accessed January 20, 2015.
27. Sullivan MP, McHale KJ, Parvizi J, Mehta S. Nanotechnology: current concepts in orthopaedic surgery and future directions. Bone Joint J. 2014;96-B(5):569-573.
28. Pleshko N, Grande DA, Myers KR. Nanotechnology in orthopaedics. J Am Acad Orthop Surg. 2012;20(1):60-62.
The orthopedic community is increasingly deluged with advancements in the basic sciences. With each step, we must evaluate the necessity of new information and the relevance of these topics for clinical practice. Since the late 1990s, the promise of nanotechnology to effect significant changes in the medical field has been heralded. However, in this coming decade, we as a profession will see unprecedented advances in the movement of this technology “from the bench to the bedside.” Not unlike many other basic science advancements in our field, nanotechnology is poorly understood among clinicians and residents. As the use of biologics and drug delivery systems expands in orthopedics, nanoparticle-based devices will become more prevalent and have a momentous impact on the way we treat and diagnose orthopedic patients.
A nanoparticle is generally defined as a particle in which at least 1 dimension is between 1 to 100 nanometers and has material properties consistent with quantum mechanics.1 Nanomaterials can be composed of organic and inorganic chemical elements that enable basic chemical processes to create more complex systems. Individual nanoparticle units can be synthesized to form nanostructures, including nanotubes, nanoscaffolds, nanofibers, and even nanodiamonds.2-4 Nanoparticles at this scale display unique optical, chemical, and physical properties that can be manipulated to create specific end-use applications. Such uses may include glass fabrication, optical probes, television screens, drug delivery, gene delivery, and multiplex diagnostic assays.5-7 By crossing disciplines of physics, engineering, and medical sciences, we can create novel technology that includes nanomanufacturing, targeted drug delivery, nanorobotics in conjunction with artificial intelligence, and point-of-care diagnostics.7-9
The field of orthopedics has benefited from nanotechnologic advances, such as new therapeutics and implant-related technology. Nanotubes are hollow nanosized cylinders that are commonly created from titania, silica, or carbon-based substrates. They have garnered significant interest for their high tensile and shear strength, favorable microstructure for bony ingrowth, and their capacity to hold antibiotics or growth factors, such as bone morphogenic proteins (BMPs).10 The current local delivery limitations of BMPs via a collagen sponge have the potential to be maximized and better controlled with a nanotechnology-based approach. The size, internal structure, and shape of the nanoparticle can be manipulated to control the release of these growth factors, and certain nanoparticles can be dual-layered, allowing for release of multiple growth factors at once or in succession.11,12 A more powerful and targeted delivery system of these types of growth factors may result in improved or more robust outcomes, and further research is warranted.
It is possible that carbon-based nanotubes can be categorized as a biomedical implant secondary to their mechanical properties.13 Their strength and ability to be augmented with osteogenic materials has made them an attractive area of research as alternative implant surfaces and stand-alone implants. Nanotubes are capable of acting as a scaffold for antibiotic-loaded, carbon-based nanodiamonds for localized treatment of periprosthetic infection, and research has been directed toward controlled release of the nanodiamond-antibiotic construct from these scaffolds or hydrogels.4,14 Technologies like this may allow the clinician to treat periprosthetic infections locally and minimize the use of systemic antibiotics. The perfection of this type of delivery system may augment the role of antibiotic-laden cement and improve our treatment success rates, even in traditionally hard-to-treat organisms.
Nanoscaffolds and nanofibers are created from nanosized polymers and rendered into a 3-dimensional structure that can be loaded with biologic particles or acting as a scaffold/template for tissue or bone ingrowth. Nanofibers created using biodegradable substrates such as poly(lactic-co-glycolic acid) (PLGA) and chitosan have been extensively studied for their delayed-release properties and biocompatibility.15 These scaffolds are often soaked or loaded with chondrogenic, osteogenic, or antibacterial agents, and have been evaluated in both in vitro and in vivo studies with promising results.15,16 They have been an exciting area of research in tissue engineering, and have been accepted as an adjunct in tendon-repair treatments and local bone regeneration.3,17 As this technology is perfected, the potential to treat more effectively massive rotator cuff tears or tears with poor tissue integrity will dramatically improve and expand the indications for rotator cuff repair.
Augmentation of implant surfaces with nanomaterials that improve osseointegration, or that act as antimicrobial agents have also been a focus of research in hopes of decreasing the rates of aseptic failure and periprosthetic infection in arthroplasty procedures. Nanocrystalline surfaces made of hydroxyapatite and cobalt chromium have been evaluated for their enhanced osteoconductive properties, and may replace standard surfaces.18-20 Recent work evaluating nanoparticle-antibiotic constructs that have been covalently bound to implant surfaces for delayed release of antibiotics during the perioperative period has shown promise, and may allow a more targeted and localized treatment strategy for periprosthetic infection.21,22
Major limitations regarding successful clinical implementation of nanotechnology include both cost and regulatory processes. Currently, pharmaceutical companies estimate that, on average, successful clinical trials from phase 1 to completion for new drugs can cost hundreds of millions of dollars.23 Such high costs result partially from the laborious and capital-intensive process of conducting clinical trials that meet US Food and Drug Administration (FDA) requirements. These regulations would apply to both surface-coated implants and nanoparticle-based drug delivery systems. These types of implants would not be expedited into the market secondary to their drug delivery component and would likely require lengthy clinical studies. Implant companies may be reluctant to invest millions of dollars in multiple FDA trials when they have lucrative implants on the market.
Other limitations include the particles’ complex 3-dimensional structure, which can present challenges for mass production. Producing large quantities of nanoparticles at a consistent quality may be a major limitation to the more unique and target-based nanotherapies. Recent concerns with the toxicity profile of nanotechnology-based medicines have resulted in more intense scrutiny of the nanotechnology safety profile.24,25 Currently, nanoparticle technology is evaluated case by case with each technology requiring its own toxicology and safety profile testing if it is intended for human use. These tests can be cost-prohibitive and require extensive private and government capital for successful market entry. Despite these limitations, nanotechnology will impact the next generation of orthopedic surgeons. Current estimates project the nanomedicine market to be worth $177.6 billion by 2019.26
Advances in nanobased orthopedic technologies have expanded dramatically in the past decade, and we, as the treating physicians, must make educated decisions on how and when to use nanoparticle-based therapies and treatment options. Nanotechnology’s basic science is confusing and often burdensome, but contemporary review articles may be helpful in keeping the orthopedic resident and clinician current with advancements.10,27,28 The more we educate ourselves about evolving nanotechnologies, the less reluctance we will have when evaluating new diagnostic and therapeutic treatment modalities.
The orthopedic community is increasingly deluged with advancements in the basic sciences. With each step, we must evaluate the necessity of new information and the relevance of these topics for clinical practice. Since the late 1990s, the promise of nanotechnology to effect significant changes in the medical field has been heralded. However, in this coming decade, we as a profession will see unprecedented advances in the movement of this technology “from the bench to the bedside.” Not unlike many other basic science advancements in our field, nanotechnology is poorly understood among clinicians and residents. As the use of biologics and drug delivery systems expands in orthopedics, nanoparticle-based devices will become more prevalent and have a momentous impact on the way we treat and diagnose orthopedic patients.
A nanoparticle is generally defined as a particle in which at least 1 dimension is between 1 to 100 nanometers and has material properties consistent with quantum mechanics.1 Nanomaterials can be composed of organic and inorganic chemical elements that enable basic chemical processes to create more complex systems. Individual nanoparticle units can be synthesized to form nanostructures, including nanotubes, nanoscaffolds, nanofibers, and even nanodiamonds.2-4 Nanoparticles at this scale display unique optical, chemical, and physical properties that can be manipulated to create specific end-use applications. Such uses may include glass fabrication, optical probes, television screens, drug delivery, gene delivery, and multiplex diagnostic assays.5-7 By crossing disciplines of physics, engineering, and medical sciences, we can create novel technology that includes nanomanufacturing, targeted drug delivery, nanorobotics in conjunction with artificial intelligence, and point-of-care diagnostics.7-9
The field of orthopedics has benefited from nanotechnologic advances, such as new therapeutics and implant-related technology. Nanotubes are hollow nanosized cylinders that are commonly created from titania, silica, or carbon-based substrates. They have garnered significant interest for their high tensile and shear strength, favorable microstructure for bony ingrowth, and their capacity to hold antibiotics or growth factors, such as bone morphogenic proteins (BMPs).10 The current local delivery limitations of BMPs via a collagen sponge have the potential to be maximized and better controlled with a nanotechnology-based approach. The size, internal structure, and shape of the nanoparticle can be manipulated to control the release of these growth factors, and certain nanoparticles can be dual-layered, allowing for release of multiple growth factors at once or in succession.11,12 A more powerful and targeted delivery system of these types of growth factors may result in improved or more robust outcomes, and further research is warranted.
It is possible that carbon-based nanotubes can be categorized as a biomedical implant secondary to their mechanical properties.13 Their strength and ability to be augmented with osteogenic materials has made them an attractive area of research as alternative implant surfaces and stand-alone implants. Nanotubes are capable of acting as a scaffold for antibiotic-loaded, carbon-based nanodiamonds for localized treatment of periprosthetic infection, and research has been directed toward controlled release of the nanodiamond-antibiotic construct from these scaffolds or hydrogels.4,14 Technologies like this may allow the clinician to treat periprosthetic infections locally and minimize the use of systemic antibiotics. The perfection of this type of delivery system may augment the role of antibiotic-laden cement and improve our treatment success rates, even in traditionally hard-to-treat organisms.
Nanoscaffolds and nanofibers are created from nanosized polymers and rendered into a 3-dimensional structure that can be loaded with biologic particles or acting as a scaffold/template for tissue or bone ingrowth. Nanofibers created using biodegradable substrates such as poly(lactic-co-glycolic acid) (PLGA) and chitosan have been extensively studied for their delayed-release properties and biocompatibility.15 These scaffolds are often soaked or loaded with chondrogenic, osteogenic, or antibacterial agents, and have been evaluated in both in vitro and in vivo studies with promising results.15,16 They have been an exciting area of research in tissue engineering, and have been accepted as an adjunct in tendon-repair treatments and local bone regeneration.3,17 As this technology is perfected, the potential to treat more effectively massive rotator cuff tears or tears with poor tissue integrity will dramatically improve and expand the indications for rotator cuff repair.
Augmentation of implant surfaces with nanomaterials that improve osseointegration, or that act as antimicrobial agents have also been a focus of research in hopes of decreasing the rates of aseptic failure and periprosthetic infection in arthroplasty procedures. Nanocrystalline surfaces made of hydroxyapatite and cobalt chromium have been evaluated for their enhanced osteoconductive properties, and may replace standard surfaces.18-20 Recent work evaluating nanoparticle-antibiotic constructs that have been covalently bound to implant surfaces for delayed release of antibiotics during the perioperative period has shown promise, and may allow a more targeted and localized treatment strategy for periprosthetic infection.21,22
Major limitations regarding successful clinical implementation of nanotechnology include both cost and regulatory processes. Currently, pharmaceutical companies estimate that, on average, successful clinical trials from phase 1 to completion for new drugs can cost hundreds of millions of dollars.23 Such high costs result partially from the laborious and capital-intensive process of conducting clinical trials that meet US Food and Drug Administration (FDA) requirements. These regulations would apply to both surface-coated implants and nanoparticle-based drug delivery systems. These types of implants would not be expedited into the market secondary to their drug delivery component and would likely require lengthy clinical studies. Implant companies may be reluctant to invest millions of dollars in multiple FDA trials when they have lucrative implants on the market.
Other limitations include the particles’ complex 3-dimensional structure, which can present challenges for mass production. Producing large quantities of nanoparticles at a consistent quality may be a major limitation to the more unique and target-based nanotherapies. Recent concerns with the toxicity profile of nanotechnology-based medicines have resulted in more intense scrutiny of the nanotechnology safety profile.24,25 Currently, nanoparticle technology is evaluated case by case with each technology requiring its own toxicology and safety profile testing if it is intended for human use. These tests can be cost-prohibitive and require extensive private and government capital for successful market entry. Despite these limitations, nanotechnology will impact the next generation of orthopedic surgeons. Current estimates project the nanomedicine market to be worth $177.6 billion by 2019.26
Advances in nanobased orthopedic technologies have expanded dramatically in the past decade, and we, as the treating physicians, must make educated decisions on how and when to use nanoparticle-based therapies and treatment options. Nanotechnology’s basic science is confusing and often burdensome, but contemporary review articles may be helpful in keeping the orthopedic resident and clinician current with advancements.10,27,28 The more we educate ourselves about evolving nanotechnologies, the less reluctance we will have when evaluating new diagnostic and therapeutic treatment modalities.
1. Hewakuruppu YL, Dombrovsky LA, Chen C, et al. Plasmonic “pump-probe” method to study semi-transparent nanofluids. Appl Opt. 2013;52(24):6041-6050.
2. Balasundaram G, Webster TJ. An overview of nano-polymers for orthopedic applications. Macromol Biosci. 2007;7(5):635-642.
3. Zhang Z, Hu J, Ma PX. Nanofiber-based delivery of bioactive agents and stem cells to bone sites. Adv Drug Deliv Rev. 2012;64(12):1129-1141.
4. Mochalin VN, Shenderova O, Ho D, Gogotsi Y. The properties and applications of nanodiamonds. Nat Nanotechnol. 2012;7(1):11-23.
5. Kneipp J, Kneipp H, Rice WL, Kneipp K. Optical probes for biological applications based on surface-enhanced Raman scattering from indocyanine green on gold nanoparticles. Anal Chem. 2005;77(8):2381-2385.
6. Wang L, O’Donoghue MB, Tan W. Nanoparticles for multiplex diagnostics and imaging. Nanomedicine (Lond). 2006;1(4):413-426.
7. Krebs MD, Salter E, Chen E, Sutter KA, Alsberg E. Calcium phosphate-DNA nanoparticle gene delivery from alginate hydrogels induces in vivo osteogenesis. J Biomed Mater Res A. 2010;92(3):1131-1138.
8. Myers FB, Lee LP. Innovations in optical microfluidic technologies for point-of-care diagnostics. Lab Chip. 2008;8(12):2015-2031.
9. Sacha GM, Varona P. Artificial intelligence in nanotechnology. Nanotechnology. 2013;24(45):452002.
10. Ganguly DY, Shahbazian R, Shokuhfar T. Recent advances in nanotubes for orthopedic implants. J Nanotech Smart Mater. 2014;1:1-10.
11. Srivastava S, Kotov NA. Composite Layer-by-Layer (LBL) assembly with inorganic nanoparticles and nanowires. Acc Chem Res. 2008;41(12):1831-1841.
12. Panda HS, Srivastava R, Bahadur D. Shape and size control of nano dispersed Mg/Al layered double hydroxide. J Nanosci Nanotechnol. 2008;8(8):4218-4223.
13. Wang X, Li Q, Xie J, et al. Fabrication of ultralong and electrically uniform single-walled carbon nanotubes on clean substrates. Nano Lett. 2009;9(9):3137-3141.
14. Zhu Y, Li J, Li W, et al. The biocompatibility of nanodiamonds and their application in drug delivery systems. Theranostics. 2012;2(3):302-312.
15. Wu L, Ding J. In vitro degradation of three-dimensional porous poly(D,L-lactide-co-glycolide) scaffolds for tissue engineering. Biomaterials. 2004;25(2):5821-5830.
16. Wu X, Rabkin-Aikawa E, Guleserian KJ, et al. Tissue-engineered microvessels on three-dimensional biodegradable scaffolds using human endothelial progenitor cells. Am J Physiol Heart Circ Physiol. 2004;287(2):H480-H487.
17. Xia W, Liu W, Cui L, et al. Tissue engineering of cartilage with the use of chitosan-gelatin complex scaffolds. J Biomed Mater Res B Appl Biomater. 2004;71(2):373-380.
18. Laurencin CT, Kumbar SG, Nukavarapu SP. Nanotechnology and orthopedics: a personal perspective. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2009;1(1):6-10.
19. Webster TJ, Ejiofor JU. Increased osteoblast adhesion on nanophase metals: Ti, Ti6Al4V, and CoCrMo. Biomaterials. 2004;25(19):4731-4739.
20. Webster TJ, Ergun C, Doremus RH, Siegel RW, Bizios R. Enhanced functions of osteoblasts on nanophase ceramics. Biomaterials. 2000;21(17):1803-1810.
21. Stewart S, Barr S, Engiles J, et al. Vancomycin-modified implant surface inhibits biofilm formation and supports bone-healing in an infected osteotomy model in sheep: a proof-of-concept study. J Bone Joint Surg Am. 2012;94(15):1406-1415.
22. Hickok NJ, Shapiro IM. Immobilized antibiotics to prevent orthopaedic implant infections. Adv Drug Deliv Rev. 2012;64(12):1165-1176.
23. DiMasi JA, Hansen RW, Grabowski HG. The price of innovation: new estimates of drug development costs. J Health Econ. 2003;22(2):151-185.
24. Vines T, Faunce T. Assessing the safety and cost-effectiveness of early nanodrugs. J Law Med. 2009;16(5):822-845.
25. Nel A, Xia T, Mädler L, Li N. Toxic potential of materials at the nanolevel. Science. 2006;311(5761):622-627.
26. Nanomedicine Market (Neurology, Cardiovascular, Anti-Inflammatory, Anti-Infective, and Oncology Applications): Global Industry Analysis, Size, Share, Growth, Trends and Forecast, 2013-2019. Transparency Market Research website. http://www.transparencymarketresearch.com/nanomedicine-market.html. Published August 1, 2014. Accessed January 20, 2015.
27. Sullivan MP, McHale KJ, Parvizi J, Mehta S. Nanotechnology: current concepts in orthopaedic surgery and future directions. Bone Joint J. 2014;96-B(5):569-573.
28. Pleshko N, Grande DA, Myers KR. Nanotechnology in orthopaedics. J Am Acad Orthop Surg. 2012;20(1):60-62.
1. Hewakuruppu YL, Dombrovsky LA, Chen C, et al. Plasmonic “pump-probe” method to study semi-transparent nanofluids. Appl Opt. 2013;52(24):6041-6050.
2. Balasundaram G, Webster TJ. An overview of nano-polymers for orthopedic applications. Macromol Biosci. 2007;7(5):635-642.
3. Zhang Z, Hu J, Ma PX. Nanofiber-based delivery of bioactive agents and stem cells to bone sites. Adv Drug Deliv Rev. 2012;64(12):1129-1141.
4. Mochalin VN, Shenderova O, Ho D, Gogotsi Y. The properties and applications of nanodiamonds. Nat Nanotechnol. 2012;7(1):11-23.
5. Kneipp J, Kneipp H, Rice WL, Kneipp K. Optical probes for biological applications based on surface-enhanced Raman scattering from indocyanine green on gold nanoparticles. Anal Chem. 2005;77(8):2381-2385.
6. Wang L, O’Donoghue MB, Tan W. Nanoparticles for multiplex diagnostics and imaging. Nanomedicine (Lond). 2006;1(4):413-426.
7. Krebs MD, Salter E, Chen E, Sutter KA, Alsberg E. Calcium phosphate-DNA nanoparticle gene delivery from alginate hydrogels induces in vivo osteogenesis. J Biomed Mater Res A. 2010;92(3):1131-1138.
8. Myers FB, Lee LP. Innovations in optical microfluidic technologies for point-of-care diagnostics. Lab Chip. 2008;8(12):2015-2031.
9. Sacha GM, Varona P. Artificial intelligence in nanotechnology. Nanotechnology. 2013;24(45):452002.
10. Ganguly DY, Shahbazian R, Shokuhfar T. Recent advances in nanotubes for orthopedic implants. J Nanotech Smart Mater. 2014;1:1-10.
11. Srivastava S, Kotov NA. Composite Layer-by-Layer (LBL) assembly with inorganic nanoparticles and nanowires. Acc Chem Res. 2008;41(12):1831-1841.
12. Panda HS, Srivastava R, Bahadur D. Shape and size control of nano dispersed Mg/Al layered double hydroxide. J Nanosci Nanotechnol. 2008;8(8):4218-4223.
13. Wang X, Li Q, Xie J, et al. Fabrication of ultralong and electrically uniform single-walled carbon nanotubes on clean substrates. Nano Lett. 2009;9(9):3137-3141.
14. Zhu Y, Li J, Li W, et al. The biocompatibility of nanodiamonds and their application in drug delivery systems. Theranostics. 2012;2(3):302-312.
15. Wu L, Ding J. In vitro degradation of three-dimensional porous poly(D,L-lactide-co-glycolide) scaffolds for tissue engineering. Biomaterials. 2004;25(2):5821-5830.
16. Wu X, Rabkin-Aikawa E, Guleserian KJ, et al. Tissue-engineered microvessels on three-dimensional biodegradable scaffolds using human endothelial progenitor cells. Am J Physiol Heart Circ Physiol. 2004;287(2):H480-H487.
17. Xia W, Liu W, Cui L, et al. Tissue engineering of cartilage with the use of chitosan-gelatin complex scaffolds. J Biomed Mater Res B Appl Biomater. 2004;71(2):373-380.
18. Laurencin CT, Kumbar SG, Nukavarapu SP. Nanotechnology and orthopedics: a personal perspective. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2009;1(1):6-10.
19. Webster TJ, Ejiofor JU. Increased osteoblast adhesion on nanophase metals: Ti, Ti6Al4V, and CoCrMo. Biomaterials. 2004;25(19):4731-4739.
20. Webster TJ, Ergun C, Doremus RH, Siegel RW, Bizios R. Enhanced functions of osteoblasts on nanophase ceramics. Biomaterials. 2000;21(17):1803-1810.
21. Stewart S, Barr S, Engiles J, et al. Vancomycin-modified implant surface inhibits biofilm formation and supports bone-healing in an infected osteotomy model in sheep: a proof-of-concept study. J Bone Joint Surg Am. 2012;94(15):1406-1415.
22. Hickok NJ, Shapiro IM. Immobilized antibiotics to prevent orthopaedic implant infections. Adv Drug Deliv Rev. 2012;64(12):1165-1176.
23. DiMasi JA, Hansen RW, Grabowski HG. The price of innovation: new estimates of drug development costs. J Health Econ. 2003;22(2):151-185.
24. Vines T, Faunce T. Assessing the safety and cost-effectiveness of early nanodrugs. J Law Med. 2009;16(5):822-845.
25. Nel A, Xia T, Mädler L, Li N. Toxic potential of materials at the nanolevel. Science. 2006;311(5761):622-627.
26. Nanomedicine Market (Neurology, Cardiovascular, Anti-Inflammatory, Anti-Infective, and Oncology Applications): Global Industry Analysis, Size, Share, Growth, Trends and Forecast, 2013-2019. Transparency Market Research website. http://www.transparencymarketresearch.com/nanomedicine-market.html. Published August 1, 2014. Accessed January 20, 2015.
27. Sullivan MP, McHale KJ, Parvizi J, Mehta S. Nanotechnology: current concepts in orthopaedic surgery and future directions. Bone Joint J. 2014;96-B(5):569-573.
28. Pleshko N, Grande DA, Myers KR. Nanotechnology in orthopaedics. J Am Acad Orthop Surg. 2012;20(1):60-62.
The patient refuses to cooperate. What can you do? What should you do?
The real estate business embraces the concept of ownership using the term “bundle of rights.” Real estate agents view full, unaffected ownership of a real property as complete (ie, undivided) and, when ownership is shared, talk about percentages of that bundle.
The same principle can be applied to guardianship. Because we are our own guardians, we own a full, undivided bundle of rights, including all our constitutional rights and the right to make decisions— even bad ones. Of course, an undivided bundle also means that we are fully responsible for the decisions we make.
When a patient requires representation
There may be a situation when we would give someone else the authority to represent us for a specific reason. In this case we would authorize this person to act on our behalf as we would do ourselves—yet we still retain 100% ownership of the “bundle,” and therefore can revoke this authorization at any time. The person we hire (appoint) to represent us will become our power of attorney (POA), and because we appoint this person for a specific situation (handle certain medical affairs, manage some financial affairs, sign real estate documents, etc.), this kind or POA is called “specific” or “special.” When we give someone the right to represent us in any or all of our affairs, this POA is called “general” or “durable.”
It is important to mention that as long as we continue to have psychological capacity and are willing to continue to be our own guardians (own 100% of the bundle of rights), we can terminate any POA we have appointed previously or designate another person to represent us as a “special” or “general” POA. Because of this, if an older patient—who is legally competent but physically unable to live on his (her) own— refuses to enter a long-term care facility, he (she) cannot be sent there against his will, even if the POA insists on it. Because of this, if the patient’s primary team strongly disagrees with this patient’s decision, his (her) “decision-making capacity” should be assessed and, if necessary, a competency hearing will need to be conducted. The court will then decide if this person is able (or unable) to handle his own affairs, and if the court decides that the person cannot be responsible to provide himself with food, health care, housing, and other necessities, the guardian (relative, friend, public administrator, etc.) will be appointed to do so.
Evaluating decision-making capacity
Determining “decision-making capacity” should not be confused with the legal concept of “competence.” We, physicians, often are called to evaluate a patient and give our opinion of the current level of this patient’s functioning (including his [her] decision-making capacity), and we—ourselves and a requesting team—need to be clear that it is merely our opinion and should be used as such. We need to remember that even if a patient is judged to be legally incompetent to handle financial affairs, he (she) might retain sufficient ability to make decisions about treatments.
We also need to remember that decision-making capacity can change, depending on medical conditions (severe anxiety, delirium), successful treatments, substance intoxication, etc. Because of this, we need to communicate to the requesting team that “decision-making ability” is situation-specific and time-specific, and that failure to make a decision on one issue should not be generalized to other aspects of the patient’s life.
Any physician can evaluate patient’s decision-making ability, but traditionally the psychiatry team is called to do so. It usually happens because the primary medical team needs us to provide “a third-party validation,” or because of the common misperception that only the psychiatric team can initiate a civil involuntary detention when necessary.
In any case, regardless of who evaluates the patient, specific points need to be addressed and the following questions need to be answered:
• Does the patient understand the nature of his (her) condition?
• Does the patient understand what treatment we are proposing or what he should do?
• Does the patient understand the consequences (good or bad) if he rejects our proposed action or treatment?
When information (discharge plan, treatment plan, etc.) is presented to patients, we should ask them to repeat it in their own words. We should not expect them to understand all of the technical aspects. We should consider patients’ intelligence level and their ability to communicate; if they can clearly verbalize their understanding of information and be consistent in their wish to continue with their decision, we have to declare that they have decision-making capability and able to proceed with their chosen treatment.
More matters that need to be mentioned
Restrictions on the patient. We need to remember that, even if a patient is thought to be able to make his own decisions, there may be some situations when he can be held in the hospital against his will. These usually are the cases when the patient is psychiatrically or medically unstable (unable to care of himself), but also if the patient is at risk of harming himself or others, subject of elder abuse, or suspected of being an abuser.
Restrictions on the practitioner. Even if the patient is determined to be lacking decision-making capacity, we, physicians, cannot perform tests, procedures, or do the placements without the patient’s agreement.
Informed consent doctrine is applicable in this case, and if performing a test or procedure is necessary (except life- or limb-saving emergencies, when doctrine of physician prerogative applies), or if there a disagreement in post-discharge placement, the emergency guardianship may need to be pursued.
Disclosure
Dr. Graypel reports no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
The real estate business embraces the concept of ownership using the term “bundle of rights.” Real estate agents view full, unaffected ownership of a real property as complete (ie, undivided) and, when ownership is shared, talk about percentages of that bundle.
The same principle can be applied to guardianship. Because we are our own guardians, we own a full, undivided bundle of rights, including all our constitutional rights and the right to make decisions— even bad ones. Of course, an undivided bundle also means that we are fully responsible for the decisions we make.
When a patient requires representation
There may be a situation when we would give someone else the authority to represent us for a specific reason. In this case we would authorize this person to act on our behalf as we would do ourselves—yet we still retain 100% ownership of the “bundle,” and therefore can revoke this authorization at any time. The person we hire (appoint) to represent us will become our power of attorney (POA), and because we appoint this person for a specific situation (handle certain medical affairs, manage some financial affairs, sign real estate documents, etc.), this kind or POA is called “specific” or “special.” When we give someone the right to represent us in any or all of our affairs, this POA is called “general” or “durable.”
It is important to mention that as long as we continue to have psychological capacity and are willing to continue to be our own guardians (own 100% of the bundle of rights), we can terminate any POA we have appointed previously or designate another person to represent us as a “special” or “general” POA. Because of this, if an older patient—who is legally competent but physically unable to live on his (her) own— refuses to enter a long-term care facility, he (she) cannot be sent there against his will, even if the POA insists on it. Because of this, if the patient’s primary team strongly disagrees with this patient’s decision, his (her) “decision-making capacity” should be assessed and, if necessary, a competency hearing will need to be conducted. The court will then decide if this person is able (or unable) to handle his own affairs, and if the court decides that the person cannot be responsible to provide himself with food, health care, housing, and other necessities, the guardian (relative, friend, public administrator, etc.) will be appointed to do so.
Evaluating decision-making capacity
Determining “decision-making capacity” should not be confused with the legal concept of “competence.” We, physicians, often are called to evaluate a patient and give our opinion of the current level of this patient’s functioning (including his [her] decision-making capacity), and we—ourselves and a requesting team—need to be clear that it is merely our opinion and should be used as such. We need to remember that even if a patient is judged to be legally incompetent to handle financial affairs, he (she) might retain sufficient ability to make decisions about treatments.
We also need to remember that decision-making capacity can change, depending on medical conditions (severe anxiety, delirium), successful treatments, substance intoxication, etc. Because of this, we need to communicate to the requesting team that “decision-making ability” is situation-specific and time-specific, and that failure to make a decision on one issue should not be generalized to other aspects of the patient’s life.
Any physician can evaluate patient’s decision-making ability, but traditionally the psychiatry team is called to do so. It usually happens because the primary medical team needs us to provide “a third-party validation,” or because of the common misperception that only the psychiatric team can initiate a civil involuntary detention when necessary.
In any case, regardless of who evaluates the patient, specific points need to be addressed and the following questions need to be answered:
• Does the patient understand the nature of his (her) condition?
• Does the patient understand what treatment we are proposing or what he should do?
• Does the patient understand the consequences (good or bad) if he rejects our proposed action or treatment?
When information (discharge plan, treatment plan, etc.) is presented to patients, we should ask them to repeat it in their own words. We should not expect them to understand all of the technical aspects. We should consider patients’ intelligence level and their ability to communicate; if they can clearly verbalize their understanding of information and be consistent in their wish to continue with their decision, we have to declare that they have decision-making capability and able to proceed with their chosen treatment.
More matters that need to be mentioned
Restrictions on the patient. We need to remember that, even if a patient is thought to be able to make his own decisions, there may be some situations when he can be held in the hospital against his will. These usually are the cases when the patient is psychiatrically or medically unstable (unable to care of himself), but also if the patient is at risk of harming himself or others, subject of elder abuse, or suspected of being an abuser.
Restrictions on the practitioner. Even if the patient is determined to be lacking decision-making capacity, we, physicians, cannot perform tests, procedures, or do the placements without the patient’s agreement.
Informed consent doctrine is applicable in this case, and if performing a test or procedure is necessary (except life- or limb-saving emergencies, when doctrine of physician prerogative applies), or if there a disagreement in post-discharge placement, the emergency guardianship may need to be pursued.
Disclosure
Dr. Graypel reports no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
The real estate business embraces the concept of ownership using the term “bundle of rights.” Real estate agents view full, unaffected ownership of a real property as complete (ie, undivided) and, when ownership is shared, talk about percentages of that bundle.
The same principle can be applied to guardianship. Because we are our own guardians, we own a full, undivided bundle of rights, including all our constitutional rights and the right to make decisions— even bad ones. Of course, an undivided bundle also means that we are fully responsible for the decisions we make.
When a patient requires representation
There may be a situation when we would give someone else the authority to represent us for a specific reason. In this case we would authorize this person to act on our behalf as we would do ourselves—yet we still retain 100% ownership of the “bundle,” and therefore can revoke this authorization at any time. The person we hire (appoint) to represent us will become our power of attorney (POA), and because we appoint this person for a specific situation (handle certain medical affairs, manage some financial affairs, sign real estate documents, etc.), this kind or POA is called “specific” or “special.” When we give someone the right to represent us in any or all of our affairs, this POA is called “general” or “durable.”
It is important to mention that as long as we continue to have psychological capacity and are willing to continue to be our own guardians (own 100% of the bundle of rights), we can terminate any POA we have appointed previously or designate another person to represent us as a “special” or “general” POA. Because of this, if an older patient—who is legally competent but physically unable to live on his (her) own— refuses to enter a long-term care facility, he (she) cannot be sent there against his will, even if the POA insists on it. Because of this, if the patient’s primary team strongly disagrees with this patient’s decision, his (her) “decision-making capacity” should be assessed and, if necessary, a competency hearing will need to be conducted. The court will then decide if this person is able (or unable) to handle his own affairs, and if the court decides that the person cannot be responsible to provide himself with food, health care, housing, and other necessities, the guardian (relative, friend, public administrator, etc.) will be appointed to do so.
Evaluating decision-making capacity
Determining “decision-making capacity” should not be confused with the legal concept of “competence.” We, physicians, often are called to evaluate a patient and give our opinion of the current level of this patient’s functioning (including his [her] decision-making capacity), and we—ourselves and a requesting team—need to be clear that it is merely our opinion and should be used as such. We need to remember that even if a patient is judged to be legally incompetent to handle financial affairs, he (she) might retain sufficient ability to make decisions about treatments.
We also need to remember that decision-making capacity can change, depending on medical conditions (severe anxiety, delirium), successful treatments, substance intoxication, etc. Because of this, we need to communicate to the requesting team that “decision-making ability” is situation-specific and time-specific, and that failure to make a decision on one issue should not be generalized to other aspects of the patient’s life.
Any physician can evaluate patient’s decision-making ability, but traditionally the psychiatry team is called to do so. It usually happens because the primary medical team needs us to provide “a third-party validation,” or because of the common misperception that only the psychiatric team can initiate a civil involuntary detention when necessary.
In any case, regardless of who evaluates the patient, specific points need to be addressed and the following questions need to be answered:
• Does the patient understand the nature of his (her) condition?
• Does the patient understand what treatment we are proposing or what he should do?
• Does the patient understand the consequences (good or bad) if he rejects our proposed action or treatment?
When information (discharge plan, treatment plan, etc.) is presented to patients, we should ask them to repeat it in their own words. We should not expect them to understand all of the technical aspects. We should consider patients’ intelligence level and their ability to communicate; if they can clearly verbalize their understanding of information and be consistent in their wish to continue with their decision, we have to declare that they have decision-making capability and able to proceed with their chosen treatment.
More matters that need to be mentioned
Restrictions on the patient. We need to remember that, even if a patient is thought to be able to make his own decisions, there may be some situations when he can be held in the hospital against his will. These usually are the cases when the patient is psychiatrically or medically unstable (unable to care of himself), but also if the patient is at risk of harming himself or others, subject of elder abuse, or suspected of being an abuser.
Restrictions on the practitioner. Even if the patient is determined to be lacking decision-making capacity, we, physicians, cannot perform tests, procedures, or do the placements without the patient’s agreement.
Informed consent doctrine is applicable in this case, and if performing a test or procedure is necessary (except life- or limb-saving emergencies, when doctrine of physician prerogative applies), or if there a disagreement in post-discharge placement, the emergency guardianship may need to be pursued.
Disclosure
Dr. Graypel reports no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
Dermatologic Emergencies
Dermatologic emergency may sound like an oxymoron, but there are many emergencies that dermatology residents may encounter in their careers. In some instances the skin is the primary organ that is affected, while in others cutaneous symptoms and life-threatening signs are important diagnostic clues for what may lie beneath the skin.
As residents who are occasionally on call or on consultation services, it is important for us to recognize dermatologic emergencies quickly because some of these conditions can acutely evolve and become lethal if a diagnosis is not made early in the disease course with the appropriate treatment administered. Dermatologic emergencies can range from severe drug reactions, infections, autoimmune exacerbations, and inflammatory conditions (eg, erythroderma) to environmental insults such as burns (Figure 1) and child abuse.1
Critical Infections
Some dermatologic emergencies are infectious in origin, and although these infections are most commonly bacterial (eg, necrotizing fasciitis), they also can range from viral to fungal (eg, mucormycosis) in nature. Some areas with large populations of immunocompromised patients (eg, human immunodeficiency virus–positive patients, organ transplant recipients) may warrant a high index of suspicion for possible zebras (rare conditions) and opportunistic infections that may quickly escalate to life-threatening situations.
Although few cutaneous manifestations in emergent infections are pathognomonic, they sometimes can be categorized according to the appearance of the primary lesion: erythrodermic (eg, staphylococcal scalded skin syndrome), maculopapular (eg, Lyme disease), purpuric/petechial (eg, Rocky Mountain spotted fever), pustular (eg, disseminated candidiasis), or vesicular (eg, neonatal herpes simplex virus)(Table). On consultations, dermatology residents frequently get called to evaluate hemorrhagic and ischemic lesions in inpatients (Figure 2). Aside from infectious causes, the differential diagnosis may include coagulation abnormalities (eg, concurrent anticoagulant therapies), vasculitides, poisoning, vascular disease, or Stevens-Johnson syndrome and toxic epidermal necrolysis, which can occasionally present with hemorrhagic lesions.1,2
Necrotizing Fasciitis
Dermatology residents may frequently encounter necrotizing fasciitis, either in clinic or on the wards (Figure 3). Recognition of the skin signs in this condition is essential to patient survival. As an intern, I once had an attending teach me that patients with necrotizing fasciitis only have a couple of hours to live. The rapid unfolding of this flesh-eating disease and its high morbidity and mortality has led to recent attention in the press and media.
Although necrotizing fasciitis may be caused by several different bacterial organisms (eg, gram positive, gram negative, polymicrobial), it usually is rapidly progressive, destroying muscle and subcutaneous tissues in a matter of hours.3 Bacteria usually enter through a traumatic or present wound and quickly move along fascial planes, destroying blood vessels and whatever subcutaneous tissues happen to be in the way. Within the first few hours, the involved area that was initially erythematous becomes indurated, woody, extremely painful, and dusky, indicating a lack of circulation to the area. Extensive debridement is required until reaching noninfected tissue that is no longer purulent, necrotic, or woody to the touch. If necrotizing fasciitis is not diagnosed and treated early, patients may lose one or several limbs and death may occur.
Key findings of necrotizing fasciitis include systemic toxicity, localized painful induration, well-defined dusky blue discoloration, and a lack of bleeding or purulent discharge on incision and squeezing of the affected tissue. Crepitation or a crackling sensation can occasionally be felt when palpating the area secondary to gas formation in the tissue, though it is not always present. Patients with necrotizing fasciitis often initially present to dermatology clinics because the first manifestation happens to be in the skin. The role of dermatologists in treating this critical condition may prompt recognition and collaboration with other specialists to reach a viable outcome for the patient.3
Drug Reactions
Cutaneous drug eruptions usually are relatively benign, consisting of a morbilliform eruption often without any other accompanying symptoms. However, sometimes these reactions can present as exfoliative dermatitis or red man syndrome in which patients can develop total body erythema with diffuse scaling and pruritus.4 Aside from drug reactions, other causes of exfoliative dermatitis such as psoriasis, atopic and seborrheic dermatitis, mycosis fungoides, and lymphoma should be ruled out. Other drug eruptions that can be classified as dermatologic emergencies include leukocytoclastic vasculitis, severe urticaria or angioedema, erythema multiforme, or Stevens-Johnson syndrome and toxic epidermal necrolysis.
Severe Acne
If not treated promptly, serious cases of acne can lead to severe scarring and psychologic problems. Acne fulminans is characterized by a rapid eruption of suppurative and large, highly inflamed nodules, plaques, and cysts that result in ragged ulcerations and cicatrization of the chest, back, and occasionally the face. Systemic symptoms of fever, arthralgia, leukocytosis, and myalgia suggest an upregulation of the immune system in affected patients.
Final Comment
In summary, dermatologic emergencies do exist and some may present with characteristic skin findings. In almost all cases, collaboration with other departments such as trauma, burn, internal medicine, rheumatology, and infectious diseases is extremely helpful in diagnosing and treating these medical emergencies. Collaboration can provide insight into how brainstorming through different approaches can lead to a better outcome whether it be solving the cause of a puzzling rash in a patient with multiple comorbidities or surgically removing a bullet from a trauma patient (Figure 4). Recognition of specific cutaneous manifestations and early diagnosis of dermatologic emergencies can be lifesaving.
1. McQueen A, Martin SA, Lio PA. Derm emergencies: detecting early signs of trouble. J Fam Pract. 2012;61:71-78.
2. Bennion S. Dermatologic emergencies. In: Fitzpatrick J, Morelli J, eds. Dermatology Secrets Plus. 4th ed. Philadelphia, PA: Mosby; 2011:442-452.
3. Sarani B, Strong M, Pascual J, et al. Necrotizing fasciitis: current concepts and review of the literature. J Am Coll Surg. 2009;208:279-288.
4. Wolf R, Orion E, Marcos B, et al. Life-threatening acute adverse cutaneous drug reactions. Clin Dermatol. 2005;23:171-181.
Dermatologic emergency may sound like an oxymoron, but there are many emergencies that dermatology residents may encounter in their careers. In some instances the skin is the primary organ that is affected, while in others cutaneous symptoms and life-threatening signs are important diagnostic clues for what may lie beneath the skin.
As residents who are occasionally on call or on consultation services, it is important for us to recognize dermatologic emergencies quickly because some of these conditions can acutely evolve and become lethal if a diagnosis is not made early in the disease course with the appropriate treatment administered. Dermatologic emergencies can range from severe drug reactions, infections, autoimmune exacerbations, and inflammatory conditions (eg, erythroderma) to environmental insults such as burns (Figure 1) and child abuse.1
Critical Infections
Some dermatologic emergencies are infectious in origin, and although these infections are most commonly bacterial (eg, necrotizing fasciitis), they also can range from viral to fungal (eg, mucormycosis) in nature. Some areas with large populations of immunocompromised patients (eg, human immunodeficiency virus–positive patients, organ transplant recipients) may warrant a high index of suspicion for possible zebras (rare conditions) and opportunistic infections that may quickly escalate to life-threatening situations.
Although few cutaneous manifestations in emergent infections are pathognomonic, they sometimes can be categorized according to the appearance of the primary lesion: erythrodermic (eg, staphylococcal scalded skin syndrome), maculopapular (eg, Lyme disease), purpuric/petechial (eg, Rocky Mountain spotted fever), pustular (eg, disseminated candidiasis), or vesicular (eg, neonatal herpes simplex virus)(Table). On consultations, dermatology residents frequently get called to evaluate hemorrhagic and ischemic lesions in inpatients (Figure 2). Aside from infectious causes, the differential diagnosis may include coagulation abnormalities (eg, concurrent anticoagulant therapies), vasculitides, poisoning, vascular disease, or Stevens-Johnson syndrome and toxic epidermal necrolysis, which can occasionally present with hemorrhagic lesions.1,2
Necrotizing Fasciitis
Dermatology residents may frequently encounter necrotizing fasciitis, either in clinic or on the wards (Figure 3). Recognition of the skin signs in this condition is essential to patient survival. As an intern, I once had an attending teach me that patients with necrotizing fasciitis only have a couple of hours to live. The rapid unfolding of this flesh-eating disease and its high morbidity and mortality has led to recent attention in the press and media.
Although necrotizing fasciitis may be caused by several different bacterial organisms (eg, gram positive, gram negative, polymicrobial), it usually is rapidly progressive, destroying muscle and subcutaneous tissues in a matter of hours.3 Bacteria usually enter through a traumatic or present wound and quickly move along fascial planes, destroying blood vessels and whatever subcutaneous tissues happen to be in the way. Within the first few hours, the involved area that was initially erythematous becomes indurated, woody, extremely painful, and dusky, indicating a lack of circulation to the area. Extensive debridement is required until reaching noninfected tissue that is no longer purulent, necrotic, or woody to the touch. If necrotizing fasciitis is not diagnosed and treated early, patients may lose one or several limbs and death may occur.
Key findings of necrotizing fasciitis include systemic toxicity, localized painful induration, well-defined dusky blue discoloration, and a lack of bleeding or purulent discharge on incision and squeezing of the affected tissue. Crepitation or a crackling sensation can occasionally be felt when palpating the area secondary to gas formation in the tissue, though it is not always present. Patients with necrotizing fasciitis often initially present to dermatology clinics because the first manifestation happens to be in the skin. The role of dermatologists in treating this critical condition may prompt recognition and collaboration with other specialists to reach a viable outcome for the patient.3
Drug Reactions
Cutaneous drug eruptions usually are relatively benign, consisting of a morbilliform eruption often without any other accompanying symptoms. However, sometimes these reactions can present as exfoliative dermatitis or red man syndrome in which patients can develop total body erythema with diffuse scaling and pruritus.4 Aside from drug reactions, other causes of exfoliative dermatitis such as psoriasis, atopic and seborrheic dermatitis, mycosis fungoides, and lymphoma should be ruled out. Other drug eruptions that can be classified as dermatologic emergencies include leukocytoclastic vasculitis, severe urticaria or angioedema, erythema multiforme, or Stevens-Johnson syndrome and toxic epidermal necrolysis.
Severe Acne
If not treated promptly, serious cases of acne can lead to severe scarring and psychologic problems. Acne fulminans is characterized by a rapid eruption of suppurative and large, highly inflamed nodules, plaques, and cysts that result in ragged ulcerations and cicatrization of the chest, back, and occasionally the face. Systemic symptoms of fever, arthralgia, leukocytosis, and myalgia suggest an upregulation of the immune system in affected patients.
Final Comment
In summary, dermatologic emergencies do exist and some may present with characteristic skin findings. In almost all cases, collaboration with other departments such as trauma, burn, internal medicine, rheumatology, and infectious diseases is extremely helpful in diagnosing and treating these medical emergencies. Collaboration can provide insight into how brainstorming through different approaches can lead to a better outcome whether it be solving the cause of a puzzling rash in a patient with multiple comorbidities or surgically removing a bullet from a trauma patient (Figure 4). Recognition of specific cutaneous manifestations and early diagnosis of dermatologic emergencies can be lifesaving.
Dermatologic emergency may sound like an oxymoron, but there are many emergencies that dermatology residents may encounter in their careers. In some instances the skin is the primary organ that is affected, while in others cutaneous symptoms and life-threatening signs are important diagnostic clues for what may lie beneath the skin.
As residents who are occasionally on call or on consultation services, it is important for us to recognize dermatologic emergencies quickly because some of these conditions can acutely evolve and become lethal if a diagnosis is not made early in the disease course with the appropriate treatment administered. Dermatologic emergencies can range from severe drug reactions, infections, autoimmune exacerbations, and inflammatory conditions (eg, erythroderma) to environmental insults such as burns (Figure 1) and child abuse.1
Critical Infections
Some dermatologic emergencies are infectious in origin, and although these infections are most commonly bacterial (eg, necrotizing fasciitis), they also can range from viral to fungal (eg, mucormycosis) in nature. Some areas with large populations of immunocompromised patients (eg, human immunodeficiency virus–positive patients, organ transplant recipients) may warrant a high index of suspicion for possible zebras (rare conditions) and opportunistic infections that may quickly escalate to life-threatening situations.
Although few cutaneous manifestations in emergent infections are pathognomonic, they sometimes can be categorized according to the appearance of the primary lesion: erythrodermic (eg, staphylococcal scalded skin syndrome), maculopapular (eg, Lyme disease), purpuric/petechial (eg, Rocky Mountain spotted fever), pustular (eg, disseminated candidiasis), or vesicular (eg, neonatal herpes simplex virus)(Table). On consultations, dermatology residents frequently get called to evaluate hemorrhagic and ischemic lesions in inpatients (Figure 2). Aside from infectious causes, the differential diagnosis may include coagulation abnormalities (eg, concurrent anticoagulant therapies), vasculitides, poisoning, vascular disease, or Stevens-Johnson syndrome and toxic epidermal necrolysis, which can occasionally present with hemorrhagic lesions.1,2
Necrotizing Fasciitis
Dermatology residents may frequently encounter necrotizing fasciitis, either in clinic or on the wards (Figure 3). Recognition of the skin signs in this condition is essential to patient survival. As an intern, I once had an attending teach me that patients with necrotizing fasciitis only have a couple of hours to live. The rapid unfolding of this flesh-eating disease and its high morbidity and mortality has led to recent attention in the press and media.
Although necrotizing fasciitis may be caused by several different bacterial organisms (eg, gram positive, gram negative, polymicrobial), it usually is rapidly progressive, destroying muscle and subcutaneous tissues in a matter of hours.3 Bacteria usually enter through a traumatic or present wound and quickly move along fascial planes, destroying blood vessels and whatever subcutaneous tissues happen to be in the way. Within the first few hours, the involved area that was initially erythematous becomes indurated, woody, extremely painful, and dusky, indicating a lack of circulation to the area. Extensive debridement is required until reaching noninfected tissue that is no longer purulent, necrotic, or woody to the touch. If necrotizing fasciitis is not diagnosed and treated early, patients may lose one or several limbs and death may occur.
Key findings of necrotizing fasciitis include systemic toxicity, localized painful induration, well-defined dusky blue discoloration, and a lack of bleeding or purulent discharge on incision and squeezing of the affected tissue. Crepitation or a crackling sensation can occasionally be felt when palpating the area secondary to gas formation in the tissue, though it is not always present. Patients with necrotizing fasciitis often initially present to dermatology clinics because the first manifestation happens to be in the skin. The role of dermatologists in treating this critical condition may prompt recognition and collaboration with other specialists to reach a viable outcome for the patient.3
Drug Reactions
Cutaneous drug eruptions usually are relatively benign, consisting of a morbilliform eruption often without any other accompanying symptoms. However, sometimes these reactions can present as exfoliative dermatitis or red man syndrome in which patients can develop total body erythema with diffuse scaling and pruritus.4 Aside from drug reactions, other causes of exfoliative dermatitis such as psoriasis, atopic and seborrheic dermatitis, mycosis fungoides, and lymphoma should be ruled out. Other drug eruptions that can be classified as dermatologic emergencies include leukocytoclastic vasculitis, severe urticaria or angioedema, erythema multiforme, or Stevens-Johnson syndrome and toxic epidermal necrolysis.
Severe Acne
If not treated promptly, serious cases of acne can lead to severe scarring and psychologic problems. Acne fulminans is characterized by a rapid eruption of suppurative and large, highly inflamed nodules, plaques, and cysts that result in ragged ulcerations and cicatrization of the chest, back, and occasionally the face. Systemic symptoms of fever, arthralgia, leukocytosis, and myalgia suggest an upregulation of the immune system in affected patients.
Final Comment
In summary, dermatologic emergencies do exist and some may present with characteristic skin findings. In almost all cases, collaboration with other departments such as trauma, burn, internal medicine, rheumatology, and infectious diseases is extremely helpful in diagnosing and treating these medical emergencies. Collaboration can provide insight into how brainstorming through different approaches can lead to a better outcome whether it be solving the cause of a puzzling rash in a patient with multiple comorbidities or surgically removing a bullet from a trauma patient (Figure 4). Recognition of specific cutaneous manifestations and early diagnosis of dermatologic emergencies can be lifesaving.
1. McQueen A, Martin SA, Lio PA. Derm emergencies: detecting early signs of trouble. J Fam Pract. 2012;61:71-78.
2. Bennion S. Dermatologic emergencies. In: Fitzpatrick J, Morelli J, eds. Dermatology Secrets Plus. 4th ed. Philadelphia, PA: Mosby; 2011:442-452.
3. Sarani B, Strong M, Pascual J, et al. Necrotizing fasciitis: current concepts and review of the literature. J Am Coll Surg. 2009;208:279-288.
4. Wolf R, Orion E, Marcos B, et al. Life-threatening acute adverse cutaneous drug reactions. Clin Dermatol. 2005;23:171-181.
1. McQueen A, Martin SA, Lio PA. Derm emergencies: detecting early signs of trouble. J Fam Pract. 2012;61:71-78.
2. Bennion S. Dermatologic emergencies. In: Fitzpatrick J, Morelli J, eds. Dermatology Secrets Plus. 4th ed. Philadelphia, PA: Mosby; 2011:442-452.
3. Sarani B, Strong M, Pascual J, et al. Necrotizing fasciitis: current concepts and review of the literature. J Am Coll Surg. 2009;208:279-288.
4. Wolf R, Orion E, Marcos B, et al. Life-threatening acute adverse cutaneous drug reactions. Clin Dermatol. 2005;23:171-181.