User login
Pennsylvania joins states with ‘I’m sorry’ laws
Pennsylvania physicians can now apologize to patients after a poor medical outcome without fear that it will be used against them in court.
The Benevolent Gesture Medical Professional Liability Act unanimously passed the Pennsylvania House of Representatives Oct. 22 and was signed by Gov. Tom Corbett, a Republican, the following day. The legislation is in line with other "I’m sorry" laws across the country, said Dr. C. Richard Schott, former president of the Pennsylvania Medical Society (PAMED).
"Physicians, not only in Pennsylvania but around the country, have been concerned that if they offer a benevolent gesture in the setting of a bad outcome, that they can be held liable for their expression of sympathy or empathy to the patient," Dr. Schott said. "It becomes a barrier to otherwise appropriate communication. Other states have this type of legislation in various forms, and we are delighted that it passed" in Pennsylvania.
More than 30 states have some form of apology statute aimed at physicians and other health care providers. The language and extent of legal protection however, depends on the jurisdiction, said Dr. Marlynn Wei, a psychiatrist and attorney in New York City.
Pennsylvania law’s protects any action, conduct, or statement that conveys a sense of apology, condolence, explanation, compassion, or commiseration "emanating from humane impulses." The statute does not protect factual statements or admissions of guilt for a medical outcome or error.
Other states such as Colorado and Idaho shield physicians against admissions of fault being used against them in court.
"The goals of [apology] laws are to be on the side with the patient as opposed to pitting the doctor and patient against each other," said Dr. Wei, who authored an article on the use and effectiveness of state apology laws (J. Health Law 2007;40:107-59). "These laws allow us to be compassionate."
Whether apology laws actually reduce litigation is unclear, Dr. Wei said. Most patients who experience a poor medical outcome want an apology, but whether they ultimately sue depends on the severity of the injury and whether they believe negligence was involved.
"Most apology laws say these [sympathetic] statements cannot be used against you in court, but they don’t prevent a lawsuit from happening," she said.
In Pennsylvania, both physicians and plaintiffs’ attorneys supported the new law . Proponents said agreement between the two sides led to the legislation’s swift passage.
"Stakeholders were able to put aside their competing interests to come to a compromise," Pennsylvania Sen. Pat Vance (R-Cumberland) said in a statement.
The law’s simple intent made the statute easy for doctors and plaintiffs’ attorneys to agree upon, said George B. Faller Jr., a personal injury attorney in Carlisle, Pa.
"The lawyers defending doctors are O.K. with it because [physicians] can say they’re sorry without it being admissible," he said. "The plaintiffs’ lawyers on the other hand, like the fact that if there are factual statements made or admissions of guilt, those are admissible."
Pennsylvania physicians can now apologize to patients after a poor medical outcome without fear that it will be used against them in court.
The Benevolent Gesture Medical Professional Liability Act unanimously passed the Pennsylvania House of Representatives Oct. 22 and was signed by Gov. Tom Corbett, a Republican, the following day. The legislation is in line with other "I’m sorry" laws across the country, said Dr. C. Richard Schott, former president of the Pennsylvania Medical Society (PAMED).
"Physicians, not only in Pennsylvania but around the country, have been concerned that if they offer a benevolent gesture in the setting of a bad outcome, that they can be held liable for their expression of sympathy or empathy to the patient," Dr. Schott said. "It becomes a barrier to otherwise appropriate communication. Other states have this type of legislation in various forms, and we are delighted that it passed" in Pennsylvania.
More than 30 states have some form of apology statute aimed at physicians and other health care providers. The language and extent of legal protection however, depends on the jurisdiction, said Dr. Marlynn Wei, a psychiatrist and attorney in New York City.
Pennsylvania law’s protects any action, conduct, or statement that conveys a sense of apology, condolence, explanation, compassion, or commiseration "emanating from humane impulses." The statute does not protect factual statements or admissions of guilt for a medical outcome or error.
Other states such as Colorado and Idaho shield physicians against admissions of fault being used against them in court.
"The goals of [apology] laws are to be on the side with the patient as opposed to pitting the doctor and patient against each other," said Dr. Wei, who authored an article on the use and effectiveness of state apology laws (J. Health Law 2007;40:107-59). "These laws allow us to be compassionate."
Whether apology laws actually reduce litigation is unclear, Dr. Wei said. Most patients who experience a poor medical outcome want an apology, but whether they ultimately sue depends on the severity of the injury and whether they believe negligence was involved.
"Most apology laws say these [sympathetic] statements cannot be used against you in court, but they don’t prevent a lawsuit from happening," she said.
In Pennsylvania, both physicians and plaintiffs’ attorneys supported the new law . Proponents said agreement between the two sides led to the legislation’s swift passage.
"Stakeholders were able to put aside their competing interests to come to a compromise," Pennsylvania Sen. Pat Vance (R-Cumberland) said in a statement.
The law’s simple intent made the statute easy for doctors and plaintiffs’ attorneys to agree upon, said George B. Faller Jr., a personal injury attorney in Carlisle, Pa.
"The lawyers defending doctors are O.K. with it because [physicians] can say they’re sorry without it being admissible," he said. "The plaintiffs’ lawyers on the other hand, like the fact that if there are factual statements made or admissions of guilt, those are admissible."
Pennsylvania physicians can now apologize to patients after a poor medical outcome without fear that it will be used against them in court.
The Benevolent Gesture Medical Professional Liability Act unanimously passed the Pennsylvania House of Representatives Oct. 22 and was signed by Gov. Tom Corbett, a Republican, the following day. The legislation is in line with other "I’m sorry" laws across the country, said Dr. C. Richard Schott, former president of the Pennsylvania Medical Society (PAMED).
"Physicians, not only in Pennsylvania but around the country, have been concerned that if they offer a benevolent gesture in the setting of a bad outcome, that they can be held liable for their expression of sympathy or empathy to the patient," Dr. Schott said. "It becomes a barrier to otherwise appropriate communication. Other states have this type of legislation in various forms, and we are delighted that it passed" in Pennsylvania.
More than 30 states have some form of apology statute aimed at physicians and other health care providers. The language and extent of legal protection however, depends on the jurisdiction, said Dr. Marlynn Wei, a psychiatrist and attorney in New York City.
Pennsylvania law’s protects any action, conduct, or statement that conveys a sense of apology, condolence, explanation, compassion, or commiseration "emanating from humane impulses." The statute does not protect factual statements or admissions of guilt for a medical outcome or error.
Other states such as Colorado and Idaho shield physicians against admissions of fault being used against them in court.
"The goals of [apology] laws are to be on the side with the patient as opposed to pitting the doctor and patient against each other," said Dr. Wei, who authored an article on the use and effectiveness of state apology laws (J. Health Law 2007;40:107-59). "These laws allow us to be compassionate."
Whether apology laws actually reduce litigation is unclear, Dr. Wei said. Most patients who experience a poor medical outcome want an apology, but whether they ultimately sue depends on the severity of the injury and whether they believe negligence was involved.
"Most apology laws say these [sympathetic] statements cannot be used against you in court, but they don’t prevent a lawsuit from happening," she said.
In Pennsylvania, both physicians and plaintiffs’ attorneys supported the new law . Proponents said agreement between the two sides led to the legislation’s swift passage.
"Stakeholders were able to put aside their competing interests to come to a compromise," Pennsylvania Sen. Pat Vance (R-Cumberland) said in a statement.
The law’s simple intent made the statute easy for doctors and plaintiffs’ attorneys to agree upon, said George B. Faller Jr., a personal injury attorney in Carlisle, Pa.
"The lawyers defending doctors are O.K. with it because [physicians] can say they’re sorry without it being admissible," he said. "The plaintiffs’ lawyers on the other hand, like the fact that if there are factual statements made or admissions of guilt, those are admissible."
ACA could pose unintentional legal dangers for physicians
Federal quality metrics – integrated into the Affordable Care Act to improve and standardize care – are posing unintended legal risks for physicians, medical malpractice experts noted.
"The Affordable Care Act itself doesn’t identify medical malpractice issues," according to Aldo Leiva, a health law attorney in Coral Gables, Fla. "The concern that has arisen has been whether or not the content or language in the [ACA] can be used by plaintiffs’ lawyers against doctors by creating an additional standard of care."
Attorneys and insurers already are hearing reports of federal reimbursement decisions being introduced into medical malpractice cases. In such instances, lawyers use federal payment denials to bolster their claims of negligence, according to Mike Stinson, director of government relations for PIAA, a national trade association representing medical liability insurers.
Hospital readmission standards are one such federal quality measurement that could unfairly impact doctors in court, said Brian K. Atchinson, PIAA president and CEO. The ACA reduces payments to hospitals considered to have excessive readmissions.
"The mere fact that there will be many thousands of people that will be readmitted to a hospital within 30 days, that should not be evidence of inadequate care," Mr. Atchinson said.
Penalties regarding hospital-acquired conditions and value-based incentive payments also could impact lawsuits.
Lawyers are creative and will search for clever ways to further their argument, Mr. Leiva said. "There’s nothing to stop [them] from using that to move the needle a little further" to support their claim."
State legislatures are working to remedy this growing legal jeopardy. Georgia recently passed laws banning federal quality measures from being used to create standards in medical liability cases. Florida has had a similar law on the books since 2011.
Before Georgia’s law was enacted in May, plaintiffs’ attorneys had sought to have information about Medicare reimbursement denials presented to medical malpractice juries, according to Joseph L. Cregan, senior vice president and general counsel for MAG Mutual Insurance, a medical liability insurer that operates in the Southeast.
"Obviously, placing these issues before the jury could adversely affect our physician insureds, because it ... encourages a juror to impose a simplistic analysis instead of the proper legal analysis," Mr. Cregan said. "Our desire was that H.B. 499 would clarify that traditional ‘reasonable physician’ standard of care analysis remains the fundamental key to judging our Georgia physicians."
Since Georgia’s enactment of H.B. 499, other states, including Alabama and Mississippi, have expressed interest in considering similar laws, Mr. Cregan said.
"I think you are going to see the H.B. 499 idea introduced in other state legislatures in 2014, and I think you will see the issue debated and passed in a number of states," he said.
Others are looking toward federal legislation as a solution to unintentional ACA legal traps.
H.R. 1473, the Standard of Care Protection Act, was introduced by Rep. Phil Gingrey (R-Ga.), and is supported by the American Medical Association. It is currently under consideration by subcommittees of the Energy and Commerce Commiteee and the Judiciary Committee; no Senate companion has been introduced.
The bill would prevent ACA guidelines and standards from being construed to establish a standard of care in medical malpractice cases. It also would prevent those provisions from preempting state laws that govern medical liability.
"There should be no room for misinterpretation of the [ACA] to create new causes of action or trump state medical malpractice laws," Rep. Gloria Negrete McLeod (D-Calif.), a cosponsor of the bill, said in a statement.
The bill also was included in H.R. 2810, a bill to replace the Medicare Sustainable Growth Rate formula, which was passed out of the Energy and Commerce Committee in July. Proponents are cautiously optimistic about the bill’s success.
"Over the autumn, we’re going to be paying close attention to how this legislation proceeds," Mr. Atchinson of PIAA said. "We certainly believe it would be good for both the physicians and the nation’s patients."
Federal quality metrics – integrated into the Affordable Care Act to improve and standardize care – are posing unintended legal risks for physicians, medical malpractice experts noted.
"The Affordable Care Act itself doesn’t identify medical malpractice issues," according to Aldo Leiva, a health law attorney in Coral Gables, Fla. "The concern that has arisen has been whether or not the content or language in the [ACA] can be used by plaintiffs’ lawyers against doctors by creating an additional standard of care."
Attorneys and insurers already are hearing reports of federal reimbursement decisions being introduced into medical malpractice cases. In such instances, lawyers use federal payment denials to bolster their claims of negligence, according to Mike Stinson, director of government relations for PIAA, a national trade association representing medical liability insurers.
Hospital readmission standards are one such federal quality measurement that could unfairly impact doctors in court, said Brian K. Atchinson, PIAA president and CEO. The ACA reduces payments to hospitals considered to have excessive readmissions.
"The mere fact that there will be many thousands of people that will be readmitted to a hospital within 30 days, that should not be evidence of inadequate care," Mr. Atchinson said.
Penalties regarding hospital-acquired conditions and value-based incentive payments also could impact lawsuits.
Lawyers are creative and will search for clever ways to further their argument, Mr. Leiva said. "There’s nothing to stop [them] from using that to move the needle a little further" to support their claim."
State legislatures are working to remedy this growing legal jeopardy. Georgia recently passed laws banning federal quality measures from being used to create standards in medical liability cases. Florida has had a similar law on the books since 2011.
Before Georgia’s law was enacted in May, plaintiffs’ attorneys had sought to have information about Medicare reimbursement denials presented to medical malpractice juries, according to Joseph L. Cregan, senior vice president and general counsel for MAG Mutual Insurance, a medical liability insurer that operates in the Southeast.
"Obviously, placing these issues before the jury could adversely affect our physician insureds, because it ... encourages a juror to impose a simplistic analysis instead of the proper legal analysis," Mr. Cregan said. "Our desire was that H.B. 499 would clarify that traditional ‘reasonable physician’ standard of care analysis remains the fundamental key to judging our Georgia physicians."
Since Georgia’s enactment of H.B. 499, other states, including Alabama and Mississippi, have expressed interest in considering similar laws, Mr. Cregan said.
"I think you are going to see the H.B. 499 idea introduced in other state legislatures in 2014, and I think you will see the issue debated and passed in a number of states," he said.
Others are looking toward federal legislation as a solution to unintentional ACA legal traps.
H.R. 1473, the Standard of Care Protection Act, was introduced by Rep. Phil Gingrey (R-Ga.), and is supported by the American Medical Association. It is currently under consideration by subcommittees of the Energy and Commerce Commiteee and the Judiciary Committee; no Senate companion has been introduced.
The bill would prevent ACA guidelines and standards from being construed to establish a standard of care in medical malpractice cases. It also would prevent those provisions from preempting state laws that govern medical liability.
"There should be no room for misinterpretation of the [ACA] to create new causes of action or trump state medical malpractice laws," Rep. Gloria Negrete McLeod (D-Calif.), a cosponsor of the bill, said in a statement.
The bill also was included in H.R. 2810, a bill to replace the Medicare Sustainable Growth Rate formula, which was passed out of the Energy and Commerce Committee in July. Proponents are cautiously optimistic about the bill’s success.
"Over the autumn, we’re going to be paying close attention to how this legislation proceeds," Mr. Atchinson of PIAA said. "We certainly believe it would be good for both the physicians and the nation’s patients."
Federal quality metrics – integrated into the Affordable Care Act to improve and standardize care – are posing unintended legal risks for physicians, medical malpractice experts noted.
"The Affordable Care Act itself doesn’t identify medical malpractice issues," according to Aldo Leiva, a health law attorney in Coral Gables, Fla. "The concern that has arisen has been whether or not the content or language in the [ACA] can be used by plaintiffs’ lawyers against doctors by creating an additional standard of care."
Attorneys and insurers already are hearing reports of federal reimbursement decisions being introduced into medical malpractice cases. In such instances, lawyers use federal payment denials to bolster their claims of negligence, according to Mike Stinson, director of government relations for PIAA, a national trade association representing medical liability insurers.
Hospital readmission standards are one such federal quality measurement that could unfairly impact doctors in court, said Brian K. Atchinson, PIAA president and CEO. The ACA reduces payments to hospitals considered to have excessive readmissions.
"The mere fact that there will be many thousands of people that will be readmitted to a hospital within 30 days, that should not be evidence of inadequate care," Mr. Atchinson said.
Penalties regarding hospital-acquired conditions and value-based incentive payments also could impact lawsuits.
Lawyers are creative and will search for clever ways to further their argument, Mr. Leiva said. "There’s nothing to stop [them] from using that to move the needle a little further" to support their claim."
State legislatures are working to remedy this growing legal jeopardy. Georgia recently passed laws banning federal quality measures from being used to create standards in medical liability cases. Florida has had a similar law on the books since 2011.
Before Georgia’s law was enacted in May, plaintiffs’ attorneys had sought to have information about Medicare reimbursement denials presented to medical malpractice juries, according to Joseph L. Cregan, senior vice president and general counsel for MAG Mutual Insurance, a medical liability insurer that operates in the Southeast.
"Obviously, placing these issues before the jury could adversely affect our physician insureds, because it ... encourages a juror to impose a simplistic analysis instead of the proper legal analysis," Mr. Cregan said. "Our desire was that H.B. 499 would clarify that traditional ‘reasonable physician’ standard of care analysis remains the fundamental key to judging our Georgia physicians."
Since Georgia’s enactment of H.B. 499, other states, including Alabama and Mississippi, have expressed interest in considering similar laws, Mr. Cregan said.
"I think you are going to see the H.B. 499 idea introduced in other state legislatures in 2014, and I think you will see the issue debated and passed in a number of states," he said.
Others are looking toward federal legislation as a solution to unintentional ACA legal traps.
H.R. 1473, the Standard of Care Protection Act, was introduced by Rep. Phil Gingrey (R-Ga.), and is supported by the American Medical Association. It is currently under consideration by subcommittees of the Energy and Commerce Commiteee and the Judiciary Committee; no Senate companion has been introduced.
The bill would prevent ACA guidelines and standards from being construed to establish a standard of care in medical malpractice cases. It also would prevent those provisions from preempting state laws that govern medical liability.
"There should be no room for misinterpretation of the [ACA] to create new causes of action or trump state medical malpractice laws," Rep. Gloria Negrete McLeod (D-Calif.), a cosponsor of the bill, said in a statement.
The bill also was included in H.R. 2810, a bill to replace the Medicare Sustainable Growth Rate formula, which was passed out of the Energy and Commerce Committee in July. Proponents are cautiously optimistic about the bill’s success.
"Over the autumn, we’re going to be paying close attention to how this legislation proceeds," Mr. Atchinson of PIAA said. "We certainly believe it would be good for both the physicians and the nation’s patients."
Tavenner apologizes for healthcare.gov woes
WASHINGTON – Appearing before a House committee, Medicare Chief Marilyn Tavenner apologized for the problems Americans are experiencing with healthcare.gov.
Thirty-six states are using the federal health insurance exchange, which relies on healthcare.gov as the main avenue for shopping and enrolling in the health insurance made available under the Affordable Care Act. The website has been plagued by glitches since it went live on Oct. 1. Ms. Tavenner, administrator of the Centers for Medicare and Medicaid Services (CMS), is the first administration official to appear before Congress to discuss the rollout of the exchanges.
"To the millions of Americans who’ve attempted to use healthcare.gov to shop and enroll in health care coverage, I want to apologize to you that the website has not worked as well as it should," Ms. Tavenner said at an Oct. 29 hearing of the House Ways and Means Committee. "I want to assure you that healthcare.gov can and will be fixed."
Committee Chairman Dave Camp (R-Mich.) said that "three years should have been enough" time to make sure the website functioned properly, and that the website problems were small compared to overall issues with the ACA [Affordable Care Act]. He said that it appeared that many thousands of Americans were being dropped by their current plans – more, apparently, than were getting signed up for new coverage.
"The numbers I’m hearing from insurers in my home state of Michigan are not good," said Rep. Camp, referring to how many had enrolled in coverage through the exchange there, which is being run in partnership with the state. "In fact, I think I could have a meeting in my office and have all of them fit in it."
When asked by Rep. Camp and other Republicans for details on the numbers of people who have actually enrolled in health insurance via the federal exchange, Ms. Tavenner repeatedly said that those figures would be available in mid-November.
Rep. Camp expressed concerns that given the website problems, it seemed unlikely that the administration would be able to enroll 7 million Americans by late March, and more importantly, to attract large numbers of young and healthy enrollees. Without them, the exchange plans might end up being overly populated with higher-risk, sicker enrollees, he said.
Ms. Tavenner said that premiums were "locked in" for 2014, but that "obviously, the next 6 months of enrollment are critical."
Ways and Means Committee Democrats largely defended the law and said the website would be fixed soon enough. But some were not as quick to apologize for the failures. Rep. Lloyd Doggett (D-Tex.) said that while Republican delaying tactics might be harmful, "I recognize that the promise of affordable health care could also be denied through management failure."
He asked Ms. Tavenner whether both the small business exchanges and a Spanish-language version of healthcare.gov would be up and running by November. She said they would.
Ms. Tavenner emphasized that the system is working. "It’s just not working at the speed that we want and at the success rate that we wanted."
At a press briefing the same day, Julie Bataille, a CMS spokeswoman, said the agency had stabilized the problems that consumers were previously having creating accounts on healthcare.gov. After doubling the number of servers and making software enhancements, the agency is now able to process nearly 17,000 registrants per hour through the account creation step with few errors, she said.
aault@frontlinemedcom.com On Twitter @aliciaault
Committee Chairman Dave Camp,
WASHINGTON – Appearing before a House committee, Medicare Chief Marilyn Tavenner apologized for the problems Americans are experiencing with healthcare.gov.
Thirty-six states are using the federal health insurance exchange, which relies on healthcare.gov as the main avenue for shopping and enrolling in the health insurance made available under the Affordable Care Act. The website has been plagued by glitches since it went live on Oct. 1. Ms. Tavenner, administrator of the Centers for Medicare and Medicaid Services (CMS), is the first administration official to appear before Congress to discuss the rollout of the exchanges.
"To the millions of Americans who’ve attempted to use healthcare.gov to shop and enroll in health care coverage, I want to apologize to you that the website has not worked as well as it should," Ms. Tavenner said at an Oct. 29 hearing of the House Ways and Means Committee. "I want to assure you that healthcare.gov can and will be fixed."
Committee Chairman Dave Camp (R-Mich.) said that "three years should have been enough" time to make sure the website functioned properly, and that the website problems were small compared to overall issues with the ACA [Affordable Care Act]. He said that it appeared that many thousands of Americans were being dropped by their current plans – more, apparently, than were getting signed up for new coverage.
"The numbers I’m hearing from insurers in my home state of Michigan are not good," said Rep. Camp, referring to how many had enrolled in coverage through the exchange there, which is being run in partnership with the state. "In fact, I think I could have a meeting in my office and have all of them fit in it."
When asked by Rep. Camp and other Republicans for details on the numbers of people who have actually enrolled in health insurance via the federal exchange, Ms. Tavenner repeatedly said that those figures would be available in mid-November.
Rep. Camp expressed concerns that given the website problems, it seemed unlikely that the administration would be able to enroll 7 million Americans by late March, and more importantly, to attract large numbers of young and healthy enrollees. Without them, the exchange plans might end up being overly populated with higher-risk, sicker enrollees, he said.
Ms. Tavenner said that premiums were "locked in" for 2014, but that "obviously, the next 6 months of enrollment are critical."
Ways and Means Committee Democrats largely defended the law and said the website would be fixed soon enough. But some were not as quick to apologize for the failures. Rep. Lloyd Doggett (D-Tex.) said that while Republican delaying tactics might be harmful, "I recognize that the promise of affordable health care could also be denied through management failure."
He asked Ms. Tavenner whether both the small business exchanges and a Spanish-language version of healthcare.gov would be up and running by November. She said they would.
Ms. Tavenner emphasized that the system is working. "It’s just not working at the speed that we want and at the success rate that we wanted."
At a press briefing the same day, Julie Bataille, a CMS spokeswoman, said the agency had stabilized the problems that consumers were previously having creating accounts on healthcare.gov. After doubling the number of servers and making software enhancements, the agency is now able to process nearly 17,000 registrants per hour through the account creation step with few errors, she said.
aault@frontlinemedcom.com On Twitter @aliciaault
WASHINGTON – Appearing before a House committee, Medicare Chief Marilyn Tavenner apologized for the problems Americans are experiencing with healthcare.gov.
Thirty-six states are using the federal health insurance exchange, which relies on healthcare.gov as the main avenue for shopping and enrolling in the health insurance made available under the Affordable Care Act. The website has been plagued by glitches since it went live on Oct. 1. Ms. Tavenner, administrator of the Centers for Medicare and Medicaid Services (CMS), is the first administration official to appear before Congress to discuss the rollout of the exchanges.
"To the millions of Americans who’ve attempted to use healthcare.gov to shop and enroll in health care coverage, I want to apologize to you that the website has not worked as well as it should," Ms. Tavenner said at an Oct. 29 hearing of the House Ways and Means Committee. "I want to assure you that healthcare.gov can and will be fixed."
Committee Chairman Dave Camp (R-Mich.) said that "three years should have been enough" time to make sure the website functioned properly, and that the website problems were small compared to overall issues with the ACA [Affordable Care Act]. He said that it appeared that many thousands of Americans were being dropped by their current plans – more, apparently, than were getting signed up for new coverage.
"The numbers I’m hearing from insurers in my home state of Michigan are not good," said Rep. Camp, referring to how many had enrolled in coverage through the exchange there, which is being run in partnership with the state. "In fact, I think I could have a meeting in my office and have all of them fit in it."
When asked by Rep. Camp and other Republicans for details on the numbers of people who have actually enrolled in health insurance via the federal exchange, Ms. Tavenner repeatedly said that those figures would be available in mid-November.
Rep. Camp expressed concerns that given the website problems, it seemed unlikely that the administration would be able to enroll 7 million Americans by late March, and more importantly, to attract large numbers of young and healthy enrollees. Without them, the exchange plans might end up being overly populated with higher-risk, sicker enrollees, he said.
Ms. Tavenner said that premiums were "locked in" for 2014, but that "obviously, the next 6 months of enrollment are critical."
Ways and Means Committee Democrats largely defended the law and said the website would be fixed soon enough. But some were not as quick to apologize for the failures. Rep. Lloyd Doggett (D-Tex.) said that while Republican delaying tactics might be harmful, "I recognize that the promise of affordable health care could also be denied through management failure."
He asked Ms. Tavenner whether both the small business exchanges and a Spanish-language version of healthcare.gov would be up and running by November. She said they would.
Ms. Tavenner emphasized that the system is working. "It’s just not working at the speed that we want and at the success rate that we wanted."
At a press briefing the same day, Julie Bataille, a CMS spokeswoman, said the agency had stabilized the problems that consumers were previously having creating accounts on healthcare.gov. After doubling the number of servers and making software enhancements, the agency is now able to process nearly 17,000 registrants per hour through the account creation step with few errors, she said.
aault@frontlinemedcom.com On Twitter @aliciaault
Committee Chairman Dave Camp,
Committee Chairman Dave Camp,
AT A HOUSE WAYS AND MEANS COMMITTEE HEARING
Med school applications spike
The number of students applying to medical school hit an all-time high this year, according to figures from the Association of American Medical Colleges.
More than 48,000 students applied to enter U.S. medical schools in 2013, a jump of 6% from the previous year. Most of the aspiring doctors – more than 35,000 – are first-time applicants.
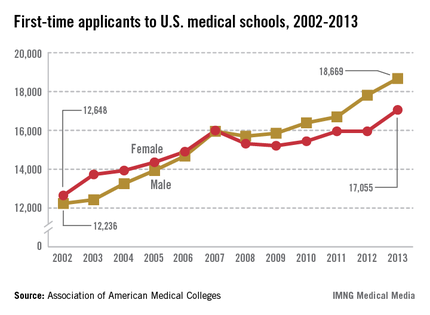
"We haven’t seen a level like this since 1996," said Dr. Darrell G. Kirch, president and CEO of the Association of American Medical Colleges (AAMC).
First-time medical school enrollment also set records this year. More than 20,000 students enrolled in their first year of medical school, a nearly 3% rise over the 2012 figures.
Dr. Kirch said college students are "voting with their feet" because they view medicine as an attractive career choice.
Medical schools have been able to accommodate this renewed interested in medicine because existing programs have expanded and some new medical schools have opened. This year, 14 medical schools increased their class sizes by more than 10% and four new medical schools opened their doors, according to the AAMC.
The enrollment and application numbers are good news as the nation faces a shortage of physicians, Dr. Kirch said. But the surge in students entering medical school won’t mean anything if they don’t have residency programs to go to after graduation, he said.
The problem is that residency training programs aren’t growing because Congress capped federal funding for residency training back in 1997. The funding restrictions severely limit the number of new slots that academic medical centers can afford to support, he said.
Dr. Kirch called on lawmakers to pass federal legislation that would lift the funding caps.
"If they don’t do it, we could face a serious shortage of physicians across the board," he said.
mschneider@frontlinemedcom.com
On Twitter @MaryEllenNY
The number of students applying to medical school hit an all-time high this year, according to figures from the Association of American Medical Colleges.
More than 48,000 students applied to enter U.S. medical schools in 2013, a jump of 6% from the previous year. Most of the aspiring doctors – more than 35,000 – are first-time applicants.

"We haven’t seen a level like this since 1996," said Dr. Darrell G. Kirch, president and CEO of the Association of American Medical Colleges (AAMC).
First-time medical school enrollment also set records this year. More than 20,000 students enrolled in their first year of medical school, a nearly 3% rise over the 2012 figures.
Dr. Kirch said college students are "voting with their feet" because they view medicine as an attractive career choice.
Medical schools have been able to accommodate this renewed interested in medicine because existing programs have expanded and some new medical schools have opened. This year, 14 medical schools increased their class sizes by more than 10% and four new medical schools opened their doors, according to the AAMC.
The enrollment and application numbers are good news as the nation faces a shortage of physicians, Dr. Kirch said. But the surge in students entering medical school won’t mean anything if they don’t have residency programs to go to after graduation, he said.
The problem is that residency training programs aren’t growing because Congress capped federal funding for residency training back in 1997. The funding restrictions severely limit the number of new slots that academic medical centers can afford to support, he said.
Dr. Kirch called on lawmakers to pass federal legislation that would lift the funding caps.
"If they don’t do it, we could face a serious shortage of physicians across the board," he said.
mschneider@frontlinemedcom.com
On Twitter @MaryEllenNY
The number of students applying to medical school hit an all-time high this year, according to figures from the Association of American Medical Colleges.
More than 48,000 students applied to enter U.S. medical schools in 2013, a jump of 6% from the previous year. Most of the aspiring doctors – more than 35,000 – are first-time applicants.

"We haven’t seen a level like this since 1996," said Dr. Darrell G. Kirch, president and CEO of the Association of American Medical Colleges (AAMC).
First-time medical school enrollment also set records this year. More than 20,000 students enrolled in their first year of medical school, a nearly 3% rise over the 2012 figures.
Dr. Kirch said college students are "voting with their feet" because they view medicine as an attractive career choice.
Medical schools have been able to accommodate this renewed interested in medicine because existing programs have expanded and some new medical schools have opened. This year, 14 medical schools increased their class sizes by more than 10% and four new medical schools opened their doors, according to the AAMC.
The enrollment and application numbers are good news as the nation faces a shortage of physicians, Dr. Kirch said. But the surge in students entering medical school won’t mean anything if they don’t have residency programs to go to after graduation, he said.
The problem is that residency training programs aren’t growing because Congress capped federal funding for residency training back in 1997. The funding restrictions severely limit the number of new slots that academic medical centers can afford to support, he said.
Dr. Kirch called on lawmakers to pass federal legislation that would lift the funding caps.
"If they don’t do it, we could face a serious shortage of physicians across the board," he said.
mschneider@frontlinemedcom.com
On Twitter @MaryEllenNY
Physicians alarmed by high rate of Medicare claims denied in error
As Medicare auditors have increased their workloads in recent years, doctors and hospitals are appealing more decisions that deny claims for beneficiary services.
Physicians sought to overturn denials 1.5 million times in 2012, which represented a 9% increase since 2008, according to an October report from the Health and Human Services department’s Office of Inspector General (OIG). Hospitals have appealed far fewer denied claims, but Medicare auditors, such as Recovery Audit Contractors (RACs), are driving more inpatient providers to enter the redetermination process, the OIG reported.
Physicians continue to have serious concerns about the inaccuracy of Medicare audit contractors, according to Dr. Ardis Dee Hoven, president of the American Medical Association.
The OIG’s report pointed out that "physicians who invest the time and expense to dispute contractor determinations prevail 54% of the time at the first level of appeal," Dr. Hoven said. "This contractor error rate is far too high, and physician practices should not have to undergo burdensome audits – including RAC audits – while this problem remains unaddressed."
Medicare processed more than 1.2 billion claims and denied nearly 140 million of them in 2012. Of those, 2.6% were appealed via the first-level appeals process. The OIG report did not provide details on why physician claims were denied; however, many Part A hospital benefit appeals stemmed from a RAC decision regarding short-term inpatient hospital stays.
The Centers for Medicare and Medicaid Services is implementing a new appeals system that soon will offer more information about appeals. Four contractors have begun using the system to process Part A redeterminations this year, CMS administrator Marilyn Tavenner said in an August memo to the OIG.
Contractors largely meet the timeframes established for redetermination requests, which is good news to practices that actively appeal claims, said Kent Moore, senior physician payment strategist at the American Academy of Family Physicians.
"If you appeal and you’re successful, you can get your money in a timely manner," Mr. Moore said. "If you’re not successful, then it might take longer than expected."
The OIG report found for favorably appealed claims, contractors paid physicians within 30 days 93% of the time. However, denied claims that are appealed to a second level can take 6 months to decide. In 2012, contractors transferred more than 280,000 Part B claims to the second level.
As Medicare auditors have increased their workloads in recent years, doctors and hospitals are appealing more decisions that deny claims for beneficiary services.
Physicians sought to overturn denials 1.5 million times in 2012, which represented a 9% increase since 2008, according to an October report from the Health and Human Services department’s Office of Inspector General (OIG). Hospitals have appealed far fewer denied claims, but Medicare auditors, such as Recovery Audit Contractors (RACs), are driving more inpatient providers to enter the redetermination process, the OIG reported.
Physicians continue to have serious concerns about the inaccuracy of Medicare audit contractors, according to Dr. Ardis Dee Hoven, president of the American Medical Association.
The OIG’s report pointed out that "physicians who invest the time and expense to dispute contractor determinations prevail 54% of the time at the first level of appeal," Dr. Hoven said. "This contractor error rate is far too high, and physician practices should not have to undergo burdensome audits – including RAC audits – while this problem remains unaddressed."
Medicare processed more than 1.2 billion claims and denied nearly 140 million of them in 2012. Of those, 2.6% were appealed via the first-level appeals process. The OIG report did not provide details on why physician claims were denied; however, many Part A hospital benefit appeals stemmed from a RAC decision regarding short-term inpatient hospital stays.
The Centers for Medicare and Medicaid Services is implementing a new appeals system that soon will offer more information about appeals. Four contractors have begun using the system to process Part A redeterminations this year, CMS administrator Marilyn Tavenner said in an August memo to the OIG.
Contractors largely meet the timeframes established for redetermination requests, which is good news to practices that actively appeal claims, said Kent Moore, senior physician payment strategist at the American Academy of Family Physicians.
"If you appeal and you’re successful, you can get your money in a timely manner," Mr. Moore said. "If you’re not successful, then it might take longer than expected."
The OIG report found for favorably appealed claims, contractors paid physicians within 30 days 93% of the time. However, denied claims that are appealed to a second level can take 6 months to decide. In 2012, contractors transferred more than 280,000 Part B claims to the second level.
As Medicare auditors have increased their workloads in recent years, doctors and hospitals are appealing more decisions that deny claims for beneficiary services.
Physicians sought to overturn denials 1.5 million times in 2012, which represented a 9% increase since 2008, according to an October report from the Health and Human Services department’s Office of Inspector General (OIG). Hospitals have appealed far fewer denied claims, but Medicare auditors, such as Recovery Audit Contractors (RACs), are driving more inpatient providers to enter the redetermination process, the OIG reported.
Physicians continue to have serious concerns about the inaccuracy of Medicare audit contractors, according to Dr. Ardis Dee Hoven, president of the American Medical Association.
The OIG’s report pointed out that "physicians who invest the time and expense to dispute contractor determinations prevail 54% of the time at the first level of appeal," Dr. Hoven said. "This contractor error rate is far too high, and physician practices should not have to undergo burdensome audits – including RAC audits – while this problem remains unaddressed."
Medicare processed more than 1.2 billion claims and denied nearly 140 million of them in 2012. Of those, 2.6% were appealed via the first-level appeals process. The OIG report did not provide details on why physician claims were denied; however, many Part A hospital benefit appeals stemmed from a RAC decision regarding short-term inpatient hospital stays.
The Centers for Medicare and Medicaid Services is implementing a new appeals system that soon will offer more information about appeals. Four contractors have begun using the system to process Part A redeterminations this year, CMS administrator Marilyn Tavenner said in an August memo to the OIG.
Contractors largely meet the timeframes established for redetermination requests, which is good news to practices that actively appeal claims, said Kent Moore, senior physician payment strategist at the American Academy of Family Physicians.
"If you appeal and you’re successful, you can get your money in a timely manner," Mr. Moore said. "If you’re not successful, then it might take longer than expected."
The OIG report found for favorably appealed claims, contractors paid physicians within 30 days 93% of the time. However, denied claims that are appealed to a second level can take 6 months to decide. In 2012, contractors transferred more than 280,000 Part B claims to the second level.
HHS: Healthcare.gov fixes underway
Three weeks after the launch of the health insurance exchanges, federal officials said they are working around-the-clock to improve the functionality of the healthcare.gov website.
The Health and Human Services (HHS) department has been working to increase the site’s bandwidth to accommodate heavier-than-expected web traffic, improve the site’s architecture to make it easier for consumers to create online accounts, and ramp up testing of the system as each of the changes is made, Centers for Medicare and Medicaid Services (CMS) spokeswoman Julie Bataille said during a press conference Oct. 24.
And the federal data hub, which provides information used in determining eligibility for tax credits, is performing well with responses in seconds, she said.
"It is getting better every day," Ms. Bataille said. "We are making incremental improvements."
Consumers will notice a smoother and more consistent experience on healthcare.gov over time, Ms. Bataille said. And she added that consumers would be able to enroll in health insurance by Dec. 15 in order have coverage in place on Jan. 1.
The healthcare.gov site, which is the portal for accessing the 34 federally run health exchanges, has been plagued by glitches since its launch on Oct. 1, with consumers complaining that the site would freeze, go blank, or send error messages when they tried to create an account.
Some of those problems have been resolved, and more consumers are now able to create online accounts to apply for insurance, Ms. Bataille said.
In the first 3 weeks, consumers have completed about 700,000 applications for insurance across the state- and federally run health insurance exchanges, she said. That means that they have completed the application phase and received information on whether they are eligible for federal premium subsidies. However, Ms. Bataille said CMS would not release information on the number of people who have actually enrolled in health plans until November.
Federal officials are urging consumers who have trouble with the website to call 1-800-318-2596 instead. That hotline has received about 1.6 million calls since Oct. 1.
Three weeks after the launch of the health insurance exchanges, federal officials said they are working around-the-clock to improve the functionality of the healthcare.gov website.
The Health and Human Services (HHS) department has been working to increase the site’s bandwidth to accommodate heavier-than-expected web traffic, improve the site’s architecture to make it easier for consumers to create online accounts, and ramp up testing of the system as each of the changes is made, Centers for Medicare and Medicaid Services (CMS) spokeswoman Julie Bataille said during a press conference Oct. 24.
And the federal data hub, which provides information used in determining eligibility for tax credits, is performing well with responses in seconds, she said.
"It is getting better every day," Ms. Bataille said. "We are making incremental improvements."
Consumers will notice a smoother and more consistent experience on healthcare.gov over time, Ms. Bataille said. And she added that consumers would be able to enroll in health insurance by Dec. 15 in order have coverage in place on Jan. 1.
The healthcare.gov site, which is the portal for accessing the 34 federally run health exchanges, has been plagued by glitches since its launch on Oct. 1, with consumers complaining that the site would freeze, go blank, or send error messages when they tried to create an account.
Some of those problems have been resolved, and more consumers are now able to create online accounts to apply for insurance, Ms. Bataille said.
In the first 3 weeks, consumers have completed about 700,000 applications for insurance across the state- and federally run health insurance exchanges, she said. That means that they have completed the application phase and received information on whether they are eligible for federal premium subsidies. However, Ms. Bataille said CMS would not release information on the number of people who have actually enrolled in health plans until November.
Federal officials are urging consumers who have trouble with the website to call 1-800-318-2596 instead. That hotline has received about 1.6 million calls since Oct. 1.
Three weeks after the launch of the health insurance exchanges, federal officials said they are working around-the-clock to improve the functionality of the healthcare.gov website.
The Health and Human Services (HHS) department has been working to increase the site’s bandwidth to accommodate heavier-than-expected web traffic, improve the site’s architecture to make it easier for consumers to create online accounts, and ramp up testing of the system as each of the changes is made, Centers for Medicare and Medicaid Services (CMS) spokeswoman Julie Bataille said during a press conference Oct. 24.
And the federal data hub, which provides information used in determining eligibility for tax credits, is performing well with responses in seconds, she said.
"It is getting better every day," Ms. Bataille said. "We are making incremental improvements."
Consumers will notice a smoother and more consistent experience on healthcare.gov over time, Ms. Bataille said. And she added that consumers would be able to enroll in health insurance by Dec. 15 in order have coverage in place on Jan. 1.
The healthcare.gov site, which is the portal for accessing the 34 federally run health exchanges, has been plagued by glitches since its launch on Oct. 1, with consumers complaining that the site would freeze, go blank, or send error messages when they tried to create an account.
Some of those problems have been resolved, and more consumers are now able to create online accounts to apply for insurance, Ms. Bataille said.
In the first 3 weeks, consumers have completed about 700,000 applications for insurance across the state- and federally run health insurance exchanges, she said. That means that they have completed the application phase and received information on whether they are eligible for federal premium subsidies. However, Ms. Bataille said CMS would not release information on the number of people who have actually enrolled in health plans until November.
Federal officials are urging consumers who have trouble with the website to call 1-800-318-2596 instead. That hotline has received about 1.6 million calls since Oct. 1.
Contractors blame CMS for healthcare.gov woes
Contractors who helped develop healthcare.gov suggested that officials at the Centers for Medicare and Medicaid Services made decisions that contributed to the federal insurance exchange website’s woes over the last few weeks.
Representatives from four federal contractors testified about healthcare.gov at a 4-hour House Energy and Commerce Committee hearing on Oct. 24. Cheryl Campbell, senior vice president at CGI Federal, said that 2 weeks before the website’s official launch, CMS officials asked the company to turn off a feature that would let consumers shop for health plans before going through an official registration process.
The administration acknowledged on Oct. 20 that consumers "have had trouble creating accounts and logging in to the site, while others have received confusing error messages, or had to wait for slow page loads or forms that failed to respond in a timely fashion."
The website has been altered so that it is now possible to see available plans without registering.
At the hearing, Republicans and some Democrats wanted to know how the problems arose, what kinds of problems were being encountered, who had made decisions at the CMS, and when the problems would be fixed. And some noted that their concern went beyond just the functioning of healthcare.gov.
"This is more than a website problem – and frankly, the website should have been the easy part," said Committee Chairman Fred Upton (R-Mich.). "I’m also concerned about what happens next. Will enrollment glitches become provider payment glitches? Will patients show up at their doctor’s office or hospital only to be told they, or their coverage, aren’t in the system?"
Rep. Henry Waxman (D-Calif.) said website problems were just that. "The Affordable Care Act is an enormous success, with one obvious exception – it has a poorly designed website."
The contractors testified that normally, testing of such a large website would have been conducted for months before launch, but that healthcare.gov only received about 2 weeks of prelaunch testing. They also said that they were not aware of major problems until Oct. 1, the day the site opened to the public.
Andrew Slavitt, group executive vice president of Optum/QSSI, said that on Oct. 1, "the registration system was overwhelmed by concurrent users."
But the contractors also said that fixes to problems are being made on a real-time basis. Ms. Campbell said "the system is working," and that she expected that everything would be resolved in time for consumers to start coverage by Jan. 1.
Several Republicans said they did not have confidence in those predictions. Referring to the administration’s bringing in technology experts to help fix the website, Rep. Tim Murphy (R-Penn.) said, "Congress should press pause on the tech surge," until it is determined what went wrong.
Others called on the Obama administration to delay penalties for individuals who can’t get insurance coverage. Sen. Marco Rubio (R-Fla.) said he will introduce a bill soon to put off the penalties until healthcare.gov is "certified to be functioning."
On Twitter @aliciaault
Contractors who helped develop healthcare.gov suggested that officials at the Centers for Medicare and Medicaid Services made decisions that contributed to the federal insurance exchange website’s woes over the last few weeks.
Representatives from four federal contractors testified about healthcare.gov at a 4-hour House Energy and Commerce Committee hearing on Oct. 24. Cheryl Campbell, senior vice president at CGI Federal, said that 2 weeks before the website’s official launch, CMS officials asked the company to turn off a feature that would let consumers shop for health plans before going through an official registration process.
The administration acknowledged on Oct. 20 that consumers "have had trouble creating accounts and logging in to the site, while others have received confusing error messages, or had to wait for slow page loads or forms that failed to respond in a timely fashion."
The website has been altered so that it is now possible to see available plans without registering.
At the hearing, Republicans and some Democrats wanted to know how the problems arose, what kinds of problems were being encountered, who had made decisions at the CMS, and when the problems would be fixed. And some noted that their concern went beyond just the functioning of healthcare.gov.
"This is more than a website problem – and frankly, the website should have been the easy part," said Committee Chairman Fred Upton (R-Mich.). "I’m also concerned about what happens next. Will enrollment glitches become provider payment glitches? Will patients show up at their doctor’s office or hospital only to be told they, or their coverage, aren’t in the system?"
Rep. Henry Waxman (D-Calif.) said website problems were just that. "The Affordable Care Act is an enormous success, with one obvious exception – it has a poorly designed website."
The contractors testified that normally, testing of such a large website would have been conducted for months before launch, but that healthcare.gov only received about 2 weeks of prelaunch testing. They also said that they were not aware of major problems until Oct. 1, the day the site opened to the public.
Andrew Slavitt, group executive vice president of Optum/QSSI, said that on Oct. 1, "the registration system was overwhelmed by concurrent users."
But the contractors also said that fixes to problems are being made on a real-time basis. Ms. Campbell said "the system is working," and that she expected that everything would be resolved in time for consumers to start coverage by Jan. 1.
Several Republicans said they did not have confidence in those predictions. Referring to the administration’s bringing in technology experts to help fix the website, Rep. Tim Murphy (R-Penn.) said, "Congress should press pause on the tech surge," until it is determined what went wrong.
Others called on the Obama administration to delay penalties for individuals who can’t get insurance coverage. Sen. Marco Rubio (R-Fla.) said he will introduce a bill soon to put off the penalties until healthcare.gov is "certified to be functioning."
On Twitter @aliciaault
Contractors who helped develop healthcare.gov suggested that officials at the Centers for Medicare and Medicaid Services made decisions that contributed to the federal insurance exchange website’s woes over the last few weeks.
Representatives from four federal contractors testified about healthcare.gov at a 4-hour House Energy and Commerce Committee hearing on Oct. 24. Cheryl Campbell, senior vice president at CGI Federal, said that 2 weeks before the website’s official launch, CMS officials asked the company to turn off a feature that would let consumers shop for health plans before going through an official registration process.
The administration acknowledged on Oct. 20 that consumers "have had trouble creating accounts and logging in to the site, while others have received confusing error messages, or had to wait for slow page loads or forms that failed to respond in a timely fashion."
The website has been altered so that it is now possible to see available plans without registering.
At the hearing, Republicans and some Democrats wanted to know how the problems arose, what kinds of problems were being encountered, who had made decisions at the CMS, and when the problems would be fixed. And some noted that their concern went beyond just the functioning of healthcare.gov.
"This is more than a website problem – and frankly, the website should have been the easy part," said Committee Chairman Fred Upton (R-Mich.). "I’m also concerned about what happens next. Will enrollment glitches become provider payment glitches? Will patients show up at their doctor’s office or hospital only to be told they, or their coverage, aren’t in the system?"
Rep. Henry Waxman (D-Calif.) said website problems were just that. "The Affordable Care Act is an enormous success, with one obvious exception – it has a poorly designed website."
The contractors testified that normally, testing of such a large website would have been conducted for months before launch, but that healthcare.gov only received about 2 weeks of prelaunch testing. They also said that they were not aware of major problems until Oct. 1, the day the site opened to the public.
Andrew Slavitt, group executive vice president of Optum/QSSI, said that on Oct. 1, "the registration system was overwhelmed by concurrent users."
But the contractors also said that fixes to problems are being made on a real-time basis. Ms. Campbell said "the system is working," and that she expected that everything would be resolved in time for consumers to start coverage by Jan. 1.
Several Republicans said they did not have confidence in those predictions. Referring to the administration’s bringing in technology experts to help fix the website, Rep. Tim Murphy (R-Penn.) said, "Congress should press pause on the tech surge," until it is determined what went wrong.
Others called on the Obama administration to delay penalties for individuals who can’t get insurance coverage. Sen. Marco Rubio (R-Fla.) said he will introduce a bill soon to put off the penalties until healthcare.gov is "certified to be functioning."
On Twitter @aliciaault
FROM A HOUSE ENERGY AND COMMERCE COMMITTEE HEARING
Experts call for broad sharing of clinical trial data
Clinical trial data should be shared as broadly as possible to help spur scientific innovation and answer questions of importance to public health.
"The question is not whether, but how, these data should be broadly shared," wrote Michelle M. Mello and her colleagues from Harvard University, Boston, the Pharmaceutical Research and Manufacturers of America, and several consulting companies, in an article published online Oct. 21 in the New England Journal of Medicine.
At a minimum, data-sharing should be available for trials of all approved prescription drugs, devices, and biologics in any country that has adequate intellectual property protection. A system has to ensure responsible use of data, protect privacy of research participants, and treat "all qualified data requesters and trial sponsors evenhandedly," requiring both generators and requesters to work according to the same rigorous scientific principles (N. Engl. J. Med. 2013 Oct. 21 [doi: 10.1056/NEJMhle1309073]).
The demand for more data from and about clinical trials – including protocol designs, results summaries, and more recently, raw input data – has grown over the past 15 years. Some medical journals have pushed for disclosure of more data upon request. The Food and Drug Administration increasingly has been requiring disclosure, and pharmaceutical manufacturers in the United States and Europe have made commitments to making more information public. Beginning in March 2014, the European Medicines Agency will require disclosure of some raw data, individual case report forms, and other data.
The authors envision at least four potential models for sharing data. With purely open access, everything would be available for download for free. This is the riskiest model, they said, since it would provide the least accountability.
Another model: The data generator would keep the data but answer very specific requests. A third model would have the clinical trial sponsor review data requests and decide whether and how to release the data.
In the last model, an independent review board would determine whether the data should be released. The board would collect the data from the sponsor and issue it to the requester, on a limited, need-to-know basis.
This model would likely best balance all of the competing needs, according to the authors. The independent board promises "to ensure accountability on the part of data generators and users and allow trial sponsors a voice while precluding them from denying access to data for reasons the public would not consider legitimate," they wrote.
An independent board also would help protect research participants and make sure that the playing field is level among all stakeholders, they added.
The authors noted many benefits to allowing wider access to patient-level data, such as independent analyses of safety and effectiveness, new lines of inquiry that could expose product flaws or trial design flaws, and the potential to answer questions that might affect public health but that weren’t explored in the original study.
The biggest downside is that individual participants’ privacy could be compromised, according to the authors. The risk of exposure "raises critical questions about how to ensure that participants understand the potential ramifications of data sharing," they wrote.
Mandatory disclosure could also discourage investment in research and development if manufacturers believe that the data could be used by competitors. Wider data-sharing could also lead to second-guessing of approvals by regulatory agencies.
Clinical trial data-sharing received attention from the World Medical Association in its most recent update of the Declaration of Helsinki, published online Oct. 19 in JAMA (doi: 10.1001/jama.2013.281053). Three general principles addressed data sharing:
• Number 9: It is the duty of physicians who are involved in medical research to protect the life, health, dignity, integrity, right to self-determination, privacy, and confidentiality of personal information of research subjects. The responsibility for the protection of research subjects must always rest with the physician or other health care professionals and never with the research subjects, even though they have given consent.
• Number 24. Every precaution must be taken to protect the privacy of research subjects and the confidentiality of their personal information.
• Number 32. For medical research using identifiable human material or data, such as research on material or data contained in biobanks or similar repositories, physicians must seek informed consent for its collection, storage and/or reuse. There may be exceptional situations where consent would be impossible or impracticable to obtain for such research. In such situations the research may be done only after consideration and approval of a research ethics committee.
Several of the authors of the paper published in the New England Journal of Medicine disclosed that they work for consulting companies that receive fees from various manufacturers and from academic medical centers. The working group was convened through the Multi-Regional Clinical Trials Center at Harvard University, which receives funds from pharmaceutical companies and not-for-profit entities.
On Twitter @aliciaault
Contrary to industry fears, we argue that access to full – though appropriately deidentified – data sets from clinical trials will benefit the research-based biopharmaceutical industry. We predict that it will help to increase the efficiency of drug development, improve cost-effectiveness, improve comparative-effectiveness analysis, and reduce duplication of effort among trial sponsors.
A managed-release environment that allows sharing of patient-level data while ensuring patient privacy would create a level playing field for all stakeholders. What is sometimes labeled as "free riding" may ultimately pay dividends for innovative companies and for public health. It is ironic that the organizations that most resist wider access to data are the ones that stand to benefit so much from greater transparency.
Dr. Hans-Georg Eichler, Frank Petavy, Dr. Francesco Pignatti, and Dr. Guido Rasi are all with the European Medicines Agency in London. Their remarks are taken from an accompanying editorial(N. Eng. J. Med. 2013 Oct. 21 [doi: 10.1056/NEJMp1310771]).
Contrary to industry fears, we argue that access to full – though appropriately deidentified – data sets from clinical trials will benefit the research-based biopharmaceutical industry. We predict that it will help to increase the efficiency of drug development, improve cost-effectiveness, improve comparative-effectiveness analysis, and reduce duplication of effort among trial sponsors.
A managed-release environment that allows sharing of patient-level data while ensuring patient privacy would create a level playing field for all stakeholders. What is sometimes labeled as "free riding" may ultimately pay dividends for innovative companies and for public health. It is ironic that the organizations that most resist wider access to data are the ones that stand to benefit so much from greater transparency.
Dr. Hans-Georg Eichler, Frank Petavy, Dr. Francesco Pignatti, and Dr. Guido Rasi are all with the European Medicines Agency in London. Their remarks are taken from an accompanying editorial(N. Eng. J. Med. 2013 Oct. 21 [doi: 10.1056/NEJMp1310771]).
Contrary to industry fears, we argue that access to full – though appropriately deidentified – data sets from clinical trials will benefit the research-based biopharmaceutical industry. We predict that it will help to increase the efficiency of drug development, improve cost-effectiveness, improve comparative-effectiveness analysis, and reduce duplication of effort among trial sponsors.
A managed-release environment that allows sharing of patient-level data while ensuring patient privacy would create a level playing field for all stakeholders. What is sometimes labeled as "free riding" may ultimately pay dividends for innovative companies and for public health. It is ironic that the organizations that most resist wider access to data are the ones that stand to benefit so much from greater transparency.
Dr. Hans-Georg Eichler, Frank Petavy, Dr. Francesco Pignatti, and Dr. Guido Rasi are all with the European Medicines Agency in London. Their remarks are taken from an accompanying editorial(N. Eng. J. Med. 2013 Oct. 21 [doi: 10.1056/NEJMp1310771]).
Clinical trial data should be shared as broadly as possible to help spur scientific innovation and answer questions of importance to public health.
"The question is not whether, but how, these data should be broadly shared," wrote Michelle M. Mello and her colleagues from Harvard University, Boston, the Pharmaceutical Research and Manufacturers of America, and several consulting companies, in an article published online Oct. 21 in the New England Journal of Medicine.
At a minimum, data-sharing should be available for trials of all approved prescription drugs, devices, and biologics in any country that has adequate intellectual property protection. A system has to ensure responsible use of data, protect privacy of research participants, and treat "all qualified data requesters and trial sponsors evenhandedly," requiring both generators and requesters to work according to the same rigorous scientific principles (N. Engl. J. Med. 2013 Oct. 21 [doi: 10.1056/NEJMhle1309073]).
The demand for more data from and about clinical trials – including protocol designs, results summaries, and more recently, raw input data – has grown over the past 15 years. Some medical journals have pushed for disclosure of more data upon request. The Food and Drug Administration increasingly has been requiring disclosure, and pharmaceutical manufacturers in the United States and Europe have made commitments to making more information public. Beginning in March 2014, the European Medicines Agency will require disclosure of some raw data, individual case report forms, and other data.
The authors envision at least four potential models for sharing data. With purely open access, everything would be available for download for free. This is the riskiest model, they said, since it would provide the least accountability.
Another model: The data generator would keep the data but answer very specific requests. A third model would have the clinical trial sponsor review data requests and decide whether and how to release the data.
In the last model, an independent review board would determine whether the data should be released. The board would collect the data from the sponsor and issue it to the requester, on a limited, need-to-know basis.
This model would likely best balance all of the competing needs, according to the authors. The independent board promises "to ensure accountability on the part of data generators and users and allow trial sponsors a voice while precluding them from denying access to data for reasons the public would not consider legitimate," they wrote.
An independent board also would help protect research participants and make sure that the playing field is level among all stakeholders, they added.
The authors noted many benefits to allowing wider access to patient-level data, such as independent analyses of safety and effectiveness, new lines of inquiry that could expose product flaws or trial design flaws, and the potential to answer questions that might affect public health but that weren’t explored in the original study.
The biggest downside is that individual participants’ privacy could be compromised, according to the authors. The risk of exposure "raises critical questions about how to ensure that participants understand the potential ramifications of data sharing," they wrote.
Mandatory disclosure could also discourage investment in research and development if manufacturers believe that the data could be used by competitors. Wider data-sharing could also lead to second-guessing of approvals by regulatory agencies.
Clinical trial data-sharing received attention from the World Medical Association in its most recent update of the Declaration of Helsinki, published online Oct. 19 in JAMA (doi: 10.1001/jama.2013.281053). Three general principles addressed data sharing:
• Number 9: It is the duty of physicians who are involved in medical research to protect the life, health, dignity, integrity, right to self-determination, privacy, and confidentiality of personal information of research subjects. The responsibility for the protection of research subjects must always rest with the physician or other health care professionals and never with the research subjects, even though they have given consent.
• Number 24. Every precaution must be taken to protect the privacy of research subjects and the confidentiality of their personal information.
• Number 32. For medical research using identifiable human material or data, such as research on material or data contained in biobanks or similar repositories, physicians must seek informed consent for its collection, storage and/or reuse. There may be exceptional situations where consent would be impossible or impracticable to obtain for such research. In such situations the research may be done only after consideration and approval of a research ethics committee.
Several of the authors of the paper published in the New England Journal of Medicine disclosed that they work for consulting companies that receive fees from various manufacturers and from academic medical centers. The working group was convened through the Multi-Regional Clinical Trials Center at Harvard University, which receives funds from pharmaceutical companies and not-for-profit entities.
On Twitter @aliciaault
Clinical trial data should be shared as broadly as possible to help spur scientific innovation and answer questions of importance to public health.
"The question is not whether, but how, these data should be broadly shared," wrote Michelle M. Mello and her colleagues from Harvard University, Boston, the Pharmaceutical Research and Manufacturers of America, and several consulting companies, in an article published online Oct. 21 in the New England Journal of Medicine.
At a minimum, data-sharing should be available for trials of all approved prescription drugs, devices, and biologics in any country that has adequate intellectual property protection. A system has to ensure responsible use of data, protect privacy of research participants, and treat "all qualified data requesters and trial sponsors evenhandedly," requiring both generators and requesters to work according to the same rigorous scientific principles (N. Engl. J. Med. 2013 Oct. 21 [doi: 10.1056/NEJMhle1309073]).
The demand for more data from and about clinical trials – including protocol designs, results summaries, and more recently, raw input data – has grown over the past 15 years. Some medical journals have pushed for disclosure of more data upon request. The Food and Drug Administration increasingly has been requiring disclosure, and pharmaceutical manufacturers in the United States and Europe have made commitments to making more information public. Beginning in March 2014, the European Medicines Agency will require disclosure of some raw data, individual case report forms, and other data.
The authors envision at least four potential models for sharing data. With purely open access, everything would be available for download for free. This is the riskiest model, they said, since it would provide the least accountability.
Another model: The data generator would keep the data but answer very specific requests. A third model would have the clinical trial sponsor review data requests and decide whether and how to release the data.
In the last model, an independent review board would determine whether the data should be released. The board would collect the data from the sponsor and issue it to the requester, on a limited, need-to-know basis.
This model would likely best balance all of the competing needs, according to the authors. The independent board promises "to ensure accountability on the part of data generators and users and allow trial sponsors a voice while precluding them from denying access to data for reasons the public would not consider legitimate," they wrote.
An independent board also would help protect research participants and make sure that the playing field is level among all stakeholders, they added.
The authors noted many benefits to allowing wider access to patient-level data, such as independent analyses of safety and effectiveness, new lines of inquiry that could expose product flaws or trial design flaws, and the potential to answer questions that might affect public health but that weren’t explored in the original study.
The biggest downside is that individual participants’ privacy could be compromised, according to the authors. The risk of exposure "raises critical questions about how to ensure that participants understand the potential ramifications of data sharing," they wrote.
Mandatory disclosure could also discourage investment in research and development if manufacturers believe that the data could be used by competitors. Wider data-sharing could also lead to second-guessing of approvals by regulatory agencies.
Clinical trial data-sharing received attention from the World Medical Association in its most recent update of the Declaration of Helsinki, published online Oct. 19 in JAMA (doi: 10.1001/jama.2013.281053). Three general principles addressed data sharing:
• Number 9: It is the duty of physicians who are involved in medical research to protect the life, health, dignity, integrity, right to self-determination, privacy, and confidentiality of personal information of research subjects. The responsibility for the protection of research subjects must always rest with the physician or other health care professionals and never with the research subjects, even though they have given consent.
• Number 24. Every precaution must be taken to protect the privacy of research subjects and the confidentiality of their personal information.
• Number 32. For medical research using identifiable human material or data, such as research on material or data contained in biobanks or similar repositories, physicians must seek informed consent for its collection, storage and/or reuse. There may be exceptional situations where consent would be impossible or impracticable to obtain for such research. In such situations the research may be done only after consideration and approval of a research ethics committee.
Several of the authors of the paper published in the New England Journal of Medicine disclosed that they work for consulting companies that receive fees from various manufacturers and from academic medical centers. The working group was convened through the Multi-Regional Clinical Trials Center at Harvard University, which receives funds from pharmaceutical companies and not-for-profit entities.
On Twitter @aliciaault
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Obama addresses health exchange website woes
President Obama acknowledged that ongoing problems with the healthcare.gov website – the health insurance exchange for Americans in most states – were giving consumers pause, making supporters nervous, and helping adversaries discredit the Affordable Care Act.
"There’s no sugarcoating it. The website has been too slow," President Obama said during an Oct. 21 White House speech. He said that consumers were having trouble logging on to the website and have gotten bogged down during the application process.
"Nobody’s more frustrated by that than I am," he said, adding that "I want the cash registers to work; I want the checkout lines to be smooth. So I want people to be able to get this great product."
That product, according to the president, is high-quality health insurance that’s offered at a good price.
He said there has been "massive demand," citing nearly 20 million visitors to healthcare.gov. Thirty-four states are directing their residents to buy coverage through that federal website. The president also said there was a lot of demand for plans offered through the exchanges being run by 16 states and the District of Columbia.
Overall, half a million consumers have submitted applications through the federal and state websites, President Obama said. "People don’t just want it; they’re showing up to buy it," he said.
For consumers who have difficulty in accessing the website, President Obama referred them to a toll-free phone number, 800-318-2596. He also said that people could apply in-person at various locations.
"Nobody’s madder than me about the fact that the website isn’t working as well as it should, which means it’s going to get fixed," he said.
On Oct. 20, Health and Human Services department officials said that some of those fixes were underway. The site has been sluggish because "the initial wave of interest stressed the account service," according to HHS officials. But they said that the "data hub" was working, meaning that consumers would be told if they were eligible for subsidies. They also noted that the agency is bringing in technical help to fix the website – something President Obama repeated in his speech.
But the president also said he was frustrated that there was so much focus on the website, given that it was only 3 weeks into a 6-month enrollment process, and that many of the ACA’s benefits were already in place.
He addressed what he called "some of the politics that have swirled around the Affordable Care Act," noting Republican efforts to defund or repeal the ACA.
"And I’m sure that, given the problems with the website so far, they’re going to be looking to go after it even harder," he said.
But, he said, "We did not wage this long and contentious battle just around a website." The battle was "to make sure that millions of Americans in the wealthiest nation on Earth finally have the same chance to get the same security of affordable quality health care as anybody else."
Speaker of the House John Boehner (R-Ohio) was not impressed. "If the president is frustrated by the mounting failures of his health care law, it wasn’t apparent today," said Rep. Boehner, in a statement. "Instead of answers, we got well-worn talking points. Instead of explanations, we got excuses."
He added that "the House’s oversight of this failure is just beginning."
On Oct. 24, the House Energy and Commerce Committee will hold the first of what is likely to be many hearings on the website rollout. HHS Secretary Kathleen Sebelius was invited to testify, but declined because of a scheduling conflict.
Rep. Boehner said that "Secretary Sebelius must change her mind and appear at this week’s hearing."
On Twitter @aliciaault
*This story was updated 10/22/13.
President Obama acknowledged that ongoing problems with the healthcare.gov website – the health insurance exchange for Americans in most states – were giving consumers pause, making supporters nervous, and helping adversaries discredit the Affordable Care Act.
"There’s no sugarcoating it. The website has been too slow," President Obama said during an Oct. 21 White House speech. He said that consumers were having trouble logging on to the website and have gotten bogged down during the application process.
"Nobody’s more frustrated by that than I am," he said, adding that "I want the cash registers to work; I want the checkout lines to be smooth. So I want people to be able to get this great product."
That product, according to the president, is high-quality health insurance that’s offered at a good price.
He said there has been "massive demand," citing nearly 20 million visitors to healthcare.gov. Thirty-four states are directing their residents to buy coverage through that federal website. The president also said there was a lot of demand for plans offered through the exchanges being run by 16 states and the District of Columbia.
Overall, half a million consumers have submitted applications through the federal and state websites, President Obama said. "People don’t just want it; they’re showing up to buy it," he said.
For consumers who have difficulty in accessing the website, President Obama referred them to a toll-free phone number, 800-318-2596. He also said that people could apply in-person at various locations.
"Nobody’s madder than me about the fact that the website isn’t working as well as it should, which means it’s going to get fixed," he said.
On Oct. 20, Health and Human Services department officials said that some of those fixes were underway. The site has been sluggish because "the initial wave of interest stressed the account service," according to HHS officials. But they said that the "data hub" was working, meaning that consumers would be told if they were eligible for subsidies. They also noted that the agency is bringing in technical help to fix the website – something President Obama repeated in his speech.
But the president also said he was frustrated that there was so much focus on the website, given that it was only 3 weeks into a 6-month enrollment process, and that many of the ACA’s benefits were already in place.
He addressed what he called "some of the politics that have swirled around the Affordable Care Act," noting Republican efforts to defund or repeal the ACA.
"And I’m sure that, given the problems with the website so far, they’re going to be looking to go after it even harder," he said.
But, he said, "We did not wage this long and contentious battle just around a website." The battle was "to make sure that millions of Americans in the wealthiest nation on Earth finally have the same chance to get the same security of affordable quality health care as anybody else."
Speaker of the House John Boehner (R-Ohio) was not impressed. "If the president is frustrated by the mounting failures of his health care law, it wasn’t apparent today," said Rep. Boehner, in a statement. "Instead of answers, we got well-worn talking points. Instead of explanations, we got excuses."
He added that "the House’s oversight of this failure is just beginning."
On Oct. 24, the House Energy and Commerce Committee will hold the first of what is likely to be many hearings on the website rollout. HHS Secretary Kathleen Sebelius was invited to testify, but declined because of a scheduling conflict.
Rep. Boehner said that "Secretary Sebelius must change her mind and appear at this week’s hearing."
On Twitter @aliciaault
*This story was updated 10/22/13.
President Obama acknowledged that ongoing problems with the healthcare.gov website – the health insurance exchange for Americans in most states – were giving consumers pause, making supporters nervous, and helping adversaries discredit the Affordable Care Act.
"There’s no sugarcoating it. The website has been too slow," President Obama said during an Oct. 21 White House speech. He said that consumers were having trouble logging on to the website and have gotten bogged down during the application process.
"Nobody’s more frustrated by that than I am," he said, adding that "I want the cash registers to work; I want the checkout lines to be smooth. So I want people to be able to get this great product."
That product, according to the president, is high-quality health insurance that’s offered at a good price.
He said there has been "massive demand," citing nearly 20 million visitors to healthcare.gov. Thirty-four states are directing their residents to buy coverage through that federal website. The president also said there was a lot of demand for plans offered through the exchanges being run by 16 states and the District of Columbia.
Overall, half a million consumers have submitted applications through the federal and state websites, President Obama said. "People don’t just want it; they’re showing up to buy it," he said.
For consumers who have difficulty in accessing the website, President Obama referred them to a toll-free phone number, 800-318-2596. He also said that people could apply in-person at various locations.
"Nobody’s madder than me about the fact that the website isn’t working as well as it should, which means it’s going to get fixed," he said.
On Oct. 20, Health and Human Services department officials said that some of those fixes were underway. The site has been sluggish because "the initial wave of interest stressed the account service," according to HHS officials. But they said that the "data hub" was working, meaning that consumers would be told if they were eligible for subsidies. They also noted that the agency is bringing in technical help to fix the website – something President Obama repeated in his speech.
But the president also said he was frustrated that there was so much focus on the website, given that it was only 3 weeks into a 6-month enrollment process, and that many of the ACA’s benefits were already in place.
He addressed what he called "some of the politics that have swirled around the Affordable Care Act," noting Republican efforts to defund or repeal the ACA.
"And I’m sure that, given the problems with the website so far, they’re going to be looking to go after it even harder," he said.
But, he said, "We did not wage this long and contentious battle just around a website." The battle was "to make sure that millions of Americans in the wealthiest nation on Earth finally have the same chance to get the same security of affordable quality health care as anybody else."
Speaker of the House John Boehner (R-Ohio) was not impressed. "If the president is frustrated by the mounting failures of his health care law, it wasn’t apparent today," said Rep. Boehner, in a statement. "Instead of answers, we got well-worn talking points. Instead of explanations, we got excuses."
He added that "the House’s oversight of this failure is just beginning."
On Oct. 24, the House Energy and Commerce Committee will hold the first of what is likely to be many hearings on the website rollout. HHS Secretary Kathleen Sebelius was invited to testify, but declined because of a scheduling conflict.
Rep. Boehner said that "Secretary Sebelius must change her mind and appear at this week’s hearing."
On Twitter @aliciaault
*This story was updated 10/22/13.
Congress approves deal to open government, leaves health reform intact
Congress has approved a deal to fully reopen the federal government and keep the nation from defaulting on its debt. The agreement does not delay or defund the Affordable Care Act.
On Oct. 16 – 16 days into the government shutdown and about a day before the United States was set to breach its debt ceiling – the Senate voted 81-18 to approve a bipartisan compromise brokered by Senate Majority Leader Harry Reid (D-Nev.) and Minority Leader Mitch McConnell (R-Ky.). Later that night, the House approved the bill 285-144. The president signed the legislation in the early hours of Oct. 17.
The law funds the federal government at its current levels until Jan. 15, 2014, keeping the sequestration cuts in effect. It also allows the government to continue to borrow money to meet its financial obligations until Feb. 7, 2014. In the meantime, lawmakers have agreed to convene a budget conference to work out a long-range budget plan by mid-December.
Although the bill does not fundamentally change the Affordable Care Act (ACA), it does require the Department of Health and Human Services to beef up its income verification for individuals seeking subsidies to purchase insurance on the exchanges.
Last July, the agency announced that it would not verify the income of every applicant who applied for federal subsidies through the exchanges. Instead, federal officials planned to audit a sample of applications to ensure that individuals were getting the appropriate federal insurance subsidies. GOP lawmakers objected to this approach, saying that relying too heavily on the honor system in determining federal subsidies would leave the government open to fraud.
Under the new law, HHS must submit a report to Congress on its verification procedures by Jan. 1, 2014, and the HHS Inspector General must submit a progress report by July 2014 on the effectiveness of the new procedures.
Although the new law doesn’t do anything to hamper the implementation of the ACA, opponents have not given up. Sen. McConnell, who negotiated the deal with Democrats, said that Republicans will keep trying to repeal the ACA.
"This law is ravaging our economy, killing jobs, driving up premiums, and driving people off the health care plans they have and like in droves," he said on the Senate floor. "Its disastrous rollout is a sign of even worse things to come. And the Democrat refusal to delay it reflects a stubborn ideological obsession that will do untold damage to our country."
mschneider@frontlinemedcom.com
On Twitter @MaryEllenNY
Congress has approved a deal to fully reopen the federal government and keep the nation from defaulting on its debt. The agreement does not delay or defund the Affordable Care Act.
On Oct. 16 – 16 days into the government shutdown and about a day before the United States was set to breach its debt ceiling – the Senate voted 81-18 to approve a bipartisan compromise brokered by Senate Majority Leader Harry Reid (D-Nev.) and Minority Leader Mitch McConnell (R-Ky.). Later that night, the House approved the bill 285-144. The president signed the legislation in the early hours of Oct. 17.
The law funds the federal government at its current levels until Jan. 15, 2014, keeping the sequestration cuts in effect. It also allows the government to continue to borrow money to meet its financial obligations until Feb. 7, 2014. In the meantime, lawmakers have agreed to convene a budget conference to work out a long-range budget plan by mid-December.
Although the bill does not fundamentally change the Affordable Care Act (ACA), it does require the Department of Health and Human Services to beef up its income verification for individuals seeking subsidies to purchase insurance on the exchanges.
Last July, the agency announced that it would not verify the income of every applicant who applied for federal subsidies through the exchanges. Instead, federal officials planned to audit a sample of applications to ensure that individuals were getting the appropriate federal insurance subsidies. GOP lawmakers objected to this approach, saying that relying too heavily on the honor system in determining federal subsidies would leave the government open to fraud.
Under the new law, HHS must submit a report to Congress on its verification procedures by Jan. 1, 2014, and the HHS Inspector General must submit a progress report by July 2014 on the effectiveness of the new procedures.
Although the new law doesn’t do anything to hamper the implementation of the ACA, opponents have not given up. Sen. McConnell, who negotiated the deal with Democrats, said that Republicans will keep trying to repeal the ACA.
"This law is ravaging our economy, killing jobs, driving up premiums, and driving people off the health care plans they have and like in droves," he said on the Senate floor. "Its disastrous rollout is a sign of even worse things to come. And the Democrat refusal to delay it reflects a stubborn ideological obsession that will do untold damage to our country."
mschneider@frontlinemedcom.com
On Twitter @MaryEllenNY
Congress has approved a deal to fully reopen the federal government and keep the nation from defaulting on its debt. The agreement does not delay or defund the Affordable Care Act.
On Oct. 16 – 16 days into the government shutdown and about a day before the United States was set to breach its debt ceiling – the Senate voted 81-18 to approve a bipartisan compromise brokered by Senate Majority Leader Harry Reid (D-Nev.) and Minority Leader Mitch McConnell (R-Ky.). Later that night, the House approved the bill 285-144. The president signed the legislation in the early hours of Oct. 17.
The law funds the federal government at its current levels until Jan. 15, 2014, keeping the sequestration cuts in effect. It also allows the government to continue to borrow money to meet its financial obligations until Feb. 7, 2014. In the meantime, lawmakers have agreed to convene a budget conference to work out a long-range budget plan by mid-December.
Although the bill does not fundamentally change the Affordable Care Act (ACA), it does require the Department of Health and Human Services to beef up its income verification for individuals seeking subsidies to purchase insurance on the exchanges.
Last July, the agency announced that it would not verify the income of every applicant who applied for federal subsidies through the exchanges. Instead, federal officials planned to audit a sample of applications to ensure that individuals were getting the appropriate federal insurance subsidies. GOP lawmakers objected to this approach, saying that relying too heavily on the honor system in determining federal subsidies would leave the government open to fraud.
Under the new law, HHS must submit a report to Congress on its verification procedures by Jan. 1, 2014, and the HHS Inspector General must submit a progress report by July 2014 on the effectiveness of the new procedures.
Although the new law doesn’t do anything to hamper the implementation of the ACA, opponents have not given up. Sen. McConnell, who negotiated the deal with Democrats, said that Republicans will keep trying to repeal the ACA.
"This law is ravaging our economy, killing jobs, driving up premiums, and driving people off the health care plans they have and like in droves," he said on the Senate floor. "Its disastrous rollout is a sign of even worse things to come. And the Democrat refusal to delay it reflects a stubborn ideological obsession that will do untold damage to our country."
mschneider@frontlinemedcom.com
On Twitter @MaryEllenNY