User login
Vaccine exemptions more common in western states
For the 2016-2017 school year, 2% of American kindergarten students had an exemption from one or more vaccines, said Ranee Seither and associates at the National Center of Immunization and Respiratory Disease, Centers for Disease Control and Prevention, Atlanta.
Among the 46 states – including the District of Columbia – that reported data, nine times as many exemptions were granted on religious or philosophical grounds (1.8%) as were granted for medical reasons (0.2%), they said.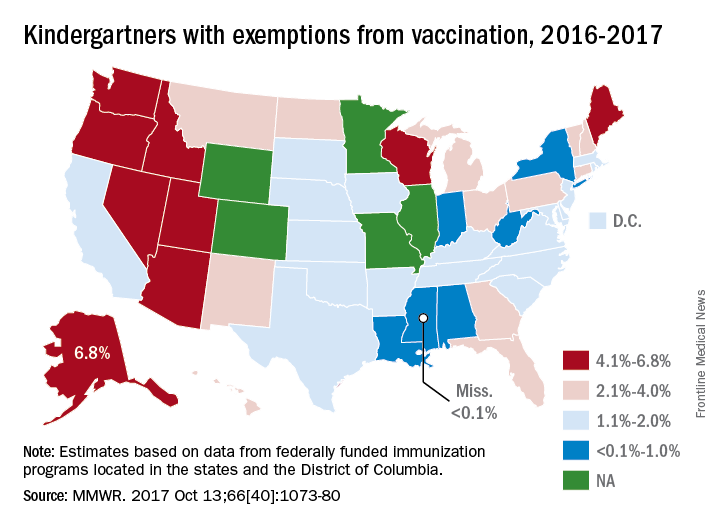
Alaska had the highest rate of medical exemptions at 1.5% and Oregon had the highest rate of religious/philosophical exemptions at 6.5%. Thirty states do not allow philosophical exemptions, Arizona and Mississippi do not allow religious exemptions, and West Virginia does not allow either, they noted.
Exemption data were reported for 3,666,870 kindergartners for the 2016-2017 school year and collected by federally funded immunization programs in the 50 states and D.C.
For the 2016-2017 school year, 2% of American kindergarten students had an exemption from one or more vaccines, said Ranee Seither and associates at the National Center of Immunization and Respiratory Disease, Centers for Disease Control and Prevention, Atlanta.
Among the 46 states – including the District of Columbia – that reported data, nine times as many exemptions were granted on religious or philosophical grounds (1.8%) as were granted for medical reasons (0.2%), they said.
Alaska had the highest rate of medical exemptions at 1.5% and Oregon had the highest rate of religious/philosophical exemptions at 6.5%. Thirty states do not allow philosophical exemptions, Arizona and Mississippi do not allow religious exemptions, and West Virginia does not allow either, they noted.
Exemption data were reported for 3,666,870 kindergartners for the 2016-2017 school year and collected by federally funded immunization programs in the 50 states and D.C.
For the 2016-2017 school year, 2% of American kindergarten students had an exemption from one or more vaccines, said Ranee Seither and associates at the National Center of Immunization and Respiratory Disease, Centers for Disease Control and Prevention, Atlanta.
Among the 46 states – including the District of Columbia – that reported data, nine times as many exemptions were granted on religious or philosophical grounds (1.8%) as were granted for medical reasons (0.2%), they said.
Alaska had the highest rate of medical exemptions at 1.5% and Oregon had the highest rate of religious/philosophical exemptions at 6.5%. Thirty states do not allow philosophical exemptions, Arizona and Mississippi do not allow religious exemptions, and West Virginia does not allow either, they noted.
Exemption data were reported for 3,666,870 kindergartners for the 2016-2017 school year and collected by federally funded immunization programs in the 50 states and D.C.
FROM MMWR
Health disparities in rural America: Chronic conditions
Among rural adults, multiple chronic health conditions are most common in non-Hispanic blacks and American Indians/Alaska Natives (AI/ANs) and least common among Asians and Native Hawaiians/other Pacific Islanders (NHOPIs), according to the Centers for Disease Control and Prevention.
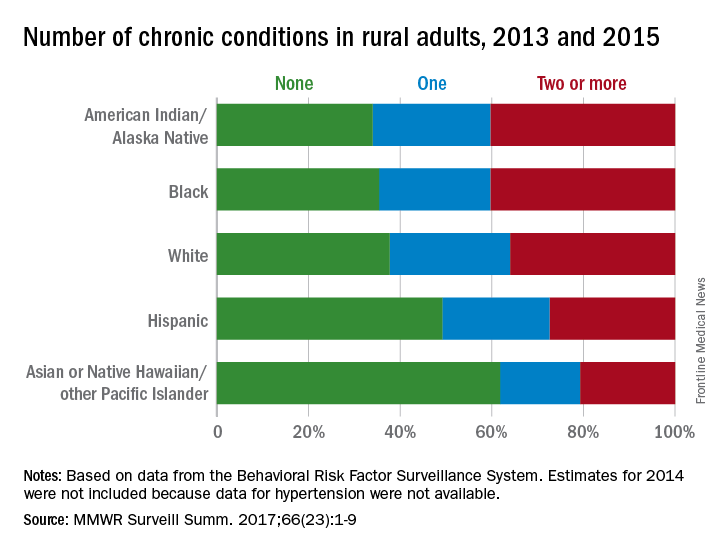
The order was reversed for adults reporting no chronic conditions: Asians and NHOPIs at 61.8%, Hispanics at 49.2%, whites at 37.8%, blacks at 35.4%, and AI/ANs at 34.0%, the researchers said.
For the chronic health conditions included separately in the report, blacks had the highest rate (45.9%) and Asians and NHOPIs had the lowest rate (15.5%) of obesity; AI/ANs were most likely (23.2%) and Asians and NHOPIs were least likely (5.8%) to report depressive disorder. Other conditions considered in the estimates were myocardial infarction; coronary heart disease; stroke; hypertension; asthma; skin cancer; other types of cancer; chronic obstructive pulmonary disease; kidney disease; some form of arthritis, rheumatoid arthritis, gout, lupus, or fibromyalgia; and diabetes. Estimates for 2014 were not included because data for hypertension were not available, the investigators noted.
Of the 3,143 counties categorized by the National Center for Health Statistics’ Urban-Rural Classification Scheme for Counties, a total of 1,325 were considered rural and included 6.1% of the U.S. population, they said.
Among rural adults, multiple chronic health conditions are most common in non-Hispanic blacks and American Indians/Alaska Natives (AI/ANs) and least common among Asians and Native Hawaiians/other Pacific Islanders (NHOPIs), according to the Centers for Disease Control and Prevention.

The order was reversed for adults reporting no chronic conditions: Asians and NHOPIs at 61.8%, Hispanics at 49.2%, whites at 37.8%, blacks at 35.4%, and AI/ANs at 34.0%, the researchers said.
For the chronic health conditions included separately in the report, blacks had the highest rate (45.9%) and Asians and NHOPIs had the lowest rate (15.5%) of obesity; AI/ANs were most likely (23.2%) and Asians and NHOPIs were least likely (5.8%) to report depressive disorder. Other conditions considered in the estimates were myocardial infarction; coronary heart disease; stroke; hypertension; asthma; skin cancer; other types of cancer; chronic obstructive pulmonary disease; kidney disease; some form of arthritis, rheumatoid arthritis, gout, lupus, or fibromyalgia; and diabetes. Estimates for 2014 were not included because data for hypertension were not available, the investigators noted.
Of the 3,143 counties categorized by the National Center for Health Statistics’ Urban-Rural Classification Scheme for Counties, a total of 1,325 were considered rural and included 6.1% of the U.S. population, they said.
Among rural adults, multiple chronic health conditions are most common in non-Hispanic blacks and American Indians/Alaska Natives (AI/ANs) and least common among Asians and Native Hawaiians/other Pacific Islanders (NHOPIs), according to the Centers for Disease Control and Prevention.

The order was reversed for adults reporting no chronic conditions: Asians and NHOPIs at 61.8%, Hispanics at 49.2%, whites at 37.8%, blacks at 35.4%, and AI/ANs at 34.0%, the researchers said.
For the chronic health conditions included separately in the report, blacks had the highest rate (45.9%) and Asians and NHOPIs had the lowest rate (15.5%) of obesity; AI/ANs were most likely (23.2%) and Asians and NHOPIs were least likely (5.8%) to report depressive disorder. Other conditions considered in the estimates were myocardial infarction; coronary heart disease; stroke; hypertension; asthma; skin cancer; other types of cancer; chronic obstructive pulmonary disease; kidney disease; some form of arthritis, rheumatoid arthritis, gout, lupus, or fibromyalgia; and diabetes. Estimates for 2014 were not included because data for hypertension were not available, the investigators noted.
Of the 3,143 counties categorized by the National Center for Health Statistics’ Urban-Rural Classification Scheme for Counties, a total of 1,325 were considered rural and included 6.1% of the U.S. population, they said.
FROM MMWR SURVEILLANCE SUMMARIES
FDA approves first trastuzumab biosimilar
The Food and Drug Administration has approved trastuzumab-dkst (Ogivri) as a biosimilar to trastuzumab (Herceptin) for the treatment of patients with HER2+ breast or metastatic gastric or gastroesophageal junction adenocarcinoma.
This is the first biosimilar approved in the United States for the treatment of breast cancer or gastric cancer and the second biosimilar approved for the treatment of cancer, the FDA said in a statement.
The approval of trastuzumab-dkst is based on structural and functional characterization, animal study data, human pharmacokinetic and pharmacodynamic data, clinical immunogenicity data, and other clinical safety and effectiveness data.
Common expected side effects of trastuzumab-dkst for the treatment of HER2+ breast cancer include headache, diarrhea, nausea, chills, fever, infection, congestive heart failure, insomnia, cough, and rash. Common expected side effects for the treatment of HER2+ metastatic gastric cancer include neutropenia, diarrhea, fatigue, anemia, stomatitis, weight loss, upper respiratory tract infections, fever, thrombocytopenia, mucosal inflammation, nasopharyngitis, and dysgeusia.
The biosimilar label contains a Boxed Warning – as trastuzumab does – about increased risks of cardiomyopathy, infusion reactions, pulmonary toxicity, and fetal toxicity.
The FDA’s Oncologic Drugs Advisory Committee voted unanimously in July to recommend approval of the biosimilar, made by Mylan and Biocon.
To prepare for the entry of biosimilars to the market, AGA is taking the lead in educating health-care professionals and patients about biosimilars. Visit www.gastro.org/biosimilars to learn more.
The Food and Drug Administration has approved trastuzumab-dkst (Ogivri) as a biosimilar to trastuzumab (Herceptin) for the treatment of patients with HER2+ breast or metastatic gastric or gastroesophageal junction adenocarcinoma.
This is the first biosimilar approved in the United States for the treatment of breast cancer or gastric cancer and the second biosimilar approved for the treatment of cancer, the FDA said in a statement.
The approval of trastuzumab-dkst is based on structural and functional characterization, animal study data, human pharmacokinetic and pharmacodynamic data, clinical immunogenicity data, and other clinical safety and effectiveness data.
Common expected side effects of trastuzumab-dkst for the treatment of HER2+ breast cancer include headache, diarrhea, nausea, chills, fever, infection, congestive heart failure, insomnia, cough, and rash. Common expected side effects for the treatment of HER2+ metastatic gastric cancer include neutropenia, diarrhea, fatigue, anemia, stomatitis, weight loss, upper respiratory tract infections, fever, thrombocytopenia, mucosal inflammation, nasopharyngitis, and dysgeusia.
The biosimilar label contains a Boxed Warning – as trastuzumab does – about increased risks of cardiomyopathy, infusion reactions, pulmonary toxicity, and fetal toxicity.
The FDA’s Oncologic Drugs Advisory Committee voted unanimously in July to recommend approval of the biosimilar, made by Mylan and Biocon.
To prepare for the entry of biosimilars to the market, AGA is taking the lead in educating health-care professionals and patients about biosimilars. Visit www.gastro.org/biosimilars to learn more.
The Food and Drug Administration has approved trastuzumab-dkst (Ogivri) as a biosimilar to trastuzumab (Herceptin) for the treatment of patients with HER2+ breast or metastatic gastric or gastroesophageal junction adenocarcinoma.
This is the first biosimilar approved in the United States for the treatment of breast cancer or gastric cancer and the second biosimilar approved for the treatment of cancer, the FDA said in a statement.
The approval of trastuzumab-dkst is based on structural and functional characterization, animal study data, human pharmacokinetic and pharmacodynamic data, clinical immunogenicity data, and other clinical safety and effectiveness data.
Common expected side effects of trastuzumab-dkst for the treatment of HER2+ breast cancer include headache, diarrhea, nausea, chills, fever, infection, congestive heart failure, insomnia, cough, and rash. Common expected side effects for the treatment of HER2+ metastatic gastric cancer include neutropenia, diarrhea, fatigue, anemia, stomatitis, weight loss, upper respiratory tract infections, fever, thrombocytopenia, mucosal inflammation, nasopharyngitis, and dysgeusia.
The biosimilar label contains a Boxed Warning – as trastuzumab does – about increased risks of cardiomyopathy, infusion reactions, pulmonary toxicity, and fetal toxicity.
The FDA’s Oncologic Drugs Advisory Committee voted unanimously in July to recommend approval of the biosimilar, made by Mylan and Biocon.
To prepare for the entry of biosimilars to the market, AGA is taking the lead in educating health-care professionals and patients about biosimilars. Visit www.gastro.org/biosimilars to learn more.
New buprenorphine formulation approved for medication-assisted treatment
The Food and Drug Administration has approved an extended-release, subcutaneous injection formulation of buprenorphine for use in treating moderate to severe opioid use disorder (OUD), the manufacturer of the drug announced Nov. 30.
The new product, called Sublocade, is a monthly injection intended for use in patients who have already begun treatment of OUD with transmucosal buprenorphine products, followed by a dose adjustment for a minimum of 7 days. Sublocade contains the partial mu-opioid agonist buprenorphine. By administering a consistent level of buprenorphine into the body, it ensures that levels of buprenorphine are delivered to the mu-opioid receptors, diminishing the effects of opioids, including the euphoric sensations associated with opioid use. During the clinical trial program, buprenorphine plasma concentrations of 2-3 ng/mL were found to bind to greater than 70% of mu-opioid receptors.
According to a statement from the FDA, Sublocade will be distributed only to health care providers as part of a Risk Evaluation and Mitigation Strategy to ensure that the product is not distributed directly to patients. Sublocade should be administered only by a health care professional. Self-injection of Sublocade into the blood stream instead of subcutaneously could lead to occlusion of blood vessels and embolism, according to one of the drug’s boxed warnings. It also should be used as part of a complete treatment program that includes counseling and psychosocial support.
The FDA is also requiring the manufacturer to conduct postmarketing studies to assess which patients would benefit from a higher dosing regimen, to determine whether Sublocade can be safely initiated without a dose stabilization period of sublingual buprenorphine, to assess the feasibility of administering Sublocade at a longer inter-dose interval than once monthly, and to determine a process for transitioning patients with long-term stability on a transmucosal buprenorphine dose to a monthly dose of Sublocade without the use of a higher dose for the first 2 months of treatment.
At recent joint meetings of the FDA’s Psychopharmacologic Drugs and Drug Safety and Risk Management advisory committees, panelists voted on Oct. 31 to recommend approval of Sublocade and on Nov. 1 for another subcutaneous buprenorphine injection formulation. These actions have not gone unnoticed by the American Medical Association.
“The AMA enthusiastically supports Food and Drug Administration Commissioner Scott Gottlieb’s efforts to advance policies and actions to treat those suffering from an opioid use disorder,” Patrice Harris, MD, immediate past chair of the American Medical Association Board of Trustees and a member of the AMA Opioid Task Force, said in a statement. “We also second his bold acknowledgment that criminal justice systems should offer [medication-assisted treatment] to those being detained. As he points out, ‘At the very moment when the criminal justice system could be dramatically lowering the risk of overdose, it is creating the conditions of reduced tolerance to opioids that substantially raises the risk of death upon release.’ With his clear explanation of the problem and solution, this situation can be remedied.”
The Food and Drug Administration has approved an extended-release, subcutaneous injection formulation of buprenorphine for use in treating moderate to severe opioid use disorder (OUD), the manufacturer of the drug announced Nov. 30.
The new product, called Sublocade, is a monthly injection intended for use in patients who have already begun treatment of OUD with transmucosal buprenorphine products, followed by a dose adjustment for a minimum of 7 days. Sublocade contains the partial mu-opioid agonist buprenorphine. By administering a consistent level of buprenorphine into the body, it ensures that levels of buprenorphine are delivered to the mu-opioid receptors, diminishing the effects of opioids, including the euphoric sensations associated with opioid use. During the clinical trial program, buprenorphine plasma concentrations of 2-3 ng/mL were found to bind to greater than 70% of mu-opioid receptors.
According to a statement from the FDA, Sublocade will be distributed only to health care providers as part of a Risk Evaluation and Mitigation Strategy to ensure that the product is not distributed directly to patients. Sublocade should be administered only by a health care professional. Self-injection of Sublocade into the blood stream instead of subcutaneously could lead to occlusion of blood vessels and embolism, according to one of the drug’s boxed warnings. It also should be used as part of a complete treatment program that includes counseling and psychosocial support.
The FDA is also requiring the manufacturer to conduct postmarketing studies to assess which patients would benefit from a higher dosing regimen, to determine whether Sublocade can be safely initiated without a dose stabilization period of sublingual buprenorphine, to assess the feasibility of administering Sublocade at a longer inter-dose interval than once monthly, and to determine a process for transitioning patients with long-term stability on a transmucosal buprenorphine dose to a monthly dose of Sublocade without the use of a higher dose for the first 2 months of treatment.
At recent joint meetings of the FDA’s Psychopharmacologic Drugs and Drug Safety and Risk Management advisory committees, panelists voted on Oct. 31 to recommend approval of Sublocade and on Nov. 1 for another subcutaneous buprenorphine injection formulation. These actions have not gone unnoticed by the American Medical Association.
“The AMA enthusiastically supports Food and Drug Administration Commissioner Scott Gottlieb’s efforts to advance policies and actions to treat those suffering from an opioid use disorder,” Patrice Harris, MD, immediate past chair of the American Medical Association Board of Trustees and a member of the AMA Opioid Task Force, said in a statement. “We also second his bold acknowledgment that criminal justice systems should offer [medication-assisted treatment] to those being detained. As he points out, ‘At the very moment when the criminal justice system could be dramatically lowering the risk of overdose, it is creating the conditions of reduced tolerance to opioids that substantially raises the risk of death upon release.’ With his clear explanation of the problem and solution, this situation can be remedied.”
The Food and Drug Administration has approved an extended-release, subcutaneous injection formulation of buprenorphine for use in treating moderate to severe opioid use disorder (OUD), the manufacturer of the drug announced Nov. 30.
The new product, called Sublocade, is a monthly injection intended for use in patients who have already begun treatment of OUD with transmucosal buprenorphine products, followed by a dose adjustment for a minimum of 7 days. Sublocade contains the partial mu-opioid agonist buprenorphine. By administering a consistent level of buprenorphine into the body, it ensures that levels of buprenorphine are delivered to the mu-opioid receptors, diminishing the effects of opioids, including the euphoric sensations associated with opioid use. During the clinical trial program, buprenorphine plasma concentrations of 2-3 ng/mL were found to bind to greater than 70% of mu-opioid receptors.
According to a statement from the FDA, Sublocade will be distributed only to health care providers as part of a Risk Evaluation and Mitigation Strategy to ensure that the product is not distributed directly to patients. Sublocade should be administered only by a health care professional. Self-injection of Sublocade into the blood stream instead of subcutaneously could lead to occlusion of blood vessels and embolism, according to one of the drug’s boxed warnings. It also should be used as part of a complete treatment program that includes counseling and psychosocial support.
The FDA is also requiring the manufacturer to conduct postmarketing studies to assess which patients would benefit from a higher dosing regimen, to determine whether Sublocade can be safely initiated without a dose stabilization period of sublingual buprenorphine, to assess the feasibility of administering Sublocade at a longer inter-dose interval than once monthly, and to determine a process for transitioning patients with long-term stability on a transmucosal buprenorphine dose to a monthly dose of Sublocade without the use of a higher dose for the first 2 months of treatment.
At recent joint meetings of the FDA’s Psychopharmacologic Drugs and Drug Safety and Risk Management advisory committees, panelists voted on Oct. 31 to recommend approval of Sublocade and on Nov. 1 for another subcutaneous buprenorphine injection formulation. These actions have not gone unnoticed by the American Medical Association.
“The AMA enthusiastically supports Food and Drug Administration Commissioner Scott Gottlieb’s efforts to advance policies and actions to treat those suffering from an opioid use disorder,” Patrice Harris, MD, immediate past chair of the American Medical Association Board of Trustees and a member of the AMA Opioid Task Force, said in a statement. “We also second his bold acknowledgment that criminal justice systems should offer [medication-assisted treatment] to those being detained. As he points out, ‘At the very moment when the criminal justice system could be dramatically lowering the risk of overdose, it is creating the conditions of reduced tolerance to opioids that substantially raises the risk of death upon release.’ With his clear explanation of the problem and solution, this situation can be remedied.”
Mississippi has highest varicella vaccination rate
Coverage for two doses of varicella vaccine among kindergarten students was highest in Mississippi and lowest in the District of Columbia, said Ranee Seither and associates at the National Center of Immunization and Respiratory Disease at the Centers for Disease Control and Prevention, Atlanta.
For the 2016-2017 school year, 99.4% of Mississippi children enrolled in kindergarten received the state-required two doses of varicella vaccine, compared with 84.6% in D.C. The median was 93.8% for the 42 states that require two doses and 96.5% for those 42 plus the 7 states that reported and only require one dose. Oklahoma and Wyoming “did not report data because of widespread problems with the quality of data reported by schools,” the CDC investigators wrote (MMWR 2017;66[40]:1073-80).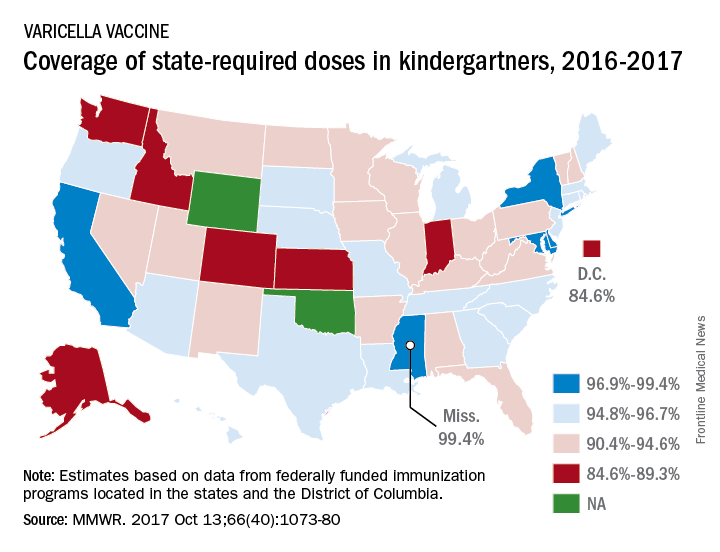
The data for the CDC analysis, which included 3,973,172 kindergartners for the 2016-2017 school year, were collected by federally funded immunization programs in the 50 states and D.C.
Coverage for two doses of varicella vaccine among kindergarten students was highest in Mississippi and lowest in the District of Columbia, said Ranee Seither and associates at the National Center of Immunization and Respiratory Disease at the Centers for Disease Control and Prevention, Atlanta.
For the 2016-2017 school year, 99.4% of Mississippi children enrolled in kindergarten received the state-required two doses of varicella vaccine, compared with 84.6% in D.C. The median was 93.8% for the 42 states that require two doses and 96.5% for those 42 plus the 7 states that reported and only require one dose. Oklahoma and Wyoming “did not report data because of widespread problems with the quality of data reported by schools,” the CDC investigators wrote (MMWR 2017;66[40]:1073-80).
The data for the CDC analysis, which included 3,973,172 kindergartners for the 2016-2017 school year, were collected by federally funded immunization programs in the 50 states and D.C.
Coverage for two doses of varicella vaccine among kindergarten students was highest in Mississippi and lowest in the District of Columbia, said Ranee Seither and associates at the National Center of Immunization and Respiratory Disease at the Centers for Disease Control and Prevention, Atlanta.
For the 2016-2017 school year, 99.4% of Mississippi children enrolled in kindergarten received the state-required two doses of varicella vaccine, compared with 84.6% in D.C. The median was 93.8% for the 42 states that require two doses and 96.5% for those 42 plus the 7 states that reported and only require one dose. Oklahoma and Wyoming “did not report data because of widespread problems with the quality of data reported by schools,” the CDC investigators wrote (MMWR 2017;66[40]:1073-80).
The data for the CDC analysis, which included 3,973,172 kindergartners for the 2016-2017 school year, were collected by federally funded immunization programs in the 50 states and D.C.
FROM MMWR
Two-thirds of abortions occur by 8 weeks’ gestation
, although there was variation by maternal age and race/ethnicity, according to the Centers for Disease Control and Prevention.
That year, 65.3% of abortions were performed at a gestational age of 8 weeks or earlier, with 25.6% occurring at 9-13 weeks. Gestational distribution of the remaining abortions was fairly even: 3.4% at 14-15 weeks, 2.2% at 16-17 weeks, 2.0% at 18-20 weeks, and 1.4% at 21 weeks or later, the CDC investigators reported (MMWR Surveill Summ. 2017 Nov 25;66[24]:1-48).
The percentage of abortions occurring at 8 weeks or earlier was lowest for the youngest age group and increased along with maternal age: 43% for those under 15 years of age and progressing up to 72.5% for women over age 40. That scenario was basically reversed for all of the other gestational periods, as the under-15 group had the highest percentage for 9-13 weeks (34.4%), 14-15 (6.7%), 16-17 (3.6%), 18-20 (5.0%), and 21 weeks and later (7.3%). Those over age 40 had the lowest or almost the lowest percentage in each period, reported Tara C. Jatlaoui, MD, and her associates.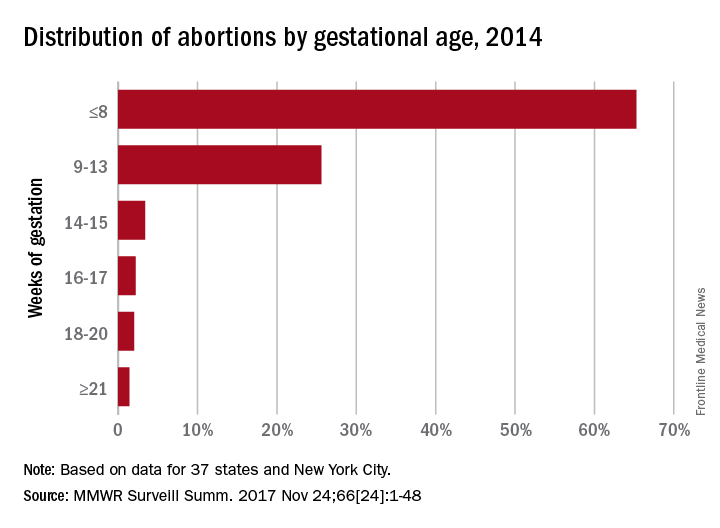
The data for gestational period analysis came from 37 states and New York City. New York State, along with 12 other states, did not report, did not report by gestational age, or did not meet reporting standards.
, although there was variation by maternal age and race/ethnicity, according to the Centers for Disease Control and Prevention.
That year, 65.3% of abortions were performed at a gestational age of 8 weeks or earlier, with 25.6% occurring at 9-13 weeks. Gestational distribution of the remaining abortions was fairly even: 3.4% at 14-15 weeks, 2.2% at 16-17 weeks, 2.0% at 18-20 weeks, and 1.4% at 21 weeks or later, the CDC investigators reported (MMWR Surveill Summ. 2017 Nov 25;66[24]:1-48).
The percentage of abortions occurring at 8 weeks or earlier was lowest for the youngest age group and increased along with maternal age: 43% for those under 15 years of age and progressing up to 72.5% for women over age 40. That scenario was basically reversed for all of the other gestational periods, as the under-15 group had the highest percentage for 9-13 weeks (34.4%), 14-15 (6.7%), 16-17 (3.6%), 18-20 (5.0%), and 21 weeks and later (7.3%). Those over age 40 had the lowest or almost the lowest percentage in each period, reported Tara C. Jatlaoui, MD, and her associates.
The data for gestational period analysis came from 37 states and New York City. New York State, along with 12 other states, did not report, did not report by gestational age, or did not meet reporting standards.
, although there was variation by maternal age and race/ethnicity, according to the Centers for Disease Control and Prevention.
That year, 65.3% of abortions were performed at a gestational age of 8 weeks or earlier, with 25.6% occurring at 9-13 weeks. Gestational distribution of the remaining abortions was fairly even: 3.4% at 14-15 weeks, 2.2% at 16-17 weeks, 2.0% at 18-20 weeks, and 1.4% at 21 weeks or later, the CDC investigators reported (MMWR Surveill Summ. 2017 Nov 25;66[24]:1-48).
The percentage of abortions occurring at 8 weeks or earlier was lowest for the youngest age group and increased along with maternal age: 43% for those under 15 years of age and progressing up to 72.5% for women over age 40. That scenario was basically reversed for all of the other gestational periods, as the under-15 group had the highest percentage for 9-13 weeks (34.4%), 14-15 (6.7%), 16-17 (3.6%), 18-20 (5.0%), and 21 weeks and later (7.3%). Those over age 40 had the lowest or almost the lowest percentage in each period, reported Tara C. Jatlaoui, MD, and her associates.
The data for gestational period analysis came from 37 states and New York City. New York State, along with 12 other states, did not report, did not report by gestational age, or did not meet reporting standards.
FROM MMWR SURVEILLANCE SUMMARIES
FDA grants orphan drug status to rofecoxib for hemophilic arthropathy
The Food and Drug Administration on Nov. 21 granted orphan drug designation to rofecoxib (TRM-201), a cyclooxygenase 2–selective nonsteroidal anti-inflammatory drug (NSAID) intended to treat patients with hemophilic arthropathy (HA).
HA, a joint disease caused by hemarthrosis, is the largest cause of morbidity for hemophilia patients. There are currently no approved treatments in the United States.
The attempt at a reintroduction of rofecoxib specifically for the treatment of HA is being developed by Tremeau Pharmaceuticals.
Patients with hemophilia look to avoid traditional NSAIDs, as those drugs risk gastrointestinal ulcers and impair platelet aggregation. The current standard of care for HA is opioid treatment.
Rofecoxib and other NSAIDs cause an increased risk of serious cardiovascular thrombotic events and gastrointestinal adverse events.
Orphan drug status is available to treatments for rare disorders and provides a 7-year marketing exclusivity period against competition, along with tax credits and a waiver of Prescription Drug User Fee Act filing fees.
The Food and Drug Administration on Nov. 21 granted orphan drug designation to rofecoxib (TRM-201), a cyclooxygenase 2–selective nonsteroidal anti-inflammatory drug (NSAID) intended to treat patients with hemophilic arthropathy (HA).
HA, a joint disease caused by hemarthrosis, is the largest cause of morbidity for hemophilia patients. There are currently no approved treatments in the United States.
The attempt at a reintroduction of rofecoxib specifically for the treatment of HA is being developed by Tremeau Pharmaceuticals.
Patients with hemophilia look to avoid traditional NSAIDs, as those drugs risk gastrointestinal ulcers and impair platelet aggregation. The current standard of care for HA is opioid treatment.
Rofecoxib and other NSAIDs cause an increased risk of serious cardiovascular thrombotic events and gastrointestinal adverse events.
Orphan drug status is available to treatments for rare disorders and provides a 7-year marketing exclusivity period against competition, along with tax credits and a waiver of Prescription Drug User Fee Act filing fees.
The Food and Drug Administration on Nov. 21 granted orphan drug designation to rofecoxib (TRM-201), a cyclooxygenase 2–selective nonsteroidal anti-inflammatory drug (NSAID) intended to treat patients with hemophilic arthropathy (HA).
HA, a joint disease caused by hemarthrosis, is the largest cause of morbidity for hemophilia patients. There are currently no approved treatments in the United States.
The attempt at a reintroduction of rofecoxib specifically for the treatment of HA is being developed by Tremeau Pharmaceuticals.
Patients with hemophilia look to avoid traditional NSAIDs, as those drugs risk gastrointestinal ulcers and impair platelet aggregation. The current standard of care for HA is opioid treatment.
Rofecoxib and other NSAIDs cause an increased risk of serious cardiovascular thrombotic events and gastrointestinal adverse events.
Orphan drug status is available to treatments for rare disorders and provides a 7-year marketing exclusivity period against competition, along with tax credits and a waiver of Prescription Drug User Fee Act filing fees.
FDA approves epinephrine autoinjector for infants, small children
The Food and Drug Administration approved an epinephrine autoinjector constructed specifically to treat life-threatening allergic reactions in infants and small children weighing 16.5-33 pounds.
The Auvi-Q 0.1 mg autoinjector by kaléo was approved after a priority review by the FDA, with features such as “a voice prompt system that guides a user with step-by-step instructions through the delivery process,” according to a written statement from the company. This auto-injector has a shorter needle length and lower dose of epinephrine than other FDA-approved 0.15-mg and 0.3-mg epinephrine autoinjectors.
In a previous study of 51 infants with a mean weight of 24 pounds who were treated with a 0.15-mg epinephrine auto-injector with a standard 12.7-mm needle length, 43% were at risk of having the needle strike the bone. Unintentional injection of epinephrine into the intraosseous space can cause systemic absorption of the epinephrine and possible cardiac complications (Ann Allergy Asthma Immunol. 2017 Jun;118[6]:719-25.e1).
This new autoinjector with a shorter needle length was designed to obviate this problem, according to kaléo’s statement.
The Auvi-Q 0.1 mg autoinjector should be available to patients in the first half of 2018, the company said.
The Food and Drug Administration approved an epinephrine autoinjector constructed specifically to treat life-threatening allergic reactions in infants and small children weighing 16.5-33 pounds.
The Auvi-Q 0.1 mg autoinjector by kaléo was approved after a priority review by the FDA, with features such as “a voice prompt system that guides a user with step-by-step instructions through the delivery process,” according to a written statement from the company. This auto-injector has a shorter needle length and lower dose of epinephrine than other FDA-approved 0.15-mg and 0.3-mg epinephrine autoinjectors.
In a previous study of 51 infants with a mean weight of 24 pounds who were treated with a 0.15-mg epinephrine auto-injector with a standard 12.7-mm needle length, 43% were at risk of having the needle strike the bone. Unintentional injection of epinephrine into the intraosseous space can cause systemic absorption of the epinephrine and possible cardiac complications (Ann Allergy Asthma Immunol. 2017 Jun;118[6]:719-25.e1).
This new autoinjector with a shorter needle length was designed to obviate this problem, according to kaléo’s statement.
The Auvi-Q 0.1 mg autoinjector should be available to patients in the first half of 2018, the company said.
The Food and Drug Administration approved an epinephrine autoinjector constructed specifically to treat life-threatening allergic reactions in infants and small children weighing 16.5-33 pounds.
The Auvi-Q 0.1 mg autoinjector by kaléo was approved after a priority review by the FDA, with features such as “a voice prompt system that guides a user with step-by-step instructions through the delivery process,” according to a written statement from the company. This auto-injector has a shorter needle length and lower dose of epinephrine than other FDA-approved 0.15-mg and 0.3-mg epinephrine autoinjectors.
In a previous study of 51 infants with a mean weight of 24 pounds who were treated with a 0.15-mg epinephrine auto-injector with a standard 12.7-mm needle length, 43% were at risk of having the needle strike the bone. Unintentional injection of epinephrine into the intraosseous space can cause systemic absorption of the epinephrine and possible cardiac complications (Ann Allergy Asthma Immunol. 2017 Jun;118[6]:719-25.e1).
This new autoinjector with a shorter needle length was designed to obviate this problem, according to kaléo’s statement.
The Auvi-Q 0.1 mg autoinjector should be available to patients in the first half of 2018, the company said.
MMR vaccine coverage at 94% in kindergartners
, according to the Centers for Disease Control and Prevention.
A look at the map shows that state coverage of required MMR doses varied considerably. Mississippi (99.4%), Maryland (99.3%), Delaware (98.5%), California (97.3%), New York (97.3%), Texas (97.3%), and Louisiana (97.1%) were the furthest above the national median. Occupying the low end of the range were the District of Columbia (85.6%), Colorado (87.3%), Indiana (88.9%), Alaska (89.0%), Kansas (89.5%), and Idaho (89.9%), reported Ranee Seither, MPH, of the National Center for Immunization and Respiratory Disease, and associates at the CDC, Atlanta (MMWR 2017 Oct 13;66[40]:1073-80).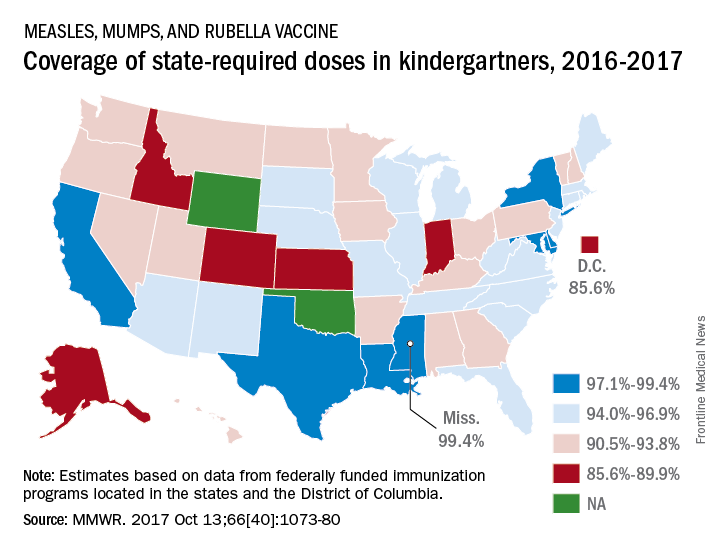
The data for the CDC analysis, which included 3,973,172 kindergartners for the 2016-2017 school year, were collected by federally funded immunization programs in the 50 states and D.C.
, according to the Centers for Disease Control and Prevention.
A look at the map shows that state coverage of required MMR doses varied considerably. Mississippi (99.4%), Maryland (99.3%), Delaware (98.5%), California (97.3%), New York (97.3%), Texas (97.3%), and Louisiana (97.1%) were the furthest above the national median. Occupying the low end of the range were the District of Columbia (85.6%), Colorado (87.3%), Indiana (88.9%), Alaska (89.0%), Kansas (89.5%), and Idaho (89.9%), reported Ranee Seither, MPH, of the National Center for Immunization and Respiratory Disease, and associates at the CDC, Atlanta (MMWR 2017 Oct 13;66[40]:1073-80).
The data for the CDC analysis, which included 3,973,172 kindergartners for the 2016-2017 school year, were collected by federally funded immunization programs in the 50 states and D.C.
, according to the Centers for Disease Control and Prevention.
A look at the map shows that state coverage of required MMR doses varied considerably. Mississippi (99.4%), Maryland (99.3%), Delaware (98.5%), California (97.3%), New York (97.3%), Texas (97.3%), and Louisiana (97.1%) were the furthest above the national median. Occupying the low end of the range were the District of Columbia (85.6%), Colorado (87.3%), Indiana (88.9%), Alaska (89.0%), Kansas (89.5%), and Idaho (89.9%), reported Ranee Seither, MPH, of the National Center for Immunization and Respiratory Disease, and associates at the CDC, Atlanta (MMWR 2017 Oct 13;66[40]:1073-80).
The data for the CDC analysis, which included 3,973,172 kindergartners for the 2016-2017 school year, were collected by federally funded immunization programs in the 50 states and D.C.
FROM MMWR
Benralizumab approved for eosinophilic asthma
This is the only respiratory biologic to provide fast and “near-complete depletion of eosinophils within 24 hours,” according to the statement from AstraZeneca.
Similar adverse events were seen in patients who took benralizumab and the placebo. AstraZeneca will market benralizumab under the trade name Fasenra.
“This is an important day for severe, eosinophilic asthma patients who have had limited treatment options for far too long,” said Eugene Bleecker, MD, professor and codirector of genetics, genomics, and precision medicine at the University of Arizona in Tucson, in the statement.
This is the only respiratory biologic to provide fast and “near-complete depletion of eosinophils within 24 hours,” according to the statement from AstraZeneca.
Similar adverse events were seen in patients who took benralizumab and the placebo. AstraZeneca will market benralizumab under the trade name Fasenra.
“This is an important day for severe, eosinophilic asthma patients who have had limited treatment options for far too long,” said Eugene Bleecker, MD, professor and codirector of genetics, genomics, and precision medicine at the University of Arizona in Tucson, in the statement.
This is the only respiratory biologic to provide fast and “near-complete depletion of eosinophils within 24 hours,” according to the statement from AstraZeneca.
Similar adverse events were seen in patients who took benralizumab and the placebo. AstraZeneca will market benralizumab under the trade name Fasenra.
“This is an important day for severe, eosinophilic asthma patients who have had limited treatment options for far too long,” said Eugene Bleecker, MD, professor and codirector of genetics, genomics, and precision medicine at the University of Arizona in Tucson, in the statement.

