User login
Zika-linked birth defects climbing in U.S. hot spots
The prevalence of birth defects strongly linked with congenital Zika virus infection increased 21% from the first to the second half of 2016 in areas of the United States with local, endemic transmission: Puerto Rico, south Florida, and southern Texas, according to a report in the Jan. 26 edition of Morbidity and Mortality Weekly Report.
In those areas, complications strongly associated with Zika – including microcephaly; brain and eye abnormalities; and neurogenic hip dislocation, clubfoot, hearing loss, and arthrogryposis – jumped from 2.0 to 2.4 cases per 1,000 live births, with 140 cases in the first half of the year and 169 cases in the second (P = .009). Microcephaly and brain abnormalities were the most common problems.
In places with less than one confirmed Zika case from travel per 100,000 residents, such as Hawaii and Utah, the prevalence of birth defects strongly linked to Zika actually dropped from 2.8 cases per 1,000 live births to 2.4 in 2016.
The 15 U.S. jurisdictions in the study included nearly 1 million live births, representing approximately one fourth of the total live births in the United States in 2016. The live birth rate was 92% among the 2,962 infants and fetuses with Zika-associated birth defects.
All the jurisdictions had existing birth defects surveillance systems that quickly adapted to monitor for potential Zika defects. However, although strongly associated with Zika, there’s no guarantee that the birth defects in the study were actually caused by the virus, the researchers noted.
“These data will help communities plan for needed resources to care for affected patients and families and can serve as a foundation for linking and evaluating health and developmental outcomes of affected children,” said the investigators, led by Augustina Delaney, PhD, of the National Center on Birth Defects and Developmental Disabilities at the Centers for Disease Control and Prevention in Atlanta.
The work was the first population-based birth defect surveillance report for Zika. The CDC follows confirmed Zika cases in pregnant women and their offspring closely, but only a small portion of women are actually tested, so there’s likely far more cases of congenital Zika infection than show up in registries. Despite its limits, birth defect surveillance likely provides a more accurate picture of the actual extent of the problem.
It’s not known why Zika-linked birth defects dropped off in areas with low or no travel-associated cases. “However ... further case ascertainment from the final quarter of 2016 is anticipated in all jurisdictions,” so the numbers could change, the authors said.
They had no conflicts of interest.
SOURCE: Delaney A, et. al. MMWR Morb Mortal Wkly Rep. 2018 Jan 26;67(3):91-6
Although these birth defects are not specific to congenital Zika virus infection, only those defects found previously to be most closely aligned with congenital Zika infection had increased prevalence.
It is critical that public health surveillance programs continue reporting the occurrence of these birth defects to monitor for trends following the Zika virus outbreak.
Brenda Fitzgerald , MD, is the director of the Centers for Disease Control and Prevention in Atlanta. Coleen A. Boyle , PhD, is the director of the CDC National Center on Birth Defects and Developmental Disabilities, and Margaret Honein , PhD, is chief of the agency’s Birth Defects Branch. They made their comments Jan. 25 in JAMA, and had no conflicts of interest (Jama. 2018 Jan 25. doi: 10.1001/jama.2018.0126).
Although these birth defects are not specific to congenital Zika virus infection, only those defects found previously to be most closely aligned with congenital Zika infection had increased prevalence.
It is critical that public health surveillance programs continue reporting the occurrence of these birth defects to monitor for trends following the Zika virus outbreak.
Brenda Fitzgerald , MD, is the director of the Centers for Disease Control and Prevention in Atlanta. Coleen A. Boyle , PhD, is the director of the CDC National Center on Birth Defects and Developmental Disabilities, and Margaret Honein , PhD, is chief of the agency’s Birth Defects Branch. They made their comments Jan. 25 in JAMA, and had no conflicts of interest (Jama. 2018 Jan 25. doi: 10.1001/jama.2018.0126).
Although these birth defects are not specific to congenital Zika virus infection, only those defects found previously to be most closely aligned with congenital Zika infection had increased prevalence.
It is critical that public health surveillance programs continue reporting the occurrence of these birth defects to monitor for trends following the Zika virus outbreak.
Brenda Fitzgerald , MD, is the director of the Centers for Disease Control and Prevention in Atlanta. Coleen A. Boyle , PhD, is the director of the CDC National Center on Birth Defects and Developmental Disabilities, and Margaret Honein , PhD, is chief of the agency’s Birth Defects Branch. They made their comments Jan. 25 in JAMA, and had no conflicts of interest (Jama. 2018 Jan 25. doi: 10.1001/jama.2018.0126).
The prevalence of birth defects strongly linked with congenital Zika virus infection increased 21% from the first to the second half of 2016 in areas of the United States with local, endemic transmission: Puerto Rico, south Florida, and southern Texas, according to a report in the Jan. 26 edition of Morbidity and Mortality Weekly Report.
In those areas, complications strongly associated with Zika – including microcephaly; brain and eye abnormalities; and neurogenic hip dislocation, clubfoot, hearing loss, and arthrogryposis – jumped from 2.0 to 2.4 cases per 1,000 live births, with 140 cases in the first half of the year and 169 cases in the second (P = .009). Microcephaly and brain abnormalities were the most common problems.
In places with less than one confirmed Zika case from travel per 100,000 residents, such as Hawaii and Utah, the prevalence of birth defects strongly linked to Zika actually dropped from 2.8 cases per 1,000 live births to 2.4 in 2016.
The 15 U.S. jurisdictions in the study included nearly 1 million live births, representing approximately one fourth of the total live births in the United States in 2016. The live birth rate was 92% among the 2,962 infants and fetuses with Zika-associated birth defects.
All the jurisdictions had existing birth defects surveillance systems that quickly adapted to monitor for potential Zika defects. However, although strongly associated with Zika, there’s no guarantee that the birth defects in the study were actually caused by the virus, the researchers noted.
“These data will help communities plan for needed resources to care for affected patients and families and can serve as a foundation for linking and evaluating health and developmental outcomes of affected children,” said the investigators, led by Augustina Delaney, PhD, of the National Center on Birth Defects and Developmental Disabilities at the Centers for Disease Control and Prevention in Atlanta.
The work was the first population-based birth defect surveillance report for Zika. The CDC follows confirmed Zika cases in pregnant women and their offspring closely, but only a small portion of women are actually tested, so there’s likely far more cases of congenital Zika infection than show up in registries. Despite its limits, birth defect surveillance likely provides a more accurate picture of the actual extent of the problem.
It’s not known why Zika-linked birth defects dropped off in areas with low or no travel-associated cases. “However ... further case ascertainment from the final quarter of 2016 is anticipated in all jurisdictions,” so the numbers could change, the authors said.
They had no conflicts of interest.
SOURCE: Delaney A, et. al. MMWR Morb Mortal Wkly Rep. 2018 Jan 26;67(3):91-6
The prevalence of birth defects strongly linked with congenital Zika virus infection increased 21% from the first to the second half of 2016 in areas of the United States with local, endemic transmission: Puerto Rico, south Florida, and southern Texas, according to a report in the Jan. 26 edition of Morbidity and Mortality Weekly Report.
In those areas, complications strongly associated with Zika – including microcephaly; brain and eye abnormalities; and neurogenic hip dislocation, clubfoot, hearing loss, and arthrogryposis – jumped from 2.0 to 2.4 cases per 1,000 live births, with 140 cases in the first half of the year and 169 cases in the second (P = .009). Microcephaly and brain abnormalities were the most common problems.
In places with less than one confirmed Zika case from travel per 100,000 residents, such as Hawaii and Utah, the prevalence of birth defects strongly linked to Zika actually dropped from 2.8 cases per 1,000 live births to 2.4 in 2016.
The 15 U.S. jurisdictions in the study included nearly 1 million live births, representing approximately one fourth of the total live births in the United States in 2016. The live birth rate was 92% among the 2,962 infants and fetuses with Zika-associated birth defects.
All the jurisdictions had existing birth defects surveillance systems that quickly adapted to monitor for potential Zika defects. However, although strongly associated with Zika, there’s no guarantee that the birth defects in the study were actually caused by the virus, the researchers noted.
“These data will help communities plan for needed resources to care for affected patients and families and can serve as a foundation for linking and evaluating health and developmental outcomes of affected children,” said the investigators, led by Augustina Delaney, PhD, of the National Center on Birth Defects and Developmental Disabilities at the Centers for Disease Control and Prevention in Atlanta.
The work was the first population-based birth defect surveillance report for Zika. The CDC follows confirmed Zika cases in pregnant women and their offspring closely, but only a small portion of women are actually tested, so there’s likely far more cases of congenital Zika infection than show up in registries. Despite its limits, birth defect surveillance likely provides a more accurate picture of the actual extent of the problem.
It’s not known why Zika-linked birth defects dropped off in areas with low or no travel-associated cases. “However ... further case ascertainment from the final quarter of 2016 is anticipated in all jurisdictions,” so the numbers could change, the authors said.
They had no conflicts of interest.
SOURCE: Delaney A, et. al. MMWR Morb Mortal Wkly Rep. 2018 Jan 26;67(3):91-6
FROM MMWR
Key clinical point: Although microcephaly and other birth defects strongly associated with Zika virus are holding steady or even decreasing elsewhere in the United States, there was an uptick in 2016 in areas with endemic transmission.
Major finding: The prevalence of birth defects strongly related to congenital Zika virus infection increased 21% from the first to the second half of 2016 in southern Texas, south Florida, and Puerto Rico.
Study details: Birth defects surveillance in about a quarter of the infants born in the United States in 2016.
Disclosures: The investigators had no conflicts of interest.
Source: Delaney A, et. al. MMWR Morb Mortal Wkly Rep. 2018 Jan 26;67(3):91-6
Women filling more ADHD prescriptions
, according to the Centers for Disease Control and Prevention.
In 2003, 0.9% of women aged 15-44 years with private employer–sponsored insurance filled a prescription for an ADHD medication. By 2015, that figure had gone up to 4.0% for an increase of 344% that was unevenly split by medication class: prescriptions for stimulants were up by 388%, but nonstimulants had no change, wrote Kayla N. Anderson, PhD, and her associates in the Morbidity and Mortality Weekly Report.
“The substantial increase in the percentage of reproductive-aged women filling ADHD medication prescriptions from 2003 to 2015 ... is of public health concern given the high percentage of unintended pregnancies and uncertainty concerning the safety of ADHD medication exposure before and during pregnancy,” they wrote.
This analysis was restricted to women with at least 11 months of enrollment in a private health insurance plan that included prescription drug coverage during the year of interest. The sample included a median of 4.6 million women each year.
SOURCE: Anderson K et al. MMWR. 2018 Jan 19;76(2):66-70.
, according to the Centers for Disease Control and Prevention.
In 2003, 0.9% of women aged 15-44 years with private employer–sponsored insurance filled a prescription for an ADHD medication. By 2015, that figure had gone up to 4.0% for an increase of 344% that was unevenly split by medication class: prescriptions for stimulants were up by 388%, but nonstimulants had no change, wrote Kayla N. Anderson, PhD, and her associates in the Morbidity and Mortality Weekly Report.
“The substantial increase in the percentage of reproductive-aged women filling ADHD medication prescriptions from 2003 to 2015 ... is of public health concern given the high percentage of unintended pregnancies and uncertainty concerning the safety of ADHD medication exposure before and during pregnancy,” they wrote.
This analysis was restricted to women with at least 11 months of enrollment in a private health insurance plan that included prescription drug coverage during the year of interest. The sample included a median of 4.6 million women each year.
SOURCE: Anderson K et al. MMWR. 2018 Jan 19;76(2):66-70.
, according to the Centers for Disease Control and Prevention.
In 2003, 0.9% of women aged 15-44 years with private employer–sponsored insurance filled a prescription for an ADHD medication. By 2015, that figure had gone up to 4.0% for an increase of 344% that was unevenly split by medication class: prescriptions for stimulants were up by 388%, but nonstimulants had no change, wrote Kayla N. Anderson, PhD, and her associates in the Morbidity and Mortality Weekly Report.
“The substantial increase in the percentage of reproductive-aged women filling ADHD medication prescriptions from 2003 to 2015 ... is of public health concern given the high percentage of unintended pregnancies and uncertainty concerning the safety of ADHD medication exposure before and during pregnancy,” they wrote.
This analysis was restricted to women with at least 11 months of enrollment in a private health insurance plan that included prescription drug coverage during the year of interest. The sample included a median of 4.6 million women each year.
SOURCE: Anderson K et al. MMWR. 2018 Jan 19;76(2):66-70.
FROM MMWR
Flu season takes another turn for the worse
By one measure at least – the proportion of outpatient visits for influenza-like illness (ILI) – this flu season is now the worst in almost a decade, according to data from the Centers for Disease Control and Prevention.
which hit an early peak of 7.7% in October of 2009. The slight pause that occurred in the first week of January as the rate only rose from 5.7% to 5.8% now looks more like the earlier trend from December, when the level of outpatient visits more than doubled over a 3-week period, data from the CDC FluView website show.
“The geographic spread of influenza in Puerto Rico and 49 states was reported as widespread” for the week ending Jan. 13, and 24 states had the highest level of ILI activity on the CDC’s 1-10 scale, the CDC influenza division reported Jan 19.
There were 10 flu-related pediatric deaths reported during the week, with two occurring in the week ending Jan. 13. A total of 30 deaths in children have been associated with influenza so far for the 2017-2018 season, the CDC said.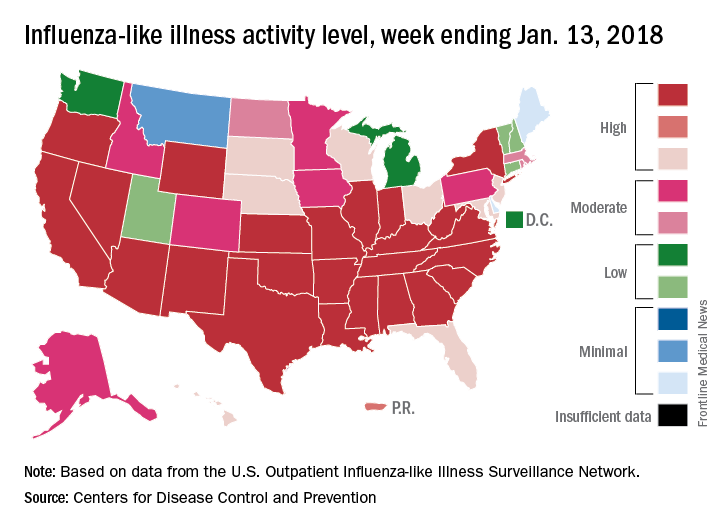
By one measure at least – the proportion of outpatient visits for influenza-like illness (ILI) – this flu season is now the worst in almost a decade, according to data from the Centers for Disease Control and Prevention.
which hit an early peak of 7.7% in October of 2009. The slight pause that occurred in the first week of January as the rate only rose from 5.7% to 5.8% now looks more like the earlier trend from December, when the level of outpatient visits more than doubled over a 3-week period, data from the CDC FluView website show.
“The geographic spread of influenza in Puerto Rico and 49 states was reported as widespread” for the week ending Jan. 13, and 24 states had the highest level of ILI activity on the CDC’s 1-10 scale, the CDC influenza division reported Jan 19.
There were 10 flu-related pediatric deaths reported during the week, with two occurring in the week ending Jan. 13. A total of 30 deaths in children have been associated with influenza so far for the 2017-2018 season, the CDC said.
By one measure at least – the proportion of outpatient visits for influenza-like illness (ILI) – this flu season is now the worst in almost a decade, according to data from the Centers for Disease Control and Prevention.
which hit an early peak of 7.7% in October of 2009. The slight pause that occurred in the first week of January as the rate only rose from 5.7% to 5.8% now looks more like the earlier trend from December, when the level of outpatient visits more than doubled over a 3-week period, data from the CDC FluView website show.
“The geographic spread of influenza in Puerto Rico and 49 states was reported as widespread” for the week ending Jan. 13, and 24 states had the highest level of ILI activity on the CDC’s 1-10 scale, the CDC influenza division reported Jan 19.
There were 10 flu-related pediatric deaths reported during the week, with two occurring in the week ending Jan. 13. A total of 30 deaths in children have been associated with influenza so far for the 2017-2018 season, the CDC said.
Listen up: Acoustic device useful for diabetic foot ulcers
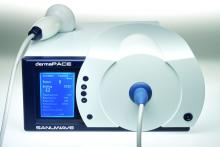
The Food & Drug Administration has approved the marketing of a device that uses acoustic shock waves to boost wound closure in patients with diabetic foot ulcers (DFUs), an especially stubborn and dangerous condition.
The treatment is experimental, and only limited research into its effectiveness has been published. Still, representatives of its manufacturer say the device, known as dermaPACE, has produced promising results as a secondary treatment in stubborn cases.
A wound care specialist said in an interview that the shock wave technology appears to hold promise.
“A shortcoming in the field of wound care is that providers are typically not trained in a standardized fashion on when and how to a perform meticulous excisional sharp debridement of a wound,” said Bill Tettelbach, MD, systems medical director of Wound Care & Hyperbaric Medicine Services at Intermountain Healthcare in Salt Lake City. “In the majority of cases, the better the debridement, the more rapidly the patient will obtain wound closure.”
This new therapy may provide a benefit as a secondary treatment, especially when the patient cannot tolerate extensive sharp debridement, he said. It also could potentially improve biofilm penetration of antimicrobial topical treatments, he said.
DFUs are believed to affect as many as 1 in 4 people with diabetes over the course of their lifetimes. A 2014 report estimated that care of these wounds costs insurers as much as $13 billion a year in the U.S. alone (Diabetes Care. 2014 Mar;37[3]:651-8).
Treatment options include debridement and, in more extreme cases, hyperbaric oxygen treatment. Amputation can be required if treatment is unsuccessful.
According to Mr. Stegagno, the shock wave device is about the size of a desktop computer from a decade ago. A high-voltage generator box is connected to a handheld therapy head and delivers an acoustic pulse to the patient. The system “is like a spark plug that you see in your automobile,” he said. “It’s pretty much the same technology as lithotripsy, just downsized significantly. The key part is a highly focused, high-energy pulse.”
In a news release, the FDA said it examined the results of two studies of patients with diabetes who received usual DFU care along with either the shock wave therapy or a sham therapy. A total of 336 patients took part in the multicenter, randomized, double-blind studies.
According to the FDA, the studies found a 44% wound closure rate at 24 weeks in patients who had undergone 1-7 shock wave treatments, compared with the 30% wound closure rate in those who received the sham treatment.
Side effects included pain while the device was applied, bruising and numbness, migraines, nausea, fainting, wound infection, fever, and infection beyond the wound such as cellulitis and osteomyelitis.
“There were no meaningful statistical differences in the adverse event rates between the dermaPACE-treated patients and the sham-control group,” Mr. Stegagno said. “There were no issues regarding the tolerability of the treatment, which suggests that a second course of treatment, if needed, is a clinically viable option.”
Mr. Stegagno said the FDA expressed concern about “increased incidences of osteomyelitis at later points in the trials, particularly at the 10-week mark and later.” In response to the agency’s concerns, warning statements were added to labeling, he said.
According to Mr. Stegagno, only one study into the shock wave treatment for DFU has been published, although research has been released through posters and abstracts. The small published study favorably compared shock wave therapy with hyperbaric oxygen therapy. (Diabetes Res Clin Pract. 2011 May;92[2]:187-93)
“Sanuwave will be sponsoring additional studies later this year in the [United States] as follow-on studies to the just-completed DFU trials,” Mr. Stegagno said.
The FDA says the device is intended to be used in adults aged 22 and up with certain types of chronic DFUs. The Sanuwave company says patients should be treated with 4-8 applications over 2-10 weeks.
The shock wave process appears to boost healing through a process that leads to inflammatory responses and oxygenation, Mr. Stegagno said, by first creating an initial compression phase that “squeezes the cell and creates a microtrauma.”
“The cell wakes up and says, ‘Something just punched me,’ ” he said. “This tissue and cellular disruption is believed to initiate the cellular signaling for growth factors and other proteins noted in studies.”
The effects of negative pressure also play a role in stimulation of the wound, he said.
The shock wave therapy will cost an estimated $3,000-$4,000 per protocol of 8 treatments, said Kevin A. Richardson II, the CEO and chairman of the board at Sanuwave, in an interview. The initial plan is for the company to place the devices with doctors while the firm still owns the machines, he said.
The FDA approved the marketing of the device as part of its de novo premarket review pathway, which allows certain new types of devices to be approved when approved similar devices don’t yet exist for the purposes of comparison.
Mr. Stegagno and Mr. Richardson work for Sanuwave. Dr. Tettelbach reported no relevant disclosures.
The Food & Drug Administration has approved the marketing of a device that uses acoustic shock waves to boost wound closure in patients with diabetic foot ulcers (DFUs), an especially stubborn and dangerous condition.
The treatment is experimental, and only limited research into its effectiveness has been published. Still, representatives of its manufacturer say the device, known as dermaPACE, has produced promising results as a secondary treatment in stubborn cases.
A wound care specialist said in an interview that the shock wave technology appears to hold promise.
“A shortcoming in the field of wound care is that providers are typically not trained in a standardized fashion on when and how to a perform meticulous excisional sharp debridement of a wound,” said Bill Tettelbach, MD, systems medical director of Wound Care & Hyperbaric Medicine Services at Intermountain Healthcare in Salt Lake City. “In the majority of cases, the better the debridement, the more rapidly the patient will obtain wound closure.”
This new therapy may provide a benefit as a secondary treatment, especially when the patient cannot tolerate extensive sharp debridement, he said. It also could potentially improve biofilm penetration of antimicrobial topical treatments, he said.
DFUs are believed to affect as many as 1 in 4 people with diabetes over the course of their lifetimes. A 2014 report estimated that care of these wounds costs insurers as much as $13 billion a year in the U.S. alone (Diabetes Care. 2014 Mar;37[3]:651-8).
Treatment options include debridement and, in more extreme cases, hyperbaric oxygen treatment. Amputation can be required if treatment is unsuccessful.
According to Mr. Stegagno, the shock wave device is about the size of a desktop computer from a decade ago. A high-voltage generator box is connected to a handheld therapy head and delivers an acoustic pulse to the patient. The system “is like a spark plug that you see in your automobile,” he said. “It’s pretty much the same technology as lithotripsy, just downsized significantly. The key part is a highly focused, high-energy pulse.”
In a news release, the FDA said it examined the results of two studies of patients with diabetes who received usual DFU care along with either the shock wave therapy or a sham therapy. A total of 336 patients took part in the multicenter, randomized, double-blind studies.
According to the FDA, the studies found a 44% wound closure rate at 24 weeks in patients who had undergone 1-7 shock wave treatments, compared with the 30% wound closure rate in those who received the sham treatment.
Side effects included pain while the device was applied, bruising and numbness, migraines, nausea, fainting, wound infection, fever, and infection beyond the wound such as cellulitis and osteomyelitis.
“There were no meaningful statistical differences in the adverse event rates between the dermaPACE-treated patients and the sham-control group,” Mr. Stegagno said. “There were no issues regarding the tolerability of the treatment, which suggests that a second course of treatment, if needed, is a clinically viable option.”
Mr. Stegagno said the FDA expressed concern about “increased incidences of osteomyelitis at later points in the trials, particularly at the 10-week mark and later.” In response to the agency’s concerns, warning statements were added to labeling, he said.
According to Mr. Stegagno, only one study into the shock wave treatment for DFU has been published, although research has been released through posters and abstracts. The small published study favorably compared shock wave therapy with hyperbaric oxygen therapy. (Diabetes Res Clin Pract. 2011 May;92[2]:187-93)
“Sanuwave will be sponsoring additional studies later this year in the [United States] as follow-on studies to the just-completed DFU trials,” Mr. Stegagno said.
The FDA says the device is intended to be used in adults aged 22 and up with certain types of chronic DFUs. The Sanuwave company says patients should be treated with 4-8 applications over 2-10 weeks.
The shock wave process appears to boost healing through a process that leads to inflammatory responses and oxygenation, Mr. Stegagno said, by first creating an initial compression phase that “squeezes the cell and creates a microtrauma.”
“The cell wakes up and says, ‘Something just punched me,’ ” he said. “This tissue and cellular disruption is believed to initiate the cellular signaling for growth factors and other proteins noted in studies.”
The effects of negative pressure also play a role in stimulation of the wound, he said.
The shock wave therapy will cost an estimated $3,000-$4,000 per protocol of 8 treatments, said Kevin A. Richardson II, the CEO and chairman of the board at Sanuwave, in an interview. The initial plan is for the company to place the devices with doctors while the firm still owns the machines, he said.
The FDA approved the marketing of the device as part of its de novo premarket review pathway, which allows certain new types of devices to be approved when approved similar devices don’t yet exist for the purposes of comparison.
Mr. Stegagno and Mr. Richardson work for Sanuwave. Dr. Tettelbach reported no relevant disclosures.
The Food & Drug Administration has approved the marketing of a device that uses acoustic shock waves to boost wound closure in patients with diabetic foot ulcers (DFUs), an especially stubborn and dangerous condition.
The treatment is experimental, and only limited research into its effectiveness has been published. Still, representatives of its manufacturer say the device, known as dermaPACE, has produced promising results as a secondary treatment in stubborn cases.
A wound care specialist said in an interview that the shock wave technology appears to hold promise.
“A shortcoming in the field of wound care is that providers are typically not trained in a standardized fashion on when and how to a perform meticulous excisional sharp debridement of a wound,” said Bill Tettelbach, MD, systems medical director of Wound Care & Hyperbaric Medicine Services at Intermountain Healthcare in Salt Lake City. “In the majority of cases, the better the debridement, the more rapidly the patient will obtain wound closure.”
This new therapy may provide a benefit as a secondary treatment, especially when the patient cannot tolerate extensive sharp debridement, he said. It also could potentially improve biofilm penetration of antimicrobial topical treatments, he said.
DFUs are believed to affect as many as 1 in 4 people with diabetes over the course of their lifetimes. A 2014 report estimated that care of these wounds costs insurers as much as $13 billion a year in the U.S. alone (Diabetes Care. 2014 Mar;37[3]:651-8).
Treatment options include debridement and, in more extreme cases, hyperbaric oxygen treatment. Amputation can be required if treatment is unsuccessful.
According to Mr. Stegagno, the shock wave device is about the size of a desktop computer from a decade ago. A high-voltage generator box is connected to a handheld therapy head and delivers an acoustic pulse to the patient. The system “is like a spark plug that you see in your automobile,” he said. “It’s pretty much the same technology as lithotripsy, just downsized significantly. The key part is a highly focused, high-energy pulse.”
In a news release, the FDA said it examined the results of two studies of patients with diabetes who received usual DFU care along with either the shock wave therapy or a sham therapy. A total of 336 patients took part in the multicenter, randomized, double-blind studies.
According to the FDA, the studies found a 44% wound closure rate at 24 weeks in patients who had undergone 1-7 shock wave treatments, compared with the 30% wound closure rate in those who received the sham treatment.
Side effects included pain while the device was applied, bruising and numbness, migraines, nausea, fainting, wound infection, fever, and infection beyond the wound such as cellulitis and osteomyelitis.
“There were no meaningful statistical differences in the adverse event rates between the dermaPACE-treated patients and the sham-control group,” Mr. Stegagno said. “There were no issues regarding the tolerability of the treatment, which suggests that a second course of treatment, if needed, is a clinically viable option.”
Mr. Stegagno said the FDA expressed concern about “increased incidences of osteomyelitis at later points in the trials, particularly at the 10-week mark and later.” In response to the agency’s concerns, warning statements were added to labeling, he said.
According to Mr. Stegagno, only one study into the shock wave treatment for DFU has been published, although research has been released through posters and abstracts. The small published study favorably compared shock wave therapy with hyperbaric oxygen therapy. (Diabetes Res Clin Pract. 2011 May;92[2]:187-93)
“Sanuwave will be sponsoring additional studies later this year in the [United States] as follow-on studies to the just-completed DFU trials,” Mr. Stegagno said.
The FDA says the device is intended to be used in adults aged 22 and up with certain types of chronic DFUs. The Sanuwave company says patients should be treated with 4-8 applications over 2-10 weeks.
The shock wave process appears to boost healing through a process that leads to inflammatory responses and oxygenation, Mr. Stegagno said, by first creating an initial compression phase that “squeezes the cell and creates a microtrauma.”
“The cell wakes up and says, ‘Something just punched me,’ ” he said. “This tissue and cellular disruption is believed to initiate the cellular signaling for growth factors and other proteins noted in studies.”
The effects of negative pressure also play a role in stimulation of the wound, he said.
The shock wave therapy will cost an estimated $3,000-$4,000 per protocol of 8 treatments, said Kevin A. Richardson II, the CEO and chairman of the board at Sanuwave, in an interview. The initial plan is for the company to place the devices with doctors while the firm still owns the machines, he said.
The FDA approved the marketing of the device as part of its de novo premarket review pathway, which allows certain new types of devices to be approved when approved similar devices don’t yet exist for the purposes of comparison.
Mr. Stegagno and Mr. Richardson work for Sanuwave. Dr. Tettelbach reported no relevant disclosures.
FDA: LifeVest wearable defibrillator has safety issue
The Zoll LifeVest 4000, a wearable defibrillator, could fail to deliver a treatment shock after displaying the message “Call for service: Device has a problem that may require service. Call ZOLL for service, Message Code 102,” according to the FDA.
“Failure to contact Zoll and immediately replace the device after Message Code 102 appears on the device screen may result in serious patient harm or death of the patient because the device may fail to deliver therapy appropriately when needed” according to an FDA press release.
Only one death associated with “Message Code 102” malfunction of LifeVest has been reported, but about 0.1% of devices have displayed the “Message Code 102” error. According to Zoll, roughly 33,670 devices have been distributed as of Nov. 14, 2017, with nearly 75% of them distributed in the United States.
The FDA has indicated that it will continue to work with Zoll to monitor adverse events associated with the “Message Code 102” error and work on finding a permanent solution to this problem. Recommendations for physicians, caregivers, and patients regarding how to respond to error messages can be found here.
The Zoll LifeVest 4000, a wearable defibrillator, could fail to deliver a treatment shock after displaying the message “Call for service: Device has a problem that may require service. Call ZOLL for service, Message Code 102,” according to the FDA.
“Failure to contact Zoll and immediately replace the device after Message Code 102 appears on the device screen may result in serious patient harm or death of the patient because the device may fail to deliver therapy appropriately when needed” according to an FDA press release.
Only one death associated with “Message Code 102” malfunction of LifeVest has been reported, but about 0.1% of devices have displayed the “Message Code 102” error. According to Zoll, roughly 33,670 devices have been distributed as of Nov. 14, 2017, with nearly 75% of them distributed in the United States.
The FDA has indicated that it will continue to work with Zoll to monitor adverse events associated with the “Message Code 102” error and work on finding a permanent solution to this problem. Recommendations for physicians, caregivers, and patients regarding how to respond to error messages can be found here.
The Zoll LifeVest 4000, a wearable defibrillator, could fail to deliver a treatment shock after displaying the message “Call for service: Device has a problem that may require service. Call ZOLL for service, Message Code 102,” according to the FDA.
“Failure to contact Zoll and immediately replace the device after Message Code 102 appears on the device screen may result in serious patient harm or death of the patient because the device may fail to deliver therapy appropriately when needed” according to an FDA press release.
Only one death associated with “Message Code 102” malfunction of LifeVest has been reported, but about 0.1% of devices have displayed the “Message Code 102” error. According to Zoll, roughly 33,670 devices have been distributed as of Nov. 14, 2017, with nearly 75% of them distributed in the United States.
The FDA has indicated that it will continue to work with Zoll to monitor adverse events associated with the “Message Code 102” error and work on finding a permanent solution to this problem. Recommendations for physicians, caregivers, and patients regarding how to respond to error messages can be found here.
U.S. influenza activity widespread to start 2018
As far as the influenza virus is concerned, the new year started in the same way as the old one ended: with almost half of the states at the highest level of flu activity, according to the Centers for Disease Control and Prevention.
For the week ending Jan. 6, 2018, there were 23 states – including California, Illinois, and Texas – at level 10 on the CDC’s 1-10 scale for influenza-like illness (ILI) activity, which was up from 22 for the last full week of 2017. Joining the 23 states in the “high” range were New Jersey and Ohio at level 9 and Colorado at level 8, the CDC’s influenza division reported Jan. 12.
Seven flu-related pediatric deaths were reported during the week ending Jan. 6, although one occurred during the week ending Dec. 16 and two were during the week ending Dec. 23. There have been a total of 20 pediatric deaths related to influenza so far for the 2017-2018 season, the CDC said. In 2016-2017, there were 110 pediatric deaths from the flu.
As far as the influenza virus is concerned, the new year started in the same way as the old one ended: with almost half of the states at the highest level of flu activity, according to the Centers for Disease Control and Prevention.
For the week ending Jan. 6, 2018, there were 23 states – including California, Illinois, and Texas – at level 10 on the CDC’s 1-10 scale for influenza-like illness (ILI) activity, which was up from 22 for the last full week of 2017. Joining the 23 states in the “high” range were New Jersey and Ohio at level 9 and Colorado at level 8, the CDC’s influenza division reported Jan. 12.
Seven flu-related pediatric deaths were reported during the week ending Jan. 6, although one occurred during the week ending Dec. 16 and two were during the week ending Dec. 23. There have been a total of 20 pediatric deaths related to influenza so far for the 2017-2018 season, the CDC said. In 2016-2017, there were 110 pediatric deaths from the flu.
As far as the influenza virus is concerned, the new year started in the same way as the old one ended: with almost half of the states at the highest level of flu activity, according to the Centers for Disease Control and Prevention.
For the week ending Jan. 6, 2018, there were 23 states – including California, Illinois, and Texas – at level 10 on the CDC’s 1-10 scale for influenza-like illness (ILI) activity, which was up from 22 for the last full week of 2017. Joining the 23 states in the “high” range were New Jersey and Ohio at level 9 and Colorado at level 8, the CDC’s influenza division reported Jan. 12.
Seven flu-related pediatric deaths were reported during the week ending Jan. 6, although one occurred during the week ending Dec. 16 and two were during the week ending Dec. 23. There have been a total of 20 pediatric deaths related to influenza so far for the 2017-2018 season, the CDC said. In 2016-2017, there were 110 pediatric deaths from the flu.
Office visits have a chronic problem
according to the National Center for Health Statistics.
That year, chronic problems were the major reason for 40% of all office-based physician visits, making them significantly more common than the other four broad categories of visit types: new problem (26%), preventive care (19%), injury (9%), and pre- or postsurgery care (6%), the NCHS said in a recent Data Brief.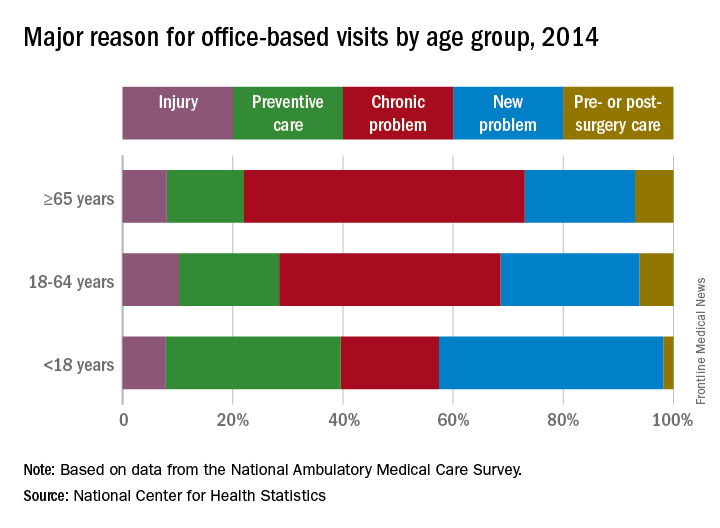
Children were significantly more likely to have a new problem (41% of visits) or get preventive care (32%) than were the other age groups and were significantly less likely to have a chronic problem (18%) or to receive pre- or postsurgery care (2%), according to data from the 2014 National Ambulatory Medical Care Survey, which included 45,710 visit records from 9,989 physicians.
SOURCE: Ashman JJ et al. NCHS Data Brief. 2017;(292):1-8e.
according to the National Center for Health Statistics.
That year, chronic problems were the major reason for 40% of all office-based physician visits, making them significantly more common than the other four broad categories of visit types: new problem (26%), preventive care (19%), injury (9%), and pre- or postsurgery care (6%), the NCHS said in a recent Data Brief.
Children were significantly more likely to have a new problem (41% of visits) or get preventive care (32%) than were the other age groups and were significantly less likely to have a chronic problem (18%) or to receive pre- or postsurgery care (2%), according to data from the 2014 National Ambulatory Medical Care Survey, which included 45,710 visit records from 9,989 physicians.
SOURCE: Ashman JJ et al. NCHS Data Brief. 2017;(292):1-8e.
according to the National Center for Health Statistics.
That year, chronic problems were the major reason for 40% of all office-based physician visits, making them significantly more common than the other four broad categories of visit types: new problem (26%), preventive care (19%), injury (9%), and pre- or postsurgery care (6%), the NCHS said in a recent Data Brief.
Children were significantly more likely to have a new problem (41% of visits) or get preventive care (32%) than were the other age groups and were significantly less likely to have a chronic problem (18%) or to receive pre- or postsurgery care (2%), according to data from the 2014 National Ambulatory Medical Care Survey, which included 45,710 visit records from 9,989 physicians.
SOURCE: Ashman JJ et al. NCHS Data Brief. 2017;(292):1-8e.
Normal prepregnancy weight becoming less normal
according to the Centers for Disease Control and Prevention.
The overall prevalence of normal prepregnancy weight declined from 47.3% to 45.1% over that period in 36 states, the District of Columbia, and New York City, which reports natality data separately from New York state. The decreases were statistically significant in 26 states and New York City, the CDC investigators reported (MMWR. 2018 Jan 5;66[51-2]:1402-7).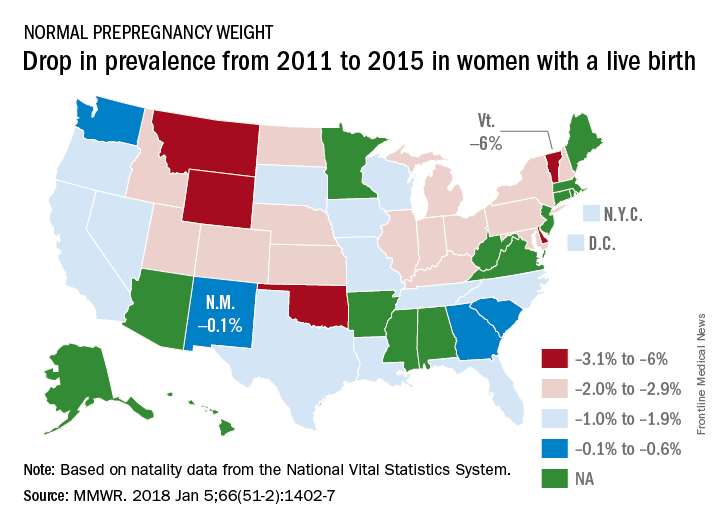
Based on data from 48 states, D.C., and New York City, the distribution of prevalence for the BMI categories in 2015 was 3.6% underweight, 45% normal weight, 25.8% overweight, and 25.6% obese, the investigators said.
The CDC analysis was based on natality data from the National Vital Statistics System. The standard birth certificate was revised in 2003 to include maternal height and prepregnancy weight, but only 38 jurisdictions were using it by 2011. By 2015, all states except Connecticut and New Jersey had adopted its use.
according to the Centers for Disease Control and Prevention.
The overall prevalence of normal prepregnancy weight declined from 47.3% to 45.1% over that period in 36 states, the District of Columbia, and New York City, which reports natality data separately from New York state. The decreases were statistically significant in 26 states and New York City, the CDC investigators reported (MMWR. 2018 Jan 5;66[51-2]:1402-7).
Based on data from 48 states, D.C., and New York City, the distribution of prevalence for the BMI categories in 2015 was 3.6% underweight, 45% normal weight, 25.8% overweight, and 25.6% obese, the investigators said.
The CDC analysis was based on natality data from the National Vital Statistics System. The standard birth certificate was revised in 2003 to include maternal height and prepregnancy weight, but only 38 jurisdictions were using it by 2011. By 2015, all states except Connecticut and New Jersey had adopted its use.
according to the Centers for Disease Control and Prevention.
The overall prevalence of normal prepregnancy weight declined from 47.3% to 45.1% over that period in 36 states, the District of Columbia, and New York City, which reports natality data separately from New York state. The decreases were statistically significant in 26 states and New York City, the CDC investigators reported (MMWR. 2018 Jan 5;66[51-2]:1402-7).
Based on data from 48 states, D.C., and New York City, the distribution of prevalence for the BMI categories in 2015 was 3.6% underweight, 45% normal weight, 25.8% overweight, and 25.6% obese, the investigators said.
The CDC analysis was based on natality data from the National Vital Statistics System. The standard birth certificate was revised in 2003 to include maternal height and prepregnancy weight, but only 38 jurisdictions were using it by 2011. By 2015, all states except Connecticut and New Jersey had adopted its use.
FROM MMWR
FDA bans 24 ingredients from OTC health care antiseptic products
in hospital settings and other health care situations outside the hospital, the U.S. Food and Drug Administration announced in a final rule.
The affected products include health care personnel hand washes and hand rubs, surgical hand scrubs and hand rubs, and patient antiseptic skin preparations. The final rule was published Dec. 20 in the Federal Register and becomes effective in December 2018.
The agency determined that a deferral is warranted for six health care antiseptic active ingredients – benzalkonium chloride, benzethonium chloride, chloroxylenol, alcohol, isopropyl alcohol, and povidone-iodine – to allow more time for interested parties to complete the studies necessary to fill the safety and effectiveness data gaps identified for these ingredients.
“The FDA expects that this information may help better inform us on antiseptic resistance and antibiotic cross-resistance in the health care setting,” FDA Commissioner Scott Gottlieb, MD, said in a statement. “Importantly, this doesn’t mean that products containing these six ingredients are ineffective or unsafe. These antiseptic products remain an important resource in health care settings. Personnel should continue to use these products consistent with infection control guidelines while the additional data are gathered.”
No additional data was provided for another 24 products, which were deemed not generally recognized as safe and effective. The minimum data needed to demonstrate safety for all health care antiseptic active ingredients fall into four broad categories: human safety studies, nonclinical safety studies (developmental and reproductive toxicity studies and carcinogenicity studies), data to characterize potential hormonal effects, and data to evaluate the development of antimicrobial resistance, the final rule states.
The FDA noted that manufacturers started to remove nearly all of these 24 active ingredients from their products following a 2015 proposed rule. Triclosan is currently being used in available products.
The active ingredients affected are chlorhexidine gluconate; cloflucarban; fluorosalan; hexachlorophene; hexylresorcinol; iodophors (iodine-containing ingredients including iodine complex [ammonium ether sulfate and polyoxyethylene sorbitan monolaurate], iodine complex [phosphate ester of alkylaryloxy polyethylene glycol], iodine tincture USP, iodine topical solution USP, nonylphenoxypoly [ethyleneoxy] ethanoliodine, poloxamer–iodine complex, undecoylium chloride iodine complex); mercufenol chloride; methylbenzethonium chloride; phenol; secondary amyltricresols; sodium oxychlorosene; tribromsalan; triclocarban; triclosan; triple dye; combination of calomel, oxyquinoline benzoate, triethanolamine, and phenol derivative; and combination of mercufenol chloride and secondary amyltricresols in 50% alcohol.
If manufacturers want to use one or more of these 24 active ingredients in future OTC health care antiseptic drug products, those products will be considered new drugs for which a new drug application approval will be required, the agency said.
The rule does not affect health care antiseptics that are currently marketed under new drug applications and abbreviated new drug applications.
FDA’s action follows a similar final rule published Sept. 6, 2016, which removed triclosan and 18 other active ingredients from consumer antiseptic products.
in hospital settings and other health care situations outside the hospital, the U.S. Food and Drug Administration announced in a final rule.
The affected products include health care personnel hand washes and hand rubs, surgical hand scrubs and hand rubs, and patient antiseptic skin preparations. The final rule was published Dec. 20 in the Federal Register and becomes effective in December 2018.
The agency determined that a deferral is warranted for six health care antiseptic active ingredients – benzalkonium chloride, benzethonium chloride, chloroxylenol, alcohol, isopropyl alcohol, and povidone-iodine – to allow more time for interested parties to complete the studies necessary to fill the safety and effectiveness data gaps identified for these ingredients.
“The FDA expects that this information may help better inform us on antiseptic resistance and antibiotic cross-resistance in the health care setting,” FDA Commissioner Scott Gottlieb, MD, said in a statement. “Importantly, this doesn’t mean that products containing these six ingredients are ineffective or unsafe. These antiseptic products remain an important resource in health care settings. Personnel should continue to use these products consistent with infection control guidelines while the additional data are gathered.”
No additional data was provided for another 24 products, which were deemed not generally recognized as safe and effective. The minimum data needed to demonstrate safety for all health care antiseptic active ingredients fall into four broad categories: human safety studies, nonclinical safety studies (developmental and reproductive toxicity studies and carcinogenicity studies), data to characterize potential hormonal effects, and data to evaluate the development of antimicrobial resistance, the final rule states.
The FDA noted that manufacturers started to remove nearly all of these 24 active ingredients from their products following a 2015 proposed rule. Triclosan is currently being used in available products.
The active ingredients affected are chlorhexidine gluconate; cloflucarban; fluorosalan; hexachlorophene; hexylresorcinol; iodophors (iodine-containing ingredients including iodine complex [ammonium ether sulfate and polyoxyethylene sorbitan monolaurate], iodine complex [phosphate ester of alkylaryloxy polyethylene glycol], iodine tincture USP, iodine topical solution USP, nonylphenoxypoly [ethyleneoxy] ethanoliodine, poloxamer–iodine complex, undecoylium chloride iodine complex); mercufenol chloride; methylbenzethonium chloride; phenol; secondary amyltricresols; sodium oxychlorosene; tribromsalan; triclocarban; triclosan; triple dye; combination of calomel, oxyquinoline benzoate, triethanolamine, and phenol derivative; and combination of mercufenol chloride and secondary amyltricresols in 50% alcohol.
If manufacturers want to use one or more of these 24 active ingredients in future OTC health care antiseptic drug products, those products will be considered new drugs for which a new drug application approval will be required, the agency said.
The rule does not affect health care antiseptics that are currently marketed under new drug applications and abbreviated new drug applications.
FDA’s action follows a similar final rule published Sept. 6, 2016, which removed triclosan and 18 other active ingredients from consumer antiseptic products.
in hospital settings and other health care situations outside the hospital, the U.S. Food and Drug Administration announced in a final rule.
The affected products include health care personnel hand washes and hand rubs, surgical hand scrubs and hand rubs, and patient antiseptic skin preparations. The final rule was published Dec. 20 in the Federal Register and becomes effective in December 2018.
The agency determined that a deferral is warranted for six health care antiseptic active ingredients – benzalkonium chloride, benzethonium chloride, chloroxylenol, alcohol, isopropyl alcohol, and povidone-iodine – to allow more time for interested parties to complete the studies necessary to fill the safety and effectiveness data gaps identified for these ingredients.
“The FDA expects that this information may help better inform us on antiseptic resistance and antibiotic cross-resistance in the health care setting,” FDA Commissioner Scott Gottlieb, MD, said in a statement. “Importantly, this doesn’t mean that products containing these six ingredients are ineffective or unsafe. These antiseptic products remain an important resource in health care settings. Personnel should continue to use these products consistent with infection control guidelines while the additional data are gathered.”
No additional data was provided for another 24 products, which were deemed not generally recognized as safe and effective. The minimum data needed to demonstrate safety for all health care antiseptic active ingredients fall into four broad categories: human safety studies, nonclinical safety studies (developmental and reproductive toxicity studies and carcinogenicity studies), data to characterize potential hormonal effects, and data to evaluate the development of antimicrobial resistance, the final rule states.
The FDA noted that manufacturers started to remove nearly all of these 24 active ingredients from their products following a 2015 proposed rule. Triclosan is currently being used in available products.
The active ingredients affected are chlorhexidine gluconate; cloflucarban; fluorosalan; hexachlorophene; hexylresorcinol; iodophors (iodine-containing ingredients including iodine complex [ammonium ether sulfate and polyoxyethylene sorbitan monolaurate], iodine complex [phosphate ester of alkylaryloxy polyethylene glycol], iodine tincture USP, iodine topical solution USP, nonylphenoxypoly [ethyleneoxy] ethanoliodine, poloxamer–iodine complex, undecoylium chloride iodine complex); mercufenol chloride; methylbenzethonium chloride; phenol; secondary amyltricresols; sodium oxychlorosene; tribromsalan; triclocarban; triclosan; triple dye; combination of calomel, oxyquinoline benzoate, triethanolamine, and phenol derivative; and combination of mercufenol chloride and secondary amyltricresols in 50% alcohol.
If manufacturers want to use one or more of these 24 active ingredients in future OTC health care antiseptic drug products, those products will be considered new drugs for which a new drug application approval will be required, the agency said.
The rule does not affect health care antiseptics that are currently marketed under new drug applications and abbreviated new drug applications.
FDA’s action follows a similar final rule published Sept. 6, 2016, which removed triclosan and 18 other active ingredients from consumer antiseptic products.
FDA approves angiotensin II for shock patients
Angiotensin II has been approved for use in intravenous infusions to increase blood pressure in adults with septic or other distributive shock, the Food and Drug Administration announced.
Shock-related drops in blood pressure can restrict blood flow to vital organs and can result in organ failure and death. “There is a need for treatment options for critically ill hypotensive patients who do not adequately respond to available therapies,” Norman Stockbridge, MD, PhD, director of the division of cardiovascular and renal products in the FDA’s Center for Drug Evaluation and Research, said in a written statement.
The effectiveness of angiotensin II for treating critically low blood pressure was confirmed in a clinical trial of 321 patients who were in shock. A significant number of patients responded to angiotensin II treatment, compared with those given placebo. In combination with conventional treatments, angiotensin II increased blood pressure safely and effectively, according to the FDA statement.
Angiotensin II can cause serious blood clots, and prophylactic treatment for such issues is recommended.
The application for angiontensin II was received under Priority Review. The goal of Priority Review is for the FDA to take action on the application within 6 months if the agency determines that an approved drug would improve the safety and effectiveness of treating a serious medical condition.
Angiotensin II injections will be marketed as Giapreza by La Jolla Pharmaceutical Company.
Angiotensin II has been approved for use in intravenous infusions to increase blood pressure in adults with septic or other distributive shock, the Food and Drug Administration announced.
Shock-related drops in blood pressure can restrict blood flow to vital organs and can result in organ failure and death. “There is a need for treatment options for critically ill hypotensive patients who do not adequately respond to available therapies,” Norman Stockbridge, MD, PhD, director of the division of cardiovascular and renal products in the FDA’s Center for Drug Evaluation and Research, said in a written statement.
The effectiveness of angiotensin II for treating critically low blood pressure was confirmed in a clinical trial of 321 patients who were in shock. A significant number of patients responded to angiotensin II treatment, compared with those given placebo. In combination with conventional treatments, angiotensin II increased blood pressure safely and effectively, according to the FDA statement.
Angiotensin II can cause serious blood clots, and prophylactic treatment for such issues is recommended.
The application for angiontensin II was received under Priority Review. The goal of Priority Review is for the FDA to take action on the application within 6 months if the agency determines that an approved drug would improve the safety and effectiveness of treating a serious medical condition.
Angiotensin II injections will be marketed as Giapreza by La Jolla Pharmaceutical Company.
Angiotensin II has been approved for use in intravenous infusions to increase blood pressure in adults with septic or other distributive shock, the Food and Drug Administration announced.
Shock-related drops in blood pressure can restrict blood flow to vital organs and can result in organ failure and death. “There is a need for treatment options for critically ill hypotensive patients who do not adequately respond to available therapies,” Norman Stockbridge, MD, PhD, director of the division of cardiovascular and renal products in the FDA’s Center for Drug Evaluation and Research, said in a written statement.
The effectiveness of angiotensin II for treating critically low blood pressure was confirmed in a clinical trial of 321 patients who were in shock. A significant number of patients responded to angiotensin II treatment, compared with those given placebo. In combination with conventional treatments, angiotensin II increased blood pressure safely and effectively, according to the FDA statement.
Angiotensin II can cause serious blood clots, and prophylactic treatment for such issues is recommended.
The application for angiontensin II was received under Priority Review. The goal of Priority Review is for the FDA to take action on the application within 6 months if the agency determines that an approved drug would improve the safety and effectiveness of treating a serious medical condition.
Angiotensin II injections will be marketed as Giapreza by La Jolla Pharmaceutical Company.