User login
Psychiatric issues common among hepatitis C inpatients
Adult inpatients with hepatitis C are much more likely to have mental health comorbidities, compared with those who do not have hepatitis C, according to the Agency for Healthcare Research and Quality.
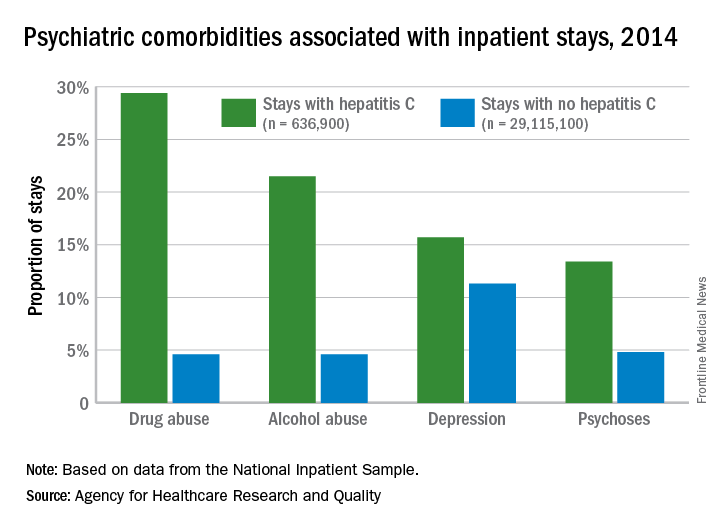
All four comorbidities skewed younger, and the oldest patients (73 years and older) with hepatitis C presented with each condition at about the same rate as the non–hepatitis C population. The proportions of hepatitis C–related inpatient stays with alcohol abuse by age, for example, were 20.5% for 18-51 years, 23.3% for those aged 52-72, and 5.8% for the 73-and-older group, according to data from the National Inpatient Sample, which includes more than 95% of all discharges from community (short-term, nonfederal, nonrehabilitation) hospitals in the United States.
Adult inpatients with hepatitis C are much more likely to have mental health comorbidities, compared with those who do not have hepatitis C, according to the Agency for Healthcare Research and Quality.

All four comorbidities skewed younger, and the oldest patients (73 years and older) with hepatitis C presented with each condition at about the same rate as the non–hepatitis C population. The proportions of hepatitis C–related inpatient stays with alcohol abuse by age, for example, were 20.5% for 18-51 years, 23.3% for those aged 52-72, and 5.8% for the 73-and-older group, according to data from the National Inpatient Sample, which includes more than 95% of all discharges from community (short-term, nonfederal, nonrehabilitation) hospitals in the United States.
Adult inpatients with hepatitis C are much more likely to have mental health comorbidities, compared with those who do not have hepatitis C, according to the Agency for Healthcare Research and Quality.

All four comorbidities skewed younger, and the oldest patients (73 years and older) with hepatitis C presented with each condition at about the same rate as the non–hepatitis C population. The proportions of hepatitis C–related inpatient stays with alcohol abuse by age, for example, were 20.5% for 18-51 years, 23.3% for those aged 52-72, and 5.8% for the 73-and-older group, according to data from the National Inpatient Sample, which includes more than 95% of all discharges from community (short-term, nonfederal, nonrehabilitation) hospitals in the United States.
Drugs identified after suicide deaths
Antidepressants and alcohol were each identified in more than 40% of tests performed after suicide deaths in 2014, according to data from the Centers for Disease Control and Prevention’s National Violent Death Reporting System.
Of the 14,834 suicide deaths reported that year in the system’s 18 participating states, tests for alcohol – conducted for 53% (7,883) of decedents – were the most commonly performed and were the second most likely to be positive among drugs with data available: The rate was 40.2%. Among the tests for blood alcohol concentration, a level of 0.08 g/dL or higher, which is over the legal limit in all states, was seen in almost 70% of positive results, Katherine A. Fowler, PhD, and her associates at the CDC’s National Center for Injury Prevention and Control reported in Morbidity and Mortality Weekly Report Surveillance Summaries.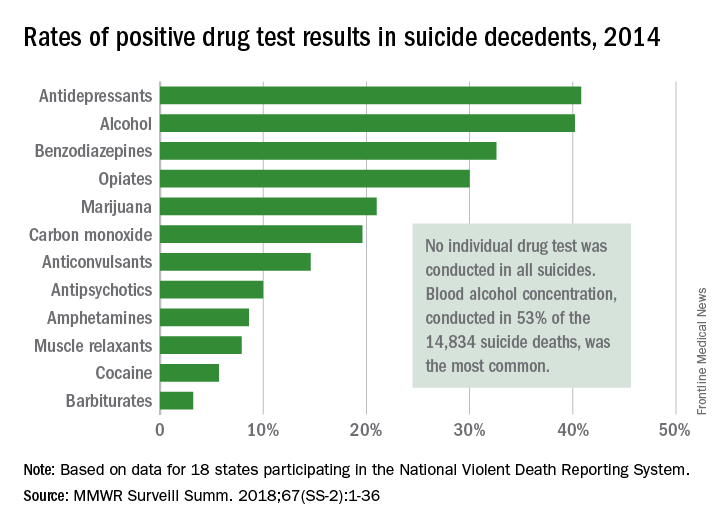
The 18 states that collected statewide data for 2014 – Alaska, Colorado, Georgia, Kentucky, Maryland, Massachusetts, Michigan, New Jersey, New Mexico, North Carolina, Ohio, Oklahoma, Oregon, Rhode Island, South Carolina, Utah, Virginia, and Wisconsin – represent just over 33% of the U.S. population.
SOURCE: Fowler KA et al. MMWR Surveill Summ. 2018;67(SS-2):1-36. doi: 10.15585/mmwr.ss6702a1.
Antidepressants and alcohol were each identified in more than 40% of tests performed after suicide deaths in 2014, according to data from the Centers for Disease Control and Prevention’s National Violent Death Reporting System.
Of the 14,834 suicide deaths reported that year in the system’s 18 participating states, tests for alcohol – conducted for 53% (7,883) of decedents – were the most commonly performed and were the second most likely to be positive among drugs with data available: The rate was 40.2%. Among the tests for blood alcohol concentration, a level of 0.08 g/dL or higher, which is over the legal limit in all states, was seen in almost 70% of positive results, Katherine A. Fowler, PhD, and her associates at the CDC’s National Center for Injury Prevention and Control reported in Morbidity and Mortality Weekly Report Surveillance Summaries.
The 18 states that collected statewide data for 2014 – Alaska, Colorado, Georgia, Kentucky, Maryland, Massachusetts, Michigan, New Jersey, New Mexico, North Carolina, Ohio, Oklahoma, Oregon, Rhode Island, South Carolina, Utah, Virginia, and Wisconsin – represent just over 33% of the U.S. population.
SOURCE: Fowler KA et al. MMWR Surveill Summ. 2018;67(SS-2):1-36. doi: 10.15585/mmwr.ss6702a1.
Antidepressants and alcohol were each identified in more than 40% of tests performed after suicide deaths in 2014, according to data from the Centers for Disease Control and Prevention’s National Violent Death Reporting System.
Of the 14,834 suicide deaths reported that year in the system’s 18 participating states, tests for alcohol – conducted for 53% (7,883) of decedents – were the most commonly performed and were the second most likely to be positive among drugs with data available: The rate was 40.2%. Among the tests for blood alcohol concentration, a level of 0.08 g/dL or higher, which is over the legal limit in all states, was seen in almost 70% of positive results, Katherine A. Fowler, PhD, and her associates at the CDC’s National Center for Injury Prevention and Control reported in Morbidity and Mortality Weekly Report Surveillance Summaries.
The 18 states that collected statewide data for 2014 – Alaska, Colorado, Georgia, Kentucky, Maryland, Massachusetts, Michigan, New Jersey, New Mexico, North Carolina, Ohio, Oklahoma, Oregon, Rhode Island, South Carolina, Utah, Virginia, and Wisconsin – represent just over 33% of the U.S. population.
SOURCE: Fowler KA et al. MMWR Surveill Summ. 2018;67(SS-2):1-36. doi: 10.15585/mmwr.ss6702a1.
FROM MMWR
Drug combo indicated for bacterial pneumonia
(Avycaz) to include hospital-acquired bacterial pneumonia and ventilator-associated bacterial pneumonia (HABP/VABP) in adults.
Specifically, the approved indication is for infections caused by certain Gram-negative bacteria – some of which are increasingly resistant to available antibiotics – including, Klebsiella pneumoniae, Enterobacter cloacae, Escherichia coli, Serratia marcescens, Proteus mirabilis, Pseudomonas aeruginosa, and Haemophilus influenzae.
There have not been new treatment options for HABP/VABP caused by Gram-negative bacteria in more than 15 years, according to Allergan, the drug’s manufacturer.
This is the third approved indication for ceftazidime/avibactam; the other two indications are for complicated intra-abdominal infections (in combination with metronidazole) and for complicated urinary tract infections.
(Avycaz) to include hospital-acquired bacterial pneumonia and ventilator-associated bacterial pneumonia (HABP/VABP) in adults.
Specifically, the approved indication is for infections caused by certain Gram-negative bacteria – some of which are increasingly resistant to available antibiotics – including, Klebsiella pneumoniae, Enterobacter cloacae, Escherichia coli, Serratia marcescens, Proteus mirabilis, Pseudomonas aeruginosa, and Haemophilus influenzae.
There have not been new treatment options for HABP/VABP caused by Gram-negative bacteria in more than 15 years, according to Allergan, the drug’s manufacturer.
This is the third approved indication for ceftazidime/avibactam; the other two indications are for complicated intra-abdominal infections (in combination with metronidazole) and for complicated urinary tract infections.
(Avycaz) to include hospital-acquired bacterial pneumonia and ventilator-associated bacterial pneumonia (HABP/VABP) in adults.
Specifically, the approved indication is for infections caused by certain Gram-negative bacteria – some of which are increasingly resistant to available antibiotics – including, Klebsiella pneumoniae, Enterobacter cloacae, Escherichia coli, Serratia marcescens, Proteus mirabilis, Pseudomonas aeruginosa, and Haemophilus influenzae.
There have not been new treatment options for HABP/VABP caused by Gram-negative bacteria in more than 15 years, according to Allergan, the drug’s manufacturer.
This is the third approved indication for ceftazidime/avibactam; the other two indications are for complicated intra-abdominal infections (in combination with metronidazole) and for complicated urinary tract infections.
Hospitals filling as flu season worsens
Through the last full week of January, the cumulative “hospitalization rate is the highest we’ve seen,” acting Centers for Disease Control and Prevention director Anne Schuchat, MD, said. For the current season so far, the hospitalization rate stands at 51.4 per 100,000 population, putting it on pace to top the total of 710,000 flu-related admissions that occurred during the 2014-2015 season, she said in a weekly briefing Feb. 2.
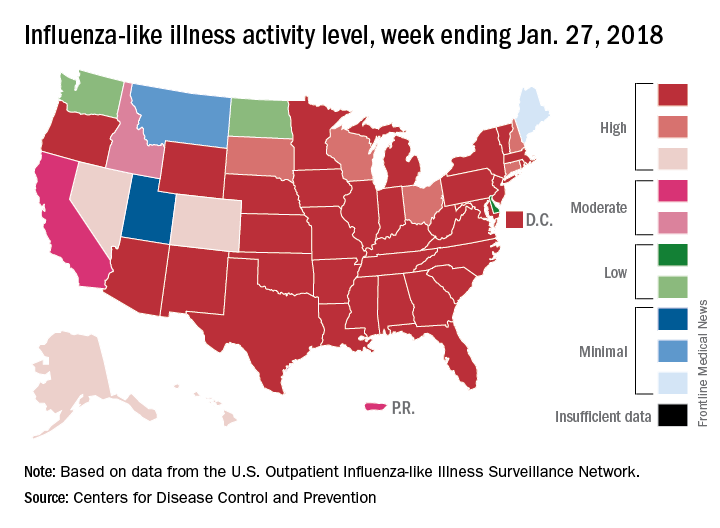
Flu-related pediatric deaths also took a big jump for the week as another 16 were reported, which brings the total for the season to 53. Of the children who have died so far, only 20% were vaccinated, said Dan Jernigan, MD, MPH, director of the influenza division at the CDC’s National Center for Immunization and Respiratory Diseases, Atlanta. He also noted that half of the children who have been hospitalized did not had an underlying condition.
The one bit of good news for the week was that activity in the West seems to be easing up, Dr. Schuchat said. The geographic spread of ILI was reported as widespread in 48 states, which is down from 49 the previous week because Oregon dropped off the list. To go along with that, the ILI activity level in California has dropped 2 weeks in a row and now stands at level 7, the CDC data show.
Through the last full week of January, the cumulative “hospitalization rate is the highest we’ve seen,” acting Centers for Disease Control and Prevention director Anne Schuchat, MD, said. For the current season so far, the hospitalization rate stands at 51.4 per 100,000 population, putting it on pace to top the total of 710,000 flu-related admissions that occurred during the 2014-2015 season, she said in a weekly briefing Feb. 2.

Flu-related pediatric deaths also took a big jump for the week as another 16 were reported, which brings the total for the season to 53. Of the children who have died so far, only 20% were vaccinated, said Dan Jernigan, MD, MPH, director of the influenza division at the CDC’s National Center for Immunization and Respiratory Diseases, Atlanta. He also noted that half of the children who have been hospitalized did not had an underlying condition.
The one bit of good news for the week was that activity in the West seems to be easing up, Dr. Schuchat said. The geographic spread of ILI was reported as widespread in 48 states, which is down from 49 the previous week because Oregon dropped off the list. To go along with that, the ILI activity level in California has dropped 2 weeks in a row and now stands at level 7, the CDC data show.
Through the last full week of January, the cumulative “hospitalization rate is the highest we’ve seen,” acting Centers for Disease Control and Prevention director Anne Schuchat, MD, said. For the current season so far, the hospitalization rate stands at 51.4 per 100,000 population, putting it on pace to top the total of 710,000 flu-related admissions that occurred during the 2014-2015 season, she said in a weekly briefing Feb. 2.

Flu-related pediatric deaths also took a big jump for the week as another 16 were reported, which brings the total for the season to 53. Of the children who have died so far, only 20% were vaccinated, said Dan Jernigan, MD, MPH, director of the influenza division at the CDC’s National Center for Immunization and Respiratory Diseases, Atlanta. He also noted that half of the children who have been hospitalized did not had an underlying condition.
The one bit of good news for the week was that activity in the West seems to be easing up, Dr. Schuchat said. The geographic spread of ILI was reported as widespread in 48 states, which is down from 49 the previous week because Oregon dropped off the list. To go along with that, the ILI activity level in California has dropped 2 weeks in a row and now stands at level 7, the CDC data show.
FDA approves implantable therapy for PAH
to treat adult patients with New York Heart Association (NYHA) Class I, II and III pulmonary arterial hypertension.
This infusion system is implanted into a patient for intravenous delivery of treprostinil (Remodulin) and is designed to help supply blood to the lungs and keep a patient’s blood pressure within a healthy range. The system comprises three parts: the pump, the programmer, and the catheter.
The implant should not be used for patients with NYHA Class IV heart failure, a known or suspected infection, bacteremia, or sepsis requiring antibiotics; vasculature that is inadequate for an 8 French introducer or catheter advancement without stylet guidance; implanted leads or catheters (active or abandoned) in the superior vena cava that cannot be removed prior to or at system implant; a body size not sufficient to accept the pump; or skin or soft tissue that would heal poorly or increase susceptibility to infections. Patients who are unable to tolerate a sudden cessation of treprostinil therapy also would not be able to receive the implantable device.
Read the full approval on the FDA’s website.
to treat adult patients with New York Heart Association (NYHA) Class I, II and III pulmonary arterial hypertension.
This infusion system is implanted into a patient for intravenous delivery of treprostinil (Remodulin) and is designed to help supply blood to the lungs and keep a patient’s blood pressure within a healthy range. The system comprises three parts: the pump, the programmer, and the catheter.
The implant should not be used for patients with NYHA Class IV heart failure, a known or suspected infection, bacteremia, or sepsis requiring antibiotics; vasculature that is inadequate for an 8 French introducer or catheter advancement without stylet guidance; implanted leads or catheters (active or abandoned) in the superior vena cava that cannot be removed prior to or at system implant; a body size not sufficient to accept the pump; or skin or soft tissue that would heal poorly or increase susceptibility to infections. Patients who are unable to tolerate a sudden cessation of treprostinil therapy also would not be able to receive the implantable device.
Read the full approval on the FDA’s website.
to treat adult patients with New York Heart Association (NYHA) Class I, II and III pulmonary arterial hypertension.
This infusion system is implanted into a patient for intravenous delivery of treprostinil (Remodulin) and is designed to help supply blood to the lungs and keep a patient’s blood pressure within a healthy range. The system comprises three parts: the pump, the programmer, and the catheter.
The implant should not be used for patients with NYHA Class IV heart failure, a known or suspected infection, bacteremia, or sepsis requiring antibiotics; vasculature that is inadequate for an 8 French introducer or catheter advancement without stylet guidance; implanted leads or catheters (active or abandoned) in the superior vena cava that cannot be removed prior to or at system implant; a body size not sufficient to accept the pump; or skin or soft tissue that would heal poorly or increase susceptibility to infections. Patients who are unable to tolerate a sudden cessation of treprostinil therapy also would not be able to receive the implantable device.
Read the full approval on the FDA’s website.
Baby boomers are the hepatitis C generation
Increases in hepatitis C–related inpatient stays for baby boomers from 2005 to 2014 far outpaced those of older adults, while younger adults saw their admissions drop over that period, according to the Agency for Healthcare Research and Quality.
For the baby boomers (adults aged 52-72 years), the rate of inpatient stays involving hepatitis C with or without hepatitis B, HIV, or alcoholic liver disease rose from 300.7 per 100,000 population in 2005 to 503.1 per 100,000 in 2014 – an increase of over 67%. For patients aged 73 years and older, that rate went from 104.4 in 2005 to 117.1 in 2014, which translates to a 12% increase, and for patients aged 18-51 years, it dropped 15%, from 182.5 to 155.4, the AHRQ said in a statistical brief.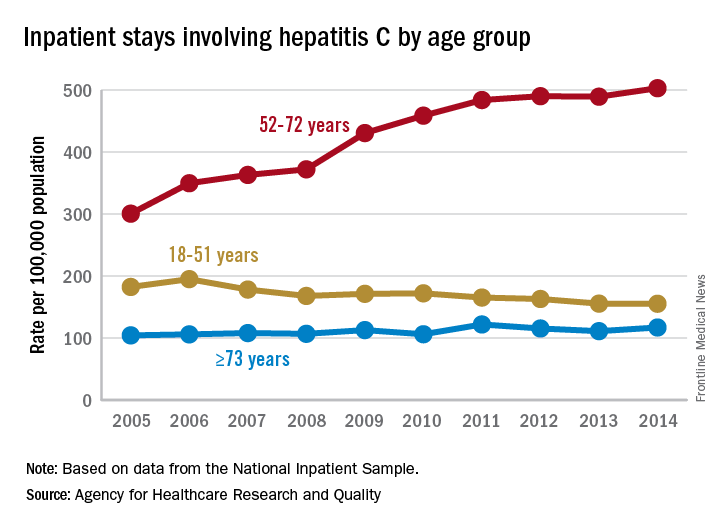
Along with the increased hospitalizations, “acute hepatitis C cases nearly tripled from 2010 through 2015,” the report noted, which was “likely the result of increasing injection drug use due to the growing opioid epidemic.”
Increases in hepatitis C–related inpatient stays for baby boomers from 2005 to 2014 far outpaced those of older adults, while younger adults saw their admissions drop over that period, according to the Agency for Healthcare Research and Quality.
For the baby boomers (adults aged 52-72 years), the rate of inpatient stays involving hepatitis C with or without hepatitis B, HIV, or alcoholic liver disease rose from 300.7 per 100,000 population in 2005 to 503.1 per 100,000 in 2014 – an increase of over 67%. For patients aged 73 years and older, that rate went from 104.4 in 2005 to 117.1 in 2014, which translates to a 12% increase, and for patients aged 18-51 years, it dropped 15%, from 182.5 to 155.4, the AHRQ said in a statistical brief.
Along with the increased hospitalizations, “acute hepatitis C cases nearly tripled from 2010 through 2015,” the report noted, which was “likely the result of increasing injection drug use due to the growing opioid epidemic.”
Increases in hepatitis C–related inpatient stays for baby boomers from 2005 to 2014 far outpaced those of older adults, while younger adults saw their admissions drop over that period, according to the Agency for Healthcare Research and Quality.
For the baby boomers (adults aged 52-72 years), the rate of inpatient stays involving hepatitis C with or without hepatitis B, HIV, or alcoholic liver disease rose from 300.7 per 100,000 population in 2005 to 503.1 per 100,000 in 2014 – an increase of over 67%. For patients aged 73 years and older, that rate went from 104.4 in 2005 to 117.1 in 2014, which translates to a 12% increase, and for patients aged 18-51 years, it dropped 15%, from 182.5 to 155.4, the AHRQ said in a statistical brief.
Along with the increased hospitalizations, “acute hepatitis C cases nearly tripled from 2010 through 2015,” the report noted, which was “likely the result of increasing injection drug use due to the growing opioid epidemic.”
FDA issues safety alert for loperamide
The Food and Drug Administration announced Jan. 30 that is has issued a MedWatch safety alert on the use of the over-the-counter (OTC) antidiarrhea drug, loperamide.
Currently, the FDA is working with manufacturers to use blister packs or other single-dose packaging and to limit the number of doses in a package.
The alert comes after receiving continuous reports of serious heart problems and deaths with the use of much higher than recommended doses of loperamide, mainly among people who are intentionally misusing or abusing the product, regardless of the addition of a warning to the medicine label and a previous communication. The FDA states that loperamide is a safe drug when used as directed.
Loperamide is approved to help control symptoms of diarrhea. The maximum recommended daily dose for adults is 8 mg per day for OTC use and 16 mg per day for prescription use. It acts on opioid receptors in the gut to slow the movement in the intestines and decrease the number of bowel movements.
It is noted that much higher than recommended doses of loperamide, either intentionally or unintentionally, can result in serious cardiac adverse events, including QT interval prolongation, torsade de pointes or other ventricular arrhythmias, syncope, and cardiac arrest. Health care professionals and patients can report adverse events or side effects related to the use of these products to the FDA’s MedWatch Safety Information and Adverse Event Reporting Program.
In 2016, the FDA issued a Drug Safety Communication and added warnings about serious heart problems to the drug label of prescription loperamide and to the Drug Facts label of OTC loperamide products. The FDA is working to evaluate this safety issue and will update the public when more information is available.
Read the full safety alert here.
The Food and Drug Administration announced Jan. 30 that is has issued a MedWatch safety alert on the use of the over-the-counter (OTC) antidiarrhea drug, loperamide.
Currently, the FDA is working with manufacturers to use blister packs or other single-dose packaging and to limit the number of doses in a package.
The alert comes after receiving continuous reports of serious heart problems and deaths with the use of much higher than recommended doses of loperamide, mainly among people who are intentionally misusing or abusing the product, regardless of the addition of a warning to the medicine label and a previous communication. The FDA states that loperamide is a safe drug when used as directed.
Loperamide is approved to help control symptoms of diarrhea. The maximum recommended daily dose for adults is 8 mg per day for OTC use and 16 mg per day for prescription use. It acts on opioid receptors in the gut to slow the movement in the intestines and decrease the number of bowel movements.
It is noted that much higher than recommended doses of loperamide, either intentionally or unintentionally, can result in serious cardiac adverse events, including QT interval prolongation, torsade de pointes or other ventricular arrhythmias, syncope, and cardiac arrest. Health care professionals and patients can report adverse events or side effects related to the use of these products to the FDA’s MedWatch Safety Information and Adverse Event Reporting Program.
In 2016, the FDA issued a Drug Safety Communication and added warnings about serious heart problems to the drug label of prescription loperamide and to the Drug Facts label of OTC loperamide products. The FDA is working to evaluate this safety issue and will update the public when more information is available.
Read the full safety alert here.
The Food and Drug Administration announced Jan. 30 that is has issued a MedWatch safety alert on the use of the over-the-counter (OTC) antidiarrhea drug, loperamide.
Currently, the FDA is working with manufacturers to use blister packs or other single-dose packaging and to limit the number of doses in a package.
The alert comes after receiving continuous reports of serious heart problems and deaths with the use of much higher than recommended doses of loperamide, mainly among people who are intentionally misusing or abusing the product, regardless of the addition of a warning to the medicine label and a previous communication. The FDA states that loperamide is a safe drug when used as directed.
Loperamide is approved to help control symptoms of diarrhea. The maximum recommended daily dose for adults is 8 mg per day for OTC use and 16 mg per day for prescription use. It acts on opioid receptors in the gut to slow the movement in the intestines and decrease the number of bowel movements.
It is noted that much higher than recommended doses of loperamide, either intentionally or unintentionally, can result in serious cardiac adverse events, including QT interval prolongation, torsade de pointes or other ventricular arrhythmias, syncope, and cardiac arrest. Health care professionals and patients can report adverse events or side effects related to the use of these products to the FDA’s MedWatch Safety Information and Adverse Event Reporting Program.
In 2016, the FDA issued a Drug Safety Communication and added warnings about serious heart problems to the drug label of prescription loperamide and to the Drug Facts label of OTC loperamide products. The FDA is working to evaluate this safety issue and will update the public when more information is available.
Read the full safety alert here.
Birth cohort affected 2015-2016 flu vaccine effectiveness
The influenza vaccine introduced in 2009 showed reduced effectiveness during the 2015-2016 influenza season, but only in adults born between 1958 and 1979, according to an analysis published online in the Journal of Infectious Diseases.
Using the Influenza Vaccine Effectiveness Network, researchers analyzed data from 2,115 patients with medically attended acute respiratory illness who tested positive for A(H1N1)pdm09 influenza virus, and 14,696 patients who tested negative for the influenza virus, from 2010-2011 to 2015-2016 (excluding the 2014-2015 influenza season).
Overall, 48% of the influenza virus–negative patients and 28% of the virus-positive patients had received at least one dose of the seasonal inactivated influenza vaccine more than 2 weeks before they fell ill.
However, the vaccine, which was based on the A/California/07/2009 strain of the A(H1N1)pdm09 virus, was only 47% effective during the 2015-2016 season, compared with 61% effectiveness during the 2010-2011 season through to the 2013-2014 season.
When researchers looked at vaccine effectiveness by birth cohort, they found that one particular cohort – individuals born between 1958 and 1979 – showed a significantly reduced vaccine effectiveness (22%) during the 2015-2016 season. By comparison, vaccine effectiveness in this cohort was 61% during the 2010-2013 seasons, and 56% during the 2013-2014 season.
When this birth cohort was excluded from analysis of the 2015-2016 season, the overall vaccine effectiveness for that season was 61%.
While the vaccine was based on an early reference strain of A(H1N1)pdm09, the virus itself later acquired mutations in the hemagglutinin gene, leading to the emergence of new genetic clades, including 6B, which dominated in the 2013-2014 influenza season, and 6B.1, which dominated in 2015-2016.
“Limited serologic data suggest that some adults born during 1958-1979 (age range in 2015-2016, 36-57 years) have decreased antibody titers against A(H1N1)pdm09 group 6B and 6B.1 viruses,” wrote Brendan Flannery, PhD, from the Centers for Disease Control and Prevention, and his coauthors.
They suggested that individuals in this cohort may have been immunologically primed with A/USSR/90/1977-like viruses, which were the first group of A(H1N1) viruses that this cohort would have been exposed to. A(H1N1) strains didn’t circulate between 1958 and 1977. Vaccination with A(H1N1)pdm09 viruses may have induced antibodies against shared antigenic components found on early versions of A(H1N1)pdm09.
If these shared antigenic epitopes were then altered in the later 6B and 6B.1 viruses, that might account for decreased antibody titers in this age group.
“Replacement of the A/California/07/2009(H1N1)pdm09 vaccine reference strain with A/Michigan/45/2015 (group 6B.1) should lead to improved [vaccine effectiveness] against circulating A(H1N1)pdm09 viruses,” the investigators noted.
The study was supported by the Centers for Disease Control and Prevention, the National Institutes of Health, and the National Center for Advancing Translational Sciences. Eight authors declared funding, grants, and consultancies with the pharmaceutical industry, with five also declaring funding from the CDC.
SOURCE: Flannery B et al. J Infect Dis. 2018 Jan 18. doi: 10.1093/infdis/jix634.
This study proposes that influenza virus strains encountered early in life focus the immune response to later infection or vaccination on shared epitopes between the early and later strains. Supporting this hypothesis is evidence from other studies showing that 60% of the serological response to inactivated influenza vaccines is the result of boosting pre-existing antibodies, rather than the creation of new, vaccine-induced antibodies.
However there are also some flaws to this argument, and we should be careful to avoid confirmation bias. For example, the reduction in effectiveness of vaccines against A(H1N1) has been observed in North America, where this study is located, but to a lesser extent in studies conducted in other regions. Reductions in vaccine effectiveness have also been observed in other birth cohorts and during other influenza seasons.
That aside, accumulating evidence suggests that the vaccine strain be updated from A/California/7/2009 to A/Michigan/45/2015 (a clade 6B.1 strain) for the 2016-2017 influenza seasons.
Allen C. Cheng, PhD, is from the School of Public Health and Preventive Medicine at Monash University, Melbourne, and Kanta Subbarao, MBBS, is from the World Health Organization Collaborating Centre for Reference and Research on Influenza and the Peter Doherty Institute for Infection and Immunity, Australia. These comments are taken from an accompanying editorial (J Infect Dis. 2018, Jan 18. doi: 10.1093/infdis/jix635). The authors declared support from the Australian Department of Health and the Australian National Health and Medical Research Council. No conflicts of interest were declared.
This study proposes that influenza virus strains encountered early in life focus the immune response to later infection or vaccination on shared epitopes between the early and later strains. Supporting this hypothesis is evidence from other studies showing that 60% of the serological response to inactivated influenza vaccines is the result of boosting pre-existing antibodies, rather than the creation of new, vaccine-induced antibodies.
However there are also some flaws to this argument, and we should be careful to avoid confirmation bias. For example, the reduction in effectiveness of vaccines against A(H1N1) has been observed in North America, where this study is located, but to a lesser extent in studies conducted in other regions. Reductions in vaccine effectiveness have also been observed in other birth cohorts and during other influenza seasons.
That aside, accumulating evidence suggests that the vaccine strain be updated from A/California/7/2009 to A/Michigan/45/2015 (a clade 6B.1 strain) for the 2016-2017 influenza seasons.
Allen C. Cheng, PhD, is from the School of Public Health and Preventive Medicine at Monash University, Melbourne, and Kanta Subbarao, MBBS, is from the World Health Organization Collaborating Centre for Reference and Research on Influenza and the Peter Doherty Institute for Infection and Immunity, Australia. These comments are taken from an accompanying editorial (J Infect Dis. 2018, Jan 18. doi: 10.1093/infdis/jix635). The authors declared support from the Australian Department of Health and the Australian National Health and Medical Research Council. No conflicts of interest were declared.
This study proposes that influenza virus strains encountered early in life focus the immune response to later infection or vaccination on shared epitopes between the early and later strains. Supporting this hypothesis is evidence from other studies showing that 60% of the serological response to inactivated influenza vaccines is the result of boosting pre-existing antibodies, rather than the creation of new, vaccine-induced antibodies.
However there are also some flaws to this argument, and we should be careful to avoid confirmation bias. For example, the reduction in effectiveness of vaccines against A(H1N1) has been observed in North America, where this study is located, but to a lesser extent in studies conducted in other regions. Reductions in vaccine effectiveness have also been observed in other birth cohorts and during other influenza seasons.
That aside, accumulating evidence suggests that the vaccine strain be updated from A/California/7/2009 to A/Michigan/45/2015 (a clade 6B.1 strain) for the 2016-2017 influenza seasons.
Allen C. Cheng, PhD, is from the School of Public Health and Preventive Medicine at Monash University, Melbourne, and Kanta Subbarao, MBBS, is from the World Health Organization Collaborating Centre for Reference and Research on Influenza and the Peter Doherty Institute for Infection and Immunity, Australia. These comments are taken from an accompanying editorial (J Infect Dis. 2018, Jan 18. doi: 10.1093/infdis/jix635). The authors declared support from the Australian Department of Health and the Australian National Health and Medical Research Council. No conflicts of interest were declared.
The influenza vaccine introduced in 2009 showed reduced effectiveness during the 2015-2016 influenza season, but only in adults born between 1958 and 1979, according to an analysis published online in the Journal of Infectious Diseases.
Using the Influenza Vaccine Effectiveness Network, researchers analyzed data from 2,115 patients with medically attended acute respiratory illness who tested positive for A(H1N1)pdm09 influenza virus, and 14,696 patients who tested negative for the influenza virus, from 2010-2011 to 2015-2016 (excluding the 2014-2015 influenza season).
Overall, 48% of the influenza virus–negative patients and 28% of the virus-positive patients had received at least one dose of the seasonal inactivated influenza vaccine more than 2 weeks before they fell ill.
However, the vaccine, which was based on the A/California/07/2009 strain of the A(H1N1)pdm09 virus, was only 47% effective during the 2015-2016 season, compared with 61% effectiveness during the 2010-2011 season through to the 2013-2014 season.
When researchers looked at vaccine effectiveness by birth cohort, they found that one particular cohort – individuals born between 1958 and 1979 – showed a significantly reduced vaccine effectiveness (22%) during the 2015-2016 season. By comparison, vaccine effectiveness in this cohort was 61% during the 2010-2013 seasons, and 56% during the 2013-2014 season.
When this birth cohort was excluded from analysis of the 2015-2016 season, the overall vaccine effectiveness for that season was 61%.
While the vaccine was based on an early reference strain of A(H1N1)pdm09, the virus itself later acquired mutations in the hemagglutinin gene, leading to the emergence of new genetic clades, including 6B, which dominated in the 2013-2014 influenza season, and 6B.1, which dominated in 2015-2016.
“Limited serologic data suggest that some adults born during 1958-1979 (age range in 2015-2016, 36-57 years) have decreased antibody titers against A(H1N1)pdm09 group 6B and 6B.1 viruses,” wrote Brendan Flannery, PhD, from the Centers for Disease Control and Prevention, and his coauthors.
They suggested that individuals in this cohort may have been immunologically primed with A/USSR/90/1977-like viruses, which were the first group of A(H1N1) viruses that this cohort would have been exposed to. A(H1N1) strains didn’t circulate between 1958 and 1977. Vaccination with A(H1N1)pdm09 viruses may have induced antibodies against shared antigenic components found on early versions of A(H1N1)pdm09.
If these shared antigenic epitopes were then altered in the later 6B and 6B.1 viruses, that might account for decreased antibody titers in this age group.
“Replacement of the A/California/07/2009(H1N1)pdm09 vaccine reference strain with A/Michigan/45/2015 (group 6B.1) should lead to improved [vaccine effectiveness] against circulating A(H1N1)pdm09 viruses,” the investigators noted.
The study was supported by the Centers for Disease Control and Prevention, the National Institutes of Health, and the National Center for Advancing Translational Sciences. Eight authors declared funding, grants, and consultancies with the pharmaceutical industry, with five also declaring funding from the CDC.
SOURCE: Flannery B et al. J Infect Dis. 2018 Jan 18. doi: 10.1093/infdis/jix634.
The influenza vaccine introduced in 2009 showed reduced effectiveness during the 2015-2016 influenza season, but only in adults born between 1958 and 1979, according to an analysis published online in the Journal of Infectious Diseases.
Using the Influenza Vaccine Effectiveness Network, researchers analyzed data from 2,115 patients with medically attended acute respiratory illness who tested positive for A(H1N1)pdm09 influenza virus, and 14,696 patients who tested negative for the influenza virus, from 2010-2011 to 2015-2016 (excluding the 2014-2015 influenza season).
Overall, 48% of the influenza virus–negative patients and 28% of the virus-positive patients had received at least one dose of the seasonal inactivated influenza vaccine more than 2 weeks before they fell ill.
However, the vaccine, which was based on the A/California/07/2009 strain of the A(H1N1)pdm09 virus, was only 47% effective during the 2015-2016 season, compared with 61% effectiveness during the 2010-2011 season through to the 2013-2014 season.
When researchers looked at vaccine effectiveness by birth cohort, they found that one particular cohort – individuals born between 1958 and 1979 – showed a significantly reduced vaccine effectiveness (22%) during the 2015-2016 season. By comparison, vaccine effectiveness in this cohort was 61% during the 2010-2013 seasons, and 56% during the 2013-2014 season.
When this birth cohort was excluded from analysis of the 2015-2016 season, the overall vaccine effectiveness for that season was 61%.
While the vaccine was based on an early reference strain of A(H1N1)pdm09, the virus itself later acquired mutations in the hemagglutinin gene, leading to the emergence of new genetic clades, including 6B, which dominated in the 2013-2014 influenza season, and 6B.1, which dominated in 2015-2016.
“Limited serologic data suggest that some adults born during 1958-1979 (age range in 2015-2016, 36-57 years) have decreased antibody titers against A(H1N1)pdm09 group 6B and 6B.1 viruses,” wrote Brendan Flannery, PhD, from the Centers for Disease Control and Prevention, and his coauthors.
They suggested that individuals in this cohort may have been immunologically primed with A/USSR/90/1977-like viruses, which were the first group of A(H1N1) viruses that this cohort would have been exposed to. A(H1N1) strains didn’t circulate between 1958 and 1977. Vaccination with A(H1N1)pdm09 viruses may have induced antibodies against shared antigenic components found on early versions of A(H1N1)pdm09.
If these shared antigenic epitopes were then altered in the later 6B and 6B.1 viruses, that might account for decreased antibody titers in this age group.
“Replacement of the A/California/07/2009(H1N1)pdm09 vaccine reference strain with A/Michigan/45/2015 (group 6B.1) should lead to improved [vaccine effectiveness] against circulating A(H1N1)pdm09 viruses,” the investigators noted.
The study was supported by the Centers for Disease Control and Prevention, the National Institutes of Health, and the National Center for Advancing Translational Sciences. Eight authors declared funding, grants, and consultancies with the pharmaceutical industry, with five also declaring funding from the CDC.
SOURCE: Flannery B et al. J Infect Dis. 2018 Jan 18. doi: 10.1093/infdis/jix634.
FROM THE JOURNAL OF INFECTIOUS DISEASES
Key clinical point:
Major finding: The influenza vaccine effectiveness during the 2015-2016 season was just 22% in individuals born between 1958 and 1979.
Data source: A retrospective case-control study of 2,115 patients who tested positive for A(H1N1)pdm09 influenza virus, and 14,696 negative controls.
Disclosures: The study was supported by the Centers for Disease Control and Prevention, the National Institutes of Health, and the National Center for Advancing Translational Sciences. Eight authors declared funding, grants, and consultancies with the pharmaceutical industry, with five also declaring funding from the CDC.
Source: Flannery B et al. J Infect Dis. 2018 Jan 18. doi: 10.1093/infdis/jix634.
CDC: Flu levels highest since pandemic year 2009
according to data from the Centers for Disease Control and Prevention.
That season was dominated by influenza A (H3N2), and the 2017-2018 season seems to be going down that same path. For the week ending Jan. 20, the proportion of outpatient visits for influenza-like illness increased to 6.6%, which is, for the second consecutive week, the highest level reported since October of – you guessed it – 2009, when it hit 7.7%, the CDC said in its weekly flu surveillance report.
The level reported last week, 6.3%, has been revised downward and now stands at an even 6%.
It turns out that 2018 is something of a milestone for the H3N2 virus. The virus first emerged in 1968, so it has reached its 50th anniversary, Dan Jernigan, MD, director of the influenza division at the CDC’s National Center for Immunization and Respiratory Diseases, Atlanta, said on Jan. 26 in a weekly briefing.
H3N2 must not be happy about hitting the big 5-0, however, because the map of influenza-like illness activity looks pretty red and angry. For the week ending Jan. 20, there were 30 states at the highest level of flu activity on the CDC’s 1-10 scale, with another nine in the “high” range at levels 8 and 9.
Dr. Jernigan did suggest that activity may have peaked in some areas of the country, with California among them.
There were seven pediatric deaths reported for the week ending Jan. 20, although six occurred in previous weeks. There have been 37 flu-related deaths among children so far during the 2017-2018 season, the CDC said.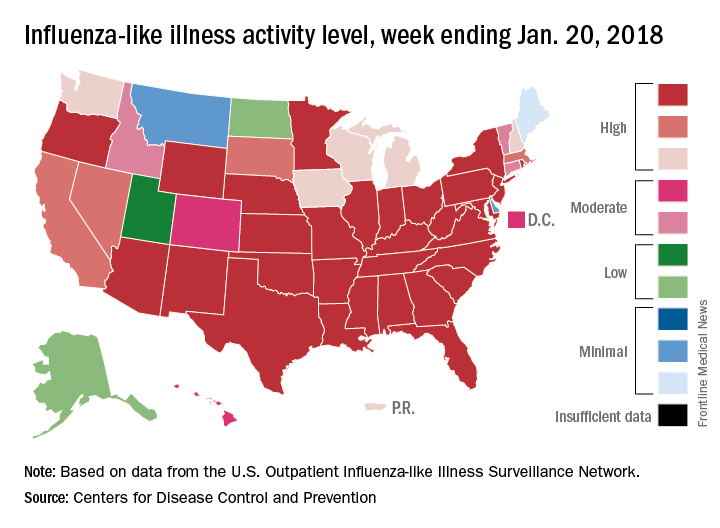
according to data from the Centers for Disease Control and Prevention.
That season was dominated by influenza A (H3N2), and the 2017-2018 season seems to be going down that same path. For the week ending Jan. 20, the proportion of outpatient visits for influenza-like illness increased to 6.6%, which is, for the second consecutive week, the highest level reported since October of – you guessed it – 2009, when it hit 7.7%, the CDC said in its weekly flu surveillance report.
The level reported last week, 6.3%, has been revised downward and now stands at an even 6%.
It turns out that 2018 is something of a milestone for the H3N2 virus. The virus first emerged in 1968, so it has reached its 50th anniversary, Dan Jernigan, MD, director of the influenza division at the CDC’s National Center for Immunization and Respiratory Diseases, Atlanta, said on Jan. 26 in a weekly briefing.
H3N2 must not be happy about hitting the big 5-0, however, because the map of influenza-like illness activity looks pretty red and angry. For the week ending Jan. 20, there were 30 states at the highest level of flu activity on the CDC’s 1-10 scale, with another nine in the “high” range at levels 8 and 9.
Dr. Jernigan did suggest that activity may have peaked in some areas of the country, with California among them.
There were seven pediatric deaths reported for the week ending Jan. 20, although six occurred in previous weeks. There have been 37 flu-related deaths among children so far during the 2017-2018 season, the CDC said.
according to data from the Centers for Disease Control and Prevention.
That season was dominated by influenza A (H3N2), and the 2017-2018 season seems to be going down that same path. For the week ending Jan. 20, the proportion of outpatient visits for influenza-like illness increased to 6.6%, which is, for the second consecutive week, the highest level reported since October of – you guessed it – 2009, when it hit 7.7%, the CDC said in its weekly flu surveillance report.
The level reported last week, 6.3%, has been revised downward and now stands at an even 6%.
It turns out that 2018 is something of a milestone for the H3N2 virus. The virus first emerged in 1968, so it has reached its 50th anniversary, Dan Jernigan, MD, director of the influenza division at the CDC’s National Center for Immunization and Respiratory Diseases, Atlanta, said on Jan. 26 in a weekly briefing.
H3N2 must not be happy about hitting the big 5-0, however, because the map of influenza-like illness activity looks pretty red and angry. For the week ending Jan. 20, there were 30 states at the highest level of flu activity on the CDC’s 1-10 scale, with another nine in the “high” range at levels 8 and 9.
Dr. Jernigan did suggest that activity may have peaked in some areas of the country, with California among them.
There were seven pediatric deaths reported for the week ending Jan. 20, although six occurred in previous weeks. There have been 37 flu-related deaths among children so far during the 2017-2018 season, the CDC said.
FDA approves starting dose of roflumilast
The Food and Drug Administration has approved the use of a 250-mcg dose of roflumilast for patients with chronic obstructive pulmonary disease (COPD) for 4 weeks, followed by the use of 500-mcg therapeutic doses, according to a statement from the drug’s marketer, AstraZeneca.
The larger doses of roflumilast (Daliresp) are currently indicated for reducing the risk of COPD exacerbations in patients with severe COPD associated with chronic bronchitis and a history of exacerbations, according to the statement. The selective phosphodiesterase-4 inhibitor, roflumilast, was approved for this use in 500-mcg doses in 2011. The new smaller doses of the drug are being offered to help reduce the rate of treatment discontinuation with use of the higher therapeutic dosing. The 250-mcg doses of roflumilast are not to be used as treatment for COPD.
“As the only once-daily tablet to provide enhanced protection against COPD exacerbations when added to current bronchodilator therapy, this is an important new dosing option to help patients start and stay on treatment. Exacerbations are associated with hospitalizations and an accelerated decline in lung function, and these patients living with COPD need effective treatment options,” Tosh Butt, vice president, respiratory, at AstraZeneca, said in the press release.
The approval of use of the 250-mcg doses was based on data from the OPTIMIZE study (Evaluation of Tolerability and Pharmacokinetics of Roflumilast trial, 250 mcg and 500 mcg, as an add-on to Standard COPD Treatment to Treat Severe COPD), according to the statement.
Over 12 weeks, the percentage of patients stopping treatment was significantly lower in those first given 250 mcg of roflumilast daily for 4 weeks, followed by 500 mcg once a week for 8 weeks (18.4%), compared with those given 500 mcg of roflumilast daily for 12 weeks (24.6%; odds ratio, 0.66; 95% confidence interval, 0.47-0.93; P = .017).
In eight controlled clinical trials, the most common adverse effects were diarrhea, weight loss, nausea, headache, back pain, influenza, insomnia, dizziness, and decreased appetite.
The Food and Drug Administration has approved the use of a 250-mcg dose of roflumilast for patients with chronic obstructive pulmonary disease (COPD) for 4 weeks, followed by the use of 500-mcg therapeutic doses, according to a statement from the drug’s marketer, AstraZeneca.
The larger doses of roflumilast (Daliresp) are currently indicated for reducing the risk of COPD exacerbations in patients with severe COPD associated with chronic bronchitis and a history of exacerbations, according to the statement. The selective phosphodiesterase-4 inhibitor, roflumilast, was approved for this use in 500-mcg doses in 2011. The new smaller doses of the drug are being offered to help reduce the rate of treatment discontinuation with use of the higher therapeutic dosing. The 250-mcg doses of roflumilast are not to be used as treatment for COPD.
“As the only once-daily tablet to provide enhanced protection against COPD exacerbations when added to current bronchodilator therapy, this is an important new dosing option to help patients start and stay on treatment. Exacerbations are associated with hospitalizations and an accelerated decline in lung function, and these patients living with COPD need effective treatment options,” Tosh Butt, vice president, respiratory, at AstraZeneca, said in the press release.
The approval of use of the 250-mcg doses was based on data from the OPTIMIZE study (Evaluation of Tolerability and Pharmacokinetics of Roflumilast trial, 250 mcg and 500 mcg, as an add-on to Standard COPD Treatment to Treat Severe COPD), according to the statement.
Over 12 weeks, the percentage of patients stopping treatment was significantly lower in those first given 250 mcg of roflumilast daily for 4 weeks, followed by 500 mcg once a week for 8 weeks (18.4%), compared with those given 500 mcg of roflumilast daily for 12 weeks (24.6%; odds ratio, 0.66; 95% confidence interval, 0.47-0.93; P = .017).
In eight controlled clinical trials, the most common adverse effects were diarrhea, weight loss, nausea, headache, back pain, influenza, insomnia, dizziness, and decreased appetite.
The Food and Drug Administration has approved the use of a 250-mcg dose of roflumilast for patients with chronic obstructive pulmonary disease (COPD) for 4 weeks, followed by the use of 500-mcg therapeutic doses, according to a statement from the drug’s marketer, AstraZeneca.
The larger doses of roflumilast (Daliresp) are currently indicated for reducing the risk of COPD exacerbations in patients with severe COPD associated with chronic bronchitis and a history of exacerbations, according to the statement. The selective phosphodiesterase-4 inhibitor, roflumilast, was approved for this use in 500-mcg doses in 2011. The new smaller doses of the drug are being offered to help reduce the rate of treatment discontinuation with use of the higher therapeutic dosing. The 250-mcg doses of roflumilast are not to be used as treatment for COPD.
“As the only once-daily tablet to provide enhanced protection against COPD exacerbations when added to current bronchodilator therapy, this is an important new dosing option to help patients start and stay on treatment. Exacerbations are associated with hospitalizations and an accelerated decline in lung function, and these patients living with COPD need effective treatment options,” Tosh Butt, vice president, respiratory, at AstraZeneca, said in the press release.
The approval of use of the 250-mcg doses was based on data from the OPTIMIZE study (Evaluation of Tolerability and Pharmacokinetics of Roflumilast trial, 250 mcg and 500 mcg, as an add-on to Standard COPD Treatment to Treat Severe COPD), according to the statement.
Over 12 weeks, the percentage of patients stopping treatment was significantly lower in those first given 250 mcg of roflumilast daily for 4 weeks, followed by 500 mcg once a week for 8 weeks (18.4%), compared with those given 500 mcg of roflumilast daily for 12 weeks (24.6%; odds ratio, 0.66; 95% confidence interval, 0.47-0.93; P = .017).
In eight controlled clinical trials, the most common adverse effects were diarrhea, weight loss, nausea, headache, back pain, influenza, insomnia, dizziness, and decreased appetite.