User login
Buy the IBD, get the comorbidity for free
Chronic conditions, such as arthritis and respiratory disease, are significantly more common in adults with inflammatory bowel disease than in those without IBD, according to the Centers for Disease Control and Prevention.
The age-adjusted prevalence of arthritis in adults with IBD is 36.3%, compared with 21.1% for those without IBD, and the prevalence of respiratory disease is 27.3% for IBD patients and 16.6% for non-IBD patients, CDC investigators reported in the Morbidity and Mortality Weekly Report.
Other comorbid chronic conditions with a significantly higher prevalence in patients with IBD than in those without were ulcer (26% vs. 5.5%), cardiovascular disease (19.2% vs. 12%), and cancer (13.7% vs. 8.1%), said Fang Xu, PhD, of the CDC’s National Center for Chronic Disease Prevention and Health Promotion and associates.
Serious psychological distress in the past 30 days was significantly more prevalent in adults with IBD (7.4%) than in those without it (3.4%), and those with IBD were also significantly more likely to report averaging less than 7 hours of sleep than were those without IBD (38.2% vs. 32.2%), according to their analysis of the 2015 and 2016 National Health Interview Surveys.
“Given the disease’s complexity and the effects of chronic conditions and symptoms, optimal IBD care might require a multidisciplinary approach that includes gastroenterologists, preventive medicine specialists, and other medical practitioners,” the investigators wrote.
SOURCE: Xu F et al. MMWR. 2018 Feb 16;67(6);190-5.
Chronic conditions, such as arthritis and respiratory disease, are significantly more common in adults with inflammatory bowel disease than in those without IBD, according to the Centers for Disease Control and Prevention.
The age-adjusted prevalence of arthritis in adults with IBD is 36.3%, compared with 21.1% for those without IBD, and the prevalence of respiratory disease is 27.3% for IBD patients and 16.6% for non-IBD patients, CDC investigators reported in the Morbidity and Mortality Weekly Report.
Other comorbid chronic conditions with a significantly higher prevalence in patients with IBD than in those without were ulcer (26% vs. 5.5%), cardiovascular disease (19.2% vs. 12%), and cancer (13.7% vs. 8.1%), said Fang Xu, PhD, of the CDC’s National Center for Chronic Disease Prevention and Health Promotion and associates.
Serious psychological distress in the past 30 days was significantly more prevalent in adults with IBD (7.4%) than in those without it (3.4%), and those with IBD were also significantly more likely to report averaging less than 7 hours of sleep than were those without IBD (38.2% vs. 32.2%), according to their analysis of the 2015 and 2016 National Health Interview Surveys.
“Given the disease’s complexity and the effects of chronic conditions and symptoms, optimal IBD care might require a multidisciplinary approach that includes gastroenterologists, preventive medicine specialists, and other medical practitioners,” the investigators wrote.
SOURCE: Xu F et al. MMWR. 2018 Feb 16;67(6);190-5.
Chronic conditions, such as arthritis and respiratory disease, are significantly more common in adults with inflammatory bowel disease than in those without IBD, according to the Centers for Disease Control and Prevention.
The age-adjusted prevalence of arthritis in adults with IBD is 36.3%, compared with 21.1% for those without IBD, and the prevalence of respiratory disease is 27.3% for IBD patients and 16.6% for non-IBD patients, CDC investigators reported in the Morbidity and Mortality Weekly Report.
Other comorbid chronic conditions with a significantly higher prevalence in patients with IBD than in those without were ulcer (26% vs. 5.5%), cardiovascular disease (19.2% vs. 12%), and cancer (13.7% vs. 8.1%), said Fang Xu, PhD, of the CDC’s National Center for Chronic Disease Prevention and Health Promotion and associates.
Serious psychological distress in the past 30 days was significantly more prevalent in adults with IBD (7.4%) than in those without it (3.4%), and those with IBD were also significantly more likely to report averaging less than 7 hours of sleep than were those without IBD (38.2% vs. 32.2%), according to their analysis of the 2015 and 2016 National Health Interview Surveys.
“Given the disease’s complexity and the effects of chronic conditions and symptoms, optimal IBD care might require a multidisciplinary approach that includes gastroenterologists, preventive medicine specialists, and other medical practitioners,” the investigators wrote.
SOURCE: Xu F et al. MMWR. 2018 Feb 16;67(6);190-5.
FROM MMWR
Lurasidone approved for bipolar I depression for children aged 10-17
The Food and Drug Administration has approved lurasidone HCI (Latuda) for treating bipolar I depression in children and adolescents, according to a March 6 statement from the drug’s manufacturer.
“We know that children who have been diagnosed with bipolar depression can be at risk for poor school performance and impairments in social functioning,” said Robert L. Findling, MD, professor of psychiatry and behavioral sciences at Johns Hopkins University, Baltimore, in the statement.
Approval of the atypical antipsychotic is based on results of a 6-week, randomized placebo-controlled phase 3 study of 347 children and adolescents diagnosed with bipolar I depression. Patients received either 20-80 mg/day of lurasidone or placebo.
Patients who received lurasidone reportedly experienced improved bipolar depression symptoms, compared with placebo, based on “the primary efficacy endpoint of change from baseline to week 6 on the Children’s Depression Rating Scale–Revised total score (–21.0 vs. –15.3; effect size = 0.45; P less than .0001),” the statement said. Clinically relevant changes also were found among patients who took the medication on other measures, including the Clinical Global Impressions-Bipolar Scale.
The most common adverse effects were nausea (16% vs. 5.8%), weight gain (6.9% vs. 1.7%), and insomnia (5.1% vs. 2.3%).
Lurasidone also has been approved for treating schizophrenia and bipolar I depression in adults. Last year, the drug was approved for treating schizophrenia in adolescents.
The Food and Drug Administration has approved lurasidone HCI (Latuda) for treating bipolar I depression in children and adolescents, according to a March 6 statement from the drug’s manufacturer.
“We know that children who have been diagnosed with bipolar depression can be at risk for poor school performance and impairments in social functioning,” said Robert L. Findling, MD, professor of psychiatry and behavioral sciences at Johns Hopkins University, Baltimore, in the statement.
Approval of the atypical antipsychotic is based on results of a 6-week, randomized placebo-controlled phase 3 study of 347 children and adolescents diagnosed with bipolar I depression. Patients received either 20-80 mg/day of lurasidone or placebo.
Patients who received lurasidone reportedly experienced improved bipolar depression symptoms, compared with placebo, based on “the primary efficacy endpoint of change from baseline to week 6 on the Children’s Depression Rating Scale–Revised total score (–21.0 vs. –15.3; effect size = 0.45; P less than .0001),” the statement said. Clinically relevant changes also were found among patients who took the medication on other measures, including the Clinical Global Impressions-Bipolar Scale.
The most common adverse effects were nausea (16% vs. 5.8%), weight gain (6.9% vs. 1.7%), and insomnia (5.1% vs. 2.3%).
Lurasidone also has been approved for treating schizophrenia and bipolar I depression in adults. Last year, the drug was approved for treating schizophrenia in adolescents.
The Food and Drug Administration has approved lurasidone HCI (Latuda) for treating bipolar I depression in children and adolescents, according to a March 6 statement from the drug’s manufacturer.
“We know that children who have been diagnosed with bipolar depression can be at risk for poor school performance and impairments in social functioning,” said Robert L. Findling, MD, professor of psychiatry and behavioral sciences at Johns Hopkins University, Baltimore, in the statement.
Approval of the atypical antipsychotic is based on results of a 6-week, randomized placebo-controlled phase 3 study of 347 children and adolescents diagnosed with bipolar I depression. Patients received either 20-80 mg/day of lurasidone or placebo.
Patients who received lurasidone reportedly experienced improved bipolar depression symptoms, compared with placebo, based on “the primary efficacy endpoint of change from baseline to week 6 on the Children’s Depression Rating Scale–Revised total score (–21.0 vs. –15.3; effect size = 0.45; P less than .0001),” the statement said. Clinically relevant changes also were found among patients who took the medication on other measures, including the Clinical Global Impressions-Bipolar Scale.
The most common adverse effects were nausea (16% vs. 5.8%), weight gain (6.9% vs. 1.7%), and insomnia (5.1% vs. 2.3%).
Lurasidone also has been approved for treating schizophrenia and bipolar I depression in adults. Last year, the drug was approved for treating schizophrenia in adolescents.
Flu activity takes another turn for the better
Outpatient influenza-like illness activity continues to drop, but pediatric deaths for 2017-2018 are already higher than either of the last two entire seasons, according to the Centers for Disease and Prevention.
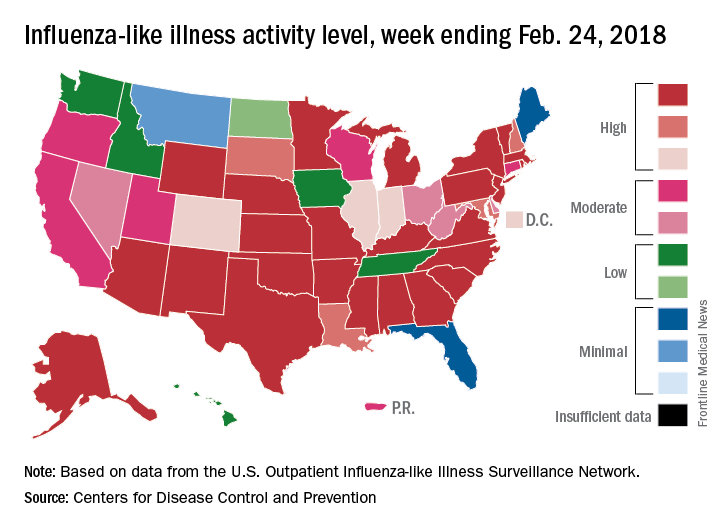
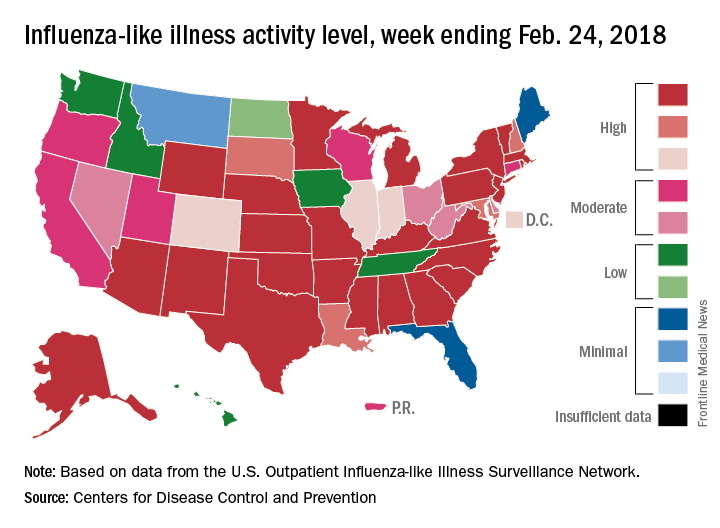
An additional 17 influenza-like illness-related (ILI) pediatric deaths were reported during the week ending Feb. 24, eight of which occurred in previous weeks. That brings the total to 114 for the 2017-2018 flu season so far, compared with 110 for the entire 2016-2017 season and 93 for the 2015-2016 season, the CDC reported Mar. 2.
The proportion of outpatient visits for ILI took another big drop, falling to 5.0% for the week, which was down from 6.4% the previous week and the seasonal high of 7.4% the 2 weeks before that (Feb. 10 and Feb. 3), CDC data show.
Flu-related hospitalizations, however, continued to rise to new highs, as the cumulative rate hit 81.7 per 100,000 population. In 2014-2015, the season with the highest number of hospitalizations since the CDC started keeping track, the cumulative rate for the corresponding week was 55.9 per 100,000, according to the CDC’s Fluview website.
The map of state-reported ILI activity shows that 25 states are at level 10 on the CDC’s 1-10 scale, which is down from 33 the week before. Eight other states and the District of Columbia were in the “high” range with activity at levels 8 and 9 for the week ending Feb. 24, the CDC said.
Outpatient influenza-like illness activity continues to drop, but pediatric deaths for 2017-2018 are already higher than either of the last two entire seasons, according to the Centers for Disease and Prevention.

An additional 17 influenza-like illness-related (ILI) pediatric deaths were reported during the week ending Feb. 24, eight of which occurred in previous weeks. That brings the total to 114 for the 2017-2018 flu season so far, compared with 110 for the entire 2016-2017 season and 93 for the 2015-2016 season, the CDC reported Mar. 2.
The proportion of outpatient visits for ILI took another big drop, falling to 5.0% for the week, which was down from 6.4% the previous week and the seasonal high of 7.4% the 2 weeks before that (Feb. 10 and Feb. 3), CDC data show.
Flu-related hospitalizations, however, continued to rise to new highs, as the cumulative rate hit 81.7 per 100,000 population. In 2014-2015, the season with the highest number of hospitalizations since the CDC started keeping track, the cumulative rate for the corresponding week was 55.9 per 100,000, according to the CDC’s Fluview website.
The map of state-reported ILI activity shows that 25 states are at level 10 on the CDC’s 1-10 scale, which is down from 33 the week before. Eight other states and the District of Columbia were in the “high” range with activity at levels 8 and 9 for the week ending Feb. 24, the CDC said.
Outpatient influenza-like illness activity continues to drop, but pediatric deaths for 2017-2018 are already higher than either of the last two entire seasons, according to the Centers for Disease and Prevention.

An additional 17 influenza-like illness-related (ILI) pediatric deaths were reported during the week ending Feb. 24, eight of which occurred in previous weeks. That brings the total to 114 for the 2017-2018 flu season so far, compared with 110 for the entire 2016-2017 season and 93 for the 2015-2016 season, the CDC reported Mar. 2.
The proportion of outpatient visits for ILI took another big drop, falling to 5.0% for the week, which was down from 6.4% the previous week and the seasonal high of 7.4% the 2 weeks before that (Feb. 10 and Feb. 3), CDC data show.
Flu-related hospitalizations, however, continued to rise to new highs, as the cumulative rate hit 81.7 per 100,000 population. In 2014-2015, the season with the highest number of hospitalizations since the CDC started keeping track, the cumulative rate for the corresponding week was 55.9 per 100,000, according to the CDC’s Fluview website.
The map of state-reported ILI activity shows that 25 states are at level 10 on the CDC’s 1-10 scale, which is down from 33 the week before. Eight other states and the District of Columbia were in the “high” range with activity at levels 8 and 9 for the week ending Feb. 24, the CDC said.
Opioid deaths in the ED increase nationally
Opioid-related deaths in emergency departments increased by approximately 30% across all regions of the United States between 2016 and 2017, according to the Centers for Disease Control and Prevention.
Analysis of 91 million ED visits from the CDC’s National Syndromic Surveillance Program and Enhanced State Opioid Overdose Surveillance database found significant increases in opioid overdose deaths in 16 states, reaching as high as 109% in Wisconsin and 106% in Delaware, CDC officials said during a press briefing.
“We are currently seeing the highest drug overdose death rate ever recorded in the United States, driven by prescription opioids and by illicit opioids such as heroin and illicitly manufactured fentanyl,” said Anne Schuchat, MD, acting CDC director. “In 2016, there were more than 63,000 drug overdose deaths, and more than 42,000 of those deaths involved an opioid.”
Of the 91 million visits, a total of 261,755 were suspected of opioid overdoses across both databases.
The greatest increase was seen in the Midwest region (69.7%), followed by the West (40.3%), Northeast (21.3%), Southwest (20.2%), and Southeast (14%).
Death rates rose across all demographics, regardless of sex or age.
While Delaware recorded some of the highest increases in deaths, Massachusetts, New Hampshire, and Rhode Island decreased, although not within statistical significance.
“These decreases may possibly be related to implementation of interventions, including expansion of access to medication-assisted treatment,” said Dr. Schuchat. “The decrease in Kentucky during this period of time may reflect some fluctuations in drug supply.”
In a comparison of urban and rural areas, large and medium metropolitan communities had the sharpest increase, at 45%.
To combat the rise in deaths, the CDC is encouraging an increase in naloxone distribution and training for first responders and community members.
The agency also recommends that local health departments begin using ED data to alert local communities when opioid-related deaths rise.
“This is a very difficult and fast-moving epidemic, and there are no easy solutions,” Dr. Schuchat said. [These data send] “a wake-up call about the need to improve what happens when patients leave the emergency department; all of us working together, government, public health, the medical community, law enforcement, and community members themselves can help fight this epidemic and save lives.”
ezimmerman@frontlinemedcom.com
SOURCE: Vivolo-Kantor AM et al. MMWR Morb Mortal Wkly Rep. 6 Mar 2018. doi: 10.15585/mmwr.mm6709e1.
Opioid-related deaths in emergency departments increased by approximately 30% across all regions of the United States between 2016 and 2017, according to the Centers for Disease Control and Prevention.
Analysis of 91 million ED visits from the CDC’s National Syndromic Surveillance Program and Enhanced State Opioid Overdose Surveillance database found significant increases in opioid overdose deaths in 16 states, reaching as high as 109% in Wisconsin and 106% in Delaware, CDC officials said during a press briefing.
“We are currently seeing the highest drug overdose death rate ever recorded in the United States, driven by prescription opioids and by illicit opioids such as heroin and illicitly manufactured fentanyl,” said Anne Schuchat, MD, acting CDC director. “In 2016, there were more than 63,000 drug overdose deaths, and more than 42,000 of those deaths involved an opioid.”
Of the 91 million visits, a total of 261,755 were suspected of opioid overdoses across both databases.
The greatest increase was seen in the Midwest region (69.7%), followed by the West (40.3%), Northeast (21.3%), Southwest (20.2%), and Southeast (14%).
Death rates rose across all demographics, regardless of sex or age.
While Delaware recorded some of the highest increases in deaths, Massachusetts, New Hampshire, and Rhode Island decreased, although not within statistical significance.
“These decreases may possibly be related to implementation of interventions, including expansion of access to medication-assisted treatment,” said Dr. Schuchat. “The decrease in Kentucky during this period of time may reflect some fluctuations in drug supply.”
In a comparison of urban and rural areas, large and medium metropolitan communities had the sharpest increase, at 45%.
To combat the rise in deaths, the CDC is encouraging an increase in naloxone distribution and training for first responders and community members.
The agency also recommends that local health departments begin using ED data to alert local communities when opioid-related deaths rise.
“This is a very difficult and fast-moving epidemic, and there are no easy solutions,” Dr. Schuchat said. [These data send] “a wake-up call about the need to improve what happens when patients leave the emergency department; all of us working together, government, public health, the medical community, law enforcement, and community members themselves can help fight this epidemic and save lives.”
ezimmerman@frontlinemedcom.com
SOURCE: Vivolo-Kantor AM et al. MMWR Morb Mortal Wkly Rep. 6 Mar 2018. doi: 10.15585/mmwr.mm6709e1.
Opioid-related deaths in emergency departments increased by approximately 30% across all regions of the United States between 2016 and 2017, according to the Centers for Disease Control and Prevention.
Analysis of 91 million ED visits from the CDC’s National Syndromic Surveillance Program and Enhanced State Opioid Overdose Surveillance database found significant increases in opioid overdose deaths in 16 states, reaching as high as 109% in Wisconsin and 106% in Delaware, CDC officials said during a press briefing.
“We are currently seeing the highest drug overdose death rate ever recorded in the United States, driven by prescription opioids and by illicit opioids such as heroin and illicitly manufactured fentanyl,” said Anne Schuchat, MD, acting CDC director. “In 2016, there were more than 63,000 drug overdose deaths, and more than 42,000 of those deaths involved an opioid.”
Of the 91 million visits, a total of 261,755 were suspected of opioid overdoses across both databases.
The greatest increase was seen in the Midwest region (69.7%), followed by the West (40.3%), Northeast (21.3%), Southwest (20.2%), and Southeast (14%).
Death rates rose across all demographics, regardless of sex or age.
While Delaware recorded some of the highest increases in deaths, Massachusetts, New Hampshire, and Rhode Island decreased, although not within statistical significance.
“These decreases may possibly be related to implementation of interventions, including expansion of access to medication-assisted treatment,” said Dr. Schuchat. “The decrease in Kentucky during this period of time may reflect some fluctuations in drug supply.”
In a comparison of urban and rural areas, large and medium metropolitan communities had the sharpest increase, at 45%.
To combat the rise in deaths, the CDC is encouraging an increase in naloxone distribution and training for first responders and community members.
The agency also recommends that local health departments begin using ED data to alert local communities when opioid-related deaths rise.
“This is a very difficult and fast-moving epidemic, and there are no easy solutions,” Dr. Schuchat said. [These data send] “a wake-up call about the need to improve what happens when patients leave the emergency department; all of us working together, government, public health, the medical community, law enforcement, and community members themselves can help fight this epidemic and save lives.”
ezimmerman@frontlinemedcom.com
SOURCE: Vivolo-Kantor AM et al. MMWR Morb Mortal Wkly Rep. 6 Mar 2018. doi: 10.15585/mmwr.mm6709e1.
FROM MMWR
Flu season shows signs of slowing
Flu-related outpatient activity dropped for the second week in a row as the cumulative hospitalization rate continues to rise, according to data from the Centers for Disease Control and Prevention.
For the week ending Feb. 17, the proportion of outpatient visits for influenza-like illness (ILI) was 6.4%, which was down from 7.4% the previous week (Feb. 10) and down from the seasonal high of 7.5% set 2 weeks earlier, the CDC said in its weekly flu surveillance report. The rate for the week ending Feb. 10 was reported last week as 7.5%, but it has been revised downward.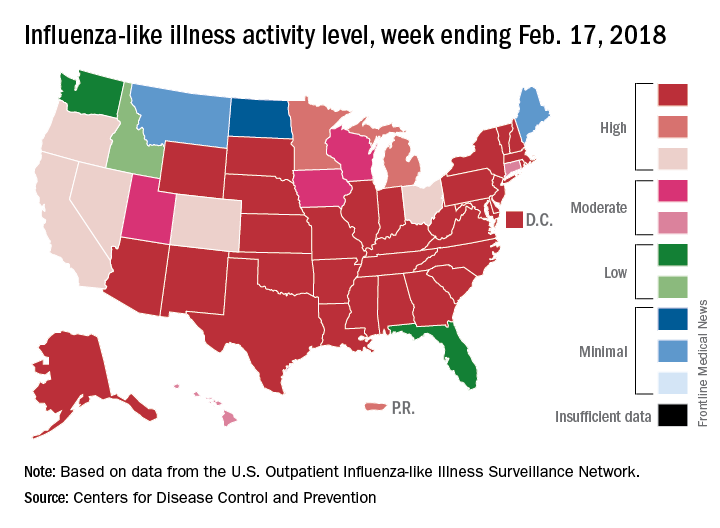
State reports of ILI activity support the decreases seen in the national outpatient rate. There were 33 states at level 10 on the CDC’s 1-10 scale for the week ending Feb. 17 – down from 39 the week before – and a total of 41 states in the “high” range from levels 8-10, compared with 45 the previous week, CDC’s FluView website shows.
Reports of flu-related pediatric deaths continued: 13 deaths were reported during the week, although 9 occurred in previous weeks. The total for the 2017-2018 season is now 97. There were 110 pediatric deaths in the entire 2016-2017 season, 93 during the 2015-2016 season, and 149 in 2014-2015, the CDC said.
Flu-related outpatient activity dropped for the second week in a row as the cumulative hospitalization rate continues to rise, according to data from the Centers for Disease Control and Prevention.
For the week ending Feb. 17, the proportion of outpatient visits for influenza-like illness (ILI) was 6.4%, which was down from 7.4% the previous week (Feb. 10) and down from the seasonal high of 7.5% set 2 weeks earlier, the CDC said in its weekly flu surveillance report. The rate for the week ending Feb. 10 was reported last week as 7.5%, but it has been revised downward.
State reports of ILI activity support the decreases seen in the national outpatient rate. There were 33 states at level 10 on the CDC’s 1-10 scale for the week ending Feb. 17 – down from 39 the week before – and a total of 41 states in the “high” range from levels 8-10, compared with 45 the previous week, CDC’s FluView website shows.
Reports of flu-related pediatric deaths continued: 13 deaths were reported during the week, although 9 occurred in previous weeks. The total for the 2017-2018 season is now 97. There were 110 pediatric deaths in the entire 2016-2017 season, 93 during the 2015-2016 season, and 149 in 2014-2015, the CDC said.
Flu-related outpatient activity dropped for the second week in a row as the cumulative hospitalization rate continues to rise, according to data from the Centers for Disease Control and Prevention.
For the week ending Feb. 17, the proportion of outpatient visits for influenza-like illness (ILI) was 6.4%, which was down from 7.4% the previous week (Feb. 10) and down from the seasonal high of 7.5% set 2 weeks earlier, the CDC said in its weekly flu surveillance report. The rate for the week ending Feb. 10 was reported last week as 7.5%, but it has been revised downward.
State reports of ILI activity support the decreases seen in the national outpatient rate. There were 33 states at level 10 on the CDC’s 1-10 scale for the week ending Feb. 17 – down from 39 the week before – and a total of 41 states in the “high” range from levels 8-10, compared with 45 the previous week, CDC’s FluView website shows.
Reports of flu-related pediatric deaths continued: 13 deaths were reported during the week, although 9 occurred in previous weeks. The total for the 2017-2018 season is now 97. There were 110 pediatric deaths in the entire 2016-2017 season, 93 during the 2015-2016 season, and 149 in 2014-2015, the CDC said.
FDA warns against clarithromycin use in patients with heart disease
The Food and Drug Administration has added a new warning for an increased risk of death in patients with heart disease who have used clarithromycin (Biaxin), on the basis of results of a 10-year follow-up from the CLARICOR trial.
The CLARICOR trial followed 4,372 randomized patients for at least 2 years after undergoing 14 days of treatment with daily doses of 500 mg clarithromycin. Among these patients, researchers observed an unexpected increase in deaths in patients with coronary heart disease. (The Feb. 22 FDA statement announcing the alert did not provide data from CLARICOR.) As of yet, there is no clear explanation of how clarithromycin would lead to more deaths, compared with a placebo, the agency said.
Regardless, two of the six observational studies published found a link between clarithromycin use and long-term risks; four did not. The CLARICOR trial provides the strongest evidence of increased health risks, the statement said.
The FDA is recommending that health care professionals be aware of the risks associated with clarithromycin use and consider the benefits and risks of use in patients with heart disease. If at all possible, the use of other antibiotics may be a better option. Doctors should advise patients to be aware of signs and symptoms associated with cardiovascular issues.
Patients are also an important piece of the puzzle and should communicate with their health care providers about heart disease, particularly when taking antibiotics to treat for an infection.
The FDA has added the results of the CLARICOR trial to the clarithromycin drug labels. The agency will continue to monitor the safety reports in patients using clarithromycin.
Serious adverse events associated with clarithromycin should be reported to the FDA’s MedWatch program at 800-332-1088 or www.fda.gov/medwatch/.
The Food and Drug Administration has added a new warning for an increased risk of death in patients with heart disease who have used clarithromycin (Biaxin), on the basis of results of a 10-year follow-up from the CLARICOR trial.
The CLARICOR trial followed 4,372 randomized patients for at least 2 years after undergoing 14 days of treatment with daily doses of 500 mg clarithromycin. Among these patients, researchers observed an unexpected increase in deaths in patients with coronary heart disease. (The Feb. 22 FDA statement announcing the alert did not provide data from CLARICOR.) As of yet, there is no clear explanation of how clarithromycin would lead to more deaths, compared with a placebo, the agency said.
Regardless, two of the six observational studies published found a link between clarithromycin use and long-term risks; four did not. The CLARICOR trial provides the strongest evidence of increased health risks, the statement said.
The FDA is recommending that health care professionals be aware of the risks associated with clarithromycin use and consider the benefits and risks of use in patients with heart disease. If at all possible, the use of other antibiotics may be a better option. Doctors should advise patients to be aware of signs and symptoms associated with cardiovascular issues.
Patients are also an important piece of the puzzle and should communicate with their health care providers about heart disease, particularly when taking antibiotics to treat for an infection.
The FDA has added the results of the CLARICOR trial to the clarithromycin drug labels. The agency will continue to monitor the safety reports in patients using clarithromycin.
Serious adverse events associated with clarithromycin should be reported to the FDA’s MedWatch program at 800-332-1088 or www.fda.gov/medwatch/.
The Food and Drug Administration has added a new warning for an increased risk of death in patients with heart disease who have used clarithromycin (Biaxin), on the basis of results of a 10-year follow-up from the CLARICOR trial.
The CLARICOR trial followed 4,372 randomized patients for at least 2 years after undergoing 14 days of treatment with daily doses of 500 mg clarithromycin. Among these patients, researchers observed an unexpected increase in deaths in patients with coronary heart disease. (The Feb. 22 FDA statement announcing the alert did not provide data from CLARICOR.) As of yet, there is no clear explanation of how clarithromycin would lead to more deaths, compared with a placebo, the agency said.
Regardless, two of the six observational studies published found a link between clarithromycin use and long-term risks; four did not. The CLARICOR trial provides the strongest evidence of increased health risks, the statement said.
The FDA is recommending that health care professionals be aware of the risks associated with clarithromycin use and consider the benefits and risks of use in patients with heart disease. If at all possible, the use of other antibiotics may be a better option. Doctors should advise patients to be aware of signs and symptoms associated with cardiovascular issues.
Patients are also an important piece of the puzzle and should communicate with their health care providers about heart disease, particularly when taking antibiotics to treat for an infection.
The FDA has added the results of the CLARICOR trial to the clarithromycin drug labels. The agency will continue to monitor the safety reports in patients using clarithromycin.
Serious adverse events associated with clarithromycin should be reported to the FDA’s MedWatch program at 800-332-1088 or www.fda.gov/medwatch/.
FDA grants priority review for AML drug
The Food and Drug Administration has granted priority review status to ivosidenib for the treatment of patients with relapsed or refractory acute myeloid leukemia with an isocitrate dehydrogenase 1 mutation.
The drug, marketed by Agios Pharmaceuticals, was given a Prescription Drug User Free Act action date of Aug. 21, 2018.
Results from a phase 1 dose-escalation and expansion study (AG120-C-001) presented at the annual meeting of the American Society of Hematology showed a complete response and complete response with partial hematologic recovery rate of 30.4% in 125 patients with relapsed/refractory AML who received the drug, according to Agios.
The Food and Drug Administration has granted priority review status to ivosidenib for the treatment of patients with relapsed or refractory acute myeloid leukemia with an isocitrate dehydrogenase 1 mutation.
The drug, marketed by Agios Pharmaceuticals, was given a Prescription Drug User Free Act action date of Aug. 21, 2018.
Results from a phase 1 dose-escalation and expansion study (AG120-C-001) presented at the annual meeting of the American Society of Hematology showed a complete response and complete response with partial hematologic recovery rate of 30.4% in 125 patients with relapsed/refractory AML who received the drug, according to Agios.
The Food and Drug Administration has granted priority review status to ivosidenib for the treatment of patients with relapsed or refractory acute myeloid leukemia with an isocitrate dehydrogenase 1 mutation.
The drug, marketed by Agios Pharmaceuticals, was given a Prescription Drug User Free Act action date of Aug. 21, 2018.
Results from a phase 1 dose-escalation and expansion study (AG120-C-001) presented at the annual meeting of the American Society of Hematology showed a complete response and complete response with partial hematologic recovery rate of 30.4% in 125 patients with relapsed/refractory AML who received the drug, according to Agios.
Flu increase may be slowing
A bit of revisionist history has outpatient influenza activity at a lower level than was reported last week, even though it hasn’t dropped.
The proportion of outpatient visits for influenza-like illness (ILI) for the week ending Feb. 10 was 7.5%, according to the Centers for Disease Control. That is lower than the 7.7% previously reported for the week ending Feb. 3, which would seem to be a drop, but the CDC also has revised that earlier number to 7.5%, so there is no change. (This is not the first time an earlier ILI level has been retroactively lowered: The figure reported for the week ending Jan. 13 was revised in the following report from 6.3% down to 6.0%.)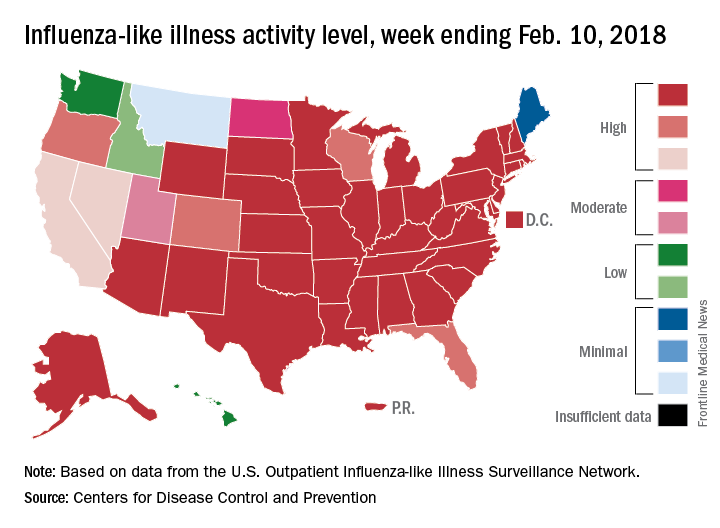
Hospital visits, however, continue to rise at record levels. The cumulative rate for the week ending Feb. 10 was 67.9 visits per 100,000 population, which is higher than the same week for the 2014-2015 (52.9 per 100,000) when flu hospitalizations for the season hit a high of 710,000. Flu-related pediatric deaths also went up, with 22 new reports; this brings the total to 84 for the 2017-2018 season.
A bit of revisionist history has outpatient influenza activity at a lower level than was reported last week, even though it hasn’t dropped.
The proportion of outpatient visits for influenza-like illness (ILI) for the week ending Feb. 10 was 7.5%, according to the Centers for Disease Control. That is lower than the 7.7% previously reported for the week ending Feb. 3, which would seem to be a drop, but the CDC also has revised that earlier number to 7.5%, so there is no change. (This is not the first time an earlier ILI level has been retroactively lowered: The figure reported for the week ending Jan. 13 was revised in the following report from 6.3% down to 6.0%.)
Hospital visits, however, continue to rise at record levels. The cumulative rate for the week ending Feb. 10 was 67.9 visits per 100,000 population, which is higher than the same week for the 2014-2015 (52.9 per 100,000) when flu hospitalizations for the season hit a high of 710,000. Flu-related pediatric deaths also went up, with 22 new reports; this brings the total to 84 for the 2017-2018 season.
A bit of revisionist history has outpatient influenza activity at a lower level than was reported last week, even though it hasn’t dropped.
The proportion of outpatient visits for influenza-like illness (ILI) for the week ending Feb. 10 was 7.5%, according to the Centers for Disease Control. That is lower than the 7.7% previously reported for the week ending Feb. 3, which would seem to be a drop, but the CDC also has revised that earlier number to 7.5%, so there is no change. (This is not the first time an earlier ILI level has been retroactively lowered: The figure reported for the week ending Jan. 13 was revised in the following report from 6.3% down to 6.0%.)
Hospital visits, however, continue to rise at record levels. The cumulative rate for the week ending Feb. 10 was 67.9 visits per 100,000 population, which is higher than the same week for the 2014-2015 (52.9 per 100,000) when flu hospitalizations for the season hit a high of 710,000. Flu-related pediatric deaths also went up, with 22 new reports; this brings the total to 84 for the 2017-2018 season.
FROM THE CDC WEEKLY U.S. INFLUENZA SURVEILLANCE REPORT
MMWR: Current flu vaccine does not protect elderly
, according to the Feb. 16 issue of Morbidity and Mortality Weekly Report.
The elderly are not among them. Although the vaccine was somewhat protective in children and adults up to 49 years old, “no statistically significant protection was observed in other age groups,” including people 65 years and older, reported investigators led by Brendan Flannery, PhD, of the Centers for Disease Control and Prevention influenza division.
They also reported that the cumulative hospitalization rate attributed to laboratory-confirmed influenza for the week ending Feb. 3, 2018 (59.9/100,000), exceeded the rate for the same week in 2014-2015 (50.9/100,000), an A(H3N2) virus–predominant season, and is the highest rate observed for this week since the system expanded to include adults during the 2005-2006 season.
This year’s overall effectiveness rating was in contrast to the 2016-2017 seasonal effectiveness of 48% (MMWR. 2017 Feb 17;66[6];167-71).
The CDC noted that influenza is going to be active for several more weeks, so “vaccination is still recommended,” but “treatment with influenza antiviral medications, where appropriate, is especially important this season.” Meanwhile, “influenza vaccines with improved effectiveness are needed,” the CDC said.
The estimates are based on 4,562 patients 6 months to over 65 years old presenting with acute respiratory illness in 2018 from Nov. 2 to Feb. 3 at five outpatient medical clinics scattered across the United States. Nasal and oropharyngeal swabs were tested with reverse transcription polymerase chain reaction for the presence of influenza viruses; 413 subjects were 65 years or older.
Vaccine effectiveness against the less common virus A(H1N1)pdm09 was 67%, and 42% against the even rarer influenza B viruses. Estimates were adjusted for a range of confounders, including study site, age, general health, and week of illness. Vaccination rates ranged from 45% to 59% across the study sites; 38% of the subjects tested positive for influenza, most for type A viruses. The shot didn’t work too well: 43% of the influenza cases had gotten it.
The 25% effectiveness against A(H3N2) is a bit higher than recent reports of 17% from Canada and 10% from Australia, but similar to the 32% efficacy reported in the United States for the 2016-2017 season.
“These interim estimates reflect ongoing challenges with the A(H3N2) vaccine component since the 2011-12 season,” the investigators wrote. “Multiple factors might be contributing to the reported [vaccine effectiveness] against A(H3N2) viruses this season. … Genetic changes in the vaccine virus hemagglutinin protein that arise during passage in eggs might result in a vaccine immune response that is less effective against circulating viruses.”
On a related note, on Feb. 18, Senators Edward J. Markey (D-Mass.), Richard Blumenthal (D-Conn.), and Amy Klobuchar (D-Minn.) held a press conference to announce they were introducing the Flu Vaccine Bill to dedicate $1 billion over a 5-year period in order to develop a flu vaccine that could provide lifetime protection.
The investigators had no conflicts of interest.
SOURCE: Flannery B. et al. MMWR. 2018 Feb 16;67(6):180-5; Budd A. et al. MMWR. 2018 Feb 16;67(6):169-79.
, according to the Feb. 16 issue of Morbidity and Mortality Weekly Report.
The elderly are not among them. Although the vaccine was somewhat protective in children and adults up to 49 years old, “no statistically significant protection was observed in other age groups,” including people 65 years and older, reported investigators led by Brendan Flannery, PhD, of the Centers for Disease Control and Prevention influenza division.
They also reported that the cumulative hospitalization rate attributed to laboratory-confirmed influenza for the week ending Feb. 3, 2018 (59.9/100,000), exceeded the rate for the same week in 2014-2015 (50.9/100,000), an A(H3N2) virus–predominant season, and is the highest rate observed for this week since the system expanded to include adults during the 2005-2006 season.
This year’s overall effectiveness rating was in contrast to the 2016-2017 seasonal effectiveness of 48% (MMWR. 2017 Feb 17;66[6];167-71).
The CDC noted that influenza is going to be active for several more weeks, so “vaccination is still recommended,” but “treatment with influenza antiviral medications, where appropriate, is especially important this season.” Meanwhile, “influenza vaccines with improved effectiveness are needed,” the CDC said.
The estimates are based on 4,562 patients 6 months to over 65 years old presenting with acute respiratory illness in 2018 from Nov. 2 to Feb. 3 at five outpatient medical clinics scattered across the United States. Nasal and oropharyngeal swabs were tested with reverse transcription polymerase chain reaction for the presence of influenza viruses; 413 subjects were 65 years or older.
Vaccine effectiveness against the less common virus A(H1N1)pdm09 was 67%, and 42% against the even rarer influenza B viruses. Estimates were adjusted for a range of confounders, including study site, age, general health, and week of illness. Vaccination rates ranged from 45% to 59% across the study sites; 38% of the subjects tested positive for influenza, most for type A viruses. The shot didn’t work too well: 43% of the influenza cases had gotten it.
The 25% effectiveness against A(H3N2) is a bit higher than recent reports of 17% from Canada and 10% from Australia, but similar to the 32% efficacy reported in the United States for the 2016-2017 season.
“These interim estimates reflect ongoing challenges with the A(H3N2) vaccine component since the 2011-12 season,” the investigators wrote. “Multiple factors might be contributing to the reported [vaccine effectiveness] against A(H3N2) viruses this season. … Genetic changes in the vaccine virus hemagglutinin protein that arise during passage in eggs might result in a vaccine immune response that is less effective against circulating viruses.”
On a related note, on Feb. 18, Senators Edward J. Markey (D-Mass.), Richard Blumenthal (D-Conn.), and Amy Klobuchar (D-Minn.) held a press conference to announce they were introducing the Flu Vaccine Bill to dedicate $1 billion over a 5-year period in order to develop a flu vaccine that could provide lifetime protection.
The investigators had no conflicts of interest.
SOURCE: Flannery B. et al. MMWR. 2018 Feb 16;67(6):180-5; Budd A. et al. MMWR. 2018 Feb 16;67(6):169-79.
, according to the Feb. 16 issue of Morbidity and Mortality Weekly Report.
The elderly are not among them. Although the vaccine was somewhat protective in children and adults up to 49 years old, “no statistically significant protection was observed in other age groups,” including people 65 years and older, reported investigators led by Brendan Flannery, PhD, of the Centers for Disease Control and Prevention influenza division.
They also reported that the cumulative hospitalization rate attributed to laboratory-confirmed influenza for the week ending Feb. 3, 2018 (59.9/100,000), exceeded the rate for the same week in 2014-2015 (50.9/100,000), an A(H3N2) virus–predominant season, and is the highest rate observed for this week since the system expanded to include adults during the 2005-2006 season.
This year’s overall effectiveness rating was in contrast to the 2016-2017 seasonal effectiveness of 48% (MMWR. 2017 Feb 17;66[6];167-71).
The CDC noted that influenza is going to be active for several more weeks, so “vaccination is still recommended,” but “treatment with influenza antiviral medications, where appropriate, is especially important this season.” Meanwhile, “influenza vaccines with improved effectiveness are needed,” the CDC said.
The estimates are based on 4,562 patients 6 months to over 65 years old presenting with acute respiratory illness in 2018 from Nov. 2 to Feb. 3 at five outpatient medical clinics scattered across the United States. Nasal and oropharyngeal swabs were tested with reverse transcription polymerase chain reaction for the presence of influenza viruses; 413 subjects were 65 years or older.
Vaccine effectiveness against the less common virus A(H1N1)pdm09 was 67%, and 42% against the even rarer influenza B viruses. Estimates were adjusted for a range of confounders, including study site, age, general health, and week of illness. Vaccination rates ranged from 45% to 59% across the study sites; 38% of the subjects tested positive for influenza, most for type A viruses. The shot didn’t work too well: 43% of the influenza cases had gotten it.
The 25% effectiveness against A(H3N2) is a bit higher than recent reports of 17% from Canada and 10% from Australia, but similar to the 32% efficacy reported in the United States for the 2016-2017 season.
“These interim estimates reflect ongoing challenges with the A(H3N2) vaccine component since the 2011-12 season,” the investigators wrote. “Multiple factors might be contributing to the reported [vaccine effectiveness] against A(H3N2) viruses this season. … Genetic changes in the vaccine virus hemagglutinin protein that arise during passage in eggs might result in a vaccine immune response that is less effective against circulating viruses.”
On a related note, on Feb. 18, Senators Edward J. Markey (D-Mass.), Richard Blumenthal (D-Conn.), and Amy Klobuchar (D-Minn.) held a press conference to announce they were introducing the Flu Vaccine Bill to dedicate $1 billion over a 5-year period in order to develop a flu vaccine that could provide lifetime protection.
The investigators had no conflicts of interest.
SOURCE: Flannery B. et al. MMWR. 2018 Feb 16;67(6):180-5; Budd A. et al. MMWR. 2018 Feb 16;67(6):169-79.
FROM MORBIDITY AND MORTALITY WEEKLY REPORT
This is what a flu pandemic looks like
For the week ending Feb. 3, 2018, the proportion of outpatient visits for influenza-like illness (ILI) was 7.7%, which would appear to equal the mark of 7.7% set in October of 2009. The earlier 7.7%, however, is rounded down from 7.715%, while the current mark is rounded up from 7.653%, data from the CDC’s Fluview website show.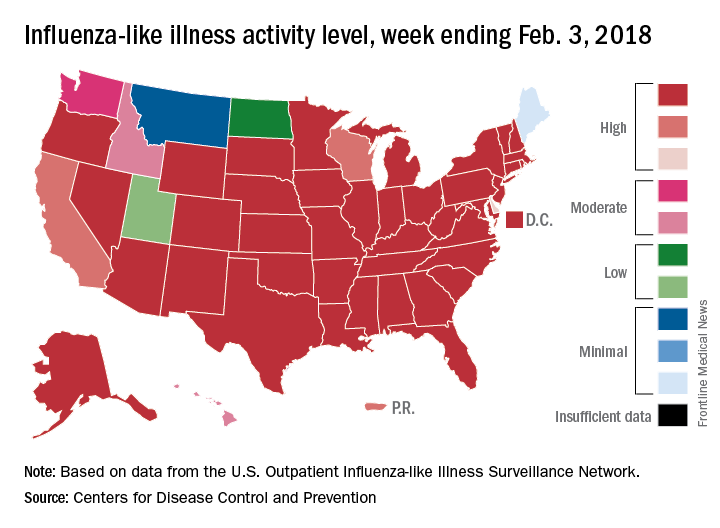
Deaths attributed to pneumonia and influenza were above the epidemic threshold set by the National Center for Health Statistics Mortality Surveillance system, acting CDC director Anne Schuchat, MD, said in a teleconference sponsored by the agency.
ILI activity was at level 10 on the CDC’s 1-10 scale in 41 states, compared with 34 the week before, and was categorized in the “high” range (levels 8-10) in another 3 states and Puerto Rico, according to data from the CDC’s Outpatient Influenza-like Illness Surveillance Network. In California, which was noted as a possible bright spot last week by Dr. Schuchat because activity there had been decreasing, the ILI level went back up to level 9 after being at 7 the week before.
Flu-related hospitalizations are continuing to rise at a record clip, with the cumulative rate for the week of Feb. 3 at 59.9 per 100,000 population, the CDC reported. A total of 1 in 10 hospital-based deaths last week were related to influenza. At this point in the 2014-2015 flu season – which has the highest number of hospitalizations at 710,000 – the hospitalization rate was only 50.9 per 100,000 population.
There were 10 pediatric deaths reported for the week ending Feb. 3, although 9 occurred in previous weeks. There have been 63 flu-related deaths among children so far during the 2017-2018 season.
Dr. Schuchat continued to recommend members of the public to get a flu shot and to stay home if they are feeling sick.
“What could be mild symptoms for you could be deadly for someone else,” Dr. Schuchat said, adding that antiviral medications remain important. “Physicians do not have to wait for confirmatory flu testing. They should begin treatment with antiviral drugs immediately in they suspect they have a severely ill or a high risk patient.”
“Flu vaccines often have lower effectiveness against H3N1 viruses. However, some protection is better than none. The vaccine’s effectiveness against other flu viruses, like B and H1N1, is better. Because of the ongoing intensity of the flu season and the increasing circulation of influenza B and h1n1, we do continue to recommend vaccination even this late in the season.”
Dr. Schuchat stressed the importance of the pneumococcal pneumonia vaccine. “Flu can make people more vulnerable to secondary infections like bacterial pneumonia. We recommend people aged 65 and over get a pneumococcal pneumonia vaccine,” she said.
For the week ending Feb. 3, 2018, the proportion of outpatient visits for influenza-like illness (ILI) was 7.7%, which would appear to equal the mark of 7.7% set in October of 2009. The earlier 7.7%, however, is rounded down from 7.715%, while the current mark is rounded up from 7.653%, data from the CDC’s Fluview website show.
Deaths attributed to pneumonia and influenza were above the epidemic threshold set by the National Center for Health Statistics Mortality Surveillance system, acting CDC director Anne Schuchat, MD, said in a teleconference sponsored by the agency.
ILI activity was at level 10 on the CDC’s 1-10 scale in 41 states, compared with 34 the week before, and was categorized in the “high” range (levels 8-10) in another 3 states and Puerto Rico, according to data from the CDC’s Outpatient Influenza-like Illness Surveillance Network. In California, which was noted as a possible bright spot last week by Dr. Schuchat because activity there had been decreasing, the ILI level went back up to level 9 after being at 7 the week before.
Flu-related hospitalizations are continuing to rise at a record clip, with the cumulative rate for the week of Feb. 3 at 59.9 per 100,000 population, the CDC reported. A total of 1 in 10 hospital-based deaths last week were related to influenza. At this point in the 2014-2015 flu season – which has the highest number of hospitalizations at 710,000 – the hospitalization rate was only 50.9 per 100,000 population.
There were 10 pediatric deaths reported for the week ending Feb. 3, although 9 occurred in previous weeks. There have been 63 flu-related deaths among children so far during the 2017-2018 season.
Dr. Schuchat continued to recommend members of the public to get a flu shot and to stay home if they are feeling sick.
“What could be mild symptoms for you could be deadly for someone else,” Dr. Schuchat said, adding that antiviral medications remain important. “Physicians do not have to wait for confirmatory flu testing. They should begin treatment with antiviral drugs immediately in they suspect they have a severely ill or a high risk patient.”
“Flu vaccines often have lower effectiveness against H3N1 viruses. However, some protection is better than none. The vaccine’s effectiveness against other flu viruses, like B and H1N1, is better. Because of the ongoing intensity of the flu season and the increasing circulation of influenza B and h1n1, we do continue to recommend vaccination even this late in the season.”
Dr. Schuchat stressed the importance of the pneumococcal pneumonia vaccine. “Flu can make people more vulnerable to secondary infections like bacterial pneumonia. We recommend people aged 65 and over get a pneumococcal pneumonia vaccine,” she said.
For the week ending Feb. 3, 2018, the proportion of outpatient visits for influenza-like illness (ILI) was 7.7%, which would appear to equal the mark of 7.7% set in October of 2009. The earlier 7.7%, however, is rounded down from 7.715%, while the current mark is rounded up from 7.653%, data from the CDC’s Fluview website show.
Deaths attributed to pneumonia and influenza were above the epidemic threshold set by the National Center for Health Statistics Mortality Surveillance system, acting CDC director Anne Schuchat, MD, said in a teleconference sponsored by the agency.
ILI activity was at level 10 on the CDC’s 1-10 scale in 41 states, compared with 34 the week before, and was categorized in the “high” range (levels 8-10) in another 3 states and Puerto Rico, according to data from the CDC’s Outpatient Influenza-like Illness Surveillance Network. In California, which was noted as a possible bright spot last week by Dr. Schuchat because activity there had been decreasing, the ILI level went back up to level 9 after being at 7 the week before.
Flu-related hospitalizations are continuing to rise at a record clip, with the cumulative rate for the week of Feb. 3 at 59.9 per 100,000 population, the CDC reported. A total of 1 in 10 hospital-based deaths last week were related to influenza. At this point in the 2014-2015 flu season – which has the highest number of hospitalizations at 710,000 – the hospitalization rate was only 50.9 per 100,000 population.
There were 10 pediatric deaths reported for the week ending Feb. 3, although 9 occurred in previous weeks. There have been 63 flu-related deaths among children so far during the 2017-2018 season.
Dr. Schuchat continued to recommend members of the public to get a flu shot and to stay home if they are feeling sick.
“What could be mild symptoms for you could be deadly for someone else,” Dr. Schuchat said, adding that antiviral medications remain important. “Physicians do not have to wait for confirmatory flu testing. They should begin treatment with antiviral drugs immediately in they suspect they have a severely ill or a high risk patient.”
“Flu vaccines often have lower effectiveness against H3N1 viruses. However, some protection is better than none. The vaccine’s effectiveness against other flu viruses, like B and H1N1, is better. Because of the ongoing intensity of the flu season and the increasing circulation of influenza B and h1n1, we do continue to recommend vaccination even this late in the season.”
Dr. Schuchat stressed the importance of the pneumococcal pneumonia vaccine. “Flu can make people more vulnerable to secondary infections like bacterial pneumonia. We recommend people aged 65 and over get a pneumococcal pneumonia vaccine,” she said.
FROM A CDC TELECONFERENCE