User login
Lucas Franki is an associate editor for MDedge News, and has been with the company since 2014. He has a BA in English from Penn State University and is an Eagle Scout.
FDA approves low-dose meloxicam medication for osteoarthritis
The Food and Drug Administration has approved a low-dose formulation of meloxicam for the treatment of osteoarthritis pain, according to manufacturer, Iroko Pharmaceuticals.
The new medication, Vivlodex, is made using a proprietary technology for producing submicron-size particles of meloxicam that are 10 times smaller than their traditional size, decreasing dissolution time. In a 12-week, phase III trial of 402 osteoarthritis patients aged 40 years or older, Vivlodex in 5- and 10-mg doses achieved efficacy at 33% lower doses than with commercially available meloxicam medications. Vivlodex will be available as a once-daily medication in 5- or 10-mg doses.
The FDA and professional medical associations have recommended administering nonsteroidal anti-inflammatory drugs in as small a dose and for as short a duration as possible. Serious adverse events stemming from NSAID treatments are related to dose and risk can increase as treatment continues.
“Meloxicam is the second most commonly prescribed NSAID in the [United States]. The approval of Vivlodex is a welcome option that offers patients an effective, low-dose NSAID,” Dr. Byron Cryer, associate dean at the University of Texas Southwestern Medical Center at Dallas, said in a statement from Iroko.
The Food and Drug Administration has approved a low-dose formulation of meloxicam for the treatment of osteoarthritis pain, according to manufacturer, Iroko Pharmaceuticals.
The new medication, Vivlodex, is made using a proprietary technology for producing submicron-size particles of meloxicam that are 10 times smaller than their traditional size, decreasing dissolution time. In a 12-week, phase III trial of 402 osteoarthritis patients aged 40 years or older, Vivlodex in 5- and 10-mg doses achieved efficacy at 33% lower doses than with commercially available meloxicam medications. Vivlodex will be available as a once-daily medication in 5- or 10-mg doses.
The FDA and professional medical associations have recommended administering nonsteroidal anti-inflammatory drugs in as small a dose and for as short a duration as possible. Serious adverse events stemming from NSAID treatments are related to dose and risk can increase as treatment continues.
“Meloxicam is the second most commonly prescribed NSAID in the [United States]. The approval of Vivlodex is a welcome option that offers patients an effective, low-dose NSAID,” Dr. Byron Cryer, associate dean at the University of Texas Southwestern Medical Center at Dallas, said in a statement from Iroko.
The Food and Drug Administration has approved a low-dose formulation of meloxicam for the treatment of osteoarthritis pain, according to manufacturer, Iroko Pharmaceuticals.
The new medication, Vivlodex, is made using a proprietary technology for producing submicron-size particles of meloxicam that are 10 times smaller than their traditional size, decreasing dissolution time. In a 12-week, phase III trial of 402 osteoarthritis patients aged 40 years or older, Vivlodex in 5- and 10-mg doses achieved efficacy at 33% lower doses than with commercially available meloxicam medications. Vivlodex will be available as a once-daily medication in 5- or 10-mg doses.
The FDA and professional medical associations have recommended administering nonsteroidal anti-inflammatory drugs in as small a dose and for as short a duration as possible. Serious adverse events stemming from NSAID treatments are related to dose and risk can increase as treatment continues.
“Meloxicam is the second most commonly prescribed NSAID in the [United States]. The approval of Vivlodex is a welcome option that offers patients an effective, low-dose NSAID,” Dr. Byron Cryer, associate dean at the University of Texas Southwestern Medical Center at Dallas, said in a statement from Iroko.
HHS awards $2.2 billion for HIV/AIDS care in FY 2015
Over $2.2 billion in grants from the Ryan White HIV/AIDS Program were awarded to states, cities, and local organizations in fiscal year 2015, according to the U.S. Department of Health & Human Services.
U.S. cities, states, and territories were the main recipients of Ryan White HIV/AIDS Program grants. Under Part A of the program, $624.7 million was awarded to 53 metropolitan areas for core medical support to HIV/AIDS patients. Under Part B of the program, over $1.3 billion was given to 59 states and territories for core medical support and the AIDS Drug Assistance Program.
The remaining grant money, under Parts C, D, and F of the program, went to local, community-based organizations that provide core medical and support services to HIV/AIDS patients; local organizations that provide family-oriented comprehensive care to women, children, and infants; and to programs supporting development of innovative models, technical assistance, and clinical training for people living with HIV.
“Over the last quarter century, the Ryan White HIV/AIDS Program has played a critical role in the United States’ public health response to HIV,” HHS Secretary Sylvia M. Burwell said in a related statement. “These grants will make a difference for the most vulnerable Americans who lack adequate health care coverage or financial resources to pay for treatment.”
Find the full statement on the HHS website.
Over $2.2 billion in grants from the Ryan White HIV/AIDS Program were awarded to states, cities, and local organizations in fiscal year 2015, according to the U.S. Department of Health & Human Services.
U.S. cities, states, and territories were the main recipients of Ryan White HIV/AIDS Program grants. Under Part A of the program, $624.7 million was awarded to 53 metropolitan areas for core medical support to HIV/AIDS patients. Under Part B of the program, over $1.3 billion was given to 59 states and territories for core medical support and the AIDS Drug Assistance Program.
The remaining grant money, under Parts C, D, and F of the program, went to local, community-based organizations that provide core medical and support services to HIV/AIDS patients; local organizations that provide family-oriented comprehensive care to women, children, and infants; and to programs supporting development of innovative models, technical assistance, and clinical training for people living with HIV.
“Over the last quarter century, the Ryan White HIV/AIDS Program has played a critical role in the United States’ public health response to HIV,” HHS Secretary Sylvia M. Burwell said in a related statement. “These grants will make a difference for the most vulnerable Americans who lack adequate health care coverage or financial resources to pay for treatment.”
Find the full statement on the HHS website.
Over $2.2 billion in grants from the Ryan White HIV/AIDS Program were awarded to states, cities, and local organizations in fiscal year 2015, according to the U.S. Department of Health & Human Services.
U.S. cities, states, and territories were the main recipients of Ryan White HIV/AIDS Program grants. Under Part A of the program, $624.7 million was awarded to 53 metropolitan areas for core medical support to HIV/AIDS patients. Under Part B of the program, over $1.3 billion was given to 59 states and territories for core medical support and the AIDS Drug Assistance Program.
The remaining grant money, under Parts C, D, and F of the program, went to local, community-based organizations that provide core medical and support services to HIV/AIDS patients; local organizations that provide family-oriented comprehensive care to women, children, and infants; and to programs supporting development of innovative models, technical assistance, and clinical training for people living with HIV.
“Over the last quarter century, the Ryan White HIV/AIDS Program has played a critical role in the United States’ public health response to HIV,” HHS Secretary Sylvia M. Burwell said in a related statement. “These grants will make a difference for the most vulnerable Americans who lack adequate health care coverage or financial resources to pay for treatment.”
Find the full statement on the HHS website.
Louisiana goes two for one on controlled substance prescriptions
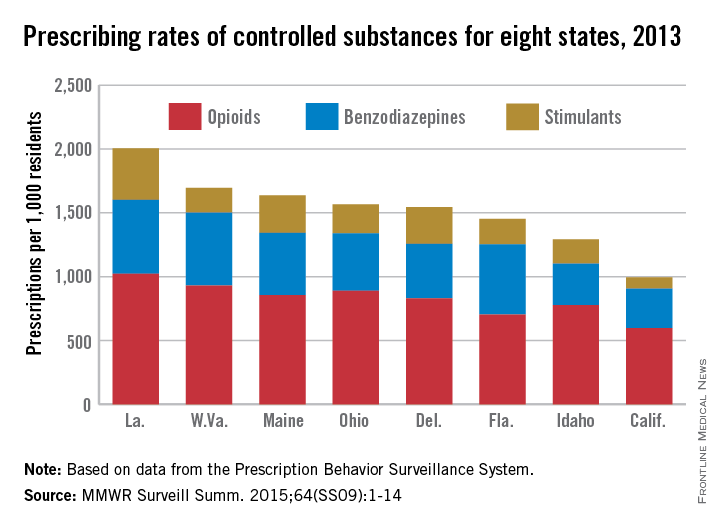
In 2013, enough controlled substances were prescribed in Louisiana – 2006.2 prescriptions per 1,000 population – that each and every person in the state could have received two, according to a report from the Centers for Disease Control and Prevention.
The combined prescribing rate for opioids, benzodiazepines, and stimulants in Louisiana topped the eight states included in the CDC report, with West Virginia in second at 1,695.7 prescriptions per 1,000. At the low end, California was the only one of the eight states where the average controlled substance prescription rate was less than one per person, but just barely at 994.8 per 1,000 people, followed by Idaho at 1,292.3 per 1,000, according to the CDC investigators in a Morbidity and Mortality Weekly Report Surveillance Summary (2015 Oct;64[SS09]:1-14).
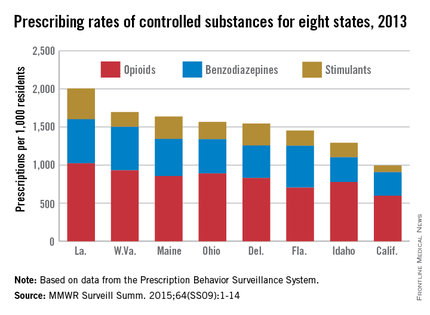
Of the three categories of controlled substances, opioids were by far the most commonly prescribed. At 1,021.7 prescriptions per 1,000 people, the opioid prescription rate in Louisiana was greater than the total controlled substance prescription rate in California. West Virginia had the second highest rate at 929.3, and California had the lowest at 596.3.
Prescription rates for stimulants and benzodiazepines were highest in Louisiana at 403.9 and 580.6 per 1,000 people, respectively. Maine had the next-highest stimulant prescription rate at 293.9, and West Virginia had the second-highest benzodiazepine prescription rate at 572.1. California had the lowest rate in both categories at 87.7 and 310.8, respectively.
Women received opioids and benzodiazepines at much higher rates than did men in every state, but stimulant-prescribing rates were higher for men in five states.
Opioid use was highest in people aged 55-64 years, though use spiked dramatically past the age of 25. People in Louisiana aged 55-64 receiving opioids had the highest controlled substance prescription rate of any measured age group, at 1,715.7 per 1,000 people. Benzodiazepine use was most common in people over 65 years, and stimulant prescriptions were highest in people younger than 18, likely because of the prevalence of childhood attention-deficit/hyperactivity disorder, the CDC investigators said.
The MMWR report used data collected by the Prescription Behavior Surveillance System. The eight states were included because they submitted data to the Prescription Behavior Surveillance System in time for the report, and they represent about one-quarter of the U.S. population.
In 2013, enough controlled substances were prescribed in Louisiana – 2006.2 prescriptions per 1,000 population – that each and every person in the state could have received two, according to a report from the Centers for Disease Control and Prevention.
The combined prescribing rate for opioids, benzodiazepines, and stimulants in Louisiana topped the eight states included in the CDC report, with West Virginia in second at 1,695.7 prescriptions per 1,000. At the low end, California was the only one of the eight states where the average controlled substance prescription rate was less than one per person, but just barely at 994.8 per 1,000 people, followed by Idaho at 1,292.3 per 1,000, according to the CDC investigators in a Morbidity and Mortality Weekly Report Surveillance Summary (2015 Oct;64[SS09]:1-14).

Of the three categories of controlled substances, opioids were by far the most commonly prescribed. At 1,021.7 prescriptions per 1,000 people, the opioid prescription rate in Louisiana was greater than the total controlled substance prescription rate in California. West Virginia had the second highest rate at 929.3, and California had the lowest at 596.3.
Prescription rates for stimulants and benzodiazepines were highest in Louisiana at 403.9 and 580.6 per 1,000 people, respectively. Maine had the next-highest stimulant prescription rate at 293.9, and West Virginia had the second-highest benzodiazepine prescription rate at 572.1. California had the lowest rate in both categories at 87.7 and 310.8, respectively.
Women received opioids and benzodiazepines at much higher rates than did men in every state, but stimulant-prescribing rates were higher for men in five states.
Opioid use was highest in people aged 55-64 years, though use spiked dramatically past the age of 25. People in Louisiana aged 55-64 receiving opioids had the highest controlled substance prescription rate of any measured age group, at 1,715.7 per 1,000 people. Benzodiazepine use was most common in people over 65 years, and stimulant prescriptions were highest in people younger than 18, likely because of the prevalence of childhood attention-deficit/hyperactivity disorder, the CDC investigators said.
The MMWR report used data collected by the Prescription Behavior Surveillance System. The eight states were included because they submitted data to the Prescription Behavior Surveillance System in time for the report, and they represent about one-quarter of the U.S. population.
In 2013, enough controlled substances were prescribed in Louisiana – 2006.2 prescriptions per 1,000 population – that each and every person in the state could have received two, according to a report from the Centers for Disease Control and Prevention.
The combined prescribing rate for opioids, benzodiazepines, and stimulants in Louisiana topped the eight states included in the CDC report, with West Virginia in second at 1,695.7 prescriptions per 1,000. At the low end, California was the only one of the eight states where the average controlled substance prescription rate was less than one per person, but just barely at 994.8 per 1,000 people, followed by Idaho at 1,292.3 per 1,000, according to the CDC investigators in a Morbidity and Mortality Weekly Report Surveillance Summary (2015 Oct;64[SS09]:1-14).

Of the three categories of controlled substances, opioids were by far the most commonly prescribed. At 1,021.7 prescriptions per 1,000 people, the opioid prescription rate in Louisiana was greater than the total controlled substance prescription rate in California. West Virginia had the second highest rate at 929.3, and California had the lowest at 596.3.
Prescription rates for stimulants and benzodiazepines were highest in Louisiana at 403.9 and 580.6 per 1,000 people, respectively. Maine had the next-highest stimulant prescription rate at 293.9, and West Virginia had the second-highest benzodiazepine prescription rate at 572.1. California had the lowest rate in both categories at 87.7 and 310.8, respectively.
Women received opioids and benzodiazepines at much higher rates than did men in every state, but stimulant-prescribing rates were higher for men in five states.
Opioid use was highest in people aged 55-64 years, though use spiked dramatically past the age of 25. People in Louisiana aged 55-64 receiving opioids had the highest controlled substance prescription rate of any measured age group, at 1,715.7 per 1,000 people. Benzodiazepine use was most common in people over 65 years, and stimulant prescriptions were highest in people younger than 18, likely because of the prevalence of childhood attention-deficit/hyperactivity disorder, the CDC investigators said.
The MMWR report used data collected by the Prescription Behavior Surveillance System. The eight states were included because they submitted data to the Prescription Behavior Surveillance System in time for the report, and they represent about one-quarter of the U.S. population.
FROM MORBIDITY AND MORTALITY WEEKLY REPORT
Effects of ADHD on the brain differ between genders
The effect of attention-deficit/hyperactivity disorder on white matter in the primary motor and medial orbitofrontal cortices differs significantly between boys and girls, according to Lisa Jacobson, Ph.D., and her associates.
For the study, 60 children aged 8-12, split evenly between boys and girls, were compared to a typically developing control group of similar size and makeup via diffusion tensor imaging. For boys with ADHD, fractional anisotropy (FA) was reduced in the primary motor cortex, compared with the control group. But FA was similar in the medial orbitofrontal cortex among the boys. For girls with ADHD, FA was similar in the primary motor cortex to that of the control group but higher in the medial orbitofrontal cortex, reported Dr. Jacobson, a psychologist and pediatric neuropsychologist at Kennedy Krieger Institute, Baltimore.
Low FA in the primary motor cortex was associated with a higher reaction time variability (CVRT) in boys, and higher commission error rates in girls. Low FA in the medial orbitofrontal cortex was associated with higher CVRT and higher commission error rates in both boys and girls.
The study results “suggest a sexually dimorphic pattern of frontal WM abnormalities in children with ADHD, such that boys show greater involvement of motor regions crucial to more basic aspects of motor response control, whereas girls show greater involvement of prefrontal regions important to top-down regulation of higher order emotional and behavioral responses,” the investigators said.
Find the study in Journal of the American Academy of Child & Adolescent Psychiatry (doi: 10.1016/j.jaac.2015.08.014).
The effect of attention-deficit/hyperactivity disorder on white matter in the primary motor and medial orbitofrontal cortices differs significantly between boys and girls, according to Lisa Jacobson, Ph.D., and her associates.
For the study, 60 children aged 8-12, split evenly between boys and girls, were compared to a typically developing control group of similar size and makeup via diffusion tensor imaging. For boys with ADHD, fractional anisotropy (FA) was reduced in the primary motor cortex, compared with the control group. But FA was similar in the medial orbitofrontal cortex among the boys. For girls with ADHD, FA was similar in the primary motor cortex to that of the control group but higher in the medial orbitofrontal cortex, reported Dr. Jacobson, a psychologist and pediatric neuropsychologist at Kennedy Krieger Institute, Baltimore.
Low FA in the primary motor cortex was associated with a higher reaction time variability (CVRT) in boys, and higher commission error rates in girls. Low FA in the medial orbitofrontal cortex was associated with higher CVRT and higher commission error rates in both boys and girls.
The study results “suggest a sexually dimorphic pattern of frontal WM abnormalities in children with ADHD, such that boys show greater involvement of motor regions crucial to more basic aspects of motor response control, whereas girls show greater involvement of prefrontal regions important to top-down regulation of higher order emotional and behavioral responses,” the investigators said.
Find the study in Journal of the American Academy of Child & Adolescent Psychiatry (doi: 10.1016/j.jaac.2015.08.014).
The effect of attention-deficit/hyperactivity disorder on white matter in the primary motor and medial orbitofrontal cortices differs significantly between boys and girls, according to Lisa Jacobson, Ph.D., and her associates.
For the study, 60 children aged 8-12, split evenly between boys and girls, were compared to a typically developing control group of similar size and makeup via diffusion tensor imaging. For boys with ADHD, fractional anisotropy (FA) was reduced in the primary motor cortex, compared with the control group. But FA was similar in the medial orbitofrontal cortex among the boys. For girls with ADHD, FA was similar in the primary motor cortex to that of the control group but higher in the medial orbitofrontal cortex, reported Dr. Jacobson, a psychologist and pediatric neuropsychologist at Kennedy Krieger Institute, Baltimore.
Low FA in the primary motor cortex was associated with a higher reaction time variability (CVRT) in boys, and higher commission error rates in girls. Low FA in the medial orbitofrontal cortex was associated with higher CVRT and higher commission error rates in both boys and girls.
The study results “suggest a sexually dimorphic pattern of frontal WM abnormalities in children with ADHD, such that boys show greater involvement of motor regions crucial to more basic aspects of motor response control, whereas girls show greater involvement of prefrontal regions important to top-down regulation of higher order emotional and behavioral responses,” the investigators said.
Find the study in Journal of the American Academy of Child & Adolescent Psychiatry (doi: 10.1016/j.jaac.2015.08.014).
Poor perception of physical health worsens bipolar symptoms
Patients with bipolar disorder who perceive their overall physical health as poor experience worse disease outcomes, according to Emily E. Bernstein of Harvard University, Cambridge, Mass., and her associates.
Data were collected from the Systematic Treatment Enhancement Program for Bipolar Disorder and were analyzed via the Short Form Health Survey (SF-36). An increased sense of role limitation increased the risk of depressive symptoms, while increased physical pain raised the risk of mania/hypomania. “Reports of specific or concrete physical limitations in daily life showed no associations with psychiatric symptoms at concurrent assessments, but did predict worse course of illness” 1 year later, the researchers wrote.
“Though further research is warranted, changes in subjective physical health–related quality of life, even independent of objective health changes, may offer important insight into global well-being and be targets of psychotherapy treatment,” they concluded.
Find the study in the Journal of Affective Disorders (doi: 10.1016/j.jad.2015.09.052).
Patients with bipolar disorder who perceive their overall physical health as poor experience worse disease outcomes, according to Emily E. Bernstein of Harvard University, Cambridge, Mass., and her associates.
Data were collected from the Systematic Treatment Enhancement Program for Bipolar Disorder and were analyzed via the Short Form Health Survey (SF-36). An increased sense of role limitation increased the risk of depressive symptoms, while increased physical pain raised the risk of mania/hypomania. “Reports of specific or concrete physical limitations in daily life showed no associations with psychiatric symptoms at concurrent assessments, but did predict worse course of illness” 1 year later, the researchers wrote.
“Though further research is warranted, changes in subjective physical health–related quality of life, even independent of objective health changes, may offer important insight into global well-being and be targets of psychotherapy treatment,” they concluded.
Find the study in the Journal of Affective Disorders (doi: 10.1016/j.jad.2015.09.052).
Patients with bipolar disorder who perceive their overall physical health as poor experience worse disease outcomes, according to Emily E. Bernstein of Harvard University, Cambridge, Mass., and her associates.
Data were collected from the Systematic Treatment Enhancement Program for Bipolar Disorder and were analyzed via the Short Form Health Survey (SF-36). An increased sense of role limitation increased the risk of depressive symptoms, while increased physical pain raised the risk of mania/hypomania. “Reports of specific or concrete physical limitations in daily life showed no associations with psychiatric symptoms at concurrent assessments, but did predict worse course of illness” 1 year later, the researchers wrote.
“Though further research is warranted, changes in subjective physical health–related quality of life, even independent of objective health changes, may offer important insight into global well-being and be targets of psychotherapy treatment,” they concluded.
Find the study in the Journal of Affective Disorders (doi: 10.1016/j.jad.2015.09.052).
Dupilumab effective for atopic dermatitis
Dupilumab, an investigational monoclonal antibody directed against interleukin-4 and interleukin-13, was effective for treatment of moderate to severe atopic dermatitis in adults, and higher doses were associated with better outcomes, according to Dr. Diamant Thaçi, and associates.
In the international, randomized, placebo-controlled study, 379 patients were split into six groups and treated for 16 weeks, receiving either 300 mg dupilumab once a week, 300 mg every 2 weeks, 200 mg every 2 weeks, 300 mg every 4 weeks, 100 mg every 4 weeks, or placebo.
Eczema Area and Severity Index (EASI) scores were most improved in the 300-mg-weekly group, with a reduction of 74%, followed by the 300-mg-every-2-weeks group with a 68% reduction. (Dupilumab is administered subcutaneously). EASI scores were similarly improved in the 200-mg-every-2-weeks and 300-mg-every-4-weeks groups, with reductions of 65% and 64%, respectively. The reduction was 45% in the 100-mg-every-4-weeks group and 18% in the placebo group.
Adverse events related to treatment were similar in both groups, with 80% of the placebo group and 81% of the dupilumab group reporting at least one adverse event. The most common side effect was nasopharyngitis, reported in 28% of the dupilumab group and in 26% of the placebo group.
“The emerging data with dupilumab in multiple atopic diseases provides the first compelling clinical data to support a single unifying hypothesis regarding the drivers of allergic and atopic diseases in general (i.e., that IL-4 and IL-13 are key drivers of signs and symptoms in these clinical settings), and suggests that their blockade might also be effective in other atopic settings,” the investigators concluded.
This was a dose-ranging phase IIb study. Results from phase III studies of dupilumab in adults with moderate to severe atopic dermatitis that is not adequately controlled with topical atopic dermatitis medications are expected in the first half of 2016. Dupilumab is currently under clinical development and its safety and efficacy have not been fully evaluated by any regulatory authority, according to a press statement issued by Regeneron Pharmaceuticals and Sanofi, which are developing the drug.
Find the full study in the Lancet (doi: 10.1016/S0140-6736[15]00388-8).
Dr. Thaçi is with the Comprehensive Center for Inflammation Medicine, at University Hospital Schleswig- Holstein, Campus Lübeck (Germany).
Dupilumab, an investigational monoclonal antibody directed against interleukin-4 and interleukin-13, was effective for treatment of moderate to severe atopic dermatitis in adults, and higher doses were associated with better outcomes, according to Dr. Diamant Thaçi, and associates.
In the international, randomized, placebo-controlled study, 379 patients were split into six groups and treated for 16 weeks, receiving either 300 mg dupilumab once a week, 300 mg every 2 weeks, 200 mg every 2 weeks, 300 mg every 4 weeks, 100 mg every 4 weeks, or placebo.
Eczema Area and Severity Index (EASI) scores were most improved in the 300-mg-weekly group, with a reduction of 74%, followed by the 300-mg-every-2-weeks group with a 68% reduction. (Dupilumab is administered subcutaneously). EASI scores were similarly improved in the 200-mg-every-2-weeks and 300-mg-every-4-weeks groups, with reductions of 65% and 64%, respectively. The reduction was 45% in the 100-mg-every-4-weeks group and 18% in the placebo group.
Adverse events related to treatment were similar in both groups, with 80% of the placebo group and 81% of the dupilumab group reporting at least one adverse event. The most common side effect was nasopharyngitis, reported in 28% of the dupilumab group and in 26% of the placebo group.
“The emerging data with dupilumab in multiple atopic diseases provides the first compelling clinical data to support a single unifying hypothesis regarding the drivers of allergic and atopic diseases in general (i.e., that IL-4 and IL-13 are key drivers of signs and symptoms in these clinical settings), and suggests that their blockade might also be effective in other atopic settings,” the investigators concluded.
This was a dose-ranging phase IIb study. Results from phase III studies of dupilumab in adults with moderate to severe atopic dermatitis that is not adequately controlled with topical atopic dermatitis medications are expected in the first half of 2016. Dupilumab is currently under clinical development and its safety and efficacy have not been fully evaluated by any regulatory authority, according to a press statement issued by Regeneron Pharmaceuticals and Sanofi, which are developing the drug.
Find the full study in the Lancet (doi: 10.1016/S0140-6736[15]00388-8).
Dr. Thaçi is with the Comprehensive Center for Inflammation Medicine, at University Hospital Schleswig- Holstein, Campus Lübeck (Germany).
Dupilumab, an investigational monoclonal antibody directed against interleukin-4 and interleukin-13, was effective for treatment of moderate to severe atopic dermatitis in adults, and higher doses were associated with better outcomes, according to Dr. Diamant Thaçi, and associates.
In the international, randomized, placebo-controlled study, 379 patients were split into six groups and treated for 16 weeks, receiving either 300 mg dupilumab once a week, 300 mg every 2 weeks, 200 mg every 2 weeks, 300 mg every 4 weeks, 100 mg every 4 weeks, or placebo.
Eczema Area and Severity Index (EASI) scores were most improved in the 300-mg-weekly group, with a reduction of 74%, followed by the 300-mg-every-2-weeks group with a 68% reduction. (Dupilumab is administered subcutaneously). EASI scores were similarly improved in the 200-mg-every-2-weeks and 300-mg-every-4-weeks groups, with reductions of 65% and 64%, respectively. The reduction was 45% in the 100-mg-every-4-weeks group and 18% in the placebo group.
Adverse events related to treatment were similar in both groups, with 80% of the placebo group and 81% of the dupilumab group reporting at least one adverse event. The most common side effect was nasopharyngitis, reported in 28% of the dupilumab group and in 26% of the placebo group.
“The emerging data with dupilumab in multiple atopic diseases provides the first compelling clinical data to support a single unifying hypothesis regarding the drivers of allergic and atopic diseases in general (i.e., that IL-4 and IL-13 are key drivers of signs and symptoms in these clinical settings), and suggests that their blockade might also be effective in other atopic settings,” the investigators concluded.
This was a dose-ranging phase IIb study. Results from phase III studies of dupilumab in adults with moderate to severe atopic dermatitis that is not adequately controlled with topical atopic dermatitis medications are expected in the first half of 2016. Dupilumab is currently under clinical development and its safety and efficacy have not been fully evaluated by any regulatory authority, according to a press statement issued by Regeneron Pharmaceuticals and Sanofi, which are developing the drug.
Find the full study in the Lancet (doi: 10.1016/S0140-6736[15]00388-8).
Dr. Thaçi is with the Comprehensive Center for Inflammation Medicine, at University Hospital Schleswig- Holstein, Campus Lübeck (Germany).
Decline in antibiotic effectiveness could harm surgical, chemotherapy patients
An increase of surgical site infections (SSIs) stemming from pathogens resistant to antibiotic prophylaxis could result in thousands of infection-related deaths in surgical and chemotherapy patients, according to a new study published in the Lancet Infectious Diseases.
A total of 31 published meta-analyses of randomized or quasi–randomized controlled trials were included in the study by Dr. Ramanan Laxminarayan of the Center for Disease Dynamics, Economics & Policy in Washington, and his associates. The researchers surveyed the 10 most common surgeries in which antibiotic prophylaxis provides the greatest benefit. The infection rate in surgical patients receiving prophylaxis was 4.2%, and was 11.1% in patients who did not receive prophylaxis. Relative risk reduction for infection was least in cancer chemotherapy at 35% and greatest in pacemaker implantation at 86%.
Between 38.7% and 50.9% of SSIs and 26.8% of infections after chemotherapy are caused by antibiotic-resistant pathogens. A decrease in prophylaxis effectiveness of 10% would cause 40,000 additional infections and 2,100 additional deaths, while a decrease in effectiveness of 70% would cause 280,000 additional infections and 15,000 additional deaths.
The authors say more data are needed to establish how antibiotic prophylaxis recommendations should be modified in the context of increasing rates of resistance.
In a related comment, Dr. Joshua Wolf from St. Jude Children’s Research Hospital, Memphis, said, “To improve stewardship outcomes, we need more research that focuses on understanding impediments to appropriate antibiotic prescribing, strategies that target these impediments, resources to implement the strategies, and leadership that understands the urgency and complexity of the task. In view of the lack of progress so far, mandatory implementation of these steps could be necessary to achieve notable change.”
Find the full study in the Lancet Infectious Diseases (doi: 10.1016/S1473-3099[15]00270-4).
An increase of surgical site infections (SSIs) stemming from pathogens resistant to antibiotic prophylaxis could result in thousands of infection-related deaths in surgical and chemotherapy patients, according to a new study published in the Lancet Infectious Diseases.
A total of 31 published meta-analyses of randomized or quasi–randomized controlled trials were included in the study by Dr. Ramanan Laxminarayan of the Center for Disease Dynamics, Economics & Policy in Washington, and his associates. The researchers surveyed the 10 most common surgeries in which antibiotic prophylaxis provides the greatest benefit. The infection rate in surgical patients receiving prophylaxis was 4.2%, and was 11.1% in patients who did not receive prophylaxis. Relative risk reduction for infection was least in cancer chemotherapy at 35% and greatest in pacemaker implantation at 86%.
Between 38.7% and 50.9% of SSIs and 26.8% of infections after chemotherapy are caused by antibiotic-resistant pathogens. A decrease in prophylaxis effectiveness of 10% would cause 40,000 additional infections and 2,100 additional deaths, while a decrease in effectiveness of 70% would cause 280,000 additional infections and 15,000 additional deaths.
The authors say more data are needed to establish how antibiotic prophylaxis recommendations should be modified in the context of increasing rates of resistance.
In a related comment, Dr. Joshua Wolf from St. Jude Children’s Research Hospital, Memphis, said, “To improve stewardship outcomes, we need more research that focuses on understanding impediments to appropriate antibiotic prescribing, strategies that target these impediments, resources to implement the strategies, and leadership that understands the urgency and complexity of the task. In view of the lack of progress so far, mandatory implementation of these steps could be necessary to achieve notable change.”
Find the full study in the Lancet Infectious Diseases (doi: 10.1016/S1473-3099[15]00270-4).
An increase of surgical site infections (SSIs) stemming from pathogens resistant to antibiotic prophylaxis could result in thousands of infection-related deaths in surgical and chemotherapy patients, according to a new study published in the Lancet Infectious Diseases.
A total of 31 published meta-analyses of randomized or quasi–randomized controlled trials were included in the study by Dr. Ramanan Laxminarayan of the Center for Disease Dynamics, Economics & Policy in Washington, and his associates. The researchers surveyed the 10 most common surgeries in which antibiotic prophylaxis provides the greatest benefit. The infection rate in surgical patients receiving prophylaxis was 4.2%, and was 11.1% in patients who did not receive prophylaxis. Relative risk reduction for infection was least in cancer chemotherapy at 35% and greatest in pacemaker implantation at 86%.
Between 38.7% and 50.9% of SSIs and 26.8% of infections after chemotherapy are caused by antibiotic-resistant pathogens. A decrease in prophylaxis effectiveness of 10% would cause 40,000 additional infections and 2,100 additional deaths, while a decrease in effectiveness of 70% would cause 280,000 additional infections and 15,000 additional deaths.
The authors say more data are needed to establish how antibiotic prophylaxis recommendations should be modified in the context of increasing rates of resistance.
In a related comment, Dr. Joshua Wolf from St. Jude Children’s Research Hospital, Memphis, said, “To improve stewardship outcomes, we need more research that focuses on understanding impediments to appropriate antibiotic prescribing, strategies that target these impediments, resources to implement the strategies, and leadership that understands the urgency and complexity of the task. In view of the lack of progress so far, mandatory implementation of these steps could be necessary to achieve notable change.”
Find the full study in the Lancet Infectious Diseases (doi: 10.1016/S1473-3099[15]00270-4).
Alcohol-related cirrhosis deaths tied to state alcohol policies
States with strong policies regarding alcohol have lower rates of alcohol-related cirrhosis deaths, especially in women, according to Dr. Scott Hadland and his associates.
A 10-point increase in Alcohol Policy Scale (APS) score was associated with a 9% decrease in alcohol-related cirrhosis deaths in women; however, when American Indian (AI)/Alaska Natives (AN) were excluded, the association between APS score and cirrhosis deaths increased to 11% in both genders and to 18% in women aged 23-66. The association between cirrhosis death rate and APS score was strongest in the Northeast.
High APS scores were not as related to AI/AN cirrhosis deaths, which were significantly higher than in other populations. This is likely due to differing tribal policies regarding alcohol and the isolation of AI/AN communities, which prevents state alcohol policies from working effectively, the investigators said.
“Future studies should continue to assess the differential effects of alcohol policies on other health concerns that are linked to excessive alcohol use for specific population subgroups according to sex, race/ethnicity, and geographical location,” the investigators said.
Find the full study in Preventing Chronic Disease (doi: 10.5888/pcd12.150200).
States with strong policies regarding alcohol have lower rates of alcohol-related cirrhosis deaths, especially in women, according to Dr. Scott Hadland and his associates.
A 10-point increase in Alcohol Policy Scale (APS) score was associated with a 9% decrease in alcohol-related cirrhosis deaths in women; however, when American Indian (AI)/Alaska Natives (AN) were excluded, the association between APS score and cirrhosis deaths increased to 11% in both genders and to 18% in women aged 23-66. The association between cirrhosis death rate and APS score was strongest in the Northeast.
High APS scores were not as related to AI/AN cirrhosis deaths, which were significantly higher than in other populations. This is likely due to differing tribal policies regarding alcohol and the isolation of AI/AN communities, which prevents state alcohol policies from working effectively, the investigators said.
“Future studies should continue to assess the differential effects of alcohol policies on other health concerns that are linked to excessive alcohol use for specific population subgroups according to sex, race/ethnicity, and geographical location,” the investigators said.
Find the full study in Preventing Chronic Disease (doi: 10.5888/pcd12.150200).
States with strong policies regarding alcohol have lower rates of alcohol-related cirrhosis deaths, especially in women, according to Dr. Scott Hadland and his associates.
A 10-point increase in Alcohol Policy Scale (APS) score was associated with a 9% decrease in alcohol-related cirrhosis deaths in women; however, when American Indian (AI)/Alaska Natives (AN) were excluded, the association between APS score and cirrhosis deaths increased to 11% in both genders and to 18% in women aged 23-66. The association between cirrhosis death rate and APS score was strongest in the Northeast.
High APS scores were not as related to AI/AN cirrhosis deaths, which were significantly higher than in other populations. This is likely due to differing tribal policies regarding alcohol and the isolation of AI/AN communities, which prevents state alcohol policies from working effectively, the investigators said.
“Future studies should continue to assess the differential effects of alcohol policies on other health concerns that are linked to excessive alcohol use for specific population subgroups according to sex, race/ethnicity, and geographical location,” the investigators said.
Find the full study in Preventing Chronic Disease (doi: 10.5888/pcd12.150200).
Children with metabolic syndrome at higher risk for diabetes
Adults who had metabolic syndrome when they were children were at higher risk for developing type 2 diabetes, especially if the disease was more severe, according to Dr. Mark D. DeBoer of the University of Virginia, Charlottesville, and Matthew J. Gurka, Ph.D., of West Virginia University, Morgantown, and their associates.
A total of 354 people completed the study, which started during 1973-1976 and had two follow-ups, one during 1998-2003 and the second from 2010 to 2014. Average age at baseline was 12.9 years, and average age at study completion was 49.6 years.
Among the individuals who had metabolic syndrome (MetS) at baseline, the odds ratio for developing diabetes at the first follow-up was 4.4, and the OR for diabetes at the second follow-up was 7.8. For each unit increase in MetS severity at baseline, the OR for diabetes development was 2.7 at first follow-up and 2.8 at second follow-up.
“These data provide evidence for a role for MetS severity as a marker of disease risk and suggest potential clinical utility in following MetS severity over time,” the investigators noted. They had no disclosures to report.
Find the full study in Diabetologia (doi: 10.1007/s00125-015-3759-5).
Adults who had metabolic syndrome when they were children were at higher risk for developing type 2 diabetes, especially if the disease was more severe, according to Dr. Mark D. DeBoer of the University of Virginia, Charlottesville, and Matthew J. Gurka, Ph.D., of West Virginia University, Morgantown, and their associates.
A total of 354 people completed the study, which started during 1973-1976 and had two follow-ups, one during 1998-2003 and the second from 2010 to 2014. Average age at baseline was 12.9 years, and average age at study completion was 49.6 years.
Among the individuals who had metabolic syndrome (MetS) at baseline, the odds ratio for developing diabetes at the first follow-up was 4.4, and the OR for diabetes at the second follow-up was 7.8. For each unit increase in MetS severity at baseline, the OR for diabetes development was 2.7 at first follow-up and 2.8 at second follow-up.
“These data provide evidence for a role for MetS severity as a marker of disease risk and suggest potential clinical utility in following MetS severity over time,” the investigators noted. They had no disclosures to report.
Find the full study in Diabetologia (doi: 10.1007/s00125-015-3759-5).
Adults who had metabolic syndrome when they were children were at higher risk for developing type 2 diabetes, especially if the disease was more severe, according to Dr. Mark D. DeBoer of the University of Virginia, Charlottesville, and Matthew J. Gurka, Ph.D., of West Virginia University, Morgantown, and their associates.
A total of 354 people completed the study, which started during 1973-1976 and had two follow-ups, one during 1998-2003 and the second from 2010 to 2014. Average age at baseline was 12.9 years, and average age at study completion was 49.6 years.
Among the individuals who had metabolic syndrome (MetS) at baseline, the odds ratio for developing diabetes at the first follow-up was 4.4, and the OR for diabetes at the second follow-up was 7.8. For each unit increase in MetS severity at baseline, the OR for diabetes development was 2.7 at first follow-up and 2.8 at second follow-up.
“These data provide evidence for a role for MetS severity as a marker of disease risk and suggest potential clinical utility in following MetS severity over time,” the investigators noted. They had no disclosures to report.
Find the full study in Diabetologia (doi: 10.1007/s00125-015-3759-5).
CDC supports new WHO guideline for HIV
The Centers for Disease Control and Prevention supports a new guideline from the World Health Organization regarding human immunodeficiency virus treatment and prevention, according to a press release.
The Early Release HIV Treatment and Pre-Exposure Prophylaxis guideline now allows for antiretroviral treatment (ART) in all people living with HIV. Previously, the scope of ART was limited to those with compromised immune systems (CD4 levels lower than 500) and to vulnerable populations such as children, pregnant women, and tuberculosis patients. Early ART has been shown to reduce transmission rate and improve outcomes.
Studies and real-world usage have indicated that preexposure prophylaxis (PrEP) can reduce HIV risk by as much as 90% in people who take the medication regularly. The WHO guideline now recommends daily oral PrEP as an extra choice for prevention in people at an increased HIV risk.
“While the science is clear, today’s guidelines are a call to policymakers and the public health community worldwide to translate that science into action. The WHO Early Release Guidelines support countries in expanding access to HIV treatment and affirm the promise of PrEP as an important part of a comprehensive response to HIV,” the CDC noted in the press release.
Find the full press release here.
The Centers for Disease Control and Prevention supports a new guideline from the World Health Organization regarding human immunodeficiency virus treatment and prevention, according to a press release.
The Early Release HIV Treatment and Pre-Exposure Prophylaxis guideline now allows for antiretroviral treatment (ART) in all people living with HIV. Previously, the scope of ART was limited to those with compromised immune systems (CD4 levels lower than 500) and to vulnerable populations such as children, pregnant women, and tuberculosis patients. Early ART has been shown to reduce transmission rate and improve outcomes.
Studies and real-world usage have indicated that preexposure prophylaxis (PrEP) can reduce HIV risk by as much as 90% in people who take the medication regularly. The WHO guideline now recommends daily oral PrEP as an extra choice for prevention in people at an increased HIV risk.
“While the science is clear, today’s guidelines are a call to policymakers and the public health community worldwide to translate that science into action. The WHO Early Release Guidelines support countries in expanding access to HIV treatment and affirm the promise of PrEP as an important part of a comprehensive response to HIV,” the CDC noted in the press release.
Find the full press release here.
The Centers for Disease Control and Prevention supports a new guideline from the World Health Organization regarding human immunodeficiency virus treatment and prevention, according to a press release.
The Early Release HIV Treatment and Pre-Exposure Prophylaxis guideline now allows for antiretroviral treatment (ART) in all people living with HIV. Previously, the scope of ART was limited to those with compromised immune systems (CD4 levels lower than 500) and to vulnerable populations such as children, pregnant women, and tuberculosis patients. Early ART has been shown to reduce transmission rate and improve outcomes.
Studies and real-world usage have indicated that preexposure prophylaxis (PrEP) can reduce HIV risk by as much as 90% in people who take the medication regularly. The WHO guideline now recommends daily oral PrEP as an extra choice for prevention in people at an increased HIV risk.
“While the science is clear, today’s guidelines are a call to policymakers and the public health community worldwide to translate that science into action. The WHO Early Release Guidelines support countries in expanding access to HIV treatment and affirm the promise of PrEP as an important part of a comprehensive response to HIV,” the CDC noted in the press release.
Find the full press release here.