User login
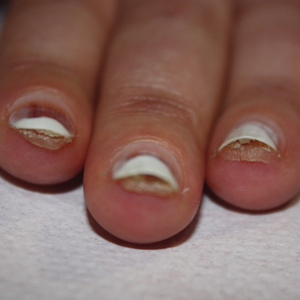
Pseudoleukonychia of the Distal Fingernails
Pseudoleukonychia of the Distal Fingernails
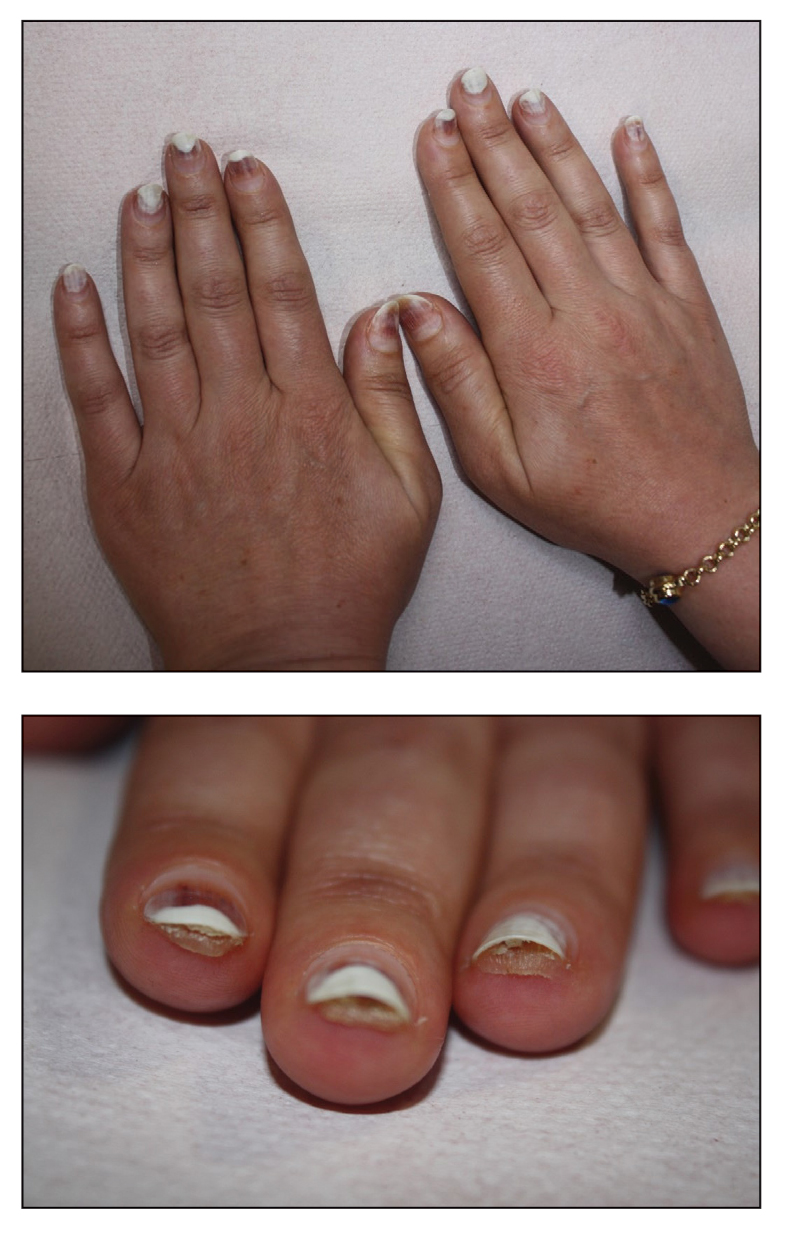
THE DIAGNOSIS: Pseudopsoriatic Nails With Pterygium Inversum Unguis
Based on the clinical findings and the patient’s history of gel manicures, a diagnosis of pseudopsoriatic nails with pterygium inversum unguis (PIU) was made. The patient was advised to avoid gel manicures and any other chemical or mechanical trauma to the nails. No other treatment was administered. Improvements including healthy nail growth and disappearing color and structure changes within the nail plates were noted at 2 months’ follow-up.
The durability and availability of gel manicures has been increasingly popular due to their ideal cosmetic results. A gel manicure involves applying a gel nail polish (GNP) containing acrylate or methacrylate monomers that harden after exposure to UV light through a photopolymerization reaction. Acrylate polymers including ethylene glycol dimethacrylate, 2-hydroxyethyl acrylate, 2-hydroxyethyl methacrylate, 2-hydroxypropyl methacrylate, methyl methacrylate, and tetrahydrofurfuryl methacrylate are known to cause allergic contact dermatitis in patients who wear acrylate-based GNP.1Hydroxyethyl methacrylate is the most common sensitizer among these acrylates. Fingertip dry dermatitis, fissured painful pulpitis of the fingers, and periungual erythema are the most common manifestations of methacrylate allergy; however, there also are reports of onycholysis and onychodystrophy in patients with severe allergic contact dermatitis caused by acrylates.2,3
In contrast to common public misconception that GNP may strengthen the nails, scientific evidence has shown otherwise. Besides allergic contact dermatitis, mechanical damage and UV-induced skin manifestations have been reported in association with GNP.1,3,4 Pseudopsoriatic nails are characterized by onycholysis accompanied by subungual hyperkeratosis, closely resembling the nail findings seen in psoriasis. This condition may occur due to mechanical damage and acrylate sensitization.2,4 Pterygium inversum unguis, also known as ventral pterygium, occurs as a result of hyponychium trauma due to either application or removal processes of GNP and/ or exposure to chemical ingredients and is one of the most striking clinical manifestations of GNP use.5 In our patient, all fingernails were affected by PIU.
Patients presenting with pseudopsoriatic nail changes and/or PIU should be questioned about potential exposure to GNP and/or sculpted nails, also known as custom artificial nails or nail prostheses. Diagnosis primarily is made clinically, but microbial cultures or skin biopsy may be required to exclude psoriasis and fungal infections in some patients. Patch testing with acrylate series in particular also is necessary in patients presenting with hand dermatitis. As it is the most common contact sensitizer in the acrylic material of the GNPs, screening for 2-hydroxyethyl methacrylate allergy is recommended in these patients.1 Almost all adverse effects related to use of GNP may be reversible upon discontinuation of exposure.
- Litaiem N, Baklouti M, Zeglaoui F. Side effects of gel nail polish: a systematic review. Clin Dermatol. 2022;40:706-715. doi:10.1016 /j.clindermatol.2022.07.008
- Engelina S, Shim TN. Atypical cases of pseudo-psoriatic nails associated with acrylate contact allergy. Contact Dermatitis. 2021; 84:342-344. doi:10.1111/cod.13741
- Draelos ZD. Nail cosmetics and adornment. Dermatol Clinics. 2021;39:351-359. doi:10.1016/j.det.2021.01.001
- Rieder EA, Tosti A. Cosmetically induced disorders of the nail with update on contemporary nail manicures. J Clin Aesthet Dermatol. 2016;9:39-44.
- Cervantes J, Sanchez M, Eber AE, et al. Pterygium inversum unguis secondary to gel polish. J Eur Acad Dermatol Venereol. 2018;32:160-163. doi:10.1111/jdv.14603
THE DIAGNOSIS: Pseudopsoriatic Nails With Pterygium Inversum Unguis
Based on the clinical findings and the patient’s history of gel manicures, a diagnosis of pseudopsoriatic nails with pterygium inversum unguis (PIU) was made. The patient was advised to avoid gel manicures and any other chemical or mechanical trauma to the nails. No other treatment was administered. Improvements including healthy nail growth and disappearing color and structure changes within the nail plates were noted at 2 months’ follow-up.
The durability and availability of gel manicures has been increasingly popular due to their ideal cosmetic results. A gel manicure involves applying a gel nail polish (GNP) containing acrylate or methacrylate monomers that harden after exposure to UV light through a photopolymerization reaction. Acrylate polymers including ethylene glycol dimethacrylate, 2-hydroxyethyl acrylate, 2-hydroxyethyl methacrylate, 2-hydroxypropyl methacrylate, methyl methacrylate, and tetrahydrofurfuryl methacrylate are known to cause allergic contact dermatitis in patients who wear acrylate-based GNP.1Hydroxyethyl methacrylate is the most common sensitizer among these acrylates. Fingertip dry dermatitis, fissured painful pulpitis of the fingers, and periungual erythema are the most common manifestations of methacrylate allergy; however, there also are reports of onycholysis and onychodystrophy in patients with severe allergic contact dermatitis caused by acrylates.2,3
In contrast to common public misconception that GNP may strengthen the nails, scientific evidence has shown otherwise. Besides allergic contact dermatitis, mechanical damage and UV-induced skin manifestations have been reported in association with GNP.1,3,4 Pseudopsoriatic nails are characterized by onycholysis accompanied by subungual hyperkeratosis, closely resembling the nail findings seen in psoriasis. This condition may occur due to mechanical damage and acrylate sensitization.2,4 Pterygium inversum unguis, also known as ventral pterygium, occurs as a result of hyponychium trauma due to either application or removal processes of GNP and/ or exposure to chemical ingredients and is one of the most striking clinical manifestations of GNP use.5 In our patient, all fingernails were affected by PIU.
Patients presenting with pseudopsoriatic nail changes and/or PIU should be questioned about potential exposure to GNP and/or sculpted nails, also known as custom artificial nails or nail prostheses. Diagnosis primarily is made clinically, but microbial cultures or skin biopsy may be required to exclude psoriasis and fungal infections in some patients. Patch testing with acrylate series in particular also is necessary in patients presenting with hand dermatitis. As it is the most common contact sensitizer in the acrylic material of the GNPs, screening for 2-hydroxyethyl methacrylate allergy is recommended in these patients.1 Almost all adverse effects related to use of GNP may be reversible upon discontinuation of exposure.
THE DIAGNOSIS: Pseudopsoriatic Nails With Pterygium Inversum Unguis
Based on the clinical findings and the patient’s history of gel manicures, a diagnosis of pseudopsoriatic nails with pterygium inversum unguis (PIU) was made. The patient was advised to avoid gel manicures and any other chemical or mechanical trauma to the nails. No other treatment was administered. Improvements including healthy nail growth and disappearing color and structure changes within the nail plates were noted at 2 months’ follow-up.
The durability and availability of gel manicures has been increasingly popular due to their ideal cosmetic results. A gel manicure involves applying a gel nail polish (GNP) containing acrylate or methacrylate monomers that harden after exposure to UV light through a photopolymerization reaction. Acrylate polymers including ethylene glycol dimethacrylate, 2-hydroxyethyl acrylate, 2-hydroxyethyl methacrylate, 2-hydroxypropyl methacrylate, methyl methacrylate, and tetrahydrofurfuryl methacrylate are known to cause allergic contact dermatitis in patients who wear acrylate-based GNP.1Hydroxyethyl methacrylate is the most common sensitizer among these acrylates. Fingertip dry dermatitis, fissured painful pulpitis of the fingers, and periungual erythema are the most common manifestations of methacrylate allergy; however, there also are reports of onycholysis and onychodystrophy in patients with severe allergic contact dermatitis caused by acrylates.2,3
In contrast to common public misconception that GNP may strengthen the nails, scientific evidence has shown otherwise. Besides allergic contact dermatitis, mechanical damage and UV-induced skin manifestations have been reported in association with GNP.1,3,4 Pseudopsoriatic nails are characterized by onycholysis accompanied by subungual hyperkeratosis, closely resembling the nail findings seen in psoriasis. This condition may occur due to mechanical damage and acrylate sensitization.2,4 Pterygium inversum unguis, also known as ventral pterygium, occurs as a result of hyponychium trauma due to either application or removal processes of GNP and/ or exposure to chemical ingredients and is one of the most striking clinical manifestations of GNP use.5 In our patient, all fingernails were affected by PIU.
Patients presenting with pseudopsoriatic nail changes and/or PIU should be questioned about potential exposure to GNP and/or sculpted nails, also known as custom artificial nails or nail prostheses. Diagnosis primarily is made clinically, but microbial cultures or skin biopsy may be required to exclude psoriasis and fungal infections in some patients. Patch testing with acrylate series in particular also is necessary in patients presenting with hand dermatitis. As it is the most common contact sensitizer in the acrylic material of the GNPs, screening for 2-hydroxyethyl methacrylate allergy is recommended in these patients.1 Almost all adverse effects related to use of GNP may be reversible upon discontinuation of exposure.
- Litaiem N, Baklouti M, Zeglaoui F. Side effects of gel nail polish: a systematic review. Clin Dermatol. 2022;40:706-715. doi:10.1016 /j.clindermatol.2022.07.008
- Engelina S, Shim TN. Atypical cases of pseudo-psoriatic nails associated with acrylate contact allergy. Contact Dermatitis. 2021; 84:342-344. doi:10.1111/cod.13741
- Draelos ZD. Nail cosmetics and adornment. Dermatol Clinics. 2021;39:351-359. doi:10.1016/j.det.2021.01.001
- Rieder EA, Tosti A. Cosmetically induced disorders of the nail with update on contemporary nail manicures. J Clin Aesthet Dermatol. 2016;9:39-44.
- Cervantes J, Sanchez M, Eber AE, et al. Pterygium inversum unguis secondary to gel polish. J Eur Acad Dermatol Venereol. 2018;32:160-163. doi:10.1111/jdv.14603
- Litaiem N, Baklouti M, Zeglaoui F. Side effects of gel nail polish: a systematic review. Clin Dermatol. 2022;40:706-715. doi:10.1016 /j.clindermatol.2022.07.008
- Engelina S, Shim TN. Atypical cases of pseudo-psoriatic nails associated with acrylate contact allergy. Contact Dermatitis. 2021; 84:342-344. doi:10.1111/cod.13741
- Draelos ZD. Nail cosmetics and adornment. Dermatol Clinics. 2021;39:351-359. doi:10.1016/j.det.2021.01.001
- Rieder EA, Tosti A. Cosmetically induced disorders of the nail with update on contemporary nail manicures. J Clin Aesthet Dermatol. 2016;9:39-44.
- Cervantes J, Sanchez M, Eber AE, et al. Pterygium inversum unguis secondary to gel polish. J Eur Acad Dermatol Venereol. 2018;32:160-163. doi:10.1111/jdv.14603
Pseudoleukonychia of the Distal Fingernails
Pseudoleukonychia of the Distal Fingernails
An otherwise healthy 36-year-old woman presented to the dermatology department for evaluation of disfiguring nail changes and subungual verrucous skin lesions of 3 weeks’ duration. A review of systems and the patient’s personal and family history were unremarkable. She denied any recent trauma or chemical exposure but noted that she had regularly been patronizing a beauty salon for gel manicures over the past year; her most recent visit was 6 weeks prior to the current presentation. She previously was treated at another dermatology clinic with local corticosteroid creams without any improvement. Dermatologic examination revealed pseudoleukonychia of the distal fingernails surrounded by an erythematous and/or haemorrhagic border. Overgrowth and adherence of the hyponychium to the nail plate also was noted in almost all the fingernails. A prior complete blood cell count and biochemistry panel were within reference range.
Leaders Helping Leaders: The American Dermatological Association as a Model of Lifelong Professional Community
Leaders Helping Leaders: The American Dermatological Association as a Model of Lifelong Professional Community
Have you ever heard of the American Dermatological Association (ADA)? While many residents may not yet be familiar with this group, its members are among the most respected leaders in dermatology. They serve as current and past presidents of influential organizations including the American Academy of Dermatology (Susan C. Taylor, MD [Philadelphia, Pennsylvania]), the American Society for Dermatologic Surgery (M. Laurin Council, MD, MBA [Creve Coeur, Missouri]), and the Association of Professors of Dermatology (Sewon Kang, MD [Baltimore, Maryland]). Others lead certification boards or serve as editors of key journals like the Journal of the American Academy of Dermatology (Dirk Elston, MD [Charleston, South Carolina]), JAMA Dermatology (Kanade Shinkai, MD [San Francisco, California], and Cutis (Vincent A. DeLeo, MD [Los Angeles, California]).
The ADA is celebrating its 150th anniversary in 2026. What makes the organization so enduring is not just its history, but its culture. The members of the ADA foster deep, long-lasting relationships, and its meetings are purposefully designed to balance structured scientific sessions with unscheduled time for reflection, conversation, and connection. That intentional design cultivates learning, innovation, and wellness.
Steven Covey’s The 7 Habits of Highly Effective People1 highlights the importance of renewal and relationship building, as does the Harvard Study of Adult Development, one of the longest-running research projects on well-being.2-4 The key conclusion? Relationships are the strongest predictors of long, healthy, and fulfilling lives, not wealth or achievement. Medical training is intense, and the emphasis often falls squarely on achievement. But the friendships you form in medical school, residency, and early career are just as formative. Membership with the ADA continues this spirit of connection throughout one’s professional life, with meetings that welcome spouses and partners and encourage engagement across generations.
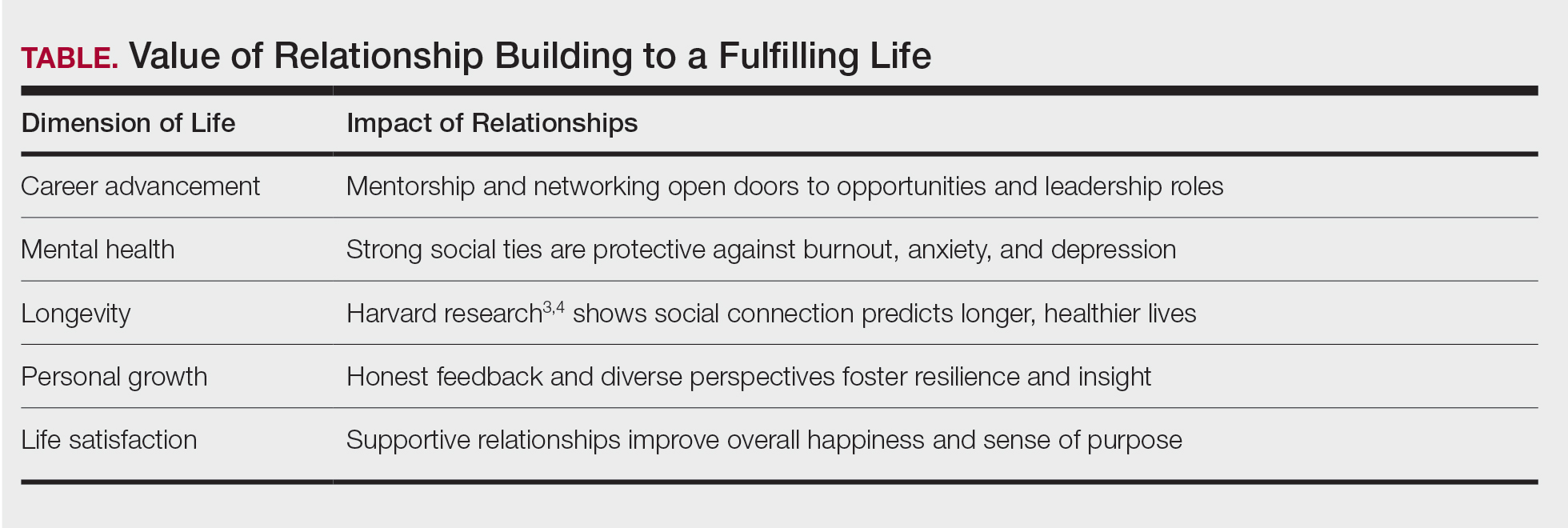
A hallmark of ADA culture is its commitment to mentoring and mutual support. Need advice about transitioning from private practice to academia? Navigating department leadership? Applying for a grant? Considering industry, editorial, or global health roles? Within the ADA, there’s someone who has done it and is eager to help. Recent meetings have addressed future-facing topics such as artificial intelligence, bedside diagnostics, workforce advocacy, and global health while also carving out time for rejuvenating activities: book clubs with best-selling authors, sessions on the arts, storytelling, wellness, and travel. This holistic programming reflects the ADA’s belief in supporting the whole physician.3 Members understand the value of relationships and appreciate these opportunities to learn about the passions and interests of their colleagues (Table).
Candidates are nominated by current members and must be board certified and at least 10 years beyond completion of their training. Members vote upon candidates in a rank voting system each year. If someone is nominated and not selected, they did not fail—they may be nominated again. The idea behind this membership process is to keep the organization small enough that members can get to know one another—there are currently 552 active members. Importantly, the ADA has embraced diversity and inclusion. While historically male- and White-dominated, recent inductee classes now reflect gender parity and a broader range of backgrounds, enriching the organization with fresh perspectives.5-8
For residents and fellows, the lesson is clear: friendships, mentorship, and time for reflection are not luxuries—they are essential. Burnout stems from relentless output in isolation; however, in cultures that prioritize renewal, authenticity, and community, physicians can flourish.9 Membership in small professional organizations is an important step towards avoiding isolation. We encourage you to be active in your local, state, and national organizations.
The ADA stands as a powerful example of how professional societies can help you build the kind of life and career you want, not just a résumé. From informal beachside conversations to high-level scientific discussions, its enduring strength is this: leaders helping others lead.
- Covey SR. The 7 Habits of Highly Effective People: Powerful Lessons in Personal Change. Simon & Schuster; 1989.
- Waldinger R, Schulz M. The Good Life: Lessons From the World’s Longest Scientific Study of Happiness. Simon & Schuster; 2023.
- Malone JC, Liu SR, Vaillant GE, et al. Midlife Eriksonian psychosocial development: setting the stage for late-life cognitive and emotional health. Dev Psychol. 2016;52:496-508. doi:10.1037/a0039875
- Vaillant GE, Milofsky E. Natural history of male psychological health: IX. Empirical evidence for Erikson’s model of the life cycle. Am J Psychiatry. 1980;137:1348-59. doi:10.1176/ajp.137.11.1348
- American Dermatological Association. Accessed August 8, 2025. https://www.ada1.org
- Pariser DM. Illustrated History of the American Dermatological Association, 1876–2020. American Dermatological Association; 2020.
- Smith JG Jr, Johnson ML. 125th anniversary of the American Dermatological Association. Arch Dermatol. 2001;137:1520. doi:10.1001/archderm.137.11.1520
- Rodriguez R, Anderson L, Woolhiser E, et al. Diversity among American Dermatological Association members by sex and geographic region. JMIR Dermatol. 2024;7:E47802. doi:10.2196/47802
- Grant A. Think Again: The Power of Knowing What You Don’t Know. Viking; 2021.
Have you ever heard of the American Dermatological Association (ADA)? While many residents may not yet be familiar with this group, its members are among the most respected leaders in dermatology. They serve as current and past presidents of influential organizations including the American Academy of Dermatology (Susan C. Taylor, MD [Philadelphia, Pennsylvania]), the American Society for Dermatologic Surgery (M. Laurin Council, MD, MBA [Creve Coeur, Missouri]), and the Association of Professors of Dermatology (Sewon Kang, MD [Baltimore, Maryland]). Others lead certification boards or serve as editors of key journals like the Journal of the American Academy of Dermatology (Dirk Elston, MD [Charleston, South Carolina]), JAMA Dermatology (Kanade Shinkai, MD [San Francisco, California], and Cutis (Vincent A. DeLeo, MD [Los Angeles, California]).
The ADA is celebrating its 150th anniversary in 2026. What makes the organization so enduring is not just its history, but its culture. The members of the ADA foster deep, long-lasting relationships, and its meetings are purposefully designed to balance structured scientific sessions with unscheduled time for reflection, conversation, and connection. That intentional design cultivates learning, innovation, and wellness.
Steven Covey’s The 7 Habits of Highly Effective People1 highlights the importance of renewal and relationship building, as does the Harvard Study of Adult Development, one of the longest-running research projects on well-being.2-4 The key conclusion? Relationships are the strongest predictors of long, healthy, and fulfilling lives, not wealth or achievement. Medical training is intense, and the emphasis often falls squarely on achievement. But the friendships you form in medical school, residency, and early career are just as formative. Membership with the ADA continues this spirit of connection throughout one’s professional life, with meetings that welcome spouses and partners and encourage engagement across generations.
A hallmark of ADA culture is its commitment to mentoring and mutual support. Need advice about transitioning from private practice to academia? Navigating department leadership? Applying for a grant? Considering industry, editorial, or global health roles? Within the ADA, there’s someone who has done it and is eager to help. Recent meetings have addressed future-facing topics such as artificial intelligence, bedside diagnostics, workforce advocacy, and global health while also carving out time for rejuvenating activities: book clubs with best-selling authors, sessions on the arts, storytelling, wellness, and travel. This holistic programming reflects the ADA’s belief in supporting the whole physician.3 Members understand the value of relationships and appreciate these opportunities to learn about the passions and interests of their colleagues (Table).

Candidates are nominated by current members and must be board certified and at least 10 years beyond completion of their training. Members vote upon candidates in a rank voting system each year. If someone is nominated and not selected, they did not fail—they may be nominated again. The idea behind this membership process is to keep the organization small enough that members can get to know one another—there are currently 552 active members. Importantly, the ADA has embraced diversity and inclusion. While historically male- and White-dominated, recent inductee classes now reflect gender parity and a broader range of backgrounds, enriching the organization with fresh perspectives.5-8
For residents and fellows, the lesson is clear: friendships, mentorship, and time for reflection are not luxuries—they are essential. Burnout stems from relentless output in isolation; however, in cultures that prioritize renewal, authenticity, and community, physicians can flourish.9 Membership in small professional organizations is an important step towards avoiding isolation. We encourage you to be active in your local, state, and national organizations.
The ADA stands as a powerful example of how professional societies can help you build the kind of life and career you want, not just a résumé. From informal beachside conversations to high-level scientific discussions, its enduring strength is this: leaders helping others lead.
Have you ever heard of the American Dermatological Association (ADA)? While many residents may not yet be familiar with this group, its members are among the most respected leaders in dermatology. They serve as current and past presidents of influential organizations including the American Academy of Dermatology (Susan C. Taylor, MD [Philadelphia, Pennsylvania]), the American Society for Dermatologic Surgery (M. Laurin Council, MD, MBA [Creve Coeur, Missouri]), and the Association of Professors of Dermatology (Sewon Kang, MD [Baltimore, Maryland]). Others lead certification boards or serve as editors of key journals like the Journal of the American Academy of Dermatology (Dirk Elston, MD [Charleston, South Carolina]), JAMA Dermatology (Kanade Shinkai, MD [San Francisco, California], and Cutis (Vincent A. DeLeo, MD [Los Angeles, California]).
The ADA is celebrating its 150th anniversary in 2026. What makes the organization so enduring is not just its history, but its culture. The members of the ADA foster deep, long-lasting relationships, and its meetings are purposefully designed to balance structured scientific sessions with unscheduled time for reflection, conversation, and connection. That intentional design cultivates learning, innovation, and wellness.
Steven Covey’s The 7 Habits of Highly Effective People1 highlights the importance of renewal and relationship building, as does the Harvard Study of Adult Development, one of the longest-running research projects on well-being.2-4 The key conclusion? Relationships are the strongest predictors of long, healthy, and fulfilling lives, not wealth or achievement. Medical training is intense, and the emphasis often falls squarely on achievement. But the friendships you form in medical school, residency, and early career are just as formative. Membership with the ADA continues this spirit of connection throughout one’s professional life, with meetings that welcome spouses and partners and encourage engagement across generations.
A hallmark of ADA culture is its commitment to mentoring and mutual support. Need advice about transitioning from private practice to academia? Navigating department leadership? Applying for a grant? Considering industry, editorial, or global health roles? Within the ADA, there’s someone who has done it and is eager to help. Recent meetings have addressed future-facing topics such as artificial intelligence, bedside diagnostics, workforce advocacy, and global health while also carving out time for rejuvenating activities: book clubs with best-selling authors, sessions on the arts, storytelling, wellness, and travel. This holistic programming reflects the ADA’s belief in supporting the whole physician.3 Members understand the value of relationships and appreciate these opportunities to learn about the passions and interests of their colleagues (Table).

Candidates are nominated by current members and must be board certified and at least 10 years beyond completion of their training. Members vote upon candidates in a rank voting system each year. If someone is nominated and not selected, they did not fail—they may be nominated again. The idea behind this membership process is to keep the organization small enough that members can get to know one another—there are currently 552 active members. Importantly, the ADA has embraced diversity and inclusion. While historically male- and White-dominated, recent inductee classes now reflect gender parity and a broader range of backgrounds, enriching the organization with fresh perspectives.5-8
For residents and fellows, the lesson is clear: friendships, mentorship, and time for reflection are not luxuries—they are essential. Burnout stems from relentless output in isolation; however, in cultures that prioritize renewal, authenticity, and community, physicians can flourish.9 Membership in small professional organizations is an important step towards avoiding isolation. We encourage you to be active in your local, state, and national organizations.
The ADA stands as a powerful example of how professional societies can help you build the kind of life and career you want, not just a résumé. From informal beachside conversations to high-level scientific discussions, its enduring strength is this: leaders helping others lead.
- Covey SR. The 7 Habits of Highly Effective People: Powerful Lessons in Personal Change. Simon & Schuster; 1989.
- Waldinger R, Schulz M. The Good Life: Lessons From the World’s Longest Scientific Study of Happiness. Simon & Schuster; 2023.
- Malone JC, Liu SR, Vaillant GE, et al. Midlife Eriksonian psychosocial development: setting the stage for late-life cognitive and emotional health. Dev Psychol. 2016;52:496-508. doi:10.1037/a0039875
- Vaillant GE, Milofsky E. Natural history of male psychological health: IX. Empirical evidence for Erikson’s model of the life cycle. Am J Psychiatry. 1980;137:1348-59. doi:10.1176/ajp.137.11.1348
- American Dermatological Association. Accessed August 8, 2025. https://www.ada1.org
- Pariser DM. Illustrated History of the American Dermatological Association, 1876–2020. American Dermatological Association; 2020.
- Smith JG Jr, Johnson ML. 125th anniversary of the American Dermatological Association. Arch Dermatol. 2001;137:1520. doi:10.1001/archderm.137.11.1520
- Rodriguez R, Anderson L, Woolhiser E, et al. Diversity among American Dermatological Association members by sex and geographic region. JMIR Dermatol. 2024;7:E47802. doi:10.2196/47802
- Grant A. Think Again: The Power of Knowing What You Don’t Know. Viking; 2021.
- Covey SR. The 7 Habits of Highly Effective People: Powerful Lessons in Personal Change. Simon & Schuster; 1989.
- Waldinger R, Schulz M. The Good Life: Lessons From the World’s Longest Scientific Study of Happiness. Simon & Schuster; 2023.
- Malone JC, Liu SR, Vaillant GE, et al. Midlife Eriksonian psychosocial development: setting the stage for late-life cognitive and emotional health. Dev Psychol. 2016;52:496-508. doi:10.1037/a0039875
- Vaillant GE, Milofsky E. Natural history of male psychological health: IX. Empirical evidence for Erikson’s model of the life cycle. Am J Psychiatry. 1980;137:1348-59. doi:10.1176/ajp.137.11.1348
- American Dermatological Association. Accessed August 8, 2025. https://www.ada1.org
- Pariser DM. Illustrated History of the American Dermatological Association, 1876–2020. American Dermatological Association; 2020.
- Smith JG Jr, Johnson ML. 125th anniversary of the American Dermatological Association. Arch Dermatol. 2001;137:1520. doi:10.1001/archderm.137.11.1520
- Rodriguez R, Anderson L, Woolhiser E, et al. Diversity among American Dermatological Association members by sex and geographic region. JMIR Dermatol. 2024;7:E47802. doi:10.2196/47802
- Grant A. Think Again: The Power of Knowing What You Don’t Know. Viking; 2021.
Leaders Helping Leaders: The American Dermatological Association as a Model of Lifelong Professional Community
Leaders Helping Leaders: The American Dermatological Association as a Model of Lifelong Professional Community
Practice Points
- Professional relationships are a critical determinant of career longevity and personal well-being. Structured opportunities for mentorship, reflection, and intergenerational dialogue within professional societies can help mitigate burnout and foster sustained leadership development.
- Medical societies serve as leadership incubators. Through selective membership, cross-organizational representation, and sustained engagement, the American Dermatological Association models how professional community strengthens the specialty beyond scientific exchange alone.
- Physicians benefit from intentional engagement in organized dermatology. Participation in local, state, and national societies cultivates mentorship networks, broadens perspective, and lays the foundation for future leadership opportunities.
Hospital Dermatology: Review of Research in 2024-2025
Hospital Dermatology: Review of Research in 2024-2025
Dermatologists play a central role in the care of hospitalized patients with skin disease. This review summarizes research from January 2024 to December 2025 on severe cutaneous adverse drug reactions, emerging infectious diseases, hidradenitis suppurativa (HS), and inpatient dermatology workforce issues. Key developments include improved recognition and management of drug reactions; updated diagnostic and prognostic tools for Stevens-Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN); and guidance for emerging infections such as measles, dengue, mpox, orthopoxviruses, and resistant dermatophytes. Evidence-based strategies for HS aim to reduce unnecessary admissions and optimize care. Workforce challenges, including limited access, high call burden, and potential for artificial intelligence (AI)–assisted diagnosis, are also highlighted. These findings emphasize the critical contributions of dermatologists to hospital-based care and provide emerging evidence to guide clinical practice.
Dermatologists play a critical role in the care of hospitalized patients. Herein, we review the research developments between January 2024 and December 2025 most relevant to the care of hospitalized patients with skin disease, including severe cutaneous adverse reactions (SCARs), emerging and re-emerging infectious diseases, hidradenitis suppurativa (HS), and access to inpatient dermatology services.
Severe Cutaneous Adverse Drug Reactions
Severe cutaneous adverse drug reactions are among the most frequent reasons for inpatient dermatology consultation. A National Inpatient Sample study identified more than 160,000 cases of drug rash with eosinophilia and systemic symptoms (DRESS syndrome) between January 2016 and December 2020.1 The overall mortality rate was 2.0%, substantially lower than the rates of up to 10% reported in earlier studies.2 Case burden and mortality peaked during the fall months, possibly due to either increased use of antibiotics or increased viral infection or reactivation during these months.1
A retrospective cohort study of patients with probable or definite DRESS syndrome showed that, among 93 patients with at least 1 viral marker tested, human herpesvirus (HHV) reactivation was found in 42% (39/93), including HHV-6 (28%)(24/85), Epstein-Barr virus (17%)(15/87), and cytomegalovirus (20%)(18/89); furthermore, viral reactivation was associated with higher 1-year mortality (odds ratio, 3.9), dialysis initiation, flares of disease, and longer hospital stay (all P<.05).1 Multiple reactivations were associated with higher inpatient mortality and 1-year mortality; however, despite apparent prognostic importance, the role of screening for viral reactivation in DRESS syndrome is undefined.
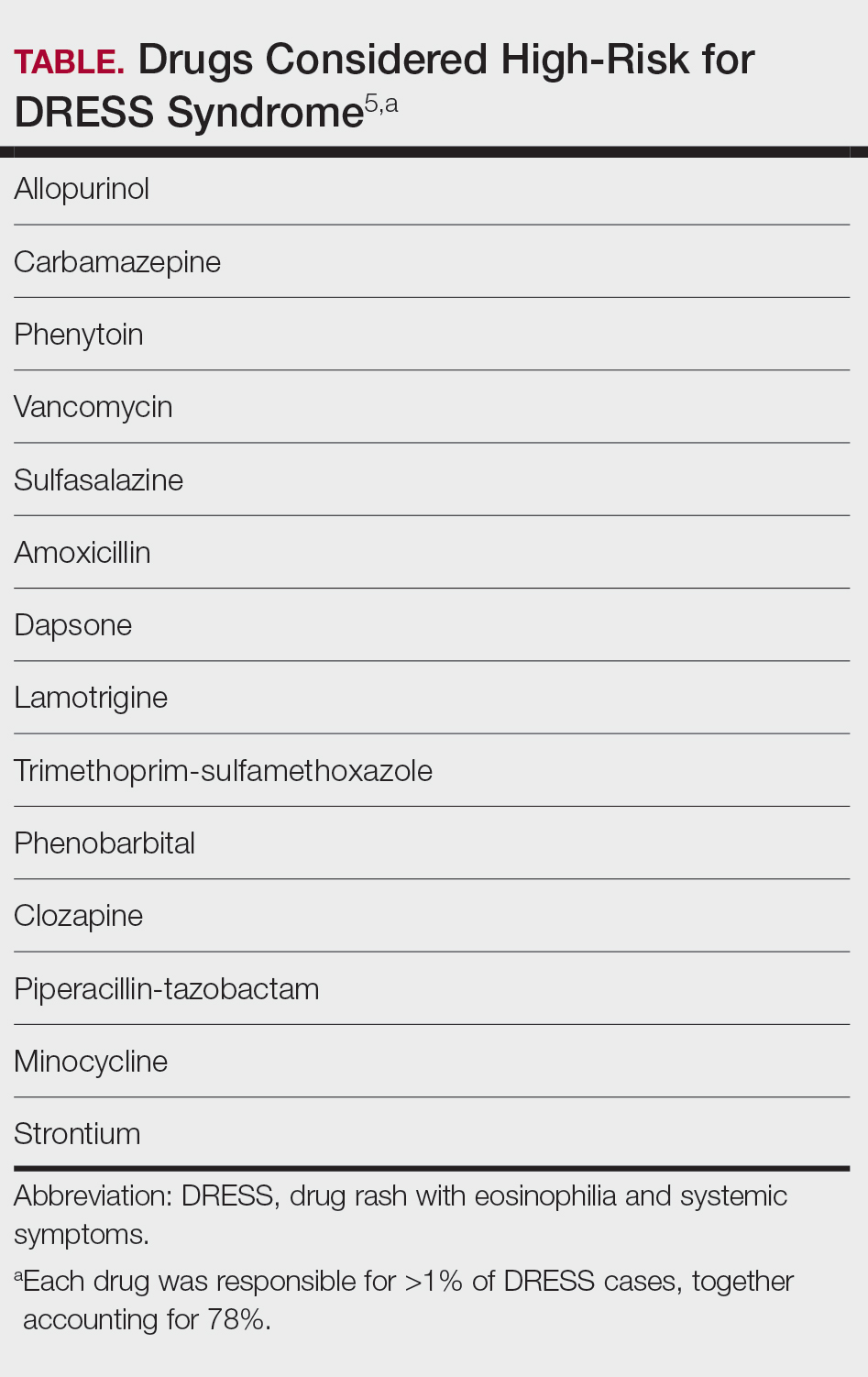
A systematic review of 610 studies including 2122 patients with DRESS syndrome demonstrated that, among 193 causal agents identified, 14 drugs accounted for more than 1% of cases each and therefore were considered high risk. Seventy-eight percent of cases were attributed to these 14 drugs (Table).5 A TriNetX Query study analyzed antibiotic exposures across SCARs and reported that sulfonamides (hazard ratio [HR], 7.5), aminoglycosides (HR, 3.7), and tetracyclines (HR, 1.7) were associated with an elevated risk for SCARs. Sulfonamides had the highest absolute incidence of SCARs, followed by cephalosporins and penicillins.6
A multicenter randomized clinical trial7 compared high-potency topical corticosteroids (clobetasol 30 g/d) to systemic corticosteroids (prednisone 0.5 mg/kg/d) for treatment of moderate DRESS syndrome. On day 30, 53.8% (14/26) of patients in the topical group had achieved remission of visceral involvement, compared to 72.0% (18/25) in the systemic group. Before day 30, 23.1% (6/26) of patients in the topical group worsened, necessitating transition to high-dose systemic steroids. When inpatient monitoring is available, low-dose systemic corticosteroids or high-potency topical steroids may be reasonable management strategies for moderate DRESS syndrome7; however, the frequent need for treatment intensification suggests limitations to this strategy.
Since prolonged courses of systemic steroids generally are necessary for management of DRESS syndrome, steroid-sparing options are needed. A retrospective case series examined interleukin 5 inhibition in patients with possible DRESS syndrome (Registry of Severe Cutaneous Adverse Reactions score ≥3). All patients demonstrated rapid eosinophil reduction within 1 to 3 days (mean [SD] time to resolution, 1.4 [0.9] days) after treatment with mepolizumab or benralizumab, with clinical improvement occurring at a mean (SD) of 16 (3.7) days (range, 13-21 days).8
A French cohort study of 1221 adult patients with Stevens-Johnson syndrome (SJS)/toxic epidermal necrolysis (TEN) reported in-hospital mortality of 19% and a total mortality of 34% at 1 year.9 Risk factors contributing to in-hospital mortality included age, history of/current diagnosis of cancer, dementia, and liver disease, while postdischarge mortality was associated with acute kidney injury and sepsis. Long-term complications included ophthalmologic and mood disorders.9
A new set of diagnostic criteria for SJS/TEN, known as the Niigata criteria,10 includes 3 main items: severe mucosal lesions in cutaneous-mucosal transition zones (eg, eyes, lips, vulva) or generalized erythema with necrotic lesions; fever of 38.5 °C or higher; and necrosis of the epidermis seen on histopathology. Because epidermal detachment involving 10% of the body surface area (BSA) is an important mortality risk predicter, SJS is defined as less than 10% BSA involvement, and TEN has been redefined as 10% or more BSA involvement (not ≥30%). A new prognostic score—clinical risk score for TEN (CRISTEN)—can be tabulated at the point of care without laboratory values. It was developed based on the 10 most important risk factors for death in a retrospective study of 382 patients, which included age 65 years or older; epidermal detachment involving 10% BSA or higher; an antibiotic as causative agent; systemic corticosteroid therapy before the onset of SJS/TEN; involvement of all 3 mucosal surfaces; and medical comorbidities such as renal impairment, diabetes, cardiac disease, active cancer, and bacterial infection.11
New potential therapeutic targets for SJS/TEN include PC111 (monoclonal antibody to Fas ligand), formyl peptide receptor 1 antagonists (which inhibit necroptosis induced by formyl peptide receptor 1–annexin A1 interaction), daratumumab (which depletes cytotoxic CD8-positive and CD38-positive T cells), and Janus kinase (JAK) inhibitors.10 Spatial proteomics showed marked enrichment of type I and type II interferon signatures as well as activation of signal transducer and activator of transcription 1. In vitro, tofacitinib reduced keratinocyte-directed cytotoxicity, and in vivo JAK inhibitors ameliorated disease severity in 2 TEN mouse models. Patients with TEN that was refractory to corticosteroid therapy received rescue treatment with JAK inhibitors and had re-epithelization within several days with marked reduction in levels of phosphorylated signal transducer and activator of transcription 1.12 Controlled studies are needed to assess the potential role of JAK inhibitors for SJS/TEN.
Emerging and Re-emerging Infectious Diseases
Dermatologists may encounter emerging or re-emerging infections, performing an essential public health role in the process. In 2025, a total of 2281 confirmed cases of measles had been reported across 45 of the United States.13 During the COVID-19 pandemic, measles vaccine coverage in the United States dropped to 93%—down from 95% to 97% prepandemic. Worldwide, 2022 saw an increase of 1.4 million measles cases (18% increase) and 41,200 excess deaths (43% increase) compared to the previous year. Complications of measles include pneumonia, blindness, otitis media, and encephalitis, with 1 in 5 (20%) unvaccinated people with measles in the United States requiring hospitalization.14 A vaccine coverage rate higher than 95% is needed to prevent community spread of disease. Since efforts to detect and rapidly isolate cases of measles are critical, dermatologists should consider measles in the differential of morbilliform eruptions with viral symptoms and ask about vaccination status.
Since 2023, dengue infection rates have tripled in the Americas, representing the highest levels recorded since tracking began in 1980. In 2024, there were more than 12 million cases, with approximately 8000 deaths reported. Ninety percent of cases occur in Argentina, Brazil, Colombia, and Mexico, but local transmission has been reported in Arizona, California, Florida, Hawaii, and Texas.15 The characteristic exanthem of dengue is diffuse erythema with islands of sparing.<
Unlike during the 2022 outbreak of mpox clade II, which predominantly impacted men who have sex with men, there now is an ongoing outbreak of mpox clades 1a and 1b in the Democratic Republic of the Congo and surrounding countries that more commonly affects children and heterosexual adults. It is also more transmissible and virulent. Cases of mpox clade I have been reported in several European countries and across the United States, mostly among travelers from areas of active transmission. Vaccination of at-risk individuals is considered effective; however, tecovirimat is not.16
Outbreaks of 2 emerging zoonotic orthopoxviruses recently have been reported. Buffalopox virus (BPXV) is transmitted via direct contact with the skin of infected cattle and buffalo as well as fomites and has been responsible for human cases in South Asia. Characteristics of BPXV include macules, umbilicated papules, vesicles, pustules, and eschars that evolve over several weeks, with a predilection for the hands and face. It can manifest with prodromal symptoms of fever, malaise, and lymphadenopathy.17 Borealpox virus (formerly known as Alaskapox) has similar manifestations. Its reservoir includes small mammals such as voles and shrews, but it also has been found in cats and dogs and has been responsible for at least one human fatality. Cidofovir may be an effective therapy for both BPXV and borealpox virus, and prior smallpox vaccination may provide protection.18 These outbreaks demonstrate the continued importance of research for more effective vaccines and therapies against smallpox and other orthopoxviruses.19 A recent review provided a detailed overview of the epidemiology, transmission, dermatologic findings, and management strategies associated with smallpox and other bioweapons.20
In 2023, a case was reported of a patient in a New York City hospital with tinea that was refractory to multiple rounds of topical antifungals, which called attention to the presence of Trichophyton indotineae in the United States.21 Since then, additional reports and case series have characterized the clinical presentation of T indotineae as widespread and atypical, refractory to traditional therapies, and most often encountered in travelers returning from Bangladesh or elsewhere in South Asia.22 The diagnosis should be confirmed via DNA testing of fungal culture. Itraconazole 100 to 200 mg/d is the antifungal therapy of choice.23
Other series have reported cases of tinea genitalis caused by Trichophyton mentagrophytes type VII seen predominately in sex workers and others engaging in high-risk sexual contact, highlighting the spread of dermatophytes through sexual activity.24-26 Lastly, it is important to culture pustules and consider atypical pathogens in patients with chronic folliculitis not responding to typical therapies such as tetracycline antibiotics. A case series reported the presence of pustules in the beard area of 7 men who have sex with men, with culture data showing Klebsiella aerogenes. Prolonged courses of fluoroquinolones were necessary for clearance.27
Reducing HS Admissions Through Evidence-Based Management
Hidradenitis suppurativa is a frequent cause of emergency department visits and hospital admissions. In an analysis of the Nationwide Readmissions Database, 17.8% (392/2204) of patients admitted to the hospital with HS were readmitted within 30 days, a number comparable to that of heart failure.28
Flaring HS can produce symptoms that mimic sepsis. A retrospective cohort study examining sepsislike features in HS showed that more than 50% (30/58) of those admitted to the hospital with an HS flare were misdiagnosed with sepsis, and more than 80% (53/64) of those patients received intravenous antibiotics.29 A National Inpatient Sample (January 2016-December 2018) study demonstrated minimal rates of true infection in patients admitted with HS flares,30 while patients with HS diagnosed as sepsis do not sustain the mortality expected from true sepsis. Improving recognition of HS and differentiation of the disease from true sepsis could decrease unnecessary antibiotic use, hospital admissions, and cost, underscoring the need for a framework to reliably and reproducibly distinguish sepsis from HS flare.31
While severe HS is difficult to manage, there may be a window of opportunity in which appropriate treatment of early disease may prevent progression and decrease inpatient utilization. A prospective cohort study of 335 biologic-naïve patients with mild to moderate HS (Hurley stages I and II) followed over a median of 2 years showed that active smoking, body mass index higher than 25, and the presence of disease in 2 or more anatomic areas were factors predictive of progression to severe disease.32
Despite high utilization of emergency and inpatient care, there has been no consensus on inpatient management of HS. A Delphi consensus exercise including 26 expert dermatologists reached consensus on 40 statements.33 Specific recommendations involve multidisciplinary care, including from a dermatologist; consideration of comorbid medical conditions; supportive care measures (wound care, pain control); evidence-based medical management, including initiation or adjustment of biologic therapies; targeted surgical intervention; nutritional support and maintenance of glycemic control; and attention to transitional care at discharge, including home health services, verification of insurance status, and timely outpatient dermatology follow-up.34 A retrospective review of 98 patients treated with intravenous ertapenem for a mean duration of 13 weeks demonstrated improvement in clinical and inflammatory markers.35 Patients with severe or treatment-refractory HS, including those admitted to the hospital, may benefit from initiation of this therapy in select circumstances.
Hospital Dermatology Workforce
Inpatient dermatology consultations are extremely valuable for improving diagnostic accuracy, reducing admissions for pseudocellulitis and inflammatory skin conditions, and keeping cancer patients on needed therapies.36-38 Despite this clear value added, a cross-sectional analysis of inpatient Medicare claims data from January 2013 to December 2019 found that the number of dermatologists performing more than 10 inpatient consults per year decreased from 356 to 281.39 Additionally, medical centers in which dermatology encounters occurred decreased from 239 to 157 during the same period. Ninety-eight percent of inpatient dermatologists were in metropolitan areas, with large regions lacking access to inpatient dermatology consultation altogether.39
A survey of Society for Pediatric Dermatology members similarly characterized the state of the pediatric dermatology workforce performing hospital consultation.40 Seventy-five percent reported a high call burden, defined as more than 11 days or nights per month, more than 1 weekend per month, and/or more than 5 hours per week seeing patients. Ninety-one percent of consultation services are based within academic institutions, reflecting disparities in access.40 A prospective cohort study of academic pediatric dermatologists reported that 310 curbside consultations were performed over 24 weeks; of these calls, 17% occurred during weeknights and 23% on weekends. None of these curbside interactions was reimbursed.41 These findings underscore the burden of uncompensated time a subset of pediatric dermatologists dedicates to inpatient consultations, highlighting the need for improved financial and administrative support and an increased number of physicians performing this role.
A survey study42 suggested that unfamiliarity with the inpatient setting, rather than medical knowledge, is the most important barrier to inpatient work among clinical dermatologists. Proposed interventions include resource guides (eg, hospital maps, pager numbers for key individuals, and protocols for urgent specimens). Reference guides and refresher courses may decrease gaps in knowledge or awareness among dermatologists in ambulatory practice.42 Another way to bolster the inpatient dermatology workforce may be to provide more guidance to qualified advanced practice providers to triage and address dermatologic emergencies.43
Artificial intelligence (AI) also has been explored as a tool for diagnosing complex dermatologic conditions. One study presented 15 published inpatient dermatology cases to 7 dermatologists. Participants were asked to formulate their top 3 differential diagnoses and were then shown AI-generated differentials and asked to submit a revised differential. Participants showed a diagnostic accuracy of 69% before seeing the AI-generated differential diagnosis and 79% after; however, in cases in which the AI differential was incorrect, diagnostic accuracy of the dermatologists decreased after being shown the AI model.44
Final Thoughts
This January 2024 to December 2025 review of research relevant to hospital dermatology highlights important developments and ongoing challenges in SCARs, emerging and re-emerging infectious diseases, HS, and the inpatient dermatology workforce. Dermatologists continue to play a critical role in the care of hospitalized patients with skin disease.
- Desai AD, Thomas C. Seasonal trends in drug reaction with eosinophilia and systemic symptoms. J Am Acad Dermatol. 2025;92:183-185.
- Wei BM, Fox LP, Kaffenberger BH, et al. Drug-induced hypersensitivity syndrome/drug reaction with eosinophilia and systemic symptoms. Part I. Epidemiology, pathogenesis, clinicopathological features, and prognosis. J Am Acad Dermatol. 2024;90:885-908. doi:10.1016/j.jaad.2023.02.072
- Chan LCE, Sultana R, Choo KJL, et al. Viral reactivation and clinical outcomes in drug reaction with eosinophilia and systemic symptoms (DRESS). Sci Rep. 2024;14:28492.
- Brüggen MC, Walsh S, Ameri MM, et al. Management of adult patients with drug reaction with eosinophilia and systemic symptoms: a Delphi-based international consensus. JAMA Dermatol. 2024;160:37-44
- Hansen E, Gallardo M, Yan A, et al. Risk assessment of drugs associated with DRESS syndrome based on publication frequency: a systematic review. J Am Acad Dermatol. 2024;91:962-966.
- Neubauer ZJK, Chan R, Singal A, et al. SCAR-ed by antibiotics: a retrospective cohort study of severe cutaneous adverse reactions (SCAR) relative risk. J Am Acad Dermatol. 2025;92:1143-1145.
- Ingen-Housz-Oro S, Guichard E, Milpied B, et al. Topical versus oral corticosteroids in moderate drug reaction with eosinophilia and systemic symptoms: a multicenter randomized clinical trial. J Am Acad Dermatol. 2024;91:544-547.
- Hijaz B, Nambudiri VE, Imadojemu S. IL-5 inhibitor treatment in drug reaction with eosinophilia and systemic symptoms. JAMA Dermatol. 2025;161:661-663.
- Bettuzzi T, Lebrun-Vignes B, Ingen-Housz-Oro S, et al. Incidence, in-hospital and long-term mortality, and sequelae of epidermal necrolysis in adults. JAMA Dermatol. 2024;160:1288-1296.
- Hama N, Aoki S, Chen CB, et al. Recent progress in Stevens-Johnson syndrome/toxic epidermal necrolysis: diagnostic criteria, pathogenesis and treatment. Br J Dermatol. 2024;192:9-18.
- Hama N, Sunaga Y, Ochiai H, et al. Development and validation of a novel score to predict mortality in Stevens-Johnson syndrome and toxic epidermal necrolysis: CRISTEN. J Allergy Clin Immunol Pract. 2023;11:3161-3168.e2.
- Nordmann TM, Anderton H, Hasegawa A, et al. Spatial proteomics identifies JAKi as treatment for a lethal skin disease. Nature. 2024;635:1001-1009.
- Centers for Disease Control and Prevention. Measles cases and outbreaks. Updated January 7, 2026. Accessed January 12, 2026. https://www.cdc.gov/measles/data-research/
- Rubin R. Despite safe and effective vaccine, measles cases and deaths increased worldwide from 2021 to 2022. JAMA. 2024;331:188-189.
- Orrall A. Dengue cases in the Americas highest recorded. JAMA. 2025;333:452.
- Harris E. As mpox cases surge in Africa, WHO declares a global emergency-here’s what to know. JAMA. 2024;332:862-864.
- Burningham KM, Hinojosa T, Cavazos A, et al. Buffalopox: an emerging cutaneous disease in humans. J Eur Acad Dermatol Venereol. 2025;39:404-406.
- Parker ER. Emergence of Alaskapox infection: what dermatologists need to know. J Am Acad Dermatol. 2024;91:397-399.
- Gostin LO, Singaravelu S, Hynes N. Smallpox readiness: modern strategies against an ancient disease. JAMA. 2024;332:873-874.
- Osborne S, Kam O, Thacker S, et al. Review of category A bioweapons with cutaneous features: epidemiology, clinical presentation, and contemporary management strategies. J Am Acad Dermatol. 2025;93:165-175.
- Caplan AS, Chaturvedi S, Zhu Y, et al. Notes from the field: first reported U.S. cases of tinea caused by Trichophyton indotineae - New York City, December 2021-March 2023. MMWR Morb Mortal Wkly Rep. 2023;72:536-537.
- McKenna M. Why the rise of this drug-resistant fungus is raising international concern. JAMA. 2024;332:859-861.
- Caplan AS, Todd GC, Zhu Y, et al. Clinical course, antifungal susceptibility, and genomic sequencing of Trichophyton indotineae. JAMA Dermatol. 2024;160:701-709.
- Jabet A, Bérot V, Chiarabini T, et al. Trichophyton mentagrophytes ITS genotype VII infections among men who have sex with men in France: an ongoing phenomenon. J Eur Acad Dermatol Venereol. 2025;39:407-415.
- Luchsinger I, Bosshard PP, Kasper RS, et al. Tinea genitalis: a new entity of sexually transmitted infection? Case series and review of the literature. Sex Transm Infect. 2015;91:493-496.
- Khurana A, Sharath S, Sardana K, et al. Therapeutic updates on the management of tinea corporis or cruris in the era of Trichophyton indotineae: separating evidence from hype-a narrative review. Indian J Dermatol. 2023;68:525-540.
- Bérot V, Monsel G, Dauendorffer JN, et al; Groupe Infectiologie Dermatologique et Infections Sexuellement Transmissibles (GrIDIST) de la Société Française de Dermatologie. Klebsiella aerogenes-related facial folliculitis in men having sex with men: a hypothetical new STI?J Eur Acad Dermatol Venereol. 2025;39:E10-E12.
- Edigin E, Kaul S, Eseaton PO, et al. At 180 days hidradenitis suppurativa readmission rate is comparable to heart failure: analysis of the Nationwide Readmissions Database. J Am Acad Dermatol. 2022;87:188-192.
- AbdelHameid D, Wang L, Mauskar MM, et al. Sepsis-like features in hidradenitis suppurativa flares requiring admission: a retrospective cohort study. J Am Acad Dermatol. 2024;90:1291-1294.
- Ehizogie E, Maghari I, Lo S, et al. Hidradenitis suppurativa, systemic inflammatory response syndrome and sepsis: a database study. Br J Dermatol. 2024;191:451-453.
- Maghari I, Abiad H, Griffin T, et al. Hidradenitis suppurativa (HS), systemic inflammatory response syndrome and sepsis, sepsis caused by HS: an empty systematic review. Br J Dermatol. 2024;191:449-450.
- Kjærsgaard Andersen R, Pedersen O, Eidsmo L, et al. Initial steps towards developing a predictive algorithm of disease progression for hidradenitis suppurativa (HS): results from a Cox proportional hazard regression analysis on disease progression among a cohort of 335 Danish patients with HS. Br J Dermatol. 2024;190:904-914.
- Needham M, Pichardo R, Alavi A, et al. Inpatient management of hidradenitis suppurativa: a Delphi consensus study. Cutis. 2024;113:251-254.
- Maskan Bermudez N, Elman SA, Kirsner RS, et al. Management of hidradenitis suppurativa in the inpatient setting: a clinical guide. Arch Dermatol Res. 2025;317:202.
- Nosrati A, Ch’en PY, Torpey ME, et al. Efficacy and durability of intravenous ertapenem therapy for recalcitrant hidradenitis suppurativa. JAMA Dermatol. 2024;160:312-318.
- Tracey EH, Forrestel A, Rosenbach M, et al. Inpatient dermatology consultation in patients with hematologic malignancies. J Am Acad Dermatol. 2016;75:835-836.
- Li DG, Xia FD, Khosravi H, et al. Outcomes of early dermatology consultation for inpatients diagnosed with cellulitis. JAMA Dermatol. 2018;154:537-543.
- Jacoby TV, Shah N, Asdourian MS, et al. Dermatology evaluation for cutaneous immune-related adverse events is associated with improved survival in cancer patients treated with checkpoint inhibition. J Am Acad Dermatol. 2023;88:711-714.
- Hydol-Smith JA, Gallardo MA, Korman A, et al. The United States dermatology inpatient workforce between 2013 and 2019: a Medicare analysis reveals contraction of the workforce and vast access deserts-a cross-sectional analysis. Arch Dermatol Res. 2024;316:103.
- Pineider JL, Rangu SA, Shaw KS, et al. Pediatric consultative dermatology: a survey of the Society for Pediatric Dermatology workforce reveals shortcomings in existing practice models of pediatric dermatology consult services in the United States. Pediatr Dermatol. 2024;41:270-274.
- Puar NK, Canty KM, Newell BD, et al. An evaluation of pediatric dermatology curbside consultations in an academic center: a prospective cohort study. J Am Acad Dermatol. 2024;90:1258-1260.
- Lau CB, Smith GP. Strategies for improving dermatologist comfort and quality of patient care in inpatient settings: a cross-sectional survey study. Arch Dermatol Res. 2024;316:575.
- Hazim AH. Empowering advanced clinical practitioners in managing acute dermatological emergencies. Br J Nurs. 2024;33:448-455.
- Macklis P, Kaffenberger B, Kirven R, et al. Dermatology diagnostic accuracy is improved by artificial intelligence-generated differential diagnoses. Int J Dermatol. 2025;64:960-962.
Dermatologists play a central role in the care of hospitalized patients with skin disease. This review summarizes research from January 2024 to December 2025 on severe cutaneous adverse drug reactions, emerging infectious diseases, hidradenitis suppurativa (HS), and inpatient dermatology workforce issues. Key developments include improved recognition and management of drug reactions; updated diagnostic and prognostic tools for Stevens-Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN); and guidance for emerging infections such as measles, dengue, mpox, orthopoxviruses, and resistant dermatophytes. Evidence-based strategies for HS aim to reduce unnecessary admissions and optimize care. Workforce challenges, including limited access, high call burden, and potential for artificial intelligence (AI)–assisted diagnosis, are also highlighted. These findings emphasize the critical contributions of dermatologists to hospital-based care and provide emerging evidence to guide clinical practice.
Dermatologists play a critical role in the care of hospitalized patients. Herein, we review the research developments between January 2024 and December 2025 most relevant to the care of hospitalized patients with skin disease, including severe cutaneous adverse reactions (SCARs), emerging and re-emerging infectious diseases, hidradenitis suppurativa (HS), and access to inpatient dermatology services.
Severe Cutaneous Adverse Drug Reactions
Severe cutaneous adverse drug reactions are among the most frequent reasons for inpatient dermatology consultation. A National Inpatient Sample study identified more than 160,000 cases of drug rash with eosinophilia and systemic symptoms (DRESS syndrome) between January 2016 and December 2020.1 The overall mortality rate was 2.0%, substantially lower than the rates of up to 10% reported in earlier studies.2 Case burden and mortality peaked during the fall months, possibly due to either increased use of antibiotics or increased viral infection or reactivation during these months.1
A retrospective cohort study of patients with probable or definite DRESS syndrome showed that, among 93 patients with at least 1 viral marker tested, human herpesvirus (HHV) reactivation was found in 42% (39/93), including HHV-6 (28%)(24/85), Epstein-Barr virus (17%)(15/87), and cytomegalovirus (20%)(18/89); furthermore, viral reactivation was associated with higher 1-year mortality (odds ratio, 3.9), dialysis initiation, flares of disease, and longer hospital stay (all P<.05).1 Multiple reactivations were associated with higher inpatient mortality and 1-year mortality; however, despite apparent prognostic importance, the role of screening for viral reactivation in DRESS syndrome is undefined.
A systematic review of 610 studies including 2122 patients with DRESS syndrome demonstrated that, among 193 causal agents identified, 14 drugs accounted for more than 1% of cases each and therefore were considered high risk. Seventy-eight percent of cases were attributed to these 14 drugs (Table).5 A TriNetX Query study analyzed antibiotic exposures across SCARs and reported that sulfonamides (hazard ratio [HR], 7.5), aminoglycosides (HR, 3.7), and tetracyclines (HR, 1.7) were associated with an elevated risk for SCARs. Sulfonamides had the highest absolute incidence of SCARs, followed by cephalosporins and penicillins.6

A multicenter randomized clinical trial7 compared high-potency topical corticosteroids (clobetasol 30 g/d) to systemic corticosteroids (prednisone 0.5 mg/kg/d) for treatment of moderate DRESS syndrome. On day 30, 53.8% (14/26) of patients in the topical group had achieved remission of visceral involvement, compared to 72.0% (18/25) in the systemic group. Before day 30, 23.1% (6/26) of patients in the topical group worsened, necessitating transition to high-dose systemic steroids. When inpatient monitoring is available, low-dose systemic corticosteroids or high-potency topical steroids may be reasonable management strategies for moderate DRESS syndrome7; however, the frequent need for treatment intensification suggests limitations to this strategy.
Since prolonged courses of systemic steroids generally are necessary for management of DRESS syndrome, steroid-sparing options are needed. A retrospective case series examined interleukin 5 inhibition in patients with possible DRESS syndrome (Registry of Severe Cutaneous Adverse Reactions score ≥3). All patients demonstrated rapid eosinophil reduction within 1 to 3 days (mean [SD] time to resolution, 1.4 [0.9] days) after treatment with mepolizumab or benralizumab, with clinical improvement occurring at a mean (SD) of 16 (3.7) days (range, 13-21 days).8
A French cohort study of 1221 adult patients with Stevens-Johnson syndrome (SJS)/toxic epidermal necrolysis (TEN) reported in-hospital mortality of 19% and a total mortality of 34% at 1 year.9 Risk factors contributing to in-hospital mortality included age, history of/current diagnosis of cancer, dementia, and liver disease, while postdischarge mortality was associated with acute kidney injury and sepsis. Long-term complications included ophthalmologic and mood disorders.9
A new set of diagnostic criteria for SJS/TEN, known as the Niigata criteria,10 includes 3 main items: severe mucosal lesions in cutaneous-mucosal transition zones (eg, eyes, lips, vulva) or generalized erythema with necrotic lesions; fever of 38.5 °C or higher; and necrosis of the epidermis seen on histopathology. Because epidermal detachment involving 10% of the body surface area (BSA) is an important mortality risk predicter, SJS is defined as less than 10% BSA involvement, and TEN has been redefined as 10% or more BSA involvement (not ≥30%). A new prognostic score—clinical risk score for TEN (CRISTEN)—can be tabulated at the point of care without laboratory values. It was developed based on the 10 most important risk factors for death in a retrospective study of 382 patients, which included age 65 years or older; epidermal detachment involving 10% BSA or higher; an antibiotic as causative agent; systemic corticosteroid therapy before the onset of SJS/TEN; involvement of all 3 mucosal surfaces; and medical comorbidities such as renal impairment, diabetes, cardiac disease, active cancer, and bacterial infection.11
New potential therapeutic targets for SJS/TEN include PC111 (monoclonal antibody to Fas ligand), formyl peptide receptor 1 antagonists (which inhibit necroptosis induced by formyl peptide receptor 1–annexin A1 interaction), daratumumab (which depletes cytotoxic CD8-positive and CD38-positive T cells), and Janus kinase (JAK) inhibitors.10 Spatial proteomics showed marked enrichment of type I and type II interferon signatures as well as activation of signal transducer and activator of transcription 1. In vitro, tofacitinib reduced keratinocyte-directed cytotoxicity, and in vivo JAK inhibitors ameliorated disease severity in 2 TEN mouse models. Patients with TEN that was refractory to corticosteroid therapy received rescue treatment with JAK inhibitors and had re-epithelization within several days with marked reduction in levels of phosphorylated signal transducer and activator of transcription 1.12 Controlled studies are needed to assess the potential role of JAK inhibitors for SJS/TEN.
Emerging and Re-emerging Infectious Diseases
Dermatologists may encounter emerging or re-emerging infections, performing an essential public health role in the process. In 2025, a total of 2281 confirmed cases of measles had been reported across 45 of the United States.13 During the COVID-19 pandemic, measles vaccine coverage in the United States dropped to 93%—down from 95% to 97% prepandemic. Worldwide, 2022 saw an increase of 1.4 million measles cases (18% increase) and 41,200 excess deaths (43% increase) compared to the previous year. Complications of measles include pneumonia, blindness, otitis media, and encephalitis, with 1 in 5 (20%) unvaccinated people with measles in the United States requiring hospitalization.14 A vaccine coverage rate higher than 95% is needed to prevent community spread of disease. Since efforts to detect and rapidly isolate cases of measles are critical, dermatologists should consider measles in the differential of morbilliform eruptions with viral symptoms and ask about vaccination status.
Since 2023, dengue infection rates have tripled in the Americas, representing the highest levels recorded since tracking began in 1980. In 2024, there were more than 12 million cases, with approximately 8000 deaths reported. Ninety percent of cases occur in Argentina, Brazil, Colombia, and Mexico, but local transmission has been reported in Arizona, California, Florida, Hawaii, and Texas.15 The characteristic exanthem of dengue is diffuse erythema with islands of sparing.<
Unlike during the 2022 outbreak of mpox clade II, which predominantly impacted men who have sex with men, there now is an ongoing outbreak of mpox clades 1a and 1b in the Democratic Republic of the Congo and surrounding countries that more commonly affects children and heterosexual adults. It is also more transmissible and virulent. Cases of mpox clade I have been reported in several European countries and across the United States, mostly among travelers from areas of active transmission. Vaccination of at-risk individuals is considered effective; however, tecovirimat is not.16
Outbreaks of 2 emerging zoonotic orthopoxviruses recently have been reported. Buffalopox virus (BPXV) is transmitted via direct contact with the skin of infected cattle and buffalo as well as fomites and has been responsible for human cases in South Asia. Characteristics of BPXV include macules, umbilicated papules, vesicles, pustules, and eschars that evolve over several weeks, with a predilection for the hands and face. It can manifest with prodromal symptoms of fever, malaise, and lymphadenopathy.17 Borealpox virus (formerly known as Alaskapox) has similar manifestations. Its reservoir includes small mammals such as voles and shrews, but it also has been found in cats and dogs and has been responsible for at least one human fatality. Cidofovir may be an effective therapy for both BPXV and borealpox virus, and prior smallpox vaccination may provide protection.18 These outbreaks demonstrate the continued importance of research for more effective vaccines and therapies against smallpox and other orthopoxviruses.19 A recent review provided a detailed overview of the epidemiology, transmission, dermatologic findings, and management strategies associated with smallpox and other bioweapons.20
In 2023, a case was reported of a patient in a New York City hospital with tinea that was refractory to multiple rounds of topical antifungals, which called attention to the presence of Trichophyton indotineae in the United States.21 Since then, additional reports and case series have characterized the clinical presentation of T indotineae as widespread and atypical, refractory to traditional therapies, and most often encountered in travelers returning from Bangladesh or elsewhere in South Asia.22 The diagnosis should be confirmed via DNA testing of fungal culture. Itraconazole 100 to 200 mg/d is the antifungal therapy of choice.23
Other series have reported cases of tinea genitalis caused by Trichophyton mentagrophytes type VII seen predominately in sex workers and others engaging in high-risk sexual contact, highlighting the spread of dermatophytes through sexual activity.24-26 Lastly, it is important to culture pustules and consider atypical pathogens in patients with chronic folliculitis not responding to typical therapies such as tetracycline antibiotics. A case series reported the presence of pustules in the beard area of 7 men who have sex with men, with culture data showing Klebsiella aerogenes. Prolonged courses of fluoroquinolones were necessary for clearance.27
Reducing HS Admissions Through Evidence-Based Management
Hidradenitis suppurativa is a frequent cause of emergency department visits and hospital admissions. In an analysis of the Nationwide Readmissions Database, 17.8% (392/2204) of patients admitted to the hospital with HS were readmitted within 30 days, a number comparable to that of heart failure.28
Flaring HS can produce symptoms that mimic sepsis. A retrospective cohort study examining sepsislike features in HS showed that more than 50% (30/58) of those admitted to the hospital with an HS flare were misdiagnosed with sepsis, and more than 80% (53/64) of those patients received intravenous antibiotics.29 A National Inpatient Sample (January 2016-December 2018) study demonstrated minimal rates of true infection in patients admitted with HS flares,30 while patients with HS diagnosed as sepsis do not sustain the mortality expected from true sepsis. Improving recognition of HS and differentiation of the disease from true sepsis could decrease unnecessary antibiotic use, hospital admissions, and cost, underscoring the need for a framework to reliably and reproducibly distinguish sepsis from HS flare.31
While severe HS is difficult to manage, there may be a window of opportunity in which appropriate treatment of early disease may prevent progression and decrease inpatient utilization. A prospective cohort study of 335 biologic-naïve patients with mild to moderate HS (Hurley stages I and II) followed over a median of 2 years showed that active smoking, body mass index higher than 25, and the presence of disease in 2 or more anatomic areas were factors predictive of progression to severe disease.32
Despite high utilization of emergency and inpatient care, there has been no consensus on inpatient management of HS. A Delphi consensus exercise including 26 expert dermatologists reached consensus on 40 statements.33 Specific recommendations involve multidisciplinary care, including from a dermatologist; consideration of comorbid medical conditions; supportive care measures (wound care, pain control); evidence-based medical management, including initiation or adjustment of biologic therapies; targeted surgical intervention; nutritional support and maintenance of glycemic control; and attention to transitional care at discharge, including home health services, verification of insurance status, and timely outpatient dermatology follow-up.34 A retrospective review of 98 patients treated with intravenous ertapenem for a mean duration of 13 weeks demonstrated improvement in clinical and inflammatory markers.35 Patients with severe or treatment-refractory HS, including those admitted to the hospital, may benefit from initiation of this therapy in select circumstances.
Hospital Dermatology Workforce
Inpatient dermatology consultations are extremely valuable for improving diagnostic accuracy, reducing admissions for pseudocellulitis and inflammatory skin conditions, and keeping cancer patients on needed therapies.36-38 Despite this clear value added, a cross-sectional analysis of inpatient Medicare claims data from January 2013 to December 2019 found that the number of dermatologists performing more than 10 inpatient consults per year decreased from 356 to 281.39 Additionally, medical centers in which dermatology encounters occurred decreased from 239 to 157 during the same period. Ninety-eight percent of inpatient dermatologists were in metropolitan areas, with large regions lacking access to inpatient dermatology consultation altogether.39
A survey of Society for Pediatric Dermatology members similarly characterized the state of the pediatric dermatology workforce performing hospital consultation.40 Seventy-five percent reported a high call burden, defined as more than 11 days or nights per month, more than 1 weekend per month, and/or more than 5 hours per week seeing patients. Ninety-one percent of consultation services are based within academic institutions, reflecting disparities in access.40 A prospective cohort study of academic pediatric dermatologists reported that 310 curbside consultations were performed over 24 weeks; of these calls, 17% occurred during weeknights and 23% on weekends. None of these curbside interactions was reimbursed.41 These findings underscore the burden of uncompensated time a subset of pediatric dermatologists dedicates to inpatient consultations, highlighting the need for improved financial and administrative support and an increased number of physicians performing this role.
A survey study42 suggested that unfamiliarity with the inpatient setting, rather than medical knowledge, is the most important barrier to inpatient work among clinical dermatologists. Proposed interventions include resource guides (eg, hospital maps, pager numbers for key individuals, and protocols for urgent specimens). Reference guides and refresher courses may decrease gaps in knowledge or awareness among dermatologists in ambulatory practice.42 Another way to bolster the inpatient dermatology workforce may be to provide more guidance to qualified advanced practice providers to triage and address dermatologic emergencies.43
Artificial intelligence (AI) also has been explored as a tool for diagnosing complex dermatologic conditions. One study presented 15 published inpatient dermatology cases to 7 dermatologists. Participants were asked to formulate their top 3 differential diagnoses and were then shown AI-generated differentials and asked to submit a revised differential. Participants showed a diagnostic accuracy of 69% before seeing the AI-generated differential diagnosis and 79% after; however, in cases in which the AI differential was incorrect, diagnostic accuracy of the dermatologists decreased after being shown the AI model.44
Final Thoughts
This January 2024 to December 2025 review of research relevant to hospital dermatology highlights important developments and ongoing challenges in SCARs, emerging and re-emerging infectious diseases, HS, and the inpatient dermatology workforce. Dermatologists continue to play a critical role in the care of hospitalized patients with skin disease.
Dermatologists play a central role in the care of hospitalized patients with skin disease. This review summarizes research from January 2024 to December 2025 on severe cutaneous adverse drug reactions, emerging infectious diseases, hidradenitis suppurativa (HS), and inpatient dermatology workforce issues. Key developments include improved recognition and management of drug reactions; updated diagnostic and prognostic tools for Stevens-Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN); and guidance for emerging infections such as measles, dengue, mpox, orthopoxviruses, and resistant dermatophytes. Evidence-based strategies for HS aim to reduce unnecessary admissions and optimize care. Workforce challenges, including limited access, high call burden, and potential for artificial intelligence (AI)–assisted diagnosis, are also highlighted. These findings emphasize the critical contributions of dermatologists to hospital-based care and provide emerging evidence to guide clinical practice.
Dermatologists play a critical role in the care of hospitalized patients. Herein, we review the research developments between January 2024 and December 2025 most relevant to the care of hospitalized patients with skin disease, including severe cutaneous adverse reactions (SCARs), emerging and re-emerging infectious diseases, hidradenitis suppurativa (HS), and access to inpatient dermatology services.
Severe Cutaneous Adverse Drug Reactions
Severe cutaneous adverse drug reactions are among the most frequent reasons for inpatient dermatology consultation. A National Inpatient Sample study identified more than 160,000 cases of drug rash with eosinophilia and systemic symptoms (DRESS syndrome) between January 2016 and December 2020.1 The overall mortality rate was 2.0%, substantially lower than the rates of up to 10% reported in earlier studies.2 Case burden and mortality peaked during the fall months, possibly due to either increased use of antibiotics or increased viral infection or reactivation during these months.1
A retrospective cohort study of patients with probable or definite DRESS syndrome showed that, among 93 patients with at least 1 viral marker tested, human herpesvirus (HHV) reactivation was found in 42% (39/93), including HHV-6 (28%)(24/85), Epstein-Barr virus (17%)(15/87), and cytomegalovirus (20%)(18/89); furthermore, viral reactivation was associated with higher 1-year mortality (odds ratio, 3.9), dialysis initiation, flares of disease, and longer hospital stay (all P<.05).1 Multiple reactivations were associated with higher inpatient mortality and 1-year mortality; however, despite apparent prognostic importance, the role of screening for viral reactivation in DRESS syndrome is undefined.
A systematic review of 610 studies including 2122 patients with DRESS syndrome demonstrated that, among 193 causal agents identified, 14 drugs accounted for more than 1% of cases each and therefore were considered high risk. Seventy-eight percent of cases were attributed to these 14 drugs (Table).5 A TriNetX Query study analyzed antibiotic exposures across SCARs and reported that sulfonamides (hazard ratio [HR], 7.5), aminoglycosides (HR, 3.7), and tetracyclines (HR, 1.7) were associated with an elevated risk for SCARs. Sulfonamides had the highest absolute incidence of SCARs, followed by cephalosporins and penicillins.6

A multicenter randomized clinical trial7 compared high-potency topical corticosteroids (clobetasol 30 g/d) to systemic corticosteroids (prednisone 0.5 mg/kg/d) for treatment of moderate DRESS syndrome. On day 30, 53.8% (14/26) of patients in the topical group had achieved remission of visceral involvement, compared to 72.0% (18/25) in the systemic group. Before day 30, 23.1% (6/26) of patients in the topical group worsened, necessitating transition to high-dose systemic steroids. When inpatient monitoring is available, low-dose systemic corticosteroids or high-potency topical steroids may be reasonable management strategies for moderate DRESS syndrome7; however, the frequent need for treatment intensification suggests limitations to this strategy.
Since prolonged courses of systemic steroids generally are necessary for management of DRESS syndrome, steroid-sparing options are needed. A retrospective case series examined interleukin 5 inhibition in patients with possible DRESS syndrome (Registry of Severe Cutaneous Adverse Reactions score ≥3). All patients demonstrated rapid eosinophil reduction within 1 to 3 days (mean [SD] time to resolution, 1.4 [0.9] days) after treatment with mepolizumab or benralizumab, with clinical improvement occurring at a mean (SD) of 16 (3.7) days (range, 13-21 days).8
A French cohort study of 1221 adult patients with Stevens-Johnson syndrome (SJS)/toxic epidermal necrolysis (TEN) reported in-hospital mortality of 19% and a total mortality of 34% at 1 year.9 Risk factors contributing to in-hospital mortality included age, history of/current diagnosis of cancer, dementia, and liver disease, while postdischarge mortality was associated with acute kidney injury and sepsis. Long-term complications included ophthalmologic and mood disorders.9
A new set of diagnostic criteria for SJS/TEN, known as the Niigata criteria,10 includes 3 main items: severe mucosal lesions in cutaneous-mucosal transition zones (eg, eyes, lips, vulva) or generalized erythema with necrotic lesions; fever of 38.5 °C or higher; and necrosis of the epidermis seen on histopathology. Because epidermal detachment involving 10% of the body surface area (BSA) is an important mortality risk predicter, SJS is defined as less than 10% BSA involvement, and TEN has been redefined as 10% or more BSA involvement (not ≥30%). A new prognostic score—clinical risk score for TEN (CRISTEN)—can be tabulated at the point of care without laboratory values. It was developed based on the 10 most important risk factors for death in a retrospective study of 382 patients, which included age 65 years or older; epidermal detachment involving 10% BSA or higher; an antibiotic as causative agent; systemic corticosteroid therapy before the onset of SJS/TEN; involvement of all 3 mucosal surfaces; and medical comorbidities such as renal impairment, diabetes, cardiac disease, active cancer, and bacterial infection.11
New potential therapeutic targets for SJS/TEN include PC111 (monoclonal antibody to Fas ligand), formyl peptide receptor 1 antagonists (which inhibit necroptosis induced by formyl peptide receptor 1–annexin A1 interaction), daratumumab (which depletes cytotoxic CD8-positive and CD38-positive T cells), and Janus kinase (JAK) inhibitors.10 Spatial proteomics showed marked enrichment of type I and type II interferon signatures as well as activation of signal transducer and activator of transcription 1. In vitro, tofacitinib reduced keratinocyte-directed cytotoxicity, and in vivo JAK inhibitors ameliorated disease severity in 2 TEN mouse models. Patients with TEN that was refractory to corticosteroid therapy received rescue treatment with JAK inhibitors and had re-epithelization within several days with marked reduction in levels of phosphorylated signal transducer and activator of transcription 1.12 Controlled studies are needed to assess the potential role of JAK inhibitors for SJS/TEN.
Emerging and Re-emerging Infectious Diseases
Dermatologists may encounter emerging or re-emerging infections, performing an essential public health role in the process. In 2025, a total of 2281 confirmed cases of measles had been reported across 45 of the United States.13 During the COVID-19 pandemic, measles vaccine coverage in the United States dropped to 93%—down from 95% to 97% prepandemic. Worldwide, 2022 saw an increase of 1.4 million measles cases (18% increase) and 41,200 excess deaths (43% increase) compared to the previous year. Complications of measles include pneumonia, blindness, otitis media, and encephalitis, with 1 in 5 (20%) unvaccinated people with measles in the United States requiring hospitalization.14 A vaccine coverage rate higher than 95% is needed to prevent community spread of disease. Since efforts to detect and rapidly isolate cases of measles are critical, dermatologists should consider measles in the differential of morbilliform eruptions with viral symptoms and ask about vaccination status.
Since 2023, dengue infection rates have tripled in the Americas, representing the highest levels recorded since tracking began in 1980. In 2024, there were more than 12 million cases, with approximately 8000 deaths reported. Ninety percent of cases occur in Argentina, Brazil, Colombia, and Mexico, but local transmission has been reported in Arizona, California, Florida, Hawaii, and Texas.15 The characteristic exanthem of dengue is diffuse erythema with islands of sparing.<
Unlike during the 2022 outbreak of mpox clade II, which predominantly impacted men who have sex with men, there now is an ongoing outbreak of mpox clades 1a and 1b in the Democratic Republic of the Congo and surrounding countries that more commonly affects children and heterosexual adults. It is also more transmissible and virulent. Cases of mpox clade I have been reported in several European countries and across the United States, mostly among travelers from areas of active transmission. Vaccination of at-risk individuals is considered effective; however, tecovirimat is not.16
Outbreaks of 2 emerging zoonotic orthopoxviruses recently have been reported. Buffalopox virus (BPXV) is transmitted via direct contact with the skin of infected cattle and buffalo as well as fomites and has been responsible for human cases in South Asia. Characteristics of BPXV include macules, umbilicated papules, vesicles, pustules, and eschars that evolve over several weeks, with a predilection for the hands and face. It can manifest with prodromal symptoms of fever, malaise, and lymphadenopathy.17 Borealpox virus (formerly known as Alaskapox) has similar manifestations. Its reservoir includes small mammals such as voles and shrews, but it also has been found in cats and dogs and has been responsible for at least one human fatality. Cidofovir may be an effective therapy for both BPXV and borealpox virus, and prior smallpox vaccination may provide protection.18 These outbreaks demonstrate the continued importance of research for more effective vaccines and therapies against smallpox and other orthopoxviruses.19 A recent review provided a detailed overview of the epidemiology, transmission, dermatologic findings, and management strategies associated with smallpox and other bioweapons.20
In 2023, a case was reported of a patient in a New York City hospital with tinea that was refractory to multiple rounds of topical antifungals, which called attention to the presence of Trichophyton indotineae in the United States.21 Since then, additional reports and case series have characterized the clinical presentation of T indotineae as widespread and atypical, refractory to traditional therapies, and most often encountered in travelers returning from Bangladesh or elsewhere in South Asia.22 The diagnosis should be confirmed via DNA testing of fungal culture. Itraconazole 100 to 200 mg/d is the antifungal therapy of choice.23
Other series have reported cases of tinea genitalis caused by Trichophyton mentagrophytes type VII seen predominately in sex workers and others engaging in high-risk sexual contact, highlighting the spread of dermatophytes through sexual activity.24-26 Lastly, it is important to culture pustules and consider atypical pathogens in patients with chronic folliculitis not responding to typical therapies such as tetracycline antibiotics. A case series reported the presence of pustules in the beard area of 7 men who have sex with men, with culture data showing Klebsiella aerogenes. Prolonged courses of fluoroquinolones were necessary for clearance.27
Reducing HS Admissions Through Evidence-Based Management
Hidradenitis suppurativa is a frequent cause of emergency department visits and hospital admissions. In an analysis of the Nationwide Readmissions Database, 17.8% (392/2204) of patients admitted to the hospital with HS were readmitted within 30 days, a number comparable to that of heart failure.28
Flaring HS can produce symptoms that mimic sepsis. A retrospective cohort study examining sepsislike features in HS showed that more than 50% (30/58) of those admitted to the hospital with an HS flare were misdiagnosed with sepsis, and more than 80% (53/64) of those patients received intravenous antibiotics.29 A National Inpatient Sample (January 2016-December 2018) study demonstrated minimal rates of true infection in patients admitted with HS flares,30 while patients with HS diagnosed as sepsis do not sustain the mortality expected from true sepsis. Improving recognition of HS and differentiation of the disease from true sepsis could decrease unnecessary antibiotic use, hospital admissions, and cost, underscoring the need for a framework to reliably and reproducibly distinguish sepsis from HS flare.31
While severe HS is difficult to manage, there may be a window of opportunity in which appropriate treatment of early disease may prevent progression and decrease inpatient utilization. A prospective cohort study of 335 biologic-naïve patients with mild to moderate HS (Hurley stages I and II) followed over a median of 2 years showed that active smoking, body mass index higher than 25, and the presence of disease in 2 or more anatomic areas were factors predictive of progression to severe disease.32
Despite high utilization of emergency and inpatient care, there has been no consensus on inpatient management of HS. A Delphi consensus exercise including 26 expert dermatologists reached consensus on 40 statements.33 Specific recommendations involve multidisciplinary care, including from a dermatologist; consideration of comorbid medical conditions; supportive care measures (wound care, pain control); evidence-based medical management, including initiation or adjustment of biologic therapies; targeted surgical intervention; nutritional support and maintenance of glycemic control; and attention to transitional care at discharge, including home health services, verification of insurance status, and timely outpatient dermatology follow-up.34 A retrospective review of 98 patients treated with intravenous ertapenem for a mean duration of 13 weeks demonstrated improvement in clinical and inflammatory markers.35 Patients with severe or treatment-refractory HS, including those admitted to the hospital, may benefit from initiation of this therapy in select circumstances.
Hospital Dermatology Workforce
Inpatient dermatology consultations are extremely valuable for improving diagnostic accuracy, reducing admissions for pseudocellulitis and inflammatory skin conditions, and keeping cancer patients on needed therapies.36-38 Despite this clear value added, a cross-sectional analysis of inpatient Medicare claims data from January 2013 to December 2019 found that the number of dermatologists performing more than 10 inpatient consults per year decreased from 356 to 281.39 Additionally, medical centers in which dermatology encounters occurred decreased from 239 to 157 during the same period. Ninety-eight percent of inpatient dermatologists were in metropolitan areas, with large regions lacking access to inpatient dermatology consultation altogether.39
A survey of Society for Pediatric Dermatology members similarly characterized the state of the pediatric dermatology workforce performing hospital consultation.40 Seventy-five percent reported a high call burden, defined as more than 11 days or nights per month, more than 1 weekend per month, and/or more than 5 hours per week seeing patients. Ninety-one percent of consultation services are based within academic institutions, reflecting disparities in access.40 A prospective cohort study of academic pediatric dermatologists reported that 310 curbside consultations were performed over 24 weeks; of these calls, 17% occurred during weeknights and 23% on weekends. None of these curbside interactions was reimbursed.41 These findings underscore the burden of uncompensated time a subset of pediatric dermatologists dedicates to inpatient consultations, highlighting the need for improved financial and administrative support and an increased number of physicians performing this role.
A survey study42 suggested that unfamiliarity with the inpatient setting, rather than medical knowledge, is the most important barrier to inpatient work among clinical dermatologists. Proposed interventions include resource guides (eg, hospital maps, pager numbers for key individuals, and protocols for urgent specimens). Reference guides and refresher courses may decrease gaps in knowledge or awareness among dermatologists in ambulatory practice.42 Another way to bolster the inpatient dermatology workforce may be to provide more guidance to qualified advanced practice providers to triage and address dermatologic emergencies.43
Artificial intelligence (AI) also has been explored as a tool for diagnosing complex dermatologic conditions. One study presented 15 published inpatient dermatology cases to 7 dermatologists. Participants were asked to formulate their top 3 differential diagnoses and were then shown AI-generated differentials and asked to submit a revised differential. Participants showed a diagnostic accuracy of 69% before seeing the AI-generated differential diagnosis and 79% after; however, in cases in which the AI differential was incorrect, diagnostic accuracy of the dermatologists decreased after being shown the AI model.44
Final Thoughts
This January 2024 to December 2025 review of research relevant to hospital dermatology highlights important developments and ongoing challenges in SCARs, emerging and re-emerging infectious diseases, HS, and the inpatient dermatology workforce. Dermatologists continue to play a critical role in the care of hospitalized patients with skin disease.
- Desai AD, Thomas C. Seasonal trends in drug reaction with eosinophilia and systemic symptoms. J Am Acad Dermatol. 2025;92:183-185.
- Wei BM, Fox LP, Kaffenberger BH, et al. Drug-induced hypersensitivity syndrome/drug reaction with eosinophilia and systemic symptoms. Part I. Epidemiology, pathogenesis, clinicopathological features, and prognosis. J Am Acad Dermatol. 2024;90:885-908. doi:10.1016/j.jaad.2023.02.072
- Chan LCE, Sultana R, Choo KJL, et al. Viral reactivation and clinical outcomes in drug reaction with eosinophilia and systemic symptoms (DRESS). Sci Rep. 2024;14:28492.
- Brüggen MC, Walsh S, Ameri MM, et al. Management of adult patients with drug reaction with eosinophilia and systemic symptoms: a Delphi-based international consensus. JAMA Dermatol. 2024;160:37-44
- Hansen E, Gallardo M, Yan A, et al. Risk assessment of drugs associated with DRESS syndrome based on publication frequency: a systematic review. J Am Acad Dermatol. 2024;91:962-966.
- Neubauer ZJK, Chan R, Singal A, et al. SCAR-ed by antibiotics: a retrospective cohort study of severe cutaneous adverse reactions (SCAR) relative risk. J Am Acad Dermatol. 2025;92:1143-1145.
- Ingen-Housz-Oro S, Guichard E, Milpied B, et al. Topical versus oral corticosteroids in moderate drug reaction with eosinophilia and systemic symptoms: a multicenter randomized clinical trial. J Am Acad Dermatol. 2024;91:544-547.
- Hijaz B, Nambudiri VE, Imadojemu S. IL-5 inhibitor treatment in drug reaction with eosinophilia and systemic symptoms. JAMA Dermatol. 2025;161:661-663.
- Bettuzzi T, Lebrun-Vignes B, Ingen-Housz-Oro S, et al. Incidence, in-hospital and long-term mortality, and sequelae of epidermal necrolysis in adults. JAMA Dermatol. 2024;160:1288-1296.
- Hama N, Aoki S, Chen CB, et al. Recent progress in Stevens-Johnson syndrome/toxic epidermal necrolysis: diagnostic criteria, pathogenesis and treatment. Br J Dermatol. 2024;192:9-18.
- Hama N, Sunaga Y, Ochiai H, et al. Development and validation of a novel score to predict mortality in Stevens-Johnson syndrome and toxic epidermal necrolysis: CRISTEN. J Allergy Clin Immunol Pract. 2023;11:3161-3168.e2.
- Nordmann TM, Anderton H, Hasegawa A, et al. Spatial proteomics identifies JAKi as treatment for a lethal skin disease. Nature. 2024;635:1001-1009.
- Centers for Disease Control and Prevention. Measles cases and outbreaks. Updated January 7, 2026. Accessed January 12, 2026. https://www.cdc.gov/measles/data-research/
- Rubin R. Despite safe and effective vaccine, measles cases and deaths increased worldwide from 2021 to 2022. JAMA. 2024;331:188-189.
- Orrall A. Dengue cases in the Americas highest recorded. JAMA. 2025;333:452.
- Harris E. As mpox cases surge in Africa, WHO declares a global emergency-here’s what to know. JAMA. 2024;332:862-864.
- Burningham KM, Hinojosa T, Cavazos A, et al. Buffalopox: an emerging cutaneous disease in humans. J Eur Acad Dermatol Venereol. 2025;39:404-406.
- Parker ER. Emergence of Alaskapox infection: what dermatologists need to know. J Am Acad Dermatol. 2024;91:397-399.
- Gostin LO, Singaravelu S, Hynes N. Smallpox readiness: modern strategies against an ancient disease. JAMA. 2024;332:873-874.
- Osborne S, Kam O, Thacker S, et al. Review of category A bioweapons with cutaneous features: epidemiology, clinical presentation, and contemporary management strategies. J Am Acad Dermatol. 2025;93:165-175.
- Caplan AS, Chaturvedi S, Zhu Y, et al. Notes from the field: first reported U.S. cases of tinea caused by Trichophyton indotineae - New York City, December 2021-March 2023. MMWR Morb Mortal Wkly Rep. 2023;72:536-537.
- McKenna M. Why the rise of this drug-resistant fungus is raising international concern. JAMA. 2024;332:859-861.
- Caplan AS, Todd GC, Zhu Y, et al. Clinical course, antifungal susceptibility, and genomic sequencing of Trichophyton indotineae. JAMA Dermatol. 2024;160:701-709.
- Jabet A, Bérot V, Chiarabini T, et al. Trichophyton mentagrophytes ITS genotype VII infections among men who have sex with men in France: an ongoing phenomenon. J Eur Acad Dermatol Venereol. 2025;39:407-415.
- Luchsinger I, Bosshard PP, Kasper RS, et al. Tinea genitalis: a new entity of sexually transmitted infection? Case series and review of the literature. Sex Transm Infect. 2015;91:493-496.
- Khurana A, Sharath S, Sardana K, et al. Therapeutic updates on the management of tinea corporis or cruris in the era of Trichophyton indotineae: separating evidence from hype-a narrative review. Indian J Dermatol. 2023;68:525-540.
- Bérot V, Monsel G, Dauendorffer JN, et al; Groupe Infectiologie Dermatologique et Infections Sexuellement Transmissibles (GrIDIST) de la Société Française de Dermatologie. Klebsiella aerogenes-related facial folliculitis in men having sex with men: a hypothetical new STI?J Eur Acad Dermatol Venereol. 2025;39:E10-E12.
- Edigin E, Kaul S, Eseaton PO, et al. At 180 days hidradenitis suppurativa readmission rate is comparable to heart failure: analysis of the Nationwide Readmissions Database. J Am Acad Dermatol. 2022;87:188-192.
- AbdelHameid D, Wang L, Mauskar MM, et al. Sepsis-like features in hidradenitis suppurativa flares requiring admission: a retrospective cohort study. J Am Acad Dermatol. 2024;90:1291-1294.
- Ehizogie E, Maghari I, Lo S, et al. Hidradenitis suppurativa, systemic inflammatory response syndrome and sepsis: a database study. Br J Dermatol. 2024;191:451-453.
- Maghari I, Abiad H, Griffin T, et al. Hidradenitis suppurativa (HS), systemic inflammatory response syndrome and sepsis, sepsis caused by HS: an empty systematic review. Br J Dermatol. 2024;191:449-450.
- Kjærsgaard Andersen R, Pedersen O, Eidsmo L, et al. Initial steps towards developing a predictive algorithm of disease progression for hidradenitis suppurativa (HS): results from a Cox proportional hazard regression analysis on disease progression among a cohort of 335 Danish patients with HS. Br J Dermatol. 2024;190:904-914.
- Needham M, Pichardo R, Alavi A, et al. Inpatient management of hidradenitis suppurativa: a Delphi consensus study. Cutis. 2024;113:251-254.
- Maskan Bermudez N, Elman SA, Kirsner RS, et al. Management of hidradenitis suppurativa in the inpatient setting: a clinical guide. Arch Dermatol Res. 2025;317:202.
- Nosrati A, Ch’en PY, Torpey ME, et al. Efficacy and durability of intravenous ertapenem therapy for recalcitrant hidradenitis suppurativa. JAMA Dermatol. 2024;160:312-318.
- Tracey EH, Forrestel A, Rosenbach M, et al. Inpatient dermatology consultation in patients with hematologic malignancies. J Am Acad Dermatol. 2016;75:835-836.
- Li DG, Xia FD, Khosravi H, et al. Outcomes of early dermatology consultation for inpatients diagnosed with cellulitis. JAMA Dermatol. 2018;154:537-543.
- Jacoby TV, Shah N, Asdourian MS, et al. Dermatology evaluation for cutaneous immune-related adverse events is associated with improved survival in cancer patients treated with checkpoint inhibition. J Am Acad Dermatol. 2023;88:711-714.
- Hydol-Smith JA, Gallardo MA, Korman A, et al. The United States dermatology inpatient workforce between 2013 and 2019: a Medicare analysis reveals contraction of the workforce and vast access deserts-a cross-sectional analysis. Arch Dermatol Res. 2024;316:103.
- Pineider JL, Rangu SA, Shaw KS, et al. Pediatric consultative dermatology: a survey of the Society for Pediatric Dermatology workforce reveals shortcomings in existing practice models of pediatric dermatology consult services in the United States. Pediatr Dermatol. 2024;41:270-274.
- Puar NK, Canty KM, Newell BD, et al. An evaluation of pediatric dermatology curbside consultations in an academic center: a prospective cohort study. J Am Acad Dermatol. 2024;90:1258-1260.
- Lau CB, Smith GP. Strategies for improving dermatologist comfort and quality of patient care in inpatient settings: a cross-sectional survey study. Arch Dermatol Res. 2024;316:575.
- Hazim AH. Empowering advanced clinical practitioners in managing acute dermatological emergencies. Br J Nurs. 2024;33:448-455.
- Macklis P, Kaffenberger B, Kirven R, et al. Dermatology diagnostic accuracy is improved by artificial intelligence-generated differential diagnoses. Int J Dermatol. 2025;64:960-962.
- Desai AD, Thomas C. Seasonal trends in drug reaction with eosinophilia and systemic symptoms. J Am Acad Dermatol. 2025;92:183-185.
- Wei BM, Fox LP, Kaffenberger BH, et al. Drug-induced hypersensitivity syndrome/drug reaction with eosinophilia and systemic symptoms. Part I. Epidemiology, pathogenesis, clinicopathological features, and prognosis. J Am Acad Dermatol. 2024;90:885-908. doi:10.1016/j.jaad.2023.02.072
- Chan LCE, Sultana R, Choo KJL, et al. Viral reactivation and clinical outcomes in drug reaction with eosinophilia and systemic symptoms (DRESS). Sci Rep. 2024;14:28492.
- Brüggen MC, Walsh S, Ameri MM, et al. Management of adult patients with drug reaction with eosinophilia and systemic symptoms: a Delphi-based international consensus. JAMA Dermatol. 2024;160:37-44
- Hansen E, Gallardo M, Yan A, et al. Risk assessment of drugs associated with DRESS syndrome based on publication frequency: a systematic review. J Am Acad Dermatol. 2024;91:962-966.
- Neubauer ZJK, Chan R, Singal A, et al. SCAR-ed by antibiotics: a retrospective cohort study of severe cutaneous adverse reactions (SCAR) relative risk. J Am Acad Dermatol. 2025;92:1143-1145.
- Ingen-Housz-Oro S, Guichard E, Milpied B, et al. Topical versus oral corticosteroids in moderate drug reaction with eosinophilia and systemic symptoms: a multicenter randomized clinical trial. J Am Acad Dermatol. 2024;91:544-547.
- Hijaz B, Nambudiri VE, Imadojemu S. IL-5 inhibitor treatment in drug reaction with eosinophilia and systemic symptoms. JAMA Dermatol. 2025;161:661-663.
- Bettuzzi T, Lebrun-Vignes B, Ingen-Housz-Oro S, et al. Incidence, in-hospital and long-term mortality, and sequelae of epidermal necrolysis in adults. JAMA Dermatol. 2024;160:1288-1296.
- Hama N, Aoki S, Chen CB, et al. Recent progress in Stevens-Johnson syndrome/toxic epidermal necrolysis: diagnostic criteria, pathogenesis and treatment. Br J Dermatol. 2024;192:9-18.
- Hama N, Sunaga Y, Ochiai H, et al. Development and validation of a novel score to predict mortality in Stevens-Johnson syndrome and toxic epidermal necrolysis: CRISTEN. J Allergy Clin Immunol Pract. 2023;11:3161-3168.e2.
- Nordmann TM, Anderton H, Hasegawa A, et al. Spatial proteomics identifies JAKi as treatment for a lethal skin disease. Nature. 2024;635:1001-1009.
- Centers for Disease Control and Prevention. Measles cases and outbreaks. Updated January 7, 2026. Accessed January 12, 2026. https://www.cdc.gov/measles/data-research/
- Rubin R. Despite safe and effective vaccine, measles cases and deaths increased worldwide from 2021 to 2022. JAMA. 2024;331:188-189.
- Orrall A. Dengue cases in the Americas highest recorded. JAMA. 2025;333:452.
- Harris E. As mpox cases surge in Africa, WHO declares a global emergency-here’s what to know. JAMA. 2024;332:862-864.
- Burningham KM, Hinojosa T, Cavazos A, et al. Buffalopox: an emerging cutaneous disease in humans. J Eur Acad Dermatol Venereol. 2025;39:404-406.
- Parker ER. Emergence of Alaskapox infection: what dermatologists need to know. J Am Acad Dermatol. 2024;91:397-399.
- Gostin LO, Singaravelu S, Hynes N. Smallpox readiness: modern strategies against an ancient disease. JAMA. 2024;332:873-874.
- Osborne S, Kam O, Thacker S, et al. Review of category A bioweapons with cutaneous features: epidemiology, clinical presentation, and contemporary management strategies. J Am Acad Dermatol. 2025;93:165-175.
- Caplan AS, Chaturvedi S, Zhu Y, et al. Notes from the field: first reported U.S. cases of tinea caused by Trichophyton indotineae - New York City, December 2021-March 2023. MMWR Morb Mortal Wkly Rep. 2023;72:536-537.
- McKenna M. Why the rise of this drug-resistant fungus is raising international concern. JAMA. 2024;332:859-861.
- Caplan AS, Todd GC, Zhu Y, et al. Clinical course, antifungal susceptibility, and genomic sequencing of Trichophyton indotineae. JAMA Dermatol. 2024;160:701-709.
- Jabet A, Bérot V, Chiarabini T, et al. Trichophyton mentagrophytes ITS genotype VII infections among men who have sex with men in France: an ongoing phenomenon. J Eur Acad Dermatol Venereol. 2025;39:407-415.
- Luchsinger I, Bosshard PP, Kasper RS, et al. Tinea genitalis: a new entity of sexually transmitted infection? Case series and review of the literature. Sex Transm Infect. 2015;91:493-496.
- Khurana A, Sharath S, Sardana K, et al. Therapeutic updates on the management of tinea corporis or cruris in the era of Trichophyton indotineae: separating evidence from hype-a narrative review. Indian J Dermatol. 2023;68:525-540.
- Bérot V, Monsel G, Dauendorffer JN, et al; Groupe Infectiologie Dermatologique et Infections Sexuellement Transmissibles (GrIDIST) de la Société Française de Dermatologie. Klebsiella aerogenes-related facial folliculitis in men having sex with men: a hypothetical new STI?J Eur Acad Dermatol Venereol. 2025;39:E10-E12.
- Edigin E, Kaul S, Eseaton PO, et al. At 180 days hidradenitis suppurativa readmission rate is comparable to heart failure: analysis of the Nationwide Readmissions Database. J Am Acad Dermatol. 2022;87:188-192.
- AbdelHameid D, Wang L, Mauskar MM, et al. Sepsis-like features in hidradenitis suppurativa flares requiring admission: a retrospective cohort study. J Am Acad Dermatol. 2024;90:1291-1294.
- Ehizogie E, Maghari I, Lo S, et al. Hidradenitis suppurativa, systemic inflammatory response syndrome and sepsis: a database study. Br J Dermatol. 2024;191:451-453.
- Maghari I, Abiad H, Griffin T, et al. Hidradenitis suppurativa (HS), systemic inflammatory response syndrome and sepsis, sepsis caused by HS: an empty systematic review. Br J Dermatol. 2024;191:449-450.
- Kjærsgaard Andersen R, Pedersen O, Eidsmo L, et al. Initial steps towards developing a predictive algorithm of disease progression for hidradenitis suppurativa (HS): results from a Cox proportional hazard regression analysis on disease progression among a cohort of 335 Danish patients with HS. Br J Dermatol. 2024;190:904-914.
- Needham M, Pichardo R, Alavi A, et al. Inpatient management of hidradenitis suppurativa: a Delphi consensus study. Cutis. 2024;113:251-254.
- Maskan Bermudez N, Elman SA, Kirsner RS, et al. Management of hidradenitis suppurativa in the inpatient setting: a clinical guide. Arch Dermatol Res. 2025;317:202.
- Nosrati A, Ch’en PY, Torpey ME, et al. Efficacy and durability of intravenous ertapenem therapy for recalcitrant hidradenitis suppurativa. JAMA Dermatol. 2024;160:312-318.
- Tracey EH, Forrestel A, Rosenbach M, et al. Inpatient dermatology consultation in patients with hematologic malignancies. J Am Acad Dermatol. 2016;75:835-836.
- Li DG, Xia FD, Khosravi H, et al. Outcomes of early dermatology consultation for inpatients diagnosed with cellulitis. JAMA Dermatol. 2018;154:537-543.
- Jacoby TV, Shah N, Asdourian MS, et al. Dermatology evaluation for cutaneous immune-related adverse events is associated with improved survival in cancer patients treated with checkpoint inhibition. J Am Acad Dermatol. 2023;88:711-714.
- Hydol-Smith JA, Gallardo MA, Korman A, et al. The United States dermatology inpatient workforce between 2013 and 2019: a Medicare analysis reveals contraction of the workforce and vast access deserts-a cross-sectional analysis. Arch Dermatol Res. 2024;316:103.
- Pineider JL, Rangu SA, Shaw KS, et al. Pediatric consultative dermatology: a survey of the Society for Pediatric Dermatology workforce reveals shortcomings in existing practice models of pediatric dermatology consult services in the United States. Pediatr Dermatol. 2024;41:270-274.
- Puar NK, Canty KM, Newell BD, et al. An evaluation of pediatric dermatology curbside consultations in an academic center: a prospective cohort study. J Am Acad Dermatol. 2024;90:1258-1260.
- Lau CB, Smith GP. Strategies for improving dermatologist comfort and quality of patient care in inpatient settings: a cross-sectional survey study. Arch Dermatol Res. 2024;316:575.
- Hazim AH. Empowering advanced clinical practitioners in managing acute dermatological emergencies. Br J Nurs. 2024;33:448-455.
- Macklis P, Kaffenberger B, Kirven R, et al. Dermatology diagnostic accuracy is improved by artificial intelligence-generated differential diagnoses. Int J Dermatol. 2025;64:960-962.
Hospital Dermatology: Review of Research in 2024-2025
Hospital Dermatology: Review of Research in 2024-2025
Practice Points
- In suspected drug reaction with eosinophilia and systemic symptoms, discontinue the offending drug; test for human herpesvirus 6, Epstein-Barr virus, and cytomegalovirus when available; and treat moderate cases with low-dose corticosteroids. Reserve interleukin 5 inhibitors for refractory disease.
- For Stevens-Johnson syndrome and toxic epidermal necrolysis (TEN), apply Niigata diagnostic criteria and clinical risk score for TEN, refer patients with 10% or more body surface area detachment to higher-level or burn care, and consider targeted therapies for refractory cases.
- When assessing infectious rashes, consider measles, dengue, mpox, orthopoxviruses, and resistant dermatophytes. Review the patient’s vaccination and travel history, isolate suspected measles cases, and confirm atypical tinea with culture or DNA testing.
- To reduce unnecessary admissions and optimize care for hidradenitis suppurativa, avoid misdiagnosing flares as sepsis, implement multidisciplinary protocols, consider selective intravenous antibiotics, and support expanded inpatient dermatology coverage.
Phototherapy for Pediatric Segmental Vitiligo
Phototherapy for Pediatric Segmental Vitiligo
To the Editor:
Segmental vitiligo (SV) accounts for a minority of vitiligo cases and most frequently occurs in children.1 It characteristically manifests unilaterally and affects a single body area with a sharp midline demarcation. In contrast to nonsegmental vitiligo (NSV), SV typically stabilizes early in the disease progression.1 The pathophysiology of this vitiligo subtype is not well established, but possible autoinflammatory mechanisms associated with somatic mosaicism, neuronal mechanisms, and/or microvascular skin-homing have been proposed.2 We present the case of a pediatric patient with segmental vitiligo of the right hemiface treated with a combination of a topical calcineurin inhibitor and narrow-band UVB (NB-UVB) phototherapy.
An otherwise healthy 7-year-old boy presented to the dermatology department for evaluation of depigmented macules and patches affecting the right hemiface (temporal, periorbital, malar, perioral, preauricular, and mandibular regions) and neck associated with homolateral leukotrichia of the scalp and facial hair as well as the eyelashes of 5 years’ duration. The findings were consistent with SV (Figure 1). The patient previously had been diagnosed based on the clinical findings and treated with continuous application of topical calcineurin inhibitors plus oral cyclosporine (3 mg/kg/d) for 1 year, but the response was poor. The condition had a severe impact on the patient’s quality of life and social relationships. Therapeutic options were discussed with the patient’s caregivers, and ultimately NB-UVB phototherapy was started twice weekly with 10% increases in the dose at each treatment. Topical tacrolimus ointment (1 mg/g) also was started, and the cyclosporine was stopped. Evaluation of treatment progress occurred every 3 months, with progressive repigmentation of the patches following a perifollicular pattern. After 6 months of phototherapy, there was notable repigmentation of the affected areas, particularly in the malar, perioral, and perinasal regions (Figure 2) and the therapeutic response improved after 1 year of treatment (Figure 3). No adverse events were noted during the treatment period.
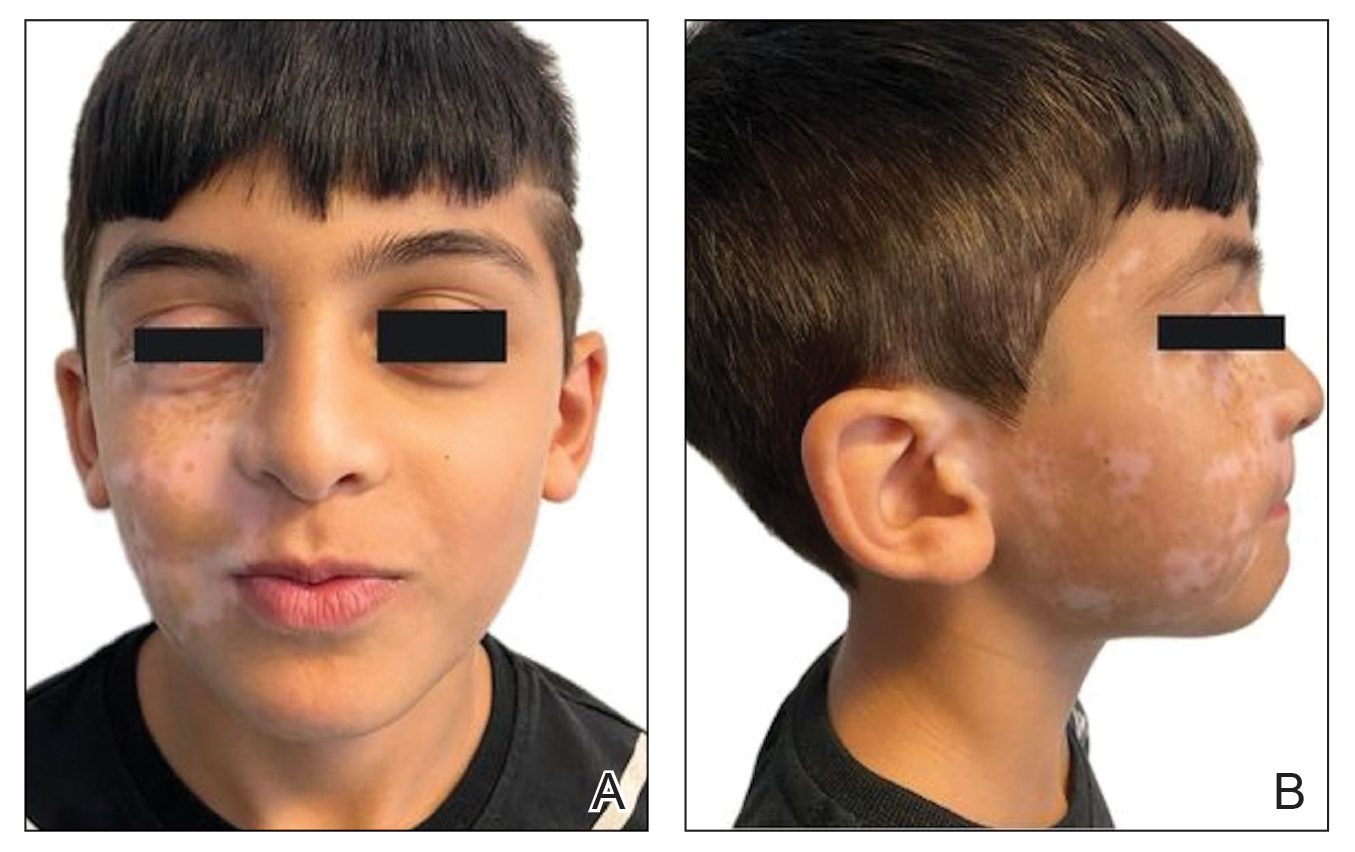
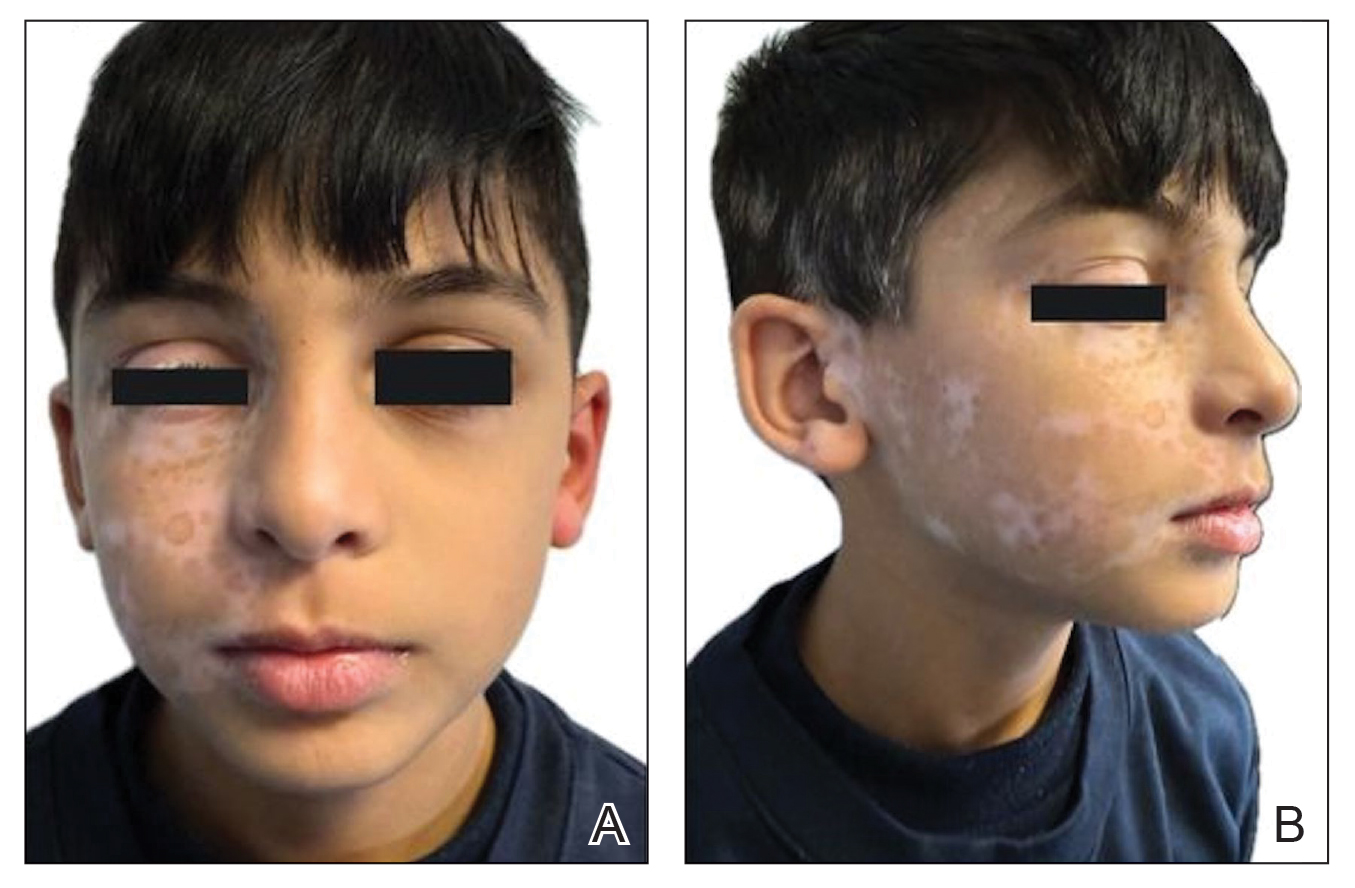
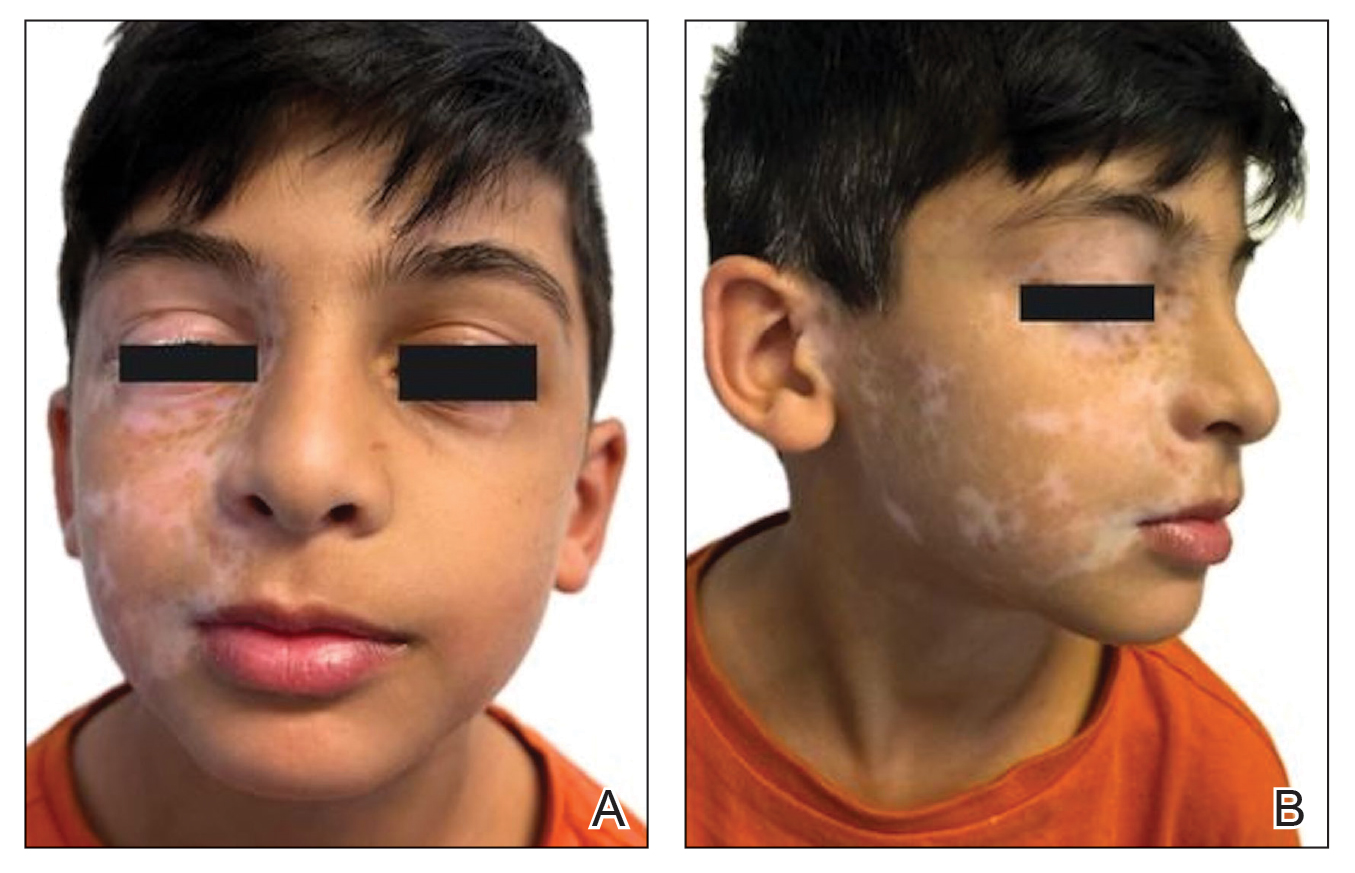
Segmental vitiligo lacks consistently effective treatment options. This subtype of vitiligo is classically resistant to conventional therapeutic options. Surgery may be a more effective and long-lasting treatment option but is not suitable for every patient.1,3 Janus kinase (JAK) inhibitors are the newest treatment options being explored for topical and systemic treatment of vitiligo, with promising results in active and stable NSV lesions4,5; however, SV rarely is represented in case reports and clinical trials. The topical JAK inhibitor ruxolitinib has been approved for use in NSV,5 and a phase 2 trial with oral ritlecitinib only included patients with NSV.4 Furthermore, JAK inhibitors have been studied and approved for children aged 12 years or older as well as for adults,4,5 but younger age groups (4-10 years)—in whom SV most frequently manifests, as in our patient—have been excluded from these studies.1 We present a novel case of SV of the right hemiface in a child that was successfully treated with NB-UVB phototherapy in association with topical calcineurin inhibitors.
The role of phototherapy for the treatment of vitiligo has been well documented, and it frequently is combined with other therapeutic modalities, such as topical anti-inflammatory drugs or, most recently, laser and micrografting techniques.6,7 The most frequently used modality is NB-UVB. In the active phase, it performs an immunomodulatory role, while in the stable phase, it stimulates migration and activity of perilesional and hair follicle melanocytes.8 Initiating therapy early is advisable, particularly during the first 6 months of progression, as there is a higher probability of response1,3,8; nevertheless, a good response was achieved despite the 5-year evolution of vitiligo in our patient. This is a safe option for a skin condition that may begin early in life and require long-term treatment.8 A main concern would be an increased risk for skin cancer associated with repeated NB-UVB exposure, which has not been verified in a recent analysis.9
Segmental vitiligo can considerably impact the patient’s quality of life, affecting social interactions and self-perception, particularly in younger patients with facial involvement; thus, effective and safe therapeutic strategies adapted to the individual and their vitiligo lesions should be discussed. Classical treatment options remain valid and provide good results for some patients; therefore, they should not be disregarded even with the rise of innovative therapies.
- Speeckaert R, Lambert J, Bulat V, et al. Autoimmunity in segmental vitiligo. Front Immunol. 2020;11:568447. doi:10.3389/fimmu.2020.568447
- Lin X, Meng X, Lin J. Segmental vitiligo: autoimmune pathogenesis, neuronal mechanisms, and somatic mosaicism. Int J Dermatol. 2025;64:490-498. doi:10.1111/ijd.17627
- Khalili M, Amiri R, Mohammadi S, et al. Efficacy and safety of traditional and surgical treatment modalities in segmental vitiligo: a review article. J Cosmet Dermatol. 2022;21:2360-2373. doi:10.1111/jocd.14899
- Yamaguchi Y, Peeva E, Del Duca E, et al. Ritlecitinib, a JAK3/TEC family kinase inhibitor, stabilizes active lesions and repigments stable lesions in vitiligo. Arch Dermatol Res. 2024;316:478. doi:10.1007/s00403-024-03182-y
- Rosmarin D, Passeron T, Pandya AG, et al. Two phase 3, randomized, controlled trials of ruxolitinib cream for vitiligo. N Engl J Med. 2022;387:1445-1455. doi:10.1056/NEJMoa2118828
- Chavez-Alvarez S, Herz-Ruelas M, Ocampo-Candiani J, et al. Stable segmental vitiligo treated with punch mini-grafts and narrow band UVB phototherapy. Australas J Dermatol. 2020;61:83-85. doi:10.1111/ajd.13105
- Kim WI, Kim S, Lee SH, et al. The efficacy of fractional carbon dioxide laser combined with narrow-band ultraviolet B phototherapy for non-segmental vitiligo: a systematic review and meta-analysis. Lasers Med Sci. 2021;36:165-173. doi:10.1007/s10103-020-03069-0
- Esmat S, Hegazy RA, Shalaby S, et al. Phototherapy and combination therapies for vitiligo. Dermatol Clin. 2017;35:171-192. doi:10.1016/j.det.2016.11.008
- Mimouni I, Shulman J, Unes AA, et al. Frequency of skin cancer among psoriasis, vitiligo, and mycosis fungoides patients treated with narrowband ultraviolet B phototherapy. Photodermatol Photoimmunol Photomed. 2024;40:E12936. doi:10.1111/phpp.12936
To the Editor:
Segmental vitiligo (SV) accounts for a minority of vitiligo cases and most frequently occurs in children.1 It characteristically manifests unilaterally and affects a single body area with a sharp midline demarcation. In contrast to nonsegmental vitiligo (NSV), SV typically stabilizes early in the disease progression.1 The pathophysiology of this vitiligo subtype is not well established, but possible autoinflammatory mechanisms associated with somatic mosaicism, neuronal mechanisms, and/or microvascular skin-homing have been proposed.2 We present the case of a pediatric patient with segmental vitiligo of the right hemiface treated with a combination of a topical calcineurin inhibitor and narrow-band UVB (NB-UVB) phototherapy.
An otherwise healthy 7-year-old boy presented to the dermatology department for evaluation of depigmented macules and patches affecting the right hemiface (temporal, periorbital, malar, perioral, preauricular, and mandibular regions) and neck associated with homolateral leukotrichia of the scalp and facial hair as well as the eyelashes of 5 years’ duration. The findings were consistent with SV (Figure 1). The patient previously had been diagnosed based on the clinical findings and treated with continuous application of topical calcineurin inhibitors plus oral cyclosporine (3 mg/kg/d) for 1 year, but the response was poor. The condition had a severe impact on the patient’s quality of life and social relationships. Therapeutic options were discussed with the patient’s caregivers, and ultimately NB-UVB phototherapy was started twice weekly with 10% increases in the dose at each treatment. Topical tacrolimus ointment (1 mg/g) also was started, and the cyclosporine was stopped. Evaluation of treatment progress occurred every 3 months, with progressive repigmentation of the patches following a perifollicular pattern. After 6 months of phototherapy, there was notable repigmentation of the affected areas, particularly in the malar, perioral, and perinasal regions (Figure 2) and the therapeutic response improved after 1 year of treatment (Figure 3). No adverse events were noted during the treatment period.



Segmental vitiligo lacks consistently effective treatment options. This subtype of vitiligo is classically resistant to conventional therapeutic options. Surgery may be a more effective and long-lasting treatment option but is not suitable for every patient.1,3 Janus kinase (JAK) inhibitors are the newest treatment options being explored for topical and systemic treatment of vitiligo, with promising results in active and stable NSV lesions4,5; however, SV rarely is represented in case reports and clinical trials. The topical JAK inhibitor ruxolitinib has been approved for use in NSV,5 and a phase 2 trial with oral ritlecitinib only included patients with NSV.4 Furthermore, JAK inhibitors have been studied and approved for children aged 12 years or older as well as for adults,4,5 but younger age groups (4-10 years)—in whom SV most frequently manifests, as in our patient—have been excluded from these studies.1 We present a novel case of SV of the right hemiface in a child that was successfully treated with NB-UVB phototherapy in association with topical calcineurin inhibitors.
The role of phototherapy for the treatment of vitiligo has been well documented, and it frequently is combined with other therapeutic modalities, such as topical anti-inflammatory drugs or, most recently, laser and micrografting techniques.6,7 The most frequently used modality is NB-UVB. In the active phase, it performs an immunomodulatory role, while in the stable phase, it stimulates migration and activity of perilesional and hair follicle melanocytes.8 Initiating therapy early is advisable, particularly during the first 6 months of progression, as there is a higher probability of response1,3,8; nevertheless, a good response was achieved despite the 5-year evolution of vitiligo in our patient. This is a safe option for a skin condition that may begin early in life and require long-term treatment.8 A main concern would be an increased risk for skin cancer associated with repeated NB-UVB exposure, which has not been verified in a recent analysis.9
Segmental vitiligo can considerably impact the patient’s quality of life, affecting social interactions and self-perception, particularly in younger patients with facial involvement; thus, effective and safe therapeutic strategies adapted to the individual and their vitiligo lesions should be discussed. Classical treatment options remain valid and provide good results for some patients; therefore, they should not be disregarded even with the rise of innovative therapies.
To the Editor:
Segmental vitiligo (SV) accounts for a minority of vitiligo cases and most frequently occurs in children.1 It characteristically manifests unilaterally and affects a single body area with a sharp midline demarcation. In contrast to nonsegmental vitiligo (NSV), SV typically stabilizes early in the disease progression.1 The pathophysiology of this vitiligo subtype is not well established, but possible autoinflammatory mechanisms associated with somatic mosaicism, neuronal mechanisms, and/or microvascular skin-homing have been proposed.2 We present the case of a pediatric patient with segmental vitiligo of the right hemiface treated with a combination of a topical calcineurin inhibitor and narrow-band UVB (NB-UVB) phototherapy.
An otherwise healthy 7-year-old boy presented to the dermatology department for evaluation of depigmented macules and patches affecting the right hemiface (temporal, periorbital, malar, perioral, preauricular, and mandibular regions) and neck associated with homolateral leukotrichia of the scalp and facial hair as well as the eyelashes of 5 years’ duration. The findings were consistent with SV (Figure 1). The patient previously had been diagnosed based on the clinical findings and treated with continuous application of topical calcineurin inhibitors plus oral cyclosporine (3 mg/kg/d) for 1 year, but the response was poor. The condition had a severe impact on the patient’s quality of life and social relationships. Therapeutic options were discussed with the patient’s caregivers, and ultimately NB-UVB phototherapy was started twice weekly with 10% increases in the dose at each treatment. Topical tacrolimus ointment (1 mg/g) also was started, and the cyclosporine was stopped. Evaluation of treatment progress occurred every 3 months, with progressive repigmentation of the patches following a perifollicular pattern. After 6 months of phototherapy, there was notable repigmentation of the affected areas, particularly in the malar, perioral, and perinasal regions (Figure 2) and the therapeutic response improved after 1 year of treatment (Figure 3). No adverse events were noted during the treatment period.



Segmental vitiligo lacks consistently effective treatment options. This subtype of vitiligo is classically resistant to conventional therapeutic options. Surgery may be a more effective and long-lasting treatment option but is not suitable for every patient.1,3 Janus kinase (JAK) inhibitors are the newest treatment options being explored for topical and systemic treatment of vitiligo, with promising results in active and stable NSV lesions4,5; however, SV rarely is represented in case reports and clinical trials. The topical JAK inhibitor ruxolitinib has been approved for use in NSV,5 and a phase 2 trial with oral ritlecitinib only included patients with NSV.4 Furthermore, JAK inhibitors have been studied and approved for children aged 12 years or older as well as for adults,4,5 but younger age groups (4-10 years)—in whom SV most frequently manifests, as in our patient—have been excluded from these studies.1 We present a novel case of SV of the right hemiface in a child that was successfully treated with NB-UVB phototherapy in association with topical calcineurin inhibitors.
The role of phototherapy for the treatment of vitiligo has been well documented, and it frequently is combined with other therapeutic modalities, such as topical anti-inflammatory drugs or, most recently, laser and micrografting techniques.6,7 The most frequently used modality is NB-UVB. In the active phase, it performs an immunomodulatory role, while in the stable phase, it stimulates migration and activity of perilesional and hair follicle melanocytes.8 Initiating therapy early is advisable, particularly during the first 6 months of progression, as there is a higher probability of response1,3,8; nevertheless, a good response was achieved despite the 5-year evolution of vitiligo in our patient. This is a safe option for a skin condition that may begin early in life and require long-term treatment.8 A main concern would be an increased risk for skin cancer associated with repeated NB-UVB exposure, which has not been verified in a recent analysis.9
Segmental vitiligo can considerably impact the patient’s quality of life, affecting social interactions and self-perception, particularly in younger patients with facial involvement; thus, effective and safe therapeutic strategies adapted to the individual and their vitiligo lesions should be discussed. Classical treatment options remain valid and provide good results for some patients; therefore, they should not be disregarded even with the rise of innovative therapies.
- Speeckaert R, Lambert J, Bulat V, et al. Autoimmunity in segmental vitiligo. Front Immunol. 2020;11:568447. doi:10.3389/fimmu.2020.568447
- Lin X, Meng X, Lin J. Segmental vitiligo: autoimmune pathogenesis, neuronal mechanisms, and somatic mosaicism. Int J Dermatol. 2025;64:490-498. doi:10.1111/ijd.17627
- Khalili M, Amiri R, Mohammadi S, et al. Efficacy and safety of traditional and surgical treatment modalities in segmental vitiligo: a review article. J Cosmet Dermatol. 2022;21:2360-2373. doi:10.1111/jocd.14899
- Yamaguchi Y, Peeva E, Del Duca E, et al. Ritlecitinib, a JAK3/TEC family kinase inhibitor, stabilizes active lesions and repigments stable lesions in vitiligo. Arch Dermatol Res. 2024;316:478. doi:10.1007/s00403-024-03182-y
- Rosmarin D, Passeron T, Pandya AG, et al. Two phase 3, randomized, controlled trials of ruxolitinib cream for vitiligo. N Engl J Med. 2022;387:1445-1455. doi:10.1056/NEJMoa2118828
- Chavez-Alvarez S, Herz-Ruelas M, Ocampo-Candiani J, et al. Stable segmental vitiligo treated with punch mini-grafts and narrow band UVB phototherapy. Australas J Dermatol. 2020;61:83-85. doi:10.1111/ajd.13105
- Kim WI, Kim S, Lee SH, et al. The efficacy of fractional carbon dioxide laser combined with narrow-band ultraviolet B phototherapy for non-segmental vitiligo: a systematic review and meta-analysis. Lasers Med Sci. 2021;36:165-173. doi:10.1007/s10103-020-03069-0
- Esmat S, Hegazy RA, Shalaby S, et al. Phototherapy and combination therapies for vitiligo. Dermatol Clin. 2017;35:171-192. doi:10.1016/j.det.2016.11.008
- Mimouni I, Shulman J, Unes AA, et al. Frequency of skin cancer among psoriasis, vitiligo, and mycosis fungoides patients treated with narrowband ultraviolet B phototherapy. Photodermatol Photoimmunol Photomed. 2024;40:E12936. doi:10.1111/phpp.12936
- Speeckaert R, Lambert J, Bulat V, et al. Autoimmunity in segmental vitiligo. Front Immunol. 2020;11:568447. doi:10.3389/fimmu.2020.568447
- Lin X, Meng X, Lin J. Segmental vitiligo: autoimmune pathogenesis, neuronal mechanisms, and somatic mosaicism. Int J Dermatol. 2025;64:490-498. doi:10.1111/ijd.17627
- Khalili M, Amiri R, Mohammadi S, et al. Efficacy and safety of traditional and surgical treatment modalities in segmental vitiligo: a review article. J Cosmet Dermatol. 2022;21:2360-2373. doi:10.1111/jocd.14899
- Yamaguchi Y, Peeva E, Del Duca E, et al. Ritlecitinib, a JAK3/TEC family kinase inhibitor, stabilizes active lesions and repigments stable lesions in vitiligo. Arch Dermatol Res. 2024;316:478. doi:10.1007/s00403-024-03182-y
- Rosmarin D, Passeron T, Pandya AG, et al. Two phase 3, randomized, controlled trials of ruxolitinib cream for vitiligo. N Engl J Med. 2022;387:1445-1455. doi:10.1056/NEJMoa2118828
- Chavez-Alvarez S, Herz-Ruelas M, Ocampo-Candiani J, et al. Stable segmental vitiligo treated with punch mini-grafts and narrow band UVB phototherapy. Australas J Dermatol. 2020;61:83-85. doi:10.1111/ajd.13105
- Kim WI, Kim S, Lee SH, et al. The efficacy of fractional carbon dioxide laser combined with narrow-band ultraviolet B phototherapy for non-segmental vitiligo: a systematic review and meta-analysis. Lasers Med Sci. 2021;36:165-173. doi:10.1007/s10103-020-03069-0
- Esmat S, Hegazy RA, Shalaby S, et al. Phototherapy and combination therapies for vitiligo. Dermatol Clin. 2017;35:171-192. doi:10.1016/j.det.2016.11.008
- Mimouni I, Shulman J, Unes AA, et al. Frequency of skin cancer among psoriasis, vitiligo, and mycosis fungoides patients treated with narrowband ultraviolet B phototherapy. Photodermatol Photoimmunol Photomed. 2024;40:E12936. doi:10.1111/phpp.12936
Phototherapy for Pediatric Segmental Vitiligo
Phototherapy for Pediatric Segmental Vitiligo
Practice Points
- Narrow-band UVB in combination with topical tacrolimus may be an effective treatment option for pediatric segmental vitiligo (SV), even in longstanding disease.
- Current evidence for Janus kinase (JAK) inhibitors largely is derived from studies in nonsegmental vitiligo and in patients aged 12 years or older.
- Segmental vitiligo is underrepresented in clinical trials, and topical and systemic JAK inhibitors are not approved for younger children, in whom SV most commonly occurs.
Cancer Data Trends 2026
The annual issue of Cancer Data Trends, produced in collaboration with the Association of VA Hematology/Oncology (AVAHO), highlights the latest research in some of the top cancers impacting US veterans.
In this issue:
- Prostate Cancer: Exposures, Racial Differences, and Treatment Trends
- Expanding Breast Cancer Care for Women Veterans: Genetic Testing and New Therapies
- Multiple Myeloma: Herbicide Exposure, BMI, and Novel Quadruplet Regimens
- Melanoma in Veterans: Higher Risk, Delayed Diagnosis, and Evolving Solutions
- Colorectal Cancer Trends and Digital Interventions in Veterans
- Advancing HCC Management: Updated Guidelines and VHA's Innovative Screening Trial
- Mental Health Care in Veterans With Cancer
The annual issue of Cancer Data Trends, produced in collaboration with the Association of VA Hematology/Oncology (AVAHO), highlights the latest research in some of the top cancers impacting US veterans.
In this issue:
- Prostate Cancer: Exposures, Racial Differences, and Treatment Trends
- Expanding Breast Cancer Care for Women Veterans: Genetic Testing and New Therapies
- Multiple Myeloma: Herbicide Exposure, BMI, and Novel Quadruplet Regimens
- Melanoma in Veterans: Higher Risk, Delayed Diagnosis, and Evolving Solutions
- Colorectal Cancer Trends and Digital Interventions in Veterans
- Advancing HCC Management: Updated Guidelines and VHA's Innovative Screening Trial
- Mental Health Care in Veterans With Cancer
The annual issue of Cancer Data Trends, produced in collaboration with the Association of VA Hematology/Oncology (AVAHO), highlights the latest research in some of the top cancers impacting US veterans.
In this issue:
- Prostate Cancer: Exposures, Racial Differences, and Treatment Trends
- Expanding Breast Cancer Care for Women Veterans: Genetic Testing and New Therapies
- Multiple Myeloma: Herbicide Exposure, BMI, and Novel Quadruplet Regimens
- Melanoma in Veterans: Higher Risk, Delayed Diagnosis, and Evolving Solutions
- Colorectal Cancer Trends and Digital Interventions in Veterans
- Advancing HCC Management: Updated Guidelines and VHA's Innovative Screening Trial
- Mental Health Care in Veterans With Cancer
Black Patches on the Angles of the Mandible
Black Patches on the Angles of the Mandible
THE DIAGNOSIS: Black Dermographism
Black dermographism is characterized by asymptomatic black discoloration on the skin caused by contact with various metals, most commonly gold but also silver, nickel, zinc, lead, and aluminum.1 These metallic particles have a black appearance as they do not reflect light.2 Our patient was wearing gold hoop earrings at presentation, which were near the black patches. Certain topical products (eg, makeup, sunscreens [especially those containing zinc oxide or titanium oxide], toothpaste) can abrade metal, causing it to deposit on the skin and absorb light.3 The black discoloration is not permanent and can be prevented by avoiding contact between inciting products and metals.2 No further diagnostic testing is necessary, and the patches will self-resolve if contact with the product is avoided.
Our patient noted that she wore a physical sunscreen daily, but the black patches were present only when she wore the gold hoop earrings. Given this history and physical examination findings in the office, it was suspected she had black dermographism due to her gold earrings and topical sunscreen. The patient was advised to avoid wearing the gold earrings.
Black dermographism is a misnomer because it is not a true urticarial reaction but rather a false dermographism; therefore, patients will not experience pruritus or erythema.1 True dermographism is an inducible urticarial eruption from pressure or trauma to the skin. The clinical appearance is notable for erythematous wheals in the shape of the external force applied.4 Two other types of false dermographism include white dermographism, which occurs secondary to allergic contact dermatitis, and yellow dermographism, which is caused by bile deposits on the skin.4
Additional diagnoses were able to be ruled out for the following reasons: cutaneous mastocytosis can manifest with red-brown maculopapular lesions often accompanied by the Darier sign, which includes swelling, pruritus, and erythema but was not present in our patient.4 Allergic contact dermatitis manifests as a delayed eczematous reaction around 48 to 72 hours after exposure to an allergen. Our patient’s lesions formed while wearing gold earrings but did not manifest with a hypersensitivity reaction. Of note, symptomatic dermographism has been reported to mimic latex allergy.5 Ecchymosis may appear as erythematous, violaceous, or yellow-green patches depending on the stage but develops due to leakage from broken blood vessels secondary to trauma, which was not reported in our patient. Type I hypersensitivity reactions can occur minutes to hours after exposure to an allergen but typically manifest with a wheal-and-flare presentation.
Black dermographism from gold earrings can mimic concerning skin disorders or poor hygiene, causing unnecessary anxiety. Understanding that it is a harmless reaction between gold and certain topical products can reassure patients and prevent unnecessary testing or treatments.
- Zawar V, Kumavat S, Pawar M. Black dermographism: an uncommon cause of skin discoloration. Indian Dermatol Online J. 2018;9:216-217. doi:10.4103/idoj.IDOJ_228_17
- Lowe E, Lim S. Black dermographism. JAMA Dermatol. 2017; 153:352-353.
- Fisher AA. Black dermographism: mechanism for formation of black color. Cutis. 1993;52(1):17-19.
- Nobles T, Muse ME, Schmieder GJ. Dermatographism. In: StatPearls [Internet]. StatPearls Publishing; February 20, 2023.
- Golberg O, Johnston GA, Wilkinson M. Symptomatic dermographism mimicking latex allergy. Dermatitis. 2014;25:101-103. doi:10.1097 /DER.0000000000000016
THE DIAGNOSIS: Black Dermographism
Black dermographism is characterized by asymptomatic black discoloration on the skin caused by contact with various metals, most commonly gold but also silver, nickel, zinc, lead, and aluminum.1 These metallic particles have a black appearance as they do not reflect light.2 Our patient was wearing gold hoop earrings at presentation, which were near the black patches. Certain topical products (eg, makeup, sunscreens [especially those containing zinc oxide or titanium oxide], toothpaste) can abrade metal, causing it to deposit on the skin and absorb light.3 The black discoloration is not permanent and can be prevented by avoiding contact between inciting products and metals.2 No further diagnostic testing is necessary, and the patches will self-resolve if contact with the product is avoided.
Our patient noted that she wore a physical sunscreen daily, but the black patches were present only when she wore the gold hoop earrings. Given this history and physical examination findings in the office, it was suspected she had black dermographism due to her gold earrings and topical sunscreen. The patient was advised to avoid wearing the gold earrings.
Black dermographism is a misnomer because it is not a true urticarial reaction but rather a false dermographism; therefore, patients will not experience pruritus or erythema.1 True dermographism is an inducible urticarial eruption from pressure or trauma to the skin. The clinical appearance is notable for erythematous wheals in the shape of the external force applied.4 Two other types of false dermographism include white dermographism, which occurs secondary to allergic contact dermatitis, and yellow dermographism, which is caused by bile deposits on the skin.4
Additional diagnoses were able to be ruled out for the following reasons: cutaneous mastocytosis can manifest with red-brown maculopapular lesions often accompanied by the Darier sign, which includes swelling, pruritus, and erythema but was not present in our patient.4 Allergic contact dermatitis manifests as a delayed eczematous reaction around 48 to 72 hours after exposure to an allergen. Our patient’s lesions formed while wearing gold earrings but did not manifest with a hypersensitivity reaction. Of note, symptomatic dermographism has been reported to mimic latex allergy.5 Ecchymosis may appear as erythematous, violaceous, or yellow-green patches depending on the stage but develops due to leakage from broken blood vessels secondary to trauma, which was not reported in our patient. Type I hypersensitivity reactions can occur minutes to hours after exposure to an allergen but typically manifest with a wheal-and-flare presentation.
Black dermographism from gold earrings can mimic concerning skin disorders or poor hygiene, causing unnecessary anxiety. Understanding that it is a harmless reaction between gold and certain topical products can reassure patients and prevent unnecessary testing or treatments.
THE DIAGNOSIS: Black Dermographism
Black dermographism is characterized by asymptomatic black discoloration on the skin caused by contact with various metals, most commonly gold but also silver, nickel, zinc, lead, and aluminum.1 These metallic particles have a black appearance as they do not reflect light.2 Our patient was wearing gold hoop earrings at presentation, which were near the black patches. Certain topical products (eg, makeup, sunscreens [especially those containing zinc oxide or titanium oxide], toothpaste) can abrade metal, causing it to deposit on the skin and absorb light.3 The black discoloration is not permanent and can be prevented by avoiding contact between inciting products and metals.2 No further diagnostic testing is necessary, and the patches will self-resolve if contact with the product is avoided.
Our patient noted that she wore a physical sunscreen daily, but the black patches were present only when she wore the gold hoop earrings. Given this history and physical examination findings in the office, it was suspected she had black dermographism due to her gold earrings and topical sunscreen. The patient was advised to avoid wearing the gold earrings.
Black dermographism is a misnomer because it is not a true urticarial reaction but rather a false dermographism; therefore, patients will not experience pruritus or erythema.1 True dermographism is an inducible urticarial eruption from pressure or trauma to the skin. The clinical appearance is notable for erythematous wheals in the shape of the external force applied.4 Two other types of false dermographism include white dermographism, which occurs secondary to allergic contact dermatitis, and yellow dermographism, which is caused by bile deposits on the skin.4
Additional diagnoses were able to be ruled out for the following reasons: cutaneous mastocytosis can manifest with red-brown maculopapular lesions often accompanied by the Darier sign, which includes swelling, pruritus, and erythema but was not present in our patient.4 Allergic contact dermatitis manifests as a delayed eczematous reaction around 48 to 72 hours after exposure to an allergen. Our patient’s lesions formed while wearing gold earrings but did not manifest with a hypersensitivity reaction. Of note, symptomatic dermographism has been reported to mimic latex allergy.5 Ecchymosis may appear as erythematous, violaceous, or yellow-green patches depending on the stage but develops due to leakage from broken blood vessels secondary to trauma, which was not reported in our patient. Type I hypersensitivity reactions can occur minutes to hours after exposure to an allergen but typically manifest with a wheal-and-flare presentation.
Black dermographism from gold earrings can mimic concerning skin disorders or poor hygiene, causing unnecessary anxiety. Understanding that it is a harmless reaction between gold and certain topical products can reassure patients and prevent unnecessary testing or treatments.
- Zawar V, Kumavat S, Pawar M. Black dermographism: an uncommon cause of skin discoloration. Indian Dermatol Online J. 2018;9:216-217. doi:10.4103/idoj.IDOJ_228_17
- Lowe E, Lim S. Black dermographism. JAMA Dermatol. 2017; 153:352-353.
- Fisher AA. Black dermographism: mechanism for formation of black color. Cutis. 1993;52(1):17-19.
- Nobles T, Muse ME, Schmieder GJ. Dermatographism. In: StatPearls [Internet]. StatPearls Publishing; February 20, 2023.
- Golberg O, Johnston GA, Wilkinson M. Symptomatic dermographism mimicking latex allergy. Dermatitis. 2014;25:101-103. doi:10.1097 /DER.0000000000000016
- Zawar V, Kumavat S, Pawar M. Black dermographism: an uncommon cause of skin discoloration. Indian Dermatol Online J. 2018;9:216-217. doi:10.4103/idoj.IDOJ_228_17
- Lowe E, Lim S. Black dermographism. JAMA Dermatol. 2017; 153:352-353.
- Fisher AA. Black dermographism: mechanism for formation of black color. Cutis. 1993;52(1):17-19.
- Nobles T, Muse ME, Schmieder GJ. Dermatographism. In: StatPearls [Internet]. StatPearls Publishing; February 20, 2023.
- Golberg O, Johnston GA, Wilkinson M. Symptomatic dermographism mimicking latex allergy. Dermatitis. 2014;25:101-103. doi:10.1097 /DER.0000000000000016
Black Patches on the Angles of the Mandible
Black Patches on the Angles of the Mandible
A 30-year-old woman presented for evaluation of intermittent pigmented patches on the face of several months’ duration. The patches would form during the day and disappear when the patient woke up the next morning. She denied any associated pruritus, pain, redness, or recent trauma to the area. Her medical history was otherwise unremarkable. Physical examination revealed ill-defined black patches on both mandibular angles (top). The following day, the patient sent a photograph from home, and the patch was absent (bottom).
Diffusely Scattered Linear Folliculopapular Eruption
Diffusely Scattered Linear Folliculopapular Eruption
THE DIAGNOSIS: Disseminate and Recurrent Infundibulofolliculitis
Histopathology demonstrated a lymphocyte-predominant infundibular infiltrate with mild spongiosis and lymphocytic exocytosis; a mild, superficial perivascular infiltrate also was present. The surrounding skin was largely normal with no notable papillomatosis, acanthosis, or hyperkeratosis (Figure 1). The clinical presentation and histopathologic findings led to the diagnosis of disseminate and recurrent infundibulofolliculitis (DRIF). The patient was started on a 2-week course of once-daily ammonium lactate lotion 12% and urea cream 40% and twice-daily triamcinolone ointment 0.1%. The patient was instructed to take a 1-week break before this regimen was repeated. Isotretinoin 0.5 mg/kg/d for 2 to 4 months was considered and will be an option if there is no improvement at follow-up.
Disseminate and recurrent infundibulofolliculitis is a rare noninfectious folliculitis that initially was described by Hitch and Lund1 in 1968. Males of African descent are most commonly affected by DRIF, but the condition is not limited to this population.2,3 It manifests as asymptomatic, flesh-colored, monomorphic, follicular papules distributed on the trunk and proximal extremities. Pustules can be present, and hair may be seen protruding from them. As the name suggests, DRIF is associated with histopathologic changes that are prominent at the infundibulum of hair follicles.3,4 Disseminate and recurrent infundibulofolliculitis can persist for months to years because it often is resistant to treatment. Treatments include topical monotherapies such as corticosteroids, calcineurin inhibitors, or retinoids; combination topical treatments; antibiotics; and isotretinoin.2 Recurrent remission and exacerbation occurs in many patients.3
The classic manifestations of DRIF, including follicular, monomorphic, flesh-colored papules distributed on the neck, trunk, and proximal upper extremities, were seen in our patient (Figure 2). These findings along with the skin biopsy identifying a lymphocytic infundibular infiltrate led to the diagnosis of DRIF. The papules associated with DRIF can be recurrent or chronic. The lesions in this patient were chronic and persistent.
Despite limited evidence, it has been suggested that DRIF may be a manifestation of atopic dermatitis in patients with darker skin tones. In our case, the patient had a history of childhood eczema. Other hypotheses have proposed that DRIF could be a nonspecific reaction to a currently unknown antigen. A causative infectious agent has not been identified, although the search continues. There is speculation that DRIF could be an overt expression of normal follicular prominence, but the presence of occasional pustules and lymphocyte- predominant infundibular infiltrate negates that.3
Confluent and reticulated papillomatosis was included in the differential for our patient and manifests as asymptomatic hyperpigmented papules and plaques frequently occurring on the upper trunk, neck, and axilla; however, these lesions have a peripheral netlike configuration, as the name suggests. Additionally, this condition is thought to have an infectious component (Dietzia papillomatosis) and responds to antibiotic treatment.5 Follicular eczema also was high in the differential diagnosis but usually is seasonal and pruritic, and histopathology typically shows the features of spongiotic dermatitis. It also would respond well to topical steroids.6 Another condition high on the differential was juxtaclavicular beaded lines, which also manifests as flesh-colored follicular papules distributed on the upper trunk; however, histopathology usually shows features of hyperplastic pilosebaceous units along with spongiosis and exocytosis.7 Pityrosporum folliculitis initially was considered, but the patient only endorsed occasional pruritus. Additionally, no fungal elements were observed.
Currently, there are no definitive treatments for DRIF. The topical treatments available include midpotency corticosteroids, tretinoin, calcineurin inhibitors, 12% lactic acid, and 20% to 40% urea. The systemic therapies are high-dose oral vitamin A (100,000 IU/d), isotretinoin, and psoralen plus UVA.8-10
- Hitch JM, Lund HZ. Disseminate and recurrent infundibulo-folliculitis: report of a case. Arch Dermatol. 1968;97:432-435.
- Ma BC, Sahni VN, Sahni DR, et al. Disseminate and recurrent infundibulofolliculitis: an under-recognized yet treatable entity. J Drugs Dermatol. 2021;20:1353-1354. doi:10.36849/jdd.6173
- Nair SP, Gomathy M, Kumar GN. Disseminate and recurrent infundibulo- folliculitis in an Indian patient: a case report with review of literature. Indian Dermatol Online J. 2017;8:39-41. doi:10.4103/2229- 5178.198775
- Rekha S, Kumar V, Rao P, et al. Disseminate and recurrent infundibulofolliculitis. Indian J Dermatol. 2019;64:404-406. doi:10.4103/ijd.IJD_77_18
- Jones AL, Koerner RJ, Natarajan S, et al. Dietzia papillomatosis sp. nov., a novel actinomycete isolated from the skin of an immunocompetent patient with confluent and reticulated papillomatosis. Int J Syst Evol Microbiol. 2008;58(pt 1):68-72. doi:10.1099/ijs.0.65178-0
- Cohen PR. Follicular contact dermatitis revisited: a review emphasizing neomycin-associated follicular contact dermatitis. World J Clin Cases. 2014;2:815-821. doi:10.12998/wjcc.v2.i12.815
- Butterworth T, Johnson WC. Justa-clavicular beaded lines. Arch Dermatol. 1974;110:891-893.
- Calka O, Metin A, Ozen S. A case of disseminated and recurrent infundibulo-folliculitis responsive to treatment with isotretinoin. J Dermatol. 2002;29:431-434.
- Goihman-Yahr M. Disseminate and recurrent infundibulofolliculitis: response to psoralen plus UVA therapy. Int J Dermatol. 1999;38:75-76.
- Hinds GA, Heald PW. A case of disseminate and recurrent infundibulofolliculitis responsive to treatment with topical steroids. Dermatol Online J. 2008;14:11.
THE DIAGNOSIS: Disseminate and Recurrent Infundibulofolliculitis
Histopathology demonstrated a lymphocyte-predominant infundibular infiltrate with mild spongiosis and lymphocytic exocytosis; a mild, superficial perivascular infiltrate also was present. The surrounding skin was largely normal with no notable papillomatosis, acanthosis, or hyperkeratosis (Figure 1). The clinical presentation and histopathologic findings led to the diagnosis of disseminate and recurrent infundibulofolliculitis (DRIF). The patient was started on a 2-week course of once-daily ammonium lactate lotion 12% and urea cream 40% and twice-daily triamcinolone ointment 0.1%. The patient was instructed to take a 1-week break before this regimen was repeated. Isotretinoin 0.5 mg/kg/d for 2 to 4 months was considered and will be an option if there is no improvement at follow-up.

Disseminate and recurrent infundibulofolliculitis is a rare noninfectious folliculitis that initially was described by Hitch and Lund1 in 1968. Males of African descent are most commonly affected by DRIF, but the condition is not limited to this population.2,3 It manifests as asymptomatic, flesh-colored, monomorphic, follicular papules distributed on the trunk and proximal extremities. Pustules can be present, and hair may be seen protruding from them. As the name suggests, DRIF is associated with histopathologic changes that are prominent at the infundibulum of hair follicles.3,4 Disseminate and recurrent infundibulofolliculitis can persist for months to years because it often is resistant to treatment. Treatments include topical monotherapies such as corticosteroids, calcineurin inhibitors, or retinoids; combination topical treatments; antibiotics; and isotretinoin.2 Recurrent remission and exacerbation occurs in many patients.3
The classic manifestations of DRIF, including follicular, monomorphic, flesh-colored papules distributed on the neck, trunk, and proximal upper extremities, were seen in our patient (Figure 2). These findings along with the skin biopsy identifying a lymphocytic infundibular infiltrate led to the diagnosis of DRIF. The papules associated with DRIF can be recurrent or chronic. The lesions in this patient were chronic and persistent.

Despite limited evidence, it has been suggested that DRIF may be a manifestation of atopic dermatitis in patients with darker skin tones. In our case, the patient had a history of childhood eczema. Other hypotheses have proposed that DRIF could be a nonspecific reaction to a currently unknown antigen. A causative infectious agent has not been identified, although the search continues. There is speculation that DRIF could be an overt expression of normal follicular prominence, but the presence of occasional pustules and lymphocyte- predominant infundibular infiltrate negates that.3
Confluent and reticulated papillomatosis was included in the differential for our patient and manifests as asymptomatic hyperpigmented papules and plaques frequently occurring on the upper trunk, neck, and axilla; however, these lesions have a peripheral netlike configuration, as the name suggests. Additionally, this condition is thought to have an infectious component (Dietzia papillomatosis) and responds to antibiotic treatment.5 Follicular eczema also was high in the differential diagnosis but usually is seasonal and pruritic, and histopathology typically shows the features of spongiotic dermatitis. It also would respond well to topical steroids.6 Another condition high on the differential was juxtaclavicular beaded lines, which also manifests as flesh-colored follicular papules distributed on the upper trunk; however, histopathology usually shows features of hyperplastic pilosebaceous units along with spongiosis and exocytosis.7 Pityrosporum folliculitis initially was considered, but the patient only endorsed occasional pruritus. Additionally, no fungal elements were observed.
Currently, there are no definitive treatments for DRIF. The topical treatments available include midpotency corticosteroids, tretinoin, calcineurin inhibitors, 12% lactic acid, and 20% to 40% urea. The systemic therapies are high-dose oral vitamin A (100,000 IU/d), isotretinoin, and psoralen plus UVA.8-10
THE DIAGNOSIS: Disseminate and Recurrent Infundibulofolliculitis
Histopathology demonstrated a lymphocyte-predominant infundibular infiltrate with mild spongiosis and lymphocytic exocytosis; a mild, superficial perivascular infiltrate also was present. The surrounding skin was largely normal with no notable papillomatosis, acanthosis, or hyperkeratosis (Figure 1). The clinical presentation and histopathologic findings led to the diagnosis of disseminate and recurrent infundibulofolliculitis (DRIF). The patient was started on a 2-week course of once-daily ammonium lactate lotion 12% and urea cream 40% and twice-daily triamcinolone ointment 0.1%. The patient was instructed to take a 1-week break before this regimen was repeated. Isotretinoin 0.5 mg/kg/d for 2 to 4 months was considered and will be an option if there is no improvement at follow-up.

Disseminate and recurrent infundibulofolliculitis is a rare noninfectious folliculitis that initially was described by Hitch and Lund1 in 1968. Males of African descent are most commonly affected by DRIF, but the condition is not limited to this population.2,3 It manifests as asymptomatic, flesh-colored, monomorphic, follicular papules distributed on the trunk and proximal extremities. Pustules can be present, and hair may be seen protruding from them. As the name suggests, DRIF is associated with histopathologic changes that are prominent at the infundibulum of hair follicles.3,4 Disseminate and recurrent infundibulofolliculitis can persist for months to years because it often is resistant to treatment. Treatments include topical monotherapies such as corticosteroids, calcineurin inhibitors, or retinoids; combination topical treatments; antibiotics; and isotretinoin.2 Recurrent remission and exacerbation occurs in many patients.3
The classic manifestations of DRIF, including follicular, monomorphic, flesh-colored papules distributed on the neck, trunk, and proximal upper extremities, were seen in our patient (Figure 2). These findings along with the skin biopsy identifying a lymphocytic infundibular infiltrate led to the diagnosis of DRIF. The papules associated with DRIF can be recurrent or chronic. The lesions in this patient were chronic and persistent.

Despite limited evidence, it has been suggested that DRIF may be a manifestation of atopic dermatitis in patients with darker skin tones. In our case, the patient had a history of childhood eczema. Other hypotheses have proposed that DRIF could be a nonspecific reaction to a currently unknown antigen. A causative infectious agent has not been identified, although the search continues. There is speculation that DRIF could be an overt expression of normal follicular prominence, but the presence of occasional pustules and lymphocyte- predominant infundibular infiltrate negates that.3
Confluent and reticulated papillomatosis was included in the differential for our patient and manifests as asymptomatic hyperpigmented papules and plaques frequently occurring on the upper trunk, neck, and axilla; however, these lesions have a peripheral netlike configuration, as the name suggests. Additionally, this condition is thought to have an infectious component (Dietzia papillomatosis) and responds to antibiotic treatment.5 Follicular eczema also was high in the differential diagnosis but usually is seasonal and pruritic, and histopathology typically shows the features of spongiotic dermatitis. It also would respond well to topical steroids.6 Another condition high on the differential was juxtaclavicular beaded lines, which also manifests as flesh-colored follicular papules distributed on the upper trunk; however, histopathology usually shows features of hyperplastic pilosebaceous units along with spongiosis and exocytosis.7 Pityrosporum folliculitis initially was considered, but the patient only endorsed occasional pruritus. Additionally, no fungal elements were observed.
Currently, there are no definitive treatments for DRIF. The topical treatments available include midpotency corticosteroids, tretinoin, calcineurin inhibitors, 12% lactic acid, and 20% to 40% urea. The systemic therapies are high-dose oral vitamin A (100,000 IU/d), isotretinoin, and psoralen plus UVA.8-10
- Hitch JM, Lund HZ. Disseminate and recurrent infundibulo-folliculitis: report of a case. Arch Dermatol. 1968;97:432-435.
- Ma BC, Sahni VN, Sahni DR, et al. Disseminate and recurrent infundibulofolliculitis: an under-recognized yet treatable entity. J Drugs Dermatol. 2021;20:1353-1354. doi:10.36849/jdd.6173
- Nair SP, Gomathy M, Kumar GN. Disseminate and recurrent infundibulo- folliculitis in an Indian patient: a case report with review of literature. Indian Dermatol Online J. 2017;8:39-41. doi:10.4103/2229- 5178.198775
- Rekha S, Kumar V, Rao P, et al. Disseminate and recurrent infundibulofolliculitis. Indian J Dermatol. 2019;64:404-406. doi:10.4103/ijd.IJD_77_18
- Jones AL, Koerner RJ, Natarajan S, et al. Dietzia papillomatosis sp. nov., a novel actinomycete isolated from the skin of an immunocompetent patient with confluent and reticulated papillomatosis. Int J Syst Evol Microbiol. 2008;58(pt 1):68-72. doi:10.1099/ijs.0.65178-0
- Cohen PR. Follicular contact dermatitis revisited: a review emphasizing neomycin-associated follicular contact dermatitis. World J Clin Cases. 2014;2:815-821. doi:10.12998/wjcc.v2.i12.815
- Butterworth T, Johnson WC. Justa-clavicular beaded lines. Arch Dermatol. 1974;110:891-893.
- Calka O, Metin A, Ozen S. A case of disseminated and recurrent infundibulo-folliculitis responsive to treatment with isotretinoin. J Dermatol. 2002;29:431-434.
- Goihman-Yahr M. Disseminate and recurrent infundibulofolliculitis: response to psoralen plus UVA therapy. Int J Dermatol. 1999;38:75-76.
- Hinds GA, Heald PW. A case of disseminate and recurrent infundibulofolliculitis responsive to treatment with topical steroids. Dermatol Online J. 2008;14:11.
- Hitch JM, Lund HZ. Disseminate and recurrent infundibulo-folliculitis: report of a case. Arch Dermatol. 1968;97:432-435.
- Ma BC, Sahni VN, Sahni DR, et al. Disseminate and recurrent infundibulofolliculitis: an under-recognized yet treatable entity. J Drugs Dermatol. 2021;20:1353-1354. doi:10.36849/jdd.6173
- Nair SP, Gomathy M, Kumar GN. Disseminate and recurrent infundibulo- folliculitis in an Indian patient: a case report with review of literature. Indian Dermatol Online J. 2017;8:39-41. doi:10.4103/2229- 5178.198775
- Rekha S, Kumar V, Rao P, et al. Disseminate and recurrent infundibulofolliculitis. Indian J Dermatol. 2019;64:404-406. doi:10.4103/ijd.IJD_77_18
- Jones AL, Koerner RJ, Natarajan S, et al. Dietzia papillomatosis sp. nov., a novel actinomycete isolated from the skin of an immunocompetent patient with confluent and reticulated papillomatosis. Int J Syst Evol Microbiol. 2008;58(pt 1):68-72. doi:10.1099/ijs.0.65178-0
- Cohen PR. Follicular contact dermatitis revisited: a review emphasizing neomycin-associated follicular contact dermatitis. World J Clin Cases. 2014;2:815-821. doi:10.12998/wjcc.v2.i12.815
- Butterworth T, Johnson WC. Justa-clavicular beaded lines. Arch Dermatol. 1974;110:891-893.
- Calka O, Metin A, Ozen S. A case of disseminated and recurrent infundibulo-folliculitis responsive to treatment with isotretinoin. J Dermatol. 2002;29:431-434.
- Goihman-Yahr M. Disseminate and recurrent infundibulofolliculitis: response to psoralen plus UVA therapy. Int J Dermatol. 1999;38:75-76.
- Hinds GA, Heald PW. A case of disseminate and recurrent infundibulofolliculitis responsive to treatment with topical steroids. Dermatol Online J. 2008;14:11.
Diffusely Scattered Linear Folliculopapular Eruption
Diffusely Scattered Linear Folliculopapular Eruption
A 31-year-old man with a darker skin tone and a history of childhood eczema presented with papules on the trunk and upper arms of several years’ duration. The papules were persistent and were generally asymptomatic but occasionally pruritic. The patient previously had self-treated with over-the counter lotions and topical hydrocortisone with no appreciable changes. On physical examination, a hyperpigmented patch with follicular monomorphic papules was noted across the upper back along with confluent papules and plaques predominantly on the trunk and upper arms. Additionally, the patient had several monomorphic papules in a linear distribution on the neck. Review of systems and examination of the remaining skin were unremarkable. A biopsy from a representative papule on the left upper back was performed.
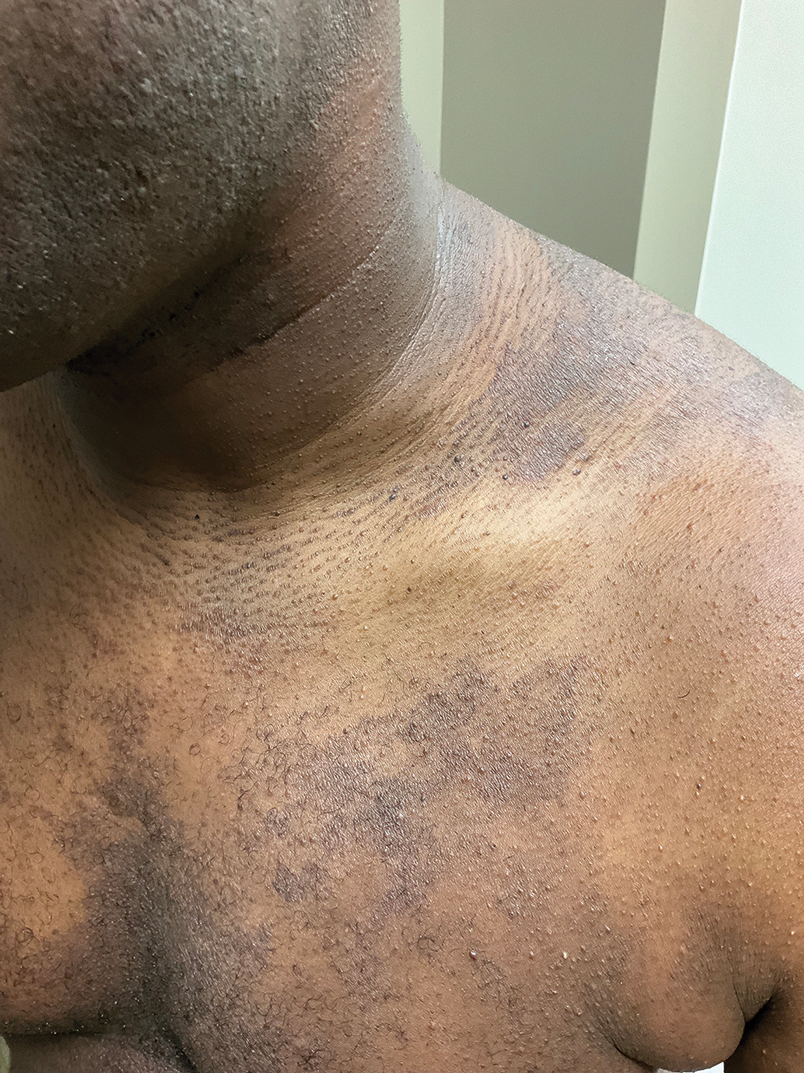
Mental Health Prescribers’ Perceptions of Patients With Substance Use Disorders and Harm Reduction Services
Mental Health Prescribers’ Perceptions of Patients With Substance Use Disorders and Harm Reduction Services
The Public Health and Welfare Act of 1988 prohibited the use of federal funds to “provide individuals with hypodermic needles or syringes so that such individuals may use illegal drugs.”1 Although the Act included the caveat that the US Surgeon General may determine that “a demonstration needle exchange program would be effective in reducing drug abuse,” and thus federal funds could be used, the legislation prohibited federal, state, and local agencies from funding syringe services programs (SSPs). SSPs use various harm reduction tools to improve public safety and reduce the potential harmful consequences of risky behaviors, similar to how using a seat belt while driving reduces the risk of injury or death.2 SSPs are rooted in evidence-based practices, and several studies, according to the Centers for Disease Control and Prevention, have found that people who use drugs (PWUDs) who use community-based SSPs are 5 times more likely to enter treatment than those who do not use these programs. Additionally, these programs have shown an estimated 50% reduction in HIV and hepatitis C infections.3
Amid a 2015 HIV outbreak in Indiana among individuals sharing needles for injection drug use, Congress passed an omnibus spending bill that partially lifted the federal funding restriction. Federal funds now may be used for operational costs that support SSPs but may not be used to purchase syringes themselves.4
Following the 2015 legislation, federal agencies began implementing SSPs. The Veterans Health Administration (VHA) established SSPs at 3 medical centers in 2017.5 Veterans who participated in the programs were able to access supplies (eg, syringes, fentanyl test strips, wound care kits, and condoms) through donations to US Department of Veterans Affairs (VA) medical centers (VAMCs). The success of these programs laid the foundation for the VHA to implement SSPs nationally. VHA SSPs provided access to naloxone (an opioid overdose reversal medication), fentanyl test strips, condoms, sterile syringe distribution, testing for blood-borne viruses, HIV pre-exposure prophylaxis, as well as educational materials and resources, and low-barrier access to drug treatment (eg, medications for opioid use disorder [OUD]).
In 2020, the Biden Administration outlined 7 drug policy priorities, which included enhancing evidence-based harm reduction efforts. 6 This policy also discussed mandates for federal agencies to remove barriers to federal funding for purchasing syringes and other harm reduction supplies. The VHA responded to the policy by publishing guidance that recommended VAMCs develop and/or ensure veterans have access to harm reduction services in the community, where state law is not legally more stringent.7
In 2025 the Trump administration Statement of Drug Policy Priorities encouraged local jurisdictions to increase the availability of drug test strips and naloxone.8 These significant policy shifts moved SSPs from being housed mostly in local public health departments and community-based organizations to also being available at health care facilities. 9 VAMCs have unique opportunities to provide universal health care that includes both prevention services and other medical management to PWUD.
One study assessed staff perceptions of PWUD at a VAMC in preparation for a training program about harm reduction. The results indicated an overall positive staff perception of PWUD, although only the Drug and Drug Problems Perceptions Questionnaire (DDPPQ) was administered, which assessed comfort of working with this population and not explicitly the use of harm reduction.10 Another study interviewed clinical pharmacists, primary care clinicians, social workers, and directors of addiction and mental health services to determine barriers and facilitators (ie, potential opportunities to promote change) to implementing harm reduction at the VHA. The study identified barriers to be a lack of knowledge, time, and comfort, while suggesting opportunities for improvement were engagement of champions, communication and educational strategies, and adaptation of existing infrastructure.11
While these findings are insightful for the VHA to disseminate a harm reduction program, there remains a gap in assessing staff willingness to provide harm reduction services. Evidence on harm reduction services among veterans is limited and more research is needed to better understand the role of these services and acceptance among enrolled veterans and VHA staff. Specifically, more research is needed on health care practitioners’ (HCPs) perceptions of harm reduction use.
Mental health care practitioners frequently treat patients with substance use disorders (SUDs), making them an ideal initial cohort to assess willingness to provide harm reduction to this population. By analyzing mental HCPs’ perceptions, additional interventions could be identified, implemented, and evaluated to improve their willingness to provide harm reduction tools.
This project focused on mental health clinicians with prescribing privileges: physicians (allopathic and osteopathic physicians), nurse practitioners, physician assistants, and clinical pharmacist practitioners. Mental health prescribers were selected because they are uniquely positioned at the intersection of prevention and treatment in drug use. Furthermore, mental health prescribers at the VAMCs included in this study are usually the primary point of entry to SUD clinics. This mixed-methods study used an anonymous online survey and voluntary postsurvey discussions with mental health care prescribers to elaborate on their beliefs and attitudes, providing deeper insight into their responses regarding harm reduction.
Methods
This project was conducted by the Veterans Integrated Services Network (VISN) 5 academic detailing team. VISN 5 serves veterans from economically and demographically diverse areas in Maryland; Washington, DC; West Virginia; and portions of Virginia, Pennsylvania, Ohio, and Kentucky. VAMCs in Baltimore, Maryland, and Washington, DC, serve a largely urban population while the 4 West Virginia facilities in Martinsburg, Huntington, Beckley, and Clarksburg, serve a largely rural population. West Virginia has been the epicenter of the opioid crisis and consistently has the highest drug overdose deaths per capita in the United States.12 Among cities, Baltimore, Maryland, has the highest number of drug overdose deaths per capita with 174.1 per 100,000 people.12,13
At the time of this project, the 6 VISN 5 VAMCs had established overdose education and naloxone distribution (OEND) programs. Although OEND programs have existed since 2013, VISN 5 SSPs and harm reduction services that provided fentanyl test strips were only available at the Martinsburg, Beckley, and Huntington VAMCs. All 6 VAMCs had substance use treatment programs with a variety of inpatient and outpatient mental health services. The Washington, DC and Baltimore VAMCs had opioid treatment programs that provided methadone maintenance.
The VISN 5 academic detailing team consists of 7 clinical pharmacists. These academic detailers plan annual systematic interventions to provide medical knowledge translation services on health-related campaigns. Academic detailers are trained in change management and motivational interviewing. They uniquely facilitate conversations with HCPs on various topics or campaigns, aiming for quality improvement and behavioral change through positive relationships and sharing resources.14 Academic detailing conversations and relationships with HCPs involve assessing and understanding HCP behaviors, including barriers and readiness to change to align with the goal of improving patient outcomes. Academic detailing has improved practice behaviors around providing OEND in VHA.15
To prepare for a harm reduction campaign, the academic detailers sought to gain insight from target VISN 5 mental health prescribers. Figure 1 outlines the project timeline, which started with emails inviting mental health prescribers to complete an anonymous online survey. Academic detailers from each site emailed mental health prescribers who completed the survey to determine interest in expanding on survey findings. Mental health prescribers who completed the survey could participate in a postsurvey discussion.
Surveys
Between January 29, 2024, and February 22, 2024, the academic detailers emailed facility mental health prescribers (N = 156) a link to an anonymous 15-question survey. The email informed recipients of the survey’s purpose: to gain a better understanding of prescriber perceptions of veterans with SUD and harm reduction programs and their willingness to provide harm reduction tools, to better determine interventions that could be implemented.
The survey collected prescriber demographic data and their perceptions of PWUD and harm reduction tools and education. Survey questions were extrapolated from validated surveys (eg, DDPPQ) and survey-based implicit association test.16,17 The survey used multiple choice and 5-point Likert scale questions. Mental health prescribers were asked about their role at the VHA, years in practice, medical center affiliation, type of SUDs treated (eg, opioid, stimulant, alcohol, cannabis, or other), and whether they had previously met with academic detailers about harm reduction.
Respondents read statements about patients with or without SUD and provided Likert scale responses describing their regard, level of comfort, and preferences. The survey included Likert scale questions about respondents’ comfort in providing harm reduction education and supplies. Respondents also noted whether they believed harm reduction reduced substance use, harm reduction tools encourage people with SUD to continue using drugs, and whether HCPs can impact clinical change.
Postsurvey interviews with predetermined questions were conducted in-person or via video conference with ≥ 1 prescriber at each VAMC by an academic detailer. The postsurvey discussion offered an opportunity for respondents to further elaborate and describe previous experiences and current beliefs that may affect their attitudes toward people with SUD and their views on harm reduction. Participants received no compensation for survey completion or interviews.
Analysis
The Washington VAMC Institutional Review Board reviewed and approved this project as quality improvement with potential publication. No inferential statistics were calculated. Survey participant demographics were reported using frequencies and proportions reported for categorical variables. Notes from follow-up interviews were analyzed using the Prosci Awareness, Desire, Knowledge, Ability, and Reinforcement (ADKAR) Model for Change Management.18 This framework is used by academic detailers to determine a prescriber’s stage of change, which helps select the appropriate resources to move the clinician along a change framework. Completed postsurvey interview sheets, including notes written by the academic detailer, were analyzed by the project lead (NJ) who reviewed each interview sheet and analysis with the academic detailer who led the discussion.
Results
Sixty-six respondents completed the online survey (42% response rate), and 7 mental health prescribers participated in a postsurvey discussion. Thirty-one participants (47%) were physicians and 17 (26%) were in practice for > 20 years. Response rates reflected the size of mental health staff at each VAMC at the time of the survey: 17 respondents (26%) worked at each of the Martinsburg and Baltimore VAMCs, with fewer at the other VAMCs (Table 1). Alcohol use disorder was the most commonly reported SUD treated (n = 62; 33%), followed by cannabis use disorder (n = 40; 21%), OUD (n = 38; 20%), and stimulant use disorder (n = 37; 20%).
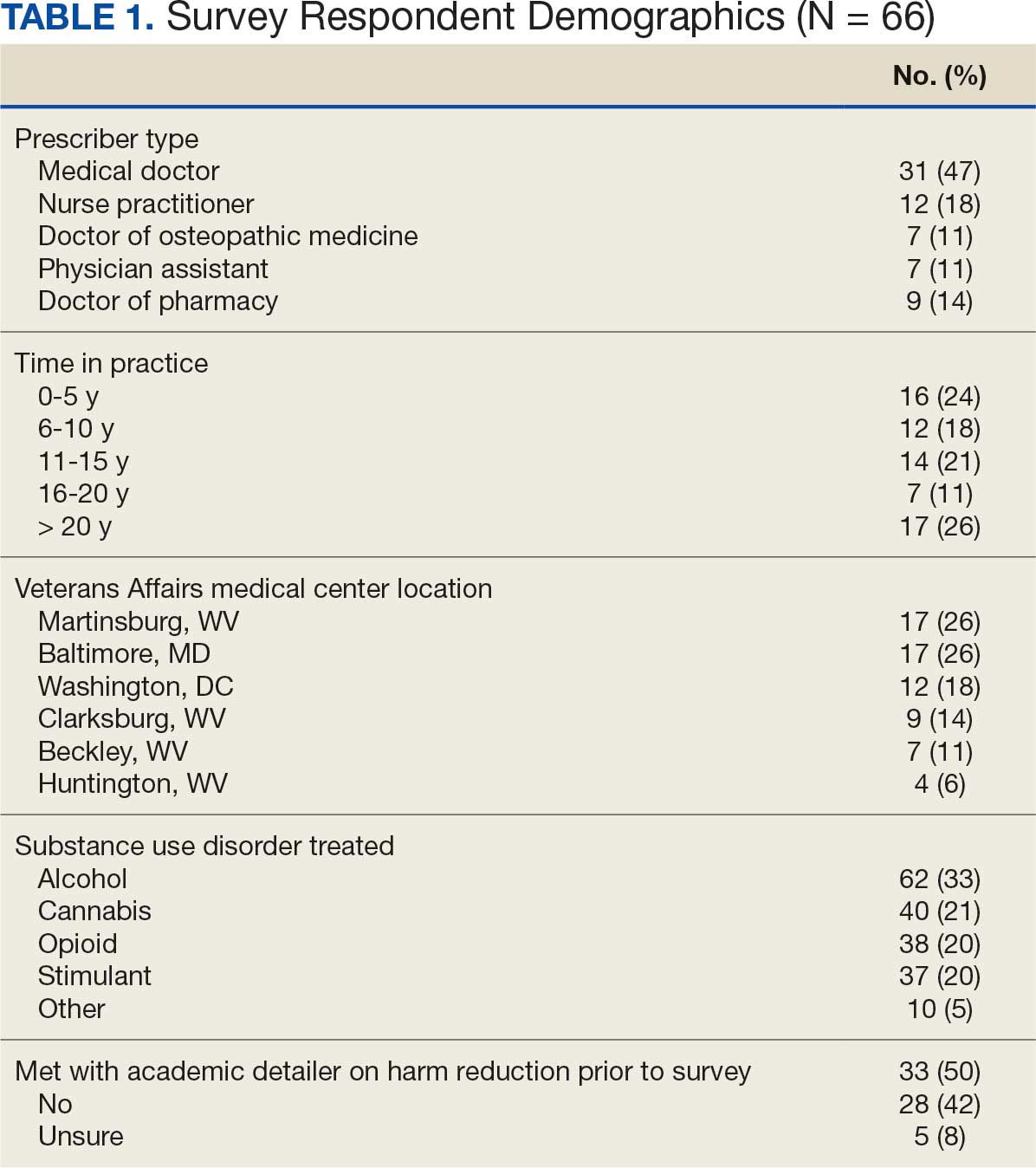
Respondents felt comfortable and confident educating patients on ways to reduce harm related to substance use (91%; mean [SD], 4.24 [0.84]). Most prescribers surveyed (97%; mean [SD], 1.59 [0.68]) disagreed or strongly disagreed that harm reduction encourages patients with SUD to continue using drugs, and all prescribers surveyed disagreed that there is nothing they can do to encourage harm reduction. Survey results were mixed for personal comfort in working with people with SUD vs people without SUD (Figure 2). Respondents were most willing to provide naloxone (95%; mean [SD], 4.71 [0.78]), compared to fentanyl test strips (61%; mean [SD], 3.61 [1.41]) or syringes (39%; mean [SD], 3.18 [1.39]). Respondents were neutral or least willing to provide syringes (Figure 3).
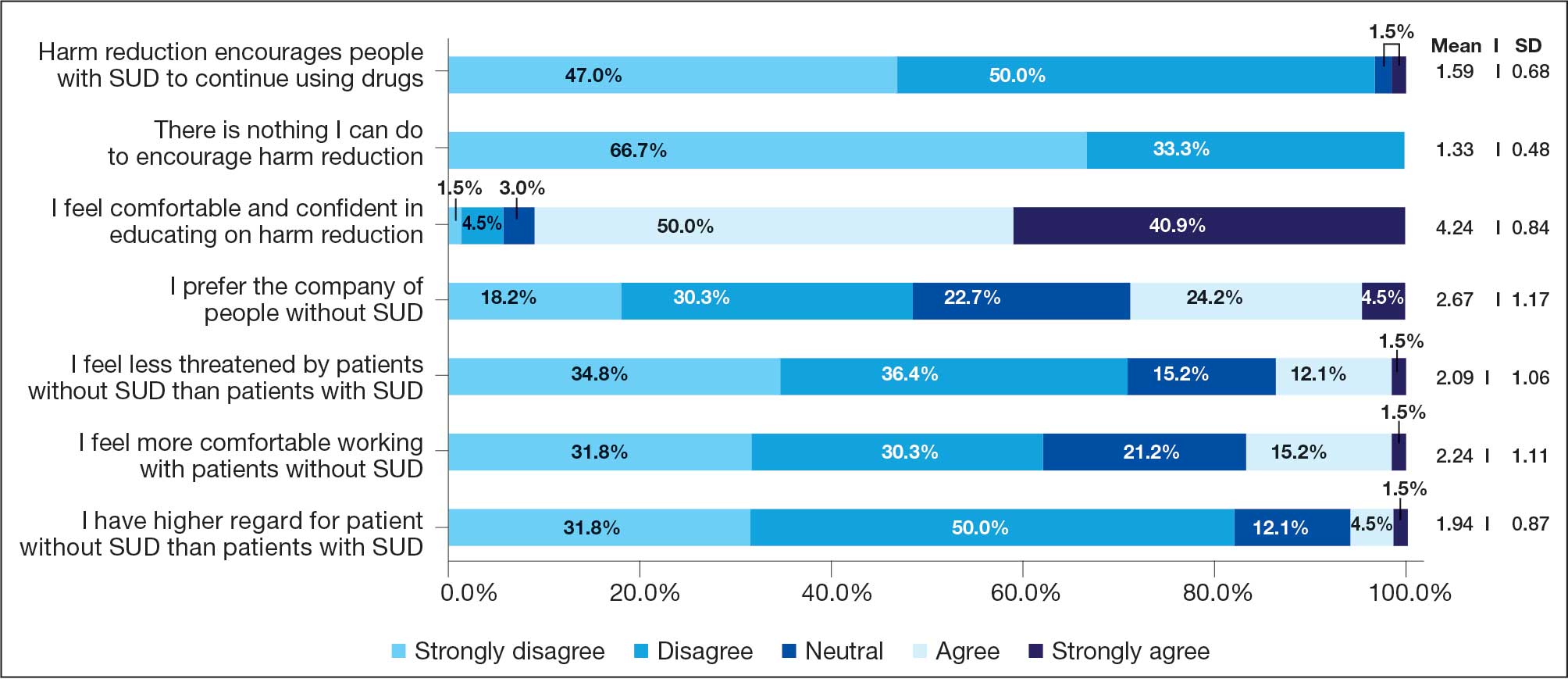
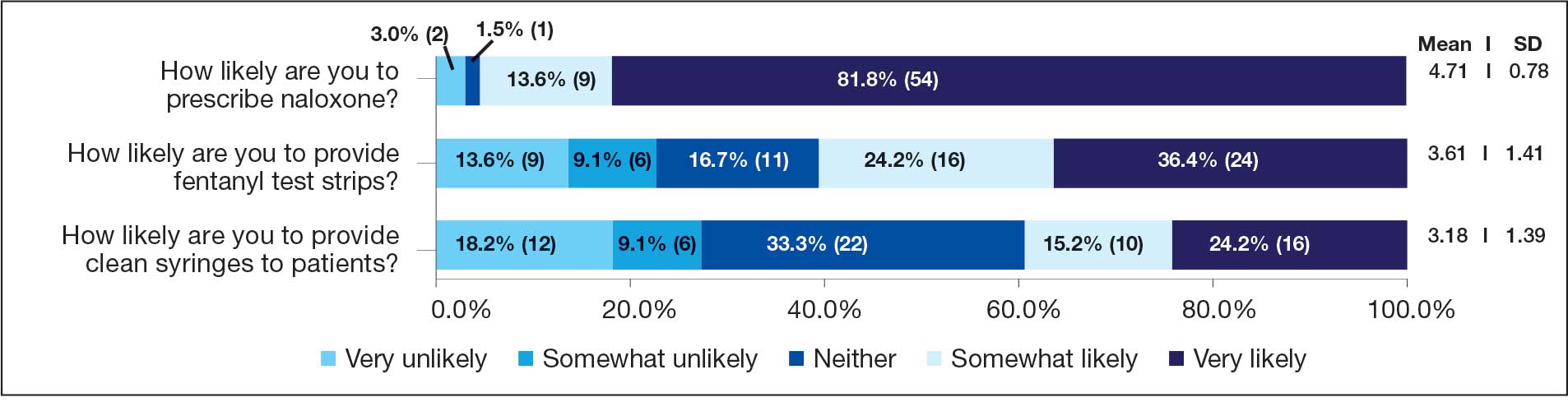
Seven postsurvey interviews were completed between academic detailers and mental health clinicians across the 6 VAMCs. Respondents included 1 physician assistant, 1 nurse practitioner, 1 pharmacist, and 4 physicians. Notes were analyzed using the ADKAR Change Competency Model to organize clinician stages of change (Table 2).
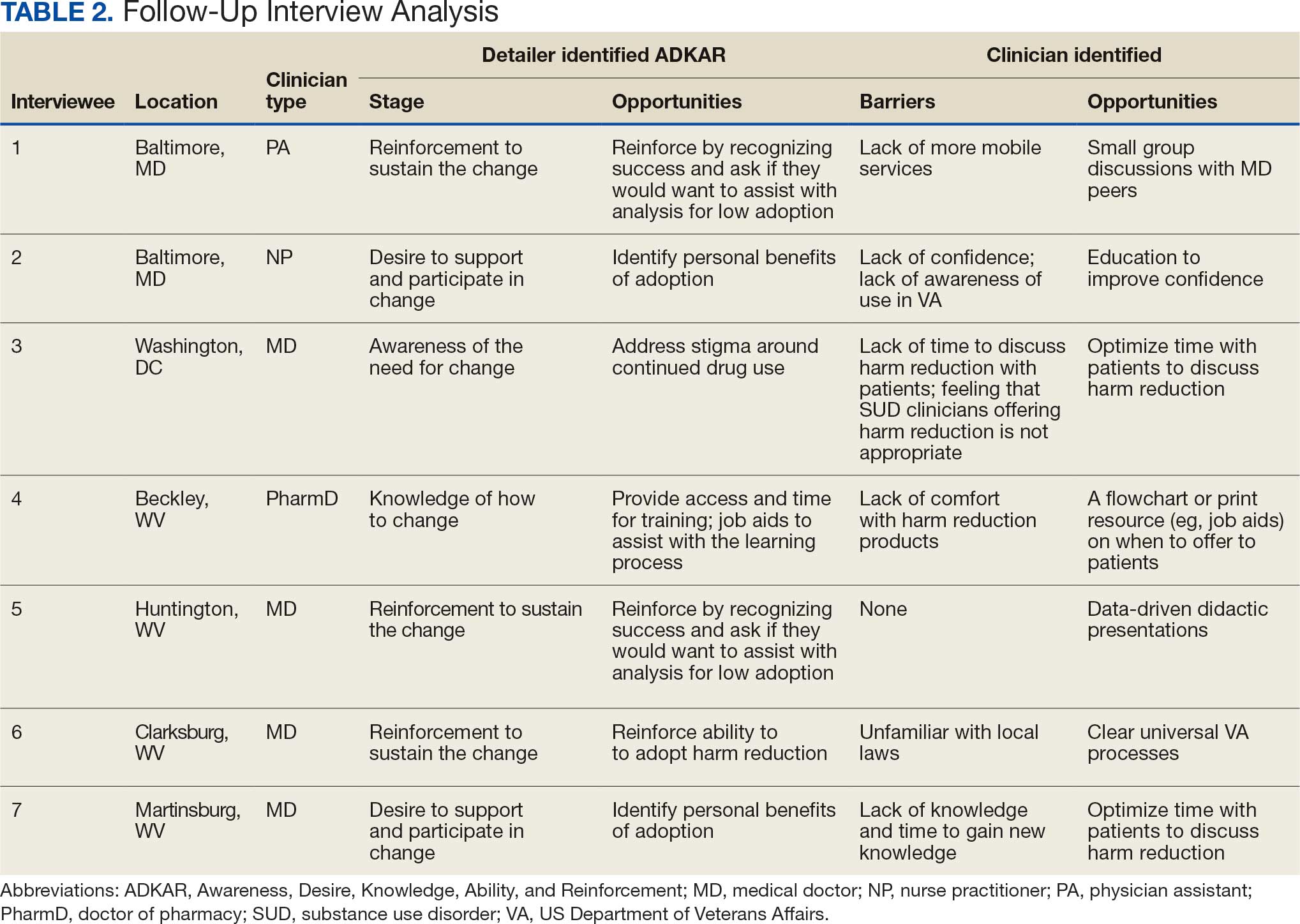
Barriers identified by interviewees included lack of mobile services, lack of confidence and awareness of the availability of harm reduction at their respective medical center, lack of time to discuss harm reduction, negative sentiments toward providing SUD-related harm reduction, discomfort with harm reduction products, and lack of knowledge and time to learn about harm reduction services. Opportunities identified to drive change in practice included additional time allotted during patient appointments, educational discussions and presentations to increase knowledge of and comfort with harm reduction tools, a clear clinical patient care workflow and process for harm reduction services, and reinforcement strategies to recognize success.
Discussion
This project investigated mental health prescribers’ perceptions of harm reduction at VAMCs in West Virginia, Maryland, and Washington, DC. While previous studies have demonstrated the efficacy of harm reduction tools, there is a lack of research on HCPs willingness to use these resources. This study suggests that while most respondents feel confident in and see the value of offering harm reduction resources to patients, a disparity exists between which resources HCPs are more likely to use and factors that would further enhance their ability to integrate harm reduction into practice. The follow-up interviews provided additional insight into the survey results.
Most respondents met the awareness and desire stage and moved to the knowledge, ability, or reinforcement ADKAR stage. It would be reasonable to extrapolate that most of the respondents felt comfortable with and were very likely to offer certain harm reduction tools. In the ADKAR interview analysis, the most common factors needed to drive change included having more time during patient appointments, additional education, clear processes for harm reduction services, and reinforcement strategies to sustain change. Respondents noted that harm reduction discussions took extra time in their already limited appointments with patients, which may have limited time for discussions surrounding all other mental health concerns. These discussions often necessitate in-depth conversations to accurately understand the patients’ needs. Given HCP time constraints, they may view harm reduction as lower in urgency and priority relative to other concerns. While most respondents were in the reinforcement phase, it is important to note the ADKAR model is fluid, and therefore an HCP could move forward or backward. This movement can be noted in the postsurvey interviews where, for example, prescriber 6 was determined to be in the reinforcement stage since they had already discussed harm reduction with patients. However, prescriber 6 also noted a barrier of unfamiliarity with local laws, which could shift them to the ADKAR knowledge stage.
Respondents noted that education through didactic sessions could lead to better incorporation of harm reduction into patient care. While harm reduction has evidence supporting its effectiveness, the respondents noted willingness to discuss harm reduction when treatment fails or the patient refuses treatment or referrals. Respondents expressed mixed opinions on use of harm reduction tools among patients with SUDs as some prescribers viewed harm reduction as part of a treatment plan and others viewed a return to drug use as a failure of treatment. Furthermore, respondents expressed hesitancy surrounding certain harm reduction tools, such as fentanyl test strips or syringes, and perceived these supplies as intended for medical use rather than harm reduction. HCPs may feel uncomfortable offering these supplies for drug use, despite their use for reducing risk.
Most responses were received from VAMCs with large mental health substance use programs. Respondents at larger, urban facilities (Washington, DC, and Baltimore, Maryland) expressed more hesitancy around using harm reduction tools despite having more harm reduction resources available compared to smaller or rural sites. These results align with previous studies that found no difference in prescribers providing medications for OUD in rural and urban VAMCs, showing urban sites, despite more resources, are not more willing to provide harm reduction or other addiction services.19 This evidence might indicate that urban sites may not use available resources (eg, methadone clinics) or that rural sites can provide just as robust medications for OUD care as urban sites.
Follow-up interview analysis indicated that HCPs lack knowledge of certain harm reduction tools. One-on-one peer discussions, like academic detailing, can facilitate discussions around a prescriber’s role in harm reduction, address gaps in knowledge by sharing what is available at the facilities for harm reduction, and suggest conversation points to help prescribers start harm reduction discussions with patients unwilling to begin treatment. Additionally, academic detailing can connect prescribers to available resources in the community to provide pragmatic approaches and suggestions. A clear and consistent treatment process may reduce barriers by reassuring prescribers they have support and by providing consistent directions so that prescribers do not waste time.
Reinforcement is important for sustaining change. VAMCs could consider positive feedback and other evidence-based reinforcement strategies (eg, social recognition, continuing education) to communicate that these changes are noticed and appreciated.20 Late adopters may also be influenced by seeing positive feedback and results for peers. Systematic changes can be the catalyst for and sustain individual change.
Shifting perceptions and adopting change may be challenging, especially for SUD, which can be highly stigmatized. Promotion of successful change should be multifaceted and include both system and individual approaches. VHA systemic changes that could contribute to positive change include provision of time and access to SUD treatment training, a clear and sustainable treatment process, and reinforcement by recognizing success. In addition, facility leadership could provide support through dedicated time and resources during the workday for SUD treatment and harm reduction training. Support could empower HCPs and convey leadership support for harm reduction. This dedicated time could be used for didactic lecture sessions or individual meetings with academic detailers who can tailor discussions to the prescriber’s practice.
Strengths and Limitations
This survey included prescribers from a range of mental health care practice settings (eg, inpatient, outpatient clinic, rural, urban) and varied years of experience. This variety resulted in diverse perspectives and knowledge bases. Postsurvey interviews allowed academic detailers to gain deeper insight into answers in the survey, which can guide future interventions. Postsurvey interviews and application of the ADKAR model provided additional viewpoints on harm reduction.
A limitation of this project is the absence of an assessment of respondents’ harm reduction knowledge accuracy. Although respondents reported confidence in discussing harm reduction with patients, the survey did not assess whether their knowledge was accurate. Additionally, the survey did not ask about the availability of syringes and test strips at the prescribers’ VAMC, which could explain discrepancies in responses between naloxone and other forms of harm reduction (drug test strips and syringes were not available to all HCPs in the VISN). This lack of availability may have skewed responses. West Virginia SSPs, for example, were closed following legislative changes, which may contribute to stigma.21
Not all respondents were asked to do a follow-up interview, which limited the perspectives included in this study. Each site had ≥ 1 follow-up interview to limit the academic detailer’s workload. The initial survey included the phrase clean syringe, which can be stigmatizing and insinuate that PWUD are not clean. The preferred term would have been sterile syringe.22
Conclusions
This survey of mental health prescribers found that most respondents are comfortable treating patients with SUD and confident in educating patients on harm reduction. Additionally, most respondents were more willing to provide naloxone vs fentanyl test strips or sterile syringes. A lack of time and awareness was the most frequently cited barrier to harm reduction services. As the VHA continues to expand access to harm reduction programs, which have proven to increase treatment rates and reduce disease, it will be imperative for HCPs, including mental health prescribers, to recognize the benefit of these programs for veterans with SUD. Future interventions should be designed and evaluated in collaboration with all HCPs and patients. This project determined ways to promote change for prescribers, but it will be important for further research to continue those conversations and incorporate patient perspectives.
- Use of funds to supply hypodermic needles or syringes for illegal drug use; prohibition, 42 USC § 300ee-5 (1988). Accessed January 5, 2026. https://www.law.cornell.edu /uscode/text/42/300ee-5
- OD2A Case Study: Harm Reduction. Centers for Disease Control and Prevention. June 9, 2025. Accessed January 5, 2026. https://www.cdc.gov/overdose-prevention/php /od2a/harm-reduction.html
- Strengthening Syringe Services Programs (SSPs). Centers for Disease Control and Prevention. March 20, 2024. Accessed January 5, 2026. https://www.cdc.gov/hepatitis -syringe-services/php/about/index.html
- Weinmeyer R. Needle exchange programs’ status in US politics. AMA J Ethics. 2016;18:252-257. doi:10.1001/journalofethics.2016.18.3.hlaw1-1603
- Rife-Pennington T, Dinges E, Ho MQ. Implementing syringe services programs within the Veterans Health Administration: facility experiences and next steps. J Am Pharm Assoc (2003). 2023;63:234-240. doi:10.1016/j.japh.2022.10.019
- The Biden-Harris Administration’s Statement of Drug Policy Priorities for Year One. Executive Office of the President, Office of National Drug Control Policy. April 1, 2021. Accessed January 5, 2026. https://bidenwhitehouse.archives.gov/wp-content/uploads/2021/03/BidenHarris -Statement-of-Drug-Policy-Priorities-April-1.pdf
- HIV - for veterans and the public syringe services programs. US Department of Veterans Affairs. Updated August 16, 2021. Accessed January 5, 2026. https://www.hiv .va.gov/patient/ssp.asp
- Trump Administration’s Statement of Drug Policy Priorities. White House. April 1, 2025. Accessed January 7, 2026. https://www.whitehouse.gov/wp-content /uploads/2025/04/2025-Trump-Administration-Drug-Policy -Priorities.pdf
- Health Centers and Syringe Services Programs. National Health Care for the Homeless Council. May 2023. Accessed January 5, 2026. https://nhchc.org/wp-content /uploads/2023/06/Health-Centers-SSPs-Final.pdf
- Lynch RD, Biederman DJ, Silva S, Demasi K. A syringe service program within a federal system: foundations for implementation. J Addict Nurs. 2021;32:152-158. doi:10.1097/JAN.0000000000000402
- Harvey LH, Sliwinski SK, Flike K, et al. The integration of harm reduction services in the Veterans Health Administration (VHA): a qualitative analysis of barriers and facilitators. J Addict Dis. 2024;42:326-334. doi:10.1080/10550887.2023.2210021
- Drug Overdose Death Rates. National Center for Drug Abuse Statistics. Accessed January 5, 2026. https:// drugabusestatistics.org/drug-overdose-deaths
- Ng G. New database shows Baltimore greatly devastated by opioid epidemic. Updated August 21, 2023. Accessed January 5, 2026. https://www.wbaltv.com/article/opioid -epidemic-database-baltimore-deaths/44869671
- Introductory Guide to Academic Detailing. National Resource Center for Academic Detailing. 2017. Accessed January 5, 2026. https://www.narcad.org /uploads/5/7/9/5/57955981/introductory_guide_to_ad.pdf
- Zhang J. Can educational outreach improve experts’ decision making? Evidence from a national opioid academic detailing program. SSRN. 2023;4297398. doi:10.2139/ssrn.4297398
- Watson H, Maclaren W, Kerr S. Staff attitudes towards working with drug users: development of the Drug Problems Perceptions Questionnaire. Addiction. 2007;102:206- 215. doi:10.1111/j.1360-0443.2006.01686.x
- Dahl RA, Vakkalanka JP, Harland KK, Radke J. Investigating healthcare provider bias toward patients who use drugs using a survey-based implicit association test: pilot study. J Addict Med. 2022;16:557-562. doi:10.1097/ADM.0000000000000970
- Hiatt JM, Creasey TJ. Change Management: The People Side of Change. Prosci Learning Center Publications; 2012.
- Wyse JJ, Shull S, Lindner S, et al. Access to medications for opioid use disorder in rural versus urban Veterans Health Administration facilities. J Gen Intern Med. 2023;38:1871-1876. doi:10.1007/s11606-023-08027-4
- Mostofian F, Ruban C, Simunovic N, Bhandari M. Changing physician behavior: what works?. Am J Manag Care. 2015;21(1):75-84.
- Bergdorf-Smith K, Bridge Initiative for S&T Policy, Leadership, and Communications. Syringe Service Programs and HIV Prevention in West Virginia. West Virginia University. February 5, 2024. Accessed January 5, 2026. https:// scitechpolicy.wvu.edu/science-and-technology-notes -articles/2024/02/05/syringe-service-programs-and-hiv -prevention-in-west-virginia
- Brunsdon N. Stop saying ‘clean’. Injecting Advice. February 7, 2011. Accessed January 5, 2026. https:// injectingadvice.com/stop-saying-clean/
The Public Health and Welfare Act of 1988 prohibited the use of federal funds to “provide individuals with hypodermic needles or syringes so that such individuals may use illegal drugs.”1 Although the Act included the caveat that the US Surgeon General may determine that “a demonstration needle exchange program would be effective in reducing drug abuse,” and thus federal funds could be used, the legislation prohibited federal, state, and local agencies from funding syringe services programs (SSPs). SSPs use various harm reduction tools to improve public safety and reduce the potential harmful consequences of risky behaviors, similar to how using a seat belt while driving reduces the risk of injury or death.2 SSPs are rooted in evidence-based practices, and several studies, according to the Centers for Disease Control and Prevention, have found that people who use drugs (PWUDs) who use community-based SSPs are 5 times more likely to enter treatment than those who do not use these programs. Additionally, these programs have shown an estimated 50% reduction in HIV and hepatitis C infections.3
Amid a 2015 HIV outbreak in Indiana among individuals sharing needles for injection drug use, Congress passed an omnibus spending bill that partially lifted the federal funding restriction. Federal funds now may be used for operational costs that support SSPs but may not be used to purchase syringes themselves.4
Following the 2015 legislation, federal agencies began implementing SSPs. The Veterans Health Administration (VHA) established SSPs at 3 medical centers in 2017.5 Veterans who participated in the programs were able to access supplies (eg, syringes, fentanyl test strips, wound care kits, and condoms) through donations to US Department of Veterans Affairs (VA) medical centers (VAMCs). The success of these programs laid the foundation for the VHA to implement SSPs nationally. VHA SSPs provided access to naloxone (an opioid overdose reversal medication), fentanyl test strips, condoms, sterile syringe distribution, testing for blood-borne viruses, HIV pre-exposure prophylaxis, as well as educational materials and resources, and low-barrier access to drug treatment (eg, medications for opioid use disorder [OUD]).
In 2020, the Biden Administration outlined 7 drug policy priorities, which included enhancing evidence-based harm reduction efforts. 6 This policy also discussed mandates for federal agencies to remove barriers to federal funding for purchasing syringes and other harm reduction supplies. The VHA responded to the policy by publishing guidance that recommended VAMCs develop and/or ensure veterans have access to harm reduction services in the community, where state law is not legally more stringent.7
In 2025 the Trump administration Statement of Drug Policy Priorities encouraged local jurisdictions to increase the availability of drug test strips and naloxone.8 These significant policy shifts moved SSPs from being housed mostly in local public health departments and community-based organizations to also being available at health care facilities. 9 VAMCs have unique opportunities to provide universal health care that includes both prevention services and other medical management to PWUD.
One study assessed staff perceptions of PWUD at a VAMC in preparation for a training program about harm reduction. The results indicated an overall positive staff perception of PWUD, although only the Drug and Drug Problems Perceptions Questionnaire (DDPPQ) was administered, which assessed comfort of working with this population and not explicitly the use of harm reduction.10 Another study interviewed clinical pharmacists, primary care clinicians, social workers, and directors of addiction and mental health services to determine barriers and facilitators (ie, potential opportunities to promote change) to implementing harm reduction at the VHA. The study identified barriers to be a lack of knowledge, time, and comfort, while suggesting opportunities for improvement were engagement of champions, communication and educational strategies, and adaptation of existing infrastructure.11
While these findings are insightful for the VHA to disseminate a harm reduction program, there remains a gap in assessing staff willingness to provide harm reduction services. Evidence on harm reduction services among veterans is limited and more research is needed to better understand the role of these services and acceptance among enrolled veterans and VHA staff. Specifically, more research is needed on health care practitioners’ (HCPs) perceptions of harm reduction use.
Mental health care practitioners frequently treat patients with substance use disorders (SUDs), making them an ideal initial cohort to assess willingness to provide harm reduction to this population. By analyzing mental HCPs’ perceptions, additional interventions could be identified, implemented, and evaluated to improve their willingness to provide harm reduction tools.
This project focused on mental health clinicians with prescribing privileges: physicians (allopathic and osteopathic physicians), nurse practitioners, physician assistants, and clinical pharmacist practitioners. Mental health prescribers were selected because they are uniquely positioned at the intersection of prevention and treatment in drug use. Furthermore, mental health prescribers at the VAMCs included in this study are usually the primary point of entry to SUD clinics. This mixed-methods study used an anonymous online survey and voluntary postsurvey discussions with mental health care prescribers to elaborate on their beliefs and attitudes, providing deeper insight into their responses regarding harm reduction.
Methods
This project was conducted by the Veterans Integrated Services Network (VISN) 5 academic detailing team. VISN 5 serves veterans from economically and demographically diverse areas in Maryland; Washington, DC; West Virginia; and portions of Virginia, Pennsylvania, Ohio, and Kentucky. VAMCs in Baltimore, Maryland, and Washington, DC, serve a largely urban population while the 4 West Virginia facilities in Martinsburg, Huntington, Beckley, and Clarksburg, serve a largely rural population. West Virginia has been the epicenter of the opioid crisis and consistently has the highest drug overdose deaths per capita in the United States.12 Among cities, Baltimore, Maryland, has the highest number of drug overdose deaths per capita with 174.1 per 100,000 people.12,13
At the time of this project, the 6 VISN 5 VAMCs had established overdose education and naloxone distribution (OEND) programs. Although OEND programs have existed since 2013, VISN 5 SSPs and harm reduction services that provided fentanyl test strips were only available at the Martinsburg, Beckley, and Huntington VAMCs. All 6 VAMCs had substance use treatment programs with a variety of inpatient and outpatient mental health services. The Washington, DC and Baltimore VAMCs had opioid treatment programs that provided methadone maintenance.
The VISN 5 academic detailing team consists of 7 clinical pharmacists. These academic detailers plan annual systematic interventions to provide medical knowledge translation services on health-related campaigns. Academic detailers are trained in change management and motivational interviewing. They uniquely facilitate conversations with HCPs on various topics or campaigns, aiming for quality improvement and behavioral change through positive relationships and sharing resources.14 Academic detailing conversations and relationships with HCPs involve assessing and understanding HCP behaviors, including barriers and readiness to change to align with the goal of improving patient outcomes. Academic detailing has improved practice behaviors around providing OEND in VHA.15
To prepare for a harm reduction campaign, the academic detailers sought to gain insight from target VISN 5 mental health prescribers. Figure 1 outlines the project timeline, which started with emails inviting mental health prescribers to complete an anonymous online survey. Academic detailers from each site emailed mental health prescribers who completed the survey to determine interest in expanding on survey findings. Mental health prescribers who completed the survey could participate in a postsurvey discussion.

Surveys
Between January 29, 2024, and February 22, 2024, the academic detailers emailed facility mental health prescribers (N = 156) a link to an anonymous 15-question survey. The email informed recipients of the survey’s purpose: to gain a better understanding of prescriber perceptions of veterans with SUD and harm reduction programs and their willingness to provide harm reduction tools, to better determine interventions that could be implemented.
The survey collected prescriber demographic data and their perceptions of PWUD and harm reduction tools and education. Survey questions were extrapolated from validated surveys (eg, DDPPQ) and survey-based implicit association test.16,17 The survey used multiple choice and 5-point Likert scale questions. Mental health prescribers were asked about their role at the VHA, years in practice, medical center affiliation, type of SUDs treated (eg, opioid, stimulant, alcohol, cannabis, or other), and whether they had previously met with academic detailers about harm reduction.
Respondents read statements about patients with or without SUD and provided Likert scale responses describing their regard, level of comfort, and preferences. The survey included Likert scale questions about respondents’ comfort in providing harm reduction education and supplies. Respondents also noted whether they believed harm reduction reduced substance use, harm reduction tools encourage people with SUD to continue using drugs, and whether HCPs can impact clinical change.
Postsurvey interviews with predetermined questions were conducted in-person or via video conference with ≥ 1 prescriber at each VAMC by an academic detailer. The postsurvey discussion offered an opportunity for respondents to further elaborate and describe previous experiences and current beliefs that may affect their attitudes toward people with SUD and their views on harm reduction. Participants received no compensation for survey completion or interviews.
Analysis
The Washington VAMC Institutional Review Board reviewed and approved this project as quality improvement with potential publication. No inferential statistics were calculated. Survey participant demographics were reported using frequencies and proportions reported for categorical variables. Notes from follow-up interviews were analyzed using the Prosci Awareness, Desire, Knowledge, Ability, and Reinforcement (ADKAR) Model for Change Management.18 This framework is used by academic detailers to determine a prescriber’s stage of change, which helps select the appropriate resources to move the clinician along a change framework. Completed postsurvey interview sheets, including notes written by the academic detailer, were analyzed by the project lead (NJ) who reviewed each interview sheet and analysis with the academic detailer who led the discussion.
Results
Sixty-six respondents completed the online survey (42% response rate), and 7 mental health prescribers participated in a postsurvey discussion. Thirty-one participants (47%) were physicians and 17 (26%) were in practice for > 20 years. Response rates reflected the size of mental health staff at each VAMC at the time of the survey: 17 respondents (26%) worked at each of the Martinsburg and Baltimore VAMCs, with fewer at the other VAMCs (Table 1). Alcohol use disorder was the most commonly reported SUD treated (n = 62; 33%), followed by cannabis use disorder (n = 40; 21%), OUD (n = 38; 20%), and stimulant use disorder (n = 37; 20%).

Respondents felt comfortable and confident educating patients on ways to reduce harm related to substance use (91%; mean [SD], 4.24 [0.84]). Most prescribers surveyed (97%; mean [SD], 1.59 [0.68]) disagreed or strongly disagreed that harm reduction encourages patients with SUD to continue using drugs, and all prescribers surveyed disagreed that there is nothing they can do to encourage harm reduction. Survey results were mixed for personal comfort in working with people with SUD vs people without SUD (Figure 2). Respondents were most willing to provide naloxone (95%; mean [SD], 4.71 [0.78]), compared to fentanyl test strips (61%; mean [SD], 3.61 [1.41]) or syringes (39%; mean [SD], 3.18 [1.39]). Respondents were neutral or least willing to provide syringes (Figure 3).


Seven postsurvey interviews were completed between academic detailers and mental health clinicians across the 6 VAMCs. Respondents included 1 physician assistant, 1 nurse practitioner, 1 pharmacist, and 4 physicians. Notes were analyzed using the ADKAR Change Competency Model to organize clinician stages of change (Table 2).

Barriers identified by interviewees included lack of mobile services, lack of confidence and awareness of the availability of harm reduction at their respective medical center, lack of time to discuss harm reduction, negative sentiments toward providing SUD-related harm reduction, discomfort with harm reduction products, and lack of knowledge and time to learn about harm reduction services. Opportunities identified to drive change in practice included additional time allotted during patient appointments, educational discussions and presentations to increase knowledge of and comfort with harm reduction tools, a clear clinical patient care workflow and process for harm reduction services, and reinforcement strategies to recognize success.
Discussion
This project investigated mental health prescribers’ perceptions of harm reduction at VAMCs in West Virginia, Maryland, and Washington, DC. While previous studies have demonstrated the efficacy of harm reduction tools, there is a lack of research on HCPs willingness to use these resources. This study suggests that while most respondents feel confident in and see the value of offering harm reduction resources to patients, a disparity exists between which resources HCPs are more likely to use and factors that would further enhance their ability to integrate harm reduction into practice. The follow-up interviews provided additional insight into the survey results.
Most respondents met the awareness and desire stage and moved to the knowledge, ability, or reinforcement ADKAR stage. It would be reasonable to extrapolate that most of the respondents felt comfortable with and were very likely to offer certain harm reduction tools. In the ADKAR interview analysis, the most common factors needed to drive change included having more time during patient appointments, additional education, clear processes for harm reduction services, and reinforcement strategies to sustain change. Respondents noted that harm reduction discussions took extra time in their already limited appointments with patients, which may have limited time for discussions surrounding all other mental health concerns. These discussions often necessitate in-depth conversations to accurately understand the patients’ needs. Given HCP time constraints, they may view harm reduction as lower in urgency and priority relative to other concerns. While most respondents were in the reinforcement phase, it is important to note the ADKAR model is fluid, and therefore an HCP could move forward or backward. This movement can be noted in the postsurvey interviews where, for example, prescriber 6 was determined to be in the reinforcement stage since they had already discussed harm reduction with patients. However, prescriber 6 also noted a barrier of unfamiliarity with local laws, which could shift them to the ADKAR knowledge stage.
Respondents noted that education through didactic sessions could lead to better incorporation of harm reduction into patient care. While harm reduction has evidence supporting its effectiveness, the respondents noted willingness to discuss harm reduction when treatment fails or the patient refuses treatment or referrals. Respondents expressed mixed opinions on use of harm reduction tools among patients with SUDs as some prescribers viewed harm reduction as part of a treatment plan and others viewed a return to drug use as a failure of treatment. Furthermore, respondents expressed hesitancy surrounding certain harm reduction tools, such as fentanyl test strips or syringes, and perceived these supplies as intended for medical use rather than harm reduction. HCPs may feel uncomfortable offering these supplies for drug use, despite their use for reducing risk.
Most responses were received from VAMCs with large mental health substance use programs. Respondents at larger, urban facilities (Washington, DC, and Baltimore, Maryland) expressed more hesitancy around using harm reduction tools despite having more harm reduction resources available compared to smaller or rural sites. These results align with previous studies that found no difference in prescribers providing medications for OUD in rural and urban VAMCs, showing urban sites, despite more resources, are not more willing to provide harm reduction or other addiction services.19 This evidence might indicate that urban sites may not use available resources (eg, methadone clinics) or that rural sites can provide just as robust medications for OUD care as urban sites.
Follow-up interview analysis indicated that HCPs lack knowledge of certain harm reduction tools. One-on-one peer discussions, like academic detailing, can facilitate discussions around a prescriber’s role in harm reduction, address gaps in knowledge by sharing what is available at the facilities for harm reduction, and suggest conversation points to help prescribers start harm reduction discussions with patients unwilling to begin treatment. Additionally, academic detailing can connect prescribers to available resources in the community to provide pragmatic approaches and suggestions. A clear and consistent treatment process may reduce barriers by reassuring prescribers they have support and by providing consistent directions so that prescribers do not waste time.
Reinforcement is important for sustaining change. VAMCs could consider positive feedback and other evidence-based reinforcement strategies (eg, social recognition, continuing education) to communicate that these changes are noticed and appreciated.20 Late adopters may also be influenced by seeing positive feedback and results for peers. Systematic changes can be the catalyst for and sustain individual change.
Shifting perceptions and adopting change may be challenging, especially for SUD, which can be highly stigmatized. Promotion of successful change should be multifaceted and include both system and individual approaches. VHA systemic changes that could contribute to positive change include provision of time and access to SUD treatment training, a clear and sustainable treatment process, and reinforcement by recognizing success. In addition, facility leadership could provide support through dedicated time and resources during the workday for SUD treatment and harm reduction training. Support could empower HCPs and convey leadership support for harm reduction. This dedicated time could be used for didactic lecture sessions or individual meetings with academic detailers who can tailor discussions to the prescriber’s practice.
Strengths and Limitations
This survey included prescribers from a range of mental health care practice settings (eg, inpatient, outpatient clinic, rural, urban) and varied years of experience. This variety resulted in diverse perspectives and knowledge bases. Postsurvey interviews allowed academic detailers to gain deeper insight into answers in the survey, which can guide future interventions. Postsurvey interviews and application of the ADKAR model provided additional viewpoints on harm reduction.
A limitation of this project is the absence of an assessment of respondents’ harm reduction knowledge accuracy. Although respondents reported confidence in discussing harm reduction with patients, the survey did not assess whether their knowledge was accurate. Additionally, the survey did not ask about the availability of syringes and test strips at the prescribers’ VAMC, which could explain discrepancies in responses between naloxone and other forms of harm reduction (drug test strips and syringes were not available to all HCPs in the VISN). This lack of availability may have skewed responses. West Virginia SSPs, for example, were closed following legislative changes, which may contribute to stigma.21
Not all respondents were asked to do a follow-up interview, which limited the perspectives included in this study. Each site had ≥ 1 follow-up interview to limit the academic detailer’s workload. The initial survey included the phrase clean syringe, which can be stigmatizing and insinuate that PWUD are not clean. The preferred term would have been sterile syringe.22
Conclusions
This survey of mental health prescribers found that most respondents are comfortable treating patients with SUD and confident in educating patients on harm reduction. Additionally, most respondents were more willing to provide naloxone vs fentanyl test strips or sterile syringes. A lack of time and awareness was the most frequently cited barrier to harm reduction services. As the VHA continues to expand access to harm reduction programs, which have proven to increase treatment rates and reduce disease, it will be imperative for HCPs, including mental health prescribers, to recognize the benefit of these programs for veterans with SUD. Future interventions should be designed and evaluated in collaboration with all HCPs and patients. This project determined ways to promote change for prescribers, but it will be important for further research to continue those conversations and incorporate patient perspectives.
The Public Health and Welfare Act of 1988 prohibited the use of federal funds to “provide individuals with hypodermic needles or syringes so that such individuals may use illegal drugs.”1 Although the Act included the caveat that the US Surgeon General may determine that “a demonstration needle exchange program would be effective in reducing drug abuse,” and thus federal funds could be used, the legislation prohibited federal, state, and local agencies from funding syringe services programs (SSPs). SSPs use various harm reduction tools to improve public safety and reduce the potential harmful consequences of risky behaviors, similar to how using a seat belt while driving reduces the risk of injury or death.2 SSPs are rooted in evidence-based practices, and several studies, according to the Centers for Disease Control and Prevention, have found that people who use drugs (PWUDs) who use community-based SSPs are 5 times more likely to enter treatment than those who do not use these programs. Additionally, these programs have shown an estimated 50% reduction in HIV and hepatitis C infections.3
Amid a 2015 HIV outbreak in Indiana among individuals sharing needles for injection drug use, Congress passed an omnibus spending bill that partially lifted the federal funding restriction. Federal funds now may be used for operational costs that support SSPs but may not be used to purchase syringes themselves.4
Following the 2015 legislation, federal agencies began implementing SSPs. The Veterans Health Administration (VHA) established SSPs at 3 medical centers in 2017.5 Veterans who participated in the programs were able to access supplies (eg, syringes, fentanyl test strips, wound care kits, and condoms) through donations to US Department of Veterans Affairs (VA) medical centers (VAMCs). The success of these programs laid the foundation for the VHA to implement SSPs nationally. VHA SSPs provided access to naloxone (an opioid overdose reversal medication), fentanyl test strips, condoms, sterile syringe distribution, testing for blood-borne viruses, HIV pre-exposure prophylaxis, as well as educational materials and resources, and low-barrier access to drug treatment (eg, medications for opioid use disorder [OUD]).
In 2020, the Biden Administration outlined 7 drug policy priorities, which included enhancing evidence-based harm reduction efforts. 6 This policy also discussed mandates for federal agencies to remove barriers to federal funding for purchasing syringes and other harm reduction supplies. The VHA responded to the policy by publishing guidance that recommended VAMCs develop and/or ensure veterans have access to harm reduction services in the community, where state law is not legally more stringent.7
In 2025 the Trump administration Statement of Drug Policy Priorities encouraged local jurisdictions to increase the availability of drug test strips and naloxone.8 These significant policy shifts moved SSPs from being housed mostly in local public health departments and community-based organizations to also being available at health care facilities. 9 VAMCs have unique opportunities to provide universal health care that includes both prevention services and other medical management to PWUD.
One study assessed staff perceptions of PWUD at a VAMC in preparation for a training program about harm reduction. The results indicated an overall positive staff perception of PWUD, although only the Drug and Drug Problems Perceptions Questionnaire (DDPPQ) was administered, which assessed comfort of working with this population and not explicitly the use of harm reduction.10 Another study interviewed clinical pharmacists, primary care clinicians, social workers, and directors of addiction and mental health services to determine barriers and facilitators (ie, potential opportunities to promote change) to implementing harm reduction at the VHA. The study identified barriers to be a lack of knowledge, time, and comfort, while suggesting opportunities for improvement were engagement of champions, communication and educational strategies, and adaptation of existing infrastructure.11
While these findings are insightful for the VHA to disseminate a harm reduction program, there remains a gap in assessing staff willingness to provide harm reduction services. Evidence on harm reduction services among veterans is limited and more research is needed to better understand the role of these services and acceptance among enrolled veterans and VHA staff. Specifically, more research is needed on health care practitioners’ (HCPs) perceptions of harm reduction use.
Mental health care practitioners frequently treat patients with substance use disorders (SUDs), making them an ideal initial cohort to assess willingness to provide harm reduction to this population. By analyzing mental HCPs’ perceptions, additional interventions could be identified, implemented, and evaluated to improve their willingness to provide harm reduction tools.
This project focused on mental health clinicians with prescribing privileges: physicians (allopathic and osteopathic physicians), nurse practitioners, physician assistants, and clinical pharmacist practitioners. Mental health prescribers were selected because they are uniquely positioned at the intersection of prevention and treatment in drug use. Furthermore, mental health prescribers at the VAMCs included in this study are usually the primary point of entry to SUD clinics. This mixed-methods study used an anonymous online survey and voluntary postsurvey discussions with mental health care prescribers to elaborate on their beliefs and attitudes, providing deeper insight into their responses regarding harm reduction.
Methods
This project was conducted by the Veterans Integrated Services Network (VISN) 5 academic detailing team. VISN 5 serves veterans from economically and demographically diverse areas in Maryland; Washington, DC; West Virginia; and portions of Virginia, Pennsylvania, Ohio, and Kentucky. VAMCs in Baltimore, Maryland, and Washington, DC, serve a largely urban population while the 4 West Virginia facilities in Martinsburg, Huntington, Beckley, and Clarksburg, serve a largely rural population. West Virginia has been the epicenter of the opioid crisis and consistently has the highest drug overdose deaths per capita in the United States.12 Among cities, Baltimore, Maryland, has the highest number of drug overdose deaths per capita with 174.1 per 100,000 people.12,13
At the time of this project, the 6 VISN 5 VAMCs had established overdose education and naloxone distribution (OEND) programs. Although OEND programs have existed since 2013, VISN 5 SSPs and harm reduction services that provided fentanyl test strips were only available at the Martinsburg, Beckley, and Huntington VAMCs. All 6 VAMCs had substance use treatment programs with a variety of inpatient and outpatient mental health services. The Washington, DC and Baltimore VAMCs had opioid treatment programs that provided methadone maintenance.
The VISN 5 academic detailing team consists of 7 clinical pharmacists. These academic detailers plan annual systematic interventions to provide medical knowledge translation services on health-related campaigns. Academic detailers are trained in change management and motivational interviewing. They uniquely facilitate conversations with HCPs on various topics or campaigns, aiming for quality improvement and behavioral change through positive relationships and sharing resources.14 Academic detailing conversations and relationships with HCPs involve assessing and understanding HCP behaviors, including barriers and readiness to change to align with the goal of improving patient outcomes. Academic detailing has improved practice behaviors around providing OEND in VHA.15
To prepare for a harm reduction campaign, the academic detailers sought to gain insight from target VISN 5 mental health prescribers. Figure 1 outlines the project timeline, which started with emails inviting mental health prescribers to complete an anonymous online survey. Academic detailers from each site emailed mental health prescribers who completed the survey to determine interest in expanding on survey findings. Mental health prescribers who completed the survey could participate in a postsurvey discussion.

Surveys
Between January 29, 2024, and February 22, 2024, the academic detailers emailed facility mental health prescribers (N = 156) a link to an anonymous 15-question survey. The email informed recipients of the survey’s purpose: to gain a better understanding of prescriber perceptions of veterans with SUD and harm reduction programs and their willingness to provide harm reduction tools, to better determine interventions that could be implemented.
The survey collected prescriber demographic data and their perceptions of PWUD and harm reduction tools and education. Survey questions were extrapolated from validated surveys (eg, DDPPQ) and survey-based implicit association test.16,17 The survey used multiple choice and 5-point Likert scale questions. Mental health prescribers were asked about their role at the VHA, years in practice, medical center affiliation, type of SUDs treated (eg, opioid, stimulant, alcohol, cannabis, or other), and whether they had previously met with academic detailers about harm reduction.
Respondents read statements about patients with or without SUD and provided Likert scale responses describing their regard, level of comfort, and preferences. The survey included Likert scale questions about respondents’ comfort in providing harm reduction education and supplies. Respondents also noted whether they believed harm reduction reduced substance use, harm reduction tools encourage people with SUD to continue using drugs, and whether HCPs can impact clinical change.
Postsurvey interviews with predetermined questions were conducted in-person or via video conference with ≥ 1 prescriber at each VAMC by an academic detailer. The postsurvey discussion offered an opportunity for respondents to further elaborate and describe previous experiences and current beliefs that may affect their attitudes toward people with SUD and their views on harm reduction. Participants received no compensation for survey completion or interviews.
Analysis
The Washington VAMC Institutional Review Board reviewed and approved this project as quality improvement with potential publication. No inferential statistics were calculated. Survey participant demographics were reported using frequencies and proportions reported for categorical variables. Notes from follow-up interviews were analyzed using the Prosci Awareness, Desire, Knowledge, Ability, and Reinforcement (ADKAR) Model for Change Management.18 This framework is used by academic detailers to determine a prescriber’s stage of change, which helps select the appropriate resources to move the clinician along a change framework. Completed postsurvey interview sheets, including notes written by the academic detailer, were analyzed by the project lead (NJ) who reviewed each interview sheet and analysis with the academic detailer who led the discussion.
Results
Sixty-six respondents completed the online survey (42% response rate), and 7 mental health prescribers participated in a postsurvey discussion. Thirty-one participants (47%) were physicians and 17 (26%) were in practice for > 20 years. Response rates reflected the size of mental health staff at each VAMC at the time of the survey: 17 respondents (26%) worked at each of the Martinsburg and Baltimore VAMCs, with fewer at the other VAMCs (Table 1). Alcohol use disorder was the most commonly reported SUD treated (n = 62; 33%), followed by cannabis use disorder (n = 40; 21%), OUD (n = 38; 20%), and stimulant use disorder (n = 37; 20%).

Respondents felt comfortable and confident educating patients on ways to reduce harm related to substance use (91%; mean [SD], 4.24 [0.84]). Most prescribers surveyed (97%; mean [SD], 1.59 [0.68]) disagreed or strongly disagreed that harm reduction encourages patients with SUD to continue using drugs, and all prescribers surveyed disagreed that there is nothing they can do to encourage harm reduction. Survey results were mixed for personal comfort in working with people with SUD vs people without SUD (Figure 2). Respondents were most willing to provide naloxone (95%; mean [SD], 4.71 [0.78]), compared to fentanyl test strips (61%; mean [SD], 3.61 [1.41]) or syringes (39%; mean [SD], 3.18 [1.39]). Respondents were neutral or least willing to provide syringes (Figure 3).


Seven postsurvey interviews were completed between academic detailers and mental health clinicians across the 6 VAMCs. Respondents included 1 physician assistant, 1 nurse practitioner, 1 pharmacist, and 4 physicians. Notes were analyzed using the ADKAR Change Competency Model to organize clinician stages of change (Table 2).

Barriers identified by interviewees included lack of mobile services, lack of confidence and awareness of the availability of harm reduction at their respective medical center, lack of time to discuss harm reduction, negative sentiments toward providing SUD-related harm reduction, discomfort with harm reduction products, and lack of knowledge and time to learn about harm reduction services. Opportunities identified to drive change in practice included additional time allotted during patient appointments, educational discussions and presentations to increase knowledge of and comfort with harm reduction tools, a clear clinical patient care workflow and process for harm reduction services, and reinforcement strategies to recognize success.
Discussion
This project investigated mental health prescribers’ perceptions of harm reduction at VAMCs in West Virginia, Maryland, and Washington, DC. While previous studies have demonstrated the efficacy of harm reduction tools, there is a lack of research on HCPs willingness to use these resources. This study suggests that while most respondents feel confident in and see the value of offering harm reduction resources to patients, a disparity exists between which resources HCPs are more likely to use and factors that would further enhance their ability to integrate harm reduction into practice. The follow-up interviews provided additional insight into the survey results.
Most respondents met the awareness and desire stage and moved to the knowledge, ability, or reinforcement ADKAR stage. It would be reasonable to extrapolate that most of the respondents felt comfortable with and were very likely to offer certain harm reduction tools. In the ADKAR interview analysis, the most common factors needed to drive change included having more time during patient appointments, additional education, clear processes for harm reduction services, and reinforcement strategies to sustain change. Respondents noted that harm reduction discussions took extra time in their already limited appointments with patients, which may have limited time for discussions surrounding all other mental health concerns. These discussions often necessitate in-depth conversations to accurately understand the patients’ needs. Given HCP time constraints, they may view harm reduction as lower in urgency and priority relative to other concerns. While most respondents were in the reinforcement phase, it is important to note the ADKAR model is fluid, and therefore an HCP could move forward or backward. This movement can be noted in the postsurvey interviews where, for example, prescriber 6 was determined to be in the reinforcement stage since they had already discussed harm reduction with patients. However, prescriber 6 also noted a barrier of unfamiliarity with local laws, which could shift them to the ADKAR knowledge stage.
Respondents noted that education through didactic sessions could lead to better incorporation of harm reduction into patient care. While harm reduction has evidence supporting its effectiveness, the respondents noted willingness to discuss harm reduction when treatment fails or the patient refuses treatment or referrals. Respondents expressed mixed opinions on use of harm reduction tools among patients with SUDs as some prescribers viewed harm reduction as part of a treatment plan and others viewed a return to drug use as a failure of treatment. Furthermore, respondents expressed hesitancy surrounding certain harm reduction tools, such as fentanyl test strips or syringes, and perceived these supplies as intended for medical use rather than harm reduction. HCPs may feel uncomfortable offering these supplies for drug use, despite their use for reducing risk.
Most responses were received from VAMCs with large mental health substance use programs. Respondents at larger, urban facilities (Washington, DC, and Baltimore, Maryland) expressed more hesitancy around using harm reduction tools despite having more harm reduction resources available compared to smaller or rural sites. These results align with previous studies that found no difference in prescribers providing medications for OUD in rural and urban VAMCs, showing urban sites, despite more resources, are not more willing to provide harm reduction or other addiction services.19 This evidence might indicate that urban sites may not use available resources (eg, methadone clinics) or that rural sites can provide just as robust medications for OUD care as urban sites.
Follow-up interview analysis indicated that HCPs lack knowledge of certain harm reduction tools. One-on-one peer discussions, like academic detailing, can facilitate discussions around a prescriber’s role in harm reduction, address gaps in knowledge by sharing what is available at the facilities for harm reduction, and suggest conversation points to help prescribers start harm reduction discussions with patients unwilling to begin treatment. Additionally, academic detailing can connect prescribers to available resources in the community to provide pragmatic approaches and suggestions. A clear and consistent treatment process may reduce barriers by reassuring prescribers they have support and by providing consistent directions so that prescribers do not waste time.
Reinforcement is important for sustaining change. VAMCs could consider positive feedback and other evidence-based reinforcement strategies (eg, social recognition, continuing education) to communicate that these changes are noticed and appreciated.20 Late adopters may also be influenced by seeing positive feedback and results for peers. Systematic changes can be the catalyst for and sustain individual change.
Shifting perceptions and adopting change may be challenging, especially for SUD, which can be highly stigmatized. Promotion of successful change should be multifaceted and include both system and individual approaches. VHA systemic changes that could contribute to positive change include provision of time and access to SUD treatment training, a clear and sustainable treatment process, and reinforcement by recognizing success. In addition, facility leadership could provide support through dedicated time and resources during the workday for SUD treatment and harm reduction training. Support could empower HCPs and convey leadership support for harm reduction. This dedicated time could be used for didactic lecture sessions or individual meetings with academic detailers who can tailor discussions to the prescriber’s practice.
Strengths and Limitations
This survey included prescribers from a range of mental health care practice settings (eg, inpatient, outpatient clinic, rural, urban) and varied years of experience. This variety resulted in diverse perspectives and knowledge bases. Postsurvey interviews allowed academic detailers to gain deeper insight into answers in the survey, which can guide future interventions. Postsurvey interviews and application of the ADKAR model provided additional viewpoints on harm reduction.
A limitation of this project is the absence of an assessment of respondents’ harm reduction knowledge accuracy. Although respondents reported confidence in discussing harm reduction with patients, the survey did not assess whether their knowledge was accurate. Additionally, the survey did not ask about the availability of syringes and test strips at the prescribers’ VAMC, which could explain discrepancies in responses between naloxone and other forms of harm reduction (drug test strips and syringes were not available to all HCPs in the VISN). This lack of availability may have skewed responses. West Virginia SSPs, for example, were closed following legislative changes, which may contribute to stigma.21
Not all respondents were asked to do a follow-up interview, which limited the perspectives included in this study. Each site had ≥ 1 follow-up interview to limit the academic detailer’s workload. The initial survey included the phrase clean syringe, which can be stigmatizing and insinuate that PWUD are not clean. The preferred term would have been sterile syringe.22
Conclusions
This survey of mental health prescribers found that most respondents are comfortable treating patients with SUD and confident in educating patients on harm reduction. Additionally, most respondents were more willing to provide naloxone vs fentanyl test strips or sterile syringes. A lack of time and awareness was the most frequently cited barrier to harm reduction services. As the VHA continues to expand access to harm reduction programs, which have proven to increase treatment rates and reduce disease, it will be imperative for HCPs, including mental health prescribers, to recognize the benefit of these programs for veterans with SUD. Future interventions should be designed and evaluated in collaboration with all HCPs and patients. This project determined ways to promote change for prescribers, but it will be important for further research to continue those conversations and incorporate patient perspectives.
- Use of funds to supply hypodermic needles or syringes for illegal drug use; prohibition, 42 USC § 300ee-5 (1988). Accessed January 5, 2026. https://www.law.cornell.edu /uscode/text/42/300ee-5
- OD2A Case Study: Harm Reduction. Centers for Disease Control and Prevention. June 9, 2025. Accessed January 5, 2026. https://www.cdc.gov/overdose-prevention/php /od2a/harm-reduction.html
- Strengthening Syringe Services Programs (SSPs). Centers for Disease Control and Prevention. March 20, 2024. Accessed January 5, 2026. https://www.cdc.gov/hepatitis -syringe-services/php/about/index.html
- Weinmeyer R. Needle exchange programs’ status in US politics. AMA J Ethics. 2016;18:252-257. doi:10.1001/journalofethics.2016.18.3.hlaw1-1603
- Rife-Pennington T, Dinges E, Ho MQ. Implementing syringe services programs within the Veterans Health Administration: facility experiences and next steps. J Am Pharm Assoc (2003). 2023;63:234-240. doi:10.1016/j.japh.2022.10.019
- The Biden-Harris Administration’s Statement of Drug Policy Priorities for Year One. Executive Office of the President, Office of National Drug Control Policy. April 1, 2021. Accessed January 5, 2026. https://bidenwhitehouse.archives.gov/wp-content/uploads/2021/03/BidenHarris -Statement-of-Drug-Policy-Priorities-April-1.pdf
- HIV - for veterans and the public syringe services programs. US Department of Veterans Affairs. Updated August 16, 2021. Accessed January 5, 2026. https://www.hiv .va.gov/patient/ssp.asp
- Trump Administration’s Statement of Drug Policy Priorities. White House. April 1, 2025. Accessed January 7, 2026. https://www.whitehouse.gov/wp-content /uploads/2025/04/2025-Trump-Administration-Drug-Policy -Priorities.pdf
- Health Centers and Syringe Services Programs. National Health Care for the Homeless Council. May 2023. Accessed January 5, 2026. https://nhchc.org/wp-content /uploads/2023/06/Health-Centers-SSPs-Final.pdf
- Lynch RD, Biederman DJ, Silva S, Demasi K. A syringe service program within a federal system: foundations for implementation. J Addict Nurs. 2021;32:152-158. doi:10.1097/JAN.0000000000000402
- Harvey LH, Sliwinski SK, Flike K, et al. The integration of harm reduction services in the Veterans Health Administration (VHA): a qualitative analysis of barriers and facilitators. J Addict Dis. 2024;42:326-334. doi:10.1080/10550887.2023.2210021
- Drug Overdose Death Rates. National Center for Drug Abuse Statistics. Accessed January 5, 2026. https:// drugabusestatistics.org/drug-overdose-deaths
- Ng G. New database shows Baltimore greatly devastated by opioid epidemic. Updated August 21, 2023. Accessed January 5, 2026. https://www.wbaltv.com/article/opioid -epidemic-database-baltimore-deaths/44869671
- Introductory Guide to Academic Detailing. National Resource Center for Academic Detailing. 2017. Accessed January 5, 2026. https://www.narcad.org /uploads/5/7/9/5/57955981/introductory_guide_to_ad.pdf
- Zhang J. Can educational outreach improve experts’ decision making? Evidence from a national opioid academic detailing program. SSRN. 2023;4297398. doi:10.2139/ssrn.4297398
- Watson H, Maclaren W, Kerr S. Staff attitudes towards working with drug users: development of the Drug Problems Perceptions Questionnaire. Addiction. 2007;102:206- 215. doi:10.1111/j.1360-0443.2006.01686.x
- Dahl RA, Vakkalanka JP, Harland KK, Radke J. Investigating healthcare provider bias toward patients who use drugs using a survey-based implicit association test: pilot study. J Addict Med. 2022;16:557-562. doi:10.1097/ADM.0000000000000970
- Hiatt JM, Creasey TJ. Change Management: The People Side of Change. Prosci Learning Center Publications; 2012.
- Wyse JJ, Shull S, Lindner S, et al. Access to medications for opioid use disorder in rural versus urban Veterans Health Administration facilities. J Gen Intern Med. 2023;38:1871-1876. doi:10.1007/s11606-023-08027-4
- Mostofian F, Ruban C, Simunovic N, Bhandari M. Changing physician behavior: what works?. Am J Manag Care. 2015;21(1):75-84.
- Bergdorf-Smith K, Bridge Initiative for S&T Policy, Leadership, and Communications. Syringe Service Programs and HIV Prevention in West Virginia. West Virginia University. February 5, 2024. Accessed January 5, 2026. https:// scitechpolicy.wvu.edu/science-and-technology-notes -articles/2024/02/05/syringe-service-programs-and-hiv -prevention-in-west-virginia
- Brunsdon N. Stop saying ‘clean’. Injecting Advice. February 7, 2011. Accessed January 5, 2026. https:// injectingadvice.com/stop-saying-clean/
- Use of funds to supply hypodermic needles or syringes for illegal drug use; prohibition, 42 USC § 300ee-5 (1988). Accessed January 5, 2026. https://www.law.cornell.edu /uscode/text/42/300ee-5
- OD2A Case Study: Harm Reduction. Centers for Disease Control and Prevention. June 9, 2025. Accessed January 5, 2026. https://www.cdc.gov/overdose-prevention/php /od2a/harm-reduction.html
- Strengthening Syringe Services Programs (SSPs). Centers for Disease Control and Prevention. March 20, 2024. Accessed January 5, 2026. https://www.cdc.gov/hepatitis -syringe-services/php/about/index.html
- Weinmeyer R. Needle exchange programs’ status in US politics. AMA J Ethics. 2016;18:252-257. doi:10.1001/journalofethics.2016.18.3.hlaw1-1603
- Rife-Pennington T, Dinges E, Ho MQ. Implementing syringe services programs within the Veterans Health Administration: facility experiences and next steps. J Am Pharm Assoc (2003). 2023;63:234-240. doi:10.1016/j.japh.2022.10.019
- The Biden-Harris Administration’s Statement of Drug Policy Priorities for Year One. Executive Office of the President, Office of National Drug Control Policy. April 1, 2021. Accessed January 5, 2026. https://bidenwhitehouse.archives.gov/wp-content/uploads/2021/03/BidenHarris -Statement-of-Drug-Policy-Priorities-April-1.pdf
- HIV - for veterans and the public syringe services programs. US Department of Veterans Affairs. Updated August 16, 2021. Accessed January 5, 2026. https://www.hiv .va.gov/patient/ssp.asp
- Trump Administration’s Statement of Drug Policy Priorities. White House. April 1, 2025. Accessed January 7, 2026. https://www.whitehouse.gov/wp-content /uploads/2025/04/2025-Trump-Administration-Drug-Policy -Priorities.pdf
- Health Centers and Syringe Services Programs. National Health Care for the Homeless Council. May 2023. Accessed January 5, 2026. https://nhchc.org/wp-content /uploads/2023/06/Health-Centers-SSPs-Final.pdf
- Lynch RD, Biederman DJ, Silva S, Demasi K. A syringe service program within a federal system: foundations for implementation. J Addict Nurs. 2021;32:152-158. doi:10.1097/JAN.0000000000000402
- Harvey LH, Sliwinski SK, Flike K, et al. The integration of harm reduction services in the Veterans Health Administration (VHA): a qualitative analysis of barriers and facilitators. J Addict Dis. 2024;42:326-334. doi:10.1080/10550887.2023.2210021
- Drug Overdose Death Rates. National Center for Drug Abuse Statistics. Accessed January 5, 2026. https:// drugabusestatistics.org/drug-overdose-deaths
- Ng G. New database shows Baltimore greatly devastated by opioid epidemic. Updated August 21, 2023. Accessed January 5, 2026. https://www.wbaltv.com/article/opioid -epidemic-database-baltimore-deaths/44869671
- Introductory Guide to Academic Detailing. National Resource Center for Academic Detailing. 2017. Accessed January 5, 2026. https://www.narcad.org /uploads/5/7/9/5/57955981/introductory_guide_to_ad.pdf
- Zhang J. Can educational outreach improve experts’ decision making? Evidence from a national opioid academic detailing program. SSRN. 2023;4297398. doi:10.2139/ssrn.4297398
- Watson H, Maclaren W, Kerr S. Staff attitudes towards working with drug users: development of the Drug Problems Perceptions Questionnaire. Addiction. 2007;102:206- 215. doi:10.1111/j.1360-0443.2006.01686.x
- Dahl RA, Vakkalanka JP, Harland KK, Radke J. Investigating healthcare provider bias toward patients who use drugs using a survey-based implicit association test: pilot study. J Addict Med. 2022;16:557-562. doi:10.1097/ADM.0000000000000970
- Hiatt JM, Creasey TJ. Change Management: The People Side of Change. Prosci Learning Center Publications; 2012.
- Wyse JJ, Shull S, Lindner S, et al. Access to medications for opioid use disorder in rural versus urban Veterans Health Administration facilities. J Gen Intern Med. 2023;38:1871-1876. doi:10.1007/s11606-023-08027-4
- Mostofian F, Ruban C, Simunovic N, Bhandari M. Changing physician behavior: what works?. Am J Manag Care. 2015;21(1):75-84.
- Bergdorf-Smith K, Bridge Initiative for S&T Policy, Leadership, and Communications. Syringe Service Programs and HIV Prevention in West Virginia. West Virginia University. February 5, 2024. Accessed January 5, 2026. https:// scitechpolicy.wvu.edu/science-and-technology-notes -articles/2024/02/05/syringe-service-programs-and-hiv -prevention-in-west-virginia
- Brunsdon N. Stop saying ‘clean’. Injecting Advice. February 7, 2011. Accessed January 5, 2026. https:// injectingadvice.com/stop-saying-clean/
Mental Health Prescribers’ Perceptions of Patients With Substance Use Disorders and Harm Reduction Services
Mental Health Prescribers’ Perceptions of Patients With Substance Use Disorders and Harm Reduction Services
Thermal Therapy is Associated With Decreased Pain Treatment Use: A Retrospective Cohort Study
Thermal Therapy is Associated With Decreased Pain Treatment Use: A Retrospective Cohort Study
Growing recognition of the limitations of long-term opioid therapy for chronic noncancer pain has highlighted the importance of nonpharmacologic approaches to pain treatment.1,2 These treatments are varied and may include psychological and behavioral therapies (eg, cognitive behavioral therapy for chronic pain), exercise and movement therapies (eg, yoga), and manual therapies (eg, chiropractic). Body surface cold therapy, while predominantly used to reduce postoperative pain and inflammation,3,4 pain subsequent to acute musculoskeletal injury,5 and pain related to muscle soreness,6 is a nonpharmacologic treatment that has shown pain-reducing benefits for chronic low back pain and knee osteoarthritis, as has its counterpart, superficial heat therapy.7-9 Heat therapy has also been shown to improve strength, flexibility, and activities of daily living in patients with chronic low back pain.10,11 Cold and heat therapies are commonly used complementarily. Cold therapies aim to reduce blood flow and inflammation and are often used immediately following trauma to an affected area, whereas heat therapies increase blood flow and metabolic activity and are commonly used to promote healing.5
Heat and cold therapies (also known as thermal therapies) benefit resource-limited health care systems, as most devices require a single expenditure and can be self-administered by patients at home as part of their pain self-management plan. In addition, these pain self-management tools may attenuate the need for more expensive specialty pain care and ongoing analgesic pharmacotherapy. Despite their potential, few studies have characterized the benefits of thermal therapies for patients with heterogeneous chronic pain syndromes.
The purpose of this retrospective cohort study was to examine the potential clinical and health system benefits of patient-administered thermal therapy. Our primary hypothesis was that patients with chronic musculoskeletal pain who received a thermal therapy self-management device would have fewer days of opioid prescriptions compared with a sample of matched control patients. Secondarily, we hypothesized that patients who received a thermal therapy device would have lower utilization of specialty pain care, fewer potentially hazardous opioid prescriptions (eg, high-dose opioid therapy and concurrent opioid and benzodiazepine prescriptions), fewer prescriptions for nonopioid analgesic medications, and decreased pain intensity when compared with matched controls.
Methods
This retrospective cohort study compared pain pharmacotherapy, pain treatment utilization, and pain intensity outcomes between patients who received a thermal therapy device and matched patients who did not. The study was approved by the US Department of Veterans Affairs (VA) Portland Health Care System Institutional Review Board and was granted a waiver of informed consent to access patient electronic health records (EHRs).
Pain Care
The VA uses ThermaZone thermal therapy devices (Innovative Medical Equipment) for chronic pain treatment. The device uses thermoelectric technology to provide point-of-contact cooling and heating therapy through site-specific pads (eg, ankle, knee, hip, back, elbow, shoulder). Patients place pads on sites where they experience pain, and temperature regulated water circulates through the device and to the pad, providing consistent, localized thermal therapy. The pads range in temperature from 1 °C to 52 °C, and temperatures are self-monitored and controlled by the patient.
Standard pain care in this study followed the VA stepped model of pain care, which builds on a foundation of patient education for pain self-management approaches (eg, exercise, mindfulness, relaxation, social support).12 According to the VA stepped model of pain care, all patients with chronic pain should engage in these foundational self-management approaches. However, some patients require more intensive care. The VA stepped-up treatment engages primary and specialty care services—such as physical therapy, pharmacy, complementary and integrative health approaches, mental health, and substance use services—and, when indicated, may escalate treatment to interdisciplinary pain teams or tertiary pain centers. In this retrospective cohort study, treatment patients received a thermal therapy device and standard of care, whereas control patients received standard care only.
Sample Selection
Eligible patients were aged ≥ 18 years, had a musculoskeletal pain diagnosis documented in the EHR in the year prior to thermal therapy device receipt (or during the same period for a treatment patient’s matched control), and were enrolled in VA health care during the study period. Patients who died during the study period were excluded. Treatment patients received a thermal therapy device from the VA between January 1, 2017, and December 31, 2018, when complete data on thermal therapy devices were available. For control patients, the VA Corporate Data Warehouse (CDW) was used to identify VA patients with characteristics similar to those of treated patients.
We modeled the probability that a patient would receive a thermal therapy device using logistic regression. Predictor variables were measured in the year prior to device receipt and included variables associated with pain treatment utilization and analgesic pharmacotherapy receipt, as recommended by Brookhart et al.13 These included age, sex, race, ethnicity, VA service-connected disability status, comorbidities, receipt of medications for opioid use disorder, pain diagnoses, mental health diagnoses, and substance use disorder diagnoses.14-19
The resulting propensity scores (eg, predicted probabilities) were used to match treatment patients 1:1 with control patients using a nearest-neighbor matching algorithm.20 This procedure matches a control patient with the closest propensity score to that of the corresponding treatment patient. An exact match on VA facility was required (eg, treatment patients and matched controls received care at the same VA facility). Standardized differences were used to assess covariate balance between the matched groups, and kernel density plots of propensity scores tested for sufficient overlap.21 Control patients were selected from a pool of 1,150,149 patients.
Study Variables
The index date was the date the thermal therapy device was released to treatment patients or the same date for the matched controls. Data were extracted from the CDW over a 24-month period: 12 months prior to the index date through 12 months afterwards. Collecting data in the 12 months prior to treatment initiation allowed us to adjust for covariates and provided greater precision, as recommended for observational study designs.22
Treatment conditions were defined dichotomously as receipt vs nonreceipt of a thermal therapy device. The primary outcome was the number of days of opioid use in the 12 months following the index date. Additional outcomes included days of high-dose opioid therapy (≥ 50 mg morphine equivalent [MME] daily), concurrent opioid and benzodiazepine prescriptions, and nonopioid analgesic pharmacotherapy (eg, nonsteroidal anti-inflammatory drugs, acetaminophen, muscle relaxants). All prescription data were based on medication fills obtained from VA pharmacy records. Additional outcomes included the number of visits to physical therapy, occupational therapy, physical medicine and rehabilitation, and interdisciplinary pain clinics, including interventional pain medicine.
Pain intensity ratings were collected as part of routine VA care using a numeric scale from 0 (no pain) to 10 (worst possible pain). Pain intensity trajectories were computed using all available EHR-derived pain intensity score data for each patient in the 12 months prior to and following the index date.
Covariates were extracted from the EHR and evaluated in the year prior to the index date, unless otherwise noted. They included age at the index date; self-reported sex, and race and ethnicity; service-connected disability status (disability awarded as a result of military service-related trauma or injury); Charlson Comorbidity Index; and diagnoses of opioid use disorder, alcohol use disorder, other substance use disorder, mood disorder, posttraumatic stress disorder, other anxiety disorder, psychotic disorder, neuropathic pain, and headache pain.23 All diagnoses were coded as yes if identified in the EHR as a focus of treatment during any clinical encounter in the year prior to the index date or no if not identified as a focus of treatment.
The number of days patients had been living with pain was calculated and defined as the number of days from the first pain diagnosis available in a patient’s EHR to the index date. Finally, the number of pain-related surgeries (eg, surgeries with ≥ 1 pain diagnoses associated with the clinical encounter) and average pain intensity were computed for the pre- and postindex date evaluation periods and included as model covariates.
Statistical Analyses
For the 4 pharmacotherapy and 4 nonpharmacologic treatment usage outcomes, we tested the fit of linear models and several models with count distributions using the Bayesian information criterion.24 Count distributions included Poisson, zero-inflated Poisson, negative binomial, and zero-inflated negative binomial. With 1 exception (days of high-dose opioid use), a negative binomial distribution best fit the data. For days of high-dose opioid use, a Gaussian distribution best fit the data.
Eight separate mixed-effects regression analyses compared changes in each outcome from the 12-month preindex period through the 12-month postindex period between treatment and control patients by testing the Time × Treatment interaction. This approach statistically accounts for observed pretreatment differences in outcome variables. Statistics for the main effects of time and treatment are also presented. To reduce bias, models controlled for covariates specified previously.
For pain intensity, we used random-effects growth modeling to quantify both fixed and random effects of pain intensity at the index date (eg, the model intercept), which estimates pain at the time of treatment initiation, and change in pain during the 12 months following the index date (eg, the model slope), which characterizes the trajectory of pain intensity ratings.25 The model included piecewise components of pain score trajectories in the 12 months prior to the index date and in the 12 months following the index date. Several types of change for the 12-month postindex observation period were explored—including quadratic and cubic curvilinear change. A linear model for change in pain over time provided the best fit based on the Bayesian information criterion and parsimony of model parameters.26 We report estimates of change in pain over time in monthly intervals for ease of interpretation. However, models used all individual pain scores rather than computing monthly averages when > 1 pain score was available within a month, as recommended in previous research.27 This approach makes optimal use of all available data. Both random effects (intercept and slope) were regressed onto the set of covariates described previously.
This study used data available in the EHR over the 24-month observation period. We characterize the density (eg, frequency) of all outcome variables by treatment condition in the Appendix. Because the hypotheses were directional, the authors used a 2-sided α = 0.10 and applied a Bonferroni correction for the 9 statistical tests performed, resulting in an adjusted α of 0.01. Treatment utilization and pharmacotherapy outcome analyses were performed in Stata, version 16.1. Random-effects growth modeling of pain score trajectories was performed using Mplus, version 8.8.
Results
There were 2182 patients in the treatment group and 2182 matched controls. The mean (SD) age was 54 (15) years; 81% were male, and about two-thirds (68%) identified as White and non-Hispanic. Mental health comorbidities were common, with > 40% of the sample having diagnoses of a mood disorder and/or posttraumatic stress disorder. Nearly all patients (90%) had VA service-connected disability ratings. Among patients with chronic musculoskeletal pain, 8% had comorbid neuropathic pain and 14% had headache. The mean (SD) duration of chronic pain across both patient groups was 3416 (2016) days, or about 9.4 years (Table 1).
Pharmacotherapy
High-dose opioid use (> 50 MME daily dose), days of opioid use, and concurrent opioid and benzodiazepine use decreased for all patients from the pre- to posttreatment period (Table 2). However, high-dose opioid use (Time × Treatment interaction, 3.24; 99% CI, 0.34 to 6.14) (Figure 1A) and concurrent opioid and benzodiazepine use (Time × Treatment interaction, 0.76; 99% CI, 0.67 to 0.86) (Figure 1B) had a larger decrease for the treatment vs matched control group. Treatment and matched control patients had comparable reductions in days of opioid use (Time × Treatment interaction, 0.98; 99% CI, 0.91 to 1.05) (Figure 1C). Neither group showed changes in nonopioid analgesic pharmacotherapy over time (main effect of time, incidence rate ratio [IRR], 1.03; 99% CI, 0.99 to 1.07; Time × Treatment interaction, 0.95; 99% CI, 0.90 to 1.01) (Figure 1D).
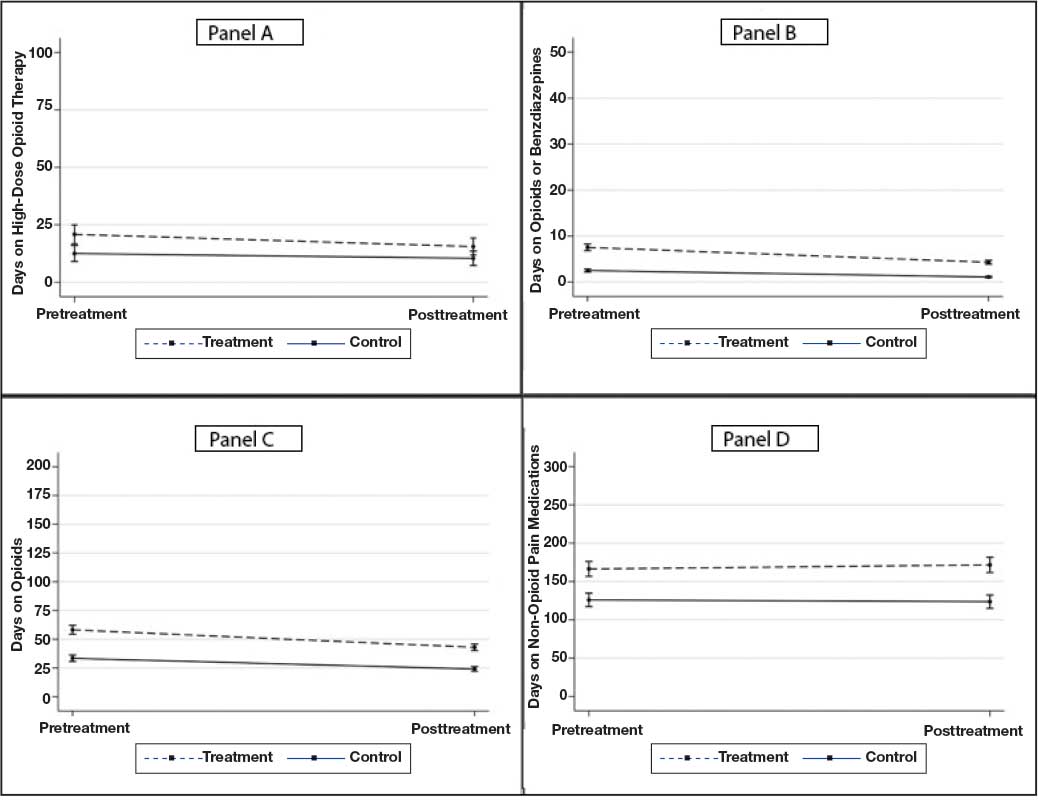
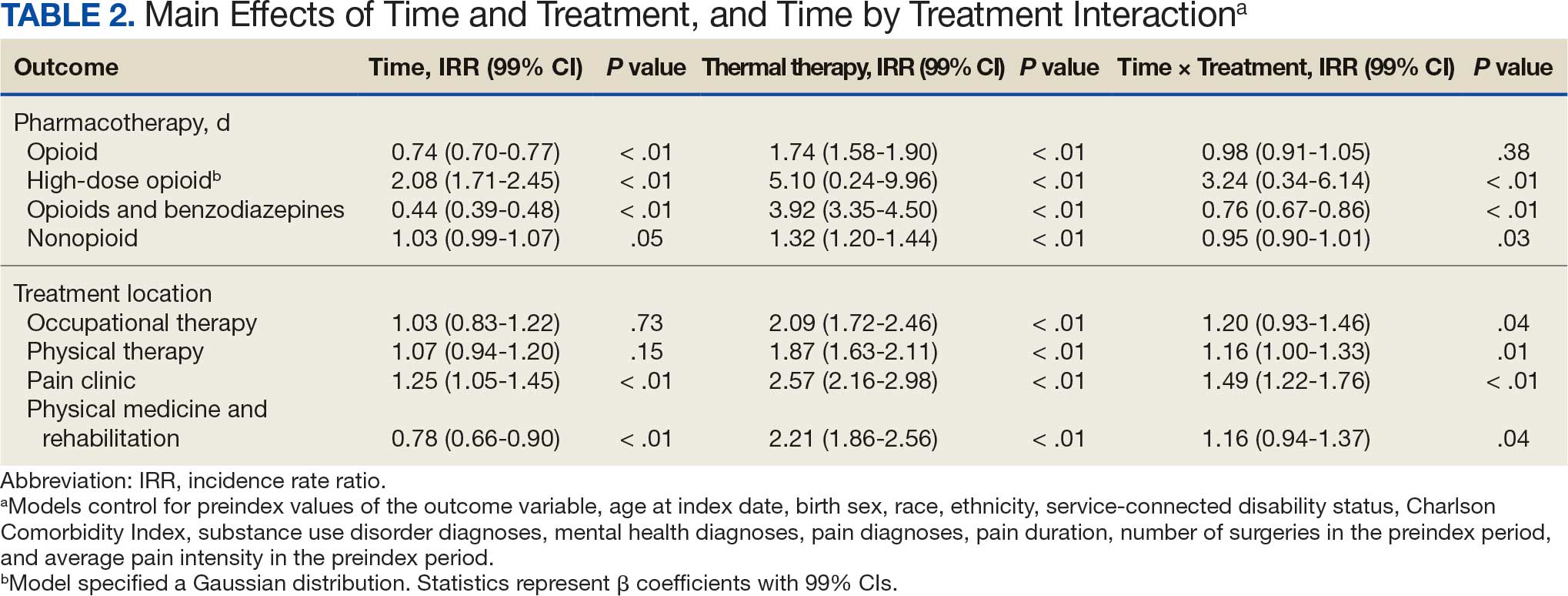
Nonpharmacologic Pain Treatment
The number of physical therapy and pain clinic visits declined for treatment patients and increased slightly for matched control patients (Figure 2A and 2B). For occupational therapy visits, neither group showed changes over time (main effect of time IRR, 1.03; 99% CI, 0.83 to 1.22; Time × Treatment interaction, 1.20; 99% CI, 0.93 to 1.46) (Figure 2C). For physical medicine and rehabilitation visits, both groups decreased use over time (main effect of time IRR, 0.78; 99% CI, 0.66 to 0.90), but this decrease did not differ between treatment and control patients (Time × Treatment interaction, 1.16; 99% CI, 0.94 to 1.37) (Figure 2D).
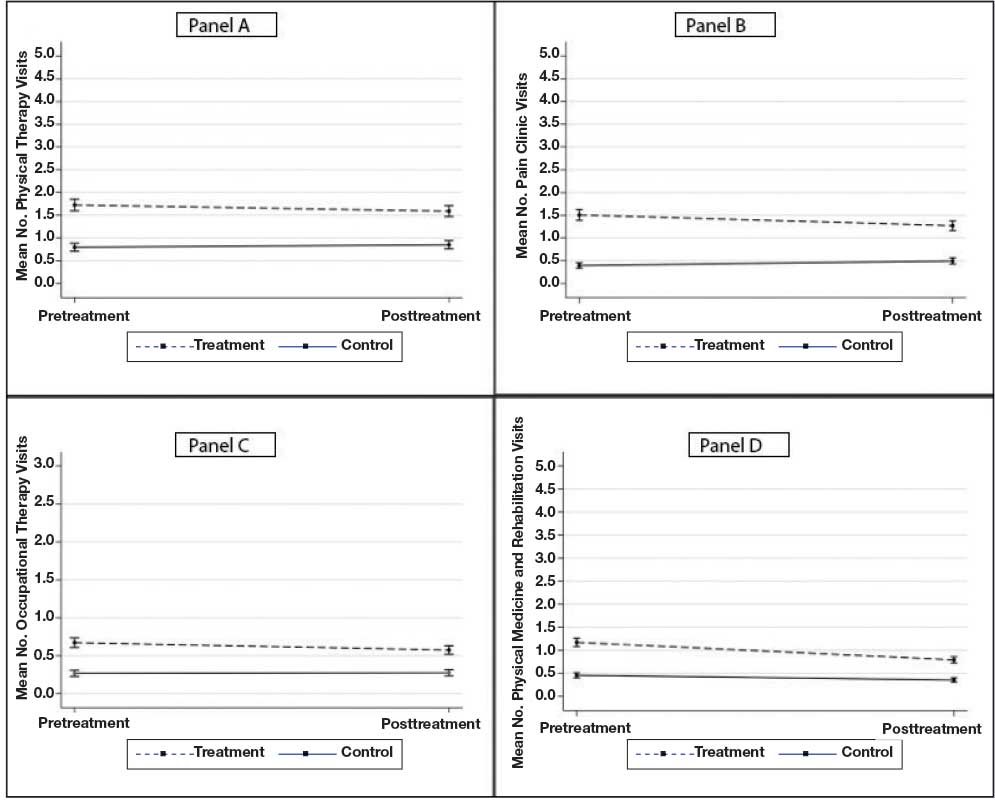
Pain Intensity
Pain intensity decreased for both groups by an estimated 0.02 points per month (99% CI, -0.04 to -0.01; P < .01), or 0.24 points over the 12-month postindex follow-up period (Figure 3). There were no statistically significant differences between treatment and control patients in pain intensity changes over the follow-up period (B = -0.02; 99% CI, -0.06 to 0.02; P = .15).
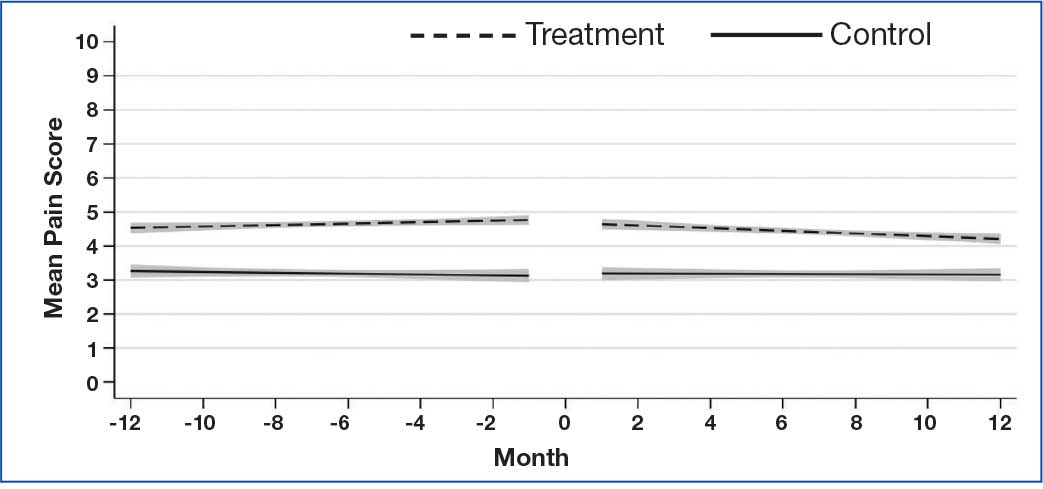
Discussion
Patients with musculoskeletal pain who received a thermal therapy device showed a larger decline in use of some specialty pain treatments, including physical therapy and specialty pain clinic services, when compared with matched control patients. One possible explanation is that patients who used the device may have had their pain adequately managed and thus required less specialty pain care. However, the absence of clinically significant changes in pain intensity over time suggests that pain intensity alone did not account for the observed changes in pain treatment use. We were unable to measure constructs of pain interference or functioning, which may be better predictors of functional restoration, as these data are not routinely collected within the VA and documented in the EHR. Future studies could clarify these findings by including measures of pain interference and functioning.
Although the overall declines in physical therapy and specialty pain clinic use associated with thermal therapy were modest (< 1 visit per patient), the impact of these reductions can be profound. In resource-limited health care settings, even small reductions in high-cost care utilization could be of great value in that health systems could offset costs associated with treating chronic pain without compromising quality of care or key clinical outcomes, such as pain intensity. This study, however, did not include a cost analysis. Future studies should incorporate formal cost analyses to quantify cost offsets that may result from decreased specialty pain treatment use.
Patients who received thermal therapy devices did not show clinically meaningful reductions in pain intensity over time, defined as reductions in pain intensity of 1.7 to 2.0 on a 0 to 10 scale.28,29 This finding is consistent with prior research that demonstrates relatively stable pain intensity self-ratings longitudinally by patients with chronic pain diagnoses, when assessed in the context of usual clinical care.30 This finding, however, is inconsistent with prior literature that demonstrates pain-reducing benefits of thermal therapy for low back pain and knee osteoarthritis.7-9
In this study, pain intensity ratings were derived from the EHR during routine outpatient clinical encounters and not at the time thermal therapy was self-administered, as has been done in prior clinical trials.7-9 Pain location was not specified at the time of pain ratings, and it is possible that patients may have been endorsing pain in areas of the body that had not been treated by thermal therapy. Patient-level variability in pain intensity ratings (eg, within-patient range over time) was not examined, although prior research indicates substantial variability.30 While average pain intensity ratings in the current study did not change, an examination of patient variability warrants further study, as a narrowing of pain intensity ratings can be perceived, by patients, as demonstrable improvement and has been associated with improved physical and psychological outcomes.31 Furthermore, pain intensity does not characterize physical or emotional functioning that can be captured with more comprehensive validated measures, some of which are recommended outcomes in pain clinical trials.32
Our findings point to reductions in all forms of opioid use across both treatment and control patients. Data from the VA and in the US more generally point to downward trends in opioid prescribing during the study period.33 This decline is likely due to increased use of risk mitigation approaches, such as routine urine drug screens and review of prescription drug monitoring databases.34 These state-level databases track prescribing of controlled substances, including opioids and benzodiazepines, within a state. Implementation of these practices has been associated with declines in higher risk opioid prescribing.34 Findings from this study further point to associations of reduced higher risk opioid use among patients who received thermal therapy devices. In the full sample of patients, reductions in days of opioid use, high-dose opioid use, and co-use of opioids and benzodiazepines were observed across all patients, with greater reductions observed in high-dose opioid use and concurrent opioid and benzodiazepine use among patients who received a thermal therapy device. Experimental studies indicate that the endogenous opioid system is implicated in pain relief and activated by thermal therapies. 35 Differential reductions in higher-risk opioid use among patients who received thermal therapy devices in our study may be associated with endogenous opioid activation, though this was not specifically measured. It is also unclear whether thermal therapy was provided by clinicians in the context of opioid tapering or other risk mitigation efforts, or patients reduced higher risk opioid use of their own volition. Prior research has identified both patient- and clinician-initiated opioid tapering and discontinuation.36 While a thorough explication of opioid dose reduction was beyond the scope of this study, future qualitative work could help explain potential benefits of thermal therapy in the context of analgesic pharmacotherapy use, including opioid medications.
Limitations
The extent to which patients used the thermal therapy device could not be measured; therefore, device receipt was used as a proxy for use. However, it was not possible to determine whether the frequency and duration of device use was associated with study outcomes. Treatment and control groups demonstrated some differences in outcome variables at the index date. Potential known biases were addressed using propensity score matching procedures and statistical procedures that controlled for patient demographic and clinical characteristics, as well as pretreatment values of all outcome variables. Nevertheless, as an observational study, this analysis cannot account for all known and unknown confounders, and a randomized controlled trial is needed to make claims of causality. The study population consisted of US veterans and included a low proportion of women. As a result, the findings may not be generalizable to other patient populations. Finally, prescription dispensing data, used as a proxy for pharmacotherapy use, do not necessarily reflect actual medication use.
Conclusions
This study is among the first to examine associations between thermal therapy and specialty pain treatment and analgesic pharmacotherapy use among US veterans. Although the retrospective cohort study design does not allow causal inferences regarding the efficacy of thermal therapy for veterans with chronic musculoskeletal pain, confidence in the findings is strengthened by methodological and statistical control of known confounders. Future trials employing experimental designs are needed to further clarify the clinical and health systems benefits of thermal therapy for musculoskeletal pain syndromes.
- Krebs EE, Gravely A, Nugent S, et al. Effect of opioid vs nonopioid medications on pain-related function in patients with chronic back pain or hip or knee osteoarthritis pain: the SPACE randomized clinical trial. JAMA. 2018;2018:872-882. doi:10.1001/jama.2018.0899
- Becker WC, DeBar LL, Heapy AA, et al. A research agenda for advancing non-pharmacological management of chronic musculoskeletal pain: findings from a VHA state-of-the-art conference. J Gen Intern Med. 2018;33:1-15. doi:10.1007/s11606-018-4345-6
- Fernandes IA, Armond ACV, Falci SGM. The effectiveness of the cold therapy (cryotherapy) in the management of inflammatory parameters after removal of mandibular third molars: a meta-analysis. Int Arch Otorhinolaryngol. 2019;23:221-228. doi:10.1055/s-0039-1677755
- Quinlan P, Davis J, Fields K, et al. Effects of localized cold therapy on pain in postoperative spinal fusion patients: a randomized controlled trial. Orthop Nurs. 2017;36:344-349. doi:10.1097/NOR.0000000000000382
- Malanga GA, Yan N, Stark J. Mechanisms and efficacy of heat and cold therapies for musculoskeletal injury. Postgrad Med. 2015;127:57-65. doi:10.1080/00325481.2015.992719
- Wang Y, Li S, Zhang Y, et al. Heat and cold therapy reduce pain in patients with delayed onset muscle soreness: a systematic review and meta-analysis of 32 randomized controlled trials. Phys Ther Sport. 2021;48:177-187. doi:10.1016/j.ptsp.2021.01.004
- Ariana M, Afrasiabifar A, Doulatabad SN, et al. The effect of local heat therapy versus cold rub gel on pain and joint functions in patients with knee osteoarthritis. Clin Nurs Res. 2022;31:1014-1022. doi:10.1177/10547738211035502
- French SD, Cameron M, Walker BF, et al. Superficial heat or cold for low back pain. Cochrane Database Syst Rev. 2006;1:CD004750. doi:10.1002/14651858.CD004750.pub2
- Tao XG, Bernacki EJ. A randomized clinical trial of continuous low-level heat therapy for acute muscular low back pain in the workplace. J Occup Environ Med. 2005;47:1298- 1306. doi:10.1097/01.jom.0000184877.01691.a3
- Freiwald J, Hoppe MW, Beermann W, et al. Effects of supplemental heat therapy in multimodal treated chronic low back pain patients on strength and flexibility. Clin Biomech. 2018;57:107-113. doi:10.1016/j.clinbiomech.2018.06.008
- Freiwald J, Magni A, Fanlo-Mazas P, et al. A role for superficial heat therapy in the management of nonspecific, mild-to-moderate low back pain in current clinical practice: a narrative review. Life. 2021;11:780. doi:10.3390/life11080780
- Kerns RD, Philip EJ, Lee AW, et al. Implementation of the Veterans Health Administration National Pain Management Strategy. Transl Behav Med. 2011;1:635-643. doi:10.1007/s13142-011-0094-3
- Brookhart MA, Schneeweiss S, Rothman KJ, et al. Variable selection for propensity score models. Am J Epidemiol. 2006;163:1149-1156. doi:10.1093/aje/kwj149
- Edlund MJ, Martin BC, Devries A, et al. Trends in use of opioids for chronic non-cancer pain among individuals with mental health and substance use disorders: the TROUP study. Clin J Pain. 2010;26:1-8. doi:10.1097/AJP.0b013e3181b99f35
- Kaur S, Stechuchak KM, Coffman CJ, et al. Gender differences in health care utilization among veterans with chronic pain. J Gen Intern Med. 2007;22:228-233. doi:10.1007/s11606-006-0048-5
- Meghani SH, Cho E. Self-reported pain and utilization of pain treatment between minorities and non-minorities in the United States. Public Health Nurs. 2009;26:307-316. doi:10.1111/j.1525-1446.2009.00785.x
- Morasco BJ, Duckart JP, Carr TP, et al. Clinical characteristics of veterans prescribed high doses of opioid medications for chronic non-cancer pain. Pain. 2010;151:625-632. doi:10.1016/j.pain.2010.08.002
- Nielsen S, Lintzeris N, Bruno R, et al. Benzodiazepine use among chronic pain patients prescribed opioids: associations with pain, physical and mental health, and health service utilization. Pain Med. 2015;16:356-366. doi:10.1111/pme.12594
- Quinn PD, Hur K, Chang Z, et al. Incident and long-term opioid therapy among patients with psychiatric conditions and medications: a national study of commercial health care claims. Pain. 2017;158:140-148. doi:10.1097/j.pain.0000000000000730
- Rubin DB. Matching to remove bias in observational studies. Biometrics. 1973;29:159-183.
- Rosenbaum PR, Rubin DB. Constructing a control group using multivariate matched sampling methods that incorporate the propensity score. Am Stat. 1985;39:33-38. doi:10.1007/s11596-025-00052-0
- Steiner PM, Cook TD, Shadish WR, et al. The importance of covariate selection in controlling for selection bias in observational studies. Psychol Methods. 2010;15:250-267. doi:10.1037/a0018719
- Charlson ME, Pompei P, Ales KL, et al. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40:373- 383. doi:10.1016/0021-9681(87)90171-8
- Long JS, Freese J. Regression Models for Categorical Dependent Variables Using Stata. 3rd ed. Stata Press; 2014.
- Muthén LK, Muthén BO. Mplus User’s Guide. 8th ed. Muthén & Muthén; 1998-2017.
- Hedeker D, Gibbons RD. Longitudinal Data Analysis. Wiley; 2006.
- Dobscha SK, Morasco BJ, Kovas AE, et al. Short-term variability in outpatient pain intensity scores in a national sample of older veterans with chronic pain. Pain Med. 2015;16:855-865. doi:10.1111/pme.12643
- Bahreini M, Safaie A, Mirfazaelian H, et al. How much change in pain score does really matter to patients? Am J Emerg Med. 2020;38:1641-1646. doi:10.1016/j.ajem.2019.158489
- Suzuki H, Aono S, Inoue S, et al. Clinically significant changes in pain along the pain intensity numerical rating scale in patients with chronic low back pain. PLoS One. 2020;15:e0229228. doi:10.1371/journal.pone.0229228
- McPherson SM, Smith CL, Dobscha SK, et al. Changes in pain intensity after discontinuation of long-term opioid therapy for chronic noncancer pain. Pain. 2018;159:2097- 2104. doi:10.1097/j.pain.0000000000001315
- Andrews NE, Strong J, Meredith PJ. Activity pacing, avoidance, endurance, and associations with patient functioning in chronic pain: a systematic and meta-analysis. Arch Phys Med Rehabil. 2012;93:2109-121. doi:10.1016/j.apmr.2012.05.029
- Dworkin RH, Turk DC, Farrar JT, et al. Core outcome measures for chronic pain clinical trials: IMMPACT recommendations. Pain. 2005;113:9-19. doi:10.1016/j.pain.2004.09.012
- Gellad WF, Good CB, Shulkin DJ. Addressing the opioid epidemic in the United States: Lessons from the Department of Veterans Affairs. JAMA Intern Med. 2017;177:611- 612. doi:10.1001/jamainternmed.2017.0147
- Lin LA, Bohnert ASB, Kerns RD, et al. Impact of the Opioid Safety Initiative on opioid-related prescribing in veterans. Pain. 2017;158:833-839. doi:10.1097/j.pain.0000000000000837
- Sirucek L, Price RC, Gandhi W, et al. Endogenous opioids contribute to the feeling of pain relief in humans. Pain. 2021;162:2821-2831. doi:10.1097/j.pain.0000000000002285
- Lovejoy TI, Morasco BJ, Demidenko MI, et al. Reasons for discontinuation of long-term opioid therapy in patients with and without substance use disorders. Pain. 2017;158:526- 534. doi:10.1097/j.pain.0000000000000796
Growing recognition of the limitations of long-term opioid therapy for chronic noncancer pain has highlighted the importance of nonpharmacologic approaches to pain treatment.1,2 These treatments are varied and may include psychological and behavioral therapies (eg, cognitive behavioral therapy for chronic pain), exercise and movement therapies (eg, yoga), and manual therapies (eg, chiropractic). Body surface cold therapy, while predominantly used to reduce postoperative pain and inflammation,3,4 pain subsequent to acute musculoskeletal injury,5 and pain related to muscle soreness,6 is a nonpharmacologic treatment that has shown pain-reducing benefits for chronic low back pain and knee osteoarthritis, as has its counterpart, superficial heat therapy.7-9 Heat therapy has also been shown to improve strength, flexibility, and activities of daily living in patients with chronic low back pain.10,11 Cold and heat therapies are commonly used complementarily. Cold therapies aim to reduce blood flow and inflammation and are often used immediately following trauma to an affected area, whereas heat therapies increase blood flow and metabolic activity and are commonly used to promote healing.5
Heat and cold therapies (also known as thermal therapies) benefit resource-limited health care systems, as most devices require a single expenditure and can be self-administered by patients at home as part of their pain self-management plan. In addition, these pain self-management tools may attenuate the need for more expensive specialty pain care and ongoing analgesic pharmacotherapy. Despite their potential, few studies have characterized the benefits of thermal therapies for patients with heterogeneous chronic pain syndromes.
The purpose of this retrospective cohort study was to examine the potential clinical and health system benefits of patient-administered thermal therapy. Our primary hypothesis was that patients with chronic musculoskeletal pain who received a thermal therapy self-management device would have fewer days of opioid prescriptions compared with a sample of matched control patients. Secondarily, we hypothesized that patients who received a thermal therapy device would have lower utilization of specialty pain care, fewer potentially hazardous opioid prescriptions (eg, high-dose opioid therapy and concurrent opioid and benzodiazepine prescriptions), fewer prescriptions for nonopioid analgesic medications, and decreased pain intensity when compared with matched controls.
Methods
This retrospective cohort study compared pain pharmacotherapy, pain treatment utilization, and pain intensity outcomes between patients who received a thermal therapy device and matched patients who did not. The study was approved by the US Department of Veterans Affairs (VA) Portland Health Care System Institutional Review Board and was granted a waiver of informed consent to access patient electronic health records (EHRs).
Pain Care
The VA uses ThermaZone thermal therapy devices (Innovative Medical Equipment) for chronic pain treatment. The device uses thermoelectric technology to provide point-of-contact cooling and heating therapy through site-specific pads (eg, ankle, knee, hip, back, elbow, shoulder). Patients place pads on sites where they experience pain, and temperature regulated water circulates through the device and to the pad, providing consistent, localized thermal therapy. The pads range in temperature from 1 °C to 52 °C, and temperatures are self-monitored and controlled by the patient.
Standard pain care in this study followed the VA stepped model of pain care, which builds on a foundation of patient education for pain self-management approaches (eg, exercise, mindfulness, relaxation, social support).12 According to the VA stepped model of pain care, all patients with chronic pain should engage in these foundational self-management approaches. However, some patients require more intensive care. The VA stepped-up treatment engages primary and specialty care services—such as physical therapy, pharmacy, complementary and integrative health approaches, mental health, and substance use services—and, when indicated, may escalate treatment to interdisciplinary pain teams or tertiary pain centers. In this retrospective cohort study, treatment patients received a thermal therapy device and standard of care, whereas control patients received standard care only.
Sample Selection
Eligible patients were aged ≥ 18 years, had a musculoskeletal pain diagnosis documented in the EHR in the year prior to thermal therapy device receipt (or during the same period for a treatment patient’s matched control), and were enrolled in VA health care during the study period. Patients who died during the study period were excluded. Treatment patients received a thermal therapy device from the VA between January 1, 2017, and December 31, 2018, when complete data on thermal therapy devices were available. For control patients, the VA Corporate Data Warehouse (CDW) was used to identify VA patients with characteristics similar to those of treated patients.
We modeled the probability that a patient would receive a thermal therapy device using logistic regression. Predictor variables were measured in the year prior to device receipt and included variables associated with pain treatment utilization and analgesic pharmacotherapy receipt, as recommended by Brookhart et al.13 These included age, sex, race, ethnicity, VA service-connected disability status, comorbidities, receipt of medications for opioid use disorder, pain diagnoses, mental health diagnoses, and substance use disorder diagnoses.14-19
The resulting propensity scores (eg, predicted probabilities) were used to match treatment patients 1:1 with control patients using a nearest-neighbor matching algorithm.20 This procedure matches a control patient with the closest propensity score to that of the corresponding treatment patient. An exact match on VA facility was required (eg, treatment patients and matched controls received care at the same VA facility). Standardized differences were used to assess covariate balance between the matched groups, and kernel density plots of propensity scores tested for sufficient overlap.21 Control patients were selected from a pool of 1,150,149 patients.
Study Variables
The index date was the date the thermal therapy device was released to treatment patients or the same date for the matched controls. Data were extracted from the CDW over a 24-month period: 12 months prior to the index date through 12 months afterwards. Collecting data in the 12 months prior to treatment initiation allowed us to adjust for covariates and provided greater precision, as recommended for observational study designs.22
Treatment conditions were defined dichotomously as receipt vs nonreceipt of a thermal therapy device. The primary outcome was the number of days of opioid use in the 12 months following the index date. Additional outcomes included days of high-dose opioid therapy (≥ 50 mg morphine equivalent [MME] daily), concurrent opioid and benzodiazepine prescriptions, and nonopioid analgesic pharmacotherapy (eg, nonsteroidal anti-inflammatory drugs, acetaminophen, muscle relaxants). All prescription data were based on medication fills obtained from VA pharmacy records. Additional outcomes included the number of visits to physical therapy, occupational therapy, physical medicine and rehabilitation, and interdisciplinary pain clinics, including interventional pain medicine.
Pain intensity ratings were collected as part of routine VA care using a numeric scale from 0 (no pain) to 10 (worst possible pain). Pain intensity trajectories were computed using all available EHR-derived pain intensity score data for each patient in the 12 months prior to and following the index date.
Covariates were extracted from the EHR and evaluated in the year prior to the index date, unless otherwise noted. They included age at the index date; self-reported sex, and race and ethnicity; service-connected disability status (disability awarded as a result of military service-related trauma or injury); Charlson Comorbidity Index; and diagnoses of opioid use disorder, alcohol use disorder, other substance use disorder, mood disorder, posttraumatic stress disorder, other anxiety disorder, psychotic disorder, neuropathic pain, and headache pain.23 All diagnoses were coded as yes if identified in the EHR as a focus of treatment during any clinical encounter in the year prior to the index date or no if not identified as a focus of treatment.
The number of days patients had been living with pain was calculated and defined as the number of days from the first pain diagnosis available in a patient’s EHR to the index date. Finally, the number of pain-related surgeries (eg, surgeries with ≥ 1 pain diagnoses associated with the clinical encounter) and average pain intensity were computed for the pre- and postindex date evaluation periods and included as model covariates.
Statistical Analyses
For the 4 pharmacotherapy and 4 nonpharmacologic treatment usage outcomes, we tested the fit of linear models and several models with count distributions using the Bayesian information criterion.24 Count distributions included Poisson, zero-inflated Poisson, negative binomial, and zero-inflated negative binomial. With 1 exception (days of high-dose opioid use), a negative binomial distribution best fit the data. For days of high-dose opioid use, a Gaussian distribution best fit the data.
Eight separate mixed-effects regression analyses compared changes in each outcome from the 12-month preindex period through the 12-month postindex period between treatment and control patients by testing the Time × Treatment interaction. This approach statistically accounts for observed pretreatment differences in outcome variables. Statistics for the main effects of time and treatment are also presented. To reduce bias, models controlled for covariates specified previously.
For pain intensity, we used random-effects growth modeling to quantify both fixed and random effects of pain intensity at the index date (eg, the model intercept), which estimates pain at the time of treatment initiation, and change in pain during the 12 months following the index date (eg, the model slope), which characterizes the trajectory of pain intensity ratings.25 The model included piecewise components of pain score trajectories in the 12 months prior to the index date and in the 12 months following the index date. Several types of change for the 12-month postindex observation period were explored—including quadratic and cubic curvilinear change. A linear model for change in pain over time provided the best fit based on the Bayesian information criterion and parsimony of model parameters.26 We report estimates of change in pain over time in monthly intervals for ease of interpretation. However, models used all individual pain scores rather than computing monthly averages when > 1 pain score was available within a month, as recommended in previous research.27 This approach makes optimal use of all available data. Both random effects (intercept and slope) were regressed onto the set of covariates described previously.
This study used data available in the EHR over the 24-month observation period. We characterize the density (eg, frequency) of all outcome variables by treatment condition in the Appendix. Because the hypotheses were directional, the authors used a 2-sided α = 0.10 and applied a Bonferroni correction for the 9 statistical tests performed, resulting in an adjusted α of 0.01. Treatment utilization and pharmacotherapy outcome analyses were performed in Stata, version 16.1. Random-effects growth modeling of pain score trajectories was performed using Mplus, version 8.8.

Results
There were 2182 patients in the treatment group and 2182 matched controls. The mean (SD) age was 54 (15) years; 81% were male, and about two-thirds (68%) identified as White and non-Hispanic. Mental health comorbidities were common, with > 40% of the sample having diagnoses of a mood disorder and/or posttraumatic stress disorder. Nearly all patients (90%) had VA service-connected disability ratings. Among patients with chronic musculoskeletal pain, 8% had comorbid neuropathic pain and 14% had headache. The mean (SD) duration of chronic pain across both patient groups was 3416 (2016) days, or about 9.4 years (Table 1).

Pharmacotherapy
High-dose opioid use (> 50 MME daily dose), days of opioid use, and concurrent opioid and benzodiazepine use decreased for all patients from the pre- to posttreatment period (Table 2). However, high-dose opioid use (Time × Treatment interaction, 3.24; 99% CI, 0.34 to 6.14) (Figure 1A) and concurrent opioid and benzodiazepine use (Time × Treatment interaction, 0.76; 99% CI, 0.67 to 0.86) (Figure 1B) had a larger decrease for the treatment vs matched control group. Treatment and matched control patients had comparable reductions in days of opioid use (Time × Treatment interaction, 0.98; 99% CI, 0.91 to 1.05) (Figure 1C). Neither group showed changes in nonopioid analgesic pharmacotherapy over time (main effect of time, incidence rate ratio [IRR], 1.03; 99% CI, 0.99 to 1.07; Time × Treatment interaction, 0.95; 99% CI, 0.90 to 1.01) (Figure 1D).


Nonpharmacologic Pain Treatment
The number of physical therapy and pain clinic visits declined for treatment patients and increased slightly for matched control patients (Figure 2A and 2B). For occupational therapy visits, neither group showed changes over time (main effect of time IRR, 1.03; 99% CI, 0.83 to 1.22; Time × Treatment interaction, 1.20; 99% CI, 0.93 to 1.46) (Figure 2C). For physical medicine and rehabilitation visits, both groups decreased use over time (main effect of time IRR, 0.78; 99% CI, 0.66 to 0.90), but this decrease did not differ between treatment and control patients (Time × Treatment interaction, 1.16; 99% CI, 0.94 to 1.37) (Figure 2D).

Pain Intensity
Pain intensity decreased for both groups by an estimated 0.02 points per month (99% CI, -0.04 to -0.01; P < .01), or 0.24 points over the 12-month postindex follow-up period (Figure 3). There were no statistically significant differences between treatment and control patients in pain intensity changes over the follow-up period (B = -0.02; 99% CI, -0.06 to 0.02; P = .15).

Discussion
Patients with musculoskeletal pain who received a thermal therapy device showed a larger decline in use of some specialty pain treatments, including physical therapy and specialty pain clinic services, when compared with matched control patients. One possible explanation is that patients who used the device may have had their pain adequately managed and thus required less specialty pain care. However, the absence of clinically significant changes in pain intensity over time suggests that pain intensity alone did not account for the observed changes in pain treatment use. We were unable to measure constructs of pain interference or functioning, which may be better predictors of functional restoration, as these data are not routinely collected within the VA and documented in the EHR. Future studies could clarify these findings by including measures of pain interference and functioning.
Although the overall declines in physical therapy and specialty pain clinic use associated with thermal therapy were modest (< 1 visit per patient), the impact of these reductions can be profound. In resource-limited health care settings, even small reductions in high-cost care utilization could be of great value in that health systems could offset costs associated with treating chronic pain without compromising quality of care or key clinical outcomes, such as pain intensity. This study, however, did not include a cost analysis. Future studies should incorporate formal cost analyses to quantify cost offsets that may result from decreased specialty pain treatment use.
Patients who received thermal therapy devices did not show clinically meaningful reductions in pain intensity over time, defined as reductions in pain intensity of 1.7 to 2.0 on a 0 to 10 scale.28,29 This finding is consistent with prior research that demonstrates relatively stable pain intensity self-ratings longitudinally by patients with chronic pain diagnoses, when assessed in the context of usual clinical care.30 This finding, however, is inconsistent with prior literature that demonstrates pain-reducing benefits of thermal therapy for low back pain and knee osteoarthritis.7-9
In this study, pain intensity ratings were derived from the EHR during routine outpatient clinical encounters and not at the time thermal therapy was self-administered, as has been done in prior clinical trials.7-9 Pain location was not specified at the time of pain ratings, and it is possible that patients may have been endorsing pain in areas of the body that had not been treated by thermal therapy. Patient-level variability in pain intensity ratings (eg, within-patient range over time) was not examined, although prior research indicates substantial variability.30 While average pain intensity ratings in the current study did not change, an examination of patient variability warrants further study, as a narrowing of pain intensity ratings can be perceived, by patients, as demonstrable improvement and has been associated with improved physical and psychological outcomes.31 Furthermore, pain intensity does not characterize physical or emotional functioning that can be captured with more comprehensive validated measures, some of which are recommended outcomes in pain clinical trials.32
Our findings point to reductions in all forms of opioid use across both treatment and control patients. Data from the VA and in the US more generally point to downward trends in opioid prescribing during the study period.33 This decline is likely due to increased use of risk mitigation approaches, such as routine urine drug screens and review of prescription drug monitoring databases.34 These state-level databases track prescribing of controlled substances, including opioids and benzodiazepines, within a state. Implementation of these practices has been associated with declines in higher risk opioid prescribing.34 Findings from this study further point to associations of reduced higher risk opioid use among patients who received thermal therapy devices. In the full sample of patients, reductions in days of opioid use, high-dose opioid use, and co-use of opioids and benzodiazepines were observed across all patients, with greater reductions observed in high-dose opioid use and concurrent opioid and benzodiazepine use among patients who received a thermal therapy device. Experimental studies indicate that the endogenous opioid system is implicated in pain relief and activated by thermal therapies. 35 Differential reductions in higher-risk opioid use among patients who received thermal therapy devices in our study may be associated with endogenous opioid activation, though this was not specifically measured. It is also unclear whether thermal therapy was provided by clinicians in the context of opioid tapering or other risk mitigation efforts, or patients reduced higher risk opioid use of their own volition. Prior research has identified both patient- and clinician-initiated opioid tapering and discontinuation.36 While a thorough explication of opioid dose reduction was beyond the scope of this study, future qualitative work could help explain potential benefits of thermal therapy in the context of analgesic pharmacotherapy use, including opioid medications.
Limitations
The extent to which patients used the thermal therapy device could not be measured; therefore, device receipt was used as a proxy for use. However, it was not possible to determine whether the frequency and duration of device use was associated with study outcomes. Treatment and control groups demonstrated some differences in outcome variables at the index date. Potential known biases were addressed using propensity score matching procedures and statistical procedures that controlled for patient demographic and clinical characteristics, as well as pretreatment values of all outcome variables. Nevertheless, as an observational study, this analysis cannot account for all known and unknown confounders, and a randomized controlled trial is needed to make claims of causality. The study population consisted of US veterans and included a low proportion of women. As a result, the findings may not be generalizable to other patient populations. Finally, prescription dispensing data, used as a proxy for pharmacotherapy use, do not necessarily reflect actual medication use.
Conclusions
This study is among the first to examine associations between thermal therapy and specialty pain treatment and analgesic pharmacotherapy use among US veterans. Although the retrospective cohort study design does not allow causal inferences regarding the efficacy of thermal therapy for veterans with chronic musculoskeletal pain, confidence in the findings is strengthened by methodological and statistical control of known confounders. Future trials employing experimental designs are needed to further clarify the clinical and health systems benefits of thermal therapy for musculoskeletal pain syndromes.
Growing recognition of the limitations of long-term opioid therapy for chronic noncancer pain has highlighted the importance of nonpharmacologic approaches to pain treatment.1,2 These treatments are varied and may include psychological and behavioral therapies (eg, cognitive behavioral therapy for chronic pain), exercise and movement therapies (eg, yoga), and manual therapies (eg, chiropractic). Body surface cold therapy, while predominantly used to reduce postoperative pain and inflammation,3,4 pain subsequent to acute musculoskeletal injury,5 and pain related to muscle soreness,6 is a nonpharmacologic treatment that has shown pain-reducing benefits for chronic low back pain and knee osteoarthritis, as has its counterpart, superficial heat therapy.7-9 Heat therapy has also been shown to improve strength, flexibility, and activities of daily living in patients with chronic low back pain.10,11 Cold and heat therapies are commonly used complementarily. Cold therapies aim to reduce blood flow and inflammation and are often used immediately following trauma to an affected area, whereas heat therapies increase blood flow and metabolic activity and are commonly used to promote healing.5
Heat and cold therapies (also known as thermal therapies) benefit resource-limited health care systems, as most devices require a single expenditure and can be self-administered by patients at home as part of their pain self-management plan. In addition, these pain self-management tools may attenuate the need for more expensive specialty pain care and ongoing analgesic pharmacotherapy. Despite their potential, few studies have characterized the benefits of thermal therapies for patients with heterogeneous chronic pain syndromes.
The purpose of this retrospective cohort study was to examine the potential clinical and health system benefits of patient-administered thermal therapy. Our primary hypothesis was that patients with chronic musculoskeletal pain who received a thermal therapy self-management device would have fewer days of opioid prescriptions compared with a sample of matched control patients. Secondarily, we hypothesized that patients who received a thermal therapy device would have lower utilization of specialty pain care, fewer potentially hazardous opioid prescriptions (eg, high-dose opioid therapy and concurrent opioid and benzodiazepine prescriptions), fewer prescriptions for nonopioid analgesic medications, and decreased pain intensity when compared with matched controls.
Methods
This retrospective cohort study compared pain pharmacotherapy, pain treatment utilization, and pain intensity outcomes between patients who received a thermal therapy device and matched patients who did not. The study was approved by the US Department of Veterans Affairs (VA) Portland Health Care System Institutional Review Board and was granted a waiver of informed consent to access patient electronic health records (EHRs).
Pain Care
The VA uses ThermaZone thermal therapy devices (Innovative Medical Equipment) for chronic pain treatment. The device uses thermoelectric technology to provide point-of-contact cooling and heating therapy through site-specific pads (eg, ankle, knee, hip, back, elbow, shoulder). Patients place pads on sites where they experience pain, and temperature regulated water circulates through the device and to the pad, providing consistent, localized thermal therapy. The pads range in temperature from 1 °C to 52 °C, and temperatures are self-monitored and controlled by the patient.
Standard pain care in this study followed the VA stepped model of pain care, which builds on a foundation of patient education for pain self-management approaches (eg, exercise, mindfulness, relaxation, social support).12 According to the VA stepped model of pain care, all patients with chronic pain should engage in these foundational self-management approaches. However, some patients require more intensive care. The VA stepped-up treatment engages primary and specialty care services—such as physical therapy, pharmacy, complementary and integrative health approaches, mental health, and substance use services—and, when indicated, may escalate treatment to interdisciplinary pain teams or tertiary pain centers. In this retrospective cohort study, treatment patients received a thermal therapy device and standard of care, whereas control patients received standard care only.
Sample Selection
Eligible patients were aged ≥ 18 years, had a musculoskeletal pain diagnosis documented in the EHR in the year prior to thermal therapy device receipt (or during the same period for a treatment patient’s matched control), and were enrolled in VA health care during the study period. Patients who died during the study period were excluded. Treatment patients received a thermal therapy device from the VA between January 1, 2017, and December 31, 2018, when complete data on thermal therapy devices were available. For control patients, the VA Corporate Data Warehouse (CDW) was used to identify VA patients with characteristics similar to those of treated patients.
We modeled the probability that a patient would receive a thermal therapy device using logistic regression. Predictor variables were measured in the year prior to device receipt and included variables associated with pain treatment utilization and analgesic pharmacotherapy receipt, as recommended by Brookhart et al.13 These included age, sex, race, ethnicity, VA service-connected disability status, comorbidities, receipt of medications for opioid use disorder, pain diagnoses, mental health diagnoses, and substance use disorder diagnoses.14-19
The resulting propensity scores (eg, predicted probabilities) were used to match treatment patients 1:1 with control patients using a nearest-neighbor matching algorithm.20 This procedure matches a control patient with the closest propensity score to that of the corresponding treatment patient. An exact match on VA facility was required (eg, treatment patients and matched controls received care at the same VA facility). Standardized differences were used to assess covariate balance between the matched groups, and kernel density plots of propensity scores tested for sufficient overlap.21 Control patients were selected from a pool of 1,150,149 patients.
Study Variables
The index date was the date the thermal therapy device was released to treatment patients or the same date for the matched controls. Data were extracted from the CDW over a 24-month period: 12 months prior to the index date through 12 months afterwards. Collecting data in the 12 months prior to treatment initiation allowed us to adjust for covariates and provided greater precision, as recommended for observational study designs.22
Treatment conditions were defined dichotomously as receipt vs nonreceipt of a thermal therapy device. The primary outcome was the number of days of opioid use in the 12 months following the index date. Additional outcomes included days of high-dose opioid therapy (≥ 50 mg morphine equivalent [MME] daily), concurrent opioid and benzodiazepine prescriptions, and nonopioid analgesic pharmacotherapy (eg, nonsteroidal anti-inflammatory drugs, acetaminophen, muscle relaxants). All prescription data were based on medication fills obtained from VA pharmacy records. Additional outcomes included the number of visits to physical therapy, occupational therapy, physical medicine and rehabilitation, and interdisciplinary pain clinics, including interventional pain medicine.
Pain intensity ratings were collected as part of routine VA care using a numeric scale from 0 (no pain) to 10 (worst possible pain). Pain intensity trajectories were computed using all available EHR-derived pain intensity score data for each patient in the 12 months prior to and following the index date.
Covariates were extracted from the EHR and evaluated in the year prior to the index date, unless otherwise noted. They included age at the index date; self-reported sex, and race and ethnicity; service-connected disability status (disability awarded as a result of military service-related trauma or injury); Charlson Comorbidity Index; and diagnoses of opioid use disorder, alcohol use disorder, other substance use disorder, mood disorder, posttraumatic stress disorder, other anxiety disorder, psychotic disorder, neuropathic pain, and headache pain.23 All diagnoses were coded as yes if identified in the EHR as a focus of treatment during any clinical encounter in the year prior to the index date or no if not identified as a focus of treatment.
The number of days patients had been living with pain was calculated and defined as the number of days from the first pain diagnosis available in a patient’s EHR to the index date. Finally, the number of pain-related surgeries (eg, surgeries with ≥ 1 pain diagnoses associated with the clinical encounter) and average pain intensity were computed for the pre- and postindex date evaluation periods and included as model covariates.
Statistical Analyses
For the 4 pharmacotherapy and 4 nonpharmacologic treatment usage outcomes, we tested the fit of linear models and several models with count distributions using the Bayesian information criterion.24 Count distributions included Poisson, zero-inflated Poisson, negative binomial, and zero-inflated negative binomial. With 1 exception (days of high-dose opioid use), a negative binomial distribution best fit the data. For days of high-dose opioid use, a Gaussian distribution best fit the data.
Eight separate mixed-effects regression analyses compared changes in each outcome from the 12-month preindex period through the 12-month postindex period between treatment and control patients by testing the Time × Treatment interaction. This approach statistically accounts for observed pretreatment differences in outcome variables. Statistics for the main effects of time and treatment are also presented. To reduce bias, models controlled for covariates specified previously.
For pain intensity, we used random-effects growth modeling to quantify both fixed and random effects of pain intensity at the index date (eg, the model intercept), which estimates pain at the time of treatment initiation, and change in pain during the 12 months following the index date (eg, the model slope), which characterizes the trajectory of pain intensity ratings.25 The model included piecewise components of pain score trajectories in the 12 months prior to the index date and in the 12 months following the index date. Several types of change for the 12-month postindex observation period were explored—including quadratic and cubic curvilinear change. A linear model for change in pain over time provided the best fit based on the Bayesian information criterion and parsimony of model parameters.26 We report estimates of change in pain over time in monthly intervals for ease of interpretation. However, models used all individual pain scores rather than computing monthly averages when > 1 pain score was available within a month, as recommended in previous research.27 This approach makes optimal use of all available data. Both random effects (intercept and slope) were regressed onto the set of covariates described previously.
This study used data available in the EHR over the 24-month observation period. We characterize the density (eg, frequency) of all outcome variables by treatment condition in the Appendix. Because the hypotheses were directional, the authors used a 2-sided α = 0.10 and applied a Bonferroni correction for the 9 statistical tests performed, resulting in an adjusted α of 0.01. Treatment utilization and pharmacotherapy outcome analyses were performed in Stata, version 16.1. Random-effects growth modeling of pain score trajectories was performed using Mplus, version 8.8.

Results
There were 2182 patients in the treatment group and 2182 matched controls. The mean (SD) age was 54 (15) years; 81% were male, and about two-thirds (68%) identified as White and non-Hispanic. Mental health comorbidities were common, with > 40% of the sample having diagnoses of a mood disorder and/or posttraumatic stress disorder. Nearly all patients (90%) had VA service-connected disability ratings. Among patients with chronic musculoskeletal pain, 8% had comorbid neuropathic pain and 14% had headache. The mean (SD) duration of chronic pain across both patient groups was 3416 (2016) days, or about 9.4 years (Table 1).

Pharmacotherapy
High-dose opioid use (> 50 MME daily dose), days of opioid use, and concurrent opioid and benzodiazepine use decreased for all patients from the pre- to posttreatment period (Table 2). However, high-dose opioid use (Time × Treatment interaction, 3.24; 99% CI, 0.34 to 6.14) (Figure 1A) and concurrent opioid and benzodiazepine use (Time × Treatment interaction, 0.76; 99% CI, 0.67 to 0.86) (Figure 1B) had a larger decrease for the treatment vs matched control group. Treatment and matched control patients had comparable reductions in days of opioid use (Time × Treatment interaction, 0.98; 99% CI, 0.91 to 1.05) (Figure 1C). Neither group showed changes in nonopioid analgesic pharmacotherapy over time (main effect of time, incidence rate ratio [IRR], 1.03; 99% CI, 0.99 to 1.07; Time × Treatment interaction, 0.95; 99% CI, 0.90 to 1.01) (Figure 1D).


Nonpharmacologic Pain Treatment
The number of physical therapy and pain clinic visits declined for treatment patients and increased slightly for matched control patients (Figure 2A and 2B). For occupational therapy visits, neither group showed changes over time (main effect of time IRR, 1.03; 99% CI, 0.83 to 1.22; Time × Treatment interaction, 1.20; 99% CI, 0.93 to 1.46) (Figure 2C). For physical medicine and rehabilitation visits, both groups decreased use over time (main effect of time IRR, 0.78; 99% CI, 0.66 to 0.90), but this decrease did not differ between treatment and control patients (Time × Treatment interaction, 1.16; 99% CI, 0.94 to 1.37) (Figure 2D).

Pain Intensity
Pain intensity decreased for both groups by an estimated 0.02 points per month (99% CI, -0.04 to -0.01; P < .01), or 0.24 points over the 12-month postindex follow-up period (Figure 3). There were no statistically significant differences between treatment and control patients in pain intensity changes over the follow-up period (B = -0.02; 99% CI, -0.06 to 0.02; P = .15).

Discussion
Patients with musculoskeletal pain who received a thermal therapy device showed a larger decline in use of some specialty pain treatments, including physical therapy and specialty pain clinic services, when compared with matched control patients. One possible explanation is that patients who used the device may have had their pain adequately managed and thus required less specialty pain care. However, the absence of clinically significant changes in pain intensity over time suggests that pain intensity alone did not account for the observed changes in pain treatment use. We were unable to measure constructs of pain interference or functioning, which may be better predictors of functional restoration, as these data are not routinely collected within the VA and documented in the EHR. Future studies could clarify these findings by including measures of pain interference and functioning.
Although the overall declines in physical therapy and specialty pain clinic use associated with thermal therapy were modest (< 1 visit per patient), the impact of these reductions can be profound. In resource-limited health care settings, even small reductions in high-cost care utilization could be of great value in that health systems could offset costs associated with treating chronic pain without compromising quality of care or key clinical outcomes, such as pain intensity. This study, however, did not include a cost analysis. Future studies should incorporate formal cost analyses to quantify cost offsets that may result from decreased specialty pain treatment use.
Patients who received thermal therapy devices did not show clinically meaningful reductions in pain intensity over time, defined as reductions in pain intensity of 1.7 to 2.0 on a 0 to 10 scale.28,29 This finding is consistent with prior research that demonstrates relatively stable pain intensity self-ratings longitudinally by patients with chronic pain diagnoses, when assessed in the context of usual clinical care.30 This finding, however, is inconsistent with prior literature that demonstrates pain-reducing benefits of thermal therapy for low back pain and knee osteoarthritis.7-9
In this study, pain intensity ratings were derived from the EHR during routine outpatient clinical encounters and not at the time thermal therapy was self-administered, as has been done in prior clinical trials.7-9 Pain location was not specified at the time of pain ratings, and it is possible that patients may have been endorsing pain in areas of the body that had not been treated by thermal therapy. Patient-level variability in pain intensity ratings (eg, within-patient range over time) was not examined, although prior research indicates substantial variability.30 While average pain intensity ratings in the current study did not change, an examination of patient variability warrants further study, as a narrowing of pain intensity ratings can be perceived, by patients, as demonstrable improvement and has been associated with improved physical and psychological outcomes.31 Furthermore, pain intensity does not characterize physical or emotional functioning that can be captured with more comprehensive validated measures, some of which are recommended outcomes in pain clinical trials.32
Our findings point to reductions in all forms of opioid use across both treatment and control patients. Data from the VA and in the US more generally point to downward trends in opioid prescribing during the study period.33 This decline is likely due to increased use of risk mitigation approaches, such as routine urine drug screens and review of prescription drug monitoring databases.34 These state-level databases track prescribing of controlled substances, including opioids and benzodiazepines, within a state. Implementation of these practices has been associated with declines in higher risk opioid prescribing.34 Findings from this study further point to associations of reduced higher risk opioid use among patients who received thermal therapy devices. In the full sample of patients, reductions in days of opioid use, high-dose opioid use, and co-use of opioids and benzodiazepines were observed across all patients, with greater reductions observed in high-dose opioid use and concurrent opioid and benzodiazepine use among patients who received a thermal therapy device. Experimental studies indicate that the endogenous opioid system is implicated in pain relief and activated by thermal therapies. 35 Differential reductions in higher-risk opioid use among patients who received thermal therapy devices in our study may be associated with endogenous opioid activation, though this was not specifically measured. It is also unclear whether thermal therapy was provided by clinicians in the context of opioid tapering or other risk mitigation efforts, or patients reduced higher risk opioid use of their own volition. Prior research has identified both patient- and clinician-initiated opioid tapering and discontinuation.36 While a thorough explication of opioid dose reduction was beyond the scope of this study, future qualitative work could help explain potential benefits of thermal therapy in the context of analgesic pharmacotherapy use, including opioid medications.
Limitations
The extent to which patients used the thermal therapy device could not be measured; therefore, device receipt was used as a proxy for use. However, it was not possible to determine whether the frequency and duration of device use was associated with study outcomes. Treatment and control groups demonstrated some differences in outcome variables at the index date. Potential known biases were addressed using propensity score matching procedures and statistical procedures that controlled for patient demographic and clinical characteristics, as well as pretreatment values of all outcome variables. Nevertheless, as an observational study, this analysis cannot account for all known and unknown confounders, and a randomized controlled trial is needed to make claims of causality. The study population consisted of US veterans and included a low proportion of women. As a result, the findings may not be generalizable to other patient populations. Finally, prescription dispensing data, used as a proxy for pharmacotherapy use, do not necessarily reflect actual medication use.
Conclusions
This study is among the first to examine associations between thermal therapy and specialty pain treatment and analgesic pharmacotherapy use among US veterans. Although the retrospective cohort study design does not allow causal inferences regarding the efficacy of thermal therapy for veterans with chronic musculoskeletal pain, confidence in the findings is strengthened by methodological and statistical control of known confounders. Future trials employing experimental designs are needed to further clarify the clinical and health systems benefits of thermal therapy for musculoskeletal pain syndromes.
- Krebs EE, Gravely A, Nugent S, et al. Effect of opioid vs nonopioid medications on pain-related function in patients with chronic back pain or hip or knee osteoarthritis pain: the SPACE randomized clinical trial. JAMA. 2018;2018:872-882. doi:10.1001/jama.2018.0899
- Becker WC, DeBar LL, Heapy AA, et al. A research agenda for advancing non-pharmacological management of chronic musculoskeletal pain: findings from a VHA state-of-the-art conference. J Gen Intern Med. 2018;33:1-15. doi:10.1007/s11606-018-4345-6
- Fernandes IA, Armond ACV, Falci SGM. The effectiveness of the cold therapy (cryotherapy) in the management of inflammatory parameters after removal of mandibular third molars: a meta-analysis. Int Arch Otorhinolaryngol. 2019;23:221-228. doi:10.1055/s-0039-1677755
- Quinlan P, Davis J, Fields K, et al. Effects of localized cold therapy on pain in postoperative spinal fusion patients: a randomized controlled trial. Orthop Nurs. 2017;36:344-349. doi:10.1097/NOR.0000000000000382
- Malanga GA, Yan N, Stark J. Mechanisms and efficacy of heat and cold therapies for musculoskeletal injury. Postgrad Med. 2015;127:57-65. doi:10.1080/00325481.2015.992719
- Wang Y, Li S, Zhang Y, et al. Heat and cold therapy reduce pain in patients with delayed onset muscle soreness: a systematic review and meta-analysis of 32 randomized controlled trials. Phys Ther Sport. 2021;48:177-187. doi:10.1016/j.ptsp.2021.01.004
- Ariana M, Afrasiabifar A, Doulatabad SN, et al. The effect of local heat therapy versus cold rub gel on pain and joint functions in patients with knee osteoarthritis. Clin Nurs Res. 2022;31:1014-1022. doi:10.1177/10547738211035502
- French SD, Cameron M, Walker BF, et al. Superficial heat or cold for low back pain. Cochrane Database Syst Rev. 2006;1:CD004750. doi:10.1002/14651858.CD004750.pub2
- Tao XG, Bernacki EJ. A randomized clinical trial of continuous low-level heat therapy for acute muscular low back pain in the workplace. J Occup Environ Med. 2005;47:1298- 1306. doi:10.1097/01.jom.0000184877.01691.a3
- Freiwald J, Hoppe MW, Beermann W, et al. Effects of supplemental heat therapy in multimodal treated chronic low back pain patients on strength and flexibility. Clin Biomech. 2018;57:107-113. doi:10.1016/j.clinbiomech.2018.06.008
- Freiwald J, Magni A, Fanlo-Mazas P, et al. A role for superficial heat therapy in the management of nonspecific, mild-to-moderate low back pain in current clinical practice: a narrative review. Life. 2021;11:780. doi:10.3390/life11080780
- Kerns RD, Philip EJ, Lee AW, et al. Implementation of the Veterans Health Administration National Pain Management Strategy. Transl Behav Med. 2011;1:635-643. doi:10.1007/s13142-011-0094-3
- Brookhart MA, Schneeweiss S, Rothman KJ, et al. Variable selection for propensity score models. Am J Epidemiol. 2006;163:1149-1156. doi:10.1093/aje/kwj149
- Edlund MJ, Martin BC, Devries A, et al. Trends in use of opioids for chronic non-cancer pain among individuals with mental health and substance use disorders: the TROUP study. Clin J Pain. 2010;26:1-8. doi:10.1097/AJP.0b013e3181b99f35
- Kaur S, Stechuchak KM, Coffman CJ, et al. Gender differences in health care utilization among veterans with chronic pain. J Gen Intern Med. 2007;22:228-233. doi:10.1007/s11606-006-0048-5
- Meghani SH, Cho E. Self-reported pain and utilization of pain treatment between minorities and non-minorities in the United States. Public Health Nurs. 2009;26:307-316. doi:10.1111/j.1525-1446.2009.00785.x
- Morasco BJ, Duckart JP, Carr TP, et al. Clinical characteristics of veterans prescribed high doses of opioid medications for chronic non-cancer pain. Pain. 2010;151:625-632. doi:10.1016/j.pain.2010.08.002
- Nielsen S, Lintzeris N, Bruno R, et al. Benzodiazepine use among chronic pain patients prescribed opioids: associations with pain, physical and mental health, and health service utilization. Pain Med. 2015;16:356-366. doi:10.1111/pme.12594
- Quinn PD, Hur K, Chang Z, et al. Incident and long-term opioid therapy among patients with psychiatric conditions and medications: a national study of commercial health care claims. Pain. 2017;158:140-148. doi:10.1097/j.pain.0000000000000730
- Rubin DB. Matching to remove bias in observational studies. Biometrics. 1973;29:159-183.
- Rosenbaum PR, Rubin DB. Constructing a control group using multivariate matched sampling methods that incorporate the propensity score. Am Stat. 1985;39:33-38. doi:10.1007/s11596-025-00052-0
- Steiner PM, Cook TD, Shadish WR, et al. The importance of covariate selection in controlling for selection bias in observational studies. Psychol Methods. 2010;15:250-267. doi:10.1037/a0018719
- Charlson ME, Pompei P, Ales KL, et al. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40:373- 383. doi:10.1016/0021-9681(87)90171-8
- Long JS, Freese J. Regression Models for Categorical Dependent Variables Using Stata. 3rd ed. Stata Press; 2014.
- Muthén LK, Muthén BO. Mplus User’s Guide. 8th ed. Muthén & Muthén; 1998-2017.
- Hedeker D, Gibbons RD. Longitudinal Data Analysis. Wiley; 2006.
- Dobscha SK, Morasco BJ, Kovas AE, et al. Short-term variability in outpatient pain intensity scores in a national sample of older veterans with chronic pain. Pain Med. 2015;16:855-865. doi:10.1111/pme.12643
- Bahreini M, Safaie A, Mirfazaelian H, et al. How much change in pain score does really matter to patients? Am J Emerg Med. 2020;38:1641-1646. doi:10.1016/j.ajem.2019.158489
- Suzuki H, Aono S, Inoue S, et al. Clinically significant changes in pain along the pain intensity numerical rating scale in patients with chronic low back pain. PLoS One. 2020;15:e0229228. doi:10.1371/journal.pone.0229228
- McPherson SM, Smith CL, Dobscha SK, et al. Changes in pain intensity after discontinuation of long-term opioid therapy for chronic noncancer pain. Pain. 2018;159:2097- 2104. doi:10.1097/j.pain.0000000000001315
- Andrews NE, Strong J, Meredith PJ. Activity pacing, avoidance, endurance, and associations with patient functioning in chronic pain: a systematic and meta-analysis. Arch Phys Med Rehabil. 2012;93:2109-121. doi:10.1016/j.apmr.2012.05.029
- Dworkin RH, Turk DC, Farrar JT, et al. Core outcome measures for chronic pain clinical trials: IMMPACT recommendations. Pain. 2005;113:9-19. doi:10.1016/j.pain.2004.09.012
- Gellad WF, Good CB, Shulkin DJ. Addressing the opioid epidemic in the United States: Lessons from the Department of Veterans Affairs. JAMA Intern Med. 2017;177:611- 612. doi:10.1001/jamainternmed.2017.0147
- Lin LA, Bohnert ASB, Kerns RD, et al. Impact of the Opioid Safety Initiative on opioid-related prescribing in veterans. Pain. 2017;158:833-839. doi:10.1097/j.pain.0000000000000837
- Sirucek L, Price RC, Gandhi W, et al. Endogenous opioids contribute to the feeling of pain relief in humans. Pain. 2021;162:2821-2831. doi:10.1097/j.pain.0000000000002285
- Lovejoy TI, Morasco BJ, Demidenko MI, et al. Reasons for discontinuation of long-term opioid therapy in patients with and without substance use disorders. Pain. 2017;158:526- 534. doi:10.1097/j.pain.0000000000000796
- Krebs EE, Gravely A, Nugent S, et al. Effect of opioid vs nonopioid medications on pain-related function in patients with chronic back pain or hip or knee osteoarthritis pain: the SPACE randomized clinical trial. JAMA. 2018;2018:872-882. doi:10.1001/jama.2018.0899
- Becker WC, DeBar LL, Heapy AA, et al. A research agenda for advancing non-pharmacological management of chronic musculoskeletal pain: findings from a VHA state-of-the-art conference. J Gen Intern Med. 2018;33:1-15. doi:10.1007/s11606-018-4345-6
- Fernandes IA, Armond ACV, Falci SGM. The effectiveness of the cold therapy (cryotherapy) in the management of inflammatory parameters after removal of mandibular third molars: a meta-analysis. Int Arch Otorhinolaryngol. 2019;23:221-228. doi:10.1055/s-0039-1677755
- Quinlan P, Davis J, Fields K, et al. Effects of localized cold therapy on pain in postoperative spinal fusion patients: a randomized controlled trial. Orthop Nurs. 2017;36:344-349. doi:10.1097/NOR.0000000000000382
- Malanga GA, Yan N, Stark J. Mechanisms and efficacy of heat and cold therapies for musculoskeletal injury. Postgrad Med. 2015;127:57-65. doi:10.1080/00325481.2015.992719
- Wang Y, Li S, Zhang Y, et al. Heat and cold therapy reduce pain in patients with delayed onset muscle soreness: a systematic review and meta-analysis of 32 randomized controlled trials. Phys Ther Sport. 2021;48:177-187. doi:10.1016/j.ptsp.2021.01.004
- Ariana M, Afrasiabifar A, Doulatabad SN, et al. The effect of local heat therapy versus cold rub gel on pain and joint functions in patients with knee osteoarthritis. Clin Nurs Res. 2022;31:1014-1022. doi:10.1177/10547738211035502
- French SD, Cameron M, Walker BF, et al. Superficial heat or cold for low back pain. Cochrane Database Syst Rev. 2006;1:CD004750. doi:10.1002/14651858.CD004750.pub2
- Tao XG, Bernacki EJ. A randomized clinical trial of continuous low-level heat therapy for acute muscular low back pain in the workplace. J Occup Environ Med. 2005;47:1298- 1306. doi:10.1097/01.jom.0000184877.01691.a3
- Freiwald J, Hoppe MW, Beermann W, et al. Effects of supplemental heat therapy in multimodal treated chronic low back pain patients on strength and flexibility. Clin Biomech. 2018;57:107-113. doi:10.1016/j.clinbiomech.2018.06.008
- Freiwald J, Magni A, Fanlo-Mazas P, et al. A role for superficial heat therapy in the management of nonspecific, mild-to-moderate low back pain in current clinical practice: a narrative review. Life. 2021;11:780. doi:10.3390/life11080780
- Kerns RD, Philip EJ, Lee AW, et al. Implementation of the Veterans Health Administration National Pain Management Strategy. Transl Behav Med. 2011;1:635-643. doi:10.1007/s13142-011-0094-3
- Brookhart MA, Schneeweiss S, Rothman KJ, et al. Variable selection for propensity score models. Am J Epidemiol. 2006;163:1149-1156. doi:10.1093/aje/kwj149
- Edlund MJ, Martin BC, Devries A, et al. Trends in use of opioids for chronic non-cancer pain among individuals with mental health and substance use disorders: the TROUP study. Clin J Pain. 2010;26:1-8. doi:10.1097/AJP.0b013e3181b99f35
- Kaur S, Stechuchak KM, Coffman CJ, et al. Gender differences in health care utilization among veterans with chronic pain. J Gen Intern Med. 2007;22:228-233. doi:10.1007/s11606-006-0048-5
- Meghani SH, Cho E. Self-reported pain and utilization of pain treatment between minorities and non-minorities in the United States. Public Health Nurs. 2009;26:307-316. doi:10.1111/j.1525-1446.2009.00785.x
- Morasco BJ, Duckart JP, Carr TP, et al. Clinical characteristics of veterans prescribed high doses of opioid medications for chronic non-cancer pain. Pain. 2010;151:625-632. doi:10.1016/j.pain.2010.08.002
- Nielsen S, Lintzeris N, Bruno R, et al. Benzodiazepine use among chronic pain patients prescribed opioids: associations with pain, physical and mental health, and health service utilization. Pain Med. 2015;16:356-366. doi:10.1111/pme.12594
- Quinn PD, Hur K, Chang Z, et al. Incident and long-term opioid therapy among patients with psychiatric conditions and medications: a national study of commercial health care claims. Pain. 2017;158:140-148. doi:10.1097/j.pain.0000000000000730
- Rubin DB. Matching to remove bias in observational studies. Biometrics. 1973;29:159-183.
- Rosenbaum PR, Rubin DB. Constructing a control group using multivariate matched sampling methods that incorporate the propensity score. Am Stat. 1985;39:33-38. doi:10.1007/s11596-025-00052-0
- Steiner PM, Cook TD, Shadish WR, et al. The importance of covariate selection in controlling for selection bias in observational studies. Psychol Methods. 2010;15:250-267. doi:10.1037/a0018719
- Charlson ME, Pompei P, Ales KL, et al. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40:373- 383. doi:10.1016/0021-9681(87)90171-8
- Long JS, Freese J. Regression Models for Categorical Dependent Variables Using Stata. 3rd ed. Stata Press; 2014.
- Muthén LK, Muthén BO. Mplus User’s Guide. 8th ed. Muthén & Muthén; 1998-2017.
- Hedeker D, Gibbons RD. Longitudinal Data Analysis. Wiley; 2006.
- Dobscha SK, Morasco BJ, Kovas AE, et al. Short-term variability in outpatient pain intensity scores in a national sample of older veterans with chronic pain. Pain Med. 2015;16:855-865. doi:10.1111/pme.12643
- Bahreini M, Safaie A, Mirfazaelian H, et al. How much change in pain score does really matter to patients? Am J Emerg Med. 2020;38:1641-1646. doi:10.1016/j.ajem.2019.158489
- Suzuki H, Aono S, Inoue S, et al. Clinically significant changes in pain along the pain intensity numerical rating scale in patients with chronic low back pain. PLoS One. 2020;15:e0229228. doi:10.1371/journal.pone.0229228
- McPherson SM, Smith CL, Dobscha SK, et al. Changes in pain intensity after discontinuation of long-term opioid therapy for chronic noncancer pain. Pain. 2018;159:2097- 2104. doi:10.1097/j.pain.0000000000001315
- Andrews NE, Strong J, Meredith PJ. Activity pacing, avoidance, endurance, and associations with patient functioning in chronic pain: a systematic and meta-analysis. Arch Phys Med Rehabil. 2012;93:2109-121. doi:10.1016/j.apmr.2012.05.029
- Dworkin RH, Turk DC, Farrar JT, et al. Core outcome measures for chronic pain clinical trials: IMMPACT recommendations. Pain. 2005;113:9-19. doi:10.1016/j.pain.2004.09.012
- Gellad WF, Good CB, Shulkin DJ. Addressing the opioid epidemic in the United States: Lessons from the Department of Veterans Affairs. JAMA Intern Med. 2017;177:611- 612. doi:10.1001/jamainternmed.2017.0147
- Lin LA, Bohnert ASB, Kerns RD, et al. Impact of the Opioid Safety Initiative on opioid-related prescribing in veterans. Pain. 2017;158:833-839. doi:10.1097/j.pain.0000000000000837
- Sirucek L, Price RC, Gandhi W, et al. Endogenous opioids contribute to the feeling of pain relief in humans. Pain. 2021;162:2821-2831. doi:10.1097/j.pain.0000000000002285
- Lovejoy TI, Morasco BJ, Demidenko MI, et al. Reasons for discontinuation of long-term opioid therapy in patients with and without substance use disorders. Pain. 2017;158:526- 534. doi:10.1097/j.pain.0000000000000796
Thermal Therapy is Associated With Decreased Pain Treatment Use: A Retrospective Cohort Study
Thermal Therapy is Associated With Decreased Pain Treatment Use: A Retrospective Cohort Study
Implementation of a Pharmacist-Led Penicillin Allergy Interview at a Veterans Care Facility
Implementation of a Pharmacist-Led Penicillin Allergy Interview at a Veterans Care Facility
Self-reported penicillin allergies are common, with a prevalence of about 10% of patients, according to the Centers for Disease Control and Prevention (CDC).1 However, only about 1% of patients have a true immunoglobulin E (IgE)-mediated allergy. This issue is often further complicated by inaccurate classification of nonallergic adverse effects as an allergy, resulting in incomplete allergy documentation in the electronic health record (EHR). The cross-reactivity rate with cephalosporins (Β-lactam antibiotics) in patients reporting a penicillin allergy is < 1%, which suggests that many patients with reported penicillin allergies can safely receive them.2 Despite this, patients with self-reported penicillin allergies often receive non–Β-lactam antibiotic agents, which may be associated with an increased risk of adverse drug reactions (ADRs), increased health care costs, and inferior clinical outcomes.3
Several strategies are recommended to assess patients with self-reported penicillin allergies. According to the CDC, evaluating a patient who reports a penicillin or other Β-lactam antibiotic allergy involves 3 steps: (1) obtaining a thorough medical history, including previous exposures to penicillin or other Β-lactam antibiotic; (2) performing a skin test using the penicillin major and minor determinants; and (3) among those who have a negative penicillin skin test, performing an observed oral challenge with 250 mg amoxicillin before proceeding directly to treatment with the indicated Β-lactam therapy.4
Most existing clinical guidance for assessing patients with self-reported penicillin allergies stems from site-specific policies and primarily focuses on oral amoxicillin challenges or penicillin skin testing (PST). However, performing these tests may not be feasible at all facilities due to time constraints and lack of allergists. Therefore, alternative strategies are necessary, such as conducting detailed patient interviews. Few studies have evaluated switching to Β-lactam agents following a penicillin allergy interview alone. However, with thorough patient histories and detailed interviews, patients with reported penicillin allergies can safely use Β-lactam antibiotics.5 Implementing this procedure provides a cost-savings opportunity by not having to administer additional antibiotics for testing in addition to improving antibiotic stewardship.
The Memphis Veterans Affairs Medical Center (MVAMC) created the Allergy to Β-Lactam Evaluation (ABLE) process to clarify and remove penicillin allergies. The process involves conducting a thorough chart review and patient interview followed by completion of a note template that provides recommendations about patient allergies and Β-lactam prescribing. Mitchell et al found that the pharmacist-led process to be beneficial for addressing Β-lactam allergy clearance.6 As a result, the ABLE process was implemented at several other US Department of Veterans Affairs (VA) medical centers (VAMCs). Using the ABLE template, the purpose of this study was to evaluate the impact of a pharmacist-led penicillin allergy initiative on penicillin allergy delabeling with an interview process alone.
Methods
Prior to ABLE process implementation, there were no standardized procedures for documenting allergy histories. ABLE was implemented at the Robley Rex VAMC (RRVAMC) in November 2022. During the interview phase, patients were initially identified during admission via TheraDoc as having either a penicillin allergy or ADR. The infectious disease pharmacist or pharmacy resident interviewed patients with documented penicillin allergies or ADRs using a standardized questionnaire (eAppendix 1). Not all identified patients could be interviewed. Patients currently receiving an antibiotic were prioritized for interviews. Patients were excluded if they declined or were unable to be interviewed, although a patient’s caregiver(s) could be interviewed in person or via telephone, if the patient was not available.
Following the interview, pharmacists used guidance from the ABLE process in addition to a detailed EHR review to determine whether the patient was eligible for an allergy update or removal and/or switch to a Β-lactam antibiotic (Figure). If eligible for modification, the interviewing pharmacist made the necessary changes. A templated process note with patient-specific recommendations was entered into the Computerized Patient Record System (CPRS) and the primary care team attending physician was added as an additional signer to be alerted in the system note (eAppendix 2).
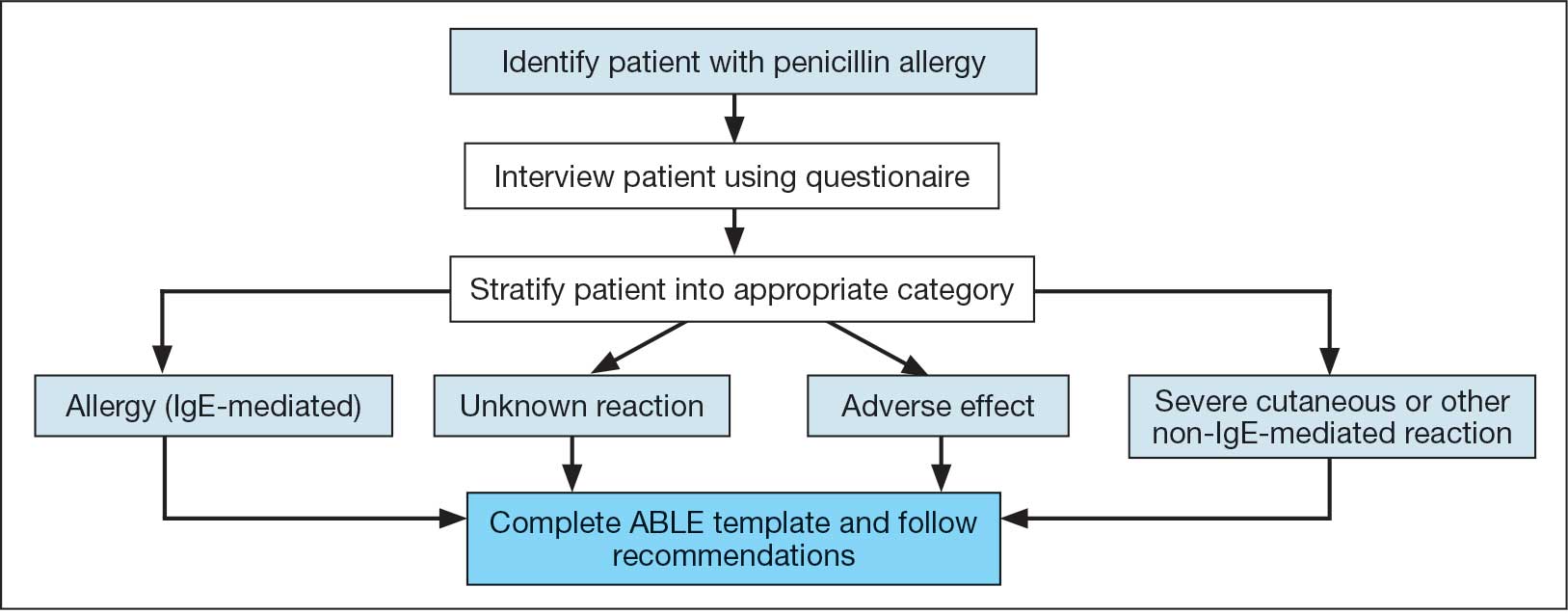
This single-center, retrospective cohort study involved review of CPRS notes and clinical interviews in the interviewed group. Hospitalized patients at the RRVAMC aged ≥ 18 years with a documented penicillin allergy or ADR were included. The historical control group consisted of patients admitted between October 31, 2019, and October 31, 2022, and the intervention group consisted of patients admitted between November 1, 2022, and March 1, 2023. Patients in the historical control group were matched 1:1 to the intervention group for penicillin allergy severity (allergy [IgE-mediated], unknown, adverse effect, severe cutaneous or other non–IgE-mediated reaction) and whether they received a noncarbapenem non–Β-lactam antibiotic.
The primary outcome was the number of patient allergies/ADRs removed or changed on patient profiles regardless of whether their antibiotic regimen was changed. This outcome was further assessed by evaluating the number of patient allergies or ADRs removed or changed on patient profiles with or without a change in antibiotic regimen. Primary outcomes were analyzed using χ2 and/ or Fisher exact tests, as appropriate to determine statistically significant differences between the interviewed group and the historical control.
Results
Seventy patients were included: 35 patients in the interviewed group and 35 patients in the historical control group, respectively. Both groups had a mean age of 72 years and predominantly included White male patients (Table 1). Following the interview, the allergy profile was modified for 6 patients (17%) in the interview group vs 0 patients in the control group (P = .03) (Table 2). The primary outcome was analyzed separately regardless of an antibiotic regimen change. There was not a statistically significant difference between groups when assessing patients for change in therapy (P > .99). All 6 patients with an allergy profile modification had no change in antibiotic regimen.
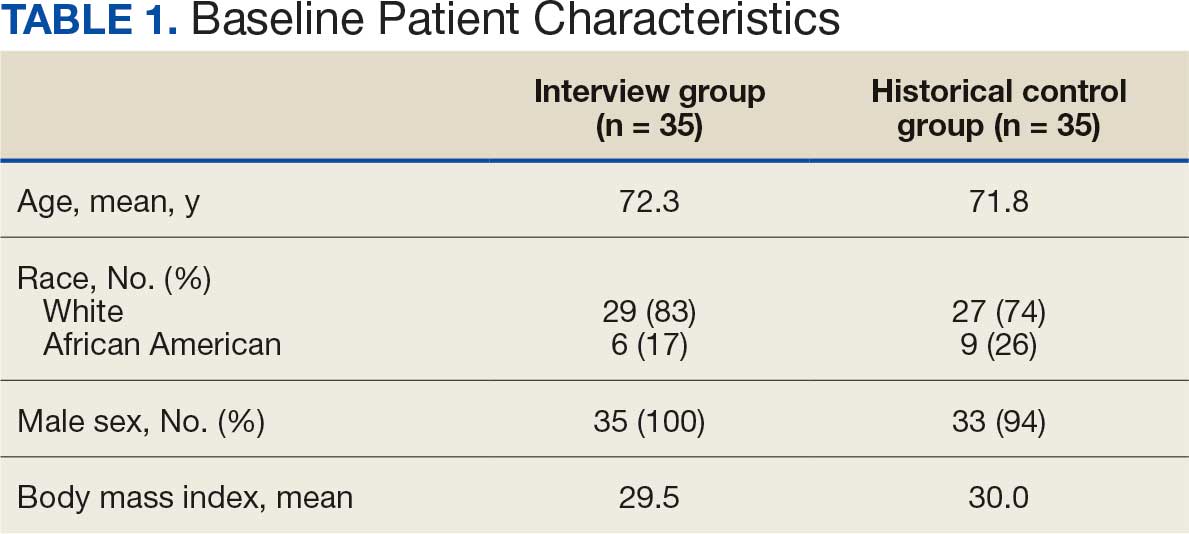

Discussion
This study suggests the ABLE process may be a valuable tool for adjusting penicillin allergies or ADRs within patient EHRs. In the interview group, allergies were modified in 6 (17%) patients while no patients in the control group had allergy modifications. Of the 6 allergy profile modifications, 4 allergy labels were changed from an allergy to an ADR. These patients were cleared to receive future Β-lactam antibiotics after clinicians recognized the lack of a true IgE-mediated allergic reaction. In addition, 2 of the modified allergy profiles removed the allergy designation. Although this represents a small subset of interviewed patients, it illustrates the clinical effectiveness of an interview process alone to remove penicillin allergy designations.
Previous research has assessed the impact of pharmacist intervention on penicillin allergy clarification. Mitchell et al implemented a pharmacist-driven Β-lactam allergy assessment and penicillin allergy clinic (PAC) at the MVAMC with the goal of evaluating its impact on allergy clearance. In their study, clinical pharmacy specialists evaluated patients with Β-lactam allergies, and those deemed eligible were later seen in the PAC. Among the 246 patients evaluated using the Β-lactam allergy assessment alone and who were not seen in the PAC, 25% had their penicillin allergy removed following a detailed assessment.6
Song et al evaluated the effectiveness and feasibility of a pharmacist-driven penicillin allergy delabeling pilot program without skin testing or oral challenges. Patients with penicillin allergies were interviewed by a pharmacy resident using a standardized checklist. Among the 66 patients interviewed, 12 (18%) met the criteria for delabeling and consented to removal of their allergy.7 The delabeling rates in these 2 studies are similar to the 17% rate of allergy modification in our study, although this study is the only one to compare results to a historical control group.
Harper et al evaluated the impact of a penicillin allergy assessment, including penicillin skin testing and oral amoxicillin challenges, on delabeling penicillin allergies. Pharmacists completed a penicillin allergy assessment and performed penicillin skin testing and/or oral amoxicillin challenges for eligible patients. Of 35 patients, 31 (89%) had their penicillin allergies delabeled in the EHR.8 The rate of penicillin allergy delabeling in Harper et al was likely higher than that seen in our study due to the use of oral challenge and skin testing. Regardless, a detailed penicillin allergy interview alone was effective at RRVAMC, resulting in a significant rate of allergy removal or change. This supports the use of detailed penicillin allergy assessments in settings where penicillin skin testing or oral challenges may not be feasible.
Mann et al demonstrated the effectiveness of penicillin allergy assessments in switching eligible patients to Β-lactam antibiotics. Their single-center, prospective study assessed the impact of a pharmacist-driven detailed penicillin allergy interview initiative. Interviews that evaluated potential changes to allergy profiles were conducted with 175 patients. Of these patients, 135 (77.1%) were on antimicrobial therapy and 42 (31.1%) patients receiving therapy met criteria to switch to a noncarbapenem Β-lactam antibiotic. Thirty-one patients (73.8%) switched with no signs or symptoms of intolerance demonstrating that an interview can be a valuable tool for antibiotic optimization, specifically in patients with penicillin allergy.9 No patients in our study switched antibiotic therapy, likely because only a small number of patients were eligible for transition to a noncarbapenem Β-lactam antibiotic. In the Mann et al study, non–Β-lactam antibiotics, such as fluoroquinolones and carbapenems, accounted for > 75% of the antibiotics used.
Limitations
The sample size of this study was small and its duration was short. There is a risk for selection bias as not all identified patients were able to be interviewed while admitted, but patients on antibiotics were prioritized as they were most likely to directly benefit during their current admission from a modification of their allergy. Most patients in the study were White and male, which may limit the generalizability of the results. Additionally, recommendations regarding antibiotic changes were primarily communicated to the treatment team based on a templated note in CPRS alone. Therefore, implementation of these recommendations largely relied upon nonverbal communication. Direct pharmacist-physician communication could have led to a larger impact on antimicrobial therapy changes. The interviewer’s participation in daily rounds with time allotted to discuss this topic can be considered in the future to improve these processes.
Conclusions
This study found that the ABLE process identified patients for penicillin allergy delabeling. With the high prevalence of inaccurate penicillin allergy documentation, this tool offers VA health care systems a way to empower pharmacists in allergy clarification, leading to improvements in antibiotic stewardship. Although the sample size was small, the ABLE process may provide a framework for VA clinicians. Future research has the potential to demonstrate the practicality and effectiveness this pharmacist-led penicillin allergy interview process can offer clinicians.
- Health care providers. Clinical features of penicillin allergy. Centers for Disease Control and Prevention. August 25, 2025. Accessed February 4, 2026. https://www.cdc.gov /antibiotic-use/hcp/clinical-signs/index.html
- Wrynn AF. Penicillin allergies: A guide for NPs. Nurse Pract. 2022;47:30-36. doi:10.1097/01.NPR.0000855312.11145.78
- Mohsen S, Dickinson JA, Somayaji R. Update on the adverse effects of antimicrobial therapies in community practice. Can Fam Physician. 2020;66:651-659.
- Sexually Transmitted Infections Treatment Guidelines, 2021. Managing persons who have a history of penicillin allergy. Centers for Disease Control and Prevention. September 21, 2022. Accessed February 4, 2026. https:// www.cdc.gov/std/treatment-guidelines/penicillin-allergy .htm
- Holmes AK, Bennett NT, Berry TP. Pharmacy driven assessment of appropriate antibiotic selection in patients with reported beta-lactam allergy. J Am Coll Clin Pharm. 2019;2:509-514. doi:10.1002/jac5.1135
- Mitchell AB, Ness RA, Bennett JG, et al. Implementation and impact of a Β-lactam allergy assessment protocol in a veteran population. Fed Pract. 2021;38:420-425. doi:10.12788/fp.0172
- Song YC, Nelson ZJ, Wankum MA, et al. Effectiveness and feasibility of pharmacist-driven penicillin allergy de-labeling pilot program without skin testing or oral challenges. Pharmacy (Basel). 2021;9:127. doi:10.3390/pharmacy9030127
- Harper HM, Sanchez M. Review of pharmacist driven penicillin allergy assessments and skin testing: a multicenter case-series. Hosp Pharm. 2022;57:469-473. doi:10.1177/00185787211046862
- Mann KL, Wu JY, Shah SS. Implementation of a pharmacist- driven detailed penicillin allergy interview. Ann Pharmacother. 2020;54:364-370. doi:10.1177/1060028019884874
Self-reported penicillin allergies are common, with a prevalence of about 10% of patients, according to the Centers for Disease Control and Prevention (CDC).1 However, only about 1% of patients have a true immunoglobulin E (IgE)-mediated allergy. This issue is often further complicated by inaccurate classification of nonallergic adverse effects as an allergy, resulting in incomplete allergy documentation in the electronic health record (EHR). The cross-reactivity rate with cephalosporins (Β-lactam antibiotics) in patients reporting a penicillin allergy is < 1%, which suggests that many patients with reported penicillin allergies can safely receive them.2 Despite this, patients with self-reported penicillin allergies often receive non–Β-lactam antibiotic agents, which may be associated with an increased risk of adverse drug reactions (ADRs), increased health care costs, and inferior clinical outcomes.3
Several strategies are recommended to assess patients with self-reported penicillin allergies. According to the CDC, evaluating a patient who reports a penicillin or other Β-lactam antibiotic allergy involves 3 steps: (1) obtaining a thorough medical history, including previous exposures to penicillin or other Β-lactam antibiotic; (2) performing a skin test using the penicillin major and minor determinants; and (3) among those who have a negative penicillin skin test, performing an observed oral challenge with 250 mg amoxicillin before proceeding directly to treatment with the indicated Β-lactam therapy.4
Most existing clinical guidance for assessing patients with self-reported penicillin allergies stems from site-specific policies and primarily focuses on oral amoxicillin challenges or penicillin skin testing (PST). However, performing these tests may not be feasible at all facilities due to time constraints and lack of allergists. Therefore, alternative strategies are necessary, such as conducting detailed patient interviews. Few studies have evaluated switching to Β-lactam agents following a penicillin allergy interview alone. However, with thorough patient histories and detailed interviews, patients with reported penicillin allergies can safely use Β-lactam antibiotics.5 Implementing this procedure provides a cost-savings opportunity by not having to administer additional antibiotics for testing in addition to improving antibiotic stewardship.
The Memphis Veterans Affairs Medical Center (MVAMC) created the Allergy to Β-Lactam Evaluation (ABLE) process to clarify and remove penicillin allergies. The process involves conducting a thorough chart review and patient interview followed by completion of a note template that provides recommendations about patient allergies and Β-lactam prescribing. Mitchell et al found that the pharmacist-led process to be beneficial for addressing Β-lactam allergy clearance.6 As a result, the ABLE process was implemented at several other US Department of Veterans Affairs (VA) medical centers (VAMCs). Using the ABLE template, the purpose of this study was to evaluate the impact of a pharmacist-led penicillin allergy initiative on penicillin allergy delabeling with an interview process alone.
Methods
Prior to ABLE process implementation, there were no standardized procedures for documenting allergy histories. ABLE was implemented at the Robley Rex VAMC (RRVAMC) in November 2022. During the interview phase, patients were initially identified during admission via TheraDoc as having either a penicillin allergy or ADR. The infectious disease pharmacist or pharmacy resident interviewed patients with documented penicillin allergies or ADRs using a standardized questionnaire (eAppendix 1). Not all identified patients could be interviewed. Patients currently receiving an antibiotic were prioritized for interviews. Patients were excluded if they declined or were unable to be interviewed, although a patient’s caregiver(s) could be interviewed in person or via telephone, if the patient was not available.
Following the interview, pharmacists used guidance from the ABLE process in addition to a detailed EHR review to determine whether the patient was eligible for an allergy update or removal and/or switch to a Β-lactam antibiotic (Figure). If eligible for modification, the interviewing pharmacist made the necessary changes. A templated process note with patient-specific recommendations was entered into the Computerized Patient Record System (CPRS) and the primary care team attending physician was added as an additional signer to be alerted in the system note (eAppendix 2).

This single-center, retrospective cohort study involved review of CPRS notes and clinical interviews in the interviewed group. Hospitalized patients at the RRVAMC aged ≥ 18 years with a documented penicillin allergy or ADR were included. The historical control group consisted of patients admitted between October 31, 2019, and October 31, 2022, and the intervention group consisted of patients admitted between November 1, 2022, and March 1, 2023. Patients in the historical control group were matched 1:1 to the intervention group for penicillin allergy severity (allergy [IgE-mediated], unknown, adverse effect, severe cutaneous or other non–IgE-mediated reaction) and whether they received a noncarbapenem non–Β-lactam antibiotic.
The primary outcome was the number of patient allergies/ADRs removed or changed on patient profiles regardless of whether their antibiotic regimen was changed. This outcome was further assessed by evaluating the number of patient allergies or ADRs removed or changed on patient profiles with or without a change in antibiotic regimen. Primary outcomes were analyzed using χ2 and/ or Fisher exact tests, as appropriate to determine statistically significant differences between the interviewed group and the historical control.
Results
Seventy patients were included: 35 patients in the interviewed group and 35 patients in the historical control group, respectively. Both groups had a mean age of 72 years and predominantly included White male patients (Table 1). Following the interview, the allergy profile was modified for 6 patients (17%) in the interview group vs 0 patients in the control group (P = .03) (Table 2). The primary outcome was analyzed separately regardless of an antibiotic regimen change. There was not a statistically significant difference between groups when assessing patients for change in therapy (P > .99). All 6 patients with an allergy profile modification had no change in antibiotic regimen.


Discussion
This study suggests the ABLE process may be a valuable tool for adjusting penicillin allergies or ADRs within patient EHRs. In the interview group, allergies were modified in 6 (17%) patients while no patients in the control group had allergy modifications. Of the 6 allergy profile modifications, 4 allergy labels were changed from an allergy to an ADR. These patients were cleared to receive future Β-lactam antibiotics after clinicians recognized the lack of a true IgE-mediated allergic reaction. In addition, 2 of the modified allergy profiles removed the allergy designation. Although this represents a small subset of interviewed patients, it illustrates the clinical effectiveness of an interview process alone to remove penicillin allergy designations.
Previous research has assessed the impact of pharmacist intervention on penicillin allergy clarification. Mitchell et al implemented a pharmacist-driven Β-lactam allergy assessment and penicillin allergy clinic (PAC) at the MVAMC with the goal of evaluating its impact on allergy clearance. In their study, clinical pharmacy specialists evaluated patients with Β-lactam allergies, and those deemed eligible were later seen in the PAC. Among the 246 patients evaluated using the Β-lactam allergy assessment alone and who were not seen in the PAC, 25% had their penicillin allergy removed following a detailed assessment.6
Song et al evaluated the effectiveness and feasibility of a pharmacist-driven penicillin allergy delabeling pilot program without skin testing or oral challenges. Patients with penicillin allergies were interviewed by a pharmacy resident using a standardized checklist. Among the 66 patients interviewed, 12 (18%) met the criteria for delabeling and consented to removal of their allergy.7 The delabeling rates in these 2 studies are similar to the 17% rate of allergy modification in our study, although this study is the only one to compare results to a historical control group.
Harper et al evaluated the impact of a penicillin allergy assessment, including penicillin skin testing and oral amoxicillin challenges, on delabeling penicillin allergies. Pharmacists completed a penicillin allergy assessment and performed penicillin skin testing and/or oral amoxicillin challenges for eligible patients. Of 35 patients, 31 (89%) had their penicillin allergies delabeled in the EHR.8 The rate of penicillin allergy delabeling in Harper et al was likely higher than that seen in our study due to the use of oral challenge and skin testing. Regardless, a detailed penicillin allergy interview alone was effective at RRVAMC, resulting in a significant rate of allergy removal or change. This supports the use of detailed penicillin allergy assessments in settings where penicillin skin testing or oral challenges may not be feasible.
Mann et al demonstrated the effectiveness of penicillin allergy assessments in switching eligible patients to Β-lactam antibiotics. Their single-center, prospective study assessed the impact of a pharmacist-driven detailed penicillin allergy interview initiative. Interviews that evaluated potential changes to allergy profiles were conducted with 175 patients. Of these patients, 135 (77.1%) were on antimicrobial therapy and 42 (31.1%) patients receiving therapy met criteria to switch to a noncarbapenem Β-lactam antibiotic. Thirty-one patients (73.8%) switched with no signs or symptoms of intolerance demonstrating that an interview can be a valuable tool for antibiotic optimization, specifically in patients with penicillin allergy.9 No patients in our study switched antibiotic therapy, likely because only a small number of patients were eligible for transition to a noncarbapenem Β-lactam antibiotic. In the Mann et al study, non–Β-lactam antibiotics, such as fluoroquinolones and carbapenems, accounted for > 75% of the antibiotics used.
Limitations
The sample size of this study was small and its duration was short. There is a risk for selection bias as not all identified patients were able to be interviewed while admitted, but patients on antibiotics were prioritized as they were most likely to directly benefit during their current admission from a modification of their allergy. Most patients in the study were White and male, which may limit the generalizability of the results. Additionally, recommendations regarding antibiotic changes were primarily communicated to the treatment team based on a templated note in CPRS alone. Therefore, implementation of these recommendations largely relied upon nonverbal communication. Direct pharmacist-physician communication could have led to a larger impact on antimicrobial therapy changes. The interviewer’s participation in daily rounds with time allotted to discuss this topic can be considered in the future to improve these processes.
Conclusions
This study found that the ABLE process identified patients for penicillin allergy delabeling. With the high prevalence of inaccurate penicillin allergy documentation, this tool offers VA health care systems a way to empower pharmacists in allergy clarification, leading to improvements in antibiotic stewardship. Although the sample size was small, the ABLE process may provide a framework for VA clinicians. Future research has the potential to demonstrate the practicality and effectiveness this pharmacist-led penicillin allergy interview process can offer clinicians.
Self-reported penicillin allergies are common, with a prevalence of about 10% of patients, according to the Centers for Disease Control and Prevention (CDC).1 However, only about 1% of patients have a true immunoglobulin E (IgE)-mediated allergy. This issue is often further complicated by inaccurate classification of nonallergic adverse effects as an allergy, resulting in incomplete allergy documentation in the electronic health record (EHR). The cross-reactivity rate with cephalosporins (Β-lactam antibiotics) in patients reporting a penicillin allergy is < 1%, which suggests that many patients with reported penicillin allergies can safely receive them.2 Despite this, patients with self-reported penicillin allergies often receive non–Β-lactam antibiotic agents, which may be associated with an increased risk of adverse drug reactions (ADRs), increased health care costs, and inferior clinical outcomes.3
Several strategies are recommended to assess patients with self-reported penicillin allergies. According to the CDC, evaluating a patient who reports a penicillin or other Β-lactam antibiotic allergy involves 3 steps: (1) obtaining a thorough medical history, including previous exposures to penicillin or other Β-lactam antibiotic; (2) performing a skin test using the penicillin major and minor determinants; and (3) among those who have a negative penicillin skin test, performing an observed oral challenge with 250 mg amoxicillin before proceeding directly to treatment with the indicated Β-lactam therapy.4
Most existing clinical guidance for assessing patients with self-reported penicillin allergies stems from site-specific policies and primarily focuses on oral amoxicillin challenges or penicillin skin testing (PST). However, performing these tests may not be feasible at all facilities due to time constraints and lack of allergists. Therefore, alternative strategies are necessary, such as conducting detailed patient interviews. Few studies have evaluated switching to Β-lactam agents following a penicillin allergy interview alone. However, with thorough patient histories and detailed interviews, patients with reported penicillin allergies can safely use Β-lactam antibiotics.5 Implementing this procedure provides a cost-savings opportunity by not having to administer additional antibiotics for testing in addition to improving antibiotic stewardship.
The Memphis Veterans Affairs Medical Center (MVAMC) created the Allergy to Β-Lactam Evaluation (ABLE) process to clarify and remove penicillin allergies. The process involves conducting a thorough chart review and patient interview followed by completion of a note template that provides recommendations about patient allergies and Β-lactam prescribing. Mitchell et al found that the pharmacist-led process to be beneficial for addressing Β-lactam allergy clearance.6 As a result, the ABLE process was implemented at several other US Department of Veterans Affairs (VA) medical centers (VAMCs). Using the ABLE template, the purpose of this study was to evaluate the impact of a pharmacist-led penicillin allergy initiative on penicillin allergy delabeling with an interview process alone.
Methods
Prior to ABLE process implementation, there were no standardized procedures for documenting allergy histories. ABLE was implemented at the Robley Rex VAMC (RRVAMC) in November 2022. During the interview phase, patients were initially identified during admission via TheraDoc as having either a penicillin allergy or ADR. The infectious disease pharmacist or pharmacy resident interviewed patients with documented penicillin allergies or ADRs using a standardized questionnaire (eAppendix 1). Not all identified patients could be interviewed. Patients currently receiving an antibiotic were prioritized for interviews. Patients were excluded if they declined or were unable to be interviewed, although a patient’s caregiver(s) could be interviewed in person or via telephone, if the patient was not available.
Following the interview, pharmacists used guidance from the ABLE process in addition to a detailed EHR review to determine whether the patient was eligible for an allergy update or removal and/or switch to a Β-lactam antibiotic (Figure). If eligible for modification, the interviewing pharmacist made the necessary changes. A templated process note with patient-specific recommendations was entered into the Computerized Patient Record System (CPRS) and the primary care team attending physician was added as an additional signer to be alerted in the system note (eAppendix 2).

This single-center, retrospective cohort study involved review of CPRS notes and clinical interviews in the interviewed group. Hospitalized patients at the RRVAMC aged ≥ 18 years with a documented penicillin allergy or ADR were included. The historical control group consisted of patients admitted between October 31, 2019, and October 31, 2022, and the intervention group consisted of patients admitted between November 1, 2022, and March 1, 2023. Patients in the historical control group were matched 1:1 to the intervention group for penicillin allergy severity (allergy [IgE-mediated], unknown, adverse effect, severe cutaneous or other non–IgE-mediated reaction) and whether they received a noncarbapenem non–Β-lactam antibiotic.
The primary outcome was the number of patient allergies/ADRs removed or changed on patient profiles regardless of whether their antibiotic regimen was changed. This outcome was further assessed by evaluating the number of patient allergies or ADRs removed or changed on patient profiles with or without a change in antibiotic regimen. Primary outcomes were analyzed using χ2 and/ or Fisher exact tests, as appropriate to determine statistically significant differences between the interviewed group and the historical control.
Results
Seventy patients were included: 35 patients in the interviewed group and 35 patients in the historical control group, respectively. Both groups had a mean age of 72 years and predominantly included White male patients (Table 1). Following the interview, the allergy profile was modified for 6 patients (17%) in the interview group vs 0 patients in the control group (P = .03) (Table 2). The primary outcome was analyzed separately regardless of an antibiotic regimen change. There was not a statistically significant difference between groups when assessing patients for change in therapy (P > .99). All 6 patients with an allergy profile modification had no change in antibiotic regimen.


Discussion
This study suggests the ABLE process may be a valuable tool for adjusting penicillin allergies or ADRs within patient EHRs. In the interview group, allergies were modified in 6 (17%) patients while no patients in the control group had allergy modifications. Of the 6 allergy profile modifications, 4 allergy labels were changed from an allergy to an ADR. These patients were cleared to receive future Β-lactam antibiotics after clinicians recognized the lack of a true IgE-mediated allergic reaction. In addition, 2 of the modified allergy profiles removed the allergy designation. Although this represents a small subset of interviewed patients, it illustrates the clinical effectiveness of an interview process alone to remove penicillin allergy designations.
Previous research has assessed the impact of pharmacist intervention on penicillin allergy clarification. Mitchell et al implemented a pharmacist-driven Β-lactam allergy assessment and penicillin allergy clinic (PAC) at the MVAMC with the goal of evaluating its impact on allergy clearance. In their study, clinical pharmacy specialists evaluated patients with Β-lactam allergies, and those deemed eligible were later seen in the PAC. Among the 246 patients evaluated using the Β-lactam allergy assessment alone and who were not seen in the PAC, 25% had their penicillin allergy removed following a detailed assessment.6
Song et al evaluated the effectiveness and feasibility of a pharmacist-driven penicillin allergy delabeling pilot program without skin testing or oral challenges. Patients with penicillin allergies were interviewed by a pharmacy resident using a standardized checklist. Among the 66 patients interviewed, 12 (18%) met the criteria for delabeling and consented to removal of their allergy.7 The delabeling rates in these 2 studies are similar to the 17% rate of allergy modification in our study, although this study is the only one to compare results to a historical control group.
Harper et al evaluated the impact of a penicillin allergy assessment, including penicillin skin testing and oral amoxicillin challenges, on delabeling penicillin allergies. Pharmacists completed a penicillin allergy assessment and performed penicillin skin testing and/or oral amoxicillin challenges for eligible patients. Of 35 patients, 31 (89%) had their penicillin allergies delabeled in the EHR.8 The rate of penicillin allergy delabeling in Harper et al was likely higher than that seen in our study due to the use of oral challenge and skin testing. Regardless, a detailed penicillin allergy interview alone was effective at RRVAMC, resulting in a significant rate of allergy removal or change. This supports the use of detailed penicillin allergy assessments in settings where penicillin skin testing or oral challenges may not be feasible.
Mann et al demonstrated the effectiveness of penicillin allergy assessments in switching eligible patients to Β-lactam antibiotics. Their single-center, prospective study assessed the impact of a pharmacist-driven detailed penicillin allergy interview initiative. Interviews that evaluated potential changes to allergy profiles were conducted with 175 patients. Of these patients, 135 (77.1%) were on antimicrobial therapy and 42 (31.1%) patients receiving therapy met criteria to switch to a noncarbapenem Β-lactam antibiotic. Thirty-one patients (73.8%) switched with no signs or symptoms of intolerance demonstrating that an interview can be a valuable tool for antibiotic optimization, specifically in patients with penicillin allergy.9 No patients in our study switched antibiotic therapy, likely because only a small number of patients were eligible for transition to a noncarbapenem Β-lactam antibiotic. In the Mann et al study, non–Β-lactam antibiotics, such as fluoroquinolones and carbapenems, accounted for > 75% of the antibiotics used.
Limitations
The sample size of this study was small and its duration was short. There is a risk for selection bias as not all identified patients were able to be interviewed while admitted, but patients on antibiotics were prioritized as they were most likely to directly benefit during their current admission from a modification of their allergy. Most patients in the study were White and male, which may limit the generalizability of the results. Additionally, recommendations regarding antibiotic changes were primarily communicated to the treatment team based on a templated note in CPRS alone. Therefore, implementation of these recommendations largely relied upon nonverbal communication. Direct pharmacist-physician communication could have led to a larger impact on antimicrobial therapy changes. The interviewer’s participation in daily rounds with time allotted to discuss this topic can be considered in the future to improve these processes.
Conclusions
This study found that the ABLE process identified patients for penicillin allergy delabeling. With the high prevalence of inaccurate penicillin allergy documentation, this tool offers VA health care systems a way to empower pharmacists in allergy clarification, leading to improvements in antibiotic stewardship. Although the sample size was small, the ABLE process may provide a framework for VA clinicians. Future research has the potential to demonstrate the practicality and effectiveness this pharmacist-led penicillin allergy interview process can offer clinicians.
- Health care providers. Clinical features of penicillin allergy. Centers for Disease Control and Prevention. August 25, 2025. Accessed February 4, 2026. https://www.cdc.gov /antibiotic-use/hcp/clinical-signs/index.html
- Wrynn AF. Penicillin allergies: A guide for NPs. Nurse Pract. 2022;47:30-36. doi:10.1097/01.NPR.0000855312.11145.78
- Mohsen S, Dickinson JA, Somayaji R. Update on the adverse effects of antimicrobial therapies in community practice. Can Fam Physician. 2020;66:651-659.
- Sexually Transmitted Infections Treatment Guidelines, 2021. Managing persons who have a history of penicillin allergy. Centers for Disease Control and Prevention. September 21, 2022. Accessed February 4, 2026. https:// www.cdc.gov/std/treatment-guidelines/penicillin-allergy .htm
- Holmes AK, Bennett NT, Berry TP. Pharmacy driven assessment of appropriate antibiotic selection in patients with reported beta-lactam allergy. J Am Coll Clin Pharm. 2019;2:509-514. doi:10.1002/jac5.1135
- Mitchell AB, Ness RA, Bennett JG, et al. Implementation and impact of a Β-lactam allergy assessment protocol in a veteran population. Fed Pract. 2021;38:420-425. doi:10.12788/fp.0172
- Song YC, Nelson ZJ, Wankum MA, et al. Effectiveness and feasibility of pharmacist-driven penicillin allergy de-labeling pilot program without skin testing or oral challenges. Pharmacy (Basel). 2021;9:127. doi:10.3390/pharmacy9030127
- Harper HM, Sanchez M. Review of pharmacist driven penicillin allergy assessments and skin testing: a multicenter case-series. Hosp Pharm. 2022;57:469-473. doi:10.1177/00185787211046862
- Mann KL, Wu JY, Shah SS. Implementation of a pharmacist- driven detailed penicillin allergy interview. Ann Pharmacother. 2020;54:364-370. doi:10.1177/1060028019884874
- Health care providers. Clinical features of penicillin allergy. Centers for Disease Control and Prevention. August 25, 2025. Accessed February 4, 2026. https://www.cdc.gov /antibiotic-use/hcp/clinical-signs/index.html
- Wrynn AF. Penicillin allergies: A guide for NPs. Nurse Pract. 2022;47:30-36. doi:10.1097/01.NPR.0000855312.11145.78
- Mohsen S, Dickinson JA, Somayaji R. Update on the adverse effects of antimicrobial therapies in community practice. Can Fam Physician. 2020;66:651-659.
- Sexually Transmitted Infections Treatment Guidelines, 2021. Managing persons who have a history of penicillin allergy. Centers for Disease Control and Prevention. September 21, 2022. Accessed February 4, 2026. https:// www.cdc.gov/std/treatment-guidelines/penicillin-allergy .htm
- Holmes AK, Bennett NT, Berry TP. Pharmacy driven assessment of appropriate antibiotic selection in patients with reported beta-lactam allergy. J Am Coll Clin Pharm. 2019;2:509-514. doi:10.1002/jac5.1135
- Mitchell AB, Ness RA, Bennett JG, et al. Implementation and impact of a Β-lactam allergy assessment protocol in a veteran population. Fed Pract. 2021;38:420-425. doi:10.12788/fp.0172
- Song YC, Nelson ZJ, Wankum MA, et al. Effectiveness and feasibility of pharmacist-driven penicillin allergy de-labeling pilot program without skin testing or oral challenges. Pharmacy (Basel). 2021;9:127. doi:10.3390/pharmacy9030127
- Harper HM, Sanchez M. Review of pharmacist driven penicillin allergy assessments and skin testing: a multicenter case-series. Hosp Pharm. 2022;57:469-473. doi:10.1177/00185787211046862
- Mann KL, Wu JY, Shah SS. Implementation of a pharmacist- driven detailed penicillin allergy interview. Ann Pharmacother. 2020;54:364-370. doi:10.1177/1060028019884874
Implementation of a Pharmacist-Led Penicillin Allergy Interview at a Veterans Care Facility
Implementation of a Pharmacist-Led Penicillin Allergy Interview at a Veterans Care Facility