User login
Dupilumab for Bullous Pemphigoid: To Treat or Not to Treat?
Dupilumab for Bullous Pemphigoid: To Treat or Not to Treat?
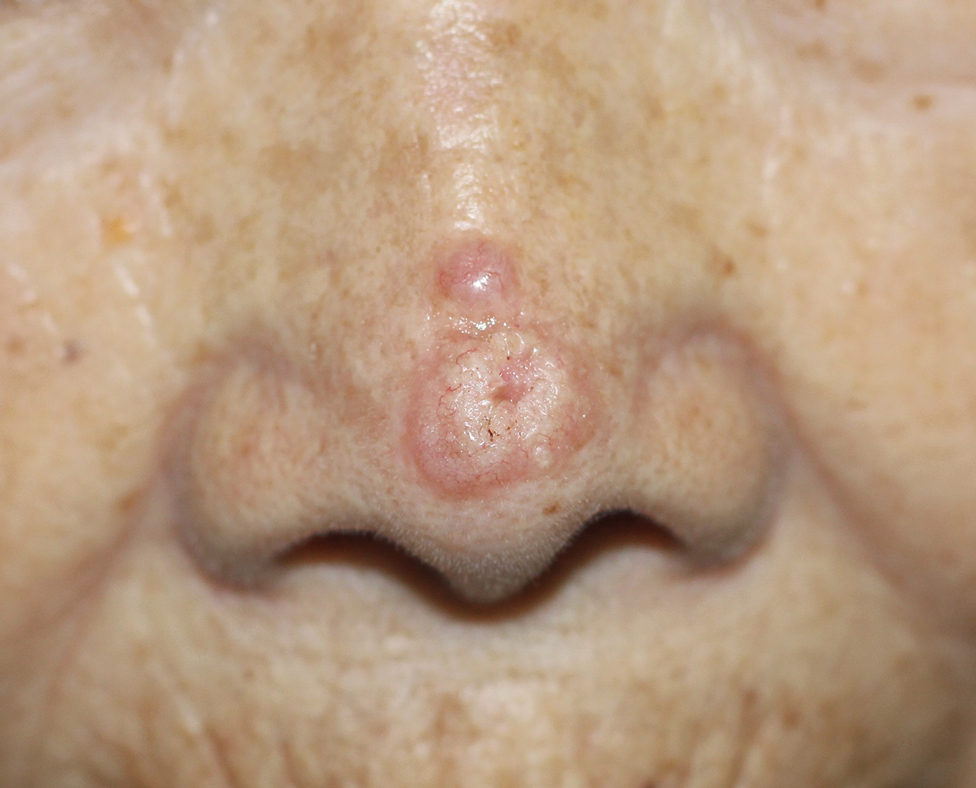
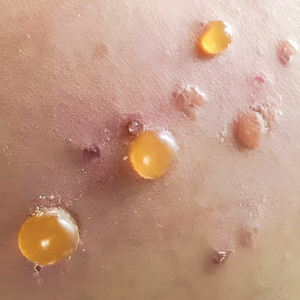
Bullous pemphigoid (BP) is a chronic autoimmune skin condition characterized by large, fluid-filled blisters (bullae) on the skin. There usually is severe itching, which may antedate the bullae by several weeks. Bullous pemphigoid most commonly affects adults aged 60 years and older with comorbidities such as metabolic syndrome or other autoimmune diseases. The typical blistering is subepidermal, and its occurrence seems to be driven by autoantibodies targeting the epidermal basement membrane zone.
Conventional treatment for BP has long included systemic corticosteroids and immunosuppressants; this combination of drugs effectively controls inflammation but also is associated with considerable risks such as infection, hyperglycemia with worsening diabetes, hypertension, osteoporosis and fracture risk, and delirium, particularly in older patients. Recent developments in biologic therapy have supported the utilization of potentially safer alternatives. Specifically, dupilumab has demonstrated efficacy in the treatment of atopic dermatitis. Case reports and small research studies included in a recent review have suggested that dupilumab could be effective in managing BP, potentially offering a safer treatment option.1 Importantly, dupilumab recently was approved for BP by the US Food and Drug Administration, making it the first biologic approved for treating this condition.2 In this article, we discuss dupilumab as an option for clinicians to prescribe as an initial therapy for BP.
Pathophysiology of Bullous Pemphigoid and Dupilumab
Bullous pemphigoid pathophysiology is driven by 2 main components: immunologic and inflammatory processes. The immune-mediated attack consists of autoantibodies against the hemidesmosomal proteins BP180 and BP230 in the basement membrane zone, which anchor the epidermis to the dermis.1 When these proteins are targeted, a cascade of responses is triggered, including the release of inflammatory cells, particularly eosinophils and neutrophils, into the skin. These cells release inflammatory mediators that break down the basement membrane, resulting in the separation of the epidermis from the dermis and leading to subepidermal blister formation.
The inflammation associated with BP largely is driven by a T helper 2–dominant immune response, with increased levels of cytokines such as interleukin (IL) 4 and IL-13.1 The chronic inflammation contributes to the characteristic blisters and intense pruritus seen in patients with BP. Traditional treatments such as corticosteroids work broadly to suppress this inflammation by reducing the production of all proinflammatory cytokines; however, they lack specificity, which dampens the targeted immune response against BP and broadly suppresses the immune system, increasing the risk for infection and other adverse effects. For older patients with multiple health conditions, these adverse effects can impact quality of life and pose health risks.
Interleukin 4 and IL-13 contribute to eosinophilia by upregulating the expression of eosinophil chemoattractant cytokines that promote eosinophil migration and adhesion.1 This is where dupilumab presents a promising alternative. Dupilumab is a monoclonal antibody that selectively targets the key cytokines implicated in BP. It binds to the IL-4 receptor alpha subunit, which IL-4 and IL-13 share, thereby inhibiting the signaling of both cytokines.1
Dupilumab Reports and Case Series
Current evidence supporting the use of dupilumab for BP consists primarily of case series and uncontrolled clinical observations, both of which are useful to formulate but not to test hypotheses.3 One case series of 13 patients diagnosed with BP reported that 92.3% (12/13) of patients achieved disease clearance or satisfactory control, with 53.8% (7/13) achieving total lesion clearance and no adverse events observed.4 In a larger case series, 87.0% of 146 patients achieved disease control with an initial dose of dupilumab 600 mg followed by 300 mg every 2 weeks for 4 weeks, though the study also looked at secondary outcomes for up to 64 weeks.5 Both these findings are compatible with the idea that dupilumab has efficacy in BP, but they cannot be used to test hypotheses.3,4
In a single case report of refractory BP, the combination of dupilumab and omalizumab—a monoclonal antibody that blocks the binding of IgE to mast cells, basophils, and dendritic cells—has shown efficacy, particularly when BP is unresponsive to standard therapies.6 The case involved a 72-year-old woman with unresponsive BP who was treated with a combination of dupilumab and omalizumab. She experienced marked improvement in pruritus and bullae accompanied by normalization of anti-BP180 autoantibody levels. This result points to the possibility that IL-4 and IL-13 blockade could influence autoantibody production. The authors attributed the reduction in pruritus to dupilumab, although the drug was administered in combination with omalizumab.6
Small Randomized Trial of Dupilumab
In addition to these uncontrolled clinical observations, dupilumab recently demonstrated positive results in a small randomized trial.7 Patients who were assigned to receive dupilumab were 5 times more likely to achieve sustained remission compared to those assigned to a placebo. All randomized patients also were treated with a standard-of-care oral corticosteroid regimen, which complicated the ability to isolate the specific effects of dupilumab.7 Finally, a completed totality of evidence on the prescription of dupilumab for refractory BP should include more reliable results from larger randomized trials.8
Final Thoughts
Dupilumab is now approved by the US Food and Drug Administration for the treatment of BP, marking an important milestone as the first targeted biologic therapy for this condition. While the totality of evidence is still evolving, the available data indicate both efficacy and a favorable safety profile when compared to traditional corticosteroids and immunosuppressants. Based on the available data, we suggest that health care providers consider dupilumab as a first-line monotherapy in appropriate patients with BP without specific contraindications, particularly as a practical and safe alternative to corticosteroids and immunosuppressive therapies in older adults with comorbidities. If the patient demonstrates clinical improvement in symptom relief, the therapy can be continued. This strategy minimizes initial exposure to more potent biologics or therapies with higher toxicity profiles. For patients without adequate disease control or with severe initial presentations requiring rapid improvement, clinicians may consider escalation to adjunctive or alternative therapies, such as rituximab, omalizumab, or a short course of systemic corticosteroids for acute management.
Emerging data suggest that dupilumab is a promising therapy for BP. We suggest that it may be a safer targeted alternative to traditional corticosteroids and immunosuppressive therapies. The ability of dupilumab to mitigate inflammation without broadly suppressing the immune system suggests that it may turn out to be an especially valuable option for older patients and those with comorbid conditions.
Acknowledgement—The authors thank Jodi Fiedler, MD (Boca Raton, FL), for her expert advice and assistance.
- Zhang L, Chen Z, Wang L, et al. Bullous pemphigoid: the role of type 2 inflammation in its pathogenesis and the prospect of targeted therapy. Front Immunol. 2023;14:1115083. doi:10.3389/fimmu.2023.1115083
- Sanofi. Dupixent approved in the US as the only targeted medicine to treat patients with bullous pemphigoid. News release. June 20, 2025. Accessed September 8, 2025. https://www.sanofi.com/en/media-room/press-releases/2025/2025-06-20-05-00-00-3102518
- Hennekens CH, DeMets D. Statistical association and causation: contributions of different types of evidence. JAMA. 2011;306:1134-1136. doi:10.1001/jama.2011.322
- Abdat R, Waldman RA, de Bedout V, et al. Dupilumab as a novel therapy for bullous pemphigoid: a multicenter case series. J Am Acad Dermatol. 2020;83:46-52. doi:10.1016/j.jaad.2020.01.089
- Zhao L, Wang Q, Liang G, et al. Evaluation of dupilumab in patients with bullous pemphigoid. JAMA Dermatol. 2023;159:953-960. doi:10.1001/jamadermatol.2023.2428
- Seyed Jafari SM, Feldmeyer L, Bossart S, et al. Case report: combination of omalizumab and dupilumab for recalcitrant bullous pemphigoid. Front Immunol. 2021;11:611549. doi:10.3389/fimmu.2020.611549
- US National Library of Medicine. A multicenter, randomized, double-blind, placebo-controlled, parallel-group study to evaluate the efficacy and safety of dupilumab in adult patients with bullous pemphigoid. ClinicalTrials.gov. Published 2019. Accessed March 10, 2026. https://clinicaltrials.gov/ct2/show/NCT04206553
- Hennekens CH, DeMets D. The need for large scale randomized evidence without undue emphasis on small trials, meta-analyses or subgroup analyses. JAMA. 2009;302:2361-2362. doi:10.1001/jama.2009.1829
Bullous pemphigoid (BP) is a chronic autoimmune skin condition characterized by large, fluid-filled blisters (bullae) on the skin. There usually is severe itching, which may antedate the bullae by several weeks. Bullous pemphigoid most commonly affects adults aged 60 years and older with comorbidities such as metabolic syndrome or other autoimmune diseases. The typical blistering is subepidermal, and its occurrence seems to be driven by autoantibodies targeting the epidermal basement membrane zone.
Conventional treatment for BP has long included systemic corticosteroids and immunosuppressants; this combination of drugs effectively controls inflammation but also is associated with considerable risks such as infection, hyperglycemia with worsening diabetes, hypertension, osteoporosis and fracture risk, and delirium, particularly in older patients. Recent developments in biologic therapy have supported the utilization of potentially safer alternatives. Specifically, dupilumab has demonstrated efficacy in the treatment of atopic dermatitis. Case reports and small research studies included in a recent review have suggested that dupilumab could be effective in managing BP, potentially offering a safer treatment option.1 Importantly, dupilumab recently was approved for BP by the US Food and Drug Administration, making it the first biologic approved for treating this condition.2 In this article, we discuss dupilumab as an option for clinicians to prescribe as an initial therapy for BP.
Pathophysiology of Bullous Pemphigoid and Dupilumab
Bullous pemphigoid pathophysiology is driven by 2 main components: immunologic and inflammatory processes. The immune-mediated attack consists of autoantibodies against the hemidesmosomal proteins BP180 and BP230 in the basement membrane zone, which anchor the epidermis to the dermis.1 When these proteins are targeted, a cascade of responses is triggered, including the release of inflammatory cells, particularly eosinophils and neutrophils, into the skin. These cells release inflammatory mediators that break down the basement membrane, resulting in the separation of the epidermis from the dermis and leading to subepidermal blister formation.
The inflammation associated with BP largely is driven by a T helper 2–dominant immune response, with increased levels of cytokines such as interleukin (IL) 4 and IL-13.1 The chronic inflammation contributes to the characteristic blisters and intense pruritus seen in patients with BP. Traditional treatments such as corticosteroids work broadly to suppress this inflammation by reducing the production of all proinflammatory cytokines; however, they lack specificity, which dampens the targeted immune response against BP and broadly suppresses the immune system, increasing the risk for infection and other adverse effects. For older patients with multiple health conditions, these adverse effects can impact quality of life and pose health risks.
Interleukin 4 and IL-13 contribute to eosinophilia by upregulating the expression of eosinophil chemoattractant cytokines that promote eosinophil migration and adhesion.1 This is where dupilumab presents a promising alternative. Dupilumab is a monoclonal antibody that selectively targets the key cytokines implicated in BP. It binds to the IL-4 receptor alpha subunit, which IL-4 and IL-13 share, thereby inhibiting the signaling of both cytokines.1
Dupilumab Reports and Case Series
Current evidence supporting the use of dupilumab for BP consists primarily of case series and uncontrolled clinical observations, both of which are useful to formulate but not to test hypotheses.3 One case series of 13 patients diagnosed with BP reported that 92.3% (12/13) of patients achieved disease clearance or satisfactory control, with 53.8% (7/13) achieving total lesion clearance and no adverse events observed.4 In a larger case series, 87.0% of 146 patients achieved disease control with an initial dose of dupilumab 600 mg followed by 300 mg every 2 weeks for 4 weeks, though the study also looked at secondary outcomes for up to 64 weeks.5 Both these findings are compatible with the idea that dupilumab has efficacy in BP, but they cannot be used to test hypotheses.3,4
In a single case report of refractory BP, the combination of dupilumab and omalizumab—a monoclonal antibody that blocks the binding of IgE to mast cells, basophils, and dendritic cells—has shown efficacy, particularly when BP is unresponsive to standard therapies.6 The case involved a 72-year-old woman with unresponsive BP who was treated with a combination of dupilumab and omalizumab. She experienced marked improvement in pruritus and bullae accompanied by normalization of anti-BP180 autoantibody levels. This result points to the possibility that IL-4 and IL-13 blockade could influence autoantibody production. The authors attributed the reduction in pruritus to dupilumab, although the drug was administered in combination with omalizumab.6
Small Randomized Trial of Dupilumab
In addition to these uncontrolled clinical observations, dupilumab recently demonstrated positive results in a small randomized trial.7 Patients who were assigned to receive dupilumab were 5 times more likely to achieve sustained remission compared to those assigned to a placebo. All randomized patients also were treated with a standard-of-care oral corticosteroid regimen, which complicated the ability to isolate the specific effects of dupilumab.7 Finally, a completed totality of evidence on the prescription of dupilumab for refractory BP should include more reliable results from larger randomized trials.8
Final Thoughts
Dupilumab is now approved by the US Food and Drug Administration for the treatment of BP, marking an important milestone as the first targeted biologic therapy for this condition. While the totality of evidence is still evolving, the available data indicate both efficacy and a favorable safety profile when compared to traditional corticosteroids and immunosuppressants. Based on the available data, we suggest that health care providers consider dupilumab as a first-line monotherapy in appropriate patients with BP without specific contraindications, particularly as a practical and safe alternative to corticosteroids and immunosuppressive therapies in older adults with comorbidities. If the patient demonstrates clinical improvement in symptom relief, the therapy can be continued. This strategy minimizes initial exposure to more potent biologics or therapies with higher toxicity profiles. For patients without adequate disease control or with severe initial presentations requiring rapid improvement, clinicians may consider escalation to adjunctive or alternative therapies, such as rituximab, omalizumab, or a short course of systemic corticosteroids for acute management.
Emerging data suggest that dupilumab is a promising therapy for BP. We suggest that it may be a safer targeted alternative to traditional corticosteroids and immunosuppressive therapies. The ability of dupilumab to mitigate inflammation without broadly suppressing the immune system suggests that it may turn out to be an especially valuable option for older patients and those with comorbid conditions.
Acknowledgement—The authors thank Jodi Fiedler, MD (Boca Raton, FL), for her expert advice and assistance.
Bullous pemphigoid (BP) is a chronic autoimmune skin condition characterized by large, fluid-filled blisters (bullae) on the skin. There usually is severe itching, which may antedate the bullae by several weeks. Bullous pemphigoid most commonly affects adults aged 60 years and older with comorbidities such as metabolic syndrome or other autoimmune diseases. The typical blistering is subepidermal, and its occurrence seems to be driven by autoantibodies targeting the epidermal basement membrane zone.
Conventional treatment for BP has long included systemic corticosteroids and immunosuppressants; this combination of drugs effectively controls inflammation but also is associated with considerable risks such as infection, hyperglycemia with worsening diabetes, hypertension, osteoporosis and fracture risk, and delirium, particularly in older patients. Recent developments in biologic therapy have supported the utilization of potentially safer alternatives. Specifically, dupilumab has demonstrated efficacy in the treatment of atopic dermatitis. Case reports and small research studies included in a recent review have suggested that dupilumab could be effective in managing BP, potentially offering a safer treatment option.1 Importantly, dupilumab recently was approved for BP by the US Food and Drug Administration, making it the first biologic approved for treating this condition.2 In this article, we discuss dupilumab as an option for clinicians to prescribe as an initial therapy for BP.
Pathophysiology of Bullous Pemphigoid and Dupilumab
Bullous pemphigoid pathophysiology is driven by 2 main components: immunologic and inflammatory processes. The immune-mediated attack consists of autoantibodies against the hemidesmosomal proteins BP180 and BP230 in the basement membrane zone, which anchor the epidermis to the dermis.1 When these proteins are targeted, a cascade of responses is triggered, including the release of inflammatory cells, particularly eosinophils and neutrophils, into the skin. These cells release inflammatory mediators that break down the basement membrane, resulting in the separation of the epidermis from the dermis and leading to subepidermal blister formation.
The inflammation associated with BP largely is driven by a T helper 2–dominant immune response, with increased levels of cytokines such as interleukin (IL) 4 and IL-13.1 The chronic inflammation contributes to the characteristic blisters and intense pruritus seen in patients with BP. Traditional treatments such as corticosteroids work broadly to suppress this inflammation by reducing the production of all proinflammatory cytokines; however, they lack specificity, which dampens the targeted immune response against BP and broadly suppresses the immune system, increasing the risk for infection and other adverse effects. For older patients with multiple health conditions, these adverse effects can impact quality of life and pose health risks.
Interleukin 4 and IL-13 contribute to eosinophilia by upregulating the expression of eosinophil chemoattractant cytokines that promote eosinophil migration and adhesion.1 This is where dupilumab presents a promising alternative. Dupilumab is a monoclonal antibody that selectively targets the key cytokines implicated in BP. It binds to the IL-4 receptor alpha subunit, which IL-4 and IL-13 share, thereby inhibiting the signaling of both cytokines.1
Dupilumab Reports and Case Series
Current evidence supporting the use of dupilumab for BP consists primarily of case series and uncontrolled clinical observations, both of which are useful to formulate but not to test hypotheses.3 One case series of 13 patients diagnosed with BP reported that 92.3% (12/13) of patients achieved disease clearance or satisfactory control, with 53.8% (7/13) achieving total lesion clearance and no adverse events observed.4 In a larger case series, 87.0% of 146 patients achieved disease control with an initial dose of dupilumab 600 mg followed by 300 mg every 2 weeks for 4 weeks, though the study also looked at secondary outcomes for up to 64 weeks.5 Both these findings are compatible with the idea that dupilumab has efficacy in BP, but they cannot be used to test hypotheses.3,4
In a single case report of refractory BP, the combination of dupilumab and omalizumab—a monoclonal antibody that blocks the binding of IgE to mast cells, basophils, and dendritic cells—has shown efficacy, particularly when BP is unresponsive to standard therapies.6 The case involved a 72-year-old woman with unresponsive BP who was treated with a combination of dupilumab and omalizumab. She experienced marked improvement in pruritus and bullae accompanied by normalization of anti-BP180 autoantibody levels. This result points to the possibility that IL-4 and IL-13 blockade could influence autoantibody production. The authors attributed the reduction in pruritus to dupilumab, although the drug was administered in combination with omalizumab.6
Small Randomized Trial of Dupilumab
In addition to these uncontrolled clinical observations, dupilumab recently demonstrated positive results in a small randomized trial.7 Patients who were assigned to receive dupilumab were 5 times more likely to achieve sustained remission compared to those assigned to a placebo. All randomized patients also were treated with a standard-of-care oral corticosteroid regimen, which complicated the ability to isolate the specific effects of dupilumab.7 Finally, a completed totality of evidence on the prescription of dupilumab for refractory BP should include more reliable results from larger randomized trials.8
Final Thoughts
Dupilumab is now approved by the US Food and Drug Administration for the treatment of BP, marking an important milestone as the first targeted biologic therapy for this condition. While the totality of evidence is still evolving, the available data indicate both efficacy and a favorable safety profile when compared to traditional corticosteroids and immunosuppressants. Based on the available data, we suggest that health care providers consider dupilumab as a first-line monotherapy in appropriate patients with BP without specific contraindications, particularly as a practical and safe alternative to corticosteroids and immunosuppressive therapies in older adults with comorbidities. If the patient demonstrates clinical improvement in symptom relief, the therapy can be continued. This strategy minimizes initial exposure to more potent biologics or therapies with higher toxicity profiles. For patients without adequate disease control or with severe initial presentations requiring rapid improvement, clinicians may consider escalation to adjunctive or alternative therapies, such as rituximab, omalizumab, or a short course of systemic corticosteroids for acute management.
Emerging data suggest that dupilumab is a promising therapy for BP. We suggest that it may be a safer targeted alternative to traditional corticosteroids and immunosuppressive therapies. The ability of dupilumab to mitigate inflammation without broadly suppressing the immune system suggests that it may turn out to be an especially valuable option for older patients and those with comorbid conditions.
Acknowledgement—The authors thank Jodi Fiedler, MD (Boca Raton, FL), for her expert advice and assistance.
- Zhang L, Chen Z, Wang L, et al. Bullous pemphigoid: the role of type 2 inflammation in its pathogenesis and the prospect of targeted therapy. Front Immunol. 2023;14:1115083. doi:10.3389/fimmu.2023.1115083
- Sanofi. Dupixent approved in the US as the only targeted medicine to treat patients with bullous pemphigoid. News release. June 20, 2025. Accessed September 8, 2025. https://www.sanofi.com/en/media-room/press-releases/2025/2025-06-20-05-00-00-3102518
- Hennekens CH, DeMets D. Statistical association and causation: contributions of different types of evidence. JAMA. 2011;306:1134-1136. doi:10.1001/jama.2011.322
- Abdat R, Waldman RA, de Bedout V, et al. Dupilumab as a novel therapy for bullous pemphigoid: a multicenter case series. J Am Acad Dermatol. 2020;83:46-52. doi:10.1016/j.jaad.2020.01.089
- Zhao L, Wang Q, Liang G, et al. Evaluation of dupilumab in patients with bullous pemphigoid. JAMA Dermatol. 2023;159:953-960. doi:10.1001/jamadermatol.2023.2428
- Seyed Jafari SM, Feldmeyer L, Bossart S, et al. Case report: combination of omalizumab and dupilumab for recalcitrant bullous pemphigoid. Front Immunol. 2021;11:611549. doi:10.3389/fimmu.2020.611549
- US National Library of Medicine. A multicenter, randomized, double-blind, placebo-controlled, parallel-group study to evaluate the efficacy and safety of dupilumab in adult patients with bullous pemphigoid. ClinicalTrials.gov. Published 2019. Accessed March 10, 2026. https://clinicaltrials.gov/ct2/show/NCT04206553
- Hennekens CH, DeMets D. The need for large scale randomized evidence without undue emphasis on small trials, meta-analyses or subgroup analyses. JAMA. 2009;302:2361-2362. doi:10.1001/jama.2009.1829
- Zhang L, Chen Z, Wang L, et al. Bullous pemphigoid: the role of type 2 inflammation in its pathogenesis and the prospect of targeted therapy. Front Immunol. 2023;14:1115083. doi:10.3389/fimmu.2023.1115083
- Sanofi. Dupixent approved in the US as the only targeted medicine to treat patients with bullous pemphigoid. News release. June 20, 2025. Accessed September 8, 2025. https://www.sanofi.com/en/media-room/press-releases/2025/2025-06-20-05-00-00-3102518
- Hennekens CH, DeMets D. Statistical association and causation: contributions of different types of evidence. JAMA. 2011;306:1134-1136. doi:10.1001/jama.2011.322
- Abdat R, Waldman RA, de Bedout V, et al. Dupilumab as a novel therapy for bullous pemphigoid: a multicenter case series. J Am Acad Dermatol. 2020;83:46-52. doi:10.1016/j.jaad.2020.01.089
- Zhao L, Wang Q, Liang G, et al. Evaluation of dupilumab in patients with bullous pemphigoid. JAMA Dermatol. 2023;159:953-960. doi:10.1001/jamadermatol.2023.2428
- Seyed Jafari SM, Feldmeyer L, Bossart S, et al. Case report: combination of omalizumab and dupilumab for recalcitrant bullous pemphigoid. Front Immunol. 2021;11:611549. doi:10.3389/fimmu.2020.611549
- US National Library of Medicine. A multicenter, randomized, double-blind, placebo-controlled, parallel-group study to evaluate the efficacy and safety of dupilumab in adult patients with bullous pemphigoid. ClinicalTrials.gov. Published 2019. Accessed March 10, 2026. https://clinicaltrials.gov/ct2/show/NCT04206553
- Hennekens CH, DeMets D. The need for large scale randomized evidence without undue emphasis on small trials, meta-analyses or subgroup analyses. JAMA. 2009;302:2361-2362. doi:10.1001/jama.2009.1829
Dupilumab for Bullous Pemphigoid: To Treat or Not to Treat?
Dupilumab for Bullous Pemphigoid: To Treat or Not to Treat?
PRACTICE POINTS
- Bullous pemphigoid (BP) is a pruritic autoimmune blistering disease in older adults that often requires systemic therapy with associated risks.
- Dupilumab targets interleukin (IL) 4/IL-13–driven inflammation and shows promising efficacy in BP.
- As the first approved biologic for BP, dupilumab may offer a safer alternative to corticosteroids in select patients.
Social Drivers of Health Curriculum for Dermatology Residents: the UCSF Experience
Social Drivers of Health Curriculum for Dermatology Residents: the UCSF Experience
Social drivers of health (SDH) describe the conditions in which an individual is born, grows, lives, works, and ages—all of which collectively influence their health. Examples of SDH include employment status, literacy level, education level, housing status, food access, income level, and social cohesion. Social drivers of health are critical catalysts to attaining health equity. Effectively applying an understanding of how SDH affect the care of all patients is an essential competency for physicians practicing in the modern era of rising income inequality and housing instability and increasing racial, ethnic, language, religious, and cultural diversity in the United States; however, in dermatology residency, this skill set often is developed by the hidden curriculum (ie, the informal curriculum that is based on what patient scenarios a resident happens to face) rather than one represented by formal educational objectives.2
Adding to this challenge of limited formal curricula is that caring for minoritized, marginalized, and other populations facing specific barriers can evoke feelings of frustration, helplessness, and even anger. These feelings can test the limits of a physician’s identity as a healer, leading to burnout and self-protective attitudes such as distancing (emotionally, physically, or both) from these patients.3 This is particularly relevant given that the majority (76%-79% each year from 2007-2019) of medical student matriculants come from families with incomes in the top 2 quintiles nationwide, and fewer than 6% come from the lowest quintile earners.4,5 These data indicate that most trainees have not experienced (and may even have a hard time imagining) the degree of economic and housing instability faced by many of their low-income patients, the care of whom disproportionately falls to large academic medical centers, which sponsor dermatology training programs.6 Many trainees may feel uncomfortable communicating across the broad range of racial, socioeconomic, linguistic, and cultural differences they encounter during training and in practice. Structured opportunities to provide care in a supervised supportive environment combined with didactics that emphasize practical, evidence-based strategies can build empathy, improve attitudes toward patients from diverse backgrounds, and strengthen self-efficacy in challenging scenarios.3
In the past decade, there has been a push toward integrating our understanding of SDH into formal medical training.7 Other specialty training programs—including psychiatry,8 internal medicine,9 pediatrics,10 and family medicine11—have incorporated these elements into their curricula and competency evaluations. In dermatology, as in other specialties, making and implementing effective, patient-centered care plans requires attention to the various social and structural drivers that may influence outcomes. Dermatologists therefore should be educated about SDH during their training programs and empowered to address the ways they affect patient care.
At the University of California San Francisco (UCSF)(San Francisco, California), our dermatology trainees care for patients in several hospital systems citywide, including a tertiary academic medical center with multiple locations, a county hospital, and a Veterans Affairs medical center. Given the diversity of patient populations across our training sites—including many racially and ethnically minoritized individuals, immigrants, patients with limited English proficiency, people experiencing homelessness, and sexual and gender diverse individuals—we identified a critical opportunity to enhance our training through formal didactics and hands-on experiences that integrate SDH into existing curricula and strengthen trainees’ ability to provide high-quality care to all patients.
Implementing an SDH Curriculum
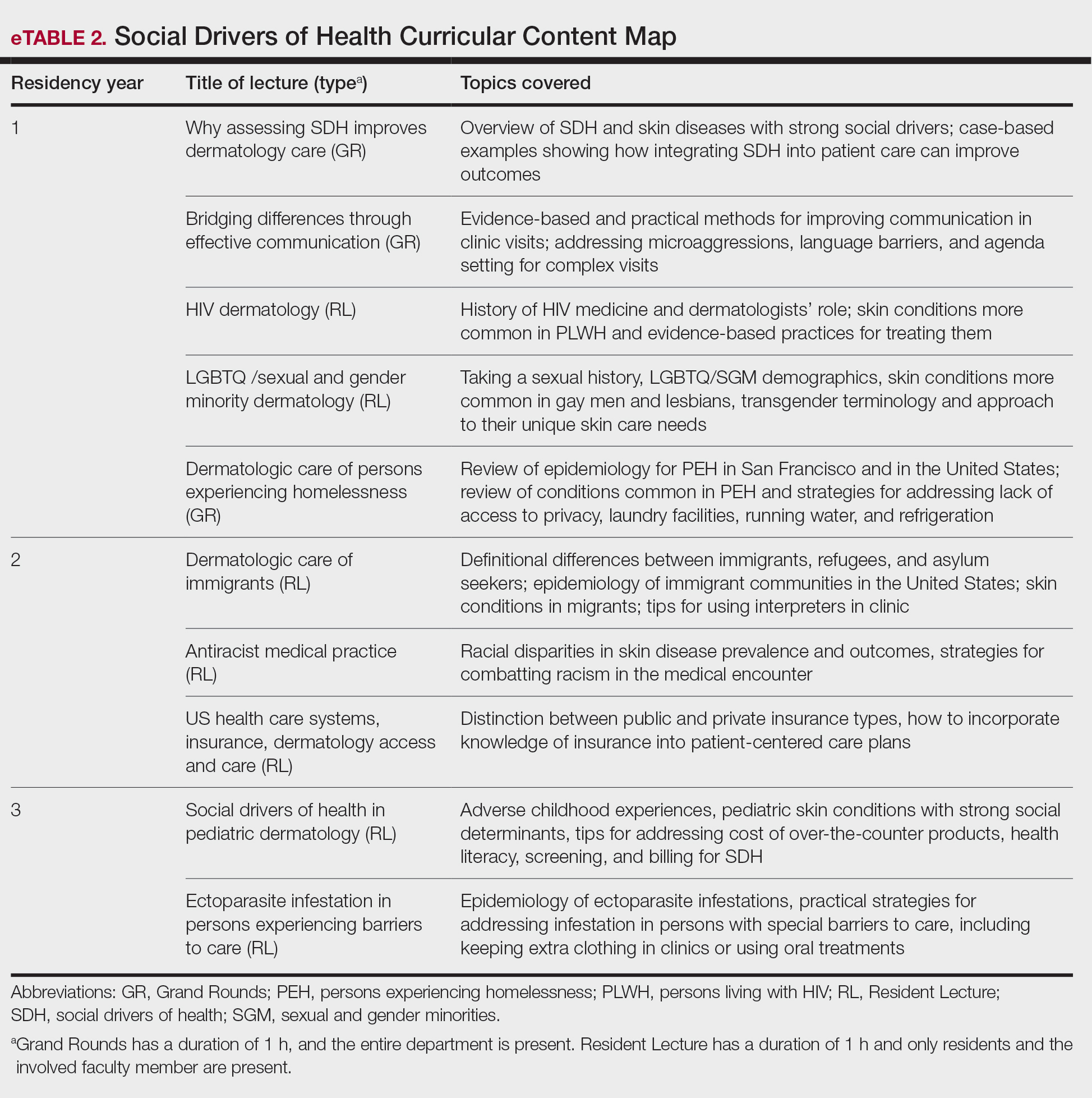
In May 2020, UCSF dermatology faculty with an interest in SDH collaborated with departmental educational leadership to develop a formal SDH curriculum centered around 8 core learning objectives for residents (eTable 1). To achieve these objectives, we organized a 3-year didactic and experiential curriculum consisting of lectures (eTable 2), grand rounds sessions, journal clubs, and community engagement opportunities. Residents also spend 7 months during their training rotating at San Francisco’s city and county hospital (Zuckerberg San Francisco General Hospital [San Francisco, California]) where all faculty are members of the core SDH curriculum development team and where residents can put into practice many of the skills learned in formal didactics to develop patient-centered care plans for low-income patients, approximately 40% of whom have limited English proficiency.

To further center the importance of SDH and health equity in our training program, we developed a Health Equity Chief leadership role for senior dermatology residents. Each year, 2 to 4 residents volunteer for and serve in this role, wherein they work with core faculty to review and improve SDH curriculum elements. They also work to enhance community engagement opportunities for residents (eg, pathway programs aimed at diversifying the dermatology workforce by introducing historically excluded local high school and college students to dermatology as a career path) and improve dermatology trainees’ awareness of the history and health needs of the specific communities we serve in San Francisco. They also are prepared to become leaders in the field of health equity and to improve the care of diverse patient populations after residency. Our faculty curriculum leaders meet quarterly with our Health Equity Chiefs to review their individual and collective goals and strategize ways to improve learner and community engagement. Departmental funds are made available to support these efforts.
Leadership at our safety-net county hospital also developed a patient navigator position to improve our ability to care for patients with the most complex medical conditions and social needs. This role is held by a medical student taking a funded gap year and incorporates aspects of social work (eg, identifying barriers to care and connecting patients with resources such as transportation), quality improvement, and clinical research.12
Assessing Residents’ Experience of a New SDH Curriculum
Prior to curriculum implementation, we surveyed graduating UCSF dermatology residents in June 2020 to assess their familiarity with SDH and the social and medical needs of various populations facing barriers to care, their comfort level with specific challenging clinical situations, and their desire for additional training. Responses were measured using a 5-part Likert scale, with additional options for free-text response. After initiating the SDH curriculum in July 2020, we sent the same survey each year to all senior residents immediately prior to their graduation, offering a small financial incentive ($15 cash gift card) to those who completed the survey. We obtained UCSF Institutional Review Board approval to utilize these survey data to better understand and to enhance residents’ experience of the SDH curriculum.
All 8 residents invited in 2020 completed the survey assessing curriculum efficacy (100% response rate). For the 2023 and 2024 classes, data were analyzed in aggregate (n=14), with a 50% response rate. After implementation of the SDH curriculum, there was improvement in learners’ awareness of challenges faced by every patient population, from a mean (SD) of 3.12 (0.66) to 4.52 (SD, 0.69)(P<.05). Learners were more comfortable handling hypothetical clinical scenarios requiring them to identify and address specific SDH after vs before implementation of the curriculum (mean [SD], 3.5 [1.06] before vs 4.0 [1.16] after)(P>.05), though this difference was not statistically learners’ awareness of challenges faced by every patient population, from a mean (SD) of 3.12 (0.66) to 4.52 (0.69)(P<.05). Learners were more comfortable handling hypothetical clinical scenarios requiring them to identify and address specific SDH after vs before implementation of the curriculum (mean [SD], 3.5 [1.06] before vs 4.0 [1.16] after)(P>.05), though this difference was not statistically significant. Finally, many respondents expressed appreciation that our curriculum improved their ability to care for patients in complex social circumstances. Residents suggested in the free-text responses that learning more about the historical underpinnings of health disparities, opportunities for grassroots activism, and how to provide more culturally competent care of Native American populations could improve our curriculum.
Implications for Dermatology Training
Our survey results indicate that a formal SDH curriculum can improve dermatology residents’ ability to care for populations with complex social needs. We advocate for implementing SDH curricula into dermatology training programs nationwide, as has been recommended by others.13,14 We also propose that structural competency should eventually be a key dermatologic competency as determined by the Accreditation Council for Graduate Medical Education, in line with the American Medical Association’s recommendation that structural competency is a learned skill required to end health inequity.15 The Accreditation Council for Graduate Medical Education specialty program requirements currently are being revised; interested individuals can engage in this process by submitting this suggestion for public comment (https://www.acgme.org/programs-and-institutions/programs/review-and-comment/).
Limitations of a survey include the relatively small sample size (7-8 per year) and variable response rates. In addition, we did not survey each class of residents at the beginning and end of their training; our comparisons therefore were limited by comparing different individuals with distinct backgrounds and experiences. Furthermore, we acknowledge that the experience of developing this curriculum in San Francisco may be distinct from other communities, where access to dermatologic care may vary according to both the availability of public health insurance and the treatments covered by public insurers. In San Francisco, insurance coverage is near universal, such that residents in our training program regularly care for undocumented immigrants, persons experiencing homelessness, and other populations that might find it challenging to present to dermatology clinics in other settings nationwide.
Final Thoughts
Future directions of our curriculum include exploration of novel curriculum delivery methods (including a problem-based curriculum approach and other more experiential didactics), increased opportunities for community engagement, greater focus on advocacy with an emphasis on broader social and structural policies and their downstream effects, and focusing more specifically on the history and needs of specific low-income San Francisco neighborhoods and diverse patient populations.
- US Department of Health and Human Services. Health Equity in Healthy People 2030. Accessed January 7, 2025. https://odphp.healtwh.gov/healthypeople/priority-areas/health-equity-healthy-people-2030
- Axelson DJ, Stull MJ, Coates WC. Social determinants of health: a missing link in emergency medicine training. AEM Educ Train. 2018;2:66-68. doi:10.1002/aet2.10056
- Brenner AM, Guerrero APS, Beresin EV, et al. Teaching medical students and residents about homelessness: complex, evidence-based, and imperative. Acad Psychiatry. 2016;40:572-575. doi:10.1007/s40596-016-0571-6
- Youngclaus J, Roskovensky L. An Updated Look at the Economic Diversity of U.S. Medical Students. American Association of Medical Colleges Analysis in Brief. 2018;18. https://www.aamc.org/media/9596/download?attachment
- Shahriar AA, Puram VV, Miller JM, et al. Socioeconomic diversity of the matriculating US medical student body by race, ethnicity, and sex, 2017-2019. JAMA Netw Open. 2022;5:E222621. doi:10.1001/jamanetworkopen.2022.2621
- Williams JC, Maxey AE, Wei ML, et al. A cross-sectional analysis of Medicaid acceptance among US dermatology residency training programs. J Am Acad Dermatol. 2022;86:453-455. doi:10.1016/j.jaad.2021.09.046
- Daniel H, Bornstein S, Kane G; Health and Public Policy Committee of the American College of Physicians. Addressing social determinants to improve patient care and promote health equity: an American College of Physicians position paper. Ann Intern Med. 2018;168:577-578. doi:10.2105/AJPH
- Hansen H, Kline N, Braslow J, et al. From cultural to structural competency—training psychiatry residents to act on social determinants of health and institutional racism. JAMA Psychiatry. 2018;75:117-118. doi:10.1001/jamapsychiatry.2017.3894
- Schmidt S, Higgins S, George M, et al. An experiential resident module for understanding social determinants of health at an academic safety-net hospital. MedEdPORTAL. 2017;26:10647. doi:10.15766/mep_2374-8265.10647
- Hoffman BD, Rose J, Best D, et al. The community pediatrics training initiative project planning tool: a practical approach to community-based advocacy. MedEdPORTAL. 2017;13:10630.
- Chrisman-Khawam L, Abdullah N, Dhoopar A. Teaching health-care trainees empathy and homelessness IQ through service learning, reflective practice, and altruistic attribution. Int J Psychiatry Med. 2017;52:245-254. doi:10.1177/0091217417730288
- Sanchez-Anguiano ME, Klufas D, Amerson E. Screening for cardiometabolic risk factors in patients with psoriasis and hidradenitis suppurativa: a pilot study in a safety net population. J Am Acad Dermatol. 2024;91:1269-1272. doi:10.1016/j.jaad.2024.07.1518
- Riley C, Vasquez R, Pritchett EN. Equipping dermatologists to address structural and social drivers of inequities—structural competency. JAMA Dermatol. 2024;160:1037-1038. doi:10.1001/jamadermatol.2024.2351
- Crawl-Bey A, Pritchett EN, Riley C. 54338 Structural competency in dermatology: a pilot curriculum for equipping residents to address structural factors that contribute to health inequity. J Am Acad Dermatol. 2024;91(3 suppl):AB318. doi:10.1016/j.jaad.2024.07.1264
- Smith TM. New competency focus involves structural factors in health. American Medical Association. December 18, 202. Accessed March 23, 2026. https://www.ama-assn.org/education/changemeded-initiative/new-competency-focus-involves-structural-factors-health
Social drivers of health (SDH) describe the conditions in which an individual is born, grows, lives, works, and ages—all of which collectively influence their health. Examples of SDH include employment status, literacy level, education level, housing status, food access, income level, and social cohesion. Social drivers of health are critical catalysts to attaining health equity. Effectively applying an understanding of how SDH affect the care of all patients is an essential competency for physicians practicing in the modern era of rising income inequality and housing instability and increasing racial, ethnic, language, religious, and cultural diversity in the United States; however, in dermatology residency, this skill set often is developed by the hidden curriculum (ie, the informal curriculum that is based on what patient scenarios a resident happens to face) rather than one represented by formal educational objectives.2
Adding to this challenge of limited formal curricula is that caring for minoritized, marginalized, and other populations facing specific barriers can evoke feelings of frustration, helplessness, and even anger. These feelings can test the limits of a physician’s identity as a healer, leading to burnout and self-protective attitudes such as distancing (emotionally, physically, or both) from these patients.3 This is particularly relevant given that the majority (76%-79% each year from 2007-2019) of medical student matriculants come from families with incomes in the top 2 quintiles nationwide, and fewer than 6% come from the lowest quintile earners.4,5 These data indicate that most trainees have not experienced (and may even have a hard time imagining) the degree of economic and housing instability faced by many of their low-income patients, the care of whom disproportionately falls to large academic medical centers, which sponsor dermatology training programs.6 Many trainees may feel uncomfortable communicating across the broad range of racial, socioeconomic, linguistic, and cultural differences they encounter during training and in practice. Structured opportunities to provide care in a supervised supportive environment combined with didactics that emphasize practical, evidence-based strategies can build empathy, improve attitudes toward patients from diverse backgrounds, and strengthen self-efficacy in challenging scenarios.3
In the past decade, there has been a push toward integrating our understanding of SDH into formal medical training.7 Other specialty training programs—including psychiatry,8 internal medicine,9 pediatrics,10 and family medicine11—have incorporated these elements into their curricula and competency evaluations. In dermatology, as in other specialties, making and implementing effective, patient-centered care plans requires attention to the various social and structural drivers that may influence outcomes. Dermatologists therefore should be educated about SDH during their training programs and empowered to address the ways they affect patient care.
At the University of California San Francisco (UCSF)(San Francisco, California), our dermatology trainees care for patients in several hospital systems citywide, including a tertiary academic medical center with multiple locations, a county hospital, and a Veterans Affairs medical center. Given the diversity of patient populations across our training sites—including many racially and ethnically minoritized individuals, immigrants, patients with limited English proficiency, people experiencing homelessness, and sexual and gender diverse individuals—we identified a critical opportunity to enhance our training through formal didactics and hands-on experiences that integrate SDH into existing curricula and strengthen trainees’ ability to provide high-quality care to all patients.
Implementing an SDH Curriculum
In May 2020, UCSF dermatology faculty with an interest in SDH collaborated with departmental educational leadership to develop a formal SDH curriculum centered around 8 core learning objectives for residents (eTable 1). To achieve these objectives, we organized a 3-year didactic and experiential curriculum consisting of lectures (eTable 2), grand rounds sessions, journal clubs, and community engagement opportunities. Residents also spend 7 months during their training rotating at San Francisco’s city and county hospital (Zuckerberg San Francisco General Hospital [San Francisco, California]) where all faculty are members of the core SDH curriculum development team and where residents can put into practice many of the skills learned in formal didactics to develop patient-centered care plans for low-income patients, approximately 40% of whom have limited English proficiency.


To further center the importance of SDH and health equity in our training program, we developed a Health Equity Chief leadership role for senior dermatology residents. Each year, 2 to 4 residents volunteer for and serve in this role, wherein they work with core faculty to review and improve SDH curriculum elements. They also work to enhance community engagement opportunities for residents (eg, pathway programs aimed at diversifying the dermatology workforce by introducing historically excluded local high school and college students to dermatology as a career path) and improve dermatology trainees’ awareness of the history and health needs of the specific communities we serve in San Francisco. They also are prepared to become leaders in the field of health equity and to improve the care of diverse patient populations after residency. Our faculty curriculum leaders meet quarterly with our Health Equity Chiefs to review their individual and collective goals and strategize ways to improve learner and community engagement. Departmental funds are made available to support these efforts.
Leadership at our safety-net county hospital also developed a patient navigator position to improve our ability to care for patients with the most complex medical conditions and social needs. This role is held by a medical student taking a funded gap year and incorporates aspects of social work (eg, identifying barriers to care and connecting patients with resources such as transportation), quality improvement, and clinical research.12
Assessing Residents’ Experience of a New SDH Curriculum
Prior to curriculum implementation, we surveyed graduating UCSF dermatology residents in June 2020 to assess their familiarity with SDH and the social and medical needs of various populations facing barriers to care, their comfort level with specific challenging clinical situations, and their desire for additional training. Responses were measured using a 5-part Likert scale, with additional options for free-text response. After initiating the SDH curriculum in July 2020, we sent the same survey each year to all senior residents immediately prior to their graduation, offering a small financial incentive ($15 cash gift card) to those who completed the survey. We obtained UCSF Institutional Review Board approval to utilize these survey data to better understand and to enhance residents’ experience of the SDH curriculum.
All 8 residents invited in 2020 completed the survey assessing curriculum efficacy (100% response rate). For the 2023 and 2024 classes, data were analyzed in aggregate (n=14), with a 50% response rate. After implementation of the SDH curriculum, there was improvement in learners’ awareness of challenges faced by every patient population, from a mean (SD) of 3.12 (0.66) to 4.52 (SD, 0.69)(P<.05). Learners were more comfortable handling hypothetical clinical scenarios requiring them to identify and address specific SDH after vs before implementation of the curriculum (mean [SD], 3.5 [1.06] before vs 4.0 [1.16] after)(P>.05), though this difference was not statistically learners’ awareness of challenges faced by every patient population, from a mean (SD) of 3.12 (0.66) to 4.52 (0.69)(P<.05). Learners were more comfortable handling hypothetical clinical scenarios requiring them to identify and address specific SDH after vs before implementation of the curriculum (mean [SD], 3.5 [1.06] before vs 4.0 [1.16] after)(P>.05), though this difference was not statistically significant. Finally, many respondents expressed appreciation that our curriculum improved their ability to care for patients in complex social circumstances. Residents suggested in the free-text responses that learning more about the historical underpinnings of health disparities, opportunities for grassroots activism, and how to provide more culturally competent care of Native American populations could improve our curriculum.
Implications for Dermatology Training
Our survey results indicate that a formal SDH curriculum can improve dermatology residents’ ability to care for populations with complex social needs. We advocate for implementing SDH curricula into dermatology training programs nationwide, as has been recommended by others.13,14 We also propose that structural competency should eventually be a key dermatologic competency as determined by the Accreditation Council for Graduate Medical Education, in line with the American Medical Association’s recommendation that structural competency is a learned skill required to end health inequity.15 The Accreditation Council for Graduate Medical Education specialty program requirements currently are being revised; interested individuals can engage in this process by submitting this suggestion for public comment (https://www.acgme.org/programs-and-institutions/programs/review-and-comment/).
Limitations of a survey include the relatively small sample size (7-8 per year) and variable response rates. In addition, we did not survey each class of residents at the beginning and end of their training; our comparisons therefore were limited by comparing different individuals with distinct backgrounds and experiences. Furthermore, we acknowledge that the experience of developing this curriculum in San Francisco may be distinct from other communities, where access to dermatologic care may vary according to both the availability of public health insurance and the treatments covered by public insurers. In San Francisco, insurance coverage is near universal, such that residents in our training program regularly care for undocumented immigrants, persons experiencing homelessness, and other populations that might find it challenging to present to dermatology clinics in other settings nationwide.
Final Thoughts
Future directions of our curriculum include exploration of novel curriculum delivery methods (including a problem-based curriculum approach and other more experiential didactics), increased opportunities for community engagement, greater focus on advocacy with an emphasis on broader social and structural policies and their downstream effects, and focusing more specifically on the history and needs of specific low-income San Francisco neighborhoods and diverse patient populations.
Social drivers of health (SDH) describe the conditions in which an individual is born, grows, lives, works, and ages—all of which collectively influence their health. Examples of SDH include employment status, literacy level, education level, housing status, food access, income level, and social cohesion. Social drivers of health are critical catalysts to attaining health equity. Effectively applying an understanding of how SDH affect the care of all patients is an essential competency for physicians practicing in the modern era of rising income inequality and housing instability and increasing racial, ethnic, language, religious, and cultural diversity in the United States; however, in dermatology residency, this skill set often is developed by the hidden curriculum (ie, the informal curriculum that is based on what patient scenarios a resident happens to face) rather than one represented by formal educational objectives.2
Adding to this challenge of limited formal curricula is that caring for minoritized, marginalized, and other populations facing specific barriers can evoke feelings of frustration, helplessness, and even anger. These feelings can test the limits of a physician’s identity as a healer, leading to burnout and self-protective attitudes such as distancing (emotionally, physically, or both) from these patients.3 This is particularly relevant given that the majority (76%-79% each year from 2007-2019) of medical student matriculants come from families with incomes in the top 2 quintiles nationwide, and fewer than 6% come from the lowest quintile earners.4,5 These data indicate that most trainees have not experienced (and may even have a hard time imagining) the degree of economic and housing instability faced by many of their low-income patients, the care of whom disproportionately falls to large academic medical centers, which sponsor dermatology training programs.6 Many trainees may feel uncomfortable communicating across the broad range of racial, socioeconomic, linguistic, and cultural differences they encounter during training and in practice. Structured opportunities to provide care in a supervised supportive environment combined with didactics that emphasize practical, evidence-based strategies can build empathy, improve attitudes toward patients from diverse backgrounds, and strengthen self-efficacy in challenging scenarios.3
In the past decade, there has been a push toward integrating our understanding of SDH into formal medical training.7 Other specialty training programs—including psychiatry,8 internal medicine,9 pediatrics,10 and family medicine11—have incorporated these elements into their curricula and competency evaluations. In dermatology, as in other specialties, making and implementing effective, patient-centered care plans requires attention to the various social and structural drivers that may influence outcomes. Dermatologists therefore should be educated about SDH during their training programs and empowered to address the ways they affect patient care.
At the University of California San Francisco (UCSF)(San Francisco, California), our dermatology trainees care for patients in several hospital systems citywide, including a tertiary academic medical center with multiple locations, a county hospital, and a Veterans Affairs medical center. Given the diversity of patient populations across our training sites—including many racially and ethnically minoritized individuals, immigrants, patients with limited English proficiency, people experiencing homelessness, and sexual and gender diverse individuals—we identified a critical opportunity to enhance our training through formal didactics and hands-on experiences that integrate SDH into existing curricula and strengthen trainees’ ability to provide high-quality care to all patients.
Implementing an SDH Curriculum
In May 2020, UCSF dermatology faculty with an interest in SDH collaborated with departmental educational leadership to develop a formal SDH curriculum centered around 8 core learning objectives for residents (eTable 1). To achieve these objectives, we organized a 3-year didactic and experiential curriculum consisting of lectures (eTable 2), grand rounds sessions, journal clubs, and community engagement opportunities. Residents also spend 7 months during their training rotating at San Francisco’s city and county hospital (Zuckerberg San Francisco General Hospital [San Francisco, California]) where all faculty are members of the core SDH curriculum development team and where residents can put into practice many of the skills learned in formal didactics to develop patient-centered care plans for low-income patients, approximately 40% of whom have limited English proficiency.


To further center the importance of SDH and health equity in our training program, we developed a Health Equity Chief leadership role for senior dermatology residents. Each year, 2 to 4 residents volunteer for and serve in this role, wherein they work with core faculty to review and improve SDH curriculum elements. They also work to enhance community engagement opportunities for residents (eg, pathway programs aimed at diversifying the dermatology workforce by introducing historically excluded local high school and college students to dermatology as a career path) and improve dermatology trainees’ awareness of the history and health needs of the specific communities we serve in San Francisco. They also are prepared to become leaders in the field of health equity and to improve the care of diverse patient populations after residency. Our faculty curriculum leaders meet quarterly with our Health Equity Chiefs to review their individual and collective goals and strategize ways to improve learner and community engagement. Departmental funds are made available to support these efforts.
Leadership at our safety-net county hospital also developed a patient navigator position to improve our ability to care for patients with the most complex medical conditions and social needs. This role is held by a medical student taking a funded gap year and incorporates aspects of social work (eg, identifying barriers to care and connecting patients with resources such as transportation), quality improvement, and clinical research.12
Assessing Residents’ Experience of a New SDH Curriculum
Prior to curriculum implementation, we surveyed graduating UCSF dermatology residents in June 2020 to assess their familiarity with SDH and the social and medical needs of various populations facing barriers to care, their comfort level with specific challenging clinical situations, and their desire for additional training. Responses were measured using a 5-part Likert scale, with additional options for free-text response. After initiating the SDH curriculum in July 2020, we sent the same survey each year to all senior residents immediately prior to their graduation, offering a small financial incentive ($15 cash gift card) to those who completed the survey. We obtained UCSF Institutional Review Board approval to utilize these survey data to better understand and to enhance residents’ experience of the SDH curriculum.
All 8 residents invited in 2020 completed the survey assessing curriculum efficacy (100% response rate). For the 2023 and 2024 classes, data were analyzed in aggregate (n=14), with a 50% response rate. After implementation of the SDH curriculum, there was improvement in learners’ awareness of challenges faced by every patient population, from a mean (SD) of 3.12 (0.66) to 4.52 (SD, 0.69)(P<.05). Learners were more comfortable handling hypothetical clinical scenarios requiring them to identify and address specific SDH after vs before implementation of the curriculum (mean [SD], 3.5 [1.06] before vs 4.0 [1.16] after)(P>.05), though this difference was not statistically learners’ awareness of challenges faced by every patient population, from a mean (SD) of 3.12 (0.66) to 4.52 (0.69)(P<.05). Learners were more comfortable handling hypothetical clinical scenarios requiring them to identify and address specific SDH after vs before implementation of the curriculum (mean [SD], 3.5 [1.06] before vs 4.0 [1.16] after)(P>.05), though this difference was not statistically significant. Finally, many respondents expressed appreciation that our curriculum improved their ability to care for patients in complex social circumstances. Residents suggested in the free-text responses that learning more about the historical underpinnings of health disparities, opportunities for grassroots activism, and how to provide more culturally competent care of Native American populations could improve our curriculum.
Implications for Dermatology Training
Our survey results indicate that a formal SDH curriculum can improve dermatology residents’ ability to care for populations with complex social needs. We advocate for implementing SDH curricula into dermatology training programs nationwide, as has been recommended by others.13,14 We also propose that structural competency should eventually be a key dermatologic competency as determined by the Accreditation Council for Graduate Medical Education, in line with the American Medical Association’s recommendation that structural competency is a learned skill required to end health inequity.15 The Accreditation Council for Graduate Medical Education specialty program requirements currently are being revised; interested individuals can engage in this process by submitting this suggestion for public comment (https://www.acgme.org/programs-and-institutions/programs/review-and-comment/).
Limitations of a survey include the relatively small sample size (7-8 per year) and variable response rates. In addition, we did not survey each class of residents at the beginning and end of their training; our comparisons therefore were limited by comparing different individuals with distinct backgrounds and experiences. Furthermore, we acknowledge that the experience of developing this curriculum in San Francisco may be distinct from other communities, where access to dermatologic care may vary according to both the availability of public health insurance and the treatments covered by public insurers. In San Francisco, insurance coverage is near universal, such that residents in our training program regularly care for undocumented immigrants, persons experiencing homelessness, and other populations that might find it challenging to present to dermatology clinics in other settings nationwide.
Final Thoughts
Future directions of our curriculum include exploration of novel curriculum delivery methods (including a problem-based curriculum approach and other more experiential didactics), increased opportunities for community engagement, greater focus on advocacy with an emphasis on broader social and structural policies and their downstream effects, and focusing more specifically on the history and needs of specific low-income San Francisco neighborhoods and diverse patient populations.
- US Department of Health and Human Services. Health Equity in Healthy People 2030. Accessed January 7, 2025. https://odphp.healtwh.gov/healthypeople/priority-areas/health-equity-healthy-people-2030
- Axelson DJ, Stull MJ, Coates WC. Social determinants of health: a missing link in emergency medicine training. AEM Educ Train. 2018;2:66-68. doi:10.1002/aet2.10056
- Brenner AM, Guerrero APS, Beresin EV, et al. Teaching medical students and residents about homelessness: complex, evidence-based, and imperative. Acad Psychiatry. 2016;40:572-575. doi:10.1007/s40596-016-0571-6
- Youngclaus J, Roskovensky L. An Updated Look at the Economic Diversity of U.S. Medical Students. American Association of Medical Colleges Analysis in Brief. 2018;18. https://www.aamc.org/media/9596/download?attachment
- Shahriar AA, Puram VV, Miller JM, et al. Socioeconomic diversity of the matriculating US medical student body by race, ethnicity, and sex, 2017-2019. JAMA Netw Open. 2022;5:E222621. doi:10.1001/jamanetworkopen.2022.2621
- Williams JC, Maxey AE, Wei ML, et al. A cross-sectional analysis of Medicaid acceptance among US dermatology residency training programs. J Am Acad Dermatol. 2022;86:453-455. doi:10.1016/j.jaad.2021.09.046
- Daniel H, Bornstein S, Kane G; Health and Public Policy Committee of the American College of Physicians. Addressing social determinants to improve patient care and promote health equity: an American College of Physicians position paper. Ann Intern Med. 2018;168:577-578. doi:10.2105/AJPH
- Hansen H, Kline N, Braslow J, et al. From cultural to structural competency—training psychiatry residents to act on social determinants of health and institutional racism. JAMA Psychiatry. 2018;75:117-118. doi:10.1001/jamapsychiatry.2017.3894
- Schmidt S, Higgins S, George M, et al. An experiential resident module for understanding social determinants of health at an academic safety-net hospital. MedEdPORTAL. 2017;26:10647. doi:10.15766/mep_2374-8265.10647
- Hoffman BD, Rose J, Best D, et al. The community pediatrics training initiative project planning tool: a practical approach to community-based advocacy. MedEdPORTAL. 2017;13:10630.
- Chrisman-Khawam L, Abdullah N, Dhoopar A. Teaching health-care trainees empathy and homelessness IQ through service learning, reflective practice, and altruistic attribution. Int J Psychiatry Med. 2017;52:245-254. doi:10.1177/0091217417730288
- Sanchez-Anguiano ME, Klufas D, Amerson E. Screening for cardiometabolic risk factors in patients with psoriasis and hidradenitis suppurativa: a pilot study in a safety net population. J Am Acad Dermatol. 2024;91:1269-1272. doi:10.1016/j.jaad.2024.07.1518
- Riley C, Vasquez R, Pritchett EN. Equipping dermatologists to address structural and social drivers of inequities—structural competency. JAMA Dermatol. 2024;160:1037-1038. doi:10.1001/jamadermatol.2024.2351
- Crawl-Bey A, Pritchett EN, Riley C. 54338 Structural competency in dermatology: a pilot curriculum for equipping residents to address structural factors that contribute to health inequity. J Am Acad Dermatol. 2024;91(3 suppl):AB318. doi:10.1016/j.jaad.2024.07.1264
- Smith TM. New competency focus involves structural factors in health. American Medical Association. December 18, 202. Accessed March 23, 2026. https://www.ama-assn.org/education/changemeded-initiative/new-competency-focus-involves-structural-factors-health
- US Department of Health and Human Services. Health Equity in Healthy People 2030. Accessed January 7, 2025. https://odphp.healtwh.gov/healthypeople/priority-areas/health-equity-healthy-people-2030
- Axelson DJ, Stull MJ, Coates WC. Social determinants of health: a missing link in emergency medicine training. AEM Educ Train. 2018;2:66-68. doi:10.1002/aet2.10056
- Brenner AM, Guerrero APS, Beresin EV, et al. Teaching medical students and residents about homelessness: complex, evidence-based, and imperative. Acad Psychiatry. 2016;40:572-575. doi:10.1007/s40596-016-0571-6
- Youngclaus J, Roskovensky L. An Updated Look at the Economic Diversity of U.S. Medical Students. American Association of Medical Colleges Analysis in Brief. 2018;18. https://www.aamc.org/media/9596/download?attachment
- Shahriar AA, Puram VV, Miller JM, et al. Socioeconomic diversity of the matriculating US medical student body by race, ethnicity, and sex, 2017-2019. JAMA Netw Open. 2022;5:E222621. doi:10.1001/jamanetworkopen.2022.2621
- Williams JC, Maxey AE, Wei ML, et al. A cross-sectional analysis of Medicaid acceptance among US dermatology residency training programs. J Am Acad Dermatol. 2022;86:453-455. doi:10.1016/j.jaad.2021.09.046
- Daniel H, Bornstein S, Kane G; Health and Public Policy Committee of the American College of Physicians. Addressing social determinants to improve patient care and promote health equity: an American College of Physicians position paper. Ann Intern Med. 2018;168:577-578. doi:10.2105/AJPH
- Hansen H, Kline N, Braslow J, et al. From cultural to structural competency—training psychiatry residents to act on social determinants of health and institutional racism. JAMA Psychiatry. 2018;75:117-118. doi:10.1001/jamapsychiatry.2017.3894
- Schmidt S, Higgins S, George M, et al. An experiential resident module for understanding social determinants of health at an academic safety-net hospital. MedEdPORTAL. 2017;26:10647. doi:10.15766/mep_2374-8265.10647
- Hoffman BD, Rose J, Best D, et al. The community pediatrics training initiative project planning tool: a practical approach to community-based advocacy. MedEdPORTAL. 2017;13:10630.
- Chrisman-Khawam L, Abdullah N, Dhoopar A. Teaching health-care trainees empathy and homelessness IQ through service learning, reflective practice, and altruistic attribution. Int J Psychiatry Med. 2017;52:245-254. doi:10.1177/0091217417730288
- Sanchez-Anguiano ME, Klufas D, Amerson E. Screening for cardiometabolic risk factors in patients with psoriasis and hidradenitis suppurativa: a pilot study in a safety net population. J Am Acad Dermatol. 2024;91:1269-1272. doi:10.1016/j.jaad.2024.07.1518
- Riley C, Vasquez R, Pritchett EN. Equipping dermatologists to address structural and social drivers of inequities—structural competency. JAMA Dermatol. 2024;160:1037-1038. doi:10.1001/jamadermatol.2024.2351
- Crawl-Bey A, Pritchett EN, Riley C. 54338 Structural competency in dermatology: a pilot curriculum for equipping residents to address structural factors that contribute to health inequity. J Am Acad Dermatol. 2024;91(3 suppl):AB318. doi:10.1016/j.jaad.2024.07.1264
- Smith TM. New competency focus involves structural factors in health. American Medical Association. December 18, 202. Accessed March 23, 2026. https://www.ama-assn.org/education/changemeded-initiative/new-competency-focus-involves-structural-factors-health
Social Drivers of Health Curriculum for Dermatology Residents: the UCSF Experience
Social Drivers of Health Curriculum for Dermatology Residents: the UCSF Experience
Practice Points
- Integrating a formal curriculum on social drivers of health, including didactics on structural racism, cultural humility, communication, and practical strategies, can help residents learn to routinely assess social needs and develop feasible patient-centered care plans.
- Classroom learning paired with experiential rotations and community engagement in safety-net hospitals can help to build empathy, practical skills, and comfort managing real-world social barriers.
- Creation of trainee leadership roles and dedicated program support (eg, departmental funding) can sustain curriculum improvements, foster advocacy skills, and diversify the workforce pipeline.
Choosing the Right Electronic Medical Record: Key Features and Considerations for Dermatology Practices
Choosing the Right Electronic Medical Record: Key Features and Considerations for Dermatology Practices
Choosing an electronic medical record (EMR) is one of the most important clinical and financial decisions a dermatology practice will make. An effective system can help streamline workflows, support high-quality patient care, and protect revenue, while the wrong choice can slow clinicians down and add to the administrative burden.
Dermatology workflows involve unique documentation, imaging, and billing needs that are not always well served by generic EMR platforms. To help guide the selection of an EMR, the following framework outlines key features and practice considerations specific to dermatology practices.
While many general EMRs offer customization, dermatology practices benefit greatly from ready-built, specialty-specific documentation tools. Key elements to evaluate include the following:
- Preconfigured dermatology templates for common conditions and procedures (eg, acne, psoriasis, melanoma, biopsies, cosmetic treatments)
- Smart-phrase libraries tailored to dermatologic language and examinations
- Ability to create, modify, and share custom templates across providers
Why It Matters—Efficient charting reduces documentation time, improves consistency, and supports accurate coding.
Dermatology is a highly visual specialty, making clinical photography and image management essential. Important capabilities of an EMR include the following:
- Easy capture, annotation, and longitudinal tracking of clinical images
- Seamless embedding of photographs directly into the patient chart
- Side-by-side comparison of current and prior images
- Secure image storage and camera integration
- Body-mapping tools to mark and track lesion locations visually
Why It Matters—A high-quality image workflow supports diagnosis, treatment planning, patient education, and medicolegal documentation.
For insurance-based practices, robust billing and revenue cycle management (RCM) tools are critical. For direct-care models, some of these items may be prioritized lower. Key features to compare include the following:
- Support for International Classification of Diseases, 10th Revision, Clinical Modification; Current Procedural Terminology; and dermatology-specific code sets
- Automated coding suggestions tied to clinical documentation
- Reviewing claims for errors and inconsistencies prior to submitting to payers’ insurance eligibility verification
- Electronic Remittance Advice/Explanation of Benefits posting and denial management workflows
- Support for cosmetic and self-pay billing
- Ability to generate superbills (itemized receipts for medical services that include International Classification of Diseases Tenth Revision and Current Procedural Terminology codes; patients can submit these directly to their insurance company for reimbursement) for direct-pay practices
Why It Matters—Strong RCM functionality protects revenue, reduces denials, and minimizes staff workload.
The most effective EMRs tightly integrate clinical charting with daily practice operations. Features to evaluate include the following:
- Integrated scheduling with color-coded calendars
- Appointment-type templates and block scheduling
- Automated patient reminders via text or email
- Support for multiprovider and multilocation practices
- Integration with outside pathology or lab services
Why It Matters—Clear and templated scheduling and practice integration help practices run more smoothly by reducing administrative workload and errors and coordinating communication between providers and even ancillary services.
Patient communication and virtual care are increasingly important in dermatology. When evaluating EMRs, compare the following:
- Built-in telehealth functionality vs third-party integrations
- Automated appointment reminders
- Patient portal features (forms, messaging, results)
- Online booking capabilities
Why It Matters—Integrated telehealth and patient communication tools improve access to care, enhance patient engagement, and streamline scheduling, messaging, and virtual visits within dermatology workflows.
Reporting capabilities support clinical quality, compliance, and business decision-making. Key reporting areas include the following:
- Clinical reports (outcomes, lesion tracking, disease management)
- Financial reports (revenue per provider, payer mix, visit types)
- Customizable or exportable reporting tools
Why It Matters—Robust reporting and analytics help dermatology practices track clinical outcomes, monitor financial performance, and make data-driven decisions to improve both patient care and practice management.
The user experience after implementation of the EMR is just as important as the software itself. Evaluate the following after the EMR is implemented:
- Initial training and onboarding resources
- Availability of dermatology-specific support teams
- Ongoing education, help centers, or user communities
- Access to dedicated implementation or success managers
Why It Matters—Strong training and support resources help ensure a smoother EMR implementation, faster staff adoption, and ongoing optimization of the system for dermatology workflows.
Finally, look beyond the sticker price. The total cost of ownership includes far more than monthly fees. Compare the following:
- Upfront costs (implementation, data migration, training)
- Subscription pricing (per provider or per user)
- Billing or RCM fees (including percentages of collections if applicable) and payment processing fees
- Costs for add-on modules (telehealth, imaging, analytics)
- Contract length and termination terms
Why It Matters—Understanding the full cost of ownership helps dermatology practices choose an EMR that fits their budget long-term while avoiding unexpected fees and contractual limitations.
There is no single “best” EMR for every dermatology practice. The right choice depends on your practice model, payer mix, clinical focus, and growth plans. By evaluating EMRs through a dermatology-specific lens and asking the right questions, you can choose a system that supports both excellent patient care and long-term practice success.
Choosing an electronic medical record (EMR) is one of the most important clinical and financial decisions a dermatology practice will make. An effective system can help streamline workflows, support high-quality patient care, and protect revenue, while the wrong choice can slow clinicians down and add to the administrative burden.
Dermatology workflows involve unique documentation, imaging, and billing needs that are not always well served by generic EMR platforms. To help guide the selection of an EMR, the following framework outlines key features and practice considerations specific to dermatology practices.
While many general EMRs offer customization, dermatology practices benefit greatly from ready-built, specialty-specific documentation tools. Key elements to evaluate include the following:
- Preconfigured dermatology templates for common conditions and procedures (eg, acne, psoriasis, melanoma, biopsies, cosmetic treatments)
- Smart-phrase libraries tailored to dermatologic language and examinations
- Ability to create, modify, and share custom templates across providers
Why It Matters—Efficient charting reduces documentation time, improves consistency, and supports accurate coding.
Dermatology is a highly visual specialty, making clinical photography and image management essential. Important capabilities of an EMR include the following:
- Easy capture, annotation, and longitudinal tracking of clinical images
- Seamless embedding of photographs directly into the patient chart
- Side-by-side comparison of current and prior images
- Secure image storage and camera integration
- Body-mapping tools to mark and track lesion locations visually
Why It Matters—A high-quality image workflow supports diagnosis, treatment planning, patient education, and medicolegal documentation.
For insurance-based practices, robust billing and revenue cycle management (RCM) tools are critical. For direct-care models, some of these items may be prioritized lower. Key features to compare include the following:
- Support for International Classification of Diseases, 10th Revision, Clinical Modification; Current Procedural Terminology; and dermatology-specific code sets
- Automated coding suggestions tied to clinical documentation
- Reviewing claims for errors and inconsistencies prior to submitting to payers’ insurance eligibility verification
- Electronic Remittance Advice/Explanation of Benefits posting and denial management workflows
- Support for cosmetic and self-pay billing
- Ability to generate superbills (itemized receipts for medical services that include International Classification of Diseases Tenth Revision and Current Procedural Terminology codes; patients can submit these directly to their insurance company for reimbursement) for direct-pay practices
Why It Matters—Strong RCM functionality protects revenue, reduces denials, and minimizes staff workload.
The most effective EMRs tightly integrate clinical charting with daily practice operations. Features to evaluate include the following:
- Integrated scheduling with color-coded calendars
- Appointment-type templates and block scheduling
- Automated patient reminders via text or email
- Support for multiprovider and multilocation practices
- Integration with outside pathology or lab services
Why It Matters—Clear and templated scheduling and practice integration help practices run more smoothly by reducing administrative workload and errors and coordinating communication between providers and even ancillary services.
Patient communication and virtual care are increasingly important in dermatology. When evaluating EMRs, compare the following:
- Built-in telehealth functionality vs third-party integrations
- Automated appointment reminders
- Patient portal features (forms, messaging, results)
- Online booking capabilities
Why It Matters—Integrated telehealth and patient communication tools improve access to care, enhance patient engagement, and streamline scheduling, messaging, and virtual visits within dermatology workflows.
Reporting capabilities support clinical quality, compliance, and business decision-making. Key reporting areas include the following:
- Clinical reports (outcomes, lesion tracking, disease management)
- Financial reports (revenue per provider, payer mix, visit types)
- Customizable or exportable reporting tools
Why It Matters—Robust reporting and analytics help dermatology practices track clinical outcomes, monitor financial performance, and make data-driven decisions to improve both patient care and practice management.
The user experience after implementation of the EMR is just as important as the software itself. Evaluate the following after the EMR is implemented:
- Initial training and onboarding resources
- Availability of dermatology-specific support teams
- Ongoing education, help centers, or user communities
- Access to dedicated implementation or success managers
Why It Matters—Strong training and support resources help ensure a smoother EMR implementation, faster staff adoption, and ongoing optimization of the system for dermatology workflows.
Finally, look beyond the sticker price. The total cost of ownership includes far more than monthly fees. Compare the following:
- Upfront costs (implementation, data migration, training)
- Subscription pricing (per provider or per user)
- Billing or RCM fees (including percentages of collections if applicable) and payment processing fees
- Costs for add-on modules (telehealth, imaging, analytics)
- Contract length and termination terms
Why It Matters—Understanding the full cost of ownership helps dermatology practices choose an EMR that fits their budget long-term while avoiding unexpected fees and contractual limitations.
There is no single “best” EMR for every dermatology practice. The right choice depends on your practice model, payer mix, clinical focus, and growth plans. By evaluating EMRs through a dermatology-specific lens and asking the right questions, you can choose a system that supports both excellent patient care and long-term practice success.
Choosing an electronic medical record (EMR) is one of the most important clinical and financial decisions a dermatology practice will make. An effective system can help streamline workflows, support high-quality patient care, and protect revenue, while the wrong choice can slow clinicians down and add to the administrative burden.
Dermatology workflows involve unique documentation, imaging, and billing needs that are not always well served by generic EMR platforms. To help guide the selection of an EMR, the following framework outlines key features and practice considerations specific to dermatology practices.
While many general EMRs offer customization, dermatology practices benefit greatly from ready-built, specialty-specific documentation tools. Key elements to evaluate include the following:
- Preconfigured dermatology templates for common conditions and procedures (eg, acne, psoriasis, melanoma, biopsies, cosmetic treatments)
- Smart-phrase libraries tailored to dermatologic language and examinations
- Ability to create, modify, and share custom templates across providers
Why It Matters—Efficient charting reduces documentation time, improves consistency, and supports accurate coding.
Dermatology is a highly visual specialty, making clinical photography and image management essential. Important capabilities of an EMR include the following:
- Easy capture, annotation, and longitudinal tracking of clinical images
- Seamless embedding of photographs directly into the patient chart
- Side-by-side comparison of current and prior images
- Secure image storage and camera integration
- Body-mapping tools to mark and track lesion locations visually
Why It Matters—A high-quality image workflow supports diagnosis, treatment planning, patient education, and medicolegal documentation.
For insurance-based practices, robust billing and revenue cycle management (RCM) tools are critical. For direct-care models, some of these items may be prioritized lower. Key features to compare include the following:
- Support for International Classification of Diseases, 10th Revision, Clinical Modification; Current Procedural Terminology; and dermatology-specific code sets
- Automated coding suggestions tied to clinical documentation
- Reviewing claims for errors and inconsistencies prior to submitting to payers’ insurance eligibility verification
- Electronic Remittance Advice/Explanation of Benefits posting and denial management workflows
- Support for cosmetic and self-pay billing
- Ability to generate superbills (itemized receipts for medical services that include International Classification of Diseases Tenth Revision and Current Procedural Terminology codes; patients can submit these directly to their insurance company for reimbursement) for direct-pay practices
Why It Matters—Strong RCM functionality protects revenue, reduces denials, and minimizes staff workload.
The most effective EMRs tightly integrate clinical charting with daily practice operations. Features to evaluate include the following:
- Integrated scheduling with color-coded calendars
- Appointment-type templates and block scheduling
- Automated patient reminders via text or email
- Support for multiprovider and multilocation practices
- Integration with outside pathology or lab services
Why It Matters—Clear and templated scheduling and practice integration help practices run more smoothly by reducing administrative workload and errors and coordinating communication between providers and even ancillary services.
Patient communication and virtual care are increasingly important in dermatology. When evaluating EMRs, compare the following:
- Built-in telehealth functionality vs third-party integrations
- Automated appointment reminders
- Patient portal features (forms, messaging, results)
- Online booking capabilities
Why It Matters—Integrated telehealth and patient communication tools improve access to care, enhance patient engagement, and streamline scheduling, messaging, and virtual visits within dermatology workflows.
Reporting capabilities support clinical quality, compliance, and business decision-making. Key reporting areas include the following:
- Clinical reports (outcomes, lesion tracking, disease management)
- Financial reports (revenue per provider, payer mix, visit types)
- Customizable or exportable reporting tools
Why It Matters—Robust reporting and analytics help dermatology practices track clinical outcomes, monitor financial performance, and make data-driven decisions to improve both patient care and practice management.
The user experience after implementation of the EMR is just as important as the software itself. Evaluate the following after the EMR is implemented:
- Initial training and onboarding resources
- Availability of dermatology-specific support teams
- Ongoing education, help centers, or user communities
- Access to dedicated implementation or success managers
Why It Matters—Strong training and support resources help ensure a smoother EMR implementation, faster staff adoption, and ongoing optimization of the system for dermatology workflows.
Finally, look beyond the sticker price. The total cost of ownership includes far more than monthly fees. Compare the following:
- Upfront costs (implementation, data migration, training)
- Subscription pricing (per provider or per user)
- Billing or RCM fees (including percentages of collections if applicable) and payment processing fees
- Costs for add-on modules (telehealth, imaging, analytics)
- Contract length and termination terms
Why It Matters—Understanding the full cost of ownership helps dermatology practices choose an EMR that fits their budget long-term while avoiding unexpected fees and contractual limitations.
There is no single “best” EMR for every dermatology practice. The right choice depends on your practice model, payer mix, clinical focus, and growth plans. By evaluating EMRs through a dermatology-specific lens and asking the right questions, you can choose a system that supports both excellent patient care and long-term practice success.
Choosing the Right Electronic Medical Record: Key Features and Considerations for Dermatology Practices
Choosing the Right Electronic Medical Record: Key Features and Considerations for Dermatology Practices
PRACTICE POINTS
- Choosing an electronic medical record (EMR) built for dermatology workflow is a critical part of practice management.
- Features of an EMR that should be evaluated include support for clinical documentation, scheduling and billing, and customer support.
- The proper EMR can reduce administrative tasks and protect practice revenue, but there is no one-size-fits-all option.
Beyond Conventional Paradigms: Rethinking the Evaluation and Management of Melasma
Beyond Conventional Paradigms: Rethinking the Evaluation and Management of Melasma
Melasma is a chronic hyperpigmentation disorder with a high recurrence rate. Advances in pathogenesis, diagnosis, and treatment have shifted dermatologic approaches for both initial management and long-term care. These advances highlight the need to move beyond conventional paradigms for melasma to adopt a more comprehensive and holistic approach to evaluation and management.
Diagnosis: Beyond the Wood Lamp
Diagnosis of melasma is primarily clinical, based on visual examination, with tools such as a Wood lamp (320-400 nm) aiding in classification as epidermal, dermal, or mixed types.1 Dermoscopy is another useful tool that commonly demonstrates a brown reticular pseudonetwork, vascular patterns, and brown clods.2 Reflectance confocal microscopy allows detailed assessment of pigment distribution across skin layers and may serve as a useful diagnostic adjunct.3 Two-photon excitation microscopy also has shown concordance with reflectance confocal microscopy in identifying key pathologic features of melasma and allows quantitative assessment of pigment burden.4,5 Biopsy remains warranted in cases when the diagnosis is unclear.6,7 These advanced tools provide additional options for noninvasive imaging of melasma, which may be useful during both diagnosis and treatment.
Pathogenesis: Beyond the Melanocyte
Recent insights into pathogenesis have shifted the view of melasma from a purely melanocyte-driven disorder to one involving complex epidermal-dermal interactions influenced by chronic UV and visible light exposure and sustained cutaneous inflammation. Evidence suggests involvement of additional cell types and structural components, including epidermal barrier dysfunction, basement membrane alterations, senescent fibroblasts, mast cell activity, vascular remodeling, and dermal solar elastosis. Senescent fibroblasts secrete melanogenic and angiogenic mediators (including stem cell factor, vascular endothelial growth factor, endothelin 1, and matrix metalloproteinases) while mast cell degranulation releases histamine, tryptase, and other proteases that drive melanocyte activation, extracellular matrix degradation, and persistent hyperpigmentation.8 New research highlights the importance of epidermal-dermal communication in melasma disease activity.8,9 Exposure to UV and visible light leads to keratinocyte injury and fibroblast dysfunction, promoting the release of inflammatory and melanogenic mediators that stimulate melanocytes and weaken the basement membrane.8 Mast cell activation and vascular signaling also have been implicated, reinforcing the contribution of inflammation and dermal remodeling to ongoing pigmentation.9 This broader understanding helps explain the difficulty of sustaining long-term remission.8,9 The multifaceted and evolving understanding of melasma’s complex pathogenesis highlights areas for future therapeutic targets beyond melanin, which may help lead to greater clearance and remission periods.
Treatment: Beyond Hydroquinone
Melasma treatment has shifted toward a longitudinal approach reflecting its chronic relapsing nature. Effective management should include combination therapy of broad-spectrum photoprotection, topical therapies, systemic therapies (when appropriate), and adjunctive procedural modalities, while emphasizing the importance of maintenance therapy and relapse prevention.10,11 Growing evidence demonstrates that UV and visible light contribute to melanogenesis and prolonged pigmentation, particularly in individuals with skin of color.12,13 Visible light, particularly within the high-energy blue wavelength spectrum (400-500 nm), induces reactive oxygen species and stimulates melanogenesis, resulting in persistent hyperpigmentation and exacerbation of melasma in darker skin types.13 Iron oxide, a mineral-based physical filter commonly used in tinted mineral sunscreens, enhances protection from visible light, thus improving melasma outcomes.12,13 In a randomized double-blind placebo-controlled trial, oral polypodium leucotomos extract, an antioxidant with photoprotective effects, improved Melasma Area and Severity Index (MASI)/modified MASI (mMASI) scores and melasma-rated quality of life measures when used as an adjunct to sunscreen with or without topical hydroquinone.14
Topical therapy serves as the mainstay of melasma treatment. Hydroquinone- and retinoid-based regimens including triple-combination therapy remain an effective gold standard for active disease; however, there still are concerns about irritant dermatitis with both as well as ochronosis with long-term use of hydroquinone.15 Recent studies highlight the efficacy of nonhydroquinone topical therapies such as azelaic acid, tranexamic acid (TXA), thiamidol, cysteamine, metformin, malassezin, 2-mercaptonicotinoyl glycine, niacinamide, kojic acid, ascorbic acid, and botanical-derived compounds, many of which demonstrate reductions in disease severity and more favorable tolerability profiles.16-19 Combinations of these ingredients can allow for addressing the various pathogenic factors of melasma beyond pigmentation, including vascularity, inflammation, and solar damage.
Systemic therapy, particularly oral TXA, has emerged as an option for patients with moderate to severe or refractory melasma. Clinical trials and observational studies demonstrate improvement with oral TXA; however, relapse after discontinuation is common, emphasizing the importance of careful patient selection, established treatment duration, and concurrent maintenance therapy.16,20 Although it is well tolerated, adverse effects include gastrointestinal discomfort, menstrual irregularities, and headache. Additionally, prescreening for contraindications and clotting risk factors is needed to avoid thromboembolic events.21
Procedural interventions are best used as adjunctive therapies rather than primary treatment. Superficial and medium peels can be used depending on baseline skin type.22 Platelet-rich plasma is a promising adjunctive therapy for melasma, demonstrating reductions in MASI scores following intradermal or microneedling-assisted delivery, likely mediated through growth factor–driven inhibition of melanogenesis and modulation of tyrosinase activity. Platelet-rich plasma promotes dermal remodeling through angiogenesis, collagen synthesis, and extracellular matrix production improving skin texture and pigmentation.23 Botulinum toxin A may improve pigmentation by modulating melanogenesis as well as inflammatory and vascular pathways.24
Radiofrequency microneedling provides a dermal-targeting therapeutic approach, with some evidence showing that monthly treatments can help maintain improvements achieved with conventional therapy by modulating photoaged dermal structures impacted by melanogenic signaling.25 Histopathologic correlation studies suggest that microneedling used alone or as a delivery platform alongside topical agents may be beneficial in dermal-predominant disease, while laser-based therapies should be reserved for recalcitrant cases due to the risk for postinflammatory hyperpigmentation and melasma recurrence, particularly in those with darker skin types.26 Lasers such as Q-switched 1064-nm Nd:YAG as well as nonablative lasers used in conservative settings are options for melasma treatment, including in patients with skin of color.26,27 Laser toning with serial low-fluence (<3 J/cm2), large-spot 1064-nm Nd:YAG has been used to reduce inflammation and risk for postinflammatory hyperpigmentation.28 Additionally, randomized split-face data demonstrate that picosecond 1064-nm devices achieve comparable reductions in mMASI scores to traditional nanosecond devices, and combination strategies incorporating intense pulsed light (IPL) have shown greater MASI improvement compared with IPL alone.28,29 However, IPL generally is limited to lighter skin phototypes due to the risk for postinflammatory hyperpigmentation in darker skin.30
Lastly, long-term management requires patient education on the importance of year-round maintenance therapy, consistency, and compliance. Reinforcing adherence to photoprotection, supporting barrier repair, and setting expectations regarding prognosis are essential components to optimizing melasma treatment outcomes.12
Future Insights: Beyond the Current State of Melasma Research and Treatment
Future advances in melasma management likely will focus on improving disease assessment and long-term treatment durability. Tools such as the MASI and mMASI are widely used but are limited by interobserver variability and reduced sensitivity in darker skin types, highlighting the need to incorporate objective measures such as colorimetry.10,26 There also is a need for studies that relate clinical improvement to underlying pathology. Histopathologic correlation remains limited across most therapeutic trials, and more studies should assess long-term outcomes and quality-of-life measures given the relapsing nature of melasma.26
Future interventions will continue to move past the treatment of pigment alone. Increasing recognition of vascular, inflammatory, and barrier-related contributions to melasma, as well as targeting of other cell types affected by melasma, will be important considerations in future treatment innovations.8,11 In summary, improved inclusion of patients with skin of color in clinical trials, expansion of clinical outcomes measures and objective parameters of disease evaluation, novel approaches to treatment and relapse prevention, and greater attention to patient adherence and education will be essential to achieving advancements in melasma care.12
- Sanchez NP, Pathak MA, Sato S, et al. Melasma: a clinical, light microscopic, ultrastructural, and immunofluorescence study. J Am Acad Dermatol. 1981;4:698-710.
- Bostan E, Cakir A. The dermoscopic characteristics of melasma in relation to different skin phototypes, distribution patterns and wood lamp findings: a cross-sectional study of 236 melasma lesions. Arch Dermatol Res. 2023;315:1927-1938.
- Guida S, Longo C, Ronga R, et al. Melasma and reflectance confocal microscopy: from baseline to treatment monitoring. Int J Dermatol. 2024;63:1007-1012.
- Ning X, Yang J, Ouyang H, et al. In vivo evaluation of melasma pathologic features and treatment response by 2-photon microscopy. JAMA Dermatol. 2025;161:1039-1047.
- Chen K-J, Han Y, Wang Z-Y, et al. Submicron resolution techniques: multiphoton microscopy in skin disease. Exp Dermatol. 2023;32:1613-1623.
- Philipp-Dormston WG. Melasma: a step-by-step approach towards a multimodal combination therapy. Clin Cosmet Investig Dermatol. 2024;17:1203-1216.
- Doolan B, Gupta M. Melasma. Aust J Gen Pract. 2021;50:880-885.
- Ali L, Al Niaimi F. Pathogenesis of melasma explained. Int J Dermatol. 2025;64:1201-1212.
- Artzi O, Horovitz T, Bar-Ilan E, et al. The pathogenesis of melasma and implications for treatment. J Cosmet Dermatol. 2021;20:3432-3445.
- Gan C, Rodrigues M. An update on new and existing treatments for the management of melasma. Am J Clin Dermatol. 2024;25:717-733.
- Sarkar R, Desai SR, Sinha S, et al. Delphi consensus on melasma management by international experts and pigmentary disorders society. J Eur Acad Dermatol Venereol. Published online September 25, 2025.
- Desai SR, Alexis AF, Elbuluk N, et al. Best practices in the treatment of melasma with a focus on patients with skin of color. J Am Acad Dermatol. 2024;90:269-279.
- Lim HW, Kohli I, Ruvolo E, et al. Impact of visible light on skin health: the role of antioxidants and free radical quenchers in skin protection. J Am Acad Deramtol. 2022;86(3 suppl):S27-S37.
- Goh CL, Chuah SY, Tien S, et al. Double-blind, placebo-controlled trial to evaluate the effectiveness of polypodium leucotomos extract in the treatment of melasma in Asian skin: a pilot study. J Clin Aesthet Dermatol. 2018;11:14-19.
- Mahajan VK, Patil A, Blicharz L, et al. Medical therapies for melasma. J Cosmet Dermatol. 2022;21:3707-3728.
- Suliman RS, Alhuwayshil J, Almuflehi AA, et al. Emerging topical therapies for melasma: a comparative analysis of efficacy and safety. J Dermatolog Treat. 2025;36:2591502.
- Austin E, Nguyen JK, Jagdeo J. Topical treatments for melasma: a systematic review of randomized controlled trials. J Drugs Dermatol. 2019;18:S1545961619P1156X.
- Grimes PE, Dias S, Oparaugo NC, et al. A randomized, controlled, split-face, double-blind study comparing topical malassezin to hydroquinone 4% for melasma. J Drugs Dermatol. 2026;25:25-29.
- Passeron T, Kerob D, Le Dantec G, et al. Efficacy and tolerability of a new facial 2-mercaptonicotinoyl glycine-containing depigmenting serum versus hydroquinone 4% over 3-month treatment of facial melasma. Dermatol Ther (Heidelb). 2025;15:2379-2390.
- McKesey J, Tovar-Garza A, Pandya AG. Melasma treatment: an evidence-based review. Am J Clin Dermatol. 2020;21:173-225.
- Bala HR, Lee S, Wong C, et al. Oral tranexamic acid for the treatment of melasma: a review. Dermatol Surg. 2018;44:814-825.
- Sarkar R, Katoch S. Chemical peels in treatment of melasma. Dermatol Clin. 2024;42:21-32.
- Sarkar R, Gupta M. Platelet-rich plasma in melasma—a systematic review. Dermatol Surg. 2022;48:131-134.
- Pongklaokam J, Manuskiatti W, Wanitphakdeedecha R, et al. Efficacy and safety of letibotulinum toxin A for the treatment of melasma in two different dilutions: a randomized double-blind split-face study. Toxins (Basel). 2025;17:349.
- Han HJ, Kim JC, Park YJ, et al. Targeting the dermis for melasma maintenance treatment. Sci Rep. 2024;14:949.
- Zhang AD, Lazar M, Akhundova E, et al. A scoping review on melasma treatments and their histopathologic correlates. Dermatopathology (Basel). 2025;12:13.
- Goldman MP, Wu DC, Khetarpal S, et al. Dual 1550-nm erbium glass fiber and 1927-nm thulium fiber non-ablative fractional laser system treatment in patients with skin of color: a review of clinical studies and unmet needs. Lasers Surg Med. Published online February 10, 2026.
- Chen J, Yu N, Peng L, et al. Efficacy of low-fluence 1064 nm Q-switched Nd: YAG laser for the treatment of melasma: a meta-analysis and systematic review. J Cosmet Dermatol. 2022;21.
- Feng J, Huang L. Comparison of picosecond and nanosecond Nd:YAG 1064-nm lasers in the treatment of melasma: a split-face randomized clinical trial. Plast Reconstr Surg. 2023;151:772-777.
- Passeron T, Genedy R, Salah L, et al., Laser treatment of hyperpigmented lesions: position statement of the European Society of Laser in Dermatology. J Eur Acad Dermatol Venereol. 2019;33:987-1005.
Melasma is a chronic hyperpigmentation disorder with a high recurrence rate. Advances in pathogenesis, diagnosis, and treatment have shifted dermatologic approaches for both initial management and long-term care. These advances highlight the need to move beyond conventional paradigms for melasma to adopt a more comprehensive and holistic approach to evaluation and management.
Diagnosis: Beyond the Wood Lamp
Diagnosis of melasma is primarily clinical, based on visual examination, with tools such as a Wood lamp (320-400 nm) aiding in classification as epidermal, dermal, or mixed types.1 Dermoscopy is another useful tool that commonly demonstrates a brown reticular pseudonetwork, vascular patterns, and brown clods.2 Reflectance confocal microscopy allows detailed assessment of pigment distribution across skin layers and may serve as a useful diagnostic adjunct.3 Two-photon excitation microscopy also has shown concordance with reflectance confocal microscopy in identifying key pathologic features of melasma and allows quantitative assessment of pigment burden.4,5 Biopsy remains warranted in cases when the diagnosis is unclear.6,7 These advanced tools provide additional options for noninvasive imaging of melasma, which may be useful during both diagnosis and treatment.
Pathogenesis: Beyond the Melanocyte
Recent insights into pathogenesis have shifted the view of melasma from a purely melanocyte-driven disorder to one involving complex epidermal-dermal interactions influenced by chronic UV and visible light exposure and sustained cutaneous inflammation. Evidence suggests involvement of additional cell types and structural components, including epidermal barrier dysfunction, basement membrane alterations, senescent fibroblasts, mast cell activity, vascular remodeling, and dermal solar elastosis. Senescent fibroblasts secrete melanogenic and angiogenic mediators (including stem cell factor, vascular endothelial growth factor, endothelin 1, and matrix metalloproteinases) while mast cell degranulation releases histamine, tryptase, and other proteases that drive melanocyte activation, extracellular matrix degradation, and persistent hyperpigmentation.8 New research highlights the importance of epidermal-dermal communication in melasma disease activity.8,9 Exposure to UV and visible light leads to keratinocyte injury and fibroblast dysfunction, promoting the release of inflammatory and melanogenic mediators that stimulate melanocytes and weaken the basement membrane.8 Mast cell activation and vascular signaling also have been implicated, reinforcing the contribution of inflammation and dermal remodeling to ongoing pigmentation.9 This broader understanding helps explain the difficulty of sustaining long-term remission.8,9 The multifaceted and evolving understanding of melasma’s complex pathogenesis highlights areas for future therapeutic targets beyond melanin, which may help lead to greater clearance and remission periods.
Treatment: Beyond Hydroquinone
Melasma treatment has shifted toward a longitudinal approach reflecting its chronic relapsing nature. Effective management should include combination therapy of broad-spectrum photoprotection, topical therapies, systemic therapies (when appropriate), and adjunctive procedural modalities, while emphasizing the importance of maintenance therapy and relapse prevention.10,11 Growing evidence demonstrates that UV and visible light contribute to melanogenesis and prolonged pigmentation, particularly in individuals with skin of color.12,13 Visible light, particularly within the high-energy blue wavelength spectrum (400-500 nm), induces reactive oxygen species and stimulates melanogenesis, resulting in persistent hyperpigmentation and exacerbation of melasma in darker skin types.13 Iron oxide, a mineral-based physical filter commonly used in tinted mineral sunscreens, enhances protection from visible light, thus improving melasma outcomes.12,13 In a randomized double-blind placebo-controlled trial, oral polypodium leucotomos extract, an antioxidant with photoprotective effects, improved Melasma Area and Severity Index (MASI)/modified MASI (mMASI) scores and melasma-rated quality of life measures when used as an adjunct to sunscreen with or without topical hydroquinone.14
Topical therapy serves as the mainstay of melasma treatment. Hydroquinone- and retinoid-based regimens including triple-combination therapy remain an effective gold standard for active disease; however, there still are concerns about irritant dermatitis with both as well as ochronosis with long-term use of hydroquinone.15 Recent studies highlight the efficacy of nonhydroquinone topical therapies such as azelaic acid, tranexamic acid (TXA), thiamidol, cysteamine, metformin, malassezin, 2-mercaptonicotinoyl glycine, niacinamide, kojic acid, ascorbic acid, and botanical-derived compounds, many of which demonstrate reductions in disease severity and more favorable tolerability profiles.16-19 Combinations of these ingredients can allow for addressing the various pathogenic factors of melasma beyond pigmentation, including vascularity, inflammation, and solar damage.
Systemic therapy, particularly oral TXA, has emerged as an option for patients with moderate to severe or refractory melasma. Clinical trials and observational studies demonstrate improvement with oral TXA; however, relapse after discontinuation is common, emphasizing the importance of careful patient selection, established treatment duration, and concurrent maintenance therapy.16,20 Although it is well tolerated, adverse effects include gastrointestinal discomfort, menstrual irregularities, and headache. Additionally, prescreening for contraindications and clotting risk factors is needed to avoid thromboembolic events.21
Procedural interventions are best used as adjunctive therapies rather than primary treatment. Superficial and medium peels can be used depending on baseline skin type.22 Platelet-rich plasma is a promising adjunctive therapy for melasma, demonstrating reductions in MASI scores following intradermal or microneedling-assisted delivery, likely mediated through growth factor–driven inhibition of melanogenesis and modulation of tyrosinase activity. Platelet-rich plasma promotes dermal remodeling through angiogenesis, collagen synthesis, and extracellular matrix production improving skin texture and pigmentation.23 Botulinum toxin A may improve pigmentation by modulating melanogenesis as well as inflammatory and vascular pathways.24
Radiofrequency microneedling provides a dermal-targeting therapeutic approach, with some evidence showing that monthly treatments can help maintain improvements achieved with conventional therapy by modulating photoaged dermal structures impacted by melanogenic signaling.25 Histopathologic correlation studies suggest that microneedling used alone or as a delivery platform alongside topical agents may be beneficial in dermal-predominant disease, while laser-based therapies should be reserved for recalcitrant cases due to the risk for postinflammatory hyperpigmentation and melasma recurrence, particularly in those with darker skin types.26 Lasers such as Q-switched 1064-nm Nd:YAG as well as nonablative lasers used in conservative settings are options for melasma treatment, including in patients with skin of color.26,27 Laser toning with serial low-fluence (<3 J/cm2), large-spot 1064-nm Nd:YAG has been used to reduce inflammation and risk for postinflammatory hyperpigmentation.28 Additionally, randomized split-face data demonstrate that picosecond 1064-nm devices achieve comparable reductions in mMASI scores to traditional nanosecond devices, and combination strategies incorporating intense pulsed light (IPL) have shown greater MASI improvement compared with IPL alone.28,29 However, IPL generally is limited to lighter skin phototypes due to the risk for postinflammatory hyperpigmentation in darker skin.30
Lastly, long-term management requires patient education on the importance of year-round maintenance therapy, consistency, and compliance. Reinforcing adherence to photoprotection, supporting barrier repair, and setting expectations regarding prognosis are essential components to optimizing melasma treatment outcomes.12
Future Insights: Beyond the Current State of Melasma Research and Treatment
Future advances in melasma management likely will focus on improving disease assessment and long-term treatment durability. Tools such as the MASI and mMASI are widely used but are limited by interobserver variability and reduced sensitivity in darker skin types, highlighting the need to incorporate objective measures such as colorimetry.10,26 There also is a need for studies that relate clinical improvement to underlying pathology. Histopathologic correlation remains limited across most therapeutic trials, and more studies should assess long-term outcomes and quality-of-life measures given the relapsing nature of melasma.26
Future interventions will continue to move past the treatment of pigment alone. Increasing recognition of vascular, inflammatory, and barrier-related contributions to melasma, as well as targeting of other cell types affected by melasma, will be important considerations in future treatment innovations.8,11 In summary, improved inclusion of patients with skin of color in clinical trials, expansion of clinical outcomes measures and objective parameters of disease evaluation, novel approaches to treatment and relapse prevention, and greater attention to patient adherence and education will be essential to achieving advancements in melasma care.12
Melasma is a chronic hyperpigmentation disorder with a high recurrence rate. Advances in pathogenesis, diagnosis, and treatment have shifted dermatologic approaches for both initial management and long-term care. These advances highlight the need to move beyond conventional paradigms for melasma to adopt a more comprehensive and holistic approach to evaluation and management.
Diagnosis: Beyond the Wood Lamp
Diagnosis of melasma is primarily clinical, based on visual examination, with tools such as a Wood lamp (320-400 nm) aiding in classification as epidermal, dermal, or mixed types.1 Dermoscopy is another useful tool that commonly demonstrates a brown reticular pseudonetwork, vascular patterns, and brown clods.2 Reflectance confocal microscopy allows detailed assessment of pigment distribution across skin layers and may serve as a useful diagnostic adjunct.3 Two-photon excitation microscopy also has shown concordance with reflectance confocal microscopy in identifying key pathologic features of melasma and allows quantitative assessment of pigment burden.4,5 Biopsy remains warranted in cases when the diagnosis is unclear.6,7 These advanced tools provide additional options for noninvasive imaging of melasma, which may be useful during both diagnosis and treatment.
Pathogenesis: Beyond the Melanocyte
Recent insights into pathogenesis have shifted the view of melasma from a purely melanocyte-driven disorder to one involving complex epidermal-dermal interactions influenced by chronic UV and visible light exposure and sustained cutaneous inflammation. Evidence suggests involvement of additional cell types and structural components, including epidermal barrier dysfunction, basement membrane alterations, senescent fibroblasts, mast cell activity, vascular remodeling, and dermal solar elastosis. Senescent fibroblasts secrete melanogenic and angiogenic mediators (including stem cell factor, vascular endothelial growth factor, endothelin 1, and matrix metalloproteinases) while mast cell degranulation releases histamine, tryptase, and other proteases that drive melanocyte activation, extracellular matrix degradation, and persistent hyperpigmentation.8 New research highlights the importance of epidermal-dermal communication in melasma disease activity.8,9 Exposure to UV and visible light leads to keratinocyte injury and fibroblast dysfunction, promoting the release of inflammatory and melanogenic mediators that stimulate melanocytes and weaken the basement membrane.8 Mast cell activation and vascular signaling also have been implicated, reinforcing the contribution of inflammation and dermal remodeling to ongoing pigmentation.9 This broader understanding helps explain the difficulty of sustaining long-term remission.8,9 The multifaceted and evolving understanding of melasma’s complex pathogenesis highlights areas for future therapeutic targets beyond melanin, which may help lead to greater clearance and remission periods.
Treatment: Beyond Hydroquinone
Melasma treatment has shifted toward a longitudinal approach reflecting its chronic relapsing nature. Effective management should include combination therapy of broad-spectrum photoprotection, topical therapies, systemic therapies (when appropriate), and adjunctive procedural modalities, while emphasizing the importance of maintenance therapy and relapse prevention.10,11 Growing evidence demonstrates that UV and visible light contribute to melanogenesis and prolonged pigmentation, particularly in individuals with skin of color.12,13 Visible light, particularly within the high-energy blue wavelength spectrum (400-500 nm), induces reactive oxygen species and stimulates melanogenesis, resulting in persistent hyperpigmentation and exacerbation of melasma in darker skin types.13 Iron oxide, a mineral-based physical filter commonly used in tinted mineral sunscreens, enhances protection from visible light, thus improving melasma outcomes.12,13 In a randomized double-blind placebo-controlled trial, oral polypodium leucotomos extract, an antioxidant with photoprotective effects, improved Melasma Area and Severity Index (MASI)/modified MASI (mMASI) scores and melasma-rated quality of life measures when used as an adjunct to sunscreen with or without topical hydroquinone.14
Topical therapy serves as the mainstay of melasma treatment. Hydroquinone- and retinoid-based regimens including triple-combination therapy remain an effective gold standard for active disease; however, there still are concerns about irritant dermatitis with both as well as ochronosis with long-term use of hydroquinone.15 Recent studies highlight the efficacy of nonhydroquinone topical therapies such as azelaic acid, tranexamic acid (TXA), thiamidol, cysteamine, metformin, malassezin, 2-mercaptonicotinoyl glycine, niacinamide, kojic acid, ascorbic acid, and botanical-derived compounds, many of which demonstrate reductions in disease severity and more favorable tolerability profiles.16-19 Combinations of these ingredients can allow for addressing the various pathogenic factors of melasma beyond pigmentation, including vascularity, inflammation, and solar damage.
Systemic therapy, particularly oral TXA, has emerged as an option for patients with moderate to severe or refractory melasma. Clinical trials and observational studies demonstrate improvement with oral TXA; however, relapse after discontinuation is common, emphasizing the importance of careful patient selection, established treatment duration, and concurrent maintenance therapy.16,20 Although it is well tolerated, adverse effects include gastrointestinal discomfort, menstrual irregularities, and headache. Additionally, prescreening for contraindications and clotting risk factors is needed to avoid thromboembolic events.21
Procedural interventions are best used as adjunctive therapies rather than primary treatment. Superficial and medium peels can be used depending on baseline skin type.22 Platelet-rich plasma is a promising adjunctive therapy for melasma, demonstrating reductions in MASI scores following intradermal or microneedling-assisted delivery, likely mediated through growth factor–driven inhibition of melanogenesis and modulation of tyrosinase activity. Platelet-rich plasma promotes dermal remodeling through angiogenesis, collagen synthesis, and extracellular matrix production improving skin texture and pigmentation.23 Botulinum toxin A may improve pigmentation by modulating melanogenesis as well as inflammatory and vascular pathways.24
Radiofrequency microneedling provides a dermal-targeting therapeutic approach, with some evidence showing that monthly treatments can help maintain improvements achieved with conventional therapy by modulating photoaged dermal structures impacted by melanogenic signaling.25 Histopathologic correlation studies suggest that microneedling used alone or as a delivery platform alongside topical agents may be beneficial in dermal-predominant disease, while laser-based therapies should be reserved for recalcitrant cases due to the risk for postinflammatory hyperpigmentation and melasma recurrence, particularly in those with darker skin types.26 Lasers such as Q-switched 1064-nm Nd:YAG as well as nonablative lasers used in conservative settings are options for melasma treatment, including in patients with skin of color.26,27 Laser toning with serial low-fluence (<3 J/cm2), large-spot 1064-nm Nd:YAG has been used to reduce inflammation and risk for postinflammatory hyperpigmentation.28 Additionally, randomized split-face data demonstrate that picosecond 1064-nm devices achieve comparable reductions in mMASI scores to traditional nanosecond devices, and combination strategies incorporating intense pulsed light (IPL) have shown greater MASI improvement compared with IPL alone.28,29 However, IPL generally is limited to lighter skin phototypes due to the risk for postinflammatory hyperpigmentation in darker skin.30
Lastly, long-term management requires patient education on the importance of year-round maintenance therapy, consistency, and compliance. Reinforcing adherence to photoprotection, supporting barrier repair, and setting expectations regarding prognosis are essential components to optimizing melasma treatment outcomes.12
Future Insights: Beyond the Current State of Melasma Research and Treatment
Future advances in melasma management likely will focus on improving disease assessment and long-term treatment durability. Tools such as the MASI and mMASI are widely used but are limited by interobserver variability and reduced sensitivity in darker skin types, highlighting the need to incorporate objective measures such as colorimetry.10,26 There also is a need for studies that relate clinical improvement to underlying pathology. Histopathologic correlation remains limited across most therapeutic trials, and more studies should assess long-term outcomes and quality-of-life measures given the relapsing nature of melasma.26
Future interventions will continue to move past the treatment of pigment alone. Increasing recognition of vascular, inflammatory, and barrier-related contributions to melasma, as well as targeting of other cell types affected by melasma, will be important considerations in future treatment innovations.8,11 In summary, improved inclusion of patients with skin of color in clinical trials, expansion of clinical outcomes measures and objective parameters of disease evaluation, novel approaches to treatment and relapse prevention, and greater attention to patient adherence and education will be essential to achieving advancements in melasma care.12
- Sanchez NP, Pathak MA, Sato S, et al. Melasma: a clinical, light microscopic, ultrastructural, and immunofluorescence study. J Am Acad Dermatol. 1981;4:698-710.
- Bostan E, Cakir A. The dermoscopic characteristics of melasma in relation to different skin phototypes, distribution patterns and wood lamp findings: a cross-sectional study of 236 melasma lesions. Arch Dermatol Res. 2023;315:1927-1938.
- Guida S, Longo C, Ronga R, et al. Melasma and reflectance confocal microscopy: from baseline to treatment monitoring. Int J Dermatol. 2024;63:1007-1012.
- Ning X, Yang J, Ouyang H, et al. In vivo evaluation of melasma pathologic features and treatment response by 2-photon microscopy. JAMA Dermatol. 2025;161:1039-1047.
- Chen K-J, Han Y, Wang Z-Y, et al. Submicron resolution techniques: multiphoton microscopy in skin disease. Exp Dermatol. 2023;32:1613-1623.
- Philipp-Dormston WG. Melasma: a step-by-step approach towards a multimodal combination therapy. Clin Cosmet Investig Dermatol. 2024;17:1203-1216.
- Doolan B, Gupta M. Melasma. Aust J Gen Pract. 2021;50:880-885.
- Ali L, Al Niaimi F. Pathogenesis of melasma explained. Int J Dermatol. 2025;64:1201-1212.
- Artzi O, Horovitz T, Bar-Ilan E, et al. The pathogenesis of melasma and implications for treatment. J Cosmet Dermatol. 2021;20:3432-3445.
- Gan C, Rodrigues M. An update on new and existing treatments for the management of melasma. Am J Clin Dermatol. 2024;25:717-733.
- Sarkar R, Desai SR, Sinha S, et al. Delphi consensus on melasma management by international experts and pigmentary disorders society. J Eur Acad Dermatol Venereol. Published online September 25, 2025.
- Desai SR, Alexis AF, Elbuluk N, et al. Best practices in the treatment of melasma with a focus on patients with skin of color. J Am Acad Dermatol. 2024;90:269-279.
- Lim HW, Kohli I, Ruvolo E, et al. Impact of visible light on skin health: the role of antioxidants and free radical quenchers in skin protection. J Am Acad Deramtol. 2022;86(3 suppl):S27-S37.
- Goh CL, Chuah SY, Tien S, et al. Double-blind, placebo-controlled trial to evaluate the effectiveness of polypodium leucotomos extract in the treatment of melasma in Asian skin: a pilot study. J Clin Aesthet Dermatol. 2018;11:14-19.
- Mahajan VK, Patil A, Blicharz L, et al. Medical therapies for melasma. J Cosmet Dermatol. 2022;21:3707-3728.
- Suliman RS, Alhuwayshil J, Almuflehi AA, et al. Emerging topical therapies for melasma: a comparative analysis of efficacy and safety. J Dermatolog Treat. 2025;36:2591502.
- Austin E, Nguyen JK, Jagdeo J. Topical treatments for melasma: a systematic review of randomized controlled trials. J Drugs Dermatol. 2019;18:S1545961619P1156X.
- Grimes PE, Dias S, Oparaugo NC, et al. A randomized, controlled, split-face, double-blind study comparing topical malassezin to hydroquinone 4% for melasma. J Drugs Dermatol. 2026;25:25-29.
- Passeron T, Kerob D, Le Dantec G, et al. Efficacy and tolerability of a new facial 2-mercaptonicotinoyl glycine-containing depigmenting serum versus hydroquinone 4% over 3-month treatment of facial melasma. Dermatol Ther (Heidelb). 2025;15:2379-2390.
- McKesey J, Tovar-Garza A, Pandya AG. Melasma treatment: an evidence-based review. Am J Clin Dermatol. 2020;21:173-225.
- Bala HR, Lee S, Wong C, et al. Oral tranexamic acid for the treatment of melasma: a review. Dermatol Surg. 2018;44:814-825.
- Sarkar R, Katoch S. Chemical peels in treatment of melasma. Dermatol Clin. 2024;42:21-32.
- Sarkar R, Gupta M. Platelet-rich plasma in melasma—a systematic review. Dermatol Surg. 2022;48:131-134.
- Pongklaokam J, Manuskiatti W, Wanitphakdeedecha R, et al. Efficacy and safety of letibotulinum toxin A for the treatment of melasma in two different dilutions: a randomized double-blind split-face study. Toxins (Basel). 2025;17:349.
- Han HJ, Kim JC, Park YJ, et al. Targeting the dermis for melasma maintenance treatment. Sci Rep. 2024;14:949.
- Zhang AD, Lazar M, Akhundova E, et al. A scoping review on melasma treatments and their histopathologic correlates. Dermatopathology (Basel). 2025;12:13.
- Goldman MP, Wu DC, Khetarpal S, et al. Dual 1550-nm erbium glass fiber and 1927-nm thulium fiber non-ablative fractional laser system treatment in patients with skin of color: a review of clinical studies and unmet needs. Lasers Surg Med. Published online February 10, 2026.
- Chen J, Yu N, Peng L, et al. Efficacy of low-fluence 1064 nm Q-switched Nd: YAG laser for the treatment of melasma: a meta-analysis and systematic review. J Cosmet Dermatol. 2022;21.
- Feng J, Huang L. Comparison of picosecond and nanosecond Nd:YAG 1064-nm lasers in the treatment of melasma: a split-face randomized clinical trial. Plast Reconstr Surg. 2023;151:772-777.
- Passeron T, Genedy R, Salah L, et al., Laser treatment of hyperpigmented lesions: position statement of the European Society of Laser in Dermatology. J Eur Acad Dermatol Venereol. 2019;33:987-1005.
- Sanchez NP, Pathak MA, Sato S, et al. Melasma: a clinical, light microscopic, ultrastructural, and immunofluorescence study. J Am Acad Dermatol. 1981;4:698-710.
- Bostan E, Cakir A. The dermoscopic characteristics of melasma in relation to different skin phototypes, distribution patterns and wood lamp findings: a cross-sectional study of 236 melasma lesions. Arch Dermatol Res. 2023;315:1927-1938.
- Guida S, Longo C, Ronga R, et al. Melasma and reflectance confocal microscopy: from baseline to treatment monitoring. Int J Dermatol. 2024;63:1007-1012.
- Ning X, Yang J, Ouyang H, et al. In vivo evaluation of melasma pathologic features and treatment response by 2-photon microscopy. JAMA Dermatol. 2025;161:1039-1047.
- Chen K-J, Han Y, Wang Z-Y, et al. Submicron resolution techniques: multiphoton microscopy in skin disease. Exp Dermatol. 2023;32:1613-1623.
- Philipp-Dormston WG. Melasma: a step-by-step approach towards a multimodal combination therapy. Clin Cosmet Investig Dermatol. 2024;17:1203-1216.
- Doolan B, Gupta M. Melasma. Aust J Gen Pract. 2021;50:880-885.
- Ali L, Al Niaimi F. Pathogenesis of melasma explained. Int J Dermatol. 2025;64:1201-1212.
- Artzi O, Horovitz T, Bar-Ilan E, et al. The pathogenesis of melasma and implications for treatment. J Cosmet Dermatol. 2021;20:3432-3445.
- Gan C, Rodrigues M. An update on new and existing treatments for the management of melasma. Am J Clin Dermatol. 2024;25:717-733.
- Sarkar R, Desai SR, Sinha S, et al. Delphi consensus on melasma management by international experts and pigmentary disorders society. J Eur Acad Dermatol Venereol. Published online September 25, 2025.
- Desai SR, Alexis AF, Elbuluk N, et al. Best practices in the treatment of melasma with a focus on patients with skin of color. J Am Acad Dermatol. 2024;90:269-279.
- Lim HW, Kohli I, Ruvolo E, et al. Impact of visible light on skin health: the role of antioxidants and free radical quenchers in skin protection. J Am Acad Deramtol. 2022;86(3 suppl):S27-S37.
- Goh CL, Chuah SY, Tien S, et al. Double-blind, placebo-controlled trial to evaluate the effectiveness of polypodium leucotomos extract in the treatment of melasma in Asian skin: a pilot study. J Clin Aesthet Dermatol. 2018;11:14-19.
- Mahajan VK, Patil A, Blicharz L, et al. Medical therapies for melasma. J Cosmet Dermatol. 2022;21:3707-3728.
- Suliman RS, Alhuwayshil J, Almuflehi AA, et al. Emerging topical therapies for melasma: a comparative analysis of efficacy and safety. J Dermatolog Treat. 2025;36:2591502.
- Austin E, Nguyen JK, Jagdeo J. Topical treatments for melasma: a systematic review of randomized controlled trials. J Drugs Dermatol. 2019;18:S1545961619P1156X.
- Grimes PE, Dias S, Oparaugo NC, et al. A randomized, controlled, split-face, double-blind study comparing topical malassezin to hydroquinone 4% for melasma. J Drugs Dermatol. 2026;25:25-29.
- Passeron T, Kerob D, Le Dantec G, et al. Efficacy and tolerability of a new facial 2-mercaptonicotinoyl glycine-containing depigmenting serum versus hydroquinone 4% over 3-month treatment of facial melasma. Dermatol Ther (Heidelb). 2025;15:2379-2390.
- McKesey J, Tovar-Garza A, Pandya AG. Melasma treatment: an evidence-based review. Am J Clin Dermatol. 2020;21:173-225.
- Bala HR, Lee S, Wong C, et al. Oral tranexamic acid for the treatment of melasma: a review. Dermatol Surg. 2018;44:814-825.
- Sarkar R, Katoch S. Chemical peels in treatment of melasma. Dermatol Clin. 2024;42:21-32.
- Sarkar R, Gupta M. Platelet-rich plasma in melasma—a systematic review. Dermatol Surg. 2022;48:131-134.
- Pongklaokam J, Manuskiatti W, Wanitphakdeedecha R, et al. Efficacy and safety of letibotulinum toxin A for the treatment of melasma in two different dilutions: a randomized double-blind split-face study. Toxins (Basel). 2025;17:349.
- Han HJ, Kim JC, Park YJ, et al. Targeting the dermis for melasma maintenance treatment. Sci Rep. 2024;14:949.
- Zhang AD, Lazar M, Akhundova E, et al. A scoping review on melasma treatments and their histopathologic correlates. Dermatopathology (Basel). 2025;12:13.
- Goldman MP, Wu DC, Khetarpal S, et al. Dual 1550-nm erbium glass fiber and 1927-nm thulium fiber non-ablative fractional laser system treatment in patients with skin of color: a review of clinical studies and unmet needs. Lasers Surg Med. Published online February 10, 2026.
- Chen J, Yu N, Peng L, et al. Efficacy of low-fluence 1064 nm Q-switched Nd: YAG laser for the treatment of melasma: a meta-analysis and systematic review. J Cosmet Dermatol. 2022;21.
- Feng J, Huang L. Comparison of picosecond and nanosecond Nd:YAG 1064-nm lasers in the treatment of melasma: a split-face randomized clinical trial. Plast Reconstr Surg. 2023;151:772-777.
- Passeron T, Genedy R, Salah L, et al., Laser treatment of hyperpigmented lesions: position statement of the European Society of Laser in Dermatology. J Eur Acad Dermatol Venereol. 2019;33:987-1005.
Beyond Conventional Paradigms: Rethinking the Evaluation and Management of Melasma
Beyond Conventional Paradigms: Rethinking the Evaluation and Management of Melasma
Painful, Purpuric, Nodular Lesion With an Irregular Surface on the Shoulder
Painful, Purpuric, Nodular Lesion With an Irregular Surface on the Shoulder
THE DIAGNOSIS: Cutaneous Leiomyosarcoma
Based on the clinical and histopathologic findings, our patient was diagnosed with primary cutaneous leiomyosarcoma (LMS), a rare soft-tissue neoplasm that arises from smooth muscle and typically manifests as a firm pink nodule.1 The neoplasm may occur in the area of a prior traumatic injury or develop spontaneously without an identifiable cause.1-3 Cutaneous LMS represents 2% to 3% of all soft-tissue sarcomas worldwide, with an estimated incidence of 1 in 500,000 annually.1,4 Men who are in their fifth to seventh decades of life are at the highest risk for LMS.1
Histologically, cutaneous LMS can be subclassified as dermal, which has a low metastatic risk and excellent prognosis, or subcutaneous, which is associated with poorer outcomes and vascular muscle origin.1 In our case, hematoxylin and eosin staining revealed fascicles of smooth muscle fibers with hypercellularity, atypia, and mitotic figures (Figure). The neoplasm stained positive for desmin, vimentin, and smooth muscle actin and negative for SOX10, Melan-A, PRAME (preferentially expressed antigen in melanoma), CD34, and Factor XIIIa.1
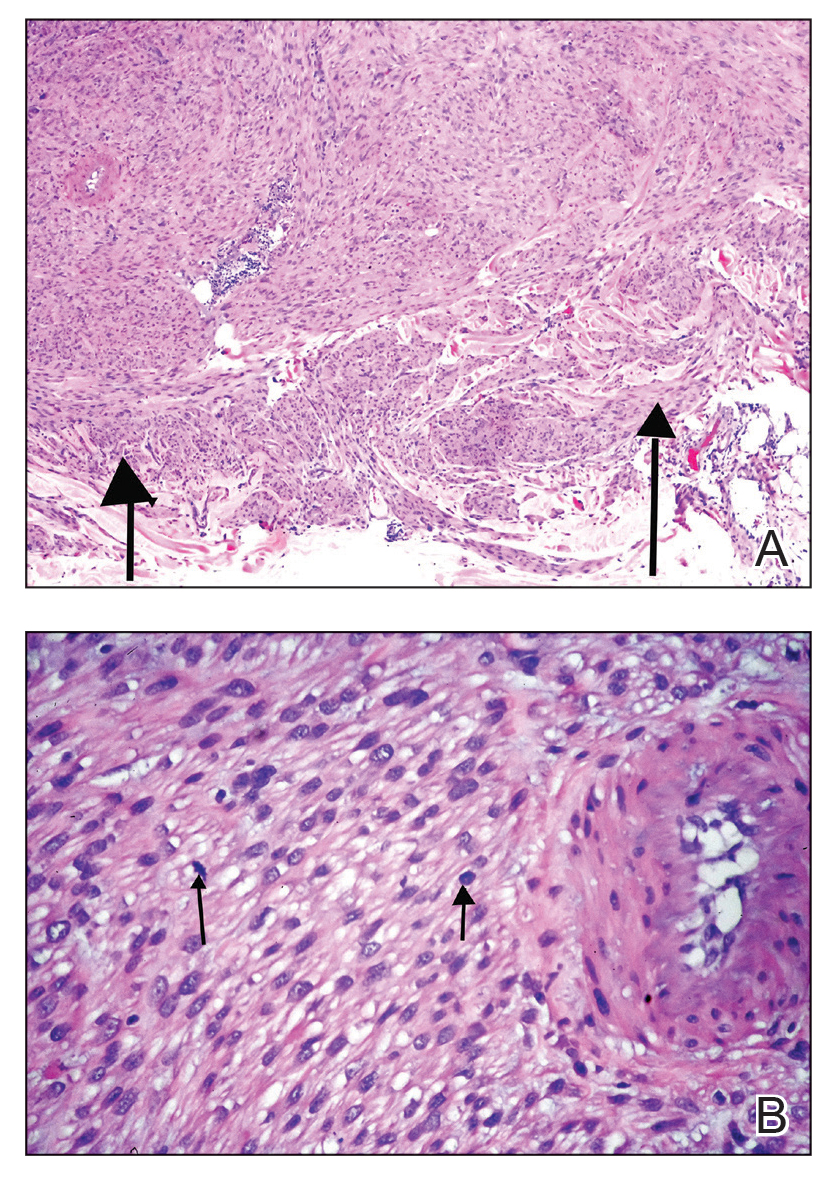
Standard treatment for LMS is surgical excision.5 Poor prognostic factors include lesions with a diameter of 5 cm or larger, deep subcutaneous tumor invasion, and distant metastases.2,5
The differential diagnosis may include dermatofibrosarcoma protuberans, which can have a similar pink nodular appearance and also may manifest after injury6; however, this lesion would stain positive for CD34 on histopathology.1 Nodular melanoma also can manifest as a solitary red, raised lesion, but it would stain positive for SOX10, PRAME, and Melan-A on histopathology.7 Basal cell carcinoma, which also may have a similar clinical appearance, is associated with nests of basaloid cells and palisading nuclei histologically.8 Lastly, atypical fibroxanthoma also manifests as a red nodule or plaque and is associated with atypical mitotic figures on histology; however, it notably stains negative for desmin.9
In summary, cutaneous LMS should be included in the differential diagnosis for raised, pink nodules. Given its nonspecific clinical presentation, this rare and malignant neoplasm requires biopsy and immunohistochemical staining for accurate diagnosis.
- Soares Queirós C, Filipe P, Soares de Almeida L. Cutaneous leiomyosarcoma: a 20-year retrospective study and review of the literature. Anais Brasileiros de Dermatologia. 2021;96:278-283. doi:10.1016/j.abd.2020.10.003
- Kim NG, Kim JO, Park YJ, et al. Cutaneous leiomyosarcoma of the face. Arch Craniofac Surg. 2017;18:145-148. doi:10.7181/acfs.2017.18.2.145
- Litaiem N, Tabka M, Nefiss M, et al. Cutaneous leiomyosarcoma mimicking arteriovenous malformation. Clin Case Rep. 2020;8:2538-2540. doi:10.1002/ccr3.3222
- Hmida L, Letaief F, Doghri R, et al. Cutaneous leiomyosarcoma on the trunk: an unusual presentation with an aggressive course - case report and review of literature. Pan Afr Med J. 2018;31:190. doi:10.11604/pamj.2018.31.190.16682
- Cazzato G, Sergi MC, Sablone S, et al. Advanced cutaneous leiomyosarcoma of the forearm. Dermatopathology (Basel). 2021;8:40-44. doi:10.3390/dermatopathology8010008
- Li Y, Wang C, Xiang B, et al. Clinical features, pathological findings and treatment of recurrent dermatofibrosarcoma protuberans. J Cancer. 2017;8:1319-1323. doi:10.7150/jca.17988
- Hernandez LE, Frech FS, Mohsin N, et al. Nodular melanoma: a review of pathogenesis, presentation, diagnosis and treatment. J Dermatol & Skin Sci. 2021;3:25-30. doi:10.29245/2767-5092/2021/3.1144
- Steele RB, Puckette Y. Basal cell carcinoma. StatPearls [Internet]. StatPearls Publishing; 2026. Updated November 7, 2025. Accessed March 3, 2026. https://www.ncbi.nlm.nih.gov/books/NBK482439/
- Kolb L, Schmieder GJ. Atypical fibroxanthoma. StatPearls [Internet]. StatPearls Publishing; 2026. Updated September 18, 2022. Accessed March 3, 2026. http://www.ncbi.nlm.nih.gov/books/NBK459342/
THE DIAGNOSIS: Cutaneous Leiomyosarcoma
Based on the clinical and histopathologic findings, our patient was diagnosed with primary cutaneous leiomyosarcoma (LMS), a rare soft-tissue neoplasm that arises from smooth muscle and typically manifests as a firm pink nodule.1 The neoplasm may occur in the area of a prior traumatic injury or develop spontaneously without an identifiable cause.1-3 Cutaneous LMS represents 2% to 3% of all soft-tissue sarcomas worldwide, with an estimated incidence of 1 in 500,000 annually.1,4 Men who are in their fifth to seventh decades of life are at the highest risk for LMS.1
Histologically, cutaneous LMS can be subclassified as dermal, which has a low metastatic risk and excellent prognosis, or subcutaneous, which is associated with poorer outcomes and vascular muscle origin.1 In our case, hematoxylin and eosin staining revealed fascicles of smooth muscle fibers with hypercellularity, atypia, and mitotic figures (Figure). The neoplasm stained positive for desmin, vimentin, and smooth muscle actin and negative for SOX10, Melan-A, PRAME (preferentially expressed antigen in melanoma), CD34, and Factor XIIIa.1

Standard treatment for LMS is surgical excision.5 Poor prognostic factors include lesions with a diameter of 5 cm or larger, deep subcutaneous tumor invasion, and distant metastases.2,5
The differential diagnosis may include dermatofibrosarcoma protuberans, which can have a similar pink nodular appearance and also may manifest after injury6; however, this lesion would stain positive for CD34 on histopathology.1 Nodular melanoma also can manifest as a solitary red, raised lesion, but it would stain positive for SOX10, PRAME, and Melan-A on histopathology.7 Basal cell carcinoma, which also may have a similar clinical appearance, is associated with nests of basaloid cells and palisading nuclei histologically.8 Lastly, atypical fibroxanthoma also manifests as a red nodule or plaque and is associated with atypical mitotic figures on histology; however, it notably stains negative for desmin.9
In summary, cutaneous LMS should be included in the differential diagnosis for raised, pink nodules. Given its nonspecific clinical presentation, this rare and malignant neoplasm requires biopsy and immunohistochemical staining for accurate diagnosis.
THE DIAGNOSIS: Cutaneous Leiomyosarcoma
Based on the clinical and histopathologic findings, our patient was diagnosed with primary cutaneous leiomyosarcoma (LMS), a rare soft-tissue neoplasm that arises from smooth muscle and typically manifests as a firm pink nodule.1 The neoplasm may occur in the area of a prior traumatic injury or develop spontaneously without an identifiable cause.1-3 Cutaneous LMS represents 2% to 3% of all soft-tissue sarcomas worldwide, with an estimated incidence of 1 in 500,000 annually.1,4 Men who are in their fifth to seventh decades of life are at the highest risk for LMS.1
Histologically, cutaneous LMS can be subclassified as dermal, which has a low metastatic risk and excellent prognosis, or subcutaneous, which is associated with poorer outcomes and vascular muscle origin.1 In our case, hematoxylin and eosin staining revealed fascicles of smooth muscle fibers with hypercellularity, atypia, and mitotic figures (Figure). The neoplasm stained positive for desmin, vimentin, and smooth muscle actin and negative for SOX10, Melan-A, PRAME (preferentially expressed antigen in melanoma), CD34, and Factor XIIIa.1

Standard treatment for LMS is surgical excision.5 Poor prognostic factors include lesions with a diameter of 5 cm or larger, deep subcutaneous tumor invasion, and distant metastases.2,5
The differential diagnosis may include dermatofibrosarcoma protuberans, which can have a similar pink nodular appearance and also may manifest after injury6; however, this lesion would stain positive for CD34 on histopathology.1 Nodular melanoma also can manifest as a solitary red, raised lesion, but it would stain positive for SOX10, PRAME, and Melan-A on histopathology.7 Basal cell carcinoma, which also may have a similar clinical appearance, is associated with nests of basaloid cells and palisading nuclei histologically.8 Lastly, atypical fibroxanthoma also manifests as a red nodule or plaque and is associated with atypical mitotic figures on histology; however, it notably stains negative for desmin.9
In summary, cutaneous LMS should be included in the differential diagnosis for raised, pink nodules. Given its nonspecific clinical presentation, this rare and malignant neoplasm requires biopsy and immunohistochemical staining for accurate diagnosis.
- Soares Queirós C, Filipe P, Soares de Almeida L. Cutaneous leiomyosarcoma: a 20-year retrospective study and review of the literature. Anais Brasileiros de Dermatologia. 2021;96:278-283. doi:10.1016/j.abd.2020.10.003
- Kim NG, Kim JO, Park YJ, et al. Cutaneous leiomyosarcoma of the face. Arch Craniofac Surg. 2017;18:145-148. doi:10.7181/acfs.2017.18.2.145
- Litaiem N, Tabka M, Nefiss M, et al. Cutaneous leiomyosarcoma mimicking arteriovenous malformation. Clin Case Rep. 2020;8:2538-2540. doi:10.1002/ccr3.3222
- Hmida L, Letaief F, Doghri R, et al. Cutaneous leiomyosarcoma on the trunk: an unusual presentation with an aggressive course - case report and review of literature. Pan Afr Med J. 2018;31:190. doi:10.11604/pamj.2018.31.190.16682
- Cazzato G, Sergi MC, Sablone S, et al. Advanced cutaneous leiomyosarcoma of the forearm. Dermatopathology (Basel). 2021;8:40-44. doi:10.3390/dermatopathology8010008
- Li Y, Wang C, Xiang B, et al. Clinical features, pathological findings and treatment of recurrent dermatofibrosarcoma protuberans. J Cancer. 2017;8:1319-1323. doi:10.7150/jca.17988
- Hernandez LE, Frech FS, Mohsin N, et al. Nodular melanoma: a review of pathogenesis, presentation, diagnosis and treatment. J Dermatol & Skin Sci. 2021;3:25-30. doi:10.29245/2767-5092/2021/3.1144
- Steele RB, Puckette Y. Basal cell carcinoma. StatPearls [Internet]. StatPearls Publishing; 2026. Updated November 7, 2025. Accessed March 3, 2026. https://www.ncbi.nlm.nih.gov/books/NBK482439/
- Kolb L, Schmieder GJ. Atypical fibroxanthoma. StatPearls [Internet]. StatPearls Publishing; 2026. Updated September 18, 2022. Accessed March 3, 2026. http://www.ncbi.nlm.nih.gov/books/NBK459342/
- Soares Queirós C, Filipe P, Soares de Almeida L. Cutaneous leiomyosarcoma: a 20-year retrospective study and review of the literature. Anais Brasileiros de Dermatologia. 2021;96:278-283. doi:10.1016/j.abd.2020.10.003
- Kim NG, Kim JO, Park YJ, et al. Cutaneous leiomyosarcoma of the face. Arch Craniofac Surg. 2017;18:145-148. doi:10.7181/acfs.2017.18.2.145
- Litaiem N, Tabka M, Nefiss M, et al. Cutaneous leiomyosarcoma mimicking arteriovenous malformation. Clin Case Rep. 2020;8:2538-2540. doi:10.1002/ccr3.3222
- Hmida L, Letaief F, Doghri R, et al. Cutaneous leiomyosarcoma on the trunk: an unusual presentation with an aggressive course - case report and review of literature. Pan Afr Med J. 2018;31:190. doi:10.11604/pamj.2018.31.190.16682
- Cazzato G, Sergi MC, Sablone S, et al. Advanced cutaneous leiomyosarcoma of the forearm. Dermatopathology (Basel). 2021;8:40-44. doi:10.3390/dermatopathology8010008
- Li Y, Wang C, Xiang B, et al. Clinical features, pathological findings and treatment of recurrent dermatofibrosarcoma protuberans. J Cancer. 2017;8:1319-1323. doi:10.7150/jca.17988
- Hernandez LE, Frech FS, Mohsin N, et al. Nodular melanoma: a review of pathogenesis, presentation, diagnosis and treatment. J Dermatol & Skin Sci. 2021;3:25-30. doi:10.29245/2767-5092/2021/3.1144
- Steele RB, Puckette Y. Basal cell carcinoma. StatPearls [Internet]. StatPearls Publishing; 2026. Updated November 7, 2025. Accessed March 3, 2026. https://www.ncbi.nlm.nih.gov/books/NBK482439/
- Kolb L, Schmieder GJ. Atypical fibroxanthoma. StatPearls [Internet]. StatPearls Publishing; 2026. Updated September 18, 2022. Accessed March 3, 2026. http://www.ncbi.nlm.nih.gov/books/NBK459342/
Painful, Purpuric, Nodular Lesion With an Irregular Surface on the Shoulder
Painful, Purpuric, Nodular Lesion With an Irregular Surface on the Shoulder
A 53-year-old man presented to the dermatology clinic for evaluation of a painful, purpuric, nodular lesion on the left shoulder of 3 months’ duration. The lesion had an irregular surface that was surrounded by an erythematous ring. Biopsy revealed fascicles of eosinophilic cells within the dermis. The nuclei were heterogeneous in size and shape and had blunted ends. Frequent atypia and mitotic figures were observed, and the lesion extended into the subcutis. Immunostaining was positive for desmin and smooth muscle actin and negative for SOX10, Melan-A, PRAME (preferentially expressed antigen in melanoma), CD34, and Factor XIIIa.
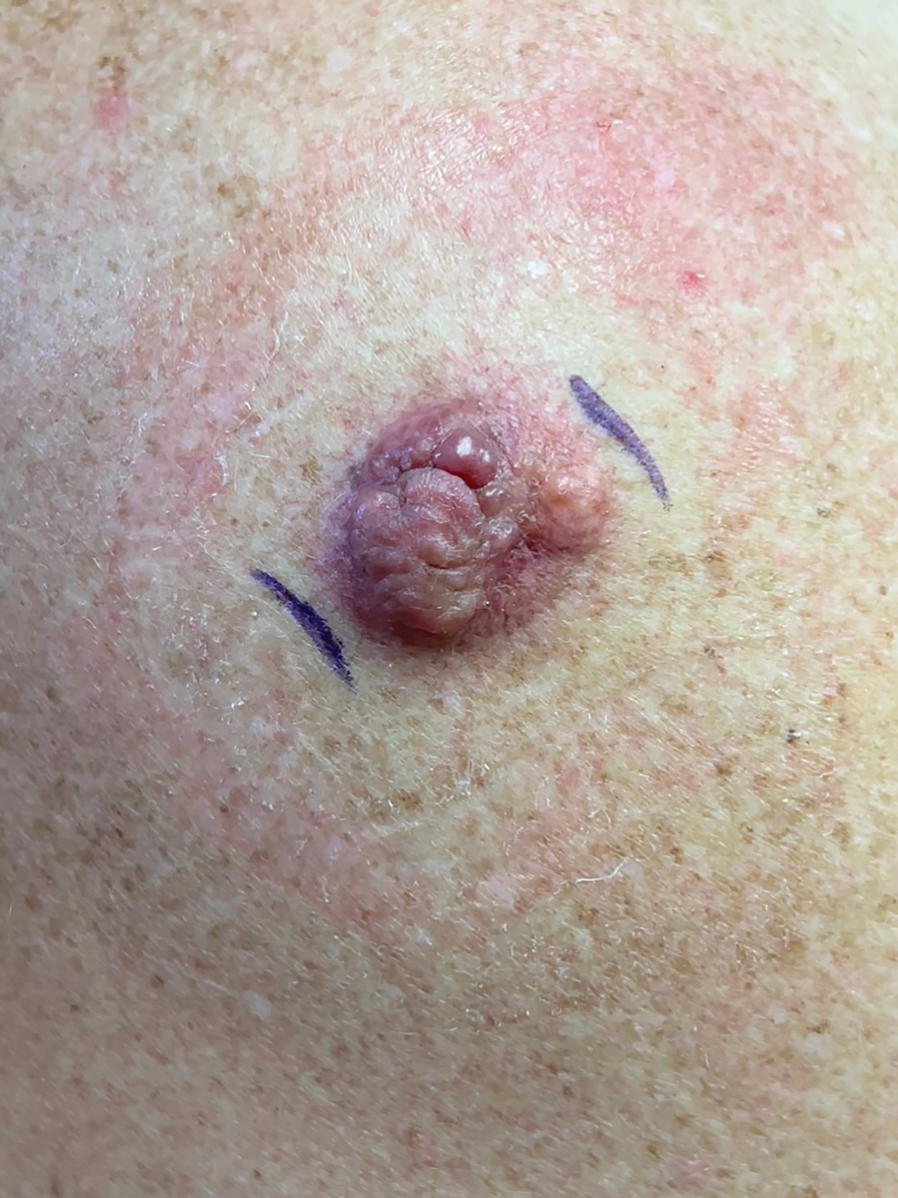
A Case of Birt-Hogg-Dubé Syndrome: A Rare but Essential Diagnosis to Consider
A Case of Birt-Hogg-Dubé Syndrome: A Rare but Essential Diagnosis to Consider
Birt-Hogg-Dubé syndrome (BHD) is an autosomal dominant disease that arises from loss-of-function mutations in the FLCN gene. FLCN encodes folliculin, which is presumed to function as a tumor suppressor, though its precise role is incompletely understood.1,2 BHD is characterized by multiple pulmonary cysts leading to recurrent spontaneous pneumothoraces, cutaneous lesions—specifically fibrofolliculomas—and an increased risk of renal malignancies. Diagnosing BHD is challenging due to the variable presentation of the disease. Some patients may only have cystic lung diseases, while others may not have characteristic skin lesions.3-5 It is important to maintain awareness of BHD, especially when the diagnosis dictates the need for genetic counseling.
Case Presentation
A male veteran in his 60s, who was a lifelong nonsmoker with a history of extensive bullous emphysema and recurrent pneumothoraces, presented to the Veterans Affairs Greater Los Angeles Healthcare System pulmonary clinic while transferring care from a separate institution.
According to the patient, the first pneumothorax episode occurred about 20 years before presentation, followed by a recurrence a few years later after he was diagnosed with emphysema. He underwent pleurodesis of the right lung during his service abroad. Another episode nearly a decade after the first pneumothorax necessitated pleurodesis of the left lung (Figure 1). The patient's family history revealed pulmonary cysts in 1 immediate family member but no history of renal tumors. Notably, his mother passed away at a young age due to tuberculosis.
On physical examination, numerous skin tags and acrochordons on the face were observed, which had been stable for > 30 years. Despite a slow decline in exercise capacity following pleurodesis, the patient could still walk multiple miles daily and climb 3 flights of stairs before needing to rest. Pulmonary function testing (PFT) showed a forced expiratory volume in 1 second (FEV1)/forced vital capacity ratio of 0.84 with reduced FEV1, total lung capacity (TLC), and diffusion capacity for carbon monoxide (DLCO), indicating a mild restrictive ventilatory defect and reduced diffusing capacity.
Laboratory results revealed a normal α-1 antitrypsin level: 133 mg/dL (reference, 83-199 mg/dL), with a Pi*MS phenotype and undetectable antinuclear antibodies. The most recent chest computed tomography (CT) in 2019, displayed paraseptal and centrilobular emphysema, scattered blebs, and scarring consistent with prior pleurodesis procedures (Figure 2).
Genetic testing for the FLCN gene revealed heterozygous pathogenic mutation: c.1285del and p.His429Thrfs*39, which confirmed the diagnosis of BHD. A shave biopsy of a postauricular papular lesion confirmed a histologic pattern of fibrofolliculoma/trichodiscoma.
Follow-up and Outcomes
After confirmation of the BHD diagnosis, the patient was referred to genetic counseling and scheduled for annual magnetic resonance imaging (MRI) of the abdomen and pelvis to screen for renal malignancies. As the patient was able to establish care with a new long-term primary care practitioner in the outpatient setting, he continues regular follow-up visits in the pulmonary clinic with stable respiratory symptoms and no recurrent pneumothoraces thus far.
Discussion
Differential Diagnoses of Cystic Pulmonary Lesions
BHD is an important differential diagnosis to consider in the presentation of diffuse cystic lung diseases. Still, 2 other crucial considerations are pulmonary Langerhans cell histiocytosis (PLCH) and lymphangioleiomyomatosis (LAM), which occur at slightly higher frequencies than BHD.6
One of the first steps in radiographically evaluating cystic lung diseases is to characterize the cysts. The Fleischner Society defines true cysts as a “round parenchymal lucency or low-attenuating area with a well-defined interface with normal lung.”7 Mimics of cystic lesions may include cavitary lung lesions, thick-walled spaces within another area of mass, nodule, or consolidation. Another mimic is a pneumatocele, a pseudocyst that lacks epithelial lining and may be secondary to bacterial pneumonia, pneumocystis infections, trauma, or prior mechanical ventilation.8After characterizing true cysts, different patterns of cystic lesions can also be associated with specific diseases. Cysts in PLCH are commonly more uniform and round, whereas the cysts in LAM may be more irregularly shaped. 9 Cysts in BHD may be larger and predominantly located in basal and paramediastinal areas.4LAM is associated with tuberous sclerosis, which can also present with skin lesions (angiofibromas) and renal tumors (angiomyolipomas), thus creating a very similar picture to BHD. Therefore, tissue biopsies of skin lesions are essential as histopathology can identify characteristic fibrofolliculomas specific to BHD. While genetic testing would also strongly support the diagnosis of BHD, it is essential to note that negative genetic testing does not rule out BHD.4Lastly, lymphoid interstitial pneumonia (LIP) is another important consideration in the differential diagnosis of cystic lung diseases. LIP presents with not only perivascular cysts and centrilobular nodules but also diffuse ground-glass attenuation.10 In contrast to BHD, LIP is associated with autoimmune diseases such as Sjögren syndrome and infectious diseases such as HIV; thus, it may be differentiated from BHD by the presence of underlying disease processes and may warrant serologic testing for potential rheumatologic disorders.
Characteristics and Diagnostic Criteria
Cystic lung disease is the most common presentation of BHD. It presents in > 80% of cases and confers a 50-fold increase in the risk of spontaneous pneumothorax compared with the general population.4,11 Recurrent pneumothoraces are observed in about 25% to 30% of patients with BHD, typically occurring between the third and fifth decades of life and at significantly decreased rates after 50 years of age.12 A spontaneous pneumothorax might serve as the initial and perhaps the sole clinical presentation for some patients with BHD, but others may present with other respiratory symptoms such as cough and exertional dyspnea. PFT results may be normal or reveal a mild restrictive ventilatory defect and reduced DLCO, as reported in a few cases.6 The management of pulmonary complications primarily revolves around reducing the risk of pneumothoraces, which includes precautions such as avoiding positive pressure ventilation and air travel. Early pleurodesis with the first occurrence of a spontaneous pneumothorax is considered in some cases.13
The distinctive dermatologic features associated with BHD include multiple white papules primarily found on the nose and face. Pathologically, these manifestations have a range of histologic distinctions, from fibrofolliculomas to benign hamartomas of the hair follicles and trichodiscomas.5 The diagnostic criteria outlined by Menko et al note that confirmation of BHD requires the presence of either ≥ 5 pathologically confirmed fibrofolliculomas or trichodiscomas, a documented pathogenic FLCN gene mutation, or the fulfillment of 2 minor criteria. These minor criteria include the presence of multiple lung cysts, early-onset renal cancer, or a first-degree relative with BHD.5
Recurrent Pneumothoraces Management
After the first episode of spontaneous pneumothorax, early pleurodesis is indicated as the risk of recurrence can be as high as 75%.4,14 Specific pleurodesis modalities have shown promising results, such as total pleural covering with cellulose mesh. In a small retrospective review, cellulose mesh demonstrated a significant reduction in the recurrence rate of pneumothorax at 7.5 years for patients with BHD compared with partial covering.15 Apart from preventing further pneumothorax episodes in the affected lung, it is also important to highlight patient education and monitoring after initial pleurodesis, as the contralateral lung is also at risk. As demonstrated in this case, the patient had received pleurodesis of his right lung but experienced another pneumothorax of his contralateral lung a few years later.
Lastly, the patient was advised to avoid air travel altogether; however, current data may suggest that air travel may not be an absolute contraindication for patients with BHD. Although the literature on this topic is limited, a retrospective study by Johannesma et al involving 158 patients with BHD surveyed on pneumothorax occurrence after air travel indicated a calculated risk of 0.63% per flight. Notably, only 3 of 13 patients with BHD and recurrent pneumothoraces after travel had undergone pleurodesis in the past.16 Therefore, counseling patients on the potential risks of air travel and allowing essential flights while diligently monitoring for symptoms during and after travel may be a reasonable, patient-centered approach in contrast to a complete restriction on air travel.
Timing to Diagnosis
Diagnosing BHD is challenging and often delayed. In a 2022 study by Steinlein et al, the average delay in BHD diagnoses in their cohort was 9.3 years, with 4 patients also diagnosed with renal malignancy during the study period.17 The difficulty in diagnosis can be attributed to the heterogeneous presentation among affected family members, some of whom may exclusively exhibit pulmonary cystic lesions without dermatologic findings.
A lack of longitudinal care for this patient may have contributed to the diagnostic delay. The patient had pneumothorax events across separate care settings and locations, and due to employment-related relocations, he often re-established care at various health care systems. This case highlights the importance of continuity of care, especially in BHD, where monitoring for renal tumors is also essential to long-term management.17,18
Renal Tumor Monitoring
Finally, once BHD is diagnosed, one of the most important considerations is to begin routine monitoring for renal malignancies. Current recommendations advise starting lifelong renal cancer screening, even as early as age 20 years, with annual MRIs, as renal ultrasound may not be sufficiently sensitive to detect smaller lesions.19 The screening interval can be extended to every 2 years for patients without a family history of renal tumors or suspicious renal lesions. If tumors are found, then nephron-sparing surgery is recommended, given the potential for the development of chronic renal insufficiency in patients with BHD.20
Conclusions
BHD is a rare and complex syndrome in which early recognition and diagnosis play a pivotal role in preventing potentially severe complications such as renal malignancies. Suspicion of a genetic disorder, such as BHD, LAM, or PLCH, should arise in patients who experience spontaneous pneumothorax, especially in the presence of multiple cystic lesions or a family history of pneumothoraces. Early consideration of pleurodesis after the first spontaneous pneumothorax is advisable. The complex presentation of BHD contributes to the difficulty of diagnosis and may delay recognition, which can be exacerbated by variable continuity of care.
- Schmidt LS, Linehan WM. Molecular genetics and clinical features of Birt-Hogg-Dubé-Syndrome. Nat Rev Urol. 2015;12:558-569. doi:10.1038/nrurol.2015.206
- Lim DHK, Rehal PK, Nahorski MS, et al. A new locus-specific database (LSDB) for mutations in the folliculin (FLCN) gene. Hum Mutat. 2010;31:E1043-1051. doi:10.1002/humu.21130
- Aivaz O, Berkman S, Middelton L, et al. Comedonal and cystic fibrofolliculomas in Birt-Hogg-Dube syndrome. JAMA Dermatology. 2015;151:770-774. doi:10.1001/jamadermatol.2015.0215
- Daccord C, Good JM, Morren MA, et al. Birt–Hogg–Dubé syndrome. Eur Respir Rev. 2020;29:200042. doi:10.1183/16000617.0042-2020
- Menko FH, van Steensel MA, Giraud S, et al. Birt-Hogg-Dubé syndrome: diagnosis and management. The Lancet Oncology. 2009;10:1199-1206. doi:10.1016/S1470-2045(09)70188-3
- Daccord C, Cottin V, Prévot G, et al. Lung function in Birt-Hogg-Dubé syndrome: a retrospective analysis of 96 patients. Orphanet J Rare Dis. 2020;15:120. doi:10.1186/s13023-020-01402-y
- Hansell DM, Bankier AA, MacMahon H, et al. Fleischner Society: glossary of terms for thoracic imaging. Radiology. 2008;246:697-722. doi:10.1148/radiol.2462070712
- Jamil A, Kasi A. Pneumatocele. In: StatPearls. StatPearls Publishing; 2024. Accessed March 2, 2026. http://www.ncbi.nlm.nih.gov/books/NBK556146/
- Bhardwaj H, Bhardwaj B. Differentiating pulmonary lymphangioleiomyomatosis from pulmonary langerhans cell histiocytosis and Birt-Hogg-Dube syndrome. Lung India. 2013;30:372-373. doi:10.4103/0970-2113.120611
- Swigris JJ, Berry GJ, Raffin TA, et al. Lymphoid interstitial pneumonia: a narrative review. Chest. 2002;122:2150-2164. doi:10.1378/chest.122.6.2150
- Zbar B, Alvord WG, Glenn G, et al. Risk of renal and colonic neoplasms and spontaneous pneumothorax in the Birt-Hogg-Dubé syndrome. Cancer Epidemiol Biomarkers Prev. 2002;11:393-400.
- Sattler EC, Steinlein OK. Delayed diagnosis of Birt-Hogg-Dubé syndrome due to marked intrafamilial clinical variability: a case report. BMC Med Genet. 2018;19:45. doi:10.1186/s12881-018-0558-0
- Gupta N, Seyama K, McCormack FX. Pulmonary manifestations of Birt-Hogg-Dubé syndrome. Fam Cancer. 2013;12:387-396. doi:10.1007/s10689-013-9660-9
- Gupta N, Kopras EJ, Henske EP, et al. Spontaneous pneumothoraces in patients with Birt–Hogg–Dubé syndrome. Ann Am Thorac Soc. 2017;14:706-713. doi:10.1513/AnnalsATS.201611-886OC
- Mizobuchi T, Kurihara M, Ebana H, et al. A total pleural covering of absorbable cellulose mesh prevents pneumothorax recurrence in patients with Birt-Hogg-Dubé syndrome. Orphanet J Rare Dis. 2018;13:78. doi:10.1186/s13023-018-0790-x
- Johannesma PC, van de Beek I, van der Wel JWT, et al. Risk of spontaneous pneumothorax due to air travel and diving in patients with Birt–Hogg–Dubé syndrome. Springerplus. 2016;5:1506. doi:10.1186/s40064-016-3009-4
- Steinlein OK, Reithmair M, Syunyaeva Z, et al. Delayed diagnosis of Birt-Hogg-Dubé syndrome might be aggravated by gender bias. eClinicalMedicine. 2022;51:101572. doi:10.1016/j.eclinm.2022.101572
- Pereira Gray DJ, Sidaway-Lee K, White E, et al. Continuity of care with doctors—a matter of life and death? A systematic review of continuity of care and mortality. BMJ Open. 2018;8:e021161. doi:10.1136/bmjopen-2017-021161
- Sattler EC, Steinlein OK. GeneReviews Birt-Hogg-Dubé syndrome. January 30, 2020. Accessed March 2, 2026. https://www.ncbi.nlm.nih.gov/books/NBK1522/table
- Stamatakis L, Metwalli AR, Middelton LA, et al. Diagnosis and management of BHD-associated kidney cancer. Fam Cancer. 2013;12:397-402. doi:10.1007/s10689-013-9657-4
Birt-Hogg-Dubé syndrome (BHD) is an autosomal dominant disease that arises from loss-of-function mutations in the FLCN gene. FLCN encodes folliculin, which is presumed to function as a tumor suppressor, though its precise role is incompletely understood.1,2 BHD is characterized by multiple pulmonary cysts leading to recurrent spontaneous pneumothoraces, cutaneous lesions—specifically fibrofolliculomas—and an increased risk of renal malignancies. Diagnosing BHD is challenging due to the variable presentation of the disease. Some patients may only have cystic lung diseases, while others may not have characteristic skin lesions.3-5 It is important to maintain awareness of BHD, especially when the diagnosis dictates the need for genetic counseling.
Case Presentation
A male veteran in his 60s, who was a lifelong nonsmoker with a history of extensive bullous emphysema and recurrent pneumothoraces, presented to the Veterans Affairs Greater Los Angeles Healthcare System pulmonary clinic while transferring care from a separate institution.
According to the patient, the first pneumothorax episode occurred about 20 years before presentation, followed by a recurrence a few years later after he was diagnosed with emphysema. He underwent pleurodesis of the right lung during his service abroad. Another episode nearly a decade after the first pneumothorax necessitated pleurodesis of the left lung (Figure 1). The patient's family history revealed pulmonary cysts in 1 immediate family member but no history of renal tumors. Notably, his mother passed away at a young age due to tuberculosis.

On physical examination, numerous skin tags and acrochordons on the face were observed, which had been stable for > 30 years. Despite a slow decline in exercise capacity following pleurodesis, the patient could still walk multiple miles daily and climb 3 flights of stairs before needing to rest. Pulmonary function testing (PFT) showed a forced expiratory volume in 1 second (FEV1)/forced vital capacity ratio of 0.84 with reduced FEV1, total lung capacity (TLC), and diffusion capacity for carbon monoxide (DLCO), indicating a mild restrictive ventilatory defect and reduced diffusing capacity.
Laboratory results revealed a normal α-1 antitrypsin level: 133 mg/dL (reference, 83-199 mg/dL), with a Pi*MS phenotype and undetectable antinuclear antibodies. The most recent chest computed tomography (CT) in 2019, displayed paraseptal and centrilobular emphysema, scattered blebs, and scarring consistent with prior pleurodesis procedures (Figure 2).

Genetic testing for the FLCN gene revealed heterozygous pathogenic mutation: c.1285del and p.His429Thrfs*39, which confirmed the diagnosis of BHD. A shave biopsy of a postauricular papular lesion confirmed a histologic pattern of fibrofolliculoma/trichodiscoma.
Follow-up and Outcomes
After confirmation of the BHD diagnosis, the patient was referred to genetic counseling and scheduled for annual magnetic resonance imaging (MRI) of the abdomen and pelvis to screen for renal malignancies. As the patient was able to establish care with a new long-term primary care practitioner in the outpatient setting, he continues regular follow-up visits in the pulmonary clinic with stable respiratory symptoms and no recurrent pneumothoraces thus far.
Discussion
Differential Diagnoses of Cystic Pulmonary Lesions
BHD is an important differential diagnosis to consider in the presentation of diffuse cystic lung diseases. Still, 2 other crucial considerations are pulmonary Langerhans cell histiocytosis (PLCH) and lymphangioleiomyomatosis (LAM), which occur at slightly higher frequencies than BHD.6
One of the first steps in radiographically evaluating cystic lung diseases is to characterize the cysts. The Fleischner Society defines true cysts as a “round parenchymal lucency or low-attenuating area with a well-defined interface with normal lung.”7 Mimics of cystic lesions may include cavitary lung lesions, thick-walled spaces within another area of mass, nodule, or consolidation. Another mimic is a pneumatocele, a pseudocyst that lacks epithelial lining and may be secondary to bacterial pneumonia, pneumocystis infections, trauma, or prior mechanical ventilation.8After characterizing true cysts, different patterns of cystic lesions can also be associated with specific diseases. Cysts in PLCH are commonly more uniform and round, whereas the cysts in LAM may be more irregularly shaped. 9 Cysts in BHD may be larger and predominantly located in basal and paramediastinal areas.4LAM is associated with tuberous sclerosis, which can also present with skin lesions (angiofibromas) and renal tumors (angiomyolipomas), thus creating a very similar picture to BHD. Therefore, tissue biopsies of skin lesions are essential as histopathology can identify characteristic fibrofolliculomas specific to BHD. While genetic testing would also strongly support the diagnosis of BHD, it is essential to note that negative genetic testing does not rule out BHD.4Lastly, lymphoid interstitial pneumonia (LIP) is another important consideration in the differential diagnosis of cystic lung diseases. LIP presents with not only perivascular cysts and centrilobular nodules but also diffuse ground-glass attenuation.10 In contrast to BHD, LIP is associated with autoimmune diseases such as Sjögren syndrome and infectious diseases such as HIV; thus, it may be differentiated from BHD by the presence of underlying disease processes and may warrant serologic testing for potential rheumatologic disorders.
Characteristics and Diagnostic Criteria
Cystic lung disease is the most common presentation of BHD. It presents in > 80% of cases and confers a 50-fold increase in the risk of spontaneous pneumothorax compared with the general population.4,11 Recurrent pneumothoraces are observed in about 25% to 30% of patients with BHD, typically occurring between the third and fifth decades of life and at significantly decreased rates after 50 years of age.12 A spontaneous pneumothorax might serve as the initial and perhaps the sole clinical presentation for some patients with BHD, but others may present with other respiratory symptoms such as cough and exertional dyspnea. PFT results may be normal or reveal a mild restrictive ventilatory defect and reduced DLCO, as reported in a few cases.6 The management of pulmonary complications primarily revolves around reducing the risk of pneumothoraces, which includes precautions such as avoiding positive pressure ventilation and air travel. Early pleurodesis with the first occurrence of a spontaneous pneumothorax is considered in some cases.13
The distinctive dermatologic features associated with BHD include multiple white papules primarily found on the nose and face. Pathologically, these manifestations have a range of histologic distinctions, from fibrofolliculomas to benign hamartomas of the hair follicles and trichodiscomas.5 The diagnostic criteria outlined by Menko et al note that confirmation of BHD requires the presence of either ≥ 5 pathologically confirmed fibrofolliculomas or trichodiscomas, a documented pathogenic FLCN gene mutation, or the fulfillment of 2 minor criteria. These minor criteria include the presence of multiple lung cysts, early-onset renal cancer, or a first-degree relative with BHD.5
Recurrent Pneumothoraces Management
After the first episode of spontaneous pneumothorax, early pleurodesis is indicated as the risk of recurrence can be as high as 75%.4,14 Specific pleurodesis modalities have shown promising results, such as total pleural covering with cellulose mesh. In a small retrospective review, cellulose mesh demonstrated a significant reduction in the recurrence rate of pneumothorax at 7.5 years for patients with BHD compared with partial covering.15 Apart from preventing further pneumothorax episodes in the affected lung, it is also important to highlight patient education and monitoring after initial pleurodesis, as the contralateral lung is also at risk. As demonstrated in this case, the patient had received pleurodesis of his right lung but experienced another pneumothorax of his contralateral lung a few years later.
Lastly, the patient was advised to avoid air travel altogether; however, current data may suggest that air travel may not be an absolute contraindication for patients with BHD. Although the literature on this topic is limited, a retrospective study by Johannesma et al involving 158 patients with BHD surveyed on pneumothorax occurrence after air travel indicated a calculated risk of 0.63% per flight. Notably, only 3 of 13 patients with BHD and recurrent pneumothoraces after travel had undergone pleurodesis in the past.16 Therefore, counseling patients on the potential risks of air travel and allowing essential flights while diligently monitoring for symptoms during and after travel may be a reasonable, patient-centered approach in contrast to a complete restriction on air travel.
Timing to Diagnosis
Diagnosing BHD is challenging and often delayed. In a 2022 study by Steinlein et al, the average delay in BHD diagnoses in their cohort was 9.3 years, with 4 patients also diagnosed with renal malignancy during the study period.17 The difficulty in diagnosis can be attributed to the heterogeneous presentation among affected family members, some of whom may exclusively exhibit pulmonary cystic lesions without dermatologic findings.
A lack of longitudinal care for this patient may have contributed to the diagnostic delay. The patient had pneumothorax events across separate care settings and locations, and due to employment-related relocations, he often re-established care at various health care systems. This case highlights the importance of continuity of care, especially in BHD, where monitoring for renal tumors is also essential to long-term management.17,18
Renal Tumor Monitoring
Finally, once BHD is diagnosed, one of the most important considerations is to begin routine monitoring for renal malignancies. Current recommendations advise starting lifelong renal cancer screening, even as early as age 20 years, with annual MRIs, as renal ultrasound may not be sufficiently sensitive to detect smaller lesions.19 The screening interval can be extended to every 2 years for patients without a family history of renal tumors or suspicious renal lesions. If tumors are found, then nephron-sparing surgery is recommended, given the potential for the development of chronic renal insufficiency in patients with BHD.20
Conclusions
BHD is a rare and complex syndrome in which early recognition and diagnosis play a pivotal role in preventing potentially severe complications such as renal malignancies. Suspicion of a genetic disorder, such as BHD, LAM, or PLCH, should arise in patients who experience spontaneous pneumothorax, especially in the presence of multiple cystic lesions or a family history of pneumothoraces. Early consideration of pleurodesis after the first spontaneous pneumothorax is advisable. The complex presentation of BHD contributes to the difficulty of diagnosis and may delay recognition, which can be exacerbated by variable continuity of care.
Birt-Hogg-Dubé syndrome (BHD) is an autosomal dominant disease that arises from loss-of-function mutations in the FLCN gene. FLCN encodes folliculin, which is presumed to function as a tumor suppressor, though its precise role is incompletely understood.1,2 BHD is characterized by multiple pulmonary cysts leading to recurrent spontaneous pneumothoraces, cutaneous lesions—specifically fibrofolliculomas—and an increased risk of renal malignancies. Diagnosing BHD is challenging due to the variable presentation of the disease. Some patients may only have cystic lung diseases, while others may not have characteristic skin lesions.3-5 It is important to maintain awareness of BHD, especially when the diagnosis dictates the need for genetic counseling.
Case Presentation
A male veteran in his 60s, who was a lifelong nonsmoker with a history of extensive bullous emphysema and recurrent pneumothoraces, presented to the Veterans Affairs Greater Los Angeles Healthcare System pulmonary clinic while transferring care from a separate institution.
According to the patient, the first pneumothorax episode occurred about 20 years before presentation, followed by a recurrence a few years later after he was diagnosed with emphysema. He underwent pleurodesis of the right lung during his service abroad. Another episode nearly a decade after the first pneumothorax necessitated pleurodesis of the left lung (Figure 1). The patient's family history revealed pulmonary cysts in 1 immediate family member but no history of renal tumors. Notably, his mother passed away at a young age due to tuberculosis.

On physical examination, numerous skin tags and acrochordons on the face were observed, which had been stable for > 30 years. Despite a slow decline in exercise capacity following pleurodesis, the patient could still walk multiple miles daily and climb 3 flights of stairs before needing to rest. Pulmonary function testing (PFT) showed a forced expiratory volume in 1 second (FEV1)/forced vital capacity ratio of 0.84 with reduced FEV1, total lung capacity (TLC), and diffusion capacity for carbon monoxide (DLCO), indicating a mild restrictive ventilatory defect and reduced diffusing capacity.
Laboratory results revealed a normal α-1 antitrypsin level: 133 mg/dL (reference, 83-199 mg/dL), with a Pi*MS phenotype and undetectable antinuclear antibodies. The most recent chest computed tomography (CT) in 2019, displayed paraseptal and centrilobular emphysema, scattered blebs, and scarring consistent with prior pleurodesis procedures (Figure 2).

Genetic testing for the FLCN gene revealed heterozygous pathogenic mutation: c.1285del and p.His429Thrfs*39, which confirmed the diagnosis of BHD. A shave biopsy of a postauricular papular lesion confirmed a histologic pattern of fibrofolliculoma/trichodiscoma.
Follow-up and Outcomes
After confirmation of the BHD diagnosis, the patient was referred to genetic counseling and scheduled for annual magnetic resonance imaging (MRI) of the abdomen and pelvis to screen for renal malignancies. As the patient was able to establish care with a new long-term primary care practitioner in the outpatient setting, he continues regular follow-up visits in the pulmonary clinic with stable respiratory symptoms and no recurrent pneumothoraces thus far.
Discussion
Differential Diagnoses of Cystic Pulmonary Lesions
BHD is an important differential diagnosis to consider in the presentation of diffuse cystic lung diseases. Still, 2 other crucial considerations are pulmonary Langerhans cell histiocytosis (PLCH) and lymphangioleiomyomatosis (LAM), which occur at slightly higher frequencies than BHD.6
One of the first steps in radiographically evaluating cystic lung diseases is to characterize the cysts. The Fleischner Society defines true cysts as a “round parenchymal lucency or low-attenuating area with a well-defined interface with normal lung.”7 Mimics of cystic lesions may include cavitary lung lesions, thick-walled spaces within another area of mass, nodule, or consolidation. Another mimic is a pneumatocele, a pseudocyst that lacks epithelial lining and may be secondary to bacterial pneumonia, pneumocystis infections, trauma, or prior mechanical ventilation.8After characterizing true cysts, different patterns of cystic lesions can also be associated with specific diseases. Cysts in PLCH are commonly more uniform and round, whereas the cysts in LAM may be more irregularly shaped. 9 Cysts in BHD may be larger and predominantly located in basal and paramediastinal areas.4LAM is associated with tuberous sclerosis, which can also present with skin lesions (angiofibromas) and renal tumors (angiomyolipomas), thus creating a very similar picture to BHD. Therefore, tissue biopsies of skin lesions are essential as histopathology can identify characteristic fibrofolliculomas specific to BHD. While genetic testing would also strongly support the diagnosis of BHD, it is essential to note that negative genetic testing does not rule out BHD.4Lastly, lymphoid interstitial pneumonia (LIP) is another important consideration in the differential diagnosis of cystic lung diseases. LIP presents with not only perivascular cysts and centrilobular nodules but also diffuse ground-glass attenuation.10 In contrast to BHD, LIP is associated with autoimmune diseases such as Sjögren syndrome and infectious diseases such as HIV; thus, it may be differentiated from BHD by the presence of underlying disease processes and may warrant serologic testing for potential rheumatologic disorders.
Characteristics and Diagnostic Criteria
Cystic lung disease is the most common presentation of BHD. It presents in > 80% of cases and confers a 50-fold increase in the risk of spontaneous pneumothorax compared with the general population.4,11 Recurrent pneumothoraces are observed in about 25% to 30% of patients with BHD, typically occurring between the third and fifth decades of life and at significantly decreased rates after 50 years of age.12 A spontaneous pneumothorax might serve as the initial and perhaps the sole clinical presentation for some patients with BHD, but others may present with other respiratory symptoms such as cough and exertional dyspnea. PFT results may be normal or reveal a mild restrictive ventilatory defect and reduced DLCO, as reported in a few cases.6 The management of pulmonary complications primarily revolves around reducing the risk of pneumothoraces, which includes precautions such as avoiding positive pressure ventilation and air travel. Early pleurodesis with the first occurrence of a spontaneous pneumothorax is considered in some cases.13
The distinctive dermatologic features associated with BHD include multiple white papules primarily found on the nose and face. Pathologically, these manifestations have a range of histologic distinctions, from fibrofolliculomas to benign hamartomas of the hair follicles and trichodiscomas.5 The diagnostic criteria outlined by Menko et al note that confirmation of BHD requires the presence of either ≥ 5 pathologically confirmed fibrofolliculomas or trichodiscomas, a documented pathogenic FLCN gene mutation, or the fulfillment of 2 minor criteria. These minor criteria include the presence of multiple lung cysts, early-onset renal cancer, or a first-degree relative with BHD.5
Recurrent Pneumothoraces Management
After the first episode of spontaneous pneumothorax, early pleurodesis is indicated as the risk of recurrence can be as high as 75%.4,14 Specific pleurodesis modalities have shown promising results, such as total pleural covering with cellulose mesh. In a small retrospective review, cellulose mesh demonstrated a significant reduction in the recurrence rate of pneumothorax at 7.5 years for patients with BHD compared with partial covering.15 Apart from preventing further pneumothorax episodes in the affected lung, it is also important to highlight patient education and monitoring after initial pleurodesis, as the contralateral lung is also at risk. As demonstrated in this case, the patient had received pleurodesis of his right lung but experienced another pneumothorax of his contralateral lung a few years later.
Lastly, the patient was advised to avoid air travel altogether; however, current data may suggest that air travel may not be an absolute contraindication for patients with BHD. Although the literature on this topic is limited, a retrospective study by Johannesma et al involving 158 patients with BHD surveyed on pneumothorax occurrence after air travel indicated a calculated risk of 0.63% per flight. Notably, only 3 of 13 patients with BHD and recurrent pneumothoraces after travel had undergone pleurodesis in the past.16 Therefore, counseling patients on the potential risks of air travel and allowing essential flights while diligently monitoring for symptoms during and after travel may be a reasonable, patient-centered approach in contrast to a complete restriction on air travel.
Timing to Diagnosis
Diagnosing BHD is challenging and often delayed. In a 2022 study by Steinlein et al, the average delay in BHD diagnoses in their cohort was 9.3 years, with 4 patients also diagnosed with renal malignancy during the study period.17 The difficulty in diagnosis can be attributed to the heterogeneous presentation among affected family members, some of whom may exclusively exhibit pulmonary cystic lesions without dermatologic findings.
A lack of longitudinal care for this patient may have contributed to the diagnostic delay. The patient had pneumothorax events across separate care settings and locations, and due to employment-related relocations, he often re-established care at various health care systems. This case highlights the importance of continuity of care, especially in BHD, where monitoring for renal tumors is also essential to long-term management.17,18
Renal Tumor Monitoring
Finally, once BHD is diagnosed, one of the most important considerations is to begin routine monitoring for renal malignancies. Current recommendations advise starting lifelong renal cancer screening, even as early as age 20 years, with annual MRIs, as renal ultrasound may not be sufficiently sensitive to detect smaller lesions.19 The screening interval can be extended to every 2 years for patients without a family history of renal tumors or suspicious renal lesions. If tumors are found, then nephron-sparing surgery is recommended, given the potential for the development of chronic renal insufficiency in patients with BHD.20
Conclusions
BHD is a rare and complex syndrome in which early recognition and diagnosis play a pivotal role in preventing potentially severe complications such as renal malignancies. Suspicion of a genetic disorder, such as BHD, LAM, or PLCH, should arise in patients who experience spontaneous pneumothorax, especially in the presence of multiple cystic lesions or a family history of pneumothoraces. Early consideration of pleurodesis after the first spontaneous pneumothorax is advisable. The complex presentation of BHD contributes to the difficulty of diagnosis and may delay recognition, which can be exacerbated by variable continuity of care.
- Schmidt LS, Linehan WM. Molecular genetics and clinical features of Birt-Hogg-Dubé-Syndrome. Nat Rev Urol. 2015;12:558-569. doi:10.1038/nrurol.2015.206
- Lim DHK, Rehal PK, Nahorski MS, et al. A new locus-specific database (LSDB) for mutations in the folliculin (FLCN) gene. Hum Mutat. 2010;31:E1043-1051. doi:10.1002/humu.21130
- Aivaz O, Berkman S, Middelton L, et al. Comedonal and cystic fibrofolliculomas in Birt-Hogg-Dube syndrome. JAMA Dermatology. 2015;151:770-774. doi:10.1001/jamadermatol.2015.0215
- Daccord C, Good JM, Morren MA, et al. Birt–Hogg–Dubé syndrome. Eur Respir Rev. 2020;29:200042. doi:10.1183/16000617.0042-2020
- Menko FH, van Steensel MA, Giraud S, et al. Birt-Hogg-Dubé syndrome: diagnosis and management. The Lancet Oncology. 2009;10:1199-1206. doi:10.1016/S1470-2045(09)70188-3
- Daccord C, Cottin V, Prévot G, et al. Lung function in Birt-Hogg-Dubé syndrome: a retrospective analysis of 96 patients. Orphanet J Rare Dis. 2020;15:120. doi:10.1186/s13023-020-01402-y
- Hansell DM, Bankier AA, MacMahon H, et al. Fleischner Society: glossary of terms for thoracic imaging. Radiology. 2008;246:697-722. doi:10.1148/radiol.2462070712
- Jamil A, Kasi A. Pneumatocele. In: StatPearls. StatPearls Publishing; 2024. Accessed March 2, 2026. http://www.ncbi.nlm.nih.gov/books/NBK556146/
- Bhardwaj H, Bhardwaj B. Differentiating pulmonary lymphangioleiomyomatosis from pulmonary langerhans cell histiocytosis and Birt-Hogg-Dube syndrome. Lung India. 2013;30:372-373. doi:10.4103/0970-2113.120611
- Swigris JJ, Berry GJ, Raffin TA, et al. Lymphoid interstitial pneumonia: a narrative review. Chest. 2002;122:2150-2164. doi:10.1378/chest.122.6.2150
- Zbar B, Alvord WG, Glenn G, et al. Risk of renal and colonic neoplasms and spontaneous pneumothorax in the Birt-Hogg-Dubé syndrome. Cancer Epidemiol Biomarkers Prev. 2002;11:393-400.
- Sattler EC, Steinlein OK. Delayed diagnosis of Birt-Hogg-Dubé syndrome due to marked intrafamilial clinical variability: a case report. BMC Med Genet. 2018;19:45. doi:10.1186/s12881-018-0558-0
- Gupta N, Seyama K, McCormack FX. Pulmonary manifestations of Birt-Hogg-Dubé syndrome. Fam Cancer. 2013;12:387-396. doi:10.1007/s10689-013-9660-9
- Gupta N, Kopras EJ, Henske EP, et al. Spontaneous pneumothoraces in patients with Birt–Hogg–Dubé syndrome. Ann Am Thorac Soc. 2017;14:706-713. doi:10.1513/AnnalsATS.201611-886OC
- Mizobuchi T, Kurihara M, Ebana H, et al. A total pleural covering of absorbable cellulose mesh prevents pneumothorax recurrence in patients with Birt-Hogg-Dubé syndrome. Orphanet J Rare Dis. 2018;13:78. doi:10.1186/s13023-018-0790-x
- Johannesma PC, van de Beek I, van der Wel JWT, et al. Risk of spontaneous pneumothorax due to air travel and diving in patients with Birt–Hogg–Dubé syndrome. Springerplus. 2016;5:1506. doi:10.1186/s40064-016-3009-4
- Steinlein OK, Reithmair M, Syunyaeva Z, et al. Delayed diagnosis of Birt-Hogg-Dubé syndrome might be aggravated by gender bias. eClinicalMedicine. 2022;51:101572. doi:10.1016/j.eclinm.2022.101572
- Pereira Gray DJ, Sidaway-Lee K, White E, et al. Continuity of care with doctors—a matter of life and death? A systematic review of continuity of care and mortality. BMJ Open. 2018;8:e021161. doi:10.1136/bmjopen-2017-021161
- Sattler EC, Steinlein OK. GeneReviews Birt-Hogg-Dubé syndrome. January 30, 2020. Accessed March 2, 2026. https://www.ncbi.nlm.nih.gov/books/NBK1522/table
- Stamatakis L, Metwalli AR, Middelton LA, et al. Diagnosis and management of BHD-associated kidney cancer. Fam Cancer. 2013;12:397-402. doi:10.1007/s10689-013-9657-4
- Schmidt LS, Linehan WM. Molecular genetics and clinical features of Birt-Hogg-Dubé-Syndrome. Nat Rev Urol. 2015;12:558-569. doi:10.1038/nrurol.2015.206
- Lim DHK, Rehal PK, Nahorski MS, et al. A new locus-specific database (LSDB) for mutations in the folliculin (FLCN) gene. Hum Mutat. 2010;31:E1043-1051. doi:10.1002/humu.21130
- Aivaz O, Berkman S, Middelton L, et al. Comedonal and cystic fibrofolliculomas in Birt-Hogg-Dube syndrome. JAMA Dermatology. 2015;151:770-774. doi:10.1001/jamadermatol.2015.0215
- Daccord C, Good JM, Morren MA, et al. Birt–Hogg–Dubé syndrome. Eur Respir Rev. 2020;29:200042. doi:10.1183/16000617.0042-2020
- Menko FH, van Steensel MA, Giraud S, et al. Birt-Hogg-Dubé syndrome: diagnosis and management. The Lancet Oncology. 2009;10:1199-1206. doi:10.1016/S1470-2045(09)70188-3
- Daccord C, Cottin V, Prévot G, et al. Lung function in Birt-Hogg-Dubé syndrome: a retrospective analysis of 96 patients. Orphanet J Rare Dis. 2020;15:120. doi:10.1186/s13023-020-01402-y
- Hansell DM, Bankier AA, MacMahon H, et al. Fleischner Society: glossary of terms for thoracic imaging. Radiology. 2008;246:697-722. doi:10.1148/radiol.2462070712
- Jamil A, Kasi A. Pneumatocele. In: StatPearls. StatPearls Publishing; 2024. Accessed March 2, 2026. http://www.ncbi.nlm.nih.gov/books/NBK556146/
- Bhardwaj H, Bhardwaj B. Differentiating pulmonary lymphangioleiomyomatosis from pulmonary langerhans cell histiocytosis and Birt-Hogg-Dube syndrome. Lung India. 2013;30:372-373. doi:10.4103/0970-2113.120611
- Swigris JJ, Berry GJ, Raffin TA, et al. Lymphoid interstitial pneumonia: a narrative review. Chest. 2002;122:2150-2164. doi:10.1378/chest.122.6.2150
- Zbar B, Alvord WG, Glenn G, et al. Risk of renal and colonic neoplasms and spontaneous pneumothorax in the Birt-Hogg-Dubé syndrome. Cancer Epidemiol Biomarkers Prev. 2002;11:393-400.
- Sattler EC, Steinlein OK. Delayed diagnosis of Birt-Hogg-Dubé syndrome due to marked intrafamilial clinical variability: a case report. BMC Med Genet. 2018;19:45. doi:10.1186/s12881-018-0558-0
- Gupta N, Seyama K, McCormack FX. Pulmonary manifestations of Birt-Hogg-Dubé syndrome. Fam Cancer. 2013;12:387-396. doi:10.1007/s10689-013-9660-9
- Gupta N, Kopras EJ, Henske EP, et al. Spontaneous pneumothoraces in patients with Birt–Hogg–Dubé syndrome. Ann Am Thorac Soc. 2017;14:706-713. doi:10.1513/AnnalsATS.201611-886OC
- Mizobuchi T, Kurihara M, Ebana H, et al. A total pleural covering of absorbable cellulose mesh prevents pneumothorax recurrence in patients with Birt-Hogg-Dubé syndrome. Orphanet J Rare Dis. 2018;13:78. doi:10.1186/s13023-018-0790-x
- Johannesma PC, van de Beek I, van der Wel JWT, et al. Risk of spontaneous pneumothorax due to air travel and diving in patients with Birt–Hogg–Dubé syndrome. Springerplus. 2016;5:1506. doi:10.1186/s40064-016-3009-4
- Steinlein OK, Reithmair M, Syunyaeva Z, et al. Delayed diagnosis of Birt-Hogg-Dubé syndrome might be aggravated by gender bias. eClinicalMedicine. 2022;51:101572. doi:10.1016/j.eclinm.2022.101572
- Pereira Gray DJ, Sidaway-Lee K, White E, et al. Continuity of care with doctors—a matter of life and death? A systematic review of continuity of care and mortality. BMJ Open. 2018;8:e021161. doi:10.1136/bmjopen-2017-021161
- Sattler EC, Steinlein OK. GeneReviews Birt-Hogg-Dubé syndrome. January 30, 2020. Accessed March 2, 2026. https://www.ncbi.nlm.nih.gov/books/NBK1522/table
- Stamatakis L, Metwalli AR, Middelton LA, et al. Diagnosis and management of BHD-associated kidney cancer. Fam Cancer. 2013;12:397-402. doi:10.1007/s10689-013-9657-4
A Case of Birt-Hogg-Dubé Syndrome: A Rare but Essential Diagnosis to Consider
A Case of Birt-Hogg-Dubé Syndrome: A Rare but Essential Diagnosis to Consider
Recliner Butt
Recliner Butt
DIAGNOSIS
Senile gluteal dermatosis (SGD). SGD is a friction- related skin injury, also known as recliner butt, chronic tissue injury, or grandfather’s disease.1-4 The hallmarks include blanchable erythematous plaques and/or purplish discoloration of the fleshy part of the buttocks or posterior thighs, with little to no change over months to years. Additional findings may include skin erosions, lichenification, and ridging. SGD is most commonly seen in older adults with impaired mobility who spend prolonged periods in a reclined position, particularly those who slide down in a chair, “scoot,” or drag the buttocks during transfers or repositioning.
The pathogenesis of SGD is thought to involve microischemia associated with prolonged sitting.4 Histopathologic findings are nonspecific and may include hyperkeratosis, psoriasiform epidermal hyperplasia, vascular dilatation or proliferation in the superficial dermis, and reactive lymphohistiocytic perivascular infiltrate.4 The condition is poorly recognized and is likely underreported. Treatment involves reducing frictional injury by avoiding the reclined position, minimizing sliding during transfers, and frequent repositioning. Petroleum-based ointments may be applied to reduce friction and protect the skin barrier. Heat-dissipating chair cushions can be used to offload pressure and improve the local microclimate. Friction-related skin injuries need to be differentiated from pressure injuries, in which pressure and shear are the driving forces, and lesions are located over bony prominences.
Unlike SGD, chronic lichen sclerosus typically occurs in the anogenital area, including the scrotum and vulva, and is typically intensely pruritic, with white, atrophic plaques.
A stage 2 pressure injury is characterized by an area of partial-thickness skin loss with exposed dermis, usually overlying a bony prominence. Although friction-related skin injuries may contain erosions, they are often maroon or purple and are not located over a bony prominence.
Deep tissue injury (DTI) is characterized by nonblanchable dark red or purple skin discoloration, with intact or nonintact skin. While friction injuries may mimic DTIs, they lack the characteristic anatomic location over a bony prominence and the predictable evolution pattern seen in DTIs.
Incontinence-associated dermatitis (IAD) results from prolonged exposure to urine and/or feces and presents with erythema, inflammation, and epidermal erosion. Although IAD can look similar or coincide with SGD, the affected area is typically red, not purple. Skin ridging and lichenification are also not seen in IAD cases.
Sedentary behavior is prevalent among older adults, with nearly 60% spending > 4 hours per day sitting.5 Prolonged sitting puts them at risk for friction-related skin injuries. Even though friction-related skin injuries are typically nonprogressive, these patients are also at risk for pressure injuries that are typically acquired in a sitting position (eg, ischial and sacrococcygeal). Therefore, it is imperative that clinicians not only address SGD but also implement a pressure injury prevention plan.
- Berke CT. Pathology and clinical presentation of friction injuries case series and literature review. J Wound Ostomy Continence Nurs. 2105;42:47-61. doi:10.1097/WON.0000000000000087
- Mahoney MF, Rozenboom BJ. Definition and characteristics of chronic tissue injury: a unique form of skin damage. J Wound Ostomy Continence Nurs. 2019;46:187-191. doi:10.1097/WON.0000000000000527
- Kelechi, TJ. Commentary: chronic tissue injury. Making the case for a new form of skin damage. J Wound Ostomy Continence Nurs. 2019;46:192-193. doi:10.1097/WON.0000000000000533
- Majid I, Jairam D, Baheti K, et al. Senile guletal dermatosis: update on etiopathogenesis, Diagnostic Criteria, and Management. Dermatol Ther. 2024;37:e5556190.
- Harvey JA, Chastin SF, Skelton DA. Prevalence of sedentary behavior in older adults: a systematic review. Int J Environ Res Public Health. 2013;10:6645-6661. doi:10.3390/ijerph10126645
DIAGNOSIS
Senile gluteal dermatosis (SGD). SGD is a friction- related skin injury, also known as recliner butt, chronic tissue injury, or grandfather’s disease.1-4 The hallmarks include blanchable erythematous plaques and/or purplish discoloration of the fleshy part of the buttocks or posterior thighs, with little to no change over months to years. Additional findings may include skin erosions, lichenification, and ridging. SGD is most commonly seen in older adults with impaired mobility who spend prolonged periods in a reclined position, particularly those who slide down in a chair, “scoot,” or drag the buttocks during transfers or repositioning.
The pathogenesis of SGD is thought to involve microischemia associated with prolonged sitting.4 Histopathologic findings are nonspecific and may include hyperkeratosis, psoriasiform epidermal hyperplasia, vascular dilatation or proliferation in the superficial dermis, and reactive lymphohistiocytic perivascular infiltrate.4 The condition is poorly recognized and is likely underreported. Treatment involves reducing frictional injury by avoiding the reclined position, minimizing sliding during transfers, and frequent repositioning. Petroleum-based ointments may be applied to reduce friction and protect the skin barrier. Heat-dissipating chair cushions can be used to offload pressure and improve the local microclimate. Friction-related skin injuries need to be differentiated from pressure injuries, in which pressure and shear are the driving forces, and lesions are located over bony prominences.
Unlike SGD, chronic lichen sclerosus typically occurs in the anogenital area, including the scrotum and vulva, and is typically intensely pruritic, with white, atrophic plaques.
A stage 2 pressure injury is characterized by an area of partial-thickness skin loss with exposed dermis, usually overlying a bony prominence. Although friction-related skin injuries may contain erosions, they are often maroon or purple and are not located over a bony prominence.
Deep tissue injury (DTI) is characterized by nonblanchable dark red or purple skin discoloration, with intact or nonintact skin. While friction injuries may mimic DTIs, they lack the characteristic anatomic location over a bony prominence and the predictable evolution pattern seen in DTIs.
Incontinence-associated dermatitis (IAD) results from prolonged exposure to urine and/or feces and presents with erythema, inflammation, and epidermal erosion. Although IAD can look similar or coincide with SGD, the affected area is typically red, not purple. Skin ridging and lichenification are also not seen in IAD cases.
Sedentary behavior is prevalent among older adults, with nearly 60% spending > 4 hours per day sitting.5 Prolonged sitting puts them at risk for friction-related skin injuries. Even though friction-related skin injuries are typically nonprogressive, these patients are also at risk for pressure injuries that are typically acquired in a sitting position (eg, ischial and sacrococcygeal). Therefore, it is imperative that clinicians not only address SGD but also implement a pressure injury prevention plan.
DIAGNOSIS
Senile gluteal dermatosis (SGD). SGD is a friction- related skin injury, also known as recliner butt, chronic tissue injury, or grandfather’s disease.1-4 The hallmarks include blanchable erythematous plaques and/or purplish discoloration of the fleshy part of the buttocks or posterior thighs, with little to no change over months to years. Additional findings may include skin erosions, lichenification, and ridging. SGD is most commonly seen in older adults with impaired mobility who spend prolonged periods in a reclined position, particularly those who slide down in a chair, “scoot,” or drag the buttocks during transfers or repositioning.
The pathogenesis of SGD is thought to involve microischemia associated with prolonged sitting.4 Histopathologic findings are nonspecific and may include hyperkeratosis, psoriasiform epidermal hyperplasia, vascular dilatation or proliferation in the superficial dermis, and reactive lymphohistiocytic perivascular infiltrate.4 The condition is poorly recognized and is likely underreported. Treatment involves reducing frictional injury by avoiding the reclined position, minimizing sliding during transfers, and frequent repositioning. Petroleum-based ointments may be applied to reduce friction and protect the skin barrier. Heat-dissipating chair cushions can be used to offload pressure and improve the local microclimate. Friction-related skin injuries need to be differentiated from pressure injuries, in which pressure and shear are the driving forces, and lesions are located over bony prominences.
Unlike SGD, chronic lichen sclerosus typically occurs in the anogenital area, including the scrotum and vulva, and is typically intensely pruritic, with white, atrophic plaques.
A stage 2 pressure injury is characterized by an area of partial-thickness skin loss with exposed dermis, usually overlying a bony prominence. Although friction-related skin injuries may contain erosions, they are often maroon or purple and are not located over a bony prominence.
Deep tissue injury (DTI) is characterized by nonblanchable dark red or purple skin discoloration, with intact or nonintact skin. While friction injuries may mimic DTIs, they lack the characteristic anatomic location over a bony prominence and the predictable evolution pattern seen in DTIs.
Incontinence-associated dermatitis (IAD) results from prolonged exposure to urine and/or feces and presents with erythema, inflammation, and epidermal erosion. Although IAD can look similar or coincide with SGD, the affected area is typically red, not purple. Skin ridging and lichenification are also not seen in IAD cases.
Sedentary behavior is prevalent among older adults, with nearly 60% spending > 4 hours per day sitting.5 Prolonged sitting puts them at risk for friction-related skin injuries. Even though friction-related skin injuries are typically nonprogressive, these patients are also at risk for pressure injuries that are typically acquired in a sitting position (eg, ischial and sacrococcygeal). Therefore, it is imperative that clinicians not only address SGD but also implement a pressure injury prevention plan.
- Berke CT. Pathology and clinical presentation of friction injuries case series and literature review. J Wound Ostomy Continence Nurs. 2105;42:47-61. doi:10.1097/WON.0000000000000087
- Mahoney MF, Rozenboom BJ. Definition and characteristics of chronic tissue injury: a unique form of skin damage. J Wound Ostomy Continence Nurs. 2019;46:187-191. doi:10.1097/WON.0000000000000527
- Kelechi, TJ. Commentary: chronic tissue injury. Making the case for a new form of skin damage. J Wound Ostomy Continence Nurs. 2019;46:192-193. doi:10.1097/WON.0000000000000533
- Majid I, Jairam D, Baheti K, et al. Senile guletal dermatosis: update on etiopathogenesis, Diagnostic Criteria, and Management. Dermatol Ther. 2024;37:e5556190.
- Harvey JA, Chastin SF, Skelton DA. Prevalence of sedentary behavior in older adults: a systematic review. Int J Environ Res Public Health. 2013;10:6645-6661. doi:10.3390/ijerph10126645
- Berke CT. Pathology and clinical presentation of friction injuries case series and literature review. J Wound Ostomy Continence Nurs. 2105;42:47-61. doi:10.1097/WON.0000000000000087
- Mahoney MF, Rozenboom BJ. Definition and characteristics of chronic tissue injury: a unique form of skin damage. J Wound Ostomy Continence Nurs. 2019;46:187-191. doi:10.1097/WON.0000000000000527
- Kelechi, TJ. Commentary: chronic tissue injury. Making the case for a new form of skin damage. J Wound Ostomy Continence Nurs. 2019;46:192-193. doi:10.1097/WON.0000000000000533
- Majid I, Jairam D, Baheti K, et al. Senile guletal dermatosis: update on etiopathogenesis, Diagnostic Criteria, and Management. Dermatol Ther. 2024;37:e5556190.
- Harvey JA, Chastin SF, Skelton DA. Prevalence of sedentary behavior in older adults: a systematic review. Int J Environ Res Public Health. 2013;10:6645-6661. doi:10.3390/ijerph10126645
Recliner Butt
Recliner Butt
An 89-year-old male veteran with a history of obesity (body mass index, 33), osteoarthritis, anemia, pulmonary embolism, and urinary incontinence presented for evaluation of gluteal skin lesions (Figure). The patient had poor mobility and spent most of the day in a recliner chair. He also slept in the recliner due to chronic dyspnea and orthopnea.
The gluteal region demonstrated purplish discoloration with blanchable erythema and superficial ulcerations. The affected area was not pruritic and had remained unchanged for 3 months.
A punch biopsy of the discolored gluteal area was performed. Histopathologic examination revealed hyperkeratosis, orthokeratosis, irregular acanthosis, and mild spongiosis. Vascular proliferation and papillary dermal edema were also noted.
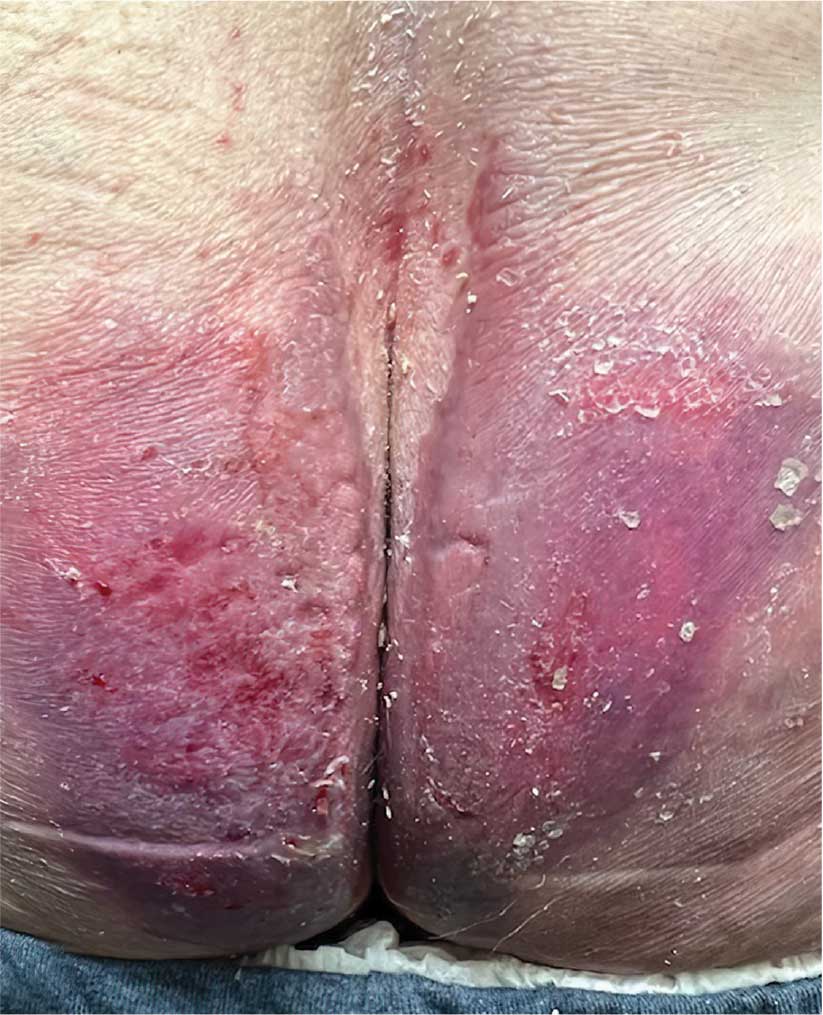
Predictors of Unplanned Postoperative Visits in a Veterans Affairs Hand Surgery Practice
Predictors of Unplanned Postoperative Visits in a Veterans Affairs Hand Surgery Practice
Patients make unplanned appointments after elective soft tissue hand surgery for real or perceived complications when they experience pain, anxiety, or fear. Unplanned appointments can create travel and financial burdens for patients and families. These appointments take time away from scheduled appointments and can contribute to late arrivals and delays in other clinics. Unscheduled appointments contribute to poor access when staff are diverted from scheduled appointments. If predictive factors can be identified, unplanned appointments may either be ameliorated or avoided with better perioperative risk management or education.
Methods
The US Department of Veterans Affairs (VA) North Florida/South Georgia Veterans Health System (NFSGVAHS) and University of Florida Institutional Review Board approved a retrospective chart review of all plastic surgery cases performed at the Malcom Randall VA Medical Center (MRVAMC) and Lake City VAMC operating rooms from July 1, 2018, through December 31, 2019, and January 1, 2021, through June 30, 2022 (nonurgent surgeries were discouraged during the COVID-19 pandemic). Elective soft tissue hand surgery cases were identified based on the operative description found in the Surgical Service Surgeon Staffing Report reviewed monthly by the Service Chief. Potential indicators of unplanned visits were recorded, including age; sex; diagnosis of diabetes, depression, anxiety, or posttraumatic stress disorder (PTSD); current smoking status; and residential zip code. We used the first 3 digits of the patients’ zip codes, which indicate region, as an estimate of proximity to the MRVAMC, which has a 50-county catchment area across North Florida and South Georgia. Diagnoses were found on the “problem list” from the electronic health record documented in the history and physical examinations before surgery. Clinic notes were examined for 3 months postsurgery to identify unplanned postoperative visits and the reason for the appointment. A χ2 analysis was conducted using Excel Version 2402. P < .05 was used to determine whether age (> 60 years), sex, proximity to MRVAMC, diabetes, smoking, depression, anxiety, or PTSD were statistically significant independent risk factors for these appointments.
Results
A total of 1009 elective soft tissue hand surgeries at MRVAMC were reviewed. The patients median age was 61 years. Patients included 173 women (17.1%) and 836 men (82.9%). Eighty-one patients (8.0%) returned for unplanned visits. Age (P = .82); proximity to MRVAMC (P = .34); and diabetes (P = .60), smoking (P = .55), anxiety (P = .33), or PTSD (P = .37) were not statistically significant predictors of unplanned appointments. Depression diagnosis (P = .04) and female sex (P = .03) were found to be independent risk factors for an unplanned appointment (Table 1). The most common indication for the requested appointment was pain-related, followed closely by noninfectious wound concerns and persistent symptoms (Table 2).

Discussion
Improved access, quality, and efficiency for patients are goals for the VA.1-3 The MRVAMC Plastic and Hand Surgery service provides care for the NFSGVAHS and receives an average of 15 to 20 consultation requests daily. The Veterans Health Administration is frequently challenged by staff shortages, and surgical services struggle to respond to consultation requests and treat patients within reasonable time frames.4,5
The objective of this study was to identify risk factors for unplanned postoperative appointments following elective hand surgery. Unplanned appointments prevent scheduled patients from being seen on time and contribute to backlogs and delays. When patients schedule multiple appointments on the same day, delays in the first clinic’s scheduled appointments create delays for the second and third clinics. Hand surgery clinics can provide a better experience for patients and staff by identifying and mitigating factors prompting unplanned visits.
We anticipated that wound complications would prompt unscheduled visits. Diabetes is a known risk factor for wound healing complications after plastic and hand surgery.6,7 A hemoglobin A1c (HbA1c) screening protocol used by the NFSGVAHS plastic surgery service since 2015 to identify poorly controlled patients with diabetes before surgery may partially explain this finding.8 We did not find a statistically significant difference between patients with diabetes and patients without diabetes for scheduling unplanned appointments. The plastic surgery service does not perform elective hand surgery unless the patient’s HbA1c level is < 9%, or violate the flexor sheath unless HbA1c level is < 8%. However, Zhuang et al found an increase in soft tissue infections after hand surgery with HbA1c levels ≥ 7%.9
Smoking is a potential factor in postoperative hand surgery complications.10,11 Lans et al found an increased incidence of 30-day emergency room visits in current and former smokers after outpatient upper extremity fracture surgery.12 The MRVAMC Plastic Surgery Service counsels patients about the risk of skin necrosis and delayed wound healing, but does not cancel cases or obtain laboratory values to verify abstinence in patients undergoing hand surgery. The VA has multiple resources available for patients interested in smoking cessation through mental health services.13
MRVAMC patients have been known to resist returning for scheduled appointments due to the costs or availability of transportation. We suspected that patients who lived further from MRVAMC would be less likely to return for unscheduled visits. We used the first 3 digits of the patients’ mailing zip code to estimate residential proximity to MRVAMC. An acknowledged limitation to this approach is that some veterans have primary addresses in other regions but still spend significant time in the MRVAMC catchment area and use the facility for their health care during the winter months. These “snowbirds” might reside near the facility despite having official addresses that are more distant. Additionally, there was no increased risk of unplanned visits after hand surgery in patients aged > 61 years (the median age of study participants) (P = .82). Dependence on a third party for transportation in older veterans could impact this finding.
Based on the observation that most patients needed reassurance rather than an intervention when they returned for unscheduled appointments, diagnoses of depression, anxiety, and PTSD were evaluated as separate predictive factors. In previous research, anxiety was found to be a risk factor for problematic recovery following carpal tunnel surgery.14 In the current study, depression was found to be a statistically significant predictor of unscheduled postoperative appointments (P = .04), while anxiety (P = .33) and PTSD (P = .37) were not statistically significant predictors. This is consistent with other studies that have found preexisting depression can predict complications after hand surgery.15,16 Vranceanu et al found that depression predicted pain intensity and disability after elective hand surgery.16 Similarly, Oflazoglu et al found a 12% incidence of depression based on the Patient Health Questionnaire-9 in new and returning hand patients who presented to an academic practice.17 They suggest patients should be assessed at all levels of care and that those with poor responses to surgical or nonsurgical management should be evaluated for depression. MRVAMC has a large mental health service consisting of psychiatrists, psychologists, addiction specialists, social workers, and homeless outreach, and patients tend to already have a diagnosis and mental health practitioner when they present to the clinic.
Recent studies found that wound problems, pain, and stiffness were the most common reasons for return visits.18,19 Shetty et al identified younger age, worse preoperative pain scores, and poor access to transportation as predictors of preventable emergency room visits, which generate higher health care expenditures than an office visit.19 Our study’s top reasons for appointments (pain, wound/scar concerns, persistent symptoms) can be addressed with additional presurgery patient and family education. Additionally, clinicians encourage nonnarcotic pain management strategies including anti-inflammatories, acetaminophen, elevation, splinting, and hand therapy, and the hospital employs experienced, fellowship-trained anesthesia block faculty who help limit perioperative narcotic use. Patients are advised that pain can be used to guide them through the postoperative recovery by preventing overuse and alerting them to a problem that would be masked with narcotics, and long-standing problems such as chronic nerve compressions may continue to cause pain after surgery.
Patients and families can be given consistent and repetitive verbal and written information, instructions, and expectations at the initial consultation, preoperative appointment, and on the day of surgery. Postoperatively, outside their scheduled appointments, patients are encouraged to use the My HealtheVet secure messaging system or call the clinic to access an experienced registered nurse before making a long drive. Access to virtual or phone visits can reduce emergent in-person visits in a VA population.20
Ozdag et al found that 42% of patients who had elective carpal tunnel surgery made unplanned electronic messages or phone contact within 2 weeks postsurgery. The authors point out the uncompensated administrative burden on the staff answering these messages and suggest pre-empting the contacts with more up-front education regarding postoperative pain expectations and management strategies.21
Fisher et al found that attending hand therapy reduced the number of emergency department visits in postoperative infection cases.22 At MRVAMC, a postoperative emergency department visit for a patient prompts an urgent unplanned appointment to the plastic surgery clinic, often on the same day. The MRVAMC occupational therapy clinic employed 3 on-site certified hand therapists during the study period. Because all hand surgery patients at the clinic receive hand therapy on the same day as their first postoperative appointment, attendance at hand therapy was not evaluated as a predictor of unplanned visits. Scheduled hand therapy is another point of contact where the clinic can provide reassurance and patient education.
While females made up 17.1% of the patients in this study, they constituted 12.5% of all veterans in Florida in fiscal year 2023.23 This study found that women were more likely to present for unplanned postoperative appointments (P = .03). This is consistent with existing literature which has found that women are higher users of health care and office-based appointments.24,25 This finding suggests the need for further study into whether our methods of communicating instructions to female patients undergoing plastic surgery may not be optimal.
Strengths and Limitations
As a retrospective review, the authors used information documented by multiple different health care practitioners, including trainees. The electronic medical record problem lists and templates provide consistency of information; however, less seasoned clinicians may interpret what they see and hear differently from more experienced clinicians in the postoperative setting. This study occurred in one part of the country with demographics that may not mirror other VA systems or the general population. The authors hope this study can be a starting point for other health care facilities to investigate ways to minimize the burden of unscheduled appointments. A strength of the study is that it was conducted within a closed system, as patients tend to stay within the VA system and documentation and communication among clinicians, even outside the immediate facility, are easily accessed through the electronic health record.
Conclusions
This study found that depression diagnosis and female sex are statistically significant predictors of unplanned postoperative visits after elective soft tissue hand surgery. More effective patient education during the preoperative period, particularly in patients with depression, may be warranted.
- Apaydin EA, Paige NM, Begashaw MM, et al. Veterans Health Administration (VA) vs. non-VA healthcare quality: a systematic review. J Gen Intern Med. 2023;38:2179-2188. doi:10.1007/s11606-023-08207-2
- Blegen M, Ko J, Salzman G, et al. Comparing quality of surgical care between the US Department of Veterans Affairs and non-Veterans Affairs settings: a systematic review. J Am Coll Surg. 2023;237:352-361. doi:10.1097/XCS.0000000000000720
- Valsangkar NP, Eppstein AC, Lawson RA, et al. Effect of lean processes on surgical wait times and efficiency in a tertiary care veterans affairs medical center. JAMA Surg. 2017;152:42-47. doi:10.1001/jamasurg.2016.2808
- National Association of Veterans Affairs Physicians and Dentists. Physicians remain at top of staffing shortage in VA. NAVAPD. December 20, 2023. Accessed March 16, 2026. https://www.navapd.org/news/physicians-remain-at-top-of-staffing-shortage-in-va
- OIG Determination of Veterans Health Administration’s severe occupational staffing shortages fiscal year 2024. Veterans Affairs Office of Inspector General. August 7, 2024. Accessed February 4, 2026. https://www.vaoig.gov/reports/national-healthcare-review/oig-determination-veterans-health-administrations-severe-0
- Goltsman D, Morrison KA, Ascherman JA. Defining the association between diabetes and plastic surgery outcomes: an analysis of nearly 40,000 patients. Plast Reconstr Surg Glob Open. 2017;5:e1461. doi:10.1097/GOX.0000000000001461 7.
- Cox CT, Sierra S, Egan A, et al. Elevated hemoglobin A1c and the risk of postoperative complications in elective hand and upper extremity surgery. Cureus. 2023;15:e48373. doi:10.7759/cureus.48373
- Coady-Fariborzian L, Anstead C. HbA1c and infection in diabetic elective hand surgery: a Veterans Affair Medical Center experience 2012-2018. Hand (NY). 2023;18:994-998. doi:10.1177/1558944720937363<
- Zhuang T, Shapiro LM, Fogel N, et al. Perioperative laboratory markers as risk factors for surgical site infection after elective hand surgery. J Hand Surg Am. 2021;46:675-684. doi:10.1016/j.jhsa.2021.04.001
- Cho BH, Aziz KT, Giladi AM. The impact of smoking on early postoperative complications in hand surgery. J Hand Surg Am. 2021;46:336.e1-336.e11. doi:10.1016/j.jhsa.2020.07.01411.
- Del Core MA, Ahn J, Golden AS, et al. Effect of smoking on short-term postoperative complications after elective upper extremity surgery. Hand (N Y). 2022;17:231-238. doi:10.1177/1558944720926638
- Lans J, Beagles CB, Watkins IT, et al. Unplanned postoperative emergency department visits after upper extremity fracture surgery. J Orthop Trauma. 2025;39:22-27. doi:10.1097/BOT.0000000000002925
- Tobacco and health - how to quit. US Dept of Veterans Affairs. Updated October 29, 2025. Accessed February 4, 2026. https://www.mentalhealth.va.gov/quit-tobacco/how-to-quit.asp
- Ryan C, Miner H, Ramachandran S, et al. General anxiety is associated with problematic initial recovery after carpal tunnel release. Clin Orthop Relat Res. 2022;480:1576-1581. doi:10.1097/CORR.0000000000002115
- Crijns TJ, Bernstein DN, Ring D, et al. Depression and pain interference correlate with physical function in patients recovering from hand surgery. Hand (N Y). 2019;14:830-835. doi:10.1177/1558944718777814
- Vranceanu AM, Jupiter JB, Mudgal CS, et al. Predictors of pain intensity and disability after minor hand surgery. J Hand Surg Am. 2010;35:956-960. doi:10.1016/j.jhsa.2010.02.00117.
- Oflazoglu K, Mellema JJ, Menendez ME, et al. Prevalence of and factors associated with major depression in patients with upper extremity conditions. J Hand Surg Am. 2016;41:263-269. doi:10.1016/j.jhsa.2015.11.019
- Townsend CB, Henry TW, Lutsky KF, et al. Unplanned office visits following outpatient hand surgery. Hand (N Y). 2022;17:1264-1268. doi:10.1177/15589447211028932
- Shetty PN, Guarino GM, Zhang G, et al. Risk factors for preventable emergency department use after outpatient hand surgery. J Hand Surg Am. 2022;47:855-864. doi:10.1016/j.jhsa.2022.05.012
- Sommers-Olson B, Christianson J, Neumann T, et al. Reducing nonemergent visits to the emergency department in a Veterans Affairs multistate system. J Emerg Nurs. 2023;49:539-545. doi:10.1016/j.jen.2023.02.010
- Ozdag Y, Manzar S, El Koussaify J, et al. Unplanned postoperative phone calls and electronic messages for patients with and without opioid prescriptions after carpal tunnel release. J Hand Surg Glob Online. 2024;6:363-368. doi:10.1016/j.jhsg.2024.02.006
- Fisher AH, Gandhi J, Nelson Z, et al. Immediate interventions after surgery to reduce readmission for upper extremity infections. Ann Plast Surg. 2022;88:S163-S169. doi:10.1097/SAP.0000000000003141
- Florida Department of Veterans Affairs Fast Facts. Florida Department of Veterans Affairs. Accessed February 4, 2026. https://floridavets.org/our-veterans/profilefast-facts/
- Bertakis KD, Azari R, Helms LJ, et al. Gender differences in the utilization of health care services. J Fam Pract. 2000;49:147-152.
- Ashman JJ, Santo L, Okeyode T. Characteristics of office-based physician visits, 2018. NCHS Data Brief. 2021;408:1-8.
Patients make unplanned appointments after elective soft tissue hand surgery for real or perceived complications when they experience pain, anxiety, or fear. Unplanned appointments can create travel and financial burdens for patients and families. These appointments take time away from scheduled appointments and can contribute to late arrivals and delays in other clinics. Unscheduled appointments contribute to poor access when staff are diverted from scheduled appointments. If predictive factors can be identified, unplanned appointments may either be ameliorated or avoided with better perioperative risk management or education.
Methods
The US Department of Veterans Affairs (VA) North Florida/South Georgia Veterans Health System (NFSGVAHS) and University of Florida Institutional Review Board approved a retrospective chart review of all plastic surgery cases performed at the Malcom Randall VA Medical Center (MRVAMC) and Lake City VAMC operating rooms from July 1, 2018, through December 31, 2019, and January 1, 2021, through June 30, 2022 (nonurgent surgeries were discouraged during the COVID-19 pandemic). Elective soft tissue hand surgery cases were identified based on the operative description found in the Surgical Service Surgeon Staffing Report reviewed monthly by the Service Chief. Potential indicators of unplanned visits were recorded, including age; sex; diagnosis of diabetes, depression, anxiety, or posttraumatic stress disorder (PTSD); current smoking status; and residential zip code. We used the first 3 digits of the patients’ zip codes, which indicate region, as an estimate of proximity to the MRVAMC, which has a 50-county catchment area across North Florida and South Georgia. Diagnoses were found on the “problem list” from the electronic health record documented in the history and physical examinations before surgery. Clinic notes were examined for 3 months postsurgery to identify unplanned postoperative visits and the reason for the appointment. A χ2 analysis was conducted using Excel Version 2402. P < .05 was used to determine whether age (> 60 years), sex, proximity to MRVAMC, diabetes, smoking, depression, anxiety, or PTSD were statistically significant independent risk factors for these appointments.
Results
A total of 1009 elective soft tissue hand surgeries at MRVAMC were reviewed. The patients median age was 61 years. Patients included 173 women (17.1%) and 836 men (82.9%). Eighty-one patients (8.0%) returned for unplanned visits. Age (P = .82); proximity to MRVAMC (P = .34); and diabetes (P = .60), smoking (P = .55), anxiety (P = .33), or PTSD (P = .37) were not statistically significant predictors of unplanned appointments. Depression diagnosis (P = .04) and female sex (P = .03) were found to be independent risk factors for an unplanned appointment (Table 1). The most common indication for the requested appointment was pain-related, followed closely by noninfectious wound concerns and persistent symptoms (Table 2).


Discussion
Improved access, quality, and efficiency for patients are goals for the VA.1-3 The MRVAMC Plastic and Hand Surgery service provides care for the NFSGVAHS and receives an average of 15 to 20 consultation requests daily. The Veterans Health Administration is frequently challenged by staff shortages, and surgical services struggle to respond to consultation requests and treat patients within reasonable time frames.4,5
The objective of this study was to identify risk factors for unplanned postoperative appointments following elective hand surgery. Unplanned appointments prevent scheduled patients from being seen on time and contribute to backlogs and delays. When patients schedule multiple appointments on the same day, delays in the first clinic’s scheduled appointments create delays for the second and third clinics. Hand surgery clinics can provide a better experience for patients and staff by identifying and mitigating factors prompting unplanned visits.
We anticipated that wound complications would prompt unscheduled visits. Diabetes is a known risk factor for wound healing complications after plastic and hand surgery.6,7 A hemoglobin A1c (HbA1c) screening protocol used by the NFSGVAHS plastic surgery service since 2015 to identify poorly controlled patients with diabetes before surgery may partially explain this finding.8 We did not find a statistically significant difference between patients with diabetes and patients without diabetes for scheduling unplanned appointments. The plastic surgery service does not perform elective hand surgery unless the patient’s HbA1c level is < 9%, or violate the flexor sheath unless HbA1c level is < 8%. However, Zhuang et al found an increase in soft tissue infections after hand surgery with HbA1c levels ≥ 7%.9
Smoking is a potential factor in postoperative hand surgery complications.10,11 Lans et al found an increased incidence of 30-day emergency room visits in current and former smokers after outpatient upper extremity fracture surgery.12 The MRVAMC Plastic Surgery Service counsels patients about the risk of skin necrosis and delayed wound healing, but does not cancel cases or obtain laboratory values to verify abstinence in patients undergoing hand surgery. The VA has multiple resources available for patients interested in smoking cessation through mental health services.13
MRVAMC patients have been known to resist returning for scheduled appointments due to the costs or availability of transportation. We suspected that patients who lived further from MRVAMC would be less likely to return for unscheduled visits. We used the first 3 digits of the patients’ mailing zip code to estimate residential proximity to MRVAMC. An acknowledged limitation to this approach is that some veterans have primary addresses in other regions but still spend significant time in the MRVAMC catchment area and use the facility for their health care during the winter months. These “snowbirds” might reside near the facility despite having official addresses that are more distant. Additionally, there was no increased risk of unplanned visits after hand surgery in patients aged > 61 years (the median age of study participants) (P = .82). Dependence on a third party for transportation in older veterans could impact this finding.
Based on the observation that most patients needed reassurance rather than an intervention when they returned for unscheduled appointments, diagnoses of depression, anxiety, and PTSD were evaluated as separate predictive factors. In previous research, anxiety was found to be a risk factor for problematic recovery following carpal tunnel surgery.14 In the current study, depression was found to be a statistically significant predictor of unscheduled postoperative appointments (P = .04), while anxiety (P = .33) and PTSD (P = .37) were not statistically significant predictors. This is consistent with other studies that have found preexisting depression can predict complications after hand surgery.15,16 Vranceanu et al found that depression predicted pain intensity and disability after elective hand surgery.16 Similarly, Oflazoglu et al found a 12% incidence of depression based on the Patient Health Questionnaire-9 in new and returning hand patients who presented to an academic practice.17 They suggest patients should be assessed at all levels of care and that those with poor responses to surgical or nonsurgical management should be evaluated for depression. MRVAMC has a large mental health service consisting of psychiatrists, psychologists, addiction specialists, social workers, and homeless outreach, and patients tend to already have a diagnosis and mental health practitioner when they present to the clinic.
Recent studies found that wound problems, pain, and stiffness were the most common reasons for return visits.18,19 Shetty et al identified younger age, worse preoperative pain scores, and poor access to transportation as predictors of preventable emergency room visits, which generate higher health care expenditures than an office visit.19 Our study’s top reasons for appointments (pain, wound/scar concerns, persistent symptoms) can be addressed with additional presurgery patient and family education. Additionally, clinicians encourage nonnarcotic pain management strategies including anti-inflammatories, acetaminophen, elevation, splinting, and hand therapy, and the hospital employs experienced, fellowship-trained anesthesia block faculty who help limit perioperative narcotic use. Patients are advised that pain can be used to guide them through the postoperative recovery by preventing overuse and alerting them to a problem that would be masked with narcotics, and long-standing problems such as chronic nerve compressions may continue to cause pain after surgery.
Patients and families can be given consistent and repetitive verbal and written information, instructions, and expectations at the initial consultation, preoperative appointment, and on the day of surgery. Postoperatively, outside their scheduled appointments, patients are encouraged to use the My HealtheVet secure messaging system or call the clinic to access an experienced registered nurse before making a long drive. Access to virtual or phone visits can reduce emergent in-person visits in a VA population.20
Ozdag et al found that 42% of patients who had elective carpal tunnel surgery made unplanned electronic messages or phone contact within 2 weeks postsurgery. The authors point out the uncompensated administrative burden on the staff answering these messages and suggest pre-empting the contacts with more up-front education regarding postoperative pain expectations and management strategies.21
Fisher et al found that attending hand therapy reduced the number of emergency department visits in postoperative infection cases.22 At MRVAMC, a postoperative emergency department visit for a patient prompts an urgent unplanned appointment to the plastic surgery clinic, often on the same day. The MRVAMC occupational therapy clinic employed 3 on-site certified hand therapists during the study period. Because all hand surgery patients at the clinic receive hand therapy on the same day as their first postoperative appointment, attendance at hand therapy was not evaluated as a predictor of unplanned visits. Scheduled hand therapy is another point of contact where the clinic can provide reassurance and patient education.
While females made up 17.1% of the patients in this study, they constituted 12.5% of all veterans in Florida in fiscal year 2023.23 This study found that women were more likely to present for unplanned postoperative appointments (P = .03). This is consistent with existing literature which has found that women are higher users of health care and office-based appointments.24,25 This finding suggests the need for further study into whether our methods of communicating instructions to female patients undergoing plastic surgery may not be optimal.
Strengths and Limitations
As a retrospective review, the authors used information documented by multiple different health care practitioners, including trainees. The electronic medical record problem lists and templates provide consistency of information; however, less seasoned clinicians may interpret what they see and hear differently from more experienced clinicians in the postoperative setting. This study occurred in one part of the country with demographics that may not mirror other VA systems or the general population. The authors hope this study can be a starting point for other health care facilities to investigate ways to minimize the burden of unscheduled appointments. A strength of the study is that it was conducted within a closed system, as patients tend to stay within the VA system and documentation and communication among clinicians, even outside the immediate facility, are easily accessed through the electronic health record.
Conclusions
This study found that depression diagnosis and female sex are statistically significant predictors of unplanned postoperative visits after elective soft tissue hand surgery. More effective patient education during the preoperative period, particularly in patients with depression, may be warranted.
Patients make unplanned appointments after elective soft tissue hand surgery for real or perceived complications when they experience pain, anxiety, or fear. Unplanned appointments can create travel and financial burdens for patients and families. These appointments take time away from scheduled appointments and can contribute to late arrivals and delays in other clinics. Unscheduled appointments contribute to poor access when staff are diverted from scheduled appointments. If predictive factors can be identified, unplanned appointments may either be ameliorated or avoided with better perioperative risk management or education.
Methods
The US Department of Veterans Affairs (VA) North Florida/South Georgia Veterans Health System (NFSGVAHS) and University of Florida Institutional Review Board approved a retrospective chart review of all plastic surgery cases performed at the Malcom Randall VA Medical Center (MRVAMC) and Lake City VAMC operating rooms from July 1, 2018, through December 31, 2019, and January 1, 2021, through June 30, 2022 (nonurgent surgeries were discouraged during the COVID-19 pandemic). Elective soft tissue hand surgery cases were identified based on the operative description found in the Surgical Service Surgeon Staffing Report reviewed monthly by the Service Chief. Potential indicators of unplanned visits were recorded, including age; sex; diagnosis of diabetes, depression, anxiety, or posttraumatic stress disorder (PTSD); current smoking status; and residential zip code. We used the first 3 digits of the patients’ zip codes, which indicate region, as an estimate of proximity to the MRVAMC, which has a 50-county catchment area across North Florida and South Georgia. Diagnoses were found on the “problem list” from the electronic health record documented in the history and physical examinations before surgery. Clinic notes were examined for 3 months postsurgery to identify unplanned postoperative visits and the reason for the appointment. A χ2 analysis was conducted using Excel Version 2402. P < .05 was used to determine whether age (> 60 years), sex, proximity to MRVAMC, diabetes, smoking, depression, anxiety, or PTSD were statistically significant independent risk factors for these appointments.
Results
A total of 1009 elective soft tissue hand surgeries at MRVAMC were reviewed. The patients median age was 61 years. Patients included 173 women (17.1%) and 836 men (82.9%). Eighty-one patients (8.0%) returned for unplanned visits. Age (P = .82); proximity to MRVAMC (P = .34); and diabetes (P = .60), smoking (P = .55), anxiety (P = .33), or PTSD (P = .37) were not statistically significant predictors of unplanned appointments. Depression diagnosis (P = .04) and female sex (P = .03) were found to be independent risk factors for an unplanned appointment (Table 1). The most common indication for the requested appointment was pain-related, followed closely by noninfectious wound concerns and persistent symptoms (Table 2).


Discussion
Improved access, quality, and efficiency for patients are goals for the VA.1-3 The MRVAMC Plastic and Hand Surgery service provides care for the NFSGVAHS and receives an average of 15 to 20 consultation requests daily. The Veterans Health Administration is frequently challenged by staff shortages, and surgical services struggle to respond to consultation requests and treat patients within reasonable time frames.4,5
The objective of this study was to identify risk factors for unplanned postoperative appointments following elective hand surgery. Unplanned appointments prevent scheduled patients from being seen on time and contribute to backlogs and delays. When patients schedule multiple appointments on the same day, delays in the first clinic’s scheduled appointments create delays for the second and third clinics. Hand surgery clinics can provide a better experience for patients and staff by identifying and mitigating factors prompting unplanned visits.
We anticipated that wound complications would prompt unscheduled visits. Diabetes is a known risk factor for wound healing complications after plastic and hand surgery.6,7 A hemoglobin A1c (HbA1c) screening protocol used by the NFSGVAHS plastic surgery service since 2015 to identify poorly controlled patients with diabetes before surgery may partially explain this finding.8 We did not find a statistically significant difference between patients with diabetes and patients without diabetes for scheduling unplanned appointments. The plastic surgery service does not perform elective hand surgery unless the patient’s HbA1c level is < 9%, or violate the flexor sheath unless HbA1c level is < 8%. However, Zhuang et al found an increase in soft tissue infections after hand surgery with HbA1c levels ≥ 7%.9
Smoking is a potential factor in postoperative hand surgery complications.10,11 Lans et al found an increased incidence of 30-day emergency room visits in current and former smokers after outpatient upper extremity fracture surgery.12 The MRVAMC Plastic Surgery Service counsels patients about the risk of skin necrosis and delayed wound healing, but does not cancel cases or obtain laboratory values to verify abstinence in patients undergoing hand surgery. The VA has multiple resources available for patients interested in smoking cessation through mental health services.13
MRVAMC patients have been known to resist returning for scheduled appointments due to the costs or availability of transportation. We suspected that patients who lived further from MRVAMC would be less likely to return for unscheduled visits. We used the first 3 digits of the patients’ mailing zip code to estimate residential proximity to MRVAMC. An acknowledged limitation to this approach is that some veterans have primary addresses in other regions but still spend significant time in the MRVAMC catchment area and use the facility for their health care during the winter months. These “snowbirds” might reside near the facility despite having official addresses that are more distant. Additionally, there was no increased risk of unplanned visits after hand surgery in patients aged > 61 years (the median age of study participants) (P = .82). Dependence on a third party for transportation in older veterans could impact this finding.
Based on the observation that most patients needed reassurance rather than an intervention when they returned for unscheduled appointments, diagnoses of depression, anxiety, and PTSD were evaluated as separate predictive factors. In previous research, anxiety was found to be a risk factor for problematic recovery following carpal tunnel surgery.14 In the current study, depression was found to be a statistically significant predictor of unscheduled postoperative appointments (P = .04), while anxiety (P = .33) and PTSD (P = .37) were not statistically significant predictors. This is consistent with other studies that have found preexisting depression can predict complications after hand surgery.15,16 Vranceanu et al found that depression predicted pain intensity and disability after elective hand surgery.16 Similarly, Oflazoglu et al found a 12% incidence of depression based on the Patient Health Questionnaire-9 in new and returning hand patients who presented to an academic practice.17 They suggest patients should be assessed at all levels of care and that those with poor responses to surgical or nonsurgical management should be evaluated for depression. MRVAMC has a large mental health service consisting of psychiatrists, psychologists, addiction specialists, social workers, and homeless outreach, and patients tend to already have a diagnosis and mental health practitioner when they present to the clinic.
Recent studies found that wound problems, pain, and stiffness were the most common reasons for return visits.18,19 Shetty et al identified younger age, worse preoperative pain scores, and poor access to transportation as predictors of preventable emergency room visits, which generate higher health care expenditures than an office visit.19 Our study’s top reasons for appointments (pain, wound/scar concerns, persistent symptoms) can be addressed with additional presurgery patient and family education. Additionally, clinicians encourage nonnarcotic pain management strategies including anti-inflammatories, acetaminophen, elevation, splinting, and hand therapy, and the hospital employs experienced, fellowship-trained anesthesia block faculty who help limit perioperative narcotic use. Patients are advised that pain can be used to guide them through the postoperative recovery by preventing overuse and alerting them to a problem that would be masked with narcotics, and long-standing problems such as chronic nerve compressions may continue to cause pain after surgery.
Patients and families can be given consistent and repetitive verbal and written information, instructions, and expectations at the initial consultation, preoperative appointment, and on the day of surgery. Postoperatively, outside their scheduled appointments, patients are encouraged to use the My HealtheVet secure messaging system or call the clinic to access an experienced registered nurse before making a long drive. Access to virtual or phone visits can reduce emergent in-person visits in a VA population.20
Ozdag et al found that 42% of patients who had elective carpal tunnel surgery made unplanned electronic messages or phone contact within 2 weeks postsurgery. The authors point out the uncompensated administrative burden on the staff answering these messages and suggest pre-empting the contacts with more up-front education regarding postoperative pain expectations and management strategies.21
Fisher et al found that attending hand therapy reduced the number of emergency department visits in postoperative infection cases.22 At MRVAMC, a postoperative emergency department visit for a patient prompts an urgent unplanned appointment to the plastic surgery clinic, often on the same day. The MRVAMC occupational therapy clinic employed 3 on-site certified hand therapists during the study period. Because all hand surgery patients at the clinic receive hand therapy on the same day as their first postoperative appointment, attendance at hand therapy was not evaluated as a predictor of unplanned visits. Scheduled hand therapy is another point of contact where the clinic can provide reassurance and patient education.
While females made up 17.1% of the patients in this study, they constituted 12.5% of all veterans in Florida in fiscal year 2023.23 This study found that women were more likely to present for unplanned postoperative appointments (P = .03). This is consistent with existing literature which has found that women are higher users of health care and office-based appointments.24,25 This finding suggests the need for further study into whether our methods of communicating instructions to female patients undergoing plastic surgery may not be optimal.
Strengths and Limitations
As a retrospective review, the authors used information documented by multiple different health care practitioners, including trainees. The electronic medical record problem lists and templates provide consistency of information; however, less seasoned clinicians may interpret what they see and hear differently from more experienced clinicians in the postoperative setting. This study occurred in one part of the country with demographics that may not mirror other VA systems or the general population. The authors hope this study can be a starting point for other health care facilities to investigate ways to minimize the burden of unscheduled appointments. A strength of the study is that it was conducted within a closed system, as patients tend to stay within the VA system and documentation and communication among clinicians, even outside the immediate facility, are easily accessed through the electronic health record.
Conclusions
This study found that depression diagnosis and female sex are statistically significant predictors of unplanned postoperative visits after elective soft tissue hand surgery. More effective patient education during the preoperative period, particularly in patients with depression, may be warranted.
- Apaydin EA, Paige NM, Begashaw MM, et al. Veterans Health Administration (VA) vs. non-VA healthcare quality: a systematic review. J Gen Intern Med. 2023;38:2179-2188. doi:10.1007/s11606-023-08207-2
- Blegen M, Ko J, Salzman G, et al. Comparing quality of surgical care between the US Department of Veterans Affairs and non-Veterans Affairs settings: a systematic review. J Am Coll Surg. 2023;237:352-361. doi:10.1097/XCS.0000000000000720
- Valsangkar NP, Eppstein AC, Lawson RA, et al. Effect of lean processes on surgical wait times and efficiency in a tertiary care veterans affairs medical center. JAMA Surg. 2017;152:42-47. doi:10.1001/jamasurg.2016.2808
- National Association of Veterans Affairs Physicians and Dentists. Physicians remain at top of staffing shortage in VA. NAVAPD. December 20, 2023. Accessed March 16, 2026. https://www.navapd.org/news/physicians-remain-at-top-of-staffing-shortage-in-va
- OIG Determination of Veterans Health Administration’s severe occupational staffing shortages fiscal year 2024. Veterans Affairs Office of Inspector General. August 7, 2024. Accessed February 4, 2026. https://www.vaoig.gov/reports/national-healthcare-review/oig-determination-veterans-health-administrations-severe-0
- Goltsman D, Morrison KA, Ascherman JA. Defining the association between diabetes and plastic surgery outcomes: an analysis of nearly 40,000 patients. Plast Reconstr Surg Glob Open. 2017;5:e1461. doi:10.1097/GOX.0000000000001461 7.
- Cox CT, Sierra S, Egan A, et al. Elevated hemoglobin A1c and the risk of postoperative complications in elective hand and upper extremity surgery. Cureus. 2023;15:e48373. doi:10.7759/cureus.48373
- Coady-Fariborzian L, Anstead C. HbA1c and infection in diabetic elective hand surgery: a Veterans Affair Medical Center experience 2012-2018. Hand (NY). 2023;18:994-998. doi:10.1177/1558944720937363<
- Zhuang T, Shapiro LM, Fogel N, et al. Perioperative laboratory markers as risk factors for surgical site infection after elective hand surgery. J Hand Surg Am. 2021;46:675-684. doi:10.1016/j.jhsa.2021.04.001
- Cho BH, Aziz KT, Giladi AM. The impact of smoking on early postoperative complications in hand surgery. J Hand Surg Am. 2021;46:336.e1-336.e11. doi:10.1016/j.jhsa.2020.07.01411.
- Del Core MA, Ahn J, Golden AS, et al. Effect of smoking on short-term postoperative complications after elective upper extremity surgery. Hand (N Y). 2022;17:231-238. doi:10.1177/1558944720926638
- Lans J, Beagles CB, Watkins IT, et al. Unplanned postoperative emergency department visits after upper extremity fracture surgery. J Orthop Trauma. 2025;39:22-27. doi:10.1097/BOT.0000000000002925
- Tobacco and health - how to quit. US Dept of Veterans Affairs. Updated October 29, 2025. Accessed February 4, 2026. https://www.mentalhealth.va.gov/quit-tobacco/how-to-quit.asp
- Ryan C, Miner H, Ramachandran S, et al. General anxiety is associated with problematic initial recovery after carpal tunnel release. Clin Orthop Relat Res. 2022;480:1576-1581. doi:10.1097/CORR.0000000000002115
- Crijns TJ, Bernstein DN, Ring D, et al. Depression and pain interference correlate with physical function in patients recovering from hand surgery. Hand (N Y). 2019;14:830-835. doi:10.1177/1558944718777814
- Vranceanu AM, Jupiter JB, Mudgal CS, et al. Predictors of pain intensity and disability after minor hand surgery. J Hand Surg Am. 2010;35:956-960. doi:10.1016/j.jhsa.2010.02.00117.
- Oflazoglu K, Mellema JJ, Menendez ME, et al. Prevalence of and factors associated with major depression in patients with upper extremity conditions. J Hand Surg Am. 2016;41:263-269. doi:10.1016/j.jhsa.2015.11.019
- Townsend CB, Henry TW, Lutsky KF, et al. Unplanned office visits following outpatient hand surgery. Hand (N Y). 2022;17:1264-1268. doi:10.1177/15589447211028932
- Shetty PN, Guarino GM, Zhang G, et al. Risk factors for preventable emergency department use after outpatient hand surgery. J Hand Surg Am. 2022;47:855-864. doi:10.1016/j.jhsa.2022.05.012
- Sommers-Olson B, Christianson J, Neumann T, et al. Reducing nonemergent visits to the emergency department in a Veterans Affairs multistate system. J Emerg Nurs. 2023;49:539-545. doi:10.1016/j.jen.2023.02.010
- Ozdag Y, Manzar S, El Koussaify J, et al. Unplanned postoperative phone calls and electronic messages for patients with and without opioid prescriptions after carpal tunnel release. J Hand Surg Glob Online. 2024;6:363-368. doi:10.1016/j.jhsg.2024.02.006
- Fisher AH, Gandhi J, Nelson Z, et al. Immediate interventions after surgery to reduce readmission for upper extremity infections. Ann Plast Surg. 2022;88:S163-S169. doi:10.1097/SAP.0000000000003141
- Florida Department of Veterans Affairs Fast Facts. Florida Department of Veterans Affairs. Accessed February 4, 2026. https://floridavets.org/our-veterans/profilefast-facts/
- Bertakis KD, Azari R, Helms LJ, et al. Gender differences in the utilization of health care services. J Fam Pract. 2000;49:147-152.
- Ashman JJ, Santo L, Okeyode T. Characteristics of office-based physician visits, 2018. NCHS Data Brief. 2021;408:1-8.
- Apaydin EA, Paige NM, Begashaw MM, et al. Veterans Health Administration (VA) vs. non-VA healthcare quality: a systematic review. J Gen Intern Med. 2023;38:2179-2188. doi:10.1007/s11606-023-08207-2
- Blegen M, Ko J, Salzman G, et al. Comparing quality of surgical care between the US Department of Veterans Affairs and non-Veterans Affairs settings: a systematic review. J Am Coll Surg. 2023;237:352-361. doi:10.1097/XCS.0000000000000720
- Valsangkar NP, Eppstein AC, Lawson RA, et al. Effect of lean processes on surgical wait times and efficiency in a tertiary care veterans affairs medical center. JAMA Surg. 2017;152:42-47. doi:10.1001/jamasurg.2016.2808
- National Association of Veterans Affairs Physicians and Dentists. Physicians remain at top of staffing shortage in VA. NAVAPD. December 20, 2023. Accessed March 16, 2026. https://www.navapd.org/news/physicians-remain-at-top-of-staffing-shortage-in-va
- OIG Determination of Veterans Health Administration’s severe occupational staffing shortages fiscal year 2024. Veterans Affairs Office of Inspector General. August 7, 2024. Accessed February 4, 2026. https://www.vaoig.gov/reports/national-healthcare-review/oig-determination-veterans-health-administrations-severe-0
- Goltsman D, Morrison KA, Ascherman JA. Defining the association between diabetes and plastic surgery outcomes: an analysis of nearly 40,000 patients. Plast Reconstr Surg Glob Open. 2017;5:e1461. doi:10.1097/GOX.0000000000001461 7.
- Cox CT, Sierra S, Egan A, et al. Elevated hemoglobin A1c and the risk of postoperative complications in elective hand and upper extremity surgery. Cureus. 2023;15:e48373. doi:10.7759/cureus.48373
- Coady-Fariborzian L, Anstead C. HbA1c and infection in diabetic elective hand surgery: a Veterans Affair Medical Center experience 2012-2018. Hand (NY). 2023;18:994-998. doi:10.1177/1558944720937363<
- Zhuang T, Shapiro LM, Fogel N, et al. Perioperative laboratory markers as risk factors for surgical site infection after elective hand surgery. J Hand Surg Am. 2021;46:675-684. doi:10.1016/j.jhsa.2021.04.001
- Cho BH, Aziz KT, Giladi AM. The impact of smoking on early postoperative complications in hand surgery. J Hand Surg Am. 2021;46:336.e1-336.e11. doi:10.1016/j.jhsa.2020.07.01411.
- Del Core MA, Ahn J, Golden AS, et al. Effect of smoking on short-term postoperative complications after elective upper extremity surgery. Hand (N Y). 2022;17:231-238. doi:10.1177/1558944720926638
- Lans J, Beagles CB, Watkins IT, et al. Unplanned postoperative emergency department visits after upper extremity fracture surgery. J Orthop Trauma. 2025;39:22-27. doi:10.1097/BOT.0000000000002925
- Tobacco and health - how to quit. US Dept of Veterans Affairs. Updated October 29, 2025. Accessed February 4, 2026. https://www.mentalhealth.va.gov/quit-tobacco/how-to-quit.asp
- Ryan C, Miner H, Ramachandran S, et al. General anxiety is associated with problematic initial recovery after carpal tunnel release. Clin Orthop Relat Res. 2022;480:1576-1581. doi:10.1097/CORR.0000000000002115
- Crijns TJ, Bernstein DN, Ring D, et al. Depression and pain interference correlate with physical function in patients recovering from hand surgery. Hand (N Y). 2019;14:830-835. doi:10.1177/1558944718777814
- Vranceanu AM, Jupiter JB, Mudgal CS, et al. Predictors of pain intensity and disability after minor hand surgery. J Hand Surg Am. 2010;35:956-960. doi:10.1016/j.jhsa.2010.02.00117.
- Oflazoglu K, Mellema JJ, Menendez ME, et al. Prevalence of and factors associated with major depression in patients with upper extremity conditions. J Hand Surg Am. 2016;41:263-269. doi:10.1016/j.jhsa.2015.11.019
- Townsend CB, Henry TW, Lutsky KF, et al. Unplanned office visits following outpatient hand surgery. Hand (N Y). 2022;17:1264-1268. doi:10.1177/15589447211028932
- Shetty PN, Guarino GM, Zhang G, et al. Risk factors for preventable emergency department use after outpatient hand surgery. J Hand Surg Am. 2022;47:855-864. doi:10.1016/j.jhsa.2022.05.012
- Sommers-Olson B, Christianson J, Neumann T, et al. Reducing nonemergent visits to the emergency department in a Veterans Affairs multistate system. J Emerg Nurs. 2023;49:539-545. doi:10.1016/j.jen.2023.02.010
- Ozdag Y, Manzar S, El Koussaify J, et al. Unplanned postoperative phone calls and electronic messages for patients with and without opioid prescriptions after carpal tunnel release. J Hand Surg Glob Online. 2024;6:363-368. doi:10.1016/j.jhsg.2024.02.006
- Fisher AH, Gandhi J, Nelson Z, et al. Immediate interventions after surgery to reduce readmission for upper extremity infections. Ann Plast Surg. 2022;88:S163-S169. doi:10.1097/SAP.0000000000003141
- Florida Department of Veterans Affairs Fast Facts. Florida Department of Veterans Affairs. Accessed February 4, 2026. https://floridavets.org/our-veterans/profilefast-facts/
- Bertakis KD, Azari R, Helms LJ, et al. Gender differences in the utilization of health care services. J Fam Pract. 2000;49:147-152.
- Ashman JJ, Santo L, Okeyode T. Characteristics of office-based physician visits, 2018. NCHS Data Brief. 2021;408:1-8.
Predictors of Unplanned Postoperative Visits in a Veterans Affairs Hand Surgery Practice
Predictors of Unplanned Postoperative Visits in a Veterans Affairs Hand Surgery Practice
Is Ken Kizer’s Legacy at Risk? The Future of the Veterans Health Administration
Is Ken Kizer’s Legacy at Risk? The Future of the Veterans Health Administration
Mostly it is loss that teaches us about the worth of things.
Arthur Schopenhauer1
One of the leaders I most respected in my US Department of Veterans Affairs (VA) career was a man who had worked his way up through the ranks to become a medical center director. Usually calm, cool, and collected, he would wax poetic when discussing the hero of the VA Health Care System revolution, Ken Kizer, MD, MPH.
In this issue of Federal Practitioner, journalist Randy Dotinga interviews Kizer about the current challenges facing the VA. Though many readers will have participated in or at least know about Kizer’s unprecedented overhaul of the agency, many others, especially those new to VA, may not. It seemed a fitting time to offer an outline of the immense and positive accomplishments that occurred in the agency during his tenure, especially as, under the current administration, many of his most forward-thinking initiatives seem to be moving backward.2
When President Clinton nominated Kizer to serve as the Under Secretary for Health for the Veterans Health Administration in 1994, the poor quality care the agency delivered was castigated in popular movies like Born on the Fourth of July. Veterans who were seen in that era, and who eventually returned to a far better, kinder VA thanks to Kizer, would often tell me, “Doc, the VA was really bad then, and I was afraid to come back.” The critique of VA health care in the mid-1990s sounds like a bureaucratic déjà vu of many of the concerns Kizer raised in his interview, including fragmentation of care, access barriers, and poor coordination of treatment.3
If anyone was prepared and qualified to take on this seeming mission impossible, it was Kizer. A US Navy veteran with 6 board certifications, he came to the VA following a brave and innovative stint as the top health official in California, where he successfully took on the tobacco lobby and dramatically reduced the state’s rates of smoking and related diseases.4
Long before it was the subject of reality TV shows, Kizer dubbed his major renovation of the VA’s antiquated structure an “extreme makeover.”3 Though this description is an oversimplification of Kizer’s monumental efforts, the makeover can be considered in 4 to 6 buckets, depending on how various health policy experts parse the re-engineering efforts.5-7
Decentralization. Kizer instituted the Veterans Integrated Service Network (VISN) system to coordinate the management and operations of all the hospitals, clinics, and other VA health care entities in what is roughly a region. The locus of decision-making shifted from the VA Central Office to the VISNs, intended to promote more efficient, economical, and streamlined health care delivery.
Capitation. Accompanying this restructuring was a shift to a capitated system focused on preventive care. The Veterans Equitable Resource Allocation system was designed to logically link workload and funding. This was a major shift away from VA’s previous emphasis on inpatient and specialty care and resulted in the closing of multiple hospitals.4
Information Systems. I can still remember the first time I sat down at a prehistoric computer to use the Computerized Patient Record System (CPRS). Though now much maligned, then it was like something out of Star Trek, at a time when almost every other health care institution was buried in paper charts. With CPRS, VA suddenly had a pioneering and much-envied electronic medical record that facilitated continuity of care, communication between professionals, and accuracy and completeness of documentation.
Data Driven Performance Improvement. The VISNs and information systems inaugurated a new era of data-driven quality improvement. The assembly and analysis of data enabled VISNs to have real-time input about comparative facility performance.
Performance Measures. The data enabled evidence-based performance measures to be developed and monitored. Though these have now become the bane of many Federal Practitioner readers’ existence, they were originally intended for VISN directors and members of the senior executive service at VA central office. These were tied to incentives that, though recently the subject of watchdog investigation, were intended to motivate and reward high-quality care.6
Even this cursory look at Kizer’s accomplishments is more than enough to demonstrate the magnitude of the makeover, and when the time frame of the achievements is factored in, the transformation is the equivalent of a planet changing its orbit at light speed. Rhetoric aside, there are now hundreds of research articles published in top medical and health policy journals, many of them authored by Kizer,7,8 that have amply demonstrated that when he departed the VA in 1999, it had become “the best care anywhere.” 9 For example, a 2000 New England Journal of Medicine article found that from 1994 to 2000, the percentage of veterans whose care met ≥ 90% of 9 of 17 quality standards was > 70% for 13 of the measures, outperforming fee-for-service Medicare.10
There had been uncertainty about whether Kizer would seek a second term as Under Secretary when he announced that he was leaving. With concise modesty, Kizer said he had met his charge to, “re-engineer the veterans’ health care system so that it could effectively function in the 21st century.”11
Despite openly and critically discussing the many difficulties the VA currently confronts, Kizer ends his interview on a note of hope. Since he likely knows more about VA than any person alive, we need to trust his judgment that his legacy, which currently seems more in jeopardy than ever before, will somehow prevail. Perhaps I am too melancholic, but I believe it will take a professional of the stature of Dr. Kizer to take us back to that future, and I fear we will not see his likes again.
- Schopenhaur A, translated by Saunders TB. Parerga and Paralipomena: A Collection of Philosophical Essays. Cosimo Classics: 2007.
- Spotswood, S. Massive VA restructuring would cut number of VISNS, reduce high-level leadership. U.S. Medicine. January 15, 2026. Accessed March 23, 2026. https://www .usmedicine.com/non-clinical-topics/policy/massive -va-restructuring-would-cut-number-of-visns-reduce-high -level-leadership/
- Kizer KW, Dudley RA. Extreme makeover: transformation of the veterans health ca re system. Annu Rev Public Health. 2009;30:313-339. doi:10.1146/annurev.publhealth.29.020907.090940
- Payne D. How Kizer healed the VA. BMJ. 2012;344:e3324. doi:10.1136/bmj.e3324
- Jha AK. What can the rest of the health care system learn from VA’s quality and safety transformation? Agency for Healthcare Research and Quality. Patient Safety Network. September 1, 2006. Accessed March 23, 2026. https://psnet .ahrq.gov/perspective/what-can-rest-health-care-system -learn-vas-quality-and-safety-transformation
- US Department of Veterans Affairs, Office of Inspector General. VA improperly awarded 10.8 million in incentives to central office senior executives. Report # 23-03773-169. May 9, 2024. Accessed March 23, 2026. https://www.vaoig.gov /reports/administrative-investigation/va-improperly-awarded -108-million-incentives-central-office
- Kizer KW. The “new VA”: a national laboratory for health care quality management. Am J Med Qual. 1999;14:3-20. doi:10.1177/106286069901400103
- Kizer KW, Pane GA. The “new VA”: delivering health care value through integrated service networks. Ann Emerg Med. 1997;30:804-807. doi:10.1016/s0196-0644(97)70053-2
- Longman P. Best Care Anywhere: Why VA Health Care is Better than Yours. 3rd ed. Berrett-Koehler Publishers; 2012.
- Jha AK, Perlin JB, Kizer KW, Dudley RA. Effect of the transformation of the Veterans Affairs Health Care System on the quality of care. N Engl J Med. 2003;348:2218-2227. doi:10.1056/NEJMsa021899
- US Department of Veterans Affairs. Kizer withdraws name from consideration for second term as VA Under Secretary for Health. Vanguard. June/July 1999. Accessed March 23, 2026. https://www.va.gov/opa/publications/archives /vanguard/99junjulvg.pdf
Mostly it is loss that teaches us about the worth of things.
Arthur Schopenhauer1
One of the leaders I most respected in my US Department of Veterans Affairs (VA) career was a man who had worked his way up through the ranks to become a medical center director. Usually calm, cool, and collected, he would wax poetic when discussing the hero of the VA Health Care System revolution, Ken Kizer, MD, MPH.
In this issue of Federal Practitioner, journalist Randy Dotinga interviews Kizer about the current challenges facing the VA. Though many readers will have participated in or at least know about Kizer’s unprecedented overhaul of the agency, many others, especially those new to VA, may not. It seemed a fitting time to offer an outline of the immense and positive accomplishments that occurred in the agency during his tenure, especially as, under the current administration, many of his most forward-thinking initiatives seem to be moving backward.2
When President Clinton nominated Kizer to serve as the Under Secretary for Health for the Veterans Health Administration in 1994, the poor quality care the agency delivered was castigated in popular movies like Born on the Fourth of July. Veterans who were seen in that era, and who eventually returned to a far better, kinder VA thanks to Kizer, would often tell me, “Doc, the VA was really bad then, and I was afraid to come back.” The critique of VA health care in the mid-1990s sounds like a bureaucratic déjà vu of many of the concerns Kizer raised in his interview, including fragmentation of care, access barriers, and poor coordination of treatment.3
If anyone was prepared and qualified to take on this seeming mission impossible, it was Kizer. A US Navy veteran with 6 board certifications, he came to the VA following a brave and innovative stint as the top health official in California, where he successfully took on the tobacco lobby and dramatically reduced the state’s rates of smoking and related diseases.4
Long before it was the subject of reality TV shows, Kizer dubbed his major renovation of the VA’s antiquated structure an “extreme makeover.”3 Though this description is an oversimplification of Kizer’s monumental efforts, the makeover can be considered in 4 to 6 buckets, depending on how various health policy experts parse the re-engineering efforts.5-7
Decentralization. Kizer instituted the Veterans Integrated Service Network (VISN) system to coordinate the management and operations of all the hospitals, clinics, and other VA health care entities in what is roughly a region. The locus of decision-making shifted from the VA Central Office to the VISNs, intended to promote more efficient, economical, and streamlined health care delivery.
Capitation. Accompanying this restructuring was a shift to a capitated system focused on preventive care. The Veterans Equitable Resource Allocation system was designed to logically link workload and funding. This was a major shift away from VA’s previous emphasis on inpatient and specialty care and resulted in the closing of multiple hospitals.4
Information Systems. I can still remember the first time I sat down at a prehistoric computer to use the Computerized Patient Record System (CPRS). Though now much maligned, then it was like something out of Star Trek, at a time when almost every other health care institution was buried in paper charts. With CPRS, VA suddenly had a pioneering and much-envied electronic medical record that facilitated continuity of care, communication between professionals, and accuracy and completeness of documentation.
Data Driven Performance Improvement. The VISNs and information systems inaugurated a new era of data-driven quality improvement. The assembly and analysis of data enabled VISNs to have real-time input about comparative facility performance.
Performance Measures. The data enabled evidence-based performance measures to be developed and monitored. Though these have now become the bane of many Federal Practitioner readers’ existence, they were originally intended for VISN directors and members of the senior executive service at VA central office. These were tied to incentives that, though recently the subject of watchdog investigation, were intended to motivate and reward high-quality care.6
Even this cursory look at Kizer’s accomplishments is more than enough to demonstrate the magnitude of the makeover, and when the time frame of the achievements is factored in, the transformation is the equivalent of a planet changing its orbit at light speed. Rhetoric aside, there are now hundreds of research articles published in top medical and health policy journals, many of them authored by Kizer,7,8 that have amply demonstrated that when he departed the VA in 1999, it had become “the best care anywhere.” 9 For example, a 2000 New England Journal of Medicine article found that from 1994 to 2000, the percentage of veterans whose care met ≥ 90% of 9 of 17 quality standards was > 70% for 13 of the measures, outperforming fee-for-service Medicare.10
There had been uncertainty about whether Kizer would seek a second term as Under Secretary when he announced that he was leaving. With concise modesty, Kizer said he had met his charge to, “re-engineer the veterans’ health care system so that it could effectively function in the 21st century.”11
Despite openly and critically discussing the many difficulties the VA currently confronts, Kizer ends his interview on a note of hope. Since he likely knows more about VA than any person alive, we need to trust his judgment that his legacy, which currently seems more in jeopardy than ever before, will somehow prevail. Perhaps I am too melancholic, but I believe it will take a professional of the stature of Dr. Kizer to take us back to that future, and I fear we will not see his likes again.
Mostly it is loss that teaches us about the worth of things.
Arthur Schopenhauer1
One of the leaders I most respected in my US Department of Veterans Affairs (VA) career was a man who had worked his way up through the ranks to become a medical center director. Usually calm, cool, and collected, he would wax poetic when discussing the hero of the VA Health Care System revolution, Ken Kizer, MD, MPH.
In this issue of Federal Practitioner, journalist Randy Dotinga interviews Kizer about the current challenges facing the VA. Though many readers will have participated in or at least know about Kizer’s unprecedented overhaul of the agency, many others, especially those new to VA, may not. It seemed a fitting time to offer an outline of the immense and positive accomplishments that occurred in the agency during his tenure, especially as, under the current administration, many of his most forward-thinking initiatives seem to be moving backward.2
When President Clinton nominated Kizer to serve as the Under Secretary for Health for the Veterans Health Administration in 1994, the poor quality care the agency delivered was castigated in popular movies like Born on the Fourth of July. Veterans who were seen in that era, and who eventually returned to a far better, kinder VA thanks to Kizer, would often tell me, “Doc, the VA was really bad then, and I was afraid to come back.” The critique of VA health care in the mid-1990s sounds like a bureaucratic déjà vu of many of the concerns Kizer raised in his interview, including fragmentation of care, access barriers, and poor coordination of treatment.3
If anyone was prepared and qualified to take on this seeming mission impossible, it was Kizer. A US Navy veteran with 6 board certifications, he came to the VA following a brave and innovative stint as the top health official in California, where he successfully took on the tobacco lobby and dramatically reduced the state’s rates of smoking and related diseases.4
Long before it was the subject of reality TV shows, Kizer dubbed his major renovation of the VA’s antiquated structure an “extreme makeover.”3 Though this description is an oversimplification of Kizer’s monumental efforts, the makeover can be considered in 4 to 6 buckets, depending on how various health policy experts parse the re-engineering efforts.5-7
Decentralization. Kizer instituted the Veterans Integrated Service Network (VISN) system to coordinate the management and operations of all the hospitals, clinics, and other VA health care entities in what is roughly a region. The locus of decision-making shifted from the VA Central Office to the VISNs, intended to promote more efficient, economical, and streamlined health care delivery.
Capitation. Accompanying this restructuring was a shift to a capitated system focused on preventive care. The Veterans Equitable Resource Allocation system was designed to logically link workload and funding. This was a major shift away from VA’s previous emphasis on inpatient and specialty care and resulted in the closing of multiple hospitals.4
Information Systems. I can still remember the first time I sat down at a prehistoric computer to use the Computerized Patient Record System (CPRS). Though now much maligned, then it was like something out of Star Trek, at a time when almost every other health care institution was buried in paper charts. With CPRS, VA suddenly had a pioneering and much-envied electronic medical record that facilitated continuity of care, communication between professionals, and accuracy and completeness of documentation.
Data Driven Performance Improvement. The VISNs and information systems inaugurated a new era of data-driven quality improvement. The assembly and analysis of data enabled VISNs to have real-time input about comparative facility performance.
Performance Measures. The data enabled evidence-based performance measures to be developed and monitored. Though these have now become the bane of many Federal Practitioner readers’ existence, they were originally intended for VISN directors and members of the senior executive service at VA central office. These were tied to incentives that, though recently the subject of watchdog investigation, were intended to motivate and reward high-quality care.6
Even this cursory look at Kizer’s accomplishments is more than enough to demonstrate the magnitude of the makeover, and when the time frame of the achievements is factored in, the transformation is the equivalent of a planet changing its orbit at light speed. Rhetoric aside, there are now hundreds of research articles published in top medical and health policy journals, many of them authored by Kizer,7,8 that have amply demonstrated that when he departed the VA in 1999, it had become “the best care anywhere.” 9 For example, a 2000 New England Journal of Medicine article found that from 1994 to 2000, the percentage of veterans whose care met ≥ 90% of 9 of 17 quality standards was > 70% for 13 of the measures, outperforming fee-for-service Medicare.10
There had been uncertainty about whether Kizer would seek a second term as Under Secretary when he announced that he was leaving. With concise modesty, Kizer said he had met his charge to, “re-engineer the veterans’ health care system so that it could effectively function in the 21st century.”11
Despite openly and critically discussing the many difficulties the VA currently confronts, Kizer ends his interview on a note of hope. Since he likely knows more about VA than any person alive, we need to trust his judgment that his legacy, which currently seems more in jeopardy than ever before, will somehow prevail. Perhaps I am too melancholic, but I believe it will take a professional of the stature of Dr. Kizer to take us back to that future, and I fear we will not see his likes again.
- Schopenhaur A, translated by Saunders TB. Parerga and Paralipomena: A Collection of Philosophical Essays. Cosimo Classics: 2007.
- Spotswood, S. Massive VA restructuring would cut number of VISNS, reduce high-level leadership. U.S. Medicine. January 15, 2026. Accessed March 23, 2026. https://www .usmedicine.com/non-clinical-topics/policy/massive -va-restructuring-would-cut-number-of-visns-reduce-high -level-leadership/
- Kizer KW, Dudley RA. Extreme makeover: transformation of the veterans health ca re system. Annu Rev Public Health. 2009;30:313-339. doi:10.1146/annurev.publhealth.29.020907.090940
- Payne D. How Kizer healed the VA. BMJ. 2012;344:e3324. doi:10.1136/bmj.e3324
- Jha AK. What can the rest of the health care system learn from VA’s quality and safety transformation? Agency for Healthcare Research and Quality. Patient Safety Network. September 1, 2006. Accessed March 23, 2026. https://psnet .ahrq.gov/perspective/what-can-rest-health-care-system -learn-vas-quality-and-safety-transformation
- US Department of Veterans Affairs, Office of Inspector General. VA improperly awarded 10.8 million in incentives to central office senior executives. Report # 23-03773-169. May 9, 2024. Accessed March 23, 2026. https://www.vaoig.gov /reports/administrative-investigation/va-improperly-awarded -108-million-incentives-central-office
- Kizer KW. The “new VA”: a national laboratory for health care quality management. Am J Med Qual. 1999;14:3-20. doi:10.1177/106286069901400103
- Kizer KW, Pane GA. The “new VA”: delivering health care value through integrated service networks. Ann Emerg Med. 1997;30:804-807. doi:10.1016/s0196-0644(97)70053-2
- Longman P. Best Care Anywhere: Why VA Health Care is Better than Yours. 3rd ed. Berrett-Koehler Publishers; 2012.
- Jha AK, Perlin JB, Kizer KW, Dudley RA. Effect of the transformation of the Veterans Affairs Health Care System on the quality of care. N Engl J Med. 2003;348:2218-2227. doi:10.1056/NEJMsa021899
- US Department of Veterans Affairs. Kizer withdraws name from consideration for second term as VA Under Secretary for Health. Vanguard. June/July 1999. Accessed March 23, 2026. https://www.va.gov/opa/publications/archives /vanguard/99junjulvg.pdf
- Schopenhaur A, translated by Saunders TB. Parerga and Paralipomena: A Collection of Philosophical Essays. Cosimo Classics: 2007.
- Spotswood, S. Massive VA restructuring would cut number of VISNS, reduce high-level leadership. U.S. Medicine. January 15, 2026. Accessed March 23, 2026. https://www .usmedicine.com/non-clinical-topics/policy/massive -va-restructuring-would-cut-number-of-visns-reduce-high -level-leadership/
- Kizer KW, Dudley RA. Extreme makeover: transformation of the veterans health ca re system. Annu Rev Public Health. 2009;30:313-339. doi:10.1146/annurev.publhealth.29.020907.090940
- Payne D. How Kizer healed the VA. BMJ. 2012;344:e3324. doi:10.1136/bmj.e3324
- Jha AK. What can the rest of the health care system learn from VA’s quality and safety transformation? Agency for Healthcare Research and Quality. Patient Safety Network. September 1, 2006. Accessed March 23, 2026. https://psnet .ahrq.gov/perspective/what-can-rest-health-care-system -learn-vas-quality-and-safety-transformation
- US Department of Veterans Affairs, Office of Inspector General. VA improperly awarded 10.8 million in incentives to central office senior executives. Report # 23-03773-169. May 9, 2024. Accessed March 23, 2026. https://www.vaoig.gov /reports/administrative-investigation/va-improperly-awarded -108-million-incentives-central-office
- Kizer KW. The “new VA”: a national laboratory for health care quality management. Am J Med Qual. 1999;14:3-20. doi:10.1177/106286069901400103
- Kizer KW, Pane GA. The “new VA”: delivering health care value through integrated service networks. Ann Emerg Med. 1997;30:804-807. doi:10.1016/s0196-0644(97)70053-2
- Longman P. Best Care Anywhere: Why VA Health Care is Better than Yours. 3rd ed. Berrett-Koehler Publishers; 2012.
- Jha AK, Perlin JB, Kizer KW, Dudley RA. Effect of the transformation of the Veterans Affairs Health Care System on the quality of care. N Engl J Med. 2003;348:2218-2227. doi:10.1056/NEJMsa021899
- US Department of Veterans Affairs. Kizer withdraws name from consideration for second term as VA Under Secretary for Health. Vanguard. June/July 1999. Accessed March 23, 2026. https://www.va.gov/opa/publications/archives /vanguard/99junjulvg.pdf
Is Ken Kizer’s Legacy at Risk? The Future of the Veterans Health Administration
Is Ken Kizer’s Legacy at Risk? The Future of the Veterans Health Administration
Asymptomatic Plaque and Nodule on the Nose
Asymptomatic Plaque and Nodule on the Nose
THE DIAGNOSIS: Coexisting Squamous Cell Carcinoma and Basal Cell Carcinoma
Dermoscopy of the plaque showed central ulceration with blood spots surrounded by branched linear vessels, which was suggestive of squamous cell carcinoma (SCC)(Figure 1A). The nodule showed shiny, white-red, structureless areas with small gray spots, bright white crystalline streaks, and short fine telangiectasias suggestive of basal cell carcinoma (BCC)(Figure 1B). Histopathology showed that the plaque had irregular nests, cords, and sheets of neoplastic keratinocytes invading the dermis (Figure 2A) and the nodule had discrete nests of basaloid cells with peripheral palisading in the dermis (Figure 2B), which confirmed the diagnosis of coexisting SCC and BCC. The patient underwent surgical excision of the lesions, which achieved clear margins. At the 2-year follow-up, there was no sign of recurrence.
Squamous cell carcinoma is the second most frequent cancer in humans. Older patients are more susceptible due to chronic UV exposure.1 Basal cell carcinoma is the most common human cancer worldwide.2 These skin cancers have different clinical manifestations, pathologic features, treatment methods, and prognoses. The coexistence of 2 types of skin cancer presents a diagnostic challenge. Possible causes of this phenomenon are not clear. It may simply be a coincidence since the lesions typically occur in sun-exposed areas such as the nose, which may be affected by photodamage.3 According to the field cancerization theory, chronically sun-exposed areas are at higher risk for development of coexisting skin cancers.4 A more interesting explanation is the interaction theory, which suggests that one tumor produces epidermal or stromal changes that induce the formation of a second independent tumor via the paracrine effect (ie, growth mediators from nearby cells).4
Dermoscopy is an important noninvasive diagnostic tool for the evaluation of skin cancer, particularly early detection. Dermoscopic findings of blood vessels, ulcers, the fiber sign, blood spots, white structureless areas, keratin, and centered vessels indicate a diagnosis of SCC.5 In contrast, common dermoscopic findings for BCC include arborizing vessels, ulceration, shiny white structures, and blue-gray ovoid nests or globules.6
Irritated seborrheic keratosis is an inflammatory variant of seborrheic keratosis, which often is challenging to identify clinically due to its similar features with SCC; however, SCC is more likely to demonstrate dotted or branched vessels, white structureless areas, white circles around follicles, irregular or peripheral vessel patterns, and central scales on dermoscopy. In contrast, irritated seborrheic keratosis is more likely to have hairpin vessels, regular vessel patterns, and white halos around vessels, which may aid in the differentiation between the two entities.6
Due to the higher sensitivity of dermoscopy for detecting pigmented BCC compared to nonpigmented BCC, it holds substantial diagnostic value in Asian populations, in whom pigmented BCC is the most common subtype.6,8 However, the lack of pigmentation in the nodule in our case posed a diagnostic challenge, as the diagnosis of BCC had to rely on subtle vascular and shiny white structures rather than more obvious pigment clues. This absence of pigment, however, also helped rule out pigmented BCC as a diagnosis for the nodule. Short fine telangiectasias is the second most common vascular pattern in BCC, and bright white structures are highly suggestive of nonpigmented BCC.6 Therefore, dermoscopic findings of bright-white structures with fine telangiectasias should be alerted to the possibility of nonpigmented BCC.
Basosquamous carcinoma has clinical and dermoscopic features between SCC and BCC, and the presence of dermatoscopic features from both BCC and SCC should raise suspicion, but the diagnosis is particularly challenging because its presentation is nonspecific.9 We need to be vigilant about the possibility of coexistence of 2 types of skin cancer, and that regular physical examination and dermatoscopy are very important for early detection and diagnosis.
- Corchado-Cobos R, García-Sancha N, González-Sarmiento R, et al. Cutaneous squamous cell carcinoma: from biology to therapy. Int J Mol Sci. 2020;21:2956. doi:10.3390/ijms21082956
- Cameron MC, Lee E, Hibler BP, et al. Basal cell carcinoma: epidemiology; pathophysiology; clinical and histological subtypes; and disease associations. J Am Acad Dermatol. 2019;80:303-317. doi:10.1016/j.jaad.2018.03.060
- Kraemer KH, Lee MM, Scotto J. Xeroderma pigmentosum. Cutaneous, ocular, and neurologic abnormalities in 830 published cases. Arch Dermatol. 1987;123:241-250. doi:10.1001/archderm.123.2.241
- Cornejo KM, Deng AC. Malignant melanoma within squamous cell carcinoma and basal cell carcinoma: is it a combined or collision tumor? a case report and review of the literature. Am J Dermatopathol. 2013;35:226-34. doi:10.1097/DAD.0b013e3182545e27
- Ertop Dog˘an P, Akay BN, Okçu Heper A, et al. Dermatoscopic findings and dermatopathological correlates in clinical variants of actinic keratosis, Bowen’s disease, keratoacanthoma, and squamous cell carcinoma. Dermatol Ther. 2021;34:E14877. doi:10.1111/dth.14877.
- Álvarez-Salafranca M, Ara M, Zaballos P. Dermoscopy in basal cell carcinoma: an updated review. Actas Dermosifiliogr (Engl Ed). 2021;112:330-338. doi:10.1016/j.ad.2020.11.011
- Papageorgiou C, Spyridis I, Manoli SM, et al. Accuracy of dermoscopic criteria for the differential diagnosis between irritated seborrheic keratosis and squamous cell carcinoma. J Am Acad Dermatol. 2021;85:1143-1150. doi:10.1016/j.jaad.2020.02.019
- Cheng SY, Luk NM, Chong LY. Special features of non-melanoma skin cancer in Hong Kong Chinese patients: 10-year retrospective study. Hong Kong Med J. 2001;7:22-28.
- Murgia G, Denaro N, Boggio F, et al. Basosquamous carcinoma: comprehensive clinical and histopathological aspects, novel imaging tools, and therapeutic approaches. Cells. 2023;23:2737. doi:10.3390/cells12232737
THE DIAGNOSIS: Coexisting Squamous Cell Carcinoma and Basal Cell Carcinoma
Dermoscopy of the plaque showed central ulceration with blood spots surrounded by branched linear vessels, which was suggestive of squamous cell carcinoma (SCC)(Figure 1A). The nodule showed shiny, white-red, structureless areas with small gray spots, bright white crystalline streaks, and short fine telangiectasias suggestive of basal cell carcinoma (BCC)(Figure 1B). Histopathology showed that the plaque had irregular nests, cords, and sheets of neoplastic keratinocytes invading the dermis (Figure 2A) and the nodule had discrete nests of basaloid cells with peripheral palisading in the dermis (Figure 2B), which confirmed the diagnosis of coexisting SCC and BCC. The patient underwent surgical excision of the lesions, which achieved clear margins. At the 2-year follow-up, there was no sign of recurrence.


Squamous cell carcinoma is the second most frequent cancer in humans. Older patients are more susceptible due to chronic UV exposure.1 Basal cell carcinoma is the most common human cancer worldwide.2 These skin cancers have different clinical manifestations, pathologic features, treatment methods, and prognoses. The coexistence of 2 types of skin cancer presents a diagnostic challenge. Possible causes of this phenomenon are not clear. It may simply be a coincidence since the lesions typically occur in sun-exposed areas such as the nose, which may be affected by photodamage.3 According to the field cancerization theory, chronically sun-exposed areas are at higher risk for development of coexisting skin cancers.4 A more interesting explanation is the interaction theory, which suggests that one tumor produces epidermal or stromal changes that induce the formation of a second independent tumor via the paracrine effect (ie, growth mediators from nearby cells).4
Dermoscopy is an important noninvasive diagnostic tool for the evaluation of skin cancer, particularly early detection. Dermoscopic findings of blood vessels, ulcers, the fiber sign, blood spots, white structureless areas, keratin, and centered vessels indicate a diagnosis of SCC.5 In contrast, common dermoscopic findings for BCC include arborizing vessels, ulceration, shiny white structures, and blue-gray ovoid nests or globules.6
Irritated seborrheic keratosis is an inflammatory variant of seborrheic keratosis, which often is challenging to identify clinically due to its similar features with SCC; however, SCC is more likely to demonstrate dotted or branched vessels, white structureless areas, white circles around follicles, irregular or peripheral vessel patterns, and central scales on dermoscopy. In contrast, irritated seborrheic keratosis is more likely to have hairpin vessels, regular vessel patterns, and white halos around vessels, which may aid in the differentiation between the two entities.6
Due to the higher sensitivity of dermoscopy for detecting pigmented BCC compared to nonpigmented BCC, it holds substantial diagnostic value in Asian populations, in whom pigmented BCC is the most common subtype.6,8 However, the lack of pigmentation in the nodule in our case posed a diagnostic challenge, as the diagnosis of BCC had to rely on subtle vascular and shiny white structures rather than more obvious pigment clues. This absence of pigment, however, also helped rule out pigmented BCC as a diagnosis for the nodule. Short fine telangiectasias is the second most common vascular pattern in BCC, and bright white structures are highly suggestive of nonpigmented BCC.6 Therefore, dermoscopic findings of bright-white structures with fine telangiectasias should be alerted to the possibility of nonpigmented BCC.
Basosquamous carcinoma has clinical and dermoscopic features between SCC and BCC, and the presence of dermatoscopic features from both BCC and SCC should raise suspicion, but the diagnosis is particularly challenging because its presentation is nonspecific.9 We need to be vigilant about the possibility of coexistence of 2 types of skin cancer, and that regular physical examination and dermatoscopy are very important for early detection and diagnosis.
THE DIAGNOSIS: Coexisting Squamous Cell Carcinoma and Basal Cell Carcinoma
Dermoscopy of the plaque showed central ulceration with blood spots surrounded by branched linear vessels, which was suggestive of squamous cell carcinoma (SCC)(Figure 1A). The nodule showed shiny, white-red, structureless areas with small gray spots, bright white crystalline streaks, and short fine telangiectasias suggestive of basal cell carcinoma (BCC)(Figure 1B). Histopathology showed that the plaque had irregular nests, cords, and sheets of neoplastic keratinocytes invading the dermis (Figure 2A) and the nodule had discrete nests of basaloid cells with peripheral palisading in the dermis (Figure 2B), which confirmed the diagnosis of coexisting SCC and BCC. The patient underwent surgical excision of the lesions, which achieved clear margins. At the 2-year follow-up, there was no sign of recurrence.


Squamous cell carcinoma is the second most frequent cancer in humans. Older patients are more susceptible due to chronic UV exposure.1 Basal cell carcinoma is the most common human cancer worldwide.2 These skin cancers have different clinical manifestations, pathologic features, treatment methods, and prognoses. The coexistence of 2 types of skin cancer presents a diagnostic challenge. Possible causes of this phenomenon are not clear. It may simply be a coincidence since the lesions typically occur in sun-exposed areas such as the nose, which may be affected by photodamage.3 According to the field cancerization theory, chronically sun-exposed areas are at higher risk for development of coexisting skin cancers.4 A more interesting explanation is the interaction theory, which suggests that one tumor produces epidermal or stromal changes that induce the formation of a second independent tumor via the paracrine effect (ie, growth mediators from nearby cells).4
Dermoscopy is an important noninvasive diagnostic tool for the evaluation of skin cancer, particularly early detection. Dermoscopic findings of blood vessels, ulcers, the fiber sign, blood spots, white structureless areas, keratin, and centered vessels indicate a diagnosis of SCC.5 In contrast, common dermoscopic findings for BCC include arborizing vessels, ulceration, shiny white structures, and blue-gray ovoid nests or globules.6
Irritated seborrheic keratosis is an inflammatory variant of seborrheic keratosis, which often is challenging to identify clinically due to its similar features with SCC; however, SCC is more likely to demonstrate dotted or branched vessels, white structureless areas, white circles around follicles, irregular or peripheral vessel patterns, and central scales on dermoscopy. In contrast, irritated seborrheic keratosis is more likely to have hairpin vessels, regular vessel patterns, and white halos around vessels, which may aid in the differentiation between the two entities.6
Due to the higher sensitivity of dermoscopy for detecting pigmented BCC compared to nonpigmented BCC, it holds substantial diagnostic value in Asian populations, in whom pigmented BCC is the most common subtype.6,8 However, the lack of pigmentation in the nodule in our case posed a diagnostic challenge, as the diagnosis of BCC had to rely on subtle vascular and shiny white structures rather than more obvious pigment clues. This absence of pigment, however, also helped rule out pigmented BCC as a diagnosis for the nodule. Short fine telangiectasias is the second most common vascular pattern in BCC, and bright white structures are highly suggestive of nonpigmented BCC.6 Therefore, dermoscopic findings of bright-white structures with fine telangiectasias should be alerted to the possibility of nonpigmented BCC.
Basosquamous carcinoma has clinical and dermoscopic features between SCC and BCC, and the presence of dermatoscopic features from both BCC and SCC should raise suspicion, but the diagnosis is particularly challenging because its presentation is nonspecific.9 We need to be vigilant about the possibility of coexistence of 2 types of skin cancer, and that regular physical examination and dermatoscopy are very important for early detection and diagnosis.
- Corchado-Cobos R, García-Sancha N, González-Sarmiento R, et al. Cutaneous squamous cell carcinoma: from biology to therapy. Int J Mol Sci. 2020;21:2956. doi:10.3390/ijms21082956
- Cameron MC, Lee E, Hibler BP, et al. Basal cell carcinoma: epidemiology; pathophysiology; clinical and histological subtypes; and disease associations. J Am Acad Dermatol. 2019;80:303-317. doi:10.1016/j.jaad.2018.03.060
- Kraemer KH, Lee MM, Scotto J. Xeroderma pigmentosum. Cutaneous, ocular, and neurologic abnormalities in 830 published cases. Arch Dermatol. 1987;123:241-250. doi:10.1001/archderm.123.2.241
- Cornejo KM, Deng AC. Malignant melanoma within squamous cell carcinoma and basal cell carcinoma: is it a combined or collision tumor? a case report and review of the literature. Am J Dermatopathol. 2013;35:226-34. doi:10.1097/DAD.0b013e3182545e27
- Ertop Dog˘an P, Akay BN, Okçu Heper A, et al. Dermatoscopic findings and dermatopathological correlates in clinical variants of actinic keratosis, Bowen’s disease, keratoacanthoma, and squamous cell carcinoma. Dermatol Ther. 2021;34:E14877. doi:10.1111/dth.14877.
- Álvarez-Salafranca M, Ara M, Zaballos P. Dermoscopy in basal cell carcinoma: an updated review. Actas Dermosifiliogr (Engl Ed). 2021;112:330-338. doi:10.1016/j.ad.2020.11.011
- Papageorgiou C, Spyridis I, Manoli SM, et al. Accuracy of dermoscopic criteria for the differential diagnosis between irritated seborrheic keratosis and squamous cell carcinoma. J Am Acad Dermatol. 2021;85:1143-1150. doi:10.1016/j.jaad.2020.02.019
- Cheng SY, Luk NM, Chong LY. Special features of non-melanoma skin cancer in Hong Kong Chinese patients: 10-year retrospective study. Hong Kong Med J. 2001;7:22-28.
- Murgia G, Denaro N, Boggio F, et al. Basosquamous carcinoma: comprehensive clinical and histopathological aspects, novel imaging tools, and therapeutic approaches. Cells. 2023;23:2737. doi:10.3390/cells12232737
- Corchado-Cobos R, García-Sancha N, González-Sarmiento R, et al. Cutaneous squamous cell carcinoma: from biology to therapy. Int J Mol Sci. 2020;21:2956. doi:10.3390/ijms21082956
- Cameron MC, Lee E, Hibler BP, et al. Basal cell carcinoma: epidemiology; pathophysiology; clinical and histological subtypes; and disease associations. J Am Acad Dermatol. 2019;80:303-317. doi:10.1016/j.jaad.2018.03.060
- Kraemer KH, Lee MM, Scotto J. Xeroderma pigmentosum. Cutaneous, ocular, and neurologic abnormalities in 830 published cases. Arch Dermatol. 1987;123:241-250. doi:10.1001/archderm.123.2.241
- Cornejo KM, Deng AC. Malignant melanoma within squamous cell carcinoma and basal cell carcinoma: is it a combined or collision tumor? a case report and review of the literature. Am J Dermatopathol. 2013;35:226-34. doi:10.1097/DAD.0b013e3182545e27
- Ertop Dog˘an P, Akay BN, Okçu Heper A, et al. Dermatoscopic findings and dermatopathological correlates in clinical variants of actinic keratosis, Bowen’s disease, keratoacanthoma, and squamous cell carcinoma. Dermatol Ther. 2021;34:E14877. doi:10.1111/dth.14877.
- Álvarez-Salafranca M, Ara M, Zaballos P. Dermoscopy in basal cell carcinoma: an updated review. Actas Dermosifiliogr (Engl Ed). 2021;112:330-338. doi:10.1016/j.ad.2020.11.011
- Papageorgiou C, Spyridis I, Manoli SM, et al. Accuracy of dermoscopic criteria for the differential diagnosis between irritated seborrheic keratosis and squamous cell carcinoma. J Am Acad Dermatol. 2021;85:1143-1150. doi:10.1016/j.jaad.2020.02.019
- Cheng SY, Luk NM, Chong LY. Special features of non-melanoma skin cancer in Hong Kong Chinese patients: 10-year retrospective study. Hong Kong Med J. 2001;7:22-28.
- Murgia G, Denaro N, Boggio F, et al. Basosquamous carcinoma: comprehensive clinical and histopathological aspects, novel imaging tools, and therapeutic approaches. Cells. 2023;23:2737. doi:10.3390/cells12232737
Asymptomatic Plaque and Nodule on the Nose
Asymptomatic Plaque and Nodule on the Nose
An 80-year-old Asian woman presented to the hospital dermatology clinic for evaluation of 2 lesions on the nose of 2 years’ duration. The patient reported that the lesions had initially manifested as an asymptomatic red rash, but within the past month, the affected area had grown in diameter with a rough surface and occasional bleeding. Additionally, a smaller new rash appeared around the original plaque. She had no personal or family history of skin cancer. Physical examination revealed a 1.5-cm reddish plaque on the tip of the nose with a central ulcer filled with viscous exudate. Another 0.5-cm reddish nodule with a smooth surface also was noted adjacent to the plaque. Dermoscopy and a skin biopsy of both lesions were performed.