User login
Thermal Therapy is Associated With Decreased Pain Treatment Use: A Retrospective Cohort Study
Thermal Therapy is Associated With Decreased Pain Treatment Use: A Retrospective Cohort Study
Growing recognition of the limitations of long-term opioid therapy for chronic noncancer pain has highlighted the importance of nonpharmacologic approaches to pain treatment.1,2 These treatments are varied and may include psychological and behavioral therapies (eg, cognitive behavioral therapy for chronic pain), exercise and movement therapies (eg, yoga), and manual therapies (eg, chiropractic). Body surface cold therapy, while predominantly used to reduce postoperative pain and inflammation,3,4 pain subsequent to acute musculoskeletal injury,5 and pain related to muscle soreness,6 is a nonpharmacologic treatment that has shown pain-reducing benefits for chronic low back pain and knee osteoarthritis, as has its counterpart, superficial heat therapy.7-9 Heat therapy has also been shown to improve strength, flexibility, and activities of daily living in patients with chronic low back pain.10,11 Cold and heat therapies are commonly used complementarily. Cold therapies aim to reduce blood flow and inflammation and are often used immediately following trauma to an affected area, whereas heat therapies increase blood flow and metabolic activity and are commonly used to promote healing.5
Heat and cold therapies (also known as thermal therapies) benefit resource-limited health care systems, as most devices require a single expenditure and can be self-administered by patients at home as part of their pain self-management plan. In addition, these pain self-management tools may attenuate the need for more expensive specialty pain care and ongoing analgesic pharmacotherapy. Despite their potential, few studies have characterized the benefits of thermal therapies for patients with heterogeneous chronic pain syndromes.
The purpose of this retrospective cohort study was to examine the potential clinical and health system benefits of patient-administered thermal therapy. Our primary hypothesis was that patients with chronic musculoskeletal pain who received a thermal therapy self-management device would have fewer days of opioid prescriptions compared with a sample of matched control patients. Secondarily, we hypothesized that patients who received a thermal therapy device would have lower utilization of specialty pain care, fewer potentially hazardous opioid prescriptions (eg, high-dose opioid therapy and concurrent opioid and benzodiazepine prescriptions), fewer prescriptions for nonopioid analgesic medications, and decreased pain intensity when compared with matched controls.
Methods
This retrospective cohort study compared pain pharmacotherapy, pain treatment utilization, and pain intensity outcomes between patients who received a thermal therapy device and matched patients who did not. The study was approved by the US Department of Veterans Affairs (VA) Portland Health Care System Institutional Review Board and was granted a waiver of informed consent to access patient electronic health records (EHRs).
Pain Care
The VA uses ThermaZone thermal therapy devices (Innovative Medical Equipment) for chronic pain treatment. The device uses thermoelectric technology to provide point-of-contact cooling and heating therapy through site-specific pads (eg, ankle, knee, hip, back, elbow, shoulder). Patients place pads on sites where they experience pain, and temperature regulated water circulates through the device and to the pad, providing consistent, localized thermal therapy. The pads range in temperature from 1 °C to 52 °C, and temperatures are self-monitored and controlled by the patient.
Standard pain care in this study followed the VA stepped model of pain care, which builds on a foundation of patient education for pain self-management approaches (eg, exercise, mindfulness, relaxation, social support).12 According to the VA stepped model of pain care, all patients with chronic pain should engage in these foundational self-management approaches. However, some patients require more intensive care. The VA stepped-up treatment engages primary and specialty care services—such as physical therapy, pharmacy, complementary and integrative health approaches, mental health, and substance use services—and, when indicated, may escalate treatment to interdisciplinary pain teams or tertiary pain centers. In this retrospective cohort study, treatment patients received a thermal therapy device and standard of care, whereas control patients received standard care only.
Sample Selection
Eligible patients were aged ≥ 18 years, had a musculoskeletal pain diagnosis documented in the EHR in the year prior to thermal therapy device receipt (or during the same period for a treatment patient’s matched control), and were enrolled in VA health care during the study period. Patients who died during the study period were excluded. Treatment patients received a thermal therapy device from the VA between January 1, 2017, and December 31, 2018, when complete data on thermal therapy devices were available. For control patients, the VA Corporate Data Warehouse (CDW) was used to identify VA patients with characteristics similar to those of treated patients.
We modeled the probability that a patient would receive a thermal therapy device using logistic regression. Predictor variables were measured in the year prior to device receipt and included variables associated with pain treatment utilization and analgesic pharmacotherapy receipt, as recommended by Brookhart et al.13 These included age, sex, race, ethnicity, VA service-connected disability status, comorbidities, receipt of medications for opioid use disorder, pain diagnoses, mental health diagnoses, and substance use disorder diagnoses.14-19
The resulting propensity scores (eg, predicted probabilities) were used to match treatment patients 1:1 with control patients using a nearest-neighbor matching algorithm.20 This procedure matches a control patient with the closest propensity score to that of the corresponding treatment patient. An exact match on VA facility was required (eg, treatment patients and matched controls received care at the same VA facility). Standardized differences were used to assess covariate balance between the matched groups, and kernel density plots of propensity scores tested for sufficient overlap.21 Control patients were selected from a pool of 1,150,149 patients.
Study Variables
The index date was the date the thermal therapy device was released to treatment patients or the same date for the matched controls. Data were extracted from the CDW over a 24-month period: 12 months prior to the index date through 12 months afterwards. Collecting data in the 12 months prior to treatment initiation allowed us to adjust for covariates and provided greater precision, as recommended for observational study designs.22
Treatment conditions were defined dichotomously as receipt vs nonreceipt of a thermal therapy device. The primary outcome was the number of days of opioid use in the 12 months following the index date. Additional outcomes included days of high-dose opioid therapy (≥ 50 mg morphine equivalent [MME] daily), concurrent opioid and benzodiazepine prescriptions, and nonopioid analgesic pharmacotherapy (eg, nonsteroidal anti-inflammatory drugs, acetaminophen, muscle relaxants). All prescription data were based on medication fills obtained from VA pharmacy records. Additional outcomes included the number of visits to physical therapy, occupational therapy, physical medicine and rehabilitation, and interdisciplinary pain clinics, including interventional pain medicine.
Pain intensity ratings were collected as part of routine VA care using a numeric scale from 0 (no pain) to 10 (worst possible pain). Pain intensity trajectories were computed using all available EHR-derived pain intensity score data for each patient in the 12 months prior to and following the index date.
Covariates were extracted from the EHR and evaluated in the year prior to the index date, unless otherwise noted. They included age at the index date; self-reported sex, and race and ethnicity; service-connected disability status (disability awarded as a result of military service-related trauma or injury); Charlson Comorbidity Index; and diagnoses of opioid use disorder, alcohol use disorder, other substance use disorder, mood disorder, posttraumatic stress disorder, other anxiety disorder, psychotic disorder, neuropathic pain, and headache pain.23 All diagnoses were coded as yes if identified in the EHR as a focus of treatment during any clinical encounter in the year prior to the index date or no if not identified as a focus of treatment.
The number of days patients had been living with pain was calculated and defined as the number of days from the first pain diagnosis available in a patient’s EHR to the index date. Finally, the number of pain-related surgeries (eg, surgeries with ≥ 1 pain diagnoses associated with the clinical encounter) and average pain intensity were computed for the pre- and postindex date evaluation periods and included as model covariates.
Statistical Analyses
For the 4 pharmacotherapy and 4 nonpharmacologic treatment usage outcomes, we tested the fit of linear models and several models with count distributions using the Bayesian information criterion.24 Count distributions included Poisson, zero-inflated Poisson, negative binomial, and zero-inflated negative binomial. With 1 exception (days of high-dose opioid use), a negative binomial distribution best fit the data. For days of high-dose opioid use, a Gaussian distribution best fit the data.
Eight separate mixed-effects regression analyses compared changes in each outcome from the 12-month preindex period through the 12-month postindex period between treatment and control patients by testing the Time × Treatment interaction. This approach statistically accounts for observed pretreatment differences in outcome variables. Statistics for the main effects of time and treatment are also presented. To reduce bias, models controlled for covariates specified previously.
For pain intensity, we used random-effects growth modeling to quantify both fixed and random effects of pain intensity at the index date (eg, the model intercept), which estimates pain at the time of treatment initiation, and change in pain during the 12 months following the index date (eg, the model slope), which characterizes the trajectory of pain intensity ratings.25 The model included piecewise components of pain score trajectories in the 12 months prior to the index date and in the 12 months following the index date. Several types of change for the 12-month postindex observation period were explored—including quadratic and cubic curvilinear change. A linear model for change in pain over time provided the best fit based on the Bayesian information criterion and parsimony of model parameters.26 We report estimates of change in pain over time in monthly intervals for ease of interpretation. However, models used all individual pain scores rather than computing monthly averages when > 1 pain score was available within a month, as recommended in previous research.27 This approach makes optimal use of all available data. Both random effects (intercept and slope) were regressed onto the set of covariates described previously.
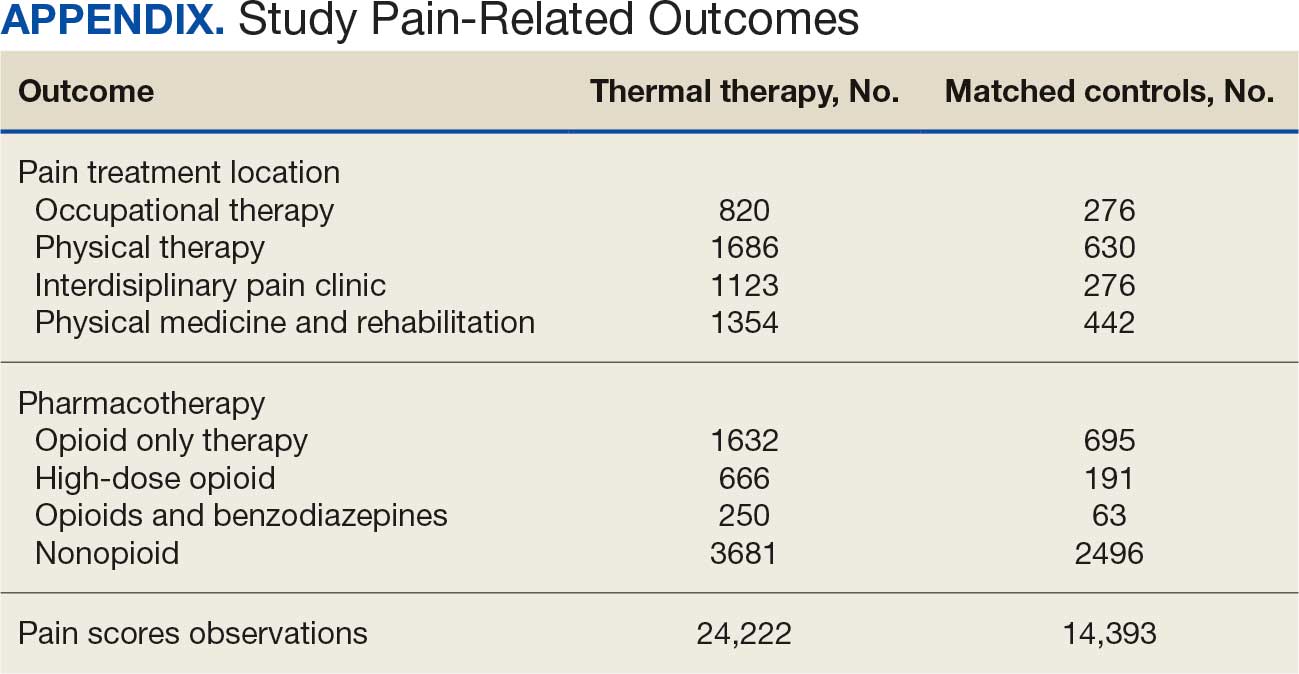
This study used data available in the EHR over the 24-month observation period. We characterize the density (eg, frequency) of all outcome variables by treatment condition in the Appendix. Because the hypotheses were directional, the authors used a 2-sided α = 0.10 and applied a Bonferroni correction for the 9 statistical tests performed, resulting in an adjusted α of 0.01. Treatment utilization and pharmacotherapy outcome analyses were performed in Stata, version 16.1. Random-effects growth modeling of pain score trajectories was performed using Mplus, version 8.8.
Results
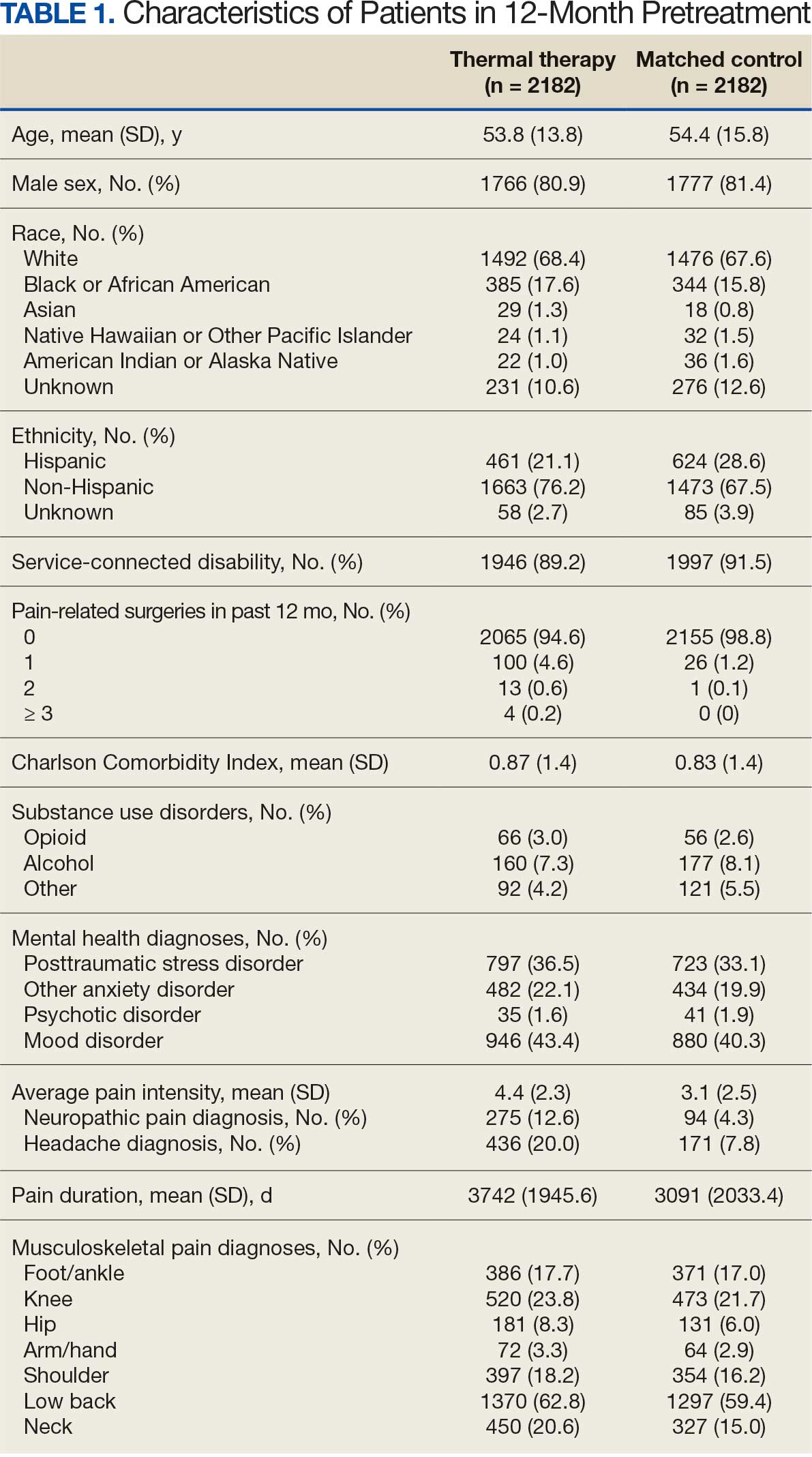
There were 2182 patients in the treatment group and 2182 matched controls. The mean (SD) age was 54 (15) years; 81% were male, and about two-thirds (68%) identified as White and non-Hispanic. Mental health comorbidities were common, with > 40% of the sample having diagnoses of a mood disorder and/or posttraumatic stress disorder. Nearly all patients (90%) had VA service-connected disability ratings. Among patients with chronic musculoskeletal pain, 8% had comorbid neuropathic pain and 14% had headache. The mean (SD) duration of chronic pain across both patient groups was 3416 (2016) days, or about 9.4 years (Table 1).
Pharmacotherapy
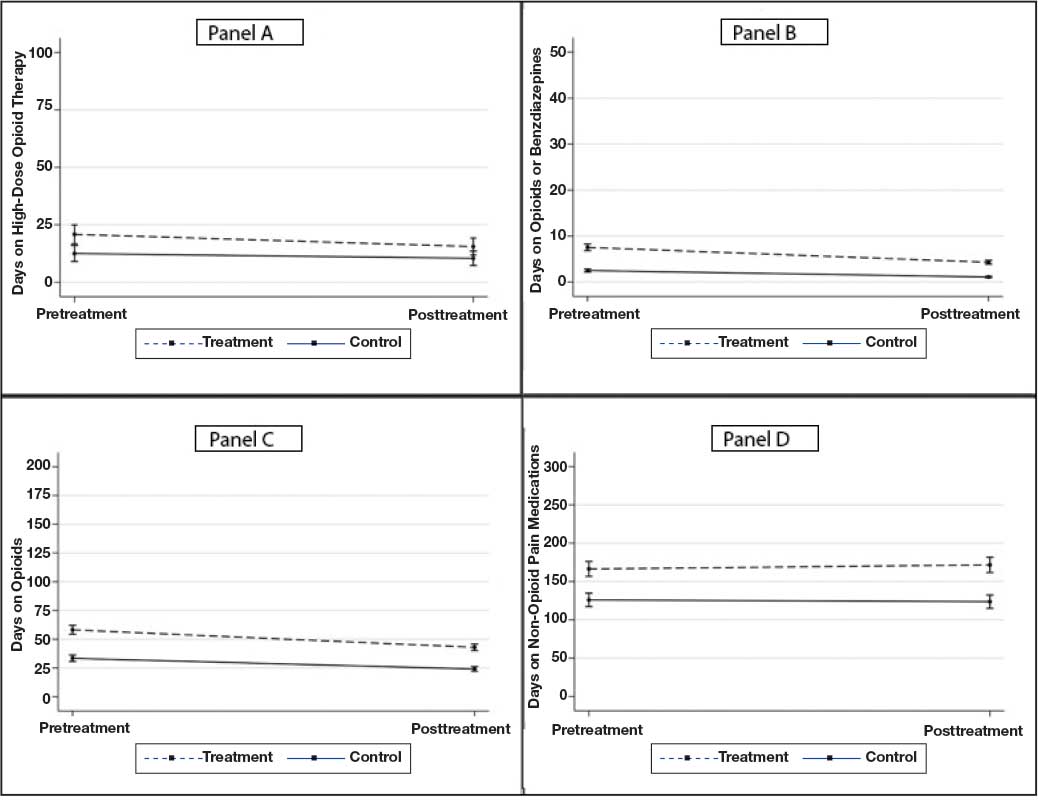
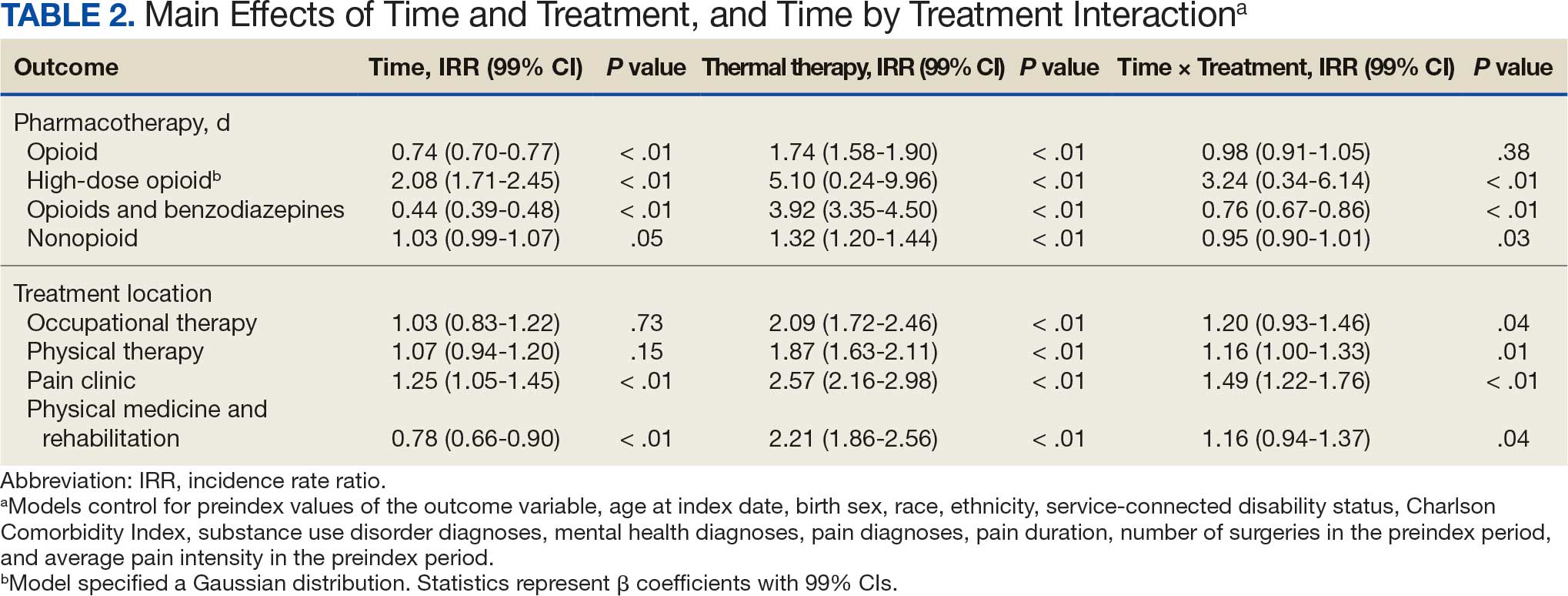
High-dose opioid use (> 50 MME daily dose), days of opioid use, and concurrent opioid and benzodiazepine use decreased for all patients from the pre- to posttreatment period (Table 2). However, high-dose opioid use (Time × Treatment interaction, 3.24; 99% CI, 0.34 to 6.14) (Figure 1A) and concurrent opioid and benzodiazepine use (Time × Treatment interaction, 0.76; 99% CI, 0.67 to 0.86) (Figure 1B) had a larger decrease for the treatment vs matched control group. Treatment and matched control patients had comparable reductions in days of opioid use (Time × Treatment interaction, 0.98; 99% CI, 0.91 to 1.05) (Figure 1C). Neither group showed changes in nonopioid analgesic pharmacotherapy over time (main effect of time, incidence rate ratio [IRR], 1.03; 99% CI, 0.99 to 1.07; Time × Treatment interaction, 0.95; 99% CI, 0.90 to 1.01) (Figure 1D).
Nonpharmacologic Pain Treatment
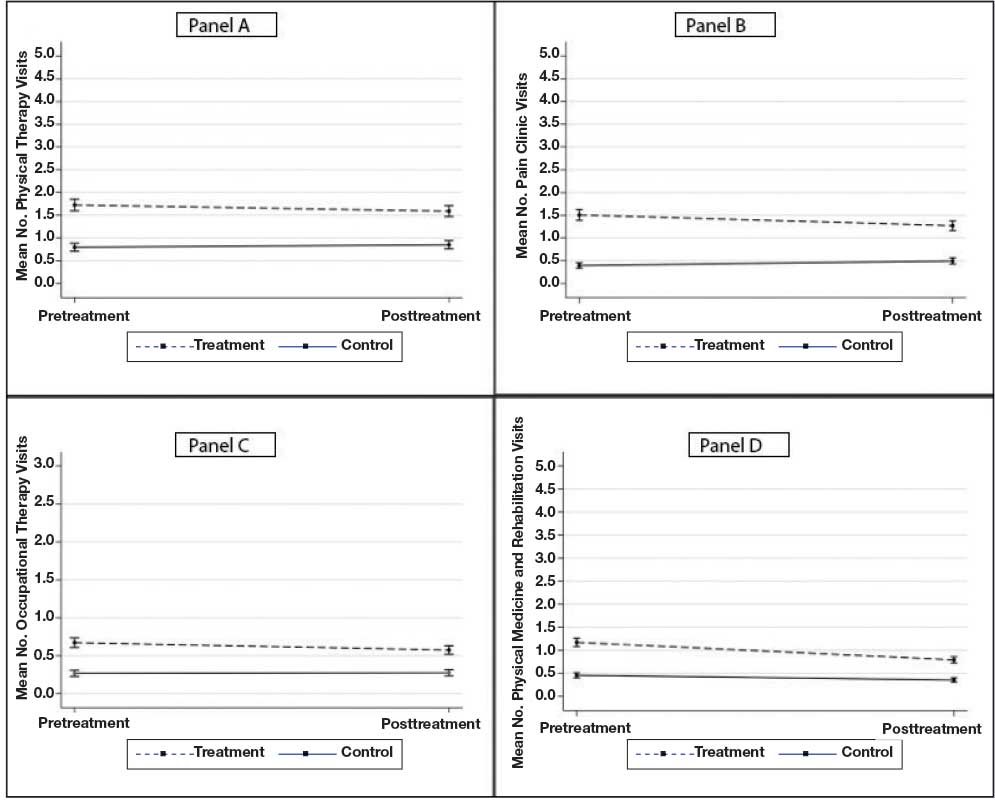
The number of physical therapy and pain clinic visits declined for treatment patients and increased slightly for matched control patients (Figure 2A and 2B). For occupational therapy visits, neither group showed changes over time (main effect of time IRR, 1.03; 99% CI, 0.83 to 1.22; Time × Treatment interaction, 1.20; 99% CI, 0.93 to 1.46) (Figure 2C). For physical medicine and rehabilitation visits, both groups decreased use over time (main effect of time IRR, 0.78; 99% CI, 0.66 to 0.90), but this decrease did not differ between treatment and control patients (Time × Treatment interaction, 1.16; 99% CI, 0.94 to 1.37) (Figure 2D).
Pain Intensity
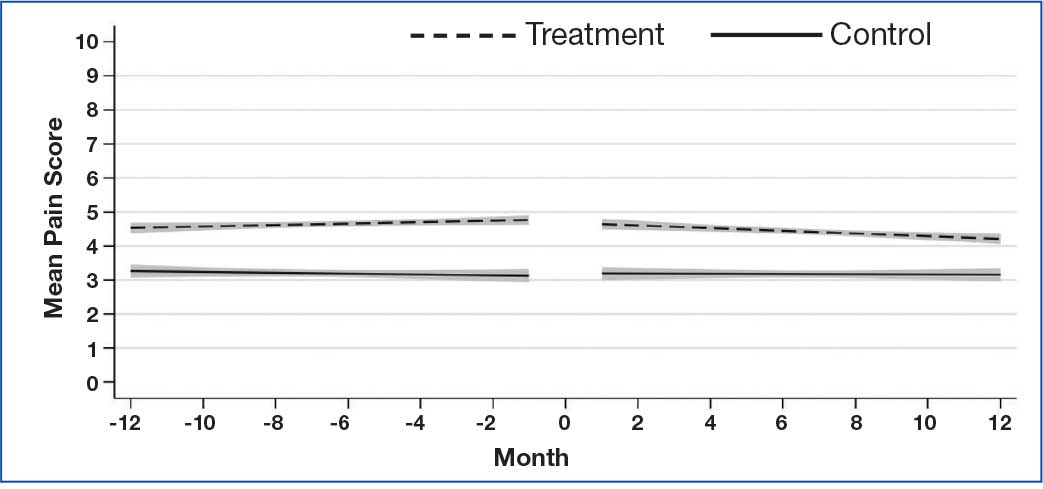
Pain intensity decreased for both groups by an estimated 0.02 points per month (99% CI, -0.04 to -0.01; P < .01), or 0.24 points over the 12-month postindex follow-up period (Figure 3). There were no statistically significant differences between treatment and control patients in pain intensity changes over the follow-up period (B = -0.02; 99% CI, -0.06 to 0.02; P = .15).
Discussion
Patients with musculoskeletal pain who received a thermal therapy device showed a larger decline in use of some specialty pain treatments, including physical therapy and specialty pain clinic services, when compared with matched control patients. One possible explanation is that patients who used the device may have had their pain adequately managed and thus required less specialty pain care. However, the absence of clinically significant changes in pain intensity over time suggests that pain intensity alone did not account for the observed changes in pain treatment use. We were unable to measure constructs of pain interference or functioning, which may be better predictors of functional restoration, as these data are not routinely collected within the VA and documented in the EHR. Future studies could clarify these findings by including measures of pain interference and functioning.
Although the overall declines in physical therapy and specialty pain clinic use associated with thermal therapy were modest (< 1 visit per patient), the impact of these reductions can be profound. In resource-limited health care settings, even small reductions in high-cost care utilization could be of great value in that health systems could offset costs associated with treating chronic pain without compromising quality of care or key clinical outcomes, such as pain intensity. This study, however, did not include a cost analysis. Future studies should incorporate formal cost analyses to quantify cost offsets that may result from decreased specialty pain treatment use.
Patients who received thermal therapy devices did not show clinically meaningful reductions in pain intensity over time, defined as reductions in pain intensity of 1.7 to 2.0 on a 0 to 10 scale.28,29 This finding is consistent with prior research that demonstrates relatively stable pain intensity self-ratings longitudinally by patients with chronic pain diagnoses, when assessed in the context of usual clinical care.30 This finding, however, is inconsistent with prior literature that demonstrates pain-reducing benefits of thermal therapy for low back pain and knee osteoarthritis.7-9
In this study, pain intensity ratings were derived from the EHR during routine outpatient clinical encounters and not at the time thermal therapy was self-administered, as has been done in prior clinical trials.7-9 Pain location was not specified at the time of pain ratings, and it is possible that patients may have been endorsing pain in areas of the body that had not been treated by thermal therapy. Patient-level variability in pain intensity ratings (eg, within-patient range over time) was not examined, although prior research indicates substantial variability.30 While average pain intensity ratings in the current study did not change, an examination of patient variability warrants further study, as a narrowing of pain intensity ratings can be perceived, by patients, as demonstrable improvement and has been associated with improved physical and psychological outcomes.31 Furthermore, pain intensity does not characterize physical or emotional functioning that can be captured with more comprehensive validated measures, some of which are recommended outcomes in pain clinical trials.32
Our findings point to reductions in all forms of opioid use across both treatment and control patients. Data from the VA and in the US more generally point to downward trends in opioid prescribing during the study period.33 This decline is likely due to increased use of risk mitigation approaches, such as routine urine drug screens and review of prescription drug monitoring databases.34 These state-level databases track prescribing of controlled substances, including opioids and benzodiazepines, within a state. Implementation of these practices has been associated with declines in higher risk opioid prescribing.34 Findings from this study further point to associations of reduced higher risk opioid use among patients who received thermal therapy devices. In the full sample of patients, reductions in days of opioid use, high-dose opioid use, and co-use of opioids and benzodiazepines were observed across all patients, with greater reductions observed in high-dose opioid use and concurrent opioid and benzodiazepine use among patients who received a thermal therapy device. Experimental studies indicate that the endogenous opioid system is implicated in pain relief and activated by thermal therapies. 35 Differential reductions in higher-risk opioid use among patients who received thermal therapy devices in our study may be associated with endogenous opioid activation, though this was not specifically measured. It is also unclear whether thermal therapy was provided by clinicians in the context of opioid tapering or other risk mitigation efforts, or patients reduced higher risk opioid use of their own volition. Prior research has identified both patient- and clinician-initiated opioid tapering and discontinuation.36 While a thorough explication of opioid dose reduction was beyond the scope of this study, future qualitative work could help explain potential benefits of thermal therapy in the context of analgesic pharmacotherapy use, including opioid medications.
Limitations
The extent to which patients used the thermal therapy device could not be measured; therefore, device receipt was used as a proxy for use. However, it was not possible to determine whether the frequency and duration of device use was associated with study outcomes. Treatment and control groups demonstrated some differences in outcome variables at the index date. Potential known biases were addressed using propensity score matching procedures and statistical procedures that controlled for patient demographic and clinical characteristics, as well as pretreatment values of all outcome variables. Nevertheless, as an observational study, this analysis cannot account for all known and unknown confounders, and a randomized controlled trial is needed to make claims of causality. The study population consisted of US veterans and included a low proportion of women. As a result, the findings may not be generalizable to other patient populations. Finally, prescription dispensing data, used as a proxy for pharmacotherapy use, do not necessarily reflect actual medication use.
Conclusions
This study is among the first to examine associations between thermal therapy and specialty pain treatment and analgesic pharmacotherapy use among US veterans. Although the retrospective cohort study design does not allow causal inferences regarding the efficacy of thermal therapy for veterans with chronic musculoskeletal pain, confidence in the findings is strengthened by methodological and statistical control of known confounders. Future trials employing experimental designs are needed to further clarify the clinical and health systems benefits of thermal therapy for musculoskeletal pain syndromes.
- Krebs EE, Gravely A, Nugent S, et al. Effect of opioid vs nonopioid medications on pain-related function in patients with chronic back pain or hip or knee osteoarthritis pain: the SPACE randomized clinical trial. JAMA. 2018;2018:872-882. doi:10.1001/jama.2018.0899
- Becker WC, DeBar LL, Heapy AA, et al. A research agenda for advancing non-pharmacological management of chronic musculoskeletal pain: findings from a VHA state-of-the-art conference. J Gen Intern Med. 2018;33:1-15. doi:10.1007/s11606-018-4345-6
- Fernandes IA, Armond ACV, Falci SGM. The effectiveness of the cold therapy (cryotherapy) in the management of inflammatory parameters after removal of mandibular third molars: a meta-analysis. Int Arch Otorhinolaryngol. 2019;23:221-228. doi:10.1055/s-0039-1677755
- Quinlan P, Davis J, Fields K, et al. Effects of localized cold therapy on pain in postoperative spinal fusion patients: a randomized controlled trial. Orthop Nurs. 2017;36:344-349. doi:10.1097/NOR.0000000000000382
- Malanga GA, Yan N, Stark J. Mechanisms and efficacy of heat and cold therapies for musculoskeletal injury. Postgrad Med. 2015;127:57-65. doi:10.1080/00325481.2015.992719
- Wang Y, Li S, Zhang Y, et al. Heat and cold therapy reduce pain in patients with delayed onset muscle soreness: a systematic review and meta-analysis of 32 randomized controlled trials. Phys Ther Sport. 2021;48:177-187. doi:10.1016/j.ptsp.2021.01.004
- Ariana M, Afrasiabifar A, Doulatabad SN, et al. The effect of local heat therapy versus cold rub gel on pain and joint functions in patients with knee osteoarthritis. Clin Nurs Res. 2022;31:1014-1022. doi:10.1177/10547738211035502
- French SD, Cameron M, Walker BF, et al. Superficial heat or cold for low back pain. Cochrane Database Syst Rev. 2006;1:CD004750. doi:10.1002/14651858.CD004750.pub2
- Tao XG, Bernacki EJ. A randomized clinical trial of continuous low-level heat therapy for acute muscular low back pain in the workplace. J Occup Environ Med. 2005;47:1298- 1306. doi:10.1097/01.jom.0000184877.01691.a3
- Freiwald J, Hoppe MW, Beermann W, et al. Effects of supplemental heat therapy in multimodal treated chronic low back pain patients on strength and flexibility. Clin Biomech. 2018;57:107-113. doi:10.1016/j.clinbiomech.2018.06.008
- Freiwald J, Magni A, Fanlo-Mazas P, et al. A role for superficial heat therapy in the management of nonspecific, mild-to-moderate low back pain in current clinical practice: a narrative review. Life. 2021;11:780. doi:10.3390/life11080780
- Kerns RD, Philip EJ, Lee AW, et al. Implementation of the Veterans Health Administration National Pain Management Strategy. Transl Behav Med. 2011;1:635-643. doi:10.1007/s13142-011-0094-3
- Brookhart MA, Schneeweiss S, Rothman KJ, et al. Variable selection for propensity score models. Am J Epidemiol. 2006;163:1149-1156. doi:10.1093/aje/kwj149
- Edlund MJ, Martin BC, Devries A, et al. Trends in use of opioids for chronic non-cancer pain among individuals with mental health and substance use disorders: the TROUP study. Clin J Pain. 2010;26:1-8. doi:10.1097/AJP.0b013e3181b99f35
- Kaur S, Stechuchak KM, Coffman CJ, et al. Gender differences in health care utilization among veterans with chronic pain. J Gen Intern Med. 2007;22:228-233. doi:10.1007/s11606-006-0048-5
- Meghani SH, Cho E. Self-reported pain and utilization of pain treatment between minorities and non-minorities in the United States. Public Health Nurs. 2009;26:307-316. doi:10.1111/j.1525-1446.2009.00785.x
- Morasco BJ, Duckart JP, Carr TP, et al. Clinical characteristics of veterans prescribed high doses of opioid medications for chronic non-cancer pain. Pain. 2010;151:625-632. doi:10.1016/j.pain.2010.08.002
- Nielsen S, Lintzeris N, Bruno R, et al. Benzodiazepine use among chronic pain patients prescribed opioids: associations with pain, physical and mental health, and health service utilization. Pain Med. 2015;16:356-366. doi:10.1111/pme.12594
- Quinn PD, Hur K, Chang Z, et al. Incident and long-term opioid therapy among patients with psychiatric conditions and medications: a national study of commercial health care claims. Pain. 2017;158:140-148. doi:10.1097/j.pain.0000000000000730
- Rubin DB. Matching to remove bias in observational studies. Biometrics. 1973;29:159-183.
- Rosenbaum PR, Rubin DB. Constructing a control group using multivariate matched sampling methods that incorporate the propensity score. Am Stat. 1985;39:33-38. doi:10.1007/s11596-025-00052-0
- Steiner PM, Cook TD, Shadish WR, et al. The importance of covariate selection in controlling for selection bias in observational studies. Psychol Methods. 2010;15:250-267. doi:10.1037/a0018719
- Charlson ME, Pompei P, Ales KL, et al. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40:373- 383. doi:10.1016/0021-9681(87)90171-8
- Long JS, Freese J. Regression Models for Categorical Dependent Variables Using Stata. 3rd ed. Stata Press; 2014.
- Muthén LK, Muthén BO. Mplus User’s Guide. 8th ed. Muthén & Muthén; 1998-2017.
- Hedeker D, Gibbons RD. Longitudinal Data Analysis. Wiley; 2006.
- Dobscha SK, Morasco BJ, Kovas AE, et al. Short-term variability in outpatient pain intensity scores in a national sample of older veterans with chronic pain. Pain Med. 2015;16:855-865. doi:10.1111/pme.12643
- Bahreini M, Safaie A, Mirfazaelian H, et al. How much change in pain score does really matter to patients? Am J Emerg Med. 2020;38:1641-1646. doi:10.1016/j.ajem.2019.158489
- Suzuki H, Aono S, Inoue S, et al. Clinically significant changes in pain along the pain intensity numerical rating scale in patients with chronic low back pain. PLoS One. 2020;15:e0229228. doi:10.1371/journal.pone.0229228
- McPherson SM, Smith CL, Dobscha SK, et al. Changes in pain intensity after discontinuation of long-term opioid therapy for chronic noncancer pain. Pain. 2018;159:2097- 2104. doi:10.1097/j.pain.0000000000001315
- Andrews NE, Strong J, Meredith PJ. Activity pacing, avoidance, endurance, and associations with patient functioning in chronic pain: a systematic and meta-analysis. Arch Phys Med Rehabil. 2012;93:2109-121. doi:10.1016/j.apmr.2012.05.029
- Dworkin RH, Turk DC, Farrar JT, et al. Core outcome measures for chronic pain clinical trials: IMMPACT recommendations. Pain. 2005;113:9-19. doi:10.1016/j.pain.2004.09.012
- Gellad WF, Good CB, Shulkin DJ. Addressing the opioid epidemic in the United States: Lessons from the Department of Veterans Affairs. JAMA Intern Med. 2017;177:611- 612. doi:10.1001/jamainternmed.2017.0147
- Lin LA, Bohnert ASB, Kerns RD, et al. Impact of the Opioid Safety Initiative on opioid-related prescribing in veterans. Pain. 2017;158:833-839. doi:10.1097/j.pain.0000000000000837
- Sirucek L, Price RC, Gandhi W, et al. Endogenous opioids contribute to the feeling of pain relief in humans. Pain. 2021;162:2821-2831. doi:10.1097/j.pain.0000000000002285
- Lovejoy TI, Morasco BJ, Demidenko MI, et al. Reasons for discontinuation of long-term opioid therapy in patients with and without substance use disorders. Pain. 2017;158:526- 534. doi:10.1097/j.pain.0000000000000796
Growing recognition of the limitations of long-term opioid therapy for chronic noncancer pain has highlighted the importance of nonpharmacologic approaches to pain treatment.1,2 These treatments are varied and may include psychological and behavioral therapies (eg, cognitive behavioral therapy for chronic pain), exercise and movement therapies (eg, yoga), and manual therapies (eg, chiropractic). Body surface cold therapy, while predominantly used to reduce postoperative pain and inflammation,3,4 pain subsequent to acute musculoskeletal injury,5 and pain related to muscle soreness,6 is a nonpharmacologic treatment that has shown pain-reducing benefits for chronic low back pain and knee osteoarthritis, as has its counterpart, superficial heat therapy.7-9 Heat therapy has also been shown to improve strength, flexibility, and activities of daily living in patients with chronic low back pain.10,11 Cold and heat therapies are commonly used complementarily. Cold therapies aim to reduce blood flow and inflammation and are often used immediately following trauma to an affected area, whereas heat therapies increase blood flow and metabolic activity and are commonly used to promote healing.5
Heat and cold therapies (also known as thermal therapies) benefit resource-limited health care systems, as most devices require a single expenditure and can be self-administered by patients at home as part of their pain self-management plan. In addition, these pain self-management tools may attenuate the need for more expensive specialty pain care and ongoing analgesic pharmacotherapy. Despite their potential, few studies have characterized the benefits of thermal therapies for patients with heterogeneous chronic pain syndromes.
The purpose of this retrospective cohort study was to examine the potential clinical and health system benefits of patient-administered thermal therapy. Our primary hypothesis was that patients with chronic musculoskeletal pain who received a thermal therapy self-management device would have fewer days of opioid prescriptions compared with a sample of matched control patients. Secondarily, we hypothesized that patients who received a thermal therapy device would have lower utilization of specialty pain care, fewer potentially hazardous opioid prescriptions (eg, high-dose opioid therapy and concurrent opioid and benzodiazepine prescriptions), fewer prescriptions for nonopioid analgesic medications, and decreased pain intensity when compared with matched controls.
Methods
This retrospective cohort study compared pain pharmacotherapy, pain treatment utilization, and pain intensity outcomes between patients who received a thermal therapy device and matched patients who did not. The study was approved by the US Department of Veterans Affairs (VA) Portland Health Care System Institutional Review Board and was granted a waiver of informed consent to access patient electronic health records (EHRs).
Pain Care
The VA uses ThermaZone thermal therapy devices (Innovative Medical Equipment) for chronic pain treatment. The device uses thermoelectric technology to provide point-of-contact cooling and heating therapy through site-specific pads (eg, ankle, knee, hip, back, elbow, shoulder). Patients place pads on sites where they experience pain, and temperature regulated water circulates through the device and to the pad, providing consistent, localized thermal therapy. The pads range in temperature from 1 °C to 52 °C, and temperatures are self-monitored and controlled by the patient.
Standard pain care in this study followed the VA stepped model of pain care, which builds on a foundation of patient education for pain self-management approaches (eg, exercise, mindfulness, relaxation, social support).12 According to the VA stepped model of pain care, all patients with chronic pain should engage in these foundational self-management approaches. However, some patients require more intensive care. The VA stepped-up treatment engages primary and specialty care services—such as physical therapy, pharmacy, complementary and integrative health approaches, mental health, and substance use services—and, when indicated, may escalate treatment to interdisciplinary pain teams or tertiary pain centers. In this retrospective cohort study, treatment patients received a thermal therapy device and standard of care, whereas control patients received standard care only.
Sample Selection
Eligible patients were aged ≥ 18 years, had a musculoskeletal pain diagnosis documented in the EHR in the year prior to thermal therapy device receipt (or during the same period for a treatment patient’s matched control), and were enrolled in VA health care during the study period. Patients who died during the study period were excluded. Treatment patients received a thermal therapy device from the VA between January 1, 2017, and December 31, 2018, when complete data on thermal therapy devices were available. For control patients, the VA Corporate Data Warehouse (CDW) was used to identify VA patients with characteristics similar to those of treated patients.
We modeled the probability that a patient would receive a thermal therapy device using logistic regression. Predictor variables were measured in the year prior to device receipt and included variables associated with pain treatment utilization and analgesic pharmacotherapy receipt, as recommended by Brookhart et al.13 These included age, sex, race, ethnicity, VA service-connected disability status, comorbidities, receipt of medications for opioid use disorder, pain diagnoses, mental health diagnoses, and substance use disorder diagnoses.14-19
The resulting propensity scores (eg, predicted probabilities) were used to match treatment patients 1:1 with control patients using a nearest-neighbor matching algorithm.20 This procedure matches a control patient with the closest propensity score to that of the corresponding treatment patient. An exact match on VA facility was required (eg, treatment patients and matched controls received care at the same VA facility). Standardized differences were used to assess covariate balance between the matched groups, and kernel density plots of propensity scores tested for sufficient overlap.21 Control patients were selected from a pool of 1,150,149 patients.
Study Variables
The index date was the date the thermal therapy device was released to treatment patients or the same date for the matched controls. Data were extracted from the CDW over a 24-month period: 12 months prior to the index date through 12 months afterwards. Collecting data in the 12 months prior to treatment initiation allowed us to adjust for covariates and provided greater precision, as recommended for observational study designs.22
Treatment conditions were defined dichotomously as receipt vs nonreceipt of a thermal therapy device. The primary outcome was the number of days of opioid use in the 12 months following the index date. Additional outcomes included days of high-dose opioid therapy (≥ 50 mg morphine equivalent [MME] daily), concurrent opioid and benzodiazepine prescriptions, and nonopioid analgesic pharmacotherapy (eg, nonsteroidal anti-inflammatory drugs, acetaminophen, muscle relaxants). All prescription data were based on medication fills obtained from VA pharmacy records. Additional outcomes included the number of visits to physical therapy, occupational therapy, physical medicine and rehabilitation, and interdisciplinary pain clinics, including interventional pain medicine.
Pain intensity ratings were collected as part of routine VA care using a numeric scale from 0 (no pain) to 10 (worst possible pain). Pain intensity trajectories were computed using all available EHR-derived pain intensity score data for each patient in the 12 months prior to and following the index date.
Covariates were extracted from the EHR and evaluated in the year prior to the index date, unless otherwise noted. They included age at the index date; self-reported sex, and race and ethnicity; service-connected disability status (disability awarded as a result of military service-related trauma or injury); Charlson Comorbidity Index; and diagnoses of opioid use disorder, alcohol use disorder, other substance use disorder, mood disorder, posttraumatic stress disorder, other anxiety disorder, psychotic disorder, neuropathic pain, and headache pain.23 All diagnoses were coded as yes if identified in the EHR as a focus of treatment during any clinical encounter in the year prior to the index date or no if not identified as a focus of treatment.
The number of days patients had been living with pain was calculated and defined as the number of days from the first pain diagnosis available in a patient’s EHR to the index date. Finally, the number of pain-related surgeries (eg, surgeries with ≥ 1 pain diagnoses associated with the clinical encounter) and average pain intensity were computed for the pre- and postindex date evaluation periods and included as model covariates.
Statistical Analyses
For the 4 pharmacotherapy and 4 nonpharmacologic treatment usage outcomes, we tested the fit of linear models and several models with count distributions using the Bayesian information criterion.24 Count distributions included Poisson, zero-inflated Poisson, negative binomial, and zero-inflated negative binomial. With 1 exception (days of high-dose opioid use), a negative binomial distribution best fit the data. For days of high-dose opioid use, a Gaussian distribution best fit the data.
Eight separate mixed-effects regression analyses compared changes in each outcome from the 12-month preindex period through the 12-month postindex period between treatment and control patients by testing the Time × Treatment interaction. This approach statistically accounts for observed pretreatment differences in outcome variables. Statistics for the main effects of time and treatment are also presented. To reduce bias, models controlled for covariates specified previously.
For pain intensity, we used random-effects growth modeling to quantify both fixed and random effects of pain intensity at the index date (eg, the model intercept), which estimates pain at the time of treatment initiation, and change in pain during the 12 months following the index date (eg, the model slope), which characterizes the trajectory of pain intensity ratings.25 The model included piecewise components of pain score trajectories in the 12 months prior to the index date and in the 12 months following the index date. Several types of change for the 12-month postindex observation period were explored—including quadratic and cubic curvilinear change. A linear model for change in pain over time provided the best fit based on the Bayesian information criterion and parsimony of model parameters.26 We report estimates of change in pain over time in monthly intervals for ease of interpretation. However, models used all individual pain scores rather than computing monthly averages when > 1 pain score was available within a month, as recommended in previous research.27 This approach makes optimal use of all available data. Both random effects (intercept and slope) were regressed onto the set of covariates described previously.
This study used data available in the EHR over the 24-month observation period. We characterize the density (eg, frequency) of all outcome variables by treatment condition in the Appendix. Because the hypotheses were directional, the authors used a 2-sided α = 0.10 and applied a Bonferroni correction for the 9 statistical tests performed, resulting in an adjusted α of 0.01. Treatment utilization and pharmacotherapy outcome analyses were performed in Stata, version 16.1. Random-effects growth modeling of pain score trajectories was performed using Mplus, version 8.8.

Results
There were 2182 patients in the treatment group and 2182 matched controls. The mean (SD) age was 54 (15) years; 81% were male, and about two-thirds (68%) identified as White and non-Hispanic. Mental health comorbidities were common, with > 40% of the sample having diagnoses of a mood disorder and/or posttraumatic stress disorder. Nearly all patients (90%) had VA service-connected disability ratings. Among patients with chronic musculoskeletal pain, 8% had comorbid neuropathic pain and 14% had headache. The mean (SD) duration of chronic pain across both patient groups was 3416 (2016) days, or about 9.4 years (Table 1).

Pharmacotherapy
High-dose opioid use (> 50 MME daily dose), days of opioid use, and concurrent opioid and benzodiazepine use decreased for all patients from the pre- to posttreatment period (Table 2). However, high-dose opioid use (Time × Treatment interaction, 3.24; 99% CI, 0.34 to 6.14) (Figure 1A) and concurrent opioid and benzodiazepine use (Time × Treatment interaction, 0.76; 99% CI, 0.67 to 0.86) (Figure 1B) had a larger decrease for the treatment vs matched control group. Treatment and matched control patients had comparable reductions in days of opioid use (Time × Treatment interaction, 0.98; 99% CI, 0.91 to 1.05) (Figure 1C). Neither group showed changes in nonopioid analgesic pharmacotherapy over time (main effect of time, incidence rate ratio [IRR], 1.03; 99% CI, 0.99 to 1.07; Time × Treatment interaction, 0.95; 99% CI, 0.90 to 1.01) (Figure 1D).


Nonpharmacologic Pain Treatment
The number of physical therapy and pain clinic visits declined for treatment patients and increased slightly for matched control patients (Figure 2A and 2B). For occupational therapy visits, neither group showed changes over time (main effect of time IRR, 1.03; 99% CI, 0.83 to 1.22; Time × Treatment interaction, 1.20; 99% CI, 0.93 to 1.46) (Figure 2C). For physical medicine and rehabilitation visits, both groups decreased use over time (main effect of time IRR, 0.78; 99% CI, 0.66 to 0.90), but this decrease did not differ between treatment and control patients (Time × Treatment interaction, 1.16; 99% CI, 0.94 to 1.37) (Figure 2D).

Pain Intensity
Pain intensity decreased for both groups by an estimated 0.02 points per month (99% CI, -0.04 to -0.01; P < .01), or 0.24 points over the 12-month postindex follow-up period (Figure 3). There were no statistically significant differences between treatment and control patients in pain intensity changes over the follow-up period (B = -0.02; 99% CI, -0.06 to 0.02; P = .15).

Discussion
Patients with musculoskeletal pain who received a thermal therapy device showed a larger decline in use of some specialty pain treatments, including physical therapy and specialty pain clinic services, when compared with matched control patients. One possible explanation is that patients who used the device may have had their pain adequately managed and thus required less specialty pain care. However, the absence of clinically significant changes in pain intensity over time suggests that pain intensity alone did not account for the observed changes in pain treatment use. We were unable to measure constructs of pain interference or functioning, which may be better predictors of functional restoration, as these data are not routinely collected within the VA and documented in the EHR. Future studies could clarify these findings by including measures of pain interference and functioning.
Although the overall declines in physical therapy and specialty pain clinic use associated with thermal therapy were modest (< 1 visit per patient), the impact of these reductions can be profound. In resource-limited health care settings, even small reductions in high-cost care utilization could be of great value in that health systems could offset costs associated with treating chronic pain without compromising quality of care or key clinical outcomes, such as pain intensity. This study, however, did not include a cost analysis. Future studies should incorporate formal cost analyses to quantify cost offsets that may result from decreased specialty pain treatment use.
Patients who received thermal therapy devices did not show clinically meaningful reductions in pain intensity over time, defined as reductions in pain intensity of 1.7 to 2.0 on a 0 to 10 scale.28,29 This finding is consistent with prior research that demonstrates relatively stable pain intensity self-ratings longitudinally by patients with chronic pain diagnoses, when assessed in the context of usual clinical care.30 This finding, however, is inconsistent with prior literature that demonstrates pain-reducing benefits of thermal therapy for low back pain and knee osteoarthritis.7-9
In this study, pain intensity ratings were derived from the EHR during routine outpatient clinical encounters and not at the time thermal therapy was self-administered, as has been done in prior clinical trials.7-9 Pain location was not specified at the time of pain ratings, and it is possible that patients may have been endorsing pain in areas of the body that had not been treated by thermal therapy. Patient-level variability in pain intensity ratings (eg, within-patient range over time) was not examined, although prior research indicates substantial variability.30 While average pain intensity ratings in the current study did not change, an examination of patient variability warrants further study, as a narrowing of pain intensity ratings can be perceived, by patients, as demonstrable improvement and has been associated with improved physical and psychological outcomes.31 Furthermore, pain intensity does not characterize physical or emotional functioning that can be captured with more comprehensive validated measures, some of which are recommended outcomes in pain clinical trials.32
Our findings point to reductions in all forms of opioid use across both treatment and control patients. Data from the VA and in the US more generally point to downward trends in opioid prescribing during the study period.33 This decline is likely due to increased use of risk mitigation approaches, such as routine urine drug screens and review of prescription drug monitoring databases.34 These state-level databases track prescribing of controlled substances, including opioids and benzodiazepines, within a state. Implementation of these practices has been associated with declines in higher risk opioid prescribing.34 Findings from this study further point to associations of reduced higher risk opioid use among patients who received thermal therapy devices. In the full sample of patients, reductions in days of opioid use, high-dose opioid use, and co-use of opioids and benzodiazepines were observed across all patients, with greater reductions observed in high-dose opioid use and concurrent opioid and benzodiazepine use among patients who received a thermal therapy device. Experimental studies indicate that the endogenous opioid system is implicated in pain relief and activated by thermal therapies. 35 Differential reductions in higher-risk opioid use among patients who received thermal therapy devices in our study may be associated with endogenous opioid activation, though this was not specifically measured. It is also unclear whether thermal therapy was provided by clinicians in the context of opioid tapering or other risk mitigation efforts, or patients reduced higher risk opioid use of their own volition. Prior research has identified both patient- and clinician-initiated opioid tapering and discontinuation.36 While a thorough explication of opioid dose reduction was beyond the scope of this study, future qualitative work could help explain potential benefits of thermal therapy in the context of analgesic pharmacotherapy use, including opioid medications.
Limitations
The extent to which patients used the thermal therapy device could not be measured; therefore, device receipt was used as a proxy for use. However, it was not possible to determine whether the frequency and duration of device use was associated with study outcomes. Treatment and control groups demonstrated some differences in outcome variables at the index date. Potential known biases were addressed using propensity score matching procedures and statistical procedures that controlled for patient demographic and clinical characteristics, as well as pretreatment values of all outcome variables. Nevertheless, as an observational study, this analysis cannot account for all known and unknown confounders, and a randomized controlled trial is needed to make claims of causality. The study population consisted of US veterans and included a low proportion of women. As a result, the findings may not be generalizable to other patient populations. Finally, prescription dispensing data, used as a proxy for pharmacotherapy use, do not necessarily reflect actual medication use.
Conclusions
This study is among the first to examine associations between thermal therapy and specialty pain treatment and analgesic pharmacotherapy use among US veterans. Although the retrospective cohort study design does not allow causal inferences regarding the efficacy of thermal therapy for veterans with chronic musculoskeletal pain, confidence in the findings is strengthened by methodological and statistical control of known confounders. Future trials employing experimental designs are needed to further clarify the clinical and health systems benefits of thermal therapy for musculoskeletal pain syndromes.
Growing recognition of the limitations of long-term opioid therapy for chronic noncancer pain has highlighted the importance of nonpharmacologic approaches to pain treatment.1,2 These treatments are varied and may include psychological and behavioral therapies (eg, cognitive behavioral therapy for chronic pain), exercise and movement therapies (eg, yoga), and manual therapies (eg, chiropractic). Body surface cold therapy, while predominantly used to reduce postoperative pain and inflammation,3,4 pain subsequent to acute musculoskeletal injury,5 and pain related to muscle soreness,6 is a nonpharmacologic treatment that has shown pain-reducing benefits for chronic low back pain and knee osteoarthritis, as has its counterpart, superficial heat therapy.7-9 Heat therapy has also been shown to improve strength, flexibility, and activities of daily living in patients with chronic low back pain.10,11 Cold and heat therapies are commonly used complementarily. Cold therapies aim to reduce blood flow and inflammation and are often used immediately following trauma to an affected area, whereas heat therapies increase blood flow and metabolic activity and are commonly used to promote healing.5
Heat and cold therapies (also known as thermal therapies) benefit resource-limited health care systems, as most devices require a single expenditure and can be self-administered by patients at home as part of their pain self-management plan. In addition, these pain self-management tools may attenuate the need for more expensive specialty pain care and ongoing analgesic pharmacotherapy. Despite their potential, few studies have characterized the benefits of thermal therapies for patients with heterogeneous chronic pain syndromes.
The purpose of this retrospective cohort study was to examine the potential clinical and health system benefits of patient-administered thermal therapy. Our primary hypothesis was that patients with chronic musculoskeletal pain who received a thermal therapy self-management device would have fewer days of opioid prescriptions compared with a sample of matched control patients. Secondarily, we hypothesized that patients who received a thermal therapy device would have lower utilization of specialty pain care, fewer potentially hazardous opioid prescriptions (eg, high-dose opioid therapy and concurrent opioid and benzodiazepine prescriptions), fewer prescriptions for nonopioid analgesic medications, and decreased pain intensity when compared with matched controls.
Methods
This retrospective cohort study compared pain pharmacotherapy, pain treatment utilization, and pain intensity outcomes between patients who received a thermal therapy device and matched patients who did not. The study was approved by the US Department of Veterans Affairs (VA) Portland Health Care System Institutional Review Board and was granted a waiver of informed consent to access patient electronic health records (EHRs).
Pain Care
The VA uses ThermaZone thermal therapy devices (Innovative Medical Equipment) for chronic pain treatment. The device uses thermoelectric technology to provide point-of-contact cooling and heating therapy through site-specific pads (eg, ankle, knee, hip, back, elbow, shoulder). Patients place pads on sites where they experience pain, and temperature regulated water circulates through the device and to the pad, providing consistent, localized thermal therapy. The pads range in temperature from 1 °C to 52 °C, and temperatures are self-monitored and controlled by the patient.
Standard pain care in this study followed the VA stepped model of pain care, which builds on a foundation of patient education for pain self-management approaches (eg, exercise, mindfulness, relaxation, social support).12 According to the VA stepped model of pain care, all patients with chronic pain should engage in these foundational self-management approaches. However, some patients require more intensive care. The VA stepped-up treatment engages primary and specialty care services—such as physical therapy, pharmacy, complementary and integrative health approaches, mental health, and substance use services—and, when indicated, may escalate treatment to interdisciplinary pain teams or tertiary pain centers. In this retrospective cohort study, treatment patients received a thermal therapy device and standard of care, whereas control patients received standard care only.
Sample Selection
Eligible patients were aged ≥ 18 years, had a musculoskeletal pain diagnosis documented in the EHR in the year prior to thermal therapy device receipt (or during the same period for a treatment patient’s matched control), and were enrolled in VA health care during the study period. Patients who died during the study period were excluded. Treatment patients received a thermal therapy device from the VA between January 1, 2017, and December 31, 2018, when complete data on thermal therapy devices were available. For control patients, the VA Corporate Data Warehouse (CDW) was used to identify VA patients with characteristics similar to those of treated patients.
We modeled the probability that a patient would receive a thermal therapy device using logistic regression. Predictor variables were measured in the year prior to device receipt and included variables associated with pain treatment utilization and analgesic pharmacotherapy receipt, as recommended by Brookhart et al.13 These included age, sex, race, ethnicity, VA service-connected disability status, comorbidities, receipt of medications for opioid use disorder, pain diagnoses, mental health diagnoses, and substance use disorder diagnoses.14-19
The resulting propensity scores (eg, predicted probabilities) were used to match treatment patients 1:1 with control patients using a nearest-neighbor matching algorithm.20 This procedure matches a control patient with the closest propensity score to that of the corresponding treatment patient. An exact match on VA facility was required (eg, treatment patients and matched controls received care at the same VA facility). Standardized differences were used to assess covariate balance between the matched groups, and kernel density plots of propensity scores tested for sufficient overlap.21 Control patients were selected from a pool of 1,150,149 patients.
Study Variables
The index date was the date the thermal therapy device was released to treatment patients or the same date for the matched controls. Data were extracted from the CDW over a 24-month period: 12 months prior to the index date through 12 months afterwards. Collecting data in the 12 months prior to treatment initiation allowed us to adjust for covariates and provided greater precision, as recommended for observational study designs.22
Treatment conditions were defined dichotomously as receipt vs nonreceipt of a thermal therapy device. The primary outcome was the number of days of opioid use in the 12 months following the index date. Additional outcomes included days of high-dose opioid therapy (≥ 50 mg morphine equivalent [MME] daily), concurrent opioid and benzodiazepine prescriptions, and nonopioid analgesic pharmacotherapy (eg, nonsteroidal anti-inflammatory drugs, acetaminophen, muscle relaxants). All prescription data were based on medication fills obtained from VA pharmacy records. Additional outcomes included the number of visits to physical therapy, occupational therapy, physical medicine and rehabilitation, and interdisciplinary pain clinics, including interventional pain medicine.
Pain intensity ratings were collected as part of routine VA care using a numeric scale from 0 (no pain) to 10 (worst possible pain). Pain intensity trajectories were computed using all available EHR-derived pain intensity score data for each patient in the 12 months prior to and following the index date.
Covariates were extracted from the EHR and evaluated in the year prior to the index date, unless otherwise noted. They included age at the index date; self-reported sex, and race and ethnicity; service-connected disability status (disability awarded as a result of military service-related trauma or injury); Charlson Comorbidity Index; and diagnoses of opioid use disorder, alcohol use disorder, other substance use disorder, mood disorder, posttraumatic stress disorder, other anxiety disorder, psychotic disorder, neuropathic pain, and headache pain.23 All diagnoses were coded as yes if identified in the EHR as a focus of treatment during any clinical encounter in the year prior to the index date or no if not identified as a focus of treatment.
The number of days patients had been living with pain was calculated and defined as the number of days from the first pain diagnosis available in a patient’s EHR to the index date. Finally, the number of pain-related surgeries (eg, surgeries with ≥ 1 pain diagnoses associated with the clinical encounter) and average pain intensity were computed for the pre- and postindex date evaluation periods and included as model covariates.
Statistical Analyses
For the 4 pharmacotherapy and 4 nonpharmacologic treatment usage outcomes, we tested the fit of linear models and several models with count distributions using the Bayesian information criterion.24 Count distributions included Poisson, zero-inflated Poisson, negative binomial, and zero-inflated negative binomial. With 1 exception (days of high-dose opioid use), a negative binomial distribution best fit the data. For days of high-dose opioid use, a Gaussian distribution best fit the data.
Eight separate mixed-effects regression analyses compared changes in each outcome from the 12-month preindex period through the 12-month postindex period between treatment and control patients by testing the Time × Treatment interaction. This approach statistically accounts for observed pretreatment differences in outcome variables. Statistics for the main effects of time and treatment are also presented. To reduce bias, models controlled for covariates specified previously.
For pain intensity, we used random-effects growth modeling to quantify both fixed and random effects of pain intensity at the index date (eg, the model intercept), which estimates pain at the time of treatment initiation, and change in pain during the 12 months following the index date (eg, the model slope), which characterizes the trajectory of pain intensity ratings.25 The model included piecewise components of pain score trajectories in the 12 months prior to the index date and in the 12 months following the index date. Several types of change for the 12-month postindex observation period were explored—including quadratic and cubic curvilinear change. A linear model for change in pain over time provided the best fit based on the Bayesian information criterion and parsimony of model parameters.26 We report estimates of change in pain over time in monthly intervals for ease of interpretation. However, models used all individual pain scores rather than computing monthly averages when > 1 pain score was available within a month, as recommended in previous research.27 This approach makes optimal use of all available data. Both random effects (intercept and slope) were regressed onto the set of covariates described previously.
This study used data available in the EHR over the 24-month observation period. We characterize the density (eg, frequency) of all outcome variables by treatment condition in the Appendix. Because the hypotheses were directional, the authors used a 2-sided α = 0.10 and applied a Bonferroni correction for the 9 statistical tests performed, resulting in an adjusted α of 0.01. Treatment utilization and pharmacotherapy outcome analyses were performed in Stata, version 16.1. Random-effects growth modeling of pain score trajectories was performed using Mplus, version 8.8.

Results
There were 2182 patients in the treatment group and 2182 matched controls. The mean (SD) age was 54 (15) years; 81% were male, and about two-thirds (68%) identified as White and non-Hispanic. Mental health comorbidities were common, with > 40% of the sample having diagnoses of a mood disorder and/or posttraumatic stress disorder. Nearly all patients (90%) had VA service-connected disability ratings. Among patients with chronic musculoskeletal pain, 8% had comorbid neuropathic pain and 14% had headache. The mean (SD) duration of chronic pain across both patient groups was 3416 (2016) days, or about 9.4 years (Table 1).

Pharmacotherapy
High-dose opioid use (> 50 MME daily dose), days of opioid use, and concurrent opioid and benzodiazepine use decreased for all patients from the pre- to posttreatment period (Table 2). However, high-dose opioid use (Time × Treatment interaction, 3.24; 99% CI, 0.34 to 6.14) (Figure 1A) and concurrent opioid and benzodiazepine use (Time × Treatment interaction, 0.76; 99% CI, 0.67 to 0.86) (Figure 1B) had a larger decrease for the treatment vs matched control group. Treatment and matched control patients had comparable reductions in days of opioid use (Time × Treatment interaction, 0.98; 99% CI, 0.91 to 1.05) (Figure 1C). Neither group showed changes in nonopioid analgesic pharmacotherapy over time (main effect of time, incidence rate ratio [IRR], 1.03; 99% CI, 0.99 to 1.07; Time × Treatment interaction, 0.95; 99% CI, 0.90 to 1.01) (Figure 1D).


Nonpharmacologic Pain Treatment
The number of physical therapy and pain clinic visits declined for treatment patients and increased slightly for matched control patients (Figure 2A and 2B). For occupational therapy visits, neither group showed changes over time (main effect of time IRR, 1.03; 99% CI, 0.83 to 1.22; Time × Treatment interaction, 1.20; 99% CI, 0.93 to 1.46) (Figure 2C). For physical medicine and rehabilitation visits, both groups decreased use over time (main effect of time IRR, 0.78; 99% CI, 0.66 to 0.90), but this decrease did not differ between treatment and control patients (Time × Treatment interaction, 1.16; 99% CI, 0.94 to 1.37) (Figure 2D).

Pain Intensity
Pain intensity decreased for both groups by an estimated 0.02 points per month (99% CI, -0.04 to -0.01; P < .01), or 0.24 points over the 12-month postindex follow-up period (Figure 3). There were no statistically significant differences between treatment and control patients in pain intensity changes over the follow-up period (B = -0.02; 99% CI, -0.06 to 0.02; P = .15).

Discussion
Patients with musculoskeletal pain who received a thermal therapy device showed a larger decline in use of some specialty pain treatments, including physical therapy and specialty pain clinic services, when compared with matched control patients. One possible explanation is that patients who used the device may have had their pain adequately managed and thus required less specialty pain care. However, the absence of clinically significant changes in pain intensity over time suggests that pain intensity alone did not account for the observed changes in pain treatment use. We were unable to measure constructs of pain interference or functioning, which may be better predictors of functional restoration, as these data are not routinely collected within the VA and documented in the EHR. Future studies could clarify these findings by including measures of pain interference and functioning.
Although the overall declines in physical therapy and specialty pain clinic use associated with thermal therapy were modest (< 1 visit per patient), the impact of these reductions can be profound. In resource-limited health care settings, even small reductions in high-cost care utilization could be of great value in that health systems could offset costs associated with treating chronic pain without compromising quality of care or key clinical outcomes, such as pain intensity. This study, however, did not include a cost analysis. Future studies should incorporate formal cost analyses to quantify cost offsets that may result from decreased specialty pain treatment use.
Patients who received thermal therapy devices did not show clinically meaningful reductions in pain intensity over time, defined as reductions in pain intensity of 1.7 to 2.0 on a 0 to 10 scale.28,29 This finding is consistent with prior research that demonstrates relatively stable pain intensity self-ratings longitudinally by patients with chronic pain diagnoses, when assessed in the context of usual clinical care.30 This finding, however, is inconsistent with prior literature that demonstrates pain-reducing benefits of thermal therapy for low back pain and knee osteoarthritis.7-9
In this study, pain intensity ratings were derived from the EHR during routine outpatient clinical encounters and not at the time thermal therapy was self-administered, as has been done in prior clinical trials.7-9 Pain location was not specified at the time of pain ratings, and it is possible that patients may have been endorsing pain in areas of the body that had not been treated by thermal therapy. Patient-level variability in pain intensity ratings (eg, within-patient range over time) was not examined, although prior research indicates substantial variability.30 While average pain intensity ratings in the current study did not change, an examination of patient variability warrants further study, as a narrowing of pain intensity ratings can be perceived, by patients, as demonstrable improvement and has been associated with improved physical and psychological outcomes.31 Furthermore, pain intensity does not characterize physical or emotional functioning that can be captured with more comprehensive validated measures, some of which are recommended outcomes in pain clinical trials.32
Our findings point to reductions in all forms of opioid use across both treatment and control patients. Data from the VA and in the US more generally point to downward trends in opioid prescribing during the study period.33 This decline is likely due to increased use of risk mitigation approaches, such as routine urine drug screens and review of prescription drug monitoring databases.34 These state-level databases track prescribing of controlled substances, including opioids and benzodiazepines, within a state. Implementation of these practices has been associated with declines in higher risk opioid prescribing.34 Findings from this study further point to associations of reduced higher risk opioid use among patients who received thermal therapy devices. In the full sample of patients, reductions in days of opioid use, high-dose opioid use, and co-use of opioids and benzodiazepines were observed across all patients, with greater reductions observed in high-dose opioid use and concurrent opioid and benzodiazepine use among patients who received a thermal therapy device. Experimental studies indicate that the endogenous opioid system is implicated in pain relief and activated by thermal therapies. 35 Differential reductions in higher-risk opioid use among patients who received thermal therapy devices in our study may be associated with endogenous opioid activation, though this was not specifically measured. It is also unclear whether thermal therapy was provided by clinicians in the context of opioid tapering or other risk mitigation efforts, or patients reduced higher risk opioid use of their own volition. Prior research has identified both patient- and clinician-initiated opioid tapering and discontinuation.36 While a thorough explication of opioid dose reduction was beyond the scope of this study, future qualitative work could help explain potential benefits of thermal therapy in the context of analgesic pharmacotherapy use, including opioid medications.
Limitations
The extent to which patients used the thermal therapy device could not be measured; therefore, device receipt was used as a proxy for use. However, it was not possible to determine whether the frequency and duration of device use was associated with study outcomes. Treatment and control groups demonstrated some differences in outcome variables at the index date. Potential known biases were addressed using propensity score matching procedures and statistical procedures that controlled for patient demographic and clinical characteristics, as well as pretreatment values of all outcome variables. Nevertheless, as an observational study, this analysis cannot account for all known and unknown confounders, and a randomized controlled trial is needed to make claims of causality. The study population consisted of US veterans and included a low proportion of women. As a result, the findings may not be generalizable to other patient populations. Finally, prescription dispensing data, used as a proxy for pharmacotherapy use, do not necessarily reflect actual medication use.
Conclusions
This study is among the first to examine associations between thermal therapy and specialty pain treatment and analgesic pharmacotherapy use among US veterans. Although the retrospective cohort study design does not allow causal inferences regarding the efficacy of thermal therapy for veterans with chronic musculoskeletal pain, confidence in the findings is strengthened by methodological and statistical control of known confounders. Future trials employing experimental designs are needed to further clarify the clinical and health systems benefits of thermal therapy for musculoskeletal pain syndromes.
- Krebs EE, Gravely A, Nugent S, et al. Effect of opioid vs nonopioid medications on pain-related function in patients with chronic back pain or hip or knee osteoarthritis pain: the SPACE randomized clinical trial. JAMA. 2018;2018:872-882. doi:10.1001/jama.2018.0899
- Becker WC, DeBar LL, Heapy AA, et al. A research agenda for advancing non-pharmacological management of chronic musculoskeletal pain: findings from a VHA state-of-the-art conference. J Gen Intern Med. 2018;33:1-15. doi:10.1007/s11606-018-4345-6
- Fernandes IA, Armond ACV, Falci SGM. The effectiveness of the cold therapy (cryotherapy) in the management of inflammatory parameters after removal of mandibular third molars: a meta-analysis. Int Arch Otorhinolaryngol. 2019;23:221-228. doi:10.1055/s-0039-1677755
- Quinlan P, Davis J, Fields K, et al. Effects of localized cold therapy on pain in postoperative spinal fusion patients: a randomized controlled trial. Orthop Nurs. 2017;36:344-349. doi:10.1097/NOR.0000000000000382
- Malanga GA, Yan N, Stark J. Mechanisms and efficacy of heat and cold therapies for musculoskeletal injury. Postgrad Med. 2015;127:57-65. doi:10.1080/00325481.2015.992719
- Wang Y, Li S, Zhang Y, et al. Heat and cold therapy reduce pain in patients with delayed onset muscle soreness: a systematic review and meta-analysis of 32 randomized controlled trials. Phys Ther Sport. 2021;48:177-187. doi:10.1016/j.ptsp.2021.01.004
- Ariana M, Afrasiabifar A, Doulatabad SN, et al. The effect of local heat therapy versus cold rub gel on pain and joint functions in patients with knee osteoarthritis. Clin Nurs Res. 2022;31:1014-1022. doi:10.1177/10547738211035502
- French SD, Cameron M, Walker BF, et al. Superficial heat or cold for low back pain. Cochrane Database Syst Rev. 2006;1:CD004750. doi:10.1002/14651858.CD004750.pub2
- Tao XG, Bernacki EJ. A randomized clinical trial of continuous low-level heat therapy for acute muscular low back pain in the workplace. J Occup Environ Med. 2005;47:1298- 1306. doi:10.1097/01.jom.0000184877.01691.a3
- Freiwald J, Hoppe MW, Beermann W, et al. Effects of supplemental heat therapy in multimodal treated chronic low back pain patients on strength and flexibility. Clin Biomech. 2018;57:107-113. doi:10.1016/j.clinbiomech.2018.06.008
- Freiwald J, Magni A, Fanlo-Mazas P, et al. A role for superficial heat therapy in the management of nonspecific, mild-to-moderate low back pain in current clinical practice: a narrative review. Life. 2021;11:780. doi:10.3390/life11080780
- Kerns RD, Philip EJ, Lee AW, et al. Implementation of the Veterans Health Administration National Pain Management Strategy. Transl Behav Med. 2011;1:635-643. doi:10.1007/s13142-011-0094-3
- Brookhart MA, Schneeweiss S, Rothman KJ, et al. Variable selection for propensity score models. Am J Epidemiol. 2006;163:1149-1156. doi:10.1093/aje/kwj149
- Edlund MJ, Martin BC, Devries A, et al. Trends in use of opioids for chronic non-cancer pain among individuals with mental health and substance use disorders: the TROUP study. Clin J Pain. 2010;26:1-8. doi:10.1097/AJP.0b013e3181b99f35
- Kaur S, Stechuchak KM, Coffman CJ, et al. Gender differences in health care utilization among veterans with chronic pain. J Gen Intern Med. 2007;22:228-233. doi:10.1007/s11606-006-0048-5
- Meghani SH, Cho E. Self-reported pain and utilization of pain treatment between minorities and non-minorities in the United States. Public Health Nurs. 2009;26:307-316. doi:10.1111/j.1525-1446.2009.00785.x
- Morasco BJ, Duckart JP, Carr TP, et al. Clinical characteristics of veterans prescribed high doses of opioid medications for chronic non-cancer pain. Pain. 2010;151:625-632. doi:10.1016/j.pain.2010.08.002
- Nielsen S, Lintzeris N, Bruno R, et al. Benzodiazepine use among chronic pain patients prescribed opioids: associations with pain, physical and mental health, and health service utilization. Pain Med. 2015;16:356-366. doi:10.1111/pme.12594
- Quinn PD, Hur K, Chang Z, et al. Incident and long-term opioid therapy among patients with psychiatric conditions and medications: a national study of commercial health care claims. Pain. 2017;158:140-148. doi:10.1097/j.pain.0000000000000730
- Rubin DB. Matching to remove bias in observational studies. Biometrics. 1973;29:159-183.
- Rosenbaum PR, Rubin DB. Constructing a control group using multivariate matched sampling methods that incorporate the propensity score. Am Stat. 1985;39:33-38. doi:10.1007/s11596-025-00052-0
- Steiner PM, Cook TD, Shadish WR, et al. The importance of covariate selection in controlling for selection bias in observational studies. Psychol Methods. 2010;15:250-267. doi:10.1037/a0018719
- Charlson ME, Pompei P, Ales KL, et al. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40:373- 383. doi:10.1016/0021-9681(87)90171-8
- Long JS, Freese J. Regression Models for Categorical Dependent Variables Using Stata. 3rd ed. Stata Press; 2014.
- Muthén LK, Muthén BO. Mplus User’s Guide. 8th ed. Muthén & Muthén; 1998-2017.
- Hedeker D, Gibbons RD. Longitudinal Data Analysis. Wiley; 2006.
- Dobscha SK, Morasco BJ, Kovas AE, et al. Short-term variability in outpatient pain intensity scores in a national sample of older veterans with chronic pain. Pain Med. 2015;16:855-865. doi:10.1111/pme.12643
- Bahreini M, Safaie A, Mirfazaelian H, et al. How much change in pain score does really matter to patients? Am J Emerg Med. 2020;38:1641-1646. doi:10.1016/j.ajem.2019.158489
- Suzuki H, Aono S, Inoue S, et al. Clinically significant changes in pain along the pain intensity numerical rating scale in patients with chronic low back pain. PLoS One. 2020;15:e0229228. doi:10.1371/journal.pone.0229228
- McPherson SM, Smith CL, Dobscha SK, et al. Changes in pain intensity after discontinuation of long-term opioid therapy for chronic noncancer pain. Pain. 2018;159:2097- 2104. doi:10.1097/j.pain.0000000000001315
- Andrews NE, Strong J, Meredith PJ. Activity pacing, avoidance, endurance, and associations with patient functioning in chronic pain: a systematic and meta-analysis. Arch Phys Med Rehabil. 2012;93:2109-121. doi:10.1016/j.apmr.2012.05.029
- Dworkin RH, Turk DC, Farrar JT, et al. Core outcome measures for chronic pain clinical trials: IMMPACT recommendations. Pain. 2005;113:9-19. doi:10.1016/j.pain.2004.09.012
- Gellad WF, Good CB, Shulkin DJ. Addressing the opioid epidemic in the United States: Lessons from the Department of Veterans Affairs. JAMA Intern Med. 2017;177:611- 612. doi:10.1001/jamainternmed.2017.0147
- Lin LA, Bohnert ASB, Kerns RD, et al. Impact of the Opioid Safety Initiative on opioid-related prescribing in veterans. Pain. 2017;158:833-839. doi:10.1097/j.pain.0000000000000837
- Sirucek L, Price RC, Gandhi W, et al. Endogenous opioids contribute to the feeling of pain relief in humans. Pain. 2021;162:2821-2831. doi:10.1097/j.pain.0000000000002285
- Lovejoy TI, Morasco BJ, Demidenko MI, et al. Reasons for discontinuation of long-term opioid therapy in patients with and without substance use disorders. Pain. 2017;158:526- 534. doi:10.1097/j.pain.0000000000000796
- Krebs EE, Gravely A, Nugent S, et al. Effect of opioid vs nonopioid medications on pain-related function in patients with chronic back pain or hip or knee osteoarthritis pain: the SPACE randomized clinical trial. JAMA. 2018;2018:872-882. doi:10.1001/jama.2018.0899
- Becker WC, DeBar LL, Heapy AA, et al. A research agenda for advancing non-pharmacological management of chronic musculoskeletal pain: findings from a VHA state-of-the-art conference. J Gen Intern Med. 2018;33:1-15. doi:10.1007/s11606-018-4345-6
- Fernandes IA, Armond ACV, Falci SGM. The effectiveness of the cold therapy (cryotherapy) in the management of inflammatory parameters after removal of mandibular third molars: a meta-analysis. Int Arch Otorhinolaryngol. 2019;23:221-228. doi:10.1055/s-0039-1677755
- Quinlan P, Davis J, Fields K, et al. Effects of localized cold therapy on pain in postoperative spinal fusion patients: a randomized controlled trial. Orthop Nurs. 2017;36:344-349. doi:10.1097/NOR.0000000000000382
- Malanga GA, Yan N, Stark J. Mechanisms and efficacy of heat and cold therapies for musculoskeletal injury. Postgrad Med. 2015;127:57-65. doi:10.1080/00325481.2015.992719
- Wang Y, Li S, Zhang Y, et al. Heat and cold therapy reduce pain in patients with delayed onset muscle soreness: a systematic review and meta-analysis of 32 randomized controlled trials. Phys Ther Sport. 2021;48:177-187. doi:10.1016/j.ptsp.2021.01.004
- Ariana M, Afrasiabifar A, Doulatabad SN, et al. The effect of local heat therapy versus cold rub gel on pain and joint functions in patients with knee osteoarthritis. Clin Nurs Res. 2022;31:1014-1022. doi:10.1177/10547738211035502
- French SD, Cameron M, Walker BF, et al. Superficial heat or cold for low back pain. Cochrane Database Syst Rev. 2006;1:CD004750. doi:10.1002/14651858.CD004750.pub2
- Tao XG, Bernacki EJ. A randomized clinical trial of continuous low-level heat therapy for acute muscular low back pain in the workplace. J Occup Environ Med. 2005;47:1298- 1306. doi:10.1097/01.jom.0000184877.01691.a3
- Freiwald J, Hoppe MW, Beermann W, et al. Effects of supplemental heat therapy in multimodal treated chronic low back pain patients on strength and flexibility. Clin Biomech. 2018;57:107-113. doi:10.1016/j.clinbiomech.2018.06.008
- Freiwald J, Magni A, Fanlo-Mazas P, et al. A role for superficial heat therapy in the management of nonspecific, mild-to-moderate low back pain in current clinical practice: a narrative review. Life. 2021;11:780. doi:10.3390/life11080780
- Kerns RD, Philip EJ, Lee AW, et al. Implementation of the Veterans Health Administration National Pain Management Strategy. Transl Behav Med. 2011;1:635-643. doi:10.1007/s13142-011-0094-3
- Brookhart MA, Schneeweiss S, Rothman KJ, et al. Variable selection for propensity score models. Am J Epidemiol. 2006;163:1149-1156. doi:10.1093/aje/kwj149
- Edlund MJ, Martin BC, Devries A, et al. Trends in use of opioids for chronic non-cancer pain among individuals with mental health and substance use disorders: the TROUP study. Clin J Pain. 2010;26:1-8. doi:10.1097/AJP.0b013e3181b99f35
- Kaur S, Stechuchak KM, Coffman CJ, et al. Gender differences in health care utilization among veterans with chronic pain. J Gen Intern Med. 2007;22:228-233. doi:10.1007/s11606-006-0048-5
- Meghani SH, Cho E. Self-reported pain and utilization of pain treatment between minorities and non-minorities in the United States. Public Health Nurs. 2009;26:307-316. doi:10.1111/j.1525-1446.2009.00785.x
- Morasco BJ, Duckart JP, Carr TP, et al. Clinical characteristics of veterans prescribed high doses of opioid medications for chronic non-cancer pain. Pain. 2010;151:625-632. doi:10.1016/j.pain.2010.08.002
- Nielsen S, Lintzeris N, Bruno R, et al. Benzodiazepine use among chronic pain patients prescribed opioids: associations with pain, physical and mental health, and health service utilization. Pain Med. 2015;16:356-366. doi:10.1111/pme.12594
- Quinn PD, Hur K, Chang Z, et al. Incident and long-term opioid therapy among patients with psychiatric conditions and medications: a national study of commercial health care claims. Pain. 2017;158:140-148. doi:10.1097/j.pain.0000000000000730
- Rubin DB. Matching to remove bias in observational studies. Biometrics. 1973;29:159-183.
- Rosenbaum PR, Rubin DB. Constructing a control group using multivariate matched sampling methods that incorporate the propensity score. Am Stat. 1985;39:33-38. doi:10.1007/s11596-025-00052-0
- Steiner PM, Cook TD, Shadish WR, et al. The importance of covariate selection in controlling for selection bias in observational studies. Psychol Methods. 2010;15:250-267. doi:10.1037/a0018719
- Charlson ME, Pompei P, Ales KL, et al. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40:373- 383. doi:10.1016/0021-9681(87)90171-8
- Long JS, Freese J. Regression Models for Categorical Dependent Variables Using Stata. 3rd ed. Stata Press; 2014.
- Muthén LK, Muthén BO. Mplus User’s Guide. 8th ed. Muthén & Muthén; 1998-2017.
- Hedeker D, Gibbons RD. Longitudinal Data Analysis. Wiley; 2006.
- Dobscha SK, Morasco BJ, Kovas AE, et al. Short-term variability in outpatient pain intensity scores in a national sample of older veterans with chronic pain. Pain Med. 2015;16:855-865. doi:10.1111/pme.12643
- Bahreini M, Safaie A, Mirfazaelian H, et al. How much change in pain score does really matter to patients? Am J Emerg Med. 2020;38:1641-1646. doi:10.1016/j.ajem.2019.158489
- Suzuki H, Aono S, Inoue S, et al. Clinically significant changes in pain along the pain intensity numerical rating scale in patients with chronic low back pain. PLoS One. 2020;15:e0229228. doi:10.1371/journal.pone.0229228
- McPherson SM, Smith CL, Dobscha SK, et al. Changes in pain intensity after discontinuation of long-term opioid therapy for chronic noncancer pain. Pain. 2018;159:2097- 2104. doi:10.1097/j.pain.0000000000001315
- Andrews NE, Strong J, Meredith PJ. Activity pacing, avoidance, endurance, and associations with patient functioning in chronic pain: a systematic and meta-analysis. Arch Phys Med Rehabil. 2012;93:2109-121. doi:10.1016/j.apmr.2012.05.029
- Dworkin RH, Turk DC, Farrar JT, et al. Core outcome measures for chronic pain clinical trials: IMMPACT recommendations. Pain. 2005;113:9-19. doi:10.1016/j.pain.2004.09.012
- Gellad WF, Good CB, Shulkin DJ. Addressing the opioid epidemic in the United States: Lessons from the Department of Veterans Affairs. JAMA Intern Med. 2017;177:611- 612. doi:10.1001/jamainternmed.2017.0147
- Lin LA, Bohnert ASB, Kerns RD, et al. Impact of the Opioid Safety Initiative on opioid-related prescribing in veterans. Pain. 2017;158:833-839. doi:10.1097/j.pain.0000000000000837
- Sirucek L, Price RC, Gandhi W, et al. Endogenous opioids contribute to the feeling of pain relief in humans. Pain. 2021;162:2821-2831. doi:10.1097/j.pain.0000000000002285
- Lovejoy TI, Morasco BJ, Demidenko MI, et al. Reasons for discontinuation of long-term opioid therapy in patients with and without substance use disorders. Pain. 2017;158:526- 534. doi:10.1097/j.pain.0000000000000796
Thermal Therapy is Associated With Decreased Pain Treatment Use: A Retrospective Cohort Study
Thermal Therapy is Associated With Decreased Pain Treatment Use: A Retrospective Cohort Study
Time to Reconsider Tramadol for Chronic Pain?
Time to Reconsider Tramadol for Chronic Pain?
Tramadol, a commonly prescribed opioid often viewed as a safer option for chronic pain, provided limited pain relief while increasing the risk for serious adverse effects, results of a new analysis showed.
In a systematic review and meta-analysis, investigators found that tramadol offered clinically insignificant pain relief, while doubling the likelihood of serious adverse events, most commonly cardiac complications.
"Given the limited analgesic benefits and increased risk of harm, tramadol use for chronic pain should be reconsidered," Jehad Ahmad Barakji, MD, of the Centre for Clinical Intervention Research at Rigshospitalet in Copenhagen, Denmark, told Medscape Medical News.
"Across different chronic pain conditions, tramadol's pain-relieving effect appears modest, and while some patients may experience relief, most will not gain substantial benefit," he added.
However, the researchers cautioned that the certainty of the evidence was low-to-moderate and that the quality of the underlying trials varied substantially.
The study was published on October 7 in BMJ Evidence-Based Medicine.
Popularity Outpacing Proof
Tramadol, a dual-action opioid that modulates serotonin and norepinephrine pathways, has long been promoted as a middle-ground analgesic - less addictive than morphine but stronger than nonopioid medications. It is approved for moderate-to-severe pain, including postoperative and chronic conditions.
Prescriptions have risen sharply worldwide, fueled by perceptions of safety and a belief that tramadol carries a lower risk for dependence. A recent global analysis estimated that nearly 18% of adults have used tramadol in their lifetime, and > 80% of those users combined it with at least 1 other substance.
Despite its widespread use, evidence supporting tramadol's long-term effectiveness and safety in chronic pain has been limited and inconsistent. Previous research has largely focused on short-term or condition-specific outcomes, such as osteoarthritis or neuropathic pain, leaving uncertainty about the drug's overall risk-benefit profile.
Small Benefit, Big Risk
The current study is the first comprehensive systematic review to evaluate tramadol alone for chronic pain using both meta-analysis and trail sequential analysis, provided a broader view of efficacy and safety across pain types.
"We sought to fill this gap by evaluating the totality of evidence to guide clinical practice," Barakji said.
The analysis included 19 randomized, placebo-controlled trials with 6569 adults. The average participant age was 56 years, and study durations ranged from 4 to 16 weeks. Pain intensity was typically assessed with the 0-10 numeric rating scale (NRS), while function and quality of life were measured with validated patient-reported tools.
Across the pooled analysis, participants receiving tramadol experienced an average pain reduction of 0.9 points on the NRS compared with placebo - a difference below the 1-point threshold considered clinically meaningful. About 7% more patients in the tramadol groups achieved noticeable pain relief, but investigators said the benefit was modest and uncertain.
Serious adverse events were twice as common among tramadol users, most often involving cardiac complications such as chest pain, coronary artery disease, or heart failure. Nonserious adverse effects, including nausea, dizziness, constipation, and drowsiness, were frequent and contributed to higher discontinuation rates among tramadol recipients.
The researchers acknowledged that most included trials were at a high risk for bias due to incomplete outcome reporting, small sample sizes, and inconsistent assessment methods, factors that may have exaggerated benefits and underestimated harms.
Reevaluating Tramadol's Role
Commenting on the research, Jessica Otte, MD, clinical associate professor in the Department of Family Medicine at The University of British Columbia, Vancouver, British Columbia, Canada, said the new review stands out for examining tramadol's use across a range of chronic pain conditions, an area where clinicians often struggle to help patients achieve meaningful and sustained relief.
Otte, who has studied the drug's safety and effectiveness through The University of British Columbia's Therapeutics Initiative, said the Danish team's analysis expands on earlier research that largely focused on single pain conditions. The findings, she said, add weight to growing evidence that challenges tramadol's perceived advantages over other analgesics.
"This review doesn't change the narrative but strengthens it: Tramdol's reputation as a safer or uniquely effective opioid is increasingly difficult to defend," she told Medscape Medical News.
While she believes the study makes an important contribution, Otte said the results should be interpreted with caution because of gaps in the underlying evidence. The Danish authors noted similar concerns, and Otte agreed that many of the included trials had methodological shortcomings.
"A lot of the studies had biases that made use less certain about what was reported," she said. "Many outcomes that matter to patients - like quality of life, functional improvement, or withdrawal - were either inconsistently measured or not reported at all."
She added that patients assigned to tramadol were more likely to discontinue early, both overall and because of adverse effects, raising concern about tolerability and attrition bias.
Also commenting, Houman Danesh, MD, professor and medical director of pain management at the Icahn School of Medicine at Mount Sinai in New York City, said the review provides moderate-certainty evidence that tramadol increases the risk for serious adverse events; but its broad approach - evaluating efficacy and safety across multiple chronic pain conditions - could be a confounding factor.
"Tramadol may benefit some conditions more than others," he noted, which could alter the overall risk-benefit profile.
In his clinical experience, Danesh said severe complications such as cardiac events are uncommon. He explained that heart rhythm disturbances occasionally associated with tramadol generally arise when patients are taking other medications that affect cardiac conduction.
Danesh emphasized that clinicians should weigh the study alongside other research and their own experience when deciding whether to prescribe the drug.
"It's important to take this study into consideration," he said, "but there are multiple studies that support the use of tramadol, and we have to look at the totality of the evidence."
Written by Carla Cantor.
The authors declared no competing interests and received no specific grant from any funding agency. Otte and Danesh reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Tramadol, a commonly prescribed opioid often viewed as a safer option for chronic pain, provided limited pain relief while increasing the risk for serious adverse effects, results of a new analysis showed.
In a systematic review and meta-analysis, investigators found that tramadol offered clinically insignificant pain relief, while doubling the likelihood of serious adverse events, most commonly cardiac complications.
"Given the limited analgesic benefits and increased risk of harm, tramadol use for chronic pain should be reconsidered," Jehad Ahmad Barakji, MD, of the Centre for Clinical Intervention Research at Rigshospitalet in Copenhagen, Denmark, told Medscape Medical News.
"Across different chronic pain conditions, tramadol's pain-relieving effect appears modest, and while some patients may experience relief, most will not gain substantial benefit," he added.
However, the researchers cautioned that the certainty of the evidence was low-to-moderate and that the quality of the underlying trials varied substantially.
The study was published on October 7 in BMJ Evidence-Based Medicine.
Popularity Outpacing Proof
Tramadol, a dual-action opioid that modulates serotonin and norepinephrine pathways, has long been promoted as a middle-ground analgesic - less addictive than morphine but stronger than nonopioid medications. It is approved for moderate-to-severe pain, including postoperative and chronic conditions.
Prescriptions have risen sharply worldwide, fueled by perceptions of safety and a belief that tramadol carries a lower risk for dependence. A recent global analysis estimated that nearly 18% of adults have used tramadol in their lifetime, and > 80% of those users combined it with at least 1 other substance.
Despite its widespread use, evidence supporting tramadol's long-term effectiveness and safety in chronic pain has been limited and inconsistent. Previous research has largely focused on short-term or condition-specific outcomes, such as osteoarthritis or neuropathic pain, leaving uncertainty about the drug's overall risk-benefit profile.
Small Benefit, Big Risk
The current study is the first comprehensive systematic review to evaluate tramadol alone for chronic pain using both meta-analysis and trail sequential analysis, provided a broader view of efficacy and safety across pain types.
"We sought to fill this gap by evaluating the totality of evidence to guide clinical practice," Barakji said.
The analysis included 19 randomized, placebo-controlled trials with 6569 adults. The average participant age was 56 years, and study durations ranged from 4 to 16 weeks. Pain intensity was typically assessed with the 0-10 numeric rating scale (NRS), while function and quality of life were measured with validated patient-reported tools.
Across the pooled analysis, participants receiving tramadol experienced an average pain reduction of 0.9 points on the NRS compared with placebo - a difference below the 1-point threshold considered clinically meaningful. About 7% more patients in the tramadol groups achieved noticeable pain relief, but investigators said the benefit was modest and uncertain.
Serious adverse events were twice as common among tramadol users, most often involving cardiac complications such as chest pain, coronary artery disease, or heart failure. Nonserious adverse effects, including nausea, dizziness, constipation, and drowsiness, were frequent and contributed to higher discontinuation rates among tramadol recipients.
The researchers acknowledged that most included trials were at a high risk for bias due to incomplete outcome reporting, small sample sizes, and inconsistent assessment methods, factors that may have exaggerated benefits and underestimated harms.
Reevaluating Tramadol's Role
Commenting on the research, Jessica Otte, MD, clinical associate professor in the Department of Family Medicine at The University of British Columbia, Vancouver, British Columbia, Canada, said the new review stands out for examining tramadol's use across a range of chronic pain conditions, an area where clinicians often struggle to help patients achieve meaningful and sustained relief.
Otte, who has studied the drug's safety and effectiveness through The University of British Columbia's Therapeutics Initiative, said the Danish team's analysis expands on earlier research that largely focused on single pain conditions. The findings, she said, add weight to growing evidence that challenges tramadol's perceived advantages over other analgesics.
"This review doesn't change the narrative but strengthens it: Tramdol's reputation as a safer or uniquely effective opioid is increasingly difficult to defend," she told Medscape Medical News.
While she believes the study makes an important contribution, Otte said the results should be interpreted with caution because of gaps in the underlying evidence. The Danish authors noted similar concerns, and Otte agreed that many of the included trials had methodological shortcomings.
"A lot of the studies had biases that made use less certain about what was reported," she said. "Many outcomes that matter to patients - like quality of life, functional improvement, or withdrawal - were either inconsistently measured or not reported at all."
She added that patients assigned to tramadol were more likely to discontinue early, both overall and because of adverse effects, raising concern about tolerability and attrition bias.
Also commenting, Houman Danesh, MD, professor and medical director of pain management at the Icahn School of Medicine at Mount Sinai in New York City, said the review provides moderate-certainty evidence that tramadol increases the risk for serious adverse events; but its broad approach - evaluating efficacy and safety across multiple chronic pain conditions - could be a confounding factor.
"Tramadol may benefit some conditions more than others," he noted, which could alter the overall risk-benefit profile.
In his clinical experience, Danesh said severe complications such as cardiac events are uncommon. He explained that heart rhythm disturbances occasionally associated with tramadol generally arise when patients are taking other medications that affect cardiac conduction.
Danesh emphasized that clinicians should weigh the study alongside other research and their own experience when deciding whether to prescribe the drug.
"It's important to take this study into consideration," he said, "but there are multiple studies that support the use of tramadol, and we have to look at the totality of the evidence."
Written by Carla Cantor.
The authors declared no competing interests and received no specific grant from any funding agency. Otte and Danesh reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Tramadol, a commonly prescribed opioid often viewed as a safer option for chronic pain, provided limited pain relief while increasing the risk for serious adverse effects, results of a new analysis showed.
In a systematic review and meta-analysis, investigators found that tramadol offered clinically insignificant pain relief, while doubling the likelihood of serious adverse events, most commonly cardiac complications.
"Given the limited analgesic benefits and increased risk of harm, tramadol use for chronic pain should be reconsidered," Jehad Ahmad Barakji, MD, of the Centre for Clinical Intervention Research at Rigshospitalet in Copenhagen, Denmark, told Medscape Medical News.
"Across different chronic pain conditions, tramadol's pain-relieving effect appears modest, and while some patients may experience relief, most will not gain substantial benefit," he added.
However, the researchers cautioned that the certainty of the evidence was low-to-moderate and that the quality of the underlying trials varied substantially.
The study was published on October 7 in BMJ Evidence-Based Medicine.
Popularity Outpacing Proof
Tramadol, a dual-action opioid that modulates serotonin and norepinephrine pathways, has long been promoted as a middle-ground analgesic - less addictive than morphine but stronger than nonopioid medications. It is approved for moderate-to-severe pain, including postoperative and chronic conditions.
Prescriptions have risen sharply worldwide, fueled by perceptions of safety and a belief that tramadol carries a lower risk for dependence. A recent global analysis estimated that nearly 18% of adults have used tramadol in their lifetime, and > 80% of those users combined it with at least 1 other substance.
Despite its widespread use, evidence supporting tramadol's long-term effectiveness and safety in chronic pain has been limited and inconsistent. Previous research has largely focused on short-term or condition-specific outcomes, such as osteoarthritis or neuropathic pain, leaving uncertainty about the drug's overall risk-benefit profile.
Small Benefit, Big Risk
The current study is the first comprehensive systematic review to evaluate tramadol alone for chronic pain using both meta-analysis and trail sequential analysis, provided a broader view of efficacy and safety across pain types.
"We sought to fill this gap by evaluating the totality of evidence to guide clinical practice," Barakji said.
The analysis included 19 randomized, placebo-controlled trials with 6569 adults. The average participant age was 56 years, and study durations ranged from 4 to 16 weeks. Pain intensity was typically assessed with the 0-10 numeric rating scale (NRS), while function and quality of life were measured with validated patient-reported tools.
Across the pooled analysis, participants receiving tramadol experienced an average pain reduction of 0.9 points on the NRS compared with placebo - a difference below the 1-point threshold considered clinically meaningful. About 7% more patients in the tramadol groups achieved noticeable pain relief, but investigators said the benefit was modest and uncertain.
Serious adverse events were twice as common among tramadol users, most often involving cardiac complications such as chest pain, coronary artery disease, or heart failure. Nonserious adverse effects, including nausea, dizziness, constipation, and drowsiness, were frequent and contributed to higher discontinuation rates among tramadol recipients.
The researchers acknowledged that most included trials were at a high risk for bias due to incomplete outcome reporting, small sample sizes, and inconsistent assessment methods, factors that may have exaggerated benefits and underestimated harms.
Reevaluating Tramadol's Role
Commenting on the research, Jessica Otte, MD, clinical associate professor in the Department of Family Medicine at The University of British Columbia, Vancouver, British Columbia, Canada, said the new review stands out for examining tramadol's use across a range of chronic pain conditions, an area where clinicians often struggle to help patients achieve meaningful and sustained relief.
Otte, who has studied the drug's safety and effectiveness through The University of British Columbia's Therapeutics Initiative, said the Danish team's analysis expands on earlier research that largely focused on single pain conditions. The findings, she said, add weight to growing evidence that challenges tramadol's perceived advantages over other analgesics.
"This review doesn't change the narrative but strengthens it: Tramdol's reputation as a safer or uniquely effective opioid is increasingly difficult to defend," she told Medscape Medical News.
While she believes the study makes an important contribution, Otte said the results should be interpreted with caution because of gaps in the underlying evidence. The Danish authors noted similar concerns, and Otte agreed that many of the included trials had methodological shortcomings.
"A lot of the studies had biases that made use less certain about what was reported," she said. "Many outcomes that matter to patients - like quality of life, functional improvement, or withdrawal - were either inconsistently measured or not reported at all."
She added that patients assigned to tramadol were more likely to discontinue early, both overall and because of adverse effects, raising concern about tolerability and attrition bias.
Also commenting, Houman Danesh, MD, professor and medical director of pain management at the Icahn School of Medicine at Mount Sinai in New York City, said the review provides moderate-certainty evidence that tramadol increases the risk for serious adverse events; but its broad approach - evaluating efficacy and safety across multiple chronic pain conditions - could be a confounding factor.
"Tramadol may benefit some conditions more than others," he noted, which could alter the overall risk-benefit profile.
In his clinical experience, Danesh said severe complications such as cardiac events are uncommon. He explained that heart rhythm disturbances occasionally associated with tramadol generally arise when patients are taking other medications that affect cardiac conduction.
Danesh emphasized that clinicians should weigh the study alongside other research and their own experience when deciding whether to prescribe the drug.
"It's important to take this study into consideration," he said, "but there are multiple studies that support the use of tramadol, and we have to look at the totality of the evidence."
Written by Carla Cantor.
The authors declared no competing interests and received no specific grant from any funding agency. Otte and Danesh reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Time to Reconsider Tramadol for Chronic Pain?
Time to Reconsider Tramadol for Chronic Pain?
Is low-dose naltrexone effective in chronic pain management?
Evidence summary
Naltrexone is comparable to amitriptyline for diabetic neuropathy pain
A 2021 randomized, double-blind, active-comparator, crossover clinical trial conducted in India examined the efficacy of low-dose naltrexone vs standard-of-care amitriptyline in patients (N = 67) with painful diabetic neuropathy. Participants were adults (ages 18 to 75 years) with painful diabetic neuropathy who had been on a stable dose of nonopioid pain medication for at least 1 month.1
Patients were randomly assigned to start receiving naltrexone 2 mg (n = 33) or amitriptyline 10 mg (n = 34). They received their starting medication for 6 weeks (with follow-up every 2 weeks), then completed a 2-week washout period, and then switched to the other study medication for 6 weeks (same follow-up schedule). If patients reported < 20% pain reduction on the Visual Analog Scale (VAS; 0-100 scoring system with 0 = no pain and 100 = worst pain) at a follow-up visit, their medication dose was titrated up, to a maximum of 4 mg of naltrexone or 25 to 50 mg of amitriptyline.1
The primary outcome of interest was the mean change in VAS pain score following 6 weeks of treatment. There was no statistically different change from baseline VAS pain score between the amitriptyline and naltrexone groups (mean difference [MD] = 1.6; 95% CI, –0.9 to 4.2; P = 0.21). These findings were consistent across the secondary endpoints (Likert 5-point pain scale and McGill Pain Questionnaire scores). There was no statistically significant difference in Hamilton Depression Rating Scale scores (13 in the naltrexone group vs 11 in the amitriptyline group; P = .81), no reports of decreased sleep quality in either group, and no significant difference in Patients’ Global Impression of Change scores at 6-week evaluation.1
The naltrexone cohort experienced 8 adverse events (most commonly, mild diarrhea), while the amitriptyline cohort experienced 52 adverse events (most commonly, somnolence) (P < .001). The limitations of the study include the lack of a placebo arm and a relatively small sample size.1
Greater reduction in pain scores with naltrexone
A 2022 retrospective cohort study evaluated the effectiveness of naltrexone for patients treated at a single outpatient integrative pain management practice in Alaska between 2014 and 2019. The exposure group (n = 36) included patients who had completed at least a 2-month continuous regimen of oral naltrexone 4.5 mg. Controls (n = 42) were selected from the remaining practice population receiving standard care and were primarily matched by diagnosis code, followed by gender, then age +/– 5 years. Patients were divided into subgroups for inflammatory and neuropathic pain.2
The primary outcome measured was the mean change in VAS score or numeric rating score (NRS; both used a 1-10 rating system), which was assessed during a patient’s appointment from initiation of treatment to the most recent visit or at the termination of therapy (intervention interquartile range, 12-14 months). There was no statistically significant difference in VAS/NRS between the low-dose naltrexone and control groups at baseline (6.09 vs 6.38; P = .454). The low-dose naltrexone group experienced a greater reduction in VAS/NRS pain scores compared to the control group (–37.8% vs –4.3%; P < .001).2
Compared with control patients in each group, patients in the inflammatory pain subgroup and the neuropathic pain subgroup who received low-dose naltrexone reported reductions in pain scores of 32% (P < .001) and 44% (P = .048), respectively. There was no statistically significant difference in mean change in VAS/NRS scores between the inflammatory and neuropathic subgroups (P = .763). A multivariate linear regression analysis did not identify significant variables other than low-dose naltrexone that correlated with pain improvement. The number needed to treat to observe a ≥ 50% reduction in pain scores was 3.2.2
Continue to: Limitations for this study...
Limitations for this study include its small sample size and open-label design.2
Low-dose naltrexone is effective for fibromyalgia pain
A 2020 single-blind prospective dose-response study utilized the up-and-down method to identify effective naltrexone dose for patients in a Danish university hospital pain clinic. Patients were White women ages 18 to 60 years (N = 25) who had a diagnosis of fibromyalgia unresponsive to traditional pharmacologic treatment. All patients received treatment with low-dose naltrexone (ranging from 0.75 mg to 6.0 mg) but were blinded to dose.3
Patients were evaluated for improvement in fibromyalgia symptoms using the Patient Global Impression of Improvement (PGI-I) scale—which ranges from 1 (very much improved) to 7 (very much worse), with 4 being “no change”—at baseline and after 2 to 3 weeks of treatment with low-dose naltrexone. A patient was considered a responder if they scored 1 to 3 on the follow-up PGI-I scale or if they experienced a > 30% pain reduction on the VAS. If a patient did not respond to their dose, the next patient began treatment at a dose 0.75 mg higher than the previous patient’s ending dose. If a patient did respond to low-dose naltrexone treatment, the next patient’s starting dose was 0.75 mg less than the previous patient’s. Eleven of 25 patients were considered responders.3
The primary outcomes were effective dose for 50% of fibromyalgia patients (3.88 mg; 95% CI, 3.39-4.35) and effective dose for 95% of fibromyalgia patients (5.4 mg; 95% CI, 4.66-6.13). Secondary outcomes were fibromyalgia symptoms as evaluated on the Fibromyalgia Impact Questionnaire Revised. Five of the 11 responders reported a > 30% improvement in tenderness and 8 of the 11 responders reported a > 30% decrease in waking unrefreshed.3
Limitations of the study include the short time period of treatment before response was assessed and the decision to use low test doses, which may have hindered detection of effective doses > 6 mg in fibromyalgia.3
Editor’s takeaway
Low-dose naltrexone, a less-often-used form of pain management, is a welcome option. Studies show some effectiveness in a variety of pain conditions with few adverse effects. The small number of studies, the small sample sizes, and the limited follow-up duration should encourage more investigation into how to best use this intervention.
1. Srinivasan A, Dutta P, Bansal D, et al. Efficacy and safety of low-dose naltrexone in painful diabetic neuropathy: a randomized, double-blind, active-control, crossover clinical trial. J Diabetes. 2021;13:770-778. doi: 10.1111/1753-0407.13202
2. Martin SJ, McAnally HB, Okediji P, et al. Low-dose naltrexone, an opioid-receptor antagonist, is a broad-spectrum analgesic: a retrospective cohort study. Pain Management. 2022;12:699-709. doi: 10.2217/pmt-2021-0122
3. Bruun-Plesner K, Blichfeldt-Eckhardt MR, Vaegter HB, et al. Low-dose naltrexone for the treatment of fibromyalgia: investigation of dose-response relationships. Pain Med. 2020;21:2253-2261. doi: 10.1093/pm/pnaa001
Evidence summary
Naltrexone is comparable to amitriptyline for diabetic neuropathy pain
A 2021 randomized, double-blind, active-comparator, crossover clinical trial conducted in India examined the efficacy of low-dose naltrexone vs standard-of-care amitriptyline in patients (N = 67) with painful diabetic neuropathy. Participants were adults (ages 18 to 75 years) with painful diabetic neuropathy who had been on a stable dose of nonopioid pain medication for at least 1 month.1
Patients were randomly assigned to start receiving naltrexone 2 mg (n = 33) or amitriptyline 10 mg (n = 34). They received their starting medication for 6 weeks (with follow-up every 2 weeks), then completed a 2-week washout period, and then switched to the other study medication for 6 weeks (same follow-up schedule). If patients reported < 20% pain reduction on the Visual Analog Scale (VAS; 0-100 scoring system with 0 = no pain and 100 = worst pain) at a follow-up visit, their medication dose was titrated up, to a maximum of 4 mg of naltrexone or 25 to 50 mg of amitriptyline.1
The primary outcome of interest was the mean change in VAS pain score following 6 weeks of treatment. There was no statistically different change from baseline VAS pain score between the amitriptyline and naltrexone groups (mean difference [MD] = 1.6; 95% CI, –0.9 to 4.2; P = 0.21). These findings were consistent across the secondary endpoints (Likert 5-point pain scale and McGill Pain Questionnaire scores). There was no statistically significant difference in Hamilton Depression Rating Scale scores (13 in the naltrexone group vs 11 in the amitriptyline group; P = .81), no reports of decreased sleep quality in either group, and no significant difference in Patients’ Global Impression of Change scores at 6-week evaluation.1
The naltrexone cohort experienced 8 adverse events (most commonly, mild diarrhea), while the amitriptyline cohort experienced 52 adverse events (most commonly, somnolence) (P < .001). The limitations of the study include the lack of a placebo arm and a relatively small sample size.1
Greater reduction in pain scores with naltrexone
A 2022 retrospective cohort study evaluated the effectiveness of naltrexone for patients treated at a single outpatient integrative pain management practice in Alaska between 2014 and 2019. The exposure group (n = 36) included patients who had completed at least a 2-month continuous regimen of oral naltrexone 4.5 mg. Controls (n = 42) were selected from the remaining practice population receiving standard care and were primarily matched by diagnosis code, followed by gender, then age +/– 5 years. Patients were divided into subgroups for inflammatory and neuropathic pain.2
The primary outcome measured was the mean change in VAS score or numeric rating score (NRS; both used a 1-10 rating system), which was assessed during a patient’s appointment from initiation of treatment to the most recent visit or at the termination of therapy (intervention interquartile range, 12-14 months). There was no statistically significant difference in VAS/NRS between the low-dose naltrexone and control groups at baseline (6.09 vs 6.38; P = .454). The low-dose naltrexone group experienced a greater reduction in VAS/NRS pain scores compared to the control group (–37.8% vs –4.3%; P < .001).2
Compared with control patients in each group, patients in the inflammatory pain subgroup and the neuropathic pain subgroup who received low-dose naltrexone reported reductions in pain scores of 32% (P < .001) and 44% (P = .048), respectively. There was no statistically significant difference in mean change in VAS/NRS scores between the inflammatory and neuropathic subgroups (P = .763). A multivariate linear regression analysis did not identify significant variables other than low-dose naltrexone that correlated with pain improvement. The number needed to treat to observe a ≥ 50% reduction in pain scores was 3.2.2
Continue to: Limitations for this study...
Limitations for this study include its small sample size and open-label design.2
Low-dose naltrexone is effective for fibromyalgia pain
A 2020 single-blind prospective dose-response study utilized the up-and-down method to identify effective naltrexone dose for patients in a Danish university hospital pain clinic. Patients were White women ages 18 to 60 years (N = 25) who had a diagnosis of fibromyalgia unresponsive to traditional pharmacologic treatment. All patients received treatment with low-dose naltrexone (ranging from 0.75 mg to 6.0 mg) but were blinded to dose.3
Patients were evaluated for improvement in fibromyalgia symptoms using the Patient Global Impression of Improvement (PGI-I) scale—which ranges from 1 (very much improved) to 7 (very much worse), with 4 being “no change”—at baseline and after 2 to 3 weeks of treatment with low-dose naltrexone. A patient was considered a responder if they scored 1 to 3 on the follow-up PGI-I scale or if they experienced a > 30% pain reduction on the VAS. If a patient did not respond to their dose, the next patient began treatment at a dose 0.75 mg higher than the previous patient’s ending dose. If a patient did respond to low-dose naltrexone treatment, the next patient’s starting dose was 0.75 mg less than the previous patient’s. Eleven of 25 patients were considered responders.3
The primary outcomes were effective dose for 50% of fibromyalgia patients (3.88 mg; 95% CI, 3.39-4.35) and effective dose for 95% of fibromyalgia patients (5.4 mg; 95% CI, 4.66-6.13). Secondary outcomes were fibromyalgia symptoms as evaluated on the Fibromyalgia Impact Questionnaire Revised. Five of the 11 responders reported a > 30% improvement in tenderness and 8 of the 11 responders reported a > 30% decrease in waking unrefreshed.3
Limitations of the study include the short time period of treatment before response was assessed and the decision to use low test doses, which may have hindered detection of effective doses > 6 mg in fibromyalgia.3
Editor’s takeaway
Low-dose naltrexone, a less-often-used form of pain management, is a welcome option. Studies show some effectiveness in a variety of pain conditions with few adverse effects. The small number of studies, the small sample sizes, and the limited follow-up duration should encourage more investigation into how to best use this intervention.
Evidence summary
Naltrexone is comparable to amitriptyline for diabetic neuropathy pain
A 2021 randomized, double-blind, active-comparator, crossover clinical trial conducted in India examined the efficacy of low-dose naltrexone vs standard-of-care amitriptyline in patients (N = 67) with painful diabetic neuropathy. Participants were adults (ages 18 to 75 years) with painful diabetic neuropathy who had been on a stable dose of nonopioid pain medication for at least 1 month.1
Patients were randomly assigned to start receiving naltrexone 2 mg (n = 33) or amitriptyline 10 mg (n = 34). They received their starting medication for 6 weeks (with follow-up every 2 weeks), then completed a 2-week washout period, and then switched to the other study medication for 6 weeks (same follow-up schedule). If patients reported < 20% pain reduction on the Visual Analog Scale (VAS; 0-100 scoring system with 0 = no pain and 100 = worst pain) at a follow-up visit, their medication dose was titrated up, to a maximum of 4 mg of naltrexone or 25 to 50 mg of amitriptyline.1
The primary outcome of interest was the mean change in VAS pain score following 6 weeks of treatment. There was no statistically different change from baseline VAS pain score between the amitriptyline and naltrexone groups (mean difference [MD] = 1.6; 95% CI, –0.9 to 4.2; P = 0.21). These findings were consistent across the secondary endpoints (Likert 5-point pain scale and McGill Pain Questionnaire scores). There was no statistically significant difference in Hamilton Depression Rating Scale scores (13 in the naltrexone group vs 11 in the amitriptyline group; P = .81), no reports of decreased sleep quality in either group, and no significant difference in Patients’ Global Impression of Change scores at 6-week evaluation.1
The naltrexone cohort experienced 8 adverse events (most commonly, mild diarrhea), while the amitriptyline cohort experienced 52 adverse events (most commonly, somnolence) (P < .001). The limitations of the study include the lack of a placebo arm and a relatively small sample size.1
Greater reduction in pain scores with naltrexone
A 2022 retrospective cohort study evaluated the effectiveness of naltrexone for patients treated at a single outpatient integrative pain management practice in Alaska between 2014 and 2019. The exposure group (n = 36) included patients who had completed at least a 2-month continuous regimen of oral naltrexone 4.5 mg. Controls (n = 42) were selected from the remaining practice population receiving standard care and were primarily matched by diagnosis code, followed by gender, then age +/– 5 years. Patients were divided into subgroups for inflammatory and neuropathic pain.2
The primary outcome measured was the mean change in VAS score or numeric rating score (NRS; both used a 1-10 rating system), which was assessed during a patient’s appointment from initiation of treatment to the most recent visit or at the termination of therapy (intervention interquartile range, 12-14 months). There was no statistically significant difference in VAS/NRS between the low-dose naltrexone and control groups at baseline (6.09 vs 6.38; P = .454). The low-dose naltrexone group experienced a greater reduction in VAS/NRS pain scores compared to the control group (–37.8% vs –4.3%; P < .001).2
Compared with control patients in each group, patients in the inflammatory pain subgroup and the neuropathic pain subgroup who received low-dose naltrexone reported reductions in pain scores of 32% (P < .001) and 44% (P = .048), respectively. There was no statistically significant difference in mean change in VAS/NRS scores between the inflammatory and neuropathic subgroups (P = .763). A multivariate linear regression analysis did not identify significant variables other than low-dose naltrexone that correlated with pain improvement. The number needed to treat to observe a ≥ 50% reduction in pain scores was 3.2.2
Continue to: Limitations for this study...
Limitations for this study include its small sample size and open-label design.2
Low-dose naltrexone is effective for fibromyalgia pain
A 2020 single-blind prospective dose-response study utilized the up-and-down method to identify effective naltrexone dose for patients in a Danish university hospital pain clinic. Patients were White women ages 18 to 60 years (N = 25) who had a diagnosis of fibromyalgia unresponsive to traditional pharmacologic treatment. All patients received treatment with low-dose naltrexone (ranging from 0.75 mg to 6.0 mg) but were blinded to dose.3
Patients were evaluated for improvement in fibromyalgia symptoms using the Patient Global Impression of Improvement (PGI-I) scale—which ranges from 1 (very much improved) to 7 (very much worse), with 4 being “no change”—at baseline and after 2 to 3 weeks of treatment with low-dose naltrexone. A patient was considered a responder if they scored 1 to 3 on the follow-up PGI-I scale or if they experienced a > 30% pain reduction on the VAS. If a patient did not respond to their dose, the next patient began treatment at a dose 0.75 mg higher than the previous patient’s ending dose. If a patient did respond to low-dose naltrexone treatment, the next patient’s starting dose was 0.75 mg less than the previous patient’s. Eleven of 25 patients were considered responders.3
The primary outcomes were effective dose for 50% of fibromyalgia patients (3.88 mg; 95% CI, 3.39-4.35) and effective dose for 95% of fibromyalgia patients (5.4 mg; 95% CI, 4.66-6.13). Secondary outcomes were fibromyalgia symptoms as evaluated on the Fibromyalgia Impact Questionnaire Revised. Five of the 11 responders reported a > 30% improvement in tenderness and 8 of the 11 responders reported a > 30% decrease in waking unrefreshed.3
Limitations of the study include the short time period of treatment before response was assessed and the decision to use low test doses, which may have hindered detection of effective doses > 6 mg in fibromyalgia.3
Editor’s takeaway
Low-dose naltrexone, a less-often-used form of pain management, is a welcome option. Studies show some effectiveness in a variety of pain conditions with few adverse effects. The small number of studies, the small sample sizes, and the limited follow-up duration should encourage more investigation into how to best use this intervention.
1. Srinivasan A, Dutta P, Bansal D, et al. Efficacy and safety of low-dose naltrexone in painful diabetic neuropathy: a randomized, double-blind, active-control, crossover clinical trial. J Diabetes. 2021;13:770-778. doi: 10.1111/1753-0407.13202
2. Martin SJ, McAnally HB, Okediji P, et al. Low-dose naltrexone, an opioid-receptor antagonist, is a broad-spectrum analgesic: a retrospective cohort study. Pain Management. 2022;12:699-709. doi: 10.2217/pmt-2021-0122
3. Bruun-Plesner K, Blichfeldt-Eckhardt MR, Vaegter HB, et al. Low-dose naltrexone for the treatment of fibromyalgia: investigation of dose-response relationships. Pain Med. 2020;21:2253-2261. doi: 10.1093/pm/pnaa001
1. Srinivasan A, Dutta P, Bansal D, et al. Efficacy and safety of low-dose naltrexone in painful diabetic neuropathy: a randomized, double-blind, active-control, crossover clinical trial. J Diabetes. 2021;13:770-778. doi: 10.1111/1753-0407.13202
2. Martin SJ, McAnally HB, Okediji P, et al. Low-dose naltrexone, an opioid-receptor antagonist, is a broad-spectrum analgesic: a retrospective cohort study. Pain Management. 2022;12:699-709. doi: 10.2217/pmt-2021-0122
3. Bruun-Plesner K, Blichfeldt-Eckhardt MR, Vaegter HB, et al. Low-dose naltrexone for the treatment of fibromyalgia: investigation of dose-response relationships. Pain Med. 2020;21:2253-2261. doi: 10.1093/pm/pnaa001
EVIDENCE-BASED ANSWER:
YES. Low-dose naltrexone is as effective as amitriptyline in the treatment of painful diabetic neuropathy and has a superior safety profile (strength of recommendation [SOR], B; single randomized controlled trial [RCT]).
Low-dose naltrexone significantly reduced pain by 32% in inflammatory conditions and 44% in neuropathic conditions (SOR, B; single retrospective cohort study).
Doses as low as 5.4 mg were found to reduce pain in 95% of patients with fibromyalgia (SOR, B; single prospective dose-response study).
