User login
Table Salt Method Following Cryotherapy for Recurrent Pyogenic Granuloma on the Fingertip
Table Salt Method Following Cryotherapy for Recurrent Pyogenic Granuloma on the Fingertip
Practice Gap
Pyogenic granulomas (PGs) are benign endothelial tumors of the skin or mucosae that frequently become ulcerated and may cause patients substantial discomfort or distress due to rapid enlargement and bleeding.1 These lesions often manifest as solitary red papules or polyps following localized trauma or irritation. They can grow up to 1 cm over a few weeks to several months. Pyogenic granulomas can develop at any age, but they most commonly are seen in children and young adults, with a slight male predominance.1,2 The differential diagnosis for PG includes amelanotic melanoma, bacillary angiomatosis, Kaposi sarcoma, glomus tumor, infantile hemangioma, and irritated melanocytic nevus.1 Histologically, PGs are well-circumscribed exophytic or pedunculated proliferations of small capillaries that often are arranged in a lobular pattern. Early lesions show packed endothelial cells, while advanced lesions display more ectatic vessels, erosion, and crusting.3 The term pyogenic granuloma is a misnomer, as these lesions display neither an infectious etiology nor granulomatous tissue on dermatopathologic examination.4 A more accurate clinical description for this lesion is a lobular capillary hemangioma.
Numerous surgical and laser techniques have been used to treat PGs, with varying degrees of success. Treatment often consists of either shave excision followed by electrosurgery at the base or full excision with suturing under local anesthesia for patients who can tolerate anesthetic injections.1 Pulsed dye laser has been proven to be a safe alternative treatment option, particularly in children who otherwise would not tolerate surgical procedures.5 Topical beta-blockers, silver nitrate cauterization, sclerotherapy, and liquid nitrogen all have been used as alternative treatment methods.1
Pyogenic granulomas often recur after first-line treatments, and patients may hesitate to try more invasive techniques when the first choice has failed. Children may not be amenable to any of these curative techniques, as they may not tolerate the pain associated with lidocaine injection and/or have a fear of needles or surgical intervention; even adults may be reluctant to have a procedure they perceive as painful. We present a less invasive technique for treatment of recurrent PGs using table salt and cryotherapy.
The Technique
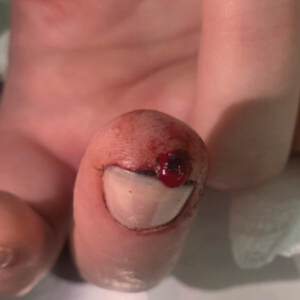
A 51-year-old woman with no notable medical history presented to the emergency department for evaluation of a black dot on the pulp of the right third fingertip of 1 week’s duration. The patient reported rapid progression to an ulcerated red nodule with associated bleeding for the past 3 days (Figure 1). Direct pressure temporarily alleviated the bleeding, but it started again upon cessation of pressure. She denied any preceding trauma to the area or any associated systemic symptoms such as fever, chills, nausea, or vomiting.
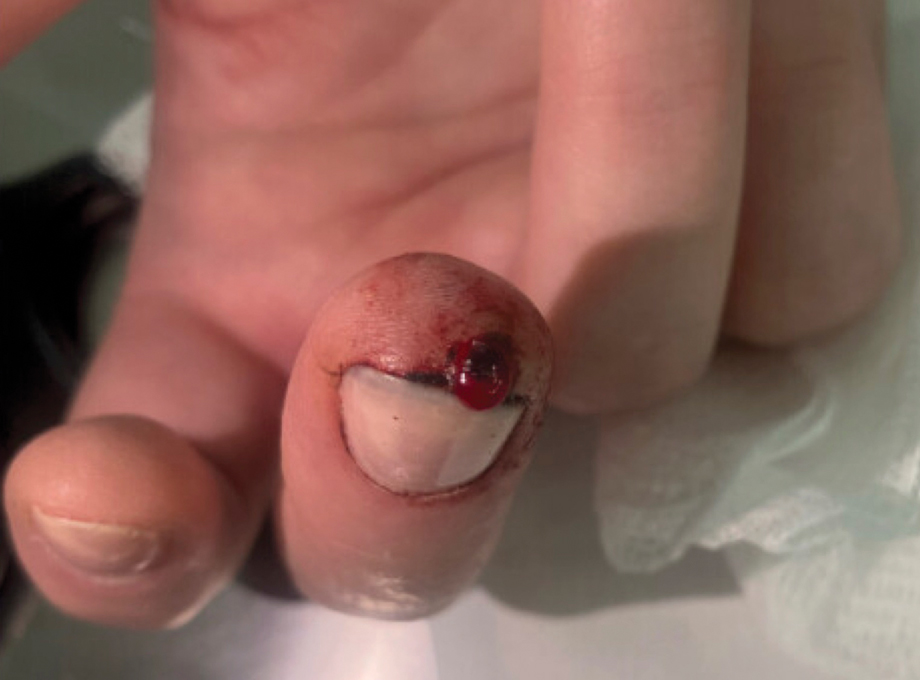
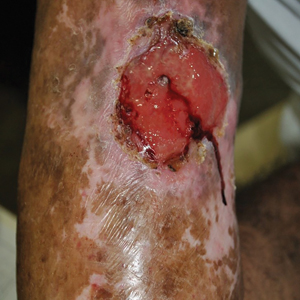
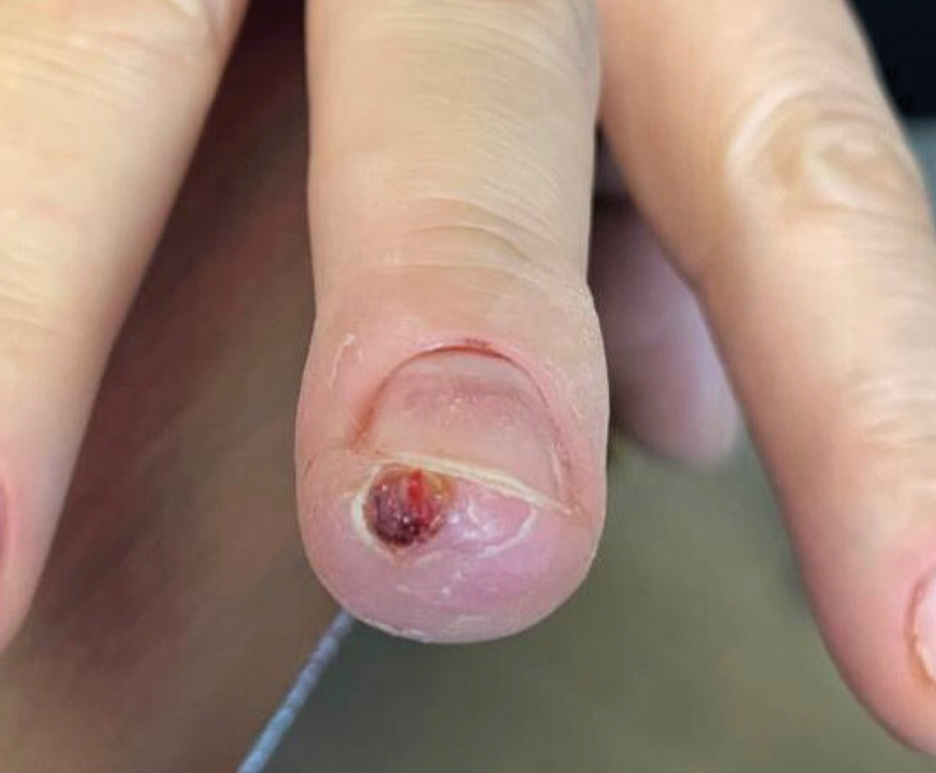
The inpatient dermatology team recommended that the patient be discharged following silver nitrate cautery, with a referral sent to outpatient dermatology; however, the patient returned to the dermatology clinic 4 days later, at which time physical examination revealed a well-circumscribed, 5-mm, bright-red, erosive papule with overlying hemorrhagic crust that was not actively bleeding, as well as fissuring of the surrounding skin. The entire lesion was removed using tangential excision followed by electrodesiccation at the base. Pathology revealed small capillaries arranged in a lobular pattern, confirming the diagnosis of PG. At a 2-week follow-up visit, the patient noted that the lesion had recurred within 24 hours after the procedure and was larger (Figure 2). At this visit, management was switched to a single treatment of cryotherapy (3 cycles for 5 seconds per cycle), and the table salt method was recommended based on a literature review for alternative nonpainful approaches for PG.6-11 We used this technique in our patient as an adjuvant to cryotherapy with the goal of reducing the need for additional painful procedures, but table salt also can be used as a standalone treatment without prior cryotherapy.
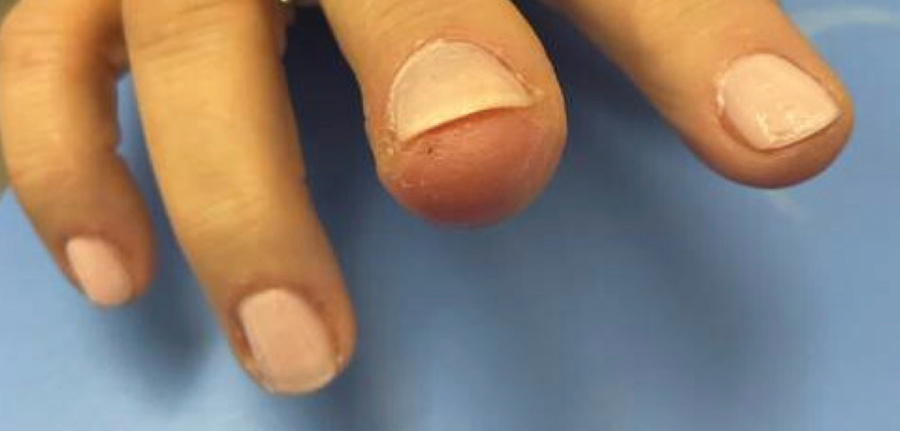
The patient was instructed to apply table salt to the lesion once daily for 2 weeks by pressing the lesion into a small amount of salt placed on a clean plate and then applying an occlusive dressing such as surgical or paper tape. She also was advised to apply petroleum jelly around the periphery of the lesion prior to salt application to protect the unaffected skin from irritation. Complete resolution of the lesion was seen when the patient followed up 2 weeks later (Figure 3). At the most recent follow-up 2 months after treatment, no further recurrence of the PG was reported.
Practice Implication
Pyogenic granulomas can be distressing for both patients and providers because they are cosmetically bothersome and prone to spontaneous bleeding. Various medical and surgical options exist to treat PGs, but there is no clear consensus on a superior modality. A 2019 study by Daruwalla and Dhurat6 highlighted a less invasive treatment option for PGs using table salt applied once daily for 2 weeks under an occlusive dressing with good outcomes and without involving other treatments such as cryotherapy. Several other case reports have endorsed this approach, adding anecdotal evidence for its utility.7-11 Topical sodium chloride may treat PGs primarily through osmotic desiccation, drawing water out of the lesion and leading to endothelial cell shrinkage and collapse of its capillary network. This hyperosmolar environment also may induce microvascular thrombosis and ischemia, promoting lesion necrosis. Additionally, repeated application creates a dry, mildly irritative surface that may suppress angiogenesis and encourage regression of the vascular proliferation.
Consider topical application of table salt for treatment of PGs in certain subsets of patients; for example, patients who are not amenable to surgery or are too young for advanced surgical techniques may be good candidates for this method, as it does not require anesthetic injections and generally is pain free. Patients with resistant or recurrent PGs that did not respond to first-line treatments such as cryotherapy, tangential excision, or electrodesiccation may be more amenable to a less invasive secondary approach.
Importantly, we used a dual-therapy approach in our patient, initially using a single application of cryotherapy followed by the table salt method once daily for 2 weeks. This imposes limitations on the generalizability of table salt as a standalone approach for treating PGs. In this case, we did not have prior practical experience using table salt for this condition and only had small reports to justify its use. As a result, we attempted a more traditional treatment initially (cryotherapy) to avoid potential delays in resolution. The clinicians recommended table salt as an adjuvant prior to seeing the cryotherapy results because this treatment was benign and offered potential additive results, and therefore waiting was not necessary. However, various other cases have reported similar success using table salt as monotherapy.6-9,11 Patients should be advised of potential mild adverse events, such as irritation to the surrounding skin. Higher-level evidence studies are required to further vet the utility of the table salt method for treatment of PGs.
- Bolognia JL, Schaffer JV, Cerroni L. Vascular neoplasms and neoplastic‑like proliferations. In: Dermatology. Elsevier; 2018.
- Harris MN, Desai R, Chuang TY, et al. Lobular capillary hemangiomas: an epidemiologic report, with emphasis on cutaneous lesions. J Am Acad Dermatol. 2000;42:1012-1016.
- Ferringer TK, DiCaudo DJ, Elston D, et al. Dermatopathology. W.B. Saunders; 2008.
- Gomes SR, Shakir QJ, Thaker PV, et al. Pyogenic granuloma of the gingiva: a misnomer? - a case report and review of literature. J Indian Soc Periodontol. 2013;17:514-519. doi:10.4103/0972-124X.118327
- Sud AR, Tan ST. Pyogenic granuloma-treatment by shave-excision and/or pulsed-dye laser. J Plast Reconstr Aesthet Surg. 2010;63:1364-1368. doi:10.1016/j.bjps.2009.06.031
- Daruwalla SB, Dhurat RS. A pinch of salt is all it takes! the novel use of table salt for the effective treatment of pyogenic granuloma. J Am Acad Dermatol. 2020;83:E107-E108. doi:10.1016/j.jaad.2019.12.013
- Alhammad G, Albaraka M, Alotaibi H, et al. The use of common salt for the treatment of pyogenic granuloma. JAAD Case Rep. 2024;53:40-42. doi:10.1016/j.jdcr.2024.08.016
- Weiss ES, Wood D. Simple, safe, and effective treatment for pyogenic granuloma. Can Fam Physician. 2023;69:479-480. doi:10.46747/cfp.6907479
- Bernales Salinas A, Toro Sepúlveda A, Meier Pincheira H, et al. Case report: pyogenic granuloma-just salt, a simple and pain-free treatment. Dermatol Ther. 2022;35:E15194. doi:10.1111/dth.15194
- Martín-Nieto González J, Rodríguez-Sánchez B, Berna-Rico E, et al. Pyogenic granuloma resolved with timolol and table salt. An Pediatr (Engl Ed). 2025;102:503706. doi:10.1016/j.anpede.2025.503706
- Bin Rubaian NF. Complete resolution of a refractory pyogenic granuloma following topical salt treatment. Open Access Emerg Med. 2021;13:445-448. doi:10.2147/OAEM.S323793
Practice Gap
Pyogenic granulomas (PGs) are benign endothelial tumors of the skin or mucosae that frequently become ulcerated and may cause patients substantial discomfort or distress due to rapid enlargement and bleeding.1 These lesions often manifest as solitary red papules or polyps following localized trauma or irritation. They can grow up to 1 cm over a few weeks to several months. Pyogenic granulomas can develop at any age, but they most commonly are seen in children and young adults, with a slight male predominance.1,2 The differential diagnosis for PG includes amelanotic melanoma, bacillary angiomatosis, Kaposi sarcoma, glomus tumor, infantile hemangioma, and irritated melanocytic nevus.1 Histologically, PGs are well-circumscribed exophytic or pedunculated proliferations of small capillaries that often are arranged in a lobular pattern. Early lesions show packed endothelial cells, while advanced lesions display more ectatic vessels, erosion, and crusting.3 The term pyogenic granuloma is a misnomer, as these lesions display neither an infectious etiology nor granulomatous tissue on dermatopathologic examination.4 A more accurate clinical description for this lesion is a lobular capillary hemangioma.
Numerous surgical and laser techniques have been used to treat PGs, with varying degrees of success. Treatment often consists of either shave excision followed by electrosurgery at the base or full excision with suturing under local anesthesia for patients who can tolerate anesthetic injections.1 Pulsed dye laser has been proven to be a safe alternative treatment option, particularly in children who otherwise would not tolerate surgical procedures.5 Topical beta-blockers, silver nitrate cauterization, sclerotherapy, and liquid nitrogen all have been used as alternative treatment methods.1
Pyogenic granulomas often recur after first-line treatments, and patients may hesitate to try more invasive techniques when the first choice has failed. Children may not be amenable to any of these curative techniques, as they may not tolerate the pain associated with lidocaine injection and/or have a fear of needles or surgical intervention; even adults may be reluctant to have a procedure they perceive as painful. We present a less invasive technique for treatment of recurrent PGs using table salt and cryotherapy.
The Technique
A 51-year-old woman with no notable medical history presented to the emergency department for evaluation of a black dot on the pulp of the right third fingertip of 1 week’s duration. The patient reported rapid progression to an ulcerated red nodule with associated bleeding for the past 3 days (Figure 1). Direct pressure temporarily alleviated the bleeding, but it started again upon cessation of pressure. She denied any preceding trauma to the area or any associated systemic symptoms such as fever, chills, nausea, or vomiting.

The inpatient dermatology team recommended that the patient be discharged following silver nitrate cautery, with a referral sent to outpatient dermatology; however, the patient returned to the dermatology clinic 4 days later, at which time physical examination revealed a well-circumscribed, 5-mm, bright-red, erosive papule with overlying hemorrhagic crust that was not actively bleeding, as well as fissuring of the surrounding skin. The entire lesion was removed using tangential excision followed by electrodesiccation at the base. Pathology revealed small capillaries arranged in a lobular pattern, confirming the diagnosis of PG. At a 2-week follow-up visit, the patient noted that the lesion had recurred within 24 hours after the procedure and was larger (Figure 2). At this visit, management was switched to a single treatment of cryotherapy (3 cycles for 5 seconds per cycle), and the table salt method was recommended based on a literature review for alternative nonpainful approaches for PG.6-11 We used this technique in our patient as an adjuvant to cryotherapy with the goal of reducing the need for additional painful procedures, but table salt also can be used as a standalone treatment without prior cryotherapy.

The patient was instructed to apply table salt to the lesion once daily for 2 weeks by pressing the lesion into a small amount of salt placed on a clean plate and then applying an occlusive dressing such as surgical or paper tape. She also was advised to apply petroleum jelly around the periphery of the lesion prior to salt application to protect the unaffected skin from irritation. Complete resolution of the lesion was seen when the patient followed up 2 weeks later (Figure 3). At the most recent follow-up 2 months after treatment, no further recurrence of the PG was reported.

Practice Implication
Pyogenic granulomas can be distressing for both patients and providers because they are cosmetically bothersome and prone to spontaneous bleeding. Various medical and surgical options exist to treat PGs, but there is no clear consensus on a superior modality. A 2019 study by Daruwalla and Dhurat6 highlighted a less invasive treatment option for PGs using table salt applied once daily for 2 weeks under an occlusive dressing with good outcomes and without involving other treatments such as cryotherapy. Several other case reports have endorsed this approach, adding anecdotal evidence for its utility.7-11 Topical sodium chloride may treat PGs primarily through osmotic desiccation, drawing water out of the lesion and leading to endothelial cell shrinkage and collapse of its capillary network. This hyperosmolar environment also may induce microvascular thrombosis and ischemia, promoting lesion necrosis. Additionally, repeated application creates a dry, mildly irritative surface that may suppress angiogenesis and encourage regression of the vascular proliferation.
Consider topical application of table salt for treatment of PGs in certain subsets of patients; for example, patients who are not amenable to surgery or are too young for advanced surgical techniques may be good candidates for this method, as it does not require anesthetic injections and generally is pain free. Patients with resistant or recurrent PGs that did not respond to first-line treatments such as cryotherapy, tangential excision, or electrodesiccation may be more amenable to a less invasive secondary approach.
Importantly, we used a dual-therapy approach in our patient, initially using a single application of cryotherapy followed by the table salt method once daily for 2 weeks. This imposes limitations on the generalizability of table salt as a standalone approach for treating PGs. In this case, we did not have prior practical experience using table salt for this condition and only had small reports to justify its use. As a result, we attempted a more traditional treatment initially (cryotherapy) to avoid potential delays in resolution. The clinicians recommended table salt as an adjuvant prior to seeing the cryotherapy results because this treatment was benign and offered potential additive results, and therefore waiting was not necessary. However, various other cases have reported similar success using table salt as monotherapy.6-9,11 Patients should be advised of potential mild adverse events, such as irritation to the surrounding skin. Higher-level evidence studies are required to further vet the utility of the table salt method for treatment of PGs.
Practice Gap
Pyogenic granulomas (PGs) are benign endothelial tumors of the skin or mucosae that frequently become ulcerated and may cause patients substantial discomfort or distress due to rapid enlargement and bleeding.1 These lesions often manifest as solitary red papules or polyps following localized trauma or irritation. They can grow up to 1 cm over a few weeks to several months. Pyogenic granulomas can develop at any age, but they most commonly are seen in children and young adults, with a slight male predominance.1,2 The differential diagnosis for PG includes amelanotic melanoma, bacillary angiomatosis, Kaposi sarcoma, glomus tumor, infantile hemangioma, and irritated melanocytic nevus.1 Histologically, PGs are well-circumscribed exophytic or pedunculated proliferations of small capillaries that often are arranged in a lobular pattern. Early lesions show packed endothelial cells, while advanced lesions display more ectatic vessels, erosion, and crusting.3 The term pyogenic granuloma is a misnomer, as these lesions display neither an infectious etiology nor granulomatous tissue on dermatopathologic examination.4 A more accurate clinical description for this lesion is a lobular capillary hemangioma.
Numerous surgical and laser techniques have been used to treat PGs, with varying degrees of success. Treatment often consists of either shave excision followed by electrosurgery at the base or full excision with suturing under local anesthesia for patients who can tolerate anesthetic injections.1 Pulsed dye laser has been proven to be a safe alternative treatment option, particularly in children who otherwise would not tolerate surgical procedures.5 Topical beta-blockers, silver nitrate cauterization, sclerotherapy, and liquid nitrogen all have been used as alternative treatment methods.1
Pyogenic granulomas often recur after first-line treatments, and patients may hesitate to try more invasive techniques when the first choice has failed. Children may not be amenable to any of these curative techniques, as they may not tolerate the pain associated with lidocaine injection and/or have a fear of needles or surgical intervention; even adults may be reluctant to have a procedure they perceive as painful. We present a less invasive technique for treatment of recurrent PGs using table salt and cryotherapy.
The Technique
A 51-year-old woman with no notable medical history presented to the emergency department for evaluation of a black dot on the pulp of the right third fingertip of 1 week’s duration. The patient reported rapid progression to an ulcerated red nodule with associated bleeding for the past 3 days (Figure 1). Direct pressure temporarily alleviated the bleeding, but it started again upon cessation of pressure. She denied any preceding trauma to the area or any associated systemic symptoms such as fever, chills, nausea, or vomiting.

The inpatient dermatology team recommended that the patient be discharged following silver nitrate cautery, with a referral sent to outpatient dermatology; however, the patient returned to the dermatology clinic 4 days later, at which time physical examination revealed a well-circumscribed, 5-mm, bright-red, erosive papule with overlying hemorrhagic crust that was not actively bleeding, as well as fissuring of the surrounding skin. The entire lesion was removed using tangential excision followed by electrodesiccation at the base. Pathology revealed small capillaries arranged in a lobular pattern, confirming the diagnosis of PG. At a 2-week follow-up visit, the patient noted that the lesion had recurred within 24 hours after the procedure and was larger (Figure 2). At this visit, management was switched to a single treatment of cryotherapy (3 cycles for 5 seconds per cycle), and the table salt method was recommended based on a literature review for alternative nonpainful approaches for PG.6-11 We used this technique in our patient as an adjuvant to cryotherapy with the goal of reducing the need for additional painful procedures, but table salt also can be used as a standalone treatment without prior cryotherapy.

The patient was instructed to apply table salt to the lesion once daily for 2 weeks by pressing the lesion into a small amount of salt placed on a clean plate and then applying an occlusive dressing such as surgical or paper tape. She also was advised to apply petroleum jelly around the periphery of the lesion prior to salt application to protect the unaffected skin from irritation. Complete resolution of the lesion was seen when the patient followed up 2 weeks later (Figure 3). At the most recent follow-up 2 months after treatment, no further recurrence of the PG was reported.

Practice Implication
Pyogenic granulomas can be distressing for both patients and providers because they are cosmetically bothersome and prone to spontaneous bleeding. Various medical and surgical options exist to treat PGs, but there is no clear consensus on a superior modality. A 2019 study by Daruwalla and Dhurat6 highlighted a less invasive treatment option for PGs using table salt applied once daily for 2 weeks under an occlusive dressing with good outcomes and without involving other treatments such as cryotherapy. Several other case reports have endorsed this approach, adding anecdotal evidence for its utility.7-11 Topical sodium chloride may treat PGs primarily through osmotic desiccation, drawing water out of the lesion and leading to endothelial cell shrinkage and collapse of its capillary network. This hyperosmolar environment also may induce microvascular thrombosis and ischemia, promoting lesion necrosis. Additionally, repeated application creates a dry, mildly irritative surface that may suppress angiogenesis and encourage regression of the vascular proliferation.
Consider topical application of table salt for treatment of PGs in certain subsets of patients; for example, patients who are not amenable to surgery or are too young for advanced surgical techniques may be good candidates for this method, as it does not require anesthetic injections and generally is pain free. Patients with resistant or recurrent PGs that did not respond to first-line treatments such as cryotherapy, tangential excision, or electrodesiccation may be more amenable to a less invasive secondary approach.
Importantly, we used a dual-therapy approach in our patient, initially using a single application of cryotherapy followed by the table salt method once daily for 2 weeks. This imposes limitations on the generalizability of table salt as a standalone approach for treating PGs. In this case, we did not have prior practical experience using table salt for this condition and only had small reports to justify its use. As a result, we attempted a more traditional treatment initially (cryotherapy) to avoid potential delays in resolution. The clinicians recommended table salt as an adjuvant prior to seeing the cryotherapy results because this treatment was benign and offered potential additive results, and therefore waiting was not necessary. However, various other cases have reported similar success using table salt as monotherapy.6-9,11 Patients should be advised of potential mild adverse events, such as irritation to the surrounding skin. Higher-level evidence studies are required to further vet the utility of the table salt method for treatment of PGs.
- Bolognia JL, Schaffer JV, Cerroni L. Vascular neoplasms and neoplastic‑like proliferations. In: Dermatology. Elsevier; 2018.
- Harris MN, Desai R, Chuang TY, et al. Lobular capillary hemangiomas: an epidemiologic report, with emphasis on cutaneous lesions. J Am Acad Dermatol. 2000;42:1012-1016.
- Ferringer TK, DiCaudo DJ, Elston D, et al. Dermatopathology. W.B. Saunders; 2008.
- Gomes SR, Shakir QJ, Thaker PV, et al. Pyogenic granuloma of the gingiva: a misnomer? - a case report and review of literature. J Indian Soc Periodontol. 2013;17:514-519. doi:10.4103/0972-124X.118327
- Sud AR, Tan ST. Pyogenic granuloma-treatment by shave-excision and/or pulsed-dye laser. J Plast Reconstr Aesthet Surg. 2010;63:1364-1368. doi:10.1016/j.bjps.2009.06.031
- Daruwalla SB, Dhurat RS. A pinch of salt is all it takes! the novel use of table salt for the effective treatment of pyogenic granuloma. J Am Acad Dermatol. 2020;83:E107-E108. doi:10.1016/j.jaad.2019.12.013
- Alhammad G, Albaraka M, Alotaibi H, et al. The use of common salt for the treatment of pyogenic granuloma. JAAD Case Rep. 2024;53:40-42. doi:10.1016/j.jdcr.2024.08.016
- Weiss ES, Wood D. Simple, safe, and effective treatment for pyogenic granuloma. Can Fam Physician. 2023;69:479-480. doi:10.46747/cfp.6907479
- Bernales Salinas A, Toro Sepúlveda A, Meier Pincheira H, et al. Case report: pyogenic granuloma-just salt, a simple and pain-free treatment. Dermatol Ther. 2022;35:E15194. doi:10.1111/dth.15194
- Martín-Nieto González J, Rodríguez-Sánchez B, Berna-Rico E, et al. Pyogenic granuloma resolved with timolol and table salt. An Pediatr (Engl Ed). 2025;102:503706. doi:10.1016/j.anpede.2025.503706
- Bin Rubaian NF. Complete resolution of a refractory pyogenic granuloma following topical salt treatment. Open Access Emerg Med. 2021;13:445-448. doi:10.2147/OAEM.S323793
- Bolognia JL, Schaffer JV, Cerroni L. Vascular neoplasms and neoplastic‑like proliferations. In: Dermatology. Elsevier; 2018.
- Harris MN, Desai R, Chuang TY, et al. Lobular capillary hemangiomas: an epidemiologic report, with emphasis on cutaneous lesions. J Am Acad Dermatol. 2000;42:1012-1016.
- Ferringer TK, DiCaudo DJ, Elston D, et al. Dermatopathology. W.B. Saunders; 2008.
- Gomes SR, Shakir QJ, Thaker PV, et al. Pyogenic granuloma of the gingiva: a misnomer? - a case report and review of literature. J Indian Soc Periodontol. 2013;17:514-519. doi:10.4103/0972-124X.118327
- Sud AR, Tan ST. Pyogenic granuloma-treatment by shave-excision and/or pulsed-dye laser. J Plast Reconstr Aesthet Surg. 2010;63:1364-1368. doi:10.1016/j.bjps.2009.06.031
- Daruwalla SB, Dhurat RS. A pinch of salt is all it takes! the novel use of table salt for the effective treatment of pyogenic granuloma. J Am Acad Dermatol. 2020;83:E107-E108. doi:10.1016/j.jaad.2019.12.013
- Alhammad G, Albaraka M, Alotaibi H, et al. The use of common salt for the treatment of pyogenic granuloma. JAAD Case Rep. 2024;53:40-42. doi:10.1016/j.jdcr.2024.08.016
- Weiss ES, Wood D. Simple, safe, and effective treatment for pyogenic granuloma. Can Fam Physician. 2023;69:479-480. doi:10.46747/cfp.6907479
- Bernales Salinas A, Toro Sepúlveda A, Meier Pincheira H, et al. Case report: pyogenic granuloma-just salt, a simple and pain-free treatment. Dermatol Ther. 2022;35:E15194. doi:10.1111/dth.15194
- Martín-Nieto González J, Rodríguez-Sánchez B, Berna-Rico E, et al. Pyogenic granuloma resolved with timolol and table salt. An Pediatr (Engl Ed). 2025;102:503706. doi:10.1016/j.anpede.2025.503706
- Bin Rubaian NF. Complete resolution of a refractory pyogenic granuloma following topical salt treatment. Open Access Emerg Med. 2021;13:445-448. doi:10.2147/OAEM.S323793
Table Salt Method Following Cryotherapy for Recurrent Pyogenic Granuloma on the Fingertip
Table Salt Method Following Cryotherapy for Recurrent Pyogenic Granuloma on the Fingertip
AAD 2026 Annual Meeting Highlights
AAD 2026 Annual Meeting Highlights
The American Academy of Dermatology’s 2026 Annual Meeting in Denver, Colorado, showcased advances in clinical practice and dermatology research. Selected key updates are summarized here for concise review of emerging dermatology data relevant to clinical practice.
AI Holds Promise in Dermatology, Issues Remain to be Addressed
Artificial intelligence (AI) is rapidly advancing in dermatology, improving image analysis, clinical decision support, and workflow efficiency; however, concerns remain about ethical use, training gaps, and potential skill loss among clinicians. While AI may enhance productivity and care, experts emphasize the need for cautious implementation, education, and ongoing evaluation of real-world performance.
Phase 2b Findings Support Novel Agent to Treat Alopecia Areata
A phase 2b trial of rezpegaldesleukin for severe alopecia areata showed considerably greater reductions in SALT scores vs placebo over 36 weeks, with higher response rates and no treatment plateau. The biologic, which enhances regulatory T-cell activity, demonstrated a favorable safety profile, with mainly mild injection-site reactions and no new safety signals.
JAK Inhibitors: Identifying Ideal Candidates and Putting Real-World Risks in Context
Emerging evidence suggests Janus kinase (JAK) inhibitors are safer in dermatology than early rheumatoid arthritis data indicated. Risks for cardiovascular events, thrombosis, and malignancy appear low and largely driven by baseline patient factors. With appropriate screening and monitoring, these agents can be used safely in most patients with inflammatory skin diseases.
Nemolizumab Phase 2 Findings Positive for Children 2-11 Years Old With Atopic Dermatitis
A phase 2 open-label study of nemolizumab in children aged 2 to 11 years with moderate to severe atopic dermatitis showed notable improvements in skin clearance, disease severity, and itch with weight-based dosing. Responses were rapid, durable through 52 weeks, and consistent with prior data, with no new safety signals identified in this population.
Melasma: A New Era of Topical Treatment Options Galore
Melasma treatment is rapidly expanding beyond traditional agents such as hydroquinone and triple combination therapy, with newer topicals including tranexamic acid, cysteamine, azelaic acid, thiamidol, and emerging compounds showing variable efficacy. While promising, evidence is still evolving, and combination regimens plus strict photoprotection remain the cornerstone of management.
Weight-Loss Drug–Biologic Combination Boosts Relief in Psoriatic Arthritis
In a phase 3b trial, combining tirzepatide with ixekizumab significantly improved joint and skin outcomes in patients with psoriatic arthritis and overweight/obesity (P<.05) compared with ixekizumab alone (P<.001). The combination yielded higher American College of Rheumatology and Psoriasis Area and Severity Index response rates, early symptom improvement, and meaningful weight loss, with safety profiles consistent with known effects.
Tips on Using Biologics for Psoriasis in Context of HIV
Evidence for biologic use in HIV-positive patients with moderate to severe psoriasis is limited, but available case reports suggest tumor necrosis factor inhibitors and newer IL-targeted biologics are generally effective without major impacts on viral load or CD4 counts. Experts recommend prioritizing nonimmunosuppressive options and coordinating care with HIV specialists due to potential infection risks.
Upadacitinib Results in Significant Improvements in Nonsegmental Vitiligo in Phase 3 Studies
Two phase 3 trials showed that the Janus kinase 1 inhibitor upadacitinib significantly improved repigmentation outcomes in adolescents and adults with nonsegmental vitiligo vs placebo over 48 weeks (P<.0001 for both), with a higher proportion achieving clinically meaningful reductions in Vitiligo Area and Severity Index scores. Benefits increased over time without plateau, and no new safety signals were identified.
The American Academy of Dermatology’s 2026 Annual Meeting in Denver, Colorado, showcased advances in clinical practice and dermatology research. Selected key updates are summarized here for concise review of emerging dermatology data relevant to clinical practice.
AI Holds Promise in Dermatology, Issues Remain to be Addressed
Artificial intelligence (AI) is rapidly advancing in dermatology, improving image analysis, clinical decision support, and workflow efficiency; however, concerns remain about ethical use, training gaps, and potential skill loss among clinicians. While AI may enhance productivity and care, experts emphasize the need for cautious implementation, education, and ongoing evaluation of real-world performance.
Phase 2b Findings Support Novel Agent to Treat Alopecia Areata
A phase 2b trial of rezpegaldesleukin for severe alopecia areata showed considerably greater reductions in SALT scores vs placebo over 36 weeks, with higher response rates and no treatment plateau. The biologic, which enhances regulatory T-cell activity, demonstrated a favorable safety profile, with mainly mild injection-site reactions and no new safety signals.
JAK Inhibitors: Identifying Ideal Candidates and Putting Real-World Risks in Context
Emerging evidence suggests Janus kinase (JAK) inhibitors are safer in dermatology than early rheumatoid arthritis data indicated. Risks for cardiovascular events, thrombosis, and malignancy appear low and largely driven by baseline patient factors. With appropriate screening and monitoring, these agents can be used safely in most patients with inflammatory skin diseases.
Nemolizumab Phase 2 Findings Positive for Children 2-11 Years Old With Atopic Dermatitis
A phase 2 open-label study of nemolizumab in children aged 2 to 11 years with moderate to severe atopic dermatitis showed notable improvements in skin clearance, disease severity, and itch with weight-based dosing. Responses were rapid, durable through 52 weeks, and consistent with prior data, with no new safety signals identified in this population.
Melasma: A New Era of Topical Treatment Options Galore
Melasma treatment is rapidly expanding beyond traditional agents such as hydroquinone and triple combination therapy, with newer topicals including tranexamic acid, cysteamine, azelaic acid, thiamidol, and emerging compounds showing variable efficacy. While promising, evidence is still evolving, and combination regimens plus strict photoprotection remain the cornerstone of management.
Weight-Loss Drug–Biologic Combination Boosts Relief in Psoriatic Arthritis
In a phase 3b trial, combining tirzepatide with ixekizumab significantly improved joint and skin outcomes in patients with psoriatic arthritis and overweight/obesity (P<.05) compared with ixekizumab alone (P<.001). The combination yielded higher American College of Rheumatology and Psoriasis Area and Severity Index response rates, early symptom improvement, and meaningful weight loss, with safety profiles consistent with known effects.
Tips on Using Biologics for Psoriasis in Context of HIV
Evidence for biologic use in HIV-positive patients with moderate to severe psoriasis is limited, but available case reports suggest tumor necrosis factor inhibitors and newer IL-targeted biologics are generally effective without major impacts on viral load or CD4 counts. Experts recommend prioritizing nonimmunosuppressive options and coordinating care with HIV specialists due to potential infection risks.
Upadacitinib Results in Significant Improvements in Nonsegmental Vitiligo in Phase 3 Studies
Two phase 3 trials showed that the Janus kinase 1 inhibitor upadacitinib significantly improved repigmentation outcomes in adolescents and adults with nonsegmental vitiligo vs placebo over 48 weeks (P<.0001 for both), with a higher proportion achieving clinically meaningful reductions in Vitiligo Area and Severity Index scores. Benefits increased over time without plateau, and no new safety signals were identified.
The American Academy of Dermatology’s 2026 Annual Meeting in Denver, Colorado, showcased advances in clinical practice and dermatology research. Selected key updates are summarized here for concise review of emerging dermatology data relevant to clinical practice.
AI Holds Promise in Dermatology, Issues Remain to be Addressed
Artificial intelligence (AI) is rapidly advancing in dermatology, improving image analysis, clinical decision support, and workflow efficiency; however, concerns remain about ethical use, training gaps, and potential skill loss among clinicians. While AI may enhance productivity and care, experts emphasize the need for cautious implementation, education, and ongoing evaluation of real-world performance.
Phase 2b Findings Support Novel Agent to Treat Alopecia Areata
A phase 2b trial of rezpegaldesleukin for severe alopecia areata showed considerably greater reductions in SALT scores vs placebo over 36 weeks, with higher response rates and no treatment plateau. The biologic, which enhances regulatory T-cell activity, demonstrated a favorable safety profile, with mainly mild injection-site reactions and no new safety signals.
JAK Inhibitors: Identifying Ideal Candidates and Putting Real-World Risks in Context
Emerging evidence suggests Janus kinase (JAK) inhibitors are safer in dermatology than early rheumatoid arthritis data indicated. Risks for cardiovascular events, thrombosis, and malignancy appear low and largely driven by baseline patient factors. With appropriate screening and monitoring, these agents can be used safely in most patients with inflammatory skin diseases.
Nemolizumab Phase 2 Findings Positive for Children 2-11 Years Old With Atopic Dermatitis
A phase 2 open-label study of nemolizumab in children aged 2 to 11 years with moderate to severe atopic dermatitis showed notable improvements in skin clearance, disease severity, and itch with weight-based dosing. Responses were rapid, durable through 52 weeks, and consistent with prior data, with no new safety signals identified in this population.
Melasma: A New Era of Topical Treatment Options Galore
Melasma treatment is rapidly expanding beyond traditional agents such as hydroquinone and triple combination therapy, with newer topicals including tranexamic acid, cysteamine, azelaic acid, thiamidol, and emerging compounds showing variable efficacy. While promising, evidence is still evolving, and combination regimens plus strict photoprotection remain the cornerstone of management.
Weight-Loss Drug–Biologic Combination Boosts Relief in Psoriatic Arthritis
In a phase 3b trial, combining tirzepatide with ixekizumab significantly improved joint and skin outcomes in patients with psoriatic arthritis and overweight/obesity (P<.05) compared with ixekizumab alone (P<.001). The combination yielded higher American College of Rheumatology and Psoriasis Area and Severity Index response rates, early symptom improvement, and meaningful weight loss, with safety profiles consistent with known effects.
Tips on Using Biologics for Psoriasis in Context of HIV
Evidence for biologic use in HIV-positive patients with moderate to severe psoriasis is limited, but available case reports suggest tumor necrosis factor inhibitors and newer IL-targeted biologics are generally effective without major impacts on viral load or CD4 counts. Experts recommend prioritizing nonimmunosuppressive options and coordinating care with HIV specialists due to potential infection risks.
Upadacitinib Results in Significant Improvements in Nonsegmental Vitiligo in Phase 3 Studies
Two phase 3 trials showed that the Janus kinase 1 inhibitor upadacitinib significantly improved repigmentation outcomes in adolescents and adults with nonsegmental vitiligo vs placebo over 48 weeks (P<.0001 for both), with a higher proportion achieving clinically meaningful reductions in Vitiligo Area and Severity Index scores. Benefits increased over time without plateau, and no new safety signals were identified.
AAD 2026 Annual Meeting Highlights
AAD 2026 Annual Meeting Highlights
Evaluating Drug Eruptions Using AI: Tips From Alina G. Bridges, DO
Evaluating Drug Eruptions Using AI: Tips From Alina G. Bridges, DO
How might AI enhance the detection of key histologic features in drug eruptions compared to traditional microscopy?
DR. BRIDGES: AI offers the potential to enhance detection of histologic features in drug eruptions by systematically analyzing entire whole-slide images. Convolutional neural networks and attention-based models can identify subtle or focal findings such as scattered dyskeratotic keratinocytes, focal spongiosis, early interface change, rare eosinophils, or microvascular injury, which may be overlooked during routine microscopy due to sampling limitations. This capability is particularly relevant in drug eruptions, where histologic changes often are heterogeneous and patchy.
AI-generated attention heatmaps can highlight diagnostically relevant regions across the slide, improving consistency and completeness of slide reviews. While AI has demonstrated high sensitivity and specificity in broader dermatopathology tasks, particularly neoplastic conditions, drug eruption–specific validation data are currently lacking. As such, the most realistic application at present is AI functioning as a sensitivity-enhancing adjunct or “second reader,” improving consistency and completeness of slide review while preserving expert human interpretation.
Which histologic patterns in drug eruptions are hardest to quantify, and how could AI help standardize their assessment?
DR. BRIDGES: AI-based image analysis can standardize the assessment of histologic patterns through objective reproducible quantification. Deep learning algorithms can segment epidermal and dermal compartments, identify inflammatory cell types, and calculate metrics such as eosinophil density per unit area, percentage of epidermis with vacuolar alteration, or number of affected vessels. Studies in quantitative immunohistochemistry demonstrate high accuracy for tissue segmentation and cell counting, suggesting feasibility for similar applications in inflammatory dermatopathology. While these tools would not replace diagnostic interpretation, they could provide standardized measurements that enhance reproducibility and improve clinicopathologic correlation.
What training challenges must be addressed in AI and drug eruption histology?
DR. BRIDGES: Training AI models for drug eruption histopathology faces several challenges, including the limited availability of high-quality, well-annotated datasets, as most existing AI dermatopathology research focuses on neoplastic conditions. Drug eruptions also exhibit marked histologic heterogeneity, ranging from spongiotic and lichenoid to vasculitic and cytotoxic patterns, often with significant overlap. Accurate labeling, therefore, requires robust clinicopathologic correlation, including medication history, timing, laboratory data, and clinical outcomes—information that is often incomplete or retrospective.
Inaccurate or inconsistent annotations can significantly degrade model performance, and expert disagreement in borderline cases further complicates the creation of reliable ground truth. Additionally, training data may reflect institutional or demographic biases, risking unequal performance across patient populations. Addressing these challenges will require multicenter collaboration, standardized annotation protocols, inclusion of diverse patient cohorts, and careful attention to bias mitigation. At present, these barriers place drug eruption AI firmly in the investigational rather than clinical domain.
How important is AI explainability in the interpretation of diagnostic suggestions?
DR. BRIDGES: Explainability is essential for trust, particularly in the evaluation of drug eruptions, where diagnostic decisions can have serious clinical consequences. Dermatopathologists must understand which histologic features are driving an AI model’s assessment to ensure that conclusions align with morphologic reality and clinicopathologic reasoning. Explainable AI tools (such as attention heatmaps, feature importance rankings, and methods like Shapley Additive Explanations or Local Interpretable Model-Agnostic Explanations) can help clarify which histologic features are driving the AI model’s assessment.
Without transparency, AI systems function as “black boxes,” limiting their utility in high-stakes settings where diagnostic accountability and clinical communication are paramount. Explainability also supports appropriate skepticism, allowing pathologists to recognize when model outputs may be unreliable due to artifacts, atypical patterns, or out-of-distribution cases. In cases of drug eruptions—where diagnosis relies on combining histology, clinical timing, and medication history—explainability is essential for proper use.
How could AI pattern recognition be integrated into your workflow to enhance diagnostic efficiency and accuracy? What safeguards would be required?
DR. BRIDGES: In the near term, AI pattern recognition can be useful as an assistive tool rather than a diagnostic authority. One potential application is pre-screening whole-slide images to flag cases with features such as prominent interface change, increased keratinocyte necrosis, eosinophil-rich infiltrates, or vascular injury, prompting expedited review in clinically concerning scenarios. During sign-out, AI overlays could aid efficiency by highlighting rare but relevant features and providing quantitative summaries that support standardized reporting.
Safeguards are essential. AI systems must be validated across diverse practice settings, staining protocols, and scanning platforms. Human oversight is mandatory, with the dermatopathologist retaining full diagnostic responsibility. AI involvement should be clearly documented for medicolegal transparency, and performance should be continuously monitored to detect algorithmic drift as new drug eruption patterns emerge. Given current limitations, AI is best viewed as a tool to refine and support expert judgment, not replace it.
What data-sharing or privacy challenges must be addressed to develop robust AI models for diverse drug-eruption histopathology?
DR. BRIDGES: Developing robust AI models for drug eruptions requires large diverse datasets, raising significant privacy and governance challenges. Rigorous de-identification protocols, clear informed consent frameworks, and strong institutional oversight are therefore essential. Multicenter collaborations must employ secure data-use agreements and governance structures that clearly define access, ownership, and downstream use of data.
Ensuring equitable representation is equally critical, as underrepresentation of certain populations may lead to biased performance and disparities in care. Standardized data formats and interoperable systems are needed to facilitate collaboration while preserving security. Transparent governance structures, clear rules regarding data use, and trust-building with patients and institutions will ultimately determine willingness to participate. Addressing these challenges is foundational to advancing AI research in drug eruptions responsibly and ethically.
How might AI enhance the detection of key histologic features in drug eruptions compared to traditional microscopy?
DR. BRIDGES: AI offers the potential to enhance detection of histologic features in drug eruptions by systematically analyzing entire whole-slide images. Convolutional neural networks and attention-based models can identify subtle or focal findings such as scattered dyskeratotic keratinocytes, focal spongiosis, early interface change, rare eosinophils, or microvascular injury, which may be overlooked during routine microscopy due to sampling limitations. This capability is particularly relevant in drug eruptions, where histologic changes often are heterogeneous and patchy.
AI-generated attention heatmaps can highlight diagnostically relevant regions across the slide, improving consistency and completeness of slide reviews. While AI has demonstrated high sensitivity and specificity in broader dermatopathology tasks, particularly neoplastic conditions, drug eruption–specific validation data are currently lacking. As such, the most realistic application at present is AI functioning as a sensitivity-enhancing adjunct or “second reader,” improving consistency and completeness of slide review while preserving expert human interpretation.
Which histologic patterns in drug eruptions are hardest to quantify, and how could AI help standardize their assessment?
DR. BRIDGES: AI-based image analysis can standardize the assessment of histologic patterns through objective reproducible quantification. Deep learning algorithms can segment epidermal and dermal compartments, identify inflammatory cell types, and calculate metrics such as eosinophil density per unit area, percentage of epidermis with vacuolar alteration, or number of affected vessels. Studies in quantitative immunohistochemistry demonstrate high accuracy for tissue segmentation and cell counting, suggesting feasibility for similar applications in inflammatory dermatopathology. While these tools would not replace diagnostic interpretation, they could provide standardized measurements that enhance reproducibility and improve clinicopathologic correlation.
What training challenges must be addressed in AI and drug eruption histology?
DR. BRIDGES: Training AI models for drug eruption histopathology faces several challenges, including the limited availability of high-quality, well-annotated datasets, as most existing AI dermatopathology research focuses on neoplastic conditions. Drug eruptions also exhibit marked histologic heterogeneity, ranging from spongiotic and lichenoid to vasculitic and cytotoxic patterns, often with significant overlap. Accurate labeling, therefore, requires robust clinicopathologic correlation, including medication history, timing, laboratory data, and clinical outcomes—information that is often incomplete or retrospective.
Inaccurate or inconsistent annotations can significantly degrade model performance, and expert disagreement in borderline cases further complicates the creation of reliable ground truth. Additionally, training data may reflect institutional or demographic biases, risking unequal performance across patient populations. Addressing these challenges will require multicenter collaboration, standardized annotation protocols, inclusion of diverse patient cohorts, and careful attention to bias mitigation. At present, these barriers place drug eruption AI firmly in the investigational rather than clinical domain.
How important is AI explainability in the interpretation of diagnostic suggestions?
DR. BRIDGES: Explainability is essential for trust, particularly in the evaluation of drug eruptions, where diagnostic decisions can have serious clinical consequences. Dermatopathologists must understand which histologic features are driving an AI model’s assessment to ensure that conclusions align with morphologic reality and clinicopathologic reasoning. Explainable AI tools (such as attention heatmaps, feature importance rankings, and methods like Shapley Additive Explanations or Local Interpretable Model-Agnostic Explanations) can help clarify which histologic features are driving the AI model’s assessment.
Without transparency, AI systems function as “black boxes,” limiting their utility in high-stakes settings where diagnostic accountability and clinical communication are paramount. Explainability also supports appropriate skepticism, allowing pathologists to recognize when model outputs may be unreliable due to artifacts, atypical patterns, or out-of-distribution cases. In cases of drug eruptions—where diagnosis relies on combining histology, clinical timing, and medication history—explainability is essential for proper use.
How could AI pattern recognition be integrated into your workflow to enhance diagnostic efficiency and accuracy? What safeguards would be required?
DR. BRIDGES: In the near term, AI pattern recognition can be useful as an assistive tool rather than a diagnostic authority. One potential application is pre-screening whole-slide images to flag cases with features such as prominent interface change, increased keratinocyte necrosis, eosinophil-rich infiltrates, or vascular injury, prompting expedited review in clinically concerning scenarios. During sign-out, AI overlays could aid efficiency by highlighting rare but relevant features and providing quantitative summaries that support standardized reporting.
Safeguards are essential. AI systems must be validated across diverse practice settings, staining protocols, and scanning platforms. Human oversight is mandatory, with the dermatopathologist retaining full diagnostic responsibility. AI involvement should be clearly documented for medicolegal transparency, and performance should be continuously monitored to detect algorithmic drift as new drug eruption patterns emerge. Given current limitations, AI is best viewed as a tool to refine and support expert judgment, not replace it.
What data-sharing or privacy challenges must be addressed to develop robust AI models for diverse drug-eruption histopathology?
DR. BRIDGES: Developing robust AI models for drug eruptions requires large diverse datasets, raising significant privacy and governance challenges. Rigorous de-identification protocols, clear informed consent frameworks, and strong institutional oversight are therefore essential. Multicenter collaborations must employ secure data-use agreements and governance structures that clearly define access, ownership, and downstream use of data.
Ensuring equitable representation is equally critical, as underrepresentation of certain populations may lead to biased performance and disparities in care. Standardized data formats and interoperable systems are needed to facilitate collaboration while preserving security. Transparent governance structures, clear rules regarding data use, and trust-building with patients and institutions will ultimately determine willingness to participate. Addressing these challenges is foundational to advancing AI research in drug eruptions responsibly and ethically.
How might AI enhance the detection of key histologic features in drug eruptions compared to traditional microscopy?
DR. BRIDGES: AI offers the potential to enhance detection of histologic features in drug eruptions by systematically analyzing entire whole-slide images. Convolutional neural networks and attention-based models can identify subtle or focal findings such as scattered dyskeratotic keratinocytes, focal spongiosis, early interface change, rare eosinophils, or microvascular injury, which may be overlooked during routine microscopy due to sampling limitations. This capability is particularly relevant in drug eruptions, where histologic changes often are heterogeneous and patchy.
AI-generated attention heatmaps can highlight diagnostically relevant regions across the slide, improving consistency and completeness of slide reviews. While AI has demonstrated high sensitivity and specificity in broader dermatopathology tasks, particularly neoplastic conditions, drug eruption–specific validation data are currently lacking. As such, the most realistic application at present is AI functioning as a sensitivity-enhancing adjunct or “second reader,” improving consistency and completeness of slide review while preserving expert human interpretation.
Which histologic patterns in drug eruptions are hardest to quantify, and how could AI help standardize their assessment?
DR. BRIDGES: AI-based image analysis can standardize the assessment of histologic patterns through objective reproducible quantification. Deep learning algorithms can segment epidermal and dermal compartments, identify inflammatory cell types, and calculate metrics such as eosinophil density per unit area, percentage of epidermis with vacuolar alteration, or number of affected vessels. Studies in quantitative immunohistochemistry demonstrate high accuracy for tissue segmentation and cell counting, suggesting feasibility for similar applications in inflammatory dermatopathology. While these tools would not replace diagnostic interpretation, they could provide standardized measurements that enhance reproducibility and improve clinicopathologic correlation.
What training challenges must be addressed in AI and drug eruption histology?
DR. BRIDGES: Training AI models for drug eruption histopathology faces several challenges, including the limited availability of high-quality, well-annotated datasets, as most existing AI dermatopathology research focuses on neoplastic conditions. Drug eruptions also exhibit marked histologic heterogeneity, ranging from spongiotic and lichenoid to vasculitic and cytotoxic patterns, often with significant overlap. Accurate labeling, therefore, requires robust clinicopathologic correlation, including medication history, timing, laboratory data, and clinical outcomes—information that is often incomplete or retrospective.
Inaccurate or inconsistent annotations can significantly degrade model performance, and expert disagreement in borderline cases further complicates the creation of reliable ground truth. Additionally, training data may reflect institutional or demographic biases, risking unequal performance across patient populations. Addressing these challenges will require multicenter collaboration, standardized annotation protocols, inclusion of diverse patient cohorts, and careful attention to bias mitigation. At present, these barriers place drug eruption AI firmly in the investigational rather than clinical domain.
How important is AI explainability in the interpretation of diagnostic suggestions?
DR. BRIDGES: Explainability is essential for trust, particularly in the evaluation of drug eruptions, where diagnostic decisions can have serious clinical consequences. Dermatopathologists must understand which histologic features are driving an AI model’s assessment to ensure that conclusions align with morphologic reality and clinicopathologic reasoning. Explainable AI tools (such as attention heatmaps, feature importance rankings, and methods like Shapley Additive Explanations or Local Interpretable Model-Agnostic Explanations) can help clarify which histologic features are driving the AI model’s assessment.
Without transparency, AI systems function as “black boxes,” limiting their utility in high-stakes settings where diagnostic accountability and clinical communication are paramount. Explainability also supports appropriate skepticism, allowing pathologists to recognize when model outputs may be unreliable due to artifacts, atypical patterns, or out-of-distribution cases. In cases of drug eruptions—where diagnosis relies on combining histology, clinical timing, and medication history—explainability is essential for proper use.
How could AI pattern recognition be integrated into your workflow to enhance diagnostic efficiency and accuracy? What safeguards would be required?
DR. BRIDGES: In the near term, AI pattern recognition can be useful as an assistive tool rather than a diagnostic authority. One potential application is pre-screening whole-slide images to flag cases with features such as prominent interface change, increased keratinocyte necrosis, eosinophil-rich infiltrates, or vascular injury, prompting expedited review in clinically concerning scenarios. During sign-out, AI overlays could aid efficiency by highlighting rare but relevant features and providing quantitative summaries that support standardized reporting.
Safeguards are essential. AI systems must be validated across diverse practice settings, staining protocols, and scanning platforms. Human oversight is mandatory, with the dermatopathologist retaining full diagnostic responsibility. AI involvement should be clearly documented for medicolegal transparency, and performance should be continuously monitored to detect algorithmic drift as new drug eruption patterns emerge. Given current limitations, AI is best viewed as a tool to refine and support expert judgment, not replace it.
What data-sharing or privacy challenges must be addressed to develop robust AI models for diverse drug-eruption histopathology?
DR. BRIDGES: Developing robust AI models for drug eruptions requires large diverse datasets, raising significant privacy and governance challenges. Rigorous de-identification protocols, clear informed consent frameworks, and strong institutional oversight are therefore essential. Multicenter collaborations must employ secure data-use agreements and governance structures that clearly define access, ownership, and downstream use of data.
Ensuring equitable representation is equally critical, as underrepresentation of certain populations may lead to biased performance and disparities in care. Standardized data formats and interoperable systems are needed to facilitate collaboration while preserving security. Transparent governance structures, clear rules regarding data use, and trust-building with patients and institutions will ultimately determine willingness to participate. Addressing these challenges is foundational to advancing AI research in drug eruptions responsibly and ethically.
Evaluating Drug Eruptions Using AI: Tips From Alina G. Bridges, DO
Evaluating Drug Eruptions Using AI: Tips From Alina G. Bridges, DO
Sunscreen in 2026: Doping, Regulatory Changes, and Impact on Vitamin D Levels
Sunscreen in 2026: Doping, Regulatory Changes, and Impact on Vitamin D Levels
Sunscreen continues to be the foundation of successful skin cancer prevention. Daily sunscreen application and reapplication are recommended to prevent all types of skin cancer, with the strongest body of evidence supporting prevention of squamous cell carcinoma.1 Sunscreens have been used safely for decades; since my last update in 2024,2 no evidence of harm has emerged, despite the fact that organic (chemical) sunscreen filters are absorbed systemically.3
So, what’s happening with sunscreen in 2026? Let’s review some hot news and fresh controversies.
Sunscreen Doping Is Pervasive
Sunscreen “doping” signifies the sneaky addition of UV filters that have not been approved by the US Food and Drug Administration (FDA) into sunscreens under the guise of inactive ingredients. Why would a manufacturer do such a thing? To enhance a sunscreen’s UV absorption without having to increase the concentration of zinc oxide/titanium dioxide (which creates an undesirable white cast) or exceeding the maximum permitted concentration of chemical active ingredients.4,5 In a 2025 analysis of the top 150 sunscreens sold on Amazon, 48.3% contained these covert UV filters, including almost half of those marketed as mineral-only products.6 The most prevalent doping ingredient was butyloctyl salicylate, which is chemically and functionally related to the FDA-approved chemical UV filter octisalate (ethylhexyl salicylate).5
The practice of sunscreen doping is deceptive. Can a product be accurately marketed as mineral sunscreen if it contains ingredients that function as chemical UV filters but are not classified as active ingredients by the FDA? The bigger picture is that sunscreen doping is a symptom of regulatory malaise specific to the United States. Regulation of sunscreens as over-the-counter drugs plus the FDA’s stringent requirements for UV filters to be generally recognized as safe and effective (GRASE) have stymied the approval process to the extent that no new active ingredients have been approved since 1999.2 The FDA allows 16 active ingredients compared to about 30 in Europe and Asia—not for lack of safety evaluations prior to approval in those regions.7 In the United States, getting a new active sunscreen ingredient approved is far more onerous and costly than the streamlined processes that are in place abroad. This restricts sunscreen innovation; in particular, the US market lacks the wide variety of international options for protection against long-wave UVA radiation, remaining limited to just avobenzone and zinc oxide. Since long-wave UVA plays a major role in photoaging, this represents a gap in protection compared to international sunscreen offerings.1,7 Due to domestic sunscreen limitations, some Americans have turned to purchasing non–FDA-approved sunscreens abroad or through online channels.8
New Sunscreen Filter Pending Approval, and Hope for Regulatory Changes
Let’s move on to a more positive development. A new sunscreen filter is actually nearing approval in the United States! Bemotrizinol, also known as bis-ethylhexyloxyphenol methoxyphenyl triazine, is a broad-spectrum chemical UVA/UVB blocker that would represent the first new active ingredient to become available in the United States since 1999.9 It satisfies the FDA requirement for minimal systemic absorption and GRASE status and has been used with a clean safety record since 2000 in Europe.10 The icing on top is that bemotrizinol seems to be minimally allergenic, with only a few published reports of contact dermatitis over several decades of use.11,12
Yes, as I write, the FDA is on the cusp of approving bemotrizinol, a great broad-spectrum sunscreen ingredient, to one day be added to the products on our shelves. The cynic in me can’t help but point out that it took more than 20 years of effort and an estimated $20 million to get us to this point of near-approval of one new sunscreen filter.13
Perhaps things won’t be so difficult in the future. In late 2025, the bipartisan Supporting Accessible, Flexible, and Effective (SAFE) Sunscreen Standards Act was signed into law.14 The SAFE Sunscreen Standards Act calls on the FDA to be more flexible and allow for the use of real-world evidence and observational studies to demonstrate safety and effectiveness of active ingredients used in sunscreens. We can only hope that real change is forthcoming and that future sunscreen approvals won’t require decades of work and millions of dollars, as in the case of bemotrizinol.
Daily Sunscreen Use Linked to Reduction in Vitamin D Levels
The UVB wavelengths that cause sunburn overlap with those that initiate vitamin D production in the skin, generating concerns about sunscreen use reducing vitamin D levels. Nevertheless, in 2019, expert opinion and a systematic literature review determined that routine use of sunscreen was unlikely to be associated with a reduction in vitamin D levels.15,16 However, a major limitation at that time was a lack of studies examining vitamin D status in individuals using high–sun protection factor (SPF) sunscreens.
Now we have results from the first field study assessing the impact of long-term daily application of higher SPF sunscreen on vitamin D levels. In the Australian Sun-D Trial, Tran et al17 randomly assigned 639 participants to either an intervention group (routine application of SPF 50+ sunscreen on days forecasted to have a UV index ≥3) or a control group (discretionary sunscreen use). Vitamin D levels were measured at baseline in the winter/spring, at the end of summer, and then at the end of the following winter. At the end of summer, vitamin D levels increased in both groups but less in the intervention group, then decreased similarly in both groups by winter. Routine sunscreen application was associated with a decrease of 5.2 nmol/L (2.1 ng/mL) in vitamin D levels, which the authors rightfully considered to be modest. Additionally, vitamin D deficiency (defined as <50 nmol/L [<20 ng/mL]) was detected in more of the intervention group compared to the control group (45.7% vs 36.9%). The study reasonably concluded that sunscreen continues to be essential in preventing skin cancers but regular users may require vitamin D testing and/or supplementation.17
Looking Ahead
In this update, I discussed several important pieces of sunscreen news. If you check your favorite mineral sunscreen’s ingredients list, odds are you will find it also contains inactive doping ingredients shown to secretly enhance UV protection. Perhaps manufacturers won’t have to dope sunscreens in the United States forever if regulatory reforms facilitate the approval of active ingredients such as bemotrizinol used safely in other countries without huge investments of time and money. For daily sunscreen users, consider checking and/or empirically supplementing vitamin D.
None of this should discourage us from recommending regular consistent sunscreen application and reapplication to our patients. There continues to be a lack of evidence of harms associated with systemic absorption of chemical UV filters in humans, and sunscreen will continue to function as an indispensable component of skin cancer prevention for the foreseeable future.
- Abdel Azim S, Bainvoll L, Vecerek N, et al. Sunscreens part 1: mechanisms and efficacy. J Am Acad Dermatol. 2025;92:677-686.
- Adler BL. Sunscreen safety: 2024 updates. Cutis. 2024;113:195-196.
- Abdel Azim S, Bainvoll L, Vecerek N, et al. Sunscreens part 2: regulation and safety. J Am Acad Dermatol. 2025;92:689-698.
- Gawey L, Gunnell L, Shi VY. Sunscreen doping: a misnomer. J Invest Dermatol. 2025;145:2941-2942.
- Moradi Tuchayi S, Wang Z, Yan J, et al. Sunscreens: misconceptions and misinformation. J Invest Dermatol. 2023;143:1406-1411.
- Gawey L, Gunnell L, Joshi A, et al. Hidden UV filters: an analysis of the 150 most popular sunscreens. J Am Acad Dermatol. 2025;93:1594-1596.
- Pantelic MN, Wong N, Kwa M, et al. Ultraviolet filters in the United States and European Union: a review of safety and implications for the future of US sunscreens. J Am Acad Dermatol. 2023;88:632-646.
- Scaturro M. Other countries have better sunscreens. here’s why we can’t get them in the U.S. NPR. May 18, 2024. Accessed April 9, 2026. https://www.npr.org/sections/health-shots/2024/05/18/1251919831/sunscreen-effective-better-ingredients-fda
- US Food & Drug Administration. FDA proposes expanding sunscreen active ingredient list [news release]. December 11, 2025. Accessed February 24, 2026. https://www.fda.gov/news-events/press-announcements/fda-proposes-expanding-sunscreen-active-ingredient-list
- D’Ruiz CD, Plautz JR, Schuetz R, et al. Preliminary clinical pharmacokinetic evaluation of bemotrizinol—a new sunscreen active ingredient being considered for inclusion under FDA’s over-the-counter (OTC) sunscreen monograph. Regul Toxicol Pharmacol. 2023;139:105344.
- Badaoui A. Allergic contact dermatitis to bis-ethylhexyloxyphenol methoxyphenyl triazine (Tinosorb S) in a sunscreen: an emerging allergen? Contact Dermatitis. 2024;91:443-445.
- European Multicentre Photopatch Test Study (EMCPPTS) Taskforce. A European multicentre photopatch test study. Br J Dermatol. 2012;166:1002-1009.
- Nesvig K. The FDA finally proposed the approval of a new sunscreen filter. December 11, 2025. Accessed February 22, 2026. https://www.allure.com/story/fda-proposes-bemotrizinol-suncreen-filter-approval
- SAFE Sunscreen Standards Act, HR 3686, 119th Congress (2025-2026). Accessed February 23, 2026. https://www.congress.gov/bill/119th-congress/house-bill/3686/text
- Neale RE, Khan SR, Lucas RM, et al. The effect of sunscreen on vitamin D: a review. Br J Dermatol. 2019;181:907-915.
- Passeron T, Bouillon R, Callender V, et al. Sunscreen photoprotection and vitamin D status. Br J Dermatol. 2019;181:916-931.
- Tran V, Duarte Romero BL, Andersen H, et al. Effect of daily sunscreen application on vitamin D: findings from the open-label randomized controlled Sun-D Trial. Br J Dermatol. 2025;193:1128-1137.
Sunscreen continues to be the foundation of successful skin cancer prevention. Daily sunscreen application and reapplication are recommended to prevent all types of skin cancer, with the strongest body of evidence supporting prevention of squamous cell carcinoma.1 Sunscreens have been used safely for decades; since my last update in 2024,2 no evidence of harm has emerged, despite the fact that organic (chemical) sunscreen filters are absorbed systemically.3
So, what’s happening with sunscreen in 2026? Let’s review some hot news and fresh controversies.
Sunscreen Doping Is Pervasive
Sunscreen “doping” signifies the sneaky addition of UV filters that have not been approved by the US Food and Drug Administration (FDA) into sunscreens under the guise of inactive ingredients. Why would a manufacturer do such a thing? To enhance a sunscreen’s UV absorption without having to increase the concentration of zinc oxide/titanium dioxide (which creates an undesirable white cast) or exceeding the maximum permitted concentration of chemical active ingredients.4,5 In a 2025 analysis of the top 150 sunscreens sold on Amazon, 48.3% contained these covert UV filters, including almost half of those marketed as mineral-only products.6 The most prevalent doping ingredient was butyloctyl salicylate, which is chemically and functionally related to the FDA-approved chemical UV filter octisalate (ethylhexyl salicylate).5
The practice of sunscreen doping is deceptive. Can a product be accurately marketed as mineral sunscreen if it contains ingredients that function as chemical UV filters but are not classified as active ingredients by the FDA? The bigger picture is that sunscreen doping is a symptom of regulatory malaise specific to the United States. Regulation of sunscreens as over-the-counter drugs plus the FDA’s stringent requirements for UV filters to be generally recognized as safe and effective (GRASE) have stymied the approval process to the extent that no new active ingredients have been approved since 1999.2 The FDA allows 16 active ingredients compared to about 30 in Europe and Asia—not for lack of safety evaluations prior to approval in those regions.7 In the United States, getting a new active sunscreen ingredient approved is far more onerous and costly than the streamlined processes that are in place abroad. This restricts sunscreen innovation; in particular, the US market lacks the wide variety of international options for protection against long-wave UVA radiation, remaining limited to just avobenzone and zinc oxide. Since long-wave UVA plays a major role in photoaging, this represents a gap in protection compared to international sunscreen offerings.1,7 Due to domestic sunscreen limitations, some Americans have turned to purchasing non–FDA-approved sunscreens abroad or through online channels.8
New Sunscreen Filter Pending Approval, and Hope for Regulatory Changes
Let’s move on to a more positive development. A new sunscreen filter is actually nearing approval in the United States! Bemotrizinol, also known as bis-ethylhexyloxyphenol methoxyphenyl triazine, is a broad-spectrum chemical UVA/UVB blocker that would represent the first new active ingredient to become available in the United States since 1999.9 It satisfies the FDA requirement for minimal systemic absorption and GRASE status and has been used with a clean safety record since 2000 in Europe.10 The icing on top is that bemotrizinol seems to be minimally allergenic, with only a few published reports of contact dermatitis over several decades of use.11,12
Yes, as I write, the FDA is on the cusp of approving bemotrizinol, a great broad-spectrum sunscreen ingredient, to one day be added to the products on our shelves. The cynic in me can’t help but point out that it took more than 20 years of effort and an estimated $20 million to get us to this point of near-approval of one new sunscreen filter.13
Perhaps things won’t be so difficult in the future. In late 2025, the bipartisan Supporting Accessible, Flexible, and Effective (SAFE) Sunscreen Standards Act was signed into law.14 The SAFE Sunscreen Standards Act calls on the FDA to be more flexible and allow for the use of real-world evidence and observational studies to demonstrate safety and effectiveness of active ingredients used in sunscreens. We can only hope that real change is forthcoming and that future sunscreen approvals won’t require decades of work and millions of dollars, as in the case of bemotrizinol.
Daily Sunscreen Use Linked to Reduction in Vitamin D Levels
The UVB wavelengths that cause sunburn overlap with those that initiate vitamin D production in the skin, generating concerns about sunscreen use reducing vitamin D levels. Nevertheless, in 2019, expert opinion and a systematic literature review determined that routine use of sunscreen was unlikely to be associated with a reduction in vitamin D levels.15,16 However, a major limitation at that time was a lack of studies examining vitamin D status in individuals using high–sun protection factor (SPF) sunscreens.
Now we have results from the first field study assessing the impact of long-term daily application of higher SPF sunscreen on vitamin D levels. In the Australian Sun-D Trial, Tran et al17 randomly assigned 639 participants to either an intervention group (routine application of SPF 50+ sunscreen on days forecasted to have a UV index ≥3) or a control group (discretionary sunscreen use). Vitamin D levels were measured at baseline in the winter/spring, at the end of summer, and then at the end of the following winter. At the end of summer, vitamin D levels increased in both groups but less in the intervention group, then decreased similarly in both groups by winter. Routine sunscreen application was associated with a decrease of 5.2 nmol/L (2.1 ng/mL) in vitamin D levels, which the authors rightfully considered to be modest. Additionally, vitamin D deficiency (defined as <50 nmol/L [<20 ng/mL]) was detected in more of the intervention group compared to the control group (45.7% vs 36.9%). The study reasonably concluded that sunscreen continues to be essential in preventing skin cancers but regular users may require vitamin D testing and/or supplementation.17
Looking Ahead
In this update, I discussed several important pieces of sunscreen news. If you check your favorite mineral sunscreen’s ingredients list, odds are you will find it also contains inactive doping ingredients shown to secretly enhance UV protection. Perhaps manufacturers won’t have to dope sunscreens in the United States forever if regulatory reforms facilitate the approval of active ingredients such as bemotrizinol used safely in other countries without huge investments of time and money. For daily sunscreen users, consider checking and/or empirically supplementing vitamin D.
None of this should discourage us from recommending regular consistent sunscreen application and reapplication to our patients. There continues to be a lack of evidence of harms associated with systemic absorption of chemical UV filters in humans, and sunscreen will continue to function as an indispensable component of skin cancer prevention for the foreseeable future.
Sunscreen continues to be the foundation of successful skin cancer prevention. Daily sunscreen application and reapplication are recommended to prevent all types of skin cancer, with the strongest body of evidence supporting prevention of squamous cell carcinoma.1 Sunscreens have been used safely for decades; since my last update in 2024,2 no evidence of harm has emerged, despite the fact that organic (chemical) sunscreen filters are absorbed systemically.3
So, what’s happening with sunscreen in 2026? Let’s review some hot news and fresh controversies.
Sunscreen Doping Is Pervasive
Sunscreen “doping” signifies the sneaky addition of UV filters that have not been approved by the US Food and Drug Administration (FDA) into sunscreens under the guise of inactive ingredients. Why would a manufacturer do such a thing? To enhance a sunscreen’s UV absorption without having to increase the concentration of zinc oxide/titanium dioxide (which creates an undesirable white cast) or exceeding the maximum permitted concentration of chemical active ingredients.4,5 In a 2025 analysis of the top 150 sunscreens sold on Amazon, 48.3% contained these covert UV filters, including almost half of those marketed as mineral-only products.6 The most prevalent doping ingredient was butyloctyl salicylate, which is chemically and functionally related to the FDA-approved chemical UV filter octisalate (ethylhexyl salicylate).5
The practice of sunscreen doping is deceptive. Can a product be accurately marketed as mineral sunscreen if it contains ingredients that function as chemical UV filters but are not classified as active ingredients by the FDA? The bigger picture is that sunscreen doping is a symptom of regulatory malaise specific to the United States. Regulation of sunscreens as over-the-counter drugs plus the FDA’s stringent requirements for UV filters to be generally recognized as safe and effective (GRASE) have stymied the approval process to the extent that no new active ingredients have been approved since 1999.2 The FDA allows 16 active ingredients compared to about 30 in Europe and Asia—not for lack of safety evaluations prior to approval in those regions.7 In the United States, getting a new active sunscreen ingredient approved is far more onerous and costly than the streamlined processes that are in place abroad. This restricts sunscreen innovation; in particular, the US market lacks the wide variety of international options for protection against long-wave UVA radiation, remaining limited to just avobenzone and zinc oxide. Since long-wave UVA plays a major role in photoaging, this represents a gap in protection compared to international sunscreen offerings.1,7 Due to domestic sunscreen limitations, some Americans have turned to purchasing non–FDA-approved sunscreens abroad or through online channels.8
New Sunscreen Filter Pending Approval, and Hope for Regulatory Changes
Let’s move on to a more positive development. A new sunscreen filter is actually nearing approval in the United States! Bemotrizinol, also known as bis-ethylhexyloxyphenol methoxyphenyl triazine, is a broad-spectrum chemical UVA/UVB blocker that would represent the first new active ingredient to become available in the United States since 1999.9 It satisfies the FDA requirement for minimal systemic absorption and GRASE status and has been used with a clean safety record since 2000 in Europe.10 The icing on top is that bemotrizinol seems to be minimally allergenic, with only a few published reports of contact dermatitis over several decades of use.11,12
Yes, as I write, the FDA is on the cusp of approving bemotrizinol, a great broad-spectrum sunscreen ingredient, to one day be added to the products on our shelves. The cynic in me can’t help but point out that it took more than 20 years of effort and an estimated $20 million to get us to this point of near-approval of one new sunscreen filter.13
Perhaps things won’t be so difficult in the future. In late 2025, the bipartisan Supporting Accessible, Flexible, and Effective (SAFE) Sunscreen Standards Act was signed into law.14 The SAFE Sunscreen Standards Act calls on the FDA to be more flexible and allow for the use of real-world evidence and observational studies to demonstrate safety and effectiveness of active ingredients used in sunscreens. We can only hope that real change is forthcoming and that future sunscreen approvals won’t require decades of work and millions of dollars, as in the case of bemotrizinol.
Daily Sunscreen Use Linked to Reduction in Vitamin D Levels
The UVB wavelengths that cause sunburn overlap with those that initiate vitamin D production in the skin, generating concerns about sunscreen use reducing vitamin D levels. Nevertheless, in 2019, expert opinion and a systematic literature review determined that routine use of sunscreen was unlikely to be associated with a reduction in vitamin D levels.15,16 However, a major limitation at that time was a lack of studies examining vitamin D status in individuals using high–sun protection factor (SPF) sunscreens.
Now we have results from the first field study assessing the impact of long-term daily application of higher SPF sunscreen on vitamin D levels. In the Australian Sun-D Trial, Tran et al17 randomly assigned 639 participants to either an intervention group (routine application of SPF 50+ sunscreen on days forecasted to have a UV index ≥3) or a control group (discretionary sunscreen use). Vitamin D levels were measured at baseline in the winter/spring, at the end of summer, and then at the end of the following winter. At the end of summer, vitamin D levels increased in both groups but less in the intervention group, then decreased similarly in both groups by winter. Routine sunscreen application was associated with a decrease of 5.2 nmol/L (2.1 ng/mL) in vitamin D levels, which the authors rightfully considered to be modest. Additionally, vitamin D deficiency (defined as <50 nmol/L [<20 ng/mL]) was detected in more of the intervention group compared to the control group (45.7% vs 36.9%). The study reasonably concluded that sunscreen continues to be essential in preventing skin cancers but regular users may require vitamin D testing and/or supplementation.17
Looking Ahead
In this update, I discussed several important pieces of sunscreen news. If you check your favorite mineral sunscreen’s ingredients list, odds are you will find it also contains inactive doping ingredients shown to secretly enhance UV protection. Perhaps manufacturers won’t have to dope sunscreens in the United States forever if regulatory reforms facilitate the approval of active ingredients such as bemotrizinol used safely in other countries without huge investments of time and money. For daily sunscreen users, consider checking and/or empirically supplementing vitamin D.
None of this should discourage us from recommending regular consistent sunscreen application and reapplication to our patients. There continues to be a lack of evidence of harms associated with systemic absorption of chemical UV filters in humans, and sunscreen will continue to function as an indispensable component of skin cancer prevention for the foreseeable future.
- Abdel Azim S, Bainvoll L, Vecerek N, et al. Sunscreens part 1: mechanisms and efficacy. J Am Acad Dermatol. 2025;92:677-686.
- Adler BL. Sunscreen safety: 2024 updates. Cutis. 2024;113:195-196.
- Abdel Azim S, Bainvoll L, Vecerek N, et al. Sunscreens part 2: regulation and safety. J Am Acad Dermatol. 2025;92:689-698.
- Gawey L, Gunnell L, Shi VY. Sunscreen doping: a misnomer. J Invest Dermatol. 2025;145:2941-2942.
- Moradi Tuchayi S, Wang Z, Yan J, et al. Sunscreens: misconceptions and misinformation. J Invest Dermatol. 2023;143:1406-1411.
- Gawey L, Gunnell L, Joshi A, et al. Hidden UV filters: an analysis of the 150 most popular sunscreens. J Am Acad Dermatol. 2025;93:1594-1596.
- Pantelic MN, Wong N, Kwa M, et al. Ultraviolet filters in the United States and European Union: a review of safety and implications for the future of US sunscreens. J Am Acad Dermatol. 2023;88:632-646.
- Scaturro M. Other countries have better sunscreens. here’s why we can’t get them in the U.S. NPR. May 18, 2024. Accessed April 9, 2026. https://www.npr.org/sections/health-shots/2024/05/18/1251919831/sunscreen-effective-better-ingredients-fda
- US Food & Drug Administration. FDA proposes expanding sunscreen active ingredient list [news release]. December 11, 2025. Accessed February 24, 2026. https://www.fda.gov/news-events/press-announcements/fda-proposes-expanding-sunscreen-active-ingredient-list
- D’Ruiz CD, Plautz JR, Schuetz R, et al. Preliminary clinical pharmacokinetic evaluation of bemotrizinol—a new sunscreen active ingredient being considered for inclusion under FDA’s over-the-counter (OTC) sunscreen monograph. Regul Toxicol Pharmacol. 2023;139:105344.
- Badaoui A. Allergic contact dermatitis to bis-ethylhexyloxyphenol methoxyphenyl triazine (Tinosorb S) in a sunscreen: an emerging allergen? Contact Dermatitis. 2024;91:443-445.
- European Multicentre Photopatch Test Study (EMCPPTS) Taskforce. A European multicentre photopatch test study. Br J Dermatol. 2012;166:1002-1009.
- Nesvig K. The FDA finally proposed the approval of a new sunscreen filter. December 11, 2025. Accessed February 22, 2026. https://www.allure.com/story/fda-proposes-bemotrizinol-suncreen-filter-approval
- SAFE Sunscreen Standards Act, HR 3686, 119th Congress (2025-2026). Accessed February 23, 2026. https://www.congress.gov/bill/119th-congress/house-bill/3686/text
- Neale RE, Khan SR, Lucas RM, et al. The effect of sunscreen on vitamin D: a review. Br J Dermatol. 2019;181:907-915.
- Passeron T, Bouillon R, Callender V, et al. Sunscreen photoprotection and vitamin D status. Br J Dermatol. 2019;181:916-931.
- Tran V, Duarte Romero BL, Andersen H, et al. Effect of daily sunscreen application on vitamin D: findings from the open-label randomized controlled Sun-D Trial. Br J Dermatol. 2025;193:1128-1137.
- Abdel Azim S, Bainvoll L, Vecerek N, et al. Sunscreens part 1: mechanisms and efficacy. J Am Acad Dermatol. 2025;92:677-686.
- Adler BL. Sunscreen safety: 2024 updates. Cutis. 2024;113:195-196.
- Abdel Azim S, Bainvoll L, Vecerek N, et al. Sunscreens part 2: regulation and safety. J Am Acad Dermatol. 2025;92:689-698.
- Gawey L, Gunnell L, Shi VY. Sunscreen doping: a misnomer. J Invest Dermatol. 2025;145:2941-2942.
- Moradi Tuchayi S, Wang Z, Yan J, et al. Sunscreens: misconceptions and misinformation. J Invest Dermatol. 2023;143:1406-1411.
- Gawey L, Gunnell L, Joshi A, et al. Hidden UV filters: an analysis of the 150 most popular sunscreens. J Am Acad Dermatol. 2025;93:1594-1596.
- Pantelic MN, Wong N, Kwa M, et al. Ultraviolet filters in the United States and European Union: a review of safety and implications for the future of US sunscreens. J Am Acad Dermatol. 2023;88:632-646.
- Scaturro M. Other countries have better sunscreens. here’s why we can’t get them in the U.S. NPR. May 18, 2024. Accessed April 9, 2026. https://www.npr.org/sections/health-shots/2024/05/18/1251919831/sunscreen-effective-better-ingredients-fda
- US Food & Drug Administration. FDA proposes expanding sunscreen active ingredient list [news release]. December 11, 2025. Accessed February 24, 2026. https://www.fda.gov/news-events/press-announcements/fda-proposes-expanding-sunscreen-active-ingredient-list
- D’Ruiz CD, Plautz JR, Schuetz R, et al. Preliminary clinical pharmacokinetic evaluation of bemotrizinol—a new sunscreen active ingredient being considered for inclusion under FDA’s over-the-counter (OTC) sunscreen monograph. Regul Toxicol Pharmacol. 2023;139:105344.
- Badaoui A. Allergic contact dermatitis to bis-ethylhexyloxyphenol methoxyphenyl triazine (Tinosorb S) in a sunscreen: an emerging allergen? Contact Dermatitis. 2024;91:443-445.
- European Multicentre Photopatch Test Study (EMCPPTS) Taskforce. A European multicentre photopatch test study. Br J Dermatol. 2012;166:1002-1009.
- Nesvig K. The FDA finally proposed the approval of a new sunscreen filter. December 11, 2025. Accessed February 22, 2026. https://www.allure.com/story/fda-proposes-bemotrizinol-suncreen-filter-approval
- SAFE Sunscreen Standards Act, HR 3686, 119th Congress (2025-2026). Accessed February 23, 2026. https://www.congress.gov/bill/119th-congress/house-bill/3686/text
- Neale RE, Khan SR, Lucas RM, et al. The effect of sunscreen on vitamin D: a review. Br J Dermatol. 2019;181:907-915.
- Passeron T, Bouillon R, Callender V, et al. Sunscreen photoprotection and vitamin D status. Br J Dermatol. 2019;181:916-931.
- Tran V, Duarte Romero BL, Andersen H, et al. Effect of daily sunscreen application on vitamin D: findings from the open-label randomized controlled Sun-D Trial. Br J Dermatol. 2025;193:1128-1137.
Sunscreen in 2026: Doping, Regulatory Changes, and Impact on Vitamin D Levels
Sunscreen in 2026: Doping, Regulatory Changes, and Impact on Vitamin D Levels
Predictors of Lidocaine Volume Used During Mohs Micrographic Surgery
Predictors of Lidocaine Volume Used During Mohs Micrographic Surgery
To the Editor:
Mohs micrographic surgery (MMS) is performed in stages and often requires repeated administration of a local anesthetic, most commonly lidocaine. While generally safe, lidocaine administration carries the potential for cumulative toxicity, particularly in patients who have large or multiple lesions or medical comorbidities or who require extensive repair. Current safety guidelines suggest upper limits of 7 mg/kg (or 500 mg) of lidocaine with epinephrine and 4.5 mg/kg (or 300 mg) without epinephrine for adults.1 However, concerns have been raised about the relevance of these thresholds to MMS, in which anesthetic administration may be prolonged, cumulative, and influenced by surgical complexity.2-5 While clinical experience often guides anesthetic planning, limited data exist identifying predictors of lidocaine use during MMS.
We performed an institutional review board–approved retrospective chart review of 149 patients who underwent 170 MMS procedures at a single academic dermatologic surgery center between July 2022 and June 2023. The aim of our study was to identify clinical and surgical predictors of lidocaine volume used during MMS. All procedures were performed by board-certified dermatologic surgeons (including A.J.). All patients received 1% lidocaine with epinephrine as the primary anesthetic agent. We collected patient demographic variables (age, sex, race, weight), procedural characteristics (anatomic site, number of Mohs stages, skin cancer type, number of surgical sites treated in one day, preoperative and postoperative lesion size, surgeon, repair type), comorbid conditions (hypertension, diabetes), and time from diagnosis to surgery. Data were extracted from the institutional REDCap system. We used t tests and analysis of variance for categorical variables and linear regression for continuous predictors, with statistical significance set at P<.05.
Baseline characteristics of the study patients are outlined in Table 1. The mean (SD) age was 74.2 (9.4) years, and most patients (98.7% [147/149]) were White. The mean (SD) weight was 83.1 (19.1) kg. Most lesions were either basal cell carcinoma (BCC)(50.6%) or squamous cell carcinoma (SCC)(44.1%), with 5.3% of lesions representing melanoma. The mean (SD) total lidocaine volume administered was 11.8 (8.3) mL. The majority (123/149 [72.4%]) of cases required one Mohs stage, but a subset required multiple stages, with a maximum of 5.
Several procedural and patient factors were significantly associated with the volume of lidocaine used. As expected, lesion size strongly influenced lidocaine volume. Both preoperative and postoperative lesion sizes were highly significant linear predictors (R2=0.28 and 0.41, respectively; P<.001), and postoperative lesion size demonstrated the strongest correlation of all tested variables. Patient weight was also significantly associated with lidocaine use (R2=.03, P=.0202), though the proportion of explained variance was modest. The operating surgeon also was significantly associated with lidocaine use (P=.006), suggesting potential variation in anesthetic technique or threshold for reinfiltration. The number of surgical sites treated in a single session also was significantly associated with greater lidocaine volume (P<.001).
Skin cancer type was a notable categorical predictor. Melanomas required substantially more lidocaine than BCCs or SCCs, with a mean (SD) volume of 25.6 (12.1) mL compared with 10.8 (6.0) mL for BCC and 11.4 (8.8) mL for SCC (P<.001). This difference may reflect disparities in surgical margin requirements, tumor depth, or intraoperative technique. While lesion location and number of stages were not statistically significant overall, mean lidocaine volumes trended higher in lesions on the trunk (18.2 mL) and in procedures requiring 3 or more stages (up to 22.0 mL for a single 4-stage case), though small sample sizes limited the ability to detect statistically significant differences in these subgroups. Detailed comparisons are presented in Table 2.
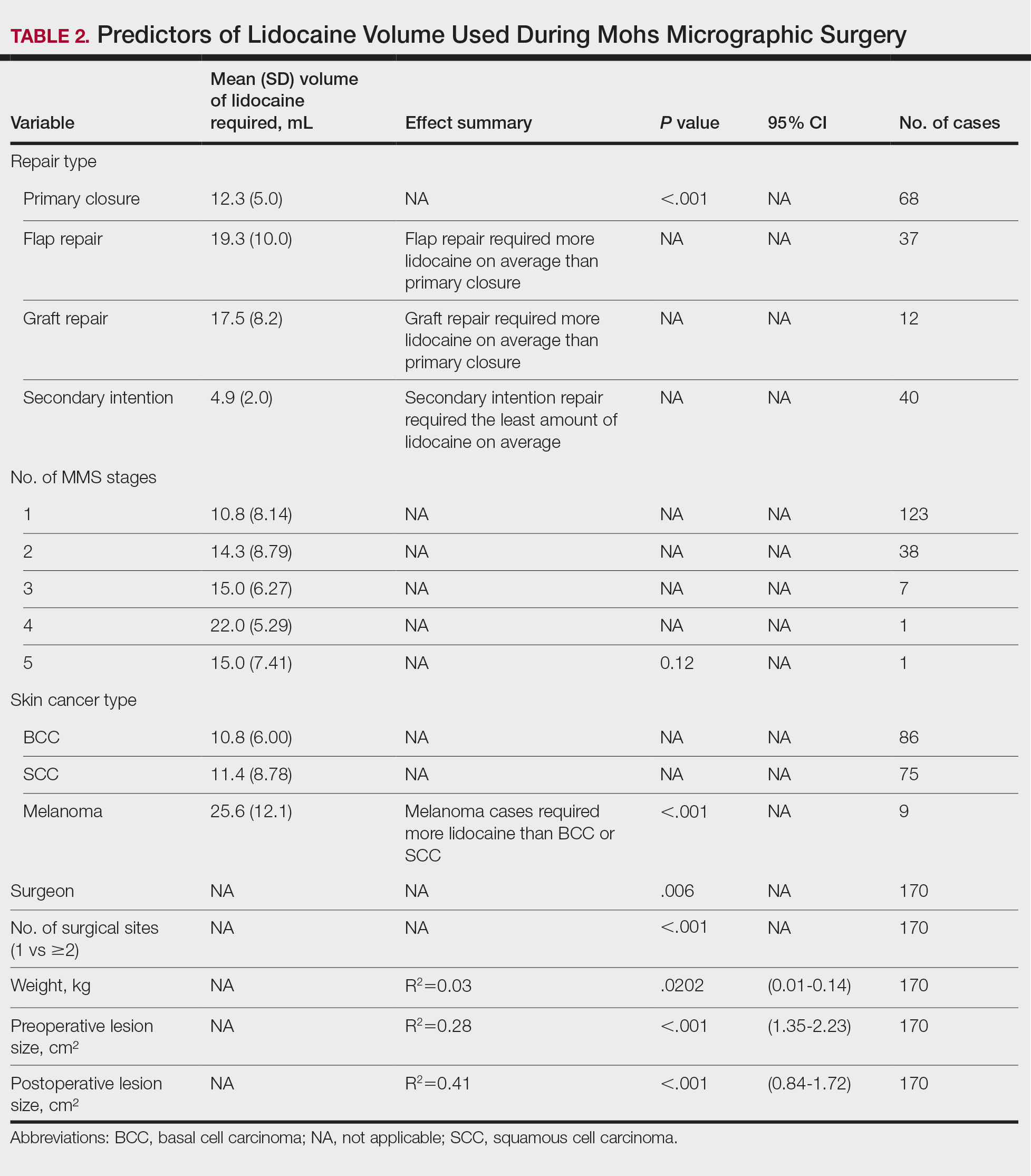
Wound repair type also was significantly associated with lidocaine volume requirements. Primary closures required a mean (SD) volume of 12.3 (5.0) mL, whereas flap repairs required 19.3 (10.0) mL and graft repairs required 17.5 (8.2) mL. Secondary-intention healing used the lowest lidocaine volumes (mean [SD], 4.9 [2.0] mL). Differences across repair types were statistically significant (analysis of variance, P<.001). These findings indicate that more complex reconstructions, such as flaps and grafts, are associated with higher anesthetic needs when compared with primary closures or secondary-intention healing.
Several other predictors, including age, time from diagnosis to surgery, and comorbid conditions such as hypertension or diabetes, were not significantly associated with anesthetic volume in our cohort. Time from diagnosis to surgery ranged widely but did not correlate with lesion size or lidocaine use, possibly due to scheduling variability or biopsy technique.
These findings offer practical implications for clinical planning. While most MMS cases fall well within safe limits for lidocaine administration, some patients—particularly those with melanoma, large lesions, or multiple surgical sites—may approach thresholds at which further monitoring or dose tracking becomes relevant. Anticipating higher anesthetic requirements may help surgical teams plan procedure length, anesthesia restocking, or sequencing of multisite cases. Our analysis also showed that the type of wound repair meaningfully influences anesthetic use, with flap and graft repairs requiring substantially higher lidocaine volumes than primary closures and secondary-intention healing. Considering both tumor characteristics and the planned reconstruction may therefore improve the accuracy of anesthetic forecasting during preoperative planning.
We also observed surgeon-level variation in lidocaine volume despite standardized tumor types and case complexity. This suggests a role for individual technique (eg, depth of field block, number of reinfiltrations) and highlights the need for ongoing education around anesthetic optimization.
Our study was limited by its retrospective design, single-institution setting, and demographically homogeneous population. With 98.8% of patients identifying as White, generalizability to skin of color populations may be limited. In addition, lidocaine metabolism may vary across patient factors not captured here (eg, hepatic or renal function). Finally, although lidocaine volume was the outcome of interest, we did not measure patient-reported pain control, which may further clarify anesthetic adequacy. Nonetheless, our analysis demonstrated that routinely available clinical and procedural data can predict lidocaine volume requirements with reasonable reliability. Although no patient in our cohort approached the maximum recommended lidocaine dose, understanding these predictors may help anticipate scenarios nearing maximum dosing thresholds. In future studies, integrating weight-based thresholds (eg, mL/kg received) or serum lidocaine levels may improve safety monitoring and validate toxicity thresholds in complex cases.
In conclusion, we identified several key factors that predict lidocaine volume during MMS, including lesion size, melanoma diagnosis, number of surgical sites, patient weight, planned reconstruction type, and the operating surgeon. Among these factors, melanoma cases required more than twice the volume of lidocaine compared to BCC and SCC cases, and flap and graft repairs demonstrated the highest anesthetic requirements among closure types. Taken together, these findings reinforce the need for advanced anesthetic planning in aggressive, anatomically complex, or reconstruction-intensive cases and may support more informed intraoperative decision-making.
- Kouba DJ, LoPiccolo MC, Alam M, et al. Guidelines for the use of local anesthesia in office-based dermatologic surgery. J Am Acad Dermatol. 2016;74:1201-1219. doi:10.1016/j.jaad.2016.01.022
- Wang A, Grushchak S, Kaul S, et al. Toxicity of infiltrative lidocaine in dermatologic surgery: are current limits valid? Dermatol Pract Concept. 2021;11:e2021120. doi:10.5826/dpc.1104a120
- Patrinely JR Jr, Darragh C, Frank N, et al. Risk of adverse events due to high volumes of local anesthesia during Mohs micrographic surgery. Arch Dermatol Res. 2021;313:679-684. doi:10.1007/s00403-020-02155-1
- Butterwick KJ, Goldman MP, Sriprachya-Anunt S. Lidocaine levels during the first two hours of infiltration of dilute anesthetic solution for tumescent liposuction: rapid versus slow delivery. Dermatol Surg. 1999;25:681-685. doi:10.1046/j.1524-4725.1999.98275.x
- Flanagan K, McLean R, Goldberg D. Is it time to redefine lidocaine administration guidelines in Mohs surgery? J Drugs Dermatol. 2020;19:433.
To the Editor:
Mohs micrographic surgery (MMS) is performed in stages and often requires repeated administration of a local anesthetic, most commonly lidocaine. While generally safe, lidocaine administration carries the potential for cumulative toxicity, particularly in patients who have large or multiple lesions or medical comorbidities or who require extensive repair. Current safety guidelines suggest upper limits of 7 mg/kg (or 500 mg) of lidocaine with epinephrine and 4.5 mg/kg (or 300 mg) without epinephrine for adults.1 However, concerns have been raised about the relevance of these thresholds to MMS, in which anesthetic administration may be prolonged, cumulative, and influenced by surgical complexity.2-5 While clinical experience often guides anesthetic planning, limited data exist identifying predictors of lidocaine use during MMS.
We performed an institutional review board–approved retrospective chart review of 149 patients who underwent 170 MMS procedures at a single academic dermatologic surgery center between July 2022 and June 2023. The aim of our study was to identify clinical and surgical predictors of lidocaine volume used during MMS. All procedures were performed by board-certified dermatologic surgeons (including A.J.). All patients received 1% lidocaine with epinephrine as the primary anesthetic agent. We collected patient demographic variables (age, sex, race, weight), procedural characteristics (anatomic site, number of Mohs stages, skin cancer type, number of surgical sites treated in one day, preoperative and postoperative lesion size, surgeon, repair type), comorbid conditions (hypertension, diabetes), and time from diagnosis to surgery. Data were extracted from the institutional REDCap system. We used t tests and analysis of variance for categorical variables and linear regression for continuous predictors, with statistical significance set at P<.05.
Baseline characteristics of the study patients are outlined in Table 1. The mean (SD) age was 74.2 (9.4) years, and most patients (98.7% [147/149]) were White. The mean (SD) weight was 83.1 (19.1) kg. Most lesions were either basal cell carcinoma (BCC)(50.6%) or squamous cell carcinoma (SCC)(44.1%), with 5.3% of lesions representing melanoma. The mean (SD) total lidocaine volume administered was 11.8 (8.3) mL. The majority (123/149 [72.4%]) of cases required one Mohs stage, but a subset required multiple stages, with a maximum of 5.

Several procedural and patient factors were significantly associated with the volume of lidocaine used. As expected, lesion size strongly influenced lidocaine volume. Both preoperative and postoperative lesion sizes were highly significant linear predictors (R2=0.28 and 0.41, respectively; P<.001), and postoperative lesion size demonstrated the strongest correlation of all tested variables. Patient weight was also significantly associated with lidocaine use (R2=.03, P=.0202), though the proportion of explained variance was modest. The operating surgeon also was significantly associated with lidocaine use (P=.006), suggesting potential variation in anesthetic technique or threshold for reinfiltration. The number of surgical sites treated in a single session also was significantly associated with greater lidocaine volume (P<.001).
Skin cancer type was a notable categorical predictor. Melanomas required substantially more lidocaine than BCCs or SCCs, with a mean (SD) volume of 25.6 (12.1) mL compared with 10.8 (6.0) mL for BCC and 11.4 (8.8) mL for SCC (P<.001). This difference may reflect disparities in surgical margin requirements, tumor depth, or intraoperative technique. While lesion location and number of stages were not statistically significant overall, mean lidocaine volumes trended higher in lesions on the trunk (18.2 mL) and in procedures requiring 3 or more stages (up to 22.0 mL for a single 4-stage case), though small sample sizes limited the ability to detect statistically significant differences in these subgroups. Detailed comparisons are presented in Table 2.

Wound repair type also was significantly associated with lidocaine volume requirements. Primary closures required a mean (SD) volume of 12.3 (5.0) mL, whereas flap repairs required 19.3 (10.0) mL and graft repairs required 17.5 (8.2) mL. Secondary-intention healing used the lowest lidocaine volumes (mean [SD], 4.9 [2.0] mL). Differences across repair types were statistically significant (analysis of variance, P<.001). These findings indicate that more complex reconstructions, such as flaps and grafts, are associated with higher anesthetic needs when compared with primary closures or secondary-intention healing.
Several other predictors, including age, time from diagnosis to surgery, and comorbid conditions such as hypertension or diabetes, were not significantly associated with anesthetic volume in our cohort. Time from diagnosis to surgery ranged widely but did not correlate with lesion size or lidocaine use, possibly due to scheduling variability or biopsy technique.
These findings offer practical implications for clinical planning. While most MMS cases fall well within safe limits for lidocaine administration, some patients—particularly those with melanoma, large lesions, or multiple surgical sites—may approach thresholds at which further monitoring or dose tracking becomes relevant. Anticipating higher anesthetic requirements may help surgical teams plan procedure length, anesthesia restocking, or sequencing of multisite cases. Our analysis also showed that the type of wound repair meaningfully influences anesthetic use, with flap and graft repairs requiring substantially higher lidocaine volumes than primary closures and secondary-intention healing. Considering both tumor characteristics and the planned reconstruction may therefore improve the accuracy of anesthetic forecasting during preoperative planning.
We also observed surgeon-level variation in lidocaine volume despite standardized tumor types and case complexity. This suggests a role for individual technique (eg, depth of field block, number of reinfiltrations) and highlights the need for ongoing education around anesthetic optimization.
Our study was limited by its retrospective design, single-institution setting, and demographically homogeneous population. With 98.8% of patients identifying as White, generalizability to skin of color populations may be limited. In addition, lidocaine metabolism may vary across patient factors not captured here (eg, hepatic or renal function). Finally, although lidocaine volume was the outcome of interest, we did not measure patient-reported pain control, which may further clarify anesthetic adequacy. Nonetheless, our analysis demonstrated that routinely available clinical and procedural data can predict lidocaine volume requirements with reasonable reliability. Although no patient in our cohort approached the maximum recommended lidocaine dose, understanding these predictors may help anticipate scenarios nearing maximum dosing thresholds. In future studies, integrating weight-based thresholds (eg, mL/kg received) or serum lidocaine levels may improve safety monitoring and validate toxicity thresholds in complex cases.
In conclusion, we identified several key factors that predict lidocaine volume during MMS, including lesion size, melanoma diagnosis, number of surgical sites, patient weight, planned reconstruction type, and the operating surgeon. Among these factors, melanoma cases required more than twice the volume of lidocaine compared to BCC and SCC cases, and flap and graft repairs demonstrated the highest anesthetic requirements among closure types. Taken together, these findings reinforce the need for advanced anesthetic planning in aggressive, anatomically complex, or reconstruction-intensive cases and may support more informed intraoperative decision-making.
To the Editor:
Mohs micrographic surgery (MMS) is performed in stages and often requires repeated administration of a local anesthetic, most commonly lidocaine. While generally safe, lidocaine administration carries the potential for cumulative toxicity, particularly in patients who have large or multiple lesions or medical comorbidities or who require extensive repair. Current safety guidelines suggest upper limits of 7 mg/kg (or 500 mg) of lidocaine with epinephrine and 4.5 mg/kg (or 300 mg) without epinephrine for adults.1 However, concerns have been raised about the relevance of these thresholds to MMS, in which anesthetic administration may be prolonged, cumulative, and influenced by surgical complexity.2-5 While clinical experience often guides anesthetic planning, limited data exist identifying predictors of lidocaine use during MMS.
We performed an institutional review board–approved retrospective chart review of 149 patients who underwent 170 MMS procedures at a single academic dermatologic surgery center between July 2022 and June 2023. The aim of our study was to identify clinical and surgical predictors of lidocaine volume used during MMS. All procedures were performed by board-certified dermatologic surgeons (including A.J.). All patients received 1% lidocaine with epinephrine as the primary anesthetic agent. We collected patient demographic variables (age, sex, race, weight), procedural characteristics (anatomic site, number of Mohs stages, skin cancer type, number of surgical sites treated in one day, preoperative and postoperative lesion size, surgeon, repair type), comorbid conditions (hypertension, diabetes), and time from diagnosis to surgery. Data were extracted from the institutional REDCap system. We used t tests and analysis of variance for categorical variables and linear regression for continuous predictors, with statistical significance set at P<.05.
Baseline characteristics of the study patients are outlined in Table 1. The mean (SD) age was 74.2 (9.4) years, and most patients (98.7% [147/149]) were White. The mean (SD) weight was 83.1 (19.1) kg. Most lesions were either basal cell carcinoma (BCC)(50.6%) or squamous cell carcinoma (SCC)(44.1%), with 5.3% of lesions representing melanoma. The mean (SD) total lidocaine volume administered was 11.8 (8.3) mL. The majority (123/149 [72.4%]) of cases required one Mohs stage, but a subset required multiple stages, with a maximum of 5.

Several procedural and patient factors were significantly associated with the volume of lidocaine used. As expected, lesion size strongly influenced lidocaine volume. Both preoperative and postoperative lesion sizes were highly significant linear predictors (R2=0.28 and 0.41, respectively; P<.001), and postoperative lesion size demonstrated the strongest correlation of all tested variables. Patient weight was also significantly associated with lidocaine use (R2=.03, P=.0202), though the proportion of explained variance was modest. The operating surgeon also was significantly associated with lidocaine use (P=.006), suggesting potential variation in anesthetic technique or threshold for reinfiltration. The number of surgical sites treated in a single session also was significantly associated with greater lidocaine volume (P<.001).
Skin cancer type was a notable categorical predictor. Melanomas required substantially more lidocaine than BCCs or SCCs, with a mean (SD) volume of 25.6 (12.1) mL compared with 10.8 (6.0) mL for BCC and 11.4 (8.8) mL for SCC (P<.001). This difference may reflect disparities in surgical margin requirements, tumor depth, or intraoperative technique. While lesion location and number of stages were not statistically significant overall, mean lidocaine volumes trended higher in lesions on the trunk (18.2 mL) and in procedures requiring 3 or more stages (up to 22.0 mL for a single 4-stage case), though small sample sizes limited the ability to detect statistically significant differences in these subgroups. Detailed comparisons are presented in Table 2.

Wound repair type also was significantly associated with lidocaine volume requirements. Primary closures required a mean (SD) volume of 12.3 (5.0) mL, whereas flap repairs required 19.3 (10.0) mL and graft repairs required 17.5 (8.2) mL. Secondary-intention healing used the lowest lidocaine volumes (mean [SD], 4.9 [2.0] mL). Differences across repair types were statistically significant (analysis of variance, P<.001). These findings indicate that more complex reconstructions, such as flaps and grafts, are associated with higher anesthetic needs when compared with primary closures or secondary-intention healing.
Several other predictors, including age, time from diagnosis to surgery, and comorbid conditions such as hypertension or diabetes, were not significantly associated with anesthetic volume in our cohort. Time from diagnosis to surgery ranged widely but did not correlate with lesion size or lidocaine use, possibly due to scheduling variability or biopsy technique.
These findings offer practical implications for clinical planning. While most MMS cases fall well within safe limits for lidocaine administration, some patients—particularly those with melanoma, large lesions, or multiple surgical sites—may approach thresholds at which further monitoring or dose tracking becomes relevant. Anticipating higher anesthetic requirements may help surgical teams plan procedure length, anesthesia restocking, or sequencing of multisite cases. Our analysis also showed that the type of wound repair meaningfully influences anesthetic use, with flap and graft repairs requiring substantially higher lidocaine volumes than primary closures and secondary-intention healing. Considering both tumor characteristics and the planned reconstruction may therefore improve the accuracy of anesthetic forecasting during preoperative planning.
We also observed surgeon-level variation in lidocaine volume despite standardized tumor types and case complexity. This suggests a role for individual technique (eg, depth of field block, number of reinfiltrations) and highlights the need for ongoing education around anesthetic optimization.
Our study was limited by its retrospective design, single-institution setting, and demographically homogeneous population. With 98.8% of patients identifying as White, generalizability to skin of color populations may be limited. In addition, lidocaine metabolism may vary across patient factors not captured here (eg, hepatic or renal function). Finally, although lidocaine volume was the outcome of interest, we did not measure patient-reported pain control, which may further clarify anesthetic adequacy. Nonetheless, our analysis demonstrated that routinely available clinical and procedural data can predict lidocaine volume requirements with reasonable reliability. Although no patient in our cohort approached the maximum recommended lidocaine dose, understanding these predictors may help anticipate scenarios nearing maximum dosing thresholds. In future studies, integrating weight-based thresholds (eg, mL/kg received) or serum lidocaine levels may improve safety monitoring and validate toxicity thresholds in complex cases.
In conclusion, we identified several key factors that predict lidocaine volume during MMS, including lesion size, melanoma diagnosis, number of surgical sites, patient weight, planned reconstruction type, and the operating surgeon. Among these factors, melanoma cases required more than twice the volume of lidocaine compared to BCC and SCC cases, and flap and graft repairs demonstrated the highest anesthetic requirements among closure types. Taken together, these findings reinforce the need for advanced anesthetic planning in aggressive, anatomically complex, or reconstruction-intensive cases and may support more informed intraoperative decision-making.
- Kouba DJ, LoPiccolo MC, Alam M, et al. Guidelines for the use of local anesthesia in office-based dermatologic surgery. J Am Acad Dermatol. 2016;74:1201-1219. doi:10.1016/j.jaad.2016.01.022
- Wang A, Grushchak S, Kaul S, et al. Toxicity of infiltrative lidocaine in dermatologic surgery: are current limits valid? Dermatol Pract Concept. 2021;11:e2021120. doi:10.5826/dpc.1104a120
- Patrinely JR Jr, Darragh C, Frank N, et al. Risk of adverse events due to high volumes of local anesthesia during Mohs micrographic surgery. Arch Dermatol Res. 2021;313:679-684. doi:10.1007/s00403-020-02155-1
- Butterwick KJ, Goldman MP, Sriprachya-Anunt S. Lidocaine levels during the first two hours of infiltration of dilute anesthetic solution for tumescent liposuction: rapid versus slow delivery. Dermatol Surg. 1999;25:681-685. doi:10.1046/j.1524-4725.1999.98275.x
- Flanagan K, McLean R, Goldberg D. Is it time to redefine lidocaine administration guidelines in Mohs surgery? J Drugs Dermatol. 2020;19:433.
- Kouba DJ, LoPiccolo MC, Alam M, et al. Guidelines for the use of local anesthesia in office-based dermatologic surgery. J Am Acad Dermatol. 2016;74:1201-1219. doi:10.1016/j.jaad.2016.01.022
- Wang A, Grushchak S, Kaul S, et al. Toxicity of infiltrative lidocaine in dermatologic surgery: are current limits valid? Dermatol Pract Concept. 2021;11:e2021120. doi:10.5826/dpc.1104a120
- Patrinely JR Jr, Darragh C, Frank N, et al. Risk of adverse events due to high volumes of local anesthesia during Mohs micrographic surgery. Arch Dermatol Res. 2021;313:679-684. doi:10.1007/s00403-020-02155-1
- Butterwick KJ, Goldman MP, Sriprachya-Anunt S. Lidocaine levels during the first two hours of infiltration of dilute anesthetic solution for tumescent liposuction: rapid versus slow delivery. Dermatol Surg. 1999;25:681-685. doi:10.1046/j.1524-4725.1999.98275.x
- Flanagan K, McLean R, Goldberg D. Is it time to redefine lidocaine administration guidelines in Mohs surgery? J Drugs Dermatol. 2020;19:433.
Predictors of Lidocaine Volume Used During Mohs Micrographic Surgery
Predictors of Lidocaine Volume Used During Mohs Micrographic Surgery
Practice Points
- Larger lesion size, melanoma diagnosis, and multiple surgical sites are associated with higher lidocaine volume requirements during Mohs micrographic surgery.
- Melanomas required more than twice the average lidocaine volume compared with basal cell carcinomas and squamous cell carcinomas.
- Flap and graft repairs require substantially more lidocaine than primary closures, while secondary-intention healing uses the least, making reconstruction type an important predictor of total anesthetic needs.
Chromoblastomycosis Leading to Squamous Cell Carcinoma: An Overlooked Outcome of a Neglected Tropical Disease
Chromoblastomycosis Leading to Squamous Cell Carcinoma: An Overlooked Outcome of a Neglected Tropical Disease
Chromoblastomycosis is a neglected tropical implantation mycosis caused by dematiaceous fungi that leads to substantial morbidity. This condition is diagnosed microscopically by visualizing the characteristic thick-walled, single, or multicellular clusters of pigmented fungal cells (also known as medlar bodies, muriform cells, or sclerotic bodies).1 The main causative fungi varies by geographic region, but most commonly is caused by Cladophialophora carrionii, Fonsecaea species, Phialophora verrucosa species complex, and Rhinocladiella aquaspersa.2-4 Standardized treatment guidelines have not been established, but itraconazole typically is considered first-line regardless of causative fungi.5 Terbinafine, other azoles, and topical immunomodulators, either as monotherapy or in combination, may be appropriate alternative or adjunctive options for refractory disease, although supporting data are limited.6-9
Complications from chromoblastomycosis are common, particularly in long-standing, severe, or refractory disease. An analysis using billing codes in the United States found 14% (35/255) of hospitalized patients with chromoblastomycosis had lymphedema.10 In Mexico, 63% (32/51) of patients with chromoblastomycosis developed secondary bacterial infections.11 Skin fibrosis and ankylosis also can occur and cause mobility issues and decreased quality of life. An infrequent but potentially life-threatening complication12 is the development of squamous cell carcinoma (SCC) associated with chronic lesions, representing a preventable end-stage complication of delayed diagnosis and treatment (Figure).
In this review, we summarize reported epidemiology and clinical risk factors for SCC complicating chromoblastomycosis. We also discuss plausible inflammatory mechanisms of malignant transformation and propose pragmatic clinical and public health interventions, including decentralized microscopy-based diagnosis, timely antifungal access, and biopsy-triggered surveillance of chronically inflamed lesions, to reduce preventable morbidity.
Epidemiology and Risk Factors
The epidemiology of SCC developing from chromoblastomycosis is not well understood due to gaps in national and global surveillance. Some studies have found that 2% to 13% of patients with chromoblastomycosis developed SCC.4,11,13-15 Based on case reports and case series, a symptom duration of more than 10 years appears to be the most substantial risk factor for the development of SCC rather than host immune status.16-18 Severity, specifically the size of the injury, and vegetating lesions also have been suggested as risk factors for the development of SCC.16 Additionally, the appearance of new lesions (mainly ulcers not related to secondary infection) that appear during the healing phase should raise the suspicion of SCC and warrant a biopsy for evaluation.16
Pathophysiology
The exact mechanism of malignant transformation has not been elucidated, but histopathologic features suggest substantial epidermal proliferation. In some cases, this leads to pseudoepitheliomatous hyperplasia, a nonmalignant hyperproliferative state that is an important differential HPV to leishmaniasis and lupus vulgaris.19 The chronic inflammation from long-standing chromoblastomycosis likely contributes to the further malignant transformation to SCC.
Polymorphonuclear cells and activated macrophages seen in chronic inflammation can promote the release of enzymes and free radicals that has led to malignant transformation in vitro but has not been investigated specifically in chromoblastomycosis.16 Additionally, chronic inflammation and metabolic products of phagocytosis often are accompanied by excessive production of reactive oxygen and nitrogen species, which can damage DNA, lipoproteins, and cell membranes. Other potential contributors include the expression of cyclooxygenase 2 and release of arachidonic acid metabolites (eg, prostaglandins, leukotrienes), which can damage the cell and promote carcinogenesis. It is not clear whether similar mechanisms account for the development of SCCs in other chronic skin inflammations or infections such as cutaneous tuberculosis or Marjolin ulcers.20
Clinical and Public Health Interventions
Squamous cell carcinoma arising in the setting of chromoblastomycosis warrants prompt oncologic evaluation and definitive surgical management, which may require extensive surgical excision and, in advanced disease, amputation.14,17,18 Advanced malignant tumors can be difficult to manage and can result in death.21,22 Additionally, clinicians should maintain a low threshold for biopsy in long-standing chromoblastomycosis, particularly when lesions demonstrate new ulceration, rapid growth, bleeding, pain, malodor, or failure to improve with appropriate antifungal therapy.16 Recurrent or new lesions after amputation may indicate persistent or recurrent infection and may require continued antifungal management alongside cancer care.16
Squamous cell carcinoma arising from chromoblastomycosis results after substantial diagnostic delays, allowing chronic inflammation to transform infection into malignancy. Separating benign inflammation-associated epidermal proliferation from transformation to SCC requires histopathologic skill. An assay based on increased expression of chromosome 15 open reading frame 48 (C15orf48), an immune regulatory protein, has been developed to aid in this distinction; however, it is not widely available.23
Raising awareness of chromoblastomycosis among clinicians and communities, particularly in rural areas where the disease is more common, is critical to improve health care–seeking behaviors and expedite access to care pathways.2 Furthermore, access and training on microscopy to diagnose chromoblastomycosis in decentralized areas can facilitate earlier diagnosis in primary health care settings rather than waiting for diagnosis in tertiary care settings, at which point disease usually is advanced. Global implementation of existing programs that use microscopy (eg, malaria in rural areas) can be partnered with frontline health worker cross-training on chromoblastomycosis diagnosis to improve appropriate identification of disease.24 Finally, improving access to affordable antifungals, particularly itraconazole, is necessary along with further research into novel therapeutic strategies. Approaches that utilize local manufacturing and pooled procurement could help expand treatment availability in parallel with diagnostic improvement initiatives.25
Final Thoughts
Squamous cell carcinoma resulting from chromoblastomycosis is a devastating complication, often leading to limb amputation. The true prevalence is unknown, but it occurs more commonly in long-standing disease without appropriate therapy. The appearance of new lesions or ulcers after initial improvement should increase suspicion and lead to biopsy and careful pathologic evaluation. Prevention of SCC requires increased clinical awareness, early diagnosis, and timely initiation of antifungal treatment. Enhanced surveillance among individuals with chromoblastomycosis would help to better understand its prevalence, associated risk factors, and impact on quality of life.
- Queiroz-Telles F, de Hoog S, Santos DWCL, et al. Chromoblastomycosis. Clin Microbiol Rev. 2017;30:233-276.
- Smith DJ, Queiroz-Telles F, Rabenja FR, et al. A global chromoblastomycosis strategy and development of the global chromoblastomycosis working group. PLoS Negl Trop Dis. 2024;18:E0012562.
- Yen JS, Shih IH, Chung WH, et al. Chromoblastomycosis in northern Taiwan from 2017 to 2024: unique characteristics. Clin Exp Dermatol. Published online July 18, 2025. doi:10.1093/ced/llaf329
- Santos DWCL, Vicente VA, Weiss VA, et al. Chromoblastomycosis in an endemic area of Brazil: a clinical-epidemiological analysis and a worldwide haplotype network. J Fungi. 2020;6:204.
- Smith DJ, Melhem MSC, Dirven J, et al. Establishment of epidemiological cutoff values for Fonsecaea pedrosoi, the primary etiologic agent of chromoblastomycosis, and eight antifungal medications. J Clin Microbiol. 2025;63:E01903-24.
- Logan C, Singh M, Fox N, et al. Chromoblastomycosis treated with posaconazole and adjunctive imiquimod: lending innate immunity a helping hand. Open Forum Infect Dis. 2023;10:ofad124.
- de Sousa M da GT, Belda W, Spina R, et al. Topical application of imiquimod as a treatment for chromoblastomycosis. Clin Infect Dis. 2014;58:1734-1737.
- Criado PR, Careta MF, Valente NYS, et al. Extensive long-standing chromomycosis due to Fonsecaea pedrosoi: three cases with relevant improvement under voriconazole therapy. J Dermatol Treat. 2011;22:167-174.
- Esterre P, Inzan CK, Ramarcel ER, et al. Treatment of chromomycosis with terbinafine: preliminary results of an open pilot study. Br J Dermatol. 1996;134:33-36.
- Smith DJ, Benedict K, Lockhart SR, et al. Chromoblastomycosis and phaeohyphomycotic abscess-associated hospitalizations, United States, 2016–2021. PLoS Negl Trop Dis. 2025;19:E0013499.
- Bonifaz A, Carrasco‐Gerard E, Saúl A. Chromoblastomycosis: clinical and mycologic experience of 51 cases. Mycoses. 2001;44:1-7.
- Torres E, Beristain JG, Lievanos Z, et al. Chromoblastomycosis associated with a lethal squamous cell carcinoma. An Bras Dermatol. 2010;85:267-270.
- Verma S, Thakur BK, Raphael V, et al. Epidemiology of subcutaneous mycoses in northeast India: a retrospective study. Indian J Dermatol. 2018;63:496-501.
- Siregar GO, Harianja M, Rinonce HT, et al. Chromoblastomycosis: a case series from Sumba, eastern Indonesia. Clin Exp Dermatol. 2025;50:1447-1450.
- Valentin J, Grotta G, Muller T, et al. Chromoblastomycosis in French Guiana: epidemiology and practices, 1955-2023. J Fungi. 2024;10:168.
- Azevedo CMPS, Marques SG, Santos DWCL, et al. Squamous cell carcinoma derived from chronic chromoblastomycosis in Brazil. Clin Infect Dis. 2015;60:1500-1504.
- Belda Jr W, Criado PR, Casteleti P, et al. Chromoblastomycosis evolving to sarcomatoid squamous cell carcinoma: a case report. Dermatol Rep. 2021;13:9009.
- Jamil A, Lee YY, Thevarajah S. Invasive squamous cell carcinoma arising from chromoblastomycosis. Med Mycol. 2012;50:99-102.
- Delahaye T, Orduz-Robledo M, Beltran A M, et al. Pseudo-epitheliomatous hyperplasia and skin infections. Open Dermatol J. 2024;18:E18743722304513.
- Fania L, Didona D, Di Pietro FR, et al. Cutaneous squamous cell carcinoma: from pathophysiology to novel therapeutic approaches. Biomedicines. 2021;9:171.
- Torres E, Beristain JG, Lievanos Z, et al. Carcinoma epidermoide como complicação letal de lesões crônicas de cromoblastomicose. An Bras Dermatol. 2010;85:267-270.
- Rojas OC, González GM, Moreno-Treviño M, et al. Chromoblastomycosis by Cladophialophora carrionii associated with squamous cell carcinoma and review of published reports. Mycopathologia. 2015;179:153-157.
- Su A, Ra S, Li X, et al. Differentiating cutaneous squamous cell carcinoma and pseudoepitheliomatous hyperplasia by multiplex qRT-PCR. Mod Pathol. 2013;26:1433-1437.
- Siregar GO, Harianja M, Smith DJ, et al. Leveraging malaria microscopy infrastructure to diagnose common and neglected skin diseases using direct microscopy in Sumba, Indonesia. Lancet Reg Health - West Pac. 2025;64:101739.
- Smith DJ, Soebono H, Parajuli N, et al. South-East Asia regional neglected tropical disease framework: improving control of mycetoma, chromoblastomycosis, and sporotrichosis. Lancet Reg Health Southeast Asia. 2025;35:100561.
Chromoblastomycosis is a neglected tropical implantation mycosis caused by dematiaceous fungi that leads to substantial morbidity. This condition is diagnosed microscopically by visualizing the characteristic thick-walled, single, or multicellular clusters of pigmented fungal cells (also known as medlar bodies, muriform cells, or sclerotic bodies).1 The main causative fungi varies by geographic region, but most commonly is caused by Cladophialophora carrionii, Fonsecaea species, Phialophora verrucosa species complex, and Rhinocladiella aquaspersa.2-4 Standardized treatment guidelines have not been established, but itraconazole typically is considered first-line regardless of causative fungi.5 Terbinafine, other azoles, and topical immunomodulators, either as monotherapy or in combination, may be appropriate alternative or adjunctive options for refractory disease, although supporting data are limited.6-9
Complications from chromoblastomycosis are common, particularly in long-standing, severe, or refractory disease. An analysis using billing codes in the United States found 14% (35/255) of hospitalized patients with chromoblastomycosis had lymphedema.10 In Mexico, 63% (32/51) of patients with chromoblastomycosis developed secondary bacterial infections.11 Skin fibrosis and ankylosis also can occur and cause mobility issues and decreased quality of life. An infrequent but potentially life-threatening complication12 is the development of squamous cell carcinoma (SCC) associated with chronic lesions, representing a preventable end-stage complication of delayed diagnosis and treatment (Figure).

In this review, we summarize reported epidemiology and clinical risk factors for SCC complicating chromoblastomycosis. We also discuss plausible inflammatory mechanisms of malignant transformation and propose pragmatic clinical and public health interventions, including decentralized microscopy-based diagnosis, timely antifungal access, and biopsy-triggered surveillance of chronically inflamed lesions, to reduce preventable morbidity.
Epidemiology and Risk Factors
The epidemiology of SCC developing from chromoblastomycosis is not well understood due to gaps in national and global surveillance. Some studies have found that 2% to 13% of patients with chromoblastomycosis developed SCC.4,11,13-15 Based on case reports and case series, a symptom duration of more than 10 years appears to be the most substantial risk factor for the development of SCC rather than host immune status.16-18 Severity, specifically the size of the injury, and vegetating lesions also have been suggested as risk factors for the development of SCC.16 Additionally, the appearance of new lesions (mainly ulcers not related to secondary infection) that appear during the healing phase should raise the suspicion of SCC and warrant a biopsy for evaluation.16
Pathophysiology
The exact mechanism of malignant transformation has not been elucidated, but histopathologic features suggest substantial epidermal proliferation. In some cases, this leads to pseudoepitheliomatous hyperplasia, a nonmalignant hyperproliferative state that is an important differential HPV to leishmaniasis and lupus vulgaris.19 The chronic inflammation from long-standing chromoblastomycosis likely contributes to the further malignant transformation to SCC.
Polymorphonuclear cells and activated macrophages seen in chronic inflammation can promote the release of enzymes and free radicals that has led to malignant transformation in vitro but has not been investigated specifically in chromoblastomycosis.16 Additionally, chronic inflammation and metabolic products of phagocytosis often are accompanied by excessive production of reactive oxygen and nitrogen species, which can damage DNA, lipoproteins, and cell membranes. Other potential contributors include the expression of cyclooxygenase 2 and release of arachidonic acid metabolites (eg, prostaglandins, leukotrienes), which can damage the cell and promote carcinogenesis. It is not clear whether similar mechanisms account for the development of SCCs in other chronic skin inflammations or infections such as cutaneous tuberculosis or Marjolin ulcers.20
Clinical and Public Health Interventions
Squamous cell carcinoma arising in the setting of chromoblastomycosis warrants prompt oncologic evaluation and definitive surgical management, which may require extensive surgical excision and, in advanced disease, amputation.14,17,18 Advanced malignant tumors can be difficult to manage and can result in death.21,22 Additionally, clinicians should maintain a low threshold for biopsy in long-standing chromoblastomycosis, particularly when lesions demonstrate new ulceration, rapid growth, bleeding, pain, malodor, or failure to improve with appropriate antifungal therapy.16 Recurrent or new lesions after amputation may indicate persistent or recurrent infection and may require continued antifungal management alongside cancer care.16
Squamous cell carcinoma arising from chromoblastomycosis results after substantial diagnostic delays, allowing chronic inflammation to transform infection into malignancy. Separating benign inflammation-associated epidermal proliferation from transformation to SCC requires histopathologic skill. An assay based on increased expression of chromosome 15 open reading frame 48 (C15orf48), an immune regulatory protein, has been developed to aid in this distinction; however, it is not widely available.23
Raising awareness of chromoblastomycosis among clinicians and communities, particularly in rural areas where the disease is more common, is critical to improve health care–seeking behaviors and expedite access to care pathways.2 Furthermore, access and training on microscopy to diagnose chromoblastomycosis in decentralized areas can facilitate earlier diagnosis in primary health care settings rather than waiting for diagnosis in tertiary care settings, at which point disease usually is advanced. Global implementation of existing programs that use microscopy (eg, malaria in rural areas) can be partnered with frontline health worker cross-training on chromoblastomycosis diagnosis to improve appropriate identification of disease.24 Finally, improving access to affordable antifungals, particularly itraconazole, is necessary along with further research into novel therapeutic strategies. Approaches that utilize local manufacturing and pooled procurement could help expand treatment availability in parallel with diagnostic improvement initiatives.25
Final Thoughts
Squamous cell carcinoma resulting from chromoblastomycosis is a devastating complication, often leading to limb amputation. The true prevalence is unknown, but it occurs more commonly in long-standing disease without appropriate therapy. The appearance of new lesions or ulcers after initial improvement should increase suspicion and lead to biopsy and careful pathologic evaluation. Prevention of SCC requires increased clinical awareness, early diagnosis, and timely initiation of antifungal treatment. Enhanced surveillance among individuals with chromoblastomycosis would help to better understand its prevalence, associated risk factors, and impact on quality of life.
Chromoblastomycosis is a neglected tropical implantation mycosis caused by dematiaceous fungi that leads to substantial morbidity. This condition is diagnosed microscopically by visualizing the characteristic thick-walled, single, or multicellular clusters of pigmented fungal cells (also known as medlar bodies, muriform cells, or sclerotic bodies).1 The main causative fungi varies by geographic region, but most commonly is caused by Cladophialophora carrionii, Fonsecaea species, Phialophora verrucosa species complex, and Rhinocladiella aquaspersa.2-4 Standardized treatment guidelines have not been established, but itraconazole typically is considered first-line regardless of causative fungi.5 Terbinafine, other azoles, and topical immunomodulators, either as monotherapy or in combination, may be appropriate alternative or adjunctive options for refractory disease, although supporting data are limited.6-9
Complications from chromoblastomycosis are common, particularly in long-standing, severe, or refractory disease. An analysis using billing codes in the United States found 14% (35/255) of hospitalized patients with chromoblastomycosis had lymphedema.10 In Mexico, 63% (32/51) of patients with chromoblastomycosis developed secondary bacterial infections.11 Skin fibrosis and ankylosis also can occur and cause mobility issues and decreased quality of life. An infrequent but potentially life-threatening complication12 is the development of squamous cell carcinoma (SCC) associated with chronic lesions, representing a preventable end-stage complication of delayed diagnosis and treatment (Figure).

In this review, we summarize reported epidemiology and clinical risk factors for SCC complicating chromoblastomycosis. We also discuss plausible inflammatory mechanisms of malignant transformation and propose pragmatic clinical and public health interventions, including decentralized microscopy-based diagnosis, timely antifungal access, and biopsy-triggered surveillance of chronically inflamed lesions, to reduce preventable morbidity.
Epidemiology and Risk Factors
The epidemiology of SCC developing from chromoblastomycosis is not well understood due to gaps in national and global surveillance. Some studies have found that 2% to 13% of patients with chromoblastomycosis developed SCC.4,11,13-15 Based on case reports and case series, a symptom duration of more than 10 years appears to be the most substantial risk factor for the development of SCC rather than host immune status.16-18 Severity, specifically the size of the injury, and vegetating lesions also have been suggested as risk factors for the development of SCC.16 Additionally, the appearance of new lesions (mainly ulcers not related to secondary infection) that appear during the healing phase should raise the suspicion of SCC and warrant a biopsy for evaluation.16
Pathophysiology
The exact mechanism of malignant transformation has not been elucidated, but histopathologic features suggest substantial epidermal proliferation. In some cases, this leads to pseudoepitheliomatous hyperplasia, a nonmalignant hyperproliferative state that is an important differential HPV to leishmaniasis and lupus vulgaris.19 The chronic inflammation from long-standing chromoblastomycosis likely contributes to the further malignant transformation to SCC.
Polymorphonuclear cells and activated macrophages seen in chronic inflammation can promote the release of enzymes and free radicals that has led to malignant transformation in vitro but has not been investigated specifically in chromoblastomycosis.16 Additionally, chronic inflammation and metabolic products of phagocytosis often are accompanied by excessive production of reactive oxygen and nitrogen species, which can damage DNA, lipoproteins, and cell membranes. Other potential contributors include the expression of cyclooxygenase 2 and release of arachidonic acid metabolites (eg, prostaglandins, leukotrienes), which can damage the cell and promote carcinogenesis. It is not clear whether similar mechanisms account for the development of SCCs in other chronic skin inflammations or infections such as cutaneous tuberculosis or Marjolin ulcers.20
Clinical and Public Health Interventions
Squamous cell carcinoma arising in the setting of chromoblastomycosis warrants prompt oncologic evaluation and definitive surgical management, which may require extensive surgical excision and, in advanced disease, amputation.14,17,18 Advanced malignant tumors can be difficult to manage and can result in death.21,22 Additionally, clinicians should maintain a low threshold for biopsy in long-standing chromoblastomycosis, particularly when lesions demonstrate new ulceration, rapid growth, bleeding, pain, malodor, or failure to improve with appropriate antifungal therapy.16 Recurrent or new lesions after amputation may indicate persistent or recurrent infection and may require continued antifungal management alongside cancer care.16
Squamous cell carcinoma arising from chromoblastomycosis results after substantial diagnostic delays, allowing chronic inflammation to transform infection into malignancy. Separating benign inflammation-associated epidermal proliferation from transformation to SCC requires histopathologic skill. An assay based on increased expression of chromosome 15 open reading frame 48 (C15orf48), an immune regulatory protein, has been developed to aid in this distinction; however, it is not widely available.23
Raising awareness of chromoblastomycosis among clinicians and communities, particularly in rural areas where the disease is more common, is critical to improve health care–seeking behaviors and expedite access to care pathways.2 Furthermore, access and training on microscopy to diagnose chromoblastomycosis in decentralized areas can facilitate earlier diagnosis in primary health care settings rather than waiting for diagnosis in tertiary care settings, at which point disease usually is advanced. Global implementation of existing programs that use microscopy (eg, malaria in rural areas) can be partnered with frontline health worker cross-training on chromoblastomycosis diagnosis to improve appropriate identification of disease.24 Finally, improving access to affordable antifungals, particularly itraconazole, is necessary along with further research into novel therapeutic strategies. Approaches that utilize local manufacturing and pooled procurement could help expand treatment availability in parallel with diagnostic improvement initiatives.25
Final Thoughts
Squamous cell carcinoma resulting from chromoblastomycosis is a devastating complication, often leading to limb amputation. The true prevalence is unknown, but it occurs more commonly in long-standing disease without appropriate therapy. The appearance of new lesions or ulcers after initial improvement should increase suspicion and lead to biopsy and careful pathologic evaluation. Prevention of SCC requires increased clinical awareness, early diagnosis, and timely initiation of antifungal treatment. Enhanced surveillance among individuals with chromoblastomycosis would help to better understand its prevalence, associated risk factors, and impact on quality of life.
- Queiroz-Telles F, de Hoog S, Santos DWCL, et al. Chromoblastomycosis. Clin Microbiol Rev. 2017;30:233-276.
- Smith DJ, Queiroz-Telles F, Rabenja FR, et al. A global chromoblastomycosis strategy and development of the global chromoblastomycosis working group. PLoS Negl Trop Dis. 2024;18:E0012562.
- Yen JS, Shih IH, Chung WH, et al. Chromoblastomycosis in northern Taiwan from 2017 to 2024: unique characteristics. Clin Exp Dermatol. Published online July 18, 2025. doi:10.1093/ced/llaf329
- Santos DWCL, Vicente VA, Weiss VA, et al. Chromoblastomycosis in an endemic area of Brazil: a clinical-epidemiological analysis and a worldwide haplotype network. J Fungi. 2020;6:204.
- Smith DJ, Melhem MSC, Dirven J, et al. Establishment of epidemiological cutoff values for Fonsecaea pedrosoi, the primary etiologic agent of chromoblastomycosis, and eight antifungal medications. J Clin Microbiol. 2025;63:E01903-24.
- Logan C, Singh M, Fox N, et al. Chromoblastomycosis treated with posaconazole and adjunctive imiquimod: lending innate immunity a helping hand. Open Forum Infect Dis. 2023;10:ofad124.
- de Sousa M da GT, Belda W, Spina R, et al. Topical application of imiquimod as a treatment for chromoblastomycosis. Clin Infect Dis. 2014;58:1734-1737.
- Criado PR, Careta MF, Valente NYS, et al. Extensive long-standing chromomycosis due to Fonsecaea pedrosoi: three cases with relevant improvement under voriconazole therapy. J Dermatol Treat. 2011;22:167-174.
- Esterre P, Inzan CK, Ramarcel ER, et al. Treatment of chromomycosis with terbinafine: preliminary results of an open pilot study. Br J Dermatol. 1996;134:33-36.
- Smith DJ, Benedict K, Lockhart SR, et al. Chromoblastomycosis and phaeohyphomycotic abscess-associated hospitalizations, United States, 2016–2021. PLoS Negl Trop Dis. 2025;19:E0013499.
- Bonifaz A, Carrasco‐Gerard E, Saúl A. Chromoblastomycosis: clinical and mycologic experience of 51 cases. Mycoses. 2001;44:1-7.
- Torres E, Beristain JG, Lievanos Z, et al. Chromoblastomycosis associated with a lethal squamous cell carcinoma. An Bras Dermatol. 2010;85:267-270.
- Verma S, Thakur BK, Raphael V, et al. Epidemiology of subcutaneous mycoses in northeast India: a retrospective study. Indian J Dermatol. 2018;63:496-501.
- Siregar GO, Harianja M, Rinonce HT, et al. Chromoblastomycosis: a case series from Sumba, eastern Indonesia. Clin Exp Dermatol. 2025;50:1447-1450.
- Valentin J, Grotta G, Muller T, et al. Chromoblastomycosis in French Guiana: epidemiology and practices, 1955-2023. J Fungi. 2024;10:168.
- Azevedo CMPS, Marques SG, Santos DWCL, et al. Squamous cell carcinoma derived from chronic chromoblastomycosis in Brazil. Clin Infect Dis. 2015;60:1500-1504.
- Belda Jr W, Criado PR, Casteleti P, et al. Chromoblastomycosis evolving to sarcomatoid squamous cell carcinoma: a case report. Dermatol Rep. 2021;13:9009.
- Jamil A, Lee YY, Thevarajah S. Invasive squamous cell carcinoma arising from chromoblastomycosis. Med Mycol. 2012;50:99-102.
- Delahaye T, Orduz-Robledo M, Beltran A M, et al. Pseudo-epitheliomatous hyperplasia and skin infections. Open Dermatol J. 2024;18:E18743722304513.
- Fania L, Didona D, Di Pietro FR, et al. Cutaneous squamous cell carcinoma: from pathophysiology to novel therapeutic approaches. Biomedicines. 2021;9:171.
- Torres E, Beristain JG, Lievanos Z, et al. Carcinoma epidermoide como complicação letal de lesões crônicas de cromoblastomicose. An Bras Dermatol. 2010;85:267-270.
- Rojas OC, González GM, Moreno-Treviño M, et al. Chromoblastomycosis by Cladophialophora carrionii associated with squamous cell carcinoma and review of published reports. Mycopathologia. 2015;179:153-157.
- Su A, Ra S, Li X, et al. Differentiating cutaneous squamous cell carcinoma and pseudoepitheliomatous hyperplasia by multiplex qRT-PCR. Mod Pathol. 2013;26:1433-1437.
- Siregar GO, Harianja M, Smith DJ, et al. Leveraging malaria microscopy infrastructure to diagnose common and neglected skin diseases using direct microscopy in Sumba, Indonesia. Lancet Reg Health - West Pac. 2025;64:101739.
- Smith DJ, Soebono H, Parajuli N, et al. South-East Asia regional neglected tropical disease framework: improving control of mycetoma, chromoblastomycosis, and sporotrichosis. Lancet Reg Health Southeast Asia. 2025;35:100561.
- Queiroz-Telles F, de Hoog S, Santos DWCL, et al. Chromoblastomycosis. Clin Microbiol Rev. 2017;30:233-276.
- Smith DJ, Queiroz-Telles F, Rabenja FR, et al. A global chromoblastomycosis strategy and development of the global chromoblastomycosis working group. PLoS Negl Trop Dis. 2024;18:E0012562.
- Yen JS, Shih IH, Chung WH, et al. Chromoblastomycosis in northern Taiwan from 2017 to 2024: unique characteristics. Clin Exp Dermatol. Published online July 18, 2025. doi:10.1093/ced/llaf329
- Santos DWCL, Vicente VA, Weiss VA, et al. Chromoblastomycosis in an endemic area of Brazil: a clinical-epidemiological analysis and a worldwide haplotype network. J Fungi. 2020;6:204.
- Smith DJ, Melhem MSC, Dirven J, et al. Establishment of epidemiological cutoff values for Fonsecaea pedrosoi, the primary etiologic agent of chromoblastomycosis, and eight antifungal medications. J Clin Microbiol. 2025;63:E01903-24.
- Logan C, Singh M, Fox N, et al. Chromoblastomycosis treated with posaconazole and adjunctive imiquimod: lending innate immunity a helping hand. Open Forum Infect Dis. 2023;10:ofad124.
- de Sousa M da GT, Belda W, Spina R, et al. Topical application of imiquimod as a treatment for chromoblastomycosis. Clin Infect Dis. 2014;58:1734-1737.
- Criado PR, Careta MF, Valente NYS, et al. Extensive long-standing chromomycosis due to Fonsecaea pedrosoi: three cases with relevant improvement under voriconazole therapy. J Dermatol Treat. 2011;22:167-174.
- Esterre P, Inzan CK, Ramarcel ER, et al. Treatment of chromomycosis with terbinafine: preliminary results of an open pilot study. Br J Dermatol. 1996;134:33-36.
- Smith DJ, Benedict K, Lockhart SR, et al. Chromoblastomycosis and phaeohyphomycotic abscess-associated hospitalizations, United States, 2016–2021. PLoS Negl Trop Dis. 2025;19:E0013499.
- Bonifaz A, Carrasco‐Gerard E, Saúl A. Chromoblastomycosis: clinical and mycologic experience of 51 cases. Mycoses. 2001;44:1-7.
- Torres E, Beristain JG, Lievanos Z, et al. Chromoblastomycosis associated with a lethal squamous cell carcinoma. An Bras Dermatol. 2010;85:267-270.
- Verma S, Thakur BK, Raphael V, et al. Epidemiology of subcutaneous mycoses in northeast India: a retrospective study. Indian J Dermatol. 2018;63:496-501.
- Siregar GO, Harianja M, Rinonce HT, et al. Chromoblastomycosis: a case series from Sumba, eastern Indonesia. Clin Exp Dermatol. 2025;50:1447-1450.
- Valentin J, Grotta G, Muller T, et al. Chromoblastomycosis in French Guiana: epidemiology and practices, 1955-2023. J Fungi. 2024;10:168.
- Azevedo CMPS, Marques SG, Santos DWCL, et al. Squamous cell carcinoma derived from chronic chromoblastomycosis in Brazil. Clin Infect Dis. 2015;60:1500-1504.
- Belda Jr W, Criado PR, Casteleti P, et al. Chromoblastomycosis evolving to sarcomatoid squamous cell carcinoma: a case report. Dermatol Rep. 2021;13:9009.
- Jamil A, Lee YY, Thevarajah S. Invasive squamous cell carcinoma arising from chromoblastomycosis. Med Mycol. 2012;50:99-102.
- Delahaye T, Orduz-Robledo M, Beltran A M, et al. Pseudo-epitheliomatous hyperplasia and skin infections. Open Dermatol J. 2024;18:E18743722304513.
- Fania L, Didona D, Di Pietro FR, et al. Cutaneous squamous cell carcinoma: from pathophysiology to novel therapeutic approaches. Biomedicines. 2021;9:171.
- Torres E, Beristain JG, Lievanos Z, et al. Carcinoma epidermoide como complicação letal de lesões crônicas de cromoblastomicose. An Bras Dermatol. 2010;85:267-270.
- Rojas OC, González GM, Moreno-Treviño M, et al. Chromoblastomycosis by Cladophialophora carrionii associated with squamous cell carcinoma and review of published reports. Mycopathologia. 2015;179:153-157.
- Su A, Ra S, Li X, et al. Differentiating cutaneous squamous cell carcinoma and pseudoepitheliomatous hyperplasia by multiplex qRT-PCR. Mod Pathol. 2013;26:1433-1437.
- Siregar GO, Harianja M, Smith DJ, et al. Leveraging malaria microscopy infrastructure to diagnose common and neglected skin diseases using direct microscopy in Sumba, Indonesia. Lancet Reg Health - West Pac. 2025;64:101739.
- Smith DJ, Soebono H, Parajuli N, et al. South-East Asia regional neglected tropical disease framework: improving control of mycetoma, chromoblastomycosis, and sporotrichosis. Lancet Reg Health Southeast Asia. 2025;35:100561.
Chromoblastomycosis Leading to Squamous Cell Carcinoma: An Overlooked Outcome of a Neglected Tropical Disease
Chromoblastomycosis Leading to Squamous Cell Carcinoma: An Overlooked Outcome of a Neglected Tropical Disease
Practice Points
- Chromoblastomycosis is recognized by the World Health Organization as a neglected tropical disease and principally affects agricultural workers in tropical and subtropical regions.
- Long-standing or refractory chromoblastomycosis can lead to substantial morbidity, including lymphedema, secondary bacterial infections, extensive scarring, functional impairment, and squamous cell carcinoma (SCC).
- The development of SCC is thought to be related to chronic inflammation and prolonged disease duration (Mathematical Pi LT Std>10 years). Advanced cases may require extensive surgical excision or amputation.
- Early recognition with support of direct microscopy or histopathology, timely antifungal treatment (often with itraconazole), and a low threshold for repeat biopsy of new ulceration or rapidly changing lesions may prevent malignant transformation and disability.
Enlarging Mass on the Scalp
Enlarging Mass on the Scalp
THE DIAGNOSIS: Malignant Proliferating Trichilemmal Tumor
Histologic examination revealed atypical keratinocytes, nuclear pleomorphism, and lobulating epithelial masses with trichilemmal keratinization (Figure). The presence of CD34 positivity, a marker of outer follicular root sheath–derived cells, supported the diagnosis of a malignant proliferating trichilemmal tumor (MPTT). Imaging also revealed signs of bone invasion, further supporting a malignant process. Based on these findings, the patient underwent complete excision of the mass with scalp reconstruction, lymph node dissection, and systemic evaluation for metastases. Final pathology confirmed negative surgical margins and no lymph node involvement. Adjuvant radiation was not required, given the absence of skull invasion or confirmed distant metastasis.
The differential diagnosis for rapidly enlarging scalp tumors can be broad and includes both benign and malignant processes. In this patient, the differential diagnoses included trichilemmal carcinoma, cutaneous squamous cell carcinoma (SCC), sebaceous carcinoma (SC), proliferating trichilemmal tumor (PTT), and MPTT. Due to the notable clinical and histologic overlap among these lesions, definitive diagnosis required histopathologic evaluation in our patient.
Proliferating trichilemmal tumors were first described in 1966 by Wilson-Jones,1 who used the term proliferating epidermoid cysts, noting their distinct histologic features and resemblance to SCC.2 These tumors generally are benign and arise from the isthmus of the outer root sheath of the hair follicle; however, malignant transformation can occur, resulting in a rare entity known as MPTT. This malignant variant was first described in 1983 by Saida et al,3 who emphasized its distinct clinical behavior, including infiltrative growth, high mitotic activity, and potential for local recurrence and metastasis.
A recent literature review identified 60 reported cases of MPTT, with an average patient age of 57 years and a female predominance.4 Clinically, MPTTs often manifest as large (>5 cm) lobulated masses located on sun-exposed, hair-bearing areas of the skin, especially the scalp. These lesions may be flesh-colored to pink and often exhibit ulceration, necrosis, or calcification.5 Typically, MPTTs follow a biphasic course, beginning with a slow-growing phase followed by a period of rapid growth. Due to their aggressive behavior and resemblance to other cutaneous malignancies, accurate differentiation of MPTT from benign PTTs, cutaneous SCCs, SCs, and trichilemmal carcinomas is critical.
Malignant proliferating trichilemmal tumors demonstrate a substantially higher metastatic potential than either benign PTTs or cutaneous SCCs. While cutaneous SCCs carry a metastasis rate of approximately 1.9% to 2.6%, MPTTs carry a considerably higher rate of approximately 25.0%.6 Regional lymphatic spread is the most common route of dissemination, making comprehensive lymph node assessment—both radiographic and clinical—an important component of tumor staging. When lymph node involvement is suspected, surgical dissection may be indicated, along with consideration of adjuvant therapies.
Histopathologically, MPTT is characterized by nuclear atypia, mitotic figures, and lobulated masses of proliferating epithelium showing trichilemmal differentiation and infiltrative growth.4 The presence of CD34 positivity, reflecting outer follicular root sheath differentiation, helps distinguish MPTT from cutaneous SCC and SC, which typically lack this marker.6,7 Immunohistochemistry is therefore a valuable adjunct in differentiating these lesions.
The mainstay of treatment for MPTT is wide local excision with clear margins. Margins of at least 1 cm generally are recommended. Although Mohs micrographic surgery may be used in anatomically sensitive areas, it typically is not preferred due to the potential for skip lesions in MPTT, which may lead to incomplete excision and recurrence.8 In cases with evidence of regional or distant metastasis or when clear margins cannot be achieved confidently, adjunctive treatments such as radiation therapy and systemic chemotherapy may be indicated. Preoperative imaging is used to evaluate for local invasion (skull or bone involvement) and regional lymph node status, which may inform adjuvant therapy postoperatively.
The prognosis for MPTT is variable and depends largely on early recognition, accurate histopathologic diagnosis, complete surgical excision with clear margins, and the presence or absence of metastasis. When the tumor is fully excised with negative margins and no lymph node involvement, the risk for recurrence is substantially reduced; however, MPTT is known for its potential aggressive behavior. Delays in diagnosis or incomplete resection can lead to local recurrence, regional spread, or even distant metastasis. In the literature review discussed previously, the mortality rate of patients with MPTT was 11.7%,4 which is notably higher than that of more common cutaneous malignancies such as cutaneous SCC, which is reported at 1.2%.9
The clinical course of MPTT remains difficult to predict due to its rarity and the limited availability of large-scale studies. Most published data are derived from isolated case reports or small case series, making standardized treatment guidelines challenging. Given this uncertainty, long-term follow-up is strongly recommended to monitor for recurrence or metastatic progression.2
This case highlights the critical role of clinicopathologic correlation in the evaluation of atypical or rapidly growing scalp lesions. The expertise of dermatologists in recognizing atypical presentations, combined with precise histopathologic analysis, including immunohistochemical staining, is vital to ensuring accurate diagnosis and optimal treatment. Early intervention can improve patient outcomes by reducing the risk for local recurrence and metastatic progression as well as the need for more intensive therapies.
- Jones EW. Proliferating epidermoid cysts. Arch Dermatol. 1966;94:11-19.
- Kemaloglu CA, Öztürk M, Aydın B, et al. Malignant proliferating trichilemmal tumor of the scalp: report of 4 cases and a short review of the literature. Case Reports Plast Surg Hand Surg. 2022;9:158-164. doi:10.1080/23320885.2022.2077208
- Saida T, Oohard K, Hori Y, et al. Development of a malignant proliferating trichilemmal cyst in a patient with multiple trichilemmal cysts. Dermatology. 1983;166:203-208. doi:10.1159/000249868
- Abdelhammed MH, Siatecka H, Diwan AH, et al. A rare case of a malignant proliferating trichilemmal tumor: a molecular study harboring potential therapeutic significance and a review of literature. Dermatopathology (Basel). 2024;11:354-363. doi:10.3390 /dermatopathology11040038
- Fronek L, Brahs A, Farsi M, et al. A rare case of trichilemmal carcinoma: histology and management. J Clin Aesthet Dermatol. 2021;14:25-30.
- Osto M, Parry N, Rehman R, et al. Malignant proliferating trichilemmal tumor of the scalp: a systematic review. Am J Dermatopathol. 2021;43:851-866. doi:10.1097/DAD.0000000000001991
- Plaza JA, Mackinnon A, Carrillo L, et al. Role of immunohistochemistry in the diagnosis of sebaceous carcinoma: a clinicopathologic and immunohistochemical study. Am J Dermatopathol. 2015;37:809-821. doi:10.1097/DAD.0000000000000255
- Singh P, Usman A, Motta L, et al. Malignant proliferating trichilemmal tumour. BMJ Case Rep. Published online August 17, 2018. doi:10.1136/bcr-2018-224460
- Ran NA, Granger EE, Brodland DG, et al. Risk factor number and recurrence, metastasis, and disease-related death in cutaneous squamous cell carcinoma. JAMA Dermatol. 2025;161:597-604. doi:10.1001/jamadermatol.2025.0128
THE DIAGNOSIS: Malignant Proliferating Trichilemmal Tumor
Histologic examination revealed atypical keratinocytes, nuclear pleomorphism, and lobulating epithelial masses with trichilemmal keratinization (Figure). The presence of CD34 positivity, a marker of outer follicular root sheath–derived cells, supported the diagnosis of a malignant proliferating trichilemmal tumor (MPTT). Imaging also revealed signs of bone invasion, further supporting a malignant process. Based on these findings, the patient underwent complete excision of the mass with scalp reconstruction, lymph node dissection, and systemic evaluation for metastases. Final pathology confirmed negative surgical margins and no lymph node involvement. Adjuvant radiation was not required, given the absence of skull invasion or confirmed distant metastasis.

The differential diagnosis for rapidly enlarging scalp tumors can be broad and includes both benign and malignant processes. In this patient, the differential diagnoses included trichilemmal carcinoma, cutaneous squamous cell carcinoma (SCC), sebaceous carcinoma (SC), proliferating trichilemmal tumor (PTT), and MPTT. Due to the notable clinical and histologic overlap among these lesions, definitive diagnosis required histopathologic evaluation in our patient.
Proliferating trichilemmal tumors were first described in 1966 by Wilson-Jones,1 who used the term proliferating epidermoid cysts, noting their distinct histologic features and resemblance to SCC.2 These tumors generally are benign and arise from the isthmus of the outer root sheath of the hair follicle; however, malignant transformation can occur, resulting in a rare entity known as MPTT. This malignant variant was first described in 1983 by Saida et al,3 who emphasized its distinct clinical behavior, including infiltrative growth, high mitotic activity, and potential for local recurrence and metastasis.
A recent literature review identified 60 reported cases of MPTT, with an average patient age of 57 years and a female predominance.4 Clinically, MPTTs often manifest as large (>5 cm) lobulated masses located on sun-exposed, hair-bearing areas of the skin, especially the scalp. These lesions may be flesh-colored to pink and often exhibit ulceration, necrosis, or calcification.5 Typically, MPTTs follow a biphasic course, beginning with a slow-growing phase followed by a period of rapid growth. Due to their aggressive behavior and resemblance to other cutaneous malignancies, accurate differentiation of MPTT from benign PTTs, cutaneous SCCs, SCs, and trichilemmal carcinomas is critical.
Malignant proliferating trichilemmal tumors demonstrate a substantially higher metastatic potential than either benign PTTs or cutaneous SCCs. While cutaneous SCCs carry a metastasis rate of approximately 1.9% to 2.6%, MPTTs carry a considerably higher rate of approximately 25.0%.6 Regional lymphatic spread is the most common route of dissemination, making comprehensive lymph node assessment—both radiographic and clinical—an important component of tumor staging. When lymph node involvement is suspected, surgical dissection may be indicated, along with consideration of adjuvant therapies.
Histopathologically, MPTT is characterized by nuclear atypia, mitotic figures, and lobulated masses of proliferating epithelium showing trichilemmal differentiation and infiltrative growth.4 The presence of CD34 positivity, reflecting outer follicular root sheath differentiation, helps distinguish MPTT from cutaneous SCC and SC, which typically lack this marker.6,7 Immunohistochemistry is therefore a valuable adjunct in differentiating these lesions.
The mainstay of treatment for MPTT is wide local excision with clear margins. Margins of at least 1 cm generally are recommended. Although Mohs micrographic surgery may be used in anatomically sensitive areas, it typically is not preferred due to the potential for skip lesions in MPTT, which may lead to incomplete excision and recurrence.8 In cases with evidence of regional or distant metastasis or when clear margins cannot be achieved confidently, adjunctive treatments such as radiation therapy and systemic chemotherapy may be indicated. Preoperative imaging is used to evaluate for local invasion (skull or bone involvement) and regional lymph node status, which may inform adjuvant therapy postoperatively.
The prognosis for MPTT is variable and depends largely on early recognition, accurate histopathologic diagnosis, complete surgical excision with clear margins, and the presence or absence of metastasis. When the tumor is fully excised with negative margins and no lymph node involvement, the risk for recurrence is substantially reduced; however, MPTT is known for its potential aggressive behavior. Delays in diagnosis or incomplete resection can lead to local recurrence, regional spread, or even distant metastasis. In the literature review discussed previously, the mortality rate of patients with MPTT was 11.7%,4 which is notably higher than that of more common cutaneous malignancies such as cutaneous SCC, which is reported at 1.2%.9
The clinical course of MPTT remains difficult to predict due to its rarity and the limited availability of large-scale studies. Most published data are derived from isolated case reports or small case series, making standardized treatment guidelines challenging. Given this uncertainty, long-term follow-up is strongly recommended to monitor for recurrence or metastatic progression.2
This case highlights the critical role of clinicopathologic correlation in the evaluation of atypical or rapidly growing scalp lesions. The expertise of dermatologists in recognizing atypical presentations, combined with precise histopathologic analysis, including immunohistochemical staining, is vital to ensuring accurate diagnosis and optimal treatment. Early intervention can improve patient outcomes by reducing the risk for local recurrence and metastatic progression as well as the need for more intensive therapies.
THE DIAGNOSIS: Malignant Proliferating Trichilemmal Tumor
Histologic examination revealed atypical keratinocytes, nuclear pleomorphism, and lobulating epithelial masses with trichilemmal keratinization (Figure). The presence of CD34 positivity, a marker of outer follicular root sheath–derived cells, supported the diagnosis of a malignant proliferating trichilemmal tumor (MPTT). Imaging also revealed signs of bone invasion, further supporting a malignant process. Based on these findings, the patient underwent complete excision of the mass with scalp reconstruction, lymph node dissection, and systemic evaluation for metastases. Final pathology confirmed negative surgical margins and no lymph node involvement. Adjuvant radiation was not required, given the absence of skull invasion or confirmed distant metastasis.

The differential diagnosis for rapidly enlarging scalp tumors can be broad and includes both benign and malignant processes. In this patient, the differential diagnoses included trichilemmal carcinoma, cutaneous squamous cell carcinoma (SCC), sebaceous carcinoma (SC), proliferating trichilemmal tumor (PTT), and MPTT. Due to the notable clinical and histologic overlap among these lesions, definitive diagnosis required histopathologic evaluation in our patient.
Proliferating trichilemmal tumors were first described in 1966 by Wilson-Jones,1 who used the term proliferating epidermoid cysts, noting their distinct histologic features and resemblance to SCC.2 These tumors generally are benign and arise from the isthmus of the outer root sheath of the hair follicle; however, malignant transformation can occur, resulting in a rare entity known as MPTT. This malignant variant was first described in 1983 by Saida et al,3 who emphasized its distinct clinical behavior, including infiltrative growth, high mitotic activity, and potential for local recurrence and metastasis.
A recent literature review identified 60 reported cases of MPTT, with an average patient age of 57 years and a female predominance.4 Clinically, MPTTs often manifest as large (>5 cm) lobulated masses located on sun-exposed, hair-bearing areas of the skin, especially the scalp. These lesions may be flesh-colored to pink and often exhibit ulceration, necrosis, or calcification.5 Typically, MPTTs follow a biphasic course, beginning with a slow-growing phase followed by a period of rapid growth. Due to their aggressive behavior and resemblance to other cutaneous malignancies, accurate differentiation of MPTT from benign PTTs, cutaneous SCCs, SCs, and trichilemmal carcinomas is critical.
Malignant proliferating trichilemmal tumors demonstrate a substantially higher metastatic potential than either benign PTTs or cutaneous SCCs. While cutaneous SCCs carry a metastasis rate of approximately 1.9% to 2.6%, MPTTs carry a considerably higher rate of approximately 25.0%.6 Regional lymphatic spread is the most common route of dissemination, making comprehensive lymph node assessment—both radiographic and clinical—an important component of tumor staging. When lymph node involvement is suspected, surgical dissection may be indicated, along with consideration of adjuvant therapies.
Histopathologically, MPTT is characterized by nuclear atypia, mitotic figures, and lobulated masses of proliferating epithelium showing trichilemmal differentiation and infiltrative growth.4 The presence of CD34 positivity, reflecting outer follicular root sheath differentiation, helps distinguish MPTT from cutaneous SCC and SC, which typically lack this marker.6,7 Immunohistochemistry is therefore a valuable adjunct in differentiating these lesions.
The mainstay of treatment for MPTT is wide local excision with clear margins. Margins of at least 1 cm generally are recommended. Although Mohs micrographic surgery may be used in anatomically sensitive areas, it typically is not preferred due to the potential for skip lesions in MPTT, which may lead to incomplete excision and recurrence.8 In cases with evidence of regional or distant metastasis or when clear margins cannot be achieved confidently, adjunctive treatments such as radiation therapy and systemic chemotherapy may be indicated. Preoperative imaging is used to evaluate for local invasion (skull or bone involvement) and regional lymph node status, which may inform adjuvant therapy postoperatively.
The prognosis for MPTT is variable and depends largely on early recognition, accurate histopathologic diagnosis, complete surgical excision with clear margins, and the presence or absence of metastasis. When the tumor is fully excised with negative margins and no lymph node involvement, the risk for recurrence is substantially reduced; however, MPTT is known for its potential aggressive behavior. Delays in diagnosis or incomplete resection can lead to local recurrence, regional spread, or even distant metastasis. In the literature review discussed previously, the mortality rate of patients with MPTT was 11.7%,4 which is notably higher than that of more common cutaneous malignancies such as cutaneous SCC, which is reported at 1.2%.9
The clinical course of MPTT remains difficult to predict due to its rarity and the limited availability of large-scale studies. Most published data are derived from isolated case reports or small case series, making standardized treatment guidelines challenging. Given this uncertainty, long-term follow-up is strongly recommended to monitor for recurrence or metastatic progression.2
This case highlights the critical role of clinicopathologic correlation in the evaluation of atypical or rapidly growing scalp lesions. The expertise of dermatologists in recognizing atypical presentations, combined with precise histopathologic analysis, including immunohistochemical staining, is vital to ensuring accurate diagnosis and optimal treatment. Early intervention can improve patient outcomes by reducing the risk for local recurrence and metastatic progression as well as the need for more intensive therapies.
- Jones EW. Proliferating epidermoid cysts. Arch Dermatol. 1966;94:11-19.
- Kemaloglu CA, Öztürk M, Aydın B, et al. Malignant proliferating trichilemmal tumor of the scalp: report of 4 cases and a short review of the literature. Case Reports Plast Surg Hand Surg. 2022;9:158-164. doi:10.1080/23320885.2022.2077208
- Saida T, Oohard K, Hori Y, et al. Development of a malignant proliferating trichilemmal cyst in a patient with multiple trichilemmal cysts. Dermatology. 1983;166:203-208. doi:10.1159/000249868
- Abdelhammed MH, Siatecka H, Diwan AH, et al. A rare case of a malignant proliferating trichilemmal tumor: a molecular study harboring potential therapeutic significance and a review of literature. Dermatopathology (Basel). 2024;11:354-363. doi:10.3390 /dermatopathology11040038
- Fronek L, Brahs A, Farsi M, et al. A rare case of trichilemmal carcinoma: histology and management. J Clin Aesthet Dermatol. 2021;14:25-30.
- Osto M, Parry N, Rehman R, et al. Malignant proliferating trichilemmal tumor of the scalp: a systematic review. Am J Dermatopathol. 2021;43:851-866. doi:10.1097/DAD.0000000000001991
- Plaza JA, Mackinnon A, Carrillo L, et al. Role of immunohistochemistry in the diagnosis of sebaceous carcinoma: a clinicopathologic and immunohistochemical study. Am J Dermatopathol. 2015;37:809-821. doi:10.1097/DAD.0000000000000255
- Singh P, Usman A, Motta L, et al. Malignant proliferating trichilemmal tumour. BMJ Case Rep. Published online August 17, 2018. doi:10.1136/bcr-2018-224460
- Ran NA, Granger EE, Brodland DG, et al. Risk factor number and recurrence, metastasis, and disease-related death in cutaneous squamous cell carcinoma. JAMA Dermatol. 2025;161:597-604. doi:10.1001/jamadermatol.2025.0128
- Jones EW. Proliferating epidermoid cysts. Arch Dermatol. 1966;94:11-19.
- Kemaloglu CA, Öztürk M, Aydın B, et al. Malignant proliferating trichilemmal tumor of the scalp: report of 4 cases and a short review of the literature. Case Reports Plast Surg Hand Surg. 2022;9:158-164. doi:10.1080/23320885.2022.2077208
- Saida T, Oohard K, Hori Y, et al. Development of a malignant proliferating trichilemmal cyst in a patient with multiple trichilemmal cysts. Dermatology. 1983;166:203-208. doi:10.1159/000249868
- Abdelhammed MH, Siatecka H, Diwan AH, et al. A rare case of a malignant proliferating trichilemmal tumor: a molecular study harboring potential therapeutic significance and a review of literature. Dermatopathology (Basel). 2024;11:354-363. doi:10.3390 /dermatopathology11040038
- Fronek L, Brahs A, Farsi M, et al. A rare case of trichilemmal carcinoma: histology and management. J Clin Aesthet Dermatol. 2021;14:25-30.
- Osto M, Parry N, Rehman R, et al. Malignant proliferating trichilemmal tumor of the scalp: a systematic review. Am J Dermatopathol. 2021;43:851-866. doi:10.1097/DAD.0000000000001991
- Plaza JA, Mackinnon A, Carrillo L, et al. Role of immunohistochemistry in the diagnosis of sebaceous carcinoma: a clinicopathologic and immunohistochemical study. Am J Dermatopathol. 2015;37:809-821. doi:10.1097/DAD.0000000000000255
- Singh P, Usman A, Motta L, et al. Malignant proliferating trichilemmal tumour. BMJ Case Rep. Published online August 17, 2018. doi:10.1136/bcr-2018-224460
- Ran NA, Granger EE, Brodland DG, et al. Risk factor number and recurrence, metastasis, and disease-related death in cutaneous squamous cell carcinoma. JAMA Dermatol. 2025;161:597-604. doi:10.1001/jamadermatol.2025.0128
Enlarging Mass on the Scalp
Enlarging Mass on the Scalp
A 61-year-old woman presented to the emergency department with worsening pain and bleeding from a scalp tumor of 16 years’ duration. Initially noted as a small nodule on the left parietal scalp on computed tomography of the head, the mass had grown rapidly in recent years and currently measured 22×10×15 cm. At prior consultations with plastic and general surgery, the patient had declined surgical intervention. At the current presentation, biopsies were performed by plastic surgery, and a dermatopathology consultation was ordered. Histopathology revealed atypical keratinocytes, nuclear pleomorphism, lobulating epithelial masses with trichilemmal keratinization, and CD34 positivity. Subsequent computed tomography and positron emission tomography of the head showed occipital skull erosion and bilateral cervical lymphadenopathy, suggesting metastasis.
Perioperative Considerations for Orthopedic Surgery in a Geriatric Population
Perioperative Considerations for Orthopedic Surgery in a Geriatric Population
More than 40 million surgeries are performed annually in the United States, of which > 18 million are orthopedic, including > 1 million emergency orthopedic surgeries and > 2 million joint replacements.1-4 Notably, > 50% of patients undergoing orthopedic surgery are aged ≥ 65 years, a demographic shift driven by longer life expectancies and an increasing number of older adults remaining physically active for extended periods.5 Osteoarthritis, the most common joint disease, affects 10% of men and 18% of women aged > 60 years, often necessitating an orthopedic joint replacement.6 Perioperative morbidity and mortality are 2.9- to 6.7-times higher in older adults compared with younger adults.7 These risks include infection, venous thromboembolism (VTE), pressure ulcers, reduced mobility, and increased mortality. Due to the high incidence of these complications in older surgical patients, special perioperative protocols and considerations are needed when preparing an older patient for surgery. This review aims to establish concrete considerations and guidelines for perioperative management.
METHODOLOGY
A literature review of PubMed, Google Scholar, and IEEE Xplore identified research on perioperative challenges in geriatric orthopedic surgery. Keywords included geriatrics and orthopedic surgery, perioperative care in geriatric populations, and orthopedic perioperative care. Inclusion criteria were strictly defined to ensure relevance to the geriatric population, with studies focusing on patients aged ≥ 65 years. Exclusion criteria were applied to remove studies that did not involve geriatric populations or orthopedic surgeries or that lacked a clear perioperative focus. Studies were analyzed for design, interventions, and outcomes. Special attention was given to identifying common challenges and trends related to perioperative considerations. We developed a narrative report providing a comprehensive overview of the current understanding of perioperative care for geriatric orthopedic patients to offer practical recommendations for clinicians to use in their practice.
RESULTS
Consistent with the narrative review methodology described, the literature search yielded a broad range of publications addressing perioperative considerations in geriatric orthopedic patients. Articles were screened for relevance to patients aged ≥ 65 years undergoing orthopedic surgery and for applicability to perioperative optimization and postoperative outcomes. Given the heterogeneity in study design, population characteristics, and outcome reporting, findings are presented descriptively rather than being quantitatively pooled. Studies not focused on geriatric populations, orthopedic procedures, or perioperative management were excluded. Key themes included multimorbidity and comorbidity optimization, age-related physiologic changes, frailty assessment and fracture risk stratification, nutritional and bone health management, mechanism of injury considerations, prevention of postoperative complications, and the role of multidisciplinary perioperative care.
Unique Physiological Challenges
The aging process induces a range of physiological changes that can increase morbidity and mortality following surgery. One of the most essential elements to surgical recovery is wound healing, as impairments in this process can lead to adverse events, including infection, cosmetic deformity, and wound dehiscence. The general paradigm of aging involves cell senescence resulting in slower or disorganized functional capacity of these cells.8 While wound healing in older individuals was once thought to be defective, recent research has demonstrated that this process is not absent, but delayed.9
Wound healing is a tightly regulated and evolutionarily conserved process that proceeds through 3 main phases: inflammation, proliferation, and remodeling. Re-epithelialization begins with the migration of epithelial cells from hair follicles, sweat glands, or wound margins (depending on wound depth) and is influenced by oxygen levels, moisture, and growth factors.9 Several characteristics of aged skin contribute to the delayed healing process. Aged skin has fewer hair follicles and eccrine sweat glands, as well as decreased follicle thickness.10 This results in fewer proliferating cells for wound healing and lower amounts of sebum production for skin moisture.11 Furthermore, aged fibroblasts are fewer in number and less effective in synthesizing extracellular matrices, resulting in slower and less tensile wound healing.12 Additionally, microvascular changes associated with aging result in disorganized vasculature, which impairs oxygen delivery to the wound bed and diminishes the influx of proinflammatory cells necessary for effective healing.13 These senescent traits of aged skin contribute to the delayed wound healing process found in geriatric patients.
Compounding these age-related factors is the prevalence of multimorbidity, or coexisting chronic diagnoses, in 55% to 98% of older patients.14 Common comorbidities include peripheral arterial disease, chronic venous insufficiency, type 1 and type 2 diabetes, neoplasms, atherosclerotic disease, and hypertension. Older patients are more likely to be prescribed corticosteroids and chemotherapeutic agents that impair the function of inflammatory cells necessary for wound healing.15,16 Additionally, decreased mobility is more common in geriatric patients, which can increase the risk of wound formation, particularly pressure ulcers.17
Perioperative Considerations
All surgical patients undergo a formal or informal preoperative evaluation to assess their fitness for surgery, with the goal of minimizing both anesthesia-related risks and postoperative complications. A widely used tool in this assessment is the American Society of Anesthesiologists (ASA) physical status classification, which stratifies patients into 6 categories based on their medical history and overall health status.18 Classes range from healthy patients (Class I) to organ donors who are brain-dead (Class VI).
Cardiac optimization is an essential component of preoperative evaluation for older adults due to their higher risk of underlying cardiovascular disease.19 This process involves an in-depth review of the patient’s cardiac history, including the timing and nature of any prior interventions and the recurrence rate. Functional capacity is assessed through metabolic equivalents, where a threshold of > 4 metabolic equivalents (the ability to walk up a flight of stairs) is considered adequate for surgery. Risk is assessed based on the specific surgical procedure, and nonemergent orthopedic procedures are considered intermediate risk. If a patient is deemed high risk at any stage of this evaluation, further cardiac testing is indicated.
Pulmonary optimization is typically necessary for geriatric patients, who are more likely to have conditions such as chronic obstructive pulmonary disease or interstitial lung disease.14,20 In patients without severe systemic lung disease, pulmonary optimization involves assessing the functional expiratory volume and diffusing capacity for carbon monoxide. In addition, aggressive modification of risk factors, such as smoking cessation, is strongly recommended.
Additional perioperative conditions are disease-specific and involve evaluation of comorbid illnesses and recognition of absolute contraindications to noncardiac surgery. For instance, an ejection fraction of < 35%, a history of myocardial infarction within 6 months, or active diabetic ketoacidosis are all absolute contraindications to elective surgery. For orthopedic procedures, additional contraindications include symptomatic bacteremia, active joint or local tissue infection, severe malnutrition, uncontrolled metabolic syndrome or chronic disease, untreated immunodeficiency, and active deep venous thrombosis (DVT) or pulmonary embolism.21
Bone Health and Nutrition
In the context of orthopedic surgery, the hallmark of clinically defined optimal bone health is a musculoskeletal system that provides the ability for pain-free functional and occupational tasks with an adequate capacity to withstand the mechanical forces imparted by everyday life. Back pain and arthritis are the fourth- and sixth-most common complaints in primary care, underscoring suboptimal bone health management in developed countries.22
Optimizing bone health through proper nutrition is crucial in the perioperative management of geriatric orthopedic patients. The clinical diagnosis of malnutrition has well-studied associations with worse outcomes after orthopedic surgery, which include increased mortality, hospital length of stay, readmission rates, and health institution spending.23-25 Some studies show that up to 60% of geriatric patients may be malnourished.26
Regarding vitamin and mineral supplements, the general consensus before orthopedic surgery is that vitamins A, C, D, and E, and zinc are predictive in determining postoperative health.27 However, Curtis et al state that therapy should be targeted at correcting relative deficiencies; supraphysiologic concentrations of these vitamins do not appear to be helpful.27 This claim may merit serum studies to rule out deficiencies. Dietitians should be involved in the creation of a patient care plan in the spirit of multidisciplinary orthopedic surgery approaches, which have proven to result in superior patient outcomes.28 Additionally, directive counseling should be provided when necessary.
In patients with adequately managed nutrition, 7 to 10 days of diet optimization is typically sufficient, but patients with malnutrition may require sustained nutritional support for up to 6 weeks; a standardized time for adequate nutrition supplementation has not been identified.25-27 Postoperative management is similar in older patients who are malnourished and those receiving adequate nutrition after orthopedic surgery, which typically involves 3 weeks of a high-protein diet.26
Evaluating Mechanisms of Injury
Assessing the mechanism of injury (MOI) is essential to developing an appropriate and successful orthopedic treatment plan. MOI is typically categorized as low energy, which consists of ground-level falls and other minor trauma, or high energy, which can include motor vehicle crashes or falls from a height.29 Unlike younger patients who typically experience trauma from high-energy MOIs, geriatric patients often sustain fractures from low-energy MOIs. The importance of assessing MOI for the geriatric population is magnified as it provides vital clues that not only help determine the nature of the injury, but also highlight underlying frailty, comorbidities, and potential complications. Weakness or deconditioning related to older age is often not discovered before trauma, which is why assessing the MOI can provide valuable information regarding overall patient health.30
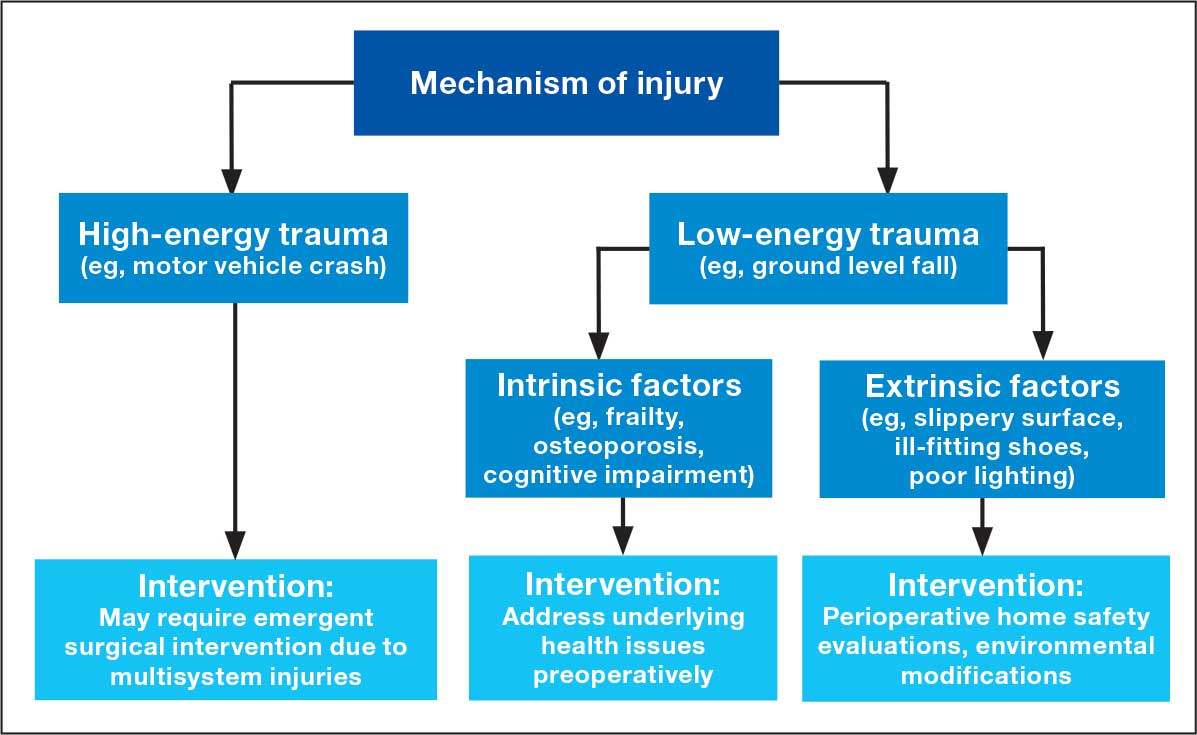
The MOI of trauma also is correlated with factors that influence postoperative recovery and overall prognosis (Figure). Falls comprise more than three-quarters of the MOI in geriatric patients with trauma, and > 90% of these falls are ground-level or other simple falls.30 Falls secondary to an intrinsic disorder, rather than an extrinsic environmental hazard, are more common in geriatric patients.31
These events may be associated with an underlying medical condition, such as osteopenia, osteoporosis, or neuromuscular disorders, such as Parkinson disease.32 They may also be attributed to normal age-related changes, such as decreased visual acuity, reduced reaction time, or mild cognitive impairment.30 An estimated 6% to 35% of geriatric patients who present to the emergency department have some degree of cognitive dysfunction.33 Accordingly, a thorough understanding of the events leading up to injury is vital for the management of older patients. Knowing the specific circumstances of a fall can provide insight into the patient’s gait, balance, and need for further investigations such as cognitive screening or evaluation of home safety. This information can guide decisions regarding preoperative optimization of medications and postoperative rehabilitation interventions.
Frailty and Risk of Fracture
Frailty is a clinical syndrome defined by overall decreased capacity for the body’s adaptive changes to various stressors.34 It is a common condition in geriatric populations due to cumulative degenerative changes and multisystem decline over a lifetime’s worth of disruptions to natural homeostasis.34 In orthopedics, frailty typically refers to musculoskeletal durability and resilience in response to mechanical forces (ie, falls, trauma, and high-acceleration movements). Globally, > 200 million people have osteoporotic frailty, leading to 9 million hip fractures annually.35 More than 30% of people aged ≥ 65 years fall ≥ 1 time per calendar year.36
Assessing frailty in geriatric patients undergoing orthopedic surgery is vital, as it predisposes patients to higher rates of morbidity, mortality, and institutionalization, particularly from falls and resultant fragility fractures.37-39 This is true for a wide range of orthopedic procedures, spanning elective to urgent surgeries and involving the axial and appendicular skeleton.40,41 Given the high rates of fractures, subsequent patient morbidity, and financial burden on the health care system, effective frailty screening is essential.
There are many strategies to assess frailty risk and subsequent fracture risk.42 Questionnaires or online medical calculators serve as easy-to-use tools for assessment of frailty or associated predictors of fragility fractures. Validated assessment tools are provided in Table 1.
Dual-emission X-ray absorptiometry is a well-established way to determine bone density and establish fracture risk. The Fried Frailty Phenotype score and Short Performance Physical Battery test are clinically applicable methods of assessing frailty in older outpatient populations. Although these examinations focus on different aspects of the patient, they have moderate agreeability, are sensitive, and can be readily performed in the clinical setting as demonstrated by a > 90% patient participation rate for both methods.42 Finally, several serum studies can be predictive of frailty, the most readily modifiable of which are vitamin D3, ferritin, albumin, and calcium.43 Although they are more invasive for the patient, serum studies can provide additional modifiable targets for perioperative optimization and contribute to risk stratification.
Risk stratification should take place around 6 weeks before surgery, which should provide adequate time for rectification of preoperative barriers to elective surgical intervention—namely nutritional status. In cases of urgent or emergent procedures (ie, femoral neck fracture with concern for avascular necrosis of the femoral head), this may not be possible but should be conducted nonetheless for patient-specific postoperative rehabilitation and risk reduction.
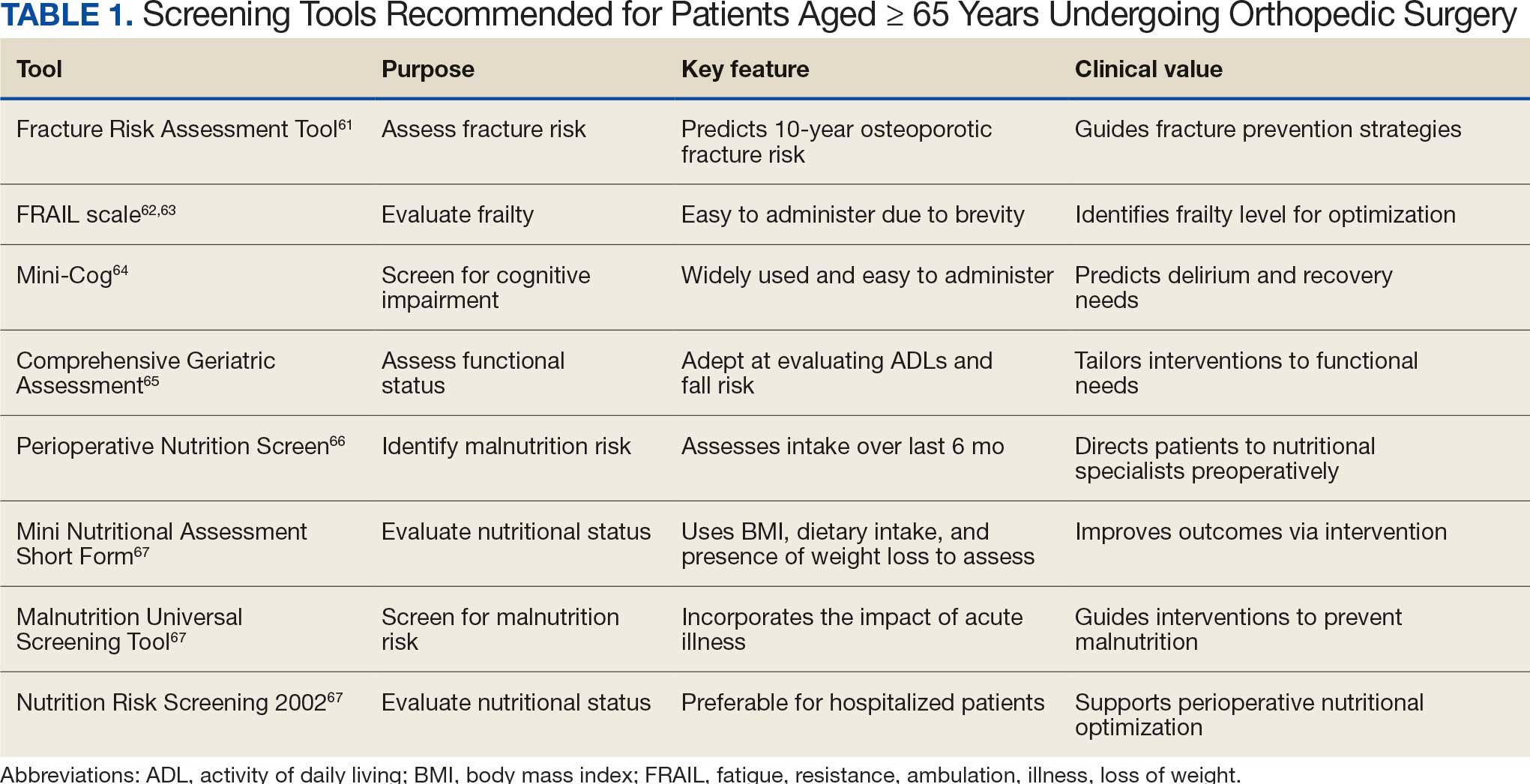
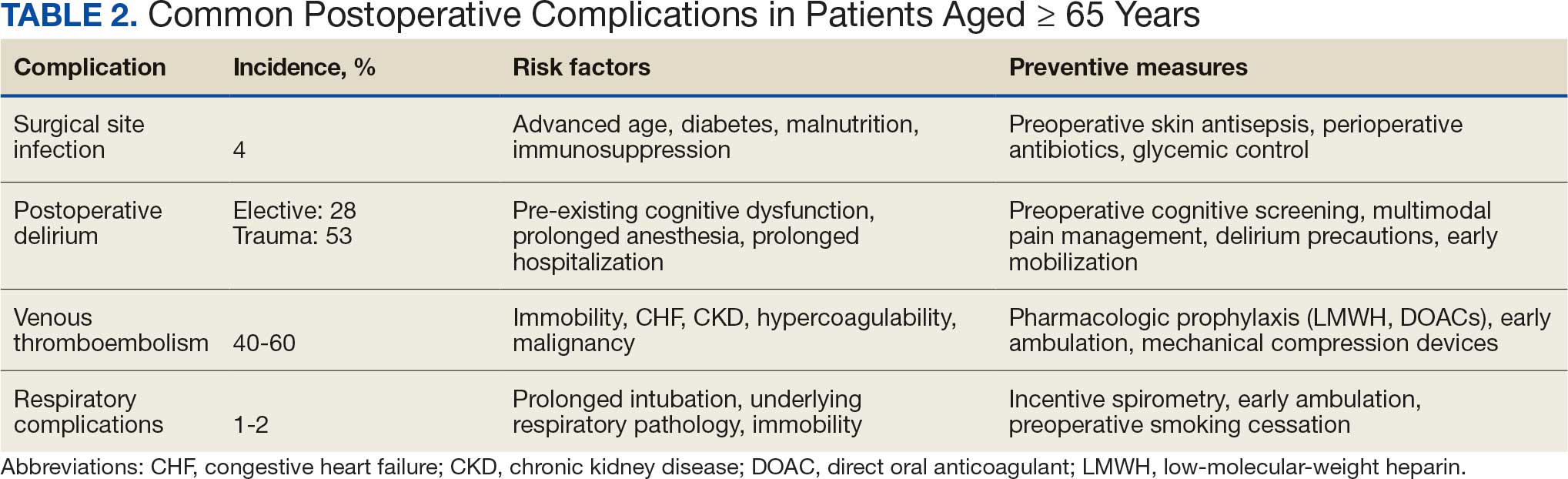
Postoperative Complication Risks
Postoperative complications affect nearly 15% of geriatric orthopedic patients, highlighting the need for comprehensive preoperative evaluations to assess risk factors.44 Age-related physiological changes, frailty, and comorbidities complicate recovery and management (Table 2).
Wound healing is impaired in older individuals due to suboptimal circulation and decreased oxygenation that is secondary to age-related changes, as well as the increased likelihood of comorbid conditions (eg, diabetes).7 Surgical site infections can be particularly malicious in geriatric patients, with a 4% incidence.45,46 Hospitalization can be prolonged by a mean 2 weeks, which increases the risk of hospital-associated delirium and iatrogenic complications.46 Both the mortality rate and costs associated with hospitalization are higher for older patients who develop surgical site infections compared with patients aged < 65 years, underscoring the importance of vigilant monitoring, early detection, and effective preoperative screening to identify and manage modifiable risk factors.47
Postoperative delirium is another common complication of orthopedic surgery in the geriatric population, increasing morbidity and mortality. The incidence is reported to be as high as 53.3% in the trauma setting and 28.3% in the elective setting, indicating a need to assess patient risk preoperatively.48,49 Several factors contribute to the high incidence of delirium, including advanced age, longer surgical durations, intraoperative hypotension and hypercapnia, pre-existing cognitive dysfunction, and postoperative sleep disorders.50
VTE is another common cause of complications following orthopedic surgery. The development of DVT can lead to subsequent pulmonary embolism, which can be fatal. Orthopedic surgery patients are already at higher risk of DVT and VTE than other surgical patients, with an incidence as high as 40% to 60%, though it is frequently asymptomatic.51,52 Geriatric patients may be more likely to have concurrent comorbidities that increase hypercoagulability.53 Congestive heart failure, chronic kidney disease, and cardiovascular disease are all more common in older individuals and can increase the risk of VTE by 2-fold.53 While anticoagulation is the standard of care to prevent VTE after orthopedic surgery, geriatric patients require more careful monitoring due to the higher incidence of bleeding complications. Additionally, early postoperative mobilization is critical to reduce the risk of DVT without significantly increasing pain or causing other adverse events.54
Respiratory complications are common after orthopedic surgery, particularly atelectasis and bronchospasm, which can result from intraoperative mechanical ventilation.55 While these conditions are typically self-limiting, more severe respiratory issues such as pneumonia are a significant concern because they may lead to the need for mechanical ventilation and admission to the intensive care unit (ICU). The more severe complications have an incidence of about 1% to 2% in orthopedic surgery patients.56 Preventive strategies, such as respiratory physiotherapy and guided breathing exercises, are crucial to minimize perioperative pulmonary complications and promote optimal recovery. Addressing these challenges through early intervention is essential to improve outcomes.
Multidisciplinary Perioperative Care
Multidisciplinary care in orthopedic surgery involves collaborative management of patient care by general practitioners, surgeons, anesthesiologists, dietitians, physical and occupational therapists, inpatient health care practitioners (HCPs), and social services. The goal of this form of care is to provide a longitudinal sequence of health-optimization tactics that prepare a patient for surgery and give them the best chance of postoperative recovery.
Given that many aspects of geriatric health play a role in orthopedic postoperative outcomes, there are many preoperative factors to consider. As previously discussed, preoperative evaluation of geriatric patients should include nutritional and fragility screening to determine surgical candidacy and target modifiable risk factors for risk reduction. This screening can be conducted by primary care practitioners and orthopedic surgeons in an outpatient setting. A multidisciplinary approach benefits patients by decreasing time to surgery.35
Several large studies have demonstrated the positive influence of a multidisciplinary approach on patient-oriented outcomes in orthopedic patients. Incorporation of this style of care in contrast to surgeon-led perioperative optimization leads to fewer floor and ICU admissions, shorter lengths of stay, and decreased mortality rates.35,57 These findings are broadly applicable to a wide range of orthopedic surgeries and even surgeries outside of the musculoskeletal system.58,59 In addition, this strategy has demonstrated reduced in-hospital health care costs due to shorter lengths of stay and fewer ICU admissions. Physical and occupational therapy also have irreplaceable roles in outcomes after orthopedic surgeries. They have independently been shown to decrease pain, increase range of motion, and increase functionality in daily life.60 These aspects of recovery are essential for geriatric well-being.
Screening Tools
The World Health Organization FRAX fracture risk assessment tool (www.fraxplus.org/calculation-tool) was developed to identify patients at high risk of fracture and subsequent complications and to guide clinical decision-making regarding pharmacologic interventions.61 FRAX calculates the 10-year probability of fracture based on demographic factors, such as age and body mass index, clinical measures (eg, femoral neck bone mineral density), and risk factors (eg, prior fragility fractures, substance use history, and prolonged glucocorticoid use).61 The online tool is easy to use, making it a valuable resource for assessing fracture risk and determining appropriate treatment strategies.
The fatigue, resistance, ambulation, illnesses, loss of weight (FRAIL) scale assesses frailty in older adults. The scale classifies patients into 3 categories: robust, prefrail, and frail. The frail category is associated with an increased frequency of hip fracture and an elevated ASA class.62 Additionally, the FRAIL scale has demonstrated value in predicting hospital length of stay and the risk of postoperative complications.62 It also has shown utility in quantifying frailty status, which is traditionally challenging to assess systematically.63
The Mini-Cog is commonly used in geriatric populations to screen for cognitive impairment. Preoperative Mini-Cog screening has been shown to predict the development of postoperative complications.64 Geriatric patients who screened positive for cognitive impairment prior to orthopedic surgery were more likely to develop postoperative delirium, require alternative discharge disposition, and have a longer hospital length of stay.64 Mini-Cog serves as an important preoperative tool for identifying patients who may benefit from closer postoperative monitoring and tailored care.
The Comprehensive Geriatric Assessment (CGA) is a multidimensional evaluation that has been validated for use in geriatric patients undergoing orthopedic surgery.65 The CGA assesses functional status and the ability to perform activities of daily living (ADLs), such as eating, dressing, and ambulating. Poor ADLs are associated with increased risk of falls and cardiopulmonary complications. The CGA allows HCPs to identify patients at higher risk of complications and tailor interventions that optimize functional recovery during the perioperative period.
Nutritional screening is another component of preoperative evaluation in older adults undergoing orthopedic surgery. The Perioperative Nutrition Screen is a preoperative phone assessment of unintentional weight loss in the past 6 months.66 Patients who screen positive are asked to come in for a preoperative visit with a registered dietitian who can further evaluate the nutritional status of the patient.
The Mini Nutritional Assessment Short Form (MNA-SF), Malnutrition Universal Screening Tool, and Nutrition Risk Screening 2002 have all been validated for use in older patients undergoing orthopedic surgery. Among these, the MNA-SF has demonstrated superior utility in predicting hospital readmission and mortality.67 Given the established links between malnutrition and poor surgical outcomes, routine nutritional screening is important for identifying geriatric patients who may require preoperative nutritional interventions.
CONCLUSIONS AND RECOMMENDATIONS
Perioperative management of geriatric patients undergoing orthopedic surgery requires an assessment and strategy focused on risk stratification, patient optimization, and mitigation of potential complications and mortality. Due to the complexity and comprehensive nature of an optimal perioperative plan, creating the plan early is essential to ensure adequate time for patient optimization and care coordination.
Nutrition plays a critical role in the success of surgical procedures, and orthopedics is no exception. Extra care should be taken to preoperatively optimize patient bone health before surgical intervention to enhance recovery and reduce the risk of complications. After an appropriate patient history and clinical picture are gathered, screening tools should be used on a case-by-case basis to further characterize comorbid conditions that may contribute to suboptimal outcomes. Additionally, given the proven association between frailty and fracture risk, frailty serves as a readily quantifiable predictor of patient-oriented outcomes. This should be assessed preoperatively with appropriate risk-stratification tools to determine appropriate postoperative measures to prevent morbidity and mortality.
Orthopedic surgery is increasingly common in geriatric patients, who face higher perioperative risks due to age-related physiological changes, multimorbidity, and frailty. Optimizing preoperative assessment and adopting a multidisciplinary approach—integrating surgeons, anesthesiologists, physical therapists, and dietitians—can improve outcomes, reduce complications, and enhance recovery. The successful use of the tools and strategies outlined in this article by primary care should facilitate access to and recovery from orthopedic surgery in the geriatric population.
Dobson GP. Trauma of major surgery: a global problem that is not going away. Int J Surg. 2020;81:47-54. doi:10.1016/j.ijsu.2020.07.017
United States (US) orthopedic procedures count by segments and forecast to 2030. GlobalData. February 17, 2023. Accessed April 29, 2026. https://www.globaldata.com/store/report/usa-orthopedic-procedures-analysis/
Jarman MP, Weaver MJ, Haider AH, Salim A, Harris MB. The national burden of orthopedic injury: cross-sectional estimates for trauma system planning and optimization. J Surg Res. 2020;249:197-204. doi:10.1016/j.jss.2019.12.023
Hegde V, Stambough JB, Levine BR, et al. Highlights of the 2022 American Joint Replacement Registry Annual Report. Arthroplast Today. 2023;21:101137. doi:10.1016/j.artd.2023.101137
Nakamura K, Ogata T. Locomotive syndrome: definition and management. Clin Rev Bone Miner Metab. 2016;14:56-67. doi:10.1007/s12018-016-9208-2
Glyn-Jones S, Palmer AJR, Agricola R, et al. Osteoarthritis. Lancet. 2015;386:376-387. doi:10.1016/S0140-6736(14)60802-3
Hughes S, Leary A, Zweizig S, Cain J. Surgery in elderly people: preoperative, operative and postoperative care to assist healing. Best Pract Res Clin Obstet Gynaecol. 2013;27:753-765. doi:10.1016/j.bpobgyn.2013.02.006
Regulski MJ. Cellular senescence: what, why, and how. Wounds. 2017;29:168-174.
Kremer M, Burkemper N. Aging skin and wound healing. Clin Geriatr Med. 2024;40:1-10. doi:10.1016/j.cger.2023.06.001
Fenske NA, Lober CW. Structural and functional changes of normal aging skin. J Am Acad Dermatol. 1986;15:571-585. doi:10.1016/S0190-9622(86)70208-9
Van Neste D, Tobin DJ. Hair cycle and hair pigmentation: dynamic interactions and changes associated with aging. Micron. 2004;35:193-200. doi:10.1016/j.micron.2003.11.006
Salzer MC, Lafzi A, Berenguer-Llergo A, et al. Identity noise and adipogenic traits characterize dermal fibroblast aging. Cell. 2018;175:1575-1590.e22. doi:10.1016/j.cell.2018.10.012
Jin K. A microcirculatory theory of aging. Aging Dis. 2019;10:676-683. doi:10.14336/AD.2019.0315
Marengoni A, Angleman S, Melis R, et al. Aging with multimorbidity: a systematic review of the literature. Ageing Res Rev. 2011;10:430-439. doi:10.1016/j.arr.2011.03.003
Waljee AK, Rogers MAM, Lin P, et al. Short term use of oral corticosteroids and related harms among adults in the United States: population based cohort study. BMJ. 2017;357:j1415. doi:10.1136/bmj.j1415
Given B, Given CW. Older adults and cancer treatment. Cancer. 2008;113:3505-3511. doi:10.1002/cncr.23939
Ferrucci L, Cooper R, Shardell M, Simonsick EM, Schrack JA, Kuh D. Age-related change in mobility: perspectives from life course epidemiology and geroscience. J Gerontol A Biol Sci Med Sci. 2016;71:1184-1194. doi:10.1093/gerona/glw043
Mayhew D, Mendonca V, Murthy BVS. A review of ASA physical status - historical perspectives and modern developments. Anaesthesia. 2019;74:373-379. doi:10.1111/anae.14569
Eagle KA, Berger PB, Calkins H, et al. ACC/AHA guideline update for perioperative cardiovascular evaluation for noncardiac surgery—executive summary a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee to Update the 1996 Guidelines on Perioperative Cardiovascular Evaluation for Noncardiac Surgery). Circulation. 2002;105:1257-1267. doi:10.1161/circ.105.10.1257
Bapoje SR, Whitaker JF, Schulz T, Chu ES, Albert RK. Preoperative evaluation of the patient with pulmonary disease. Chest. 2007;132:1637-1645. doi:10.1378/chest.07-0347
Choe H, Indelli PF, Ricciardi B, et al. What are the absolute contraindications for elective total knee or hip arthroplasty? J Arthroplasty. 2025;40(2 suppl 1):S45-S47. doi:10.1016/j.arth.2024.10.041
Finley CR, Chan DS, Garrison S, et al. What are the most common conditions in primary care? Systematic review. Can Fam Physician. 2018;64:832-840.
Vaid S, Bell T, Grim R, Ahuja V. Predicting risk of death in general surgery patients on the basis of preoperative variables using American College of Surgeons National Surgical Quality Improvement Program data. Perm J. 2012;16:10-17. doi:10.7812/TPP/12-019
Correia MTD, Waitzberg DL. The impact of malnutrition on morbidity, mortality, length of hospital stay and costs evaluated through a multivariate model analysis. Clin Nutr. 2003;22:235-239. doi:10.1016/S0261-5614(02)00215-7
Friedman J, Lussiez A, Sullivan J, Wang S, Englesbe M. Implications of sarcopenia in major surgery. Nutr Clin Pract. 2015;30:175-179. doi:10.1177/0884533615569888
Hirsch KR, Wolfe RR, Ferrando AA. Pre- and post-surgical nutrition for preservation of muscle mass, strength, and functionality following orthopedic surgery. Nutrients. 2021;13:1675. doi:10.3390/nu13051675
Curtis W, Choi T, Ahmad A, Shultz C. Perioperative nutritional considerations in orthopaedic surgery: a review of the literature. West J Orthop. 2023;12:1. https://digitalrepository.unm.edu/wjo/vol12/iss1/1
Wischmeyer PE, Carli F, Evans DC, et al. American Society for Enhanced Recovery and Perioperative Quality Initiative joint consensus statement on nutrition screening and therapy within a surgical enhanced recovery pathway. Anesth Analg. 2018;126:1883-1895. doi:10.1213/ANE.0000000000002743
Mun F, Ringenbach K, Baer B, et al. Factors influencing geriatric orthopaedic trauma mortality. Injury. 2022;53:919-924. doi:10.1016/j.injury.2022.01.005
Bonne S, Schuerer DJE. Trauma in the older adult: epidemiology and evolving geriatric trauma principles. Clin Geriatr Med. 2013;29:137-150. doi:10.1016/j.cger.2012.10.008
Montero-Odasso MM. Falls as a geriatric syndrome: mechanisms and risk identification. In: Duque G, Kiel DP, eds. Osteoporosis in Older Persons: Advances in Pathophysiology and Therapeutic Approaches. 2nd ed. Springer International Publishing; 2016:171-186. doi:10.1007/978-3-319-25976-5_10
Lach HW, Reed AT, Arfken CL, et al. Falls in the elderly: reliability of a classification system. J Am Geriatr Soc. 1991;39:197-202. doi:10.1111/j.1532-5415.1991.tb01626.x
Carpenter CR, DesPain B, Keeling TN, Shah M, Rothenberger M. The six-item screener and AD8 for the detection of cognitive impairment in geriatric emergency department patients. Ann Emerg Med. 2011;57:653-661. doi:10.1016/j.annemergmed.2010.06.560
Clegg A, Young J, Iliffe S, Rikkert MO, Rockwood K. Frailty in elderly people. Lancet. 2013;381:752-762. doi:10.1016/S0140-6736(12)62167-9
Patel JN, Klein DS, Sreekumar S, Liporace FA, Yoon RS. Outcomes in multidisciplinary team-based approach in geriatric hip fracture care: a systematic review. J Am Acad Orthop Surg. 2020;28:128-133. doi:10.5435/JAAOS-D-18-00425
Amador LF, Loera JA. Preventing postoperative falls in the older adult. J Am Coll Surg. 2007;204:447-453. doi:10.1016/j.jamcollsurg.2006.12.010
Tembo MC, Holloway-Kew KL, Mohebbi M, et al. The association between a fracture risk tool and frailty: Geelong Osteoporosis Study. BMC Geriatr. 2020;20:196. doi:10.1186/s12877-020-01595-8
Demiris¸ B, Basat S, Kurt F, Aksakal B, Basat O. Evaluation of the relationship between frailty and fracture risk using Fracture Risk Assessment Tool in patients 65 years and over. South Clin Istanb Eurasia. 2023;34:42-48. doi:10.14744/scie.2022.66564
Partridge JSL, Harari D, Dhesi JK. Frailty in the older surgical patient: a review. Age Ageing. 2012;41:142-147. doi:10.1093/ageing/afr182
Mamtora PH, Fortier MA, Barnett SR, Schmid LN, Kain ZN. Peri-operative management of frailty in the orthopedic patient. J Orthop. 2020;22:304-307. doi:10.1016/j.jor.2020.05.024
Leven DM, Lee NJ, Kim JS, et al. Frailty is predictive of adverse postoperative events in patients undergoing lumbar fusion. Global Spine J. 2017;7:529-535. doi:10.1177/2192568217700099
Pritchard JM, Kennedy CC, Karampatos S, et al. Measuring frailty in clinical practice: a comparison of physical frailty assessment methods in a geriatric out-patient clinic. BMC Geriatr. 2017;17:264. doi:10.1186/s12877-017-0623-0
Kumar A, Dhar M, Agarwal M, Mukherjee A, Saxena V. Predictors of frailty in the elderly population: a cross-sectional study at a tertiary care center. Cureus. 2022;14:e30557. doi:10.7759/cureus.30557
Scarano KA, Philp FH, Westrick ER, Altman GT, Altman DT. Evaluating postoperative complications and outcomes of orthopedic fracture repair in nonagenarian patients. Geriatr Orthop Surg Rehabil. 2018;9:2151459318758106. doi:10.1177/2151459318758106
Liang Z, Rong K, Gu W, et al. Surgical site infection following elective orthopaedic surgeries in geriatric patients: incidence and associated risk factors. Int Wound J. 2019;16:773-780. doi:10.1111/iwj.13096
Ren M, Liang W, Wu Z, Zhao H, Wang J. Risk factors of surgical site infection in geriatric orthopedic surgery: a retrospective multicenter cohort study. Geriatr Gerontol Int. 2019;19:213-217. doi:10.1111/ggi.13590
Kaye KS, Schmader KE, Sawyer R. Surgical site infection in the elderly population. Clin Infect Dis. 2004;39:1835-1841. doi:10.1086/425744
Bruce AJ, Ritchie CW, Blizard R, Lai R, Raven P. The incidence of delirium associated with orthopedic surgery: a meta-analytic review. Int Psychogeriatr. 2007;19:197-214. doi:10.1017/S104161020600425X
Williams-Russo P, Urquhart BL, Sharrock NE, Charlson ME. Post-operative delirium: predictors and prognosis in elderly orthopedic patients. J Am Geriatr Soc. 1992;40:759-767. doi:10.1111/j.1532-5415.1992.tb01846.x
Wang J, Li Z, Yu Y, Li B, Shao G, Wang Q. Risk factors contributing to postoperative delirium in geriatric patients postorthopedic surgery. Asia Pac Psychiatry. 2015;7:375-382. doi:10.1111/appy.12193
Geerts WH, Pineo GF, Heit JA, et al. Prevention of venous thromboembolism: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest. 2004;126:338S-400S. doi:10.1378/chest.126.3_suppl.338S
Kahn SR, Shivakumar S. What’s new in VTE risk and prevention in orthopedic surgery. Res Pract Thromb Haemost. 2020;4:366-376. doi:10.1002/rth2.12323
Uzel K, Azboy I·, Parvizi J. Venous thromboembolism in orthopedic surgery: global guidelines. Acta Orthop Traumatol Turc. 2023;57:192-203. doi:10.5152/j.aott.2023.23074
Peck M, Holthaus A, Kingsbury K, Salsberry MG, Duggirala V. Mobility in acute care for geriatric patients with orthopedic conditions: a review of recent literature. Curr Geri Rep. 2020;9:300-310. doi:10.1007/s13670-020-00347-1
Leme LEG, Sitta MC, Toledo M, Henriques SS. Orthopedic surgery among the elderly: clinical characteristics. Rev Bras Ortop. 2015;46:238-246. doi:10.1016/S2255-4971(15)30189-0
Malcolm TL, Knezevic NN, Zouki CC, Tharian AR. Pulmonary complications after hip and knee arthroplasty in the United States, 2004-2014. Anesth Analg. 2020;130:917-924. doi:10.1213/ANE.0000000000004265
Kamal T, Conway RM, Littlejohn I, Ricketts D. The role of a multidisciplinary pre-assessment clinic in reducing mortality after complex orthopaedic surgery. Ann R Coll Surg Engl. 2011;93:149-151. doi:10.1308/003588411X561026
Davis MJ, Luu BC, Raj S, Abu-Ghname A, Buchanan EP. Multidisciplinary care in surgery: Are team-based interventions cost-effective? Surgeon. 2021;19:49-60. doi:10.1016/j.surge.2020.02.005
Frassanito L, Vergari A, Nestorini R, et al. Enhanced recovery after surgery (ERAS) in hip and knee replacement surgery: description of a multidisciplinary program to improve management of the patients undergoing major orthopedic surgery. Musculoskelet Surg. 2020;104:87-92. doi:10.1007/s12306-019-00603-4
Reddy RS, Alahmari KA, Alshahrani MS, et al. Exploring the impact of physiotherapy on health outcomes in older adults with chronic diseases: a cross-sectional analysis. Front Public Health. 2024;12:1415882. doi:10.3389/fpubh.2024.1415882
Watts NB. The Fracture Risk Assessment Tool (FRAX®): applications in clinical practice. J Womens Health (Larchmt). 2011;20:525-531. doi:10.1089/jwh.2010.2294
Gleason LJ, Benton EA, Alvarez-Nebreda ML, Weaver MJ, Harris MB, Javedan H. FRAIL questionnaire screening tool and short-term outcomes in geriatric fracture patients. J Am Med Dir Assoc. 2017;18:1082-1086. doi:10.1016/j.jamda.2017.07.005
Kojima G. Frailty defined by FRAIL scale as a predictor of mortality: a systematic review and meta-analysis. J Am Med Dir Assoc. 2018;19:480-483. doi:10.1016/j.jamda.2018.04.006
Culley DJ, Flaherty D, Fahey MC, et al. Poor performance on a preoperative cognitive screening test predicts postoperative complications in older orthopedic surgical patients. Anesthesiology. 2017;127:765-774. doi:10.1097/ALN.0000000000001859
Kong C, Zhang Y, Wang C, et al. Comprehensive geriatric assessment for older orthopedic patients and analysis of risk factors for postoperative complications. BMC Geriatr. 2022;22:644. doi:10.1186/s12877-022-03328-5
Williams DGA, Wischmeyer PE. Perioperative nutrition care of orthopedic surgery patient. Tech Orthop. 2020;35:15-18. doi:10.1097/BTO.0000000000000412
Koren-Hakim T, Weiss A, Hershkovitz A, et al. Comparing the adequacy of the MNA-SF, NRS-2002 and MUST nutritional tools in assessing malnutrition in hip fracture operated elderly patients. Clin Nutr. 2016;35:1053-1058. doi:10.1016/j.clnu.2015.07.014
More than 40 million surgeries are performed annually in the United States, of which > 18 million are orthopedic, including > 1 million emergency orthopedic surgeries and > 2 million joint replacements.1-4 Notably, > 50% of patients undergoing orthopedic surgery are aged ≥ 65 years, a demographic shift driven by longer life expectancies and an increasing number of older adults remaining physically active for extended periods.5 Osteoarthritis, the most common joint disease, affects 10% of men and 18% of women aged > 60 years, often necessitating an orthopedic joint replacement.6 Perioperative morbidity and mortality are 2.9- to 6.7-times higher in older adults compared with younger adults.7 These risks include infection, venous thromboembolism (VTE), pressure ulcers, reduced mobility, and increased mortality. Due to the high incidence of these complications in older surgical patients, special perioperative protocols and considerations are needed when preparing an older patient for surgery. This review aims to establish concrete considerations and guidelines for perioperative management.
METHODOLOGY
A literature review of PubMed, Google Scholar, and IEEE Xplore identified research on perioperative challenges in geriatric orthopedic surgery. Keywords included geriatrics and orthopedic surgery, perioperative care in geriatric populations, and orthopedic perioperative care. Inclusion criteria were strictly defined to ensure relevance to the geriatric population, with studies focusing on patients aged ≥ 65 years. Exclusion criteria were applied to remove studies that did not involve geriatric populations or orthopedic surgeries or that lacked a clear perioperative focus. Studies were analyzed for design, interventions, and outcomes. Special attention was given to identifying common challenges and trends related to perioperative considerations. We developed a narrative report providing a comprehensive overview of the current understanding of perioperative care for geriatric orthopedic patients to offer practical recommendations for clinicians to use in their practice.
RESULTS
Consistent with the narrative review methodology described, the literature search yielded a broad range of publications addressing perioperative considerations in geriatric orthopedic patients. Articles were screened for relevance to patients aged ≥ 65 years undergoing orthopedic surgery and for applicability to perioperative optimization and postoperative outcomes. Given the heterogeneity in study design, population characteristics, and outcome reporting, findings are presented descriptively rather than being quantitatively pooled. Studies not focused on geriatric populations, orthopedic procedures, or perioperative management were excluded. Key themes included multimorbidity and comorbidity optimization, age-related physiologic changes, frailty assessment and fracture risk stratification, nutritional and bone health management, mechanism of injury considerations, prevention of postoperative complications, and the role of multidisciplinary perioperative care.
Unique Physiological Challenges
The aging process induces a range of physiological changes that can increase morbidity and mortality following surgery. One of the most essential elements to surgical recovery is wound healing, as impairments in this process can lead to adverse events, including infection, cosmetic deformity, and wound dehiscence. The general paradigm of aging involves cell senescence resulting in slower or disorganized functional capacity of these cells.8 While wound healing in older individuals was once thought to be defective, recent research has demonstrated that this process is not absent, but delayed.9
Wound healing is a tightly regulated and evolutionarily conserved process that proceeds through 3 main phases: inflammation, proliferation, and remodeling. Re-epithelialization begins with the migration of epithelial cells from hair follicles, sweat glands, or wound margins (depending on wound depth) and is influenced by oxygen levels, moisture, and growth factors.9 Several characteristics of aged skin contribute to the delayed healing process. Aged skin has fewer hair follicles and eccrine sweat glands, as well as decreased follicle thickness.10 This results in fewer proliferating cells for wound healing and lower amounts of sebum production for skin moisture.11 Furthermore, aged fibroblasts are fewer in number and less effective in synthesizing extracellular matrices, resulting in slower and less tensile wound healing.12 Additionally, microvascular changes associated with aging result in disorganized vasculature, which impairs oxygen delivery to the wound bed and diminishes the influx of proinflammatory cells necessary for effective healing.13 These senescent traits of aged skin contribute to the delayed wound healing process found in geriatric patients.
Compounding these age-related factors is the prevalence of multimorbidity, or coexisting chronic diagnoses, in 55% to 98% of older patients.14 Common comorbidities include peripheral arterial disease, chronic venous insufficiency, type 1 and type 2 diabetes, neoplasms, atherosclerotic disease, and hypertension. Older patients are more likely to be prescribed corticosteroids and chemotherapeutic agents that impair the function of inflammatory cells necessary for wound healing.15,16 Additionally, decreased mobility is more common in geriatric patients, which can increase the risk of wound formation, particularly pressure ulcers.17
Perioperative Considerations
All surgical patients undergo a formal or informal preoperative evaluation to assess their fitness for surgery, with the goal of minimizing both anesthesia-related risks and postoperative complications. A widely used tool in this assessment is the American Society of Anesthesiologists (ASA) physical status classification, which stratifies patients into 6 categories based on their medical history and overall health status.18 Classes range from healthy patients (Class I) to organ donors who are brain-dead (Class VI).
Cardiac optimization is an essential component of preoperative evaluation for older adults due to their higher risk of underlying cardiovascular disease.19 This process involves an in-depth review of the patient’s cardiac history, including the timing and nature of any prior interventions and the recurrence rate. Functional capacity is assessed through metabolic equivalents, where a threshold of > 4 metabolic equivalents (the ability to walk up a flight of stairs) is considered adequate for surgery. Risk is assessed based on the specific surgical procedure, and nonemergent orthopedic procedures are considered intermediate risk. If a patient is deemed high risk at any stage of this evaluation, further cardiac testing is indicated.
Pulmonary optimization is typically necessary for geriatric patients, who are more likely to have conditions such as chronic obstructive pulmonary disease or interstitial lung disease.14,20 In patients without severe systemic lung disease, pulmonary optimization involves assessing the functional expiratory volume and diffusing capacity for carbon monoxide. In addition, aggressive modification of risk factors, such as smoking cessation, is strongly recommended.
Additional perioperative conditions are disease-specific and involve evaluation of comorbid illnesses and recognition of absolute contraindications to noncardiac surgery. For instance, an ejection fraction of < 35%, a history of myocardial infarction within 6 months, or active diabetic ketoacidosis are all absolute contraindications to elective surgery. For orthopedic procedures, additional contraindications include symptomatic bacteremia, active joint or local tissue infection, severe malnutrition, uncontrolled metabolic syndrome or chronic disease, untreated immunodeficiency, and active deep venous thrombosis (DVT) or pulmonary embolism.21
Bone Health and Nutrition
In the context of orthopedic surgery, the hallmark of clinically defined optimal bone health is a musculoskeletal system that provides the ability for pain-free functional and occupational tasks with an adequate capacity to withstand the mechanical forces imparted by everyday life. Back pain and arthritis are the fourth- and sixth-most common complaints in primary care, underscoring suboptimal bone health management in developed countries.22
Optimizing bone health through proper nutrition is crucial in the perioperative management of geriatric orthopedic patients. The clinical diagnosis of malnutrition has well-studied associations with worse outcomes after orthopedic surgery, which include increased mortality, hospital length of stay, readmission rates, and health institution spending.23-25 Some studies show that up to 60% of geriatric patients may be malnourished.26
Regarding vitamin and mineral supplements, the general consensus before orthopedic surgery is that vitamins A, C, D, and E, and zinc are predictive in determining postoperative health.27 However, Curtis et al state that therapy should be targeted at correcting relative deficiencies; supraphysiologic concentrations of these vitamins do not appear to be helpful.27 This claim may merit serum studies to rule out deficiencies. Dietitians should be involved in the creation of a patient care plan in the spirit of multidisciplinary orthopedic surgery approaches, which have proven to result in superior patient outcomes.28 Additionally, directive counseling should be provided when necessary.
In patients with adequately managed nutrition, 7 to 10 days of diet optimization is typically sufficient, but patients with malnutrition may require sustained nutritional support for up to 6 weeks; a standardized time for adequate nutrition supplementation has not been identified.25-27 Postoperative management is similar in older patients who are malnourished and those receiving adequate nutrition after orthopedic surgery, which typically involves 3 weeks of a high-protein diet.26
Evaluating Mechanisms of Injury
Assessing the mechanism of injury (MOI) is essential to developing an appropriate and successful orthopedic treatment plan. MOI is typically categorized as low energy, which consists of ground-level falls and other minor trauma, or high energy, which can include motor vehicle crashes or falls from a height.29 Unlike younger patients who typically experience trauma from high-energy MOIs, geriatric patients often sustain fractures from low-energy MOIs. The importance of assessing MOI for the geriatric population is magnified as it provides vital clues that not only help determine the nature of the injury, but also highlight underlying frailty, comorbidities, and potential complications. Weakness or deconditioning related to older age is often not discovered before trauma, which is why assessing the MOI can provide valuable information regarding overall patient health.30

The MOI of trauma also is correlated with factors that influence postoperative recovery and overall prognosis (Figure). Falls comprise more than three-quarters of the MOI in geriatric patients with trauma, and > 90% of these falls are ground-level or other simple falls.30 Falls secondary to an intrinsic disorder, rather than an extrinsic environmental hazard, are more common in geriatric patients.31
These events may be associated with an underlying medical condition, such as osteopenia, osteoporosis, or neuromuscular disorders, such as Parkinson disease.32 They may also be attributed to normal age-related changes, such as decreased visual acuity, reduced reaction time, or mild cognitive impairment.30 An estimated 6% to 35% of geriatric patients who present to the emergency department have some degree of cognitive dysfunction.33 Accordingly, a thorough understanding of the events leading up to injury is vital for the management of older patients. Knowing the specific circumstances of a fall can provide insight into the patient’s gait, balance, and need for further investigations such as cognitive screening or evaluation of home safety. This information can guide decisions regarding preoperative optimization of medications and postoperative rehabilitation interventions.
Frailty and Risk of Fracture
Frailty is a clinical syndrome defined by overall decreased capacity for the body’s adaptive changes to various stressors.34 It is a common condition in geriatric populations due to cumulative degenerative changes and multisystem decline over a lifetime’s worth of disruptions to natural homeostasis.34 In orthopedics, frailty typically refers to musculoskeletal durability and resilience in response to mechanical forces (ie, falls, trauma, and high-acceleration movements). Globally, > 200 million people have osteoporotic frailty, leading to 9 million hip fractures annually.35 More than 30% of people aged ≥ 65 years fall ≥ 1 time per calendar year.36
Assessing frailty in geriatric patients undergoing orthopedic surgery is vital, as it predisposes patients to higher rates of morbidity, mortality, and institutionalization, particularly from falls and resultant fragility fractures.37-39 This is true for a wide range of orthopedic procedures, spanning elective to urgent surgeries and involving the axial and appendicular skeleton.40,41 Given the high rates of fractures, subsequent patient morbidity, and financial burden on the health care system, effective frailty screening is essential.
There are many strategies to assess frailty risk and subsequent fracture risk.42 Questionnaires or online medical calculators serve as easy-to-use tools for assessment of frailty or associated predictors of fragility fractures. Validated assessment tools are provided in Table 1.
Dual-emission X-ray absorptiometry is a well-established way to determine bone density and establish fracture risk. The Fried Frailty Phenotype score and Short Performance Physical Battery test are clinically applicable methods of assessing frailty in older outpatient populations. Although these examinations focus on different aspects of the patient, they have moderate agreeability, are sensitive, and can be readily performed in the clinical setting as demonstrated by a > 90% patient participation rate for both methods.42 Finally, several serum studies can be predictive of frailty, the most readily modifiable of which are vitamin D3, ferritin, albumin, and calcium.43 Although they are more invasive for the patient, serum studies can provide additional modifiable targets for perioperative optimization and contribute to risk stratification.
Risk stratification should take place around 6 weeks before surgery, which should provide adequate time for rectification of preoperative barriers to elective surgical intervention—namely nutritional status. In cases of urgent or emergent procedures (ie, femoral neck fracture with concern for avascular necrosis of the femoral head), this may not be possible but should be conducted nonetheless for patient-specific postoperative rehabilitation and risk reduction.


Postoperative Complication Risks
Postoperative complications affect nearly 15% of geriatric orthopedic patients, highlighting the need for comprehensive preoperative evaluations to assess risk factors.44 Age-related physiological changes, frailty, and comorbidities complicate recovery and management (Table 2).
Wound healing is impaired in older individuals due to suboptimal circulation and decreased oxygenation that is secondary to age-related changes, as well as the increased likelihood of comorbid conditions (eg, diabetes).7 Surgical site infections can be particularly malicious in geriatric patients, with a 4% incidence.45,46 Hospitalization can be prolonged by a mean 2 weeks, which increases the risk of hospital-associated delirium and iatrogenic complications.46 Both the mortality rate and costs associated with hospitalization are higher for older patients who develop surgical site infections compared with patients aged < 65 years, underscoring the importance of vigilant monitoring, early detection, and effective preoperative screening to identify and manage modifiable risk factors.47
Postoperative delirium is another common complication of orthopedic surgery in the geriatric population, increasing morbidity and mortality. The incidence is reported to be as high as 53.3% in the trauma setting and 28.3% in the elective setting, indicating a need to assess patient risk preoperatively.48,49 Several factors contribute to the high incidence of delirium, including advanced age, longer surgical durations, intraoperative hypotension and hypercapnia, pre-existing cognitive dysfunction, and postoperative sleep disorders.50
VTE is another common cause of complications following orthopedic surgery. The development of DVT can lead to subsequent pulmonary embolism, which can be fatal. Orthopedic surgery patients are already at higher risk of DVT and VTE than other surgical patients, with an incidence as high as 40% to 60%, though it is frequently asymptomatic.51,52 Geriatric patients may be more likely to have concurrent comorbidities that increase hypercoagulability.53 Congestive heart failure, chronic kidney disease, and cardiovascular disease are all more common in older individuals and can increase the risk of VTE by 2-fold.53 While anticoagulation is the standard of care to prevent VTE after orthopedic surgery, geriatric patients require more careful monitoring due to the higher incidence of bleeding complications. Additionally, early postoperative mobilization is critical to reduce the risk of DVT without significantly increasing pain or causing other adverse events.54
Respiratory complications are common after orthopedic surgery, particularly atelectasis and bronchospasm, which can result from intraoperative mechanical ventilation.55 While these conditions are typically self-limiting, more severe respiratory issues such as pneumonia are a significant concern because they may lead to the need for mechanical ventilation and admission to the intensive care unit (ICU). The more severe complications have an incidence of about 1% to 2% in orthopedic surgery patients.56 Preventive strategies, such as respiratory physiotherapy and guided breathing exercises, are crucial to minimize perioperative pulmonary complications and promote optimal recovery. Addressing these challenges through early intervention is essential to improve outcomes.
Multidisciplinary Perioperative Care
Multidisciplinary care in orthopedic surgery involves collaborative management of patient care by general practitioners, surgeons, anesthesiologists, dietitians, physical and occupational therapists, inpatient health care practitioners (HCPs), and social services. The goal of this form of care is to provide a longitudinal sequence of health-optimization tactics that prepare a patient for surgery and give them the best chance of postoperative recovery.
Given that many aspects of geriatric health play a role in orthopedic postoperative outcomes, there are many preoperative factors to consider. As previously discussed, preoperative evaluation of geriatric patients should include nutritional and fragility screening to determine surgical candidacy and target modifiable risk factors for risk reduction. This screening can be conducted by primary care practitioners and orthopedic surgeons in an outpatient setting. A multidisciplinary approach benefits patients by decreasing time to surgery.35
Several large studies have demonstrated the positive influence of a multidisciplinary approach on patient-oriented outcomes in orthopedic patients. Incorporation of this style of care in contrast to surgeon-led perioperative optimization leads to fewer floor and ICU admissions, shorter lengths of stay, and decreased mortality rates.35,57 These findings are broadly applicable to a wide range of orthopedic surgeries and even surgeries outside of the musculoskeletal system.58,59 In addition, this strategy has demonstrated reduced in-hospital health care costs due to shorter lengths of stay and fewer ICU admissions. Physical and occupational therapy also have irreplaceable roles in outcomes after orthopedic surgeries. They have independently been shown to decrease pain, increase range of motion, and increase functionality in daily life.60 These aspects of recovery are essential for geriatric well-being.
Screening Tools
The World Health Organization FRAX fracture risk assessment tool (www.fraxplus.org/calculation-tool) was developed to identify patients at high risk of fracture and subsequent complications and to guide clinical decision-making regarding pharmacologic interventions.61 FRAX calculates the 10-year probability of fracture based on demographic factors, such as age and body mass index, clinical measures (eg, femoral neck bone mineral density), and risk factors (eg, prior fragility fractures, substance use history, and prolonged glucocorticoid use).61 The online tool is easy to use, making it a valuable resource for assessing fracture risk and determining appropriate treatment strategies.
The fatigue, resistance, ambulation, illnesses, loss of weight (FRAIL) scale assesses frailty in older adults. The scale classifies patients into 3 categories: robust, prefrail, and frail. The frail category is associated with an increased frequency of hip fracture and an elevated ASA class.62 Additionally, the FRAIL scale has demonstrated value in predicting hospital length of stay and the risk of postoperative complications.62 It also has shown utility in quantifying frailty status, which is traditionally challenging to assess systematically.63
The Mini-Cog is commonly used in geriatric populations to screen for cognitive impairment. Preoperative Mini-Cog screening has been shown to predict the development of postoperative complications.64 Geriatric patients who screened positive for cognitive impairment prior to orthopedic surgery were more likely to develop postoperative delirium, require alternative discharge disposition, and have a longer hospital length of stay.64 Mini-Cog serves as an important preoperative tool for identifying patients who may benefit from closer postoperative monitoring and tailored care.
The Comprehensive Geriatric Assessment (CGA) is a multidimensional evaluation that has been validated for use in geriatric patients undergoing orthopedic surgery.65 The CGA assesses functional status and the ability to perform activities of daily living (ADLs), such as eating, dressing, and ambulating. Poor ADLs are associated with increased risk of falls and cardiopulmonary complications. The CGA allows HCPs to identify patients at higher risk of complications and tailor interventions that optimize functional recovery during the perioperative period.
Nutritional screening is another component of preoperative evaluation in older adults undergoing orthopedic surgery. The Perioperative Nutrition Screen is a preoperative phone assessment of unintentional weight loss in the past 6 months.66 Patients who screen positive are asked to come in for a preoperative visit with a registered dietitian who can further evaluate the nutritional status of the patient.
The Mini Nutritional Assessment Short Form (MNA-SF), Malnutrition Universal Screening Tool, and Nutrition Risk Screening 2002 have all been validated for use in older patients undergoing orthopedic surgery. Among these, the MNA-SF has demonstrated superior utility in predicting hospital readmission and mortality.67 Given the established links between malnutrition and poor surgical outcomes, routine nutritional screening is important for identifying geriatric patients who may require preoperative nutritional interventions.
CONCLUSIONS AND RECOMMENDATIONS
Perioperative management of geriatric patients undergoing orthopedic surgery requires an assessment and strategy focused on risk stratification, patient optimization, and mitigation of potential complications and mortality. Due to the complexity and comprehensive nature of an optimal perioperative plan, creating the plan early is essential to ensure adequate time for patient optimization and care coordination.
Nutrition plays a critical role in the success of surgical procedures, and orthopedics is no exception. Extra care should be taken to preoperatively optimize patient bone health before surgical intervention to enhance recovery and reduce the risk of complications. After an appropriate patient history and clinical picture are gathered, screening tools should be used on a case-by-case basis to further characterize comorbid conditions that may contribute to suboptimal outcomes. Additionally, given the proven association between frailty and fracture risk, frailty serves as a readily quantifiable predictor of patient-oriented outcomes. This should be assessed preoperatively with appropriate risk-stratification tools to determine appropriate postoperative measures to prevent morbidity and mortality.
Orthopedic surgery is increasingly common in geriatric patients, who face higher perioperative risks due to age-related physiological changes, multimorbidity, and frailty. Optimizing preoperative assessment and adopting a multidisciplinary approach—integrating surgeons, anesthesiologists, physical therapists, and dietitians—can improve outcomes, reduce complications, and enhance recovery. The successful use of the tools and strategies outlined in this article by primary care should facilitate access to and recovery from orthopedic surgery in the geriatric population.
More than 40 million surgeries are performed annually in the United States, of which > 18 million are orthopedic, including > 1 million emergency orthopedic surgeries and > 2 million joint replacements.1-4 Notably, > 50% of patients undergoing orthopedic surgery are aged ≥ 65 years, a demographic shift driven by longer life expectancies and an increasing number of older adults remaining physically active for extended periods.5 Osteoarthritis, the most common joint disease, affects 10% of men and 18% of women aged > 60 years, often necessitating an orthopedic joint replacement.6 Perioperative morbidity and mortality are 2.9- to 6.7-times higher in older adults compared with younger adults.7 These risks include infection, venous thromboembolism (VTE), pressure ulcers, reduced mobility, and increased mortality. Due to the high incidence of these complications in older surgical patients, special perioperative protocols and considerations are needed when preparing an older patient for surgery. This review aims to establish concrete considerations and guidelines for perioperative management.
METHODOLOGY
A literature review of PubMed, Google Scholar, and IEEE Xplore identified research on perioperative challenges in geriatric orthopedic surgery. Keywords included geriatrics and orthopedic surgery, perioperative care in geriatric populations, and orthopedic perioperative care. Inclusion criteria were strictly defined to ensure relevance to the geriatric population, with studies focusing on patients aged ≥ 65 years. Exclusion criteria were applied to remove studies that did not involve geriatric populations or orthopedic surgeries or that lacked a clear perioperative focus. Studies were analyzed for design, interventions, and outcomes. Special attention was given to identifying common challenges and trends related to perioperative considerations. We developed a narrative report providing a comprehensive overview of the current understanding of perioperative care for geriatric orthopedic patients to offer practical recommendations for clinicians to use in their practice.
RESULTS
Consistent with the narrative review methodology described, the literature search yielded a broad range of publications addressing perioperative considerations in geriatric orthopedic patients. Articles were screened for relevance to patients aged ≥ 65 years undergoing orthopedic surgery and for applicability to perioperative optimization and postoperative outcomes. Given the heterogeneity in study design, population characteristics, and outcome reporting, findings are presented descriptively rather than being quantitatively pooled. Studies not focused on geriatric populations, orthopedic procedures, or perioperative management were excluded. Key themes included multimorbidity and comorbidity optimization, age-related physiologic changes, frailty assessment and fracture risk stratification, nutritional and bone health management, mechanism of injury considerations, prevention of postoperative complications, and the role of multidisciplinary perioperative care.
Unique Physiological Challenges
The aging process induces a range of physiological changes that can increase morbidity and mortality following surgery. One of the most essential elements to surgical recovery is wound healing, as impairments in this process can lead to adverse events, including infection, cosmetic deformity, and wound dehiscence. The general paradigm of aging involves cell senescence resulting in slower or disorganized functional capacity of these cells.8 While wound healing in older individuals was once thought to be defective, recent research has demonstrated that this process is not absent, but delayed.9
Wound healing is a tightly regulated and evolutionarily conserved process that proceeds through 3 main phases: inflammation, proliferation, and remodeling. Re-epithelialization begins with the migration of epithelial cells from hair follicles, sweat glands, or wound margins (depending on wound depth) and is influenced by oxygen levels, moisture, and growth factors.9 Several characteristics of aged skin contribute to the delayed healing process. Aged skin has fewer hair follicles and eccrine sweat glands, as well as decreased follicle thickness.10 This results in fewer proliferating cells for wound healing and lower amounts of sebum production for skin moisture.11 Furthermore, aged fibroblasts are fewer in number and less effective in synthesizing extracellular matrices, resulting in slower and less tensile wound healing.12 Additionally, microvascular changes associated with aging result in disorganized vasculature, which impairs oxygen delivery to the wound bed and diminishes the influx of proinflammatory cells necessary for effective healing.13 These senescent traits of aged skin contribute to the delayed wound healing process found in geriatric patients.
Compounding these age-related factors is the prevalence of multimorbidity, or coexisting chronic diagnoses, in 55% to 98% of older patients.14 Common comorbidities include peripheral arterial disease, chronic venous insufficiency, type 1 and type 2 diabetes, neoplasms, atherosclerotic disease, and hypertension. Older patients are more likely to be prescribed corticosteroids and chemotherapeutic agents that impair the function of inflammatory cells necessary for wound healing.15,16 Additionally, decreased mobility is more common in geriatric patients, which can increase the risk of wound formation, particularly pressure ulcers.17
Perioperative Considerations
All surgical patients undergo a formal or informal preoperative evaluation to assess their fitness for surgery, with the goal of minimizing both anesthesia-related risks and postoperative complications. A widely used tool in this assessment is the American Society of Anesthesiologists (ASA) physical status classification, which stratifies patients into 6 categories based on their medical history and overall health status.18 Classes range from healthy patients (Class I) to organ donors who are brain-dead (Class VI).
Cardiac optimization is an essential component of preoperative evaluation for older adults due to their higher risk of underlying cardiovascular disease.19 This process involves an in-depth review of the patient’s cardiac history, including the timing and nature of any prior interventions and the recurrence rate. Functional capacity is assessed through metabolic equivalents, where a threshold of > 4 metabolic equivalents (the ability to walk up a flight of stairs) is considered adequate for surgery. Risk is assessed based on the specific surgical procedure, and nonemergent orthopedic procedures are considered intermediate risk. If a patient is deemed high risk at any stage of this evaluation, further cardiac testing is indicated.
Pulmonary optimization is typically necessary for geriatric patients, who are more likely to have conditions such as chronic obstructive pulmonary disease or interstitial lung disease.14,20 In patients without severe systemic lung disease, pulmonary optimization involves assessing the functional expiratory volume and diffusing capacity for carbon monoxide. In addition, aggressive modification of risk factors, such as smoking cessation, is strongly recommended.
Additional perioperative conditions are disease-specific and involve evaluation of comorbid illnesses and recognition of absolute contraindications to noncardiac surgery. For instance, an ejection fraction of < 35%, a history of myocardial infarction within 6 months, or active diabetic ketoacidosis are all absolute contraindications to elective surgery. For orthopedic procedures, additional contraindications include symptomatic bacteremia, active joint or local tissue infection, severe malnutrition, uncontrolled metabolic syndrome or chronic disease, untreated immunodeficiency, and active deep venous thrombosis (DVT) or pulmonary embolism.21
Bone Health and Nutrition
In the context of orthopedic surgery, the hallmark of clinically defined optimal bone health is a musculoskeletal system that provides the ability for pain-free functional and occupational tasks with an adequate capacity to withstand the mechanical forces imparted by everyday life. Back pain and arthritis are the fourth- and sixth-most common complaints in primary care, underscoring suboptimal bone health management in developed countries.22
Optimizing bone health through proper nutrition is crucial in the perioperative management of geriatric orthopedic patients. The clinical diagnosis of malnutrition has well-studied associations with worse outcomes after orthopedic surgery, which include increased mortality, hospital length of stay, readmission rates, and health institution spending.23-25 Some studies show that up to 60% of geriatric patients may be malnourished.26
Regarding vitamin and mineral supplements, the general consensus before orthopedic surgery is that vitamins A, C, D, and E, and zinc are predictive in determining postoperative health.27 However, Curtis et al state that therapy should be targeted at correcting relative deficiencies; supraphysiologic concentrations of these vitamins do not appear to be helpful.27 This claim may merit serum studies to rule out deficiencies. Dietitians should be involved in the creation of a patient care plan in the spirit of multidisciplinary orthopedic surgery approaches, which have proven to result in superior patient outcomes.28 Additionally, directive counseling should be provided when necessary.
In patients with adequately managed nutrition, 7 to 10 days of diet optimization is typically sufficient, but patients with malnutrition may require sustained nutritional support for up to 6 weeks; a standardized time for adequate nutrition supplementation has not been identified.25-27 Postoperative management is similar in older patients who are malnourished and those receiving adequate nutrition after orthopedic surgery, which typically involves 3 weeks of a high-protein diet.26
Evaluating Mechanisms of Injury
Assessing the mechanism of injury (MOI) is essential to developing an appropriate and successful orthopedic treatment plan. MOI is typically categorized as low energy, which consists of ground-level falls and other minor trauma, or high energy, which can include motor vehicle crashes or falls from a height.29 Unlike younger patients who typically experience trauma from high-energy MOIs, geriatric patients often sustain fractures from low-energy MOIs. The importance of assessing MOI for the geriatric population is magnified as it provides vital clues that not only help determine the nature of the injury, but also highlight underlying frailty, comorbidities, and potential complications. Weakness or deconditioning related to older age is often not discovered before trauma, which is why assessing the MOI can provide valuable information regarding overall patient health.30

The MOI of trauma also is correlated with factors that influence postoperative recovery and overall prognosis (Figure). Falls comprise more than three-quarters of the MOI in geriatric patients with trauma, and > 90% of these falls are ground-level or other simple falls.30 Falls secondary to an intrinsic disorder, rather than an extrinsic environmental hazard, are more common in geriatric patients.31
These events may be associated with an underlying medical condition, such as osteopenia, osteoporosis, or neuromuscular disorders, such as Parkinson disease.32 They may also be attributed to normal age-related changes, such as decreased visual acuity, reduced reaction time, or mild cognitive impairment.30 An estimated 6% to 35% of geriatric patients who present to the emergency department have some degree of cognitive dysfunction.33 Accordingly, a thorough understanding of the events leading up to injury is vital for the management of older patients. Knowing the specific circumstances of a fall can provide insight into the patient’s gait, balance, and need for further investigations such as cognitive screening or evaluation of home safety. This information can guide decisions regarding preoperative optimization of medications and postoperative rehabilitation interventions.
Frailty and Risk of Fracture
Frailty is a clinical syndrome defined by overall decreased capacity for the body’s adaptive changes to various stressors.34 It is a common condition in geriatric populations due to cumulative degenerative changes and multisystem decline over a lifetime’s worth of disruptions to natural homeostasis.34 In orthopedics, frailty typically refers to musculoskeletal durability and resilience in response to mechanical forces (ie, falls, trauma, and high-acceleration movements). Globally, > 200 million people have osteoporotic frailty, leading to 9 million hip fractures annually.35 More than 30% of people aged ≥ 65 years fall ≥ 1 time per calendar year.36
Assessing frailty in geriatric patients undergoing orthopedic surgery is vital, as it predisposes patients to higher rates of morbidity, mortality, and institutionalization, particularly from falls and resultant fragility fractures.37-39 This is true for a wide range of orthopedic procedures, spanning elective to urgent surgeries and involving the axial and appendicular skeleton.40,41 Given the high rates of fractures, subsequent patient morbidity, and financial burden on the health care system, effective frailty screening is essential.
There are many strategies to assess frailty risk and subsequent fracture risk.42 Questionnaires or online medical calculators serve as easy-to-use tools for assessment of frailty or associated predictors of fragility fractures. Validated assessment tools are provided in Table 1.
Dual-emission X-ray absorptiometry is a well-established way to determine bone density and establish fracture risk. The Fried Frailty Phenotype score and Short Performance Physical Battery test are clinically applicable methods of assessing frailty in older outpatient populations. Although these examinations focus on different aspects of the patient, they have moderate agreeability, are sensitive, and can be readily performed in the clinical setting as demonstrated by a > 90% patient participation rate for both methods.42 Finally, several serum studies can be predictive of frailty, the most readily modifiable of which are vitamin D3, ferritin, albumin, and calcium.43 Although they are more invasive for the patient, serum studies can provide additional modifiable targets for perioperative optimization and contribute to risk stratification.
Risk stratification should take place around 6 weeks before surgery, which should provide adequate time for rectification of preoperative barriers to elective surgical intervention—namely nutritional status. In cases of urgent or emergent procedures (ie, femoral neck fracture with concern for avascular necrosis of the femoral head), this may not be possible but should be conducted nonetheless for patient-specific postoperative rehabilitation and risk reduction.


Postoperative Complication Risks
Postoperative complications affect nearly 15% of geriatric orthopedic patients, highlighting the need for comprehensive preoperative evaluations to assess risk factors.44 Age-related physiological changes, frailty, and comorbidities complicate recovery and management (Table 2).
Wound healing is impaired in older individuals due to suboptimal circulation and decreased oxygenation that is secondary to age-related changes, as well as the increased likelihood of comorbid conditions (eg, diabetes).7 Surgical site infections can be particularly malicious in geriatric patients, with a 4% incidence.45,46 Hospitalization can be prolonged by a mean 2 weeks, which increases the risk of hospital-associated delirium and iatrogenic complications.46 Both the mortality rate and costs associated with hospitalization are higher for older patients who develop surgical site infections compared with patients aged < 65 years, underscoring the importance of vigilant monitoring, early detection, and effective preoperative screening to identify and manage modifiable risk factors.47
Postoperative delirium is another common complication of orthopedic surgery in the geriatric population, increasing morbidity and mortality. The incidence is reported to be as high as 53.3% in the trauma setting and 28.3% in the elective setting, indicating a need to assess patient risk preoperatively.48,49 Several factors contribute to the high incidence of delirium, including advanced age, longer surgical durations, intraoperative hypotension and hypercapnia, pre-existing cognitive dysfunction, and postoperative sleep disorders.50
VTE is another common cause of complications following orthopedic surgery. The development of DVT can lead to subsequent pulmonary embolism, which can be fatal. Orthopedic surgery patients are already at higher risk of DVT and VTE than other surgical patients, with an incidence as high as 40% to 60%, though it is frequently asymptomatic.51,52 Geriatric patients may be more likely to have concurrent comorbidities that increase hypercoagulability.53 Congestive heart failure, chronic kidney disease, and cardiovascular disease are all more common in older individuals and can increase the risk of VTE by 2-fold.53 While anticoagulation is the standard of care to prevent VTE after orthopedic surgery, geriatric patients require more careful monitoring due to the higher incidence of bleeding complications. Additionally, early postoperative mobilization is critical to reduce the risk of DVT without significantly increasing pain or causing other adverse events.54
Respiratory complications are common after orthopedic surgery, particularly atelectasis and bronchospasm, which can result from intraoperative mechanical ventilation.55 While these conditions are typically self-limiting, more severe respiratory issues such as pneumonia are a significant concern because they may lead to the need for mechanical ventilation and admission to the intensive care unit (ICU). The more severe complications have an incidence of about 1% to 2% in orthopedic surgery patients.56 Preventive strategies, such as respiratory physiotherapy and guided breathing exercises, are crucial to minimize perioperative pulmonary complications and promote optimal recovery. Addressing these challenges through early intervention is essential to improve outcomes.
Multidisciplinary Perioperative Care
Multidisciplinary care in orthopedic surgery involves collaborative management of patient care by general practitioners, surgeons, anesthesiologists, dietitians, physical and occupational therapists, inpatient health care practitioners (HCPs), and social services. The goal of this form of care is to provide a longitudinal sequence of health-optimization tactics that prepare a patient for surgery and give them the best chance of postoperative recovery.
Given that many aspects of geriatric health play a role in orthopedic postoperative outcomes, there are many preoperative factors to consider. As previously discussed, preoperative evaluation of geriatric patients should include nutritional and fragility screening to determine surgical candidacy and target modifiable risk factors for risk reduction. This screening can be conducted by primary care practitioners and orthopedic surgeons in an outpatient setting. A multidisciplinary approach benefits patients by decreasing time to surgery.35
Several large studies have demonstrated the positive influence of a multidisciplinary approach on patient-oriented outcomes in orthopedic patients. Incorporation of this style of care in contrast to surgeon-led perioperative optimization leads to fewer floor and ICU admissions, shorter lengths of stay, and decreased mortality rates.35,57 These findings are broadly applicable to a wide range of orthopedic surgeries and even surgeries outside of the musculoskeletal system.58,59 In addition, this strategy has demonstrated reduced in-hospital health care costs due to shorter lengths of stay and fewer ICU admissions. Physical and occupational therapy also have irreplaceable roles in outcomes after orthopedic surgeries. They have independently been shown to decrease pain, increase range of motion, and increase functionality in daily life.60 These aspects of recovery are essential for geriatric well-being.
Screening Tools
The World Health Organization FRAX fracture risk assessment tool (www.fraxplus.org/calculation-tool) was developed to identify patients at high risk of fracture and subsequent complications and to guide clinical decision-making regarding pharmacologic interventions.61 FRAX calculates the 10-year probability of fracture based on demographic factors, such as age and body mass index, clinical measures (eg, femoral neck bone mineral density), and risk factors (eg, prior fragility fractures, substance use history, and prolonged glucocorticoid use).61 The online tool is easy to use, making it a valuable resource for assessing fracture risk and determining appropriate treatment strategies.
The fatigue, resistance, ambulation, illnesses, loss of weight (FRAIL) scale assesses frailty in older adults. The scale classifies patients into 3 categories: robust, prefrail, and frail. The frail category is associated with an increased frequency of hip fracture and an elevated ASA class.62 Additionally, the FRAIL scale has demonstrated value in predicting hospital length of stay and the risk of postoperative complications.62 It also has shown utility in quantifying frailty status, which is traditionally challenging to assess systematically.63
The Mini-Cog is commonly used in geriatric populations to screen for cognitive impairment. Preoperative Mini-Cog screening has been shown to predict the development of postoperative complications.64 Geriatric patients who screened positive for cognitive impairment prior to orthopedic surgery were more likely to develop postoperative delirium, require alternative discharge disposition, and have a longer hospital length of stay.64 Mini-Cog serves as an important preoperative tool for identifying patients who may benefit from closer postoperative monitoring and tailored care.
The Comprehensive Geriatric Assessment (CGA) is a multidimensional evaluation that has been validated for use in geriatric patients undergoing orthopedic surgery.65 The CGA assesses functional status and the ability to perform activities of daily living (ADLs), such as eating, dressing, and ambulating. Poor ADLs are associated with increased risk of falls and cardiopulmonary complications. The CGA allows HCPs to identify patients at higher risk of complications and tailor interventions that optimize functional recovery during the perioperative period.
Nutritional screening is another component of preoperative evaluation in older adults undergoing orthopedic surgery. The Perioperative Nutrition Screen is a preoperative phone assessment of unintentional weight loss in the past 6 months.66 Patients who screen positive are asked to come in for a preoperative visit with a registered dietitian who can further evaluate the nutritional status of the patient.
The Mini Nutritional Assessment Short Form (MNA-SF), Malnutrition Universal Screening Tool, and Nutrition Risk Screening 2002 have all been validated for use in older patients undergoing orthopedic surgery. Among these, the MNA-SF has demonstrated superior utility in predicting hospital readmission and mortality.67 Given the established links between malnutrition and poor surgical outcomes, routine nutritional screening is important for identifying geriatric patients who may require preoperative nutritional interventions.
CONCLUSIONS AND RECOMMENDATIONS
Perioperative management of geriatric patients undergoing orthopedic surgery requires an assessment and strategy focused on risk stratification, patient optimization, and mitigation of potential complications and mortality. Due to the complexity and comprehensive nature of an optimal perioperative plan, creating the plan early is essential to ensure adequate time for patient optimization and care coordination.
Nutrition plays a critical role in the success of surgical procedures, and orthopedics is no exception. Extra care should be taken to preoperatively optimize patient bone health before surgical intervention to enhance recovery and reduce the risk of complications. After an appropriate patient history and clinical picture are gathered, screening tools should be used on a case-by-case basis to further characterize comorbid conditions that may contribute to suboptimal outcomes. Additionally, given the proven association between frailty and fracture risk, frailty serves as a readily quantifiable predictor of patient-oriented outcomes. This should be assessed preoperatively with appropriate risk-stratification tools to determine appropriate postoperative measures to prevent morbidity and mortality.
Orthopedic surgery is increasingly common in geriatric patients, who face higher perioperative risks due to age-related physiological changes, multimorbidity, and frailty. Optimizing preoperative assessment and adopting a multidisciplinary approach—integrating surgeons, anesthesiologists, physical therapists, and dietitians—can improve outcomes, reduce complications, and enhance recovery. The successful use of the tools and strategies outlined in this article by primary care should facilitate access to and recovery from orthopedic surgery in the geriatric population.
Dobson GP. Trauma of major surgery: a global problem that is not going away. Int J Surg. 2020;81:47-54. doi:10.1016/j.ijsu.2020.07.017
United States (US) orthopedic procedures count by segments and forecast to 2030. GlobalData. February 17, 2023. Accessed April 29, 2026. https://www.globaldata.com/store/report/usa-orthopedic-procedures-analysis/
Jarman MP, Weaver MJ, Haider AH, Salim A, Harris MB. The national burden of orthopedic injury: cross-sectional estimates for trauma system planning and optimization. J Surg Res. 2020;249:197-204. doi:10.1016/j.jss.2019.12.023
Hegde V, Stambough JB, Levine BR, et al. Highlights of the 2022 American Joint Replacement Registry Annual Report. Arthroplast Today. 2023;21:101137. doi:10.1016/j.artd.2023.101137
Nakamura K, Ogata T. Locomotive syndrome: definition and management. Clin Rev Bone Miner Metab. 2016;14:56-67. doi:10.1007/s12018-016-9208-2
Glyn-Jones S, Palmer AJR, Agricola R, et al. Osteoarthritis. Lancet. 2015;386:376-387. doi:10.1016/S0140-6736(14)60802-3
Hughes S, Leary A, Zweizig S, Cain J. Surgery in elderly people: preoperative, operative and postoperative care to assist healing. Best Pract Res Clin Obstet Gynaecol. 2013;27:753-765. doi:10.1016/j.bpobgyn.2013.02.006
Regulski MJ. Cellular senescence: what, why, and how. Wounds. 2017;29:168-174.
Kremer M, Burkemper N. Aging skin and wound healing. Clin Geriatr Med. 2024;40:1-10. doi:10.1016/j.cger.2023.06.001
Fenske NA, Lober CW. Structural and functional changes of normal aging skin. J Am Acad Dermatol. 1986;15:571-585. doi:10.1016/S0190-9622(86)70208-9
Van Neste D, Tobin DJ. Hair cycle and hair pigmentation: dynamic interactions and changes associated with aging. Micron. 2004;35:193-200. doi:10.1016/j.micron.2003.11.006
Salzer MC, Lafzi A, Berenguer-Llergo A, et al. Identity noise and adipogenic traits characterize dermal fibroblast aging. Cell. 2018;175:1575-1590.e22. doi:10.1016/j.cell.2018.10.012
Jin K. A microcirculatory theory of aging. Aging Dis. 2019;10:676-683. doi:10.14336/AD.2019.0315
Marengoni A, Angleman S, Melis R, et al. Aging with multimorbidity: a systematic review of the literature. Ageing Res Rev. 2011;10:430-439. doi:10.1016/j.arr.2011.03.003
Waljee AK, Rogers MAM, Lin P, et al. Short term use of oral corticosteroids and related harms among adults in the United States: population based cohort study. BMJ. 2017;357:j1415. doi:10.1136/bmj.j1415
Given B, Given CW. Older adults and cancer treatment. Cancer. 2008;113:3505-3511. doi:10.1002/cncr.23939
Ferrucci L, Cooper R, Shardell M, Simonsick EM, Schrack JA, Kuh D. Age-related change in mobility: perspectives from life course epidemiology and geroscience. J Gerontol A Biol Sci Med Sci. 2016;71:1184-1194. doi:10.1093/gerona/glw043
Mayhew D, Mendonca V, Murthy BVS. A review of ASA physical status - historical perspectives and modern developments. Anaesthesia. 2019;74:373-379. doi:10.1111/anae.14569
Eagle KA, Berger PB, Calkins H, et al. ACC/AHA guideline update for perioperative cardiovascular evaluation for noncardiac surgery—executive summary a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee to Update the 1996 Guidelines on Perioperative Cardiovascular Evaluation for Noncardiac Surgery). Circulation. 2002;105:1257-1267. doi:10.1161/circ.105.10.1257
Bapoje SR, Whitaker JF, Schulz T, Chu ES, Albert RK. Preoperative evaluation of the patient with pulmonary disease. Chest. 2007;132:1637-1645. doi:10.1378/chest.07-0347
Choe H, Indelli PF, Ricciardi B, et al. What are the absolute contraindications for elective total knee or hip arthroplasty? J Arthroplasty. 2025;40(2 suppl 1):S45-S47. doi:10.1016/j.arth.2024.10.041
Finley CR, Chan DS, Garrison S, et al. What are the most common conditions in primary care? Systematic review. Can Fam Physician. 2018;64:832-840.
Vaid S, Bell T, Grim R, Ahuja V. Predicting risk of death in general surgery patients on the basis of preoperative variables using American College of Surgeons National Surgical Quality Improvement Program data. Perm J. 2012;16:10-17. doi:10.7812/TPP/12-019
Correia MTD, Waitzberg DL. The impact of malnutrition on morbidity, mortality, length of hospital stay and costs evaluated through a multivariate model analysis. Clin Nutr. 2003;22:235-239. doi:10.1016/S0261-5614(02)00215-7
Friedman J, Lussiez A, Sullivan J, Wang S, Englesbe M. Implications of sarcopenia in major surgery. Nutr Clin Pract. 2015;30:175-179. doi:10.1177/0884533615569888
Hirsch KR, Wolfe RR, Ferrando AA. Pre- and post-surgical nutrition for preservation of muscle mass, strength, and functionality following orthopedic surgery. Nutrients. 2021;13:1675. doi:10.3390/nu13051675
Curtis W, Choi T, Ahmad A, Shultz C. Perioperative nutritional considerations in orthopaedic surgery: a review of the literature. West J Orthop. 2023;12:1. https://digitalrepository.unm.edu/wjo/vol12/iss1/1
Wischmeyer PE, Carli F, Evans DC, et al. American Society for Enhanced Recovery and Perioperative Quality Initiative joint consensus statement on nutrition screening and therapy within a surgical enhanced recovery pathway. Anesth Analg. 2018;126:1883-1895. doi:10.1213/ANE.0000000000002743
Mun F, Ringenbach K, Baer B, et al. Factors influencing geriatric orthopaedic trauma mortality. Injury. 2022;53:919-924. doi:10.1016/j.injury.2022.01.005
Bonne S, Schuerer DJE. Trauma in the older adult: epidemiology and evolving geriatric trauma principles. Clin Geriatr Med. 2013;29:137-150. doi:10.1016/j.cger.2012.10.008
Montero-Odasso MM. Falls as a geriatric syndrome: mechanisms and risk identification. In: Duque G, Kiel DP, eds. Osteoporosis in Older Persons: Advances in Pathophysiology and Therapeutic Approaches. 2nd ed. Springer International Publishing; 2016:171-186. doi:10.1007/978-3-319-25976-5_10
Lach HW, Reed AT, Arfken CL, et al. Falls in the elderly: reliability of a classification system. J Am Geriatr Soc. 1991;39:197-202. doi:10.1111/j.1532-5415.1991.tb01626.x
Carpenter CR, DesPain B, Keeling TN, Shah M, Rothenberger M. The six-item screener and AD8 for the detection of cognitive impairment in geriatric emergency department patients. Ann Emerg Med. 2011;57:653-661. doi:10.1016/j.annemergmed.2010.06.560
Clegg A, Young J, Iliffe S, Rikkert MO, Rockwood K. Frailty in elderly people. Lancet. 2013;381:752-762. doi:10.1016/S0140-6736(12)62167-9
Patel JN, Klein DS, Sreekumar S, Liporace FA, Yoon RS. Outcomes in multidisciplinary team-based approach in geriatric hip fracture care: a systematic review. J Am Acad Orthop Surg. 2020;28:128-133. doi:10.5435/JAAOS-D-18-00425
Amador LF, Loera JA. Preventing postoperative falls in the older adult. J Am Coll Surg. 2007;204:447-453. doi:10.1016/j.jamcollsurg.2006.12.010
Tembo MC, Holloway-Kew KL, Mohebbi M, et al. The association between a fracture risk tool and frailty: Geelong Osteoporosis Study. BMC Geriatr. 2020;20:196. doi:10.1186/s12877-020-01595-8
Demiris¸ B, Basat S, Kurt F, Aksakal B, Basat O. Evaluation of the relationship between frailty and fracture risk using Fracture Risk Assessment Tool in patients 65 years and over. South Clin Istanb Eurasia. 2023;34:42-48. doi:10.14744/scie.2022.66564
Partridge JSL, Harari D, Dhesi JK. Frailty in the older surgical patient: a review. Age Ageing. 2012;41:142-147. doi:10.1093/ageing/afr182
Mamtora PH, Fortier MA, Barnett SR, Schmid LN, Kain ZN. Peri-operative management of frailty in the orthopedic patient. J Orthop. 2020;22:304-307. doi:10.1016/j.jor.2020.05.024
Leven DM, Lee NJ, Kim JS, et al. Frailty is predictive of adverse postoperative events in patients undergoing lumbar fusion. Global Spine J. 2017;7:529-535. doi:10.1177/2192568217700099
Pritchard JM, Kennedy CC, Karampatos S, et al. Measuring frailty in clinical practice: a comparison of physical frailty assessment methods in a geriatric out-patient clinic. BMC Geriatr. 2017;17:264. doi:10.1186/s12877-017-0623-0
Kumar A, Dhar M, Agarwal M, Mukherjee A, Saxena V. Predictors of frailty in the elderly population: a cross-sectional study at a tertiary care center. Cureus. 2022;14:e30557. doi:10.7759/cureus.30557
Scarano KA, Philp FH, Westrick ER, Altman GT, Altman DT. Evaluating postoperative complications and outcomes of orthopedic fracture repair in nonagenarian patients. Geriatr Orthop Surg Rehabil. 2018;9:2151459318758106. doi:10.1177/2151459318758106
Liang Z, Rong K, Gu W, et al. Surgical site infection following elective orthopaedic surgeries in geriatric patients: incidence and associated risk factors. Int Wound J. 2019;16:773-780. doi:10.1111/iwj.13096
Ren M, Liang W, Wu Z, Zhao H, Wang J. Risk factors of surgical site infection in geriatric orthopedic surgery: a retrospective multicenter cohort study. Geriatr Gerontol Int. 2019;19:213-217. doi:10.1111/ggi.13590
Kaye KS, Schmader KE, Sawyer R. Surgical site infection in the elderly population. Clin Infect Dis. 2004;39:1835-1841. doi:10.1086/425744
Bruce AJ, Ritchie CW, Blizard R, Lai R, Raven P. The incidence of delirium associated with orthopedic surgery: a meta-analytic review. Int Psychogeriatr. 2007;19:197-214. doi:10.1017/S104161020600425X
Williams-Russo P, Urquhart BL, Sharrock NE, Charlson ME. Post-operative delirium: predictors and prognosis in elderly orthopedic patients. J Am Geriatr Soc. 1992;40:759-767. doi:10.1111/j.1532-5415.1992.tb01846.x
Wang J, Li Z, Yu Y, Li B, Shao G, Wang Q. Risk factors contributing to postoperative delirium in geriatric patients postorthopedic surgery. Asia Pac Psychiatry. 2015;7:375-382. doi:10.1111/appy.12193
Geerts WH, Pineo GF, Heit JA, et al. Prevention of venous thromboembolism: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest. 2004;126:338S-400S. doi:10.1378/chest.126.3_suppl.338S
Kahn SR, Shivakumar S. What’s new in VTE risk and prevention in orthopedic surgery. Res Pract Thromb Haemost. 2020;4:366-376. doi:10.1002/rth2.12323
Uzel K, Azboy I·, Parvizi J. Venous thromboembolism in orthopedic surgery: global guidelines. Acta Orthop Traumatol Turc. 2023;57:192-203. doi:10.5152/j.aott.2023.23074
Peck M, Holthaus A, Kingsbury K, Salsberry MG, Duggirala V. Mobility in acute care for geriatric patients with orthopedic conditions: a review of recent literature. Curr Geri Rep. 2020;9:300-310. doi:10.1007/s13670-020-00347-1
Leme LEG, Sitta MC, Toledo M, Henriques SS. Orthopedic surgery among the elderly: clinical characteristics. Rev Bras Ortop. 2015;46:238-246. doi:10.1016/S2255-4971(15)30189-0
Malcolm TL, Knezevic NN, Zouki CC, Tharian AR. Pulmonary complications after hip and knee arthroplasty in the United States, 2004-2014. Anesth Analg. 2020;130:917-924. doi:10.1213/ANE.0000000000004265
Kamal T, Conway RM, Littlejohn I, Ricketts D. The role of a multidisciplinary pre-assessment clinic in reducing mortality after complex orthopaedic surgery. Ann R Coll Surg Engl. 2011;93:149-151. doi:10.1308/003588411X561026
Davis MJ, Luu BC, Raj S, Abu-Ghname A, Buchanan EP. Multidisciplinary care in surgery: Are team-based interventions cost-effective? Surgeon. 2021;19:49-60. doi:10.1016/j.surge.2020.02.005
Frassanito L, Vergari A, Nestorini R, et al. Enhanced recovery after surgery (ERAS) in hip and knee replacement surgery: description of a multidisciplinary program to improve management of the patients undergoing major orthopedic surgery. Musculoskelet Surg. 2020;104:87-92. doi:10.1007/s12306-019-00603-4
Reddy RS, Alahmari KA, Alshahrani MS, et al. Exploring the impact of physiotherapy on health outcomes in older adults with chronic diseases: a cross-sectional analysis. Front Public Health. 2024;12:1415882. doi:10.3389/fpubh.2024.1415882
Watts NB. The Fracture Risk Assessment Tool (FRAX®): applications in clinical practice. J Womens Health (Larchmt). 2011;20:525-531. doi:10.1089/jwh.2010.2294
Gleason LJ, Benton EA, Alvarez-Nebreda ML, Weaver MJ, Harris MB, Javedan H. FRAIL questionnaire screening tool and short-term outcomes in geriatric fracture patients. J Am Med Dir Assoc. 2017;18:1082-1086. doi:10.1016/j.jamda.2017.07.005
Kojima G. Frailty defined by FRAIL scale as a predictor of mortality: a systematic review and meta-analysis. J Am Med Dir Assoc. 2018;19:480-483. doi:10.1016/j.jamda.2018.04.006
Culley DJ, Flaherty D, Fahey MC, et al. Poor performance on a preoperative cognitive screening test predicts postoperative complications in older orthopedic surgical patients. Anesthesiology. 2017;127:765-774. doi:10.1097/ALN.0000000000001859
Kong C, Zhang Y, Wang C, et al. Comprehensive geriatric assessment for older orthopedic patients and analysis of risk factors for postoperative complications. BMC Geriatr. 2022;22:644. doi:10.1186/s12877-022-03328-5
Williams DGA, Wischmeyer PE. Perioperative nutrition care of orthopedic surgery patient. Tech Orthop. 2020;35:15-18. doi:10.1097/BTO.0000000000000412
Koren-Hakim T, Weiss A, Hershkovitz A, et al. Comparing the adequacy of the MNA-SF, NRS-2002 and MUST nutritional tools in assessing malnutrition in hip fracture operated elderly patients. Clin Nutr. 2016;35:1053-1058. doi:10.1016/j.clnu.2015.07.014
Dobson GP. Trauma of major surgery: a global problem that is not going away. Int J Surg. 2020;81:47-54. doi:10.1016/j.ijsu.2020.07.017
United States (US) orthopedic procedures count by segments and forecast to 2030. GlobalData. February 17, 2023. Accessed April 29, 2026. https://www.globaldata.com/store/report/usa-orthopedic-procedures-analysis/
Jarman MP, Weaver MJ, Haider AH, Salim A, Harris MB. The national burden of orthopedic injury: cross-sectional estimates for trauma system planning and optimization. J Surg Res. 2020;249:197-204. doi:10.1016/j.jss.2019.12.023
Hegde V, Stambough JB, Levine BR, et al. Highlights of the 2022 American Joint Replacement Registry Annual Report. Arthroplast Today. 2023;21:101137. doi:10.1016/j.artd.2023.101137
Nakamura K, Ogata T. Locomotive syndrome: definition and management. Clin Rev Bone Miner Metab. 2016;14:56-67. doi:10.1007/s12018-016-9208-2
Glyn-Jones S, Palmer AJR, Agricola R, et al. Osteoarthritis. Lancet. 2015;386:376-387. doi:10.1016/S0140-6736(14)60802-3
Hughes S, Leary A, Zweizig S, Cain J. Surgery in elderly people: preoperative, operative and postoperative care to assist healing. Best Pract Res Clin Obstet Gynaecol. 2013;27:753-765. doi:10.1016/j.bpobgyn.2013.02.006
Regulski MJ. Cellular senescence: what, why, and how. Wounds. 2017;29:168-174.
Kremer M, Burkemper N. Aging skin and wound healing. Clin Geriatr Med. 2024;40:1-10. doi:10.1016/j.cger.2023.06.001
Fenske NA, Lober CW. Structural and functional changes of normal aging skin. J Am Acad Dermatol. 1986;15:571-585. doi:10.1016/S0190-9622(86)70208-9
Van Neste D, Tobin DJ. Hair cycle and hair pigmentation: dynamic interactions and changes associated with aging. Micron. 2004;35:193-200. doi:10.1016/j.micron.2003.11.006
Salzer MC, Lafzi A, Berenguer-Llergo A, et al. Identity noise and adipogenic traits characterize dermal fibroblast aging. Cell. 2018;175:1575-1590.e22. doi:10.1016/j.cell.2018.10.012
Jin K. A microcirculatory theory of aging. Aging Dis. 2019;10:676-683. doi:10.14336/AD.2019.0315
Marengoni A, Angleman S, Melis R, et al. Aging with multimorbidity: a systematic review of the literature. Ageing Res Rev. 2011;10:430-439. doi:10.1016/j.arr.2011.03.003
Waljee AK, Rogers MAM, Lin P, et al. Short term use of oral corticosteroids and related harms among adults in the United States: population based cohort study. BMJ. 2017;357:j1415. doi:10.1136/bmj.j1415
Given B, Given CW. Older adults and cancer treatment. Cancer. 2008;113:3505-3511. doi:10.1002/cncr.23939
Ferrucci L, Cooper R, Shardell M, Simonsick EM, Schrack JA, Kuh D. Age-related change in mobility: perspectives from life course epidemiology and geroscience. J Gerontol A Biol Sci Med Sci. 2016;71:1184-1194. doi:10.1093/gerona/glw043
Mayhew D, Mendonca V, Murthy BVS. A review of ASA physical status - historical perspectives and modern developments. Anaesthesia. 2019;74:373-379. doi:10.1111/anae.14569
Eagle KA, Berger PB, Calkins H, et al. ACC/AHA guideline update for perioperative cardiovascular evaluation for noncardiac surgery—executive summary a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee to Update the 1996 Guidelines on Perioperative Cardiovascular Evaluation for Noncardiac Surgery). Circulation. 2002;105:1257-1267. doi:10.1161/circ.105.10.1257
Bapoje SR, Whitaker JF, Schulz T, Chu ES, Albert RK. Preoperative evaluation of the patient with pulmonary disease. Chest. 2007;132:1637-1645. doi:10.1378/chest.07-0347
Choe H, Indelli PF, Ricciardi B, et al. What are the absolute contraindications for elective total knee or hip arthroplasty? J Arthroplasty. 2025;40(2 suppl 1):S45-S47. doi:10.1016/j.arth.2024.10.041
Finley CR, Chan DS, Garrison S, et al. What are the most common conditions in primary care? Systematic review. Can Fam Physician. 2018;64:832-840.
Vaid S, Bell T, Grim R, Ahuja V. Predicting risk of death in general surgery patients on the basis of preoperative variables using American College of Surgeons National Surgical Quality Improvement Program data. Perm J. 2012;16:10-17. doi:10.7812/TPP/12-019
Correia MTD, Waitzberg DL. The impact of malnutrition on morbidity, mortality, length of hospital stay and costs evaluated through a multivariate model analysis. Clin Nutr. 2003;22:235-239. doi:10.1016/S0261-5614(02)00215-7
Friedman J, Lussiez A, Sullivan J, Wang S, Englesbe M. Implications of sarcopenia in major surgery. Nutr Clin Pract. 2015;30:175-179. doi:10.1177/0884533615569888
Hirsch KR, Wolfe RR, Ferrando AA. Pre- and post-surgical nutrition for preservation of muscle mass, strength, and functionality following orthopedic surgery. Nutrients. 2021;13:1675. doi:10.3390/nu13051675
Curtis W, Choi T, Ahmad A, Shultz C. Perioperative nutritional considerations in orthopaedic surgery: a review of the literature. West J Orthop. 2023;12:1. https://digitalrepository.unm.edu/wjo/vol12/iss1/1
Wischmeyer PE, Carli F, Evans DC, et al. American Society for Enhanced Recovery and Perioperative Quality Initiative joint consensus statement on nutrition screening and therapy within a surgical enhanced recovery pathway. Anesth Analg. 2018;126:1883-1895. doi:10.1213/ANE.0000000000002743
Mun F, Ringenbach K, Baer B, et al. Factors influencing geriatric orthopaedic trauma mortality. Injury. 2022;53:919-924. doi:10.1016/j.injury.2022.01.005
Bonne S, Schuerer DJE. Trauma in the older adult: epidemiology and evolving geriatric trauma principles. Clin Geriatr Med. 2013;29:137-150. doi:10.1016/j.cger.2012.10.008
Montero-Odasso MM. Falls as a geriatric syndrome: mechanisms and risk identification. In: Duque G, Kiel DP, eds. Osteoporosis in Older Persons: Advances in Pathophysiology and Therapeutic Approaches. 2nd ed. Springer International Publishing; 2016:171-186. doi:10.1007/978-3-319-25976-5_10
Lach HW, Reed AT, Arfken CL, et al. Falls in the elderly: reliability of a classification system. J Am Geriatr Soc. 1991;39:197-202. doi:10.1111/j.1532-5415.1991.tb01626.x
Carpenter CR, DesPain B, Keeling TN, Shah M, Rothenberger M. The six-item screener and AD8 for the detection of cognitive impairment in geriatric emergency department patients. Ann Emerg Med. 2011;57:653-661. doi:10.1016/j.annemergmed.2010.06.560
Clegg A, Young J, Iliffe S, Rikkert MO, Rockwood K. Frailty in elderly people. Lancet. 2013;381:752-762. doi:10.1016/S0140-6736(12)62167-9
Patel JN, Klein DS, Sreekumar S, Liporace FA, Yoon RS. Outcomes in multidisciplinary team-based approach in geriatric hip fracture care: a systematic review. J Am Acad Orthop Surg. 2020;28:128-133. doi:10.5435/JAAOS-D-18-00425
Amador LF, Loera JA. Preventing postoperative falls in the older adult. J Am Coll Surg. 2007;204:447-453. doi:10.1016/j.jamcollsurg.2006.12.010
Tembo MC, Holloway-Kew KL, Mohebbi M, et al. The association between a fracture risk tool and frailty: Geelong Osteoporosis Study. BMC Geriatr. 2020;20:196. doi:10.1186/s12877-020-01595-8
Demiris¸ B, Basat S, Kurt F, Aksakal B, Basat O. Evaluation of the relationship between frailty and fracture risk using Fracture Risk Assessment Tool in patients 65 years and over. South Clin Istanb Eurasia. 2023;34:42-48. doi:10.14744/scie.2022.66564
Partridge JSL, Harari D, Dhesi JK. Frailty in the older surgical patient: a review. Age Ageing. 2012;41:142-147. doi:10.1093/ageing/afr182
Mamtora PH, Fortier MA, Barnett SR, Schmid LN, Kain ZN. Peri-operative management of frailty in the orthopedic patient. J Orthop. 2020;22:304-307. doi:10.1016/j.jor.2020.05.024
Leven DM, Lee NJ, Kim JS, et al. Frailty is predictive of adverse postoperative events in patients undergoing lumbar fusion. Global Spine J. 2017;7:529-535. doi:10.1177/2192568217700099
Pritchard JM, Kennedy CC, Karampatos S, et al. Measuring frailty in clinical practice: a comparison of physical frailty assessment methods in a geriatric out-patient clinic. BMC Geriatr. 2017;17:264. doi:10.1186/s12877-017-0623-0
Kumar A, Dhar M, Agarwal M, Mukherjee A, Saxena V. Predictors of frailty in the elderly population: a cross-sectional study at a tertiary care center. Cureus. 2022;14:e30557. doi:10.7759/cureus.30557
Scarano KA, Philp FH, Westrick ER, Altman GT, Altman DT. Evaluating postoperative complications and outcomes of orthopedic fracture repair in nonagenarian patients. Geriatr Orthop Surg Rehabil. 2018;9:2151459318758106. doi:10.1177/2151459318758106
Liang Z, Rong K, Gu W, et al. Surgical site infection following elective orthopaedic surgeries in geriatric patients: incidence and associated risk factors. Int Wound J. 2019;16:773-780. doi:10.1111/iwj.13096
Ren M, Liang W, Wu Z, Zhao H, Wang J. Risk factors of surgical site infection in geriatric orthopedic surgery: a retrospective multicenter cohort study. Geriatr Gerontol Int. 2019;19:213-217. doi:10.1111/ggi.13590
Kaye KS, Schmader KE, Sawyer R. Surgical site infection in the elderly population. Clin Infect Dis. 2004;39:1835-1841. doi:10.1086/425744
Bruce AJ, Ritchie CW, Blizard R, Lai R, Raven P. The incidence of delirium associated with orthopedic surgery: a meta-analytic review. Int Psychogeriatr. 2007;19:197-214. doi:10.1017/S104161020600425X
Williams-Russo P, Urquhart BL, Sharrock NE, Charlson ME. Post-operative delirium: predictors and prognosis in elderly orthopedic patients. J Am Geriatr Soc. 1992;40:759-767. doi:10.1111/j.1532-5415.1992.tb01846.x
Wang J, Li Z, Yu Y, Li B, Shao G, Wang Q. Risk factors contributing to postoperative delirium in geriatric patients postorthopedic surgery. Asia Pac Psychiatry. 2015;7:375-382. doi:10.1111/appy.12193
Geerts WH, Pineo GF, Heit JA, et al. Prevention of venous thromboembolism: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest. 2004;126:338S-400S. doi:10.1378/chest.126.3_suppl.338S
Kahn SR, Shivakumar S. What’s new in VTE risk and prevention in orthopedic surgery. Res Pract Thromb Haemost. 2020;4:366-376. doi:10.1002/rth2.12323
Uzel K, Azboy I·, Parvizi J. Venous thromboembolism in orthopedic surgery: global guidelines. Acta Orthop Traumatol Turc. 2023;57:192-203. doi:10.5152/j.aott.2023.23074
Peck M, Holthaus A, Kingsbury K, Salsberry MG, Duggirala V. Mobility in acute care for geriatric patients with orthopedic conditions: a review of recent literature. Curr Geri Rep. 2020;9:300-310. doi:10.1007/s13670-020-00347-1
Leme LEG, Sitta MC, Toledo M, Henriques SS. Orthopedic surgery among the elderly: clinical characteristics. Rev Bras Ortop. 2015;46:238-246. doi:10.1016/S2255-4971(15)30189-0
Malcolm TL, Knezevic NN, Zouki CC, Tharian AR. Pulmonary complications after hip and knee arthroplasty in the United States, 2004-2014. Anesth Analg. 2020;130:917-924. doi:10.1213/ANE.0000000000004265
Kamal T, Conway RM, Littlejohn I, Ricketts D. The role of a multidisciplinary pre-assessment clinic in reducing mortality after complex orthopaedic surgery. Ann R Coll Surg Engl. 2011;93:149-151. doi:10.1308/003588411X561026
Davis MJ, Luu BC, Raj S, Abu-Ghname A, Buchanan EP. Multidisciplinary care in surgery: Are team-based interventions cost-effective? Surgeon. 2021;19:49-60. doi:10.1016/j.surge.2020.02.005
Frassanito L, Vergari A, Nestorini R, et al. Enhanced recovery after surgery (ERAS) in hip and knee replacement surgery: description of a multidisciplinary program to improve management of the patients undergoing major orthopedic surgery. Musculoskelet Surg. 2020;104:87-92. doi:10.1007/s12306-019-00603-4
Reddy RS, Alahmari KA, Alshahrani MS, et al. Exploring the impact of physiotherapy on health outcomes in older adults with chronic diseases: a cross-sectional analysis. Front Public Health. 2024;12:1415882. doi:10.3389/fpubh.2024.1415882
Watts NB. The Fracture Risk Assessment Tool (FRAX®): applications in clinical practice. J Womens Health (Larchmt). 2011;20:525-531. doi:10.1089/jwh.2010.2294
Gleason LJ, Benton EA, Alvarez-Nebreda ML, Weaver MJ, Harris MB, Javedan H. FRAIL questionnaire screening tool and short-term outcomes in geriatric fracture patients. J Am Med Dir Assoc. 2017;18:1082-1086. doi:10.1016/j.jamda.2017.07.005
Kojima G. Frailty defined by FRAIL scale as a predictor of mortality: a systematic review and meta-analysis. J Am Med Dir Assoc. 2018;19:480-483. doi:10.1016/j.jamda.2018.04.006
Culley DJ, Flaherty D, Fahey MC, et al. Poor performance on a preoperative cognitive screening test predicts postoperative complications in older orthopedic surgical patients. Anesthesiology. 2017;127:765-774. doi:10.1097/ALN.0000000000001859
Kong C, Zhang Y, Wang C, et al. Comprehensive geriatric assessment for older orthopedic patients and analysis of risk factors for postoperative complications. BMC Geriatr. 2022;22:644. doi:10.1186/s12877-022-03328-5
Williams DGA, Wischmeyer PE. Perioperative nutrition care of orthopedic surgery patient. Tech Orthop. 2020;35:15-18. doi:10.1097/BTO.0000000000000412
Koren-Hakim T, Weiss A, Hershkovitz A, et al. Comparing the adequacy of the MNA-SF, NRS-2002 and MUST nutritional tools in assessing malnutrition in hip fracture operated elderly patients. Clin Nutr. 2016;35:1053-1058. doi:10.1016/j.clnu.2015.07.014
Perioperative Considerations for Orthopedic Surgery in a Geriatric Population
Perioperative Considerations for Orthopedic Surgery in a Geriatric Population
Involving Concerned Significant Others in Firearm Suicide Prevention: Development of the Family FireArms Secure Storage Training Intervention
Involving Concerned Significant Others in Firearm Suicide Prevention: Development of the Family FireArms Secure Storage Training Intervention
Veterans are at higher risk for suicide compared with civilian populations.1 Firearms are the most frequent cause of death in veteran deaths by suicide, likely because about 51% of veterans own ≥ 1 firearms and firearms are the most lethal and readily available mechanism.1-3 Unsecure firearm storage practices (eg, storing firearms unlocked, in an unsecure location, or loaded with ammunition) are associated with increased suicide risk.4 Conversely, secure firearm storage (ie, storing firearms locked and unloaded) is associated with lower suicide risk.5
A 2019 study of veterans who own firearms found that only 22.2% store all their firearms unloaded and locked, while 32.7% store ≥ 1 firearm unlocked and loaded, and 45.2% store firearms both unlocked and loaded or locked and unloaded. Only 6.3% of veterans strongly agreed that having a firearm at home increased suicide risk among household members; however, 77.2% indicated they would ensure a household member could not access firearms if they were concerned about their suicidal ideation.6
Another study found that 9.2% of veterans receive lethal means safety counseling from their US Department of Veterans Affairs (VA)-affiliated or non-VA health care professional.7 These data highlight a need to educate veterans about the increased risk for suicide associated with storing an unsecured firearm in the household and to connect this understanding to their values of service and protection of others, while simultaneously preparing them and their family members for a potential mental health crisis.
Consistent with the government’s public health approach to suicide prevention, prevention efforts should also enlist the participation of individuals outside health care.8 For example, prior research has found that family members are considered highly credible, and engaging them could expand the reach of lethal means safety conversations. A qualitative analysis of 29 veterans found that 17 (57%) said they preferred having a concerned significant other (CSO) (eg, spouse, adult friend, or relative) involved in their suicide prevention care, while 21 (72%) said they would prefer having a CSO assisting in the secure storage of firearms.9,10 Some veterans may be more amenable to a conversation about firearm access and suicide risk concerns initiated by a CSO rather than by a clinician, indicating the potential benefits of educating and involving CSOs in suicide prevention.11 Involving CSOs in secure firearm storage planning may also strengthen the veteran’s sense of social support, a key protective factor against suicidal ideation.12
CSO involvement in secure firearm storage can provide the following benefits: (1) helping the veteran create a secure storage plan, including developing approaches to secure storage; (2) understanding warning signs of suicide; (3) helping the veteran limit access to firearms during a suicidal crisis; (4) helping the veteran remember the secure storage plan; (5) helping the veteran connect with mental health services; and (6) enhancing social support. In most instances, CSOs are physically close to the veteran (eg, live in the same household) and have a greater practical ability to support and affect change with respect to changes in firearm storage practices.
This article describes the development of an intervention that incorporates CSO involvement in firearms safety efforts for veterans with guidance from VA mental health care practitioners (HCPs). The goal is to provide HCPs and other key stakeholders with a detailed description of the intervention and to suggest potential strategies for how to involve CSOs in suicide prevention.
This article follows the Guideline for Reporting Evidence-based Practice Educational interventions and Teaching checklist, which was developed to facilitate standardized reporting and replication for education interventions.13 Applicable portions of the checklist are outlined, with others (ie, incentives, planned/unplanned changes, attendance, and other outcomes) to be addressed in future research.
FFAST INTERVENTION
Training (FFAST) intervention promotes voluntary secure firearm storage, engages CSOs in veteran mental health care, and provides psychoeducation and skills to support crisis management. The intervention was developed for all veterans who do not securely store firearms.
Theory
The intervention incorporates motivational interviewing techniques, as ambivalence about changing firearm storage behaviors is common, particularly when veterans own firearms for safety or protection.6,14 Motivational interviewing is a collaborative approach that addresses a client’s ambivalence to change by eliciting and exploring the client’s own arguments related to change.14 An important aspect of developing this intervention was to ensure it would be culturally relevant to veteran firearm owners and their CSOs.15 Further, involvement of the CSO is intentional and meant to boost social support, a known buffering factor against suicide risk.12
Objectives
This intervention’s primary objective was for veteran participants to identify secure firearm storage practices and develop a plan for implementing them, including when a veteran or other household member experiences a mental health crisis. For CSOs, the primary objective is to learn how to help the veteran connect with mental health resources if needed and support secure firearm storage as necessary. The overall goal is to learn how to identify warning signs for suicide and how to respond to a mental health crisis through a collaborative process, including securing firearms in a crisis situation.
Materials, Educational Strategies, and Instructors
Training for delivering the intervention was provided via direct consultation with the developer of the intervention and manual. The manual contains pertinent background information to provide context for the intervention’s significance and rationale, including the role of firearms in suicides and current lethal means safety initiatives. It also describes the purpose and objective of each intervention component in detail in addition to providing a script for interventionists to follow to complete each objective.
Training materials for veterans and CSOs include a single Firearms Secure Storage Planning worksheet completed during the intervention, with which the interventionist guides participants through the creation of a secure firearm storage plan (Table). Educational strategies include psychoeducation and Socratic questioning (eg, questioning focused on guiding participants toward the intervention goals) delivered verbally by the interventionist.
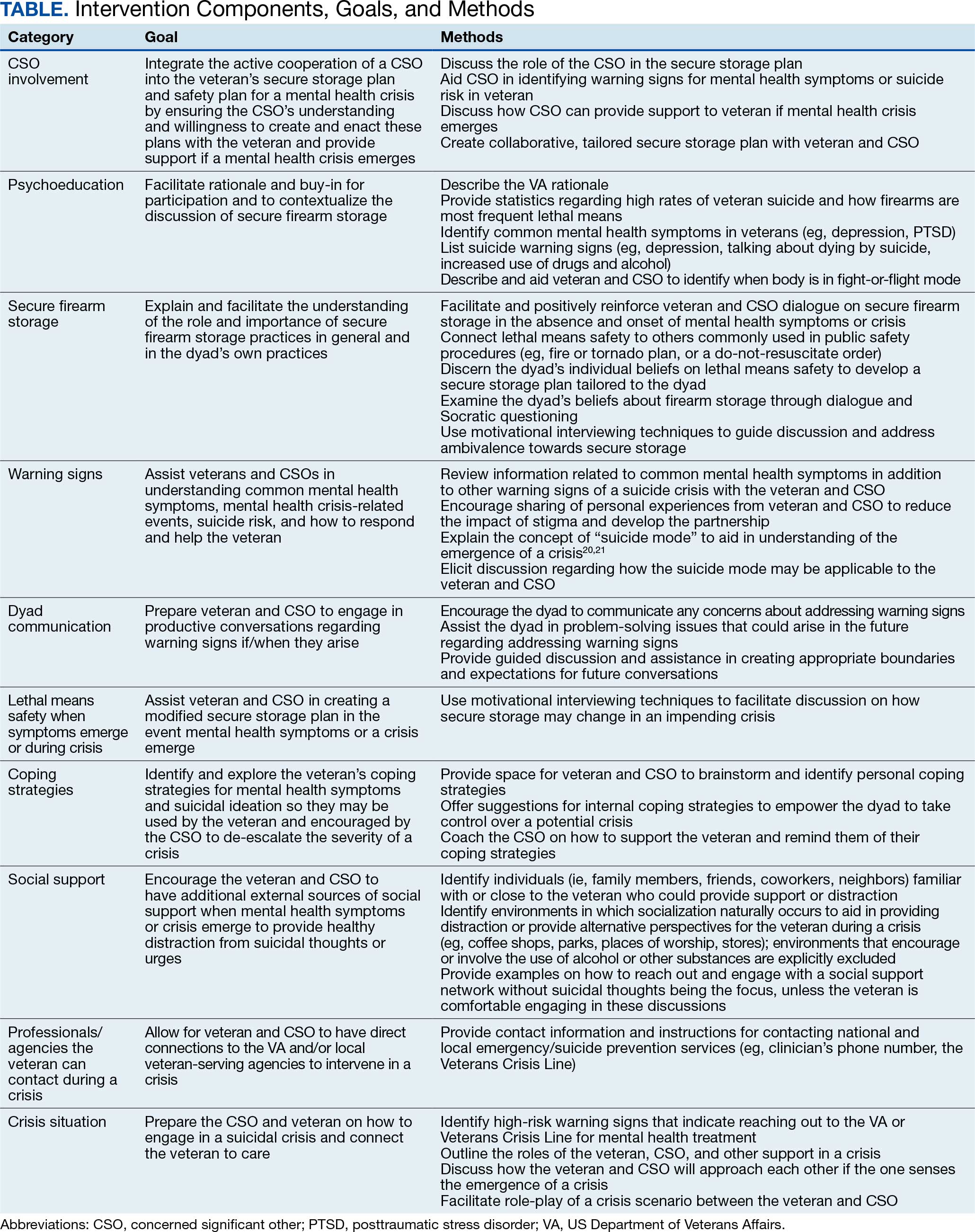
The intervention is delivered in person or virtually during a single 90-minute session with a veteran and CSO. Veterans and CSOs work with the interventionist to complete collaborative activities during the session and have self-directive learning activities or homework.
The intervention has 4 primary components: (1) CSO involvement; (2) psychoeducation; (3) secure firearm storage; and (4) how to respond to a mental health crisis. Each CSO should have an established relationship with the veteran, be willing and able to be present during the intervention, and remain an encouraging support person for the veteran. The interventionist emphasizes that it is part of the VA mission for staff to care about the veteran, and that initiating such contact with a CSO is meant to prioritize veteran safety and the safety of their family. Psychoeducation on mental health symptoms, suicide warning signs, veteran suicide rates and lethal means, and the benefits of secure firearm storage, is incorporated in the intervention.
The secure firearm storage component consists of 7 subcomponents: (1) general lethal means secure storage; (2) warning signs; (3) dyad communication; (4) lethal means safety when symptoms emerge; (5) coping strategies; (6) social support; and (7) emergency contacts. A lethal means safety worksheet rooted in the Stanley and Brown suicide safety plan model and implemented in VA health care settings is used to facilitate discussions of secure storage (Appendix).16
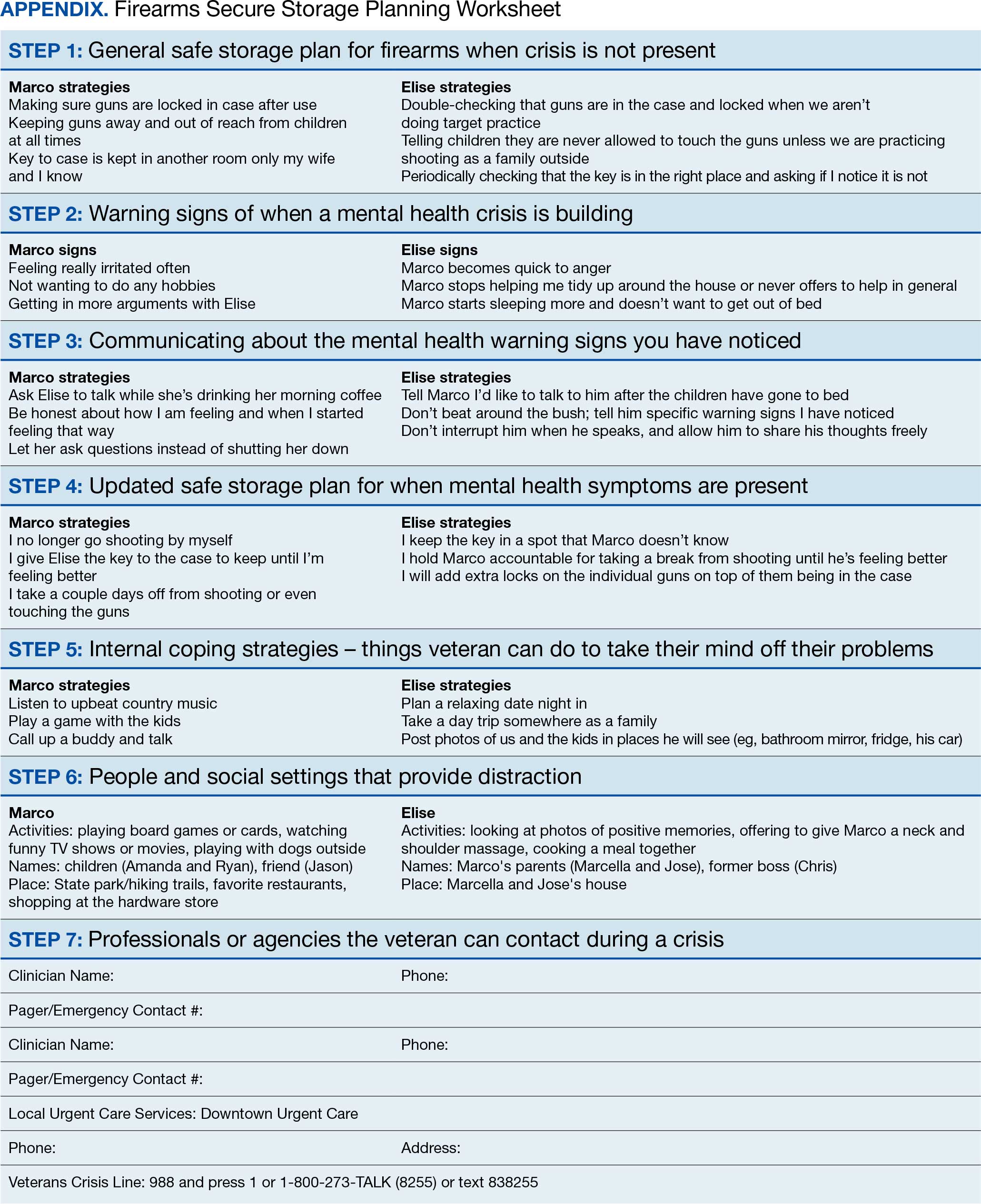
CSOs typically have little or no suicidal crisis response training, yet they likely have more interaction with the veteran on a daily basis than HCPs, putting them in a vital position to identify a crisis early and connect the veteran with the proper care. The crisis component prepares the CSO and veteran to navigate a crisis scenario so they can practice their newly developed safety plan and increase their comfort in discussing mental health and suicidal crisis.
FICTIONAL CASE STUDY
Cole, aged 59 years, is a Persian Gulf War veteran and retired police officer. His medical history includes hypothyroidism, hypertension, type 2 diabetes mellitus, chronic posttraumatic stress disorder, major depressive disorder, and insomnia.
Cole's wife of > 30 years, Sheila, joined him for the FFAST intervention. They report having 4 firearms in the home, 3 of which are loaded but stored in a lockbox and 1 that Cole reports is kept on his person for protection. Cole reports passive suicidal ideation, but no plans or intent. When discussing warning signs that a mental health crisis is building, Cole describes feeling anxious, having a change in his speech patterns, and isolating himself. Sheila agrees, but also mentions that Cole is easily angered and becomes nonverbal. Cole and Sheila express difficulty communicating and appear to have a breakthrough moment when Cole says he does not like when Sheila repeats herself, as he feels like she is “poking” at him. Sheila shares concerns for his safety and that she only repeats herself because he refuses to talk.
Cole agrees to verbalize that he is safe but needs time to process his thoughts. Sheila agrees to give him space with a plan to revisit the conversation within an agreed upon timeline. When discussing an updated secure storage plan for their firearms when a mental health crisis is building, Cole commits to allowing Sheila to store the firearm currently on his person in their gun safe, with the ammunition stored separately, and to giving her the gun safe key. They agree to implement this practice until the mental health crisis has passed.
To mitigate a potential crisis, the interventionist discusses possible internal coping strategies for Cole, including writing, reading, walking the dog, listening to music, and baking. People and social settings that could provide distraction involve going to the gym, talking to his friend Carl or his daughter Kelly, and attending the men’s ministry at church. The intervention concludes by discussing professionals or agencies that Cole and Sheila could contact during a crisis. After the intervention, Cole and Sheila are asked to rate their likelihood of using the plan they established during the conversation on a scale of 0 to 10, with 0 being highly unlikely and 10 being extremely likely. Cole responds with 9 and Sheila responds with 10.
DISCUSSION
Lethal means safety remains a critical component of veteran suicide prevention. However, lethal means safety discussions are often implemented after suicide risk has been identified, which may be too late. Thus, having these conversations early and before a crisis may be imperative. Veterans have expressed a desire to have CSOs involved in their suicide prevention treatment, and CSOs can play a key role in recognizing risk factors during everyday life. The FFAST intervention addresses many of these gaps.
Having discussions in advance of a crisis allows veterans to consider an effective secure firearm storage plan outside of the context of a crisis. Including a CSO galvanizes another person to understand a veteran’s needs and assist with secure firearm storage, identify warning signs, and support them during a crisis. These discussions occur in a context where there is less pressure than during a crisis. Features that were more appealing to veterans and their CSOs were also incorporated, such as having the dyad build a plan that is conceptually similar to other public safety initiatives (eg, a fire safety plan, tornado plan, or hurricane plan). Previous research demonstrates that veterans appreciated the nonjudgmental approach and some preferred that clinicians approach the discussion of secure firearm storage within the context of general home and family safety.17 Additionally, this intervention can build on veterans’ prior military training in preparedness.
Other potential benefits associated with the FFAST intervention include creating an opportunity to strengthen communication between the veteran and CSO. While FFAST is intended to be used with all types of CSOs, this work is consistent with preliminary data from a couples-based suicide prevention study that indicated veterans and their partners reported increases in relationship functioning and marginal decreases in suicidal ideation.18 It is possible that communication strategies gained from the current intervention could improve veterans’ relationships with their CSOs, which are associated with a greater sense of social support and reduced suicide risk.12
The intervention is a brief, single session that may be appealing to veterans and CSOs with full schedules. Evidence suggests that even brief, single-session interventions have a significant impact on beliefs about secure firearm storage, knowledge of lethal means safety, and confidence in having secure firearm storage conversations.19 However, clinicians should be cautious when extrapolating from the findings of the current case example, which was a one-time intervention with no follow-up.
Future Directions
Pilot testing of the proposed intervention is underway, and future research will include feedback from veterans and CSOs, as well as feasibility and acceptability data collected during the pilot process. The pilot study uses a successive cohort design with an initial 2 sets of 5 veteran and CSO dyads, and subsequent funding has expanded the pilot study to include an additional 30 dyads. Qualitative interviews will be conducted separately with each veteran and CSO, and additional constructs such as feasibility, acceptability, barriers and facilitators to implementation, and changes in secure storage will be examined. This future research may provide a deeper understanding of the broader acceptability, feasibility, and satisfaction associated with a suicide prevention intervention focused on securing firearms and involving veterans and their CSOs. These data could be used to inform future implementation trials and inform the development of an implementation strategy. In the interim, the nature of the manual is summarized in the context of the urgency of suicide prevention in this at-risk population.
Conclusions
FFAST is a novel approach to veteran firearm suicide prevention. By involving CSOs and emphasizing mental health crisis preparedness between them and veterans, the dyad can work in association with HCPs to establish and exercise secure firearm storage practices as part of an at-home safety plan. Implementation of FFAST may be beneficial for all veterans, not only those who have been identified as being at high suicide risk.
- US Dept of Veterans Affairs Office of Suicide Prevention. 2024 national veteran suicide prevention annual report. December 2024. Accessed February 5, 2026. https://www.mentalhealth.va.gov/docs/data-sheets/2024/2024-Annual-Report-Part-2-of-2_508.pdf
- Fischer IC, Aunon FM, Nichter B, et al. Firearm ownership among a nationally representative sample of U.S. veterans. Am J Prev Med. 2023;65:1129-1133. doi:10.1016/j.amepre.2023.06.013
- Conner A, Azrael D, Miller M. Suicide case-fatality rates in the United States, 2007-2014: a nationwide population-based study. Ann Intern Med. 2019;171(12):885-895. doi:10.7326/M19-1324
- Dempsey CL, Benedek DM, Zuromski KL, et al. Association of firearm ownership, use, accessibility, and storage practices with suicide risk among US army soldiers. JAMA Netw Open. 2019;2:e195383. doi:10.1001/jamanetworkopen.2019.5383
- Butterworth SE, Daruwala SE, Anestis MD. Firearm storage and shooting experience: factors relevant to the practical capability for suicide. J Psychiatr Res. 2018;102:52-56. doi:10.1016/j.jpsychires.2018.03.010
- Simonetti JA, Azrael D, Miller M. Firearm storage practices and risk perceptions among a nationally representative sample of U.S. veterans with and without self-harm risk factors. Suicide Life Threat Behav. 2019;49:653-664. doi:10.1111/sltb.12463
- Simonetti JA, Azrael D, Zhang W, Miller M. Receipt of clinician-delivered firearm safety counseling among U.S. veterans: results from a 2019 national survey. Suicide Life Threat Behav. 2022;52:1121-1125. doi:10.1111/sltb.12906
- US Office of the Surgeon General. The surgeon general’s call to action to implement the national strategy for suicide prevention. January 2021. Accessed February 5, 2026. https://www.hhs.gov/sites/default/files/sprc-call-to-action.pdf
- DeBeer BB, Matthieu MM, Kittel JA, et al. Quality Improvement Evaluation of the Feasibility and Acceptability of Adding a Concerned Significant Other to Safety Planning for Suicide Prevention With Veterans. J Ment Health Couns. 2019;41:4-20. doi:10.17744/mehc.41.1.02
- DeBeer BB, Matthieu MM, Degutis LC, et al. Firearms lethal means safety among veterans: attitudes toward involving a concerned significant other. J Mil Veteran Fam Health. 2025;11:23-31.
- Monteith LL, Holliday R, Dorsey Holliman BA, et al. Understanding female veterans’ experiences and perspectives of firearms. J Clin Psychol. 2020;76:1736-1753. doi:10.1002/jclp.22952
- DeBeer BB, Kimbrel NA, Meyer EC, et al. Combined PTSD and depressive symptoms interact with post-deployment social support to predict suicidal ideation in Operation Enduring Freedom and Operation Iraqi Freedom veterans. Psychiatry Res. 2014;216:357-362. doi:10.1016/j.psychres.2014.02.010
- Phillips AC, Lewis LK, McEvoy MP, et al. Development and validation of the guideline for reporting evidence-based practice educational interventions and teaching (GREET). BMC Med Educ. 2016;16:237. doi:10.1186/s12909-016-0759-1
- Miller WR, Rollnick S. Motivational Interviewing: Helping People Change. 3rd ed. Guilford Press; 2013.
- Khazanov GK, Keddem S, Hoskins K, et al. Stakeholder perceptions of lethal means safety counseling: a qualitative systematic review. Front Psychiatry. 2022;13:993415. doi:10.3389/fpsyt.2022.993415
- Stanley B, Brown GK, Karlin B, et al. US Dept of Veterans Affairs. Safety plan treatment manual to reduce suicide risk: veteran version. August 20, 2008. Accessed February 5, 2026. https://www.mentalhealth.va.gov/mentalhealth/docs/va_safety_planning_manual.doc
- Dobscha SK, Clark KD, Newell S, et al. Strategies for discussing firearms storage safety in primary care: veteran perspectives. J Gen Intern Med. 2021;36:1492-1502. doi:10.1007/s11606-020-06412-x
- Khalifian CE, Leifker FR, Knopp K, et al. Utilizing the couple relationship to prevent suicide: a preliminary examination of treatment for relationships and safety together. J Clin Psych. 2022;78:747-757. doi:10.1002/jclp.23251
- Walsh A, Friedman K, Morrissey BH, et al. Project Safe Guard: evaluating a lethal means safety intervention to reduce firearm suicide in the National Guard. Mil Med. 2024;189:510-516. doi:10.1093/milmed/usae172
- Beck AT. Beyond belief: a theory of modes, personality, and psychopathology. In: Salkovkis PM, ed. Frontiers of Cognitive Therapy. Guilford Press;1996:1-25.
- Rudd MD. The suicidal mode: a cognitive-behavioral model of suicidality. Suicide Life Threat Behav. 2000;30(1):18-33.
Veterans are at higher risk for suicide compared with civilian populations.1 Firearms are the most frequent cause of death in veteran deaths by suicide, likely because about 51% of veterans own ≥ 1 firearms and firearms are the most lethal and readily available mechanism.1-3 Unsecure firearm storage practices (eg, storing firearms unlocked, in an unsecure location, or loaded with ammunition) are associated with increased suicide risk.4 Conversely, secure firearm storage (ie, storing firearms locked and unloaded) is associated with lower suicide risk.5
A 2019 study of veterans who own firearms found that only 22.2% store all their firearms unloaded and locked, while 32.7% store ≥ 1 firearm unlocked and loaded, and 45.2% store firearms both unlocked and loaded or locked and unloaded. Only 6.3% of veterans strongly agreed that having a firearm at home increased suicide risk among household members; however, 77.2% indicated they would ensure a household member could not access firearms if they were concerned about their suicidal ideation.6
Another study found that 9.2% of veterans receive lethal means safety counseling from their US Department of Veterans Affairs (VA)-affiliated or non-VA health care professional.7 These data highlight a need to educate veterans about the increased risk for suicide associated with storing an unsecured firearm in the household and to connect this understanding to their values of service and protection of others, while simultaneously preparing them and their family members for a potential mental health crisis.
Consistent with the government’s public health approach to suicide prevention, prevention efforts should also enlist the participation of individuals outside health care.8 For example, prior research has found that family members are considered highly credible, and engaging them could expand the reach of lethal means safety conversations. A qualitative analysis of 29 veterans found that 17 (57%) said they preferred having a concerned significant other (CSO) (eg, spouse, adult friend, or relative) involved in their suicide prevention care, while 21 (72%) said they would prefer having a CSO assisting in the secure storage of firearms.9,10 Some veterans may be more amenable to a conversation about firearm access and suicide risk concerns initiated by a CSO rather than by a clinician, indicating the potential benefits of educating and involving CSOs in suicide prevention.11 Involving CSOs in secure firearm storage planning may also strengthen the veteran’s sense of social support, a key protective factor against suicidal ideation.12
CSO involvement in secure firearm storage can provide the following benefits: (1) helping the veteran create a secure storage plan, including developing approaches to secure storage; (2) understanding warning signs of suicide; (3) helping the veteran limit access to firearms during a suicidal crisis; (4) helping the veteran remember the secure storage plan; (5) helping the veteran connect with mental health services; and (6) enhancing social support. In most instances, CSOs are physically close to the veteran (eg, live in the same household) and have a greater practical ability to support and affect change with respect to changes in firearm storage practices.
This article describes the development of an intervention that incorporates CSO involvement in firearms safety efforts for veterans with guidance from VA mental health care practitioners (HCPs). The goal is to provide HCPs and other key stakeholders with a detailed description of the intervention and to suggest potential strategies for how to involve CSOs in suicide prevention.
This article follows the Guideline for Reporting Evidence-based Practice Educational interventions and Teaching checklist, which was developed to facilitate standardized reporting and replication for education interventions.13 Applicable portions of the checklist are outlined, with others (ie, incentives, planned/unplanned changes, attendance, and other outcomes) to be addressed in future research.
FFAST INTERVENTION
Training (FFAST) intervention promotes voluntary secure firearm storage, engages CSOs in veteran mental health care, and provides psychoeducation and skills to support crisis management. The intervention was developed for all veterans who do not securely store firearms.
Theory
The intervention incorporates motivational interviewing techniques, as ambivalence about changing firearm storage behaviors is common, particularly when veterans own firearms for safety or protection.6,14 Motivational interviewing is a collaborative approach that addresses a client’s ambivalence to change by eliciting and exploring the client’s own arguments related to change.14 An important aspect of developing this intervention was to ensure it would be culturally relevant to veteran firearm owners and their CSOs.15 Further, involvement of the CSO is intentional and meant to boost social support, a known buffering factor against suicide risk.12
Objectives
This intervention’s primary objective was for veteran participants to identify secure firearm storage practices and develop a plan for implementing them, including when a veteran or other household member experiences a mental health crisis. For CSOs, the primary objective is to learn how to help the veteran connect with mental health resources if needed and support secure firearm storage as necessary. The overall goal is to learn how to identify warning signs for suicide and how to respond to a mental health crisis through a collaborative process, including securing firearms in a crisis situation.
Materials, Educational Strategies, and Instructors
Training for delivering the intervention was provided via direct consultation with the developer of the intervention and manual. The manual contains pertinent background information to provide context for the intervention’s significance and rationale, including the role of firearms in suicides and current lethal means safety initiatives. It also describes the purpose and objective of each intervention component in detail in addition to providing a script for interventionists to follow to complete each objective.
Training materials for veterans and CSOs include a single Firearms Secure Storage Planning worksheet completed during the intervention, with which the interventionist guides participants through the creation of a secure firearm storage plan (Table). Educational strategies include psychoeducation and Socratic questioning (eg, questioning focused on guiding participants toward the intervention goals) delivered verbally by the interventionist.

The intervention is delivered in person or virtually during a single 90-minute session with a veteran and CSO. Veterans and CSOs work with the interventionist to complete collaborative activities during the session and have self-directive learning activities or homework.
The intervention has 4 primary components: (1) CSO involvement; (2) psychoeducation; (3) secure firearm storage; and (4) how to respond to a mental health crisis. Each CSO should have an established relationship with the veteran, be willing and able to be present during the intervention, and remain an encouraging support person for the veteran. The interventionist emphasizes that it is part of the VA mission for staff to care about the veteran, and that initiating such contact with a CSO is meant to prioritize veteran safety and the safety of their family. Psychoeducation on mental health symptoms, suicide warning signs, veteran suicide rates and lethal means, and the benefits of secure firearm storage, is incorporated in the intervention.
The secure firearm storage component consists of 7 subcomponents: (1) general lethal means secure storage; (2) warning signs; (3) dyad communication; (4) lethal means safety when symptoms emerge; (5) coping strategies; (6) social support; and (7) emergency contacts. A lethal means safety worksheet rooted in the Stanley and Brown suicide safety plan model and implemented in VA health care settings is used to facilitate discussions of secure storage (Appendix).16

CSOs typically have little or no suicidal crisis response training, yet they likely have more interaction with the veteran on a daily basis than HCPs, putting them in a vital position to identify a crisis early and connect the veteran with the proper care. The crisis component prepares the CSO and veteran to navigate a crisis scenario so they can practice their newly developed safety plan and increase their comfort in discussing mental health and suicidal crisis.
FICTIONAL CASE STUDY
Cole, aged 59 years, is a Persian Gulf War veteran and retired police officer. His medical history includes hypothyroidism, hypertension, type 2 diabetes mellitus, chronic posttraumatic stress disorder, major depressive disorder, and insomnia.
Cole's wife of > 30 years, Sheila, joined him for the FFAST intervention. They report having 4 firearms in the home, 3 of which are loaded but stored in a lockbox and 1 that Cole reports is kept on his person for protection. Cole reports passive suicidal ideation, but no plans or intent. When discussing warning signs that a mental health crisis is building, Cole describes feeling anxious, having a change in his speech patterns, and isolating himself. Sheila agrees, but also mentions that Cole is easily angered and becomes nonverbal. Cole and Sheila express difficulty communicating and appear to have a breakthrough moment when Cole says he does not like when Sheila repeats herself, as he feels like she is “poking” at him. Sheila shares concerns for his safety and that she only repeats herself because he refuses to talk.
Cole agrees to verbalize that he is safe but needs time to process his thoughts. Sheila agrees to give him space with a plan to revisit the conversation within an agreed upon timeline. When discussing an updated secure storage plan for their firearms when a mental health crisis is building, Cole commits to allowing Sheila to store the firearm currently on his person in their gun safe, with the ammunition stored separately, and to giving her the gun safe key. They agree to implement this practice until the mental health crisis has passed.
To mitigate a potential crisis, the interventionist discusses possible internal coping strategies for Cole, including writing, reading, walking the dog, listening to music, and baking. People and social settings that could provide distraction involve going to the gym, talking to his friend Carl or his daughter Kelly, and attending the men’s ministry at church. The intervention concludes by discussing professionals or agencies that Cole and Sheila could contact during a crisis. After the intervention, Cole and Sheila are asked to rate their likelihood of using the plan they established during the conversation on a scale of 0 to 10, with 0 being highly unlikely and 10 being extremely likely. Cole responds with 9 and Sheila responds with 10.
DISCUSSION
Lethal means safety remains a critical component of veteran suicide prevention. However, lethal means safety discussions are often implemented after suicide risk has been identified, which may be too late. Thus, having these conversations early and before a crisis may be imperative. Veterans have expressed a desire to have CSOs involved in their suicide prevention treatment, and CSOs can play a key role in recognizing risk factors during everyday life. The FFAST intervention addresses many of these gaps.
Having discussions in advance of a crisis allows veterans to consider an effective secure firearm storage plan outside of the context of a crisis. Including a CSO galvanizes another person to understand a veteran’s needs and assist with secure firearm storage, identify warning signs, and support them during a crisis. These discussions occur in a context where there is less pressure than during a crisis. Features that were more appealing to veterans and their CSOs were also incorporated, such as having the dyad build a plan that is conceptually similar to other public safety initiatives (eg, a fire safety plan, tornado plan, or hurricane plan). Previous research demonstrates that veterans appreciated the nonjudgmental approach and some preferred that clinicians approach the discussion of secure firearm storage within the context of general home and family safety.17 Additionally, this intervention can build on veterans’ prior military training in preparedness.
Other potential benefits associated with the FFAST intervention include creating an opportunity to strengthen communication between the veteran and CSO. While FFAST is intended to be used with all types of CSOs, this work is consistent with preliminary data from a couples-based suicide prevention study that indicated veterans and their partners reported increases in relationship functioning and marginal decreases in suicidal ideation.18 It is possible that communication strategies gained from the current intervention could improve veterans’ relationships with their CSOs, which are associated with a greater sense of social support and reduced suicide risk.12
The intervention is a brief, single session that may be appealing to veterans and CSOs with full schedules. Evidence suggests that even brief, single-session interventions have a significant impact on beliefs about secure firearm storage, knowledge of lethal means safety, and confidence in having secure firearm storage conversations.19 However, clinicians should be cautious when extrapolating from the findings of the current case example, which was a one-time intervention with no follow-up.
Future Directions
Pilot testing of the proposed intervention is underway, and future research will include feedback from veterans and CSOs, as well as feasibility and acceptability data collected during the pilot process. The pilot study uses a successive cohort design with an initial 2 sets of 5 veteran and CSO dyads, and subsequent funding has expanded the pilot study to include an additional 30 dyads. Qualitative interviews will be conducted separately with each veteran and CSO, and additional constructs such as feasibility, acceptability, barriers and facilitators to implementation, and changes in secure storage will be examined. This future research may provide a deeper understanding of the broader acceptability, feasibility, and satisfaction associated with a suicide prevention intervention focused on securing firearms and involving veterans and their CSOs. These data could be used to inform future implementation trials and inform the development of an implementation strategy. In the interim, the nature of the manual is summarized in the context of the urgency of suicide prevention in this at-risk population.
Conclusions
FFAST is a novel approach to veteran firearm suicide prevention. By involving CSOs and emphasizing mental health crisis preparedness between them and veterans, the dyad can work in association with HCPs to establish and exercise secure firearm storage practices as part of an at-home safety plan. Implementation of FFAST may be beneficial for all veterans, not only those who have been identified as being at high suicide risk.
Veterans are at higher risk for suicide compared with civilian populations.1 Firearms are the most frequent cause of death in veteran deaths by suicide, likely because about 51% of veterans own ≥ 1 firearms and firearms are the most lethal and readily available mechanism.1-3 Unsecure firearm storage practices (eg, storing firearms unlocked, in an unsecure location, or loaded with ammunition) are associated with increased suicide risk.4 Conversely, secure firearm storage (ie, storing firearms locked and unloaded) is associated with lower suicide risk.5
A 2019 study of veterans who own firearms found that only 22.2% store all their firearms unloaded and locked, while 32.7% store ≥ 1 firearm unlocked and loaded, and 45.2% store firearms both unlocked and loaded or locked and unloaded. Only 6.3% of veterans strongly agreed that having a firearm at home increased suicide risk among household members; however, 77.2% indicated they would ensure a household member could not access firearms if they were concerned about their suicidal ideation.6
Another study found that 9.2% of veterans receive lethal means safety counseling from their US Department of Veterans Affairs (VA)-affiliated or non-VA health care professional.7 These data highlight a need to educate veterans about the increased risk for suicide associated with storing an unsecured firearm in the household and to connect this understanding to their values of service and protection of others, while simultaneously preparing them and their family members for a potential mental health crisis.
Consistent with the government’s public health approach to suicide prevention, prevention efforts should also enlist the participation of individuals outside health care.8 For example, prior research has found that family members are considered highly credible, and engaging them could expand the reach of lethal means safety conversations. A qualitative analysis of 29 veterans found that 17 (57%) said they preferred having a concerned significant other (CSO) (eg, spouse, adult friend, or relative) involved in their suicide prevention care, while 21 (72%) said they would prefer having a CSO assisting in the secure storage of firearms.9,10 Some veterans may be more amenable to a conversation about firearm access and suicide risk concerns initiated by a CSO rather than by a clinician, indicating the potential benefits of educating and involving CSOs in suicide prevention.11 Involving CSOs in secure firearm storage planning may also strengthen the veteran’s sense of social support, a key protective factor against suicidal ideation.12
CSO involvement in secure firearm storage can provide the following benefits: (1) helping the veteran create a secure storage plan, including developing approaches to secure storage; (2) understanding warning signs of suicide; (3) helping the veteran limit access to firearms during a suicidal crisis; (4) helping the veteran remember the secure storage plan; (5) helping the veteran connect with mental health services; and (6) enhancing social support. In most instances, CSOs are physically close to the veteran (eg, live in the same household) and have a greater practical ability to support and affect change with respect to changes in firearm storage practices.
This article describes the development of an intervention that incorporates CSO involvement in firearms safety efforts for veterans with guidance from VA mental health care practitioners (HCPs). The goal is to provide HCPs and other key stakeholders with a detailed description of the intervention and to suggest potential strategies for how to involve CSOs in suicide prevention.
This article follows the Guideline for Reporting Evidence-based Practice Educational interventions and Teaching checklist, which was developed to facilitate standardized reporting and replication for education interventions.13 Applicable portions of the checklist are outlined, with others (ie, incentives, planned/unplanned changes, attendance, and other outcomes) to be addressed in future research.
FFAST INTERVENTION
Training (FFAST) intervention promotes voluntary secure firearm storage, engages CSOs in veteran mental health care, and provides psychoeducation and skills to support crisis management. The intervention was developed for all veterans who do not securely store firearms.
Theory
The intervention incorporates motivational interviewing techniques, as ambivalence about changing firearm storage behaviors is common, particularly when veterans own firearms for safety or protection.6,14 Motivational interviewing is a collaborative approach that addresses a client’s ambivalence to change by eliciting and exploring the client’s own arguments related to change.14 An important aspect of developing this intervention was to ensure it would be culturally relevant to veteran firearm owners and their CSOs.15 Further, involvement of the CSO is intentional and meant to boost social support, a known buffering factor against suicide risk.12
Objectives
This intervention’s primary objective was for veteran participants to identify secure firearm storage practices and develop a plan for implementing them, including when a veteran or other household member experiences a mental health crisis. For CSOs, the primary objective is to learn how to help the veteran connect with mental health resources if needed and support secure firearm storage as necessary. The overall goal is to learn how to identify warning signs for suicide and how to respond to a mental health crisis through a collaborative process, including securing firearms in a crisis situation.
Materials, Educational Strategies, and Instructors
Training for delivering the intervention was provided via direct consultation with the developer of the intervention and manual. The manual contains pertinent background information to provide context for the intervention’s significance and rationale, including the role of firearms in suicides and current lethal means safety initiatives. It also describes the purpose and objective of each intervention component in detail in addition to providing a script for interventionists to follow to complete each objective.
Training materials for veterans and CSOs include a single Firearms Secure Storage Planning worksheet completed during the intervention, with which the interventionist guides participants through the creation of a secure firearm storage plan (Table). Educational strategies include psychoeducation and Socratic questioning (eg, questioning focused on guiding participants toward the intervention goals) delivered verbally by the interventionist.

The intervention is delivered in person or virtually during a single 90-minute session with a veteran and CSO. Veterans and CSOs work with the interventionist to complete collaborative activities during the session and have self-directive learning activities or homework.
The intervention has 4 primary components: (1) CSO involvement; (2) psychoeducation; (3) secure firearm storage; and (4) how to respond to a mental health crisis. Each CSO should have an established relationship with the veteran, be willing and able to be present during the intervention, and remain an encouraging support person for the veteran. The interventionist emphasizes that it is part of the VA mission for staff to care about the veteran, and that initiating such contact with a CSO is meant to prioritize veteran safety and the safety of their family. Psychoeducation on mental health symptoms, suicide warning signs, veteran suicide rates and lethal means, and the benefits of secure firearm storage, is incorporated in the intervention.
The secure firearm storage component consists of 7 subcomponents: (1) general lethal means secure storage; (2) warning signs; (3) dyad communication; (4) lethal means safety when symptoms emerge; (5) coping strategies; (6) social support; and (7) emergency contacts. A lethal means safety worksheet rooted in the Stanley and Brown suicide safety plan model and implemented in VA health care settings is used to facilitate discussions of secure storage (Appendix).16

CSOs typically have little or no suicidal crisis response training, yet they likely have more interaction with the veteran on a daily basis than HCPs, putting them in a vital position to identify a crisis early and connect the veteran with the proper care. The crisis component prepares the CSO and veteran to navigate a crisis scenario so they can practice their newly developed safety plan and increase their comfort in discussing mental health and suicidal crisis.
FICTIONAL CASE STUDY
Cole, aged 59 years, is a Persian Gulf War veteran and retired police officer. His medical history includes hypothyroidism, hypertension, type 2 diabetes mellitus, chronic posttraumatic stress disorder, major depressive disorder, and insomnia.
Cole's wife of > 30 years, Sheila, joined him for the FFAST intervention. They report having 4 firearms in the home, 3 of which are loaded but stored in a lockbox and 1 that Cole reports is kept on his person for protection. Cole reports passive suicidal ideation, but no plans or intent. When discussing warning signs that a mental health crisis is building, Cole describes feeling anxious, having a change in his speech patterns, and isolating himself. Sheila agrees, but also mentions that Cole is easily angered and becomes nonverbal. Cole and Sheila express difficulty communicating and appear to have a breakthrough moment when Cole says he does not like when Sheila repeats herself, as he feels like she is “poking” at him. Sheila shares concerns for his safety and that she only repeats herself because he refuses to talk.
Cole agrees to verbalize that he is safe but needs time to process his thoughts. Sheila agrees to give him space with a plan to revisit the conversation within an agreed upon timeline. When discussing an updated secure storage plan for their firearms when a mental health crisis is building, Cole commits to allowing Sheila to store the firearm currently on his person in their gun safe, with the ammunition stored separately, and to giving her the gun safe key. They agree to implement this practice until the mental health crisis has passed.
To mitigate a potential crisis, the interventionist discusses possible internal coping strategies for Cole, including writing, reading, walking the dog, listening to music, and baking. People and social settings that could provide distraction involve going to the gym, talking to his friend Carl or his daughter Kelly, and attending the men’s ministry at church. The intervention concludes by discussing professionals or agencies that Cole and Sheila could contact during a crisis. After the intervention, Cole and Sheila are asked to rate their likelihood of using the plan they established during the conversation on a scale of 0 to 10, with 0 being highly unlikely and 10 being extremely likely. Cole responds with 9 and Sheila responds with 10.
DISCUSSION
Lethal means safety remains a critical component of veteran suicide prevention. However, lethal means safety discussions are often implemented after suicide risk has been identified, which may be too late. Thus, having these conversations early and before a crisis may be imperative. Veterans have expressed a desire to have CSOs involved in their suicide prevention treatment, and CSOs can play a key role in recognizing risk factors during everyday life. The FFAST intervention addresses many of these gaps.
Having discussions in advance of a crisis allows veterans to consider an effective secure firearm storage plan outside of the context of a crisis. Including a CSO galvanizes another person to understand a veteran’s needs and assist with secure firearm storage, identify warning signs, and support them during a crisis. These discussions occur in a context where there is less pressure than during a crisis. Features that were more appealing to veterans and their CSOs were also incorporated, such as having the dyad build a plan that is conceptually similar to other public safety initiatives (eg, a fire safety plan, tornado plan, or hurricane plan). Previous research demonstrates that veterans appreciated the nonjudgmental approach and some preferred that clinicians approach the discussion of secure firearm storage within the context of general home and family safety.17 Additionally, this intervention can build on veterans’ prior military training in preparedness.
Other potential benefits associated with the FFAST intervention include creating an opportunity to strengthen communication between the veteran and CSO. While FFAST is intended to be used with all types of CSOs, this work is consistent with preliminary data from a couples-based suicide prevention study that indicated veterans and their partners reported increases in relationship functioning and marginal decreases in suicidal ideation.18 It is possible that communication strategies gained from the current intervention could improve veterans’ relationships with their CSOs, which are associated with a greater sense of social support and reduced suicide risk.12
The intervention is a brief, single session that may be appealing to veterans and CSOs with full schedules. Evidence suggests that even brief, single-session interventions have a significant impact on beliefs about secure firearm storage, knowledge of lethal means safety, and confidence in having secure firearm storage conversations.19 However, clinicians should be cautious when extrapolating from the findings of the current case example, which was a one-time intervention with no follow-up.
Future Directions
Pilot testing of the proposed intervention is underway, and future research will include feedback from veterans and CSOs, as well as feasibility and acceptability data collected during the pilot process. The pilot study uses a successive cohort design with an initial 2 sets of 5 veteran and CSO dyads, and subsequent funding has expanded the pilot study to include an additional 30 dyads. Qualitative interviews will be conducted separately with each veteran and CSO, and additional constructs such as feasibility, acceptability, barriers and facilitators to implementation, and changes in secure storage will be examined. This future research may provide a deeper understanding of the broader acceptability, feasibility, and satisfaction associated with a suicide prevention intervention focused on securing firearms and involving veterans and their CSOs. These data could be used to inform future implementation trials and inform the development of an implementation strategy. In the interim, the nature of the manual is summarized in the context of the urgency of suicide prevention in this at-risk population.
Conclusions
FFAST is a novel approach to veteran firearm suicide prevention. By involving CSOs and emphasizing mental health crisis preparedness between them and veterans, the dyad can work in association with HCPs to establish and exercise secure firearm storage practices as part of an at-home safety plan. Implementation of FFAST may be beneficial for all veterans, not only those who have been identified as being at high suicide risk.
- US Dept of Veterans Affairs Office of Suicide Prevention. 2024 national veteran suicide prevention annual report. December 2024. Accessed February 5, 2026. https://www.mentalhealth.va.gov/docs/data-sheets/2024/2024-Annual-Report-Part-2-of-2_508.pdf
- Fischer IC, Aunon FM, Nichter B, et al. Firearm ownership among a nationally representative sample of U.S. veterans. Am J Prev Med. 2023;65:1129-1133. doi:10.1016/j.amepre.2023.06.013
- Conner A, Azrael D, Miller M. Suicide case-fatality rates in the United States, 2007-2014: a nationwide population-based study. Ann Intern Med. 2019;171(12):885-895. doi:10.7326/M19-1324
- Dempsey CL, Benedek DM, Zuromski KL, et al. Association of firearm ownership, use, accessibility, and storage practices with suicide risk among US army soldiers. JAMA Netw Open. 2019;2:e195383. doi:10.1001/jamanetworkopen.2019.5383
- Butterworth SE, Daruwala SE, Anestis MD. Firearm storage and shooting experience: factors relevant to the practical capability for suicide. J Psychiatr Res. 2018;102:52-56. doi:10.1016/j.jpsychires.2018.03.010
- Simonetti JA, Azrael D, Miller M. Firearm storage practices and risk perceptions among a nationally representative sample of U.S. veterans with and without self-harm risk factors. Suicide Life Threat Behav. 2019;49:653-664. doi:10.1111/sltb.12463
- Simonetti JA, Azrael D, Zhang W, Miller M. Receipt of clinician-delivered firearm safety counseling among U.S. veterans: results from a 2019 national survey. Suicide Life Threat Behav. 2022;52:1121-1125. doi:10.1111/sltb.12906
- US Office of the Surgeon General. The surgeon general’s call to action to implement the national strategy for suicide prevention. January 2021. Accessed February 5, 2026. https://www.hhs.gov/sites/default/files/sprc-call-to-action.pdf
- DeBeer BB, Matthieu MM, Kittel JA, et al. Quality Improvement Evaluation of the Feasibility and Acceptability of Adding a Concerned Significant Other to Safety Planning for Suicide Prevention With Veterans. J Ment Health Couns. 2019;41:4-20. doi:10.17744/mehc.41.1.02
- DeBeer BB, Matthieu MM, Degutis LC, et al. Firearms lethal means safety among veterans: attitudes toward involving a concerned significant other. J Mil Veteran Fam Health. 2025;11:23-31.
- Monteith LL, Holliday R, Dorsey Holliman BA, et al. Understanding female veterans’ experiences and perspectives of firearms. J Clin Psychol. 2020;76:1736-1753. doi:10.1002/jclp.22952
- DeBeer BB, Kimbrel NA, Meyer EC, et al. Combined PTSD and depressive symptoms interact with post-deployment social support to predict suicidal ideation in Operation Enduring Freedom and Operation Iraqi Freedom veterans. Psychiatry Res. 2014;216:357-362. doi:10.1016/j.psychres.2014.02.010
- Phillips AC, Lewis LK, McEvoy MP, et al. Development and validation of the guideline for reporting evidence-based practice educational interventions and teaching (GREET). BMC Med Educ. 2016;16:237. doi:10.1186/s12909-016-0759-1
- Miller WR, Rollnick S. Motivational Interviewing: Helping People Change. 3rd ed. Guilford Press; 2013.
- Khazanov GK, Keddem S, Hoskins K, et al. Stakeholder perceptions of lethal means safety counseling: a qualitative systematic review. Front Psychiatry. 2022;13:993415. doi:10.3389/fpsyt.2022.993415
- Stanley B, Brown GK, Karlin B, et al. US Dept of Veterans Affairs. Safety plan treatment manual to reduce suicide risk: veteran version. August 20, 2008. Accessed February 5, 2026. https://www.mentalhealth.va.gov/mentalhealth/docs/va_safety_planning_manual.doc
- Dobscha SK, Clark KD, Newell S, et al. Strategies for discussing firearms storage safety in primary care: veteran perspectives. J Gen Intern Med. 2021;36:1492-1502. doi:10.1007/s11606-020-06412-x
- Khalifian CE, Leifker FR, Knopp K, et al. Utilizing the couple relationship to prevent suicide: a preliminary examination of treatment for relationships and safety together. J Clin Psych. 2022;78:747-757. doi:10.1002/jclp.23251
- Walsh A, Friedman K, Morrissey BH, et al. Project Safe Guard: evaluating a lethal means safety intervention to reduce firearm suicide in the National Guard. Mil Med. 2024;189:510-516. doi:10.1093/milmed/usae172
- Beck AT. Beyond belief: a theory of modes, personality, and psychopathology. In: Salkovkis PM, ed. Frontiers of Cognitive Therapy. Guilford Press;1996:1-25.
- Rudd MD. The suicidal mode: a cognitive-behavioral model of suicidality. Suicide Life Threat Behav. 2000;30(1):18-33.
- US Dept of Veterans Affairs Office of Suicide Prevention. 2024 national veteran suicide prevention annual report. December 2024. Accessed February 5, 2026. https://www.mentalhealth.va.gov/docs/data-sheets/2024/2024-Annual-Report-Part-2-of-2_508.pdf
- Fischer IC, Aunon FM, Nichter B, et al. Firearm ownership among a nationally representative sample of U.S. veterans. Am J Prev Med. 2023;65:1129-1133. doi:10.1016/j.amepre.2023.06.013
- Conner A, Azrael D, Miller M. Suicide case-fatality rates in the United States, 2007-2014: a nationwide population-based study. Ann Intern Med. 2019;171(12):885-895. doi:10.7326/M19-1324
- Dempsey CL, Benedek DM, Zuromski KL, et al. Association of firearm ownership, use, accessibility, and storage practices with suicide risk among US army soldiers. JAMA Netw Open. 2019;2:e195383. doi:10.1001/jamanetworkopen.2019.5383
- Butterworth SE, Daruwala SE, Anestis MD. Firearm storage and shooting experience: factors relevant to the practical capability for suicide. J Psychiatr Res. 2018;102:52-56. doi:10.1016/j.jpsychires.2018.03.010
- Simonetti JA, Azrael D, Miller M. Firearm storage practices and risk perceptions among a nationally representative sample of U.S. veterans with and without self-harm risk factors. Suicide Life Threat Behav. 2019;49:653-664. doi:10.1111/sltb.12463
- Simonetti JA, Azrael D, Zhang W, Miller M. Receipt of clinician-delivered firearm safety counseling among U.S. veterans: results from a 2019 national survey. Suicide Life Threat Behav. 2022;52:1121-1125. doi:10.1111/sltb.12906
- US Office of the Surgeon General. The surgeon general’s call to action to implement the national strategy for suicide prevention. January 2021. Accessed February 5, 2026. https://www.hhs.gov/sites/default/files/sprc-call-to-action.pdf
- DeBeer BB, Matthieu MM, Kittel JA, et al. Quality Improvement Evaluation of the Feasibility and Acceptability of Adding a Concerned Significant Other to Safety Planning for Suicide Prevention With Veterans. J Ment Health Couns. 2019;41:4-20. doi:10.17744/mehc.41.1.02
- DeBeer BB, Matthieu MM, Degutis LC, et al. Firearms lethal means safety among veterans: attitudes toward involving a concerned significant other. J Mil Veteran Fam Health. 2025;11:23-31.
- Monteith LL, Holliday R, Dorsey Holliman BA, et al. Understanding female veterans’ experiences and perspectives of firearms. J Clin Psychol. 2020;76:1736-1753. doi:10.1002/jclp.22952
- DeBeer BB, Kimbrel NA, Meyer EC, et al. Combined PTSD and depressive symptoms interact with post-deployment social support to predict suicidal ideation in Operation Enduring Freedom and Operation Iraqi Freedom veterans. Psychiatry Res. 2014;216:357-362. doi:10.1016/j.psychres.2014.02.010
- Phillips AC, Lewis LK, McEvoy MP, et al. Development and validation of the guideline for reporting evidence-based practice educational interventions and teaching (GREET). BMC Med Educ. 2016;16:237. doi:10.1186/s12909-016-0759-1
- Miller WR, Rollnick S. Motivational Interviewing: Helping People Change. 3rd ed. Guilford Press; 2013.
- Khazanov GK, Keddem S, Hoskins K, et al. Stakeholder perceptions of lethal means safety counseling: a qualitative systematic review. Front Psychiatry. 2022;13:993415. doi:10.3389/fpsyt.2022.993415
- Stanley B, Brown GK, Karlin B, et al. US Dept of Veterans Affairs. Safety plan treatment manual to reduce suicide risk: veteran version. August 20, 2008. Accessed February 5, 2026. https://www.mentalhealth.va.gov/mentalhealth/docs/va_safety_planning_manual.doc
- Dobscha SK, Clark KD, Newell S, et al. Strategies for discussing firearms storage safety in primary care: veteran perspectives. J Gen Intern Med. 2021;36:1492-1502. doi:10.1007/s11606-020-06412-x
- Khalifian CE, Leifker FR, Knopp K, et al. Utilizing the couple relationship to prevent suicide: a preliminary examination of treatment for relationships and safety together. J Clin Psych. 2022;78:747-757. doi:10.1002/jclp.23251
- Walsh A, Friedman K, Morrissey BH, et al. Project Safe Guard: evaluating a lethal means safety intervention to reduce firearm suicide in the National Guard. Mil Med. 2024;189:510-516. doi:10.1093/milmed/usae172
- Beck AT. Beyond belief: a theory of modes, personality, and psychopathology. In: Salkovkis PM, ed. Frontiers of Cognitive Therapy. Guilford Press;1996:1-25.
- Rudd MD. The suicidal mode: a cognitive-behavioral model of suicidality. Suicide Life Threat Behav. 2000;30(1):18-33.
Involving Concerned Significant Others in Firearm Suicide Prevention: Development of the Family FireArms Secure Storage Training Intervention
Involving Concerned Significant Others in Firearm Suicide Prevention: Development of the Family FireArms Secure Storage Training Intervention
Ask the Expert Geriatric Psychiatry: A VHA Email Consultation Program to Support Clinicians
Ask the Expert Geriatric Psychiatry: A VHA Email Consultation Program to Support Clinicians
T he US Census Bureau projects that the number of older adults (aged ≥ 65 years) will exceed 49 million by 2030, and an estimated 20% (nearly 10 million) of this population will experience cognitive or mental health disorders.1,2 The mental health workforce is not equipped to address the specialized mental health care needs of many older adults.2,3 For example, geriatric psychiatrists specialize in the diagnosis and treatment of mental illness and cognitive disorders in the later stages of life, but their numbers are few and declining. Only 33.5% of geriatric psychiatry fellowship training slots were filled from 2017 to 2021, and only 62 fellows trained during the 2021-2022 academic year.4 Board-certified geriatric psychiatrists also tend to be concentrated in larger, urban, academically-affiliated medical centers, often leaving rural areas and smaller facilities without access, including facilities in the Veterans Health Administration (VHA).5
The VHA has been optimizing access to specialty geriatric mental health services via regional and national virtual consultation services. Seven of 19 Veterans Integrated Service Network (VISN) Clinical Resource Hubs (CRHs) have geriatric mental health teams.6 These provide interdisciplinary geriatric mental telehealth services, including geriatric psychiatry, for older veterans with complex care needs.7,8 Likewise, the VHA National Expert Consultation & Specialized Services-Mental Health (NEXCSS- MH, formerly known as the National Telemental Health Center) sponsors video teleconsultations with board-certified geriatric psychiatrists and an Ask the Expert email consultation program.
This article describes the Ask the Expert Geriatric Psychiatry email program (one of several similar programs at NEXCSS-MH), building upon a symposium presented at the American Association for Geriatric Psychiatry (AAGP) annual meeting in March 2022.9 The program was initiated in June 2021 as a result of discussions between the National Mental Health Director, Geriatric Mental Health in the VHA Office of Mental Health and Suicide Prevention (now known as the Office of Mental Health [OMH]), and National Telemental Health Center leadership. VHA board certified geriatric psychiatrists were recruited to serve as expert consultants and respond to email questions submitted by VHA clinicians regarding the psychiatric care of older adult veterans. The results of this program identify educational needs among clinical staff and may inform the development of program materials for a range of clinicians.
Program Description
The national geriatric mental health director recruited prospective experts and met with each to assess interest and qualifications, consulting with OMH psychiatrist leaders before making selections. Five experts were initially selected; 1 later stepped down and was replaced by another, who also stepped down. The experts were board certified in psychiatry and geriatric psychiatry and held a variety of local and national leadership positions, including geriatric psychiatry fellowship director, US Department of Veterans Affairs (VA) research and clinical leader, and various roles in the AAGP; some had received teaching awards.
Operations
The national geriatric mental health director announced the program in June 2021 to VHA mental health and geriatric program email groups with reminders sent every few months. The announcement included information about the types of questions appropriate to submit, including examples of general clinical management questions that did not share patient-specific protected health information, and clarified that experts would not be conducting chart reviews because the time required for detailed chart reviews was not feasible for volunteer experts to integrate into their otherwise full-time jobs at their respective VA medical centers. The announcement also included brief biographies of the experts.
The Figure describes the daily operations of the Ask the Expert Geriatric Psychiatry email consultation program. The NEXCSS- MH developed a Microsoft Outlook mailbox and group email address where clinicians from across the VHA could submit questions. The experts, as well as the national geriatric mental health director and NEXCSS-MH staff, had access to this mailbox to track and/or respond to questions. One expert volunteered to be the program’s primary mailbox coordinator. The coordinator checked the inbox daily and assigned each question to one of the experts on a rotating basis using the color-coding feature in Outlook. The other experts were advised to check the email account at least once weekly and reply to any assigned questions.
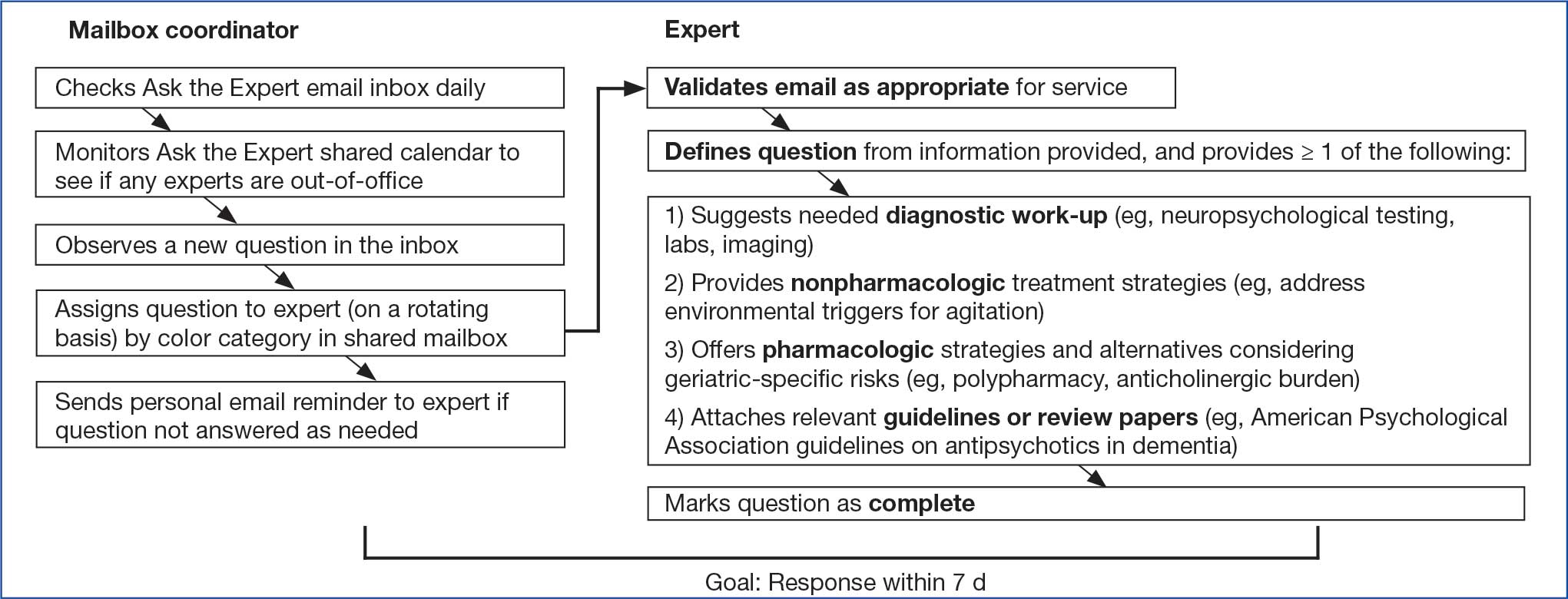
Responding to a question entailed first determining whether the question was appropriate for the service. For example, if a question requested a chart review, the expert replied that experts could not provide chart reviews and requested that the question be reframed. Next, the expert often needed to define a specific clinical question from the information provided, as email questions often touched upon several topics. The expert provided personalized advice on diagnostic testing, nonpharmacologic treatment strategies, and/or pharmacologic treatment options. Experts also often attached relevant guidelines or review articles. The goal was to provide a response within 7 business days.
All email responses included a disclaimer indicating that the program was not intended for urgent or immediate medical advice and that the information provided was for VHA clinician education purposes only. The disclaimer explained that email communication did not establish a doctor-patient relationship between the expert and a specific veteran and that, if desired, a request for a clinical consultation could be submitted on a specific case (ie, a video teleconsultation).
Methods for Reviewing Questions
Descriptive statistics, including frequencies, means, and minimum and maximum ranges, were used to capture the number of questions the program received, type of requester, and length of time prior to response for emailed questions.9 Conventional content analysis procedures were used between January and October 2024 to analyze clinicians’ questions.10 Four subject matter experts (3 geriatric psychiatrists and 1 geropsychologist) served as coders, assigned in groups of 2 to review questions. Each coder independently reviewed assigned questions and identified preliminary themes. Themes were reviewed and revised using an iterative process during regular team meetings with coders to clarify and confirm interpretations. Discrepancies were discussed within team meetings to achieve consensus.
Questions received. Between February 2022 and December 2023, the program received 101 email questions. Requesters included 39 physicians, 17 nurse practitioners or physician assistants, 15 social workers, 14 psychologists, 9 nurses, 5 pharmacists, 1 dietitian, and 1 who was undetermined. Experts responded to the questions an average of 6 days after receipt (range, < 1-19); 73 responses (72%) met the 7-day goal.
Iterative changes to coded themes were made during group discussions. Multiple clinical questions were often posed within the same email. Initially, some coders identified themes solely based on reported symptoms; others identified themes based on reported and/or potential diagnostic conditions attributed to the symptom(s) described within the email. For example, some coders selected a primary theme of behavioral and psychological symptoms of dementia (BPSD) only if a behavior contributing to distress in the veteran or others was described, while others selected this theme when any psychiatric symptom (eg, psychosis) was present in the context of dementia. The group identified 1 primary theme per question based on the main clinical symptom or main concern presented. Co-occurring diagnostic conditions highlighted in the email requests were included as secondary themes, and each question could have > 1 secondary theme.
The most frequent requests related to clinical symptoms included questions about agitated behaviors, sleep and/or nightmares, and depression symptoms (Table 1). Twenty-seven of 33 email requests on agitated behaviors were related to a dementia diagnosis, as were several questions about sleep/nightmares, depression, psychosis/mania, and anxiety. Many diagnostic conditions were described in the email requests (Table 2). The most frequent condition was dementia, followed by a medical condition, depressive disorder, posttraumatic stress disorder, and/or serious mental illness.
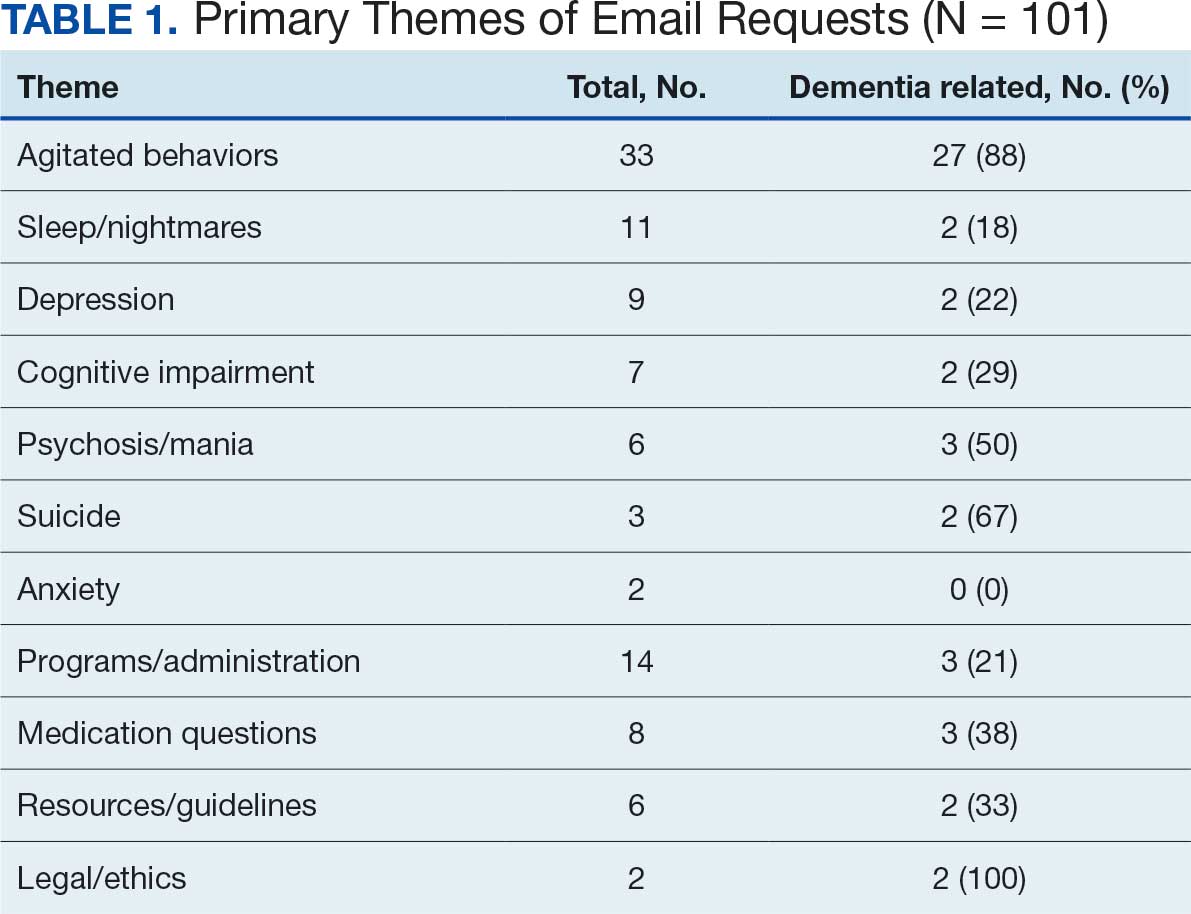

Request for Feedback. In February 2022, an email request was sent to the 64 clinicians who asked email questions from the start of the program in June 2021 through December 2021. A second request included 11 clinicians who asked questions from January through February 2022. These requests were sent as part of preparations for the symposium on the program presented at the AAGP annual meeting in March 2022.9 In May 2024, feedback was requested from 37 clinicians who submitted questions from May 1, 2023, through May 15, 2024.
Requests for feedback included 6 closed-ended and 1 open-ended question: (1) Did the answer you received help inform clinical practice? (2) Did you receive a timely response? (3) What type of information was useful to you in addressing your question (ie, direct/specific answer to a clinical scenario, guidelines, articles, VA resources)? (4) Do you have access to a geriatric psychiatrist at your facility? (5) Are you likely to use Ask the Expert Geriatric Psychiatry in the future? (6) Would you use a geriatric psychiatry teleconsultation service? (7) Share suggestions for improvement. Frequencies of response selection were obtained for each question. Text responses to the open-ended question asking for suggestions for improvement were reviewed and summarized.
Responses
Thirty users responded to the feedback request (27% response rate). Respondents considered the answers received extremely (n = 14; 47%) or very much (n = 12; 40%) helpful for their clinical practice. Twenty-three respondents (77%) felt an answer was provided promptly, 7 respondents (23%) felt the answer was not timely but still useful, and none felt that the answer was too late. Respondents reported that the most useful type of information in addressing their questions was a direct/specific answer to a clinical scenario (n = 27; 90%), followed by guidelines (n = 12; 40%), articles (n = 7; 23%), and VA resources (n = 4; 13%).
Sixteen respondents (53%) reported that they rarely had ready access to a geriatric psychiatrist at their facility, 3 (10%) had access sometimes, 4 (14%) had access usually, 3 (10%) had access regularly, and 3 (10%) never had access. Twenty-seven respondents (90%) indicated they would be very likely to use the service again. If geriatric psychiatry teleconsultation and/or e-consultation were offered, many respondents indicated they would be extremely (n = 10; 33%) or very (n = 12; 40%) likely to use teleconsultation and/or e-consultation.
Suggestions for improvement included supporting experts to perform chart reviews for email questions, developing a template or consult form, holding a biweekly drop-in meeting to present questions to and discuss cases with a panel of experts, and providing further help addressing complex decisional capacity issues, delirium, and care or placement for veterans with severe behavioral issues in a rural setting.
Discussion
Although many older adults experience cognitive and mental health disorders that may benefit from management by a geriatric psychiatrist, the number of trained geriatric psychiatrists available is insufficient to allow for direct care for each patient. The Ask the Expert Geriatric Psychiatry email consultation program is one aspect of a multicomponent strategy within the VHA to increase access to specialty geriatric mental health services for veterans. A key advantage of the program is that it is not resource intensive. Experts can participate voluntarily, providing timely feedback to clinicians around the country while continuing other duties at their respective VA medical centers. Email replies to the experts’ answers elicited positive feedback on the program, include: “I found this service to be extremely helpful and I have shared the information they sent me with several other coworkers!”, “It was great!”, and “I endorsed the service to our VISN Rehabilitation and Extended Care group.”
The coding of primary and secondary themes from 101 email questions that were retained revealed the range and relative frequencies of clinical and administrative topics with which clinicians needed help. The most common (33%) theme was agitated behaviors. Nearly half of the questions (48%) were related to underlying dementia, and 29% were related to a patient’s medical comorbidities. These findings suggest that the expertise of a geriatric psychiatrist is particularly relevant when caring for older patients experiencing BPSD or patients with complex, overlapping psychiatric and medical conditions.
Despite a 27% response rate, participant feedback has been helpful. The program reached its intended audience of clinicians in rural areas and at smaller facilities with 53% of requesters reporting they rarely had access to a geriatric psychiatrist. Suggestions for improvement indicated that some clinicians desired additional support, including chart reviews, meetings with experts, and a video teleconsultation service (available through NEXCSS-MH).
Many clinicians without training in specialty geriatric mental health may require help with complex clinical presentations. For example, 39 clinicians who submitted questions to the program were physicians. Accreditation Council for Graduate Medical Education program requirements for general psychiatry residency include 4 weeks of geriatric psychiatry.11 The findings of this study suggest that this level of training may not be adequate to independently care for every patient who experiences dementia or multimorbidity. Several training and mentoring initiatives have been developed to address the professional development need for psychiatrists.12-14
The need for geriatric workforce development is significant across health care, including other mental health professions.15,16 The VHA Geriatric Scholars program trains rural primary care practitioners, psychologists, and psychiatrists.17,18 Likewise, consultative geriatric specialty support for primary care practitioners in rural areas is provided via the Geriatric Research Education and Clinical Center Connect program.19 The Ask the Expert Geriatric Psychiatry email program is an additional economical model to support clinician educational development and provide rapid educational responses to inform patient care.
Ask the Expert received fewer email questions than anticipated. Enhanced optimization may require more frequent and widespread announcements about the program. Clinical staff may not be aware of the program due to an overload of email communications. Likewise, it may be challenging for busy clinicians to take the time to seek consultation or recognize a potential gap in their knowledge or skills. Had more questions been submitted, the 5 volunteer experts may have had more difficulty addressing the demand. Feedback from this project may inform development of a frequently asked questions document to share with VHA teams and a drop-in office hour to pose clinical questions of geriatric psychiatry experts, as recommended by a clinician who participated in the program.
Limitations
Not all requesters were sent a request for feedback, and the response rate for the request for feedback was only 27%. As the program has evolved, it began sending a request for feedback immediately after answering each question, which may increase the odds of response. The goal of experts answering questions within 7 business days was met 72% of the time, likely an artifact of experts integrating question answering with many other duties. The mailbox coordinator has since provided email prompts to experts immediately upon being assigned a question with the goal of improving timeliness. The program did not include chart reviews or patient consultations, as neither was feasible for volunteer experts. The email consultation service is a single component of virtual consultative specialty geriatric mental health services within the VHA, including video consultations via NEXCSS-MH and regional geriatric mental health teams.
Conclusions
The need for specialty geriatric mental health services is increasing in the VHA and across the US. However, there are too few board-certified geriatric psychiatrists to provide direct patient care to all older adults with cognitive and mental health disorders. The VHA has leveraged telehealth to improve access to geriatric mental health care. The VHA Ask the Expert Geriatric Psychiatry email consultation program is a low-resource service which provides rapid feedback to clinicians nationwide on challenging clinical scenarios, many of which are dementia-related. Most users of the service who responded to requests for feedback reported that answers to their questions were helpful and timely. The email consultation program should continue to be supplemented by more comprehensive geriatric telemental health services for particularly complex cases to meet the needs of older veterans.
- 2023 population projections for the nation by age, sex, race, Hispanic origin and nativity. United States Census Bureau. November 9, 2023. Accessed December 11, 2025. https://www.census.gov/newsroom/press-kits/2023/population-projections.html
- National Academies of Sciences Engineering and Medicine. Addressing the rising mental health needs of an aging population: proceedings of a workshop. 2024. Accessed December 11, 2025. doi.org:10.17226/27340
- Institute of Medicine. The mental health and substance use workforce for older adults: in whose hands? The National Academies Press; 2012. Accessed December 11, 2025. doi:10.17226/13400
- American Psychiatric Association. 2022 resident/fellow census. November 2023. Accessed December 11, 2025. https://www.psychiatry.org/getmedia/d80438af-f760-40f3-9d33-f91309b09564/APA-Resident-Census-2022.pdf
- Juul D, Colenda CC, Lyness JM, et al. Subspecialty training and certification in geriatric psychiatry: a 25-year overview. Am J Geriatr Psychiatry. 2017;25:445-453. doi:10.1016/j.jagp.2016.12.018
- Jaske E, Wheat CL, Rubenstein LV, et al. Understanding how contingency staffing programs can support mental health services in the Veterans Health Administration. Telemed J E Health. 2024;30:1857-1865. doi:10.1089/tmj.2023.0573
- Gould CE, Carlson C, Alfaro AJ, et al. Supporting veterans, caregivers, and providers in rural regions with tele-geriatric psychiatry consultation: a mixed methods pilot study. Am J Geriatr Psychiatry. 2023;31:279-290. doi:10.1016/j.jagp.2023.01.005
- Gould CE, Paiko L, Carlson C, et al. Implementation of tele-geriatricmental healthcare for rural veterans: factors influencing care models. Front Health Serv. 2024;4:1221899. doi:10.3389/frhs.2024.1221899
- Padala P, Schultz S, Khatkhate G, et al. Ask the expert geriatric psychiatry: VA program to support clinicians. Am J Geriatr Psychiatry. 2022;30:S18. doi:10.1016/j.jagp.2022.01.279
- Hsieh HF, Shannon SE. Three approaches to qualitative content analysis. Qual Health Res. 2005;15:1277-1288. doi:10.1177/1049732305276687
- Accreditation Council for Graduate Medical Education. Program requirements for graduate medical education in psychiatry. Revised September 3, 2025. Accessed December 11, 2025. https://www.acgme.org/globalassets/pfassets/programrequirements/2025-reformatted-requirements/400_psychiatry_2025_reformatted.pdf
- Fernandez J, Agarwal KS, Amspoker AB, et al. Outcomes from an interprofessional, dementia-focused, telementoring program: a brief report. Gerontol Geriatr Educ. 2024;45:601-606. doi:10.1080/02701960.2023.2253175
- Conroy ML, Garcia-Pittman EC, van Dyck LI, et al. The COVID-19 American Association for Geriatric Psychiatry (AAGP) online trainee curriculum: program evaluation and future directions. Am J Geriatr Psychiatry. 2025;33:308-314. doi:10.1016/j.jagp.2024.10.010
- Conroy ML, Garcia-Pittman EC, Ali H, et al. The COVID-19 AAGP online trainee curriculum: development and method of initial evaluation. Am J Geriatr Psychiatry. 2020;28:1004-1008. doi:10.1016/j.jagp.2020.06.003
- Flaherty E, Busby-Whitehead J, Potter J, et al. The geriatric workforce enhancement program: review of the coordinating center and examples of the GWEP in practice. Am J Geriatr Psychiatry. 2019;27:675-686. doi:10.1016/j.jagp.2019.04.010
- Hoge MA, Karel MJ, Zeiss AM, et al. Strengthening psychology’s workforce for older adults: implications of the Institute of Medicine’s report to Congress. Am Psychol. 2015;70:265-278. doi:10.1037/a0038927
- Kramer BJ, Creekmur B, Howe JL, et al. Veterans Affairs geriatric scholars program: enhancing existing primary care clinician skills in caring for older veterans. J Am Geriatr Soc. 2016;64:2343-2348. doi:10.1111/jgs.14382
- Gould CE, Rodriguez RL, Gregg JJ, et al. Preparing Veterans Health Administration psychologists to meet the complex needs of aging veterans. Fed Pract. 2024;41:S10-S15. doi:10.12788/fp.0466
- Pimentel CB, Gately M, Barczi SR, et al. GRECC Connect: Geriatrics telehealth to empower health care providers and improve management of older veterans in rural communities. Fed Pract. 2019;36:464-470.
T he US Census Bureau projects that the number of older adults (aged ≥ 65 years) will exceed 49 million by 2030, and an estimated 20% (nearly 10 million) of this population will experience cognitive or mental health disorders.1,2 The mental health workforce is not equipped to address the specialized mental health care needs of many older adults.2,3 For example, geriatric psychiatrists specialize in the diagnosis and treatment of mental illness and cognitive disorders in the later stages of life, but their numbers are few and declining. Only 33.5% of geriatric psychiatry fellowship training slots were filled from 2017 to 2021, and only 62 fellows trained during the 2021-2022 academic year.4 Board-certified geriatric psychiatrists also tend to be concentrated in larger, urban, academically-affiliated medical centers, often leaving rural areas and smaller facilities without access, including facilities in the Veterans Health Administration (VHA).5
The VHA has been optimizing access to specialty geriatric mental health services via regional and national virtual consultation services. Seven of 19 Veterans Integrated Service Network (VISN) Clinical Resource Hubs (CRHs) have geriatric mental health teams.6 These provide interdisciplinary geriatric mental telehealth services, including geriatric psychiatry, for older veterans with complex care needs.7,8 Likewise, the VHA National Expert Consultation & Specialized Services-Mental Health (NEXCSS- MH, formerly known as the National Telemental Health Center) sponsors video teleconsultations with board-certified geriatric psychiatrists and an Ask the Expert email consultation program.
This article describes the Ask the Expert Geriatric Psychiatry email program (one of several similar programs at NEXCSS-MH), building upon a symposium presented at the American Association for Geriatric Psychiatry (AAGP) annual meeting in March 2022.9 The program was initiated in June 2021 as a result of discussions between the National Mental Health Director, Geriatric Mental Health in the VHA Office of Mental Health and Suicide Prevention (now known as the Office of Mental Health [OMH]), and National Telemental Health Center leadership. VHA board certified geriatric psychiatrists were recruited to serve as expert consultants and respond to email questions submitted by VHA clinicians regarding the psychiatric care of older adult veterans. The results of this program identify educational needs among clinical staff and may inform the development of program materials for a range of clinicians.
Program Description
The national geriatric mental health director recruited prospective experts and met with each to assess interest and qualifications, consulting with OMH psychiatrist leaders before making selections. Five experts were initially selected; 1 later stepped down and was replaced by another, who also stepped down. The experts were board certified in psychiatry and geriatric psychiatry and held a variety of local and national leadership positions, including geriatric psychiatry fellowship director, US Department of Veterans Affairs (VA) research and clinical leader, and various roles in the AAGP; some had received teaching awards.
Operations
The national geriatric mental health director announced the program in June 2021 to VHA mental health and geriatric program email groups with reminders sent every few months. The announcement included information about the types of questions appropriate to submit, including examples of general clinical management questions that did not share patient-specific protected health information, and clarified that experts would not be conducting chart reviews because the time required for detailed chart reviews was not feasible for volunteer experts to integrate into their otherwise full-time jobs at their respective VA medical centers. The announcement also included brief biographies of the experts.
The Figure describes the daily operations of the Ask the Expert Geriatric Psychiatry email consultation program. The NEXCSS- MH developed a Microsoft Outlook mailbox and group email address where clinicians from across the VHA could submit questions. The experts, as well as the national geriatric mental health director and NEXCSS-MH staff, had access to this mailbox to track and/or respond to questions. One expert volunteered to be the program’s primary mailbox coordinator. The coordinator checked the inbox daily and assigned each question to one of the experts on a rotating basis using the color-coding feature in Outlook. The other experts were advised to check the email account at least once weekly and reply to any assigned questions.

Responding to a question entailed first determining whether the question was appropriate for the service. For example, if a question requested a chart review, the expert replied that experts could not provide chart reviews and requested that the question be reframed. Next, the expert often needed to define a specific clinical question from the information provided, as email questions often touched upon several topics. The expert provided personalized advice on diagnostic testing, nonpharmacologic treatment strategies, and/or pharmacologic treatment options. Experts also often attached relevant guidelines or review articles. The goal was to provide a response within 7 business days.
All email responses included a disclaimer indicating that the program was not intended for urgent or immediate medical advice and that the information provided was for VHA clinician education purposes only. The disclaimer explained that email communication did not establish a doctor-patient relationship between the expert and a specific veteran and that, if desired, a request for a clinical consultation could be submitted on a specific case (ie, a video teleconsultation).
Methods for Reviewing Questions
Descriptive statistics, including frequencies, means, and minimum and maximum ranges, were used to capture the number of questions the program received, type of requester, and length of time prior to response for emailed questions.9 Conventional content analysis procedures were used between January and October 2024 to analyze clinicians’ questions.10 Four subject matter experts (3 geriatric psychiatrists and 1 geropsychologist) served as coders, assigned in groups of 2 to review questions. Each coder independently reviewed assigned questions and identified preliminary themes. Themes were reviewed and revised using an iterative process during regular team meetings with coders to clarify and confirm interpretations. Discrepancies were discussed within team meetings to achieve consensus.
Questions received. Between February 2022 and December 2023, the program received 101 email questions. Requesters included 39 physicians, 17 nurse practitioners or physician assistants, 15 social workers, 14 psychologists, 9 nurses, 5 pharmacists, 1 dietitian, and 1 who was undetermined. Experts responded to the questions an average of 6 days after receipt (range, < 1-19); 73 responses (72%) met the 7-day goal.
Iterative changes to coded themes were made during group discussions. Multiple clinical questions were often posed within the same email. Initially, some coders identified themes solely based on reported symptoms; others identified themes based on reported and/or potential diagnostic conditions attributed to the symptom(s) described within the email. For example, some coders selected a primary theme of behavioral and psychological symptoms of dementia (BPSD) only if a behavior contributing to distress in the veteran or others was described, while others selected this theme when any psychiatric symptom (eg, psychosis) was present in the context of dementia. The group identified 1 primary theme per question based on the main clinical symptom or main concern presented. Co-occurring diagnostic conditions highlighted in the email requests were included as secondary themes, and each question could have > 1 secondary theme.
The most frequent requests related to clinical symptoms included questions about agitated behaviors, sleep and/or nightmares, and depression symptoms (Table 1). Twenty-seven of 33 email requests on agitated behaviors were related to a dementia diagnosis, as were several questions about sleep/nightmares, depression, psychosis/mania, and anxiety. Many diagnostic conditions were described in the email requests (Table 2). The most frequent condition was dementia, followed by a medical condition, depressive disorder, posttraumatic stress disorder, and/or serious mental illness.


Request for Feedback. In February 2022, an email request was sent to the 64 clinicians who asked email questions from the start of the program in June 2021 through December 2021. A second request included 11 clinicians who asked questions from January through February 2022. These requests were sent as part of preparations for the symposium on the program presented at the AAGP annual meeting in March 2022.9 In May 2024, feedback was requested from 37 clinicians who submitted questions from May 1, 2023, through May 15, 2024.
Requests for feedback included 6 closed-ended and 1 open-ended question: (1) Did the answer you received help inform clinical practice? (2) Did you receive a timely response? (3) What type of information was useful to you in addressing your question (ie, direct/specific answer to a clinical scenario, guidelines, articles, VA resources)? (4) Do you have access to a geriatric psychiatrist at your facility? (5) Are you likely to use Ask the Expert Geriatric Psychiatry in the future? (6) Would you use a geriatric psychiatry teleconsultation service? (7) Share suggestions for improvement. Frequencies of response selection were obtained for each question. Text responses to the open-ended question asking for suggestions for improvement were reviewed and summarized.
Responses
Thirty users responded to the feedback request (27% response rate). Respondents considered the answers received extremely (n = 14; 47%) or very much (n = 12; 40%) helpful for their clinical practice. Twenty-three respondents (77%) felt an answer was provided promptly, 7 respondents (23%) felt the answer was not timely but still useful, and none felt that the answer was too late. Respondents reported that the most useful type of information in addressing their questions was a direct/specific answer to a clinical scenario (n = 27; 90%), followed by guidelines (n = 12; 40%), articles (n = 7; 23%), and VA resources (n = 4; 13%).
Sixteen respondents (53%) reported that they rarely had ready access to a geriatric psychiatrist at their facility, 3 (10%) had access sometimes, 4 (14%) had access usually, 3 (10%) had access regularly, and 3 (10%) never had access. Twenty-seven respondents (90%) indicated they would be very likely to use the service again. If geriatric psychiatry teleconsultation and/or e-consultation were offered, many respondents indicated they would be extremely (n = 10; 33%) or very (n = 12; 40%) likely to use teleconsultation and/or e-consultation.
Suggestions for improvement included supporting experts to perform chart reviews for email questions, developing a template or consult form, holding a biweekly drop-in meeting to present questions to and discuss cases with a panel of experts, and providing further help addressing complex decisional capacity issues, delirium, and care or placement for veterans with severe behavioral issues in a rural setting.
Discussion
Although many older adults experience cognitive and mental health disorders that may benefit from management by a geriatric psychiatrist, the number of trained geriatric psychiatrists available is insufficient to allow for direct care for each patient. The Ask the Expert Geriatric Psychiatry email consultation program is one aspect of a multicomponent strategy within the VHA to increase access to specialty geriatric mental health services for veterans. A key advantage of the program is that it is not resource intensive. Experts can participate voluntarily, providing timely feedback to clinicians around the country while continuing other duties at their respective VA medical centers. Email replies to the experts’ answers elicited positive feedback on the program, include: “I found this service to be extremely helpful and I have shared the information they sent me with several other coworkers!”, “It was great!”, and “I endorsed the service to our VISN Rehabilitation and Extended Care group.”
The coding of primary and secondary themes from 101 email questions that were retained revealed the range and relative frequencies of clinical and administrative topics with which clinicians needed help. The most common (33%) theme was agitated behaviors. Nearly half of the questions (48%) were related to underlying dementia, and 29% were related to a patient’s medical comorbidities. These findings suggest that the expertise of a geriatric psychiatrist is particularly relevant when caring for older patients experiencing BPSD or patients with complex, overlapping psychiatric and medical conditions.
Despite a 27% response rate, participant feedback has been helpful. The program reached its intended audience of clinicians in rural areas and at smaller facilities with 53% of requesters reporting they rarely had access to a geriatric psychiatrist. Suggestions for improvement indicated that some clinicians desired additional support, including chart reviews, meetings with experts, and a video teleconsultation service (available through NEXCSS-MH).
Many clinicians without training in specialty geriatric mental health may require help with complex clinical presentations. For example, 39 clinicians who submitted questions to the program were physicians. Accreditation Council for Graduate Medical Education program requirements for general psychiatry residency include 4 weeks of geriatric psychiatry.11 The findings of this study suggest that this level of training may not be adequate to independently care for every patient who experiences dementia or multimorbidity. Several training and mentoring initiatives have been developed to address the professional development need for psychiatrists.12-14
The need for geriatric workforce development is significant across health care, including other mental health professions.15,16 The VHA Geriatric Scholars program trains rural primary care practitioners, psychologists, and psychiatrists.17,18 Likewise, consultative geriatric specialty support for primary care practitioners in rural areas is provided via the Geriatric Research Education and Clinical Center Connect program.19 The Ask the Expert Geriatric Psychiatry email program is an additional economical model to support clinician educational development and provide rapid educational responses to inform patient care.
Ask the Expert received fewer email questions than anticipated. Enhanced optimization may require more frequent and widespread announcements about the program. Clinical staff may not be aware of the program due to an overload of email communications. Likewise, it may be challenging for busy clinicians to take the time to seek consultation or recognize a potential gap in their knowledge or skills. Had more questions been submitted, the 5 volunteer experts may have had more difficulty addressing the demand. Feedback from this project may inform development of a frequently asked questions document to share with VHA teams and a drop-in office hour to pose clinical questions of geriatric psychiatry experts, as recommended by a clinician who participated in the program.
Limitations
Not all requesters were sent a request for feedback, and the response rate for the request for feedback was only 27%. As the program has evolved, it began sending a request for feedback immediately after answering each question, which may increase the odds of response. The goal of experts answering questions within 7 business days was met 72% of the time, likely an artifact of experts integrating question answering with many other duties. The mailbox coordinator has since provided email prompts to experts immediately upon being assigned a question with the goal of improving timeliness. The program did not include chart reviews or patient consultations, as neither was feasible for volunteer experts. The email consultation service is a single component of virtual consultative specialty geriatric mental health services within the VHA, including video consultations via NEXCSS-MH and regional geriatric mental health teams.
Conclusions
The need for specialty geriatric mental health services is increasing in the VHA and across the US. However, there are too few board-certified geriatric psychiatrists to provide direct patient care to all older adults with cognitive and mental health disorders. The VHA has leveraged telehealth to improve access to geriatric mental health care. The VHA Ask the Expert Geriatric Psychiatry email consultation program is a low-resource service which provides rapid feedback to clinicians nationwide on challenging clinical scenarios, many of which are dementia-related. Most users of the service who responded to requests for feedback reported that answers to their questions were helpful and timely. The email consultation program should continue to be supplemented by more comprehensive geriatric telemental health services for particularly complex cases to meet the needs of older veterans.
T he US Census Bureau projects that the number of older adults (aged ≥ 65 years) will exceed 49 million by 2030, and an estimated 20% (nearly 10 million) of this population will experience cognitive or mental health disorders.1,2 The mental health workforce is not equipped to address the specialized mental health care needs of many older adults.2,3 For example, geriatric psychiatrists specialize in the diagnosis and treatment of mental illness and cognitive disorders in the later stages of life, but their numbers are few and declining. Only 33.5% of geriatric psychiatry fellowship training slots were filled from 2017 to 2021, and only 62 fellows trained during the 2021-2022 academic year.4 Board-certified geriatric psychiatrists also tend to be concentrated in larger, urban, academically-affiliated medical centers, often leaving rural areas and smaller facilities without access, including facilities in the Veterans Health Administration (VHA).5
The VHA has been optimizing access to specialty geriatric mental health services via regional and national virtual consultation services. Seven of 19 Veterans Integrated Service Network (VISN) Clinical Resource Hubs (CRHs) have geriatric mental health teams.6 These provide interdisciplinary geriatric mental telehealth services, including geriatric psychiatry, for older veterans with complex care needs.7,8 Likewise, the VHA National Expert Consultation & Specialized Services-Mental Health (NEXCSS- MH, formerly known as the National Telemental Health Center) sponsors video teleconsultations with board-certified geriatric psychiatrists and an Ask the Expert email consultation program.
This article describes the Ask the Expert Geriatric Psychiatry email program (one of several similar programs at NEXCSS-MH), building upon a symposium presented at the American Association for Geriatric Psychiatry (AAGP) annual meeting in March 2022.9 The program was initiated in June 2021 as a result of discussions between the National Mental Health Director, Geriatric Mental Health in the VHA Office of Mental Health and Suicide Prevention (now known as the Office of Mental Health [OMH]), and National Telemental Health Center leadership. VHA board certified geriatric psychiatrists were recruited to serve as expert consultants and respond to email questions submitted by VHA clinicians regarding the psychiatric care of older adult veterans. The results of this program identify educational needs among clinical staff and may inform the development of program materials for a range of clinicians.
Program Description
The national geriatric mental health director recruited prospective experts and met with each to assess interest and qualifications, consulting with OMH psychiatrist leaders before making selections. Five experts were initially selected; 1 later stepped down and was replaced by another, who also stepped down. The experts were board certified in psychiatry and geriatric psychiatry and held a variety of local and national leadership positions, including geriatric psychiatry fellowship director, US Department of Veterans Affairs (VA) research and clinical leader, and various roles in the AAGP; some had received teaching awards.
Operations
The national geriatric mental health director announced the program in June 2021 to VHA mental health and geriatric program email groups with reminders sent every few months. The announcement included information about the types of questions appropriate to submit, including examples of general clinical management questions that did not share patient-specific protected health information, and clarified that experts would not be conducting chart reviews because the time required for detailed chart reviews was not feasible for volunteer experts to integrate into their otherwise full-time jobs at their respective VA medical centers. The announcement also included brief biographies of the experts.
The Figure describes the daily operations of the Ask the Expert Geriatric Psychiatry email consultation program. The NEXCSS- MH developed a Microsoft Outlook mailbox and group email address where clinicians from across the VHA could submit questions. The experts, as well as the national geriatric mental health director and NEXCSS-MH staff, had access to this mailbox to track and/or respond to questions. One expert volunteered to be the program’s primary mailbox coordinator. The coordinator checked the inbox daily and assigned each question to one of the experts on a rotating basis using the color-coding feature in Outlook. The other experts were advised to check the email account at least once weekly and reply to any assigned questions.

Responding to a question entailed first determining whether the question was appropriate for the service. For example, if a question requested a chart review, the expert replied that experts could not provide chart reviews and requested that the question be reframed. Next, the expert often needed to define a specific clinical question from the information provided, as email questions often touched upon several topics. The expert provided personalized advice on diagnostic testing, nonpharmacologic treatment strategies, and/or pharmacologic treatment options. Experts also often attached relevant guidelines or review articles. The goal was to provide a response within 7 business days.
All email responses included a disclaimer indicating that the program was not intended for urgent or immediate medical advice and that the information provided was for VHA clinician education purposes only. The disclaimer explained that email communication did not establish a doctor-patient relationship between the expert and a specific veteran and that, if desired, a request for a clinical consultation could be submitted on a specific case (ie, a video teleconsultation).
Methods for Reviewing Questions
Descriptive statistics, including frequencies, means, and minimum and maximum ranges, were used to capture the number of questions the program received, type of requester, and length of time prior to response for emailed questions.9 Conventional content analysis procedures were used between January and October 2024 to analyze clinicians’ questions.10 Four subject matter experts (3 geriatric psychiatrists and 1 geropsychologist) served as coders, assigned in groups of 2 to review questions. Each coder independently reviewed assigned questions and identified preliminary themes. Themes were reviewed and revised using an iterative process during regular team meetings with coders to clarify and confirm interpretations. Discrepancies were discussed within team meetings to achieve consensus.
Questions received. Between February 2022 and December 2023, the program received 101 email questions. Requesters included 39 physicians, 17 nurse practitioners or physician assistants, 15 social workers, 14 psychologists, 9 nurses, 5 pharmacists, 1 dietitian, and 1 who was undetermined. Experts responded to the questions an average of 6 days after receipt (range, < 1-19); 73 responses (72%) met the 7-day goal.
Iterative changes to coded themes were made during group discussions. Multiple clinical questions were often posed within the same email. Initially, some coders identified themes solely based on reported symptoms; others identified themes based on reported and/or potential diagnostic conditions attributed to the symptom(s) described within the email. For example, some coders selected a primary theme of behavioral and psychological symptoms of dementia (BPSD) only if a behavior contributing to distress in the veteran or others was described, while others selected this theme when any psychiatric symptom (eg, psychosis) was present in the context of dementia. The group identified 1 primary theme per question based on the main clinical symptom or main concern presented. Co-occurring diagnostic conditions highlighted in the email requests were included as secondary themes, and each question could have > 1 secondary theme.
The most frequent requests related to clinical symptoms included questions about agitated behaviors, sleep and/or nightmares, and depression symptoms (Table 1). Twenty-seven of 33 email requests on agitated behaviors were related to a dementia diagnosis, as were several questions about sleep/nightmares, depression, psychosis/mania, and anxiety. Many diagnostic conditions were described in the email requests (Table 2). The most frequent condition was dementia, followed by a medical condition, depressive disorder, posttraumatic stress disorder, and/or serious mental illness.


Request for Feedback. In February 2022, an email request was sent to the 64 clinicians who asked email questions from the start of the program in June 2021 through December 2021. A second request included 11 clinicians who asked questions from January through February 2022. These requests were sent as part of preparations for the symposium on the program presented at the AAGP annual meeting in March 2022.9 In May 2024, feedback was requested from 37 clinicians who submitted questions from May 1, 2023, through May 15, 2024.
Requests for feedback included 6 closed-ended and 1 open-ended question: (1) Did the answer you received help inform clinical practice? (2) Did you receive a timely response? (3) What type of information was useful to you in addressing your question (ie, direct/specific answer to a clinical scenario, guidelines, articles, VA resources)? (4) Do you have access to a geriatric psychiatrist at your facility? (5) Are you likely to use Ask the Expert Geriatric Psychiatry in the future? (6) Would you use a geriatric psychiatry teleconsultation service? (7) Share suggestions for improvement. Frequencies of response selection were obtained for each question. Text responses to the open-ended question asking for suggestions for improvement were reviewed and summarized.
Responses
Thirty users responded to the feedback request (27% response rate). Respondents considered the answers received extremely (n = 14; 47%) or very much (n = 12; 40%) helpful for their clinical practice. Twenty-three respondents (77%) felt an answer was provided promptly, 7 respondents (23%) felt the answer was not timely but still useful, and none felt that the answer was too late. Respondents reported that the most useful type of information in addressing their questions was a direct/specific answer to a clinical scenario (n = 27; 90%), followed by guidelines (n = 12; 40%), articles (n = 7; 23%), and VA resources (n = 4; 13%).
Sixteen respondents (53%) reported that they rarely had ready access to a geriatric psychiatrist at their facility, 3 (10%) had access sometimes, 4 (14%) had access usually, 3 (10%) had access regularly, and 3 (10%) never had access. Twenty-seven respondents (90%) indicated they would be very likely to use the service again. If geriatric psychiatry teleconsultation and/or e-consultation were offered, many respondents indicated they would be extremely (n = 10; 33%) or very (n = 12; 40%) likely to use teleconsultation and/or e-consultation.
Suggestions for improvement included supporting experts to perform chart reviews for email questions, developing a template or consult form, holding a biweekly drop-in meeting to present questions to and discuss cases with a panel of experts, and providing further help addressing complex decisional capacity issues, delirium, and care or placement for veterans with severe behavioral issues in a rural setting.
Discussion
Although many older adults experience cognitive and mental health disorders that may benefit from management by a geriatric psychiatrist, the number of trained geriatric psychiatrists available is insufficient to allow for direct care for each patient. The Ask the Expert Geriatric Psychiatry email consultation program is one aspect of a multicomponent strategy within the VHA to increase access to specialty geriatric mental health services for veterans. A key advantage of the program is that it is not resource intensive. Experts can participate voluntarily, providing timely feedback to clinicians around the country while continuing other duties at their respective VA medical centers. Email replies to the experts’ answers elicited positive feedback on the program, include: “I found this service to be extremely helpful and I have shared the information they sent me with several other coworkers!”, “It was great!”, and “I endorsed the service to our VISN Rehabilitation and Extended Care group.”
The coding of primary and secondary themes from 101 email questions that were retained revealed the range and relative frequencies of clinical and administrative topics with which clinicians needed help. The most common (33%) theme was agitated behaviors. Nearly half of the questions (48%) were related to underlying dementia, and 29% were related to a patient’s medical comorbidities. These findings suggest that the expertise of a geriatric psychiatrist is particularly relevant when caring for older patients experiencing BPSD or patients with complex, overlapping psychiatric and medical conditions.
Despite a 27% response rate, participant feedback has been helpful. The program reached its intended audience of clinicians in rural areas and at smaller facilities with 53% of requesters reporting they rarely had access to a geriatric psychiatrist. Suggestions for improvement indicated that some clinicians desired additional support, including chart reviews, meetings with experts, and a video teleconsultation service (available through NEXCSS-MH).
Many clinicians without training in specialty geriatric mental health may require help with complex clinical presentations. For example, 39 clinicians who submitted questions to the program were physicians. Accreditation Council for Graduate Medical Education program requirements for general psychiatry residency include 4 weeks of geriatric psychiatry.11 The findings of this study suggest that this level of training may not be adequate to independently care for every patient who experiences dementia or multimorbidity. Several training and mentoring initiatives have been developed to address the professional development need for psychiatrists.12-14
The need for geriatric workforce development is significant across health care, including other mental health professions.15,16 The VHA Geriatric Scholars program trains rural primary care practitioners, psychologists, and psychiatrists.17,18 Likewise, consultative geriatric specialty support for primary care practitioners in rural areas is provided via the Geriatric Research Education and Clinical Center Connect program.19 The Ask the Expert Geriatric Psychiatry email program is an additional economical model to support clinician educational development and provide rapid educational responses to inform patient care.
Ask the Expert received fewer email questions than anticipated. Enhanced optimization may require more frequent and widespread announcements about the program. Clinical staff may not be aware of the program due to an overload of email communications. Likewise, it may be challenging for busy clinicians to take the time to seek consultation or recognize a potential gap in their knowledge or skills. Had more questions been submitted, the 5 volunteer experts may have had more difficulty addressing the demand. Feedback from this project may inform development of a frequently asked questions document to share with VHA teams and a drop-in office hour to pose clinical questions of geriatric psychiatry experts, as recommended by a clinician who participated in the program.
Limitations
Not all requesters were sent a request for feedback, and the response rate for the request for feedback was only 27%. As the program has evolved, it began sending a request for feedback immediately after answering each question, which may increase the odds of response. The goal of experts answering questions within 7 business days was met 72% of the time, likely an artifact of experts integrating question answering with many other duties. The mailbox coordinator has since provided email prompts to experts immediately upon being assigned a question with the goal of improving timeliness. The program did not include chart reviews or patient consultations, as neither was feasible for volunteer experts. The email consultation service is a single component of virtual consultative specialty geriatric mental health services within the VHA, including video consultations via NEXCSS-MH and regional geriatric mental health teams.
Conclusions
The need for specialty geriatric mental health services is increasing in the VHA and across the US. However, there are too few board-certified geriatric psychiatrists to provide direct patient care to all older adults with cognitive and mental health disorders. The VHA has leveraged telehealth to improve access to geriatric mental health care. The VHA Ask the Expert Geriatric Psychiatry email consultation program is a low-resource service which provides rapid feedback to clinicians nationwide on challenging clinical scenarios, many of which are dementia-related. Most users of the service who responded to requests for feedback reported that answers to their questions were helpful and timely. The email consultation program should continue to be supplemented by more comprehensive geriatric telemental health services for particularly complex cases to meet the needs of older veterans.
- 2023 population projections for the nation by age, sex, race, Hispanic origin and nativity. United States Census Bureau. November 9, 2023. Accessed December 11, 2025. https://www.census.gov/newsroom/press-kits/2023/population-projections.html
- National Academies of Sciences Engineering and Medicine. Addressing the rising mental health needs of an aging population: proceedings of a workshop. 2024. Accessed December 11, 2025. doi.org:10.17226/27340
- Institute of Medicine. The mental health and substance use workforce for older adults: in whose hands? The National Academies Press; 2012. Accessed December 11, 2025. doi:10.17226/13400
- American Psychiatric Association. 2022 resident/fellow census. November 2023. Accessed December 11, 2025. https://www.psychiatry.org/getmedia/d80438af-f760-40f3-9d33-f91309b09564/APA-Resident-Census-2022.pdf
- Juul D, Colenda CC, Lyness JM, et al. Subspecialty training and certification in geriatric psychiatry: a 25-year overview. Am J Geriatr Psychiatry. 2017;25:445-453. doi:10.1016/j.jagp.2016.12.018
- Jaske E, Wheat CL, Rubenstein LV, et al. Understanding how contingency staffing programs can support mental health services in the Veterans Health Administration. Telemed J E Health. 2024;30:1857-1865. doi:10.1089/tmj.2023.0573
- Gould CE, Carlson C, Alfaro AJ, et al. Supporting veterans, caregivers, and providers in rural regions with tele-geriatric psychiatry consultation: a mixed methods pilot study. Am J Geriatr Psychiatry. 2023;31:279-290. doi:10.1016/j.jagp.2023.01.005
- Gould CE, Paiko L, Carlson C, et al. Implementation of tele-geriatricmental healthcare for rural veterans: factors influencing care models. Front Health Serv. 2024;4:1221899. doi:10.3389/frhs.2024.1221899
- Padala P, Schultz S, Khatkhate G, et al. Ask the expert geriatric psychiatry: VA program to support clinicians. Am J Geriatr Psychiatry. 2022;30:S18. doi:10.1016/j.jagp.2022.01.279
- Hsieh HF, Shannon SE. Three approaches to qualitative content analysis. Qual Health Res. 2005;15:1277-1288. doi:10.1177/1049732305276687
- Accreditation Council for Graduate Medical Education. Program requirements for graduate medical education in psychiatry. Revised September 3, 2025. Accessed December 11, 2025. https://www.acgme.org/globalassets/pfassets/programrequirements/2025-reformatted-requirements/400_psychiatry_2025_reformatted.pdf
- Fernandez J, Agarwal KS, Amspoker AB, et al. Outcomes from an interprofessional, dementia-focused, telementoring program: a brief report. Gerontol Geriatr Educ. 2024;45:601-606. doi:10.1080/02701960.2023.2253175
- Conroy ML, Garcia-Pittman EC, van Dyck LI, et al. The COVID-19 American Association for Geriatric Psychiatry (AAGP) online trainee curriculum: program evaluation and future directions. Am J Geriatr Psychiatry. 2025;33:308-314. doi:10.1016/j.jagp.2024.10.010
- Conroy ML, Garcia-Pittman EC, Ali H, et al. The COVID-19 AAGP online trainee curriculum: development and method of initial evaluation. Am J Geriatr Psychiatry. 2020;28:1004-1008. doi:10.1016/j.jagp.2020.06.003
- Flaherty E, Busby-Whitehead J, Potter J, et al. The geriatric workforce enhancement program: review of the coordinating center and examples of the GWEP in practice. Am J Geriatr Psychiatry. 2019;27:675-686. doi:10.1016/j.jagp.2019.04.010
- Hoge MA, Karel MJ, Zeiss AM, et al. Strengthening psychology’s workforce for older adults: implications of the Institute of Medicine’s report to Congress. Am Psychol. 2015;70:265-278. doi:10.1037/a0038927
- Kramer BJ, Creekmur B, Howe JL, et al. Veterans Affairs geriatric scholars program: enhancing existing primary care clinician skills in caring for older veterans. J Am Geriatr Soc. 2016;64:2343-2348. doi:10.1111/jgs.14382
- Gould CE, Rodriguez RL, Gregg JJ, et al. Preparing Veterans Health Administration psychologists to meet the complex needs of aging veterans. Fed Pract. 2024;41:S10-S15. doi:10.12788/fp.0466
- Pimentel CB, Gately M, Barczi SR, et al. GRECC Connect: Geriatrics telehealth to empower health care providers and improve management of older veterans in rural communities. Fed Pract. 2019;36:464-470.
- 2023 population projections for the nation by age, sex, race, Hispanic origin and nativity. United States Census Bureau. November 9, 2023. Accessed December 11, 2025. https://www.census.gov/newsroom/press-kits/2023/population-projections.html
- National Academies of Sciences Engineering and Medicine. Addressing the rising mental health needs of an aging population: proceedings of a workshop. 2024. Accessed December 11, 2025. doi.org:10.17226/27340
- Institute of Medicine. The mental health and substance use workforce for older adults: in whose hands? The National Academies Press; 2012. Accessed December 11, 2025. doi:10.17226/13400
- American Psychiatric Association. 2022 resident/fellow census. November 2023. Accessed December 11, 2025. https://www.psychiatry.org/getmedia/d80438af-f760-40f3-9d33-f91309b09564/APA-Resident-Census-2022.pdf
- Juul D, Colenda CC, Lyness JM, et al. Subspecialty training and certification in geriatric psychiatry: a 25-year overview. Am J Geriatr Psychiatry. 2017;25:445-453. doi:10.1016/j.jagp.2016.12.018
- Jaske E, Wheat CL, Rubenstein LV, et al. Understanding how contingency staffing programs can support mental health services in the Veterans Health Administration. Telemed J E Health. 2024;30:1857-1865. doi:10.1089/tmj.2023.0573
- Gould CE, Carlson C, Alfaro AJ, et al. Supporting veterans, caregivers, and providers in rural regions with tele-geriatric psychiatry consultation: a mixed methods pilot study. Am J Geriatr Psychiatry. 2023;31:279-290. doi:10.1016/j.jagp.2023.01.005
- Gould CE, Paiko L, Carlson C, et al. Implementation of tele-geriatricmental healthcare for rural veterans: factors influencing care models. Front Health Serv. 2024;4:1221899. doi:10.3389/frhs.2024.1221899
- Padala P, Schultz S, Khatkhate G, et al. Ask the expert geriatric psychiatry: VA program to support clinicians. Am J Geriatr Psychiatry. 2022;30:S18. doi:10.1016/j.jagp.2022.01.279
- Hsieh HF, Shannon SE. Three approaches to qualitative content analysis. Qual Health Res. 2005;15:1277-1288. doi:10.1177/1049732305276687
- Accreditation Council for Graduate Medical Education. Program requirements for graduate medical education in psychiatry. Revised September 3, 2025. Accessed December 11, 2025. https://www.acgme.org/globalassets/pfassets/programrequirements/2025-reformatted-requirements/400_psychiatry_2025_reformatted.pdf
- Fernandez J, Agarwal KS, Amspoker AB, et al. Outcomes from an interprofessional, dementia-focused, telementoring program: a brief report. Gerontol Geriatr Educ. 2024;45:601-606. doi:10.1080/02701960.2023.2253175
- Conroy ML, Garcia-Pittman EC, van Dyck LI, et al. The COVID-19 American Association for Geriatric Psychiatry (AAGP) online trainee curriculum: program evaluation and future directions. Am J Geriatr Psychiatry. 2025;33:308-314. doi:10.1016/j.jagp.2024.10.010
- Conroy ML, Garcia-Pittman EC, Ali H, et al. The COVID-19 AAGP online trainee curriculum: development and method of initial evaluation. Am J Geriatr Psychiatry. 2020;28:1004-1008. doi:10.1016/j.jagp.2020.06.003
- Flaherty E, Busby-Whitehead J, Potter J, et al. The geriatric workforce enhancement program: review of the coordinating center and examples of the GWEP in practice. Am J Geriatr Psychiatry. 2019;27:675-686. doi:10.1016/j.jagp.2019.04.010
- Hoge MA, Karel MJ, Zeiss AM, et al. Strengthening psychology’s workforce for older adults: implications of the Institute of Medicine’s report to Congress. Am Psychol. 2015;70:265-278. doi:10.1037/a0038927
- Kramer BJ, Creekmur B, Howe JL, et al. Veterans Affairs geriatric scholars program: enhancing existing primary care clinician skills in caring for older veterans. J Am Geriatr Soc. 2016;64:2343-2348. doi:10.1111/jgs.14382
- Gould CE, Rodriguez RL, Gregg JJ, et al. Preparing Veterans Health Administration psychologists to meet the complex needs of aging veterans. Fed Pract. 2024;41:S10-S15. doi:10.12788/fp.0466
- Pimentel CB, Gately M, Barczi SR, et al. GRECC Connect: Geriatrics telehealth to empower health care providers and improve management of older veterans in rural communities. Fed Pract. 2019;36:464-470.
Ask the Expert Geriatric Psychiatry: A VHA Email Consultation Program to Support Clinicians
Ask the Expert Geriatric Psychiatry: A VHA Email Consultation Program to Support Clinicians