User login
Chromoblastomycosis Leading to Squamous Cell Carcinoma: An Overlooked Outcome of a Neglected Tropical Disease
Chromoblastomycosis Leading to Squamous Cell Carcinoma: An Overlooked Outcome of a Neglected Tropical Disease
Chromoblastomycosis is a neglected tropical implantation mycosis caused by dematiaceous fungi that leads to substantial morbidity. This condition is diagnosed microscopically by visualizing the characteristic thick-walled, single, or multicellular clusters of pigmented fungal cells (also known as medlar bodies, muriform cells, or sclerotic bodies).1 The main causative fungi varies by geographic region, but most commonly is caused by Cladophialophora carrionii, Fonsecaea species, Phialophora verrucosa species complex, and Rhinocladiella aquaspersa.2-4 Standardized treatment guidelines have not been established, but itraconazole typically is considered first-line regardless of causative fungi.5 Terbinafine, other azoles, and topical immunomodulators, either as monotherapy or in combination, may be appropriate alternative or adjunctive options for refractory disease, although supporting data are limited.6-9
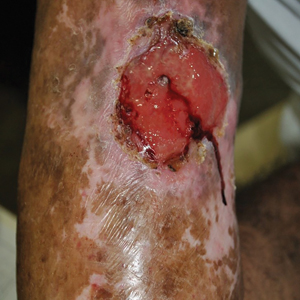
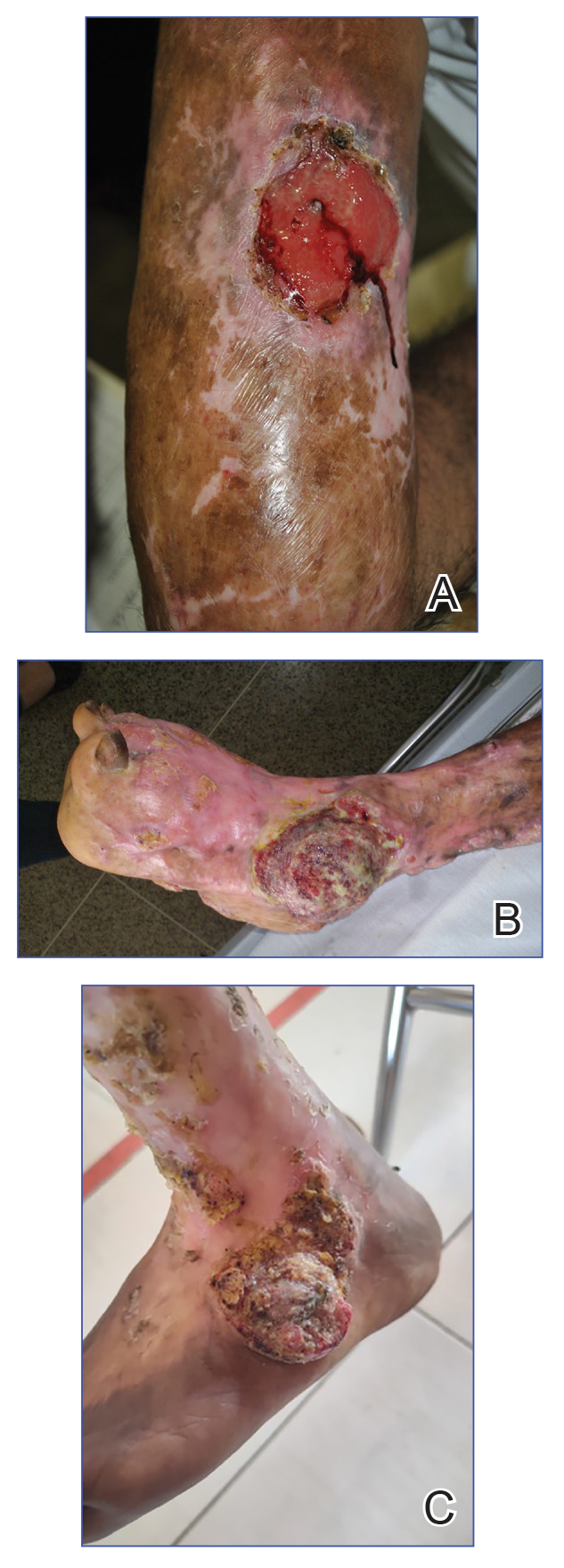
Complications from chromoblastomycosis are common, particularly in long-standing, severe, or refractory disease. An analysis using billing codes in the United States found 14% (35/255) of hospitalized patients with chromoblastomycosis had lymphedema.10 In Mexico, 63% (32/51) of patients with chromoblastomycosis developed secondary bacterial infections.11 Skin fibrosis and ankylosis also can occur and cause mobility issues and decreased quality of life. An infrequent but potentially life-threatening complication12 is the development of squamous cell carcinoma (SCC) associated with chronic lesions, representing a preventable end-stage complication of delayed diagnosis and treatment (Figure).
In this review, we summarize reported epidemiology and clinical risk factors for SCC complicating chromoblastomycosis. We also discuss plausible inflammatory mechanisms of malignant transformation and propose pragmatic clinical and public health interventions, including decentralized microscopy-based diagnosis, timely antifungal access, and biopsy-triggered surveillance of chronically inflamed lesions, to reduce preventable morbidity.
Epidemiology and Risk Factors
The epidemiology of SCC developing from chromoblastomycosis is not well understood due to gaps in national and global surveillance. Some studies have found that 2% to 13% of patients with chromoblastomycosis developed SCC.4,11,13-15 Based on case reports and case series, a symptom duration of more than 10 years appears to be the most substantial risk factor for the development of SCC rather than host immune status.16-18 Severity, specifically the size of the injury, and vegetating lesions also have been suggested as risk factors for the development of SCC.16 Additionally, the appearance of new lesions (mainly ulcers not related to secondary infection) that appear during the healing phase should raise the suspicion of SCC and warrant a biopsy for evaluation.16
Pathophysiology
The exact mechanism of malignant transformation has not been elucidated, but histopathologic features suggest substantial epidermal proliferation. In some cases, this leads to pseudoepitheliomatous hyperplasia, a nonmalignant hyperproliferative state that is an important differential HPV to leishmaniasis and lupus vulgaris.19 The chronic inflammation from long-standing chromoblastomycosis likely contributes to the further malignant transformation to SCC.
Polymorphonuclear cells and activated macrophages seen in chronic inflammation can promote the release of enzymes and free radicals that has led to malignant transformation in vitro but has not been investigated specifically in chromoblastomycosis.16 Additionally, chronic inflammation and metabolic products of phagocytosis often are accompanied by excessive production of reactive oxygen and nitrogen species, which can damage DNA, lipoproteins, and cell membranes. Other potential contributors include the expression of cyclooxygenase 2 and release of arachidonic acid metabolites (eg, prostaglandins, leukotrienes), which can damage the cell and promote carcinogenesis. It is not clear whether similar mechanisms account for the development of SCCs in other chronic skin inflammations or infections such as cutaneous tuberculosis or Marjolin ulcers.20
Clinical and Public Health Interventions
Squamous cell carcinoma arising in the setting of chromoblastomycosis warrants prompt oncologic evaluation and definitive surgical management, which may require extensive surgical excision and, in advanced disease, amputation.14,17,18 Advanced malignant tumors can be difficult to manage and can result in death.21,22 Additionally, clinicians should maintain a low threshold for biopsy in long-standing chromoblastomycosis, particularly when lesions demonstrate new ulceration, rapid growth, bleeding, pain, malodor, or failure to improve with appropriate antifungal therapy.16 Recurrent or new lesions after amputation may indicate persistent or recurrent infection and may require continued antifungal management alongside cancer care.16
Squamous cell carcinoma arising from chromoblastomycosis results after substantial diagnostic delays, allowing chronic inflammation to transform infection into malignancy. Separating benign inflammation-associated epidermal proliferation from transformation to SCC requires histopathologic skill. An assay based on increased expression of chromosome 15 open reading frame 48 (C15orf48), an immune regulatory protein, has been developed to aid in this distinction; however, it is not widely available.23
Raising awareness of chromoblastomycosis among clinicians and communities, particularly in rural areas where the disease is more common, is critical to improve health care–seeking behaviors and expedite access to care pathways.2 Furthermore, access and training on microscopy to diagnose chromoblastomycosis in decentralized areas can facilitate earlier diagnosis in primary health care settings rather than waiting for diagnosis in tertiary care settings, at which point disease usually is advanced. Global implementation of existing programs that use microscopy (eg, malaria in rural areas) can be partnered with frontline health worker cross-training on chromoblastomycosis diagnosis to improve appropriate identification of disease.24 Finally, improving access to affordable antifungals, particularly itraconazole, is necessary along with further research into novel therapeutic strategies. Approaches that utilize local manufacturing and pooled procurement could help expand treatment availability in parallel with diagnostic improvement initiatives.25
Final Thoughts
Squamous cell carcinoma resulting from chromoblastomycosis is a devastating complication, often leading to limb amputation. The true prevalence is unknown, but it occurs more commonly in long-standing disease without appropriate therapy. The appearance of new lesions or ulcers after initial improvement should increase suspicion and lead to biopsy and careful pathologic evaluation. Prevention of SCC requires increased clinical awareness, early diagnosis, and timely initiation of antifungal treatment. Enhanced surveillance among individuals with chromoblastomycosis would help to better understand its prevalence, associated risk factors, and impact on quality of life.
- Queiroz-Telles F, de Hoog S, Santos DWCL, et al. Chromoblastomycosis. Clin Microbiol Rev. 2017;30:233-276.
- Smith DJ, Queiroz-Telles F, Rabenja FR, et al. A global chromoblastomycosis strategy and development of the global chromoblastomycosis working group. PLoS Negl Trop Dis. 2024;18:E0012562.
- Yen JS, Shih IH, Chung WH, et al. Chromoblastomycosis in northern Taiwan from 2017 to 2024: unique characteristics. Clin Exp Dermatol. Published online July 18, 2025. doi:10.1093/ced/llaf329
- Santos DWCL, Vicente VA, Weiss VA, et al. Chromoblastomycosis in an endemic area of Brazil: a clinical-epidemiological analysis and a worldwide haplotype network. J Fungi. 2020;6:204.
- Smith DJ, Melhem MSC, Dirven J, et al. Establishment of epidemiological cutoff values for Fonsecaea pedrosoi, the primary etiologic agent of chromoblastomycosis, and eight antifungal medications. J Clin Microbiol. 2025;63:E01903-24.
- Logan C, Singh M, Fox N, et al. Chromoblastomycosis treated with posaconazole and adjunctive imiquimod: lending innate immunity a helping hand. Open Forum Infect Dis. 2023;10:ofad124.
- de Sousa M da GT, Belda W, Spina R, et al. Topical application of imiquimod as a treatment for chromoblastomycosis. Clin Infect Dis. 2014;58:1734-1737.
- Criado PR, Careta MF, Valente NYS, et al. Extensive long-standing chromomycosis due to Fonsecaea pedrosoi: three cases with relevant improvement under voriconazole therapy. J Dermatol Treat. 2011;22:167-174.
- Esterre P, Inzan CK, Ramarcel ER, et al. Treatment of chromomycosis with terbinafine: preliminary results of an open pilot study. Br J Dermatol. 1996;134:33-36.
- Smith DJ, Benedict K, Lockhart SR, et al. Chromoblastomycosis and phaeohyphomycotic abscess-associated hospitalizations, United States, 2016–2021. PLoS Negl Trop Dis. 2025;19:E0013499.
- Bonifaz A, Carrasco‐Gerard E, Saúl A. Chromoblastomycosis: clinical and mycologic experience of 51 cases. Mycoses. 2001;44:1-7.
- Torres E, Beristain JG, Lievanos Z, et al. Chromoblastomycosis associated with a lethal squamous cell carcinoma. An Bras Dermatol. 2010;85:267-270.
- Verma S, Thakur BK, Raphael V, et al. Epidemiology of subcutaneous mycoses in northeast India: a retrospective study. Indian J Dermatol. 2018;63:496-501.
- Siregar GO, Harianja M, Rinonce HT, et al. Chromoblastomycosis: a case series from Sumba, eastern Indonesia. Clin Exp Dermatol. 2025;50:1447-1450.
- Valentin J, Grotta G, Muller T, et al. Chromoblastomycosis in French Guiana: epidemiology and practices, 1955-2023. J Fungi. 2024;10:168.
- Azevedo CMPS, Marques SG, Santos DWCL, et al. Squamous cell carcinoma derived from chronic chromoblastomycosis in Brazil. Clin Infect Dis. 2015;60:1500-1504.
- Belda Jr W, Criado PR, Casteleti P, et al. Chromoblastomycosis evolving to sarcomatoid squamous cell carcinoma: a case report. Dermatol Rep. 2021;13:9009.
- Jamil A, Lee YY, Thevarajah S. Invasive squamous cell carcinoma arising from chromoblastomycosis. Med Mycol. 2012;50:99-102.
- Delahaye T, Orduz-Robledo M, Beltran A M, et al. Pseudo-epitheliomatous hyperplasia and skin infections. Open Dermatol J. 2024;18:E18743722304513.
- Fania L, Didona D, Di Pietro FR, et al. Cutaneous squamous cell carcinoma: from pathophysiology to novel therapeutic approaches. Biomedicines. 2021;9:171.
- Torres E, Beristain JG, Lievanos Z, et al. Carcinoma epidermoide como complicação letal de lesões crônicas de cromoblastomicose. An Bras Dermatol. 2010;85:267-270.
- Rojas OC, González GM, Moreno-Treviño M, et al. Chromoblastomycosis by Cladophialophora carrionii associated with squamous cell carcinoma and review of published reports. Mycopathologia. 2015;179:153-157.
- Su A, Ra S, Li X, et al. Differentiating cutaneous squamous cell carcinoma and pseudoepitheliomatous hyperplasia by multiplex qRT-PCR. Mod Pathol. 2013;26:1433-1437.
- Siregar GO, Harianja M, Smith DJ, et al. Leveraging malaria microscopy infrastructure to diagnose common and neglected skin diseases using direct microscopy in Sumba, Indonesia. Lancet Reg Health - West Pac. 2025;64:101739.
- Smith DJ, Soebono H, Parajuli N, et al. South-East Asia regional neglected tropical disease framework: improving control of mycetoma, chromoblastomycosis, and sporotrichosis. Lancet Reg Health Southeast Asia. 2025;35:100561.
Chromoblastomycosis is a neglected tropical implantation mycosis caused by dematiaceous fungi that leads to substantial morbidity. This condition is diagnosed microscopically by visualizing the characteristic thick-walled, single, or multicellular clusters of pigmented fungal cells (also known as medlar bodies, muriform cells, or sclerotic bodies).1 The main causative fungi varies by geographic region, but most commonly is caused by Cladophialophora carrionii, Fonsecaea species, Phialophora verrucosa species complex, and Rhinocladiella aquaspersa.2-4 Standardized treatment guidelines have not been established, but itraconazole typically is considered first-line regardless of causative fungi.5 Terbinafine, other azoles, and topical immunomodulators, either as monotherapy or in combination, may be appropriate alternative or adjunctive options for refractory disease, although supporting data are limited.6-9
Complications from chromoblastomycosis are common, particularly in long-standing, severe, or refractory disease. An analysis using billing codes in the United States found 14% (35/255) of hospitalized patients with chromoblastomycosis had lymphedema.10 In Mexico, 63% (32/51) of patients with chromoblastomycosis developed secondary bacterial infections.11 Skin fibrosis and ankylosis also can occur and cause mobility issues and decreased quality of life. An infrequent but potentially life-threatening complication12 is the development of squamous cell carcinoma (SCC) associated with chronic lesions, representing a preventable end-stage complication of delayed diagnosis and treatment (Figure).

In this review, we summarize reported epidemiology and clinical risk factors for SCC complicating chromoblastomycosis. We also discuss plausible inflammatory mechanisms of malignant transformation and propose pragmatic clinical and public health interventions, including decentralized microscopy-based diagnosis, timely antifungal access, and biopsy-triggered surveillance of chronically inflamed lesions, to reduce preventable morbidity.
Epidemiology and Risk Factors
The epidemiology of SCC developing from chromoblastomycosis is not well understood due to gaps in national and global surveillance. Some studies have found that 2% to 13% of patients with chromoblastomycosis developed SCC.4,11,13-15 Based on case reports and case series, a symptom duration of more than 10 years appears to be the most substantial risk factor for the development of SCC rather than host immune status.16-18 Severity, specifically the size of the injury, and vegetating lesions also have been suggested as risk factors for the development of SCC.16 Additionally, the appearance of new lesions (mainly ulcers not related to secondary infection) that appear during the healing phase should raise the suspicion of SCC and warrant a biopsy for evaluation.16
Pathophysiology
The exact mechanism of malignant transformation has not been elucidated, but histopathologic features suggest substantial epidermal proliferation. In some cases, this leads to pseudoepitheliomatous hyperplasia, a nonmalignant hyperproliferative state that is an important differential HPV to leishmaniasis and lupus vulgaris.19 The chronic inflammation from long-standing chromoblastomycosis likely contributes to the further malignant transformation to SCC.
Polymorphonuclear cells and activated macrophages seen in chronic inflammation can promote the release of enzymes and free radicals that has led to malignant transformation in vitro but has not been investigated specifically in chromoblastomycosis.16 Additionally, chronic inflammation and metabolic products of phagocytosis often are accompanied by excessive production of reactive oxygen and nitrogen species, which can damage DNA, lipoproteins, and cell membranes. Other potential contributors include the expression of cyclooxygenase 2 and release of arachidonic acid metabolites (eg, prostaglandins, leukotrienes), which can damage the cell and promote carcinogenesis. It is not clear whether similar mechanisms account for the development of SCCs in other chronic skin inflammations or infections such as cutaneous tuberculosis or Marjolin ulcers.20
Clinical and Public Health Interventions
Squamous cell carcinoma arising in the setting of chromoblastomycosis warrants prompt oncologic evaluation and definitive surgical management, which may require extensive surgical excision and, in advanced disease, amputation.14,17,18 Advanced malignant tumors can be difficult to manage and can result in death.21,22 Additionally, clinicians should maintain a low threshold for biopsy in long-standing chromoblastomycosis, particularly when lesions demonstrate new ulceration, rapid growth, bleeding, pain, malodor, or failure to improve with appropriate antifungal therapy.16 Recurrent or new lesions after amputation may indicate persistent or recurrent infection and may require continued antifungal management alongside cancer care.16
Squamous cell carcinoma arising from chromoblastomycosis results after substantial diagnostic delays, allowing chronic inflammation to transform infection into malignancy. Separating benign inflammation-associated epidermal proliferation from transformation to SCC requires histopathologic skill. An assay based on increased expression of chromosome 15 open reading frame 48 (C15orf48), an immune regulatory protein, has been developed to aid in this distinction; however, it is not widely available.23
Raising awareness of chromoblastomycosis among clinicians and communities, particularly in rural areas where the disease is more common, is critical to improve health care–seeking behaviors and expedite access to care pathways.2 Furthermore, access and training on microscopy to diagnose chromoblastomycosis in decentralized areas can facilitate earlier diagnosis in primary health care settings rather than waiting for diagnosis in tertiary care settings, at which point disease usually is advanced. Global implementation of existing programs that use microscopy (eg, malaria in rural areas) can be partnered with frontline health worker cross-training on chromoblastomycosis diagnosis to improve appropriate identification of disease.24 Finally, improving access to affordable antifungals, particularly itraconazole, is necessary along with further research into novel therapeutic strategies. Approaches that utilize local manufacturing and pooled procurement could help expand treatment availability in parallel with diagnostic improvement initiatives.25
Final Thoughts
Squamous cell carcinoma resulting from chromoblastomycosis is a devastating complication, often leading to limb amputation. The true prevalence is unknown, but it occurs more commonly in long-standing disease without appropriate therapy. The appearance of new lesions or ulcers after initial improvement should increase suspicion and lead to biopsy and careful pathologic evaluation. Prevention of SCC requires increased clinical awareness, early diagnosis, and timely initiation of antifungal treatment. Enhanced surveillance among individuals with chromoblastomycosis would help to better understand its prevalence, associated risk factors, and impact on quality of life.
Chromoblastomycosis is a neglected tropical implantation mycosis caused by dematiaceous fungi that leads to substantial morbidity. This condition is diagnosed microscopically by visualizing the characteristic thick-walled, single, or multicellular clusters of pigmented fungal cells (also known as medlar bodies, muriform cells, or sclerotic bodies).1 The main causative fungi varies by geographic region, but most commonly is caused by Cladophialophora carrionii, Fonsecaea species, Phialophora verrucosa species complex, and Rhinocladiella aquaspersa.2-4 Standardized treatment guidelines have not been established, but itraconazole typically is considered first-line regardless of causative fungi.5 Terbinafine, other azoles, and topical immunomodulators, either as monotherapy or in combination, may be appropriate alternative or adjunctive options for refractory disease, although supporting data are limited.6-9
Complications from chromoblastomycosis are common, particularly in long-standing, severe, or refractory disease. An analysis using billing codes in the United States found 14% (35/255) of hospitalized patients with chromoblastomycosis had lymphedema.10 In Mexico, 63% (32/51) of patients with chromoblastomycosis developed secondary bacterial infections.11 Skin fibrosis and ankylosis also can occur and cause mobility issues and decreased quality of life. An infrequent but potentially life-threatening complication12 is the development of squamous cell carcinoma (SCC) associated with chronic lesions, representing a preventable end-stage complication of delayed diagnosis and treatment (Figure).

In this review, we summarize reported epidemiology and clinical risk factors for SCC complicating chromoblastomycosis. We also discuss plausible inflammatory mechanisms of malignant transformation and propose pragmatic clinical and public health interventions, including decentralized microscopy-based diagnosis, timely antifungal access, and biopsy-triggered surveillance of chronically inflamed lesions, to reduce preventable morbidity.
Epidemiology and Risk Factors
The epidemiology of SCC developing from chromoblastomycosis is not well understood due to gaps in national and global surveillance. Some studies have found that 2% to 13% of patients with chromoblastomycosis developed SCC.4,11,13-15 Based on case reports and case series, a symptom duration of more than 10 years appears to be the most substantial risk factor for the development of SCC rather than host immune status.16-18 Severity, specifically the size of the injury, and vegetating lesions also have been suggested as risk factors for the development of SCC.16 Additionally, the appearance of new lesions (mainly ulcers not related to secondary infection) that appear during the healing phase should raise the suspicion of SCC and warrant a biopsy for evaluation.16
Pathophysiology
The exact mechanism of malignant transformation has not been elucidated, but histopathologic features suggest substantial epidermal proliferation. In some cases, this leads to pseudoepitheliomatous hyperplasia, a nonmalignant hyperproliferative state that is an important differential HPV to leishmaniasis and lupus vulgaris.19 The chronic inflammation from long-standing chromoblastomycosis likely contributes to the further malignant transformation to SCC.
Polymorphonuclear cells and activated macrophages seen in chronic inflammation can promote the release of enzymes and free radicals that has led to malignant transformation in vitro but has not been investigated specifically in chromoblastomycosis.16 Additionally, chronic inflammation and metabolic products of phagocytosis often are accompanied by excessive production of reactive oxygen and nitrogen species, which can damage DNA, lipoproteins, and cell membranes. Other potential contributors include the expression of cyclooxygenase 2 and release of arachidonic acid metabolites (eg, prostaglandins, leukotrienes), which can damage the cell and promote carcinogenesis. It is not clear whether similar mechanisms account for the development of SCCs in other chronic skin inflammations or infections such as cutaneous tuberculosis or Marjolin ulcers.20
Clinical and Public Health Interventions
Squamous cell carcinoma arising in the setting of chromoblastomycosis warrants prompt oncologic evaluation and definitive surgical management, which may require extensive surgical excision and, in advanced disease, amputation.14,17,18 Advanced malignant tumors can be difficult to manage and can result in death.21,22 Additionally, clinicians should maintain a low threshold for biopsy in long-standing chromoblastomycosis, particularly when lesions demonstrate new ulceration, rapid growth, bleeding, pain, malodor, or failure to improve with appropriate antifungal therapy.16 Recurrent or new lesions after amputation may indicate persistent or recurrent infection and may require continued antifungal management alongside cancer care.16
Squamous cell carcinoma arising from chromoblastomycosis results after substantial diagnostic delays, allowing chronic inflammation to transform infection into malignancy. Separating benign inflammation-associated epidermal proliferation from transformation to SCC requires histopathologic skill. An assay based on increased expression of chromosome 15 open reading frame 48 (C15orf48), an immune regulatory protein, has been developed to aid in this distinction; however, it is not widely available.23
Raising awareness of chromoblastomycosis among clinicians and communities, particularly in rural areas where the disease is more common, is critical to improve health care–seeking behaviors and expedite access to care pathways.2 Furthermore, access and training on microscopy to diagnose chromoblastomycosis in decentralized areas can facilitate earlier diagnosis in primary health care settings rather than waiting for diagnosis in tertiary care settings, at which point disease usually is advanced. Global implementation of existing programs that use microscopy (eg, malaria in rural areas) can be partnered with frontline health worker cross-training on chromoblastomycosis diagnosis to improve appropriate identification of disease.24 Finally, improving access to affordable antifungals, particularly itraconazole, is necessary along with further research into novel therapeutic strategies. Approaches that utilize local manufacturing and pooled procurement could help expand treatment availability in parallel with diagnostic improvement initiatives.25
Final Thoughts
Squamous cell carcinoma resulting from chromoblastomycosis is a devastating complication, often leading to limb amputation. The true prevalence is unknown, but it occurs more commonly in long-standing disease without appropriate therapy. The appearance of new lesions or ulcers after initial improvement should increase suspicion and lead to biopsy and careful pathologic evaluation. Prevention of SCC requires increased clinical awareness, early diagnosis, and timely initiation of antifungal treatment. Enhanced surveillance among individuals with chromoblastomycosis would help to better understand its prevalence, associated risk factors, and impact on quality of life.
- Queiroz-Telles F, de Hoog S, Santos DWCL, et al. Chromoblastomycosis. Clin Microbiol Rev. 2017;30:233-276.
- Smith DJ, Queiroz-Telles F, Rabenja FR, et al. A global chromoblastomycosis strategy and development of the global chromoblastomycosis working group. PLoS Negl Trop Dis. 2024;18:E0012562.
- Yen JS, Shih IH, Chung WH, et al. Chromoblastomycosis in northern Taiwan from 2017 to 2024: unique characteristics. Clin Exp Dermatol. Published online July 18, 2025. doi:10.1093/ced/llaf329
- Santos DWCL, Vicente VA, Weiss VA, et al. Chromoblastomycosis in an endemic area of Brazil: a clinical-epidemiological analysis and a worldwide haplotype network. J Fungi. 2020;6:204.
- Smith DJ, Melhem MSC, Dirven J, et al. Establishment of epidemiological cutoff values for Fonsecaea pedrosoi, the primary etiologic agent of chromoblastomycosis, and eight antifungal medications. J Clin Microbiol. 2025;63:E01903-24.
- Logan C, Singh M, Fox N, et al. Chromoblastomycosis treated with posaconazole and adjunctive imiquimod: lending innate immunity a helping hand. Open Forum Infect Dis. 2023;10:ofad124.
- de Sousa M da GT, Belda W, Spina R, et al. Topical application of imiquimod as a treatment for chromoblastomycosis. Clin Infect Dis. 2014;58:1734-1737.
- Criado PR, Careta MF, Valente NYS, et al. Extensive long-standing chromomycosis due to Fonsecaea pedrosoi: three cases with relevant improvement under voriconazole therapy. J Dermatol Treat. 2011;22:167-174.
- Esterre P, Inzan CK, Ramarcel ER, et al. Treatment of chromomycosis with terbinafine: preliminary results of an open pilot study. Br J Dermatol. 1996;134:33-36.
- Smith DJ, Benedict K, Lockhart SR, et al. Chromoblastomycosis and phaeohyphomycotic abscess-associated hospitalizations, United States, 2016–2021. PLoS Negl Trop Dis. 2025;19:E0013499.
- Bonifaz A, Carrasco‐Gerard E, Saúl A. Chromoblastomycosis: clinical and mycologic experience of 51 cases. Mycoses. 2001;44:1-7.
- Torres E, Beristain JG, Lievanos Z, et al. Chromoblastomycosis associated with a lethal squamous cell carcinoma. An Bras Dermatol. 2010;85:267-270.
- Verma S, Thakur BK, Raphael V, et al. Epidemiology of subcutaneous mycoses in northeast India: a retrospective study. Indian J Dermatol. 2018;63:496-501.
- Siregar GO, Harianja M, Rinonce HT, et al. Chromoblastomycosis: a case series from Sumba, eastern Indonesia. Clin Exp Dermatol. 2025;50:1447-1450.
- Valentin J, Grotta G, Muller T, et al. Chromoblastomycosis in French Guiana: epidemiology and practices, 1955-2023. J Fungi. 2024;10:168.
- Azevedo CMPS, Marques SG, Santos DWCL, et al. Squamous cell carcinoma derived from chronic chromoblastomycosis in Brazil. Clin Infect Dis. 2015;60:1500-1504.
- Belda Jr W, Criado PR, Casteleti P, et al. Chromoblastomycosis evolving to sarcomatoid squamous cell carcinoma: a case report. Dermatol Rep. 2021;13:9009.
- Jamil A, Lee YY, Thevarajah S. Invasive squamous cell carcinoma arising from chromoblastomycosis. Med Mycol. 2012;50:99-102.
- Delahaye T, Orduz-Robledo M, Beltran A M, et al. Pseudo-epitheliomatous hyperplasia and skin infections. Open Dermatol J. 2024;18:E18743722304513.
- Fania L, Didona D, Di Pietro FR, et al. Cutaneous squamous cell carcinoma: from pathophysiology to novel therapeutic approaches. Biomedicines. 2021;9:171.
- Torres E, Beristain JG, Lievanos Z, et al. Carcinoma epidermoide como complicação letal de lesões crônicas de cromoblastomicose. An Bras Dermatol. 2010;85:267-270.
- Rojas OC, González GM, Moreno-Treviño M, et al. Chromoblastomycosis by Cladophialophora carrionii associated with squamous cell carcinoma and review of published reports. Mycopathologia. 2015;179:153-157.
- Su A, Ra S, Li X, et al. Differentiating cutaneous squamous cell carcinoma and pseudoepitheliomatous hyperplasia by multiplex qRT-PCR. Mod Pathol. 2013;26:1433-1437.
- Siregar GO, Harianja M, Smith DJ, et al. Leveraging malaria microscopy infrastructure to diagnose common and neglected skin diseases using direct microscopy in Sumba, Indonesia. Lancet Reg Health - West Pac. 2025;64:101739.
- Smith DJ, Soebono H, Parajuli N, et al. South-East Asia regional neglected tropical disease framework: improving control of mycetoma, chromoblastomycosis, and sporotrichosis. Lancet Reg Health Southeast Asia. 2025;35:100561.
- Queiroz-Telles F, de Hoog S, Santos DWCL, et al. Chromoblastomycosis. Clin Microbiol Rev. 2017;30:233-276.
- Smith DJ, Queiroz-Telles F, Rabenja FR, et al. A global chromoblastomycosis strategy and development of the global chromoblastomycosis working group. PLoS Negl Trop Dis. 2024;18:E0012562.
- Yen JS, Shih IH, Chung WH, et al. Chromoblastomycosis in northern Taiwan from 2017 to 2024: unique characteristics. Clin Exp Dermatol. Published online July 18, 2025. doi:10.1093/ced/llaf329
- Santos DWCL, Vicente VA, Weiss VA, et al. Chromoblastomycosis in an endemic area of Brazil: a clinical-epidemiological analysis and a worldwide haplotype network. J Fungi. 2020;6:204.
- Smith DJ, Melhem MSC, Dirven J, et al. Establishment of epidemiological cutoff values for Fonsecaea pedrosoi, the primary etiologic agent of chromoblastomycosis, and eight antifungal medications. J Clin Microbiol. 2025;63:E01903-24.
- Logan C, Singh M, Fox N, et al. Chromoblastomycosis treated with posaconazole and adjunctive imiquimod: lending innate immunity a helping hand. Open Forum Infect Dis. 2023;10:ofad124.
- de Sousa M da GT, Belda W, Spina R, et al. Topical application of imiquimod as a treatment for chromoblastomycosis. Clin Infect Dis. 2014;58:1734-1737.
- Criado PR, Careta MF, Valente NYS, et al. Extensive long-standing chromomycosis due to Fonsecaea pedrosoi: three cases with relevant improvement under voriconazole therapy. J Dermatol Treat. 2011;22:167-174.
- Esterre P, Inzan CK, Ramarcel ER, et al. Treatment of chromomycosis with terbinafine: preliminary results of an open pilot study. Br J Dermatol. 1996;134:33-36.
- Smith DJ, Benedict K, Lockhart SR, et al. Chromoblastomycosis and phaeohyphomycotic abscess-associated hospitalizations, United States, 2016–2021. PLoS Negl Trop Dis. 2025;19:E0013499.
- Bonifaz A, Carrasco‐Gerard E, Saúl A. Chromoblastomycosis: clinical and mycologic experience of 51 cases. Mycoses. 2001;44:1-7.
- Torres E, Beristain JG, Lievanos Z, et al. Chromoblastomycosis associated with a lethal squamous cell carcinoma. An Bras Dermatol. 2010;85:267-270.
- Verma S, Thakur BK, Raphael V, et al. Epidemiology of subcutaneous mycoses in northeast India: a retrospective study. Indian J Dermatol. 2018;63:496-501.
- Siregar GO, Harianja M, Rinonce HT, et al. Chromoblastomycosis: a case series from Sumba, eastern Indonesia. Clin Exp Dermatol. 2025;50:1447-1450.
- Valentin J, Grotta G, Muller T, et al. Chromoblastomycosis in French Guiana: epidemiology and practices, 1955-2023. J Fungi. 2024;10:168.
- Azevedo CMPS, Marques SG, Santos DWCL, et al. Squamous cell carcinoma derived from chronic chromoblastomycosis in Brazil. Clin Infect Dis. 2015;60:1500-1504.
- Belda Jr W, Criado PR, Casteleti P, et al. Chromoblastomycosis evolving to sarcomatoid squamous cell carcinoma: a case report. Dermatol Rep. 2021;13:9009.
- Jamil A, Lee YY, Thevarajah S. Invasive squamous cell carcinoma arising from chromoblastomycosis. Med Mycol. 2012;50:99-102.
- Delahaye T, Orduz-Robledo M, Beltran A M, et al. Pseudo-epitheliomatous hyperplasia and skin infections. Open Dermatol J. 2024;18:E18743722304513.
- Fania L, Didona D, Di Pietro FR, et al. Cutaneous squamous cell carcinoma: from pathophysiology to novel therapeutic approaches. Biomedicines. 2021;9:171.
- Torres E, Beristain JG, Lievanos Z, et al. Carcinoma epidermoide como complicação letal de lesões crônicas de cromoblastomicose. An Bras Dermatol. 2010;85:267-270.
- Rojas OC, González GM, Moreno-Treviño M, et al. Chromoblastomycosis by Cladophialophora carrionii associated with squamous cell carcinoma and review of published reports. Mycopathologia. 2015;179:153-157.
- Su A, Ra S, Li X, et al. Differentiating cutaneous squamous cell carcinoma and pseudoepitheliomatous hyperplasia by multiplex qRT-PCR. Mod Pathol. 2013;26:1433-1437.
- Siregar GO, Harianja M, Smith DJ, et al. Leveraging malaria microscopy infrastructure to diagnose common and neglected skin diseases using direct microscopy in Sumba, Indonesia. Lancet Reg Health - West Pac. 2025;64:101739.
- Smith DJ, Soebono H, Parajuli N, et al. South-East Asia regional neglected tropical disease framework: improving control of mycetoma, chromoblastomycosis, and sporotrichosis. Lancet Reg Health Southeast Asia. 2025;35:100561.
Chromoblastomycosis Leading to Squamous Cell Carcinoma: An Overlooked Outcome of a Neglected Tropical Disease
Chromoblastomycosis Leading to Squamous Cell Carcinoma: An Overlooked Outcome of a Neglected Tropical Disease
Practice Points
- Chromoblastomycosis is recognized by the World Health Organization as a neglected tropical disease and principally affects agricultural workers in tropical and subtropical regions.
- Long-standing or refractory chromoblastomycosis can lead to substantial morbidity, including lymphedema, secondary bacterial infections, extensive scarring, functional impairment, and squamous cell carcinoma (SCC).
- The development of SCC is thought to be related to chronic inflammation and prolonged disease duration (Mathematical Pi LT Std>10 years). Advanced cases may require extensive surgical excision or amputation.
- Early recognition with support of direct microscopy or histopathology, timely antifungal treatment (often with itraconazole), and a low threshold for repeat biopsy of new ulceration or rapidly changing lesions may prevent malignant transformation and disability.