User login
Optimizing Care for Veterans at Risk of Cancer From Camp Lejeune Water Exposure
Optimizing Care for Veterans at Risk of Cancer From Camp Lejeune Water Exposure
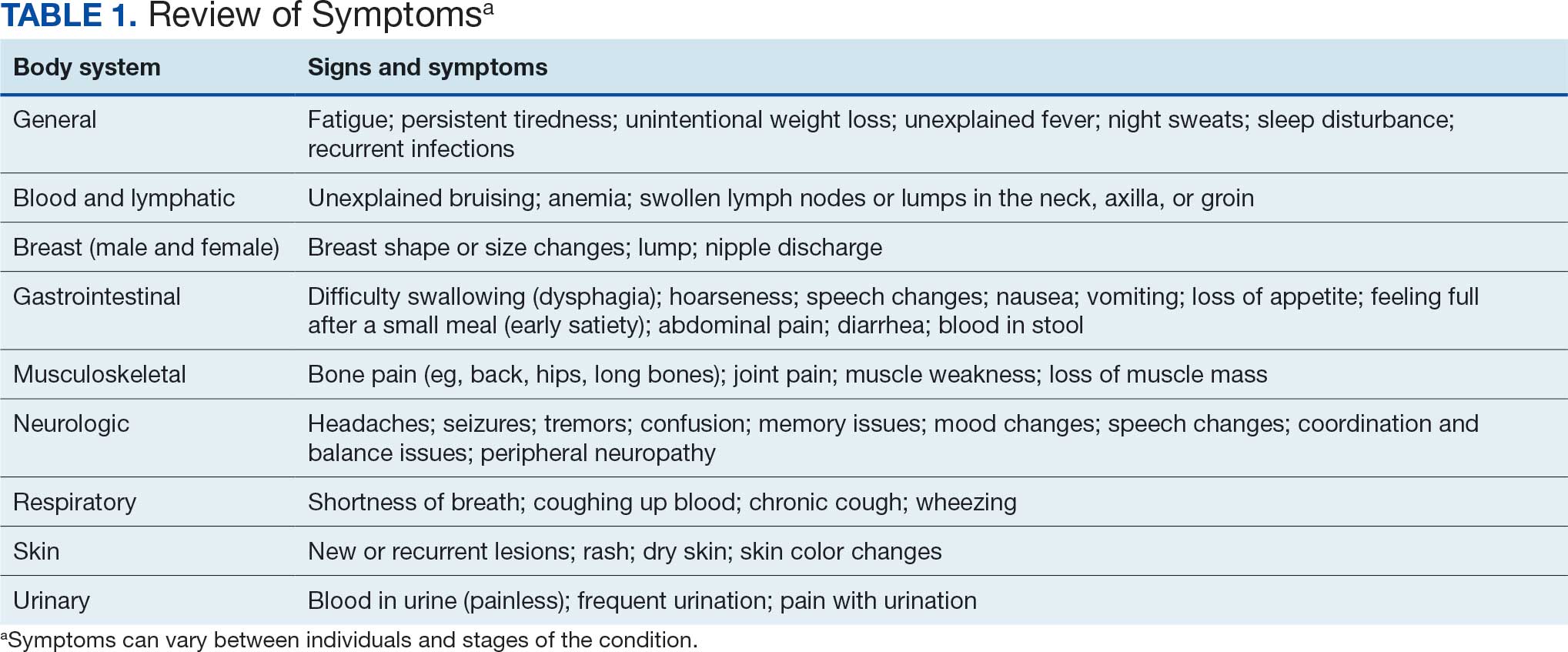
Clinical awareness of cancers associated with Camp Lejeune water contamination exposure remains limited despite legal and policy advances. Gaps persist in early symptom recognition and timely diagnostic evaluation before a definitive cancer diagnosis among exposed personnel. This may represent missed opportunities for earlier identification of volatile organic compounds (VOCs)-related cancers and for less invasive treatment options for veterans in this high-risk population.
Federal health care practitioners (HCPs), especially those in primary care and internal medicine, are uniquely positioned to bridge this gap. By improving the recognition of symptoms, pertinent physical examination findings, and implementing a diagnostic screening panel, HCPs can support accurate diagnoses and facilitate earlier treatment to improve health and quality of life for this population.
From 1953 to 1985, as many as 1 million military personnel, civilian workers, and their families stationed at US Marine Corps Base Camp Lejeune were unknowingly exposed to toxic and carcinogenic chemicals in drinking and bathing water.1 Three of the 8 main water sources on base were contaminated with VOCs, which are associated with multiple cancers.1-3
The US Department of Veterans Affairs (VA) recognizes 15 conditions associated with Camp Lejeune contaminated water exposure for VA benefits, including 10 cancers: adult leukemia; aplastic anemia and other myelodysplastic syndromes (MDS); bladder, esophageal, kidney, liver, breast (male and female), and lung cancers; multiple myeloma; and non-Hodgkin lymphoma (NHL).4
BACKGROUND
Established in 1942, Camp Lejeune is an important Marine Corps training installation. Between 1953 and 1985, multiple on-base water systems were contaminated with VOCs, including trichloroethylene (TCE), perchloroethylene (PCE), benzene, and vinyl chloride, due to improper waste disposal and industrial runoff from on- and off-base sources.5 Tarawa Terrace water treatment plant (WTP) was contaminated primarily with PCE from November 1957 to February 1987. Hadnot Point WTP was contaminated with TCE from August 1953 to December 1984, along with PCE, and benzene, toluene, ethylbenzene, and xylene (BTEX). Holcomb Boulevard WTP, established in 1972, was contaminated with TCE from June 1972 to February 1985.2 These contaminants entered the drinking and bathing water supply over decades, and exposure often occurred concurrently across = 1 VOC, compounding health risks.2,3 This prolonged 32-year VOC exposure window underlies current concerns regarding long-term cancer risk among affected service members, civilian employees, and family members. Epidemiologic research has found statistically significant associations between VOC exposure and multiple cancers, neurologic conditions, and reproductive issues.6 Specifically, TCE is associated with higher risks of hematologic cancers, multiple myeloma, NHL, and kidney cancer.3 PCE is linked with kidney cancer, benzene with multiple myeloma and NHL, and vinyl chloride with hepatobiliary cancers.3 A cohort mortality study compared Camp Lejeune personnel with a control group at Camp Pendleton from 1972 to 1985 and found a 3-fold higher incidence or mortality rate for kidney, esophageal, and female breast cancers, leukemia, and lymphoma among exposed Camp Lejeune personnel.6 Notably, personnel assigned to Camp Lejeune for as little as 6 months faced up to a 6-fold increase in cancer risk; the average military assignment between 1975 and 1985 was 18 months.3,6
Honoring America's Veterans and Caring for Camp Lejeune Families Act of 2012, the Sergeant First Class Heath Robinson Honoring Our Promise to Address Comprehensive Toxics (PACT) Act of 2022, the Camp Lejeune Justice Act of 2022, and the pending Ensuring Justice for Camp Lejeune Victims Act of 2025 provide health care and legal resources for personnel and families affected by Camp Lejeune’s contaminated water.6-8 These laws acknowledge associations between exposure and specific health conditions and expanded health care, benefits, and legal recourse for affected veterans, survivors, and their families.8,9
CANCERS LINKED TO CAMP LEJEUNE
Camp Lejeune VOC-contaminated water exposure is associated with solid tumor and hematologic cancers. Symptoms, physical examination findings, and diagnostic considerations vary by cancer type (Table 1).
Bladder Cancer
The US incidence rate of bladder cancer for both males and females is 18 per 100,000 individuals per year, with a death rate of 4.1 per 100,000 individuals per year, and a 2.1% lifetime diagnosis risk.10 Personnel exposed to VOCs at Camp Lejeune had a 9% higher risk of developing bladder cancer and a 2% increased mortality compared with an unexposed control group at Camp Pendleton.1,7 Other bladder cancer subtypes at increased risk are papillary transitional cell carcinoma, nonpapillary transition cell carcinoma, and urothelial carcinoma.7 This is consistent with prior research that found PCE exposure is associated with an increased risk for bladder cancer.3,7,11 Smoking and tobacco use remain significant risk factors for bladder cancer.12
Symptomatology. The most common symptom associated with bladder cancer is painless hematuria (gross or microscopic). Other often delayed symptoms include urinary frequency, urgency, or nocturia.13,14
Diagnostics. Screening tests include urinalysis for hematuria, urine cytology, and cystoscopy with biopsy as the gold standard for diagnosis and staging.15,16
Kidney Cancer
The US incidence rate of kidney cancer and renal pelvis cancer for both males and females is 17.5 per 100,000 individuals per year, with a death rate of 3.4 per 100,000, and a 1.8% lifetime diagnosis risk.17 Camp Lejeune personnel exposed to VOCs had a 6% increased risk of developing kidney cancer and renal pelvis cancer and a 21% higher mortality risk compared with Camp Pendleton controls.1,7 Subtypes at risk include renal cell carcinoma and papillary carcinoma.7 This is consistent with prior research that found exposures to TCE and PCE are associated with a 3-fold increased risk of kidney cancer.3,7
Symptomatology. Hematuria, flank pain, and a palpable abdominal mass are common symptoms associated with kidney cancer. In advanced stages, other symptoms may include left-sided varicocele, anemia, weight loss, fatigue, fever, and night sweats.18
Diagnostics. Screening tests include urinalysis to assess the presence of blood, complete blood count (CBC) to assess anemia, calcium (elevated), and lactate dehydrogenase (LDH), which may be elevated. Imaging strategies include abdominal computed tomography (CT), magnetic resonance imaging (MRI), or ultrasound.19
Esophageal Cancer
The US incidence rate of esophageal cancer for both males and females is 4.2 per 100,000 individuals per year, the death rate is 3.7 per 100,000 individuals per year, and a 0.5% lifetime diagnosis risk.20 VOC-exposed Camp Lejeune personnel had a 27% increased incidence and 25% increased mortality compared with the control group.1,7 Esophageal cancer subtypes at elevated risk include squamous cell carcinoma and adenocarcinoma. This is consistent with prior research that found Camp Lejeune water exposure is associated with a 3-fold increased risk for esophageal cancer.7 Additional risk factors include history of smoking and alcohol use.21
Symptomatology. Esophageal cancer is often asymptomatic with potential symptoms that include dysphagia, hoarseness, and weight loss in advanced disease.22
Diagnostics. Endoscopy with biopsy is the definitive method for diagnosis.23
Liver Cancer
The US incidence rate of liver cancer and intrahepatic bile duct cancer for both males and females is 9.4 per 100,000 individuals per year, with a death rate of 6.6 per 100,000 individuals per year, and a 1.1% lifetime diagnosis risk.24 VOC-exposed personnel had a 1% higher mortality than controls.1
Symptomatology. Liver cancer is often asymptomatic and appears in late stages.25 Common symptoms include right upper quadrant pain, early satiety, nausea, vomiting, loss of appetite, weight loss, ascites, jaundice, and abnormal bleeding or bruising.25,26
Diagnostics. Diagnostic tests may include an ultrasound, CT, or MRI. Additional laboratory testing may include liver function, a-fetoprotein blood, CBC, renal function, calcium, and hepatitis panel screening for hepatitis B and C.27,28
Lung Cancer
The US incidence rate of lung cancer for both males and females is 47.8 per 100,000 individuals per year, with a death rate of 31.5 per 100,000 individuals per year, and a 5.4% lifetime diagnosis risk.29 VOC-exposed personnel had a 16% increased risk and 19% higher mortality.1,7 Subtypes include large cell, small cell, non-small cell, squamous cell, and adenocarcinoma.7 Smoking is an additional risk factor.30
Symptomatology. Symptoms of lung cancer include cough, shortness of breath, chest pain worse with deep breathing, unexplained weight loss, fatigue, night sweats, and recurrent fevers. Advanced stages may metastasize or spread to the liver, bones, and brain.31
Diagnostics. Low-dose CT and chest X-ray are used for screening.32
Breast Cancer
The US incidence rate of female breast cancer is 130.8 per 100,000 individuals per year, with a death rate of 19.2 per 100,000 individuals per year, and a 13.0% lifetime risk of diagnosis.33 For female VOC-exposed personnel, there was an equal risk of developing breast cancer as the control group.1 However, exposed females at Camp Lejeune had a 23% higher mortality risk compared to the control group.7 Breast cancer subtypes among females include ductal carcinoma, lobular carcinoma, and ductal-lobular carcinoma.1
The US incidence rate of male breast cancer is 1.3 per 100,000 individuals per year, with a death rate of 0.3 per 100,000 individuals per year.34,35 The lifetime risk for males developing breast cancer is 137.7 per 100,000 and about 70 to 100 times less common in men than women.36
Male personnel exposed at Camp Lejeune had a 4% increased risk for developing breast cancer compared to Camp Pendleton.7 However, mortality was lower in the Camp Lejeune group.1 Although male breast cancer is rare, males at Camp Lejeune had a higher incidence, indicating a link between TCE, PCE, vinyl chloride exposures and male breast cancer.37 Male breast cancer is more often diagnosed in advanced stages than female breast cancer due to the lack of awareness or absence of routine screenings.38 The most common breast cancer type in males is invasive ductal carcinoma, accounting for 85% to 90% of cases; lobular carcinoma is the second most common type.39
Symptomatology. In both females and males, breast cancer symptoms include painless, firm mass or lump in the breast (left breast slightly more common than right), skin changes or dimpling, nipple retraction or turning inward, and nipple discharge. Breast cancer can spread to the lymph nodes and can be appreciated in axilla or clavicular regions.40
Diagnostics. The diagnostic evaluation for breast cancer is similar for females and males. It includes a clinical breast examination, diagnostic mammogram, and ultrasound.41 Mammograms can distinguish between gynecomastia and cancer, especially in males.42 A core or fine needle biopsy is needed to confirm diagnosis.41
Adult Leukemia
The US incidence rate of leukemia for both male and female was 14.4 per 100,000 individuals per year, with a death rate of 5.8 per 100,000 individuals per year, and a 1.5% lifetime diagnosis risk.43
VOC-exposed personnel had a 7% higher risk of developing leukemia and a 13% increased mortality risk compared with the control group.1,7 Subtypes of leukemia at risk included a 38% increased incidence of acute myeloid/monocytic leukemia (AML) and a 2% increased incidence of chronic lymphocytic leukemia (CLL).1 Benzene and TCE exposures are known risk factors for AML and other leukemias.7 Personnel at Camp Lejeune had 3 times the incidence or mortality for leukemia, specifically AML mortality at 20%.7 Smoking is an additional risk factor for certain leukemias, especially AML.30
Symptomatology. Symptoms associated with leukemia are often nonspecific and may include fatigue, pallor, easy bruising or bleeding (skin or gums), recurrent infections secondary to neutropenia, fever, night sweats, pain or feeling full after a small meal due to enlarged spleen or liver, and weight loss.44,45
Diagnostics. An initial screening includes a CBC with differential, a peripheral smear to detect the presence of blast cells, as well as Auer rods in myeloid blast cells in AML or smudge cells in CLL. Confirmatory tests may include bone marrow biopsy or flow cytometry. A referral to a hematologist is recommended for any suspected leukemia.46,47
Myelodysplastic Syndromes
Aplastic anemia and MDS are considered rare disorders.48 Aplastic anemia is a nonmalignant bone marrow failure disorder with pancytopenia and hypocellular bone marrow due to the loss of hematopoietic stem cells.48 MDS is a type of hematopoietic cancer where the bone marrow produces abnormal blood cells or does not make enough healthy cells.49 This can lead to an increased risk for infection, cytopenias, neutropenia, refractory anemia, and thrombocytopenia, and progression to AML in some patients.49
The reported US incidence of MDS from 1975 to 2013 was 6.7 per 100,000 for males and 3.7 per 100,000 for females.50 Benzene exposure is linked to MDS and a known cause of AML.1 VOC-exposed personnel had a 68% increased risk of developing MDS and a 2.3-fold increased mortality risk compared to controls.1,7
Symptomatology. Some patients are asymptomatic at diagnosis.51 Symptoms related to cytopenia include fatigue, pallor, purpura, petechiae, bleeding of skin, gum, or nose, recurrent infections, fever, bone pain, loss of appetite, and weight loss.50,51
Diagnostics. Initial workup includes a CBC with differential to assess for anemia, white blood cell and absolute neutrophil counts (low), and thrombocytopenia.52 A peripheral blood smear may show myeloid blast cells. A bone marrow aspiration and biopsy, flow cytometry, and cytogenetic or molecular testing may be performed. If MDS is suspected, a referral to a hematologist should be considered.52
Multiple Myeloma
The US incidence rate of multiple myeloma for both males and females is 7.3 per 100,000 individuals per year, with a mortality rate of 2.9 per 100,000 individuals per year, and a 0.8% lifetime diagnosis risk.53 VOC-exposed personnel had a 13% increased risk of developing multiple myeloma and an 8% increased mortality risk compared to unexposed personnel.1,7
Symptomatology. Multiple myeloma may be asymptomatic in early stages. The most common presenting symptom is bone pain, especially in the back, hips, and long bones, due to hypercalcemia from increased reabsorption, plasma cell tumor overgrowth in the bone marrow, and lytic lesions.54 Additional symptoms include fatigue and pallor related to anemia, leukopenia, thrombocytopenia, recurrent infections, extreme thirst, frequent urination, dehydration, confusion associated with hypercalcemia, peripheral neuropathy, loss of appetite, weight loss, and renal impairment or failure.54
Diagnostics. Testing considerations include a CBC with a peripheral blood smear to evaluate anemia and rouleaux formation of red blood cells (seen in > 50% of patients with multiple myeloma), comprehensive metabolic panel (CMP) to assess kidney function, calcium levels (elevated), serum and urine protein electrophoresis with immunofixation to detect monoclonal protein (detected in > 80% of patients with multiple myeloma) and Bence-Jones proteins, serum free light chain assay, and a bone marrow biopsy for diagnosis.55,56
MRI of the spine and pelvis is the most sensitive to detecting bone marrow involvement and focal lesions before lytic lesion progression occurs and for assessing spinal cord compression.57 PET/CT is more sensitive at detecting extramedullary disease, outside of the spine, and for patients that cannot undergo MRI.57 A whole-body low-dose CT, either alone or with PET, is more sensitive than an X-ray at detecting lytic lesions, fractures, or osteoporosis associated with multiple myeloma.57
Non-Hodgkin Lymphoma
The US incidence rate of NHL for both males and females are 18.7 per 100,000 individuals per year, the death rate is 4.9 per 100,000 individuals per year, and a 2% lifetime diagnosis risk.58 VOC-exposed personnel had a 1% higher risk of developing NHL and a decreased mortality risk compared to the control group.1,7 Specific NHL subtypes with increased risk in the exposed cohort are mantle cell (26%), follicular (7%), Burkitt (53%), and marginal zone B-cell (45%).7
Symptomatology. NHL often presents with painless lymphadenopathy or enlarged lymph nodes involving the cervical, axillary, inguinal regions.59,60 Other symptoms include frequent infections, unexplained bruising, weight loss, and “B symptoms,” such as fever and night sweats.59,60 Some patients develop a mediastinal mass in the thorax, which if large may lead to cough or shortness of breath.59
Diagnostics. The initial diagnostic workup includes CBC with differential and LDH, which may be elevated.60,61 Imaging may begin with a chest X-ray to assess for a mediastinal mass; however, CTs of the chest, abdomen, and pelvis provide more detail to better assess for NHL. Whole body PET/CT is considered the gold standard for assessing and staging systemic involvement. If enlarged lymph nodes are present, a biopsy can confirm the subtype of NHL.60,61
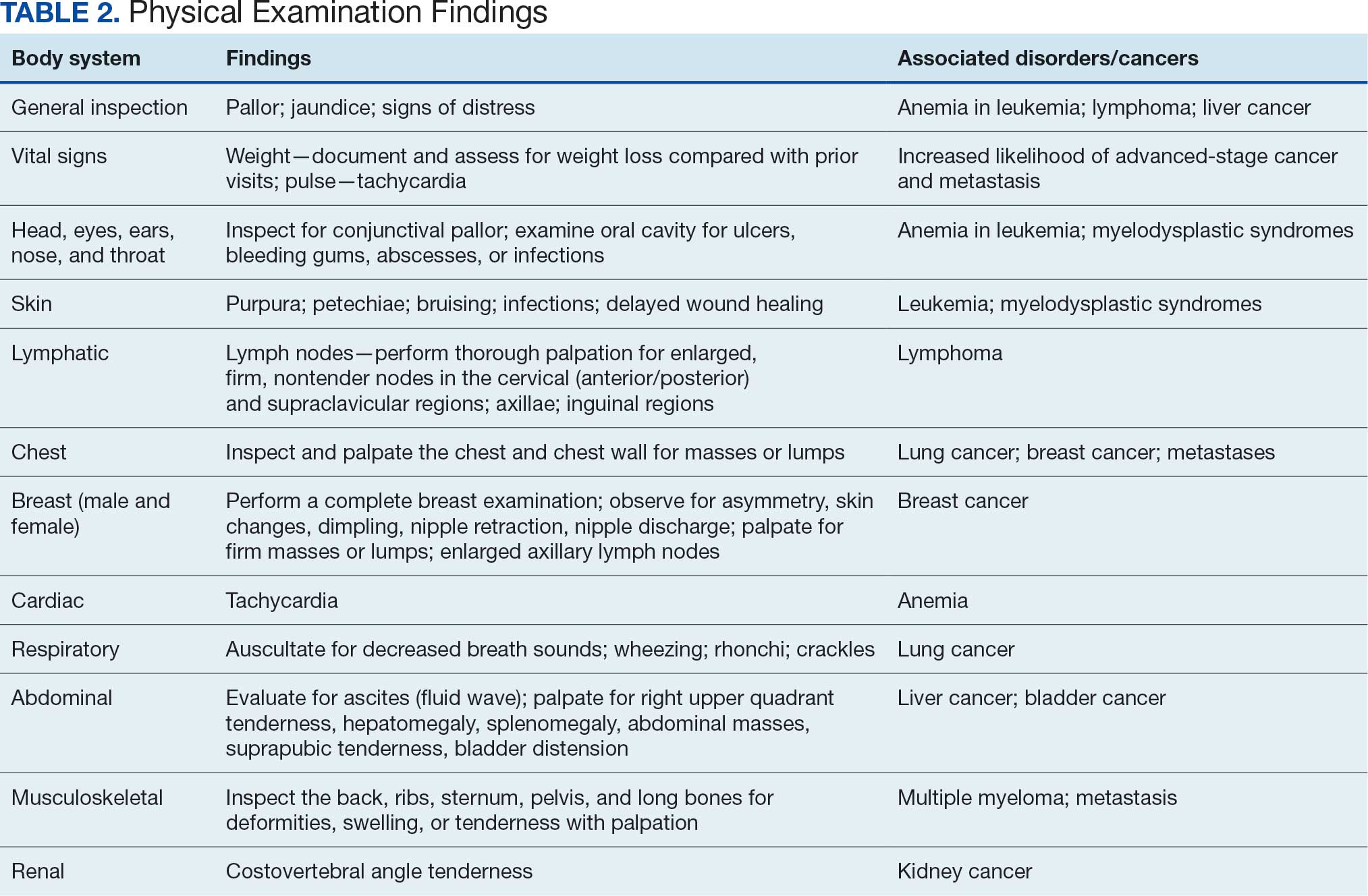
PHYSICAL EXAMINATION
A focused physical examination may aid HCPs in early detection of the cancers associated with Camp Lejeune (Table 2). The physical examination can guide diagnostic testing and imaging for further assessment and workup for VOC-related cancers.
Proposed Diagnostic Screening Panel
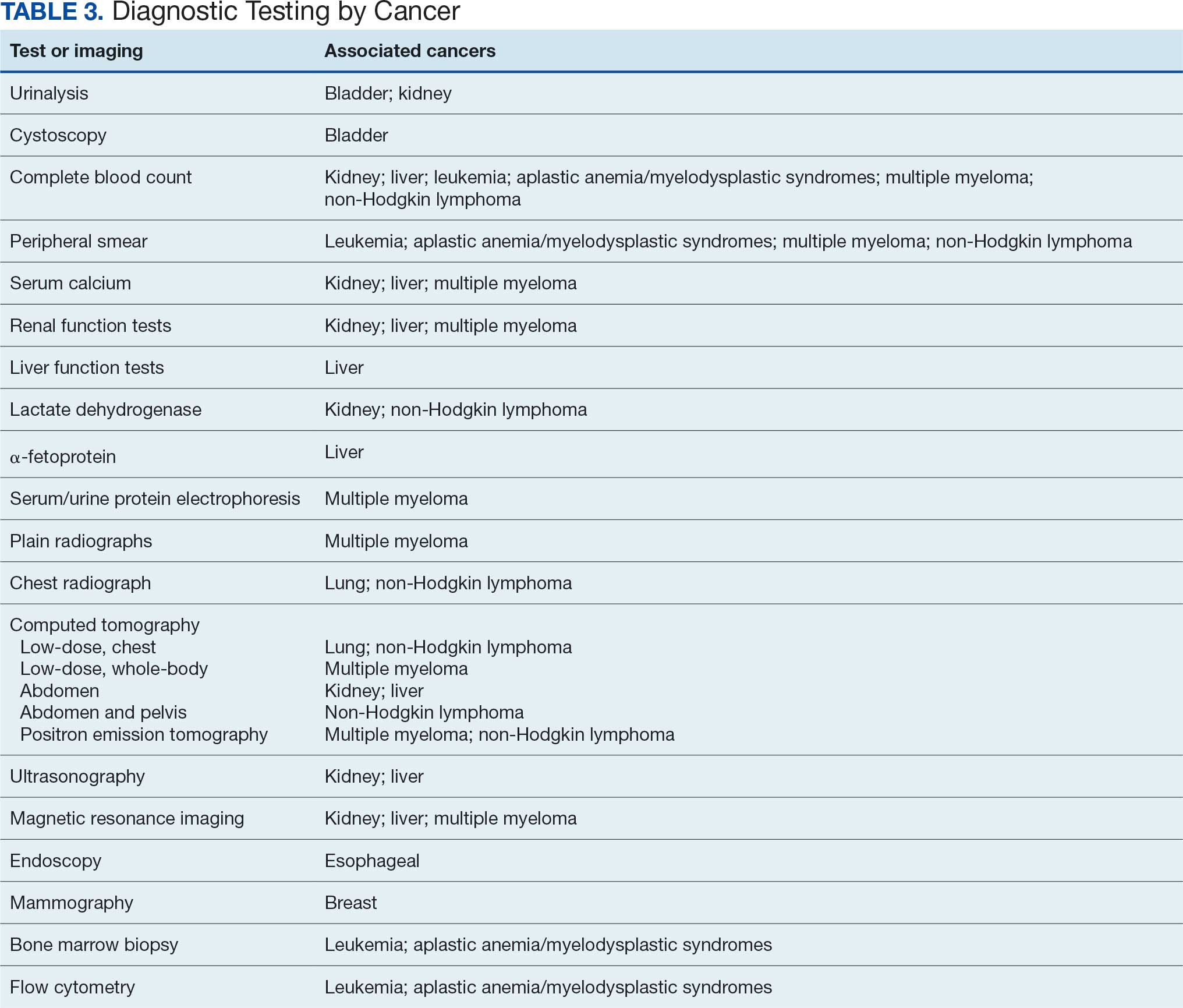
Primary care and internal medicine HCPs have the opportunity to improve patient health outcomes by implementing a targeted diagnostic screening panel for identified veterans previously stationed at Camp Lejeune. Early identification of cancers associated with VOCs exposure can facilitate earlier treatment interventions and improve health and quality of life outcomes. The following diagnostic screening panel outlines a potential cost-effective strategy for evaluating and detecting the 10 cancers associated with VOC exposure in Camp Lejeune water.
Baseline Screening
Implementing a diagnostic screening panel in this high-risk cohort can lead to earlier diagnosis, reduce mortality, and improve patient outcomes through early intervention, which in turn may result in less invasive treatment. This approach may also reduce health care costs by avoiding costs associated with delayed diagnosis and advanced-stage cancer care (Tables 3 and 4).
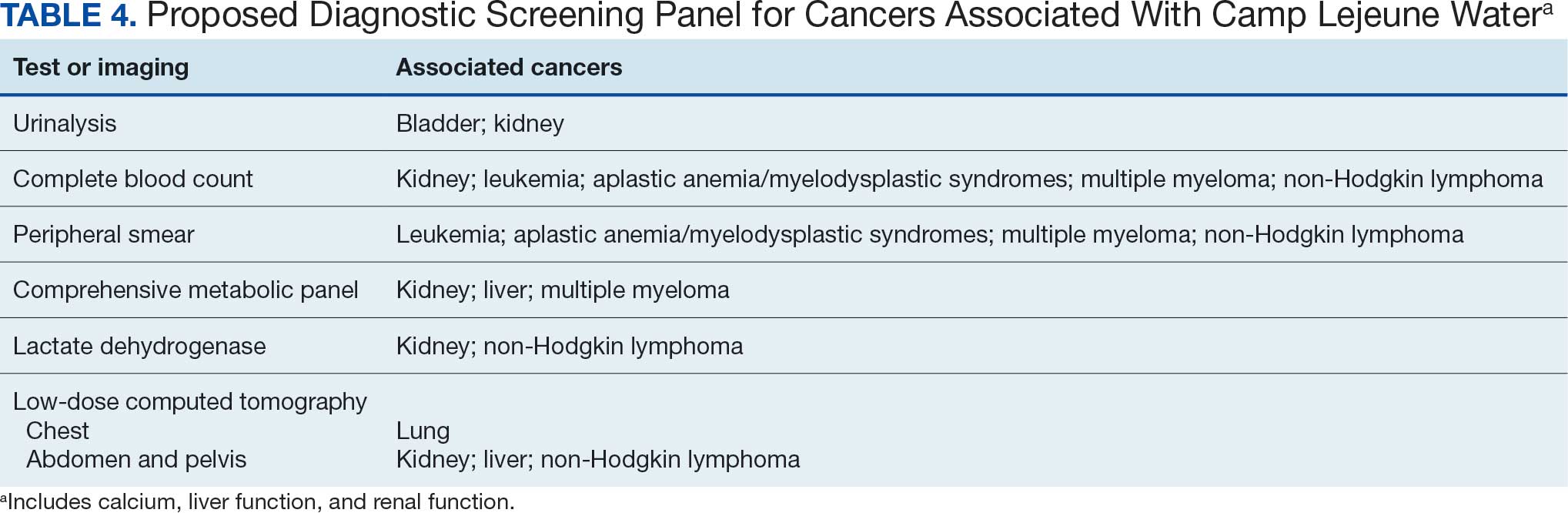
A baseline panel of tests for exposed veterans could include:
- A CBC with differential and peripheral smear to assess for anemia, leukemia, thrombocytopenia, and blast cells associated with leukemias, MDS, multiple myeloma, and NHL.19,46,47,52,55,56,60,61
- CMP evaluates calcium, total protein, renal and liver renal function. Elevated test results may indicate kidney or liver cancer or multiple myeloma.19,27,28,55,56
- LDH testing may reveal levels that are elevated from tissue damage or high cell turnover in kidney cancer, multiple myeloma, and NHL.19,55,56,60,61
- Urinalysis with microscopy may detect hematuria, proteinuria and cellular casts in bladder and kidney cancers.13,24,19
- Low-dose CTs of the chest, abdomen, and pelvis are recommended for early identification of any masses or lymphadenopathy in lung, kidney, liver cancers, and NHL.19,27,28,32,60,61
COST EFFICIENCY
Screening Panel Cost
According to the Medicare Clinical Laboratory Fee Schedule payment cap for 2018, the mean cost for the proposed blood workup was $35 (CBC, $10; CMP, $13; LDH, $8; urinalysis, $4).62 Medicare procedure price schedule for 2025 includes $351 for a CT of the abdomen and pelvis with and without contrast (Current Procedural Terminology [CPT] code 74177) and $187 for a CT of the chest with and without contrast (CPT code 71270).63,64 The total proposed diagnostic screening panel payment cost about $572.
Cancer Care Cost
The average cost for initial cancer care across all cancer sites from 2007 to 2013 was $43,516 per patient; Camp Lejeune-associated cancers ranged from $26,443 for bladder cancer to $89,947 for esophageal cancer care.64 Further, the last year of life cost across all cancer sites averaged $109,727, and Camp Lejeune-associated cancer types ranged from $76,101 for breast cancer to $169,588 for leukemia.65
CONCLUSIONS
From 1953 to 1985, up to 1 million military personnel, civilian workers, and their families stationed at Camp Lejeune were unknowingly exposed to toxic and carcinogenic VOCs, which are associated with = 10 cancers, including bladder, kidney, esophageal, liver, lung, breast, and hematologic malignancies.1-4 Some veterans may be asymptomatic, whereas others present with subtle or specific symptoms that can vary by individual and the type and stage of cancer. HCPs have an opportunity to improve patient outcomes through awareness in identifying symptoms associated with Camp Lejeune water exposure and performing a thorough baseline physical examination, especially noting lymphadenopathy, unexplained weight loss, or masses, which can guide further diagnostic evaluation. Timely screening can identify cancers earlier, reducing delays in care, mitigating the cost burden associated with advanced-stage cancer treatment, improving survival outcomes, and enhancing quality of life. Primary care and internal medicine HCPs specifically play a crucial role in early recognition, physical assessment, and appropriate screening tools. A proposed panel includes CBC with differential and peripheral smear, CMP, LDH, urinalysis, and low-dose CTs of the chest, abdomen and pelvis. Implementation should be guided by clinical judgment and patient-specific risk factors. The proposed diagnostic screening panel is a small price to pay for those who served in any capacity at Camp Lejeune.
- Bove FJ, Greek A, Gatiba R, et al. Cancer incidence among Marines and Navy personnel and civilian workers exposed to industrial solvents in drinking water at US Marine Corps Base Camp Lejeune: a cohort study. Environ Health Perspect. 2024;132:107008. doi:10.1289/EHP14966
- Maslia ML, Aral MM, Ruckart PZ, Bove FJ. Reconstructing historical VOC concentrations in drinking water for epidemiological studies at a US military base: summary of results. Water (Basel). 2016;8:449. doi:10.3390/w8100449
- Rosenfeld PE, Spaeth KR, McCarthy SJ, et al. Camp Lejeune Marine cancer risk assessment for exposure to contaminated drinking water from 1955 to 1987. Water Air Soil Pollut. 2024;235(2). doi:10.1007/s11270-023-06863-y
- US Department of Veterans Affairs, Veterans Health Administration. Camp Lejeune: past water contamination. Updated April 15, 2025. Accessed March 3, 2026. https://www.publichealth.va.gov/exposures/camp-lejeune/
- Jung K, Khan A, Mocharnuk R, et al. Clinical encounter with three cancer patients affected by groundwater contamination at Camp Lejeune: a case series and review of the literature. J Med Case Rep. 2022;16(1):272. doi:10.1186/s13256-022-03501-9
- Honoring America's Veterans and Caring for Camp Lejeune Familes Act of 2012, Pub L No. 112-154. Janey Ensminger Act. Congress.gov. Accessed April 15, 2026. https://ww.congress.gov/bill/112th-congress/house-bill/1627
- Bove FJ, Greek A, Gatiba R, et al. Evaluation of mortality among Marines, Navy personnel, and civilian workers exposed to contaminated drinking water at USMC Base Camp Lejeune: a cohort study. Environ Health. 2024;23(1):61. doi:10.1186/s12940-024-01099-7
- Honoring our PACT Act of 2022 (Pub L No. 117-168): expansion of health care eligibility and toxic exposure screenings. Congress.gov. Accessed March 3, 2026. https://www.congress.gov/bill/117th-congress/house-bill/3967
- Ensuring Justice for Camp Lejeune Victims Act of 2025. Congress.gov. Accessed March 24, 2026. https://www.congress.gov/bill/119th-congress/house-bill/4145
- SEER. Cancer stat facts: bladder cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/urinb.html
- Agency for Toxic Substances and Disease Registry. ATSDR assessment of the evidence for the drinking water contaminants at Camp Lejeune and specific cancers and other diseases. Published January 13, 2017. Accessed March 3, 2026. https://www.atsdr.cdc.gov/camp-lejeune/media/pdfs/2024/10/ATSDR_summary_of_the_evidence_for_causality_TCE_PCE_508.pdf
- National Cancer Institute. What is bladder cancer? Updated February 16, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder
- National Cancer Institute. Bladder cancer symptoms. Updated February 16, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder/symptoms
- American Cancer Society. Bladder cancer signs and symptoms. Updated March 12, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/bladder-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- National Cancer Institute. Bladder cancer screening. Updated April 27, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder/screening
- American Cancer Society. Tests for bladder cancer. Updated March 12, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/bladder-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: kidney and renal pelvis cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/kidrp.html
- American Cancer Society. Kidney cancer signs and symptoms. Updated May 1, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/kidney-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- American Cancer Society. Tests for kidney cancer. Updated May 1, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/kidney-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: esophageal cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/esoph.html
- Engel LS, Chow WH, Vaughan TL, et al. Population attributable risks of esophageal and gastric cancers.
J Natl Cancer Inst. 2003;95(18):1404-1413. doi:10.1093/jnci/djg047 - American Cancer Society. Signs and symptoms of esophageal cancer. Updated March 20, 2020. Accessed March 3, 2026. https://www.cancer.org/cancer/types/esophagus-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- American Cancer Society. Tests for esophageal cancer. Updated March 20, 2020. Accessed March 3, 2026. https://www.cancer.org/cancer/types/esophagus-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: liver and intrahepatic bile duct cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/livibd.html
- American Cancer Society. Signs and symptoms of liver cancer. Updated February 11, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/liver-cancer/detection-diagnosis-staging/signs-symptoms.html
- National Cancer Institute. What is liver cancer? Updated May 15, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/liver/what-is-liver-cancer
- American Cancer Society. Tests for liver cancer. Updated February 11, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/liver-cancer/detection-diagnosis-staging/how-diagnosed.html
- National Cancer Institute. Liver cancer screening. Updated May 15, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/liver/what-is-liver-cancer/screening
- SEER. Cancer stat facts: lung cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/lungb.html
- US Department of Health and Human Services. The Health Consequences of Smoking: A Report of the Surgeon General. Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health; 2004. Accessed March 3, 2026. https://www.ncbi.nlm.nih.gov/books/NBK44695/pdf/Bookshelf_NBK44695.pdf
- American Cancer Society. Lung cancer signs and symptoms. Updated February 27, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/lung-cancer/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for lung cancer. Updated January 29, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/lung-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: female breast cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/breast.html
- SEER. SEER*Explorer breast incidence and mortality comparison. Updated July 2, 2025. Accessed March 3, 2026. https://seer.cancer.gov/statistics-network/explorer/application.html
- Susan G. Komen. Male breast cancer. Updated June 3, 2025. Accessed March 3, 2026. https://www.komen.org/breast-cancer/facts-statistics/male-breast-cancer/
- American Cancer Society. Key statistics for breast cancer in men. Updated January 16, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/about/key-statistics.html
- Ruckart PZ, Bove FJ, Shanley E 3rd, et al. Evaluation of contaminated drinking water and male breast cancer at Marine Corps Base Camp Lejeune, North Carolina: a case-control study. Environ Health. 2015;14:74. doi:10.1186/s12940-015-0061-4
- Culell P, Solernou L, Tarazona J, et al. Male breast cancer: a multicentric study. Breast J. 2007;13:213-215. doi:10.1111/j.1524-4741.2007.00412.x
- Giordano SH, Cohen DS, Buzdar AU, et al. Breast carcinoma in men: a population-based study. Cancer. 2004;101:51-57. doi:10.1002/cncr.20312
- American Cancer Society. Signs and symptoms of breast cancer in men. Updated April 27, 2018. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests to help diagnose breast cancer in men. Updated December 20, 2021. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/detection-diagnosis-staging/how-diagnosed.html
- Evans GF, Anthony T, Turnage RH, et al. The diagnostic accuracy of mammography in the evaluation of male breast disease. Am J Surg. 2001;181:96-100. doi:10.1016/S0002-9610(00)00571-7
- SEER. Cancer stat facts: leukemia. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/leuks.html
- American Cancer Society. Signs and symptoms of acute myeloid leukemia (AML). Updated March 20, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/acute-myeloid-leukemia/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Signs and symptoms of chronic lymphocytic leukemia (CLL). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/chronic-lymphocytic-leukemia/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for acute myeloid leukemia (AML). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/acute-myeloid-leukemia/detection-diagnosis-staging/how-diagnosed.html
- American Cancer Society. Tests for chronic lymphocytic leukemia (CLL). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/chronic-lymphocytic-leukemia/detection-diagnosis-staging/how-diagnosed.html
- Giudice V, Selleri C. Aplastic anemia: pathophysiology. Semin Hematol. 2022;59:13-20. doi:10.1053/j.seminhematol.2021.12.002
- National Cancer Institute. Myelodysplastic syndromes treatment (PDQ®)–patient version. Updated October 4, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/myeloproliferative/patient/myelodysplastic-treatment-pdq
- SEER. Cancer statistics review (CSR) 1975-2013: myelodysplastic syndromes. Accessed March 3, 2026. https://seer.cancer.gov/archive/csr/1975_2013/browse_csr.php?sectionSEL=30&pageSEL=sect_30_table.01
- American Cancer Society. Signs and symptoms of myelodysplastic syndrome (MDS). Updated November 21, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/myelodysplastic-syndrome/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for myelodysplastic syndromes (MDS). Updated November 21, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/myelodysplastic-syndrome/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: myeloma. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/mulmy.html
- American Cancer Society. Signs and symptoms of multiple myeloma. Updated February 28, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/multiple-myeloma/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for multiple myeloma. Updated February 28, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/multiple-myeloma/detection-diagnosis-staging/testing.html
- Laubach JP. Multiple myeloma: clinical features, laboratory manifestations, and diagnosis. UpToDate. Updated April 28, 2025. Accessed March 3, 2026. https://www.uptodate.com/contents/multiple-myeloma-clinical-features-laboratory-manifestations-and-diagnosis
- Hillengass J, Usmani S, Rajkumar SV, et al. International Myeloma Working Group consensus recommendations on imaging in monoclonal plasma cell disorders. Lancet Oncol. 2019;20:e302-e312. doi:10.1016/S1470-2045(19)30309-2
- SEER. Cancer stat facts: non-Hodgkin lymphoma. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/nhl.html
- American Cancer Society. Signs and symptoms of non-Hodgkin lymphoma. Updated February 15, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/non-hodgkin-lymphoma/detection-diagnosis-staging/signs-symptoms.html
- National Cancer Institute. Non-Hodgkin lymphoma treatment (PDQ®)–patient version. Updated August 22, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/lymphoma/patient/adult-nhl-treatment-pdq
- American Cancer Society. Tests for non-Hodgkin lymphoma. Updated February 15, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/non-hodgkin-lymphoma/detection-diagnosis-staging/how-diagnosed.html
- College of American Pathologists. Medicare clinical laboratory fee schedule. Published November 2017. Accessed March 3, 2026. https://documents.cap.org/documents/2018-final-medicare-clfs-rates.pdf
- Medicare.gov. Procedure price lookup for outpatient services. Accessed March 3, 2026. https://www.medicare.gov/procedure-price-lookup/cost/71270/
- Medicare.gov. Procedure price lookup for outpatient services. Accessed March 3, 2026. https://www.medicare.gov/procedure-price-lookup/cost/74177/
- National Cancer Institute. Cancer trends progress report: financial burden of cancer care. Updated April 2025. Accessed March 3, 2026. https://progressreport.cancer.gov/after/economic_burden
Clinical awareness of cancers associated with Camp Lejeune water contamination exposure remains limited despite legal and policy advances. Gaps persist in early symptom recognition and timely diagnostic evaluation before a definitive cancer diagnosis among exposed personnel. This may represent missed opportunities for earlier identification of volatile organic compounds (VOCs)-related cancers and for less invasive treatment options for veterans in this high-risk population.
Federal health care practitioners (HCPs), especially those in primary care and internal medicine, are uniquely positioned to bridge this gap. By improving the recognition of symptoms, pertinent physical examination findings, and implementing a diagnostic screening panel, HCPs can support accurate diagnoses and facilitate earlier treatment to improve health and quality of life for this population.
From 1953 to 1985, as many as 1 million military personnel, civilian workers, and their families stationed at US Marine Corps Base Camp Lejeune were unknowingly exposed to toxic and carcinogenic chemicals in drinking and bathing water.1 Three of the 8 main water sources on base were contaminated with VOCs, which are associated with multiple cancers.1-3
The US Department of Veterans Affairs (VA) recognizes 15 conditions associated with Camp Lejeune contaminated water exposure for VA benefits, including 10 cancers: adult leukemia; aplastic anemia and other myelodysplastic syndromes (MDS); bladder, esophageal, kidney, liver, breast (male and female), and lung cancers; multiple myeloma; and non-Hodgkin lymphoma (NHL).4
BACKGROUND
Established in 1942, Camp Lejeune is an important Marine Corps training installation. Between 1953 and 1985, multiple on-base water systems were contaminated with VOCs, including trichloroethylene (TCE), perchloroethylene (PCE), benzene, and vinyl chloride, due to improper waste disposal and industrial runoff from on- and off-base sources.5 Tarawa Terrace water treatment plant (WTP) was contaminated primarily with PCE from November 1957 to February 1987. Hadnot Point WTP was contaminated with TCE from August 1953 to December 1984, along with PCE, and benzene, toluene, ethylbenzene, and xylene (BTEX). Holcomb Boulevard WTP, established in 1972, was contaminated with TCE from June 1972 to February 1985.2 These contaminants entered the drinking and bathing water supply over decades, and exposure often occurred concurrently across = 1 VOC, compounding health risks.2,3 This prolonged 32-year VOC exposure window underlies current concerns regarding long-term cancer risk among affected service members, civilian employees, and family members. Epidemiologic research has found statistically significant associations between VOC exposure and multiple cancers, neurologic conditions, and reproductive issues.6 Specifically, TCE is associated with higher risks of hematologic cancers, multiple myeloma, NHL, and kidney cancer.3 PCE is linked with kidney cancer, benzene with multiple myeloma and NHL, and vinyl chloride with hepatobiliary cancers.3 A cohort mortality study compared Camp Lejeune personnel with a control group at Camp Pendleton from 1972 to 1985 and found a 3-fold higher incidence or mortality rate for kidney, esophageal, and female breast cancers, leukemia, and lymphoma among exposed Camp Lejeune personnel.6 Notably, personnel assigned to Camp Lejeune for as little as 6 months faced up to a 6-fold increase in cancer risk; the average military assignment between 1975 and 1985 was 18 months.3,6
Honoring America's Veterans and Caring for Camp Lejeune Families Act of 2012, the Sergeant First Class Heath Robinson Honoring Our Promise to Address Comprehensive Toxics (PACT) Act of 2022, the Camp Lejeune Justice Act of 2022, and the pending Ensuring Justice for Camp Lejeune Victims Act of 2025 provide health care and legal resources for personnel and families affected by Camp Lejeune’s contaminated water.6-8 These laws acknowledge associations between exposure and specific health conditions and expanded health care, benefits, and legal recourse for affected veterans, survivors, and their families.8,9
CANCERS LINKED TO CAMP LEJEUNE
Camp Lejeune VOC-contaminated water exposure is associated with solid tumor and hematologic cancers. Symptoms, physical examination findings, and diagnostic considerations vary by cancer type (Table 1).

Bladder Cancer
The US incidence rate of bladder cancer for both males and females is 18 per 100,000 individuals per year, with a death rate of 4.1 per 100,000 individuals per year, and a 2.1% lifetime diagnosis risk.10 Personnel exposed to VOCs at Camp Lejeune had a 9% higher risk of developing bladder cancer and a 2% increased mortality compared with an unexposed control group at Camp Pendleton.1,7 Other bladder cancer subtypes at increased risk are papillary transitional cell carcinoma, nonpapillary transition cell carcinoma, and urothelial carcinoma.7 This is consistent with prior research that found PCE exposure is associated with an increased risk for bladder cancer.3,7,11 Smoking and tobacco use remain significant risk factors for bladder cancer.12
Symptomatology. The most common symptom associated with bladder cancer is painless hematuria (gross or microscopic). Other often delayed symptoms include urinary frequency, urgency, or nocturia.13,14
Diagnostics. Screening tests include urinalysis for hematuria, urine cytology, and cystoscopy with biopsy as the gold standard for diagnosis and staging.15,16
Kidney Cancer
The US incidence rate of kidney cancer and renal pelvis cancer for both males and females is 17.5 per 100,000 individuals per year, with a death rate of 3.4 per 100,000, and a 1.8% lifetime diagnosis risk.17 Camp Lejeune personnel exposed to VOCs had a 6% increased risk of developing kidney cancer and renal pelvis cancer and a 21% higher mortality risk compared with Camp Pendleton controls.1,7 Subtypes at risk include renal cell carcinoma and papillary carcinoma.7 This is consistent with prior research that found exposures to TCE and PCE are associated with a 3-fold increased risk of kidney cancer.3,7
Symptomatology. Hematuria, flank pain, and a palpable abdominal mass are common symptoms associated with kidney cancer. In advanced stages, other symptoms may include left-sided varicocele, anemia, weight loss, fatigue, fever, and night sweats.18
Diagnostics. Screening tests include urinalysis to assess the presence of blood, complete blood count (CBC) to assess anemia, calcium (elevated), and lactate dehydrogenase (LDH), which may be elevated. Imaging strategies include abdominal computed tomography (CT), magnetic resonance imaging (MRI), or ultrasound.19
Esophageal Cancer
The US incidence rate of esophageal cancer for both males and females is 4.2 per 100,000 individuals per year, the death rate is 3.7 per 100,000 individuals per year, and a 0.5% lifetime diagnosis risk.20 VOC-exposed Camp Lejeune personnel had a 27% increased incidence and 25% increased mortality compared with the control group.1,7 Esophageal cancer subtypes at elevated risk include squamous cell carcinoma and adenocarcinoma. This is consistent with prior research that found Camp Lejeune water exposure is associated with a 3-fold increased risk for esophageal cancer.7 Additional risk factors include history of smoking and alcohol use.21
Symptomatology. Esophageal cancer is often asymptomatic with potential symptoms that include dysphagia, hoarseness, and weight loss in advanced disease.22
Diagnostics. Endoscopy with biopsy is the definitive method for diagnosis.23
Liver Cancer
The US incidence rate of liver cancer and intrahepatic bile duct cancer for both males and females is 9.4 per 100,000 individuals per year, with a death rate of 6.6 per 100,000 individuals per year, and a 1.1% lifetime diagnosis risk.24 VOC-exposed personnel had a 1% higher mortality than controls.1
Symptomatology. Liver cancer is often asymptomatic and appears in late stages.25 Common symptoms include right upper quadrant pain, early satiety, nausea, vomiting, loss of appetite, weight loss, ascites, jaundice, and abnormal bleeding or bruising.25,26
Diagnostics. Diagnostic tests may include an ultrasound, CT, or MRI. Additional laboratory testing may include liver function, a-fetoprotein blood, CBC, renal function, calcium, and hepatitis panel screening for hepatitis B and C.27,28
Lung Cancer
The US incidence rate of lung cancer for both males and females is 47.8 per 100,000 individuals per year, with a death rate of 31.5 per 100,000 individuals per year, and a 5.4% lifetime diagnosis risk.29 VOC-exposed personnel had a 16% increased risk and 19% higher mortality.1,7 Subtypes include large cell, small cell, non-small cell, squamous cell, and adenocarcinoma.7 Smoking is an additional risk factor.30
Symptomatology. Symptoms of lung cancer include cough, shortness of breath, chest pain worse with deep breathing, unexplained weight loss, fatigue, night sweats, and recurrent fevers. Advanced stages may metastasize or spread to the liver, bones, and brain.31
Diagnostics. Low-dose CT and chest X-ray are used for screening.32
Breast Cancer
The US incidence rate of female breast cancer is 130.8 per 100,000 individuals per year, with a death rate of 19.2 per 100,000 individuals per year, and a 13.0% lifetime risk of diagnosis.33 For female VOC-exposed personnel, there was an equal risk of developing breast cancer as the control group.1 However, exposed females at Camp Lejeune had a 23% higher mortality risk compared to the control group.7 Breast cancer subtypes among females include ductal carcinoma, lobular carcinoma, and ductal-lobular carcinoma.1
The US incidence rate of male breast cancer is 1.3 per 100,000 individuals per year, with a death rate of 0.3 per 100,000 individuals per year.34,35 The lifetime risk for males developing breast cancer is 137.7 per 100,000 and about 70 to 100 times less common in men than women.36
Male personnel exposed at Camp Lejeune had a 4% increased risk for developing breast cancer compared to Camp Pendleton.7 However, mortality was lower in the Camp Lejeune group.1 Although male breast cancer is rare, males at Camp Lejeune had a higher incidence, indicating a link between TCE, PCE, vinyl chloride exposures and male breast cancer.37 Male breast cancer is more often diagnosed in advanced stages than female breast cancer due to the lack of awareness or absence of routine screenings.38 The most common breast cancer type in males is invasive ductal carcinoma, accounting for 85% to 90% of cases; lobular carcinoma is the second most common type.39
Symptomatology. In both females and males, breast cancer symptoms include painless, firm mass or lump in the breast (left breast slightly more common than right), skin changes or dimpling, nipple retraction or turning inward, and nipple discharge. Breast cancer can spread to the lymph nodes and can be appreciated in axilla or clavicular regions.40
Diagnostics. The diagnostic evaluation for breast cancer is similar for females and males. It includes a clinical breast examination, diagnostic mammogram, and ultrasound.41 Mammograms can distinguish between gynecomastia and cancer, especially in males.42 A core or fine needle biopsy is needed to confirm diagnosis.41
Adult Leukemia
The US incidence rate of leukemia for both male and female was 14.4 per 100,000 individuals per year, with a death rate of 5.8 per 100,000 individuals per year, and a 1.5% lifetime diagnosis risk.43
VOC-exposed personnel had a 7% higher risk of developing leukemia and a 13% increased mortality risk compared with the control group.1,7 Subtypes of leukemia at risk included a 38% increased incidence of acute myeloid/monocytic leukemia (AML) and a 2% increased incidence of chronic lymphocytic leukemia (CLL).1 Benzene and TCE exposures are known risk factors for AML and other leukemias.7 Personnel at Camp Lejeune had 3 times the incidence or mortality for leukemia, specifically AML mortality at 20%.7 Smoking is an additional risk factor for certain leukemias, especially AML.30
Symptomatology. Symptoms associated with leukemia are often nonspecific and may include fatigue, pallor, easy bruising or bleeding (skin or gums), recurrent infections secondary to neutropenia, fever, night sweats, pain or feeling full after a small meal due to enlarged spleen or liver, and weight loss.44,45
Diagnostics. An initial screening includes a CBC with differential, a peripheral smear to detect the presence of blast cells, as well as Auer rods in myeloid blast cells in AML or smudge cells in CLL. Confirmatory tests may include bone marrow biopsy or flow cytometry. A referral to a hematologist is recommended for any suspected leukemia.46,47
Myelodysplastic Syndromes
Aplastic anemia and MDS are considered rare disorders.48 Aplastic anemia is a nonmalignant bone marrow failure disorder with pancytopenia and hypocellular bone marrow due to the loss of hematopoietic stem cells.48 MDS is a type of hematopoietic cancer where the bone marrow produces abnormal blood cells or does not make enough healthy cells.49 This can lead to an increased risk for infection, cytopenias, neutropenia, refractory anemia, and thrombocytopenia, and progression to AML in some patients.49
The reported US incidence of MDS from 1975 to 2013 was 6.7 per 100,000 for males and 3.7 per 100,000 for females.50 Benzene exposure is linked to MDS and a known cause of AML.1 VOC-exposed personnel had a 68% increased risk of developing MDS and a 2.3-fold increased mortality risk compared to controls.1,7
Symptomatology. Some patients are asymptomatic at diagnosis.51 Symptoms related to cytopenia include fatigue, pallor, purpura, petechiae, bleeding of skin, gum, or nose, recurrent infections, fever, bone pain, loss of appetite, and weight loss.50,51
Diagnostics. Initial workup includes a CBC with differential to assess for anemia, white blood cell and absolute neutrophil counts (low), and thrombocytopenia.52 A peripheral blood smear may show myeloid blast cells. A bone marrow aspiration and biopsy, flow cytometry, and cytogenetic or molecular testing may be performed. If MDS is suspected, a referral to a hematologist should be considered.52
Multiple Myeloma
The US incidence rate of multiple myeloma for both males and females is 7.3 per 100,000 individuals per year, with a mortality rate of 2.9 per 100,000 individuals per year, and a 0.8% lifetime diagnosis risk.53 VOC-exposed personnel had a 13% increased risk of developing multiple myeloma and an 8% increased mortality risk compared to unexposed personnel.1,7
Symptomatology. Multiple myeloma may be asymptomatic in early stages. The most common presenting symptom is bone pain, especially in the back, hips, and long bones, due to hypercalcemia from increased reabsorption, plasma cell tumor overgrowth in the bone marrow, and lytic lesions.54 Additional symptoms include fatigue and pallor related to anemia, leukopenia, thrombocytopenia, recurrent infections, extreme thirst, frequent urination, dehydration, confusion associated with hypercalcemia, peripheral neuropathy, loss of appetite, weight loss, and renal impairment or failure.54
Diagnostics. Testing considerations include a CBC with a peripheral blood smear to evaluate anemia and rouleaux formation of red blood cells (seen in > 50% of patients with multiple myeloma), comprehensive metabolic panel (CMP) to assess kidney function, calcium levels (elevated), serum and urine protein electrophoresis with immunofixation to detect monoclonal protein (detected in > 80% of patients with multiple myeloma) and Bence-Jones proteins, serum free light chain assay, and a bone marrow biopsy for diagnosis.55,56
MRI of the spine and pelvis is the most sensitive to detecting bone marrow involvement and focal lesions before lytic lesion progression occurs and for assessing spinal cord compression.57 PET/CT is more sensitive at detecting extramedullary disease, outside of the spine, and for patients that cannot undergo MRI.57 A whole-body low-dose CT, either alone or with PET, is more sensitive than an X-ray at detecting lytic lesions, fractures, or osteoporosis associated with multiple myeloma.57
Non-Hodgkin Lymphoma
The US incidence rate of NHL for both males and females are 18.7 per 100,000 individuals per year, the death rate is 4.9 per 100,000 individuals per year, and a 2% lifetime diagnosis risk.58 VOC-exposed personnel had a 1% higher risk of developing NHL and a decreased mortality risk compared to the control group.1,7 Specific NHL subtypes with increased risk in the exposed cohort are mantle cell (26%), follicular (7%), Burkitt (53%), and marginal zone B-cell (45%).7
Symptomatology. NHL often presents with painless lymphadenopathy or enlarged lymph nodes involving the cervical, axillary, inguinal regions.59,60 Other symptoms include frequent infections, unexplained bruising, weight loss, and “B symptoms,” such as fever and night sweats.59,60 Some patients develop a mediastinal mass in the thorax, which if large may lead to cough or shortness of breath.59
Diagnostics. The initial diagnostic workup includes CBC with differential and LDH, which may be elevated.60,61 Imaging may begin with a chest X-ray to assess for a mediastinal mass; however, CTs of the chest, abdomen, and pelvis provide more detail to better assess for NHL. Whole body PET/CT is considered the gold standard for assessing and staging systemic involvement. If enlarged lymph nodes are present, a biopsy can confirm the subtype of NHL.60,61
PHYSICAL EXAMINATION
A focused physical examination may aid HCPs in early detection of the cancers associated with Camp Lejeune (Table 2). The physical examination can guide diagnostic testing and imaging for further assessment and workup for VOC-related cancers.

Proposed Diagnostic Screening Panel
Primary care and internal medicine HCPs have the opportunity to improve patient health outcomes by implementing a targeted diagnostic screening panel for identified veterans previously stationed at Camp Lejeune. Early identification of cancers associated with VOCs exposure can facilitate earlier treatment interventions and improve health and quality of life outcomes. The following diagnostic screening panel outlines a potential cost-effective strategy for evaluating and detecting the 10 cancers associated with VOC exposure in Camp Lejeune water.
Baseline Screening
Implementing a diagnostic screening panel in this high-risk cohort can lead to earlier diagnosis, reduce mortality, and improve patient outcomes through early intervention, which in turn may result in less invasive treatment. This approach may also reduce health care costs by avoiding costs associated with delayed diagnosis and advanced-stage cancer care (Tables 3 and 4).


A baseline panel of tests for exposed veterans could include:
- A CBC with differential and peripheral smear to assess for anemia, leukemia, thrombocytopenia, and blast cells associated with leukemias, MDS, multiple myeloma, and NHL.19,46,47,52,55,56,60,61
- CMP evaluates calcium, total protein, renal and liver renal function. Elevated test results may indicate kidney or liver cancer or multiple myeloma.19,27,28,55,56
- LDH testing may reveal levels that are elevated from tissue damage or high cell turnover in kidney cancer, multiple myeloma, and NHL.19,55,56,60,61
- Urinalysis with microscopy may detect hematuria, proteinuria and cellular casts in bladder and kidney cancers.13,24,19
- Low-dose CTs of the chest, abdomen, and pelvis are recommended for early identification of any masses or lymphadenopathy in lung, kidney, liver cancers, and NHL.19,27,28,32,60,61
COST EFFICIENCY
Screening Panel Cost
According to the Medicare Clinical Laboratory Fee Schedule payment cap for 2018, the mean cost for the proposed blood workup was $35 (CBC, $10; CMP, $13; LDH, $8; urinalysis, $4).62 Medicare procedure price schedule for 2025 includes $351 for a CT of the abdomen and pelvis with and without contrast (Current Procedural Terminology [CPT] code 74177) and $187 for a CT of the chest with and without contrast (CPT code 71270).63,64 The total proposed diagnostic screening panel payment cost about $572.
Cancer Care Cost
The average cost for initial cancer care across all cancer sites from 2007 to 2013 was $43,516 per patient; Camp Lejeune-associated cancers ranged from $26,443 for bladder cancer to $89,947 for esophageal cancer care.64 Further, the last year of life cost across all cancer sites averaged $109,727, and Camp Lejeune-associated cancer types ranged from $76,101 for breast cancer to $169,588 for leukemia.65
CONCLUSIONS
From 1953 to 1985, up to 1 million military personnel, civilian workers, and their families stationed at Camp Lejeune were unknowingly exposed to toxic and carcinogenic VOCs, which are associated with = 10 cancers, including bladder, kidney, esophageal, liver, lung, breast, and hematologic malignancies.1-4 Some veterans may be asymptomatic, whereas others present with subtle or specific symptoms that can vary by individual and the type and stage of cancer. HCPs have an opportunity to improve patient outcomes through awareness in identifying symptoms associated with Camp Lejeune water exposure and performing a thorough baseline physical examination, especially noting lymphadenopathy, unexplained weight loss, or masses, which can guide further diagnostic evaluation. Timely screening can identify cancers earlier, reducing delays in care, mitigating the cost burden associated with advanced-stage cancer treatment, improving survival outcomes, and enhancing quality of life. Primary care and internal medicine HCPs specifically play a crucial role in early recognition, physical assessment, and appropriate screening tools. A proposed panel includes CBC with differential and peripheral smear, CMP, LDH, urinalysis, and low-dose CTs of the chest, abdomen and pelvis. Implementation should be guided by clinical judgment and patient-specific risk factors. The proposed diagnostic screening panel is a small price to pay for those who served in any capacity at Camp Lejeune.
Clinical awareness of cancers associated with Camp Lejeune water contamination exposure remains limited despite legal and policy advances. Gaps persist in early symptom recognition and timely diagnostic evaluation before a definitive cancer diagnosis among exposed personnel. This may represent missed opportunities for earlier identification of volatile organic compounds (VOCs)-related cancers and for less invasive treatment options for veterans in this high-risk population.
Federal health care practitioners (HCPs), especially those in primary care and internal medicine, are uniquely positioned to bridge this gap. By improving the recognition of symptoms, pertinent physical examination findings, and implementing a diagnostic screening panel, HCPs can support accurate diagnoses and facilitate earlier treatment to improve health and quality of life for this population.
From 1953 to 1985, as many as 1 million military personnel, civilian workers, and their families stationed at US Marine Corps Base Camp Lejeune were unknowingly exposed to toxic and carcinogenic chemicals in drinking and bathing water.1 Three of the 8 main water sources on base were contaminated with VOCs, which are associated with multiple cancers.1-3
The US Department of Veterans Affairs (VA) recognizes 15 conditions associated with Camp Lejeune contaminated water exposure for VA benefits, including 10 cancers: adult leukemia; aplastic anemia and other myelodysplastic syndromes (MDS); bladder, esophageal, kidney, liver, breast (male and female), and lung cancers; multiple myeloma; and non-Hodgkin lymphoma (NHL).4
BACKGROUND
Established in 1942, Camp Lejeune is an important Marine Corps training installation. Between 1953 and 1985, multiple on-base water systems were contaminated with VOCs, including trichloroethylene (TCE), perchloroethylene (PCE), benzene, and vinyl chloride, due to improper waste disposal and industrial runoff from on- and off-base sources.5 Tarawa Terrace water treatment plant (WTP) was contaminated primarily with PCE from November 1957 to February 1987. Hadnot Point WTP was contaminated with TCE from August 1953 to December 1984, along with PCE, and benzene, toluene, ethylbenzene, and xylene (BTEX). Holcomb Boulevard WTP, established in 1972, was contaminated with TCE from June 1972 to February 1985.2 These contaminants entered the drinking and bathing water supply over decades, and exposure often occurred concurrently across = 1 VOC, compounding health risks.2,3 This prolonged 32-year VOC exposure window underlies current concerns regarding long-term cancer risk among affected service members, civilian employees, and family members. Epidemiologic research has found statistically significant associations between VOC exposure and multiple cancers, neurologic conditions, and reproductive issues.6 Specifically, TCE is associated with higher risks of hematologic cancers, multiple myeloma, NHL, and kidney cancer.3 PCE is linked with kidney cancer, benzene with multiple myeloma and NHL, and vinyl chloride with hepatobiliary cancers.3 A cohort mortality study compared Camp Lejeune personnel with a control group at Camp Pendleton from 1972 to 1985 and found a 3-fold higher incidence or mortality rate for kidney, esophageal, and female breast cancers, leukemia, and lymphoma among exposed Camp Lejeune personnel.6 Notably, personnel assigned to Camp Lejeune for as little as 6 months faced up to a 6-fold increase in cancer risk; the average military assignment between 1975 and 1985 was 18 months.3,6
Honoring America's Veterans and Caring for Camp Lejeune Families Act of 2012, the Sergeant First Class Heath Robinson Honoring Our Promise to Address Comprehensive Toxics (PACT) Act of 2022, the Camp Lejeune Justice Act of 2022, and the pending Ensuring Justice for Camp Lejeune Victims Act of 2025 provide health care and legal resources for personnel and families affected by Camp Lejeune’s contaminated water.6-8 These laws acknowledge associations between exposure and specific health conditions and expanded health care, benefits, and legal recourse for affected veterans, survivors, and their families.8,9
CANCERS LINKED TO CAMP LEJEUNE
Camp Lejeune VOC-contaminated water exposure is associated with solid tumor and hematologic cancers. Symptoms, physical examination findings, and diagnostic considerations vary by cancer type (Table 1).

Bladder Cancer
The US incidence rate of bladder cancer for both males and females is 18 per 100,000 individuals per year, with a death rate of 4.1 per 100,000 individuals per year, and a 2.1% lifetime diagnosis risk.10 Personnel exposed to VOCs at Camp Lejeune had a 9% higher risk of developing bladder cancer and a 2% increased mortality compared with an unexposed control group at Camp Pendleton.1,7 Other bladder cancer subtypes at increased risk are papillary transitional cell carcinoma, nonpapillary transition cell carcinoma, and urothelial carcinoma.7 This is consistent with prior research that found PCE exposure is associated with an increased risk for bladder cancer.3,7,11 Smoking and tobacco use remain significant risk factors for bladder cancer.12
Symptomatology. The most common symptom associated with bladder cancer is painless hematuria (gross or microscopic). Other often delayed symptoms include urinary frequency, urgency, or nocturia.13,14
Diagnostics. Screening tests include urinalysis for hematuria, urine cytology, and cystoscopy with biopsy as the gold standard for diagnosis and staging.15,16
Kidney Cancer
The US incidence rate of kidney cancer and renal pelvis cancer for both males and females is 17.5 per 100,000 individuals per year, with a death rate of 3.4 per 100,000, and a 1.8% lifetime diagnosis risk.17 Camp Lejeune personnel exposed to VOCs had a 6% increased risk of developing kidney cancer and renal pelvis cancer and a 21% higher mortality risk compared with Camp Pendleton controls.1,7 Subtypes at risk include renal cell carcinoma and papillary carcinoma.7 This is consistent with prior research that found exposures to TCE and PCE are associated with a 3-fold increased risk of kidney cancer.3,7
Symptomatology. Hematuria, flank pain, and a palpable abdominal mass are common symptoms associated with kidney cancer. In advanced stages, other symptoms may include left-sided varicocele, anemia, weight loss, fatigue, fever, and night sweats.18
Diagnostics. Screening tests include urinalysis to assess the presence of blood, complete blood count (CBC) to assess anemia, calcium (elevated), and lactate dehydrogenase (LDH), which may be elevated. Imaging strategies include abdominal computed tomography (CT), magnetic resonance imaging (MRI), or ultrasound.19
Esophageal Cancer
The US incidence rate of esophageal cancer for both males and females is 4.2 per 100,000 individuals per year, the death rate is 3.7 per 100,000 individuals per year, and a 0.5% lifetime diagnosis risk.20 VOC-exposed Camp Lejeune personnel had a 27% increased incidence and 25% increased mortality compared with the control group.1,7 Esophageal cancer subtypes at elevated risk include squamous cell carcinoma and adenocarcinoma. This is consistent with prior research that found Camp Lejeune water exposure is associated with a 3-fold increased risk for esophageal cancer.7 Additional risk factors include history of smoking and alcohol use.21
Symptomatology. Esophageal cancer is often asymptomatic with potential symptoms that include dysphagia, hoarseness, and weight loss in advanced disease.22
Diagnostics. Endoscopy with biopsy is the definitive method for diagnosis.23
Liver Cancer
The US incidence rate of liver cancer and intrahepatic bile duct cancer for both males and females is 9.4 per 100,000 individuals per year, with a death rate of 6.6 per 100,000 individuals per year, and a 1.1% lifetime diagnosis risk.24 VOC-exposed personnel had a 1% higher mortality than controls.1
Symptomatology. Liver cancer is often asymptomatic and appears in late stages.25 Common symptoms include right upper quadrant pain, early satiety, nausea, vomiting, loss of appetite, weight loss, ascites, jaundice, and abnormal bleeding or bruising.25,26
Diagnostics. Diagnostic tests may include an ultrasound, CT, or MRI. Additional laboratory testing may include liver function, a-fetoprotein blood, CBC, renal function, calcium, and hepatitis panel screening for hepatitis B and C.27,28
Lung Cancer
The US incidence rate of lung cancer for both males and females is 47.8 per 100,000 individuals per year, with a death rate of 31.5 per 100,000 individuals per year, and a 5.4% lifetime diagnosis risk.29 VOC-exposed personnel had a 16% increased risk and 19% higher mortality.1,7 Subtypes include large cell, small cell, non-small cell, squamous cell, and adenocarcinoma.7 Smoking is an additional risk factor.30
Symptomatology. Symptoms of lung cancer include cough, shortness of breath, chest pain worse with deep breathing, unexplained weight loss, fatigue, night sweats, and recurrent fevers. Advanced stages may metastasize or spread to the liver, bones, and brain.31
Diagnostics. Low-dose CT and chest X-ray are used for screening.32
Breast Cancer
The US incidence rate of female breast cancer is 130.8 per 100,000 individuals per year, with a death rate of 19.2 per 100,000 individuals per year, and a 13.0% lifetime risk of diagnosis.33 For female VOC-exposed personnel, there was an equal risk of developing breast cancer as the control group.1 However, exposed females at Camp Lejeune had a 23% higher mortality risk compared to the control group.7 Breast cancer subtypes among females include ductal carcinoma, lobular carcinoma, and ductal-lobular carcinoma.1
The US incidence rate of male breast cancer is 1.3 per 100,000 individuals per year, with a death rate of 0.3 per 100,000 individuals per year.34,35 The lifetime risk for males developing breast cancer is 137.7 per 100,000 and about 70 to 100 times less common in men than women.36
Male personnel exposed at Camp Lejeune had a 4% increased risk for developing breast cancer compared to Camp Pendleton.7 However, mortality was lower in the Camp Lejeune group.1 Although male breast cancer is rare, males at Camp Lejeune had a higher incidence, indicating a link between TCE, PCE, vinyl chloride exposures and male breast cancer.37 Male breast cancer is more often diagnosed in advanced stages than female breast cancer due to the lack of awareness or absence of routine screenings.38 The most common breast cancer type in males is invasive ductal carcinoma, accounting for 85% to 90% of cases; lobular carcinoma is the second most common type.39
Symptomatology. In both females and males, breast cancer symptoms include painless, firm mass or lump in the breast (left breast slightly more common than right), skin changes or dimpling, nipple retraction or turning inward, and nipple discharge. Breast cancer can spread to the lymph nodes and can be appreciated in axilla or clavicular regions.40
Diagnostics. The diagnostic evaluation for breast cancer is similar for females and males. It includes a clinical breast examination, diagnostic mammogram, and ultrasound.41 Mammograms can distinguish between gynecomastia and cancer, especially in males.42 A core or fine needle biopsy is needed to confirm diagnosis.41
Adult Leukemia
The US incidence rate of leukemia for both male and female was 14.4 per 100,000 individuals per year, with a death rate of 5.8 per 100,000 individuals per year, and a 1.5% lifetime diagnosis risk.43
VOC-exposed personnel had a 7% higher risk of developing leukemia and a 13% increased mortality risk compared with the control group.1,7 Subtypes of leukemia at risk included a 38% increased incidence of acute myeloid/monocytic leukemia (AML) and a 2% increased incidence of chronic lymphocytic leukemia (CLL).1 Benzene and TCE exposures are known risk factors for AML and other leukemias.7 Personnel at Camp Lejeune had 3 times the incidence or mortality for leukemia, specifically AML mortality at 20%.7 Smoking is an additional risk factor for certain leukemias, especially AML.30
Symptomatology. Symptoms associated with leukemia are often nonspecific and may include fatigue, pallor, easy bruising or bleeding (skin or gums), recurrent infections secondary to neutropenia, fever, night sweats, pain or feeling full after a small meal due to enlarged spleen or liver, and weight loss.44,45
Diagnostics. An initial screening includes a CBC with differential, a peripheral smear to detect the presence of blast cells, as well as Auer rods in myeloid blast cells in AML or smudge cells in CLL. Confirmatory tests may include bone marrow biopsy or flow cytometry. A referral to a hematologist is recommended for any suspected leukemia.46,47
Myelodysplastic Syndromes
Aplastic anemia and MDS are considered rare disorders.48 Aplastic anemia is a nonmalignant bone marrow failure disorder with pancytopenia and hypocellular bone marrow due to the loss of hematopoietic stem cells.48 MDS is a type of hematopoietic cancer where the bone marrow produces abnormal blood cells or does not make enough healthy cells.49 This can lead to an increased risk for infection, cytopenias, neutropenia, refractory anemia, and thrombocytopenia, and progression to AML in some patients.49
The reported US incidence of MDS from 1975 to 2013 was 6.7 per 100,000 for males and 3.7 per 100,000 for females.50 Benzene exposure is linked to MDS and a known cause of AML.1 VOC-exposed personnel had a 68% increased risk of developing MDS and a 2.3-fold increased mortality risk compared to controls.1,7
Symptomatology. Some patients are asymptomatic at diagnosis.51 Symptoms related to cytopenia include fatigue, pallor, purpura, petechiae, bleeding of skin, gum, or nose, recurrent infections, fever, bone pain, loss of appetite, and weight loss.50,51
Diagnostics. Initial workup includes a CBC with differential to assess for anemia, white blood cell and absolute neutrophil counts (low), and thrombocytopenia.52 A peripheral blood smear may show myeloid blast cells. A bone marrow aspiration and biopsy, flow cytometry, and cytogenetic or molecular testing may be performed. If MDS is suspected, a referral to a hematologist should be considered.52
Multiple Myeloma
The US incidence rate of multiple myeloma for both males and females is 7.3 per 100,000 individuals per year, with a mortality rate of 2.9 per 100,000 individuals per year, and a 0.8% lifetime diagnosis risk.53 VOC-exposed personnel had a 13% increased risk of developing multiple myeloma and an 8% increased mortality risk compared to unexposed personnel.1,7
Symptomatology. Multiple myeloma may be asymptomatic in early stages. The most common presenting symptom is bone pain, especially in the back, hips, and long bones, due to hypercalcemia from increased reabsorption, plasma cell tumor overgrowth in the bone marrow, and lytic lesions.54 Additional symptoms include fatigue and pallor related to anemia, leukopenia, thrombocytopenia, recurrent infections, extreme thirst, frequent urination, dehydration, confusion associated with hypercalcemia, peripheral neuropathy, loss of appetite, weight loss, and renal impairment or failure.54
Diagnostics. Testing considerations include a CBC with a peripheral blood smear to evaluate anemia and rouleaux formation of red blood cells (seen in > 50% of patients with multiple myeloma), comprehensive metabolic panel (CMP) to assess kidney function, calcium levels (elevated), serum and urine protein electrophoresis with immunofixation to detect monoclonal protein (detected in > 80% of patients with multiple myeloma) and Bence-Jones proteins, serum free light chain assay, and a bone marrow biopsy for diagnosis.55,56
MRI of the spine and pelvis is the most sensitive to detecting bone marrow involvement and focal lesions before lytic lesion progression occurs and for assessing spinal cord compression.57 PET/CT is more sensitive at detecting extramedullary disease, outside of the spine, and for patients that cannot undergo MRI.57 A whole-body low-dose CT, either alone or with PET, is more sensitive than an X-ray at detecting lytic lesions, fractures, or osteoporosis associated with multiple myeloma.57
Non-Hodgkin Lymphoma
The US incidence rate of NHL for both males and females are 18.7 per 100,000 individuals per year, the death rate is 4.9 per 100,000 individuals per year, and a 2% lifetime diagnosis risk.58 VOC-exposed personnel had a 1% higher risk of developing NHL and a decreased mortality risk compared to the control group.1,7 Specific NHL subtypes with increased risk in the exposed cohort are mantle cell (26%), follicular (7%), Burkitt (53%), and marginal zone B-cell (45%).7
Symptomatology. NHL often presents with painless lymphadenopathy or enlarged lymph nodes involving the cervical, axillary, inguinal regions.59,60 Other symptoms include frequent infections, unexplained bruising, weight loss, and “B symptoms,” such as fever and night sweats.59,60 Some patients develop a mediastinal mass in the thorax, which if large may lead to cough or shortness of breath.59
Diagnostics. The initial diagnostic workup includes CBC with differential and LDH, which may be elevated.60,61 Imaging may begin with a chest X-ray to assess for a mediastinal mass; however, CTs of the chest, abdomen, and pelvis provide more detail to better assess for NHL. Whole body PET/CT is considered the gold standard for assessing and staging systemic involvement. If enlarged lymph nodes are present, a biopsy can confirm the subtype of NHL.60,61
PHYSICAL EXAMINATION
A focused physical examination may aid HCPs in early detection of the cancers associated with Camp Lejeune (Table 2). The physical examination can guide diagnostic testing and imaging for further assessment and workup for VOC-related cancers.

Proposed Diagnostic Screening Panel
Primary care and internal medicine HCPs have the opportunity to improve patient health outcomes by implementing a targeted diagnostic screening panel for identified veterans previously stationed at Camp Lejeune. Early identification of cancers associated with VOCs exposure can facilitate earlier treatment interventions and improve health and quality of life outcomes. The following diagnostic screening panel outlines a potential cost-effective strategy for evaluating and detecting the 10 cancers associated with VOC exposure in Camp Lejeune water.
Baseline Screening
Implementing a diagnostic screening panel in this high-risk cohort can lead to earlier diagnosis, reduce mortality, and improve patient outcomes through early intervention, which in turn may result in less invasive treatment. This approach may also reduce health care costs by avoiding costs associated with delayed diagnosis and advanced-stage cancer care (Tables 3 and 4).


A baseline panel of tests for exposed veterans could include:
- A CBC with differential and peripheral smear to assess for anemia, leukemia, thrombocytopenia, and blast cells associated with leukemias, MDS, multiple myeloma, and NHL.19,46,47,52,55,56,60,61
- CMP evaluates calcium, total protein, renal and liver renal function. Elevated test results may indicate kidney or liver cancer or multiple myeloma.19,27,28,55,56
- LDH testing may reveal levels that are elevated from tissue damage or high cell turnover in kidney cancer, multiple myeloma, and NHL.19,55,56,60,61
- Urinalysis with microscopy may detect hematuria, proteinuria and cellular casts in bladder and kidney cancers.13,24,19
- Low-dose CTs of the chest, abdomen, and pelvis are recommended for early identification of any masses or lymphadenopathy in lung, kidney, liver cancers, and NHL.19,27,28,32,60,61
COST EFFICIENCY
Screening Panel Cost
According to the Medicare Clinical Laboratory Fee Schedule payment cap for 2018, the mean cost for the proposed blood workup was $35 (CBC, $10; CMP, $13; LDH, $8; urinalysis, $4).62 Medicare procedure price schedule for 2025 includes $351 for a CT of the abdomen and pelvis with and without contrast (Current Procedural Terminology [CPT] code 74177) and $187 for a CT of the chest with and without contrast (CPT code 71270).63,64 The total proposed diagnostic screening panel payment cost about $572.
Cancer Care Cost
The average cost for initial cancer care across all cancer sites from 2007 to 2013 was $43,516 per patient; Camp Lejeune-associated cancers ranged from $26,443 for bladder cancer to $89,947 for esophageal cancer care.64 Further, the last year of life cost across all cancer sites averaged $109,727, and Camp Lejeune-associated cancer types ranged from $76,101 for breast cancer to $169,588 for leukemia.65
CONCLUSIONS
From 1953 to 1985, up to 1 million military personnel, civilian workers, and their families stationed at Camp Lejeune were unknowingly exposed to toxic and carcinogenic VOCs, which are associated with = 10 cancers, including bladder, kidney, esophageal, liver, lung, breast, and hematologic malignancies.1-4 Some veterans may be asymptomatic, whereas others present with subtle or specific symptoms that can vary by individual and the type and stage of cancer. HCPs have an opportunity to improve patient outcomes through awareness in identifying symptoms associated with Camp Lejeune water exposure and performing a thorough baseline physical examination, especially noting lymphadenopathy, unexplained weight loss, or masses, which can guide further diagnostic evaluation. Timely screening can identify cancers earlier, reducing delays in care, mitigating the cost burden associated with advanced-stage cancer treatment, improving survival outcomes, and enhancing quality of life. Primary care and internal medicine HCPs specifically play a crucial role in early recognition, physical assessment, and appropriate screening tools. A proposed panel includes CBC with differential and peripheral smear, CMP, LDH, urinalysis, and low-dose CTs of the chest, abdomen and pelvis. Implementation should be guided by clinical judgment and patient-specific risk factors. The proposed diagnostic screening panel is a small price to pay for those who served in any capacity at Camp Lejeune.
- Bove FJ, Greek A, Gatiba R, et al. Cancer incidence among Marines and Navy personnel and civilian workers exposed to industrial solvents in drinking water at US Marine Corps Base Camp Lejeune: a cohort study. Environ Health Perspect. 2024;132:107008. doi:10.1289/EHP14966
- Maslia ML, Aral MM, Ruckart PZ, Bove FJ. Reconstructing historical VOC concentrations in drinking water for epidemiological studies at a US military base: summary of results. Water (Basel). 2016;8:449. doi:10.3390/w8100449
- Rosenfeld PE, Spaeth KR, McCarthy SJ, et al. Camp Lejeune Marine cancer risk assessment for exposure to contaminated drinking water from 1955 to 1987. Water Air Soil Pollut. 2024;235(2). doi:10.1007/s11270-023-06863-y
- US Department of Veterans Affairs, Veterans Health Administration. Camp Lejeune: past water contamination. Updated April 15, 2025. Accessed March 3, 2026. https://www.publichealth.va.gov/exposures/camp-lejeune/
- Jung K, Khan A, Mocharnuk R, et al. Clinical encounter with three cancer patients affected by groundwater contamination at Camp Lejeune: a case series and review of the literature. J Med Case Rep. 2022;16(1):272. doi:10.1186/s13256-022-03501-9
- Honoring America's Veterans and Caring for Camp Lejeune Familes Act of 2012, Pub L No. 112-154. Janey Ensminger Act. Congress.gov. Accessed April 15, 2026. https://ww.congress.gov/bill/112th-congress/house-bill/1627
- Bove FJ, Greek A, Gatiba R, et al. Evaluation of mortality among Marines, Navy personnel, and civilian workers exposed to contaminated drinking water at USMC Base Camp Lejeune: a cohort study. Environ Health. 2024;23(1):61. doi:10.1186/s12940-024-01099-7
- Honoring our PACT Act of 2022 (Pub L No. 117-168): expansion of health care eligibility and toxic exposure screenings. Congress.gov. Accessed March 3, 2026. https://www.congress.gov/bill/117th-congress/house-bill/3967
- Ensuring Justice for Camp Lejeune Victims Act of 2025. Congress.gov. Accessed March 24, 2026. https://www.congress.gov/bill/119th-congress/house-bill/4145
- SEER. Cancer stat facts: bladder cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/urinb.html
- Agency for Toxic Substances and Disease Registry. ATSDR assessment of the evidence for the drinking water contaminants at Camp Lejeune and specific cancers and other diseases. Published January 13, 2017. Accessed March 3, 2026. https://www.atsdr.cdc.gov/camp-lejeune/media/pdfs/2024/10/ATSDR_summary_of_the_evidence_for_causality_TCE_PCE_508.pdf
- National Cancer Institute. What is bladder cancer? Updated February 16, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder
- National Cancer Institute. Bladder cancer symptoms. Updated February 16, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder/symptoms
- American Cancer Society. Bladder cancer signs and symptoms. Updated March 12, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/bladder-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- National Cancer Institute. Bladder cancer screening. Updated April 27, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder/screening
- American Cancer Society. Tests for bladder cancer. Updated March 12, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/bladder-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: kidney and renal pelvis cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/kidrp.html
- American Cancer Society. Kidney cancer signs and symptoms. Updated May 1, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/kidney-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- American Cancer Society. Tests for kidney cancer. Updated May 1, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/kidney-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: esophageal cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/esoph.html
- Engel LS, Chow WH, Vaughan TL, et al. Population attributable risks of esophageal and gastric cancers.
J Natl Cancer Inst. 2003;95(18):1404-1413. doi:10.1093/jnci/djg047 - American Cancer Society. Signs and symptoms of esophageal cancer. Updated March 20, 2020. Accessed March 3, 2026. https://www.cancer.org/cancer/types/esophagus-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- American Cancer Society. Tests for esophageal cancer. Updated March 20, 2020. Accessed March 3, 2026. https://www.cancer.org/cancer/types/esophagus-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: liver and intrahepatic bile duct cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/livibd.html
- American Cancer Society. Signs and symptoms of liver cancer. Updated February 11, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/liver-cancer/detection-diagnosis-staging/signs-symptoms.html
- National Cancer Institute. What is liver cancer? Updated May 15, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/liver/what-is-liver-cancer
- American Cancer Society. Tests for liver cancer. Updated February 11, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/liver-cancer/detection-diagnosis-staging/how-diagnosed.html
- National Cancer Institute. Liver cancer screening. Updated May 15, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/liver/what-is-liver-cancer/screening
- SEER. Cancer stat facts: lung cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/lungb.html
- US Department of Health and Human Services. The Health Consequences of Smoking: A Report of the Surgeon General. Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health; 2004. Accessed March 3, 2026. https://www.ncbi.nlm.nih.gov/books/NBK44695/pdf/Bookshelf_NBK44695.pdf
- American Cancer Society. Lung cancer signs and symptoms. Updated February 27, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/lung-cancer/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for lung cancer. Updated January 29, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/lung-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: female breast cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/breast.html
- SEER. SEER*Explorer breast incidence and mortality comparison. Updated July 2, 2025. Accessed March 3, 2026. https://seer.cancer.gov/statistics-network/explorer/application.html
- Susan G. Komen. Male breast cancer. Updated June 3, 2025. Accessed March 3, 2026. https://www.komen.org/breast-cancer/facts-statistics/male-breast-cancer/
- American Cancer Society. Key statistics for breast cancer in men. Updated January 16, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/about/key-statistics.html
- Ruckart PZ, Bove FJ, Shanley E 3rd, et al. Evaluation of contaminated drinking water and male breast cancer at Marine Corps Base Camp Lejeune, North Carolina: a case-control study. Environ Health. 2015;14:74. doi:10.1186/s12940-015-0061-4
- Culell P, Solernou L, Tarazona J, et al. Male breast cancer: a multicentric study. Breast J. 2007;13:213-215. doi:10.1111/j.1524-4741.2007.00412.x
- Giordano SH, Cohen DS, Buzdar AU, et al. Breast carcinoma in men: a population-based study. Cancer. 2004;101:51-57. doi:10.1002/cncr.20312
- American Cancer Society. Signs and symptoms of breast cancer in men. Updated April 27, 2018. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests to help diagnose breast cancer in men. Updated December 20, 2021. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/detection-diagnosis-staging/how-diagnosed.html
- Evans GF, Anthony T, Turnage RH, et al. The diagnostic accuracy of mammography in the evaluation of male breast disease. Am J Surg. 2001;181:96-100. doi:10.1016/S0002-9610(00)00571-7
- SEER. Cancer stat facts: leukemia. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/leuks.html
- American Cancer Society. Signs and symptoms of acute myeloid leukemia (AML). Updated March 20, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/acute-myeloid-leukemia/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Signs and symptoms of chronic lymphocytic leukemia (CLL). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/chronic-lymphocytic-leukemia/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for acute myeloid leukemia (AML). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/acute-myeloid-leukemia/detection-diagnosis-staging/how-diagnosed.html
- American Cancer Society. Tests for chronic lymphocytic leukemia (CLL). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/chronic-lymphocytic-leukemia/detection-diagnosis-staging/how-diagnosed.html
- Giudice V, Selleri C. Aplastic anemia: pathophysiology. Semin Hematol. 2022;59:13-20. doi:10.1053/j.seminhematol.2021.12.002
- National Cancer Institute. Myelodysplastic syndromes treatment (PDQ®)–patient version. Updated October 4, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/myeloproliferative/patient/myelodysplastic-treatment-pdq
- SEER. Cancer statistics review (CSR) 1975-2013: myelodysplastic syndromes. Accessed March 3, 2026. https://seer.cancer.gov/archive/csr/1975_2013/browse_csr.php?sectionSEL=30&pageSEL=sect_30_table.01
- American Cancer Society. Signs and symptoms of myelodysplastic syndrome (MDS). Updated November 21, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/myelodysplastic-syndrome/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for myelodysplastic syndromes (MDS). Updated November 21, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/myelodysplastic-syndrome/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: myeloma. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/mulmy.html
- American Cancer Society. Signs and symptoms of multiple myeloma. Updated February 28, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/multiple-myeloma/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for multiple myeloma. Updated February 28, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/multiple-myeloma/detection-diagnosis-staging/testing.html
- Laubach JP. Multiple myeloma: clinical features, laboratory manifestations, and diagnosis. UpToDate. Updated April 28, 2025. Accessed March 3, 2026. https://www.uptodate.com/contents/multiple-myeloma-clinical-features-laboratory-manifestations-and-diagnosis
- Hillengass J, Usmani S, Rajkumar SV, et al. International Myeloma Working Group consensus recommendations on imaging in monoclonal plasma cell disorders. Lancet Oncol. 2019;20:e302-e312. doi:10.1016/S1470-2045(19)30309-2
- SEER. Cancer stat facts: non-Hodgkin lymphoma. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/nhl.html
- American Cancer Society. Signs and symptoms of non-Hodgkin lymphoma. Updated February 15, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/non-hodgkin-lymphoma/detection-diagnosis-staging/signs-symptoms.html
- National Cancer Institute. Non-Hodgkin lymphoma treatment (PDQ®)–patient version. Updated August 22, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/lymphoma/patient/adult-nhl-treatment-pdq
- American Cancer Society. Tests for non-Hodgkin lymphoma. Updated February 15, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/non-hodgkin-lymphoma/detection-diagnosis-staging/how-diagnosed.html
- College of American Pathologists. Medicare clinical laboratory fee schedule. Published November 2017. Accessed March 3, 2026. https://documents.cap.org/documents/2018-final-medicare-clfs-rates.pdf
- Medicare.gov. Procedure price lookup for outpatient services. Accessed March 3, 2026. https://www.medicare.gov/procedure-price-lookup/cost/71270/
- Medicare.gov. Procedure price lookup for outpatient services. Accessed March 3, 2026. https://www.medicare.gov/procedure-price-lookup/cost/74177/
- National Cancer Institute. Cancer trends progress report: financial burden of cancer care. Updated April 2025. Accessed March 3, 2026. https://progressreport.cancer.gov/after/economic_burden
- Bove FJ, Greek A, Gatiba R, et al. Cancer incidence among Marines and Navy personnel and civilian workers exposed to industrial solvents in drinking water at US Marine Corps Base Camp Lejeune: a cohort study. Environ Health Perspect. 2024;132:107008. doi:10.1289/EHP14966
- Maslia ML, Aral MM, Ruckart PZ, Bove FJ. Reconstructing historical VOC concentrations in drinking water for epidemiological studies at a US military base: summary of results. Water (Basel). 2016;8:449. doi:10.3390/w8100449
- Rosenfeld PE, Spaeth KR, McCarthy SJ, et al. Camp Lejeune Marine cancer risk assessment for exposure to contaminated drinking water from 1955 to 1987. Water Air Soil Pollut. 2024;235(2). doi:10.1007/s11270-023-06863-y
- US Department of Veterans Affairs, Veterans Health Administration. Camp Lejeune: past water contamination. Updated April 15, 2025. Accessed March 3, 2026. https://www.publichealth.va.gov/exposures/camp-lejeune/
- Jung K, Khan A, Mocharnuk R, et al. Clinical encounter with three cancer patients affected by groundwater contamination at Camp Lejeune: a case series and review of the literature. J Med Case Rep. 2022;16(1):272. doi:10.1186/s13256-022-03501-9
- Honoring America's Veterans and Caring for Camp Lejeune Familes Act of 2012, Pub L No. 112-154. Janey Ensminger Act. Congress.gov. Accessed April 15, 2026. https://ww.congress.gov/bill/112th-congress/house-bill/1627
- Bove FJ, Greek A, Gatiba R, et al. Evaluation of mortality among Marines, Navy personnel, and civilian workers exposed to contaminated drinking water at USMC Base Camp Lejeune: a cohort study. Environ Health. 2024;23(1):61. doi:10.1186/s12940-024-01099-7
- Honoring our PACT Act of 2022 (Pub L No. 117-168): expansion of health care eligibility and toxic exposure screenings. Congress.gov. Accessed March 3, 2026. https://www.congress.gov/bill/117th-congress/house-bill/3967
- Ensuring Justice for Camp Lejeune Victims Act of 2025. Congress.gov. Accessed March 24, 2026. https://www.congress.gov/bill/119th-congress/house-bill/4145
- SEER. Cancer stat facts: bladder cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/urinb.html
- Agency for Toxic Substances and Disease Registry. ATSDR assessment of the evidence for the drinking water contaminants at Camp Lejeune and specific cancers and other diseases. Published January 13, 2017. Accessed March 3, 2026. https://www.atsdr.cdc.gov/camp-lejeune/media/pdfs/2024/10/ATSDR_summary_of_the_evidence_for_causality_TCE_PCE_508.pdf
- National Cancer Institute. What is bladder cancer? Updated February 16, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder
- National Cancer Institute. Bladder cancer symptoms. Updated February 16, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder/symptoms
- American Cancer Society. Bladder cancer signs and symptoms. Updated March 12, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/bladder-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- National Cancer Institute. Bladder cancer screening. Updated April 27, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder/screening
- American Cancer Society. Tests for bladder cancer. Updated March 12, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/bladder-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: kidney and renal pelvis cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/kidrp.html
- American Cancer Society. Kidney cancer signs and symptoms. Updated May 1, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/kidney-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- American Cancer Society. Tests for kidney cancer. Updated May 1, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/kidney-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: esophageal cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/esoph.html
- Engel LS, Chow WH, Vaughan TL, et al. Population attributable risks of esophageal and gastric cancers.
J Natl Cancer Inst. 2003;95(18):1404-1413. doi:10.1093/jnci/djg047 - American Cancer Society. Signs and symptoms of esophageal cancer. Updated March 20, 2020. Accessed March 3, 2026. https://www.cancer.org/cancer/types/esophagus-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- American Cancer Society. Tests for esophageal cancer. Updated March 20, 2020. Accessed March 3, 2026. https://www.cancer.org/cancer/types/esophagus-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: liver and intrahepatic bile duct cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/livibd.html
- American Cancer Society. Signs and symptoms of liver cancer. Updated February 11, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/liver-cancer/detection-diagnosis-staging/signs-symptoms.html
- National Cancer Institute. What is liver cancer? Updated May 15, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/liver/what-is-liver-cancer
- American Cancer Society. Tests for liver cancer. Updated February 11, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/liver-cancer/detection-diagnosis-staging/how-diagnosed.html
- National Cancer Institute. Liver cancer screening. Updated May 15, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/liver/what-is-liver-cancer/screening
- SEER. Cancer stat facts: lung cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/lungb.html
- US Department of Health and Human Services. The Health Consequences of Smoking: A Report of the Surgeon General. Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health; 2004. Accessed March 3, 2026. https://www.ncbi.nlm.nih.gov/books/NBK44695/pdf/Bookshelf_NBK44695.pdf
- American Cancer Society. Lung cancer signs and symptoms. Updated February 27, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/lung-cancer/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for lung cancer. Updated January 29, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/lung-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: female breast cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/breast.html
- SEER. SEER*Explorer breast incidence and mortality comparison. Updated July 2, 2025. Accessed March 3, 2026. https://seer.cancer.gov/statistics-network/explorer/application.html
- Susan G. Komen. Male breast cancer. Updated June 3, 2025. Accessed March 3, 2026. https://www.komen.org/breast-cancer/facts-statistics/male-breast-cancer/
- American Cancer Society. Key statistics for breast cancer in men. Updated January 16, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/about/key-statistics.html
- Ruckart PZ, Bove FJ, Shanley E 3rd, et al. Evaluation of contaminated drinking water and male breast cancer at Marine Corps Base Camp Lejeune, North Carolina: a case-control study. Environ Health. 2015;14:74. doi:10.1186/s12940-015-0061-4
- Culell P, Solernou L, Tarazona J, et al. Male breast cancer: a multicentric study. Breast J. 2007;13:213-215. doi:10.1111/j.1524-4741.2007.00412.x
- Giordano SH, Cohen DS, Buzdar AU, et al. Breast carcinoma in men: a population-based study. Cancer. 2004;101:51-57. doi:10.1002/cncr.20312
- American Cancer Society. Signs and symptoms of breast cancer in men. Updated April 27, 2018. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests to help diagnose breast cancer in men. Updated December 20, 2021. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/detection-diagnosis-staging/how-diagnosed.html
- Evans GF, Anthony T, Turnage RH, et al. The diagnostic accuracy of mammography in the evaluation of male breast disease. Am J Surg. 2001;181:96-100. doi:10.1016/S0002-9610(00)00571-7
- SEER. Cancer stat facts: leukemia. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/leuks.html
- American Cancer Society. Signs and symptoms of acute myeloid leukemia (AML). Updated March 20, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/acute-myeloid-leukemia/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Signs and symptoms of chronic lymphocytic leukemia (CLL). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/chronic-lymphocytic-leukemia/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for acute myeloid leukemia (AML). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/acute-myeloid-leukemia/detection-diagnosis-staging/how-diagnosed.html
- American Cancer Society. Tests for chronic lymphocytic leukemia (CLL). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/chronic-lymphocytic-leukemia/detection-diagnosis-staging/how-diagnosed.html
- Giudice V, Selleri C. Aplastic anemia: pathophysiology. Semin Hematol. 2022;59:13-20. doi:10.1053/j.seminhematol.2021.12.002
- National Cancer Institute. Myelodysplastic syndromes treatment (PDQ®)–patient version. Updated October 4, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/myeloproliferative/patient/myelodysplastic-treatment-pdq
- SEER. Cancer statistics review (CSR) 1975-2013: myelodysplastic syndromes. Accessed March 3, 2026. https://seer.cancer.gov/archive/csr/1975_2013/browse_csr.php?sectionSEL=30&pageSEL=sect_30_table.01
- American Cancer Society. Signs and symptoms of myelodysplastic syndrome (MDS). Updated November 21, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/myelodysplastic-syndrome/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for myelodysplastic syndromes (MDS). Updated November 21, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/myelodysplastic-syndrome/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: myeloma. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/mulmy.html
- American Cancer Society. Signs and symptoms of multiple myeloma. Updated February 28, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/multiple-myeloma/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for multiple myeloma. Updated February 28, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/multiple-myeloma/detection-diagnosis-staging/testing.html
- Laubach JP. Multiple myeloma: clinical features, laboratory manifestations, and diagnosis. UpToDate. Updated April 28, 2025. Accessed March 3, 2026. https://www.uptodate.com/contents/multiple-myeloma-clinical-features-laboratory-manifestations-and-diagnosis
- Hillengass J, Usmani S, Rajkumar SV, et al. International Myeloma Working Group consensus recommendations on imaging in monoclonal plasma cell disorders. Lancet Oncol. 2019;20:e302-e312. doi:10.1016/S1470-2045(19)30309-2
- SEER. Cancer stat facts: non-Hodgkin lymphoma. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/nhl.html
- American Cancer Society. Signs and symptoms of non-Hodgkin lymphoma. Updated February 15, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/non-hodgkin-lymphoma/detection-diagnosis-staging/signs-symptoms.html
- National Cancer Institute. Non-Hodgkin lymphoma treatment (PDQ®)–patient version. Updated August 22, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/lymphoma/patient/adult-nhl-treatment-pdq
- American Cancer Society. Tests for non-Hodgkin lymphoma. Updated February 15, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/non-hodgkin-lymphoma/detection-diagnosis-staging/how-diagnosed.html
- College of American Pathologists. Medicare clinical laboratory fee schedule. Published November 2017. Accessed March 3, 2026. https://documents.cap.org/documents/2018-final-medicare-clfs-rates.pdf
- Medicare.gov. Procedure price lookup for outpatient services. Accessed March 3, 2026. https://www.medicare.gov/procedure-price-lookup/cost/71270/
- Medicare.gov. Procedure price lookup for outpatient services. Accessed March 3, 2026. https://www.medicare.gov/procedure-price-lookup/cost/74177/
- National Cancer Institute. Cancer trends progress report: financial burden of cancer care. Updated April 2025. Accessed March 3, 2026. https://progressreport.cancer.gov/after/economic_burden
Optimizing Care for Veterans at Risk of Cancer From Camp Lejeune Water Exposure
Optimizing Care for Veterans at Risk of Cancer From Camp Lejeune Water Exposure
Early Outcomes of Stereotactic Body Radiotherapy for Localized Prostate Cancer: A Retrospective Analysis
Early Outcomes of Stereotactic Body Radiotherapy for Localized Prostate Cancer: A Retrospective Analysis
Prostate cancer is the most common cancer in US males, with an estimated 313,780 new cases and 35,770 deaths in 2025.1 Several treatment options are available for localized prostate cancer that have similar outcomes, including active surveillance for low-risk cancers, surgery, or radiotherapy.2,3 Conventional fractionation radiotherapy (CFRT) with 40 to 45 fractions over 8 to 9 weeks has been used for decades. Over the past 2 decades, moderate hypofractionation schedules with 2.4 to 3.4 Gy per fraction over 20 to 28 fractions have become standard, as many noninferiority randomized clinical trials (RCTs) such as CHHiP (UK),4 PROFIT (Canada and Europe),5 NRG Oncology RTOG 0415 (US),6 HYPRO (Netherlands),7,8 and HYPO-RT-PC (Sweden and Denmark),9 have shown the noninferiority of moderately hypofractionated radiotherapy compared with CFRT. Notably, most of these noninferiority studies primarily included patients with low- or intermediate-risk prostate cancer, except for the HYPO-RT-PC trial,9 which also included patients with intermediate- and high-risk prostate cancer.
These noninferiority studies, along with technological advances in radiotherapy, such as intensity-modulated radiotherapy (IMRT), volumetric modulated arc therapy (VMAT), and image-guided radiotherapy (IGRT), paved the path to ultrahypofractionated stereotactic body radiotherapy (SBRT) that is delivered in 5 fractions of ≥ 6 Gy. This high dose per fraction may have a radiobiologic advantage over conventional fractionation. The relatively low a/ß ratio of prostate cancer, estimated to be between 1 and 2, suggests that tumor cells may be particularly sensitive to the high doses per fraction delivered in SBRT.10-13 Compared with CFRT, SBRT-induced tumor cell death may also be mediated through different pathways; this pathway appears to be generated in a dose-dependent manner, particularly with doses > 8 Gy per fraction.14,15 Additionally, the higher a/ß ratio for the surrounding organs at risk, such as the bladder and rectum, theoretically allows for an improved therapeutic ratio window that maximizes tumor control while minimizing damage to healthy tissues.
A substantial body of evidence from prospective studies and meta-analyses supports the use of SBRT for localized prostate cancer. HYPO-RT-PC, a significant phase 3 noninferiority study, enrolled 1200 patients with intermediate (89%) and high-risk (11%) prostate cancer randomized between 2 arms, including CFRT to 78 Gy in 39 fractions and SBRT to 42.7 Gy in 7 fractions, treated 3 days weekly. After a median follow-up of 60 months, the estimated 5-year biochemical relapse-free survival rate was 84% in both groups.9 This trial was notable because it was the first randomized study to demonstrate that SBRT was noninferior to CFRT in intermediate- and high-risk prostate cancer patients. Another pivotal phase 3 trial, the PACE-B study, enrolled 874 patients to compare SBRT (36.25 Gy to the prostate gland, with a secondary dose of 40 Gy to the gross tumor volume where applicable, in 5 fractions) with CFRT (78 Gy in 39 fractions) and moderately hypofractionated radiotherapy (HFRT) (62 Gy in 20 fractions) in patients with low- or intermediate-risk prostate cancer. With a 74-month median follow-up, the study reported 5-year biochemical free rates of 94.6% for CFRT and 95.8% for SBRT, confirming the noninferiority of SBRT to CFRT.15
SBRT offers short, effective, and convenient treatment to many patients with localized prostate cancer. While previous guidelines were more restrictive, the March 2026 National Comprehensive Cancer Network (NCCN) guidelines now list SBRT as a preferred treatment modality for high-risk prostate cancer.16
Given the growing body of evidence supporting the efficacy and safety of SBRT, we implemented an SBRT program in 2014 at a tertiary care center for veterans. This retrospective study was undertaken to evaluate the early efficacy and toxicity of SBRT in patients with localized prostate cancer treated at our institution, including patients across all risk stratifications.
METHODS
We identified 242 patients diagnosed with prostate cancer who underwent SBRT treatment between November 2014 and October 2024 at Overland Park Veterans Affairs Radiation Oncology Clinic. For the final analysis, 46 patients with < 2 years of follow-up and 22 patients who died from causes other than prostate cancer were excluded, resulting in a cohort of 174 patients with ≥ 24-month follow-up.
Treatment
Patients eligible for staging underwent imaging according to NCCN guidelines, including computed tomography (CT) of the abdomen and pelvis, bone scintigraphy, or, in recent years, prostate-specific membrane antigen positron emission tomography, primarily used for unfavorable intermediate-risk (UIR) and high-risk (HR) cancers. Patients with a negative staging work-up for nodal or skeletal disease were included. Prior to planning the CT simulation, patients were given bowel preparation instructions, including a low-fiber and low-gas-producing diet, simethicone, and enemas, the night before and morning of the simulation. Patients were instructed to arrive with a comfortably full bladder, having not voided for 2 to 3 hours prior to the procedure. At Kansas City Veterans Affairs Medical Center (KCVAMC), SBRT treatment was generally restricted to patients with a baseline American Urological Association symptom score of 15 to 20 out of 35 and a prostate gland size < 80 mL to minimize the risk of acute urinary toxicity. We did not use intraprostatic fiducials, hydrogel rectal spacers, or intravenous contrast agents for planning CT simulation.
Patients were placed in a supine position, and a vacuum bag was used for immobilization. Following the CT simulation, the images were transferred to the Eclipse treatment planning system. The clinical target volume (CTV) encompassed the prostate and the proximal 1.0 cm of the seminal vesicles for Gleason score (GS) 1 to 2, and the entire seminal vesicle was included for GS 3 to 5, which is consistent with KCVAMC practice and established safety protocols. The planning target volume (PTV) was created by uniformly expanding the CTV by 5 to 7 mm, except for the posterior margin, which was limited to 3 to 5 mm. When elective nodal radiotherapy was planned for HR prostate cancer, the pelvic field for CT simulation started at the L-2 upper border, with the lower border extending to the lesser trochanter. The pelvic nodes were delineated per Radiation Therapy Oncology Group (RTOG) guidelines.17 The CTV nodes (CTVn), including common iliac, external and internal iliac nodes, obturator, and presacral nodes, were created by uniformly expanding the CTVn by 2 to 3 mm. Slice-by-slice corrections were made to avoid bowel overlap in these patients.
The use of androgen deprivation therapy (ADT) for a duration of 6 to 24 months was prescribed for patients with UIR or HR prostate cancer per NCCN guidelines.16 The prescribed dose to the PTV was 36.25 to 40 Gy (40 Gy was mostly used as a boost to the dominant lesion) in 5 fractions, with each fraction ranging from 7.25 to 8 Gy. For elective nodal radiotherapy in patients at HR, the prescribed dose was 25 Gy in 5 fractions. All patients were planned for VMAT, which aims to deliver ≥ 95% of the prescription dose to 95% of the PTV. Once the physician approved the treatment plan and physics quality assessment was completed, treatments commenced on an every-other-day schedule. Patients received the same bowel preparation instructions for each treatment as for the planning CT simulation. Daily treatment accuracy was confirmed via daily 3-dimensional cone-beam CT (CBCT) for IGRT. No fiducials or hydrogel rectal spacers were used.
Follow-up Schedule and Toxicity Assessment
Follow-up assessments were conducted 4 to 6 weeks after radiation therapy and then repeated every 6 months for 2 to 5 years, and annually thereafter. At each follow-up visit, patients were evaluated for genitourinary (GU) and gastrointestinal (GI) toxicity, according to RTOG toxicity criteria. Prostate-specific antigen (PSA) levels were monitored; in patients receiving ADT, testosterone levels were also checked.
Statistical Analysis
Biochemical failure was defined using the Phoenix definition (nadir PSA + 2 ng/mL). Differences between dose cohorts were assessed using the log-rank test for survival outcomes and X2 testing for categorical variables. GU and GI toxicities were summarized as cumulative incidences of RTOG grade ≥ II events. Statistical significance was set at P < .05.
RESULTS
One hundred seventy-four patients were included in the retrospective review. Patients had a median follow-up of 45 months (range, 24-111) (Figure). The median age at treatment was 74 years (range, 51-88), and the median pretreatment PSA level was 11.9 ng/mL (range, 0.6-69.5). Twenty-six patients (14.9%) had a GS 1, 77 (44.3%) had GS 2, 41 (23.6%) had GS 3, 18 (10.3%) had GS 4, and 12 (6.9%) had GS 5. Fifty-one patients (29.3%) received elective pelvic nodal radiotherapy, and 93 patients (53.4%) received ADT (Table 1).
At 24 months follow-up, 6 patients (3.4%) had biochemical failures. One patient died from metastatic prostate cancer, and 5 patients are living with biochemical failure (Table 2). The actuarial 5-year overall survival (OS) rate was 99.4%, and the 5-year disease-free survival (DFS) rate was 96.6%. We performed a subanalysis comparing outcomes of the 36.25 Gy vs 40 Gy SBRT cohorts. There was no statistically significant difference in DFS, OS, or the cumulative incidence of grade II/III toxicity between patients treated with 40 Gy vs 36.25 Gy. Outcomes stratified by NCCN risk groups (low, intermediate, high/very high) are detailed in Table 3. As expected, DFS was slightly lower in the high-risk group, but overall disease control remained high across all stratifications.
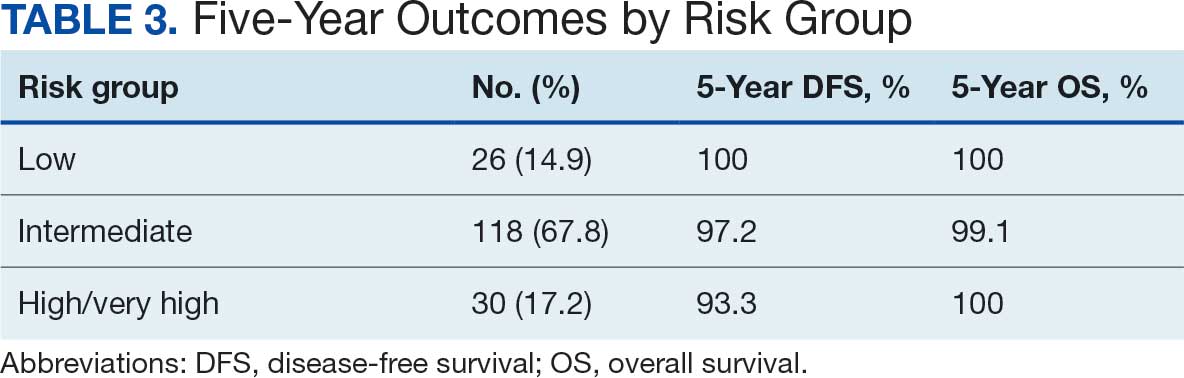
The cumulative incidence of RTOG grade II and higher GU toxicity was 28.2% (Table 4). This included 46 patients (26.4%) with grade II GU toxicity and 2 patients (1.2%) who developed grade III GU complications (1 requiring self-catheterization and another a suprapubic catheter for urinary retention). One patient (0.6%) treated with a 40 Gy dose regimen experienced a grade IV GU complication in the form of a rectovesical fistula necessitating surgical intervention.
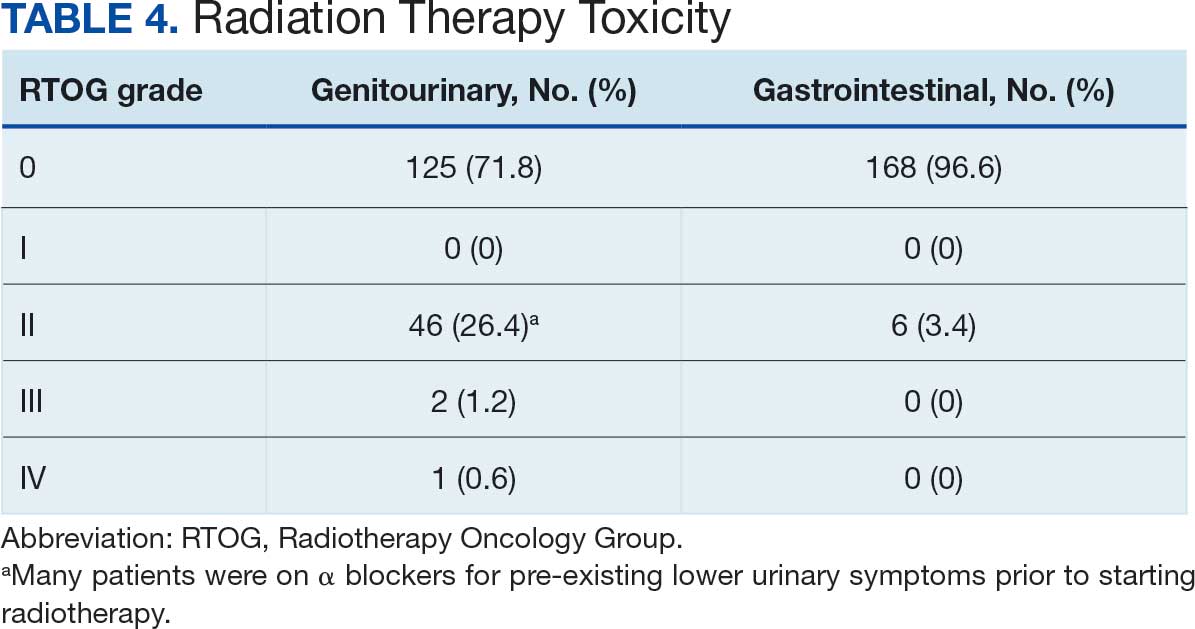
The cumulative incidence of RTOG grade II or higher GI toxicity was 3.4%, and no grade III or IV gastrointestinal toxicities were observed during the follow-up period. Importantly, intraprostatic fiducials, hydrogel rectal spacers, or intravenous contrast were not routinely used in this cohort of patients.
The high rates of actuarial 5-year DFS and OS observed suggest a favorable initial response to the SBRT regimen employed at KCVAMC. However, given the potential for late recurrence in patients with prostate cancer, longer follow-up is essential to determine the durability of these outcomes. The observed GU toxicity rate of 28.2% for grade II and higher events warrants careful consideration and compares with other published data on SBRT for prostate cancer.15 The occurrence of a grade IV rectovesical fistula, although rare, is a notable adverse event that warrants discussion in the context of the treatment approach. The low incidence of grade II or higher GI toxicity is an encouraging finding, particularly given that hydrogel rectal spacers are not routinely used to minimize rectal exposure.
DISCUSSION
The primary objective of this retrospective study was to evaluate the outcomes of SBRT for patients with localized prostate cancer treated at KCVAMC and to compare these results with those reported in the literature. Our findings demonstrate promising intermediate-term efficacy, with an estimated 5-year DFS of 96.6% and OS of 99.4% at a median follow-up of 45 months. Furthermore, the observed toxicity profile appears acceptable, with a cumulative grade II and higher GU toxicity rate of 28.2% and a grade II or higher GI toxicity rate of 3.4%. Notably, these outcomes were achieved without the routine use of intraprostatic fiducials or hydrogel rectal spacers.
Two pivotal randomized phase 3 trials have established the noninferiority of ultrahypofractionated radiotherapy (UHRT) with SBRT over conventional fractionation. The HYPO-RT-PC trial compared SBRT (42.7 Gy in 7 fractions) with conventional fractionation (78 Gy in 39 fractions) in intermediate- and high-risk patients with prostate cancer and reported a 5-year biochemical relapse-free survival of 84% in both arms.9 The PACE-B trial, which included patients at low- and intermediate-risk, compared SBRT (36.25 Gy in 5 fractions) with conventional or moderate HFRT and reported a 5-year biochemical control rate of 95.8% in the SBRT arm and 94.6% in the control arm.15
A comprehensive review and meta-analysis of 7 phase 3 studies involving 6795 patients compared different radiotherapy regimens, namely, UHRT, HFRT, and CFRT, and reported that after 5 years, the DFS rates were 85.1% for CFRT, 86% for HFRT, and 85% for UHRT, with no significant difference in toxicity among the 3 different treatment approaches.18 This suggests that shorter, more intense radiotherapy schedules (UHRT and HFRT) may be as effective and safe as traditional, longer courses of radiation.
There are multiple published nonrandomized prospective trials in which thousands of patients with extreme hypofractionation have been treated with different doses, fractions, and techniques. While heterogeneity and limited long-term follow-up in the existing evidence are acknowledged, these data suggest that prostate SBRT provides appropriate biochemical control with few high-grade toxicities, supporting its ongoing global use and justifying further prospective investigations. Comparative data are shown in Table 5. Several ongoing studies are evaluating noninferiority, superiority, and cost-effectiveness using different methodologies (Table 6).9,15,19-24
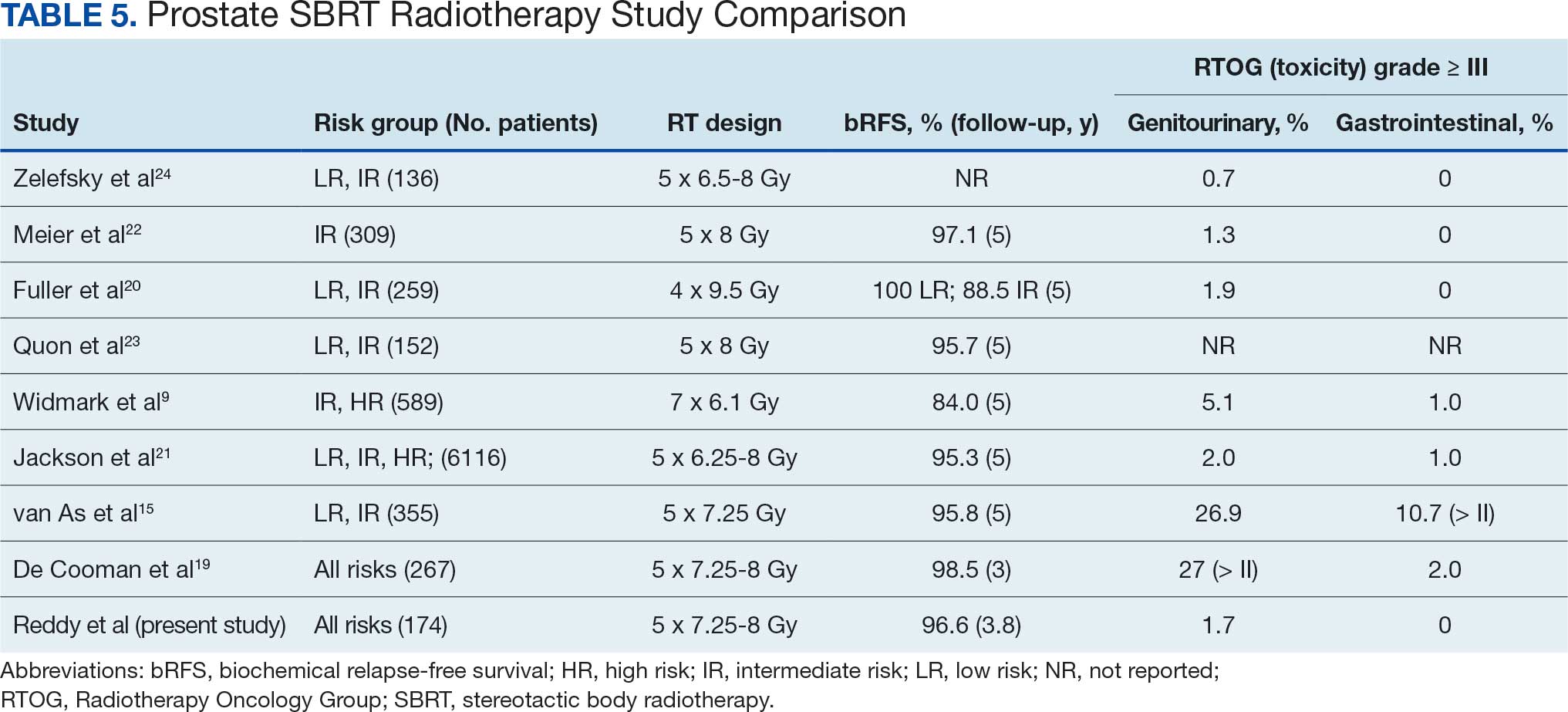
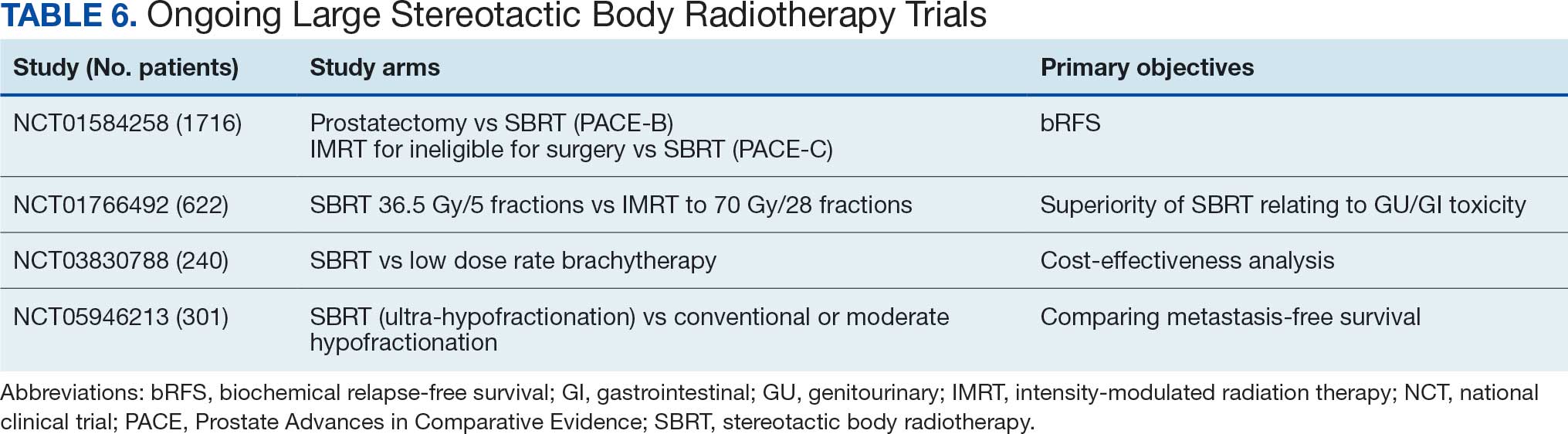
This study’s efficacy outcomes, particularly the high DFS rate, are consistent with the findings from these landmark trials, suggesting that the SBRT regimen used at KCVAMC is effective in achieving early disease control despite 17.2% of patients having high-risk disease. The GU toxicity observed in this study, with a 28.2% rate of grade II or higher events, is also comparable with the 26.9% reported in the 5-fraction SBRT arm of the PACE-B trial, which had a longer median follow-up of 74 months.15 It is important to note that a portion of these grade II events occurred in patients who were already on a blockers for pre-existing lower urinary tract symptoms before starting radiotherapy, which may inflate the observed cumulative acute toxicity score.
A critical comparison is how SBRT toxicity aligns with moderate hypofractionation (eg, 60 Gy in 20 fractions or 70 Gy in 28 fractions as reported by others).4,6 Our observed grade III and higher GU toxicity rate (1.7%) and grade III and higher GI toxicity rate (0%) are highly favorable when compared with historical moderate hypofractionation data, which typically report grade III GU toxicity in the range of 2% to 3% and grade III GI toxicity around 1% to 2%. This suggests that despite the higher dose per fraction, SBRT does not necessarily lead to increased severe acute toxicity, potentially offering a superior therapeutic ratio for GI and GU sparing.
However, the occurrence of a grade IV rectovesical fistula in 1 patient (0.6%)—who received the 40 Gy dose—was a serious complication that warrants careful consideration. This rare, but severe, complication in the higher dose cohort underscores the potential for increased organ-at-risk toxicity, particularly in the absence of a hydrogel rectal spacer, which is designed to mitigate high-dose rectal exposure. While the overall rate of significant GU toxicity remains low, this event highlights the potential risks associated with SBRT. Hydrogel rectal spacers are designed to increase the distance between the prostate and the rectum, which can reduce the rectal radiation dose and potentially mitigate the risk of such fistulas. The low rate of grade II or worse GI toxicity (3.4%) in our study is noteworthy, especially considering that hydrogel spacers were not routinely used. This finding aligns with the 2.5% GI toxicity rate reported in the SBRT arm of the PACE-B trial, suggesting that careful treatment planning and delivery techniques, such as VMAT-IMRT and daily CBCT for IGRT, may contribute to minimizing GI toxicity even without the use of rectal spacers.15 The exclusive use of 3-dimensional CBCT for IGRT in our study, without the use of fiducial markers, suggests that accurate target localization can be achieved with this approach, contributing to the observed efficacy and reduced toxicity.
Strengths and Limitations
This study’s retrospective, single-center design may have introduced selection bias. The median follow-up of 45 months, while substantial, is still relatively short for assessing very late toxicities and long-term oncologic outcomes in prostate cancer, which is known for late recurrences. Additionally, the lack of a direct comparison group within KCVAMC limits the ability to definitively attribute the observed outcomes solely to SBRT treatment. However, the strengths of this study include the inclusion of a consecutive series of veteran patients with localized prostate cancer across all risk categories, providing a real-world perspective on SBRT outcomes in a diverse patient population. Furthermore, the detailed assessment of efficacy and toxicity via standardized RTOG criteria enhances the comparability of our findings with those of other published prospective studies, despite the retrospective nature of the data.
CONCLUSIONS
This single-institution retrospective analysis revealed that short-term SBRT (36.25 to 40 Gy in 5 fractions), with a minimum follow-up of 24 months and a median follow-up of 45 months, for localized prostate cancer, including patients at HR, is associated with promising early efficacy and acceptable toxicity, even in the absence of routine fiducial or hydrogel spacer use. The favorable actuarial 5-year DFS and OS rates, coupled with a manageable toxicity profile, suggest that SBRT is a safe and convenient treatment option for many patients with localized prostate cancer. However, a longer follow-up is necessary to confirm these findings and fully characterize the long-term efficacy and toxicity of this SBRT regimen. Nevertheless, the results contribute to the growing body of evidence suggesting that SBRT is a safe and convenient treatment option for many patients with localized prostate cancer.
- Siegel RL, Kratzer TB, Giaquinto AN, et al. Cancer statistics, 2025. CA Cancer J Clin. 2025;75:10-45. doi:10.3322/caac.21871
- Donovan JL, Hamdy FC, Lane JA, et al. Patient-reported outcomes after monitoring, surgery, or radiotherapy for prostate cancer. N Engl J Med. 2016;375:1425-1437. doi:10.1056/NEJMoa1606221
- Hamdy FC, Donovan JL, Lane JA, et al. 10-year outcomes after monitoring, surgery, or radiotherapy for localized prostate cancer. N Engl J Med. 2016;375:1415-1424. doi:10.1056/NEJMoa1606220
- Dearnaley D, Syndikus I, Mossop H, et al. Conventional versus hypofractionated high-dose intensity-modulated radiotherapy for prostate cancer: 5-year outcomes of the randomised, non-inferiority, phase 3 CHHiP trial. Lancet Oncol. 2016;17:1047-1060. doi:10.1016/S1470-2045(16)30102-4
- Catton CN, Lukka H, Gu CS, et al. Randomized trial of a hypofractionated radiation regimen for the treatment of localized prostate cancer. J Clin Oncol. 2017;35:1884-1890. doi:10.1200/JCO.2016.71.7397
- Lee WR, Dignam JJ, Amin MB, et al. Long-term analysis of NRG Oncology RTOG 0415: a randomized phase III noninferiority study comparing two fractionation schedules in patients with low-risk prostate cancer. J Clin Oncol. 2024;42:2377-2381. doi:10.1200/JCO.23.02445
- de Vries KC, Wortel RC, Oomen-de Hoop E, et al. Hypofractionated versus conventionally fractionated radiation therapy for patients with intermediate- or high-risk, localized, prostate cancer: 7-year outcomes from the randomized, multicenter, open-label, phase 3 HYPRO trial. Int J Radiat Oncol Biol Phys. 2020;106:108-115. doi:10.1016/j.ijrobp.2019.09.007
- Incrocci L, Wortel RC, Alemayehu WG, et al. Hypofractionated versus conventionally fractionated radiotherapy for patients with localised prostate cancer (HYPRO): final efficacy results from a randomised, multicentre, open-label, phase 3 trial. Lancet Oncol. 2016;17:1061-1069. doi:10.1016/S1470-2045(16)30070-5
- Widmark A, Gunnlaugsson A, Beckman L, et al. Ultra-hypofractionated versus conventionally fractionated radiotherapy for prostate cancer: 5-year outcomes of the HYPO-RT-PC randomised, non-inferiority, phase 3 trial. Lancet. 2019;394:385-395. doi:10.1016/S0140-6736(19)31131-6
- Brenner DJ, Hall EJ. Fractionation and protraction for radiotherapy of prostate carcinoma. Int J Radiat Oncol Biol Phys. 1999;43:1095-101. doi:10.1016/s0360-3016(98)00438-6
- Dasu A. Is the alpha/beta value for prostate tumours low enough to be safely used in clinical trials? Clin Oncol (R Coll Radiol). 2007;19:289-301. doi:10.1016/j.clon.2007.02.007
- Garcia-Barros M, Paris F, Cordon-Cardo C, et al. Tumor response to radiotherapy regulated by endothelial cell apoptosis. Science. 2003;300:1155-1159. doi:10.1126/science.1082504
- Gulliford S, Hall E, Dearnaley D. Hypofractionation trials and radiobiology of prostate cancer. Oncoscience. 2017;4:27-28. doi:10.18632/oncoscience.347
- Fuks Z, Kolesnick R. Engaging the vascular component of the tumor response. Cancer Cell. 2005;8:89-91. doi:10.1016/j.ccr.2005.07.014
- van As N, Griffin C, Tree A, et al. Phase 3 Trial of stereotactic body radiotherapy in localized prostate cancer. N Engl J Med. Oct 17 2024;391:1413-1425. doi:10.1056/NEJMoa2403365
- National Comprehensive Cancer Network. NCCN Guidelines Version 5. 2026 Prostate Cancer. Accessed March 24, 2026. https://www.nccn.org/professionals/physician_gls/pdf/prostate.pdf
- Lawton CA, Michalski J, El-Naqa I, et al. RTOG GU radiation oncology specialists reach consensus on pelvic lymph node volumes for high-risk prostate cancer. Int J Radiat Oncol Biol Phys. 2009;74:383-387. doi:10.1016/j.ijrobp.2008.08.002
- Lehrer EJ, Kishan AU, Yu JB, et al. Ultrahypofractionated versus hypofractionated and conventionally fractionated radiation therapy for localized prostate cancer: a systematic review and meta-analysis of phase III randomized trials. Radiother Oncol. 2020;148:235-242. doi:10.1016/j.radonc.2020.04.037
- De Cooman B, Debacker T, Adams T, et al. Stereotactic body radiotherapy (SBRT) as a treatment for localized prostate cancer: a retrospective analysis. Radiat Oncol. 2025;20:25. doi:10.1186/s13014-025-02598-8
- Fuller DB, Falchook AD, Crabtree T, et al. Phase 2 multicenter trial of heterogeneous-dosing stereotactic body radiotherapy for low- and intermediate-risk prostate cancer: 5-year outcomes. Eur Urol Oncol. 2018;1:540-547. doi:10.1016/j.euo.2018.06.013
- Jackson WC, Silva J, Hartman HE, et al. Stereotactic body radiation therapy for localized prostate cancer: a systematic review and meta-analysis of over 6,000 patients treated on prospective studies. Int J Radiat Oncol Biol Phys. 2019;104:778-789. doi:10.1016/j.ijrobp.2019.03.051
- Meier RM, Bloch DA, Cotrutz C, et al. Multicenter trial of stereotactic body radiation therapy for low- and intermediate-risk prostate cancer: survival and toxicity endpoints. nt J Radiat Oncol Biol Phys. 2018;102:296-303. doi:10.1016/j.ijrobp.2018.05.040
- Quon HC, Ong A, Cheung P, et al. Once-weekly versus every-other-day stereotactic body radiotherapy in patients with prostate cancer (PATRIOT): a phase 2 randomized trial. Radiother Oncol. 2018;127:206-212. doi:10.1016/j.radonc.2018.02.029
- Zelefsky MJ, Kollmeier M, McBride S, et al. Five-year outcomes of a phase 1 dose-escalation study using stereotactic body radiosurgery for patients with low-risk and intermediate-risk prostate cancer. Int J Radiat Oncol Biol Phys. 2019;104:42-49. doi:10.1016/j.ijrobp.2018.12.045
Prostate cancer is the most common cancer in US males, with an estimated 313,780 new cases and 35,770 deaths in 2025.1 Several treatment options are available for localized prostate cancer that have similar outcomes, including active surveillance for low-risk cancers, surgery, or radiotherapy.2,3 Conventional fractionation radiotherapy (CFRT) with 40 to 45 fractions over 8 to 9 weeks has been used for decades. Over the past 2 decades, moderate hypofractionation schedules with 2.4 to 3.4 Gy per fraction over 20 to 28 fractions have become standard, as many noninferiority randomized clinical trials (RCTs) such as CHHiP (UK),4 PROFIT (Canada and Europe),5 NRG Oncology RTOG 0415 (US),6 HYPRO (Netherlands),7,8 and HYPO-RT-PC (Sweden and Denmark),9 have shown the noninferiority of moderately hypofractionated radiotherapy compared with CFRT. Notably, most of these noninferiority studies primarily included patients with low- or intermediate-risk prostate cancer, except for the HYPO-RT-PC trial,9 which also included patients with intermediate- and high-risk prostate cancer.
These noninferiority studies, along with technological advances in radiotherapy, such as intensity-modulated radiotherapy (IMRT), volumetric modulated arc therapy (VMAT), and image-guided radiotherapy (IGRT), paved the path to ultrahypofractionated stereotactic body radiotherapy (SBRT) that is delivered in 5 fractions of ≥ 6 Gy. This high dose per fraction may have a radiobiologic advantage over conventional fractionation. The relatively low a/ß ratio of prostate cancer, estimated to be between 1 and 2, suggests that tumor cells may be particularly sensitive to the high doses per fraction delivered in SBRT.10-13 Compared with CFRT, SBRT-induced tumor cell death may also be mediated through different pathways; this pathway appears to be generated in a dose-dependent manner, particularly with doses > 8 Gy per fraction.14,15 Additionally, the higher a/ß ratio for the surrounding organs at risk, such as the bladder and rectum, theoretically allows for an improved therapeutic ratio window that maximizes tumor control while minimizing damage to healthy tissues.
A substantial body of evidence from prospective studies and meta-analyses supports the use of SBRT for localized prostate cancer. HYPO-RT-PC, a significant phase 3 noninferiority study, enrolled 1200 patients with intermediate (89%) and high-risk (11%) prostate cancer randomized between 2 arms, including CFRT to 78 Gy in 39 fractions and SBRT to 42.7 Gy in 7 fractions, treated 3 days weekly. After a median follow-up of 60 months, the estimated 5-year biochemical relapse-free survival rate was 84% in both groups.9 This trial was notable because it was the first randomized study to demonstrate that SBRT was noninferior to CFRT in intermediate- and high-risk prostate cancer patients. Another pivotal phase 3 trial, the PACE-B study, enrolled 874 patients to compare SBRT (36.25 Gy to the prostate gland, with a secondary dose of 40 Gy to the gross tumor volume where applicable, in 5 fractions) with CFRT (78 Gy in 39 fractions) and moderately hypofractionated radiotherapy (HFRT) (62 Gy in 20 fractions) in patients with low- or intermediate-risk prostate cancer. With a 74-month median follow-up, the study reported 5-year biochemical free rates of 94.6% for CFRT and 95.8% for SBRT, confirming the noninferiority of SBRT to CFRT.15
SBRT offers short, effective, and convenient treatment to many patients with localized prostate cancer. While previous guidelines were more restrictive, the March 2026 National Comprehensive Cancer Network (NCCN) guidelines now list SBRT as a preferred treatment modality for high-risk prostate cancer.16
Given the growing body of evidence supporting the efficacy and safety of SBRT, we implemented an SBRT program in 2014 at a tertiary care center for veterans. This retrospective study was undertaken to evaluate the early efficacy and toxicity of SBRT in patients with localized prostate cancer treated at our institution, including patients across all risk stratifications.
METHODS
We identified 242 patients diagnosed with prostate cancer who underwent SBRT treatment between November 2014 and October 2024 at Overland Park Veterans Affairs Radiation Oncology Clinic. For the final analysis, 46 patients with < 2 years of follow-up and 22 patients who died from causes other than prostate cancer were excluded, resulting in a cohort of 174 patients with ≥ 24-month follow-up.
Treatment
Patients eligible for staging underwent imaging according to NCCN guidelines, including computed tomography (CT) of the abdomen and pelvis, bone scintigraphy, or, in recent years, prostate-specific membrane antigen positron emission tomography, primarily used for unfavorable intermediate-risk (UIR) and high-risk (HR) cancers. Patients with a negative staging work-up for nodal or skeletal disease were included. Prior to planning the CT simulation, patients were given bowel preparation instructions, including a low-fiber and low-gas-producing diet, simethicone, and enemas, the night before and morning of the simulation. Patients were instructed to arrive with a comfortably full bladder, having not voided for 2 to 3 hours prior to the procedure. At Kansas City Veterans Affairs Medical Center (KCVAMC), SBRT treatment was generally restricted to patients with a baseline American Urological Association symptom score of 15 to 20 out of 35 and a prostate gland size < 80 mL to minimize the risk of acute urinary toxicity. We did not use intraprostatic fiducials, hydrogel rectal spacers, or intravenous contrast agents for planning CT simulation.
Patients were placed in a supine position, and a vacuum bag was used for immobilization. Following the CT simulation, the images were transferred to the Eclipse treatment planning system. The clinical target volume (CTV) encompassed the prostate and the proximal 1.0 cm of the seminal vesicles for Gleason score (GS) 1 to 2, and the entire seminal vesicle was included for GS 3 to 5, which is consistent with KCVAMC practice and established safety protocols. The planning target volume (PTV) was created by uniformly expanding the CTV by 5 to 7 mm, except for the posterior margin, which was limited to 3 to 5 mm. When elective nodal radiotherapy was planned for HR prostate cancer, the pelvic field for CT simulation started at the L-2 upper border, with the lower border extending to the lesser trochanter. The pelvic nodes were delineated per Radiation Therapy Oncology Group (RTOG) guidelines.17 The CTV nodes (CTVn), including common iliac, external and internal iliac nodes, obturator, and presacral nodes, were created by uniformly expanding the CTVn by 2 to 3 mm. Slice-by-slice corrections were made to avoid bowel overlap in these patients.
The use of androgen deprivation therapy (ADT) for a duration of 6 to 24 months was prescribed for patients with UIR or HR prostate cancer per NCCN guidelines.16 The prescribed dose to the PTV was 36.25 to 40 Gy (40 Gy was mostly used as a boost to the dominant lesion) in 5 fractions, with each fraction ranging from 7.25 to 8 Gy. For elective nodal radiotherapy in patients at HR, the prescribed dose was 25 Gy in 5 fractions. All patients were planned for VMAT, which aims to deliver ≥ 95% of the prescription dose to 95% of the PTV. Once the physician approved the treatment plan and physics quality assessment was completed, treatments commenced on an every-other-day schedule. Patients received the same bowel preparation instructions for each treatment as for the planning CT simulation. Daily treatment accuracy was confirmed via daily 3-dimensional cone-beam CT (CBCT) for IGRT. No fiducials or hydrogel rectal spacers were used.
Follow-up Schedule and Toxicity Assessment
Follow-up assessments were conducted 4 to 6 weeks after radiation therapy and then repeated every 6 months for 2 to 5 years, and annually thereafter. At each follow-up visit, patients were evaluated for genitourinary (GU) and gastrointestinal (GI) toxicity, according to RTOG toxicity criteria. Prostate-specific antigen (PSA) levels were monitored; in patients receiving ADT, testosterone levels were also checked.
Statistical Analysis
Biochemical failure was defined using the Phoenix definition (nadir PSA + 2 ng/mL). Differences between dose cohorts were assessed using the log-rank test for survival outcomes and X2 testing for categorical variables. GU and GI toxicities were summarized as cumulative incidences of RTOG grade ≥ II events. Statistical significance was set at P < .05.
RESULTS
One hundred seventy-four patients were included in the retrospective review. Patients had a median follow-up of 45 months (range, 24-111) (Figure). The median age at treatment was 74 years (range, 51-88), and the median pretreatment PSA level was 11.9 ng/mL (range, 0.6-69.5). Twenty-six patients (14.9%) had a GS 1, 77 (44.3%) had GS 2, 41 (23.6%) had GS 3, 18 (10.3%) had GS 4, and 12 (6.9%) had GS 5. Fifty-one patients (29.3%) received elective pelvic nodal radiotherapy, and 93 patients (53.4%) received ADT (Table 1).


At 24 months follow-up, 6 patients (3.4%) had biochemical failures. One patient died from metastatic prostate cancer, and 5 patients are living with biochemical failure (Table 2). The actuarial 5-year overall survival (OS) rate was 99.4%, and the 5-year disease-free survival (DFS) rate was 96.6%. We performed a subanalysis comparing outcomes of the 36.25 Gy vs 40 Gy SBRT cohorts. There was no statistically significant difference in DFS, OS, or the cumulative incidence of grade II/III toxicity between patients treated with 40 Gy vs 36.25 Gy. Outcomes stratified by NCCN risk groups (low, intermediate, high/very high) are detailed in Table 3. As expected, DFS was slightly lower in the high-risk group, but overall disease control remained high across all stratifications.


The cumulative incidence of RTOG grade II and higher GU toxicity was 28.2% (Table 4). This included 46 patients (26.4%) with grade II GU toxicity and 2 patients (1.2%) who developed grade III GU complications (1 requiring self-catheterization and another a suprapubic catheter for urinary retention). One patient (0.6%) treated with a 40 Gy dose regimen experienced a grade IV GU complication in the form of a rectovesical fistula necessitating surgical intervention.

The cumulative incidence of RTOG grade II or higher GI toxicity was 3.4%, and no grade III or IV gastrointestinal toxicities were observed during the follow-up period. Importantly, intraprostatic fiducials, hydrogel rectal spacers, or intravenous contrast were not routinely used in this cohort of patients.
The high rates of actuarial 5-year DFS and OS observed suggest a favorable initial response to the SBRT regimen employed at KCVAMC. However, given the potential for late recurrence in patients with prostate cancer, longer follow-up is essential to determine the durability of these outcomes. The observed GU toxicity rate of 28.2% for grade II and higher events warrants careful consideration and compares with other published data on SBRT for prostate cancer.15 The occurrence of a grade IV rectovesical fistula, although rare, is a notable adverse event that warrants discussion in the context of the treatment approach. The low incidence of grade II or higher GI toxicity is an encouraging finding, particularly given that hydrogel rectal spacers are not routinely used to minimize rectal exposure.
DISCUSSION
The primary objective of this retrospective study was to evaluate the outcomes of SBRT for patients with localized prostate cancer treated at KCVAMC and to compare these results with those reported in the literature. Our findings demonstrate promising intermediate-term efficacy, with an estimated 5-year DFS of 96.6% and OS of 99.4% at a median follow-up of 45 months. Furthermore, the observed toxicity profile appears acceptable, with a cumulative grade II and higher GU toxicity rate of 28.2% and a grade II or higher GI toxicity rate of 3.4%. Notably, these outcomes were achieved without the routine use of intraprostatic fiducials or hydrogel rectal spacers.
Two pivotal randomized phase 3 trials have established the noninferiority of ultrahypofractionated radiotherapy (UHRT) with SBRT over conventional fractionation. The HYPO-RT-PC trial compared SBRT (42.7 Gy in 7 fractions) with conventional fractionation (78 Gy in 39 fractions) in intermediate- and high-risk patients with prostate cancer and reported a 5-year biochemical relapse-free survival of 84% in both arms.9 The PACE-B trial, which included patients at low- and intermediate-risk, compared SBRT (36.25 Gy in 5 fractions) with conventional or moderate HFRT and reported a 5-year biochemical control rate of 95.8% in the SBRT arm and 94.6% in the control arm.15
A comprehensive review and meta-analysis of 7 phase 3 studies involving 6795 patients compared different radiotherapy regimens, namely, UHRT, HFRT, and CFRT, and reported that after 5 years, the DFS rates were 85.1% for CFRT, 86% for HFRT, and 85% for UHRT, with no significant difference in toxicity among the 3 different treatment approaches.18 This suggests that shorter, more intense radiotherapy schedules (UHRT and HFRT) may be as effective and safe as traditional, longer courses of radiation.
There are multiple published nonrandomized prospective trials in which thousands of patients with extreme hypofractionation have been treated with different doses, fractions, and techniques. While heterogeneity and limited long-term follow-up in the existing evidence are acknowledged, these data suggest that prostate SBRT provides appropriate biochemical control with few high-grade toxicities, supporting its ongoing global use and justifying further prospective investigations. Comparative data are shown in Table 5. Several ongoing studies are evaluating noninferiority, superiority, and cost-effectiveness using different methodologies (Table 6).9,15,19-24


This study’s efficacy outcomes, particularly the high DFS rate, are consistent with the findings from these landmark trials, suggesting that the SBRT regimen used at KCVAMC is effective in achieving early disease control despite 17.2% of patients having high-risk disease. The GU toxicity observed in this study, with a 28.2% rate of grade II or higher events, is also comparable with the 26.9% reported in the 5-fraction SBRT arm of the PACE-B trial, which had a longer median follow-up of 74 months.15 It is important to note that a portion of these grade II events occurred in patients who were already on a blockers for pre-existing lower urinary tract symptoms before starting radiotherapy, which may inflate the observed cumulative acute toxicity score.
A critical comparison is how SBRT toxicity aligns with moderate hypofractionation (eg, 60 Gy in 20 fractions or 70 Gy in 28 fractions as reported by others).4,6 Our observed grade III and higher GU toxicity rate (1.7%) and grade III and higher GI toxicity rate (0%) are highly favorable when compared with historical moderate hypofractionation data, which typically report grade III GU toxicity in the range of 2% to 3% and grade III GI toxicity around 1% to 2%. This suggests that despite the higher dose per fraction, SBRT does not necessarily lead to increased severe acute toxicity, potentially offering a superior therapeutic ratio for GI and GU sparing.
However, the occurrence of a grade IV rectovesical fistula in 1 patient (0.6%)—who received the 40 Gy dose—was a serious complication that warrants careful consideration. This rare, but severe, complication in the higher dose cohort underscores the potential for increased organ-at-risk toxicity, particularly in the absence of a hydrogel rectal spacer, which is designed to mitigate high-dose rectal exposure. While the overall rate of significant GU toxicity remains low, this event highlights the potential risks associated with SBRT. Hydrogel rectal spacers are designed to increase the distance between the prostate and the rectum, which can reduce the rectal radiation dose and potentially mitigate the risk of such fistulas. The low rate of grade II or worse GI toxicity (3.4%) in our study is noteworthy, especially considering that hydrogel spacers were not routinely used. This finding aligns with the 2.5% GI toxicity rate reported in the SBRT arm of the PACE-B trial, suggesting that careful treatment planning and delivery techniques, such as VMAT-IMRT and daily CBCT for IGRT, may contribute to minimizing GI toxicity even without the use of rectal spacers.15 The exclusive use of 3-dimensional CBCT for IGRT in our study, without the use of fiducial markers, suggests that accurate target localization can be achieved with this approach, contributing to the observed efficacy and reduced toxicity.
Strengths and Limitations
This study’s retrospective, single-center design may have introduced selection bias. The median follow-up of 45 months, while substantial, is still relatively short for assessing very late toxicities and long-term oncologic outcomes in prostate cancer, which is known for late recurrences. Additionally, the lack of a direct comparison group within KCVAMC limits the ability to definitively attribute the observed outcomes solely to SBRT treatment. However, the strengths of this study include the inclusion of a consecutive series of veteran patients with localized prostate cancer across all risk categories, providing a real-world perspective on SBRT outcomes in a diverse patient population. Furthermore, the detailed assessment of efficacy and toxicity via standardized RTOG criteria enhances the comparability of our findings with those of other published prospective studies, despite the retrospective nature of the data.
CONCLUSIONS
This single-institution retrospective analysis revealed that short-term SBRT (36.25 to 40 Gy in 5 fractions), with a minimum follow-up of 24 months and a median follow-up of 45 months, for localized prostate cancer, including patients at HR, is associated with promising early efficacy and acceptable toxicity, even in the absence of routine fiducial or hydrogel spacer use. The favorable actuarial 5-year DFS and OS rates, coupled with a manageable toxicity profile, suggest that SBRT is a safe and convenient treatment option for many patients with localized prostate cancer. However, a longer follow-up is necessary to confirm these findings and fully characterize the long-term efficacy and toxicity of this SBRT regimen. Nevertheless, the results contribute to the growing body of evidence suggesting that SBRT is a safe and convenient treatment option for many patients with localized prostate cancer.
Prostate cancer is the most common cancer in US males, with an estimated 313,780 new cases and 35,770 deaths in 2025.1 Several treatment options are available for localized prostate cancer that have similar outcomes, including active surveillance for low-risk cancers, surgery, or radiotherapy.2,3 Conventional fractionation radiotherapy (CFRT) with 40 to 45 fractions over 8 to 9 weeks has been used for decades. Over the past 2 decades, moderate hypofractionation schedules with 2.4 to 3.4 Gy per fraction over 20 to 28 fractions have become standard, as many noninferiority randomized clinical trials (RCTs) such as CHHiP (UK),4 PROFIT (Canada and Europe),5 NRG Oncology RTOG 0415 (US),6 HYPRO (Netherlands),7,8 and HYPO-RT-PC (Sweden and Denmark),9 have shown the noninferiority of moderately hypofractionated radiotherapy compared with CFRT. Notably, most of these noninferiority studies primarily included patients with low- or intermediate-risk prostate cancer, except for the HYPO-RT-PC trial,9 which also included patients with intermediate- and high-risk prostate cancer.
These noninferiority studies, along with technological advances in radiotherapy, such as intensity-modulated radiotherapy (IMRT), volumetric modulated arc therapy (VMAT), and image-guided radiotherapy (IGRT), paved the path to ultrahypofractionated stereotactic body radiotherapy (SBRT) that is delivered in 5 fractions of ≥ 6 Gy. This high dose per fraction may have a radiobiologic advantage over conventional fractionation. The relatively low a/ß ratio of prostate cancer, estimated to be between 1 and 2, suggests that tumor cells may be particularly sensitive to the high doses per fraction delivered in SBRT.10-13 Compared with CFRT, SBRT-induced tumor cell death may also be mediated through different pathways; this pathway appears to be generated in a dose-dependent manner, particularly with doses > 8 Gy per fraction.14,15 Additionally, the higher a/ß ratio for the surrounding organs at risk, such as the bladder and rectum, theoretically allows for an improved therapeutic ratio window that maximizes tumor control while minimizing damage to healthy tissues.
A substantial body of evidence from prospective studies and meta-analyses supports the use of SBRT for localized prostate cancer. HYPO-RT-PC, a significant phase 3 noninferiority study, enrolled 1200 patients with intermediate (89%) and high-risk (11%) prostate cancer randomized between 2 arms, including CFRT to 78 Gy in 39 fractions and SBRT to 42.7 Gy in 7 fractions, treated 3 days weekly. After a median follow-up of 60 months, the estimated 5-year biochemical relapse-free survival rate was 84% in both groups.9 This trial was notable because it was the first randomized study to demonstrate that SBRT was noninferior to CFRT in intermediate- and high-risk prostate cancer patients. Another pivotal phase 3 trial, the PACE-B study, enrolled 874 patients to compare SBRT (36.25 Gy to the prostate gland, with a secondary dose of 40 Gy to the gross tumor volume where applicable, in 5 fractions) with CFRT (78 Gy in 39 fractions) and moderately hypofractionated radiotherapy (HFRT) (62 Gy in 20 fractions) in patients with low- or intermediate-risk prostate cancer. With a 74-month median follow-up, the study reported 5-year biochemical free rates of 94.6% for CFRT and 95.8% for SBRT, confirming the noninferiority of SBRT to CFRT.15
SBRT offers short, effective, and convenient treatment to many patients with localized prostate cancer. While previous guidelines were more restrictive, the March 2026 National Comprehensive Cancer Network (NCCN) guidelines now list SBRT as a preferred treatment modality for high-risk prostate cancer.16
Given the growing body of evidence supporting the efficacy and safety of SBRT, we implemented an SBRT program in 2014 at a tertiary care center for veterans. This retrospective study was undertaken to evaluate the early efficacy and toxicity of SBRT in patients with localized prostate cancer treated at our institution, including patients across all risk stratifications.
METHODS
We identified 242 patients diagnosed with prostate cancer who underwent SBRT treatment between November 2014 and October 2024 at Overland Park Veterans Affairs Radiation Oncology Clinic. For the final analysis, 46 patients with < 2 years of follow-up and 22 patients who died from causes other than prostate cancer were excluded, resulting in a cohort of 174 patients with ≥ 24-month follow-up.
Treatment
Patients eligible for staging underwent imaging according to NCCN guidelines, including computed tomography (CT) of the abdomen and pelvis, bone scintigraphy, or, in recent years, prostate-specific membrane antigen positron emission tomography, primarily used for unfavorable intermediate-risk (UIR) and high-risk (HR) cancers. Patients with a negative staging work-up for nodal or skeletal disease were included. Prior to planning the CT simulation, patients were given bowel preparation instructions, including a low-fiber and low-gas-producing diet, simethicone, and enemas, the night before and morning of the simulation. Patients were instructed to arrive with a comfortably full bladder, having not voided for 2 to 3 hours prior to the procedure. At Kansas City Veterans Affairs Medical Center (KCVAMC), SBRT treatment was generally restricted to patients with a baseline American Urological Association symptom score of 15 to 20 out of 35 and a prostate gland size < 80 mL to minimize the risk of acute urinary toxicity. We did not use intraprostatic fiducials, hydrogel rectal spacers, or intravenous contrast agents for planning CT simulation.
Patients were placed in a supine position, and a vacuum bag was used for immobilization. Following the CT simulation, the images were transferred to the Eclipse treatment planning system. The clinical target volume (CTV) encompassed the prostate and the proximal 1.0 cm of the seminal vesicles for Gleason score (GS) 1 to 2, and the entire seminal vesicle was included for GS 3 to 5, which is consistent with KCVAMC practice and established safety protocols. The planning target volume (PTV) was created by uniformly expanding the CTV by 5 to 7 mm, except for the posterior margin, which was limited to 3 to 5 mm. When elective nodal radiotherapy was planned for HR prostate cancer, the pelvic field for CT simulation started at the L-2 upper border, with the lower border extending to the lesser trochanter. The pelvic nodes were delineated per Radiation Therapy Oncology Group (RTOG) guidelines.17 The CTV nodes (CTVn), including common iliac, external and internal iliac nodes, obturator, and presacral nodes, were created by uniformly expanding the CTVn by 2 to 3 mm. Slice-by-slice corrections were made to avoid bowel overlap in these patients.
The use of androgen deprivation therapy (ADT) for a duration of 6 to 24 months was prescribed for patients with UIR or HR prostate cancer per NCCN guidelines.16 The prescribed dose to the PTV was 36.25 to 40 Gy (40 Gy was mostly used as a boost to the dominant lesion) in 5 fractions, with each fraction ranging from 7.25 to 8 Gy. For elective nodal radiotherapy in patients at HR, the prescribed dose was 25 Gy in 5 fractions. All patients were planned for VMAT, which aims to deliver ≥ 95% of the prescription dose to 95% of the PTV. Once the physician approved the treatment plan and physics quality assessment was completed, treatments commenced on an every-other-day schedule. Patients received the same bowel preparation instructions for each treatment as for the planning CT simulation. Daily treatment accuracy was confirmed via daily 3-dimensional cone-beam CT (CBCT) for IGRT. No fiducials or hydrogel rectal spacers were used.
Follow-up Schedule and Toxicity Assessment
Follow-up assessments were conducted 4 to 6 weeks after radiation therapy and then repeated every 6 months for 2 to 5 years, and annually thereafter. At each follow-up visit, patients were evaluated for genitourinary (GU) and gastrointestinal (GI) toxicity, according to RTOG toxicity criteria. Prostate-specific antigen (PSA) levels were monitored; in patients receiving ADT, testosterone levels were also checked.
Statistical Analysis
Biochemical failure was defined using the Phoenix definition (nadir PSA + 2 ng/mL). Differences between dose cohorts were assessed using the log-rank test for survival outcomes and X2 testing for categorical variables. GU and GI toxicities were summarized as cumulative incidences of RTOG grade ≥ II events. Statistical significance was set at P < .05.
RESULTS
One hundred seventy-four patients were included in the retrospective review. Patients had a median follow-up of 45 months (range, 24-111) (Figure). The median age at treatment was 74 years (range, 51-88), and the median pretreatment PSA level was 11.9 ng/mL (range, 0.6-69.5). Twenty-six patients (14.9%) had a GS 1, 77 (44.3%) had GS 2, 41 (23.6%) had GS 3, 18 (10.3%) had GS 4, and 12 (6.9%) had GS 5. Fifty-one patients (29.3%) received elective pelvic nodal radiotherapy, and 93 patients (53.4%) received ADT (Table 1).


At 24 months follow-up, 6 patients (3.4%) had biochemical failures. One patient died from metastatic prostate cancer, and 5 patients are living with biochemical failure (Table 2). The actuarial 5-year overall survival (OS) rate was 99.4%, and the 5-year disease-free survival (DFS) rate was 96.6%. We performed a subanalysis comparing outcomes of the 36.25 Gy vs 40 Gy SBRT cohorts. There was no statistically significant difference in DFS, OS, or the cumulative incidence of grade II/III toxicity between patients treated with 40 Gy vs 36.25 Gy. Outcomes stratified by NCCN risk groups (low, intermediate, high/very high) are detailed in Table 3. As expected, DFS was slightly lower in the high-risk group, but overall disease control remained high across all stratifications.


The cumulative incidence of RTOG grade II and higher GU toxicity was 28.2% (Table 4). This included 46 patients (26.4%) with grade II GU toxicity and 2 patients (1.2%) who developed grade III GU complications (1 requiring self-catheterization and another a suprapubic catheter for urinary retention). One patient (0.6%) treated with a 40 Gy dose regimen experienced a grade IV GU complication in the form of a rectovesical fistula necessitating surgical intervention.

The cumulative incidence of RTOG grade II or higher GI toxicity was 3.4%, and no grade III or IV gastrointestinal toxicities were observed during the follow-up period. Importantly, intraprostatic fiducials, hydrogel rectal spacers, or intravenous contrast were not routinely used in this cohort of patients.
The high rates of actuarial 5-year DFS and OS observed suggest a favorable initial response to the SBRT regimen employed at KCVAMC. However, given the potential for late recurrence in patients with prostate cancer, longer follow-up is essential to determine the durability of these outcomes. The observed GU toxicity rate of 28.2% for grade II and higher events warrants careful consideration and compares with other published data on SBRT for prostate cancer.15 The occurrence of a grade IV rectovesical fistula, although rare, is a notable adverse event that warrants discussion in the context of the treatment approach. The low incidence of grade II or higher GI toxicity is an encouraging finding, particularly given that hydrogel rectal spacers are not routinely used to minimize rectal exposure.
DISCUSSION
The primary objective of this retrospective study was to evaluate the outcomes of SBRT for patients with localized prostate cancer treated at KCVAMC and to compare these results with those reported in the literature. Our findings demonstrate promising intermediate-term efficacy, with an estimated 5-year DFS of 96.6% and OS of 99.4% at a median follow-up of 45 months. Furthermore, the observed toxicity profile appears acceptable, with a cumulative grade II and higher GU toxicity rate of 28.2% and a grade II or higher GI toxicity rate of 3.4%. Notably, these outcomes were achieved without the routine use of intraprostatic fiducials or hydrogel rectal spacers.
Two pivotal randomized phase 3 trials have established the noninferiority of ultrahypofractionated radiotherapy (UHRT) with SBRT over conventional fractionation. The HYPO-RT-PC trial compared SBRT (42.7 Gy in 7 fractions) with conventional fractionation (78 Gy in 39 fractions) in intermediate- and high-risk patients with prostate cancer and reported a 5-year biochemical relapse-free survival of 84% in both arms.9 The PACE-B trial, which included patients at low- and intermediate-risk, compared SBRT (36.25 Gy in 5 fractions) with conventional or moderate HFRT and reported a 5-year biochemical control rate of 95.8% in the SBRT arm and 94.6% in the control arm.15
A comprehensive review and meta-analysis of 7 phase 3 studies involving 6795 patients compared different radiotherapy regimens, namely, UHRT, HFRT, and CFRT, and reported that after 5 years, the DFS rates were 85.1% for CFRT, 86% for HFRT, and 85% for UHRT, with no significant difference in toxicity among the 3 different treatment approaches.18 This suggests that shorter, more intense radiotherapy schedules (UHRT and HFRT) may be as effective and safe as traditional, longer courses of radiation.
There are multiple published nonrandomized prospective trials in which thousands of patients with extreme hypofractionation have been treated with different doses, fractions, and techniques. While heterogeneity and limited long-term follow-up in the existing evidence are acknowledged, these data suggest that prostate SBRT provides appropriate biochemical control with few high-grade toxicities, supporting its ongoing global use and justifying further prospective investigations. Comparative data are shown in Table 5. Several ongoing studies are evaluating noninferiority, superiority, and cost-effectiveness using different methodologies (Table 6).9,15,19-24


This study’s efficacy outcomes, particularly the high DFS rate, are consistent with the findings from these landmark trials, suggesting that the SBRT regimen used at KCVAMC is effective in achieving early disease control despite 17.2% of patients having high-risk disease. The GU toxicity observed in this study, with a 28.2% rate of grade II or higher events, is also comparable with the 26.9% reported in the 5-fraction SBRT arm of the PACE-B trial, which had a longer median follow-up of 74 months.15 It is important to note that a portion of these grade II events occurred in patients who were already on a blockers for pre-existing lower urinary tract symptoms before starting radiotherapy, which may inflate the observed cumulative acute toxicity score.
A critical comparison is how SBRT toxicity aligns with moderate hypofractionation (eg, 60 Gy in 20 fractions or 70 Gy in 28 fractions as reported by others).4,6 Our observed grade III and higher GU toxicity rate (1.7%) and grade III and higher GI toxicity rate (0%) are highly favorable when compared with historical moderate hypofractionation data, which typically report grade III GU toxicity in the range of 2% to 3% and grade III GI toxicity around 1% to 2%. This suggests that despite the higher dose per fraction, SBRT does not necessarily lead to increased severe acute toxicity, potentially offering a superior therapeutic ratio for GI and GU sparing.
However, the occurrence of a grade IV rectovesical fistula in 1 patient (0.6%)—who received the 40 Gy dose—was a serious complication that warrants careful consideration. This rare, but severe, complication in the higher dose cohort underscores the potential for increased organ-at-risk toxicity, particularly in the absence of a hydrogel rectal spacer, which is designed to mitigate high-dose rectal exposure. While the overall rate of significant GU toxicity remains low, this event highlights the potential risks associated with SBRT. Hydrogel rectal spacers are designed to increase the distance between the prostate and the rectum, which can reduce the rectal radiation dose and potentially mitigate the risk of such fistulas. The low rate of grade II or worse GI toxicity (3.4%) in our study is noteworthy, especially considering that hydrogel spacers were not routinely used. This finding aligns with the 2.5% GI toxicity rate reported in the SBRT arm of the PACE-B trial, suggesting that careful treatment planning and delivery techniques, such as VMAT-IMRT and daily CBCT for IGRT, may contribute to minimizing GI toxicity even without the use of rectal spacers.15 The exclusive use of 3-dimensional CBCT for IGRT in our study, without the use of fiducial markers, suggests that accurate target localization can be achieved with this approach, contributing to the observed efficacy and reduced toxicity.
Strengths and Limitations
This study’s retrospective, single-center design may have introduced selection bias. The median follow-up of 45 months, while substantial, is still relatively short for assessing very late toxicities and long-term oncologic outcomes in prostate cancer, which is known for late recurrences. Additionally, the lack of a direct comparison group within KCVAMC limits the ability to definitively attribute the observed outcomes solely to SBRT treatment. However, the strengths of this study include the inclusion of a consecutive series of veteran patients with localized prostate cancer across all risk categories, providing a real-world perspective on SBRT outcomes in a diverse patient population. Furthermore, the detailed assessment of efficacy and toxicity via standardized RTOG criteria enhances the comparability of our findings with those of other published prospective studies, despite the retrospective nature of the data.
CONCLUSIONS
This single-institution retrospective analysis revealed that short-term SBRT (36.25 to 40 Gy in 5 fractions), with a minimum follow-up of 24 months and a median follow-up of 45 months, for localized prostate cancer, including patients at HR, is associated with promising early efficacy and acceptable toxicity, even in the absence of routine fiducial or hydrogel spacer use. The favorable actuarial 5-year DFS and OS rates, coupled with a manageable toxicity profile, suggest that SBRT is a safe and convenient treatment option for many patients with localized prostate cancer. However, a longer follow-up is necessary to confirm these findings and fully characterize the long-term efficacy and toxicity of this SBRT regimen. Nevertheless, the results contribute to the growing body of evidence suggesting that SBRT is a safe and convenient treatment option for many patients with localized prostate cancer.
- Siegel RL, Kratzer TB, Giaquinto AN, et al. Cancer statistics, 2025. CA Cancer J Clin. 2025;75:10-45. doi:10.3322/caac.21871
- Donovan JL, Hamdy FC, Lane JA, et al. Patient-reported outcomes after monitoring, surgery, or radiotherapy for prostate cancer. N Engl J Med. 2016;375:1425-1437. doi:10.1056/NEJMoa1606221
- Hamdy FC, Donovan JL, Lane JA, et al. 10-year outcomes after monitoring, surgery, or radiotherapy for localized prostate cancer. N Engl J Med. 2016;375:1415-1424. doi:10.1056/NEJMoa1606220
- Dearnaley D, Syndikus I, Mossop H, et al. Conventional versus hypofractionated high-dose intensity-modulated radiotherapy for prostate cancer: 5-year outcomes of the randomised, non-inferiority, phase 3 CHHiP trial. Lancet Oncol. 2016;17:1047-1060. doi:10.1016/S1470-2045(16)30102-4
- Catton CN, Lukka H, Gu CS, et al. Randomized trial of a hypofractionated radiation regimen for the treatment of localized prostate cancer. J Clin Oncol. 2017;35:1884-1890. doi:10.1200/JCO.2016.71.7397
- Lee WR, Dignam JJ, Amin MB, et al. Long-term analysis of NRG Oncology RTOG 0415: a randomized phase III noninferiority study comparing two fractionation schedules in patients with low-risk prostate cancer. J Clin Oncol. 2024;42:2377-2381. doi:10.1200/JCO.23.02445
- de Vries KC, Wortel RC, Oomen-de Hoop E, et al. Hypofractionated versus conventionally fractionated radiation therapy for patients with intermediate- or high-risk, localized, prostate cancer: 7-year outcomes from the randomized, multicenter, open-label, phase 3 HYPRO trial. Int J Radiat Oncol Biol Phys. 2020;106:108-115. doi:10.1016/j.ijrobp.2019.09.007
- Incrocci L, Wortel RC, Alemayehu WG, et al. Hypofractionated versus conventionally fractionated radiotherapy for patients with localised prostate cancer (HYPRO): final efficacy results from a randomised, multicentre, open-label, phase 3 trial. Lancet Oncol. 2016;17:1061-1069. doi:10.1016/S1470-2045(16)30070-5
- Widmark A, Gunnlaugsson A, Beckman L, et al. Ultra-hypofractionated versus conventionally fractionated radiotherapy for prostate cancer: 5-year outcomes of the HYPO-RT-PC randomised, non-inferiority, phase 3 trial. Lancet. 2019;394:385-395. doi:10.1016/S0140-6736(19)31131-6
- Brenner DJ, Hall EJ. Fractionation and protraction for radiotherapy of prostate carcinoma. Int J Radiat Oncol Biol Phys. 1999;43:1095-101. doi:10.1016/s0360-3016(98)00438-6
- Dasu A. Is the alpha/beta value for prostate tumours low enough to be safely used in clinical trials? Clin Oncol (R Coll Radiol). 2007;19:289-301. doi:10.1016/j.clon.2007.02.007
- Garcia-Barros M, Paris F, Cordon-Cardo C, et al. Tumor response to radiotherapy regulated by endothelial cell apoptosis. Science. 2003;300:1155-1159. doi:10.1126/science.1082504
- Gulliford S, Hall E, Dearnaley D. Hypofractionation trials and radiobiology of prostate cancer. Oncoscience. 2017;4:27-28. doi:10.18632/oncoscience.347
- Fuks Z, Kolesnick R. Engaging the vascular component of the tumor response. Cancer Cell. 2005;8:89-91. doi:10.1016/j.ccr.2005.07.014
- van As N, Griffin C, Tree A, et al. Phase 3 Trial of stereotactic body radiotherapy in localized prostate cancer. N Engl J Med. Oct 17 2024;391:1413-1425. doi:10.1056/NEJMoa2403365
- National Comprehensive Cancer Network. NCCN Guidelines Version 5. 2026 Prostate Cancer. Accessed March 24, 2026. https://www.nccn.org/professionals/physician_gls/pdf/prostate.pdf
- Lawton CA, Michalski J, El-Naqa I, et al. RTOG GU radiation oncology specialists reach consensus on pelvic lymph node volumes for high-risk prostate cancer. Int J Radiat Oncol Biol Phys. 2009;74:383-387. doi:10.1016/j.ijrobp.2008.08.002
- Lehrer EJ, Kishan AU, Yu JB, et al. Ultrahypofractionated versus hypofractionated and conventionally fractionated radiation therapy for localized prostate cancer: a systematic review and meta-analysis of phase III randomized trials. Radiother Oncol. 2020;148:235-242. doi:10.1016/j.radonc.2020.04.037
- De Cooman B, Debacker T, Adams T, et al. Stereotactic body radiotherapy (SBRT) as a treatment for localized prostate cancer: a retrospective analysis. Radiat Oncol. 2025;20:25. doi:10.1186/s13014-025-02598-8
- Fuller DB, Falchook AD, Crabtree T, et al. Phase 2 multicenter trial of heterogeneous-dosing stereotactic body radiotherapy for low- and intermediate-risk prostate cancer: 5-year outcomes. Eur Urol Oncol. 2018;1:540-547. doi:10.1016/j.euo.2018.06.013
- Jackson WC, Silva J, Hartman HE, et al. Stereotactic body radiation therapy for localized prostate cancer: a systematic review and meta-analysis of over 6,000 patients treated on prospective studies. Int J Radiat Oncol Biol Phys. 2019;104:778-789. doi:10.1016/j.ijrobp.2019.03.051
- Meier RM, Bloch DA, Cotrutz C, et al. Multicenter trial of stereotactic body radiation therapy for low- and intermediate-risk prostate cancer: survival and toxicity endpoints. nt J Radiat Oncol Biol Phys. 2018;102:296-303. doi:10.1016/j.ijrobp.2018.05.040
- Quon HC, Ong A, Cheung P, et al. Once-weekly versus every-other-day stereotactic body radiotherapy in patients with prostate cancer (PATRIOT): a phase 2 randomized trial. Radiother Oncol. 2018;127:206-212. doi:10.1016/j.radonc.2018.02.029
- Zelefsky MJ, Kollmeier M, McBride S, et al. Five-year outcomes of a phase 1 dose-escalation study using stereotactic body radiosurgery for patients with low-risk and intermediate-risk prostate cancer. Int J Radiat Oncol Biol Phys. 2019;104:42-49. doi:10.1016/j.ijrobp.2018.12.045
- Siegel RL, Kratzer TB, Giaquinto AN, et al. Cancer statistics, 2025. CA Cancer J Clin. 2025;75:10-45. doi:10.3322/caac.21871
- Donovan JL, Hamdy FC, Lane JA, et al. Patient-reported outcomes after monitoring, surgery, or radiotherapy for prostate cancer. N Engl J Med. 2016;375:1425-1437. doi:10.1056/NEJMoa1606221
- Hamdy FC, Donovan JL, Lane JA, et al. 10-year outcomes after monitoring, surgery, or radiotherapy for localized prostate cancer. N Engl J Med. 2016;375:1415-1424. doi:10.1056/NEJMoa1606220
- Dearnaley D, Syndikus I, Mossop H, et al. Conventional versus hypofractionated high-dose intensity-modulated radiotherapy for prostate cancer: 5-year outcomes of the randomised, non-inferiority, phase 3 CHHiP trial. Lancet Oncol. 2016;17:1047-1060. doi:10.1016/S1470-2045(16)30102-4
- Catton CN, Lukka H, Gu CS, et al. Randomized trial of a hypofractionated radiation regimen for the treatment of localized prostate cancer. J Clin Oncol. 2017;35:1884-1890. doi:10.1200/JCO.2016.71.7397
- Lee WR, Dignam JJ, Amin MB, et al. Long-term analysis of NRG Oncology RTOG 0415: a randomized phase III noninferiority study comparing two fractionation schedules in patients with low-risk prostate cancer. J Clin Oncol. 2024;42:2377-2381. doi:10.1200/JCO.23.02445
- de Vries KC, Wortel RC, Oomen-de Hoop E, et al. Hypofractionated versus conventionally fractionated radiation therapy for patients with intermediate- or high-risk, localized, prostate cancer: 7-year outcomes from the randomized, multicenter, open-label, phase 3 HYPRO trial. Int J Radiat Oncol Biol Phys. 2020;106:108-115. doi:10.1016/j.ijrobp.2019.09.007
- Incrocci L, Wortel RC, Alemayehu WG, et al. Hypofractionated versus conventionally fractionated radiotherapy for patients with localised prostate cancer (HYPRO): final efficacy results from a randomised, multicentre, open-label, phase 3 trial. Lancet Oncol. 2016;17:1061-1069. doi:10.1016/S1470-2045(16)30070-5
- Widmark A, Gunnlaugsson A, Beckman L, et al. Ultra-hypofractionated versus conventionally fractionated radiotherapy for prostate cancer: 5-year outcomes of the HYPO-RT-PC randomised, non-inferiority, phase 3 trial. Lancet. 2019;394:385-395. doi:10.1016/S0140-6736(19)31131-6
- Brenner DJ, Hall EJ. Fractionation and protraction for radiotherapy of prostate carcinoma. Int J Radiat Oncol Biol Phys. 1999;43:1095-101. doi:10.1016/s0360-3016(98)00438-6
- Dasu A. Is the alpha/beta value for prostate tumours low enough to be safely used in clinical trials? Clin Oncol (R Coll Radiol). 2007;19:289-301. doi:10.1016/j.clon.2007.02.007
- Garcia-Barros M, Paris F, Cordon-Cardo C, et al. Tumor response to radiotherapy regulated by endothelial cell apoptosis. Science. 2003;300:1155-1159. doi:10.1126/science.1082504
- Gulliford S, Hall E, Dearnaley D. Hypofractionation trials and radiobiology of prostate cancer. Oncoscience. 2017;4:27-28. doi:10.18632/oncoscience.347
- Fuks Z, Kolesnick R. Engaging the vascular component of the tumor response. Cancer Cell. 2005;8:89-91. doi:10.1016/j.ccr.2005.07.014
- van As N, Griffin C, Tree A, et al. Phase 3 Trial of stereotactic body radiotherapy in localized prostate cancer. N Engl J Med. Oct 17 2024;391:1413-1425. doi:10.1056/NEJMoa2403365
- National Comprehensive Cancer Network. NCCN Guidelines Version 5. 2026 Prostate Cancer. Accessed March 24, 2026. https://www.nccn.org/professionals/physician_gls/pdf/prostate.pdf
- Lawton CA, Michalski J, El-Naqa I, et al. RTOG GU radiation oncology specialists reach consensus on pelvic lymph node volumes for high-risk prostate cancer. Int J Radiat Oncol Biol Phys. 2009;74:383-387. doi:10.1016/j.ijrobp.2008.08.002
- Lehrer EJ, Kishan AU, Yu JB, et al. Ultrahypofractionated versus hypofractionated and conventionally fractionated radiation therapy for localized prostate cancer: a systematic review and meta-analysis of phase III randomized trials. Radiother Oncol. 2020;148:235-242. doi:10.1016/j.radonc.2020.04.037
- De Cooman B, Debacker T, Adams T, et al. Stereotactic body radiotherapy (SBRT) as a treatment for localized prostate cancer: a retrospective analysis. Radiat Oncol. 2025;20:25. doi:10.1186/s13014-025-02598-8
- Fuller DB, Falchook AD, Crabtree T, et al. Phase 2 multicenter trial of heterogeneous-dosing stereotactic body radiotherapy for low- and intermediate-risk prostate cancer: 5-year outcomes. Eur Urol Oncol. 2018;1:540-547. doi:10.1016/j.euo.2018.06.013
- Jackson WC, Silva J, Hartman HE, et al. Stereotactic body radiation therapy for localized prostate cancer: a systematic review and meta-analysis of over 6,000 patients treated on prospective studies. Int J Radiat Oncol Biol Phys. 2019;104:778-789. doi:10.1016/j.ijrobp.2019.03.051
- Meier RM, Bloch DA, Cotrutz C, et al. Multicenter trial of stereotactic body radiation therapy for low- and intermediate-risk prostate cancer: survival and toxicity endpoints. nt J Radiat Oncol Biol Phys. 2018;102:296-303. doi:10.1016/j.ijrobp.2018.05.040
- Quon HC, Ong A, Cheung P, et al. Once-weekly versus every-other-day stereotactic body radiotherapy in patients with prostate cancer (PATRIOT): a phase 2 randomized trial. Radiother Oncol. 2018;127:206-212. doi:10.1016/j.radonc.2018.02.029
- Zelefsky MJ, Kollmeier M, McBride S, et al. Five-year outcomes of a phase 1 dose-escalation study using stereotactic body radiosurgery for patients with low-risk and intermediate-risk prostate cancer. Int J Radiat Oncol Biol Phys. 2019;104:42-49. doi:10.1016/j.ijrobp.2018.12.045
Early Outcomes of Stereotactic Body Radiotherapy for Localized Prostate Cancer: A Retrospective Analysis
Early Outcomes of Stereotactic Body Radiotherapy for Localized Prostate Cancer: A Retrospective Analysis
Capturing Pathology Workload Associated With Precision Oncology
Capturing Pathology Workload Associated With Precision Oncology
Precision oncology (PO) is cancer treatment individualized to the special characteristics of a patient’s tumor. It has become standard care for most patients with advanced cancer. Advances in molecular cell biology and immunology have identified numerous targets and many therapies have been developed as a result. Molecular testing and targeted therapy are typically covered by insurance, even when inflation-adjusted price growth is considered.1 Barriers remain, however, and pathologists are uniquely qualified to address some of the challenges.2
Most US laboratories do not perform molecular diagnostic tests for PO, particularly comprehensive evaluation of multiple targets by next-generation sequencing, or other techniques. Instead, these tests are sent to reference laboratories. The workload associated with referral testing is an obstacle to increased use of such tests. Despite guideline recommendations, a minority of indicated tests are performed.3 This is true even when testing costs are covered by clinical trials or grants, such as those in the Veterans Health Administration (VHA).
The main characteristic of successful PO programs is a multidisciplinary commitment, including pathology involvement in molecular tumor boards and assistance with test selection, tissue collection, and result interpretation.2 This, however, adds to the workload for the pathology department, an underappreciated phenomenon in the context of pathology workforce shortages.4
Workforce shortages impact all occupations in the laboratory setting. Though the shortage of medical technologists in clinical pathology laboratories has repeatedly been identified as critical at the VHA as well as in the private sector, the same cannot be said for staff shortages in anatomic pathology laboratories. Thus, the hospital laboratory divisions are concerned with biopsy or resection tissue specimens as opposed to the bodily fluid specimens that predominate in clinical laboratories.5 The lack of accurate data on histopathology technicians and technologists has precluded the degree of recognition seen for medical technologists. In labor statistics, these occupations are often obscured by inclusion with other jobs in broad categories such as medical and clinical laboratory technologists and technicians.6 Vacancy—the principal metric used to assess medical laboratory workforce shortage—fails to account for positions that are eventually eliminated after remaining vacant for prolonged periods, positions not replaced as a result of ambitious efficiency measures, or positions that were never created due to insufficient funding, reasons for administrative disapproval, or coverage by laboratory professionals working extra shifts or second jobs.7
Increased demand for pathologists is suggested by a 42% increase in workload per pathologist over the last decade, while a shortage is suggested by decreases in absolute and population-adjusted numbers of pathologists.8,9 An influx of pathologists is not an expected remedy due to the global decline in medical graduates pursuing careers in the field.8
Approximations for required labor and potential revenue generation are necessary to justify creation of pathology positions. This work mostly has gone uncaptured due to the limitations of Current Procedural Terminology (CPT) codes. Few laboratories have consistently used the 88363, 88325, and G0452 CPT codes. The pathology clinical consultation CPT codes (80503-80506) released in 2022 enhance acquisition of this work. The new codes replace 80500 and 80502 and allow for more precise identification of any work requiring medical judgment that a pathologist does at the request of another qualified health care professional (HCP) or as required by federal or state regulation.
The codes can be used to bill for associated time spent reviewing health records, communicating with other HCPs, placing orders, and documentation. An HCP can bill according to level of medical decision-making (MDM) or time spent by the consulting pathologist. Code 80503 can be billed for 5 to 20 minutes of a pathologist's time, 80504 for 21 to 40 minutes, 80505 for 41 to 60 minutes, and 80506 for each additional 30 minutes after the first hour. Levels of MDM (low, moderate, and high) are defined as for other evaluation and management services. A consultation report must be generated and contain documentation of the consultation request, pathologist interpretation, and justification for the level of service associated with the chosen code. Relative value units (RVUs) and reimbursement associated with each as well as other consultation-related codes are available in Table 1.
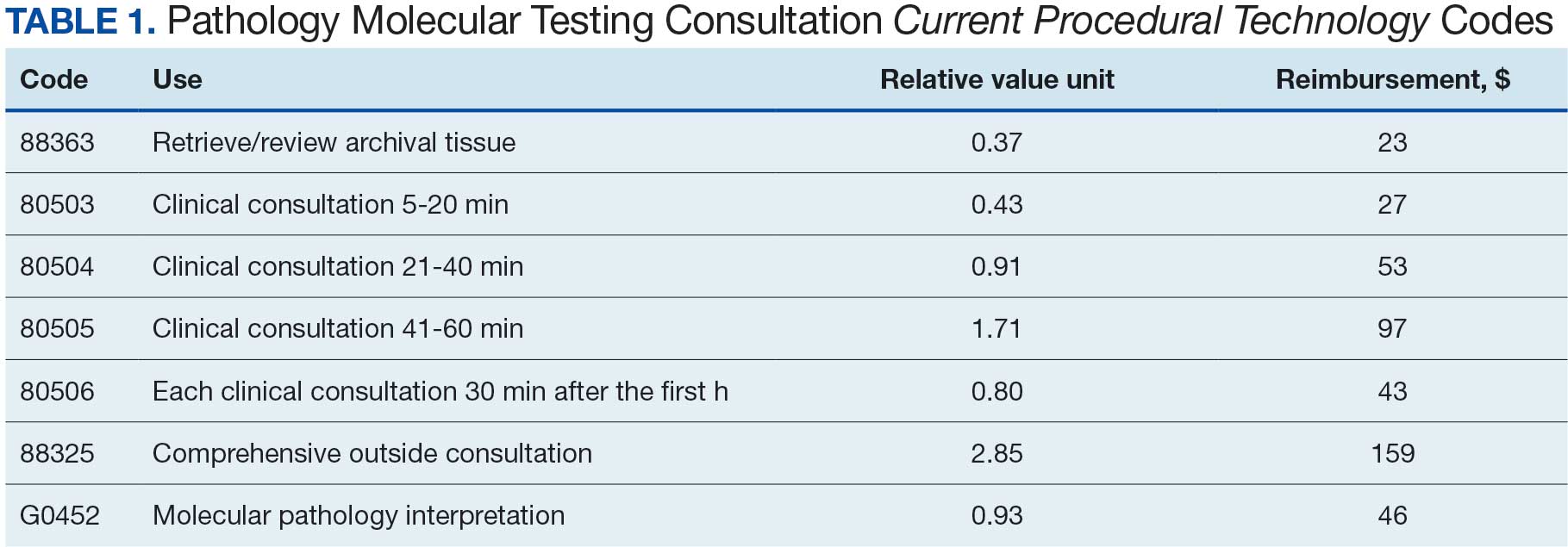
This article outlines how the pathology time investment associated with anatomic pathology molecular testing at the Kansas City Veterans Affairs Medical Center (KCVAMC) can be captured using the consultation process and new CPT codes. Staff included 4 pathologists, 3 histotechnologists, 1 histology supervisor, 1 grossing room technician, and 1 cytotechnologist, 1 cytology technician.
METHODS
The AP molecular testing consultation process at the KCVAMC was mapped, with the average time measured for each step (Figure). AP records for 2021 were reviewed to determine the number of AP molecular send out tests. Cumulative time investment was calculated in hours and a theoretical number of RVUs was calculated using the new pathology clinical consultation CPT codes (80503-80506). This theoretical number of RVUs was compared with the total AP RVUs generated in 2021 to determine a potential increase in RVUs with use of the new CPT codes to capture pathology work associated with AP molecular testing consultations.
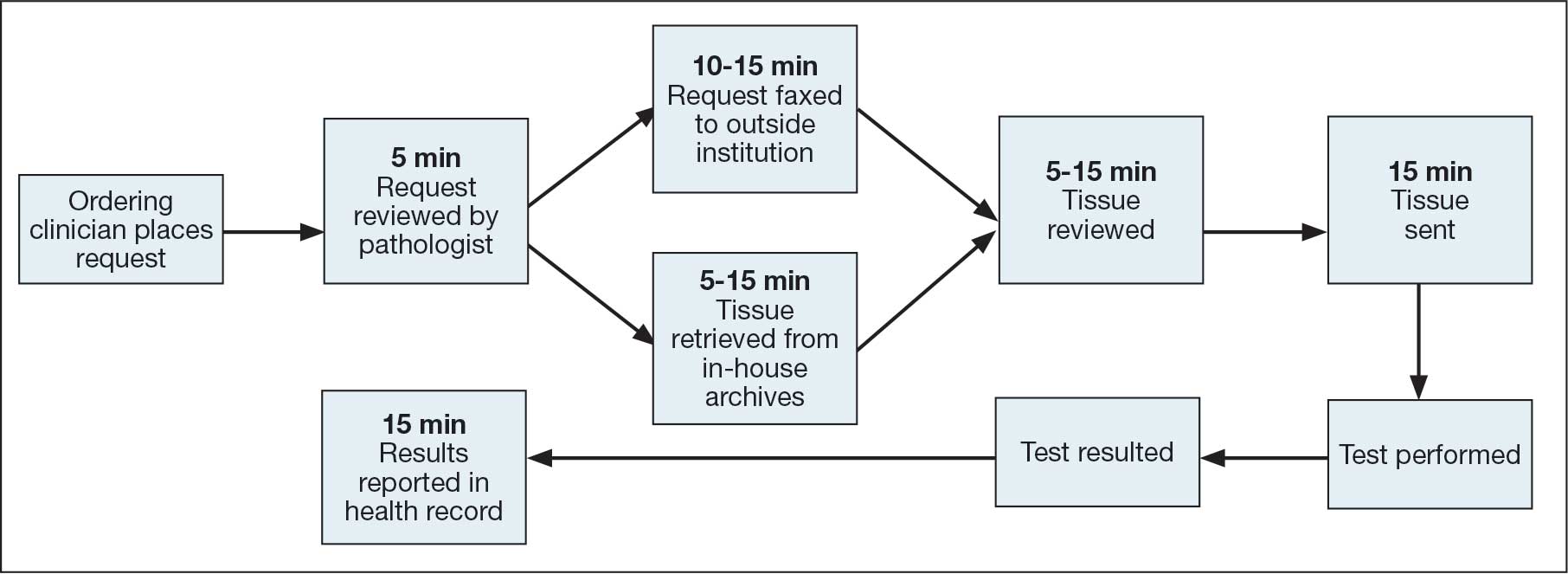
RESULTS
From 2021 to 2023, there were 21,021 AP cases at the KCVAMC. Basal cell carcinomas and squamous cell carcinomas of the skin were excluded because they comprise most cancer cases but almost never necessitate AP molecular test consultations. A total of 2118 cancer cases were included, representing 10.1% of all cases. Ancillary AP molecular send-out tests were performed on 1338 (6.4%) cases. Since ancillary tissue tests are requested by consultation at the KCVAMC, this resulted in 1338 consultations (Table 2).
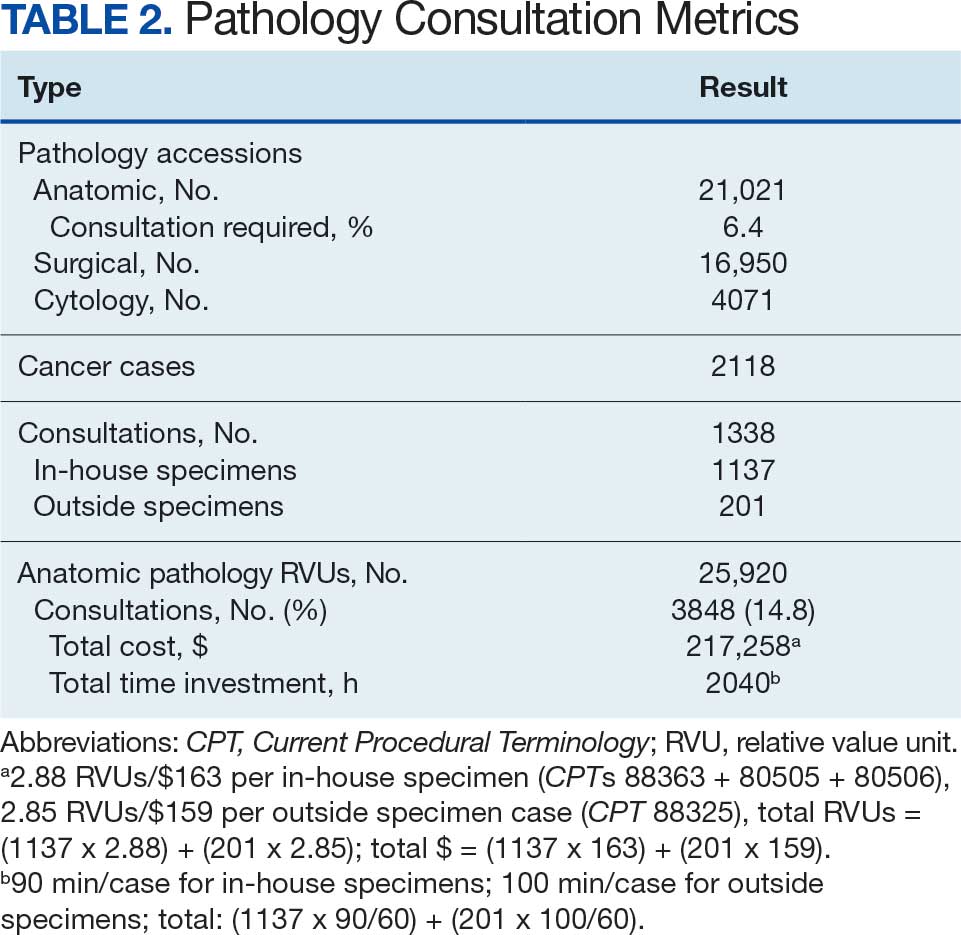
The time to complete a consultation was measured by calculating the mean time required to complete each step (Table 3). With in-house specimen consultations requiring 90 minutes each and outside specimen consultations requiring 100 minutes each, a total of 2040 hours of pathology staff time was necessary to complete associated consultations. Billing for this time with the new pathology clinical consultation CPT codes would generate 3847 RVUs, which would have equated to 14.8% (3847/25,920) of the anatomic pathology RVUs.

DISCUSSION
When considering the lengths of time for tasks associated with each consultation, it is important to remember that the volume (2-3 daily), was insufficient to meaningfully benefit from batching. Thus, waiting to perform a particular task until it was needed for multiple cases reduced the inefficiency associated with starting and switching between tasks. Both the Computerized Patient Record System and VistA had to be reopened, reauthenticated, and reloaded for each step that required use of the health record, printer, or fax machine. Faxes also required waiting for transmission and printed confirmation of successful transmission. As a result, the time values denoted for each step are likely underestimated, as the effect of interruptions is significant and not reflected in the estimates recorded.10
This analysis has demonstrated that PO entails a significant amount of work for pathology departments. To determine and maintain appropriate staffing models, this work must be captured and reimbursed. Unlike other pathology work, which is performed in-house and reimbursed for the associated test, a significant proportion of PO testing is sent out. Even if more reliable assays are developed, the physical processes of sending out samples and reporting test results cannot be outsourced. Independent and commensurate reimbursement methods are necessary to allow for this work and PO.
CMS included new pathology clinical consultation codes that may be used to bill for some of this work according to the 2022 physician fee schedule due to advocacy work by the College of American Pathologists and the American Medical Association CPT editorial panel.11
CONCLUSIONS
This analysis found that adoption of PO may present a significant amount of additional work for pathology departments. To determine and maintain appropriate staffing models, work completed by pathologists in this manner must be recorded and reimbursed. Pathologists need to be trained and encouraged to use these CPT codes and bill for the work described in this article. The increased revenue will allow for additional positions to alleviate the burdens imposed by understaffing so that pathology can function as a facilitator of PO rather than as a barrier to it.
- Wilson LE, Greiner MA, Altomare I, et al. Rapid rise in the cost of targeted cancer therapies for Medicare patients with solid tumors from 2006 to 2015. J Geriatr Oncol. 2021;12:375-380. doi:10.1016/j.jgo.2020.11.007
- Ersek JL, Black LJ, Thompson MA, et al. Implementing precision medicine programs and clinical trials in the community-based oncology practice: barriers and best practices. Am Soc Clin Oncol Educ Book. 2018;38:188-196. doi:10.1200/EDBK_200633
- Inal C, Yilmaz E, Cheng H, et al. Effect of reflex testing by pathologists on molecular testing rates in lung cancer patients: experience from a community-based academic center. J Clin Oncol. 2014;32:8098. doi:10.1200/jco.2014.32.15_suppl.8098
- Robboy SJ, Gupta S, Crawford JM, et al. The pathologist workforce in the United States: II. an interactive modeling tool for analyzing future qualitative and quantitative staffing demands for services. Arch Pathol Lab Med. 2015;139:1413-1430. doi:10.5858/arpa.2014-0559-OA
- OIG determination of Veterans Health Administration’s occupational staffing shortages fiscal year 2021. Department of Veterans Affairs OIG. September 28, 2021. Accessed January 30, 2026. https://www.oversight.gov/report/VA/OIG-determination-veterans-health-administrations-occupational-staffing-shortages-fiscal
- Zanto S, Cremeans L, Deutsch-Keahey D, et al. Addressing the clinical laboratory workforce shortage. The American Society for Clinical Laboratory Science. July 2, 2020. Accessed January 30, 2026. https://ascls.org/addressing-the-clinical-laboratory-workforce-shortage/
- Bennett A, Garcia E, Schulze M, et al. Building a laboratory workforce to meet the future: ASCP Task Force on the Laboratory Professionals Workforce. Am J Clin Pathol. 2014;141:154-167. doi:10.1309/AJCPIV2OG8TEGHHZ
- Fielder T, Watts F, Howden C, et al. Why choose a pathology career? Arch Pathol Lab Med. 2022;146:903-910. doi:10.5858/arpa.2021-0118-OA
- Metter DM, Colgan TJ, Leung ST, et al. Trends in the US and Canadian pathologist workforces from 2007 to 2017. JAMA Netw Open. 2019;2:e194337. doi:10.1001/jamanetworkopen.2019.4337
- Schulte B. Work interruptions can cost you 6 hours a day. An efficiency expert explains how to avoid them. The Washington Post. June 1, 2015. Accessed January 30, 2026. https://www.washingtonpost.com/news/inspired-life/wp/2015/06/01/interruptions-at-work-can-cost-you-up-to-6-hours-a-day-heres-how-to-avoid-them/
- Fiegl C. Medicare adopts new clinical consult billing codes. College of American Pathologists Today. December 2021. Accessed January 30, 2026. https://www.captodayonline.com/medicare-adopts-new-clinical-consult-billing-code
Precision oncology (PO) is cancer treatment individualized to the special characteristics of a patient’s tumor. It has become standard care for most patients with advanced cancer. Advances in molecular cell biology and immunology have identified numerous targets and many therapies have been developed as a result. Molecular testing and targeted therapy are typically covered by insurance, even when inflation-adjusted price growth is considered.1 Barriers remain, however, and pathologists are uniquely qualified to address some of the challenges.2
Most US laboratories do not perform molecular diagnostic tests for PO, particularly comprehensive evaluation of multiple targets by next-generation sequencing, or other techniques. Instead, these tests are sent to reference laboratories. The workload associated with referral testing is an obstacle to increased use of such tests. Despite guideline recommendations, a minority of indicated tests are performed.3 This is true even when testing costs are covered by clinical trials or grants, such as those in the Veterans Health Administration (VHA).
The main characteristic of successful PO programs is a multidisciplinary commitment, including pathology involvement in molecular tumor boards and assistance with test selection, tissue collection, and result interpretation.2 This, however, adds to the workload for the pathology department, an underappreciated phenomenon in the context of pathology workforce shortages.4
Workforce shortages impact all occupations in the laboratory setting. Though the shortage of medical technologists in clinical pathology laboratories has repeatedly been identified as critical at the VHA as well as in the private sector, the same cannot be said for staff shortages in anatomic pathology laboratories. Thus, the hospital laboratory divisions are concerned with biopsy or resection tissue specimens as opposed to the bodily fluid specimens that predominate in clinical laboratories.5 The lack of accurate data on histopathology technicians and technologists has precluded the degree of recognition seen for medical technologists. In labor statistics, these occupations are often obscured by inclusion with other jobs in broad categories such as medical and clinical laboratory technologists and technicians.6 Vacancy—the principal metric used to assess medical laboratory workforce shortage—fails to account for positions that are eventually eliminated after remaining vacant for prolonged periods, positions not replaced as a result of ambitious efficiency measures, or positions that were never created due to insufficient funding, reasons for administrative disapproval, or coverage by laboratory professionals working extra shifts or second jobs.7
Increased demand for pathologists is suggested by a 42% increase in workload per pathologist over the last decade, while a shortage is suggested by decreases in absolute and population-adjusted numbers of pathologists.8,9 An influx of pathologists is not an expected remedy due to the global decline in medical graduates pursuing careers in the field.8
Approximations for required labor and potential revenue generation are necessary to justify creation of pathology positions. This work mostly has gone uncaptured due to the limitations of Current Procedural Terminology (CPT) codes. Few laboratories have consistently used the 88363, 88325, and G0452 CPT codes. The pathology clinical consultation CPT codes (80503-80506) released in 2022 enhance acquisition of this work. The new codes replace 80500 and 80502 and allow for more precise identification of any work requiring medical judgment that a pathologist does at the request of another qualified health care professional (HCP) or as required by federal or state regulation.
The codes can be used to bill for associated time spent reviewing health records, communicating with other HCPs, placing orders, and documentation. An HCP can bill according to level of medical decision-making (MDM) or time spent by the consulting pathologist. Code 80503 can be billed for 5 to 20 minutes of a pathologist's time, 80504 for 21 to 40 minutes, 80505 for 41 to 60 minutes, and 80506 for each additional 30 minutes after the first hour. Levels of MDM (low, moderate, and high) are defined as for other evaluation and management services. A consultation report must be generated and contain documentation of the consultation request, pathologist interpretation, and justification for the level of service associated with the chosen code. Relative value units (RVUs) and reimbursement associated with each as well as other consultation-related codes are available in Table 1.

This article outlines how the pathology time investment associated with anatomic pathology molecular testing at the Kansas City Veterans Affairs Medical Center (KCVAMC) can be captured using the consultation process and new CPT codes. Staff included 4 pathologists, 3 histotechnologists, 1 histology supervisor, 1 grossing room technician, and 1 cytotechnologist, 1 cytology technician.
METHODS
The AP molecular testing consultation process at the KCVAMC was mapped, with the average time measured for each step (Figure). AP records for 2021 were reviewed to determine the number of AP molecular send out tests. Cumulative time investment was calculated in hours and a theoretical number of RVUs was calculated using the new pathology clinical consultation CPT codes (80503-80506). This theoretical number of RVUs was compared with the total AP RVUs generated in 2021 to determine a potential increase in RVUs with use of the new CPT codes to capture pathology work associated with AP molecular testing consultations.

RESULTS
From 2021 to 2023, there were 21,021 AP cases at the KCVAMC. Basal cell carcinomas and squamous cell carcinomas of the skin were excluded because they comprise most cancer cases but almost never necessitate AP molecular test consultations. A total of 2118 cancer cases were included, representing 10.1% of all cases. Ancillary AP molecular send-out tests were performed on 1338 (6.4%) cases. Since ancillary tissue tests are requested by consultation at the KCVAMC, this resulted in 1338 consultations (Table 2).

The time to complete a consultation was measured by calculating the mean time required to complete each step (Table 3). With in-house specimen consultations requiring 90 minutes each and outside specimen consultations requiring 100 minutes each, a total of 2040 hours of pathology staff time was necessary to complete associated consultations. Billing for this time with the new pathology clinical consultation CPT codes would generate 3847 RVUs, which would have equated to 14.8% (3847/25,920) of the anatomic pathology RVUs.

DISCUSSION
When considering the lengths of time for tasks associated with each consultation, it is important to remember that the volume (2-3 daily), was insufficient to meaningfully benefit from batching. Thus, waiting to perform a particular task until it was needed for multiple cases reduced the inefficiency associated with starting and switching between tasks. Both the Computerized Patient Record System and VistA had to be reopened, reauthenticated, and reloaded for each step that required use of the health record, printer, or fax machine. Faxes also required waiting for transmission and printed confirmation of successful transmission. As a result, the time values denoted for each step are likely underestimated, as the effect of interruptions is significant and not reflected in the estimates recorded.10
This analysis has demonstrated that PO entails a significant amount of work for pathology departments. To determine and maintain appropriate staffing models, this work must be captured and reimbursed. Unlike other pathology work, which is performed in-house and reimbursed for the associated test, a significant proportion of PO testing is sent out. Even if more reliable assays are developed, the physical processes of sending out samples and reporting test results cannot be outsourced. Independent and commensurate reimbursement methods are necessary to allow for this work and PO.
CMS included new pathology clinical consultation codes that may be used to bill for some of this work according to the 2022 physician fee schedule due to advocacy work by the College of American Pathologists and the American Medical Association CPT editorial panel.11
CONCLUSIONS
This analysis found that adoption of PO may present a significant amount of additional work for pathology departments. To determine and maintain appropriate staffing models, work completed by pathologists in this manner must be recorded and reimbursed. Pathologists need to be trained and encouraged to use these CPT codes and bill for the work described in this article. The increased revenue will allow for additional positions to alleviate the burdens imposed by understaffing so that pathology can function as a facilitator of PO rather than as a barrier to it.
Precision oncology (PO) is cancer treatment individualized to the special characteristics of a patient’s tumor. It has become standard care for most patients with advanced cancer. Advances in molecular cell biology and immunology have identified numerous targets and many therapies have been developed as a result. Molecular testing and targeted therapy are typically covered by insurance, even when inflation-adjusted price growth is considered.1 Barriers remain, however, and pathologists are uniquely qualified to address some of the challenges.2
Most US laboratories do not perform molecular diagnostic tests for PO, particularly comprehensive evaluation of multiple targets by next-generation sequencing, or other techniques. Instead, these tests are sent to reference laboratories. The workload associated with referral testing is an obstacle to increased use of such tests. Despite guideline recommendations, a minority of indicated tests are performed.3 This is true even when testing costs are covered by clinical trials or grants, such as those in the Veterans Health Administration (VHA).
The main characteristic of successful PO programs is a multidisciplinary commitment, including pathology involvement in molecular tumor boards and assistance with test selection, tissue collection, and result interpretation.2 This, however, adds to the workload for the pathology department, an underappreciated phenomenon in the context of pathology workforce shortages.4
Workforce shortages impact all occupations in the laboratory setting. Though the shortage of medical technologists in clinical pathology laboratories has repeatedly been identified as critical at the VHA as well as in the private sector, the same cannot be said for staff shortages in anatomic pathology laboratories. Thus, the hospital laboratory divisions are concerned with biopsy or resection tissue specimens as opposed to the bodily fluid specimens that predominate in clinical laboratories.5 The lack of accurate data on histopathology technicians and technologists has precluded the degree of recognition seen for medical technologists. In labor statistics, these occupations are often obscured by inclusion with other jobs in broad categories such as medical and clinical laboratory technologists and technicians.6 Vacancy—the principal metric used to assess medical laboratory workforce shortage—fails to account for positions that are eventually eliminated after remaining vacant for prolonged periods, positions not replaced as a result of ambitious efficiency measures, or positions that were never created due to insufficient funding, reasons for administrative disapproval, or coverage by laboratory professionals working extra shifts or second jobs.7
Increased demand for pathologists is suggested by a 42% increase in workload per pathologist over the last decade, while a shortage is suggested by decreases in absolute and population-adjusted numbers of pathologists.8,9 An influx of pathologists is not an expected remedy due to the global decline in medical graduates pursuing careers in the field.8
Approximations for required labor and potential revenue generation are necessary to justify creation of pathology positions. This work mostly has gone uncaptured due to the limitations of Current Procedural Terminology (CPT) codes. Few laboratories have consistently used the 88363, 88325, and G0452 CPT codes. The pathology clinical consultation CPT codes (80503-80506) released in 2022 enhance acquisition of this work. The new codes replace 80500 and 80502 and allow for more precise identification of any work requiring medical judgment that a pathologist does at the request of another qualified health care professional (HCP) or as required by federal or state regulation.
The codes can be used to bill for associated time spent reviewing health records, communicating with other HCPs, placing orders, and documentation. An HCP can bill according to level of medical decision-making (MDM) or time spent by the consulting pathologist. Code 80503 can be billed for 5 to 20 minutes of a pathologist's time, 80504 for 21 to 40 minutes, 80505 for 41 to 60 minutes, and 80506 for each additional 30 minutes after the first hour. Levels of MDM (low, moderate, and high) are defined as for other evaluation and management services. A consultation report must be generated and contain documentation of the consultation request, pathologist interpretation, and justification for the level of service associated with the chosen code. Relative value units (RVUs) and reimbursement associated with each as well as other consultation-related codes are available in Table 1.

This article outlines how the pathology time investment associated with anatomic pathology molecular testing at the Kansas City Veterans Affairs Medical Center (KCVAMC) can be captured using the consultation process and new CPT codes. Staff included 4 pathologists, 3 histotechnologists, 1 histology supervisor, 1 grossing room technician, and 1 cytotechnologist, 1 cytology technician.
METHODS
The AP molecular testing consultation process at the KCVAMC was mapped, with the average time measured for each step (Figure). AP records for 2021 were reviewed to determine the number of AP molecular send out tests. Cumulative time investment was calculated in hours and a theoretical number of RVUs was calculated using the new pathology clinical consultation CPT codes (80503-80506). This theoretical number of RVUs was compared with the total AP RVUs generated in 2021 to determine a potential increase in RVUs with use of the new CPT codes to capture pathology work associated with AP molecular testing consultations.

RESULTS
From 2021 to 2023, there were 21,021 AP cases at the KCVAMC. Basal cell carcinomas and squamous cell carcinomas of the skin were excluded because they comprise most cancer cases but almost never necessitate AP molecular test consultations. A total of 2118 cancer cases were included, representing 10.1% of all cases. Ancillary AP molecular send-out tests were performed on 1338 (6.4%) cases. Since ancillary tissue tests are requested by consultation at the KCVAMC, this resulted in 1338 consultations (Table 2).

The time to complete a consultation was measured by calculating the mean time required to complete each step (Table 3). With in-house specimen consultations requiring 90 minutes each and outside specimen consultations requiring 100 minutes each, a total of 2040 hours of pathology staff time was necessary to complete associated consultations. Billing for this time with the new pathology clinical consultation CPT codes would generate 3847 RVUs, which would have equated to 14.8% (3847/25,920) of the anatomic pathology RVUs.

DISCUSSION
When considering the lengths of time for tasks associated with each consultation, it is important to remember that the volume (2-3 daily), was insufficient to meaningfully benefit from batching. Thus, waiting to perform a particular task until it was needed for multiple cases reduced the inefficiency associated with starting and switching between tasks. Both the Computerized Patient Record System and VistA had to be reopened, reauthenticated, and reloaded for each step that required use of the health record, printer, or fax machine. Faxes also required waiting for transmission and printed confirmation of successful transmission. As a result, the time values denoted for each step are likely underestimated, as the effect of interruptions is significant and not reflected in the estimates recorded.10
This analysis has demonstrated that PO entails a significant amount of work for pathology departments. To determine and maintain appropriate staffing models, this work must be captured and reimbursed. Unlike other pathology work, which is performed in-house and reimbursed for the associated test, a significant proportion of PO testing is sent out. Even if more reliable assays are developed, the physical processes of sending out samples and reporting test results cannot be outsourced. Independent and commensurate reimbursement methods are necessary to allow for this work and PO.
CMS included new pathology clinical consultation codes that may be used to bill for some of this work according to the 2022 physician fee schedule due to advocacy work by the College of American Pathologists and the American Medical Association CPT editorial panel.11
CONCLUSIONS
This analysis found that adoption of PO may present a significant amount of additional work for pathology departments. To determine and maintain appropriate staffing models, work completed by pathologists in this manner must be recorded and reimbursed. Pathologists need to be trained and encouraged to use these CPT codes and bill for the work described in this article. The increased revenue will allow for additional positions to alleviate the burdens imposed by understaffing so that pathology can function as a facilitator of PO rather than as a barrier to it.
- Wilson LE, Greiner MA, Altomare I, et al. Rapid rise in the cost of targeted cancer therapies for Medicare patients with solid tumors from 2006 to 2015. J Geriatr Oncol. 2021;12:375-380. doi:10.1016/j.jgo.2020.11.007
- Ersek JL, Black LJ, Thompson MA, et al. Implementing precision medicine programs and clinical trials in the community-based oncology practice: barriers and best practices. Am Soc Clin Oncol Educ Book. 2018;38:188-196. doi:10.1200/EDBK_200633
- Inal C, Yilmaz E, Cheng H, et al. Effect of reflex testing by pathologists on molecular testing rates in lung cancer patients: experience from a community-based academic center. J Clin Oncol. 2014;32:8098. doi:10.1200/jco.2014.32.15_suppl.8098
- Robboy SJ, Gupta S, Crawford JM, et al. The pathologist workforce in the United States: II. an interactive modeling tool for analyzing future qualitative and quantitative staffing demands for services. Arch Pathol Lab Med. 2015;139:1413-1430. doi:10.5858/arpa.2014-0559-OA
- OIG determination of Veterans Health Administration’s occupational staffing shortages fiscal year 2021. Department of Veterans Affairs OIG. September 28, 2021. Accessed January 30, 2026. https://www.oversight.gov/report/VA/OIG-determination-veterans-health-administrations-occupational-staffing-shortages-fiscal
- Zanto S, Cremeans L, Deutsch-Keahey D, et al. Addressing the clinical laboratory workforce shortage. The American Society for Clinical Laboratory Science. July 2, 2020. Accessed January 30, 2026. https://ascls.org/addressing-the-clinical-laboratory-workforce-shortage/
- Bennett A, Garcia E, Schulze M, et al. Building a laboratory workforce to meet the future: ASCP Task Force on the Laboratory Professionals Workforce. Am J Clin Pathol. 2014;141:154-167. doi:10.1309/AJCPIV2OG8TEGHHZ
- Fielder T, Watts F, Howden C, et al. Why choose a pathology career? Arch Pathol Lab Med. 2022;146:903-910. doi:10.5858/arpa.2021-0118-OA
- Metter DM, Colgan TJ, Leung ST, et al. Trends in the US and Canadian pathologist workforces from 2007 to 2017. JAMA Netw Open. 2019;2:e194337. doi:10.1001/jamanetworkopen.2019.4337
- Schulte B. Work interruptions can cost you 6 hours a day. An efficiency expert explains how to avoid them. The Washington Post. June 1, 2015. Accessed January 30, 2026. https://www.washingtonpost.com/news/inspired-life/wp/2015/06/01/interruptions-at-work-can-cost-you-up-to-6-hours-a-day-heres-how-to-avoid-them/
- Fiegl C. Medicare adopts new clinical consult billing codes. College of American Pathologists Today. December 2021. Accessed January 30, 2026. https://www.captodayonline.com/medicare-adopts-new-clinical-consult-billing-code
- Wilson LE, Greiner MA, Altomare I, et al. Rapid rise in the cost of targeted cancer therapies for Medicare patients with solid tumors from 2006 to 2015. J Geriatr Oncol. 2021;12:375-380. doi:10.1016/j.jgo.2020.11.007
- Ersek JL, Black LJ, Thompson MA, et al. Implementing precision medicine programs and clinical trials in the community-based oncology practice: barriers and best practices. Am Soc Clin Oncol Educ Book. 2018;38:188-196. doi:10.1200/EDBK_200633
- Inal C, Yilmaz E, Cheng H, et al. Effect of reflex testing by pathologists on molecular testing rates in lung cancer patients: experience from a community-based academic center. J Clin Oncol. 2014;32:8098. doi:10.1200/jco.2014.32.15_suppl.8098
- Robboy SJ, Gupta S, Crawford JM, et al. The pathologist workforce in the United States: II. an interactive modeling tool for analyzing future qualitative and quantitative staffing demands for services. Arch Pathol Lab Med. 2015;139:1413-1430. doi:10.5858/arpa.2014-0559-OA
- OIG determination of Veterans Health Administration’s occupational staffing shortages fiscal year 2021. Department of Veterans Affairs OIG. September 28, 2021. Accessed January 30, 2026. https://www.oversight.gov/report/VA/OIG-determination-veterans-health-administrations-occupational-staffing-shortages-fiscal
- Zanto S, Cremeans L, Deutsch-Keahey D, et al. Addressing the clinical laboratory workforce shortage. The American Society for Clinical Laboratory Science. July 2, 2020. Accessed January 30, 2026. https://ascls.org/addressing-the-clinical-laboratory-workforce-shortage/
- Bennett A, Garcia E, Schulze M, et al. Building a laboratory workforce to meet the future: ASCP Task Force on the Laboratory Professionals Workforce. Am J Clin Pathol. 2014;141:154-167. doi:10.1309/AJCPIV2OG8TEGHHZ
- Fielder T, Watts F, Howden C, et al. Why choose a pathology career? Arch Pathol Lab Med. 2022;146:903-910. doi:10.5858/arpa.2021-0118-OA
- Metter DM, Colgan TJ, Leung ST, et al. Trends in the US and Canadian pathologist workforces from 2007 to 2017. JAMA Netw Open. 2019;2:e194337. doi:10.1001/jamanetworkopen.2019.4337
- Schulte B. Work interruptions can cost you 6 hours a day. An efficiency expert explains how to avoid them. The Washington Post. June 1, 2015. Accessed January 30, 2026. https://www.washingtonpost.com/news/inspired-life/wp/2015/06/01/interruptions-at-work-can-cost-you-up-to-6-hours-a-day-heres-how-to-avoid-them/
- Fiegl C. Medicare adopts new clinical consult billing codes. College of American Pathologists Today. December 2021. Accessed January 30, 2026. https://www.captodayonline.com/medicare-adopts-new-clinical-consult-billing-code
Capturing Pathology Workload Associated With Precision Oncology
Capturing Pathology Workload Associated With Precision Oncology
Investigating Real-World Tolerance and Dose Reductions of Oncology Multikinase Inhibitors in a VA Population
Investigating Real-World Tolerance and Dose Reductions of Oncology Multikinase Inhibitors in a VA Population
The US Department of Veterans Affairs (VA) annually treats around 450,000 veterans with cancer and diagnoses an additional 56,000.1,2 Oral multikinase inhibitors (MKIs) are widely used as targeted therapies for many different malignancies. Despite the ease of oral administration, these agents are often accompanied by significant adverse effects (AEs) and drug-drug interactions.3,4 Common AEs include hypertension, cutaneous reactions, gastrointestinal disturbances, proteinuria, and fatigue. Some serious outcomes that may occur are myocardial infarction, thrombosis, nephrotic syndrome, hemorrhage, hepatotoxicity, and gastrointestinal events.5,6 Due to poor tolerability of these AEs, dose reductions, frequent therapy holds, and discontinuation of therapy may occur.
The US Food and Drug Administration recognizes dosing challenges with novel therapies and has created the Oncology Center of Excellence (OCE) Project Optimus initiative to reform dose optimization in oncology drug development. The initiative aims to shift the focus from establishing dose regimens based on the maximum tolerated doses of cytotoxic chemotherapeutics to an emphasis on maximum efficacy, safety, and tolerability, which better reflect real-world dosing.7,8
MKIs can be challenging to manage because of the frequent toxicity-related dose reductions, interruptions, and discontinuations. In a multicenter retrospective study, Schnadig et al investigated dosing characteristics of first-line sunitinib for advanced renal cell carcinoma (RCC) and found that, among 114 patients who experienced AEs while taking sunitinib, 39.5% had dose reductions, 5.3% delayed therapy, 18.4% required additional supportive medications, and 22.8% discontinued sunitinib.9 Overall survival and median progression-free survival of these patients were lower than reported by Motzer et al in a phase 3 clinical trial.10 Schnadig et al concluded that patients treated with sunitinib for RCC in the community setting required more frequent dose reductions and had less time on therapy compared with patients in clinical trials, which ultimately impacted clinical outcomes.9
At the VA North Texas Health Care System (VANTHCS), patients with cancer have difficulty tolerating MKIs and often require dose alterations and/or discontinuation because of drug intolerance rather than discontinuation due to progression. Frequent dose adjustments for toxicity management can place more strain on patients and health care resources because of additional appointments, clinician time, and emergency department visits. Escalating drug costs can also cause concern when prescription doses are unused or changed frequently.
To capture and quantify prescribing practices and dose adjustments, this study evaluated the tolerability of MKIs at VANTHCS. This analysis may also guide clinicians in the selection of starting doses as well as dose titration expectations to optimize MKI therapy.
METHODS
This single-center, retrospective chart review analyzed patients receiving oral oncology MKIs for various malignancies at VANTHCS between January 1, 2014, and October 31, 2024. Participants included adults aged ≥ 18 years with a prescription for axitinib, cabozantinib, lenvatinib, pazopanib, regorafenib, sorafenib, or sunitinib initiated by the hematology/oncology service at VANTHCS. Patients were included if they had follow-up documentation with the hematology/oncology service and/or other VANTHCS clinicians outlining their course of therapy after MKI initiation. Patients were excluded if they did not have sufficient follow-up documentation (eg, transferred care to a non-VA health care practitioner [HCP], moved to another VA health care system), were enrolled in clinical trials, or were prescribed an MKI from a Care in the Community (CITC) prescriber. Electronic health record review and data collection were performed using the VA Computerized Patient Record System and Research Electronic Data Capture. Data were collected from the time of initiation to cessation of therapy and included information regarding therapy changes, progressive disease, and date of death, when available. Data collected included age, sex, race, comorbidities, date of death, type of malignancy and subtypes, cancer stage, MKI used (ie, drug, dose, frequency, schedule, and indication), dates of medication changes (ie, start, adjustment, hold, discontinuation), concurrent antineoplastic treatments, and AEs documented at times of dose change or interruption.
The primary outcome was MKI tolerance determined using relative dose intensity (RDI) and mean and median time on therapy. Two methods are used to calculate RDI that vary in how they approach time on therapy as outlined in Hawn et al.11 This study used method 2, which accounts for holds in therapy by comparing the actual duration of treatment with the duration expected according to treatment protocol. Method 1 compares the prescribed dose with the administered dose and does not adjust for holds.11 Using method 2, the RDI in this study was calculated by dividing the total actual dose given by the total indicated dose for the malignancy being treated, which accounts for duration of treatment.

The total actual dose was the strength, frequency, and days on therapy for each time frame of treatment multiplied together. This method accounted for all dose adjustments and time periods of treatment holds, including patient self-adjustments, prescriber-directed adjustments, and nonadherence determined by HCP documentation and/or prescription data. Similarly, the indicated total dose was calculated by multiplying the indicated strength, frequency, and all days that treatment should have occurred (time from start to finish). Indicated doses were derived from the prescribing information for each malignancy with the exception of sunitinib, for which the off-label dose of 37.5 mg daily was considered a full dose.12,13 The total indicated dose for axitinib was calculated by considering the dose escalation schedule from the prescribing information.
Patients who required dose reductions due to renal/hepatic impairments or drug-drug interactions had their total indicated dose calculated using dose adjustments listed in the prescribing information. The mean RDI for each MKI agent was calculated by averaging the RDI for each prescription. The overall combined mean RDI included the means of all the MKIs reviewed to avoid skewing the results toward an MKI with more prescriptions. RDIs were also calculated for each cancer type for each agent. Additional descriptive secondary outcomes included rates of AEs and adjustments in doses.
RESULTS
Electronic data extraction identified 278 patients with 366 MKI prescriptions, of which 108 veterans with 158 MKI prescriptions were excluded. The top reason for exclusion was patients managed through CITC. Ultimately, 170 veterans with 208 MKI prescriptions managed by the VANTHCS hematology/oncology clinic were included (Table 1). Among patients receiving MKIs, the mean age was 72.7 years, 98% were male, and 99% had metastatic disease.
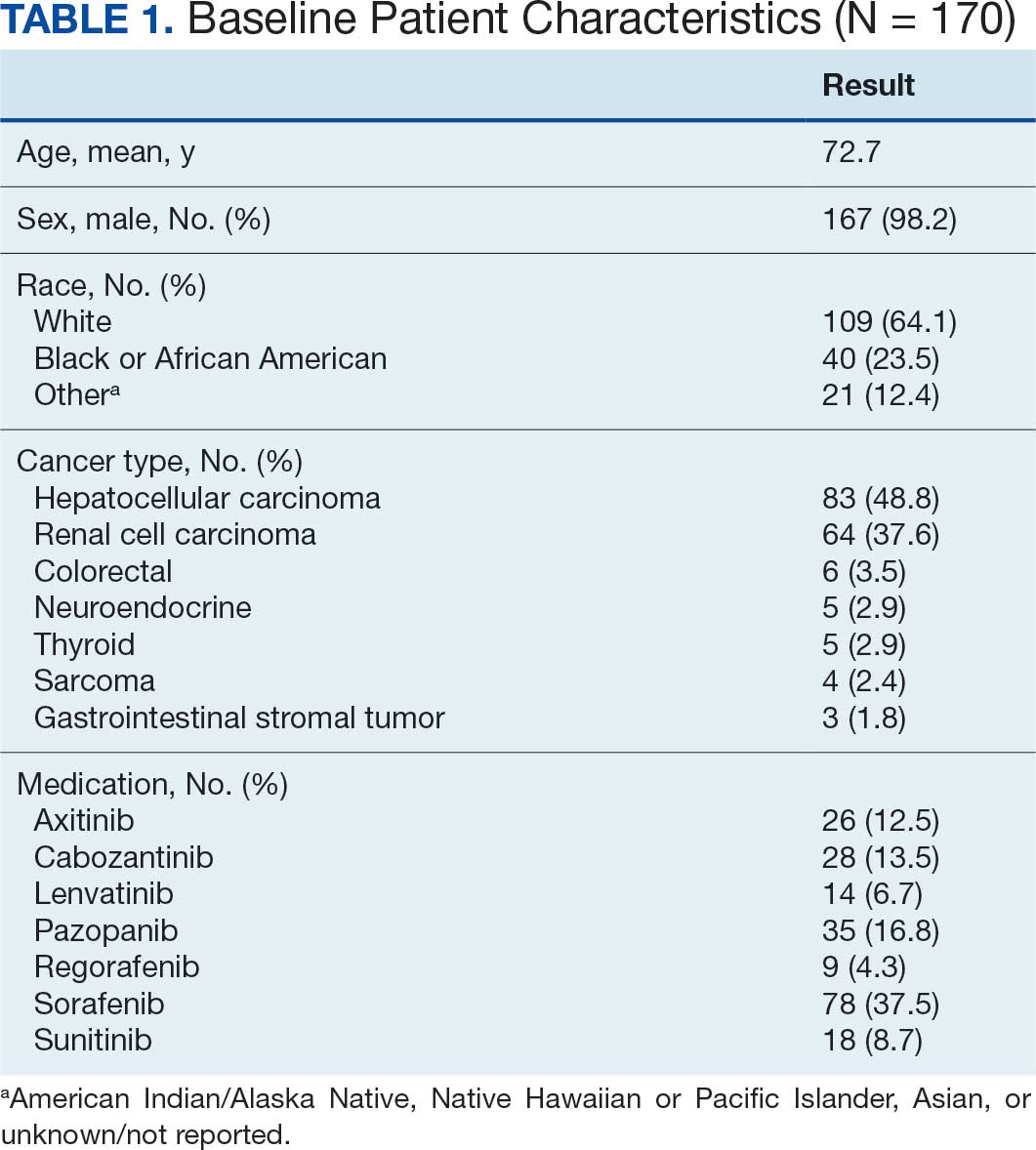
The overall combined mean MKI RDI was 67.5% using method 2 and ranged from 85.5% for sunitinib to 49.0% for sorafenib (Figure 1). Additional information regarding mean and median RDIs using method 2 is shown in Figure 1 and further subdivided by cancer type in Table 2. Median RDIs overall were similar to mean RDIs for most agents. Figure 2 indicates the mean and median time on therapy, reflecting time on therapy excluding days therapy was held. The overall combined mean and median days on therapy for all MKIs were 155 days and 95 days, respectively. Mean time on therapy depended on the agent used and ranged from 35 days (regorafenib) to 237 days (cabozantinib).
Of 208 MKI prescriptions, 127 (61.1%) were initiated at a reduced dose due to baseline concerns for tolerance such as performance status, frailty, and prior intolerance of other treatments. Eighty-one prescriptions (38.9%) were initiated at their indicated doses. Ninety prescriptions (43.3%) required dose reductions during treatment. Some MKI prescriptions had multiple dose increases and decreases, which is why RDI more accurately reflects dose adjustments. A total of 376 AEs that contributed to a dose adjustment, hold, or discontinuation occurred across all MKI prescriptions. The most common AEs were 82 failure-to-thrive events (21.8%) (fatigue, malaise, loss of appetite, reduced mobility, global decline), 79 gastrointestinal events (21.0%) (nausea, vomiting, diarrhea, abdominal pain), 62 dermatologic events (16.5%) (rash, hand-foot skin reactions, allergic response), 61 hepatic dysfunction events (16.2%) (liver enzyme elevations, hyperbilirubinemia), 40 cardiovascular events (10.6%) (hypertension, heart failure exacerbations, edema), and 33 renal dysfunction events (8.8%) (acute kidney injury, proteinuria) (Appendix 1).
DISCUSSION
The mean RDI of MKI prescriptions used in the veteran population at VANTHCS was about two-thirds of the indicated dose. These results indicate that most veterans required dose reductions and/or holds due to concerns over initial tolerance/performance status, worsening clinical condition, and/or intolerable AEs attributed to treatment. A retrospective study conducted by Denduluri et al suggested that an RDI of < 85% is a clinically meaningful reduction for traditional chemotherapy based on previous literature.14 However, it is less clear what RDI should be expected specifically for MKIs in real-world populations. The MKI phase 3 approval trials in RCC for axitinib, lenvatinib, and sunitinib found median RDIs of 89.4%, 69.6% to 70.4%, and 83.9%, respectively. Each trial cited dose reductions most commonly as the result of treatment-related AEs.15,16
Studies on the impact of RDIs on survival outcomes found that higher RDIs may improve overall and progression-free survival. Retrospective studies inspecting lenvatinib in hepatocellular carcinoma (HCC) indicated that an RDI > 70% in the initial 4 weeks resulted in favorable survival outcomes.17 Similarly, a retrospective study investigating sunitinib in RCC found that an RDI > 60% conferred favorable survival outcomes.18 Alghamdi et al noted that patients taking sorafenib for HCC who had RDI > 50% had a favorable trend in survival characteristics. Interestingly, the study found an RDI of 50% to 75% appeared to have better survival than an RDI > 75%.19 The authors of these studies hypothesized that additional dose reductions allowed for longer total time on therapy due to improved tolerability.17-19
This analysis found that the RDIs for most MKI agents at VANTHCS were < 85% and lower than the RDIs found in other review articles and phase 3 trials, with the exceptions of pazopanib in thyroid cancer and sunitinib in gastrointestinal stromal tumor (GIST), thyroid cancer, and neuroendocrine cancer. The reasons for the lower RDIs in this study are likely multifactorial, reflecting patient population characteristics, off-label dosing practices, and HCP experiences with these agents. Many veterans have chronic comorbidities that could contribute to reduced performance status and ability to tolerate these therapies. Despite attempts to preemptively reduce doses for patients and account for potential impaired tolerance, there were patients who required further dose reductions in our study.
Failure to thrive was the most common AE leading to dose adjustment or discontinuation, which illustrates the extensive effects these agents have on patient functioning in a real-world population. Notably though, the RDI for sunitinib was higher in this population because about half of patients were dosed using the off-label recommendation, whereas the prescribing information recommends a more intensive 6-week dosing cycle for certain cancer types.12,13,20 Sorafenib was also often dose-adjusted based on a pharmacokinetic study of sorafenib in renal/hepatic dysfunction, and the RDI likely reflects the off-label prescribing pattern.21
Patients with thyroid cancer were found to have higher RDIs compared with those receiving the same agents for other cancer types. Improved tolerability of MKIs in thyroid cancer may be due to a generally more tolerable disease course. Thyroid cancer is the most common cancer in individuals aged < 40 years, a population that is often more robust with fewer comorbidities. Moreover, the 5-year relative survival rate for thyroid cancer remains > 98%.22 This rate is in contrast to those for other cancer types such as HCC, with a 5-year relative survival rate of only 15%.23
It is challenging to compare the mean and median times on therapy found in this study with those in current literature, as this review included multiple different cancer types for each agent. However, the numbers are generally lower than durations of therapy found across the different disease states and further emphasize the difficulty in tolerating MKIs in the VANTHCS population. Regorafenib had a short duration of time on therapy, which highlights the importance of trials like ReDOS and initiatives such as OCE Project Optimus in helping improve tolerance.7,8,24
Comparing our results with other studies proved challenging because the RDI calculation methods were not specified. Calculating RDIs in this study using method 1, which does not account for holds, resulted in higher RDIs (Appendix 2). Using method 1, all MKIs had RDIs < 85%, except for pazopanib in thyroid cancer (100%) and RCC (87.9%), and sunitinib in GIST (93.6%), thyroid cancer (100%), and neuroendocrine cancer (93.7%). Notably, using method 1 increased the RDI for pazopanib in neuroendocrine cancer from 5.4% to 50.0%. The low RDI was attributed to a single veteran with a long hold duration, which demonstrates the discrepancy that can occur between the 2 methods.
Limitations
The retrospective design, lack of survival outcomes, and difficulty comparing results with other literature were limitations of this study. Because survival outcomes were not evaluated, future research should seek to investigate how RDIs and dose adjustments made among MKIs can affect survival outcomes in real-world populations. This veteran population with cancer often had multiple chronic comorbidities, which may have contributed to difficulty tolerating MKIs and could have impacted results. Disease-related factors may have influenced the poor tolerance of the MKIs and were not specifically accounted for. Adjustment for comorbidities was not possible because of discrepancies and/or incomplete diagnosis codes and Eastern Cooperative Oncology Group performance status scores documented in patient charts. Therefore, we decided not to report these findings due to potential inaccuracies.
CONCLUSIONS
Results of this study demonstrate that oncology MKI agents used at VANTHCS were difficult for patients to tolerate, leading to suboptimal dosing compared with indicated doses established in clinical trials and prescribing information. Clinicians may use these data to help guide clinical decision-making whenever initiating and managing MKI agents in this population. These findings reinforce that MKI agents are often difficult to tolerate in real-world practice, and indicated doses are often not achieved. Further studies should aim to investigate the effect that various RDIs have on overall survival. Further investigation into different dosing schemes for MKIs to improve tolerability and longer-term use may also prove beneficial.
This analysis may help guide clinicians to carefully approach dosing MKI agents in the veteran population. Given the RDI and AEs, more clinicians may consider starting at lower than indicated doses with the goal to titrate up as tolerated. Additionally, the results highlight the importance of considering palliative care consults and ensuring appropriate supportive care agents are preemptively engaged and adjusted as needed. Approaching dosing and titrations cautiously may help reduce the burden of management on the health care system.
- Frequently asked questions. VA National Oncology Program. 2025. Accessed December 15, 2025. https://www.cancer.va.gov/CANCER/faqs.html
- Torez L. Reigniting the cancer moonshot to beat cancer. VA News. April 20, 2023. Accessed April 6, 2026. https://news.va.gov/118378/reigniting-the-cancer-moonshot-to-beat-cancer
- Shah NN, Casella E, Capozzi D, et al. Improving the safety of oral chemotherapy at an academic medical center. J Oncol Pract. 2016;12:e71-e76. doi:10.1200/JOP.2015.007260
- Hussaarts KGAM, Veerman GDM, Jansman FGA, et al. Clinically relevant drug interactions with multikinase inhibitors: a review. Ther Adv Med Oncol. 2019;11:1758835918818347. doi:10.1177/1758835918818347
- Shyam Sunder S, Sharma UC, Pokharel S. Adverse effects of tyrosine kinase inhibitors in cancer therapy: pathophysiology, mechanisms and clinical management. Signal Transduct Target Ther. 2023;8:262. doi:10.1038/s41392-023-01469-6
- Thomson RJ, Moshirfar M, Ronquillo Y. Tyrosine kinase inhibitors. In: StatPearls [Internet]. StatPearls Publishing; updated July 18, 2023. Accessed December 15, 2025. https://www.ncbi.nlm.nih.gov/books/NBK563322/
- Project Optimus. US Food and Drug Administration. Updated December 6, 2024. Accessed December 15, 2025. https://www.fda.gov/about-fda/oncology-center-excellence/project-optimus
- Optimizing the dosage of human prescription drugs and biological products for the treatment of oncologic diseases: Guidance for Industry. Docket number FDA-2022-D-2827. US Food and Drug Administration. August 2024. Accessed December 15, 2025. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/optimizing-dosage-human-prescription-drugs-and-biological-products-treatment-oncologic-diseases
- Schnadig ID, Hutson TE, Chung H, et al. Dosing patterns, toxicity, and outcomes in patients treated with first-line sunitinib for advanced renal cell carcinoma in community-based practices. Clin Genitourin Cancer. 2014;12:413-421. doi:10.1016/j.clgc.2014.06.015
- Motzer RJ, Hutson TE, Tomczak P, et al. Sunitinib versus interferon alfa in metastatic renal-cell carcinoma. N Engl J Med. 2007;356:115-124. doi:10.1056/nejmoa065044
- Hawn C, Bansal D. Relative dose intensity in oncology trials: a discussion of two approaches. PharmaSUG. 2024. Accessed April 6, 2026. https://pharmasug.org/proceedings/2024/ST/PharmaSUG-2024-ST-297.pdf
- George S, Merriam P, Maki RG, et al. Multicenter phase II trial of sunitinib in the treatment of nongastrointestinal stromal tumor sarcomas. J Clin Oncol. 2009;27:3154-3160. doi:10.1200/jco.2008.20.9890
- George S, Blay JY, Casali PG, et al. Clinical evaluation of continuous daily dosing of sunitinib malate in patients with advanced gastrointestinal stromal tumour after imatinib failure. Eur J Cancer. 2009;45:1959-1968. doi:10.1016/j.ejca.2009.02.011
- Denduluri N, Patt DA, Wang Y, et al. Dose delays, dose reductions, and relative dose intensity in patients with cancer who received adjuvant or neoadjuvant chemotherapy in community oncology practices. J Natl Compr Canc Netw. 2015;13:1383-1393. doi:10.6004/jnccn.2015.0166
- Motzer RJ, Penkov K, Haanen J, et al. Avelumab plus axitinib versus sunitinib for advanced renal-cell carcinoma. N Engl J Med. 2019;380:1103-1115. doi:10.1056/nejmoa1816047
- Motzer R, Alekseev B, Rha SY, et al. Lenvatinib plus pembrolizumab or everolimus for advanced renal cell carcinoma. N Engl J Med. 2021;384:1289-1300. doi:10.1056/nejmoa2035716
- Kirino S, Tsuchiya K, Kurosaki M, et al. Relative dose intensity over the first four weeks of lenvatinib therapy is a factor of favorable response and overall survival in patients with unresectable hepatocellular carcinoma. PloS One. 2020;15:e0231828. doi:10.1371/journal.pone.0231828
- Ishihara H, Takagi T, Kondo T, et al. Decreased relative dose intensity during the early phase of treatment impacts the therapeutic efficacy of sunitinib in metastatic renal cell carcinoma. Jpn J Clin Oncol. 2018;48:667-672. doi:10.1093/jjco/hyy078
- Alghamdi MA, Amaro CP, Lee-Ying R, et al. Effect of sorafenib starting dose and dose intensity on survival in patients with hepatocellular carcinoma: results from a Canadian Multicenter Database. Cancer Med. 2020;9:4918-4928. doi:10.1002/cam4.3228
- Motzer RJ, Rini BI, Bukowski RM, et al. Sunitinib in patients with metastatic renal cell carcinoma. JAMA. 2006;295:2516-2524. doi:10.1001/jama.295.21.2516
- Miller AA, Murry DJ, Owzar K, et al. Phase I and pharmacokinetic study of sorafenib in patients with hepatic or renal dysfunction: CALGB 60301. J Clin Oncol. 2009;27:1800-1805. doi:10.1200/jco.2008.20.0931
- Boucai L, Zafereo M, Cabanillas ME. Thyroid cancer: a review. JAMA. 2024;331:425-435. doi:10.1001/jama.2023.26348
- Amin N, Anwar J, Sulaiman A, et al. Hepatocellular carcinoma: a comprehensive review. Diseases. 2025;13:207. doi:10.3390/diseases13070207
- Bekaii-Saab TS, Ou FS, Ahn DH, et al. Regorafenib dose-optimisation in patients with refractory metastatic colorectal cancer (ReDOS): a randomised, multicentre, open-label, phase 2 study. Lancet Oncol. 2019;20:1070-1082. doi:10.1016/s1470-2045(19)30272-4
The US Department of Veterans Affairs (VA) annually treats around 450,000 veterans with cancer and diagnoses an additional 56,000.1,2 Oral multikinase inhibitors (MKIs) are widely used as targeted therapies for many different malignancies. Despite the ease of oral administration, these agents are often accompanied by significant adverse effects (AEs) and drug-drug interactions.3,4 Common AEs include hypertension, cutaneous reactions, gastrointestinal disturbances, proteinuria, and fatigue. Some serious outcomes that may occur are myocardial infarction, thrombosis, nephrotic syndrome, hemorrhage, hepatotoxicity, and gastrointestinal events.5,6 Due to poor tolerability of these AEs, dose reductions, frequent therapy holds, and discontinuation of therapy may occur.
The US Food and Drug Administration recognizes dosing challenges with novel therapies and has created the Oncology Center of Excellence (OCE) Project Optimus initiative to reform dose optimization in oncology drug development. The initiative aims to shift the focus from establishing dose regimens based on the maximum tolerated doses of cytotoxic chemotherapeutics to an emphasis on maximum efficacy, safety, and tolerability, which better reflect real-world dosing.7,8
MKIs can be challenging to manage because of the frequent toxicity-related dose reductions, interruptions, and discontinuations. In a multicenter retrospective study, Schnadig et al investigated dosing characteristics of first-line sunitinib for advanced renal cell carcinoma (RCC) and found that, among 114 patients who experienced AEs while taking sunitinib, 39.5% had dose reductions, 5.3% delayed therapy, 18.4% required additional supportive medications, and 22.8% discontinued sunitinib.9 Overall survival and median progression-free survival of these patients were lower than reported by Motzer et al in a phase 3 clinical trial.10 Schnadig et al concluded that patients treated with sunitinib for RCC in the community setting required more frequent dose reductions and had less time on therapy compared with patients in clinical trials, which ultimately impacted clinical outcomes.9
At the VA North Texas Health Care System (VANTHCS), patients with cancer have difficulty tolerating MKIs and often require dose alterations and/or discontinuation because of drug intolerance rather than discontinuation due to progression. Frequent dose adjustments for toxicity management can place more strain on patients and health care resources because of additional appointments, clinician time, and emergency department visits. Escalating drug costs can also cause concern when prescription doses are unused or changed frequently.
To capture and quantify prescribing practices and dose adjustments, this study evaluated the tolerability of MKIs at VANTHCS. This analysis may also guide clinicians in the selection of starting doses as well as dose titration expectations to optimize MKI therapy.
METHODS
This single-center, retrospective chart review analyzed patients receiving oral oncology MKIs for various malignancies at VANTHCS between January 1, 2014, and October 31, 2024. Participants included adults aged ≥ 18 years with a prescription for axitinib, cabozantinib, lenvatinib, pazopanib, regorafenib, sorafenib, or sunitinib initiated by the hematology/oncology service at VANTHCS. Patients were included if they had follow-up documentation with the hematology/oncology service and/or other VANTHCS clinicians outlining their course of therapy after MKI initiation. Patients were excluded if they did not have sufficient follow-up documentation (eg, transferred care to a non-VA health care practitioner [HCP], moved to another VA health care system), were enrolled in clinical trials, or were prescribed an MKI from a Care in the Community (CITC) prescriber. Electronic health record review and data collection were performed using the VA Computerized Patient Record System and Research Electronic Data Capture. Data were collected from the time of initiation to cessation of therapy and included information regarding therapy changes, progressive disease, and date of death, when available. Data collected included age, sex, race, comorbidities, date of death, type of malignancy and subtypes, cancer stage, MKI used (ie, drug, dose, frequency, schedule, and indication), dates of medication changes (ie, start, adjustment, hold, discontinuation), concurrent antineoplastic treatments, and AEs documented at times of dose change or interruption.
The primary outcome was MKI tolerance determined using relative dose intensity (RDI) and mean and median time on therapy. Two methods are used to calculate RDI that vary in how they approach time on therapy as outlined in Hawn et al.11 This study used method 2, which accounts for holds in therapy by comparing the actual duration of treatment with the duration expected according to treatment protocol. Method 1 compares the prescribed dose with the administered dose and does not adjust for holds.11 Using method 2, the RDI in this study was calculated by dividing the total actual dose given by the total indicated dose for the malignancy being treated, which accounts for duration of treatment.

The total actual dose was the strength, frequency, and days on therapy for each time frame of treatment multiplied together. This method accounted for all dose adjustments and time periods of treatment holds, including patient self-adjustments, prescriber-directed adjustments, and nonadherence determined by HCP documentation and/or prescription data. Similarly, the indicated total dose was calculated by multiplying the indicated strength, frequency, and all days that treatment should have occurred (time from start to finish). Indicated doses were derived from the prescribing information for each malignancy with the exception of sunitinib, for which the off-label dose of 37.5 mg daily was considered a full dose.12,13 The total indicated dose for axitinib was calculated by considering the dose escalation schedule from the prescribing information.
Patients who required dose reductions due to renal/hepatic impairments or drug-drug interactions had their total indicated dose calculated using dose adjustments listed in the prescribing information. The mean RDI for each MKI agent was calculated by averaging the RDI for each prescription. The overall combined mean RDI included the means of all the MKIs reviewed to avoid skewing the results toward an MKI with more prescriptions. RDIs were also calculated for each cancer type for each agent. Additional descriptive secondary outcomes included rates of AEs and adjustments in doses.
RESULTS
Electronic data extraction identified 278 patients with 366 MKI prescriptions, of which 108 veterans with 158 MKI prescriptions were excluded. The top reason for exclusion was patients managed through CITC. Ultimately, 170 veterans with 208 MKI prescriptions managed by the VANTHCS hematology/oncology clinic were included (Table 1). Among patients receiving MKIs, the mean age was 72.7 years, 98% were male, and 99% had metastatic disease.

The overall combined mean MKI RDI was 67.5% using method 2 and ranged from 85.5% for sunitinib to 49.0% for sorafenib (Figure 1). Additional information regarding mean and median RDIs using method 2 is shown in Figure 1 and further subdivided by cancer type in Table 2. Median RDIs overall were similar to mean RDIs for most agents. Figure 2 indicates the mean and median time on therapy, reflecting time on therapy excluding days therapy was held. The overall combined mean and median days on therapy for all MKIs were 155 days and 95 days, respectively. Mean time on therapy depended on the agent used and ranged from 35 days (regorafenib) to 237 days (cabozantinib).



Of 208 MKI prescriptions, 127 (61.1%) were initiated at a reduced dose due to baseline concerns for tolerance such as performance status, frailty, and prior intolerance of other treatments. Eighty-one prescriptions (38.9%) were initiated at their indicated doses. Ninety prescriptions (43.3%) required dose reductions during treatment. Some MKI prescriptions had multiple dose increases and decreases, which is why RDI more accurately reflects dose adjustments. A total of 376 AEs that contributed to a dose adjustment, hold, or discontinuation occurred across all MKI prescriptions. The most common AEs were 82 failure-to-thrive events (21.8%) (fatigue, malaise, loss of appetite, reduced mobility, global decline), 79 gastrointestinal events (21.0%) (nausea, vomiting, diarrhea, abdominal pain), 62 dermatologic events (16.5%) (rash, hand-foot skin reactions, allergic response), 61 hepatic dysfunction events (16.2%) (liver enzyme elevations, hyperbilirubinemia), 40 cardiovascular events (10.6%) (hypertension, heart failure exacerbations, edema), and 33 renal dysfunction events (8.8%) (acute kidney injury, proteinuria) (Appendix 1).

DISCUSSION
The mean RDI of MKI prescriptions used in the veteran population at VANTHCS was about two-thirds of the indicated dose. These results indicate that most veterans required dose reductions and/or holds due to concerns over initial tolerance/performance status, worsening clinical condition, and/or intolerable AEs attributed to treatment. A retrospective study conducted by Denduluri et al suggested that an RDI of < 85% is a clinically meaningful reduction for traditional chemotherapy based on previous literature.14 However, it is less clear what RDI should be expected specifically for MKIs in real-world populations. The MKI phase 3 approval trials in RCC for axitinib, lenvatinib, and sunitinib found median RDIs of 89.4%, 69.6% to 70.4%, and 83.9%, respectively. Each trial cited dose reductions most commonly as the result of treatment-related AEs.15,16
Studies on the impact of RDIs on survival outcomes found that higher RDIs may improve overall and progression-free survival. Retrospective studies inspecting lenvatinib in hepatocellular carcinoma (HCC) indicated that an RDI > 70% in the initial 4 weeks resulted in favorable survival outcomes.17 Similarly, a retrospective study investigating sunitinib in RCC found that an RDI > 60% conferred favorable survival outcomes.18 Alghamdi et al noted that patients taking sorafenib for HCC who had RDI > 50% had a favorable trend in survival characteristics. Interestingly, the study found an RDI of 50% to 75% appeared to have better survival than an RDI > 75%.19 The authors of these studies hypothesized that additional dose reductions allowed for longer total time on therapy due to improved tolerability.17-19
This analysis found that the RDIs for most MKI agents at VANTHCS were < 85% and lower than the RDIs found in other review articles and phase 3 trials, with the exceptions of pazopanib in thyroid cancer and sunitinib in gastrointestinal stromal tumor (GIST), thyroid cancer, and neuroendocrine cancer. The reasons for the lower RDIs in this study are likely multifactorial, reflecting patient population characteristics, off-label dosing practices, and HCP experiences with these agents. Many veterans have chronic comorbidities that could contribute to reduced performance status and ability to tolerate these therapies. Despite attempts to preemptively reduce doses for patients and account for potential impaired tolerance, there were patients who required further dose reductions in our study.
Failure to thrive was the most common AE leading to dose adjustment or discontinuation, which illustrates the extensive effects these agents have on patient functioning in a real-world population. Notably though, the RDI for sunitinib was higher in this population because about half of patients were dosed using the off-label recommendation, whereas the prescribing information recommends a more intensive 6-week dosing cycle for certain cancer types.12,13,20 Sorafenib was also often dose-adjusted based on a pharmacokinetic study of sorafenib in renal/hepatic dysfunction, and the RDI likely reflects the off-label prescribing pattern.21
Patients with thyroid cancer were found to have higher RDIs compared with those receiving the same agents for other cancer types. Improved tolerability of MKIs in thyroid cancer may be due to a generally more tolerable disease course. Thyroid cancer is the most common cancer in individuals aged < 40 years, a population that is often more robust with fewer comorbidities. Moreover, the 5-year relative survival rate for thyroid cancer remains > 98%.22 This rate is in contrast to those for other cancer types such as HCC, with a 5-year relative survival rate of only 15%.23
It is challenging to compare the mean and median times on therapy found in this study with those in current literature, as this review included multiple different cancer types for each agent. However, the numbers are generally lower than durations of therapy found across the different disease states and further emphasize the difficulty in tolerating MKIs in the VANTHCS population. Regorafenib had a short duration of time on therapy, which highlights the importance of trials like ReDOS and initiatives such as OCE Project Optimus in helping improve tolerance.7,8,24
Comparing our results with other studies proved challenging because the RDI calculation methods were not specified. Calculating RDIs in this study using method 1, which does not account for holds, resulted in higher RDIs (Appendix 2). Using method 1, all MKIs had RDIs < 85%, except for pazopanib in thyroid cancer (100%) and RCC (87.9%), and sunitinib in GIST (93.6%), thyroid cancer (100%), and neuroendocrine cancer (93.7%). Notably, using method 1 increased the RDI for pazopanib in neuroendocrine cancer from 5.4% to 50.0%. The low RDI was attributed to a single veteran with a long hold duration, which demonstrates the discrepancy that can occur between the 2 methods.

Limitations
The retrospective design, lack of survival outcomes, and difficulty comparing results with other literature were limitations of this study. Because survival outcomes were not evaluated, future research should seek to investigate how RDIs and dose adjustments made among MKIs can affect survival outcomes in real-world populations. This veteran population with cancer often had multiple chronic comorbidities, which may have contributed to difficulty tolerating MKIs and could have impacted results. Disease-related factors may have influenced the poor tolerance of the MKIs and were not specifically accounted for. Adjustment for comorbidities was not possible because of discrepancies and/or incomplete diagnosis codes and Eastern Cooperative Oncology Group performance status scores documented in patient charts. Therefore, we decided not to report these findings due to potential inaccuracies.
CONCLUSIONS
Results of this study demonstrate that oncology MKI agents used at VANTHCS were difficult for patients to tolerate, leading to suboptimal dosing compared with indicated doses established in clinical trials and prescribing information. Clinicians may use these data to help guide clinical decision-making whenever initiating and managing MKI agents in this population. These findings reinforce that MKI agents are often difficult to tolerate in real-world practice, and indicated doses are often not achieved. Further studies should aim to investigate the effect that various RDIs have on overall survival. Further investigation into different dosing schemes for MKIs to improve tolerability and longer-term use may also prove beneficial.
This analysis may help guide clinicians to carefully approach dosing MKI agents in the veteran population. Given the RDI and AEs, more clinicians may consider starting at lower than indicated doses with the goal to titrate up as tolerated. Additionally, the results highlight the importance of considering palliative care consults and ensuring appropriate supportive care agents are preemptively engaged and adjusted as needed. Approaching dosing and titrations cautiously may help reduce the burden of management on the health care system.
The US Department of Veterans Affairs (VA) annually treats around 450,000 veterans with cancer and diagnoses an additional 56,000.1,2 Oral multikinase inhibitors (MKIs) are widely used as targeted therapies for many different malignancies. Despite the ease of oral administration, these agents are often accompanied by significant adverse effects (AEs) and drug-drug interactions.3,4 Common AEs include hypertension, cutaneous reactions, gastrointestinal disturbances, proteinuria, and fatigue. Some serious outcomes that may occur are myocardial infarction, thrombosis, nephrotic syndrome, hemorrhage, hepatotoxicity, and gastrointestinal events.5,6 Due to poor tolerability of these AEs, dose reductions, frequent therapy holds, and discontinuation of therapy may occur.
The US Food and Drug Administration recognizes dosing challenges with novel therapies and has created the Oncology Center of Excellence (OCE) Project Optimus initiative to reform dose optimization in oncology drug development. The initiative aims to shift the focus from establishing dose regimens based on the maximum tolerated doses of cytotoxic chemotherapeutics to an emphasis on maximum efficacy, safety, and tolerability, which better reflect real-world dosing.7,8
MKIs can be challenging to manage because of the frequent toxicity-related dose reductions, interruptions, and discontinuations. In a multicenter retrospective study, Schnadig et al investigated dosing characteristics of first-line sunitinib for advanced renal cell carcinoma (RCC) and found that, among 114 patients who experienced AEs while taking sunitinib, 39.5% had dose reductions, 5.3% delayed therapy, 18.4% required additional supportive medications, and 22.8% discontinued sunitinib.9 Overall survival and median progression-free survival of these patients were lower than reported by Motzer et al in a phase 3 clinical trial.10 Schnadig et al concluded that patients treated with sunitinib for RCC in the community setting required more frequent dose reductions and had less time on therapy compared with patients in clinical trials, which ultimately impacted clinical outcomes.9
At the VA North Texas Health Care System (VANTHCS), patients with cancer have difficulty tolerating MKIs and often require dose alterations and/or discontinuation because of drug intolerance rather than discontinuation due to progression. Frequent dose adjustments for toxicity management can place more strain on patients and health care resources because of additional appointments, clinician time, and emergency department visits. Escalating drug costs can also cause concern when prescription doses are unused or changed frequently.
To capture and quantify prescribing practices and dose adjustments, this study evaluated the tolerability of MKIs at VANTHCS. This analysis may also guide clinicians in the selection of starting doses as well as dose titration expectations to optimize MKI therapy.
METHODS
This single-center, retrospective chart review analyzed patients receiving oral oncology MKIs for various malignancies at VANTHCS between January 1, 2014, and October 31, 2024. Participants included adults aged ≥ 18 years with a prescription for axitinib, cabozantinib, lenvatinib, pazopanib, regorafenib, sorafenib, or sunitinib initiated by the hematology/oncology service at VANTHCS. Patients were included if they had follow-up documentation with the hematology/oncology service and/or other VANTHCS clinicians outlining their course of therapy after MKI initiation. Patients were excluded if they did not have sufficient follow-up documentation (eg, transferred care to a non-VA health care practitioner [HCP], moved to another VA health care system), were enrolled in clinical trials, or were prescribed an MKI from a Care in the Community (CITC) prescriber. Electronic health record review and data collection were performed using the VA Computerized Patient Record System and Research Electronic Data Capture. Data were collected from the time of initiation to cessation of therapy and included information regarding therapy changes, progressive disease, and date of death, when available. Data collected included age, sex, race, comorbidities, date of death, type of malignancy and subtypes, cancer stage, MKI used (ie, drug, dose, frequency, schedule, and indication), dates of medication changes (ie, start, adjustment, hold, discontinuation), concurrent antineoplastic treatments, and AEs documented at times of dose change or interruption.
The primary outcome was MKI tolerance determined using relative dose intensity (RDI) and mean and median time on therapy. Two methods are used to calculate RDI that vary in how they approach time on therapy as outlined in Hawn et al.11 This study used method 2, which accounts for holds in therapy by comparing the actual duration of treatment with the duration expected according to treatment protocol. Method 1 compares the prescribed dose with the administered dose and does not adjust for holds.11 Using method 2, the RDI in this study was calculated by dividing the total actual dose given by the total indicated dose for the malignancy being treated, which accounts for duration of treatment.

The total actual dose was the strength, frequency, and days on therapy for each time frame of treatment multiplied together. This method accounted for all dose adjustments and time periods of treatment holds, including patient self-adjustments, prescriber-directed adjustments, and nonadherence determined by HCP documentation and/or prescription data. Similarly, the indicated total dose was calculated by multiplying the indicated strength, frequency, and all days that treatment should have occurred (time from start to finish). Indicated doses were derived from the prescribing information for each malignancy with the exception of sunitinib, for which the off-label dose of 37.5 mg daily was considered a full dose.12,13 The total indicated dose for axitinib was calculated by considering the dose escalation schedule from the prescribing information.
Patients who required dose reductions due to renal/hepatic impairments or drug-drug interactions had their total indicated dose calculated using dose adjustments listed in the prescribing information. The mean RDI for each MKI agent was calculated by averaging the RDI for each prescription. The overall combined mean RDI included the means of all the MKIs reviewed to avoid skewing the results toward an MKI with more prescriptions. RDIs were also calculated for each cancer type for each agent. Additional descriptive secondary outcomes included rates of AEs and adjustments in doses.
RESULTS
Electronic data extraction identified 278 patients with 366 MKI prescriptions, of which 108 veterans with 158 MKI prescriptions were excluded. The top reason for exclusion was patients managed through CITC. Ultimately, 170 veterans with 208 MKI prescriptions managed by the VANTHCS hematology/oncology clinic were included (Table 1). Among patients receiving MKIs, the mean age was 72.7 years, 98% were male, and 99% had metastatic disease.

The overall combined mean MKI RDI was 67.5% using method 2 and ranged from 85.5% for sunitinib to 49.0% for sorafenib (Figure 1). Additional information regarding mean and median RDIs using method 2 is shown in Figure 1 and further subdivided by cancer type in Table 2. Median RDIs overall were similar to mean RDIs for most agents. Figure 2 indicates the mean and median time on therapy, reflecting time on therapy excluding days therapy was held. The overall combined mean and median days on therapy for all MKIs were 155 days and 95 days, respectively. Mean time on therapy depended on the agent used and ranged from 35 days (regorafenib) to 237 days (cabozantinib).



Of 208 MKI prescriptions, 127 (61.1%) were initiated at a reduced dose due to baseline concerns for tolerance such as performance status, frailty, and prior intolerance of other treatments. Eighty-one prescriptions (38.9%) were initiated at their indicated doses. Ninety prescriptions (43.3%) required dose reductions during treatment. Some MKI prescriptions had multiple dose increases and decreases, which is why RDI more accurately reflects dose adjustments. A total of 376 AEs that contributed to a dose adjustment, hold, or discontinuation occurred across all MKI prescriptions. The most common AEs were 82 failure-to-thrive events (21.8%) (fatigue, malaise, loss of appetite, reduced mobility, global decline), 79 gastrointestinal events (21.0%) (nausea, vomiting, diarrhea, abdominal pain), 62 dermatologic events (16.5%) (rash, hand-foot skin reactions, allergic response), 61 hepatic dysfunction events (16.2%) (liver enzyme elevations, hyperbilirubinemia), 40 cardiovascular events (10.6%) (hypertension, heart failure exacerbations, edema), and 33 renal dysfunction events (8.8%) (acute kidney injury, proteinuria) (Appendix 1).

DISCUSSION
The mean RDI of MKI prescriptions used in the veteran population at VANTHCS was about two-thirds of the indicated dose. These results indicate that most veterans required dose reductions and/or holds due to concerns over initial tolerance/performance status, worsening clinical condition, and/or intolerable AEs attributed to treatment. A retrospective study conducted by Denduluri et al suggested that an RDI of < 85% is a clinically meaningful reduction for traditional chemotherapy based on previous literature.14 However, it is less clear what RDI should be expected specifically for MKIs in real-world populations. The MKI phase 3 approval trials in RCC for axitinib, lenvatinib, and sunitinib found median RDIs of 89.4%, 69.6% to 70.4%, and 83.9%, respectively. Each trial cited dose reductions most commonly as the result of treatment-related AEs.15,16
Studies on the impact of RDIs on survival outcomes found that higher RDIs may improve overall and progression-free survival. Retrospective studies inspecting lenvatinib in hepatocellular carcinoma (HCC) indicated that an RDI > 70% in the initial 4 weeks resulted in favorable survival outcomes.17 Similarly, a retrospective study investigating sunitinib in RCC found that an RDI > 60% conferred favorable survival outcomes.18 Alghamdi et al noted that patients taking sorafenib for HCC who had RDI > 50% had a favorable trend in survival characteristics. Interestingly, the study found an RDI of 50% to 75% appeared to have better survival than an RDI > 75%.19 The authors of these studies hypothesized that additional dose reductions allowed for longer total time on therapy due to improved tolerability.17-19
This analysis found that the RDIs for most MKI agents at VANTHCS were < 85% and lower than the RDIs found in other review articles and phase 3 trials, with the exceptions of pazopanib in thyroid cancer and sunitinib in gastrointestinal stromal tumor (GIST), thyroid cancer, and neuroendocrine cancer. The reasons for the lower RDIs in this study are likely multifactorial, reflecting patient population characteristics, off-label dosing practices, and HCP experiences with these agents. Many veterans have chronic comorbidities that could contribute to reduced performance status and ability to tolerate these therapies. Despite attempts to preemptively reduce doses for patients and account for potential impaired tolerance, there were patients who required further dose reductions in our study.
Failure to thrive was the most common AE leading to dose adjustment or discontinuation, which illustrates the extensive effects these agents have on patient functioning in a real-world population. Notably though, the RDI for sunitinib was higher in this population because about half of patients were dosed using the off-label recommendation, whereas the prescribing information recommends a more intensive 6-week dosing cycle for certain cancer types.12,13,20 Sorafenib was also often dose-adjusted based on a pharmacokinetic study of sorafenib in renal/hepatic dysfunction, and the RDI likely reflects the off-label prescribing pattern.21
Patients with thyroid cancer were found to have higher RDIs compared with those receiving the same agents for other cancer types. Improved tolerability of MKIs in thyroid cancer may be due to a generally more tolerable disease course. Thyroid cancer is the most common cancer in individuals aged < 40 years, a population that is often more robust with fewer comorbidities. Moreover, the 5-year relative survival rate for thyroid cancer remains > 98%.22 This rate is in contrast to those for other cancer types such as HCC, with a 5-year relative survival rate of only 15%.23
It is challenging to compare the mean and median times on therapy found in this study with those in current literature, as this review included multiple different cancer types for each agent. However, the numbers are generally lower than durations of therapy found across the different disease states and further emphasize the difficulty in tolerating MKIs in the VANTHCS population. Regorafenib had a short duration of time on therapy, which highlights the importance of trials like ReDOS and initiatives such as OCE Project Optimus in helping improve tolerance.7,8,24
Comparing our results with other studies proved challenging because the RDI calculation methods were not specified. Calculating RDIs in this study using method 1, which does not account for holds, resulted in higher RDIs (Appendix 2). Using method 1, all MKIs had RDIs < 85%, except for pazopanib in thyroid cancer (100%) and RCC (87.9%), and sunitinib in GIST (93.6%), thyroid cancer (100%), and neuroendocrine cancer (93.7%). Notably, using method 1 increased the RDI for pazopanib in neuroendocrine cancer from 5.4% to 50.0%. The low RDI was attributed to a single veteran with a long hold duration, which demonstrates the discrepancy that can occur between the 2 methods.

Limitations
The retrospective design, lack of survival outcomes, and difficulty comparing results with other literature were limitations of this study. Because survival outcomes were not evaluated, future research should seek to investigate how RDIs and dose adjustments made among MKIs can affect survival outcomes in real-world populations. This veteran population with cancer often had multiple chronic comorbidities, which may have contributed to difficulty tolerating MKIs and could have impacted results. Disease-related factors may have influenced the poor tolerance of the MKIs and were not specifically accounted for. Adjustment for comorbidities was not possible because of discrepancies and/or incomplete diagnosis codes and Eastern Cooperative Oncology Group performance status scores documented in patient charts. Therefore, we decided not to report these findings due to potential inaccuracies.
CONCLUSIONS
Results of this study demonstrate that oncology MKI agents used at VANTHCS were difficult for patients to tolerate, leading to suboptimal dosing compared with indicated doses established in clinical trials and prescribing information. Clinicians may use these data to help guide clinical decision-making whenever initiating and managing MKI agents in this population. These findings reinforce that MKI agents are often difficult to tolerate in real-world practice, and indicated doses are often not achieved. Further studies should aim to investigate the effect that various RDIs have on overall survival. Further investigation into different dosing schemes for MKIs to improve tolerability and longer-term use may also prove beneficial.
This analysis may help guide clinicians to carefully approach dosing MKI agents in the veteran population. Given the RDI and AEs, more clinicians may consider starting at lower than indicated doses with the goal to titrate up as tolerated. Additionally, the results highlight the importance of considering palliative care consults and ensuring appropriate supportive care agents are preemptively engaged and adjusted as needed. Approaching dosing and titrations cautiously may help reduce the burden of management on the health care system.
- Frequently asked questions. VA National Oncology Program. 2025. Accessed December 15, 2025. https://www.cancer.va.gov/CANCER/faqs.html
- Torez L. Reigniting the cancer moonshot to beat cancer. VA News. April 20, 2023. Accessed April 6, 2026. https://news.va.gov/118378/reigniting-the-cancer-moonshot-to-beat-cancer
- Shah NN, Casella E, Capozzi D, et al. Improving the safety of oral chemotherapy at an academic medical center. J Oncol Pract. 2016;12:e71-e76. doi:10.1200/JOP.2015.007260
- Hussaarts KGAM, Veerman GDM, Jansman FGA, et al. Clinically relevant drug interactions with multikinase inhibitors: a review. Ther Adv Med Oncol. 2019;11:1758835918818347. doi:10.1177/1758835918818347
- Shyam Sunder S, Sharma UC, Pokharel S. Adverse effects of tyrosine kinase inhibitors in cancer therapy: pathophysiology, mechanisms and clinical management. Signal Transduct Target Ther. 2023;8:262. doi:10.1038/s41392-023-01469-6
- Thomson RJ, Moshirfar M, Ronquillo Y. Tyrosine kinase inhibitors. In: StatPearls [Internet]. StatPearls Publishing; updated July 18, 2023. Accessed December 15, 2025. https://www.ncbi.nlm.nih.gov/books/NBK563322/
- Project Optimus. US Food and Drug Administration. Updated December 6, 2024. Accessed December 15, 2025. https://www.fda.gov/about-fda/oncology-center-excellence/project-optimus
- Optimizing the dosage of human prescription drugs and biological products for the treatment of oncologic diseases: Guidance for Industry. Docket number FDA-2022-D-2827. US Food and Drug Administration. August 2024. Accessed December 15, 2025. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/optimizing-dosage-human-prescription-drugs-and-biological-products-treatment-oncologic-diseases
- Schnadig ID, Hutson TE, Chung H, et al. Dosing patterns, toxicity, and outcomes in patients treated with first-line sunitinib for advanced renal cell carcinoma in community-based practices. Clin Genitourin Cancer. 2014;12:413-421. doi:10.1016/j.clgc.2014.06.015
- Motzer RJ, Hutson TE, Tomczak P, et al. Sunitinib versus interferon alfa in metastatic renal-cell carcinoma. N Engl J Med. 2007;356:115-124. doi:10.1056/nejmoa065044
- Hawn C, Bansal D. Relative dose intensity in oncology trials: a discussion of two approaches. PharmaSUG. 2024. Accessed April 6, 2026. https://pharmasug.org/proceedings/2024/ST/PharmaSUG-2024-ST-297.pdf
- George S, Merriam P, Maki RG, et al. Multicenter phase II trial of sunitinib in the treatment of nongastrointestinal stromal tumor sarcomas. J Clin Oncol. 2009;27:3154-3160. doi:10.1200/jco.2008.20.9890
- George S, Blay JY, Casali PG, et al. Clinical evaluation of continuous daily dosing of sunitinib malate in patients with advanced gastrointestinal stromal tumour after imatinib failure. Eur J Cancer. 2009;45:1959-1968. doi:10.1016/j.ejca.2009.02.011
- Denduluri N, Patt DA, Wang Y, et al. Dose delays, dose reductions, and relative dose intensity in patients with cancer who received adjuvant or neoadjuvant chemotherapy in community oncology practices. J Natl Compr Canc Netw. 2015;13:1383-1393. doi:10.6004/jnccn.2015.0166
- Motzer RJ, Penkov K, Haanen J, et al. Avelumab plus axitinib versus sunitinib for advanced renal-cell carcinoma. N Engl J Med. 2019;380:1103-1115. doi:10.1056/nejmoa1816047
- Motzer R, Alekseev B, Rha SY, et al. Lenvatinib plus pembrolizumab or everolimus for advanced renal cell carcinoma. N Engl J Med. 2021;384:1289-1300. doi:10.1056/nejmoa2035716
- Kirino S, Tsuchiya K, Kurosaki M, et al. Relative dose intensity over the first four weeks of lenvatinib therapy is a factor of favorable response and overall survival in patients with unresectable hepatocellular carcinoma. PloS One. 2020;15:e0231828. doi:10.1371/journal.pone.0231828
- Ishihara H, Takagi T, Kondo T, et al. Decreased relative dose intensity during the early phase of treatment impacts the therapeutic efficacy of sunitinib in metastatic renal cell carcinoma. Jpn J Clin Oncol. 2018;48:667-672. doi:10.1093/jjco/hyy078
- Alghamdi MA, Amaro CP, Lee-Ying R, et al. Effect of sorafenib starting dose and dose intensity on survival in patients with hepatocellular carcinoma: results from a Canadian Multicenter Database. Cancer Med. 2020;9:4918-4928. doi:10.1002/cam4.3228
- Motzer RJ, Rini BI, Bukowski RM, et al. Sunitinib in patients with metastatic renal cell carcinoma. JAMA. 2006;295:2516-2524. doi:10.1001/jama.295.21.2516
- Miller AA, Murry DJ, Owzar K, et al. Phase I and pharmacokinetic study of sorafenib in patients with hepatic or renal dysfunction: CALGB 60301. J Clin Oncol. 2009;27:1800-1805. doi:10.1200/jco.2008.20.0931
- Boucai L, Zafereo M, Cabanillas ME. Thyroid cancer: a review. JAMA. 2024;331:425-435. doi:10.1001/jama.2023.26348
- Amin N, Anwar J, Sulaiman A, et al. Hepatocellular carcinoma: a comprehensive review. Diseases. 2025;13:207. doi:10.3390/diseases13070207
- Bekaii-Saab TS, Ou FS, Ahn DH, et al. Regorafenib dose-optimisation in patients with refractory metastatic colorectal cancer (ReDOS): a randomised, multicentre, open-label, phase 2 study. Lancet Oncol. 2019;20:1070-1082. doi:10.1016/s1470-2045(19)30272-4
- Frequently asked questions. VA National Oncology Program. 2025. Accessed December 15, 2025. https://www.cancer.va.gov/CANCER/faqs.html
- Torez L. Reigniting the cancer moonshot to beat cancer. VA News. April 20, 2023. Accessed April 6, 2026. https://news.va.gov/118378/reigniting-the-cancer-moonshot-to-beat-cancer
- Shah NN, Casella E, Capozzi D, et al. Improving the safety of oral chemotherapy at an academic medical center. J Oncol Pract. 2016;12:e71-e76. doi:10.1200/JOP.2015.007260
- Hussaarts KGAM, Veerman GDM, Jansman FGA, et al. Clinically relevant drug interactions with multikinase inhibitors: a review. Ther Adv Med Oncol. 2019;11:1758835918818347. doi:10.1177/1758835918818347
- Shyam Sunder S, Sharma UC, Pokharel S. Adverse effects of tyrosine kinase inhibitors in cancer therapy: pathophysiology, mechanisms and clinical management. Signal Transduct Target Ther. 2023;8:262. doi:10.1038/s41392-023-01469-6
- Thomson RJ, Moshirfar M, Ronquillo Y. Tyrosine kinase inhibitors. In: StatPearls [Internet]. StatPearls Publishing; updated July 18, 2023. Accessed December 15, 2025. https://www.ncbi.nlm.nih.gov/books/NBK563322/
- Project Optimus. US Food and Drug Administration. Updated December 6, 2024. Accessed December 15, 2025. https://www.fda.gov/about-fda/oncology-center-excellence/project-optimus
- Optimizing the dosage of human prescription drugs and biological products for the treatment of oncologic diseases: Guidance for Industry. Docket number FDA-2022-D-2827. US Food and Drug Administration. August 2024. Accessed December 15, 2025. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/optimizing-dosage-human-prescription-drugs-and-biological-products-treatment-oncologic-diseases
- Schnadig ID, Hutson TE, Chung H, et al. Dosing patterns, toxicity, and outcomes in patients treated with first-line sunitinib for advanced renal cell carcinoma in community-based practices. Clin Genitourin Cancer. 2014;12:413-421. doi:10.1016/j.clgc.2014.06.015
- Motzer RJ, Hutson TE, Tomczak P, et al. Sunitinib versus interferon alfa in metastatic renal-cell carcinoma. N Engl J Med. 2007;356:115-124. doi:10.1056/nejmoa065044
- Hawn C, Bansal D. Relative dose intensity in oncology trials: a discussion of two approaches. PharmaSUG. 2024. Accessed April 6, 2026. https://pharmasug.org/proceedings/2024/ST/PharmaSUG-2024-ST-297.pdf
- George S, Merriam P, Maki RG, et al. Multicenter phase II trial of sunitinib in the treatment of nongastrointestinal stromal tumor sarcomas. J Clin Oncol. 2009;27:3154-3160. doi:10.1200/jco.2008.20.9890
- George S, Blay JY, Casali PG, et al. Clinical evaluation of continuous daily dosing of sunitinib malate in patients with advanced gastrointestinal stromal tumour after imatinib failure. Eur J Cancer. 2009;45:1959-1968. doi:10.1016/j.ejca.2009.02.011
- Denduluri N, Patt DA, Wang Y, et al. Dose delays, dose reductions, and relative dose intensity in patients with cancer who received adjuvant or neoadjuvant chemotherapy in community oncology practices. J Natl Compr Canc Netw. 2015;13:1383-1393. doi:10.6004/jnccn.2015.0166
- Motzer RJ, Penkov K, Haanen J, et al. Avelumab plus axitinib versus sunitinib for advanced renal-cell carcinoma. N Engl J Med. 2019;380:1103-1115. doi:10.1056/nejmoa1816047
- Motzer R, Alekseev B, Rha SY, et al. Lenvatinib plus pembrolizumab or everolimus for advanced renal cell carcinoma. N Engl J Med. 2021;384:1289-1300. doi:10.1056/nejmoa2035716
- Kirino S, Tsuchiya K, Kurosaki M, et al. Relative dose intensity over the first four weeks of lenvatinib therapy is a factor of favorable response and overall survival in patients with unresectable hepatocellular carcinoma. PloS One. 2020;15:e0231828. doi:10.1371/journal.pone.0231828
- Ishihara H, Takagi T, Kondo T, et al. Decreased relative dose intensity during the early phase of treatment impacts the therapeutic efficacy of sunitinib in metastatic renal cell carcinoma. Jpn J Clin Oncol. 2018;48:667-672. doi:10.1093/jjco/hyy078
- Alghamdi MA, Amaro CP, Lee-Ying R, et al. Effect of sorafenib starting dose and dose intensity on survival in patients with hepatocellular carcinoma: results from a Canadian Multicenter Database. Cancer Med. 2020;9:4918-4928. doi:10.1002/cam4.3228
- Motzer RJ, Rini BI, Bukowski RM, et al. Sunitinib in patients with metastatic renal cell carcinoma. JAMA. 2006;295:2516-2524. doi:10.1001/jama.295.21.2516
- Miller AA, Murry DJ, Owzar K, et al. Phase I and pharmacokinetic study of sorafenib in patients with hepatic or renal dysfunction: CALGB 60301. J Clin Oncol. 2009;27:1800-1805. doi:10.1200/jco.2008.20.0931
- Boucai L, Zafereo M, Cabanillas ME. Thyroid cancer: a review. JAMA. 2024;331:425-435. doi:10.1001/jama.2023.26348
- Amin N, Anwar J, Sulaiman A, et al. Hepatocellular carcinoma: a comprehensive review. Diseases. 2025;13:207. doi:10.3390/diseases13070207
- Bekaii-Saab TS, Ou FS, Ahn DH, et al. Regorafenib dose-optimisation in patients with refractory metastatic colorectal cancer (ReDOS): a randomised, multicentre, open-label, phase 2 study. Lancet Oncol. 2019;20:1070-1082. doi:10.1016/s1470-2045(19)30272-4
Investigating Real-World Tolerance and Dose Reductions of Oncology Multikinase Inhibitors in a VA Population
Investigating Real-World Tolerance and Dose Reductions of Oncology Multikinase Inhibitors in a VA Population
Potential Tyrosine Kinase Inhibitor Therapy Discontinuation for Patients With Chronic Myeloid Leukemia in a VA Regional Network
Potential Tyrosine Kinase Inhibitor Therapy Discontinuation for Patients With Chronic Myeloid Leukemia in a VA Regional Network
Chronic myeloid leukemia (CML) is a hematologic malignancy resulting from an acquired mutation. The mutation results in a reciprocal translocation between the long arms of chromosomes 9 and 22 and is known as the Philadelphia chromosome (Ph), or Ph-positive (Ph+) when present. The translocation results in the formation of a BCR-ABL fusion oncogene, which leads to continuous cell cycling and proliferation, altered differentiation, and a loss of apoptosis.1,2
Until the 1980s, CML was considered fatal.3 The mainstay of treatment consisted of 2 oral chemotherapeutic agents, busulfan and hydroxyurea. These medications did not prevent blast crisis, a fatal form of leukemia.4,5 The introduction of tyrosine kinase inhibitors (TKIs) transformed CML management and improved 10-year overall survival from about 20% to > 80% by delaying the transition to blast crisis. Now, the risk of death from general health conditions or comorbidities is higher than that of CML.6
TKIs target the root cause of CML through inhibition of the BCR-ABL oncoprotein.1,2 For CML, the goals of treatment include maintaining hematologic, cytogenetic, and molecular remission; preventing progression to accelerated phase or blast crisis; minimizing toxicity; and enabling potential cessation of therapy in carefully selected patients.7,8
Small cohort studies suggest that dose reduction of TKIs in patients who achieve optimal responses may reduce the risk of long-term adverse effects (AEs). However, optimal dose-reduction and minimum effective dose of each agent are unknown.7 The ability to maintain undetectable minimal residual disease or disease detectable at a stable low level after TKI discontinuation has been called treatment-free remission. Studies suggest that about 40% to 50% of patients who have achieved a stable deep molecular response remain in treatment-free remission after stopping first-line treatment.9,10 Of the patients who relapse following TKI discontinuation, 80% relapse within the first 6 months of treatment cessation. Molecular response is regained in almost all patients when treatment is resumed with the same TKI.11
The National Comprehensive Cancer Network (NCCN) recommends considering discontinuation of TKI therapy only outside the setting of a clinical trial and only in patients who consent to discontinuation after a thorough discussion of the potential risks and benefits. The NCCN criteria for patients who may be eligible for discontinuation are listed in Table 1. The Life After Stopping TKIs study reported that 80% of patients with well-controlled chronic phase CML who discontinued TKIs had a clinically meaningful improvement in fatigue. Patients also reported clinically meaningful improvements in depression, diarrhea, sleep disturbance, and pain interference. These symptoms worsened after restarting TKI therapy.12

TKI DISCONTINUATION
Electronic health record data were extracted using structured query language from the US Department of Veterans Affairs (VA) Corporate Data Warehouse (CDW). To be eligible for discontinuation, veterans had to be aged > 18 years, receive oncology care within a Veterans Integrated Services Network (VISN) 21 health care system (HCS) (VA Sierra Nevada HCS, VA Southern Nevada HCS, VA Central California HCS, VA Palo Alto HCS, VA Northern California HCS, and VA San Francisco HCS) or be a veteran referred to a community-based oncology practitioner. Patients had to have a documented diagnosis of chronic phase CML, have an active order for a TKI, be on TKI therapy for ≥ 3 years, and have a stable molecular response (BCR-ABL1 ≤ 0.01% on the International Scale for ≥ 2 years with ≥ 4 tests done ≥ 3 months apart) as of October 1, 2024. Veterans were excluded if they had a history of advanced accelerated phase CML, previous TKI discontinuation trials, nonadherence to the TKI, or if they did not want to consider TKI discontinuation.
This analysis evaluated the potential cost avoidance associated with TKI discontinuation. Cost avoidance was calculated using the average wholesale price of each TKI. Secondary objectives evaluated health outcomes of TKI discontinuation including CML relapse, reported AEs, long-term remission, and TKI withdrawal syndrome. Health outcomes were determined through chart review of AEs and clinic notes documented in the electronic health record during the study time frame.
Baseline information for eligible patients was collected, including age, sex, and race, and chart reviews were completed to evaluate reported AEs associated with therapy. Oncology clinical pharmacy practitioners (CPPs) at each VISN 21 facility were notified of eligible patients to facilitate discussion with oncologists and establish monitoring if therapy was discontinued. Following TKI discontinuation, health outcomes were evaluated, including CML relapse, changes in reported AEs, long-term remission, and TKI withdrawal syndrome. Descriptive statistics were used to analyze the baseline characteristics. Cost avoidance was calculated using the average wholesale price for each TKI. The number of tablets required to reach each patient’s individual dose was taken into consideration when determining the cost avoidance. A dashboard was created using the query from the CDW and was developed in Microsoft Power BI.
Preliminary Results
In FY 2024, VISN 21 had 3819 oncology patients. Twenty-four patients had taken a TKI for ≥ 3 years, 20 had a stable molecular response, and 15 had not previously attempted to discontinue their TKI (Figure 1). Fifteen veterans were eligible for therapy discontinuation for a total potential annual cost avoidance of $1.2 million (Figure 2). Most of the cost avoidance, $935,057 (78%), was attributed to 3 patients on nilotinib. The mean age of the population was 74 years. All patients were male, and 12 (80%) were White. (Table 2). At baseline, 11 patients (73%) were taking imatinib. One patient received oncology care from a community care clinician. All 15 patients decided to remain on therapy.
Abbreviations: CML, chronic myeloid leukemia; TKI, tyrosine kinase inhibitor;
VISN, Veterans Integrated Service Network.
for 15 patients at Veterans Integrated Services Network 21.
DISCUSSION
As a multisite quality improvement initiative, this project raised awareness of TKI therapy discontinuation in select patients with CML. It also sparked collaboration among oncology CPPs and clinicians and stimulated conversations about CML treatment. The development of the TKI discontinuation dashboard provides a population health management tool for CPPs and clinicians to identify eligible patients in the future.
Adherence to TKIs is crucial for disease control and survival in patients with CML. Patients are counseled that poor adherence to therapy may contribute to worsening disease or suboptimal response, the development of resistance, and greater health care costs.13 Therefore, it was a challenge for patients to understand and accept that they could stop TKI therapy after achieving a stable deep molecular response. Discussions with patients about the goal of therapy—suppressing the BCR-ABL oncogene, which they have achieved—could encourage patients to trial therapy discontinuation.
Only small cohort studies have been completed to evaluate the outcomes of therapy discontinuation. Much remains unknown regarding the optimal dose-reduction strategy and the minimum effective dose of each agent. Additionally, understanding the qualities of a good candidate for TKI discontinuation remains a barrier. A similar project was conducted in VISN 17. Five patients were counseled on TKI discontinuation; however, only 1 discontinued TKI therapy. Unfortunately, soon after discontinuing treatment, the patient had to restart therapy. Additional literature will enhance understanding of therapy discontinuation.
An unexpected finding of TKI discontinuation trials has been a reversible phenomenon known as TKI withdrawal syndrome.9 It can occur regardless of the TKI used and results in pruritus and new or worsening musculoskeletal pain within several weeks of TKI discontinuation in about 30% of patients. Symptoms may last several months and may require acetaminophen or nonsteroidal anti-inflammatory drugs for pain control.9,10,14
The potential cost avoidance of $1.2 million is an underestimation because VA contracts allow for greater cost savings. However, that information is confidential and therefore average wholesale price had to be used for this project. Most of the cost avoidance was due to 4 patients who could not tolerate imatinib and used nilotinib, which is more expensive.
Limitations
The small sample size presented some limitations. Of the 3819 oncology patients within VISN 21 in FY 2024, 186 received a TKI and only 15 were eligible for discontinuation. Additionally, challenges emerged when discussing discontinuation with community care clinicians and patients. Community care clinicians were difficult to contact, making it challenging to discuss the project with them. CPPs noted hesitancy among VA clinicians and patients to discontinue a medication for which adherence was continually emphasized.
Conclusions
Discussions about CML TKI discontinuation led to collaboration with the oncology care team and could lead to significant cost avoidance. Barriers to TKI discontinuation included patients’ concern for relapse, risk of discontinuation syndrome, the requirement for close monitoring, and clinician buy-in. Outcome studies are needed to gain a greater understanding of the benefits and risks of therapy discontinuation. In the future, evaluation of possible clinical and biological predictors of successful TKI discontinuation may be beneficial.
- Schiffer CA. BCR-ABL tyrosine kinase inhibitors for chronic myelogenous leukemia. N Engl J Med. 2007;357:258-265. doi:10.1056/NEJMct071828
- Hehlmann R, Hochhaus A, Baccarani M; European LeukemiaNet. Chronic myeloid leukaemia. Lancet. 2007;370:342-350. doi:10.1016/S0140-6736(07)61165-9
- Goldman JM, Melo JV. Chronic myeloid leukemia--advances in biology and new approaches to treatment. N Engl J Med. 2003;349:1451-1464. doi:10.1056/NEJMra020777
- Pasic I, Lipton JH. Current approach to the treatment of chronic myeloid leukaemia. Leuk Res. 2017;55:65-78. doi:10.1016/j.leukres.2017.01.005
- Rao KV, Iannucci A, Jabbour E. Current and future clinical strategies in the management of chronic myeloid leukemia. Pharmacotherapy. 2010;30:77S-101S. doi:10.1592/phco.30.pt2.77S
- Cortes J, Pavlovsky C, Saußele S. Chronic myeloid leukaemia. Lancet. 2021;398:1914-1926. doi:10.1016/S0140-6736(21)01204-6
- National Comprehensive Cancer Network (NCCN). NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®). Chronic myeloid leukemia. Version 1.2026. July 16, 2025. Accessed February 8, 2026. https://www.nccn.org /guidelines/guidelines-detail?id=1427
- Hochhaus A, Baccarani M, Silver RT, et al. European LeukemiaNet 2020 recommendations for treating chronic myeloid leukemia. Leukemia. 2020;34:966-984. doi:10.1038/s41375-020-0776-2
- Saußele S, Richter J, Hochhaus A, Mahon F-X. The concept of treatment-free remission in chronic myeloid leukemia. Leukemia. 2016;30:1638-1647. doi:10.1038/leu.2016.115
- Atallah E, Sweet K. Treatment-free remission: the new goal in CML therapy. Curr Hematol Malig Rep. 2021;16:433-439. doi:10.1007/s11899-021-00653-1
- Hehlmann R. The new ELN recommendations for treating CML. J Clin Med. 2020;9:3671. doi:10.3390/jcm9113671
- Atallah E, Schiffer CA, Radich JP , et al. Assessment of outcomes after stopping tyrosine kinase inhibitors among patients with chronic myeloid leukemia: a non-randomized clinical trial. JAMA Oncol. 2021;7:42-50. doi:10.1001/jamaoncol.2020.5774
- Breccia M, Efficace F, Alimena G. Imatinib treatment in chronic myelogenous leukemia: what have we learned so far? Cancer Lett. 2011;300:115-121. doi:10.1016/j.canlet.2010.10.018
- Berman E. How I treat chronic-phase chronic myelogenous leukemia. Blood. 2022;139:3138-3147. doi:10.1182/blood.2021011722
Chronic myeloid leukemia (CML) is a hematologic malignancy resulting from an acquired mutation. The mutation results in a reciprocal translocation between the long arms of chromosomes 9 and 22 and is known as the Philadelphia chromosome (Ph), or Ph-positive (Ph+) when present. The translocation results in the formation of a BCR-ABL fusion oncogene, which leads to continuous cell cycling and proliferation, altered differentiation, and a loss of apoptosis.1,2
Until the 1980s, CML was considered fatal.3 The mainstay of treatment consisted of 2 oral chemotherapeutic agents, busulfan and hydroxyurea. These medications did not prevent blast crisis, a fatal form of leukemia.4,5 The introduction of tyrosine kinase inhibitors (TKIs) transformed CML management and improved 10-year overall survival from about 20% to > 80% by delaying the transition to blast crisis. Now, the risk of death from general health conditions or comorbidities is higher than that of CML.6
TKIs target the root cause of CML through inhibition of the BCR-ABL oncoprotein.1,2 For CML, the goals of treatment include maintaining hematologic, cytogenetic, and molecular remission; preventing progression to accelerated phase or blast crisis; minimizing toxicity; and enabling potential cessation of therapy in carefully selected patients.7,8
Small cohort studies suggest that dose reduction of TKIs in patients who achieve optimal responses may reduce the risk of long-term adverse effects (AEs). However, optimal dose-reduction and minimum effective dose of each agent are unknown.7 The ability to maintain undetectable minimal residual disease or disease detectable at a stable low level after TKI discontinuation has been called treatment-free remission. Studies suggest that about 40% to 50% of patients who have achieved a stable deep molecular response remain in treatment-free remission after stopping first-line treatment.9,10 Of the patients who relapse following TKI discontinuation, 80% relapse within the first 6 months of treatment cessation. Molecular response is regained in almost all patients when treatment is resumed with the same TKI.11
The National Comprehensive Cancer Network (NCCN) recommends considering discontinuation of TKI therapy only outside the setting of a clinical trial and only in patients who consent to discontinuation after a thorough discussion of the potential risks and benefits. The NCCN criteria for patients who may be eligible for discontinuation are listed in Table 1. The Life After Stopping TKIs study reported that 80% of patients with well-controlled chronic phase CML who discontinued TKIs had a clinically meaningful improvement in fatigue. Patients also reported clinically meaningful improvements in depression, diarrhea, sleep disturbance, and pain interference. These symptoms worsened after restarting TKI therapy.12

TKI DISCONTINUATION
Electronic health record data were extracted using structured query language from the US Department of Veterans Affairs (VA) Corporate Data Warehouse (CDW). To be eligible for discontinuation, veterans had to be aged > 18 years, receive oncology care within a Veterans Integrated Services Network (VISN) 21 health care system (HCS) (VA Sierra Nevada HCS, VA Southern Nevada HCS, VA Central California HCS, VA Palo Alto HCS, VA Northern California HCS, and VA San Francisco HCS) or be a veteran referred to a community-based oncology practitioner. Patients had to have a documented diagnosis of chronic phase CML, have an active order for a TKI, be on TKI therapy for ≥ 3 years, and have a stable molecular response (BCR-ABL1 ≤ 0.01% on the International Scale for ≥ 2 years with ≥ 4 tests done ≥ 3 months apart) as of October 1, 2024. Veterans were excluded if they had a history of advanced accelerated phase CML, previous TKI discontinuation trials, nonadherence to the TKI, or if they did not want to consider TKI discontinuation.
This analysis evaluated the potential cost avoidance associated with TKI discontinuation. Cost avoidance was calculated using the average wholesale price of each TKI. Secondary objectives evaluated health outcomes of TKI discontinuation including CML relapse, reported AEs, long-term remission, and TKI withdrawal syndrome. Health outcomes were determined through chart review of AEs and clinic notes documented in the electronic health record during the study time frame.
Baseline information for eligible patients was collected, including age, sex, and race, and chart reviews were completed to evaluate reported AEs associated with therapy. Oncology clinical pharmacy practitioners (CPPs) at each VISN 21 facility were notified of eligible patients to facilitate discussion with oncologists and establish monitoring if therapy was discontinued. Following TKI discontinuation, health outcomes were evaluated, including CML relapse, changes in reported AEs, long-term remission, and TKI withdrawal syndrome. Descriptive statistics were used to analyze the baseline characteristics. Cost avoidance was calculated using the average wholesale price for each TKI. The number of tablets required to reach each patient’s individual dose was taken into consideration when determining the cost avoidance. A dashboard was created using the query from the CDW and was developed in Microsoft Power BI.
Preliminary Results
In FY 2024, VISN 21 had 3819 oncology patients. Twenty-four patients had taken a TKI for ≥ 3 years, 20 had a stable molecular response, and 15 had not previously attempted to discontinue their TKI (Figure 1). Fifteen veterans were eligible for therapy discontinuation for a total potential annual cost avoidance of $1.2 million (Figure 2). Most of the cost avoidance, $935,057 (78%), was attributed to 3 patients on nilotinib. The mean age of the population was 74 years. All patients were male, and 12 (80%) were White. (Table 2). At baseline, 11 patients (73%) were taking imatinib. One patient received oncology care from a community care clinician. All 15 patients decided to remain on therapy.

Abbreviations: CML, chronic myeloid leukemia; TKI, tyrosine kinase inhibitor;
VISN, Veterans Integrated Service Network.

for 15 patients at Veterans Integrated Services Network 21.

DISCUSSION
As a multisite quality improvement initiative, this project raised awareness of TKI therapy discontinuation in select patients with CML. It also sparked collaboration among oncology CPPs and clinicians and stimulated conversations about CML treatment. The development of the TKI discontinuation dashboard provides a population health management tool for CPPs and clinicians to identify eligible patients in the future.
Adherence to TKIs is crucial for disease control and survival in patients with CML. Patients are counseled that poor adherence to therapy may contribute to worsening disease or suboptimal response, the development of resistance, and greater health care costs.13 Therefore, it was a challenge for patients to understand and accept that they could stop TKI therapy after achieving a stable deep molecular response. Discussions with patients about the goal of therapy—suppressing the BCR-ABL oncogene, which they have achieved—could encourage patients to trial therapy discontinuation.
Only small cohort studies have been completed to evaluate the outcomes of therapy discontinuation. Much remains unknown regarding the optimal dose-reduction strategy and the minimum effective dose of each agent. Additionally, understanding the qualities of a good candidate for TKI discontinuation remains a barrier. A similar project was conducted in VISN 17. Five patients were counseled on TKI discontinuation; however, only 1 discontinued TKI therapy. Unfortunately, soon after discontinuing treatment, the patient had to restart therapy. Additional literature will enhance understanding of therapy discontinuation.
An unexpected finding of TKI discontinuation trials has been a reversible phenomenon known as TKI withdrawal syndrome.9 It can occur regardless of the TKI used and results in pruritus and new or worsening musculoskeletal pain within several weeks of TKI discontinuation in about 30% of patients. Symptoms may last several months and may require acetaminophen or nonsteroidal anti-inflammatory drugs for pain control.9,10,14
The potential cost avoidance of $1.2 million is an underestimation because VA contracts allow for greater cost savings. However, that information is confidential and therefore average wholesale price had to be used for this project. Most of the cost avoidance was due to 4 patients who could not tolerate imatinib and used nilotinib, which is more expensive.
Limitations
The small sample size presented some limitations. Of the 3819 oncology patients within VISN 21 in FY 2024, 186 received a TKI and only 15 were eligible for discontinuation. Additionally, challenges emerged when discussing discontinuation with community care clinicians and patients. Community care clinicians were difficult to contact, making it challenging to discuss the project with them. CPPs noted hesitancy among VA clinicians and patients to discontinue a medication for which adherence was continually emphasized.
Conclusions
Discussions about CML TKI discontinuation led to collaboration with the oncology care team and could lead to significant cost avoidance. Barriers to TKI discontinuation included patients’ concern for relapse, risk of discontinuation syndrome, the requirement for close monitoring, and clinician buy-in. Outcome studies are needed to gain a greater understanding of the benefits and risks of therapy discontinuation. In the future, evaluation of possible clinical and biological predictors of successful TKI discontinuation may be beneficial.
Chronic myeloid leukemia (CML) is a hematologic malignancy resulting from an acquired mutation. The mutation results in a reciprocal translocation between the long arms of chromosomes 9 and 22 and is known as the Philadelphia chromosome (Ph), or Ph-positive (Ph+) when present. The translocation results in the formation of a BCR-ABL fusion oncogene, which leads to continuous cell cycling and proliferation, altered differentiation, and a loss of apoptosis.1,2
Until the 1980s, CML was considered fatal.3 The mainstay of treatment consisted of 2 oral chemotherapeutic agents, busulfan and hydroxyurea. These medications did not prevent blast crisis, a fatal form of leukemia.4,5 The introduction of tyrosine kinase inhibitors (TKIs) transformed CML management and improved 10-year overall survival from about 20% to > 80% by delaying the transition to blast crisis. Now, the risk of death from general health conditions or comorbidities is higher than that of CML.6
TKIs target the root cause of CML through inhibition of the BCR-ABL oncoprotein.1,2 For CML, the goals of treatment include maintaining hematologic, cytogenetic, and molecular remission; preventing progression to accelerated phase or blast crisis; minimizing toxicity; and enabling potential cessation of therapy in carefully selected patients.7,8
Small cohort studies suggest that dose reduction of TKIs in patients who achieve optimal responses may reduce the risk of long-term adverse effects (AEs). However, optimal dose-reduction and minimum effective dose of each agent are unknown.7 The ability to maintain undetectable minimal residual disease or disease detectable at a stable low level after TKI discontinuation has been called treatment-free remission. Studies suggest that about 40% to 50% of patients who have achieved a stable deep molecular response remain in treatment-free remission after stopping first-line treatment.9,10 Of the patients who relapse following TKI discontinuation, 80% relapse within the first 6 months of treatment cessation. Molecular response is regained in almost all patients when treatment is resumed with the same TKI.11
The National Comprehensive Cancer Network (NCCN) recommends considering discontinuation of TKI therapy only outside the setting of a clinical trial and only in patients who consent to discontinuation after a thorough discussion of the potential risks and benefits. The NCCN criteria for patients who may be eligible for discontinuation are listed in Table 1. The Life After Stopping TKIs study reported that 80% of patients with well-controlled chronic phase CML who discontinued TKIs had a clinically meaningful improvement in fatigue. Patients also reported clinically meaningful improvements in depression, diarrhea, sleep disturbance, and pain interference. These symptoms worsened after restarting TKI therapy.12

TKI DISCONTINUATION
Electronic health record data were extracted using structured query language from the US Department of Veterans Affairs (VA) Corporate Data Warehouse (CDW). To be eligible for discontinuation, veterans had to be aged > 18 years, receive oncology care within a Veterans Integrated Services Network (VISN) 21 health care system (HCS) (VA Sierra Nevada HCS, VA Southern Nevada HCS, VA Central California HCS, VA Palo Alto HCS, VA Northern California HCS, and VA San Francisco HCS) or be a veteran referred to a community-based oncology practitioner. Patients had to have a documented diagnosis of chronic phase CML, have an active order for a TKI, be on TKI therapy for ≥ 3 years, and have a stable molecular response (BCR-ABL1 ≤ 0.01% on the International Scale for ≥ 2 years with ≥ 4 tests done ≥ 3 months apart) as of October 1, 2024. Veterans were excluded if they had a history of advanced accelerated phase CML, previous TKI discontinuation trials, nonadherence to the TKI, or if they did not want to consider TKI discontinuation.
This analysis evaluated the potential cost avoidance associated with TKI discontinuation. Cost avoidance was calculated using the average wholesale price of each TKI. Secondary objectives evaluated health outcomes of TKI discontinuation including CML relapse, reported AEs, long-term remission, and TKI withdrawal syndrome. Health outcomes were determined through chart review of AEs and clinic notes documented in the electronic health record during the study time frame.
Baseline information for eligible patients was collected, including age, sex, and race, and chart reviews were completed to evaluate reported AEs associated with therapy. Oncology clinical pharmacy practitioners (CPPs) at each VISN 21 facility were notified of eligible patients to facilitate discussion with oncologists and establish monitoring if therapy was discontinued. Following TKI discontinuation, health outcomes were evaluated, including CML relapse, changes in reported AEs, long-term remission, and TKI withdrawal syndrome. Descriptive statistics were used to analyze the baseline characteristics. Cost avoidance was calculated using the average wholesale price for each TKI. The number of tablets required to reach each patient’s individual dose was taken into consideration when determining the cost avoidance. A dashboard was created using the query from the CDW and was developed in Microsoft Power BI.
Preliminary Results
In FY 2024, VISN 21 had 3819 oncology patients. Twenty-four patients had taken a TKI for ≥ 3 years, 20 had a stable molecular response, and 15 had not previously attempted to discontinue their TKI (Figure 1). Fifteen veterans were eligible for therapy discontinuation for a total potential annual cost avoidance of $1.2 million (Figure 2). Most of the cost avoidance, $935,057 (78%), was attributed to 3 patients on nilotinib. The mean age of the population was 74 years. All patients were male, and 12 (80%) were White. (Table 2). At baseline, 11 patients (73%) were taking imatinib. One patient received oncology care from a community care clinician. All 15 patients decided to remain on therapy.

Abbreviations: CML, chronic myeloid leukemia; TKI, tyrosine kinase inhibitor;
VISN, Veterans Integrated Service Network.

for 15 patients at Veterans Integrated Services Network 21.

DISCUSSION
As a multisite quality improvement initiative, this project raised awareness of TKI therapy discontinuation in select patients with CML. It also sparked collaboration among oncology CPPs and clinicians and stimulated conversations about CML treatment. The development of the TKI discontinuation dashboard provides a population health management tool for CPPs and clinicians to identify eligible patients in the future.
Adherence to TKIs is crucial for disease control and survival in patients with CML. Patients are counseled that poor adherence to therapy may contribute to worsening disease or suboptimal response, the development of resistance, and greater health care costs.13 Therefore, it was a challenge for patients to understand and accept that they could stop TKI therapy after achieving a stable deep molecular response. Discussions with patients about the goal of therapy—suppressing the BCR-ABL oncogene, which they have achieved—could encourage patients to trial therapy discontinuation.
Only small cohort studies have been completed to evaluate the outcomes of therapy discontinuation. Much remains unknown regarding the optimal dose-reduction strategy and the minimum effective dose of each agent. Additionally, understanding the qualities of a good candidate for TKI discontinuation remains a barrier. A similar project was conducted in VISN 17. Five patients were counseled on TKI discontinuation; however, only 1 discontinued TKI therapy. Unfortunately, soon after discontinuing treatment, the patient had to restart therapy. Additional literature will enhance understanding of therapy discontinuation.
An unexpected finding of TKI discontinuation trials has been a reversible phenomenon known as TKI withdrawal syndrome.9 It can occur regardless of the TKI used and results in pruritus and new or worsening musculoskeletal pain within several weeks of TKI discontinuation in about 30% of patients. Symptoms may last several months and may require acetaminophen or nonsteroidal anti-inflammatory drugs for pain control.9,10,14
The potential cost avoidance of $1.2 million is an underestimation because VA contracts allow for greater cost savings. However, that information is confidential and therefore average wholesale price had to be used for this project. Most of the cost avoidance was due to 4 patients who could not tolerate imatinib and used nilotinib, which is more expensive.
Limitations
The small sample size presented some limitations. Of the 3819 oncology patients within VISN 21 in FY 2024, 186 received a TKI and only 15 were eligible for discontinuation. Additionally, challenges emerged when discussing discontinuation with community care clinicians and patients. Community care clinicians were difficult to contact, making it challenging to discuss the project with them. CPPs noted hesitancy among VA clinicians and patients to discontinue a medication for which adherence was continually emphasized.
Conclusions
Discussions about CML TKI discontinuation led to collaboration with the oncology care team and could lead to significant cost avoidance. Barriers to TKI discontinuation included patients’ concern for relapse, risk of discontinuation syndrome, the requirement for close monitoring, and clinician buy-in. Outcome studies are needed to gain a greater understanding of the benefits and risks of therapy discontinuation. In the future, evaluation of possible clinical and biological predictors of successful TKI discontinuation may be beneficial.
- Schiffer CA. BCR-ABL tyrosine kinase inhibitors for chronic myelogenous leukemia. N Engl J Med. 2007;357:258-265. doi:10.1056/NEJMct071828
- Hehlmann R, Hochhaus A, Baccarani M; European LeukemiaNet. Chronic myeloid leukaemia. Lancet. 2007;370:342-350. doi:10.1016/S0140-6736(07)61165-9
- Goldman JM, Melo JV. Chronic myeloid leukemia--advances in biology and new approaches to treatment. N Engl J Med. 2003;349:1451-1464. doi:10.1056/NEJMra020777
- Pasic I, Lipton JH. Current approach to the treatment of chronic myeloid leukaemia. Leuk Res. 2017;55:65-78. doi:10.1016/j.leukres.2017.01.005
- Rao KV, Iannucci A, Jabbour E. Current and future clinical strategies in the management of chronic myeloid leukemia. Pharmacotherapy. 2010;30:77S-101S. doi:10.1592/phco.30.pt2.77S
- Cortes J, Pavlovsky C, Saußele S. Chronic myeloid leukaemia. Lancet. 2021;398:1914-1926. doi:10.1016/S0140-6736(21)01204-6
- National Comprehensive Cancer Network (NCCN). NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®). Chronic myeloid leukemia. Version 1.2026. July 16, 2025. Accessed February 8, 2026. https://www.nccn.org /guidelines/guidelines-detail?id=1427
- Hochhaus A, Baccarani M, Silver RT, et al. European LeukemiaNet 2020 recommendations for treating chronic myeloid leukemia. Leukemia. 2020;34:966-984. doi:10.1038/s41375-020-0776-2
- Saußele S, Richter J, Hochhaus A, Mahon F-X. The concept of treatment-free remission in chronic myeloid leukemia. Leukemia. 2016;30:1638-1647. doi:10.1038/leu.2016.115
- Atallah E, Sweet K. Treatment-free remission: the new goal in CML therapy. Curr Hematol Malig Rep. 2021;16:433-439. doi:10.1007/s11899-021-00653-1
- Hehlmann R. The new ELN recommendations for treating CML. J Clin Med. 2020;9:3671. doi:10.3390/jcm9113671
- Atallah E, Schiffer CA, Radich JP , et al. Assessment of outcomes after stopping tyrosine kinase inhibitors among patients with chronic myeloid leukemia: a non-randomized clinical trial. JAMA Oncol. 2021;7:42-50. doi:10.1001/jamaoncol.2020.5774
- Breccia M, Efficace F, Alimena G. Imatinib treatment in chronic myelogenous leukemia: what have we learned so far? Cancer Lett. 2011;300:115-121. doi:10.1016/j.canlet.2010.10.018
- Berman E. How I treat chronic-phase chronic myelogenous leukemia. Blood. 2022;139:3138-3147. doi:10.1182/blood.2021011722
- Schiffer CA. BCR-ABL tyrosine kinase inhibitors for chronic myelogenous leukemia. N Engl J Med. 2007;357:258-265. doi:10.1056/NEJMct071828
- Hehlmann R, Hochhaus A, Baccarani M; European LeukemiaNet. Chronic myeloid leukaemia. Lancet. 2007;370:342-350. doi:10.1016/S0140-6736(07)61165-9
- Goldman JM, Melo JV. Chronic myeloid leukemia--advances in biology and new approaches to treatment. N Engl J Med. 2003;349:1451-1464. doi:10.1056/NEJMra020777
- Pasic I, Lipton JH. Current approach to the treatment of chronic myeloid leukaemia. Leuk Res. 2017;55:65-78. doi:10.1016/j.leukres.2017.01.005
- Rao KV, Iannucci A, Jabbour E. Current and future clinical strategies in the management of chronic myeloid leukemia. Pharmacotherapy. 2010;30:77S-101S. doi:10.1592/phco.30.pt2.77S
- Cortes J, Pavlovsky C, Saußele S. Chronic myeloid leukaemia. Lancet. 2021;398:1914-1926. doi:10.1016/S0140-6736(21)01204-6
- National Comprehensive Cancer Network (NCCN). NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®). Chronic myeloid leukemia. Version 1.2026. July 16, 2025. Accessed February 8, 2026. https://www.nccn.org /guidelines/guidelines-detail?id=1427
- Hochhaus A, Baccarani M, Silver RT, et al. European LeukemiaNet 2020 recommendations for treating chronic myeloid leukemia. Leukemia. 2020;34:966-984. doi:10.1038/s41375-020-0776-2
- Saußele S, Richter J, Hochhaus A, Mahon F-X. The concept of treatment-free remission in chronic myeloid leukemia. Leukemia. 2016;30:1638-1647. doi:10.1038/leu.2016.115
- Atallah E, Sweet K. Treatment-free remission: the new goal in CML therapy. Curr Hematol Malig Rep. 2021;16:433-439. doi:10.1007/s11899-021-00653-1
- Hehlmann R. The new ELN recommendations for treating CML. J Clin Med. 2020;9:3671. doi:10.3390/jcm9113671
- Atallah E, Schiffer CA, Radich JP , et al. Assessment of outcomes after stopping tyrosine kinase inhibitors among patients with chronic myeloid leukemia: a non-randomized clinical trial. JAMA Oncol. 2021;7:42-50. doi:10.1001/jamaoncol.2020.5774
- Breccia M, Efficace F, Alimena G. Imatinib treatment in chronic myelogenous leukemia: what have we learned so far? Cancer Lett. 2011;300:115-121. doi:10.1016/j.canlet.2010.10.018
- Berman E. How I treat chronic-phase chronic myelogenous leukemia. Blood. 2022;139:3138-3147. doi:10.1182/blood.2021011722
Potential Tyrosine Kinase Inhibitor Therapy Discontinuation for Patients With Chronic Myeloid Leukemia in a VA Regional Network
Potential Tyrosine Kinase Inhibitor Therapy Discontinuation for Patients With Chronic Myeloid Leukemia in a VA Regional Network
A Legacy in Dermatology: Dr. Vincent A. DeLeo Named AAD Master Dermatologist
A Legacy in Dermatology: Dr. Vincent A. DeLeo Named AAD Master Dermatologist
The Cutis editorial staff is proud to announce that Vincent A. DeLeo, MD, Editor-in-Chief, was honored with the Master Dermatologist Award at the 2026 Annual Meeting of the American Academy of Dermatology (AAD) in Denver, Colorado.
Presented as part of the AAD’s “Stars of the Academy” program, this award is reserved for physicians whose careers have advanced dermatology through leadership, service, and meaningful contributions to patient care, education, and research. The award reflects Dr. DeLeo’s impact across the specialty.

“Vince’s passion for dermatology has impacted all aspects of our specialty. He has been at the forefront of basic science research, clinical dermatology, education, mentoring, and leadership of specialty organizations and societies.” –Susan C. Taylor, MD
During the presentation, outgoing AAD president Susan C. Taylor, MD, emphasized Dr. DeLeo’s wide-ranging influence, noting his reputation as a researcher, compassionate physician, and skilled diagnostician. He is adept at managing complex cases and improving patient outcomes. Dr. DeLeo is widely recognized for his expertise in contact dermatitis, photomedicine, and photoprotection, as well as for his contributions to dermatologic education.
Beyond his clinical and editorial leadership of Cutis for the past 25 years, Dr. DeLeo is committed to mentorship and leadership by serving on the AAD Board of Directors as well as other specialty organizations such as the American Contact Dermatitis Society.
We congratulate Dr. DeLeo on this well-deserved distinction and thank him for his continued vision and dedication to our readers and the specialty at large.
The Cutis editorial staff is proud to announce that Vincent A. DeLeo, MD, Editor-in-Chief, was honored with the Master Dermatologist Award at the 2026 Annual Meeting of the American Academy of Dermatology (AAD) in Denver, Colorado.
Presented as part of the AAD’s “Stars of the Academy” program, this award is reserved for physicians whose careers have advanced dermatology through leadership, service, and meaningful contributions to patient care, education, and research. The award reflects Dr. DeLeo’s impact across the specialty.

“Vince’s passion for dermatology has impacted all aspects of our specialty. He has been at the forefront of basic science research, clinical dermatology, education, mentoring, and leadership of specialty organizations and societies.” –Susan C. Taylor, MD
During the presentation, outgoing AAD president Susan C. Taylor, MD, emphasized Dr. DeLeo’s wide-ranging influence, noting his reputation as a researcher, compassionate physician, and skilled diagnostician. He is adept at managing complex cases and improving patient outcomes. Dr. DeLeo is widely recognized for his expertise in contact dermatitis, photomedicine, and photoprotection, as well as for his contributions to dermatologic education.
Beyond his clinical and editorial leadership of Cutis for the past 25 years, Dr. DeLeo is committed to mentorship and leadership by serving on the AAD Board of Directors as well as other specialty organizations such as the American Contact Dermatitis Society.
We congratulate Dr. DeLeo on this well-deserved distinction and thank him for his continued vision and dedication to our readers and the specialty at large.
The Cutis editorial staff is proud to announce that Vincent A. DeLeo, MD, Editor-in-Chief, was honored with the Master Dermatologist Award at the 2026 Annual Meeting of the American Academy of Dermatology (AAD) in Denver, Colorado.
Presented as part of the AAD’s “Stars of the Academy” program, this award is reserved for physicians whose careers have advanced dermatology through leadership, service, and meaningful contributions to patient care, education, and research. The award reflects Dr. DeLeo’s impact across the specialty.

“Vince’s passion for dermatology has impacted all aspects of our specialty. He has been at the forefront of basic science research, clinical dermatology, education, mentoring, and leadership of specialty organizations and societies.” –Susan C. Taylor, MD
During the presentation, outgoing AAD president Susan C. Taylor, MD, emphasized Dr. DeLeo’s wide-ranging influence, noting his reputation as a researcher, compassionate physician, and skilled diagnostician. He is adept at managing complex cases and improving patient outcomes. Dr. DeLeo is widely recognized for his expertise in contact dermatitis, photomedicine, and photoprotection, as well as for his contributions to dermatologic education.
Beyond his clinical and editorial leadership of Cutis for the past 25 years, Dr. DeLeo is committed to mentorship and leadership by serving on the AAD Board of Directors as well as other specialty organizations such as the American Contact Dermatitis Society.
We congratulate Dr. DeLeo on this well-deserved distinction and thank him for his continued vision and dedication to our readers and the specialty at large.
A Legacy in Dermatology: Dr. Vincent A. DeLeo Named AAD Master Dermatologist
A Legacy in Dermatology: Dr. Vincent A. DeLeo Named AAD Master Dermatologist
Tinted vs Nontinted Sunscreens: Attenuation of Near-Visible UVA and Visible Light and Implications for Skin of Color
Tinted vs Nontinted Sunscreens: Attenuation of Near-Visible UVA and Visible Light and Implications for Skin of Color
Individuals with skin of color (SOC) are disproportionately affected by hyperpigmentation disorders such as melasma and postinflammatory hyperpigmentation following sun exposure. Although epidermal melanin provides UVB protection, susceptibility to pigmentary responses from longer UVA wavelengths and visible light (VL) remains, particularly the highest energy wavelengths of blue light (BL) between 400 and 450 nm.1 Blue light can induce immediate and persistent pigment darkening in those with Fitzpatrick skin types IV to VI, and trace amounts of near-visible UVA (NV-UVA) between 370 and 400 nm can synergize with VL to amplify pigmentation and erythema responses.2
Current photoprotection recommendations emphasize sun protection factor (SPF) ratings of 30+ and broad-spectrum labeling; however, under the US Food and Drug Administration standards, the broad-spectrum designation is based solely on achieving a mean critical wavelength of 370 nm or higher, which does not ensure meaningful attenuation of NV-UVA or VL wavelengths.3 Tinted sunscreens containing iron oxides (FeO) have been shown to improve protection against these pigment-inducing wavelengths,4 yet quantitative comparisons between tinted and nontinted commercial sunscreen products remain limited.
To address the gap in understanding about tinted vs nontinted commercial sunscreen products, we conducted an in vitro spectrophotometric comparative analysis. The objectives were to quantify NV-UVA and BL attenuation across products and evaluate whether formulation characteristics (eg, SPF rating, filter types and concentration, the presence and depth of tint, antioxidant content) would correlate with improved photoprotection in pigment-sensitive wavelengths. We hypothesized that formulation features such as higher SPF, inorganic filters, and the presence of tint antioxidants would be associated with superior NV-UVA and BL attenuation compared with nontinted formulations.
Methods
Sunscreen Selection—A convenience sample of 23 broad-spectrum sunscreens commercially available at drugstores was selected to reflect easily accessible options. Six sunscreen brands with tinted (n=13) and nontinted (n=10) counterpart formulations were included. Filter category (mineral and/or chemical), SPF, UV filter type and concentration, tint shade (light, medium, medium/deep, deep), number of photoprotective antioxidants (diethylhexyl syringylidenemalonate, vitamin E, vitamin C, licochalcone A, and glycyrrhetinic acid), and presence of FeO were recorded.
Substrate Preparation—Testing was performed using standardized polymethyl methacrylate (PMMA) plates. Sunscreens were mixed prior to application and applied at 1.3 mg/cm² per the European Cosmetic and Perfumery Association (COLIPA) UVA testing guidelines.5 Plates were reweighed to confirm dosing and dried in a dark environment for at least 15 minutes prior to testing.
Spectrophotometric Measurements—Spectral transmittance was measured from 250 to 450 nm using a spectrophotometer equipped with a xenon flash lamp (energy <0.2 J/cm²). Baseline transmission was recorded using untreated PMMA plates. Five scans were averaged per plate. Analyses focused on NV-UVA transmittance from 380 to 400 nm and peak BL transmission at 450 nm.
Mean NV-UVA transmittance was calculated as the arithmetic mean of percent transmittance measured at 1-nm increments from 380 to 400 nm (n=21). Because of the steep rise in transmittance between 380 and 400 nm and subsequent plateau into the visible range, this approach was used to approximate the area under the transmittance-wavelength curve over the specified interval, enabling direct comparison of NV-UVA penetration between formulations.
Statistical Analysis—Descriptive statistics were used to summarize transmittance values. Spearman rank correlation was used to assess associations between formulation variables and spectral attenuation. Analysis of covariance was used to evaluate the effect of FeO on transmittance while adjusting for SPF or filter type. The Mann-Whitney U test was used to compare NV-UVA and blue light transmittance between FeO-containing mineral and chemical formulations. Statistical significance was set at P<.05.
Results
Across broad-spectrum sunscreen formulations (N=23), mean SPF values were 40.4 (range, 30-70), and the mean number of antioxidants in the ingredient list was 1.5 (range, 0-4). Mean NV-UVA transmittance was 16.7% (range, 0.1%-55.0%) and mean BL transmittance was 44.3% (range, 0.3%-97.5%)(eTable 1).
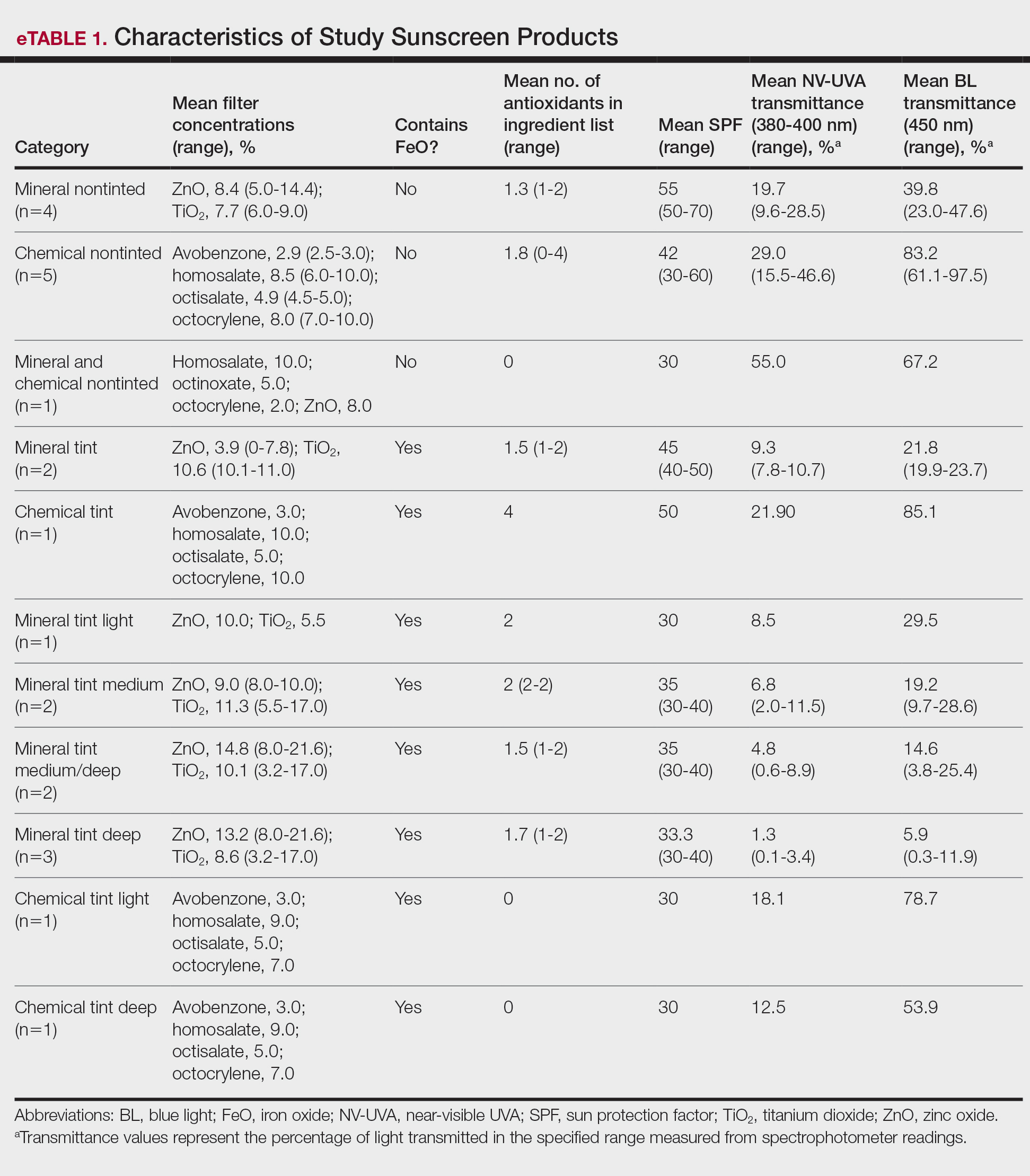
The mean labeled zinc oxide (ZnO) concentration among ZnO-containing formulations (n=14) was 10.5% (range, 5.0%-21.6%), with mean NV-UVA and BL transmittance of 12.6% (range, 0.1%-55.0%) and 25.8% (range, 0.3%-67.2%), respectively. Mean NV-UVA and BL transmittance were 26.7% (range, 9.6%-55.0%) and 45.6% (range, 23.0%-67.2%) among ZnO formulations without FeO (n=5), compared with lower transmittance of 4.8% (range, 0.1%-11.5%) and 14.9% (range, 0.3%-29.5%) in ZnO formulations containing FeO (n=9).
The mean labeled titanium dioxide (TiO2) concentration among TiO2-containing formulations (n=14) was 9.0% (range, 3.2%-17.0%), with corresponding mean NV-UVA and BL transmittance of 9.5% (range, 0.1%-28.5%) and 22.7% (range, 0.3%-47.6%), respectively. Among TiO2 formulations without FeO (n=4), mean NV-UVA and BL transmittance was 19.7% (range, 9.6%-28.5%) and 39.8% (range, 23.0%-47.6%), while FeO-containing TiO2 formulations (n=10) showed lower mean NV-UVA and BL transmittance of 5.4% (range, 0.1%-11.5%) and 15.8% (range, 0.3%-29.5%), respectively. The mean labeled avobenzone concentration among avobenzone-containing formulations (n=8) was 2.9% (range, 2.5%-3%), with mean NV-UVA and BL transmittance of 24.7% (range, 10.2%-46.6%) and 79.2% (range, 53.9%-97.5%). Formulations without FeO (n=5) had mean NV-UVA and BL transmittance of 29.0% (range, 10.2%-46.6%) and 83.2% (range, 61.1%-97.5%), whereas FeO-containing products (n=3) demonstrated lower mean NV-UVA and BL transmittance of 17.5% (range, 12.5%-21.9%) and 72.6% (range, 53.9%-85.1%), respectively.
Among products containing ZnO, TiO2, and avobenzone, the specific UV filter concentrations showed no statistically significant correlation with NV-UVA or BL transmittance (all P>.05). Iron oxide presence significantly correlated with lower NV-UVA (r=–0.67; P=.00042) and lower BL transmittance (r=–0.57; P=.0046). The number of antioxidants in the ingredient list did not correlate with NV-UVA transmittance (r=–0.28; P=.19) or BL transmittance (r=–0.16; P=.47). Sun protection factor was not significantly correlated with either wavelength range (Table 1).
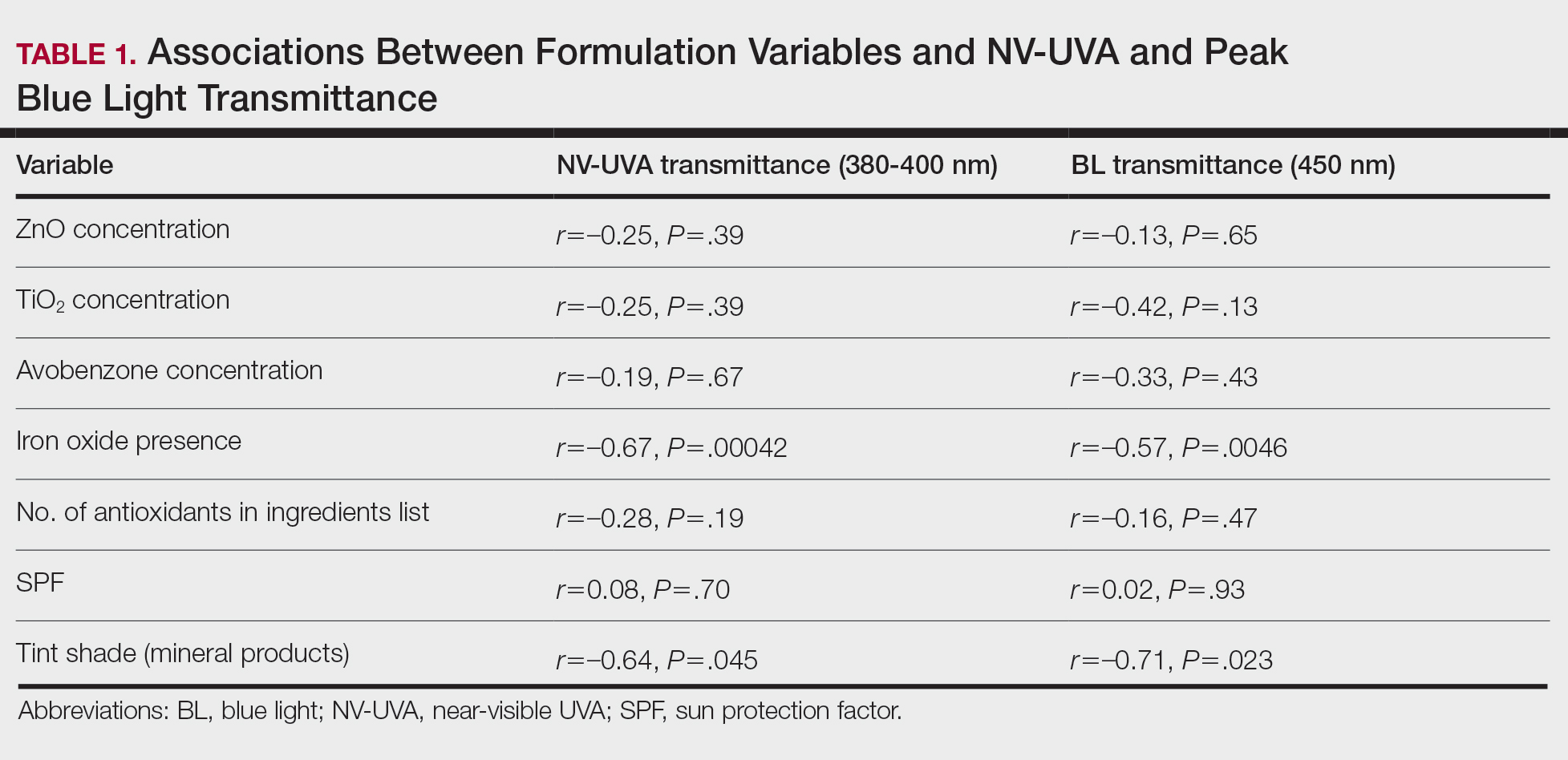
Tint shade was treated as an ordinal variable (light, medium, medium/deep, and deep; medium was considered the universal shade). Increasing tint shade depth was significantly associated with reduced NV-UVA (r=–0.64; P=.045) and BL (r=–0.71; P=.023), suggesting a dose-response relationship wherein darker tints exhibited greater attenuation of pigment-relevant wavelengths. Among mineral filter formulations, tinted products demonstrated lower NV-UVA and BL transmittance compared with their nontinted counterparts, with deeper tints providing the greatest reduction in transmittance (eFigure 1). Similar results were observed for chemical filter formulations with greater attenuation in the NV-UVA and BL range for tinted versus nontinted products with greater variability across shade depths (eFigure 2).
After adjusting for SPF, FeO presence remained significantly associated with reduced NV-UVA (F[1,20]=26.9; P<.001) and BL transmittance (F[1,20]=11.7; P=.003). After adjusting for filter type (mineral vs chemical), FeO remained significantly associated with NV-UVA (F[1,19]=10.1; P=.004) and BL transmittance (F[1,19]=10.4; P=.005)(Table 2).

Among FeO-containing products, mineral filters demonstrated significantly lower NV-UVA transmittance compared with chemical filters (median, 5.58% [interquartile range (IQR), 0.59%-9.35%] vs 18.10% [IQR, 12.47%-21.90%]; U=0.00; P=.007). The same was true for BL transmittance (median, 15.90% [IQR, 5.00%-26.20%] vs 78.70% [IQR, 53.90%-85.10%]; U=0.00; P=.007). The differences in spectral transmittance between FeO-containing mineral and chemical filter formulations are illustrated in eFigure 3, with mineral-based products demonstrating lower transmittance, particularly across the upper NV-UVA range and across the BL range. These results indicated greater pigment-relevant photoprotection with mineral vs chemical filters (eTable 2).
Comment
Our initial hypothesis proposed that tinted sunscreens would provide greater NV-UVA and BL attenuation than nontinted formulations, and that characteristics such as inorganic filter content, SPF rating, and antioxidants would correlate with improved protection in pigment-sensitive wavelengths. Our findings partially supported this hypothesis. In this analysis, substantial variability in the NV-UVA and BL transmittance was observed despite all products meeting broad-spectrum criteria. Nontinted mineral and chemical sunscreens exhibited high transmittance in these pigment-related wavelengths, reaching values as high as 55.0% for NV-UVA and 97.5% for BL. These findings align with prior analysis demonstrating that while broad-spectrum sunscreens available in the United States may meet the current critical wavelength criteria for protection in the UVA range, they still may transmit 30% to 66% of available UVA over 2 hours between formulations with equivalent SPF label values.6
Recent analyses show that sunscreen recommendations in lay media rarely incorporate input from board-certified dermatologists for individuals with SOC and disproportionately favor nontinted chemical formulations, despite the high prevalence of pigmentary disorders in this population.7 Near-visible UVA and BL have been demonstrated to be biologically relevant pigment-inducing wavelengths, both in vitro and in vivo, particularly in individuals with SOC, yet broad-spectrum labeling does not ensure protection against these spectra.8 Pigmentary tints such as FeO have demonstrated enhanced attenuation in this spectral region in vivo and may provide more reliable coverage than products with broad-spectrum designation alone.4,9 Treatment options for pigmentary disorders such as melasma tend to be palliative and costly, making optimized photoprotection a critical component of care to reduce ongoing pigmentary stimuli.10
Formulations containing FeO demonstrated significantly lower NV-UVA (P<.001) and BL transmittance (P=.003) on average; however, transmittance values ranged widely (NV-UVA: 0.10%-21.90%, BL: 0.30%-85.10%), indicating that FeO presence alone does not determine the magnitude of attenuation. Notably, among FeO-containing products, mineral filters provided significantly lower NV-UVA and BL transmittance compared with chemical filters (P=.007 for both), suggesting that filter type further modulates pigment-relevant photoprotection. Tinted formulations may improve compliance with product use by reducing the white cast and improve shade matching to find suitable options for deeper skin tones,11 but the highly variable photoprotection offered raises concerns about clinical benefit. Although deeper tints showed greater attenuation, pigment concentrations and combinations are not disclosed by manufacturers as FeO is not considered an active ingredient. Darker shades are not practical across all skin tones in individuals with SOC, which underscores the need for standardized pigment metrics and shade-inclusive options.
While avobenzone and ZnO are the only US Food and Drug Administration–approved sunscreen active ingredients that extend protection beyond 360 nm,12 both exhibited reduced attenuation beyond the longer end of the UVA spectrum. Formulation characteristics, including the concentration of ZnO, TiO2, and/or avobenzone as well as SPF, did not correlate with NV-UVA or BL attenuation. In the adjusted analysis, FeO presence remained significantly associated with reduced transmittance after adjusting for SPF (NV-UVA: P<.001, BL: P=.003) or filter type (NV-UVA: P=.004, BL: P=.005). These findings suggest that the presence of FeO, rather than UV filters or SPF ratings, supports attenuation in the 380 to 450–nm range, indicating a functional benefit in addition to improved cosmesis.13
Although antioxidants in specific combinations have shown promise in vivo, no association was observed between the number of antioxidants present and NV-UVA or BL attenuation compared with added tint.14 This suggests that specific antioxidant combinations and their concentrations may be more relevant than the total count.
Several study limitations need to be considered in interpreting our results, including a modest number of products, controlled in vitro testing conditions, and an incomplete representation of products with pigment concentrations and shade ranges marketed to individuals with SOC across all price categories, despite our focus on affordable, commercially available options. Moreover, PMMA-based spectrophotometry does not account for skin surface heterogeneity, photodegradation, sweat, oil, friction, or application variability, which may alter real-world performance. Additionally, FeO concentrations could not be quantified beyond labeling of tint shade depth, preventing a true assessment of dose-response effects. These limitations may reduce generalizability and highlight the need for complementary in vivo studies to assess clinically relevant outcomes such as persistent pigment darkening. For this reason, caution is warranted in extrapolating these spectral findings to clinical efficacy.
Conclusion
Given the susceptibility of individuals with SOC to pigmentary disorders driven by NV-UVA and BL, our findings support further development and study of FeO-containing sunscreens that address clinically relevant wavelengths. Wide variability in photo-attenuation among tinted formulations underscores the need for evidence-based recommendations, with further studies needed to guide photoprotection strategies for populations with SOC.
- Marionnet C, Piffaut V, Sasai J, et al. A precise analysis of the relative contribution of UVA1 and visible light colour domains in solar light-induced skin pigmentation. J Eur Acad Dermatol Venereol. 2023;37(suppl 4):3-11. doi:10.1111/jdv.18948
- Kohli I, Chaowattanapanit S, Mohammad TF, et al. Synergistic effects of long-wavelength ultraviolet A1 and visible light on pigmentation and erythema. Br J Dermatol. 2018;178:1173-1180. doi: 10.1111/bjd.15940
- US Food and Drug Administration. Over-the-counter monograph M020: sunscreen drug products for over-the-counter human use. September 24, 2021. Accessed April 7, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/omuf/monographs/OTCMonograph_M020-SunscreenDrugProductsforOTCHumanUse09242021.pdf
- Grimes PE, Paturi J, Chen Y, et al. Photoprotection efficacy of sun protection factor and iron oxide formulations in diverse skin with melasma and photodamage. J Drugs Dermatol. 2025;24:662-667. doi:10.36849/JDD.9240
- Moyal D, Alard V, Bertin C, et al. The revised COLIPA in vitro UVA method. Int J Cosmet Sci. 2013;35:35-40. doi:10.1111/j.1468-2494.2012.00748.x
- Coelho SG, Rua D, Miller SA, et al. Suboptimal UVA attenuation by broad spectrum sunscreens under outdoor solar conditions contributes to lifetime UVA burden. Photodermatol Photoimmunol Photomed. 2020;36:42-52. doi:10.1111/phpp.12503
- Song H, Beckles A, Salian P, et al. Sunscreen recommendations for patients with skin of color in the popular press and in the dermatology clinic. Int J Womens Dermatol. 2020;7:165-170. doi:10.1016/j.ijwd.2020.10.008
- Lawrence KP, Douki T, Sarkany RPE, et al. The UV/visible radiation boundary region (385-405 nm) damages skin cells and induces “dark” cyclobutane pyrimidine dimers in human skin in vivo. Sci Rep. 2018;8:12722. doi:10.1038/s41598-018-30738-6
- Ezekwe N, Pourang A, Lyons AB, et al. Evaluation of the protection of sunscreen products against long wavelength ultraviolet A1 and visible light-induced biological effects. Photodermatol Photoimmunol Photomed. 2024;40:E12937. doi:10.1111/phpp.12937
- Mpofana N, Chibi B, Gqaleni N, et al. Melasma in people with darker skin types: a scoping review protocol on prevalence, treatment options for melasma and impact on quality of life. Syst Rev. 2023;12:139. doi:10.1186/s13643-023-02300-7
- Wang JY, Patel P, Philip R, et al. Sunscreen practices and preferences of skin of color patients. J Drugs Dermatol. 2024;23:456-462. doi:10.36849/JDD.8268
- Beasley DG, Meyer TA. Characterization of the UVA protection provided by avobenzone, zinc oxide, and titanium dioxide in broad-spectrum sunscreen products. Am J Clin Dermatol. 2010;11:413-421. doi:10.2165/11537050-000000000-00000
- Morgado-Carrasco D, Delgado J, Prudkin-Silva Let al. Sunscreens prescribed to patients with skin of color and/or with melasma: a survey of 221 dermatologists and dermatology residents in Spain. Photodermatol Photoimmunol Photomed. 2024;40:E12996. doi:10.1111/phpp.12996
- Ruvolo E, Boothby-Shoemaker W, Kumar N, et al. Evaluation of efficacy of antioxidant-enriched sunscreen prodcuts against long wavelength ultraviolet A1 and visible light. Int J Cosmet Sci. 2022;44:394-402. doi:10.1111/ics.12785
Individuals with skin of color (SOC) are disproportionately affected by hyperpigmentation disorders such as melasma and postinflammatory hyperpigmentation following sun exposure. Although epidermal melanin provides UVB protection, susceptibility to pigmentary responses from longer UVA wavelengths and visible light (VL) remains, particularly the highest energy wavelengths of blue light (BL) between 400 and 450 nm.1 Blue light can induce immediate and persistent pigment darkening in those with Fitzpatrick skin types IV to VI, and trace amounts of near-visible UVA (NV-UVA) between 370 and 400 nm can synergize with VL to amplify pigmentation and erythema responses.2
Current photoprotection recommendations emphasize sun protection factor (SPF) ratings of 30+ and broad-spectrum labeling; however, under the US Food and Drug Administration standards, the broad-spectrum designation is based solely on achieving a mean critical wavelength of 370 nm or higher, which does not ensure meaningful attenuation of NV-UVA or VL wavelengths.3 Tinted sunscreens containing iron oxides (FeO) have been shown to improve protection against these pigment-inducing wavelengths,4 yet quantitative comparisons between tinted and nontinted commercial sunscreen products remain limited.
To address the gap in understanding about tinted vs nontinted commercial sunscreen products, we conducted an in vitro spectrophotometric comparative analysis. The objectives were to quantify NV-UVA and BL attenuation across products and evaluate whether formulation characteristics (eg, SPF rating, filter types and concentration, the presence and depth of tint, antioxidant content) would correlate with improved photoprotection in pigment-sensitive wavelengths. We hypothesized that formulation features such as higher SPF, inorganic filters, and the presence of tint antioxidants would be associated with superior NV-UVA and BL attenuation compared with nontinted formulations.
Methods
Sunscreen Selection—A convenience sample of 23 broad-spectrum sunscreens commercially available at drugstores was selected to reflect easily accessible options. Six sunscreen brands with tinted (n=13) and nontinted (n=10) counterpart formulations were included. Filter category (mineral and/or chemical), SPF, UV filter type and concentration, tint shade (light, medium, medium/deep, deep), number of photoprotective antioxidants (diethylhexyl syringylidenemalonate, vitamin E, vitamin C, licochalcone A, and glycyrrhetinic acid), and presence of FeO were recorded.
Substrate Preparation—Testing was performed using standardized polymethyl methacrylate (PMMA) plates. Sunscreens were mixed prior to application and applied at 1.3 mg/cm² per the European Cosmetic and Perfumery Association (COLIPA) UVA testing guidelines.5 Plates were reweighed to confirm dosing and dried in a dark environment for at least 15 minutes prior to testing.
Spectrophotometric Measurements—Spectral transmittance was measured from 250 to 450 nm using a spectrophotometer equipped with a xenon flash lamp (energy <0.2 J/cm²). Baseline transmission was recorded using untreated PMMA plates. Five scans were averaged per plate. Analyses focused on NV-UVA transmittance from 380 to 400 nm and peak BL transmission at 450 nm.
Mean NV-UVA transmittance was calculated as the arithmetic mean of percent transmittance measured at 1-nm increments from 380 to 400 nm (n=21). Because of the steep rise in transmittance between 380 and 400 nm and subsequent plateau into the visible range, this approach was used to approximate the area under the transmittance-wavelength curve over the specified interval, enabling direct comparison of NV-UVA penetration between formulations.
Statistical Analysis—Descriptive statistics were used to summarize transmittance values. Spearman rank correlation was used to assess associations between formulation variables and spectral attenuation. Analysis of covariance was used to evaluate the effect of FeO on transmittance while adjusting for SPF or filter type. The Mann-Whitney U test was used to compare NV-UVA and blue light transmittance between FeO-containing mineral and chemical formulations. Statistical significance was set at P<.05.
Results
Across broad-spectrum sunscreen formulations (N=23), mean SPF values were 40.4 (range, 30-70), and the mean number of antioxidants in the ingredient list was 1.5 (range, 0-4). Mean NV-UVA transmittance was 16.7% (range, 0.1%-55.0%) and mean BL transmittance was 44.3% (range, 0.3%-97.5%)(eTable 1).

The mean labeled zinc oxide (ZnO) concentration among ZnO-containing formulations (n=14) was 10.5% (range, 5.0%-21.6%), with mean NV-UVA and BL transmittance of 12.6% (range, 0.1%-55.0%) and 25.8% (range, 0.3%-67.2%), respectively. Mean NV-UVA and BL transmittance were 26.7% (range, 9.6%-55.0%) and 45.6% (range, 23.0%-67.2%) among ZnO formulations without FeO (n=5), compared with lower transmittance of 4.8% (range, 0.1%-11.5%) and 14.9% (range, 0.3%-29.5%) in ZnO formulations containing FeO (n=9).
The mean labeled titanium dioxide (TiO2) concentration among TiO2-containing formulations (n=14) was 9.0% (range, 3.2%-17.0%), with corresponding mean NV-UVA and BL transmittance of 9.5% (range, 0.1%-28.5%) and 22.7% (range, 0.3%-47.6%), respectively. Among TiO2 formulations without FeO (n=4), mean NV-UVA and BL transmittance was 19.7% (range, 9.6%-28.5%) and 39.8% (range, 23.0%-47.6%), while FeO-containing TiO2 formulations (n=10) showed lower mean NV-UVA and BL transmittance of 5.4% (range, 0.1%-11.5%) and 15.8% (range, 0.3%-29.5%), respectively. The mean labeled avobenzone concentration among avobenzone-containing formulations (n=8) was 2.9% (range, 2.5%-3%), with mean NV-UVA and BL transmittance of 24.7% (range, 10.2%-46.6%) and 79.2% (range, 53.9%-97.5%). Formulations without FeO (n=5) had mean NV-UVA and BL transmittance of 29.0% (range, 10.2%-46.6%) and 83.2% (range, 61.1%-97.5%), whereas FeO-containing products (n=3) demonstrated lower mean NV-UVA and BL transmittance of 17.5% (range, 12.5%-21.9%) and 72.6% (range, 53.9%-85.1%), respectively.
Among products containing ZnO, TiO2, and avobenzone, the specific UV filter concentrations showed no statistically significant correlation with NV-UVA or BL transmittance (all P>.05). Iron oxide presence significantly correlated with lower NV-UVA (r=–0.67; P=.00042) and lower BL transmittance (r=–0.57; P=.0046). The number of antioxidants in the ingredient list did not correlate with NV-UVA transmittance (r=–0.28; P=.19) or BL transmittance (r=–0.16; P=.47). Sun protection factor was not significantly correlated with either wavelength range (Table 1).

Tint shade was treated as an ordinal variable (light, medium, medium/deep, and deep; medium was considered the universal shade). Increasing tint shade depth was significantly associated with reduced NV-UVA (r=–0.64; P=.045) and BL (r=–0.71; P=.023), suggesting a dose-response relationship wherein darker tints exhibited greater attenuation of pigment-relevant wavelengths. Among mineral filter formulations, tinted products demonstrated lower NV-UVA and BL transmittance compared with their nontinted counterparts, with deeper tints providing the greatest reduction in transmittance (eFigure 1). Similar results were observed for chemical filter formulations with greater attenuation in the NV-UVA and BL range for tinted versus nontinted products with greater variability across shade depths (eFigure 2).


After adjusting for SPF, FeO presence remained significantly associated with reduced NV-UVA (F[1,20]=26.9; P<.001) and BL transmittance (F[1,20]=11.7; P=.003). After adjusting for filter type (mineral vs chemical), FeO remained significantly associated with NV-UVA (F[1,19]=10.1; P=.004) and BL transmittance (F[1,19]=10.4; P=.005)(Table 2).

Among FeO-containing products, mineral filters demonstrated significantly lower NV-UVA transmittance compared with chemical filters (median, 5.58% [interquartile range (IQR), 0.59%-9.35%] vs 18.10% [IQR, 12.47%-21.90%]; U=0.00; P=.007). The same was true for BL transmittance (median, 15.90% [IQR, 5.00%-26.20%] vs 78.70% [IQR, 53.90%-85.10%]; U=0.00; P=.007). The differences in spectral transmittance between FeO-containing mineral and chemical filter formulations are illustrated in eFigure 3, with mineral-based products demonstrating lower transmittance, particularly across the upper NV-UVA range and across the BL range. These results indicated greater pigment-relevant photoprotection with mineral vs chemical filters (eTable 2).


Comment
Our initial hypothesis proposed that tinted sunscreens would provide greater NV-UVA and BL attenuation than nontinted formulations, and that characteristics such as inorganic filter content, SPF rating, and antioxidants would correlate with improved protection in pigment-sensitive wavelengths. Our findings partially supported this hypothesis. In this analysis, substantial variability in the NV-UVA and BL transmittance was observed despite all products meeting broad-spectrum criteria. Nontinted mineral and chemical sunscreens exhibited high transmittance in these pigment-related wavelengths, reaching values as high as 55.0% for NV-UVA and 97.5% for BL. These findings align with prior analysis demonstrating that while broad-spectrum sunscreens available in the United States may meet the current critical wavelength criteria for protection in the UVA range, they still may transmit 30% to 66% of available UVA over 2 hours between formulations with equivalent SPF label values.6
Recent analyses show that sunscreen recommendations in lay media rarely incorporate input from board-certified dermatologists for individuals with SOC and disproportionately favor nontinted chemical formulations, despite the high prevalence of pigmentary disorders in this population.7 Near-visible UVA and BL have been demonstrated to be biologically relevant pigment-inducing wavelengths, both in vitro and in vivo, particularly in individuals with SOC, yet broad-spectrum labeling does not ensure protection against these spectra.8 Pigmentary tints such as FeO have demonstrated enhanced attenuation in this spectral region in vivo and may provide more reliable coverage than products with broad-spectrum designation alone.4,9 Treatment options for pigmentary disorders such as melasma tend to be palliative and costly, making optimized photoprotection a critical component of care to reduce ongoing pigmentary stimuli.10
Formulations containing FeO demonstrated significantly lower NV-UVA (P<.001) and BL transmittance (P=.003) on average; however, transmittance values ranged widely (NV-UVA: 0.10%-21.90%, BL: 0.30%-85.10%), indicating that FeO presence alone does not determine the magnitude of attenuation. Notably, among FeO-containing products, mineral filters provided significantly lower NV-UVA and BL transmittance compared with chemical filters (P=.007 for both), suggesting that filter type further modulates pigment-relevant photoprotection. Tinted formulations may improve compliance with product use by reducing the white cast and improve shade matching to find suitable options for deeper skin tones,11 but the highly variable photoprotection offered raises concerns about clinical benefit. Although deeper tints showed greater attenuation, pigment concentrations and combinations are not disclosed by manufacturers as FeO is not considered an active ingredient. Darker shades are not practical across all skin tones in individuals with SOC, which underscores the need for standardized pigment metrics and shade-inclusive options.
While avobenzone and ZnO are the only US Food and Drug Administration–approved sunscreen active ingredients that extend protection beyond 360 nm,12 both exhibited reduced attenuation beyond the longer end of the UVA spectrum. Formulation characteristics, including the concentration of ZnO, TiO2, and/or avobenzone as well as SPF, did not correlate with NV-UVA or BL attenuation. In the adjusted analysis, FeO presence remained significantly associated with reduced transmittance after adjusting for SPF (NV-UVA: P<.001, BL: P=.003) or filter type (NV-UVA: P=.004, BL: P=.005). These findings suggest that the presence of FeO, rather than UV filters or SPF ratings, supports attenuation in the 380 to 450–nm range, indicating a functional benefit in addition to improved cosmesis.13
Although antioxidants in specific combinations have shown promise in vivo, no association was observed between the number of antioxidants present and NV-UVA or BL attenuation compared with added tint.14 This suggests that specific antioxidant combinations and their concentrations may be more relevant than the total count.
Several study limitations need to be considered in interpreting our results, including a modest number of products, controlled in vitro testing conditions, and an incomplete representation of products with pigment concentrations and shade ranges marketed to individuals with SOC across all price categories, despite our focus on affordable, commercially available options. Moreover, PMMA-based spectrophotometry does not account for skin surface heterogeneity, photodegradation, sweat, oil, friction, or application variability, which may alter real-world performance. Additionally, FeO concentrations could not be quantified beyond labeling of tint shade depth, preventing a true assessment of dose-response effects. These limitations may reduce generalizability and highlight the need for complementary in vivo studies to assess clinically relevant outcomes such as persistent pigment darkening. For this reason, caution is warranted in extrapolating these spectral findings to clinical efficacy.
Conclusion
Given the susceptibility of individuals with SOC to pigmentary disorders driven by NV-UVA and BL, our findings support further development and study of FeO-containing sunscreens that address clinically relevant wavelengths. Wide variability in photo-attenuation among tinted formulations underscores the need for evidence-based recommendations, with further studies needed to guide photoprotection strategies for populations with SOC.
Individuals with skin of color (SOC) are disproportionately affected by hyperpigmentation disorders such as melasma and postinflammatory hyperpigmentation following sun exposure. Although epidermal melanin provides UVB protection, susceptibility to pigmentary responses from longer UVA wavelengths and visible light (VL) remains, particularly the highest energy wavelengths of blue light (BL) between 400 and 450 nm.1 Blue light can induce immediate and persistent pigment darkening in those with Fitzpatrick skin types IV to VI, and trace amounts of near-visible UVA (NV-UVA) between 370 and 400 nm can synergize with VL to amplify pigmentation and erythema responses.2
Current photoprotection recommendations emphasize sun protection factor (SPF) ratings of 30+ and broad-spectrum labeling; however, under the US Food and Drug Administration standards, the broad-spectrum designation is based solely on achieving a mean critical wavelength of 370 nm or higher, which does not ensure meaningful attenuation of NV-UVA or VL wavelengths.3 Tinted sunscreens containing iron oxides (FeO) have been shown to improve protection against these pigment-inducing wavelengths,4 yet quantitative comparisons between tinted and nontinted commercial sunscreen products remain limited.
To address the gap in understanding about tinted vs nontinted commercial sunscreen products, we conducted an in vitro spectrophotometric comparative analysis. The objectives were to quantify NV-UVA and BL attenuation across products and evaluate whether formulation characteristics (eg, SPF rating, filter types and concentration, the presence and depth of tint, antioxidant content) would correlate with improved photoprotection in pigment-sensitive wavelengths. We hypothesized that formulation features such as higher SPF, inorganic filters, and the presence of tint antioxidants would be associated with superior NV-UVA and BL attenuation compared with nontinted formulations.
Methods
Sunscreen Selection—A convenience sample of 23 broad-spectrum sunscreens commercially available at drugstores was selected to reflect easily accessible options. Six sunscreen brands with tinted (n=13) and nontinted (n=10) counterpart formulations were included. Filter category (mineral and/or chemical), SPF, UV filter type and concentration, tint shade (light, medium, medium/deep, deep), number of photoprotective antioxidants (diethylhexyl syringylidenemalonate, vitamin E, vitamin C, licochalcone A, and glycyrrhetinic acid), and presence of FeO were recorded.
Substrate Preparation—Testing was performed using standardized polymethyl methacrylate (PMMA) plates. Sunscreens were mixed prior to application and applied at 1.3 mg/cm² per the European Cosmetic and Perfumery Association (COLIPA) UVA testing guidelines.5 Plates were reweighed to confirm dosing and dried in a dark environment for at least 15 minutes prior to testing.
Spectrophotometric Measurements—Spectral transmittance was measured from 250 to 450 nm using a spectrophotometer equipped with a xenon flash lamp (energy <0.2 J/cm²). Baseline transmission was recorded using untreated PMMA plates. Five scans were averaged per plate. Analyses focused on NV-UVA transmittance from 380 to 400 nm and peak BL transmission at 450 nm.
Mean NV-UVA transmittance was calculated as the arithmetic mean of percent transmittance measured at 1-nm increments from 380 to 400 nm (n=21). Because of the steep rise in transmittance between 380 and 400 nm and subsequent plateau into the visible range, this approach was used to approximate the area under the transmittance-wavelength curve over the specified interval, enabling direct comparison of NV-UVA penetration between formulations.
Statistical Analysis—Descriptive statistics were used to summarize transmittance values. Spearman rank correlation was used to assess associations between formulation variables and spectral attenuation. Analysis of covariance was used to evaluate the effect of FeO on transmittance while adjusting for SPF or filter type. The Mann-Whitney U test was used to compare NV-UVA and blue light transmittance between FeO-containing mineral and chemical formulations. Statistical significance was set at P<.05.
Results
Across broad-spectrum sunscreen formulations (N=23), mean SPF values were 40.4 (range, 30-70), and the mean number of antioxidants in the ingredient list was 1.5 (range, 0-4). Mean NV-UVA transmittance was 16.7% (range, 0.1%-55.0%) and mean BL transmittance was 44.3% (range, 0.3%-97.5%)(eTable 1).

The mean labeled zinc oxide (ZnO) concentration among ZnO-containing formulations (n=14) was 10.5% (range, 5.0%-21.6%), with mean NV-UVA and BL transmittance of 12.6% (range, 0.1%-55.0%) and 25.8% (range, 0.3%-67.2%), respectively. Mean NV-UVA and BL transmittance were 26.7% (range, 9.6%-55.0%) and 45.6% (range, 23.0%-67.2%) among ZnO formulations without FeO (n=5), compared with lower transmittance of 4.8% (range, 0.1%-11.5%) and 14.9% (range, 0.3%-29.5%) in ZnO formulations containing FeO (n=9).
The mean labeled titanium dioxide (TiO2) concentration among TiO2-containing formulations (n=14) was 9.0% (range, 3.2%-17.0%), with corresponding mean NV-UVA and BL transmittance of 9.5% (range, 0.1%-28.5%) and 22.7% (range, 0.3%-47.6%), respectively. Among TiO2 formulations without FeO (n=4), mean NV-UVA and BL transmittance was 19.7% (range, 9.6%-28.5%) and 39.8% (range, 23.0%-47.6%), while FeO-containing TiO2 formulations (n=10) showed lower mean NV-UVA and BL transmittance of 5.4% (range, 0.1%-11.5%) and 15.8% (range, 0.3%-29.5%), respectively. The mean labeled avobenzone concentration among avobenzone-containing formulations (n=8) was 2.9% (range, 2.5%-3%), with mean NV-UVA and BL transmittance of 24.7% (range, 10.2%-46.6%) and 79.2% (range, 53.9%-97.5%). Formulations without FeO (n=5) had mean NV-UVA and BL transmittance of 29.0% (range, 10.2%-46.6%) and 83.2% (range, 61.1%-97.5%), whereas FeO-containing products (n=3) demonstrated lower mean NV-UVA and BL transmittance of 17.5% (range, 12.5%-21.9%) and 72.6% (range, 53.9%-85.1%), respectively.
Among products containing ZnO, TiO2, and avobenzone, the specific UV filter concentrations showed no statistically significant correlation with NV-UVA or BL transmittance (all P>.05). Iron oxide presence significantly correlated with lower NV-UVA (r=–0.67; P=.00042) and lower BL transmittance (r=–0.57; P=.0046). The number of antioxidants in the ingredient list did not correlate with NV-UVA transmittance (r=–0.28; P=.19) or BL transmittance (r=–0.16; P=.47). Sun protection factor was not significantly correlated with either wavelength range (Table 1).

Tint shade was treated as an ordinal variable (light, medium, medium/deep, and deep; medium was considered the universal shade). Increasing tint shade depth was significantly associated with reduced NV-UVA (r=–0.64; P=.045) and BL (r=–0.71; P=.023), suggesting a dose-response relationship wherein darker tints exhibited greater attenuation of pigment-relevant wavelengths. Among mineral filter formulations, tinted products demonstrated lower NV-UVA and BL transmittance compared with their nontinted counterparts, with deeper tints providing the greatest reduction in transmittance (eFigure 1). Similar results were observed for chemical filter formulations with greater attenuation in the NV-UVA and BL range for tinted versus nontinted products with greater variability across shade depths (eFigure 2).


After adjusting for SPF, FeO presence remained significantly associated with reduced NV-UVA (F[1,20]=26.9; P<.001) and BL transmittance (F[1,20]=11.7; P=.003). After adjusting for filter type (mineral vs chemical), FeO remained significantly associated with NV-UVA (F[1,19]=10.1; P=.004) and BL transmittance (F[1,19]=10.4; P=.005)(Table 2).

Among FeO-containing products, mineral filters demonstrated significantly lower NV-UVA transmittance compared with chemical filters (median, 5.58% [interquartile range (IQR), 0.59%-9.35%] vs 18.10% [IQR, 12.47%-21.90%]; U=0.00; P=.007). The same was true for BL transmittance (median, 15.90% [IQR, 5.00%-26.20%] vs 78.70% [IQR, 53.90%-85.10%]; U=0.00; P=.007). The differences in spectral transmittance between FeO-containing mineral and chemical filter formulations are illustrated in eFigure 3, with mineral-based products demonstrating lower transmittance, particularly across the upper NV-UVA range and across the BL range. These results indicated greater pigment-relevant photoprotection with mineral vs chemical filters (eTable 2).


Comment
Our initial hypothesis proposed that tinted sunscreens would provide greater NV-UVA and BL attenuation than nontinted formulations, and that characteristics such as inorganic filter content, SPF rating, and antioxidants would correlate with improved protection in pigment-sensitive wavelengths. Our findings partially supported this hypothesis. In this analysis, substantial variability in the NV-UVA and BL transmittance was observed despite all products meeting broad-spectrum criteria. Nontinted mineral and chemical sunscreens exhibited high transmittance in these pigment-related wavelengths, reaching values as high as 55.0% for NV-UVA and 97.5% for BL. These findings align with prior analysis demonstrating that while broad-spectrum sunscreens available in the United States may meet the current critical wavelength criteria for protection in the UVA range, they still may transmit 30% to 66% of available UVA over 2 hours between formulations with equivalent SPF label values.6
Recent analyses show that sunscreen recommendations in lay media rarely incorporate input from board-certified dermatologists for individuals with SOC and disproportionately favor nontinted chemical formulations, despite the high prevalence of pigmentary disorders in this population.7 Near-visible UVA and BL have been demonstrated to be biologically relevant pigment-inducing wavelengths, both in vitro and in vivo, particularly in individuals with SOC, yet broad-spectrum labeling does not ensure protection against these spectra.8 Pigmentary tints such as FeO have demonstrated enhanced attenuation in this spectral region in vivo and may provide more reliable coverage than products with broad-spectrum designation alone.4,9 Treatment options for pigmentary disorders such as melasma tend to be palliative and costly, making optimized photoprotection a critical component of care to reduce ongoing pigmentary stimuli.10
Formulations containing FeO demonstrated significantly lower NV-UVA (P<.001) and BL transmittance (P=.003) on average; however, transmittance values ranged widely (NV-UVA: 0.10%-21.90%, BL: 0.30%-85.10%), indicating that FeO presence alone does not determine the magnitude of attenuation. Notably, among FeO-containing products, mineral filters provided significantly lower NV-UVA and BL transmittance compared with chemical filters (P=.007 for both), suggesting that filter type further modulates pigment-relevant photoprotection. Tinted formulations may improve compliance with product use by reducing the white cast and improve shade matching to find suitable options for deeper skin tones,11 but the highly variable photoprotection offered raises concerns about clinical benefit. Although deeper tints showed greater attenuation, pigment concentrations and combinations are not disclosed by manufacturers as FeO is not considered an active ingredient. Darker shades are not practical across all skin tones in individuals with SOC, which underscores the need for standardized pigment metrics and shade-inclusive options.
While avobenzone and ZnO are the only US Food and Drug Administration–approved sunscreen active ingredients that extend protection beyond 360 nm,12 both exhibited reduced attenuation beyond the longer end of the UVA spectrum. Formulation characteristics, including the concentration of ZnO, TiO2, and/or avobenzone as well as SPF, did not correlate with NV-UVA or BL attenuation. In the adjusted analysis, FeO presence remained significantly associated with reduced transmittance after adjusting for SPF (NV-UVA: P<.001, BL: P=.003) or filter type (NV-UVA: P=.004, BL: P=.005). These findings suggest that the presence of FeO, rather than UV filters or SPF ratings, supports attenuation in the 380 to 450–nm range, indicating a functional benefit in addition to improved cosmesis.13
Although antioxidants in specific combinations have shown promise in vivo, no association was observed between the number of antioxidants present and NV-UVA or BL attenuation compared with added tint.14 This suggests that specific antioxidant combinations and their concentrations may be more relevant than the total count.
Several study limitations need to be considered in interpreting our results, including a modest number of products, controlled in vitro testing conditions, and an incomplete representation of products with pigment concentrations and shade ranges marketed to individuals with SOC across all price categories, despite our focus on affordable, commercially available options. Moreover, PMMA-based spectrophotometry does not account for skin surface heterogeneity, photodegradation, sweat, oil, friction, or application variability, which may alter real-world performance. Additionally, FeO concentrations could not be quantified beyond labeling of tint shade depth, preventing a true assessment of dose-response effects. These limitations may reduce generalizability and highlight the need for complementary in vivo studies to assess clinically relevant outcomes such as persistent pigment darkening. For this reason, caution is warranted in extrapolating these spectral findings to clinical efficacy.
Conclusion
Given the susceptibility of individuals with SOC to pigmentary disorders driven by NV-UVA and BL, our findings support further development and study of FeO-containing sunscreens that address clinically relevant wavelengths. Wide variability in photo-attenuation among tinted formulations underscores the need for evidence-based recommendations, with further studies needed to guide photoprotection strategies for populations with SOC.
- Marionnet C, Piffaut V, Sasai J, et al. A precise analysis of the relative contribution of UVA1 and visible light colour domains in solar light-induced skin pigmentation. J Eur Acad Dermatol Venereol. 2023;37(suppl 4):3-11. doi:10.1111/jdv.18948
- Kohli I, Chaowattanapanit S, Mohammad TF, et al. Synergistic effects of long-wavelength ultraviolet A1 and visible light on pigmentation and erythema. Br J Dermatol. 2018;178:1173-1180. doi: 10.1111/bjd.15940
- US Food and Drug Administration. Over-the-counter monograph M020: sunscreen drug products for over-the-counter human use. September 24, 2021. Accessed April 7, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/omuf/monographs/OTCMonograph_M020-SunscreenDrugProductsforOTCHumanUse09242021.pdf
- Grimes PE, Paturi J, Chen Y, et al. Photoprotection efficacy of sun protection factor and iron oxide formulations in diverse skin with melasma and photodamage. J Drugs Dermatol. 2025;24:662-667. doi:10.36849/JDD.9240
- Moyal D, Alard V, Bertin C, et al. The revised COLIPA in vitro UVA method. Int J Cosmet Sci. 2013;35:35-40. doi:10.1111/j.1468-2494.2012.00748.x
- Coelho SG, Rua D, Miller SA, et al. Suboptimal UVA attenuation by broad spectrum sunscreens under outdoor solar conditions contributes to lifetime UVA burden. Photodermatol Photoimmunol Photomed. 2020;36:42-52. doi:10.1111/phpp.12503
- Song H, Beckles A, Salian P, et al. Sunscreen recommendations for patients with skin of color in the popular press and in the dermatology clinic. Int J Womens Dermatol. 2020;7:165-170. doi:10.1016/j.ijwd.2020.10.008
- Lawrence KP, Douki T, Sarkany RPE, et al. The UV/visible radiation boundary region (385-405 nm) damages skin cells and induces “dark” cyclobutane pyrimidine dimers in human skin in vivo. Sci Rep. 2018;8:12722. doi:10.1038/s41598-018-30738-6
- Ezekwe N, Pourang A, Lyons AB, et al. Evaluation of the protection of sunscreen products against long wavelength ultraviolet A1 and visible light-induced biological effects. Photodermatol Photoimmunol Photomed. 2024;40:E12937. doi:10.1111/phpp.12937
- Mpofana N, Chibi B, Gqaleni N, et al. Melasma in people with darker skin types: a scoping review protocol on prevalence, treatment options for melasma and impact on quality of life. Syst Rev. 2023;12:139. doi:10.1186/s13643-023-02300-7
- Wang JY, Patel P, Philip R, et al. Sunscreen practices and preferences of skin of color patients. J Drugs Dermatol. 2024;23:456-462. doi:10.36849/JDD.8268
- Beasley DG, Meyer TA. Characterization of the UVA protection provided by avobenzone, zinc oxide, and titanium dioxide in broad-spectrum sunscreen products. Am J Clin Dermatol. 2010;11:413-421. doi:10.2165/11537050-000000000-00000
- Morgado-Carrasco D, Delgado J, Prudkin-Silva Let al. Sunscreens prescribed to patients with skin of color and/or with melasma: a survey of 221 dermatologists and dermatology residents in Spain. Photodermatol Photoimmunol Photomed. 2024;40:E12996. doi:10.1111/phpp.12996
- Ruvolo E, Boothby-Shoemaker W, Kumar N, et al. Evaluation of efficacy of antioxidant-enriched sunscreen prodcuts against long wavelength ultraviolet A1 and visible light. Int J Cosmet Sci. 2022;44:394-402. doi:10.1111/ics.12785
- Marionnet C, Piffaut V, Sasai J, et al. A precise analysis of the relative contribution of UVA1 and visible light colour domains in solar light-induced skin pigmentation. J Eur Acad Dermatol Venereol. 2023;37(suppl 4):3-11. doi:10.1111/jdv.18948
- Kohli I, Chaowattanapanit S, Mohammad TF, et al. Synergistic effects of long-wavelength ultraviolet A1 and visible light on pigmentation and erythema. Br J Dermatol. 2018;178:1173-1180. doi: 10.1111/bjd.15940
- US Food and Drug Administration. Over-the-counter monograph M020: sunscreen drug products for over-the-counter human use. September 24, 2021. Accessed April 7, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/omuf/monographs/OTCMonograph_M020-SunscreenDrugProductsforOTCHumanUse09242021.pdf
- Grimes PE, Paturi J, Chen Y, et al. Photoprotection efficacy of sun protection factor and iron oxide formulations in diverse skin with melasma and photodamage. J Drugs Dermatol. 2025;24:662-667. doi:10.36849/JDD.9240
- Moyal D, Alard V, Bertin C, et al. The revised COLIPA in vitro UVA method. Int J Cosmet Sci. 2013;35:35-40. doi:10.1111/j.1468-2494.2012.00748.x
- Coelho SG, Rua D, Miller SA, et al. Suboptimal UVA attenuation by broad spectrum sunscreens under outdoor solar conditions contributes to lifetime UVA burden. Photodermatol Photoimmunol Photomed. 2020;36:42-52. doi:10.1111/phpp.12503
- Song H, Beckles A, Salian P, et al. Sunscreen recommendations for patients with skin of color in the popular press and in the dermatology clinic. Int J Womens Dermatol. 2020;7:165-170. doi:10.1016/j.ijwd.2020.10.008
- Lawrence KP, Douki T, Sarkany RPE, et al. The UV/visible radiation boundary region (385-405 nm) damages skin cells and induces “dark” cyclobutane pyrimidine dimers in human skin in vivo. Sci Rep. 2018;8:12722. doi:10.1038/s41598-018-30738-6
- Ezekwe N, Pourang A, Lyons AB, et al. Evaluation of the protection of sunscreen products against long wavelength ultraviolet A1 and visible light-induced biological effects. Photodermatol Photoimmunol Photomed. 2024;40:E12937. doi:10.1111/phpp.12937
- Mpofana N, Chibi B, Gqaleni N, et al. Melasma in people with darker skin types: a scoping review protocol on prevalence, treatment options for melasma and impact on quality of life. Syst Rev. 2023;12:139. doi:10.1186/s13643-023-02300-7
- Wang JY, Patel P, Philip R, et al. Sunscreen practices and preferences of skin of color patients. J Drugs Dermatol. 2024;23:456-462. doi:10.36849/JDD.8268
- Beasley DG, Meyer TA. Characterization of the UVA protection provided by avobenzone, zinc oxide, and titanium dioxide in broad-spectrum sunscreen products. Am J Clin Dermatol. 2010;11:413-421. doi:10.2165/11537050-000000000-00000
- Morgado-Carrasco D, Delgado J, Prudkin-Silva Let al. Sunscreens prescribed to patients with skin of color and/or with melasma: a survey of 221 dermatologists and dermatology residents in Spain. Photodermatol Photoimmunol Photomed. 2024;40:E12996. doi:10.1111/phpp.12996
- Ruvolo E, Boothby-Shoemaker W, Kumar N, et al. Evaluation of efficacy of antioxidant-enriched sunscreen prodcuts against long wavelength ultraviolet A1 and visible light. Int J Cosmet Sci. 2022;44:394-402. doi:10.1111/ics.12785
Tinted vs Nontinted Sunscreens: Attenuation of Near-Visible UVA and Visible Light and Implications for Skin of Color
Tinted vs Nontinted Sunscreens: Attenuation of Near-Visible UVA and Visible Light and Implications for Skin of Color
PRACTICE POINTS
- There is substantial variability in spectral attenuation among tinted sunscreens, highlighting the need for standardized pigment metrics and evidence-based photoprotection guidance for individuals with skin of color (SOC).
- Broad-spectrum labeling, sun protection factor values, UV filter type (mineral vs chemical), and antioxidant inclusion do not reliably predict protection against pigment-inducing UV and visible light wavelengths for individuals with SOC.
- Iron oxide–containing tinted sunscreens demonstrate lower near-visible UVA and blue light transmittance than nontinted formulations, with greater attenuation in mineral vs chemical products. Both pigment and filter type should inform photoprotection recommendations in individuals with SOC.
Cutaneous Reactions to Triatomine (Kissing Bug) Bites and the Risk for Chagas Disease
Cutaneous Reactions to Triatomine (Kissing Bug) Bites and the Risk for Chagas Disease
Triatome bugs cause painful bites and serve as vectors for Chagas disease. In this article, we will address diagnosis and vector identification.
Key Morphologic Features
Insects from the subfamily Triatominae are identifiable by their long legs and a shieldlike abdomen behind a platelike pronotum that covers the thorax. Their half-membranous wings overlap, covering the central abdomen but leaving the lateral portions visible. Tigerlike stripes are characteristically prominent on the visible portions of the lateral abdomen. The stalklike head has an articulated beaklike mouth that can be retracted and used to deliver a powerful bite (Figure 1).

Feeding Mechanisms and Host Reactions
Triatome bugs are blood-feeding arthropods that hide in cracks and crevices in domestic structures by day and feed at night. They are shy feeders, and laboratory colonies have been known to die rather than feed in daylight. They are particularly common in thatched or wattle-and-daub dwellings, where they can be present in great numbers and descend on sleeping inhabitants at night. Triatome bugs require regular blood meals throughout the 5 developmental nymph stages in order to undergo successful molting.
In the wild, triatome bugs feed on a range of animals with little specificity, but in domestic settings they feed largely on humans. Thermosensors in the antennae help them locate blood vessels under the skin, which they penetrate easily due to their long mouthparts. Like other blood-sucking arthropods, they release an anticoagulant that facilitates continuous blood flow while feeding, which accounts for many of the cutaneous reactions observed after the host sustains a triatomine bite.1
Triatomine bugs have trouble feeding through clothing and seek out exposed skin, particularly the eyelids, producing the characteristic unilateral eyelid swelling known as the Romaña sign. Other bite reactions include purpura; macular erythema; and vesiculobullous, papular, and urticarial lesions (Figure 2).2 Associated lymphangitis or lymphadenopathy may be noted, and anaphylaxis has been reported. Similar to those of cockroaches, triatome antigens have been associated with atopic dermatitis and asthma.3
Chagas Disease Risk and Transmission
Triatomine reduviids are the primary vector of Chagas disease, and the geographic range of both continues to expand, particularly in North America. The disease remains endemic in Latin America, with the highest incidence now reported in Brazil.4 An estimated 240,000 to 350,000 individuals in the United States are infected, primarily immigrants from Mexico, Central America, and South America; approximately 30% of those infected will develop cardiac and/or gastrointestinal complications.4 If left untreated, Chagas disease leads to autonomic ganglion destruction and subsequent gastrointestinal and cardiac complications, including megacolon, dilated cardiomyopathy, and heart failure.5
Trypanosoma cruzi, the microorganism responsible for Chagas disease, is spread to humans through triatomine fecal matter scratched into the bite wound.6 Triatomine bugs have a highly developed gastrocolic reflex and defecate liberally as they feed. Fecal volume is heavily dependent on species and sex, with fifth-stage female nymphs producing the highest volume of excrement and thereby acting as particularly adept disease vectors.6 Triatoma infestans and members of the genus Mepraia are key vectors of T cruzi.1 In areas of South America where populations of T infestans are controlled through public health measures, Mepraia emerge as a largely uncontrolled disease vector.1,7 While endemic to the southern United States and South America, T cruzi has spread to much of North America and Europe by way of Triatominae as naturalized or invasive species.8
There are 3 phases of Chagas disease: acute, indeterminate, and chronic. A chagoma is a localized erythematous swelling at the site of the bite. The acute phase often lacks systemic symptoms but may include fever, myalgia, and headache. The intermediate phase may include fatigue and recurrent fevers. The most serious manifestations occur in the chronic phase and include cardiomyopathy with signs of congestive heart failure, irregular heartbeat, cardiac arrest, abdominal pain, constipation, and dysphagia.
Deforestation has been identified as a driving factor in the spread of Chagas disease, as the disease vectors shift from wilderness areas and animal hosts to inhabited areas where humans are the most readily available food source. Triatome bugs in areas experiencing higher levels of development or forest harvesting are forced into human-populated areas. As a result, instances of Chagas disease are on the rise in these communities.7 Salvador, Bahia, Brazil, has been identified as one such target of increased vector presence due to heavy deforestation, and the hottest months were identified as having the greatest threat of vector exposure.9 Brazil became the leading geographic area for the disease partly because of heavy loss of forested land.10
Vector Control and Prevention Strategies
Elimination of cracks and crevices in walls; replacement of wattle and daub with stucco, plaster, and other solid building materials; and the use of insecticides with durability in the environment have been used to reduce triatome bug infestation in homes. However, highly persistent insecticides carry greater environmental risk and may drive resistance as declining concentrations select for resistant arthropods. Repellents have less environmental impact and play an important role in vector control. Citronella essential oil has been observed to repel several species of triatome bugs that are common in Arizona; specifically, the component alcohols geraniol and citronellol were found to be effective at inhibiting triatome feeding.11
Early detection of Chagas disease is essential, as end-stage cardiomyopathy and megacolon are difficult to treat. Newly developed multiantigen testing has shown promising results, suggesting a potential for more accurate testing for Chagas disease.8 Geospatial tracking and mapping of T cruzi vectors now are employed to track seasonal vector changes and disease patterns.9 Researchers also have developed a dedicated dichotomous key for the identification of triatome bugs endemic in Brazil with the hope of better identification and mapping of disease vector presence and density.10 The key consists of a series of statements with 2 choices in each step. It uses observable features of the arthropod to lead users to the correct identification.
Final Thoughts
Identification of triatome bugs can help with public health efforts to control the spread of disease. Patients with unilateral eyelid swelling should be evaluated for possible bedbug or triatome exposure.
- Egaña C, Pinto R, Vergara F, et al. Fluctuations in Trypanosoma cruzi discrete typing unit composition in two naturally infected triatomines: Mepraia gajardoi and M. spinolai after laboratory feeding. Acta Trop. 2016;160:9-14. Erratum in: Acta Trop. 2016;162:248. doi:10.1016/j.actatropica.2016.04.008
- Moffitt JE, Venarske D, Goddard J, et al. Allergic reactions to Triatoma bites. Ann Allergy Asthma Immunol. 2003;91:122-128.
- Alonso A, Potenza M, Mouchián K, et al. Proteinase and gelatinolytic properties of a Triatoma infestans extract. Allergol Immunopathol (Madr). 2004;32:223-227.
- Hochberg NS, Montgomery SP. Chagas disease. Ann Intern Med. 2023;176:ITC17-ITC32. doi:10.7326/AITC202302210
- Pless M, Juranek D, Kozarsky P, et al. The epidemiology of Chagas’ disease in a hyperendemic area of Cochabamba, Bolivia: a clinical study including electrocardiography, seroreactivity to Trypanosoma cruzi, xenodiagnosis, and domiciliary triatomine distribution. Am J Trop Med Hyg. 1992;47:539-546.
- Piesman J, Sherlock IA. Factors controlling the volume of feces produced by triatomine vectors of Chagas’ disease. Acta Trop. 1983;40:351-358.
- Steverding D. The history of Chagas disease. Parasit Vectors. 2014;10:317.
- Granjon E, Dichtel-Danjoy ML, Saba E, et al. Development of a novel multiplex immunoassay multi-cruzi for the serological confirmation of Chagas disease. PLoS Negl Trop Dis. 2016;10:e0004596.
- Santana Kde S, Bavia ME, Lima AD, et al. Spatial distribution of triatomines (Reduviidae: Triatominae) in urban areas of the city of Salvador, Bahia, Brazil. Geospat Health. 2011;5:199-203.
- de Mello DV, Nhapulo EF, Cesaretto LP, et al. Dichotomous keys based on cytogenetic data for triatomines reported in Brazilian regions with outbreaks of orally transmitted Chagas disease (Pernambuco and Rio Grande Do Norte). Trop Med Infect Dis. 2023;8:196.
- Zamora D, Klotz SA, Meister EA, et al. Repellency of the components of the essential oil, citronella, to Triatoma rubida, Triatoma protracta, and Triatoma recurva (Hemiptera: Reduviidae: Triatominae). J Med Entomol. 2015;52:719-721.
Triatome bugs cause painful bites and serve as vectors for Chagas disease. In this article, we will address diagnosis and vector identification.
Key Morphologic Features
Insects from the subfamily Triatominae are identifiable by their long legs and a shieldlike abdomen behind a platelike pronotum that covers the thorax. Their half-membranous wings overlap, covering the central abdomen but leaving the lateral portions visible. Tigerlike stripes are characteristically prominent on the visible portions of the lateral abdomen. The stalklike head has an articulated beaklike mouth that can be retracted and used to deliver a powerful bite (Figure 1).

Feeding Mechanisms and Host Reactions
Triatome bugs are blood-feeding arthropods that hide in cracks and crevices in domestic structures by day and feed at night. They are shy feeders, and laboratory colonies have been known to die rather than feed in daylight. They are particularly common in thatched or wattle-and-daub dwellings, where they can be present in great numbers and descend on sleeping inhabitants at night. Triatome bugs require regular blood meals throughout the 5 developmental nymph stages in order to undergo successful molting.
In the wild, triatome bugs feed on a range of animals with little specificity, but in domestic settings they feed largely on humans. Thermosensors in the antennae help them locate blood vessels under the skin, which they penetrate easily due to their long mouthparts. Like other blood-sucking arthropods, they release an anticoagulant that facilitates continuous blood flow while feeding, which accounts for many of the cutaneous reactions observed after the host sustains a triatomine bite.1
Triatomine bugs have trouble feeding through clothing and seek out exposed skin, particularly the eyelids, producing the characteristic unilateral eyelid swelling known as the Romaña sign. Other bite reactions include purpura; macular erythema; and vesiculobullous, papular, and urticarial lesions (Figure 2).2 Associated lymphangitis or lymphadenopathy may be noted, and anaphylaxis has been reported. Similar to those of cockroaches, triatome antigens have been associated with atopic dermatitis and asthma.3

Chagas Disease Risk and Transmission
Triatomine reduviids are the primary vector of Chagas disease, and the geographic range of both continues to expand, particularly in North America. The disease remains endemic in Latin America, with the highest incidence now reported in Brazil.4 An estimated 240,000 to 350,000 individuals in the United States are infected, primarily immigrants from Mexico, Central America, and South America; approximately 30% of those infected will develop cardiac and/or gastrointestinal complications.4 If left untreated, Chagas disease leads to autonomic ganglion destruction and subsequent gastrointestinal and cardiac complications, including megacolon, dilated cardiomyopathy, and heart failure.5
Trypanosoma cruzi, the microorganism responsible for Chagas disease, is spread to humans through triatomine fecal matter scratched into the bite wound.6 Triatomine bugs have a highly developed gastrocolic reflex and defecate liberally as they feed. Fecal volume is heavily dependent on species and sex, with fifth-stage female nymphs producing the highest volume of excrement and thereby acting as particularly adept disease vectors.6 Triatoma infestans and members of the genus Mepraia are key vectors of T cruzi.1 In areas of South America where populations of T infestans are controlled through public health measures, Mepraia emerge as a largely uncontrolled disease vector.1,7 While endemic to the southern United States and South America, T cruzi has spread to much of North America and Europe by way of Triatominae as naturalized or invasive species.8
There are 3 phases of Chagas disease: acute, indeterminate, and chronic. A chagoma is a localized erythematous swelling at the site of the bite. The acute phase often lacks systemic symptoms but may include fever, myalgia, and headache. The intermediate phase may include fatigue and recurrent fevers. The most serious manifestations occur in the chronic phase and include cardiomyopathy with signs of congestive heart failure, irregular heartbeat, cardiac arrest, abdominal pain, constipation, and dysphagia.
Deforestation has been identified as a driving factor in the spread of Chagas disease, as the disease vectors shift from wilderness areas and animal hosts to inhabited areas where humans are the most readily available food source. Triatome bugs in areas experiencing higher levels of development or forest harvesting are forced into human-populated areas. As a result, instances of Chagas disease are on the rise in these communities.7 Salvador, Bahia, Brazil, has been identified as one such target of increased vector presence due to heavy deforestation, and the hottest months were identified as having the greatest threat of vector exposure.9 Brazil became the leading geographic area for the disease partly because of heavy loss of forested land.10
Vector Control and Prevention Strategies
Elimination of cracks and crevices in walls; replacement of wattle and daub with stucco, plaster, and other solid building materials; and the use of insecticides with durability in the environment have been used to reduce triatome bug infestation in homes. However, highly persistent insecticides carry greater environmental risk and may drive resistance as declining concentrations select for resistant arthropods. Repellents have less environmental impact and play an important role in vector control. Citronella essential oil has been observed to repel several species of triatome bugs that are common in Arizona; specifically, the component alcohols geraniol and citronellol were found to be effective at inhibiting triatome feeding.11
Early detection of Chagas disease is essential, as end-stage cardiomyopathy and megacolon are difficult to treat. Newly developed multiantigen testing has shown promising results, suggesting a potential for more accurate testing for Chagas disease.8 Geospatial tracking and mapping of T cruzi vectors now are employed to track seasonal vector changes and disease patterns.9 Researchers also have developed a dedicated dichotomous key for the identification of triatome bugs endemic in Brazil with the hope of better identification and mapping of disease vector presence and density.10 The key consists of a series of statements with 2 choices in each step. It uses observable features of the arthropod to lead users to the correct identification.
Final Thoughts
Identification of triatome bugs can help with public health efforts to control the spread of disease. Patients with unilateral eyelid swelling should be evaluated for possible bedbug or triatome exposure.
Triatome bugs cause painful bites and serve as vectors for Chagas disease. In this article, we will address diagnosis and vector identification.
Key Morphologic Features
Insects from the subfamily Triatominae are identifiable by their long legs and a shieldlike abdomen behind a platelike pronotum that covers the thorax. Their half-membranous wings overlap, covering the central abdomen but leaving the lateral portions visible. Tigerlike stripes are characteristically prominent on the visible portions of the lateral abdomen. The stalklike head has an articulated beaklike mouth that can be retracted and used to deliver a powerful bite (Figure 1).

Feeding Mechanisms and Host Reactions
Triatome bugs are blood-feeding arthropods that hide in cracks and crevices in domestic structures by day and feed at night. They are shy feeders, and laboratory colonies have been known to die rather than feed in daylight. They are particularly common in thatched or wattle-and-daub dwellings, where they can be present in great numbers and descend on sleeping inhabitants at night. Triatome bugs require regular blood meals throughout the 5 developmental nymph stages in order to undergo successful molting.
In the wild, triatome bugs feed on a range of animals with little specificity, but in domestic settings they feed largely on humans. Thermosensors in the antennae help them locate blood vessels under the skin, which they penetrate easily due to their long mouthparts. Like other blood-sucking arthropods, they release an anticoagulant that facilitates continuous blood flow while feeding, which accounts for many of the cutaneous reactions observed after the host sustains a triatomine bite.1
Triatomine bugs have trouble feeding through clothing and seek out exposed skin, particularly the eyelids, producing the characteristic unilateral eyelid swelling known as the Romaña sign. Other bite reactions include purpura; macular erythema; and vesiculobullous, papular, and urticarial lesions (Figure 2).2 Associated lymphangitis or lymphadenopathy may be noted, and anaphylaxis has been reported. Similar to those of cockroaches, triatome antigens have been associated with atopic dermatitis and asthma.3

Chagas Disease Risk and Transmission
Triatomine reduviids are the primary vector of Chagas disease, and the geographic range of both continues to expand, particularly in North America. The disease remains endemic in Latin America, with the highest incidence now reported in Brazil.4 An estimated 240,000 to 350,000 individuals in the United States are infected, primarily immigrants from Mexico, Central America, and South America; approximately 30% of those infected will develop cardiac and/or gastrointestinal complications.4 If left untreated, Chagas disease leads to autonomic ganglion destruction and subsequent gastrointestinal and cardiac complications, including megacolon, dilated cardiomyopathy, and heart failure.5
Trypanosoma cruzi, the microorganism responsible for Chagas disease, is spread to humans through triatomine fecal matter scratched into the bite wound.6 Triatomine bugs have a highly developed gastrocolic reflex and defecate liberally as they feed. Fecal volume is heavily dependent on species and sex, with fifth-stage female nymphs producing the highest volume of excrement and thereby acting as particularly adept disease vectors.6 Triatoma infestans and members of the genus Mepraia are key vectors of T cruzi.1 In areas of South America where populations of T infestans are controlled through public health measures, Mepraia emerge as a largely uncontrolled disease vector.1,7 While endemic to the southern United States and South America, T cruzi has spread to much of North America and Europe by way of Triatominae as naturalized or invasive species.8
There are 3 phases of Chagas disease: acute, indeterminate, and chronic. A chagoma is a localized erythematous swelling at the site of the bite. The acute phase often lacks systemic symptoms but may include fever, myalgia, and headache. The intermediate phase may include fatigue and recurrent fevers. The most serious manifestations occur in the chronic phase and include cardiomyopathy with signs of congestive heart failure, irregular heartbeat, cardiac arrest, abdominal pain, constipation, and dysphagia.
Deforestation has been identified as a driving factor in the spread of Chagas disease, as the disease vectors shift from wilderness areas and animal hosts to inhabited areas where humans are the most readily available food source. Triatome bugs in areas experiencing higher levels of development or forest harvesting are forced into human-populated areas. As a result, instances of Chagas disease are on the rise in these communities.7 Salvador, Bahia, Brazil, has been identified as one such target of increased vector presence due to heavy deforestation, and the hottest months were identified as having the greatest threat of vector exposure.9 Brazil became the leading geographic area for the disease partly because of heavy loss of forested land.10
Vector Control and Prevention Strategies
Elimination of cracks and crevices in walls; replacement of wattle and daub with stucco, plaster, and other solid building materials; and the use of insecticides with durability in the environment have been used to reduce triatome bug infestation in homes. However, highly persistent insecticides carry greater environmental risk and may drive resistance as declining concentrations select for resistant arthropods. Repellents have less environmental impact and play an important role in vector control. Citronella essential oil has been observed to repel several species of triatome bugs that are common in Arizona; specifically, the component alcohols geraniol and citronellol were found to be effective at inhibiting triatome feeding.11
Early detection of Chagas disease is essential, as end-stage cardiomyopathy and megacolon are difficult to treat. Newly developed multiantigen testing has shown promising results, suggesting a potential for more accurate testing for Chagas disease.8 Geospatial tracking and mapping of T cruzi vectors now are employed to track seasonal vector changes and disease patterns.9 Researchers also have developed a dedicated dichotomous key for the identification of triatome bugs endemic in Brazil with the hope of better identification and mapping of disease vector presence and density.10 The key consists of a series of statements with 2 choices in each step. It uses observable features of the arthropod to lead users to the correct identification.
Final Thoughts
Identification of triatome bugs can help with public health efforts to control the spread of disease. Patients with unilateral eyelid swelling should be evaluated for possible bedbug or triatome exposure.
- Egaña C, Pinto R, Vergara F, et al. Fluctuations in Trypanosoma cruzi discrete typing unit composition in two naturally infected triatomines: Mepraia gajardoi and M. spinolai after laboratory feeding. Acta Trop. 2016;160:9-14. Erratum in: Acta Trop. 2016;162:248. doi:10.1016/j.actatropica.2016.04.008
- Moffitt JE, Venarske D, Goddard J, et al. Allergic reactions to Triatoma bites. Ann Allergy Asthma Immunol. 2003;91:122-128.
- Alonso A, Potenza M, Mouchián K, et al. Proteinase and gelatinolytic properties of a Triatoma infestans extract. Allergol Immunopathol (Madr). 2004;32:223-227.
- Hochberg NS, Montgomery SP. Chagas disease. Ann Intern Med. 2023;176:ITC17-ITC32. doi:10.7326/AITC202302210
- Pless M, Juranek D, Kozarsky P, et al. The epidemiology of Chagas’ disease in a hyperendemic area of Cochabamba, Bolivia: a clinical study including electrocardiography, seroreactivity to Trypanosoma cruzi, xenodiagnosis, and domiciliary triatomine distribution. Am J Trop Med Hyg. 1992;47:539-546.
- Piesman J, Sherlock IA. Factors controlling the volume of feces produced by triatomine vectors of Chagas’ disease. Acta Trop. 1983;40:351-358.
- Steverding D. The history of Chagas disease. Parasit Vectors. 2014;10:317.
- Granjon E, Dichtel-Danjoy ML, Saba E, et al. Development of a novel multiplex immunoassay multi-cruzi for the serological confirmation of Chagas disease. PLoS Negl Trop Dis. 2016;10:e0004596.
- Santana Kde S, Bavia ME, Lima AD, et al. Spatial distribution of triatomines (Reduviidae: Triatominae) in urban areas of the city of Salvador, Bahia, Brazil. Geospat Health. 2011;5:199-203.
- de Mello DV, Nhapulo EF, Cesaretto LP, et al. Dichotomous keys based on cytogenetic data for triatomines reported in Brazilian regions with outbreaks of orally transmitted Chagas disease (Pernambuco and Rio Grande Do Norte). Trop Med Infect Dis. 2023;8:196.
- Zamora D, Klotz SA, Meister EA, et al. Repellency of the components of the essential oil, citronella, to Triatoma rubida, Triatoma protracta, and Triatoma recurva (Hemiptera: Reduviidae: Triatominae). J Med Entomol. 2015;52:719-721.
- Egaña C, Pinto R, Vergara F, et al. Fluctuations in Trypanosoma cruzi discrete typing unit composition in two naturally infected triatomines: Mepraia gajardoi and M. spinolai after laboratory feeding. Acta Trop. 2016;160:9-14. Erratum in: Acta Trop. 2016;162:248. doi:10.1016/j.actatropica.2016.04.008
- Moffitt JE, Venarske D, Goddard J, et al. Allergic reactions to Triatoma bites. Ann Allergy Asthma Immunol. 2003;91:122-128.
- Alonso A, Potenza M, Mouchián K, et al. Proteinase and gelatinolytic properties of a Triatoma infestans extract. Allergol Immunopathol (Madr). 2004;32:223-227.
- Hochberg NS, Montgomery SP. Chagas disease. Ann Intern Med. 2023;176:ITC17-ITC32. doi:10.7326/AITC202302210
- Pless M, Juranek D, Kozarsky P, et al. The epidemiology of Chagas’ disease in a hyperendemic area of Cochabamba, Bolivia: a clinical study including electrocardiography, seroreactivity to Trypanosoma cruzi, xenodiagnosis, and domiciliary triatomine distribution. Am J Trop Med Hyg. 1992;47:539-546.
- Piesman J, Sherlock IA. Factors controlling the volume of feces produced by triatomine vectors of Chagas’ disease. Acta Trop. 1983;40:351-358.
- Steverding D. The history of Chagas disease. Parasit Vectors. 2014;10:317.
- Granjon E, Dichtel-Danjoy ML, Saba E, et al. Development of a novel multiplex immunoassay multi-cruzi for the serological confirmation of Chagas disease. PLoS Negl Trop Dis. 2016;10:e0004596.
- Santana Kde S, Bavia ME, Lima AD, et al. Spatial distribution of triatomines (Reduviidae: Triatominae) in urban areas of the city of Salvador, Bahia, Brazil. Geospat Health. 2011;5:199-203.
- de Mello DV, Nhapulo EF, Cesaretto LP, et al. Dichotomous keys based on cytogenetic data for triatomines reported in Brazilian regions with outbreaks of orally transmitted Chagas disease (Pernambuco and Rio Grande Do Norte). Trop Med Infect Dis. 2023;8:196.
- Zamora D, Klotz SA, Meister EA, et al. Repellency of the components of the essential oil, citronella, to Triatoma rubida, Triatoma protracta, and Triatoma recurva (Hemiptera: Reduviidae: Triatominae). J Med Entomol. 2015;52:719-721.
Cutaneous Reactions to Triatomine (Kissing Bug) Bites and the Risk for Chagas Disease
Cutaneous Reactions to Triatomine (Kissing Bug) Bites and the Risk for Chagas Disease
Practice Points
- Triatomine bugs, commonly known as kissing bugs, are widespread, especially in warmer climates, and their geographic range is expanding.
- The Romaña sign, characterized by unilateral swelling of the eyelid, is common in triatomine bites.
- Triatomine bugs are the primary vector for transmission of the parasite Trypanosoma cruzi, the causative agent of Chagas disease.
- In recent years, T cruzi has been detected in triatomine reduviids in suburban areas of the southwestern United States.
Alignment of ChatGPT Responses With AAD Guidelines for Cutaneous Melanoma
Alignment of ChatGPT Responses With AAD Guidelines for Cutaneous Melanoma
To the Editor:
ChatGPT (OpenAI), a popular large language model that generates responses to user queries, has attracted substantial attention as a potential resource for patient education.1 While prior studies have shown that ChatGPT can provide reliable and general patient information, its alignment with the American Academy of Dermatology’s (AAD’s) guidelines for primary cutaneous melanoma (CM) compared to evidence in the recent literature has not been evaluated.2,3 In this study, we compared ChatGPT’s responses to the 25 evidence-based questions utilized by the AAD to establish its 2019 recommendations for primary CM. Because the 2019 AAD guidelines included literature only through April 2017, we conducted an additional search (May 2017–February 2024) to assess ChatGPT’s alignment with more recent evidence not captured in the guidelines.
On April 17, 2024, 2 authors (D.P. and A.F.) prompted ChatGPT with 25 evidence-based questions from the 2019 AAD guidelines for the management of primary CM.4 ChatGPT’s responses were compared with the AAD’s published recommendations and were cross-referenced with responses gathered from our own search of PubMed articles indexed for MEDLINE using the phrase melanoma (cutaneous) and treatment, which included studies from May 2017 to February 2024.
ChatGPT’s answers to 23 of the questions aligned with the AAD’s guidelines (Table 1); in instances when the guidelines were inconclusive regarding pathology, the model provided recommendations supported by our contemporary PubMed literature search. Of the 3 questions related to CM pathology, the AAD guidelines had sufficient evidence to provide recommendations for 2 questions. The first question evaluated the clinical information necessary to help the pathologist improve diagnosis (Table 2). ChatGPT’s response to one question about staged excision and Mohs micrographic surgery for melanoma in situ did not align with the AAD guidelines (Table 3).

Our results showed that ChatGPT provided comprehensive responses aligned with current evidence on CM treatment, except for one surgery question for which its response differed from the AAD guidelines. Our findings are consistent with an observational study that reported board-certified dermatologists rated ChatGPT’s responses on melanoma-related questions as 4.88 on a scale of 1 to 5 (1 indicated completely inaccurate information, 5 indicated complete accuracy for clinical sufficiency in practice). The authors also found that ChatGPT gave vague advice, such as to “get regular skin exams,” which is less specific than dermatologists’ recommendations for annual, biannual, or more frequent examinations.5 ChatGPT’s limitations in offering comprehensive answers for some questions aligned with our findings, specifically the omission of key information in the surgical-related question, highlighting the challenge of relying on AI for nuanced clinical guidance.
We found that ChatGPT considered immunosuppression an important risk factor for CM. Similarly, a 2023 cohort study of 93 patients with melanoma and a history of immunosuppression reported that these patients had a higher risk for CM compared with a control group from the National Cancer Institute’s Surveillance, Epidemiology and End Results Program (standardized incidence ratio, 1.53; 95% CI, 1.12-2.04), indicating that incidence of CM in immunocompromised patients was 53% higher than an age- and sex-matched population cohort.6
Our findings also demonstrated that both ChatGPT’s responses and the AAD guidelines aligned in indicating that evidence linking pregnancy to an increased risk for CM remains inconclusive and that pregnant women should still undergo surveillance. A 2022 retrospective cohort study of 1406 women comparing pregnancy-associated melanoma to non–pregnancy-associated CM had no difference in overall survival (hazard ratio, 0.75; 95% CI, 0.54-1.05).7 However, tumor thickness (2.01-4.00 mm) was greater in postpartum cases compared with cases in nonpregnant women (odds ratio, 1.75; 95% CI, 1.03-2.98), suggesting that pregnancy may affect tumor characteristics.7 These findings underscore the importance of using AI tools such as ChatGPT as a supplement to—rather than as a replacement for—expert clinical judgment and up-to-date medical guidelines.
- Kung TH, Cheatham M, Medenilla A, et al. Performance of ChatGPT on USMLE: potential for AI-assisted medical education using large language models. PLOS Digit Health. 2023;2:E0000198.
- Roster K, Kann RB, Farabi B, et al. Readability and health literacy scores for ChatGPT-generated dermatology public education materials: cross-sectional analysis of sunscreen and melanoma questions. JMIR Dermatol. 2024;7:E50163.
- Dihan Q, Chauhan M, Eleiwa T, et al. Using large language models to generate educational materials on childhood glaucoma. Am J Ophthalmol. 2024;265:28-38.
- Swetter SM, Tsao H, Bichakjian CK, et al. Guidelines of care for the management of primary cutaneous melanoma. J Am Acad Dermatol. 2019;80:208-250.
- Young JN, O’Hagan R, Poplausky D, et al. The utility of ChatGPT in generating patient-facing and clinical responses for melanoma. J Am Acad Dermatol. 2023;89:602-604.
- Killeen TF, Shanley R, Ramesh V, et al. Malignant melanoma in a retrospective cohort of immunocompromised patients: a statistical and pathologic analysis. Cancers (Basel). 2023;15:3600.
- Kiuru M, Li Q, Zhu G, et al. Melanoma in women of childbearing age and in pregnancy in California, 1994-2015: a population-based cohort study. J Eur Acad Dermatol Venereol. 2022;36:2025-2035.
To the Editor:
ChatGPT (OpenAI), a popular large language model that generates responses to user queries, has attracted substantial attention as a potential resource for patient education.1 While prior studies have shown that ChatGPT can provide reliable and general patient information, its alignment with the American Academy of Dermatology’s (AAD’s) guidelines for primary cutaneous melanoma (CM) compared to evidence in the recent literature has not been evaluated.2,3 In this study, we compared ChatGPT’s responses to the 25 evidence-based questions utilized by the AAD to establish its 2019 recommendations for primary CM. Because the 2019 AAD guidelines included literature only through April 2017, we conducted an additional search (May 2017–February 2024) to assess ChatGPT’s alignment with more recent evidence not captured in the guidelines.
On April 17, 2024, 2 authors (D.P. and A.F.) prompted ChatGPT with 25 evidence-based questions from the 2019 AAD guidelines for the management of primary CM.4 ChatGPT’s responses were compared with the AAD’s published recommendations and were cross-referenced with responses gathered from our own search of PubMed articles indexed for MEDLINE using the phrase melanoma (cutaneous) and treatment, which included studies from May 2017 to February 2024.
ChatGPT’s answers to 23 of the questions aligned with the AAD’s guidelines (Table 1); in instances when the guidelines were inconclusive regarding pathology, the model provided recommendations supported by our contemporary PubMed literature search. Of the 3 questions related to CM pathology, the AAD guidelines had sufficient evidence to provide recommendations for 2 questions. The first question evaluated the clinical information necessary to help the pathologist improve diagnosis (Table 2). ChatGPT’s response to one question about staged excision and Mohs micrographic surgery for melanoma in situ did not align with the AAD guidelines (Table 3).



Our results showed that ChatGPT provided comprehensive responses aligned with current evidence on CM treatment, except for one surgery question for which its response differed from the AAD guidelines. Our findings are consistent with an observational study that reported board-certified dermatologists rated ChatGPT’s responses on melanoma-related questions as 4.88 on a scale of 1 to 5 (1 indicated completely inaccurate information, 5 indicated complete accuracy for clinical sufficiency in practice). The authors also found that ChatGPT gave vague advice, such as to “get regular skin exams,” which is less specific than dermatologists’ recommendations for annual, biannual, or more frequent examinations.5 ChatGPT’s limitations in offering comprehensive answers for some questions aligned with our findings, specifically the omission of key information in the surgical-related question, highlighting the challenge of relying on AI for nuanced clinical guidance.
We found that ChatGPT considered immunosuppression an important risk factor for CM. Similarly, a 2023 cohort study of 93 patients with melanoma and a history of immunosuppression reported that these patients had a higher risk for CM compared with a control group from the National Cancer Institute’s Surveillance, Epidemiology and End Results Program (standardized incidence ratio, 1.53; 95% CI, 1.12-2.04), indicating that incidence of CM in immunocompromised patients was 53% higher than an age- and sex-matched population cohort.6
Our findings also demonstrated that both ChatGPT’s responses and the AAD guidelines aligned in indicating that evidence linking pregnancy to an increased risk for CM remains inconclusive and that pregnant women should still undergo surveillance. A 2022 retrospective cohort study of 1406 women comparing pregnancy-associated melanoma to non–pregnancy-associated CM had no difference in overall survival (hazard ratio, 0.75; 95% CI, 0.54-1.05).7 However, tumor thickness (2.01-4.00 mm) was greater in postpartum cases compared with cases in nonpregnant women (odds ratio, 1.75; 95% CI, 1.03-2.98), suggesting that pregnancy may affect tumor characteristics.7 These findings underscore the importance of using AI tools such as ChatGPT as a supplement to—rather than as a replacement for—expert clinical judgment and up-to-date medical guidelines.
To the Editor:
ChatGPT (OpenAI), a popular large language model that generates responses to user queries, has attracted substantial attention as a potential resource for patient education.1 While prior studies have shown that ChatGPT can provide reliable and general patient information, its alignment with the American Academy of Dermatology’s (AAD’s) guidelines for primary cutaneous melanoma (CM) compared to evidence in the recent literature has not been evaluated.2,3 In this study, we compared ChatGPT’s responses to the 25 evidence-based questions utilized by the AAD to establish its 2019 recommendations for primary CM. Because the 2019 AAD guidelines included literature only through April 2017, we conducted an additional search (May 2017–February 2024) to assess ChatGPT’s alignment with more recent evidence not captured in the guidelines.
On April 17, 2024, 2 authors (D.P. and A.F.) prompted ChatGPT with 25 evidence-based questions from the 2019 AAD guidelines for the management of primary CM.4 ChatGPT’s responses were compared with the AAD’s published recommendations and were cross-referenced with responses gathered from our own search of PubMed articles indexed for MEDLINE using the phrase melanoma (cutaneous) and treatment, which included studies from May 2017 to February 2024.
ChatGPT’s answers to 23 of the questions aligned with the AAD’s guidelines (Table 1); in instances when the guidelines were inconclusive regarding pathology, the model provided recommendations supported by our contemporary PubMed literature search. Of the 3 questions related to CM pathology, the AAD guidelines had sufficient evidence to provide recommendations for 2 questions. The first question evaluated the clinical information necessary to help the pathologist improve diagnosis (Table 2). ChatGPT’s response to one question about staged excision and Mohs micrographic surgery for melanoma in situ did not align with the AAD guidelines (Table 3).



Our results showed that ChatGPT provided comprehensive responses aligned with current evidence on CM treatment, except for one surgery question for which its response differed from the AAD guidelines. Our findings are consistent with an observational study that reported board-certified dermatologists rated ChatGPT’s responses on melanoma-related questions as 4.88 on a scale of 1 to 5 (1 indicated completely inaccurate information, 5 indicated complete accuracy for clinical sufficiency in practice). The authors also found that ChatGPT gave vague advice, such as to “get regular skin exams,” which is less specific than dermatologists’ recommendations for annual, biannual, or more frequent examinations.5 ChatGPT’s limitations in offering comprehensive answers for some questions aligned with our findings, specifically the omission of key information in the surgical-related question, highlighting the challenge of relying on AI for nuanced clinical guidance.
We found that ChatGPT considered immunosuppression an important risk factor for CM. Similarly, a 2023 cohort study of 93 patients with melanoma and a history of immunosuppression reported that these patients had a higher risk for CM compared with a control group from the National Cancer Institute’s Surveillance, Epidemiology and End Results Program (standardized incidence ratio, 1.53; 95% CI, 1.12-2.04), indicating that incidence of CM in immunocompromised patients was 53% higher than an age- and sex-matched population cohort.6
Our findings also demonstrated that both ChatGPT’s responses and the AAD guidelines aligned in indicating that evidence linking pregnancy to an increased risk for CM remains inconclusive and that pregnant women should still undergo surveillance. A 2022 retrospective cohort study of 1406 women comparing pregnancy-associated melanoma to non–pregnancy-associated CM had no difference in overall survival (hazard ratio, 0.75; 95% CI, 0.54-1.05).7 However, tumor thickness (2.01-4.00 mm) was greater in postpartum cases compared with cases in nonpregnant women (odds ratio, 1.75; 95% CI, 1.03-2.98), suggesting that pregnancy may affect tumor characteristics.7 These findings underscore the importance of using AI tools such as ChatGPT as a supplement to—rather than as a replacement for—expert clinical judgment and up-to-date medical guidelines.
- Kung TH, Cheatham M, Medenilla A, et al. Performance of ChatGPT on USMLE: potential for AI-assisted medical education using large language models. PLOS Digit Health. 2023;2:E0000198.
- Roster K, Kann RB, Farabi B, et al. Readability and health literacy scores for ChatGPT-generated dermatology public education materials: cross-sectional analysis of sunscreen and melanoma questions. JMIR Dermatol. 2024;7:E50163.
- Dihan Q, Chauhan M, Eleiwa T, et al. Using large language models to generate educational materials on childhood glaucoma. Am J Ophthalmol. 2024;265:28-38.
- Swetter SM, Tsao H, Bichakjian CK, et al. Guidelines of care for the management of primary cutaneous melanoma. J Am Acad Dermatol. 2019;80:208-250.
- Young JN, O’Hagan R, Poplausky D, et al. The utility of ChatGPT in generating patient-facing and clinical responses for melanoma. J Am Acad Dermatol. 2023;89:602-604.
- Killeen TF, Shanley R, Ramesh V, et al. Malignant melanoma in a retrospective cohort of immunocompromised patients: a statistical and pathologic analysis. Cancers (Basel). 2023;15:3600.
- Kiuru M, Li Q, Zhu G, et al. Melanoma in women of childbearing age and in pregnancy in California, 1994-2015: a population-based cohort study. J Eur Acad Dermatol Venereol. 2022;36:2025-2035.
- Kung TH, Cheatham M, Medenilla A, et al. Performance of ChatGPT on USMLE: potential for AI-assisted medical education using large language models. PLOS Digit Health. 2023;2:E0000198.
- Roster K, Kann RB, Farabi B, et al. Readability and health literacy scores for ChatGPT-generated dermatology public education materials: cross-sectional analysis of sunscreen and melanoma questions. JMIR Dermatol. 2024;7:E50163.
- Dihan Q, Chauhan M, Eleiwa T, et al. Using large language models to generate educational materials on childhood glaucoma. Am J Ophthalmol. 2024;265:28-38.
- Swetter SM, Tsao H, Bichakjian CK, et al. Guidelines of care for the management of primary cutaneous melanoma. J Am Acad Dermatol. 2019;80:208-250.
- Young JN, O’Hagan R, Poplausky D, et al. The utility of ChatGPT in generating patient-facing and clinical responses for melanoma. J Am Acad Dermatol. 2023;89:602-604.
- Killeen TF, Shanley R, Ramesh V, et al. Malignant melanoma in a retrospective cohort of immunocompromised patients: a statistical and pathologic analysis. Cancers (Basel). 2023;15:3600.
- Kiuru M, Li Q, Zhu G, et al. Melanoma in women of childbearing age and in pregnancy in California, 1994-2015: a population-based cohort study. J Eur Acad Dermatol Venereol. 2022;36:2025-2035.
Alignment of ChatGPT Responses With AAD Guidelines for Cutaneous Melanoma
Alignment of ChatGPT Responses With AAD Guidelines for Cutaneous Melanoma
PRACTICE POINTS
- ChatGPT provides structured, educational-style responses with broad contextual detail but may omit key clinical nuances such as specific surgical considerations, including staged excision or Mohs micrographic surgery for melanoma in situ.
- Large language models should be viewed as a tool to supplement expert clinical judgment and established guidelines rather than as a standalone replacement for dermatologic decision-making.
Sunscreen Access and Skin Cancer Prevention: Availability at the Marine Corps Exchange
Sunscreen Access and Skin Cancer Prevention: Availability at the Marine Corps Exchange
Military service members are at high risk for skin cancer due to unique occupational and environmental exposures, particularly in the aviation community, in which high-altitude flying, prolonged outdoor aircraft maintenance, physical training, field exercises, and deployments limit access to shade and opportunities for sunscreen reapplication. During deployment or field operations, service members may operate in environments with limited access to SPF products, particularly if sunscreen is not included among personal items.
Research on sun protection strategies and skin cancer risk factors in military personnel is critical to improving prevention, particularly given the higher incidence of melanoma in this population. A 2010 retrospective tumor registry review from the Department of Defense and the National Cancer Institute found higher melanoma rates in military personnel compared with the general population among individuals aged 45 to 49 years (33.62 vs 27.49), 50 to 54 years (49.76 vs 32.18), and 55 to 59 years (178.48 vs 39.17).1
This article discusses barriers to sun protection in military populations, evaluates sunscreen availability in military exchanges, and considers implications for policy and prevention.
Barriers to Sun Protection and Sunscreen Use
According to Rosenberg et al,2 the cause of higher rates of skin cancer among military service members may be multifactorial, including financial barriers to sunscreen use, limited education on photodamage, and insufficient emphasis on sun protection during demanding operational or training activities. Veterans of Operation Enduring Freedom and Operation Iraqi Freedom who were surveyed about UV exposure and sunscreen indicated that 23% (49/211) received education about skin cancer but less than 30% (60/211) used sunscreen consistently during deployment due to lack of access, which has been reported previously.3 Sunscreen adherence also may be reduced in this population due to factors such as skin irritation, cost, poor cosmetic acceptability, and lower utilization among male service members. In their literature review of 9 publications pertaining to skin cancer risk through December 2016, Riemenschneider et al1 noted that male service members comprised 85% of the US military in 2014, and men statistically have lower rates of sunscreen use than women.
Sunscreen Availability and Product Analysis in Military Exchanges
Sunscreen is an important component of skin care for skin cancer prevention. More consistent use has been noted in households with annual incomes of $60,000 or higher.4 Sunscreen product availability has not been evaluated in the military community. Exchange stores are military equivalents of commercial chain stores where service members can purchase tax-free items. The Marine Corps Exchange (MCX) operates on 18 large active-duty bases worldwide. Patrons include active-duty service members from any branch, veterans, and family members. Officials from the MCX headquarters approve and maintain items sold on base. Although product availability may vary by location, standardization is maintained through vendor agreements influenced by customer demand and includes both exchange-branded and private-label products.5
In a review of 96 sunscreen products at Marine Corps Air Station Cherry Point MCX, 62.5% (60/96) met American Academy of Dermatology guideline criteria (SPF ≥30, broad-spectrum UVA/UVB protection, and water resistance of 40-80 minutes).6 Of all products, 79.1% (76/96) were SPF 30 or higher, 76.0% (73/96) were water-resistant, and all provided broad-spectrum protection. Lotion formulations comprised 62.5% (60/96), and the mean price per ounce was $11.96. Opportunities for product expansion include increased availability of options for sensitive skin, as mineral sunscreens comprised 14.6% (14/96) of products; greater variety of products marketed to men, which accounted for 5.2% (5/96); and improved representation for service members with skin of color, as tinted formulations comprised 2.1% (2/96).6
Implications for Policy and Operational Readiness
Given these data, future studies should evaluate sunscreen purchasing behaviors among US service members to determine MCX utilization and whether product selection is driven by active-duty demand or broader consumer purchasing patterns. If product offerings are driven by the civilian customer base, this may result in a lack of tailored options for military service members who are most at risk for high UV exposure. If the MCX does not meet the needs of service members adequately or is inaccessible due to cost or inventory limitations, it highlights a weakness in skin cancer prevention.
Future research should explore not only sunscreen purchasing behavior among service members but also barriers to access and compliance with sun protection measures, as these insights are critical for informing effective policy that balances personal responsibility with institutional support. This could help with advocacy efforts for more effective, readily available options on base. It also could strengthen the argument for alternative strategies to complement sunscreen use, such as a sunscreen allowance, inclusion of sunscreen with provided uniforms and equipment, patient education, work breaks, sun-protective uniform items, and designated shade areas at work.6
Final Thoughts
Policy changes such as routine provision of sunscreen through supply chains, issuing sunscreen with uniforms, or providing a sunscreen stipend could remove financial and logistical barriers to consistent use of sunscreen in military populations. These measures could be impactful during field operations, deployments, and training in austere environments, where commercial purchasing options are limited and UV exposure is high. A proactive approach to sun safety could demonstrate a commitment to preserving the current health and operational readiness of active-duty service members while reducing future financial burdens of skin disease and helping promote wellness in this population during retirement. As with ear protection, uniforms, and eyewear, sunscreen should be considered a standard component of operational readiness.
- Riemenschneider K, Liu J, Powers JG. Skin cancer in the military: a systematic review of melanoma and nonmelanoma skin cancer incidence, prevention, and screening among active duty and veteran personnel. J Am Acad Dermatol. 2018;78:1185-1192. doi:10.1016/j.jaad.2017.11.062
- Rosenberg A, Cho S. We can do better at protecting our service members from skin cancer. Mil Med. 2022;187:311-313. doi:10.1093/milmed/usac198
- Powers JG, Patel NA, Powers EM, et al. Skin cancer risk factors and preventative behaviors among United States military veterans deployed to Iraq and Afghanistan. J Invest Dermatol. 2015;135:2871-2873. doi:10.1038/jid.2015.238
- Tahir S, Ihebom D, Garcia E, et al. Sunscreen access, availability, and quality in dollar store chains. J Am Acad of Dermatol. 2024;90:1284-1285. doi:10.1016/j.jaad.2024.02.018
- mymcx.com. Published 2025. Accessed May 15, 2025. https://www.mymcx.com/
- American Academy of Dermatology Association. How to select a sunscreen. Accessed April 23, 2026. https://www.aad.org/public/everyday-care/sun-protection/shade-clothing-sunscreen/how-to-select-sunscreen.
- Anderson S, Zhang S, Crotty A, et al. Bridging the knowledge-action gap in skin cancer prevention among US military personnel. Cutis. 2025;115:146-149. doi:10.12788/cutis.1207
Military service members are at high risk for skin cancer due to unique occupational and environmental exposures, particularly in the aviation community, in which high-altitude flying, prolonged outdoor aircraft maintenance, physical training, field exercises, and deployments limit access to shade and opportunities for sunscreen reapplication. During deployment or field operations, service members may operate in environments with limited access to SPF products, particularly if sunscreen is not included among personal items.
Research on sun protection strategies and skin cancer risk factors in military personnel is critical to improving prevention, particularly given the higher incidence of melanoma in this population. A 2010 retrospective tumor registry review from the Department of Defense and the National Cancer Institute found higher melanoma rates in military personnel compared with the general population among individuals aged 45 to 49 years (33.62 vs 27.49), 50 to 54 years (49.76 vs 32.18), and 55 to 59 years (178.48 vs 39.17).1
This article discusses barriers to sun protection in military populations, evaluates sunscreen availability in military exchanges, and considers implications for policy and prevention.
Barriers to Sun Protection and Sunscreen Use
According to Rosenberg et al,2 the cause of higher rates of skin cancer among military service members may be multifactorial, including financial barriers to sunscreen use, limited education on photodamage, and insufficient emphasis on sun protection during demanding operational or training activities. Veterans of Operation Enduring Freedom and Operation Iraqi Freedom who were surveyed about UV exposure and sunscreen indicated that 23% (49/211) received education about skin cancer but less than 30% (60/211) used sunscreen consistently during deployment due to lack of access, which has been reported previously.3 Sunscreen adherence also may be reduced in this population due to factors such as skin irritation, cost, poor cosmetic acceptability, and lower utilization among male service members. In their literature review of 9 publications pertaining to skin cancer risk through December 2016, Riemenschneider et al1 noted that male service members comprised 85% of the US military in 2014, and men statistically have lower rates of sunscreen use than women.
Sunscreen Availability and Product Analysis in Military Exchanges
Sunscreen is an important component of skin care for skin cancer prevention. More consistent use has been noted in households with annual incomes of $60,000 or higher.4 Sunscreen product availability has not been evaluated in the military community. Exchange stores are military equivalents of commercial chain stores where service members can purchase tax-free items. The Marine Corps Exchange (MCX) operates on 18 large active-duty bases worldwide. Patrons include active-duty service members from any branch, veterans, and family members. Officials from the MCX headquarters approve and maintain items sold on base. Although product availability may vary by location, standardization is maintained through vendor agreements influenced by customer demand and includes both exchange-branded and private-label products.5
In a review of 96 sunscreen products at Marine Corps Air Station Cherry Point MCX, 62.5% (60/96) met American Academy of Dermatology guideline criteria (SPF ≥30, broad-spectrum UVA/UVB protection, and water resistance of 40-80 minutes).6 Of all products, 79.1% (76/96) were SPF 30 or higher, 76.0% (73/96) were water-resistant, and all provided broad-spectrum protection. Lotion formulations comprised 62.5% (60/96), and the mean price per ounce was $11.96. Opportunities for product expansion include increased availability of options for sensitive skin, as mineral sunscreens comprised 14.6% (14/96) of products; greater variety of products marketed to men, which accounted for 5.2% (5/96); and improved representation for service members with skin of color, as tinted formulations comprised 2.1% (2/96).6
Implications for Policy and Operational Readiness
Given these data, future studies should evaluate sunscreen purchasing behaviors among US service members to determine MCX utilization and whether product selection is driven by active-duty demand or broader consumer purchasing patterns. If product offerings are driven by the civilian customer base, this may result in a lack of tailored options for military service members who are most at risk for high UV exposure. If the MCX does not meet the needs of service members adequately or is inaccessible due to cost or inventory limitations, it highlights a weakness in skin cancer prevention.
Future research should explore not only sunscreen purchasing behavior among service members but also barriers to access and compliance with sun protection measures, as these insights are critical for informing effective policy that balances personal responsibility with institutional support. This could help with advocacy efforts for more effective, readily available options on base. It also could strengthen the argument for alternative strategies to complement sunscreen use, such as a sunscreen allowance, inclusion of sunscreen with provided uniforms and equipment, patient education, work breaks, sun-protective uniform items, and designated shade areas at work.6
Final Thoughts
Policy changes such as routine provision of sunscreen through supply chains, issuing sunscreen with uniforms, or providing a sunscreen stipend could remove financial and logistical barriers to consistent use of sunscreen in military populations. These measures could be impactful during field operations, deployments, and training in austere environments, where commercial purchasing options are limited and UV exposure is high. A proactive approach to sun safety could demonstrate a commitment to preserving the current health and operational readiness of active-duty service members while reducing future financial burdens of skin disease and helping promote wellness in this population during retirement. As with ear protection, uniforms, and eyewear, sunscreen should be considered a standard component of operational readiness.
Military service members are at high risk for skin cancer due to unique occupational and environmental exposures, particularly in the aviation community, in which high-altitude flying, prolonged outdoor aircraft maintenance, physical training, field exercises, and deployments limit access to shade and opportunities for sunscreen reapplication. During deployment or field operations, service members may operate in environments with limited access to SPF products, particularly if sunscreen is not included among personal items.
Research on sun protection strategies and skin cancer risk factors in military personnel is critical to improving prevention, particularly given the higher incidence of melanoma in this population. A 2010 retrospective tumor registry review from the Department of Defense and the National Cancer Institute found higher melanoma rates in military personnel compared with the general population among individuals aged 45 to 49 years (33.62 vs 27.49), 50 to 54 years (49.76 vs 32.18), and 55 to 59 years (178.48 vs 39.17).1
This article discusses barriers to sun protection in military populations, evaluates sunscreen availability in military exchanges, and considers implications for policy and prevention.
Barriers to Sun Protection and Sunscreen Use
According to Rosenberg et al,2 the cause of higher rates of skin cancer among military service members may be multifactorial, including financial barriers to sunscreen use, limited education on photodamage, and insufficient emphasis on sun protection during demanding operational or training activities. Veterans of Operation Enduring Freedom and Operation Iraqi Freedom who were surveyed about UV exposure and sunscreen indicated that 23% (49/211) received education about skin cancer but less than 30% (60/211) used sunscreen consistently during deployment due to lack of access, which has been reported previously.3 Sunscreen adherence also may be reduced in this population due to factors such as skin irritation, cost, poor cosmetic acceptability, and lower utilization among male service members. In their literature review of 9 publications pertaining to skin cancer risk through December 2016, Riemenschneider et al1 noted that male service members comprised 85% of the US military in 2014, and men statistically have lower rates of sunscreen use than women.
Sunscreen Availability and Product Analysis in Military Exchanges
Sunscreen is an important component of skin care for skin cancer prevention. More consistent use has been noted in households with annual incomes of $60,000 or higher.4 Sunscreen product availability has not been evaluated in the military community. Exchange stores are military equivalents of commercial chain stores where service members can purchase tax-free items. The Marine Corps Exchange (MCX) operates on 18 large active-duty bases worldwide. Patrons include active-duty service members from any branch, veterans, and family members. Officials from the MCX headquarters approve and maintain items sold on base. Although product availability may vary by location, standardization is maintained through vendor agreements influenced by customer demand and includes both exchange-branded and private-label products.5
In a review of 96 sunscreen products at Marine Corps Air Station Cherry Point MCX, 62.5% (60/96) met American Academy of Dermatology guideline criteria (SPF ≥30, broad-spectrum UVA/UVB protection, and water resistance of 40-80 minutes).6 Of all products, 79.1% (76/96) were SPF 30 or higher, 76.0% (73/96) were water-resistant, and all provided broad-spectrum protection. Lotion formulations comprised 62.5% (60/96), and the mean price per ounce was $11.96. Opportunities for product expansion include increased availability of options for sensitive skin, as mineral sunscreens comprised 14.6% (14/96) of products; greater variety of products marketed to men, which accounted for 5.2% (5/96); and improved representation for service members with skin of color, as tinted formulations comprised 2.1% (2/96).6
Implications for Policy and Operational Readiness
Given these data, future studies should evaluate sunscreen purchasing behaviors among US service members to determine MCX utilization and whether product selection is driven by active-duty demand or broader consumer purchasing patterns. If product offerings are driven by the civilian customer base, this may result in a lack of tailored options for military service members who are most at risk for high UV exposure. If the MCX does not meet the needs of service members adequately or is inaccessible due to cost or inventory limitations, it highlights a weakness in skin cancer prevention.
Future research should explore not only sunscreen purchasing behavior among service members but also barriers to access and compliance with sun protection measures, as these insights are critical for informing effective policy that balances personal responsibility with institutional support. This could help with advocacy efforts for more effective, readily available options on base. It also could strengthen the argument for alternative strategies to complement sunscreen use, such as a sunscreen allowance, inclusion of sunscreen with provided uniforms and equipment, patient education, work breaks, sun-protective uniform items, and designated shade areas at work.6
Final Thoughts
Policy changes such as routine provision of sunscreen through supply chains, issuing sunscreen with uniforms, or providing a sunscreen stipend could remove financial and logistical barriers to consistent use of sunscreen in military populations. These measures could be impactful during field operations, deployments, and training in austere environments, where commercial purchasing options are limited and UV exposure is high. A proactive approach to sun safety could demonstrate a commitment to preserving the current health and operational readiness of active-duty service members while reducing future financial burdens of skin disease and helping promote wellness in this population during retirement. As with ear protection, uniforms, and eyewear, sunscreen should be considered a standard component of operational readiness.
- Riemenschneider K, Liu J, Powers JG. Skin cancer in the military: a systematic review of melanoma and nonmelanoma skin cancer incidence, prevention, and screening among active duty and veteran personnel. J Am Acad Dermatol. 2018;78:1185-1192. doi:10.1016/j.jaad.2017.11.062
- Rosenberg A, Cho S. We can do better at protecting our service members from skin cancer. Mil Med. 2022;187:311-313. doi:10.1093/milmed/usac198
- Powers JG, Patel NA, Powers EM, et al. Skin cancer risk factors and preventative behaviors among United States military veterans deployed to Iraq and Afghanistan. J Invest Dermatol. 2015;135:2871-2873. doi:10.1038/jid.2015.238
- Tahir S, Ihebom D, Garcia E, et al. Sunscreen access, availability, and quality in dollar store chains. J Am Acad of Dermatol. 2024;90:1284-1285. doi:10.1016/j.jaad.2024.02.018
- mymcx.com. Published 2025. Accessed May 15, 2025. https://www.mymcx.com/
- American Academy of Dermatology Association. How to select a sunscreen. Accessed April 23, 2026. https://www.aad.org/public/everyday-care/sun-protection/shade-clothing-sunscreen/how-to-select-sunscreen.
- Anderson S, Zhang S, Crotty A, et al. Bridging the knowledge-action gap in skin cancer prevention among US military personnel. Cutis. 2025;115:146-149. doi:10.12788/cutis.1207
- Riemenschneider K, Liu J, Powers JG. Skin cancer in the military: a systematic review of melanoma and nonmelanoma skin cancer incidence, prevention, and screening among active duty and veteran personnel. J Am Acad Dermatol. 2018;78:1185-1192. doi:10.1016/j.jaad.2017.11.062
- Rosenberg A, Cho S. We can do better at protecting our service members from skin cancer. Mil Med. 2022;187:311-313. doi:10.1093/milmed/usac198
- Powers JG, Patel NA, Powers EM, et al. Skin cancer risk factors and preventative behaviors among United States military veterans deployed to Iraq and Afghanistan. J Invest Dermatol. 2015;135:2871-2873. doi:10.1038/jid.2015.238
- Tahir S, Ihebom D, Garcia E, et al. Sunscreen access, availability, and quality in dollar store chains. J Am Acad of Dermatol. 2024;90:1284-1285. doi:10.1016/j.jaad.2024.02.018
- mymcx.com. Published 2025. Accessed May 15, 2025. https://www.mymcx.com/
- American Academy of Dermatology Association. How to select a sunscreen. Accessed April 23, 2026. https://www.aad.org/public/everyday-care/sun-protection/shade-clothing-sunscreen/how-to-select-sunscreen.
- Anderson S, Zhang S, Crotty A, et al. Bridging the knowledge-action gap in skin cancer prevention among US military personnel. Cutis. 2025;115:146-149. doi:10.12788/cutis.1207
Sunscreen Access and Skin Cancer Prevention: Availability at the Marine Corps Exchange
Sunscreen Access and Skin Cancer Prevention: Availability at the Marine Corps Exchange
Practice Points
- Military service members face increased UV exposure from occupational and deployment conditions, but sunscreen use is limited by logistical, educational, and behavioral barriers.
- Sunscreen availability in military exchanges partially meets dermatologic guidelines, highlighting opportunities to expand tailored options and integrate sun protection into operational readiness policy.