User login
Investigating Real-World Tolerance and Dose Reductions of Oncology Multikinase Inhibitors in a VA Population
Investigating Real-World Tolerance and Dose Reductions of Oncology Multikinase Inhibitors in a VA Population
The US Department of Veterans Affairs (VA) annually treats around 450,000 veterans with cancer and diagnoses an additional 56,000.1,2 Oral multikinase inhibitors (MKIs) are widely used as targeted therapies for many different malignancies. Despite the ease of oral administration, these agents are often accompanied by significant adverse effects (AEs) and drug-drug interactions.3,4 Common AEs include hypertension, cutaneous reactions, gastrointestinal disturbances, proteinuria, and fatigue. Some serious outcomes that may occur are myocardial infarction, thrombosis, nephrotic syndrome, hemorrhage, hepatotoxicity, and gastrointestinal events.5,6 Due to poor tolerability of these AEs, dose reductions, frequent therapy holds, and discontinuation of therapy may occur.
The US Food and Drug Administration recognizes dosing challenges with novel therapies and has created the Oncology Center of Excellence (OCE) Project Optimus initiative to reform dose optimization in oncology drug development. The initiative aims to shift the focus from establishing dose regimens based on the maximum tolerated doses of cytotoxic chemotherapeutics to an emphasis on maximum efficacy, safety, and tolerability, which better reflect real-world dosing.7,8
MKIs can be challenging to manage because of the frequent toxicity-related dose reductions, interruptions, and discontinuations. In a multicenter retrospective study, Schnadig et al investigated dosing characteristics of first-line sunitinib for advanced renal cell carcinoma (RCC) and found that, among 114 patients who experienced AEs while taking sunitinib, 39.5% had dose reductions, 5.3% delayed therapy, 18.4% required additional supportive medications, and 22.8% discontinued sunitinib.9 Overall survival and median progression-free survival of these patients were lower than reported by Motzer et al in a phase 3 clinical trial.10 Schnadig et al concluded that patients treated with sunitinib for RCC in the community setting required more frequent dose reductions and had less time on therapy compared with patients in clinical trials, which ultimately impacted clinical outcomes.9
At the VA North Texas Health Care System (VANTHCS), patients with cancer have difficulty tolerating MKIs and often require dose alterations and/or discontinuation because of drug intolerance rather than discontinuation due to progression. Frequent dose adjustments for toxicity management can place more strain on patients and health care resources because of additional appointments, clinician time, and emergency department visits. Escalating drug costs can also cause concern when prescription doses are unused or changed frequently.
To capture and quantify prescribing practices and dose adjustments, this study evaluated the tolerability of MKIs at VANTHCS. This analysis may also guide clinicians in the selection of starting doses as well as dose titration expectations to optimize MKI therapy.
METHODS
This single-center, retrospective chart review analyzed patients receiving oral oncology MKIs for various malignancies at VANTHCS between January 1, 2014, and October 31, 2024. Participants included adults aged ≥ 18 years with a prescription for axitinib, cabozantinib, lenvatinib, pazopanib, regorafenib, sorafenib, or sunitinib initiated by the hematology/oncology service at VANTHCS. Patients were included if they had follow-up documentation with the hematology/oncology service and/or other VANTHCS clinicians outlining their course of therapy after MKI initiation. Patients were excluded if they did not have sufficient follow-up documentation (eg, transferred care to a non-VA health care practitioner [HCP], moved to another VA health care system), were enrolled in clinical trials, or were prescribed an MKI from a Care in the Community (CITC) prescriber. Electronic health record review and data collection were performed using the VA Computerized Patient Record System and Research Electronic Data Capture. Data were collected from the time of initiation to cessation of therapy and included information regarding therapy changes, progressive disease, and date of death, when available. Data collected included age, sex, race, comorbidities, date of death, type of malignancy and subtypes, cancer stage, MKI used (ie, drug, dose, frequency, schedule, and indication), dates of medication changes (ie, start, adjustment, hold, discontinuation), concurrent antineoplastic treatments, and AEs documented at times of dose change or interruption.
The primary outcome was MKI tolerance determined using relative dose intensity (RDI) and mean and median time on therapy. Two methods are used to calculate RDI that vary in how they approach time on therapy as outlined in Hawn et al.11 This study used method 2, which accounts for holds in therapy by comparing the actual duration of treatment with the duration expected according to treatment protocol. Method 1 compares the prescribed dose with the administered dose and does not adjust for holds.11 Using method 2, the RDI in this study was calculated by dividing the total actual dose given by the total indicated dose for the malignancy being treated, which accounts for duration of treatment.

The total actual dose was the strength, frequency, and days on therapy for each time frame of treatment multiplied together. This method accounted for all dose adjustments and time periods of treatment holds, including patient self-adjustments, prescriber-directed adjustments, and nonadherence determined by HCP documentation and/or prescription data. Similarly, the indicated total dose was calculated by multiplying the indicated strength, frequency, and all days that treatment should have occurred (time from start to finish). Indicated doses were derived from the prescribing information for each malignancy with the exception of sunitinib, for which the off-label dose of 37.5 mg daily was considered a full dose.12,13 The total indicated dose for axitinib was calculated by considering the dose escalation schedule from the prescribing information.
Patients who required dose reductions due to renal/hepatic impairments or drug-drug interactions had their total indicated dose calculated using dose adjustments listed in the prescribing information. The mean RDI for each MKI agent was calculated by averaging the RDI for each prescription. The overall combined mean RDI included the means of all the MKIs reviewed to avoid skewing the results toward an MKI with more prescriptions. RDIs were also calculated for each cancer type for each agent. Additional descriptive secondary outcomes included rates of AEs and adjustments in doses.
RESULTS
Electronic data extraction identified 278 patients with 366 MKI prescriptions, of which 108 veterans with 158 MKI prescriptions were excluded. The top reason for exclusion was patients managed through CITC. Ultimately, 170 veterans with 208 MKI prescriptions managed by the VANTHCS hematology/oncology clinic were included (Table 1). Among patients receiving MKIs, the mean age was 72.7 years, 98% were male, and 99% had metastatic disease.
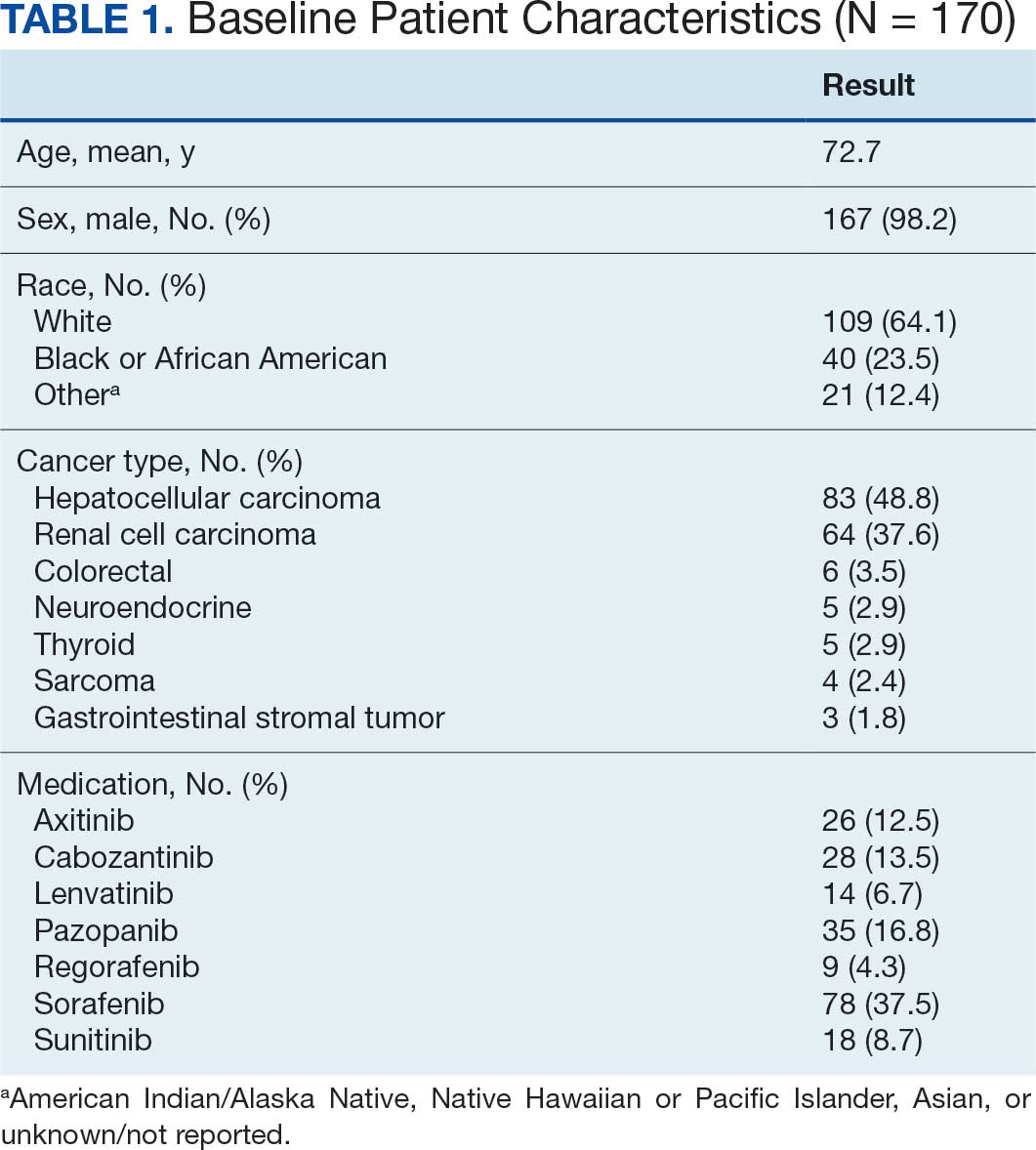
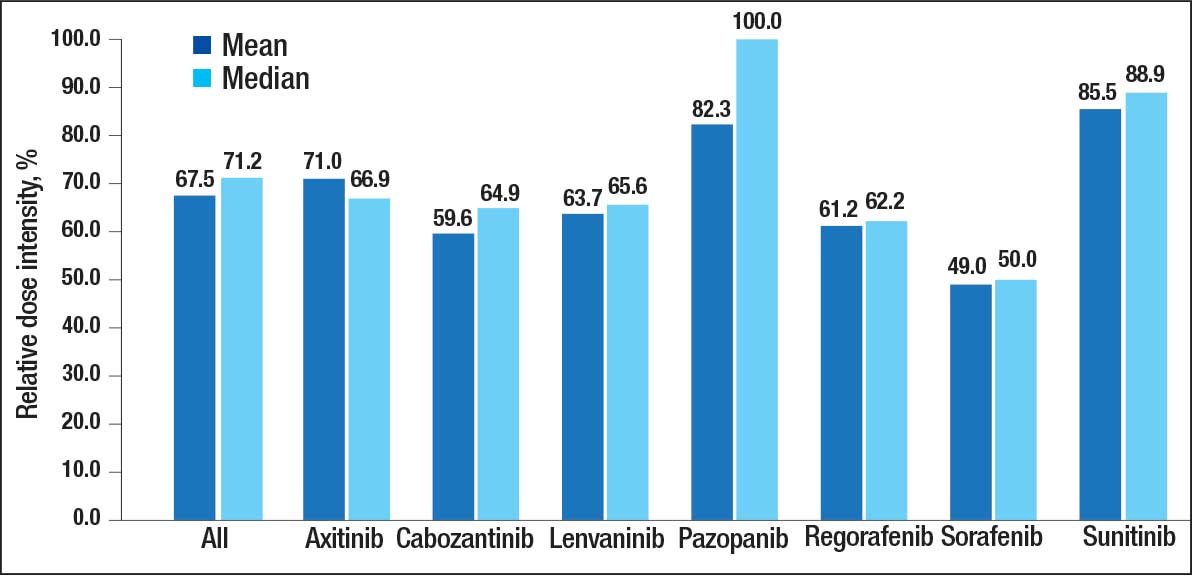
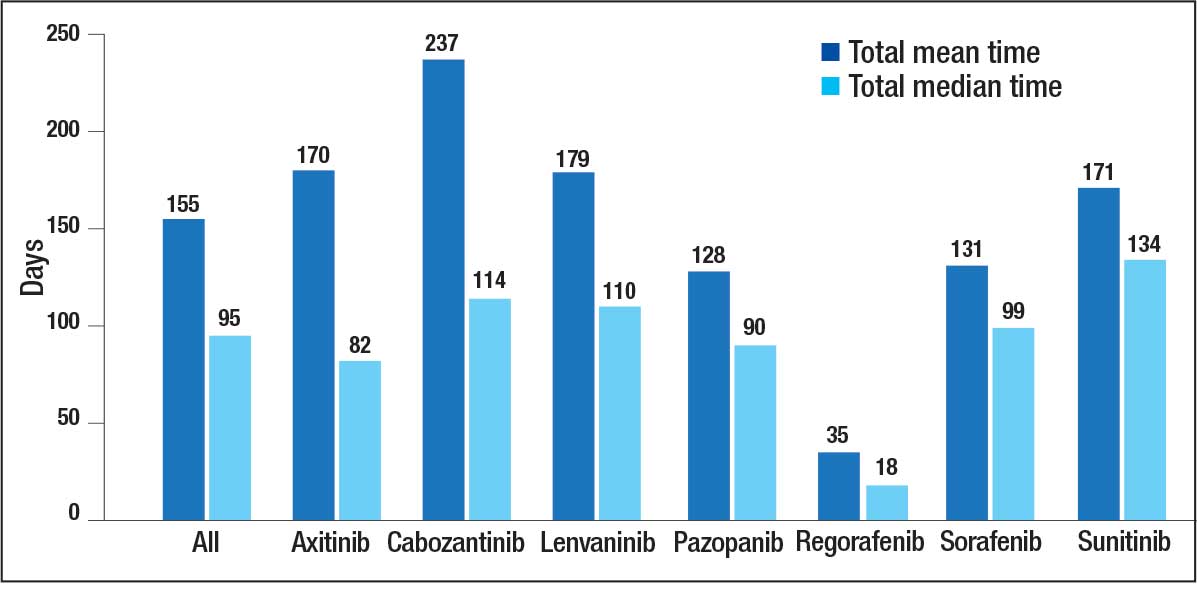
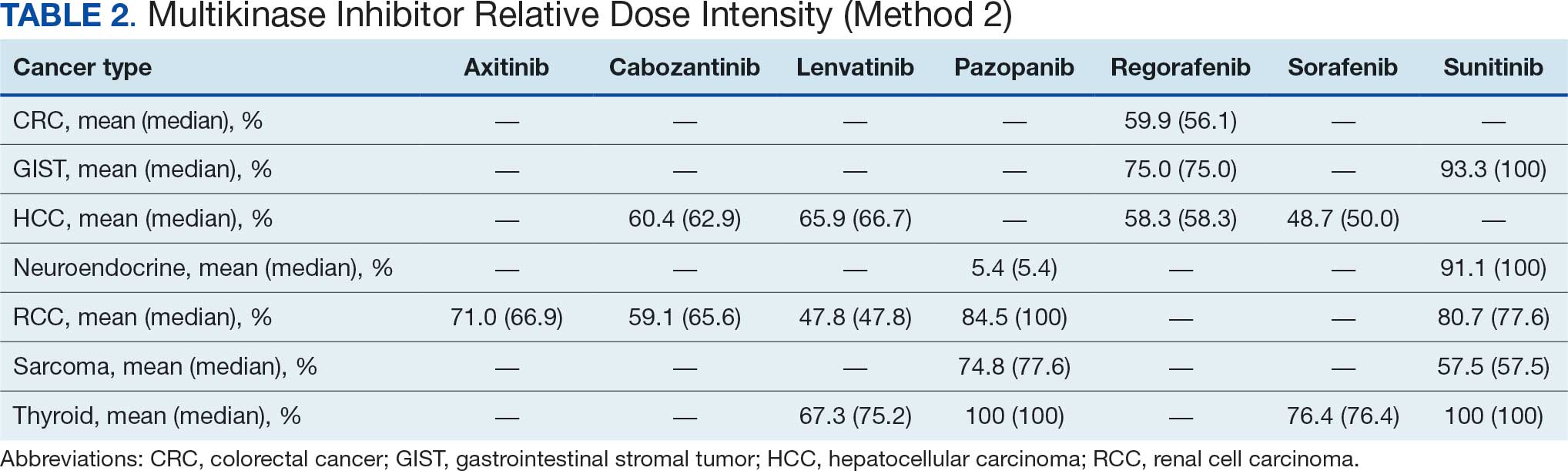
The overall combined mean MKI RDI was 67.5% using method 2 and ranged from 85.5% for sunitinib to 49.0% for sorafenib (Figure 1). Additional information regarding mean and median RDIs using method 2 is shown in Figure 1 and further subdivided by cancer type in Table 2. Median RDIs overall were similar to mean RDIs for most agents. Figure 2 indicates the mean and median time on therapy, reflecting time on therapy excluding days therapy was held. The overall combined mean and median days on therapy for all MKIs were 155 days and 95 days, respectively. Mean time on therapy depended on the agent used and ranged from 35 days (regorafenib) to 237 days (cabozantinib).
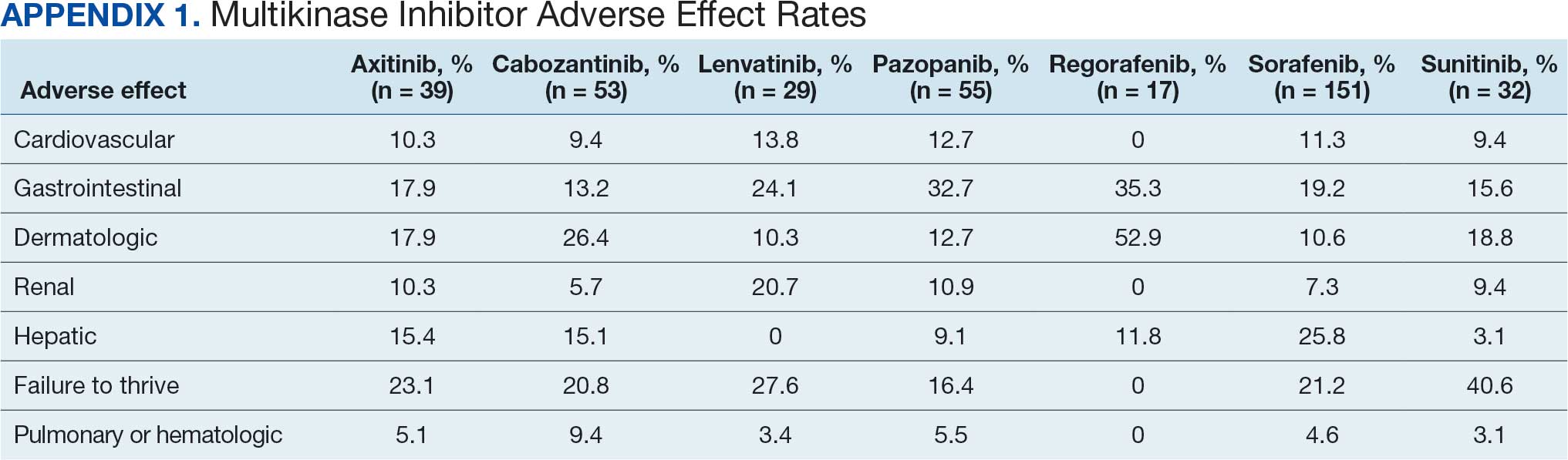
Of 208 MKI prescriptions, 127 (61.1%) were initiated at a reduced dose due to baseline concerns for tolerance such as performance status, frailty, and prior intolerance of other treatments. Eighty-one prescriptions (38.9%) were initiated at their indicated doses. Ninety prescriptions (43.3%) required dose reductions during treatment. Some MKI prescriptions had multiple dose increases and decreases, which is why RDI more accurately reflects dose adjustments. A total of 376 AEs that contributed to a dose adjustment, hold, or discontinuation occurred across all MKI prescriptions. The most common AEs were 82 failure-to-thrive events (21.8%) (fatigue, malaise, loss of appetite, reduced mobility, global decline), 79 gastrointestinal events (21.0%) (nausea, vomiting, diarrhea, abdominal pain), 62 dermatologic events (16.5%) (rash, hand-foot skin reactions, allergic response), 61 hepatic dysfunction events (16.2%) (liver enzyme elevations, hyperbilirubinemia), 40 cardiovascular events (10.6%) (hypertension, heart failure exacerbations, edema), and 33 renal dysfunction events (8.8%) (acute kidney injury, proteinuria) (Appendix 1).
DISCUSSION
The mean RDI of MKI prescriptions used in the veteran population at VANTHCS was about two-thirds of the indicated dose. These results indicate that most veterans required dose reductions and/or holds due to concerns over initial tolerance/performance status, worsening clinical condition, and/or intolerable AEs attributed to treatment. A retrospective study conducted by Denduluri et al suggested that an RDI of < 85% is a clinically meaningful reduction for traditional chemotherapy based on previous literature.14 However, it is less clear what RDI should be expected specifically for MKIs in real-world populations. The MKI phase 3 approval trials in RCC for axitinib, lenvatinib, and sunitinib found median RDIs of 89.4%, 69.6% to 70.4%, and 83.9%, respectively. Each trial cited dose reductions most commonly as the result of treatment-related AEs.15,16
Studies on the impact of RDIs on survival outcomes found that higher RDIs may improve overall and progression-free survival. Retrospective studies inspecting lenvatinib in hepatocellular carcinoma (HCC) indicated that an RDI > 70% in the initial 4 weeks resulted in favorable survival outcomes.17 Similarly, a retrospective study investigating sunitinib in RCC found that an RDI > 60% conferred favorable survival outcomes.18 Alghamdi et al noted that patients taking sorafenib for HCC who had RDI > 50% had a favorable trend in survival characteristics. Interestingly, the study found an RDI of 50% to 75% appeared to have better survival than an RDI > 75%.19 The authors of these studies hypothesized that additional dose reductions allowed for longer total time on therapy due to improved tolerability.17-19
This analysis found that the RDIs for most MKI agents at VANTHCS were < 85% and lower than the RDIs found in other review articles and phase 3 trials, with the exceptions of pazopanib in thyroid cancer and sunitinib in gastrointestinal stromal tumor (GIST), thyroid cancer, and neuroendocrine cancer. The reasons for the lower RDIs in this study are likely multifactorial, reflecting patient population characteristics, off-label dosing practices, and HCP experiences with these agents. Many veterans have chronic comorbidities that could contribute to reduced performance status and ability to tolerate these therapies. Despite attempts to preemptively reduce doses for patients and account for potential impaired tolerance, there were patients who required further dose reductions in our study.
Failure to thrive was the most common AE leading to dose adjustment or discontinuation, which illustrates the extensive effects these agents have on patient functioning in a real-world population. Notably though, the RDI for sunitinib was higher in this population because about half of patients were dosed using the off-label recommendation, whereas the prescribing information recommends a more intensive 6-week dosing cycle for certain cancer types.12,13,20 Sorafenib was also often dose-adjusted based on a pharmacokinetic study of sorafenib in renal/hepatic dysfunction, and the RDI likely reflects the off-label prescribing pattern.21
Patients with thyroid cancer were found to have higher RDIs compared with those receiving the same agents for other cancer types. Improved tolerability of MKIs in thyroid cancer may be due to a generally more tolerable disease course. Thyroid cancer is the most common cancer in individuals aged < 40 years, a population that is often more robust with fewer comorbidities. Moreover, the 5-year relative survival rate for thyroid cancer remains > 98%.22 This rate is in contrast to those for other cancer types such as HCC, with a 5-year relative survival rate of only 15%.23
It is challenging to compare the mean and median times on therapy found in this study with those in current literature, as this review included multiple different cancer types for each agent. However, the numbers are generally lower than durations of therapy found across the different disease states and further emphasize the difficulty in tolerating MKIs in the VANTHCS population. Regorafenib had a short duration of time on therapy, which highlights the importance of trials like ReDOS and initiatives such as OCE Project Optimus in helping improve tolerance.7,8,24
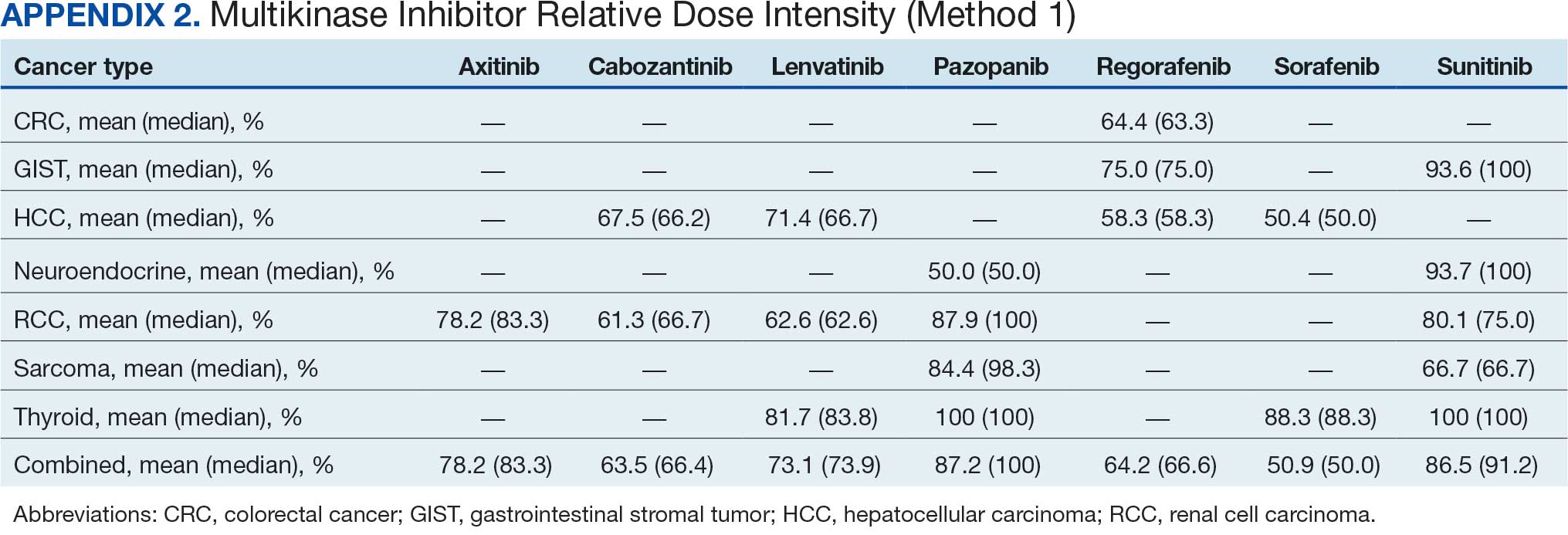
Comparing our results with other studies proved challenging because the RDI calculation methods were not specified. Calculating RDIs in this study using method 1, which does not account for holds, resulted in higher RDIs (Appendix 2). Using method 1, all MKIs had RDIs < 85%, except for pazopanib in thyroid cancer (100%) and RCC (87.9%), and sunitinib in GIST (93.6%), thyroid cancer (100%), and neuroendocrine cancer (93.7%). Notably, using method 1 increased the RDI for pazopanib in neuroendocrine cancer from 5.4% to 50.0%. The low RDI was attributed to a single veteran with a long hold duration, which demonstrates the discrepancy that can occur between the 2 methods.
Limitations
The retrospective design, lack of survival outcomes, and difficulty comparing results with other literature were limitations of this study. Because survival outcomes were not evaluated, future research should seek to investigate how RDIs and dose adjustments made among MKIs can affect survival outcomes in real-world populations. This veteran population with cancer often had multiple chronic comorbidities, which may have contributed to difficulty tolerating MKIs and could have impacted results. Disease-related factors may have influenced the poor tolerance of the MKIs and were not specifically accounted for. Adjustment for comorbidities was not possible because of discrepancies and/or incomplete diagnosis codes and Eastern Cooperative Oncology Group performance status scores documented in patient charts. Therefore, we decided not to report these findings due to potential inaccuracies.
CONCLUSIONS
Results of this study demonstrate that oncology MKI agents used at VANTHCS were difficult for patients to tolerate, leading to suboptimal dosing compared with indicated doses established in clinical trials and prescribing information. Clinicians may use these data to help guide clinical decision-making whenever initiating and managing MKI agents in this population. These findings reinforce that MKI agents are often difficult to tolerate in real-world practice, and indicated doses are often not achieved. Further studies should aim to investigate the effect that various RDIs have on overall survival. Further investigation into different dosing schemes for MKIs to improve tolerability and longer-term use may also prove beneficial.
This analysis may help guide clinicians to carefully approach dosing MKI agents in the veteran population. Given the RDI and AEs, more clinicians may consider starting at lower than indicated doses with the goal to titrate up as tolerated. Additionally, the results highlight the importance of considering palliative care consults and ensuring appropriate supportive care agents are preemptively engaged and adjusted as needed. Approaching dosing and titrations cautiously may help reduce the burden of management on the health care system.
- Frequently asked questions. VA National Oncology Program. 2025. Accessed December 15, 2025. https://www.cancer.va.gov/CANCER/faqs.html
- Torez L. Reigniting the cancer moonshot to beat cancer. VA News. April 20, 2023. Accessed April 6, 2026. https://news.va.gov/118378/reigniting-the-cancer-moonshot-to-beat-cancer
- Shah NN, Casella E, Capozzi D, et al. Improving the safety of oral chemotherapy at an academic medical center. J Oncol Pract. 2016;12:e71-e76. doi:10.1200/JOP.2015.007260
- Hussaarts KGAM, Veerman GDM, Jansman FGA, et al. Clinically relevant drug interactions with multikinase inhibitors: a review. Ther Adv Med Oncol. 2019;11:1758835918818347. doi:10.1177/1758835918818347
- Shyam Sunder S, Sharma UC, Pokharel S. Adverse effects of tyrosine kinase inhibitors in cancer therapy: pathophysiology, mechanisms and clinical management. Signal Transduct Target Ther. 2023;8:262. doi:10.1038/s41392-023-01469-6
- Thomson RJ, Moshirfar M, Ronquillo Y. Tyrosine kinase inhibitors. In: StatPearls [Internet]. StatPearls Publishing; updated July 18, 2023. Accessed December 15, 2025. https://www.ncbi.nlm.nih.gov/books/NBK563322/
- Project Optimus. US Food and Drug Administration. Updated December 6, 2024. Accessed December 15, 2025. https://www.fda.gov/about-fda/oncology-center-excellence/project-optimus
- Optimizing the dosage of human prescription drugs and biological products for the treatment of oncologic diseases: Guidance for Industry. Docket number FDA-2022-D-2827. US Food and Drug Administration. August 2024. Accessed December 15, 2025. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/optimizing-dosage-human-prescription-drugs-and-biological-products-treatment-oncologic-diseases
- Schnadig ID, Hutson TE, Chung H, et al. Dosing patterns, toxicity, and outcomes in patients treated with first-line sunitinib for advanced renal cell carcinoma in community-based practices. Clin Genitourin Cancer. 2014;12:413-421. doi:10.1016/j.clgc.2014.06.015
- Motzer RJ, Hutson TE, Tomczak P, et al. Sunitinib versus interferon alfa in metastatic renal-cell carcinoma. N Engl J Med. 2007;356:115-124. doi:10.1056/nejmoa065044
- Hawn C, Bansal D. Relative dose intensity in oncology trials: a discussion of two approaches. PharmaSUG. 2024. Accessed April 6, 2026. https://pharmasug.org/proceedings/2024/ST/PharmaSUG-2024-ST-297.pdf
- George S, Merriam P, Maki RG, et al. Multicenter phase II trial of sunitinib in the treatment of nongastrointestinal stromal tumor sarcomas. J Clin Oncol. 2009;27:3154-3160. doi:10.1200/jco.2008.20.9890
- George S, Blay JY, Casali PG, et al. Clinical evaluation of continuous daily dosing of sunitinib malate in patients with advanced gastrointestinal stromal tumour after imatinib failure. Eur J Cancer. 2009;45:1959-1968. doi:10.1016/j.ejca.2009.02.011
- Denduluri N, Patt DA, Wang Y, et al. Dose delays, dose reductions, and relative dose intensity in patients with cancer who received adjuvant or neoadjuvant chemotherapy in community oncology practices. J Natl Compr Canc Netw. 2015;13:1383-1393. doi:10.6004/jnccn.2015.0166
- Motzer RJ, Penkov K, Haanen J, et al. Avelumab plus axitinib versus sunitinib for advanced renal-cell carcinoma. N Engl J Med. 2019;380:1103-1115. doi:10.1056/nejmoa1816047
- Motzer R, Alekseev B, Rha SY, et al. Lenvatinib plus pembrolizumab or everolimus for advanced renal cell carcinoma. N Engl J Med. 2021;384:1289-1300. doi:10.1056/nejmoa2035716
- Kirino S, Tsuchiya K, Kurosaki M, et al. Relative dose intensity over the first four weeks of lenvatinib therapy is a factor of favorable response and overall survival in patients with unresectable hepatocellular carcinoma. PloS One. 2020;15:e0231828. doi:10.1371/journal.pone.0231828
- Ishihara H, Takagi T, Kondo T, et al. Decreased relative dose intensity during the early phase of treatment impacts the therapeutic efficacy of sunitinib in metastatic renal cell carcinoma. Jpn J Clin Oncol. 2018;48:667-672. doi:10.1093/jjco/hyy078
- Alghamdi MA, Amaro CP, Lee-Ying R, et al. Effect of sorafenib starting dose and dose intensity on survival in patients with hepatocellular carcinoma: results from a Canadian Multicenter Database. Cancer Med. 2020;9:4918-4928. doi:10.1002/cam4.3228
- Motzer RJ, Rini BI, Bukowski RM, et al. Sunitinib in patients with metastatic renal cell carcinoma. JAMA. 2006;295:2516-2524. doi:10.1001/jama.295.21.2516
- Miller AA, Murry DJ, Owzar K, et al. Phase I and pharmacokinetic study of sorafenib in patients with hepatic or renal dysfunction: CALGB 60301. J Clin Oncol. 2009;27:1800-1805. doi:10.1200/jco.2008.20.0931
- Boucai L, Zafereo M, Cabanillas ME. Thyroid cancer: a review. JAMA. 2024;331:425-435. doi:10.1001/jama.2023.26348
- Amin N, Anwar J, Sulaiman A, et al. Hepatocellular carcinoma: a comprehensive review. Diseases. 2025;13:207. doi:10.3390/diseases13070207
- Bekaii-Saab TS, Ou FS, Ahn DH, et al. Regorafenib dose-optimisation in patients with refractory metastatic colorectal cancer (ReDOS): a randomised, multicentre, open-label, phase 2 study. Lancet Oncol. 2019;20:1070-1082. doi:10.1016/s1470-2045(19)30272-4
The US Department of Veterans Affairs (VA) annually treats around 450,000 veterans with cancer and diagnoses an additional 56,000.1,2 Oral multikinase inhibitors (MKIs) are widely used as targeted therapies for many different malignancies. Despite the ease of oral administration, these agents are often accompanied by significant adverse effects (AEs) and drug-drug interactions.3,4 Common AEs include hypertension, cutaneous reactions, gastrointestinal disturbances, proteinuria, and fatigue. Some serious outcomes that may occur are myocardial infarction, thrombosis, nephrotic syndrome, hemorrhage, hepatotoxicity, and gastrointestinal events.5,6 Due to poor tolerability of these AEs, dose reductions, frequent therapy holds, and discontinuation of therapy may occur.
The US Food and Drug Administration recognizes dosing challenges with novel therapies and has created the Oncology Center of Excellence (OCE) Project Optimus initiative to reform dose optimization in oncology drug development. The initiative aims to shift the focus from establishing dose regimens based on the maximum tolerated doses of cytotoxic chemotherapeutics to an emphasis on maximum efficacy, safety, and tolerability, which better reflect real-world dosing.7,8
MKIs can be challenging to manage because of the frequent toxicity-related dose reductions, interruptions, and discontinuations. In a multicenter retrospective study, Schnadig et al investigated dosing characteristics of first-line sunitinib for advanced renal cell carcinoma (RCC) and found that, among 114 patients who experienced AEs while taking sunitinib, 39.5% had dose reductions, 5.3% delayed therapy, 18.4% required additional supportive medications, and 22.8% discontinued sunitinib.9 Overall survival and median progression-free survival of these patients were lower than reported by Motzer et al in a phase 3 clinical trial.10 Schnadig et al concluded that patients treated with sunitinib for RCC in the community setting required more frequent dose reductions and had less time on therapy compared with patients in clinical trials, which ultimately impacted clinical outcomes.9
At the VA North Texas Health Care System (VANTHCS), patients with cancer have difficulty tolerating MKIs and often require dose alterations and/or discontinuation because of drug intolerance rather than discontinuation due to progression. Frequent dose adjustments for toxicity management can place more strain on patients and health care resources because of additional appointments, clinician time, and emergency department visits. Escalating drug costs can also cause concern when prescription doses are unused or changed frequently.
To capture and quantify prescribing practices and dose adjustments, this study evaluated the tolerability of MKIs at VANTHCS. This analysis may also guide clinicians in the selection of starting doses as well as dose titration expectations to optimize MKI therapy.
METHODS
This single-center, retrospective chart review analyzed patients receiving oral oncology MKIs for various malignancies at VANTHCS between January 1, 2014, and October 31, 2024. Participants included adults aged ≥ 18 years with a prescription for axitinib, cabozantinib, lenvatinib, pazopanib, regorafenib, sorafenib, or sunitinib initiated by the hematology/oncology service at VANTHCS. Patients were included if they had follow-up documentation with the hematology/oncology service and/or other VANTHCS clinicians outlining their course of therapy after MKI initiation. Patients were excluded if they did not have sufficient follow-up documentation (eg, transferred care to a non-VA health care practitioner [HCP], moved to another VA health care system), were enrolled in clinical trials, or were prescribed an MKI from a Care in the Community (CITC) prescriber. Electronic health record review and data collection were performed using the VA Computerized Patient Record System and Research Electronic Data Capture. Data were collected from the time of initiation to cessation of therapy and included information regarding therapy changes, progressive disease, and date of death, when available. Data collected included age, sex, race, comorbidities, date of death, type of malignancy and subtypes, cancer stage, MKI used (ie, drug, dose, frequency, schedule, and indication), dates of medication changes (ie, start, adjustment, hold, discontinuation), concurrent antineoplastic treatments, and AEs documented at times of dose change or interruption.
The primary outcome was MKI tolerance determined using relative dose intensity (RDI) and mean and median time on therapy. Two methods are used to calculate RDI that vary in how they approach time on therapy as outlined in Hawn et al.11 This study used method 2, which accounts for holds in therapy by comparing the actual duration of treatment with the duration expected according to treatment protocol. Method 1 compares the prescribed dose with the administered dose and does not adjust for holds.11 Using method 2, the RDI in this study was calculated by dividing the total actual dose given by the total indicated dose for the malignancy being treated, which accounts for duration of treatment.

The total actual dose was the strength, frequency, and days on therapy for each time frame of treatment multiplied together. This method accounted for all dose adjustments and time periods of treatment holds, including patient self-adjustments, prescriber-directed adjustments, and nonadherence determined by HCP documentation and/or prescription data. Similarly, the indicated total dose was calculated by multiplying the indicated strength, frequency, and all days that treatment should have occurred (time from start to finish). Indicated doses were derived from the prescribing information for each malignancy with the exception of sunitinib, for which the off-label dose of 37.5 mg daily was considered a full dose.12,13 The total indicated dose for axitinib was calculated by considering the dose escalation schedule from the prescribing information.
Patients who required dose reductions due to renal/hepatic impairments or drug-drug interactions had their total indicated dose calculated using dose adjustments listed in the prescribing information. The mean RDI for each MKI agent was calculated by averaging the RDI for each prescription. The overall combined mean RDI included the means of all the MKIs reviewed to avoid skewing the results toward an MKI with more prescriptions. RDIs were also calculated for each cancer type for each agent. Additional descriptive secondary outcomes included rates of AEs and adjustments in doses.
RESULTS
Electronic data extraction identified 278 patients with 366 MKI prescriptions, of which 108 veterans with 158 MKI prescriptions were excluded. The top reason for exclusion was patients managed through CITC. Ultimately, 170 veterans with 208 MKI prescriptions managed by the VANTHCS hematology/oncology clinic were included (Table 1). Among patients receiving MKIs, the mean age was 72.7 years, 98% were male, and 99% had metastatic disease.

The overall combined mean MKI RDI was 67.5% using method 2 and ranged from 85.5% for sunitinib to 49.0% for sorafenib (Figure 1). Additional information regarding mean and median RDIs using method 2 is shown in Figure 1 and further subdivided by cancer type in Table 2. Median RDIs overall were similar to mean RDIs for most agents. Figure 2 indicates the mean and median time on therapy, reflecting time on therapy excluding days therapy was held. The overall combined mean and median days on therapy for all MKIs were 155 days and 95 days, respectively. Mean time on therapy depended on the agent used and ranged from 35 days (regorafenib) to 237 days (cabozantinib).



Of 208 MKI prescriptions, 127 (61.1%) were initiated at a reduced dose due to baseline concerns for tolerance such as performance status, frailty, and prior intolerance of other treatments. Eighty-one prescriptions (38.9%) were initiated at their indicated doses. Ninety prescriptions (43.3%) required dose reductions during treatment. Some MKI prescriptions had multiple dose increases and decreases, which is why RDI more accurately reflects dose adjustments. A total of 376 AEs that contributed to a dose adjustment, hold, or discontinuation occurred across all MKI prescriptions. The most common AEs were 82 failure-to-thrive events (21.8%) (fatigue, malaise, loss of appetite, reduced mobility, global decline), 79 gastrointestinal events (21.0%) (nausea, vomiting, diarrhea, abdominal pain), 62 dermatologic events (16.5%) (rash, hand-foot skin reactions, allergic response), 61 hepatic dysfunction events (16.2%) (liver enzyme elevations, hyperbilirubinemia), 40 cardiovascular events (10.6%) (hypertension, heart failure exacerbations, edema), and 33 renal dysfunction events (8.8%) (acute kidney injury, proteinuria) (Appendix 1).

DISCUSSION
The mean RDI of MKI prescriptions used in the veteran population at VANTHCS was about two-thirds of the indicated dose. These results indicate that most veterans required dose reductions and/or holds due to concerns over initial tolerance/performance status, worsening clinical condition, and/or intolerable AEs attributed to treatment. A retrospective study conducted by Denduluri et al suggested that an RDI of < 85% is a clinically meaningful reduction for traditional chemotherapy based on previous literature.14 However, it is less clear what RDI should be expected specifically for MKIs in real-world populations. The MKI phase 3 approval trials in RCC for axitinib, lenvatinib, and sunitinib found median RDIs of 89.4%, 69.6% to 70.4%, and 83.9%, respectively. Each trial cited dose reductions most commonly as the result of treatment-related AEs.15,16
Studies on the impact of RDIs on survival outcomes found that higher RDIs may improve overall and progression-free survival. Retrospective studies inspecting lenvatinib in hepatocellular carcinoma (HCC) indicated that an RDI > 70% in the initial 4 weeks resulted in favorable survival outcomes.17 Similarly, a retrospective study investigating sunitinib in RCC found that an RDI > 60% conferred favorable survival outcomes.18 Alghamdi et al noted that patients taking sorafenib for HCC who had RDI > 50% had a favorable trend in survival characteristics. Interestingly, the study found an RDI of 50% to 75% appeared to have better survival than an RDI > 75%.19 The authors of these studies hypothesized that additional dose reductions allowed for longer total time on therapy due to improved tolerability.17-19
This analysis found that the RDIs for most MKI agents at VANTHCS were < 85% and lower than the RDIs found in other review articles and phase 3 trials, with the exceptions of pazopanib in thyroid cancer and sunitinib in gastrointestinal stromal tumor (GIST), thyroid cancer, and neuroendocrine cancer. The reasons for the lower RDIs in this study are likely multifactorial, reflecting patient population characteristics, off-label dosing practices, and HCP experiences with these agents. Many veterans have chronic comorbidities that could contribute to reduced performance status and ability to tolerate these therapies. Despite attempts to preemptively reduce doses for patients and account for potential impaired tolerance, there were patients who required further dose reductions in our study.
Failure to thrive was the most common AE leading to dose adjustment or discontinuation, which illustrates the extensive effects these agents have on patient functioning in a real-world population. Notably though, the RDI for sunitinib was higher in this population because about half of patients were dosed using the off-label recommendation, whereas the prescribing information recommends a more intensive 6-week dosing cycle for certain cancer types.12,13,20 Sorafenib was also often dose-adjusted based on a pharmacokinetic study of sorafenib in renal/hepatic dysfunction, and the RDI likely reflects the off-label prescribing pattern.21
Patients with thyroid cancer were found to have higher RDIs compared with those receiving the same agents for other cancer types. Improved tolerability of MKIs in thyroid cancer may be due to a generally more tolerable disease course. Thyroid cancer is the most common cancer in individuals aged < 40 years, a population that is often more robust with fewer comorbidities. Moreover, the 5-year relative survival rate for thyroid cancer remains > 98%.22 This rate is in contrast to those for other cancer types such as HCC, with a 5-year relative survival rate of only 15%.23
It is challenging to compare the mean and median times on therapy found in this study with those in current literature, as this review included multiple different cancer types for each agent. However, the numbers are generally lower than durations of therapy found across the different disease states and further emphasize the difficulty in tolerating MKIs in the VANTHCS population. Regorafenib had a short duration of time on therapy, which highlights the importance of trials like ReDOS and initiatives such as OCE Project Optimus in helping improve tolerance.7,8,24
Comparing our results with other studies proved challenging because the RDI calculation methods were not specified. Calculating RDIs in this study using method 1, which does not account for holds, resulted in higher RDIs (Appendix 2). Using method 1, all MKIs had RDIs < 85%, except for pazopanib in thyroid cancer (100%) and RCC (87.9%), and sunitinib in GIST (93.6%), thyroid cancer (100%), and neuroendocrine cancer (93.7%). Notably, using method 1 increased the RDI for pazopanib in neuroendocrine cancer from 5.4% to 50.0%. The low RDI was attributed to a single veteran with a long hold duration, which demonstrates the discrepancy that can occur between the 2 methods.

Limitations
The retrospective design, lack of survival outcomes, and difficulty comparing results with other literature were limitations of this study. Because survival outcomes were not evaluated, future research should seek to investigate how RDIs and dose adjustments made among MKIs can affect survival outcomes in real-world populations. This veteran population with cancer often had multiple chronic comorbidities, which may have contributed to difficulty tolerating MKIs and could have impacted results. Disease-related factors may have influenced the poor tolerance of the MKIs and were not specifically accounted for. Adjustment for comorbidities was not possible because of discrepancies and/or incomplete diagnosis codes and Eastern Cooperative Oncology Group performance status scores documented in patient charts. Therefore, we decided not to report these findings due to potential inaccuracies.
CONCLUSIONS
Results of this study demonstrate that oncology MKI agents used at VANTHCS were difficult for patients to tolerate, leading to suboptimal dosing compared with indicated doses established in clinical trials and prescribing information. Clinicians may use these data to help guide clinical decision-making whenever initiating and managing MKI agents in this population. These findings reinforce that MKI agents are often difficult to tolerate in real-world practice, and indicated doses are often not achieved. Further studies should aim to investigate the effect that various RDIs have on overall survival. Further investigation into different dosing schemes for MKIs to improve tolerability and longer-term use may also prove beneficial.
This analysis may help guide clinicians to carefully approach dosing MKI agents in the veteran population. Given the RDI and AEs, more clinicians may consider starting at lower than indicated doses with the goal to titrate up as tolerated. Additionally, the results highlight the importance of considering palliative care consults and ensuring appropriate supportive care agents are preemptively engaged and adjusted as needed. Approaching dosing and titrations cautiously may help reduce the burden of management on the health care system.
The US Department of Veterans Affairs (VA) annually treats around 450,000 veterans with cancer and diagnoses an additional 56,000.1,2 Oral multikinase inhibitors (MKIs) are widely used as targeted therapies for many different malignancies. Despite the ease of oral administration, these agents are often accompanied by significant adverse effects (AEs) and drug-drug interactions.3,4 Common AEs include hypertension, cutaneous reactions, gastrointestinal disturbances, proteinuria, and fatigue. Some serious outcomes that may occur are myocardial infarction, thrombosis, nephrotic syndrome, hemorrhage, hepatotoxicity, and gastrointestinal events.5,6 Due to poor tolerability of these AEs, dose reductions, frequent therapy holds, and discontinuation of therapy may occur.
The US Food and Drug Administration recognizes dosing challenges with novel therapies and has created the Oncology Center of Excellence (OCE) Project Optimus initiative to reform dose optimization in oncology drug development. The initiative aims to shift the focus from establishing dose regimens based on the maximum tolerated doses of cytotoxic chemotherapeutics to an emphasis on maximum efficacy, safety, and tolerability, which better reflect real-world dosing.7,8
MKIs can be challenging to manage because of the frequent toxicity-related dose reductions, interruptions, and discontinuations. In a multicenter retrospective study, Schnadig et al investigated dosing characteristics of first-line sunitinib for advanced renal cell carcinoma (RCC) and found that, among 114 patients who experienced AEs while taking sunitinib, 39.5% had dose reductions, 5.3% delayed therapy, 18.4% required additional supportive medications, and 22.8% discontinued sunitinib.9 Overall survival and median progression-free survival of these patients were lower than reported by Motzer et al in a phase 3 clinical trial.10 Schnadig et al concluded that patients treated with sunitinib for RCC in the community setting required more frequent dose reductions and had less time on therapy compared with patients in clinical trials, which ultimately impacted clinical outcomes.9
At the VA North Texas Health Care System (VANTHCS), patients with cancer have difficulty tolerating MKIs and often require dose alterations and/or discontinuation because of drug intolerance rather than discontinuation due to progression. Frequent dose adjustments for toxicity management can place more strain on patients and health care resources because of additional appointments, clinician time, and emergency department visits. Escalating drug costs can also cause concern when prescription doses are unused or changed frequently.
To capture and quantify prescribing practices and dose adjustments, this study evaluated the tolerability of MKIs at VANTHCS. This analysis may also guide clinicians in the selection of starting doses as well as dose titration expectations to optimize MKI therapy.
METHODS
This single-center, retrospective chart review analyzed patients receiving oral oncology MKIs for various malignancies at VANTHCS between January 1, 2014, and October 31, 2024. Participants included adults aged ≥ 18 years with a prescription for axitinib, cabozantinib, lenvatinib, pazopanib, regorafenib, sorafenib, or sunitinib initiated by the hematology/oncology service at VANTHCS. Patients were included if they had follow-up documentation with the hematology/oncology service and/or other VANTHCS clinicians outlining their course of therapy after MKI initiation. Patients were excluded if they did not have sufficient follow-up documentation (eg, transferred care to a non-VA health care practitioner [HCP], moved to another VA health care system), were enrolled in clinical trials, or were prescribed an MKI from a Care in the Community (CITC) prescriber. Electronic health record review and data collection were performed using the VA Computerized Patient Record System and Research Electronic Data Capture. Data were collected from the time of initiation to cessation of therapy and included information regarding therapy changes, progressive disease, and date of death, when available. Data collected included age, sex, race, comorbidities, date of death, type of malignancy and subtypes, cancer stage, MKI used (ie, drug, dose, frequency, schedule, and indication), dates of medication changes (ie, start, adjustment, hold, discontinuation), concurrent antineoplastic treatments, and AEs documented at times of dose change or interruption.
The primary outcome was MKI tolerance determined using relative dose intensity (RDI) and mean and median time on therapy. Two methods are used to calculate RDI that vary in how they approach time on therapy as outlined in Hawn et al.11 This study used method 2, which accounts for holds in therapy by comparing the actual duration of treatment with the duration expected according to treatment protocol. Method 1 compares the prescribed dose with the administered dose and does not adjust for holds.11 Using method 2, the RDI in this study was calculated by dividing the total actual dose given by the total indicated dose for the malignancy being treated, which accounts for duration of treatment.

The total actual dose was the strength, frequency, and days on therapy for each time frame of treatment multiplied together. This method accounted for all dose adjustments and time periods of treatment holds, including patient self-adjustments, prescriber-directed adjustments, and nonadherence determined by HCP documentation and/or prescription data. Similarly, the indicated total dose was calculated by multiplying the indicated strength, frequency, and all days that treatment should have occurred (time from start to finish). Indicated doses were derived from the prescribing information for each malignancy with the exception of sunitinib, for which the off-label dose of 37.5 mg daily was considered a full dose.12,13 The total indicated dose for axitinib was calculated by considering the dose escalation schedule from the prescribing information.
Patients who required dose reductions due to renal/hepatic impairments or drug-drug interactions had their total indicated dose calculated using dose adjustments listed in the prescribing information. The mean RDI for each MKI agent was calculated by averaging the RDI for each prescription. The overall combined mean RDI included the means of all the MKIs reviewed to avoid skewing the results toward an MKI with more prescriptions. RDIs were also calculated for each cancer type for each agent. Additional descriptive secondary outcomes included rates of AEs and adjustments in doses.
RESULTS
Electronic data extraction identified 278 patients with 366 MKI prescriptions, of which 108 veterans with 158 MKI prescriptions were excluded. The top reason for exclusion was patients managed through CITC. Ultimately, 170 veterans with 208 MKI prescriptions managed by the VANTHCS hematology/oncology clinic were included (Table 1). Among patients receiving MKIs, the mean age was 72.7 years, 98% were male, and 99% had metastatic disease.

The overall combined mean MKI RDI was 67.5% using method 2 and ranged from 85.5% for sunitinib to 49.0% for sorafenib (Figure 1). Additional information regarding mean and median RDIs using method 2 is shown in Figure 1 and further subdivided by cancer type in Table 2. Median RDIs overall were similar to mean RDIs for most agents. Figure 2 indicates the mean and median time on therapy, reflecting time on therapy excluding days therapy was held. The overall combined mean and median days on therapy for all MKIs were 155 days and 95 days, respectively. Mean time on therapy depended on the agent used and ranged from 35 days (regorafenib) to 237 days (cabozantinib).



Of 208 MKI prescriptions, 127 (61.1%) were initiated at a reduced dose due to baseline concerns for tolerance such as performance status, frailty, and prior intolerance of other treatments. Eighty-one prescriptions (38.9%) were initiated at their indicated doses. Ninety prescriptions (43.3%) required dose reductions during treatment. Some MKI prescriptions had multiple dose increases and decreases, which is why RDI more accurately reflects dose adjustments. A total of 376 AEs that contributed to a dose adjustment, hold, or discontinuation occurred across all MKI prescriptions. The most common AEs were 82 failure-to-thrive events (21.8%) (fatigue, malaise, loss of appetite, reduced mobility, global decline), 79 gastrointestinal events (21.0%) (nausea, vomiting, diarrhea, abdominal pain), 62 dermatologic events (16.5%) (rash, hand-foot skin reactions, allergic response), 61 hepatic dysfunction events (16.2%) (liver enzyme elevations, hyperbilirubinemia), 40 cardiovascular events (10.6%) (hypertension, heart failure exacerbations, edema), and 33 renal dysfunction events (8.8%) (acute kidney injury, proteinuria) (Appendix 1).

DISCUSSION
The mean RDI of MKI prescriptions used in the veteran population at VANTHCS was about two-thirds of the indicated dose. These results indicate that most veterans required dose reductions and/or holds due to concerns over initial tolerance/performance status, worsening clinical condition, and/or intolerable AEs attributed to treatment. A retrospective study conducted by Denduluri et al suggested that an RDI of < 85% is a clinically meaningful reduction for traditional chemotherapy based on previous literature.14 However, it is less clear what RDI should be expected specifically for MKIs in real-world populations. The MKI phase 3 approval trials in RCC for axitinib, lenvatinib, and sunitinib found median RDIs of 89.4%, 69.6% to 70.4%, and 83.9%, respectively. Each trial cited dose reductions most commonly as the result of treatment-related AEs.15,16
Studies on the impact of RDIs on survival outcomes found that higher RDIs may improve overall and progression-free survival. Retrospective studies inspecting lenvatinib in hepatocellular carcinoma (HCC) indicated that an RDI > 70% in the initial 4 weeks resulted in favorable survival outcomes.17 Similarly, a retrospective study investigating sunitinib in RCC found that an RDI > 60% conferred favorable survival outcomes.18 Alghamdi et al noted that patients taking sorafenib for HCC who had RDI > 50% had a favorable trend in survival characteristics. Interestingly, the study found an RDI of 50% to 75% appeared to have better survival than an RDI > 75%.19 The authors of these studies hypothesized that additional dose reductions allowed for longer total time on therapy due to improved tolerability.17-19
This analysis found that the RDIs for most MKI agents at VANTHCS were < 85% and lower than the RDIs found in other review articles and phase 3 trials, with the exceptions of pazopanib in thyroid cancer and sunitinib in gastrointestinal stromal tumor (GIST), thyroid cancer, and neuroendocrine cancer. The reasons for the lower RDIs in this study are likely multifactorial, reflecting patient population characteristics, off-label dosing practices, and HCP experiences with these agents. Many veterans have chronic comorbidities that could contribute to reduced performance status and ability to tolerate these therapies. Despite attempts to preemptively reduce doses for patients and account for potential impaired tolerance, there were patients who required further dose reductions in our study.
Failure to thrive was the most common AE leading to dose adjustment or discontinuation, which illustrates the extensive effects these agents have on patient functioning in a real-world population. Notably though, the RDI for sunitinib was higher in this population because about half of patients were dosed using the off-label recommendation, whereas the prescribing information recommends a more intensive 6-week dosing cycle for certain cancer types.12,13,20 Sorafenib was also often dose-adjusted based on a pharmacokinetic study of sorafenib in renal/hepatic dysfunction, and the RDI likely reflects the off-label prescribing pattern.21
Patients with thyroid cancer were found to have higher RDIs compared with those receiving the same agents for other cancer types. Improved tolerability of MKIs in thyroid cancer may be due to a generally more tolerable disease course. Thyroid cancer is the most common cancer in individuals aged < 40 years, a population that is often more robust with fewer comorbidities. Moreover, the 5-year relative survival rate for thyroid cancer remains > 98%.22 This rate is in contrast to those for other cancer types such as HCC, with a 5-year relative survival rate of only 15%.23
It is challenging to compare the mean and median times on therapy found in this study with those in current literature, as this review included multiple different cancer types for each agent. However, the numbers are generally lower than durations of therapy found across the different disease states and further emphasize the difficulty in tolerating MKIs in the VANTHCS population. Regorafenib had a short duration of time on therapy, which highlights the importance of trials like ReDOS and initiatives such as OCE Project Optimus in helping improve tolerance.7,8,24
Comparing our results with other studies proved challenging because the RDI calculation methods were not specified. Calculating RDIs in this study using method 1, which does not account for holds, resulted in higher RDIs (Appendix 2). Using method 1, all MKIs had RDIs < 85%, except for pazopanib in thyroid cancer (100%) and RCC (87.9%), and sunitinib in GIST (93.6%), thyroid cancer (100%), and neuroendocrine cancer (93.7%). Notably, using method 1 increased the RDI for pazopanib in neuroendocrine cancer from 5.4% to 50.0%. The low RDI was attributed to a single veteran with a long hold duration, which demonstrates the discrepancy that can occur between the 2 methods.

Limitations
The retrospective design, lack of survival outcomes, and difficulty comparing results with other literature were limitations of this study. Because survival outcomes were not evaluated, future research should seek to investigate how RDIs and dose adjustments made among MKIs can affect survival outcomes in real-world populations. This veteran population with cancer often had multiple chronic comorbidities, which may have contributed to difficulty tolerating MKIs and could have impacted results. Disease-related factors may have influenced the poor tolerance of the MKIs and were not specifically accounted for. Adjustment for comorbidities was not possible because of discrepancies and/or incomplete diagnosis codes and Eastern Cooperative Oncology Group performance status scores documented in patient charts. Therefore, we decided not to report these findings due to potential inaccuracies.
CONCLUSIONS
Results of this study demonstrate that oncology MKI agents used at VANTHCS were difficult for patients to tolerate, leading to suboptimal dosing compared with indicated doses established in clinical trials and prescribing information. Clinicians may use these data to help guide clinical decision-making whenever initiating and managing MKI agents in this population. These findings reinforce that MKI agents are often difficult to tolerate in real-world practice, and indicated doses are often not achieved. Further studies should aim to investigate the effect that various RDIs have on overall survival. Further investigation into different dosing schemes for MKIs to improve tolerability and longer-term use may also prove beneficial.
This analysis may help guide clinicians to carefully approach dosing MKI agents in the veteran population. Given the RDI and AEs, more clinicians may consider starting at lower than indicated doses with the goal to titrate up as tolerated. Additionally, the results highlight the importance of considering palliative care consults and ensuring appropriate supportive care agents are preemptively engaged and adjusted as needed. Approaching dosing and titrations cautiously may help reduce the burden of management on the health care system.
- Frequently asked questions. VA National Oncology Program. 2025. Accessed December 15, 2025. https://www.cancer.va.gov/CANCER/faqs.html
- Torez L. Reigniting the cancer moonshot to beat cancer. VA News. April 20, 2023. Accessed April 6, 2026. https://news.va.gov/118378/reigniting-the-cancer-moonshot-to-beat-cancer
- Shah NN, Casella E, Capozzi D, et al. Improving the safety of oral chemotherapy at an academic medical center. J Oncol Pract. 2016;12:e71-e76. doi:10.1200/JOP.2015.007260
- Hussaarts KGAM, Veerman GDM, Jansman FGA, et al. Clinically relevant drug interactions with multikinase inhibitors: a review. Ther Adv Med Oncol. 2019;11:1758835918818347. doi:10.1177/1758835918818347
- Shyam Sunder S, Sharma UC, Pokharel S. Adverse effects of tyrosine kinase inhibitors in cancer therapy: pathophysiology, mechanisms and clinical management. Signal Transduct Target Ther. 2023;8:262. doi:10.1038/s41392-023-01469-6
- Thomson RJ, Moshirfar M, Ronquillo Y. Tyrosine kinase inhibitors. In: StatPearls [Internet]. StatPearls Publishing; updated July 18, 2023. Accessed December 15, 2025. https://www.ncbi.nlm.nih.gov/books/NBK563322/
- Project Optimus. US Food and Drug Administration. Updated December 6, 2024. Accessed December 15, 2025. https://www.fda.gov/about-fda/oncology-center-excellence/project-optimus
- Optimizing the dosage of human prescription drugs and biological products for the treatment of oncologic diseases: Guidance for Industry. Docket number FDA-2022-D-2827. US Food and Drug Administration. August 2024. Accessed December 15, 2025. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/optimizing-dosage-human-prescription-drugs-and-biological-products-treatment-oncologic-diseases
- Schnadig ID, Hutson TE, Chung H, et al. Dosing patterns, toxicity, and outcomes in patients treated with first-line sunitinib for advanced renal cell carcinoma in community-based practices. Clin Genitourin Cancer. 2014;12:413-421. doi:10.1016/j.clgc.2014.06.015
- Motzer RJ, Hutson TE, Tomczak P, et al. Sunitinib versus interferon alfa in metastatic renal-cell carcinoma. N Engl J Med. 2007;356:115-124. doi:10.1056/nejmoa065044
- Hawn C, Bansal D. Relative dose intensity in oncology trials: a discussion of two approaches. PharmaSUG. 2024. Accessed April 6, 2026. https://pharmasug.org/proceedings/2024/ST/PharmaSUG-2024-ST-297.pdf
- George S, Merriam P, Maki RG, et al. Multicenter phase II trial of sunitinib in the treatment of nongastrointestinal stromal tumor sarcomas. J Clin Oncol. 2009;27:3154-3160. doi:10.1200/jco.2008.20.9890
- George S, Blay JY, Casali PG, et al. Clinical evaluation of continuous daily dosing of sunitinib malate in patients with advanced gastrointestinal stromal tumour after imatinib failure. Eur J Cancer. 2009;45:1959-1968. doi:10.1016/j.ejca.2009.02.011
- Denduluri N, Patt DA, Wang Y, et al. Dose delays, dose reductions, and relative dose intensity in patients with cancer who received adjuvant or neoadjuvant chemotherapy in community oncology practices. J Natl Compr Canc Netw. 2015;13:1383-1393. doi:10.6004/jnccn.2015.0166
- Motzer RJ, Penkov K, Haanen J, et al. Avelumab plus axitinib versus sunitinib for advanced renal-cell carcinoma. N Engl J Med. 2019;380:1103-1115. doi:10.1056/nejmoa1816047
- Motzer R, Alekseev B, Rha SY, et al. Lenvatinib plus pembrolizumab or everolimus for advanced renal cell carcinoma. N Engl J Med. 2021;384:1289-1300. doi:10.1056/nejmoa2035716
- Kirino S, Tsuchiya K, Kurosaki M, et al. Relative dose intensity over the first four weeks of lenvatinib therapy is a factor of favorable response and overall survival in patients with unresectable hepatocellular carcinoma. PloS One. 2020;15:e0231828. doi:10.1371/journal.pone.0231828
- Ishihara H, Takagi T, Kondo T, et al. Decreased relative dose intensity during the early phase of treatment impacts the therapeutic efficacy of sunitinib in metastatic renal cell carcinoma. Jpn J Clin Oncol. 2018;48:667-672. doi:10.1093/jjco/hyy078
- Alghamdi MA, Amaro CP, Lee-Ying R, et al. Effect of sorafenib starting dose and dose intensity on survival in patients with hepatocellular carcinoma: results from a Canadian Multicenter Database. Cancer Med. 2020;9:4918-4928. doi:10.1002/cam4.3228
- Motzer RJ, Rini BI, Bukowski RM, et al. Sunitinib in patients with metastatic renal cell carcinoma. JAMA. 2006;295:2516-2524. doi:10.1001/jama.295.21.2516
- Miller AA, Murry DJ, Owzar K, et al. Phase I and pharmacokinetic study of sorafenib in patients with hepatic or renal dysfunction: CALGB 60301. J Clin Oncol. 2009;27:1800-1805. doi:10.1200/jco.2008.20.0931
- Boucai L, Zafereo M, Cabanillas ME. Thyroid cancer: a review. JAMA. 2024;331:425-435. doi:10.1001/jama.2023.26348
- Amin N, Anwar J, Sulaiman A, et al. Hepatocellular carcinoma: a comprehensive review. Diseases. 2025;13:207. doi:10.3390/diseases13070207
- Bekaii-Saab TS, Ou FS, Ahn DH, et al. Regorafenib dose-optimisation in patients with refractory metastatic colorectal cancer (ReDOS): a randomised, multicentre, open-label, phase 2 study. Lancet Oncol. 2019;20:1070-1082. doi:10.1016/s1470-2045(19)30272-4
- Frequently asked questions. VA National Oncology Program. 2025. Accessed December 15, 2025. https://www.cancer.va.gov/CANCER/faqs.html
- Torez L. Reigniting the cancer moonshot to beat cancer. VA News. April 20, 2023. Accessed April 6, 2026. https://news.va.gov/118378/reigniting-the-cancer-moonshot-to-beat-cancer
- Shah NN, Casella E, Capozzi D, et al. Improving the safety of oral chemotherapy at an academic medical center. J Oncol Pract. 2016;12:e71-e76. doi:10.1200/JOP.2015.007260
- Hussaarts KGAM, Veerman GDM, Jansman FGA, et al. Clinically relevant drug interactions with multikinase inhibitors: a review. Ther Adv Med Oncol. 2019;11:1758835918818347. doi:10.1177/1758835918818347
- Shyam Sunder S, Sharma UC, Pokharel S. Adverse effects of tyrosine kinase inhibitors in cancer therapy: pathophysiology, mechanisms and clinical management. Signal Transduct Target Ther. 2023;8:262. doi:10.1038/s41392-023-01469-6
- Thomson RJ, Moshirfar M, Ronquillo Y. Tyrosine kinase inhibitors. In: StatPearls [Internet]. StatPearls Publishing; updated July 18, 2023. Accessed December 15, 2025. https://www.ncbi.nlm.nih.gov/books/NBK563322/
- Project Optimus. US Food and Drug Administration. Updated December 6, 2024. Accessed December 15, 2025. https://www.fda.gov/about-fda/oncology-center-excellence/project-optimus
- Optimizing the dosage of human prescription drugs and biological products for the treatment of oncologic diseases: Guidance for Industry. Docket number FDA-2022-D-2827. US Food and Drug Administration. August 2024. Accessed December 15, 2025. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/optimizing-dosage-human-prescription-drugs-and-biological-products-treatment-oncologic-diseases
- Schnadig ID, Hutson TE, Chung H, et al. Dosing patterns, toxicity, and outcomes in patients treated with first-line sunitinib for advanced renal cell carcinoma in community-based practices. Clin Genitourin Cancer. 2014;12:413-421. doi:10.1016/j.clgc.2014.06.015
- Motzer RJ, Hutson TE, Tomczak P, et al. Sunitinib versus interferon alfa in metastatic renal-cell carcinoma. N Engl J Med. 2007;356:115-124. doi:10.1056/nejmoa065044
- Hawn C, Bansal D. Relative dose intensity in oncology trials: a discussion of two approaches. PharmaSUG. 2024. Accessed April 6, 2026. https://pharmasug.org/proceedings/2024/ST/PharmaSUG-2024-ST-297.pdf
- George S, Merriam P, Maki RG, et al. Multicenter phase II trial of sunitinib in the treatment of nongastrointestinal stromal tumor sarcomas. J Clin Oncol. 2009;27:3154-3160. doi:10.1200/jco.2008.20.9890
- George S, Blay JY, Casali PG, et al. Clinical evaluation of continuous daily dosing of sunitinib malate in patients with advanced gastrointestinal stromal tumour after imatinib failure. Eur J Cancer. 2009;45:1959-1968. doi:10.1016/j.ejca.2009.02.011
- Denduluri N, Patt DA, Wang Y, et al. Dose delays, dose reductions, and relative dose intensity in patients with cancer who received adjuvant or neoadjuvant chemotherapy in community oncology practices. J Natl Compr Canc Netw. 2015;13:1383-1393. doi:10.6004/jnccn.2015.0166
- Motzer RJ, Penkov K, Haanen J, et al. Avelumab plus axitinib versus sunitinib for advanced renal-cell carcinoma. N Engl J Med. 2019;380:1103-1115. doi:10.1056/nejmoa1816047
- Motzer R, Alekseev B, Rha SY, et al. Lenvatinib plus pembrolizumab or everolimus for advanced renal cell carcinoma. N Engl J Med. 2021;384:1289-1300. doi:10.1056/nejmoa2035716
- Kirino S, Tsuchiya K, Kurosaki M, et al. Relative dose intensity over the first four weeks of lenvatinib therapy is a factor of favorable response and overall survival in patients with unresectable hepatocellular carcinoma. PloS One. 2020;15:e0231828. doi:10.1371/journal.pone.0231828
- Ishihara H, Takagi T, Kondo T, et al. Decreased relative dose intensity during the early phase of treatment impacts the therapeutic efficacy of sunitinib in metastatic renal cell carcinoma. Jpn J Clin Oncol. 2018;48:667-672. doi:10.1093/jjco/hyy078
- Alghamdi MA, Amaro CP, Lee-Ying R, et al. Effect of sorafenib starting dose and dose intensity on survival in patients with hepatocellular carcinoma: results from a Canadian Multicenter Database. Cancer Med. 2020;9:4918-4928. doi:10.1002/cam4.3228
- Motzer RJ, Rini BI, Bukowski RM, et al. Sunitinib in patients with metastatic renal cell carcinoma. JAMA. 2006;295:2516-2524. doi:10.1001/jama.295.21.2516
- Miller AA, Murry DJ, Owzar K, et al. Phase I and pharmacokinetic study of sorafenib in patients with hepatic or renal dysfunction: CALGB 60301. J Clin Oncol. 2009;27:1800-1805. doi:10.1200/jco.2008.20.0931
- Boucai L, Zafereo M, Cabanillas ME. Thyroid cancer: a review. JAMA. 2024;331:425-435. doi:10.1001/jama.2023.26348
- Amin N, Anwar J, Sulaiman A, et al. Hepatocellular carcinoma: a comprehensive review. Diseases. 2025;13:207. doi:10.3390/diseases13070207
- Bekaii-Saab TS, Ou FS, Ahn DH, et al. Regorafenib dose-optimisation in patients with refractory metastatic colorectal cancer (ReDOS): a randomised, multicentre, open-label, phase 2 study. Lancet Oncol. 2019;20:1070-1082. doi:10.1016/s1470-2045(19)30272-4
Investigating Real-World Tolerance and Dose Reductions of Oncology Multikinase Inhibitors in a VA Population
Investigating Real-World Tolerance and Dose Reductions of Oncology Multikinase Inhibitors in a VA Population