User login
Continued Dosing of Oritavancin for Complicated Gram-Positive Infections
Oritavancin is a lipoglycopeptide antibiotic. The US Food and Drug Administration (FDA) approved oritavancin in 2014 for adults with acute bacterial skin and skin structure infections (ABSSSI).1 The antibiotic is currently FDA approved for infections caused by Gram-positive organisms, including methicillin-resistant and methicillinsusceptible Staphylococcus aureus (MRSA, MSSA), a variety of Streptococcus species, and vancomycin-susceptible Enterococcus faecalis (VSE). Oritavancin demonstrates concentrationdependent bactericidal activity and has a half-life of 245 hours. This half-life allows for treatment of ABSSSI with a single 1,200 mg IV dose, which has been shown to be noninferior to vancomycin dosed twice daily for 7 to 10 days.1-3
Proposal for Expanded Uses
Although the approved indication for oritavancin is narrow, in vitro studies have shown that oritavancin also has activity against vancomycin-resistant enterococci (VRE), and rabbit studies have demonstrated its excellent bone penetration.4,5 These findings have raised the question of whether oritavancin can be safely and effectively used for infections such as endocarditis, osteomyelitis, and bacteremia, which are often caused by invasive Grampositive organisms. These types of invasive infections, particularly when MRSA is implicated, generally require IV antibiotic therapy for several weeks, often with vancomycin.6
To avoid long hospital stays solely for antibiotic administration, health care practitioners will often use outpatient parenteral antimicrobial therapy (OPAT). However, using OPAT presents many challenges due to the need for frequent dosing, the risk of peripheral or central-line infections, and therapeutic drug monitoring when using vancomycin; additionally, administration and line care oftentimes require caregiver support, which may not be present for all patients.7 Concerns also have been raised regarding the use of OPAT in patients with a history of IV drug use due to the potential increased risk of line infections or line abuse. Few studies have explored OPAT in this population, and the Infectious Diseases Society of America OPAT guidelines recommend that the decision to use OPAT should be made on a case-by-case basis.7 Thus, patients who are deemed inappropriate for OPAT oftentimes remain hospitalized or reside briefly in nursing facilities solely for antibiotic administration
Oritavancin’s long half-life and potent activity against Gram-positive organisms has led to increased interest in off-label use of infrequent dosing intervals, such as weekly, to treat complicated and invasive infections. Weekly rather than daily dosing would allow for less burdensome antibiotic administration regimens and shorter hospital stays especially for patients who are not candidates for OPAT.
Efficacy of Continued Dosing
This proposed weekly dosing pattern, referred to as continued dosing or a multiple-dose regimen, has gained traction in the literature. To date, no randomized controlled trials have been conducted to assess oritavancin’s efficacy in off-label indications or continued dosing, but several case reports and retrospective cohort analyses show promising outcomes.8-16 In an analysis of data from the Clinical and Historic Registry and Orbactiv Medical Evaluation (CHROME) patient registry, 32 patients received multiple doses of oritavancin for complicated Gram-positive infections with a 93.8% overall clinical success rate, including success rates of 90.9% (10/11) for general bone and joint infections and 87.5% (7/8) for patients diagnosed specifically with osteomyelitis.8
Patients received between 2 and 10 doses of 1,200 mg IV given every 6 to 14 days. Johnson and colleagues report using oritavancin 1,200 mg IV every other day for 3 doses followed by 1,200 mg IV once weekly for a patient with daptomycin- and vancomycin-resistant Enterococcus endocarditis, resulting in negative blood cultures while on therapy.9 However, source control via valve replacement and postoperative oritavancin 1,200 mg IV twice weekly for 10 weeks was required to fully clear the infection.
Schulz and colleagues published a retrospective cohort analysis of 17 patients who received multiple doses of oritavancin for complicated bacterial infections, including osteomyelitis, pneumonia, and bacteremia.10 They reported 100% of patients were either successfully cured or had demonstrable improvements in their infections by using a 1,200 mg IV loading dose followed by 800 mg IV if the second dose was given within 7 days or 1,200 mg IV if the second dose was given more than 10 days later. Patients received between 2 and 18 total doses, with 6 out of 17 (35%) receiving only 2 doses. One patient who received 18 doses was an outlier, as her treatment goal was palliative suppression due to an infected endovascular graft that could not be removed.
In a published case series, 1 of 10 patients receiving oritavancin for invasive Grampositive infections received multiple doses of oritavancin for an MSSA deep tissue infection.11 The 3 total doses (strength not reported) were separated by 19 days and 14 days and resulted in cure. Several case reports and a retrospective chart review study specifically show the effectiveness of oritavancin for osteomyelitis caused by MSSA, MRSA, and VRE.12-16 However, dosing strategies varied widely after the initial 1,200 mg IV loading dose.
Drug Interactions, Safety, and Tolerability
Oritavancin has minimal drug-drug interactions, the most notable being with anticoagulants. 1 Use of IV heparin within 120 hours of oritavancin administration can falsely elevate activated partial thromboplastin time (aPTT) levels; therefore, heparin should not be monitored with aPTT during this period. Oritavancin also can artificially prolong international normalized ratio (INR) values for up to 12 hours, and dose adjustments based on INRs during this window are not recommended. Of note, factor Xa laboratory monitoring is unaffected by oritavancin, as it does not depend on phospholipid reagents as do aPTT and INR measurements.
Oritavancin has been shown to be well tolerated when dosed according to both the package insert and continued dosing strategies. The most common adverse effects (AEs) (≥ 3%), occurring at similar rates to vancomycin, are nausea, vomiting, diarrhea, headache, and limb and subcutaneous abscesses.1 Infusion reactions also have been reported, although they are usually reversible on slowing or stopping the infusion. It is worth noting that the use of oritavancin for osteomyelitis is not recommended in the product labeling, as an increased rate of osteomyelitis was observed in the oritavancin vs IV vancomycin groups for the treatment of patients with acute bacterial skin and skin structure infection (SOLO) trials (0.6% in oritavancin group vs 0.1% in vancomycin group, statistical significance not reported).17 However, it was postulated that these osteomyelitis cases were likely present, yet not recognized, at baseline and were not the result of administering oritavancin. This conclusion is further corroborated by previously presented research demonstrating successful cure of osteomyelitis with continued dosing strategies.12-16
Many patients receiving multiple doses of oritavancin did not experience AEs or laboratory abnormalities.13,15 Four of 17 patients (24%) in one retrospective review experienced AEs, including infusion reactions, anemia, and leukopenia; all were reversible on discontinuation of oritavancin, and contributions of other antibiotics in some cases could not be ruled out.10 One patient experienced taste disturbance for several hours after each infusion, and a second had documented hearing loss after 3 doses of oritavancin in a 33-day period, though she had received 6 weeks of IV vancomycin prior to oritavancin.11,12 A patient treated for daptomycin- and vancomycinresistant Enterococcus faecium prosthetic valve endocarditis experienced nausea, anorexia, and minor liver function test (LFT) abnormalities after cumulative oritavancin exposure over 18 weeks.9 On discontinuation of the drug, nausea and anorexia improved, and LFTs normalized 11 months later. Overall, AEs reported with continued dosing of oritavancin have been minimal and largely reversible, mimicking the AEs in the product labeling for traditional dosing. This suggests that using a continued dosing strategy may not result in worse or more frequent AEs, though randomized controlled trials are needed to fully ascertain these preliminary findings.
Conclusions
The literature supporting the use of oritavancin beyond single-dose administration for ABSSSI is growing. Continued dosing regimens have been well tolerated and have resulted in clinical cure for many patients with barriers to first-line treatment and complicated or invasive infections. While randomized controlled trials are needed to concretely demonstrate the efficacy and safety of continued dosing of oritavancin, it may fill an important treatment niche in this era of growing antibiotic resistance and increasing complexity of patient cases.
1. Orbactiv [package insert]. Parsippany, NJ: The Medicines Company; 2019.
2. Corey GR, Kabler H, Mehra P, et al. Single-dose oritavancin in the treatment of acute bacterial skin infections. N Engl J Med. 2014;370(23):2180-2190. doi:10.1056/NEJMoa1310422
3. Corey GR, Good S, Jiang H, et al. Single-dose oritavancin versus 7-10 days of vancomycin in the treatment of gram-positive acute bacterial skin and skin structure infections: the SOLO II noninferiority study. Clin Infect Dis. 2015;60(2):254-262. doi:10.1093/cid/ciu778
4. Sweeney D, Stoneburner A, Shinabarger DL, et al. Comparative in vitro activity of oritavancin and other agents against vancomycin-susceptible and -resistant enterococci. J Antimicrob Chemother. 2017;72(2):622-624. doi.10.1093/jac/dkw451
5. Lehoux D, Ostiguy V, Vadieux C, et al. Oritavancin pharmacokinetics and bone penetration in rabbits. Antimicrob Agents Chemother. 2015;59(10):6501-6505. doi:10.1128/AAC.00981-15
6. Liu C, Bayer A, Cosgrove SE, et al. Clinical practice guidelines by the Infectious Diseases Society of America for the treatment of methicillin-resistant Staphylococcus aureus infections in adults and children. Clin Infect Dis. 2011;52(3):e18-e55. doi:10.1093/cid/ciq146
7. Norris AH, Shrestha NK, Allison GM, et al. 2018 Infectious Diseases Society of America clinical practice guideline for the management of outpatient parenteral antimicrobial therapy. Clin Infect Dis. 2019;68(1):e1-e35. doi:10.1093/cid/ciy745
8. Redell M, Seirra-Hoffman M, Assi Maha, et al. The CHROME study, a real-world experience of single- and multiple-dose oritavancin for treatment of gram-positive infections. Open Forum Infect Dis. 2019;6(11):ofz479. doi:10.1093/ofid/ofz479
9. Johnson JA, Feeney ER, Kubiak DW, Corey GR. Prolonged use of oritavancin for vancomycin-resistant Enterococcus faecium prosthetic valve endocarditis. Open Forum Infect Dis. 2015;2(4):ofv156. doi:10.1093/ofid/ofv156
10. Schulz LT, Dworkin E, Dela-Pena J, Rose WE. Multipledose oritavancin evaluation in a retrospective cohort of patients with complicated infections. Pharmacotherapy. 2018;38(1):152-159. doi:10.1002/phar.2057
11. Stewart CL, Turner MS, Frens JJ, Snider CB, Smith JR. Real-world experience with oritavancin therapy in invasive gram-positive infections. Infect Dis Ther. 2017;6(2):277-289. doi:10.1007/s40121-017-0156-z
12. Delaportas DJ, Estrada SJ, Darmelio M. Successful treatment of methicillin susceptible Staphylococcus aureus osteomyelitis with oritavancin. Pharmacotherapy. 2017;37(8):e90-e92. doi:10.1002/phar.1957
13. Chastain DB, Davis A. Treatment of chronic osteomyelitis with multidose oritavancin: a case series and literature review. Int J Antimicrob Agents. 2019;53(4):429-434. doi:10.1016/j.ijantimicag.2018.11.023
14. Dahesh S, Wong B, Nizet V, Sakoulas G, Tran TT, Aitken SL. Treatment of multidrug-resistant vancomycinresistant Enterococcus faecium hardware-associated vertebral osteomyelitis with oritavancin plus ampicillin. Antimicrob Agents Chemother. 2019;63(7):e02622-18. doi:10.1128/AAC.02622-18
15. Foster RA, Philavong KP, Weissman S, Tang X, Bookstaver PB. Oritavancin for the treatment of daptomycin nonsusceptible vancomycin-resistant Enterococci osteomyelitis. Infect Dis Clin Pract. 2018;26(2):97-99. doi:10.1097/IPC.0000000000000517
16. Ruggero M, Ziegler M, Tebas P, Binkley A, Kelly B. Successful treatment of methicillin-resistant Staphylococcus aureus vertebral osteomyelitis with outpatient oritavancin therapy. Infect Dis Clin Pract. 2018;26(3):141-144. doi:10.1097/IPC.0000000000000599
17. Corey GR, Loutit J, Moeck G, et al. Single intravenous dose of oritavancin for treatment of acute skin and skin structure infections caused by gram-positive bacteria: summary of safety analysis from the phase 3 SOLO studies. Antimicrob Agents Chemother. 2018;62(4):e01919- 17. doi:10.1128/AAC.01919-17
Oritavancin is a lipoglycopeptide antibiotic. The US Food and Drug Administration (FDA) approved oritavancin in 2014 for adults with acute bacterial skin and skin structure infections (ABSSSI).1 The antibiotic is currently FDA approved for infections caused by Gram-positive organisms, including methicillin-resistant and methicillinsusceptible Staphylococcus aureus (MRSA, MSSA), a variety of Streptococcus species, and vancomycin-susceptible Enterococcus faecalis (VSE). Oritavancin demonstrates concentrationdependent bactericidal activity and has a half-life of 245 hours. This half-life allows for treatment of ABSSSI with a single 1,200 mg IV dose, which has been shown to be noninferior to vancomycin dosed twice daily for 7 to 10 days.1-3
Proposal for Expanded Uses
Although the approved indication for oritavancin is narrow, in vitro studies have shown that oritavancin also has activity against vancomycin-resistant enterococci (VRE), and rabbit studies have demonstrated its excellent bone penetration.4,5 These findings have raised the question of whether oritavancin can be safely and effectively used for infections such as endocarditis, osteomyelitis, and bacteremia, which are often caused by invasive Grampositive organisms. These types of invasive infections, particularly when MRSA is implicated, generally require IV antibiotic therapy for several weeks, often with vancomycin.6
To avoid long hospital stays solely for antibiotic administration, health care practitioners will often use outpatient parenteral antimicrobial therapy (OPAT). However, using OPAT presents many challenges due to the need for frequent dosing, the risk of peripheral or central-line infections, and therapeutic drug monitoring when using vancomycin; additionally, administration and line care oftentimes require caregiver support, which may not be present for all patients.7 Concerns also have been raised regarding the use of OPAT in patients with a history of IV drug use due to the potential increased risk of line infections or line abuse. Few studies have explored OPAT in this population, and the Infectious Diseases Society of America OPAT guidelines recommend that the decision to use OPAT should be made on a case-by-case basis.7 Thus, patients who are deemed inappropriate for OPAT oftentimes remain hospitalized or reside briefly in nursing facilities solely for antibiotic administration
Oritavancin’s long half-life and potent activity against Gram-positive organisms has led to increased interest in off-label use of infrequent dosing intervals, such as weekly, to treat complicated and invasive infections. Weekly rather than daily dosing would allow for less burdensome antibiotic administration regimens and shorter hospital stays especially for patients who are not candidates for OPAT.
Efficacy of Continued Dosing
This proposed weekly dosing pattern, referred to as continued dosing or a multiple-dose regimen, has gained traction in the literature. To date, no randomized controlled trials have been conducted to assess oritavancin’s efficacy in off-label indications or continued dosing, but several case reports and retrospective cohort analyses show promising outcomes.8-16 In an analysis of data from the Clinical and Historic Registry and Orbactiv Medical Evaluation (CHROME) patient registry, 32 patients received multiple doses of oritavancin for complicated Gram-positive infections with a 93.8% overall clinical success rate, including success rates of 90.9% (10/11) for general bone and joint infections and 87.5% (7/8) for patients diagnosed specifically with osteomyelitis.8
Patients received between 2 and 10 doses of 1,200 mg IV given every 6 to 14 days. Johnson and colleagues report using oritavancin 1,200 mg IV every other day for 3 doses followed by 1,200 mg IV once weekly for a patient with daptomycin- and vancomycin-resistant Enterococcus endocarditis, resulting in negative blood cultures while on therapy.9 However, source control via valve replacement and postoperative oritavancin 1,200 mg IV twice weekly for 10 weeks was required to fully clear the infection.
Schulz and colleagues published a retrospective cohort analysis of 17 patients who received multiple doses of oritavancin for complicated bacterial infections, including osteomyelitis, pneumonia, and bacteremia.10 They reported 100% of patients were either successfully cured or had demonstrable improvements in their infections by using a 1,200 mg IV loading dose followed by 800 mg IV if the second dose was given within 7 days or 1,200 mg IV if the second dose was given more than 10 days later. Patients received between 2 and 18 total doses, with 6 out of 17 (35%) receiving only 2 doses. One patient who received 18 doses was an outlier, as her treatment goal was palliative suppression due to an infected endovascular graft that could not be removed.
In a published case series, 1 of 10 patients receiving oritavancin for invasive Grampositive infections received multiple doses of oritavancin for an MSSA deep tissue infection.11 The 3 total doses (strength not reported) were separated by 19 days and 14 days and resulted in cure. Several case reports and a retrospective chart review study specifically show the effectiveness of oritavancin for osteomyelitis caused by MSSA, MRSA, and VRE.12-16 However, dosing strategies varied widely after the initial 1,200 mg IV loading dose.
Drug Interactions, Safety, and Tolerability
Oritavancin has minimal drug-drug interactions, the most notable being with anticoagulants. 1 Use of IV heparin within 120 hours of oritavancin administration can falsely elevate activated partial thromboplastin time (aPTT) levels; therefore, heparin should not be monitored with aPTT during this period. Oritavancin also can artificially prolong international normalized ratio (INR) values for up to 12 hours, and dose adjustments based on INRs during this window are not recommended. Of note, factor Xa laboratory monitoring is unaffected by oritavancin, as it does not depend on phospholipid reagents as do aPTT and INR measurements.
Oritavancin has been shown to be well tolerated when dosed according to both the package insert and continued dosing strategies. The most common adverse effects (AEs) (≥ 3%), occurring at similar rates to vancomycin, are nausea, vomiting, diarrhea, headache, and limb and subcutaneous abscesses.1 Infusion reactions also have been reported, although they are usually reversible on slowing or stopping the infusion. It is worth noting that the use of oritavancin for osteomyelitis is not recommended in the product labeling, as an increased rate of osteomyelitis was observed in the oritavancin vs IV vancomycin groups for the treatment of patients with acute bacterial skin and skin structure infection (SOLO) trials (0.6% in oritavancin group vs 0.1% in vancomycin group, statistical significance not reported).17 However, it was postulated that these osteomyelitis cases were likely present, yet not recognized, at baseline and were not the result of administering oritavancin. This conclusion is further corroborated by previously presented research demonstrating successful cure of osteomyelitis with continued dosing strategies.12-16
Many patients receiving multiple doses of oritavancin did not experience AEs or laboratory abnormalities.13,15 Four of 17 patients (24%) in one retrospective review experienced AEs, including infusion reactions, anemia, and leukopenia; all were reversible on discontinuation of oritavancin, and contributions of other antibiotics in some cases could not be ruled out.10 One patient experienced taste disturbance for several hours after each infusion, and a second had documented hearing loss after 3 doses of oritavancin in a 33-day period, though she had received 6 weeks of IV vancomycin prior to oritavancin.11,12 A patient treated for daptomycin- and vancomycinresistant Enterococcus faecium prosthetic valve endocarditis experienced nausea, anorexia, and minor liver function test (LFT) abnormalities after cumulative oritavancin exposure over 18 weeks.9 On discontinuation of the drug, nausea and anorexia improved, and LFTs normalized 11 months later. Overall, AEs reported with continued dosing of oritavancin have been minimal and largely reversible, mimicking the AEs in the product labeling for traditional dosing. This suggests that using a continued dosing strategy may not result in worse or more frequent AEs, though randomized controlled trials are needed to fully ascertain these preliminary findings.
Conclusions
The literature supporting the use of oritavancin beyond single-dose administration for ABSSSI is growing. Continued dosing regimens have been well tolerated and have resulted in clinical cure for many patients with barriers to first-line treatment and complicated or invasive infections. While randomized controlled trials are needed to concretely demonstrate the efficacy and safety of continued dosing of oritavancin, it may fill an important treatment niche in this era of growing antibiotic resistance and increasing complexity of patient cases.
Oritavancin is a lipoglycopeptide antibiotic. The US Food and Drug Administration (FDA) approved oritavancin in 2014 for adults with acute bacterial skin and skin structure infections (ABSSSI).1 The antibiotic is currently FDA approved for infections caused by Gram-positive organisms, including methicillin-resistant and methicillinsusceptible Staphylococcus aureus (MRSA, MSSA), a variety of Streptococcus species, and vancomycin-susceptible Enterococcus faecalis (VSE). Oritavancin demonstrates concentrationdependent bactericidal activity and has a half-life of 245 hours. This half-life allows for treatment of ABSSSI with a single 1,200 mg IV dose, which has been shown to be noninferior to vancomycin dosed twice daily for 7 to 10 days.1-3
Proposal for Expanded Uses
Although the approved indication for oritavancin is narrow, in vitro studies have shown that oritavancin also has activity against vancomycin-resistant enterococci (VRE), and rabbit studies have demonstrated its excellent bone penetration.4,5 These findings have raised the question of whether oritavancin can be safely and effectively used for infections such as endocarditis, osteomyelitis, and bacteremia, which are often caused by invasive Grampositive organisms. These types of invasive infections, particularly when MRSA is implicated, generally require IV antibiotic therapy for several weeks, often with vancomycin.6
To avoid long hospital stays solely for antibiotic administration, health care practitioners will often use outpatient parenteral antimicrobial therapy (OPAT). However, using OPAT presents many challenges due to the need for frequent dosing, the risk of peripheral or central-line infections, and therapeutic drug monitoring when using vancomycin; additionally, administration and line care oftentimes require caregiver support, which may not be present for all patients.7 Concerns also have been raised regarding the use of OPAT in patients with a history of IV drug use due to the potential increased risk of line infections or line abuse. Few studies have explored OPAT in this population, and the Infectious Diseases Society of America OPAT guidelines recommend that the decision to use OPAT should be made on a case-by-case basis.7 Thus, patients who are deemed inappropriate for OPAT oftentimes remain hospitalized or reside briefly in nursing facilities solely for antibiotic administration
Oritavancin’s long half-life and potent activity against Gram-positive organisms has led to increased interest in off-label use of infrequent dosing intervals, such as weekly, to treat complicated and invasive infections. Weekly rather than daily dosing would allow for less burdensome antibiotic administration regimens and shorter hospital stays especially for patients who are not candidates for OPAT.
Efficacy of Continued Dosing
This proposed weekly dosing pattern, referred to as continued dosing or a multiple-dose regimen, has gained traction in the literature. To date, no randomized controlled trials have been conducted to assess oritavancin’s efficacy in off-label indications or continued dosing, but several case reports and retrospective cohort analyses show promising outcomes.8-16 In an analysis of data from the Clinical and Historic Registry and Orbactiv Medical Evaluation (CHROME) patient registry, 32 patients received multiple doses of oritavancin for complicated Gram-positive infections with a 93.8% overall clinical success rate, including success rates of 90.9% (10/11) for general bone and joint infections and 87.5% (7/8) for patients diagnosed specifically with osteomyelitis.8
Patients received between 2 and 10 doses of 1,200 mg IV given every 6 to 14 days. Johnson and colleagues report using oritavancin 1,200 mg IV every other day for 3 doses followed by 1,200 mg IV once weekly for a patient with daptomycin- and vancomycin-resistant Enterococcus endocarditis, resulting in negative blood cultures while on therapy.9 However, source control via valve replacement and postoperative oritavancin 1,200 mg IV twice weekly for 10 weeks was required to fully clear the infection.
Schulz and colleagues published a retrospective cohort analysis of 17 patients who received multiple doses of oritavancin for complicated bacterial infections, including osteomyelitis, pneumonia, and bacteremia.10 They reported 100% of patients were either successfully cured or had demonstrable improvements in their infections by using a 1,200 mg IV loading dose followed by 800 mg IV if the second dose was given within 7 days or 1,200 mg IV if the second dose was given more than 10 days later. Patients received between 2 and 18 total doses, with 6 out of 17 (35%) receiving only 2 doses. One patient who received 18 doses was an outlier, as her treatment goal was palliative suppression due to an infected endovascular graft that could not be removed.
In a published case series, 1 of 10 patients receiving oritavancin for invasive Grampositive infections received multiple doses of oritavancin for an MSSA deep tissue infection.11 The 3 total doses (strength not reported) were separated by 19 days and 14 days and resulted in cure. Several case reports and a retrospective chart review study specifically show the effectiveness of oritavancin for osteomyelitis caused by MSSA, MRSA, and VRE.12-16 However, dosing strategies varied widely after the initial 1,200 mg IV loading dose.
Drug Interactions, Safety, and Tolerability
Oritavancin has minimal drug-drug interactions, the most notable being with anticoagulants. 1 Use of IV heparin within 120 hours of oritavancin administration can falsely elevate activated partial thromboplastin time (aPTT) levels; therefore, heparin should not be monitored with aPTT during this period. Oritavancin also can artificially prolong international normalized ratio (INR) values for up to 12 hours, and dose adjustments based on INRs during this window are not recommended. Of note, factor Xa laboratory monitoring is unaffected by oritavancin, as it does not depend on phospholipid reagents as do aPTT and INR measurements.
Oritavancin has been shown to be well tolerated when dosed according to both the package insert and continued dosing strategies. The most common adverse effects (AEs) (≥ 3%), occurring at similar rates to vancomycin, are nausea, vomiting, diarrhea, headache, and limb and subcutaneous abscesses.1 Infusion reactions also have been reported, although they are usually reversible on slowing or stopping the infusion. It is worth noting that the use of oritavancin for osteomyelitis is not recommended in the product labeling, as an increased rate of osteomyelitis was observed in the oritavancin vs IV vancomycin groups for the treatment of patients with acute bacterial skin and skin structure infection (SOLO) trials (0.6% in oritavancin group vs 0.1% in vancomycin group, statistical significance not reported).17 However, it was postulated that these osteomyelitis cases were likely present, yet not recognized, at baseline and were not the result of administering oritavancin. This conclusion is further corroborated by previously presented research demonstrating successful cure of osteomyelitis with continued dosing strategies.12-16
Many patients receiving multiple doses of oritavancin did not experience AEs or laboratory abnormalities.13,15 Four of 17 patients (24%) in one retrospective review experienced AEs, including infusion reactions, anemia, and leukopenia; all were reversible on discontinuation of oritavancin, and contributions of other antibiotics in some cases could not be ruled out.10 One patient experienced taste disturbance for several hours after each infusion, and a second had documented hearing loss after 3 doses of oritavancin in a 33-day period, though she had received 6 weeks of IV vancomycin prior to oritavancin.11,12 A patient treated for daptomycin- and vancomycinresistant Enterococcus faecium prosthetic valve endocarditis experienced nausea, anorexia, and minor liver function test (LFT) abnormalities after cumulative oritavancin exposure over 18 weeks.9 On discontinuation of the drug, nausea and anorexia improved, and LFTs normalized 11 months later. Overall, AEs reported with continued dosing of oritavancin have been minimal and largely reversible, mimicking the AEs in the product labeling for traditional dosing. This suggests that using a continued dosing strategy may not result in worse or more frequent AEs, though randomized controlled trials are needed to fully ascertain these preliminary findings.
Conclusions
The literature supporting the use of oritavancin beyond single-dose administration for ABSSSI is growing. Continued dosing regimens have been well tolerated and have resulted in clinical cure for many patients with barriers to first-line treatment and complicated or invasive infections. While randomized controlled trials are needed to concretely demonstrate the efficacy and safety of continued dosing of oritavancin, it may fill an important treatment niche in this era of growing antibiotic resistance and increasing complexity of patient cases.
1. Orbactiv [package insert]. Parsippany, NJ: The Medicines Company; 2019.
2. Corey GR, Kabler H, Mehra P, et al. Single-dose oritavancin in the treatment of acute bacterial skin infections. N Engl J Med. 2014;370(23):2180-2190. doi:10.1056/NEJMoa1310422
3. Corey GR, Good S, Jiang H, et al. Single-dose oritavancin versus 7-10 days of vancomycin in the treatment of gram-positive acute bacterial skin and skin structure infections: the SOLO II noninferiority study. Clin Infect Dis. 2015;60(2):254-262. doi:10.1093/cid/ciu778
4. Sweeney D, Stoneburner A, Shinabarger DL, et al. Comparative in vitro activity of oritavancin and other agents against vancomycin-susceptible and -resistant enterococci. J Antimicrob Chemother. 2017;72(2):622-624. doi.10.1093/jac/dkw451
5. Lehoux D, Ostiguy V, Vadieux C, et al. Oritavancin pharmacokinetics and bone penetration in rabbits. Antimicrob Agents Chemother. 2015;59(10):6501-6505. doi:10.1128/AAC.00981-15
6. Liu C, Bayer A, Cosgrove SE, et al. Clinical practice guidelines by the Infectious Diseases Society of America for the treatment of methicillin-resistant Staphylococcus aureus infections in adults and children. Clin Infect Dis. 2011;52(3):e18-e55. doi:10.1093/cid/ciq146
7. Norris AH, Shrestha NK, Allison GM, et al. 2018 Infectious Diseases Society of America clinical practice guideline for the management of outpatient parenteral antimicrobial therapy. Clin Infect Dis. 2019;68(1):e1-e35. doi:10.1093/cid/ciy745
8. Redell M, Seirra-Hoffman M, Assi Maha, et al. The CHROME study, a real-world experience of single- and multiple-dose oritavancin for treatment of gram-positive infections. Open Forum Infect Dis. 2019;6(11):ofz479. doi:10.1093/ofid/ofz479
9. Johnson JA, Feeney ER, Kubiak DW, Corey GR. Prolonged use of oritavancin for vancomycin-resistant Enterococcus faecium prosthetic valve endocarditis. Open Forum Infect Dis. 2015;2(4):ofv156. doi:10.1093/ofid/ofv156
10. Schulz LT, Dworkin E, Dela-Pena J, Rose WE. Multipledose oritavancin evaluation in a retrospective cohort of patients with complicated infections. Pharmacotherapy. 2018;38(1):152-159. doi:10.1002/phar.2057
11. Stewart CL, Turner MS, Frens JJ, Snider CB, Smith JR. Real-world experience with oritavancin therapy in invasive gram-positive infections. Infect Dis Ther. 2017;6(2):277-289. doi:10.1007/s40121-017-0156-z
12. Delaportas DJ, Estrada SJ, Darmelio M. Successful treatment of methicillin susceptible Staphylococcus aureus osteomyelitis with oritavancin. Pharmacotherapy. 2017;37(8):e90-e92. doi:10.1002/phar.1957
13. Chastain DB, Davis A. Treatment of chronic osteomyelitis with multidose oritavancin: a case series and literature review. Int J Antimicrob Agents. 2019;53(4):429-434. doi:10.1016/j.ijantimicag.2018.11.023
14. Dahesh S, Wong B, Nizet V, Sakoulas G, Tran TT, Aitken SL. Treatment of multidrug-resistant vancomycinresistant Enterococcus faecium hardware-associated vertebral osteomyelitis with oritavancin plus ampicillin. Antimicrob Agents Chemother. 2019;63(7):e02622-18. doi:10.1128/AAC.02622-18
15. Foster RA, Philavong KP, Weissman S, Tang X, Bookstaver PB. Oritavancin for the treatment of daptomycin nonsusceptible vancomycin-resistant Enterococci osteomyelitis. Infect Dis Clin Pract. 2018;26(2):97-99. doi:10.1097/IPC.0000000000000517
16. Ruggero M, Ziegler M, Tebas P, Binkley A, Kelly B. Successful treatment of methicillin-resistant Staphylococcus aureus vertebral osteomyelitis with outpatient oritavancin therapy. Infect Dis Clin Pract. 2018;26(3):141-144. doi:10.1097/IPC.0000000000000599
17. Corey GR, Loutit J, Moeck G, et al. Single intravenous dose of oritavancin for treatment of acute skin and skin structure infections caused by gram-positive bacteria: summary of safety analysis from the phase 3 SOLO studies. Antimicrob Agents Chemother. 2018;62(4):e01919- 17. doi:10.1128/AAC.01919-17
1. Orbactiv [package insert]. Parsippany, NJ: The Medicines Company; 2019.
2. Corey GR, Kabler H, Mehra P, et al. Single-dose oritavancin in the treatment of acute bacterial skin infections. N Engl J Med. 2014;370(23):2180-2190. doi:10.1056/NEJMoa1310422
3. Corey GR, Good S, Jiang H, et al. Single-dose oritavancin versus 7-10 days of vancomycin in the treatment of gram-positive acute bacterial skin and skin structure infections: the SOLO II noninferiority study. Clin Infect Dis. 2015;60(2):254-262. doi:10.1093/cid/ciu778
4. Sweeney D, Stoneburner A, Shinabarger DL, et al. Comparative in vitro activity of oritavancin and other agents against vancomycin-susceptible and -resistant enterococci. J Antimicrob Chemother. 2017;72(2):622-624. doi.10.1093/jac/dkw451
5. Lehoux D, Ostiguy V, Vadieux C, et al. Oritavancin pharmacokinetics and bone penetration in rabbits. Antimicrob Agents Chemother. 2015;59(10):6501-6505. doi:10.1128/AAC.00981-15
6. Liu C, Bayer A, Cosgrove SE, et al. Clinical practice guidelines by the Infectious Diseases Society of America for the treatment of methicillin-resistant Staphylococcus aureus infections in adults and children. Clin Infect Dis. 2011;52(3):e18-e55. doi:10.1093/cid/ciq146
7. Norris AH, Shrestha NK, Allison GM, et al. 2018 Infectious Diseases Society of America clinical practice guideline for the management of outpatient parenteral antimicrobial therapy. Clin Infect Dis. 2019;68(1):e1-e35. doi:10.1093/cid/ciy745
8. Redell M, Seirra-Hoffman M, Assi Maha, et al. The CHROME study, a real-world experience of single- and multiple-dose oritavancin for treatment of gram-positive infections. Open Forum Infect Dis. 2019;6(11):ofz479. doi:10.1093/ofid/ofz479
9. Johnson JA, Feeney ER, Kubiak DW, Corey GR. Prolonged use of oritavancin for vancomycin-resistant Enterococcus faecium prosthetic valve endocarditis. Open Forum Infect Dis. 2015;2(4):ofv156. doi:10.1093/ofid/ofv156
10. Schulz LT, Dworkin E, Dela-Pena J, Rose WE. Multipledose oritavancin evaluation in a retrospective cohort of patients with complicated infections. Pharmacotherapy. 2018;38(1):152-159. doi:10.1002/phar.2057
11. Stewart CL, Turner MS, Frens JJ, Snider CB, Smith JR. Real-world experience with oritavancin therapy in invasive gram-positive infections. Infect Dis Ther. 2017;6(2):277-289. doi:10.1007/s40121-017-0156-z
12. Delaportas DJ, Estrada SJ, Darmelio M. Successful treatment of methicillin susceptible Staphylococcus aureus osteomyelitis with oritavancin. Pharmacotherapy. 2017;37(8):e90-e92. doi:10.1002/phar.1957
13. Chastain DB, Davis A. Treatment of chronic osteomyelitis with multidose oritavancin: a case series and literature review. Int J Antimicrob Agents. 2019;53(4):429-434. doi:10.1016/j.ijantimicag.2018.11.023
14. Dahesh S, Wong B, Nizet V, Sakoulas G, Tran TT, Aitken SL. Treatment of multidrug-resistant vancomycinresistant Enterococcus faecium hardware-associated vertebral osteomyelitis with oritavancin plus ampicillin. Antimicrob Agents Chemother. 2019;63(7):e02622-18. doi:10.1128/AAC.02622-18
15. Foster RA, Philavong KP, Weissman S, Tang X, Bookstaver PB. Oritavancin for the treatment of daptomycin nonsusceptible vancomycin-resistant Enterococci osteomyelitis. Infect Dis Clin Pract. 2018;26(2):97-99. doi:10.1097/IPC.0000000000000517
16. Ruggero M, Ziegler M, Tebas P, Binkley A, Kelly B. Successful treatment of methicillin-resistant Staphylococcus aureus vertebral osteomyelitis with outpatient oritavancin therapy. Infect Dis Clin Pract. 2018;26(3):141-144. doi:10.1097/IPC.0000000000000599
17. Corey GR, Loutit J, Moeck G, et al. Single intravenous dose of oritavancin for treatment of acute skin and skin structure infections caused by gram-positive bacteria: summary of safety analysis from the phase 3 SOLO studies. Antimicrob Agents Chemother. 2018;62(4):e01919- 17. doi:10.1128/AAC.01919-17
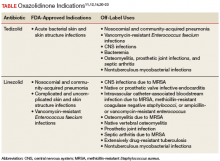
Probiotic Use for the Prevention of Antibiotic- Associated Clostridium difficile Infection
Clostridium difficile (C difficile) is a gram-positive, toxin-producing bacterium that is of increasing concern among health care providers and patients. Infection with C difficile can have manifestations ranging from mild diarrhea to severe toxic megacolon and can result in prolonged hospitalization with severe cases requiring admission to an intensive care unit.1 In 2014, the US was estimated to have more than 600,000 cases of C difficile infection (CDI), previously known as C difficile–associated diarrhea, and more than 44,000 associated deaths. The annual economic cost of CDI is thought to exceed $5 billion.1 According to studies of health care–associated illness, CDI rates are comparable to or have surpassed rates of methicillin-resistant Staphylococcus aureus infection within the US, including at US Department of Veterans Affairs (VA) acute care centers nationwide.2,3
C difficile has been shown to be the causative agent in 10% to 20% of antibiotic-associated diarrhea episodes.4 Colonization of C difficile is uncommon in healthy adults, but colonization rates are as high as 21% in hospitalized patients, with increasing rates proportional to increasing hospital length of stay.5,6 Although not all colonized patients develop clinically significant CDI, those who do may require multiple treatment courses, over months to years, because of the high risk of disease recurrence. An estimated 25% of patients have a single recurrent episode of CDI within 30 days after treatment completion, and 45% of those patients have additional recurrent infections.7,8 Although probiotics do not have an approved US Food and Drug Administration (FDA) indication, these supplements are often used to try to prevent CDI from developing during concomitant antibiotic use. Probiotics are microorganisms with potential health benefits, but the mechanisms of these benefits are not fully understood. Proposed mechanisms include reduced growth of pathogenic bacteria, modulation of the immune system, and support of the intestinal wall barrier.9 The many probiotic formulations currently marketed include Lactobacillus acidophilus (L acidophilus) capsules and various combinations of L acidophilus, Lactobacillus casei, Bifidobacterium lactis, Bifidobacterium longum, Streptococcus thermophilus, and other bacterial strains.
Dosing and Guidelines
Manufacturers’ suggested dosing for their Lactobacillus capsules, tablets, and packets varies from 1 unit daily to 4 units 4 times daily for dietary supplementation; the products’ labeling does not include any information regarding treatment duration.10-13 In addition, there are no published recommendations or product labeling guiding the dosing of probiotics or their duration of use in the primary prevention of CDI.
In 2017, the Infectious Diseases Society of America (IDSA) and the Society for Healthcare Epidemiology of America (SHEA) updated their CDI treatment guidelines.14 As these guidelines indicate that the data on probiotic use in CDI are inadequate, IDSA and SHEA make no recommendation for or against probiotic use in primary prevention of the disease. The guidelines point to several limitations in the literature, including variability in probiotic formulations studied, duration of probiotic administration, definitions of CDI, and duration of study follow-up.
Given the lack of consensus guidelines that clinicians can use when deciding which probiotic dosing and duration are appropriate for a patient for primary prevention of CDI, we evaluated the literature on the topic and summarize their findings here.
Review of Probiotoc Literature
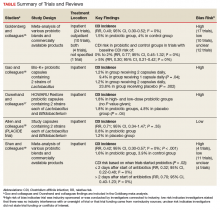
Conflicting data exist about probiotics and their effect on CDI prevention. The literature reviewed was selected based on our assessment of its contribution to the topic and its potential utility to clinicians in determining appropriate probiotic therapies and recommendations. Included in our discussion is a large Cochrane Review of probiotic efficacy, 2 trials of probiotic dosing, the PLACIDE trial, and a systematic review of timely probiotic initiation. All of these studies attempted to determine the effect of probiotics on CDI incidence (Table).
In their 2017 Cochrane Review, Goldenberg and colleagues reviewed 39 trials that investigated the efficacy of probiotics in CDI prevention in 9,955 immunocompetent patients receiving antibiotics.15 The incidence of CDI was significantly lower in patients who received a probiotic than in patients who received placebo or no treatment (1.5% vs 4.0%; relative risk [RR], 0.40; 95% CI, 0.30-0.52; I2 = 0%). It is important to note that trials with a control-group CDI incidence of 0% to 2% (baseline CDI risk) found no statistically significant difference in CDI risk between patients using and not using probiotics (RR, 0.77; 95% CI, 0.45-1.32; I2 = 0%) and that the preceding statistically significant result may have been driven by the inclusion of trials with high baseline CDI risk (> 5%). Trials that enrolled patients who were at this risk level found a statistically significant 70% reduction in CDI risk in those using probiotics (vs no probiotics) while on concomitant antibiotic therapy (RR, 0.30; 95% CI, 0.21-0.42; I2 = 0%).
Probiotic therapy seems to be effective in reducing CDI risk in immunocompetent patients and may be particularly beneficial in patients at higher CDI risk, though Goldenberg and colleagues did not elaborate on what constitutes higher risk and based their conclusion on their control group’s high CDI incidence (> 5%). The most common adverse events (AEs) attributable to probiotics included abdominal cramping, nausea, fever, soft stools, flatulence, and taste disturbance. The review’s findings are limited in that the inclusion of many small trials with high baseline CDI risk likely contributed to a statistically significant result, and 17 of the review’s 39 trials were industry-sponsored or were conducted by investigators with industry associations; another 12 lacked statements about funding or sponsorship.
Two of the trials in the Cochrane Review investigated whether probiotics have a dose effect on CDI prevention. Gao and colleagues randomly assigned 255 hospitalized Asian patients to 3 groups: those receiving placebo, 1 probiotic capsule daily, and 2 probiotic capsules daily.16 Each probiotic capsule contained 50 billion colony-forming units (CFUs) of Lactobacillus. Incidence of CDI was lower in patients taking 2 probiotic capsules daily than in those taking 1 probiotic capsule daily (1.2% vs 9.4%; P = .04) or placebo (1.2% vs 23.8%; P = .002). In the other trial, Ouwehand and colleagues randomly assigned 503 hospitalized Asian patients to 3 groups as well: those receiving placebo, low-dose probiotic (4.17 billion CFUs of Lactobacillus and Bifidobacterium), and high-dose probiotic (17 billion CFUs).17 The incidence of CDI in each probiotic group (low-dose, high-dose) was 1.8%, which was significantly lower than the 4.8% in the placebo group (P = .04).
The Cochrane Review’s largest and most rigorous trial was PLACIDE, a 2013 randomized controlled study of the effect of probiotics on CDI.18 Allen and colleagues randomly assigned 2,981 inpatients (aged ≥ 65 years; exposed to antibiotics within preceding 7 days) to 2 groups: those receiving either 1 probiotic capsule daily, or 1 placebo capsule daily, for 21 days. Results showed no difference in CDI incidence between the probiotic and placebo groups (0.8% vs 1.2%; RR, 0.71; 95% CI, 0.34-1.47; P = .35). Of note, this trial is free of industry sponsorship, is the largest probiotic trial to date, has a control-group baseline CDI rate consistent with the rate in hospital and ambulatory settings in the US, and found a negative result regarding probiotic use in CDI prevention. Findings are limited in that the study allowed for initiating probiotic therapy up to 7 days after the start of antibiotics, and patients were given 1 relatively low-dose capsule daily, which may have contributed to lack of an effect on CDI prevention. No serious AEs were attributed to probiotic use.
In a 2017 systematic meta-analysis of 19 studies, Shen and colleagues investigated whether timely use of probiotics prevented CDI in 6,261 hospitalized patients receiving antibiotics.19 The incidence of CDI was significantly lower in patients receiving vs not receiving probiotics (1.6% vs 3.9%; RR, 0.42; 95% CI, 0.30-0.57; I2 = 0%; P < .001).19 A subgroup analysis was performed to compare studies initiating probiotics within 2 days after the start of antibiotics with studies initiating probiotics more than 2 days after the start. CDI risk was reduced by 68% when probiotics were started within 2 days, vs 30% when started after 2 days (RR, 0.32; 95% CI, 0.22-0.48; I2 = 0% vs RR, 0.70; 95% CI, 0.40-1.23; I2 = 0%; P = .02). Of note, no difference was found in efficacy among the various probiotic formulations, and no significant AEs were noted in any study group.
Trials included in the Cochrane Review used many different probiotic regimens over various durations.15 All these trials continued probiotics for at least the duration of antibiotic therapy. The 2 trials that evaluated the effect of probiotic therapy over an extended period required probiotics be started within 48 hours after initiation of antibiotic therapy; one trial continued probiotics for 5 days after completion of antibiotics, and the other for 7 days after completion.16,20 In both trials, CDI was statistically significantly reduced among adults using probiotics compared with adults receiving placebo.
Probiotic Safety
The FDA has not approved probiotics for the prevention or treatment of any health problems. Most probiotics are FDA-regulated as dietary supplements and do not have to meet stringent drug-approval requirements. The FDA has given many strains of common probiotics the Generally Recognized as Safe designation for use in commercially available products and foods.21-23 Probiotic use has not been associated with significant AEs in clinical trials and generally has been considered safe in immunocompetent and otherwise healthy persons.15-19 However, clinical trials have been inadequate in reporting or investigating AEs; the alternative for evaluating the risks of probiotic therapy is case reports.24,25 Theoretical risks associated with probiotics include sepsis, deleterious effects on normal gut digestion, excessive immune stimulation, and possible transfer of antimicrobial resistance genes among microorganisms.26 Boyle and colleagues further described a handful of case reports of sepsis caused by probiotics in immunocompromised individuals; the other theoretical risks have not been reported outside animal studies.26
CDI Risk Factors
Many factors can increase a patient’s CDI risk. Specific antibiotics (eg, ampicillin, amoxicillin, cephalosporins, clindamycin, fluoroquinolones) confer higher risk.27,28 Other factors include inflammatory bowel disease, organ transplantation, chemotherapy, chronic kidney disease, and immunodeficiency. Advanced age increases CDI risk and can increase the severity of infection. The evidence regarding acid suppression and CDI risk is conflicting, though a recent meta-analysis found that use of proton pump inhibitors is associated with a 2-fold higher risk of developing CDI.29 Patient-specific risk factors should be evaluated when the risk–benefit ratio for probiotic use is being considered.
Conclusion
CDIs are becoming increasingly burdensome to the health care system. More research is needed on the role of probiotics in CDI prevention in patients taking antibiotics. Given the limited risk for AEs when probiotics are used in immunocompetent patients and the relatively low cost of these supplements, the risks likely are outweighed by the postulated benefits, and probiotics may be recommended in select patient populations.
The PLACIDE trial found no benefit of probiotics in preventing CDI in a population similar to that of a typical US hospital or ambulatory setting, but its intervention allowed late initiation of relatively low doses of probiotics. Therefore, probiotics may be recommended for CDI prevention in patients taking antibiotics, especially patients at high risk for developing CDI. When clinicians recommend probiotic use in this setting, the probiotic should be initiated within 2 days after the start of antibiotics and should be continued for the duration of antibiotic therapy and for up to 7 days after that therapy is completed. Optimal probiotic dosing, likely dependent on the product used, remains unclear. PLACIDE trial results suggest that a dosage of at least 1 probiotic capsule 2 times daily may confer additional efficacy.
1. Desai K, Gupta SB, Dubberke ER, Prabhu VS, Browne C, Mast TC. Epidemiological and economic burden of Clostridium difficile in the United States: estimates from a modeling approach. BMC Infect Dis. 2016;16:303.
2. Miller BA, Chen LF, Sexton DJ, Anderson DJ. Comparison of the burdens of hospital-onset, healthcare facility-associated Clostridium difficile infection and of healthcare-associated infection due to methicillin-resistant Staphylococcus aureus in community hospitals. Infect Control Hosp Epidemiol. 2011;32(4):387-390.
3. Evans ME, Kralovic SM, Simbartl LA, Jain R, Roselle GA. Effect of a Clostridium difficile infection prevention initiative in Veterans Affairs acute care facilities. Infect Control Hosp Epidemiol. 2016;37(6):720-722.
4. Bartlett JG. Clinical practice. Antibiotic-associated diarrhea. N Engl J Med. 2002;346(5):334-339.
5. Johnson S, Clabots CR, Linn FV, Olson MM, Peterson LR, Gerding DN. Nosocomial Clostridium difficile colonisation and disease. Lancet. 1990;336(8707):97-100.
6. McFarland LV, Mulligan ME, Kwok RY, Stamm WE. Nosocomial acquisition of Clostridium difficile infection. N Engl J Med. 1989;320(4):204-210.
7. McFarland LV, Elmer GW, Surawicz CM. Breaking the cycle: treatment strategies for 163 cases of recurrent Clostridium difficile disease. Am J Gastroenterol. 2002;97(7):1769-1775.
8. Kelly CP. Can we identify patients at high risk of recurrent Clostridium difficile infection? Clin Microbiol Infect. 2012;18(suppl 6):21-27.
9. Sartor RB. Probiotics for gastrointestinal diseases. https://www.uptodate.com/contents/probiotics-for-gastrointestinal-diseases. Updated September 4, 2018. Accessed April 4, 2019.
10. VSL#3 (Lactobacillus) [prescribing information]. Covington, LA: Alfasigma USA Inc; July 2017.
11. Culturelle Digestive Health Probiotic Capsules (Lactobacillus) [prescribing information]. Cromwell, CT: I-Health, Inc; 2015.
12. Flora-Q (Lactobacillus) [prescribing information]. Melville, NY: PharmaDerm; May 2012.
13. Lactinex (Lactobacillus) [prescribing information]. Franklin Lakes, NJ: Becton, Dickinson and Company; 2015
14. McDonald LC, Gerding DN, Johnson S, et al. Clinical practice guidelines for Clostridium difficile infection in adults and children: 2017 update by the Infectious Diseases Society of America (IDSA) and Society for Healthcare Epidemiology of America (SHEA). Clin Infect Dis. 2018;66(7):987-994.
15. Goldenberg JZ, Yap C, Lytvyn L, et al. Probiotics for the prevention of Clostridium difficile–associated diarrhea in adults and children. Cochrane Database Syst Rev. 2017;(12):CD006095.
16. Gao XW, Mubasher M, Fang CY, Reifer C, Miller LE. Dose–response efficacy of a proprietary probiotic formula of Lactobacillus acidophilus CL1285 and Lactobacillus casei LBC80R for antibiotic-associated diarrhea and Clostridium difficile–associated diarrhea prophylaxis in adult patients. Am J Gastroenterol. 2010;105(7):1636-1641.
17. Ouwehand AC, DongLian C, Weijian X, et al. Probiotics reduce symptoms of antibiotic use in a hospital setting: a randomized dose response study. Vaccine. 2014;32(4):458-463.
18. Allen SJ, Wareham K, Wang D, et al. Lactobacilli and bifidobacteria in the prevention of antibiotic-associated diarrhoea and Clostridium difficile diarrhoea in older inpatients (PLACIDE): a randomised, double-blind, placebo-controlled, multicentre trial. Lancet. 2013;382(9900):1249-1257.
19. Shen NT, Maw A, Tmanova LL, et al. Timely use of probiotics in hospitalized adults prevents Clostridium difficile infection: a systematic review with meta-regression analysis. Gastroenterology. 2017;152(8):1889-1900.
20. Hickson M, D’Souza AL, Muthu N, et al. Use of probiotic Lactobacillus preparation to prevent diarrhoea associated with antibiotics: randomised double blind placebo controlled trial. BMJ. 2007;335(7610):80.
21. Center for Food Safety and Applied Nutrition. GRAS notice inventory. https://www.fda.gov/Food/IngredientsPackagingLabeling/GRAS/NoticeInventory/default.htm. Updated September 26, 2018. Accessed November 1, 2018.
22. Mattia A, Merker R. Regulation of probiotic substances as ingredients in foods: premarket approval or “generally recognized as safe” notification. Clin Infect Dis. 2008;46(suppl 2):S115-S118.
23. Probiotics: in depth. https://nccih.nih.gov/health/probiotics/introduction.htm. Updated October 2016. Accessed January 15, 2019.
24. Doron S, Snydman DR. Risk and safety of probiotics. Clin Infect Dis. 2015;60(suppl 2):S129-S134.
25. Bafeta A, Koh M, Riveros C, Ravaud P. Harms reporting in randomized controlled trials of interventions aimed at modifying microbiota: a systematic review. Ann Intern Med. 2018;169(4):240-247.
26. Boyle RJ, Robins-Browne RM, Tang ML. Probiotic use in clinical practice: what are the risks? Am J Clin Nutr. 2006;83(6):1256-1264.
27. Leffler DA, Lamont JT. Clostridium difficile infection. N Engl J Med. 2015;372(16):1539-1548.
28. Brown KA, Khanafer N, Daneman N, Fisman DN. Meta-analysis of antibiotics and the risk of community-associated Clostridium difficile infection. Antimicrob Agents Chemoth. 2013;57(5):2326-2332.
29. Oshima T, Wu L, Li M, Fukui H, Watari J, Miwa H. Magnitude and direction of the association between Clostridium difficile infection and proton pump inhibitors in adults and pediatric patients: a systematic review and meta-analysis. J Gastroenterol. 2018;53(1):84-94.
Clostridium difficile (C difficile) is a gram-positive, toxin-producing bacterium that is of increasing concern among health care providers and patients. Infection with C difficile can have manifestations ranging from mild diarrhea to severe toxic megacolon and can result in prolonged hospitalization with severe cases requiring admission to an intensive care unit.1 In 2014, the US was estimated to have more than 600,000 cases of C difficile infection (CDI), previously known as C difficile–associated diarrhea, and more than 44,000 associated deaths. The annual economic cost of CDI is thought to exceed $5 billion.1 According to studies of health care–associated illness, CDI rates are comparable to or have surpassed rates of methicillin-resistant Staphylococcus aureus infection within the US, including at US Department of Veterans Affairs (VA) acute care centers nationwide.2,3
C difficile has been shown to be the causative agent in 10% to 20% of antibiotic-associated diarrhea episodes.4 Colonization of C difficile is uncommon in healthy adults, but colonization rates are as high as 21% in hospitalized patients, with increasing rates proportional to increasing hospital length of stay.5,6 Although not all colonized patients develop clinically significant CDI, those who do may require multiple treatment courses, over months to years, because of the high risk of disease recurrence. An estimated 25% of patients have a single recurrent episode of CDI within 30 days after treatment completion, and 45% of those patients have additional recurrent infections.7,8 Although probiotics do not have an approved US Food and Drug Administration (FDA) indication, these supplements are often used to try to prevent CDI from developing during concomitant antibiotic use. Probiotics are microorganisms with potential health benefits, but the mechanisms of these benefits are not fully understood. Proposed mechanisms include reduced growth of pathogenic bacteria, modulation of the immune system, and support of the intestinal wall barrier.9 The many probiotic formulations currently marketed include Lactobacillus acidophilus (L acidophilus) capsules and various combinations of L acidophilus, Lactobacillus casei, Bifidobacterium lactis, Bifidobacterium longum, Streptococcus thermophilus, and other bacterial strains.
Dosing and Guidelines
Manufacturers’ suggested dosing for their Lactobacillus capsules, tablets, and packets varies from 1 unit daily to 4 units 4 times daily for dietary supplementation; the products’ labeling does not include any information regarding treatment duration.10-13 In addition, there are no published recommendations or product labeling guiding the dosing of probiotics or their duration of use in the primary prevention of CDI.
In 2017, the Infectious Diseases Society of America (IDSA) and the Society for Healthcare Epidemiology of America (SHEA) updated their CDI treatment guidelines.14 As these guidelines indicate that the data on probiotic use in CDI are inadequate, IDSA and SHEA make no recommendation for or against probiotic use in primary prevention of the disease. The guidelines point to several limitations in the literature, including variability in probiotic formulations studied, duration of probiotic administration, definitions of CDI, and duration of study follow-up.
Given the lack of consensus guidelines that clinicians can use when deciding which probiotic dosing and duration are appropriate for a patient for primary prevention of CDI, we evaluated the literature on the topic and summarize their findings here.
Review of Probiotoc Literature
Conflicting data exist about probiotics and their effect on CDI prevention. The literature reviewed was selected based on our assessment of its contribution to the topic and its potential utility to clinicians in determining appropriate probiotic therapies and recommendations. Included in our discussion is a large Cochrane Review of probiotic efficacy, 2 trials of probiotic dosing, the PLACIDE trial, and a systematic review of timely probiotic initiation. All of these studies attempted to determine the effect of probiotics on CDI incidence (Table).
In their 2017 Cochrane Review, Goldenberg and colleagues reviewed 39 trials that investigated the efficacy of probiotics in CDI prevention in 9,955 immunocompetent patients receiving antibiotics.15 The incidence of CDI was significantly lower in patients who received a probiotic than in patients who received placebo or no treatment (1.5% vs 4.0%; relative risk [RR], 0.40; 95% CI, 0.30-0.52; I2 = 0%). It is important to note that trials with a control-group CDI incidence of 0% to 2% (baseline CDI risk) found no statistically significant difference in CDI risk between patients using and not using probiotics (RR, 0.77; 95% CI, 0.45-1.32; I2 = 0%) and that the preceding statistically significant result may have been driven by the inclusion of trials with high baseline CDI risk (> 5%). Trials that enrolled patients who were at this risk level found a statistically significant 70% reduction in CDI risk in those using probiotics (vs no probiotics) while on concomitant antibiotic therapy (RR, 0.30; 95% CI, 0.21-0.42; I2 = 0%).
Probiotic therapy seems to be effective in reducing CDI risk in immunocompetent patients and may be particularly beneficial in patients at higher CDI risk, though Goldenberg and colleagues did not elaborate on what constitutes higher risk and based their conclusion on their control group’s high CDI incidence (> 5%). The most common adverse events (AEs) attributable to probiotics included abdominal cramping, nausea, fever, soft stools, flatulence, and taste disturbance. The review’s findings are limited in that the inclusion of many small trials with high baseline CDI risk likely contributed to a statistically significant result, and 17 of the review’s 39 trials were industry-sponsored or were conducted by investigators with industry associations; another 12 lacked statements about funding or sponsorship.
Two of the trials in the Cochrane Review investigated whether probiotics have a dose effect on CDI prevention. Gao and colleagues randomly assigned 255 hospitalized Asian patients to 3 groups: those receiving placebo, 1 probiotic capsule daily, and 2 probiotic capsules daily.16 Each probiotic capsule contained 50 billion colony-forming units (CFUs) of Lactobacillus. Incidence of CDI was lower in patients taking 2 probiotic capsules daily than in those taking 1 probiotic capsule daily (1.2% vs 9.4%; P = .04) or placebo (1.2% vs 23.8%; P = .002). In the other trial, Ouwehand and colleagues randomly assigned 503 hospitalized Asian patients to 3 groups as well: those receiving placebo, low-dose probiotic (4.17 billion CFUs of Lactobacillus and Bifidobacterium), and high-dose probiotic (17 billion CFUs).17 The incidence of CDI in each probiotic group (low-dose, high-dose) was 1.8%, which was significantly lower than the 4.8% in the placebo group (P = .04).
The Cochrane Review’s largest and most rigorous trial was PLACIDE, a 2013 randomized controlled study of the effect of probiotics on CDI.18 Allen and colleagues randomly assigned 2,981 inpatients (aged ≥ 65 years; exposed to antibiotics within preceding 7 days) to 2 groups: those receiving either 1 probiotic capsule daily, or 1 placebo capsule daily, for 21 days. Results showed no difference in CDI incidence between the probiotic and placebo groups (0.8% vs 1.2%; RR, 0.71; 95% CI, 0.34-1.47; P = .35). Of note, this trial is free of industry sponsorship, is the largest probiotic trial to date, has a control-group baseline CDI rate consistent with the rate in hospital and ambulatory settings in the US, and found a negative result regarding probiotic use in CDI prevention. Findings are limited in that the study allowed for initiating probiotic therapy up to 7 days after the start of antibiotics, and patients were given 1 relatively low-dose capsule daily, which may have contributed to lack of an effect on CDI prevention. No serious AEs were attributed to probiotic use.
In a 2017 systematic meta-analysis of 19 studies, Shen and colleagues investigated whether timely use of probiotics prevented CDI in 6,261 hospitalized patients receiving antibiotics.19 The incidence of CDI was significantly lower in patients receiving vs not receiving probiotics (1.6% vs 3.9%; RR, 0.42; 95% CI, 0.30-0.57; I2 = 0%; P < .001).19 A subgroup analysis was performed to compare studies initiating probiotics within 2 days after the start of antibiotics with studies initiating probiotics more than 2 days after the start. CDI risk was reduced by 68% when probiotics were started within 2 days, vs 30% when started after 2 days (RR, 0.32; 95% CI, 0.22-0.48; I2 = 0% vs RR, 0.70; 95% CI, 0.40-1.23; I2 = 0%; P = .02). Of note, no difference was found in efficacy among the various probiotic formulations, and no significant AEs were noted in any study group.
Trials included in the Cochrane Review used many different probiotic regimens over various durations.15 All these trials continued probiotics for at least the duration of antibiotic therapy. The 2 trials that evaluated the effect of probiotic therapy over an extended period required probiotics be started within 48 hours after initiation of antibiotic therapy; one trial continued probiotics for 5 days after completion of antibiotics, and the other for 7 days after completion.16,20 In both trials, CDI was statistically significantly reduced among adults using probiotics compared with adults receiving placebo.
Probiotic Safety
The FDA has not approved probiotics for the prevention or treatment of any health problems. Most probiotics are FDA-regulated as dietary supplements and do not have to meet stringent drug-approval requirements. The FDA has given many strains of common probiotics the Generally Recognized as Safe designation for use in commercially available products and foods.21-23 Probiotic use has not been associated with significant AEs in clinical trials and generally has been considered safe in immunocompetent and otherwise healthy persons.15-19 However, clinical trials have been inadequate in reporting or investigating AEs; the alternative for evaluating the risks of probiotic therapy is case reports.24,25 Theoretical risks associated with probiotics include sepsis, deleterious effects on normal gut digestion, excessive immune stimulation, and possible transfer of antimicrobial resistance genes among microorganisms.26 Boyle and colleagues further described a handful of case reports of sepsis caused by probiotics in immunocompromised individuals; the other theoretical risks have not been reported outside animal studies.26
CDI Risk Factors
Many factors can increase a patient’s CDI risk. Specific antibiotics (eg, ampicillin, amoxicillin, cephalosporins, clindamycin, fluoroquinolones) confer higher risk.27,28 Other factors include inflammatory bowel disease, organ transplantation, chemotherapy, chronic kidney disease, and immunodeficiency. Advanced age increases CDI risk and can increase the severity of infection. The evidence regarding acid suppression and CDI risk is conflicting, though a recent meta-analysis found that use of proton pump inhibitors is associated with a 2-fold higher risk of developing CDI.29 Patient-specific risk factors should be evaluated when the risk–benefit ratio for probiotic use is being considered.
Conclusion
CDIs are becoming increasingly burdensome to the health care system. More research is needed on the role of probiotics in CDI prevention in patients taking antibiotics. Given the limited risk for AEs when probiotics are used in immunocompetent patients and the relatively low cost of these supplements, the risks likely are outweighed by the postulated benefits, and probiotics may be recommended in select patient populations.
The PLACIDE trial found no benefit of probiotics in preventing CDI in a population similar to that of a typical US hospital or ambulatory setting, but its intervention allowed late initiation of relatively low doses of probiotics. Therefore, probiotics may be recommended for CDI prevention in patients taking antibiotics, especially patients at high risk for developing CDI. When clinicians recommend probiotic use in this setting, the probiotic should be initiated within 2 days after the start of antibiotics and should be continued for the duration of antibiotic therapy and for up to 7 days after that therapy is completed. Optimal probiotic dosing, likely dependent on the product used, remains unclear. PLACIDE trial results suggest that a dosage of at least 1 probiotic capsule 2 times daily may confer additional efficacy.
Clostridium difficile (C difficile) is a gram-positive, toxin-producing bacterium that is of increasing concern among health care providers and patients. Infection with C difficile can have manifestations ranging from mild diarrhea to severe toxic megacolon and can result in prolonged hospitalization with severe cases requiring admission to an intensive care unit.1 In 2014, the US was estimated to have more than 600,000 cases of C difficile infection (CDI), previously known as C difficile–associated diarrhea, and more than 44,000 associated deaths. The annual economic cost of CDI is thought to exceed $5 billion.1 According to studies of health care–associated illness, CDI rates are comparable to or have surpassed rates of methicillin-resistant Staphylococcus aureus infection within the US, including at US Department of Veterans Affairs (VA) acute care centers nationwide.2,3
C difficile has been shown to be the causative agent in 10% to 20% of antibiotic-associated diarrhea episodes.4 Colonization of C difficile is uncommon in healthy adults, but colonization rates are as high as 21% in hospitalized patients, with increasing rates proportional to increasing hospital length of stay.5,6 Although not all colonized patients develop clinically significant CDI, those who do may require multiple treatment courses, over months to years, because of the high risk of disease recurrence. An estimated 25% of patients have a single recurrent episode of CDI within 30 days after treatment completion, and 45% of those patients have additional recurrent infections.7,8 Although probiotics do not have an approved US Food and Drug Administration (FDA) indication, these supplements are often used to try to prevent CDI from developing during concomitant antibiotic use. Probiotics are microorganisms with potential health benefits, but the mechanisms of these benefits are not fully understood. Proposed mechanisms include reduced growth of pathogenic bacteria, modulation of the immune system, and support of the intestinal wall barrier.9 The many probiotic formulations currently marketed include Lactobacillus acidophilus (L acidophilus) capsules and various combinations of L acidophilus, Lactobacillus casei, Bifidobacterium lactis, Bifidobacterium longum, Streptococcus thermophilus, and other bacterial strains.
Dosing and Guidelines
Manufacturers’ suggested dosing for their Lactobacillus capsules, tablets, and packets varies from 1 unit daily to 4 units 4 times daily for dietary supplementation; the products’ labeling does not include any information regarding treatment duration.10-13 In addition, there are no published recommendations or product labeling guiding the dosing of probiotics or their duration of use in the primary prevention of CDI.
In 2017, the Infectious Diseases Society of America (IDSA) and the Society for Healthcare Epidemiology of America (SHEA) updated their CDI treatment guidelines.14 As these guidelines indicate that the data on probiotic use in CDI are inadequate, IDSA and SHEA make no recommendation for or against probiotic use in primary prevention of the disease. The guidelines point to several limitations in the literature, including variability in probiotic formulations studied, duration of probiotic administration, definitions of CDI, and duration of study follow-up.
Given the lack of consensus guidelines that clinicians can use when deciding which probiotic dosing and duration are appropriate for a patient for primary prevention of CDI, we evaluated the literature on the topic and summarize their findings here.
Review of Probiotoc Literature
Conflicting data exist about probiotics and their effect on CDI prevention. The literature reviewed was selected based on our assessment of its contribution to the topic and its potential utility to clinicians in determining appropriate probiotic therapies and recommendations. Included in our discussion is a large Cochrane Review of probiotic efficacy, 2 trials of probiotic dosing, the PLACIDE trial, and a systematic review of timely probiotic initiation. All of these studies attempted to determine the effect of probiotics on CDI incidence (Table).
In their 2017 Cochrane Review, Goldenberg and colleagues reviewed 39 trials that investigated the efficacy of probiotics in CDI prevention in 9,955 immunocompetent patients receiving antibiotics.15 The incidence of CDI was significantly lower in patients who received a probiotic than in patients who received placebo or no treatment (1.5% vs 4.0%; relative risk [RR], 0.40; 95% CI, 0.30-0.52; I2 = 0%). It is important to note that trials with a control-group CDI incidence of 0% to 2% (baseline CDI risk) found no statistically significant difference in CDI risk between patients using and not using probiotics (RR, 0.77; 95% CI, 0.45-1.32; I2 = 0%) and that the preceding statistically significant result may have been driven by the inclusion of trials with high baseline CDI risk (> 5%). Trials that enrolled patients who were at this risk level found a statistically significant 70% reduction in CDI risk in those using probiotics (vs no probiotics) while on concomitant antibiotic therapy (RR, 0.30; 95% CI, 0.21-0.42; I2 = 0%).
Probiotic therapy seems to be effective in reducing CDI risk in immunocompetent patients and may be particularly beneficial in patients at higher CDI risk, though Goldenberg and colleagues did not elaborate on what constitutes higher risk and based their conclusion on their control group’s high CDI incidence (> 5%). The most common adverse events (AEs) attributable to probiotics included abdominal cramping, nausea, fever, soft stools, flatulence, and taste disturbance. The review’s findings are limited in that the inclusion of many small trials with high baseline CDI risk likely contributed to a statistically significant result, and 17 of the review’s 39 trials were industry-sponsored or were conducted by investigators with industry associations; another 12 lacked statements about funding or sponsorship.
Two of the trials in the Cochrane Review investigated whether probiotics have a dose effect on CDI prevention. Gao and colleagues randomly assigned 255 hospitalized Asian patients to 3 groups: those receiving placebo, 1 probiotic capsule daily, and 2 probiotic capsules daily.16 Each probiotic capsule contained 50 billion colony-forming units (CFUs) of Lactobacillus. Incidence of CDI was lower in patients taking 2 probiotic capsules daily than in those taking 1 probiotic capsule daily (1.2% vs 9.4%; P = .04) or placebo (1.2% vs 23.8%; P = .002). In the other trial, Ouwehand and colleagues randomly assigned 503 hospitalized Asian patients to 3 groups as well: those receiving placebo, low-dose probiotic (4.17 billion CFUs of Lactobacillus and Bifidobacterium), and high-dose probiotic (17 billion CFUs).17 The incidence of CDI in each probiotic group (low-dose, high-dose) was 1.8%, which was significantly lower than the 4.8% in the placebo group (P = .04).
The Cochrane Review’s largest and most rigorous trial was PLACIDE, a 2013 randomized controlled study of the effect of probiotics on CDI.18 Allen and colleagues randomly assigned 2,981 inpatients (aged ≥ 65 years; exposed to antibiotics within preceding 7 days) to 2 groups: those receiving either 1 probiotic capsule daily, or 1 placebo capsule daily, for 21 days. Results showed no difference in CDI incidence between the probiotic and placebo groups (0.8% vs 1.2%; RR, 0.71; 95% CI, 0.34-1.47; P = .35). Of note, this trial is free of industry sponsorship, is the largest probiotic trial to date, has a control-group baseline CDI rate consistent with the rate in hospital and ambulatory settings in the US, and found a negative result regarding probiotic use in CDI prevention. Findings are limited in that the study allowed for initiating probiotic therapy up to 7 days after the start of antibiotics, and patients were given 1 relatively low-dose capsule daily, which may have contributed to lack of an effect on CDI prevention. No serious AEs were attributed to probiotic use.
In a 2017 systematic meta-analysis of 19 studies, Shen and colleagues investigated whether timely use of probiotics prevented CDI in 6,261 hospitalized patients receiving antibiotics.19 The incidence of CDI was significantly lower in patients receiving vs not receiving probiotics (1.6% vs 3.9%; RR, 0.42; 95% CI, 0.30-0.57; I2 = 0%; P < .001).19 A subgroup analysis was performed to compare studies initiating probiotics within 2 days after the start of antibiotics with studies initiating probiotics more than 2 days after the start. CDI risk was reduced by 68% when probiotics were started within 2 days, vs 30% when started after 2 days (RR, 0.32; 95% CI, 0.22-0.48; I2 = 0% vs RR, 0.70; 95% CI, 0.40-1.23; I2 = 0%; P = .02). Of note, no difference was found in efficacy among the various probiotic formulations, and no significant AEs were noted in any study group.
Trials included in the Cochrane Review used many different probiotic regimens over various durations.15 All these trials continued probiotics for at least the duration of antibiotic therapy. The 2 trials that evaluated the effect of probiotic therapy over an extended period required probiotics be started within 48 hours after initiation of antibiotic therapy; one trial continued probiotics for 5 days after completion of antibiotics, and the other for 7 days after completion.16,20 In both trials, CDI was statistically significantly reduced among adults using probiotics compared with adults receiving placebo.
Probiotic Safety
The FDA has not approved probiotics for the prevention or treatment of any health problems. Most probiotics are FDA-regulated as dietary supplements and do not have to meet stringent drug-approval requirements. The FDA has given many strains of common probiotics the Generally Recognized as Safe designation for use in commercially available products and foods.21-23 Probiotic use has not been associated with significant AEs in clinical trials and generally has been considered safe in immunocompetent and otherwise healthy persons.15-19 However, clinical trials have been inadequate in reporting or investigating AEs; the alternative for evaluating the risks of probiotic therapy is case reports.24,25 Theoretical risks associated with probiotics include sepsis, deleterious effects on normal gut digestion, excessive immune stimulation, and possible transfer of antimicrobial resistance genes among microorganisms.26 Boyle and colleagues further described a handful of case reports of sepsis caused by probiotics in immunocompromised individuals; the other theoretical risks have not been reported outside animal studies.26
CDI Risk Factors
Many factors can increase a patient’s CDI risk. Specific antibiotics (eg, ampicillin, amoxicillin, cephalosporins, clindamycin, fluoroquinolones) confer higher risk.27,28 Other factors include inflammatory bowel disease, organ transplantation, chemotherapy, chronic kidney disease, and immunodeficiency. Advanced age increases CDI risk and can increase the severity of infection. The evidence regarding acid suppression and CDI risk is conflicting, though a recent meta-analysis found that use of proton pump inhibitors is associated with a 2-fold higher risk of developing CDI.29 Patient-specific risk factors should be evaluated when the risk–benefit ratio for probiotic use is being considered.
Conclusion
CDIs are becoming increasingly burdensome to the health care system. More research is needed on the role of probiotics in CDI prevention in patients taking antibiotics. Given the limited risk for AEs when probiotics are used in immunocompetent patients and the relatively low cost of these supplements, the risks likely are outweighed by the postulated benefits, and probiotics may be recommended in select patient populations.
The PLACIDE trial found no benefit of probiotics in preventing CDI in a population similar to that of a typical US hospital or ambulatory setting, but its intervention allowed late initiation of relatively low doses of probiotics. Therefore, probiotics may be recommended for CDI prevention in patients taking antibiotics, especially patients at high risk for developing CDI. When clinicians recommend probiotic use in this setting, the probiotic should be initiated within 2 days after the start of antibiotics and should be continued for the duration of antibiotic therapy and for up to 7 days after that therapy is completed. Optimal probiotic dosing, likely dependent on the product used, remains unclear. PLACIDE trial results suggest that a dosage of at least 1 probiotic capsule 2 times daily may confer additional efficacy.
1. Desai K, Gupta SB, Dubberke ER, Prabhu VS, Browne C, Mast TC. Epidemiological and economic burden of Clostridium difficile in the United States: estimates from a modeling approach. BMC Infect Dis. 2016;16:303.
2. Miller BA, Chen LF, Sexton DJ, Anderson DJ. Comparison of the burdens of hospital-onset, healthcare facility-associated Clostridium difficile infection and of healthcare-associated infection due to methicillin-resistant Staphylococcus aureus in community hospitals. Infect Control Hosp Epidemiol. 2011;32(4):387-390.
3. Evans ME, Kralovic SM, Simbartl LA, Jain R, Roselle GA. Effect of a Clostridium difficile infection prevention initiative in Veterans Affairs acute care facilities. Infect Control Hosp Epidemiol. 2016;37(6):720-722.
4. Bartlett JG. Clinical practice. Antibiotic-associated diarrhea. N Engl J Med. 2002;346(5):334-339.
5. Johnson S, Clabots CR, Linn FV, Olson MM, Peterson LR, Gerding DN. Nosocomial Clostridium difficile colonisation and disease. Lancet. 1990;336(8707):97-100.
6. McFarland LV, Mulligan ME, Kwok RY, Stamm WE. Nosocomial acquisition of Clostridium difficile infection. N Engl J Med. 1989;320(4):204-210.
7. McFarland LV, Elmer GW, Surawicz CM. Breaking the cycle: treatment strategies for 163 cases of recurrent Clostridium difficile disease. Am J Gastroenterol. 2002;97(7):1769-1775.
8. Kelly CP. Can we identify patients at high risk of recurrent Clostridium difficile infection? Clin Microbiol Infect. 2012;18(suppl 6):21-27.
9. Sartor RB. Probiotics for gastrointestinal diseases. https://www.uptodate.com/contents/probiotics-for-gastrointestinal-diseases. Updated September 4, 2018. Accessed April 4, 2019.
10. VSL#3 (Lactobacillus) [prescribing information]. Covington, LA: Alfasigma USA Inc; July 2017.
11. Culturelle Digestive Health Probiotic Capsules (Lactobacillus) [prescribing information]. Cromwell, CT: I-Health, Inc; 2015.
12. Flora-Q (Lactobacillus) [prescribing information]. Melville, NY: PharmaDerm; May 2012.
13. Lactinex (Lactobacillus) [prescribing information]. Franklin Lakes, NJ: Becton, Dickinson and Company; 2015
14. McDonald LC, Gerding DN, Johnson S, et al. Clinical practice guidelines for Clostridium difficile infection in adults and children: 2017 update by the Infectious Diseases Society of America (IDSA) and Society for Healthcare Epidemiology of America (SHEA). Clin Infect Dis. 2018;66(7):987-994.
15. Goldenberg JZ, Yap C, Lytvyn L, et al. Probiotics for the prevention of Clostridium difficile–associated diarrhea in adults and children. Cochrane Database Syst Rev. 2017;(12):CD006095.
16. Gao XW, Mubasher M, Fang CY, Reifer C, Miller LE. Dose–response efficacy of a proprietary probiotic formula of Lactobacillus acidophilus CL1285 and Lactobacillus casei LBC80R for antibiotic-associated diarrhea and Clostridium difficile–associated diarrhea prophylaxis in adult patients. Am J Gastroenterol. 2010;105(7):1636-1641.
17. Ouwehand AC, DongLian C, Weijian X, et al. Probiotics reduce symptoms of antibiotic use in a hospital setting: a randomized dose response study. Vaccine. 2014;32(4):458-463.
18. Allen SJ, Wareham K, Wang D, et al. Lactobacilli and bifidobacteria in the prevention of antibiotic-associated diarrhoea and Clostridium difficile diarrhoea in older inpatients (PLACIDE): a randomised, double-blind, placebo-controlled, multicentre trial. Lancet. 2013;382(9900):1249-1257.
19. Shen NT, Maw A, Tmanova LL, et al. Timely use of probiotics in hospitalized adults prevents Clostridium difficile infection: a systematic review with meta-regression analysis. Gastroenterology. 2017;152(8):1889-1900.
20. Hickson M, D’Souza AL, Muthu N, et al. Use of probiotic Lactobacillus preparation to prevent diarrhoea associated with antibiotics: randomised double blind placebo controlled trial. BMJ. 2007;335(7610):80.
21. Center for Food Safety and Applied Nutrition. GRAS notice inventory. https://www.fda.gov/Food/IngredientsPackagingLabeling/GRAS/NoticeInventory/default.htm. Updated September 26, 2018. Accessed November 1, 2018.
22. Mattia A, Merker R. Regulation of probiotic substances as ingredients in foods: premarket approval or “generally recognized as safe” notification. Clin Infect Dis. 2008;46(suppl 2):S115-S118.
23. Probiotics: in depth. https://nccih.nih.gov/health/probiotics/introduction.htm. Updated October 2016. Accessed January 15, 2019.
24. Doron S, Snydman DR. Risk and safety of probiotics. Clin Infect Dis. 2015;60(suppl 2):S129-S134.
25. Bafeta A, Koh M, Riveros C, Ravaud P. Harms reporting in randomized controlled trials of interventions aimed at modifying microbiota: a systematic review. Ann Intern Med. 2018;169(4):240-247.
26. Boyle RJ, Robins-Browne RM, Tang ML. Probiotic use in clinical practice: what are the risks? Am J Clin Nutr. 2006;83(6):1256-1264.
27. Leffler DA, Lamont JT. Clostridium difficile infection. N Engl J Med. 2015;372(16):1539-1548.
28. Brown KA, Khanafer N, Daneman N, Fisman DN. Meta-analysis of antibiotics and the risk of community-associated Clostridium difficile infection. Antimicrob Agents Chemoth. 2013;57(5):2326-2332.
29. Oshima T, Wu L, Li M, Fukui H, Watari J, Miwa H. Magnitude and direction of the association between Clostridium difficile infection and proton pump inhibitors in adults and pediatric patients: a systematic review and meta-analysis. J Gastroenterol. 2018;53(1):84-94.
1. Desai K, Gupta SB, Dubberke ER, Prabhu VS, Browne C, Mast TC. Epidemiological and economic burden of Clostridium difficile in the United States: estimates from a modeling approach. BMC Infect Dis. 2016;16:303.
2. Miller BA, Chen LF, Sexton DJ, Anderson DJ. Comparison of the burdens of hospital-onset, healthcare facility-associated Clostridium difficile infection and of healthcare-associated infection due to methicillin-resistant Staphylococcus aureus in community hospitals. Infect Control Hosp Epidemiol. 2011;32(4):387-390.
3. Evans ME, Kralovic SM, Simbartl LA, Jain R, Roselle GA. Effect of a Clostridium difficile infection prevention initiative in Veterans Affairs acute care facilities. Infect Control Hosp Epidemiol. 2016;37(6):720-722.
4. Bartlett JG. Clinical practice. Antibiotic-associated diarrhea. N Engl J Med. 2002;346(5):334-339.
5. Johnson S, Clabots CR, Linn FV, Olson MM, Peterson LR, Gerding DN. Nosocomial Clostridium difficile colonisation and disease. Lancet. 1990;336(8707):97-100.
6. McFarland LV, Mulligan ME, Kwok RY, Stamm WE. Nosocomial acquisition of Clostridium difficile infection. N Engl J Med. 1989;320(4):204-210.
7. McFarland LV, Elmer GW, Surawicz CM. Breaking the cycle: treatment strategies for 163 cases of recurrent Clostridium difficile disease. Am J Gastroenterol. 2002;97(7):1769-1775.
8. Kelly CP. Can we identify patients at high risk of recurrent Clostridium difficile infection? Clin Microbiol Infect. 2012;18(suppl 6):21-27.
9. Sartor RB. Probiotics for gastrointestinal diseases. https://www.uptodate.com/contents/probiotics-for-gastrointestinal-diseases. Updated September 4, 2018. Accessed April 4, 2019.
10. VSL#3 (Lactobacillus) [prescribing information]. Covington, LA: Alfasigma USA Inc; July 2017.
11. Culturelle Digestive Health Probiotic Capsules (Lactobacillus) [prescribing information]. Cromwell, CT: I-Health, Inc; 2015.
12. Flora-Q (Lactobacillus) [prescribing information]. Melville, NY: PharmaDerm; May 2012.
13. Lactinex (Lactobacillus) [prescribing information]. Franklin Lakes, NJ: Becton, Dickinson and Company; 2015
14. McDonald LC, Gerding DN, Johnson S, et al. Clinical practice guidelines for Clostridium difficile infection in adults and children: 2017 update by the Infectious Diseases Society of America (IDSA) and Society for Healthcare Epidemiology of America (SHEA). Clin Infect Dis. 2018;66(7):987-994.
15. Goldenberg JZ, Yap C, Lytvyn L, et al. Probiotics for the prevention of Clostridium difficile–associated diarrhea in adults and children. Cochrane Database Syst Rev. 2017;(12):CD006095.
16. Gao XW, Mubasher M, Fang CY, Reifer C, Miller LE. Dose–response efficacy of a proprietary probiotic formula of Lactobacillus acidophilus CL1285 and Lactobacillus casei LBC80R for antibiotic-associated diarrhea and Clostridium difficile–associated diarrhea prophylaxis in adult patients. Am J Gastroenterol. 2010;105(7):1636-1641.
17. Ouwehand AC, DongLian C, Weijian X, et al. Probiotics reduce symptoms of antibiotic use in a hospital setting: a randomized dose response study. Vaccine. 2014;32(4):458-463.
18. Allen SJ, Wareham K, Wang D, et al. Lactobacilli and bifidobacteria in the prevention of antibiotic-associated diarrhoea and Clostridium difficile diarrhoea in older inpatients (PLACIDE): a randomised, double-blind, placebo-controlled, multicentre trial. Lancet. 2013;382(9900):1249-1257.
19. Shen NT, Maw A, Tmanova LL, et al. Timely use of probiotics in hospitalized adults prevents Clostridium difficile infection: a systematic review with meta-regression analysis. Gastroenterology. 2017;152(8):1889-1900.
20. Hickson M, D’Souza AL, Muthu N, et al. Use of probiotic Lactobacillus preparation to prevent diarrhoea associated with antibiotics: randomised double blind placebo controlled trial. BMJ. 2007;335(7610):80.
21. Center for Food Safety and Applied Nutrition. GRAS notice inventory. https://www.fda.gov/Food/IngredientsPackagingLabeling/GRAS/NoticeInventory/default.htm. Updated September 26, 2018. Accessed November 1, 2018.
22. Mattia A, Merker R. Regulation of probiotic substances as ingredients in foods: premarket approval or “generally recognized as safe” notification. Clin Infect Dis. 2008;46(suppl 2):S115-S118.
23. Probiotics: in depth. https://nccih.nih.gov/health/probiotics/introduction.htm. Updated October 2016. Accessed January 15, 2019.
24. Doron S, Snydman DR. Risk and safety of probiotics. Clin Infect Dis. 2015;60(suppl 2):S129-S134.
25. Bafeta A, Koh M, Riveros C, Ravaud P. Harms reporting in randomized controlled trials of interventions aimed at modifying microbiota: a systematic review. Ann Intern Med. 2018;169(4):240-247.
26. Boyle RJ, Robins-Browne RM, Tang ML. Probiotic use in clinical practice: what are the risks? Am J Clin Nutr. 2006;83(6):1256-1264.
27. Leffler DA, Lamont JT. Clostridium difficile infection. N Engl J Med. 2015;372(16):1539-1548.
28. Brown KA, Khanafer N, Daneman N, Fisman DN. Meta-analysis of antibiotics and the risk of community-associated Clostridium difficile infection. Antimicrob Agents Chemoth. 2013;57(5):2326-2332.
29. Oshima T, Wu L, Li M, Fukui H, Watari J, Miwa H. Magnitude and direction of the association between Clostridium difficile infection and proton pump inhibitors in adults and pediatric patients: a systematic review and meta-analysis. J Gastroenterol. 2018;53(1):84-94.
Tedizolid Use in Immunocompromised Patients
Immunocompromised patients are often susceptible to opportunistic infections, including those caused by multidrug-resistant organisms (MDROs). Transplant recipients are at high risk for developing infections due to lifelong immunosuppressive therapy.1,2 Additionally, patients receiving chemotherapy and those with HIV and AIDS are in an immunocompromised state.3-8
Regardless of the etiology for immunosuppression, decreased absolute neutrophil and platelet counts are seen in this condition. Although immunosuppressed individuals may be at increased risk of Gram-negative or Gram-positive infections, this review focuses on the treatment of Gram-positive bacterial infections. Of particular concern are opportunistic infections caused by Gram-positive MDROs, including methicillin-resistant Staphylococcus aureus (MRSA), vancomycin-resistant Enterococcus species (VRE), penicillin-resistant Streptococcus pneumoniae, and Nocardia species. Treatment of infections in the immunocompromised patient population warrants careful antimicrobial selection to ensure that a patient’s immune system is not further compromised due to adverse effects (AEs) secondary to therapy. As such, clinicians are exploring alternative antimicrobials, such as tedizolid, to treat various opportunistic infections.
Recently, requests at the William S. Middleton Memorial Veterans Hospital in Madison, Wisconsin, for off-label use of tedizolid have increased despite having other cheaper alternatives with comparable Gram-positive coverage. This review examines available literature regarding off-label use of tedizolid with a focus on use in immunocompromised patients.
Tedizolid phosphate (Sivextro) is an oxazolidinone antibiotic prodrug that joined linezolid as the second in its class in 2014. Oxazolidinones inhibit bacterial protein synthesis by binding to the 50S subunit of bacterial ribosomes in Gram-positive bacteria and are often used to treat MRSA and VRE infections.9 In vitro, oxazolidinones have shown bacteriostatic activity against Enterococcus and Staphylococcus species while exhibiting bactericidal activity against most Streptococcus species.10 Tedizolid has a US Food and Drug Administration (FDA) -approved, simplified dosing profile of 200 mg daily for 6 days compared with linezolid 400 to 600 mg twice daily for 10 to 14 days. Both medications are highly bioavailable with direct IV to oral conversion.11,12 Potential, expanded use of tedizolid against Gram-positive MDROs rests on a more favorable AE profile than does its linezolid predecessor. Tedizolid has been associated with less antibiotic-induced myelosuppression, which could prove valuable for immunocompromised patients.13
Tedizolid is approved for the sole indication of acute bacterial skin and skin structure infections (ABSSSI), whereas its predecessor has many approved indications and has been used extensively for off-label indications (Table).
Adverse reactions, as determined by 2 phase 2 and 2 phase 3 clinical trials evaluating 1,050 patients treated with tedizolid and 662 patients treated with linezolid, were similar between the oxazolidinones. Nausea was the most common AE and was reported in 8% and 12% of patients taking tedizolid and linezolid, respectively. Other common AEs (1%-6%) reported for both agents included vomiting, diarrhea, headache, and dizziness.11 Myelosuppression, peripheral neuropathy, and optic nerve disorders were the most common severe AEs reported with oxazolidinones. Tedizolid demonstrated a significantly decreased incidence of neutropenia (3%), defined by absolute neutrophil count 9/L compared with that of linezolid (7%) (P = .024).13 Evaluation of peripheral neuropathy and optic nerve disorders within the tedizolid and linezolid groups revealed similar incidences (peripheral neuropathy 1.2% vs 0.6%; optic nerve disorders 0.3% vs 0.2%, respectively).11
There is one preclinical trial that described the use of tedizolid in a murine model. A murine model study compared the antistaphylococcal killing effect of doses of tedizolid equivalent to human exposures ranging from 200 to 3,200 mg/d in both granulocytopenic and normal mice. The mice were evaluated at 24, 48, and 72 hours after therapy initiation. The presence of granulocytes had a dramatic effect on the antimicrobial effect of tedizolid. Dose response, demonstrated by the ratio of the area under the curve over the minimum inhibitory concentration, was on average > 25-fold for nonneutropenic vs neutropenic models. Near maximal effect of the nonneutropenic group, irrespective of duration of therapy, was achieved at the lowest dose tested (an exposure of about 200-mg tedizolid phosphate per day in humans).This study suggests that immunocompromised patients may warrant higher doses of tedizolid than the currently FDA-approved dose due to a decreased number of granulocytes available for modulating bacterial infections.15
Use of tedizolid doses higher than that which is FDA-approved may negate the favorable AE profile. A phase 1 clinical study was conducted to evaluate the safety, tolerability, and pharmacokinetics of tedizolid compared with those of linezolid in 40 healthy volunteers in a 21-day multiple ascending dose study.16 Subjects were stratified into 5 treatment cohorts: 200-, 300-, or 400-mg tedizolid orally once a day, 600-mg linezolid orally twice a day, and placebo. Tedizolid given at 200 mg had a hematologic safety profile similar to that of placebo. However, mean platelet counts decreased over time in a dose-dependent manner for tedizolid, with the 400-mg tedizolid and linezolid groups reporting similar reductions in platelet counts.16
Some evidence is available examining linezolid in neutropenic patients. Rafailidis and colleagues reviewed available literature regarding linezolid in neutropenic patients with Gram-positive infections. Evaluation of linezolid administration at usual doses to 438 neutropenic patients from 2 prospective comparative studies, a prospective cohort study, 2 retrospective studies, and 8 case reports was performed. Results of the evaluation revealed a clinical cure rate between 57% and 87% in the intention-to-treat population of the prospective studies.17 Given the similarities in bacterial spectrum of activity between linezolid and tedizolid, it may be reasonable to infer that tedizolid’s decreased myelosuppression profile would make it useful in the setting of neutropenia in immunocompromised patients.
There is little evidence regarding the use of tedizolid in immunocompromised patients, as only 2 case reports were found. The first described a 60-year-old male postrenal transplant complicated with VRE bacteremia, rhabdomyolysis, and thrombocytopenia. This patient was treated with prolonged tedizolid 200 mg daily due to multiple contraindications for treatment with other antibiotics. The patient was cured with a 14-day course of tedizolid without any noted AEs.18
The second identified case report described the use of tedizolid for the treatment of central nervous system (CNS) manifestations secondary to nocardiosis. Effective treatment of CNS nocardiosis requires high concentrations and prolonged duration of antimicrobial exposure. This case report described a 68-year-old, chronically immunocompromised female patient with multiple myeloma who was hospitalized for 3 months for the treatment of a CNS nocardiosis infection. After discharge, the patient was treated with an oral regimen of 200-mg tedizolid daily in combination with sulfamethoxazole/trimethoprim (800 mg/160 mg) 3 times daily. After 6 months of combination therapy, magnetic resonance imaging revealed complete resolution of nocardiosis-related central lesions. Although the patient’s malignancy advanced during combination antibiotic therapy, the patient’s absolute neutrophil count remained stable and showed an increase in absolute CD4+ cell counts with no other documented AEs.19
Tedizolid is the latest FDA-approved oxazolidinone antibiotic for susceptible Gram-positive acute bacterial skin and skin structure infections. It has a simplified and shorter duration of treatment and imparts similar AEs at improved rates compared with that of linezolid, most notably in relation to hematologic AEs. Due to the lack of established literature and an agreed-upon dosing strategy for the use of tedizolid in immunocompromised patients, tedizolid therapy for Gram-positive infections in immunocompromised patients should be reserved for salvage therapy when more established Gram-positive antibiotic agents lack efficacy or when patient contraindications to their use exist.
1. Fishman JA, Issa NC. Infection in organ transplantation: risk factors and evolving patterns of infection. Infect Dis Clin North Am. 2010;24(2):273-283.
2. Fishman JA. Infection in solid-organ transplant recipients. N Engl J Med. 2007;357(25):2601-2614.
3. Nightingale SD, Byrd LT, Southern PM, Jockusch JD, Cal SX, Wynne BA. Incidence of mycobacterium avium-intracellulare complex bacteremia in human immunodeficiency virus-positive patients. J Infect Dis. 1992;165(6):1082-1085.
4. O’Brien S, Kantarjian H, Beran M, et al. Results of fludarabine and prednisone therapy in 264 patients with chronic lymphocytic leukemia with multivariate analysis-derived prognostic model for response to treatment. Blood. 1993;82(6):1695-1700.
5. Anaissie E, Kontoyiannis DP, Kantarjian H, Elting L, Robertson LE, Keating M. Listeriosis in patients with chronic lymphocytic leukemia who were treated with fludarabine and prednisone. Ann Intern Med. 1992;117(6):466-469.
6. Morrison VA, Rai KR, Peterson BL, et al. Impact of therapy with chlorambucil, fludarabine, or fludarabine plus chlorambucil on infections in patients with chronic lymphocytic leukemia: Intergroup Study Cancer and Leukemia Group B 9011. J Clin Oncol. 2001;19(16):3611-3621.
7. Nucci M, Anaissie E. Infections in patients with multiple myeloma in the era of high-dose therapy and novel agents. Clin Infect Dis. 2009;49(8):1211-1225.
8. Naseer M, Dailey FE, Juboori AA, Samiullah S, Tahan V. Epidemiology, determinants, and managements of AIDS cholangiopathy: a review. World J Gastroenterol. 2018;24(7):767-774.
9. Radunz S, Juntermanns B, Kaiser GM, et al. Efficacy and safety of linezolid in liver transplant patients. Transpl Infect Dis. 2011;13(4):353-358.
10. Roger C, Roberts JA, Muller L. Clinical pharmacokinetics and pharmacodynamics of oxazolidinones. Clin Pharmacokinet. 2018;57(5):559-575.
11. Sivextro [package insert]. Whitehouse Station, NJ: Merck & Co Inc; 2016.
12. Zyvox [package insert]. New York, NY: Pfizer Inc; 2018.
13. Moran GJ, Fang E, Corey GR, Das AF, De Anda C, Prokocimer P. Tedizolid for 6 days versus linezolid for 10 days for acute bacterial skin and skin-structure infections (ESTABLISH-2): a randomized, double-blind, phase 3, non-inferiority trial. Lancet Infect Dis. 2014;14(8):696-705.
14. Liu C, Bayer A, Cosgrove S, et al; Infectious Diseases Society of America. Clinical practice guidelines by the Infectious Diseases Society of America for the treatment of methicillin-resistant Staphylococcus aureus infections in adults and children. Clin Infect Dis. 2011;52(3):e18-e55.
15. Drusano GL, Liu W, Kulawy R, Louie A. Impact of granulocytes on the antimicrobial effect of tedizolid in a mouse thigh infection model. Antimicrob Agents Chemother. 2011;55(11):5300-5305.
16. Lodise TP, Bidell MR, Flanagan SD, Zasowski EJ, Minassian SL, Prokocimer P. Characterization of the haematological profile of 21 days of tedizolid in healthy subjects. J Antimicrob Chemother. 2016;71(9):2553-2558.
17. Rafailidis PI, Kouranos VD, Christodoulou C, Falagas ME. Linezolid for patients with neutropenia: are bacteriostatic agents appropriate? Expert Rev Anti Infect Ther. 2009;7(4):415-422.
18. Sudhindra P, Lee L, Wang G, Dhand A. Tedizolid for treatment of enterococcal bacteremia. Open Forum Infect Dis. 2016;3(suppl 1):1344.
19. Matin A, Sharma S, Mathur P, Apewokin SK. Myelosuppression-sparing treatment of central nervous system nocardiosis in a multiple myeloma patient utilizing a tedizolid-based regimen: a case report. Int J Antimicrob Agents. 2017;49(4):488-492.
20. Dryden MS. Alternative clinical indications for novel antibiotics licensed for skin and soft tissue infections? Curr Opin Infect Dis. 2015;28(2):117-124.
21. Milstein M, Brzezinski A, Varaine F, Mitnick CD. (Re)moving the needle: prospects for all-oral treatment for multidrug-resistant tuberculosis. Int J Tuberc Lung Dis. 2016;20(12):18-23.
22. Winthrop KL, Ku JH, Marras TK, et al. The tolerability of linezolid in the treatment of nontuberculous mycobacterial disease. Eur Respir J. 2015;45(4):1177-1179.
23. Yuste JR, Bertó J, Del Pozo JL, Leiva J. Prolonged use of tedizolid in a pulmonary non-tuberculosis mycobacterial infection after linezlid-induced toxicity. J Antimicrob Chemother. 2017;72(2):625-628.
Immunocompromised patients are often susceptible to opportunistic infections, including those caused by multidrug-resistant organisms (MDROs). Transplant recipients are at high risk for developing infections due to lifelong immunosuppressive therapy.1,2 Additionally, patients receiving chemotherapy and those with HIV and AIDS are in an immunocompromised state.3-8
Regardless of the etiology for immunosuppression, decreased absolute neutrophil and platelet counts are seen in this condition. Although immunosuppressed individuals may be at increased risk of Gram-negative or Gram-positive infections, this review focuses on the treatment of Gram-positive bacterial infections. Of particular concern are opportunistic infections caused by Gram-positive MDROs, including methicillin-resistant Staphylococcus aureus (MRSA), vancomycin-resistant Enterococcus species (VRE), penicillin-resistant Streptococcus pneumoniae, and Nocardia species. Treatment of infections in the immunocompromised patient population warrants careful antimicrobial selection to ensure that a patient’s immune system is not further compromised due to adverse effects (AEs) secondary to therapy. As such, clinicians are exploring alternative antimicrobials, such as tedizolid, to treat various opportunistic infections.
Recently, requests at the William S. Middleton Memorial Veterans Hospital in Madison, Wisconsin, for off-label use of tedizolid have increased despite having other cheaper alternatives with comparable Gram-positive coverage. This review examines available literature regarding off-label use of tedizolid with a focus on use in immunocompromised patients.
Tedizolid phosphate (Sivextro) is an oxazolidinone antibiotic prodrug that joined linezolid as the second in its class in 2014. Oxazolidinones inhibit bacterial protein synthesis by binding to the 50S subunit of bacterial ribosomes in Gram-positive bacteria and are often used to treat MRSA and VRE infections.9 In vitro, oxazolidinones have shown bacteriostatic activity against Enterococcus and Staphylococcus species while exhibiting bactericidal activity against most Streptococcus species.10 Tedizolid has a US Food and Drug Administration (FDA) -approved, simplified dosing profile of 200 mg daily for 6 days compared with linezolid 400 to 600 mg twice daily for 10 to 14 days. Both medications are highly bioavailable with direct IV to oral conversion.11,12 Potential, expanded use of tedizolid against Gram-positive MDROs rests on a more favorable AE profile than does its linezolid predecessor. Tedizolid has been associated with less antibiotic-induced myelosuppression, which could prove valuable for immunocompromised patients.13
Tedizolid is approved for the sole indication of acute bacterial skin and skin structure infections (ABSSSI), whereas its predecessor has many approved indications and has been used extensively for off-label indications (Table).
Adverse reactions, as determined by 2 phase 2 and 2 phase 3 clinical trials evaluating 1,050 patients treated with tedizolid and 662 patients treated with linezolid, were similar between the oxazolidinones. Nausea was the most common AE and was reported in 8% and 12% of patients taking tedizolid and linezolid, respectively. Other common AEs (1%-6%) reported for both agents included vomiting, diarrhea, headache, and dizziness.11 Myelosuppression, peripheral neuropathy, and optic nerve disorders were the most common severe AEs reported with oxazolidinones. Tedizolid demonstrated a significantly decreased incidence of neutropenia (3%), defined by absolute neutrophil count 9/L compared with that of linezolid (7%) (P = .024).13 Evaluation of peripheral neuropathy and optic nerve disorders within the tedizolid and linezolid groups revealed similar incidences (peripheral neuropathy 1.2% vs 0.6%; optic nerve disorders 0.3% vs 0.2%, respectively).11
There is one preclinical trial that described the use of tedizolid in a murine model. A murine model study compared the antistaphylococcal killing effect of doses of tedizolid equivalent to human exposures ranging from 200 to 3,200 mg/d in both granulocytopenic and normal mice. The mice were evaluated at 24, 48, and 72 hours after therapy initiation. The presence of granulocytes had a dramatic effect on the antimicrobial effect of tedizolid. Dose response, demonstrated by the ratio of the area under the curve over the minimum inhibitory concentration, was on average > 25-fold for nonneutropenic vs neutropenic models. Near maximal effect of the nonneutropenic group, irrespective of duration of therapy, was achieved at the lowest dose tested (an exposure of about 200-mg tedizolid phosphate per day in humans).This study suggests that immunocompromised patients may warrant higher doses of tedizolid than the currently FDA-approved dose due to a decreased number of granulocytes available for modulating bacterial infections.15
Use of tedizolid doses higher than that which is FDA-approved may negate the favorable AE profile. A phase 1 clinical study was conducted to evaluate the safety, tolerability, and pharmacokinetics of tedizolid compared with those of linezolid in 40 healthy volunteers in a 21-day multiple ascending dose study.16 Subjects were stratified into 5 treatment cohorts: 200-, 300-, or 400-mg tedizolid orally once a day, 600-mg linezolid orally twice a day, and placebo. Tedizolid given at 200 mg had a hematologic safety profile similar to that of placebo. However, mean platelet counts decreased over time in a dose-dependent manner for tedizolid, with the 400-mg tedizolid and linezolid groups reporting similar reductions in platelet counts.16
Some evidence is available examining linezolid in neutropenic patients. Rafailidis and colleagues reviewed available literature regarding linezolid in neutropenic patients with Gram-positive infections. Evaluation of linezolid administration at usual doses to 438 neutropenic patients from 2 prospective comparative studies, a prospective cohort study, 2 retrospective studies, and 8 case reports was performed. Results of the evaluation revealed a clinical cure rate between 57% and 87% in the intention-to-treat population of the prospective studies.17 Given the similarities in bacterial spectrum of activity between linezolid and tedizolid, it may be reasonable to infer that tedizolid’s decreased myelosuppression profile would make it useful in the setting of neutropenia in immunocompromised patients.
There is little evidence regarding the use of tedizolid in immunocompromised patients, as only 2 case reports were found. The first described a 60-year-old male postrenal transplant complicated with VRE bacteremia, rhabdomyolysis, and thrombocytopenia. This patient was treated with prolonged tedizolid 200 mg daily due to multiple contraindications for treatment with other antibiotics. The patient was cured with a 14-day course of tedizolid without any noted AEs.18
The second identified case report described the use of tedizolid for the treatment of central nervous system (CNS) manifestations secondary to nocardiosis. Effective treatment of CNS nocardiosis requires high concentrations and prolonged duration of antimicrobial exposure. This case report described a 68-year-old, chronically immunocompromised female patient with multiple myeloma who was hospitalized for 3 months for the treatment of a CNS nocardiosis infection. After discharge, the patient was treated with an oral regimen of 200-mg tedizolid daily in combination with sulfamethoxazole/trimethoprim (800 mg/160 mg) 3 times daily. After 6 months of combination therapy, magnetic resonance imaging revealed complete resolution of nocardiosis-related central lesions. Although the patient’s malignancy advanced during combination antibiotic therapy, the patient’s absolute neutrophil count remained stable and showed an increase in absolute CD4+ cell counts with no other documented AEs.19
Tedizolid is the latest FDA-approved oxazolidinone antibiotic for susceptible Gram-positive acute bacterial skin and skin structure infections. It has a simplified and shorter duration of treatment and imparts similar AEs at improved rates compared with that of linezolid, most notably in relation to hematologic AEs. Due to the lack of established literature and an agreed-upon dosing strategy for the use of tedizolid in immunocompromised patients, tedizolid therapy for Gram-positive infections in immunocompromised patients should be reserved for salvage therapy when more established Gram-positive antibiotic agents lack efficacy or when patient contraindications to their use exist.
Immunocompromised patients are often susceptible to opportunistic infections, including those caused by multidrug-resistant organisms (MDROs). Transplant recipients are at high risk for developing infections due to lifelong immunosuppressive therapy.1,2 Additionally, patients receiving chemotherapy and those with HIV and AIDS are in an immunocompromised state.3-8
Regardless of the etiology for immunosuppression, decreased absolute neutrophil and platelet counts are seen in this condition. Although immunosuppressed individuals may be at increased risk of Gram-negative or Gram-positive infections, this review focuses on the treatment of Gram-positive bacterial infections. Of particular concern are opportunistic infections caused by Gram-positive MDROs, including methicillin-resistant Staphylococcus aureus (MRSA), vancomycin-resistant Enterococcus species (VRE), penicillin-resistant Streptococcus pneumoniae, and Nocardia species. Treatment of infections in the immunocompromised patient population warrants careful antimicrobial selection to ensure that a patient’s immune system is not further compromised due to adverse effects (AEs) secondary to therapy. As such, clinicians are exploring alternative antimicrobials, such as tedizolid, to treat various opportunistic infections.
Recently, requests at the William S. Middleton Memorial Veterans Hospital in Madison, Wisconsin, for off-label use of tedizolid have increased despite having other cheaper alternatives with comparable Gram-positive coverage. This review examines available literature regarding off-label use of tedizolid with a focus on use in immunocompromised patients.
Tedizolid phosphate (Sivextro) is an oxazolidinone antibiotic prodrug that joined linezolid as the second in its class in 2014. Oxazolidinones inhibit bacterial protein synthesis by binding to the 50S subunit of bacterial ribosomes in Gram-positive bacteria and are often used to treat MRSA and VRE infections.9 In vitro, oxazolidinones have shown bacteriostatic activity against Enterococcus and Staphylococcus species while exhibiting bactericidal activity against most Streptococcus species.10 Tedizolid has a US Food and Drug Administration (FDA) -approved, simplified dosing profile of 200 mg daily for 6 days compared with linezolid 400 to 600 mg twice daily for 10 to 14 days. Both medications are highly bioavailable with direct IV to oral conversion.11,12 Potential, expanded use of tedizolid against Gram-positive MDROs rests on a more favorable AE profile than does its linezolid predecessor. Tedizolid has been associated with less antibiotic-induced myelosuppression, which could prove valuable for immunocompromised patients.13
Tedizolid is approved for the sole indication of acute bacterial skin and skin structure infections (ABSSSI), whereas its predecessor has many approved indications and has been used extensively for off-label indications (Table).
Adverse reactions, as determined by 2 phase 2 and 2 phase 3 clinical trials evaluating 1,050 patients treated with tedizolid and 662 patients treated with linezolid, were similar between the oxazolidinones. Nausea was the most common AE and was reported in 8% and 12% of patients taking tedizolid and linezolid, respectively. Other common AEs (1%-6%) reported for both agents included vomiting, diarrhea, headache, and dizziness.11 Myelosuppression, peripheral neuropathy, and optic nerve disorders were the most common severe AEs reported with oxazolidinones. Tedizolid demonstrated a significantly decreased incidence of neutropenia (3%), defined by absolute neutrophil count 9/L compared with that of linezolid (7%) (P = .024).13 Evaluation of peripheral neuropathy and optic nerve disorders within the tedizolid and linezolid groups revealed similar incidences (peripheral neuropathy 1.2% vs 0.6%; optic nerve disorders 0.3% vs 0.2%, respectively).11
There is one preclinical trial that described the use of tedizolid in a murine model. A murine model study compared the antistaphylococcal killing effect of doses of tedizolid equivalent to human exposures ranging from 200 to 3,200 mg/d in both granulocytopenic and normal mice. The mice were evaluated at 24, 48, and 72 hours after therapy initiation. The presence of granulocytes had a dramatic effect on the antimicrobial effect of tedizolid. Dose response, demonstrated by the ratio of the area under the curve over the minimum inhibitory concentration, was on average > 25-fold for nonneutropenic vs neutropenic models. Near maximal effect of the nonneutropenic group, irrespective of duration of therapy, was achieved at the lowest dose tested (an exposure of about 200-mg tedizolid phosphate per day in humans).This study suggests that immunocompromised patients may warrant higher doses of tedizolid than the currently FDA-approved dose due to a decreased number of granulocytes available for modulating bacterial infections.15
Use of tedizolid doses higher than that which is FDA-approved may negate the favorable AE profile. A phase 1 clinical study was conducted to evaluate the safety, tolerability, and pharmacokinetics of tedizolid compared with those of linezolid in 40 healthy volunteers in a 21-day multiple ascending dose study.16 Subjects were stratified into 5 treatment cohorts: 200-, 300-, or 400-mg tedizolid orally once a day, 600-mg linezolid orally twice a day, and placebo. Tedizolid given at 200 mg had a hematologic safety profile similar to that of placebo. However, mean platelet counts decreased over time in a dose-dependent manner for tedizolid, with the 400-mg tedizolid and linezolid groups reporting similar reductions in platelet counts.16
Some evidence is available examining linezolid in neutropenic patients. Rafailidis and colleagues reviewed available literature regarding linezolid in neutropenic patients with Gram-positive infections. Evaluation of linezolid administration at usual doses to 438 neutropenic patients from 2 prospective comparative studies, a prospective cohort study, 2 retrospective studies, and 8 case reports was performed. Results of the evaluation revealed a clinical cure rate between 57% and 87% in the intention-to-treat population of the prospective studies.17 Given the similarities in bacterial spectrum of activity between linezolid and tedizolid, it may be reasonable to infer that tedizolid’s decreased myelosuppression profile would make it useful in the setting of neutropenia in immunocompromised patients.
There is little evidence regarding the use of tedizolid in immunocompromised patients, as only 2 case reports were found. The first described a 60-year-old male postrenal transplant complicated with VRE bacteremia, rhabdomyolysis, and thrombocytopenia. This patient was treated with prolonged tedizolid 200 mg daily due to multiple contraindications for treatment with other antibiotics. The patient was cured with a 14-day course of tedizolid without any noted AEs.18
The second identified case report described the use of tedizolid for the treatment of central nervous system (CNS) manifestations secondary to nocardiosis. Effective treatment of CNS nocardiosis requires high concentrations and prolonged duration of antimicrobial exposure. This case report described a 68-year-old, chronically immunocompromised female patient with multiple myeloma who was hospitalized for 3 months for the treatment of a CNS nocardiosis infection. After discharge, the patient was treated with an oral regimen of 200-mg tedizolid daily in combination with sulfamethoxazole/trimethoprim (800 mg/160 mg) 3 times daily. After 6 months of combination therapy, magnetic resonance imaging revealed complete resolution of nocardiosis-related central lesions. Although the patient’s malignancy advanced during combination antibiotic therapy, the patient’s absolute neutrophil count remained stable and showed an increase in absolute CD4+ cell counts with no other documented AEs.19
Tedizolid is the latest FDA-approved oxazolidinone antibiotic for susceptible Gram-positive acute bacterial skin and skin structure infections. It has a simplified and shorter duration of treatment and imparts similar AEs at improved rates compared with that of linezolid, most notably in relation to hematologic AEs. Due to the lack of established literature and an agreed-upon dosing strategy for the use of tedizolid in immunocompromised patients, tedizolid therapy for Gram-positive infections in immunocompromised patients should be reserved for salvage therapy when more established Gram-positive antibiotic agents lack efficacy or when patient contraindications to their use exist.
1. Fishman JA, Issa NC. Infection in organ transplantation: risk factors and evolving patterns of infection. Infect Dis Clin North Am. 2010;24(2):273-283.
2. Fishman JA. Infection in solid-organ transplant recipients. N Engl J Med. 2007;357(25):2601-2614.
3. Nightingale SD, Byrd LT, Southern PM, Jockusch JD, Cal SX, Wynne BA. Incidence of mycobacterium avium-intracellulare complex bacteremia in human immunodeficiency virus-positive patients. J Infect Dis. 1992;165(6):1082-1085.
4. O’Brien S, Kantarjian H, Beran M, et al. Results of fludarabine and prednisone therapy in 264 patients with chronic lymphocytic leukemia with multivariate analysis-derived prognostic model for response to treatment. Blood. 1993;82(6):1695-1700.
5. Anaissie E, Kontoyiannis DP, Kantarjian H, Elting L, Robertson LE, Keating M. Listeriosis in patients with chronic lymphocytic leukemia who were treated with fludarabine and prednisone. Ann Intern Med. 1992;117(6):466-469.
6. Morrison VA, Rai KR, Peterson BL, et al. Impact of therapy with chlorambucil, fludarabine, or fludarabine plus chlorambucil on infections in patients with chronic lymphocytic leukemia: Intergroup Study Cancer and Leukemia Group B 9011. J Clin Oncol. 2001;19(16):3611-3621.
7. Nucci M, Anaissie E. Infections in patients with multiple myeloma in the era of high-dose therapy and novel agents. Clin Infect Dis. 2009;49(8):1211-1225.
8. Naseer M, Dailey FE, Juboori AA, Samiullah S, Tahan V. Epidemiology, determinants, and managements of AIDS cholangiopathy: a review. World J Gastroenterol. 2018;24(7):767-774.
9. Radunz S, Juntermanns B, Kaiser GM, et al. Efficacy and safety of linezolid in liver transplant patients. Transpl Infect Dis. 2011;13(4):353-358.
10. Roger C, Roberts JA, Muller L. Clinical pharmacokinetics and pharmacodynamics of oxazolidinones. Clin Pharmacokinet. 2018;57(5):559-575.
11. Sivextro [package insert]. Whitehouse Station, NJ: Merck & Co Inc; 2016.
12. Zyvox [package insert]. New York, NY: Pfizer Inc; 2018.
13. Moran GJ, Fang E, Corey GR, Das AF, De Anda C, Prokocimer P. Tedizolid for 6 days versus linezolid for 10 days for acute bacterial skin and skin-structure infections (ESTABLISH-2): a randomized, double-blind, phase 3, non-inferiority trial. Lancet Infect Dis. 2014;14(8):696-705.
14. Liu C, Bayer A, Cosgrove S, et al; Infectious Diseases Society of America. Clinical practice guidelines by the Infectious Diseases Society of America for the treatment of methicillin-resistant Staphylococcus aureus infections in adults and children. Clin Infect Dis. 2011;52(3):e18-e55.
15. Drusano GL, Liu W, Kulawy R, Louie A. Impact of granulocytes on the antimicrobial effect of tedizolid in a mouse thigh infection model. Antimicrob Agents Chemother. 2011;55(11):5300-5305.
16. Lodise TP, Bidell MR, Flanagan SD, Zasowski EJ, Minassian SL, Prokocimer P. Characterization of the haematological profile of 21 days of tedizolid in healthy subjects. J Antimicrob Chemother. 2016;71(9):2553-2558.
17. Rafailidis PI, Kouranos VD, Christodoulou C, Falagas ME. Linezolid for patients with neutropenia: are bacteriostatic agents appropriate? Expert Rev Anti Infect Ther. 2009;7(4):415-422.
18. Sudhindra P, Lee L, Wang G, Dhand A. Tedizolid for treatment of enterococcal bacteremia. Open Forum Infect Dis. 2016;3(suppl 1):1344.
19. Matin A, Sharma S, Mathur P, Apewokin SK. Myelosuppression-sparing treatment of central nervous system nocardiosis in a multiple myeloma patient utilizing a tedizolid-based regimen: a case report. Int J Antimicrob Agents. 2017;49(4):488-492.
20. Dryden MS. Alternative clinical indications for novel antibiotics licensed for skin and soft tissue infections? Curr Opin Infect Dis. 2015;28(2):117-124.
21. Milstein M, Brzezinski A, Varaine F, Mitnick CD. (Re)moving the needle: prospects for all-oral treatment for multidrug-resistant tuberculosis. Int J Tuberc Lung Dis. 2016;20(12):18-23.
22. Winthrop KL, Ku JH, Marras TK, et al. The tolerability of linezolid in the treatment of nontuberculous mycobacterial disease. Eur Respir J. 2015;45(4):1177-1179.
23. Yuste JR, Bertó J, Del Pozo JL, Leiva J. Prolonged use of tedizolid in a pulmonary non-tuberculosis mycobacterial infection after linezlid-induced toxicity. J Antimicrob Chemother. 2017;72(2):625-628.
1. Fishman JA, Issa NC. Infection in organ transplantation: risk factors and evolving patterns of infection. Infect Dis Clin North Am. 2010;24(2):273-283.
2. Fishman JA. Infection in solid-organ transplant recipients. N Engl J Med. 2007;357(25):2601-2614.
3. Nightingale SD, Byrd LT, Southern PM, Jockusch JD, Cal SX, Wynne BA. Incidence of mycobacterium avium-intracellulare complex bacteremia in human immunodeficiency virus-positive patients. J Infect Dis. 1992;165(6):1082-1085.
4. O’Brien S, Kantarjian H, Beran M, et al. Results of fludarabine and prednisone therapy in 264 patients with chronic lymphocytic leukemia with multivariate analysis-derived prognostic model for response to treatment. Blood. 1993;82(6):1695-1700.
5. Anaissie E, Kontoyiannis DP, Kantarjian H, Elting L, Robertson LE, Keating M. Listeriosis in patients with chronic lymphocytic leukemia who were treated with fludarabine and prednisone. Ann Intern Med. 1992;117(6):466-469.
6. Morrison VA, Rai KR, Peterson BL, et al. Impact of therapy with chlorambucil, fludarabine, or fludarabine plus chlorambucil on infections in patients with chronic lymphocytic leukemia: Intergroup Study Cancer and Leukemia Group B 9011. J Clin Oncol. 2001;19(16):3611-3621.
7. Nucci M, Anaissie E. Infections in patients with multiple myeloma in the era of high-dose therapy and novel agents. Clin Infect Dis. 2009;49(8):1211-1225.
8. Naseer M, Dailey FE, Juboori AA, Samiullah S, Tahan V. Epidemiology, determinants, and managements of AIDS cholangiopathy: a review. World J Gastroenterol. 2018;24(7):767-774.
9. Radunz S, Juntermanns B, Kaiser GM, et al. Efficacy and safety of linezolid in liver transplant patients. Transpl Infect Dis. 2011;13(4):353-358.
10. Roger C, Roberts JA, Muller L. Clinical pharmacokinetics and pharmacodynamics of oxazolidinones. Clin Pharmacokinet. 2018;57(5):559-575.
11. Sivextro [package insert]. Whitehouse Station, NJ: Merck & Co Inc; 2016.
12. Zyvox [package insert]. New York, NY: Pfizer Inc; 2018.
13. Moran GJ, Fang E, Corey GR, Das AF, De Anda C, Prokocimer P. Tedizolid for 6 days versus linezolid for 10 days for acute bacterial skin and skin-structure infections (ESTABLISH-2): a randomized, double-blind, phase 3, non-inferiority trial. Lancet Infect Dis. 2014;14(8):696-705.
14. Liu C, Bayer A, Cosgrove S, et al; Infectious Diseases Society of America. Clinical practice guidelines by the Infectious Diseases Society of America for the treatment of methicillin-resistant Staphylococcus aureus infections in adults and children. Clin Infect Dis. 2011;52(3):e18-e55.
15. Drusano GL, Liu W, Kulawy R, Louie A. Impact of granulocytes on the antimicrobial effect of tedizolid in a mouse thigh infection model. Antimicrob Agents Chemother. 2011;55(11):5300-5305.
16. Lodise TP, Bidell MR, Flanagan SD, Zasowski EJ, Minassian SL, Prokocimer P. Characterization of the haematological profile of 21 days of tedizolid in healthy subjects. J Antimicrob Chemother. 2016;71(9):2553-2558.
17. Rafailidis PI, Kouranos VD, Christodoulou C, Falagas ME. Linezolid for patients with neutropenia: are bacteriostatic agents appropriate? Expert Rev Anti Infect Ther. 2009;7(4):415-422.
18. Sudhindra P, Lee L, Wang G, Dhand A. Tedizolid for treatment of enterococcal bacteremia. Open Forum Infect Dis. 2016;3(suppl 1):1344.
19. Matin A, Sharma S, Mathur P, Apewokin SK. Myelosuppression-sparing treatment of central nervous system nocardiosis in a multiple myeloma patient utilizing a tedizolid-based regimen: a case report. Int J Antimicrob Agents. 2017;49(4):488-492.
20. Dryden MS. Alternative clinical indications for novel antibiotics licensed for skin and soft tissue infections? Curr Opin Infect Dis. 2015;28(2):117-124.
21. Milstein M, Brzezinski A, Varaine F, Mitnick CD. (Re)moving the needle: prospects for all-oral treatment for multidrug-resistant tuberculosis. Int J Tuberc Lung Dis. 2016;20(12):18-23.
22. Winthrop KL, Ku JH, Marras TK, et al. The tolerability of linezolid in the treatment of nontuberculous mycobacterial disease. Eur Respir J. 2015;45(4):1177-1179.
23. Yuste JR, Bertó J, Del Pozo JL, Leiva J. Prolonged use of tedizolid in a pulmonary non-tuberculosis mycobacterial infection after linezlid-induced toxicity. J Antimicrob Chemother. 2017;72(2):625-628.
Risk Factors Associated With Multidrug-Resistant Pneumonia in Nonhospitalized Patients
Successful treatment of pneumonia depends on timely diagnosis and administration of antibiotics. Multidrug-resistant organisms (MDROs) complicate antibiotic therapies by rendering some antibiotic agents ineffective. Inappropriate initial therapy has been associated with a more than 2-fold increase in the risk of mortality.1 Because culture results are not available immediately, clinicians prescribe antibiotics empirically and must rely on guidelines and knowledge of risk factors associated with MDRO infection to make these selections.
Treatment guidelines exist for hospital-acquired and ventilator-associated pneumonia (HAP/VAP) and community-acquired pneumonia (CAP) to assist with empiric antibiotic selection. For HAP/VAP, 2 to 3 antibiotics with a broad-spectrum of activity are used due to increased prevalence of MDROs in hospitals, whereastreatment of CAP involves more narrow coverage because bacteria that cause this infection typically have fewer antibiotic resistances.2,3 The HAP/VAP guidelines stratify the risk of pneumonia due to the presence of a MDRO acquired during a hospitalization. However, neither the CAP nor HAP/VAP guidelines offer risk-stratification guidance for nonhospitalized patients who develop pneumonia but who may have become colonized with a MDRO during a previous hospitalization or from another exposure to a health care facility.
Health care-associated pneumonia (HCAP) was first described in the 2005 American Thoracic Society and the Infectious Diseases Society of America (ATS/IDSA) nosocomial pneumonia guidelines and was associated with criteria intended to aid clinician identification of nonhospitalized patients at risk for MDRO pneumonia, which warranted empiric broad-spectrum antibiotic therapy.2 According to these guidelines, patients were classified as having HCAP if they had been hospitalized for at least 48 hours in the past 90 days, admitted from a nursing home, received recent intravenous antibiotics, had hemodialysis in the past 30 days, had a history of home infusion therapy or wound care, received intravenous chemotherapy, or had a family member with MDRO colonization.
Since publication of the 2005 guidelines, HCAP has been criticized as being a poor predictor of MDRO infection. A 2014 meta-analysis of 24 studies investigated the discriminating ability of HCAP and reported that the specificity and sensitivity for MDRO infections was 71.2% and 53.7%, respectively.3 In 2016, the ATS/IDSA guidelines were updated to remove HCAP due to the risk of antibiotic overprescribing.4
Literature Review
Although criteria previously defining a patient as having HCAP have been shown to be a poor discriminator of MDRO pneumonia as a whole, MDRO infections still pose a threat to nonhospitalized patients who have exposure to the health care system. A literature review was performed to identify independent HCAP risk factors that may increase the risk of MDRO pneumonia infecting a nonhospitalized patient needing empiric broad-spectrum antibiotic therapy. All included studies were prospective or retrospective observational cohort studies that performed logistic regression analyses to assess the association between MDRO isolation and the previously defined HCAP risk factors (Table 1).
Five studies examined the risk of MDRO infection in patients with a previous hospital admission of 2 days or more in the past 90 days. Shindo and colleagues found a significant increase in MDRO infections by about 2-fold (adjusted odds ratio [AOR], 2.1; 95% confidence interval [CI], 1.2-3.4).5 Shorr and colleagues found a 4-fold increase in likelihood of identifying a MDRO in HCAP (AOR, 4.2; 95% CI, 2.9-6.3).6 Nseir and colleagues and Jung and colleagues found similar results (AOR 3.9, 95% CI 1.7-8.8; AOR 2.7, 95% CI 1.3-5.5, respectively).7,8 Conflicting results were reported by Gross and colleagues who did not find a significant relationship between previous hospitalization and MDRO isolation (AOR 1.2, 95% CI, 0.5-3.2).9
In patients with pneumonia admitted from a nursing home, MDRO infection risk also was evaluated in these 5 studies. Shorr and colleagues, Nseir and colleagues, and Gross and colleagues found significant AORs of 2.7 (95% CI 1.7-4.3), 2.0 (95% CI 1.1-3.7), and 4.2 (95% CI 1.6-11.3), respectively.6,7,9 Shindo and colleagues (AOR 1.1; 95% CI, 0.6-2.0) and Jung and colleagues (AOR 1.9, 95% CI, 0.5-6.9) found this risk factor not significant.5
Receipt of antibiotics within the previous 90 days was assessed in 3 studies. Shindo and colleagues, Nseir and colleagues, and Gross and colleagues all found significant AORs of 2.5 (95% CI 1.2-4.0), 2.3 (95% CI 1.2-4.3), and 2.9 (95% CI 1.1-7.5), respectively.5,7,9 Antibiotic therapy within the previous 90 days is an established risk factor for MDRO pneumonia, and the 2016 ATS/IDSA guidelines consider this a risk factor for HAP and VAP, including pneumonia caused by methicillin resistant Staphylococcus aureus and Pseudomonas aeruginosa.4
The impact of hemodialysis in the previous month on acquisition of MDRO pneumonia was investigated in 4 studies. Shindo and colleagues, Jung and colleagues, and Gross and colleagues concluded that this risk factor was not significantly related to MDRO infection, reporting AORs of 2.2 (95% CI 0.5-9.7), 2.8 (95% CI 0.9-9.2) and 0.7 (95% CI 0.1-5.1), respectively.5,8,9 Shorr and colleagues, however, found a significant AOR of 2.1 (95% CI 1.0-4.3).6
Shindo and colleagues investigated the impact of home infusion therapy on acquisition of pneumonia due to a MDRO and reported a nonsignificant AOR of 0.8 (95% CI 0.4-1.8).5 Gross and colleagues also found a nonsignificant AOR of 0 (P = .1).9 In the Shindo and colleagues study, resistance was found in 107 of 679 patients who did not receive infusion therapy, and 12 of 55 patients who were receiving infusion therapy.5 Gross and colleagues reported that home-infusion therapy was received by 0 of 20 patients with MDRO infection and 4 of the 501 patients without MDRO infection.9
Shindo and colleagues reported that home wound care was not found to be significantly related to MDRO pneumonia as well as did Gross and colleagues: AORs of 3.8 (0.8-18.4) and 1.4 (95% CI 0.5-4.4), respectively.5,9 Jung and colleagues examined IV chemotherapy in the past 30 days, and found this to not significantly impact the odds of MDRO isolation (AOR = 0.62, 95% CI 0.2-1.8).8 No data were available reflecting the risk of a family member with a MDRO.
Limitations
The variables on which logistic regression were performed differed among the studies. Therefore, results cannot be averaged or compared quantitatively, as AORs varied, depending on the variables included. In addition, data were drawn from multiple geographic locations that may impact MDRO prevalence within each patient population. Finally, this review examines the utility of the risk factors formerly included in HCAP. However, other risk factors for MDRO pneumonia outlined by the ATS/IDSA guidelines still should be considered when evaluating patient risk. The 2016 guidelines recommend local incidence of resistant strains be considered when initiating empiric therapy. Review of medical records for previous positive cultures and duration of current hospitalization also should be considered. Although the 2016 ATS/IDSA HAP guidelines are not intended for immunosuppressed patients, this risk factor also may be taken into account.
Conclusion
Review and synthesis of published literature found previous hospital admission (of ≥ 2 days in the past 90 days), admission from a nursing home, and IV antibiotic therapy in the last 90 days to be independent risk factors for identification of MDRO pneumonia in previously nonhospitalized patients (Table 2). Additionally, although no data were found to support this risk factor, existence of an in-home (close contact) source of MDROs would provide ample opportunity for transmission, so evaluation of known exposure to MDROs from contacts should be considered. When choosing empiric antibiotic therapy for patients admitted to the hospital for treatment of pneumonia, consideration of patient history and risk factors that may contribute to infection with a MDRO are recommended.
1. Kollef MH, Sherman G, Ward S, Fraser VJ. Inadequate antimicrobial treatment of infections: a risk factor for hospital mortality among critically ill patients. Chest. 1999;115(2):462-474.
2. American Thoracic Society; Infectious Diseases Society of America. Guidelines for the management of adults with hospital-acquired, ventilator-associated, and healthcare-associated pneumonia. Am J Respir Crit Care Med. 2005;171(4):388-416.
3. Chalmers JD, Rother C, Salih W, Ewig S. Healthcare-associated pneumonia does not accurately identify potentially resistant pathogens: a systematic review and meta-analysis. Clin Infect Dis. 2014;58(3):330-339.
4. Kalil AC, Metersky ML, Klompas M, et al. Management of adults with hospital-acquired and ventilator-associated pneumonia: 2016 clinical practice guidelines by the Infectious Diseases Society of America and the American Thoracic Society. Clin Infect Dis. 2016;63(5):e61-e111.
5. Shindo Y, Ito R, Kobayashi D, et al. Risk factors for drug-resistant pathogens in community-acquired and healthcare-associated pneumonia. Am J Respir Crit Care Med. 2013;188(8):985-995.
6. Shorr AF, Zilberberg MD, Micek ST, Kollef MH. Prediction of infection due to antibiotic-resistant bacteria by select risk factors for health care–associated pneumonia. Arch Intern Med. 2008;168(20):2205-2210.
7. Nseir S, Grailles G, Soury-Lavergne A, Minacori F, Alves I, Durocher A. Accuracy of American Thoracic Society/Infectious Diseases Society of America criteria in predicting infection or colonization with multidrug-resistant bacteria at intensive-care unit admission. Clin Microbiol Infect. 2010;16(7):902-908.
8. Jung JY, Park MS, Kim YS, et al. Healthcare-associated pneumonia among hospitalized patients in a Korean tertiary hospital. BMC Infectious Diseases. 2011;11:61.
9. Gross AE, Van Schooneveld TC, Olsen KM, et al. Epidemiology and predictors of multidrug-resistant community-acquired and health care-associated pneumonia. Antimicrob Agents Chemother. 2014;58(9):5262-5268.
Successful treatment of pneumonia depends on timely diagnosis and administration of antibiotics. Multidrug-resistant organisms (MDROs) complicate antibiotic therapies by rendering some antibiotic agents ineffective. Inappropriate initial therapy has been associated with a more than 2-fold increase in the risk of mortality.1 Because culture results are not available immediately, clinicians prescribe antibiotics empirically and must rely on guidelines and knowledge of risk factors associated with MDRO infection to make these selections.
Treatment guidelines exist for hospital-acquired and ventilator-associated pneumonia (HAP/VAP) and community-acquired pneumonia (CAP) to assist with empiric antibiotic selection. For HAP/VAP, 2 to 3 antibiotics with a broad-spectrum of activity are used due to increased prevalence of MDROs in hospitals, whereastreatment of CAP involves more narrow coverage because bacteria that cause this infection typically have fewer antibiotic resistances.2,3 The HAP/VAP guidelines stratify the risk of pneumonia due to the presence of a MDRO acquired during a hospitalization. However, neither the CAP nor HAP/VAP guidelines offer risk-stratification guidance for nonhospitalized patients who develop pneumonia but who may have become colonized with a MDRO during a previous hospitalization or from another exposure to a health care facility.
Health care-associated pneumonia (HCAP) was first described in the 2005 American Thoracic Society and the Infectious Diseases Society of America (ATS/IDSA) nosocomial pneumonia guidelines and was associated with criteria intended to aid clinician identification of nonhospitalized patients at risk for MDRO pneumonia, which warranted empiric broad-spectrum antibiotic therapy.2 According to these guidelines, patients were classified as having HCAP if they had been hospitalized for at least 48 hours in the past 90 days, admitted from a nursing home, received recent intravenous antibiotics, had hemodialysis in the past 30 days, had a history of home infusion therapy or wound care, received intravenous chemotherapy, or had a family member with MDRO colonization.
Since publication of the 2005 guidelines, HCAP has been criticized as being a poor predictor of MDRO infection. A 2014 meta-analysis of 24 studies investigated the discriminating ability of HCAP and reported that the specificity and sensitivity for MDRO infections was 71.2% and 53.7%, respectively.3 In 2016, the ATS/IDSA guidelines were updated to remove HCAP due to the risk of antibiotic overprescribing.4
Literature Review
Although criteria previously defining a patient as having HCAP have been shown to be a poor discriminator of MDRO pneumonia as a whole, MDRO infections still pose a threat to nonhospitalized patients who have exposure to the health care system. A literature review was performed to identify independent HCAP risk factors that may increase the risk of MDRO pneumonia infecting a nonhospitalized patient needing empiric broad-spectrum antibiotic therapy. All included studies were prospective or retrospective observational cohort studies that performed logistic regression analyses to assess the association between MDRO isolation and the previously defined HCAP risk factors (Table 1).
Five studies examined the risk of MDRO infection in patients with a previous hospital admission of 2 days or more in the past 90 days. Shindo and colleagues found a significant increase in MDRO infections by about 2-fold (adjusted odds ratio [AOR], 2.1; 95% confidence interval [CI], 1.2-3.4).5 Shorr and colleagues found a 4-fold increase in likelihood of identifying a MDRO in HCAP (AOR, 4.2; 95% CI, 2.9-6.3).6 Nseir and colleagues and Jung and colleagues found similar results (AOR 3.9, 95% CI 1.7-8.8; AOR 2.7, 95% CI 1.3-5.5, respectively).7,8 Conflicting results were reported by Gross and colleagues who did not find a significant relationship between previous hospitalization and MDRO isolation (AOR 1.2, 95% CI, 0.5-3.2).9
In patients with pneumonia admitted from a nursing home, MDRO infection risk also was evaluated in these 5 studies. Shorr and colleagues, Nseir and colleagues, and Gross and colleagues found significant AORs of 2.7 (95% CI 1.7-4.3), 2.0 (95% CI 1.1-3.7), and 4.2 (95% CI 1.6-11.3), respectively.6,7,9 Shindo and colleagues (AOR 1.1; 95% CI, 0.6-2.0) and Jung and colleagues (AOR 1.9, 95% CI, 0.5-6.9) found this risk factor not significant.5
Receipt of antibiotics within the previous 90 days was assessed in 3 studies. Shindo and colleagues, Nseir and colleagues, and Gross and colleagues all found significant AORs of 2.5 (95% CI 1.2-4.0), 2.3 (95% CI 1.2-4.3), and 2.9 (95% CI 1.1-7.5), respectively.5,7,9 Antibiotic therapy within the previous 90 days is an established risk factor for MDRO pneumonia, and the 2016 ATS/IDSA guidelines consider this a risk factor for HAP and VAP, including pneumonia caused by methicillin resistant Staphylococcus aureus and Pseudomonas aeruginosa.4
The impact of hemodialysis in the previous month on acquisition of MDRO pneumonia was investigated in 4 studies. Shindo and colleagues, Jung and colleagues, and Gross and colleagues concluded that this risk factor was not significantly related to MDRO infection, reporting AORs of 2.2 (95% CI 0.5-9.7), 2.8 (95% CI 0.9-9.2) and 0.7 (95% CI 0.1-5.1), respectively.5,8,9 Shorr and colleagues, however, found a significant AOR of 2.1 (95% CI 1.0-4.3).6
Shindo and colleagues investigated the impact of home infusion therapy on acquisition of pneumonia due to a MDRO and reported a nonsignificant AOR of 0.8 (95% CI 0.4-1.8).5 Gross and colleagues also found a nonsignificant AOR of 0 (P = .1).9 In the Shindo and colleagues study, resistance was found in 107 of 679 patients who did not receive infusion therapy, and 12 of 55 patients who were receiving infusion therapy.5 Gross and colleagues reported that home-infusion therapy was received by 0 of 20 patients with MDRO infection and 4 of the 501 patients without MDRO infection.9
Shindo and colleagues reported that home wound care was not found to be significantly related to MDRO pneumonia as well as did Gross and colleagues: AORs of 3.8 (0.8-18.4) and 1.4 (95% CI 0.5-4.4), respectively.5,9 Jung and colleagues examined IV chemotherapy in the past 30 days, and found this to not significantly impact the odds of MDRO isolation (AOR = 0.62, 95% CI 0.2-1.8).8 No data were available reflecting the risk of a family member with a MDRO.
Limitations
The variables on which logistic regression were performed differed among the studies. Therefore, results cannot be averaged or compared quantitatively, as AORs varied, depending on the variables included. In addition, data were drawn from multiple geographic locations that may impact MDRO prevalence within each patient population. Finally, this review examines the utility of the risk factors formerly included in HCAP. However, other risk factors for MDRO pneumonia outlined by the ATS/IDSA guidelines still should be considered when evaluating patient risk. The 2016 guidelines recommend local incidence of resistant strains be considered when initiating empiric therapy. Review of medical records for previous positive cultures and duration of current hospitalization also should be considered. Although the 2016 ATS/IDSA HAP guidelines are not intended for immunosuppressed patients, this risk factor also may be taken into account.
Conclusion
Review and synthesis of published literature found previous hospital admission (of ≥ 2 days in the past 90 days), admission from a nursing home, and IV antibiotic therapy in the last 90 days to be independent risk factors for identification of MDRO pneumonia in previously nonhospitalized patients (Table 2). Additionally, although no data were found to support this risk factor, existence of an in-home (close contact) source of MDROs would provide ample opportunity for transmission, so evaluation of known exposure to MDROs from contacts should be considered. When choosing empiric antibiotic therapy for patients admitted to the hospital for treatment of pneumonia, consideration of patient history and risk factors that may contribute to infection with a MDRO are recommended.
Successful treatment of pneumonia depends on timely diagnosis and administration of antibiotics. Multidrug-resistant organisms (MDROs) complicate antibiotic therapies by rendering some antibiotic agents ineffective. Inappropriate initial therapy has been associated with a more than 2-fold increase in the risk of mortality.1 Because culture results are not available immediately, clinicians prescribe antibiotics empirically and must rely on guidelines and knowledge of risk factors associated with MDRO infection to make these selections.
Treatment guidelines exist for hospital-acquired and ventilator-associated pneumonia (HAP/VAP) and community-acquired pneumonia (CAP) to assist with empiric antibiotic selection. For HAP/VAP, 2 to 3 antibiotics with a broad-spectrum of activity are used due to increased prevalence of MDROs in hospitals, whereastreatment of CAP involves more narrow coverage because bacteria that cause this infection typically have fewer antibiotic resistances.2,3 The HAP/VAP guidelines stratify the risk of pneumonia due to the presence of a MDRO acquired during a hospitalization. However, neither the CAP nor HAP/VAP guidelines offer risk-stratification guidance for nonhospitalized patients who develop pneumonia but who may have become colonized with a MDRO during a previous hospitalization or from another exposure to a health care facility.
Health care-associated pneumonia (HCAP) was first described in the 2005 American Thoracic Society and the Infectious Diseases Society of America (ATS/IDSA) nosocomial pneumonia guidelines and was associated with criteria intended to aid clinician identification of nonhospitalized patients at risk for MDRO pneumonia, which warranted empiric broad-spectrum antibiotic therapy.2 According to these guidelines, patients were classified as having HCAP if they had been hospitalized for at least 48 hours in the past 90 days, admitted from a nursing home, received recent intravenous antibiotics, had hemodialysis in the past 30 days, had a history of home infusion therapy or wound care, received intravenous chemotherapy, or had a family member with MDRO colonization.
Since publication of the 2005 guidelines, HCAP has been criticized as being a poor predictor of MDRO infection. A 2014 meta-analysis of 24 studies investigated the discriminating ability of HCAP and reported that the specificity and sensitivity for MDRO infections was 71.2% and 53.7%, respectively.3 In 2016, the ATS/IDSA guidelines were updated to remove HCAP due to the risk of antibiotic overprescribing.4
Literature Review
Although criteria previously defining a patient as having HCAP have been shown to be a poor discriminator of MDRO pneumonia as a whole, MDRO infections still pose a threat to nonhospitalized patients who have exposure to the health care system. A literature review was performed to identify independent HCAP risk factors that may increase the risk of MDRO pneumonia infecting a nonhospitalized patient needing empiric broad-spectrum antibiotic therapy. All included studies were prospective or retrospective observational cohort studies that performed logistic regression analyses to assess the association between MDRO isolation and the previously defined HCAP risk factors (Table 1).
Five studies examined the risk of MDRO infection in patients with a previous hospital admission of 2 days or more in the past 90 days. Shindo and colleagues found a significant increase in MDRO infections by about 2-fold (adjusted odds ratio [AOR], 2.1; 95% confidence interval [CI], 1.2-3.4).5 Shorr and colleagues found a 4-fold increase in likelihood of identifying a MDRO in HCAP (AOR, 4.2; 95% CI, 2.9-6.3).6 Nseir and colleagues and Jung and colleagues found similar results (AOR 3.9, 95% CI 1.7-8.8; AOR 2.7, 95% CI 1.3-5.5, respectively).7,8 Conflicting results were reported by Gross and colleagues who did not find a significant relationship between previous hospitalization and MDRO isolation (AOR 1.2, 95% CI, 0.5-3.2).9
In patients with pneumonia admitted from a nursing home, MDRO infection risk also was evaluated in these 5 studies. Shorr and colleagues, Nseir and colleagues, and Gross and colleagues found significant AORs of 2.7 (95% CI 1.7-4.3), 2.0 (95% CI 1.1-3.7), and 4.2 (95% CI 1.6-11.3), respectively.6,7,9 Shindo and colleagues (AOR 1.1; 95% CI, 0.6-2.0) and Jung and colleagues (AOR 1.9, 95% CI, 0.5-6.9) found this risk factor not significant.5
Receipt of antibiotics within the previous 90 days was assessed in 3 studies. Shindo and colleagues, Nseir and colleagues, and Gross and colleagues all found significant AORs of 2.5 (95% CI 1.2-4.0), 2.3 (95% CI 1.2-4.3), and 2.9 (95% CI 1.1-7.5), respectively.5,7,9 Antibiotic therapy within the previous 90 days is an established risk factor for MDRO pneumonia, and the 2016 ATS/IDSA guidelines consider this a risk factor for HAP and VAP, including pneumonia caused by methicillin resistant Staphylococcus aureus and Pseudomonas aeruginosa.4
The impact of hemodialysis in the previous month on acquisition of MDRO pneumonia was investigated in 4 studies. Shindo and colleagues, Jung and colleagues, and Gross and colleagues concluded that this risk factor was not significantly related to MDRO infection, reporting AORs of 2.2 (95% CI 0.5-9.7), 2.8 (95% CI 0.9-9.2) and 0.7 (95% CI 0.1-5.1), respectively.5,8,9 Shorr and colleagues, however, found a significant AOR of 2.1 (95% CI 1.0-4.3).6
Shindo and colleagues investigated the impact of home infusion therapy on acquisition of pneumonia due to a MDRO and reported a nonsignificant AOR of 0.8 (95% CI 0.4-1.8).5 Gross and colleagues also found a nonsignificant AOR of 0 (P = .1).9 In the Shindo and colleagues study, resistance was found in 107 of 679 patients who did not receive infusion therapy, and 12 of 55 patients who were receiving infusion therapy.5 Gross and colleagues reported that home-infusion therapy was received by 0 of 20 patients with MDRO infection and 4 of the 501 patients without MDRO infection.9
Shindo and colleagues reported that home wound care was not found to be significantly related to MDRO pneumonia as well as did Gross and colleagues: AORs of 3.8 (0.8-18.4) and 1.4 (95% CI 0.5-4.4), respectively.5,9 Jung and colleagues examined IV chemotherapy in the past 30 days, and found this to not significantly impact the odds of MDRO isolation (AOR = 0.62, 95% CI 0.2-1.8).8 No data were available reflecting the risk of a family member with a MDRO.
Limitations
The variables on which logistic regression were performed differed among the studies. Therefore, results cannot be averaged or compared quantitatively, as AORs varied, depending on the variables included. In addition, data were drawn from multiple geographic locations that may impact MDRO prevalence within each patient population. Finally, this review examines the utility of the risk factors formerly included in HCAP. However, other risk factors for MDRO pneumonia outlined by the ATS/IDSA guidelines still should be considered when evaluating patient risk. The 2016 guidelines recommend local incidence of resistant strains be considered when initiating empiric therapy. Review of medical records for previous positive cultures and duration of current hospitalization also should be considered. Although the 2016 ATS/IDSA HAP guidelines are not intended for immunosuppressed patients, this risk factor also may be taken into account.
Conclusion
Review and synthesis of published literature found previous hospital admission (of ≥ 2 days in the past 90 days), admission from a nursing home, and IV antibiotic therapy in the last 90 days to be independent risk factors for identification of MDRO pneumonia in previously nonhospitalized patients (Table 2). Additionally, although no data were found to support this risk factor, existence of an in-home (close contact) source of MDROs would provide ample opportunity for transmission, so evaluation of known exposure to MDROs from contacts should be considered. When choosing empiric antibiotic therapy for patients admitted to the hospital for treatment of pneumonia, consideration of patient history and risk factors that may contribute to infection with a MDRO are recommended.
1. Kollef MH, Sherman G, Ward S, Fraser VJ. Inadequate antimicrobial treatment of infections: a risk factor for hospital mortality among critically ill patients. Chest. 1999;115(2):462-474.
2. American Thoracic Society; Infectious Diseases Society of America. Guidelines for the management of adults with hospital-acquired, ventilator-associated, and healthcare-associated pneumonia. Am J Respir Crit Care Med. 2005;171(4):388-416.
3. Chalmers JD, Rother C, Salih W, Ewig S. Healthcare-associated pneumonia does not accurately identify potentially resistant pathogens: a systematic review and meta-analysis. Clin Infect Dis. 2014;58(3):330-339.
4. Kalil AC, Metersky ML, Klompas M, et al. Management of adults with hospital-acquired and ventilator-associated pneumonia: 2016 clinical practice guidelines by the Infectious Diseases Society of America and the American Thoracic Society. Clin Infect Dis. 2016;63(5):e61-e111.
5. Shindo Y, Ito R, Kobayashi D, et al. Risk factors for drug-resistant pathogens in community-acquired and healthcare-associated pneumonia. Am J Respir Crit Care Med. 2013;188(8):985-995.
6. Shorr AF, Zilberberg MD, Micek ST, Kollef MH. Prediction of infection due to antibiotic-resistant bacteria by select risk factors for health care–associated pneumonia. Arch Intern Med. 2008;168(20):2205-2210.
7. Nseir S, Grailles G, Soury-Lavergne A, Minacori F, Alves I, Durocher A. Accuracy of American Thoracic Society/Infectious Diseases Society of America criteria in predicting infection or colonization with multidrug-resistant bacteria at intensive-care unit admission. Clin Microbiol Infect. 2010;16(7):902-908.
8. Jung JY, Park MS, Kim YS, et al. Healthcare-associated pneumonia among hospitalized patients in a Korean tertiary hospital. BMC Infectious Diseases. 2011;11:61.
9. Gross AE, Van Schooneveld TC, Olsen KM, et al. Epidemiology and predictors of multidrug-resistant community-acquired and health care-associated pneumonia. Antimicrob Agents Chemother. 2014;58(9):5262-5268.
1. Kollef MH, Sherman G, Ward S, Fraser VJ. Inadequate antimicrobial treatment of infections: a risk factor for hospital mortality among critically ill patients. Chest. 1999;115(2):462-474.
2. American Thoracic Society; Infectious Diseases Society of America. Guidelines for the management of adults with hospital-acquired, ventilator-associated, and healthcare-associated pneumonia. Am J Respir Crit Care Med. 2005;171(4):388-416.
3. Chalmers JD, Rother C, Salih W, Ewig S. Healthcare-associated pneumonia does not accurately identify potentially resistant pathogens: a systematic review and meta-analysis. Clin Infect Dis. 2014;58(3):330-339.
4. Kalil AC, Metersky ML, Klompas M, et al. Management of adults with hospital-acquired and ventilator-associated pneumonia: 2016 clinical practice guidelines by the Infectious Diseases Society of America and the American Thoracic Society. Clin Infect Dis. 2016;63(5):e61-e111.
5. Shindo Y, Ito R, Kobayashi D, et al. Risk factors for drug-resistant pathogens in community-acquired and healthcare-associated pneumonia. Am J Respir Crit Care Med. 2013;188(8):985-995.
6. Shorr AF, Zilberberg MD, Micek ST, Kollef MH. Prediction of infection due to antibiotic-resistant bacteria by select risk factors for health care–associated pneumonia. Arch Intern Med. 2008;168(20):2205-2210.
7. Nseir S, Grailles G, Soury-Lavergne A, Minacori F, Alves I, Durocher A. Accuracy of American Thoracic Society/Infectious Diseases Society of America criteria in predicting infection or colonization with multidrug-resistant bacteria at intensive-care unit admission. Clin Microbiol Infect. 2010;16(7):902-908.
8. Jung JY, Park MS, Kim YS, et al. Healthcare-associated pneumonia among hospitalized patients in a Korean tertiary hospital. BMC Infectious Diseases. 2011;11:61.
9. Gross AE, Van Schooneveld TC, Olsen KM, et al. Epidemiology and predictors of multidrug-resistant community-acquired and health care-associated pneumonia. Antimicrob Agents Chemother. 2014;58(9):5262-5268.
The Role of Methicillin-Resistant Staphylococcus aureus Polymerase Chain Reaction Nasal Swabs in Clinical Decision Making
Methicillin-resistant Staphylococcus aureus (MRSA) is a Gram positive, round bacterium. The bacteria has evolved to withstand attacks from antibiotics and has made MRSA resistant to traditional antibiotics, such as β-lactams, resulting in difficult-to-treat infections. The presence of a genetic mutation within the mecA gene, which codes for the penicillin-binding protein 2a (PBP2a), differentiates MRSA from methicillin-susceptible Staphylococcus aureus (MSSA). Presence of the PBP2a protein allows Staphylococcus aureus (S aureus)to overcome β-lactam antibiotics’ method of killing by allowing the bacteria to continue to divide and grow.
β-lactam antibiotics cause cell death in susceptible isolates by binding to penicillin-binding proteins, which inhibits transpeptidation within the cell wall via inactivation of the penicillin-binding protein. By inhibiting cell wall synthesis, the cell loses its integrity and leaks its contents, causing cell death. Penicillin-binding protein 2a is a modified protein that has a low affinity for β-lactam antibiotics, allowing MRSA to survive and making it dangerous and difficult to eradicate.
First described in 1961, MRSA’s prevalence steadily increased in the following decades. It is the most common cause of skin and soft tissue infections presenting to emergency departments in the U.S.1 About 20% of bloodstream infections are caused by S aureus, and in 2003, nearly two-thirds of hospital-onset S aureus infections were methicillin-resistant in U.S. intensive-care units (ICUs).2 It has been shown that patients with MRSA bacteremia have worse overall outcomes, including increased mortality, greater lengths of stay, and increased costs, compared with those with MSSA infections.2,3 In 2011, MRSA infections caused an estimated 11,000 deaths, making fast and accurate detection of MRSA a crucial step in appropriate antimicrobial therapy selection.4
Currently, the Clinical and Laboratory Standards Institute (CLSI) recommends testing for MRSA by using phenotypic or genotypic methods. Phenotypic methods test for the observable characteristics of an organism, whereas a genotypic method identifies the specific gene that the organism carries. Recommended phenotypic methods include the latex agglutination test for PBP2a, the cefoxitin disk screen test, and a plate containing 6 μg/mL of oxacillin in Mueller-Hinton agar supplemented with sodium chloride.5 These methods have varying sensitivity and specificity and take between 48 to 72 hours to provide a result.
Within the past 15 years, a newer, genotypic, method of MRSA detection was approved by the FDA with high sensitivity and specificity. This method uses polymerase chain reaction (PCR) to identify the mecA gene. Polymerase chain reaction is a technique used to copy and amplify a specific segment of DNA, making thousands to millions of copies. If present, the MRSA PCR amplifies the mecA gene that makes S aureus resistant to methicillin and other β-lactams, which confirms that the specimen contains MRSA. The FDA has approved the use of MRSA PCR nasal swabs to detect MRSA in patients at risk of nasal colonization. While previously discussed methods may take between 2 and 3 days to confirm presence of MRSA, PCR can identify MRSA in about 1 hour.6
If a S aureus infection is suspected, empiric therapy often includes coverage of both MSSA and MRSA, due to the high morbidity and mortality associated with these infections. However, continuing an unneeded or unduly broad antibiotic, such as those that cover MRSA, can cause unintended consequences, such as toxicities, emerging resistance, or selection for pathogenic organisms.7 Therefore, empiric broad antibiotic therapy should be de-escalated as soon as possible, which further emphasizes the need for quick and accurate detection of the infecting organism. De-escalation of therapy can lead to a shorter length of stay and decreased mortality.8,9 Conversely, quick identification of infections caused by MRSA would allow therapy to be broadened to cover MRSA in infected patients, which could potentially decrease patient morbidity and mortality.
Nasal MRSA PCR Colonization
Rapid identification of a causative organism is crucial to determine appropriate antibiotic therapy. Fortunately, PCR is a very rapid method of detecting MRSA, and the use of MRSA PCR nasal swabs may be an effective way to predict whether MRSA is the organism causing an infection at various anatomical sites. If a patient has a suspected infection on admission, a MRSA PCR nasal swab often is completed to determine whether a patient’s nares are colonized with MRSA. However, there is no clear consensus in the literature regarding the correlation between MRSA nasal colonization and an infection caused by MRSA, making it difficult for clinicians to confidently de-escalate therapy on a negative MRSA PCR or broaden therapy on a positive result. The purpose of this literature review was to determine whether a MRSA PCR nasal swab can be used as a surrogate marker for MRSA infections at various sites.
Pneumonia has many potential causative organisms, many of which are covered empirically with guideline-directed therapy. The predictive power of MRSA PCR nasal swabs may allow clinicians to prescribe earlier directed therapy. A retrospective cohort study performed at a tertiary care center looked at the clinical usefulness of a MRSA PCR nasal swab in the treatment of pneumonia.10 Patients were included in the trial if they had a MRSA PCR nasal swab within 1 month of their blood or sputum culture as well as confirmed pneumonia. After analysis of 435 patients, the MRSA PCR nasal swab showed the following performance characteristics for detecting culture-proven MRSA: 88.0% sensitivity, 90.1% specificity, 35.4% positive predictive value (PPV), and 99.2% negative predictive value (NPV). Due to the high negative predictive value, the results indicated that discontinuation of MRSA antibiotic coverage would be appropriate for noncritically ill patients with pneumonia who had a negative MRSA PCR nasal swab.
Another retrospective study was performed by Johnson and colleagues to determine the association between MRSA PCR nasal swabs and the causative organism in pneumonia.11 Patients were included in the trial if they had a MRSA PCR nasal swab and a lower respiratory culture yielding S aureus within 48 hours of hospital admission. After analysis of 72 patients, MRSA PCR nasal swabs demonstrated the following diagnostic characteristics for detecting culture-proven MRSA: 93.3% sensitivity, 95.2% specificity, 93.3%PPV, and 95.2% NPV. These results suggest that early nasal swab MRSA PCR tests can predict the absence of MRSA reliably and may help guide the discontinuation of MRSA-directed empiric antibiotic therapy.
In addition, Giancola retrospectively studied the relationship between MRSA PCR nasal swabs and the likelihood of pneumonia caused by MRSA in intensive and intermediate care units.12 An analysis of 200 patients revealed high concordance between respiratory cultures and MRSA PCR nasal swab results with the following characteristics: 90.5% sensitivity, 79.9% specificity, 34.5% PPV, and 98.6% NPV. These test characteristics suggested that MRSA PCR nasal swabs might be a useful stewardship tool to allow for discontinuation of anti-MRSA therapy in critically ill patients with confirmed pneumonia.
Another retrospective analysis conducted by Baby and colleagues took a different approach to determine the clinical usefulness of MRSA PCR nasal swabs in the treatment of pneumonia.13 The primary outcome, mean duration of MRSA-targeted therapy, was reduced by 46.6 hours in the group who received a pharmacist-ordered MRSA PCR nasal swab compared with the group that did not receive a MRSA PCR nasal swab (P < .01) Per protocol, pharmacists were authorized to order a MRSA PCR nasal swab for patients who were prescribed vancomycin or linezolid for pneumonia. On receipt of the MRSA PCR nasal swab results, pharmacists were instructed to recommend discontinuation of anti-MRSA therapy if the PCR was negative for MRSA.
Results of this study indicated there were no significant differences in time to clinical improvement between preprotocol and postprotocol implementation (1.8 days vs 2.3 days, respectively; P = .54), length of stay (11.0 days vs 8.2 days, respectively; P = .22), or mortality (14.8% vs 6.7%, respectively; P = .41). The MRSA PCR nasal swabs allowed for a reduction in duration of anti-MRSA therapy without adverse effects on outcomes and provided a statistically significant reduction in the incidence of acute kidney injury during therapy in the postprotocol implementation group (26% vs 3.3%; P = .02), likely due to decreased exposure to vancomycin. Collectively, these studies indicate that MRSA PCR nasal swabs can be clinically useful in making decisions regarding discontinuation of MRSA-targeted therapy in pneumonia when MRSA PCR nasal swabs are negative.
A wider variety of infection sites were studied in a 2008 retrospective review of nearly 5,800 MRSA PCR nasal swabs taken within 24 hours (before or after) of a clinical culture that resulted growth of any organism.14 The goal of this study was to determine whether MRSA nasal colonization could predict MRSA involvement at various suspected infection sites. Overall, 217 patients (67.2%) with positive MRSA clinical cultures had a positive MRSA PCR nasal swab. The concordance between MRSA PCR nasal swabs and infection sites was highest with positive urine cultures (77%) and lowest in “other” infection sites (60%, primarily abdomen, buttock, and breast). Respiratory infections showed a 75% concordance between MRSA PCR nasal swabs and infection sites, as well as the following characteristics: 75% sensitivity, 90% specificity, 30% PPV, and 98% NPV. Additionally, infection site concordance was higher when clinical cultures grew clindamycin-resistant MRSA (71.3%) vs clindamycin-susceptible MRSA (59.3%; P = .04).
Overall, a positive MRSA PCR nasal swab increased the likelihood of MRSA at the primary infection site but was not clinically significant or consistent across infection sites. As seen in other studies, a negative MRSA PCR nasal swab could be useful for lowering concern for MRSA involvement in the primary infection, as evidenced by the following characteristics for all infection sites: 67% sensitivity, 90% specificity, 27% PPV, and 98% NPV.
Sarkionda and colleagues evaluated the clinical usefulness of MRSA PCR nasal swabs in the ICU setting in patients with a lower respiratory tract infection (RTI) or bloodstream infection.15 A total of 749 patients received a MRSA PCR nasal swab before admission to the ICU and were included in this study. The concordance between MRSA PCR nasal swabs and the causative organism was analyzed in patients who developed a MRSA lower respiratory infection (N = 120) and a MRSA bloodstream infection (N = 78) and demonstrated the following characteristics: 24.2% sensitivity, 78.5% specificity, 17.7% PPV, and 84.4% NPV; and 23.1% sensitivity, 78.2% specificity, 11.0% PPV, and 89.7% NPV, respectively. The authors concluded that the MRSA nasal swab results are not useful for making decisions regarding the need of empiric antimicrobial therapy targeting MRSA infections in lower respiratory infections and bloodstream infections. However, due to the high NPV in this study, one might conclude that negative MRSA PCR nasal swabs could still be used to de-escalate therapy, which is in agreement with the results from Dangerfield and Johnson.10,11
Similarly, results from a retrospective chart review demonstrated a lack of predictive value by the MRSA PCR nasal swab.16 Of 1,203 adult patients admitted to an ICU at a single center, 57 positive MRSA colonized and 122 negative MRSA colonized patients’ charts were randomly selected. The presence of MRSA lower RTI or bloodstream infections was found to be 3.51% vs 2.46% in the colonized and noncolonized groups, respectively (P = .46). These results led to the conclusion that a positive MRSA PCR nasal swab alone should not be used to make decisions regarding empiric MRSA antibiotic coverage.
An alternative approach to MRSA surveillance was taken by Harris in a prospective cohort of 12,080 adults with a suspected infection on admission to a non-ICU.17 Patients were screened with a 2-question tool to determine whether they were high risk for a MRSA infection. The 2 questions were “Have you been admitted to any health care facility in the last 12 months?” and “Do you have a skin infection (eg, boil, abscess, spider bite, or cellulitis) at this time?” If patients answered yes to either question, they were considered high risk, and a MRSA PCR nasal swab was ordered.
Patients who answered no to both questions were considered low risk and did not receive a MRSA PCR nasal swab. In total, 623 of 5,609 patients (11.1%) identified as high risk had a positive MRSA PCR nasal swab, and 148 of these 623 patients (23.8%) developed a MRSA-positive clinical culture. Only 121 of 4,986 patients (2.4%) who were high risk and had a negative MRSA PCR nasal swab went on to develop a MRSA-positive clinical culture (98% NPV). Additionally, 104 of 6,741 patients (1.6%) who answered no to both screening questions developed a MRSA-positive clinical culture (98% NPV). Results indicated that a high percentage of patients who were at high risk for MRSA (yes response to either question) and had a positive MRSA PCR nasal swab also had a positive clinical culture for MRSA. Conversely, a very small percentage of high-risk patients with a negative MRSA PCR nasal swab developed a positive clinical culture for MRSA.
The screening tool proved very effective as the low-risk group had the lowest number of patients (1.6%) develop a positive clinical culture for MRSA. It may be deduced that combination use of MRSA colonization testing via PCR nasal swabs in conjunction with a screening tool may be an effective method to identify patients in whom anti-MRSA therapy can be safely discontinued.
Conclusion
Based on the results of previously described studies, sufficient data may exist to support the discontinuation of MRSA-targeted therapy in noncritically ill patients with confirmed or suspected pneumonia and a negative MRSA PCR nasal swab. Insufficient evidence exists, however, to support a broadening of antimicrobial therapy to include anti-MRSA coverage in individuals with a positive MRSA PCR nasal swab, regardless of the infection site.
Clinical judgment should be used when determining empiric antimicrobial therapy and for appropriateness of de-escalation of therapy in critically ill patients. Once a patient stabilizes, a negative MRSA PCR nasal swab could be considered as supporting evidence to discontinue anti-MRSA therapy, especially in patients with lower respiratory infections, such as pneumonia.
1. Moran GJ, Krishnadasan A, Gorwitz RJ, et al; EMERGEncy ID Net Study Group. Methicillin-resistant S. aureus infections among patients in the emergency department. N Engl J Med. 2006;355(7):666-674.
2. Klevens RM, Morrison MA, Nadle J, et al; Active Bacterial Core surveillance (ABCs) MRSA Investigators. Invasive methicillin-resistant Staphylococcus aureus infections in the United States. JAMA. 2007;298(15):1763-1771.
3. Cosgrove SE, Fowler VG Jr. Management of methicillin-resistant Staphylococcus aureus bacteremia. Clin Infect Dis. 2008;46(suppl 5):S386-S393.
4. Dantes R, Mu Y, Belflower R, et al; Emerging Infections Program-Active Bacterial Core Surveillance MRSA Surveillance Investigators. National burden of invasive methicillin-resistant Staphylococcus aureus infections, United States, 2011. JAMA Intern Med. 2013;173(21):1970-1978.
5. Pillai MM, Latha R, Sarkar G. Detection of methicillin resistance in Staphylococcus aureus by polymerase chain reaction and conventional methods: a comparative study. J Lab Physicians. 2012;4(2):83-88.
6. Peterson LR, Liesenfeld O, Woods CW, et al. Multicenter evaluation of the LightCycler methicillin-resistant Staphylococcus aureus (MRSA) advanced test as a rapid method for detection of MRSA in nasal surveillance swabs. J Clin Microbiol. 2010;48(5):1661-1666.
7. File TM Jr, Srinivasan A, Bartlett JG. Antimicrobial stewardship: important for patient and public health. Clin Infect Dis. 2014;59(suppl 3):S93-S96.
8. Viasus D, Simonetti AF, Garcia-Vidal C, Niubó J, Dorca J, Carratalà J. Impact of antibiotic de-escalation on clinical outcomes in community-acquired pneumococcal pneumonia. J Antimicrob Chemother. 2017;72(2):547-5553.
9. Paul M, Dickstein Y, Raz-Pasteur A. Antibiotic de-escalation for bloodstream infections and pneumonia: a systematic review and meta-analysis. Clin Microbiol Infect. 2016;22(12):960-967.
10. Dangerfield B, Chung A, Webb B, Seville MT. Predictive value of methicillin-resistant Staphylococcus aureus (MRSA) nasal swab PCR assay for MRSA pneumonia. Antimicrob Agents Chemother. 2014;58(2):859-864.
11. Johnson JA, Wright ME, Sheperd LA, Musher DM, Dang BN. Nasal methicillin-resistant Staphylococcus aureus polymerase chain reaction a potential use in guiding antibiotic therapy for pneumonia. Perm J. 2015;19(1):34-36.
12. Giancola SE, Nguyen AT, Le B, et al. Clinical utility of a nasal swab methicillin-resistant Staphylococcus aureus polymerase chain reaction test in intensive and intermediate care unit patients with pneumonia. Diagn Microbiol Infect Dis. 2016;86(3):307-310.
13. Baby N, Faust AC, Smith T, Sheperd LA, Knoll L, Goodman EL. Nasal methicillin-resistant Staphylococcus aureus (MRSA) PCR testing reduces the duration of MRSA-targeted therapy in patients with suspected MRSA pneumonia. Antimicrob Agents Chemother. 2017;61(4):e02432-16.
14. Robicsek A, Suseno M, Beaumont JL, Thomson RB Jr, Peterson LR. Prediction of methicillin-resistant Staphylococcus aureus involvement in disease sites by concomitant nasal sampling. J Clin Microbiol. 2008;46(2):588-592.
15. Sarkionda KV, Micek ST, Dohery JA, Reichley RM, Warren D, Kollef MH. Methicillin-resistant Staphylococcus aureus nasal colonization is a poor predictor of intensive care unit-acquired methicillin-resistant Staphylococcus aureus infections requiring antibiotic treatment. Crit Care Med. 2010;38(10):1991-1995.
16. Ghidey F, Igbinosa O, Igbinosa E. Nasal colonization of methicillin resistant Staphylococcus aureus (MRSA) does not predict subsequent infection in the intensive care unit. Beni-Seuf University J Basic Appl Sci
17. Harris, AD, Furuno JP, Roghmann MC, et al. Targeted surveillance of methicillin-resistant Staphylococcus aureus and its potential use to guide empiric antibiotic therapy. Antimicrob Agents Chemother. 2010;54(8):3143-3148.
Methicillin-resistant Staphylococcus aureus (MRSA) is a Gram positive, round bacterium. The bacteria has evolved to withstand attacks from antibiotics and has made MRSA resistant to traditional antibiotics, such as β-lactams, resulting in difficult-to-treat infections. The presence of a genetic mutation within the mecA gene, which codes for the penicillin-binding protein 2a (PBP2a), differentiates MRSA from methicillin-susceptible Staphylococcus aureus (MSSA). Presence of the PBP2a protein allows Staphylococcus aureus (S aureus)to overcome β-lactam antibiotics’ method of killing by allowing the bacteria to continue to divide and grow.
β-lactam antibiotics cause cell death in susceptible isolates by binding to penicillin-binding proteins, which inhibits transpeptidation within the cell wall via inactivation of the penicillin-binding protein. By inhibiting cell wall synthesis, the cell loses its integrity and leaks its contents, causing cell death. Penicillin-binding protein 2a is a modified protein that has a low affinity for β-lactam antibiotics, allowing MRSA to survive and making it dangerous and difficult to eradicate.
First described in 1961, MRSA’s prevalence steadily increased in the following decades. It is the most common cause of skin and soft tissue infections presenting to emergency departments in the U.S.1 About 20% of bloodstream infections are caused by S aureus, and in 2003, nearly two-thirds of hospital-onset S aureus infections were methicillin-resistant in U.S. intensive-care units (ICUs).2 It has been shown that patients with MRSA bacteremia have worse overall outcomes, including increased mortality, greater lengths of stay, and increased costs, compared with those with MSSA infections.2,3 In 2011, MRSA infections caused an estimated 11,000 deaths, making fast and accurate detection of MRSA a crucial step in appropriate antimicrobial therapy selection.4
Currently, the Clinical and Laboratory Standards Institute (CLSI) recommends testing for MRSA by using phenotypic or genotypic methods. Phenotypic methods test for the observable characteristics of an organism, whereas a genotypic method identifies the specific gene that the organism carries. Recommended phenotypic methods include the latex agglutination test for PBP2a, the cefoxitin disk screen test, and a plate containing 6 μg/mL of oxacillin in Mueller-Hinton agar supplemented with sodium chloride.5 These methods have varying sensitivity and specificity and take between 48 to 72 hours to provide a result.
Within the past 15 years, a newer, genotypic, method of MRSA detection was approved by the FDA with high sensitivity and specificity. This method uses polymerase chain reaction (PCR) to identify the mecA gene. Polymerase chain reaction is a technique used to copy and amplify a specific segment of DNA, making thousands to millions of copies. If present, the MRSA PCR amplifies the mecA gene that makes S aureus resistant to methicillin and other β-lactams, which confirms that the specimen contains MRSA. The FDA has approved the use of MRSA PCR nasal swabs to detect MRSA in patients at risk of nasal colonization. While previously discussed methods may take between 2 and 3 days to confirm presence of MRSA, PCR can identify MRSA in about 1 hour.6
If a S aureus infection is suspected, empiric therapy often includes coverage of both MSSA and MRSA, due to the high morbidity and mortality associated with these infections. However, continuing an unneeded or unduly broad antibiotic, such as those that cover MRSA, can cause unintended consequences, such as toxicities, emerging resistance, or selection for pathogenic organisms.7 Therefore, empiric broad antibiotic therapy should be de-escalated as soon as possible, which further emphasizes the need for quick and accurate detection of the infecting organism. De-escalation of therapy can lead to a shorter length of stay and decreased mortality.8,9 Conversely, quick identification of infections caused by MRSA would allow therapy to be broadened to cover MRSA in infected patients, which could potentially decrease patient morbidity and mortality.
Nasal MRSA PCR Colonization
Rapid identification of a causative organism is crucial to determine appropriate antibiotic therapy. Fortunately, PCR is a very rapid method of detecting MRSA, and the use of MRSA PCR nasal swabs may be an effective way to predict whether MRSA is the organism causing an infection at various anatomical sites. If a patient has a suspected infection on admission, a MRSA PCR nasal swab often is completed to determine whether a patient’s nares are colonized with MRSA. However, there is no clear consensus in the literature regarding the correlation between MRSA nasal colonization and an infection caused by MRSA, making it difficult for clinicians to confidently de-escalate therapy on a negative MRSA PCR or broaden therapy on a positive result. The purpose of this literature review was to determine whether a MRSA PCR nasal swab can be used as a surrogate marker for MRSA infections at various sites.
Pneumonia has many potential causative organisms, many of which are covered empirically with guideline-directed therapy. The predictive power of MRSA PCR nasal swabs may allow clinicians to prescribe earlier directed therapy. A retrospective cohort study performed at a tertiary care center looked at the clinical usefulness of a MRSA PCR nasal swab in the treatment of pneumonia.10 Patients were included in the trial if they had a MRSA PCR nasal swab within 1 month of their blood or sputum culture as well as confirmed pneumonia. After analysis of 435 patients, the MRSA PCR nasal swab showed the following performance characteristics for detecting culture-proven MRSA: 88.0% sensitivity, 90.1% specificity, 35.4% positive predictive value (PPV), and 99.2% negative predictive value (NPV). Due to the high negative predictive value, the results indicated that discontinuation of MRSA antibiotic coverage would be appropriate for noncritically ill patients with pneumonia who had a negative MRSA PCR nasal swab.
Another retrospective study was performed by Johnson and colleagues to determine the association between MRSA PCR nasal swabs and the causative organism in pneumonia.11 Patients were included in the trial if they had a MRSA PCR nasal swab and a lower respiratory culture yielding S aureus within 48 hours of hospital admission. After analysis of 72 patients, MRSA PCR nasal swabs demonstrated the following diagnostic characteristics for detecting culture-proven MRSA: 93.3% sensitivity, 95.2% specificity, 93.3%PPV, and 95.2% NPV. These results suggest that early nasal swab MRSA PCR tests can predict the absence of MRSA reliably and may help guide the discontinuation of MRSA-directed empiric antibiotic therapy.
In addition, Giancola retrospectively studied the relationship between MRSA PCR nasal swabs and the likelihood of pneumonia caused by MRSA in intensive and intermediate care units.12 An analysis of 200 patients revealed high concordance between respiratory cultures and MRSA PCR nasal swab results with the following characteristics: 90.5% sensitivity, 79.9% specificity, 34.5% PPV, and 98.6% NPV. These test characteristics suggested that MRSA PCR nasal swabs might be a useful stewardship tool to allow for discontinuation of anti-MRSA therapy in critically ill patients with confirmed pneumonia.
Another retrospective analysis conducted by Baby and colleagues took a different approach to determine the clinical usefulness of MRSA PCR nasal swabs in the treatment of pneumonia.13 The primary outcome, mean duration of MRSA-targeted therapy, was reduced by 46.6 hours in the group who received a pharmacist-ordered MRSA PCR nasal swab compared with the group that did not receive a MRSA PCR nasal swab (P < .01) Per protocol, pharmacists were authorized to order a MRSA PCR nasal swab for patients who were prescribed vancomycin or linezolid for pneumonia. On receipt of the MRSA PCR nasal swab results, pharmacists were instructed to recommend discontinuation of anti-MRSA therapy if the PCR was negative for MRSA.
Results of this study indicated there were no significant differences in time to clinical improvement between preprotocol and postprotocol implementation (1.8 days vs 2.3 days, respectively; P = .54), length of stay (11.0 days vs 8.2 days, respectively; P = .22), or mortality (14.8% vs 6.7%, respectively; P = .41). The MRSA PCR nasal swabs allowed for a reduction in duration of anti-MRSA therapy without adverse effects on outcomes and provided a statistically significant reduction in the incidence of acute kidney injury during therapy in the postprotocol implementation group (26% vs 3.3%; P = .02), likely due to decreased exposure to vancomycin. Collectively, these studies indicate that MRSA PCR nasal swabs can be clinically useful in making decisions regarding discontinuation of MRSA-targeted therapy in pneumonia when MRSA PCR nasal swabs are negative.
A wider variety of infection sites were studied in a 2008 retrospective review of nearly 5,800 MRSA PCR nasal swabs taken within 24 hours (before or after) of a clinical culture that resulted growth of any organism.14 The goal of this study was to determine whether MRSA nasal colonization could predict MRSA involvement at various suspected infection sites. Overall, 217 patients (67.2%) with positive MRSA clinical cultures had a positive MRSA PCR nasal swab. The concordance between MRSA PCR nasal swabs and infection sites was highest with positive urine cultures (77%) and lowest in “other” infection sites (60%, primarily abdomen, buttock, and breast). Respiratory infections showed a 75% concordance between MRSA PCR nasal swabs and infection sites, as well as the following characteristics: 75% sensitivity, 90% specificity, 30% PPV, and 98% NPV. Additionally, infection site concordance was higher when clinical cultures grew clindamycin-resistant MRSA (71.3%) vs clindamycin-susceptible MRSA (59.3%; P = .04).
Overall, a positive MRSA PCR nasal swab increased the likelihood of MRSA at the primary infection site but was not clinically significant or consistent across infection sites. As seen in other studies, a negative MRSA PCR nasal swab could be useful for lowering concern for MRSA involvement in the primary infection, as evidenced by the following characteristics for all infection sites: 67% sensitivity, 90% specificity, 27% PPV, and 98% NPV.
Sarkionda and colleagues evaluated the clinical usefulness of MRSA PCR nasal swabs in the ICU setting in patients with a lower respiratory tract infection (RTI) or bloodstream infection.15 A total of 749 patients received a MRSA PCR nasal swab before admission to the ICU and were included in this study. The concordance between MRSA PCR nasal swabs and the causative organism was analyzed in patients who developed a MRSA lower respiratory infection (N = 120) and a MRSA bloodstream infection (N = 78) and demonstrated the following characteristics: 24.2% sensitivity, 78.5% specificity, 17.7% PPV, and 84.4% NPV; and 23.1% sensitivity, 78.2% specificity, 11.0% PPV, and 89.7% NPV, respectively. The authors concluded that the MRSA nasal swab results are not useful for making decisions regarding the need of empiric antimicrobial therapy targeting MRSA infections in lower respiratory infections and bloodstream infections. However, due to the high NPV in this study, one might conclude that negative MRSA PCR nasal swabs could still be used to de-escalate therapy, which is in agreement with the results from Dangerfield and Johnson.10,11
Similarly, results from a retrospective chart review demonstrated a lack of predictive value by the MRSA PCR nasal swab.16 Of 1,203 adult patients admitted to an ICU at a single center, 57 positive MRSA colonized and 122 negative MRSA colonized patients’ charts were randomly selected. The presence of MRSA lower RTI or bloodstream infections was found to be 3.51% vs 2.46% in the colonized and noncolonized groups, respectively (P = .46). These results led to the conclusion that a positive MRSA PCR nasal swab alone should not be used to make decisions regarding empiric MRSA antibiotic coverage.
An alternative approach to MRSA surveillance was taken by Harris in a prospective cohort of 12,080 adults with a suspected infection on admission to a non-ICU.17 Patients were screened with a 2-question tool to determine whether they were high risk for a MRSA infection. The 2 questions were “Have you been admitted to any health care facility in the last 12 months?” and “Do you have a skin infection (eg, boil, abscess, spider bite, or cellulitis) at this time?” If patients answered yes to either question, they were considered high risk, and a MRSA PCR nasal swab was ordered.
Patients who answered no to both questions were considered low risk and did not receive a MRSA PCR nasal swab. In total, 623 of 5,609 patients (11.1%) identified as high risk had a positive MRSA PCR nasal swab, and 148 of these 623 patients (23.8%) developed a MRSA-positive clinical culture. Only 121 of 4,986 patients (2.4%) who were high risk and had a negative MRSA PCR nasal swab went on to develop a MRSA-positive clinical culture (98% NPV). Additionally, 104 of 6,741 patients (1.6%) who answered no to both screening questions developed a MRSA-positive clinical culture (98% NPV). Results indicated that a high percentage of patients who were at high risk for MRSA (yes response to either question) and had a positive MRSA PCR nasal swab also had a positive clinical culture for MRSA. Conversely, a very small percentage of high-risk patients with a negative MRSA PCR nasal swab developed a positive clinical culture for MRSA.
The screening tool proved very effective as the low-risk group had the lowest number of patients (1.6%) develop a positive clinical culture for MRSA. It may be deduced that combination use of MRSA colonization testing via PCR nasal swabs in conjunction with a screening tool may be an effective method to identify patients in whom anti-MRSA therapy can be safely discontinued.
Conclusion
Based on the results of previously described studies, sufficient data may exist to support the discontinuation of MRSA-targeted therapy in noncritically ill patients with confirmed or suspected pneumonia and a negative MRSA PCR nasal swab. Insufficient evidence exists, however, to support a broadening of antimicrobial therapy to include anti-MRSA coverage in individuals with a positive MRSA PCR nasal swab, regardless of the infection site.
Clinical judgment should be used when determining empiric antimicrobial therapy and for appropriateness of de-escalation of therapy in critically ill patients. Once a patient stabilizes, a negative MRSA PCR nasal swab could be considered as supporting evidence to discontinue anti-MRSA therapy, especially in patients with lower respiratory infections, such as pneumonia.
Methicillin-resistant Staphylococcus aureus (MRSA) is a Gram positive, round bacterium. The bacteria has evolved to withstand attacks from antibiotics and has made MRSA resistant to traditional antibiotics, such as β-lactams, resulting in difficult-to-treat infections. The presence of a genetic mutation within the mecA gene, which codes for the penicillin-binding protein 2a (PBP2a), differentiates MRSA from methicillin-susceptible Staphylococcus aureus (MSSA). Presence of the PBP2a protein allows Staphylococcus aureus (S aureus)to overcome β-lactam antibiotics’ method of killing by allowing the bacteria to continue to divide and grow.
β-lactam antibiotics cause cell death in susceptible isolates by binding to penicillin-binding proteins, which inhibits transpeptidation within the cell wall via inactivation of the penicillin-binding protein. By inhibiting cell wall synthesis, the cell loses its integrity and leaks its contents, causing cell death. Penicillin-binding protein 2a is a modified protein that has a low affinity for β-lactam antibiotics, allowing MRSA to survive and making it dangerous and difficult to eradicate.
First described in 1961, MRSA’s prevalence steadily increased in the following decades. It is the most common cause of skin and soft tissue infections presenting to emergency departments in the U.S.1 About 20% of bloodstream infections are caused by S aureus, and in 2003, nearly two-thirds of hospital-onset S aureus infections were methicillin-resistant in U.S. intensive-care units (ICUs).2 It has been shown that patients with MRSA bacteremia have worse overall outcomes, including increased mortality, greater lengths of stay, and increased costs, compared with those with MSSA infections.2,3 In 2011, MRSA infections caused an estimated 11,000 deaths, making fast and accurate detection of MRSA a crucial step in appropriate antimicrobial therapy selection.4
Currently, the Clinical and Laboratory Standards Institute (CLSI) recommends testing for MRSA by using phenotypic or genotypic methods. Phenotypic methods test for the observable characteristics of an organism, whereas a genotypic method identifies the specific gene that the organism carries. Recommended phenotypic methods include the latex agglutination test for PBP2a, the cefoxitin disk screen test, and a plate containing 6 μg/mL of oxacillin in Mueller-Hinton agar supplemented with sodium chloride.5 These methods have varying sensitivity and specificity and take between 48 to 72 hours to provide a result.
Within the past 15 years, a newer, genotypic, method of MRSA detection was approved by the FDA with high sensitivity and specificity. This method uses polymerase chain reaction (PCR) to identify the mecA gene. Polymerase chain reaction is a technique used to copy and amplify a specific segment of DNA, making thousands to millions of copies. If present, the MRSA PCR amplifies the mecA gene that makes S aureus resistant to methicillin and other β-lactams, which confirms that the specimen contains MRSA. The FDA has approved the use of MRSA PCR nasal swabs to detect MRSA in patients at risk of nasal colonization. While previously discussed methods may take between 2 and 3 days to confirm presence of MRSA, PCR can identify MRSA in about 1 hour.6
If a S aureus infection is suspected, empiric therapy often includes coverage of both MSSA and MRSA, due to the high morbidity and mortality associated with these infections. However, continuing an unneeded or unduly broad antibiotic, such as those that cover MRSA, can cause unintended consequences, such as toxicities, emerging resistance, or selection for pathogenic organisms.7 Therefore, empiric broad antibiotic therapy should be de-escalated as soon as possible, which further emphasizes the need for quick and accurate detection of the infecting organism. De-escalation of therapy can lead to a shorter length of stay and decreased mortality.8,9 Conversely, quick identification of infections caused by MRSA would allow therapy to be broadened to cover MRSA in infected patients, which could potentially decrease patient morbidity and mortality.
Nasal MRSA PCR Colonization
Rapid identification of a causative organism is crucial to determine appropriate antibiotic therapy. Fortunately, PCR is a very rapid method of detecting MRSA, and the use of MRSA PCR nasal swabs may be an effective way to predict whether MRSA is the organism causing an infection at various anatomical sites. If a patient has a suspected infection on admission, a MRSA PCR nasal swab often is completed to determine whether a patient’s nares are colonized with MRSA. However, there is no clear consensus in the literature regarding the correlation between MRSA nasal colonization and an infection caused by MRSA, making it difficult for clinicians to confidently de-escalate therapy on a negative MRSA PCR or broaden therapy on a positive result. The purpose of this literature review was to determine whether a MRSA PCR nasal swab can be used as a surrogate marker for MRSA infections at various sites.
Pneumonia has many potential causative organisms, many of which are covered empirically with guideline-directed therapy. The predictive power of MRSA PCR nasal swabs may allow clinicians to prescribe earlier directed therapy. A retrospective cohort study performed at a tertiary care center looked at the clinical usefulness of a MRSA PCR nasal swab in the treatment of pneumonia.10 Patients were included in the trial if they had a MRSA PCR nasal swab within 1 month of their blood or sputum culture as well as confirmed pneumonia. After analysis of 435 patients, the MRSA PCR nasal swab showed the following performance characteristics for detecting culture-proven MRSA: 88.0% sensitivity, 90.1% specificity, 35.4% positive predictive value (PPV), and 99.2% negative predictive value (NPV). Due to the high negative predictive value, the results indicated that discontinuation of MRSA antibiotic coverage would be appropriate for noncritically ill patients with pneumonia who had a negative MRSA PCR nasal swab.
Another retrospective study was performed by Johnson and colleagues to determine the association between MRSA PCR nasal swabs and the causative organism in pneumonia.11 Patients were included in the trial if they had a MRSA PCR nasal swab and a lower respiratory culture yielding S aureus within 48 hours of hospital admission. After analysis of 72 patients, MRSA PCR nasal swabs demonstrated the following diagnostic characteristics for detecting culture-proven MRSA: 93.3% sensitivity, 95.2% specificity, 93.3%PPV, and 95.2% NPV. These results suggest that early nasal swab MRSA PCR tests can predict the absence of MRSA reliably and may help guide the discontinuation of MRSA-directed empiric antibiotic therapy.
In addition, Giancola retrospectively studied the relationship between MRSA PCR nasal swabs and the likelihood of pneumonia caused by MRSA in intensive and intermediate care units.12 An analysis of 200 patients revealed high concordance between respiratory cultures and MRSA PCR nasal swab results with the following characteristics: 90.5% sensitivity, 79.9% specificity, 34.5% PPV, and 98.6% NPV. These test characteristics suggested that MRSA PCR nasal swabs might be a useful stewardship tool to allow for discontinuation of anti-MRSA therapy in critically ill patients with confirmed pneumonia.
Another retrospective analysis conducted by Baby and colleagues took a different approach to determine the clinical usefulness of MRSA PCR nasal swabs in the treatment of pneumonia.13 The primary outcome, mean duration of MRSA-targeted therapy, was reduced by 46.6 hours in the group who received a pharmacist-ordered MRSA PCR nasal swab compared with the group that did not receive a MRSA PCR nasal swab (P < .01) Per protocol, pharmacists were authorized to order a MRSA PCR nasal swab for patients who were prescribed vancomycin or linezolid for pneumonia. On receipt of the MRSA PCR nasal swab results, pharmacists were instructed to recommend discontinuation of anti-MRSA therapy if the PCR was negative for MRSA.
Results of this study indicated there were no significant differences in time to clinical improvement between preprotocol and postprotocol implementation (1.8 days vs 2.3 days, respectively; P = .54), length of stay (11.0 days vs 8.2 days, respectively; P = .22), or mortality (14.8% vs 6.7%, respectively; P = .41). The MRSA PCR nasal swabs allowed for a reduction in duration of anti-MRSA therapy without adverse effects on outcomes and provided a statistically significant reduction in the incidence of acute kidney injury during therapy in the postprotocol implementation group (26% vs 3.3%; P = .02), likely due to decreased exposure to vancomycin. Collectively, these studies indicate that MRSA PCR nasal swabs can be clinically useful in making decisions regarding discontinuation of MRSA-targeted therapy in pneumonia when MRSA PCR nasal swabs are negative.
A wider variety of infection sites were studied in a 2008 retrospective review of nearly 5,800 MRSA PCR nasal swabs taken within 24 hours (before or after) of a clinical culture that resulted growth of any organism.14 The goal of this study was to determine whether MRSA nasal colonization could predict MRSA involvement at various suspected infection sites. Overall, 217 patients (67.2%) with positive MRSA clinical cultures had a positive MRSA PCR nasal swab. The concordance between MRSA PCR nasal swabs and infection sites was highest with positive urine cultures (77%) and lowest in “other” infection sites (60%, primarily abdomen, buttock, and breast). Respiratory infections showed a 75% concordance between MRSA PCR nasal swabs and infection sites, as well as the following characteristics: 75% sensitivity, 90% specificity, 30% PPV, and 98% NPV. Additionally, infection site concordance was higher when clinical cultures grew clindamycin-resistant MRSA (71.3%) vs clindamycin-susceptible MRSA (59.3%; P = .04).
Overall, a positive MRSA PCR nasal swab increased the likelihood of MRSA at the primary infection site but was not clinically significant or consistent across infection sites. As seen in other studies, a negative MRSA PCR nasal swab could be useful for lowering concern for MRSA involvement in the primary infection, as evidenced by the following characteristics for all infection sites: 67% sensitivity, 90% specificity, 27% PPV, and 98% NPV.
Sarkionda and colleagues evaluated the clinical usefulness of MRSA PCR nasal swabs in the ICU setting in patients with a lower respiratory tract infection (RTI) or bloodstream infection.15 A total of 749 patients received a MRSA PCR nasal swab before admission to the ICU and were included in this study. The concordance between MRSA PCR nasal swabs and the causative organism was analyzed in patients who developed a MRSA lower respiratory infection (N = 120) and a MRSA bloodstream infection (N = 78) and demonstrated the following characteristics: 24.2% sensitivity, 78.5% specificity, 17.7% PPV, and 84.4% NPV; and 23.1% sensitivity, 78.2% specificity, 11.0% PPV, and 89.7% NPV, respectively. The authors concluded that the MRSA nasal swab results are not useful for making decisions regarding the need of empiric antimicrobial therapy targeting MRSA infections in lower respiratory infections and bloodstream infections. However, due to the high NPV in this study, one might conclude that negative MRSA PCR nasal swabs could still be used to de-escalate therapy, which is in agreement with the results from Dangerfield and Johnson.10,11
Similarly, results from a retrospective chart review demonstrated a lack of predictive value by the MRSA PCR nasal swab.16 Of 1,203 adult patients admitted to an ICU at a single center, 57 positive MRSA colonized and 122 negative MRSA colonized patients’ charts were randomly selected. The presence of MRSA lower RTI or bloodstream infections was found to be 3.51% vs 2.46% in the colonized and noncolonized groups, respectively (P = .46). These results led to the conclusion that a positive MRSA PCR nasal swab alone should not be used to make decisions regarding empiric MRSA antibiotic coverage.
An alternative approach to MRSA surveillance was taken by Harris in a prospective cohort of 12,080 adults with a suspected infection on admission to a non-ICU.17 Patients were screened with a 2-question tool to determine whether they were high risk for a MRSA infection. The 2 questions were “Have you been admitted to any health care facility in the last 12 months?” and “Do you have a skin infection (eg, boil, abscess, spider bite, or cellulitis) at this time?” If patients answered yes to either question, they were considered high risk, and a MRSA PCR nasal swab was ordered.
Patients who answered no to both questions were considered low risk and did not receive a MRSA PCR nasal swab. In total, 623 of 5,609 patients (11.1%) identified as high risk had a positive MRSA PCR nasal swab, and 148 of these 623 patients (23.8%) developed a MRSA-positive clinical culture. Only 121 of 4,986 patients (2.4%) who were high risk and had a negative MRSA PCR nasal swab went on to develop a MRSA-positive clinical culture (98% NPV). Additionally, 104 of 6,741 patients (1.6%) who answered no to both screening questions developed a MRSA-positive clinical culture (98% NPV). Results indicated that a high percentage of patients who were at high risk for MRSA (yes response to either question) and had a positive MRSA PCR nasal swab also had a positive clinical culture for MRSA. Conversely, a very small percentage of high-risk patients with a negative MRSA PCR nasal swab developed a positive clinical culture for MRSA.
The screening tool proved very effective as the low-risk group had the lowest number of patients (1.6%) develop a positive clinical culture for MRSA. It may be deduced that combination use of MRSA colonization testing via PCR nasal swabs in conjunction with a screening tool may be an effective method to identify patients in whom anti-MRSA therapy can be safely discontinued.
Conclusion
Based on the results of previously described studies, sufficient data may exist to support the discontinuation of MRSA-targeted therapy in noncritically ill patients with confirmed or suspected pneumonia and a negative MRSA PCR nasal swab. Insufficient evidence exists, however, to support a broadening of antimicrobial therapy to include anti-MRSA coverage in individuals with a positive MRSA PCR nasal swab, regardless of the infection site.
Clinical judgment should be used when determining empiric antimicrobial therapy and for appropriateness of de-escalation of therapy in critically ill patients. Once a patient stabilizes, a negative MRSA PCR nasal swab could be considered as supporting evidence to discontinue anti-MRSA therapy, especially in patients with lower respiratory infections, such as pneumonia.
1. Moran GJ, Krishnadasan A, Gorwitz RJ, et al; EMERGEncy ID Net Study Group. Methicillin-resistant S. aureus infections among patients in the emergency department. N Engl J Med. 2006;355(7):666-674.
2. Klevens RM, Morrison MA, Nadle J, et al; Active Bacterial Core surveillance (ABCs) MRSA Investigators. Invasive methicillin-resistant Staphylococcus aureus infections in the United States. JAMA. 2007;298(15):1763-1771.
3. Cosgrove SE, Fowler VG Jr. Management of methicillin-resistant Staphylococcus aureus bacteremia. Clin Infect Dis. 2008;46(suppl 5):S386-S393.
4. Dantes R, Mu Y, Belflower R, et al; Emerging Infections Program-Active Bacterial Core Surveillance MRSA Surveillance Investigators. National burden of invasive methicillin-resistant Staphylococcus aureus infections, United States, 2011. JAMA Intern Med. 2013;173(21):1970-1978.
5. Pillai MM, Latha R, Sarkar G. Detection of methicillin resistance in Staphylococcus aureus by polymerase chain reaction and conventional methods: a comparative study. J Lab Physicians. 2012;4(2):83-88.
6. Peterson LR, Liesenfeld O, Woods CW, et al. Multicenter evaluation of the LightCycler methicillin-resistant Staphylococcus aureus (MRSA) advanced test as a rapid method for detection of MRSA in nasal surveillance swabs. J Clin Microbiol. 2010;48(5):1661-1666.
7. File TM Jr, Srinivasan A, Bartlett JG. Antimicrobial stewardship: important for patient and public health. Clin Infect Dis. 2014;59(suppl 3):S93-S96.
8. Viasus D, Simonetti AF, Garcia-Vidal C, Niubó J, Dorca J, Carratalà J. Impact of antibiotic de-escalation on clinical outcomes in community-acquired pneumococcal pneumonia. J Antimicrob Chemother. 2017;72(2):547-5553.
9. Paul M, Dickstein Y, Raz-Pasteur A. Antibiotic de-escalation for bloodstream infections and pneumonia: a systematic review and meta-analysis. Clin Microbiol Infect. 2016;22(12):960-967.
10. Dangerfield B, Chung A, Webb B, Seville MT. Predictive value of methicillin-resistant Staphylococcus aureus (MRSA) nasal swab PCR assay for MRSA pneumonia. Antimicrob Agents Chemother. 2014;58(2):859-864.
11. Johnson JA, Wright ME, Sheperd LA, Musher DM, Dang BN. Nasal methicillin-resistant Staphylococcus aureus polymerase chain reaction a potential use in guiding antibiotic therapy for pneumonia. Perm J. 2015;19(1):34-36.
12. Giancola SE, Nguyen AT, Le B, et al. Clinical utility of a nasal swab methicillin-resistant Staphylococcus aureus polymerase chain reaction test in intensive and intermediate care unit patients with pneumonia. Diagn Microbiol Infect Dis. 2016;86(3):307-310.
13. Baby N, Faust AC, Smith T, Sheperd LA, Knoll L, Goodman EL. Nasal methicillin-resistant Staphylococcus aureus (MRSA) PCR testing reduces the duration of MRSA-targeted therapy in patients with suspected MRSA pneumonia. Antimicrob Agents Chemother. 2017;61(4):e02432-16.
14. Robicsek A, Suseno M, Beaumont JL, Thomson RB Jr, Peterson LR. Prediction of methicillin-resistant Staphylococcus aureus involvement in disease sites by concomitant nasal sampling. J Clin Microbiol. 2008;46(2):588-592.
15. Sarkionda KV, Micek ST, Dohery JA, Reichley RM, Warren D, Kollef MH. Methicillin-resistant Staphylococcus aureus nasal colonization is a poor predictor of intensive care unit-acquired methicillin-resistant Staphylococcus aureus infections requiring antibiotic treatment. Crit Care Med. 2010;38(10):1991-1995.
16. Ghidey F, Igbinosa O, Igbinosa E. Nasal colonization of methicillin resistant Staphylococcus aureus (MRSA) does not predict subsequent infection in the intensive care unit. Beni-Seuf University J Basic Appl Sci
17. Harris, AD, Furuno JP, Roghmann MC, et al. Targeted surveillance of methicillin-resistant Staphylococcus aureus and its potential use to guide empiric antibiotic therapy. Antimicrob Agents Chemother. 2010;54(8):3143-3148.
1. Moran GJ, Krishnadasan A, Gorwitz RJ, et al; EMERGEncy ID Net Study Group. Methicillin-resistant S. aureus infections among patients in the emergency department. N Engl J Med. 2006;355(7):666-674.
2. Klevens RM, Morrison MA, Nadle J, et al; Active Bacterial Core surveillance (ABCs) MRSA Investigators. Invasive methicillin-resistant Staphylococcus aureus infections in the United States. JAMA. 2007;298(15):1763-1771.
3. Cosgrove SE, Fowler VG Jr. Management of methicillin-resistant Staphylococcus aureus bacteremia. Clin Infect Dis. 2008;46(suppl 5):S386-S393.
4. Dantes R, Mu Y, Belflower R, et al; Emerging Infections Program-Active Bacterial Core Surveillance MRSA Surveillance Investigators. National burden of invasive methicillin-resistant Staphylococcus aureus infections, United States, 2011. JAMA Intern Med. 2013;173(21):1970-1978.
5. Pillai MM, Latha R, Sarkar G. Detection of methicillin resistance in Staphylococcus aureus by polymerase chain reaction and conventional methods: a comparative study. J Lab Physicians. 2012;4(2):83-88.
6. Peterson LR, Liesenfeld O, Woods CW, et al. Multicenter evaluation of the LightCycler methicillin-resistant Staphylococcus aureus (MRSA) advanced test as a rapid method for detection of MRSA in nasal surveillance swabs. J Clin Microbiol. 2010;48(5):1661-1666.
7. File TM Jr, Srinivasan A, Bartlett JG. Antimicrobial stewardship: important for patient and public health. Clin Infect Dis. 2014;59(suppl 3):S93-S96.
8. Viasus D, Simonetti AF, Garcia-Vidal C, Niubó J, Dorca J, Carratalà J. Impact of antibiotic de-escalation on clinical outcomes in community-acquired pneumococcal pneumonia. J Antimicrob Chemother. 2017;72(2):547-5553.
9. Paul M, Dickstein Y, Raz-Pasteur A. Antibiotic de-escalation for bloodstream infections and pneumonia: a systematic review and meta-analysis. Clin Microbiol Infect. 2016;22(12):960-967.
10. Dangerfield B, Chung A, Webb B, Seville MT. Predictive value of methicillin-resistant Staphylococcus aureus (MRSA) nasal swab PCR assay for MRSA pneumonia. Antimicrob Agents Chemother. 2014;58(2):859-864.
11. Johnson JA, Wright ME, Sheperd LA, Musher DM, Dang BN. Nasal methicillin-resistant Staphylococcus aureus polymerase chain reaction a potential use in guiding antibiotic therapy for pneumonia. Perm J. 2015;19(1):34-36.
12. Giancola SE, Nguyen AT, Le B, et al. Clinical utility of a nasal swab methicillin-resistant Staphylococcus aureus polymerase chain reaction test in intensive and intermediate care unit patients with pneumonia. Diagn Microbiol Infect Dis. 2016;86(3):307-310.
13. Baby N, Faust AC, Smith T, Sheperd LA, Knoll L, Goodman EL. Nasal methicillin-resistant Staphylococcus aureus (MRSA) PCR testing reduces the duration of MRSA-targeted therapy in patients with suspected MRSA pneumonia. Antimicrob Agents Chemother. 2017;61(4):e02432-16.
14. Robicsek A, Suseno M, Beaumont JL, Thomson RB Jr, Peterson LR. Prediction of methicillin-resistant Staphylococcus aureus involvement in disease sites by concomitant nasal sampling. J Clin Microbiol. 2008;46(2):588-592.
15. Sarkionda KV, Micek ST, Dohery JA, Reichley RM, Warren D, Kollef MH. Methicillin-resistant Staphylococcus aureus nasal colonization is a poor predictor of intensive care unit-acquired methicillin-resistant Staphylococcus aureus infections requiring antibiotic treatment. Crit Care Med. 2010;38(10):1991-1995.
16. Ghidey F, Igbinosa O, Igbinosa E. Nasal colonization of methicillin resistant Staphylococcus aureus (MRSA) does not predict subsequent infection in the intensive care unit. Beni-Seuf University J Basic Appl Sci
17. Harris, AD, Furuno JP, Roghmann MC, et al. Targeted surveillance of methicillin-resistant Staphylococcus aureus and its potential use to guide empiric antibiotic therapy. Antimicrob Agents Chemother. 2010;54(8):3143-3148.
The Role of Fidaxomicin in Clostridium difficile Infection
The incidence of Clostridium difficile (C difficile) infection (CDI) in the U.S. has been steadily increasing. In U.S. hospitals between 1996 and 2003, the rate of CDI diagnosis doubled, and in 2011, almost half a million CDIs contributed to 29,000 deaths.1,2 Recurrence rates after successful metronidazole or vancomycin treatment are as high as 35%.3-5 After a second recurrence, rates are as high as 40% to 60%.6
Historically, CDI was almost exclusively associated with the elderly, with exposure to health care facilities, or in individuals with a history of previous antibiotic use.1,7 Risk factors for CDI recurrence are similar, including the elderly, antibiotic use during or after initial CDI treatment, and an impaired immune response against C difficile toxins.8 However, more recently CDI has been linked to individuals who were previously considered low risk, including the young and previously healthy individuals without exposure to a health care environment or recent antibiotic use.9
Community-acquired CDIs occurring in populations previously at low risk may be due to increased virulence of the disease. A hypervirulent C difficile strain, the North American Pulsed field type 1 (NAP1)/B1/027 strain, has emerged. This strain is more resistant to fluoroquinolone antibiotics and has caused multiple CDI outbreaks in the U.S.7 Along with the increased rate of CDI occurrence, mortality rates due to CDI have been rising.10 Recent studies have shown increased rates of CDI recurrence and treatment failure in response to standard therapy (metronidazole or vancomycin).11-13
To combat emerging treatment challenges of CDIs, the FDA approved a new antibiotic, fidaxomicin, for the treatment of C difficile-associated diarrhea in 2011.14 Fidaxomicin is a first-in-class macrocyclic antibiotic that has low systemic absorption, low activity against intestinal flora, and high fecal concentrations.15 Fidaxomicin also has been shown to be less likely to promote vancomycin-resistant enterococci (VRE) than does vancomycin in CDI treatment.16 Fidaxomicin is an emerging treatment strategy for CDIs, and this article reviews its role in the treatment of CDI.
CDI Standard of Therapy
Before the approval of fidaxomicin, the Infectious Diseases Society of America (IDSA) and the Society for Healthcare Epidemiology of America released the 2010 update to the clinical practice guidelines for the treatment of CDI. Due to initial concern that use of oral vancomycin would select for VRE, guidelines recommend oral metronidazole for mild-to-moderate disease, oral vancomycin for severe CDI, and combination therapy of oral vancomycin with or without IV metronidazole for severe, complicated CDI (disease severity is defined in the Table).8,17
Fidaxomicin Approval
Prior to the approval of fidaxomicin for CDI in 2011, the FDA evaluated 2 noninferiority (NI) clinical trials comparing oral fidaxomicin to oral vancomycin for the treatment of CDI. Given that a clinical trial had previously demonstrated superior clinical cure rates of vancomycin over metronidazole, vancomycin was used as the comparator in the fidaxomicin NI trials.15 Louie and colleagues conducted a double-blind, randomized phase 3 trial comparing 10 days of fidaxomicin (200 mg twice daily) to vancomycin (125 mg 4 times daily) for the treatment of CDI (n = 629).3 Fidaxomicin was found to be noninferior to vancomycin for rate of clinical cure in the modified intention-to-treat (ITT) analysis (88% vs 86%, respectively) and the per-protocol analysis (92% vs 90%, respectively) with a NI margin of 10%. There were lower recurrence rates of CDI with fidaxomicin compared with that of vancomycin in the modified ITT analysis (15% vs 25%, respectively; P = .005). When infection with the NAP1/BI/027 strain was evaluated, fidaxomicin was shown to be noninferior to vancomycin for both clinical cure and recurrence rates.
The Louie and colleagues results were further validated when a second NI trial (n = 535) was published by Cornely and colleagues, which demonstrated similar clinical cure and recurrence rates with fidaxomicin compared with that of vancomycin.4 It is important to note that both trials used a modified ITT analysis, which included postrandomization exclusions that may have biased the results. Additionally, both trials were industry sponsored and had industry representation throughout the data collection, analysis, and manuscript preparation processes.
Use of Fidaxomicin
Fidaxomicin has been considered for use in the treatment of recurrent CDI. According to the European Society of Clinical Microbiology and Infectious Diseases (ESCMID) guidelines for the treatment of CDI, fidaxomicin is an option for both first and multiple recurrences (ESCMID does not recommend fidaxomicin for an initial episode of CDI). These guidelines state that either oral fidaxomicin or vancomycin for 10 days is an appropriate first recurrence treatment option. For multiple recurrences, the recommendations are oral fidaxomicin for 10 days or oral vancomycin for 10 days followed by a tapered or pulsed regimen.18 The IDSA C difficile treatment guidelines have not been updated since the approval of fidaxomicin and, therefore, do not contain recommendations for its use.
Given the low propensity for fidaxomicin to disrupt colonic flora, it may be hypothesized that its greatest benefit would be for use as first-line therapy in patients with a high risk of CDI recurrence prior to disruption of colonic flora with the treatment of vancomycin or metronidazole. A clinical prediction tool is needed to identify patients at high risk of CDI recurrence who would most benefit from initial fidaxomicin therapy. However, clinically relevant prediction tools are not currently used.19
Evidence exists that demonstrates the role of fidaxomicin for the treatment of recurrent CDI episodes. Cornely and colleagues pooled data from the 2 NI trials that led to the approval of fidaxomicin (n = 1,164).3,4,20 Of these, 128 participants had a recent CDI episode prior to study enrollment. For the treatment of first recurrence, 20% of patients treated with fidaxomicin experienced a second recurrence within 28 days compared with 36% of vancomycin patients (P = .045). Similarly, fewer fidaxomicin patients experienced an early recurrence within 14 days of treatment than with vancomycin for early recurrence within 14 days of treatment (8% vs 27%, respectively; P = .003).
Limitations of Fidaxomicin
One limitation of fidaxomicin is the paucity of data existing for its use in severe, life-threatening CDI, and it is currently not recommended in this indication.18 The main limitation behind the use of fidaxomicin is cost. The average wholesale price of a 10-day course of therapy of fidaxomicin is $3,360 compared with $1,273 for vancomycin capsules, $32 for compounded vancomycin oral solution, and $21 for oral metronidazole.21,22 Despite the price, cost-analysis studies have found that fidaxomicin compared with oral vancomycin is cost-effective for clinical cure rates and recurrences.23,24 Fidaxomicin also was found to be cost-effective in patients with mild-to-moderate CDI and in those using concomitant antibiotics.23 Given that 2 studies demonstrated that fidaxomicin has lower recurrence rates than that of oral vancomycin, the economical use of fidaxomicin would be of most benefit to patients at highest risk for CDI recurrence.3,4
Conclusion
In light of increased CDI treatment failure, recurrence rates, and virulence of CDI, fidaxomicin is an emerging treatment strategy. Through 2 pivotal trials, fidaxomicin has been shown to be a safe and effective first-line agent for CDI.3,4 New U.S. clinical guidelines for CDI are expected to be published in spring 2016, which will likely include the recommendation for fidaxomicin use in recurrent CDI. Current evidence suggests the most cost-effective use of fidaxomicin is in patients at highest risk of CDI recurrence.
1. McDonald LC, Owings M, Jernigan DB. Clostridium difficile infection in patients discharged from US short-stay hospitals, 1996-2003. Emerg Infect Dis. 2006;12(3):409-415.
2. Lessa FC, Mu Y, Bamberg WM, et al. Burden of Clostridium difficile infection in the United States. N Engl J Med. 2015;372(9):825-834.
3. Louie TJ, Miller MA, Mullane KM, et al; OPT-80-003 Clinical Study Group. Fidaxomicin versus vancomycin for Clostridium difficile infection. N Engl J Med. 2011;364(5):422-431.
4. Cornely OA, Crook DW, Esposito R, et al. Fidaxomicin versus vancomycin for infection with Clostridium difficile in Europe, Canada, and the USA: a double-blind, non-inferiority, randomised controlled trial. Lancet Infect Dis. 2012;12(4):281-289.
5. Aslam S, Hamill RJ, Musher DM. Treatment of Clostridium difficile-associated disease: old therapies and new strategies. Lancet Infect Dis. 2005;5(9):549-557.
6. Johnson S. Recurrent Clostridium difficile infection: a review of risk factors, treatments, and outcomes. J Infect. 2009;58:403-410.
7. McDonald LC, Killgore GE, Thompson A, et al. An epidemic, toxin gene—variant strain of Clostridium difficile. N Engl J Med. 2005;353(23):2433-2441.
8. Cohen SH, Gerding DN, Johnson S, et al; Society for Healthcare Epidemiology of America; Infectious Diseases Society of America. Clinical practice guidelines for Clostridium difficile infection in adults: 2010 update by the Society for Healthcare Epidemiology of America (SHEA) and the Infectious Diseases Society of America (IDSA). Infect Control Hosp Epidemiol. 2010;31(5):431-455.
9. Centers for Disease Control and Prevention. Severe Clostridium difficile-associated disease in populations previously at low risk—4 states, 2005. MMWR Morb Mortal Wkly Rep. 2005;54(47):1201-1205.
10. Redelings MD, Sorvillo F, Mascola L. Increase in Clostridium difficile-related mortality rates, United States, 1999-2004. Emerg Infect Dis. 2007;13(9):1417-1419.
11. Maroo S, Lamont JT. Recurrent Clostridium difficile. Gastroenterology. 2006;130(4):1311-1316.
12. Pépin J, Valiquette L, Gagnon S, Routhier S, Brazeau I. Outcomes of Clostridium difficile-associated disease treated with metronidazole or vancomycin before and after the emergence of NAP1/027. Am J Gastroenterol. 2007;102(12):2781-2788.
13. Musher DM, Aslam S, Logan N, et al. Relatively poor outcome after treatment of Clostridium difficile colitis with metronidazole. Clin Infect Dis. 2005;40(11):1586-1590.
14. U.S. Food and Drug Administration. FDA approves treatment for Clostridium difficile infection; May 27, 2011 [news release]. U.S. Food and Drug Administration website. http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm257024.htm. Updated April 10, 2014. Accessed March 15, 2016.
15. Shue YK, Sears PS, Shangle S, et al. Safety, tolerance, and pharmacokinetic studies of OPT-80 in healthy volunteers following single and multiple oral doses. Antimicrob Agents Chemother. 2008;52(4):1391-1395.
16. Nerandzic MM, Mullane K, Miller MA, Babakhani F, Donskey CJ. Reduced acquisition and overgrowth of vancomycin-resistant enterococci and Candida species in patients treated with fidaxomicin versus vancomycin for Clostridium difficile infection. Clin Infect Dis. 2012;55(2):S121-S126.
17. Aslam S, Hamill RJ, Musher DM. Treatment of Clostridium difficile-associated disease: old therapies and new strategies. Lancet Infect Dis. 2005;5(9):549-557.
18. Debast SB, Bauer MP, Kuijper EJ. European Society of Clinical Microbiology and Infectious Diseases: update of the treatment guidance document for Clostridium difficile infection. Clin Microbiol Infect. 2014;20(suppl 2):1-26.
19. Kelly CP. Can we identify patients at high risk of recurrent Clostridium difficile infection? Clin Microbiol Infect. 2012;18(suppl 6):21-27.
20. Cornely OA, Miller MA, Louie TJ, Crook DW, Gorbach SL. Treatment of first recurrence of Clostridium difficile infection: fidaxomicin versus vancomycin. Clin Infect Dis. 2012;55(suppl 2):S154-S161.
21. Murray L, ed. Red Book: Pharmacy’s Fundamental Reference. 2010 ed. Montvale, NJ: Thomson Reuters (Healthcare) Inc; 2010.
22. Cruz MP. Fidaxomicin (Dificid), a novel oral macrocyclic antibacterial agent for the treatment of Clostridium difficile-associated diarrhea in adults. Pharm Ther. 2012;37(5):278-281.
23. Stranges PM, Hutton DW, Collins CD. Cost-effectiveness analysis evaluating fidaxomicin versus oral vancomycin for the treatment of Clostridium difficile infection in the United States. Value Health. 2013;16(2):297-304.
24. Sclar DA, Robison LM, Oganov AM, Schmidt JM, Bowen KA, Castillo LV. Fidaxomicin for Clostridium difficile-associated diarrhoea: epidemiological method for estimation of warranted price. Clin Drug Investig. 2012;32(8):e17-e24.
The incidence of Clostridium difficile (C difficile) infection (CDI) in the U.S. has been steadily increasing. In U.S. hospitals between 1996 and 2003, the rate of CDI diagnosis doubled, and in 2011, almost half a million CDIs contributed to 29,000 deaths.1,2 Recurrence rates after successful metronidazole or vancomycin treatment are as high as 35%.3-5 After a second recurrence, rates are as high as 40% to 60%.6
Historically, CDI was almost exclusively associated with the elderly, with exposure to health care facilities, or in individuals with a history of previous antibiotic use.1,7 Risk factors for CDI recurrence are similar, including the elderly, antibiotic use during or after initial CDI treatment, and an impaired immune response against C difficile toxins.8 However, more recently CDI has been linked to individuals who were previously considered low risk, including the young and previously healthy individuals without exposure to a health care environment or recent antibiotic use.9
Community-acquired CDIs occurring in populations previously at low risk may be due to increased virulence of the disease. A hypervirulent C difficile strain, the North American Pulsed field type 1 (NAP1)/B1/027 strain, has emerged. This strain is more resistant to fluoroquinolone antibiotics and has caused multiple CDI outbreaks in the U.S.7 Along with the increased rate of CDI occurrence, mortality rates due to CDI have been rising.10 Recent studies have shown increased rates of CDI recurrence and treatment failure in response to standard therapy (metronidazole or vancomycin).11-13
To combat emerging treatment challenges of CDIs, the FDA approved a new antibiotic, fidaxomicin, for the treatment of C difficile-associated diarrhea in 2011.14 Fidaxomicin is a first-in-class macrocyclic antibiotic that has low systemic absorption, low activity against intestinal flora, and high fecal concentrations.15 Fidaxomicin also has been shown to be less likely to promote vancomycin-resistant enterococci (VRE) than does vancomycin in CDI treatment.16 Fidaxomicin is an emerging treatment strategy for CDIs, and this article reviews its role in the treatment of CDI.
CDI Standard of Therapy
Before the approval of fidaxomicin, the Infectious Diseases Society of America (IDSA) and the Society for Healthcare Epidemiology of America released the 2010 update to the clinical practice guidelines for the treatment of CDI. Due to initial concern that use of oral vancomycin would select for VRE, guidelines recommend oral metronidazole for mild-to-moderate disease, oral vancomycin for severe CDI, and combination therapy of oral vancomycin with or without IV metronidazole for severe, complicated CDI (disease severity is defined in the Table).8,17
Fidaxomicin Approval
Prior to the approval of fidaxomicin for CDI in 2011, the FDA evaluated 2 noninferiority (NI) clinical trials comparing oral fidaxomicin to oral vancomycin for the treatment of CDI. Given that a clinical trial had previously demonstrated superior clinical cure rates of vancomycin over metronidazole, vancomycin was used as the comparator in the fidaxomicin NI trials.15 Louie and colleagues conducted a double-blind, randomized phase 3 trial comparing 10 days of fidaxomicin (200 mg twice daily) to vancomycin (125 mg 4 times daily) for the treatment of CDI (n = 629).3 Fidaxomicin was found to be noninferior to vancomycin for rate of clinical cure in the modified intention-to-treat (ITT) analysis (88% vs 86%, respectively) and the per-protocol analysis (92% vs 90%, respectively) with a NI margin of 10%. There were lower recurrence rates of CDI with fidaxomicin compared with that of vancomycin in the modified ITT analysis (15% vs 25%, respectively; P = .005). When infection with the NAP1/BI/027 strain was evaluated, fidaxomicin was shown to be noninferior to vancomycin for both clinical cure and recurrence rates.
The Louie and colleagues results were further validated when a second NI trial (n = 535) was published by Cornely and colleagues, which demonstrated similar clinical cure and recurrence rates with fidaxomicin compared with that of vancomycin.4 It is important to note that both trials used a modified ITT analysis, which included postrandomization exclusions that may have biased the results. Additionally, both trials were industry sponsored and had industry representation throughout the data collection, analysis, and manuscript preparation processes.
Use of Fidaxomicin
Fidaxomicin has been considered for use in the treatment of recurrent CDI. According to the European Society of Clinical Microbiology and Infectious Diseases (ESCMID) guidelines for the treatment of CDI, fidaxomicin is an option for both first and multiple recurrences (ESCMID does not recommend fidaxomicin for an initial episode of CDI). These guidelines state that either oral fidaxomicin or vancomycin for 10 days is an appropriate first recurrence treatment option. For multiple recurrences, the recommendations are oral fidaxomicin for 10 days or oral vancomycin for 10 days followed by a tapered or pulsed regimen.18 The IDSA C difficile treatment guidelines have not been updated since the approval of fidaxomicin and, therefore, do not contain recommendations for its use.
Given the low propensity for fidaxomicin to disrupt colonic flora, it may be hypothesized that its greatest benefit would be for use as first-line therapy in patients with a high risk of CDI recurrence prior to disruption of colonic flora with the treatment of vancomycin or metronidazole. A clinical prediction tool is needed to identify patients at high risk of CDI recurrence who would most benefit from initial fidaxomicin therapy. However, clinically relevant prediction tools are not currently used.19
Evidence exists that demonstrates the role of fidaxomicin for the treatment of recurrent CDI episodes. Cornely and colleagues pooled data from the 2 NI trials that led to the approval of fidaxomicin (n = 1,164).3,4,20 Of these, 128 participants had a recent CDI episode prior to study enrollment. For the treatment of first recurrence, 20% of patients treated with fidaxomicin experienced a second recurrence within 28 days compared with 36% of vancomycin patients (P = .045). Similarly, fewer fidaxomicin patients experienced an early recurrence within 14 days of treatment than with vancomycin for early recurrence within 14 days of treatment (8% vs 27%, respectively; P = .003).
Limitations of Fidaxomicin
One limitation of fidaxomicin is the paucity of data existing for its use in severe, life-threatening CDI, and it is currently not recommended in this indication.18 The main limitation behind the use of fidaxomicin is cost. The average wholesale price of a 10-day course of therapy of fidaxomicin is $3,360 compared with $1,273 for vancomycin capsules, $32 for compounded vancomycin oral solution, and $21 for oral metronidazole.21,22 Despite the price, cost-analysis studies have found that fidaxomicin compared with oral vancomycin is cost-effective for clinical cure rates and recurrences.23,24 Fidaxomicin also was found to be cost-effective in patients with mild-to-moderate CDI and in those using concomitant antibiotics.23 Given that 2 studies demonstrated that fidaxomicin has lower recurrence rates than that of oral vancomycin, the economical use of fidaxomicin would be of most benefit to patients at highest risk for CDI recurrence.3,4
Conclusion
In light of increased CDI treatment failure, recurrence rates, and virulence of CDI, fidaxomicin is an emerging treatment strategy. Through 2 pivotal trials, fidaxomicin has been shown to be a safe and effective first-line agent for CDI.3,4 New U.S. clinical guidelines for CDI are expected to be published in spring 2016, which will likely include the recommendation for fidaxomicin use in recurrent CDI. Current evidence suggests the most cost-effective use of fidaxomicin is in patients at highest risk of CDI recurrence.
The incidence of Clostridium difficile (C difficile) infection (CDI) in the U.S. has been steadily increasing. In U.S. hospitals between 1996 and 2003, the rate of CDI diagnosis doubled, and in 2011, almost half a million CDIs contributed to 29,000 deaths.1,2 Recurrence rates after successful metronidazole or vancomycin treatment are as high as 35%.3-5 After a second recurrence, rates are as high as 40% to 60%.6
Historically, CDI was almost exclusively associated with the elderly, with exposure to health care facilities, or in individuals with a history of previous antibiotic use.1,7 Risk factors for CDI recurrence are similar, including the elderly, antibiotic use during or after initial CDI treatment, and an impaired immune response against C difficile toxins.8 However, more recently CDI has been linked to individuals who were previously considered low risk, including the young and previously healthy individuals without exposure to a health care environment or recent antibiotic use.9
Community-acquired CDIs occurring in populations previously at low risk may be due to increased virulence of the disease. A hypervirulent C difficile strain, the North American Pulsed field type 1 (NAP1)/B1/027 strain, has emerged. This strain is more resistant to fluoroquinolone antibiotics and has caused multiple CDI outbreaks in the U.S.7 Along with the increased rate of CDI occurrence, mortality rates due to CDI have been rising.10 Recent studies have shown increased rates of CDI recurrence and treatment failure in response to standard therapy (metronidazole or vancomycin).11-13
To combat emerging treatment challenges of CDIs, the FDA approved a new antibiotic, fidaxomicin, for the treatment of C difficile-associated diarrhea in 2011.14 Fidaxomicin is a first-in-class macrocyclic antibiotic that has low systemic absorption, low activity against intestinal flora, and high fecal concentrations.15 Fidaxomicin also has been shown to be less likely to promote vancomycin-resistant enterococci (VRE) than does vancomycin in CDI treatment.16 Fidaxomicin is an emerging treatment strategy for CDIs, and this article reviews its role in the treatment of CDI.
CDI Standard of Therapy
Before the approval of fidaxomicin, the Infectious Diseases Society of America (IDSA) and the Society for Healthcare Epidemiology of America released the 2010 update to the clinical practice guidelines for the treatment of CDI. Due to initial concern that use of oral vancomycin would select for VRE, guidelines recommend oral metronidazole for mild-to-moderate disease, oral vancomycin for severe CDI, and combination therapy of oral vancomycin with or without IV metronidazole for severe, complicated CDI (disease severity is defined in the Table).8,17
Fidaxomicin Approval
Prior to the approval of fidaxomicin for CDI in 2011, the FDA evaluated 2 noninferiority (NI) clinical trials comparing oral fidaxomicin to oral vancomycin for the treatment of CDI. Given that a clinical trial had previously demonstrated superior clinical cure rates of vancomycin over metronidazole, vancomycin was used as the comparator in the fidaxomicin NI trials.15 Louie and colleagues conducted a double-blind, randomized phase 3 trial comparing 10 days of fidaxomicin (200 mg twice daily) to vancomycin (125 mg 4 times daily) for the treatment of CDI (n = 629).3 Fidaxomicin was found to be noninferior to vancomycin for rate of clinical cure in the modified intention-to-treat (ITT) analysis (88% vs 86%, respectively) and the per-protocol analysis (92% vs 90%, respectively) with a NI margin of 10%. There were lower recurrence rates of CDI with fidaxomicin compared with that of vancomycin in the modified ITT analysis (15% vs 25%, respectively; P = .005). When infection with the NAP1/BI/027 strain was evaluated, fidaxomicin was shown to be noninferior to vancomycin for both clinical cure and recurrence rates.
The Louie and colleagues results were further validated when a second NI trial (n = 535) was published by Cornely and colleagues, which demonstrated similar clinical cure and recurrence rates with fidaxomicin compared with that of vancomycin.4 It is important to note that both trials used a modified ITT analysis, which included postrandomization exclusions that may have biased the results. Additionally, both trials were industry sponsored and had industry representation throughout the data collection, analysis, and manuscript preparation processes.
Use of Fidaxomicin
Fidaxomicin has been considered for use in the treatment of recurrent CDI. According to the European Society of Clinical Microbiology and Infectious Diseases (ESCMID) guidelines for the treatment of CDI, fidaxomicin is an option for both first and multiple recurrences (ESCMID does not recommend fidaxomicin for an initial episode of CDI). These guidelines state that either oral fidaxomicin or vancomycin for 10 days is an appropriate first recurrence treatment option. For multiple recurrences, the recommendations are oral fidaxomicin for 10 days or oral vancomycin for 10 days followed by a tapered or pulsed regimen.18 The IDSA C difficile treatment guidelines have not been updated since the approval of fidaxomicin and, therefore, do not contain recommendations for its use.
Given the low propensity for fidaxomicin to disrupt colonic flora, it may be hypothesized that its greatest benefit would be for use as first-line therapy in patients with a high risk of CDI recurrence prior to disruption of colonic flora with the treatment of vancomycin or metronidazole. A clinical prediction tool is needed to identify patients at high risk of CDI recurrence who would most benefit from initial fidaxomicin therapy. However, clinically relevant prediction tools are not currently used.19
Evidence exists that demonstrates the role of fidaxomicin for the treatment of recurrent CDI episodes. Cornely and colleagues pooled data from the 2 NI trials that led to the approval of fidaxomicin (n = 1,164).3,4,20 Of these, 128 participants had a recent CDI episode prior to study enrollment. For the treatment of first recurrence, 20% of patients treated with fidaxomicin experienced a second recurrence within 28 days compared with 36% of vancomycin patients (P = .045). Similarly, fewer fidaxomicin patients experienced an early recurrence within 14 days of treatment than with vancomycin for early recurrence within 14 days of treatment (8% vs 27%, respectively; P = .003).
Limitations of Fidaxomicin
One limitation of fidaxomicin is the paucity of data existing for its use in severe, life-threatening CDI, and it is currently not recommended in this indication.18 The main limitation behind the use of fidaxomicin is cost. The average wholesale price of a 10-day course of therapy of fidaxomicin is $3,360 compared with $1,273 for vancomycin capsules, $32 for compounded vancomycin oral solution, and $21 for oral metronidazole.21,22 Despite the price, cost-analysis studies have found that fidaxomicin compared with oral vancomycin is cost-effective for clinical cure rates and recurrences.23,24 Fidaxomicin also was found to be cost-effective in patients with mild-to-moderate CDI and in those using concomitant antibiotics.23 Given that 2 studies demonstrated that fidaxomicin has lower recurrence rates than that of oral vancomycin, the economical use of fidaxomicin would be of most benefit to patients at highest risk for CDI recurrence.3,4
Conclusion
In light of increased CDI treatment failure, recurrence rates, and virulence of CDI, fidaxomicin is an emerging treatment strategy. Through 2 pivotal trials, fidaxomicin has been shown to be a safe and effective first-line agent for CDI.3,4 New U.S. clinical guidelines for CDI are expected to be published in spring 2016, which will likely include the recommendation for fidaxomicin use in recurrent CDI. Current evidence suggests the most cost-effective use of fidaxomicin is in patients at highest risk of CDI recurrence.
1. McDonald LC, Owings M, Jernigan DB. Clostridium difficile infection in patients discharged from US short-stay hospitals, 1996-2003. Emerg Infect Dis. 2006;12(3):409-415.
2. Lessa FC, Mu Y, Bamberg WM, et al. Burden of Clostridium difficile infection in the United States. N Engl J Med. 2015;372(9):825-834.
3. Louie TJ, Miller MA, Mullane KM, et al; OPT-80-003 Clinical Study Group. Fidaxomicin versus vancomycin for Clostridium difficile infection. N Engl J Med. 2011;364(5):422-431.
4. Cornely OA, Crook DW, Esposito R, et al. Fidaxomicin versus vancomycin for infection with Clostridium difficile in Europe, Canada, and the USA: a double-blind, non-inferiority, randomised controlled trial. Lancet Infect Dis. 2012;12(4):281-289.
5. Aslam S, Hamill RJ, Musher DM. Treatment of Clostridium difficile-associated disease: old therapies and new strategies. Lancet Infect Dis. 2005;5(9):549-557.
6. Johnson S. Recurrent Clostridium difficile infection: a review of risk factors, treatments, and outcomes. J Infect. 2009;58:403-410.
7. McDonald LC, Killgore GE, Thompson A, et al. An epidemic, toxin gene—variant strain of Clostridium difficile. N Engl J Med. 2005;353(23):2433-2441.
8. Cohen SH, Gerding DN, Johnson S, et al; Society for Healthcare Epidemiology of America; Infectious Diseases Society of America. Clinical practice guidelines for Clostridium difficile infection in adults: 2010 update by the Society for Healthcare Epidemiology of America (SHEA) and the Infectious Diseases Society of America (IDSA). Infect Control Hosp Epidemiol. 2010;31(5):431-455.
9. Centers for Disease Control and Prevention. Severe Clostridium difficile-associated disease in populations previously at low risk—4 states, 2005. MMWR Morb Mortal Wkly Rep. 2005;54(47):1201-1205.
10. Redelings MD, Sorvillo F, Mascola L. Increase in Clostridium difficile-related mortality rates, United States, 1999-2004. Emerg Infect Dis. 2007;13(9):1417-1419.
11. Maroo S, Lamont JT. Recurrent Clostridium difficile. Gastroenterology. 2006;130(4):1311-1316.
12. Pépin J, Valiquette L, Gagnon S, Routhier S, Brazeau I. Outcomes of Clostridium difficile-associated disease treated with metronidazole or vancomycin before and after the emergence of NAP1/027. Am J Gastroenterol. 2007;102(12):2781-2788.
13. Musher DM, Aslam S, Logan N, et al. Relatively poor outcome after treatment of Clostridium difficile colitis with metronidazole. Clin Infect Dis. 2005;40(11):1586-1590.
14. U.S. Food and Drug Administration. FDA approves treatment for Clostridium difficile infection; May 27, 2011 [news release]. U.S. Food and Drug Administration website. http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm257024.htm. Updated April 10, 2014. Accessed March 15, 2016.
15. Shue YK, Sears PS, Shangle S, et al. Safety, tolerance, and pharmacokinetic studies of OPT-80 in healthy volunteers following single and multiple oral doses. Antimicrob Agents Chemother. 2008;52(4):1391-1395.
16. Nerandzic MM, Mullane K, Miller MA, Babakhani F, Donskey CJ. Reduced acquisition and overgrowth of vancomycin-resistant enterococci and Candida species in patients treated with fidaxomicin versus vancomycin for Clostridium difficile infection. Clin Infect Dis. 2012;55(2):S121-S126.
17. Aslam S, Hamill RJ, Musher DM. Treatment of Clostridium difficile-associated disease: old therapies and new strategies. Lancet Infect Dis. 2005;5(9):549-557.
18. Debast SB, Bauer MP, Kuijper EJ. European Society of Clinical Microbiology and Infectious Diseases: update of the treatment guidance document for Clostridium difficile infection. Clin Microbiol Infect. 2014;20(suppl 2):1-26.
19. Kelly CP. Can we identify patients at high risk of recurrent Clostridium difficile infection? Clin Microbiol Infect. 2012;18(suppl 6):21-27.
20. Cornely OA, Miller MA, Louie TJ, Crook DW, Gorbach SL. Treatment of first recurrence of Clostridium difficile infection: fidaxomicin versus vancomycin. Clin Infect Dis. 2012;55(suppl 2):S154-S161.
21. Murray L, ed. Red Book: Pharmacy’s Fundamental Reference. 2010 ed. Montvale, NJ: Thomson Reuters (Healthcare) Inc; 2010.
22. Cruz MP. Fidaxomicin (Dificid), a novel oral macrocyclic antibacterial agent for the treatment of Clostridium difficile-associated diarrhea in adults. Pharm Ther. 2012;37(5):278-281.
23. Stranges PM, Hutton DW, Collins CD. Cost-effectiveness analysis evaluating fidaxomicin versus oral vancomycin for the treatment of Clostridium difficile infection in the United States. Value Health. 2013;16(2):297-304.
24. Sclar DA, Robison LM, Oganov AM, Schmidt JM, Bowen KA, Castillo LV. Fidaxomicin for Clostridium difficile-associated diarrhoea: epidemiological method for estimation of warranted price. Clin Drug Investig. 2012;32(8):e17-e24.
1. McDonald LC, Owings M, Jernigan DB. Clostridium difficile infection in patients discharged from US short-stay hospitals, 1996-2003. Emerg Infect Dis. 2006;12(3):409-415.
2. Lessa FC, Mu Y, Bamberg WM, et al. Burden of Clostridium difficile infection in the United States. N Engl J Med. 2015;372(9):825-834.
3. Louie TJ, Miller MA, Mullane KM, et al; OPT-80-003 Clinical Study Group. Fidaxomicin versus vancomycin for Clostridium difficile infection. N Engl J Med. 2011;364(5):422-431.
4. Cornely OA, Crook DW, Esposito R, et al. Fidaxomicin versus vancomycin for infection with Clostridium difficile in Europe, Canada, and the USA: a double-blind, non-inferiority, randomised controlled trial. Lancet Infect Dis. 2012;12(4):281-289.
5. Aslam S, Hamill RJ, Musher DM. Treatment of Clostridium difficile-associated disease: old therapies and new strategies. Lancet Infect Dis. 2005;5(9):549-557.
6. Johnson S. Recurrent Clostridium difficile infection: a review of risk factors, treatments, and outcomes. J Infect. 2009;58:403-410.
7. McDonald LC, Killgore GE, Thompson A, et al. An epidemic, toxin gene—variant strain of Clostridium difficile. N Engl J Med. 2005;353(23):2433-2441.
8. Cohen SH, Gerding DN, Johnson S, et al; Society for Healthcare Epidemiology of America; Infectious Diseases Society of America. Clinical practice guidelines for Clostridium difficile infection in adults: 2010 update by the Society for Healthcare Epidemiology of America (SHEA) and the Infectious Diseases Society of America (IDSA). Infect Control Hosp Epidemiol. 2010;31(5):431-455.
9. Centers for Disease Control and Prevention. Severe Clostridium difficile-associated disease in populations previously at low risk—4 states, 2005. MMWR Morb Mortal Wkly Rep. 2005;54(47):1201-1205.
10. Redelings MD, Sorvillo F, Mascola L. Increase in Clostridium difficile-related mortality rates, United States, 1999-2004. Emerg Infect Dis. 2007;13(9):1417-1419.
11. Maroo S, Lamont JT. Recurrent Clostridium difficile. Gastroenterology. 2006;130(4):1311-1316.
12. Pépin J, Valiquette L, Gagnon S, Routhier S, Brazeau I. Outcomes of Clostridium difficile-associated disease treated with metronidazole or vancomycin before and after the emergence of NAP1/027. Am J Gastroenterol. 2007;102(12):2781-2788.
13. Musher DM, Aslam S, Logan N, et al. Relatively poor outcome after treatment of Clostridium difficile colitis with metronidazole. Clin Infect Dis. 2005;40(11):1586-1590.
14. U.S. Food and Drug Administration. FDA approves treatment for Clostridium difficile infection; May 27, 2011 [news release]. U.S. Food and Drug Administration website. http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm257024.htm. Updated April 10, 2014. Accessed March 15, 2016.
15. Shue YK, Sears PS, Shangle S, et al. Safety, tolerance, and pharmacokinetic studies of OPT-80 in healthy volunteers following single and multiple oral doses. Antimicrob Agents Chemother. 2008;52(4):1391-1395.
16. Nerandzic MM, Mullane K, Miller MA, Babakhani F, Donskey CJ. Reduced acquisition and overgrowth of vancomycin-resistant enterococci and Candida species in patients treated with fidaxomicin versus vancomycin for Clostridium difficile infection. Clin Infect Dis. 2012;55(2):S121-S126.
17. Aslam S, Hamill RJ, Musher DM. Treatment of Clostridium difficile-associated disease: old therapies and new strategies. Lancet Infect Dis. 2005;5(9):549-557.
18. Debast SB, Bauer MP, Kuijper EJ. European Society of Clinical Microbiology and Infectious Diseases: update of the treatment guidance document for Clostridium difficile infection. Clin Microbiol Infect. 2014;20(suppl 2):1-26.
19. Kelly CP. Can we identify patients at high risk of recurrent Clostridium difficile infection? Clin Microbiol Infect. 2012;18(suppl 6):21-27.
20. Cornely OA, Miller MA, Louie TJ, Crook DW, Gorbach SL. Treatment of first recurrence of Clostridium difficile infection: fidaxomicin versus vancomycin. Clin Infect Dis. 2012;55(suppl 2):S154-S161.
21. Murray L, ed. Red Book: Pharmacy’s Fundamental Reference. 2010 ed. Montvale, NJ: Thomson Reuters (Healthcare) Inc; 2010.
22. Cruz MP. Fidaxomicin (Dificid), a novel oral macrocyclic antibacterial agent for the treatment of Clostridium difficile-associated diarrhea in adults. Pharm Ther. 2012;37(5):278-281.
23. Stranges PM, Hutton DW, Collins CD. Cost-effectiveness analysis evaluating fidaxomicin versus oral vancomycin for the treatment of Clostridium difficile infection in the United States. Value Health. 2013;16(2):297-304.
24. Sclar DA, Robison LM, Oganov AM, Schmidt JM, Bowen KA, Castillo LV. Fidaxomicin for Clostridium difficile-associated diarrhoea: epidemiological method for estimation of warranted price. Clin Drug Investig. 2012;32(8):e17-e24.
Antimicrobial Dosing for Empiric and Documented Pseudomonas
Pseudomonas is a genus of aerobic, Gram-negative bacilli consisting of about 200 species. Pseudomonas aeruginosa (P aeruginosa) is the species most commonly associated with serious hospital-acquired infections and is commonly found in moist environments in hospitals, such as sinks, showers, and machinery/equipment. The symptoms of an infection by this bacterium are variable based on the site of infection and can manifest in various sites, such as the respiratory tract, urinary tract, ears, eyes, heart, skin, and soft tissue.1 General risk factors for infection with P aeruginosa include immunosuppression, history of lung disease, hospitalization lasting at least 5 days, history of repeated antibiotic use within 90 days, and a history of pseudomonal colonization/infection.
Related: Antibiotic Therapy and Bacterial Resistance in Patients With Spinal Cord Injury
Pseudomonas aeruginosa is a challenging organism to manage, as it is inherently resistant to many antibiotics. Furthermore, antibiotics effective against infections caused by P aeruginosa often require specific regimens as a result of the high minimum inhibitory concentration (MIC) of the organism. Two specific strategies that have been analyzed for proper coverage of P aeruginosa include the use of higher than usual doses and extended infusions. Due to significant challenges associated with obtaining patient outcomes data in human clinical trials, researchers often use Monte Carlo simulations, which are computational algorithms that simulate the variables of a study (ie, patient demographics) to be as real as possible to accurately predict therapeutic responses in patients.
Analyzing pharmacokinetic (PK) and pharmacodynamic (PD) indexes is valuable for determining therapeutic efficacy, as these indexes consider both the antibiotic dose/concentration and its effect over time in relation to response to therapy. The free-drug area under the concentration time curve (fAUC/MIC) ratio is a PK/PD value commonly used to describe the free-drug concentration over 24 hours that is above the MIC.2 The fAUC is dependent on creatinine clearance (CrCl) and, therefore, is specific to each patient. A threshold value for the fAUC/MIC is determined for an antibiotic, and a therapeutic regimen is dosed accordingly to assure fAUC/MIC attainment above the minimum threshold. The probability of target attainment (PTA), which is the probability that the threshold value of a PD index is achieved at a certain MIC, and the probability of cure (POC) for a given antibiotic regimen are used to determine the efficacy of an antibiotic in Monte Carlo simulations.2
Related: Bacteremia From an Unlikely Source
A study by Zelenitsky and colleagues evaluated the efficacy of 3 ciprofloxacin dosing regimens using Monte Carlo simulations (400 mg IV every 12 hours [standard dose], 400 mg IV every 8 hours [high dose], and a PD-targeted regimen dosed to attain an fAUC/MIC value > 86).3 An fAUC/MIC value of 86 was previously determined to predict cure rates of at least 90%.4 The Clinical and Laboratory Standards Institute defines a P aeruginosa MIC of ≤ 1 μg/mL to be susceptible and an MIC of ≥ 4 μg/mL to be resistant to ciprofloxacin.5
The researchers determined PTA and POC values for each regimen based on various MICs. The in vitro laboratory simulations revealed the PTA and POC values approached 100% for all 3 regimens when the MIC was 0.125 μg/mL. However, when the MIC was 1 μg/mL, the PTA for the standard and high dose was 0%, and the PD-targeted regimen was 40%. The POC was 27%, 40%, and 72% for the standard dose, high dose, and the PD-targeted regimen, respectively. Although the PD-targeted regimen was the most efficacious, it took doses exceeding 1,300 mg and 1,800 mg daily to achieve similar results. In addition, PD-targeted regimens are not practical for dosing due to patient variability in CrCl. From these simulations, it was concluded that the high dose of ciprofloxacin 400 mg IV every 8 hours should be recommended for treating Pseudomonas infections in patients with normal renal function.
Related: Antimicrobial Stewardship in an Outpatient Parenteral Antibiotic Therapy Program
In another study by Lodise and colleagues, researchers examined the clinical implications of an extended-infusion dosing strategy for piperacillin-tazobactam in the critically ill.6 The 2 piperacillin- tazobactam regimens evaluated were 3.375 g IV over 30 minutes given every 4 or 6 hours and 3.375 g IV over 4 hours given every 8 hours. The 14-day mortality rate in critically ill patients who received the extended- and intermittent-infusion regimens was 12.2% and 31.6%, respectively (P = .04). Additionally, patients receiving the extended-infusion regimen had a decreased in-house length of stay compared with the intermittent-infusion group (21 vs 38 days, P = .02). Despite having a lower drug concentration peak, the extended-infusion regimen maintains steady drug concentrations above the MIC for a greater period, resulting in prolonged therapeutic efficacy. Other antibiotics (cefepime7 and ceftazidime8) have been studied by using the same methodology of comparing intermittent and extended infusions and have had similar results.
Given the management challenges associated with P aeruginosa infections, it is important for clinicians to recognize patients who may have or be at risk of infection with P aeruginosa and use appropriate dosing regimens to effectively manage infections and improve patient outcomes.
Additional Note
An earlier version of this article appeared in the Pharmacy Related Newsletter: The Capsule, of the William S. Middleton Memorial Veterans Hospital.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
1. Murray PR, Pfaller MA, Rosenthal KS. Medical Microbiology. 7th ed. Philadelphia, PA: Elsevier; 2012.
2. Mouton JW, Dudley MN, Cars O, Derendorf H, Drusano GL. Standardization of pharmacokinetic/pharmacodynamic (PK/PD) terminology for anti-infective drugs: an update. J Antimicrob Chemother. 2005;55(5):601-607.
3. Zelenitsky S, Ariano R, Harding G, Forrest A. Evaluating ciprofloxacin dosing for Pseudomonas aeruginosa infection by using clinical outcome-based Monte Carlo simulations. Antimicrob Agents Chemother. 2005;49(10):4009-4014.
4. Zelenitsky SA, Harding GK, Sun S, Ubhi K, Ariano RE. Treatment and outcome of Pseudomonas aeruginosa bacteraemia: an antibiotic pharmacodynamic analysis. J Antimicrob Chemother. 2003;52(4):668-674.
5. Clinical and Laboratory Standards Institute. Performance Standards for Antimicrobial Susceptibility Testing; Twenty-Third Informational Supplement. CLSI document M100-S23. Wayne, PA: Clinical and Laboratory Standards Institute; 2013:63.
6. Lodise TP Jr, Lomaestro B, Drusano GL. Piperacillin-tazobactam for Pseudomonas aeruginosa infection: clinical implications of an extended-infusion dosing strategy. Clin Infect Dis. 2007;44(3):357-363.
7. Mouton JW, Den Hollander JG. Killing of Pseudomonas aeruginosa during continuous and intermittent infusion of ceftazidime in an in vitro pharmacokinetic model. Antimicrob Agents Chemother. 1994;38(5):931-936
8. Bauer KA, West JE, O’Brien JM, Goff DA. Extended-infusion cefepime reduces mortality in patients with Pseudomonas aeruginosa infections. Antimicrob Agents Chemother. 2013;57(7):2907-2912.
Pseudomonas is a genus of aerobic, Gram-negative bacilli consisting of about 200 species. Pseudomonas aeruginosa (P aeruginosa) is the species most commonly associated with serious hospital-acquired infections and is commonly found in moist environments in hospitals, such as sinks, showers, and machinery/equipment. The symptoms of an infection by this bacterium are variable based on the site of infection and can manifest in various sites, such as the respiratory tract, urinary tract, ears, eyes, heart, skin, and soft tissue.1 General risk factors for infection with P aeruginosa include immunosuppression, history of lung disease, hospitalization lasting at least 5 days, history of repeated antibiotic use within 90 days, and a history of pseudomonal colonization/infection.
Related: Antibiotic Therapy and Bacterial Resistance in Patients With Spinal Cord Injury
Pseudomonas aeruginosa is a challenging organism to manage, as it is inherently resistant to many antibiotics. Furthermore, antibiotics effective against infections caused by P aeruginosa often require specific regimens as a result of the high minimum inhibitory concentration (MIC) of the organism. Two specific strategies that have been analyzed for proper coverage of P aeruginosa include the use of higher than usual doses and extended infusions. Due to significant challenges associated with obtaining patient outcomes data in human clinical trials, researchers often use Monte Carlo simulations, which are computational algorithms that simulate the variables of a study (ie, patient demographics) to be as real as possible to accurately predict therapeutic responses in patients.
Analyzing pharmacokinetic (PK) and pharmacodynamic (PD) indexes is valuable for determining therapeutic efficacy, as these indexes consider both the antibiotic dose/concentration and its effect over time in relation to response to therapy. The free-drug area under the concentration time curve (fAUC/MIC) ratio is a PK/PD value commonly used to describe the free-drug concentration over 24 hours that is above the MIC.2 The fAUC is dependent on creatinine clearance (CrCl) and, therefore, is specific to each patient. A threshold value for the fAUC/MIC is determined for an antibiotic, and a therapeutic regimen is dosed accordingly to assure fAUC/MIC attainment above the minimum threshold. The probability of target attainment (PTA), which is the probability that the threshold value of a PD index is achieved at a certain MIC, and the probability of cure (POC) for a given antibiotic regimen are used to determine the efficacy of an antibiotic in Monte Carlo simulations.2
Related: Bacteremia From an Unlikely Source
A study by Zelenitsky and colleagues evaluated the efficacy of 3 ciprofloxacin dosing regimens using Monte Carlo simulations (400 mg IV every 12 hours [standard dose], 400 mg IV every 8 hours [high dose], and a PD-targeted regimen dosed to attain an fAUC/MIC value > 86).3 An fAUC/MIC value of 86 was previously determined to predict cure rates of at least 90%.4 The Clinical and Laboratory Standards Institute defines a P aeruginosa MIC of ≤ 1 μg/mL to be susceptible and an MIC of ≥ 4 μg/mL to be resistant to ciprofloxacin.5
The researchers determined PTA and POC values for each regimen based on various MICs. The in vitro laboratory simulations revealed the PTA and POC values approached 100% for all 3 regimens when the MIC was 0.125 μg/mL. However, when the MIC was 1 μg/mL, the PTA for the standard and high dose was 0%, and the PD-targeted regimen was 40%. The POC was 27%, 40%, and 72% for the standard dose, high dose, and the PD-targeted regimen, respectively. Although the PD-targeted regimen was the most efficacious, it took doses exceeding 1,300 mg and 1,800 mg daily to achieve similar results. In addition, PD-targeted regimens are not practical for dosing due to patient variability in CrCl. From these simulations, it was concluded that the high dose of ciprofloxacin 400 mg IV every 8 hours should be recommended for treating Pseudomonas infections in patients with normal renal function.
Related: Antimicrobial Stewardship in an Outpatient Parenteral Antibiotic Therapy Program
In another study by Lodise and colleagues, researchers examined the clinical implications of an extended-infusion dosing strategy for piperacillin-tazobactam in the critically ill.6 The 2 piperacillin- tazobactam regimens evaluated were 3.375 g IV over 30 minutes given every 4 or 6 hours and 3.375 g IV over 4 hours given every 8 hours. The 14-day mortality rate in critically ill patients who received the extended- and intermittent-infusion regimens was 12.2% and 31.6%, respectively (P = .04). Additionally, patients receiving the extended-infusion regimen had a decreased in-house length of stay compared with the intermittent-infusion group (21 vs 38 days, P = .02). Despite having a lower drug concentration peak, the extended-infusion regimen maintains steady drug concentrations above the MIC for a greater period, resulting in prolonged therapeutic efficacy. Other antibiotics (cefepime7 and ceftazidime8) have been studied by using the same methodology of comparing intermittent and extended infusions and have had similar results.
Given the management challenges associated with P aeruginosa infections, it is important for clinicians to recognize patients who may have or be at risk of infection with P aeruginosa and use appropriate dosing regimens to effectively manage infections and improve patient outcomes.
Additional Note
An earlier version of this article appeared in the Pharmacy Related Newsletter: The Capsule, of the William S. Middleton Memorial Veterans Hospital.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
Pseudomonas is a genus of aerobic, Gram-negative bacilli consisting of about 200 species. Pseudomonas aeruginosa (P aeruginosa) is the species most commonly associated with serious hospital-acquired infections and is commonly found in moist environments in hospitals, such as sinks, showers, and machinery/equipment. The symptoms of an infection by this bacterium are variable based on the site of infection and can manifest in various sites, such as the respiratory tract, urinary tract, ears, eyes, heart, skin, and soft tissue.1 General risk factors for infection with P aeruginosa include immunosuppression, history of lung disease, hospitalization lasting at least 5 days, history of repeated antibiotic use within 90 days, and a history of pseudomonal colonization/infection.
Related: Antibiotic Therapy and Bacterial Resistance in Patients With Spinal Cord Injury
Pseudomonas aeruginosa is a challenging organism to manage, as it is inherently resistant to many antibiotics. Furthermore, antibiotics effective against infections caused by P aeruginosa often require specific regimens as a result of the high minimum inhibitory concentration (MIC) of the organism. Two specific strategies that have been analyzed for proper coverage of P aeruginosa include the use of higher than usual doses and extended infusions. Due to significant challenges associated with obtaining patient outcomes data in human clinical trials, researchers often use Monte Carlo simulations, which are computational algorithms that simulate the variables of a study (ie, patient demographics) to be as real as possible to accurately predict therapeutic responses in patients.
Analyzing pharmacokinetic (PK) and pharmacodynamic (PD) indexes is valuable for determining therapeutic efficacy, as these indexes consider both the antibiotic dose/concentration and its effect over time in relation to response to therapy. The free-drug area under the concentration time curve (fAUC/MIC) ratio is a PK/PD value commonly used to describe the free-drug concentration over 24 hours that is above the MIC.2 The fAUC is dependent on creatinine clearance (CrCl) and, therefore, is specific to each patient. A threshold value for the fAUC/MIC is determined for an antibiotic, and a therapeutic regimen is dosed accordingly to assure fAUC/MIC attainment above the minimum threshold. The probability of target attainment (PTA), which is the probability that the threshold value of a PD index is achieved at a certain MIC, and the probability of cure (POC) for a given antibiotic regimen are used to determine the efficacy of an antibiotic in Monte Carlo simulations.2
Related: Bacteremia From an Unlikely Source
A study by Zelenitsky and colleagues evaluated the efficacy of 3 ciprofloxacin dosing regimens using Monte Carlo simulations (400 mg IV every 12 hours [standard dose], 400 mg IV every 8 hours [high dose], and a PD-targeted regimen dosed to attain an fAUC/MIC value > 86).3 An fAUC/MIC value of 86 was previously determined to predict cure rates of at least 90%.4 The Clinical and Laboratory Standards Institute defines a P aeruginosa MIC of ≤ 1 μg/mL to be susceptible and an MIC of ≥ 4 μg/mL to be resistant to ciprofloxacin.5
The researchers determined PTA and POC values for each regimen based on various MICs. The in vitro laboratory simulations revealed the PTA and POC values approached 100% for all 3 regimens when the MIC was 0.125 μg/mL. However, when the MIC was 1 μg/mL, the PTA for the standard and high dose was 0%, and the PD-targeted regimen was 40%. The POC was 27%, 40%, and 72% for the standard dose, high dose, and the PD-targeted regimen, respectively. Although the PD-targeted regimen was the most efficacious, it took doses exceeding 1,300 mg and 1,800 mg daily to achieve similar results. In addition, PD-targeted regimens are not practical for dosing due to patient variability in CrCl. From these simulations, it was concluded that the high dose of ciprofloxacin 400 mg IV every 8 hours should be recommended for treating Pseudomonas infections in patients with normal renal function.
Related: Antimicrobial Stewardship in an Outpatient Parenteral Antibiotic Therapy Program
In another study by Lodise and colleagues, researchers examined the clinical implications of an extended-infusion dosing strategy for piperacillin-tazobactam in the critically ill.6 The 2 piperacillin- tazobactam regimens evaluated were 3.375 g IV over 30 minutes given every 4 or 6 hours and 3.375 g IV over 4 hours given every 8 hours. The 14-day mortality rate in critically ill patients who received the extended- and intermittent-infusion regimens was 12.2% and 31.6%, respectively (P = .04). Additionally, patients receiving the extended-infusion regimen had a decreased in-house length of stay compared with the intermittent-infusion group (21 vs 38 days, P = .02). Despite having a lower drug concentration peak, the extended-infusion regimen maintains steady drug concentrations above the MIC for a greater period, resulting in prolonged therapeutic efficacy. Other antibiotics (cefepime7 and ceftazidime8) have been studied by using the same methodology of comparing intermittent and extended infusions and have had similar results.
Given the management challenges associated with P aeruginosa infections, it is important for clinicians to recognize patients who may have or be at risk of infection with P aeruginosa and use appropriate dosing regimens to effectively manage infections and improve patient outcomes.
Additional Note
An earlier version of this article appeared in the Pharmacy Related Newsletter: The Capsule, of the William S. Middleton Memorial Veterans Hospital.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
1. Murray PR, Pfaller MA, Rosenthal KS. Medical Microbiology. 7th ed. Philadelphia, PA: Elsevier; 2012.
2. Mouton JW, Dudley MN, Cars O, Derendorf H, Drusano GL. Standardization of pharmacokinetic/pharmacodynamic (PK/PD) terminology for anti-infective drugs: an update. J Antimicrob Chemother. 2005;55(5):601-607.
3. Zelenitsky S, Ariano R, Harding G, Forrest A. Evaluating ciprofloxacin dosing for Pseudomonas aeruginosa infection by using clinical outcome-based Monte Carlo simulations. Antimicrob Agents Chemother. 2005;49(10):4009-4014.
4. Zelenitsky SA, Harding GK, Sun S, Ubhi K, Ariano RE. Treatment and outcome of Pseudomonas aeruginosa bacteraemia: an antibiotic pharmacodynamic analysis. J Antimicrob Chemother. 2003;52(4):668-674.
5. Clinical and Laboratory Standards Institute. Performance Standards for Antimicrobial Susceptibility Testing; Twenty-Third Informational Supplement. CLSI document M100-S23. Wayne, PA: Clinical and Laboratory Standards Institute; 2013:63.
6. Lodise TP Jr, Lomaestro B, Drusano GL. Piperacillin-tazobactam for Pseudomonas aeruginosa infection: clinical implications of an extended-infusion dosing strategy. Clin Infect Dis. 2007;44(3):357-363.
7. Mouton JW, Den Hollander JG. Killing of Pseudomonas aeruginosa during continuous and intermittent infusion of ceftazidime in an in vitro pharmacokinetic model. Antimicrob Agents Chemother. 1994;38(5):931-936
8. Bauer KA, West JE, O’Brien JM, Goff DA. Extended-infusion cefepime reduces mortality in patients with Pseudomonas aeruginosa infections. Antimicrob Agents Chemother. 2013;57(7):2907-2912.
1. Murray PR, Pfaller MA, Rosenthal KS. Medical Microbiology. 7th ed. Philadelphia, PA: Elsevier; 2012.
2. Mouton JW, Dudley MN, Cars O, Derendorf H, Drusano GL. Standardization of pharmacokinetic/pharmacodynamic (PK/PD) terminology for anti-infective drugs: an update. J Antimicrob Chemother. 2005;55(5):601-607.
3. Zelenitsky S, Ariano R, Harding G, Forrest A. Evaluating ciprofloxacin dosing for Pseudomonas aeruginosa infection by using clinical outcome-based Monte Carlo simulations. Antimicrob Agents Chemother. 2005;49(10):4009-4014.
4. Zelenitsky SA, Harding GK, Sun S, Ubhi K, Ariano RE. Treatment and outcome of Pseudomonas aeruginosa bacteraemia: an antibiotic pharmacodynamic analysis. J Antimicrob Chemother. 2003;52(4):668-674.
5. Clinical and Laboratory Standards Institute. Performance Standards for Antimicrobial Susceptibility Testing; Twenty-Third Informational Supplement. CLSI document M100-S23. Wayne, PA: Clinical and Laboratory Standards Institute; 2013:63.
6. Lodise TP Jr, Lomaestro B, Drusano GL. Piperacillin-tazobactam for Pseudomonas aeruginosa infection: clinical implications of an extended-infusion dosing strategy. Clin Infect Dis. 2007;44(3):357-363.
7. Mouton JW, Den Hollander JG. Killing of Pseudomonas aeruginosa during continuous and intermittent infusion of ceftazidime in an in vitro pharmacokinetic model. Antimicrob Agents Chemother. 1994;38(5):931-936
8. Bauer KA, West JE, O’Brien JM, Goff DA. Extended-infusion cefepime reduces mortality in patients with Pseudomonas aeruginosa infections. Antimicrob Agents Chemother. 2013;57(7):2907-2912.