User login
Sharon Worcester is an award-winning medical journalist for MDedge News. She has been with the company since 1996, first as the Southeast Bureau Chief (1996-2009) when the company was known as International Medical News Group, then as a freelance writer (2010-2015) before returning as a reporter in 2015. She previously worked as a daily newspaper reporter covering health and local government. Sharon currently reports primarily on oncology and hematology. She has a BA from Eckerd College and an MA in Mass Communication/Print Journalism from the University of Florida. Connect with her via LinkedIn and follow her on twitter @SW_MedReporter.
Trastuzumab Bests Lapatinib for HER2 Breast Cancer
Trastuzumab is significantly more effective alone or in combination with lapatinib than is lapatinib alone for the treatment of human epidermal growth factor receptor 2–positive breast cancer in patients receiving neoadjuvant chemotherapy, according to findings from two randomized phase III trials.
The studies – GeparQuinto and the NeoALTTO (Neoadjuvant Lapatinib and/or Trastuzumab Treatment Optimization) trial – underscore the value of testing new therapies in the neoadjuvant setting.
In the GeparQuinto trial, 30% of 307 HER2-positive patients treated with the anti-HER2 humanized monoclonal antibody trastuzumab had a pathological complete response, compared with only 23% of 308 HER2-positive patients treated with the tyrosine kinase inhibitor lapatinib, which targets both HER1 and HER2 (odds ratio, 0.68).
Although the patients randomized to receive trastuzumab had significantly more edema (39% vs. 29%) and dyspnea (30% vs. 21%), those randomized to receive lapatinib experienced significantly more diarrhea (75% vs. 47%) and skin rash (55% vs. 32%), and significantly more patients in the lapatinib group discontinued treatment (33% vs. 14%), Dr. Michael Untch and his colleagues reported in the Jan. 17 issue of the Lancet Oncology.
Women with previously untreated unilateral or bilateral primary invasive breast carcinoma were enrolled in the study between Nov. 7, 2007, and July 9, 2010, at 126 centers in Germany and 1 center in Switzerland. Patients were eligible for inclusion if they had locally advanced tumor stages cT3 or cT4, hormone receptor (HR)-negative tumors, or HR-positive tumors with clinically positive axillary nodes (cN+ for cT2) or pNSLN+ for cT1 disease (Lancet Oncol. 2012 Jan. 17 [doi:10.1016/S1470-2045(11)70397-7]).
They received neoadjuvant treatment including four cycles of epirubicin given at 90 mg/m2 intravenously along with cyclophosphamide at a dose of 600 mg/m2 intravenously, every 3 weeks, and four cycles of docetaxel at 100 mg/m2 intravenously every 3 weeks, plus either trastuzumab or lapatinib throughout all cycles prior to surgery. Trastuzumab was given at a starting loading dose of 8 mg/kg and then at 6 mg/kg intravenously every 3 weeks; the lapatinib dose was 1,000-1,250 mg/day orally.
"Pathological complete response rates were significantly lower with lapatinib treatment than with trastuzumab, irrespective of the definitions of pathological complete response that were used. These results confirm the efficacy of a neoadjuvant regimen containing trastuzumab," wrote Dr. Untch, of Helios-Klinikum, Berlin-Buch, Berlin, and his colleagues from the German Breast Group and the Arbeitsgemeinschaft Gynäkologische Onkologie-Breast (AGO-B) Study Group.
The investigators noted that lapatinib may provide a lower pathological complete response because of a reduced ability to block the HER2 pathway, compared with trastuzumab. Also, trastuzumab may have additional antitumor efficacy "by inducing an immune response via antibody-derived cellular cytotoxicity," they wrote.
In addition to providing additional evidence of the feasibility – and low cardiac toxicity – of "exposing the tumor for as long as possible to the synergistic effect of chemotherapy and trastuzumab before surgery," these findings suggest lapatinib should not be used outside of clinical trials as single anti-HER2 treatment in patients undergoing neoadjuvant chemotherapy, the investigators concluded.
Indeed, these findings "should lead to the conclusion that this regimen [the combination of lapatinib with standard chemotherapy] does not seem to have any advantages compared with a standard trastuzumab-based regimen for the adjuvant treatment of HER2-positive, early-stage breast cancer," Dr. Stephen K. Chia of the British Columbia Cancer Agency, Vancouver, B.C., wrote in an accompanying editorial (Lancet Oncol. 2012 Jan. 17 [doi:10.106/S1470-2045(12)70013-X]).
Another lesson learned from the GeparQuinto trial, according to Dr. Chia, is that the preoperative setting, which the research community and patients "seem to have embraced as both standard of care therapy and as an important strategy to test new therapeutic regimens," is ideally suited for this purpose.
"Moving forward into the future, no adjuvant trials should be done without an adequate signal from preoperative trials showing safety, efficacy, target modulation, and, ideally, the identification of predictive biomarkers such that we no longer pick a loser to study in larger and more resource-intensive adjuvant trials," he said. Dr. Chia noted that the second study demonstrating improved efficacy of trastuzumab – NeoALTTO – is a good example of such a preoperative study supporting the rationale for adjuvant trials with the combination of these agents, in this case the ALTTO (Adjuvant Lapatinib and/or Trastuzumab Treatment Optimization) trial, which is underway. The lapatinib group for ALTTO has been closed due to the demonstration of futility.
In NeoALTTO, which was published concomitantly with GeparQuinto in the Lancet, trastuzumab was more effective – although not significantly – than lapatinib alone for the treatment of HER2-positive breast cancer, but it was most effective in combination with lapatinib, Dr. José Baselga of Massachusetts General Hospital Cancer Center, Boston, and his colleagues from the NeoALTTO Study Team reported.
Pathological complete response in this parallel groups, open-label study, occurred in 51% of 152 patients in the combination treatment group, compared with 30% of the 149 patients in the trastuzumab group and 25% of the 154 patients in the lapatinib group, for an adjusted odds ratio of 2.6 for the combination vs. trastuzumab alone (Lancet 2012 Jan. 17 [doi:10.106/S0140-6736(11)61847-3]).
As in the GeparQuinto Trial, no major cardiac dysfunction occurred, and diarrhea was more frequent in the patients receiving lapatinib (23% and 21% of those in the lapatinib and combination groups, respectively, vs. 2% in the trastuzumab group who had grade 3 diarrhea).
NeoALTTO participants were enrolled between Jan. 5, 2008, and May 27, 2010, at 86 sites in 23 countries. Participants had histologically confirmed invasive HER2-positive breast cancer with tumors at least 2 cm in diameter. Lapatinib was given at 1,500 mg orally to those in the lapatinib-only group, and at 1,000 mg for those in the combination group; trastuzumab was given at a loading dose of 4 mg/m2 followed by 2 mg/kg on subsequent doses for both groups.
The anti-HER2 treatments were given alone for 6 weeks, and paclitaxel at 80 mg/m2 weekly was then added for an additional 12 weeks.
"Dual targeting of HER2-positive tumors with trastuzumab and lapatinib is undertaken because of primary and acquired resistance to both agents, their partly non-overlapping mechanisms of action, and the well characterized synergistic interaction between them in HER2 breast-cancer models," the investigators noted.
These findings provide proof of concept that dual inhibition of HER2 is better than a single-agent approach, Dr. Baselga and his colleagues said, noting that the higher pathological complete response rate for the combination was clear across all subgroups tested, which is consistent with the findings from other studies of anti-HER2 treatment in HER2-positive tumors.
"Our study also supports investigation of novel targeted agents for breast cancer in the neoadjuvant setting, when tumors have not yet acquired resistance to therapy and when chances of clinical benefit are highest," they said.
GeparQuinto was funded by GlaxoSmithKline, Roche, and Sanofi-Aventis; lapatinib was provided free of charge. Dr. Untch said he had no relevant financial disclosures, but several other GeparQuinto investigators made financial disclosures relating to these and other pharmaceutical companies. The NeoALTTO trial was funded by GlaxoSmithKline. Dr. Baselga said he has received honoraria from Roche, and his institution has received funding from GlaxoSmithKline and Roche. Several other NeoALTTO investigators made financial disclosures involving GlaxoSmithKline and/or other pharmaceutical companies. Dr. Chia, the author of the editorial that accompanied the article in the Lancet Oncology on the GeparQuinto trial, disclosed that he has received honoraria from GlaxoSmithKline, manufacturer of lapatinib, and F. Hoffmann-La Roche, manufacturer of trastuzumab, as well as an unrestricted research grant from Hoffmann-La Roche.
In an accompanying editorial, Dr. Michael Gnant and Dr. Guenther G. Steger highlighted the NeoALTTO study’s design as a "crucial scientific strength."
Delaying chemotherapy for 6 weeks while the targeted anti-HER2 treatment was initiated enabled collection of samples for translational research, as well as assessment of early tumor response without confounding by cytotoxic therapy.
"Such approaches should be used more often in pivotal trials of new drugs that target specific biological pathways, to enable unbiased efficacy assessments to be made. Equally important, such trials could help to identify clinically useful markers of early response, with the ultimate goal of tailoring neoadjuvant treatments for individual patients," they wrote (Lancet 2012 Jan. 17 [doi:10.106/S0140-6736(12)60068-3]).
In addition, on the basis of such neoadjuvant trials, the traditional sequence of drug testing first in advanced disease, then in the neoadjuvant setting, and finally in the adjuvant setting, could be revised.
"In the future, assessment of a pathway-directed therapeutic intervention in rigorous neoadjuvant trials might be sufficient for validity in a biomarker-defined population of patients to be accepted," Dr. Gnant and Dr. Steger suggested. Trials in such a setting, after drug safety is established, could lead to large savings in drug development costs, and to much quicker availability of promising new drugs for the treatment of early breast cancer, they noted.
Dr. Gnant and Dr. Steger are with the Comprehensive Cancer Centre of the Medical University of Vienna. Dr. Gnant has served on advisory boards for and has received consulting fees from AstraZeneca and Novartis, and has received lecture fees and/or research support from Roche, Schering, Pfizer, Novartis, AstraZeneca, Sanofi-Aventis, GlaxoSmithKline, and Amgen. Dr. Steger has served on advisory boards for and has received consulting fees from AstraZeneca, Roche, and Amgen, and has received lecture fees and research support from AstraZeneca, Novartis, Roche, GlaxoSmithKline, and Amgen.
In an accompanying editorial, Dr. Michael Gnant and Dr. Guenther G. Steger highlighted the NeoALTTO study’s design as a "crucial scientific strength."
Delaying chemotherapy for 6 weeks while the targeted anti-HER2 treatment was initiated enabled collection of samples for translational research, as well as assessment of early tumor response without confounding by cytotoxic therapy.
"Such approaches should be used more often in pivotal trials of new drugs that target specific biological pathways, to enable unbiased efficacy assessments to be made. Equally important, such trials could help to identify clinically useful markers of early response, with the ultimate goal of tailoring neoadjuvant treatments for individual patients," they wrote (Lancet 2012 Jan. 17 [doi:10.106/S0140-6736(12)60068-3]).
In addition, on the basis of such neoadjuvant trials, the traditional sequence of drug testing first in advanced disease, then in the neoadjuvant setting, and finally in the adjuvant setting, could be revised.
"In the future, assessment of a pathway-directed therapeutic intervention in rigorous neoadjuvant trials might be sufficient for validity in a biomarker-defined population of patients to be accepted," Dr. Gnant and Dr. Steger suggested. Trials in such a setting, after drug safety is established, could lead to large savings in drug development costs, and to much quicker availability of promising new drugs for the treatment of early breast cancer, they noted.
Dr. Gnant and Dr. Steger are with the Comprehensive Cancer Centre of the Medical University of Vienna. Dr. Gnant has served on advisory boards for and has received consulting fees from AstraZeneca and Novartis, and has received lecture fees and/or research support from Roche, Schering, Pfizer, Novartis, AstraZeneca, Sanofi-Aventis, GlaxoSmithKline, and Amgen. Dr. Steger has served on advisory boards for and has received consulting fees from AstraZeneca, Roche, and Amgen, and has received lecture fees and research support from AstraZeneca, Novartis, Roche, GlaxoSmithKline, and Amgen.
In an accompanying editorial, Dr. Michael Gnant and Dr. Guenther G. Steger highlighted the NeoALTTO study’s design as a "crucial scientific strength."
Delaying chemotherapy for 6 weeks while the targeted anti-HER2 treatment was initiated enabled collection of samples for translational research, as well as assessment of early tumor response without confounding by cytotoxic therapy.
"Such approaches should be used more often in pivotal trials of new drugs that target specific biological pathways, to enable unbiased efficacy assessments to be made. Equally important, such trials could help to identify clinically useful markers of early response, with the ultimate goal of tailoring neoadjuvant treatments for individual patients," they wrote (Lancet 2012 Jan. 17 [doi:10.106/S0140-6736(12)60068-3]).
In addition, on the basis of such neoadjuvant trials, the traditional sequence of drug testing first in advanced disease, then in the neoadjuvant setting, and finally in the adjuvant setting, could be revised.
"In the future, assessment of a pathway-directed therapeutic intervention in rigorous neoadjuvant trials might be sufficient for validity in a biomarker-defined population of patients to be accepted," Dr. Gnant and Dr. Steger suggested. Trials in such a setting, after drug safety is established, could lead to large savings in drug development costs, and to much quicker availability of promising new drugs for the treatment of early breast cancer, they noted.
Dr. Gnant and Dr. Steger are with the Comprehensive Cancer Centre of the Medical University of Vienna. Dr. Gnant has served on advisory boards for and has received consulting fees from AstraZeneca and Novartis, and has received lecture fees and/or research support from Roche, Schering, Pfizer, Novartis, AstraZeneca, Sanofi-Aventis, GlaxoSmithKline, and Amgen. Dr. Steger has served on advisory boards for and has received consulting fees from AstraZeneca, Roche, and Amgen, and has received lecture fees and research support from AstraZeneca, Novartis, Roche, GlaxoSmithKline, and Amgen.
Trastuzumab is significantly more effective alone or in combination with lapatinib than is lapatinib alone for the treatment of human epidermal growth factor receptor 2–positive breast cancer in patients receiving neoadjuvant chemotherapy, according to findings from two randomized phase III trials.
The studies – GeparQuinto and the NeoALTTO (Neoadjuvant Lapatinib and/or Trastuzumab Treatment Optimization) trial – underscore the value of testing new therapies in the neoadjuvant setting.
In the GeparQuinto trial, 30% of 307 HER2-positive patients treated with the anti-HER2 humanized monoclonal antibody trastuzumab had a pathological complete response, compared with only 23% of 308 HER2-positive patients treated with the tyrosine kinase inhibitor lapatinib, which targets both HER1 and HER2 (odds ratio, 0.68).
Although the patients randomized to receive trastuzumab had significantly more edema (39% vs. 29%) and dyspnea (30% vs. 21%), those randomized to receive lapatinib experienced significantly more diarrhea (75% vs. 47%) and skin rash (55% vs. 32%), and significantly more patients in the lapatinib group discontinued treatment (33% vs. 14%), Dr. Michael Untch and his colleagues reported in the Jan. 17 issue of the Lancet Oncology.
Women with previously untreated unilateral or bilateral primary invasive breast carcinoma were enrolled in the study between Nov. 7, 2007, and July 9, 2010, at 126 centers in Germany and 1 center in Switzerland. Patients were eligible for inclusion if they had locally advanced tumor stages cT3 or cT4, hormone receptor (HR)-negative tumors, or HR-positive tumors with clinically positive axillary nodes (cN+ for cT2) or pNSLN+ for cT1 disease (Lancet Oncol. 2012 Jan. 17 [doi:10.1016/S1470-2045(11)70397-7]).
They received neoadjuvant treatment including four cycles of epirubicin given at 90 mg/m2 intravenously along with cyclophosphamide at a dose of 600 mg/m2 intravenously, every 3 weeks, and four cycles of docetaxel at 100 mg/m2 intravenously every 3 weeks, plus either trastuzumab or lapatinib throughout all cycles prior to surgery. Trastuzumab was given at a starting loading dose of 8 mg/kg and then at 6 mg/kg intravenously every 3 weeks; the lapatinib dose was 1,000-1,250 mg/day orally.
"Pathological complete response rates were significantly lower with lapatinib treatment than with trastuzumab, irrespective of the definitions of pathological complete response that were used. These results confirm the efficacy of a neoadjuvant regimen containing trastuzumab," wrote Dr. Untch, of Helios-Klinikum, Berlin-Buch, Berlin, and his colleagues from the German Breast Group and the Arbeitsgemeinschaft Gynäkologische Onkologie-Breast (AGO-B) Study Group.
The investigators noted that lapatinib may provide a lower pathological complete response because of a reduced ability to block the HER2 pathway, compared with trastuzumab. Also, trastuzumab may have additional antitumor efficacy "by inducing an immune response via antibody-derived cellular cytotoxicity," they wrote.
In addition to providing additional evidence of the feasibility – and low cardiac toxicity – of "exposing the tumor for as long as possible to the synergistic effect of chemotherapy and trastuzumab before surgery," these findings suggest lapatinib should not be used outside of clinical trials as single anti-HER2 treatment in patients undergoing neoadjuvant chemotherapy, the investigators concluded.
Indeed, these findings "should lead to the conclusion that this regimen [the combination of lapatinib with standard chemotherapy] does not seem to have any advantages compared with a standard trastuzumab-based regimen for the adjuvant treatment of HER2-positive, early-stage breast cancer," Dr. Stephen K. Chia of the British Columbia Cancer Agency, Vancouver, B.C., wrote in an accompanying editorial (Lancet Oncol. 2012 Jan. 17 [doi:10.106/S1470-2045(12)70013-X]).
Another lesson learned from the GeparQuinto trial, according to Dr. Chia, is that the preoperative setting, which the research community and patients "seem to have embraced as both standard of care therapy and as an important strategy to test new therapeutic regimens," is ideally suited for this purpose.
"Moving forward into the future, no adjuvant trials should be done without an adequate signal from preoperative trials showing safety, efficacy, target modulation, and, ideally, the identification of predictive biomarkers such that we no longer pick a loser to study in larger and more resource-intensive adjuvant trials," he said. Dr. Chia noted that the second study demonstrating improved efficacy of trastuzumab – NeoALTTO – is a good example of such a preoperative study supporting the rationale for adjuvant trials with the combination of these agents, in this case the ALTTO (Adjuvant Lapatinib and/or Trastuzumab Treatment Optimization) trial, which is underway. The lapatinib group for ALTTO has been closed due to the demonstration of futility.
In NeoALTTO, which was published concomitantly with GeparQuinto in the Lancet, trastuzumab was more effective – although not significantly – than lapatinib alone for the treatment of HER2-positive breast cancer, but it was most effective in combination with lapatinib, Dr. José Baselga of Massachusetts General Hospital Cancer Center, Boston, and his colleagues from the NeoALTTO Study Team reported.
Pathological complete response in this parallel groups, open-label study, occurred in 51% of 152 patients in the combination treatment group, compared with 30% of the 149 patients in the trastuzumab group and 25% of the 154 patients in the lapatinib group, for an adjusted odds ratio of 2.6 for the combination vs. trastuzumab alone (Lancet 2012 Jan. 17 [doi:10.106/S0140-6736(11)61847-3]).
As in the GeparQuinto Trial, no major cardiac dysfunction occurred, and diarrhea was more frequent in the patients receiving lapatinib (23% and 21% of those in the lapatinib and combination groups, respectively, vs. 2% in the trastuzumab group who had grade 3 diarrhea).
NeoALTTO participants were enrolled between Jan. 5, 2008, and May 27, 2010, at 86 sites in 23 countries. Participants had histologically confirmed invasive HER2-positive breast cancer with tumors at least 2 cm in diameter. Lapatinib was given at 1,500 mg orally to those in the lapatinib-only group, and at 1,000 mg for those in the combination group; trastuzumab was given at a loading dose of 4 mg/m2 followed by 2 mg/kg on subsequent doses for both groups.
The anti-HER2 treatments were given alone for 6 weeks, and paclitaxel at 80 mg/m2 weekly was then added for an additional 12 weeks.
"Dual targeting of HER2-positive tumors with trastuzumab and lapatinib is undertaken because of primary and acquired resistance to both agents, their partly non-overlapping mechanisms of action, and the well characterized synergistic interaction between them in HER2 breast-cancer models," the investigators noted.
These findings provide proof of concept that dual inhibition of HER2 is better than a single-agent approach, Dr. Baselga and his colleagues said, noting that the higher pathological complete response rate for the combination was clear across all subgroups tested, which is consistent with the findings from other studies of anti-HER2 treatment in HER2-positive tumors.
"Our study also supports investigation of novel targeted agents for breast cancer in the neoadjuvant setting, when tumors have not yet acquired resistance to therapy and when chances of clinical benefit are highest," they said.
GeparQuinto was funded by GlaxoSmithKline, Roche, and Sanofi-Aventis; lapatinib was provided free of charge. Dr. Untch said he had no relevant financial disclosures, but several other GeparQuinto investigators made financial disclosures relating to these and other pharmaceutical companies. The NeoALTTO trial was funded by GlaxoSmithKline. Dr. Baselga said he has received honoraria from Roche, and his institution has received funding from GlaxoSmithKline and Roche. Several other NeoALTTO investigators made financial disclosures involving GlaxoSmithKline and/or other pharmaceutical companies. Dr. Chia, the author of the editorial that accompanied the article in the Lancet Oncology on the GeparQuinto trial, disclosed that he has received honoraria from GlaxoSmithKline, manufacturer of lapatinib, and F. Hoffmann-La Roche, manufacturer of trastuzumab, as well as an unrestricted research grant from Hoffmann-La Roche.
Trastuzumab is significantly more effective alone or in combination with lapatinib than is lapatinib alone for the treatment of human epidermal growth factor receptor 2–positive breast cancer in patients receiving neoadjuvant chemotherapy, according to findings from two randomized phase III trials.
The studies – GeparQuinto and the NeoALTTO (Neoadjuvant Lapatinib and/or Trastuzumab Treatment Optimization) trial – underscore the value of testing new therapies in the neoadjuvant setting.
In the GeparQuinto trial, 30% of 307 HER2-positive patients treated with the anti-HER2 humanized monoclonal antibody trastuzumab had a pathological complete response, compared with only 23% of 308 HER2-positive patients treated with the tyrosine kinase inhibitor lapatinib, which targets both HER1 and HER2 (odds ratio, 0.68).
Although the patients randomized to receive trastuzumab had significantly more edema (39% vs. 29%) and dyspnea (30% vs. 21%), those randomized to receive lapatinib experienced significantly more diarrhea (75% vs. 47%) and skin rash (55% vs. 32%), and significantly more patients in the lapatinib group discontinued treatment (33% vs. 14%), Dr. Michael Untch and his colleagues reported in the Jan. 17 issue of the Lancet Oncology.
Women with previously untreated unilateral or bilateral primary invasive breast carcinoma were enrolled in the study between Nov. 7, 2007, and July 9, 2010, at 126 centers in Germany and 1 center in Switzerland. Patients were eligible for inclusion if they had locally advanced tumor stages cT3 or cT4, hormone receptor (HR)-negative tumors, or HR-positive tumors with clinically positive axillary nodes (cN+ for cT2) or pNSLN+ for cT1 disease (Lancet Oncol. 2012 Jan. 17 [doi:10.1016/S1470-2045(11)70397-7]).
They received neoadjuvant treatment including four cycles of epirubicin given at 90 mg/m2 intravenously along with cyclophosphamide at a dose of 600 mg/m2 intravenously, every 3 weeks, and four cycles of docetaxel at 100 mg/m2 intravenously every 3 weeks, plus either trastuzumab or lapatinib throughout all cycles prior to surgery. Trastuzumab was given at a starting loading dose of 8 mg/kg and then at 6 mg/kg intravenously every 3 weeks; the lapatinib dose was 1,000-1,250 mg/day orally.
"Pathological complete response rates were significantly lower with lapatinib treatment than with trastuzumab, irrespective of the definitions of pathological complete response that were used. These results confirm the efficacy of a neoadjuvant regimen containing trastuzumab," wrote Dr. Untch, of Helios-Klinikum, Berlin-Buch, Berlin, and his colleagues from the German Breast Group and the Arbeitsgemeinschaft Gynäkologische Onkologie-Breast (AGO-B) Study Group.
The investigators noted that lapatinib may provide a lower pathological complete response because of a reduced ability to block the HER2 pathway, compared with trastuzumab. Also, trastuzumab may have additional antitumor efficacy "by inducing an immune response via antibody-derived cellular cytotoxicity," they wrote.
In addition to providing additional evidence of the feasibility – and low cardiac toxicity – of "exposing the tumor for as long as possible to the synergistic effect of chemotherapy and trastuzumab before surgery," these findings suggest lapatinib should not be used outside of clinical trials as single anti-HER2 treatment in patients undergoing neoadjuvant chemotherapy, the investigators concluded.
Indeed, these findings "should lead to the conclusion that this regimen [the combination of lapatinib with standard chemotherapy] does not seem to have any advantages compared with a standard trastuzumab-based regimen for the adjuvant treatment of HER2-positive, early-stage breast cancer," Dr. Stephen K. Chia of the British Columbia Cancer Agency, Vancouver, B.C., wrote in an accompanying editorial (Lancet Oncol. 2012 Jan. 17 [doi:10.106/S1470-2045(12)70013-X]).
Another lesson learned from the GeparQuinto trial, according to Dr. Chia, is that the preoperative setting, which the research community and patients "seem to have embraced as both standard of care therapy and as an important strategy to test new therapeutic regimens," is ideally suited for this purpose.
"Moving forward into the future, no adjuvant trials should be done without an adequate signal from preoperative trials showing safety, efficacy, target modulation, and, ideally, the identification of predictive biomarkers such that we no longer pick a loser to study in larger and more resource-intensive adjuvant trials," he said. Dr. Chia noted that the second study demonstrating improved efficacy of trastuzumab – NeoALTTO – is a good example of such a preoperative study supporting the rationale for adjuvant trials with the combination of these agents, in this case the ALTTO (Adjuvant Lapatinib and/or Trastuzumab Treatment Optimization) trial, which is underway. The lapatinib group for ALTTO has been closed due to the demonstration of futility.
In NeoALTTO, which was published concomitantly with GeparQuinto in the Lancet, trastuzumab was more effective – although not significantly – than lapatinib alone for the treatment of HER2-positive breast cancer, but it was most effective in combination with lapatinib, Dr. José Baselga of Massachusetts General Hospital Cancer Center, Boston, and his colleagues from the NeoALTTO Study Team reported.
Pathological complete response in this parallel groups, open-label study, occurred in 51% of 152 patients in the combination treatment group, compared with 30% of the 149 patients in the trastuzumab group and 25% of the 154 patients in the lapatinib group, for an adjusted odds ratio of 2.6 for the combination vs. trastuzumab alone (Lancet 2012 Jan. 17 [doi:10.106/S0140-6736(11)61847-3]).
As in the GeparQuinto Trial, no major cardiac dysfunction occurred, and diarrhea was more frequent in the patients receiving lapatinib (23% and 21% of those in the lapatinib and combination groups, respectively, vs. 2% in the trastuzumab group who had grade 3 diarrhea).
NeoALTTO participants were enrolled between Jan. 5, 2008, and May 27, 2010, at 86 sites in 23 countries. Participants had histologically confirmed invasive HER2-positive breast cancer with tumors at least 2 cm in diameter. Lapatinib was given at 1,500 mg orally to those in the lapatinib-only group, and at 1,000 mg for those in the combination group; trastuzumab was given at a loading dose of 4 mg/m2 followed by 2 mg/kg on subsequent doses for both groups.
The anti-HER2 treatments were given alone for 6 weeks, and paclitaxel at 80 mg/m2 weekly was then added for an additional 12 weeks.
"Dual targeting of HER2-positive tumors with trastuzumab and lapatinib is undertaken because of primary and acquired resistance to both agents, their partly non-overlapping mechanisms of action, and the well characterized synergistic interaction between them in HER2 breast-cancer models," the investigators noted.
These findings provide proof of concept that dual inhibition of HER2 is better than a single-agent approach, Dr. Baselga and his colleagues said, noting that the higher pathological complete response rate for the combination was clear across all subgroups tested, which is consistent with the findings from other studies of anti-HER2 treatment in HER2-positive tumors.
"Our study also supports investigation of novel targeted agents for breast cancer in the neoadjuvant setting, when tumors have not yet acquired resistance to therapy and when chances of clinical benefit are highest," they said.
GeparQuinto was funded by GlaxoSmithKline, Roche, and Sanofi-Aventis; lapatinib was provided free of charge. Dr. Untch said he had no relevant financial disclosures, but several other GeparQuinto investigators made financial disclosures relating to these and other pharmaceutical companies. The NeoALTTO trial was funded by GlaxoSmithKline. Dr. Baselga said he has received honoraria from Roche, and his institution has received funding from GlaxoSmithKline and Roche. Several other NeoALTTO investigators made financial disclosures involving GlaxoSmithKline and/or other pharmaceutical companies. Dr. Chia, the author of the editorial that accompanied the article in the Lancet Oncology on the GeparQuinto trial, disclosed that he has received honoraria from GlaxoSmithKline, manufacturer of lapatinib, and F. Hoffmann-La Roche, manufacturer of trastuzumab, as well as an unrestricted research grant from Hoffmann-La Roche.
FROM THE LANCET AND THE LANCET ONCOLOGY
Major Finding: In the GeparQuinto trial, 30% of 307 HER2-positive patients treated with trastuzumab had a pathological complete response, compared with only 23% of 308 HER2-positive patients treated with lapatinib. In NeoALTTO, trastuzumab was more effective than lapatinib alone for the treatment of HER2-positive breast cancer, but it was most effective in combination with lapatinib. Pathological complete response occurred in 51% of 152 patients in the combination treatment group, compared with 30% of the 149 patients in the trastuzumab group and 25% of the 154 patients in the lapatinib group, for an adjusted odds ratio of 2.6 for the combination vs. trastuzumab alone.
Data Source: Two randomized phase III trials – GeparQuinto and NeoALTTO.
Disclosures: GeparQuinto was funded by GlaxoSmithKline, Roche, and Sanofi-Aventis; lapatinib was provided free of charge. Dr. Untch said he had no relevant financial disclosures, but several other GeparQuinto investigators made financial disclosures relating to these and other pharmaceutical companies. The NeoALTTO trial was funded by GlaxoSmithKline. Dr. Baselga said he has received honoraria from Roche, and his institution has received funding from GlaxoSmithKline and Roche. Several other NeoALTTO investigators made financial disclosures involving GlaxoSmithKline and/or other pharmaceutical companies. Dr. Chia, the author of the editorial that accompanied the article in the Lancet Oncology on the GeparQuinto trial, disclosed that he has received honoraria from GlaxoSmithKline, manufacturer of lapatinib, and F. Hoffmann-La Roche, manufacturer of trastuzumab, as well as an unrestricted research grant from Hoffmann-La Roche.
Obesity in U.S. Appears to Be Leveling Off
The prevalence of obesity in the United States appears to have plateaued, according to data from the 2009-2010 National Health and Nutrition Examination Survey (NHANES) conducted by the U.S. Centers for Disease Control and Prevention.
Following dramatic increases in the prevalence of obesity in adults, children, and adolescents in the 1980s and 1990s, as well as changes in the distribution of body mass index, no significant changes were seen in 2009-2010, compared with 2003-2008 figures in adults, and compared with 2007-2008 prevalence rates in children and adolescents.
For example, based on data from the 5,926 men and women with measured weight and height who participated in the 2009-2010 NHANES, the age-adjusted prevalence of obesity was roughly 35% for both men and women, which was not significantly different overall compared with the prevalence from 2003-2008, Katherine M. Flegal, Ph.D., and her colleagues from the National Center for Health Statistics, CDC, Hyattsville, Md., report online in the Jan. 17 JAMA.
Despite the lack of change overall, the analysis of adult data did indicate, however, that obesity increased in certain segments of the population. While no significant increase was seen among women overall (age- and race-adjusted annual change in odds ratio from 1999-2010, 1.01), statistically significant increases were seen among non-Hispanic black women (OR, 1.03) and Mexican American women (1.03), the investigators noted (JAMA 2012 Jan. 17 [doi:10.1001/jama.2012.39]).
A significant linear trend was also seen in men over the 12-year period (annual change in OR, 1.04).
As for BMI, the age-adjusted mean in both men and women was 28.7, and the trends over time in this study were similar to those seen with obesity, with a significant increase seen in men, but not in women, over the 12 years, the investigators said.
In a separate cross-sectional analysis of data from 4,111 children and adolescents who participated in NHANES, the 2009-2010 obesity prevalence of 9.7% in infants and toddlers up to age 2 years, and 16.9% for those aged 2-19 years, did not differ significantly from the 2007-2008 prevalence, and no difference was seen between males and females in regard to obesity prevalence, Cynthia L. Ogden, Ph.D., and her colleagues, also from the National Center for Health Statistics, CDC, reported in the same issue of JAMA.
A trend analysis over the 12-year study period did indicate, however, that the obesity prevalence among males aged 2-19 years increased significantly between 1999-2000 and 2009-2010 (OR, 1.05) per 2-year survey cycle, and that there was a significant increasing trend for non-Hispanic black males (OR, 1.10). Also, BMI increased significantly in males aged 12 through 19 years (JAMA 2012 Jan. 17 [doi:10.1001/jama.2012.40]).
NHANES data have been collected continuously since 1999, with reports released in 2-year cycles. Since no universally agreed upon definition of obesity exists for infants and toddlers up to age 2 years, high weight in this age group was defined as weight-for-recumbent length at or above the 95th percentile on the CDCs 2000 growth charts, the investigators explained.
Weight status in those aged 2 through 19 years is defined based on BMI; those at or above the sex-specific 85th percentile, but less than the 95th percentile, are considered overweight, and those at or above the sex-specific 95th percentile are considered obese. For adults, those with a BMI of 25.0 to 29.9 are considered overweight, and those with a BMI of 30.0 or higher are considered obese, with further subdivision into grades 1, 2, and 3 obesity based on BMI of 30.0 to less than 35.0, 35.0 to less than 40.0, and 40.0 or greater, respectively.
Investigators for both analyses noted that it is important to keep in mind that BMI is an "imperfect measure of body fat."
Since racial and ethnic differences in the level of body fat at specific BMIs exist, the differences in obesity prevalence by race and ethnicity in these studies may not represent actual differences in body fat, they said.
Overall, the findings in both adults and children/adolescents suggest that increases in the prevalence of obesity that have previously been observed are not continuing and have leveled off.
Although it’s difficult to predict whether these trends will continue in the same direction, the findings suggest that previous models that predict continuing increases in obesity prevalence in all age groups may be based on invalid assumptions.
None of the authors indicated having relevant conflicts of interest.
The prevalence of obesity in the United States appears to have plateaued, according to data from the 2009-2010 National Health and Nutrition Examination Survey (NHANES) conducted by the U.S. Centers for Disease Control and Prevention.
Following dramatic increases in the prevalence of obesity in adults, children, and adolescents in the 1980s and 1990s, as well as changes in the distribution of body mass index, no significant changes were seen in 2009-2010, compared with 2003-2008 figures in adults, and compared with 2007-2008 prevalence rates in children and adolescents.
For example, based on data from the 5,926 men and women with measured weight and height who participated in the 2009-2010 NHANES, the age-adjusted prevalence of obesity was roughly 35% for both men and women, which was not significantly different overall compared with the prevalence from 2003-2008, Katherine M. Flegal, Ph.D., and her colleagues from the National Center for Health Statistics, CDC, Hyattsville, Md., report online in the Jan. 17 JAMA.
Despite the lack of change overall, the analysis of adult data did indicate, however, that obesity increased in certain segments of the population. While no significant increase was seen among women overall (age- and race-adjusted annual change in odds ratio from 1999-2010, 1.01), statistically significant increases were seen among non-Hispanic black women (OR, 1.03) and Mexican American women (1.03), the investigators noted (JAMA 2012 Jan. 17 [doi:10.1001/jama.2012.39]).
A significant linear trend was also seen in men over the 12-year period (annual change in OR, 1.04).
As for BMI, the age-adjusted mean in both men and women was 28.7, and the trends over time in this study were similar to those seen with obesity, with a significant increase seen in men, but not in women, over the 12 years, the investigators said.
In a separate cross-sectional analysis of data from 4,111 children and adolescents who participated in NHANES, the 2009-2010 obesity prevalence of 9.7% in infants and toddlers up to age 2 years, and 16.9% for those aged 2-19 years, did not differ significantly from the 2007-2008 prevalence, and no difference was seen between males and females in regard to obesity prevalence, Cynthia L. Ogden, Ph.D., and her colleagues, also from the National Center for Health Statistics, CDC, reported in the same issue of JAMA.
A trend analysis over the 12-year study period did indicate, however, that the obesity prevalence among males aged 2-19 years increased significantly between 1999-2000 and 2009-2010 (OR, 1.05) per 2-year survey cycle, and that there was a significant increasing trend for non-Hispanic black males (OR, 1.10). Also, BMI increased significantly in males aged 12 through 19 years (JAMA 2012 Jan. 17 [doi:10.1001/jama.2012.40]).
NHANES data have been collected continuously since 1999, with reports released in 2-year cycles. Since no universally agreed upon definition of obesity exists for infants and toddlers up to age 2 years, high weight in this age group was defined as weight-for-recumbent length at or above the 95th percentile on the CDCs 2000 growth charts, the investigators explained.
Weight status in those aged 2 through 19 years is defined based on BMI; those at or above the sex-specific 85th percentile, but less than the 95th percentile, are considered overweight, and those at or above the sex-specific 95th percentile are considered obese. For adults, those with a BMI of 25.0 to 29.9 are considered overweight, and those with a BMI of 30.0 or higher are considered obese, with further subdivision into grades 1, 2, and 3 obesity based on BMI of 30.0 to less than 35.0, 35.0 to less than 40.0, and 40.0 or greater, respectively.
Investigators for both analyses noted that it is important to keep in mind that BMI is an "imperfect measure of body fat."
Since racial and ethnic differences in the level of body fat at specific BMIs exist, the differences in obesity prevalence by race and ethnicity in these studies may not represent actual differences in body fat, they said.
Overall, the findings in both adults and children/adolescents suggest that increases in the prevalence of obesity that have previously been observed are not continuing and have leveled off.
Although it’s difficult to predict whether these trends will continue in the same direction, the findings suggest that previous models that predict continuing increases in obesity prevalence in all age groups may be based on invalid assumptions.
None of the authors indicated having relevant conflicts of interest.
The prevalence of obesity in the United States appears to have plateaued, according to data from the 2009-2010 National Health and Nutrition Examination Survey (NHANES) conducted by the U.S. Centers for Disease Control and Prevention.
Following dramatic increases in the prevalence of obesity in adults, children, and adolescents in the 1980s and 1990s, as well as changes in the distribution of body mass index, no significant changes were seen in 2009-2010, compared with 2003-2008 figures in adults, and compared with 2007-2008 prevalence rates in children and adolescents.
For example, based on data from the 5,926 men and women with measured weight and height who participated in the 2009-2010 NHANES, the age-adjusted prevalence of obesity was roughly 35% for both men and women, which was not significantly different overall compared with the prevalence from 2003-2008, Katherine M. Flegal, Ph.D., and her colleagues from the National Center for Health Statistics, CDC, Hyattsville, Md., report online in the Jan. 17 JAMA.
Despite the lack of change overall, the analysis of adult data did indicate, however, that obesity increased in certain segments of the population. While no significant increase was seen among women overall (age- and race-adjusted annual change in odds ratio from 1999-2010, 1.01), statistically significant increases were seen among non-Hispanic black women (OR, 1.03) and Mexican American women (1.03), the investigators noted (JAMA 2012 Jan. 17 [doi:10.1001/jama.2012.39]).
A significant linear trend was also seen in men over the 12-year period (annual change in OR, 1.04).
As for BMI, the age-adjusted mean in both men and women was 28.7, and the trends over time in this study were similar to those seen with obesity, with a significant increase seen in men, but not in women, over the 12 years, the investigators said.
In a separate cross-sectional analysis of data from 4,111 children and adolescents who participated in NHANES, the 2009-2010 obesity prevalence of 9.7% in infants and toddlers up to age 2 years, and 16.9% for those aged 2-19 years, did not differ significantly from the 2007-2008 prevalence, and no difference was seen between males and females in regard to obesity prevalence, Cynthia L. Ogden, Ph.D., and her colleagues, also from the National Center for Health Statistics, CDC, reported in the same issue of JAMA.
A trend analysis over the 12-year study period did indicate, however, that the obesity prevalence among males aged 2-19 years increased significantly between 1999-2000 and 2009-2010 (OR, 1.05) per 2-year survey cycle, and that there was a significant increasing trend for non-Hispanic black males (OR, 1.10). Also, BMI increased significantly in males aged 12 through 19 years (JAMA 2012 Jan. 17 [doi:10.1001/jama.2012.40]).
NHANES data have been collected continuously since 1999, with reports released in 2-year cycles. Since no universally agreed upon definition of obesity exists for infants and toddlers up to age 2 years, high weight in this age group was defined as weight-for-recumbent length at or above the 95th percentile on the CDCs 2000 growth charts, the investigators explained.
Weight status in those aged 2 through 19 years is defined based on BMI; those at or above the sex-specific 85th percentile, but less than the 95th percentile, are considered overweight, and those at or above the sex-specific 95th percentile are considered obese. For adults, those with a BMI of 25.0 to 29.9 are considered overweight, and those with a BMI of 30.0 or higher are considered obese, with further subdivision into grades 1, 2, and 3 obesity based on BMI of 30.0 to less than 35.0, 35.0 to less than 40.0, and 40.0 or greater, respectively.
Investigators for both analyses noted that it is important to keep in mind that BMI is an "imperfect measure of body fat."
Since racial and ethnic differences in the level of body fat at specific BMIs exist, the differences in obesity prevalence by race and ethnicity in these studies may not represent actual differences in body fat, they said.
Overall, the findings in both adults and children/adolescents suggest that increases in the prevalence of obesity that have previously been observed are not continuing and have leveled off.
Although it’s difficult to predict whether these trends will continue in the same direction, the findings suggest that previous models that predict continuing increases in obesity prevalence in all age groups may be based on invalid assumptions.
None of the authors indicated having relevant conflicts of interest.
FROM JAMA
Major Finding: Following dramatic increases in the prevalence of obesity in adults, children, and adolescents in the 1980s and 1990s, as well as changes in the distribution of body mass index, no significant changes were seen in 2009-2010, compared with 2003-2008 in adults, and compared with 2007-2008 in children and adolescents.
Data Source: Two analyses of data from the 1999-2010 National Health and Nutrition Examination Surveys (NHANES).
Disclosures: The study authors reported having no relevant conflicts of interest.
Memantine Disappoints for Dementia in Down Syndrome
Memantine is not an effective treatment for dementia in adults older than 40 years with Down syndrome, according to findings from a randomized, placebo-controlled study.
In fact, all of the 173 participants in the Memantine for Dementia in Adults Older than 40 Years with Down Syndrome (MEADOWS) trial, regardless of whether they received placebo or active treatment, experienced declines in cognition and function during the 52-week study. Moreover, although the rates of decline did not differ significantly between the groups, they were slightly lower in the placebo group, senior author Dr. Clive Ballard of King’s College London (England) and his colleagues reported online in the January 10 issue of The Lancet.
The findings suggest that therapies that are effective for Alzheimer’s disease in other patient populations are not necessarily effective in those with Down syndrome.
After adjustment for baseline score, participants in the placebo group and treatment group experienced mean changes in scores on the Down syndrome attention, memory, and executive function scales (DAMES) of -1.9 and -5.6 at 52 weeks, respectively. There also was no difference in the mean changes in scores between the placebo and memantine groups in part I of the Adaptive Behavior Scale (-1.7 and -10.7, respectively), which measures independent functioning, or part II (0 and 1.0, respectively), which measures challenging behavior (Lancet 2011 Jan. 10 [doi:10.1016/S0140-6736(11)61676-0]).
The results were similar regardless of the presence of dementia, which had been diagnosed in 35% of individuals in each group at the time of randomization.
The rate of serious adverse events also did not differ significantly between the placebo and treatment groups at 7% and 11%, respectively.
The findings have implications for research strategy in development of treatments for cognitive decline and dementia in individuals with Down syndrome, the investigators said.
Dementia is very prevalent in this population, with nearly 40% of those with Down syndrome receiving a diagnosis of dementia after age 60 years. Additionally, clinically significant Alzheimer’s disease–like pathological features are "almost ubiquitous" by age 40 years in those with Down syndrome.
"Although there is therefore a rationale for the efficacy of pharmacological treatments for Alzheimer’s disease in people with Down’s syndrome, there are some key differences in pathological features that should not be overlooked. For example, people with Down’s syndrome have lifelong overproduction of amyloid-beta compounded with dysregulation of many genes, most of which are not associated with Alzheimer’s disease," the investigators explained, adding that differences in the neuropathologic features of people with Down syndrome compared with Alzheimer’s disease might affect responses to pharmacologic therapies.
The N-methyl-d-aspartate glutamate receptor antagonist memantine (Namenda) is licensed for the treatment of moderate to severe Alzheimer’s disease and showed promise in two studies in a Down syndrome model in mice. But in the current study it was found to confer no benefit, regardless of whether a formal diagnosis of dementia was available, they said.
Study participants were men and women with Down syndrome aged 40 years or older or individuals of any age with Down syndrome and an established diagnosis of dementia. They were enrolled at four learning disability centers throughout the United Kingdom and Norway. The randomization process involved a computer-generated allocation sequence that ensured balanced allocation based on the prognostic factors of sex, presence of dementia, age group, DAMES score, and center. The investigators escalated the dose of memantine over 8 weeks from 5 mg per day to the optimal therapeutic dose of 10 mg per day with fixed titration.
"The findings of this study are robust and represent an important step in the evidence base for treatment of people with Down syndrome with cognitive decline," the investigators wrote, concluding that research into specific therapies for this patient population needs to be based on an understanding of underlying pathologic processes.
In particular, an adequately powered randomized controlled trial of cholinesterase inhibitors is needed to determine if this class of drug has any role in the treatment of Down syndrome patients with dementia. Cholinesterase inhibitors are approved in the United Kingdom for people with dementia in the context of Down syndrome, but very little evidence exists to support this guidance, they said.
This study was funded by Lundbeck and sponsored by King’s College London. Dr. Ballard reported receiving consultancy and speaking fees from Lundbeck, Janssen, Novartis, and Acadia and grants from Lundbeck and Acadia. Other study authors reported receiving consultancy fees, speaking fees, honoraria, and/or grants from Lundbeck, Novartis, and/or Roche, as well as serving on the advisory board for Lundbeck, providing paid expert testimony for Novartis and Eisai, and receiving funding from the Alzheimer’s Society and the Henry Smith Trust. Detailed disclosures are available with the full text of the article at www.thelancet.com.
Though disappointing, the findings of the MEADOWS trial do serve to exclude treatments of little or no benefit for Down syndrome patients with dementia, and thereby prevent potential side effects and depletion of scarce resources, according to Dr. Gill Livingston and Dr. Andre Strydom.
The complexity of Down syndrome likely means that the amelioration of associated pathology will require a combination of treatments, rather than a single drug.
While there is optimism that the cognitive problems and neurodegeneration associated with Down's syndrome can be improved with pharmacologic intervention, researchers need a much better understanding of the neurobiology of Down syndrome to design such interventions, they said, adding that potential treatments must be identified through genetic, animal, and cellular studies, and that findings of experiments will need to be interpreted by mapping animal behavioral paradigms to human cognitive functioning.
"Sophisticated outcome measures and practical biomarkers are needed that can be used in trials for accurate measurement of changes in people with severe disability," they wrote, noting that large scale studies will require international coordination and collaboration through linked Down syndrome research networks, and that the potential effects of new treatment options should be explored thoroughly before undertaking clinical trials.
Dr. Livingston is a professor of psychiatry of older people and Dr. Strydom is a clinical senior lecturer in the department of mental health sciences at University College London (England). They wrote these comments in an editorial accompanying the trial report (Lancet 2011 Jan. 10 [doi:10.1016/S0140-6736(11)61929-6]). Dr. Livingston has been funded by Lundbeck for unrelated research in Alzheimer’s disease. Dr. Strydom is an investigator in a Roche-sponsored trial to improve cognition in adults with Down syndrome.
Though disappointing, the findings of the MEADOWS trial do serve to exclude treatments of little or no benefit for Down syndrome patients with dementia, and thereby prevent potential side effects and depletion of scarce resources, according to Dr. Gill Livingston and Dr. Andre Strydom.
The complexity of Down syndrome likely means that the amelioration of associated pathology will require a combination of treatments, rather than a single drug.
While there is optimism that the cognitive problems and neurodegeneration associated with Down's syndrome can be improved with pharmacologic intervention, researchers need a much better understanding of the neurobiology of Down syndrome to design such interventions, they said, adding that potential treatments must be identified through genetic, animal, and cellular studies, and that findings of experiments will need to be interpreted by mapping animal behavioral paradigms to human cognitive functioning.
"Sophisticated outcome measures and practical biomarkers are needed that can be used in trials for accurate measurement of changes in people with severe disability," they wrote, noting that large scale studies will require international coordination and collaboration through linked Down syndrome research networks, and that the potential effects of new treatment options should be explored thoroughly before undertaking clinical trials.
Dr. Livingston is a professor of psychiatry of older people and Dr. Strydom is a clinical senior lecturer in the department of mental health sciences at University College London (England). They wrote these comments in an editorial accompanying the trial report (Lancet 2011 Jan. 10 [doi:10.1016/S0140-6736(11)61929-6]). Dr. Livingston has been funded by Lundbeck for unrelated research in Alzheimer’s disease. Dr. Strydom is an investigator in a Roche-sponsored trial to improve cognition in adults with Down syndrome.
Though disappointing, the findings of the MEADOWS trial do serve to exclude treatments of little or no benefit for Down syndrome patients with dementia, and thereby prevent potential side effects and depletion of scarce resources, according to Dr. Gill Livingston and Dr. Andre Strydom.
The complexity of Down syndrome likely means that the amelioration of associated pathology will require a combination of treatments, rather than a single drug.
While there is optimism that the cognitive problems and neurodegeneration associated with Down's syndrome can be improved with pharmacologic intervention, researchers need a much better understanding of the neurobiology of Down syndrome to design such interventions, they said, adding that potential treatments must be identified through genetic, animal, and cellular studies, and that findings of experiments will need to be interpreted by mapping animal behavioral paradigms to human cognitive functioning.
"Sophisticated outcome measures and practical biomarkers are needed that can be used in trials for accurate measurement of changes in people with severe disability," they wrote, noting that large scale studies will require international coordination and collaboration through linked Down syndrome research networks, and that the potential effects of new treatment options should be explored thoroughly before undertaking clinical trials.
Dr. Livingston is a professor of psychiatry of older people and Dr. Strydom is a clinical senior lecturer in the department of mental health sciences at University College London (England). They wrote these comments in an editorial accompanying the trial report (Lancet 2011 Jan. 10 [doi:10.1016/S0140-6736(11)61929-6]). Dr. Livingston has been funded by Lundbeck for unrelated research in Alzheimer’s disease. Dr. Strydom is an investigator in a Roche-sponsored trial to improve cognition in adults with Down syndrome.
Memantine is not an effective treatment for dementia in adults older than 40 years with Down syndrome, according to findings from a randomized, placebo-controlled study.
In fact, all of the 173 participants in the Memantine for Dementia in Adults Older than 40 Years with Down Syndrome (MEADOWS) trial, regardless of whether they received placebo or active treatment, experienced declines in cognition and function during the 52-week study. Moreover, although the rates of decline did not differ significantly between the groups, they were slightly lower in the placebo group, senior author Dr. Clive Ballard of King’s College London (England) and his colleagues reported online in the January 10 issue of The Lancet.
The findings suggest that therapies that are effective for Alzheimer’s disease in other patient populations are not necessarily effective in those with Down syndrome.
After adjustment for baseline score, participants in the placebo group and treatment group experienced mean changes in scores on the Down syndrome attention, memory, and executive function scales (DAMES) of -1.9 and -5.6 at 52 weeks, respectively. There also was no difference in the mean changes in scores between the placebo and memantine groups in part I of the Adaptive Behavior Scale (-1.7 and -10.7, respectively), which measures independent functioning, or part II (0 and 1.0, respectively), which measures challenging behavior (Lancet 2011 Jan. 10 [doi:10.1016/S0140-6736(11)61676-0]).
The results were similar regardless of the presence of dementia, which had been diagnosed in 35% of individuals in each group at the time of randomization.
The rate of serious adverse events also did not differ significantly between the placebo and treatment groups at 7% and 11%, respectively.
The findings have implications for research strategy in development of treatments for cognitive decline and dementia in individuals with Down syndrome, the investigators said.
Dementia is very prevalent in this population, with nearly 40% of those with Down syndrome receiving a diagnosis of dementia after age 60 years. Additionally, clinically significant Alzheimer’s disease–like pathological features are "almost ubiquitous" by age 40 years in those with Down syndrome.
"Although there is therefore a rationale for the efficacy of pharmacological treatments for Alzheimer’s disease in people with Down’s syndrome, there are some key differences in pathological features that should not be overlooked. For example, people with Down’s syndrome have lifelong overproduction of amyloid-beta compounded with dysregulation of many genes, most of which are not associated with Alzheimer’s disease," the investigators explained, adding that differences in the neuropathologic features of people with Down syndrome compared with Alzheimer’s disease might affect responses to pharmacologic therapies.
The N-methyl-d-aspartate glutamate receptor antagonist memantine (Namenda) is licensed for the treatment of moderate to severe Alzheimer’s disease and showed promise in two studies in a Down syndrome model in mice. But in the current study it was found to confer no benefit, regardless of whether a formal diagnosis of dementia was available, they said.
Study participants were men and women with Down syndrome aged 40 years or older or individuals of any age with Down syndrome and an established diagnosis of dementia. They were enrolled at four learning disability centers throughout the United Kingdom and Norway. The randomization process involved a computer-generated allocation sequence that ensured balanced allocation based on the prognostic factors of sex, presence of dementia, age group, DAMES score, and center. The investigators escalated the dose of memantine over 8 weeks from 5 mg per day to the optimal therapeutic dose of 10 mg per day with fixed titration.
"The findings of this study are robust and represent an important step in the evidence base for treatment of people with Down syndrome with cognitive decline," the investigators wrote, concluding that research into specific therapies for this patient population needs to be based on an understanding of underlying pathologic processes.
In particular, an adequately powered randomized controlled trial of cholinesterase inhibitors is needed to determine if this class of drug has any role in the treatment of Down syndrome patients with dementia. Cholinesterase inhibitors are approved in the United Kingdom for people with dementia in the context of Down syndrome, but very little evidence exists to support this guidance, they said.
This study was funded by Lundbeck and sponsored by King’s College London. Dr. Ballard reported receiving consultancy and speaking fees from Lundbeck, Janssen, Novartis, and Acadia and grants from Lundbeck and Acadia. Other study authors reported receiving consultancy fees, speaking fees, honoraria, and/or grants from Lundbeck, Novartis, and/or Roche, as well as serving on the advisory board for Lundbeck, providing paid expert testimony for Novartis and Eisai, and receiving funding from the Alzheimer’s Society and the Henry Smith Trust. Detailed disclosures are available with the full text of the article at www.thelancet.com.
Memantine is not an effective treatment for dementia in adults older than 40 years with Down syndrome, according to findings from a randomized, placebo-controlled study.
In fact, all of the 173 participants in the Memantine for Dementia in Adults Older than 40 Years with Down Syndrome (MEADOWS) trial, regardless of whether they received placebo or active treatment, experienced declines in cognition and function during the 52-week study. Moreover, although the rates of decline did not differ significantly between the groups, they were slightly lower in the placebo group, senior author Dr. Clive Ballard of King’s College London (England) and his colleagues reported online in the January 10 issue of The Lancet.
The findings suggest that therapies that are effective for Alzheimer’s disease in other patient populations are not necessarily effective in those with Down syndrome.
After adjustment for baseline score, participants in the placebo group and treatment group experienced mean changes in scores on the Down syndrome attention, memory, and executive function scales (DAMES) of -1.9 and -5.6 at 52 weeks, respectively. There also was no difference in the mean changes in scores between the placebo and memantine groups in part I of the Adaptive Behavior Scale (-1.7 and -10.7, respectively), which measures independent functioning, or part II (0 and 1.0, respectively), which measures challenging behavior (Lancet 2011 Jan. 10 [doi:10.1016/S0140-6736(11)61676-0]).
The results were similar regardless of the presence of dementia, which had been diagnosed in 35% of individuals in each group at the time of randomization.
The rate of serious adverse events also did not differ significantly between the placebo and treatment groups at 7% and 11%, respectively.
The findings have implications for research strategy in development of treatments for cognitive decline and dementia in individuals with Down syndrome, the investigators said.
Dementia is very prevalent in this population, with nearly 40% of those with Down syndrome receiving a diagnosis of dementia after age 60 years. Additionally, clinically significant Alzheimer’s disease–like pathological features are "almost ubiquitous" by age 40 years in those with Down syndrome.
"Although there is therefore a rationale for the efficacy of pharmacological treatments for Alzheimer’s disease in people with Down’s syndrome, there are some key differences in pathological features that should not be overlooked. For example, people with Down’s syndrome have lifelong overproduction of amyloid-beta compounded with dysregulation of many genes, most of which are not associated with Alzheimer’s disease," the investigators explained, adding that differences in the neuropathologic features of people with Down syndrome compared with Alzheimer’s disease might affect responses to pharmacologic therapies.
The N-methyl-d-aspartate glutamate receptor antagonist memantine (Namenda) is licensed for the treatment of moderate to severe Alzheimer’s disease and showed promise in two studies in a Down syndrome model in mice. But in the current study it was found to confer no benefit, regardless of whether a formal diagnosis of dementia was available, they said.
Study participants were men and women with Down syndrome aged 40 years or older or individuals of any age with Down syndrome and an established diagnosis of dementia. They were enrolled at four learning disability centers throughout the United Kingdom and Norway. The randomization process involved a computer-generated allocation sequence that ensured balanced allocation based on the prognostic factors of sex, presence of dementia, age group, DAMES score, and center. The investigators escalated the dose of memantine over 8 weeks from 5 mg per day to the optimal therapeutic dose of 10 mg per day with fixed titration.
"The findings of this study are robust and represent an important step in the evidence base for treatment of people with Down syndrome with cognitive decline," the investigators wrote, concluding that research into specific therapies for this patient population needs to be based on an understanding of underlying pathologic processes.
In particular, an adequately powered randomized controlled trial of cholinesterase inhibitors is needed to determine if this class of drug has any role in the treatment of Down syndrome patients with dementia. Cholinesterase inhibitors are approved in the United Kingdom for people with dementia in the context of Down syndrome, but very little evidence exists to support this guidance, they said.
This study was funded by Lundbeck and sponsored by King’s College London. Dr. Ballard reported receiving consultancy and speaking fees from Lundbeck, Janssen, Novartis, and Acadia and grants from Lundbeck and Acadia. Other study authors reported receiving consultancy fees, speaking fees, honoraria, and/or grants from Lundbeck, Novartis, and/or Roche, as well as serving on the advisory board for Lundbeck, providing paid expert testimony for Novartis and Eisai, and receiving funding from the Alzheimer’s Society and the Henry Smith Trust. Detailed disclosures are available with the full text of the article at www.thelancet.com.
FROM THE LANCET
Major Finding: Participants in the placebo group and treatment group experienced statistically similar mean changes of -1.9 and -5.6, respectively, at 52 weeks in scores on the Down syndrome attention, memory, and executive function scales (DAMES); mean changes of -1.7 and -10.7, respectively, in part I of the Adaptive Behavior Scale; and mean changes of 0 and 1.0, respectively, in part II.
Data Source: A double-blind, randomized, placebo-controlled study of 173 patients with Down syndrome.
Disclosures: This study was funded by Lundbeck and sponsored by King’s College London. Dr. Ballard reported receiving consultancy and speaking fees from Lundbeck, Janssen, Novartis, and Acadia and grants from Lundbeck and Acadia. Other study authors reported receiving consultancy fees, speaking fees, honoraria, and/or grants from Lundbeck, Novartis, and/or Roche, as well as serving on the advisory board for Lundbeck, providing paid expert testimony for Novartis and Eisai, and receiving funding from the Alzheimer’s Society and the Henry Smith Trust. Detailed disclosures are available with the full text of the article at www.thelancet.com.
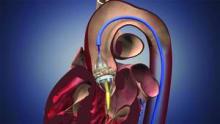
New Drug-Eluting Stents Far Safer Than Older Models
Percutaneous coronary intervention with new-generation drug-eluting stents is associated with significantly lower risk of restenosis, stent thrombosis, and death, compared with both older generation drug-eluting stents and bare-metal stents, according to findings from the Swedish Coronary Angiography and Angioplasty Registry (SCAAR).
The findings bolster the current recommendations regarding use of drug-eluting stents (DES), and are the first to show a mortality benefit with new- vs. older-generation DES. Compared with older bare-metal stents (BMS), the risk of death was reduced with older DES at 2 years, by 28%, and lower still with new DES, by 45%.
The 1- and 2-year restenosis rates among 10,551 implantations with a new-generation DES in SCAAR were 2.8% and 3.9%, respectively, compared with 4.0% and 5.8% in 19,202 implantations with an older-generation DES (cumulative adjusted hazard ratio, 0.62), and 6.3% and 7.4% in 64,631 implantations with a BMS (cumulative adjusted HR, 0.29), wrote Dr. Giovanna Sarno of Uppsala (Sweden) University and colleagues. The results are online in the Jan. 9 issue of the European Heart Journal.
The 1- and 2-year definite stent thrombosis rates among the new-generation DES implantations were 0.5% and 0.6%, respectively, compared with 0.9% and 1.3% in the older-generation DES implantations, for a risk reduction of 43%, and 1.2% and 1.4% in the bare-metal stent implantations, showing a 62% reduction in risk.
The overall mortality rate among the 61,351 patients who underwent the 94,384 stent implantations was 5.6%. Mortality was 1.9% in the new-generation DES recipients, compared with 3.4% in the older-generation DES recipients (adjusted hazard ratio, 0.77), and 6.8% in the bare-metal stent recipients (adjusted HR 0.55), the investigators said (Eur. Heart J. 2011 Jan. 9 [doi:10.1093/eurheartj/ehr479]).
"A main finding of this study is that new-generation DES are associated with a 38% lower risk of clinically relevant restenosis and a 43% lower risk of definite stent thrombosis up to 2 years, compared with older-generation DES in a large real world population," the investigators said.
Furthermore, the findings indicate that mortality with new-generation and older-generation DES is 45% and 28% lower, respectively, than with bare-metal stents, and 23% lower with new-generation DES than with older-generation DES, they said.
New-generation DES were developed with an improved design, including thinner struts and more biocompatible polymers, thought to have potential for overcoming limitations – namely those relating to long-term safety, and particularly risk of late stent thrombosis – of older generation DES. Although randomized studies demonstrated low late loss and thrombotic risk with restricted use of the new generation DES, only a few trials and reports from single-center experiences evaluating only one type of the new generation DES have looked at unrestricted use.
"Our study evaluated the performance up to 2 years of different types of new-generation DES in an unselected large real-world population – including patients with myocardial infarction, three-vessel and/or left main disease, bifurcation lesions, graft disease, restenotic lesions, and chronic total occlusions," the investigators said.
Participants in the observational cohort study included all patients in Sweden who received coronary stents from November 2006 to October 2010. In their analysis, the investigators adjusted for potential confounders, including age, sex, diabetes, hypertension, dyslipidemia, smoking status, clinical indication of the procedure, medications used at the index procedure, treated vessel and lesion type, and previous MI and revascularization.
In a press statement, study author Dr. Stefan James, also of Uppsala University, described the findings as "intriguing" and noted that, "the low rates of restenosis and stent thrombosis correspond with the results of several recent randomized trials, and this may well translate into mortality reduction in a sufficiently large study population."
However, despite the large sample size, the findings of this study should be considered within the context of the intrinsic limitations of registry data. Although the findings may be useful for the management of patients at risk for stent thrombosis and restenosis – and although they support the current strong recommendations for the use of drug-eluting stents in appropriate patients, they require confirmation by large-scale randomized studies, the investigators concluded.
New-generation DES used in this study included Endeavor Resolute (Medtronic Inc.), XienceV, Xience Prime (Abbott Laboratories), and Promus and Promus Element (Boston Scientific Corp.). Older generation DES included Cypher and Cypher Select (Cordis Corp., Miami), Taxus Express and Taxus Liberté (Boston Scientific Corp.), and Endeavor (Medtronic Inc.) Bare-metal stents include Multilink, Multilink MiniVision, and Flexmaster (Abbott Laboratories), Driver and Micro Driver coronary (Medtronic Inc.), Liberté (Boston Scientific Corp.), Braun Coroflex Blue (B.Braun Melsungen AG, Germany), and Chrono stent (CID, Saluggia, Italy).
SCAAR is sponsored by the Swedish Health Authorities. Financial support for analyses was provided by the Swedish Heart and Lung Foundation. One study author, Dr. James, disclosed that he received institutional research grants from Terumo, Medtronic, and Vascular Solutions.
Percutaneous coronary intervention with new-generation drug-eluting stents is associated with significantly lower risk of restenosis, stent thrombosis, and death, compared with both older generation drug-eluting stents and bare-metal stents, according to findings from the Swedish Coronary Angiography and Angioplasty Registry (SCAAR).
The findings bolster the current recommendations regarding use of drug-eluting stents (DES), and are the first to show a mortality benefit with new- vs. older-generation DES. Compared with older bare-metal stents (BMS), the risk of death was reduced with older DES at 2 years, by 28%, and lower still with new DES, by 45%.
The 1- and 2-year restenosis rates among 10,551 implantations with a new-generation DES in SCAAR were 2.8% and 3.9%, respectively, compared with 4.0% and 5.8% in 19,202 implantations with an older-generation DES (cumulative adjusted hazard ratio, 0.62), and 6.3% and 7.4% in 64,631 implantations with a BMS (cumulative adjusted HR, 0.29), wrote Dr. Giovanna Sarno of Uppsala (Sweden) University and colleagues. The results are online in the Jan. 9 issue of the European Heart Journal.
The 1- and 2-year definite stent thrombosis rates among the new-generation DES implantations were 0.5% and 0.6%, respectively, compared with 0.9% and 1.3% in the older-generation DES implantations, for a risk reduction of 43%, and 1.2% and 1.4% in the bare-metal stent implantations, showing a 62% reduction in risk.
The overall mortality rate among the 61,351 patients who underwent the 94,384 stent implantations was 5.6%. Mortality was 1.9% in the new-generation DES recipients, compared with 3.4% in the older-generation DES recipients (adjusted hazard ratio, 0.77), and 6.8% in the bare-metal stent recipients (adjusted HR 0.55), the investigators said (Eur. Heart J. 2011 Jan. 9 [doi:10.1093/eurheartj/ehr479]).
"A main finding of this study is that new-generation DES are associated with a 38% lower risk of clinically relevant restenosis and a 43% lower risk of definite stent thrombosis up to 2 years, compared with older-generation DES in a large real world population," the investigators said.
Furthermore, the findings indicate that mortality with new-generation and older-generation DES is 45% and 28% lower, respectively, than with bare-metal stents, and 23% lower with new-generation DES than with older-generation DES, they said.
New-generation DES were developed with an improved design, including thinner struts and more biocompatible polymers, thought to have potential for overcoming limitations – namely those relating to long-term safety, and particularly risk of late stent thrombosis – of older generation DES. Although randomized studies demonstrated low late loss and thrombotic risk with restricted use of the new generation DES, only a few trials and reports from single-center experiences evaluating only one type of the new generation DES have looked at unrestricted use.
"Our study evaluated the performance up to 2 years of different types of new-generation DES in an unselected large real-world population – including patients with myocardial infarction, three-vessel and/or left main disease, bifurcation lesions, graft disease, restenotic lesions, and chronic total occlusions," the investigators said.
Participants in the observational cohort study included all patients in Sweden who received coronary stents from November 2006 to October 2010. In their analysis, the investigators adjusted for potential confounders, including age, sex, diabetes, hypertension, dyslipidemia, smoking status, clinical indication of the procedure, medications used at the index procedure, treated vessel and lesion type, and previous MI and revascularization.
In a press statement, study author Dr. Stefan James, also of Uppsala University, described the findings as "intriguing" and noted that, "the low rates of restenosis and stent thrombosis correspond with the results of several recent randomized trials, and this may well translate into mortality reduction in a sufficiently large study population."
However, despite the large sample size, the findings of this study should be considered within the context of the intrinsic limitations of registry data. Although the findings may be useful for the management of patients at risk for stent thrombosis and restenosis – and although they support the current strong recommendations for the use of drug-eluting stents in appropriate patients, they require confirmation by large-scale randomized studies, the investigators concluded.
New-generation DES used in this study included Endeavor Resolute (Medtronic Inc.), XienceV, Xience Prime (Abbott Laboratories), and Promus and Promus Element (Boston Scientific Corp.). Older generation DES included Cypher and Cypher Select (Cordis Corp., Miami), Taxus Express and Taxus Liberté (Boston Scientific Corp.), and Endeavor (Medtronic Inc.) Bare-metal stents include Multilink, Multilink MiniVision, and Flexmaster (Abbott Laboratories), Driver and Micro Driver coronary (Medtronic Inc.), Liberté (Boston Scientific Corp.), Braun Coroflex Blue (B.Braun Melsungen AG, Germany), and Chrono stent (CID, Saluggia, Italy).
SCAAR is sponsored by the Swedish Health Authorities. Financial support for analyses was provided by the Swedish Heart and Lung Foundation. One study author, Dr. James, disclosed that he received institutional research grants from Terumo, Medtronic, and Vascular Solutions.
Percutaneous coronary intervention with new-generation drug-eluting stents is associated with significantly lower risk of restenosis, stent thrombosis, and death, compared with both older generation drug-eluting stents and bare-metal stents, according to findings from the Swedish Coronary Angiography and Angioplasty Registry (SCAAR).
The findings bolster the current recommendations regarding use of drug-eluting stents (DES), and are the first to show a mortality benefit with new- vs. older-generation DES. Compared with older bare-metal stents (BMS), the risk of death was reduced with older DES at 2 years, by 28%, and lower still with new DES, by 45%.
The 1- and 2-year restenosis rates among 10,551 implantations with a new-generation DES in SCAAR were 2.8% and 3.9%, respectively, compared with 4.0% and 5.8% in 19,202 implantations with an older-generation DES (cumulative adjusted hazard ratio, 0.62), and 6.3% and 7.4% in 64,631 implantations with a BMS (cumulative adjusted HR, 0.29), wrote Dr. Giovanna Sarno of Uppsala (Sweden) University and colleagues. The results are online in the Jan. 9 issue of the European Heart Journal.
The 1- and 2-year definite stent thrombosis rates among the new-generation DES implantations were 0.5% and 0.6%, respectively, compared with 0.9% and 1.3% in the older-generation DES implantations, for a risk reduction of 43%, and 1.2% and 1.4% in the bare-metal stent implantations, showing a 62% reduction in risk.
The overall mortality rate among the 61,351 patients who underwent the 94,384 stent implantations was 5.6%. Mortality was 1.9% in the new-generation DES recipients, compared with 3.4% in the older-generation DES recipients (adjusted hazard ratio, 0.77), and 6.8% in the bare-metal stent recipients (adjusted HR 0.55), the investigators said (Eur. Heart J. 2011 Jan. 9 [doi:10.1093/eurheartj/ehr479]).
"A main finding of this study is that new-generation DES are associated with a 38% lower risk of clinically relevant restenosis and a 43% lower risk of definite stent thrombosis up to 2 years, compared with older-generation DES in a large real world population," the investigators said.
Furthermore, the findings indicate that mortality with new-generation and older-generation DES is 45% and 28% lower, respectively, than with bare-metal stents, and 23% lower with new-generation DES than with older-generation DES, they said.
New-generation DES were developed with an improved design, including thinner struts and more biocompatible polymers, thought to have potential for overcoming limitations – namely those relating to long-term safety, and particularly risk of late stent thrombosis – of older generation DES. Although randomized studies demonstrated low late loss and thrombotic risk with restricted use of the new generation DES, only a few trials and reports from single-center experiences evaluating only one type of the new generation DES have looked at unrestricted use.
"Our study evaluated the performance up to 2 years of different types of new-generation DES in an unselected large real-world population – including patients with myocardial infarction, three-vessel and/or left main disease, bifurcation lesions, graft disease, restenotic lesions, and chronic total occlusions," the investigators said.
Participants in the observational cohort study included all patients in Sweden who received coronary stents from November 2006 to October 2010. In their analysis, the investigators adjusted for potential confounders, including age, sex, diabetes, hypertension, dyslipidemia, smoking status, clinical indication of the procedure, medications used at the index procedure, treated vessel and lesion type, and previous MI and revascularization.
In a press statement, study author Dr. Stefan James, also of Uppsala University, described the findings as "intriguing" and noted that, "the low rates of restenosis and stent thrombosis correspond with the results of several recent randomized trials, and this may well translate into mortality reduction in a sufficiently large study population."
However, despite the large sample size, the findings of this study should be considered within the context of the intrinsic limitations of registry data. Although the findings may be useful for the management of patients at risk for stent thrombosis and restenosis – and although they support the current strong recommendations for the use of drug-eluting stents in appropriate patients, they require confirmation by large-scale randomized studies, the investigators concluded.
New-generation DES used in this study included Endeavor Resolute (Medtronic Inc.), XienceV, Xience Prime (Abbott Laboratories), and Promus and Promus Element (Boston Scientific Corp.). Older generation DES included Cypher and Cypher Select (Cordis Corp., Miami), Taxus Express and Taxus Liberté (Boston Scientific Corp.), and Endeavor (Medtronic Inc.) Bare-metal stents include Multilink, Multilink MiniVision, and Flexmaster (Abbott Laboratories), Driver and Micro Driver coronary (Medtronic Inc.), Liberté (Boston Scientific Corp.), Braun Coroflex Blue (B.Braun Melsungen AG, Germany), and Chrono stent (CID, Saluggia, Italy).
SCAAR is sponsored by the Swedish Health Authorities. Financial support for analyses was provided by the Swedish Heart and Lung Foundation. One study author, Dr. James, disclosed that he received institutional research grants from Terumo, Medtronic, and Vascular Solutions.
FROM THE EUROPEAN HEART JOURNAL
Major Finding: The 1- and 2-year restenosis rates among 10,551 implantations with a new-generation DES in SCAAR were 2.8% and 3.9%, respectively, compared with 4.0% and 5.8% in 19,202 implantations with an older-generation DES (cumulative adjusted hazard ratio, 0.62), and 6.3% and 7.4% in 64,631 implantations with a bare metal stent (cumulative adjusted HR, 0.29. The 1- and 2-year definite stent thrombosis rates among the new-generation DES implantations were 0.5% and 0.6%, respectively, compared with 0.9% and 1.3% in the older-generation DES implantations (cumulative adjusted HR, 0.57), and 1.2% and 1.4% in the bare metal stent implantations (cumulative adjusted HR, 0.38).
Data Source: The Swedish Coronary Angiography and Angioplasty Registry (SCAAR).
Disclosures: SCAAR is sponsored by the Swedish Health Authorities. Financial support for analyses was provided by the Swedish Heart and Lung Foundation. One study author, Dr. James, disclosed that he received institutional research grants from Terumo, Medtronic, and Vascular Solutions.
Subclinical Shedding Persists, Despite HSV-2 Therapy
Antiviral therapy for genital herpes simplex virus type 2 is clinically effective, but even high-dose therapy did not substantially alter the frequency of viral shedding episodes in a series of three separate, but complementary, open-label crossover studies.
Suppressive therapies with greater potency are needed to prevent HSV-2 transmission and HIV-1 acquisition and transmission, Dr. Christine Johnston of the University of Washington, Seattle, and her colleagues, concluded in the Jan. 5 edition of the Lancet.
The studies – which together included 90 HSV-2-seropositive, HIV-negative adults with a median age of 43 years – evaluated within-person viral shedding in patients receiving no medication vs. standard-dose aciclovir (400 mg twice daily), those receiving high-dose aciclovir (800 mg three times daily) vs. standard-dose valaciclovir (500 mg daily), and those receiving standard-dose valaciclovir vs. high-dose valaciclovir (1 g three times daily).
Based on 23,605 swabs collected during the course of the studies, shedding frequency was reduced by all treatments, compared with no treatment.
The most significant difference occurred in the study comparing no medication vs. standard-dose acyclovir, in which 18% and 1% of the swabs indicated shedding in those groups, respectively (incidence rate ratio, 0.05), the investigators found.
However, breakthrough reactivation occurred at all drug doses. The number of episodes per person-year was highest in the no-treatment group at 28.7, and ranged from 10.0 to 22.6, with the difference reaching statistical significance only in the no-treatment vs. standard-dose aciclovir groups (28.7 vs. 10.0), the investigators said (Lancet 2011 Jan. 5 [doi:10.1016/S0140-6736(11)61750-9]).
Most of the shedding episodes during active treatment were subclinical, and the duration of episodes was shorter in the treatment groups vs. the no-treatment group (7-10 hours vs. 13 hours).
Although the available therapies for HSV have increased in use over the past 2 decades and have been successful for reducing genital lesions, treatment with daily valaciclovir has been associated with only a 48% reduction in the risk of sexual transmission of genital herpes.
"Moreover, aciclovir does not effectively reduce the risk of HIV transmission or acquisition in HSV-2 seropositive people," the investigators noted, adding that this discrepancy between potent suppression of symptoms and failure to prevent transmission has not been well understood.
Mathematical models suggest that short bursts of shedding occur frequently, and may be the cause of up to 85% of all HSV-2 reactivations, but whether treatment suppresses such short reactivation events was unknown.
To assess whether standard and/or high doses of aciclovir and valaciclovir affect these subclinical periods of HSV reactivation, they enrolled participants between November 2006 and July 2010 into the three studies, each with a 4- to 7-week initial treatment period, followed by a 1-week washout period, followed by crossover to the other treatment for an additional 4-7 weeks. Swabs were collected four times daily.
The "data suggest that anti-HSV therapy, although clinically effective, does not substantially alter the underlying pathobiology of frequent, subclinical HSV-2 reactivation. That we could not eliminate or even alter the frequency of shedding episodes with high-dose valaciclovir suggests that the maximum benefit of shedding reduction has probably been reached for currently available antiviral drugs," the investigators said.
The study was funded by the National Institutes of Health. Valaciclovir was provided for free for the third trial by GlaxoSmithKline. Dr. Johnston disclosed that she is a research investigator for AiCuris and GmbH. Two other study authors disclosed that they are consultants for those companies. These and other authors also disclosed financial relationships with Immune Design Corp., Sanofi-Pasteur, Agenus, Coridon, Vical, and/or PATH. Additionally Dr. Lawrence Corey and Dr. David M. Koelle are coinventors in several patents describing antigens and epitopes to which T-cell responses to HSV-2 are directed.
The finding that treatment does not fully prevent HSV-2 transmission underscores the need for patients to use condoms and adopt safe sex practices, and it also prompts reconsideration of "the nature of latency and reactivation of chronic HSV," Dr. Philippe Van de Perre and Dr. Nicolas Nagot said in an accompanying editorial.
Not only does low-level viral replication (which is not easily suppressed by therapy) increase the risk of transmission and deserve further attention, but the complex interactions between herpes viruses and HIV – and the conflicting results from trials evaluating interventions to reduce HIV transmission via HSV suppression – should be revisited, they argued (Lancet 2011 Jan. 5 [doi:10.1016/S0140-6736(11)61614-0]).
"What should be done to address the ineffectiveness of antiherpetic therapies for suppression of viral replication? The development of new antiherpetic drugs such as helicase-primase inhibitors is important," they said.
But even if they could stop HSV shedding, the value of these drugs would likely be hampered by a lack of the coverage and long-term adherence needed to adequately affect HSV dynamics, given that 20% of the general population is HSV-2 positive and many have no need for antiherpetic therapy.
Additionally, although "alternative control tools" (including therapeutic vaccines) are in development, adequate animal models for testing are lacking, as is a commitment from pharmaceutical companies and the public sector, they noted.
Dr. Van de Perre and Dr. Nagot are with Université Montpellier (France) and Institut National de la Santé et de la Recherche Médicale (INSERM) Unité 1058, also in Montpellier. They reported having no relevant conflicts of interest.
The finding that treatment does not fully prevent HSV-2 transmission underscores the need for patients to use condoms and adopt safe sex practices, and it also prompts reconsideration of "the nature of latency and reactivation of chronic HSV," Dr. Philippe Van de Perre and Dr. Nicolas Nagot said in an accompanying editorial.
Not only does low-level viral replication (which is not easily suppressed by therapy) increase the risk of transmission and deserve further attention, but the complex interactions between herpes viruses and HIV – and the conflicting results from trials evaluating interventions to reduce HIV transmission via HSV suppression – should be revisited, they argued (Lancet 2011 Jan. 5 [doi:10.1016/S0140-6736(11)61614-0]).
"What should be done to address the ineffectiveness of antiherpetic therapies for suppression of viral replication? The development of new antiherpetic drugs such as helicase-primase inhibitors is important," they said.
But even if they could stop HSV shedding, the value of these drugs would likely be hampered by a lack of the coverage and long-term adherence needed to adequately affect HSV dynamics, given that 20% of the general population is HSV-2 positive and many have no need for antiherpetic therapy.
Additionally, although "alternative control tools" (including therapeutic vaccines) are in development, adequate animal models for testing are lacking, as is a commitment from pharmaceutical companies and the public sector, they noted.
Dr. Van de Perre and Dr. Nagot are with Université Montpellier (France) and Institut National de la Santé et de la Recherche Médicale (INSERM) Unité 1058, also in Montpellier. They reported having no relevant conflicts of interest.
The finding that treatment does not fully prevent HSV-2 transmission underscores the need for patients to use condoms and adopt safe sex practices, and it also prompts reconsideration of "the nature of latency and reactivation of chronic HSV," Dr. Philippe Van de Perre and Dr. Nicolas Nagot said in an accompanying editorial.
Not only does low-level viral replication (which is not easily suppressed by therapy) increase the risk of transmission and deserve further attention, but the complex interactions between herpes viruses and HIV – and the conflicting results from trials evaluating interventions to reduce HIV transmission via HSV suppression – should be revisited, they argued (Lancet 2011 Jan. 5 [doi:10.1016/S0140-6736(11)61614-0]).
"What should be done to address the ineffectiveness of antiherpetic therapies for suppression of viral replication? The development of new antiherpetic drugs such as helicase-primase inhibitors is important," they said.
But even if they could stop HSV shedding, the value of these drugs would likely be hampered by a lack of the coverage and long-term adherence needed to adequately affect HSV dynamics, given that 20% of the general population is HSV-2 positive and many have no need for antiherpetic therapy.
Additionally, although "alternative control tools" (including therapeutic vaccines) are in development, adequate animal models for testing are lacking, as is a commitment from pharmaceutical companies and the public sector, they noted.
Dr. Van de Perre and Dr. Nagot are with Université Montpellier (France) and Institut National de la Santé et de la Recherche Médicale (INSERM) Unité 1058, also in Montpellier. They reported having no relevant conflicts of interest.
Antiviral therapy for genital herpes simplex virus type 2 is clinically effective, but even high-dose therapy did not substantially alter the frequency of viral shedding episodes in a series of three separate, but complementary, open-label crossover studies.
Suppressive therapies with greater potency are needed to prevent HSV-2 transmission and HIV-1 acquisition and transmission, Dr. Christine Johnston of the University of Washington, Seattle, and her colleagues, concluded in the Jan. 5 edition of the Lancet.
The studies – which together included 90 HSV-2-seropositive, HIV-negative adults with a median age of 43 years – evaluated within-person viral shedding in patients receiving no medication vs. standard-dose aciclovir (400 mg twice daily), those receiving high-dose aciclovir (800 mg three times daily) vs. standard-dose valaciclovir (500 mg daily), and those receiving standard-dose valaciclovir vs. high-dose valaciclovir (1 g three times daily).
Based on 23,605 swabs collected during the course of the studies, shedding frequency was reduced by all treatments, compared with no treatment.
The most significant difference occurred in the study comparing no medication vs. standard-dose acyclovir, in which 18% and 1% of the swabs indicated shedding in those groups, respectively (incidence rate ratio, 0.05), the investigators found.
However, breakthrough reactivation occurred at all drug doses. The number of episodes per person-year was highest in the no-treatment group at 28.7, and ranged from 10.0 to 22.6, with the difference reaching statistical significance only in the no-treatment vs. standard-dose aciclovir groups (28.7 vs. 10.0), the investigators said (Lancet 2011 Jan. 5 [doi:10.1016/S0140-6736(11)61750-9]).
Most of the shedding episodes during active treatment were subclinical, and the duration of episodes was shorter in the treatment groups vs. the no-treatment group (7-10 hours vs. 13 hours).
Although the available therapies for HSV have increased in use over the past 2 decades and have been successful for reducing genital lesions, treatment with daily valaciclovir has been associated with only a 48% reduction in the risk of sexual transmission of genital herpes.
"Moreover, aciclovir does not effectively reduce the risk of HIV transmission or acquisition in HSV-2 seropositive people," the investigators noted, adding that this discrepancy between potent suppression of symptoms and failure to prevent transmission has not been well understood.
Mathematical models suggest that short bursts of shedding occur frequently, and may be the cause of up to 85% of all HSV-2 reactivations, but whether treatment suppresses such short reactivation events was unknown.
To assess whether standard and/or high doses of aciclovir and valaciclovir affect these subclinical periods of HSV reactivation, they enrolled participants between November 2006 and July 2010 into the three studies, each with a 4- to 7-week initial treatment period, followed by a 1-week washout period, followed by crossover to the other treatment for an additional 4-7 weeks. Swabs were collected four times daily.
The "data suggest that anti-HSV therapy, although clinically effective, does not substantially alter the underlying pathobiology of frequent, subclinical HSV-2 reactivation. That we could not eliminate or even alter the frequency of shedding episodes with high-dose valaciclovir suggests that the maximum benefit of shedding reduction has probably been reached for currently available antiviral drugs," the investigators said.
The study was funded by the National Institutes of Health. Valaciclovir was provided for free for the third trial by GlaxoSmithKline. Dr. Johnston disclosed that she is a research investigator for AiCuris and GmbH. Two other study authors disclosed that they are consultants for those companies. These and other authors also disclosed financial relationships with Immune Design Corp., Sanofi-Pasteur, Agenus, Coridon, Vical, and/or PATH. Additionally Dr. Lawrence Corey and Dr. David M. Koelle are coinventors in several patents describing antigens and epitopes to which T-cell responses to HSV-2 are directed.
Antiviral therapy for genital herpes simplex virus type 2 is clinically effective, but even high-dose therapy did not substantially alter the frequency of viral shedding episodes in a series of three separate, but complementary, open-label crossover studies.
Suppressive therapies with greater potency are needed to prevent HSV-2 transmission and HIV-1 acquisition and transmission, Dr. Christine Johnston of the University of Washington, Seattle, and her colleagues, concluded in the Jan. 5 edition of the Lancet.
The studies – which together included 90 HSV-2-seropositive, HIV-negative adults with a median age of 43 years – evaluated within-person viral shedding in patients receiving no medication vs. standard-dose aciclovir (400 mg twice daily), those receiving high-dose aciclovir (800 mg three times daily) vs. standard-dose valaciclovir (500 mg daily), and those receiving standard-dose valaciclovir vs. high-dose valaciclovir (1 g three times daily).
Based on 23,605 swabs collected during the course of the studies, shedding frequency was reduced by all treatments, compared with no treatment.
The most significant difference occurred in the study comparing no medication vs. standard-dose acyclovir, in which 18% and 1% of the swabs indicated shedding in those groups, respectively (incidence rate ratio, 0.05), the investigators found.
However, breakthrough reactivation occurred at all drug doses. The number of episodes per person-year was highest in the no-treatment group at 28.7, and ranged from 10.0 to 22.6, with the difference reaching statistical significance only in the no-treatment vs. standard-dose aciclovir groups (28.7 vs. 10.0), the investigators said (Lancet 2011 Jan. 5 [doi:10.1016/S0140-6736(11)61750-9]).
Most of the shedding episodes during active treatment were subclinical, and the duration of episodes was shorter in the treatment groups vs. the no-treatment group (7-10 hours vs. 13 hours).
Although the available therapies for HSV have increased in use over the past 2 decades and have been successful for reducing genital lesions, treatment with daily valaciclovir has been associated with only a 48% reduction in the risk of sexual transmission of genital herpes.
"Moreover, aciclovir does not effectively reduce the risk of HIV transmission or acquisition in HSV-2 seropositive people," the investigators noted, adding that this discrepancy between potent suppression of symptoms and failure to prevent transmission has not been well understood.
Mathematical models suggest that short bursts of shedding occur frequently, and may be the cause of up to 85% of all HSV-2 reactivations, but whether treatment suppresses such short reactivation events was unknown.
To assess whether standard and/or high doses of aciclovir and valaciclovir affect these subclinical periods of HSV reactivation, they enrolled participants between November 2006 and July 2010 into the three studies, each with a 4- to 7-week initial treatment period, followed by a 1-week washout period, followed by crossover to the other treatment for an additional 4-7 weeks. Swabs were collected four times daily.
The "data suggest that anti-HSV therapy, although clinically effective, does not substantially alter the underlying pathobiology of frequent, subclinical HSV-2 reactivation. That we could not eliminate or even alter the frequency of shedding episodes with high-dose valaciclovir suggests that the maximum benefit of shedding reduction has probably been reached for currently available antiviral drugs," the investigators said.
The study was funded by the National Institutes of Health. Valaciclovir was provided for free for the third trial by GlaxoSmithKline. Dr. Johnston disclosed that she is a research investigator for AiCuris and GmbH. Two other study authors disclosed that they are consultants for those companies. These and other authors also disclosed financial relationships with Immune Design Corp., Sanofi-Pasteur, Agenus, Coridon, Vical, and/or PATH. Additionally Dr. Lawrence Corey and Dr. David M. Koelle are coinventors in several patents describing antigens and epitopes to which T-cell responses to HSV-2 are directed.
FROM THE LANCET
Major Finding: Based on 23,605 swabs collected during the course of the studies, viral shedding frequency was reduced by all treatments, compared with no treatment. However, breakthrough reactivation occurred at all drug doses. The number of episodes per person-year was highest in the no-treatment group at 28.7, but ranged from 10.0 to 22.6 in the treatment groups.
Data Source: Three separate but complementary open-label, crossover studies.
Disclosures: The study was funded by the National Institutes of Health. Valaciclovir was provided for free for the third trial by GlaxoSmithKline. Dr. Johnston disclosed that she is a research investigator for AiCuris and GmbH. Two other study authors disclosed that they are consultants for those companies. These and other authors also disclosed financial relationships with Immune Design Corp., Sanofi-Pasteur, Agenus, Coridon, Vical, and/or PATH. Additionally Dr. Lawrence Corey and Dr. David M. Koelle are coinventors in several patents describing antigens and epitopes to which T-cell responses to HSV-2 are directed.
Adiponectin Predicts Dementia, Alzheimer's in Women
Increased plasma adiponectin is an independent risk factor for all-cause dementia and Alzheimer’s disease in women, according to findings from the Framingham Heart Study.
In 840 dementia-free subjects from the prospective cohort study who had sera levels of biomarkers for glucose homeostasis and inflammation that were measured at a biennial examination between 1985 and 1988, and who were followed for a median of 13 years, 159 developed dementia, including 125 with Alzheimer’s disease (AD).
Adiponectin, a hormone derived from visceral fat, was the only biomarker tested to be linked with the development of all-cause dementia (hazard ratio, 1.29 per 1 standard deviation increase in adiponectin level) or AD (HR, 1.33 per 1 SD increase) after adjustment for age, education, body mass index, recent weight change, apolipoprotein E epsilon 4 allele status, and plasma docosahexaenoic acid levels, Thomas M. van Himbergen, Ph.D., of Tufts University, Boston, and his colleagues reported online in the Jan. 2 issue of Archives of Neurology.
Those with baseline adiponectin levels greater than the sex-specific median had a greater risk of both dementia and AD, compared with those with baseline values less than the median (HR, 1.63 and 1.87, respectively), the investigators found (Arch. Neurol. 2012 Jan. 2 [doi:10.1001/archneurol.2011.670]).
"We hypothesized that adiponectin contributes to or serves as a risk marker for the development of all-cause dementia and AD."
Other biomarkers of glucose homeostasis that were evaluated in this study were glucose, glycated albumin, and insulin levels. Biomarkers of inflammation that were evaluated included hsCRP (high-sensitivity C-reactive protein) and Lp-PLA2 (lipoprotein-associated phospholipase A2) levels. Because data suggest that the management of cardiovascular risk factors (such as type 2 diabetes, high blood pressure, and obesity) might reduce cognitive decline, the investigators hypothesized that factors that underlie type 2 diabetes, such as insulin signaling and inflammation, also might contribute to the development of all-cause dementia and AD.
"Furthermore, based on the role adiponectin plays in insulin signaling and the presence of adiponectin receptors in neurological tissue, we hypothesized that adiponectin contributes to or serves as a risk marker for the development of all-cause dementia and AD," the investigators wrote.
They found no indications that plasma insulin, glucose, glycated albumin, and levels of Lp-PLA2 were associated with either. Although higher plasma levels of hsCRP initially appeared to be associated with a reduced risk for Alzheimer’s and dementia, the association was no longer significant in the fully adjusted model.
Although the finding of an association between elevated adiponectin and an increased risk of dementia and AD in women supported the investigators’ hypothesis, it also raises questions.
"It is well established that insulin signaling is dysfunctional in the brains of patients with AD, and since adiponectin enhances insulin sensitivity, one would also expect beneficial actions protecting against cognitive decline," they wrote.
It is possible that adiponectin levels in the study participants might have risen as a protective response to vascular damage or unidentified changes in brain morphology; thus, studies that evaluate the correlation between adiponectin and cognitive decline over time – as well as mendelian randomization studies linking genetic variation in the adiponectin gene to plasma adiponectin levels and cognitive decline – will be of particular interest, the investigators said, noting that the results of the current study require confirmation in other samples.
In addition, Dr. van Himbergen and his colleagues reported, one limitation of the study is the "predominantly white nature" of the study sample. "Hence, our results require verification in other racial and ethnic samples," they wrote.
Funding for this study was provided by multiple sources, including Mahidol University in Bangkok, Thailand; the U.S. National Institutes of Health; the U.S. Department of Agriculture; the U.S. National Heart, Lung, and Blood Institute; the U.S. National Institute of Neurological Disorders and Stroke; and the U.S. National Institute on Aging. Dr. van Himbergen was supported by a research grant from the Unilever Food and Health Research Institute in the Netherlands.
Increased plasma adiponectin is an independent risk factor for all-cause dementia and Alzheimer’s disease in women, according to findings from the Framingham Heart Study.
In 840 dementia-free subjects from the prospective cohort study who had sera levels of biomarkers for glucose homeostasis and inflammation that were measured at a biennial examination between 1985 and 1988, and who were followed for a median of 13 years, 159 developed dementia, including 125 with Alzheimer’s disease (AD).
Adiponectin, a hormone derived from visceral fat, was the only biomarker tested to be linked with the development of all-cause dementia (hazard ratio, 1.29 per 1 standard deviation increase in adiponectin level) or AD (HR, 1.33 per 1 SD increase) after adjustment for age, education, body mass index, recent weight change, apolipoprotein E epsilon 4 allele status, and plasma docosahexaenoic acid levels, Thomas M. van Himbergen, Ph.D., of Tufts University, Boston, and his colleagues reported online in the Jan. 2 issue of Archives of Neurology.
Those with baseline adiponectin levels greater than the sex-specific median had a greater risk of both dementia and AD, compared with those with baseline values less than the median (HR, 1.63 and 1.87, respectively), the investigators found (Arch. Neurol. 2012 Jan. 2 [doi:10.1001/archneurol.2011.670]).
"We hypothesized that adiponectin contributes to or serves as a risk marker for the development of all-cause dementia and AD."
Other biomarkers of glucose homeostasis that were evaluated in this study were glucose, glycated albumin, and insulin levels. Biomarkers of inflammation that were evaluated included hsCRP (high-sensitivity C-reactive protein) and Lp-PLA2 (lipoprotein-associated phospholipase A2) levels. Because data suggest that the management of cardiovascular risk factors (such as type 2 diabetes, high blood pressure, and obesity) might reduce cognitive decline, the investigators hypothesized that factors that underlie type 2 diabetes, such as insulin signaling and inflammation, also might contribute to the development of all-cause dementia and AD.
"Furthermore, based on the role adiponectin plays in insulin signaling and the presence of adiponectin receptors in neurological tissue, we hypothesized that adiponectin contributes to or serves as a risk marker for the development of all-cause dementia and AD," the investigators wrote.
They found no indications that plasma insulin, glucose, glycated albumin, and levels of Lp-PLA2 were associated with either. Although higher plasma levels of hsCRP initially appeared to be associated with a reduced risk for Alzheimer’s and dementia, the association was no longer significant in the fully adjusted model.
Although the finding of an association between elevated adiponectin and an increased risk of dementia and AD in women supported the investigators’ hypothesis, it also raises questions.
"It is well established that insulin signaling is dysfunctional in the brains of patients with AD, and since adiponectin enhances insulin sensitivity, one would also expect beneficial actions protecting against cognitive decline," they wrote.
It is possible that adiponectin levels in the study participants might have risen as a protective response to vascular damage or unidentified changes in brain morphology; thus, studies that evaluate the correlation between adiponectin and cognitive decline over time – as well as mendelian randomization studies linking genetic variation in the adiponectin gene to plasma adiponectin levels and cognitive decline – will be of particular interest, the investigators said, noting that the results of the current study require confirmation in other samples.
In addition, Dr. van Himbergen and his colleagues reported, one limitation of the study is the "predominantly white nature" of the study sample. "Hence, our results require verification in other racial and ethnic samples," they wrote.
Funding for this study was provided by multiple sources, including Mahidol University in Bangkok, Thailand; the U.S. National Institutes of Health; the U.S. Department of Agriculture; the U.S. National Heart, Lung, and Blood Institute; the U.S. National Institute of Neurological Disorders and Stroke; and the U.S. National Institute on Aging. Dr. van Himbergen was supported by a research grant from the Unilever Food and Health Research Institute in the Netherlands.
Increased plasma adiponectin is an independent risk factor for all-cause dementia and Alzheimer’s disease in women, according to findings from the Framingham Heart Study.
In 840 dementia-free subjects from the prospective cohort study who had sera levels of biomarkers for glucose homeostasis and inflammation that were measured at a biennial examination between 1985 and 1988, and who were followed for a median of 13 years, 159 developed dementia, including 125 with Alzheimer’s disease (AD).
Adiponectin, a hormone derived from visceral fat, was the only biomarker tested to be linked with the development of all-cause dementia (hazard ratio, 1.29 per 1 standard deviation increase in adiponectin level) or AD (HR, 1.33 per 1 SD increase) after adjustment for age, education, body mass index, recent weight change, apolipoprotein E epsilon 4 allele status, and plasma docosahexaenoic acid levels, Thomas M. van Himbergen, Ph.D., of Tufts University, Boston, and his colleagues reported online in the Jan. 2 issue of Archives of Neurology.
Those with baseline adiponectin levels greater than the sex-specific median had a greater risk of both dementia and AD, compared with those with baseline values less than the median (HR, 1.63 and 1.87, respectively), the investigators found (Arch. Neurol. 2012 Jan. 2 [doi:10.1001/archneurol.2011.670]).
"We hypothesized that adiponectin contributes to or serves as a risk marker for the development of all-cause dementia and AD."
Other biomarkers of glucose homeostasis that were evaluated in this study were glucose, glycated albumin, and insulin levels. Biomarkers of inflammation that were evaluated included hsCRP (high-sensitivity C-reactive protein) and Lp-PLA2 (lipoprotein-associated phospholipase A2) levels. Because data suggest that the management of cardiovascular risk factors (such as type 2 diabetes, high blood pressure, and obesity) might reduce cognitive decline, the investigators hypothesized that factors that underlie type 2 diabetes, such as insulin signaling and inflammation, also might contribute to the development of all-cause dementia and AD.
"Furthermore, based on the role adiponectin plays in insulin signaling and the presence of adiponectin receptors in neurological tissue, we hypothesized that adiponectin contributes to or serves as a risk marker for the development of all-cause dementia and AD," the investigators wrote.
They found no indications that plasma insulin, glucose, glycated albumin, and levels of Lp-PLA2 were associated with either. Although higher plasma levels of hsCRP initially appeared to be associated with a reduced risk for Alzheimer’s and dementia, the association was no longer significant in the fully adjusted model.
Although the finding of an association between elevated adiponectin and an increased risk of dementia and AD in women supported the investigators’ hypothesis, it also raises questions.
"It is well established that insulin signaling is dysfunctional in the brains of patients with AD, and since adiponectin enhances insulin sensitivity, one would also expect beneficial actions protecting against cognitive decline," they wrote.
It is possible that adiponectin levels in the study participants might have risen as a protective response to vascular damage or unidentified changes in brain morphology; thus, studies that evaluate the correlation between adiponectin and cognitive decline over time – as well as mendelian randomization studies linking genetic variation in the adiponectin gene to plasma adiponectin levels and cognitive decline – will be of particular interest, the investigators said, noting that the results of the current study require confirmation in other samples.
In addition, Dr. van Himbergen and his colleagues reported, one limitation of the study is the "predominantly white nature" of the study sample. "Hence, our results require verification in other racial and ethnic samples," they wrote.
Funding for this study was provided by multiple sources, including Mahidol University in Bangkok, Thailand; the U.S. National Institutes of Health; the U.S. Department of Agriculture; the U.S. National Heart, Lung, and Blood Institute; the U.S. National Institute of Neurological Disorders and Stroke; and the U.S. National Institute on Aging. Dr. van Himbergen was supported by a research grant from the Unilever Food and Health Research Institute in the Netherlands.
FROM ARCHIVES OF NEUROLOGY
Major Finding: Adiponectin was the only biomarker tested to be linked with development of all-cause dementia (HR, 1.29 per 1 SD increase in adiponectin level) or Alzheimer’s disease (HR, 1.33 per 1 SD increase) after adjustment for age, education, BMI, recent weight change, apo E epsilon 4 allele status, and plasma docosahexaenoic acid levels.
Data Source: The Framingham Heart Study, a prospective cohort study.
Disclosures: Funding for this study was provided by multiple sources, including Mahidol University in Bangkok, Thailand; the U.S. National Institutes of Health; the U.S. Department of Agriculture; the U.S. National Heart, Lung, and Blood Institute; U.S. the National Institute of Neurological Disorders and Stroke; and the U.S. National Institute on Aging. Dr. van Himbergen was supported by a research grant from the Unilever Food and Health Research Institute in the Netherlands.
Greater Loss of Gray Matter Volume Seen in Early Schizophrenia
Progressive losses in gray matter volume and increases in cerebrospinal fluid in the 2 years after a first episode of psychosis in children and adolescents were greater in those eventually diagnosed with schizophrenia or "other" psychoses – but not in those diagnosed with bipolar disorder – compared with healthy controls, results from a prospective, multicenter, 2-year follow-up study show.
Furthermore, the changes in those with schizophrenia were related to markers of poorer prognosis, Dr. Celso Arango and colleagues reported in the January issue of the Archives of General Psychiatry.
The results come from the Child and Adolescent First Episode Psychosis Study (CAFEPS). Participants were 110 patients with an initial episode of psychosis of less than 6 months’ duration and 98 healthy controls who were enrolled at six psychiatric units throughout Spain. Patients and controls were matched for age (mean, 15.5 years and 15.3 years, respectively), sex, and parental socioeconomic status. MRI was performed at baseline and at the 2-year follow-up in 25 patients with schizophrenia, 16 with bipolar disorder, and 20 with "other" psychosis, as well as in 70 healthy controls.
The investigators found that schizophrenia patients, compared with controls, had significantly greater mean losses of whole brain gray matter (–37.1 vs. –14.5 cm3) and greater mean increases of left frontal cerebrospinal fluid volumes (6.7 vs. 2.4 cm3), as well as significantly greater mean losses of gray matter volume in the frontal lobe (–3.3 vs. –0.6 cm3) and in the left parietal lobe (–4.3 vs. –2.2 cm3), reported Dr. Arango of the department of child and adolescent psychiatry at the Hospital General Universitario Gregorio Marañón in Madrid, a member of CIBERSAM (Centro de Investigación Biomédica en Red de Salud Mental).
Those with "other" psychoses had significantly greater losses in whole brain gray matter volume (–37.6 vs. –14.5 cm3) and significantly greater loss of gray matter in the frontal lobes (–4.3 vs. –0.6 cm3 for the left, and –4.2 vs. –0.8 cm3 for the right) and left parietal lobes (–5.3 vs. –2.2 cm3), and significantly greater increases in cerebrospinal fluid volume in the frontal lobes (7.1 vs. 2.4 cm3 for the left, and 3.2 vs. 0.8 cm3 for the right), the investigators said (Arch. Gen. Psychiatry 2012;69:16-26).
"Progressive brain changes seem to be more marked in those patients ending with a diagnosis of schizophrenia."
No significant differences were seen in longitudinal volume changes in the bipolar disorder patients, compared with the healthy controls, although the differences that did occur were in the same direction as those that occurred in the other patient subgroups, the investigators noted.
As for associations between volume changes and clinical and outcomes variables, the investigators also noted that "significant correlations were observed between left frontal [gray matter] volume change during follow-up and number of weeks hospitalized and right frontal [cerebrospinal fluid] change, with more weeks hospitalized correlating with greater [gray matter] volume loss and CSF increase in patients with schizophrenia."
A significant association between left frontal CSF volume change during follow-up and left parietal CSF change with mean negative PANSS (Positive and Negative Syndrome Scale) score in patients with schizophrenia also was observed, and left frontal CSF volume change correlated with the mean PANSS total score.
"With regard to clinical improvement, significant correlations were found for the schizophrenia group between the PANSS negative subscale score and PANSS general change score and left temporal [gray matter] loss, with less improvement correlating with larger reductions.
"Associations were also found between frontal right CSF and PANSS general change score and parietal right CSF and PANSS general change score, suggesting that greater CSF volume increase is related to less improvement in general symptoms as measured with PANSS in schizophrenic patients," they wrote.
In the "other" psychoses group, correlations were observed between general and positive and total PANSS change scores, and left parietal CSF loss, suggesting that decreases in CSF volume are associated with less improvement on total score and the presence of positive psychotic symptoms during follow-up.
The findings – which support those from a previous smaller study that also demonstrated that schizophrenia patients with a first episode of psychosis had significant frontal gray matter reductions, compared with healthy controls – suggest a continuum between early-onset and adult-onset psychotic disorders, the investigators said.
Although the underlying mechanisms for brain volume changes in schizophrenia and other psychotic disorder are not yet understood, the findings of this study suggest that one or more active pathophysiologic processes occur in the brains of children and adolescents after a first psychotic episode – particularly in those with schizophrenia, they noted.
"Findings from cross-sectional and longitudinal studies examining patients with adolescent-onset psychosis support the concept of [early onset psychosis] as a progressive neurodevelopmental disorder with both early and later neurodevelopmental abnormalities," the investigators wrote. "Progressive brain changes seem to be more marked in those patients ending with a diagnosis of schizophrenia, although progressive changes were present in other psychoses. Some of these pathophysiologic processes seem to be markers of poorer prognosis." They added that the development of therapeutic strategies aimed at counteracting these pathological progressive brain changes requires research focused on their neurobiological underpinnings.
"The correlates of volume changes at a cellular level and the study of risk genes involved in circuitries associated with different psychoses and their relationship to developmental trajectories may be promising areas of research," they noted.
The study was supported by the Spanish Ministry of Health and Social Policy, Spanish Ministry of Science and Innovation. Dr. Arango has been a consultant to or has received honoraria from AstraZeneca, Bristol-Myers Squibb, Janssen-Cilag, Lundbeck, Otsuka, Pfizer, Roche, Servier, and Schering-Plough.
Progressive losses in gray matter volume and increases in cerebrospinal fluid in the 2 years after a first episode of psychosis in children and adolescents were greater in those eventually diagnosed with schizophrenia or "other" psychoses – but not in those diagnosed with bipolar disorder – compared with healthy controls, results from a prospective, multicenter, 2-year follow-up study show.
Furthermore, the changes in those with schizophrenia were related to markers of poorer prognosis, Dr. Celso Arango and colleagues reported in the January issue of the Archives of General Psychiatry.
The results come from the Child and Adolescent First Episode Psychosis Study (CAFEPS). Participants were 110 patients with an initial episode of psychosis of less than 6 months’ duration and 98 healthy controls who were enrolled at six psychiatric units throughout Spain. Patients and controls were matched for age (mean, 15.5 years and 15.3 years, respectively), sex, and parental socioeconomic status. MRI was performed at baseline and at the 2-year follow-up in 25 patients with schizophrenia, 16 with bipolar disorder, and 20 with "other" psychosis, as well as in 70 healthy controls.
The investigators found that schizophrenia patients, compared with controls, had significantly greater mean losses of whole brain gray matter (–37.1 vs. –14.5 cm3) and greater mean increases of left frontal cerebrospinal fluid volumes (6.7 vs. 2.4 cm3), as well as significantly greater mean losses of gray matter volume in the frontal lobe (–3.3 vs. –0.6 cm3) and in the left parietal lobe (–4.3 vs. –2.2 cm3), reported Dr. Arango of the department of child and adolescent psychiatry at the Hospital General Universitario Gregorio Marañón in Madrid, a member of CIBERSAM (Centro de Investigación Biomédica en Red de Salud Mental).
Those with "other" psychoses had significantly greater losses in whole brain gray matter volume (–37.6 vs. –14.5 cm3) and significantly greater loss of gray matter in the frontal lobes (–4.3 vs. –0.6 cm3 for the left, and –4.2 vs. –0.8 cm3 for the right) and left parietal lobes (–5.3 vs. –2.2 cm3), and significantly greater increases in cerebrospinal fluid volume in the frontal lobes (7.1 vs. 2.4 cm3 for the left, and 3.2 vs. 0.8 cm3 for the right), the investigators said (Arch. Gen. Psychiatry 2012;69:16-26).
"Progressive brain changes seem to be more marked in those patients ending with a diagnosis of schizophrenia."
No significant differences were seen in longitudinal volume changes in the bipolar disorder patients, compared with the healthy controls, although the differences that did occur were in the same direction as those that occurred in the other patient subgroups, the investigators noted.
As for associations between volume changes and clinical and outcomes variables, the investigators also noted that "significant correlations were observed between left frontal [gray matter] volume change during follow-up and number of weeks hospitalized and right frontal [cerebrospinal fluid] change, with more weeks hospitalized correlating with greater [gray matter] volume loss and CSF increase in patients with schizophrenia."
A significant association between left frontal CSF volume change during follow-up and left parietal CSF change with mean negative PANSS (Positive and Negative Syndrome Scale) score in patients with schizophrenia also was observed, and left frontal CSF volume change correlated with the mean PANSS total score.
"With regard to clinical improvement, significant correlations were found for the schizophrenia group between the PANSS negative subscale score and PANSS general change score and left temporal [gray matter] loss, with less improvement correlating with larger reductions.
"Associations were also found between frontal right CSF and PANSS general change score and parietal right CSF and PANSS general change score, suggesting that greater CSF volume increase is related to less improvement in general symptoms as measured with PANSS in schizophrenic patients," they wrote.
In the "other" psychoses group, correlations were observed between general and positive and total PANSS change scores, and left parietal CSF loss, suggesting that decreases in CSF volume are associated with less improvement on total score and the presence of positive psychotic symptoms during follow-up.
The findings – which support those from a previous smaller study that also demonstrated that schizophrenia patients with a first episode of psychosis had significant frontal gray matter reductions, compared with healthy controls – suggest a continuum between early-onset and adult-onset psychotic disorders, the investigators said.
Although the underlying mechanisms for brain volume changes in schizophrenia and other psychotic disorder are not yet understood, the findings of this study suggest that one or more active pathophysiologic processes occur in the brains of children and adolescents after a first psychotic episode – particularly in those with schizophrenia, they noted.
"Findings from cross-sectional and longitudinal studies examining patients with adolescent-onset psychosis support the concept of [early onset psychosis] as a progressive neurodevelopmental disorder with both early and later neurodevelopmental abnormalities," the investigators wrote. "Progressive brain changes seem to be more marked in those patients ending with a diagnosis of schizophrenia, although progressive changes were present in other psychoses. Some of these pathophysiologic processes seem to be markers of poorer prognosis." They added that the development of therapeutic strategies aimed at counteracting these pathological progressive brain changes requires research focused on their neurobiological underpinnings.
"The correlates of volume changes at a cellular level and the study of risk genes involved in circuitries associated with different psychoses and their relationship to developmental trajectories may be promising areas of research," they noted.
The study was supported by the Spanish Ministry of Health and Social Policy, Spanish Ministry of Science and Innovation. Dr. Arango has been a consultant to or has received honoraria from AstraZeneca, Bristol-Myers Squibb, Janssen-Cilag, Lundbeck, Otsuka, Pfizer, Roche, Servier, and Schering-Plough.
Progressive losses in gray matter volume and increases in cerebrospinal fluid in the 2 years after a first episode of psychosis in children and adolescents were greater in those eventually diagnosed with schizophrenia or "other" psychoses – but not in those diagnosed with bipolar disorder – compared with healthy controls, results from a prospective, multicenter, 2-year follow-up study show.
Furthermore, the changes in those with schizophrenia were related to markers of poorer prognosis, Dr. Celso Arango and colleagues reported in the January issue of the Archives of General Psychiatry.
The results come from the Child and Adolescent First Episode Psychosis Study (CAFEPS). Participants were 110 patients with an initial episode of psychosis of less than 6 months’ duration and 98 healthy controls who were enrolled at six psychiatric units throughout Spain. Patients and controls were matched for age (mean, 15.5 years and 15.3 years, respectively), sex, and parental socioeconomic status. MRI was performed at baseline and at the 2-year follow-up in 25 patients with schizophrenia, 16 with bipolar disorder, and 20 with "other" psychosis, as well as in 70 healthy controls.
The investigators found that schizophrenia patients, compared with controls, had significantly greater mean losses of whole brain gray matter (–37.1 vs. –14.5 cm3) and greater mean increases of left frontal cerebrospinal fluid volumes (6.7 vs. 2.4 cm3), as well as significantly greater mean losses of gray matter volume in the frontal lobe (–3.3 vs. –0.6 cm3) and in the left parietal lobe (–4.3 vs. –2.2 cm3), reported Dr. Arango of the department of child and adolescent psychiatry at the Hospital General Universitario Gregorio Marañón in Madrid, a member of CIBERSAM (Centro de Investigación Biomédica en Red de Salud Mental).
Those with "other" psychoses had significantly greater losses in whole brain gray matter volume (–37.6 vs. –14.5 cm3) and significantly greater loss of gray matter in the frontal lobes (–4.3 vs. –0.6 cm3 for the left, and –4.2 vs. –0.8 cm3 for the right) and left parietal lobes (–5.3 vs. –2.2 cm3), and significantly greater increases in cerebrospinal fluid volume in the frontal lobes (7.1 vs. 2.4 cm3 for the left, and 3.2 vs. 0.8 cm3 for the right), the investigators said (Arch. Gen. Psychiatry 2012;69:16-26).
"Progressive brain changes seem to be more marked in those patients ending with a diagnosis of schizophrenia."
No significant differences were seen in longitudinal volume changes in the bipolar disorder patients, compared with the healthy controls, although the differences that did occur were in the same direction as those that occurred in the other patient subgroups, the investigators noted.
As for associations between volume changes and clinical and outcomes variables, the investigators also noted that "significant correlations were observed between left frontal [gray matter] volume change during follow-up and number of weeks hospitalized and right frontal [cerebrospinal fluid] change, with more weeks hospitalized correlating with greater [gray matter] volume loss and CSF increase in patients with schizophrenia."
A significant association between left frontal CSF volume change during follow-up and left parietal CSF change with mean negative PANSS (Positive and Negative Syndrome Scale) score in patients with schizophrenia also was observed, and left frontal CSF volume change correlated with the mean PANSS total score.
"With regard to clinical improvement, significant correlations were found for the schizophrenia group between the PANSS negative subscale score and PANSS general change score and left temporal [gray matter] loss, with less improvement correlating with larger reductions.
"Associations were also found between frontal right CSF and PANSS general change score and parietal right CSF and PANSS general change score, suggesting that greater CSF volume increase is related to less improvement in general symptoms as measured with PANSS in schizophrenic patients," they wrote.
In the "other" psychoses group, correlations were observed between general and positive and total PANSS change scores, and left parietal CSF loss, suggesting that decreases in CSF volume are associated with less improvement on total score and the presence of positive psychotic symptoms during follow-up.
The findings – which support those from a previous smaller study that also demonstrated that schizophrenia patients with a first episode of psychosis had significant frontal gray matter reductions, compared with healthy controls – suggest a continuum between early-onset and adult-onset psychotic disorders, the investigators said.
Although the underlying mechanisms for brain volume changes in schizophrenia and other psychotic disorder are not yet understood, the findings of this study suggest that one or more active pathophysiologic processes occur in the brains of children and adolescents after a first psychotic episode – particularly in those with schizophrenia, they noted.
"Findings from cross-sectional and longitudinal studies examining patients with adolescent-onset psychosis support the concept of [early onset psychosis] as a progressive neurodevelopmental disorder with both early and later neurodevelopmental abnormalities," the investigators wrote. "Progressive brain changes seem to be more marked in those patients ending with a diagnosis of schizophrenia, although progressive changes were present in other psychoses. Some of these pathophysiologic processes seem to be markers of poorer prognosis." They added that the development of therapeutic strategies aimed at counteracting these pathological progressive brain changes requires research focused on their neurobiological underpinnings.
"The correlates of volume changes at a cellular level and the study of risk genes involved in circuitries associated with different psychoses and their relationship to developmental trajectories may be promising areas of research," they noted.
The study was supported by the Spanish Ministry of Health and Social Policy, Spanish Ministry of Science and Innovation. Dr. Arango has been a consultant to or has received honoraria from AstraZeneca, Bristol-Myers Squibb, Janssen-Cilag, Lundbeck, Otsuka, Pfizer, Roche, Servier, and Schering-Plough.
FROM THE ARCHIVES OF GENERAL PSYCHIATRY
Major Finding: Young schizophrenia patients, compared with controls, had significantly greater mean losses of whole brain gray matter (–37.1 vs. –14.5 cm3) and greater mean increases of left frontal cerebrospinal fluid volumes (6.7 vs.2.4 cm3), as well as significantly greater mean losses of gray matter volume in the frontal lobe (–3.3 vs. –0.6 cm3) and in the left parietal lobe (–4.3 vs. –2.2 cm3).
Data Source: A prospective, multicenter, naturalistic, 2-year follow-up study of 110 adolescent patients.
Disclosures: The study was supported by the Spanish Ministry of Health and Social Policy, Spanish Ministry of Science and Innovation. Dr. Arango has been a consultant to or has received honoraria from AstraZeneca, Bristol-Myers Squibb, Janssen-Cilag, Lundbeck, Otsuka, Pfizer, Roche, Servier, and Schering-Plough.
RA, Periodontal Disease May Be Bi-Directional
CHICAGO – A causal relationship between rheumatoid arthritis and periodontal disease has not been confirmed, but recent data support the concept of a bi-directional relationship between the two.
In fact, a number of investigators have attempted to characterize the relationship between rheumatoid arthritis (RA) and periodontal disease. While most research confirms that one exists, the strength and extent of that relationship remain unclear, Michele Ravenel, D.M.D., said at the annual meeting of the American College of Rheumatology.
"Periodontal disease and RA do share similar pathologic mechanisms, and the relationship appears to be bi-directional," Dr. Ravenel said. But in her own review of the literature, the only consistency she found was inconsistency, said Dr. Ravenel.
In one recent case-control study involving 57 RA patients and 52 healthy subjects, those with RA were found to have significantly greater odds of having periodontitis after adjusting for a number of variables including RA status, age, sex, alcohol use, and body mass index (J. Periodontol. 2008;79:979-86).
"Periodontal disease and RA do share similar pathologic mechanisms, and the relationship appears to be bi-directional," said Michele Ravenel, D.M.D.
And in another recent study, 65 RA patients all were found to have some form of periodontal disease, which was moderate or severe in most cases. However, rheumatoid factor levels were not found to have any influence on oral bacterial composition and/or concentration – or on severity of periodontal disease (J. Periodontol. 2011;82:1424-32).
In yet another study of patients with both moderate to severe RA and severe periodontal disease, outcomes were compared in 10 patients on no treatment, 10 on periodontal therapy, 10 on anti-TNF-alpha therapy, and 10 on both periodontal therapy and anti-TNF-alpha therapy (J. Periodontol. 2009;80:535-40). Patients who were receiving treatment for periodontal disease – either with or without anti-TNF-alpha therapy for their RA – experienced significantly greater improvements in both the periodontal disease and the RA, compared with those not on periodontal therapy, said Dr. Ravenel of the Medical University of South Carolina, Charleston.
As for the role of periodontal pathogens in RA, findings from two recent case-control studies showed that serum antibodies to known periodontal pathogens were found more frequently in those with RA and periodontal disease than in controls (MedGenMed. 2005;7:2;Clin. Exp. Rheumatol. 2006;24:656-63).
In a cross-sectional study, bacterial DNA of some of the more virulent periodontal pathogens, including Porphyromonas gingivalis, Tannerella forsythia, and Prevotella intermedia, were identified in synovial fluid of RA-affected joints (J. Clin. Periodontol. 2009;1004-10).
In another study, which was presented separately at the 2011 annual meeting of the ACR, Dr. Jose U. Scher reported on a potentially important role for "a single species-level operational taxonomic unit belonging to the genus Porphyromonas and homologous to P. gingivalis," which he said could explain the link between RA and periodontal disease.
The particular Porphyromonas species was significantly more prevalent and more abundant in 25 patients with new-onset, never-treated RA, compared with 27 patients with established disease, and 14 healthy controls. That species accounted for nearly 10% of the bacteria in the new-onset RA patients, compared with about 3% and 4% of the bacteria in control patients and patients with chronic established disease, respectively, said Dr. Scher, of New York University Hospital for Joint Diseases.
More than 90% of the patients with new-onset RA in the study had moderate to severe periodontal disease. The study findings showed that the oral microbiome in these patients – all of whom were anticitrullinated peptide antibody-positive – is distinct at disease onset and characterized by an abundance of the virulent Porphyromonas species.
Further identification of the species may provide new insight regarding the reported link between RA and periodontal disease, he concluded.
Additional research should include strict adherence to diagnostic criteria for both diseases, Dr. Ravenel said.
Dr. Ravenel and Dr. Scher both said they have no relevant disclosures.
CHICAGO – A causal relationship between rheumatoid arthritis and periodontal disease has not been confirmed, but recent data support the concept of a bi-directional relationship between the two.
In fact, a number of investigators have attempted to characterize the relationship between rheumatoid arthritis (RA) and periodontal disease. While most research confirms that one exists, the strength and extent of that relationship remain unclear, Michele Ravenel, D.M.D., said at the annual meeting of the American College of Rheumatology.
"Periodontal disease and RA do share similar pathologic mechanisms, and the relationship appears to be bi-directional," Dr. Ravenel said. But in her own review of the literature, the only consistency she found was inconsistency, said Dr. Ravenel.
In one recent case-control study involving 57 RA patients and 52 healthy subjects, those with RA were found to have significantly greater odds of having periodontitis after adjusting for a number of variables including RA status, age, sex, alcohol use, and body mass index (J. Periodontol. 2008;79:979-86).
"Periodontal disease and RA do share similar pathologic mechanisms, and the relationship appears to be bi-directional," said Michele Ravenel, D.M.D.
And in another recent study, 65 RA patients all were found to have some form of periodontal disease, which was moderate or severe in most cases. However, rheumatoid factor levels were not found to have any influence on oral bacterial composition and/or concentration – or on severity of periodontal disease (J. Periodontol. 2011;82:1424-32).
In yet another study of patients with both moderate to severe RA and severe periodontal disease, outcomes were compared in 10 patients on no treatment, 10 on periodontal therapy, 10 on anti-TNF-alpha therapy, and 10 on both periodontal therapy and anti-TNF-alpha therapy (J. Periodontol. 2009;80:535-40). Patients who were receiving treatment for periodontal disease – either with or without anti-TNF-alpha therapy for their RA – experienced significantly greater improvements in both the periodontal disease and the RA, compared with those not on periodontal therapy, said Dr. Ravenel of the Medical University of South Carolina, Charleston.
As for the role of periodontal pathogens in RA, findings from two recent case-control studies showed that serum antibodies to known periodontal pathogens were found more frequently in those with RA and periodontal disease than in controls (MedGenMed. 2005;7:2;Clin. Exp. Rheumatol. 2006;24:656-63).
In a cross-sectional study, bacterial DNA of some of the more virulent periodontal pathogens, including Porphyromonas gingivalis, Tannerella forsythia, and Prevotella intermedia, were identified in synovial fluid of RA-affected joints (J. Clin. Periodontol. 2009;1004-10).
In another study, which was presented separately at the 2011 annual meeting of the ACR, Dr. Jose U. Scher reported on a potentially important role for "a single species-level operational taxonomic unit belonging to the genus Porphyromonas and homologous to P. gingivalis," which he said could explain the link between RA and periodontal disease.
The particular Porphyromonas species was significantly more prevalent and more abundant in 25 patients with new-onset, never-treated RA, compared with 27 patients with established disease, and 14 healthy controls. That species accounted for nearly 10% of the bacteria in the new-onset RA patients, compared with about 3% and 4% of the bacteria in control patients and patients with chronic established disease, respectively, said Dr. Scher, of New York University Hospital for Joint Diseases.
More than 90% of the patients with new-onset RA in the study had moderate to severe periodontal disease. The study findings showed that the oral microbiome in these patients – all of whom were anticitrullinated peptide antibody-positive – is distinct at disease onset and characterized by an abundance of the virulent Porphyromonas species.
Further identification of the species may provide new insight regarding the reported link between RA and periodontal disease, he concluded.
Additional research should include strict adherence to diagnostic criteria for both diseases, Dr. Ravenel said.
Dr. Ravenel and Dr. Scher both said they have no relevant disclosures.
CHICAGO – A causal relationship between rheumatoid arthritis and periodontal disease has not been confirmed, but recent data support the concept of a bi-directional relationship between the two.
In fact, a number of investigators have attempted to characterize the relationship between rheumatoid arthritis (RA) and periodontal disease. While most research confirms that one exists, the strength and extent of that relationship remain unclear, Michele Ravenel, D.M.D., said at the annual meeting of the American College of Rheumatology.
"Periodontal disease and RA do share similar pathologic mechanisms, and the relationship appears to be bi-directional," Dr. Ravenel said. But in her own review of the literature, the only consistency she found was inconsistency, said Dr. Ravenel.
In one recent case-control study involving 57 RA patients and 52 healthy subjects, those with RA were found to have significantly greater odds of having periodontitis after adjusting for a number of variables including RA status, age, sex, alcohol use, and body mass index (J. Periodontol. 2008;79:979-86).
"Periodontal disease and RA do share similar pathologic mechanisms, and the relationship appears to be bi-directional," said Michele Ravenel, D.M.D.
And in another recent study, 65 RA patients all were found to have some form of periodontal disease, which was moderate or severe in most cases. However, rheumatoid factor levels were not found to have any influence on oral bacterial composition and/or concentration – or on severity of periodontal disease (J. Periodontol. 2011;82:1424-32).
In yet another study of patients with both moderate to severe RA and severe periodontal disease, outcomes were compared in 10 patients on no treatment, 10 on periodontal therapy, 10 on anti-TNF-alpha therapy, and 10 on both periodontal therapy and anti-TNF-alpha therapy (J. Periodontol. 2009;80:535-40). Patients who were receiving treatment for periodontal disease – either with or without anti-TNF-alpha therapy for their RA – experienced significantly greater improvements in both the periodontal disease and the RA, compared with those not on periodontal therapy, said Dr. Ravenel of the Medical University of South Carolina, Charleston.
As for the role of periodontal pathogens in RA, findings from two recent case-control studies showed that serum antibodies to known periodontal pathogens were found more frequently in those with RA and periodontal disease than in controls (MedGenMed. 2005;7:2;Clin. Exp. Rheumatol. 2006;24:656-63).
In a cross-sectional study, bacterial DNA of some of the more virulent periodontal pathogens, including Porphyromonas gingivalis, Tannerella forsythia, and Prevotella intermedia, were identified in synovial fluid of RA-affected joints (J. Clin. Periodontol. 2009;1004-10).
In another study, which was presented separately at the 2011 annual meeting of the ACR, Dr. Jose U. Scher reported on a potentially important role for "a single species-level operational taxonomic unit belonging to the genus Porphyromonas and homologous to P. gingivalis," which he said could explain the link between RA and periodontal disease.
The particular Porphyromonas species was significantly more prevalent and more abundant in 25 patients with new-onset, never-treated RA, compared with 27 patients with established disease, and 14 healthy controls. That species accounted for nearly 10% of the bacteria in the new-onset RA patients, compared with about 3% and 4% of the bacteria in control patients and patients with chronic established disease, respectively, said Dr. Scher, of New York University Hospital for Joint Diseases.
More than 90% of the patients with new-onset RA in the study had moderate to severe periodontal disease. The study findings showed that the oral microbiome in these patients – all of whom were anticitrullinated peptide antibody-positive – is distinct at disease onset and characterized by an abundance of the virulent Porphyromonas species.
Further identification of the species may provide new insight regarding the reported link between RA and periodontal disease, he concluded.
Additional research should include strict adherence to diagnostic criteria for both diseases, Dr. Ravenel said.
Dr. Ravenel and Dr. Scher both said they have no relevant disclosures.
FROM THE ANNUAL MEETING OF THE AMERICAN COLLEGE OF RHEUMATOLOGY
New-Onset AF Seen in One-Third of TAVI Patients
New-onset atrial fibrillation, or NOAF, occurred in nearly a third of 138 patients who underwent transcatheter aortic valve implantation in a prospective study, and the condition was associated with a significantly higher rate of cardioembolic events at 30-day and 12-month follow-up.
NOAF occurred in 44 patients at a median of 48 hours after transcatheter aortic valve implantation (TAVI), with 10 episodes resolving spontaneously within 12 hours, and 34 episodes requiring pharmacologic intervention. The rate of the combined end point of stroke and systemic embolism was 13.6% at 30 days in the NOAF group, compared with 3.2% in those who did not experience NOAF (odds ratio, 4.79); the rate was 15.9% vs. 3.2% at 12 months (OR, 5.0), Dr. Ignacio J. Amat-Santos of Laval University, Quebec City, and his colleagues reported online on Dec. 14 the Journal of the American College of Cardiology.
Significant independent predictors of NOAF included left atrial size (OR of 1.21/each increase of 1 mm/m2) and use of the transapical approach (OR, 4.08). An atrial size of 27 mm/m2 or greater as measured by echocardiography was identified as the cutoff point with the best sensitivity and specificity for predicting NOAF within 30 days of TAVI, the investigators said (J. Am. Coll. Cardiol. 2011 [doi:10/1016/j.jacc.2011.09.061]).
Furthermore, the incidence of NOAF in those with an atrial size of 27 mm/m2 or greater undergoing TAVI by the transapical approach vs. the transfemoral approach was 57% vs. 12%, respectively, they noted.
Mortality, which was 16% and 21% in those with and without NOAF at 12 months, respectively, did not differ significantly between the groups at either follow-up, they noted.
Patients in the study had severe symptomatic aortic stenosis, but no history of chronic/paroxysmal atrial fibrillation. They underwent TAVI with a balloon-expandable valve by either the transapical or transfemoral approach, were enrolled consecutively between May 2007 and May 2011, and underwent continuous electrocardiographic monitoring during hospitalization. NOAF was defined as any episode of AF lasting longer than 30 seconds.
NOAF is a known complication in patients undergoing cardiovascular interventions, but this is one of the first studies to verify an increased risk of the condition in patients undergoing TAVI, which has emerged as an alternative treatment for patients with severe symptomatic aortic stenosis and high or prohibitive operative risk. The study is also among the first to characterize predictive factors for NOAF.
"The occurrence of cerebrovascular events is probably the most worrisome complication associated with TAVI, with an incidence of about 4%, one of the highest ever reported in the field of interventional cardiology," the investigators said, noting that the fact that more than 50% of the complications occur several days after TAVI suggests that "mechanisms other than those directly related to the procedure may be involved."
Indeed, the findings of this study suggest NOAF may be an important mechanism for late neurological events after TAVI, and if confirmed in larger studies, the findings may have implications for the development of prevention trials.
"Knowing the predictive factors of NOAF following TAVI should allow us to select the patients at higher risk for NOAF preventive studies in the setting of TAVI," they investigators said.
Also of note, some of the cardioembolic events in this study seemed to be related to the non-initiation of anticoagulant therapy, the investigators said, adding that the risk of a cardioembolic event was as high as 40% in those who had NOAF and who did not receive anticoagulant therapy.
This underscores the clinical relevance of optimizing antithrombotic treatment in this high-risk subset of patients, they concluded.
Dr. Amat-Santos disclosed that he received support from the Institute of Heart Sciences. Other authors disclosed having served as consultants for Edwards Lifesciences, and/or St. Jude Medical.
The research by Dr. Amat-Santos and his colleagues deserves attention because of its originality and because of its potential implications for managing TAVI patients, according to Dr. Bernard Iung, Dr. Dominique Himbert, and Dr. Alex Vahanian.
In an accompanying editorial, they note that besides frequency and predictive factors of NOAF in patients who undergo TAVI, one of the most clinically relevant issues addressed by the study is the assessment of its impact following the procedure. While the findings should be interpreted with caution given the small number of embolic events, they represent a "relevant contribution in the analysis of the potential causes of embolic events after TAVI," they said (J. Am. Coll. Cardiol. 2012 [doi:10.1016/j.jacc2011.11.005]).
"Despite the number of mechanisms that may be involved in stroke after TAVI, the particularly strong relationship between post-procedural AF and stroke occurring after 24 h suggests that a cardioembolic origin significantly contributes to stroke after TAVI," they added, noting that the association raises questions about the most appropriate post-procedure antithrombotic therapy, and highlights the need for larger randomized trials to optimize antithrombotic therapy in TAVI patients.
The association also highlights the need for improving the identification of patients at risk, and testing pharmacological approaches to reducing atrial fibrillation incidence, they concluded.
Dr. Iung, Dr. Himbert, and Dr. Vahanian are with Bichat Hospital, Paris. Dr. Iung disclosed that he has received consulting fees from Servier, Boehringer Ingelheim, Bayer, Valtech, and Abbott, as well as speakers fees from Edwards Lifesciences, St. Jude Medical, and Sanofi-Aventis. Dr. Himbert disclosed receiving proctoring fees from Edwards Lifesciences and Medtronic. Dr. Vahanian disclosed serving as an advisory board member for Medtronic, Abbott, Valtech, and Boehringer Ingelheim, and receiving speakers fees from Edwards Lifesciences and Siemens.
The research by Dr. Amat-Santos and his colleagues deserves attention because of its originality and because of its potential implications for managing TAVI patients, according to Dr. Bernard Iung, Dr. Dominique Himbert, and Dr. Alex Vahanian.
In an accompanying editorial, they note that besides frequency and predictive factors of NOAF in patients who undergo TAVI, one of the most clinically relevant issues addressed by the study is the assessment of its impact following the procedure. While the findings should be interpreted with caution given the small number of embolic events, they represent a "relevant contribution in the analysis of the potential causes of embolic events after TAVI," they said (J. Am. Coll. Cardiol. 2012 [doi:10.1016/j.jacc2011.11.005]).
"Despite the number of mechanisms that may be involved in stroke after TAVI, the particularly strong relationship between post-procedural AF and stroke occurring after 24 h suggests that a cardioembolic origin significantly contributes to stroke after TAVI," they added, noting that the association raises questions about the most appropriate post-procedure antithrombotic therapy, and highlights the need for larger randomized trials to optimize antithrombotic therapy in TAVI patients.
The association also highlights the need for improving the identification of patients at risk, and testing pharmacological approaches to reducing atrial fibrillation incidence, they concluded.
Dr. Iung, Dr. Himbert, and Dr. Vahanian are with Bichat Hospital, Paris. Dr. Iung disclosed that he has received consulting fees from Servier, Boehringer Ingelheim, Bayer, Valtech, and Abbott, as well as speakers fees from Edwards Lifesciences, St. Jude Medical, and Sanofi-Aventis. Dr. Himbert disclosed receiving proctoring fees from Edwards Lifesciences and Medtronic. Dr. Vahanian disclosed serving as an advisory board member for Medtronic, Abbott, Valtech, and Boehringer Ingelheim, and receiving speakers fees from Edwards Lifesciences and Siemens.
The research by Dr. Amat-Santos and his colleagues deserves attention because of its originality and because of its potential implications for managing TAVI patients, according to Dr. Bernard Iung, Dr. Dominique Himbert, and Dr. Alex Vahanian.
In an accompanying editorial, they note that besides frequency and predictive factors of NOAF in patients who undergo TAVI, one of the most clinically relevant issues addressed by the study is the assessment of its impact following the procedure. While the findings should be interpreted with caution given the small number of embolic events, they represent a "relevant contribution in the analysis of the potential causes of embolic events after TAVI," they said (J. Am. Coll. Cardiol. 2012 [doi:10.1016/j.jacc2011.11.005]).
"Despite the number of mechanisms that may be involved in stroke after TAVI, the particularly strong relationship between post-procedural AF and stroke occurring after 24 h suggests that a cardioembolic origin significantly contributes to stroke after TAVI," they added, noting that the association raises questions about the most appropriate post-procedure antithrombotic therapy, and highlights the need for larger randomized trials to optimize antithrombotic therapy in TAVI patients.
The association also highlights the need for improving the identification of patients at risk, and testing pharmacological approaches to reducing atrial fibrillation incidence, they concluded.
Dr. Iung, Dr. Himbert, and Dr. Vahanian are with Bichat Hospital, Paris. Dr. Iung disclosed that he has received consulting fees from Servier, Boehringer Ingelheim, Bayer, Valtech, and Abbott, as well as speakers fees from Edwards Lifesciences, St. Jude Medical, and Sanofi-Aventis. Dr. Himbert disclosed receiving proctoring fees from Edwards Lifesciences and Medtronic. Dr. Vahanian disclosed serving as an advisory board member for Medtronic, Abbott, Valtech, and Boehringer Ingelheim, and receiving speakers fees from Edwards Lifesciences and Siemens.
New-onset atrial fibrillation, or NOAF, occurred in nearly a third of 138 patients who underwent transcatheter aortic valve implantation in a prospective study, and the condition was associated with a significantly higher rate of cardioembolic events at 30-day and 12-month follow-up.
NOAF occurred in 44 patients at a median of 48 hours after transcatheter aortic valve implantation (TAVI), with 10 episodes resolving spontaneously within 12 hours, and 34 episodes requiring pharmacologic intervention. The rate of the combined end point of stroke and systemic embolism was 13.6% at 30 days in the NOAF group, compared with 3.2% in those who did not experience NOAF (odds ratio, 4.79); the rate was 15.9% vs. 3.2% at 12 months (OR, 5.0), Dr. Ignacio J. Amat-Santos of Laval University, Quebec City, and his colleagues reported online on Dec. 14 the Journal of the American College of Cardiology.
Significant independent predictors of NOAF included left atrial size (OR of 1.21/each increase of 1 mm/m2) and use of the transapical approach (OR, 4.08). An atrial size of 27 mm/m2 or greater as measured by echocardiography was identified as the cutoff point with the best sensitivity and specificity for predicting NOAF within 30 days of TAVI, the investigators said (J. Am. Coll. Cardiol. 2011 [doi:10/1016/j.jacc.2011.09.061]).
Furthermore, the incidence of NOAF in those with an atrial size of 27 mm/m2 or greater undergoing TAVI by the transapical approach vs. the transfemoral approach was 57% vs. 12%, respectively, they noted.
Mortality, which was 16% and 21% in those with and without NOAF at 12 months, respectively, did not differ significantly between the groups at either follow-up, they noted.
Patients in the study had severe symptomatic aortic stenosis, but no history of chronic/paroxysmal atrial fibrillation. They underwent TAVI with a balloon-expandable valve by either the transapical or transfemoral approach, were enrolled consecutively between May 2007 and May 2011, and underwent continuous electrocardiographic monitoring during hospitalization. NOAF was defined as any episode of AF lasting longer than 30 seconds.
NOAF is a known complication in patients undergoing cardiovascular interventions, but this is one of the first studies to verify an increased risk of the condition in patients undergoing TAVI, which has emerged as an alternative treatment for patients with severe symptomatic aortic stenosis and high or prohibitive operative risk. The study is also among the first to characterize predictive factors for NOAF.
"The occurrence of cerebrovascular events is probably the most worrisome complication associated with TAVI, with an incidence of about 4%, one of the highest ever reported in the field of interventional cardiology," the investigators said, noting that the fact that more than 50% of the complications occur several days after TAVI suggests that "mechanisms other than those directly related to the procedure may be involved."
Indeed, the findings of this study suggest NOAF may be an important mechanism for late neurological events after TAVI, and if confirmed in larger studies, the findings may have implications for the development of prevention trials.
"Knowing the predictive factors of NOAF following TAVI should allow us to select the patients at higher risk for NOAF preventive studies in the setting of TAVI," they investigators said.
Also of note, some of the cardioembolic events in this study seemed to be related to the non-initiation of anticoagulant therapy, the investigators said, adding that the risk of a cardioembolic event was as high as 40% in those who had NOAF and who did not receive anticoagulant therapy.
This underscores the clinical relevance of optimizing antithrombotic treatment in this high-risk subset of patients, they concluded.
Dr. Amat-Santos disclosed that he received support from the Institute of Heart Sciences. Other authors disclosed having served as consultants for Edwards Lifesciences, and/or St. Jude Medical.
New-onset atrial fibrillation, or NOAF, occurred in nearly a third of 138 patients who underwent transcatheter aortic valve implantation in a prospective study, and the condition was associated with a significantly higher rate of cardioembolic events at 30-day and 12-month follow-up.
NOAF occurred in 44 patients at a median of 48 hours after transcatheter aortic valve implantation (TAVI), with 10 episodes resolving spontaneously within 12 hours, and 34 episodes requiring pharmacologic intervention. The rate of the combined end point of stroke and systemic embolism was 13.6% at 30 days in the NOAF group, compared with 3.2% in those who did not experience NOAF (odds ratio, 4.79); the rate was 15.9% vs. 3.2% at 12 months (OR, 5.0), Dr. Ignacio J. Amat-Santos of Laval University, Quebec City, and his colleagues reported online on Dec. 14 the Journal of the American College of Cardiology.
Significant independent predictors of NOAF included left atrial size (OR of 1.21/each increase of 1 mm/m2) and use of the transapical approach (OR, 4.08). An atrial size of 27 mm/m2 or greater as measured by echocardiography was identified as the cutoff point with the best sensitivity and specificity for predicting NOAF within 30 days of TAVI, the investigators said (J. Am. Coll. Cardiol. 2011 [doi:10/1016/j.jacc.2011.09.061]).
Furthermore, the incidence of NOAF in those with an atrial size of 27 mm/m2 or greater undergoing TAVI by the transapical approach vs. the transfemoral approach was 57% vs. 12%, respectively, they noted.
Mortality, which was 16% and 21% in those with and without NOAF at 12 months, respectively, did not differ significantly between the groups at either follow-up, they noted.
Patients in the study had severe symptomatic aortic stenosis, but no history of chronic/paroxysmal atrial fibrillation. They underwent TAVI with a balloon-expandable valve by either the transapical or transfemoral approach, were enrolled consecutively between May 2007 and May 2011, and underwent continuous electrocardiographic monitoring during hospitalization. NOAF was defined as any episode of AF lasting longer than 30 seconds.
NOAF is a known complication in patients undergoing cardiovascular interventions, but this is one of the first studies to verify an increased risk of the condition in patients undergoing TAVI, which has emerged as an alternative treatment for patients with severe symptomatic aortic stenosis and high or prohibitive operative risk. The study is also among the first to characterize predictive factors for NOAF.
"The occurrence of cerebrovascular events is probably the most worrisome complication associated with TAVI, with an incidence of about 4%, one of the highest ever reported in the field of interventional cardiology," the investigators said, noting that the fact that more than 50% of the complications occur several days after TAVI suggests that "mechanisms other than those directly related to the procedure may be involved."
Indeed, the findings of this study suggest NOAF may be an important mechanism for late neurological events after TAVI, and if confirmed in larger studies, the findings may have implications for the development of prevention trials.
"Knowing the predictive factors of NOAF following TAVI should allow us to select the patients at higher risk for NOAF preventive studies in the setting of TAVI," they investigators said.
Also of note, some of the cardioembolic events in this study seemed to be related to the non-initiation of anticoagulant therapy, the investigators said, adding that the risk of a cardioembolic event was as high as 40% in those who had NOAF and who did not receive anticoagulant therapy.
This underscores the clinical relevance of optimizing antithrombotic treatment in this high-risk subset of patients, they concluded.
Dr. Amat-Santos disclosed that he received support from the Institute of Heart Sciences. Other authors disclosed having served as consultants for Edwards Lifesciences, and/or St. Jude Medical.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
Major Finding: NOAF occurred in 44 patients at a median of 48 hours after transcatheter aortic valve implantation (TAVI), with 10 episodes resolving spontaneously within 12 hours, and 34 requiring pharmacologic intervention. The rate of the combined end point of stroke and systemic embolism was 13.6% at 30 days in the NOAF group, compared with 3.2% (odds ratio 4.79) in those who did not experience NOAF; the rate was 15.9% vs. 3.2% at 12 months (odds ratio 5.0).
Data Source: A prospective study of 138 patients undergoing TAVI.
Disclosures: Dr. Amat-Santos disclosed that he received support from the Institute of Heart Sciences. Other authors disclosed that they have consulted for Edwards Lifesciences Inc., and/or St. Jude Medical.
Study Characterizes Spectrum of Somatic Mutations in Chronic Lymphocytic Leukemia
Researchers using massively parallel sequencing to characterize the spectrum of somatic mutations in chronic lymphocytic leukemia identified nine genes that are mutated at significant frequencies in the disease.
Of these nine "driver" genes, which were identified from DNA samples from normal tissues and tumors in 91 patients with chronic lymphocytic leukemia, four have previously established roles in the disease (TP53, ATM, MYD88, and NOTCH1), and five do not (SF3B1, ZMYM3, MAPK1, FBXW7, and DDX3X), Dr. Lili Wang of the Dana-Farber Cancer Institute, Boston, and her colleagues reported online in the Dec. 12 issue of the New England Journal of Medicine. The findings were reported simultaneously at the annual meeting of the American Society of Hematology.
"Strikingly, the second most frequently mutated gene in our cohort was splicing factor 3b, subunit 1 (SF3B1), with missense mutations occurring in 14 of 91 patients (15%). SF3B1 is a component of the SF3B complex, which is associated with the U2 small nuclear ribonucleoprotein (snRNP), at the catalytic center of the spliceosome," the investigators said (N. Engl. J. Med. 2011 Dec. 12 [doi:10.1056/NEJMoa1109016]).
The nine driver genes identified in this study appear in five core signaling pathways, and the genes each play different, though well-established roles in these pathways, such as DNA repair and cell-cycle control, Notch signaling, inflammatory pathways, and RNA splicing (in which SF3B1, in particular, was implicated). Furthermore, each driver mutation was found to be associated with different key abnormalities; for example, the findings indicate there may be an interaction between del(11q), which is associated with aggressive disease, and SF3B1 mutation in the pathogenesis of a clinical subgroup of chronic lymphocytic leukemia.
Overall, the investigators concluded that:
• Chronic lymphocytic leukemia has a lower rate of somatic mutation than most solid tumors.
• The rate of nonsynonymous mutation was not strongly affected by therapy.
• In addition to the expected mutations in cell-cycle and DNA-repair pathways, genetic alterations in Notch signaling, inflammatory pathways, and RNA splicing and processing also exist.
• Driver mutations showed striking associations with standard prognostic markers.
The latter suggests that "particular combinations of genetic alterations may act in concert to drive cancer," the investigators said.
The findings regarding the core spliceosome component SF3B1, which they described as a "major surprise," led to further analyses that suggested that SF3B1 mutations lead to "mistakes in the splicing of ... transcripts that affect the pathogenesis of chronic lymphocytic leukemia," they said, adding that ongoing studies will evaluate how mutations in SF3B1 alter its function in the processing of critical messenger RNAs.
The study, and in particular the findings regarding SF3B1 mutations, illustrate how identification of coding mutations in chronic lymphocytic leukemia can lead to the development of mechanistic hypotheses, novel prognostic markers, and potential therapeutic targets, Dr. Wang and her associates said.
They also noted the information provides a starting point for determining "which genes within chromosomal deletions and amplifications are essential, how each mutation alters cellular networks and phenotypes, which combinations of mutations are critical in the development of cancer, and how genetic events in the host may affect the importance of specific mutations and their combinations."
In an accompanying editorial, Dr. Benjamin Ebert and Olivier A. Bernard, Ph.D., commented that the study finding regarding mutations in genes involved in RNA splicing, although highly unexpected, converge remarkably with recent published findings from studies of myelodysplastic syndromes (N. Engl. J. Med. 2011;35:1384-95; Nature 2011;478:64-9).
SF3B1 mutations were found in 20% of patients with myelodysplastic syndromes, and in 65% of patients with refractory anemia and ring sideroblasts in one study (N. Engl. J. Med. 2011;35:1384-95).
"Moreover, mutations have been reported in multiple components of the spliceosome in 45%-85% of patients with myelodysplastic syndrome. SF3B1 mutations also occur in 1%-5% of samples from a wide range of tumor types, which indicates that mutations in RNA splicing factors are a widespread cause of oncogenic transformation," they added.
These and other findings, taken together, raise the "provocative possibility" that SF3B1 mutations may sometimes occur first in hematopoietic stem cells, with "additional mutations then being acquired in either the lymphoid or the myeloid lineages and causing chronic lymphocytic leukemia or myelodysplastic syndromes, respectively," Dr. Ebert of Brigham and Women’s Hospital, Boston, and Dr. Bernard, who is with INSERM at Institut Gustave Roussy, Villejuif, France, wrote (N. Engl. J. Med. 2011 Dec. 12 [doi:10.1056/NEJMe1111584]).
The findings have implications for determining prognosis, and for identifying targets for treatment. For example, the identification of mutations in genes encoding the RNA splicing machinery raises the possibility that spliceosome could be a therapeutic target, they said.
This study was supported by grants from the National Institutes of Health, the National Cancer Institute, the Melton and Rosenbach Funds, the Blavatnik Family Foundation, the Howard Hughes Medical Institute (via an Early Career Physician-Scientist Award), and the Damon Runyon Cancer Research Foundation. Dr. Wang said she had no relevant financial disclosures. Some of the coauthors said they were consultants to numerous pharmaceutical companies or owned stock in pharmaceutical companies. Other than employment by INSERM on the part of Dr. Bernard, neither he nor Dr. Ebert had any relevant financial disclosures to report.
Researchers using massively parallel sequencing to characterize the spectrum of somatic mutations in chronic lymphocytic leukemia identified nine genes that are mutated at significant frequencies in the disease.
Of these nine "driver" genes, which were identified from DNA samples from normal tissues and tumors in 91 patients with chronic lymphocytic leukemia, four have previously established roles in the disease (TP53, ATM, MYD88, and NOTCH1), and five do not (SF3B1, ZMYM3, MAPK1, FBXW7, and DDX3X), Dr. Lili Wang of the Dana-Farber Cancer Institute, Boston, and her colleagues reported online in the Dec. 12 issue of the New England Journal of Medicine. The findings were reported simultaneously at the annual meeting of the American Society of Hematology.
"Strikingly, the second most frequently mutated gene in our cohort was splicing factor 3b, subunit 1 (SF3B1), with missense mutations occurring in 14 of 91 patients (15%). SF3B1 is a component of the SF3B complex, which is associated with the U2 small nuclear ribonucleoprotein (snRNP), at the catalytic center of the spliceosome," the investigators said (N. Engl. J. Med. 2011 Dec. 12 [doi:10.1056/NEJMoa1109016]).
The nine driver genes identified in this study appear in five core signaling pathways, and the genes each play different, though well-established roles in these pathways, such as DNA repair and cell-cycle control, Notch signaling, inflammatory pathways, and RNA splicing (in which SF3B1, in particular, was implicated). Furthermore, each driver mutation was found to be associated with different key abnormalities; for example, the findings indicate there may be an interaction between del(11q), which is associated with aggressive disease, and SF3B1 mutation in the pathogenesis of a clinical subgroup of chronic lymphocytic leukemia.
Overall, the investigators concluded that:
• Chronic lymphocytic leukemia has a lower rate of somatic mutation than most solid tumors.
• The rate of nonsynonymous mutation was not strongly affected by therapy.
• In addition to the expected mutations in cell-cycle and DNA-repair pathways, genetic alterations in Notch signaling, inflammatory pathways, and RNA splicing and processing also exist.
• Driver mutations showed striking associations with standard prognostic markers.
The latter suggests that "particular combinations of genetic alterations may act in concert to drive cancer," the investigators said.
The findings regarding the core spliceosome component SF3B1, which they described as a "major surprise," led to further analyses that suggested that SF3B1 mutations lead to "mistakes in the splicing of ... transcripts that affect the pathogenesis of chronic lymphocytic leukemia," they said, adding that ongoing studies will evaluate how mutations in SF3B1 alter its function in the processing of critical messenger RNAs.
The study, and in particular the findings regarding SF3B1 mutations, illustrate how identification of coding mutations in chronic lymphocytic leukemia can lead to the development of mechanistic hypotheses, novel prognostic markers, and potential therapeutic targets, Dr. Wang and her associates said.
They also noted the information provides a starting point for determining "which genes within chromosomal deletions and amplifications are essential, how each mutation alters cellular networks and phenotypes, which combinations of mutations are critical in the development of cancer, and how genetic events in the host may affect the importance of specific mutations and their combinations."
In an accompanying editorial, Dr. Benjamin Ebert and Olivier A. Bernard, Ph.D., commented that the study finding regarding mutations in genes involved in RNA splicing, although highly unexpected, converge remarkably with recent published findings from studies of myelodysplastic syndromes (N. Engl. J. Med. 2011;35:1384-95; Nature 2011;478:64-9).
SF3B1 mutations were found in 20% of patients with myelodysplastic syndromes, and in 65% of patients with refractory anemia and ring sideroblasts in one study (N. Engl. J. Med. 2011;35:1384-95).
"Moreover, mutations have been reported in multiple components of the spliceosome in 45%-85% of patients with myelodysplastic syndrome. SF3B1 mutations also occur in 1%-5% of samples from a wide range of tumor types, which indicates that mutations in RNA splicing factors are a widespread cause of oncogenic transformation," they added.
These and other findings, taken together, raise the "provocative possibility" that SF3B1 mutations may sometimes occur first in hematopoietic stem cells, with "additional mutations then being acquired in either the lymphoid or the myeloid lineages and causing chronic lymphocytic leukemia or myelodysplastic syndromes, respectively," Dr. Ebert of Brigham and Women’s Hospital, Boston, and Dr. Bernard, who is with INSERM at Institut Gustave Roussy, Villejuif, France, wrote (N. Engl. J. Med. 2011 Dec. 12 [doi:10.1056/NEJMe1111584]).
The findings have implications for determining prognosis, and for identifying targets for treatment. For example, the identification of mutations in genes encoding the RNA splicing machinery raises the possibility that spliceosome could be a therapeutic target, they said.
This study was supported by grants from the National Institutes of Health, the National Cancer Institute, the Melton and Rosenbach Funds, the Blavatnik Family Foundation, the Howard Hughes Medical Institute (via an Early Career Physician-Scientist Award), and the Damon Runyon Cancer Research Foundation. Dr. Wang said she had no relevant financial disclosures. Some of the coauthors said they were consultants to numerous pharmaceutical companies or owned stock in pharmaceutical companies. Other than employment by INSERM on the part of Dr. Bernard, neither he nor Dr. Ebert had any relevant financial disclosures to report.
Researchers using massively parallel sequencing to characterize the spectrum of somatic mutations in chronic lymphocytic leukemia identified nine genes that are mutated at significant frequencies in the disease.
Of these nine "driver" genes, which were identified from DNA samples from normal tissues and tumors in 91 patients with chronic lymphocytic leukemia, four have previously established roles in the disease (TP53, ATM, MYD88, and NOTCH1), and five do not (SF3B1, ZMYM3, MAPK1, FBXW7, and DDX3X), Dr. Lili Wang of the Dana-Farber Cancer Institute, Boston, and her colleagues reported online in the Dec. 12 issue of the New England Journal of Medicine. The findings were reported simultaneously at the annual meeting of the American Society of Hematology.
"Strikingly, the second most frequently mutated gene in our cohort was splicing factor 3b, subunit 1 (SF3B1), with missense mutations occurring in 14 of 91 patients (15%). SF3B1 is a component of the SF3B complex, which is associated with the U2 small nuclear ribonucleoprotein (snRNP), at the catalytic center of the spliceosome," the investigators said (N. Engl. J. Med. 2011 Dec. 12 [doi:10.1056/NEJMoa1109016]).
The nine driver genes identified in this study appear in five core signaling pathways, and the genes each play different, though well-established roles in these pathways, such as DNA repair and cell-cycle control, Notch signaling, inflammatory pathways, and RNA splicing (in which SF3B1, in particular, was implicated). Furthermore, each driver mutation was found to be associated with different key abnormalities; for example, the findings indicate there may be an interaction between del(11q), which is associated with aggressive disease, and SF3B1 mutation in the pathogenesis of a clinical subgroup of chronic lymphocytic leukemia.
Overall, the investigators concluded that:
• Chronic lymphocytic leukemia has a lower rate of somatic mutation than most solid tumors.
• The rate of nonsynonymous mutation was not strongly affected by therapy.
• In addition to the expected mutations in cell-cycle and DNA-repair pathways, genetic alterations in Notch signaling, inflammatory pathways, and RNA splicing and processing also exist.
• Driver mutations showed striking associations with standard prognostic markers.
The latter suggests that "particular combinations of genetic alterations may act in concert to drive cancer," the investigators said.
The findings regarding the core spliceosome component SF3B1, which they described as a "major surprise," led to further analyses that suggested that SF3B1 mutations lead to "mistakes in the splicing of ... transcripts that affect the pathogenesis of chronic lymphocytic leukemia," they said, adding that ongoing studies will evaluate how mutations in SF3B1 alter its function in the processing of critical messenger RNAs.
The study, and in particular the findings regarding SF3B1 mutations, illustrate how identification of coding mutations in chronic lymphocytic leukemia can lead to the development of mechanistic hypotheses, novel prognostic markers, and potential therapeutic targets, Dr. Wang and her associates said.
They also noted the information provides a starting point for determining "which genes within chromosomal deletions and amplifications are essential, how each mutation alters cellular networks and phenotypes, which combinations of mutations are critical in the development of cancer, and how genetic events in the host may affect the importance of specific mutations and their combinations."
In an accompanying editorial, Dr. Benjamin Ebert and Olivier A. Bernard, Ph.D., commented that the study finding regarding mutations in genes involved in RNA splicing, although highly unexpected, converge remarkably with recent published findings from studies of myelodysplastic syndromes (N. Engl. J. Med. 2011;35:1384-95; Nature 2011;478:64-9).
SF3B1 mutations were found in 20% of patients with myelodysplastic syndromes, and in 65% of patients with refractory anemia and ring sideroblasts in one study (N. Engl. J. Med. 2011;35:1384-95).
"Moreover, mutations have been reported in multiple components of the spliceosome in 45%-85% of patients with myelodysplastic syndrome. SF3B1 mutations also occur in 1%-5% of samples from a wide range of tumor types, which indicates that mutations in RNA splicing factors are a widespread cause of oncogenic transformation," they added.
These and other findings, taken together, raise the "provocative possibility" that SF3B1 mutations may sometimes occur first in hematopoietic stem cells, with "additional mutations then being acquired in either the lymphoid or the myeloid lineages and causing chronic lymphocytic leukemia or myelodysplastic syndromes, respectively," Dr. Ebert of Brigham and Women’s Hospital, Boston, and Dr. Bernard, who is with INSERM at Institut Gustave Roussy, Villejuif, France, wrote (N. Engl. J. Med. 2011 Dec. 12 [doi:10.1056/NEJMe1111584]).
The findings have implications for determining prognosis, and for identifying targets for treatment. For example, the identification of mutations in genes encoding the RNA splicing machinery raises the possibility that spliceosome could be a therapeutic target, they said.
This study was supported by grants from the National Institutes of Health, the National Cancer Institute, the Melton and Rosenbach Funds, the Blavatnik Family Foundation, the Howard Hughes Medical Institute (via an Early Career Physician-Scientist Award), and the Damon Runyon Cancer Research Foundation. Dr. Wang said she had no relevant financial disclosures. Some of the coauthors said they were consultants to numerous pharmaceutical companies or owned stock in pharmaceutical companies. Other than employment by INSERM on the part of Dr. Bernard, neither he nor Dr. Ebert had any relevant financial disclosures to report.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major Finding: The nine driver genes identified in this study appear in five core signaling pathways, and the genes each play different, though well-established, roles in these pathways, such as DNA repair and cell-cycle control, Notch signaling, inflammatory pathways, and RNA splicing (in which SF3B1, in particular, was implicated).
Data Source: Study using massively parallel sequencing of DNA samples from normal tissues and tumors of 91 patients with chronic lymphocytic leukemia.
Disclosures: This study was supported by grants from the National Institutes of Health, the National Cancer Institute, the Melton and Rosenbach Funds, the Blavatnik Family Foundation, the Howard Hughes Medical Institute (via an Early Career Physician-Scientist Award), and the Damon Runyon Cancer Research Foundation. Dr. Wang said she had no relevant financial disclosures. Some of the coauthors said they were consultants to numerous pharmaceutical companies or owned stock in pharmaceutical companies.