User login
Mitchel is a reporter for MDedge based in the Philadelphia area. He started with the company in 1992, when it was International Medical News Group (IMNG), and has since covered a range of medical specialties. Mitchel trained as a virologist at Roswell Park Memorial Institute in Buffalo, and then worked briefly as a researcher at Boston Children's Hospital before pivoting to journalism as a AAAS Mass Media Fellow in 1980. His first reporting job was with Science Digest magazine, and from the mid-1980s to early-1990s he was a reporter with Medical World News. @mitchelzoler
Digoxin linked to doubled mortality in atrial fib
SAN FRANCISCO – Starting digoxin treatment in patients with newly diagnosed atrial fibrillation was linked with a doubled risk of death during the subsequent 8 months, on the basis of an observation of more than 23,000 patients in a California health system.
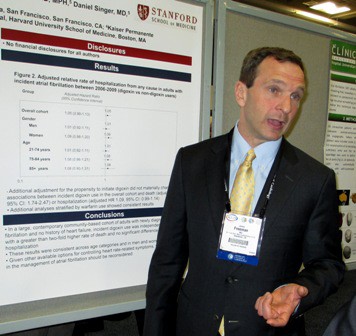
"This is a very big deal. We will tell Kaiser physicians not to prescribe digoxin to patients with atrial fibrillation," Dr. James V. Freeman said while presenting a poster at the annual meeting of the American College of Cardiology.
Although the mechanism is not known, he said he suspects it may involve arrhythmia generation and sudden death. The hospitalization rate among patients who began digoxin ran the same as in those not on the drug, even though mortality doubled.
While not widely used, digoxin received significant interest from the treating physicians in California during January 2006 through June 2009, with 13% of newly diagnosed atrial fibrillation patients receiving prescriptions. The analysis excluded patients who were already on digoxin at the time of their incident atrial fibrillation, so "these were not just physicians keeping patients on digoxin for years," Dr. Freeman said in an interview.
Existing recommendations from the ACC, last updated in 2012, list digoxin as one of eight drug choices for heart rate and rhythm control in atrial fibrillation patients. The college lists the eight drugs alphabetically and does not rate any drug as better or worse than the others.
The study presented by Dr. Freeman involved 23,272 adults aged 21 years or older with an index diagnosis of atrial fibrillation in the Kaiser Permanente Northern California and Southern California health plans during the 3.5-year period examined. Among these patients who had never used digoxin before and who were without heart failure, 2,997 filled at least one prescription for the drug.
During a median follow-up of 10 months, the mortality rate was 9.49/100 person-years of follow-up among those who received digoxin, versus 4.27/100 person-years in nonusers. After adjusting for a list of possible demographic and clinical confounders, the investigators found that patients who received digoxin had a 2.06 higher mortality rate, a statistically significant difference, reported Dr. Freeman, a cardiologist at Stanford (Calif.) University. Subgroup analyses showed similar increased mortality rates with digoxin use across both women and men and among patients younger than 75 years, 75-84, and 85 years or older.
The hospitalization rate was roughly 3 cases for every 100 person-years of follow-up in both groups.
Dr. Freeman conceded that as an observational study, the analysis was vulnerable to possible confounding factors, but in this case he thought that the risk of significant confounding was minimal. The Kaiser system collects voluminous data on comorbidities and other treatments that patients received, and the pattern showed a slight tilt toward sicker patients with more comorbidity among those who did not receive digoxin. For example, the nonusers had a 76% prevalence of hypertension, a 4% prevalence of prior coronary stenting, and a 4% rate of prior stroke, compared with prevalence rates of 74%, 3%, and 3%, respectively, among digoxin recipients. However, the digoxin recipients had slightly higher rates of cancer and lung disease, 7% compared with 6%, and 23% vs. 20%, respectively.
The patients not on digoxin also had slightly higher rates of medication with other potentially beneficial drugs. For example, treatment with an ACE inhibitor occurred in 31% of digoxin nonusers compared with 29% of the digoxin users; beta-blocker treatment occurred in 46% of digoxin nonusers, compared with 40% of the digoxin users.
Dr. Freeman said he had no relevant financial disclosures.
On Twitter @mitchelzoler
SAN FRANCISCO – Starting digoxin treatment in patients with newly diagnosed atrial fibrillation was linked with a doubled risk of death during the subsequent 8 months, on the basis of an observation of more than 23,000 patients in a California health system.
"This is a very big deal. We will tell Kaiser physicians not to prescribe digoxin to patients with atrial fibrillation," Dr. James V. Freeman said while presenting a poster at the annual meeting of the American College of Cardiology.
Although the mechanism is not known, he said he suspects it may involve arrhythmia generation and sudden death. The hospitalization rate among patients who began digoxin ran the same as in those not on the drug, even though mortality doubled.
While not widely used, digoxin received significant interest from the treating physicians in California during January 2006 through June 2009, with 13% of newly diagnosed atrial fibrillation patients receiving prescriptions. The analysis excluded patients who were already on digoxin at the time of their incident atrial fibrillation, so "these were not just physicians keeping patients on digoxin for years," Dr. Freeman said in an interview.
Existing recommendations from the ACC, last updated in 2012, list digoxin as one of eight drug choices for heart rate and rhythm control in atrial fibrillation patients. The college lists the eight drugs alphabetically and does not rate any drug as better or worse than the others.
The study presented by Dr. Freeman involved 23,272 adults aged 21 years or older with an index diagnosis of atrial fibrillation in the Kaiser Permanente Northern California and Southern California health plans during the 3.5-year period examined. Among these patients who had never used digoxin before and who were without heart failure, 2,997 filled at least one prescription for the drug.
During a median follow-up of 10 months, the mortality rate was 9.49/100 person-years of follow-up among those who received digoxin, versus 4.27/100 person-years in nonusers. After adjusting for a list of possible demographic and clinical confounders, the investigators found that patients who received digoxin had a 2.06 higher mortality rate, a statistically significant difference, reported Dr. Freeman, a cardiologist at Stanford (Calif.) University. Subgroup analyses showed similar increased mortality rates with digoxin use across both women and men and among patients younger than 75 years, 75-84, and 85 years or older.
The hospitalization rate was roughly 3 cases for every 100 person-years of follow-up in both groups.
Dr. Freeman conceded that as an observational study, the analysis was vulnerable to possible confounding factors, but in this case he thought that the risk of significant confounding was minimal. The Kaiser system collects voluminous data on comorbidities and other treatments that patients received, and the pattern showed a slight tilt toward sicker patients with more comorbidity among those who did not receive digoxin. For example, the nonusers had a 76% prevalence of hypertension, a 4% prevalence of prior coronary stenting, and a 4% rate of prior stroke, compared with prevalence rates of 74%, 3%, and 3%, respectively, among digoxin recipients. However, the digoxin recipients had slightly higher rates of cancer and lung disease, 7% compared with 6%, and 23% vs. 20%, respectively.
The patients not on digoxin also had slightly higher rates of medication with other potentially beneficial drugs. For example, treatment with an ACE inhibitor occurred in 31% of digoxin nonusers compared with 29% of the digoxin users; beta-blocker treatment occurred in 46% of digoxin nonusers, compared with 40% of the digoxin users.
Dr. Freeman said he had no relevant financial disclosures.
On Twitter @mitchelzoler
SAN FRANCISCO – Starting digoxin treatment in patients with newly diagnosed atrial fibrillation was linked with a doubled risk of death during the subsequent 8 months, on the basis of an observation of more than 23,000 patients in a California health system.
"This is a very big deal. We will tell Kaiser physicians not to prescribe digoxin to patients with atrial fibrillation," Dr. James V. Freeman said while presenting a poster at the annual meeting of the American College of Cardiology.
Although the mechanism is not known, he said he suspects it may involve arrhythmia generation and sudden death. The hospitalization rate among patients who began digoxin ran the same as in those not on the drug, even though mortality doubled.
While not widely used, digoxin received significant interest from the treating physicians in California during January 2006 through June 2009, with 13% of newly diagnosed atrial fibrillation patients receiving prescriptions. The analysis excluded patients who were already on digoxin at the time of their incident atrial fibrillation, so "these were not just physicians keeping patients on digoxin for years," Dr. Freeman said in an interview.
Existing recommendations from the ACC, last updated in 2012, list digoxin as one of eight drug choices for heart rate and rhythm control in atrial fibrillation patients. The college lists the eight drugs alphabetically and does not rate any drug as better or worse than the others.
The study presented by Dr. Freeman involved 23,272 adults aged 21 years or older with an index diagnosis of atrial fibrillation in the Kaiser Permanente Northern California and Southern California health plans during the 3.5-year period examined. Among these patients who had never used digoxin before and who were without heart failure, 2,997 filled at least one prescription for the drug.
During a median follow-up of 10 months, the mortality rate was 9.49/100 person-years of follow-up among those who received digoxin, versus 4.27/100 person-years in nonusers. After adjusting for a list of possible demographic and clinical confounders, the investigators found that patients who received digoxin had a 2.06 higher mortality rate, a statistically significant difference, reported Dr. Freeman, a cardiologist at Stanford (Calif.) University. Subgroup analyses showed similar increased mortality rates with digoxin use across both women and men and among patients younger than 75 years, 75-84, and 85 years or older.
The hospitalization rate was roughly 3 cases for every 100 person-years of follow-up in both groups.
Dr. Freeman conceded that as an observational study, the analysis was vulnerable to possible confounding factors, but in this case he thought that the risk of significant confounding was minimal. The Kaiser system collects voluminous data on comorbidities and other treatments that patients received, and the pattern showed a slight tilt toward sicker patients with more comorbidity among those who did not receive digoxin. For example, the nonusers had a 76% prevalence of hypertension, a 4% prevalence of prior coronary stenting, and a 4% rate of prior stroke, compared with prevalence rates of 74%, 3%, and 3%, respectively, among digoxin recipients. However, the digoxin recipients had slightly higher rates of cancer and lung disease, 7% compared with 6%, and 23% vs. 20%, respectively.
The patients not on digoxin also had slightly higher rates of medication with other potentially beneficial drugs. For example, treatment with an ACE inhibitor occurred in 31% of digoxin nonusers compared with 29% of the digoxin users; beta-blocker treatment occurred in 46% of digoxin nonusers, compared with 40% of the digoxin users.
Dr. Freeman said he had no relevant financial disclosures.
On Twitter @mitchelzoler
AT ACC 2013
Major finding: Newly diagnosed atrial fibrillation patients who received digoxin had twice the mortality of new a fib patients not on the drug.
Data source: An observational study of 23,272 patients with newly diagnosed atrial fibrillation and no history of digoxin use or heart failure in a U.S. health system during 2006-2009.
Disclosures: Dr. Freeman said he had no relevant financial disclosures.
New York's meningococcal disease outbreak remains contained
An outbreak of invasive meningococcal disease among men who have sex with men has raged on a small but impactful scale in New York City since 2010, but has shown no sign of spreading elsewhere, according to the epidemiologic assessment of the Centers for Disease Control and Prevention.
"There are no other [U.S.] outbreaks we know of," said Dr. Thomas A. Clark, a medical epidemiologist in the Meningitis and Vaccine-Preventable Diseases branch of the CDC "There are sporadic cases [in other U.S. locations], but outbreaks—clusters of cases--are rare. There has not been an experience in the past where an outbreak like this has spread elsewhere," Dr. Clark said in an interview.
"There have been similar clusters of meningococcal disease before, such as an outbreak in men who have sex with men (MSM) in Toronto. Because each meningococcal case is rare and has public health implications, there is a lot of communication about the cases among health departments" at the city and state level and the CDC, he said.
Awareness of the New York outbreak first surfaced last September, with a report from the city’s Department of Health and Mental Hygiene of 12 cases of invasive meningococcal disease (IMD) in New York starting in 2010 among MSM who were also infected with human immunodeficiency virus (HIV). Based on those cases, the Department recommended on Oct. 4, 2012, the meningococcal vaccine for all HIV-infected men who "had intimate contact with another man that he met through a website, digital application (‘App’), or at a bar or party since Sept. 1, 2012."
By early March, New York’s Department of Health focused its earlier vaccination recommendation and filled in some more data on the scope of the outbreak. In a March 6 alert issued to health care providers, the Department cited a total of 22 confirmed, recent cases in New York, all in MSM: 1 in 2010, 4 in 2011, 13 last year, and 4 during the first 2 months of 2013. Seventeen of the 22 cases had resided in Brooklyn or Manhattan. Cases ranged in age from 21 to 59 years; 50% had been black, 27% white, and 18% Hispanic. In addition, 7 of the 22 cases had died, including 3 of the 5 most-recent cases.
In the March alert, the DOH also revised its prior vaccination guidance, now targeting all HIV-infected MSM, and all MSM regardless of HIV status if they "regularly have close or intimate contact with men met through an online website, digital application (‘app’), or a bar or party." The Department noted that during October 2012-early March 2013, approximately 45% of the initial target population – HIV-infected MSM who also met the high-risk criteria – had received a first vaccine dose.
The relatively high, 32% mortality rate of the infections has the Department especially concerned. "We are worried about this outbreak because it can make people ill very quickly," said a Department spokesperson in an interview. "People can be protected by vaccination, but many who are at risk in this outbreak do not yet know they are at risk or do not believe that the risk is great enough to get vaccinated," the spokesperson added. The March 6 alert to providers urged them to recommend the vaccine to their targeted patients; the Department is administering meningococcal vaccine free at its clinics.
Although the 22 cases since 2010 may seem modest, it is enough to meet standards for a meningococcal outbreak, especially when the at-risk population is as tightly defined as New York has made it, commented Dr. Clark. He also agreed that vaccination was the best and only intervention.
"There are no modifiable risk factors" for meningococcal disease. "That’s why vaccination is best," he said. The only other prophylaxis is a short course of antibiotic for people identified as having been in close, household contact with a diagnosed case. The first-line antibiotic prophylaxis regimen is a single dose of ciprofloxacin, although a couple of other antibiotic alternatives are also possible.
The New York outbreak involves the serogroup C strain of Neisseria meningitidis (MMWR 2013;61:1048), which explains at least part of the reason why the outbreak has had a high mortality rate and prompted the Department of Health to refer to it as IMD. N. meningitidis serotype C has been linked to invasive infections by past reports (Epidemiol. Infect. 2006;134:1195-202). "It’s well documented that most if not all outbreaks of serogroup C have higher case-fatality rates," said Dr. Clark.
Coverage of the outbreak in the New York Times in March quoted Dr. Jay Varma, New York’s deputy commissioner for disease control, as saying IMD has been "sort of marching through the community in a way that makes us very scared."
Dr. Clark had no disclosures.
An outbreak of invasive meningococcal disease among men who have sex with men has raged on a small but impactful scale in New York City since 2010, but has shown no sign of spreading elsewhere, according to the epidemiologic assessment of the Centers for Disease Control and Prevention.
"There are no other [U.S.] outbreaks we know of," said Dr. Thomas A. Clark, a medical epidemiologist in the Meningitis and Vaccine-Preventable Diseases branch of the CDC "There are sporadic cases [in other U.S. locations], but outbreaks—clusters of cases--are rare. There has not been an experience in the past where an outbreak like this has spread elsewhere," Dr. Clark said in an interview.
"There have been similar clusters of meningococcal disease before, such as an outbreak in men who have sex with men (MSM) in Toronto. Because each meningococcal case is rare and has public health implications, there is a lot of communication about the cases among health departments" at the city and state level and the CDC, he said.
Awareness of the New York outbreak first surfaced last September, with a report from the city’s Department of Health and Mental Hygiene of 12 cases of invasive meningococcal disease (IMD) in New York starting in 2010 among MSM who were also infected with human immunodeficiency virus (HIV). Based on those cases, the Department recommended on Oct. 4, 2012, the meningococcal vaccine for all HIV-infected men who "had intimate contact with another man that he met through a website, digital application (‘App’), or at a bar or party since Sept. 1, 2012."
By early March, New York’s Department of Health focused its earlier vaccination recommendation and filled in some more data on the scope of the outbreak. In a March 6 alert issued to health care providers, the Department cited a total of 22 confirmed, recent cases in New York, all in MSM: 1 in 2010, 4 in 2011, 13 last year, and 4 during the first 2 months of 2013. Seventeen of the 22 cases had resided in Brooklyn or Manhattan. Cases ranged in age from 21 to 59 years; 50% had been black, 27% white, and 18% Hispanic. In addition, 7 of the 22 cases had died, including 3 of the 5 most-recent cases.
In the March alert, the DOH also revised its prior vaccination guidance, now targeting all HIV-infected MSM, and all MSM regardless of HIV status if they "regularly have close or intimate contact with men met through an online website, digital application (‘app’), or a bar or party." The Department noted that during October 2012-early March 2013, approximately 45% of the initial target population – HIV-infected MSM who also met the high-risk criteria – had received a first vaccine dose.
The relatively high, 32% mortality rate of the infections has the Department especially concerned. "We are worried about this outbreak because it can make people ill very quickly," said a Department spokesperson in an interview. "People can be protected by vaccination, but many who are at risk in this outbreak do not yet know they are at risk or do not believe that the risk is great enough to get vaccinated," the spokesperson added. The March 6 alert to providers urged them to recommend the vaccine to their targeted patients; the Department is administering meningococcal vaccine free at its clinics.
Although the 22 cases since 2010 may seem modest, it is enough to meet standards for a meningococcal outbreak, especially when the at-risk population is as tightly defined as New York has made it, commented Dr. Clark. He also agreed that vaccination was the best and only intervention.
"There are no modifiable risk factors" for meningococcal disease. "That’s why vaccination is best," he said. The only other prophylaxis is a short course of antibiotic for people identified as having been in close, household contact with a diagnosed case. The first-line antibiotic prophylaxis regimen is a single dose of ciprofloxacin, although a couple of other antibiotic alternatives are also possible.
The New York outbreak involves the serogroup C strain of Neisseria meningitidis (MMWR 2013;61:1048), which explains at least part of the reason why the outbreak has had a high mortality rate and prompted the Department of Health to refer to it as IMD. N. meningitidis serotype C has been linked to invasive infections by past reports (Epidemiol. Infect. 2006;134:1195-202). "It’s well documented that most if not all outbreaks of serogroup C have higher case-fatality rates," said Dr. Clark.
Coverage of the outbreak in the New York Times in March quoted Dr. Jay Varma, New York’s deputy commissioner for disease control, as saying IMD has been "sort of marching through the community in a way that makes us very scared."
Dr. Clark had no disclosures.
An outbreak of invasive meningococcal disease among men who have sex with men has raged on a small but impactful scale in New York City since 2010, but has shown no sign of spreading elsewhere, according to the epidemiologic assessment of the Centers for Disease Control and Prevention.
"There are no other [U.S.] outbreaks we know of," said Dr. Thomas A. Clark, a medical epidemiologist in the Meningitis and Vaccine-Preventable Diseases branch of the CDC "There are sporadic cases [in other U.S. locations], but outbreaks—clusters of cases--are rare. There has not been an experience in the past where an outbreak like this has spread elsewhere," Dr. Clark said in an interview.
"There have been similar clusters of meningococcal disease before, such as an outbreak in men who have sex with men (MSM) in Toronto. Because each meningococcal case is rare and has public health implications, there is a lot of communication about the cases among health departments" at the city and state level and the CDC, he said.
Awareness of the New York outbreak first surfaced last September, with a report from the city’s Department of Health and Mental Hygiene of 12 cases of invasive meningococcal disease (IMD) in New York starting in 2010 among MSM who were also infected with human immunodeficiency virus (HIV). Based on those cases, the Department recommended on Oct. 4, 2012, the meningococcal vaccine for all HIV-infected men who "had intimate contact with another man that he met through a website, digital application (‘App’), or at a bar or party since Sept. 1, 2012."
By early March, New York’s Department of Health focused its earlier vaccination recommendation and filled in some more data on the scope of the outbreak. In a March 6 alert issued to health care providers, the Department cited a total of 22 confirmed, recent cases in New York, all in MSM: 1 in 2010, 4 in 2011, 13 last year, and 4 during the first 2 months of 2013. Seventeen of the 22 cases had resided in Brooklyn or Manhattan. Cases ranged in age from 21 to 59 years; 50% had been black, 27% white, and 18% Hispanic. In addition, 7 of the 22 cases had died, including 3 of the 5 most-recent cases.
In the March alert, the DOH also revised its prior vaccination guidance, now targeting all HIV-infected MSM, and all MSM regardless of HIV status if they "regularly have close or intimate contact with men met through an online website, digital application (‘app’), or a bar or party." The Department noted that during October 2012-early March 2013, approximately 45% of the initial target population – HIV-infected MSM who also met the high-risk criteria – had received a first vaccine dose.
The relatively high, 32% mortality rate of the infections has the Department especially concerned. "We are worried about this outbreak because it can make people ill very quickly," said a Department spokesperson in an interview. "People can be protected by vaccination, but many who are at risk in this outbreak do not yet know they are at risk or do not believe that the risk is great enough to get vaccinated," the spokesperson added. The March 6 alert to providers urged them to recommend the vaccine to their targeted patients; the Department is administering meningococcal vaccine free at its clinics.
Although the 22 cases since 2010 may seem modest, it is enough to meet standards for a meningococcal outbreak, especially when the at-risk population is as tightly defined as New York has made it, commented Dr. Clark. He also agreed that vaccination was the best and only intervention.
"There are no modifiable risk factors" for meningococcal disease. "That’s why vaccination is best," he said. The only other prophylaxis is a short course of antibiotic for people identified as having been in close, household contact with a diagnosed case. The first-line antibiotic prophylaxis regimen is a single dose of ciprofloxacin, although a couple of other antibiotic alternatives are also possible.
The New York outbreak involves the serogroup C strain of Neisseria meningitidis (MMWR 2013;61:1048), which explains at least part of the reason why the outbreak has had a high mortality rate and prompted the Department of Health to refer to it as IMD. N. meningitidis serotype C has been linked to invasive infections by past reports (Epidemiol. Infect. 2006;134:1195-202). "It’s well documented that most if not all outbreaks of serogroup C have higher case-fatality rates," said Dr. Clark.
Coverage of the outbreak in the New York Times in March quoted Dr. Jay Varma, New York’s deputy commissioner for disease control, as saying IMD has been "sort of marching through the community in a way that makes us very scared."
Dr. Clark had no disclosures.
Widespread infection outbreaks pose biggest detection challenge
VIENNA – Widespread foodborne infectious-disease outbreaks pose the greatest challenge for detection, according to an analysis of 101 U.S. outbreaks during 1998-2008.
Two U.S. Salmonella outbreaks took 492 days and 251 days to detect, respectively, by far the longest lag from onset to detection of any of the outbreaks analyzed, said Heather Allen, Ph.D., at the International Meeting on Emerging Diseases and Surveillance.
In contrast, the entire group of 101 human or animal outbreaks during 1998-2008 with data available for analysis took a median 13 days to detect, and a mean 32 days, with more than three quarters of the outbreaks detected within 50 days, reported Dr. Allen, a public health analyst with LMI, a consulting company in McLean, Va. The results Dr. Allen reported came from a study she ran before becoming an LMI employee.
Median time to detection among outbreaks that extended beyond a single U.S. region was 35 days, compared with medians of 8-11 days for outbreaks confined to a single U.S. region (Northeast, Midwest, South, or West).
In cases of widespread foodborne outbreaks, "the cases trickle in and take a while for the number to build to where it is detected," Dr. Allen said.
The slow lag to report some foodborne U.S. outbreaks occurred despite use of best practices in those episodes, including PulseNet, a network of public health and food regulatory labs coordinated by the Centers for Disease Control and Prevention that apply molecular fingerprinting to isolates of Escherichia coli, Salmonella, and other foodborne pathogens.
Factors that may decrease reporting of infections and delay outbreak detection include awareness of the need to report, how busy practitioners are, the ease of reporting, and privacy concerns of patients or animal owners. The delay between a positive infection result and reporting it to a state or federal agent can be significant, and results in large differences in response times, Dr. Allen said in an interview.
"All human disease reporting to the federal level is voluntary; reporting to the state is governed by state laws and regulations."
Published records from sources such as Morbidity and Mortality Weekly Report and ProMED-Mail included 440 outbreaks during 1998-2008; Dr. Allen had data for analysis from 101 of these outbreaks. About two-thirds were in people, 16% in domestic animals, and 18% in people and animals. Initial reporting of the outbreak by a laboratory happened in 46% of the episodes, practitioners gave the initial report for 34%, and state agencies first reported 20%. State agencies had the longest average delay, 29 days, compared with roughly 10 days when either practitioners or labs made the first report.
Dr. Allen had no disclosures.
On Twitter @mitchelzoler
Detecting infectious outbreaks worldwide has gotten faster, but does that make a difference for outcomes? Although it makes intuitive sense that faster is better, right now we lack evidence to prove that.
My associates and I reported results in 2010 showing that the time to detection of infection outbreaks had dropped worldwide during 1996-2008 (Proc. Nat. Acad. Sci. 2010;107:21701-6). We studied records for 281 outbreaks verified by the World Health Organization during that period, and found that the median time from outbreak start to discovery fell from 30 days in 1996 to 14 days in 2008. Dr. Allen’s results were consistent with ours, and I think this trend is very real. We are definitely getting faster in identifying and reporting outbreaks. Part of that is likely because of improved communications and automated communications.

|
|
The problem with foodborne outbreaks is that often cases are not concentrated in one area, so you need to exceed the case threshold for the whole country before they are detected.
A big factor for outbreak detection is an astute primary-care physician who is on the lookout for an infection that doesn’t make sense. Something like severe respiratory illness in an otherwise healthy, young adult should raise a warning flag to call your local public health agency. Electronic reporting systems have been critical to reduced times for outbreak detection, but the best way to detect an outbreak is when someone recognizes something out of the ordinary, that doesn’t make sense.
Dr. Timothy F. Brewer is an infectious diseases physician and vice provost for interdisciplinary and cross-campus affairs at the University of California, Los Angeles. He said that he had no disclosures. He made these comments in an interview.
Detecting infectious outbreaks worldwide has gotten faster, but does that make a difference for outcomes? Although it makes intuitive sense that faster is better, right now we lack evidence to prove that.
My associates and I reported results in 2010 showing that the time to detection of infection outbreaks had dropped worldwide during 1996-2008 (Proc. Nat. Acad. Sci. 2010;107:21701-6). We studied records for 281 outbreaks verified by the World Health Organization during that period, and found that the median time from outbreak start to discovery fell from 30 days in 1996 to 14 days in 2008. Dr. Allen’s results were consistent with ours, and I think this trend is very real. We are definitely getting faster in identifying and reporting outbreaks. Part of that is likely because of improved communications and automated communications.

|
|
The problem with foodborne outbreaks is that often cases are not concentrated in one area, so you need to exceed the case threshold for the whole country before they are detected.
A big factor for outbreak detection is an astute primary-care physician who is on the lookout for an infection that doesn’t make sense. Something like severe respiratory illness in an otherwise healthy, young adult should raise a warning flag to call your local public health agency. Electronic reporting systems have been critical to reduced times for outbreak detection, but the best way to detect an outbreak is when someone recognizes something out of the ordinary, that doesn’t make sense.
Dr. Timothy F. Brewer is an infectious diseases physician and vice provost for interdisciplinary and cross-campus affairs at the University of California, Los Angeles. He said that he had no disclosures. He made these comments in an interview.
Detecting infectious outbreaks worldwide has gotten faster, but does that make a difference for outcomes? Although it makes intuitive sense that faster is better, right now we lack evidence to prove that.
My associates and I reported results in 2010 showing that the time to detection of infection outbreaks had dropped worldwide during 1996-2008 (Proc. Nat. Acad. Sci. 2010;107:21701-6). We studied records for 281 outbreaks verified by the World Health Organization during that period, and found that the median time from outbreak start to discovery fell from 30 days in 1996 to 14 days in 2008. Dr. Allen’s results were consistent with ours, and I think this trend is very real. We are definitely getting faster in identifying and reporting outbreaks. Part of that is likely because of improved communications and automated communications.

|
|
The problem with foodborne outbreaks is that often cases are not concentrated in one area, so you need to exceed the case threshold for the whole country before they are detected.
A big factor for outbreak detection is an astute primary-care physician who is on the lookout for an infection that doesn’t make sense. Something like severe respiratory illness in an otherwise healthy, young adult should raise a warning flag to call your local public health agency. Electronic reporting systems have been critical to reduced times for outbreak detection, but the best way to detect an outbreak is when someone recognizes something out of the ordinary, that doesn’t make sense.
Dr. Timothy F. Brewer is an infectious diseases physician and vice provost for interdisciplinary and cross-campus affairs at the University of California, Los Angeles. He said that he had no disclosures. He made these comments in an interview.
VIENNA – Widespread foodborne infectious-disease outbreaks pose the greatest challenge for detection, according to an analysis of 101 U.S. outbreaks during 1998-2008.
Two U.S. Salmonella outbreaks took 492 days and 251 days to detect, respectively, by far the longest lag from onset to detection of any of the outbreaks analyzed, said Heather Allen, Ph.D., at the International Meeting on Emerging Diseases and Surveillance.
In contrast, the entire group of 101 human or animal outbreaks during 1998-2008 with data available for analysis took a median 13 days to detect, and a mean 32 days, with more than three quarters of the outbreaks detected within 50 days, reported Dr. Allen, a public health analyst with LMI, a consulting company in McLean, Va. The results Dr. Allen reported came from a study she ran before becoming an LMI employee.
Median time to detection among outbreaks that extended beyond a single U.S. region was 35 days, compared with medians of 8-11 days for outbreaks confined to a single U.S. region (Northeast, Midwest, South, or West).
In cases of widespread foodborne outbreaks, "the cases trickle in and take a while for the number to build to where it is detected," Dr. Allen said.
The slow lag to report some foodborne U.S. outbreaks occurred despite use of best practices in those episodes, including PulseNet, a network of public health and food regulatory labs coordinated by the Centers for Disease Control and Prevention that apply molecular fingerprinting to isolates of Escherichia coli, Salmonella, and other foodborne pathogens.
Factors that may decrease reporting of infections and delay outbreak detection include awareness of the need to report, how busy practitioners are, the ease of reporting, and privacy concerns of patients or animal owners. The delay between a positive infection result and reporting it to a state or federal agent can be significant, and results in large differences in response times, Dr. Allen said in an interview.
"All human disease reporting to the federal level is voluntary; reporting to the state is governed by state laws and regulations."
Published records from sources such as Morbidity and Mortality Weekly Report and ProMED-Mail included 440 outbreaks during 1998-2008; Dr. Allen had data for analysis from 101 of these outbreaks. About two-thirds were in people, 16% in domestic animals, and 18% in people and animals. Initial reporting of the outbreak by a laboratory happened in 46% of the episodes, practitioners gave the initial report for 34%, and state agencies first reported 20%. State agencies had the longest average delay, 29 days, compared with roughly 10 days when either practitioners or labs made the first report.
Dr. Allen had no disclosures.
On Twitter @mitchelzoler
VIENNA – Widespread foodborne infectious-disease outbreaks pose the greatest challenge for detection, according to an analysis of 101 U.S. outbreaks during 1998-2008.
Two U.S. Salmonella outbreaks took 492 days and 251 days to detect, respectively, by far the longest lag from onset to detection of any of the outbreaks analyzed, said Heather Allen, Ph.D., at the International Meeting on Emerging Diseases and Surveillance.
In contrast, the entire group of 101 human or animal outbreaks during 1998-2008 with data available for analysis took a median 13 days to detect, and a mean 32 days, with more than three quarters of the outbreaks detected within 50 days, reported Dr. Allen, a public health analyst with LMI, a consulting company in McLean, Va. The results Dr. Allen reported came from a study she ran before becoming an LMI employee.
Median time to detection among outbreaks that extended beyond a single U.S. region was 35 days, compared with medians of 8-11 days for outbreaks confined to a single U.S. region (Northeast, Midwest, South, or West).
In cases of widespread foodborne outbreaks, "the cases trickle in and take a while for the number to build to where it is detected," Dr. Allen said.
The slow lag to report some foodborne U.S. outbreaks occurred despite use of best practices in those episodes, including PulseNet, a network of public health and food regulatory labs coordinated by the Centers for Disease Control and Prevention that apply molecular fingerprinting to isolates of Escherichia coli, Salmonella, and other foodborne pathogens.
Factors that may decrease reporting of infections and delay outbreak detection include awareness of the need to report, how busy practitioners are, the ease of reporting, and privacy concerns of patients or animal owners. The delay between a positive infection result and reporting it to a state or federal agent can be significant, and results in large differences in response times, Dr. Allen said in an interview.
"All human disease reporting to the federal level is voluntary; reporting to the state is governed by state laws and regulations."
Published records from sources such as Morbidity and Mortality Weekly Report and ProMED-Mail included 440 outbreaks during 1998-2008; Dr. Allen had data for analysis from 101 of these outbreaks. About two-thirds were in people, 16% in domestic animals, and 18% in people and animals. Initial reporting of the outbreak by a laboratory happened in 46% of the episodes, practitioners gave the initial report for 34%, and state agencies first reported 20%. State agencies had the longest average delay, 29 days, compared with roughly 10 days when either practitioners or labs made the first report.
Dr. Allen had no disclosures.
On Twitter @mitchelzoler
AT IMED2013
Major finding: Infection outbreaks in multiple U.S. regions took a median 35 days to detect compared with about 10 days for localized outbreaks.
Data source: Data came from analysis of published reports on 101 U.S. infection outbreaks during 1998-2008.
Disclosures: Dr. Allen had no disclosures.
FluNearYou spearheads U.S. participatory surveillance growth
VIENNA – Look for promotion of FluNearYou, a new form of crowd-source epidemiology first rolled out to the general U.S. public last September and now up to 50,000 active participants, to intensify next summer, said one of the program’s designers.
"Now that we are confident that people are interested and stay in the system, we will have a massive campaign next flu season to really build it out and see how far we can take it in the United States," Dr. Mark Smolinski said at the International Meeting on Emerging Diseases and Surveillance. FluNearYou represents the first phase of what will likely be a series of participatory surveillance tools that allow the general public to submit real-time infection epidemiology data.
"We’re thinking about engaging the public directly for all emerging infectious diseases, expanding to include more symptoms and extend the geography," said Dr. Smolinski, director of global health threats for the Skoll Global Threats Fund in San Francisco.
People who sign up for FluNearYou, via the Internet or an app, receive a weekly query that asks them to submit whether they have, in the last week, had any of 10 influenzalike symptoms (the query also asks participants if they received their seasonal flu vaccine as a reminder to do so). The FluNearYou software then analyzes the symptom information to determine whether the participant likely has flu and then maps information on probable cases, providing participants as well as public health officials a real-time map of flu prevalence at any time.
The goal is to "reduce the lag in identifying outbreaks so that everyone has access to the information at the same time," said John Brownstein, Ph.D., an epidemiologist at Children’s Hospital in Boston who has led development of Health Map, a web-based tool for tracking the status of infectious diseases worldwide. In a separate talk at the meeting, Dr. Brownstein stressed that new methods of crowd-sourced epidemiology are seen as complements rather than replacements for traditional mechanisms of infectious disease surveillance.
"We hope to continue to move the time to identify infection outbreaks down further – fast enough so that we can eliminate the threat of pandemics spreading around the world," Dr. Smolinski said. FluNearYou came about when Dr. Smolinski and his associates decided to "just ask people directly whether they have flulike symptoms."
FluNearYou debuted in 2011 with pilot testing among members of the American Public Health Association, which resulted in about 8,000 participants by early last fall. Last September, Dr. Smolinski and his associates at Skoll began publicizing the program in the San Francisco area, primarily through advertisements on public transit vehicles, which produced about 50,000 active participants by early 2013. Nationwide publicity and enrollment is the next step, he said.
Crowd-sourced epidemiology programs for flu are now in place in Europe as Influenzanet and in Australia, and the goal is to eventually merge these systems and have them cover the entire globe, Dr. Smolinski said. He also noted that the Centers for Disease Control and Prevention has launched a pilot study with a modified version of FluNearYou, with queries for a few additional symptoms, in Puerto Rico as a way to distinguish and track cases of influenza, dengue, and leptospirosis.
FluNearYou is sponsored by the Skoll Global Threats Fund and the Skoll Foundation. Dr. Smolinski is an employee of the fund. Dr. Brownstein had no disclosures.
VIENNA – Look for promotion of FluNearYou, a new form of crowd-source epidemiology first rolled out to the general U.S. public last September and now up to 50,000 active participants, to intensify next summer, said one of the program’s designers.
"Now that we are confident that people are interested and stay in the system, we will have a massive campaign next flu season to really build it out and see how far we can take it in the United States," Dr. Mark Smolinski said at the International Meeting on Emerging Diseases and Surveillance. FluNearYou represents the first phase of what will likely be a series of participatory surveillance tools that allow the general public to submit real-time infection epidemiology data.
"We’re thinking about engaging the public directly for all emerging infectious diseases, expanding to include more symptoms and extend the geography," said Dr. Smolinski, director of global health threats for the Skoll Global Threats Fund in San Francisco.
People who sign up for FluNearYou, via the Internet or an app, receive a weekly query that asks them to submit whether they have, in the last week, had any of 10 influenzalike symptoms (the query also asks participants if they received their seasonal flu vaccine as a reminder to do so). The FluNearYou software then analyzes the symptom information to determine whether the participant likely has flu and then maps information on probable cases, providing participants as well as public health officials a real-time map of flu prevalence at any time.
The goal is to "reduce the lag in identifying outbreaks so that everyone has access to the information at the same time," said John Brownstein, Ph.D., an epidemiologist at Children’s Hospital in Boston who has led development of Health Map, a web-based tool for tracking the status of infectious diseases worldwide. In a separate talk at the meeting, Dr. Brownstein stressed that new methods of crowd-sourced epidemiology are seen as complements rather than replacements for traditional mechanisms of infectious disease surveillance.
"We hope to continue to move the time to identify infection outbreaks down further – fast enough so that we can eliminate the threat of pandemics spreading around the world," Dr. Smolinski said. FluNearYou came about when Dr. Smolinski and his associates decided to "just ask people directly whether they have flulike symptoms."
FluNearYou debuted in 2011 with pilot testing among members of the American Public Health Association, which resulted in about 8,000 participants by early last fall. Last September, Dr. Smolinski and his associates at Skoll began publicizing the program in the San Francisco area, primarily through advertisements on public transit vehicles, which produced about 50,000 active participants by early 2013. Nationwide publicity and enrollment is the next step, he said.
Crowd-sourced epidemiology programs for flu are now in place in Europe as Influenzanet and in Australia, and the goal is to eventually merge these systems and have them cover the entire globe, Dr. Smolinski said. He also noted that the Centers for Disease Control and Prevention has launched a pilot study with a modified version of FluNearYou, with queries for a few additional symptoms, in Puerto Rico as a way to distinguish and track cases of influenza, dengue, and leptospirosis.
FluNearYou is sponsored by the Skoll Global Threats Fund and the Skoll Foundation. Dr. Smolinski is an employee of the fund. Dr. Brownstein had no disclosures.
VIENNA – Look for promotion of FluNearYou, a new form of crowd-source epidemiology first rolled out to the general U.S. public last September and now up to 50,000 active participants, to intensify next summer, said one of the program’s designers.
"Now that we are confident that people are interested and stay in the system, we will have a massive campaign next flu season to really build it out and see how far we can take it in the United States," Dr. Mark Smolinski said at the International Meeting on Emerging Diseases and Surveillance. FluNearYou represents the first phase of what will likely be a series of participatory surveillance tools that allow the general public to submit real-time infection epidemiology data.
"We’re thinking about engaging the public directly for all emerging infectious diseases, expanding to include more symptoms and extend the geography," said Dr. Smolinski, director of global health threats for the Skoll Global Threats Fund in San Francisco.
People who sign up for FluNearYou, via the Internet or an app, receive a weekly query that asks them to submit whether they have, in the last week, had any of 10 influenzalike symptoms (the query also asks participants if they received their seasonal flu vaccine as a reminder to do so). The FluNearYou software then analyzes the symptom information to determine whether the participant likely has flu and then maps information on probable cases, providing participants as well as public health officials a real-time map of flu prevalence at any time.
The goal is to "reduce the lag in identifying outbreaks so that everyone has access to the information at the same time," said John Brownstein, Ph.D., an epidemiologist at Children’s Hospital in Boston who has led development of Health Map, a web-based tool for tracking the status of infectious diseases worldwide. In a separate talk at the meeting, Dr. Brownstein stressed that new methods of crowd-sourced epidemiology are seen as complements rather than replacements for traditional mechanisms of infectious disease surveillance.
"We hope to continue to move the time to identify infection outbreaks down further – fast enough so that we can eliminate the threat of pandemics spreading around the world," Dr. Smolinski said. FluNearYou came about when Dr. Smolinski and his associates decided to "just ask people directly whether they have flulike symptoms."
FluNearYou debuted in 2011 with pilot testing among members of the American Public Health Association, which resulted in about 8,000 participants by early last fall. Last September, Dr. Smolinski and his associates at Skoll began publicizing the program in the San Francisco area, primarily through advertisements on public transit vehicles, which produced about 50,000 active participants by early 2013. Nationwide publicity and enrollment is the next step, he said.
Crowd-sourced epidemiology programs for flu are now in place in Europe as Influenzanet and in Australia, and the goal is to eventually merge these systems and have them cover the entire globe, Dr. Smolinski said. He also noted that the Centers for Disease Control and Prevention has launched a pilot study with a modified version of FluNearYou, with queries for a few additional symptoms, in Puerto Rico as a way to distinguish and track cases of influenza, dengue, and leptospirosis.
FluNearYou is sponsored by the Skoll Global Threats Fund and the Skoll Foundation. Dr. Smolinski is an employee of the fund. Dr. Brownstein had no disclosures.
AT IMED 2013
Major Finding: By February 2013, FluNearYou had about 50,000 active participants, up from about 8,000 in September 2012.
Data Source: Data came from FluNearYou.
Disclosures: FluNearYou is sponsored by the Skoll Global Threats Fund and the Skoll Foundation. Dr. Smolinski is an employee of the Ffund. Dr. Brownstein had no disclosures.
Cangrelor's success rose from prior failure
Designing a trial to establish the efficacy and safety of a drug may seem like science, but there is a lot of art as well. And sometimes achieving a positive trial result also requires confidence in and commitment to a concept, and the persistence to try a second time when it doesn’t work at first.
All of that happened in the unusual path that the intravenous antiplatelet drug cangrelor has navigated since researchers first devised trials in 2005 to test its efficacy in acute coronary syndrome patients who undergo coronary stenting. Arguably the most clinically important finding reported at the American College of Cardiology annual meeting earlier this month was from the CHAMPION PHOENIX trial, which showed safety and added efficacy from treatment with cangrelor, compared with clopidogrel, in this setting.
But while CHAMPION PHOENIX was a clear success, it was impossible to forget the failure from which it rose, a background that had everything to do with the trial’s myth-based name.
In 2009, results from a pair of cangrelor phase III trials, CHAMPION PCI and CHAMPION PLATFORM showed no added benefit from cangrelor in studies roughly similar in design to the PHOENIX study. The biggest difference between the success of PHOENIX and the failure of the earlier PCI and PLATFORM studies was that the researchers used a different approach to define and track the myocardial infarctions that patients developed during the studies and that served as primary end points.
Close analysis of the 2009 results from PCI and PLATFORM showed that, because many patients with acute coronary syndrome underwent coronary artery stenting so soon after becoming hospitalized, it was hard to discern new MI compared with prior events, said Dr. Robert Harrington, a Stanford (Calif.) University cardiologist and a coleader of all three studies. He, Dr. Deepak Bhatt (the other coleader), and their associates tweaked their original design three ways to address this problem, Dr. Harrington told me.
They obtained baseline measurements on the level of creatinine kinase MB in all patients, they took more frequent measures of CKMB from patients following baseline to track the development of new MIs, and they required, in addition to a further elevation in CKMB, some other indication of a new myocardial infarction: ischemic symptoms, ECG changes, or angiographic evidence. Along with their revised approach to detecting new-onset MIs, they also used a more refined definition of stent thrombosis within the first 48 hours of the study and set up the study presuming that the comparator drug, clopidogrel, would be more effective than they had anticipated the first time around.
These changes made the PHOENIX trial positive where the first two had failed.
Why did they do all this and keep at it so doggedly? Because they saw clear signals of efficacy in the two negative trials, and because they believed strongly in what a boon to cardiology practice would come from having a new drug available that interventionalists could quickly start in patients undergoing coronary catheterization and stenting but also quickly stop and clear from patients who were headed to coronary surgery following their diagnostic catheterization. That’s possible because the clinical effects from cangrelor persist for about an hour after an infusion stops, as opposed to days following a dose of an oral antiplatelet drug like clopidogrel, prasugrel, or ticagrelor.
"This drug will give us the flexibility to use a short-acting, intravenous agent," Dr. Bhatt said at the meeting. "We now give clopidogrel in the emergency department [to acute coronary syndrome patients] prior to knowing a patient’s coronary anatomy. If the patient has anatomy that needs urgent surgery, the patient has to wait a few days until the medicine clears out. By using cangrelor instead that uncomfortable situation disappears."
"The investigators thought that something was there [in CHAMPION PCI and PLATFORM], something useful for patients," added Dr. Bhatt of the Veterans Administration Boston Healthcare System.
Many interventionalists at the meeting agreed that cangrelor will bring an important new dimension of thrombotic protection and treatment flexibility to acute coronary syndrome patients undergoing coronary angiography.
"This is a blockbuster randomized controlled trial that will alter practice on numerous levels," said Dr. Gregg Stone, an interventional cardiologist at Columbia University in New York.
And it is "incredibly important for the treatment of patients undergoing catheterization who may need to have surgery," said Dr. Roxana Mehran, an interventionalist at Mount Sinai Hospital in New York.
CHAMPION "is a strikingly important study. It has global implications for the way we treat patients. The rapid on and off deals with a lot of clinical scenarios we face all the time," said Dr. Martin Leon, an interventionalist at Columbia University.
"Sometimes we need to wait 5-7 days before a patient can go to surgery because of the increased risk of bleeding" following treatment with an oral antiplatelet drug, said Dr. Cindy Grines, an interventionalist at Detroit Medical Center. "This will have a huge impact."
Dr. Bhatt, Dr. Mehran, and Dr. Grines reported financial ties with several pharmaceutical companies, including the Medicines Company, which is developing cangrelor. Dr. Harrington and Dr. Stone reported ties with numerous drug companies, including receiving a research grant from the Medicines Company. Dr. Leon disclosed having ties to two device makers but no pharmaceutical companies.
Designing a trial to establish the efficacy and safety of a drug may seem like science, but there is a lot of art as well. And sometimes achieving a positive trial result also requires confidence in and commitment to a concept, and the persistence to try a second time when it doesn’t work at first.
All of that happened in the unusual path that the intravenous antiplatelet drug cangrelor has navigated since researchers first devised trials in 2005 to test its efficacy in acute coronary syndrome patients who undergo coronary stenting. Arguably the most clinically important finding reported at the American College of Cardiology annual meeting earlier this month was from the CHAMPION PHOENIX trial, which showed safety and added efficacy from treatment with cangrelor, compared with clopidogrel, in this setting.
But while CHAMPION PHOENIX was a clear success, it was impossible to forget the failure from which it rose, a background that had everything to do with the trial’s myth-based name.
In 2009, results from a pair of cangrelor phase III trials, CHAMPION PCI and CHAMPION PLATFORM showed no added benefit from cangrelor in studies roughly similar in design to the PHOENIX study. The biggest difference between the success of PHOENIX and the failure of the earlier PCI and PLATFORM studies was that the researchers used a different approach to define and track the myocardial infarctions that patients developed during the studies and that served as primary end points.
Close analysis of the 2009 results from PCI and PLATFORM showed that, because many patients with acute coronary syndrome underwent coronary artery stenting so soon after becoming hospitalized, it was hard to discern new MI compared with prior events, said Dr. Robert Harrington, a Stanford (Calif.) University cardiologist and a coleader of all three studies. He, Dr. Deepak Bhatt (the other coleader), and their associates tweaked their original design three ways to address this problem, Dr. Harrington told me.
They obtained baseline measurements on the level of creatinine kinase MB in all patients, they took more frequent measures of CKMB from patients following baseline to track the development of new MIs, and they required, in addition to a further elevation in CKMB, some other indication of a new myocardial infarction: ischemic symptoms, ECG changes, or angiographic evidence. Along with their revised approach to detecting new-onset MIs, they also used a more refined definition of stent thrombosis within the first 48 hours of the study and set up the study presuming that the comparator drug, clopidogrel, would be more effective than they had anticipated the first time around.
These changes made the PHOENIX trial positive where the first two had failed.
Why did they do all this and keep at it so doggedly? Because they saw clear signals of efficacy in the two negative trials, and because they believed strongly in what a boon to cardiology practice would come from having a new drug available that interventionalists could quickly start in patients undergoing coronary catheterization and stenting but also quickly stop and clear from patients who were headed to coronary surgery following their diagnostic catheterization. That’s possible because the clinical effects from cangrelor persist for about an hour after an infusion stops, as opposed to days following a dose of an oral antiplatelet drug like clopidogrel, prasugrel, or ticagrelor.
"This drug will give us the flexibility to use a short-acting, intravenous agent," Dr. Bhatt said at the meeting. "We now give clopidogrel in the emergency department [to acute coronary syndrome patients] prior to knowing a patient’s coronary anatomy. If the patient has anatomy that needs urgent surgery, the patient has to wait a few days until the medicine clears out. By using cangrelor instead that uncomfortable situation disappears."
"The investigators thought that something was there [in CHAMPION PCI and PLATFORM], something useful for patients," added Dr. Bhatt of the Veterans Administration Boston Healthcare System.
Many interventionalists at the meeting agreed that cangrelor will bring an important new dimension of thrombotic protection and treatment flexibility to acute coronary syndrome patients undergoing coronary angiography.
"This is a blockbuster randomized controlled trial that will alter practice on numerous levels," said Dr. Gregg Stone, an interventional cardiologist at Columbia University in New York.
And it is "incredibly important for the treatment of patients undergoing catheterization who may need to have surgery," said Dr. Roxana Mehran, an interventionalist at Mount Sinai Hospital in New York.
CHAMPION "is a strikingly important study. It has global implications for the way we treat patients. The rapid on and off deals with a lot of clinical scenarios we face all the time," said Dr. Martin Leon, an interventionalist at Columbia University.
"Sometimes we need to wait 5-7 days before a patient can go to surgery because of the increased risk of bleeding" following treatment with an oral antiplatelet drug, said Dr. Cindy Grines, an interventionalist at Detroit Medical Center. "This will have a huge impact."
Dr. Bhatt, Dr. Mehran, and Dr. Grines reported financial ties with several pharmaceutical companies, including the Medicines Company, which is developing cangrelor. Dr. Harrington and Dr. Stone reported ties with numerous drug companies, including receiving a research grant from the Medicines Company. Dr. Leon disclosed having ties to two device makers but no pharmaceutical companies.
Designing a trial to establish the efficacy and safety of a drug may seem like science, but there is a lot of art as well. And sometimes achieving a positive trial result also requires confidence in and commitment to a concept, and the persistence to try a second time when it doesn’t work at first.
All of that happened in the unusual path that the intravenous antiplatelet drug cangrelor has navigated since researchers first devised trials in 2005 to test its efficacy in acute coronary syndrome patients who undergo coronary stenting. Arguably the most clinically important finding reported at the American College of Cardiology annual meeting earlier this month was from the CHAMPION PHOENIX trial, which showed safety and added efficacy from treatment with cangrelor, compared with clopidogrel, in this setting.
But while CHAMPION PHOENIX was a clear success, it was impossible to forget the failure from which it rose, a background that had everything to do with the trial’s myth-based name.
In 2009, results from a pair of cangrelor phase III trials, CHAMPION PCI and CHAMPION PLATFORM showed no added benefit from cangrelor in studies roughly similar in design to the PHOENIX study. The biggest difference between the success of PHOENIX and the failure of the earlier PCI and PLATFORM studies was that the researchers used a different approach to define and track the myocardial infarctions that patients developed during the studies and that served as primary end points.
Close analysis of the 2009 results from PCI and PLATFORM showed that, because many patients with acute coronary syndrome underwent coronary artery stenting so soon after becoming hospitalized, it was hard to discern new MI compared with prior events, said Dr. Robert Harrington, a Stanford (Calif.) University cardiologist and a coleader of all three studies. He, Dr. Deepak Bhatt (the other coleader), and their associates tweaked their original design three ways to address this problem, Dr. Harrington told me.
They obtained baseline measurements on the level of creatinine kinase MB in all patients, they took more frequent measures of CKMB from patients following baseline to track the development of new MIs, and they required, in addition to a further elevation in CKMB, some other indication of a new myocardial infarction: ischemic symptoms, ECG changes, or angiographic evidence. Along with their revised approach to detecting new-onset MIs, they also used a more refined definition of stent thrombosis within the first 48 hours of the study and set up the study presuming that the comparator drug, clopidogrel, would be more effective than they had anticipated the first time around.
These changes made the PHOENIX trial positive where the first two had failed.
Why did they do all this and keep at it so doggedly? Because they saw clear signals of efficacy in the two negative trials, and because they believed strongly in what a boon to cardiology practice would come from having a new drug available that interventionalists could quickly start in patients undergoing coronary catheterization and stenting but also quickly stop and clear from patients who were headed to coronary surgery following their diagnostic catheterization. That’s possible because the clinical effects from cangrelor persist for about an hour after an infusion stops, as opposed to days following a dose of an oral antiplatelet drug like clopidogrel, prasugrel, or ticagrelor.
"This drug will give us the flexibility to use a short-acting, intravenous agent," Dr. Bhatt said at the meeting. "We now give clopidogrel in the emergency department [to acute coronary syndrome patients] prior to knowing a patient’s coronary anatomy. If the patient has anatomy that needs urgent surgery, the patient has to wait a few days until the medicine clears out. By using cangrelor instead that uncomfortable situation disappears."
"The investigators thought that something was there [in CHAMPION PCI and PLATFORM], something useful for patients," added Dr. Bhatt of the Veterans Administration Boston Healthcare System.
Many interventionalists at the meeting agreed that cangrelor will bring an important new dimension of thrombotic protection and treatment flexibility to acute coronary syndrome patients undergoing coronary angiography.
"This is a blockbuster randomized controlled trial that will alter practice on numerous levels," said Dr. Gregg Stone, an interventional cardiologist at Columbia University in New York.
And it is "incredibly important for the treatment of patients undergoing catheterization who may need to have surgery," said Dr. Roxana Mehran, an interventionalist at Mount Sinai Hospital in New York.
CHAMPION "is a strikingly important study. It has global implications for the way we treat patients. The rapid on and off deals with a lot of clinical scenarios we face all the time," said Dr. Martin Leon, an interventionalist at Columbia University.
"Sometimes we need to wait 5-7 days before a patient can go to surgery because of the increased risk of bleeding" following treatment with an oral antiplatelet drug, said Dr. Cindy Grines, an interventionalist at Detroit Medical Center. "This will have a huge impact."
Dr. Bhatt, Dr. Mehran, and Dr. Grines reported financial ties with several pharmaceutical companies, including the Medicines Company, which is developing cangrelor. Dr. Harrington and Dr. Stone reported ties with numerous drug companies, including receiving a research grant from the Medicines Company. Dr. Leon disclosed having ties to two device makers but no pharmaceutical companies.
Sapien XT valve shows improvements over original
SAN FRANCISCO – A new, smaller version of the only transcatheter aortic valve available for U.S. use showed noninferior clinical outcomes and safer periprocedural results in a multicenter U.S. comparison of the new and existing devices in a total of 560 patients, raising expectations that the new valve system – the Sapien XT – will soon be on the U.S. market.
Although treatment using the Sapien XT roughly matched treatment with the existing Sapien transcatheter aortic valve in the incidence of the important adverse effect of residual moderate or severe paravalvular leak, several U.S. operators who perform transcatheter aortic valve repair (TAVR) stressed that the leak problem has begun to resolve recently as interventionalists and surgeons have found ways to minimize the issue. They also noted that availability of the new XT device will move the field a step further toward fewer leak issues, because it will allow the option of a larger, 29-mm-diameter valve, a possibility that has not been available with the original Sapien valve.
"Sapien XT represents a worthwhile advance with incremental clinical value, and it is the preferred balloon-expandable transcatheter aortic valve system," said Dr. Martin B. Leon when he presented the trial data on March 10 at the annual meeting of the American College of Cardiology.
"This second-generation device has demonstrated sufficient clinical benefit for us to say it’s the preferred therapy," said Dr. Leon, professor of medicine and director of the Center for Interventional Vascular Therapy at Columbia University in New York.
"This is an evolutionary technology that is easier to use. The results are superb," commented Dr. Gary S. Mintz, an interventional cardiologist based in Washington and medical director of the Cardiovascular Research Foundation in New York.
The PARTNER II (Placement of Aortic Transcatheter Valves) cohort B trial enrolled 560 patients with aortic stenosis deemed inoperable for surgical valve replacement at 28 U.S. centers during April 2011 through February 2012. The study randomized patients to receive the Sapien XT valve or the approved Sapien valve in a prespecified noninferiority design, with a primary outcome of all-cause death, disabling stroke, or need for repeat hospitalization at 1 year after treatment. Patients averaged 84 years old, they had an average Society of Thoracic Surgeons score of about 10%, 96% had New York Heart Association class III or IV heart failure, and about 60% met the study’s criteria for frailty.
At 1-year follow-up, the combined primary endpoint occurred in 35% of patients treated with the approved valve and in 34% of patients treated with the new XT valve, a result that was not statistically significant and that met the study’s standard for noninferiority. At 1 year, as well as at 30 days after treatment, the two patient groups showed no significant difference for any of the individual components of the clinical endpoints.
However, patients treated with the XT valve had significantly fewer procedural complications, with significantly fewer major vascular events (10% with the XT device, compared with 16% with the approved valve) and significantly fewer disabling bleeding events (8% with the XT device, compared with 13% with the first-generation system), Dr. Leon reported. The category of major vascular events included perforations, dissections, and hematomas. Patients treated with the XT device also required significantly fewer episodes when the initially placed valve had to be replaced (3 replaced valve implants, compared with 10 using the approved valve), and numerically fewer aborted procedures – 2 compared with 8 using the approved valve, although this difference just missed statistical significance (P = .06). Placement of the new valve also required an average of 14 minutes less anesthesia time, a significant difference.
"It’s a smaller device. What you would expect from a smaller device is fewer vascular complications and improved procedural events," Dr. Leon said in an interview. He also highlighted the numerical difference in 30-day mortality – 5.1% with the older valve system, compared with 3.5% using the XT device. While the difference did not reach statistical significance, "to me this difference is not trivial," he said.
The difference in disabling bleeding complications is also important, commented Dr. Mintz, who was not involved in the study. "Bleeding complications are hugely disabling, especially in this older, frail population," he said.
"The big concerns from the first PARTNER trial were vascular complications, strokes, and paravalvular leaks," said Dr. Leon. "The XT device seems to address some of these issues. The vascular complications were reduced, and the stroke rate went down" compared with the first PARTNER trial (N. Engl. J. Med. 2010;363:1597-607). "What is left is the paravalvular leak issue, which is real."
In the new trial, moderate or severe paravalvular leaks, a complication that has previously been linked with worse outcomes and increased mortality, occurred in 17% of the patients who received the approved valve and in 24% of patients who received the XT valve at 30-day follow-up, a difference that did not reach significance.
"Is there a difference in paravalvular leaks between Sapien XT and Sapien? We’re not entirely sure. There was a nonsignificant trend" that will be the subject of further analysis, Dr. Leon said. He stressed that the leak rate is confounded because significantly more patients who received the approved valve underwent a second valve placement, which often occurs because a leak occurred with the first valve. "Putting in a second valve is a big thing and is not good," he said. "No matter how you look at it, there still was a significant leak rate that is not as good as it needs to be." Dr. Leon said that he hopes the issue of paravalvular leaks will be addressed by a third-generation valve system that will enter clinical trials later this year.
But some surgeons and interventionalists involved in the PARTNER II trial noted that the incidence of paravalvular leaks has already begun to come down in the past year or so.
"We are [now] much better at sizing the valve with CT scans, and that is reducing the number of paravalvular leaks, at least anecdotally at our center," said Dr. Michael J. Mack, a cardiothoracic surgeon at the Heart Hospital in Plano, Texas, and a PARTNER II investigator. "We also underfill or overfill our [valve expansion] balloons based on the CT scans, and we’re getting better results with that. We’re fitting the valve better into the space," he said in an interview.
"We do everything we can to minimize patients leaving with a moderate or severe leak by balloon dilatation or putting in a second valve," said Dr. Jeffrey J. Popma, an interventional cardiologist and professor of medicine at Harvard University in Boston and a PARTNER II investigator. He also noted that use of the XT device reduces the number of patients who need transapical valve placement, because the smaller catheter can be more easily manipulated through a patient’s ileofemoral anatomy.
A further advantage of the XT valve for minimizing paravalvular leaks is that it is available in a 29-mm diameter, although the 29-mm valve was not included in the PARTNER II trial. The widest valve available in the original Sapien design was 26 mm.
"The 29-mm valve is a huge advance because we know that [until now] TAVR success is greater in women, probably because about 20% of American men need a larger valve, and their leak rate was higher," said Dr. Joseph E. Bavaria, a professor of surgery at the University of Pennsylvania in Philadelphia and a PARTNER II investigator. "Having the 29-mm valve available will be a big difference," he said in an interview.
The PARTNER II trial has a second cohort, cohort A, that is comparing the Sapien XT valve system against open surgical valve replacement in patients who are deemed eligible by cardiac surgeons for surgical aortic valve replacement and are at "intermediate" risk for undergoing surgery. Enrollment of the roughly 2,000 patients who will enter this study should be complete by this summer, and with a 2-year primary endpoint the results should be available sometime in 2015, Dr. Leon said.
The PARTNER II trial is sponsored by Edwards Lifesciences, which markets the XT transcatheter aortic valve system. The company has not filed an application for approval of the Sapien XT device. Dr. Leon, Dr. Mack, Dr. Popma, and Dr. Bavaria all participated in the trial. Dr. Mintz said that he has been a consultant to Boston Scientific.
SAN FRANCISCO – A new, smaller version of the only transcatheter aortic valve available for U.S. use showed noninferior clinical outcomes and safer periprocedural results in a multicenter U.S. comparison of the new and existing devices in a total of 560 patients, raising expectations that the new valve system – the Sapien XT – will soon be on the U.S. market.
Although treatment using the Sapien XT roughly matched treatment with the existing Sapien transcatheter aortic valve in the incidence of the important adverse effect of residual moderate or severe paravalvular leak, several U.S. operators who perform transcatheter aortic valve repair (TAVR) stressed that the leak problem has begun to resolve recently as interventionalists and surgeons have found ways to minimize the issue. They also noted that availability of the new XT device will move the field a step further toward fewer leak issues, because it will allow the option of a larger, 29-mm-diameter valve, a possibility that has not been available with the original Sapien valve.
"Sapien XT represents a worthwhile advance with incremental clinical value, and it is the preferred balloon-expandable transcatheter aortic valve system," said Dr. Martin B. Leon when he presented the trial data on March 10 at the annual meeting of the American College of Cardiology.
"This second-generation device has demonstrated sufficient clinical benefit for us to say it’s the preferred therapy," said Dr. Leon, professor of medicine and director of the Center for Interventional Vascular Therapy at Columbia University in New York.
"This is an evolutionary technology that is easier to use. The results are superb," commented Dr. Gary S. Mintz, an interventional cardiologist based in Washington and medical director of the Cardiovascular Research Foundation in New York.
The PARTNER II (Placement of Aortic Transcatheter Valves) cohort B trial enrolled 560 patients with aortic stenosis deemed inoperable for surgical valve replacement at 28 U.S. centers during April 2011 through February 2012. The study randomized patients to receive the Sapien XT valve or the approved Sapien valve in a prespecified noninferiority design, with a primary outcome of all-cause death, disabling stroke, or need for repeat hospitalization at 1 year after treatment. Patients averaged 84 years old, they had an average Society of Thoracic Surgeons score of about 10%, 96% had New York Heart Association class III or IV heart failure, and about 60% met the study’s criteria for frailty.
At 1-year follow-up, the combined primary endpoint occurred in 35% of patients treated with the approved valve and in 34% of patients treated with the new XT valve, a result that was not statistically significant and that met the study’s standard for noninferiority. At 1 year, as well as at 30 days after treatment, the two patient groups showed no significant difference for any of the individual components of the clinical endpoints.
However, patients treated with the XT valve had significantly fewer procedural complications, with significantly fewer major vascular events (10% with the XT device, compared with 16% with the approved valve) and significantly fewer disabling bleeding events (8% with the XT device, compared with 13% with the first-generation system), Dr. Leon reported. The category of major vascular events included perforations, dissections, and hematomas. Patients treated with the XT device also required significantly fewer episodes when the initially placed valve had to be replaced (3 replaced valve implants, compared with 10 using the approved valve), and numerically fewer aborted procedures – 2 compared with 8 using the approved valve, although this difference just missed statistical significance (P = .06). Placement of the new valve also required an average of 14 minutes less anesthesia time, a significant difference.
"It’s a smaller device. What you would expect from a smaller device is fewer vascular complications and improved procedural events," Dr. Leon said in an interview. He also highlighted the numerical difference in 30-day mortality – 5.1% with the older valve system, compared with 3.5% using the XT device. While the difference did not reach statistical significance, "to me this difference is not trivial," he said.
The difference in disabling bleeding complications is also important, commented Dr. Mintz, who was not involved in the study. "Bleeding complications are hugely disabling, especially in this older, frail population," he said.
"The big concerns from the first PARTNER trial were vascular complications, strokes, and paravalvular leaks," said Dr. Leon. "The XT device seems to address some of these issues. The vascular complications were reduced, and the stroke rate went down" compared with the first PARTNER trial (N. Engl. J. Med. 2010;363:1597-607). "What is left is the paravalvular leak issue, which is real."
In the new trial, moderate or severe paravalvular leaks, a complication that has previously been linked with worse outcomes and increased mortality, occurred in 17% of the patients who received the approved valve and in 24% of patients who received the XT valve at 30-day follow-up, a difference that did not reach significance.
"Is there a difference in paravalvular leaks between Sapien XT and Sapien? We’re not entirely sure. There was a nonsignificant trend" that will be the subject of further analysis, Dr. Leon said. He stressed that the leak rate is confounded because significantly more patients who received the approved valve underwent a second valve placement, which often occurs because a leak occurred with the first valve. "Putting in a second valve is a big thing and is not good," he said. "No matter how you look at it, there still was a significant leak rate that is not as good as it needs to be." Dr. Leon said that he hopes the issue of paravalvular leaks will be addressed by a third-generation valve system that will enter clinical trials later this year.
But some surgeons and interventionalists involved in the PARTNER II trial noted that the incidence of paravalvular leaks has already begun to come down in the past year or so.
"We are [now] much better at sizing the valve with CT scans, and that is reducing the number of paravalvular leaks, at least anecdotally at our center," said Dr. Michael J. Mack, a cardiothoracic surgeon at the Heart Hospital in Plano, Texas, and a PARTNER II investigator. "We also underfill or overfill our [valve expansion] balloons based on the CT scans, and we’re getting better results with that. We’re fitting the valve better into the space," he said in an interview.
"We do everything we can to minimize patients leaving with a moderate or severe leak by balloon dilatation or putting in a second valve," said Dr. Jeffrey J. Popma, an interventional cardiologist and professor of medicine at Harvard University in Boston and a PARTNER II investigator. He also noted that use of the XT device reduces the number of patients who need transapical valve placement, because the smaller catheter can be more easily manipulated through a patient’s ileofemoral anatomy.
A further advantage of the XT valve for minimizing paravalvular leaks is that it is available in a 29-mm diameter, although the 29-mm valve was not included in the PARTNER II trial. The widest valve available in the original Sapien design was 26 mm.
"The 29-mm valve is a huge advance because we know that [until now] TAVR success is greater in women, probably because about 20% of American men need a larger valve, and their leak rate was higher," said Dr. Joseph E. Bavaria, a professor of surgery at the University of Pennsylvania in Philadelphia and a PARTNER II investigator. "Having the 29-mm valve available will be a big difference," he said in an interview.
The PARTNER II trial has a second cohort, cohort A, that is comparing the Sapien XT valve system against open surgical valve replacement in patients who are deemed eligible by cardiac surgeons for surgical aortic valve replacement and are at "intermediate" risk for undergoing surgery. Enrollment of the roughly 2,000 patients who will enter this study should be complete by this summer, and with a 2-year primary endpoint the results should be available sometime in 2015, Dr. Leon said.
The PARTNER II trial is sponsored by Edwards Lifesciences, which markets the XT transcatheter aortic valve system. The company has not filed an application for approval of the Sapien XT device. Dr. Leon, Dr. Mack, Dr. Popma, and Dr. Bavaria all participated in the trial. Dr. Mintz said that he has been a consultant to Boston Scientific.
SAN FRANCISCO – A new, smaller version of the only transcatheter aortic valve available for U.S. use showed noninferior clinical outcomes and safer periprocedural results in a multicenter U.S. comparison of the new and existing devices in a total of 560 patients, raising expectations that the new valve system – the Sapien XT – will soon be on the U.S. market.
Although treatment using the Sapien XT roughly matched treatment with the existing Sapien transcatheter aortic valve in the incidence of the important adverse effect of residual moderate or severe paravalvular leak, several U.S. operators who perform transcatheter aortic valve repair (TAVR) stressed that the leak problem has begun to resolve recently as interventionalists and surgeons have found ways to minimize the issue. They also noted that availability of the new XT device will move the field a step further toward fewer leak issues, because it will allow the option of a larger, 29-mm-diameter valve, a possibility that has not been available with the original Sapien valve.
"Sapien XT represents a worthwhile advance with incremental clinical value, and it is the preferred balloon-expandable transcatheter aortic valve system," said Dr. Martin B. Leon when he presented the trial data on March 10 at the annual meeting of the American College of Cardiology.
"This second-generation device has demonstrated sufficient clinical benefit for us to say it’s the preferred therapy," said Dr. Leon, professor of medicine and director of the Center for Interventional Vascular Therapy at Columbia University in New York.
"This is an evolutionary technology that is easier to use. The results are superb," commented Dr. Gary S. Mintz, an interventional cardiologist based in Washington and medical director of the Cardiovascular Research Foundation in New York.
The PARTNER II (Placement of Aortic Transcatheter Valves) cohort B trial enrolled 560 patients with aortic stenosis deemed inoperable for surgical valve replacement at 28 U.S. centers during April 2011 through February 2012. The study randomized patients to receive the Sapien XT valve or the approved Sapien valve in a prespecified noninferiority design, with a primary outcome of all-cause death, disabling stroke, or need for repeat hospitalization at 1 year after treatment. Patients averaged 84 years old, they had an average Society of Thoracic Surgeons score of about 10%, 96% had New York Heart Association class III or IV heart failure, and about 60% met the study’s criteria for frailty.
At 1-year follow-up, the combined primary endpoint occurred in 35% of patients treated with the approved valve and in 34% of patients treated with the new XT valve, a result that was not statistically significant and that met the study’s standard for noninferiority. At 1 year, as well as at 30 days after treatment, the two patient groups showed no significant difference for any of the individual components of the clinical endpoints.
However, patients treated with the XT valve had significantly fewer procedural complications, with significantly fewer major vascular events (10% with the XT device, compared with 16% with the approved valve) and significantly fewer disabling bleeding events (8% with the XT device, compared with 13% with the first-generation system), Dr. Leon reported. The category of major vascular events included perforations, dissections, and hematomas. Patients treated with the XT device also required significantly fewer episodes when the initially placed valve had to be replaced (3 replaced valve implants, compared with 10 using the approved valve), and numerically fewer aborted procedures – 2 compared with 8 using the approved valve, although this difference just missed statistical significance (P = .06). Placement of the new valve also required an average of 14 minutes less anesthesia time, a significant difference.
"It’s a smaller device. What you would expect from a smaller device is fewer vascular complications and improved procedural events," Dr. Leon said in an interview. He also highlighted the numerical difference in 30-day mortality – 5.1% with the older valve system, compared with 3.5% using the XT device. While the difference did not reach statistical significance, "to me this difference is not trivial," he said.
The difference in disabling bleeding complications is also important, commented Dr. Mintz, who was not involved in the study. "Bleeding complications are hugely disabling, especially in this older, frail population," he said.
"The big concerns from the first PARTNER trial were vascular complications, strokes, and paravalvular leaks," said Dr. Leon. "The XT device seems to address some of these issues. The vascular complications were reduced, and the stroke rate went down" compared with the first PARTNER trial (N. Engl. J. Med. 2010;363:1597-607). "What is left is the paravalvular leak issue, which is real."
In the new trial, moderate or severe paravalvular leaks, a complication that has previously been linked with worse outcomes and increased mortality, occurred in 17% of the patients who received the approved valve and in 24% of patients who received the XT valve at 30-day follow-up, a difference that did not reach significance.
"Is there a difference in paravalvular leaks between Sapien XT and Sapien? We’re not entirely sure. There was a nonsignificant trend" that will be the subject of further analysis, Dr. Leon said. He stressed that the leak rate is confounded because significantly more patients who received the approved valve underwent a second valve placement, which often occurs because a leak occurred with the first valve. "Putting in a second valve is a big thing and is not good," he said. "No matter how you look at it, there still was a significant leak rate that is not as good as it needs to be." Dr. Leon said that he hopes the issue of paravalvular leaks will be addressed by a third-generation valve system that will enter clinical trials later this year.
But some surgeons and interventionalists involved in the PARTNER II trial noted that the incidence of paravalvular leaks has already begun to come down in the past year or so.
"We are [now] much better at sizing the valve with CT scans, and that is reducing the number of paravalvular leaks, at least anecdotally at our center," said Dr. Michael J. Mack, a cardiothoracic surgeon at the Heart Hospital in Plano, Texas, and a PARTNER II investigator. "We also underfill or overfill our [valve expansion] balloons based on the CT scans, and we’re getting better results with that. We’re fitting the valve better into the space," he said in an interview.
"We do everything we can to minimize patients leaving with a moderate or severe leak by balloon dilatation or putting in a second valve," said Dr. Jeffrey J. Popma, an interventional cardiologist and professor of medicine at Harvard University in Boston and a PARTNER II investigator. He also noted that use of the XT device reduces the number of patients who need transapical valve placement, because the smaller catheter can be more easily manipulated through a patient’s ileofemoral anatomy.
A further advantage of the XT valve for minimizing paravalvular leaks is that it is available in a 29-mm diameter, although the 29-mm valve was not included in the PARTNER II trial. The widest valve available in the original Sapien design was 26 mm.
"The 29-mm valve is a huge advance because we know that [until now] TAVR success is greater in women, probably because about 20% of American men need a larger valve, and their leak rate was higher," said Dr. Joseph E. Bavaria, a professor of surgery at the University of Pennsylvania in Philadelphia and a PARTNER II investigator. "Having the 29-mm valve available will be a big difference," he said in an interview.
The PARTNER II trial has a second cohort, cohort A, that is comparing the Sapien XT valve system against open surgical valve replacement in patients who are deemed eligible by cardiac surgeons for surgical aortic valve replacement and are at "intermediate" risk for undergoing surgery. Enrollment of the roughly 2,000 patients who will enter this study should be complete by this summer, and with a 2-year primary endpoint the results should be available sometime in 2015, Dr. Leon said.
The PARTNER II trial is sponsored by Edwards Lifesciences, which markets the XT transcatheter aortic valve system. The company has not filed an application for approval of the Sapien XT device. Dr. Leon, Dr. Mack, Dr. Popma, and Dr. Bavaria all participated in the trial. Dr. Mintz said that he has been a consultant to Boston Scientific.
AT ACC 13
Major finding: Aortic valve replacement with the Sapien XT valve system was noninferior to replacement with the Sapien valve system and produced fewer complications.
Data source: The PARTNER II trial, which randomized 560 patients at 28 U.S. centers.
Disclosures: The PARTNER II trial is sponsored by Edwards Lifesciences, which markets the XT transcatheter aortic valve system. Dr. Leon, Dr. Mack, Dr. Popma, and Dr. Bavaria all participated in the trial. Dr. Mintz said that he has been a consultant to Boston Scientific.
Anemia treatment not beneficial in heart failure
SAN FRANCISCO – Treatment of anemic heart failure patients with the erythropoiesis-stimulating agent darbepoetin alfa failed to produce any clinically meaningful improvements and significantly raised the incidence of ischemic stroke and thrombotic events in a randomized, multicenter trial with more than 2,200 patients.
“Our findings do not support the use of erythropoiesis-stimulating agents to reduced cardiovascular morbidity and mortality in patients with systolic heart failure and mild-to-moderately severe anemia,” Dr. Karl Swedberg said on March 10 at the annual meeting of the American College of Cardiology.
Patients with heart failure also often have anemia, and results from small studies had suggested that increasing patients’ hemoglobin levels could improve their functional capacity. In addition, results from observational studies had shown worse outcomes in heart failure patients with hemoglobin levels less than 12.0 g/dL, and that a 1.0 g/dL change in hemoglobin level linked with a difference of 15-20 percentage points in all-cause mortality.
But the results of the current study called into question whether anemia plays any primary role in worsened outcomes.
Treatment with darbepoetin had “questionable clinical impact and increased thromboembolic events.” The treatment also significantly linked with an increased rate of ischemic cerebrovascular disorders that was “worrying. Our findings suggest that the hemoglobin level, like other surrogates, is simply a marker of poor prognosis in heart failure rather than a therapeutic target," said Dr. Swedberg, professor of medicine at the University of Gothenburg, Sweden. Simultaneously with his report at the meeting, the findings were also published online (N. Engl. J. Med. 2013; 368: doi:10.1056/NEJMoa1214865).
The Reduction of Events by Darbepoetin Alfa in Heart Failure (RED-HF) trial enrolled 2,278 patients with New York Heart Association class II-IV heart failure and a hemoglobin level of 9.0-12.0 g/dL at 453 sites in 33 countries during June 2006–May 2012. All patients also had a left ventricular ejection fraction of 40% or less. The study excluded patients with iron deficiency (transferrin saturation of less than 15%), evidence or bleeding or other causes of anemia, and patients with a high serum creatinine level or blood pressure above 160/100 mm Hg. The median age of the enrolled patients was 72 years, 65% had class III or IV heart failure, and their median ejection fraction was 31%. Median hemoglobin at entry was 11.2 g/dL.
The 1,136 patients assigned to receive darbepoetin injections began at a dose of 0.75 mcg/kg body weight once every 2 weeks; once they achieved a hemoglobin level of 13.0 g/dL treatment scaled back to once monthly, and treatment was continued to maintain a hemoglobin level of 13.0-14.5 g/dL. The difference in hemoglobin levels between these patients and the 1,142 randomized to the placebo control group became statistically significant after 1 month and remained significant through the study, which had a median follow-up of 28 months. The median monthly darbepoetin dosage was 167 mcg. When patients’ transferrin saturation fell below 20% they received supplemental iron.
The study’s primary endpoint was the combined rate of all-cause death and hospitalization for worsening heart failure. This occurred in 51% of the patients treated with darbepoetin alfa and 50% of those who received placebo, a difference that was not statistically significant. The result was consistent across all subgroups examined, and was not affected by adjustment for baseline differences between the two treatment arms. Total mortality was also similar between the two groups, 42% in the darbepoetin-treated patients and 40% in the controls.
The total number of hospitalizations for heart failure was 572 in the darbepoetin arm and 695 among the placebo patients, a difference that just missed being statistically significant (P = 0.06).
The overall Summary Score of the Kansas City Cardiomyopathy Questionnaire, a measure of quality of life in heart failure patients, was 6.68 in the darbepoetin-treated patients and 4.48 in the placebo patients, an average 2.2 point difference that was statistically significant. But the percent of patients with at least a 5-point increase in their score over baseline – a clinically meaningful increase – was 53% in the darbepoetin group and 48% in the placebo arm, a difference that fell short of statistical significant (P = 0.06).
The rate of study drug discontinuations due to adverse events was similar in the two arms. The rate of any embolic and thrombotic event was 14% in the darbepoetin patients and 10% in the placebo patients, a statistically significant difference. The rate of ischemic cerebrovascular events was 5% in the darbepoetin arm and 3% in the placebo arm, a statistically significant difference, although the overall rate of all cerebrovascular disorders – ischemic or hemorrhagic – was not significantly different between the two treatment groups. Episodes of septic shock were also significantly more frequent in the darbepoetin-treatment patients.
Dr. Swedberg cautioned against extrapolating the lack of benefit from darbepoetin in this patient population to other types of patients with anemia.
"I would be very careful to extend our findings to other situations because heart failure is a very special situation, with a lot of neurohormonal activation on top of other effects on the cardiorenal axis," he said. In addition, he stressed that the findings should not dissuade clinicians from administering an erythropoiesis-stimulating agent to patients with systolic heart failure and more severe anemia.
The RED-HF study was sponsored by Amgen, the company that market darbepoetin alfa (Aranesp). Dr. Swedberg said that he has been a consultant to and lecturer on behalf of Amgen as well as Novartis and Servier.
Dr. Karl Swedberg, the American College of Cardiology,
anemia, hemoglobin levels, RED-HF, Amgen
SAN FRANCISCO – Treatment of anemic heart failure patients with the erythropoiesis-stimulating agent darbepoetin alfa failed to produce any clinically meaningful improvements and significantly raised the incidence of ischemic stroke and thrombotic events in a randomized, multicenter trial with more than 2,200 patients.
“Our findings do not support the use of erythropoiesis-stimulating agents to reduced cardiovascular morbidity and mortality in patients with systolic heart failure and mild-to-moderately severe anemia,” Dr. Karl Swedberg said on March 10 at the annual meeting of the American College of Cardiology.
Patients with heart failure also often have anemia, and results from small studies had suggested that increasing patients’ hemoglobin levels could improve their functional capacity. In addition, results from observational studies had shown worse outcomes in heart failure patients with hemoglobin levels less than 12.0 g/dL, and that a 1.0 g/dL change in hemoglobin level linked with a difference of 15-20 percentage points in all-cause mortality.
But the results of the current study called into question whether anemia plays any primary role in worsened outcomes.
Treatment with darbepoetin had “questionable clinical impact and increased thromboembolic events.” The treatment also significantly linked with an increased rate of ischemic cerebrovascular disorders that was “worrying. Our findings suggest that the hemoglobin level, like other surrogates, is simply a marker of poor prognosis in heart failure rather than a therapeutic target," said Dr. Swedberg, professor of medicine at the University of Gothenburg, Sweden. Simultaneously with his report at the meeting, the findings were also published online (N. Engl. J. Med. 2013; 368: doi:10.1056/NEJMoa1214865).
The Reduction of Events by Darbepoetin Alfa in Heart Failure (RED-HF) trial enrolled 2,278 patients with New York Heart Association class II-IV heart failure and a hemoglobin level of 9.0-12.0 g/dL at 453 sites in 33 countries during June 2006–May 2012. All patients also had a left ventricular ejection fraction of 40% or less. The study excluded patients with iron deficiency (transferrin saturation of less than 15%), evidence or bleeding or other causes of anemia, and patients with a high serum creatinine level or blood pressure above 160/100 mm Hg. The median age of the enrolled patients was 72 years, 65% had class III or IV heart failure, and their median ejection fraction was 31%. Median hemoglobin at entry was 11.2 g/dL.
The 1,136 patients assigned to receive darbepoetin injections began at a dose of 0.75 mcg/kg body weight once every 2 weeks; once they achieved a hemoglobin level of 13.0 g/dL treatment scaled back to once monthly, and treatment was continued to maintain a hemoglobin level of 13.0-14.5 g/dL. The difference in hemoglobin levels between these patients and the 1,142 randomized to the placebo control group became statistically significant after 1 month and remained significant through the study, which had a median follow-up of 28 months. The median monthly darbepoetin dosage was 167 mcg. When patients’ transferrin saturation fell below 20% they received supplemental iron.
The study’s primary endpoint was the combined rate of all-cause death and hospitalization for worsening heart failure. This occurred in 51% of the patients treated with darbepoetin alfa and 50% of those who received placebo, a difference that was not statistically significant. The result was consistent across all subgroups examined, and was not affected by adjustment for baseline differences between the two treatment arms. Total mortality was also similar between the two groups, 42% in the darbepoetin-treated patients and 40% in the controls.
The total number of hospitalizations for heart failure was 572 in the darbepoetin arm and 695 among the placebo patients, a difference that just missed being statistically significant (P = 0.06).
The overall Summary Score of the Kansas City Cardiomyopathy Questionnaire, a measure of quality of life in heart failure patients, was 6.68 in the darbepoetin-treated patients and 4.48 in the placebo patients, an average 2.2 point difference that was statistically significant. But the percent of patients with at least a 5-point increase in their score over baseline – a clinically meaningful increase – was 53% in the darbepoetin group and 48% in the placebo arm, a difference that fell short of statistical significant (P = 0.06).
The rate of study drug discontinuations due to adverse events was similar in the two arms. The rate of any embolic and thrombotic event was 14% in the darbepoetin patients and 10% in the placebo patients, a statistically significant difference. The rate of ischemic cerebrovascular events was 5% in the darbepoetin arm and 3% in the placebo arm, a statistically significant difference, although the overall rate of all cerebrovascular disorders – ischemic or hemorrhagic – was not significantly different between the two treatment groups. Episodes of septic shock were also significantly more frequent in the darbepoetin-treatment patients.
Dr. Swedberg cautioned against extrapolating the lack of benefit from darbepoetin in this patient population to other types of patients with anemia.
"I would be very careful to extend our findings to other situations because heart failure is a very special situation, with a lot of neurohormonal activation on top of other effects on the cardiorenal axis," he said. In addition, he stressed that the findings should not dissuade clinicians from administering an erythropoiesis-stimulating agent to patients with systolic heart failure and more severe anemia.
The RED-HF study was sponsored by Amgen, the company that market darbepoetin alfa (Aranesp). Dr. Swedberg said that he has been a consultant to and lecturer on behalf of Amgen as well as Novartis and Servier.
SAN FRANCISCO – Treatment of anemic heart failure patients with the erythropoiesis-stimulating agent darbepoetin alfa failed to produce any clinically meaningful improvements and significantly raised the incidence of ischemic stroke and thrombotic events in a randomized, multicenter trial with more than 2,200 patients.
“Our findings do not support the use of erythropoiesis-stimulating agents to reduced cardiovascular morbidity and mortality in patients with systolic heart failure and mild-to-moderately severe anemia,” Dr. Karl Swedberg said on March 10 at the annual meeting of the American College of Cardiology.
Patients with heart failure also often have anemia, and results from small studies had suggested that increasing patients’ hemoglobin levels could improve their functional capacity. In addition, results from observational studies had shown worse outcomes in heart failure patients with hemoglobin levels less than 12.0 g/dL, and that a 1.0 g/dL change in hemoglobin level linked with a difference of 15-20 percentage points in all-cause mortality.
But the results of the current study called into question whether anemia plays any primary role in worsened outcomes.
Treatment with darbepoetin had “questionable clinical impact and increased thromboembolic events.” The treatment also significantly linked with an increased rate of ischemic cerebrovascular disorders that was “worrying. Our findings suggest that the hemoglobin level, like other surrogates, is simply a marker of poor prognosis in heart failure rather than a therapeutic target," said Dr. Swedberg, professor of medicine at the University of Gothenburg, Sweden. Simultaneously with his report at the meeting, the findings were also published online (N. Engl. J. Med. 2013; 368: doi:10.1056/NEJMoa1214865).
The Reduction of Events by Darbepoetin Alfa in Heart Failure (RED-HF) trial enrolled 2,278 patients with New York Heart Association class II-IV heart failure and a hemoglobin level of 9.0-12.0 g/dL at 453 sites in 33 countries during June 2006–May 2012. All patients also had a left ventricular ejection fraction of 40% or less. The study excluded patients with iron deficiency (transferrin saturation of less than 15%), evidence or bleeding or other causes of anemia, and patients with a high serum creatinine level or blood pressure above 160/100 mm Hg. The median age of the enrolled patients was 72 years, 65% had class III or IV heart failure, and their median ejection fraction was 31%. Median hemoglobin at entry was 11.2 g/dL.
The 1,136 patients assigned to receive darbepoetin injections began at a dose of 0.75 mcg/kg body weight once every 2 weeks; once they achieved a hemoglobin level of 13.0 g/dL treatment scaled back to once monthly, and treatment was continued to maintain a hemoglobin level of 13.0-14.5 g/dL. The difference in hemoglobin levels between these patients and the 1,142 randomized to the placebo control group became statistically significant after 1 month and remained significant through the study, which had a median follow-up of 28 months. The median monthly darbepoetin dosage was 167 mcg. When patients’ transferrin saturation fell below 20% they received supplemental iron.
The study’s primary endpoint was the combined rate of all-cause death and hospitalization for worsening heart failure. This occurred in 51% of the patients treated with darbepoetin alfa and 50% of those who received placebo, a difference that was not statistically significant. The result was consistent across all subgroups examined, and was not affected by adjustment for baseline differences between the two treatment arms. Total mortality was also similar between the two groups, 42% in the darbepoetin-treated patients and 40% in the controls.
The total number of hospitalizations for heart failure was 572 in the darbepoetin arm and 695 among the placebo patients, a difference that just missed being statistically significant (P = 0.06).
The overall Summary Score of the Kansas City Cardiomyopathy Questionnaire, a measure of quality of life in heart failure patients, was 6.68 in the darbepoetin-treated patients and 4.48 in the placebo patients, an average 2.2 point difference that was statistically significant. But the percent of patients with at least a 5-point increase in their score over baseline – a clinically meaningful increase – was 53% in the darbepoetin group and 48% in the placebo arm, a difference that fell short of statistical significant (P = 0.06).
The rate of study drug discontinuations due to adverse events was similar in the two arms. The rate of any embolic and thrombotic event was 14% in the darbepoetin patients and 10% in the placebo patients, a statistically significant difference. The rate of ischemic cerebrovascular events was 5% in the darbepoetin arm and 3% in the placebo arm, a statistically significant difference, although the overall rate of all cerebrovascular disorders – ischemic or hemorrhagic – was not significantly different between the two treatment groups. Episodes of septic shock were also significantly more frequent in the darbepoetin-treatment patients.
Dr. Swedberg cautioned against extrapolating the lack of benefit from darbepoetin in this patient population to other types of patients with anemia.
"I would be very careful to extend our findings to other situations because heart failure is a very special situation, with a lot of neurohormonal activation on top of other effects on the cardiorenal axis," he said. In addition, he stressed that the findings should not dissuade clinicians from administering an erythropoiesis-stimulating agent to patients with systolic heart failure and more severe anemia.
The RED-HF study was sponsored by Amgen, the company that market darbepoetin alfa (Aranesp). Dr. Swedberg said that he has been a consultant to and lecturer on behalf of Amgen as well as Novartis and Servier.
Dr. Karl Swedberg, the American College of Cardiology,
anemia, hemoglobin levels, RED-HF, Amgen
Dr. Karl Swedberg, the American College of Cardiology,
anemia, hemoglobin levels, RED-HF, Amgen
Major Finding: Treatment
with darbepoetin alfa failed to reduce the rate of death or hospitalization for
worsening heart failure, compared with placebo.
Data Source: The
data came from the RED-HF trial, a multicenter, randomized, controlled trial
that enrolled 2,278 patients with heart failure and anemia.
Disclosures: The
RED-HF study was sponsored by Amgen, the company that market darbepoetin alfa
(Aranesp). Dr. Swedberg said that he has been a consultant to and lecturer on
behalf of Amgen as well as Novartis and Servier.
Heart teams inch into routine cardiac practice
The heart team has drawn fans since the franchise first took the field about 3 years ago, but how has it fared in the standings?
It may be hard to believe, given how quickly the heart team concept wormed its way into the cardiac mindset and lexicon, that the term seems to have been first coined in August 2010, when the most recent edition of the myocardial revascularization guidelines of the European Society of Cardiology was unveiled at the society’s annual meeting and then published 2 months later.
The ESC revascularization guidelines (Euro. Heart J. 2010;31:2501-55), as well as authors who have written about heart teams since then, credited the name to investigators from the Synergy Between PCI With Taxus and Cardiac Surgery (SYNTAX) trial, the group that formalized the idea of an interventional cardiologist and cardiac surgeon closely collaborating in assessing and managing patients with complex coronary artery disease who need revascularization.
The heart team approach received further support a year later, when the 2011 revascularization guidelines for percutaneous coronary intervention (Circulation 2011;124:2574-2609) and for coronary artery bypass surgery (Circulation 2011;124: 2610-42) of the American College of Cardiology, the American Heart Association, surgical societies such as the Society for Thoracic Surgery (STS), and several other groups repeated what the ESC had said and tapped heart teams – a joint consult between an interventional cardiologist and a heart surgeon – as the basic decision maker for the management of unprotected left main or complex coronary artery disease. Both the 2011 PCI and coronary artery bypass graft (CABG) guidelines said that "a heart team approach to revascularization is recommended" for these advanced-disease patients, a class I recommendation.
Further buttressing the role of heart teams, as well as muddying the waters on exactly which physicians and surgeons belong on a heart team, has been their growing role in managing other cardiac disorders, most notably for transcatheter aortic valve replacement (TAVR). Last May, the Centers for Medicare and Medicaid Services solidified the link when it ruled that it would pay for TAVR in Medicare beneficiaries only if a heart team was involved from the start. As a result, heart teams are now more tightly and consistently entwined with TAVR than they are with revascularization, where the use of a heart team tracks closely with the increased complexity of a patient’s coronary disease.
Broadening use of heart teams also makes them a little harder to define, as the interventionalist and surgeon collaboration on TAVR involves different jobs and different players than does coronary revascularization. The role heart teams play in early 2013 for U.S. revascularization of coronary disease remains a bit fuzzy. Every interventional cardiologist and cardiac surgeon have their sense of how they use heart teams and how other physicians and surgeons at their hospitals use them for revascularization cases, but a global accounting of the extent to which the heart team concept has penetrated American practice is totally lacking, and will likely stay that way.
Revascularization heart teams remain poorly defined
"The notion of encouraging multidisciplinary care in cardiovascular medicine, exemplified by the heart team concept has gained increasing traction" for revascularization, TAVR, and other complex endovascular interventions, Dr. Brahmajee K. Nallamothu and Dr. David J. Cohen wrote in a perspective article last year (Circulation Cardiovasc. Qual. Outcomes 2012;5:410-3). Examples of that traction include adding heart teams to guidelines, and sessions that focus on heart teams at meetings, Dr. Nallamothu said in an interview.
But thinking of heart teams as formal entities for coronary disease, as formal as for TAVR, is tricky. "The heart team concept always sounds like a great idea, but the real gap has been in showing people in real-world practice how they can apply the idea of a heart team to front-line decision making about patients undergoing revascularization," said Dr. Nallamothu, an interventional cardiologist at the University of Michigan in Ann Arbor. "We have not shown hospitals and physicians how to do this in a practical way. We need more discussions by multidisciplinary teams about complex patients." Heart teams "have filtered into our guidelines without showing doctors how you do it practically, and even if you can, how do you pay people for doing it?" he said.
But most heart surgeons and cardiologists will tell you that what they typically see "is not a lot of formal heart teams, but a lot of discussions, and that’s only been strengthened as TAVR and other structural heart disease programs have started to be team based," Dr. Nallamothu said.
Informal discussions between cardiologists and surgeons about revascularization cases have, of course, gone on for years, and most seem comfortable with it staying that way.
"In community hospitals, patients without complex CAD [coronary artery disease] do not routinely have cardiac surgery consultations. The traditional model is that when a patient needs surgery, the cardiologist consults a surgeon," said Dr. David C. May, an interventional cardiologist in Coppell, Tex.
"Ad hoc or provisional stenting remains the norm in about half the cases. It’s important to separate the complex and simple cases. Most ad hoc PCI is not done in complex cases. Complex cases are done as scheduled, planned procedures," Dr. May said in an interview.
"In the real world, cardiologists see surgeons all the time, every day. We interact all the time. Collegial discussions about patients are always on the table. Most of these consultations are off the cuff and not official," agreed Dr. Peter C. Block, an interventional cardiologist at Emory University in Atlanta.
"If a patient is clearly a PCI candidate, with a SYNTAX score of 15 and no diabetes, I will do PCI" without a surgical consult. "If the patient has a SYNTAX score of 28 or 30 and I think he is not a good patient for PCI, I’ll discuss it with my surgical colleagues. If we agree, I’ll either do the PCI or pass off the patient for surgery. It would be very difficult to get a surgical consult for every U.S. patient who gets PCI," Dr. Block said.
Waiting for "the pause"
But is this approach to heart team use consistent with existing guidelines, and do cardiologists usually make the right call on when they should seek a surgical consult? A critical part is what some people call "the pause," or "taking a patient off the table." An interventional cardiologist can either perform a coronary catheterization for diagnostic purposes only, obtain the patient’s coronary angiogram, and then halt the catheterization procedure while the angiogram films are reviewed by the cardiologist and surgeon, or the cardiologist can elect to continue on after angiography and proceed directly to PCI.
"Many cardiologists don’t believe a pause is necessary. If you ask most U.S. cardiologists, they will tell you that they are comfortable making the decision to stent on their own, that they don’t need a surgeon engaged in the conversation," said heart surgeon Timothy J. Gardner, medical director of the center for heart and vascular health at Christiana Care Health System in Wilmington, Del. "They don’t want to delay the stenting to another catheterization if they believe that patient needs PCI."
But at least one cardiac surgeon believes that postangiogram pauses are on the rise.
"We are definitely seeing a pause after angiography a lot more," said Dr. Mathew R. Williams, a cardiac surgeon and interventional cardiologist and codirector of the heart valve center at Columbia University in New York. "Rather than doing PCI at the time of diagnostic cath, patients are coming off the table, even when we feel the patient should have PCI. Not every case, but patients are coming off the table and that discussion is occurring for patients with complex disease, with left main and LAD [left anterior descending] involvement," Dr. Williams said.
Paying for the consult
Reimbursement is another potential challenge to the viability of surgical consults for revascularization patients.
"It’s a challenge to get people in a busy community practice together as a heart team. They are their own business entity. How do you create a heart team across these business entities?" asked Dr. Thoralf M. Sundt III, chief of cardiac surgery at Massachusetts General Hospital in Boston. "My view of community practice is limited, but I think it’s a real challenge for most community practices to do it. People agree with the heart team concept in theory, but executing it in communities will be a huge challenge."
Dr. Nallamothu of the University of Michigan said, "I’m at an academic center, and for the most part we are salaried. A lot of the heart team discussions are built into our academic responsibilities. For heart teams to really take off in the community, we need to understand how we are going to do it. You need a mechanism to value the work of evaluating cases and rendering opinions about treatment. You can’t expect physicians to do that pro bono. I think it will be a disincentive for full engagement and cooperation unless you can figure out" how to compensate physicians for just rendering their opinions.
"Community hospitals are all over the place because many community hospitals have a bunch of private groups and it’s been a challenge to get them to form heart teams. It’s very politically charged. At other community hospitals the physicians are employees of the hospital. They do a better job because they already have financial alignment," said Dr. Williams.
"Everyone recognizes that where we need to be is organized by service lines and not by academic departments," said Dr. Sundt. "A patient with heart disease wants to be cared for by physicians who deal with heart disease. The patient doesn’t care if they are cardiologists or surgeons, or who is in this group or that group. The patient needs his disease managed by physicians who focus on the patient. That’s what the heart team is. The way to reimburse it is to put all the money into the heart team bucket and then figure out how to distribute it. If CMS set up reimbursement [for revascularization] so that it depended on a heart team" as it did for TAVR, then revascularization decisions would more consistently involve heart teams, he said in an interview.
"Right now, no one pays for [a consult between a cardiologist and surgeon]. The thing that would change this the quickest would be payment reform that rewarded the [heart team] approach," said Dr. W. Douglas Weaver, medical director of the heart and vascular institute at Henry Ford Health System in Detroit.
"Employment by hospitals has driven the process of cardiologists working with surgeons on revascularization cases," Dr. Weaver added. Reimbursement issues could be resolved to some extent by more episode-based payments. What will also drive it is more integration among physicians."
But while compensation for a heart team consult on revascularization might help, and while better-integrated health systems would streamline the delivery of multidisciplinary care, inadequate compensation for participation on heart teams has not been a major roadblock up to now, many say.
"It would be awful if financial misalignment interferes with making the best decisions for patients, but I think the vast majority of cardiologists and cardiac surgeons rise to the occasion" and provide consultations even when they are not paid for it, said Dr. Patrick T. O’Gara, director of clinical cardiology at Brigham and Women’s Hospital in Boston.
"I don’t think reimbursement drives practice. Most cardiologists and surgeons try to do what’s right for the patient. They understand that sometimes they will not get reimbursed," said Dr. Gardner.
TAVR leads the way
Reimbursement for heart team members in revascularization cases may not currently be as well structured as it is for TAVR cases, but in other respects the growing number of TAVR cases and the heart teams that form to treat those cases seem to be having a spillover effect on revascularization. The TAVR experience "has catapulted the collaboration between cardiologists and surgeons in all decision making," said Dr. Weaver.
"A heart team that works together on TAVR sets the stage for collegiality" in revascularization cases, said Dr. Block. "My suspicion is that TAVR relationships will lead to closer communication [between cardiologists and surgeons] because they will see each other more often and have closer ties. The barriers [between cardiologists and surgeons] are breaking down. I don’t know how far that has gone in community hospitals, because TAVR is just beginning at most community hospitals."
It’s reached the community of Dr. May, who has been performing TAVR for a while in his Dallas-area practice.
TAVR programs "foster a significantly improved collaboration between the cardiac surgeons and cardiologists. At a higher level, it has also fostered a much closer relationship between STS and ACC because of their need to work together on guidelines and registry development. So in that regard TAVR has made a big difference," said Dr. May.
"When TAVR is done, everyone [cardiologist and surgeon] is in the room doing a piece of the work. When you talk about collaborative revascularization, you are talking about a decision. So the collaboration is different. With TAVR you work together and develop a combat-level closeness because of the care you all deliver to patients. For revascularization, it is a decision made over a cup of coffee, not in the cath lab," he added.
Dr. May and several others think the collaborative boost powered by TAVR heart teams, and perhaps reinforced in revascularization cases, has the potential to catalyze a more profound shift in cardiac care and professional relationships.
"The experience with TAVR has shown that the most fruitful way for cardiologists and surgeons to go forward is to work collaboratively," continued Dr. May. "That’s a new paradigm, and it needs to be embraced wholeheartedly. It also promotes the idea of doing what is best for patients. It’s not that cardiologists do PCI and surgeons do CABG and we don’t talk to each other. The paradigm shift we have seen with TAVR has tremendous merit."
"It’s time to bury the old PCI vs. CABG battle of the past," said Dr. Williams. If cardiologists and surgeons don’t work together, "it’s not good for them or for patients. When you work together, it’s mutually beneficial, and most importantly it’s better for patients."
What’s next for revascularization heart teams?
Have heart teams for revascularization become the standard of care, is there consensus on when they should be used, and do most U.S. interventionalists and cardiac surgeons already belong to a heart team? The short answer to all three questions is maybe.
"Heart teams may be the standard of care at many [U.S.] institutions, but I can’t speak for all. At Brigham and Women’s, it is our standard of care for patients with complicated CAD," said Dr. O’Gara. Right now "heart teams are the aspiration, but I’m not sure it is the standard. But it was a class I recommendation that had unanimous consensus behind it.
"The ACC has focused on education to proselytize the value of this approach," said Dr. O’Gara, who is currently ACC vice president. The results of the SYNTAX trial established the expectation that among patients with multivessel disease, there would be a discussion before surgeons, interventionalists, and the patient about the optimal [revascularization] approach. But aside from the educational aspect on the benefit of doing this I’m not aware of any regulatory agency that is embedding heart teams in, say, a performance metric," the better to solidify their role in practice.
Most surgeons and cardiologists fall back on calling for a heart team for any patients with "complex" coronary disease, generally defined as patients with left main disease or triple-vessel advanced coronary disease. Those were the categories of coronary disease for which PCI’s use was deemed "uncertain" by the revascularization appropriate-use criteria established last year by the ACC, STS, and several other cardiology and surgical groups (J. Am. Coll. Cardiol. 2012;59:857-81).
"I wouldn’t say that for revascularization using heart teams is the standard of care, but it’s being used in more and more places," said Dr. Weaver. A heart team "is not for every case; it takes time, and in most cases it probably won’t change the decision. But certainly for complex cases it should be routine. At many centers it is routine – but for a small subset of patients," said Dr. Weaver. "The ACC can help define what is a complex case and help convince payers that this is useful and that we need to reward this behavior as an incentive."
In general, cardiologists and surgeons likely feel little urgency to become part of revascularization heart teams, if for no other reason than most of them probably believe they are already part of one.
"I would not be surprised if most American cardiologists and surgeons feel that they are already on a heart team. They feel: ‘I practice at a place with a surgeon, or with a cardiologist – we’re a team.’ I don’t think that anyone in private practice loses sleep because they think they are not on a heart team," said Dr. Nallamothu.
But there is probably a need for a more systematic application of heart teams to appropriate cases, a clearer definition of appropriate cases, and increased leadership from professional societies to accomplish these goals, according to at least some experts.
"I wouldn’t say that societies have fallen short, but there is a lot of work to do. It’s a huge challenge, and these groups could definitely lead going forward," said Dr. Sundt.
"Societies need to take the lead on defining optimal care" and the collaborative approach to revascularization, said Dr. May. The collaborative discussions that the ACC and STS have had about TAVR is "a model" for future collaborations on revascularization and other areas of multidisciplinary cardiac care, such as mitral valve replacement.
"In the long term, I think there will be more integration of interventional cardiology and cardiac surgery, with more overlap of the procedures they use," predicted Dr. Nallamothu. As that happens, "we’ll also see more team discussions about complex revascularization cases."
Dr. Nallamothu, Dr. May, Dr. Block, Dr. Gardner, Dr. Williams, Dr. Sundt, Dr. Weaver, and Dr. O’Gara all said that they had no relevant financial disclosures.
m.zoler@elsevier.com
On Twitter @mitchelzoler
The heart team has drawn fans since the franchise first took the field about 3 years ago, but how has it fared in the standings?
It may be hard to believe, given how quickly the heart team concept wormed its way into the cardiac mindset and lexicon, that the term seems to have been first coined in August 2010, when the most recent edition of the myocardial revascularization guidelines of the European Society of Cardiology was unveiled at the society’s annual meeting and then published 2 months later.
The ESC revascularization guidelines (Euro. Heart J. 2010;31:2501-55), as well as authors who have written about heart teams since then, credited the name to investigators from the Synergy Between PCI With Taxus and Cardiac Surgery (SYNTAX) trial, the group that formalized the idea of an interventional cardiologist and cardiac surgeon closely collaborating in assessing and managing patients with complex coronary artery disease who need revascularization.
The heart team approach received further support a year later, when the 2011 revascularization guidelines for percutaneous coronary intervention (Circulation 2011;124:2574-2609) and for coronary artery bypass surgery (Circulation 2011;124: 2610-42) of the American College of Cardiology, the American Heart Association, surgical societies such as the Society for Thoracic Surgery (STS), and several other groups repeated what the ESC had said and tapped heart teams – a joint consult between an interventional cardiologist and a heart surgeon – as the basic decision maker for the management of unprotected left main or complex coronary artery disease. Both the 2011 PCI and coronary artery bypass graft (CABG) guidelines said that "a heart team approach to revascularization is recommended" for these advanced-disease patients, a class I recommendation.
Further buttressing the role of heart teams, as well as muddying the waters on exactly which physicians and surgeons belong on a heart team, has been their growing role in managing other cardiac disorders, most notably for transcatheter aortic valve replacement (TAVR). Last May, the Centers for Medicare and Medicaid Services solidified the link when it ruled that it would pay for TAVR in Medicare beneficiaries only if a heart team was involved from the start. As a result, heart teams are now more tightly and consistently entwined with TAVR than they are with revascularization, where the use of a heart team tracks closely with the increased complexity of a patient’s coronary disease.
Broadening use of heart teams also makes them a little harder to define, as the interventionalist and surgeon collaboration on TAVR involves different jobs and different players than does coronary revascularization. The role heart teams play in early 2013 for U.S. revascularization of coronary disease remains a bit fuzzy. Every interventional cardiologist and cardiac surgeon have their sense of how they use heart teams and how other physicians and surgeons at their hospitals use them for revascularization cases, but a global accounting of the extent to which the heart team concept has penetrated American practice is totally lacking, and will likely stay that way.
Revascularization heart teams remain poorly defined
"The notion of encouraging multidisciplinary care in cardiovascular medicine, exemplified by the heart team concept has gained increasing traction" for revascularization, TAVR, and other complex endovascular interventions, Dr. Brahmajee K. Nallamothu and Dr. David J. Cohen wrote in a perspective article last year (Circulation Cardiovasc. Qual. Outcomes 2012;5:410-3). Examples of that traction include adding heart teams to guidelines, and sessions that focus on heart teams at meetings, Dr. Nallamothu said in an interview.
But thinking of heart teams as formal entities for coronary disease, as formal as for TAVR, is tricky. "The heart team concept always sounds like a great idea, but the real gap has been in showing people in real-world practice how they can apply the idea of a heart team to front-line decision making about patients undergoing revascularization," said Dr. Nallamothu, an interventional cardiologist at the University of Michigan in Ann Arbor. "We have not shown hospitals and physicians how to do this in a practical way. We need more discussions by multidisciplinary teams about complex patients." Heart teams "have filtered into our guidelines without showing doctors how you do it practically, and even if you can, how do you pay people for doing it?" he said.
But most heart surgeons and cardiologists will tell you that what they typically see "is not a lot of formal heart teams, but a lot of discussions, and that’s only been strengthened as TAVR and other structural heart disease programs have started to be team based," Dr. Nallamothu said.
Informal discussions between cardiologists and surgeons about revascularization cases have, of course, gone on for years, and most seem comfortable with it staying that way.
"In community hospitals, patients without complex CAD [coronary artery disease] do not routinely have cardiac surgery consultations. The traditional model is that when a patient needs surgery, the cardiologist consults a surgeon," said Dr. David C. May, an interventional cardiologist in Coppell, Tex.
"Ad hoc or provisional stenting remains the norm in about half the cases. It’s important to separate the complex and simple cases. Most ad hoc PCI is not done in complex cases. Complex cases are done as scheduled, planned procedures," Dr. May said in an interview.
"In the real world, cardiologists see surgeons all the time, every day. We interact all the time. Collegial discussions about patients are always on the table. Most of these consultations are off the cuff and not official," agreed Dr. Peter C. Block, an interventional cardiologist at Emory University in Atlanta.
"If a patient is clearly a PCI candidate, with a SYNTAX score of 15 and no diabetes, I will do PCI" without a surgical consult. "If the patient has a SYNTAX score of 28 or 30 and I think he is not a good patient for PCI, I’ll discuss it with my surgical colleagues. If we agree, I’ll either do the PCI or pass off the patient for surgery. It would be very difficult to get a surgical consult for every U.S. patient who gets PCI," Dr. Block said.
Waiting for "the pause"
But is this approach to heart team use consistent with existing guidelines, and do cardiologists usually make the right call on when they should seek a surgical consult? A critical part is what some people call "the pause," or "taking a patient off the table." An interventional cardiologist can either perform a coronary catheterization for diagnostic purposes only, obtain the patient’s coronary angiogram, and then halt the catheterization procedure while the angiogram films are reviewed by the cardiologist and surgeon, or the cardiologist can elect to continue on after angiography and proceed directly to PCI.
"Many cardiologists don’t believe a pause is necessary. If you ask most U.S. cardiologists, they will tell you that they are comfortable making the decision to stent on their own, that they don’t need a surgeon engaged in the conversation," said heart surgeon Timothy J. Gardner, medical director of the center for heart and vascular health at Christiana Care Health System in Wilmington, Del. "They don’t want to delay the stenting to another catheterization if they believe that patient needs PCI."
But at least one cardiac surgeon believes that postangiogram pauses are on the rise.
"We are definitely seeing a pause after angiography a lot more," said Dr. Mathew R. Williams, a cardiac surgeon and interventional cardiologist and codirector of the heart valve center at Columbia University in New York. "Rather than doing PCI at the time of diagnostic cath, patients are coming off the table, even when we feel the patient should have PCI. Not every case, but patients are coming off the table and that discussion is occurring for patients with complex disease, with left main and LAD [left anterior descending] involvement," Dr. Williams said.
Paying for the consult
Reimbursement is another potential challenge to the viability of surgical consults for revascularization patients.
"It’s a challenge to get people in a busy community practice together as a heart team. They are their own business entity. How do you create a heart team across these business entities?" asked Dr. Thoralf M. Sundt III, chief of cardiac surgery at Massachusetts General Hospital in Boston. "My view of community practice is limited, but I think it’s a real challenge for most community practices to do it. People agree with the heart team concept in theory, but executing it in communities will be a huge challenge."
Dr. Nallamothu of the University of Michigan said, "I’m at an academic center, and for the most part we are salaried. A lot of the heart team discussions are built into our academic responsibilities. For heart teams to really take off in the community, we need to understand how we are going to do it. You need a mechanism to value the work of evaluating cases and rendering opinions about treatment. You can’t expect physicians to do that pro bono. I think it will be a disincentive for full engagement and cooperation unless you can figure out" how to compensate physicians for just rendering their opinions.
"Community hospitals are all over the place because many community hospitals have a bunch of private groups and it’s been a challenge to get them to form heart teams. It’s very politically charged. At other community hospitals the physicians are employees of the hospital. They do a better job because they already have financial alignment," said Dr. Williams.
"Everyone recognizes that where we need to be is organized by service lines and not by academic departments," said Dr. Sundt. "A patient with heart disease wants to be cared for by physicians who deal with heart disease. The patient doesn’t care if they are cardiologists or surgeons, or who is in this group or that group. The patient needs his disease managed by physicians who focus on the patient. That’s what the heart team is. The way to reimburse it is to put all the money into the heart team bucket and then figure out how to distribute it. If CMS set up reimbursement [for revascularization] so that it depended on a heart team" as it did for TAVR, then revascularization decisions would more consistently involve heart teams, he said in an interview.
"Right now, no one pays for [a consult between a cardiologist and surgeon]. The thing that would change this the quickest would be payment reform that rewarded the [heart team] approach," said Dr. W. Douglas Weaver, medical director of the heart and vascular institute at Henry Ford Health System in Detroit.
"Employment by hospitals has driven the process of cardiologists working with surgeons on revascularization cases," Dr. Weaver added. Reimbursement issues could be resolved to some extent by more episode-based payments. What will also drive it is more integration among physicians."
But while compensation for a heart team consult on revascularization might help, and while better-integrated health systems would streamline the delivery of multidisciplinary care, inadequate compensation for participation on heart teams has not been a major roadblock up to now, many say.
"It would be awful if financial misalignment interferes with making the best decisions for patients, but I think the vast majority of cardiologists and cardiac surgeons rise to the occasion" and provide consultations even when they are not paid for it, said Dr. Patrick T. O’Gara, director of clinical cardiology at Brigham and Women’s Hospital in Boston.
"I don’t think reimbursement drives practice. Most cardiologists and surgeons try to do what’s right for the patient. They understand that sometimes they will not get reimbursed," said Dr. Gardner.
TAVR leads the way
Reimbursement for heart team members in revascularization cases may not currently be as well structured as it is for TAVR cases, but in other respects the growing number of TAVR cases and the heart teams that form to treat those cases seem to be having a spillover effect on revascularization. The TAVR experience "has catapulted the collaboration between cardiologists and surgeons in all decision making," said Dr. Weaver.
"A heart team that works together on TAVR sets the stage for collegiality" in revascularization cases, said Dr. Block. "My suspicion is that TAVR relationships will lead to closer communication [between cardiologists and surgeons] because they will see each other more often and have closer ties. The barriers [between cardiologists and surgeons] are breaking down. I don’t know how far that has gone in community hospitals, because TAVR is just beginning at most community hospitals."
It’s reached the community of Dr. May, who has been performing TAVR for a while in his Dallas-area practice.
TAVR programs "foster a significantly improved collaboration between the cardiac surgeons and cardiologists. At a higher level, it has also fostered a much closer relationship between STS and ACC because of their need to work together on guidelines and registry development. So in that regard TAVR has made a big difference," said Dr. May.
"When TAVR is done, everyone [cardiologist and surgeon] is in the room doing a piece of the work. When you talk about collaborative revascularization, you are talking about a decision. So the collaboration is different. With TAVR you work together and develop a combat-level closeness because of the care you all deliver to patients. For revascularization, it is a decision made over a cup of coffee, not in the cath lab," he added.
Dr. May and several others think the collaborative boost powered by TAVR heart teams, and perhaps reinforced in revascularization cases, has the potential to catalyze a more profound shift in cardiac care and professional relationships.
"The experience with TAVR has shown that the most fruitful way for cardiologists and surgeons to go forward is to work collaboratively," continued Dr. May. "That’s a new paradigm, and it needs to be embraced wholeheartedly. It also promotes the idea of doing what is best for patients. It’s not that cardiologists do PCI and surgeons do CABG and we don’t talk to each other. The paradigm shift we have seen with TAVR has tremendous merit."
"It’s time to bury the old PCI vs. CABG battle of the past," said Dr. Williams. If cardiologists and surgeons don’t work together, "it’s not good for them or for patients. When you work together, it’s mutually beneficial, and most importantly it’s better for patients."
What’s next for revascularization heart teams?
Have heart teams for revascularization become the standard of care, is there consensus on when they should be used, and do most U.S. interventionalists and cardiac surgeons already belong to a heart team? The short answer to all three questions is maybe.
"Heart teams may be the standard of care at many [U.S.] institutions, but I can’t speak for all. At Brigham and Women’s, it is our standard of care for patients with complicated CAD," said Dr. O’Gara. Right now "heart teams are the aspiration, but I’m not sure it is the standard. But it was a class I recommendation that had unanimous consensus behind it.
"The ACC has focused on education to proselytize the value of this approach," said Dr. O’Gara, who is currently ACC vice president. The results of the SYNTAX trial established the expectation that among patients with multivessel disease, there would be a discussion before surgeons, interventionalists, and the patient about the optimal [revascularization] approach. But aside from the educational aspect on the benefit of doing this I’m not aware of any regulatory agency that is embedding heart teams in, say, a performance metric," the better to solidify their role in practice.
Most surgeons and cardiologists fall back on calling for a heart team for any patients with "complex" coronary disease, generally defined as patients with left main disease or triple-vessel advanced coronary disease. Those were the categories of coronary disease for which PCI’s use was deemed "uncertain" by the revascularization appropriate-use criteria established last year by the ACC, STS, and several other cardiology and surgical groups (J. Am. Coll. Cardiol. 2012;59:857-81).
"I wouldn’t say that for revascularization using heart teams is the standard of care, but it’s being used in more and more places," said Dr. Weaver. A heart team "is not for every case; it takes time, and in most cases it probably won’t change the decision. But certainly for complex cases it should be routine. At many centers it is routine – but for a small subset of patients," said Dr. Weaver. "The ACC can help define what is a complex case and help convince payers that this is useful and that we need to reward this behavior as an incentive."
In general, cardiologists and surgeons likely feel little urgency to become part of revascularization heart teams, if for no other reason than most of them probably believe they are already part of one.
"I would not be surprised if most American cardiologists and surgeons feel that they are already on a heart team. They feel: ‘I practice at a place with a surgeon, or with a cardiologist – we’re a team.’ I don’t think that anyone in private practice loses sleep because they think they are not on a heart team," said Dr. Nallamothu.
But there is probably a need for a more systematic application of heart teams to appropriate cases, a clearer definition of appropriate cases, and increased leadership from professional societies to accomplish these goals, according to at least some experts.
"I wouldn’t say that societies have fallen short, but there is a lot of work to do. It’s a huge challenge, and these groups could definitely lead going forward," said Dr. Sundt.
"Societies need to take the lead on defining optimal care" and the collaborative approach to revascularization, said Dr. May. The collaborative discussions that the ACC and STS have had about TAVR is "a model" for future collaborations on revascularization and other areas of multidisciplinary cardiac care, such as mitral valve replacement.
"In the long term, I think there will be more integration of interventional cardiology and cardiac surgery, with more overlap of the procedures they use," predicted Dr. Nallamothu. As that happens, "we’ll also see more team discussions about complex revascularization cases."
Dr. Nallamothu, Dr. May, Dr. Block, Dr. Gardner, Dr. Williams, Dr. Sundt, Dr. Weaver, and Dr. O’Gara all said that they had no relevant financial disclosures.
m.zoler@elsevier.com
On Twitter @mitchelzoler
The heart team has drawn fans since the franchise first took the field about 3 years ago, but how has it fared in the standings?
It may be hard to believe, given how quickly the heart team concept wormed its way into the cardiac mindset and lexicon, that the term seems to have been first coined in August 2010, when the most recent edition of the myocardial revascularization guidelines of the European Society of Cardiology was unveiled at the society’s annual meeting and then published 2 months later.
The ESC revascularization guidelines (Euro. Heart J. 2010;31:2501-55), as well as authors who have written about heart teams since then, credited the name to investigators from the Synergy Between PCI With Taxus and Cardiac Surgery (SYNTAX) trial, the group that formalized the idea of an interventional cardiologist and cardiac surgeon closely collaborating in assessing and managing patients with complex coronary artery disease who need revascularization.
The heart team approach received further support a year later, when the 2011 revascularization guidelines for percutaneous coronary intervention (Circulation 2011;124:2574-2609) and for coronary artery bypass surgery (Circulation 2011;124: 2610-42) of the American College of Cardiology, the American Heart Association, surgical societies such as the Society for Thoracic Surgery (STS), and several other groups repeated what the ESC had said and tapped heart teams – a joint consult between an interventional cardiologist and a heart surgeon – as the basic decision maker for the management of unprotected left main or complex coronary artery disease. Both the 2011 PCI and coronary artery bypass graft (CABG) guidelines said that "a heart team approach to revascularization is recommended" for these advanced-disease patients, a class I recommendation.
Further buttressing the role of heart teams, as well as muddying the waters on exactly which physicians and surgeons belong on a heart team, has been their growing role in managing other cardiac disorders, most notably for transcatheter aortic valve replacement (TAVR). Last May, the Centers for Medicare and Medicaid Services solidified the link when it ruled that it would pay for TAVR in Medicare beneficiaries only if a heart team was involved from the start. As a result, heart teams are now more tightly and consistently entwined with TAVR than they are with revascularization, where the use of a heart team tracks closely with the increased complexity of a patient’s coronary disease.
Broadening use of heart teams also makes them a little harder to define, as the interventionalist and surgeon collaboration on TAVR involves different jobs and different players than does coronary revascularization. The role heart teams play in early 2013 for U.S. revascularization of coronary disease remains a bit fuzzy. Every interventional cardiologist and cardiac surgeon have their sense of how they use heart teams and how other physicians and surgeons at their hospitals use them for revascularization cases, but a global accounting of the extent to which the heart team concept has penetrated American practice is totally lacking, and will likely stay that way.
Revascularization heart teams remain poorly defined
"The notion of encouraging multidisciplinary care in cardiovascular medicine, exemplified by the heart team concept has gained increasing traction" for revascularization, TAVR, and other complex endovascular interventions, Dr. Brahmajee K. Nallamothu and Dr. David J. Cohen wrote in a perspective article last year (Circulation Cardiovasc. Qual. Outcomes 2012;5:410-3). Examples of that traction include adding heart teams to guidelines, and sessions that focus on heart teams at meetings, Dr. Nallamothu said in an interview.
But thinking of heart teams as formal entities for coronary disease, as formal as for TAVR, is tricky. "The heart team concept always sounds like a great idea, but the real gap has been in showing people in real-world practice how they can apply the idea of a heart team to front-line decision making about patients undergoing revascularization," said Dr. Nallamothu, an interventional cardiologist at the University of Michigan in Ann Arbor. "We have not shown hospitals and physicians how to do this in a practical way. We need more discussions by multidisciplinary teams about complex patients." Heart teams "have filtered into our guidelines without showing doctors how you do it practically, and even if you can, how do you pay people for doing it?" he said.
But most heart surgeons and cardiologists will tell you that what they typically see "is not a lot of formal heart teams, but a lot of discussions, and that’s only been strengthened as TAVR and other structural heart disease programs have started to be team based," Dr. Nallamothu said.
Informal discussions between cardiologists and surgeons about revascularization cases have, of course, gone on for years, and most seem comfortable with it staying that way.
"In community hospitals, patients without complex CAD [coronary artery disease] do not routinely have cardiac surgery consultations. The traditional model is that when a patient needs surgery, the cardiologist consults a surgeon," said Dr. David C. May, an interventional cardiologist in Coppell, Tex.
"Ad hoc or provisional stenting remains the norm in about half the cases. It’s important to separate the complex and simple cases. Most ad hoc PCI is not done in complex cases. Complex cases are done as scheduled, planned procedures," Dr. May said in an interview.
"In the real world, cardiologists see surgeons all the time, every day. We interact all the time. Collegial discussions about patients are always on the table. Most of these consultations are off the cuff and not official," agreed Dr. Peter C. Block, an interventional cardiologist at Emory University in Atlanta.
"If a patient is clearly a PCI candidate, with a SYNTAX score of 15 and no diabetes, I will do PCI" without a surgical consult. "If the patient has a SYNTAX score of 28 or 30 and I think he is not a good patient for PCI, I’ll discuss it with my surgical colleagues. If we agree, I’ll either do the PCI or pass off the patient for surgery. It would be very difficult to get a surgical consult for every U.S. patient who gets PCI," Dr. Block said.
Waiting for "the pause"
But is this approach to heart team use consistent with existing guidelines, and do cardiologists usually make the right call on when they should seek a surgical consult? A critical part is what some people call "the pause," or "taking a patient off the table." An interventional cardiologist can either perform a coronary catheterization for diagnostic purposes only, obtain the patient’s coronary angiogram, and then halt the catheterization procedure while the angiogram films are reviewed by the cardiologist and surgeon, or the cardiologist can elect to continue on after angiography and proceed directly to PCI.
"Many cardiologists don’t believe a pause is necessary. If you ask most U.S. cardiologists, they will tell you that they are comfortable making the decision to stent on their own, that they don’t need a surgeon engaged in the conversation," said heart surgeon Timothy J. Gardner, medical director of the center for heart and vascular health at Christiana Care Health System in Wilmington, Del. "They don’t want to delay the stenting to another catheterization if they believe that patient needs PCI."
But at least one cardiac surgeon believes that postangiogram pauses are on the rise.
"We are definitely seeing a pause after angiography a lot more," said Dr. Mathew R. Williams, a cardiac surgeon and interventional cardiologist and codirector of the heart valve center at Columbia University in New York. "Rather than doing PCI at the time of diagnostic cath, patients are coming off the table, even when we feel the patient should have PCI. Not every case, but patients are coming off the table and that discussion is occurring for patients with complex disease, with left main and LAD [left anterior descending] involvement," Dr. Williams said.
Paying for the consult
Reimbursement is another potential challenge to the viability of surgical consults for revascularization patients.
"It’s a challenge to get people in a busy community practice together as a heart team. They are their own business entity. How do you create a heart team across these business entities?" asked Dr. Thoralf M. Sundt III, chief of cardiac surgery at Massachusetts General Hospital in Boston. "My view of community practice is limited, but I think it’s a real challenge for most community practices to do it. People agree with the heart team concept in theory, but executing it in communities will be a huge challenge."
Dr. Nallamothu of the University of Michigan said, "I’m at an academic center, and for the most part we are salaried. A lot of the heart team discussions are built into our academic responsibilities. For heart teams to really take off in the community, we need to understand how we are going to do it. You need a mechanism to value the work of evaluating cases and rendering opinions about treatment. You can’t expect physicians to do that pro bono. I think it will be a disincentive for full engagement and cooperation unless you can figure out" how to compensate physicians for just rendering their opinions.
"Community hospitals are all over the place because many community hospitals have a bunch of private groups and it’s been a challenge to get them to form heart teams. It’s very politically charged. At other community hospitals the physicians are employees of the hospital. They do a better job because they already have financial alignment," said Dr. Williams.
"Everyone recognizes that where we need to be is organized by service lines and not by academic departments," said Dr. Sundt. "A patient with heart disease wants to be cared for by physicians who deal with heart disease. The patient doesn’t care if they are cardiologists or surgeons, or who is in this group or that group. The patient needs his disease managed by physicians who focus on the patient. That’s what the heart team is. The way to reimburse it is to put all the money into the heart team bucket and then figure out how to distribute it. If CMS set up reimbursement [for revascularization] so that it depended on a heart team" as it did for TAVR, then revascularization decisions would more consistently involve heart teams, he said in an interview.
"Right now, no one pays for [a consult between a cardiologist and surgeon]. The thing that would change this the quickest would be payment reform that rewarded the [heart team] approach," said Dr. W. Douglas Weaver, medical director of the heart and vascular institute at Henry Ford Health System in Detroit.
"Employment by hospitals has driven the process of cardiologists working with surgeons on revascularization cases," Dr. Weaver added. Reimbursement issues could be resolved to some extent by more episode-based payments. What will also drive it is more integration among physicians."
But while compensation for a heart team consult on revascularization might help, and while better-integrated health systems would streamline the delivery of multidisciplinary care, inadequate compensation for participation on heart teams has not been a major roadblock up to now, many say.
"It would be awful if financial misalignment interferes with making the best decisions for patients, but I think the vast majority of cardiologists and cardiac surgeons rise to the occasion" and provide consultations even when they are not paid for it, said Dr. Patrick T. O’Gara, director of clinical cardiology at Brigham and Women’s Hospital in Boston.
"I don’t think reimbursement drives practice. Most cardiologists and surgeons try to do what’s right for the patient. They understand that sometimes they will not get reimbursed," said Dr. Gardner.
TAVR leads the way
Reimbursement for heart team members in revascularization cases may not currently be as well structured as it is for TAVR cases, but in other respects the growing number of TAVR cases and the heart teams that form to treat those cases seem to be having a spillover effect on revascularization. The TAVR experience "has catapulted the collaboration between cardiologists and surgeons in all decision making," said Dr. Weaver.
"A heart team that works together on TAVR sets the stage for collegiality" in revascularization cases, said Dr. Block. "My suspicion is that TAVR relationships will lead to closer communication [between cardiologists and surgeons] because they will see each other more often and have closer ties. The barriers [between cardiologists and surgeons] are breaking down. I don’t know how far that has gone in community hospitals, because TAVR is just beginning at most community hospitals."
It’s reached the community of Dr. May, who has been performing TAVR for a while in his Dallas-area practice.
TAVR programs "foster a significantly improved collaboration between the cardiac surgeons and cardiologists. At a higher level, it has also fostered a much closer relationship between STS and ACC because of their need to work together on guidelines and registry development. So in that regard TAVR has made a big difference," said Dr. May.
"When TAVR is done, everyone [cardiologist and surgeon] is in the room doing a piece of the work. When you talk about collaborative revascularization, you are talking about a decision. So the collaboration is different. With TAVR you work together and develop a combat-level closeness because of the care you all deliver to patients. For revascularization, it is a decision made over a cup of coffee, not in the cath lab," he added.
Dr. May and several others think the collaborative boost powered by TAVR heart teams, and perhaps reinforced in revascularization cases, has the potential to catalyze a more profound shift in cardiac care and professional relationships.
"The experience with TAVR has shown that the most fruitful way for cardiologists and surgeons to go forward is to work collaboratively," continued Dr. May. "That’s a new paradigm, and it needs to be embraced wholeheartedly. It also promotes the idea of doing what is best for patients. It’s not that cardiologists do PCI and surgeons do CABG and we don’t talk to each other. The paradigm shift we have seen with TAVR has tremendous merit."
"It’s time to bury the old PCI vs. CABG battle of the past," said Dr. Williams. If cardiologists and surgeons don’t work together, "it’s not good for them or for patients. When you work together, it’s mutually beneficial, and most importantly it’s better for patients."
What’s next for revascularization heart teams?
Have heart teams for revascularization become the standard of care, is there consensus on when they should be used, and do most U.S. interventionalists and cardiac surgeons already belong to a heart team? The short answer to all three questions is maybe.
"Heart teams may be the standard of care at many [U.S.] institutions, but I can’t speak for all. At Brigham and Women’s, it is our standard of care for patients with complicated CAD," said Dr. O’Gara. Right now "heart teams are the aspiration, but I’m not sure it is the standard. But it was a class I recommendation that had unanimous consensus behind it.
"The ACC has focused on education to proselytize the value of this approach," said Dr. O’Gara, who is currently ACC vice president. The results of the SYNTAX trial established the expectation that among patients with multivessel disease, there would be a discussion before surgeons, interventionalists, and the patient about the optimal [revascularization] approach. But aside from the educational aspect on the benefit of doing this I’m not aware of any regulatory agency that is embedding heart teams in, say, a performance metric," the better to solidify their role in practice.
Most surgeons and cardiologists fall back on calling for a heart team for any patients with "complex" coronary disease, generally defined as patients with left main disease or triple-vessel advanced coronary disease. Those were the categories of coronary disease for which PCI’s use was deemed "uncertain" by the revascularization appropriate-use criteria established last year by the ACC, STS, and several other cardiology and surgical groups (J. Am. Coll. Cardiol. 2012;59:857-81).
"I wouldn’t say that for revascularization using heart teams is the standard of care, but it’s being used in more and more places," said Dr. Weaver. A heart team "is not for every case; it takes time, and in most cases it probably won’t change the decision. But certainly for complex cases it should be routine. At many centers it is routine – but for a small subset of patients," said Dr. Weaver. "The ACC can help define what is a complex case and help convince payers that this is useful and that we need to reward this behavior as an incentive."
In general, cardiologists and surgeons likely feel little urgency to become part of revascularization heart teams, if for no other reason than most of them probably believe they are already part of one.
"I would not be surprised if most American cardiologists and surgeons feel that they are already on a heart team. They feel: ‘I practice at a place with a surgeon, or with a cardiologist – we’re a team.’ I don’t think that anyone in private practice loses sleep because they think they are not on a heart team," said Dr. Nallamothu.
But there is probably a need for a more systematic application of heart teams to appropriate cases, a clearer definition of appropriate cases, and increased leadership from professional societies to accomplish these goals, according to at least some experts.
"I wouldn’t say that societies have fallen short, but there is a lot of work to do. It’s a huge challenge, and these groups could definitely lead going forward," said Dr. Sundt.
"Societies need to take the lead on defining optimal care" and the collaborative approach to revascularization, said Dr. May. The collaborative discussions that the ACC and STS have had about TAVR is "a model" for future collaborations on revascularization and other areas of multidisciplinary cardiac care, such as mitral valve replacement.
"In the long term, I think there will be more integration of interventional cardiology and cardiac surgery, with more overlap of the procedures they use," predicted Dr. Nallamothu. As that happens, "we’ll also see more team discussions about complex revascularization cases."
Dr. Nallamothu, Dr. May, Dr. Block, Dr. Gardner, Dr. Williams, Dr. Sundt, Dr. Weaver, and Dr. O’Gara all said that they had no relevant financial disclosures.
m.zoler@elsevier.com
On Twitter @mitchelzoler
New respiratory coronavirus shows concerning SARS echoes
The short-lived, worldwide epidemic of severe acute respiratory syndrome that began its streak across the globe 10 years ago, starting in February 2003, has been echoed over the past 8 months by what is so far a much more limited number of cases of a new, mysterious respiratory virus closely related to the SARS pathogen.
Mostly known so far as the "novel coronavirus" (NCoV), the new agent is very similar to the severe acute respiratory syndrome (SARS)-associated coronavirus, and by late February the new virus had been identified in 13 patients worldwide – in Saudi Arabia, Jordan, the United Kingdom, and Qatar – causing seven deaths and severe illness in five of the other six patients.
Since the World Health Organization (WHO) and other epidemiology groups first became aware of the NCoV last September, and as the number of identified cases has inched up, researchers have scrambled to gather information about the novel virus and heighten surveillance for new cases. SARS left a legacy of just over 8,000 probable cases in 29 countries, including 29 probable U.S. cases, with an overall fatality rate of 10%. After bursting on the scene in early 2003, SARS quickly flamed out, with the last handful of clinical cases ever seen identified in China in early 2004.
"It’s likely we will see more" of the NCoV. "We’ll need to cast a wide net since we now know there are a dozen cases and different clinical presentations," Dr. Larry Madoff said at the International Meeting on Emerging Diseases and Surveillance. Although almost every patient with confirmed NCoV infection has had severe illness, one U.K. patient who acquired the infection from another household member had a mild, flulike illness. The milder case "calls into question the [WHO] established case definition for this illness," said Dr. Madoff, director of epidemiology and immunization at the Massachusetts Department of Public Health in Boston and chairman of the meeting’s program committee.
In the days after Dr. Madoff made that assessment at the meeting, the WHO on Feb. 21 reported the 13th case, a patient in Saudi Arabia first hospitalized in late January who died in mid-February.
An effort to track down the origin of the NCoV led to the area surrounding the Saudi Arabian city of Bisha, home of the first identified Saudi case. Suspecting a bat origin for the virus because the SARS-associated virus had been identified as a bat virus (although initial human transmission came via a civet), and because the human isolates of the NCoV could infect several bat-cell lines in the lab, U.S. epidemiologist Jonathan Epstein, D.V.M., led a team that surveyed bats from the Bisha area last fall to see if they could find coronaviruses similar to NCoV. Researchers previously reported finding NCoV-like viruses in bats from other locations that are related to the bats that live in the Bisha area, Dr. Epstein said at the meeting.*
"Everywhere you look in bats there seem to be one or two new coronaviruses, but most bat coronaviruses are not SARS-like. The novel coronavirus is the second bat coronavirus in addition to the SARS virus to cause a human infection," said Dr. Epstein, vice president for conservation medicine at EcoHealth Alliance, an environmental medical research organization based in New York.
"Based on published evidence, there is close relatedness between the novel coronavirus and the SARS virus, but SARS uses a receptor that is deep in the respiratory tract. The novel virus uses a different receptor. It’s not clear whether the novel virus can infect mucosa or the upper respiratory tract, but if it could it might be more transmissible," Dr. Epstein said in an interview.
A report from a team of Swiss researchers published on Feb. 19 (subsequent to Dr. Epstein’s comments) reported that the NCoV (which the Swiss researchers call human coronavirus [HCoV]-EMC) grew very efficiently in vitro on human bronchial epithelial cells, and that interferon treatment cut replication of the virus in these cells (mBio 2013;4:e00611-2).
The episodes of human infection by the NCoV so far that seem to be geographically disparate also have a precedent with SARS.
The pattern of cases "suggests that the source of the infection is common or widespread," Dr. Epstein said. "It’s a challenge to identify common environmental features in the case histories of the infected patients. But with SARS there were multiple spill-over events in different regions of southeast China" when the SARS virus moved from civets into people.
"There clearly are many unknowns about the epidemiology" of the NCoV, Dr. Marjorie R. Pollack, a consultant medical epidemiologist based in New York, wrote in a recent comment on the new coronavirus (ProMed Mail, 2013;Archive Number: 20130221.1554109). "Genetic studies on this NCoV place it related to coronaviruses found in bats. But how did the jump from bats to humans occur? Is there an intermediate host animal?"
According to recent guidance from the WHO, member states should "continue their surveillance for severe acute respiratory infections and to carefully review any unusual patterns. Testing for the NCoV should be considered in patients with unexplained pneumonias or in patients with unexplained severe, progressive or complicated respiratory illness not responding to treatment, particularly in persons traveling from or resident in areas of the world known to be affected."
Dr. Madoff, Dr. Epstein, and Dr. Pollack had no disclosures.
On Twitter @mitchelzoler
*Correction, 3/1/2013: In an earlier version of this story, the findings regarding the Saudi Arabian bats sampled by Dr. Jonathan Epstein and his associates were misstated. Dr. Epstein and his colleagues led a team that surveyed bats from the Bisha area last fall. They have not yet reported their findings.
The short-lived, worldwide epidemic of severe acute respiratory syndrome that began its streak across the globe 10 years ago, starting in February 2003, has been echoed over the past 8 months by what is so far a much more limited number of cases of a new, mysterious respiratory virus closely related to the SARS pathogen.
Mostly known so far as the "novel coronavirus" (NCoV), the new agent is very similar to the severe acute respiratory syndrome (SARS)-associated coronavirus, and by late February the new virus had been identified in 13 patients worldwide – in Saudi Arabia, Jordan, the United Kingdom, and Qatar – causing seven deaths and severe illness in five of the other six patients.
Since the World Health Organization (WHO) and other epidemiology groups first became aware of the NCoV last September, and as the number of identified cases has inched up, researchers have scrambled to gather information about the novel virus and heighten surveillance for new cases. SARS left a legacy of just over 8,000 probable cases in 29 countries, including 29 probable U.S. cases, with an overall fatality rate of 10%. After bursting on the scene in early 2003, SARS quickly flamed out, with the last handful of clinical cases ever seen identified in China in early 2004.
"It’s likely we will see more" of the NCoV. "We’ll need to cast a wide net since we now know there are a dozen cases and different clinical presentations," Dr. Larry Madoff said at the International Meeting on Emerging Diseases and Surveillance. Although almost every patient with confirmed NCoV infection has had severe illness, one U.K. patient who acquired the infection from another household member had a mild, flulike illness. The milder case "calls into question the [WHO] established case definition for this illness," said Dr. Madoff, director of epidemiology and immunization at the Massachusetts Department of Public Health in Boston and chairman of the meeting’s program committee.
In the days after Dr. Madoff made that assessment at the meeting, the WHO on Feb. 21 reported the 13th case, a patient in Saudi Arabia first hospitalized in late January who died in mid-February.
An effort to track down the origin of the NCoV led to the area surrounding the Saudi Arabian city of Bisha, home of the first identified Saudi case. Suspecting a bat origin for the virus because the SARS-associated virus had been identified as a bat virus (although initial human transmission came via a civet), and because the human isolates of the NCoV could infect several bat-cell lines in the lab, U.S. epidemiologist Jonathan Epstein, D.V.M., led a team that surveyed bats from the Bisha area last fall to see if they could find coronaviruses similar to NCoV. Researchers previously reported finding NCoV-like viruses in bats from other locations that are related to the bats that live in the Bisha area, Dr. Epstein said at the meeting.*
"Everywhere you look in bats there seem to be one or two new coronaviruses, but most bat coronaviruses are not SARS-like. The novel coronavirus is the second bat coronavirus in addition to the SARS virus to cause a human infection," said Dr. Epstein, vice president for conservation medicine at EcoHealth Alliance, an environmental medical research organization based in New York.
"Based on published evidence, there is close relatedness between the novel coronavirus and the SARS virus, but SARS uses a receptor that is deep in the respiratory tract. The novel virus uses a different receptor. It’s not clear whether the novel virus can infect mucosa or the upper respiratory tract, but if it could it might be more transmissible," Dr. Epstein said in an interview.
A report from a team of Swiss researchers published on Feb. 19 (subsequent to Dr. Epstein’s comments) reported that the NCoV (which the Swiss researchers call human coronavirus [HCoV]-EMC) grew very efficiently in vitro on human bronchial epithelial cells, and that interferon treatment cut replication of the virus in these cells (mBio 2013;4:e00611-2).
The episodes of human infection by the NCoV so far that seem to be geographically disparate also have a precedent with SARS.
The pattern of cases "suggests that the source of the infection is common or widespread," Dr. Epstein said. "It’s a challenge to identify common environmental features in the case histories of the infected patients. But with SARS there were multiple spill-over events in different regions of southeast China" when the SARS virus moved from civets into people.
"There clearly are many unknowns about the epidemiology" of the NCoV, Dr. Marjorie R. Pollack, a consultant medical epidemiologist based in New York, wrote in a recent comment on the new coronavirus (ProMed Mail, 2013;Archive Number: 20130221.1554109). "Genetic studies on this NCoV place it related to coronaviruses found in bats. But how did the jump from bats to humans occur? Is there an intermediate host animal?"
According to recent guidance from the WHO, member states should "continue their surveillance for severe acute respiratory infections and to carefully review any unusual patterns. Testing for the NCoV should be considered in patients with unexplained pneumonias or in patients with unexplained severe, progressive or complicated respiratory illness not responding to treatment, particularly in persons traveling from or resident in areas of the world known to be affected."
Dr. Madoff, Dr. Epstein, and Dr. Pollack had no disclosures.
On Twitter @mitchelzoler
*Correction, 3/1/2013: In an earlier version of this story, the findings regarding the Saudi Arabian bats sampled by Dr. Jonathan Epstein and his associates were misstated. Dr. Epstein and his colleagues led a team that surveyed bats from the Bisha area last fall. They have not yet reported their findings.
The short-lived, worldwide epidemic of severe acute respiratory syndrome that began its streak across the globe 10 years ago, starting in February 2003, has been echoed over the past 8 months by what is so far a much more limited number of cases of a new, mysterious respiratory virus closely related to the SARS pathogen.
Mostly known so far as the "novel coronavirus" (NCoV), the new agent is very similar to the severe acute respiratory syndrome (SARS)-associated coronavirus, and by late February the new virus had been identified in 13 patients worldwide – in Saudi Arabia, Jordan, the United Kingdom, and Qatar – causing seven deaths and severe illness in five of the other six patients.
Since the World Health Organization (WHO) and other epidemiology groups first became aware of the NCoV last September, and as the number of identified cases has inched up, researchers have scrambled to gather information about the novel virus and heighten surveillance for new cases. SARS left a legacy of just over 8,000 probable cases in 29 countries, including 29 probable U.S. cases, with an overall fatality rate of 10%. After bursting on the scene in early 2003, SARS quickly flamed out, with the last handful of clinical cases ever seen identified in China in early 2004.
"It’s likely we will see more" of the NCoV. "We’ll need to cast a wide net since we now know there are a dozen cases and different clinical presentations," Dr. Larry Madoff said at the International Meeting on Emerging Diseases and Surveillance. Although almost every patient with confirmed NCoV infection has had severe illness, one U.K. patient who acquired the infection from another household member had a mild, flulike illness. The milder case "calls into question the [WHO] established case definition for this illness," said Dr. Madoff, director of epidemiology and immunization at the Massachusetts Department of Public Health in Boston and chairman of the meeting’s program committee.
In the days after Dr. Madoff made that assessment at the meeting, the WHO on Feb. 21 reported the 13th case, a patient in Saudi Arabia first hospitalized in late January who died in mid-February.
An effort to track down the origin of the NCoV led to the area surrounding the Saudi Arabian city of Bisha, home of the first identified Saudi case. Suspecting a bat origin for the virus because the SARS-associated virus had been identified as a bat virus (although initial human transmission came via a civet), and because the human isolates of the NCoV could infect several bat-cell lines in the lab, U.S. epidemiologist Jonathan Epstein, D.V.M., led a team that surveyed bats from the Bisha area last fall to see if they could find coronaviruses similar to NCoV. Researchers previously reported finding NCoV-like viruses in bats from other locations that are related to the bats that live in the Bisha area, Dr. Epstein said at the meeting.*
"Everywhere you look in bats there seem to be one or two new coronaviruses, but most bat coronaviruses are not SARS-like. The novel coronavirus is the second bat coronavirus in addition to the SARS virus to cause a human infection," said Dr. Epstein, vice president for conservation medicine at EcoHealth Alliance, an environmental medical research organization based in New York.
"Based on published evidence, there is close relatedness between the novel coronavirus and the SARS virus, but SARS uses a receptor that is deep in the respiratory tract. The novel virus uses a different receptor. It’s not clear whether the novel virus can infect mucosa or the upper respiratory tract, but if it could it might be more transmissible," Dr. Epstein said in an interview.
A report from a team of Swiss researchers published on Feb. 19 (subsequent to Dr. Epstein’s comments) reported that the NCoV (which the Swiss researchers call human coronavirus [HCoV]-EMC) grew very efficiently in vitro on human bronchial epithelial cells, and that interferon treatment cut replication of the virus in these cells (mBio 2013;4:e00611-2).
The episodes of human infection by the NCoV so far that seem to be geographically disparate also have a precedent with SARS.
The pattern of cases "suggests that the source of the infection is common or widespread," Dr. Epstein said. "It’s a challenge to identify common environmental features in the case histories of the infected patients. But with SARS there were multiple spill-over events in different regions of southeast China" when the SARS virus moved from civets into people.
"There clearly are many unknowns about the epidemiology" of the NCoV, Dr. Marjorie R. Pollack, a consultant medical epidemiologist based in New York, wrote in a recent comment on the new coronavirus (ProMed Mail, 2013;Archive Number: 20130221.1554109). "Genetic studies on this NCoV place it related to coronaviruses found in bats. But how did the jump from bats to humans occur? Is there an intermediate host animal?"
According to recent guidance from the WHO, member states should "continue their surveillance for severe acute respiratory infections and to carefully review any unusual patterns. Testing for the NCoV should be considered in patients with unexplained pneumonias or in patients with unexplained severe, progressive or complicated respiratory illness not responding to treatment, particularly in persons traveling from or resident in areas of the world known to be affected."
Dr. Madoff, Dr. Epstein, and Dr. Pollack had no disclosures.
On Twitter @mitchelzoler
*Correction, 3/1/2013: In an earlier version of this story, the findings regarding the Saudi Arabian bats sampled by Dr. Jonathan Epstein and his associates were misstated. Dr. Epstein and his colleagues led a team that surveyed bats from the Bisha area last fall. They have not yet reported their findings.
AT IMED2013
Major Finding: As of Feb. 21, 13 people had been identified as infected with a novel coronavrius since July 2012, with seven deaths.
Data Source: World Health Organization.
Disclosures: Dr. Madoff, Dr. Epstein, and Dr. Pollack had no disclosures.
Tympanic membrane now keys otitis media diagnosis
A new, more stringent definition of acute otitis media that focuses on tympanic membrane appearance on otoscopy and downplays the diagnostic role of symptoms highlighted the new acute otitis media diagnosis and management guidelines released by the American Academy of Pediatrics on Feb. 25.
The new guidelines, the first revision since 2004, also reaffirmed and expanded the option of observation without treatment for selected children with acute otitis media (AOM) as an alternative to immediate prescription of an antibiotic. Introduction of observation as a viable alternative to antibiotics had been the major feature of the 2004 guidelines.
"The most important change from 2004 is the working definition of AOM. The appearance of the tympanic membrane [TM] is the key to an accurate diagnosis," said Dr. Allan S. Lieberthal, a pediatrician with Kaiser-Permanente in Panorama City, Calif., who chairs the AAP subcommittee on diagnosis and management of acute otitis media, which wrote the new guidelines revision (Pediatrics 2013;131;e964-e99).
"Fever, fussiness, ear pain, or a combination may be the usual reason for a doctor visit, but research done in the last several years has shown that symptoms do not differentiate AOM from an upper respiratory infection or other minor illnesses. With the requirement of a bulging TM, there should no longer be uncertainty in diagnosis," Dr. Lieberthal said.
The guidelines also emphasize pneumatic otoscopy to ensure the presence of a middle-ear effusion, calling the pneumatic otoscope the "standard tool" for diagnosing otitis media. Despite that, "relatively few physicians consistently use pneumatic otoscopy, and they may be resistant to changing their practice," Dr. Lieberthal said in an interview.
The second major change of the new guidelines is expansion of the observation option to selected children aged 6-24 months if they have unilateral AOM without otorrhea and if the child’s parent or caregiver understands and agrees with the option. Children this young with bilateral AOM, severe symptoms, or otorrhea are not recommended as candidates for deferred treatment and observation. The new guidelines also reaffirmed the observation option first recommended in 2004 for selected children older than 2 years if they have either unilateral or bilateral AOM but without otorrhea or severe symptoms.
Recent research results "reinforced the success of observation, especially in children older than 24 months who are not severely ill," said Dr. Lieberthal. "In most studies, 70% of children treated with initial observation did not require subsequent antibiotic treatment."
The guidelines also stress two important caveats: The family should be included in decisions regarding the use of observation for initial therapy, and a mechanism must be in place to ensure follow-up so that an antibiotic regimen can start if the child worsens or fails to improve within 48-72 hours after symptom onset.
Results from "two major studies using a highly specific definition of AOM in younger children showed significant improvement in patients treated with antibiotics, compared with those who were observed. The panel acknowledged this by continuing to recommend initial antibiotic treatment in children younger than 24 months who have a bulging TM. However, if the bulging is minor and the child does not appear ill, then initial observation may be used if the family agrees, and if a mechanism is in place to ensure follow-up," he said.
"Due to widespread discussion [about the observation option] in consumer media after the 2004 guidelines, parents now often ask if observation is recommended, and a growing number of pediatricians use observation," Dr. Lieberthal said, adding that "family and emergency physicians have been more resistant" to applying the observation option.
The new guidelines made no change in the recommended antibiotic regimens, with amoxicillin the top choice and amoxicillin-clavulanate an alternative for either initial or delayed treatment. For children who fail to improve after 48-72 hours of treatment, the guidelines call for switching to amoxicillin-clavulanate or ceftriaxone. The guidelines also include recommended options for children with penicillin allergy.
The new guidelines also made no changes from the 2004 recommendations for pain management, with acetaminophen and ibuprofen flagged as the "mainstays" for treating AOM pain.
This year’s guidelines also include recommendations that physicians check that their patients are up to date on their pneumococcal conjugate and influenza vaccines, that mothers are encouraged to breastfeed their infants for at least 6 months, and that physicians encourage tobacco smoke avoidance.
The guidelines also address dealing with children who develop recurrent AOM, a topic not included in the 2004 version. Recurrent AOM is defined as three episodes in 6 months or four episodes in 1 year, with one episode in the preceding 6 months. The new guidelines specifically say that clinicians should not prescribe any prophylactic antibiotics but may offer tympanostomy tubes. "Many physicians no longer use prophylactic antibiotics," Dr. Lieberthal noted. "Hopefully, the guidelines will reduce the use of unnecessary tubes," he added.
The AAP first assembled the panel to update the AOM guidelines in 2009, and the members included general pediatricians, pediatric infectious diseases specialists, otolaryngologists, an emergency medicine physician, and a family physician. The panel worked with the U.S. Agency for Healthcare Research and Quality and the Southern California Evidence-Based Research Center for its literature review. The panel designed the guidelines for children aged 6 months to 12 years who are otherwise healthy, and they include 17 individual action statements for clinicians.
The guidelines also included statements about uptake of the 2004 guidelines: "Despite significant publicity and awareness of the 2004 AOM guideline evidence shows that clinicians are hesitant to follow the guidelines recommendations." The panel added that, "for clinical practice guidelines to be effective more must be done to improve their dissemination and implementation."
When asked about these statements, Dr. Lieberthal said, "Most physicians find it difficult to change their long-standing practices. The AAP is working on an education and implementation plan" for the 2013 revision.
The revised clinical practice guidelines for acute otitis media were sponsored by the American Academy of Pediatrics. Dr. Lieberthal said that he had no relevant financial disclosures.
On Twitter @mitchelzoler
Dr. Stephen I. Pelton* comments:
The guidelines address five important areas and provide update,
clarification, and greater granularity than the original guidelines did.
They emphasize the diagnostic criteria, and strengthen
the significance of bulging as a diagnostic requirement for AOM as well as the
requirement for the presence of middle ear effusion. This will be challenging
as many clinicians are not well trained in the use of pneumatic otoscopy. The
guidelines were not written to apply to infants younger than 6 months, and in
the youngest infants, tympanometry and acoustic reflectometry may be less
accurate.
The initial approach remains assessment of the need
for antibiotic treatment based on age and severity, as well as assessment of
pain and appropriate management of pain. The choice between antibiotic
management and observation is further clarified in these guidelines based on
current evidence. The new guidelines discuss but do not highlight the results
of two recent randomized clinical trials that showed that half of the children
in the placebo group did not have a satisfactory resolution. Presumably, this
information should be included in any discussion with parents about the role
for antibiotics in treating young children with AOM. The criteria recommended
for the routine use of antibiotics in children less than 2 years old –
bilateral disease, acute otorrhea, or high fever – all are supported by
evidence.
The choice of amoxicillin or amoxicillin-clavulanate
as the first-line antibiotics is appropriate. The lack of any recommendation
for azithromycin will hopefully help educate clinicians about its limited
activity against Haemophilus in children with culture-positive AOM and
the significant prevalence of macrolide-resistant pneumococci in the community.
The pneumococcal conjugate vaccine has proven
beneficial, both for preventing AOM and recurrent AOM, and for obviating
tympanostomy tube insertion. It is important that the vaccine be administered
early in life, before recurrent AOM can develop. Influenza vaccine also is
valuable during influenza seasons for preventing flu and its AOM complication.
Although the guidelines take an absolute approach
against antibiotic prophylaxis, there may be some specific children where it
may still have value despite concerns about promoting resistance through
selective pressure.
Dr. Stephen I. Pelton is chief of pediatric
infectious disease and also is the coordinator of the maternal-child HIV
program at Boston
Medical Center.
He was not involved in development of the AAP guidelines and responded to a
request to comment on the guidelines.
Updated: 3/4/13 Dr. Pelton's misspelled first name corrected
Dr. Stephen I. Pelton* comments:
The guidelines address five important areas and provide update,
clarification, and greater granularity than the original guidelines did.
They emphasize the diagnostic criteria, and strengthen
the significance of bulging as a diagnostic requirement for AOM as well as the
requirement for the presence of middle ear effusion. This will be challenging
as many clinicians are not well trained in the use of pneumatic otoscopy. The
guidelines were not written to apply to infants younger than 6 months, and in
the youngest infants, tympanometry and acoustic reflectometry may be less
accurate.
The initial approach remains assessment of the need
for antibiotic treatment based on age and severity, as well as assessment of
pain and appropriate management of pain. The choice between antibiotic
management and observation is further clarified in these guidelines based on
current evidence. The new guidelines discuss but do not highlight the results
of two recent randomized clinical trials that showed that half of the children
in the placebo group did not have a satisfactory resolution. Presumably, this
information should be included in any discussion with parents about the role
for antibiotics in treating young children with AOM. The criteria recommended
for the routine use of antibiotics in children less than 2 years old –
bilateral disease, acute otorrhea, or high fever – all are supported by
evidence.
The choice of amoxicillin or amoxicillin-clavulanate
as the first-line antibiotics is appropriate. The lack of any recommendation
for azithromycin will hopefully help educate clinicians about its limited
activity against Haemophilus in children with culture-positive AOM and
the significant prevalence of macrolide-resistant pneumococci in the community.
The pneumococcal conjugate vaccine has proven
beneficial, both for preventing AOM and recurrent AOM, and for obviating
tympanostomy tube insertion. It is important that the vaccine be administered
early in life, before recurrent AOM can develop. Influenza vaccine also is
valuable during influenza seasons for preventing flu and its AOM complication.
Although the guidelines take an absolute approach
against antibiotic prophylaxis, there may be some specific children where it
may still have value despite concerns about promoting resistance through
selective pressure.
Dr. Stephen I. Pelton is chief of pediatric
infectious disease and also is the coordinator of the maternal-child HIV
program at Boston
Medical Center.
He was not involved in development of the AAP guidelines and responded to a
request to comment on the guidelines.
Updated: 3/4/13 Dr. Pelton's misspelled first name corrected
Dr. Stephen I. Pelton* comments:
The guidelines address five important areas and provide update,
clarification, and greater granularity than the original guidelines did.
They emphasize the diagnostic criteria, and strengthen
the significance of bulging as a diagnostic requirement for AOM as well as the
requirement for the presence of middle ear effusion. This will be challenging
as many clinicians are not well trained in the use of pneumatic otoscopy. The
guidelines were not written to apply to infants younger than 6 months, and in
the youngest infants, tympanometry and acoustic reflectometry may be less
accurate.
The initial approach remains assessment of the need
for antibiotic treatment based on age and severity, as well as assessment of
pain and appropriate management of pain. The choice between antibiotic
management and observation is further clarified in these guidelines based on
current evidence. The new guidelines discuss but do not highlight the results
of two recent randomized clinical trials that showed that half of the children
in the placebo group did not have a satisfactory resolution. Presumably, this
information should be included in any discussion with parents about the role
for antibiotics in treating young children with AOM. The criteria recommended
for the routine use of antibiotics in children less than 2 years old –
bilateral disease, acute otorrhea, or high fever – all are supported by
evidence.
The choice of amoxicillin or amoxicillin-clavulanate
as the first-line antibiotics is appropriate. The lack of any recommendation
for azithromycin will hopefully help educate clinicians about its limited
activity against Haemophilus in children with culture-positive AOM and
the significant prevalence of macrolide-resistant pneumococci in the community.
The pneumococcal conjugate vaccine has proven
beneficial, both for preventing AOM and recurrent AOM, and for obviating
tympanostomy tube insertion. It is important that the vaccine be administered
early in life, before recurrent AOM can develop. Influenza vaccine also is
valuable during influenza seasons for preventing flu and its AOM complication.
Although the guidelines take an absolute approach
against antibiotic prophylaxis, there may be some specific children where it
may still have value despite concerns about promoting resistance through
selective pressure.
Dr. Stephen I. Pelton is chief of pediatric
infectious disease and also is the coordinator of the maternal-child HIV
program at Boston
Medical Center.
He was not involved in development of the AAP guidelines and responded to a
request to comment on the guidelines.
Updated: 3/4/13 Dr. Pelton's misspelled first name corrected
A new, more stringent definition of acute otitis media that focuses on tympanic membrane appearance on otoscopy and downplays the diagnostic role of symptoms highlighted the new acute otitis media diagnosis and management guidelines released by the American Academy of Pediatrics on Feb. 25.
The new guidelines, the first revision since 2004, also reaffirmed and expanded the option of observation without treatment for selected children with acute otitis media (AOM) as an alternative to immediate prescription of an antibiotic. Introduction of observation as a viable alternative to antibiotics had been the major feature of the 2004 guidelines.
"The most important change from 2004 is the working definition of AOM. The appearance of the tympanic membrane [TM] is the key to an accurate diagnosis," said Dr. Allan S. Lieberthal, a pediatrician with Kaiser-Permanente in Panorama City, Calif., who chairs the AAP subcommittee on diagnosis and management of acute otitis media, which wrote the new guidelines revision (Pediatrics 2013;131;e964-e99).
"Fever, fussiness, ear pain, or a combination may be the usual reason for a doctor visit, but research done in the last several years has shown that symptoms do not differentiate AOM from an upper respiratory infection or other minor illnesses. With the requirement of a bulging TM, there should no longer be uncertainty in diagnosis," Dr. Lieberthal said.
The guidelines also emphasize pneumatic otoscopy to ensure the presence of a middle-ear effusion, calling the pneumatic otoscope the "standard tool" for diagnosing otitis media. Despite that, "relatively few physicians consistently use pneumatic otoscopy, and they may be resistant to changing their practice," Dr. Lieberthal said in an interview.
The second major change of the new guidelines is expansion of the observation option to selected children aged 6-24 months if they have unilateral AOM without otorrhea and if the child’s parent or caregiver understands and agrees with the option. Children this young with bilateral AOM, severe symptoms, or otorrhea are not recommended as candidates for deferred treatment and observation. The new guidelines also reaffirmed the observation option first recommended in 2004 for selected children older than 2 years if they have either unilateral or bilateral AOM but without otorrhea or severe symptoms.
Recent research results "reinforced the success of observation, especially in children older than 24 months who are not severely ill," said Dr. Lieberthal. "In most studies, 70% of children treated with initial observation did not require subsequent antibiotic treatment."
The guidelines also stress two important caveats: The family should be included in decisions regarding the use of observation for initial therapy, and a mechanism must be in place to ensure follow-up so that an antibiotic regimen can start if the child worsens or fails to improve within 48-72 hours after symptom onset.
Results from "two major studies using a highly specific definition of AOM in younger children showed significant improvement in patients treated with antibiotics, compared with those who were observed. The panel acknowledged this by continuing to recommend initial antibiotic treatment in children younger than 24 months who have a bulging TM. However, if the bulging is minor and the child does not appear ill, then initial observation may be used if the family agrees, and if a mechanism is in place to ensure follow-up," he said.
"Due to widespread discussion [about the observation option] in consumer media after the 2004 guidelines, parents now often ask if observation is recommended, and a growing number of pediatricians use observation," Dr. Lieberthal said, adding that "family and emergency physicians have been more resistant" to applying the observation option.
The new guidelines made no change in the recommended antibiotic regimens, with amoxicillin the top choice and amoxicillin-clavulanate an alternative for either initial or delayed treatment. For children who fail to improve after 48-72 hours of treatment, the guidelines call for switching to amoxicillin-clavulanate or ceftriaxone. The guidelines also include recommended options for children with penicillin allergy.
The new guidelines also made no changes from the 2004 recommendations for pain management, with acetaminophen and ibuprofen flagged as the "mainstays" for treating AOM pain.
This year’s guidelines also include recommendations that physicians check that their patients are up to date on their pneumococcal conjugate and influenza vaccines, that mothers are encouraged to breastfeed their infants for at least 6 months, and that physicians encourage tobacco smoke avoidance.
The guidelines also address dealing with children who develop recurrent AOM, a topic not included in the 2004 version. Recurrent AOM is defined as three episodes in 6 months or four episodes in 1 year, with one episode in the preceding 6 months. The new guidelines specifically say that clinicians should not prescribe any prophylactic antibiotics but may offer tympanostomy tubes. "Many physicians no longer use prophylactic antibiotics," Dr. Lieberthal noted. "Hopefully, the guidelines will reduce the use of unnecessary tubes," he added.
The AAP first assembled the panel to update the AOM guidelines in 2009, and the members included general pediatricians, pediatric infectious diseases specialists, otolaryngologists, an emergency medicine physician, and a family physician. The panel worked with the U.S. Agency for Healthcare Research and Quality and the Southern California Evidence-Based Research Center for its literature review. The panel designed the guidelines for children aged 6 months to 12 years who are otherwise healthy, and they include 17 individual action statements for clinicians.
The guidelines also included statements about uptake of the 2004 guidelines: "Despite significant publicity and awareness of the 2004 AOM guideline evidence shows that clinicians are hesitant to follow the guidelines recommendations." The panel added that, "for clinical practice guidelines to be effective more must be done to improve their dissemination and implementation."
When asked about these statements, Dr. Lieberthal said, "Most physicians find it difficult to change their long-standing practices. The AAP is working on an education and implementation plan" for the 2013 revision.
The revised clinical practice guidelines for acute otitis media were sponsored by the American Academy of Pediatrics. Dr. Lieberthal said that he had no relevant financial disclosures.
On Twitter @mitchelzoler
A new, more stringent definition of acute otitis media that focuses on tympanic membrane appearance on otoscopy and downplays the diagnostic role of symptoms highlighted the new acute otitis media diagnosis and management guidelines released by the American Academy of Pediatrics on Feb. 25.
The new guidelines, the first revision since 2004, also reaffirmed and expanded the option of observation without treatment for selected children with acute otitis media (AOM) as an alternative to immediate prescription of an antibiotic. Introduction of observation as a viable alternative to antibiotics had been the major feature of the 2004 guidelines.
"The most important change from 2004 is the working definition of AOM. The appearance of the tympanic membrane [TM] is the key to an accurate diagnosis," said Dr. Allan S. Lieberthal, a pediatrician with Kaiser-Permanente in Panorama City, Calif., who chairs the AAP subcommittee on diagnosis and management of acute otitis media, which wrote the new guidelines revision (Pediatrics 2013;131;e964-e99).
"Fever, fussiness, ear pain, or a combination may be the usual reason for a doctor visit, but research done in the last several years has shown that symptoms do not differentiate AOM from an upper respiratory infection or other minor illnesses. With the requirement of a bulging TM, there should no longer be uncertainty in diagnosis," Dr. Lieberthal said.
The guidelines also emphasize pneumatic otoscopy to ensure the presence of a middle-ear effusion, calling the pneumatic otoscope the "standard tool" for diagnosing otitis media. Despite that, "relatively few physicians consistently use pneumatic otoscopy, and they may be resistant to changing their practice," Dr. Lieberthal said in an interview.
The second major change of the new guidelines is expansion of the observation option to selected children aged 6-24 months if they have unilateral AOM without otorrhea and if the child’s parent or caregiver understands and agrees with the option. Children this young with bilateral AOM, severe symptoms, or otorrhea are not recommended as candidates for deferred treatment and observation. The new guidelines also reaffirmed the observation option first recommended in 2004 for selected children older than 2 years if they have either unilateral or bilateral AOM but without otorrhea or severe symptoms.
Recent research results "reinforced the success of observation, especially in children older than 24 months who are not severely ill," said Dr. Lieberthal. "In most studies, 70% of children treated with initial observation did not require subsequent antibiotic treatment."
The guidelines also stress two important caveats: The family should be included in decisions regarding the use of observation for initial therapy, and a mechanism must be in place to ensure follow-up so that an antibiotic regimen can start if the child worsens or fails to improve within 48-72 hours after symptom onset.
Results from "two major studies using a highly specific definition of AOM in younger children showed significant improvement in patients treated with antibiotics, compared with those who were observed. The panel acknowledged this by continuing to recommend initial antibiotic treatment in children younger than 24 months who have a bulging TM. However, if the bulging is minor and the child does not appear ill, then initial observation may be used if the family agrees, and if a mechanism is in place to ensure follow-up," he said.
"Due to widespread discussion [about the observation option] in consumer media after the 2004 guidelines, parents now often ask if observation is recommended, and a growing number of pediatricians use observation," Dr. Lieberthal said, adding that "family and emergency physicians have been more resistant" to applying the observation option.
The new guidelines made no change in the recommended antibiotic regimens, with amoxicillin the top choice and amoxicillin-clavulanate an alternative for either initial or delayed treatment. For children who fail to improve after 48-72 hours of treatment, the guidelines call for switching to amoxicillin-clavulanate or ceftriaxone. The guidelines also include recommended options for children with penicillin allergy.
The new guidelines also made no changes from the 2004 recommendations for pain management, with acetaminophen and ibuprofen flagged as the "mainstays" for treating AOM pain.
This year’s guidelines also include recommendations that physicians check that their patients are up to date on their pneumococcal conjugate and influenza vaccines, that mothers are encouraged to breastfeed their infants for at least 6 months, and that physicians encourage tobacco smoke avoidance.
The guidelines also address dealing with children who develop recurrent AOM, a topic not included in the 2004 version. Recurrent AOM is defined as three episodes in 6 months or four episodes in 1 year, with one episode in the preceding 6 months. The new guidelines specifically say that clinicians should not prescribe any prophylactic antibiotics but may offer tympanostomy tubes. "Many physicians no longer use prophylactic antibiotics," Dr. Lieberthal noted. "Hopefully, the guidelines will reduce the use of unnecessary tubes," he added.
The AAP first assembled the panel to update the AOM guidelines in 2009, and the members included general pediatricians, pediatric infectious diseases specialists, otolaryngologists, an emergency medicine physician, and a family physician. The panel worked with the U.S. Agency for Healthcare Research and Quality and the Southern California Evidence-Based Research Center for its literature review. The panel designed the guidelines for children aged 6 months to 12 years who are otherwise healthy, and they include 17 individual action statements for clinicians.
The guidelines also included statements about uptake of the 2004 guidelines: "Despite significant publicity and awareness of the 2004 AOM guideline evidence shows that clinicians are hesitant to follow the guidelines recommendations." The panel added that, "for clinical practice guidelines to be effective more must be done to improve their dissemination and implementation."
When asked about these statements, Dr. Lieberthal said, "Most physicians find it difficult to change their long-standing practices. The AAP is working on an education and implementation plan" for the 2013 revision.
The revised clinical practice guidelines for acute otitis media were sponsored by the American Academy of Pediatrics. Dr. Lieberthal said that he had no relevant financial disclosures.
On Twitter @mitchelzoler
FROM PEDIATRICS