User login
Inspection of Deep Tumor Margins for Accurate Cutaneous Squamous Cell Carcinoma Staging
To the Editor:
Histopathologic analysis of debulk specimens in Mohs micrographic surgery (MMS) may augment identification of high-risk factors in cutaneous squamous cell carcinoma (cSCC), which may warrant tumor upstaging.1 Intratumor location has not been studied when looking at these high-risk factors. Herein, we report 4 cSCCs initially categorized as well differentiated that were reclassified as moderate to poorly differentiated on analysis of debulk specimens obtained via shave removal.
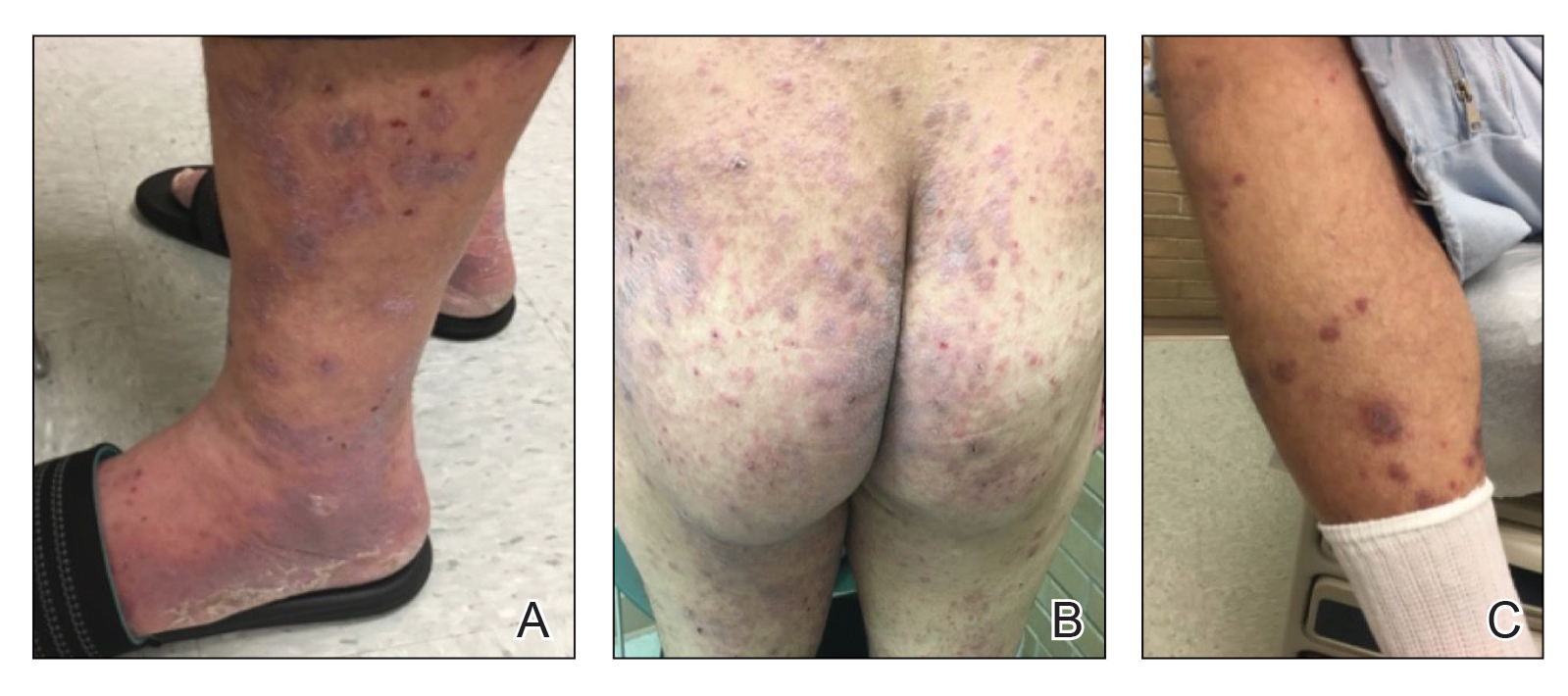
An 80-year-old man (patient 1) presented with a tender 2-cm erythematous plaque with dried hemorrhagic crusting on the frontal scalp. He had a history of nonmelanoma skin cancers. A biopsy revealed a well-differentiated cSCC, which was upgraded from a T2a tumor to T2b during MMS due to galea involvement. Debulk analysis revealed moderate to poorly differentiated cSCC, with the least-differentiated cells at the deep margin (Figure 1A). Given T2b staging, baseline imaging and radiation therapy were recommended.
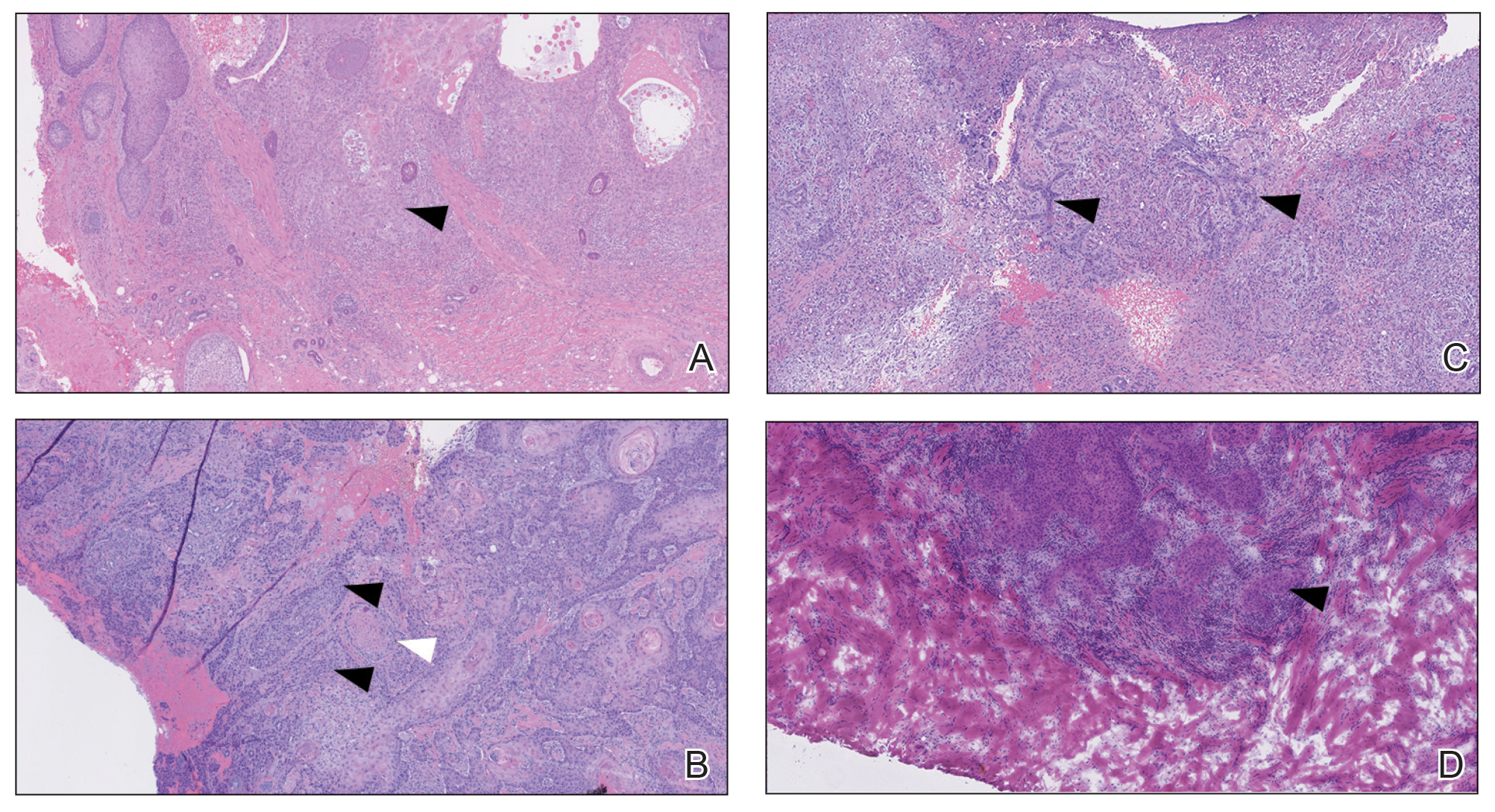
A 75-year-old man (patient 2) presented with a 2-cm erythematous plaque on the left vertex scalp with hemorrhagic crusting, yellow scale, and purulent drainage. He had a history of cSCCs. A biopsy revealed well-differentiated invasive cSCC, which was upgraded from a T2a tumor to T2b during MMS due to tumor extension beyond the subcutaneous fat. Examination of the second Mohs stage revealed moderately differentiated cSCC, with the least-differentiated cells at the deep margin, infiltration beyond the subcutaneous fat, and perineural invasion (Figure 1B). Given T2b staging, baseline imaging and radiation therapy were recommended.
An 86-year-old woman (patient 3) presented with a tender 2.4-cm plum-colored nodule on the right lower leg. She had a history of basal cell carcinoma. A biopsy revealed a well-differentiated invasive cSCC staged at T2a. Debulk analysis revealed moderately differentiated cSCC, with the least-differentiated cells at the deep margin, though the staging remained the same (Figure 1C).
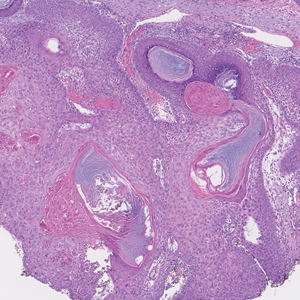
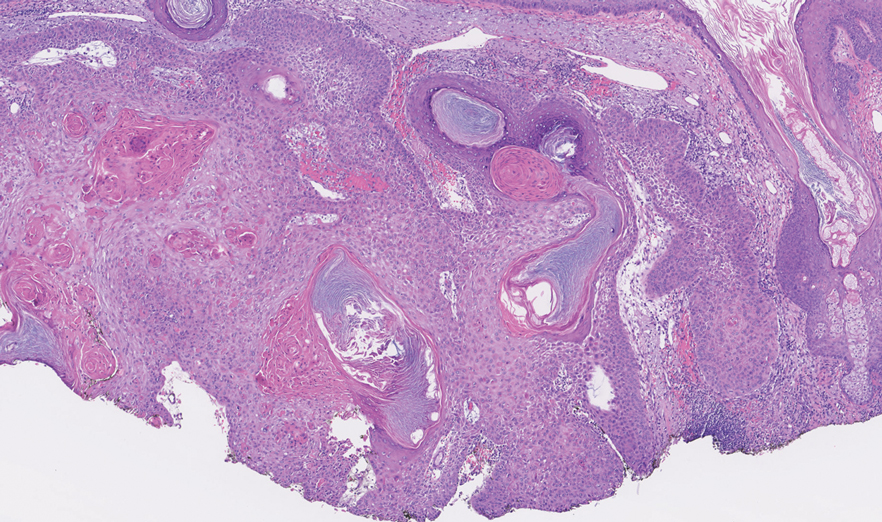
An 82-year-old man (patient 4) presented with a 2.7-cm ulcerated nodule with adjacent scaling on the vertex scalp. He had no history of skin cancer. A biopsy revealed a well-differentiated cSCC (Figure 2) that was upgraded from a T2a tumor to T2b during MMS due to tumor extension beyond the subcutaneous fat. Debulk analysis revealed moderate to poorly differentiated cSCC, with the least-differentiated cells with single-cell extension at the deep margin in the galea (Figure 1D). Given T2b staging, baseline imaging and radiation therapy were recommended.
Tumor differentiation is a factor included in the Brigham and Women’s Hospital staging system, and intratumor variability can be clinically relevant for tumor staging.1 Specifically, cSCCs may exhibit intratumor heterogeneity in which predominantly well-differentiated tumors contain focal areas of poorer differentiation.2 This intratumor heterogeneity complicates estimation of tumor risk, as a well-differentiated tumor on biopsy may exhibit poor differentiation at a deeper margin. Our cases highlight that the cells at the deeper margin indeed can show poorer differentiation or other higher-risk tumor features. Thus, the most clinically relevant cells for tumor staging and prognostication may not be visible on initial biopsy, underscoring the utility of close examination of the deep layer of the debulk specimen and Mohs layer for comprehensive staging.
Genetic studies have attempted to identify gene expression patterns in cSCCs that predispose to invasion.3 Three of the top 6 genes in this “invasion signature gene set” were matrix metalloproteases; additionally, IL-24 messenger RNA was upregulated in both the cSCC invasion front and in situ cSCCs. IL-24 has been shown to upregulate the expression of matrix metalloprotease 7 in vitro, suggesting that it may influence tumor progression.3 Although gene expression was not included in this series, the identification of genetic variability in the most poorly differentiated cells residing in the deep margins is of great interest and may reveal mutations contributing to irregular cell morphology and cSCC invasiveness.
Prior studies have indicated that a proportion of cSCCs are histopathologically upgraded from the initial biopsy during MMS due to evidence of perineural invasion, bony invasion, or lesser differentiation noted during MMS stages or debulk analysis.1,4 However, the majority of Mohs surgeons report immediately discarding debulk specimens without further evaluation.5 Herein, we highlight 4 cSCC cases in which the deep margins of the debulk specimen contained the most dedifferentiated cells. Our findings emphasize the importance of thoroughly examining deep tumor margins for complete staging yet also highlight that identifying cells at these margins may not change patient management when high-risk criteria are already met.
- McIlwee BE, Abidi NY, Ravi M, et al. Utility of debulk specimens during Mohs micrographic surgery for cutaneous squamous cell carcinoma. Dermatol Surg. 2021;47:599-604.
- Ramón y Cajal S, Sesé M, Capdevila C, et al. Clinical implications of intratumor heterogeneity: challenges and opportunities. J Mol Med. 2020;98:161-177.
- Mitsui H, Suárez-Fariñas M, Gulati N, et al. Gene expression profiling of the leading edge of cutaneous squamous cell carcinoma: IL-24-driven MMP-7. J Invest Dermatol. 2014;134:1418-1427.
- Chung E, Hoang S, McEvoy AM, et al. Histopathologic upgrading of cutaneous squamous cell carcinomas during Mohs micrographic surgery: a retrospective cohort study. J Am Acad Dermatol. 2021;85:923-930.
- Alniemi DT, Swanson AM, Lasarev M, et al. Tumor debulking trends for keratinocyte carcinomas among Mohs surgeons. Dermatol Surg. 2021;47:1660-1661.
To the Editor:
Histopathologic analysis of debulk specimens in Mohs micrographic surgery (MMS) may augment identification of high-risk factors in cutaneous squamous cell carcinoma (cSCC), which may warrant tumor upstaging.1 Intratumor location has not been studied when looking at these high-risk factors. Herein, we report 4 cSCCs initially categorized as well differentiated that were reclassified as moderate to poorly differentiated on analysis of debulk specimens obtained via shave removal.
An 80-year-old man (patient 1) presented with a tender 2-cm erythematous plaque with dried hemorrhagic crusting on the frontal scalp. He had a history of nonmelanoma skin cancers. A biopsy revealed a well-differentiated cSCC, which was upgraded from a T2a tumor to T2b during MMS due to galea involvement. Debulk analysis revealed moderate to poorly differentiated cSCC, with the least-differentiated cells at the deep margin (Figure 1A). Given T2b staging, baseline imaging and radiation therapy were recommended.

A 75-year-old man (patient 2) presented with a 2-cm erythematous plaque on the left vertex scalp with hemorrhagic crusting, yellow scale, and purulent drainage. He had a history of cSCCs. A biopsy revealed well-differentiated invasive cSCC, which was upgraded from a T2a tumor to T2b during MMS due to tumor extension beyond the subcutaneous fat. Examination of the second Mohs stage revealed moderately differentiated cSCC, with the least-differentiated cells at the deep margin, infiltration beyond the subcutaneous fat, and perineural invasion (Figure 1B). Given T2b staging, baseline imaging and radiation therapy were recommended.
An 86-year-old woman (patient 3) presented with a tender 2.4-cm plum-colored nodule on the right lower leg. She had a history of basal cell carcinoma. A biopsy revealed a well-differentiated invasive cSCC staged at T2a. Debulk analysis revealed moderately differentiated cSCC, with the least-differentiated cells at the deep margin, though the staging remained the same (Figure 1C).
An 82-year-old man (patient 4) presented with a 2.7-cm ulcerated nodule with adjacent scaling on the vertex scalp. He had no history of skin cancer. A biopsy revealed a well-differentiated cSCC (Figure 2) that was upgraded from a T2a tumor to T2b during MMS due to tumor extension beyond the subcutaneous fat. Debulk analysis revealed moderate to poorly differentiated cSCC, with the least-differentiated cells with single-cell extension at the deep margin in the galea (Figure 1D). Given T2b staging, baseline imaging and radiation therapy were recommended.

Tumor differentiation is a factor included in the Brigham and Women’s Hospital staging system, and intratumor variability can be clinically relevant for tumor staging.1 Specifically, cSCCs may exhibit intratumor heterogeneity in which predominantly well-differentiated tumors contain focal areas of poorer differentiation.2 This intratumor heterogeneity complicates estimation of tumor risk, as a well-differentiated tumor on biopsy may exhibit poor differentiation at a deeper margin. Our cases highlight that the cells at the deeper margin indeed can show poorer differentiation or other higher-risk tumor features. Thus, the most clinically relevant cells for tumor staging and prognostication may not be visible on initial biopsy, underscoring the utility of close examination of the deep layer of the debulk specimen and Mohs layer for comprehensive staging.
Genetic studies have attempted to identify gene expression patterns in cSCCs that predispose to invasion.3 Three of the top 6 genes in this “invasion signature gene set” were matrix metalloproteases; additionally, IL-24 messenger RNA was upregulated in both the cSCC invasion front and in situ cSCCs. IL-24 has been shown to upregulate the expression of matrix metalloprotease 7 in vitro, suggesting that it may influence tumor progression.3 Although gene expression was not included in this series, the identification of genetic variability in the most poorly differentiated cells residing in the deep margins is of great interest and may reveal mutations contributing to irregular cell morphology and cSCC invasiveness.
Prior studies have indicated that a proportion of cSCCs are histopathologically upgraded from the initial biopsy during MMS due to evidence of perineural invasion, bony invasion, or lesser differentiation noted during MMS stages or debulk analysis.1,4 However, the majority of Mohs surgeons report immediately discarding debulk specimens without further evaluation.5 Herein, we highlight 4 cSCC cases in which the deep margins of the debulk specimen contained the most dedifferentiated cells. Our findings emphasize the importance of thoroughly examining deep tumor margins for complete staging yet also highlight that identifying cells at these margins may not change patient management when high-risk criteria are already met.
To the Editor:
Histopathologic analysis of debulk specimens in Mohs micrographic surgery (MMS) may augment identification of high-risk factors in cutaneous squamous cell carcinoma (cSCC), which may warrant tumor upstaging.1 Intratumor location has not been studied when looking at these high-risk factors. Herein, we report 4 cSCCs initially categorized as well differentiated that were reclassified as moderate to poorly differentiated on analysis of debulk specimens obtained via shave removal.
An 80-year-old man (patient 1) presented with a tender 2-cm erythematous plaque with dried hemorrhagic crusting on the frontal scalp. He had a history of nonmelanoma skin cancers. A biopsy revealed a well-differentiated cSCC, which was upgraded from a T2a tumor to T2b during MMS due to galea involvement. Debulk analysis revealed moderate to poorly differentiated cSCC, with the least-differentiated cells at the deep margin (Figure 1A). Given T2b staging, baseline imaging and radiation therapy were recommended.

A 75-year-old man (patient 2) presented with a 2-cm erythematous plaque on the left vertex scalp with hemorrhagic crusting, yellow scale, and purulent drainage. He had a history of cSCCs. A biopsy revealed well-differentiated invasive cSCC, which was upgraded from a T2a tumor to T2b during MMS due to tumor extension beyond the subcutaneous fat. Examination of the second Mohs stage revealed moderately differentiated cSCC, with the least-differentiated cells at the deep margin, infiltration beyond the subcutaneous fat, and perineural invasion (Figure 1B). Given T2b staging, baseline imaging and radiation therapy were recommended.
An 86-year-old woman (patient 3) presented with a tender 2.4-cm plum-colored nodule on the right lower leg. She had a history of basal cell carcinoma. A biopsy revealed a well-differentiated invasive cSCC staged at T2a. Debulk analysis revealed moderately differentiated cSCC, with the least-differentiated cells at the deep margin, though the staging remained the same (Figure 1C).
An 82-year-old man (patient 4) presented with a 2.7-cm ulcerated nodule with adjacent scaling on the vertex scalp. He had no history of skin cancer. A biopsy revealed a well-differentiated cSCC (Figure 2) that was upgraded from a T2a tumor to T2b during MMS due to tumor extension beyond the subcutaneous fat. Debulk analysis revealed moderate to poorly differentiated cSCC, with the least-differentiated cells with single-cell extension at the deep margin in the galea (Figure 1D). Given T2b staging, baseline imaging and radiation therapy were recommended.

Tumor differentiation is a factor included in the Brigham and Women’s Hospital staging system, and intratumor variability can be clinically relevant for tumor staging.1 Specifically, cSCCs may exhibit intratumor heterogeneity in which predominantly well-differentiated tumors contain focal areas of poorer differentiation.2 This intratumor heterogeneity complicates estimation of tumor risk, as a well-differentiated tumor on biopsy may exhibit poor differentiation at a deeper margin. Our cases highlight that the cells at the deeper margin indeed can show poorer differentiation or other higher-risk tumor features. Thus, the most clinically relevant cells for tumor staging and prognostication may not be visible on initial biopsy, underscoring the utility of close examination of the deep layer of the debulk specimen and Mohs layer for comprehensive staging.
Genetic studies have attempted to identify gene expression patterns in cSCCs that predispose to invasion.3 Three of the top 6 genes in this “invasion signature gene set” were matrix metalloproteases; additionally, IL-24 messenger RNA was upregulated in both the cSCC invasion front and in situ cSCCs. IL-24 has been shown to upregulate the expression of matrix metalloprotease 7 in vitro, suggesting that it may influence tumor progression.3 Although gene expression was not included in this series, the identification of genetic variability in the most poorly differentiated cells residing in the deep margins is of great interest and may reveal mutations contributing to irregular cell morphology and cSCC invasiveness.
Prior studies have indicated that a proportion of cSCCs are histopathologically upgraded from the initial biopsy during MMS due to evidence of perineural invasion, bony invasion, or lesser differentiation noted during MMS stages or debulk analysis.1,4 However, the majority of Mohs surgeons report immediately discarding debulk specimens without further evaluation.5 Herein, we highlight 4 cSCC cases in which the deep margins of the debulk specimen contained the most dedifferentiated cells. Our findings emphasize the importance of thoroughly examining deep tumor margins for complete staging yet also highlight that identifying cells at these margins may not change patient management when high-risk criteria are already met.
- McIlwee BE, Abidi NY, Ravi M, et al. Utility of debulk specimens during Mohs micrographic surgery for cutaneous squamous cell carcinoma. Dermatol Surg. 2021;47:599-604.
- Ramón y Cajal S, Sesé M, Capdevila C, et al. Clinical implications of intratumor heterogeneity: challenges and opportunities. J Mol Med. 2020;98:161-177.
- Mitsui H, Suárez-Fariñas M, Gulati N, et al. Gene expression profiling of the leading edge of cutaneous squamous cell carcinoma: IL-24-driven MMP-7. J Invest Dermatol. 2014;134:1418-1427.
- Chung E, Hoang S, McEvoy AM, et al. Histopathologic upgrading of cutaneous squamous cell carcinomas during Mohs micrographic surgery: a retrospective cohort study. J Am Acad Dermatol. 2021;85:923-930.
- Alniemi DT, Swanson AM, Lasarev M, et al. Tumor debulking trends for keratinocyte carcinomas among Mohs surgeons. Dermatol Surg. 2021;47:1660-1661.
- McIlwee BE, Abidi NY, Ravi M, et al. Utility of debulk specimens during Mohs micrographic surgery for cutaneous squamous cell carcinoma. Dermatol Surg. 2021;47:599-604.
- Ramón y Cajal S, Sesé M, Capdevila C, et al. Clinical implications of intratumor heterogeneity: challenges and opportunities. J Mol Med. 2020;98:161-177.
- Mitsui H, Suárez-Fariñas M, Gulati N, et al. Gene expression profiling of the leading edge of cutaneous squamous cell carcinoma: IL-24-driven MMP-7. J Invest Dermatol. 2014;134:1418-1427.
- Chung E, Hoang S, McEvoy AM, et al. Histopathologic upgrading of cutaneous squamous cell carcinomas during Mohs micrographic surgery: a retrospective cohort study. J Am Acad Dermatol. 2021;85:923-930.
- Alniemi DT, Swanson AM, Lasarev M, et al. Tumor debulking trends for keratinocyte carcinomas among Mohs surgeons. Dermatol Surg. 2021;47:1660-1661.
Practice Points
- A proportion of cutaneous squamous cell carcinomas are upgraded from the initial biopsy during Mohs micrographic surgery due to evidence of perineural invasion, bony invasion, or lesser differentiation noted on Mohs stages or debulk analysis.
- Thorough inspection of the deep tumor margins may be required for accurate tumor staging and evaluation of metastatic risk. Cells at the deep margin of the tumor may demonstrate poorer differentiation and/or other higher-risk tumor features than those closer to the surface.
- Tumor staging may be incomplete until the deep margins are assessed to find the most dysplastic and likely clinically relevant cells, which may be missed without evaluation of the debulked tumor.
Dupilumab for the Treatment of Lichen Planus
To the Editor:
Lichen planus (LP) is an inflammatory mucocutaneous disorder that primarily affects adults aged 30 to 60 years.1 It can present across various regions such as the skin, scalp, oral cavity, genitalia, nails, and hair. It classically presents with pruritic, purple, polygonal papules or plaques. The proposed pathogenesis of this condition involves autoimmune destruction of epidermal basal keratinocytes.2 Management involves a stepwise approach, beginning with topical therapies such as corticosteroids and phototherapy and proceeding to systemic therapy including oral corticosteroids and retinoids. Additional medications with reported positive results include immunomodulators such as cyclosporine, tacrolimus, and mycophenolate mofetil.2-4 Dupilumab is a biologic immunomodulator and antagonist to the IL-4Rα on helper T cells (TH1). Although indicated for the treatment of moderate to severe atopic dermatitis, this medication’s immunomodulatory properties have been shown to aid various inflammatory cutaneous conditions, including prurigo nodularis.5-9 We present a case of dupilumab therapy for treatment-refractory LP.
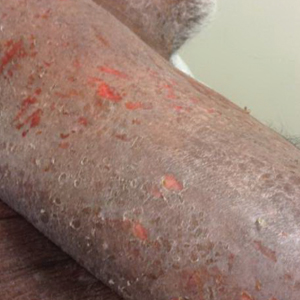
A 52-year-old man presented with a new-onset progressive rash over the prior 6 months. He reported no history of atopic dermatitis. The patient described the rash as “severely pruritic” with a numeric rating scale itch intensity of 9/10 (0 being no itch; 10 being the worst itch imaginable). Physical examination revealed purple polygonal scaly papules on the arms, hands, legs, feet, chest, and back (Figure 1).
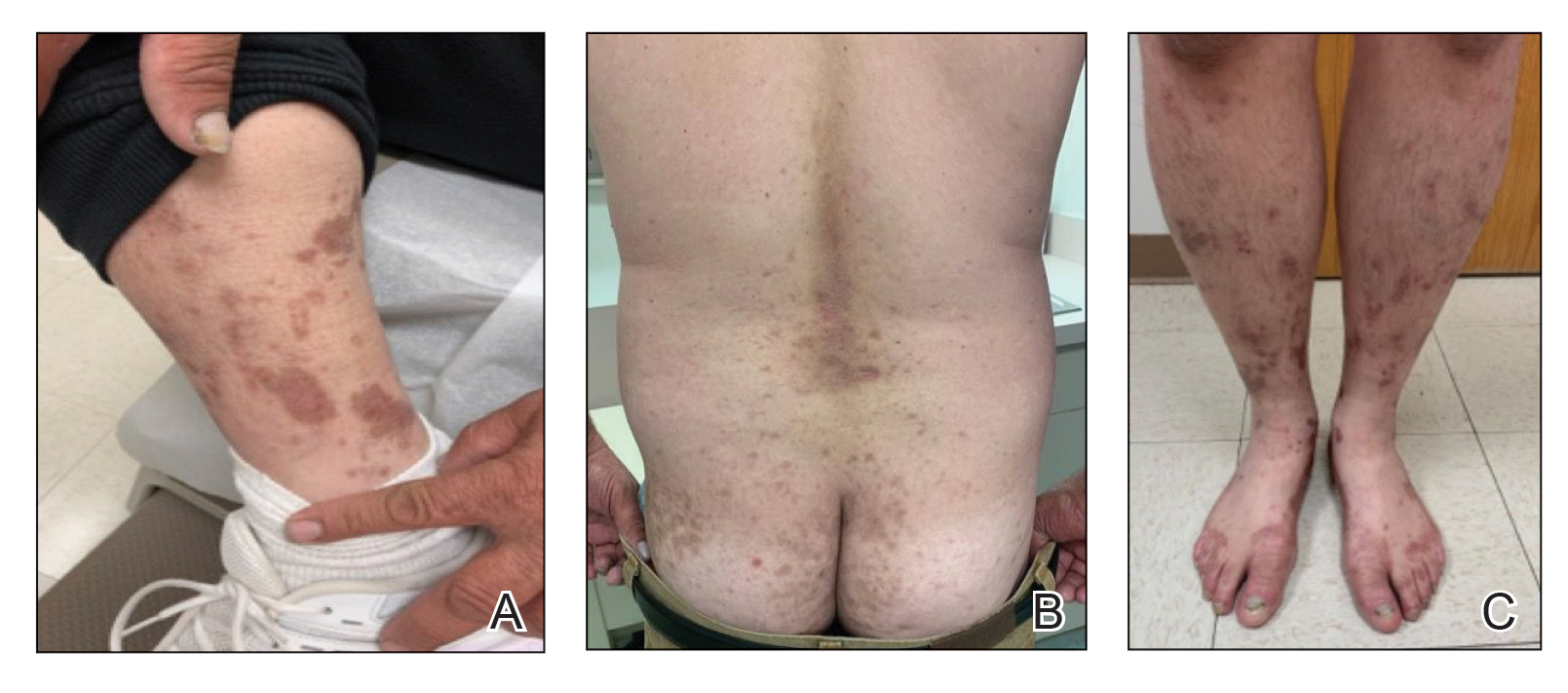
Three biopsies were taken, all indicative of lichenoid dermatitis consistent with LP. Rapid plasma reagin as well as HIV and hepatitis C virus serology tests were negative. Halobetasol ointment, tacrolimus ointment, and oral prednisone (28-day taper starting at 40 mg) all failed. Acitretin subsequently was initiated and failed to provide any benefit. The patient was unable to come to clinic 3 times a week for phototherapy due to his work schedule.
Due to the chronic, severe, and recalcitrant nature of his condition, as well as the lack of US Food and Drug Administration–approved treatments, the patient agreed to begin off-label treatment with dupilumab. Upon documentation, the patient’s primary diagnosis was listed as LP, clearly stating all commonly accepted treatments were attempted, except off-label therapy, and failed, and the plan was to treat him with dupilumab as if he had a severe form of atopic dermatitis. Dupilumab was approved with this documentation with a minimal co-pay, as the patient was on Medicaid. At 3-month follow-up (after 4 administrations of the medication), the patient showed remarkable improvement in appearance, and his numeric rating scale itch intensity score improved to 1/10.
Lichen planus is an immune-mediated, inflammatory condition that can affect the skin, hair, nails, and oral cavity. Although its etiology is not fully understood, research supports a primarily TH1 immunologic reaction.10 These T cells promote cytotoxic CD8 T-cell differentiation and migration, leading to subsequent destruction of epidermal basal keratinocytes. An important cytokine in this pathway—tumor necrosis factor α—stimulates a series of proinflammatory factors, including IL-1α, IL-8, and IL-6. IL-6 is of particular interest, as its elevation has been identified in the serum of patients with LP, with levels correlating to disease severity.11 This increase is thought to be multifactorial and a reliable predictor of disease activity.12,13 In addition to its proinflammatory role, IL-6 promotes the activity of IL-4, an essential cytokine in TH2 T-cell differentiation.
The TH2 pathway, enhanced by IL-6, increases the activity of downstream cytokines IL-4, IL-5, and IL-13. This pathway promotes IgE class switching and eosinophil maturation, pivotal factors in the development of atopic conditions such as allergic rhinitis, asthma, and atopic dermatitis. The role of IL-4 and TH2 cells in the pathogenesis of LP remains poorly understood.14 In prior basic laboratory studies, utilizing tissue sampling, RNA extraction, and real-time polymerase chain reaction assays, Yamauchi et al15 proposed that TH2-related chemokines played a pathogenic role in oral LP. Additional reports propose the pathogenic involvement of TH17, TH0, and TH2 T cells.16 These findings suggest that elevated IL-6 in those with LP may stimulate an increase in IL-4 and subsequent TH2 response. Dupilumab, a monoclonal antibody that targets IL-4Rα found on T cells, inhibits both IL-4 and IL-13 signaling, decreasing subsequent effector cell function.17,18 Several case reports have described dupilumab successfully treating various additional dermatoses, including prurigo nodularis, chronic pruritus, and bullous pemphigoid.5-9 Our case demonstrates an example of LP responsive to dupilumab. Our findings suggest that dupilumab interacts with the pathogenic cascade of LP, potentially implicating the role of TH2 in the pathophysiology of LP.
Treatment-refractory LP remains difficult to manage for both the patient and provider. Treatment regimens remain limited to small uncontrolled studies and case reports. Although primarily considered a TH1-mediated disease, the interplay of various alternative signaling pathways has been suggested. Our case of dupilumab-responsive LP suggests an underlying pathologic role of TH2-mediated activity. Dupilumab shows promise as an effective therapy for refractory LP, as evidenced by our patient’s remarkable response. Larger studies are warranted regarding the role of TH2-mediated inflammation and the use of dupilumab in LP.
- Cleach LL, Chosidow O. Clinical practice. lichen planus. N Engl J Med. 2012;266:723-732.
- Lehman, JS, Tollefson MM, Gibson LE. Lichen planus. Int J Dermatol. 2009;48:682-694.
- Frieling U, Bonsmann G, Schwarz T, et al. Treatment of severe lichen planus with mycophenolate mofetil. J Am Acad Dermatol. 2003;49:1063-1066.
- Cribier B, Frances C, Chosidow O. Treatment of lichen planus. an evidence-based medicine analysis of efficacy. Arch Dermatol. 1998;134:1521-1530.
- Calugareanu A, Jachiet C, Lepelletier C, et al. Dramatic improvement of generalized prurigo nodularis with dupilumab. J Eur Acad Dermatol Venereol. 2019;33:E303-E304.
- Kaye A, Gordon SC, Deverapalli SC, et al. Dupilumab for the treatment of recalcitrant bullous pemphigoid. JAMA Dermatol. 2018;154:1225-1226.
- Mollanazar NK, Qiu CC, Aldrich JL, et al. Use of dupilumab in HIV-positive patients: report of four cases. Br J Dermatol. 2019;181:1311-1312.
- Zhai LL, Savage KT, Qiu CC, et al. Chronic pruritus responding to dupilumab—a case series. Medicines (Basel). 2019;6:72.
- Mollanazar NK, Elgash M, Weaver L, et al. Reduced itch associated with dupilumab treatment in 4 patients with prurigo nodularis. JAMA Dermatol. 2019;155:121-122.
- Lodi G, Scully C, Carrozzo M, et al. Current controversies in oral lichen planus: report of an international consensus meeting. part 1. viral infections and etiopathogenesis. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2005;100:40-51.
- Yin M, Li G, Song H, et al. Identifying the association between interleukin-6 and lichen planus: a meta-analysis. Biomed Rep. 2017;6:571-575.
- Sun A, Chia JS, Chang YF, et al. Serum interleukin-6 level is a useful marker in evaluating therapeutic effects of levamisole and Chinese medicinal herbs on patients with oral lichen planus. J Oral Pathol Med. 2002;31:196-203.
- Rhodus NL, Cheng B, Bowles W, et al. Proinflammatory cytokine levels in saliva before and after treatment of (erosive) oral lichen planus with dexamethasone. Oral Dis. 2006;12:112-116.
- Carrozzo M. Understanding the pathobiology of oral lichen planus. Curr Oral Health Rep. 2014;1:173-179.
- Yamauchi M, Moriyama M, Hayashida JN, et al. Myeloid dendritic cells stimulated by thymic stromal lymphopoietin promote Th2 immune responses and the pathogenesis of oral lichen planus. Plos One. 2017:12:e0173017.
- Piccinni M-P, Lombardell L, Logidice F, et al. Potential pathogenetic role of Th17, Th0, and Th2 cells in erosive and reticular oral lichen planus. Oral Dis. 2013:20:212-218.
- Kidd P. Th1/Th2 balance: the hypothesis, its limitations, and implications for health and disease. Altern Med Rev. 2003;8:223-246.
- Noda S, Kruefer JG, Guttum-Yassky E. The translational revolution and use of biologics in patients with inflammatory skin diseases. J Allergy Clin Immunol. 2015;135:324-336.
To the Editor:
Lichen planus (LP) is an inflammatory mucocutaneous disorder that primarily affects adults aged 30 to 60 years.1 It can present across various regions such as the skin, scalp, oral cavity, genitalia, nails, and hair. It classically presents with pruritic, purple, polygonal papules or plaques. The proposed pathogenesis of this condition involves autoimmune destruction of epidermal basal keratinocytes.2 Management involves a stepwise approach, beginning with topical therapies such as corticosteroids and phototherapy and proceeding to systemic therapy including oral corticosteroids and retinoids. Additional medications with reported positive results include immunomodulators such as cyclosporine, tacrolimus, and mycophenolate mofetil.2-4 Dupilumab is a biologic immunomodulator and antagonist to the IL-4Rα on helper T cells (TH1). Although indicated for the treatment of moderate to severe atopic dermatitis, this medication’s immunomodulatory properties have been shown to aid various inflammatory cutaneous conditions, including prurigo nodularis.5-9 We present a case of dupilumab therapy for treatment-refractory LP.
A 52-year-old man presented with a new-onset progressive rash over the prior 6 months. He reported no history of atopic dermatitis. The patient described the rash as “severely pruritic” with a numeric rating scale itch intensity of 9/10 (0 being no itch; 10 being the worst itch imaginable). Physical examination revealed purple polygonal scaly papules on the arms, hands, legs, feet, chest, and back (Figure 1).


Three biopsies were taken, all indicative of lichenoid dermatitis consistent with LP. Rapid plasma reagin as well as HIV and hepatitis C virus serology tests were negative. Halobetasol ointment, tacrolimus ointment, and oral prednisone (28-day taper starting at 40 mg) all failed. Acitretin subsequently was initiated and failed to provide any benefit. The patient was unable to come to clinic 3 times a week for phototherapy due to his work schedule.
Due to the chronic, severe, and recalcitrant nature of his condition, as well as the lack of US Food and Drug Administration–approved treatments, the patient agreed to begin off-label treatment with dupilumab. Upon documentation, the patient’s primary diagnosis was listed as LP, clearly stating all commonly accepted treatments were attempted, except off-label therapy, and failed, and the plan was to treat him with dupilumab as if he had a severe form of atopic dermatitis. Dupilumab was approved with this documentation with a minimal co-pay, as the patient was on Medicaid. At 3-month follow-up (after 4 administrations of the medication), the patient showed remarkable improvement in appearance, and his numeric rating scale itch intensity score improved to 1/10.
Lichen planus is an immune-mediated, inflammatory condition that can affect the skin, hair, nails, and oral cavity. Although its etiology is not fully understood, research supports a primarily TH1 immunologic reaction.10 These T cells promote cytotoxic CD8 T-cell differentiation and migration, leading to subsequent destruction of epidermal basal keratinocytes. An important cytokine in this pathway—tumor necrosis factor α—stimulates a series of proinflammatory factors, including IL-1α, IL-8, and IL-6. IL-6 is of particular interest, as its elevation has been identified in the serum of patients with LP, with levels correlating to disease severity.11 This increase is thought to be multifactorial and a reliable predictor of disease activity.12,13 In addition to its proinflammatory role, IL-6 promotes the activity of IL-4, an essential cytokine in TH2 T-cell differentiation.
The TH2 pathway, enhanced by IL-6, increases the activity of downstream cytokines IL-4, IL-5, and IL-13. This pathway promotes IgE class switching and eosinophil maturation, pivotal factors in the development of atopic conditions such as allergic rhinitis, asthma, and atopic dermatitis. The role of IL-4 and TH2 cells in the pathogenesis of LP remains poorly understood.14 In prior basic laboratory studies, utilizing tissue sampling, RNA extraction, and real-time polymerase chain reaction assays, Yamauchi et al15 proposed that TH2-related chemokines played a pathogenic role in oral LP. Additional reports propose the pathogenic involvement of TH17, TH0, and TH2 T cells.16 These findings suggest that elevated IL-6 in those with LP may stimulate an increase in IL-4 and subsequent TH2 response. Dupilumab, a monoclonal antibody that targets IL-4Rα found on T cells, inhibits both IL-4 and IL-13 signaling, decreasing subsequent effector cell function.17,18 Several case reports have described dupilumab successfully treating various additional dermatoses, including prurigo nodularis, chronic pruritus, and bullous pemphigoid.5-9 Our case demonstrates an example of LP responsive to dupilumab. Our findings suggest that dupilumab interacts with the pathogenic cascade of LP, potentially implicating the role of TH2 in the pathophysiology of LP.
Treatment-refractory LP remains difficult to manage for both the patient and provider. Treatment regimens remain limited to small uncontrolled studies and case reports. Although primarily considered a TH1-mediated disease, the interplay of various alternative signaling pathways has been suggested. Our case of dupilumab-responsive LP suggests an underlying pathologic role of TH2-mediated activity. Dupilumab shows promise as an effective therapy for refractory LP, as evidenced by our patient’s remarkable response. Larger studies are warranted regarding the role of TH2-mediated inflammation and the use of dupilumab in LP.
To the Editor:
Lichen planus (LP) is an inflammatory mucocutaneous disorder that primarily affects adults aged 30 to 60 years.1 It can present across various regions such as the skin, scalp, oral cavity, genitalia, nails, and hair. It classically presents with pruritic, purple, polygonal papules or plaques. The proposed pathogenesis of this condition involves autoimmune destruction of epidermal basal keratinocytes.2 Management involves a stepwise approach, beginning with topical therapies such as corticosteroids and phototherapy and proceeding to systemic therapy including oral corticosteroids and retinoids. Additional medications with reported positive results include immunomodulators such as cyclosporine, tacrolimus, and mycophenolate mofetil.2-4 Dupilumab is a biologic immunomodulator and antagonist to the IL-4Rα on helper T cells (TH1). Although indicated for the treatment of moderate to severe atopic dermatitis, this medication’s immunomodulatory properties have been shown to aid various inflammatory cutaneous conditions, including prurigo nodularis.5-9 We present a case of dupilumab therapy for treatment-refractory LP.
A 52-year-old man presented with a new-onset progressive rash over the prior 6 months. He reported no history of atopic dermatitis. The patient described the rash as “severely pruritic” with a numeric rating scale itch intensity of 9/10 (0 being no itch; 10 being the worst itch imaginable). Physical examination revealed purple polygonal scaly papules on the arms, hands, legs, feet, chest, and back (Figure 1).


Three biopsies were taken, all indicative of lichenoid dermatitis consistent with LP. Rapid plasma reagin as well as HIV and hepatitis C virus serology tests were negative. Halobetasol ointment, tacrolimus ointment, and oral prednisone (28-day taper starting at 40 mg) all failed. Acitretin subsequently was initiated and failed to provide any benefit. The patient was unable to come to clinic 3 times a week for phototherapy due to his work schedule.
Due to the chronic, severe, and recalcitrant nature of his condition, as well as the lack of US Food and Drug Administration–approved treatments, the patient agreed to begin off-label treatment with dupilumab. Upon documentation, the patient’s primary diagnosis was listed as LP, clearly stating all commonly accepted treatments were attempted, except off-label therapy, and failed, and the plan was to treat him with dupilumab as if he had a severe form of atopic dermatitis. Dupilumab was approved with this documentation with a minimal co-pay, as the patient was on Medicaid. At 3-month follow-up (after 4 administrations of the medication), the patient showed remarkable improvement in appearance, and his numeric rating scale itch intensity score improved to 1/10.
Lichen planus is an immune-mediated, inflammatory condition that can affect the skin, hair, nails, and oral cavity. Although its etiology is not fully understood, research supports a primarily TH1 immunologic reaction.10 These T cells promote cytotoxic CD8 T-cell differentiation and migration, leading to subsequent destruction of epidermal basal keratinocytes. An important cytokine in this pathway—tumor necrosis factor α—stimulates a series of proinflammatory factors, including IL-1α, IL-8, and IL-6. IL-6 is of particular interest, as its elevation has been identified in the serum of patients with LP, with levels correlating to disease severity.11 This increase is thought to be multifactorial and a reliable predictor of disease activity.12,13 In addition to its proinflammatory role, IL-6 promotes the activity of IL-4, an essential cytokine in TH2 T-cell differentiation.
The TH2 pathway, enhanced by IL-6, increases the activity of downstream cytokines IL-4, IL-5, and IL-13. This pathway promotes IgE class switching and eosinophil maturation, pivotal factors in the development of atopic conditions such as allergic rhinitis, asthma, and atopic dermatitis. The role of IL-4 and TH2 cells in the pathogenesis of LP remains poorly understood.14 In prior basic laboratory studies, utilizing tissue sampling, RNA extraction, and real-time polymerase chain reaction assays, Yamauchi et al15 proposed that TH2-related chemokines played a pathogenic role in oral LP. Additional reports propose the pathogenic involvement of TH17, TH0, and TH2 T cells.16 These findings suggest that elevated IL-6 in those with LP may stimulate an increase in IL-4 and subsequent TH2 response. Dupilumab, a monoclonal antibody that targets IL-4Rα found on T cells, inhibits both IL-4 and IL-13 signaling, decreasing subsequent effector cell function.17,18 Several case reports have described dupilumab successfully treating various additional dermatoses, including prurigo nodularis, chronic pruritus, and bullous pemphigoid.5-9 Our case demonstrates an example of LP responsive to dupilumab. Our findings suggest that dupilumab interacts with the pathogenic cascade of LP, potentially implicating the role of TH2 in the pathophysiology of LP.
Treatment-refractory LP remains difficult to manage for both the patient and provider. Treatment regimens remain limited to small uncontrolled studies and case reports. Although primarily considered a TH1-mediated disease, the interplay of various alternative signaling pathways has been suggested. Our case of dupilumab-responsive LP suggests an underlying pathologic role of TH2-mediated activity. Dupilumab shows promise as an effective therapy for refractory LP, as evidenced by our patient’s remarkable response. Larger studies are warranted regarding the role of TH2-mediated inflammation and the use of dupilumab in LP.
- Cleach LL, Chosidow O. Clinical practice. lichen planus. N Engl J Med. 2012;266:723-732.
- Lehman, JS, Tollefson MM, Gibson LE. Lichen planus. Int J Dermatol. 2009;48:682-694.
- Frieling U, Bonsmann G, Schwarz T, et al. Treatment of severe lichen planus with mycophenolate mofetil. J Am Acad Dermatol. 2003;49:1063-1066.
- Cribier B, Frances C, Chosidow O. Treatment of lichen planus. an evidence-based medicine analysis of efficacy. Arch Dermatol. 1998;134:1521-1530.
- Calugareanu A, Jachiet C, Lepelletier C, et al. Dramatic improvement of generalized prurigo nodularis with dupilumab. J Eur Acad Dermatol Venereol. 2019;33:E303-E304.
- Kaye A, Gordon SC, Deverapalli SC, et al. Dupilumab for the treatment of recalcitrant bullous pemphigoid. JAMA Dermatol. 2018;154:1225-1226.
- Mollanazar NK, Qiu CC, Aldrich JL, et al. Use of dupilumab in HIV-positive patients: report of four cases. Br J Dermatol. 2019;181:1311-1312.
- Zhai LL, Savage KT, Qiu CC, et al. Chronic pruritus responding to dupilumab—a case series. Medicines (Basel). 2019;6:72.
- Mollanazar NK, Elgash M, Weaver L, et al. Reduced itch associated with dupilumab treatment in 4 patients with prurigo nodularis. JAMA Dermatol. 2019;155:121-122.
- Lodi G, Scully C, Carrozzo M, et al. Current controversies in oral lichen planus: report of an international consensus meeting. part 1. viral infections and etiopathogenesis. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2005;100:40-51.
- Yin M, Li G, Song H, et al. Identifying the association between interleukin-6 and lichen planus: a meta-analysis. Biomed Rep. 2017;6:571-575.
- Sun A, Chia JS, Chang YF, et al. Serum interleukin-6 level is a useful marker in evaluating therapeutic effects of levamisole and Chinese medicinal herbs on patients with oral lichen planus. J Oral Pathol Med. 2002;31:196-203.
- Rhodus NL, Cheng B, Bowles W, et al. Proinflammatory cytokine levels in saliva before and after treatment of (erosive) oral lichen planus with dexamethasone. Oral Dis. 2006;12:112-116.
- Carrozzo M. Understanding the pathobiology of oral lichen planus. Curr Oral Health Rep. 2014;1:173-179.
- Yamauchi M, Moriyama M, Hayashida JN, et al. Myeloid dendritic cells stimulated by thymic stromal lymphopoietin promote Th2 immune responses and the pathogenesis of oral lichen planus. Plos One. 2017:12:e0173017.
- Piccinni M-P, Lombardell L, Logidice F, et al. Potential pathogenetic role of Th17, Th0, and Th2 cells in erosive and reticular oral lichen planus. Oral Dis. 2013:20:212-218.
- Kidd P. Th1/Th2 balance: the hypothesis, its limitations, and implications for health and disease. Altern Med Rev. 2003;8:223-246.
- Noda S, Kruefer JG, Guttum-Yassky E. The translational revolution and use of biologics in patients with inflammatory skin diseases. J Allergy Clin Immunol. 2015;135:324-336.
- Cleach LL, Chosidow O. Clinical practice. lichen planus. N Engl J Med. 2012;266:723-732.
- Lehman, JS, Tollefson MM, Gibson LE. Lichen planus. Int J Dermatol. 2009;48:682-694.
- Frieling U, Bonsmann G, Schwarz T, et al. Treatment of severe lichen planus with mycophenolate mofetil. J Am Acad Dermatol. 2003;49:1063-1066.
- Cribier B, Frances C, Chosidow O. Treatment of lichen planus. an evidence-based medicine analysis of efficacy. Arch Dermatol. 1998;134:1521-1530.
- Calugareanu A, Jachiet C, Lepelletier C, et al. Dramatic improvement of generalized prurigo nodularis with dupilumab. J Eur Acad Dermatol Venereol. 2019;33:E303-E304.
- Kaye A, Gordon SC, Deverapalli SC, et al. Dupilumab for the treatment of recalcitrant bullous pemphigoid. JAMA Dermatol. 2018;154:1225-1226.
- Mollanazar NK, Qiu CC, Aldrich JL, et al. Use of dupilumab in HIV-positive patients: report of four cases. Br J Dermatol. 2019;181:1311-1312.
- Zhai LL, Savage KT, Qiu CC, et al. Chronic pruritus responding to dupilumab—a case series. Medicines (Basel). 2019;6:72.
- Mollanazar NK, Elgash M, Weaver L, et al. Reduced itch associated with dupilumab treatment in 4 patients with prurigo nodularis. JAMA Dermatol. 2019;155:121-122.
- Lodi G, Scully C, Carrozzo M, et al. Current controversies in oral lichen planus: report of an international consensus meeting. part 1. viral infections and etiopathogenesis. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2005;100:40-51.
- Yin M, Li G, Song H, et al. Identifying the association between interleukin-6 and lichen planus: a meta-analysis. Biomed Rep. 2017;6:571-575.
- Sun A, Chia JS, Chang YF, et al. Serum interleukin-6 level is a useful marker in evaluating therapeutic effects of levamisole and Chinese medicinal herbs on patients with oral lichen planus. J Oral Pathol Med. 2002;31:196-203.
- Rhodus NL, Cheng B, Bowles W, et al. Proinflammatory cytokine levels in saliva before and after treatment of (erosive) oral lichen planus with dexamethasone. Oral Dis. 2006;12:112-116.
- Carrozzo M. Understanding the pathobiology of oral lichen planus. Curr Oral Health Rep. 2014;1:173-179.
- Yamauchi M, Moriyama M, Hayashida JN, et al. Myeloid dendritic cells stimulated by thymic stromal lymphopoietin promote Th2 immune responses and the pathogenesis of oral lichen planus. Plos One. 2017:12:e0173017.
- Piccinni M-P, Lombardell L, Logidice F, et al. Potential pathogenetic role of Th17, Th0, and Th2 cells in erosive and reticular oral lichen planus. Oral Dis. 2013:20:212-218.
- Kidd P. Th1/Th2 balance: the hypothesis, its limitations, and implications for health and disease. Altern Med Rev. 2003;8:223-246.
- Noda S, Kruefer JG, Guttum-Yassky E. The translational revolution and use of biologics in patients with inflammatory skin diseases. J Allergy Clin Immunol. 2015;135:324-336.
Practice Points
- Lichen planus (LP) is an inflammatory mucocutaneous disorder that can present across various regions of the body with pruritic, purple, polygonal papules or plaques.
- The proposed pathogenesis of LP involves autoimmune destruction of epidermal basal keratinocytes.
- The immunomodulatory properties of dupilumab have been shown to aid various inflammatory cutaneous conditions.
Cutaneous T-cell Lymphoma and Concomitant Atopic Dermatitis Responding to Dupilumab
Patients with cutaneous T-cell lymphoma (CTCL) often are diagnosed with atopic dermatitis (AD) or psoriasis before receiving their CTCL diagnosis. The effects of new biologic therapies for AD such as dupilumab, an IL-4/IL-13 antagonist, on CTCL are unknown. Dupilumab may be beneficial in CTCL given that helper T cell (TH2) cytokines are increased in advanced CTCL.1 We present a patient with definitive CTCL and concomitant AD who was safely treated with dupilumab and experienced improvement in both CTCL and AD.
Case Report
A 68-year-old man presented with increased itching from AD and a new rash on the arms, neck, chest, back, and lower extremities (Figures 1A and 2A). He had a medical history of AD and CTCL diagnosed by biopsy and peripheral blood flow cytometry (stage IVA1 [T4N0M0B2]) that was being treated with comprehensive multimodality therapy consisting of bexarotene 375 mg daily, interferon alfa-2b 3 mIU 3 times weekly, interferon gamma-1b 2 mIU 3 times weekly, total skin electron beam therapy followed by narrowband UVB twice weekly, and extracorporeal photopheresis every 4 weeks, which resulted in a partial clinical response for 6 months. A biopsy performed at the current presentation showed focal spongiosis and features of lichen simplex chronicus with no evidence suggestive of CTCL. Peripheral blood flow cytometry showed stable B1-staged disease burden (CD4/CD8, 2.6:1); CD4+/CD7−, 12% [91/µL]; CD4+/CD26−, 21% [155/µL]). Treatment with potent and superpotent topical steroids was attempted for more than 6 months and was unsuccessful in relieving the symptoms.
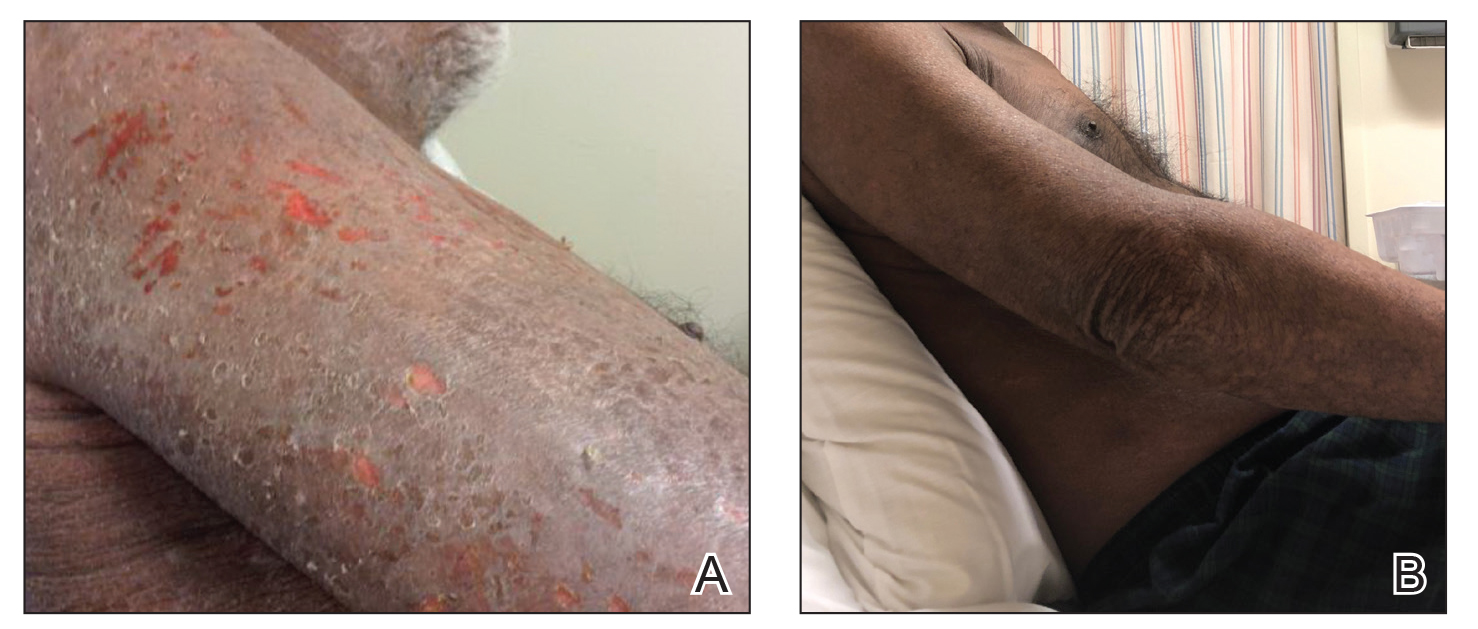
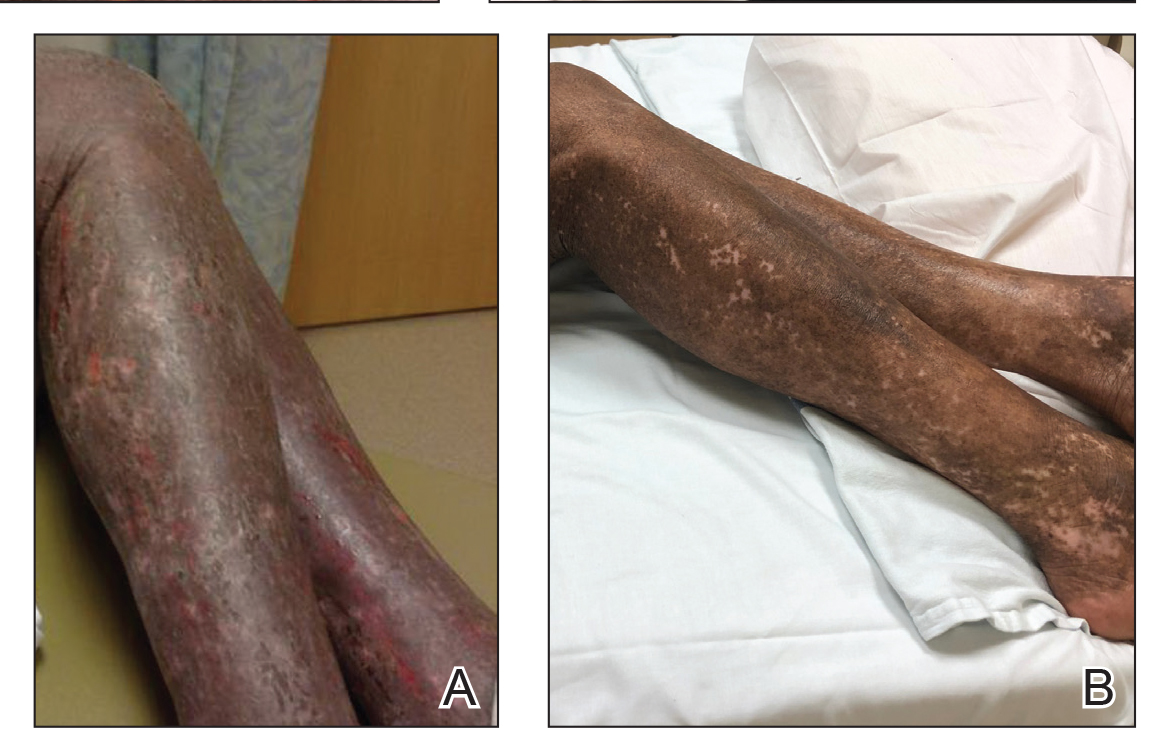
Given the recalcitrant nature of the patient’s rash and itching, dupilumab was added to his CTCL regimen. Prior to initiating dupilumab, the patient reported a numeric rating scale itch intensity of 7 out of 10. After 4 weeks of treatment with dupilumab, the patient reported a numeric rating scale itch intensity of 1. Over a 3-month period, the patient’s rash improved dramatically (Figures 1B and 2B), making it possible to decrease CTCL treatments—bexarotene decreased to 300 mg, interferon alfa-2b to 3 mIU twice weekly, interferon gamma-1b to 2 mIU twice weekly, extracorporeal photopheresis every 5 weeks, and narrowband UVB was discontinued completely. A comparison of the patient’s flow cytometry analysis from before treatment to 3 months after dupilumab showed an overall slight reduction in CTCL B1 blood involvement and normalization of the patient’s absolute eosinophil count and serum lactate dehydrogenase level. The patient tolerated the treatment well without any adverse events and has maintained clinical response for 6 months.
Comment
Cutaneous T-cell lymphomas represent a heterogeneous group of T-cell lymphoproliferative disorders involving the skin.2 The definitive diagnosis of CTCL is challenging, as the clinical and pathologic features often are nonspecific in early disease. Frequently, undiagnosed patients are treated empirically with immunosuppressive agents. Tumor necrosis factor inhibitors and cyclosporine are both associated with progression or worsening of undiagnosed CTCL.3,4 Dupilumab was the first US Food and Drug Administration–approved biologic for the treatment of moderate to severe AD. Cutaneous T-cell lymphoma has immunologic features, such as TH2 skewing, that overlap with AD; however, the effects of dupilumab in CTCL are not yet known.5,6 Our group has seen patients initially thought to have AD who received dupilumab without improvement and were subsequently diagnosed with CTCL, suggesting dupilumab did not affect CTCL tumor cells. Given these findings, there was concern that dupilumab might exacerbate undiagnosed CTCL. Our patient with definitive, severe, refractory CTCL noted marked improvement in both AD and underlying CTCL with the addition of dupilumab. No other treatments were added. The response was so dramatic that we were able to wean the doses and frequencies of several CTCL treatments. Our findings suggest that dupilumab may be beneficial in a certain subset of CTCL patients with a history of AD or known concomitant AD. Prospective studies are needed to fully investigate dupilumab safety and efficacy in CTCL and whether it has any primary effects on tumor burden in addition to benefit for itch and skin symptom relief.
- Guenova E, Watanabe R, Teague JE, et al. TH2 cytokines from malignant cells suppress TH1 responses and enforce a global TH2 bias in leukemic cutaneous T-cell lymphoma. Clin Cancer Res. 2013;19:3755-3763.
- Wilcox RA. Cutaneous T-cell lymphoma: 2016 update on diagnosis, risk-stratification, and management. Am J Hematol. 2016;91:151-165.
- Martinez-Escala ME, Posligua AL, Wickless H, et al. Progression of undiagnosed cutaneous lymphoma after anti-tumor necrosis factor-alpha therapy. J Am Acad Dermatol. 2018;78:1068-1076.
- Pielop JA, Jones D, Duvic M. Transient CD30+ nodal transformation of cutaneous T-cell lymphoma associated with cyclosporine treatment. Int J Dermatol. 2001;40:505-511.
- Saulite I, Hoetzenecker W, Weidinger S, et al. Sézary syndrome and atopic dermatitis: comparison of immunological aspects and targets [published online May 17, 2016]. BioMed Res Int. doi:10.1155/2016/9717530.
- Sigurdsson V, Toonstra J, Bihari IC, et al. Interleukin 4 and interferon-gamma expression of the dermal infiltrate in patients with erythroderma and mycosis fungoides. an immuno-histochemical study. J Cutan Pathol. 2000;27:429-435.
Patients with cutaneous T-cell lymphoma (CTCL) often are diagnosed with atopic dermatitis (AD) or psoriasis before receiving their CTCL diagnosis. The effects of new biologic therapies for AD such as dupilumab, an IL-4/IL-13 antagonist, on CTCL are unknown. Dupilumab may be beneficial in CTCL given that helper T cell (TH2) cytokines are increased in advanced CTCL.1 We present a patient with definitive CTCL and concomitant AD who was safely treated with dupilumab and experienced improvement in both CTCL and AD.
Case Report
A 68-year-old man presented with increased itching from AD and a new rash on the arms, neck, chest, back, and lower extremities (Figures 1A and 2A). He had a medical history of AD and CTCL diagnosed by biopsy and peripheral blood flow cytometry (stage IVA1 [T4N0M0B2]) that was being treated with comprehensive multimodality therapy consisting of bexarotene 375 mg daily, interferon alfa-2b 3 mIU 3 times weekly, interferon gamma-1b 2 mIU 3 times weekly, total skin electron beam therapy followed by narrowband UVB twice weekly, and extracorporeal photopheresis every 4 weeks, which resulted in a partial clinical response for 6 months. A biopsy performed at the current presentation showed focal spongiosis and features of lichen simplex chronicus with no evidence suggestive of CTCL. Peripheral blood flow cytometry showed stable B1-staged disease burden (CD4/CD8, 2.6:1); CD4+/CD7−, 12% [91/µL]; CD4+/CD26−, 21% [155/µL]). Treatment with potent and superpotent topical steroids was attempted for more than 6 months and was unsuccessful in relieving the symptoms.


Given the recalcitrant nature of the patient’s rash and itching, dupilumab was added to his CTCL regimen. Prior to initiating dupilumab, the patient reported a numeric rating scale itch intensity of 7 out of 10. After 4 weeks of treatment with dupilumab, the patient reported a numeric rating scale itch intensity of 1. Over a 3-month period, the patient’s rash improved dramatically (Figures 1B and 2B), making it possible to decrease CTCL treatments—bexarotene decreased to 300 mg, interferon alfa-2b to 3 mIU twice weekly, interferon gamma-1b to 2 mIU twice weekly, extracorporeal photopheresis every 5 weeks, and narrowband UVB was discontinued completely. A comparison of the patient’s flow cytometry analysis from before treatment to 3 months after dupilumab showed an overall slight reduction in CTCL B1 blood involvement and normalization of the patient’s absolute eosinophil count and serum lactate dehydrogenase level. The patient tolerated the treatment well without any adverse events and has maintained clinical response for 6 months.
Comment
Cutaneous T-cell lymphomas represent a heterogeneous group of T-cell lymphoproliferative disorders involving the skin.2 The definitive diagnosis of CTCL is challenging, as the clinical and pathologic features often are nonspecific in early disease. Frequently, undiagnosed patients are treated empirically with immunosuppressive agents. Tumor necrosis factor inhibitors and cyclosporine are both associated with progression or worsening of undiagnosed CTCL.3,4 Dupilumab was the first US Food and Drug Administration–approved biologic for the treatment of moderate to severe AD. Cutaneous T-cell lymphoma has immunologic features, such as TH2 skewing, that overlap with AD; however, the effects of dupilumab in CTCL are not yet known.5,6 Our group has seen patients initially thought to have AD who received dupilumab without improvement and were subsequently diagnosed with CTCL, suggesting dupilumab did not affect CTCL tumor cells. Given these findings, there was concern that dupilumab might exacerbate undiagnosed CTCL. Our patient with definitive, severe, refractory CTCL noted marked improvement in both AD and underlying CTCL with the addition of dupilumab. No other treatments were added. The response was so dramatic that we were able to wean the doses and frequencies of several CTCL treatments. Our findings suggest that dupilumab may be beneficial in a certain subset of CTCL patients with a history of AD or known concomitant AD. Prospective studies are needed to fully investigate dupilumab safety and efficacy in CTCL and whether it has any primary effects on tumor burden in addition to benefit for itch and skin symptom relief.
Patients with cutaneous T-cell lymphoma (CTCL) often are diagnosed with atopic dermatitis (AD) or psoriasis before receiving their CTCL diagnosis. The effects of new biologic therapies for AD such as dupilumab, an IL-4/IL-13 antagonist, on CTCL are unknown. Dupilumab may be beneficial in CTCL given that helper T cell (TH2) cytokines are increased in advanced CTCL.1 We present a patient with definitive CTCL and concomitant AD who was safely treated with dupilumab and experienced improvement in both CTCL and AD.
Case Report
A 68-year-old man presented with increased itching from AD and a new rash on the arms, neck, chest, back, and lower extremities (Figures 1A and 2A). He had a medical history of AD and CTCL diagnosed by biopsy and peripheral blood flow cytometry (stage IVA1 [T4N0M0B2]) that was being treated with comprehensive multimodality therapy consisting of bexarotene 375 mg daily, interferon alfa-2b 3 mIU 3 times weekly, interferon gamma-1b 2 mIU 3 times weekly, total skin electron beam therapy followed by narrowband UVB twice weekly, and extracorporeal photopheresis every 4 weeks, which resulted in a partial clinical response for 6 months. A biopsy performed at the current presentation showed focal spongiosis and features of lichen simplex chronicus with no evidence suggestive of CTCL. Peripheral blood flow cytometry showed stable B1-staged disease burden (CD4/CD8, 2.6:1); CD4+/CD7−, 12% [91/µL]; CD4+/CD26−, 21% [155/µL]). Treatment with potent and superpotent topical steroids was attempted for more than 6 months and was unsuccessful in relieving the symptoms.


Given the recalcitrant nature of the patient’s rash and itching, dupilumab was added to his CTCL regimen. Prior to initiating dupilumab, the patient reported a numeric rating scale itch intensity of 7 out of 10. After 4 weeks of treatment with dupilumab, the patient reported a numeric rating scale itch intensity of 1. Over a 3-month period, the patient’s rash improved dramatically (Figures 1B and 2B), making it possible to decrease CTCL treatments—bexarotene decreased to 300 mg, interferon alfa-2b to 3 mIU twice weekly, interferon gamma-1b to 2 mIU twice weekly, extracorporeal photopheresis every 5 weeks, and narrowband UVB was discontinued completely. A comparison of the patient’s flow cytometry analysis from before treatment to 3 months after dupilumab showed an overall slight reduction in CTCL B1 blood involvement and normalization of the patient’s absolute eosinophil count and serum lactate dehydrogenase level. The patient tolerated the treatment well without any adverse events and has maintained clinical response for 6 months.
Comment
Cutaneous T-cell lymphomas represent a heterogeneous group of T-cell lymphoproliferative disorders involving the skin.2 The definitive diagnosis of CTCL is challenging, as the clinical and pathologic features often are nonspecific in early disease. Frequently, undiagnosed patients are treated empirically with immunosuppressive agents. Tumor necrosis factor inhibitors and cyclosporine are both associated with progression or worsening of undiagnosed CTCL.3,4 Dupilumab was the first US Food and Drug Administration–approved biologic for the treatment of moderate to severe AD. Cutaneous T-cell lymphoma has immunologic features, such as TH2 skewing, that overlap with AD; however, the effects of dupilumab in CTCL are not yet known.5,6 Our group has seen patients initially thought to have AD who received dupilumab without improvement and were subsequently diagnosed with CTCL, suggesting dupilumab did not affect CTCL tumor cells. Given these findings, there was concern that dupilumab might exacerbate undiagnosed CTCL. Our patient with definitive, severe, refractory CTCL noted marked improvement in both AD and underlying CTCL with the addition of dupilumab. No other treatments were added. The response was so dramatic that we were able to wean the doses and frequencies of several CTCL treatments. Our findings suggest that dupilumab may be beneficial in a certain subset of CTCL patients with a history of AD or known concomitant AD. Prospective studies are needed to fully investigate dupilumab safety and efficacy in CTCL and whether it has any primary effects on tumor burden in addition to benefit for itch and skin symptom relief.
- Guenova E, Watanabe R, Teague JE, et al. TH2 cytokines from malignant cells suppress TH1 responses and enforce a global TH2 bias in leukemic cutaneous T-cell lymphoma. Clin Cancer Res. 2013;19:3755-3763.
- Wilcox RA. Cutaneous T-cell lymphoma: 2016 update on diagnosis, risk-stratification, and management. Am J Hematol. 2016;91:151-165.
- Martinez-Escala ME, Posligua AL, Wickless H, et al. Progression of undiagnosed cutaneous lymphoma after anti-tumor necrosis factor-alpha therapy. J Am Acad Dermatol. 2018;78:1068-1076.
- Pielop JA, Jones D, Duvic M. Transient CD30+ nodal transformation of cutaneous T-cell lymphoma associated with cyclosporine treatment. Int J Dermatol. 2001;40:505-511.
- Saulite I, Hoetzenecker W, Weidinger S, et al. Sézary syndrome and atopic dermatitis: comparison of immunological aspects and targets [published online May 17, 2016]. BioMed Res Int. doi:10.1155/2016/9717530.
- Sigurdsson V, Toonstra J, Bihari IC, et al. Interleukin 4 and interferon-gamma expression of the dermal infiltrate in patients with erythroderma and mycosis fungoides. an immuno-histochemical study. J Cutan Pathol. 2000;27:429-435.
- Guenova E, Watanabe R, Teague JE, et al. TH2 cytokines from malignant cells suppress TH1 responses and enforce a global TH2 bias in leukemic cutaneous T-cell lymphoma. Clin Cancer Res. 2013;19:3755-3763.
- Wilcox RA. Cutaneous T-cell lymphoma: 2016 update on diagnosis, risk-stratification, and management. Am J Hematol. 2016;91:151-165.
- Martinez-Escala ME, Posligua AL, Wickless H, et al. Progression of undiagnosed cutaneous lymphoma after anti-tumor necrosis factor-alpha therapy. J Am Acad Dermatol. 2018;78:1068-1076.
- Pielop JA, Jones D, Duvic M. Transient CD30+ nodal transformation of cutaneous T-cell lymphoma associated with cyclosporine treatment. Int J Dermatol. 2001;40:505-511.
- Saulite I, Hoetzenecker W, Weidinger S, et al. Sézary syndrome and atopic dermatitis: comparison of immunological aspects and targets [published online May 17, 2016]. BioMed Res Int. doi:10.1155/2016/9717530.
- Sigurdsson V, Toonstra J, Bihari IC, et al. Interleukin 4 and interferon-gamma expression of the dermal infiltrate in patients with erythroderma and mycosis fungoides. an immuno-histochemical study. J Cutan Pathol. 2000;27:429-435.
Practice Points
- The diagnosis of cutaneous T-cell lymphoma (CTCL), particularly early-stage disease, remains challenging and often requires a combination of serial clinical evaluations as well as laboratory diagnostic examinations.
- Dupilumab and its effect on helper T cell (TH2) skewing may play a role in the future management of CTCL.
Treatment of Psoriasis in Pregnancy
Historically, there have been limited data available on the management of psoriasis in pregnancy. The most comprehensive discussion of treatment guidelines is from 2012.1 In the interim, many biologics have been approved for treating psoriasis, with slow accumulation of pregnancy safety data. The 2019 American Academy of Dermatology–National Psoriasis Foundation guidelines on biologics for psoriasis contain updated information but also highlight the paucity of pregnancy safety data.2 This gap is in part a consequence of the exclusion and disenrollment of pregnant women from clinical trials.3 Additionally, lack of detection through registries contributes; pregnancy capture in registries is low compared to the expected number of pregnancies estimated from US Census data.4 Despite these shortcomings, psoriasis patients who are already pregnant or are considering becoming pregnant frequently are encountered in practice and may need treatment. This article reviews the evidence on commonly used treatments for psoriasis in pregnancy.
Background
For many patients, psoriasis improves during pregnancy5,6 and becomes worse postpartum. In a prospective study, most patients reported improvement in pregnancy corresponding to a significant decrease in
In addition to the maternal disease state, the issue of pregnancy outcomes is paramount. In the inflammatory bowel disease and rheumatology literature, it is established that uncontrolled disease is associated with poorer pregnancy outcomes.8-10 Guidelines vary among societies on the use of biologics in pregnancy generally (eTable 11,2,9,11-24), but some societies recommend systemic agents to achieve disease control during pregnancy.9,25
Assessing the potential interplay between disease severity and outcomes in pregnant women with psoriasis is further complicated by the slowly growing body of literature demonstrating that women with psoriasis have more comorbidities26 and worse pregnancy outcomes.27,28 Pregnant psoriasis patients are more likely to smoke, have depression, and be overweight or obese prior to pregnancy and are less likely to take prenatal vitamins.26 They also have an increased risk for cesarean birth, gestational diabetes, gestational hypertension, and preeclampsia.28 In contrast to these prior studies, a systematic review revealed no risk for adverse outcomes in pregnant women with psoriasis.29
Assessment of Treatments for Psoriasis in Pregnancy
In light of these issues, treatment of psoriasis during pregnancy should be assessed from several vantage points. Of note, the US Food and Drug Administration changed its classification scheme in 2015 to a more narrative format called the Pregnancy and Lactation Labeling Rule.30 Prior classifications, however, provide a reasonable starting point for categorizing the safety of drugs (Table31). Importantly, time of exposure to systemic agents also matters; first-trimester exposure is more likely to affect embryogenesis, whereas second- and third-trimester exposures are more prone to affect other aspects of fetal growth. eTable 2 provides data on the use of oral and topical medications to treat psoriasis in pregnancy.1,8,22,32-45

Topical Agents
Topical steroids are largely understood to be reasonable treatment options, though consideration of potency, formulation, area of application, and use of occlusion is important.1,46 Risk for orofacial cleft has been noted with first-trimester topical steroid exposure, though a 2015 Cochrane review update determined that the relative risk of this association was not significantly elevated.32
The impact of topical calcipotriene and salicylic acid has not been studied in human pregnancies,1 but systemic absorption can occur for both. There is potential for vitamin D toxicity with calcipotriene46; consequently, use during pregnancy is not recommended.1,46 Some authors recommend against topical salicylic acid in pregnancy; others report that limited exposure is permissible.47 In fact, as salicylic acid commonly is found in over-the-counter acne products, many women of childbearing potential likely have quotidian exposure.
Preterm delivery and low birthweight have been reported with oral tacrolimus; however, risk with topical tacrolimus is thought to be low1 because the molecular size likely prohibits notable absorption.47 Evidence for the use of anthralin and coal tar also is scarce. First-trimester coal tar use should be avoided; subsequent use in pregnancy should be restricted given concern for adverse outcomes.1
Phototherapy
Broadband or narrowband UVB therapy is recommended as second-line therapy in pregnancy. No cases of fetal risk or premature delivery associated with UVB therapy were found in our search.1 Phototherapy can exacerbate melasma47 and decrease folate levels48; as such, some authors recommend folate supplementation in females of childbearing age who are being treated with phototherapy.49 Psoralen, used in psoralen plus UVA therapy, is mutagenic and therefore contraindicated in pregnancy.1
Oral Medications
Both methotrexate, which is a teratogen, abortifacient, and mutagen,1 and systemic retinoids, which are teratogens, are contraindicated in pregnancy.1,47 Acitretin labeling recommends avoiding pregnancy for 3 years posttreatment50 because alcohol intake prolongs the medication’s half-life.22
Apremilast use is not documented in pregnant psoriasis patients51; an ongoing registry of the Organization of Tetralogy Information Specialists has not reported publicly to date.52 Animal studies of apremilast have documented dose-related decreased birthweight and fetal loss.22
Safety data for systemic steroids, used infrequently in psoriasis, are not well established. First-trimester prednisone exposure has been associated with prematurity, low birthweight, and congenital abnormalities.38 A separate evaluation of 1047 children exposed to betamethasone in utero failed to demonstrate significant change in birthweight or head circumference. However, repeat antenatal corticosteroid exposure was associated with attention problems at 2 years of age.39
Data regarding cyclosporine use, derived primarily from organ transplant recipients, suggest elevated risk for prematurity and low birthweight.53,54 A meta-analysis demonstrated that organ transplant recipients taking cyclosporine had a nonsignificantly elevated odds ratio for congenital malformations, prematurity, and low birthweight.42 Cyclosporine use for psoriasis in pregnancy is not well described; in a study, rates of prematurity and low birthweight were both 21%.43 Limited data are available for Janus kinase inhibitors, none of which are approved for psoriasis, though clinical trials in psoriasis and psoriatic arthritis are underway (ClinicalTrials.gov identifiers NCT04246372, NCT03104374, NCT03104400).
Biologics and Small-Molecule Inhibitors
Limited data on biologics in pregnancy exist25 (eTable 3). Placental transport of IgG antibodies, including biologics, increases throughout pregnancy, especially in the third trimester.82 Infants of mothers treated with a biologic with potential for placental transfer are therefore considered by some authors to be immunosuppressed during the first months of life.2
Looking globally across biologics used for psoriasis, limited safety data are encouraging. In a review of PSOLAR (Psoriasis Longitudinal Assessment and Registry), 83 pregnancies with biologic exposure resulted in 59 live births (71%); 18 spontaneous abortions (22%); 6 induced abortions (7%); no congenital abnormalities; and 7 reports of neonatal problems, including respiratory issues, ABO blood group mismatch, hospitalization, and opioid withdrawal.83
Use of tumor necrosis factor (TNF) inhibitors in pregnancy has the most data25 and is considered a reasonable treatment option. Historically, there was concern about the risk for VACTERL syndrome (vertebral defects, anal atresia, cardiac defects, tracheoesophageal fistula, renal anomalies, limb abnormalities) with exposure to a TNF inhibitor,25,84-86 but further reports have alleviated these concerns. Active transplacental transport occurs for adalimumab, infliximab, and golimumab,87 but given structural differences, transport of certolizumab and etanercept is substantially less.88,89 In the CRIB study of placental transfer of certolizumab from mother to infant (N=14), pharmacokinetic data demonstrated no quantifiable certolizumab levels in 13 infants and minimal levels in 1 infant at birth.88 There are fewer data available on the use of other biologics in pregnancy, but for those in which active placental transport is relevant, similar concerns (ie, immunosuppression) might arise (eTable 3).
Concern over biologics largely involves risk for newborn immunosuppression. A case report detailed a Crohn disease patient treated with infliximab who gave birth to an infant who died of disseminated bacille Calmette-Guérin infection at 4.5 months after receiving the vaccine at 3 months.90 This case underscores the importance of delaying live vaccination in infants born to mothers who were treated with a biologic during pregnancy. Authors have provided various data on how long to avoid vaccination; some state as long as 1 year.91
In pregnant females with inflammatory bowel disease treated with a biologic, no correlation was observed among maternal, placental, and infant serum biologic levels and neonatal infection. However, an association between preterm birth and the level of the biologic in maternal and placental (but not infant) serum and preterm birth was observed.92
In another report from the same registry, combination therapy with a TNF inhibitor and another immunomodulator led to an increased risk for infection in infants at 12 months of age, compared to infants exposed to monotherapy89 or exposed to neither agent.93 A strategy to circumvent this potential problem is to avoid treatment with actively transported molecules in the third trimester.
Conclusion
Limited data exist to guide providers who are treating pregnant women with psoriasis. Our understanding of treatment of psoriasis in pregnancy is limited as a consequence of regulations surrounding clinical trials and inadequate detection of pregnancies in registries. Further efforts are necessary to better understand the relationship between psoriasis and pregnancy and how to manage pregnant women with psoriasis.
- Bae YS, Van Voorhees AS, Hsu S, et al. Review of treatment options for psoriasis in pregnant or lactating women: from the medical board of the National Psoriasis Foundation. J Am Acad Dermatol. 2012;67:459-477.
- Menter A, Strober BE, Kaplan DH, et al. Joint AAD-NPF guidelines of care for the management and treatment of psoriasis with biologics. J Am Acad Dermatol. 2019;80:1029-1072.
- Flood KS, Porter ML, Kimball AB. Use of biologics in pregnancy: limitations stemming from clinical trials and registry experience. J Eur Acad Dermatol Venereol. 2019;33:E276-E277.
- Horn EJ, Chambers CD, Menter A, et al. Pregnancy outcomes in psoriasis: why do we know so little? J Am Acad Dermatol. 2009;61:E5-E8.
- Raychaudhuri SP, Navare T, Gross J, et al. Clinical course of psoriasis during pregnancy. Int J Dermatol. 2003;42:518-520.
- Boyd AS, Morris LF, Phillips CM, et al. Psoriasis and pregnancy: hormone and immune system interaction. Int J Dermatol. 1996;35:169-172.
- Murase JE, Chan KK, Garite TJ, et al. Hormonal effect on psoriasis in pregnancy and post partum. Arch Dermatol. 2005;14:601-606.
- Götestam Skorpen C, Hoeltzenbein M, Tincani A, et al. The EULAR points to consider for use of antirheumatic drugs before pregnancy, and during pregnancy and lactation. Ann Rheum Dis. 2016;75:795-810.
- Nguyen GC, Seow CH, Maxwell C, et al. The Toronto consensus statements for the management of inflammatory bowel disease in pregnancy. Gastroenterology. 2016;150:734-757.
- Wise J. Rheumatic diseases should be actively treated in pregnancy, new guidelines say. BMJ. 2016;532:i312.
- Puig L, Carrascosa JM, Carretero G, et al. Spanish evidence-based guidelines on the treatment of psoriasis with biologic agents, 2013. part 1: on efficacy and choice of treatment. Actas Dermosifiliogr. 2013;104:694-709.
- Girolomoni G, Altomore G, Ayala F, et al. Differential management of mild-to-severe psoriasis with biologic drugs: an Italian Delphi consensus expert panel. J Dermatolog Treat. 2015;26:128-133.
- Yeung J, Gooderham MJ, Grewal P, et al. Management of plaque psoriasis with biologic therapies in women of child-bearing potential consensus paper. J Cutan Med Surg. 2020;24:3S-14S.
- Louthrenoo W, Kasitanon N, Kathamort W, et al. 2016 updated Thai Rheumatism Association Recommendations for the use of biologic and targeted synthetic disease-modifying anti-rheumatic drugs in patients with rheumatoid arthritis. Int J Rheum Dis. 2017;20:1166-1184.
- Flint J, Panchal S, Hurrell A, et al. BSR and BHPR guideline on prescribing drugs in pregnancy and breastfeeding—part I: standard and biologic disease modifying anti-rheumatic drugs and corticosteroids. Rheumatology (Oxford). 2016;55:1693-1697.
- van der Woude CJ, Ardizzone S, Bengtson MB, et al. The second European evidenced-based consensus on reproduction and pregnancy in inflammatory bowel disease. J Crohns Colitis. 2015;9:107-124.
- Orlando A, Armuzz A, Papi C, et al. The Italian Society of Gastroenterology (SIGE) and the Italian Group for the study of Inflammatory Bowel Disease (IG-IBD) Clinical Practice Guidelines: the use of tumor necrosis factor-alpha antagonist therapy in inflammatory bowel disease. Dig Liver Dis. 2011;43:1-20.
- Puchner A, Grochenig HP, Sautner J, et al. Immunosuppressives and biologics during pregnancy and lactation. Wien Klin Wochenschr. 2019;131:29-44.
- ACOG Committee opinion no. 776: immune modulating therapies in pregnancy and lactation. Obstet Gynecol. 2019;133:E287-E297.
- Lamb CA, Kennedy NA, Raine T, et al. British Society of Gastroenterology consensus guidelines on the management of inflammatory bowel disease in adults. Gut. 2019;68:S1-S106.
- Goëb V, Ardizzone M, Arnard L, et al. Recommendations for using TNF-α antagonists and French Clinical Practice Guidelines endorsed by the French National Authority for Health. Joint Bone Spine. 2013;80:574-581.
- Rademaker M, Agnew K, Andrews M, et al. Psoriasis in those planning a family, pregnant or breast-feeding. the Australasian Psoriasis Collaboration. Australas J Dermatol. 2018;59:86-100.
- Mahadevan U, Robinson C, Bernasko N, et al. Inflammatory bowel disease in pregnancy clinical care pathway: a report from the American Gastroenterological Association IBD Parenthood Project Working Group. Gastroenterology. 2019;156:1508-1524.
- Mahadevan U, Cucchiara S, Hyam JS, et al. The London position statement of the World Congress of Gastroenterology on biological therapy for IBD with the European Crohn’s and Colitis Organisation: pregnancy and pediatrics. Am J Gastroenterol. 2011;106:214-223.
- Porter ML, Lockwood SJ, Kimball AB. Update on biologic safety for patients with psoriasis during pregnancy. Int J Womens Dermatol. 2017;3:21-25.
- Bandoli G, Johnson DL, Jones KL, et al. Potentially modifiable risk factors for adverse pregnancy outcomes in women with psoriasis. Br J Dermatol. 2010;163:334-339.
- Lima XT, Janakiraman V, Hughes MD, et al. The impact of psoriasis on pregnancy outcomes. J Invest Dermatol. 2012;132:85-91.
- Bröms G, Haerskjold A, Granath F, et al. Effect of maternal psoriasis on pregnancy and birth outcomes: a population-based cohort study from Denmark and Sweden. Acta Derm Venereol. 2018;98:728-734.
- Bobotsis R, Gulliver WP, Monaghan K, et al. Psoriasis and adverse pregnancy outcomes: a systematic review of observational studies. Br J Dermatol. 2016;175:464-472.
- Blattner CM, Danesh M, Safaee M, et al. Understanding the new FDA pregnancy and lactation labeling rules. Int J Womens Dermatol. 2016;2:5-7.
- Pernia S, DeMaagd G. The new pregnancy and lactation labeling rule. P T. 2016;4:713-715.
- Chi C-C, Wang S-H, Wojnarowska F, et al. Safety of topical corticosteroids in pregnancy. Cochrane Database Syst Rev. 2015:CD007346.
- Chi CC, Wang SH, Kirtschig G. Safety of topical corticosteroids in pregnancy. JAMA Dermatol. 2016;152:934-935.
- Dovonex (calcipotriene) Cream, 0.005% [package insert]. Dublin, Ireland: Leo Laboratories, Ltd; March 2015.
- Franssen ME, van der Wilt GJ, de Jong PC, et al. A retrospective study of the teratogenicity of dermatological coal tar products. Acta Derm Venereol. 1999;79:390-391.
- Garbis H, Elefant E, Bertolotti E, et al. Pregnancy outcome after periconceptional and first-trimester exposure to methoxsalen photochemotherapy. Arch Dermatol. 1995;131:492-493.
- Horizon Pharma USA. RAYOS (prednisone). https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/202020s000lbl.pdf https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/202020s000lbl.pdf.
- Park-Wyllie L, Mazzotta P, Pastuszak A, et al. Birth defects after maternal exposure to corticosteroids: prospective cohort study and meta-analysis of epidemiological studies. Teratology. 2000;62:385-392.
- Crowther CA, Doyle LW, Haslam RR, et al. Outcomes at 2 years of age after repeat doses of antenatal corticosteroids. N Engl J Med. 2007;357:1179-1189.
- Palmsten K, Rolland M, Herbert MF, et al. Patterns of prednisone use during pregnancy in women with rheumatoid arthritis: daily and cumulative dose. Pharmacoepidemiol Drug Saf. 2018;27:430-438.
- Groth K, Brännström M, Mölne J, et al. Cyclosporine A exposure during pregnancy in mice: effects on reproductive performance in mothers and offspring. Hum Reprod. 2010;25:697-704.
- Bar Oz B, Hackman R, Einarson T, et al. Pregnancy outcome after cyclosporine therapy during pregnancy: a meta-analysis. Transplantation. 2001;71:1051-1055.
- Paziana K, Del Monaco M, Cardonick E, et al. Ciclosporin use during pregnancy. Drug Saf. 2013;36:279-294.
- Lamarque V, Leleu MF, Monka C, et al. Analysis of 629 pregnancy outcomes in transplant recipients treated with Sandimmun. Transplant Proc. 1997;29:2480.
- Otezla (apremilast) tablets, for oral use [package insert]. Summit, NJ: Celgene Corporation; June 2017. http://www.accessdata.fda.gov/drugsatfda_docs/label/2017/205437s006lbl.pdf. Accessed July 8, 2020.
- Kurizky PS, de Castro Ferreira C, Nogueira LSC, et al. Treatment of psoriasis and psoriatic arthritis during pregnancy and breastfeeding. An Bras Dermatol. 2015;90:367-375.
- Murase JE, Heller MM, Butler DC. Safety of dermatologic medications in pregnancy and lactation. J Am Acad Dermatol. 2014;70:401.e1-401.e4.
- El-Saie LT, Rabie AR, Kamel MI, et al. Effect of narrowband ultraviolet B phototherapy on serum folic acid levels in patients with psoriasis. Lasers Med Sci. 2011;26:481-485.
- Murase JE, Koo JY, Berger TG. Narrowband ultraviolet B phototherapy influences serum folate levels in patients with vitiligo. J Am Acad Dermatol. 2010;62:710-711.
- Soriatane (acitretin) capsules [package insert]. Morrisville, NC: Stiefel Laboratories, Inc; April 2011. http://www.accessdata.fda.gov/drugsatfda_docs/label/2011/019821s018mg.pdf. Accessed July 8, 2020.
- Kaushik SB, Lebwohl MG. Psoriasis: which therapy for which patient: focus on special populations and chronic infections. J Am Acad Dermatol. 2019;80:43-53.
- Help us better understand the effects of Otezla in pregnancy. MotherToBaby website. https://mothertobaby.org/ongoing-study/otezla/. Accessed July 8, 2020.
- Bangsgaard N, Rørbye C, Skov L. Treating psoriasis during pregnancy: safety and efficacy of treatments. Am J Clin Dermatol. 2015;16:389-398.
- Tyler K. Dermatologic therapy in pregnancy. Clin Obstet Gynecol. 2015;58:112-118.
- Luu M, Benzenine E, Doret M, et al. Continuous anti–TNF-α use throughout pregnancy: possible complications for the mother but not for the fetus. a retrospective cohort on the French National Health Insurance Database (EVASION). Am J Gastroenterol. 2018;113:1669-1677.
- Bröms G, Granath F, Ekbom A, et al. Low risk of birth defects for infants whose mothers are treated with anti-tumor necrosis factor agents during pregnancy. Clin Gastroenterol Hepatol. 2016;14:234-241.
- Mirdamadi K, Salinas T, Vali R, et al. Meta-analysis of pregnancy outcomes after exposure to TNF-α inhibitors during pregnancy for the treatment of arthritic diseases. J Popul Ther Clin Pharmacol. 2018;25:E53-E56.
- Shihab Z, Yeomans ND, De Cruz P. Anti-tumour necrosis factor α therapies and inflammatory bowel disease pregnancy outcomes: a meta-analysis. J Crohns Colitis. 2016;10:979-988.
- Bröms G, Kieler H, Ekbom A, et al. Anti-TNF treatment during pregnancy and birth outcomes: a population-based study from Denmark, Finland, and Sweden. Pharmacoepidemiol Drug Saf. 2020;29:316-327.
- Diav-Citrin O, Otcheretianski-Volodarsky A, Shechtman S, et al. Pregnancy outcome following gestational exposure to TNF-alpha-inhibitors: a prospective, comparative, observational study. Reprod Toxicol. 2014;43:78-84.
- FDA determinations for pregnant and nursing women. National Psoriasis Foundation website. http://www.psoriasis.org/pregnancy/fda-determinations. Accessed July 8, 2020.
- Østensen M. Safety issues of biologics in pregnant patients with rheumatic diseases. Ann N Y Acad Sci. 2014;1317:32-38.
- Chambers CD, Johnson DL, Luo Y, et al. Pregnancy outcome in women treated with adalimumab for the treatment of rheumatoid arthritis: the OTIS Autoimmune Diseases in Pregnancy Project. Arthritis Rheum. 2012;64:2466.
- Clowse ME, Wolf DC, Forger F, et al. Pregnancy outcomes after exposure to certolizumab pegol: updated results from a pharmacovigilance safety database. Arthritis Rheumatol. 2018;70:1399-1407.
- Carman WJ, Accortt NA, Anthony MS, et al. Pregnancy and infant outcomes including major congenital malformations among women with chronic inflammatory arthritis or psoriasis, with and without etanercept use. Pharmacoepidemiol Drug Saf. 2017;26:1109-1118.
- Janssen. SIMPONI (golilumab). https://www.accessdata.fda.gov/drugsatfda_docs/label/2011/125289s0064lbl.pdf.
- Yurkon K, Guo CY, Harrison D, et al. Pregnancy outcomes in women with dermatologic conditions exposed to infliximab. J Am Acad Dermatol. 2014;70:AB179.
- Watson N, Wu K, Farr P, et al. Ustekinumab exposure during conception and pregnancy in patients with chronic plaque psoriasis: a case series of 10 pregnancies. Br J Dermatol. 2019;180:195-196.
- Naureckas S, Slater J, Gearhart N, et al. Pregnancy outcomes in women with psoriasis and psoriatic arthritis exposed to ustekinumab. J Am Acad Dermatol. 2016;74:AB264.
- Haycraft K, DiRuggiero D, Rozzo SJ, et al. Outcomes of pregnancies from tildrakizumab phases I to III clinical development program. J Clin Aesthet Dermatol. 2019;12:S27-S28.
- Tremfya (guselkumab) injection, for subcutaneous use [package insert]. Horsham, PA: Janssen Biotech, Inc; July 2017. http://www.accessdata.fda.gov/drugsatfda_docs/label/2017/761061s000lbl.pdf. Accessed Juy 8, 2020.
- Skyrizi (risankizumab-rzaa) injection, for subcutaneous use [package insert]. Northi Chicago, IL; April 2019. http://www.accessdata.fda.gov/drugsatfda_docs/label/2019/761105s000lbl.pdf. Accessed July 8, 2020.
- Siliq (brodalumab) injection, for subcutaneous use [package insert]. Bridgewater, NJ: Valeant Pharmaceuticals North America LLC; February 2017. http://www.accessdata.fda.gov/drugsatfda_docs/label/2017/761032lbl.pdf. Accessed July 8, 2020.
- Feldman S, Pangallo B, Xu W, et al. Ixekizumab and pregnancy outcome. J Am Acad Dermatol. 2017;76:AB419.
- Clarke DO, Hilbish KG, Waters DG, et al. Assessment of ixekizumab, an interleukin-17A monoclonal antibody, for potential effects on reproduction and development, including immune system function, in cynomolgus monkeys. Reprod Toxicol. 2015;58:160-173.
- Warren RB, Reich K, Langley RG, et al. Secukinumab in pregnancy: outcomes in psoriasis, psoriatic arthritis and ankylosing spondylitis from the global safety database. Br J Dermatol. 2018;179:1205-1207.
- Nardin C, Colas M, Curie V, et al. Pregnancy after tubal sterilization in a woman treated with biologics for severe psoriasis. Dermatol Ther (Heidelb). 2018;8:323-326.
- Xeljanz (tofacitinib) tablets for oral administration [package insert]. New York, NY: Pfizer; November 2012. http://www.accessdata.fda.gov/drugsatfda_docs/label/2012/203214s000lbl.pdf. Accessed July 8, 2020.
- Pfizer. Xeljanz (tofacitinib). https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/203214s018lbl.pdf.
- Mahadevan U, Dubinsky M, Su C, et al. Outcomes of pregnancies with maternal/paternal exposure in the tofacitinib safety databases for ulcerative colitis. Inflamm Bowel Dis. 2018;24:2494-2500.
- Clowse ME, Feldman SR, Isaacs JD, et al. Pregnancy outcomes in the tofacitinib safety databases for rheumatoid arthritis and psoriasis. Drug Saf. 2016;39:755-762.
- Malek A, Sager R, Kuhn P, et al. Evolution of maternofetal transport of immunoglobulins during human pregnancy. Am J Reprod Immunol. 1996;36:248-255.
- Kimball AB, Crow JA, Ridley K, et al. Pregnancy outcomes in women with moderate to severe psoriasis: the PSOLAR experience. J Am Acad Dermatol. 2014;70(suppl 1):AB179.
- Carter JD, Valeriano J, Vasey FB. Tumor necrosis factor-alpha inhibition and VATER association: a causal relationship. J Rheumatol. 2006;33:1014-1017.
- Carter JD, Ladhani A, Ricca LR, et al. A safety assessment of tumor necrosis factor antagonists during pregnancy: a review of the Food and Drug Administration database. J Rheumatol. 2009;36:635-641.
- Koren G, Inoue M. Do tumor necrosis factor inhibitors cause malformations in humans? J Rheumatol. 2009;36:465-466.
- Johansen C, Jimenez-Solem E, Haerskjold A, et al. The use and safety of TNF inhibitors during pregnancy in women with psoriasis: a review. Int J Mol Sci. 2018;19:E1349.
- Mariette X, Forger F, Abraham B, et al. Lack of placental transfer of certolizumab pegol during pregnancy: results from CRIB, a prospective, postmarketing, pharmacokinetic study. Ann Rheum Dis. 2018;77:228-233.
- Mahadevan U, Wolf DC, Dubinsky M, et al. Placental transfer of anti-tumor necrosis factor agents in pregnant patients with inflammatory bowel disease. Clin Gastroenterol Hepatol. 2013;11:286-292.
- Cheent K, Nolan J, Sharig S, et al. Case report: fatal case of disseminated BCG infection in an infant born to a mother taking infliximab for Crohn’s disease. J Crohns Colitis. 2010;4:603-605.
- Julsgaard M, Christensen LA, Gibson PR, et al. Concentrations of adalimumab and infliximab in mothers and newborns, and effects on infection. Gastroenterology. 2016;151:110-119.
- Mahadevan U, Martin C, Kane SV, et al. Do infant serum levels of biologic agents at birth correlate with risk of adverse outcomes? results from the PIANO registry. Gastroenterology. 2016;150:S91-S92.
- Mahadevan U, Martin CF, Sandler RS, et al. PIANO: a 1000 patient prospective registry of pregnancy outcomes in women with IBD exposed to immunomodulators and biologic therapy [AGA abstract 865]. Gastroenterology. 2012;142:S-149.
Historically, there have been limited data available on the management of psoriasis in pregnancy. The most comprehensive discussion of treatment guidelines is from 2012.1 In the interim, many biologics have been approved for treating psoriasis, with slow accumulation of pregnancy safety data. The 2019 American Academy of Dermatology–National Psoriasis Foundation guidelines on biologics for psoriasis contain updated information but also highlight the paucity of pregnancy safety data.2 This gap is in part a consequence of the exclusion and disenrollment of pregnant women from clinical trials.3 Additionally, lack of detection through registries contributes; pregnancy capture in registries is low compared to the expected number of pregnancies estimated from US Census data.4 Despite these shortcomings, psoriasis patients who are already pregnant or are considering becoming pregnant frequently are encountered in practice and may need treatment. This article reviews the evidence on commonly used treatments for psoriasis in pregnancy.
Background
For many patients, psoriasis improves during pregnancy5,6 and becomes worse postpartum. In a prospective study, most patients reported improvement in pregnancy corresponding to a significant decrease in
In addition to the maternal disease state, the issue of pregnancy outcomes is paramount. In the inflammatory bowel disease and rheumatology literature, it is established that uncontrolled disease is associated with poorer pregnancy outcomes.8-10 Guidelines vary among societies on the use of biologics in pregnancy generally (eTable 11,2,9,11-24), but some societies recommend systemic agents to achieve disease control during pregnancy.9,25
Assessing the potential interplay between disease severity and outcomes in pregnant women with psoriasis is further complicated by the slowly growing body of literature demonstrating that women with psoriasis have more comorbidities26 and worse pregnancy outcomes.27,28 Pregnant psoriasis patients are more likely to smoke, have depression, and be overweight or obese prior to pregnancy and are less likely to take prenatal vitamins.26 They also have an increased risk for cesarean birth, gestational diabetes, gestational hypertension, and preeclampsia.28 In contrast to these prior studies, a systematic review revealed no risk for adverse outcomes in pregnant women with psoriasis.29
Assessment of Treatments for Psoriasis in Pregnancy
In light of these issues, treatment of psoriasis during pregnancy should be assessed from several vantage points. Of note, the US Food and Drug Administration changed its classification scheme in 2015 to a more narrative format called the Pregnancy and Lactation Labeling Rule.30 Prior classifications, however, provide a reasonable starting point for categorizing the safety of drugs (Table31). Importantly, time of exposure to systemic agents also matters; first-trimester exposure is more likely to affect embryogenesis, whereas second- and third-trimester exposures are more prone to affect other aspects of fetal growth. eTable 2 provides data on the use of oral and topical medications to treat psoriasis in pregnancy.1,8,22,32-45

Topical Agents
Topical steroids are largely understood to be reasonable treatment options, though consideration of potency, formulation, area of application, and use of occlusion is important.1,46 Risk for orofacial cleft has been noted with first-trimester topical steroid exposure, though a 2015 Cochrane review update determined that the relative risk of this association was not significantly elevated.32
The impact of topical calcipotriene and salicylic acid has not been studied in human pregnancies,1 but systemic absorption can occur for both. There is potential for vitamin D toxicity with calcipotriene46; consequently, use during pregnancy is not recommended.1,46 Some authors recommend against topical salicylic acid in pregnancy; others report that limited exposure is permissible.47 In fact, as salicylic acid commonly is found in over-the-counter acne products, many women of childbearing potential likely have quotidian exposure.
Preterm delivery and low birthweight have been reported with oral tacrolimus; however, risk with topical tacrolimus is thought to be low1 because the molecular size likely prohibits notable absorption.47 Evidence for the use of anthralin and coal tar also is scarce. First-trimester coal tar use should be avoided; subsequent use in pregnancy should be restricted given concern for adverse outcomes.1
Phototherapy
Broadband or narrowband UVB therapy is recommended as second-line therapy in pregnancy. No cases of fetal risk or premature delivery associated with UVB therapy were found in our search.1 Phototherapy can exacerbate melasma47 and decrease folate levels48; as such, some authors recommend folate supplementation in females of childbearing age who are being treated with phototherapy.49 Psoralen, used in psoralen plus UVA therapy, is mutagenic and therefore contraindicated in pregnancy.1
Oral Medications
Both methotrexate, which is a teratogen, abortifacient, and mutagen,1 and systemic retinoids, which are teratogens, are contraindicated in pregnancy.1,47 Acitretin labeling recommends avoiding pregnancy for 3 years posttreatment50 because alcohol intake prolongs the medication’s half-life.22
Apremilast use is not documented in pregnant psoriasis patients51; an ongoing registry of the Organization of Tetralogy Information Specialists has not reported publicly to date.52 Animal studies of apremilast have documented dose-related decreased birthweight and fetal loss.22
Safety data for systemic steroids, used infrequently in psoriasis, are not well established. First-trimester prednisone exposure has been associated with prematurity, low birthweight, and congenital abnormalities.38 A separate evaluation of 1047 children exposed to betamethasone in utero failed to demonstrate significant change in birthweight or head circumference. However, repeat antenatal corticosteroid exposure was associated with attention problems at 2 years of age.39
Data regarding cyclosporine use, derived primarily from organ transplant recipients, suggest elevated risk for prematurity and low birthweight.53,54 A meta-analysis demonstrated that organ transplant recipients taking cyclosporine had a nonsignificantly elevated odds ratio for congenital malformations, prematurity, and low birthweight.42 Cyclosporine use for psoriasis in pregnancy is not well described; in a study, rates of prematurity and low birthweight were both 21%.43 Limited data are available for Janus kinase inhibitors, none of which are approved for psoriasis, though clinical trials in psoriasis and psoriatic arthritis are underway (ClinicalTrials.gov identifiers NCT04246372, NCT03104374, NCT03104400).
Biologics and Small-Molecule Inhibitors
Limited data on biologics in pregnancy exist25 (eTable 3). Placental transport of IgG antibodies, including biologics, increases throughout pregnancy, especially in the third trimester.82 Infants of mothers treated with a biologic with potential for placental transfer are therefore considered by some authors to be immunosuppressed during the first months of life.2
Looking globally across biologics used for psoriasis, limited safety data are encouraging. In a review of PSOLAR (Psoriasis Longitudinal Assessment and Registry), 83 pregnancies with biologic exposure resulted in 59 live births (71%); 18 spontaneous abortions (22%); 6 induced abortions (7%); no congenital abnormalities; and 7 reports of neonatal problems, including respiratory issues, ABO blood group mismatch, hospitalization, and opioid withdrawal.83
Use of tumor necrosis factor (TNF) inhibitors in pregnancy has the most data25 and is considered a reasonable treatment option. Historically, there was concern about the risk for VACTERL syndrome (vertebral defects, anal atresia, cardiac defects, tracheoesophageal fistula, renal anomalies, limb abnormalities) with exposure to a TNF inhibitor,25,84-86 but further reports have alleviated these concerns. Active transplacental transport occurs for adalimumab, infliximab, and golimumab,87 but given structural differences, transport of certolizumab and etanercept is substantially less.88,89 In the CRIB study of placental transfer of certolizumab from mother to infant (N=14), pharmacokinetic data demonstrated no quantifiable certolizumab levels in 13 infants and minimal levels in 1 infant at birth.88 There are fewer data available on the use of other biologics in pregnancy, but for those in which active placental transport is relevant, similar concerns (ie, immunosuppression) might arise (eTable 3).
Concern over biologics largely involves risk for newborn immunosuppression. A case report detailed a Crohn disease patient treated with infliximab who gave birth to an infant who died of disseminated bacille Calmette-Guérin infection at 4.5 months after receiving the vaccine at 3 months.90 This case underscores the importance of delaying live vaccination in infants born to mothers who were treated with a biologic during pregnancy. Authors have provided various data on how long to avoid vaccination; some state as long as 1 year.91
In pregnant females with inflammatory bowel disease treated with a biologic, no correlation was observed among maternal, placental, and infant serum biologic levels and neonatal infection. However, an association between preterm birth and the level of the biologic in maternal and placental (but not infant) serum and preterm birth was observed.92
In another report from the same registry, combination therapy with a TNF inhibitor and another immunomodulator led to an increased risk for infection in infants at 12 months of age, compared to infants exposed to monotherapy89 or exposed to neither agent.93 A strategy to circumvent this potential problem is to avoid treatment with actively transported molecules in the third trimester.
Conclusion
Limited data exist to guide providers who are treating pregnant women with psoriasis. Our understanding of treatment of psoriasis in pregnancy is limited as a consequence of regulations surrounding clinical trials and inadequate detection of pregnancies in registries. Further efforts are necessary to better understand the relationship between psoriasis and pregnancy and how to manage pregnant women with psoriasis.
Historically, there have been limited data available on the management of psoriasis in pregnancy. The most comprehensive discussion of treatment guidelines is from 2012.1 In the interim, many biologics have been approved for treating psoriasis, with slow accumulation of pregnancy safety data. The 2019 American Academy of Dermatology–National Psoriasis Foundation guidelines on biologics for psoriasis contain updated information but also highlight the paucity of pregnancy safety data.2 This gap is in part a consequence of the exclusion and disenrollment of pregnant women from clinical trials.3 Additionally, lack of detection through registries contributes; pregnancy capture in registries is low compared to the expected number of pregnancies estimated from US Census data.4 Despite these shortcomings, psoriasis patients who are already pregnant or are considering becoming pregnant frequently are encountered in practice and may need treatment. This article reviews the evidence on commonly used treatments for psoriasis in pregnancy.
Background
For many patients, psoriasis improves during pregnancy5,6 and becomes worse postpartum. In a prospective study, most patients reported improvement in pregnancy corresponding to a significant decrease in
In addition to the maternal disease state, the issue of pregnancy outcomes is paramount. In the inflammatory bowel disease and rheumatology literature, it is established that uncontrolled disease is associated with poorer pregnancy outcomes.8-10 Guidelines vary among societies on the use of biologics in pregnancy generally (eTable 11,2,9,11-24), but some societies recommend systemic agents to achieve disease control during pregnancy.9,25
Assessing the potential interplay between disease severity and outcomes in pregnant women with psoriasis is further complicated by the slowly growing body of literature demonstrating that women with psoriasis have more comorbidities26 and worse pregnancy outcomes.27,28 Pregnant psoriasis patients are more likely to smoke, have depression, and be overweight or obese prior to pregnancy and are less likely to take prenatal vitamins.26 They also have an increased risk for cesarean birth, gestational diabetes, gestational hypertension, and preeclampsia.28 In contrast to these prior studies, a systematic review revealed no risk for adverse outcomes in pregnant women with psoriasis.29
Assessment of Treatments for Psoriasis in Pregnancy
In light of these issues, treatment of psoriasis during pregnancy should be assessed from several vantage points. Of note, the US Food and Drug Administration changed its classification scheme in 2015 to a more narrative format called the Pregnancy and Lactation Labeling Rule.30 Prior classifications, however, provide a reasonable starting point for categorizing the safety of drugs (Table31). Importantly, time of exposure to systemic agents also matters; first-trimester exposure is more likely to affect embryogenesis, whereas second- and third-trimester exposures are more prone to affect other aspects of fetal growth. eTable 2 provides data on the use of oral and topical medications to treat psoriasis in pregnancy.1,8,22,32-45

Topical Agents
Topical steroids are largely understood to be reasonable treatment options, though consideration of potency, formulation, area of application, and use of occlusion is important.1,46 Risk for orofacial cleft has been noted with first-trimester topical steroid exposure, though a 2015 Cochrane review update determined that the relative risk of this association was not significantly elevated.32
The impact of topical calcipotriene and salicylic acid has not been studied in human pregnancies,1 but systemic absorption can occur for both. There is potential for vitamin D toxicity with calcipotriene46; consequently, use during pregnancy is not recommended.1,46 Some authors recommend against topical salicylic acid in pregnancy; others report that limited exposure is permissible.47 In fact, as salicylic acid commonly is found in over-the-counter acne products, many women of childbearing potential likely have quotidian exposure.
Preterm delivery and low birthweight have been reported with oral tacrolimus; however, risk with topical tacrolimus is thought to be low1 because the molecular size likely prohibits notable absorption.47 Evidence for the use of anthralin and coal tar also is scarce. First-trimester coal tar use should be avoided; subsequent use in pregnancy should be restricted given concern for adverse outcomes.1
Phototherapy
Broadband or narrowband UVB therapy is recommended as second-line therapy in pregnancy. No cases of fetal risk or premature delivery associated with UVB therapy were found in our search.1 Phototherapy can exacerbate melasma47 and decrease folate levels48; as such, some authors recommend folate supplementation in females of childbearing age who are being treated with phototherapy.49 Psoralen, used in psoralen plus UVA therapy, is mutagenic and therefore contraindicated in pregnancy.1
Oral Medications
Both methotrexate, which is a teratogen, abortifacient, and mutagen,1 and systemic retinoids, which are teratogens, are contraindicated in pregnancy.1,47 Acitretin labeling recommends avoiding pregnancy for 3 years posttreatment50 because alcohol intake prolongs the medication’s half-life.22
Apremilast use is not documented in pregnant psoriasis patients51; an ongoing registry of the Organization of Tetralogy Information Specialists has not reported publicly to date.52 Animal studies of apremilast have documented dose-related decreased birthweight and fetal loss.22
Safety data for systemic steroids, used infrequently in psoriasis, are not well established. First-trimester prednisone exposure has been associated with prematurity, low birthweight, and congenital abnormalities.38 A separate evaluation of 1047 children exposed to betamethasone in utero failed to demonstrate significant change in birthweight or head circumference. However, repeat antenatal corticosteroid exposure was associated with attention problems at 2 years of age.39
Data regarding cyclosporine use, derived primarily from organ transplant recipients, suggest elevated risk for prematurity and low birthweight.53,54 A meta-analysis demonstrated that organ transplant recipients taking cyclosporine had a nonsignificantly elevated odds ratio for congenital malformations, prematurity, and low birthweight.42 Cyclosporine use for psoriasis in pregnancy is not well described; in a study, rates of prematurity and low birthweight were both 21%.43 Limited data are available for Janus kinase inhibitors, none of which are approved for psoriasis, though clinical trials in psoriasis and psoriatic arthritis are underway (ClinicalTrials.gov identifiers NCT04246372, NCT03104374, NCT03104400).
Biologics and Small-Molecule Inhibitors
Limited data on biologics in pregnancy exist25 (eTable 3). Placental transport of IgG antibodies, including biologics, increases throughout pregnancy, especially in the third trimester.82 Infants of mothers treated with a biologic with potential for placental transfer are therefore considered by some authors to be immunosuppressed during the first months of life.2
Looking globally across biologics used for psoriasis, limited safety data are encouraging. In a review of PSOLAR (Psoriasis Longitudinal Assessment and Registry), 83 pregnancies with biologic exposure resulted in 59 live births (71%); 18 spontaneous abortions (22%); 6 induced abortions (7%); no congenital abnormalities; and 7 reports of neonatal problems, including respiratory issues, ABO blood group mismatch, hospitalization, and opioid withdrawal.83
Use of tumor necrosis factor (TNF) inhibitors in pregnancy has the most data25 and is considered a reasonable treatment option. Historically, there was concern about the risk for VACTERL syndrome (vertebral defects, anal atresia, cardiac defects, tracheoesophageal fistula, renal anomalies, limb abnormalities) with exposure to a TNF inhibitor,25,84-86 but further reports have alleviated these concerns. Active transplacental transport occurs for adalimumab, infliximab, and golimumab,87 but given structural differences, transport of certolizumab and etanercept is substantially less.88,89 In the CRIB study of placental transfer of certolizumab from mother to infant (N=14), pharmacokinetic data demonstrated no quantifiable certolizumab levels in 13 infants and minimal levels in 1 infant at birth.88 There are fewer data available on the use of other biologics in pregnancy, but for those in which active placental transport is relevant, similar concerns (ie, immunosuppression) might arise (eTable 3).
Concern over biologics largely involves risk for newborn immunosuppression. A case report detailed a Crohn disease patient treated with infliximab who gave birth to an infant who died of disseminated bacille Calmette-Guérin infection at 4.5 months after receiving the vaccine at 3 months.90 This case underscores the importance of delaying live vaccination in infants born to mothers who were treated with a biologic during pregnancy. Authors have provided various data on how long to avoid vaccination; some state as long as 1 year.91
In pregnant females with inflammatory bowel disease treated with a biologic, no correlation was observed among maternal, placental, and infant serum biologic levels and neonatal infection. However, an association between preterm birth and the level of the biologic in maternal and placental (but not infant) serum and preterm birth was observed.92
In another report from the same registry, combination therapy with a TNF inhibitor and another immunomodulator led to an increased risk for infection in infants at 12 months of age, compared to infants exposed to monotherapy89 or exposed to neither agent.93 A strategy to circumvent this potential problem is to avoid treatment with actively transported molecules in the third trimester.
Conclusion
Limited data exist to guide providers who are treating pregnant women with psoriasis. Our understanding of treatment of psoriasis in pregnancy is limited as a consequence of regulations surrounding clinical trials and inadequate detection of pregnancies in registries. Further efforts are necessary to better understand the relationship between psoriasis and pregnancy and how to manage pregnant women with psoriasis.
- Bae YS, Van Voorhees AS, Hsu S, et al. Review of treatment options for psoriasis in pregnant or lactating women: from the medical board of the National Psoriasis Foundation. J Am Acad Dermatol. 2012;67:459-477.
- Menter A, Strober BE, Kaplan DH, et al. Joint AAD-NPF guidelines of care for the management and treatment of psoriasis with biologics. J Am Acad Dermatol. 2019;80:1029-1072.
- Flood KS, Porter ML, Kimball AB. Use of biologics in pregnancy: limitations stemming from clinical trials and registry experience. J Eur Acad Dermatol Venereol. 2019;33:E276-E277.
- Horn EJ, Chambers CD, Menter A, et al. Pregnancy outcomes in psoriasis: why do we know so little? J Am Acad Dermatol. 2009;61:E5-E8.
- Raychaudhuri SP, Navare T, Gross J, et al. Clinical course of psoriasis during pregnancy. Int J Dermatol. 2003;42:518-520.
- Boyd AS, Morris LF, Phillips CM, et al. Psoriasis and pregnancy: hormone and immune system interaction. Int J Dermatol. 1996;35:169-172.
- Murase JE, Chan KK, Garite TJ, et al. Hormonal effect on psoriasis in pregnancy and post partum. Arch Dermatol. 2005;14:601-606.
- Götestam Skorpen C, Hoeltzenbein M, Tincani A, et al. The EULAR points to consider for use of antirheumatic drugs before pregnancy, and during pregnancy and lactation. Ann Rheum Dis. 2016;75:795-810.
- Nguyen GC, Seow CH, Maxwell C, et al. The Toronto consensus statements for the management of inflammatory bowel disease in pregnancy. Gastroenterology. 2016;150:734-757.
- Wise J. Rheumatic diseases should be actively treated in pregnancy, new guidelines say. BMJ. 2016;532:i312.
- Puig L, Carrascosa JM, Carretero G, et al. Spanish evidence-based guidelines on the treatment of psoriasis with biologic agents, 2013. part 1: on efficacy and choice of treatment. Actas Dermosifiliogr. 2013;104:694-709.
- Girolomoni G, Altomore G, Ayala F, et al. Differential management of mild-to-severe psoriasis with biologic drugs: an Italian Delphi consensus expert panel. J Dermatolog Treat. 2015;26:128-133.
- Yeung J, Gooderham MJ, Grewal P, et al. Management of plaque psoriasis with biologic therapies in women of child-bearing potential consensus paper. J Cutan Med Surg. 2020;24:3S-14S.
- Louthrenoo W, Kasitanon N, Kathamort W, et al. 2016 updated Thai Rheumatism Association Recommendations for the use of biologic and targeted synthetic disease-modifying anti-rheumatic drugs in patients with rheumatoid arthritis. Int J Rheum Dis. 2017;20:1166-1184.
- Flint J, Panchal S, Hurrell A, et al. BSR and BHPR guideline on prescribing drugs in pregnancy and breastfeeding—part I: standard and biologic disease modifying anti-rheumatic drugs and corticosteroids. Rheumatology (Oxford). 2016;55:1693-1697.
- van der Woude CJ, Ardizzone S, Bengtson MB, et al. The second European evidenced-based consensus on reproduction and pregnancy in inflammatory bowel disease. J Crohns Colitis. 2015;9:107-124.
- Orlando A, Armuzz A, Papi C, et al. The Italian Society of Gastroenterology (SIGE) and the Italian Group for the study of Inflammatory Bowel Disease (IG-IBD) Clinical Practice Guidelines: the use of tumor necrosis factor-alpha antagonist therapy in inflammatory bowel disease. Dig Liver Dis. 2011;43:1-20.
- Puchner A, Grochenig HP, Sautner J, et al. Immunosuppressives and biologics during pregnancy and lactation. Wien Klin Wochenschr. 2019;131:29-44.
- ACOG Committee opinion no. 776: immune modulating therapies in pregnancy and lactation. Obstet Gynecol. 2019;133:E287-E297.
- Lamb CA, Kennedy NA, Raine T, et al. British Society of Gastroenterology consensus guidelines on the management of inflammatory bowel disease in adults. Gut. 2019;68:S1-S106.
- Goëb V, Ardizzone M, Arnard L, et al. Recommendations for using TNF-α antagonists and French Clinical Practice Guidelines endorsed by the French National Authority for Health. Joint Bone Spine. 2013;80:574-581.
- Rademaker M, Agnew K, Andrews M, et al. Psoriasis in those planning a family, pregnant or breast-feeding. the Australasian Psoriasis Collaboration. Australas J Dermatol. 2018;59:86-100.
- Mahadevan U, Robinson C, Bernasko N, et al. Inflammatory bowel disease in pregnancy clinical care pathway: a report from the American Gastroenterological Association IBD Parenthood Project Working Group. Gastroenterology. 2019;156:1508-1524.
- Mahadevan U, Cucchiara S, Hyam JS, et al. The London position statement of the World Congress of Gastroenterology on biological therapy for IBD with the European Crohn’s and Colitis Organisation: pregnancy and pediatrics. Am J Gastroenterol. 2011;106:214-223.
- Porter ML, Lockwood SJ, Kimball AB. Update on biologic safety for patients with psoriasis during pregnancy. Int J Womens Dermatol. 2017;3:21-25.
- Bandoli G, Johnson DL, Jones KL, et al. Potentially modifiable risk factors for adverse pregnancy outcomes in women with psoriasis. Br J Dermatol. 2010;163:334-339.
- Lima XT, Janakiraman V, Hughes MD, et al. The impact of psoriasis on pregnancy outcomes. J Invest Dermatol. 2012;132:85-91.
- Bröms G, Haerskjold A, Granath F, et al. Effect of maternal psoriasis on pregnancy and birth outcomes: a population-based cohort study from Denmark and Sweden. Acta Derm Venereol. 2018;98:728-734.
- Bobotsis R, Gulliver WP, Monaghan K, et al. Psoriasis and adverse pregnancy outcomes: a systematic review of observational studies. Br J Dermatol. 2016;175:464-472.
- Blattner CM, Danesh M, Safaee M, et al. Understanding the new FDA pregnancy and lactation labeling rules. Int J Womens Dermatol. 2016;2:5-7.
- Pernia S, DeMaagd G. The new pregnancy and lactation labeling rule. P T. 2016;4:713-715.
- Chi C-C, Wang S-H, Wojnarowska F, et al. Safety of topical corticosteroids in pregnancy. Cochrane Database Syst Rev. 2015:CD007346.
- Chi CC, Wang SH, Kirtschig G. Safety of topical corticosteroids in pregnancy. JAMA Dermatol. 2016;152:934-935.
- Dovonex (calcipotriene) Cream, 0.005% [package insert]. Dublin, Ireland: Leo Laboratories, Ltd; March 2015.
- Franssen ME, van der Wilt GJ, de Jong PC, et al. A retrospective study of the teratogenicity of dermatological coal tar products. Acta Derm Venereol. 1999;79:390-391.
- Garbis H, Elefant E, Bertolotti E, et al. Pregnancy outcome after periconceptional and first-trimester exposure to methoxsalen photochemotherapy. Arch Dermatol. 1995;131:492-493.
- Horizon Pharma USA. RAYOS (prednisone). https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/202020s000lbl.pdf https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/202020s000lbl.pdf.
- Park-Wyllie L, Mazzotta P, Pastuszak A, et al. Birth defects after maternal exposure to corticosteroids: prospective cohort study and meta-analysis of epidemiological studies. Teratology. 2000;62:385-392.
- Crowther CA, Doyle LW, Haslam RR, et al. Outcomes at 2 years of age after repeat doses of antenatal corticosteroids. N Engl J Med. 2007;357:1179-1189.
- Palmsten K, Rolland M, Herbert MF, et al. Patterns of prednisone use during pregnancy in women with rheumatoid arthritis: daily and cumulative dose. Pharmacoepidemiol Drug Saf. 2018;27:430-438.
- Groth K, Brännström M, Mölne J, et al. Cyclosporine A exposure during pregnancy in mice: effects on reproductive performance in mothers and offspring. Hum Reprod. 2010;25:697-704.
- Bar Oz B, Hackman R, Einarson T, et al. Pregnancy outcome after cyclosporine therapy during pregnancy: a meta-analysis. Transplantation. 2001;71:1051-1055.
- Paziana K, Del Monaco M, Cardonick E, et al. Ciclosporin use during pregnancy. Drug Saf. 2013;36:279-294.
- Lamarque V, Leleu MF, Monka C, et al. Analysis of 629 pregnancy outcomes in transplant recipients treated with Sandimmun. Transplant Proc. 1997;29:2480.
- Otezla (apremilast) tablets, for oral use [package insert]. Summit, NJ: Celgene Corporation; June 2017. http://www.accessdata.fda.gov/drugsatfda_docs/label/2017/205437s006lbl.pdf. Accessed July 8, 2020.
- Kurizky PS, de Castro Ferreira C, Nogueira LSC, et al. Treatment of psoriasis and psoriatic arthritis during pregnancy and breastfeeding. An Bras Dermatol. 2015;90:367-375.
- Murase JE, Heller MM, Butler DC. Safety of dermatologic medications in pregnancy and lactation. J Am Acad Dermatol. 2014;70:401.e1-401.e4.
- El-Saie LT, Rabie AR, Kamel MI, et al. Effect of narrowband ultraviolet B phototherapy on serum folic acid levels in patients with psoriasis. Lasers Med Sci. 2011;26:481-485.
- Murase JE, Koo JY, Berger TG. Narrowband ultraviolet B phototherapy influences serum folate levels in patients with vitiligo. J Am Acad Dermatol. 2010;62:710-711.
- Soriatane (acitretin) capsules [package insert]. Morrisville, NC: Stiefel Laboratories, Inc; April 2011. http://www.accessdata.fda.gov/drugsatfda_docs/label/2011/019821s018mg.pdf. Accessed July 8, 2020.
- Kaushik SB, Lebwohl MG. Psoriasis: which therapy for which patient: focus on special populations and chronic infections. J Am Acad Dermatol. 2019;80:43-53.
- Help us better understand the effects of Otezla in pregnancy. MotherToBaby website. https://mothertobaby.org/ongoing-study/otezla/. Accessed July 8, 2020.
- Bangsgaard N, Rørbye C, Skov L. Treating psoriasis during pregnancy: safety and efficacy of treatments. Am J Clin Dermatol. 2015;16:389-398.
- Tyler K. Dermatologic therapy in pregnancy. Clin Obstet Gynecol. 2015;58:112-118.
- Luu M, Benzenine E, Doret M, et al. Continuous anti–TNF-α use throughout pregnancy: possible complications for the mother but not for the fetus. a retrospective cohort on the French National Health Insurance Database (EVASION). Am J Gastroenterol. 2018;113:1669-1677.
- Bröms G, Granath F, Ekbom A, et al. Low risk of birth defects for infants whose mothers are treated with anti-tumor necrosis factor agents during pregnancy. Clin Gastroenterol Hepatol. 2016;14:234-241.
- Mirdamadi K, Salinas T, Vali R, et al. Meta-analysis of pregnancy outcomes after exposure to TNF-α inhibitors during pregnancy for the treatment of arthritic diseases. J Popul Ther Clin Pharmacol. 2018;25:E53-E56.
- Shihab Z, Yeomans ND, De Cruz P. Anti-tumour necrosis factor α therapies and inflammatory bowel disease pregnancy outcomes: a meta-analysis. J Crohns Colitis. 2016;10:979-988.
- Bröms G, Kieler H, Ekbom A, et al. Anti-TNF treatment during pregnancy and birth outcomes: a population-based study from Denmark, Finland, and Sweden. Pharmacoepidemiol Drug Saf. 2020;29:316-327.
- Diav-Citrin O, Otcheretianski-Volodarsky A, Shechtman S, et al. Pregnancy outcome following gestational exposure to TNF-alpha-inhibitors: a prospective, comparative, observational study. Reprod Toxicol. 2014;43:78-84.
- FDA determinations for pregnant and nursing women. National Psoriasis Foundation website. http://www.psoriasis.org/pregnancy/fda-determinations. Accessed July 8, 2020.
- Østensen M. Safety issues of biologics in pregnant patients with rheumatic diseases. Ann N Y Acad Sci. 2014;1317:32-38.
- Chambers CD, Johnson DL, Luo Y, et al. Pregnancy outcome in women treated with adalimumab for the treatment of rheumatoid arthritis: the OTIS Autoimmune Diseases in Pregnancy Project. Arthritis Rheum. 2012;64:2466.
- Clowse ME, Wolf DC, Forger F, et al. Pregnancy outcomes after exposure to certolizumab pegol: updated results from a pharmacovigilance safety database. Arthritis Rheumatol. 2018;70:1399-1407.
- Carman WJ, Accortt NA, Anthony MS, et al. Pregnancy and infant outcomes including major congenital malformations among women with chronic inflammatory arthritis or psoriasis, with and without etanercept use. Pharmacoepidemiol Drug Saf. 2017;26:1109-1118.
- Janssen. SIMPONI (golilumab). https://www.accessdata.fda.gov/drugsatfda_docs/label/2011/125289s0064lbl.pdf.
- Yurkon K, Guo CY, Harrison D, et al. Pregnancy outcomes in women with dermatologic conditions exposed to infliximab. J Am Acad Dermatol. 2014;70:AB179.
- Watson N, Wu K, Farr P, et al. Ustekinumab exposure during conception and pregnancy in patients with chronic plaque psoriasis: a case series of 10 pregnancies. Br J Dermatol. 2019;180:195-196.
- Naureckas S, Slater J, Gearhart N, et al. Pregnancy outcomes in women with psoriasis and psoriatic arthritis exposed to ustekinumab. J Am Acad Dermatol. 2016;74:AB264.
- Haycraft K, DiRuggiero D, Rozzo SJ, et al. Outcomes of pregnancies from tildrakizumab phases I to III clinical development program. J Clin Aesthet Dermatol. 2019;12:S27-S28.
- Tremfya (guselkumab) injection, for subcutaneous use [package insert]. Horsham, PA: Janssen Biotech, Inc; July 2017. http://www.accessdata.fda.gov/drugsatfda_docs/label/2017/761061s000lbl.pdf. Accessed Juy 8, 2020.
- Skyrizi (risankizumab-rzaa) injection, for subcutaneous use [package insert]. Northi Chicago, IL; April 2019. http://www.accessdata.fda.gov/drugsatfda_docs/label/2019/761105s000lbl.pdf. Accessed July 8, 2020.
- Siliq (brodalumab) injection, for subcutaneous use [package insert]. Bridgewater, NJ: Valeant Pharmaceuticals North America LLC; February 2017. http://www.accessdata.fda.gov/drugsatfda_docs/label/2017/761032lbl.pdf. Accessed July 8, 2020.
- Feldman S, Pangallo B, Xu W, et al. Ixekizumab and pregnancy outcome. J Am Acad Dermatol. 2017;76:AB419.
- Clarke DO, Hilbish KG, Waters DG, et al. Assessment of ixekizumab, an interleukin-17A monoclonal antibody, for potential effects on reproduction and development, including immune system function, in cynomolgus monkeys. Reprod Toxicol. 2015;58:160-173.
- Warren RB, Reich K, Langley RG, et al. Secukinumab in pregnancy: outcomes in psoriasis, psoriatic arthritis and ankylosing spondylitis from the global safety database. Br J Dermatol. 2018;179:1205-1207.
- Nardin C, Colas M, Curie V, et al. Pregnancy after tubal sterilization in a woman treated with biologics for severe psoriasis. Dermatol Ther (Heidelb). 2018;8:323-326.
- Xeljanz (tofacitinib) tablets for oral administration [package insert]. New York, NY: Pfizer; November 2012. http://www.accessdata.fda.gov/drugsatfda_docs/label/2012/203214s000lbl.pdf. Accessed July 8, 2020.
- Pfizer. Xeljanz (tofacitinib). https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/203214s018lbl.pdf.
- Mahadevan U, Dubinsky M, Su C, et al. Outcomes of pregnancies with maternal/paternal exposure in the tofacitinib safety databases for ulcerative colitis. Inflamm Bowel Dis. 2018;24:2494-2500.
- Clowse ME, Feldman SR, Isaacs JD, et al. Pregnancy outcomes in the tofacitinib safety databases for rheumatoid arthritis and psoriasis. Drug Saf. 2016;39:755-762.
- Malek A, Sager R, Kuhn P, et al. Evolution of maternofetal transport of immunoglobulins during human pregnancy. Am J Reprod Immunol. 1996;36:248-255.
- Kimball AB, Crow JA, Ridley K, et al. Pregnancy outcomes in women with moderate to severe psoriasis: the PSOLAR experience. J Am Acad Dermatol. 2014;70(suppl 1):AB179.
- Carter JD, Valeriano J, Vasey FB. Tumor necrosis factor-alpha inhibition and VATER association: a causal relationship. J Rheumatol. 2006;33:1014-1017.
- Carter JD, Ladhani A, Ricca LR, et al. A safety assessment of tumor necrosis factor antagonists during pregnancy: a review of the Food and Drug Administration database. J Rheumatol. 2009;36:635-641.
- Koren G, Inoue M. Do tumor necrosis factor inhibitors cause malformations in humans? J Rheumatol. 2009;36:465-466.
- Johansen C, Jimenez-Solem E, Haerskjold A, et al. The use and safety of TNF inhibitors during pregnancy in women with psoriasis: a review. Int J Mol Sci. 2018;19:E1349.
- Mariette X, Forger F, Abraham B, et al. Lack of placental transfer of certolizumab pegol during pregnancy: results from CRIB, a prospective, postmarketing, pharmacokinetic study. Ann Rheum Dis. 2018;77:228-233.
- Mahadevan U, Wolf DC, Dubinsky M, et al. Placental transfer of anti-tumor necrosis factor agents in pregnant patients with inflammatory bowel disease. Clin Gastroenterol Hepatol. 2013;11:286-292.
- Cheent K, Nolan J, Sharig S, et al. Case report: fatal case of disseminated BCG infection in an infant born to a mother taking infliximab for Crohn’s disease. J Crohns Colitis. 2010;4:603-605.
- Julsgaard M, Christensen LA, Gibson PR, et al. Concentrations of adalimumab and infliximab in mothers and newborns, and effects on infection. Gastroenterology. 2016;151:110-119.
- Mahadevan U, Martin C, Kane SV, et al. Do infant serum levels of biologic agents at birth correlate with risk of adverse outcomes? results from the PIANO registry. Gastroenterology. 2016;150:S91-S92.
- Mahadevan U, Martin CF, Sandler RS, et al. PIANO: a 1000 patient prospective registry of pregnancy outcomes in women with IBD exposed to immunomodulators and biologic therapy [AGA abstract 865]. Gastroenterology. 2012;142:S-149.
- Bae YS, Van Voorhees AS, Hsu S, et al. Review of treatment options for psoriasis in pregnant or lactating women: from the medical board of the National Psoriasis Foundation. J Am Acad Dermatol. 2012;67:459-477.
- Menter A, Strober BE, Kaplan DH, et al. Joint AAD-NPF guidelines of care for the management and treatment of psoriasis with biologics. J Am Acad Dermatol. 2019;80:1029-1072.
- Flood KS, Porter ML, Kimball AB. Use of biologics in pregnancy: limitations stemming from clinical trials and registry experience. J Eur Acad Dermatol Venereol. 2019;33:E276-E277.
- Horn EJ, Chambers CD, Menter A, et al. Pregnancy outcomes in psoriasis: why do we know so little? J Am Acad Dermatol. 2009;61:E5-E8.
- Raychaudhuri SP, Navare T, Gross J, et al. Clinical course of psoriasis during pregnancy. Int J Dermatol. 2003;42:518-520.
- Boyd AS, Morris LF, Phillips CM, et al. Psoriasis and pregnancy: hormone and immune system interaction. Int J Dermatol. 1996;35:169-172.
- Murase JE, Chan KK, Garite TJ, et al. Hormonal effect on psoriasis in pregnancy and post partum. Arch Dermatol. 2005;14:601-606.
- Götestam Skorpen C, Hoeltzenbein M, Tincani A, et al. The EULAR points to consider for use of antirheumatic drugs before pregnancy, and during pregnancy and lactation. Ann Rheum Dis. 2016;75:795-810.
- Nguyen GC, Seow CH, Maxwell C, et al. The Toronto consensus statements for the management of inflammatory bowel disease in pregnancy. Gastroenterology. 2016;150:734-757.
- Wise J. Rheumatic diseases should be actively treated in pregnancy, new guidelines say. BMJ. 2016;532:i312.
- Puig L, Carrascosa JM, Carretero G, et al. Spanish evidence-based guidelines on the treatment of psoriasis with biologic agents, 2013. part 1: on efficacy and choice of treatment. Actas Dermosifiliogr. 2013;104:694-709.
- Girolomoni G, Altomore G, Ayala F, et al. Differential management of mild-to-severe psoriasis with biologic drugs: an Italian Delphi consensus expert panel. J Dermatolog Treat. 2015;26:128-133.
- Yeung J, Gooderham MJ, Grewal P, et al. Management of plaque psoriasis with biologic therapies in women of child-bearing potential consensus paper. J Cutan Med Surg. 2020;24:3S-14S.
- Louthrenoo W, Kasitanon N, Kathamort W, et al. 2016 updated Thai Rheumatism Association Recommendations for the use of biologic and targeted synthetic disease-modifying anti-rheumatic drugs in patients with rheumatoid arthritis. Int J Rheum Dis. 2017;20:1166-1184.
- Flint J, Panchal S, Hurrell A, et al. BSR and BHPR guideline on prescribing drugs in pregnancy and breastfeeding—part I: standard and biologic disease modifying anti-rheumatic drugs and corticosteroids. Rheumatology (Oxford). 2016;55:1693-1697.
- van der Woude CJ, Ardizzone S, Bengtson MB, et al. The second European evidenced-based consensus on reproduction and pregnancy in inflammatory bowel disease. J Crohns Colitis. 2015;9:107-124.
- Orlando A, Armuzz A, Papi C, et al. The Italian Society of Gastroenterology (SIGE) and the Italian Group for the study of Inflammatory Bowel Disease (IG-IBD) Clinical Practice Guidelines: the use of tumor necrosis factor-alpha antagonist therapy in inflammatory bowel disease. Dig Liver Dis. 2011;43:1-20.
- Puchner A, Grochenig HP, Sautner J, et al. Immunosuppressives and biologics during pregnancy and lactation. Wien Klin Wochenschr. 2019;131:29-44.
- ACOG Committee opinion no. 776: immune modulating therapies in pregnancy and lactation. Obstet Gynecol. 2019;133:E287-E297.
- Lamb CA, Kennedy NA, Raine T, et al. British Society of Gastroenterology consensus guidelines on the management of inflammatory bowel disease in adults. Gut. 2019;68:S1-S106.
- Goëb V, Ardizzone M, Arnard L, et al. Recommendations for using TNF-α antagonists and French Clinical Practice Guidelines endorsed by the French National Authority for Health. Joint Bone Spine. 2013;80:574-581.
- Rademaker M, Agnew K, Andrews M, et al. Psoriasis in those planning a family, pregnant or breast-feeding. the Australasian Psoriasis Collaboration. Australas J Dermatol. 2018;59:86-100.
- Mahadevan U, Robinson C, Bernasko N, et al. Inflammatory bowel disease in pregnancy clinical care pathway: a report from the American Gastroenterological Association IBD Parenthood Project Working Group. Gastroenterology. 2019;156:1508-1524.
- Mahadevan U, Cucchiara S, Hyam JS, et al. The London position statement of the World Congress of Gastroenterology on biological therapy for IBD with the European Crohn’s and Colitis Organisation: pregnancy and pediatrics. Am J Gastroenterol. 2011;106:214-223.
- Porter ML, Lockwood SJ, Kimball AB. Update on biologic safety for patients with psoriasis during pregnancy. Int J Womens Dermatol. 2017;3:21-25.
- Bandoli G, Johnson DL, Jones KL, et al. Potentially modifiable risk factors for adverse pregnancy outcomes in women with psoriasis. Br J Dermatol. 2010;163:334-339.
- Lima XT, Janakiraman V, Hughes MD, et al. The impact of psoriasis on pregnancy outcomes. J Invest Dermatol. 2012;132:85-91.
- Bröms G, Haerskjold A, Granath F, et al. Effect of maternal psoriasis on pregnancy and birth outcomes: a population-based cohort study from Denmark and Sweden. Acta Derm Venereol. 2018;98:728-734.
- Bobotsis R, Gulliver WP, Monaghan K, et al. Psoriasis and adverse pregnancy outcomes: a systematic review of observational studies. Br J Dermatol. 2016;175:464-472.
- Blattner CM, Danesh M, Safaee M, et al. Understanding the new FDA pregnancy and lactation labeling rules. Int J Womens Dermatol. 2016;2:5-7.
- Pernia S, DeMaagd G. The new pregnancy and lactation labeling rule. P T. 2016;4:713-715.
- Chi C-C, Wang S-H, Wojnarowska F, et al. Safety of topical corticosteroids in pregnancy. Cochrane Database Syst Rev. 2015:CD007346.
- Chi CC, Wang SH, Kirtschig G. Safety of topical corticosteroids in pregnancy. JAMA Dermatol. 2016;152:934-935.
- Dovonex (calcipotriene) Cream, 0.005% [package insert]. Dublin, Ireland: Leo Laboratories, Ltd; March 2015.
- Franssen ME, van der Wilt GJ, de Jong PC, et al. A retrospective study of the teratogenicity of dermatological coal tar products. Acta Derm Venereol. 1999;79:390-391.
- Garbis H, Elefant E, Bertolotti E, et al. Pregnancy outcome after periconceptional and first-trimester exposure to methoxsalen photochemotherapy. Arch Dermatol. 1995;131:492-493.
- Horizon Pharma USA. RAYOS (prednisone). https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/202020s000lbl.pdf https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/202020s000lbl.pdf.
- Park-Wyllie L, Mazzotta P, Pastuszak A, et al. Birth defects after maternal exposure to corticosteroids: prospective cohort study and meta-analysis of epidemiological studies. Teratology. 2000;62:385-392.
- Crowther CA, Doyle LW, Haslam RR, et al. Outcomes at 2 years of age after repeat doses of antenatal corticosteroids. N Engl J Med. 2007;357:1179-1189.
- Palmsten K, Rolland M, Herbert MF, et al. Patterns of prednisone use during pregnancy in women with rheumatoid arthritis: daily and cumulative dose. Pharmacoepidemiol Drug Saf. 2018;27:430-438.
- Groth K, Brännström M, Mölne J, et al. Cyclosporine A exposure during pregnancy in mice: effects on reproductive performance in mothers and offspring. Hum Reprod. 2010;25:697-704.
- Bar Oz B, Hackman R, Einarson T, et al. Pregnancy outcome after cyclosporine therapy during pregnancy: a meta-analysis. Transplantation. 2001;71:1051-1055.
- Paziana K, Del Monaco M, Cardonick E, et al. Ciclosporin use during pregnancy. Drug Saf. 2013;36:279-294.
- Lamarque V, Leleu MF, Monka C, et al. Analysis of 629 pregnancy outcomes in transplant recipients treated with Sandimmun. Transplant Proc. 1997;29:2480.
- Otezla (apremilast) tablets, for oral use [package insert]. Summit, NJ: Celgene Corporation; June 2017. http://www.accessdata.fda.gov/drugsatfda_docs/label/2017/205437s006lbl.pdf. Accessed July 8, 2020.
- Kurizky PS, de Castro Ferreira C, Nogueira LSC, et al. Treatment of psoriasis and psoriatic arthritis during pregnancy and breastfeeding. An Bras Dermatol. 2015;90:367-375.
- Murase JE, Heller MM, Butler DC. Safety of dermatologic medications in pregnancy and lactation. J Am Acad Dermatol. 2014;70:401.e1-401.e4.
- El-Saie LT, Rabie AR, Kamel MI, et al. Effect of narrowband ultraviolet B phototherapy on serum folic acid levels in patients with psoriasis. Lasers Med Sci. 2011;26:481-485.
- Murase JE, Koo JY, Berger TG. Narrowband ultraviolet B phototherapy influences serum folate levels in patients with vitiligo. J Am Acad Dermatol. 2010;62:710-711.
- Soriatane (acitretin) capsules [package insert]. Morrisville, NC: Stiefel Laboratories, Inc; April 2011. http://www.accessdata.fda.gov/drugsatfda_docs/label/2011/019821s018mg.pdf. Accessed July 8, 2020.
- Kaushik SB, Lebwohl MG. Psoriasis: which therapy for which patient: focus on special populations and chronic infections. J Am Acad Dermatol. 2019;80:43-53.
- Help us better understand the effects of Otezla in pregnancy. MotherToBaby website. https://mothertobaby.org/ongoing-study/otezla/. Accessed July 8, 2020.
- Bangsgaard N, Rørbye C, Skov L. Treating psoriasis during pregnancy: safety and efficacy of treatments. Am J Clin Dermatol. 2015;16:389-398.
- Tyler K. Dermatologic therapy in pregnancy. Clin Obstet Gynecol. 2015;58:112-118.
- Luu M, Benzenine E, Doret M, et al. Continuous anti–TNF-α use throughout pregnancy: possible complications for the mother but not for the fetus. a retrospective cohort on the French National Health Insurance Database (EVASION). Am J Gastroenterol. 2018;113:1669-1677.
- Bröms G, Granath F, Ekbom A, et al. Low risk of birth defects for infants whose mothers are treated with anti-tumor necrosis factor agents during pregnancy. Clin Gastroenterol Hepatol. 2016;14:234-241.
- Mirdamadi K, Salinas T, Vali R, et al. Meta-analysis of pregnancy outcomes after exposure to TNF-α inhibitors during pregnancy for the treatment of arthritic diseases. J Popul Ther Clin Pharmacol. 2018;25:E53-E56.
- Shihab Z, Yeomans ND, De Cruz P. Anti-tumour necrosis factor α therapies and inflammatory bowel disease pregnancy outcomes: a meta-analysis. J Crohns Colitis. 2016;10:979-988.
- Bröms G, Kieler H, Ekbom A, et al. Anti-TNF treatment during pregnancy and birth outcomes: a population-based study from Denmark, Finland, and Sweden. Pharmacoepidemiol Drug Saf. 2020;29:316-327.
- Diav-Citrin O, Otcheretianski-Volodarsky A, Shechtman S, et al. Pregnancy outcome following gestational exposure to TNF-alpha-inhibitors: a prospective, comparative, observational study. Reprod Toxicol. 2014;43:78-84.
- FDA determinations for pregnant and nursing women. National Psoriasis Foundation website. http://www.psoriasis.org/pregnancy/fda-determinations. Accessed July 8, 2020.
- Østensen M. Safety issues of biologics in pregnant patients with rheumatic diseases. Ann N Y Acad Sci. 2014;1317:32-38.
- Chambers CD, Johnson DL, Luo Y, et al. Pregnancy outcome in women treated with adalimumab for the treatment of rheumatoid arthritis: the OTIS Autoimmune Diseases in Pregnancy Project. Arthritis Rheum. 2012;64:2466.
- Clowse ME, Wolf DC, Forger F, et al. Pregnancy outcomes after exposure to certolizumab pegol: updated results from a pharmacovigilance safety database. Arthritis Rheumatol. 2018;70:1399-1407.
- Carman WJ, Accortt NA, Anthony MS, et al. Pregnancy and infant outcomes including major congenital malformations among women with chronic inflammatory arthritis or psoriasis, with and without etanercept use. Pharmacoepidemiol Drug Saf. 2017;26:1109-1118.
- Janssen. SIMPONI (golilumab). https://www.accessdata.fda.gov/drugsatfda_docs/label/2011/125289s0064lbl.pdf.
- Yurkon K, Guo CY, Harrison D, et al. Pregnancy outcomes in women with dermatologic conditions exposed to infliximab. J Am Acad Dermatol. 2014;70:AB179.
- Watson N, Wu K, Farr P, et al. Ustekinumab exposure during conception and pregnancy in patients with chronic plaque psoriasis: a case series of 10 pregnancies. Br J Dermatol. 2019;180:195-196.
- Naureckas S, Slater J, Gearhart N, et al. Pregnancy outcomes in women with psoriasis and psoriatic arthritis exposed to ustekinumab. J Am Acad Dermatol. 2016;74:AB264.
- Haycraft K, DiRuggiero D, Rozzo SJ, et al. Outcomes of pregnancies from tildrakizumab phases I to III clinical development program. J Clin Aesthet Dermatol. 2019;12:S27-S28.
- Tremfya (guselkumab) injection, for subcutaneous use [package insert]. Horsham, PA: Janssen Biotech, Inc; July 2017. http://www.accessdata.fda.gov/drugsatfda_docs/label/2017/761061s000lbl.pdf. Accessed Juy 8, 2020.
- Skyrizi (risankizumab-rzaa) injection, for subcutaneous use [package insert]. Northi Chicago, IL; April 2019. http://www.accessdata.fda.gov/drugsatfda_docs/label/2019/761105s000lbl.pdf. Accessed July 8, 2020.
- Siliq (brodalumab) injection, for subcutaneous use [package insert]. Bridgewater, NJ: Valeant Pharmaceuticals North America LLC; February 2017. http://www.accessdata.fda.gov/drugsatfda_docs/label/2017/761032lbl.pdf. Accessed July 8, 2020.
- Feldman S, Pangallo B, Xu W, et al. Ixekizumab and pregnancy outcome. J Am Acad Dermatol. 2017;76:AB419.
- Clarke DO, Hilbish KG, Waters DG, et al. Assessment of ixekizumab, an interleukin-17A monoclonal antibody, for potential effects on reproduction and development, including immune system function, in cynomolgus monkeys. Reprod Toxicol. 2015;58:160-173.
- Warren RB, Reich K, Langley RG, et al. Secukinumab in pregnancy: outcomes in psoriasis, psoriatic arthritis and ankylosing spondylitis from the global safety database. Br J Dermatol. 2018;179:1205-1207.
- Nardin C, Colas M, Curie V, et al. Pregnancy after tubal sterilization in a woman treated with biologics for severe psoriasis. Dermatol Ther (Heidelb). 2018;8:323-326.
- Xeljanz (tofacitinib) tablets for oral administration [package insert]. New York, NY: Pfizer; November 2012. http://www.accessdata.fda.gov/drugsatfda_docs/label/2012/203214s000lbl.pdf. Accessed July 8, 2020.
- Pfizer. Xeljanz (tofacitinib). https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/203214s018lbl.pdf.
- Mahadevan U, Dubinsky M, Su C, et al. Outcomes of pregnancies with maternal/paternal exposure in the tofacitinib safety databases for ulcerative colitis. Inflamm Bowel Dis. 2018;24:2494-2500.
- Clowse ME, Feldman SR, Isaacs JD, et al. Pregnancy outcomes in the tofacitinib safety databases for rheumatoid arthritis and psoriasis. Drug Saf. 2016;39:755-762.
- Malek A, Sager R, Kuhn P, et al. Evolution of maternofetal transport of immunoglobulins during human pregnancy. Am J Reprod Immunol. 1996;36:248-255.
- Kimball AB, Crow JA, Ridley K, et al. Pregnancy outcomes in women with moderate to severe psoriasis: the PSOLAR experience. J Am Acad Dermatol. 2014;70(suppl 1):AB179.
- Carter JD, Valeriano J, Vasey FB. Tumor necrosis factor-alpha inhibition and VATER association: a causal relationship. J Rheumatol. 2006;33:1014-1017.
- Carter JD, Ladhani A, Ricca LR, et al. A safety assessment of tumor necrosis factor antagonists during pregnancy: a review of the Food and Drug Administration database. J Rheumatol. 2009;36:635-641.
- Koren G, Inoue M. Do tumor necrosis factor inhibitors cause malformations in humans? J Rheumatol. 2009;36:465-466.
- Johansen C, Jimenez-Solem E, Haerskjold A, et al. The use and safety of TNF inhibitors during pregnancy in women with psoriasis: a review. Int J Mol Sci. 2018;19:E1349.
- Mariette X, Forger F, Abraham B, et al. Lack of placental transfer of certolizumab pegol during pregnancy: results from CRIB, a prospective, postmarketing, pharmacokinetic study. Ann Rheum Dis. 2018;77:228-233.
- Mahadevan U, Wolf DC, Dubinsky M, et al. Placental transfer of anti-tumor necrosis factor agents in pregnant patients with inflammatory bowel disease. Clin Gastroenterol Hepatol. 2013;11:286-292.
- Cheent K, Nolan J, Sharig S, et al. Case report: fatal case of disseminated BCG infection in an infant born to a mother taking infliximab for Crohn’s disease. J Crohns Colitis. 2010;4:603-605.
- Julsgaard M, Christensen LA, Gibson PR, et al. Concentrations of adalimumab and infliximab in mothers and newborns, and effects on infection. Gastroenterology. 2016;151:110-119.
- Mahadevan U, Martin C, Kane SV, et al. Do infant serum levels of biologic agents at birth correlate with risk of adverse outcomes? results from the PIANO registry. Gastroenterology. 2016;150:S91-S92.
- Mahadevan U, Martin CF, Sandler RS, et al. PIANO: a 1000 patient prospective registry of pregnancy outcomes in women with IBD exposed to immunomodulators and biologic therapy [AGA abstract 865]. Gastroenterology. 2012;142:S-149.
Practice Points
- Robust safety data often are lacking for the use of topical and systemic agents to treat psoriasis in pregnancy.
- Professional society guidelines on the use of systemic agents in pregnancy vary among dermatology, gastroenterology, and rheumatology organizations.