User login
Inherited mutations shed light on atopic dermatitis pathway
Inherited mutations in a single gene may contribute to a severe form of atopic dermatitis (AD), a study of eight patients showed.
Investigators from the National Institute of Allergy and Infectious Diseases (NIAID) and elsewhere identified eight individuals with severe AD from four unrelated families. All of the patients had a mutation in the CARD11 gene, which is part of the nuclear factor–kappa B (NF-kB) pathway.
When the mutated genes were inserted into T cells, the researchers found that the mutated copy of the gene interfered with the normal copy, preventing the activation of NF-kB and mTORC1 (mammalian target of rapamycin complex 1) – effects that may contribute to the severity of AD in these patients.
The results could have broad-ranging clinical implications, Dr. Milner said in an interview. CARD11 has been shown to be associated with AD in previous genomewide association studies. “It may not be the case that this is just found in a few rare families. This could potentially be a gene or pathway that could explain a lot of atopic dermatitis,” he noted.
The study results also point to a potential therapy for AD. The pathway can lead to a deficiency in glutamine uptake into cells, and the study suggests that glutamine supplementation could potentially restore some cells to normal functioning.
Dr. Milner also pointed out that glutamine deficiency could be an indirect consequence of the disease. “Kids with bad allergic disease are usually on a poor diet because they are avoiding foods. They may not be getting enough protein intake,” he said.
In fact, a prevention trial in premature infants sought to determine if glutamine supplementation could reduce infections. The primary endpoint failed, but researchers noted a reduction in AD, according to Dr. Milner. “That’s pretty amazing, given what we just found.”
This study is among recent studies that have highlighted potential targets for treatment of AD, including one reporting that tumor necrosis factor–like weak inducer of apoptosis, a protein, may be involved in both AD and psoriasis (Nat Commun. 2017 May 22;8:15395.).
Research identifying novel pathways involved in AD led to the development of dupilumab, which targets interleukin-4 and interleukin-13 and was recently approved by the Food and Drug Administration for moderate to severe AD. It is the first targeted biologic therapy to become available for AD. “I can’t underscore the importance of dupilumab enough,” Dr. Milner commented.
He and the other authors had no related disclosures.
Inherited mutations in a single gene may contribute to a severe form of atopic dermatitis (AD), a study of eight patients showed.
Investigators from the National Institute of Allergy and Infectious Diseases (NIAID) and elsewhere identified eight individuals with severe AD from four unrelated families. All of the patients had a mutation in the CARD11 gene, which is part of the nuclear factor–kappa B (NF-kB) pathway.
When the mutated genes were inserted into T cells, the researchers found that the mutated copy of the gene interfered with the normal copy, preventing the activation of NF-kB and mTORC1 (mammalian target of rapamycin complex 1) – effects that may contribute to the severity of AD in these patients.
The results could have broad-ranging clinical implications, Dr. Milner said in an interview. CARD11 has been shown to be associated with AD in previous genomewide association studies. “It may not be the case that this is just found in a few rare families. This could potentially be a gene or pathway that could explain a lot of atopic dermatitis,” he noted.
The study results also point to a potential therapy for AD. The pathway can lead to a deficiency in glutamine uptake into cells, and the study suggests that glutamine supplementation could potentially restore some cells to normal functioning.
Dr. Milner also pointed out that glutamine deficiency could be an indirect consequence of the disease. “Kids with bad allergic disease are usually on a poor diet because they are avoiding foods. They may not be getting enough protein intake,” he said.
In fact, a prevention trial in premature infants sought to determine if glutamine supplementation could reduce infections. The primary endpoint failed, but researchers noted a reduction in AD, according to Dr. Milner. “That’s pretty amazing, given what we just found.”
This study is among recent studies that have highlighted potential targets for treatment of AD, including one reporting that tumor necrosis factor–like weak inducer of apoptosis, a protein, may be involved in both AD and psoriasis (Nat Commun. 2017 May 22;8:15395.).
Research identifying novel pathways involved in AD led to the development of dupilumab, which targets interleukin-4 and interleukin-13 and was recently approved by the Food and Drug Administration for moderate to severe AD. It is the first targeted biologic therapy to become available for AD. “I can’t underscore the importance of dupilumab enough,” Dr. Milner commented.
He and the other authors had no related disclosures.
Inherited mutations in a single gene may contribute to a severe form of atopic dermatitis (AD), a study of eight patients showed.
Investigators from the National Institute of Allergy and Infectious Diseases (NIAID) and elsewhere identified eight individuals with severe AD from four unrelated families. All of the patients had a mutation in the CARD11 gene, which is part of the nuclear factor–kappa B (NF-kB) pathway.
When the mutated genes were inserted into T cells, the researchers found that the mutated copy of the gene interfered with the normal copy, preventing the activation of NF-kB and mTORC1 (mammalian target of rapamycin complex 1) – effects that may contribute to the severity of AD in these patients.
The results could have broad-ranging clinical implications, Dr. Milner said in an interview. CARD11 has been shown to be associated with AD in previous genomewide association studies. “It may not be the case that this is just found in a few rare families. This could potentially be a gene or pathway that could explain a lot of atopic dermatitis,” he noted.
The study results also point to a potential therapy for AD. The pathway can lead to a deficiency in glutamine uptake into cells, and the study suggests that glutamine supplementation could potentially restore some cells to normal functioning.
Dr. Milner also pointed out that glutamine deficiency could be an indirect consequence of the disease. “Kids with bad allergic disease are usually on a poor diet because they are avoiding foods. They may not be getting enough protein intake,” he said.
In fact, a prevention trial in premature infants sought to determine if glutamine supplementation could reduce infections. The primary endpoint failed, but researchers noted a reduction in AD, according to Dr. Milner. “That’s pretty amazing, given what we just found.”
This study is among recent studies that have highlighted potential targets for treatment of AD, including one reporting that tumor necrosis factor–like weak inducer of apoptosis, a protein, may be involved in both AD and psoriasis (Nat Commun. 2017 May 22;8:15395.).
Research identifying novel pathways involved in AD led to the development of dupilumab, which targets interleukin-4 and interleukin-13 and was recently approved by the Food and Drug Administration for moderate to severe AD. It is the first targeted biologic therapy to become available for AD. “I can’t underscore the importance of dupilumab enough,” Dr. Milner commented.
He and the other authors had no related disclosures.
FROM NATURE GENETICS
Key clinical point: Mutations in the CARD11 gene may play a role in severe atopic dermatitis.
Major finding: A mutation in one copy of the CARD11 gene appears to interfere with the normal functioning copy.
Data source: A case study of eight patients with severe AD, all from different families.
Disclosures: Dr. Milner reported having no relevant financial disclosures.
TWEAKing inflammation: Studies reflect potential treatment target for psoriasis, atopic dermatitis
An immunomodulatory pathway that has been linked to cancer, kidney disease, and other disease processes is becoming a focus of dermatologic research.
New evidence suggests that TNF-like weak inducer of apoptosis (TWEAK), a member of the tumor necrosis family (TNF) superfamily, may be involved in both atopic dermatitis (AD) and psoriasis (Nat Commun. 2017 May 22;8:15395. doi: 10.1038/ncomms15395). The research showed that mice engineered to have low TWEAK levels had less severe disease when both AD and psoriasis were induced.
The TWEAK receptor, Fn14, was upregulated in keratinocytes and dermal fibroblasts in mouse disease models of AD and psoriasis, and TWEAK induced production of a range of cytokines associated with both AD and psoriasis. Subcutaneous injection of recombinant TWEAK led to cutaneous inflammation, as well as histological and molecular signals of the two diseases.
The pathophysiology of both AD and psoriasis is nebulously complex, sharing a similar theme of immune dysregulation, but historically polar opposites based on the different branches of the immune response implicated.
The study is not the only recent work tying TWEAK/Fn14 to dermatologic diseases. Other recent papers have shown evidence of their involvement in chronic cutaneous lupus (J Invest Dermatol. 2015;135[8]:1986-95), UVB irradiation-induced cutaneous lupus (Exp Dermatol. 2016 Dec;25[12]:969-76), and bullous pemphigoid (J Invest Dermatol. 2017 Jul;137[7]:1512-22).
The spate of findings hint that TWEAK/Fn14 could be a novel therapeutic pathway to attack inflammatory disease. Many therapies for autoimmune disease focus on immunosuppressive agents, which are associated with an increased risk of infection. But mice engineered to lack either TWEAK or Fn14 appear normal, and a phase I trial of an anti-TWEAK antibody in patients with rheumatoid arthritis did not reveal any worrisome safety concerns. “It doesn’t seem to have the broad immunosuppressive effects which characterize the therapies we currently use,” said Chaim Putterman, MD, chief of the division of rheumatology and professor of medicine and microbiology & immunology at the Albert Einstein College of Medicine, New York.
Instead, TWEAK seems to be regulating inflammation in target organs. It almost certainly plays a role in healthy functions like wound healing and cell survival, but Dr. Putterman believes there are redundant mechanisms that can pick up the slack, as the healthy knockout mice attest. The evidence suggests that the TWEAK pathway may become overactive in some diseases and, if so, a therapeutic antibody might be able to reset it to a more normal balance. “The utopian vision is that you would block this cytokine and bring its downstream effects back to normal levels, rather than totally abrogating its homeostatic functions,” Dr. Putterman noted.
Because blocking TWEAK has no apparent immunosuppressive effects, it might be a candidate for combination therapy with existing cytotoxic drugs. “If you have a disease like psoriasis where some standard of care medications are immunosuppressive, such as methotrexate, you might not get more risk by adding an antibody targeting TWEAK, as opposed to using immunosuppressives in combination. That, I think, has potential,” he said.
Work remains, however. A proof-of-concept study in lupus nephritis, sponsored by Biogen, failed to show a benefit when an anti-TWEAK antibody was combined with the standard of care.
But the potential impact of this approach holds much promise, and the fact that TWEAK has been linked to multiple diseases should make it a more attractive drug target for drug companies. “Now we have a target, that if you knock it out, or its receptor, you can potentially affect both diseases. This may the start of a whole new direction for biologics to treat inflammatory disease, and cancer as well,” Dr. Friedman said.
Dr. Putterman and Dr. Friedman were among the authors of the 2015 JID study on TWEAK/Fn14 signaling in spontaneous lupus and the Experimental Dermatology study. Dr. Putterman has research funding from Biogen Idec. Dr. Friedman had no related disclosures. The authors of the Nature Communications study were from the La Jolla Institute for Allergy and Immunology, and Biogen.
An immunomodulatory pathway that has been linked to cancer, kidney disease, and other disease processes is becoming a focus of dermatologic research.
New evidence suggests that TNF-like weak inducer of apoptosis (TWEAK), a member of the tumor necrosis family (TNF) superfamily, may be involved in both atopic dermatitis (AD) and psoriasis (Nat Commun. 2017 May 22;8:15395. doi: 10.1038/ncomms15395). The research showed that mice engineered to have low TWEAK levels had less severe disease when both AD and psoriasis were induced.
The TWEAK receptor, Fn14, was upregulated in keratinocytes and dermal fibroblasts in mouse disease models of AD and psoriasis, and TWEAK induced production of a range of cytokines associated with both AD and psoriasis. Subcutaneous injection of recombinant TWEAK led to cutaneous inflammation, as well as histological and molecular signals of the two diseases.
The pathophysiology of both AD and psoriasis is nebulously complex, sharing a similar theme of immune dysregulation, but historically polar opposites based on the different branches of the immune response implicated.
The study is not the only recent work tying TWEAK/Fn14 to dermatologic diseases. Other recent papers have shown evidence of their involvement in chronic cutaneous lupus (J Invest Dermatol. 2015;135[8]:1986-95), UVB irradiation-induced cutaneous lupus (Exp Dermatol. 2016 Dec;25[12]:969-76), and bullous pemphigoid (J Invest Dermatol. 2017 Jul;137[7]:1512-22).
The spate of findings hint that TWEAK/Fn14 could be a novel therapeutic pathway to attack inflammatory disease. Many therapies for autoimmune disease focus on immunosuppressive agents, which are associated with an increased risk of infection. But mice engineered to lack either TWEAK or Fn14 appear normal, and a phase I trial of an anti-TWEAK antibody in patients with rheumatoid arthritis did not reveal any worrisome safety concerns. “It doesn’t seem to have the broad immunosuppressive effects which characterize the therapies we currently use,” said Chaim Putterman, MD, chief of the division of rheumatology and professor of medicine and microbiology & immunology at the Albert Einstein College of Medicine, New York.
Instead, TWEAK seems to be regulating inflammation in target organs. It almost certainly plays a role in healthy functions like wound healing and cell survival, but Dr. Putterman believes there are redundant mechanisms that can pick up the slack, as the healthy knockout mice attest. The evidence suggests that the TWEAK pathway may become overactive in some diseases and, if so, a therapeutic antibody might be able to reset it to a more normal balance. “The utopian vision is that you would block this cytokine and bring its downstream effects back to normal levels, rather than totally abrogating its homeostatic functions,” Dr. Putterman noted.
Because blocking TWEAK has no apparent immunosuppressive effects, it might be a candidate for combination therapy with existing cytotoxic drugs. “If you have a disease like psoriasis where some standard of care medications are immunosuppressive, such as methotrexate, you might not get more risk by adding an antibody targeting TWEAK, as opposed to using immunosuppressives in combination. That, I think, has potential,” he said.
Work remains, however. A proof-of-concept study in lupus nephritis, sponsored by Biogen, failed to show a benefit when an anti-TWEAK antibody was combined with the standard of care.
But the potential impact of this approach holds much promise, and the fact that TWEAK has been linked to multiple diseases should make it a more attractive drug target for drug companies. “Now we have a target, that if you knock it out, or its receptor, you can potentially affect both diseases. This may the start of a whole new direction for biologics to treat inflammatory disease, and cancer as well,” Dr. Friedman said.
Dr. Putterman and Dr. Friedman were among the authors of the 2015 JID study on TWEAK/Fn14 signaling in spontaneous lupus and the Experimental Dermatology study. Dr. Putterman has research funding from Biogen Idec. Dr. Friedman had no related disclosures. The authors of the Nature Communications study were from the La Jolla Institute for Allergy and Immunology, and Biogen.
An immunomodulatory pathway that has been linked to cancer, kidney disease, and other disease processes is becoming a focus of dermatologic research.
New evidence suggests that TNF-like weak inducer of apoptosis (TWEAK), a member of the tumor necrosis family (TNF) superfamily, may be involved in both atopic dermatitis (AD) and psoriasis (Nat Commun. 2017 May 22;8:15395. doi: 10.1038/ncomms15395). The research showed that mice engineered to have low TWEAK levels had less severe disease when both AD and psoriasis were induced.
The TWEAK receptor, Fn14, was upregulated in keratinocytes and dermal fibroblasts in mouse disease models of AD and psoriasis, and TWEAK induced production of a range of cytokines associated with both AD and psoriasis. Subcutaneous injection of recombinant TWEAK led to cutaneous inflammation, as well as histological and molecular signals of the two diseases.
The pathophysiology of both AD and psoriasis is nebulously complex, sharing a similar theme of immune dysregulation, but historically polar opposites based on the different branches of the immune response implicated.
The study is not the only recent work tying TWEAK/Fn14 to dermatologic diseases. Other recent papers have shown evidence of their involvement in chronic cutaneous lupus (J Invest Dermatol. 2015;135[8]:1986-95), UVB irradiation-induced cutaneous lupus (Exp Dermatol. 2016 Dec;25[12]:969-76), and bullous pemphigoid (J Invest Dermatol. 2017 Jul;137[7]:1512-22).
The spate of findings hint that TWEAK/Fn14 could be a novel therapeutic pathway to attack inflammatory disease. Many therapies for autoimmune disease focus on immunosuppressive agents, which are associated with an increased risk of infection. But mice engineered to lack either TWEAK or Fn14 appear normal, and a phase I trial of an anti-TWEAK antibody in patients with rheumatoid arthritis did not reveal any worrisome safety concerns. “It doesn’t seem to have the broad immunosuppressive effects which characterize the therapies we currently use,” said Chaim Putterman, MD, chief of the division of rheumatology and professor of medicine and microbiology & immunology at the Albert Einstein College of Medicine, New York.
Instead, TWEAK seems to be regulating inflammation in target organs. It almost certainly plays a role in healthy functions like wound healing and cell survival, but Dr. Putterman believes there are redundant mechanisms that can pick up the slack, as the healthy knockout mice attest. The evidence suggests that the TWEAK pathway may become overactive in some diseases and, if so, a therapeutic antibody might be able to reset it to a more normal balance. “The utopian vision is that you would block this cytokine and bring its downstream effects back to normal levels, rather than totally abrogating its homeostatic functions,” Dr. Putterman noted.
Because blocking TWEAK has no apparent immunosuppressive effects, it might be a candidate for combination therapy with existing cytotoxic drugs. “If you have a disease like psoriasis where some standard of care medications are immunosuppressive, such as methotrexate, you might not get more risk by adding an antibody targeting TWEAK, as opposed to using immunosuppressives in combination. That, I think, has potential,” he said.
Work remains, however. A proof-of-concept study in lupus nephritis, sponsored by Biogen, failed to show a benefit when an anti-TWEAK antibody was combined with the standard of care.
But the potential impact of this approach holds much promise, and the fact that TWEAK has been linked to multiple diseases should make it a more attractive drug target for drug companies. “Now we have a target, that if you knock it out, or its receptor, you can potentially affect both diseases. This may the start of a whole new direction for biologics to treat inflammatory disease, and cancer as well,” Dr. Friedman said.
Dr. Putterman and Dr. Friedman were among the authors of the 2015 JID study on TWEAK/Fn14 signaling in spontaneous lupus and the Experimental Dermatology study. Dr. Putterman has research funding from Biogen Idec. Dr. Friedman had no related disclosures. The authors of the Nature Communications study were from the La Jolla Institute for Allergy and Immunology, and Biogen.
Patch is early indicator of temperature rise after HSCT
A fever-monitoring patch was well tolerated in hospitalized patients undergoing stem cell transplant or intensive chemotherapy for leukemia, and alerted physicians to the presence of a fever much earlier than did standard temperature-taking procedures, according to findings from a study abstract that was published in conjunction with the annual meeting of the American Society of Clinical Oncology.
The patch can transmit data via Bluetooth to an iPad or smartphone, and it attempts to bring temperature recording in line with other vital signs. “It’s the only vital sign that’s not continuously monitored outside of the ICU,” said John Gannon, CEO of Blue Spark, which markets the TempTraq underarm patch used in the study.
The units are constructed with thin batteries that can be “printed” on to any surface. “Being disposable makes it a very usable work flow device,” said Mr. Gannon.
The researchers tested the TempTraq in 10 patients who had been admitted for stem cell transplant or high dose chemotherapy for leukemia.
In addition to wearing the patch, patients had their temperature measured in standard fashion every 4 hours. The researchers defined a temperature rise as a spike above 100.4° F. The device measured body temperature every 10 minutes (14,342 temperature measurements).
Standard of care measurement identified 23 temperature rise episodes, 21 of which were recorded by the TempTraq patch. The device caught temperature spikes much sooner than did standard of care measures – a median of 140.1 minutes earlier (range, 30-180 minutes).
All 10 patients continued to wear the patch throughout the hospital stay, with 9 of 10 reporting that the patch was comfortable and didn’t produce any skin irritation. Eight patients indicated interest in using the patch again, and 8 said they were completely satisfied with the patch.
The next step is to determine if the patch could be used successfully in an outpatient stem cell transplant setting.
The device could also be integrated directly with hospital central monitoring systems using Blue Spark’s TempTraq Connect, which is a HIPAA-compliant service supported by the Google Healthcare Cloud Platform, though this was not tested in the current study.
The TempTraq could be used to monitor patients for the onset of sepsis and allow faster interventions, according to Mr. Gannon. Also, patients who are susceptible to infections could be sent home with a TempTraq monitor, which would send signals to a central monitoring station.
A fever-monitoring patch was well tolerated in hospitalized patients undergoing stem cell transplant or intensive chemotherapy for leukemia, and alerted physicians to the presence of a fever much earlier than did standard temperature-taking procedures, according to findings from a study abstract that was published in conjunction with the annual meeting of the American Society of Clinical Oncology.
The patch can transmit data via Bluetooth to an iPad or smartphone, and it attempts to bring temperature recording in line with other vital signs. “It’s the only vital sign that’s not continuously monitored outside of the ICU,” said John Gannon, CEO of Blue Spark, which markets the TempTraq underarm patch used in the study.
The units are constructed with thin batteries that can be “printed” on to any surface. “Being disposable makes it a very usable work flow device,” said Mr. Gannon.
The researchers tested the TempTraq in 10 patients who had been admitted for stem cell transplant or high dose chemotherapy for leukemia.
In addition to wearing the patch, patients had their temperature measured in standard fashion every 4 hours. The researchers defined a temperature rise as a spike above 100.4° F. The device measured body temperature every 10 minutes (14,342 temperature measurements).
Standard of care measurement identified 23 temperature rise episodes, 21 of which were recorded by the TempTraq patch. The device caught temperature spikes much sooner than did standard of care measures – a median of 140.1 minutes earlier (range, 30-180 minutes).
All 10 patients continued to wear the patch throughout the hospital stay, with 9 of 10 reporting that the patch was comfortable and didn’t produce any skin irritation. Eight patients indicated interest in using the patch again, and 8 said they were completely satisfied with the patch.
The next step is to determine if the patch could be used successfully in an outpatient stem cell transplant setting.
The device could also be integrated directly with hospital central monitoring systems using Blue Spark’s TempTraq Connect, which is a HIPAA-compliant service supported by the Google Healthcare Cloud Platform, though this was not tested in the current study.
The TempTraq could be used to monitor patients for the onset of sepsis and allow faster interventions, according to Mr. Gannon. Also, patients who are susceptible to infections could be sent home with a TempTraq monitor, which would send signals to a central monitoring station.
A fever-monitoring patch was well tolerated in hospitalized patients undergoing stem cell transplant or intensive chemotherapy for leukemia, and alerted physicians to the presence of a fever much earlier than did standard temperature-taking procedures, according to findings from a study abstract that was published in conjunction with the annual meeting of the American Society of Clinical Oncology.
The patch can transmit data via Bluetooth to an iPad or smartphone, and it attempts to bring temperature recording in line with other vital signs. “It’s the only vital sign that’s not continuously monitored outside of the ICU,” said John Gannon, CEO of Blue Spark, which markets the TempTraq underarm patch used in the study.
The units are constructed with thin batteries that can be “printed” on to any surface. “Being disposable makes it a very usable work flow device,” said Mr. Gannon.
The researchers tested the TempTraq in 10 patients who had been admitted for stem cell transplant or high dose chemotherapy for leukemia.
In addition to wearing the patch, patients had their temperature measured in standard fashion every 4 hours. The researchers defined a temperature rise as a spike above 100.4° F. The device measured body temperature every 10 minutes (14,342 temperature measurements).
Standard of care measurement identified 23 temperature rise episodes, 21 of which were recorded by the TempTraq patch. The device caught temperature spikes much sooner than did standard of care measures – a median of 140.1 minutes earlier (range, 30-180 minutes).
All 10 patients continued to wear the patch throughout the hospital stay, with 9 of 10 reporting that the patch was comfortable and didn’t produce any skin irritation. Eight patients indicated interest in using the patch again, and 8 said they were completely satisfied with the patch.
The next step is to determine if the patch could be used successfully in an outpatient stem cell transplant setting.
The device could also be integrated directly with hospital central monitoring systems using Blue Spark’s TempTraq Connect, which is a HIPAA-compliant service supported by the Google Healthcare Cloud Platform, though this was not tested in the current study.
The TempTraq could be used to monitor patients for the onset of sepsis and allow faster interventions, according to Mr. Gannon. Also, patients who are susceptible to infections could be sent home with a TempTraq monitor, which would send signals to a central monitoring station.
FROM ASCO 2017
Key clinical point: The device has the potential to detect infections as well as sepsis.
Major finding: The device detected fevers a median of 140 minutes sooner than did standard hospital testing.
Data source: Prospective study of 10 patients.
Disclosures: Mr. Gannon is an employee of Blue Spark Technologies, which sponsored the study.
ACOG recommends women start mammography between ages 40 and 50 years
Woman at average risk for breast cancer should be offered their first screening mammogram at age 40, with initiation of screening beginning no later than age 50, according to new recommendations from the American College of Obstetricians and Gynecologists.
ACOG suggested a screening interval of 1-2 years based on patient values and preferences.
Screening should continue until at least age 75 years. Past that age, continuation should be considered in terms of the patient’s overall health and expected lifespan, according to the Practice Bulletin.
That’s a welcome approach, according to Ritu Salani, MD, a gynecologic oncologist at the Ohio State University, Columbus. “I do believe the ACOG guidelines have the most flexibility and are probably the most comprehensive for average-risk patients,” said Dr. Salani, MD, who was not involved in the drafting of the guidelines.
The recommendations are based in part on a review of existing recommendations from the U.S. Preventive Services Task Force, the American Cancer Society, and the National Comprehensive Cancer Network.
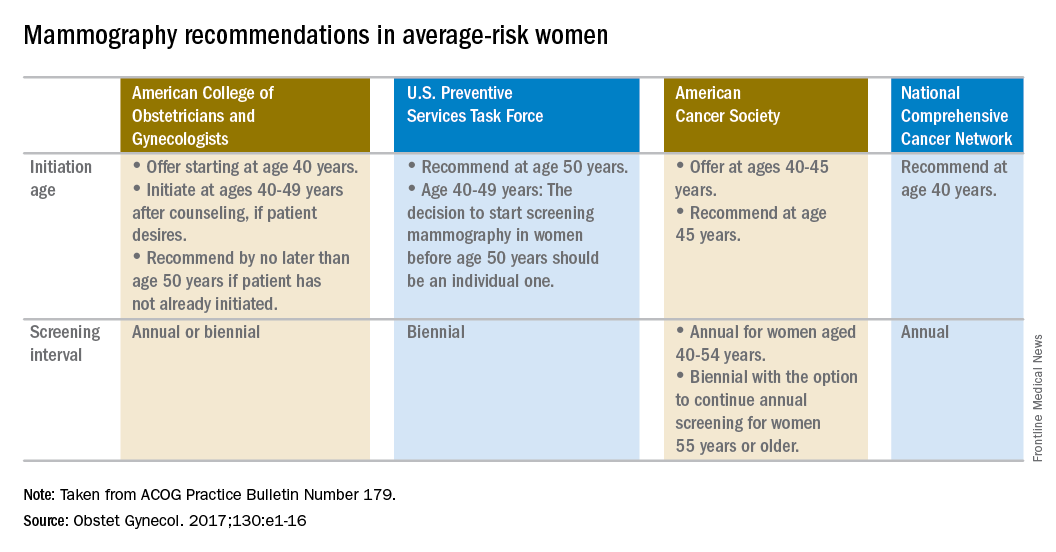
“For patients anxious about developing cancer, maybe every year would be appropriate. When you talk to patients, you have a sense of what would be beneficial for them,” said Dr. Salani, who applauded the flexibility built into the new ACOG guidelines, which says the screening interval can be annual or biennial. “It caters to shared decision-making, and that’s a nice way to frame it.”
The ACOG guidelines recommend against breast self examination because this practice carries the risk of false positives and lacks supporting evidence of benefit. But, ACOG encourages “breast self-awareness,” which means being “attuned to noticing a change or potential problem with her breasts” but without the routine examination.
Women who are asymptomatic and at average risk can be offered clinical breast examination as a screening tool, but clinicians should explain the “uncertainty of additional benefits and the possibility of adverse consequences of clinical breast examination beyond screening mammography.” If performed, ACOG recommends intervals of 1-3 years for women aged 25-39 years and annually for women aged 40 years and over.
The recommendations apply to average-risk women. To help determine risk, the Practice Bulletin also lays out a series of breast cancer risk factors that should be considered, including family history of cancer, known germline mutations, prior biopsy findings, early menarche, late menopause, breast density, and a range of lifestyle and other factors.
Woman at average risk for breast cancer should be offered their first screening mammogram at age 40, with initiation of screening beginning no later than age 50, according to new recommendations from the American College of Obstetricians and Gynecologists.
ACOG suggested a screening interval of 1-2 years based on patient values and preferences.
Screening should continue until at least age 75 years. Past that age, continuation should be considered in terms of the patient’s overall health and expected lifespan, according to the Practice Bulletin.
That’s a welcome approach, according to Ritu Salani, MD, a gynecologic oncologist at the Ohio State University, Columbus. “I do believe the ACOG guidelines have the most flexibility and are probably the most comprehensive for average-risk patients,” said Dr. Salani, MD, who was not involved in the drafting of the guidelines.
The recommendations are based in part on a review of existing recommendations from the U.S. Preventive Services Task Force, the American Cancer Society, and the National Comprehensive Cancer Network.

“For patients anxious about developing cancer, maybe every year would be appropriate. When you talk to patients, you have a sense of what would be beneficial for them,” said Dr. Salani, who applauded the flexibility built into the new ACOG guidelines, which says the screening interval can be annual or biennial. “It caters to shared decision-making, and that’s a nice way to frame it.”
The ACOG guidelines recommend against breast self examination because this practice carries the risk of false positives and lacks supporting evidence of benefit. But, ACOG encourages “breast self-awareness,” which means being “attuned to noticing a change or potential problem with her breasts” but without the routine examination.
Women who are asymptomatic and at average risk can be offered clinical breast examination as a screening tool, but clinicians should explain the “uncertainty of additional benefits and the possibility of adverse consequences of clinical breast examination beyond screening mammography.” If performed, ACOG recommends intervals of 1-3 years for women aged 25-39 years and annually for women aged 40 years and over.
The recommendations apply to average-risk women. To help determine risk, the Practice Bulletin also lays out a series of breast cancer risk factors that should be considered, including family history of cancer, known germline mutations, prior biopsy findings, early menarche, late menopause, breast density, and a range of lifestyle and other factors.
Woman at average risk for breast cancer should be offered their first screening mammogram at age 40, with initiation of screening beginning no later than age 50, according to new recommendations from the American College of Obstetricians and Gynecologists.
ACOG suggested a screening interval of 1-2 years based on patient values and preferences.
Screening should continue until at least age 75 years. Past that age, continuation should be considered in terms of the patient’s overall health and expected lifespan, according to the Practice Bulletin.
That’s a welcome approach, according to Ritu Salani, MD, a gynecologic oncologist at the Ohio State University, Columbus. “I do believe the ACOG guidelines have the most flexibility and are probably the most comprehensive for average-risk patients,” said Dr. Salani, MD, who was not involved in the drafting of the guidelines.
The recommendations are based in part on a review of existing recommendations from the U.S. Preventive Services Task Force, the American Cancer Society, and the National Comprehensive Cancer Network.

“For patients anxious about developing cancer, maybe every year would be appropriate. When you talk to patients, you have a sense of what would be beneficial for them,” said Dr. Salani, who applauded the flexibility built into the new ACOG guidelines, which says the screening interval can be annual or biennial. “It caters to shared decision-making, and that’s a nice way to frame it.”
The ACOG guidelines recommend against breast self examination because this practice carries the risk of false positives and lacks supporting evidence of benefit. But, ACOG encourages “breast self-awareness,” which means being “attuned to noticing a change or potential problem with her breasts” but without the routine examination.
Women who are asymptomatic and at average risk can be offered clinical breast examination as a screening tool, but clinicians should explain the “uncertainty of additional benefits and the possibility of adverse consequences of clinical breast examination beyond screening mammography.” If performed, ACOG recommends intervals of 1-3 years for women aged 25-39 years and annually for women aged 40 years and over.
The recommendations apply to average-risk women. To help determine risk, the Practice Bulletin also lays out a series of breast cancer risk factors that should be considered, including family history of cancer, known germline mutations, prior biopsy findings, early menarche, late menopause, breast density, and a range of lifestyle and other factors.
VIDEO: Social networking offers coping help for endometriosis patients
VANCOUVER – Nearly half of the discussion topics on the social networking site www.myendometriosisteam.com are about pain, but other uncontrolled symptoms are popular topics, including fatigue and depression.
The online social support network includes 30,000 women with endometriosis and offers them a chance to connect with other women with the condition, find a provider, and research treatments. Elise-Marie Menke, director of alliance management at MyHealthTeams, which runs the site, presented data at the World Congress on Endometriosis. Among the findings she presented was how symptoms mapped to a woman’s cycle.
In a video interview, Ms. Menke described how this type of patient-generated data can play a role in the management of disease and serve to highlight unmet needs to physicians.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
VANCOUVER – Nearly half of the discussion topics on the social networking site www.myendometriosisteam.com are about pain, but other uncontrolled symptoms are popular topics, including fatigue and depression.
The online social support network includes 30,000 women with endometriosis and offers them a chance to connect with other women with the condition, find a provider, and research treatments. Elise-Marie Menke, director of alliance management at MyHealthTeams, which runs the site, presented data at the World Congress on Endometriosis. Among the findings she presented was how symptoms mapped to a woman’s cycle.
In a video interview, Ms. Menke described how this type of patient-generated data can play a role in the management of disease and serve to highlight unmet needs to physicians.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
VANCOUVER – Nearly half of the discussion topics on the social networking site www.myendometriosisteam.com are about pain, but other uncontrolled symptoms are popular topics, including fatigue and depression.
The online social support network includes 30,000 women with endometriosis and offers them a chance to connect with other women with the condition, find a provider, and research treatments. Elise-Marie Menke, director of alliance management at MyHealthTeams, which runs the site, presented data at the World Congress on Endometriosis. Among the findings she presented was how symptoms mapped to a woman’s cycle.
In a video interview, Ms. Menke described how this type of patient-generated data can play a role in the management of disease and serve to highlight unmet needs to physicians.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT WCE 2017
Endometriosis after menopause: Weigh the treatment risks
VANCOUVER – Endometriosis, while generally considered a premenopausal condition, can also occur in women following surgical or natural menopause, and can undergo malignant transformation, although this risk is likely very small.
That was the main message from a new meta-analysis presented at the World Congress on Endometriosis. “We wanted to synthesize the case reports out there to show some common factors so physicians can be aware of them,” said Laura Gemmell, a second-year medical student at Case Western Reserve University, Cleveland, who presented the research.
The researchers surveyed the literature for studies in postmenopausal women with a confirmed or clinically suspected history of endometriosis, and who discussed the management of their menopausal symptoms. They included 33 case reports and case series (42 patients, 36 surgical menopause, 4 natural, 2 presumed natural with later oophorectomy), as well as 6 observational studies and clinical trials.
In the case reports, patients were on HT for a mean of 7.8 years, and 17 of 42 women experienced a recurrence of endometriosis. Also, 25 women had a malignant transformation and there was some overlap with the recurrence group.
Among 17 patients with recurrence, 6 had “severe” or “extensive” endometriosis, and 14 had surgical menopause, with a mean of 7.1 years between surgical menopause and presentation. Twelve of 17 received unopposed estrogen. Following surgical excision (16 of 17 cases), 10 had symptom regression without relapses.
When the researchers looked at the 25 cases of malignant transformation, they found that 13 women had endometriosis at more than one site, 22 had undergone surgical menopause, 19 were on unopposed estrogen, and the mean duration of HT was 6.7 years. Seven women presented with vaginal bleeding and nine with masses. Three died from the disease. These three women had severe endometriosis complicating factors, including older age and multiple malignancies.
The analysis also included six observational studies and clinical trials that explored recurrence of endometriosis, and whether HT should be given to women with a history of endometriosis, whether it should be given immediately after surgical menopause, and the most appropriate menopause treatments.
Predictably, the evidence could not be summed up neatly, but Ms. Gemmell emphasized the need to individually weigh the risks and benefits of HT in each patient, with consideration of characteristics such as age, previous disease severity, family history, comorbidities, and body mass index.
She also suggested that patients should be active participants in decision making.
Finally, if the decision is to go forward or continue with HT, she suggested that clinicians consider a combined treatment rather than estrogen-only, though she pointed out the increased risk for breast cancer that this presents.
Tommaso Falcone, MD, chairman of obstetrics and gynecology at the Cleveland Clinic, sounded a note of caution about the use of progestins during the question-and-answer session. “The data are not strong that it actually prevents the development of cancer in the residual disease, if there is any. Even if you take the hypothesis that progestins are going to prevent cancer of residual disease, which is a low-level risk, the main worry that women have is breast cancer, and progestin is strongly associated with breast cancer,” Dr. Falcone said in an interview.
Ms. Gemmell and Dr. Falcone reported having no financial disclosures.
VANCOUVER – Endometriosis, while generally considered a premenopausal condition, can also occur in women following surgical or natural menopause, and can undergo malignant transformation, although this risk is likely very small.
That was the main message from a new meta-analysis presented at the World Congress on Endometriosis. “We wanted to synthesize the case reports out there to show some common factors so physicians can be aware of them,” said Laura Gemmell, a second-year medical student at Case Western Reserve University, Cleveland, who presented the research.
The researchers surveyed the literature for studies in postmenopausal women with a confirmed or clinically suspected history of endometriosis, and who discussed the management of their menopausal symptoms. They included 33 case reports and case series (42 patients, 36 surgical menopause, 4 natural, 2 presumed natural with later oophorectomy), as well as 6 observational studies and clinical trials.
In the case reports, patients were on HT for a mean of 7.8 years, and 17 of 42 women experienced a recurrence of endometriosis. Also, 25 women had a malignant transformation and there was some overlap with the recurrence group.
Among 17 patients with recurrence, 6 had “severe” or “extensive” endometriosis, and 14 had surgical menopause, with a mean of 7.1 years between surgical menopause and presentation. Twelve of 17 received unopposed estrogen. Following surgical excision (16 of 17 cases), 10 had symptom regression without relapses.
When the researchers looked at the 25 cases of malignant transformation, they found that 13 women had endometriosis at more than one site, 22 had undergone surgical menopause, 19 were on unopposed estrogen, and the mean duration of HT was 6.7 years. Seven women presented with vaginal bleeding and nine with masses. Three died from the disease. These three women had severe endometriosis complicating factors, including older age and multiple malignancies.
The analysis also included six observational studies and clinical trials that explored recurrence of endometriosis, and whether HT should be given to women with a history of endometriosis, whether it should be given immediately after surgical menopause, and the most appropriate menopause treatments.
Predictably, the evidence could not be summed up neatly, but Ms. Gemmell emphasized the need to individually weigh the risks and benefits of HT in each patient, with consideration of characteristics such as age, previous disease severity, family history, comorbidities, and body mass index.
She also suggested that patients should be active participants in decision making.
Finally, if the decision is to go forward or continue with HT, she suggested that clinicians consider a combined treatment rather than estrogen-only, though she pointed out the increased risk for breast cancer that this presents.
Tommaso Falcone, MD, chairman of obstetrics and gynecology at the Cleveland Clinic, sounded a note of caution about the use of progestins during the question-and-answer session. “The data are not strong that it actually prevents the development of cancer in the residual disease, if there is any. Even if you take the hypothesis that progestins are going to prevent cancer of residual disease, which is a low-level risk, the main worry that women have is breast cancer, and progestin is strongly associated with breast cancer,” Dr. Falcone said in an interview.
Ms. Gemmell and Dr. Falcone reported having no financial disclosures.
VANCOUVER – Endometriosis, while generally considered a premenopausal condition, can also occur in women following surgical or natural menopause, and can undergo malignant transformation, although this risk is likely very small.
That was the main message from a new meta-analysis presented at the World Congress on Endometriosis. “We wanted to synthesize the case reports out there to show some common factors so physicians can be aware of them,” said Laura Gemmell, a second-year medical student at Case Western Reserve University, Cleveland, who presented the research.
The researchers surveyed the literature for studies in postmenopausal women with a confirmed or clinically suspected history of endometriosis, and who discussed the management of their menopausal symptoms. They included 33 case reports and case series (42 patients, 36 surgical menopause, 4 natural, 2 presumed natural with later oophorectomy), as well as 6 observational studies and clinical trials.
In the case reports, patients were on HT for a mean of 7.8 years, and 17 of 42 women experienced a recurrence of endometriosis. Also, 25 women had a malignant transformation and there was some overlap with the recurrence group.
Among 17 patients with recurrence, 6 had “severe” or “extensive” endometriosis, and 14 had surgical menopause, with a mean of 7.1 years between surgical menopause and presentation. Twelve of 17 received unopposed estrogen. Following surgical excision (16 of 17 cases), 10 had symptom regression without relapses.
When the researchers looked at the 25 cases of malignant transformation, they found that 13 women had endometriosis at more than one site, 22 had undergone surgical menopause, 19 were on unopposed estrogen, and the mean duration of HT was 6.7 years. Seven women presented with vaginal bleeding and nine with masses. Three died from the disease. These three women had severe endometriosis complicating factors, including older age and multiple malignancies.
The analysis also included six observational studies and clinical trials that explored recurrence of endometriosis, and whether HT should be given to women with a history of endometriosis, whether it should be given immediately after surgical menopause, and the most appropriate menopause treatments.
Predictably, the evidence could not be summed up neatly, but Ms. Gemmell emphasized the need to individually weigh the risks and benefits of HT in each patient, with consideration of characteristics such as age, previous disease severity, family history, comorbidities, and body mass index.
She also suggested that patients should be active participants in decision making.
Finally, if the decision is to go forward or continue with HT, she suggested that clinicians consider a combined treatment rather than estrogen-only, though she pointed out the increased risk for breast cancer that this presents.
Tommaso Falcone, MD, chairman of obstetrics and gynecology at the Cleveland Clinic, sounded a note of caution about the use of progestins during the question-and-answer session. “The data are not strong that it actually prevents the development of cancer in the residual disease, if there is any. Even if you take the hypothesis that progestins are going to prevent cancer of residual disease, which is a low-level risk, the main worry that women have is breast cancer, and progestin is strongly associated with breast cancer,” Dr. Falcone said in an interview.
Ms. Gemmell and Dr. Falcone reported having no financial disclosures.
EXPERT ANALYSIS AT WCE 2017
Elastrographic ultrasound could guide adenomyosis treatment
VANCOUVER – Transvaginal elastrographic (TVEG) ultrasound appears to be a better way to diagnose adenomyosis, outperforming transvaginal ultrasound in identifying lesions, according to new findings.
Researchers at Fudan University in Shanghai compared TVEG results in 152 women with adenomyosis, 89 women with fibroids, and 136 healthy controls. None of the women had received hormone therapy in the previous 6 months. Imaging was performed with both TVEG and transvaginal ultrasound, and tissue samples were taken to test for estrogen receptor (ER)-beta, progesterone receptor (PR), epithelial cadherin, and alpha–smooth muscle actin (SMA).
Image analysis showed that TVEG readily distinguished adenomyosis from fibroids or normal uterine tissue. The elastic value, representing stiffness, was highest in adenomyosis patients (3.74 plus or minus 1.01, P less than .001), followed by fibrosis (2.87 plus or minus 0.74; P less than .001), and normal tissue (1.43 plus or minus 0.59).
Elastic values correlated positively to the extent of fibrosis (r = 0.91; P less than .001), and staining levels of alpha-SMA and ER-beta (r = 0.84; P less than .001). Elasticity correlated negatively with epithelial cadherin and PR (r = –0.86; P less than .001).
The researchers concluded that TVEG outperforms transvaginal ultrasound in diagnosing adenomyosis, and that the close correlation between measurements of stiffness and fibrosis and hormone response markers suggests that it could one day help physicians choose between hormone therapy and hysterectomy.
“If we find more elastic values, maybe that means there is more fibrosis in the lesion, and it may be not as sensitive to hormone treatment, so maybe we should move on to hysterectomy,” Ding Ding, MD, PhD, associate professor of gynecology at Fudan University, said at the World Congress on Endometriosis.
But the current research does not provide those answers yet, since the elastic values weren’t linked to a clinical outcome. “We want to verify in the next step, in women who have higher elastic values, whether they are sensitive to progesterone treatment,” Dr. Ding said.
The study was sponsored by the Chinese government. Dr. Ding reported having no financial disclosures.
VANCOUVER – Transvaginal elastrographic (TVEG) ultrasound appears to be a better way to diagnose adenomyosis, outperforming transvaginal ultrasound in identifying lesions, according to new findings.
Researchers at Fudan University in Shanghai compared TVEG results in 152 women with adenomyosis, 89 women with fibroids, and 136 healthy controls. None of the women had received hormone therapy in the previous 6 months. Imaging was performed with both TVEG and transvaginal ultrasound, and tissue samples were taken to test for estrogen receptor (ER)-beta, progesterone receptor (PR), epithelial cadherin, and alpha–smooth muscle actin (SMA).
Image analysis showed that TVEG readily distinguished adenomyosis from fibroids or normal uterine tissue. The elastic value, representing stiffness, was highest in adenomyosis patients (3.74 plus or minus 1.01, P less than .001), followed by fibrosis (2.87 plus or minus 0.74; P less than .001), and normal tissue (1.43 plus or minus 0.59).
Elastic values correlated positively to the extent of fibrosis (r = 0.91; P less than .001), and staining levels of alpha-SMA and ER-beta (r = 0.84; P less than .001). Elasticity correlated negatively with epithelial cadherin and PR (r = –0.86; P less than .001).
The researchers concluded that TVEG outperforms transvaginal ultrasound in diagnosing adenomyosis, and that the close correlation between measurements of stiffness and fibrosis and hormone response markers suggests that it could one day help physicians choose between hormone therapy and hysterectomy.
“If we find more elastic values, maybe that means there is more fibrosis in the lesion, and it may be not as sensitive to hormone treatment, so maybe we should move on to hysterectomy,” Ding Ding, MD, PhD, associate professor of gynecology at Fudan University, said at the World Congress on Endometriosis.
But the current research does not provide those answers yet, since the elastic values weren’t linked to a clinical outcome. “We want to verify in the next step, in women who have higher elastic values, whether they are sensitive to progesterone treatment,” Dr. Ding said.
The study was sponsored by the Chinese government. Dr. Ding reported having no financial disclosures.
VANCOUVER – Transvaginal elastrographic (TVEG) ultrasound appears to be a better way to diagnose adenomyosis, outperforming transvaginal ultrasound in identifying lesions, according to new findings.
Researchers at Fudan University in Shanghai compared TVEG results in 152 women with adenomyosis, 89 women with fibroids, and 136 healthy controls. None of the women had received hormone therapy in the previous 6 months. Imaging was performed with both TVEG and transvaginal ultrasound, and tissue samples were taken to test for estrogen receptor (ER)-beta, progesterone receptor (PR), epithelial cadherin, and alpha–smooth muscle actin (SMA).
Image analysis showed that TVEG readily distinguished adenomyosis from fibroids or normal uterine tissue. The elastic value, representing stiffness, was highest in adenomyosis patients (3.74 plus or minus 1.01, P less than .001), followed by fibrosis (2.87 plus or minus 0.74; P less than .001), and normal tissue (1.43 plus or minus 0.59).
Elastic values correlated positively to the extent of fibrosis (r = 0.91; P less than .001), and staining levels of alpha-SMA and ER-beta (r = 0.84; P less than .001). Elasticity correlated negatively with epithelial cadherin and PR (r = –0.86; P less than .001).
The researchers concluded that TVEG outperforms transvaginal ultrasound in diagnosing adenomyosis, and that the close correlation between measurements of stiffness and fibrosis and hormone response markers suggests that it could one day help physicians choose between hormone therapy and hysterectomy.
“If we find more elastic values, maybe that means there is more fibrosis in the lesion, and it may be not as sensitive to hormone treatment, so maybe we should move on to hysterectomy,” Ding Ding, MD, PhD, associate professor of gynecology at Fudan University, said at the World Congress on Endometriosis.
But the current research does not provide those answers yet, since the elastic values weren’t linked to a clinical outcome. “We want to verify in the next step, in women who have higher elastic values, whether they are sensitive to progesterone treatment,” Dr. Ding said.
The study was sponsored by the Chinese government. Dr. Ding reported having no financial disclosures.
AT WCE 2017
Key clinical point:
Major finding: Elastic values correlated with fibrosis (r = 0.91), alpha-SMA and ER-beta (r = 0.84), and epithelial cadherin and PR (r = –0.86).
Data source: Prospective case-controlled study of 152 women with adenomyosis, 89 with fibroids, and 136 controls.
Disclosures: The study was sponsored by the Chinese government. Dr. Ding reported having no financial disclosures.
Study validates endometriosis fertility index
VANCOUVER – The Endometriosis Fertility Index accurately predicts a woman’s chances to conceive naturally with endometriosis, according to findings from a prospective cohort study.
“We’re quite confident that this is a very good system to give women a reasonable idea of their chances of conceiving naturally versus a recommendation to have IVF,” Aaron Budden, Bmed, Mmed, of the Royal Hospital for Women in Sydney, said at the World Congress on Endometriosis.
The Endometriosis Fertility Index (EFI) was first published in 2010 (Fertil Steril. 2010 Oct;94[5]:1609-15). In the recent study, researchers found that live birth rates after 3 years closely matched the rates calculated by the tool. “This is one of the most useful systems I’ve ever seen, because it takes into account historical events, as well as the time of endometriosis surgery,” Dr. Budden said.
The researchers enrolled 141 consecutive women who had undergone fertility-saving laparoscopic excision of stage III or stage IV endometriosis and attempted to conceive naturally. During follow-up, the researchers contacted the patients to determine live births, and calculated the EFI score based on a woman’s age, duration of infertility, previous pregnancy, American Society for Reproductive Medicine endometriosis classification, and their least adnexal function score. “The least function score is based off the functional capacity of the fallopian tube, fimbriae, and ovary, where the score is calculated by which of these is least functional on both right and left side,” Dr. Budden said.
The researchers included women with stage III and stage IV endometriosis because this group is often referred for in vitro fertilization (IVF). “But we found that in women with an advanced stage of endometriosis, those with EFI scores of 9 and 10 still had quite a significant chance of conceiving at 3 years, compared to women who had EFI scores of 0 to 2,” Dr. Budden said.
More than a third of the women (35%) had stage III endometriosis, and 65% had stage IV. The mean follow-up period was 56 months. Overall, 46% achieved live births.
In women with an EFI score of 9-10, the success rate was 67% at 3 years. Nearly half (48%) of women with scores of 7-8 were successful at 3 years, as were 38% with scores of 5-6, and 17% with scores of 3-4. Among women with scores of 0-2, there were no live births at 3 years.
About 58% of the women in the study had undergone previous laparoscopic surgery. The researchers found that live births were associated with complete resection of disease (hazard ratio, 2.33; P = .036) and no previous laparoscopy (HR, 2.36; P less than .001).
“One of the things we have to say is that your best chance at conceiving is if you excise all the endometriosis and you do it the first time, rather than needing a second surgery,” Dr. Budden said.
The tool can also be used to help identify patients most likely to benefit from assisted reproduction. Women with EFI scores of 0-2 had a 0% chance of conceiving naturally when followed out to 5 years, but a 39% chance with IVF. “That’s a group that you would thoroughly recommend going to IVF rather than trying to conceive naturally,” Dr. Budden said in an interview.
The study was funded by a variety of non-industry sources. Dr. Budden reported having no financial disclosures.
VANCOUVER – The Endometriosis Fertility Index accurately predicts a woman’s chances to conceive naturally with endometriosis, according to findings from a prospective cohort study.
“We’re quite confident that this is a very good system to give women a reasonable idea of their chances of conceiving naturally versus a recommendation to have IVF,” Aaron Budden, Bmed, Mmed, of the Royal Hospital for Women in Sydney, said at the World Congress on Endometriosis.
The Endometriosis Fertility Index (EFI) was first published in 2010 (Fertil Steril. 2010 Oct;94[5]:1609-15). In the recent study, researchers found that live birth rates after 3 years closely matched the rates calculated by the tool. “This is one of the most useful systems I’ve ever seen, because it takes into account historical events, as well as the time of endometriosis surgery,” Dr. Budden said.
The researchers enrolled 141 consecutive women who had undergone fertility-saving laparoscopic excision of stage III or stage IV endometriosis and attempted to conceive naturally. During follow-up, the researchers contacted the patients to determine live births, and calculated the EFI score based on a woman’s age, duration of infertility, previous pregnancy, American Society for Reproductive Medicine endometriosis classification, and their least adnexal function score. “The least function score is based off the functional capacity of the fallopian tube, fimbriae, and ovary, where the score is calculated by which of these is least functional on both right and left side,” Dr. Budden said.
The researchers included women with stage III and stage IV endometriosis because this group is often referred for in vitro fertilization (IVF). “But we found that in women with an advanced stage of endometriosis, those with EFI scores of 9 and 10 still had quite a significant chance of conceiving at 3 years, compared to women who had EFI scores of 0 to 2,” Dr. Budden said.
More than a third of the women (35%) had stage III endometriosis, and 65% had stage IV. The mean follow-up period was 56 months. Overall, 46% achieved live births.
In women with an EFI score of 9-10, the success rate was 67% at 3 years. Nearly half (48%) of women with scores of 7-8 were successful at 3 years, as were 38% with scores of 5-6, and 17% with scores of 3-4. Among women with scores of 0-2, there were no live births at 3 years.
About 58% of the women in the study had undergone previous laparoscopic surgery. The researchers found that live births were associated with complete resection of disease (hazard ratio, 2.33; P = .036) and no previous laparoscopy (HR, 2.36; P less than .001).
“One of the things we have to say is that your best chance at conceiving is if you excise all the endometriosis and you do it the first time, rather than needing a second surgery,” Dr. Budden said.
The tool can also be used to help identify patients most likely to benefit from assisted reproduction. Women with EFI scores of 0-2 had a 0% chance of conceiving naturally when followed out to 5 years, but a 39% chance with IVF. “That’s a group that you would thoroughly recommend going to IVF rather than trying to conceive naturally,” Dr. Budden said in an interview.
The study was funded by a variety of non-industry sources. Dr. Budden reported having no financial disclosures.
VANCOUVER – The Endometriosis Fertility Index accurately predicts a woman’s chances to conceive naturally with endometriosis, according to findings from a prospective cohort study.
“We’re quite confident that this is a very good system to give women a reasonable idea of their chances of conceiving naturally versus a recommendation to have IVF,” Aaron Budden, Bmed, Mmed, of the Royal Hospital for Women in Sydney, said at the World Congress on Endometriosis.
The Endometriosis Fertility Index (EFI) was first published in 2010 (Fertil Steril. 2010 Oct;94[5]:1609-15). In the recent study, researchers found that live birth rates after 3 years closely matched the rates calculated by the tool. “This is one of the most useful systems I’ve ever seen, because it takes into account historical events, as well as the time of endometriosis surgery,” Dr. Budden said.
The researchers enrolled 141 consecutive women who had undergone fertility-saving laparoscopic excision of stage III or stage IV endometriosis and attempted to conceive naturally. During follow-up, the researchers contacted the patients to determine live births, and calculated the EFI score based on a woman’s age, duration of infertility, previous pregnancy, American Society for Reproductive Medicine endometriosis classification, and their least adnexal function score. “The least function score is based off the functional capacity of the fallopian tube, fimbriae, and ovary, where the score is calculated by which of these is least functional on both right and left side,” Dr. Budden said.
The researchers included women with stage III and stage IV endometriosis because this group is often referred for in vitro fertilization (IVF). “But we found that in women with an advanced stage of endometriosis, those with EFI scores of 9 and 10 still had quite a significant chance of conceiving at 3 years, compared to women who had EFI scores of 0 to 2,” Dr. Budden said.
More than a third of the women (35%) had stage III endometriosis, and 65% had stage IV. The mean follow-up period was 56 months. Overall, 46% achieved live births.
In women with an EFI score of 9-10, the success rate was 67% at 3 years. Nearly half (48%) of women with scores of 7-8 were successful at 3 years, as were 38% with scores of 5-6, and 17% with scores of 3-4. Among women with scores of 0-2, there were no live births at 3 years.
About 58% of the women in the study had undergone previous laparoscopic surgery. The researchers found that live births were associated with complete resection of disease (hazard ratio, 2.33; P = .036) and no previous laparoscopy (HR, 2.36; P less than .001).
“One of the things we have to say is that your best chance at conceiving is if you excise all the endometriosis and you do it the first time, rather than needing a second surgery,” Dr. Budden said.
The tool can also be used to help identify patients most likely to benefit from assisted reproduction. Women with EFI scores of 0-2 had a 0% chance of conceiving naturally when followed out to 5 years, but a 39% chance with IVF. “That’s a group that you would thoroughly recommend going to IVF rather than trying to conceive naturally,” Dr. Budden said in an interview.
The study was funded by a variety of non-industry sources. Dr. Budden reported having no financial disclosures.
AT WCE 2017
Key clinical point:
Major finding: Women who scored 9-10 on the EFI had a 67% live birth rate at 3 years without IVF.
Data source: Prospective analysis of 141 women in Australia.
Disclosures: The study was funded by a variety of non-industry sources. Dr. Budden reported having no financial disclosures.
Endometriosis detection by microRNA possible in early stages
VANCOUVER – A panel of 11 microRNA biomarkers was effective in predicting endometriosis in a small validation study conducted at a Belgium hospital, showing the progress researchers are making toward a noninvasive test.
The research team at Leuven University Hospital developed the model using a population of 120 women, 82 of whom had endometriosis. After extensive analysis, they identified 11 microRNAs, which varied significantly between endometriosis patients and healthy controls and could be used as part of a panel to detect endometriosis. The panel performed well in their validation sample of 60 women with endometriosis and 30 controls, with a sensitivity of 92% and a specificity of 96%.
That validation is good news, but it is still far from being clinically useful. First, the model needs to be tested further in their own predominantly Caucasian population. Then, it needs to be validated by outside researchers, and the model will almost certainly have to be altered to account for different genetic backgrounds, lifestyle habits, and other factors, Arne Vanhie, MD, of Leuven University Hospital said at the World Congress on Endometriosis.
“We may have to tweak it a bit,” he said.
Unfortunately, that tweaking is likely to be time consuming. That’s because RNA extraction and sequencing is labor intensive and expensive, though it could be readily automated if it becomes commercialized.
It’s also technically challenging because microRNA is present in very low concentrations in plasma so researchers must spot a rare signal surrounded by a sea of noise, and that can mean a lot of technical refinement along the way. “We did a lot of testing and trial and error to see what was the best protocol,” Dr. Vanhie said.
The news isn’t all bad. With a well-funded, intensive effort, development could be swift. “There’s no intervention. You just need a tube of blood, so you could go forward with this to make it clinically relevant very easily,” Dr. Vanhie said.
The study was sponsored by the Belgian government. Dr. Vanhie reported having no financial disclosures.
VANCOUVER – A panel of 11 microRNA biomarkers was effective in predicting endometriosis in a small validation study conducted at a Belgium hospital, showing the progress researchers are making toward a noninvasive test.
The research team at Leuven University Hospital developed the model using a population of 120 women, 82 of whom had endometriosis. After extensive analysis, they identified 11 microRNAs, which varied significantly between endometriosis patients and healthy controls and could be used as part of a panel to detect endometriosis. The panel performed well in their validation sample of 60 women with endometriosis and 30 controls, with a sensitivity of 92% and a specificity of 96%.
That validation is good news, but it is still far from being clinically useful. First, the model needs to be tested further in their own predominantly Caucasian population. Then, it needs to be validated by outside researchers, and the model will almost certainly have to be altered to account for different genetic backgrounds, lifestyle habits, and other factors, Arne Vanhie, MD, of Leuven University Hospital said at the World Congress on Endometriosis.
“We may have to tweak it a bit,” he said.
Unfortunately, that tweaking is likely to be time consuming. That’s because RNA extraction and sequencing is labor intensive and expensive, though it could be readily automated if it becomes commercialized.
It’s also technically challenging because microRNA is present in very low concentrations in plasma so researchers must spot a rare signal surrounded by a sea of noise, and that can mean a lot of technical refinement along the way. “We did a lot of testing and trial and error to see what was the best protocol,” Dr. Vanhie said.
The news isn’t all bad. With a well-funded, intensive effort, development could be swift. “There’s no intervention. You just need a tube of blood, so you could go forward with this to make it clinically relevant very easily,” Dr. Vanhie said.
The study was sponsored by the Belgian government. Dr. Vanhie reported having no financial disclosures.
VANCOUVER – A panel of 11 microRNA biomarkers was effective in predicting endometriosis in a small validation study conducted at a Belgium hospital, showing the progress researchers are making toward a noninvasive test.
The research team at Leuven University Hospital developed the model using a population of 120 women, 82 of whom had endometriosis. After extensive analysis, they identified 11 microRNAs, which varied significantly between endometriosis patients and healthy controls and could be used as part of a panel to detect endometriosis. The panel performed well in their validation sample of 60 women with endometriosis and 30 controls, with a sensitivity of 92% and a specificity of 96%.
That validation is good news, but it is still far from being clinically useful. First, the model needs to be tested further in their own predominantly Caucasian population. Then, it needs to be validated by outside researchers, and the model will almost certainly have to be altered to account for different genetic backgrounds, lifestyle habits, and other factors, Arne Vanhie, MD, of Leuven University Hospital said at the World Congress on Endometriosis.
“We may have to tweak it a bit,” he said.
Unfortunately, that tweaking is likely to be time consuming. That’s because RNA extraction and sequencing is labor intensive and expensive, though it could be readily automated if it becomes commercialized.
It’s also technically challenging because microRNA is present in very low concentrations in plasma so researchers must spot a rare signal surrounded by a sea of noise, and that can mean a lot of technical refinement along the way. “We did a lot of testing and trial and error to see what was the best protocol,” Dr. Vanhie said.
The news isn’t all bad. With a well-funded, intensive effort, development could be swift. “There’s no intervention. You just need a tube of blood, so you could go forward with this to make it clinically relevant very easily,” Dr. Vanhie said.
The study was sponsored by the Belgian government. Dr. Vanhie reported having no financial disclosures.
AT WCE 2017
Key clinical point:
Major finding: The panel detected endometriosis with 92% sensitivity and 96% specificity.
Data source: The development sample included 120 women and a validation group of 90 women.
Disclosures: The study was sponsored by the Belgian government. Dr. Vanhie reported having no financial disclosures.
Oral GnRH antagonist offers endometriosis pain relief in trials
VANCOUVER – An oral agent reduced dysmenorrhea and nonmenstrual pelvic pain in endometriosis patients, with more fine control over estrogen levels than historically seen with injectable gonadotropin-releasing hormone (GnRH) agonists, according to findings from two randomized controlled trials.
Elagolix is an oral, nonpeptide, gonadotropin-releasing hormone (GnRH) antagonist being developed by Neurocrine Biosciences and AbbVie. Its dose can be adjusted in an attempt to achieve estrogen levels in the optimal “therapeutic window” that controls endometriosis pain while reducing menopausal symptoms, according to Hugh S. Taylor, MD, professor of obstetrics, gynecology and reproductive services at Yale University, New Haven, Conn., and chief of obstetrics and gynecology at Yale–New Haven Hospital, who presented the research at the World Congress on Endometriosis.
Results from two phase III clinical trials, simultaneously published in the New England Journal of Medicine, show that two different doses of elagolix – 150 mg once daily or 200 mg twice daily – improved moderate or severe endometriosis-associated pain. However, patients taking the drug experienced heightened frequencies of hot flushes and increased serum lipid levels, and decreases from baseline in bone mineral density (N Engl J Med. 2017 May 19. doi: 10.1056/NEJMoa1700089).
Still, the two doses give physicians options in tailoring the drug for their patients, Dr. Taylor said. The key is to keep the levels within the therapeutic window. “That’s always been the goal, and now we have a drug that does that. You can customize it for your patient, using the stronger dose for those who need it,” he said.
He described the results from two parallel clinical trials, one conducted in the United States and Canada (Elaris Endometriosis I, n = 872) and one conducted at 187 sites on five continents (Elaris Endometriosis II, n = 817).
Patients were allowed to use NSAIDs (500 mg of naproxen) or an opioid, or both, as needed. Dr. Taylor reported the results after 6 months of treatment.
Both doses of the drug outperformed placebo in reducing dysmenorrhea at 3 months. In Elaris EM-I, 75.8% in the high-dose group and 46.4% in the low-dose group had a clinically significant reduction in dysmenorrhea and decreased or stable use of analgesics at 3 months, compared with 19.6% in the placebo group. In Elaris EM-II, 72.4% in the high-dose group and 43.4% in the low-dose group achieved a clinically significant reduction, compared with 22.7% in the placebo group (P less than .001 for all comparisons).
For nonmenstrual pelvic pain, the two doses of elagolix again bested placebo. In Elaris EM-I, 54.5% in the high-dose group and 50.4% in the low-dose group achieved a clinically significant reduction and a decreased or stable use of analgesics, compared with 36.5% on placebo (P less than .001 for all). In Elaris EM-II, 57.8% in the high-dose group and 49.8% in the low-dose group achieved a clinically significant response, compared with 36.5% in the placebo group (P less than .001 and P = .003, respectively).
The responses were sustained at 6 months for both outcomes.
In Elaris EM-I, hot flushes were reported by 7.0% of the placebo group, 23.7% of the low-dose group, and 42.3% of the high-dose group (P less than .001). In Elaris EM-II, they were reported in 10.3% of the placebo group, 22.6% of the low-dose group, and 47.6% of the high-dose group (P less than .001).
Patients in the elagolix groups experienced increases in total cholesterol, LDL cholesterol, HDL cholesterol, and triglycerides, though the researchers noted that less than 20% of participants in each group had LDL levels higher than 160 mg/dL or triglycerides levels higher than 200 mg/dL at any point during treatment.
There were also decreases in bone mineral density in the elagolix groups, but after 6 months of treatment, a z score of –1.5 or less at the lumbar spine occurred in fewer than 5% of women in the elagolix groups.
The drug should provide a more palatable option to GnRH agonists, according to Dr. Taylor. “This is a big step forward, very effective and much more tolerable. I think having an oral, rapidly acting, reversible drug, with a couple of doses available, will make this much more widely accepted and just as effective.”
The study was sponsored by AbbVie. Dr. Taylor and other researchers on the study reported financial ties to AbbVie and other pharmaceutical companies.
VANCOUVER – An oral agent reduced dysmenorrhea and nonmenstrual pelvic pain in endometriosis patients, with more fine control over estrogen levels than historically seen with injectable gonadotropin-releasing hormone (GnRH) agonists, according to findings from two randomized controlled trials.
Elagolix is an oral, nonpeptide, gonadotropin-releasing hormone (GnRH) antagonist being developed by Neurocrine Biosciences and AbbVie. Its dose can be adjusted in an attempt to achieve estrogen levels in the optimal “therapeutic window” that controls endometriosis pain while reducing menopausal symptoms, according to Hugh S. Taylor, MD, professor of obstetrics, gynecology and reproductive services at Yale University, New Haven, Conn., and chief of obstetrics and gynecology at Yale–New Haven Hospital, who presented the research at the World Congress on Endometriosis.
Results from two phase III clinical trials, simultaneously published in the New England Journal of Medicine, show that two different doses of elagolix – 150 mg once daily or 200 mg twice daily – improved moderate or severe endometriosis-associated pain. However, patients taking the drug experienced heightened frequencies of hot flushes and increased serum lipid levels, and decreases from baseline in bone mineral density (N Engl J Med. 2017 May 19. doi: 10.1056/NEJMoa1700089).
Still, the two doses give physicians options in tailoring the drug for their patients, Dr. Taylor said. The key is to keep the levels within the therapeutic window. “That’s always been the goal, and now we have a drug that does that. You can customize it for your patient, using the stronger dose for those who need it,” he said.
He described the results from two parallel clinical trials, one conducted in the United States and Canada (Elaris Endometriosis I, n = 872) and one conducted at 187 sites on five continents (Elaris Endometriosis II, n = 817).
Patients were allowed to use NSAIDs (500 mg of naproxen) or an opioid, or both, as needed. Dr. Taylor reported the results after 6 months of treatment.
Both doses of the drug outperformed placebo in reducing dysmenorrhea at 3 months. In Elaris EM-I, 75.8% in the high-dose group and 46.4% in the low-dose group had a clinically significant reduction in dysmenorrhea and decreased or stable use of analgesics at 3 months, compared with 19.6% in the placebo group. In Elaris EM-II, 72.4% in the high-dose group and 43.4% in the low-dose group achieved a clinically significant reduction, compared with 22.7% in the placebo group (P less than .001 for all comparisons).
For nonmenstrual pelvic pain, the two doses of elagolix again bested placebo. In Elaris EM-I, 54.5% in the high-dose group and 50.4% in the low-dose group achieved a clinically significant reduction and a decreased or stable use of analgesics, compared with 36.5% on placebo (P less than .001 for all). In Elaris EM-II, 57.8% in the high-dose group and 49.8% in the low-dose group achieved a clinically significant response, compared with 36.5% in the placebo group (P less than .001 and P = .003, respectively).
The responses were sustained at 6 months for both outcomes.
In Elaris EM-I, hot flushes were reported by 7.0% of the placebo group, 23.7% of the low-dose group, and 42.3% of the high-dose group (P less than .001). In Elaris EM-II, they were reported in 10.3% of the placebo group, 22.6% of the low-dose group, and 47.6% of the high-dose group (P less than .001).
Patients in the elagolix groups experienced increases in total cholesterol, LDL cholesterol, HDL cholesterol, and triglycerides, though the researchers noted that less than 20% of participants in each group had LDL levels higher than 160 mg/dL or triglycerides levels higher than 200 mg/dL at any point during treatment.
There were also decreases in bone mineral density in the elagolix groups, but after 6 months of treatment, a z score of –1.5 or less at the lumbar spine occurred in fewer than 5% of women in the elagolix groups.
The drug should provide a more palatable option to GnRH agonists, according to Dr. Taylor. “This is a big step forward, very effective and much more tolerable. I think having an oral, rapidly acting, reversible drug, with a couple of doses available, will make this much more widely accepted and just as effective.”
The study was sponsored by AbbVie. Dr. Taylor and other researchers on the study reported financial ties to AbbVie and other pharmaceutical companies.
VANCOUVER – An oral agent reduced dysmenorrhea and nonmenstrual pelvic pain in endometriosis patients, with more fine control over estrogen levels than historically seen with injectable gonadotropin-releasing hormone (GnRH) agonists, according to findings from two randomized controlled trials.
Elagolix is an oral, nonpeptide, gonadotropin-releasing hormone (GnRH) antagonist being developed by Neurocrine Biosciences and AbbVie. Its dose can be adjusted in an attempt to achieve estrogen levels in the optimal “therapeutic window” that controls endometriosis pain while reducing menopausal symptoms, according to Hugh S. Taylor, MD, professor of obstetrics, gynecology and reproductive services at Yale University, New Haven, Conn., and chief of obstetrics and gynecology at Yale–New Haven Hospital, who presented the research at the World Congress on Endometriosis.
Results from two phase III clinical trials, simultaneously published in the New England Journal of Medicine, show that two different doses of elagolix – 150 mg once daily or 200 mg twice daily – improved moderate or severe endometriosis-associated pain. However, patients taking the drug experienced heightened frequencies of hot flushes and increased serum lipid levels, and decreases from baseline in bone mineral density (N Engl J Med. 2017 May 19. doi: 10.1056/NEJMoa1700089).
Still, the two doses give physicians options in tailoring the drug for their patients, Dr. Taylor said. The key is to keep the levels within the therapeutic window. “That’s always been the goal, and now we have a drug that does that. You can customize it for your patient, using the stronger dose for those who need it,” he said.
He described the results from two parallel clinical trials, one conducted in the United States and Canada (Elaris Endometriosis I, n = 872) and one conducted at 187 sites on five continents (Elaris Endometriosis II, n = 817).
Patients were allowed to use NSAIDs (500 mg of naproxen) or an opioid, or both, as needed. Dr. Taylor reported the results after 6 months of treatment.
Both doses of the drug outperformed placebo in reducing dysmenorrhea at 3 months. In Elaris EM-I, 75.8% in the high-dose group and 46.4% in the low-dose group had a clinically significant reduction in dysmenorrhea and decreased or stable use of analgesics at 3 months, compared with 19.6% in the placebo group. In Elaris EM-II, 72.4% in the high-dose group and 43.4% in the low-dose group achieved a clinically significant reduction, compared with 22.7% in the placebo group (P less than .001 for all comparisons).
For nonmenstrual pelvic pain, the two doses of elagolix again bested placebo. In Elaris EM-I, 54.5% in the high-dose group and 50.4% in the low-dose group achieved a clinically significant reduction and a decreased or stable use of analgesics, compared with 36.5% on placebo (P less than .001 for all). In Elaris EM-II, 57.8% in the high-dose group and 49.8% in the low-dose group achieved a clinically significant response, compared with 36.5% in the placebo group (P less than .001 and P = .003, respectively).
The responses were sustained at 6 months for both outcomes.
In Elaris EM-I, hot flushes were reported by 7.0% of the placebo group, 23.7% of the low-dose group, and 42.3% of the high-dose group (P less than .001). In Elaris EM-II, they were reported in 10.3% of the placebo group, 22.6% of the low-dose group, and 47.6% of the high-dose group (P less than .001).
Patients in the elagolix groups experienced increases in total cholesterol, LDL cholesterol, HDL cholesterol, and triglycerides, though the researchers noted that less than 20% of participants in each group had LDL levels higher than 160 mg/dL or triglycerides levels higher than 200 mg/dL at any point during treatment.
There were also decreases in bone mineral density in the elagolix groups, but after 6 months of treatment, a z score of –1.5 or less at the lumbar spine occurred in fewer than 5% of women in the elagolix groups.
The drug should provide a more palatable option to GnRH agonists, according to Dr. Taylor. “This is a big step forward, very effective and much more tolerable. I think having an oral, rapidly acting, reversible drug, with a couple of doses available, will make this much more widely accepted and just as effective.”
The study was sponsored by AbbVie. Dr. Taylor and other researchers on the study reported financial ties to AbbVie and other pharmaceutical companies.
Key clinical point:
Major finding: At 3 and 6 months, the drug achieved a clinically significant reduction in dysmenorrhea for between 43% and 77% of women, compared with placebo (P less than .001).
Data source: Two phase III controlled trials randomizing nearly 1,700 with surgically confirmed endometriosis.
Disclosures: The study was sponsored by AbbVie. Dr. Taylor and other researchers on the study reported financial ties to AbbVie and other pharmaceutical companies.