User login
Wide variability found in invasive mediastinal staging rates for lung cancer
COLORADO SPRINGS – Significant variability exists between hospitals in Washington state in their rates of invasive mediastinal staging for lung cancer, Farhood Farjah, MD, reported at the annual meeting of the Western Thoracic Surgical Association.
“We found evidence of a fivefold variation in hospital-level rates of invasive mediastinal staging not explained by chance or case mix,” according to Dr. Farjah of the University of Washington, Seattle.
“This has led to substantial concerns about quality of thoracic surgical care in the community at large,” he noted.
The Washington study is the first to show hospital-by-hospital variation in rates of invasive mediastinal staging.
Invasive mediastinal staging for lung cancer is considered important because imaging is known to have a substantial false-negative rate, and staging results have a profound impact on treatment recommendations, which can range from surgery alone to additional chemoradiation therapy.
Yet the meaning of the hospital-level huge variability in practice observed in the Washington study remains unclear.
“Our understanding of the underutilization of invasive mediastinal staging is further complicated by the fact that patterns of invasive mediastinal staging are highly variable across hospitals staffed by at least one board-certified thoracic surgeon with a noncardiac practice,” Dr. Farjah explained. “This variability could be a marker of poor-quality care. However, because the guidelines are not supported by level 1 evidence, it’s equally plausible that this variability might represent uncertainty or even disagreement with the practice guidelines – and specifically about the appropriate indication for invasive staging.”
He presented a retrospective cohort study of 406 patients whose non–small cell lung cancer was resected during July 2011–December 2013 at one of five Washington hospitals, each with at least one board-certified thoracic surgeon with a noncardiac practice on staff. The four participating community hospitals and one academic medical center were involved in a National Cancer Institute–funded, physician-led quality improvement initiative.
Overall, 66% of the 406 patients underwent any form of invasive mediastinal staging: 85% by mediastinoscopy only; 12% by mediastinoscopy plus endobronchial ultrasound-guided nodal aspiration (EBUS); 3% by EBUS only; and the remaining handful by mediastinoscopy, EBUS, and esophageal ultrasound-guided nodal aspiration. The invasive staging was performed at the time of resection in 64% of cases. A median of three nodal stations were sampled.
After statistical adjustment for random variation and between-hospital differences in clinical stage, rates of invasive staging were all over the map. While an overall mean of 66% of the lung cancer patients underwent invasive mediastinal staging, the rates at the five hospitals were 94%, 84%, 31%, 80%, and 17%.
Dr. Farjah and his coinvestigators are now conducting provider interviews and focus groups in an effort to understand what drove the participating surgeons’ wide variability in performing invasive mediastinal staging.
Discussant Jane Yanagawa, MD, of the University of California, Los Angeles, commented, “I think this is a really interesting study because, historically, lower rates of mediastinoscopy are assumed to be a reflection of low-quality care – and you suggest that might not be the case, that it might be more complicated than that.”
Dr. Yanagawa sketched one fairly common scenario that might represent a surgeon’s reasonable avoidance of guideline-recommended invasive mediastinal staging: a patient who by all preoperative imaging appears to have stage IA lung cancer and wishes to avoid the morbidity, time, and cost of needle biopsy, instead choosing to go straight to the operating room for a diagnosis by wedge resection, followed by a completion lobectomy based upon the frozen section results. Could such a pathway account for the variability seen in the Washington study?
“I think it could have,” Dr. Farjah replied. “I would say that’s probably one driver of variability.”
As for the generalizability of the findings of a five-hospital study carried out in a single state, Dr. Farjah said he thinks the results are applicable to any academic or community hospital with at least one board-certified thoracic surgeon with a noncardiac practice.
He reported having no financial conflicts of interest regarding the study.
M. Patricia Rivera, MD, FCCP, comments: Staging of lung cancer is essential to select the best treatment strategy for a given patient. However, despite multiple guideline recommendations
M. Patricia Rivera, MD, FCCP, comments: Staging of lung cancer is essential to select the best treatment strategy for a given patient. However, despite multiple guideline recommendations
M. Patricia Rivera, MD, FCCP, comments: Staging of lung cancer is essential to select the best treatment strategy for a given patient. However, despite multiple guideline recommendations
COLORADO SPRINGS – Significant variability exists between hospitals in Washington state in their rates of invasive mediastinal staging for lung cancer, Farhood Farjah, MD, reported at the annual meeting of the Western Thoracic Surgical Association.
“We found evidence of a fivefold variation in hospital-level rates of invasive mediastinal staging not explained by chance or case mix,” according to Dr. Farjah of the University of Washington, Seattle.
“This has led to substantial concerns about quality of thoracic surgical care in the community at large,” he noted.
The Washington study is the first to show hospital-by-hospital variation in rates of invasive mediastinal staging.
Invasive mediastinal staging for lung cancer is considered important because imaging is known to have a substantial false-negative rate, and staging results have a profound impact on treatment recommendations, which can range from surgery alone to additional chemoradiation therapy.
Yet the meaning of the hospital-level huge variability in practice observed in the Washington study remains unclear.
“Our understanding of the underutilization of invasive mediastinal staging is further complicated by the fact that patterns of invasive mediastinal staging are highly variable across hospitals staffed by at least one board-certified thoracic surgeon with a noncardiac practice,” Dr. Farjah explained. “This variability could be a marker of poor-quality care. However, because the guidelines are not supported by level 1 evidence, it’s equally plausible that this variability might represent uncertainty or even disagreement with the practice guidelines – and specifically about the appropriate indication for invasive staging.”
He presented a retrospective cohort study of 406 patients whose non–small cell lung cancer was resected during July 2011–December 2013 at one of five Washington hospitals, each with at least one board-certified thoracic surgeon with a noncardiac practice on staff. The four participating community hospitals and one academic medical center were involved in a National Cancer Institute–funded, physician-led quality improvement initiative.
Overall, 66% of the 406 patients underwent any form of invasive mediastinal staging: 85% by mediastinoscopy only; 12% by mediastinoscopy plus endobronchial ultrasound-guided nodal aspiration (EBUS); 3% by EBUS only; and the remaining handful by mediastinoscopy, EBUS, and esophageal ultrasound-guided nodal aspiration. The invasive staging was performed at the time of resection in 64% of cases. A median of three nodal stations were sampled.
After statistical adjustment for random variation and between-hospital differences in clinical stage, rates of invasive staging were all over the map. While an overall mean of 66% of the lung cancer patients underwent invasive mediastinal staging, the rates at the five hospitals were 94%, 84%, 31%, 80%, and 17%.
Dr. Farjah and his coinvestigators are now conducting provider interviews and focus groups in an effort to understand what drove the participating surgeons’ wide variability in performing invasive mediastinal staging.
Discussant Jane Yanagawa, MD, of the University of California, Los Angeles, commented, “I think this is a really interesting study because, historically, lower rates of mediastinoscopy are assumed to be a reflection of low-quality care – and you suggest that might not be the case, that it might be more complicated than that.”
Dr. Yanagawa sketched one fairly common scenario that might represent a surgeon’s reasonable avoidance of guideline-recommended invasive mediastinal staging: a patient who by all preoperative imaging appears to have stage IA lung cancer and wishes to avoid the morbidity, time, and cost of needle biopsy, instead choosing to go straight to the operating room for a diagnosis by wedge resection, followed by a completion lobectomy based upon the frozen section results. Could such a pathway account for the variability seen in the Washington study?
“I think it could have,” Dr. Farjah replied. “I would say that’s probably one driver of variability.”
As for the generalizability of the findings of a five-hospital study carried out in a single state, Dr. Farjah said he thinks the results are applicable to any academic or community hospital with at least one board-certified thoracic surgeon with a noncardiac practice.
He reported having no financial conflicts of interest regarding the study.
COLORADO SPRINGS – Significant variability exists between hospitals in Washington state in their rates of invasive mediastinal staging for lung cancer, Farhood Farjah, MD, reported at the annual meeting of the Western Thoracic Surgical Association.
“We found evidence of a fivefold variation in hospital-level rates of invasive mediastinal staging not explained by chance or case mix,” according to Dr. Farjah of the University of Washington, Seattle.
“This has led to substantial concerns about quality of thoracic surgical care in the community at large,” he noted.
The Washington study is the first to show hospital-by-hospital variation in rates of invasive mediastinal staging.
Invasive mediastinal staging for lung cancer is considered important because imaging is known to have a substantial false-negative rate, and staging results have a profound impact on treatment recommendations, which can range from surgery alone to additional chemoradiation therapy.
Yet the meaning of the hospital-level huge variability in practice observed in the Washington study remains unclear.
“Our understanding of the underutilization of invasive mediastinal staging is further complicated by the fact that patterns of invasive mediastinal staging are highly variable across hospitals staffed by at least one board-certified thoracic surgeon with a noncardiac practice,” Dr. Farjah explained. “This variability could be a marker of poor-quality care. However, because the guidelines are not supported by level 1 evidence, it’s equally plausible that this variability might represent uncertainty or even disagreement with the practice guidelines – and specifically about the appropriate indication for invasive staging.”
He presented a retrospective cohort study of 406 patients whose non–small cell lung cancer was resected during July 2011–December 2013 at one of five Washington hospitals, each with at least one board-certified thoracic surgeon with a noncardiac practice on staff. The four participating community hospitals and one academic medical center were involved in a National Cancer Institute–funded, physician-led quality improvement initiative.
Overall, 66% of the 406 patients underwent any form of invasive mediastinal staging: 85% by mediastinoscopy only; 12% by mediastinoscopy plus endobronchial ultrasound-guided nodal aspiration (EBUS); 3% by EBUS only; and the remaining handful by mediastinoscopy, EBUS, and esophageal ultrasound-guided nodal aspiration. The invasive staging was performed at the time of resection in 64% of cases. A median of three nodal stations were sampled.
After statistical adjustment for random variation and between-hospital differences in clinical stage, rates of invasive staging were all over the map. While an overall mean of 66% of the lung cancer patients underwent invasive mediastinal staging, the rates at the five hospitals were 94%, 84%, 31%, 80%, and 17%.
Dr. Farjah and his coinvestigators are now conducting provider interviews and focus groups in an effort to understand what drove the participating surgeons’ wide variability in performing invasive mediastinal staging.
Discussant Jane Yanagawa, MD, of the University of California, Los Angeles, commented, “I think this is a really interesting study because, historically, lower rates of mediastinoscopy are assumed to be a reflection of low-quality care – and you suggest that might not be the case, that it might be more complicated than that.”
Dr. Yanagawa sketched one fairly common scenario that might represent a surgeon’s reasonable avoidance of guideline-recommended invasive mediastinal staging: a patient who by all preoperative imaging appears to have stage IA lung cancer and wishes to avoid the morbidity, time, and cost of needle biopsy, instead choosing to go straight to the operating room for a diagnosis by wedge resection, followed by a completion lobectomy based upon the frozen section results. Could such a pathway account for the variability seen in the Washington study?
“I think it could have,” Dr. Farjah replied. “I would say that’s probably one driver of variability.”
As for the generalizability of the findings of a five-hospital study carried out in a single state, Dr. Farjah said he thinks the results are applicable to any academic or community hospital with at least one board-certified thoracic surgeon with a noncardiac practice.
He reported having no financial conflicts of interest regarding the study.
AT THE WTSA ANNUAL MEETING
Key clinical point:
Major finding: Rates of invasive mediastinal staging after adjustment for clinical stage ranged from a low of 17% at one hospital to as high as 94% at another.
Data source: This retrospective cohort study included 406 patients.
Disclosures: Dr. Farjah reported having no financial conflicts of interest.
TAVR for failed surgical valves: the VIVA study
PARIS – Transcatheter aortic valve replacement using the self-expanding Evolut R device in high-surgical-risk patients with failing surgical aortic bioprostheses showed promising 30-day safety and effectiveness results in the ongoing VIVA study, Ran Kornowski, MD, reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
“We had a lot of patients with small, failing valves in this study. Despite this, our valve gradients postprocedure were very, very low. This means to me that this platform is very well suited to deal with valve-in-valve procedures in general and with small bioprosthetic valves in particular,” observed Dr. Kornowski, chairman of the department of cardiology at Rabin Medical Center in Petah Tikva, Israel, and president of the Israel Heart Society.
The participants’ last surgical aortic valve replacement had been a mean of 9.3 years earlier. Seventy-one percent of subjects were New York Heart Association class III or IV. The mode of bioprosthetic failure was stenosis in 56% of cases, regurgitation in 23%, and both in the remainder. Ninety-three percent of their failing biosprothetic valves were stented devices. Forty-one percent of the devices were up to 21 mm and another 33% were more than 21 but less than 25 mm.
TAVR procedural access was by the iliofemoral route in 97% of cases. Local anesthesia was used in 42% of cases and conscious sedation in 35%. Fourteen percent of patients underwent preimplantation valvuloplasty; 21% postimplantation valvuloplasty. The procedural success rate was 98.5%.
The primary safety endpoint was 30-day cardiovascular mortality. The 2.0% rate was far lower than the prespecified cutoff which defined a positive outcome as less than a 10% rate in this high-surgical-risk population.
Other key 30-day outcomes:
• All-cause mortality occurred in 2.5% of patients.
• The 30-day stroke rate was 3%, with no disabling strokes.
• Major vascular complications occurred in 6.5% of the VIVA patients.
• Major bleeding occurred in 7%, minor bleeding in 7.9%. There were no cases of life-threatening bleeding.
• The incidence of Stage I acute kidney injury was rare, at 0.5%.
• Seven percent of patients received a permanent pacemaker.
• Eighty-seven percent of patients had no postprocedure paravalvular regurgitation (PVR), 11.4 had mild PVR, and 1.5% had moderate PVR.
• NYHA classification improved from baseline to 30 days in 81% of patients. At 30 days, 93% of participants were NYHA class I or II.
Turning to echocardiographic findings, the mean gradient improved from a mean baseline of 31.8 mm Hg to 12.6 mm Hg, while the effective orifice area rose from 1.0 to 1.5 cm2. The magnitude of both improvements was greater for patients with stenosis as their mode of valve failure.
“With the Evolut R, we aim for higher implantations – not more than about 4 mm below the ring – because going deeper could bring about higher gradients and functional deterioration later on,” the cardiologist explained.
The 1-year primary efficacy endpoint – lack of significant aortic stenosis as defined by a mean gradient less than 40 mm Hg – will be reported soon. The VIVA study is sponsored by Medtronic. Dr. Kornowski reported serving as a consultant to the company.
PARIS – Transcatheter aortic valve replacement using the self-expanding Evolut R device in high-surgical-risk patients with failing surgical aortic bioprostheses showed promising 30-day safety and effectiveness results in the ongoing VIVA study, Ran Kornowski, MD, reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
“We had a lot of patients with small, failing valves in this study. Despite this, our valve gradients postprocedure were very, very low. This means to me that this platform is very well suited to deal with valve-in-valve procedures in general and with small bioprosthetic valves in particular,” observed Dr. Kornowski, chairman of the department of cardiology at Rabin Medical Center in Petah Tikva, Israel, and president of the Israel Heart Society.
The participants’ last surgical aortic valve replacement had been a mean of 9.3 years earlier. Seventy-one percent of subjects were New York Heart Association class III or IV. The mode of bioprosthetic failure was stenosis in 56% of cases, regurgitation in 23%, and both in the remainder. Ninety-three percent of their failing biosprothetic valves were stented devices. Forty-one percent of the devices were up to 21 mm and another 33% were more than 21 but less than 25 mm.
TAVR procedural access was by the iliofemoral route in 97% of cases. Local anesthesia was used in 42% of cases and conscious sedation in 35%. Fourteen percent of patients underwent preimplantation valvuloplasty; 21% postimplantation valvuloplasty. The procedural success rate was 98.5%.
The primary safety endpoint was 30-day cardiovascular mortality. The 2.0% rate was far lower than the prespecified cutoff which defined a positive outcome as less than a 10% rate in this high-surgical-risk population.
Other key 30-day outcomes:
• All-cause mortality occurred in 2.5% of patients.
• The 30-day stroke rate was 3%, with no disabling strokes.
• Major vascular complications occurred in 6.5% of the VIVA patients.
• Major bleeding occurred in 7%, minor bleeding in 7.9%. There were no cases of life-threatening bleeding.
• The incidence of Stage I acute kidney injury was rare, at 0.5%.
• Seven percent of patients received a permanent pacemaker.
• Eighty-seven percent of patients had no postprocedure paravalvular regurgitation (PVR), 11.4 had mild PVR, and 1.5% had moderate PVR.
• NYHA classification improved from baseline to 30 days in 81% of patients. At 30 days, 93% of participants were NYHA class I or II.
Turning to echocardiographic findings, the mean gradient improved from a mean baseline of 31.8 mm Hg to 12.6 mm Hg, while the effective orifice area rose from 1.0 to 1.5 cm2. The magnitude of both improvements was greater for patients with stenosis as their mode of valve failure.
“With the Evolut R, we aim for higher implantations – not more than about 4 mm below the ring – because going deeper could bring about higher gradients and functional deterioration later on,” the cardiologist explained.
The 1-year primary efficacy endpoint – lack of significant aortic stenosis as defined by a mean gradient less than 40 mm Hg – will be reported soon. The VIVA study is sponsored by Medtronic. Dr. Kornowski reported serving as a consultant to the company.
PARIS – Transcatheter aortic valve replacement using the self-expanding Evolut R device in high-surgical-risk patients with failing surgical aortic bioprostheses showed promising 30-day safety and effectiveness results in the ongoing VIVA study, Ran Kornowski, MD, reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
“We had a lot of patients with small, failing valves in this study. Despite this, our valve gradients postprocedure were very, very low. This means to me that this platform is very well suited to deal with valve-in-valve procedures in general and with small bioprosthetic valves in particular,” observed Dr. Kornowski, chairman of the department of cardiology at Rabin Medical Center in Petah Tikva, Israel, and president of the Israel Heart Society.
The participants’ last surgical aortic valve replacement had been a mean of 9.3 years earlier. Seventy-one percent of subjects were New York Heart Association class III or IV. The mode of bioprosthetic failure was stenosis in 56% of cases, regurgitation in 23%, and both in the remainder. Ninety-three percent of their failing biosprothetic valves were stented devices. Forty-one percent of the devices were up to 21 mm and another 33% were more than 21 but less than 25 mm.
TAVR procedural access was by the iliofemoral route in 97% of cases. Local anesthesia was used in 42% of cases and conscious sedation in 35%. Fourteen percent of patients underwent preimplantation valvuloplasty; 21% postimplantation valvuloplasty. The procedural success rate was 98.5%.
The primary safety endpoint was 30-day cardiovascular mortality. The 2.0% rate was far lower than the prespecified cutoff which defined a positive outcome as less than a 10% rate in this high-surgical-risk population.
Other key 30-day outcomes:
• All-cause mortality occurred in 2.5% of patients.
• The 30-day stroke rate was 3%, with no disabling strokes.
• Major vascular complications occurred in 6.5% of the VIVA patients.
• Major bleeding occurred in 7%, minor bleeding in 7.9%. There were no cases of life-threatening bleeding.
• The incidence of Stage I acute kidney injury was rare, at 0.5%.
• Seven percent of patients received a permanent pacemaker.
• Eighty-seven percent of patients had no postprocedure paravalvular regurgitation (PVR), 11.4 had mild PVR, and 1.5% had moderate PVR.
• NYHA classification improved from baseline to 30 days in 81% of patients. At 30 days, 93% of participants were NYHA class I or II.
Turning to echocardiographic findings, the mean gradient improved from a mean baseline of 31.8 mm Hg to 12.6 mm Hg, while the effective orifice area rose from 1.0 to 1.5 cm2. The magnitude of both improvements was greater for patients with stenosis as their mode of valve failure.
“With the Evolut R, we aim for higher implantations – not more than about 4 mm below the ring – because going deeper could bring about higher gradients and functional deterioration later on,” the cardiologist explained.
The 1-year primary efficacy endpoint – lack of significant aortic stenosis as defined by a mean gradient less than 40 mm Hg – will be reported soon. The VIVA study is sponsored by Medtronic. Dr. Kornowski reported serving as a consultant to the company.
AT EuroPCR
Key clinical point:
Major finding: The 30-day cardiovascular mortality rate after transcatheter aortic valve replacement via a valve-in-valve procedure in patients with a failing surgically implanted bioprosthesis was 2%, compared with a projected rate of at least 10% with redo surgery.
Data source: VIVA, a prospective observational registry of 202 high-surgical-risk patients at 23 centers in four countries who underwent valve-in-valve transcatheter aortic valve replacement because of a failing surgically implanted aortic bioprosthesis.
Disclosures: VIVA is sponsored by Medtronic. The presenter reported serving as a consultant to the company.
Bailout stenting for coronary bifurcations brings ‘unacceptable’ hazards
PARIS – Bailout stenting during percutaneous coronary intervention for coronary bifurcations doubled the risk of major adverse cardiovascular events in the world’s largest registry of patients with these often-challenging lesions treated using bioactive stents, Marco Zimarino, MD, reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
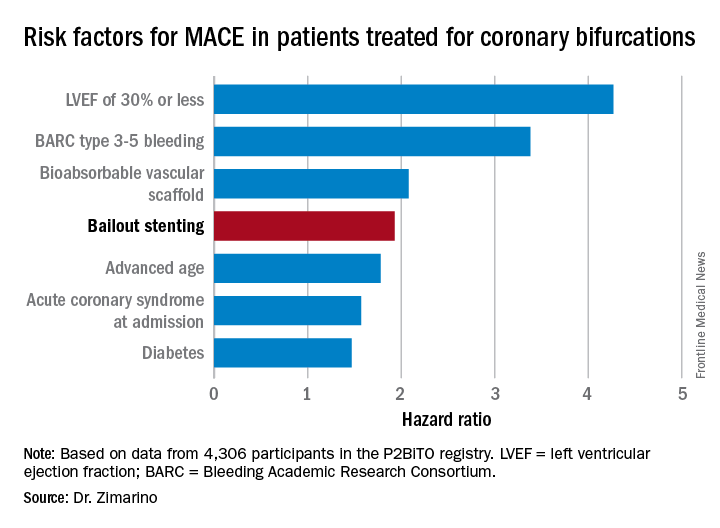
Indeed, resort to bailout stenting stood out as the major potentially modifiable risk factor for adverse outcomes among the 4,306 participants in the P2BiTO registry, an international collaboration supported by members of the EuroBifurcation Club. Most of the other independent risk factors identified in a multivariate regression analysis of the P2BiTO database were beyond operator control, including diabetes, advanced age, and presentation with an acute coronary syndrome, according to Dr. Zimarino of the University of Chieti (Italy).
Bailout stenting is largely avoidable through meticulous procedural planning, the interventional cardiologist added.
“Careful planning is always mandatory because bailout stenting is associated with an unacceptably higher risk of both in-hospital and 1-year adverse outcomes,” Dr. Zimarino emphasized. “It’s much better to leave a degraded side branch instead of using bailout stenting to get an excellent angiographic outcome that’s a predictor of a worse clinical outcome.”
Conventional wisdom holds that single stenting of either the main artery or a side branch in a patient with coronary bifurcation is safer than double stenting of both. However, that wasn’t really borne out in the P2BiTO registry provided the operator’s plan was for double stenting. The difference in 1-year major adverse cardiovascular events (MACE) between patients treated using a single- or double-stenting strategy wasn’t statistically significant, provided bailout stenting wasn’t utilized. If bailout stenting was employed, though, the risk of MACE was 2.2-fold greater than if the cardiologist stuck with the plan.
Ninety-eight percent of patients in the P2BiTO registry received drug-eluting stents. The other 2% got the Absorb bioabsorbable vascular scaffold. The percutaneous coronary intervention access site, treatment strategy, choice of stent, and duration of dual-antiplatelet therapy were left up to the operator’s discretion.
The risk of MACE was reduced by 39% in patients on dual-antiplatelet therapy for 6-12 months, compared with less than 6 months.
Discussant Graham Cassel, MD, director of the heart transplant unit at Milpark Hospital in Johannesburg, commented, “The message comes through very clearly that, if you plan your procedure well, the chance of bailout is far less – and if you do have to bail out, the results are uniformly bad. If you can avoid putting in two or three stents, that’s beneficial.”
Dr. Zimarino reported having no financial conflicts of interest regarding his presentation.
PARIS – Bailout stenting during percutaneous coronary intervention for coronary bifurcations doubled the risk of major adverse cardiovascular events in the world’s largest registry of patients with these often-challenging lesions treated using bioactive stents, Marco Zimarino, MD, reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
Indeed, resort to bailout stenting stood out as the major potentially modifiable risk factor for adverse outcomes among the 4,306 participants in the P2BiTO registry, an international collaboration supported by members of the EuroBifurcation Club. Most of the other independent risk factors identified in a multivariate regression analysis of the P2BiTO database were beyond operator control, including diabetes, advanced age, and presentation with an acute coronary syndrome, according to Dr. Zimarino of the University of Chieti (Italy).
Bailout stenting is largely avoidable through meticulous procedural planning, the interventional cardiologist added.
“Careful planning is always mandatory because bailout stenting is associated with an unacceptably higher risk of both in-hospital and 1-year adverse outcomes,” Dr. Zimarino emphasized. “It’s much better to leave a degraded side branch instead of using bailout stenting to get an excellent angiographic outcome that’s a predictor of a worse clinical outcome.”
Conventional wisdom holds that single stenting of either the main artery or a side branch in a patient with coronary bifurcation is safer than double stenting of both. However, that wasn’t really borne out in the P2BiTO registry provided the operator’s plan was for double stenting. The difference in 1-year major adverse cardiovascular events (MACE) between patients treated using a single- or double-stenting strategy wasn’t statistically significant, provided bailout stenting wasn’t utilized. If bailout stenting was employed, though, the risk of MACE was 2.2-fold greater than if the cardiologist stuck with the plan.
Ninety-eight percent of patients in the P2BiTO registry received drug-eluting stents. The other 2% got the Absorb bioabsorbable vascular scaffold. The percutaneous coronary intervention access site, treatment strategy, choice of stent, and duration of dual-antiplatelet therapy were left up to the operator’s discretion.

The risk of MACE was reduced by 39% in patients on dual-antiplatelet therapy for 6-12 months, compared with less than 6 months.
Discussant Graham Cassel, MD, director of the heart transplant unit at Milpark Hospital in Johannesburg, commented, “The message comes through very clearly that, if you plan your procedure well, the chance of bailout is far less – and if you do have to bail out, the results are uniformly bad. If you can avoid putting in two or three stents, that’s beneficial.”
Dr. Zimarino reported having no financial conflicts of interest regarding his presentation.
PARIS – Bailout stenting during percutaneous coronary intervention for coronary bifurcations doubled the risk of major adverse cardiovascular events in the world’s largest registry of patients with these often-challenging lesions treated using bioactive stents, Marco Zimarino, MD, reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
Indeed, resort to bailout stenting stood out as the major potentially modifiable risk factor for adverse outcomes among the 4,306 participants in the P2BiTO registry, an international collaboration supported by members of the EuroBifurcation Club. Most of the other independent risk factors identified in a multivariate regression analysis of the P2BiTO database were beyond operator control, including diabetes, advanced age, and presentation with an acute coronary syndrome, according to Dr. Zimarino of the University of Chieti (Italy).
Bailout stenting is largely avoidable through meticulous procedural planning, the interventional cardiologist added.
“Careful planning is always mandatory because bailout stenting is associated with an unacceptably higher risk of both in-hospital and 1-year adverse outcomes,” Dr. Zimarino emphasized. “It’s much better to leave a degraded side branch instead of using bailout stenting to get an excellent angiographic outcome that’s a predictor of a worse clinical outcome.”
Conventional wisdom holds that single stenting of either the main artery or a side branch in a patient with coronary bifurcation is safer than double stenting of both. However, that wasn’t really borne out in the P2BiTO registry provided the operator’s plan was for double stenting. The difference in 1-year major adverse cardiovascular events (MACE) between patients treated using a single- or double-stenting strategy wasn’t statistically significant, provided bailout stenting wasn’t utilized. If bailout stenting was employed, though, the risk of MACE was 2.2-fold greater than if the cardiologist stuck with the plan.
Ninety-eight percent of patients in the P2BiTO registry received drug-eluting stents. The other 2% got the Absorb bioabsorbable vascular scaffold. The percutaneous coronary intervention access site, treatment strategy, choice of stent, and duration of dual-antiplatelet therapy were left up to the operator’s discretion.

The risk of MACE was reduced by 39% in patients on dual-antiplatelet therapy for 6-12 months, compared with less than 6 months.
Discussant Graham Cassel, MD, director of the heart transplant unit at Milpark Hospital in Johannesburg, commented, “The message comes through very clearly that, if you plan your procedure well, the chance of bailout is far less – and if you do have to bail out, the results are uniformly bad. If you can avoid putting in two or three stents, that’s beneficial.”
Dr. Zimarino reported having no financial conflicts of interest regarding his presentation.
AT EUROPCR
Key clinical point:
Major finding: Bailout stenting during PCI for coronary bifurcations doubles the risk of major adverse cardiovascular events.
Data source: The P2BiTO registry includes 4,306 patients who received one or more drug-eluting stents or bioabsorbable vascular scaffolds for treatment of coronary bifurcations.
Disclosures: The study presenter reported having no financial conflicts of interest.
Revascularization of CTOs improves health status more than medical therapy
PARIS – The first randomized clinical trial to evaluate quality of life and clinical symptoms as the primary efficacy outcome in patients with coronary chronic total occlusion (CTO) showed a clear advantage for percutaneous revascularization over optimal medical therapy.
At 12 months’ follow-up in the 26-site, 396-patient EuroCTO trial, patients randomized to PCI with drug-eluting stents had significantly less angina and physical limitations coupled with greater improvement in quality of life than the optimal medical therapy (OMT) group on subscales of the Seattle Angina Questionnaire. On the angina frequency subscale, for example, the PCI group improved from a mean baseline score of 77.2 to 91.4 at 12 months, a significantly better result than the OMT group improvement from 80.6 to 87.5.
The PCI group also experienced greater mobility, better activity status, and less pain and discomfort as assessed by the EuroQOL five dimensions questionnaire (EQ-5D), Gerald S. Werner, MD, reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
“PCI of a CTO should be considered as a primary option in symptomatic patients. It is a safe and effective treatment option in expert hands,” said Dr. Werner, director of cardiology and nonsurgical intensive care at the Darmstadt (Ger.) Clinic.
He emphasized the point about “expert hands,” drawing attention to the stellar 86.3% successful revascularization rate in the EuroCTO trial, even though these were often complex procedures. Indeed, in 36% of the CTO PCIs, a retrograde approach was used.
“CTO is a special field. Just like not everybody in every hospital will do a transcatheter aortic valve replacement, not everybody should do a CTO. It can be done safely and to the benefit of the patient, but it needs to be done by someone with expertise,” the cardiologist said.
Study participants were evenly split between those with single- and multivessel disease. Patients with additional nonocclusive disease had those lesions treated by PCI before the 2:1 randomization to CTO PCI or OMT.
The periprocedural complication rate was low at 2.9%, a figure that included a 1.5% incidence of pericardial tamponade as well as vascular repairs. There were no periprocedural MIs or deaths. The 1-year major adverse cardiac event rate was roughly 6% in both study arms.
The PCI group received 6-12 months of dual-antiplatelet therapy with clopidogrel and aspirin. So did roughly 40% of the OMT group because of prior PCI.
Both study arms had comparably high rates of guideline-directed medical therapy, including statins, beta-blockers, and ACE inhibitors. However, the PCI group made significantly less use of nitrates than the OMT group during follow-up, reflecting their greater reduction in angina frequency. The crossover rate from OMT to PCI because of ongoing angina was 7.3%.
The long-term safety and durability of the two treatment strategies will be assessed at 3 years of follow-up.
The EuroCTO trial was originally planned for 600 patients. The investigators eventually settled for less because of slow enrollment. Many interventionalists who are skilled in treating CTOs proved reluctant to randomize the patients.
Dr. Werner contrasted the positive EuroCTO findings regarding clinical symptoms and quality of life to the negative results of the Korean DECISION CTO trial presented at the 2017 meeting of the American College of Cardiology. DECISION CTO found that PCI plus OMT wasn’t superior to OMT alone in reducing MI and other major adverse cardiac events in patients with at least one CTO. In Dr. Werner’s view, the Korean investigators chose the wrong endpoint.
“The quality of life improvement we’ve shown after PCI in EuroCTO is a valid clinical goal in treating stable coronary artery disease. I don’t think we can aim at improving prognosis,” according to Dr. Werner.
The impetus for EuroCTO was a recognition that CTOs are common and seriously undertreated. CTOs account for 16%-18% of all coronary lesions in patients with stable coronary artery disease, yet U.S. national data indicate only 5% of PCIs are performed to treat CTOs.
The EuroCTO trial was sponsored by the Euro CTO Club and supported by research grants from Biosensors and Asahi.
PARIS – The first randomized clinical trial to evaluate quality of life and clinical symptoms as the primary efficacy outcome in patients with coronary chronic total occlusion (CTO) showed a clear advantage for percutaneous revascularization over optimal medical therapy.
At 12 months’ follow-up in the 26-site, 396-patient EuroCTO trial, patients randomized to PCI with drug-eluting stents had significantly less angina and physical limitations coupled with greater improvement in quality of life than the optimal medical therapy (OMT) group on subscales of the Seattle Angina Questionnaire. On the angina frequency subscale, for example, the PCI group improved from a mean baseline score of 77.2 to 91.4 at 12 months, a significantly better result than the OMT group improvement from 80.6 to 87.5.
The PCI group also experienced greater mobility, better activity status, and less pain and discomfort as assessed by the EuroQOL five dimensions questionnaire (EQ-5D), Gerald S. Werner, MD, reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
“PCI of a CTO should be considered as a primary option in symptomatic patients. It is a safe and effective treatment option in expert hands,” said Dr. Werner, director of cardiology and nonsurgical intensive care at the Darmstadt (Ger.) Clinic.
He emphasized the point about “expert hands,” drawing attention to the stellar 86.3% successful revascularization rate in the EuroCTO trial, even though these were often complex procedures. Indeed, in 36% of the CTO PCIs, a retrograde approach was used.
“CTO is a special field. Just like not everybody in every hospital will do a transcatheter aortic valve replacement, not everybody should do a CTO. It can be done safely and to the benefit of the patient, but it needs to be done by someone with expertise,” the cardiologist said.
Study participants were evenly split between those with single- and multivessel disease. Patients with additional nonocclusive disease had those lesions treated by PCI before the 2:1 randomization to CTO PCI or OMT.
The periprocedural complication rate was low at 2.9%, a figure that included a 1.5% incidence of pericardial tamponade as well as vascular repairs. There were no periprocedural MIs or deaths. The 1-year major adverse cardiac event rate was roughly 6% in both study arms.
The PCI group received 6-12 months of dual-antiplatelet therapy with clopidogrel and aspirin. So did roughly 40% of the OMT group because of prior PCI.
Both study arms had comparably high rates of guideline-directed medical therapy, including statins, beta-blockers, and ACE inhibitors. However, the PCI group made significantly less use of nitrates than the OMT group during follow-up, reflecting their greater reduction in angina frequency. The crossover rate from OMT to PCI because of ongoing angina was 7.3%.
The long-term safety and durability of the two treatment strategies will be assessed at 3 years of follow-up.
The EuroCTO trial was originally planned for 600 patients. The investigators eventually settled for less because of slow enrollment. Many interventionalists who are skilled in treating CTOs proved reluctant to randomize the patients.
Dr. Werner contrasted the positive EuroCTO findings regarding clinical symptoms and quality of life to the negative results of the Korean DECISION CTO trial presented at the 2017 meeting of the American College of Cardiology. DECISION CTO found that PCI plus OMT wasn’t superior to OMT alone in reducing MI and other major adverse cardiac events in patients with at least one CTO. In Dr. Werner’s view, the Korean investigators chose the wrong endpoint.
“The quality of life improvement we’ve shown after PCI in EuroCTO is a valid clinical goal in treating stable coronary artery disease. I don’t think we can aim at improving prognosis,” according to Dr. Werner.
The impetus for EuroCTO was a recognition that CTOs are common and seriously undertreated. CTOs account for 16%-18% of all coronary lesions in patients with stable coronary artery disease, yet U.S. national data indicate only 5% of PCIs are performed to treat CTOs.
The EuroCTO trial was sponsored by the Euro CTO Club and supported by research grants from Biosensors and Asahi.
PARIS – The first randomized clinical trial to evaluate quality of life and clinical symptoms as the primary efficacy outcome in patients with coronary chronic total occlusion (CTO) showed a clear advantage for percutaneous revascularization over optimal medical therapy.
At 12 months’ follow-up in the 26-site, 396-patient EuroCTO trial, patients randomized to PCI with drug-eluting stents had significantly less angina and physical limitations coupled with greater improvement in quality of life than the optimal medical therapy (OMT) group on subscales of the Seattle Angina Questionnaire. On the angina frequency subscale, for example, the PCI group improved from a mean baseline score of 77.2 to 91.4 at 12 months, a significantly better result than the OMT group improvement from 80.6 to 87.5.
The PCI group also experienced greater mobility, better activity status, and less pain and discomfort as assessed by the EuroQOL five dimensions questionnaire (EQ-5D), Gerald S. Werner, MD, reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
“PCI of a CTO should be considered as a primary option in symptomatic patients. It is a safe and effective treatment option in expert hands,” said Dr. Werner, director of cardiology and nonsurgical intensive care at the Darmstadt (Ger.) Clinic.
He emphasized the point about “expert hands,” drawing attention to the stellar 86.3% successful revascularization rate in the EuroCTO trial, even though these were often complex procedures. Indeed, in 36% of the CTO PCIs, a retrograde approach was used.
“CTO is a special field. Just like not everybody in every hospital will do a transcatheter aortic valve replacement, not everybody should do a CTO. It can be done safely and to the benefit of the patient, but it needs to be done by someone with expertise,” the cardiologist said.
Study participants were evenly split between those with single- and multivessel disease. Patients with additional nonocclusive disease had those lesions treated by PCI before the 2:1 randomization to CTO PCI or OMT.
The periprocedural complication rate was low at 2.9%, a figure that included a 1.5% incidence of pericardial tamponade as well as vascular repairs. There were no periprocedural MIs or deaths. The 1-year major adverse cardiac event rate was roughly 6% in both study arms.
The PCI group received 6-12 months of dual-antiplatelet therapy with clopidogrel and aspirin. So did roughly 40% of the OMT group because of prior PCI.
Both study arms had comparably high rates of guideline-directed medical therapy, including statins, beta-blockers, and ACE inhibitors. However, the PCI group made significantly less use of nitrates than the OMT group during follow-up, reflecting their greater reduction in angina frequency. The crossover rate from OMT to PCI because of ongoing angina was 7.3%.
The long-term safety and durability of the two treatment strategies will be assessed at 3 years of follow-up.
The EuroCTO trial was originally planned for 600 patients. The investigators eventually settled for less because of slow enrollment. Many interventionalists who are skilled in treating CTOs proved reluctant to randomize the patients.
Dr. Werner contrasted the positive EuroCTO findings regarding clinical symptoms and quality of life to the negative results of the Korean DECISION CTO trial presented at the 2017 meeting of the American College of Cardiology. DECISION CTO found that PCI plus OMT wasn’t superior to OMT alone in reducing MI and other major adverse cardiac events in patients with at least one CTO. In Dr. Werner’s view, the Korean investigators chose the wrong endpoint.
“The quality of life improvement we’ve shown after PCI in EuroCTO is a valid clinical goal in treating stable coronary artery disease. I don’t think we can aim at improving prognosis,” according to Dr. Werner.
The impetus for EuroCTO was a recognition that CTOs are common and seriously undertreated. CTOs account for 16%-18% of all coronary lesions in patients with stable coronary artery disease, yet U.S. national data indicate only 5% of PCIs are performed to treat CTOs.
The EuroCTO trial was sponsored by the Euro CTO Club and supported by research grants from Biosensors and Asahi.
AT EUROPCR
Key clinical point:
Major finding: Patients whose chronic total occlusions were treated by PCI rather than optimal medical therapy experienced significantly less angina and greater improvement in quality of life during 12 months of follow-up.
Data source: EuroCTO, a prospective, 26-site study in which 396 patients with coronary chronic total occlusions were randomized 2:1 to PCI or optimal medical therapy.
Disclosures: EuroCTO was sponsored by the Euro CTO Club and supported by research grants from Biosensors and Asahi.
Hidden CABG costs will disrupt bundled payment systems
COLORADO SPRINGS – With bundled payment models for coronary artery bypass graft surgery looming ahead, it’s vital that cardiac surgeons take a hard look at the procedure’s hidden costs – namely, the steep price tag for postoperative complications, James H. Mehaffey, MD, said at the annual meeting of the Western Thoracic Surgical Association.
He presented a retrospective study of the 30-day hospital costs for all 36,588 patients who underwent isolated CABG during 2006-2015 at the 19 Virginia centers where the surgery is performed. This was a typical CABG population, with an average predicted risk of mortality of 1.9%. The actual 30-day mortality was 0.6%, so the surgical performance was better than expected.
“The population of patients experiencing one or more major comorbidities demonstrated a significant and dramatic increase in total hospital costs. It was an exponential increase with each additional major morbidity,” reported Dr. Mehaffey of the University of Virginia, Charlottesville.
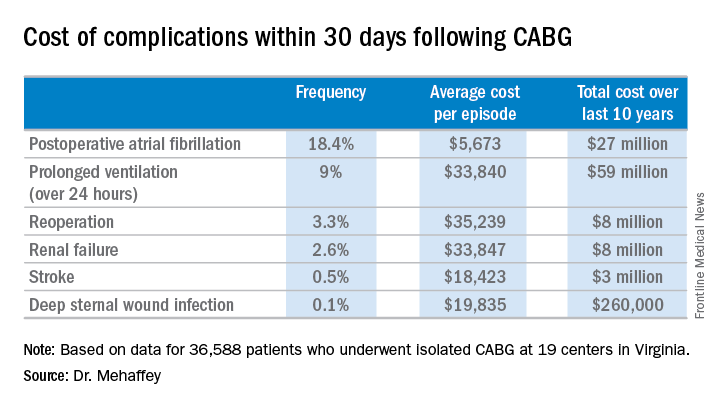
Indeed, the average cost jumped from $36,580 for uncomplicated surgery to $64,542 with one major complication, $111,239 with two, and $194,043 with three.
The two most frequent major complications were postoperative atrial fibrillation, which occurred in 18.4% of patients, and prolonged ventilation for longer than 24 hours, which occurred in 9%. Over the course of the decade-long study period, the 19 medical centers in the Virginia Cardiac Surgery Quality Initiative collectively spent roughly $59 million on prolonged ventilation and $27 million for postoperative atrial fibrillation.
The cost of CABG during the study years outpaced the CMS health care–specific inflation rate, and this escalating cost was driven primarily by postoperative complications.
For the Virginia cardiac surgery collaborative, these data on the cost of postoperative complications will be utilized to prioritize quality improvement projects.
For example, during the past decade, the Virginia collaborative made reduction in the rate of postoperative atrial fibrillation a priority. Toward that end, the collaborative developed a protocol for routine perioperative prophylactic amiodarone therapy.
“At the beginning of the study decade we had postoperative atrial fibrillation rates above 25%. The average for the entire decade was just over 18%, and in the last couple years we’ve been in the 15%-16% range. So I think we are moving the needle on this. We are making a meaningful impact,” Dr. Mehaffey said.
“We’ve already used the complication cost data to do a cost-effectiveness analysis of our prophylactic amiodarone innovation. We showed we saved an average of $250 per patient, even though we’re treating a bunch of patients who’d never get that complication,” he continued.
This sort of data on the cost of adverse events is also critical to accurately risk-adjust bundled payment models.
Discussant Richard J. Shemin, MD, asked if there was much variability in postoperative complication costs between the CABG centers in the Virginia collaborative.
The variability is enormous, Dr. Mehaffey replied. Investigators recently plugged the last 5 years worth of hospital cost and complication rate data into a proposed CABG bundled payment model and extrapolated what that would mean over the next 5 years.
“There were some institutions that would be positive by a couple million dollars from this payment system and some that were losing more than $20 million, just because of the cost variability,” said Dr. Mehaffey.
Dr. Shemin also noted that the Virginia collaborative was able to collect 30-day outcome data only through the STS database, yet the bundled payment programs are based on the 90-day postoperative experience.
“How do we capture the costs in that full 90 days that we’ll be responsible for?” asked Dr. Shemin, professor of surgery and codirector of the UCLA Cardiovascular Center.
Dr. Mehaffey said that’s indeed an important question, since a major complication such as stroke or deep sternal wound infection typically entails considerable long-term costs and repeated hospital admissions beyond the 30-day window. In Virginia, the cardiac surgery collaborative is working with payers to gain access to the 90 days worth of patient data.
He reported having no financial conflicts regarding his study.
COLORADO SPRINGS – With bundled payment models for coronary artery bypass graft surgery looming ahead, it’s vital that cardiac surgeons take a hard look at the procedure’s hidden costs – namely, the steep price tag for postoperative complications, James H. Mehaffey, MD, said at the annual meeting of the Western Thoracic Surgical Association.
He presented a retrospective study of the 30-day hospital costs for all 36,588 patients who underwent isolated CABG during 2006-2015 at the 19 Virginia centers where the surgery is performed. This was a typical CABG population, with an average predicted risk of mortality of 1.9%. The actual 30-day mortality was 0.6%, so the surgical performance was better than expected.
“The population of patients experiencing one or more major comorbidities demonstrated a significant and dramatic increase in total hospital costs. It was an exponential increase with each additional major morbidity,” reported Dr. Mehaffey of the University of Virginia, Charlottesville.
Indeed, the average cost jumped from $36,580 for uncomplicated surgery to $64,542 with one major complication, $111,239 with two, and $194,043 with three.
The two most frequent major complications were postoperative atrial fibrillation, which occurred in 18.4% of patients, and prolonged ventilation for longer than 24 hours, which occurred in 9%. Over the course of the decade-long study period, the 19 medical centers in the Virginia Cardiac Surgery Quality Initiative collectively spent roughly $59 million on prolonged ventilation and $27 million for postoperative atrial fibrillation.
The cost of CABG during the study years outpaced the CMS health care–specific inflation rate, and this escalating cost was driven primarily by postoperative complications.
For the Virginia cardiac surgery collaborative, these data on the cost of postoperative complications will be utilized to prioritize quality improvement projects.

For example, during the past decade, the Virginia collaborative made reduction in the rate of postoperative atrial fibrillation a priority. Toward that end, the collaborative developed a protocol for routine perioperative prophylactic amiodarone therapy.
“At the beginning of the study decade we had postoperative atrial fibrillation rates above 25%. The average for the entire decade was just over 18%, and in the last couple years we’ve been in the 15%-16% range. So I think we are moving the needle on this. We are making a meaningful impact,” Dr. Mehaffey said.
“We’ve already used the complication cost data to do a cost-effectiveness analysis of our prophylactic amiodarone innovation. We showed we saved an average of $250 per patient, even though we’re treating a bunch of patients who’d never get that complication,” he continued.
This sort of data on the cost of adverse events is also critical to accurately risk-adjust bundled payment models.
Discussant Richard J. Shemin, MD, asked if there was much variability in postoperative complication costs between the CABG centers in the Virginia collaborative.
The variability is enormous, Dr. Mehaffey replied. Investigators recently plugged the last 5 years worth of hospital cost and complication rate data into a proposed CABG bundled payment model and extrapolated what that would mean over the next 5 years.
“There were some institutions that would be positive by a couple million dollars from this payment system and some that were losing more than $20 million, just because of the cost variability,” said Dr. Mehaffey.
Dr. Shemin also noted that the Virginia collaborative was able to collect 30-day outcome data only through the STS database, yet the bundled payment programs are based on the 90-day postoperative experience.
“How do we capture the costs in that full 90 days that we’ll be responsible for?” asked Dr. Shemin, professor of surgery and codirector of the UCLA Cardiovascular Center.
Dr. Mehaffey said that’s indeed an important question, since a major complication such as stroke or deep sternal wound infection typically entails considerable long-term costs and repeated hospital admissions beyond the 30-day window. In Virginia, the cardiac surgery collaborative is working with payers to gain access to the 90 days worth of patient data.
He reported having no financial conflicts regarding his study.
COLORADO SPRINGS – With bundled payment models for coronary artery bypass graft surgery looming ahead, it’s vital that cardiac surgeons take a hard look at the procedure’s hidden costs – namely, the steep price tag for postoperative complications, James H. Mehaffey, MD, said at the annual meeting of the Western Thoracic Surgical Association.
He presented a retrospective study of the 30-day hospital costs for all 36,588 patients who underwent isolated CABG during 2006-2015 at the 19 Virginia centers where the surgery is performed. This was a typical CABG population, with an average predicted risk of mortality of 1.9%. The actual 30-day mortality was 0.6%, so the surgical performance was better than expected.
“The population of patients experiencing one or more major comorbidities demonstrated a significant and dramatic increase in total hospital costs. It was an exponential increase with each additional major morbidity,” reported Dr. Mehaffey of the University of Virginia, Charlottesville.
Indeed, the average cost jumped from $36,580 for uncomplicated surgery to $64,542 with one major complication, $111,239 with two, and $194,043 with three.
The two most frequent major complications were postoperative atrial fibrillation, which occurred in 18.4% of patients, and prolonged ventilation for longer than 24 hours, which occurred in 9%. Over the course of the decade-long study period, the 19 medical centers in the Virginia Cardiac Surgery Quality Initiative collectively spent roughly $59 million on prolonged ventilation and $27 million for postoperative atrial fibrillation.
The cost of CABG during the study years outpaced the CMS health care–specific inflation rate, and this escalating cost was driven primarily by postoperative complications.
For the Virginia cardiac surgery collaborative, these data on the cost of postoperative complications will be utilized to prioritize quality improvement projects.

For example, during the past decade, the Virginia collaborative made reduction in the rate of postoperative atrial fibrillation a priority. Toward that end, the collaborative developed a protocol for routine perioperative prophylactic amiodarone therapy.
“At the beginning of the study decade we had postoperative atrial fibrillation rates above 25%. The average for the entire decade was just over 18%, and in the last couple years we’ve been in the 15%-16% range. So I think we are moving the needle on this. We are making a meaningful impact,” Dr. Mehaffey said.
“We’ve already used the complication cost data to do a cost-effectiveness analysis of our prophylactic amiodarone innovation. We showed we saved an average of $250 per patient, even though we’re treating a bunch of patients who’d never get that complication,” he continued.
This sort of data on the cost of adverse events is also critical to accurately risk-adjust bundled payment models.
Discussant Richard J. Shemin, MD, asked if there was much variability in postoperative complication costs between the CABG centers in the Virginia collaborative.
The variability is enormous, Dr. Mehaffey replied. Investigators recently plugged the last 5 years worth of hospital cost and complication rate data into a proposed CABG bundled payment model and extrapolated what that would mean over the next 5 years.
“There were some institutions that would be positive by a couple million dollars from this payment system and some that were losing more than $20 million, just because of the cost variability,” said Dr. Mehaffey.
Dr. Shemin also noted that the Virginia collaborative was able to collect 30-day outcome data only through the STS database, yet the bundled payment programs are based on the 90-day postoperative experience.
“How do we capture the costs in that full 90 days that we’ll be responsible for?” asked Dr. Shemin, professor of surgery and codirector of the UCLA Cardiovascular Center.
Dr. Mehaffey said that’s indeed an important question, since a major complication such as stroke or deep sternal wound infection typically entails considerable long-term costs and repeated hospital admissions beyond the 30-day window. In Virginia, the cardiac surgery collaborative is working with payers to gain access to the 90 days worth of patient data.
He reported having no financial conflicts regarding his study.
AT THE WTSA ANNUAL MEETING
Key clinical point:
Major finding: The average 30-day total hospital cost of an isolated CABG procedure during 2006-2015 in Virginia was $36,580 if there were no postoperative complications, jumping to $64,542 with one major complication and $111,239 with two.
Data source: A retrospective study of the 30-day total hospital costs for all isolated CABG procedures performed in Virginia during 2006-2015.
Disclosures: The study presenter reported having no financial conflicts.
Thirdhand smoke shaping up as potential health hazard
DENVER – Thirdhand smoke – the persistent residue that collects on indoor surfaces where people have smoked – is “clearly” a potentially hazardous exposure, John M. Rogers, PhD, said at the annual meeting of the Teratology Society.
Everyone knows about the hazards of secondhand smoke, which have led to widespread bans on smoking in public spaces. Still, the Centers for Disease Control and Prevention estimates that 58 million nonsmokers in the United States are exposed to secondhand smoke on a regular basis. And where there is secondhand smoke, there is typically exposure to thirdhand smoke as well.
“If you walk into a hotel room you were told is a nonsmoking room and you take one breath and you know it’s not nonsmoking, that’s thirdhand smoke. Thirdhand smoke is all over the place where smokers have been,” explained Dr. Rogers, director of the toxicity assessment division at the Environmental Protection Agency in Research Triangle Park, N.C.
The main potential health risk is to young children, who ingest thirdhand smoke by the hand-to-mouth route and skin contact.
Thirdhand smoke is a much newer concept than secondhand smoke and has not yet actually been shown to pose a significant health risk. . But that is likely to change.
Thirdhand smoke has become an area of intensive research interest, with California leading the way. The Tobacco-Related Disease Research Program, a state agency funded by a tax on the sale of tobacco products, has created a research consortium on thirdhand smoke, with studies underway investigating thirdhand smoke’s precise chemical composition, cytotoxicity, genotoxicity, and true impact on public health (www.trdrp.org).
Concern regarding thirdhand smoke’s potential public health impact ramped up in response to a study in which investigators at the University of York, England, measured levels of various tobacco-specific nitrosamines, N-nitrosamines, and nicotine in house dust samples from the homes of smokers. The researchers estimated that years of early life exposure to these compounds at the levels they detected could result in one excess case of cancer per 1,000 exposed individuals (Environ Int. 2014 Oct;71:139-47).
In addition to his update on thirdhand smoke, Dr. Rogers also touched on other recent tobacco-related developments, including a determination by the Food and Drug Administration that there has been no decline in tobacco use in the last 5 years in adolescents and young adults. While cigarette smoking by young people decreased, this was offset by a large increase in the use of electronic cigarettes and a smaller rise in the use of hookah tobacco. Indeed, e-cigarette use is now about double that of cigarettes among youth.
Also of concern is evidence of a striking socioeconomic disparity in smoking prevalence: Low-education, low-income Americans have far higher tobacco use rates.
“That’s pretty alarming,” he said. “I think a lot of people in this audience probably don’t see a lot of smoking these days, but it’s still around.”
Dr. Rogers drew attention to updated evidence reviews on the reproductive and developmental effects of smoking contained in the U.S. Surgeon General’s voluminous 2014 report on the health consequences of smoking. The report concluded that there is now sufficient evidence to infer a causal relationship between maternal smoking in pregnancy, ectopic pregnancy, and orofacial clefts. The available evidence is “suggestive but not sufficient” to infer causality between maternal smoking in pregnancy and atrial septal defects, clubfoot, gastroschisis, and attention-deficit/hyperactivity disorder and other disruptive behavior disorders.
Dr. Rogers reported having no financial disclosures related to his presentation, which he noted did not necessarily reflect the views and policies of the EPA.
DENVER – Thirdhand smoke – the persistent residue that collects on indoor surfaces where people have smoked – is “clearly” a potentially hazardous exposure, John M. Rogers, PhD, said at the annual meeting of the Teratology Society.
Everyone knows about the hazards of secondhand smoke, which have led to widespread bans on smoking in public spaces. Still, the Centers for Disease Control and Prevention estimates that 58 million nonsmokers in the United States are exposed to secondhand smoke on a regular basis. And where there is secondhand smoke, there is typically exposure to thirdhand smoke as well.
“If you walk into a hotel room you were told is a nonsmoking room and you take one breath and you know it’s not nonsmoking, that’s thirdhand smoke. Thirdhand smoke is all over the place where smokers have been,” explained Dr. Rogers, director of the toxicity assessment division at the Environmental Protection Agency in Research Triangle Park, N.C.
The main potential health risk is to young children, who ingest thirdhand smoke by the hand-to-mouth route and skin contact.
Thirdhand smoke is a much newer concept than secondhand smoke and has not yet actually been shown to pose a significant health risk. . But that is likely to change.
Thirdhand smoke has become an area of intensive research interest, with California leading the way. The Tobacco-Related Disease Research Program, a state agency funded by a tax on the sale of tobacco products, has created a research consortium on thirdhand smoke, with studies underway investigating thirdhand smoke’s precise chemical composition, cytotoxicity, genotoxicity, and true impact on public health (www.trdrp.org).
Concern regarding thirdhand smoke’s potential public health impact ramped up in response to a study in which investigators at the University of York, England, measured levels of various tobacco-specific nitrosamines, N-nitrosamines, and nicotine in house dust samples from the homes of smokers. The researchers estimated that years of early life exposure to these compounds at the levels they detected could result in one excess case of cancer per 1,000 exposed individuals (Environ Int. 2014 Oct;71:139-47).
In addition to his update on thirdhand smoke, Dr. Rogers also touched on other recent tobacco-related developments, including a determination by the Food and Drug Administration that there has been no decline in tobacco use in the last 5 years in adolescents and young adults. While cigarette smoking by young people decreased, this was offset by a large increase in the use of electronic cigarettes and a smaller rise in the use of hookah tobacco. Indeed, e-cigarette use is now about double that of cigarettes among youth.
Also of concern is evidence of a striking socioeconomic disparity in smoking prevalence: Low-education, low-income Americans have far higher tobacco use rates.
“That’s pretty alarming,” he said. “I think a lot of people in this audience probably don’t see a lot of smoking these days, but it’s still around.”
Dr. Rogers drew attention to updated evidence reviews on the reproductive and developmental effects of smoking contained in the U.S. Surgeon General’s voluminous 2014 report on the health consequences of smoking. The report concluded that there is now sufficient evidence to infer a causal relationship between maternal smoking in pregnancy, ectopic pregnancy, and orofacial clefts. The available evidence is “suggestive but not sufficient” to infer causality between maternal smoking in pregnancy and atrial septal defects, clubfoot, gastroschisis, and attention-deficit/hyperactivity disorder and other disruptive behavior disorders.
Dr. Rogers reported having no financial disclosures related to his presentation, which he noted did not necessarily reflect the views and policies of the EPA.
DENVER – Thirdhand smoke – the persistent residue that collects on indoor surfaces where people have smoked – is “clearly” a potentially hazardous exposure, John M. Rogers, PhD, said at the annual meeting of the Teratology Society.
Everyone knows about the hazards of secondhand smoke, which have led to widespread bans on smoking in public spaces. Still, the Centers for Disease Control and Prevention estimates that 58 million nonsmokers in the United States are exposed to secondhand smoke on a regular basis. And where there is secondhand smoke, there is typically exposure to thirdhand smoke as well.
“If you walk into a hotel room you were told is a nonsmoking room and you take one breath and you know it’s not nonsmoking, that’s thirdhand smoke. Thirdhand smoke is all over the place where smokers have been,” explained Dr. Rogers, director of the toxicity assessment division at the Environmental Protection Agency in Research Triangle Park, N.C.
The main potential health risk is to young children, who ingest thirdhand smoke by the hand-to-mouth route and skin contact.
Thirdhand smoke is a much newer concept than secondhand smoke and has not yet actually been shown to pose a significant health risk. . But that is likely to change.
Thirdhand smoke has become an area of intensive research interest, with California leading the way. The Tobacco-Related Disease Research Program, a state agency funded by a tax on the sale of tobacco products, has created a research consortium on thirdhand smoke, with studies underway investigating thirdhand smoke’s precise chemical composition, cytotoxicity, genotoxicity, and true impact on public health (www.trdrp.org).
Concern regarding thirdhand smoke’s potential public health impact ramped up in response to a study in which investigators at the University of York, England, measured levels of various tobacco-specific nitrosamines, N-nitrosamines, and nicotine in house dust samples from the homes of smokers. The researchers estimated that years of early life exposure to these compounds at the levels they detected could result in one excess case of cancer per 1,000 exposed individuals (Environ Int. 2014 Oct;71:139-47).
In addition to his update on thirdhand smoke, Dr. Rogers also touched on other recent tobacco-related developments, including a determination by the Food and Drug Administration that there has been no decline in tobacco use in the last 5 years in adolescents and young adults. While cigarette smoking by young people decreased, this was offset by a large increase in the use of electronic cigarettes and a smaller rise in the use of hookah tobacco. Indeed, e-cigarette use is now about double that of cigarettes among youth.
Also of concern is evidence of a striking socioeconomic disparity in smoking prevalence: Low-education, low-income Americans have far higher tobacco use rates.
“That’s pretty alarming,” he said. “I think a lot of people in this audience probably don’t see a lot of smoking these days, but it’s still around.”
Dr. Rogers drew attention to updated evidence reviews on the reproductive and developmental effects of smoking contained in the U.S. Surgeon General’s voluminous 2014 report on the health consequences of smoking. The report concluded that there is now sufficient evidence to infer a causal relationship between maternal smoking in pregnancy, ectopic pregnancy, and orofacial clefts. The available evidence is “suggestive but not sufficient” to infer causality between maternal smoking in pregnancy and atrial septal defects, clubfoot, gastroschisis, and attention-deficit/hyperactivity disorder and other disruptive behavior disorders.
Dr. Rogers reported having no financial disclosures related to his presentation, which he noted did not necessarily reflect the views and policies of the EPA.
EXPERT ANALYSIS FROM TERATOLOGY SOCIETY 2017
Lessons emerge from Europe’s first enterovirus-related brain stem encephalitis outbreak
MADRID – Ninety-two percent of Spanish children sickened during the first-ever outbreak of enterovirus-associated brain stem encephalitis in western Europe survived with no long-term sequelae, Nuria Worner, MD, reported at the annual meeting of the European Society for Paediatric Infectious Diseases.
“We think that aggressive treatments should be restricted to those patients with important neurologic involvement,” declared Dr. Worner of Vall d’Hebron University Hospital in Barcelona. “We can say that no patients with milder involvement and without warning signs during the first 24 hours after onset of neurologic involvement went on to develop fulminant symptoms.”
Notable outbreaks of enterovirus A71 (EV-A71)-associated brain stem encephalitis occurred in Southeast Asia, Australia, and China in the late 1990s.
Dr. Worner reported on 196 children treated for laboratory-confirmed EV-A71–associated brain stem encephalitis at 16 Spanish hospitals in April-December 2016. Their median age was 25 months, 57% were male, and a median of 2 days of symptoms of mild viral illness transpired before neurologic symptoms arose. Prior to presenting to a hospital, 21% of the children had been diagnosed with hand-foot-and-mouth disease, and 13% with herpangina.
Initial preadmission symptoms included fever in 94% of cases, sleepiness in 86%, ataxia in 75%, tremor in 47%, myoclonus in 40%, and a rash in 26%.
Fifty-five percent of the children had EV RNA isolated from both throat and feces, 26% from the throat only, and 19% only from their feces. Eighty-seven percent of serotyped EV were EV-A71.
Ninety percent of children underwent lumbar puncture. Particularly noteworthy was the finding that EV was detected in the cerebrospinal fluid of a mere 3% of patients, although pleocytosis was present in 84%.
Brain MRI showed brain stem encephalitis along with myelitis in 50% of patients, brain stem myelitis without encephalitis in 29%, myelitis elsewhere in 2%, and normal findings in 19%.
Ground zero for the outbreak was Barcelona and the surrounding region of Catalonia; indeed, 130 of the 196 (66%) affected children came from there. The Catalan health department and pediatric infectious disease specialists quickly created standardized case severity definitions and treatment recommendations; they distributed them nationally.
Mild EV-A71–associated brain stem disease was defined as two or more of the following: tremor, myoclonus, mild ataxia, and/or significant drowsiness. The recommendation in these mild cases was for no treatment other than supportive care and careful in-hospital monitoring.
Patients with moderate involvement had to meet the definition for mild disease plus more pronounced ataxia or bulbar motor neuron involvement marked by slurred speech, drooling, dysphagia, apnea, abolition of the gag reflex, and/or an abnormal respiratory pattern. Moderately affected patients received two doses of intravenous immunoglobulin (IVIG), each dosed at 1 g/kg per 24 hours. Admission to the pediatric ICU was individualized for patients with moderate EV-A71–associated brain stem encephalitis.
Severe disease was categorized as bulbar motor neuron involvement plus neurogenic cardiorespiratory failure. Those patients were uniformly admitted to a pediatric ICU and given the two doses of IVIG. The need for systemic steroids was determined on an individual basis.
Forty percent of patients received IVIG and systemic steroids, 24% received IVIG only, 2% systemic steroids only, and 34% received no treatment other than supportive care.
Twenty-six percent of children were admitted to a pediatric ICU for a median stay of 3.5 days. Nine percent of children were placed on mechanical ventilation.
As the disease evolved, the most frequent neurologic complications included slurred speech in 15% of children, abnormal breathing pattern in 11%, seizures in 10%, acute flaccid paralysis in 9%, and cardiorespiratory failure with pulmonary edema in 9%, all occurring within the first hours after hospital admission.
The median hospital length of stay for the full study population was 6 days. The survival rate was 99.5%, with the sole death being due to cardiorespiratory failure.
With 1-6 months of follow-up since the acute episode of EV-A71–associated brain stem encephalitis, the long-term sequelae included two cases of limb paresis and two cases of paresis of a cranial nerve, one child with residual seizures, and one with hypoxic-ischemic encephalopathy.
Asked why the fatality rate in the Spanish outbreak was so much lower than in the earlier Australasian outbreaks, Dr. Worner cited Catalan physicians’ quick recognition of what was underway – and, more importantly, a difference in the EV-A71 viral subgenotype. Most of the most severe cases in Asia and Australia involved the C-4 subgenotype, while in Spain, the predominant subgenotype involved in the outbreak was C-1.
As for the curious finding that EV was detectable in the cerebrospinal fluid of a mere 3% of the Spanish children, she said the explanation is unknown. The two main possibilities are that the CNS symptoms were due to a parenchymal brain infection rather than to EV-A71 infection of meningeal tissue. Alternatively, the CNS involvement may have been a manifestation of an immunologic response to the infection, rather than being due to the virus itself.
Dr. Worner reported having no financial conflicts of interest.
MADRID – Ninety-two percent of Spanish children sickened during the first-ever outbreak of enterovirus-associated brain stem encephalitis in western Europe survived with no long-term sequelae, Nuria Worner, MD, reported at the annual meeting of the European Society for Paediatric Infectious Diseases.
“We think that aggressive treatments should be restricted to those patients with important neurologic involvement,” declared Dr. Worner of Vall d’Hebron University Hospital in Barcelona. “We can say that no patients with milder involvement and without warning signs during the first 24 hours after onset of neurologic involvement went on to develop fulminant symptoms.”
Notable outbreaks of enterovirus A71 (EV-A71)-associated brain stem encephalitis occurred in Southeast Asia, Australia, and China in the late 1990s.
Dr. Worner reported on 196 children treated for laboratory-confirmed EV-A71–associated brain stem encephalitis at 16 Spanish hospitals in April-December 2016. Their median age was 25 months, 57% were male, and a median of 2 days of symptoms of mild viral illness transpired before neurologic symptoms arose. Prior to presenting to a hospital, 21% of the children had been diagnosed with hand-foot-and-mouth disease, and 13% with herpangina.
Initial preadmission symptoms included fever in 94% of cases, sleepiness in 86%, ataxia in 75%, tremor in 47%, myoclonus in 40%, and a rash in 26%.
Fifty-five percent of the children had EV RNA isolated from both throat and feces, 26% from the throat only, and 19% only from their feces. Eighty-seven percent of serotyped EV were EV-A71.
Ninety percent of children underwent lumbar puncture. Particularly noteworthy was the finding that EV was detected in the cerebrospinal fluid of a mere 3% of patients, although pleocytosis was present in 84%.
Brain MRI showed brain stem encephalitis along with myelitis in 50% of patients, brain stem myelitis without encephalitis in 29%, myelitis elsewhere in 2%, and normal findings in 19%.
Ground zero for the outbreak was Barcelona and the surrounding region of Catalonia; indeed, 130 of the 196 (66%) affected children came from there. The Catalan health department and pediatric infectious disease specialists quickly created standardized case severity definitions and treatment recommendations; they distributed them nationally.
Mild EV-A71–associated brain stem disease was defined as two or more of the following: tremor, myoclonus, mild ataxia, and/or significant drowsiness. The recommendation in these mild cases was for no treatment other than supportive care and careful in-hospital monitoring.
Patients with moderate involvement had to meet the definition for mild disease plus more pronounced ataxia or bulbar motor neuron involvement marked by slurred speech, drooling, dysphagia, apnea, abolition of the gag reflex, and/or an abnormal respiratory pattern. Moderately affected patients received two doses of intravenous immunoglobulin (IVIG), each dosed at 1 g/kg per 24 hours. Admission to the pediatric ICU was individualized for patients with moderate EV-A71–associated brain stem encephalitis.
Severe disease was categorized as bulbar motor neuron involvement plus neurogenic cardiorespiratory failure. Those patients were uniformly admitted to a pediatric ICU and given the two doses of IVIG. The need for systemic steroids was determined on an individual basis.
Forty percent of patients received IVIG and systemic steroids, 24% received IVIG only, 2% systemic steroids only, and 34% received no treatment other than supportive care.
Twenty-six percent of children were admitted to a pediatric ICU for a median stay of 3.5 days. Nine percent of children were placed on mechanical ventilation.
As the disease evolved, the most frequent neurologic complications included slurred speech in 15% of children, abnormal breathing pattern in 11%, seizures in 10%, acute flaccid paralysis in 9%, and cardiorespiratory failure with pulmonary edema in 9%, all occurring within the first hours after hospital admission.
The median hospital length of stay for the full study population was 6 days. The survival rate was 99.5%, with the sole death being due to cardiorespiratory failure.
With 1-6 months of follow-up since the acute episode of EV-A71–associated brain stem encephalitis, the long-term sequelae included two cases of limb paresis and two cases of paresis of a cranial nerve, one child with residual seizures, and one with hypoxic-ischemic encephalopathy.
Asked why the fatality rate in the Spanish outbreak was so much lower than in the earlier Australasian outbreaks, Dr. Worner cited Catalan physicians’ quick recognition of what was underway – and, more importantly, a difference in the EV-A71 viral subgenotype. Most of the most severe cases in Asia and Australia involved the C-4 subgenotype, while in Spain, the predominant subgenotype involved in the outbreak was C-1.
As for the curious finding that EV was detectable in the cerebrospinal fluid of a mere 3% of the Spanish children, she said the explanation is unknown. The two main possibilities are that the CNS symptoms were due to a parenchymal brain infection rather than to EV-A71 infection of meningeal tissue. Alternatively, the CNS involvement may have been a manifestation of an immunologic response to the infection, rather than being due to the virus itself.
Dr. Worner reported having no financial conflicts of interest.
MADRID – Ninety-two percent of Spanish children sickened during the first-ever outbreak of enterovirus-associated brain stem encephalitis in western Europe survived with no long-term sequelae, Nuria Worner, MD, reported at the annual meeting of the European Society for Paediatric Infectious Diseases.
“We think that aggressive treatments should be restricted to those patients with important neurologic involvement,” declared Dr. Worner of Vall d’Hebron University Hospital in Barcelona. “We can say that no patients with milder involvement and without warning signs during the first 24 hours after onset of neurologic involvement went on to develop fulminant symptoms.”
Notable outbreaks of enterovirus A71 (EV-A71)-associated brain stem encephalitis occurred in Southeast Asia, Australia, and China in the late 1990s.
Dr. Worner reported on 196 children treated for laboratory-confirmed EV-A71–associated brain stem encephalitis at 16 Spanish hospitals in April-December 2016. Their median age was 25 months, 57% were male, and a median of 2 days of symptoms of mild viral illness transpired before neurologic symptoms arose. Prior to presenting to a hospital, 21% of the children had been diagnosed with hand-foot-and-mouth disease, and 13% with herpangina.
Initial preadmission symptoms included fever in 94% of cases, sleepiness in 86%, ataxia in 75%, tremor in 47%, myoclonus in 40%, and a rash in 26%.
Fifty-five percent of the children had EV RNA isolated from both throat and feces, 26% from the throat only, and 19% only from their feces. Eighty-seven percent of serotyped EV were EV-A71.
Ninety percent of children underwent lumbar puncture. Particularly noteworthy was the finding that EV was detected in the cerebrospinal fluid of a mere 3% of patients, although pleocytosis was present in 84%.
Brain MRI showed brain stem encephalitis along with myelitis in 50% of patients, brain stem myelitis without encephalitis in 29%, myelitis elsewhere in 2%, and normal findings in 19%.
Ground zero for the outbreak was Barcelona and the surrounding region of Catalonia; indeed, 130 of the 196 (66%) affected children came from there. The Catalan health department and pediatric infectious disease specialists quickly created standardized case severity definitions and treatment recommendations; they distributed them nationally.
Mild EV-A71–associated brain stem disease was defined as two or more of the following: tremor, myoclonus, mild ataxia, and/or significant drowsiness. The recommendation in these mild cases was for no treatment other than supportive care and careful in-hospital monitoring.
Patients with moderate involvement had to meet the definition for mild disease plus more pronounced ataxia or bulbar motor neuron involvement marked by slurred speech, drooling, dysphagia, apnea, abolition of the gag reflex, and/or an abnormal respiratory pattern. Moderately affected patients received two doses of intravenous immunoglobulin (IVIG), each dosed at 1 g/kg per 24 hours. Admission to the pediatric ICU was individualized for patients with moderate EV-A71–associated brain stem encephalitis.
Severe disease was categorized as bulbar motor neuron involvement plus neurogenic cardiorespiratory failure. Those patients were uniformly admitted to a pediatric ICU and given the two doses of IVIG. The need for systemic steroids was determined on an individual basis.
Forty percent of patients received IVIG and systemic steroids, 24% received IVIG only, 2% systemic steroids only, and 34% received no treatment other than supportive care.
Twenty-six percent of children were admitted to a pediatric ICU for a median stay of 3.5 days. Nine percent of children were placed on mechanical ventilation.
As the disease evolved, the most frequent neurologic complications included slurred speech in 15% of children, abnormal breathing pattern in 11%, seizures in 10%, acute flaccid paralysis in 9%, and cardiorespiratory failure with pulmonary edema in 9%, all occurring within the first hours after hospital admission.
The median hospital length of stay for the full study population was 6 days. The survival rate was 99.5%, with the sole death being due to cardiorespiratory failure.
With 1-6 months of follow-up since the acute episode of EV-A71–associated brain stem encephalitis, the long-term sequelae included two cases of limb paresis and two cases of paresis of a cranial nerve, one child with residual seizures, and one with hypoxic-ischemic encephalopathy.
Asked why the fatality rate in the Spanish outbreak was so much lower than in the earlier Australasian outbreaks, Dr. Worner cited Catalan physicians’ quick recognition of what was underway – and, more importantly, a difference in the EV-A71 viral subgenotype. Most of the most severe cases in Asia and Australia involved the C-4 subgenotype, while in Spain, the predominant subgenotype involved in the outbreak was C-1.
As for the curious finding that EV was detectable in the cerebrospinal fluid of a mere 3% of the Spanish children, she said the explanation is unknown. The two main possibilities are that the CNS symptoms were due to a parenchymal brain infection rather than to EV-A71 infection of meningeal tissue. Alternatively, the CNS involvement may have been a manifestation of an immunologic response to the infection, rather than being due to the virus itself.
Dr. Worner reported having no financial conflicts of interest.
AT ESPID 2017
Key clinical point:
Major finding: Ninety-two percent of Spanish children involved in an outbreak of enterovirus-associated brain stem encephalitis survived with no long-term sequelae.
Data source: A retrospective review of 196 children treated for laboratory-confirmed EV-A71–associated brain stem encephalitis at 16 Spanish hospitals in April-December 2016 during the first-ever outbreak of this condition in western Europe.
Disclosures: Dr. Worner reported having no financial conflicts of interest.
Is that thyroid nodule malignant?
ESTES PARK, COLO. – Before ordering any tests at all, an astute clinician can have a pretty good idea as to whether a palpable thyroid nodule is at higher or lower risk for being malignant based upon the patient’s history and physical examination, Michael T. McDermott, MD, said at a conference on internal medicine sponsored by the University of Colorado.
“The possibility of cancer is what scares people, and that’s what people want to know,” observed Dr. McDermott, professor of medicine and director of endocrinology and diabetes practice at University of Colorado Hospital.
This is a common clinical problem.
“If you felt the neck of every patient that you saw, 4%-7% would have a palpable thyroid nodule,” the endocrinologist said.
Moreover, upon imaging the thyroid by ultrasound, CT, or MRI, the prevalence of a thyroid nodule is close to 40%. And because the prevalence increases with age, thyroid nodules are found at autopsy in 50%-60% of individuals.
Ninety percent of thyroid nodules are benign. And even when they are malignant, for the most part the prognosis is very good. Ten-year survival is in excess of 90% in patients with papillary, follicular, or Hurthle-cell thyroid cancer and greater than 60% in the 2%-5% of thyroid cancers classified as medullary. The one bad actor is anaplastic thyroid cancer, with a 1-year survival rate of less than 5%; however, only 2%-5% of thyroid cancers are of the anaplastic type.
The cancer risk of a palpable thyroid nodule is increased if it is 3 cm or larger in size, of a firm and fixed consistency, or cervical lymph nodes are palpable. Clues of increased risk from the patient history include a family history of papillary or medullary thyroid cancer, compressive symptoms such as a mass sensation or difficulty swallowing, or radiation therapy to the head and neck before age 20 years for treatment of acne, ringworm, or for tonsil/adenoid shrinkage, a fairly common practice in the 1950s and 1960s.
The first test to order in evaluating a thyroid nodule is always a serum TSH. If the level is low or undetectable, that’s a relief: It is not cancer. A mildly elevated free T4 and total T3 in conjunction with the low TSH indicate an autonomously functioning nodule.
In this situation, the next test to order is a radioactive iodine uptake and scan.
“Every nuclear medicine department in the country does these,” Dr. McDermott observed.
A normal radioactive iodine uptake value is 20%-40%; however, in the setting of suppressed serum TSH, there is no physiologic stimulation for uptake, so any uptake would be considered inappropriately high and constitutes confirmation that this is a patient with hyperthyroidism.
If the serum TSH is normal or high, there is no need to order a free T4 and total T3. Instead, the next test to order is a neck ultrasound; the size and features of the nodule upon imaging will determine the need for fine needle aspiration as a next step in the evaluation. Ultrasound findings suggestive of malignancy are a dark, hypoechoic nodule with irregular margins, microcalcifications, and/or increased blood flow upon Doppler imaging.
“Patterns and combinations of findings are more informative than any single finding,” he said.
Fine needle aspiration pathology results are reported as benign, malignant, or indeterminate. If the report is benign, there is still about a 2.5% chance of malignancy due to sampling error.
Molecular markers have a role in the 30% or more of fine needle aspiration results labeled indeterminate based upon a finding of atypia of unknown significance, a follicular lesion of unknown significance, or a follicular neoplasm. The Afirma gene expression classifier (Veracyte) has a high negative predictive value because it identifies patterns of mRNA expression consistent with benign tumors. The ThyroSeq oncogene panel (Interspace Diagnostics) has a moderate negative predictive value but a high positive predictive value. The tests each run about $1,200, but they spare patients with indeterminate aspiration biopsy findings from additional invasive diagnostic procedures – and the tests are covered by insurers.
“The gene expression classifier is a good test when you’re trying to avoid surgery, and the oncogene panel tells you when you really need surgery,” Dr. McDermott explained.
He recommends as a thyroid screen palpating the thyroid as part of a routine annual physical exam and ordering a serum TSH every 5 years starting at age 50. He is opposed to proactive ultrasound imaging of the thyroid as a means of screening for thyroid cancer because that practice picks up a huge number of nonharmful nodules.
Dr. McDermott cited the current American Thyroid Association guidelines on the evaluation and management of thyroid nodules as “one of the most important recent developments in the field.” The guidelines include detailed evidence-based recommendations regarding initial evaluation, criteria for fine-needle aspiration biopsy, interpretation of the biopsy results, use of molecular markers, and management of benign thyroid nodules. The guidelines also address the initial management of thyroid cancer and surveillance for recurrent disease.
Dr. McDermott reported having no financial conflicts regarding his presentation.
ESTES PARK, COLO. – Before ordering any tests at all, an astute clinician can have a pretty good idea as to whether a palpable thyroid nodule is at higher or lower risk for being malignant based upon the patient’s history and physical examination, Michael T. McDermott, MD, said at a conference on internal medicine sponsored by the University of Colorado.
“The possibility of cancer is what scares people, and that’s what people want to know,” observed Dr. McDermott, professor of medicine and director of endocrinology and diabetes practice at University of Colorado Hospital.
This is a common clinical problem.
“If you felt the neck of every patient that you saw, 4%-7% would have a palpable thyroid nodule,” the endocrinologist said.
Moreover, upon imaging the thyroid by ultrasound, CT, or MRI, the prevalence of a thyroid nodule is close to 40%. And because the prevalence increases with age, thyroid nodules are found at autopsy in 50%-60% of individuals.
Ninety percent of thyroid nodules are benign. And even when they are malignant, for the most part the prognosis is very good. Ten-year survival is in excess of 90% in patients with papillary, follicular, or Hurthle-cell thyroid cancer and greater than 60% in the 2%-5% of thyroid cancers classified as medullary. The one bad actor is anaplastic thyroid cancer, with a 1-year survival rate of less than 5%; however, only 2%-5% of thyroid cancers are of the anaplastic type.
The cancer risk of a palpable thyroid nodule is increased if it is 3 cm or larger in size, of a firm and fixed consistency, or cervical lymph nodes are palpable. Clues of increased risk from the patient history include a family history of papillary or medullary thyroid cancer, compressive symptoms such as a mass sensation or difficulty swallowing, or radiation therapy to the head and neck before age 20 years for treatment of acne, ringworm, or for tonsil/adenoid shrinkage, a fairly common practice in the 1950s and 1960s.
The first test to order in evaluating a thyroid nodule is always a serum TSH. If the level is low or undetectable, that’s a relief: It is not cancer. A mildly elevated free T4 and total T3 in conjunction with the low TSH indicate an autonomously functioning nodule.
In this situation, the next test to order is a radioactive iodine uptake and scan.
“Every nuclear medicine department in the country does these,” Dr. McDermott observed.
A normal radioactive iodine uptake value is 20%-40%; however, in the setting of suppressed serum TSH, there is no physiologic stimulation for uptake, so any uptake would be considered inappropriately high and constitutes confirmation that this is a patient with hyperthyroidism.
If the serum TSH is normal or high, there is no need to order a free T4 and total T3. Instead, the next test to order is a neck ultrasound; the size and features of the nodule upon imaging will determine the need for fine needle aspiration as a next step in the evaluation. Ultrasound findings suggestive of malignancy are a dark, hypoechoic nodule with irregular margins, microcalcifications, and/or increased blood flow upon Doppler imaging.
“Patterns and combinations of findings are more informative than any single finding,” he said.
Fine needle aspiration pathology results are reported as benign, malignant, or indeterminate. If the report is benign, there is still about a 2.5% chance of malignancy due to sampling error.
Molecular markers have a role in the 30% or more of fine needle aspiration results labeled indeterminate based upon a finding of atypia of unknown significance, a follicular lesion of unknown significance, or a follicular neoplasm. The Afirma gene expression classifier (Veracyte) has a high negative predictive value because it identifies patterns of mRNA expression consistent with benign tumors. The ThyroSeq oncogene panel (Interspace Diagnostics) has a moderate negative predictive value but a high positive predictive value. The tests each run about $1,200, but they spare patients with indeterminate aspiration biopsy findings from additional invasive diagnostic procedures – and the tests are covered by insurers.
“The gene expression classifier is a good test when you’re trying to avoid surgery, and the oncogene panel tells you when you really need surgery,” Dr. McDermott explained.
He recommends as a thyroid screen palpating the thyroid as part of a routine annual physical exam and ordering a serum TSH every 5 years starting at age 50. He is opposed to proactive ultrasound imaging of the thyroid as a means of screening for thyroid cancer because that practice picks up a huge number of nonharmful nodules.
Dr. McDermott cited the current American Thyroid Association guidelines on the evaluation and management of thyroid nodules as “one of the most important recent developments in the field.” The guidelines include detailed evidence-based recommendations regarding initial evaluation, criteria for fine-needle aspiration biopsy, interpretation of the biopsy results, use of molecular markers, and management of benign thyroid nodules. The guidelines also address the initial management of thyroid cancer and surveillance for recurrent disease.
Dr. McDermott reported having no financial conflicts regarding his presentation.
ESTES PARK, COLO. – Before ordering any tests at all, an astute clinician can have a pretty good idea as to whether a palpable thyroid nodule is at higher or lower risk for being malignant based upon the patient’s history and physical examination, Michael T. McDermott, MD, said at a conference on internal medicine sponsored by the University of Colorado.
“The possibility of cancer is what scares people, and that’s what people want to know,” observed Dr. McDermott, professor of medicine and director of endocrinology and diabetes practice at University of Colorado Hospital.
This is a common clinical problem.
“If you felt the neck of every patient that you saw, 4%-7% would have a palpable thyroid nodule,” the endocrinologist said.
Moreover, upon imaging the thyroid by ultrasound, CT, or MRI, the prevalence of a thyroid nodule is close to 40%. And because the prevalence increases with age, thyroid nodules are found at autopsy in 50%-60% of individuals.
Ninety percent of thyroid nodules are benign. And even when they are malignant, for the most part the prognosis is very good. Ten-year survival is in excess of 90% in patients with papillary, follicular, or Hurthle-cell thyroid cancer and greater than 60% in the 2%-5% of thyroid cancers classified as medullary. The one bad actor is anaplastic thyroid cancer, with a 1-year survival rate of less than 5%; however, only 2%-5% of thyroid cancers are of the anaplastic type.
The cancer risk of a palpable thyroid nodule is increased if it is 3 cm or larger in size, of a firm and fixed consistency, or cervical lymph nodes are palpable. Clues of increased risk from the patient history include a family history of papillary or medullary thyroid cancer, compressive symptoms such as a mass sensation or difficulty swallowing, or radiation therapy to the head and neck before age 20 years for treatment of acne, ringworm, or for tonsil/adenoid shrinkage, a fairly common practice in the 1950s and 1960s.
The first test to order in evaluating a thyroid nodule is always a serum TSH. If the level is low or undetectable, that’s a relief: It is not cancer. A mildly elevated free T4 and total T3 in conjunction with the low TSH indicate an autonomously functioning nodule.
In this situation, the next test to order is a radioactive iodine uptake and scan.
“Every nuclear medicine department in the country does these,” Dr. McDermott observed.
A normal radioactive iodine uptake value is 20%-40%; however, in the setting of suppressed serum TSH, there is no physiologic stimulation for uptake, so any uptake would be considered inappropriately high and constitutes confirmation that this is a patient with hyperthyroidism.
If the serum TSH is normal or high, there is no need to order a free T4 and total T3. Instead, the next test to order is a neck ultrasound; the size and features of the nodule upon imaging will determine the need for fine needle aspiration as a next step in the evaluation. Ultrasound findings suggestive of malignancy are a dark, hypoechoic nodule with irregular margins, microcalcifications, and/or increased blood flow upon Doppler imaging.
“Patterns and combinations of findings are more informative than any single finding,” he said.
Fine needle aspiration pathology results are reported as benign, malignant, or indeterminate. If the report is benign, there is still about a 2.5% chance of malignancy due to sampling error.
Molecular markers have a role in the 30% or more of fine needle aspiration results labeled indeterminate based upon a finding of atypia of unknown significance, a follicular lesion of unknown significance, or a follicular neoplasm. The Afirma gene expression classifier (Veracyte) has a high negative predictive value because it identifies patterns of mRNA expression consistent with benign tumors. The ThyroSeq oncogene panel (Interspace Diagnostics) has a moderate negative predictive value but a high positive predictive value. The tests each run about $1,200, but they spare patients with indeterminate aspiration biopsy findings from additional invasive diagnostic procedures – and the tests are covered by insurers.
“The gene expression classifier is a good test when you’re trying to avoid surgery, and the oncogene panel tells you when you really need surgery,” Dr. McDermott explained.
He recommends as a thyroid screen palpating the thyroid as part of a routine annual physical exam and ordering a serum TSH every 5 years starting at age 50. He is opposed to proactive ultrasound imaging of the thyroid as a means of screening for thyroid cancer because that practice picks up a huge number of nonharmful nodules.
Dr. McDermott cited the current American Thyroid Association guidelines on the evaluation and management of thyroid nodules as “one of the most important recent developments in the field.” The guidelines include detailed evidence-based recommendations regarding initial evaluation, criteria for fine-needle aspiration biopsy, interpretation of the biopsy results, use of molecular markers, and management of benign thyroid nodules. The guidelines also address the initial management of thyroid cancer and surveillance for recurrent disease.
Dr. McDermott reported having no financial conflicts regarding his presentation.
EXPERT ANALYSIS FROM THE ANNUAL INTERNAL MEDICINE PROGRAM
Meningitis before age 3 months: Consider enterovirus and parechovirus
MADRID – Ninety-five percent of cases of enterovirus and parechovirus meningitis in infants younger than 90 days old in the United Kingdom and Ireland were diagnosed through lumbar puncture and identification of the virus in the CSF by PCR, Seilesh Kadambari, MD, reported at the annual meeting of the European Society for Paediatric Infectious Diseases.
“We recommend that routine testing of CSF for enterovirus and parechovirus in febrile infants should be promoted, even in the absence of pleocytosis,” said Dr. Kadambari of John Radcliffe Hospital in Oxford, England.
This active enhanced prospective surveillance study was undertaken because an earlier retrospective study concluded that the rate of viral meningitis across all pediatric and adult age groups had increased rapidly during a recent 10-year period in the United Kingdom and Ireland. Infants younger than 3 months of age were especially hard hit by enterovirus, which accounted in the earlier study for 92% of all viral meningitis cases in that age group. Dr. Kadambari and his coinvestigators decided to take a closer prospective look at the sub-3-month age group because it’s such an important time neurodevelopmentally.
During the 13-month period of June 2014 to June 2015, 710 patients younger than 90 days old were hospitalized for enterovirus or parechovirus meningitis across the United Kingdom and Republic of Ireland. Ninety-five percent were due to enterovirus. Only 6% of affected infants were born prematurely.
“One of the take-home messages for me from the study was that 12% of enterovirus cases and 23% of parechovirus meningitis cases required admission to an ICU setting,” the pediatrician observed.
Among the infants admitted to a pediatric ICU, half of those with enterovirus meningitis and all those with parechovirus meningitis required intubation and mechanical ventilation. One-fifth of the enterovirus meningitis patients in pediatric ICUs required inotropic support for cardiovascular stabilization, as did all young infants with parechovirus meningitis.
Among the 710 patients, the three most common clinical presenting features were fever, irritability, and reduced feeding, present in 85%, 66%, and 54%, respectively.
Upon physical examination, two noteworthy common findings were signs of shock, present in 27% of infants with enterovirus meningitis and 43% with parechovirus meningitis, and respiratory distress, seen in 12% and 26%, respectively.
None of the 710 infants had a secondary bacterial infection. “That has implications for antimicrobial stewardship programs,” according to Dr. Kadambari.
The majority of infants had a normal CSF WBC count and a normal-range C-reactive protein level.
“Raised inflammatory markers were not a common feature in our cohort,” he noted.
Two infants died: one of a massive pulmonary hemorrhage and the other as a result of septic shock. Of the remaining 708 patients, however, 699 (98.7%) were discharged without significant neurologic impairment. Seven infants with enterovirus meningitis and two with parechovirus meningitis were discharged with severe delay, including cases of fine motor and gross motor delay, visual abnormalities, and a single case of severe cardiac dysfunction requiring discharge on an ACE inhibitor.
This surveillance study was a collaboration between St. George’s University of London and Public Health England. Dr. Kadambari reported having no financial conflicts.
Future studies need to examine long-term neurodevelopmental outcomes in affected young infants out to age 24 months, he said.
MADRID – Ninety-five percent of cases of enterovirus and parechovirus meningitis in infants younger than 90 days old in the United Kingdom and Ireland were diagnosed through lumbar puncture and identification of the virus in the CSF by PCR, Seilesh Kadambari, MD, reported at the annual meeting of the European Society for Paediatric Infectious Diseases.
“We recommend that routine testing of CSF for enterovirus and parechovirus in febrile infants should be promoted, even in the absence of pleocytosis,” said Dr. Kadambari of John Radcliffe Hospital in Oxford, England.
This active enhanced prospective surveillance study was undertaken because an earlier retrospective study concluded that the rate of viral meningitis across all pediatric and adult age groups had increased rapidly during a recent 10-year period in the United Kingdom and Ireland. Infants younger than 3 months of age were especially hard hit by enterovirus, which accounted in the earlier study for 92% of all viral meningitis cases in that age group. Dr. Kadambari and his coinvestigators decided to take a closer prospective look at the sub-3-month age group because it’s such an important time neurodevelopmentally.
During the 13-month period of June 2014 to June 2015, 710 patients younger than 90 days old were hospitalized for enterovirus or parechovirus meningitis across the United Kingdom and Republic of Ireland. Ninety-five percent were due to enterovirus. Only 6% of affected infants were born prematurely.
“One of the take-home messages for me from the study was that 12% of enterovirus cases and 23% of parechovirus meningitis cases required admission to an ICU setting,” the pediatrician observed.
Among the infants admitted to a pediatric ICU, half of those with enterovirus meningitis and all those with parechovirus meningitis required intubation and mechanical ventilation. One-fifth of the enterovirus meningitis patients in pediatric ICUs required inotropic support for cardiovascular stabilization, as did all young infants with parechovirus meningitis.
Among the 710 patients, the three most common clinical presenting features were fever, irritability, and reduced feeding, present in 85%, 66%, and 54%, respectively.
Upon physical examination, two noteworthy common findings were signs of shock, present in 27% of infants with enterovirus meningitis and 43% with parechovirus meningitis, and respiratory distress, seen in 12% and 26%, respectively.
None of the 710 infants had a secondary bacterial infection. “That has implications for antimicrobial stewardship programs,” according to Dr. Kadambari.
The majority of infants had a normal CSF WBC count and a normal-range C-reactive protein level.
“Raised inflammatory markers were not a common feature in our cohort,” he noted.
Two infants died: one of a massive pulmonary hemorrhage and the other as a result of septic shock. Of the remaining 708 patients, however, 699 (98.7%) were discharged without significant neurologic impairment. Seven infants with enterovirus meningitis and two with parechovirus meningitis were discharged with severe delay, including cases of fine motor and gross motor delay, visual abnormalities, and a single case of severe cardiac dysfunction requiring discharge on an ACE inhibitor.
This surveillance study was a collaboration between St. George’s University of London and Public Health England. Dr. Kadambari reported having no financial conflicts.
Future studies need to examine long-term neurodevelopmental outcomes in affected young infants out to age 24 months, he said.
MADRID – Ninety-five percent of cases of enterovirus and parechovirus meningitis in infants younger than 90 days old in the United Kingdom and Ireland were diagnosed through lumbar puncture and identification of the virus in the CSF by PCR, Seilesh Kadambari, MD, reported at the annual meeting of the European Society for Paediatric Infectious Diseases.
“We recommend that routine testing of CSF for enterovirus and parechovirus in febrile infants should be promoted, even in the absence of pleocytosis,” said Dr. Kadambari of John Radcliffe Hospital in Oxford, England.
This active enhanced prospective surveillance study was undertaken because an earlier retrospective study concluded that the rate of viral meningitis across all pediatric and adult age groups had increased rapidly during a recent 10-year period in the United Kingdom and Ireland. Infants younger than 3 months of age were especially hard hit by enterovirus, which accounted in the earlier study for 92% of all viral meningitis cases in that age group. Dr. Kadambari and his coinvestigators decided to take a closer prospective look at the sub-3-month age group because it’s such an important time neurodevelopmentally.
During the 13-month period of June 2014 to June 2015, 710 patients younger than 90 days old were hospitalized for enterovirus or parechovirus meningitis across the United Kingdom and Republic of Ireland. Ninety-five percent were due to enterovirus. Only 6% of affected infants were born prematurely.
“One of the take-home messages for me from the study was that 12% of enterovirus cases and 23% of parechovirus meningitis cases required admission to an ICU setting,” the pediatrician observed.
Among the infants admitted to a pediatric ICU, half of those with enterovirus meningitis and all those with parechovirus meningitis required intubation and mechanical ventilation. One-fifth of the enterovirus meningitis patients in pediatric ICUs required inotropic support for cardiovascular stabilization, as did all young infants with parechovirus meningitis.
Among the 710 patients, the three most common clinical presenting features were fever, irritability, and reduced feeding, present in 85%, 66%, and 54%, respectively.
Upon physical examination, two noteworthy common findings were signs of shock, present in 27% of infants with enterovirus meningitis and 43% with parechovirus meningitis, and respiratory distress, seen in 12% and 26%, respectively.
None of the 710 infants had a secondary bacterial infection. “That has implications for antimicrobial stewardship programs,” according to Dr. Kadambari.
The majority of infants had a normal CSF WBC count and a normal-range C-reactive protein level.
“Raised inflammatory markers were not a common feature in our cohort,” he noted.
Two infants died: one of a massive pulmonary hemorrhage and the other as a result of septic shock. Of the remaining 708 patients, however, 699 (98.7%) were discharged without significant neurologic impairment. Seven infants with enterovirus meningitis and two with parechovirus meningitis were discharged with severe delay, including cases of fine motor and gross motor delay, visual abnormalities, and a single case of severe cardiac dysfunction requiring discharge on an ACE inhibitor.
This surveillance study was a collaboration between St. George’s University of London and Public Health England. Dr. Kadambari reported having no financial conflicts.
Future studies need to examine long-term neurodevelopmental outcomes in affected young infants out to age 24 months, he said.
AT ESPID 2017
Key clinical point:
Major finding: More than 98% of infants in the United Kingdom and Ireland younger than 90 days old with enterovirus or parechovirus meningitis were discharged without significant neurologic impairment.
Data source: A prospective active enhanced surveillance study including all 710 infants younger than 90 days old hospitalized for enterovirus or parechovirus meningitis in the United Kingdom and Ireland in a 13-month period.
Disclosures: The study was jointly sponsored by Public Health England and St. George’s University of London. The presenter reported having no financial conflicts.
Novel drug-eluting coronary stent looks good in DESSOLVE III
PARIS – A unique drug-eluting coronary stent showed positive interim results in the DESSOLVE III trial presented at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
In DESSOLVE III, 1,398 patients undergoing percutaneous coronary intervention were randomized to the widely used Xience everolimus-eluting stent or to MiStent, a novel thin-strut stent with a polymer coating that is quickly absorbed after delivering microcrystalline sirolimus into the vessel wall for prolonged release at a near-linear rate.
At an interim analysis at 12 months of follow-up, the primary endpoint – a composite of cardiac death, target vessel MI, and clinically indicated target lesion revascularization – had occurred in 5.8% of the MiStent group and 6.5% of Xience recipients in the study, which was designed as a noninferiority trial, reported Robbert J. de Winter, MD, professor of clinical cardiology at the Academic Medical Center in Amsterdam.
“The data support the hypothesis that long-term cytostatic inhibition of early neointima could prevent the late neointimal growth seen at medium and long term with conventional drug-eluting stents,” he said.
MiStent is designed to overcome a shortcoming of conventional drug-eluting stents: namely, their durable polymer coating sticks around after the cytostatic drug is finished being released. It is believed that this residual polymer, which may not disappear for 6-9 months, induces inflammation in the vessel wall, eventually leading to intimal growth, restenosis, and new atherosclerosis.
“The unique feature of the MiStent is that the polymer is bioabsorbable and is fully absorbed at 3 months, whereas the drug is present in the vessel wall out to 9 months, well after the coating has disappeared. So in theory you would expect that any inflammatory response caused by the polymer coating will be counteracted by the drug. This was seen in animal models. And in the DESSOLVE I and II studies, angiographic follow-up showed that late luminal loss was flat after 6 months, in contrast to other drug-eluting stents, which show accrual of late neointimal growth after 6 months to a year,” according to the cardiologist.
DESSOLVE III is planned as a 3-year study. Already by 6 months the curves for target lesion revascularization started to separate, with a 12-month rate of 2.6% in the MiStent group versus 3.8% for Xience. And while this difference is not yet statistically significant, Dr. de Winter and his coinvestigators expect that by 3 years the separation will have grown to the point that the difference becomes statistically significant and clinically meaningful.
The MiStent platform is composed of a cobalt/chromium alloy. The stent strut thickness is only 64 microns, in contrast to 81 microns for the Xience stent. Thinner stent struts have previously been shown to be less injurious to the vessel wall.
DESSOLVE III is an all-comers trial conducted at 20 sites in four European countries. Participants had to have a reference vessel diameter of 2.5-3.5 mm. Roughly 60% of patients had an acute coronary syndrome as their indication for PCI. The study population included, among others, patients with left main coronary artery lesions, restenotic lesions, and failed saphenous vein grafts. Dual-antiplatelet therapy was given for 6 months in patients with stable angina and 12 months for those with ACS, in accordance with European Society of Cardiology guidelines.
Discussant Robert A. Byrne, MD, of the German Heart Center in Munich, declared: “For me, this is a potentially interesting device. It’s the only device where we have a drug elution that’s more prolonged than the polymer, and this does offer the potential for later benefit.”
Dr. Byrne was a member of a European Commission–backed task force that developed European regulatory guidance for the evaluation of new coronary stents. “This MiStent trial program ticks off a lot of boxes: We had a successful first human use study, then we had a modest-size angiographic endpoint study where the late lumen loss looked good, and now we have a clinical endpoint study. This is what we want to see in the regulatory space.”
Another discussant, Chaim Lotan, MD, pronounced the MiStent “definitely another good stent on the shelf.”
It’s impossible to say whether the excellent 1-year outcomes seen with MiStent in DESSOLVE III were due to the prolonged-release microcrystalline sirolimus, the ultrathin stent struts, or both. In any case, the major adverse cardiovascular event rates seen in DESSOLVE III are so low by historical standards that it will become extremely difficult to show superiority for one contemporary drug-eluting stent over another, predicted Dr. Lotan of Hadassah Medical Center in Jerusalem.
Dr. de Winter concurred.
“I think we can now say that the benchmark for present day drug-eluting stents is a target lesion failure rate of about 6% at 12 months and a stent thrombosis rate below 1% at 12 months. It’s going to be increasingly more difficult to improve on that,” he said.
The MiStent, manufactured by Micell Technologies, is commercially available in Europe but investigational in the United States.
DESSOLVE III was sponsored by the European Cardiovascular Research Institute and supported by grants from Micell Technologies and Stentys.
Dr. de Winter reported receiving research grants from OrbusNeich, Abbott Vascular, AstraZeneca, Stentys, and Tryton.
PARIS – A unique drug-eluting coronary stent showed positive interim results in the DESSOLVE III trial presented at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
In DESSOLVE III, 1,398 patients undergoing percutaneous coronary intervention were randomized to the widely used Xience everolimus-eluting stent or to MiStent, a novel thin-strut stent with a polymer coating that is quickly absorbed after delivering microcrystalline sirolimus into the vessel wall for prolonged release at a near-linear rate.
At an interim analysis at 12 months of follow-up, the primary endpoint – a composite of cardiac death, target vessel MI, and clinically indicated target lesion revascularization – had occurred in 5.8% of the MiStent group and 6.5% of Xience recipients in the study, which was designed as a noninferiority trial, reported Robbert J. de Winter, MD, professor of clinical cardiology at the Academic Medical Center in Amsterdam.
“The data support the hypothesis that long-term cytostatic inhibition of early neointima could prevent the late neointimal growth seen at medium and long term with conventional drug-eluting stents,” he said.
MiStent is designed to overcome a shortcoming of conventional drug-eluting stents: namely, their durable polymer coating sticks around after the cytostatic drug is finished being released. It is believed that this residual polymer, which may not disappear for 6-9 months, induces inflammation in the vessel wall, eventually leading to intimal growth, restenosis, and new atherosclerosis.
“The unique feature of the MiStent is that the polymer is bioabsorbable and is fully absorbed at 3 months, whereas the drug is present in the vessel wall out to 9 months, well after the coating has disappeared. So in theory you would expect that any inflammatory response caused by the polymer coating will be counteracted by the drug. This was seen in animal models. And in the DESSOLVE I and II studies, angiographic follow-up showed that late luminal loss was flat after 6 months, in contrast to other drug-eluting stents, which show accrual of late neointimal growth after 6 months to a year,” according to the cardiologist.
DESSOLVE III is planned as a 3-year study. Already by 6 months the curves for target lesion revascularization started to separate, with a 12-month rate of 2.6% in the MiStent group versus 3.8% for Xience. And while this difference is not yet statistically significant, Dr. de Winter and his coinvestigators expect that by 3 years the separation will have grown to the point that the difference becomes statistically significant and clinically meaningful.
The MiStent platform is composed of a cobalt/chromium alloy. The stent strut thickness is only 64 microns, in contrast to 81 microns for the Xience stent. Thinner stent struts have previously been shown to be less injurious to the vessel wall.
DESSOLVE III is an all-comers trial conducted at 20 sites in four European countries. Participants had to have a reference vessel diameter of 2.5-3.5 mm. Roughly 60% of patients had an acute coronary syndrome as their indication for PCI. The study population included, among others, patients with left main coronary artery lesions, restenotic lesions, and failed saphenous vein grafts. Dual-antiplatelet therapy was given for 6 months in patients with stable angina and 12 months for those with ACS, in accordance with European Society of Cardiology guidelines.
Discussant Robert A. Byrne, MD, of the German Heart Center in Munich, declared: “For me, this is a potentially interesting device. It’s the only device where we have a drug elution that’s more prolonged than the polymer, and this does offer the potential for later benefit.”
Dr. Byrne was a member of a European Commission–backed task force that developed European regulatory guidance for the evaluation of new coronary stents. “This MiStent trial program ticks off a lot of boxes: We had a successful first human use study, then we had a modest-size angiographic endpoint study where the late lumen loss looked good, and now we have a clinical endpoint study. This is what we want to see in the regulatory space.”
Another discussant, Chaim Lotan, MD, pronounced the MiStent “definitely another good stent on the shelf.”
It’s impossible to say whether the excellent 1-year outcomes seen with MiStent in DESSOLVE III were due to the prolonged-release microcrystalline sirolimus, the ultrathin stent struts, or both. In any case, the major adverse cardiovascular event rates seen in DESSOLVE III are so low by historical standards that it will become extremely difficult to show superiority for one contemporary drug-eluting stent over another, predicted Dr. Lotan of Hadassah Medical Center in Jerusalem.
Dr. de Winter concurred.
“I think we can now say that the benchmark for present day drug-eluting stents is a target lesion failure rate of about 6% at 12 months and a stent thrombosis rate below 1% at 12 months. It’s going to be increasingly more difficult to improve on that,” he said.
The MiStent, manufactured by Micell Technologies, is commercially available in Europe but investigational in the United States.
DESSOLVE III was sponsored by the European Cardiovascular Research Institute and supported by grants from Micell Technologies and Stentys.
Dr. de Winter reported receiving research grants from OrbusNeich, Abbott Vascular, AstraZeneca, Stentys, and Tryton.
PARIS – A unique drug-eluting coronary stent showed positive interim results in the DESSOLVE III trial presented at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
In DESSOLVE III, 1,398 patients undergoing percutaneous coronary intervention were randomized to the widely used Xience everolimus-eluting stent or to MiStent, a novel thin-strut stent with a polymer coating that is quickly absorbed after delivering microcrystalline sirolimus into the vessel wall for prolonged release at a near-linear rate.
At an interim analysis at 12 months of follow-up, the primary endpoint – a composite of cardiac death, target vessel MI, and clinically indicated target lesion revascularization – had occurred in 5.8% of the MiStent group and 6.5% of Xience recipients in the study, which was designed as a noninferiority trial, reported Robbert J. de Winter, MD, professor of clinical cardiology at the Academic Medical Center in Amsterdam.
“The data support the hypothesis that long-term cytostatic inhibition of early neointima could prevent the late neointimal growth seen at medium and long term with conventional drug-eluting stents,” he said.
MiStent is designed to overcome a shortcoming of conventional drug-eluting stents: namely, their durable polymer coating sticks around after the cytostatic drug is finished being released. It is believed that this residual polymer, which may not disappear for 6-9 months, induces inflammation in the vessel wall, eventually leading to intimal growth, restenosis, and new atherosclerosis.
“The unique feature of the MiStent is that the polymer is bioabsorbable and is fully absorbed at 3 months, whereas the drug is present in the vessel wall out to 9 months, well after the coating has disappeared. So in theory you would expect that any inflammatory response caused by the polymer coating will be counteracted by the drug. This was seen in animal models. And in the DESSOLVE I and II studies, angiographic follow-up showed that late luminal loss was flat after 6 months, in contrast to other drug-eluting stents, which show accrual of late neointimal growth after 6 months to a year,” according to the cardiologist.
DESSOLVE III is planned as a 3-year study. Already by 6 months the curves for target lesion revascularization started to separate, with a 12-month rate of 2.6% in the MiStent group versus 3.8% for Xience. And while this difference is not yet statistically significant, Dr. de Winter and his coinvestigators expect that by 3 years the separation will have grown to the point that the difference becomes statistically significant and clinically meaningful.
The MiStent platform is composed of a cobalt/chromium alloy. The stent strut thickness is only 64 microns, in contrast to 81 microns for the Xience stent. Thinner stent struts have previously been shown to be less injurious to the vessel wall.
DESSOLVE III is an all-comers trial conducted at 20 sites in four European countries. Participants had to have a reference vessel diameter of 2.5-3.5 mm. Roughly 60% of patients had an acute coronary syndrome as their indication for PCI. The study population included, among others, patients with left main coronary artery lesions, restenotic lesions, and failed saphenous vein grafts. Dual-antiplatelet therapy was given for 6 months in patients with stable angina and 12 months for those with ACS, in accordance with European Society of Cardiology guidelines.
Discussant Robert A. Byrne, MD, of the German Heart Center in Munich, declared: “For me, this is a potentially interesting device. It’s the only device where we have a drug elution that’s more prolonged than the polymer, and this does offer the potential for later benefit.”
Dr. Byrne was a member of a European Commission–backed task force that developed European regulatory guidance for the evaluation of new coronary stents. “This MiStent trial program ticks off a lot of boxes: We had a successful first human use study, then we had a modest-size angiographic endpoint study where the late lumen loss looked good, and now we have a clinical endpoint study. This is what we want to see in the regulatory space.”
Another discussant, Chaim Lotan, MD, pronounced the MiStent “definitely another good stent on the shelf.”
It’s impossible to say whether the excellent 1-year outcomes seen with MiStent in DESSOLVE III were due to the prolonged-release microcrystalline sirolimus, the ultrathin stent struts, or both. In any case, the major adverse cardiovascular event rates seen in DESSOLVE III are so low by historical standards that it will become extremely difficult to show superiority for one contemporary drug-eluting stent over another, predicted Dr. Lotan of Hadassah Medical Center in Jerusalem.
Dr. de Winter concurred.
“I think we can now say that the benchmark for present day drug-eluting stents is a target lesion failure rate of about 6% at 12 months and a stent thrombosis rate below 1% at 12 months. It’s going to be increasingly more difficult to improve on that,” he said.
The MiStent, manufactured by Micell Technologies, is commercially available in Europe but investigational in the United States.
DESSOLVE III was sponsored by the European Cardiovascular Research Institute and supported by grants from Micell Technologies and Stentys.
Dr. de Winter reported receiving research grants from OrbusNeich, Abbott Vascular, AstraZeneca, Stentys, and Tryton.
AT EUROPCR
Key clinical point:
Major finding: At an interim 12-month analysis, the composite rate of cardiac death, target vessel MI, and clinically indicated target lesion revascularization was 6.5% in recipients of a Xience everolimus-eluting coronary stent and 5.8% in those randomized to the novel MiStent.
Data source: DESSOLVE III, a prospective, randomized, international trial that randomized 1,398 real-world-type all-comers undergoing PCI to the Xience device or the MiStent.
Disclosures: DESSOLVE III was sponsored by the European Cardiovascular Research Institute and supported by grants from Micell Technologies and Stentys. The presenter reported receiving research grants from both companies.
















