User login
Maternal antepartum depression creates bevy of long-term risks in offspring
PARIS – Maternal depression during pregnancy is a common occurrence that can have far-reaching effects in the offspring, according to Tiina Taka-Eilola, MD, of the University of Oulu (Finland).
Indeed, maternal antepartum depression may best be thought of as an adverse environmental factor that exacerbates the impact of any underlying genetic vulnerability to severe mental disorder that may be present in the offspring, she said at the annual congress of the European College of Neuropsychopharmacology.

At midgestation, back in the mid-1960s, 13.9% of the mothers of the Northern Finland Birth Cohort acknowledged feeling “depressed” or “very depressed,” rates consistent with those reported in other studies using standardized depression assessment instruments. Their offspring, by age 43 years, were 1.6-fold more likely to have a history of a current or past nonpsychotic mood disorder and 2-fold more likely to have had a psychotic mood disorder than did the offspring of mothers free of antepartum depression.
These risks were greatly amplified if either parent experienced a hospital-treated severe mental disorder before, during, or up to 18 years after the pregnancy. Offspring who had both a mother who experienced antepartum depression and a parent with a severe, hospital-treated mental disorder were at 3.9-fold increased risk for being diagnosed with nonpsychotic depression by age 43 years in an analysis adjusted for sex, perinatal complications, and other potential confounders. They also were at 5.6-fold increased risk for psychotic depression and a whopping 7.8-fold greater risk of bipolar disorder than offspring with neither risk factor.
Moreover, in an earlier study, among men in the Northern Finland cohort who were assessed at age 33 years, investigators found that maternal depression during pregnancy was associated with an adjusted 1.4-fold increased likelihood of having a criminal record for a nonviolent offense, a 1.6-fold increased risk of violent crime, and a 1.7-fold increase in violent recidivism. In contrast, women whose mothers were depressed during pregnancy didn’t have a significantly higher rate of criminality, compared with those whose mothers weren’t depressed (J Affect Disord. 2003 May;74[3]:273-8).
In another earlier analysis, Dr. Taka-Eilola’s senior coinvestigators demonstrated that the risk of schizophrenia in the Northern Finland offspring was 2.6-fold greater if there was parental psychosis but no maternal antepartum depression than if neither was present, while the risk was 9.4-fold higher when both risk factors were present (Am J Psychiatry. 2010 Jan;167[1]:70-7).
Dr. Taka-Eilola, a primary care physician, said that postpartum depression garners news headlines and is far more extensively researched than is antepartum depression, but as the Finnish data show, antepartum depression is at least as common and deserves to be taken seriously. It’s important to screen for it and to treat it in an effort to prevent adverse effects in the offspring, as well as out of concern for the mother’s well-being, she emphasized. She believes this is now more likely to happen as a consequence of a recent World Psychiatric Association report calling for greater clinician attention to perinatal mental health.
Dr. Taka-Eilola reported having no financial conflicts of interest regarding the Northern Finland Birth Cohort Study, which is supported by the Academy of Finland, the Finnish Cultural Foundation Lapland Regional Fund, and grants from various nonprofit foundations.
PARIS – Maternal depression during pregnancy is a common occurrence that can have far-reaching effects in the offspring, according to Tiina Taka-Eilola, MD, of the University of Oulu (Finland).
Indeed, maternal antepartum depression may best be thought of as an adverse environmental factor that exacerbates the impact of any underlying genetic vulnerability to severe mental disorder that may be present in the offspring, she said at the annual congress of the European College of Neuropsychopharmacology.

At midgestation, back in the mid-1960s, 13.9% of the mothers of the Northern Finland Birth Cohort acknowledged feeling “depressed” or “very depressed,” rates consistent with those reported in other studies using standardized depression assessment instruments. Their offspring, by age 43 years, were 1.6-fold more likely to have a history of a current or past nonpsychotic mood disorder and 2-fold more likely to have had a psychotic mood disorder than did the offspring of mothers free of antepartum depression.
These risks were greatly amplified if either parent experienced a hospital-treated severe mental disorder before, during, or up to 18 years after the pregnancy. Offspring who had both a mother who experienced antepartum depression and a parent with a severe, hospital-treated mental disorder were at 3.9-fold increased risk for being diagnosed with nonpsychotic depression by age 43 years in an analysis adjusted for sex, perinatal complications, and other potential confounders. They also were at 5.6-fold increased risk for psychotic depression and a whopping 7.8-fold greater risk of bipolar disorder than offspring with neither risk factor.
Moreover, in an earlier study, among men in the Northern Finland cohort who were assessed at age 33 years, investigators found that maternal depression during pregnancy was associated with an adjusted 1.4-fold increased likelihood of having a criminal record for a nonviolent offense, a 1.6-fold increased risk of violent crime, and a 1.7-fold increase in violent recidivism. In contrast, women whose mothers were depressed during pregnancy didn’t have a significantly higher rate of criminality, compared with those whose mothers weren’t depressed (J Affect Disord. 2003 May;74[3]:273-8).
In another earlier analysis, Dr. Taka-Eilola’s senior coinvestigators demonstrated that the risk of schizophrenia in the Northern Finland offspring was 2.6-fold greater if there was parental psychosis but no maternal antepartum depression than if neither was present, while the risk was 9.4-fold higher when both risk factors were present (Am J Psychiatry. 2010 Jan;167[1]:70-7).
Dr. Taka-Eilola, a primary care physician, said that postpartum depression garners news headlines and is far more extensively researched than is antepartum depression, but as the Finnish data show, antepartum depression is at least as common and deserves to be taken seriously. It’s important to screen for it and to treat it in an effort to prevent adverse effects in the offspring, as well as out of concern for the mother’s well-being, she emphasized. She believes this is now more likely to happen as a consequence of a recent World Psychiatric Association report calling for greater clinician attention to perinatal mental health.
Dr. Taka-Eilola reported having no financial conflicts of interest regarding the Northern Finland Birth Cohort Study, which is supported by the Academy of Finland, the Finnish Cultural Foundation Lapland Regional Fund, and grants from various nonprofit foundations.
PARIS – Maternal depression during pregnancy is a common occurrence that can have far-reaching effects in the offspring, according to Tiina Taka-Eilola, MD, of the University of Oulu (Finland).
Indeed, maternal antepartum depression may best be thought of as an adverse environmental factor that exacerbates the impact of any underlying genetic vulnerability to severe mental disorder that may be present in the offspring, she said at the annual congress of the European College of Neuropsychopharmacology.

At midgestation, back in the mid-1960s, 13.9% of the mothers of the Northern Finland Birth Cohort acknowledged feeling “depressed” or “very depressed,” rates consistent with those reported in other studies using standardized depression assessment instruments. Their offspring, by age 43 years, were 1.6-fold more likely to have a history of a current or past nonpsychotic mood disorder and 2-fold more likely to have had a psychotic mood disorder than did the offspring of mothers free of antepartum depression.
These risks were greatly amplified if either parent experienced a hospital-treated severe mental disorder before, during, or up to 18 years after the pregnancy. Offspring who had both a mother who experienced antepartum depression and a parent with a severe, hospital-treated mental disorder were at 3.9-fold increased risk for being diagnosed with nonpsychotic depression by age 43 years in an analysis adjusted for sex, perinatal complications, and other potential confounders. They also were at 5.6-fold increased risk for psychotic depression and a whopping 7.8-fold greater risk of bipolar disorder than offspring with neither risk factor.
Moreover, in an earlier study, among men in the Northern Finland cohort who were assessed at age 33 years, investigators found that maternal depression during pregnancy was associated with an adjusted 1.4-fold increased likelihood of having a criminal record for a nonviolent offense, a 1.6-fold increased risk of violent crime, and a 1.7-fold increase in violent recidivism. In contrast, women whose mothers were depressed during pregnancy didn’t have a significantly higher rate of criminality, compared with those whose mothers weren’t depressed (J Affect Disord. 2003 May;74[3]:273-8).
In another earlier analysis, Dr. Taka-Eilola’s senior coinvestigators demonstrated that the risk of schizophrenia in the Northern Finland offspring was 2.6-fold greater if there was parental psychosis but no maternal antepartum depression than if neither was present, while the risk was 9.4-fold higher when both risk factors were present (Am J Psychiatry. 2010 Jan;167[1]:70-7).
Dr. Taka-Eilola, a primary care physician, said that postpartum depression garners news headlines and is far more extensively researched than is antepartum depression, but as the Finnish data show, antepartum depression is at least as common and deserves to be taken seriously. It’s important to screen for it and to treat it in an effort to prevent adverse effects in the offspring, as well as out of concern for the mother’s well-being, she emphasized. She believes this is now more likely to happen as a consequence of a recent World Psychiatric Association report calling for greater clinician attention to perinatal mental health.
Dr. Taka-Eilola reported having no financial conflicts of interest regarding the Northern Finland Birth Cohort Study, which is supported by the Academy of Finland, the Finnish Cultural Foundation Lapland Regional Fund, and grants from various nonprofit foundations.
AT THE ECNP CONGRESS
Key clinical point:
Major finding: By midlife, offspring of an mother who had antepartum depression and a parent with a severe, hospital-treated mental disorder were at an adjusted 5.6-fold increased risk for psychotic depression and 7.8-fold greater risk of bipolar disorder than the offspring with neither risk factor.
Data source: The Northern Finland Birth Cohort 1966 is an ongoing, observational, prospective, general population-based study of the 12,058 individuals born in the two northernmost provinces of Finland during 1966.
Disclosures: The presenter reported having no financial conflicts of interest regarding the study, which is supported by the Academy of Finland, the Finnish Cultural Foundation Lapland Regional Fund, and grants from various nonprofit foundations.
Evenamide impresses for schizophrenia
PARIS – The oral selective glutamate inhibitor evenamide is moving on to advanced clinical trials on the strength of positive results in a phase 2 study, Ravi Anand, MD, reported at the annual congress of the European College of Neuropsychopharmacology.
Evenamide is a highly selective inhibitor of voltage-gated sodium channels that also attenuates glutamate release by hyperexcited neurons, explained Dr. Anand, chief medical officer at Newron Pharmaceuticals, a company based in Bresso, Italy, that is developing this novel antipsychotic agent.
The 4-week, randomized, double-blind, placebo-controlled, multicenter study included 89 patients with schizophrenia of a mean 18 years’ duration. All participants showed baseline evidence of breakthrough psychosis despite being on stable therapeutic doses of previously effective risperidone or aripiprazole. They were randomized either to add-on oral evenamide at a starting dose of 15 mg twice daily titrated to 20 or 25 mg twice daily or to placebo.
As a phase 2 study, the primary focus was on safety and tolerability. Evenamide showed no dose-limiting toxicities. Nor did those in the evenamide group experience any of the common dopaminergic side effects seen with currently available antipsychotics, such as sedation, weight gain, sexual dysfunction, cardiac abnormalities, or extrapyramidal symptoms.
The only adverse events more common in the evenamide-treated group than in placebo-treated controls were insomnia and headache, which occurred in 5 and 3 of the 50 patients on evenamide, respectively.
In terms of efficacy, 75% of the evenamide group showed improvement over baseline in terms of the Positive and Negative Syndrome Scale, compared with 44% of controls. From a mean baseline total PANSS score of 62.7, the evenamide group experienced a 5.1-point reduction at day 28, compared with a 3.7-point improvement in controls. The difference between the two study arms was already significant at the first assessment, which took place on day 8.
Furthermore, 55% of patients in the evenamide group showed significant improvement on the Clinical Global Impression Severity score, compared with 36% of controls.
Because evenamide’s efficacy was greater in younger patients, the next round of larger, longer clinical trials will focus on younger patients with more severe symptoms, Dr. Anand said.
PARIS – The oral selective glutamate inhibitor evenamide is moving on to advanced clinical trials on the strength of positive results in a phase 2 study, Ravi Anand, MD, reported at the annual congress of the European College of Neuropsychopharmacology.
Evenamide is a highly selective inhibitor of voltage-gated sodium channels that also attenuates glutamate release by hyperexcited neurons, explained Dr. Anand, chief medical officer at Newron Pharmaceuticals, a company based in Bresso, Italy, that is developing this novel antipsychotic agent.
The 4-week, randomized, double-blind, placebo-controlled, multicenter study included 89 patients with schizophrenia of a mean 18 years’ duration. All participants showed baseline evidence of breakthrough psychosis despite being on stable therapeutic doses of previously effective risperidone or aripiprazole. They were randomized either to add-on oral evenamide at a starting dose of 15 mg twice daily titrated to 20 or 25 mg twice daily or to placebo.
As a phase 2 study, the primary focus was on safety and tolerability. Evenamide showed no dose-limiting toxicities. Nor did those in the evenamide group experience any of the common dopaminergic side effects seen with currently available antipsychotics, such as sedation, weight gain, sexual dysfunction, cardiac abnormalities, or extrapyramidal symptoms.
The only adverse events more common in the evenamide-treated group than in placebo-treated controls were insomnia and headache, which occurred in 5 and 3 of the 50 patients on evenamide, respectively.
In terms of efficacy, 75% of the evenamide group showed improvement over baseline in terms of the Positive and Negative Syndrome Scale, compared with 44% of controls. From a mean baseline total PANSS score of 62.7, the evenamide group experienced a 5.1-point reduction at day 28, compared with a 3.7-point improvement in controls. The difference between the two study arms was already significant at the first assessment, which took place on day 8.
Furthermore, 55% of patients in the evenamide group showed significant improvement on the Clinical Global Impression Severity score, compared with 36% of controls.
Because evenamide’s efficacy was greater in younger patients, the next round of larger, longer clinical trials will focus on younger patients with more severe symptoms, Dr. Anand said.
PARIS – The oral selective glutamate inhibitor evenamide is moving on to advanced clinical trials on the strength of positive results in a phase 2 study, Ravi Anand, MD, reported at the annual congress of the European College of Neuropsychopharmacology.
Evenamide is a highly selective inhibitor of voltage-gated sodium channels that also attenuates glutamate release by hyperexcited neurons, explained Dr. Anand, chief medical officer at Newron Pharmaceuticals, a company based in Bresso, Italy, that is developing this novel antipsychotic agent.
The 4-week, randomized, double-blind, placebo-controlled, multicenter study included 89 patients with schizophrenia of a mean 18 years’ duration. All participants showed baseline evidence of breakthrough psychosis despite being on stable therapeutic doses of previously effective risperidone or aripiprazole. They were randomized either to add-on oral evenamide at a starting dose of 15 mg twice daily titrated to 20 or 25 mg twice daily or to placebo.
As a phase 2 study, the primary focus was on safety and tolerability. Evenamide showed no dose-limiting toxicities. Nor did those in the evenamide group experience any of the common dopaminergic side effects seen with currently available antipsychotics, such as sedation, weight gain, sexual dysfunction, cardiac abnormalities, or extrapyramidal symptoms.
The only adverse events more common in the evenamide-treated group than in placebo-treated controls were insomnia and headache, which occurred in 5 and 3 of the 50 patients on evenamide, respectively.
In terms of efficacy, 75% of the evenamide group showed improvement over baseline in terms of the Positive and Negative Syndrome Scale, compared with 44% of controls. From a mean baseline total PANSS score of 62.7, the evenamide group experienced a 5.1-point reduction at day 28, compared with a 3.7-point improvement in controls. The difference between the two study arms was already significant at the first assessment, which took place on day 8.
Furthermore, 55% of patients in the evenamide group showed significant improvement on the Clinical Global Impression Severity score, compared with 36% of controls.
Because evenamide’s efficacy was greater in younger patients, the next round of larger, longer clinical trials will focus on younger patients with more severe symptoms, Dr. Anand said.
AT THE ECNP CONGRESS
Key clinical point:
Major finding: Three-quarters of schizophrenia patients on the oral glutamate modulator evenamide showed improvement over baseline in terms of the Positive and Negative Syndrome Scale, compared with 44% of controls.
Data source: This 4-week, randomized, double-blind, placebo-controlled, multicenter study included 89 patients with schizophrenia.
Disclosures: The study presenter is the chief medical officer at Newron Pharmaceuticals, which is developing the novel antipsychotic.
Oral anticoagulation ‘reasonable’ in advanced kidney disease with A-fib
BARCELONA – Oral anticoagulation had a net overall benefit for patients with atrial fibrillation and advanced chronic kidney disease, based on results of a large observational study reported at the annual congress of the European Society of Cardiology.
The novel direct-acting oral anticoagulants (NOACs) and warfarin were all similarly effective in this study of 39,241 patients who had stage 4 or 5 chronic kidney disease (CKD), atrial fibrillation, and were not on dialysis. Compared with no oral anticoagulation, the drugs cut in half the risk of stroke or systemic embolism, with no increased risk of major bleeding.
“In patients with advanced CKD, it appears that OACs [oral anticoagulants] are reasonable,” concluded Peter A. Noseworthy, MD, of the Mayo Clinic in Rochester, Minn.
This is a potentially practice-changing finding given the “striking underutilization” of OACs in advanced CKD, he noted. Indeed, only one-third of the patients in this study were prescribed an OAC and picked up their prescriptions. And while the study has the limitations inherent to an observational study reliant upon data from a large U.S. administrative database – chiefly, the potential for residual confounding because of factors that couldn’t be adjusted for statistically – these real-world data may be as good as it gets, since patients with advanced CKD were excluded from the pivotal trials of the NOACs.
Apixaban (Eliquis) was the winner in this study: It separated itself from the pack by reducing the major bleeding risk by 57%, compared with warfarin, although it wasn’t significantly more effective than the other drugs in terms of stroke prevention. In contrast, the major bleeding rates for dabigatran (Pradaxa) and rivaroxaban (Xarelto) weren’t significantly different from warfarin in this challenging patient population.
In a related analysis of 10,712 patients with atrial fibrillation and advanced CKD who were on dialysis, use of an OAC was once again a winning strategy: It resulted not only in an impressive 58% reduction in the risk of stroke or systemic embolism, but also a 26% reduction in the risk of major bleeding, compared with no OAC.
Here again, apixaban was arguably the drug of choice. None of the 125 dialysis patients on apixaban experienced a stroke or systemic embolism. In contrast, dabigatran and rivaroxaban were associated with greater than threefold higher stroke rates than in patients on warfarin, although these differences didn’t achieve statistical significance because of small numbers, just 36 patients on dabigatran and 56 on rivaroxaban, the cardiologist continued.
For these analyses of the relationship between OAC exposure and stroke and bleeding outcomes, Dr. Noseworthy and his coinvestigators used propensity scores based upon 59 clinical and sociodemographic characteristics.
Asked why rates of utilization of OACs are so low in patients with advanced CKD, Dr. Noseworthy replied that he didn’t find that particularly surprising.
“Even if you look only at patients without renal dysfunction, there is incredible undertreatment of atrial fibrillation with OACs. And adherence is very poor,” he observed.
Moreover, in talking with nephrologists, he finds many of them have legitimate reservations about prescribing OACs for patients with end-stage renal disease on hemodialysis.
“They’re undergoing a lot of procedures. They’re having a ton of lines placed; they’re having fistulas revised; and they have very high rates of GI bleeding. In some studies the annual risk of bleeding is 20%-40% in this population. And they’re a frail population with frequent falls,” Dr. Noseworthy said.
He reported having no financial conflicts of interest regarding his study, which was conducted free of commercial support.
BARCELONA – Oral anticoagulation had a net overall benefit for patients with atrial fibrillation and advanced chronic kidney disease, based on results of a large observational study reported at the annual congress of the European Society of Cardiology.
The novel direct-acting oral anticoagulants (NOACs) and warfarin were all similarly effective in this study of 39,241 patients who had stage 4 or 5 chronic kidney disease (CKD), atrial fibrillation, and were not on dialysis. Compared with no oral anticoagulation, the drugs cut in half the risk of stroke or systemic embolism, with no increased risk of major bleeding.
“In patients with advanced CKD, it appears that OACs [oral anticoagulants] are reasonable,” concluded Peter A. Noseworthy, MD, of the Mayo Clinic in Rochester, Minn.
This is a potentially practice-changing finding given the “striking underutilization” of OACs in advanced CKD, he noted. Indeed, only one-third of the patients in this study were prescribed an OAC and picked up their prescriptions. And while the study has the limitations inherent to an observational study reliant upon data from a large U.S. administrative database – chiefly, the potential for residual confounding because of factors that couldn’t be adjusted for statistically – these real-world data may be as good as it gets, since patients with advanced CKD were excluded from the pivotal trials of the NOACs.
Apixaban (Eliquis) was the winner in this study: It separated itself from the pack by reducing the major bleeding risk by 57%, compared with warfarin, although it wasn’t significantly more effective than the other drugs in terms of stroke prevention. In contrast, the major bleeding rates for dabigatran (Pradaxa) and rivaroxaban (Xarelto) weren’t significantly different from warfarin in this challenging patient population.
In a related analysis of 10,712 patients with atrial fibrillation and advanced CKD who were on dialysis, use of an OAC was once again a winning strategy: It resulted not only in an impressive 58% reduction in the risk of stroke or systemic embolism, but also a 26% reduction in the risk of major bleeding, compared with no OAC.
Here again, apixaban was arguably the drug of choice. None of the 125 dialysis patients on apixaban experienced a stroke or systemic embolism. In contrast, dabigatran and rivaroxaban were associated with greater than threefold higher stroke rates than in patients on warfarin, although these differences didn’t achieve statistical significance because of small numbers, just 36 patients on dabigatran and 56 on rivaroxaban, the cardiologist continued.
For these analyses of the relationship between OAC exposure and stroke and bleeding outcomes, Dr. Noseworthy and his coinvestigators used propensity scores based upon 59 clinical and sociodemographic characteristics.
Asked why rates of utilization of OACs are so low in patients with advanced CKD, Dr. Noseworthy replied that he didn’t find that particularly surprising.
“Even if you look only at patients without renal dysfunction, there is incredible undertreatment of atrial fibrillation with OACs. And adherence is very poor,” he observed.
Moreover, in talking with nephrologists, he finds many of them have legitimate reservations about prescribing OACs for patients with end-stage renal disease on hemodialysis.
“They’re undergoing a lot of procedures. They’re having a ton of lines placed; they’re having fistulas revised; and they have very high rates of GI bleeding. In some studies the annual risk of bleeding is 20%-40% in this population. And they’re a frail population with frequent falls,” Dr. Noseworthy said.
He reported having no financial conflicts of interest regarding his study, which was conducted free of commercial support.
BARCELONA – Oral anticoagulation had a net overall benefit for patients with atrial fibrillation and advanced chronic kidney disease, based on results of a large observational study reported at the annual congress of the European Society of Cardiology.
The novel direct-acting oral anticoagulants (NOACs) and warfarin were all similarly effective in this study of 39,241 patients who had stage 4 or 5 chronic kidney disease (CKD), atrial fibrillation, and were not on dialysis. Compared with no oral anticoagulation, the drugs cut in half the risk of stroke or systemic embolism, with no increased risk of major bleeding.
“In patients with advanced CKD, it appears that OACs [oral anticoagulants] are reasonable,” concluded Peter A. Noseworthy, MD, of the Mayo Clinic in Rochester, Minn.
This is a potentially practice-changing finding given the “striking underutilization” of OACs in advanced CKD, he noted. Indeed, only one-third of the patients in this study were prescribed an OAC and picked up their prescriptions. And while the study has the limitations inherent to an observational study reliant upon data from a large U.S. administrative database – chiefly, the potential for residual confounding because of factors that couldn’t be adjusted for statistically – these real-world data may be as good as it gets, since patients with advanced CKD were excluded from the pivotal trials of the NOACs.
Apixaban (Eliquis) was the winner in this study: It separated itself from the pack by reducing the major bleeding risk by 57%, compared with warfarin, although it wasn’t significantly more effective than the other drugs in terms of stroke prevention. In contrast, the major bleeding rates for dabigatran (Pradaxa) and rivaroxaban (Xarelto) weren’t significantly different from warfarin in this challenging patient population.
In a related analysis of 10,712 patients with atrial fibrillation and advanced CKD who were on dialysis, use of an OAC was once again a winning strategy: It resulted not only in an impressive 58% reduction in the risk of stroke or systemic embolism, but also a 26% reduction in the risk of major bleeding, compared with no OAC.
Here again, apixaban was arguably the drug of choice. None of the 125 dialysis patients on apixaban experienced a stroke or systemic embolism. In contrast, dabigatran and rivaroxaban were associated with greater than threefold higher stroke rates than in patients on warfarin, although these differences didn’t achieve statistical significance because of small numbers, just 36 patients on dabigatran and 56 on rivaroxaban, the cardiologist continued.
For these analyses of the relationship between OAC exposure and stroke and bleeding outcomes, Dr. Noseworthy and his coinvestigators used propensity scores based upon 59 clinical and sociodemographic characteristics.
Asked why rates of utilization of OACs are so low in patients with advanced CKD, Dr. Noseworthy replied that he didn’t find that particularly surprising.
“Even if you look only at patients without renal dysfunction, there is incredible undertreatment of atrial fibrillation with OACs. And adherence is very poor,” he observed.
Moreover, in talking with nephrologists, he finds many of them have legitimate reservations about prescribing OACs for patients with end-stage renal disease on hemodialysis.
“They’re undergoing a lot of procedures. They’re having a ton of lines placed; they’re having fistulas revised; and they have very high rates of GI bleeding. In some studies the annual risk of bleeding is 20%-40% in this population. And they’re a frail population with frequent falls,” Dr. Noseworthy said.
He reported having no financial conflicts of interest regarding his study, which was conducted free of commercial support.
AT THE ESC CONGRESS 2017
Key clinical point:
Major finding: The risk of stroke/systemic embolism in patients with advanced chronic kidney disease who were on oral anticoagulation was reduced by 49% among those not on hemodialysis and by 58% in those who were, compared with similar patients not on oral anticoagulation.
Data source: This was an observational study of nearly 50,000 patients with atrial fibrillation and stage 4 or 5 chronic kidney disease in a large U.S. administrative database.
Disclosures: The presenter reported having no financial conflicts of interest regarding his study, which was conducted free of commercial support.
Methylphenidate shows enduring sleep benefits in pediatric ADHD
PARIS – Methylphenidate therapy for attention-deficit/hyperactivity disorder in medication-naive boys significantly improved their sleep quality, timing, and duration in a double-blind randomized trial, Michelle M. Solleveld, MD, reported at the annual congress of the European College of Neuropsychopharmacology.
Moreover, these salutary effects on sleep persisted for at least 1 week after methylphenidate was stopped at the end of the 16-week study, added Dr. Solleveld of the University of Amsterdam.

Indeed, while parents embrace the improvement in behavioral symptoms of ADHD provided by methylphenidate, they often express concern about the possible adverse effects of stimulant medication on their child’s sleep. The new study findings are reassuring on that score.
Sleep difficulties are a major problem in patients with ADHD: They tend to fall asleep later and have more frequent awakenings during the night, which results in decreased total sleep time and sleep efficiency, Dr. Solleveld noted.
Prior studies of methylphenidate’s effects on sleep in pediatric ADHD have yielded mixed results. The negative studies were too brief to provide meaningful results, according to Dr. Solleveld, who said at least 8 weeks of treatment are required in order to evaluate the drug’s effect on sleep problems properly.
She presented a randomized, double-blind, 16-week, placebo-controlled clinical trial involving 50 medication-naive boys with ADHD who were 10-12 years old. Their sleep was assessed via actigraphy measurements taken over 5 consecutive nights, keeping a sleep diary, and answering questionnaires, including the Epworth Sleepiness Scale, at three time points: prior to randomization, 8 weeks into the trial, and finally 1 week after the study ended.
Sleep efficiency – the primary study outcome – showed a strong 5% improvement in the methylphenidate group but was unchanged from baseline in placebo-treated controls. The boys who received methylphenidate fell asleep earlier, had a shorter latency of sleep onset, and slept for longer, compared with their baseline measures or with the sleep results in controls.
The finding that the methylphenidate-induced improvements in sleep persisted for a week after drug clearance is consistent with brain imaging studies carried out by Dr. Solleveld and her coinvestigators. They believe that the effects of stimulant therapy may be age dependent. The investigators previously have shown that adults with ADHD who began treatment with stimulants before age 16 years – when brain development is still ongoing – had lower levels of basal gamma-aminobutyric acid (GABA) and higher GABA response to an oral methylphenidate than did those who began treatment with stimulants after age 23 years. This is thought to be attributable to prolonged reductions in dopamine turnover induced by methylphenidate in the developing brain (Neuroimage Clin. 2017 Jun 2;15:812-8).
The Dutch investigators also have reported that methylphenidate therapy in children with ADHD – but not in affected adults – increased the cerebral blood flow response within the thalamus to a dopamine challenge (JAMA Psychiatry. 2016 Sep 1;73[9]:955-62).
The clinical ramifications of these apparently long-lasting, drug-related alterations in GABA neurotransmission are the subject of ongoing research.
Dr. Solleveld reported having no financial conflicts of interest.
PARIS – Methylphenidate therapy for attention-deficit/hyperactivity disorder in medication-naive boys significantly improved their sleep quality, timing, and duration in a double-blind randomized trial, Michelle M. Solleveld, MD, reported at the annual congress of the European College of Neuropsychopharmacology.
Moreover, these salutary effects on sleep persisted for at least 1 week after methylphenidate was stopped at the end of the 16-week study, added Dr. Solleveld of the University of Amsterdam.

Indeed, while parents embrace the improvement in behavioral symptoms of ADHD provided by methylphenidate, they often express concern about the possible adverse effects of stimulant medication on their child’s sleep. The new study findings are reassuring on that score.
Sleep difficulties are a major problem in patients with ADHD: They tend to fall asleep later and have more frequent awakenings during the night, which results in decreased total sleep time and sleep efficiency, Dr. Solleveld noted.
Prior studies of methylphenidate’s effects on sleep in pediatric ADHD have yielded mixed results. The negative studies were too brief to provide meaningful results, according to Dr. Solleveld, who said at least 8 weeks of treatment are required in order to evaluate the drug’s effect on sleep problems properly.
She presented a randomized, double-blind, 16-week, placebo-controlled clinical trial involving 50 medication-naive boys with ADHD who were 10-12 years old. Their sleep was assessed via actigraphy measurements taken over 5 consecutive nights, keeping a sleep diary, and answering questionnaires, including the Epworth Sleepiness Scale, at three time points: prior to randomization, 8 weeks into the trial, and finally 1 week after the study ended.
Sleep efficiency – the primary study outcome – showed a strong 5% improvement in the methylphenidate group but was unchanged from baseline in placebo-treated controls. The boys who received methylphenidate fell asleep earlier, had a shorter latency of sleep onset, and slept for longer, compared with their baseline measures or with the sleep results in controls.
The finding that the methylphenidate-induced improvements in sleep persisted for a week after drug clearance is consistent with brain imaging studies carried out by Dr. Solleveld and her coinvestigators. They believe that the effects of stimulant therapy may be age dependent. The investigators previously have shown that adults with ADHD who began treatment with stimulants before age 16 years – when brain development is still ongoing – had lower levels of basal gamma-aminobutyric acid (GABA) and higher GABA response to an oral methylphenidate than did those who began treatment with stimulants after age 23 years. This is thought to be attributable to prolonged reductions in dopamine turnover induced by methylphenidate in the developing brain (Neuroimage Clin. 2017 Jun 2;15:812-8).
The Dutch investigators also have reported that methylphenidate therapy in children with ADHD – but not in affected adults – increased the cerebral blood flow response within the thalamus to a dopamine challenge (JAMA Psychiatry. 2016 Sep 1;73[9]:955-62).
The clinical ramifications of these apparently long-lasting, drug-related alterations in GABA neurotransmission are the subject of ongoing research.
Dr. Solleveld reported having no financial conflicts of interest.
PARIS – Methylphenidate therapy for attention-deficit/hyperactivity disorder in medication-naive boys significantly improved their sleep quality, timing, and duration in a double-blind randomized trial, Michelle M. Solleveld, MD, reported at the annual congress of the European College of Neuropsychopharmacology.
Moreover, these salutary effects on sleep persisted for at least 1 week after methylphenidate was stopped at the end of the 16-week study, added Dr. Solleveld of the University of Amsterdam.

Indeed, while parents embrace the improvement in behavioral symptoms of ADHD provided by methylphenidate, they often express concern about the possible adverse effects of stimulant medication on their child’s sleep. The new study findings are reassuring on that score.
Sleep difficulties are a major problem in patients with ADHD: They tend to fall asleep later and have more frequent awakenings during the night, which results in decreased total sleep time and sleep efficiency, Dr. Solleveld noted.
Prior studies of methylphenidate’s effects on sleep in pediatric ADHD have yielded mixed results. The negative studies were too brief to provide meaningful results, according to Dr. Solleveld, who said at least 8 weeks of treatment are required in order to evaluate the drug’s effect on sleep problems properly.
She presented a randomized, double-blind, 16-week, placebo-controlled clinical trial involving 50 medication-naive boys with ADHD who were 10-12 years old. Their sleep was assessed via actigraphy measurements taken over 5 consecutive nights, keeping a sleep diary, and answering questionnaires, including the Epworth Sleepiness Scale, at three time points: prior to randomization, 8 weeks into the trial, and finally 1 week after the study ended.
Sleep efficiency – the primary study outcome – showed a strong 5% improvement in the methylphenidate group but was unchanged from baseline in placebo-treated controls. The boys who received methylphenidate fell asleep earlier, had a shorter latency of sleep onset, and slept for longer, compared with their baseline measures or with the sleep results in controls.
The finding that the methylphenidate-induced improvements in sleep persisted for a week after drug clearance is consistent with brain imaging studies carried out by Dr. Solleveld and her coinvestigators. They believe that the effects of stimulant therapy may be age dependent. The investigators previously have shown that adults with ADHD who began treatment with stimulants before age 16 years – when brain development is still ongoing – had lower levels of basal gamma-aminobutyric acid (GABA) and higher GABA response to an oral methylphenidate than did those who began treatment with stimulants after age 23 years. This is thought to be attributable to prolonged reductions in dopamine turnover induced by methylphenidate in the developing brain (Neuroimage Clin. 2017 Jun 2;15:812-8).
The Dutch investigators also have reported that methylphenidate therapy in children with ADHD – but not in affected adults – increased the cerebral blood flow response within the thalamus to a dopamine challenge (JAMA Psychiatry. 2016 Sep 1;73[9]:955-62).
The clinical ramifications of these apparently long-lasting, drug-related alterations in GABA neurotransmission are the subject of ongoing research.
Dr. Solleveld reported having no financial conflicts of interest.
AT THE ECNP CONGRESS
Key clinical point:
Major finding: Sleep efficiency in boys with ADHD improved significantly by 5% in response to methylphenidate therapy.
Data source: This randomized, double-blind, placebo-controlled clinical trial included 50 medication-naive boys aged 10-12 years with ADHD.
Disclosures: The study presenter reported having no financial conflicts of interest.
Add aggressiveness to mixed features specifier for major depressive episode
PARIS – Aggressiveness deserves to be incorporated in the next Diagnostic and Statistical Manual of Mental Disorders update as a new clinical criterion triggering application of the “with mixed features” specifier in patients diagnosed with a major depressive episode, Norma Verdolini, MD, said at the annual congress of the European College of Neuropsychopharmacology.
“Aggressiveness might be a trait component of bipolarity and a diagnostic indicator of ‘mixicity’ in patients with a major depressive episode. This has implications for the therapeutic strategy,” said Dr. Verdolini of the bipolar disorders unit at the University of Barcelona Institute of Neurosciences.
The BRIDGE-II-MIX study was a cross-sectional observational study of 2,811 adults with MDE at 239 centers in eight European countries (J Clin Psychiatry. 2015 Mar;76[3]:e351-8). Three hundred ninety-nine participants (14.2%) met the operational definition of physical or verbal aggressiveness used in Dr. Verdolini’s new post-hoc analysis.
Statistically significant and clinically meaningful differences were found between MDE patients with aggressiveness (MDE-aggro) and MDE without aggressiveness. For example, the MDE-aggro group was twice as likely to meet DSM-IV-TR criteria for bipolar disorder I. Twenty-seven percent of the MDE-aggro group met DSM-5 criteria for a mixed state, meaning both depressed mood and mania in the same episode, compared with just 4% of the MDE-no-aggro group.
The MDE-aggro patients also had a strikingly greater prevalence of comorbid borderline personality disorder, by a margin of 20% versus 4%. They had a younger mean age at their first depressive episode: 29.9 years old, compared with 36.1 in the MDE-no-aggro group. The MDE-aggro patients had more prior mood episodes and a greater number of lifetime suicide attempts. In addition, they had significantly more severe depression, mania, and bipolar disorder scores on the Clinical Global Impression Scale for Bipolar Disorder.
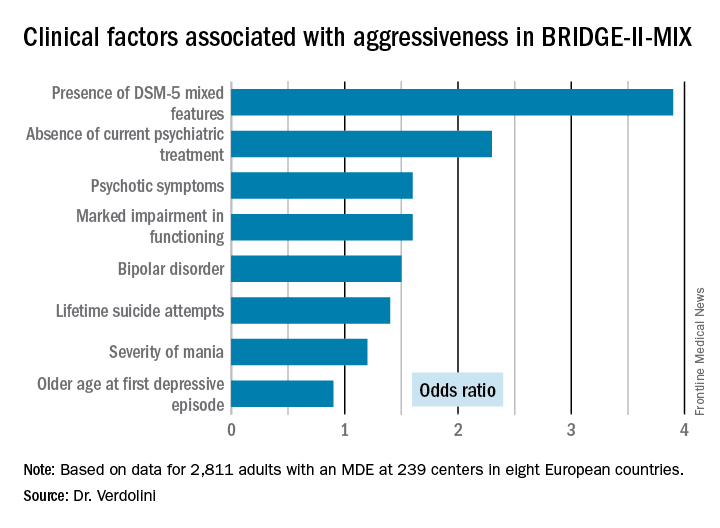
“Our results should prompt reconsideration of the diagnostic criteria for the mixed features specifier. The detection of aggression in MDE could represent a therapeutic target in personalized pharmacological treatment for bipolar disorder,” Dr. Verdolini concluded.
The BRIDGE-II-MIX study was sponsored by Sanofi-Aventis. Dr. Verdolini reported receiving research funding from the company.
PARIS – Aggressiveness deserves to be incorporated in the next Diagnostic and Statistical Manual of Mental Disorders update as a new clinical criterion triggering application of the “with mixed features” specifier in patients diagnosed with a major depressive episode, Norma Verdolini, MD, said at the annual congress of the European College of Neuropsychopharmacology.
“Aggressiveness might be a trait component of bipolarity and a diagnostic indicator of ‘mixicity’ in patients with a major depressive episode. This has implications for the therapeutic strategy,” said Dr. Verdolini of the bipolar disorders unit at the University of Barcelona Institute of Neurosciences.
The BRIDGE-II-MIX study was a cross-sectional observational study of 2,811 adults with MDE at 239 centers in eight European countries (J Clin Psychiatry. 2015 Mar;76[3]:e351-8). Three hundred ninety-nine participants (14.2%) met the operational definition of physical or verbal aggressiveness used in Dr. Verdolini’s new post-hoc analysis.
Statistically significant and clinically meaningful differences were found between MDE patients with aggressiveness (MDE-aggro) and MDE without aggressiveness. For example, the MDE-aggro group was twice as likely to meet DSM-IV-TR criteria for bipolar disorder I. Twenty-seven percent of the MDE-aggro group met DSM-5 criteria for a mixed state, meaning both depressed mood and mania in the same episode, compared with just 4% of the MDE-no-aggro group.
The MDE-aggro patients also had a strikingly greater prevalence of comorbid borderline personality disorder, by a margin of 20% versus 4%. They had a younger mean age at their first depressive episode: 29.9 years old, compared with 36.1 in the MDE-no-aggro group. The MDE-aggro patients had more prior mood episodes and a greater number of lifetime suicide attempts. In addition, they had significantly more severe depression, mania, and bipolar disorder scores on the Clinical Global Impression Scale for Bipolar Disorder.

“Our results should prompt reconsideration of the diagnostic criteria for the mixed features specifier. The detection of aggression in MDE could represent a therapeutic target in personalized pharmacological treatment for bipolar disorder,” Dr. Verdolini concluded.
The BRIDGE-II-MIX study was sponsored by Sanofi-Aventis. Dr. Verdolini reported receiving research funding from the company.
PARIS – Aggressiveness deserves to be incorporated in the next Diagnostic and Statistical Manual of Mental Disorders update as a new clinical criterion triggering application of the “with mixed features” specifier in patients diagnosed with a major depressive episode, Norma Verdolini, MD, said at the annual congress of the European College of Neuropsychopharmacology.
“Aggressiveness might be a trait component of bipolarity and a diagnostic indicator of ‘mixicity’ in patients with a major depressive episode. This has implications for the therapeutic strategy,” said Dr. Verdolini of the bipolar disorders unit at the University of Barcelona Institute of Neurosciences.
The BRIDGE-II-MIX study was a cross-sectional observational study of 2,811 adults with MDE at 239 centers in eight European countries (J Clin Psychiatry. 2015 Mar;76[3]:e351-8). Three hundred ninety-nine participants (14.2%) met the operational definition of physical or verbal aggressiveness used in Dr. Verdolini’s new post-hoc analysis.
Statistically significant and clinically meaningful differences were found between MDE patients with aggressiveness (MDE-aggro) and MDE without aggressiveness. For example, the MDE-aggro group was twice as likely to meet DSM-IV-TR criteria for bipolar disorder I. Twenty-seven percent of the MDE-aggro group met DSM-5 criteria for a mixed state, meaning both depressed mood and mania in the same episode, compared with just 4% of the MDE-no-aggro group.
The MDE-aggro patients also had a strikingly greater prevalence of comorbid borderline personality disorder, by a margin of 20% versus 4%. They had a younger mean age at their first depressive episode: 29.9 years old, compared with 36.1 in the MDE-no-aggro group. The MDE-aggro patients had more prior mood episodes and a greater number of lifetime suicide attempts. In addition, they had significantly more severe depression, mania, and bipolar disorder scores on the Clinical Global Impression Scale for Bipolar Disorder.

“Our results should prompt reconsideration of the diagnostic criteria for the mixed features specifier. The detection of aggression in MDE could represent a therapeutic target in personalized pharmacological treatment for bipolar disorder,” Dr. Verdolini concluded.
The BRIDGE-II-MIX study was sponsored by Sanofi-Aventis. Dr. Verdolini reported receiving research funding from the company.
AT THE ECNP CONGRESS
Key clinical point:
Major finding: Patients who fulfilled the DSM-5 criteria for a major depressive episode with mixed features were 3.9-fold more likely to meet investigators’ operational definition of aggressiveness.
Data source: This was a post-hoc analysis of the BRIDGE-II-MIX study, an observational cross-sectional study of 2,811 adults experiencing a major depressive episode.
Disclosures: The BRIDGE-II-MIX study was sponsored by Sanofi-Aventis. The presenter reported receiving research funding from the company.
ADHD meds don’t raise seizure risk in epilepsy patients
PARIS – The use of attention-deficit/hyperactivity disorder medications is not associated with increased risk of epileptic seizures in patients with both disorders, according to an analysis of Swedish national registry data.
“Seizure history should not exempt patients from ADHD medication treatment,” Isabell Brikell stated at the annual congress of the European College of Neuropsychopharmacology.

“That’s why it’s such an important question, whether ADHD medications increase the risk of seizures,” observed Ms. Brikell, a PhD candidate in psychiatric genetics and epidemiology at the Karolinska Institute in Stockholm.
Swedish health care registries are famously comprehensive. For example, the Swedish prescription medication registry that Ms. Brikell and her coinvestigators tapped into for their ADHD/epilepsy study contains information on 99% of all prescriptions ordered in the country since 2005.
She reported on 38,247 Swedish patients with epilepsy born during 1976-2008 and followed during 2006-2013. Forty-eight percent were female. They collectively experienced 30,093 acute epileptic seizures of sufficient severity that they presented to a hospital for an unplanned visit. When the investigators compared the rate of seizures while the patients with ADHD were on a collective 4,248 ADHD medication exposure periods to that of epilepsy patients without ADHD, they found that the seizure risk was actually 17% lower in ADHD patients while on medication. This difference fell just shy of statistical significance. The analysis was adjusted for gender, age, and time on ADHD medications.
However, Ms. Brikell and her coworkers also performed a separate analysis for each individual with ADHD in which they compared seizure rates when a given patient was on ADHD medication versus off medication, a design that controls for many of the potential confounding factors that can occur with observational data. The seizure risk proved to be 19% lower while an individual was on ADHD medication – and this difference was indeed statistically significant.
In an interview, Ms. Brikell noted that the Swedish data are confirmed by a much larger National Institute of Mental Health–sponsored American study she was involved with that is now under review for publication. The U.S. study, which used the enormous MarketScan private health insurance database, demonstrated with the power provided by very large patient numbers that the seizure risk was convincingly lower while dual-diagnosis patients were on ADHD medication than when they were off.
“It’s reassuring to see the same effect across two countries with such different health care systems,” she commented.
Epilepsy is known to be inherently associated with a threefold increased prevalence of ADHD.
Ms. Brikell’s study was funded by the Swedish Research Council, the U.S. National Institute of Mental Health, and the Swedish Initiative for Research on Microdata in the Social and Medical Sciences. She reported having no financial conflicts of interest.
PARIS – The use of attention-deficit/hyperactivity disorder medications is not associated with increased risk of epileptic seizures in patients with both disorders, according to an analysis of Swedish national registry data.
“Seizure history should not exempt patients from ADHD medication treatment,” Isabell Brikell stated at the annual congress of the European College of Neuropsychopharmacology.

“That’s why it’s such an important question, whether ADHD medications increase the risk of seizures,” observed Ms. Brikell, a PhD candidate in psychiatric genetics and epidemiology at the Karolinska Institute in Stockholm.
Swedish health care registries are famously comprehensive. For example, the Swedish prescription medication registry that Ms. Brikell and her coinvestigators tapped into for their ADHD/epilepsy study contains information on 99% of all prescriptions ordered in the country since 2005.
She reported on 38,247 Swedish patients with epilepsy born during 1976-2008 and followed during 2006-2013. Forty-eight percent were female. They collectively experienced 30,093 acute epileptic seizures of sufficient severity that they presented to a hospital for an unplanned visit. When the investigators compared the rate of seizures while the patients with ADHD were on a collective 4,248 ADHD medication exposure periods to that of epilepsy patients without ADHD, they found that the seizure risk was actually 17% lower in ADHD patients while on medication. This difference fell just shy of statistical significance. The analysis was adjusted for gender, age, and time on ADHD medications.
However, Ms. Brikell and her coworkers also performed a separate analysis for each individual with ADHD in which they compared seizure rates when a given patient was on ADHD medication versus off medication, a design that controls for many of the potential confounding factors that can occur with observational data. The seizure risk proved to be 19% lower while an individual was on ADHD medication – and this difference was indeed statistically significant.
In an interview, Ms. Brikell noted that the Swedish data are confirmed by a much larger National Institute of Mental Health–sponsored American study she was involved with that is now under review for publication. The U.S. study, which used the enormous MarketScan private health insurance database, demonstrated with the power provided by very large patient numbers that the seizure risk was convincingly lower while dual-diagnosis patients were on ADHD medication than when they were off.
“It’s reassuring to see the same effect across two countries with such different health care systems,” she commented.
Epilepsy is known to be inherently associated with a threefold increased prevalence of ADHD.
Ms. Brikell’s study was funded by the Swedish Research Council, the U.S. National Institute of Mental Health, and the Swedish Initiative for Research on Microdata in the Social and Medical Sciences. She reported having no financial conflicts of interest.
PARIS – The use of attention-deficit/hyperactivity disorder medications is not associated with increased risk of epileptic seizures in patients with both disorders, according to an analysis of Swedish national registry data.
“Seizure history should not exempt patients from ADHD medication treatment,” Isabell Brikell stated at the annual congress of the European College of Neuropsychopharmacology.

“That’s why it’s such an important question, whether ADHD medications increase the risk of seizures,” observed Ms. Brikell, a PhD candidate in psychiatric genetics and epidemiology at the Karolinska Institute in Stockholm.
Swedish health care registries are famously comprehensive. For example, the Swedish prescription medication registry that Ms. Brikell and her coinvestigators tapped into for their ADHD/epilepsy study contains information on 99% of all prescriptions ordered in the country since 2005.
She reported on 38,247 Swedish patients with epilepsy born during 1976-2008 and followed during 2006-2013. Forty-eight percent were female. They collectively experienced 30,093 acute epileptic seizures of sufficient severity that they presented to a hospital for an unplanned visit. When the investigators compared the rate of seizures while the patients with ADHD were on a collective 4,248 ADHD medication exposure periods to that of epilepsy patients without ADHD, they found that the seizure risk was actually 17% lower in ADHD patients while on medication. This difference fell just shy of statistical significance. The analysis was adjusted for gender, age, and time on ADHD medications.
However, Ms. Brikell and her coworkers also performed a separate analysis for each individual with ADHD in which they compared seizure rates when a given patient was on ADHD medication versus off medication, a design that controls for many of the potential confounding factors that can occur with observational data. The seizure risk proved to be 19% lower while an individual was on ADHD medication – and this difference was indeed statistically significant.
In an interview, Ms. Brikell noted that the Swedish data are confirmed by a much larger National Institute of Mental Health–sponsored American study she was involved with that is now under review for publication. The U.S. study, which used the enormous MarketScan private health insurance database, demonstrated with the power provided by very large patient numbers that the seizure risk was convincingly lower while dual-diagnosis patients were on ADHD medication than when they were off.
“It’s reassuring to see the same effect across two countries with such different health care systems,” she commented.
Epilepsy is known to be inherently associated with a threefold increased prevalence of ADHD.
Ms. Brikell’s study was funded by the Swedish Research Council, the U.S. National Institute of Mental Health, and the Swedish Initiative for Research on Microdata in the Social and Medical Sciences. She reported having no financial conflicts of interest.
AT THE ECNP CONGRESS
Key clinical point:
Major finding: Patients with ADHD and a history of epilepsy were at 19% lower risk of experiencing seizures while on ADHD medication than when off it.
Data source: This was an observational study of prospectively collected data on all of the nearly 40,000 Swedish patients with epilepsy born during 1976-2008.
Disclosures: The study was funded by the Swedish Research Council, the U.S. National Institute of Mental Health, and the Swedish Initiative for Research on Microdata in the Social and Medical Sciences. The presenter reported having no financial conflicts of interest.
PRIDE study supports novel approach to ECT for geriatric depression
PARIS – Results of the randomized phase 2 portion of the landmark PRIDE study of electroconvulsive therapy for severe unipolar depression in geriatric patients hold a key message for clinicians, according to Charles H. Kellner, MD.
“The clinical take-home message is that practitioners should be liberal in prescribing additional ECT past the acute course. So our recommendation is that tapering ECT courses and being liberal with continuation and maintenance ECT should be adopted in clinical practice more widely with the aim of preventing full syndromic relapse and its catastrophic consequences,” Dr. Kellner said at the annual congress of the European College of Neuropsychopharmacology.

PRIDE (Prolonging Remission in Depressed Elderly) was a National Institute of Mental Health–sponsored nine-center study of right unilateral ultrabrief pulse ECT at 0.25 msec plus supportive pharmacotherapy for treatment of geriatric depression.
Phase 1 of the study involved 240 affected patients, 62% of whom achieved remission after receiving this form of ECT at six times the seizure threshold thrice weekly for up to 1 month coupled with low- or medium-dose venlafaxine. A mean of 7.3 ECT sessions were needed to attain remission as defined by a score of 10 or less on the Hamilton Rating Scale for Depression (HAM-D24) on two consecutive occasions, down from a mean baseline score of 31.2. The scale was administered three times per week. Safety and tolerability of the ECT regimen were excellent, according to Dr. Kellner, chief of electroconvulsive therapy at New York Community Hospital and a psychiatrist at Mount Sinai School of Medicine in New York.
The PRIDE phase 1 data confirmed several points previously made in earlier studies. One is that the older patients are, the more ECT-responsive they are, even within an all-geriatric cohort such as PRIDE. Indeed, the remission rate was 55% in the 60- to 69-year-olds, compared with 72% in the 70- to 79-year-olds.
Another finding consistent with other studies: Patients with psychotic depression do particularly well with ECT. All PRIDE participants with psychotic depression achieved remission.
Also, ECT had a very rapid antisuicidal effect. At baseline, 24% of patients had a score of 1 on HAM-D24 item 3, which rates suicidality. An additional 34% had a baseline score of 2, 14% scored 3, and only 23% had a score of 0. After only a few weeks of ECT, however, 84% of patients had a score of 0 and 9% scored a 1.
“That’s one of the compelling clinical reasons to refer patients for ECT: This type of ECT is really good for treating suicidality,” Dr. Kellner noted.
Phase 2 was the more interesting part of the PRIDE study, he continued, because it evaluated in randomized fashion the efficacy and tolerability of a novel flexible, individualized strategy for as-needed maintenance ECT to sustain the mood improvement once remission was achieved. Dr. Kellner stressed that some form of maintenance therapy is essential post ECT-induced remission.
“It’s unreasonable to expect that ECT could cure the patients’ underlying illness and protect them from getting sick again for the rest of their lives. One has to understand this is a recurrent episodic illness that we’re treating. What ECT does is treat the current episode, and it does it better and more thoroughly than any other treatment in psychiatry,” Dr. Kellner said.
Post-ECT relapse rates are clearly higher in the modern era, which makes a compelling case for developing safe and effective maintenance strategies.
“We don’t quite understand why relapse rates are higher today. My belief is that for patients who come to ECT, their illness has been destabilized by having been on multiple trials of antidepressant medications beforehand. It may also be that the forms of ECT that we’re using today are somewhat less potent than the ones used in previous decades,” Dr. Kellner conceded.
He and his coinvestigators named their investigational maintenance ECT strategy STABLE, for Symptom-Titrated, Algorithm-Based Longitudinal ECT. It’s a complex algorithm described in detail in a published report (Am J Psychiatry. 2016 Nov 1; 173[11]:1110-18). Basically, it consisted of four mandatory additional ECT sessions administered once weekly for the first month post remission, followed by either one, two, or no ECT sessions per week based upon evidence of deterioration as expressed in HAM-D24 scores.
In phase 2 of PRIDE, 128 remitters from phase 1 were continued on venlafaxine at a mean dose of 192 mg/day. In addition, they were placed on lithium, achieving a mean lithium level of 0.53 mEq/L, which Dr. Kellner deemed “low but reasonable.” These 128 patients were then randomized to the STABLE arm of flexibly administered ECT or to medication only and were followed prospectively for 6 months.
The primary endpoint was change in HAM-D24 score over the course of 6 months. From a baseline mean total score of 6, the mean score climbed to 8.4 in the medication-only group while dropping to 4.2 in the STABLE arm.
“At every time point along the way in the 6-month course of phase 2 of the illness, patients who were in the STABLE arm were less symptomatic and doing better than patients in the medication-only arm,” Dr. Kellner observed.
The key secondary efficacy outcome was change in the Clinical Global Impressions Severity scale. At follow-up, the patients in the flexible ECT plus medication arm were 5.2 times more likely to be rated “not ill at all” than were those on pharmacotherapy only.
In addition, relapse occurred in 20% of the medication-only group versus 13% in the STABLE group.
Global cognitive functioning as assessed by the Mini-Mental State Examination – crudely, in Dr. Kellner’s view – did not differ between the two groups at follow-up. Results of more sophisticated tests of multiple specific domains of cognition will be forthcoming.
Of note, two-thirds of patients in the STABLE arm required no additional ECT after their four continuation ECTs during the first month.
A swipe at ECT’s critics
Dr. Kellner called ECT “a fabulous therapy,” albeit one with a serious image problem.
“I think the problem with ECT is not that we don’t know all the details of how it works or what it does clinically, it’s really a sociopolitical issue about ECT not being adequately accepted. I break down this lack of acceptance into two forms: The first is passive lack of acceptance based on our profession not having embraced ECT and continuing to fail to embrace it. The other is the active propaganda against ECT that is promulgated by the Church of Scientology, which has taken on a life of its own now because of the Internet. ECT is still fought by the Church of Scientology. They fund lots of people to say incorrect nasty things about ECT to inappropriately frighten our patients,” Dr. Kellner charged.
He cautioned that proposed new Food and Drug Administration regulations governing ECT devices would greatly limit the use of ECT, restricting it to adults with treatment-resistant major depressive disorder or bipolar disorder or who require a rapid response. Other currently cleared indications would become off-label uses.
“With off-label ECT, the potential problem is practitioners may not be covered by their malpractice insurance. And the bigger issue is health insurers may not pay for it unless the ECT is for an on-label indication,” Dr. Kellner said.
He shared that he has found extremely helpful a colleague’s advice to demystify ECT by inviting a family member to witness a patient’s treatment session. That witness can then testify to others that what goes on bears no resemblance to what happens to Jack Nicholson in the classic film “One Flew Over the Cuckoo’s Nest.”
The PRIDE study was funded by the National Institute of Mental Health. Dr. Kellner reported having no financial conflicts of interest.
PARIS – Results of the randomized phase 2 portion of the landmark PRIDE study of electroconvulsive therapy for severe unipolar depression in geriatric patients hold a key message for clinicians, according to Charles H. Kellner, MD.
“The clinical take-home message is that practitioners should be liberal in prescribing additional ECT past the acute course. So our recommendation is that tapering ECT courses and being liberal with continuation and maintenance ECT should be adopted in clinical practice more widely with the aim of preventing full syndromic relapse and its catastrophic consequences,” Dr. Kellner said at the annual congress of the European College of Neuropsychopharmacology.

PRIDE (Prolonging Remission in Depressed Elderly) was a National Institute of Mental Health–sponsored nine-center study of right unilateral ultrabrief pulse ECT at 0.25 msec plus supportive pharmacotherapy for treatment of geriatric depression.
Phase 1 of the study involved 240 affected patients, 62% of whom achieved remission after receiving this form of ECT at six times the seizure threshold thrice weekly for up to 1 month coupled with low- or medium-dose venlafaxine. A mean of 7.3 ECT sessions were needed to attain remission as defined by a score of 10 or less on the Hamilton Rating Scale for Depression (HAM-D24) on two consecutive occasions, down from a mean baseline score of 31.2. The scale was administered three times per week. Safety and tolerability of the ECT regimen were excellent, according to Dr. Kellner, chief of electroconvulsive therapy at New York Community Hospital and a psychiatrist at Mount Sinai School of Medicine in New York.
The PRIDE phase 1 data confirmed several points previously made in earlier studies. One is that the older patients are, the more ECT-responsive they are, even within an all-geriatric cohort such as PRIDE. Indeed, the remission rate was 55% in the 60- to 69-year-olds, compared with 72% in the 70- to 79-year-olds.
Another finding consistent with other studies: Patients with psychotic depression do particularly well with ECT. All PRIDE participants with psychotic depression achieved remission.
Also, ECT had a very rapid antisuicidal effect. At baseline, 24% of patients had a score of 1 on HAM-D24 item 3, which rates suicidality. An additional 34% had a baseline score of 2, 14% scored 3, and only 23% had a score of 0. After only a few weeks of ECT, however, 84% of patients had a score of 0 and 9% scored a 1.
“That’s one of the compelling clinical reasons to refer patients for ECT: This type of ECT is really good for treating suicidality,” Dr. Kellner noted.
Phase 2 was the more interesting part of the PRIDE study, he continued, because it evaluated in randomized fashion the efficacy and tolerability of a novel flexible, individualized strategy for as-needed maintenance ECT to sustain the mood improvement once remission was achieved. Dr. Kellner stressed that some form of maintenance therapy is essential post ECT-induced remission.
“It’s unreasonable to expect that ECT could cure the patients’ underlying illness and protect them from getting sick again for the rest of their lives. One has to understand this is a recurrent episodic illness that we’re treating. What ECT does is treat the current episode, and it does it better and more thoroughly than any other treatment in psychiatry,” Dr. Kellner said.
Post-ECT relapse rates are clearly higher in the modern era, which makes a compelling case for developing safe and effective maintenance strategies.
“We don’t quite understand why relapse rates are higher today. My belief is that for patients who come to ECT, their illness has been destabilized by having been on multiple trials of antidepressant medications beforehand. It may also be that the forms of ECT that we’re using today are somewhat less potent than the ones used in previous decades,” Dr. Kellner conceded.
He and his coinvestigators named their investigational maintenance ECT strategy STABLE, for Symptom-Titrated, Algorithm-Based Longitudinal ECT. It’s a complex algorithm described in detail in a published report (Am J Psychiatry. 2016 Nov 1; 173[11]:1110-18). Basically, it consisted of four mandatory additional ECT sessions administered once weekly for the first month post remission, followed by either one, two, or no ECT sessions per week based upon evidence of deterioration as expressed in HAM-D24 scores.
In phase 2 of PRIDE, 128 remitters from phase 1 were continued on venlafaxine at a mean dose of 192 mg/day. In addition, they were placed on lithium, achieving a mean lithium level of 0.53 mEq/L, which Dr. Kellner deemed “low but reasonable.” These 128 patients were then randomized to the STABLE arm of flexibly administered ECT or to medication only and were followed prospectively for 6 months.
The primary endpoint was change in HAM-D24 score over the course of 6 months. From a baseline mean total score of 6, the mean score climbed to 8.4 in the medication-only group while dropping to 4.2 in the STABLE arm.
“At every time point along the way in the 6-month course of phase 2 of the illness, patients who were in the STABLE arm were less symptomatic and doing better than patients in the medication-only arm,” Dr. Kellner observed.
The key secondary efficacy outcome was change in the Clinical Global Impressions Severity scale. At follow-up, the patients in the flexible ECT plus medication arm were 5.2 times more likely to be rated “not ill at all” than were those on pharmacotherapy only.
In addition, relapse occurred in 20% of the medication-only group versus 13% in the STABLE group.
Global cognitive functioning as assessed by the Mini-Mental State Examination – crudely, in Dr. Kellner’s view – did not differ between the two groups at follow-up. Results of more sophisticated tests of multiple specific domains of cognition will be forthcoming.
Of note, two-thirds of patients in the STABLE arm required no additional ECT after their four continuation ECTs during the first month.
A swipe at ECT’s critics
Dr. Kellner called ECT “a fabulous therapy,” albeit one with a serious image problem.
“I think the problem with ECT is not that we don’t know all the details of how it works or what it does clinically, it’s really a sociopolitical issue about ECT not being adequately accepted. I break down this lack of acceptance into two forms: The first is passive lack of acceptance based on our profession not having embraced ECT and continuing to fail to embrace it. The other is the active propaganda against ECT that is promulgated by the Church of Scientology, which has taken on a life of its own now because of the Internet. ECT is still fought by the Church of Scientology. They fund lots of people to say incorrect nasty things about ECT to inappropriately frighten our patients,” Dr. Kellner charged.
He cautioned that proposed new Food and Drug Administration regulations governing ECT devices would greatly limit the use of ECT, restricting it to adults with treatment-resistant major depressive disorder or bipolar disorder or who require a rapid response. Other currently cleared indications would become off-label uses.
“With off-label ECT, the potential problem is practitioners may not be covered by their malpractice insurance. And the bigger issue is health insurers may not pay for it unless the ECT is for an on-label indication,” Dr. Kellner said.
He shared that he has found extremely helpful a colleague’s advice to demystify ECT by inviting a family member to witness a patient’s treatment session. That witness can then testify to others that what goes on bears no resemblance to what happens to Jack Nicholson in the classic film “One Flew Over the Cuckoo’s Nest.”
The PRIDE study was funded by the National Institute of Mental Health. Dr. Kellner reported having no financial conflicts of interest.
PARIS – Results of the randomized phase 2 portion of the landmark PRIDE study of electroconvulsive therapy for severe unipolar depression in geriatric patients hold a key message for clinicians, according to Charles H. Kellner, MD.
“The clinical take-home message is that practitioners should be liberal in prescribing additional ECT past the acute course. So our recommendation is that tapering ECT courses and being liberal with continuation and maintenance ECT should be adopted in clinical practice more widely with the aim of preventing full syndromic relapse and its catastrophic consequences,” Dr. Kellner said at the annual congress of the European College of Neuropsychopharmacology.

PRIDE (Prolonging Remission in Depressed Elderly) was a National Institute of Mental Health–sponsored nine-center study of right unilateral ultrabrief pulse ECT at 0.25 msec plus supportive pharmacotherapy for treatment of geriatric depression.
Phase 1 of the study involved 240 affected patients, 62% of whom achieved remission after receiving this form of ECT at six times the seizure threshold thrice weekly for up to 1 month coupled with low- or medium-dose venlafaxine. A mean of 7.3 ECT sessions were needed to attain remission as defined by a score of 10 or less on the Hamilton Rating Scale for Depression (HAM-D24) on two consecutive occasions, down from a mean baseline score of 31.2. The scale was administered three times per week. Safety and tolerability of the ECT regimen were excellent, according to Dr. Kellner, chief of electroconvulsive therapy at New York Community Hospital and a psychiatrist at Mount Sinai School of Medicine in New York.
The PRIDE phase 1 data confirmed several points previously made in earlier studies. One is that the older patients are, the more ECT-responsive they are, even within an all-geriatric cohort such as PRIDE. Indeed, the remission rate was 55% in the 60- to 69-year-olds, compared with 72% in the 70- to 79-year-olds.
Another finding consistent with other studies: Patients with psychotic depression do particularly well with ECT. All PRIDE participants with psychotic depression achieved remission.
Also, ECT had a very rapid antisuicidal effect. At baseline, 24% of patients had a score of 1 on HAM-D24 item 3, which rates suicidality. An additional 34% had a baseline score of 2, 14% scored 3, and only 23% had a score of 0. After only a few weeks of ECT, however, 84% of patients had a score of 0 and 9% scored a 1.
“That’s one of the compelling clinical reasons to refer patients for ECT: This type of ECT is really good for treating suicidality,” Dr. Kellner noted.
Phase 2 was the more interesting part of the PRIDE study, he continued, because it evaluated in randomized fashion the efficacy and tolerability of a novel flexible, individualized strategy for as-needed maintenance ECT to sustain the mood improvement once remission was achieved. Dr. Kellner stressed that some form of maintenance therapy is essential post ECT-induced remission.
“It’s unreasonable to expect that ECT could cure the patients’ underlying illness and protect them from getting sick again for the rest of their lives. One has to understand this is a recurrent episodic illness that we’re treating. What ECT does is treat the current episode, and it does it better and more thoroughly than any other treatment in psychiatry,” Dr. Kellner said.
Post-ECT relapse rates are clearly higher in the modern era, which makes a compelling case for developing safe and effective maintenance strategies.
“We don’t quite understand why relapse rates are higher today. My belief is that for patients who come to ECT, their illness has been destabilized by having been on multiple trials of antidepressant medications beforehand. It may also be that the forms of ECT that we’re using today are somewhat less potent than the ones used in previous decades,” Dr. Kellner conceded.
He and his coinvestigators named their investigational maintenance ECT strategy STABLE, for Symptom-Titrated, Algorithm-Based Longitudinal ECT. It’s a complex algorithm described in detail in a published report (Am J Psychiatry. 2016 Nov 1; 173[11]:1110-18). Basically, it consisted of four mandatory additional ECT sessions administered once weekly for the first month post remission, followed by either one, two, or no ECT sessions per week based upon evidence of deterioration as expressed in HAM-D24 scores.
In phase 2 of PRIDE, 128 remitters from phase 1 were continued on venlafaxine at a mean dose of 192 mg/day. In addition, they were placed on lithium, achieving a mean lithium level of 0.53 mEq/L, which Dr. Kellner deemed “low but reasonable.” These 128 patients were then randomized to the STABLE arm of flexibly administered ECT or to medication only and were followed prospectively for 6 months.
The primary endpoint was change in HAM-D24 score over the course of 6 months. From a baseline mean total score of 6, the mean score climbed to 8.4 in the medication-only group while dropping to 4.2 in the STABLE arm.
“At every time point along the way in the 6-month course of phase 2 of the illness, patients who were in the STABLE arm were less symptomatic and doing better than patients in the medication-only arm,” Dr. Kellner observed.
The key secondary efficacy outcome was change in the Clinical Global Impressions Severity scale. At follow-up, the patients in the flexible ECT plus medication arm were 5.2 times more likely to be rated “not ill at all” than were those on pharmacotherapy only.
In addition, relapse occurred in 20% of the medication-only group versus 13% in the STABLE group.
Global cognitive functioning as assessed by the Mini-Mental State Examination – crudely, in Dr. Kellner’s view – did not differ between the two groups at follow-up. Results of more sophisticated tests of multiple specific domains of cognition will be forthcoming.
Of note, two-thirds of patients in the STABLE arm required no additional ECT after their four continuation ECTs during the first month.
A swipe at ECT’s critics
Dr. Kellner called ECT “a fabulous therapy,” albeit one with a serious image problem.
“I think the problem with ECT is not that we don’t know all the details of how it works or what it does clinically, it’s really a sociopolitical issue about ECT not being adequately accepted. I break down this lack of acceptance into two forms: The first is passive lack of acceptance based on our profession not having embraced ECT and continuing to fail to embrace it. The other is the active propaganda against ECT that is promulgated by the Church of Scientology, which has taken on a life of its own now because of the Internet. ECT is still fought by the Church of Scientology. They fund lots of people to say incorrect nasty things about ECT to inappropriately frighten our patients,” Dr. Kellner charged.
He cautioned that proposed new Food and Drug Administration regulations governing ECT devices would greatly limit the use of ECT, restricting it to adults with treatment-resistant major depressive disorder or bipolar disorder or who require a rapid response. Other currently cleared indications would become off-label uses.
“With off-label ECT, the potential problem is practitioners may not be covered by their malpractice insurance. And the bigger issue is health insurers may not pay for it unless the ECT is for an on-label indication,” Dr. Kellner said.
He shared that he has found extremely helpful a colleague’s advice to demystify ECT by inviting a family member to witness a patient’s treatment session. That witness can then testify to others that what goes on bears no resemblance to what happens to Jack Nicholson in the classic film “One Flew Over the Cuckoo’s Nest.”
The PRIDE study was funded by the National Institute of Mental Health. Dr. Kellner reported having no financial conflicts of interest.
AT THE ECNP CONGRESS
Key clinical point:
Major finding: At 6 months follow-up, the mean score on the Hamilton Rating Scale for Depression was 4.2 points lower in the flexible maintenance ECT group than in the medication-only group, from a baseline score of 6.
Data source: Phase 2 of the PRIDE study randomized 128 elderly patients whose severe depression remitted in response to ECT to one of two maintenance treatment strategies.
Disclosures: The PRIDE study was funded by the National Institute of Mental Health. The presenter reported having no financial conflicts of interest.
Benzodiazepines and antidepressants do not impair cognition in midlife schizophrenia
PARIS – Cumulative lifetime exposure to benzodiazepines and antidepressants in the relatively modest amounts typically prescribed in schizophrenia did not appear to adversely affect patients’ cognition in midlife, according to a first-of-its-kind study from Finland.
“These findings are reassuring,” said Anja P. Hulkko, MD, a psychiatric resident at the University of Oulu (Finland).
“Similarly, you don’t need to worry about using benzodiazepines in schizophrenia if you are using small doses or for short periods to treat a specific condition like anxiety or sleeplessness,” Dr. Hulkko said at the annual congress of the European College of Neuropsychopharmacology.
These observations were among the key findings of her analysis from the Northern Finnish Birth Cohort 1966. This ongoing unique and naturalistic observational study – or actually, more than 20 different studies conducted in the same population – was designed to study risk factors involved in preterm birth and intrauterine growth retardation, as well as the consequences of these early adverse events on later morbidity and mortality. Data including maternal health during pregnancy have been prospectively collected from first prenatal contact at weeks 10-16 well into adulthood.
The 1966 cohort included 12,058 live births in the two northernmost provinces of Finland, which was 96% of all births there in that year. DNA samples were obtained from nearly 6,000 subjects in the cohort. After birth and again every 7-15 years, the offspring were examined and underwent clinical evaluation in which a wide range of phenotypic, behavioral, and demographic data were collected. Linkage to Finland’s comprehensive national registries provides investigators with up-to-date information about participants’ education, medication, hospitalizations, significant medical diagnoses, pensions, and death. These prospective data allow investigations of the importance of genetic, biologic, social, and behavioral risk factors in the emergence of various target diseases, one of which is schizophrenia.
Dr. Hulkko’s presentation focused on 60 patients with various schizophrenia spectrum disorders who underwent an extensive battery of neuropsychological tests at age 43, when they averaged 16.5 years of illness.
Lifetime cumulative exposure to benzodiazepines or antidepressants was not significantly associated with a global composite cognition score in a multivariate analysis adjusted for potential confounders including sex, age at schizophrenia onset, and lifetime psychiatric hospital treatment days.
The comprehensive test battery included the California Verbal Learning Test, the Abstraction Inhibition and Working Memory Task, Digit Span and Matrix Reasoning, verbal fluency and vocabulary testing, and the Visual Object Learning Test.
This, she said, is the first report to look at the cognitive impact of these drug classes in midlife schizophrenia. Long-term, high-dose use of benzodiazepines has in recent years been linked to increased risk for cognitive impairment in other populations, but that’s not ordinarily how those drugs are employed in schizophrenia.
In contrast to the new Finnish antidepressant and benzodiazepine findings, higher lifetime antipsychotic dose-years in the study population were recently reported by Dr. Hulkko’s senior coinvestigators to be significantly associated with a worse adjusted cognition score, with no difference between typical and atypical antipsychotics. “It is possible that large antipsychotic doses harm cognition in schizophrenia in the long term,” the researchers concluded (Psychiatry Res. 2017 Jan;247:130-8).
At the meeting, Dr. Hulkko presented new data on the cognitive impact of cumulative exposure to antipsychotics in the midlife schizophrenia cohort. The new finding is that having an antipsychotic-free period of at least 1 year at any point since treatment started an average of more than 16 years earlier was associated with a better adjusted cognitive performance.
The same was true for having an antipsychotic-free interval of at least 11 months directly before administration of the neuropsychological test battery. This is an encouraging finding because it suggests that an antipsychotic drug holiday might promote a measurable degree of cognitive recovery. But this observation must be viewed as hypothesis generating rather than conclusive, given the relatively small size of the study population, the observational nature of the study, and the possibility that the patients on a pretest antipsychotic break had less severe illness.
“Although we have tried to control for many factors, still, it’s possible that we cannot control for everything,” she noted. “It seems likely that both the illness itself and treatment are associated with the course of cognition.”
Kamilla W. Miskowiak, PhD, a neuropsychologist at the University of Copenhagen deemed the Finnish results “highly interesting.”
“This is reassuring, since many patients are worried about taking these medications [antidepressants and benzodiazepines] because of their potential negative effects on cognition. In contrast, long-term high-dose antipsychotic medication was associated with poorer cognitive outcome. This underscores the importance of close dose monitoring of antipsychotic medication for these patients to improve their cognitive outcome,” said Dr. Miskowiak, who was not involved in the study.
The study was supported by the Academy of Finland, the Finnish Cultural Foundation Lapland Regional Fund, and grants from various nonprofit foundations. Dr. Hulkko reported having no financial conflicts of interest.
PARIS – Cumulative lifetime exposure to benzodiazepines and antidepressants in the relatively modest amounts typically prescribed in schizophrenia did not appear to adversely affect patients’ cognition in midlife, according to a first-of-its-kind study from Finland.
“These findings are reassuring,” said Anja P. Hulkko, MD, a psychiatric resident at the University of Oulu (Finland).
“Similarly, you don’t need to worry about using benzodiazepines in schizophrenia if you are using small doses or for short periods to treat a specific condition like anxiety or sleeplessness,” Dr. Hulkko said at the annual congress of the European College of Neuropsychopharmacology.
These observations were among the key findings of her analysis from the Northern Finnish Birth Cohort 1966. This ongoing unique and naturalistic observational study – or actually, more than 20 different studies conducted in the same population – was designed to study risk factors involved in preterm birth and intrauterine growth retardation, as well as the consequences of these early adverse events on later morbidity and mortality. Data including maternal health during pregnancy have been prospectively collected from first prenatal contact at weeks 10-16 well into adulthood.
The 1966 cohort included 12,058 live births in the two northernmost provinces of Finland, which was 96% of all births there in that year. DNA samples were obtained from nearly 6,000 subjects in the cohort. After birth and again every 7-15 years, the offspring were examined and underwent clinical evaluation in which a wide range of phenotypic, behavioral, and demographic data were collected. Linkage to Finland’s comprehensive national registries provides investigators with up-to-date information about participants’ education, medication, hospitalizations, significant medical diagnoses, pensions, and death. These prospective data allow investigations of the importance of genetic, biologic, social, and behavioral risk factors in the emergence of various target diseases, one of which is schizophrenia.
Dr. Hulkko’s presentation focused on 60 patients with various schizophrenia spectrum disorders who underwent an extensive battery of neuropsychological tests at age 43, when they averaged 16.5 years of illness.
Lifetime cumulative exposure to benzodiazepines or antidepressants was not significantly associated with a global composite cognition score in a multivariate analysis adjusted for potential confounders including sex, age at schizophrenia onset, and lifetime psychiatric hospital treatment days.
The comprehensive test battery included the California Verbal Learning Test, the Abstraction Inhibition and Working Memory Task, Digit Span and Matrix Reasoning, verbal fluency and vocabulary testing, and the Visual Object Learning Test.
This, she said, is the first report to look at the cognitive impact of these drug classes in midlife schizophrenia. Long-term, high-dose use of benzodiazepines has in recent years been linked to increased risk for cognitive impairment in other populations, but that’s not ordinarily how those drugs are employed in schizophrenia.
In contrast to the new Finnish antidepressant and benzodiazepine findings, higher lifetime antipsychotic dose-years in the study population were recently reported by Dr. Hulkko’s senior coinvestigators to be significantly associated with a worse adjusted cognition score, with no difference between typical and atypical antipsychotics. “It is possible that large antipsychotic doses harm cognition in schizophrenia in the long term,” the researchers concluded (Psychiatry Res. 2017 Jan;247:130-8).
At the meeting, Dr. Hulkko presented new data on the cognitive impact of cumulative exposure to antipsychotics in the midlife schizophrenia cohort. The new finding is that having an antipsychotic-free period of at least 1 year at any point since treatment started an average of more than 16 years earlier was associated with a better adjusted cognitive performance.
The same was true for having an antipsychotic-free interval of at least 11 months directly before administration of the neuropsychological test battery. This is an encouraging finding because it suggests that an antipsychotic drug holiday might promote a measurable degree of cognitive recovery. But this observation must be viewed as hypothesis generating rather than conclusive, given the relatively small size of the study population, the observational nature of the study, and the possibility that the patients on a pretest antipsychotic break had less severe illness.
“Although we have tried to control for many factors, still, it’s possible that we cannot control for everything,” she noted. “It seems likely that both the illness itself and treatment are associated with the course of cognition.”
Kamilla W. Miskowiak, PhD, a neuropsychologist at the University of Copenhagen deemed the Finnish results “highly interesting.”
“This is reassuring, since many patients are worried about taking these medications [antidepressants and benzodiazepines] because of their potential negative effects on cognition. In contrast, long-term high-dose antipsychotic medication was associated with poorer cognitive outcome. This underscores the importance of close dose monitoring of antipsychotic medication for these patients to improve their cognitive outcome,” said Dr. Miskowiak, who was not involved in the study.
The study was supported by the Academy of Finland, the Finnish Cultural Foundation Lapland Regional Fund, and grants from various nonprofit foundations. Dr. Hulkko reported having no financial conflicts of interest.
PARIS – Cumulative lifetime exposure to benzodiazepines and antidepressants in the relatively modest amounts typically prescribed in schizophrenia did not appear to adversely affect patients’ cognition in midlife, according to a first-of-its-kind study from Finland.
“These findings are reassuring,” said Anja P. Hulkko, MD, a psychiatric resident at the University of Oulu (Finland).
“Similarly, you don’t need to worry about using benzodiazepines in schizophrenia if you are using small doses or for short periods to treat a specific condition like anxiety or sleeplessness,” Dr. Hulkko said at the annual congress of the European College of Neuropsychopharmacology.
These observations were among the key findings of her analysis from the Northern Finnish Birth Cohort 1966. This ongoing unique and naturalistic observational study – or actually, more than 20 different studies conducted in the same population – was designed to study risk factors involved in preterm birth and intrauterine growth retardation, as well as the consequences of these early adverse events on later morbidity and mortality. Data including maternal health during pregnancy have been prospectively collected from first prenatal contact at weeks 10-16 well into adulthood.
The 1966 cohort included 12,058 live births in the two northernmost provinces of Finland, which was 96% of all births there in that year. DNA samples were obtained from nearly 6,000 subjects in the cohort. After birth and again every 7-15 years, the offspring were examined and underwent clinical evaluation in which a wide range of phenotypic, behavioral, and demographic data were collected. Linkage to Finland’s comprehensive national registries provides investigators with up-to-date information about participants’ education, medication, hospitalizations, significant medical diagnoses, pensions, and death. These prospective data allow investigations of the importance of genetic, biologic, social, and behavioral risk factors in the emergence of various target diseases, one of which is schizophrenia.
Dr. Hulkko’s presentation focused on 60 patients with various schizophrenia spectrum disorders who underwent an extensive battery of neuropsychological tests at age 43, when they averaged 16.5 years of illness.
Lifetime cumulative exposure to benzodiazepines or antidepressants was not significantly associated with a global composite cognition score in a multivariate analysis adjusted for potential confounders including sex, age at schizophrenia onset, and lifetime psychiatric hospital treatment days.
The comprehensive test battery included the California Verbal Learning Test, the Abstraction Inhibition and Working Memory Task, Digit Span and Matrix Reasoning, verbal fluency and vocabulary testing, and the Visual Object Learning Test.
This, she said, is the first report to look at the cognitive impact of these drug classes in midlife schizophrenia. Long-term, high-dose use of benzodiazepines has in recent years been linked to increased risk for cognitive impairment in other populations, but that’s not ordinarily how those drugs are employed in schizophrenia.
In contrast to the new Finnish antidepressant and benzodiazepine findings, higher lifetime antipsychotic dose-years in the study population were recently reported by Dr. Hulkko’s senior coinvestigators to be significantly associated with a worse adjusted cognition score, with no difference between typical and atypical antipsychotics. “It is possible that large antipsychotic doses harm cognition in schizophrenia in the long term,” the researchers concluded (Psychiatry Res. 2017 Jan;247:130-8).
At the meeting, Dr. Hulkko presented new data on the cognitive impact of cumulative exposure to antipsychotics in the midlife schizophrenia cohort. The new finding is that having an antipsychotic-free period of at least 1 year at any point since treatment started an average of more than 16 years earlier was associated with a better adjusted cognitive performance.
The same was true for having an antipsychotic-free interval of at least 11 months directly before administration of the neuropsychological test battery. This is an encouraging finding because it suggests that an antipsychotic drug holiday might promote a measurable degree of cognitive recovery. But this observation must be viewed as hypothesis generating rather than conclusive, given the relatively small size of the study population, the observational nature of the study, and the possibility that the patients on a pretest antipsychotic break had less severe illness.
“Although we have tried to control for many factors, still, it’s possible that we cannot control for everything,” she noted. “It seems likely that both the illness itself and treatment are associated with the course of cognition.”
Kamilla W. Miskowiak, PhD, a neuropsychologist at the University of Copenhagen deemed the Finnish results “highly interesting.”
“This is reassuring, since many patients are worried about taking these medications [antidepressants and benzodiazepines] because of their potential negative effects on cognition. In contrast, long-term high-dose antipsychotic medication was associated with poorer cognitive outcome. This underscores the importance of close dose monitoring of antipsychotic medication for these patients to improve their cognitive outcome,” said Dr. Miskowiak, who was not involved in the study.
The study was supported by the Academy of Finland, the Finnish Cultural Foundation Lapland Regional Fund, and grants from various nonprofit foundations. Dr. Hulkko reported having no financial conflicts of interest.
AT THE ECNP CONGRESS
Key clinical point:
Major finding: As-needed prescription of antidepressants and benzodiazepines in the dose typically used in schizophrenia does not impair midlife cognition.
Data source: An ongoing naturalistic, observational, general population-based study of more than 12,000 babies born in Northern Finland in 1966 from fetal life through middle age.
Disclosures: The study was supported by the Academy of Finland, the Finnish Cultural Foundation Lapland Regional Fund, and grants from various nonprofit charitable foundations. The presenter reported having no financial conflicts of interest.
Antipsychotics may reduce mortality in schizophrenia
PARIS – All-cause mortality was 46% lower while schizophrenia patients were on antipsychotic agents than when off therapy in a nationwide observational study of nearly 30,000 Swedes with schizophrenia – and the biggest risk reduction occurred in patients on second-generation long-acting injectables, according to Jari Tiihonen, MD, PhD.
“The guidelines say there are special situations where you should consider using depot antipsychotics. I think the guidelines got it wrong. I think the right way to think of it is there are special situations where you might consider oral agents; otherwise you should use long-acting injectables,” said Dr. Tiihonen, who holds joint appointments as professor of psychiatry at the University of Eastern Finland in Kuopio and the Karolinska Institute in Stockholm.

During a mean follow-up of 5.7 years, 8.4% of the patients died. In a multivariate Cox regression analysis adjusted for 20 potential confounding variables, including age, sex, time since diagnosis, education, and comorbid conditions, the all-cause mortality was 46% lower when patients were on an antipsychotic agent than when off therapy.
The lowest mortality was observed in patients on a second-generation long-acting injectable (LAI) antipsychotic agent. Specifically, all-cause mortality was reduced by 89% while patients were on once-monthly palpiperidone palmitate LAI (Invega Sustenna) than during periods when they were not using an antipsychotic. They were 69% less likely to die while on risperidone LAI (Risperdal Consta), and 77% less likely to die while taking oral aripiprazole (Abilify).
In a pairwise comparison between the LAI and oral versions of antipsychotic agents, the LAIs were associated with a 33% lower mortality than the equivalent oral drugs.
Compared with periods of nonuse of any antipsychotic agent, the adjusted risk of mortality was reduced by 85% while patients were on a second-generation LAI, by 47% when on second-generation oral agents, by 36% with first-generation LAIs, and by 34% for first-generation oral antipsychotics.
Also at the ECNP congress, Dr. Tiihonen presented a separate analysis of the same cohort of 29,823 patients, this time examining the real-world effectiveness of various antipsychotic agents in preventing relapse, treatment failure, and rehospitalization. For this purpose, he and his coinvestigators performed a within-individual analysis in which each patient served as his or her own control. The advantage of this analytic strategy is that it corrects for selection bias and other forms of residual confounding inherent in observational studies.
During 7 years of follow-up, 44% of patients were rehospitalized. The risk of psychiatric rehospitalization was lowest when patients were on monotherapy with once-monthly palpiperidone LAI, as evidenced by a 49% reduction, compared with no use of antipsychotic agents. Clozapine (Clozaril) monotherapy and zuclopenthixol LAI (Clopixol) were similarly effective, with each conferring a 47% reduction in rehospitalization risk, compared with periods off therapy. Perphenazine LAI (Trilafon) and olanzapine LAI (Relprevv) each brought a 42% reduction in rehospitalization risk, while the oral version of perphenazine was associated with a 14% risk reduction.
In contrast, the risk of rehospitalization when patients were on oral flupentixol (Depixol) or quetiapine (Seroquel) was not significantly different from when they were off antipsychotic agents altogether.
Collectively, the LAIs were associated with a 22% lower risk of rehospitalization, compared with their equivalent oral versions. And among the 4,603 patients newly diagnosed with schizophrenia in 2006, the LAIs were associated with a more substantial 32% risk reduction, compared with their oral formulations.
Oral clozapine had the lowest treatment failure rate: 42% less than with oral olanzapine (Zyprexa), which served as the reference standard because it was the most commonly used antipsychotic agent. Treatment failure was defined as psychiatric rehospitalization, attempted suicide, treatment discontinuation or a switch to another antipsychotic agent, or death. The various LAIs were associated with 20%-35% lower treatment failure rates than oral olanzapine.
Dr. Tiihonen’s relapse prevention analysis has been published (JAMA Psychiatry. 2017 Jul 1;74[7]:686-93).
The two studies were funded by Janssen-Cilag. Dr. Tiihonen reported serving as a consultant to that pharmaceutical company and a half-dozen others.
PARIS – All-cause mortality was 46% lower while schizophrenia patients were on antipsychotic agents than when off therapy in a nationwide observational study of nearly 30,000 Swedes with schizophrenia – and the biggest risk reduction occurred in patients on second-generation long-acting injectables, according to Jari Tiihonen, MD, PhD.
“The guidelines say there are special situations where you should consider using depot antipsychotics. I think the guidelines got it wrong. I think the right way to think of it is there are special situations where you might consider oral agents; otherwise you should use long-acting injectables,” said Dr. Tiihonen, who holds joint appointments as professor of psychiatry at the University of Eastern Finland in Kuopio and the Karolinska Institute in Stockholm.

During a mean follow-up of 5.7 years, 8.4% of the patients died. In a multivariate Cox regression analysis adjusted for 20 potential confounding variables, including age, sex, time since diagnosis, education, and comorbid conditions, the all-cause mortality was 46% lower when patients were on an antipsychotic agent than when off therapy.
The lowest mortality was observed in patients on a second-generation long-acting injectable (LAI) antipsychotic agent. Specifically, all-cause mortality was reduced by 89% while patients were on once-monthly palpiperidone palmitate LAI (Invega Sustenna) than during periods when they were not using an antipsychotic. They were 69% less likely to die while on risperidone LAI (Risperdal Consta), and 77% less likely to die while taking oral aripiprazole (Abilify).
In a pairwise comparison between the LAI and oral versions of antipsychotic agents, the LAIs were associated with a 33% lower mortality than the equivalent oral drugs.
Compared with periods of nonuse of any antipsychotic agent, the adjusted risk of mortality was reduced by 85% while patients were on a second-generation LAI, by 47% when on second-generation oral agents, by 36% with first-generation LAIs, and by 34% for first-generation oral antipsychotics.
Also at the ECNP congress, Dr. Tiihonen presented a separate analysis of the same cohort of 29,823 patients, this time examining the real-world effectiveness of various antipsychotic agents in preventing relapse, treatment failure, and rehospitalization. For this purpose, he and his coinvestigators performed a within-individual analysis in which each patient served as his or her own control. The advantage of this analytic strategy is that it corrects for selection bias and other forms of residual confounding inherent in observational studies.
During 7 years of follow-up, 44% of patients were rehospitalized. The risk of psychiatric rehospitalization was lowest when patients were on monotherapy with once-monthly palpiperidone LAI, as evidenced by a 49% reduction, compared with no use of antipsychotic agents. Clozapine (Clozaril) monotherapy and zuclopenthixol LAI (Clopixol) were similarly effective, with each conferring a 47% reduction in rehospitalization risk, compared with periods off therapy. Perphenazine LAI (Trilafon) and olanzapine LAI (Relprevv) each brought a 42% reduction in rehospitalization risk, while the oral version of perphenazine was associated with a 14% risk reduction.
In contrast, the risk of rehospitalization when patients were on oral flupentixol (Depixol) or quetiapine (Seroquel) was not significantly different from when they were off antipsychotic agents altogether.
Collectively, the LAIs were associated with a 22% lower risk of rehospitalization, compared with their equivalent oral versions. And among the 4,603 patients newly diagnosed with schizophrenia in 2006, the LAIs were associated with a more substantial 32% risk reduction, compared with their oral formulations.
Oral clozapine had the lowest treatment failure rate: 42% less than with oral olanzapine (Zyprexa), which served as the reference standard because it was the most commonly used antipsychotic agent. Treatment failure was defined as psychiatric rehospitalization, attempted suicide, treatment discontinuation or a switch to another antipsychotic agent, or death. The various LAIs were associated with 20%-35% lower treatment failure rates than oral olanzapine.
Dr. Tiihonen’s relapse prevention analysis has been published (JAMA Psychiatry. 2017 Jul 1;74[7]:686-93).
The two studies were funded by Janssen-Cilag. Dr. Tiihonen reported serving as a consultant to that pharmaceutical company and a half-dozen others.
PARIS – All-cause mortality was 46% lower while schizophrenia patients were on antipsychotic agents than when off therapy in a nationwide observational study of nearly 30,000 Swedes with schizophrenia – and the biggest risk reduction occurred in patients on second-generation long-acting injectables, according to Jari Tiihonen, MD, PhD.
“The guidelines say there are special situations where you should consider using depot antipsychotics. I think the guidelines got it wrong. I think the right way to think of it is there are special situations where you might consider oral agents; otherwise you should use long-acting injectables,” said Dr. Tiihonen, who holds joint appointments as professor of psychiatry at the University of Eastern Finland in Kuopio and the Karolinska Institute in Stockholm.

During a mean follow-up of 5.7 years, 8.4% of the patients died. In a multivariate Cox regression analysis adjusted for 20 potential confounding variables, including age, sex, time since diagnosis, education, and comorbid conditions, the all-cause mortality was 46% lower when patients were on an antipsychotic agent than when off therapy.
The lowest mortality was observed in patients on a second-generation long-acting injectable (LAI) antipsychotic agent. Specifically, all-cause mortality was reduced by 89% while patients were on once-monthly palpiperidone palmitate LAI (Invega Sustenna) than during periods when they were not using an antipsychotic. They were 69% less likely to die while on risperidone LAI (Risperdal Consta), and 77% less likely to die while taking oral aripiprazole (Abilify).
In a pairwise comparison between the LAI and oral versions of antipsychotic agents, the LAIs were associated with a 33% lower mortality than the equivalent oral drugs.
Compared with periods of nonuse of any antipsychotic agent, the adjusted risk of mortality was reduced by 85% while patients were on a second-generation LAI, by 47% when on second-generation oral agents, by 36% with first-generation LAIs, and by 34% for first-generation oral antipsychotics.
Also at the ECNP congress, Dr. Tiihonen presented a separate analysis of the same cohort of 29,823 patients, this time examining the real-world effectiveness of various antipsychotic agents in preventing relapse, treatment failure, and rehospitalization. For this purpose, he and his coinvestigators performed a within-individual analysis in which each patient served as his or her own control. The advantage of this analytic strategy is that it corrects for selection bias and other forms of residual confounding inherent in observational studies.
During 7 years of follow-up, 44% of patients were rehospitalized. The risk of psychiatric rehospitalization was lowest when patients were on monotherapy with once-monthly palpiperidone LAI, as evidenced by a 49% reduction, compared with no use of antipsychotic agents. Clozapine (Clozaril) monotherapy and zuclopenthixol LAI (Clopixol) were similarly effective, with each conferring a 47% reduction in rehospitalization risk, compared with periods off therapy. Perphenazine LAI (Trilafon) and olanzapine LAI (Relprevv) each brought a 42% reduction in rehospitalization risk, while the oral version of perphenazine was associated with a 14% risk reduction.
In contrast, the risk of rehospitalization when patients were on oral flupentixol (Depixol) or quetiapine (Seroquel) was not significantly different from when they were off antipsychotic agents altogether.
Collectively, the LAIs were associated with a 22% lower risk of rehospitalization, compared with their equivalent oral versions. And among the 4,603 patients newly diagnosed with schizophrenia in 2006, the LAIs were associated with a more substantial 32% risk reduction, compared with their oral formulations.
Oral clozapine had the lowest treatment failure rate: 42% less than with oral olanzapine (Zyprexa), which served as the reference standard because it was the most commonly used antipsychotic agent. Treatment failure was defined as psychiatric rehospitalization, attempted suicide, treatment discontinuation or a switch to another antipsychotic agent, or death. The various LAIs were associated with 20%-35% lower treatment failure rates than oral olanzapine.
Dr. Tiihonen’s relapse prevention analysis has been published (JAMA Psychiatry. 2017 Jul 1;74[7]:686-93).
The two studies were funded by Janssen-Cilag. Dr. Tiihonen reported serving as a consultant to that pharmaceutical company and a half-dozen others.
AT THE ECNP CONGRESS
Key clinical point:
Major finding: The all-cause mortality rate was 85% lower when patients with schizophrenia were on a second-generation long-acting injectable antipsychotic than when not taking an antipsychotic.
Data source: An observational study of the association between antipsychotic medication use and all-cause mortality over the course of more than half a decade in nearly 30,000 Swedish schizophrenia patients.
Disclosures: The study was funded by Janssen-Cilag. The presenter reported serving as a consultant to that pharmaceutical company and a half-dozen others.
Latest on dupilumab for atopic dermatitis: The who and how
GENEVA – The novel interleukin-4 and IL-13 signaling blocker dupilumab displayed consistently strong efficacy across all patient subgroups in a new analysis from the landmark 52-week CHRONOS trial, Andrew Blauvelt, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
“Dupilumab with concomitant topical corticosteroids improved signs and symptoms of atopic dermatitis, compared with placebo injections regardless of age, sex, BMI, or prior history of asthma, allergic rhinitis, or food allergies,” he said. Indeed, he quipped that the biologic proved to be “boring” in its broad effectiveness.
CHRONOS was a 14-country, 1-year, randomized, double-blind, placebo-controlled, phase 3 clinical trial in which 740 adults with moderate to very severe AD were assigned to mid-potency topical corticosteroids along with topical calcineurin inhibitors as needed in steroid-sensitive locations, then randomized 3:1:3 to either subcutaneous dupilumab (Dupixent) at 300 mg once weekly, placebo injections, or dupilumab at 300 mg every 2 weeks on top of the topical therapy.
Dupilumab, a fully human monoclonal antibody, was approved by the Food and Drug Administration in March 2017 at a loading dose of 600 mg, followed by 300 mg every 2 weeks for treatment of adults with moderate to severe AD. The biologic agent is soon expected to be approved across the European Union as well. CHRONOS was the first major trial designed to replicate how the biologic would likely be utilized in real-world clinical practice: that is, in conjunction with rather than as an alternative to topical therapy. Earlier pivotal phase 3 trials were required by regulatory agencies to pit dupilumab monotherapy against placebo.
CHRONOS participants were “a very tough crowd, so to speak,” the dermatologist recalled. Their median body surface area of involvement was a whopping 55%, 60% were men, the median baseline EASI (Eczema Area and Severity Index) score was 30, and fully half of patients had an IGA (Investigator’s Global Assessment) score of 4, indicative of severe disease. Plus, comorbid atopic/allergic diseases were common: half of subjects had a history of asthma, nearly half had a history of allergic rhinitis, and one-third had a history of food allergies.
Dr. Blauvelt previously presented the primary results of CHRONOS without the new subgroup analysis at the American Academy of Dermatology 2017 meeting. The primary results were subsequently published (Lancet. 2017 Jun 10;389[10086]:2287-303). To recap, the bottom line: 39% of dupilumab-treated patients achieved an IGA score of 0/1 – that is, clear or almost clear – coupled with at least a 2-point improvement from baseline at week 16, versus 12% of controls on topical steroids plus placebo injections. That’s a strikingly impressive performance, according to the dermatologist.
“An IGA of 0/1 in AD is a very high bar. It tends to correlated with an EASI 90,” Dr. Blauvelt observed.
Rates of EASI 75 – meaning at least a 75% improvement from baseline EASI scores – were achieved in 64% of patients on weekly dupilumab plus topical steroids, 69% with dupilumab every 2 weeks plus topicals, and 23% on topical steroids plus placebo. These efficacy results were essentially mirrored at week 52, with no significant changes in response rates from week 16 to week 52.
In another new finding from CHRONOS presented by Dr. Blauvelt at the EADV congress, itch also improved sharply across the board in dupilumab-treated patients, regardless of their baseline demographic and other characteristics, meaning there’s no point for clinicians to reserve the biologic for selected subgroups of patients with moderate or severe AD. From a mean baseline score of 7.6 on the 10-point peak pruritus numerical rating scale, at least a 3-point improvement was achieved in 56% of patients on biweekly dupilumab plus topical steroids, 43% on weekly dupilumab and topical steroids, and in 16% on topical steroids plus placebo injections.
Of note, 84% of patients in the two dupilumab arms remained in the CHRONOS trial through the full 52 weeks. That’s a high retention rate for a 1-year study involving a disease with a major adverse quality of life impact. In contrast, only about two-thirds of controls on topical therapy plus placebo injections stuck with the study for the duration.
The high retention rate may have had much to do with dupilumab’s generally favorable safely profile, as seen not only in CHRONOS but in earlier trials. No new safety concerns arose during the 52-week study. The dupilumab and control groups had similar rates of most side effects, with just a few exceptions: nonherpetic skin infections and worsening AD occurred more frequently in controls on topical steroids alone, while mild injection site reactions were twice as common in patients who got dupilumab than in those who received placebo injections.
Also, conjunctivitis occurred in 14% of patients on weekly dupilumab and 19% with biweekly dupilumab, compared with 8% of controls. “To me, conjunctivitis is the only significant side effect for this drug that we’ve seen thus far,” Dr. Blauvelt commented. The mechanism of the conjunctivitis is unknown. It’s clear, however, that the rate goes up with duration on therapy.
“Most of the cases have been mild to moderate and have not interfered with therapy. I’ve treated probably 80 patients with dupilumab, and I’ve had only 1 who had to stop due to her eyes,” the dermatologist recalled. The condition often responds to wetting eye drops, although topical corticosteroid eye drops are required sometimes, he added.
CAFE was a double-blind, randomized, placebo-controlled trial including 325 European patients with moderate or severe AD, all of whom were on a mid-potency topical corticosteroid with or without a topical calcineurin inhibitor. Two-thirds had previously been on cyclosporine with poor results; the drug was contraindicated in the others. Participants were randomized to subcutaneous injections of dupilumab at 300 mg weekly or biweekly or to placebo injections.
The primary outcome – an EASI-75 response at 16 weeks – was achieved in 59% of patients on weekly dupilumab, 63% of those on biweekly dupilumab, and 30% of controls on topical steroids plus placebo injections, according to Dr. De Bruin-Weller of the University Medical Center at Utrecht, the Netherlands.
A clinically meaningful 4-point or greater improvement from baseline in the Dermatology Life Quality Index occurred at 16 weeks in 79% and 88% of patients on dupilumab weekly or biweekly, respectively, compared with 44% of controls.
And among the three-quarters of CAFE participants reporting baseline moderate or severe pain or discomfort from AD, 55% of those on weekly dupilumab had none at all as assessed by the EQ-5D Pain/Discomfort Questionnaire at week 16, as did 64% on dupilumab every 2 weeks and 28% of controls on topical steroids.
At the prestigious joint EADV/AAD session of the European congress, Lawrence F. Eichenfield, MD, hailed dupilumab as evidence that “the systemic therapy revolution in atopic dermatitis is certainly happening,” with a plethora of additional agents targeting various key disease pathways now working their way through the developmental pipeline.
A key remaining question about dupilumab is, what about the safety and efficacy of the drug in the pediatric population, for whom the drug is not currently approved? Separate phase 3 randomized trials addressing this issue are now enrolling 6- to 11-year-olds and 12- to 17-year-olds.
Dr. Eichenfield is optimistic about the outcomes of these ongoing pediatric trials on the basis of an open-label, phase 2a, proof of concept study in 6- to 17-year-olds presented in a late-breaking research session at the 2017 annual meeting of the AAD. The pharmacokinetics were very similar to those seen in adults. Moreover, EASI scores improved by 32%-51% after just a single, weight-based injection of dupilumab and by up to 70% following four consecutive weekly injections.
“This is very, very exciting information. Whether the drug could actually be disease modifying is obviously a big question for us in pediatric dermatology,” observed Dr. Eichenfield, who is chief of pediatric and adolescent dermatology at Rady Children’s Hospital–San Diego and professor of dermatology and pediatrics at the University of California, San Diego.
He highlighted two important safety considerations regarding dupilumab: patients on the biologic should not be given any live vaccines, and the same IL-4 and IL-13 cytokine pathways targeted by dupilumab in patients with AD also are key in other atopic and allergic diseases. Indeed, the biologic showed positive outcomes in a recent pivotal phase 3 clinical trial for patients with uncontrolled persistent asthma, and an application for an expanded indication for dupilumab in such patients is expected to be filed with the FDA later in 2017. Dupilumab is also in phase 3 for patients with nasal polyps and in phase 2 studies for eosinophilic esophagitis.
“One of the interesting things about dupilumab is that we in dermatology are going to be in a situation where we’re able to impact diseases other than the single disease that the medication has been approved for,” Dr. Eichenfield said.
However, until such time as dupilumab actually receives an expanded indication for asthma and the details of how best to use the biologic in AD patients with that comorbidity have been worked out, it’s important for dermatologists prescribing dupilumab in those dual-diagnosis patients to discuss with them the necessity of staying on their asthma medications even though their skin is much improved, they’re feeling good, and their asthma seems to be doing better. During the dupilumab clinical trials program for AD, a fatal asthma exacerbation occurred in a patient with comorbid asthma who stopped taking asthma medications, he noted.
The CHRONOS and CAFE trials were funded by Sanofi and Regeneron Pharmaceuticals. Dr. Blauvelt, Dr. De Bruin-Weller, and Dr. Eichenfield reported serving as consultants to and researchers for those pharmaceutical companies and numerous others.
GENEVA – The novel interleukin-4 and IL-13 signaling blocker dupilumab displayed consistently strong efficacy across all patient subgroups in a new analysis from the landmark 52-week CHRONOS trial, Andrew Blauvelt, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
“Dupilumab with concomitant topical corticosteroids improved signs and symptoms of atopic dermatitis, compared with placebo injections regardless of age, sex, BMI, or prior history of asthma, allergic rhinitis, or food allergies,” he said. Indeed, he quipped that the biologic proved to be “boring” in its broad effectiveness.
CHRONOS was a 14-country, 1-year, randomized, double-blind, placebo-controlled, phase 3 clinical trial in which 740 adults with moderate to very severe AD were assigned to mid-potency topical corticosteroids along with topical calcineurin inhibitors as needed in steroid-sensitive locations, then randomized 3:1:3 to either subcutaneous dupilumab (Dupixent) at 300 mg once weekly, placebo injections, or dupilumab at 300 mg every 2 weeks on top of the topical therapy.
Dupilumab, a fully human monoclonal antibody, was approved by the Food and Drug Administration in March 2017 at a loading dose of 600 mg, followed by 300 mg every 2 weeks for treatment of adults with moderate to severe AD. The biologic agent is soon expected to be approved across the European Union as well. CHRONOS was the first major trial designed to replicate how the biologic would likely be utilized in real-world clinical practice: that is, in conjunction with rather than as an alternative to topical therapy. Earlier pivotal phase 3 trials were required by regulatory agencies to pit dupilumab monotherapy against placebo.
CHRONOS participants were “a very tough crowd, so to speak,” the dermatologist recalled. Their median body surface area of involvement was a whopping 55%, 60% were men, the median baseline EASI (Eczema Area and Severity Index) score was 30, and fully half of patients had an IGA (Investigator’s Global Assessment) score of 4, indicative of severe disease. Plus, comorbid atopic/allergic diseases were common: half of subjects had a history of asthma, nearly half had a history of allergic rhinitis, and one-third had a history of food allergies.
Dr. Blauvelt previously presented the primary results of CHRONOS without the new subgroup analysis at the American Academy of Dermatology 2017 meeting. The primary results were subsequently published (Lancet. 2017 Jun 10;389[10086]:2287-303). To recap, the bottom line: 39% of dupilumab-treated patients achieved an IGA score of 0/1 – that is, clear or almost clear – coupled with at least a 2-point improvement from baseline at week 16, versus 12% of controls on topical steroids plus placebo injections. That’s a strikingly impressive performance, according to the dermatologist.
“An IGA of 0/1 in AD is a very high bar. It tends to correlated with an EASI 90,” Dr. Blauvelt observed.
Rates of EASI 75 – meaning at least a 75% improvement from baseline EASI scores – were achieved in 64% of patients on weekly dupilumab plus topical steroids, 69% with dupilumab every 2 weeks plus topicals, and 23% on topical steroids plus placebo. These efficacy results were essentially mirrored at week 52, with no significant changes in response rates from week 16 to week 52.
In another new finding from CHRONOS presented by Dr. Blauvelt at the EADV congress, itch also improved sharply across the board in dupilumab-treated patients, regardless of their baseline demographic and other characteristics, meaning there’s no point for clinicians to reserve the biologic for selected subgroups of patients with moderate or severe AD. From a mean baseline score of 7.6 on the 10-point peak pruritus numerical rating scale, at least a 3-point improvement was achieved in 56% of patients on biweekly dupilumab plus topical steroids, 43% on weekly dupilumab and topical steroids, and in 16% on topical steroids plus placebo injections.
Of note, 84% of patients in the two dupilumab arms remained in the CHRONOS trial through the full 52 weeks. That’s a high retention rate for a 1-year study involving a disease with a major adverse quality of life impact. In contrast, only about two-thirds of controls on topical therapy plus placebo injections stuck with the study for the duration.
The high retention rate may have had much to do with dupilumab’s generally favorable safely profile, as seen not only in CHRONOS but in earlier trials. No new safety concerns arose during the 52-week study. The dupilumab and control groups had similar rates of most side effects, with just a few exceptions: nonherpetic skin infections and worsening AD occurred more frequently in controls on topical steroids alone, while mild injection site reactions were twice as common in patients who got dupilumab than in those who received placebo injections.
Also, conjunctivitis occurred in 14% of patients on weekly dupilumab and 19% with biweekly dupilumab, compared with 8% of controls. “To me, conjunctivitis is the only significant side effect for this drug that we’ve seen thus far,” Dr. Blauvelt commented. The mechanism of the conjunctivitis is unknown. It’s clear, however, that the rate goes up with duration on therapy.
“Most of the cases have been mild to moderate and have not interfered with therapy. I’ve treated probably 80 patients with dupilumab, and I’ve had only 1 who had to stop due to her eyes,” the dermatologist recalled. The condition often responds to wetting eye drops, although topical corticosteroid eye drops are required sometimes, he added.
CAFE was a double-blind, randomized, placebo-controlled trial including 325 European patients with moderate or severe AD, all of whom were on a mid-potency topical corticosteroid with or without a topical calcineurin inhibitor. Two-thirds had previously been on cyclosporine with poor results; the drug was contraindicated in the others. Participants were randomized to subcutaneous injections of dupilumab at 300 mg weekly or biweekly or to placebo injections.
The primary outcome – an EASI-75 response at 16 weeks – was achieved in 59% of patients on weekly dupilumab, 63% of those on biweekly dupilumab, and 30% of controls on topical steroids plus placebo injections, according to Dr. De Bruin-Weller of the University Medical Center at Utrecht, the Netherlands.
A clinically meaningful 4-point or greater improvement from baseline in the Dermatology Life Quality Index occurred at 16 weeks in 79% and 88% of patients on dupilumab weekly or biweekly, respectively, compared with 44% of controls.
And among the three-quarters of CAFE participants reporting baseline moderate or severe pain or discomfort from AD, 55% of those on weekly dupilumab had none at all as assessed by the EQ-5D Pain/Discomfort Questionnaire at week 16, as did 64% on dupilumab every 2 weeks and 28% of controls on topical steroids.
At the prestigious joint EADV/AAD session of the European congress, Lawrence F. Eichenfield, MD, hailed dupilumab as evidence that “the systemic therapy revolution in atopic dermatitis is certainly happening,” with a plethora of additional agents targeting various key disease pathways now working their way through the developmental pipeline.
A key remaining question about dupilumab is, what about the safety and efficacy of the drug in the pediatric population, for whom the drug is not currently approved? Separate phase 3 randomized trials addressing this issue are now enrolling 6- to 11-year-olds and 12- to 17-year-olds.
Dr. Eichenfield is optimistic about the outcomes of these ongoing pediatric trials on the basis of an open-label, phase 2a, proof of concept study in 6- to 17-year-olds presented in a late-breaking research session at the 2017 annual meeting of the AAD. The pharmacokinetics were very similar to those seen in adults. Moreover, EASI scores improved by 32%-51% after just a single, weight-based injection of dupilumab and by up to 70% following four consecutive weekly injections.
“This is very, very exciting information. Whether the drug could actually be disease modifying is obviously a big question for us in pediatric dermatology,” observed Dr. Eichenfield, who is chief of pediatric and adolescent dermatology at Rady Children’s Hospital–San Diego and professor of dermatology and pediatrics at the University of California, San Diego.
He highlighted two important safety considerations regarding dupilumab: patients on the biologic should not be given any live vaccines, and the same IL-4 and IL-13 cytokine pathways targeted by dupilumab in patients with AD also are key in other atopic and allergic diseases. Indeed, the biologic showed positive outcomes in a recent pivotal phase 3 clinical trial for patients with uncontrolled persistent asthma, and an application for an expanded indication for dupilumab in such patients is expected to be filed with the FDA later in 2017. Dupilumab is also in phase 3 for patients with nasal polyps and in phase 2 studies for eosinophilic esophagitis.
“One of the interesting things about dupilumab is that we in dermatology are going to be in a situation where we’re able to impact diseases other than the single disease that the medication has been approved for,” Dr. Eichenfield said.
However, until such time as dupilumab actually receives an expanded indication for asthma and the details of how best to use the biologic in AD patients with that comorbidity have been worked out, it’s important for dermatologists prescribing dupilumab in those dual-diagnosis patients to discuss with them the necessity of staying on their asthma medications even though their skin is much improved, they’re feeling good, and their asthma seems to be doing better. During the dupilumab clinical trials program for AD, a fatal asthma exacerbation occurred in a patient with comorbid asthma who stopped taking asthma medications, he noted.
The CHRONOS and CAFE trials were funded by Sanofi and Regeneron Pharmaceuticals. Dr. Blauvelt, Dr. De Bruin-Weller, and Dr. Eichenfield reported serving as consultants to and researchers for those pharmaceutical companies and numerous others.
GENEVA – The novel interleukin-4 and IL-13 signaling blocker dupilumab displayed consistently strong efficacy across all patient subgroups in a new analysis from the landmark 52-week CHRONOS trial, Andrew Blauvelt, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
“Dupilumab with concomitant topical corticosteroids improved signs and symptoms of atopic dermatitis, compared with placebo injections regardless of age, sex, BMI, or prior history of asthma, allergic rhinitis, or food allergies,” he said. Indeed, he quipped that the biologic proved to be “boring” in its broad effectiveness.
CHRONOS was a 14-country, 1-year, randomized, double-blind, placebo-controlled, phase 3 clinical trial in which 740 adults with moderate to very severe AD were assigned to mid-potency topical corticosteroids along with topical calcineurin inhibitors as needed in steroid-sensitive locations, then randomized 3:1:3 to either subcutaneous dupilumab (Dupixent) at 300 mg once weekly, placebo injections, or dupilumab at 300 mg every 2 weeks on top of the topical therapy.
Dupilumab, a fully human monoclonal antibody, was approved by the Food and Drug Administration in March 2017 at a loading dose of 600 mg, followed by 300 mg every 2 weeks for treatment of adults with moderate to severe AD. The biologic agent is soon expected to be approved across the European Union as well. CHRONOS was the first major trial designed to replicate how the biologic would likely be utilized in real-world clinical practice: that is, in conjunction with rather than as an alternative to topical therapy. Earlier pivotal phase 3 trials were required by regulatory agencies to pit dupilumab monotherapy against placebo.
CHRONOS participants were “a very tough crowd, so to speak,” the dermatologist recalled. Their median body surface area of involvement was a whopping 55%, 60% were men, the median baseline EASI (Eczema Area and Severity Index) score was 30, and fully half of patients had an IGA (Investigator’s Global Assessment) score of 4, indicative of severe disease. Plus, comorbid atopic/allergic diseases were common: half of subjects had a history of asthma, nearly half had a history of allergic rhinitis, and one-third had a history of food allergies.
Dr. Blauvelt previously presented the primary results of CHRONOS without the new subgroup analysis at the American Academy of Dermatology 2017 meeting. The primary results were subsequently published (Lancet. 2017 Jun 10;389[10086]:2287-303). To recap, the bottom line: 39% of dupilumab-treated patients achieved an IGA score of 0/1 – that is, clear or almost clear – coupled with at least a 2-point improvement from baseline at week 16, versus 12% of controls on topical steroids plus placebo injections. That’s a strikingly impressive performance, according to the dermatologist.
“An IGA of 0/1 in AD is a very high bar. It tends to correlated with an EASI 90,” Dr. Blauvelt observed.
Rates of EASI 75 – meaning at least a 75% improvement from baseline EASI scores – were achieved in 64% of patients on weekly dupilumab plus topical steroids, 69% with dupilumab every 2 weeks plus topicals, and 23% on topical steroids plus placebo. These efficacy results were essentially mirrored at week 52, with no significant changes in response rates from week 16 to week 52.
In another new finding from CHRONOS presented by Dr. Blauvelt at the EADV congress, itch also improved sharply across the board in dupilumab-treated patients, regardless of their baseline demographic and other characteristics, meaning there’s no point for clinicians to reserve the biologic for selected subgroups of patients with moderate or severe AD. From a mean baseline score of 7.6 on the 10-point peak pruritus numerical rating scale, at least a 3-point improvement was achieved in 56% of patients on biweekly dupilumab plus topical steroids, 43% on weekly dupilumab and topical steroids, and in 16% on topical steroids plus placebo injections.
Of note, 84% of patients in the two dupilumab arms remained in the CHRONOS trial through the full 52 weeks. That’s a high retention rate for a 1-year study involving a disease with a major adverse quality of life impact. In contrast, only about two-thirds of controls on topical therapy plus placebo injections stuck with the study for the duration.
The high retention rate may have had much to do with dupilumab’s generally favorable safely profile, as seen not only in CHRONOS but in earlier trials. No new safety concerns arose during the 52-week study. The dupilumab and control groups had similar rates of most side effects, with just a few exceptions: nonherpetic skin infections and worsening AD occurred more frequently in controls on topical steroids alone, while mild injection site reactions were twice as common in patients who got dupilumab than in those who received placebo injections.
Also, conjunctivitis occurred in 14% of patients on weekly dupilumab and 19% with biweekly dupilumab, compared with 8% of controls. “To me, conjunctivitis is the only significant side effect for this drug that we’ve seen thus far,” Dr. Blauvelt commented. The mechanism of the conjunctivitis is unknown. It’s clear, however, that the rate goes up with duration on therapy.
“Most of the cases have been mild to moderate and have not interfered with therapy. I’ve treated probably 80 patients with dupilumab, and I’ve had only 1 who had to stop due to her eyes,” the dermatologist recalled. The condition often responds to wetting eye drops, although topical corticosteroid eye drops are required sometimes, he added.
CAFE was a double-blind, randomized, placebo-controlled trial including 325 European patients with moderate or severe AD, all of whom were on a mid-potency topical corticosteroid with or without a topical calcineurin inhibitor. Two-thirds had previously been on cyclosporine with poor results; the drug was contraindicated in the others. Participants were randomized to subcutaneous injections of dupilumab at 300 mg weekly or biweekly or to placebo injections.
The primary outcome – an EASI-75 response at 16 weeks – was achieved in 59% of patients on weekly dupilumab, 63% of those on biweekly dupilumab, and 30% of controls on topical steroids plus placebo injections, according to Dr. De Bruin-Weller of the University Medical Center at Utrecht, the Netherlands.
A clinically meaningful 4-point or greater improvement from baseline in the Dermatology Life Quality Index occurred at 16 weeks in 79% and 88% of patients on dupilumab weekly or biweekly, respectively, compared with 44% of controls.
And among the three-quarters of CAFE participants reporting baseline moderate or severe pain or discomfort from AD, 55% of those on weekly dupilumab had none at all as assessed by the EQ-5D Pain/Discomfort Questionnaire at week 16, as did 64% on dupilumab every 2 weeks and 28% of controls on topical steroids.
At the prestigious joint EADV/AAD session of the European congress, Lawrence F. Eichenfield, MD, hailed dupilumab as evidence that “the systemic therapy revolution in atopic dermatitis is certainly happening,” with a plethora of additional agents targeting various key disease pathways now working their way through the developmental pipeline.
A key remaining question about dupilumab is, what about the safety and efficacy of the drug in the pediatric population, for whom the drug is not currently approved? Separate phase 3 randomized trials addressing this issue are now enrolling 6- to 11-year-olds and 12- to 17-year-olds.
Dr. Eichenfield is optimistic about the outcomes of these ongoing pediatric trials on the basis of an open-label, phase 2a, proof of concept study in 6- to 17-year-olds presented in a late-breaking research session at the 2017 annual meeting of the AAD. The pharmacokinetics were very similar to those seen in adults. Moreover, EASI scores improved by 32%-51% after just a single, weight-based injection of dupilumab and by up to 70% following four consecutive weekly injections.
“This is very, very exciting information. Whether the drug could actually be disease modifying is obviously a big question for us in pediatric dermatology,” observed Dr. Eichenfield, who is chief of pediatric and adolescent dermatology at Rady Children’s Hospital–San Diego and professor of dermatology and pediatrics at the University of California, San Diego.
He highlighted two important safety considerations regarding dupilumab: patients on the biologic should not be given any live vaccines, and the same IL-4 and IL-13 cytokine pathways targeted by dupilumab in patients with AD also are key in other atopic and allergic diseases. Indeed, the biologic showed positive outcomes in a recent pivotal phase 3 clinical trial for patients with uncontrolled persistent asthma, and an application for an expanded indication for dupilumab in such patients is expected to be filed with the FDA later in 2017. Dupilumab is also in phase 3 for patients with nasal polyps and in phase 2 studies for eosinophilic esophagitis.
“One of the interesting things about dupilumab is that we in dermatology are going to be in a situation where we’re able to impact diseases other than the single disease that the medication has been approved for,” Dr. Eichenfield said.
However, until such time as dupilumab actually receives an expanded indication for asthma and the details of how best to use the biologic in AD patients with that comorbidity have been worked out, it’s important for dermatologists prescribing dupilumab in those dual-diagnosis patients to discuss with them the necessity of staying on their asthma medications even though their skin is much improved, they’re feeling good, and their asthma seems to be doing better. During the dupilumab clinical trials program for AD, a fatal asthma exacerbation occurred in a patient with comorbid asthma who stopped taking asthma medications, he noted.
The CHRONOS and CAFE trials were funded by Sanofi and Regeneron Pharmaceuticals. Dr. Blauvelt, Dr. De Bruin-Weller, and Dr. Eichenfield reported serving as consultants to and researchers for those pharmaceutical companies and numerous others.
EXPERT ANALYSIS FROM THE EADV CONGRESS








