User login
Is MS therapy getting to be too much for general neurologists?
ORLANDO – The burgeoning treatment options for multiple sclerosis are increasingly pushing the disease beyond the comfort zone of general neurologists, according to Dr. Mariko Kita.
"MS has become a very, very active area. We’re diagnosing this condition now earlier than ever before. We are initiating treatments at the earliest sign of this condition. We’re going to be seeing, if we haven’t already, a greater influx of patients into MS centers to try to tease apart which disease-modifying therapy might be best for them. And it’s possible that the general neurologists who aren’t referring patients to us at MS centers are going to limit their discussion. They’re going to stick to their go-to drugs: They’ll say, ‘I’ve got one go-to injectable and one go-to oral, and those are the things I’m going to talk about,’ " predicted Dr. Kita, director of the Virginia Mason MS Center, Seattle, and director of the MS clinical trials unit at Virginia Mason’s Benaroya Research Institute.
There are currently nine Food and Drug Administration–approved disease-modifying therapies for MS, including three recently approved oral agents. Plenty of additional medications are advancing through the developmental pipeline.
"All of these new treatments have definitely had an impact on our clinical practice. They’ve increased our burden in a number of ways," Dr. Kita said at the fifth Cooperative Meeting of the Consortium of Multiple Sclerosis Centers and the Americas Committee for Treatment and Research in Multiple Sclerosis.
For one, patient education regarding treatment options has become quite time consuming.
"The discussion of the educational component of the injectables has already been fairly complicated, and now we’re adding to it agents that have a very wide spectrum of mechanisms of action," the neurologist said. "So the question is, how much detail do you go into? What information do you share? And if you’re not in an MS center, do you have help from a nurse educator or others to do this education?"
And then there is the numbing challenge of securing insurance authorization for treatment. "This is perhaps the single biggest unreimbursed burden on my clinic and clinic staff. This is really a major deal for MS centers," Dr. Kita said.
Requisite patient monitoring is also increasingly elaborate. This may include testing prior to putting a patient on an agent, first-dose monitoring, and ongoing monitoring for treatment adherence and the possible emergence of serious side effects.
Take, for example, fingolimod (Gilenya), approved in September 2010 as the first disease-modifying oral agent for MS, and thus the oral drug with which physicians have the most experience.
"The start-up, let’s face it, is labor intensive," Dr. Kita said.
Her personal practice before placing a patient on fingolimod is to order an electrocardiogram, pulmonary function tests, a retinal optical coherence tomography (OCT) scan, a dermatologic evaluation, and a check of varicella titers, with vaccination if appropriate. Then comes the supervised, 6-hour, first-dose observation period.
"We do that in the clinic. We have the patient bring in a companion. They take up an exam room and are not under continuous surveillance, so if issues should arise, we want a companion on hand to call for help. Plus, a medical assistant goes in every hour. And the prescribing physician checks in before the first dose and at the end of the day," she explained.
If patients are taking a medication that prolongs the QT interval, however, as many do in order to control their chronic pain, they should be hospitalized for the first dose of fingolimod, Dr. Kita added.
Monitoring needs to be done on an ongoing basis, although as yet there are no clear guidelines as to the specifics. She recommended an annual brain MRI, an ECG, pulmonary function tests, a dermatologic evaluation, and an OCT, because cases of macular edema have occurred 2 years into treatment.
"That’s not necessarily everyone’s standard practice, but it’s what we do," she continued.
And then there is the issue of monitoring circulating CD4 cell counts. Fingolimod’s mechanism of action involves sequestration of lymphocytes in lymph nodes so they can’t contribute to autoimmunity. As a result, patients maintained on fingolimod experience abnormally low levels of circulating CD4 cells, the clinical significance of which remains unclear.
"I think the immunocompromise issue hasn’t really been resolved. I know of practitioners who say, ‘I don’t want to check CD4 counts because it makes me nervous,’ and I’m not sure that’s the best practice. Having said that, it’s hard to know what to do with those exceedingly low CD4 counts," Dr. Kita said.
She added that she views the development of shingles in a patient on fingolimod as a potential red flag warranting serious consideration of a switch to another agent.
Dr. Kita reported receiving research support from Biogen Idec, Novartis (which markets Gilenya), Serono, and Acorda, and personal compensation from Biogen Idec, Bayer, and Genzyme.
ORLANDO – The burgeoning treatment options for multiple sclerosis are increasingly pushing the disease beyond the comfort zone of general neurologists, according to Dr. Mariko Kita.
"MS has become a very, very active area. We’re diagnosing this condition now earlier than ever before. We are initiating treatments at the earliest sign of this condition. We’re going to be seeing, if we haven’t already, a greater influx of patients into MS centers to try to tease apart which disease-modifying therapy might be best for them. And it’s possible that the general neurologists who aren’t referring patients to us at MS centers are going to limit their discussion. They’re going to stick to their go-to drugs: They’ll say, ‘I’ve got one go-to injectable and one go-to oral, and those are the things I’m going to talk about,’ " predicted Dr. Kita, director of the Virginia Mason MS Center, Seattle, and director of the MS clinical trials unit at Virginia Mason’s Benaroya Research Institute.
There are currently nine Food and Drug Administration–approved disease-modifying therapies for MS, including three recently approved oral agents. Plenty of additional medications are advancing through the developmental pipeline.
"All of these new treatments have definitely had an impact on our clinical practice. They’ve increased our burden in a number of ways," Dr. Kita said at the fifth Cooperative Meeting of the Consortium of Multiple Sclerosis Centers and the Americas Committee for Treatment and Research in Multiple Sclerosis.
For one, patient education regarding treatment options has become quite time consuming.
"The discussion of the educational component of the injectables has already been fairly complicated, and now we’re adding to it agents that have a very wide spectrum of mechanisms of action," the neurologist said. "So the question is, how much detail do you go into? What information do you share? And if you’re not in an MS center, do you have help from a nurse educator or others to do this education?"
And then there is the numbing challenge of securing insurance authorization for treatment. "This is perhaps the single biggest unreimbursed burden on my clinic and clinic staff. This is really a major deal for MS centers," Dr. Kita said.
Requisite patient monitoring is also increasingly elaborate. This may include testing prior to putting a patient on an agent, first-dose monitoring, and ongoing monitoring for treatment adherence and the possible emergence of serious side effects.
Take, for example, fingolimod (Gilenya), approved in September 2010 as the first disease-modifying oral agent for MS, and thus the oral drug with which physicians have the most experience.
"The start-up, let’s face it, is labor intensive," Dr. Kita said.
Her personal practice before placing a patient on fingolimod is to order an electrocardiogram, pulmonary function tests, a retinal optical coherence tomography (OCT) scan, a dermatologic evaluation, and a check of varicella titers, with vaccination if appropriate. Then comes the supervised, 6-hour, first-dose observation period.
"We do that in the clinic. We have the patient bring in a companion. They take up an exam room and are not under continuous surveillance, so if issues should arise, we want a companion on hand to call for help. Plus, a medical assistant goes in every hour. And the prescribing physician checks in before the first dose and at the end of the day," she explained.
If patients are taking a medication that prolongs the QT interval, however, as many do in order to control their chronic pain, they should be hospitalized for the first dose of fingolimod, Dr. Kita added.
Monitoring needs to be done on an ongoing basis, although as yet there are no clear guidelines as to the specifics. She recommended an annual brain MRI, an ECG, pulmonary function tests, a dermatologic evaluation, and an OCT, because cases of macular edema have occurred 2 years into treatment.
"That’s not necessarily everyone’s standard practice, but it’s what we do," she continued.
And then there is the issue of monitoring circulating CD4 cell counts. Fingolimod’s mechanism of action involves sequestration of lymphocytes in lymph nodes so they can’t contribute to autoimmunity. As a result, patients maintained on fingolimod experience abnormally low levels of circulating CD4 cells, the clinical significance of which remains unclear.
"I think the immunocompromise issue hasn’t really been resolved. I know of practitioners who say, ‘I don’t want to check CD4 counts because it makes me nervous,’ and I’m not sure that’s the best practice. Having said that, it’s hard to know what to do with those exceedingly low CD4 counts," Dr. Kita said.
She added that she views the development of shingles in a patient on fingolimod as a potential red flag warranting serious consideration of a switch to another agent.
Dr. Kita reported receiving research support from Biogen Idec, Novartis (which markets Gilenya), Serono, and Acorda, and personal compensation from Biogen Idec, Bayer, and Genzyme.
ORLANDO – The burgeoning treatment options for multiple sclerosis are increasingly pushing the disease beyond the comfort zone of general neurologists, according to Dr. Mariko Kita.
"MS has become a very, very active area. We’re diagnosing this condition now earlier than ever before. We are initiating treatments at the earliest sign of this condition. We’re going to be seeing, if we haven’t already, a greater influx of patients into MS centers to try to tease apart which disease-modifying therapy might be best for them. And it’s possible that the general neurologists who aren’t referring patients to us at MS centers are going to limit their discussion. They’re going to stick to their go-to drugs: They’ll say, ‘I’ve got one go-to injectable and one go-to oral, and those are the things I’m going to talk about,’ " predicted Dr. Kita, director of the Virginia Mason MS Center, Seattle, and director of the MS clinical trials unit at Virginia Mason’s Benaroya Research Institute.
There are currently nine Food and Drug Administration–approved disease-modifying therapies for MS, including three recently approved oral agents. Plenty of additional medications are advancing through the developmental pipeline.
"All of these new treatments have definitely had an impact on our clinical practice. They’ve increased our burden in a number of ways," Dr. Kita said at the fifth Cooperative Meeting of the Consortium of Multiple Sclerosis Centers and the Americas Committee for Treatment and Research in Multiple Sclerosis.
For one, patient education regarding treatment options has become quite time consuming.
"The discussion of the educational component of the injectables has already been fairly complicated, and now we’re adding to it agents that have a very wide spectrum of mechanisms of action," the neurologist said. "So the question is, how much detail do you go into? What information do you share? And if you’re not in an MS center, do you have help from a nurse educator or others to do this education?"
And then there is the numbing challenge of securing insurance authorization for treatment. "This is perhaps the single biggest unreimbursed burden on my clinic and clinic staff. This is really a major deal for MS centers," Dr. Kita said.
Requisite patient monitoring is also increasingly elaborate. This may include testing prior to putting a patient on an agent, first-dose monitoring, and ongoing monitoring for treatment adherence and the possible emergence of serious side effects.
Take, for example, fingolimod (Gilenya), approved in September 2010 as the first disease-modifying oral agent for MS, and thus the oral drug with which physicians have the most experience.
"The start-up, let’s face it, is labor intensive," Dr. Kita said.
Her personal practice before placing a patient on fingolimod is to order an electrocardiogram, pulmonary function tests, a retinal optical coherence tomography (OCT) scan, a dermatologic evaluation, and a check of varicella titers, with vaccination if appropriate. Then comes the supervised, 6-hour, first-dose observation period.
"We do that in the clinic. We have the patient bring in a companion. They take up an exam room and are not under continuous surveillance, so if issues should arise, we want a companion on hand to call for help. Plus, a medical assistant goes in every hour. And the prescribing physician checks in before the first dose and at the end of the day," she explained.
If patients are taking a medication that prolongs the QT interval, however, as many do in order to control their chronic pain, they should be hospitalized for the first dose of fingolimod, Dr. Kita added.
Monitoring needs to be done on an ongoing basis, although as yet there are no clear guidelines as to the specifics. She recommended an annual brain MRI, an ECG, pulmonary function tests, a dermatologic evaluation, and an OCT, because cases of macular edema have occurred 2 years into treatment.
"That’s not necessarily everyone’s standard practice, but it’s what we do," she continued.
And then there is the issue of monitoring circulating CD4 cell counts. Fingolimod’s mechanism of action involves sequestration of lymphocytes in lymph nodes so they can’t contribute to autoimmunity. As a result, patients maintained on fingolimod experience abnormally low levels of circulating CD4 cells, the clinical significance of which remains unclear.
"I think the immunocompromise issue hasn’t really been resolved. I know of practitioners who say, ‘I don’t want to check CD4 counts because it makes me nervous,’ and I’m not sure that’s the best practice. Having said that, it’s hard to know what to do with those exceedingly low CD4 counts," Dr. Kita said.
She added that she views the development of shingles in a patient on fingolimod as a potential red flag warranting serious consideration of a switch to another agent.
Dr. Kita reported receiving research support from Biogen Idec, Novartis (which markets Gilenya), Serono, and Acorda, and personal compensation from Biogen Idec, Bayer, and Genzyme.
EXPERT ANALYSIS FROM THE CMSC/ACTRIMS ANNUAL MEETING
Overtreatment common in high-risk diabetes patients
Earn 0.25 hours AMA PRA Category 1 credit: Read this article, and click the link at the end to take the post-test.
CHICAGO – Glycemic overtreatment of high-risk diabetic patients is rampant within the Veterans Affairs health care system, according to a national study.
Moreover, because many of these diabetic veterans who are at high risk for serious hypoglycemia are also Medicare eligible, it’s quite likely that overtreatment is a common problem in the Medicare population as well.
"I think these findings are directly relevant to Medicare," Dr. Leonard M. Pogach said in presenting the VA study results at the annual scientific sessions of the American Diabetes Association.
Current performance measures do not assess potential overtreatment of high-risk diabetic patients in either the VA or Medicare populations. But as a result of the VA study findings, joint federal efforts are underway to address this shortcoming, according to Dr. Pogach, who is national program director for endocrinology and diabetes at the Veterans Health Administration and professor of medicine at the New Jersey Medical School, Newark.
He cited as a major impetus for the VA study an eye-opening 2011 report by investigators at the Centers for Disease Control and Prevention that identified insulin and sulfonylurea drugs as the combined number-two cause of emergency hospitalizations for adverse drug events in the United States, second only to warfarin (N. Engl. J. Med. 2011;365:2002-12).
As part of the Choosing Wisely campaign, the American Geriatrics Society recommends that medications other than metformin not be routinely used to lower hemoglobin A1c below 7.5% in patients older than 65 years. The group further recommends an HbA1c target of 8%-9% for those with serious comorbid conditions.
Similarly, the ADA now recommends an HbA1c target of 7.5%-8.0% or slightly more in patients at increased risk for serious hypoglycemia or with reduced life expectancy, rather than its former universal goal of less than 7.0%.
For purposes of the VA study, Dr. Pogach and coinvestigators defined "high-risk" diabetes patients as those receiving insulin and/or sulfonylurea therapy and who are at least 70 years of age, have renal impairment as reflected in a serum creatinine level greater than 1.7 mg/dL, or have been diagnosed with cognitive impairment or dementia.
During the study year of 2009, a total of 285,476 of 652,738 VA patients, or 44%,with diabetes on insulin and/or sulfonylurea therapy qualified as high risk, based upon that definition. They received treatment in 139 VA facilities in 21 regions nationally.
Overall, 48% of these high-risk diabetes patients were likely being overtreated, as reflected in an HbA1c below 7.0%. Moreover, one-quarter of the high-risk group had an HbA1c below 6.5%, and 10% had an HbA1c of less than 6.0%, Dr. Pogach reported.
He added that these results likely underestimate the true extent of the glycemic overtreatment problem, because he and his coinvestigators defined "high risk" quite conservatively – based simply upon advanced age, renal dysfunction, and cognitive impairment.
Had they included other reasonable criteria – specifically, diminished life expectancy; stroke and other major neurologic disorders; cardiovascular disease; major depression; alcohol and/or drug abuse; and advanced diabetic complications – the proportion of the 652,738 diabetic VA patients on insulin and/or sulfonylurea therapy who would have qualified as being at high risk for serious hypoglycemia would have climbed from 44% all the way up to 71%.
The variation in overtreatment rates from region to region within the VA system was fairly tight. The range was much greater among facilities within a given regional district, where overtreatment rates varied from a low of 37% at a select few facilities to as high as 63%.
"We were able to identify several facilities with statistically remarkably lower rates of overtreatment than in the larger regions they lie in," Dr. Pogach noted. "Those are the sites where we might want to do site visits and qualitative studies to try to figure out what made them different. What is it about their culture or their patients? What happened in those places that we might subsequently want to replicate?"
Of note, the VA never adopted the one-size-fits-all goal of an HbA1c below 7.0% that the ADA recommended until recently. For more than a decade, VA clinical practice guidelines have included stratified glycemic targets based on comorbidities and life expectancy.
One audience member took issue with Dr. Pogach’s use of the word "overtreatment."
"Overtreatment is really a somewhat pejorative term," he argued. "I think you really have to have data showing that we are harming these people, not in terms of the surrogate outcome of hypoglycemia, but in real negative consequences."
Dr. Pogach was quick to rebut.
"I think the published results of the ACCORD and ADVANCE studies demonstrate that there’s a very strong association between cardiovascular morbidity and mortality and self-reported hypoglycemia, with adjusted odds ratios of about two and one-half," he explained. "I don’t think those data will ever be beat. We’re never going to have a randomized trial. So, I think the association with adverse outcomes is very clear, although we don’t know if hypoglycemia is the proximate cause or not."
The Department of Veterans Affairs supported the study. Dr. Pogach reported having no financial conflicts of interest.
To earn 0.25 hours AMA PRA Category 1 credit after reading this article, take the post-test here.
Earn 0.25 hours AMA PRA Category 1 credit: Read this article, and click the link at the end to take the post-test.
CHICAGO – Glycemic overtreatment of high-risk diabetic patients is rampant within the Veterans Affairs health care system, according to a national study.
Moreover, because many of these diabetic veterans who are at high risk for serious hypoglycemia are also Medicare eligible, it’s quite likely that overtreatment is a common problem in the Medicare population as well.
"I think these findings are directly relevant to Medicare," Dr. Leonard M. Pogach said in presenting the VA study results at the annual scientific sessions of the American Diabetes Association.
Current performance measures do not assess potential overtreatment of high-risk diabetic patients in either the VA or Medicare populations. But as a result of the VA study findings, joint federal efforts are underway to address this shortcoming, according to Dr. Pogach, who is national program director for endocrinology and diabetes at the Veterans Health Administration and professor of medicine at the New Jersey Medical School, Newark.
He cited as a major impetus for the VA study an eye-opening 2011 report by investigators at the Centers for Disease Control and Prevention that identified insulin and sulfonylurea drugs as the combined number-two cause of emergency hospitalizations for adverse drug events in the United States, second only to warfarin (N. Engl. J. Med. 2011;365:2002-12).
As part of the Choosing Wisely campaign, the American Geriatrics Society recommends that medications other than metformin not be routinely used to lower hemoglobin A1c below 7.5% in patients older than 65 years. The group further recommends an HbA1c target of 8%-9% for those with serious comorbid conditions.
Similarly, the ADA now recommends an HbA1c target of 7.5%-8.0% or slightly more in patients at increased risk for serious hypoglycemia or with reduced life expectancy, rather than its former universal goal of less than 7.0%.
For purposes of the VA study, Dr. Pogach and coinvestigators defined "high-risk" diabetes patients as those receiving insulin and/or sulfonylurea therapy and who are at least 70 years of age, have renal impairment as reflected in a serum creatinine level greater than 1.7 mg/dL, or have been diagnosed with cognitive impairment or dementia.
During the study year of 2009, a total of 285,476 of 652,738 VA patients, or 44%,with diabetes on insulin and/or sulfonylurea therapy qualified as high risk, based upon that definition. They received treatment in 139 VA facilities in 21 regions nationally.
Overall, 48% of these high-risk diabetes patients were likely being overtreated, as reflected in an HbA1c below 7.0%. Moreover, one-quarter of the high-risk group had an HbA1c below 6.5%, and 10% had an HbA1c of less than 6.0%, Dr. Pogach reported.
He added that these results likely underestimate the true extent of the glycemic overtreatment problem, because he and his coinvestigators defined "high risk" quite conservatively – based simply upon advanced age, renal dysfunction, and cognitive impairment.
Had they included other reasonable criteria – specifically, diminished life expectancy; stroke and other major neurologic disorders; cardiovascular disease; major depression; alcohol and/or drug abuse; and advanced diabetic complications – the proportion of the 652,738 diabetic VA patients on insulin and/or sulfonylurea therapy who would have qualified as being at high risk for serious hypoglycemia would have climbed from 44% all the way up to 71%.
The variation in overtreatment rates from region to region within the VA system was fairly tight. The range was much greater among facilities within a given regional district, where overtreatment rates varied from a low of 37% at a select few facilities to as high as 63%.
"We were able to identify several facilities with statistically remarkably lower rates of overtreatment than in the larger regions they lie in," Dr. Pogach noted. "Those are the sites where we might want to do site visits and qualitative studies to try to figure out what made them different. What is it about their culture or their patients? What happened in those places that we might subsequently want to replicate?"
Of note, the VA never adopted the one-size-fits-all goal of an HbA1c below 7.0% that the ADA recommended until recently. For more than a decade, VA clinical practice guidelines have included stratified glycemic targets based on comorbidities and life expectancy.
One audience member took issue with Dr. Pogach’s use of the word "overtreatment."
"Overtreatment is really a somewhat pejorative term," he argued. "I think you really have to have data showing that we are harming these people, not in terms of the surrogate outcome of hypoglycemia, but in real negative consequences."
Dr. Pogach was quick to rebut.
"I think the published results of the ACCORD and ADVANCE studies demonstrate that there’s a very strong association between cardiovascular morbidity and mortality and self-reported hypoglycemia, with adjusted odds ratios of about two and one-half," he explained. "I don’t think those data will ever be beat. We’re never going to have a randomized trial. So, I think the association with adverse outcomes is very clear, although we don’t know if hypoglycemia is the proximate cause or not."
The Department of Veterans Affairs supported the study. Dr. Pogach reported having no financial conflicts of interest.
To earn 0.25 hours AMA PRA Category 1 credit after reading this article, take the post-test here.
Earn 0.25 hours AMA PRA Category 1 credit: Read this article, and click the link at the end to take the post-test.
CHICAGO – Glycemic overtreatment of high-risk diabetic patients is rampant within the Veterans Affairs health care system, according to a national study.
Moreover, because many of these diabetic veterans who are at high risk for serious hypoglycemia are also Medicare eligible, it’s quite likely that overtreatment is a common problem in the Medicare population as well.
"I think these findings are directly relevant to Medicare," Dr. Leonard M. Pogach said in presenting the VA study results at the annual scientific sessions of the American Diabetes Association.
Current performance measures do not assess potential overtreatment of high-risk diabetic patients in either the VA or Medicare populations. But as a result of the VA study findings, joint federal efforts are underway to address this shortcoming, according to Dr. Pogach, who is national program director for endocrinology and diabetes at the Veterans Health Administration and professor of medicine at the New Jersey Medical School, Newark.
He cited as a major impetus for the VA study an eye-opening 2011 report by investigators at the Centers for Disease Control and Prevention that identified insulin and sulfonylurea drugs as the combined number-two cause of emergency hospitalizations for adverse drug events in the United States, second only to warfarin (N. Engl. J. Med. 2011;365:2002-12).
As part of the Choosing Wisely campaign, the American Geriatrics Society recommends that medications other than metformin not be routinely used to lower hemoglobin A1c below 7.5% in patients older than 65 years. The group further recommends an HbA1c target of 8%-9% for those with serious comorbid conditions.
Similarly, the ADA now recommends an HbA1c target of 7.5%-8.0% or slightly more in patients at increased risk for serious hypoglycemia or with reduced life expectancy, rather than its former universal goal of less than 7.0%.
For purposes of the VA study, Dr. Pogach and coinvestigators defined "high-risk" diabetes patients as those receiving insulin and/or sulfonylurea therapy and who are at least 70 years of age, have renal impairment as reflected in a serum creatinine level greater than 1.7 mg/dL, or have been diagnosed with cognitive impairment or dementia.
During the study year of 2009, a total of 285,476 of 652,738 VA patients, or 44%,with diabetes on insulin and/or sulfonylurea therapy qualified as high risk, based upon that definition. They received treatment in 139 VA facilities in 21 regions nationally.
Overall, 48% of these high-risk diabetes patients were likely being overtreated, as reflected in an HbA1c below 7.0%. Moreover, one-quarter of the high-risk group had an HbA1c below 6.5%, and 10% had an HbA1c of less than 6.0%, Dr. Pogach reported.
He added that these results likely underestimate the true extent of the glycemic overtreatment problem, because he and his coinvestigators defined "high risk" quite conservatively – based simply upon advanced age, renal dysfunction, and cognitive impairment.
Had they included other reasonable criteria – specifically, diminished life expectancy; stroke and other major neurologic disorders; cardiovascular disease; major depression; alcohol and/or drug abuse; and advanced diabetic complications – the proportion of the 652,738 diabetic VA patients on insulin and/or sulfonylurea therapy who would have qualified as being at high risk for serious hypoglycemia would have climbed from 44% all the way up to 71%.
The variation in overtreatment rates from region to region within the VA system was fairly tight. The range was much greater among facilities within a given regional district, where overtreatment rates varied from a low of 37% at a select few facilities to as high as 63%.
"We were able to identify several facilities with statistically remarkably lower rates of overtreatment than in the larger regions they lie in," Dr. Pogach noted. "Those are the sites where we might want to do site visits and qualitative studies to try to figure out what made them different. What is it about their culture or their patients? What happened in those places that we might subsequently want to replicate?"
Of note, the VA never adopted the one-size-fits-all goal of an HbA1c below 7.0% that the ADA recommended until recently. For more than a decade, VA clinical practice guidelines have included stratified glycemic targets based on comorbidities and life expectancy.
One audience member took issue with Dr. Pogach’s use of the word "overtreatment."
"Overtreatment is really a somewhat pejorative term," he argued. "I think you really have to have data showing that we are harming these people, not in terms of the surrogate outcome of hypoglycemia, but in real negative consequences."
Dr. Pogach was quick to rebut.
"I think the published results of the ACCORD and ADVANCE studies demonstrate that there’s a very strong association between cardiovascular morbidity and mortality and self-reported hypoglycemia, with adjusted odds ratios of about two and one-half," he explained. "I don’t think those data will ever be beat. We’re never going to have a randomized trial. So, I think the association with adverse outcomes is very clear, although we don’t know if hypoglycemia is the proximate cause or not."
The Department of Veterans Affairs supported the study. Dr. Pogach reported having no financial conflicts of interest.
To earn 0.25 hours AMA PRA Category 1 credit after reading this article, take the post-test here.
AT THE ADA ANNUAL SCIENTIFIC SESSIONS
Overtreatment common in high-risk diabetes patients
CHICAGO – Glycemic overtreatment of high-risk diabetic patients is rampant within the Veterans Affairs health care system, according to a national study.
Moreover, because many of these diabetic veterans who are at high risk for serious hypoglycemia are also Medicare eligible, it’s quite likely that overtreatment is a common problem in the Medicare population as well.
"I think these findings are directly relevant to Medicare," Dr. Leonard M. Pogach said in presenting the VA study results at the annual scientific sessions of the American Diabetes Association.
Current performance measures do not assess potential overtreatment of high-risk diabetic patients in either the VA or Medicare populations. But as a result of the VA study findings, joint federal efforts are underway to address this shortcoming, according to Dr. Pogach, who is national program director for endocrinology and diabetes at the Veterans Health Administration and professor of medicine at the New Jersey Medical School, Newark.
He cited as a major impetus for the VA study an eye-opening 2011 report by investigators at the Centers for Disease Control and Prevention that identified insulin and sulfonylurea drugs as the combined number-two cause of emergency hospitalizations for adverse drug events in the United States, second only to warfarin (N. Engl. J. Med. 2011;365:2002-12).
As part of the Choosing Wisely campaign, the American Geriatrics Society recommends that medications other than metformin not be routinely used to lower hemoglobin A1c below 7.5% in patients older than 65 years. The group further recommends an HbA1c target of 8%-9% for those with serious comorbid conditions.
Similarly, the ADA now recommends an HbA1c target of 7.5%-8.0% or slightly more in patients at increased risk for serious hypoglycemia or with reduced life expectancy, rather than its former universal goal of less than 7.0%.
For purposes of the VA study, Dr. Pogach and coinvestigators defined "high-risk" diabetes patients as those receiving insulin and/or sulfonylurea therapy and who are at least 70 years of age, have renal impairment as reflected in a serum creatinine level greater than 1.7 mg/dL, or have been diagnosed with cognitive impairment or dementia.
During the study year of 2009, a total of 285,476 of 652,738 VA patients, or 44%,with diabetes on insulin and/or sulfonylurea therapy qualified as high risk, based upon that definition. They received treatment in 139 VA facilities in 21 regions nationally.
Overall, 48% of these high-risk diabetes patients were likely being overtreated, as reflected in an HbA1c below 7.0%. Moreover, one-quarter of the high-risk group had an HbA1c below 6.5%, and 10% had an HbA1c of less than 6.0%, Dr. Pogach reported.
He added that these results likely underestimate the true extent of the glycemic overtreatment problem, because he and his coinvestigators defined "high risk" quite conservatively – based simply upon advanced age, renal dysfunction, and cognitive impairment.
Had they included other reasonable criteria – specifically, diminished life expectancy; stroke and other major neurologic disorders; cardiovascular disease; major depression; alcohol and/or drug abuse; and advanced diabetic complications – the proportion of the 652,738 diabetic VA patients on insulin and/or sulfonylurea therapy who would have qualified as being at high risk for serious hypoglycemia would have climbed from 44% all the way up to 71%.
The variation in overtreatment rates from region to region within the VA system was fairly tight. The range was much greater among facilities within a given regional district, where overtreatment rates varied from a low of 37% at a select few facilities to as high as 63%.
"We were able to identify several facilities with statistically remarkably lower rates of overtreatment than in the larger regions they lie in," Dr. Pogach noted. "Those are the sites where we might want to do site visits and qualitative studies to try to figure out what made them different. What is it about their culture or their patients? What happened in those places that we might subsequently want to replicate?"
Of note, the VA never adopted the one-size-fits-all goal of an HbA1c below 7.0% that the ADA recommended until recently. For more than a decade, VA clinical practice guidelines have included stratified glycemic targets based on comorbidities and life expectancy.
One audience member took issue with Dr. Pogach’s use of the word "overtreatment."
"Overtreatment is really a somewhat pejorative term," he argued. "I think you really have to have data showing that we are harming these people, not in terms of the surrogate outcome of hypoglycemia, but in real negative consequences."
Dr. Pogach was quick to rebut.
"I think the published results of the ACCORD and ADVANCE studies demonstrate that there’s a very strong association between cardiovascular morbidity and mortality and self-reported hypoglycemia, with adjusted odds ratios of about two and one-half," he explained. "I don’t think those data will ever be beat. We’re never going to have a randomized trial. So, I think the association with adverse outcomes is very clear, although we don’t know if hypoglycemia is the proximate cause or not."
The Department of Veterans Affairs supported the study. Dr. Pogach reported having no financial conflicts of interest.
CHICAGO – Glycemic overtreatment of high-risk diabetic patients is rampant within the Veterans Affairs health care system, according to a national study.
Moreover, because many of these diabetic veterans who are at high risk for serious hypoglycemia are also Medicare eligible, it’s quite likely that overtreatment is a common problem in the Medicare population as well.
"I think these findings are directly relevant to Medicare," Dr. Leonard M. Pogach said in presenting the VA study results at the annual scientific sessions of the American Diabetes Association.
Current performance measures do not assess potential overtreatment of high-risk diabetic patients in either the VA or Medicare populations. But as a result of the VA study findings, joint federal efforts are underway to address this shortcoming, according to Dr. Pogach, who is national program director for endocrinology and diabetes at the Veterans Health Administration and professor of medicine at the New Jersey Medical School, Newark.
He cited as a major impetus for the VA study an eye-opening 2011 report by investigators at the Centers for Disease Control and Prevention that identified insulin and sulfonylurea drugs as the combined number-two cause of emergency hospitalizations for adverse drug events in the United States, second only to warfarin (N. Engl. J. Med. 2011;365:2002-12).
As part of the Choosing Wisely campaign, the American Geriatrics Society recommends that medications other than metformin not be routinely used to lower hemoglobin A1c below 7.5% in patients older than 65 years. The group further recommends an HbA1c target of 8%-9% for those with serious comorbid conditions.
Similarly, the ADA now recommends an HbA1c target of 7.5%-8.0% or slightly more in patients at increased risk for serious hypoglycemia or with reduced life expectancy, rather than its former universal goal of less than 7.0%.
For purposes of the VA study, Dr. Pogach and coinvestigators defined "high-risk" diabetes patients as those receiving insulin and/or sulfonylurea therapy and who are at least 70 years of age, have renal impairment as reflected in a serum creatinine level greater than 1.7 mg/dL, or have been diagnosed with cognitive impairment or dementia.
During the study year of 2009, a total of 285,476 of 652,738 VA patients, or 44%,with diabetes on insulin and/or sulfonylurea therapy qualified as high risk, based upon that definition. They received treatment in 139 VA facilities in 21 regions nationally.
Overall, 48% of these high-risk diabetes patients were likely being overtreated, as reflected in an HbA1c below 7.0%. Moreover, one-quarter of the high-risk group had an HbA1c below 6.5%, and 10% had an HbA1c of less than 6.0%, Dr. Pogach reported.
He added that these results likely underestimate the true extent of the glycemic overtreatment problem, because he and his coinvestigators defined "high risk" quite conservatively – based simply upon advanced age, renal dysfunction, and cognitive impairment.
Had they included other reasonable criteria – specifically, diminished life expectancy; stroke and other major neurologic disorders; cardiovascular disease; major depression; alcohol and/or drug abuse; and advanced diabetic complications – the proportion of the 652,738 diabetic VA patients on insulin and/or sulfonylurea therapy who would have qualified as being at high risk for serious hypoglycemia would have climbed from 44% all the way up to 71%.
The variation in overtreatment rates from region to region within the VA system was fairly tight. The range was much greater among facilities within a given regional district, where overtreatment rates varied from a low of 37% at a select few facilities to as high as 63%.
"We were able to identify several facilities with statistically remarkably lower rates of overtreatment than in the larger regions they lie in," Dr. Pogach noted. "Those are the sites where we might want to do site visits and qualitative studies to try to figure out what made them different. What is it about their culture or their patients? What happened in those places that we might subsequently want to replicate?"
Of note, the VA never adopted the one-size-fits-all goal of an HbA1c below 7.0% that the ADA recommended until recently. For more than a decade, VA clinical practice guidelines have included stratified glycemic targets based on comorbidities and life expectancy.
One audience member took issue with Dr. Pogach’s use of the word "overtreatment."
"Overtreatment is really a somewhat pejorative term," he argued. "I think you really have to have data showing that we are harming these people, not in terms of the surrogate outcome of hypoglycemia, but in real negative consequences."
Dr. Pogach was quick to rebut.
"I think the published results of the ACCORD and ADVANCE studies demonstrate that there’s a very strong association between cardiovascular morbidity and mortality and self-reported hypoglycemia, with adjusted odds ratios of about two and one-half," he explained. "I don’t think those data will ever be beat. We’re never going to have a randomized trial. So, I think the association with adverse outcomes is very clear, although we don’t know if hypoglycemia is the proximate cause or not."
The Department of Veterans Affairs supported the study. Dr. Pogach reported having no financial conflicts of interest.
CHICAGO – Glycemic overtreatment of high-risk diabetic patients is rampant within the Veterans Affairs health care system, according to a national study.
Moreover, because many of these diabetic veterans who are at high risk for serious hypoglycemia are also Medicare eligible, it’s quite likely that overtreatment is a common problem in the Medicare population as well.
"I think these findings are directly relevant to Medicare," Dr. Leonard M. Pogach said in presenting the VA study results at the annual scientific sessions of the American Diabetes Association.
Current performance measures do not assess potential overtreatment of high-risk diabetic patients in either the VA or Medicare populations. But as a result of the VA study findings, joint federal efforts are underway to address this shortcoming, according to Dr. Pogach, who is national program director for endocrinology and diabetes at the Veterans Health Administration and professor of medicine at the New Jersey Medical School, Newark.
He cited as a major impetus for the VA study an eye-opening 2011 report by investigators at the Centers for Disease Control and Prevention that identified insulin and sulfonylurea drugs as the combined number-two cause of emergency hospitalizations for adverse drug events in the United States, second only to warfarin (N. Engl. J. Med. 2011;365:2002-12).
As part of the Choosing Wisely campaign, the American Geriatrics Society recommends that medications other than metformin not be routinely used to lower hemoglobin A1c below 7.5% in patients older than 65 years. The group further recommends an HbA1c target of 8%-9% for those with serious comorbid conditions.
Similarly, the ADA now recommends an HbA1c target of 7.5%-8.0% or slightly more in patients at increased risk for serious hypoglycemia or with reduced life expectancy, rather than its former universal goal of less than 7.0%.
For purposes of the VA study, Dr. Pogach and coinvestigators defined "high-risk" diabetes patients as those receiving insulin and/or sulfonylurea therapy and who are at least 70 years of age, have renal impairment as reflected in a serum creatinine level greater than 1.7 mg/dL, or have been diagnosed with cognitive impairment or dementia.
During the study year of 2009, a total of 285,476 of 652,738 VA patients, or 44%,with diabetes on insulin and/or sulfonylurea therapy qualified as high risk, based upon that definition. They received treatment in 139 VA facilities in 21 regions nationally.
Overall, 48% of these high-risk diabetes patients were likely being overtreated, as reflected in an HbA1c below 7.0%. Moreover, one-quarter of the high-risk group had an HbA1c below 6.5%, and 10% had an HbA1c of less than 6.0%, Dr. Pogach reported.
He added that these results likely underestimate the true extent of the glycemic overtreatment problem, because he and his coinvestigators defined "high risk" quite conservatively – based simply upon advanced age, renal dysfunction, and cognitive impairment.
Had they included other reasonable criteria – specifically, diminished life expectancy; stroke and other major neurologic disorders; cardiovascular disease; major depression; alcohol and/or drug abuse; and advanced diabetic complications – the proportion of the 652,738 diabetic VA patients on insulin and/or sulfonylurea therapy who would have qualified as being at high risk for serious hypoglycemia would have climbed from 44% all the way up to 71%.
The variation in overtreatment rates from region to region within the VA system was fairly tight. The range was much greater among facilities within a given regional district, where overtreatment rates varied from a low of 37% at a select few facilities to as high as 63%.
"We were able to identify several facilities with statistically remarkably lower rates of overtreatment than in the larger regions they lie in," Dr. Pogach noted. "Those are the sites where we might want to do site visits and qualitative studies to try to figure out what made them different. What is it about their culture or their patients? What happened in those places that we might subsequently want to replicate?"
Of note, the VA never adopted the one-size-fits-all goal of an HbA1c below 7.0% that the ADA recommended until recently. For more than a decade, VA clinical practice guidelines have included stratified glycemic targets based on comorbidities and life expectancy.
One audience member took issue with Dr. Pogach’s use of the word "overtreatment."
"Overtreatment is really a somewhat pejorative term," he argued. "I think you really have to have data showing that we are harming these people, not in terms of the surrogate outcome of hypoglycemia, but in real negative consequences."
Dr. Pogach was quick to rebut.
"I think the published results of the ACCORD and ADVANCE studies demonstrate that there’s a very strong association between cardiovascular morbidity and mortality and self-reported hypoglycemia, with adjusted odds ratios of about two and one-half," he explained. "I don’t think those data will ever be beat. We’re never going to have a randomized trial. So, I think the association with adverse outcomes is very clear, although we don’t know if hypoglycemia is the proximate cause or not."
The Department of Veterans Affairs supported the study. Dr. Pogach reported having no financial conflicts of interest.
AT THE ADA ANNUAL SCIENTIFIC SESSIONS
Major finding: Nearly half of diabetes patients at high risk for serious hypoglycemia, based upon advanced age, renal dysfunction, or cognitive impairment, had an HbA1c below 7.0%, with potential adverse consequences.
Data source: This was a cross-sectional study of nearly 300,000 diabetes patients in the Veterans Affairs health care system deemed at high risk for serious hypoglycemia.
Disclosures: The U.S. Department of Veterans Affairs supported the study. Dr. Pogach had no financial conflicts.
Medical marijuana: Tips from an expert
ORLANDO – An essential element in prescribing medical marijuana responsibly is to insist that the patient must demonstrate improved functional status to be allowed to continue with the therapy, one expert has advised.
"Functional status is really the major issue. If cannabis is going to be used as a medicine, we have to see improvement in function: return to work, improvement in daily activities, engagement in society. I tell patients, ‘If this drug is really helping you, then show me. Show me that you can come off this other medication or reduce the dose. Show me you can go out and do volunteer work or join a club. Prove to me that this is valuable to you, because I cannot continue to authorize access to a substance if you cannot show me that it’s actually helping you improve your quality of life.’ That’s the gauntlet I throw down to patients," Dr. Mark A. Ware said at the fifth Cooperative Meeting sponsored by the Consortium of Multiple Sclerosis Centers and the Americas Committee for Treatment and Research in Multiple Sclerosis.
Through this requirement, a physician can validate that a patient with chronic non-cancer pain, epilepsy, HIV, multiple sclerosis, or any of the various other conditions for which medical marijuana is often used is not merely using the prescription recreationally, sitting around the house in a fog watching "M*A*S*H" reruns on television all day, explained Dr. Ware, a family physician and anesthesiologist who is director of clinical research at the Alan Edwards Pain Management Unit at McGill University in Montreal.
Canada has had a federal program for medical marijuana in place for a dozen years. Dr. Ware has extensive experience in prescribing medical marijuana in the pain clinic, where many patients report improvement not only in pain, but in spasticity, sleep, and/or mood. In addition, he has led randomized clinical trials that demonstrated that smoked cannabis reduced pain intensity and improved sleep quality in patients with chronic neuropathic pain (CMAJ 2010;182:E694-701) and that oral nabilone (Cesamet), a synthetic cannabinoid, improved sleep and was well tolerated in patients with fibromyalgia (Anesth. Analg. 2010;110:604-10).
He offered these tips for physicians who have patients asking about medical marijuana:
The doses used are modest: A World Health Organization report estimated that the average joint contains 0.5 g of cannabis, and that the average dose in patients using marijuana medically is four joints per day, or roughly 2 g of cannabis. That equates to 20-50 mg/day of tetrahydrocannabinol, the active molecule, which is consistent with the results of clinical trials using standardized extracts.
"A watchful dose is 5 g of cannabis per day. I would be very, very cautious about anybody who’s asking for more than 5 g/day. The likelihood of diversion goes way up. There’s very little reason on pharmacologic grounds why a patient would need that much," Dr. Ware advised.
Not everyone responds to medical marijuana: As with any other medication, there are nonresponders. Because cannabis has been widely available recreationally for so long, an individual’s past recreational experience can be used as a rough predictor of the likelihood of response to medical marijuana.
"One of the tests I use when a patient with a chronic medical illness comes in asking if maybe a cannabis-based drug could be useful is I ask if they’ve ever used the drug before, say, in college or high school. If they say they did and got anxious and paranoid and hated it, that tells me they’re not cannabinoid responders. I have no scientific evidence for this, it’s just a clinical tool I use. Prior positive recreational responders, I suspect, are more likely to have a favorable effect," Dr. Ware continued.
Most patients are more concerned about medical marijuana’s safety than effectiveness: Medical cannabinoids are "overall quite safe," according to the family physician, who with coauthors has published a systemic review of the adverse effects (CMAJ 2008;178:1669-78). Cannabis has no associated toxicity even at extremely high doses. The prescription oral cannabinoids have no apparent abuse potential. While dependence is seen in some recreational marijuana smokers, it doesn’t seem to occur with clinical use.
And regarding the key safety concern for most patients and physicians – the question of smoked marijuana’s effects on the lung (see accompanying story) – a new analysis of the published literature by one of the world’s pre-eminent pulmonologists, Dr. Donald P. Tashkin, emeritus professor of medicine and medical director of the pulmonary function laboratory of the University of California, Los Angeles, concluded that "the accumulated weight of evidence" suggests regular smoking of marijuana alone doesn’t increase the risk of lung or upper airway cancer or [chronic obstructive pulmonary disease] , and the evidence is inconclusive regarding a possible associated risk of lower respiratory tract infection (Ann. Am. Thorac. Soc. 2013;10:239-47).
Medical marijuana is contraindicated in adolescents and patients with unstable ischemic heart disease or a personal or family history of psychosis: All of the clinical trials have screened for and excluded patients with a history of psychosis, either personally or in a first-degree relative. So there is no evidence supporting its safe use in such individuals.
A growing number of case reports suggest recreational cannabis use in young adolescents can trigger a latent psychotic episode in selected susceptible individuals. This is a major concern.
Cannabis is a powerful peripheral vasodilator. "The way to remember that is the red eyes of Bob Marley," Dr. Ware suggested. Peripheral vasodilation results in an increased heart rate, which could trigger an MI in a patient with unstable ischemic heart disease.
Always ask about legal issues: A surprisingly large number of patients inquiring about medical marijuana are under investigation for a crime and are seeking a stay-out-of-jail card. They won’t mention it if they’re not asked.
Consider the prescription alternatives to medical pot: Nabilone and Marinol (dronabinol, which is tetrahydrocannabinol), are approved for prescription use in the United States. The beneficial effects last longer than with smoked cannabis, and there is no uncertainty about the concentration, source, or possible contaminants.
Keep an eye out for improved technology: Smoking is a dirty delivery system for marijuana. While it’s not nearly as harmful to the lungs as smoking tobacco, as Dr. Tashkin recently concluded, cannabis smoke nevertheless does contain carcinogens and toxins. Vaporization devices are now commercially available as an alternative: a smokeless marijuana delivery system. This approach has recently been shown effective in a randomized, double-blind, placebo-controlled, crossover clinical trial conducted in patients with neuropathic pain (J. Pain 2013;14:136-48).
Dr. Ware has received lecture fees from the Canadian Consortium for the Investigation of the Cannabinoids and a research grant and honoraria from Valeant for conducting a randomized trial of nabilone in fibromyalgia patients.
ORLANDO – An essential element in prescribing medical marijuana responsibly is to insist that the patient must demonstrate improved functional status to be allowed to continue with the therapy, one expert has advised.
"Functional status is really the major issue. If cannabis is going to be used as a medicine, we have to see improvement in function: return to work, improvement in daily activities, engagement in society. I tell patients, ‘If this drug is really helping you, then show me. Show me that you can come off this other medication or reduce the dose. Show me you can go out and do volunteer work or join a club. Prove to me that this is valuable to you, because I cannot continue to authorize access to a substance if you cannot show me that it’s actually helping you improve your quality of life.’ That’s the gauntlet I throw down to patients," Dr. Mark A. Ware said at the fifth Cooperative Meeting sponsored by the Consortium of Multiple Sclerosis Centers and the Americas Committee for Treatment and Research in Multiple Sclerosis.
Through this requirement, a physician can validate that a patient with chronic non-cancer pain, epilepsy, HIV, multiple sclerosis, or any of the various other conditions for which medical marijuana is often used is not merely using the prescription recreationally, sitting around the house in a fog watching "M*A*S*H" reruns on television all day, explained Dr. Ware, a family physician and anesthesiologist who is director of clinical research at the Alan Edwards Pain Management Unit at McGill University in Montreal.
Canada has had a federal program for medical marijuana in place for a dozen years. Dr. Ware has extensive experience in prescribing medical marijuana in the pain clinic, where many patients report improvement not only in pain, but in spasticity, sleep, and/or mood. In addition, he has led randomized clinical trials that demonstrated that smoked cannabis reduced pain intensity and improved sleep quality in patients with chronic neuropathic pain (CMAJ 2010;182:E694-701) and that oral nabilone (Cesamet), a synthetic cannabinoid, improved sleep and was well tolerated in patients with fibromyalgia (Anesth. Analg. 2010;110:604-10).
He offered these tips for physicians who have patients asking about medical marijuana:
The doses used are modest: A World Health Organization report estimated that the average joint contains 0.5 g of cannabis, and that the average dose in patients using marijuana medically is four joints per day, or roughly 2 g of cannabis. That equates to 20-50 mg/day of tetrahydrocannabinol, the active molecule, which is consistent with the results of clinical trials using standardized extracts.
"A watchful dose is 5 g of cannabis per day. I would be very, very cautious about anybody who’s asking for more than 5 g/day. The likelihood of diversion goes way up. There’s very little reason on pharmacologic grounds why a patient would need that much," Dr. Ware advised.
Not everyone responds to medical marijuana: As with any other medication, there are nonresponders. Because cannabis has been widely available recreationally for so long, an individual’s past recreational experience can be used as a rough predictor of the likelihood of response to medical marijuana.
"One of the tests I use when a patient with a chronic medical illness comes in asking if maybe a cannabis-based drug could be useful is I ask if they’ve ever used the drug before, say, in college or high school. If they say they did and got anxious and paranoid and hated it, that tells me they’re not cannabinoid responders. I have no scientific evidence for this, it’s just a clinical tool I use. Prior positive recreational responders, I suspect, are more likely to have a favorable effect," Dr. Ware continued.
Most patients are more concerned about medical marijuana’s safety than effectiveness: Medical cannabinoids are "overall quite safe," according to the family physician, who with coauthors has published a systemic review of the adverse effects (CMAJ 2008;178:1669-78). Cannabis has no associated toxicity even at extremely high doses. The prescription oral cannabinoids have no apparent abuse potential. While dependence is seen in some recreational marijuana smokers, it doesn’t seem to occur with clinical use.
And regarding the key safety concern for most patients and physicians – the question of smoked marijuana’s effects on the lung (see accompanying story) – a new analysis of the published literature by one of the world’s pre-eminent pulmonologists, Dr. Donald P. Tashkin, emeritus professor of medicine and medical director of the pulmonary function laboratory of the University of California, Los Angeles, concluded that "the accumulated weight of evidence" suggests regular smoking of marijuana alone doesn’t increase the risk of lung or upper airway cancer or [chronic obstructive pulmonary disease] , and the evidence is inconclusive regarding a possible associated risk of lower respiratory tract infection (Ann. Am. Thorac. Soc. 2013;10:239-47).
Medical marijuana is contraindicated in adolescents and patients with unstable ischemic heart disease or a personal or family history of psychosis: All of the clinical trials have screened for and excluded patients with a history of psychosis, either personally or in a first-degree relative. So there is no evidence supporting its safe use in such individuals.
A growing number of case reports suggest recreational cannabis use in young adolescents can trigger a latent psychotic episode in selected susceptible individuals. This is a major concern.
Cannabis is a powerful peripheral vasodilator. "The way to remember that is the red eyes of Bob Marley," Dr. Ware suggested. Peripheral vasodilation results in an increased heart rate, which could trigger an MI in a patient with unstable ischemic heart disease.
Always ask about legal issues: A surprisingly large number of patients inquiring about medical marijuana are under investigation for a crime and are seeking a stay-out-of-jail card. They won’t mention it if they’re not asked.
Consider the prescription alternatives to medical pot: Nabilone and Marinol (dronabinol, which is tetrahydrocannabinol), are approved for prescription use in the United States. The beneficial effects last longer than with smoked cannabis, and there is no uncertainty about the concentration, source, or possible contaminants.
Keep an eye out for improved technology: Smoking is a dirty delivery system for marijuana. While it’s not nearly as harmful to the lungs as smoking tobacco, as Dr. Tashkin recently concluded, cannabis smoke nevertheless does contain carcinogens and toxins. Vaporization devices are now commercially available as an alternative: a smokeless marijuana delivery system. This approach has recently been shown effective in a randomized, double-blind, placebo-controlled, crossover clinical trial conducted in patients with neuropathic pain (J. Pain 2013;14:136-48).
Dr. Ware has received lecture fees from the Canadian Consortium for the Investigation of the Cannabinoids and a research grant and honoraria from Valeant for conducting a randomized trial of nabilone in fibromyalgia patients.
ORLANDO – An essential element in prescribing medical marijuana responsibly is to insist that the patient must demonstrate improved functional status to be allowed to continue with the therapy, one expert has advised.
"Functional status is really the major issue. If cannabis is going to be used as a medicine, we have to see improvement in function: return to work, improvement in daily activities, engagement in society. I tell patients, ‘If this drug is really helping you, then show me. Show me that you can come off this other medication or reduce the dose. Show me you can go out and do volunteer work or join a club. Prove to me that this is valuable to you, because I cannot continue to authorize access to a substance if you cannot show me that it’s actually helping you improve your quality of life.’ That’s the gauntlet I throw down to patients," Dr. Mark A. Ware said at the fifth Cooperative Meeting sponsored by the Consortium of Multiple Sclerosis Centers and the Americas Committee for Treatment and Research in Multiple Sclerosis.
Through this requirement, a physician can validate that a patient with chronic non-cancer pain, epilepsy, HIV, multiple sclerosis, or any of the various other conditions for which medical marijuana is often used is not merely using the prescription recreationally, sitting around the house in a fog watching "M*A*S*H" reruns on television all day, explained Dr. Ware, a family physician and anesthesiologist who is director of clinical research at the Alan Edwards Pain Management Unit at McGill University in Montreal.
Canada has had a federal program for medical marijuana in place for a dozen years. Dr. Ware has extensive experience in prescribing medical marijuana in the pain clinic, where many patients report improvement not only in pain, but in spasticity, sleep, and/or mood. In addition, he has led randomized clinical trials that demonstrated that smoked cannabis reduced pain intensity and improved sleep quality in patients with chronic neuropathic pain (CMAJ 2010;182:E694-701) and that oral nabilone (Cesamet), a synthetic cannabinoid, improved sleep and was well tolerated in patients with fibromyalgia (Anesth. Analg. 2010;110:604-10).
He offered these tips for physicians who have patients asking about medical marijuana:
The doses used are modest: A World Health Organization report estimated that the average joint contains 0.5 g of cannabis, and that the average dose in patients using marijuana medically is four joints per day, or roughly 2 g of cannabis. That equates to 20-50 mg/day of tetrahydrocannabinol, the active molecule, which is consistent with the results of clinical trials using standardized extracts.
"A watchful dose is 5 g of cannabis per day. I would be very, very cautious about anybody who’s asking for more than 5 g/day. The likelihood of diversion goes way up. There’s very little reason on pharmacologic grounds why a patient would need that much," Dr. Ware advised.
Not everyone responds to medical marijuana: As with any other medication, there are nonresponders. Because cannabis has been widely available recreationally for so long, an individual’s past recreational experience can be used as a rough predictor of the likelihood of response to medical marijuana.
"One of the tests I use when a patient with a chronic medical illness comes in asking if maybe a cannabis-based drug could be useful is I ask if they’ve ever used the drug before, say, in college or high school. If they say they did and got anxious and paranoid and hated it, that tells me they’re not cannabinoid responders. I have no scientific evidence for this, it’s just a clinical tool I use. Prior positive recreational responders, I suspect, are more likely to have a favorable effect," Dr. Ware continued.
Most patients are more concerned about medical marijuana’s safety than effectiveness: Medical cannabinoids are "overall quite safe," according to the family physician, who with coauthors has published a systemic review of the adverse effects (CMAJ 2008;178:1669-78). Cannabis has no associated toxicity even at extremely high doses. The prescription oral cannabinoids have no apparent abuse potential. While dependence is seen in some recreational marijuana smokers, it doesn’t seem to occur with clinical use.
And regarding the key safety concern for most patients and physicians – the question of smoked marijuana’s effects on the lung (see accompanying story) – a new analysis of the published literature by one of the world’s pre-eminent pulmonologists, Dr. Donald P. Tashkin, emeritus professor of medicine and medical director of the pulmonary function laboratory of the University of California, Los Angeles, concluded that "the accumulated weight of evidence" suggests regular smoking of marijuana alone doesn’t increase the risk of lung or upper airway cancer or [chronic obstructive pulmonary disease] , and the evidence is inconclusive regarding a possible associated risk of lower respiratory tract infection (Ann. Am. Thorac. Soc. 2013;10:239-47).
Medical marijuana is contraindicated in adolescents and patients with unstable ischemic heart disease or a personal or family history of psychosis: All of the clinical trials have screened for and excluded patients with a history of psychosis, either personally or in a first-degree relative. So there is no evidence supporting its safe use in such individuals.
A growing number of case reports suggest recreational cannabis use in young adolescents can trigger a latent psychotic episode in selected susceptible individuals. This is a major concern.
Cannabis is a powerful peripheral vasodilator. "The way to remember that is the red eyes of Bob Marley," Dr. Ware suggested. Peripheral vasodilation results in an increased heart rate, which could trigger an MI in a patient with unstable ischemic heart disease.
Always ask about legal issues: A surprisingly large number of patients inquiring about medical marijuana are under investigation for a crime and are seeking a stay-out-of-jail card. They won’t mention it if they’re not asked.
Consider the prescription alternatives to medical pot: Nabilone and Marinol (dronabinol, which is tetrahydrocannabinol), are approved for prescription use in the United States. The beneficial effects last longer than with smoked cannabis, and there is no uncertainty about the concentration, source, or possible contaminants.
Keep an eye out for improved technology: Smoking is a dirty delivery system for marijuana. While it’s not nearly as harmful to the lungs as smoking tobacco, as Dr. Tashkin recently concluded, cannabis smoke nevertheless does contain carcinogens and toxins. Vaporization devices are now commercially available as an alternative: a smokeless marijuana delivery system. This approach has recently been shown effective in a randomized, double-blind, placebo-controlled, crossover clinical trial conducted in patients with neuropathic pain (J. Pain 2013;14:136-48).
Dr. Ware has received lecture fees from the Canadian Consortium for the Investigation of the Cannabinoids and a research grant and honoraria from Valeant for conducting a randomized trial of nabilone in fibromyalgia patients.
AT THE CMSC/ACTRIMS ANNUAL MEETING
Novel insulin/GLP-1 combo succeeds in phase III trial
CHICAGO – IDegLira, a fixed-ratio combination of insulin degludec and the glucagonlike peptide-1 agonist liraglutide, markedly outperformed each medication alone in a pivotal phase III clinical trial in patients with type 2 diabetes.
IDegLira combines the effects of insulin degludec and liraglutide in one daily injection, resulting in a substantial overall improvement in glycemic control with a low risk of hypoglycemia, weight gain, and GI adverse events," Dr. John B. Buse said in presenting the results of the phase III DUAL I (Dual Action of Liraglutide and Insulin Degludec in Type 2 Diabetes) study at the annual scientific sessions of the American Diabetes Association.

The therapeutic rationale for the combination lies in the complementary characteristics of the two components. The net result is enhanced efficacy along with partial cross-cancellation of each drug’s side effects.
"Basal insulin is arguably the most powerful approach for controlling fasting blood glucose, and GLP-1 agonists have potent postprandial effects. The weight reduction associated with GLP-1 agonists could help mitigate the weight gain associated with basal insulin. The effects on alpha-cell function of GLP-agonists might reduce the hypoglycemia risk associated with basal insulin. And lastly, the process of individualized dosing that we use with basal insulin – getting the dose just right with regard to efficacy and safety – those concepts might reduce the GI side effects that are common with GLP-1 agonists," explained Dr. Buse, professor of medicine, chief of the division of endocrinology and metabolism, and executive associate dean for clinical research at the University of North Carolina, Chapel Hill.
All of these hypothesized benefits actually came to pass in DUAL I, a 26-week, international, open-label, phase III trial involving 1,663 type 2 diabetic subjects, all insulin-naive and inadequately controlled on metformin with or without pioglitazone. Eighty-three percent of patients were on metformin alone at enrollment, with the remainder on dual oral therapy.
Participants remained on their previous oral antidiabetic regimen during the 26-week study. They were randomized 2:1:1 to IDegLira, insulin degludec, or liraglutide (Victoza). Patients in the two insulin arms were started on 10 U/day of insulin degludec and titrated twice weekly in 2-unit increments, based upon their mean fasting plasma glucose. Ten units of IDegLira contain that amount of insulin degludec plus 0.36 mg of liraglutide. The maximum permitted dose of IDegLira contained 50 U of insulin degludec and 1.8 mg of liraglutide.
The primary study endpoint was reduction in hemoglobin A1c over the course of 26 weeks. Patients on IDegLira averaged a 1.91% drop to an HbA1c of 6.4%, significantly better than the 1.44% drop to 6.9% with insulin degludec and the 1.28% reduction to an HbA1c of 7.0% with liraglutide. In addition, the IDegLira group fared significantly better in terms of numerous secondary endpoints.
Particularly noteworthy, Dr. Buse continued, was the fact that fasting plasma glucose was reduced by an identical 65 mg/dL from a baseline of 165 mg/dL in the two-insulin treatment groups, even though the final mean daily dose of insulin degludec in the IDegLira group was 38 U, or 15 U/day less than in the group on insulin degludec alone. Confirmed hypoglycemic events occurred at a rate of 1.2 events over 26 weeks in the insulin degludec group and at a 32% lower rate in patients on IDegLira.
"That’s stunning in light of the fact that there was a 0.5% greater reduction in [HbA1c] in the IDegLira group," Dr. Buse observed.
Nausea was an issue for patients assigned to liraglutide. Roughly 10% of them per week reported nausea during titration, a rate that eventually fell off to about 3% per week after 14 weeks. In contrast, the proportion of patients in the IDegLira group who reported any nausea at all during the study was less than half that in the liraglutide group.
"This is likely related more to the titration program and the lower starting dose in the IDegLira group than to the final total milligrams of liraglutide," according to Dr. Buse. He noted that the final daily dose of liraglutide in the IDegLira group averaged 1.4 mg, compared with 1.8 mg in patients randomized to GLP-1 agonist monotherapy.
Earlier this year, Novo Nordisk, which is developing IDegLira, filed for marketing approval for the investigational agent from the European Medicines Agency. The company plans to file with the Food and Drug Administration later this year. However, the approval for insulin degludec was stalled after the FDA requested more cardiovascular safety data from the company in February 2013.
Session chair Julio Rosenstock of the Dallas Diabetes and Endocrine Center commented that the fixed-ratio combination is "a great concept" and hailed the phase III findings as "very, very impressive results." He wished, however, that the study had been set up blinded rather than open label.
Dr. Buse replied that a companion phase III trial, known as DUAL II,is in fact a blinded comparison. The results will be presented this fall in Barcelona at the annual meeting of the European Association for the Study of Diabetes.
Dr. Rosenstock called the reduction in hypoglycemic events in the IDegLira group particularly impressive, given the hefty decrease in HbA1c in that group. He asked for Dr. Buse’s thoughts as to the explanation.
It’s possibly in part a consequence of the IDegLira group’s lesser daily insulin dose, Dr. Buse replied, but liraglutide-induced improvement in alpha-cell function probably plays a bigger role.
He reported serving as a consultant to Novo Nordisk and more than 30 other pharmaceutical companies.
CHICAGO – IDegLira, a fixed-ratio combination of insulin degludec and the glucagonlike peptide-1 agonist liraglutide, markedly outperformed each medication alone in a pivotal phase III clinical trial in patients with type 2 diabetes.
IDegLira combines the effects of insulin degludec and liraglutide in one daily injection, resulting in a substantial overall improvement in glycemic control with a low risk of hypoglycemia, weight gain, and GI adverse events," Dr. John B. Buse said in presenting the results of the phase III DUAL I (Dual Action of Liraglutide and Insulin Degludec in Type 2 Diabetes) study at the annual scientific sessions of the American Diabetes Association.

The therapeutic rationale for the combination lies in the complementary characteristics of the two components. The net result is enhanced efficacy along with partial cross-cancellation of each drug’s side effects.
"Basal insulin is arguably the most powerful approach for controlling fasting blood glucose, and GLP-1 agonists have potent postprandial effects. The weight reduction associated with GLP-1 agonists could help mitigate the weight gain associated with basal insulin. The effects on alpha-cell function of GLP-agonists might reduce the hypoglycemia risk associated with basal insulin. And lastly, the process of individualized dosing that we use with basal insulin – getting the dose just right with regard to efficacy and safety – those concepts might reduce the GI side effects that are common with GLP-1 agonists," explained Dr. Buse, professor of medicine, chief of the division of endocrinology and metabolism, and executive associate dean for clinical research at the University of North Carolina, Chapel Hill.
All of these hypothesized benefits actually came to pass in DUAL I, a 26-week, international, open-label, phase III trial involving 1,663 type 2 diabetic subjects, all insulin-naive and inadequately controlled on metformin with or without pioglitazone. Eighty-three percent of patients were on metformin alone at enrollment, with the remainder on dual oral therapy.
Participants remained on their previous oral antidiabetic regimen during the 26-week study. They were randomized 2:1:1 to IDegLira, insulin degludec, or liraglutide (Victoza). Patients in the two insulin arms were started on 10 U/day of insulin degludec and titrated twice weekly in 2-unit increments, based upon their mean fasting plasma glucose. Ten units of IDegLira contain that amount of insulin degludec plus 0.36 mg of liraglutide. The maximum permitted dose of IDegLira contained 50 U of insulin degludec and 1.8 mg of liraglutide.
The primary study endpoint was reduction in hemoglobin A1c over the course of 26 weeks. Patients on IDegLira averaged a 1.91% drop to an HbA1c of 6.4%, significantly better than the 1.44% drop to 6.9% with insulin degludec and the 1.28% reduction to an HbA1c of 7.0% with liraglutide. In addition, the IDegLira group fared significantly better in terms of numerous secondary endpoints.
Particularly noteworthy, Dr. Buse continued, was the fact that fasting plasma glucose was reduced by an identical 65 mg/dL from a baseline of 165 mg/dL in the two-insulin treatment groups, even though the final mean daily dose of insulin degludec in the IDegLira group was 38 U, or 15 U/day less than in the group on insulin degludec alone. Confirmed hypoglycemic events occurred at a rate of 1.2 events over 26 weeks in the insulin degludec group and at a 32% lower rate in patients on IDegLira.
"That’s stunning in light of the fact that there was a 0.5% greater reduction in [HbA1c] in the IDegLira group," Dr. Buse observed.
Nausea was an issue for patients assigned to liraglutide. Roughly 10% of them per week reported nausea during titration, a rate that eventually fell off to about 3% per week after 14 weeks. In contrast, the proportion of patients in the IDegLira group who reported any nausea at all during the study was less than half that in the liraglutide group.
"This is likely related more to the titration program and the lower starting dose in the IDegLira group than to the final total milligrams of liraglutide," according to Dr. Buse. He noted that the final daily dose of liraglutide in the IDegLira group averaged 1.4 mg, compared with 1.8 mg in patients randomized to GLP-1 agonist monotherapy.
Earlier this year, Novo Nordisk, which is developing IDegLira, filed for marketing approval for the investigational agent from the European Medicines Agency. The company plans to file with the Food and Drug Administration later this year. However, the approval for insulin degludec was stalled after the FDA requested more cardiovascular safety data from the company in February 2013.
Session chair Julio Rosenstock of the Dallas Diabetes and Endocrine Center commented that the fixed-ratio combination is "a great concept" and hailed the phase III findings as "very, very impressive results." He wished, however, that the study had been set up blinded rather than open label.
Dr. Buse replied that a companion phase III trial, known as DUAL II,is in fact a blinded comparison. The results will be presented this fall in Barcelona at the annual meeting of the European Association for the Study of Diabetes.
Dr. Rosenstock called the reduction in hypoglycemic events in the IDegLira group particularly impressive, given the hefty decrease in HbA1c in that group. He asked for Dr. Buse’s thoughts as to the explanation.
It’s possibly in part a consequence of the IDegLira group’s lesser daily insulin dose, Dr. Buse replied, but liraglutide-induced improvement in alpha-cell function probably plays a bigger role.
He reported serving as a consultant to Novo Nordisk and more than 30 other pharmaceutical companies.
CHICAGO – IDegLira, a fixed-ratio combination of insulin degludec and the glucagonlike peptide-1 agonist liraglutide, markedly outperformed each medication alone in a pivotal phase III clinical trial in patients with type 2 diabetes.
IDegLira combines the effects of insulin degludec and liraglutide in one daily injection, resulting in a substantial overall improvement in glycemic control with a low risk of hypoglycemia, weight gain, and GI adverse events," Dr. John B. Buse said in presenting the results of the phase III DUAL I (Dual Action of Liraglutide and Insulin Degludec in Type 2 Diabetes) study at the annual scientific sessions of the American Diabetes Association.

The therapeutic rationale for the combination lies in the complementary characteristics of the two components. The net result is enhanced efficacy along with partial cross-cancellation of each drug’s side effects.
"Basal insulin is arguably the most powerful approach for controlling fasting blood glucose, and GLP-1 agonists have potent postprandial effects. The weight reduction associated with GLP-1 agonists could help mitigate the weight gain associated with basal insulin. The effects on alpha-cell function of GLP-agonists might reduce the hypoglycemia risk associated with basal insulin. And lastly, the process of individualized dosing that we use with basal insulin – getting the dose just right with regard to efficacy and safety – those concepts might reduce the GI side effects that are common with GLP-1 agonists," explained Dr. Buse, professor of medicine, chief of the division of endocrinology and metabolism, and executive associate dean for clinical research at the University of North Carolina, Chapel Hill.
All of these hypothesized benefits actually came to pass in DUAL I, a 26-week, international, open-label, phase III trial involving 1,663 type 2 diabetic subjects, all insulin-naive and inadequately controlled on metformin with or without pioglitazone. Eighty-three percent of patients were on metformin alone at enrollment, with the remainder on dual oral therapy.
Participants remained on their previous oral antidiabetic regimen during the 26-week study. They were randomized 2:1:1 to IDegLira, insulin degludec, or liraglutide (Victoza). Patients in the two insulin arms were started on 10 U/day of insulin degludec and titrated twice weekly in 2-unit increments, based upon their mean fasting plasma glucose. Ten units of IDegLira contain that amount of insulin degludec plus 0.36 mg of liraglutide. The maximum permitted dose of IDegLira contained 50 U of insulin degludec and 1.8 mg of liraglutide.
The primary study endpoint was reduction in hemoglobin A1c over the course of 26 weeks. Patients on IDegLira averaged a 1.91% drop to an HbA1c of 6.4%, significantly better than the 1.44% drop to 6.9% with insulin degludec and the 1.28% reduction to an HbA1c of 7.0% with liraglutide. In addition, the IDegLira group fared significantly better in terms of numerous secondary endpoints.
Particularly noteworthy, Dr. Buse continued, was the fact that fasting plasma glucose was reduced by an identical 65 mg/dL from a baseline of 165 mg/dL in the two-insulin treatment groups, even though the final mean daily dose of insulin degludec in the IDegLira group was 38 U, or 15 U/day less than in the group on insulin degludec alone. Confirmed hypoglycemic events occurred at a rate of 1.2 events over 26 weeks in the insulin degludec group and at a 32% lower rate in patients on IDegLira.
"That’s stunning in light of the fact that there was a 0.5% greater reduction in [HbA1c] in the IDegLira group," Dr. Buse observed.
Nausea was an issue for patients assigned to liraglutide. Roughly 10% of them per week reported nausea during titration, a rate that eventually fell off to about 3% per week after 14 weeks. In contrast, the proportion of patients in the IDegLira group who reported any nausea at all during the study was less than half that in the liraglutide group.
"This is likely related more to the titration program and the lower starting dose in the IDegLira group than to the final total milligrams of liraglutide," according to Dr. Buse. He noted that the final daily dose of liraglutide in the IDegLira group averaged 1.4 mg, compared with 1.8 mg in patients randomized to GLP-1 agonist monotherapy.
Earlier this year, Novo Nordisk, which is developing IDegLira, filed for marketing approval for the investigational agent from the European Medicines Agency. The company plans to file with the Food and Drug Administration later this year. However, the approval for insulin degludec was stalled after the FDA requested more cardiovascular safety data from the company in February 2013.
Session chair Julio Rosenstock of the Dallas Diabetes and Endocrine Center commented that the fixed-ratio combination is "a great concept" and hailed the phase III findings as "very, very impressive results." He wished, however, that the study had been set up blinded rather than open label.
Dr. Buse replied that a companion phase III trial, known as DUAL II,is in fact a blinded comparison. The results will be presented this fall in Barcelona at the annual meeting of the European Association for the Study of Diabetes.
Dr. Rosenstock called the reduction in hypoglycemic events in the IDegLira group particularly impressive, given the hefty decrease in HbA1c in that group. He asked for Dr. Buse’s thoughts as to the explanation.
It’s possibly in part a consequence of the IDegLira group’s lesser daily insulin dose, Dr. Buse replied, but liraglutide-induced improvement in alpha-cell function probably plays a bigger role.
He reported serving as a consultant to Novo Nordisk and more than 30 other pharmaceutical companies.
AT THE ADA ANNUAL SCIENTIFIC SESSIONS
Major finding: Patients with type 2 diabetes not adequately controlled on oral therapy experienced a mean 1.91% reduction in HbA1c to a final value of 6.4% in response to the addition of once-daily subcutaneous injections of IDegLira, a fixed-ratio combination of insulin degludec and liraglutide.
Data source: This was a pivotal phase III, 26-week, open-label study in which 1,663 adults with inadequately controlled type 2 diabetes on oral medication were randomized to IDegLira, insulin degludec, or liraglutide.
Disclosures: The study was sponsored by Novo Nordisk. The presenter is a consultant to the company.
Add-on dapagliflozin achieves combined weight/HbA1c reduction more often than glipizide
CHICAGO – Dapagliflozin and glipizide achieve comparable hemoglobin A1c reduction as add-on therapy in patients with type 2 diabetes inadequately controlled on metformin, but investigational dapagliflozin is only one-tenth as likely to produce hypoglycemia and is far more impressive in terms of weight reduction.
Those were the key findings of a 52-week, double-blind, randomized phase III trial in 814 patients who remained on their metformin following randomization to dapagliflozin at up to 10 mg/day or glipizide at up to 20 mg/day, Dr. Katja Rohwedder reported at the annual scientific sessions of the American Diabetes Association.
Dapagliflozin is a member of the emerging new class of oral antidiabetic drugs known as sodium glucose cotransporter–2 (SGLT2) inhibitors. Their novel mechanism of reducing hyperglycemia involves boosting urinary glucose excretion without affecting insulin secretion or action. Dapagliflozin was the first-in-class SGLT2 inhibitor to emerge from the developmental pipeline and seek regulatory approval. While it has been approved by the European Medicines Agency and is marketed as Forxiga in Europe for the treatment of type 2 diabetes, the Food and Drug Administration rejected the drug pending receipt of additional data to clarify an observed possible small excess of cases of breast and bladder cancer in the clinical trials. Following that action, the FDA proceeded to approve canagliflozin (Invokana) as the first SGLT2 inhibitor in the United States.
Dr. Rohwedder reported that 74.7% of the dapagliflozin group and a closely similar 73.8% of the glipizide group in the phase III add-on trial achieved an HbA1c reduction at 52 weeks. Weight reduction was a different story, however: it was achieved in 83.5% of the dapagliflozin group, compared with 26.8% with glipizide.
The combination of HbA1c and weight reduction occurred in 67% of the dapagliflozin group, a rate more than threefold greater than the 21% figure in the glipizide arm of the study, according to Dr. Rohwedder of AstraZeneca in Wedel, Germany
A second analysis utilized a more rigorous combined endpoint requiring at least a 0.5% reduction in HbA1c level and a weight loss of 3 kg or more at 52 weeks. This was achieved in one-third of the add-on dapagliflozin group, compared with less than 4% of patients on the add-on glipizide.
"I hope you will agree that this is meaningful to our patients," she said.
Hypoglycemic events occurred in 3.5% of the dapagliflozin group, compared with 40.8% with glipizide. All three serious cases involved patients on glipizide.
Genital infections occurred in 10.8% of patients on dapagliflozin and 1.7% of those on glipizide. Urinary tract infections were also more frequent with SGLT2 inhibitor therapy, by a margin of 8.6% to 5.4%.
"These events responded well to standard therapy and only rarely led to study discontinuation," according to the physician.
Session chair Dr. Ralph A. DeFronzo commented that "this study shows a pretty clear-cut distinction between these two oral agents in terms of weight gain, an important side effect that we’re all concerned about in the diabetes field."
No cases of breast or bladder cancer were reported in this 52-week study.
The time may be here "to get rid of the sulfonylureas and go to drugs that really work, without causing safety issues," suggested Dr. DeFronzo, professor of medicine and chief of the diabetes division at the University of Texas Health Science Center at San Antonio.
The phase III study was supported by Bristol-Myers Squibb and AstraZeneca, which are jointly developing dapagliflozin. Dr. Rohwedder is an employee of AstraZeneca.
CHICAGO – Dapagliflozin and glipizide achieve comparable hemoglobin A1c reduction as add-on therapy in patients with type 2 diabetes inadequately controlled on metformin, but investigational dapagliflozin is only one-tenth as likely to produce hypoglycemia and is far more impressive in terms of weight reduction.
Those were the key findings of a 52-week, double-blind, randomized phase III trial in 814 patients who remained on their metformin following randomization to dapagliflozin at up to 10 mg/day or glipizide at up to 20 mg/day, Dr. Katja Rohwedder reported at the annual scientific sessions of the American Diabetes Association.
Dapagliflozin is a member of the emerging new class of oral antidiabetic drugs known as sodium glucose cotransporter–2 (SGLT2) inhibitors. Their novel mechanism of reducing hyperglycemia involves boosting urinary glucose excretion without affecting insulin secretion or action. Dapagliflozin was the first-in-class SGLT2 inhibitor to emerge from the developmental pipeline and seek regulatory approval. While it has been approved by the European Medicines Agency and is marketed as Forxiga in Europe for the treatment of type 2 diabetes, the Food and Drug Administration rejected the drug pending receipt of additional data to clarify an observed possible small excess of cases of breast and bladder cancer in the clinical trials. Following that action, the FDA proceeded to approve canagliflozin (Invokana) as the first SGLT2 inhibitor in the United States.
Dr. Rohwedder reported that 74.7% of the dapagliflozin group and a closely similar 73.8% of the glipizide group in the phase III add-on trial achieved an HbA1c reduction at 52 weeks. Weight reduction was a different story, however: it was achieved in 83.5% of the dapagliflozin group, compared with 26.8% with glipizide.
The combination of HbA1c and weight reduction occurred in 67% of the dapagliflozin group, a rate more than threefold greater than the 21% figure in the glipizide arm of the study, according to Dr. Rohwedder of AstraZeneca in Wedel, Germany
A second analysis utilized a more rigorous combined endpoint requiring at least a 0.5% reduction in HbA1c level and a weight loss of 3 kg or more at 52 weeks. This was achieved in one-third of the add-on dapagliflozin group, compared with less than 4% of patients on the add-on glipizide.
"I hope you will agree that this is meaningful to our patients," she said.
Hypoglycemic events occurred in 3.5% of the dapagliflozin group, compared with 40.8% with glipizide. All three serious cases involved patients on glipizide.
Genital infections occurred in 10.8% of patients on dapagliflozin and 1.7% of those on glipizide. Urinary tract infections were also more frequent with SGLT2 inhibitor therapy, by a margin of 8.6% to 5.4%.
"These events responded well to standard therapy and only rarely led to study discontinuation," according to the physician.
Session chair Dr. Ralph A. DeFronzo commented that "this study shows a pretty clear-cut distinction between these two oral agents in terms of weight gain, an important side effect that we’re all concerned about in the diabetes field."
No cases of breast or bladder cancer were reported in this 52-week study.
The time may be here "to get rid of the sulfonylureas and go to drugs that really work, without causing safety issues," suggested Dr. DeFronzo, professor of medicine and chief of the diabetes division at the University of Texas Health Science Center at San Antonio.
The phase III study was supported by Bristol-Myers Squibb and AstraZeneca, which are jointly developing dapagliflozin. Dr. Rohwedder is an employee of AstraZeneca.
CHICAGO – Dapagliflozin and glipizide achieve comparable hemoglobin A1c reduction as add-on therapy in patients with type 2 diabetes inadequately controlled on metformin, but investigational dapagliflozin is only one-tenth as likely to produce hypoglycemia and is far more impressive in terms of weight reduction.
Those were the key findings of a 52-week, double-blind, randomized phase III trial in 814 patients who remained on their metformin following randomization to dapagliflozin at up to 10 mg/day or glipizide at up to 20 mg/day, Dr. Katja Rohwedder reported at the annual scientific sessions of the American Diabetes Association.
Dapagliflozin is a member of the emerging new class of oral antidiabetic drugs known as sodium glucose cotransporter–2 (SGLT2) inhibitors. Their novel mechanism of reducing hyperglycemia involves boosting urinary glucose excretion without affecting insulin secretion or action. Dapagliflozin was the first-in-class SGLT2 inhibitor to emerge from the developmental pipeline and seek regulatory approval. While it has been approved by the European Medicines Agency and is marketed as Forxiga in Europe for the treatment of type 2 diabetes, the Food and Drug Administration rejected the drug pending receipt of additional data to clarify an observed possible small excess of cases of breast and bladder cancer in the clinical trials. Following that action, the FDA proceeded to approve canagliflozin (Invokana) as the first SGLT2 inhibitor in the United States.
Dr. Rohwedder reported that 74.7% of the dapagliflozin group and a closely similar 73.8% of the glipizide group in the phase III add-on trial achieved an HbA1c reduction at 52 weeks. Weight reduction was a different story, however: it was achieved in 83.5% of the dapagliflozin group, compared with 26.8% with glipizide.
The combination of HbA1c and weight reduction occurred in 67% of the dapagliflozin group, a rate more than threefold greater than the 21% figure in the glipizide arm of the study, according to Dr. Rohwedder of AstraZeneca in Wedel, Germany
A second analysis utilized a more rigorous combined endpoint requiring at least a 0.5% reduction in HbA1c level and a weight loss of 3 kg or more at 52 weeks. This was achieved in one-third of the add-on dapagliflozin group, compared with less than 4% of patients on the add-on glipizide.
"I hope you will agree that this is meaningful to our patients," she said.
Hypoglycemic events occurred in 3.5% of the dapagliflozin group, compared with 40.8% with glipizide. All three serious cases involved patients on glipizide.
Genital infections occurred in 10.8% of patients on dapagliflozin and 1.7% of those on glipizide. Urinary tract infections were also more frequent with SGLT2 inhibitor therapy, by a margin of 8.6% to 5.4%.
"These events responded well to standard therapy and only rarely led to study discontinuation," according to the physician.
Session chair Dr. Ralph A. DeFronzo commented that "this study shows a pretty clear-cut distinction between these two oral agents in terms of weight gain, an important side effect that we’re all concerned about in the diabetes field."
No cases of breast or bladder cancer were reported in this 52-week study.
The time may be here "to get rid of the sulfonylureas and go to drugs that really work, without causing safety issues," suggested Dr. DeFronzo, professor of medicine and chief of the diabetes division at the University of Texas Health Science Center at San Antonio.
The phase III study was supported by Bristol-Myers Squibb and AstraZeneca, which are jointly developing dapagliflozin. Dr. Rohwedder is an employee of AstraZeneca.
AT THE ADA ANNUAL SCIENTIFIC SESSIONS
Major Finding: At week 52, 67% of type 2 diabetes patients on dapagliflozin had reductions in both HbA1c and weight, compared with 21% on glipizide.
Data Source: A phase III, double-blind, 52-week randomized trial of the add-on dapagliflozin or glipizide in 814 patients with type 2 diabetes not adequately controlled with metformin.
Disclosures: The phase III study was supported by Bristol-Myers Squibb and AstraZeneca, which are jointly developing dapagliflozin. Dr. Rohwedder is an employee of AstraZeneca.
Aging-related functional decline boosts diabetes risk
CHICAGO – Aging-related decline in physical functioning has emerged as a previously unrecognized independent risk factor for new-onset diabetes, according to investigators at the Centers for Disease Control and Prevention.
In a multivariate analysis involving 22,876 subjects over age 50 years at baseline in the longitudinal, prospective Health and Retirement Study, those who had mild or moderate functional decline at enrollment or who developed it during an average 8.7 years of follow-up had a 17% increased risk of subsequently being diagnosed with new-onset diabetes, compared with subjects who did not have prevalent or incident functional decline or physical disability, Barbara H. Bardenheier, Ph.D., reported at the annual scientific sessions of the American Diabetes Association.
Subjects who had severe functional decline at baseline or developed it during follow-up had a 12% excess risk of subsequent diabetes. Like mild-to-moderate functional decline, severe functional decline was also a statistically significant risk factor for later diagnosis of diabetes in a multivariate analysis adjusted for age, race, education, baseline body mass index, and socioeconomic factors. The excess diabetes risk in individuals with prevalent or incident severe functional decline would have been higher but for the fact that they also had a 2.3-fold increased risk of mortality, compared with participants without aging-related functional decline, observed Dr. Bardenheier of the CDC in Atlanta.
It has been shown consistently in longitudinal and cross-sectional studies that diabetes is associated with an increased risk of subsequent physical disability, but this analysis of the Health and Retirement Study is the first to show the converse: that aging-related functional decline and physical disability or frailty place an individual at increased risk for subsequent diabetes, she added.
The definitions of aging-related functional decline employed in this study were based on difficulty expected to last longer than 3 months in performing specific mobility measures. Five mobility measures were used: walking one block; walking several blocks; stooping, crouching, or kneeling; climbing one flight of stairs, and pushing or pulling a large object.
Mild functional decline was defined as difficulty in stooping and walking several blocks or difficulty in any two mobility measures other than stair-climbing. Moderate functional decline required either difficulty in climbing a single flight of stairs or difficulty with any three of the other mobility measures. Severe decline was defined as difficulty with at least four mobility measures.
Among the 13,143 study participants who did not have aging-related functional decline at baseline, those who developed mild functional decline during follow-up had an adjusted 19% excess risk of being diagnosed with diabetes afterward. Those who developed moderate decline had a 30% excess risk of subsequent diabetes, while those with incident severe functional decline had a 16% increased risk of diabetes along with a 2.4-fold increased mortality risk. During follow-up, 18.8% of subjects without baseline aging-related functional decline developed moderate or severe decline.
Of note, 77% of participants reported some level of functional decline during the study period. During follow-up, 15.5% of subjects developed diabetes and 25.6% of subjects died.
The clinical relevance of this new observation that aging-related functional decline is a risk factor for subsequent diabetes lies in the fact that prior studies have shown functional decline is potentially modifiable. For example, physical exercise programs geared for older patients with moderate physical frailty have been shown to be protective against further disability, according to Dr. Bardenheier.
The Health and Retirement Study is sponsored by the National Institute on Aging. Dr. Bardenheier’s analysis was funded by the CDC. She reported having no conflicts of interest.
CHICAGO – Aging-related decline in physical functioning has emerged as a previously unrecognized independent risk factor for new-onset diabetes, according to investigators at the Centers for Disease Control and Prevention.
In a multivariate analysis involving 22,876 subjects over age 50 years at baseline in the longitudinal, prospective Health and Retirement Study, those who had mild or moderate functional decline at enrollment or who developed it during an average 8.7 years of follow-up had a 17% increased risk of subsequently being diagnosed with new-onset diabetes, compared with subjects who did not have prevalent or incident functional decline or physical disability, Barbara H. Bardenheier, Ph.D., reported at the annual scientific sessions of the American Diabetes Association.
Subjects who had severe functional decline at baseline or developed it during follow-up had a 12% excess risk of subsequent diabetes. Like mild-to-moderate functional decline, severe functional decline was also a statistically significant risk factor for later diagnosis of diabetes in a multivariate analysis adjusted for age, race, education, baseline body mass index, and socioeconomic factors. The excess diabetes risk in individuals with prevalent or incident severe functional decline would have been higher but for the fact that they also had a 2.3-fold increased risk of mortality, compared with participants without aging-related functional decline, observed Dr. Bardenheier of the CDC in Atlanta.
It has been shown consistently in longitudinal and cross-sectional studies that diabetes is associated with an increased risk of subsequent physical disability, but this analysis of the Health and Retirement Study is the first to show the converse: that aging-related functional decline and physical disability or frailty place an individual at increased risk for subsequent diabetes, she added.
The definitions of aging-related functional decline employed in this study were based on difficulty expected to last longer than 3 months in performing specific mobility measures. Five mobility measures were used: walking one block; walking several blocks; stooping, crouching, or kneeling; climbing one flight of stairs, and pushing or pulling a large object.
Mild functional decline was defined as difficulty in stooping and walking several blocks or difficulty in any two mobility measures other than stair-climbing. Moderate functional decline required either difficulty in climbing a single flight of stairs or difficulty with any three of the other mobility measures. Severe decline was defined as difficulty with at least four mobility measures.
Among the 13,143 study participants who did not have aging-related functional decline at baseline, those who developed mild functional decline during follow-up had an adjusted 19% excess risk of being diagnosed with diabetes afterward. Those who developed moderate decline had a 30% excess risk of subsequent diabetes, while those with incident severe functional decline had a 16% increased risk of diabetes along with a 2.4-fold increased mortality risk. During follow-up, 18.8% of subjects without baseline aging-related functional decline developed moderate or severe decline.
Of note, 77% of participants reported some level of functional decline during the study period. During follow-up, 15.5% of subjects developed diabetes and 25.6% of subjects died.
The clinical relevance of this new observation that aging-related functional decline is a risk factor for subsequent diabetes lies in the fact that prior studies have shown functional decline is potentially modifiable. For example, physical exercise programs geared for older patients with moderate physical frailty have been shown to be protective against further disability, according to Dr. Bardenheier.
The Health and Retirement Study is sponsored by the National Institute on Aging. Dr. Bardenheier’s analysis was funded by the CDC. She reported having no conflicts of interest.
CHICAGO – Aging-related decline in physical functioning has emerged as a previously unrecognized independent risk factor for new-onset diabetes, according to investigators at the Centers for Disease Control and Prevention.
In a multivariate analysis involving 22,876 subjects over age 50 years at baseline in the longitudinal, prospective Health and Retirement Study, those who had mild or moderate functional decline at enrollment or who developed it during an average 8.7 years of follow-up had a 17% increased risk of subsequently being diagnosed with new-onset diabetes, compared with subjects who did not have prevalent or incident functional decline or physical disability, Barbara H. Bardenheier, Ph.D., reported at the annual scientific sessions of the American Diabetes Association.
Subjects who had severe functional decline at baseline or developed it during follow-up had a 12% excess risk of subsequent diabetes. Like mild-to-moderate functional decline, severe functional decline was also a statistically significant risk factor for later diagnosis of diabetes in a multivariate analysis adjusted for age, race, education, baseline body mass index, and socioeconomic factors. The excess diabetes risk in individuals with prevalent or incident severe functional decline would have been higher but for the fact that they also had a 2.3-fold increased risk of mortality, compared with participants without aging-related functional decline, observed Dr. Bardenheier of the CDC in Atlanta.
It has been shown consistently in longitudinal and cross-sectional studies that diabetes is associated with an increased risk of subsequent physical disability, but this analysis of the Health and Retirement Study is the first to show the converse: that aging-related functional decline and physical disability or frailty place an individual at increased risk for subsequent diabetes, she added.
The definitions of aging-related functional decline employed in this study were based on difficulty expected to last longer than 3 months in performing specific mobility measures. Five mobility measures were used: walking one block; walking several blocks; stooping, crouching, or kneeling; climbing one flight of stairs, and pushing or pulling a large object.
Mild functional decline was defined as difficulty in stooping and walking several blocks or difficulty in any two mobility measures other than stair-climbing. Moderate functional decline required either difficulty in climbing a single flight of stairs or difficulty with any three of the other mobility measures. Severe decline was defined as difficulty with at least four mobility measures.
Among the 13,143 study participants who did not have aging-related functional decline at baseline, those who developed mild functional decline during follow-up had an adjusted 19% excess risk of being diagnosed with diabetes afterward. Those who developed moderate decline had a 30% excess risk of subsequent diabetes, while those with incident severe functional decline had a 16% increased risk of diabetes along with a 2.4-fold increased mortality risk. During follow-up, 18.8% of subjects without baseline aging-related functional decline developed moderate or severe decline.
Of note, 77% of participants reported some level of functional decline during the study period. During follow-up, 15.5% of subjects developed diabetes and 25.6% of subjects died.
The clinical relevance of this new observation that aging-related functional decline is a risk factor for subsequent diabetes lies in the fact that prior studies have shown functional decline is potentially modifiable. For example, physical exercise programs geared for older patients with moderate physical frailty have been shown to be protective against further disability, according to Dr. Bardenheier.
The Health and Retirement Study is sponsored by the National Institute on Aging. Dr. Bardenheier’s analysis was funded by the CDC. She reported having no conflicts of interest.
AT THE ADA ANNUAL SCIENTIFIC SESSIONS
Major Finding: New-onset mild aging-related decline in physical functioning resulted in a 19% excess risk of subsequently developing diabetes, while incident moderate functional decline resulted in a 30% increased risk and severe decline resulted in a 16% excess risk.
Data Source: An analysis of nearly 23,000 participants in the prospective, longitudinal Health and Retirement Study, 15.5% of whom developed diabetes during an average 8.7 years of follow-up.
Disclosures: The Health and Retirement Study is sponsored by the National Institute on Aging. This analysis was funded by the CDC. The presenter reported having no financial conflicts.
Pilot study: Vitamin D promising for diabetic pain
CHICAGO – High-dose vitamin D2 supplementation shows promise for the treatment of chronic pain among women with type 2 diabetes and comorbid depression, according to a single-arm pilot study known as the Sunshine Study.
The vitamin D2 supplementation at 50,000 IU/week for 6 months also was associated with significant improvement in major depressive disorder in this uncontrolled study, Sue M. Penckofer, Ph.D., reported at the annual scientific sessions of the American Diabetes Association.
This was a small, hypothesis-generating study of a relatively low-cost and safe potential therapy for a common clinical condition. The encouraging results have led to a $1.5 million grant from the National Institutes of Health for a considerably larger 4-year randomized controlled trial due to begin later this year, said Dr. Penckofer, professor of nursing at Loyola University in Maywood, Ill.
The pilot study included 46 women with type 2 diabetes, major depressive disorder, and vitamin D insufficiency as evidenced by their mean baseline serum level of 18 ng/mL. They averaged 54.6 years of age, with a 7.8-year history of diabetes and a mean hemoglobin A1c of 6.8%. Twenty-eight of the women had neuropathic pain involving their legs and 34 had sensory pain, with numbness or tingling in their feet and/or legs.
After 6 months of weekly vitamin D supplementation, the subjects’ mean vitamin D blood level had climbed to 38 ng/mL. Their median score on the Center for Epidemiologic Studies Depression Scale improved from 26.8 at baseline to 12.2 and their depression rating on the Patient Health Questionnaire (PHQ-9) improved from 11.5 to 5.2, although their antidepressant medication regimens were not required to be held constant.
Scores on the neuropathic pain subscale of the Diabetes Symptom Checklist improved from a baseline of 3.2 to 1.8 at 3 months and 2.1 at 6 months. Scores on the sensory pain subscale improved from 7.3 at baseline to 5.0 at 3 months and 5.9 at 6 months. These favorable trends didn’t attain statistical significance in such a small study population, but patients in the top half of the group in terms of neuropathic or sensory pain did show statistically significant improvement over time. Those in the high baseline neuropathic pain group went from an average score of 5.2 at enrollment to 2.5 at 3 months and 2.8 at 6 months. Similarly, the high sensory pain subgroup improved from a baseline score of 10.6 to 6.8 at 3 months and 6.9 at 6 months, according to Dr. Penckofer.
The Sunshine Study was funded by the National Institute of Diabetes and Digestive and Kidney Diseases. Dr. Penckofer reported having no conflicts of interest.
CHICAGO – High-dose vitamin D2 supplementation shows promise for the treatment of chronic pain among women with type 2 diabetes and comorbid depression, according to a single-arm pilot study known as the Sunshine Study.
The vitamin D2 supplementation at 50,000 IU/week for 6 months also was associated with significant improvement in major depressive disorder in this uncontrolled study, Sue M. Penckofer, Ph.D., reported at the annual scientific sessions of the American Diabetes Association.
This was a small, hypothesis-generating study of a relatively low-cost and safe potential therapy for a common clinical condition. The encouraging results have led to a $1.5 million grant from the National Institutes of Health for a considerably larger 4-year randomized controlled trial due to begin later this year, said Dr. Penckofer, professor of nursing at Loyola University in Maywood, Ill.
The pilot study included 46 women with type 2 diabetes, major depressive disorder, and vitamin D insufficiency as evidenced by their mean baseline serum level of 18 ng/mL. They averaged 54.6 years of age, with a 7.8-year history of diabetes and a mean hemoglobin A1c of 6.8%. Twenty-eight of the women had neuropathic pain involving their legs and 34 had sensory pain, with numbness or tingling in their feet and/or legs.
After 6 months of weekly vitamin D supplementation, the subjects’ mean vitamin D blood level had climbed to 38 ng/mL. Their median score on the Center for Epidemiologic Studies Depression Scale improved from 26.8 at baseline to 12.2 and their depression rating on the Patient Health Questionnaire (PHQ-9) improved from 11.5 to 5.2, although their antidepressant medication regimens were not required to be held constant.
Scores on the neuropathic pain subscale of the Diabetes Symptom Checklist improved from a baseline of 3.2 to 1.8 at 3 months and 2.1 at 6 months. Scores on the sensory pain subscale improved from 7.3 at baseline to 5.0 at 3 months and 5.9 at 6 months. These favorable trends didn’t attain statistical significance in such a small study population, but patients in the top half of the group in terms of neuropathic or sensory pain did show statistically significant improvement over time. Those in the high baseline neuropathic pain group went from an average score of 5.2 at enrollment to 2.5 at 3 months and 2.8 at 6 months. Similarly, the high sensory pain subgroup improved from a baseline score of 10.6 to 6.8 at 3 months and 6.9 at 6 months, according to Dr. Penckofer.
The Sunshine Study was funded by the National Institute of Diabetes and Digestive and Kidney Diseases. Dr. Penckofer reported having no conflicts of interest.
CHICAGO – High-dose vitamin D2 supplementation shows promise for the treatment of chronic pain among women with type 2 diabetes and comorbid depression, according to a single-arm pilot study known as the Sunshine Study.
The vitamin D2 supplementation at 50,000 IU/week for 6 months also was associated with significant improvement in major depressive disorder in this uncontrolled study, Sue M. Penckofer, Ph.D., reported at the annual scientific sessions of the American Diabetes Association.
This was a small, hypothesis-generating study of a relatively low-cost and safe potential therapy for a common clinical condition. The encouraging results have led to a $1.5 million grant from the National Institutes of Health for a considerably larger 4-year randomized controlled trial due to begin later this year, said Dr. Penckofer, professor of nursing at Loyola University in Maywood, Ill.
The pilot study included 46 women with type 2 diabetes, major depressive disorder, and vitamin D insufficiency as evidenced by their mean baseline serum level of 18 ng/mL. They averaged 54.6 years of age, with a 7.8-year history of diabetes and a mean hemoglobin A1c of 6.8%. Twenty-eight of the women had neuropathic pain involving their legs and 34 had sensory pain, with numbness or tingling in their feet and/or legs.
After 6 months of weekly vitamin D supplementation, the subjects’ mean vitamin D blood level had climbed to 38 ng/mL. Their median score on the Center for Epidemiologic Studies Depression Scale improved from 26.8 at baseline to 12.2 and their depression rating on the Patient Health Questionnaire (PHQ-9) improved from 11.5 to 5.2, although their antidepressant medication regimens were not required to be held constant.
Scores on the neuropathic pain subscale of the Diabetes Symptom Checklist improved from a baseline of 3.2 to 1.8 at 3 months and 2.1 at 6 months. Scores on the sensory pain subscale improved from 7.3 at baseline to 5.0 at 3 months and 5.9 at 6 months. These favorable trends didn’t attain statistical significance in such a small study population, but patients in the top half of the group in terms of neuropathic or sensory pain did show statistically significant improvement over time. Those in the high baseline neuropathic pain group went from an average score of 5.2 at enrollment to 2.5 at 3 months and 2.8 at 6 months. Similarly, the high sensory pain subgroup improved from a baseline score of 10.6 to 6.8 at 3 months and 6.9 at 6 months, according to Dr. Penckofer.
The Sunshine Study was funded by the National Institute of Diabetes and Digestive and Kidney Diseases. Dr. Penckofer reported having no conflicts of interest.
AT THE ADA ANNUAL SCIENTIFIC SESSIONS
Major finding: Type 2 diabetes patients with comorbid depression and baseline elevated neuropathic and/or sensory pain symptoms showed significant improvement in pain scores after 6 months of high-dose vitamin D2 supplementation.
Data source: The Sunshine Study was a prospective, uncontrolled study in which 46 women with type 2 diabetes, comorbid major depression and chronic pain, and vitamin D insufficiency received 50,000 IU/week of vitamin D2 for 6 months.
Disclosures: The study was funded by the National Institute of Diabetes and Digestive and Kidney Diseases. Dr. Penckofer reported having no conflicts of interest.
Children of diabetic mothers at greater risk of overweight as young adults
CHICAGO – At age 17, the children of mothers with diabetes during pregnancy are more likely to be overweight, according to an Israeli longitudinal study.
This was equally the case regardless of whether the mother had gestational or pregestational diabetes, Dr. Zvi Laron reported at the annual scientific sessions of the American Diabetes Association.
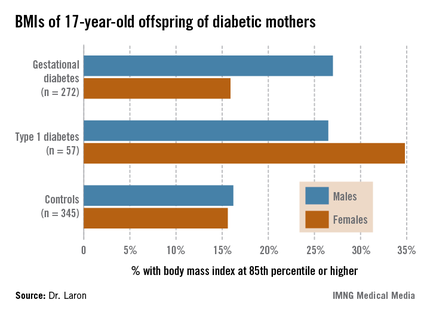
The Israeli data expand upon the earlier findings of EPOCH (Exploring Perinatal Outcomes among Children), a retrospective cohort study conducted by investigators at the University of Colorado, Denver. The Colorado investigators followed 95 children exposed to diabetes in utero – 87 whose mothers had gestational diabetes and 8 with type 1 diabetes – and 409 controls out to age 13 years, and determined that the children of diabetic mothers were more obese. Of note, the investigators found no significant differences between the children of diabetic mothers and controls in growth trajectories during infancy and early childhood, but a higher body mass index growth velocity among the offspring of diabetic mothers at ages 10-13 years (J. Pediatr. 2011;158:941-6).
The Israeli study tracked a much larger group of children of diabetic mothers later into adolescence, taking advantage of the fact that most Israeli youth have to report for compulsory military service at age 17.
Dr. Laron of Tel Aviv University and his coworkers identified 447 singleton term neonates of mothers with gestational diabetes and 97 whose mothers had type 1 diabetes born during 1987-1993 at the Helen Schneider Hospital for Women in Petach Tikva, Israel. The control group was composed of an equal number of singleton term babies born to nondiabetic mothers at the hospital during the same years.
Consistent with other reports, the average birth weight and length of the babies of diabetic mothers were significantly greater than in controls. For example, 24.3% of the baby boys and 18.4% of the girls of mothers with gestational diabetes were at or above the 90th percentile for birth weight for the Israeli population, compared with 6.8% of male and 6.0% of female controls.
Sixty-two percent of the original study population presented for military service at age 17; Arabs, as well as ultra-Orthodox Jewish men and women, are exempt from military service. The offspring exposed in utero to maternal diabetes didn’t differ from controls in terms of height, but the males with in utero exposure were significantly more likely to have a BMI at or above the 85th percentile nationally, and the females showed a similar albeit nonsignificant trend (see chart). Unlike in the Colorado study, however, the Israeli investigators didn’t have access to serial measurements that would have enabled them to determine the age at which the accelerated BMI growth trajectory occurred.
It is well established that adolescent overweight is associated with increased risk of subsequent obesity in adulthood. If the Colorado investigators are correct in their conclusion that exposure to diabetes in utero results in accelerated BMI growth occurring in late childhood, it would suggest that early childhood provides a window of opportunity for obesity prevention in these children. But a better understanding of the underlying mechanisms involved in the association between in utero exposure to diabetes and accelerated BMI growth trajectory occurring 10-13 years later may be needed before targeted preventive interventions can be developed and tested.
Dr. Laron reported having no conflicts of interest.
CHICAGO – At age 17, the children of mothers with diabetes during pregnancy are more likely to be overweight, according to an Israeli longitudinal study.
This was equally the case regardless of whether the mother had gestational or pregestational diabetes, Dr. Zvi Laron reported at the annual scientific sessions of the American Diabetes Association.

The Israeli data expand upon the earlier findings of EPOCH (Exploring Perinatal Outcomes among Children), a retrospective cohort study conducted by investigators at the University of Colorado, Denver. The Colorado investigators followed 95 children exposed to diabetes in utero – 87 whose mothers had gestational diabetes and 8 with type 1 diabetes – and 409 controls out to age 13 years, and determined that the children of diabetic mothers were more obese. Of note, the investigators found no significant differences between the children of diabetic mothers and controls in growth trajectories during infancy and early childhood, but a higher body mass index growth velocity among the offspring of diabetic mothers at ages 10-13 years (J. Pediatr. 2011;158:941-6).
The Israeli study tracked a much larger group of children of diabetic mothers later into adolescence, taking advantage of the fact that most Israeli youth have to report for compulsory military service at age 17.
Dr. Laron of Tel Aviv University and his coworkers identified 447 singleton term neonates of mothers with gestational diabetes and 97 whose mothers had type 1 diabetes born during 1987-1993 at the Helen Schneider Hospital for Women in Petach Tikva, Israel. The control group was composed of an equal number of singleton term babies born to nondiabetic mothers at the hospital during the same years.
Consistent with other reports, the average birth weight and length of the babies of diabetic mothers were significantly greater than in controls. For example, 24.3% of the baby boys and 18.4% of the girls of mothers with gestational diabetes were at or above the 90th percentile for birth weight for the Israeli population, compared with 6.8% of male and 6.0% of female controls.
Sixty-two percent of the original study population presented for military service at age 17; Arabs, as well as ultra-Orthodox Jewish men and women, are exempt from military service. The offspring exposed in utero to maternal diabetes didn’t differ from controls in terms of height, but the males with in utero exposure were significantly more likely to have a BMI at or above the 85th percentile nationally, and the females showed a similar albeit nonsignificant trend (see chart). Unlike in the Colorado study, however, the Israeli investigators didn’t have access to serial measurements that would have enabled them to determine the age at which the accelerated BMI growth trajectory occurred.
It is well established that adolescent overweight is associated with increased risk of subsequent obesity in adulthood. If the Colorado investigators are correct in their conclusion that exposure to diabetes in utero results in accelerated BMI growth occurring in late childhood, it would suggest that early childhood provides a window of opportunity for obesity prevention in these children. But a better understanding of the underlying mechanisms involved in the association between in utero exposure to diabetes and accelerated BMI growth trajectory occurring 10-13 years later may be needed before targeted preventive interventions can be developed and tested.
Dr. Laron reported having no conflicts of interest.
CHICAGO – At age 17, the children of mothers with diabetes during pregnancy are more likely to be overweight, according to an Israeli longitudinal study.
This was equally the case regardless of whether the mother had gestational or pregestational diabetes, Dr. Zvi Laron reported at the annual scientific sessions of the American Diabetes Association.

The Israeli data expand upon the earlier findings of EPOCH (Exploring Perinatal Outcomes among Children), a retrospective cohort study conducted by investigators at the University of Colorado, Denver. The Colorado investigators followed 95 children exposed to diabetes in utero – 87 whose mothers had gestational diabetes and 8 with type 1 diabetes – and 409 controls out to age 13 years, and determined that the children of diabetic mothers were more obese. Of note, the investigators found no significant differences between the children of diabetic mothers and controls in growth trajectories during infancy and early childhood, but a higher body mass index growth velocity among the offspring of diabetic mothers at ages 10-13 years (J. Pediatr. 2011;158:941-6).
The Israeli study tracked a much larger group of children of diabetic mothers later into adolescence, taking advantage of the fact that most Israeli youth have to report for compulsory military service at age 17.
Dr. Laron of Tel Aviv University and his coworkers identified 447 singleton term neonates of mothers with gestational diabetes and 97 whose mothers had type 1 diabetes born during 1987-1993 at the Helen Schneider Hospital for Women in Petach Tikva, Israel. The control group was composed of an equal number of singleton term babies born to nondiabetic mothers at the hospital during the same years.
Consistent with other reports, the average birth weight and length of the babies of diabetic mothers were significantly greater than in controls. For example, 24.3% of the baby boys and 18.4% of the girls of mothers with gestational diabetes were at or above the 90th percentile for birth weight for the Israeli population, compared with 6.8% of male and 6.0% of female controls.
Sixty-two percent of the original study population presented for military service at age 17; Arabs, as well as ultra-Orthodox Jewish men and women, are exempt from military service. The offspring exposed in utero to maternal diabetes didn’t differ from controls in terms of height, but the males with in utero exposure were significantly more likely to have a BMI at or above the 85th percentile nationally, and the females showed a similar albeit nonsignificant trend (see chart). Unlike in the Colorado study, however, the Israeli investigators didn’t have access to serial measurements that would have enabled them to determine the age at which the accelerated BMI growth trajectory occurred.
It is well established that adolescent overweight is associated with increased risk of subsequent obesity in adulthood. If the Colorado investigators are correct in their conclusion that exposure to diabetes in utero results in accelerated BMI growth occurring in late childhood, it would suggest that early childhood provides a window of opportunity for obesity prevention in these children. But a better understanding of the underlying mechanisms involved in the association between in utero exposure to diabetes and accelerated BMI growth trajectory occurring 10-13 years later may be needed before targeted preventive interventions can be developed and tested.
Dr. Laron reported having no conflicts of interest.
AT THE ADA ANNUAL SCIENTIFIC SESSIONS
Major finding: Of 17-year-old males born to diabetic mothers, 27% had a BMI at or above the 85th percentile nationally, compared with 16% of controls.
Data source: A retrospective study of 329 Israeli 17-year-olds born to diabetic mothers and 345 controls.
Disclosures: Dr. Laron reported having no conflicts of interest.
Would patients open wallets for fewer diabetes drug doses?
CHICAGO – Conventional wisdom holds that, given the option, patients prefer the convenience of less-frequent dosing of oral drugs, but just how much more money are patients with type 2 diabetes willing to pay out of pocket to change from daily to once-weekly dosing of their oral antidiabetic medications?
Not so much, apparently. Specifically, they’d be prepared to fork over amounts ranging from no more than an extra $3.64 per month to change from one pill twice daily to one pill once a day, up to $13.88 to switch from two pills once per day to one pill once a week, according to a new survey.
To reduce their pill burden from one pill per day to one per week, patients with type 2 diabetes would be willing to spend an extra $5.86 out of pocket per month, A. Brett Hauber, Ph.D., reported at the annual scientific sessions of the American Diabetes Association.
"Reducing the frequency of dosing has monetary value to patients. The value isn’t huge. It’s modest but real," declared Dr. Hauber, senior economist and vice president at RTI Health Solutions, a medical industry research and consulting firm based in Research Triangle Park, N.C.
RTI was hired by Merck to evaluate patient preferences for reducing the pill burden associated with oral antidiabetic agents relative to other patient priorities. To do so, Dr. Hauber and his coworkers employed a technique social scientists call a discrete-choice experiment.
They presented a large group of type 2 diabetes patients with a detailed survey involving a series of pairs of hypothetical oral antidiabetic drug profiles and had participants choose their preferred option for each pair. The profiles addressed reductions in average blood glucose, hypoglycemia risk, frequency of mild-to-moderate GI side effects, out-of-pocket cost, dosing frequency, weight change, and incremental risk of chronic heart failure.
Each of these attributes generated a relative importance score that enabled researchers to see what was most important to patients with type 2 diabetes. For example, the data indicated that patients considered a 13% reduction in the risk of GI side effects to be about as important as an absolute 2% reduction in the risk of developing heart failure, from 3% to 1%, Dr. Hauber explained.
The study population consisted of 923 patients with type 2 diabetes who were not on injectable therapy. A total of 197 were treatment-naive, while the rest were taking a single oral antidiabetic drug. Participants were recruited by Knowledge Networks, an online research company with access to a nationally representative sample of U.S. households. Knowledge Networks knows its recruits very, very well: The company tracks 4,500 variables on each of them.
The two strongest drivers of patient choice regarding oral antidiabetic therapy to emerge from the study were out-of-pocket cost and glucose control. Cost was hands down the most important factor. Assigning it a standardized relative importance rate of 10, glucose control came in at 5.3. This was followed by hypoglycemia at 3.1, GI side effects at 2.6, weight change at 2.5, heart failure risk at 2.0, and, lastly, daily dosing schedule at 0.8.
"What’s most interesting is every one of those relative importance scores is statistically significantly different from zero. What that means is every one of these attributes was important, at least to some extent, to patients. Some were more important than others, but everything we assessed influenced their choices," Dr. Hauber said.
Sixty-seven percent of patients preferred weekly drug dosing, but 33% actually favored daily dosing. Younger patients – those under age 45 years – particularly preferred weekly dosing over daily, by a margin of 78% to 22%. Patients not currently on treatment also expressed a stronger than average preference for weekly over daily dosing, by a margin of 75% to 25%.
"It is likely that, if we can offer more convenient, once-weekly dosing to younger patients and treatment-naive patients, it may be that they would be more likely to initiate therapy and potentially to adhere to therapy. But this is just my speculation based on the results that we have. We haven’t actually proven that here," Dr. Hauber observed.
The study was sponsored by Merck. The presenter is an executive at RTI Health Solutions, the research firm hired to conduct the investigation.
CHICAGO – Conventional wisdom holds that, given the option, patients prefer the convenience of less-frequent dosing of oral drugs, but just how much more money are patients with type 2 diabetes willing to pay out of pocket to change from daily to once-weekly dosing of their oral antidiabetic medications?
Not so much, apparently. Specifically, they’d be prepared to fork over amounts ranging from no more than an extra $3.64 per month to change from one pill twice daily to one pill once a day, up to $13.88 to switch from two pills once per day to one pill once a week, according to a new survey.
To reduce their pill burden from one pill per day to one per week, patients with type 2 diabetes would be willing to spend an extra $5.86 out of pocket per month, A. Brett Hauber, Ph.D., reported at the annual scientific sessions of the American Diabetes Association.
"Reducing the frequency of dosing has monetary value to patients. The value isn’t huge. It’s modest but real," declared Dr. Hauber, senior economist and vice president at RTI Health Solutions, a medical industry research and consulting firm based in Research Triangle Park, N.C.
RTI was hired by Merck to evaluate patient preferences for reducing the pill burden associated with oral antidiabetic agents relative to other patient priorities. To do so, Dr. Hauber and his coworkers employed a technique social scientists call a discrete-choice experiment.
They presented a large group of type 2 diabetes patients with a detailed survey involving a series of pairs of hypothetical oral antidiabetic drug profiles and had participants choose their preferred option for each pair. The profiles addressed reductions in average blood glucose, hypoglycemia risk, frequency of mild-to-moderate GI side effects, out-of-pocket cost, dosing frequency, weight change, and incremental risk of chronic heart failure.
Each of these attributes generated a relative importance score that enabled researchers to see what was most important to patients with type 2 diabetes. For example, the data indicated that patients considered a 13% reduction in the risk of GI side effects to be about as important as an absolute 2% reduction in the risk of developing heart failure, from 3% to 1%, Dr. Hauber explained.
The study population consisted of 923 patients with type 2 diabetes who were not on injectable therapy. A total of 197 were treatment-naive, while the rest were taking a single oral antidiabetic drug. Participants were recruited by Knowledge Networks, an online research company with access to a nationally representative sample of U.S. households. Knowledge Networks knows its recruits very, very well: The company tracks 4,500 variables on each of them.
The two strongest drivers of patient choice regarding oral antidiabetic therapy to emerge from the study were out-of-pocket cost and glucose control. Cost was hands down the most important factor. Assigning it a standardized relative importance rate of 10, glucose control came in at 5.3. This was followed by hypoglycemia at 3.1, GI side effects at 2.6, weight change at 2.5, heart failure risk at 2.0, and, lastly, daily dosing schedule at 0.8.
"What’s most interesting is every one of those relative importance scores is statistically significantly different from zero. What that means is every one of these attributes was important, at least to some extent, to patients. Some were more important than others, but everything we assessed influenced their choices," Dr. Hauber said.
Sixty-seven percent of patients preferred weekly drug dosing, but 33% actually favored daily dosing. Younger patients – those under age 45 years – particularly preferred weekly dosing over daily, by a margin of 78% to 22%. Patients not currently on treatment also expressed a stronger than average preference for weekly over daily dosing, by a margin of 75% to 25%.
"It is likely that, if we can offer more convenient, once-weekly dosing to younger patients and treatment-naive patients, it may be that they would be more likely to initiate therapy and potentially to adhere to therapy. But this is just my speculation based on the results that we have. We haven’t actually proven that here," Dr. Hauber observed.
The study was sponsored by Merck. The presenter is an executive at RTI Health Solutions, the research firm hired to conduct the investigation.
CHICAGO – Conventional wisdom holds that, given the option, patients prefer the convenience of less-frequent dosing of oral drugs, but just how much more money are patients with type 2 diabetes willing to pay out of pocket to change from daily to once-weekly dosing of their oral antidiabetic medications?
Not so much, apparently. Specifically, they’d be prepared to fork over amounts ranging from no more than an extra $3.64 per month to change from one pill twice daily to one pill once a day, up to $13.88 to switch from two pills once per day to one pill once a week, according to a new survey.
To reduce their pill burden from one pill per day to one per week, patients with type 2 diabetes would be willing to spend an extra $5.86 out of pocket per month, A. Brett Hauber, Ph.D., reported at the annual scientific sessions of the American Diabetes Association.
"Reducing the frequency of dosing has monetary value to patients. The value isn’t huge. It’s modest but real," declared Dr. Hauber, senior economist and vice president at RTI Health Solutions, a medical industry research and consulting firm based in Research Triangle Park, N.C.
RTI was hired by Merck to evaluate patient preferences for reducing the pill burden associated with oral antidiabetic agents relative to other patient priorities. To do so, Dr. Hauber and his coworkers employed a technique social scientists call a discrete-choice experiment.
They presented a large group of type 2 diabetes patients with a detailed survey involving a series of pairs of hypothetical oral antidiabetic drug profiles and had participants choose their preferred option for each pair. The profiles addressed reductions in average blood glucose, hypoglycemia risk, frequency of mild-to-moderate GI side effects, out-of-pocket cost, dosing frequency, weight change, and incremental risk of chronic heart failure.
Each of these attributes generated a relative importance score that enabled researchers to see what was most important to patients with type 2 diabetes. For example, the data indicated that patients considered a 13% reduction in the risk of GI side effects to be about as important as an absolute 2% reduction in the risk of developing heart failure, from 3% to 1%, Dr. Hauber explained.
The study population consisted of 923 patients with type 2 diabetes who were not on injectable therapy. A total of 197 were treatment-naive, while the rest were taking a single oral antidiabetic drug. Participants were recruited by Knowledge Networks, an online research company with access to a nationally representative sample of U.S. households. Knowledge Networks knows its recruits very, very well: The company tracks 4,500 variables on each of them.
The two strongest drivers of patient choice regarding oral antidiabetic therapy to emerge from the study were out-of-pocket cost and glucose control. Cost was hands down the most important factor. Assigning it a standardized relative importance rate of 10, glucose control came in at 5.3. This was followed by hypoglycemia at 3.1, GI side effects at 2.6, weight change at 2.5, heart failure risk at 2.0, and, lastly, daily dosing schedule at 0.8.
"What’s most interesting is every one of those relative importance scores is statistically significantly different from zero. What that means is every one of these attributes was important, at least to some extent, to patients. Some were more important than others, but everything we assessed influenced their choices," Dr. Hauber said.
Sixty-seven percent of patients preferred weekly drug dosing, but 33% actually favored daily dosing. Younger patients – those under age 45 years – particularly preferred weekly dosing over daily, by a margin of 78% to 22%. Patients not currently on treatment also expressed a stronger than average preference for weekly over daily dosing, by a margin of 75% to 25%.
"It is likely that, if we can offer more convenient, once-weekly dosing to younger patients and treatment-naive patients, it may be that they would be more likely to initiate therapy and potentially to adhere to therapy. But this is just my speculation based on the results that we have. We haven’t actually proven that here," Dr. Hauber observed.
The study was sponsored by Merck. The presenter is an executive at RTI Health Solutions, the research firm hired to conduct the investigation.
AT THE ADA ANNUAL SCIENTIFIC SESSIONS
Major Finding: Patients with type 2 diabetes would be willing to spend an extra $5.86 out of pocket per month on their oral antidiabetic medication to change from one pill per day to one per week.
Data Source: An economic discrete-choice experiment involving a detailed survey completed by 923 patients with type 2 diabetes who were not on injectable therapy.
Disclosures: The study was sponsored by Merck. The presenter is an executive at RTI Health Solutions, the research firm hired to conduct the investigation.