User login
Bariatric surgery may prevent heart failure
AMSTERDAM – New findings from the landmark Swedish Obese Subjects study "strongly suggest" that bariatric surgery reduces by roughly half the long-term risk of developing heart failure, Dr. Kristjan Karason reported at the annual congress of the European Society of Cardiology.
"This is the first study to look at the effect of bariatric surgery on heart failure risk," observed Dr. Karason of the University of Gothenburg (Sweden). "Our findings support a modulating role of excess body fat in the pathogenesis of heart failure."
The SOS (Swedish Obese Subjects) study has been a major driver of the growing enthusiasm for bariatric surgery, not only as a means of achieving sustained weight loss far beyond what can typically be accomplished medically, but also as a means of reducing obese patients’ elevated risks for a variety of serious chronic comorbid diseases.
For example, the SOS investigators have previously reported that bariatric surgery reduced the long-term risk of developing type 2 diabetes by 87% compared to matched obese controls who didn’t undergo weight-loss surgery (N. Engl. J. Med. 2012;367:695-704), that it reduced the risk of fatal or nonfatal acute MI or stroke by one-third during a median 14.7 years of follow-up (JAMA 2012;307:56-65), and it resulted in a 42% decrease in cancer incidence in women (Lancet Oncol. 2009;10:653-62).
The SOS is a nonrandomized, prospective, observational study involving 2,010 obese subjects who underwent bariatric surgery in 1987-2001, when they were 37-60 years old. A total of 68% of the bariatric surgery recipients had vertical band gastroplasty, 19% underwent gastric banding, and 13% had a Roux en-Y gastric bypass. They were extensively matched by 18 variables to 2,037 obese controls. The SOS study is being conducted at 25 surgical departments and 480 primary care clinics across Sweden. Follow-up is ongoing.
It has been known for more than a decade that increased body mass index is associated with greater risk of developing heart failure. The mechanism involved is not well defined but is probably multifactorial. Obesity imposes a greater hemodynamic load on the heart, both preload and afterload, with resultant left ventricular hypertrophy and diastolic dysfunction. Obesity is also associated with higher levels of cardiovascular risk factors and an increased risk of atrial fibrillation, Dr. Karason noted.
Mean weight loss after a median of 14.7 years of prospective follow-up in the SOS study was 18% in the bariatric surgery group and 1% in controls. During follow-up, 91 bariatric surgery patients and 152 controls were diagnosed with heart failure, for an incidence rate of 3.1 as compared with 5.2 cases per 1,000 person-years in the control subjects.
This translated to a 48% relative risk reduction in new-onset heart failure in a multivariate regression analysis adjusted for age, sex, baseline body mass index, waist circumference, blood glucose, lipids, prior cardiovascular disease, and smoking status.
One audience member asked how to interpret the new SOS findings in light of the heart failure obesity paradox, which is the observation in multiple studies that overweight and obese heart failure patients tends to fare better than leaner ones.
"There have been no studies of weight loss in obese patients with heart failure. There really should be," Dr. Karason replied. "But my feeling is that they would reduce their risk because they will improve several risk factors, and their hemodynamic situation is also improved. So my recommendation would be for those heart failure patients to lose weight. I don’t have any studies to support that."
The SOS study is funded by the Swedish Research Council. Dr. Karason reported having no germane financial interests.
AMSTERDAM – New findings from the landmark Swedish Obese Subjects study "strongly suggest" that bariatric surgery reduces by roughly half the long-term risk of developing heart failure, Dr. Kristjan Karason reported at the annual congress of the European Society of Cardiology.
"This is the first study to look at the effect of bariatric surgery on heart failure risk," observed Dr. Karason of the University of Gothenburg (Sweden). "Our findings support a modulating role of excess body fat in the pathogenesis of heart failure."
The SOS (Swedish Obese Subjects) study has been a major driver of the growing enthusiasm for bariatric surgery, not only as a means of achieving sustained weight loss far beyond what can typically be accomplished medically, but also as a means of reducing obese patients’ elevated risks for a variety of serious chronic comorbid diseases.
For example, the SOS investigators have previously reported that bariatric surgery reduced the long-term risk of developing type 2 diabetes by 87% compared to matched obese controls who didn’t undergo weight-loss surgery (N. Engl. J. Med. 2012;367:695-704), that it reduced the risk of fatal or nonfatal acute MI or stroke by one-third during a median 14.7 years of follow-up (JAMA 2012;307:56-65), and it resulted in a 42% decrease in cancer incidence in women (Lancet Oncol. 2009;10:653-62).
The SOS is a nonrandomized, prospective, observational study involving 2,010 obese subjects who underwent bariatric surgery in 1987-2001, when they were 37-60 years old. A total of 68% of the bariatric surgery recipients had vertical band gastroplasty, 19% underwent gastric banding, and 13% had a Roux en-Y gastric bypass. They were extensively matched by 18 variables to 2,037 obese controls. The SOS study is being conducted at 25 surgical departments and 480 primary care clinics across Sweden. Follow-up is ongoing.
It has been known for more than a decade that increased body mass index is associated with greater risk of developing heart failure. The mechanism involved is not well defined but is probably multifactorial. Obesity imposes a greater hemodynamic load on the heart, both preload and afterload, with resultant left ventricular hypertrophy and diastolic dysfunction. Obesity is also associated with higher levels of cardiovascular risk factors and an increased risk of atrial fibrillation, Dr. Karason noted.
Mean weight loss after a median of 14.7 years of prospective follow-up in the SOS study was 18% in the bariatric surgery group and 1% in controls. During follow-up, 91 bariatric surgery patients and 152 controls were diagnosed with heart failure, for an incidence rate of 3.1 as compared with 5.2 cases per 1,000 person-years in the control subjects.
This translated to a 48% relative risk reduction in new-onset heart failure in a multivariate regression analysis adjusted for age, sex, baseline body mass index, waist circumference, blood glucose, lipids, prior cardiovascular disease, and smoking status.
One audience member asked how to interpret the new SOS findings in light of the heart failure obesity paradox, which is the observation in multiple studies that overweight and obese heart failure patients tends to fare better than leaner ones.
"There have been no studies of weight loss in obese patients with heart failure. There really should be," Dr. Karason replied. "But my feeling is that they would reduce their risk because they will improve several risk factors, and their hemodynamic situation is also improved. So my recommendation would be for those heart failure patients to lose weight. I don’t have any studies to support that."
The SOS study is funded by the Swedish Research Council. Dr. Karason reported having no germane financial interests.
AMSTERDAM – New findings from the landmark Swedish Obese Subjects study "strongly suggest" that bariatric surgery reduces by roughly half the long-term risk of developing heart failure, Dr. Kristjan Karason reported at the annual congress of the European Society of Cardiology.
"This is the first study to look at the effect of bariatric surgery on heart failure risk," observed Dr. Karason of the University of Gothenburg (Sweden). "Our findings support a modulating role of excess body fat in the pathogenesis of heart failure."
The SOS (Swedish Obese Subjects) study has been a major driver of the growing enthusiasm for bariatric surgery, not only as a means of achieving sustained weight loss far beyond what can typically be accomplished medically, but also as a means of reducing obese patients’ elevated risks for a variety of serious chronic comorbid diseases.
For example, the SOS investigators have previously reported that bariatric surgery reduced the long-term risk of developing type 2 diabetes by 87% compared to matched obese controls who didn’t undergo weight-loss surgery (N. Engl. J. Med. 2012;367:695-704), that it reduced the risk of fatal or nonfatal acute MI or stroke by one-third during a median 14.7 years of follow-up (JAMA 2012;307:56-65), and it resulted in a 42% decrease in cancer incidence in women (Lancet Oncol. 2009;10:653-62).
The SOS is a nonrandomized, prospective, observational study involving 2,010 obese subjects who underwent bariatric surgery in 1987-2001, when they were 37-60 years old. A total of 68% of the bariatric surgery recipients had vertical band gastroplasty, 19% underwent gastric banding, and 13% had a Roux en-Y gastric bypass. They were extensively matched by 18 variables to 2,037 obese controls. The SOS study is being conducted at 25 surgical departments and 480 primary care clinics across Sweden. Follow-up is ongoing.
It has been known for more than a decade that increased body mass index is associated with greater risk of developing heart failure. The mechanism involved is not well defined but is probably multifactorial. Obesity imposes a greater hemodynamic load on the heart, both preload and afterload, with resultant left ventricular hypertrophy and diastolic dysfunction. Obesity is also associated with higher levels of cardiovascular risk factors and an increased risk of atrial fibrillation, Dr. Karason noted.
Mean weight loss after a median of 14.7 years of prospective follow-up in the SOS study was 18% in the bariatric surgery group and 1% in controls. During follow-up, 91 bariatric surgery patients and 152 controls were diagnosed with heart failure, for an incidence rate of 3.1 as compared with 5.2 cases per 1,000 person-years in the control subjects.
This translated to a 48% relative risk reduction in new-onset heart failure in a multivariate regression analysis adjusted for age, sex, baseline body mass index, waist circumference, blood glucose, lipids, prior cardiovascular disease, and smoking status.
One audience member asked how to interpret the new SOS findings in light of the heart failure obesity paradox, which is the observation in multiple studies that overweight and obese heart failure patients tends to fare better than leaner ones.
"There have been no studies of weight loss in obese patients with heart failure. There really should be," Dr. Karason replied. "But my feeling is that they would reduce their risk because they will improve several risk factors, and their hemodynamic situation is also improved. So my recommendation would be for those heart failure patients to lose weight. I don’t have any studies to support that."
The SOS study is funded by the Swedish Research Council. Dr. Karason reported having no germane financial interests.
AT THE ESC CONGRESS 2013
Major finding: The incidence rate of heart failure during a median 15 years of prospective follow-up after bariatric surgery was 3.1 cases per 1,000 person-years, compared with 5.2/1,000 person-years in obese controls.
Data source: The Swedish Obese Subjects study included 2,010 obese subjects who underwent bariatric surgery in 1987-2001 and 2,037 closely matched obese controls. It is a nonrandomized, prospective, observational study.
Disclosures: The study is funded by the Swedish Research Council. The presenter reported having no financial conflicts.
Fixed maintenance secukinumab beats as-needed dosing
ISTANBUL – Psoriasis patients who initially achieve a high-level response to secukinumab are significantly more likely to retain it over the long haul if they follow a fixed maintenance therapy schedule of once-monthly subcutaneous dosing rather than dosing as needed in the event of the start of relapse, according to the phase III SCULPTURE trial.
Secukinumab is an investigational fully human IgG1 monoclonal antibody targeting interleukin-17A, a key player in psoriasis, because it generates downstream proinflammatory cytokines and stimulates keratinocyte growth. Earlier studies indicated the investigational biologic had unprecedented clinical efficacy and rapidity of response. These observations gave rise to the hypothesis in SCULPTURE that maintenance dosing as needed upon relapse would prove noninferior to conventional fixed once-monthly maintenance therapy.
If the study hypothesis proved valid, it would mean less exposure to potential medication side effects, lower drug costs, and greater patient convenience. However, the hypothesis wasn’t borne out in the 966-patient, double-blind, randomized SCULPTURE (Study Comparing Retreatment Upon Start of Relapse), Dr. Ulrich Mrowietz reported at the annual congress of the European Academy of Dermatology and Venereology.
SCULPTURE participants had moderate-to-severe chronic plaque psoriasis despite prior systemic therapies, including biologic agents in many cases. They were randomized double blind to induction therapy involving five once-weekly subcutaneous injections of secukinumab at either 150 or 300 mg. At week 8, 843 participants with a Psoriasis Area and Severity Index (PASI) 75 response were re-randomized to maintenance therapy at the same dose, to be delivered either once monthly or as needed for relapse. The definition of relapse in this study required two elements: loss of PASI 75 response, and at least a 20% fall from the maximum PASI improvement, compared with baseline.
At week 52, 78.2% of patients randomized to 300 mg of secukinumab on a fixed once-monthly schedule still maintained a PASI 75 response. This was a significantly better outcome than the 67.7% PASI 75 rate in patients assigned to secukinumab 300 mg as needed, the 62.1% rate in those on secukinumab 150 mg once monthly, and the 52% PASI 75 rate in patients on secukinumab 150 mg as needed, reported Dr. Mrowietz of the University Medical Center Schleswig-Holstein, Kiel (Germany).
He found a bright spot in the negative results: While patients assigned to as-needed maintenance therapy achieved roughly an absolute 10% lower PASI 75 rate at 1 year, those in the secukinumab 150-mg group did so with only 46% the number of doses received by patients in the fixed monthly therapy group, while those in the retreatment-as-needed with secukinumab 300-mg group got only 39% of the number of doses, compared with patients on fixed monthly therapy.
"Fixed monthly dosing is the best maintenance regimen. But, in selected patients, there may be an opportunity with secukinumab to deviate from the usual fixed dosing regimen in favor of an as-needed approach," he said.
This tradeoff of an absolute 10% reduction in efficacy in return for a dosing regimen that entails less than half as much medication over the course of a year could prove of interest to payers, he noted.
The key, according to Dr. Mrowietz, will be to try to identify criteria helpful in selecting patients with an increased likelihood of a high-level response to the retreat-as-needed management strategy. The phase III trial was completed so recently that those necessary subanalyses have yet to be done.
However, Dr. Kristian Reich, another investigator involved in the secukinumab clinical trials program, drew a different message from SCULPTURE. He observed that these newer biologic agents are so effective that the bar has been raised with regard to patient expectations. Many patients won’t be satisfied with a PASI 75 response once a PASI 90 is achievable. And with fixed monthly maintenance secukinumab, it often is.
Indeed, the week-52 PASI 90 rate in SCULPTURE was a highly robust 59.7% in patients on fixed monthly secukinumab 300 mg and 45.8% for fixed monthly low-dose therapy, compared with the unimpressive 13.8% and 11.2% PASI 90 rates with high- and low-dose as-needed therapy.
"My take from this is that the best way to use this drug for continuous disease control is to give the drug every 4 weeks. What this study tells us is for those patients where you have to stop, where you have to use on-and-off therapy because they go away for 2 months to Africa, or they have a major operation, or for other reasons, these data are reassuring that we can use the drug safely on an intermittent basis," said Dr. Reich of Georg-August University in Göttingen, Germany.
Prior to SCULPTURE, he and other investigators were concerned that intermittent secukinumab therapy might promote the development of harmful antidrug antibodies. But antidrug antibodies arose in only three patients on fixed monthly maintenance therapy and two patients on as-needed therapy, and had no impact upon clinical efficacy or safety, he noted.
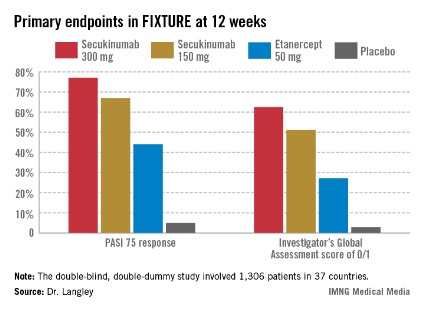
Another phase III trial presented in Istanbul was ERASURE (Efficacy and Safety of Subcutaneous Secukinumab for Moderate to Severe Chronic Plaque-Type Psoriasis for Up to 1 Year). Dr. Boni E. Elewski reported on 738 patients with moderate-to-severe chronic plaque psoriasis who were randomized double blind to secukinumab at 150 mg or 300 mg, or to placebo. Participants averaged a PASI score of 22 at baseline, along with 33% body surface area involvement. Psoriatic arthritis was present in 23% of subjects.
The co-primary endpoints in ERASURE were the week 12 PASI 75 response rates and Investigator’s Global Assessment scores of 0/1, indicative of clear or almost clear on a modified 5-point scale. The week-12 PASI 75 rates were 81.6% in the secukinumab 300 mg group, 71.6% with secukinumab 150 mg, and 4.5% with placebo. The week-12 IGA 0/1 rates were 65.3%, 51.2%, and 2.4%, said Dr. Elewski of the University of Alabama, Birmingham.
Among the other notable findings in ERASURE were the 69.8% PASI 90 and 41.6% PASI 100 responses at week 16 in the group assigned to secukinumab 300 mg, the fact that only one patient, on secukinumab 150 mg, developed transient antidrug antibodies, and the complete absence of cardiovascular events during 52 weeks on secukinumab, observed Dr. Elewski, professor of dermatology at the University of Alabama, Birmingham.
Dr. Mrowietz, Dr. Reich, and Dr. Elewski reported having received research grants from and serving as consultants to Novartis, which sponsored the secukinumab clinical trials program. The dermatologists serve in similar capacities with other pharmaceutical companies developing new medications for psoriasis.
ISTANBUL – Psoriasis patients who initially achieve a high-level response to secukinumab are significantly more likely to retain it over the long haul if they follow a fixed maintenance therapy schedule of once-monthly subcutaneous dosing rather than dosing as needed in the event of the start of relapse, according to the phase III SCULPTURE trial.
Secukinumab is an investigational fully human IgG1 monoclonal antibody targeting interleukin-17A, a key player in psoriasis, because it generates downstream proinflammatory cytokines and stimulates keratinocyte growth. Earlier studies indicated the investigational biologic had unprecedented clinical efficacy and rapidity of response. These observations gave rise to the hypothesis in SCULPTURE that maintenance dosing as needed upon relapse would prove noninferior to conventional fixed once-monthly maintenance therapy.
If the study hypothesis proved valid, it would mean less exposure to potential medication side effects, lower drug costs, and greater patient convenience. However, the hypothesis wasn’t borne out in the 966-patient, double-blind, randomized SCULPTURE (Study Comparing Retreatment Upon Start of Relapse), Dr. Ulrich Mrowietz reported at the annual congress of the European Academy of Dermatology and Venereology.
SCULPTURE participants had moderate-to-severe chronic plaque psoriasis despite prior systemic therapies, including biologic agents in many cases. They were randomized double blind to induction therapy involving five once-weekly subcutaneous injections of secukinumab at either 150 or 300 mg. At week 8, 843 participants with a Psoriasis Area and Severity Index (PASI) 75 response were re-randomized to maintenance therapy at the same dose, to be delivered either once monthly or as needed for relapse. The definition of relapse in this study required two elements: loss of PASI 75 response, and at least a 20% fall from the maximum PASI improvement, compared with baseline.
At week 52, 78.2% of patients randomized to 300 mg of secukinumab on a fixed once-monthly schedule still maintained a PASI 75 response. This was a significantly better outcome than the 67.7% PASI 75 rate in patients assigned to secukinumab 300 mg as needed, the 62.1% rate in those on secukinumab 150 mg once monthly, and the 52% PASI 75 rate in patients on secukinumab 150 mg as needed, reported Dr. Mrowietz of the University Medical Center Schleswig-Holstein, Kiel (Germany).
He found a bright spot in the negative results: While patients assigned to as-needed maintenance therapy achieved roughly an absolute 10% lower PASI 75 rate at 1 year, those in the secukinumab 150-mg group did so with only 46% the number of doses received by patients in the fixed monthly therapy group, while those in the retreatment-as-needed with secukinumab 300-mg group got only 39% of the number of doses, compared with patients on fixed monthly therapy.
"Fixed monthly dosing is the best maintenance regimen. But, in selected patients, there may be an opportunity with secukinumab to deviate from the usual fixed dosing regimen in favor of an as-needed approach," he said.
This tradeoff of an absolute 10% reduction in efficacy in return for a dosing regimen that entails less than half as much medication over the course of a year could prove of interest to payers, he noted.
The key, according to Dr. Mrowietz, will be to try to identify criteria helpful in selecting patients with an increased likelihood of a high-level response to the retreat-as-needed management strategy. The phase III trial was completed so recently that those necessary subanalyses have yet to be done.
However, Dr. Kristian Reich, another investigator involved in the secukinumab clinical trials program, drew a different message from SCULPTURE. He observed that these newer biologic agents are so effective that the bar has been raised with regard to patient expectations. Many patients won’t be satisfied with a PASI 75 response once a PASI 90 is achievable. And with fixed monthly maintenance secukinumab, it often is.
Indeed, the week-52 PASI 90 rate in SCULPTURE was a highly robust 59.7% in patients on fixed monthly secukinumab 300 mg and 45.8% for fixed monthly low-dose therapy, compared with the unimpressive 13.8% and 11.2% PASI 90 rates with high- and low-dose as-needed therapy.
"My take from this is that the best way to use this drug for continuous disease control is to give the drug every 4 weeks. What this study tells us is for those patients where you have to stop, where you have to use on-and-off therapy because they go away for 2 months to Africa, or they have a major operation, or for other reasons, these data are reassuring that we can use the drug safely on an intermittent basis," said Dr. Reich of Georg-August University in Göttingen, Germany.
Prior to SCULPTURE, he and other investigators were concerned that intermittent secukinumab therapy might promote the development of harmful antidrug antibodies. But antidrug antibodies arose in only three patients on fixed monthly maintenance therapy and two patients on as-needed therapy, and had no impact upon clinical efficacy or safety, he noted.
Another phase III trial presented in Istanbul was ERASURE (Efficacy and Safety of Subcutaneous Secukinumab for Moderate to Severe Chronic Plaque-Type Psoriasis for Up to 1 Year). Dr. Boni E. Elewski reported on 738 patients with moderate-to-severe chronic plaque psoriasis who were randomized double blind to secukinumab at 150 mg or 300 mg, or to placebo. Participants averaged a PASI score of 22 at baseline, along with 33% body surface area involvement. Psoriatic arthritis was present in 23% of subjects.
The co-primary endpoints in ERASURE were the week 12 PASI 75 response rates and Investigator’s Global Assessment scores of 0/1, indicative of clear or almost clear on a modified 5-point scale. The week-12 PASI 75 rates were 81.6% in the secukinumab 300 mg group, 71.6% with secukinumab 150 mg, and 4.5% with placebo. The week-12 IGA 0/1 rates were 65.3%, 51.2%, and 2.4%, said Dr. Elewski of the University of Alabama, Birmingham.
Among the other notable findings in ERASURE were the 69.8% PASI 90 and 41.6% PASI 100 responses at week 16 in the group assigned to secukinumab 300 mg, the fact that only one patient, on secukinumab 150 mg, developed transient antidrug antibodies, and the complete absence of cardiovascular events during 52 weeks on secukinumab, observed Dr. Elewski, professor of dermatology at the University of Alabama, Birmingham.
Dr. Mrowietz, Dr. Reich, and Dr. Elewski reported having received research grants from and serving as consultants to Novartis, which sponsored the secukinumab clinical trials program. The dermatologists serve in similar capacities with other pharmaceutical companies developing new medications for psoriasis.
ISTANBUL – Psoriasis patients who initially achieve a high-level response to secukinumab are significantly more likely to retain it over the long haul if they follow a fixed maintenance therapy schedule of once-monthly subcutaneous dosing rather than dosing as needed in the event of the start of relapse, according to the phase III SCULPTURE trial.
Secukinumab is an investigational fully human IgG1 monoclonal antibody targeting interleukin-17A, a key player in psoriasis, because it generates downstream proinflammatory cytokines and stimulates keratinocyte growth. Earlier studies indicated the investigational biologic had unprecedented clinical efficacy and rapidity of response. These observations gave rise to the hypothesis in SCULPTURE that maintenance dosing as needed upon relapse would prove noninferior to conventional fixed once-monthly maintenance therapy.
If the study hypothesis proved valid, it would mean less exposure to potential medication side effects, lower drug costs, and greater patient convenience. However, the hypothesis wasn’t borne out in the 966-patient, double-blind, randomized SCULPTURE (Study Comparing Retreatment Upon Start of Relapse), Dr. Ulrich Mrowietz reported at the annual congress of the European Academy of Dermatology and Venereology.
SCULPTURE participants had moderate-to-severe chronic plaque psoriasis despite prior systemic therapies, including biologic agents in many cases. They were randomized double blind to induction therapy involving five once-weekly subcutaneous injections of secukinumab at either 150 or 300 mg. At week 8, 843 participants with a Psoriasis Area and Severity Index (PASI) 75 response were re-randomized to maintenance therapy at the same dose, to be delivered either once monthly or as needed for relapse. The definition of relapse in this study required two elements: loss of PASI 75 response, and at least a 20% fall from the maximum PASI improvement, compared with baseline.
At week 52, 78.2% of patients randomized to 300 mg of secukinumab on a fixed once-monthly schedule still maintained a PASI 75 response. This was a significantly better outcome than the 67.7% PASI 75 rate in patients assigned to secukinumab 300 mg as needed, the 62.1% rate in those on secukinumab 150 mg once monthly, and the 52% PASI 75 rate in patients on secukinumab 150 mg as needed, reported Dr. Mrowietz of the University Medical Center Schleswig-Holstein, Kiel (Germany).
He found a bright spot in the negative results: While patients assigned to as-needed maintenance therapy achieved roughly an absolute 10% lower PASI 75 rate at 1 year, those in the secukinumab 150-mg group did so with only 46% the number of doses received by patients in the fixed monthly therapy group, while those in the retreatment-as-needed with secukinumab 300-mg group got only 39% of the number of doses, compared with patients on fixed monthly therapy.
"Fixed monthly dosing is the best maintenance regimen. But, in selected patients, there may be an opportunity with secukinumab to deviate from the usual fixed dosing regimen in favor of an as-needed approach," he said.
This tradeoff of an absolute 10% reduction in efficacy in return for a dosing regimen that entails less than half as much medication over the course of a year could prove of interest to payers, he noted.
The key, according to Dr. Mrowietz, will be to try to identify criteria helpful in selecting patients with an increased likelihood of a high-level response to the retreat-as-needed management strategy. The phase III trial was completed so recently that those necessary subanalyses have yet to be done.
However, Dr. Kristian Reich, another investigator involved in the secukinumab clinical trials program, drew a different message from SCULPTURE. He observed that these newer biologic agents are so effective that the bar has been raised with regard to patient expectations. Many patients won’t be satisfied with a PASI 75 response once a PASI 90 is achievable. And with fixed monthly maintenance secukinumab, it often is.
Indeed, the week-52 PASI 90 rate in SCULPTURE was a highly robust 59.7% in patients on fixed monthly secukinumab 300 mg and 45.8% for fixed monthly low-dose therapy, compared with the unimpressive 13.8% and 11.2% PASI 90 rates with high- and low-dose as-needed therapy.
"My take from this is that the best way to use this drug for continuous disease control is to give the drug every 4 weeks. What this study tells us is for those patients where you have to stop, where you have to use on-and-off therapy because they go away for 2 months to Africa, or they have a major operation, or for other reasons, these data are reassuring that we can use the drug safely on an intermittent basis," said Dr. Reich of Georg-August University in Göttingen, Germany.
Prior to SCULPTURE, he and other investigators were concerned that intermittent secukinumab therapy might promote the development of harmful antidrug antibodies. But antidrug antibodies arose in only three patients on fixed monthly maintenance therapy and two patients on as-needed therapy, and had no impact upon clinical efficacy or safety, he noted.
Another phase III trial presented in Istanbul was ERASURE (Efficacy and Safety of Subcutaneous Secukinumab for Moderate to Severe Chronic Plaque-Type Psoriasis for Up to 1 Year). Dr. Boni E. Elewski reported on 738 patients with moderate-to-severe chronic plaque psoriasis who were randomized double blind to secukinumab at 150 mg or 300 mg, or to placebo. Participants averaged a PASI score of 22 at baseline, along with 33% body surface area involvement. Psoriatic arthritis was present in 23% of subjects.
The co-primary endpoints in ERASURE were the week 12 PASI 75 response rates and Investigator’s Global Assessment scores of 0/1, indicative of clear or almost clear on a modified 5-point scale. The week-12 PASI 75 rates were 81.6% in the secukinumab 300 mg group, 71.6% with secukinumab 150 mg, and 4.5% with placebo. The week-12 IGA 0/1 rates were 65.3%, 51.2%, and 2.4%, said Dr. Elewski of the University of Alabama, Birmingham.
Among the other notable findings in ERASURE were the 69.8% PASI 90 and 41.6% PASI 100 responses at week 16 in the group assigned to secukinumab 300 mg, the fact that only one patient, on secukinumab 150 mg, developed transient antidrug antibodies, and the complete absence of cardiovascular events during 52 weeks on secukinumab, observed Dr. Elewski, professor of dermatology at the University of Alabama, Birmingham.
Dr. Mrowietz, Dr. Reich, and Dr. Elewski reported having received research grants from and serving as consultants to Novartis, which sponsored the secukinumab clinical trials program. The dermatologists serve in similar capacities with other pharmaceutical companies developing new medications for psoriasis.
AT THE EADV CONGRESS
Major finding: Among psoriasis patients who achieved a PASI 75 response to 5 weeks of induction therapy with subcutaneous secukinumab, 59.7% who were placed on a maintenance regimen of 300 mg every 4 weeks had a PASI 90 response at week 52, compared with 13.8% whose maintenance program involved taking 300 mg on an as-needed basis at the start of relapse.
Data source: The SCULPTURE study was a prospective, double-blind, 52-week, phase III clinical trial including 843 patients.
Disclosures: The study was sponsored by Novartis. Speakers reported having received research grants from and serving as consultants to the company.
Expanded role seen for handy new spirometers
ESTES PARK, COLO. – The new generation of portable, user-friendly spirometry devices is likely to gain widespread acceptance among primary care physicians, one pulmonary expert predicted.
"Every subspecialist has their fifth vital sign. Mine is spirometry. Some of these handheld spirometers are really easy to use, and I think they’re going to make their way into office practice," said Dr. Robert L. Keith, professor of medicine at the University of Colorado, Denver.
Spirometry is a tool crucial to diagnosing chronic obstructive pulmonary disease in symptomatic patients. Plus, the forced expiratory volume in 1 second (FEV1) as measured using spirometry is the guideline-recommended means of categorizing patients as to disease stage and the most appropriate choice of treatment in individuals with COPD (Ann. Intern. Med. 2011;155:179-91).
Moreover, spirometry also provides an objective way to assess the effectiveness of treatment, the pulmonologist observed at a conference on internal medicine sponsored by the university.
"One of the neat things about the new software is it can provide quick data on lung age," Dr. Keith explained. "I can see a guy and tell him, ‘I’ve got your spirometry results, and your FEV1 is 41% and your FEV1/forced vital capacity ratio is 0.5,’ and he’s looking at me like, ‘What does that mean, doc?’
"But if I can say, ‘You’re 61 years old and your lungs are 75,’ that’s a take-home message pretty much anyone can understand," said Dr. Keith.
"I also use spirometry to follow patients, so I can say, ‘You’ve quit smoking and are using your medications, and guess what? Your lung age has improved from 75 to age 70,’ " he added.
Examples of state-of-the-art handheld spirometry devices on the market today include the Vitalograph copd-6, the PulmoLife, and PiKo-6, Dr. Keith noted.
"We tend to have a very rural population in Colorado that flows into Denver to be seen at the VA," [Veterans Affairs] he explained. "We give patients handheld spirometers to take back home to measure their disease and report in about their lung function."
Peak expiratory flow rate underestimates COPD severity, Dr. Keith cautioned, and cannot be used to diagnose COPD. Instead, peak expiratory flow is a spirometry measurement used as a tool in managing asthma.
He reported serving on speakers bureaus for Pfizer and Boehringer-Ingelheim.
ESTES PARK, COLO. – The new generation of portable, user-friendly spirometry devices is likely to gain widespread acceptance among primary care physicians, one pulmonary expert predicted.
"Every subspecialist has their fifth vital sign. Mine is spirometry. Some of these handheld spirometers are really easy to use, and I think they’re going to make their way into office practice," said Dr. Robert L. Keith, professor of medicine at the University of Colorado, Denver.
Spirometry is a tool crucial to diagnosing chronic obstructive pulmonary disease in symptomatic patients. Plus, the forced expiratory volume in 1 second (FEV1) as measured using spirometry is the guideline-recommended means of categorizing patients as to disease stage and the most appropriate choice of treatment in individuals with COPD (Ann. Intern. Med. 2011;155:179-91).
Moreover, spirometry also provides an objective way to assess the effectiveness of treatment, the pulmonologist observed at a conference on internal medicine sponsored by the university.
"One of the neat things about the new software is it can provide quick data on lung age," Dr. Keith explained. "I can see a guy and tell him, ‘I’ve got your spirometry results, and your FEV1 is 41% and your FEV1/forced vital capacity ratio is 0.5,’ and he’s looking at me like, ‘What does that mean, doc?’
"But if I can say, ‘You’re 61 years old and your lungs are 75,’ that’s a take-home message pretty much anyone can understand," said Dr. Keith.
"I also use spirometry to follow patients, so I can say, ‘You’ve quit smoking and are using your medications, and guess what? Your lung age has improved from 75 to age 70,’ " he added.
Examples of state-of-the-art handheld spirometry devices on the market today include the Vitalograph copd-6, the PulmoLife, and PiKo-6, Dr. Keith noted.
"We tend to have a very rural population in Colorado that flows into Denver to be seen at the VA," [Veterans Affairs] he explained. "We give patients handheld spirometers to take back home to measure their disease and report in about their lung function."
Peak expiratory flow rate underestimates COPD severity, Dr. Keith cautioned, and cannot be used to diagnose COPD. Instead, peak expiratory flow is a spirometry measurement used as a tool in managing asthma.
He reported serving on speakers bureaus for Pfizer and Boehringer-Ingelheim.
ESTES PARK, COLO. – The new generation of portable, user-friendly spirometry devices is likely to gain widespread acceptance among primary care physicians, one pulmonary expert predicted.
"Every subspecialist has their fifth vital sign. Mine is spirometry. Some of these handheld spirometers are really easy to use, and I think they’re going to make their way into office practice," said Dr. Robert L. Keith, professor of medicine at the University of Colorado, Denver.
Spirometry is a tool crucial to diagnosing chronic obstructive pulmonary disease in symptomatic patients. Plus, the forced expiratory volume in 1 second (FEV1) as measured using spirometry is the guideline-recommended means of categorizing patients as to disease stage and the most appropriate choice of treatment in individuals with COPD (Ann. Intern. Med. 2011;155:179-91).
Moreover, spirometry also provides an objective way to assess the effectiveness of treatment, the pulmonologist observed at a conference on internal medicine sponsored by the university.
"One of the neat things about the new software is it can provide quick data on lung age," Dr. Keith explained. "I can see a guy and tell him, ‘I’ve got your spirometry results, and your FEV1 is 41% and your FEV1/forced vital capacity ratio is 0.5,’ and he’s looking at me like, ‘What does that mean, doc?’
"But if I can say, ‘You’re 61 years old and your lungs are 75,’ that’s a take-home message pretty much anyone can understand," said Dr. Keith.
"I also use spirometry to follow patients, so I can say, ‘You’ve quit smoking and are using your medications, and guess what? Your lung age has improved from 75 to age 70,’ " he added.
Examples of state-of-the-art handheld spirometry devices on the market today include the Vitalograph copd-6, the PulmoLife, and PiKo-6, Dr. Keith noted.
"We tend to have a very rural population in Colorado that flows into Denver to be seen at the VA," [Veterans Affairs] he explained. "We give patients handheld spirometers to take back home to measure their disease and report in about their lung function."
Peak expiratory flow rate underestimates COPD severity, Dr. Keith cautioned, and cannot be used to diagnose COPD. Instead, peak expiratory flow is a spirometry measurement used as a tool in managing asthma.
He reported serving on speakers bureaus for Pfizer and Boehringer-Ingelheim.
EXPERT ANALYSIS FROM THE ANNUAL INTERNAL MEDICINE PROGRAM
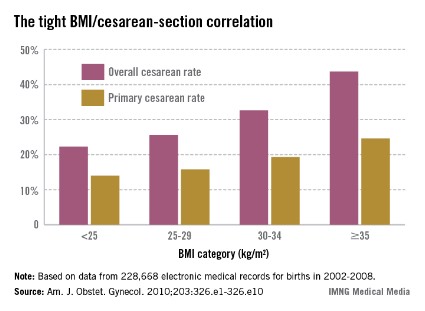
Anticipate the many challenges of cesarean section in superobese
DENVER – In current practice, the majority of superobese pregnant women – those having a prepregnancy body mass index of 50 kg/m2 or more – will end up having a cesarean section. The inherent technical challenges make it crucial to have a plan in place before heading to the operating room.
"There’s a lot to think about: patient positioning, choice of incision, antibiotic prophylaxis, deep vein thrombosis prophylaxis, wound care," Dr. Mark Alanis observed at the annual meeting of the Society of Ob/Gyn Hospitalists.
Also, it’s important to understand up front that one in three of these superobese patients will have a significant wound complication. Eighty-five percent of these are cellulitis or wound disruptions, mostly seromas, which will require packing. But one in seven of the wound complications are abscesses, and affected superobese patients require hospital readmission, said Dr. Alanis of the University of Colorado, Denver.
He presented lessons learned from his own retrospective study of 194 superobese patients who underwent cesarean section, plus several studies by other investigators. Among the highlights:
• Anesthesia evaluation. Bring the anesthesiologist in on the case early. Finding the landmarks for spinal anesthesia is tough in a superobese patient. Failed regional anesthesia is more common. In Dr. Alanis’ series, general anesthesia was required in 15% of patients – a far higher rate than in leaner women – so a careful preoperative airway assessment is essential.
• Patient positioning. Understand that positioning the superobese patient at a 20-degree tilt puts the midline far away from the surgeon, who’ll have to operate bending forward. "My back is killing me when I do these operations," the ob.gyn. said.
• Choice of incision. Dr. Alanis recommends the Pfannenstiel incision. This horizontal incision is faster than a vertical incision, the wound hurts less, healing is better, and the classic teaching that it poses an increased risk of infection in massively obese patients is a myth unsupported by data.
The key is to first mobilize the panniculus, moving it up off the suprapubic region, then securing it with a Montgomery strap tied off to the bedposts.
"It takes 5 minutes to secure the pannis. It’s the easiest thing in the world, and you don’t need an assistant for the operation once it’s done," he explained.
• Operative characteristics. The mean skin-to-delivery time in Dr. Alanis’ series was 15 minutes, with an incision-to-closure time of 64 minutes and an estimated blood loss of 1,000 mL, all considerably greater than in leaner patients.
In another investigator’s study involving 193 superobese women, the incision-to-delivery time was nearly identical at 16 minutes, and the fetal distress rate as measured by cord pH, Apgar score, and neonatal ICU admission was significantly higher than in patients with a lower body mass index (Am. J. Obstet. Gynecol. 2013;209:386.e1-386.e6).
This increased risk of fetal distress was confirmed recently in a study from the National Institutes of Health Maternal-Fetal Medicine Units Network. The analysis of 5,742 mother/singleton term neonate pairs delivered by prelabor cesarean section demonstrated that fetal distress increased with greater body mass index category. For every 10-unit increase in BMI, the cord arterial pH decreased by an adjusted value of 0.01 and the base deficit increased by 0.26 mmol/L. The relationship wasn’t linear, though; the steepest increase in fetal distress was seen in women with a prepregnancy BMI of 40 kg/L or more (Obstet. Gynecol. 2013;122:262-7).
"We want these women to deliver vaginally, but be cognizant that it’s going to take longer to take that woman to the OR, and it’s going to take longer to get that baby out," he observed.
• Antibiotic prophylaxis. A major practice trend in the past several years has been a shift to routine administration of preincision antibiotics.
• Deep vein thrombosis prophylaxis. Two-thirds of postsurgical DVTs are deemed preventable. The risk in pregnancy jumps from a fourfold increase with vaginal delivery over that in daily life, to a 13-fold increase with cesarean delivery, to a 26-fold increase with emergent cesarean section – and emergent cesarean section is considerably more common in superobese patients than in those of lesser BMI. Also, a BMI of 40 or more is an independent risk factor for DVT.
Dr. Alanis strongly recommends having an order set in place. While the risk of postpartum hemorrhage climbs with increasing BMI, this is not due to the use of anticoagulation. Nor does anticoagulation in superobese patients undergoing cesarean section raise their risk of hematoma or wound complications, he added.
• Wound closure. An audience show of hands indicated a strong preference for subcutaneous closure. Dr. Alanis said that’s probably fine for patients who are merely overweight, but in the superobese – women who often have 5-10 cm of subcutaneous thickness – it doesn’t improve the risk of seroma. He believes retention sutures are a good idea that hasn’t been well studied. Subcutaneous drains proved to be a bust in his study, as well as in every other study ever published.
"I would abandon the practice if I were you," the ob.gyn. advised.
Similarly, negative pressure dressings sound like a good idea but have proved disappointing in randomized clinical trials.
Dr. Alanis favors delayed closure. He’ll pack the wound for 3-4 days while granulation tissue forms, and then sew the wound closed in the office.
• Managing wound complications. To Dr. Alanis’ surprise, factors that proved unrelated to the risk of wound complications in his study of the superobese included labor as opposed to nonlabor, labor duration, rupture of membranes, chorioamnionitis, operative time, and emergent vs. routine vs. urgent cesarean section. Indeed, the only predictors of wound complications in his series were subcutaneous drains, associated with a 2.4-fold increased risk, and smoking, with a 2.9-fold elevated risk.
Eighty-six percent of wound complications in the superobese women were diagnosed post discharge. Wound disruption was diagnosed on median postoperative day 8.5. A total of 24% of patients with a wound complication were readmitted; 14% underwent reoperation.
Delayed closure is a very attractive way to manage seromas and hematomas. It requires healthy pink tissue and is not a technique for patients with a postoperative abscess.
Dr. Alanis reported having no financial interests germane to his presentation.
DENVER – In current practice, the majority of superobese pregnant women – those having a prepregnancy body mass index of 50 kg/m2 or more – will end up having a cesarean section. The inherent technical challenges make it crucial to have a plan in place before heading to the operating room.
"There’s a lot to think about: patient positioning, choice of incision, antibiotic prophylaxis, deep vein thrombosis prophylaxis, wound care," Dr. Mark Alanis observed at the annual meeting of the Society of Ob/Gyn Hospitalists.
Also, it’s important to understand up front that one in three of these superobese patients will have a significant wound complication. Eighty-five percent of these are cellulitis or wound disruptions, mostly seromas, which will require packing. But one in seven of the wound complications are abscesses, and affected superobese patients require hospital readmission, said Dr. Alanis of the University of Colorado, Denver.
He presented lessons learned from his own retrospective study of 194 superobese patients who underwent cesarean section, plus several studies by other investigators. Among the highlights:
• Anesthesia evaluation. Bring the anesthesiologist in on the case early. Finding the landmarks for spinal anesthesia is tough in a superobese patient. Failed regional anesthesia is more common. In Dr. Alanis’ series, general anesthesia was required in 15% of patients – a far higher rate than in leaner women – so a careful preoperative airway assessment is essential.
• Patient positioning. Understand that positioning the superobese patient at a 20-degree tilt puts the midline far away from the surgeon, who’ll have to operate bending forward. "My back is killing me when I do these operations," the ob.gyn. said.
• Choice of incision. Dr. Alanis recommends the Pfannenstiel incision. This horizontal incision is faster than a vertical incision, the wound hurts less, healing is better, and the classic teaching that it poses an increased risk of infection in massively obese patients is a myth unsupported by data.
The key is to first mobilize the panniculus, moving it up off the suprapubic region, then securing it with a Montgomery strap tied off to the bedposts.
"It takes 5 minutes to secure the pannis. It’s the easiest thing in the world, and you don’t need an assistant for the operation once it’s done," he explained.
• Operative characteristics. The mean skin-to-delivery time in Dr. Alanis’ series was 15 minutes, with an incision-to-closure time of 64 minutes and an estimated blood loss of 1,000 mL, all considerably greater than in leaner patients.
In another investigator’s study involving 193 superobese women, the incision-to-delivery time was nearly identical at 16 minutes, and the fetal distress rate as measured by cord pH, Apgar score, and neonatal ICU admission was significantly higher than in patients with a lower body mass index (Am. J. Obstet. Gynecol. 2013;209:386.e1-386.e6).
This increased risk of fetal distress was confirmed recently in a study from the National Institutes of Health Maternal-Fetal Medicine Units Network. The analysis of 5,742 mother/singleton term neonate pairs delivered by prelabor cesarean section demonstrated that fetal distress increased with greater body mass index category. For every 10-unit increase in BMI, the cord arterial pH decreased by an adjusted value of 0.01 and the base deficit increased by 0.26 mmol/L. The relationship wasn’t linear, though; the steepest increase in fetal distress was seen in women with a prepregnancy BMI of 40 kg/L or more (Obstet. Gynecol. 2013;122:262-7).
"We want these women to deliver vaginally, but be cognizant that it’s going to take longer to take that woman to the OR, and it’s going to take longer to get that baby out," he observed.
• Antibiotic prophylaxis. A major practice trend in the past several years has been a shift to routine administration of preincision antibiotics.
• Deep vein thrombosis prophylaxis. Two-thirds of postsurgical DVTs are deemed preventable. The risk in pregnancy jumps from a fourfold increase with vaginal delivery over that in daily life, to a 13-fold increase with cesarean delivery, to a 26-fold increase with emergent cesarean section – and emergent cesarean section is considerably more common in superobese patients than in those of lesser BMI. Also, a BMI of 40 or more is an independent risk factor for DVT.
Dr. Alanis strongly recommends having an order set in place. While the risk of postpartum hemorrhage climbs with increasing BMI, this is not due to the use of anticoagulation. Nor does anticoagulation in superobese patients undergoing cesarean section raise their risk of hematoma or wound complications, he added.
• Wound closure. An audience show of hands indicated a strong preference for subcutaneous closure. Dr. Alanis said that’s probably fine for patients who are merely overweight, but in the superobese – women who often have 5-10 cm of subcutaneous thickness – it doesn’t improve the risk of seroma. He believes retention sutures are a good idea that hasn’t been well studied. Subcutaneous drains proved to be a bust in his study, as well as in every other study ever published.
"I would abandon the practice if I were you," the ob.gyn. advised.
Similarly, negative pressure dressings sound like a good idea but have proved disappointing in randomized clinical trials.
Dr. Alanis favors delayed closure. He’ll pack the wound for 3-4 days while granulation tissue forms, and then sew the wound closed in the office.
• Managing wound complications. To Dr. Alanis’ surprise, factors that proved unrelated to the risk of wound complications in his study of the superobese included labor as opposed to nonlabor, labor duration, rupture of membranes, chorioamnionitis, operative time, and emergent vs. routine vs. urgent cesarean section. Indeed, the only predictors of wound complications in his series were subcutaneous drains, associated with a 2.4-fold increased risk, and smoking, with a 2.9-fold elevated risk.
Eighty-six percent of wound complications in the superobese women were diagnosed post discharge. Wound disruption was diagnosed on median postoperative day 8.5. A total of 24% of patients with a wound complication were readmitted; 14% underwent reoperation.
Delayed closure is a very attractive way to manage seromas and hematomas. It requires healthy pink tissue and is not a technique for patients with a postoperative abscess.
Dr. Alanis reported having no financial interests germane to his presentation.
DENVER – In current practice, the majority of superobese pregnant women – those having a prepregnancy body mass index of 50 kg/m2 or more – will end up having a cesarean section. The inherent technical challenges make it crucial to have a plan in place before heading to the operating room.
"There’s a lot to think about: patient positioning, choice of incision, antibiotic prophylaxis, deep vein thrombosis prophylaxis, wound care," Dr. Mark Alanis observed at the annual meeting of the Society of Ob/Gyn Hospitalists.
Also, it’s important to understand up front that one in three of these superobese patients will have a significant wound complication. Eighty-five percent of these are cellulitis or wound disruptions, mostly seromas, which will require packing. But one in seven of the wound complications are abscesses, and affected superobese patients require hospital readmission, said Dr. Alanis of the University of Colorado, Denver.
He presented lessons learned from his own retrospective study of 194 superobese patients who underwent cesarean section, plus several studies by other investigators. Among the highlights:
• Anesthesia evaluation. Bring the anesthesiologist in on the case early. Finding the landmarks for spinal anesthesia is tough in a superobese patient. Failed regional anesthesia is more common. In Dr. Alanis’ series, general anesthesia was required in 15% of patients – a far higher rate than in leaner women – so a careful preoperative airway assessment is essential.
• Patient positioning. Understand that positioning the superobese patient at a 20-degree tilt puts the midline far away from the surgeon, who’ll have to operate bending forward. "My back is killing me when I do these operations," the ob.gyn. said.
• Choice of incision. Dr. Alanis recommends the Pfannenstiel incision. This horizontal incision is faster than a vertical incision, the wound hurts less, healing is better, and the classic teaching that it poses an increased risk of infection in massively obese patients is a myth unsupported by data.
The key is to first mobilize the panniculus, moving it up off the suprapubic region, then securing it with a Montgomery strap tied off to the bedposts.
"It takes 5 minutes to secure the pannis. It’s the easiest thing in the world, and you don’t need an assistant for the operation once it’s done," he explained.
• Operative characteristics. The mean skin-to-delivery time in Dr. Alanis’ series was 15 minutes, with an incision-to-closure time of 64 minutes and an estimated blood loss of 1,000 mL, all considerably greater than in leaner patients.
In another investigator’s study involving 193 superobese women, the incision-to-delivery time was nearly identical at 16 minutes, and the fetal distress rate as measured by cord pH, Apgar score, and neonatal ICU admission was significantly higher than in patients with a lower body mass index (Am. J. Obstet. Gynecol. 2013;209:386.e1-386.e6).
This increased risk of fetal distress was confirmed recently in a study from the National Institutes of Health Maternal-Fetal Medicine Units Network. The analysis of 5,742 mother/singleton term neonate pairs delivered by prelabor cesarean section demonstrated that fetal distress increased with greater body mass index category. For every 10-unit increase in BMI, the cord arterial pH decreased by an adjusted value of 0.01 and the base deficit increased by 0.26 mmol/L. The relationship wasn’t linear, though; the steepest increase in fetal distress was seen in women with a prepregnancy BMI of 40 kg/L or more (Obstet. Gynecol. 2013;122:262-7).
"We want these women to deliver vaginally, but be cognizant that it’s going to take longer to take that woman to the OR, and it’s going to take longer to get that baby out," he observed.
• Antibiotic prophylaxis. A major practice trend in the past several years has been a shift to routine administration of preincision antibiotics.
• Deep vein thrombosis prophylaxis. Two-thirds of postsurgical DVTs are deemed preventable. The risk in pregnancy jumps from a fourfold increase with vaginal delivery over that in daily life, to a 13-fold increase with cesarean delivery, to a 26-fold increase with emergent cesarean section – and emergent cesarean section is considerably more common in superobese patients than in those of lesser BMI. Also, a BMI of 40 or more is an independent risk factor for DVT.
Dr. Alanis strongly recommends having an order set in place. While the risk of postpartum hemorrhage climbs with increasing BMI, this is not due to the use of anticoagulation. Nor does anticoagulation in superobese patients undergoing cesarean section raise their risk of hematoma or wound complications, he added.
• Wound closure. An audience show of hands indicated a strong preference for subcutaneous closure. Dr. Alanis said that’s probably fine for patients who are merely overweight, but in the superobese – women who often have 5-10 cm of subcutaneous thickness – it doesn’t improve the risk of seroma. He believes retention sutures are a good idea that hasn’t been well studied. Subcutaneous drains proved to be a bust in his study, as well as in every other study ever published.
"I would abandon the practice if I were you," the ob.gyn. advised.
Similarly, negative pressure dressings sound like a good idea but have proved disappointing in randomized clinical trials.
Dr. Alanis favors delayed closure. He’ll pack the wound for 3-4 days while granulation tissue forms, and then sew the wound closed in the office.
• Managing wound complications. To Dr. Alanis’ surprise, factors that proved unrelated to the risk of wound complications in his study of the superobese included labor as opposed to nonlabor, labor duration, rupture of membranes, chorioamnionitis, operative time, and emergent vs. routine vs. urgent cesarean section. Indeed, the only predictors of wound complications in his series were subcutaneous drains, associated with a 2.4-fold increased risk, and smoking, with a 2.9-fold elevated risk.
Eighty-six percent of wound complications in the superobese women were diagnosed post discharge. Wound disruption was diagnosed on median postoperative day 8.5. A total of 24% of patients with a wound complication were readmitted; 14% underwent reoperation.
Delayed closure is a very attractive way to manage seromas and hematomas. It requires healthy pink tissue and is not a technique for patients with a postoperative abscess.
Dr. Alanis reported having no financial interests germane to his presentation.
EXPERT ANALYSIS FROM THE SOGH ANNUAL CLINICAL MEETING
Vismodegib prolongs positive response in advanced skin cancer
ISTANBUL – Vismodegib-treated patients with locally advanced basal cell carcinoma showed triple the median response duration after 24 months’ follow-up beyond the primary analysis of the drug, Dr. Luc Dirix reported at the annual congress of the European Academy of Dermatology and Venereology.
Vismodegib, the first-in-class oral medication for treatment of advanced BCC, showed a greatly improved duration of response and no new safety signals in updated results from the ERIVANCE BCC trial, which earlier had earned the drug marketing approval from the Food and Drug Administration and European Medicines Agency.
After 24 months beyond what was included in the primary analysis of ERIVANCE BCC (N. Engl. J. Med. 2012;366:2171-9), the median duration of response by investigator assessment in patients with locally advanced BCC increased from 7.6 to 26.2 months, and increased from 7.6 to 12.9 months in those with metastatic BCC.
"The main new fact is that the median duration of response has increased importantly. That’s a very impressive improvement in disease control, especially in patients with locally advanced disease. It’s a major change from what you’ll find in the New England Journal of Medicine publication," observed Dr. Dirix of Sint-AugustinusHospital, Antwerp, Belgium.
Dr. Dirix reported that, with the additional 24 months of follow-up in the ERIVANCE BCC study, median progression-free survival now stands at 9.3 months in the group with metastatic BCC and 12.9 months in those with locally advanced disease. The objective response rate, which was 43% in patients with locally advanced BCC and 30% in those with metastatic disease in the primary analysis, hasn’t changed significantly with the additional follow-up, he said.
The median overall survival was 33 months. Twelve of the original 33 patients in the cohort with metastatic BCC have died of progressive disease, as have 4 of 63 with locally advanced BCC.
The ERIVANCE BCC trial is an open-label, phase II study in which participants were placed on vismodegib at 150 mg once daily until the occurrence of disease progression, intolerable toxicity, or patient request to withdraw. At the latest follow-up, only 13% of the original study population remained on the drug, Dr. Dirix said. Half of the patients with metastatic BCC who discontinued the drug did so because of disease progression, but only 16% of those with locally advanced BCC stopped treatment. Instead, side effects were the main reason locally advanced BCC patients discontinued therapy.
"The additional 24 months of follow-up didn’t result in any new safety signal, but there are numerous side effects, especially muscle spasms, alopecia, dysgeusia, and others, mostly mild or moderate," said Dr. Dirix.
An estimated 1.6 million new cases of BCC were diagnosed in the United States in 2006, making it the most common form of cancer in the country. Most BCCs are readily treated. However, some cases can progress to locally advanced or metastatic BCC, a disfiguring and potentially life-threatening condition that is not treatable surgically or via radiation therapy, and for which no effective therapy existed before. Vismodegib (Erivedge) provides an oral drug option that works as an inhibitor of the hedgehog pathway, which plays a key role in the pathogenesis of most BCCs.
With vismodegib intended as lifelong treatment in patients with advanced BCC, and considerable research interest in exploring the drug in other forms of BCC, reliable safety data are needed. Also at the EADV Congress, Dr. Rainer Kunstfeld presented new safety information in an interim analysis from an ongoing study known as STEVIE (Safety Events in Vismodegib), the largest study of the hedgehog pathway inhibitor. His report included the first 300 of a planned 1,200 patients with advanced BCC to be placed on vismodegib.
The data underscore the point that advanced BCC is primarily a disease of the elderly. The mean age of the 278 patients with locally advanced BCC is 70 years, and 65 years in the 22 with metastatic BCC. The median treatment duration as of the data cutoff for this analysis was 5.8 months.
Forty-five percent of patients in the locally advanced BCC group have discontinued treatment, as have 32% of those with metastatic disease, Dr. Kunstfeld said. The reasons differed for the two groups. Discontinuation due to disease progression or death was more than twice as common in patients with metastatic BCC. Patients with locally advanced disease were much more likely to halt treatment because of adverse events.
Ninety-three percent of STEVIE participants experienced at least one adverse event. Roughly two-thirds of these side effects were mild or moderate – that is, grade 1 or 2 – while the remainder were grade 3. The incidence of the most common adverse events were muscle spasms in 59% of participants, alopecia in 49%, distorted sense of taste in 41%, loss of the sense of taste in 26%, asthenia in 23%, and weight loss in 16%. These data were consistent with the safety profile seen in the ERIVANCE BCC study, observed Dr. Kunstfeld, a dermatologist at the Medical University of Vienna.
Of the 251 patients with tumor assessments available to date, more than 95% showed some degree of therapeutic benefit. A complete response was documented in 17.5% of patients, a partial response in 39.8%, and 39% had stable disease. Fewer than 3% have experienced disease progression during this early follow-up period.
The ERIVANCE BCC and STEVIE studies are funded by Genentech. Dr. Dirix and Dr. Kunstfeld reported having no financial conflicts of interest.
ISTANBUL – Vismodegib-treated patients with locally advanced basal cell carcinoma showed triple the median response duration after 24 months’ follow-up beyond the primary analysis of the drug, Dr. Luc Dirix reported at the annual congress of the European Academy of Dermatology and Venereology.
Vismodegib, the first-in-class oral medication for treatment of advanced BCC, showed a greatly improved duration of response and no new safety signals in updated results from the ERIVANCE BCC trial, which earlier had earned the drug marketing approval from the Food and Drug Administration and European Medicines Agency.
After 24 months beyond what was included in the primary analysis of ERIVANCE BCC (N. Engl. J. Med. 2012;366:2171-9), the median duration of response by investigator assessment in patients with locally advanced BCC increased from 7.6 to 26.2 months, and increased from 7.6 to 12.9 months in those with metastatic BCC.
"The main new fact is that the median duration of response has increased importantly. That’s a very impressive improvement in disease control, especially in patients with locally advanced disease. It’s a major change from what you’ll find in the New England Journal of Medicine publication," observed Dr. Dirix of Sint-AugustinusHospital, Antwerp, Belgium.
Dr. Dirix reported that, with the additional 24 months of follow-up in the ERIVANCE BCC study, median progression-free survival now stands at 9.3 months in the group with metastatic BCC and 12.9 months in those with locally advanced disease. The objective response rate, which was 43% in patients with locally advanced BCC and 30% in those with metastatic disease in the primary analysis, hasn’t changed significantly with the additional follow-up, he said.
The median overall survival was 33 months. Twelve of the original 33 patients in the cohort with metastatic BCC have died of progressive disease, as have 4 of 63 with locally advanced BCC.
The ERIVANCE BCC trial is an open-label, phase II study in which participants were placed on vismodegib at 150 mg once daily until the occurrence of disease progression, intolerable toxicity, or patient request to withdraw. At the latest follow-up, only 13% of the original study population remained on the drug, Dr. Dirix said. Half of the patients with metastatic BCC who discontinued the drug did so because of disease progression, but only 16% of those with locally advanced BCC stopped treatment. Instead, side effects were the main reason locally advanced BCC patients discontinued therapy.
"The additional 24 months of follow-up didn’t result in any new safety signal, but there are numerous side effects, especially muscle spasms, alopecia, dysgeusia, and others, mostly mild or moderate," said Dr. Dirix.
An estimated 1.6 million new cases of BCC were diagnosed in the United States in 2006, making it the most common form of cancer in the country. Most BCCs are readily treated. However, some cases can progress to locally advanced or metastatic BCC, a disfiguring and potentially life-threatening condition that is not treatable surgically or via radiation therapy, and for which no effective therapy existed before. Vismodegib (Erivedge) provides an oral drug option that works as an inhibitor of the hedgehog pathway, which plays a key role in the pathogenesis of most BCCs.
With vismodegib intended as lifelong treatment in patients with advanced BCC, and considerable research interest in exploring the drug in other forms of BCC, reliable safety data are needed. Also at the EADV Congress, Dr. Rainer Kunstfeld presented new safety information in an interim analysis from an ongoing study known as STEVIE (Safety Events in Vismodegib), the largest study of the hedgehog pathway inhibitor. His report included the first 300 of a planned 1,200 patients with advanced BCC to be placed on vismodegib.
The data underscore the point that advanced BCC is primarily a disease of the elderly. The mean age of the 278 patients with locally advanced BCC is 70 years, and 65 years in the 22 with metastatic BCC. The median treatment duration as of the data cutoff for this analysis was 5.8 months.
Forty-five percent of patients in the locally advanced BCC group have discontinued treatment, as have 32% of those with metastatic disease, Dr. Kunstfeld said. The reasons differed for the two groups. Discontinuation due to disease progression or death was more than twice as common in patients with metastatic BCC. Patients with locally advanced disease were much more likely to halt treatment because of adverse events.
Ninety-three percent of STEVIE participants experienced at least one adverse event. Roughly two-thirds of these side effects were mild or moderate – that is, grade 1 or 2 – while the remainder were grade 3. The incidence of the most common adverse events were muscle spasms in 59% of participants, alopecia in 49%, distorted sense of taste in 41%, loss of the sense of taste in 26%, asthenia in 23%, and weight loss in 16%. These data were consistent with the safety profile seen in the ERIVANCE BCC study, observed Dr. Kunstfeld, a dermatologist at the Medical University of Vienna.
Of the 251 patients with tumor assessments available to date, more than 95% showed some degree of therapeutic benefit. A complete response was documented in 17.5% of patients, a partial response in 39.8%, and 39% had stable disease. Fewer than 3% have experienced disease progression during this early follow-up period.
The ERIVANCE BCC and STEVIE studies are funded by Genentech. Dr. Dirix and Dr. Kunstfeld reported having no financial conflicts of interest.
ISTANBUL – Vismodegib-treated patients with locally advanced basal cell carcinoma showed triple the median response duration after 24 months’ follow-up beyond the primary analysis of the drug, Dr. Luc Dirix reported at the annual congress of the European Academy of Dermatology and Venereology.
Vismodegib, the first-in-class oral medication for treatment of advanced BCC, showed a greatly improved duration of response and no new safety signals in updated results from the ERIVANCE BCC trial, which earlier had earned the drug marketing approval from the Food and Drug Administration and European Medicines Agency.
After 24 months beyond what was included in the primary analysis of ERIVANCE BCC (N. Engl. J. Med. 2012;366:2171-9), the median duration of response by investigator assessment in patients with locally advanced BCC increased from 7.6 to 26.2 months, and increased from 7.6 to 12.9 months in those with metastatic BCC.
"The main new fact is that the median duration of response has increased importantly. That’s a very impressive improvement in disease control, especially in patients with locally advanced disease. It’s a major change from what you’ll find in the New England Journal of Medicine publication," observed Dr. Dirix of Sint-AugustinusHospital, Antwerp, Belgium.
Dr. Dirix reported that, with the additional 24 months of follow-up in the ERIVANCE BCC study, median progression-free survival now stands at 9.3 months in the group with metastatic BCC and 12.9 months in those with locally advanced disease. The objective response rate, which was 43% in patients with locally advanced BCC and 30% in those with metastatic disease in the primary analysis, hasn’t changed significantly with the additional follow-up, he said.
The median overall survival was 33 months. Twelve of the original 33 patients in the cohort with metastatic BCC have died of progressive disease, as have 4 of 63 with locally advanced BCC.
The ERIVANCE BCC trial is an open-label, phase II study in which participants were placed on vismodegib at 150 mg once daily until the occurrence of disease progression, intolerable toxicity, or patient request to withdraw. At the latest follow-up, only 13% of the original study population remained on the drug, Dr. Dirix said. Half of the patients with metastatic BCC who discontinued the drug did so because of disease progression, but only 16% of those with locally advanced BCC stopped treatment. Instead, side effects were the main reason locally advanced BCC patients discontinued therapy.
"The additional 24 months of follow-up didn’t result in any new safety signal, but there are numerous side effects, especially muscle spasms, alopecia, dysgeusia, and others, mostly mild or moderate," said Dr. Dirix.
An estimated 1.6 million new cases of BCC were diagnosed in the United States in 2006, making it the most common form of cancer in the country. Most BCCs are readily treated. However, some cases can progress to locally advanced or metastatic BCC, a disfiguring and potentially life-threatening condition that is not treatable surgically or via radiation therapy, and for which no effective therapy existed before. Vismodegib (Erivedge) provides an oral drug option that works as an inhibitor of the hedgehog pathway, which plays a key role in the pathogenesis of most BCCs.
With vismodegib intended as lifelong treatment in patients with advanced BCC, and considerable research interest in exploring the drug in other forms of BCC, reliable safety data are needed. Also at the EADV Congress, Dr. Rainer Kunstfeld presented new safety information in an interim analysis from an ongoing study known as STEVIE (Safety Events in Vismodegib), the largest study of the hedgehog pathway inhibitor. His report included the first 300 of a planned 1,200 patients with advanced BCC to be placed on vismodegib.
The data underscore the point that advanced BCC is primarily a disease of the elderly. The mean age of the 278 patients with locally advanced BCC is 70 years, and 65 years in the 22 with metastatic BCC. The median treatment duration as of the data cutoff for this analysis was 5.8 months.
Forty-five percent of patients in the locally advanced BCC group have discontinued treatment, as have 32% of those with metastatic disease, Dr. Kunstfeld said. The reasons differed for the two groups. Discontinuation due to disease progression or death was more than twice as common in patients with metastatic BCC. Patients with locally advanced disease were much more likely to halt treatment because of adverse events.
Ninety-three percent of STEVIE participants experienced at least one adverse event. Roughly two-thirds of these side effects were mild or moderate – that is, grade 1 or 2 – while the remainder were grade 3. The incidence of the most common adverse events were muscle spasms in 59% of participants, alopecia in 49%, distorted sense of taste in 41%, loss of the sense of taste in 26%, asthenia in 23%, and weight loss in 16%. These data were consistent with the safety profile seen in the ERIVANCE BCC study, observed Dr. Kunstfeld, a dermatologist at the Medical University of Vienna.
Of the 251 patients with tumor assessments available to date, more than 95% showed some degree of therapeutic benefit. A complete response was documented in 17.5% of patients, a partial response in 39.8%, and 39% had stable disease. Fewer than 3% have experienced disease progression during this early follow-up period.
The ERIVANCE BCC and STEVIE studies are funded by Genentech. Dr. Dirix and Dr. Kunstfeld reported having no financial conflicts of interest.
AT THE EADV CONGRESS
Major finding: With an additional 24 months of follow-up of vismodegib-treated patients with advanced basal cell carcinoma beyond previous results from the ERIVANCE study, the median response duration in patients with locally advanced disease climbed from 7.6 to 26.2 months.
Data source: The ERIVANCE BCC study is an open-label trial in which 63 patients with locally advanced BCC and 33 with metastatic BCC were placed on oral vismodegib at 150 mg/day.
Disclosures: The ERIVANCE BCC and STEVIE studies are funded by Genentech. Dr. Dirix and Dr. Kunstfeld reported having no financial conflicts of interest.
Gender-specific biomarker thresholds urged in MI diagnosis
AMSTERDAM – The use of a high-sensitivity cardiac troponin I assay and gender-specific cutoffs to define acute myocardial infarction nearly doubled the diagnosis of MI in women in an early subanalysis from the High-STEACS trial.
High-sensitivity troponin assays have identified gender differences in the normal reference range. This becomes relevant in light of the third universal definition of acute MI, released last year, which defines the biomarker threshold for the diagnosis of MI as a cardiac troponin at the 99th percentile of a healthy reference population (Eur. Heart J. 2012;33:2551-67). The conventional cardiac troponin assays in widespread use today aren’t sufficiently sensitive to detect gender differences in the normal reference range, so they rely upon a single diagnostic threshold that sets the bar so high it appears to lead to underdiagnosis of MI in women. This likely contributes to gender inequalities in treatment and outcome, Dr. Nicholas L. Mills asserted at the annual congress of the European Society of Cardiology.
The high-sensitivity cardiac troponin I assay utilized in the ongoing High-STEACS (High-Sensitivity Troponin in the Evolution of Patients with Acute Coronary Syndrome) trial uses an MI diagnostic threshold of 16 ng/L in women and 34 ng/L in men. These levels were derived by identifying the 99th percentiles in a healthy reference population composed of nearly 4,600 individuals. In contrast, the conventional troponin I assay utilized for purposes of comparison in High-STEACS employs a threshold of 50 ng/L for both men and women, explained Dr. Mills of the University of Edinburgh.
He reported on 1,126 patients with suspected acute coronary syndrome (ACS), 46% of them women, who presented to the Royal Infirmary of Edinburgh, one of the 10 Scottish medical centers participating in High-STEACS. Subjects’ plasma samples were analyzed using both the Abbott conventional sensitive cardiac troponin I assay and Abbott’s newer high-sensitivity assay, but clinicians received only the results of the conventional assay. The aim of this High-STEACS substudy was to evaluate the effect of gender-specific biomarker thresholds. The final diagnosis of acute MI was determined independently by two cardiologists on the basis of all clinical studies and 30-day outcomes.
Using the conventional assay with a requirement of a troponin level of 50 ng/L or more in both men and women, acute MI was diagnosed in 13% of women and 23% of men. Utilization of the high-sensitivity assay with sex-specific thresholds boosted the rate of diagnosis of MI from 13% to 23% in the women while having little effect in men, where the rate inched up from 23% to 24%.
The diagnostic sensitivity of the conventional assay was 77% in men and 47% in women, while the high-sensitivity assay with gender-specific thresholds had a sensitivity of 86% in men and 95% in women, according to Dr. Mills.
Women with an adjudicated diagnosis of MI were less likely than men to undergo coronary angiography, by a margin of 28% compared with 67%, and they were far less likely to undergo coronary revascularization, by a margin of 18% to 58%. That’s largely because 55 women in the study had a high-sensitivity troponin I level of 17-49 ng/L, above the level required for diagnosis of MI using the sex-specific threshold but below the 50 ng/L requirement with the conventional assay that clinicians used in their patient management decisions.
"The critical question is whether reclassification of these patients as having MI would lead to better treatment and change clinical outcomes. At 6 months, one in four of these patients had a recurrent MI or died, compared with less than 2% of women with a high-sensitivity troponin I of 16 ng/L or less. Those outcomes were similar to or worse than those in women with much larger MIs, who were treated based upon the results of the contemporary assay requiring a level of 50 ng/L or more," the cardiologist noted.
In a previous study, Dr. Mills and his coworkers demonstrated that lowering the diagnostic threshold for acute MI using a previous-generation sensitive cardiac troponin assay in patients with suspected ACS led to a 50% reduction in recurrent MI or death (JAMA 2011;305:1210-5). This was accomplished simply by reclassifying patients as having MI provided their peak troponin exceeded the new, lower threshold.
The hypothesis being tested in High-STEACS is that lowering the biomarker diagnostic threshold for MI still further while utilizing gender-specific cutoffs will result in even better clinical outcomes. High-STEACS is an ongoing randomized trial involving a planned 26,000 Scottish patients with suspected ACS who will be followed for 30 months.
"If improved sensitivity does not impinge on specificity in diagnosis, then clinical outcomes will improve through better targeting of therapies for coronary heart disease. But if increased sensitivity leads to poorer specificity, then misdiagnosis and use of inappropriate therapies may lead to detrimental clinical outcomes," Dr. Mills explained.
Discussant Dr. Eva Swahn of Linkoping (Sweden) University commented that if the results of this High-STEACS substudy showing the value of gender-specific biomarker thresholds hold up, the implications regarding diagnosis and management of MI in women could be great. But elevations in cardiac troponins can be caused by other conditions besides acute MI, including stable angina, renal failure, diabetes, and heart failure, and the substudy design leaves her unconvinced that troponin I elevations in the 17- to 49-ng/L range were necessarily due to MI.
"If you don’t have an MI you shouldn’t be treated as though you do. The management will be completely wrong, and maybe the outcome will be worse," she cautioned.
High-STEACS is funded by the British Heart Foundation. Abbott Diagnostics is supplying the cardiac troponin assay materials. Dr. Mills and Dr. Swahn reported having no financial conflicts.
The under-recognition of heart disease and differences in clinical presentation in women lead to less aggressive treatment strategies and a lower representation of women in clinical trials. Thus, as seen in this study, an improvement in the diagnosis of MI in women using the more sensitive cardiac troponin assay and different gender-based cutoffs can have a meaningful impact in outcomes in our female patients.

|
| Dr. Hiren Shah |
In a recent large meta-analysis of 11 randomized ACS trials it was shown that sex-based differences in 30-day mortality among patients with various manifestations of ACS are largely explained by clinical differences at presentation and the severity of angiographically documented disease (JAMA. 2009;302:874-82.). Thus, new assays that account for gender-based differences may lead to an earlier identification of heart disease in women to address some component of this mortality disparity.
The second phase of this study using these new thresholds will shed light on if this increased capture rate of MI for women leads to improved long term cardiac outcomes, as one hopes that the increased sensitivity seen in the first phase of the study is also balanced with an improvement in specificity. If the results of this and future studies are conclusive, gender-based interpretation of diagnostic tests will lead to earlier diagnosis and improved therapeutic strategies in women.
Dr. Hiren Shah is an assistant professor of medicine in the Feinberg School of Medicine, Northwestern University, Chicago, and a medical director of the Medicine and Cardiac Telemetry Hospitalist Unit at Northwestern Memorial Hospital in Chicago. He is on the advisory board of Hospitalist News.
The under-recognition of heart disease and differences in clinical presentation in women lead to less aggressive treatment strategies and a lower representation of women in clinical trials. Thus, as seen in this study, an improvement in the diagnosis of MI in women using the more sensitive cardiac troponin assay and different gender-based cutoffs can have a meaningful impact in outcomes in our female patients.

|
| Dr. Hiren Shah |
In a recent large meta-analysis of 11 randomized ACS trials it was shown that sex-based differences in 30-day mortality among patients with various manifestations of ACS are largely explained by clinical differences at presentation and the severity of angiographically documented disease (JAMA. 2009;302:874-82.). Thus, new assays that account for gender-based differences may lead to an earlier identification of heart disease in women to address some component of this mortality disparity.
The second phase of this study using these new thresholds will shed light on if this increased capture rate of MI for women leads to improved long term cardiac outcomes, as one hopes that the increased sensitivity seen in the first phase of the study is also balanced with an improvement in specificity. If the results of this and future studies are conclusive, gender-based interpretation of diagnostic tests will lead to earlier diagnosis and improved therapeutic strategies in women.
Dr. Hiren Shah is an assistant professor of medicine in the Feinberg School of Medicine, Northwestern University, Chicago, and a medical director of the Medicine and Cardiac Telemetry Hospitalist Unit at Northwestern Memorial Hospital in Chicago. He is on the advisory board of Hospitalist News.
The under-recognition of heart disease and differences in clinical presentation in women lead to less aggressive treatment strategies and a lower representation of women in clinical trials. Thus, as seen in this study, an improvement in the diagnosis of MI in women using the more sensitive cardiac troponin assay and different gender-based cutoffs can have a meaningful impact in outcomes in our female patients.

|
| Dr. Hiren Shah |
In a recent large meta-analysis of 11 randomized ACS trials it was shown that sex-based differences in 30-day mortality among patients with various manifestations of ACS are largely explained by clinical differences at presentation and the severity of angiographically documented disease (JAMA. 2009;302:874-82.). Thus, new assays that account for gender-based differences may lead to an earlier identification of heart disease in women to address some component of this mortality disparity.
The second phase of this study using these new thresholds will shed light on if this increased capture rate of MI for women leads to improved long term cardiac outcomes, as one hopes that the increased sensitivity seen in the first phase of the study is also balanced with an improvement in specificity. If the results of this and future studies are conclusive, gender-based interpretation of diagnostic tests will lead to earlier diagnosis and improved therapeutic strategies in women.
Dr. Hiren Shah is an assistant professor of medicine in the Feinberg School of Medicine, Northwestern University, Chicago, and a medical director of the Medicine and Cardiac Telemetry Hospitalist Unit at Northwestern Memorial Hospital in Chicago. He is on the advisory board of Hospitalist News.
AMSTERDAM – The use of a high-sensitivity cardiac troponin I assay and gender-specific cutoffs to define acute myocardial infarction nearly doubled the diagnosis of MI in women in an early subanalysis from the High-STEACS trial.
High-sensitivity troponin assays have identified gender differences in the normal reference range. This becomes relevant in light of the third universal definition of acute MI, released last year, which defines the biomarker threshold for the diagnosis of MI as a cardiac troponin at the 99th percentile of a healthy reference population (Eur. Heart J. 2012;33:2551-67). The conventional cardiac troponin assays in widespread use today aren’t sufficiently sensitive to detect gender differences in the normal reference range, so they rely upon a single diagnostic threshold that sets the bar so high it appears to lead to underdiagnosis of MI in women. This likely contributes to gender inequalities in treatment and outcome, Dr. Nicholas L. Mills asserted at the annual congress of the European Society of Cardiology.
The high-sensitivity cardiac troponin I assay utilized in the ongoing High-STEACS (High-Sensitivity Troponin in the Evolution of Patients with Acute Coronary Syndrome) trial uses an MI diagnostic threshold of 16 ng/L in women and 34 ng/L in men. These levels were derived by identifying the 99th percentiles in a healthy reference population composed of nearly 4,600 individuals. In contrast, the conventional troponin I assay utilized for purposes of comparison in High-STEACS employs a threshold of 50 ng/L for both men and women, explained Dr. Mills of the University of Edinburgh.
He reported on 1,126 patients with suspected acute coronary syndrome (ACS), 46% of them women, who presented to the Royal Infirmary of Edinburgh, one of the 10 Scottish medical centers participating in High-STEACS. Subjects’ plasma samples were analyzed using both the Abbott conventional sensitive cardiac troponin I assay and Abbott’s newer high-sensitivity assay, but clinicians received only the results of the conventional assay. The aim of this High-STEACS substudy was to evaluate the effect of gender-specific biomarker thresholds. The final diagnosis of acute MI was determined independently by two cardiologists on the basis of all clinical studies and 30-day outcomes.
Using the conventional assay with a requirement of a troponin level of 50 ng/L or more in both men and women, acute MI was diagnosed in 13% of women and 23% of men. Utilization of the high-sensitivity assay with sex-specific thresholds boosted the rate of diagnosis of MI from 13% to 23% in the women while having little effect in men, where the rate inched up from 23% to 24%.
The diagnostic sensitivity of the conventional assay was 77% in men and 47% in women, while the high-sensitivity assay with gender-specific thresholds had a sensitivity of 86% in men and 95% in women, according to Dr. Mills.
Women with an adjudicated diagnosis of MI were less likely than men to undergo coronary angiography, by a margin of 28% compared with 67%, and they were far less likely to undergo coronary revascularization, by a margin of 18% to 58%. That’s largely because 55 women in the study had a high-sensitivity troponin I level of 17-49 ng/L, above the level required for diagnosis of MI using the sex-specific threshold but below the 50 ng/L requirement with the conventional assay that clinicians used in their patient management decisions.
"The critical question is whether reclassification of these patients as having MI would lead to better treatment and change clinical outcomes. At 6 months, one in four of these patients had a recurrent MI or died, compared with less than 2% of women with a high-sensitivity troponin I of 16 ng/L or less. Those outcomes were similar to or worse than those in women with much larger MIs, who were treated based upon the results of the contemporary assay requiring a level of 50 ng/L or more," the cardiologist noted.
In a previous study, Dr. Mills and his coworkers demonstrated that lowering the diagnostic threshold for acute MI using a previous-generation sensitive cardiac troponin assay in patients with suspected ACS led to a 50% reduction in recurrent MI or death (JAMA 2011;305:1210-5). This was accomplished simply by reclassifying patients as having MI provided their peak troponin exceeded the new, lower threshold.
The hypothesis being tested in High-STEACS is that lowering the biomarker diagnostic threshold for MI still further while utilizing gender-specific cutoffs will result in even better clinical outcomes. High-STEACS is an ongoing randomized trial involving a planned 26,000 Scottish patients with suspected ACS who will be followed for 30 months.
"If improved sensitivity does not impinge on specificity in diagnosis, then clinical outcomes will improve through better targeting of therapies for coronary heart disease. But if increased sensitivity leads to poorer specificity, then misdiagnosis and use of inappropriate therapies may lead to detrimental clinical outcomes," Dr. Mills explained.
Discussant Dr. Eva Swahn of Linkoping (Sweden) University commented that if the results of this High-STEACS substudy showing the value of gender-specific biomarker thresholds hold up, the implications regarding diagnosis and management of MI in women could be great. But elevations in cardiac troponins can be caused by other conditions besides acute MI, including stable angina, renal failure, diabetes, and heart failure, and the substudy design leaves her unconvinced that troponin I elevations in the 17- to 49-ng/L range were necessarily due to MI.
"If you don’t have an MI you shouldn’t be treated as though you do. The management will be completely wrong, and maybe the outcome will be worse," she cautioned.
High-STEACS is funded by the British Heart Foundation. Abbott Diagnostics is supplying the cardiac troponin assay materials. Dr. Mills and Dr. Swahn reported having no financial conflicts.
AMSTERDAM – The use of a high-sensitivity cardiac troponin I assay and gender-specific cutoffs to define acute myocardial infarction nearly doubled the diagnosis of MI in women in an early subanalysis from the High-STEACS trial.
High-sensitivity troponin assays have identified gender differences in the normal reference range. This becomes relevant in light of the third universal definition of acute MI, released last year, which defines the biomarker threshold for the diagnosis of MI as a cardiac troponin at the 99th percentile of a healthy reference population (Eur. Heart J. 2012;33:2551-67). The conventional cardiac troponin assays in widespread use today aren’t sufficiently sensitive to detect gender differences in the normal reference range, so they rely upon a single diagnostic threshold that sets the bar so high it appears to lead to underdiagnosis of MI in women. This likely contributes to gender inequalities in treatment and outcome, Dr. Nicholas L. Mills asserted at the annual congress of the European Society of Cardiology.
The high-sensitivity cardiac troponin I assay utilized in the ongoing High-STEACS (High-Sensitivity Troponin in the Evolution of Patients with Acute Coronary Syndrome) trial uses an MI diagnostic threshold of 16 ng/L in women and 34 ng/L in men. These levels were derived by identifying the 99th percentiles in a healthy reference population composed of nearly 4,600 individuals. In contrast, the conventional troponin I assay utilized for purposes of comparison in High-STEACS employs a threshold of 50 ng/L for both men and women, explained Dr. Mills of the University of Edinburgh.
He reported on 1,126 patients with suspected acute coronary syndrome (ACS), 46% of them women, who presented to the Royal Infirmary of Edinburgh, one of the 10 Scottish medical centers participating in High-STEACS. Subjects’ plasma samples were analyzed using both the Abbott conventional sensitive cardiac troponin I assay and Abbott’s newer high-sensitivity assay, but clinicians received only the results of the conventional assay. The aim of this High-STEACS substudy was to evaluate the effect of gender-specific biomarker thresholds. The final diagnosis of acute MI was determined independently by two cardiologists on the basis of all clinical studies and 30-day outcomes.
Using the conventional assay with a requirement of a troponin level of 50 ng/L or more in both men and women, acute MI was diagnosed in 13% of women and 23% of men. Utilization of the high-sensitivity assay with sex-specific thresholds boosted the rate of diagnosis of MI from 13% to 23% in the women while having little effect in men, where the rate inched up from 23% to 24%.
The diagnostic sensitivity of the conventional assay was 77% in men and 47% in women, while the high-sensitivity assay with gender-specific thresholds had a sensitivity of 86% in men and 95% in women, according to Dr. Mills.
Women with an adjudicated diagnosis of MI were less likely than men to undergo coronary angiography, by a margin of 28% compared with 67%, and they were far less likely to undergo coronary revascularization, by a margin of 18% to 58%. That’s largely because 55 women in the study had a high-sensitivity troponin I level of 17-49 ng/L, above the level required for diagnosis of MI using the sex-specific threshold but below the 50 ng/L requirement with the conventional assay that clinicians used in their patient management decisions.
"The critical question is whether reclassification of these patients as having MI would lead to better treatment and change clinical outcomes. At 6 months, one in four of these patients had a recurrent MI or died, compared with less than 2% of women with a high-sensitivity troponin I of 16 ng/L or less. Those outcomes were similar to or worse than those in women with much larger MIs, who were treated based upon the results of the contemporary assay requiring a level of 50 ng/L or more," the cardiologist noted.
In a previous study, Dr. Mills and his coworkers demonstrated that lowering the diagnostic threshold for acute MI using a previous-generation sensitive cardiac troponin assay in patients with suspected ACS led to a 50% reduction in recurrent MI or death (JAMA 2011;305:1210-5). This was accomplished simply by reclassifying patients as having MI provided their peak troponin exceeded the new, lower threshold.
The hypothesis being tested in High-STEACS is that lowering the biomarker diagnostic threshold for MI still further while utilizing gender-specific cutoffs will result in even better clinical outcomes. High-STEACS is an ongoing randomized trial involving a planned 26,000 Scottish patients with suspected ACS who will be followed for 30 months.
"If improved sensitivity does not impinge on specificity in diagnosis, then clinical outcomes will improve through better targeting of therapies for coronary heart disease. But if increased sensitivity leads to poorer specificity, then misdiagnosis and use of inappropriate therapies may lead to detrimental clinical outcomes," Dr. Mills explained.
Discussant Dr. Eva Swahn of Linkoping (Sweden) University commented that if the results of this High-STEACS substudy showing the value of gender-specific biomarker thresholds hold up, the implications regarding diagnosis and management of MI in women could be great. But elevations in cardiac troponins can be caused by other conditions besides acute MI, including stable angina, renal failure, diabetes, and heart failure, and the substudy design leaves her unconvinced that troponin I elevations in the 17- to 49-ng/L range were necessarily due to MI.
"If you don’t have an MI you shouldn’t be treated as though you do. The management will be completely wrong, and maybe the outcome will be worse," she cautioned.
High-STEACS is funded by the British Heart Foundation. Abbott Diagnostics is supplying the cardiac troponin assay materials. Dr. Mills and Dr. Swahn reported having no financial conflicts.
AT THE ESC CONGRESS 2013
Major finding: The use of a high-sensitivity cardiac troponin I assay with novel gender-specific diagnostic thresholds boosted the MI diagnosis rate in women with suspected ACS from 13% to 23%.
Data source: A substudy of the ongoing prospective High-STEACS trial in 1,126 consecutive men and women who presented with suspected ACS.
Disclosures: High-STEACS is funded by the British Heart Foundation. Abbott Diagnostics is supplying the cardiac troponin assay materials. Dr. Mills and Dr. Swahn reported having no financial conflicts.
Why obesity spells higher cesarean section rates
DENVER – The obesity epidemic has been a major driver of the steep rise in cesarean section rates documented nationally since the year 2000.
The contemporary trend is nicely captured by data on nearly 207,000 pregnancies in the mid-to-late years of the past decade collected by the Consortium on Safe Labor, a group of 19 university and community hospitals in all nine American College of Obstetricians and Gynecologists districts. As prepregnancy body mass index increased, so did the cesarean section rate, Dr. Mark Alanis observed at the annual meeting of the Society of Ob/Gyn Hospitalists.
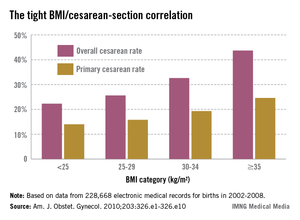
The Consortium used as their highest body mass index (BMI) category those women with a prepregnancy BMI of 35 kg/m2 or more, who made up 21% of their study population. Nationally, 7.6% of the obstetric population is extremely obese as defined by a BMI of 40 kg/m2 or more. The superobese – women with a prepregnancy BMI of 50 kg/m2 or more – make up a smaller subset, but they pose a special challenge for obstetricians. In the three studies that have examined cesarean section rates in superobese patients, one of which was conducted by Dr. Alanis, the rates were 55%, 49%, and 56%.
"It makes you wonder if it’s worth doing a randomized, controlled trial of scheduled primary cesarean section for women with a BMI of 50 kg/m2 or more. Is it more cost effective to try to do labor or just do the cesarean section? We don’t know," he said.
Why do obese women have a higher rate of cesarean section? The answer is beyond dispute, yet not widely appreciated; it’s because obese women tend to have ineffective uterine contractions in early labor, according to Dr. Alanis of the University of Colorado, Denver.
Persuasive evidence exists that greater body weight correlates with slowed cervical dilation. The first stage of labor is prolonged by about 2 hours in obese women. Dystocia occurs before 7 cm.
"You do these cesarean sections between 4 and 7 cm. You don’t do them after 7 cm very often," he noted. "The bottom line is if you can get your obese patient past 7 cm and she’s clearly in active labor, she’ll deliver just as easily as a lean patient."
Compelling data provided by the National Institutes of Health Maternal-Fetal Medicine Units Network show no differences in uterine forces or the length of the second stage of labor depending upon prepregnancy BMI in 5,341 nulliparous women.
"Maternal BMI in nulliparous women reaching the second stage is not associated with a higher incidence of cesarean delivery," according to the investigators (Obstet. Gynecol. 2011;118:1309-13).
This finding confirms earlier work by Australian/New Zealand clinical trialists in a study involving 2,629 nulliparous women who went into labor after 37 weeks’ gestation. Being overweight or obese, respectively, was associated with adjusted 39% and 2.9-fold increased likelihood of cesarean delivery during the first stage of labor, compared with normal-weight women, but no increase in second-stage cesarean delivery (Obstet. Gynecol. 2011;117:1315-22).
These findings of an overall longer duration and slower progression of the early part of the first stage of labor have led to a proposal for adoption of a separate obese labor curve (Obstet. Gynecol. 2012;120:130-5).
"We probably should," according to Dr. Alanis.
The indication for cesarean section in obese patients is disproportionately failure to progress. Fetal distress as a trigger for cesarean delivery is not more common than in normal-weight women.
The explanation for the slower course of first-stage labor in obese women remains unclear, according to Dr. Alanis. Neither smooth muscle content nor contraction strength differs between obese and leaner women.
Dr. Alanis offers vaginal birth after cesarean section to obese patients he considers to have a decent chance of success. But he explains to them that they have a higher risk of uterine rupture, are more likely to develop endometritis, and are less likely to have a successful vaginal delivery than are normal-weight women, based upon data from the Maternal-Fetal Medicine Units Network.
"So approach TOLAC [trial of labor after cesarean] cautiously," he advised.
Dr. Alanis reported having no financial interests germane to his presentation.
DENVER – The obesity epidemic has been a major driver of the steep rise in cesarean section rates documented nationally since the year 2000.
The contemporary trend is nicely captured by data on nearly 207,000 pregnancies in the mid-to-late years of the past decade collected by the Consortium on Safe Labor, a group of 19 university and community hospitals in all nine American College of Obstetricians and Gynecologists districts. As prepregnancy body mass index increased, so did the cesarean section rate, Dr. Mark Alanis observed at the annual meeting of the Society of Ob/Gyn Hospitalists.

The Consortium used as their highest body mass index (BMI) category those women with a prepregnancy BMI of 35 kg/m2 or more, who made up 21% of their study population. Nationally, 7.6% of the obstetric population is extremely obese as defined by a BMI of 40 kg/m2 or more. The superobese – women with a prepregnancy BMI of 50 kg/m2 or more – make up a smaller subset, but they pose a special challenge for obstetricians. In the three studies that have examined cesarean section rates in superobese patients, one of which was conducted by Dr. Alanis, the rates were 55%, 49%, and 56%.
"It makes you wonder if it’s worth doing a randomized, controlled trial of scheduled primary cesarean section for women with a BMI of 50 kg/m2 or more. Is it more cost effective to try to do labor or just do the cesarean section? We don’t know," he said.
Why do obese women have a higher rate of cesarean section? The answer is beyond dispute, yet not widely appreciated; it’s because obese women tend to have ineffective uterine contractions in early labor, according to Dr. Alanis of the University of Colorado, Denver.
Persuasive evidence exists that greater body weight correlates with slowed cervical dilation. The first stage of labor is prolonged by about 2 hours in obese women. Dystocia occurs before 7 cm.
"You do these cesarean sections between 4 and 7 cm. You don’t do them after 7 cm very often," he noted. "The bottom line is if you can get your obese patient past 7 cm and she’s clearly in active labor, she’ll deliver just as easily as a lean patient."
Compelling data provided by the National Institutes of Health Maternal-Fetal Medicine Units Network show no differences in uterine forces or the length of the second stage of labor depending upon prepregnancy BMI in 5,341 nulliparous women.
"Maternal BMI in nulliparous women reaching the second stage is not associated with a higher incidence of cesarean delivery," according to the investigators (Obstet. Gynecol. 2011;118:1309-13).
This finding confirms earlier work by Australian/New Zealand clinical trialists in a study involving 2,629 nulliparous women who went into labor after 37 weeks’ gestation. Being overweight or obese, respectively, was associated with adjusted 39% and 2.9-fold increased likelihood of cesarean delivery during the first stage of labor, compared with normal-weight women, but no increase in second-stage cesarean delivery (Obstet. Gynecol. 2011;117:1315-22).
These findings of an overall longer duration and slower progression of the early part of the first stage of labor have led to a proposal for adoption of a separate obese labor curve (Obstet. Gynecol. 2012;120:130-5).
"We probably should," according to Dr. Alanis.
The indication for cesarean section in obese patients is disproportionately failure to progress. Fetal distress as a trigger for cesarean delivery is not more common than in normal-weight women.
The explanation for the slower course of first-stage labor in obese women remains unclear, according to Dr. Alanis. Neither smooth muscle content nor contraction strength differs between obese and leaner women.
Dr. Alanis offers vaginal birth after cesarean section to obese patients he considers to have a decent chance of success. But he explains to them that they have a higher risk of uterine rupture, are more likely to develop endometritis, and are less likely to have a successful vaginal delivery than are normal-weight women, based upon data from the Maternal-Fetal Medicine Units Network.
"So approach TOLAC [trial of labor after cesarean] cautiously," he advised.
Dr. Alanis reported having no financial interests germane to his presentation.
DENVER – The obesity epidemic has been a major driver of the steep rise in cesarean section rates documented nationally since the year 2000.
The contemporary trend is nicely captured by data on nearly 207,000 pregnancies in the mid-to-late years of the past decade collected by the Consortium on Safe Labor, a group of 19 university and community hospitals in all nine American College of Obstetricians and Gynecologists districts. As prepregnancy body mass index increased, so did the cesarean section rate, Dr. Mark Alanis observed at the annual meeting of the Society of Ob/Gyn Hospitalists.

The Consortium used as their highest body mass index (BMI) category those women with a prepregnancy BMI of 35 kg/m2 or more, who made up 21% of their study population. Nationally, 7.6% of the obstetric population is extremely obese as defined by a BMI of 40 kg/m2 or more. The superobese – women with a prepregnancy BMI of 50 kg/m2 or more – make up a smaller subset, but they pose a special challenge for obstetricians. In the three studies that have examined cesarean section rates in superobese patients, one of which was conducted by Dr. Alanis, the rates were 55%, 49%, and 56%.
"It makes you wonder if it’s worth doing a randomized, controlled trial of scheduled primary cesarean section for women with a BMI of 50 kg/m2 or more. Is it more cost effective to try to do labor or just do the cesarean section? We don’t know," he said.
Why do obese women have a higher rate of cesarean section? The answer is beyond dispute, yet not widely appreciated; it’s because obese women tend to have ineffective uterine contractions in early labor, according to Dr. Alanis of the University of Colorado, Denver.
Persuasive evidence exists that greater body weight correlates with slowed cervical dilation. The first stage of labor is prolonged by about 2 hours in obese women. Dystocia occurs before 7 cm.
"You do these cesarean sections between 4 and 7 cm. You don’t do them after 7 cm very often," he noted. "The bottom line is if you can get your obese patient past 7 cm and she’s clearly in active labor, she’ll deliver just as easily as a lean patient."
Compelling data provided by the National Institutes of Health Maternal-Fetal Medicine Units Network show no differences in uterine forces or the length of the second stage of labor depending upon prepregnancy BMI in 5,341 nulliparous women.
"Maternal BMI in nulliparous women reaching the second stage is not associated with a higher incidence of cesarean delivery," according to the investigators (Obstet. Gynecol. 2011;118:1309-13).
This finding confirms earlier work by Australian/New Zealand clinical trialists in a study involving 2,629 nulliparous women who went into labor after 37 weeks’ gestation. Being overweight or obese, respectively, was associated with adjusted 39% and 2.9-fold increased likelihood of cesarean delivery during the first stage of labor, compared with normal-weight women, but no increase in second-stage cesarean delivery (Obstet. Gynecol. 2011;117:1315-22).
These findings of an overall longer duration and slower progression of the early part of the first stage of labor have led to a proposal for adoption of a separate obese labor curve (Obstet. Gynecol. 2012;120:130-5).
"We probably should," according to Dr. Alanis.
The indication for cesarean section in obese patients is disproportionately failure to progress. Fetal distress as a trigger for cesarean delivery is not more common than in normal-weight women.
The explanation for the slower course of first-stage labor in obese women remains unclear, according to Dr. Alanis. Neither smooth muscle content nor contraction strength differs between obese and leaner women.
Dr. Alanis offers vaginal birth after cesarean section to obese patients he considers to have a decent chance of success. But he explains to them that they have a higher risk of uterine rupture, are more likely to develop endometritis, and are less likely to have a successful vaginal delivery than are normal-weight women, based upon data from the Maternal-Fetal Medicine Units Network.
"So approach TOLAC [trial of labor after cesarean] cautiously," he advised.
Dr. Alanis reported having no financial interests germane to his presentation.
EXPERT ANALYSIS FROM THE SOGH ANNUAL CLINICAL MEETING
Secukinumab soars in phase III psoriasis studies
ISTANBUL, TURKEY – The investigational interleukin-17A inhibitor secukinumab was the talk of Europe’s premier dermatology conference as a result of a series of world-premiere presentations of not one, but three, phase III clinical trials.
The entire secukinumab pivotal phase III program results were presented together at the annual congress of the European Academy of Dermatology and Venereology. Based on these promising results, Novartis plans to file for Food and Drug Administration and European regulatory approval before the end of 2013.
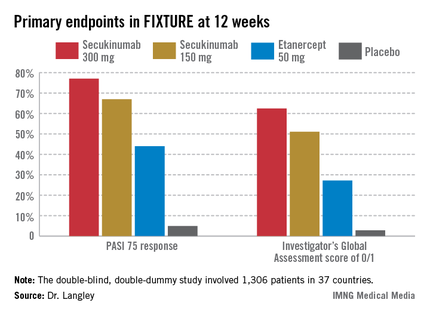
The three randomized, double-blind, multicenter pivotal trials of secukinumab in psoriasis collectively included 3,367 patients with moderate to severe chronic plaque psoriasis. All three studies were strongly positive. Efficacy rates were higher than with current biologics, clinical improvement was remarkably rapid, and the safety profile was reassuring.
"Those of us working in this area of research are really excited by this new data and what it says for the treatment of psoriasis patients," commented Dr. Richard Langley of Dalhousie University, Halifax, N.S.
He presented the centerpiece of this trio of phase III trials, known as the FIXTURE trial. FIXTURE (Full Year Investigative Examination of Secukinumab vs. Etanercept Using 2 Dosing Regimens to Determine Efficacy in Psoriasis) was a 1,306-patient, double-blind, double-dummy trial conducted by 153 investigators in 37 countries. The trial featured a 1-year head-to-head comparison of secukinumab and the widely prescribed tumor necrosis factor (TNF) inhibitor etanercept (Enbrel). The IL-17A inhibitor outperformed etanercept in all of the prespecified primary and secondary outcome measures. Moreover, the two biologics proved equally safe.
"I think it’s interesting in comparing these two molecules that when we look at the data to date, we see comparable safety results. Etanercept is one of the most widely used biologics and has an excellent safety record. That’s reassuring for those of us who are investigating IL-17 signaling," observed Dr. Langley, also the current president of the Canadian Dermatology Association.
FIXTURE participants had a mean baseline Psoriasis Area and Severity Index (PASI) score of 24, an average body mass index of 34 kg/m2, and a mean age in the mid-40s, and were previous inadequate responders to conventional systemic agents and/or biologics. The two co-primary endpoints were 12-week rates of at least a 75% reduction in PASI score, or PASI 75 response, and an Investigator’s Global Assessment (IGA) rating of 0/1, corresponding to clear or nearly clear, on a modified 5-point scale that provides more clinically meaningful information than the older 6-point IGA scale. Patients randomized to the secukinumab 300-mg or 150-mg groups did significantly better on both outcome measures than those assigned to etanercept.
The onset of benefit with secukinumab was swift; a statistically significant improvement compared with etanercept was seen within 2 weeks in terms of IGA scores and within 3 weeks for PASI 75. Secukinumab at 300 mg per subcutaneous injection – the most effective dose across all three pivotal trials – achieved roughly a 50% reduction in PASI scores after 3 weeks, compared with 8 weeks for etanercept, the dermatologist explained.
The PASI 75 response rate at 12 weeks was 77.1% in patients receiving secukinumab at 300 mg, 67% for those receiving secukinumab at 150 mg, 44% for etanercept 50 mg, and 4.9% for placebo.
The highest response rates with secukinumab were seen at week 16 rather than week 12. In the secukinumab 300-mg group, the PASI 75 rate at week 16 was 86.7%, with that high level of response being retained throughout the remainder of the 52-week study. In contrast, the PASI 75 rate at week 16 for etanercept was less than 60%. The IGA 0/1 rate at week 16 was 75.5% in the secukinumab 300-mg group and 60% with secukinumab 150 mg. The peak IGA 0/1 rate in the etanercept arm was only about 45%, and it came much later, at week 26.
Looking at more stringent secondary endpoints, the investigators found that a PASI 90 response was achieved by week 16 in 72.4% of the high-dose secukinumab group, compared with about 30% with etanercept. Twenty-four percent of patients on high-dose secukinumab had a PASI 100 response at week 12, compared with 4% on etanercept. By week 16, 36.8% of the high-dose secukinumab group had a PASI 100 response. At week 52, 65% of the secukinumab 300-mg group maintained a PASI 90 response, compared with 33% of the etanercept group.
The most common adverse event documented in FIXTURE was nasopharyngitis, which occurred in roughly one-third of patients in all four study arms. The serious infection rate was 1%-1.2% in all four study arms. No signals of an increase in malignancies or major adverse cardiovascular events were noted in any of the active treatment groups. Overall, adverse events were similar in both secukinumab arms and comparable to etanercept. Injection-site reactions occurred in 6.1% of the etanercept group and in none of the patients assigned to secukinumab or placebo.
The incidence of treatment-emergent antidrug antibodies in the secukinumab arms was 0.4%, and when it occurred, it had no impact on treatment efficacy or safety. Patients on etanercept weren’t tested for the emergence of antidrug antibodies.
Candida infections occurred in 4.7% of patients on secukinumab 300 mg, 2.3% of those on 150 mg, and 1.2% of the etanercept group. All cases in patients on secukinumab were mild or moderate and easily treated. Candida infections are a side effect of special interest because patients with a genetic defect in the IL-17 pathway can develop chronic mucocutaneous candidiasis. A theoretical concern that pharmacologic inhibition of IL-17 might create a big problem in this regard has not materialized.
The other two phase III secukinumab clinical trials presented in Istanbul – ERASURE and SCULPTURE – showed efficacy and safety results similar to those of FIXTURE. Unlike FIXTURE, neither of those trials featured a head-to-head comparison with another biologic.
These were the first completed phase III studies for any of the three biologics in this novel, highly promising class targeting IL-17 in the treatment of psoriasis and other immune-mediated inflammatory diseases. The other two agents in the pipeline targeting IL-17, brodalumab and ixekizumab, remain in ongoing phase III trials.
Because of the pivotal role IL-17 plays in generating inflammatory cytokines downstream, all three drugs are being studied for additional indications beyond psoriasis. For example, secukinumab, an IgG1 human monoclonal antibody, is in phase III clinical trials for psoriatic arthritis, ankylosing spondylitis, and rheumatoid arthritis, and in phase II for muscular sclerosis and asthma.
One audience member asked Dr. Langley for his thoughts regarding the absence of injection-site reactions in the secukinumab-treated patients. He replied that one obvious factor is that secukinumab injections are given much less frequently: once monthly during maintenance therapy as compared to once weekly with etanercept. In any event, he doesn’t consider injection-site reactions to be clinically relevant.
"Injection-site reactions don’t seem to be a major issue with any of the subcutaneous therapies. It has never in 15 years of using different biologics been an issue that’s made me stop treatment for a patient," he added.
Secukinumab was dosed at baseline, again weekly through week 4, then once monthly from week 8 through the study’s end. Etanercept was dosed at 50 mg twice weekly for 12 weeks, then once weekly in accord with the labeling instructions in most countries. Nonresponders at 12 weeks in the placebo arm were at that point randomized to secukinumab at 300 mg or 150 mg. The statistical analysis in FIXTURE was performed by nonresponder imputation.
"Those who are interested in statistics will know that that’s the most rigorous way that you can look at the data, because anybody who drops out of the study, whether they withdraw consent, have a protocol violation, or have an adverse event, even if they were responding at that time, are considered a failure or nonresponder," Dr. Langley explained.
Asked how he sees secukinumab fitting into patient management, Dr. Langley replied, "Most of us are forced to use conventional therapies and phototherapy to get approval for biologics, but after that I think the safety and efficacy of this drug put it right at the top. I could definitely use this drug as a first-line biologic for patients with plain psoriasis without psoriatic arthritis."
Coinvestigator Dr. Kristian Reich, professor of dermatology at Georg-August University in Göttingen, Germany, said he will probably continue to use TNF inhibitors in psoriasis patients with prominent arthritis, where he believes the TNF inhibitors are particularly beneficial, but he anticipates using secukinumab widely in others.
Dr. Bruce E. Strober, another active clinical trial researcher in psoriasis, said he likes what he sees in the IL-17 inhibitor data.
"I predict as a specialty we’ll become very confident in these drugs 5 years from now. A lot of us will be using them as first line in many patients. That’s my prediction. For now, though, I’m a big fan of TNF inhibition," added Dr. Strober, vice chair of dermatology at the University of Connecticut Medical Center, Farmington.
The secukinumab development program is sponsored by Novartis. Dr. Langley, Dr. Reich, and Dr. Strober disclosed having received research grants from and serving as consultants to Novartis and numerous other companies developing biologic agents for psoriasis.
ISTANBUL, TURKEY – The investigational interleukin-17A inhibitor secukinumab was the talk of Europe’s premier dermatology conference as a result of a series of world-premiere presentations of not one, but three, phase III clinical trials.
The entire secukinumab pivotal phase III program results were presented together at the annual congress of the European Academy of Dermatology and Venereology. Based on these promising results, Novartis plans to file for Food and Drug Administration and European regulatory approval before the end of 2013.

The three randomized, double-blind, multicenter pivotal trials of secukinumab in psoriasis collectively included 3,367 patients with moderate to severe chronic plaque psoriasis. All three studies were strongly positive. Efficacy rates were higher than with current biologics, clinical improvement was remarkably rapid, and the safety profile was reassuring.
"Those of us working in this area of research are really excited by this new data and what it says for the treatment of psoriasis patients," commented Dr. Richard Langley of Dalhousie University, Halifax, N.S.
He presented the centerpiece of this trio of phase III trials, known as the FIXTURE trial. FIXTURE (Full Year Investigative Examination of Secukinumab vs. Etanercept Using 2 Dosing Regimens to Determine Efficacy in Psoriasis) was a 1,306-patient, double-blind, double-dummy trial conducted by 153 investigators in 37 countries. The trial featured a 1-year head-to-head comparison of secukinumab and the widely prescribed tumor necrosis factor (TNF) inhibitor etanercept (Enbrel). The IL-17A inhibitor outperformed etanercept in all of the prespecified primary and secondary outcome measures. Moreover, the two biologics proved equally safe.
"I think it’s interesting in comparing these two molecules that when we look at the data to date, we see comparable safety results. Etanercept is one of the most widely used biologics and has an excellent safety record. That’s reassuring for those of us who are investigating IL-17 signaling," observed Dr. Langley, also the current president of the Canadian Dermatology Association.
FIXTURE participants had a mean baseline Psoriasis Area and Severity Index (PASI) score of 24, an average body mass index of 34 kg/m2, and a mean age in the mid-40s, and were previous inadequate responders to conventional systemic agents and/or biologics. The two co-primary endpoints were 12-week rates of at least a 75% reduction in PASI score, or PASI 75 response, and an Investigator’s Global Assessment (IGA) rating of 0/1, corresponding to clear or nearly clear, on a modified 5-point scale that provides more clinically meaningful information than the older 6-point IGA scale. Patients randomized to the secukinumab 300-mg or 150-mg groups did significantly better on both outcome measures than those assigned to etanercept.
The onset of benefit with secukinumab was swift; a statistically significant improvement compared with etanercept was seen within 2 weeks in terms of IGA scores and within 3 weeks for PASI 75. Secukinumab at 300 mg per subcutaneous injection – the most effective dose across all three pivotal trials – achieved roughly a 50% reduction in PASI scores after 3 weeks, compared with 8 weeks for etanercept, the dermatologist explained.
The PASI 75 response rate at 12 weeks was 77.1% in patients receiving secukinumab at 300 mg, 67% for those receiving secukinumab at 150 mg, 44% for etanercept 50 mg, and 4.9% for placebo.
The highest response rates with secukinumab were seen at week 16 rather than week 12. In the secukinumab 300-mg group, the PASI 75 rate at week 16 was 86.7%, with that high level of response being retained throughout the remainder of the 52-week study. In contrast, the PASI 75 rate at week 16 for etanercept was less than 60%. The IGA 0/1 rate at week 16 was 75.5% in the secukinumab 300-mg group and 60% with secukinumab 150 mg. The peak IGA 0/1 rate in the etanercept arm was only about 45%, and it came much later, at week 26.
Looking at more stringent secondary endpoints, the investigators found that a PASI 90 response was achieved by week 16 in 72.4% of the high-dose secukinumab group, compared with about 30% with etanercept. Twenty-four percent of patients on high-dose secukinumab had a PASI 100 response at week 12, compared with 4% on etanercept. By week 16, 36.8% of the high-dose secukinumab group had a PASI 100 response. At week 52, 65% of the secukinumab 300-mg group maintained a PASI 90 response, compared with 33% of the etanercept group.
The most common adverse event documented in FIXTURE was nasopharyngitis, which occurred in roughly one-third of patients in all four study arms. The serious infection rate was 1%-1.2% in all four study arms. No signals of an increase in malignancies or major adverse cardiovascular events were noted in any of the active treatment groups. Overall, adverse events were similar in both secukinumab arms and comparable to etanercept. Injection-site reactions occurred in 6.1% of the etanercept group and in none of the patients assigned to secukinumab or placebo.
The incidence of treatment-emergent antidrug antibodies in the secukinumab arms was 0.4%, and when it occurred, it had no impact on treatment efficacy or safety. Patients on etanercept weren’t tested for the emergence of antidrug antibodies.
Candida infections occurred in 4.7% of patients on secukinumab 300 mg, 2.3% of those on 150 mg, and 1.2% of the etanercept group. All cases in patients on secukinumab were mild or moderate and easily treated. Candida infections are a side effect of special interest because patients with a genetic defect in the IL-17 pathway can develop chronic mucocutaneous candidiasis. A theoretical concern that pharmacologic inhibition of IL-17 might create a big problem in this regard has not materialized.
The other two phase III secukinumab clinical trials presented in Istanbul – ERASURE and SCULPTURE – showed efficacy and safety results similar to those of FIXTURE. Unlike FIXTURE, neither of those trials featured a head-to-head comparison with another biologic.
These were the first completed phase III studies for any of the three biologics in this novel, highly promising class targeting IL-17 in the treatment of psoriasis and other immune-mediated inflammatory diseases. The other two agents in the pipeline targeting IL-17, brodalumab and ixekizumab, remain in ongoing phase III trials.
Because of the pivotal role IL-17 plays in generating inflammatory cytokines downstream, all three drugs are being studied for additional indications beyond psoriasis. For example, secukinumab, an IgG1 human monoclonal antibody, is in phase III clinical trials for psoriatic arthritis, ankylosing spondylitis, and rheumatoid arthritis, and in phase II for muscular sclerosis and asthma.
One audience member asked Dr. Langley for his thoughts regarding the absence of injection-site reactions in the secukinumab-treated patients. He replied that one obvious factor is that secukinumab injections are given much less frequently: once monthly during maintenance therapy as compared to once weekly with etanercept. In any event, he doesn’t consider injection-site reactions to be clinically relevant.
"Injection-site reactions don’t seem to be a major issue with any of the subcutaneous therapies. It has never in 15 years of using different biologics been an issue that’s made me stop treatment for a patient," he added.
Secukinumab was dosed at baseline, again weekly through week 4, then once monthly from week 8 through the study’s end. Etanercept was dosed at 50 mg twice weekly for 12 weeks, then once weekly in accord with the labeling instructions in most countries. Nonresponders at 12 weeks in the placebo arm were at that point randomized to secukinumab at 300 mg or 150 mg. The statistical analysis in FIXTURE was performed by nonresponder imputation.
"Those who are interested in statistics will know that that’s the most rigorous way that you can look at the data, because anybody who drops out of the study, whether they withdraw consent, have a protocol violation, or have an adverse event, even if they were responding at that time, are considered a failure or nonresponder," Dr. Langley explained.
Asked how he sees secukinumab fitting into patient management, Dr. Langley replied, "Most of us are forced to use conventional therapies and phototherapy to get approval for biologics, but after that I think the safety and efficacy of this drug put it right at the top. I could definitely use this drug as a first-line biologic for patients with plain psoriasis without psoriatic arthritis."
Coinvestigator Dr. Kristian Reich, professor of dermatology at Georg-August University in Göttingen, Germany, said he will probably continue to use TNF inhibitors in psoriasis patients with prominent arthritis, where he believes the TNF inhibitors are particularly beneficial, but he anticipates using secukinumab widely in others.
Dr. Bruce E. Strober, another active clinical trial researcher in psoriasis, said he likes what he sees in the IL-17 inhibitor data.
"I predict as a specialty we’ll become very confident in these drugs 5 years from now. A lot of us will be using them as first line in many patients. That’s my prediction. For now, though, I’m a big fan of TNF inhibition," added Dr. Strober, vice chair of dermatology at the University of Connecticut Medical Center, Farmington.
The secukinumab development program is sponsored by Novartis. Dr. Langley, Dr. Reich, and Dr. Strober disclosed having received research grants from and serving as consultants to Novartis and numerous other companies developing biologic agents for psoriasis.
ISTANBUL, TURKEY – The investigational interleukin-17A inhibitor secukinumab was the talk of Europe’s premier dermatology conference as a result of a series of world-premiere presentations of not one, but three, phase III clinical trials.
The entire secukinumab pivotal phase III program results were presented together at the annual congress of the European Academy of Dermatology and Venereology. Based on these promising results, Novartis plans to file for Food and Drug Administration and European regulatory approval before the end of 2013.

The three randomized, double-blind, multicenter pivotal trials of secukinumab in psoriasis collectively included 3,367 patients with moderate to severe chronic plaque psoriasis. All three studies were strongly positive. Efficacy rates were higher than with current biologics, clinical improvement was remarkably rapid, and the safety profile was reassuring.
"Those of us working in this area of research are really excited by this new data and what it says for the treatment of psoriasis patients," commented Dr. Richard Langley of Dalhousie University, Halifax, N.S.
He presented the centerpiece of this trio of phase III trials, known as the FIXTURE trial. FIXTURE (Full Year Investigative Examination of Secukinumab vs. Etanercept Using 2 Dosing Regimens to Determine Efficacy in Psoriasis) was a 1,306-patient, double-blind, double-dummy trial conducted by 153 investigators in 37 countries. The trial featured a 1-year head-to-head comparison of secukinumab and the widely prescribed tumor necrosis factor (TNF) inhibitor etanercept (Enbrel). The IL-17A inhibitor outperformed etanercept in all of the prespecified primary and secondary outcome measures. Moreover, the two biologics proved equally safe.
"I think it’s interesting in comparing these two molecules that when we look at the data to date, we see comparable safety results. Etanercept is one of the most widely used biologics and has an excellent safety record. That’s reassuring for those of us who are investigating IL-17 signaling," observed Dr. Langley, also the current president of the Canadian Dermatology Association.
FIXTURE participants had a mean baseline Psoriasis Area and Severity Index (PASI) score of 24, an average body mass index of 34 kg/m2, and a mean age in the mid-40s, and were previous inadequate responders to conventional systemic agents and/or biologics. The two co-primary endpoints were 12-week rates of at least a 75% reduction in PASI score, or PASI 75 response, and an Investigator’s Global Assessment (IGA) rating of 0/1, corresponding to clear or nearly clear, on a modified 5-point scale that provides more clinically meaningful information than the older 6-point IGA scale. Patients randomized to the secukinumab 300-mg or 150-mg groups did significantly better on both outcome measures than those assigned to etanercept.
The onset of benefit with secukinumab was swift; a statistically significant improvement compared with etanercept was seen within 2 weeks in terms of IGA scores and within 3 weeks for PASI 75. Secukinumab at 300 mg per subcutaneous injection – the most effective dose across all three pivotal trials – achieved roughly a 50% reduction in PASI scores after 3 weeks, compared with 8 weeks for etanercept, the dermatologist explained.
The PASI 75 response rate at 12 weeks was 77.1% in patients receiving secukinumab at 300 mg, 67% for those receiving secukinumab at 150 mg, 44% for etanercept 50 mg, and 4.9% for placebo.
The highest response rates with secukinumab were seen at week 16 rather than week 12. In the secukinumab 300-mg group, the PASI 75 rate at week 16 was 86.7%, with that high level of response being retained throughout the remainder of the 52-week study. In contrast, the PASI 75 rate at week 16 for etanercept was less than 60%. The IGA 0/1 rate at week 16 was 75.5% in the secukinumab 300-mg group and 60% with secukinumab 150 mg. The peak IGA 0/1 rate in the etanercept arm was only about 45%, and it came much later, at week 26.
Looking at more stringent secondary endpoints, the investigators found that a PASI 90 response was achieved by week 16 in 72.4% of the high-dose secukinumab group, compared with about 30% with etanercept. Twenty-four percent of patients on high-dose secukinumab had a PASI 100 response at week 12, compared with 4% on etanercept. By week 16, 36.8% of the high-dose secukinumab group had a PASI 100 response. At week 52, 65% of the secukinumab 300-mg group maintained a PASI 90 response, compared with 33% of the etanercept group.
The most common adverse event documented in FIXTURE was nasopharyngitis, which occurred in roughly one-third of patients in all four study arms. The serious infection rate was 1%-1.2% in all four study arms. No signals of an increase in malignancies or major adverse cardiovascular events were noted in any of the active treatment groups. Overall, adverse events were similar in both secukinumab arms and comparable to etanercept. Injection-site reactions occurred in 6.1% of the etanercept group and in none of the patients assigned to secukinumab or placebo.
The incidence of treatment-emergent antidrug antibodies in the secukinumab arms was 0.4%, and when it occurred, it had no impact on treatment efficacy or safety. Patients on etanercept weren’t tested for the emergence of antidrug antibodies.
Candida infections occurred in 4.7% of patients on secukinumab 300 mg, 2.3% of those on 150 mg, and 1.2% of the etanercept group. All cases in patients on secukinumab were mild or moderate and easily treated. Candida infections are a side effect of special interest because patients with a genetic defect in the IL-17 pathway can develop chronic mucocutaneous candidiasis. A theoretical concern that pharmacologic inhibition of IL-17 might create a big problem in this regard has not materialized.
The other two phase III secukinumab clinical trials presented in Istanbul – ERASURE and SCULPTURE – showed efficacy and safety results similar to those of FIXTURE. Unlike FIXTURE, neither of those trials featured a head-to-head comparison with another biologic.
These were the first completed phase III studies for any of the three biologics in this novel, highly promising class targeting IL-17 in the treatment of psoriasis and other immune-mediated inflammatory diseases. The other two agents in the pipeline targeting IL-17, brodalumab and ixekizumab, remain in ongoing phase III trials.
Because of the pivotal role IL-17 plays in generating inflammatory cytokines downstream, all three drugs are being studied for additional indications beyond psoriasis. For example, secukinumab, an IgG1 human monoclonal antibody, is in phase III clinical trials for psoriatic arthritis, ankylosing spondylitis, and rheumatoid arthritis, and in phase II for muscular sclerosis and asthma.
One audience member asked Dr. Langley for his thoughts regarding the absence of injection-site reactions in the secukinumab-treated patients. He replied that one obvious factor is that secukinumab injections are given much less frequently: once monthly during maintenance therapy as compared to once weekly with etanercept. In any event, he doesn’t consider injection-site reactions to be clinically relevant.
"Injection-site reactions don’t seem to be a major issue with any of the subcutaneous therapies. It has never in 15 years of using different biologics been an issue that’s made me stop treatment for a patient," he added.
Secukinumab was dosed at baseline, again weekly through week 4, then once monthly from week 8 through the study’s end. Etanercept was dosed at 50 mg twice weekly for 12 weeks, then once weekly in accord with the labeling instructions in most countries. Nonresponders at 12 weeks in the placebo arm were at that point randomized to secukinumab at 300 mg or 150 mg. The statistical analysis in FIXTURE was performed by nonresponder imputation.
"Those who are interested in statistics will know that that’s the most rigorous way that you can look at the data, because anybody who drops out of the study, whether they withdraw consent, have a protocol violation, or have an adverse event, even if they were responding at that time, are considered a failure or nonresponder," Dr. Langley explained.
Asked how he sees secukinumab fitting into patient management, Dr. Langley replied, "Most of us are forced to use conventional therapies and phototherapy to get approval for biologics, but after that I think the safety and efficacy of this drug put it right at the top. I could definitely use this drug as a first-line biologic for patients with plain psoriasis without psoriatic arthritis."
Coinvestigator Dr. Kristian Reich, professor of dermatology at Georg-August University in Göttingen, Germany, said he will probably continue to use TNF inhibitors in psoriasis patients with prominent arthritis, where he believes the TNF inhibitors are particularly beneficial, but he anticipates using secukinumab widely in others.
Dr. Bruce E. Strober, another active clinical trial researcher in psoriasis, said he likes what he sees in the IL-17 inhibitor data.
"I predict as a specialty we’ll become very confident in these drugs 5 years from now. A lot of us will be using them as first line in many patients. That’s my prediction. For now, though, I’m a big fan of TNF inhibition," added Dr. Strober, vice chair of dermatology at the University of Connecticut Medical Center, Farmington.
The secukinumab development program is sponsored by Novartis. Dr. Langley, Dr. Reich, and Dr. Strober disclosed having received research grants from and serving as consultants to Novartis and numerous other companies developing biologic agents for psoriasis.
AT THE EADV CONGRESS
Major finding: The PASI 75 response rate at 12 weeks in patients with moderate to severe chronic plaque psoriasis was 77.1% in those randomized to the investigational interleukin-17A inhibitor secukinumab at 300 mg, 67% for secukinumab at 150 mg, 44% for etanercept 50 mg, and 4.9% for placebo.
Data source: The FIXTURE trial was a randomized, double-blind, double-dummy, year-long, phase III trial involving 1,306 patients with chronic plaque psoriasis not adequately responsive to prior systemic therapies.
Disclosures: The secukinumab development program is sponsored by Novartis. Dr. Langley, Dr. Reich, and Dr. Strober disclosed having received research grants from and serving as consultants to Novartis and numerous other companies developing biologic agents for psoriasis.
Nix new anticoagulants with mechanical valves
AMSTERDAM -- The complete failure of the oral direct thrombin inhibitor dabigatran in the RE-ALIGN trial spells big trouble for efforts to use the novel oral factor Xa inhibitors for anticoagulation in patients with a mechanical heart valve, RE-ALIGN steering committee cochair Dr. Frans Van de Werf cautioned at the annual congress of the European Society of Cardiology.
"It would not be wise for a cardiologist to prescribe these agents for a patient with a mechanical heart valve," declared Dr. Van de Werf, professor and chairman of the department of cardiovascular medicine at the Catholic University at Leuven (Belgium).
Dabigatran (Pradaxa) is approved as a more effective and convenient alternative to warfarin for prevention of stroke and other systemic embolism in patients with nonvalvular atrial fibrillation. RE-ALIGN (Randomized, Phase II Study to Evaluate the Safety and Pharmacokinetics of Oral Dabigatran Etexilate in Patients After Heart Valve Replacement) was an effort to expand the drug's indications to include prevention of thromboembolic events in patients with mechanical heart valves.
The study was halted prematurely late last year when an interim analysis showed that dabigatran was both less safe and less protective than standard warfarin therapy. Shortly afterward, the Food and Drug Administration and the European regulatory agency warned that dabigatran is contraindicated in patients with mechanical heart valves. Dr. Van de Werf's report in Amsterdam, however, was the first presentation of the actual data.
RE-ALIGN was stopped after 252 patients with mechanical heart valves had been randomized 2:1 to dabigatran or warfarin. Dabigatran dosing was adjusted on the basis of creatinine clearance to achieve a trough plasma level of at least 50 ng/mL.
The study halt came in response to an interim analysis showing a 5% stroke rate in the dabigatran group, compared with 0% with warfarin. This was accompanied by a 4% rate of major bleeding with dabigatran, compared with 2% with warfarin, and a 27% rate of any bleeding with dabigatran versus 12% with warfarin. All dabigatran-treated patients with major bleeding had pericardial bleeding and were in the subgroup of participants who started on the direct thrombin inhibitor within 7 days post surgery.
Other unwelcome outcomes in the interim analysis included a 2% MI rate and 3% rate of valve thrombosis without symptoms in the dabigatran group, versus no such events in the warfarin arm.
"Overall, this study was clearly negative: more thrombotic events and more bleeding complications in spite of adjusting the dose of dabigatran based upon renal function," Dr. Van de Werf observed.
Simultaneous with his presentation of the RE-ALIGN findings at the ESC congress in Amsterdam, the results were published online (N. Engl. J. Med. 2013 Sept. 1 [doi:10.1056/NEJMoa1300615]).
In an accompanying editorial, Dr. Elaine M. Hylek of Boston University said the most likely explanation for the disappointing results in RE-ALIGN was that blood levels of dabigatran were inadequate (N. Engl. J. Med. 2013 Sept. 1 [doi:10.1056/NEJMe1310399]).
Dr. Van de Werf, however, rejected that as unlikely. Given that there were excesses of both thromboembolic and bleeding events, other dosing regimens would surely have sent rates of one type of complication or the other even higher, he argued.
Instead, he and his coinvestigators believe that the most likely explanation for the negative results lies in the differences in the mechanisms of action of dabigatran and warfarin. Dabigatran acts near the tail end of the coagulation cascade, on thrombin. In patients with a mechanical heart valve, thrombin is generated by exposure of blood to the artificial surface of the valve leaflets, activating what is known as the contact pathway of coagulation. Thrombin also is generated by the release of tissue factor from tissues injured during the valve surgery. Warfarin acts at both the contact pathway and tissue injury levels; dabigatran doesn't address either.
The novel oral factor Xa inhibitors don't act at the contact pathway and tissue injury levels, either, which is why Dr. Van de Werf cautioned against their off-label use in patients with a mechanical valve.
Discussant Dr. Alec Vahanian said that he "fully agrees" with Dr. Van de Werf's interpretation that the negative results most likely stemmed from dabigatran's absence of efficacy on the contact pathway.
"It sounds from this study that there is no future for dabigatran with this indication. We should not give this agent to a patient with a mechanical prosthesis, and personally I would extend that further and say we should not give this agent to patients with a bioprosthesis or other high-risk patients with severe valve disease. We don't have the evidence for that yet," said Dr. Vahanian, head of cardiology at Bichat Hospital, Paris.
Patients with a mechanical heart valve constitute a challenging population at extreme thromboembolic risk, but Dr. Vahanian offered a comforting prediction: "We are nearing the end of the era of mechanical prosthetic valves," he declared. "Patients are getting older, bioprostheses are getting better, and valve repair techniques are improving."
Dr. Harry R. Buller took issue with Dr. Van de Werf's blanket prediction that the factor Xa inhibitors will prove similarly unsafe and ineffective in patients with mechanical valves, as was dabigatran.
"We still need to find that out. One molecule of factor Xa generates about 1,000 thrombin molecules, so inhibiting coagulation at the level of Xa might actually be effective. What you see with dabigatran is inhibition at the very last stages of the whole coagulation cascade, when there are millions and millions of molecules of thrombin. That's quite difficult. If you do it higher up in the cascade it might work," argued Dr. Buller, chairman of the department of vascular medicine at the Academic Medical Center, Amsterdam.
"Would you take that risk?" challenged Dr. Van de Werf.
"I know there are people taking that risk. I love studies; I would study it," Dr. Buller replied.
Session cochair Dr. Keith A.A. Fox of the University of Edinburgh posed a provocative question to Dr. Van de Werf: "Would you extrapolate these RE-ALIGN data to a patient in atrial fibrillation with a bioprosthetic valve?"
"It's a difficult question," Dr. Van de Werf replied. "In principle, you could give one of the novel anticoagulants approved for atrial fibrillation, including dabigatran, because it is assumed there is no indication for oral anticoagulation for the bioprosthetic valve. I'm not saying you necessarily should do that. If the patient is doing well on warfarin, I don't see the need to switch to one of the new anticoagulants."
RE-ALIGN was supported by Boehringer Ingelheim. Dr. Van de Werf reported receiving research grants and speakers' fees from the company. Dr. Buller, Dr. Fox, and Dr. Vahanian reported no relevant financial interests.
AMSTERDAM -- The complete failure of the oral direct thrombin inhibitor dabigatran in the RE-ALIGN trial spells big trouble for efforts to use the novel oral factor Xa inhibitors for anticoagulation in patients with a mechanical heart valve, RE-ALIGN steering committee cochair Dr. Frans Van de Werf cautioned at the annual congress of the European Society of Cardiology.
"It would not be wise for a cardiologist to prescribe these agents for a patient with a mechanical heart valve," declared Dr. Van de Werf, professor and chairman of the department of cardiovascular medicine at the Catholic University at Leuven (Belgium).
Dabigatran (Pradaxa) is approved as a more effective and convenient alternative to warfarin for prevention of stroke and other systemic embolism in patients with nonvalvular atrial fibrillation. RE-ALIGN (Randomized, Phase II Study to Evaluate the Safety and Pharmacokinetics of Oral Dabigatran Etexilate in Patients After Heart Valve Replacement) was an effort to expand the drug's indications to include prevention of thromboembolic events in patients with mechanical heart valves.
The study was halted prematurely late last year when an interim analysis showed that dabigatran was both less safe and less protective than standard warfarin therapy. Shortly afterward, the Food and Drug Administration and the European regulatory agency warned that dabigatran is contraindicated in patients with mechanical heart valves. Dr. Van de Werf's report in Amsterdam, however, was the first presentation of the actual data.
RE-ALIGN was stopped after 252 patients with mechanical heart valves had been randomized 2:1 to dabigatran or warfarin. Dabigatran dosing was adjusted on the basis of creatinine clearance to achieve a trough plasma level of at least 50 ng/mL.
The study halt came in response to an interim analysis showing a 5% stroke rate in the dabigatran group, compared with 0% with warfarin. This was accompanied by a 4% rate of major bleeding with dabigatran, compared with 2% with warfarin, and a 27% rate of any bleeding with dabigatran versus 12% with warfarin. All dabigatran-treated patients with major bleeding had pericardial bleeding and were in the subgroup of participants who started on the direct thrombin inhibitor within 7 days post surgery.
Other unwelcome outcomes in the interim analysis included a 2% MI rate and 3% rate of valve thrombosis without symptoms in the dabigatran group, versus no such events in the warfarin arm.
"Overall, this study was clearly negative: more thrombotic events and more bleeding complications in spite of adjusting the dose of dabigatran based upon renal function," Dr. Van de Werf observed.
Simultaneous with his presentation of the RE-ALIGN findings at the ESC congress in Amsterdam, the results were published online (N. Engl. J. Med. 2013 Sept. 1 [doi:10.1056/NEJMoa1300615]).
In an accompanying editorial, Dr. Elaine M. Hylek of Boston University said the most likely explanation for the disappointing results in RE-ALIGN was that blood levels of dabigatran were inadequate (N. Engl. J. Med. 2013 Sept. 1 [doi:10.1056/NEJMe1310399]).
Dr. Van de Werf, however, rejected that as unlikely. Given that there were excesses of both thromboembolic and bleeding events, other dosing regimens would surely have sent rates of one type of complication or the other even higher, he argued.
Instead, he and his coinvestigators believe that the most likely explanation for the negative results lies in the differences in the mechanisms of action of dabigatran and warfarin. Dabigatran acts near the tail end of the coagulation cascade, on thrombin. In patients with a mechanical heart valve, thrombin is generated by exposure of blood to the artificial surface of the valve leaflets, activating what is known as the contact pathway of coagulation. Thrombin also is generated by the release of tissue factor from tissues injured during the valve surgery. Warfarin acts at both the contact pathway and tissue injury levels; dabigatran doesn't address either.
The novel oral factor Xa inhibitors don't act at the contact pathway and tissue injury levels, either, which is why Dr. Van de Werf cautioned against their off-label use in patients with a mechanical valve.
Discussant Dr. Alec Vahanian said that he "fully agrees" with Dr. Van de Werf's interpretation that the negative results most likely stemmed from dabigatran's absence of efficacy on the contact pathway.
"It sounds from this study that there is no future for dabigatran with this indication. We should not give this agent to a patient with a mechanical prosthesis, and personally I would extend that further and say we should not give this agent to patients with a bioprosthesis or other high-risk patients with severe valve disease. We don't have the evidence for that yet," said Dr. Vahanian, head of cardiology at Bichat Hospital, Paris.
Patients with a mechanical heart valve constitute a challenging population at extreme thromboembolic risk, but Dr. Vahanian offered a comforting prediction: "We are nearing the end of the era of mechanical prosthetic valves," he declared. "Patients are getting older, bioprostheses are getting better, and valve repair techniques are improving."
Dr. Harry R. Buller took issue with Dr. Van de Werf's blanket prediction that the factor Xa inhibitors will prove similarly unsafe and ineffective in patients with mechanical valves, as was dabigatran.
"We still need to find that out. One molecule of factor Xa generates about 1,000 thrombin molecules, so inhibiting coagulation at the level of Xa might actually be effective. What you see with dabigatran is inhibition at the very last stages of the whole coagulation cascade, when there are millions and millions of molecules of thrombin. That's quite difficult. If you do it higher up in the cascade it might work," argued Dr. Buller, chairman of the department of vascular medicine at the Academic Medical Center, Amsterdam.
"Would you take that risk?" challenged Dr. Van de Werf.
"I know there are people taking that risk. I love studies; I would study it," Dr. Buller replied.
Session cochair Dr. Keith A.A. Fox of the University of Edinburgh posed a provocative question to Dr. Van de Werf: "Would you extrapolate these RE-ALIGN data to a patient in atrial fibrillation with a bioprosthetic valve?"
"It's a difficult question," Dr. Van de Werf replied. "In principle, you could give one of the novel anticoagulants approved for atrial fibrillation, including dabigatran, because it is assumed there is no indication for oral anticoagulation for the bioprosthetic valve. I'm not saying you necessarily should do that. If the patient is doing well on warfarin, I don't see the need to switch to one of the new anticoagulants."
RE-ALIGN was supported by Boehringer Ingelheim. Dr. Van de Werf reported receiving research grants and speakers' fees from the company. Dr. Buller, Dr. Fox, and Dr. Vahanian reported no relevant financial interests.
AMSTERDAM -- The complete failure of the oral direct thrombin inhibitor dabigatran in the RE-ALIGN trial spells big trouble for efforts to use the novel oral factor Xa inhibitors for anticoagulation in patients with a mechanical heart valve, RE-ALIGN steering committee cochair Dr. Frans Van de Werf cautioned at the annual congress of the European Society of Cardiology.
"It would not be wise for a cardiologist to prescribe these agents for a patient with a mechanical heart valve," declared Dr. Van de Werf, professor and chairman of the department of cardiovascular medicine at the Catholic University at Leuven (Belgium).
Dabigatran (Pradaxa) is approved as a more effective and convenient alternative to warfarin for prevention of stroke and other systemic embolism in patients with nonvalvular atrial fibrillation. RE-ALIGN (Randomized, Phase II Study to Evaluate the Safety and Pharmacokinetics of Oral Dabigatran Etexilate in Patients After Heart Valve Replacement) was an effort to expand the drug's indications to include prevention of thromboembolic events in patients with mechanical heart valves.
The study was halted prematurely late last year when an interim analysis showed that dabigatran was both less safe and less protective than standard warfarin therapy. Shortly afterward, the Food and Drug Administration and the European regulatory agency warned that dabigatran is contraindicated in patients with mechanical heart valves. Dr. Van de Werf's report in Amsterdam, however, was the first presentation of the actual data.
RE-ALIGN was stopped after 252 patients with mechanical heart valves had been randomized 2:1 to dabigatran or warfarin. Dabigatran dosing was adjusted on the basis of creatinine clearance to achieve a trough plasma level of at least 50 ng/mL.
The study halt came in response to an interim analysis showing a 5% stroke rate in the dabigatran group, compared with 0% with warfarin. This was accompanied by a 4% rate of major bleeding with dabigatran, compared with 2% with warfarin, and a 27% rate of any bleeding with dabigatran versus 12% with warfarin. All dabigatran-treated patients with major bleeding had pericardial bleeding and were in the subgroup of participants who started on the direct thrombin inhibitor within 7 days post surgery.
Other unwelcome outcomes in the interim analysis included a 2% MI rate and 3% rate of valve thrombosis without symptoms in the dabigatran group, versus no such events in the warfarin arm.
"Overall, this study was clearly negative: more thrombotic events and more bleeding complications in spite of adjusting the dose of dabigatran based upon renal function," Dr. Van de Werf observed.
Simultaneous with his presentation of the RE-ALIGN findings at the ESC congress in Amsterdam, the results were published online (N. Engl. J. Med. 2013 Sept. 1 [doi:10.1056/NEJMoa1300615]).
In an accompanying editorial, Dr. Elaine M. Hylek of Boston University said the most likely explanation for the disappointing results in RE-ALIGN was that blood levels of dabigatran were inadequate (N. Engl. J. Med. 2013 Sept. 1 [doi:10.1056/NEJMe1310399]).
Dr. Van de Werf, however, rejected that as unlikely. Given that there were excesses of both thromboembolic and bleeding events, other dosing regimens would surely have sent rates of one type of complication or the other even higher, he argued.
Instead, he and his coinvestigators believe that the most likely explanation for the negative results lies in the differences in the mechanisms of action of dabigatran and warfarin. Dabigatran acts near the tail end of the coagulation cascade, on thrombin. In patients with a mechanical heart valve, thrombin is generated by exposure of blood to the artificial surface of the valve leaflets, activating what is known as the contact pathway of coagulation. Thrombin also is generated by the release of tissue factor from tissues injured during the valve surgery. Warfarin acts at both the contact pathway and tissue injury levels; dabigatran doesn't address either.
The novel oral factor Xa inhibitors don't act at the contact pathway and tissue injury levels, either, which is why Dr. Van de Werf cautioned against their off-label use in patients with a mechanical valve.
Discussant Dr. Alec Vahanian said that he "fully agrees" with Dr. Van de Werf's interpretation that the negative results most likely stemmed from dabigatran's absence of efficacy on the contact pathway.
"It sounds from this study that there is no future for dabigatran with this indication. We should not give this agent to a patient with a mechanical prosthesis, and personally I would extend that further and say we should not give this agent to patients with a bioprosthesis or other high-risk patients with severe valve disease. We don't have the evidence for that yet," said Dr. Vahanian, head of cardiology at Bichat Hospital, Paris.
Patients with a mechanical heart valve constitute a challenging population at extreme thromboembolic risk, but Dr. Vahanian offered a comforting prediction: "We are nearing the end of the era of mechanical prosthetic valves," he declared. "Patients are getting older, bioprostheses are getting better, and valve repair techniques are improving."
Dr. Harry R. Buller took issue with Dr. Van de Werf's blanket prediction that the factor Xa inhibitors will prove similarly unsafe and ineffective in patients with mechanical valves, as was dabigatran.
"We still need to find that out. One molecule of factor Xa generates about 1,000 thrombin molecules, so inhibiting coagulation at the level of Xa might actually be effective. What you see with dabigatran is inhibition at the very last stages of the whole coagulation cascade, when there are millions and millions of molecules of thrombin. That's quite difficult. If you do it higher up in the cascade it might work," argued Dr. Buller, chairman of the department of vascular medicine at the Academic Medical Center, Amsterdam.
"Would you take that risk?" challenged Dr. Van de Werf.
"I know there are people taking that risk. I love studies; I would study it," Dr. Buller replied.
Session cochair Dr. Keith A.A. Fox of the University of Edinburgh posed a provocative question to Dr. Van de Werf: "Would you extrapolate these RE-ALIGN data to a patient in atrial fibrillation with a bioprosthetic valve?"
"It's a difficult question," Dr. Van de Werf replied. "In principle, you could give one of the novel anticoagulants approved for atrial fibrillation, including dabigatran, because it is assumed there is no indication for oral anticoagulation for the bioprosthetic valve. I'm not saying you necessarily should do that. If the patient is doing well on warfarin, I don't see the need to switch to one of the new anticoagulants."
RE-ALIGN was supported by Boehringer Ingelheim. Dr. Van de Werf reported receiving research grants and speakers' fees from the company. Dr. Buller, Dr. Fox, and Dr. Vahanian reported no relevant financial interests.
Psoriasis linked to nonalcoholic fatty liver disease
ISTANBUL, TURKEY – The prevalence of psoriasis was significantly greater in subjects with nonalcoholic fatty liver disease by a margin of 7% to 4.2%, according to findings from a long-term, population-based cohort study including nearly 4,000 individuals.
This newly identified link between two common diseases – one of which manifests as visible, angry skin lesions; the other silent, insidious, and often unrecognized until its late stages – contains an important message for clinicians, said Dr. Ella van der Voort of Erasmus University in Rotterdam, the Netherlands.
"Physicians should be aware of this increased risk before prescribing for psoriasis patients medications with potential for liver toxicity – and I’m not talking about just psoriasis medications, but also medications in general, like statins," she said at the annual congress of the European Academy of Dermatology and Venereology.
Psoriasis was present in 5.1% of the study group. Most cases were mild, with affected patients having a mean psoriasis area severity index of 2.9 out of a possible 72 and no use of systemic psoriasis therapies. Nevertheless, the prevalence of nonalcoholic fatty liver disease (NAFLD) was 46.2% among the 118 psoriasis patients, compared with 33% in the 2,174 subjects without psoriasis.
NAFLD is now the most common form of chronic liver disease in Western countries. By 2020, when new well-tolerated and highly effective medications for hepatitis C are expected to have made a mark, NAFLD may be the leading reason for liver transplantation in developed countries, Dr. van der Voort observed.
The Rotterdam Study is designed to examine contributors to chronic diseases in the elderly, including cardiovascular, endocrinologic, psychiatric, neurologic, and eye diseases. The first wave of participants enrolled in 1989-93. Dr. van der Voort’s study focused on the third cohort, consisting of 3,932 elderly Rotterdam-area residents enrolled in 2006-2008. The mean age at enrollment was 76 years, and the ongoing routine evaluation of the cohort includes liver ultrasound, lab tests for liver enzyme levels and serologic evidence of hepatitis, and a skin examination.
Dr. van der Voort’s analysis was restricted to the 2,292 third-wave subjects who were not heavy drinkers and didn’t have chronic hepatitis, either of which rules out the diagnosis of NAFLD. The participants were not taking hepatotoxic medications, and there was no uncertainty as to the skin examination findings.
The hallmark of NAFLD is the accumulation of triglycerides in the hepatocytes. The previously well-described risk factors for NAFLD include the metabolic syndrome and its individual components, as well as sedentary lifestyle, smoking, and a poor-quality diet. There is also a genetic component: individuals with a mutation in the phospholipase domain-containing protein 3 (PNPLA3) gene, which codes for an enzyme functioning in hydrolyzation of triglycerides, are predisposed to NAFLD.
The prevalence of metabolic syndrome was 62% in subjects with psoriasis and 52% in those without psoriasis. Individuals with psoriasis also were more likely to be smokers by a margin of 15% to 8%. After adjusting for those variables, as well as alcohol intake, age, gender, liver enzyme levels, and other potential confounders, two strong independent predictors of NAFLD were metabolic syndrome, with an adjusted odds ratio of 3.5, and psoriasis, with an odds ratio of 1.7.
In a multivariate analysis, psoriasis was not only independently associated with a 70% increased likelihood of NAFLD, but psoriasis patients with NAFLD were also 60% more likely to have the more severe forms of NAFLD.
The Rotterdam study is funded chiefly by Dutch scientific research foundations, with secondary pharmaceutical industry support. Dr. van der Voort reported having no financial conflicts of interest.
ISTANBUL, TURKEY – The prevalence of psoriasis was significantly greater in subjects with nonalcoholic fatty liver disease by a margin of 7% to 4.2%, according to findings from a long-term, population-based cohort study including nearly 4,000 individuals.
This newly identified link between two common diseases – one of which manifests as visible, angry skin lesions; the other silent, insidious, and often unrecognized until its late stages – contains an important message for clinicians, said Dr. Ella van der Voort of Erasmus University in Rotterdam, the Netherlands.
"Physicians should be aware of this increased risk before prescribing for psoriasis patients medications with potential for liver toxicity – and I’m not talking about just psoriasis medications, but also medications in general, like statins," she said at the annual congress of the European Academy of Dermatology and Venereology.
Psoriasis was present in 5.1% of the study group. Most cases were mild, with affected patients having a mean psoriasis area severity index of 2.9 out of a possible 72 and no use of systemic psoriasis therapies. Nevertheless, the prevalence of nonalcoholic fatty liver disease (NAFLD) was 46.2% among the 118 psoriasis patients, compared with 33% in the 2,174 subjects without psoriasis.
NAFLD is now the most common form of chronic liver disease in Western countries. By 2020, when new well-tolerated and highly effective medications for hepatitis C are expected to have made a mark, NAFLD may be the leading reason for liver transplantation in developed countries, Dr. van der Voort observed.
The Rotterdam Study is designed to examine contributors to chronic diseases in the elderly, including cardiovascular, endocrinologic, psychiatric, neurologic, and eye diseases. The first wave of participants enrolled in 1989-93. Dr. van der Voort’s study focused on the third cohort, consisting of 3,932 elderly Rotterdam-area residents enrolled in 2006-2008. The mean age at enrollment was 76 years, and the ongoing routine evaluation of the cohort includes liver ultrasound, lab tests for liver enzyme levels and serologic evidence of hepatitis, and a skin examination.
Dr. van der Voort’s analysis was restricted to the 2,292 third-wave subjects who were not heavy drinkers and didn’t have chronic hepatitis, either of which rules out the diagnosis of NAFLD. The participants were not taking hepatotoxic medications, and there was no uncertainty as to the skin examination findings.
The hallmark of NAFLD is the accumulation of triglycerides in the hepatocytes. The previously well-described risk factors for NAFLD include the metabolic syndrome and its individual components, as well as sedentary lifestyle, smoking, and a poor-quality diet. There is also a genetic component: individuals with a mutation in the phospholipase domain-containing protein 3 (PNPLA3) gene, which codes for an enzyme functioning in hydrolyzation of triglycerides, are predisposed to NAFLD.
The prevalence of metabolic syndrome was 62% in subjects with psoriasis and 52% in those without psoriasis. Individuals with psoriasis also were more likely to be smokers by a margin of 15% to 8%. After adjusting for those variables, as well as alcohol intake, age, gender, liver enzyme levels, and other potential confounders, two strong independent predictors of NAFLD were metabolic syndrome, with an adjusted odds ratio of 3.5, and psoriasis, with an odds ratio of 1.7.
In a multivariate analysis, psoriasis was not only independently associated with a 70% increased likelihood of NAFLD, but psoriasis patients with NAFLD were also 60% more likely to have the more severe forms of NAFLD.
The Rotterdam study is funded chiefly by Dutch scientific research foundations, with secondary pharmaceutical industry support. Dr. van der Voort reported having no financial conflicts of interest.
ISTANBUL, TURKEY – The prevalence of psoriasis was significantly greater in subjects with nonalcoholic fatty liver disease by a margin of 7% to 4.2%, according to findings from a long-term, population-based cohort study including nearly 4,000 individuals.
This newly identified link between two common diseases – one of which manifests as visible, angry skin lesions; the other silent, insidious, and often unrecognized until its late stages – contains an important message for clinicians, said Dr. Ella van der Voort of Erasmus University in Rotterdam, the Netherlands.
"Physicians should be aware of this increased risk before prescribing for psoriasis patients medications with potential for liver toxicity – and I’m not talking about just psoriasis medications, but also medications in general, like statins," she said at the annual congress of the European Academy of Dermatology and Venereology.
Psoriasis was present in 5.1% of the study group. Most cases were mild, with affected patients having a mean psoriasis area severity index of 2.9 out of a possible 72 and no use of systemic psoriasis therapies. Nevertheless, the prevalence of nonalcoholic fatty liver disease (NAFLD) was 46.2% among the 118 psoriasis patients, compared with 33% in the 2,174 subjects without psoriasis.
NAFLD is now the most common form of chronic liver disease in Western countries. By 2020, when new well-tolerated and highly effective medications for hepatitis C are expected to have made a mark, NAFLD may be the leading reason for liver transplantation in developed countries, Dr. van der Voort observed.
The Rotterdam Study is designed to examine contributors to chronic diseases in the elderly, including cardiovascular, endocrinologic, psychiatric, neurologic, and eye diseases. The first wave of participants enrolled in 1989-93. Dr. van der Voort’s study focused on the third cohort, consisting of 3,932 elderly Rotterdam-area residents enrolled in 2006-2008. The mean age at enrollment was 76 years, and the ongoing routine evaluation of the cohort includes liver ultrasound, lab tests for liver enzyme levels and serologic evidence of hepatitis, and a skin examination.
Dr. van der Voort’s analysis was restricted to the 2,292 third-wave subjects who were not heavy drinkers and didn’t have chronic hepatitis, either of which rules out the diagnosis of NAFLD. The participants were not taking hepatotoxic medications, and there was no uncertainty as to the skin examination findings.
The hallmark of NAFLD is the accumulation of triglycerides in the hepatocytes. The previously well-described risk factors for NAFLD include the metabolic syndrome and its individual components, as well as sedentary lifestyle, smoking, and a poor-quality diet. There is also a genetic component: individuals with a mutation in the phospholipase domain-containing protein 3 (PNPLA3) gene, which codes for an enzyme functioning in hydrolyzation of triglycerides, are predisposed to NAFLD.
The prevalence of metabolic syndrome was 62% in subjects with psoriasis and 52% in those without psoriasis. Individuals with psoriasis also were more likely to be smokers by a margin of 15% to 8%. After adjusting for those variables, as well as alcohol intake, age, gender, liver enzyme levels, and other potential confounders, two strong independent predictors of NAFLD were metabolic syndrome, with an adjusted odds ratio of 3.5, and psoriasis, with an odds ratio of 1.7.
In a multivariate analysis, psoriasis was not only independently associated with a 70% increased likelihood of NAFLD, but psoriasis patients with NAFLD were also 60% more likely to have the more severe forms of NAFLD.
The Rotterdam study is funded chiefly by Dutch scientific research foundations, with secondary pharmaceutical industry support. Dr. van der Voort reported having no financial conflicts of interest.
AT THE EADV CONGRESS
Major finding: The prevalence of nonalcoholic fatty liver disease in a large cohort of elderly subjects was 46.2% among those with psoriasis and 33% in those without. In a multivariate regression analysis, psoriasis was independently associated with a 70% increased likelihood of the hepatic disease.
Data source: The Rotterdam Study is a long-standing prospective, population-based cohort study. The aim is to determine key factors in the development of a range of chronic diseases common in the elderly.
Disclosures: The Rotterdam Study is funded primarily by Dutch scientific research foundations. The presenter reported having no financial conflicts.