User login
AAAAI: Albuterol dry powder inhaler offers simplified approach for young kids
LOS ANGELES – Young asthmatic children on bronchodilator therapy may soon gain access to a novel albuterol multidose dry powder inhaler that’s already proved popular with teen and adult patients with reversible obstructive airway disease because of its ease of use.
A phase III randomized, double-blind multicenter trial of the albuterol multidose dry powder inhaler (MDPI) versus placebo in 184 asthmatic children aged 4-11 years not on systemic corticosteroids met its primary and secondary lung function endpoints, with safety and tolerability similar to placebo, Dr. Tushar P. Shah reported at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
The albuterol MDPI is already marketed by Teva Pharmaceuticals as the ProAir RespiClick in patients aged 12 and older. The purpose of this phase III clinical trial was to obtain an expanded indication in 4- to 11-year-olds. The company has submitted its request to the Food and Drug Administration and anticipates smooth sailing based upon the new data, according to Dr. Shah, senior vice president for global respiratory research and development at Teva in Frazer, Pa.
The albuterol MDPI fills an unmet need for a simplified approach to rescue medication, the allergist said in an interview.
“This is a breath-actuated inhaler. Many patients – especially kids – have a hard time coordinating a conventional multidose inhaler actuation with inhalation. They have trouble getting the timing right, so the drug doesn’t get to the distal lung. That’s why this albuterol MDPI has been very well received in adults. For kids, I think it’s going to be even better because this is a very simple and intuitive device. All they do is open the cap, inhale, [and] close the cap,” he explained.
The young study participants used the albuterol MDPI at two inhalations four times daily, with a total daily albuterol dose of 720 mcg.
The primary study endpoint was the short-term improvement in lung function seen during testing performed after the very first study dose and again after the final dose of medication 3 weeks later. This was expressed as the area under the baseline-adjusted percent-predicted forced expiratory volume in 1 second effect-time curve from predose to 6 hours post dose. On both occasions, a sharp jump in opening of the airways was demonstrated within 5 minutes of dosing, with the effect remaining significantly better than with placebo for more than 2 hours.
Moreover, the maximum change from baseline in peak expiratory flow rate seen within 2 hours after dosing was a 26% increase with the albuterol MDPI, a significantly better result than the 14% increase with placebo.
No adverse events attributable to the study drug were seen.
The study was sponsored by Teva Pharmaceuticals. The presenter is a senior company employee.
LOS ANGELES – Young asthmatic children on bronchodilator therapy may soon gain access to a novel albuterol multidose dry powder inhaler that’s already proved popular with teen and adult patients with reversible obstructive airway disease because of its ease of use.
A phase III randomized, double-blind multicenter trial of the albuterol multidose dry powder inhaler (MDPI) versus placebo in 184 asthmatic children aged 4-11 years not on systemic corticosteroids met its primary and secondary lung function endpoints, with safety and tolerability similar to placebo, Dr. Tushar P. Shah reported at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
The albuterol MDPI is already marketed by Teva Pharmaceuticals as the ProAir RespiClick in patients aged 12 and older. The purpose of this phase III clinical trial was to obtain an expanded indication in 4- to 11-year-olds. The company has submitted its request to the Food and Drug Administration and anticipates smooth sailing based upon the new data, according to Dr. Shah, senior vice president for global respiratory research and development at Teva in Frazer, Pa.
The albuterol MDPI fills an unmet need for a simplified approach to rescue medication, the allergist said in an interview.
“This is a breath-actuated inhaler. Many patients – especially kids – have a hard time coordinating a conventional multidose inhaler actuation with inhalation. They have trouble getting the timing right, so the drug doesn’t get to the distal lung. That’s why this albuterol MDPI has been very well received in adults. For kids, I think it’s going to be even better because this is a very simple and intuitive device. All they do is open the cap, inhale, [and] close the cap,” he explained.
The young study participants used the albuterol MDPI at two inhalations four times daily, with a total daily albuterol dose of 720 mcg.
The primary study endpoint was the short-term improvement in lung function seen during testing performed after the very first study dose and again after the final dose of medication 3 weeks later. This was expressed as the area under the baseline-adjusted percent-predicted forced expiratory volume in 1 second effect-time curve from predose to 6 hours post dose. On both occasions, a sharp jump in opening of the airways was demonstrated within 5 minutes of dosing, with the effect remaining significantly better than with placebo for more than 2 hours.
Moreover, the maximum change from baseline in peak expiratory flow rate seen within 2 hours after dosing was a 26% increase with the albuterol MDPI, a significantly better result than the 14% increase with placebo.
No adverse events attributable to the study drug were seen.
The study was sponsored by Teva Pharmaceuticals. The presenter is a senior company employee.
LOS ANGELES – Young asthmatic children on bronchodilator therapy may soon gain access to a novel albuterol multidose dry powder inhaler that’s already proved popular with teen and adult patients with reversible obstructive airway disease because of its ease of use.
A phase III randomized, double-blind multicenter trial of the albuterol multidose dry powder inhaler (MDPI) versus placebo in 184 asthmatic children aged 4-11 years not on systemic corticosteroids met its primary and secondary lung function endpoints, with safety and tolerability similar to placebo, Dr. Tushar P. Shah reported at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
The albuterol MDPI is already marketed by Teva Pharmaceuticals as the ProAir RespiClick in patients aged 12 and older. The purpose of this phase III clinical trial was to obtain an expanded indication in 4- to 11-year-olds. The company has submitted its request to the Food and Drug Administration and anticipates smooth sailing based upon the new data, according to Dr. Shah, senior vice president for global respiratory research and development at Teva in Frazer, Pa.
The albuterol MDPI fills an unmet need for a simplified approach to rescue medication, the allergist said in an interview.
“This is a breath-actuated inhaler. Many patients – especially kids – have a hard time coordinating a conventional multidose inhaler actuation with inhalation. They have trouble getting the timing right, so the drug doesn’t get to the distal lung. That’s why this albuterol MDPI has been very well received in adults. For kids, I think it’s going to be even better because this is a very simple and intuitive device. All they do is open the cap, inhale, [and] close the cap,” he explained.
The young study participants used the albuterol MDPI at two inhalations four times daily, with a total daily albuterol dose of 720 mcg.
The primary study endpoint was the short-term improvement in lung function seen during testing performed after the very first study dose and again after the final dose of medication 3 weeks later. This was expressed as the area under the baseline-adjusted percent-predicted forced expiratory volume in 1 second effect-time curve from predose to 6 hours post dose. On both occasions, a sharp jump in opening of the airways was demonstrated within 5 minutes of dosing, with the effect remaining significantly better than with placebo for more than 2 hours.
Moreover, the maximum change from baseline in peak expiratory flow rate seen within 2 hours after dosing was a 26% increase with the albuterol MDPI, a significantly better result than the 14% increase with placebo.
No adverse events attributable to the study drug were seen.
The study was sponsored by Teva Pharmaceuticals. The presenter is a senior company employee.
AT 2016 AAAAI ANNUAL MEETING
Key clinical point: The albuterol multidose dry powder inhaler that’s now indicated for asthma patients aged 12 years and older may become available to those aged 4-11 years.
Major finding: Lung function measurements improved sharply within 5 minutes after dosing with this bronchodilator used for acute symptom relief, with the effect lasting for longer than 2 hours.
Data source: A phase III, double-blind, multicenter, placebo-controlled randomized trial involving 184 asthmatic children aged 4-11 years.
Disclosures: The study was sponsored by Teva Pharmaceuticals. The presenter is a senior company employee.
First Sublingual Immunotherapy Tablet for House Dust Mite Allergic Rhinitis May Be US-bound
LOS ANGELES – A sublingual immunotherapy tablet for house dust mite–induced allergic rhinitis achieved clinically meaningful improvement in symptoms along with less use of rescue medications and a favorable safety profile in a pivotal phase III trial, Dr. Hendrik Nolte reported at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
The double-blind, 52-week randomized trial included 1,482 North American adults and adolescents.
“This was the largest clinical trial ever conducted with sublingual therapy in North America, and the first successful North American trial of a house dust mite sublingual immunotherapy tablet. It confirms results from previous large European trials,” noted Dr. Nolte of Merck, which is collaborating with the Danish company ALK in developing this therapy.
The sublingual immunotherapy (SLIT) tablet, known for now as the 12 SQ HDM SLIT-tablet, has been approved by European regulatory authorities for treatment of adults with house dust mite (HDM) allergic rhinitis or HDM allergic asthma. Based upon the positive findings in the phase III U.S. trial, Merck applied in February 2016 to the U.S. Food and Drug Administration for approval of the tablet as a biologic agent in patients aged 12 years or older.
HDM is the most common indoor allergen in the world. Unlike pollen and ragweed allergies, it’s not a seasonal problem. Moreover, HDM-induced allergic rhinitis is associated with increased risk of asthma. Although HDM allergic rhinitis can be treated symptomatically with oral antihistamines and nasal steroids, allergy immunotherapy has the appeal of addressing the underlying disease mechanism and potentially altering the long-term course. SLIT using a highly standardized HDM allergen extract offers a major advantage over traditional allergy immunotherapy via a lengthy program of subcutaneous injections – namely, the convenience of home self-administration.
The primary endpoint in the U.S. pivotal trial was the total combined rhinitis score (TCRS), which is the sum of the daily rhinitis symptom score and the daily rescue medication usage score averaged over the last 8 weeks of the year-long study. The FDA has set the bar for establishing clinically meaningful improvement: it requires demonstration of a reduction in the TCRS of at least 15%, compared with placebo. The 12 SQ HDM SLIT-tablet trial exceeded this standard, achieving a 17% reduction. The study also met its key prespecified secondary endpoints, including a 16% reduction in the daily rhinosinusitis symptom score and an 18% decrease in the daily medication score, compared with placebo.
Participants in the trial had a mean 18-year history of allergic rhinitis with or without conjunctivitis. Seventy-five percent of them were sensitized to other common allergens in addition to HDM, and 31% of subjects had comorbid asthma. The HDM SLIT therapy was equally effective in asthmatics and nonasthmatics, in patients allergic only to HDM and those who were polysensitized, and in subjects with and without conjunctivitis, according to Dr. Nolte.
There were no serious adverse events related to the 12 SQ HDM SLIT-tablet. A total of 9.8% of patients discontinued SLIT because of treatment-emergent adverse events, chiefly mild-to-moderate throat irritation, mouth swelling, or itchiness of the mouth or ears.
“Importantly, these events were very transient. They occurred typically within the first 8 days and lasted 14-67 hours, with a median 1-day duration of rescue medication,” he said.
Symptomatic improvement was typically seen beginning at 8-12 weeks. Adults were free to take the once-daily tablet anytime during the day. The pediatric patients were advised not to do so in the morning because they wouldn’t be under observation while on a school bus.
The tablet is based upon a formulation of allergen extracts from the two major species of HDM: Dermatophagoides pterornyssinus and D. farinae. More than 90% of HDM-sensitized patients are sensitized to both. A highly standardized manufacturing process ensures that the tablet contains 50 mcg of the four major HDM allergens in equal ratio – Der f1, Der p2, Der p1, and Der f 2 – plus other components.
In response to audience questions, Dr. Nolte said the company had tried incorporating an antihistamine into the tablet to block adverse reactions but it didn’t work. Adverse events typically occur within a couple of minutes after taking the tablet, and antihistamines are far too slow-acting to help.
“You can premedicate with antihistamines. We know European investigators and clinicians who are doing it. But I would not recommend it personally, because these tablets are taken at home after the first administration in the office, and I think it’s important that the patient has a good feel for what happens over the following days. There is a potential risk of masking side effects with premedication, which could be a concern,” Dr. Nolte said.
Merck already has two FDA-approved SLIT tablets developed with ALK on the U.S. market: Grastek, for treatment of grass pollen–induced allergic rhinitis in children and adults, and Ragwitek, for ragweed-induced allergic disease in adults.
The trial was sponsored by Merck and presented by a full-time company employee.
LOS ANGELES – A sublingual immunotherapy tablet for house dust mite–induced allergic rhinitis achieved clinically meaningful improvement in symptoms along with less use of rescue medications and a favorable safety profile in a pivotal phase III trial, Dr. Hendrik Nolte reported at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
The double-blind, 52-week randomized trial included 1,482 North American adults and adolescents.
“This was the largest clinical trial ever conducted with sublingual therapy in North America, and the first successful North American trial of a house dust mite sublingual immunotherapy tablet. It confirms results from previous large European trials,” noted Dr. Nolte of Merck, which is collaborating with the Danish company ALK in developing this therapy.
The sublingual immunotherapy (SLIT) tablet, known for now as the 12 SQ HDM SLIT-tablet, has been approved by European regulatory authorities for treatment of adults with house dust mite (HDM) allergic rhinitis or HDM allergic asthma. Based upon the positive findings in the phase III U.S. trial, Merck applied in February 2016 to the U.S. Food and Drug Administration for approval of the tablet as a biologic agent in patients aged 12 years or older.
HDM is the most common indoor allergen in the world. Unlike pollen and ragweed allergies, it’s not a seasonal problem. Moreover, HDM-induced allergic rhinitis is associated with increased risk of asthma. Although HDM allergic rhinitis can be treated symptomatically with oral antihistamines and nasal steroids, allergy immunotherapy has the appeal of addressing the underlying disease mechanism and potentially altering the long-term course. SLIT using a highly standardized HDM allergen extract offers a major advantage over traditional allergy immunotherapy via a lengthy program of subcutaneous injections – namely, the convenience of home self-administration.
The primary endpoint in the U.S. pivotal trial was the total combined rhinitis score (TCRS), which is the sum of the daily rhinitis symptom score and the daily rescue medication usage score averaged over the last 8 weeks of the year-long study. The FDA has set the bar for establishing clinically meaningful improvement: it requires demonstration of a reduction in the TCRS of at least 15%, compared with placebo. The 12 SQ HDM SLIT-tablet trial exceeded this standard, achieving a 17% reduction. The study also met its key prespecified secondary endpoints, including a 16% reduction in the daily rhinosinusitis symptom score and an 18% decrease in the daily medication score, compared with placebo.
Participants in the trial had a mean 18-year history of allergic rhinitis with or without conjunctivitis. Seventy-five percent of them were sensitized to other common allergens in addition to HDM, and 31% of subjects had comorbid asthma. The HDM SLIT therapy was equally effective in asthmatics and nonasthmatics, in patients allergic only to HDM and those who were polysensitized, and in subjects with and without conjunctivitis, according to Dr. Nolte.
There were no serious adverse events related to the 12 SQ HDM SLIT-tablet. A total of 9.8% of patients discontinued SLIT because of treatment-emergent adverse events, chiefly mild-to-moderate throat irritation, mouth swelling, or itchiness of the mouth or ears.
“Importantly, these events were very transient. They occurred typically within the first 8 days and lasted 14-67 hours, with a median 1-day duration of rescue medication,” he said.
Symptomatic improvement was typically seen beginning at 8-12 weeks. Adults were free to take the once-daily tablet anytime during the day. The pediatric patients were advised not to do so in the morning because they wouldn’t be under observation while on a school bus.
The tablet is based upon a formulation of allergen extracts from the two major species of HDM: Dermatophagoides pterornyssinus and D. farinae. More than 90% of HDM-sensitized patients are sensitized to both. A highly standardized manufacturing process ensures that the tablet contains 50 mcg of the four major HDM allergens in equal ratio – Der f1, Der p2, Der p1, and Der f 2 – plus other components.
In response to audience questions, Dr. Nolte said the company had tried incorporating an antihistamine into the tablet to block adverse reactions but it didn’t work. Adverse events typically occur within a couple of minutes after taking the tablet, and antihistamines are far too slow-acting to help.
“You can premedicate with antihistamines. We know European investigators and clinicians who are doing it. But I would not recommend it personally, because these tablets are taken at home after the first administration in the office, and I think it’s important that the patient has a good feel for what happens over the following days. There is a potential risk of masking side effects with premedication, which could be a concern,” Dr. Nolte said.
Merck already has two FDA-approved SLIT tablets developed with ALK on the U.S. market: Grastek, for treatment of grass pollen–induced allergic rhinitis in children and adults, and Ragwitek, for ragweed-induced allergic disease in adults.
The trial was sponsored by Merck and presented by a full-time company employee.
LOS ANGELES – A sublingual immunotherapy tablet for house dust mite–induced allergic rhinitis achieved clinically meaningful improvement in symptoms along with less use of rescue medications and a favorable safety profile in a pivotal phase III trial, Dr. Hendrik Nolte reported at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
The double-blind, 52-week randomized trial included 1,482 North American adults and adolescents.
“This was the largest clinical trial ever conducted with sublingual therapy in North America, and the first successful North American trial of a house dust mite sublingual immunotherapy tablet. It confirms results from previous large European trials,” noted Dr. Nolte of Merck, which is collaborating with the Danish company ALK in developing this therapy.
The sublingual immunotherapy (SLIT) tablet, known for now as the 12 SQ HDM SLIT-tablet, has been approved by European regulatory authorities for treatment of adults with house dust mite (HDM) allergic rhinitis or HDM allergic asthma. Based upon the positive findings in the phase III U.S. trial, Merck applied in February 2016 to the U.S. Food and Drug Administration for approval of the tablet as a biologic agent in patients aged 12 years or older.
HDM is the most common indoor allergen in the world. Unlike pollen and ragweed allergies, it’s not a seasonal problem. Moreover, HDM-induced allergic rhinitis is associated with increased risk of asthma. Although HDM allergic rhinitis can be treated symptomatically with oral antihistamines and nasal steroids, allergy immunotherapy has the appeal of addressing the underlying disease mechanism and potentially altering the long-term course. SLIT using a highly standardized HDM allergen extract offers a major advantage over traditional allergy immunotherapy via a lengthy program of subcutaneous injections – namely, the convenience of home self-administration.
The primary endpoint in the U.S. pivotal trial was the total combined rhinitis score (TCRS), which is the sum of the daily rhinitis symptom score and the daily rescue medication usage score averaged over the last 8 weeks of the year-long study. The FDA has set the bar for establishing clinically meaningful improvement: it requires demonstration of a reduction in the TCRS of at least 15%, compared with placebo. The 12 SQ HDM SLIT-tablet trial exceeded this standard, achieving a 17% reduction. The study also met its key prespecified secondary endpoints, including a 16% reduction in the daily rhinosinusitis symptom score and an 18% decrease in the daily medication score, compared with placebo.
Participants in the trial had a mean 18-year history of allergic rhinitis with or without conjunctivitis. Seventy-five percent of them were sensitized to other common allergens in addition to HDM, and 31% of subjects had comorbid asthma. The HDM SLIT therapy was equally effective in asthmatics and nonasthmatics, in patients allergic only to HDM and those who were polysensitized, and in subjects with and without conjunctivitis, according to Dr. Nolte.
There were no serious adverse events related to the 12 SQ HDM SLIT-tablet. A total of 9.8% of patients discontinued SLIT because of treatment-emergent adverse events, chiefly mild-to-moderate throat irritation, mouth swelling, or itchiness of the mouth or ears.
“Importantly, these events were very transient. They occurred typically within the first 8 days and lasted 14-67 hours, with a median 1-day duration of rescue medication,” he said.
Symptomatic improvement was typically seen beginning at 8-12 weeks. Adults were free to take the once-daily tablet anytime during the day. The pediatric patients were advised not to do so in the morning because they wouldn’t be under observation while on a school bus.
The tablet is based upon a formulation of allergen extracts from the two major species of HDM: Dermatophagoides pterornyssinus and D. farinae. More than 90% of HDM-sensitized patients are sensitized to both. A highly standardized manufacturing process ensures that the tablet contains 50 mcg of the four major HDM allergens in equal ratio – Der f1, Der p2, Der p1, and Der f 2 – plus other components.
In response to audience questions, Dr. Nolte said the company had tried incorporating an antihistamine into the tablet to block adverse reactions but it didn’t work. Adverse events typically occur within a couple of minutes after taking the tablet, and antihistamines are far too slow-acting to help.
“You can premedicate with antihistamines. We know European investigators and clinicians who are doing it. But I would not recommend it personally, because these tablets are taken at home after the first administration in the office, and I think it’s important that the patient has a good feel for what happens over the following days. There is a potential risk of masking side effects with premedication, which could be a concern,” Dr. Nolte said.
Merck already has two FDA-approved SLIT tablets developed with ALK on the U.S. market: Grastek, for treatment of grass pollen–induced allergic rhinitis in children and adults, and Ragwitek, for ragweed-induced allergic disease in adults.
The trial was sponsored by Merck and presented by a full-time company employee.
AT 2016 AAAAI ANNUAL MEETING
First sublingual immunotherapy tablet for house dust mite allergic rhinitis may be U.S.-bound
LOS ANGELES – A sublingual immunotherapy tablet for house dust mite–induced allergic rhinitis achieved clinically meaningful improvement in symptoms along with less use of rescue medications and a favorable safety profile in a pivotal phase III trial, Dr. Hendrik Nolte reported at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
The double-blind, 52-week randomized trial included 1,482 North American adults and adolescents.
“This was the largest clinical trial ever conducted with sublingual therapy in North America, and the first successful North American trial of a house dust mite sublingual immunotherapy tablet. It confirms results from previous large European trials,” noted Dr. Nolte of Merck, which is collaborating with the Danish company ALK in developing this therapy.
The sublingual immunotherapy (SLIT) tablet, known for now as the 12 SQ HDM SLIT-tablet, has been approved by European regulatory authorities for treatment of adults with house dust mite (HDM) allergic rhinitis or HDM allergic asthma. Based upon the positive findings in the phase III U.S. trial, Merck applied in February 2016 to the U.S. Food and Drug Administration for approval of the tablet as a biologic agent in patients aged 12 years or older.
HDM is the most common indoor allergen in the world. Unlike pollen and ragweed allergies, it’s not a seasonal problem. Moreover, HDM-induced allergic rhinitis is associated with increased risk of asthma. Although HDM allergic rhinitis can be treated symptomatically with oral antihistamines and nasal steroids, allergy immunotherapy has the appeal of addressing the underlying disease mechanism and potentially altering the long-term course. SLIT using a highly standardized HDM allergen extract offers a major advantage over traditional allergy immunotherapy via a lengthy program of subcutaneous injections – namely, the convenience of home self-administration.
The primary endpoint in the U.S. pivotal trial was the total combined rhinitis score (TCRS), which is the sum of the daily rhinitis symptom score and the daily rescue medication usage score averaged over the last 8 weeks of the year-long study. The FDA has set the bar for establishing clinically meaningful improvement: it requires demonstration of a reduction in the TCRS of at least 15%, compared with placebo. The 12 SQ HDM SLIT-tablet trial exceeded this standard, achieving a 17% reduction. The study also met its key prespecified secondary endpoints, including a 16% reduction in the daily rhinosinusitis symptom score and an 18% decrease in the daily medication score, compared with placebo.
Participants in the trial had a mean 18-year history of allergic rhinitis with or without conjunctivitis. Seventy-five percent of them were sensitized to other common allergens in addition to HDM, and 31% of subjects had comorbid asthma. The HDM SLIT therapy was equally effective in asthmatics and nonasthmatics, in patients allergic only to HDM and those who were polysensitized, and in subjects with and without conjunctivitis, according to Dr. Nolte.
There were no serious adverse events related to the 12 SQ HDM SLIT-tablet. A total of 9.8% of patients discontinued SLIT because of treatment-emergent adverse events, chiefly mild-to-moderate throat irritation, mouth swelling, or itchiness of the mouth or ears.
“Importantly, these events were very transient. They occurred typically within the first 8 days and lasted 14-67 hours, with a median 1-day duration of rescue medication,” he said.
Symptomatic improvement was typically seen beginning at 8-12 weeks. Adults were free to take the once-daily tablet anytime during the day. The pediatric patients were advised not to do so in the morning because they wouldn’t be under observation while on a school bus.
The tablet is based upon a formulation of allergen extracts from the two major species of HDM: Dermatophagoides pterornyssinus and D. farinae. More than 90% of HDM-sensitized patients are sensitized to both. A highly standardized manufacturing process ensures that the tablet contains 50 mcg of the four major HDM allergens in equal ratio – Der f1, Der p2, Der p1, and Der f 2 – plus other components.
In response to audience questions, Dr. Nolte said the company had tried incorporating an antihistamine into the tablet to block adverse reactions but it didn’t work. Adverse events typically occur within a couple of minutes after taking the tablet, and antihistamines are far too slow-acting to help.
“You can premedicate with antihistamines. We know European investigators and clinicians who are doing it. But I would not recommend it personally, because these tablets are taken at home after the first administration in the office, and I think it’s important that the patient has a good feel for what happens over the following days. There is a potential risk of masking side effects with premedication, which could be a concern,” Dr. Nolte said.
Merck already has two FDA-approved SLIT tablets developed with ALK on the U.S. market: Grastek, for treatment of grass pollen–induced allergic rhinitis in children and adults, and Ragwitek, for ragweed-induced allergic disease in adults.
The trial was sponsored by Merck and presented by a full-time company employee.
LOS ANGELES – A sublingual immunotherapy tablet for house dust mite–induced allergic rhinitis achieved clinically meaningful improvement in symptoms along with less use of rescue medications and a favorable safety profile in a pivotal phase III trial, Dr. Hendrik Nolte reported at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
The double-blind, 52-week randomized trial included 1,482 North American adults and adolescents.
“This was the largest clinical trial ever conducted with sublingual therapy in North America, and the first successful North American trial of a house dust mite sublingual immunotherapy tablet. It confirms results from previous large European trials,” noted Dr. Nolte of Merck, which is collaborating with the Danish company ALK in developing this therapy.
The sublingual immunotherapy (SLIT) tablet, known for now as the 12 SQ HDM SLIT-tablet, has been approved by European regulatory authorities for treatment of adults with house dust mite (HDM) allergic rhinitis or HDM allergic asthma. Based upon the positive findings in the phase III U.S. trial, Merck applied in February 2016 to the U.S. Food and Drug Administration for approval of the tablet as a biologic agent in patients aged 12 years or older.
HDM is the most common indoor allergen in the world. Unlike pollen and ragweed allergies, it’s not a seasonal problem. Moreover, HDM-induced allergic rhinitis is associated with increased risk of asthma. Although HDM allergic rhinitis can be treated symptomatically with oral antihistamines and nasal steroids, allergy immunotherapy has the appeal of addressing the underlying disease mechanism and potentially altering the long-term course. SLIT using a highly standardized HDM allergen extract offers a major advantage over traditional allergy immunotherapy via a lengthy program of subcutaneous injections – namely, the convenience of home self-administration.
The primary endpoint in the U.S. pivotal trial was the total combined rhinitis score (TCRS), which is the sum of the daily rhinitis symptom score and the daily rescue medication usage score averaged over the last 8 weeks of the year-long study. The FDA has set the bar for establishing clinically meaningful improvement: it requires demonstration of a reduction in the TCRS of at least 15%, compared with placebo. The 12 SQ HDM SLIT-tablet trial exceeded this standard, achieving a 17% reduction. The study also met its key prespecified secondary endpoints, including a 16% reduction in the daily rhinosinusitis symptom score and an 18% decrease in the daily medication score, compared with placebo.
Participants in the trial had a mean 18-year history of allergic rhinitis with or without conjunctivitis. Seventy-five percent of them were sensitized to other common allergens in addition to HDM, and 31% of subjects had comorbid asthma. The HDM SLIT therapy was equally effective in asthmatics and nonasthmatics, in patients allergic only to HDM and those who were polysensitized, and in subjects with and without conjunctivitis, according to Dr. Nolte.
There were no serious adverse events related to the 12 SQ HDM SLIT-tablet. A total of 9.8% of patients discontinued SLIT because of treatment-emergent adverse events, chiefly mild-to-moderate throat irritation, mouth swelling, or itchiness of the mouth or ears.
“Importantly, these events were very transient. They occurred typically within the first 8 days and lasted 14-67 hours, with a median 1-day duration of rescue medication,” he said.
Symptomatic improvement was typically seen beginning at 8-12 weeks. Adults were free to take the once-daily tablet anytime during the day. The pediatric patients were advised not to do so in the morning because they wouldn’t be under observation while on a school bus.
The tablet is based upon a formulation of allergen extracts from the two major species of HDM: Dermatophagoides pterornyssinus and D. farinae. More than 90% of HDM-sensitized patients are sensitized to both. A highly standardized manufacturing process ensures that the tablet contains 50 mcg of the four major HDM allergens in equal ratio – Der f1, Der p2, Der p1, and Der f 2 – plus other components.
In response to audience questions, Dr. Nolte said the company had tried incorporating an antihistamine into the tablet to block adverse reactions but it didn’t work. Adverse events typically occur within a couple of minutes after taking the tablet, and antihistamines are far too slow-acting to help.
“You can premedicate with antihistamines. We know European investigators and clinicians who are doing it. But I would not recommend it personally, because these tablets are taken at home after the first administration in the office, and I think it’s important that the patient has a good feel for what happens over the following days. There is a potential risk of masking side effects with premedication, which could be a concern,” Dr. Nolte said.
Merck already has two FDA-approved SLIT tablets developed with ALK on the U.S. market: Grastek, for treatment of grass pollen–induced allergic rhinitis in children and adults, and Ragwitek, for ragweed-induced allergic disease in adults.
The trial was sponsored by Merck and presented by a full-time company employee.
LOS ANGELES – A sublingual immunotherapy tablet for house dust mite–induced allergic rhinitis achieved clinically meaningful improvement in symptoms along with less use of rescue medications and a favorable safety profile in a pivotal phase III trial, Dr. Hendrik Nolte reported at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
The double-blind, 52-week randomized trial included 1,482 North American adults and adolescents.
“This was the largest clinical trial ever conducted with sublingual therapy in North America, and the first successful North American trial of a house dust mite sublingual immunotherapy tablet. It confirms results from previous large European trials,” noted Dr. Nolte of Merck, which is collaborating with the Danish company ALK in developing this therapy.
The sublingual immunotherapy (SLIT) tablet, known for now as the 12 SQ HDM SLIT-tablet, has been approved by European regulatory authorities for treatment of adults with house dust mite (HDM) allergic rhinitis or HDM allergic asthma. Based upon the positive findings in the phase III U.S. trial, Merck applied in February 2016 to the U.S. Food and Drug Administration for approval of the tablet as a biologic agent in patients aged 12 years or older.
HDM is the most common indoor allergen in the world. Unlike pollen and ragweed allergies, it’s not a seasonal problem. Moreover, HDM-induced allergic rhinitis is associated with increased risk of asthma. Although HDM allergic rhinitis can be treated symptomatically with oral antihistamines and nasal steroids, allergy immunotherapy has the appeal of addressing the underlying disease mechanism and potentially altering the long-term course. SLIT using a highly standardized HDM allergen extract offers a major advantage over traditional allergy immunotherapy via a lengthy program of subcutaneous injections – namely, the convenience of home self-administration.
The primary endpoint in the U.S. pivotal trial was the total combined rhinitis score (TCRS), which is the sum of the daily rhinitis symptom score and the daily rescue medication usage score averaged over the last 8 weeks of the year-long study. The FDA has set the bar for establishing clinically meaningful improvement: it requires demonstration of a reduction in the TCRS of at least 15%, compared with placebo. The 12 SQ HDM SLIT-tablet trial exceeded this standard, achieving a 17% reduction. The study also met its key prespecified secondary endpoints, including a 16% reduction in the daily rhinosinusitis symptom score and an 18% decrease in the daily medication score, compared with placebo.
Participants in the trial had a mean 18-year history of allergic rhinitis with or without conjunctivitis. Seventy-five percent of them were sensitized to other common allergens in addition to HDM, and 31% of subjects had comorbid asthma. The HDM SLIT therapy was equally effective in asthmatics and nonasthmatics, in patients allergic only to HDM and those who were polysensitized, and in subjects with and without conjunctivitis, according to Dr. Nolte.
There were no serious adverse events related to the 12 SQ HDM SLIT-tablet. A total of 9.8% of patients discontinued SLIT because of treatment-emergent adverse events, chiefly mild-to-moderate throat irritation, mouth swelling, or itchiness of the mouth or ears.
“Importantly, these events were very transient. They occurred typically within the first 8 days and lasted 14-67 hours, with a median 1-day duration of rescue medication,” he said.
Symptomatic improvement was typically seen beginning at 8-12 weeks. Adults were free to take the once-daily tablet anytime during the day. The pediatric patients were advised not to do so in the morning because they wouldn’t be under observation while on a school bus.
The tablet is based upon a formulation of allergen extracts from the two major species of HDM: Dermatophagoides pterornyssinus and D. farinae. More than 90% of HDM-sensitized patients are sensitized to both. A highly standardized manufacturing process ensures that the tablet contains 50 mcg of the four major HDM allergens in equal ratio – Der f1, Der p2, Der p1, and Der f 2 – plus other components.
In response to audience questions, Dr. Nolte said the company had tried incorporating an antihistamine into the tablet to block adverse reactions but it didn’t work. Adverse events typically occur within a couple of minutes after taking the tablet, and antihistamines are far too slow-acting to help.
“You can premedicate with antihistamines. We know European investigators and clinicians who are doing it. But I would not recommend it personally, because these tablets are taken at home after the first administration in the office, and I think it’s important that the patient has a good feel for what happens over the following days. There is a potential risk of masking side effects with premedication, which could be a concern,” Dr. Nolte said.
Merck already has two FDA-approved SLIT tablets developed with ALK on the U.S. market: Grastek, for treatment of grass pollen–induced allergic rhinitis in children and adults, and Ragwitek, for ragweed-induced allergic disease in adults.
The trial was sponsored by Merck and presented by a full-time company employee.
AT 2016 AAAAI ANNUAL MEETING
Key clinical point: A safe, effective, and convenient self-administered alternative to allergy shots or symptomatic medications for house dust mite allergic rhinitis may be in the works.
Major finding: A once-daily tablet containing house dust mite extract resulted in a clinically meaningful 17% improvement relative to placebo in a score combining symptomatic improvement and reduced use of rescue medications.
Data source: This pivotal phase III, double-blind, 52-week randomized trial included 1,482 North American adults and adolescents with house dust mite–induced allergic rhinitis with or without conjunctivitis.
Disclosures: The trial was sponsored by Merck and presented by a full-time company employee.
AAAAI: Early Peanut Consumption Brings Lasting Protection From Allergy
LOS ANGELES – A peanut allergy prevention strategy based upon regular consumption of peanut-containing foods from infancy to age 5 continued to provide protection even after peanut intake was halted for a full year from age 5 to 6, according to new results from an extension of the landmark LEAP trial, known as LEAP-On, presented at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
The impetus for LEAP-On was the investigators’ concern that a period of peanut avoidance might cause loss of the protective state. But that didn’t occur.
“I think there is no doubt that we have prevented peanut allergy so far in these high-risk children. Next, the LEAP-Ad Lib study will tell us whether we’ve prevented it by age 10,” said Dr. Gideon Lack of King’s College London, who headed LEAP-On.
A second major randomized trial known as EAT (Enquiring About Tolerance) presented at the meeting provided further support for early dietary introduction of allergenic foods. EAT differed from LEAP (Learning Early About Peanut Allergy) and LEAP-On in that it ambitiously randomized infants to early introduction or avoidance of not one but six allergenic foods: peanut, cooked egg, cow’s milk, fish, sesame, and wheat. Also, while LEAP and LEAP-On involved roughly 600 infants known to be at very high risk for allergy, EAT was conducted in a general population of 1,303 infants who weren’t at increased risk, all of whom were exclusively breast-fed until the intervention beginning at age 3 months.
The presentation of the LEAP-On and EAT results at the AAAAI annual meeting was a major event marked by the National Institute of Allergy and Infectious Diseases by same-day release of new NIAID-sponsored draft recommendations for the diagnosis and management of food allergies.
In a press conference held at the AAAAI annual meeting to announce the start of a 45-day public comment period for the draft update of the 2010 guidelines, Dr. Daniel Rotrosen, director of NIAID’s division of allergy, immunology and transplantation, said the new guidelines were developed largely in response to the compelling LEAP findings. That trial demonstrated that sustained consumption of peanut starting in infancy resulted in an 81% lower rate of peanut allergy at age 5 years compared to a strategy of peanut avoidance (N Engl J Med. 2015;372:803-13).
The draft guidelines, now available on the NIAID website, represent a sharp departure from the former recommendation that physicians encourage exclusive breastfeeding for the first 6 months of life followed by cautious introduction of other foods. Whereas the former orthodoxy was that delayed introduction of allergenic foods protects against development of food allergy, the new evidence-based concept supported by the LEAP and EAT findings is that just the opposite is true: that is, introduction of such foods during the period of immunologic plasticity in infancy induces tolerance.
Thus, the draft guidelines recommend that infants at high risk for peanut allergy because they have severe eczema and/or egg allergy should have introduction of peanut-containing food at 4-6 months of age to reduce their risk of peanut allergy, preceded by evaluation using peanut-specific IgE or skin prick testing to make sure it’s safe. That age window coincides with well-child visits and vaccination schedules, Dr. Rotrosen noted.
These guidelines represent the consensus of 26 organizations that participated in their development. Among them are the American Academy of Pediatrics, the American Academy of Family Physicians, the American Academy of Dermatology, the American College of Gastroenterology, and AAAAI.
“I expect the new guidelines, when finalized, to be endorsed by the leadership of all the participating organizations,” Dr. Rotrosen said.
The new paradigm will require cultural change, said Dr. James R. Baker Jr., CEO and chief medical officer of Food Allergy Research and Education, a nonprofit organization that provided partial funding for LEAP and LEAP-On.
“I think for a long time we’ve vilified these foods. There’s nothing inherently wrong with their intake, and that’s a message we need to get across to parents and physicians so they can start thinking differently,” he said.
“The good news about these studies is that they show there’s no reason not to do this,” Dr. Baker added. “There’s no harm that comes from the early introduction.”
Dr. Lack, who led the EAT trial, noted that the study didn’t meet it’s primary endpoint of a significantly lower prevalence of food allergy to any of the six intervention foods at age 3 years in the intention-to-treat analysis. But adherence to the demanding EAT early-introduction protocol was a problem. Indeed, only 43% of participants adhered to the study protocol. In a per-protocol analysis restricted to the adherent group, however, early introduction was associated with a highly significant 67% reduction in the relative risk of food allergy at 3 years of age compared to controls. And for the two most prevalent food allergies – to peanut and egg – the relative risk reductions in the early-introduction group were 100% and 75%, respectively.
The EAT results suggest that an effective preventive dose of peanut in infants at least 3 months of age is roughly 2 g of peanut protein per week, equivalent to just under 2 tsp of peanut butter, according to Dr. Lack.
Simultaneously with presentation of the LEAP-On and EAT trials in Los Angeles, the studies were published online at NEJM.org (doi: 10.1056/NEJMoa1514210 for LEAP-ON and 10.1056/NEJMoa1514209 for EAT).
LEAP-On was supported primarily by NIAID. EAT was funded mainly by the UK Foods Standards Agency and the Medical Research Council. Dr. Lack reported receiving grants from those agencies as well as Food Allergy Research and Education.
LOS ANGELES – A peanut allergy prevention strategy based upon regular consumption of peanut-containing foods from infancy to age 5 continued to provide protection even after peanut intake was halted for a full year from age 5 to 6, according to new results from an extension of the landmark LEAP trial, known as LEAP-On, presented at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
The impetus for LEAP-On was the investigators’ concern that a period of peanut avoidance might cause loss of the protective state. But that didn’t occur.
“I think there is no doubt that we have prevented peanut allergy so far in these high-risk children. Next, the LEAP-Ad Lib study will tell us whether we’ve prevented it by age 10,” said Dr. Gideon Lack of King’s College London, who headed LEAP-On.
A second major randomized trial known as EAT (Enquiring About Tolerance) presented at the meeting provided further support for early dietary introduction of allergenic foods. EAT differed from LEAP (Learning Early About Peanut Allergy) and LEAP-On in that it ambitiously randomized infants to early introduction or avoidance of not one but six allergenic foods: peanut, cooked egg, cow’s milk, fish, sesame, and wheat. Also, while LEAP and LEAP-On involved roughly 600 infants known to be at very high risk for allergy, EAT was conducted in a general population of 1,303 infants who weren’t at increased risk, all of whom were exclusively breast-fed until the intervention beginning at age 3 months.
The presentation of the LEAP-On and EAT results at the AAAAI annual meeting was a major event marked by the National Institute of Allergy and Infectious Diseases by same-day release of new NIAID-sponsored draft recommendations for the diagnosis and management of food allergies.
In a press conference held at the AAAAI annual meeting to announce the start of a 45-day public comment period for the draft update of the 2010 guidelines, Dr. Daniel Rotrosen, director of NIAID’s division of allergy, immunology and transplantation, said the new guidelines were developed largely in response to the compelling LEAP findings. That trial demonstrated that sustained consumption of peanut starting in infancy resulted in an 81% lower rate of peanut allergy at age 5 years compared to a strategy of peanut avoidance (N Engl J Med. 2015;372:803-13).
The draft guidelines, now available on the NIAID website, represent a sharp departure from the former recommendation that physicians encourage exclusive breastfeeding for the first 6 months of life followed by cautious introduction of other foods. Whereas the former orthodoxy was that delayed introduction of allergenic foods protects against development of food allergy, the new evidence-based concept supported by the LEAP and EAT findings is that just the opposite is true: that is, introduction of such foods during the period of immunologic plasticity in infancy induces tolerance.
Thus, the draft guidelines recommend that infants at high risk for peanut allergy because they have severe eczema and/or egg allergy should have introduction of peanut-containing food at 4-6 months of age to reduce their risk of peanut allergy, preceded by evaluation using peanut-specific IgE or skin prick testing to make sure it’s safe. That age window coincides with well-child visits and vaccination schedules, Dr. Rotrosen noted.
These guidelines represent the consensus of 26 organizations that participated in their development. Among them are the American Academy of Pediatrics, the American Academy of Family Physicians, the American Academy of Dermatology, the American College of Gastroenterology, and AAAAI.
“I expect the new guidelines, when finalized, to be endorsed by the leadership of all the participating organizations,” Dr. Rotrosen said.
The new paradigm will require cultural change, said Dr. James R. Baker Jr., CEO and chief medical officer of Food Allergy Research and Education, a nonprofit organization that provided partial funding for LEAP and LEAP-On.
“I think for a long time we’ve vilified these foods. There’s nothing inherently wrong with their intake, and that’s a message we need to get across to parents and physicians so they can start thinking differently,” he said.
“The good news about these studies is that they show there’s no reason not to do this,” Dr. Baker added. “There’s no harm that comes from the early introduction.”
Dr. Lack, who led the EAT trial, noted that the study didn’t meet it’s primary endpoint of a significantly lower prevalence of food allergy to any of the six intervention foods at age 3 years in the intention-to-treat analysis. But adherence to the demanding EAT early-introduction protocol was a problem. Indeed, only 43% of participants adhered to the study protocol. In a per-protocol analysis restricted to the adherent group, however, early introduction was associated with a highly significant 67% reduction in the relative risk of food allergy at 3 years of age compared to controls. And for the two most prevalent food allergies – to peanut and egg – the relative risk reductions in the early-introduction group were 100% and 75%, respectively.
The EAT results suggest that an effective preventive dose of peanut in infants at least 3 months of age is roughly 2 g of peanut protein per week, equivalent to just under 2 tsp of peanut butter, according to Dr. Lack.
Simultaneously with presentation of the LEAP-On and EAT trials in Los Angeles, the studies were published online at NEJM.org (doi: 10.1056/NEJMoa1514210 for LEAP-ON and 10.1056/NEJMoa1514209 for EAT).
LEAP-On was supported primarily by NIAID. EAT was funded mainly by the UK Foods Standards Agency and the Medical Research Council. Dr. Lack reported receiving grants from those agencies as well as Food Allergy Research and Education.
LOS ANGELES – A peanut allergy prevention strategy based upon regular consumption of peanut-containing foods from infancy to age 5 continued to provide protection even after peanut intake was halted for a full year from age 5 to 6, according to new results from an extension of the landmark LEAP trial, known as LEAP-On, presented at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
The impetus for LEAP-On was the investigators’ concern that a period of peanut avoidance might cause loss of the protective state. But that didn’t occur.
“I think there is no doubt that we have prevented peanut allergy so far in these high-risk children. Next, the LEAP-Ad Lib study will tell us whether we’ve prevented it by age 10,” said Dr. Gideon Lack of King’s College London, who headed LEAP-On.
A second major randomized trial known as EAT (Enquiring About Tolerance) presented at the meeting provided further support for early dietary introduction of allergenic foods. EAT differed from LEAP (Learning Early About Peanut Allergy) and LEAP-On in that it ambitiously randomized infants to early introduction or avoidance of not one but six allergenic foods: peanut, cooked egg, cow’s milk, fish, sesame, and wheat. Also, while LEAP and LEAP-On involved roughly 600 infants known to be at very high risk for allergy, EAT was conducted in a general population of 1,303 infants who weren’t at increased risk, all of whom were exclusively breast-fed until the intervention beginning at age 3 months.
The presentation of the LEAP-On and EAT results at the AAAAI annual meeting was a major event marked by the National Institute of Allergy and Infectious Diseases by same-day release of new NIAID-sponsored draft recommendations for the diagnosis and management of food allergies.
In a press conference held at the AAAAI annual meeting to announce the start of a 45-day public comment period for the draft update of the 2010 guidelines, Dr. Daniel Rotrosen, director of NIAID’s division of allergy, immunology and transplantation, said the new guidelines were developed largely in response to the compelling LEAP findings. That trial demonstrated that sustained consumption of peanut starting in infancy resulted in an 81% lower rate of peanut allergy at age 5 years compared to a strategy of peanut avoidance (N Engl J Med. 2015;372:803-13).
The draft guidelines, now available on the NIAID website, represent a sharp departure from the former recommendation that physicians encourage exclusive breastfeeding for the first 6 months of life followed by cautious introduction of other foods. Whereas the former orthodoxy was that delayed introduction of allergenic foods protects against development of food allergy, the new evidence-based concept supported by the LEAP and EAT findings is that just the opposite is true: that is, introduction of such foods during the period of immunologic plasticity in infancy induces tolerance.
Thus, the draft guidelines recommend that infants at high risk for peanut allergy because they have severe eczema and/or egg allergy should have introduction of peanut-containing food at 4-6 months of age to reduce their risk of peanut allergy, preceded by evaluation using peanut-specific IgE or skin prick testing to make sure it’s safe. That age window coincides with well-child visits and vaccination schedules, Dr. Rotrosen noted.
These guidelines represent the consensus of 26 organizations that participated in their development. Among them are the American Academy of Pediatrics, the American Academy of Family Physicians, the American Academy of Dermatology, the American College of Gastroenterology, and AAAAI.
“I expect the new guidelines, when finalized, to be endorsed by the leadership of all the participating organizations,” Dr. Rotrosen said.
The new paradigm will require cultural change, said Dr. James R. Baker Jr., CEO and chief medical officer of Food Allergy Research and Education, a nonprofit organization that provided partial funding for LEAP and LEAP-On.
“I think for a long time we’ve vilified these foods. There’s nothing inherently wrong with their intake, and that’s a message we need to get across to parents and physicians so they can start thinking differently,” he said.
“The good news about these studies is that they show there’s no reason not to do this,” Dr. Baker added. “There’s no harm that comes from the early introduction.”
Dr. Lack, who led the EAT trial, noted that the study didn’t meet it’s primary endpoint of a significantly lower prevalence of food allergy to any of the six intervention foods at age 3 years in the intention-to-treat analysis. But adherence to the demanding EAT early-introduction protocol was a problem. Indeed, only 43% of participants adhered to the study protocol. In a per-protocol analysis restricted to the adherent group, however, early introduction was associated with a highly significant 67% reduction in the relative risk of food allergy at 3 years of age compared to controls. And for the two most prevalent food allergies – to peanut and egg – the relative risk reductions in the early-introduction group were 100% and 75%, respectively.
The EAT results suggest that an effective preventive dose of peanut in infants at least 3 months of age is roughly 2 g of peanut protein per week, equivalent to just under 2 tsp of peanut butter, according to Dr. Lack.
Simultaneously with presentation of the LEAP-On and EAT trials in Los Angeles, the studies were published online at NEJM.org (doi: 10.1056/NEJMoa1514210 for LEAP-ON and 10.1056/NEJMoa1514209 for EAT).
LEAP-On was supported primarily by NIAID. EAT was funded mainly by the UK Foods Standards Agency and the Medical Research Council. Dr. Lack reported receiving grants from those agencies as well as Food Allergy Research and Education.
EXPERT ANALYSIS FROM THE 2016 AAAAI ANNUAL MEETING
AAAAI: Early peanut consumption brings lasting protection from allergy
LOS ANGELES – A peanut allergy prevention strategy based upon regular consumption of peanut-containing foods from infancy to age 5 continued to provide protection even after peanut intake was halted for a full year from age 5 to 6, according to new results from an extension of the landmark LEAP trial, known as LEAP-On, presented at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
The impetus for LEAP-On was the investigators’ concern that a period of peanut avoidance might cause loss of the protective state. But that didn’t occur.
“I think there is no doubt that we have prevented peanut allergy so far in these high-risk children. Next, the LEAP-Ad Lib study will tell us whether we’ve prevented it by age 10,” said Dr. Gideon Lack of King’s College London, who headed LEAP-On.
A second major randomized trial known as EAT (Enquiring About Tolerance) presented at the meeting provided further support for early dietary introduction of allergenic foods. EAT differed from LEAP (Learning Early About Peanut Allergy) and LEAP-On in that it ambitiously randomized infants to early introduction or avoidance of not one but six allergenic foods: peanut, cooked egg, cow’s milk, fish, sesame, and wheat. Also, while LEAP and LEAP-On involved roughly 600 infants known to be at very high risk for allergy, EAT was conducted in a general population of 1,303 infants who weren’t at increased risk, all of whom were exclusively breast-fed until the intervention beginning at age 3 months.
The presentation of the LEAP-On and EAT results at the AAAAI annual meeting was a major event marked by the National Institute of Allergy and Infectious Diseases by same-day release of new NIAID-sponsored draft recommendations for the diagnosis and management of food allergies.
In a press conference held at the AAAAI annual meeting to announce the start of a 45-day public comment period for the draft update of the 2010 guidelines, Dr. Daniel Rotrosen, director of NIAID’s division of allergy, immunology and transplantation, said the new guidelines were developed largely in response to the compelling LEAP findings. That trial demonstrated that sustained consumption of peanut starting in infancy resulted in an 81% lower rate of peanut allergy at age 5 years compared to a strategy of peanut avoidance (N Engl J Med. 2015;372:803-13).
The draft guidelines, now available on the NIAID website, represent a sharp departure from the former recommendation that physicians encourage exclusive breastfeeding for the first 6 months of life followed by cautious introduction of other foods. Whereas the former orthodoxy was that delayed introduction of allergenic foods protects against development of food allergy, the new evidence-based concept supported by the LEAP and EAT findings is that just the opposite is true: that is, introduction of such foods during the period of immunologic plasticity in infancy induces tolerance.
Thus, the draft guidelines recommend that infants at high risk for peanut allergy because they have severe eczema and/or egg allergy should have introduction of peanut-containing food at 4-6 months of age to reduce their risk of peanut allergy, preceded by evaluation using peanut-specific IgE or skin prick testing to make sure it’s safe. That age window coincides with well-child visits and vaccination schedules, Dr. Rotrosen noted.
These guidelines represent the consensus of 26 organizations that participated in their development. Among them are the American Academy of Pediatrics, the American Academy of Family Physicians, the American Academy of Dermatology, the American College of Gastroenterology, and AAAAI.
“I expect the new guidelines, when finalized, to be endorsed by the leadership of all the participating organizations,” Dr. Rotrosen said.
The new paradigm will require cultural change, said Dr. James R. Baker Jr., CEO and chief medical officer of Food Allergy Research and Education, a nonprofit organization that provided partial funding for LEAP and LEAP-On.
“I think for a long time we’ve vilified these foods. There’s nothing inherently wrong with their intake, and that’s a message we need to get across to parents and physicians so they can start thinking differently,” he said.
“The good news about these studies is that they show there’s no reason not to do this,” Dr. Baker added. “There’s no harm that comes from the early introduction.”
Dr. Lack, who led the EAT trial, noted that the study didn’t meet it’s primary endpoint of a significantly lower prevalence of food allergy to any of the six intervention foods at age 3 years in the intention-to-treat analysis. But adherence to the demanding EAT early-introduction protocol was a problem. Indeed, only 43% of participants adhered to the study protocol. In a per-protocol analysis restricted to the adherent group, however, early introduction was associated with a highly significant 67% reduction in the relative risk of food allergy at 3 years of age compared to controls. And for the two most prevalent food allergies – to peanut and egg – the relative risk reductions in the early-introduction group were 100% and 75%, respectively.
The EAT results suggest that an effective preventive dose of peanut in infants at least 3 months of age is roughly 2 g of peanut protein per week, equivalent to just under 2 tsp of peanut butter, according to Dr. Lack.
Simultaneously with presentation of the LEAP-On and EAT trials in Los Angeles, the studies were published online at NEJM.org (doi: 10.1056/NEJMoa1514210 for LEAP-ON and 10.1056/NEJMoa1514209 for EAT).
LEAP-On was supported primarily by NIAID. EAT was funded mainly by the UK Foods Standards Agency and the Medical Research Council. Dr. Lack reported receiving grants from those agencies as well as Food Allergy Research and Education.
LOS ANGELES – A peanut allergy prevention strategy based upon regular consumption of peanut-containing foods from infancy to age 5 continued to provide protection even after peanut intake was halted for a full year from age 5 to 6, according to new results from an extension of the landmark LEAP trial, known as LEAP-On, presented at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
The impetus for LEAP-On was the investigators’ concern that a period of peanut avoidance might cause loss of the protective state. But that didn’t occur.
“I think there is no doubt that we have prevented peanut allergy so far in these high-risk children. Next, the LEAP-Ad Lib study will tell us whether we’ve prevented it by age 10,” said Dr. Gideon Lack of King’s College London, who headed LEAP-On.
A second major randomized trial known as EAT (Enquiring About Tolerance) presented at the meeting provided further support for early dietary introduction of allergenic foods. EAT differed from LEAP (Learning Early About Peanut Allergy) and LEAP-On in that it ambitiously randomized infants to early introduction or avoidance of not one but six allergenic foods: peanut, cooked egg, cow’s milk, fish, sesame, and wheat. Also, while LEAP and LEAP-On involved roughly 600 infants known to be at very high risk for allergy, EAT was conducted in a general population of 1,303 infants who weren’t at increased risk, all of whom were exclusively breast-fed until the intervention beginning at age 3 months.
The presentation of the LEAP-On and EAT results at the AAAAI annual meeting was a major event marked by the National Institute of Allergy and Infectious Diseases by same-day release of new NIAID-sponsored draft recommendations for the diagnosis and management of food allergies.
In a press conference held at the AAAAI annual meeting to announce the start of a 45-day public comment period for the draft update of the 2010 guidelines, Dr. Daniel Rotrosen, director of NIAID’s division of allergy, immunology and transplantation, said the new guidelines were developed largely in response to the compelling LEAP findings. That trial demonstrated that sustained consumption of peanut starting in infancy resulted in an 81% lower rate of peanut allergy at age 5 years compared to a strategy of peanut avoidance (N Engl J Med. 2015;372:803-13).
The draft guidelines, now available on the NIAID website, represent a sharp departure from the former recommendation that physicians encourage exclusive breastfeeding for the first 6 months of life followed by cautious introduction of other foods. Whereas the former orthodoxy was that delayed introduction of allergenic foods protects against development of food allergy, the new evidence-based concept supported by the LEAP and EAT findings is that just the opposite is true: that is, introduction of such foods during the period of immunologic plasticity in infancy induces tolerance.
Thus, the draft guidelines recommend that infants at high risk for peanut allergy because they have severe eczema and/or egg allergy should have introduction of peanut-containing food at 4-6 months of age to reduce their risk of peanut allergy, preceded by evaluation using peanut-specific IgE or skin prick testing to make sure it’s safe. That age window coincides with well-child visits and vaccination schedules, Dr. Rotrosen noted.
These guidelines represent the consensus of 26 organizations that participated in their development. Among them are the American Academy of Pediatrics, the American Academy of Family Physicians, the American Academy of Dermatology, the American College of Gastroenterology, and AAAAI.
“I expect the new guidelines, when finalized, to be endorsed by the leadership of all the participating organizations,” Dr. Rotrosen said.
The new paradigm will require cultural change, said Dr. James R. Baker Jr., CEO and chief medical officer of Food Allergy Research and Education, a nonprofit organization that provided partial funding for LEAP and LEAP-On.
“I think for a long time we’ve vilified these foods. There’s nothing inherently wrong with their intake, and that’s a message we need to get across to parents and physicians so they can start thinking differently,” he said.
“The good news about these studies is that they show there’s no reason not to do this,” Dr. Baker added. “There’s no harm that comes from the early introduction.”
Dr. Lack, who led the EAT trial, noted that the study didn’t meet it’s primary endpoint of a significantly lower prevalence of food allergy to any of the six intervention foods at age 3 years in the intention-to-treat analysis. But adherence to the demanding EAT early-introduction protocol was a problem. Indeed, only 43% of participants adhered to the study protocol. In a per-protocol analysis restricted to the adherent group, however, early introduction was associated with a highly significant 67% reduction in the relative risk of food allergy at 3 years of age compared to controls. And for the two most prevalent food allergies – to peanut and egg – the relative risk reductions in the early-introduction group were 100% and 75%, respectively.
The EAT results suggest that an effective preventive dose of peanut in infants at least 3 months of age is roughly 2 g of peanut protein per week, equivalent to just under 2 tsp of peanut butter, according to Dr. Lack.
Simultaneously with presentation of the LEAP-On and EAT trials in Los Angeles, the studies were published online at NEJM.org (doi: 10.1056/NEJMoa1514210 for LEAP-ON and 10.1056/NEJMoa1514209 for EAT).
LEAP-On was supported primarily by NIAID. EAT was funded mainly by the UK Foods Standards Agency and the Medical Research Council. Dr. Lack reported receiving grants from those agencies as well as Food Allergy Research and Education.
LOS ANGELES – A peanut allergy prevention strategy based upon regular consumption of peanut-containing foods from infancy to age 5 continued to provide protection even after peanut intake was halted for a full year from age 5 to 6, according to new results from an extension of the landmark LEAP trial, known as LEAP-On, presented at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
The impetus for LEAP-On was the investigators’ concern that a period of peanut avoidance might cause loss of the protective state. But that didn’t occur.
“I think there is no doubt that we have prevented peanut allergy so far in these high-risk children. Next, the LEAP-Ad Lib study will tell us whether we’ve prevented it by age 10,” said Dr. Gideon Lack of King’s College London, who headed LEAP-On.
A second major randomized trial known as EAT (Enquiring About Tolerance) presented at the meeting provided further support for early dietary introduction of allergenic foods. EAT differed from LEAP (Learning Early About Peanut Allergy) and LEAP-On in that it ambitiously randomized infants to early introduction or avoidance of not one but six allergenic foods: peanut, cooked egg, cow’s milk, fish, sesame, and wheat. Also, while LEAP and LEAP-On involved roughly 600 infants known to be at very high risk for allergy, EAT was conducted in a general population of 1,303 infants who weren’t at increased risk, all of whom were exclusively breast-fed until the intervention beginning at age 3 months.
The presentation of the LEAP-On and EAT results at the AAAAI annual meeting was a major event marked by the National Institute of Allergy and Infectious Diseases by same-day release of new NIAID-sponsored draft recommendations for the diagnosis and management of food allergies.
In a press conference held at the AAAAI annual meeting to announce the start of a 45-day public comment period for the draft update of the 2010 guidelines, Dr. Daniel Rotrosen, director of NIAID’s division of allergy, immunology and transplantation, said the new guidelines were developed largely in response to the compelling LEAP findings. That trial demonstrated that sustained consumption of peanut starting in infancy resulted in an 81% lower rate of peanut allergy at age 5 years compared to a strategy of peanut avoidance (N Engl J Med. 2015;372:803-13).
The draft guidelines, now available on the NIAID website, represent a sharp departure from the former recommendation that physicians encourage exclusive breastfeeding for the first 6 months of life followed by cautious introduction of other foods. Whereas the former orthodoxy was that delayed introduction of allergenic foods protects against development of food allergy, the new evidence-based concept supported by the LEAP and EAT findings is that just the opposite is true: that is, introduction of such foods during the period of immunologic plasticity in infancy induces tolerance.
Thus, the draft guidelines recommend that infants at high risk for peanut allergy because they have severe eczema and/or egg allergy should have introduction of peanut-containing food at 4-6 months of age to reduce their risk of peanut allergy, preceded by evaluation using peanut-specific IgE or skin prick testing to make sure it’s safe. That age window coincides with well-child visits and vaccination schedules, Dr. Rotrosen noted.
These guidelines represent the consensus of 26 organizations that participated in their development. Among them are the American Academy of Pediatrics, the American Academy of Family Physicians, the American Academy of Dermatology, the American College of Gastroenterology, and AAAAI.
“I expect the new guidelines, when finalized, to be endorsed by the leadership of all the participating organizations,” Dr. Rotrosen said.
The new paradigm will require cultural change, said Dr. James R. Baker Jr., CEO and chief medical officer of Food Allergy Research and Education, a nonprofit organization that provided partial funding for LEAP and LEAP-On.
“I think for a long time we’ve vilified these foods. There’s nothing inherently wrong with their intake, and that’s a message we need to get across to parents and physicians so they can start thinking differently,” he said.
“The good news about these studies is that they show there’s no reason not to do this,” Dr. Baker added. “There’s no harm that comes from the early introduction.”
Dr. Lack, who led the EAT trial, noted that the study didn’t meet it’s primary endpoint of a significantly lower prevalence of food allergy to any of the six intervention foods at age 3 years in the intention-to-treat analysis. But adherence to the demanding EAT early-introduction protocol was a problem. Indeed, only 43% of participants adhered to the study protocol. In a per-protocol analysis restricted to the adherent group, however, early introduction was associated with a highly significant 67% reduction in the relative risk of food allergy at 3 years of age compared to controls. And for the two most prevalent food allergies – to peanut and egg – the relative risk reductions in the early-introduction group were 100% and 75%, respectively.
The EAT results suggest that an effective preventive dose of peanut in infants at least 3 months of age is roughly 2 g of peanut protein per week, equivalent to just under 2 tsp of peanut butter, according to Dr. Lack.
Simultaneously with presentation of the LEAP-On and EAT trials in Los Angeles, the studies were published online at NEJM.org (doi: 10.1056/NEJMoa1514210 for LEAP-ON and 10.1056/NEJMoa1514209 for EAT).
LEAP-On was supported primarily by NIAID. EAT was funded mainly by the UK Foods Standards Agency and the Medical Research Council. Dr. Lack reported receiving grants from those agencies as well as Food Allergy Research and Education.
EXPERT ANALYSIS FROM THE 2016 AAAAI ANNUAL MEETING
Comorbid depression worsens asthma outcomes in older adults
LOS ANGELES – Adults over age 55 with asthma and depression have nearly twice as many emergency department visits for asthma as do asthma patients without depression, based on findings in 402 asthma patients, Dr. Pooja O. Patel reported at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
Comorbid depression also is associated with more asthma-related sleep disturbances and worse health-related quality of life, even though spirometry findings are similar in asthma patients with and without depression, added Dr. Patel of the University of Michigan, Ann Arbor.
She analyzed data on 7,256 adults over age 55 who participated in the National Health and Nutrition Examination Survey during 2007-2012. The prevalence of physician-diagnosed asthma in this nationally representative group of older adults was 5.5%. And 196 of those 402 asthma patients, or fully 49%, had comorbid depression as defined by their scores on the Patient Health Questionnaire-9 (PHQ-9), a validated brief and reliable self-administered measure of depression severity.
One or more emergency department visits for asthma within the last 12 months occurred in 18.8% of the group with asthma alone, compared with 28.1% in those with comorbid depression. In a multivariate regression analysis adjusted for the demographic differences, this translated to a twofold increased likelihood of ED visits specifically for asthma in the group with asthma and depression.
These data make a compelling case for routine screening for depression in older adults with asthma. The PHQ-9 is a good, simple tool for this purpose. Future studies will need to be done in order to learn whether early identification and treatment of comorbid depression in older asthmatic adults will result in improved asthma outcomes, but that is a reasonable hope, Dr. Patel added.
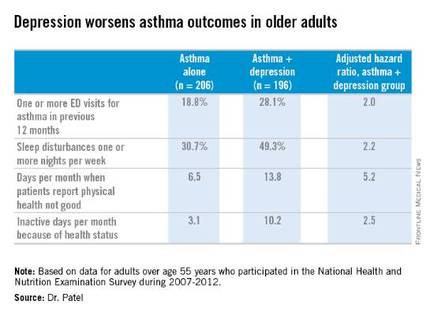
Why is asthma control worse in older adults with depression? In an interview, Dr. Patel said she thinks demographic differences may play a role. The older asthma patients with depression had less education and lower socioeconomic status than those without depression. Also, depression could adversely affect adherence to asthma controller medications, as depression is linked to worse medication adherence.
Dr. Patel reported having no financial conflicts regarding her study.
LOS ANGELES – Adults over age 55 with asthma and depression have nearly twice as many emergency department visits for asthma as do asthma patients without depression, based on findings in 402 asthma patients, Dr. Pooja O. Patel reported at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
Comorbid depression also is associated with more asthma-related sleep disturbances and worse health-related quality of life, even though spirometry findings are similar in asthma patients with and without depression, added Dr. Patel of the University of Michigan, Ann Arbor.
She analyzed data on 7,256 adults over age 55 who participated in the National Health and Nutrition Examination Survey during 2007-2012. The prevalence of physician-diagnosed asthma in this nationally representative group of older adults was 5.5%. And 196 of those 402 asthma patients, or fully 49%, had comorbid depression as defined by their scores on the Patient Health Questionnaire-9 (PHQ-9), a validated brief and reliable self-administered measure of depression severity.
One or more emergency department visits for asthma within the last 12 months occurred in 18.8% of the group with asthma alone, compared with 28.1% in those with comorbid depression. In a multivariate regression analysis adjusted for the demographic differences, this translated to a twofold increased likelihood of ED visits specifically for asthma in the group with asthma and depression.
These data make a compelling case for routine screening for depression in older adults with asthma. The PHQ-9 is a good, simple tool for this purpose. Future studies will need to be done in order to learn whether early identification and treatment of comorbid depression in older asthmatic adults will result in improved asthma outcomes, but that is a reasonable hope, Dr. Patel added.

Why is asthma control worse in older adults with depression? In an interview, Dr. Patel said she thinks demographic differences may play a role. The older asthma patients with depression had less education and lower socioeconomic status than those without depression. Also, depression could adversely affect adherence to asthma controller medications, as depression is linked to worse medication adherence.
Dr. Patel reported having no financial conflicts regarding her study.
LOS ANGELES – Adults over age 55 with asthma and depression have nearly twice as many emergency department visits for asthma as do asthma patients without depression, based on findings in 402 asthma patients, Dr. Pooja O. Patel reported at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
Comorbid depression also is associated with more asthma-related sleep disturbances and worse health-related quality of life, even though spirometry findings are similar in asthma patients with and without depression, added Dr. Patel of the University of Michigan, Ann Arbor.
She analyzed data on 7,256 adults over age 55 who participated in the National Health and Nutrition Examination Survey during 2007-2012. The prevalence of physician-diagnosed asthma in this nationally representative group of older adults was 5.5%. And 196 of those 402 asthma patients, or fully 49%, had comorbid depression as defined by their scores on the Patient Health Questionnaire-9 (PHQ-9), a validated brief and reliable self-administered measure of depression severity.
One or more emergency department visits for asthma within the last 12 months occurred in 18.8% of the group with asthma alone, compared with 28.1% in those with comorbid depression. In a multivariate regression analysis adjusted for the demographic differences, this translated to a twofold increased likelihood of ED visits specifically for asthma in the group with asthma and depression.
These data make a compelling case for routine screening for depression in older adults with asthma. The PHQ-9 is a good, simple tool for this purpose. Future studies will need to be done in order to learn whether early identification and treatment of comorbid depression in older asthmatic adults will result in improved asthma outcomes, but that is a reasonable hope, Dr. Patel added.

Why is asthma control worse in older adults with depression? In an interview, Dr. Patel said she thinks demographic differences may play a role. The older asthma patients with depression had less education and lower socioeconomic status than those without depression. Also, depression could adversely affect adherence to asthma controller medications, as depression is linked to worse medication adherence.
Dr. Patel reported having no financial conflicts regarding her study.
AT 2016 AAAAI ANNUAL MEETING
Key clinical point: Screening for depression in older adults with asthma has a high yield, with a 49% prevalence found in a nationally representative population.
Major finding: Older adults with asthma and depression were twice as likely as adults with asthma alone to have one or more emergency department visits for asthma in the past year.
Data source: An analysis of cross-sectional data on a nationally representative sample of 402 adults with physician-diagnosed asthma who participated in the National Health and Nutrition Examination Survey during 2007-2012.
Disclosures: The presenter reported having no financial conflicts regarding her study.
Cellfina 3-year data will show sustained effects on cellulite, researcher says
WAIKOLOA, HAWAII – Of the plethora of cellulite treatment devices on the market, the Food and Drug Administration has deemed only two capable of providing long-term improvement in the appearance of cellulite on the buttocks and thighs, Dr. Michael S. Kaminer said at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
These two devices are Cellulaze, a 1440 nm Nd:YAG laser typically utilized in conjunction with liposuction and marketed by Cynosure, and Cellfina, a semiautomated system for precise dermal undermining beneath cellulite dimples and marketed by Merz. Both devices target the structural problem that causes cellulite: subdermal fibrous septae that are tethered to the dermis, pulling down on the skin with resultant fat herniation and creation of cellulite dimples. The devices have the same mechanism of benefit, which entails cutting and thereby releasing the fibrous septae, albeit through two different technologies. Cellulaze cuts the fibrous septae with a laser fiber, while Cellfina uses a rapidly moving, piston-driven 18-gauge blade.
The superior efficacy of these two devices settles a long-standing controversy regarding the cause of cellulite, according to Dr. Kaminer, a dermatologist at Yale University, New Haven, Conn., and SkinCare Physicians of Chestnut Hill, Mass. “I think we now know what causes cellulite ... that if you cut the fibrous septae, things will improve.”
“There are a ton of devices (for treating cellulite) out there, but if you look at the literature none of them except Cellulaze and Cellfina have been shown to work,” he said. “Arguably the other options have, at most, limited efficacy.”
He cited a recent systematic evidence-based literature review led by Stefanie Luebberding, Ph.D., of the Rosenpark Clinic in Darmstadt, Germany. She and her coinvestigators concluded that “no clear evidence of good efficacy could be identified in any of the evaluated cellulite treatments,” which included shock wave and other forms of mechanical stimulation, massage, topical agents designed to plump up the skin, high- and low-energy laser therapies, infrared light, and radiofrequency (Am J Clin Dermatol. 2015 Aug;16[4]:243-56).
In U.S. clinical trials of Cellulaze, 68% of subjects had significant improvement at 1 year based on photographic evaluation and 65% based on Vectra three-dimensional surface imaging. Good to excellent results were reported by 76% of patients and 69% of physicians. On the downside, the Cellulaze device costs more than $100,000, recovery time is long, and the use of liposuction is falling in the United States, Dr. Kaminer observed.
He was the lead investigator in the multicenter study of Cellfina, in which 96% of patients in the open-label trial said they were satisfied or very satisfied with their results at 2 years post procedure.
From a mean baseline score of 3.4 on a 0-5 Cellulite Severity Scale, the average improvement was 2.0 points at both 1- and 2-year follow-up as assessed by independent physicians examining standardized professional photographs. Of 55 study participants, 2 experienced mild, transient adverse events (Dermatol Surg. 2015 Mar;41[3]:336-47).
Based on that trial, a single treatment session provides improvement lasting up to 1 year in the appearance of cellulite. Similar findings were noted 2 years after the procedure, said Dr. Kaminer, and he and his coinvestigators are now preparing to submit to the FDA the 3-year follow-up data, which show “essentially the same” sustained improvement.
Before the Cellfina procedure begins, the target cellulite dimples are marked on the standing patient. Roughly 20-25 dimples can be treated per session. The patient then assumes a prone position. A vacuum device is placed over the target area to control the depth and area of treatment. Tumescent anesthesia is injected, and then the piston-driven 18-gauge blade is introduced. The vacuum suction precisely controls the cutting depth at either 6 or 10 mm.
Dr. Kaminer noted that, potentially, the cellulite treatment market is colossal. One study concluded that an estimated 85% of American women age 24-54 have cellulite, and 37% of them – nearly 24 million women – say they are interested in a minimally invasive procedure to get rid of it.
“The definition of cellulite depends on what women you ask. Based on what I hear, 100% of women believe that they have cellulite and a lot of them are interested in having their cellulite treated,” he remarked.
Dr. Kaminer reported serving as a consultant to Merz, the maker of Cellfina, and receiving research funding from Cabochon, the maker of a system for improving the appearance of cellulite.
The SDEF and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – Of the plethora of cellulite treatment devices on the market, the Food and Drug Administration has deemed only two capable of providing long-term improvement in the appearance of cellulite on the buttocks and thighs, Dr. Michael S. Kaminer said at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
These two devices are Cellulaze, a 1440 nm Nd:YAG laser typically utilized in conjunction with liposuction and marketed by Cynosure, and Cellfina, a semiautomated system for precise dermal undermining beneath cellulite dimples and marketed by Merz. Both devices target the structural problem that causes cellulite: subdermal fibrous septae that are tethered to the dermis, pulling down on the skin with resultant fat herniation and creation of cellulite dimples. The devices have the same mechanism of benefit, which entails cutting and thereby releasing the fibrous septae, albeit through two different technologies. Cellulaze cuts the fibrous septae with a laser fiber, while Cellfina uses a rapidly moving, piston-driven 18-gauge blade.
The superior efficacy of these two devices settles a long-standing controversy regarding the cause of cellulite, according to Dr. Kaminer, a dermatologist at Yale University, New Haven, Conn., and SkinCare Physicians of Chestnut Hill, Mass. “I think we now know what causes cellulite ... that if you cut the fibrous septae, things will improve.”
“There are a ton of devices (for treating cellulite) out there, but if you look at the literature none of them except Cellulaze and Cellfina have been shown to work,” he said. “Arguably the other options have, at most, limited efficacy.”
He cited a recent systematic evidence-based literature review led by Stefanie Luebberding, Ph.D., of the Rosenpark Clinic in Darmstadt, Germany. She and her coinvestigators concluded that “no clear evidence of good efficacy could be identified in any of the evaluated cellulite treatments,” which included shock wave and other forms of mechanical stimulation, massage, topical agents designed to plump up the skin, high- and low-energy laser therapies, infrared light, and radiofrequency (Am J Clin Dermatol. 2015 Aug;16[4]:243-56).
In U.S. clinical trials of Cellulaze, 68% of subjects had significant improvement at 1 year based on photographic evaluation and 65% based on Vectra three-dimensional surface imaging. Good to excellent results were reported by 76% of patients and 69% of physicians. On the downside, the Cellulaze device costs more than $100,000, recovery time is long, and the use of liposuction is falling in the United States, Dr. Kaminer observed.
He was the lead investigator in the multicenter study of Cellfina, in which 96% of patients in the open-label trial said they were satisfied or very satisfied with their results at 2 years post procedure.
From a mean baseline score of 3.4 on a 0-5 Cellulite Severity Scale, the average improvement was 2.0 points at both 1- and 2-year follow-up as assessed by independent physicians examining standardized professional photographs. Of 55 study participants, 2 experienced mild, transient adverse events (Dermatol Surg. 2015 Mar;41[3]:336-47).
Based on that trial, a single treatment session provides improvement lasting up to 1 year in the appearance of cellulite. Similar findings were noted 2 years after the procedure, said Dr. Kaminer, and he and his coinvestigators are now preparing to submit to the FDA the 3-year follow-up data, which show “essentially the same” sustained improvement.
Before the Cellfina procedure begins, the target cellulite dimples are marked on the standing patient. Roughly 20-25 dimples can be treated per session. The patient then assumes a prone position. A vacuum device is placed over the target area to control the depth and area of treatment. Tumescent anesthesia is injected, and then the piston-driven 18-gauge blade is introduced. The vacuum suction precisely controls the cutting depth at either 6 or 10 mm.
Dr. Kaminer noted that, potentially, the cellulite treatment market is colossal. One study concluded that an estimated 85% of American women age 24-54 have cellulite, and 37% of them – nearly 24 million women – say they are interested in a minimally invasive procedure to get rid of it.
“The definition of cellulite depends on what women you ask. Based on what I hear, 100% of women believe that they have cellulite and a lot of them are interested in having their cellulite treated,” he remarked.
Dr. Kaminer reported serving as a consultant to Merz, the maker of Cellfina, and receiving research funding from Cabochon, the maker of a system for improving the appearance of cellulite.
The SDEF and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – Of the plethora of cellulite treatment devices on the market, the Food and Drug Administration has deemed only two capable of providing long-term improvement in the appearance of cellulite on the buttocks and thighs, Dr. Michael S. Kaminer said at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
These two devices are Cellulaze, a 1440 nm Nd:YAG laser typically utilized in conjunction with liposuction and marketed by Cynosure, and Cellfina, a semiautomated system for precise dermal undermining beneath cellulite dimples and marketed by Merz. Both devices target the structural problem that causes cellulite: subdermal fibrous septae that are tethered to the dermis, pulling down on the skin with resultant fat herniation and creation of cellulite dimples. The devices have the same mechanism of benefit, which entails cutting and thereby releasing the fibrous septae, albeit through two different technologies. Cellulaze cuts the fibrous septae with a laser fiber, while Cellfina uses a rapidly moving, piston-driven 18-gauge blade.
The superior efficacy of these two devices settles a long-standing controversy regarding the cause of cellulite, according to Dr. Kaminer, a dermatologist at Yale University, New Haven, Conn., and SkinCare Physicians of Chestnut Hill, Mass. “I think we now know what causes cellulite ... that if you cut the fibrous septae, things will improve.”
“There are a ton of devices (for treating cellulite) out there, but if you look at the literature none of them except Cellulaze and Cellfina have been shown to work,” he said. “Arguably the other options have, at most, limited efficacy.”
He cited a recent systematic evidence-based literature review led by Stefanie Luebberding, Ph.D., of the Rosenpark Clinic in Darmstadt, Germany. She and her coinvestigators concluded that “no clear evidence of good efficacy could be identified in any of the evaluated cellulite treatments,” which included shock wave and other forms of mechanical stimulation, massage, topical agents designed to plump up the skin, high- and low-energy laser therapies, infrared light, and radiofrequency (Am J Clin Dermatol. 2015 Aug;16[4]:243-56).
In U.S. clinical trials of Cellulaze, 68% of subjects had significant improvement at 1 year based on photographic evaluation and 65% based on Vectra three-dimensional surface imaging. Good to excellent results were reported by 76% of patients and 69% of physicians. On the downside, the Cellulaze device costs more than $100,000, recovery time is long, and the use of liposuction is falling in the United States, Dr. Kaminer observed.
He was the lead investigator in the multicenter study of Cellfina, in which 96% of patients in the open-label trial said they were satisfied or very satisfied with their results at 2 years post procedure.
From a mean baseline score of 3.4 on a 0-5 Cellulite Severity Scale, the average improvement was 2.0 points at both 1- and 2-year follow-up as assessed by independent physicians examining standardized professional photographs. Of 55 study participants, 2 experienced mild, transient adverse events (Dermatol Surg. 2015 Mar;41[3]:336-47).
Based on that trial, a single treatment session provides improvement lasting up to 1 year in the appearance of cellulite. Similar findings were noted 2 years after the procedure, said Dr. Kaminer, and he and his coinvestigators are now preparing to submit to the FDA the 3-year follow-up data, which show “essentially the same” sustained improvement.
Before the Cellfina procedure begins, the target cellulite dimples are marked on the standing patient. Roughly 20-25 dimples can be treated per session. The patient then assumes a prone position. A vacuum device is placed over the target area to control the depth and area of treatment. Tumescent anesthesia is injected, and then the piston-driven 18-gauge blade is introduced. The vacuum suction precisely controls the cutting depth at either 6 or 10 mm.
Dr. Kaminer noted that, potentially, the cellulite treatment market is colossal. One study concluded that an estimated 85% of American women age 24-54 have cellulite, and 37% of them – nearly 24 million women – say they are interested in a minimally invasive procedure to get rid of it.
“The definition of cellulite depends on what women you ask. Based on what I hear, 100% of women believe that they have cellulite and a lot of them are interested in having their cellulite treated,” he remarked.
Dr. Kaminer reported serving as a consultant to Merz, the maker of Cellfina, and receiving research funding from Cabochon, the maker of a system for improving the appearance of cellulite.
The SDEF and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF HAWAII DERMATOLOGY SEMINAR
New Tool Better Predicts Coronary Artery Disease in Lupus Patients
MAUI, HAWAII – Multiple studies have firmly established that the traditional Framingham Risk Score seriously underestimates the substantially increased risk of coronary artery disease present in patients with systemic lupus erythematosus (SLE). Nor is the American College of Cardiology/American Heart Association atherosclerotic cardiovascular disease risk score reliable in this population. But now physicians at the University of Toronto Lupus Clinic have come up with a more accurate risk prediction tool.
The new score is a straightforward modification of the Framingham Risk Score (FRS). It is calculated by multiplying the values for the items included in the classic FRS by two. It’s called, simply enough, the 2FRS, Dr. Dafna D. Gladman explained at the 2016 Rheumatology Winter Clinical Symposium.
In developing the 2FRS, she and her coinvestigators didn’t change the relative values of the individual components of the standard FRS. Instead, they calculated the standard baseline FRS values for 905 women with SLE who were prospectively followed at the Toronto Lupus Clinic. None had a prior history of CAD or diabetes when they enrolled in the cohort. Then the investigators tried multiplying those scores by 1.5, 2, 3, and 4 to learn which multiplier yielded the best sensitivity and specificity for prediction of coronary events, which occurred in 95 women during follow-up. Doubling the classic FRS score turned out to be the clear winner, according to Dr. Gladman, professor of medicine and codirector of the University of Toronto Lupus Clinic.
The classic FRS categorized 2.4% of the patients as being at moderate or high cardiovascular risk. With the 2FRS, this figure climbed to 17.3%. In a time-dependent covariant analysis for prediction of the first coronary event, a moderate/high score on the classic FRS was associated with a 3.2-fold increased risk, while a moderate/high score on the 2FRS conferred a 4.4-fold increased risk (J Rheumatol. 2016 Feb 15. doi: 10.3899/jrheum.150983).
The reason the classic FRS underestimates the true risk of atherosclerotic vascular events in lupus patients was illustrated in an earlier study by the Toronto group. Among 1,087 patients with no history of cardiovascular disease or diabetes when they enrolled at the clinic, 10.9% went on to experience an atherosclerotic vascular event during follow-up. In a multivariate analysis, disease-specific risk factors – namely, the presence of vasculitis or neuropsychiatric findings – proved to be more potent predictors of subsequent atherosclerotic vascular events than did the number of FRS risk factors or smoking status (J Rheumatol. 2007 Jan;34[1]:70-5).
It has long been known that mortality in SLE patients follows a bimodal pattern. Premature cardiovascular deaths explain a large portion of the second peak.
“In our total Toronto SLE cohort, we find there’s a fivefold increased risk for the development of acute MI compared to controls. The average age at which MI occurs is 69 for women in the general population, but it’s 49 for women in our lupus cohort,” according to Dr. Gladman.
In yet another study focusing on atherosclerotic comorbidity in lupus patients, the Toronto group showed that carotid total plaque area as measured by high-resolution ultrasound correlated significantly better – by nearly fivefold – with clinical ischemic cardiovascular events than did carotid intima-media thickness, suggesting total plaque area may be the better tool for investigating atherosclerotic vascular disease as expressed in SLE patients. Carotid plaque also displayed a much stronger correlation with conventional cardiovascular risk factors, including hypertension and elevated LDL cholesterol, in the lupus patients (Lupus. 2014 Oct;23[11]:1142-8).
Dr. Gladman reported receiving research grants from GlaxoSmithKline and Janssen and serving as a consultant to those pharmaceutical companies as well as to Bristol-Myers Squibb and UCB.
MAUI, HAWAII – Multiple studies have firmly established that the traditional Framingham Risk Score seriously underestimates the substantially increased risk of coronary artery disease present in patients with systemic lupus erythematosus (SLE). Nor is the American College of Cardiology/American Heart Association atherosclerotic cardiovascular disease risk score reliable in this population. But now physicians at the University of Toronto Lupus Clinic have come up with a more accurate risk prediction tool.
The new score is a straightforward modification of the Framingham Risk Score (FRS). It is calculated by multiplying the values for the items included in the classic FRS by two. It’s called, simply enough, the 2FRS, Dr. Dafna D. Gladman explained at the 2016 Rheumatology Winter Clinical Symposium.
In developing the 2FRS, she and her coinvestigators didn’t change the relative values of the individual components of the standard FRS. Instead, they calculated the standard baseline FRS values for 905 women with SLE who were prospectively followed at the Toronto Lupus Clinic. None had a prior history of CAD or diabetes when they enrolled in the cohort. Then the investigators tried multiplying those scores by 1.5, 2, 3, and 4 to learn which multiplier yielded the best sensitivity and specificity for prediction of coronary events, which occurred in 95 women during follow-up. Doubling the classic FRS score turned out to be the clear winner, according to Dr. Gladman, professor of medicine and codirector of the University of Toronto Lupus Clinic.
The classic FRS categorized 2.4% of the patients as being at moderate or high cardiovascular risk. With the 2FRS, this figure climbed to 17.3%. In a time-dependent covariant analysis for prediction of the first coronary event, a moderate/high score on the classic FRS was associated with a 3.2-fold increased risk, while a moderate/high score on the 2FRS conferred a 4.4-fold increased risk (J Rheumatol. 2016 Feb 15. doi: 10.3899/jrheum.150983).
The reason the classic FRS underestimates the true risk of atherosclerotic vascular events in lupus patients was illustrated in an earlier study by the Toronto group. Among 1,087 patients with no history of cardiovascular disease or diabetes when they enrolled at the clinic, 10.9% went on to experience an atherosclerotic vascular event during follow-up. In a multivariate analysis, disease-specific risk factors – namely, the presence of vasculitis or neuropsychiatric findings – proved to be more potent predictors of subsequent atherosclerotic vascular events than did the number of FRS risk factors or smoking status (J Rheumatol. 2007 Jan;34[1]:70-5).
It has long been known that mortality in SLE patients follows a bimodal pattern. Premature cardiovascular deaths explain a large portion of the second peak.
“In our total Toronto SLE cohort, we find there’s a fivefold increased risk for the development of acute MI compared to controls. The average age at which MI occurs is 69 for women in the general population, but it’s 49 for women in our lupus cohort,” according to Dr. Gladman.
In yet another study focusing on atherosclerotic comorbidity in lupus patients, the Toronto group showed that carotid total plaque area as measured by high-resolution ultrasound correlated significantly better – by nearly fivefold – with clinical ischemic cardiovascular events than did carotid intima-media thickness, suggesting total plaque area may be the better tool for investigating atherosclerotic vascular disease as expressed in SLE patients. Carotid plaque also displayed a much stronger correlation with conventional cardiovascular risk factors, including hypertension and elevated LDL cholesterol, in the lupus patients (Lupus. 2014 Oct;23[11]:1142-8).
Dr. Gladman reported receiving research grants from GlaxoSmithKline and Janssen and serving as a consultant to those pharmaceutical companies as well as to Bristol-Myers Squibb and UCB.
MAUI, HAWAII – Multiple studies have firmly established that the traditional Framingham Risk Score seriously underestimates the substantially increased risk of coronary artery disease present in patients with systemic lupus erythematosus (SLE). Nor is the American College of Cardiology/American Heart Association atherosclerotic cardiovascular disease risk score reliable in this population. But now physicians at the University of Toronto Lupus Clinic have come up with a more accurate risk prediction tool.
The new score is a straightforward modification of the Framingham Risk Score (FRS). It is calculated by multiplying the values for the items included in the classic FRS by two. It’s called, simply enough, the 2FRS, Dr. Dafna D. Gladman explained at the 2016 Rheumatology Winter Clinical Symposium.
In developing the 2FRS, she and her coinvestigators didn’t change the relative values of the individual components of the standard FRS. Instead, they calculated the standard baseline FRS values for 905 women with SLE who were prospectively followed at the Toronto Lupus Clinic. None had a prior history of CAD or diabetes when they enrolled in the cohort. Then the investigators tried multiplying those scores by 1.5, 2, 3, and 4 to learn which multiplier yielded the best sensitivity and specificity for prediction of coronary events, which occurred in 95 women during follow-up. Doubling the classic FRS score turned out to be the clear winner, according to Dr. Gladman, professor of medicine and codirector of the University of Toronto Lupus Clinic.
The classic FRS categorized 2.4% of the patients as being at moderate or high cardiovascular risk. With the 2FRS, this figure climbed to 17.3%. In a time-dependent covariant analysis for prediction of the first coronary event, a moderate/high score on the classic FRS was associated with a 3.2-fold increased risk, while a moderate/high score on the 2FRS conferred a 4.4-fold increased risk (J Rheumatol. 2016 Feb 15. doi: 10.3899/jrheum.150983).
The reason the classic FRS underestimates the true risk of atherosclerotic vascular events in lupus patients was illustrated in an earlier study by the Toronto group. Among 1,087 patients with no history of cardiovascular disease or diabetes when they enrolled at the clinic, 10.9% went on to experience an atherosclerotic vascular event during follow-up. In a multivariate analysis, disease-specific risk factors – namely, the presence of vasculitis or neuropsychiatric findings – proved to be more potent predictors of subsequent atherosclerotic vascular events than did the number of FRS risk factors or smoking status (J Rheumatol. 2007 Jan;34[1]:70-5).
It has long been known that mortality in SLE patients follows a bimodal pattern. Premature cardiovascular deaths explain a large portion of the second peak.
“In our total Toronto SLE cohort, we find there’s a fivefold increased risk for the development of acute MI compared to controls. The average age at which MI occurs is 69 for women in the general population, but it’s 49 for women in our lupus cohort,” according to Dr. Gladman.
In yet another study focusing on atherosclerotic comorbidity in lupus patients, the Toronto group showed that carotid total plaque area as measured by high-resolution ultrasound correlated significantly better – by nearly fivefold – with clinical ischemic cardiovascular events than did carotid intima-media thickness, suggesting total plaque area may be the better tool for investigating atherosclerotic vascular disease as expressed in SLE patients. Carotid plaque also displayed a much stronger correlation with conventional cardiovascular risk factors, including hypertension and elevated LDL cholesterol, in the lupus patients (Lupus. 2014 Oct;23[11]:1142-8).
Dr. Gladman reported receiving research grants from GlaxoSmithKline and Janssen and serving as a consultant to those pharmaceutical companies as well as to Bristol-Myers Squibb and UCB.
EXPERT ANALYSIS FROM RWCS 2016
New tool better predicts coronary artery disease in lupus patients
MAUI, HAWAII – Multiple studies have firmly established that the traditional Framingham Risk Score seriously underestimates the substantially increased risk of coronary artery disease present in patients with systemic lupus erythematosus (SLE). Nor is the American College of Cardiology/American Heart Association atherosclerotic cardiovascular disease risk score reliable in this population. But now physicians at the University of Toronto Lupus Clinic have come up with a more accurate risk prediction tool.
The new score is a straightforward modification of the Framingham Risk Score (FRS). It is calculated by multiplying the values for the items included in the classic FRS by two. It’s called, simply enough, the 2FRS, Dr. Dafna D. Gladman explained at the 2016 Rheumatology Winter Clinical Symposium.
In developing the 2FRS, she and her coinvestigators didn’t change the relative values of the individual components of the standard FRS. Instead, they calculated the standard baseline FRS values for 905 women with SLE who were prospectively followed at the Toronto Lupus Clinic. None had a prior history of CAD or diabetes when they enrolled in the cohort. Then the investigators tried multiplying those scores by 1.5, 2, 3, and 4 to learn which multiplier yielded the best sensitivity and specificity for prediction of coronary events, which occurred in 95 women during follow-up. Doubling the classic FRS score turned out to be the clear winner, according to Dr. Gladman, professor of medicine and codirector of the University of Toronto Lupus Clinic.
The classic FRS categorized 2.4% of the patients as being at moderate or high cardiovascular risk. With the 2FRS, this figure climbed to 17.3%. In a time-dependent covariant analysis for prediction of the first coronary event, a moderate/high score on the classic FRS was associated with a 3.2-fold increased risk, while a moderate/high score on the 2FRS conferred a 4.4-fold increased risk (J Rheumatol. 2016 Feb 15. doi: 10.3899/jrheum.150983).
The reason the classic FRS underestimates the true risk of atherosclerotic vascular events in lupus patients was illustrated in an earlier study by the Toronto group. Among 1,087 patients with no history of cardiovascular disease or diabetes when they enrolled at the clinic, 10.9% went on to experience an atherosclerotic vascular event during follow-up. In a multivariate analysis, disease-specific risk factors – namely, the presence of vasculitis or neuropsychiatric findings – proved to be more potent predictors of subsequent atherosclerotic vascular events than did the number of FRS risk factors or smoking status (J Rheumatol. 2007 Jan;34[1]:70-5).
It has long been known that mortality in SLE patients follows a bimodal pattern. Premature cardiovascular deaths explain a large portion of the second peak.
“In our total Toronto SLE cohort, we find there’s a fivefold increased risk for the development of acute MI compared to controls. The average age at which MI occurs is 69 for women in the general population, but it’s 49 for women in our lupus cohort,” according to Dr. Gladman.
In yet another study focusing on atherosclerotic comorbidity in lupus patients, the Toronto group showed that carotid total plaque area as measured by high-resolution ultrasound correlated significantly better – by nearly fivefold – with clinical ischemic cardiovascular events than did carotid intima-media thickness, suggesting total plaque area may be the better tool for investigating atherosclerotic vascular disease as expressed in SLE patients. Carotid plaque also displayed a much stronger correlation with conventional cardiovascular risk factors, including hypertension and elevated LDL cholesterol, in the lupus patients (Lupus. 2014 Oct;23[11]:1142-8).
Dr. Gladman reported receiving research grants from GlaxoSmithKline and Janssen and serving as a consultant to those pharmaceutical companies as well as to Bristol-Myers Squibb and UCB.
MAUI, HAWAII – Multiple studies have firmly established that the traditional Framingham Risk Score seriously underestimates the substantially increased risk of coronary artery disease present in patients with systemic lupus erythematosus (SLE). Nor is the American College of Cardiology/American Heart Association atherosclerotic cardiovascular disease risk score reliable in this population. But now physicians at the University of Toronto Lupus Clinic have come up with a more accurate risk prediction tool.
The new score is a straightforward modification of the Framingham Risk Score (FRS). It is calculated by multiplying the values for the items included in the classic FRS by two. It’s called, simply enough, the 2FRS, Dr. Dafna D. Gladman explained at the 2016 Rheumatology Winter Clinical Symposium.
In developing the 2FRS, she and her coinvestigators didn’t change the relative values of the individual components of the standard FRS. Instead, they calculated the standard baseline FRS values for 905 women with SLE who were prospectively followed at the Toronto Lupus Clinic. None had a prior history of CAD or diabetes when they enrolled in the cohort. Then the investigators tried multiplying those scores by 1.5, 2, 3, and 4 to learn which multiplier yielded the best sensitivity and specificity for prediction of coronary events, which occurred in 95 women during follow-up. Doubling the classic FRS score turned out to be the clear winner, according to Dr. Gladman, professor of medicine and codirector of the University of Toronto Lupus Clinic.
The classic FRS categorized 2.4% of the patients as being at moderate or high cardiovascular risk. With the 2FRS, this figure climbed to 17.3%. In a time-dependent covariant analysis for prediction of the first coronary event, a moderate/high score on the classic FRS was associated with a 3.2-fold increased risk, while a moderate/high score on the 2FRS conferred a 4.4-fold increased risk (J Rheumatol. 2016 Feb 15. doi: 10.3899/jrheum.150983).
The reason the classic FRS underestimates the true risk of atherosclerotic vascular events in lupus patients was illustrated in an earlier study by the Toronto group. Among 1,087 patients with no history of cardiovascular disease or diabetes when they enrolled at the clinic, 10.9% went on to experience an atherosclerotic vascular event during follow-up. In a multivariate analysis, disease-specific risk factors – namely, the presence of vasculitis or neuropsychiatric findings – proved to be more potent predictors of subsequent atherosclerotic vascular events than did the number of FRS risk factors or smoking status (J Rheumatol. 2007 Jan;34[1]:70-5).
It has long been known that mortality in SLE patients follows a bimodal pattern. Premature cardiovascular deaths explain a large portion of the second peak.
“In our total Toronto SLE cohort, we find there’s a fivefold increased risk for the development of acute MI compared to controls. The average age at which MI occurs is 69 for women in the general population, but it’s 49 for women in our lupus cohort,” according to Dr. Gladman.
In yet another study focusing on atherosclerotic comorbidity in lupus patients, the Toronto group showed that carotid total plaque area as measured by high-resolution ultrasound correlated significantly better – by nearly fivefold – with clinical ischemic cardiovascular events than did carotid intima-media thickness, suggesting total plaque area may be the better tool for investigating atherosclerotic vascular disease as expressed in SLE patients. Carotid plaque also displayed a much stronger correlation with conventional cardiovascular risk factors, including hypertension and elevated LDL cholesterol, in the lupus patients (Lupus. 2014 Oct;23[11]:1142-8).
Dr. Gladman reported receiving research grants from GlaxoSmithKline and Janssen and serving as a consultant to those pharmaceutical companies as well as to Bristol-Myers Squibb and UCB.
MAUI, HAWAII – Multiple studies have firmly established that the traditional Framingham Risk Score seriously underestimates the substantially increased risk of coronary artery disease present in patients with systemic lupus erythematosus (SLE). Nor is the American College of Cardiology/American Heart Association atherosclerotic cardiovascular disease risk score reliable in this population. But now physicians at the University of Toronto Lupus Clinic have come up with a more accurate risk prediction tool.
The new score is a straightforward modification of the Framingham Risk Score (FRS). It is calculated by multiplying the values for the items included in the classic FRS by two. It’s called, simply enough, the 2FRS, Dr. Dafna D. Gladman explained at the 2016 Rheumatology Winter Clinical Symposium.
In developing the 2FRS, she and her coinvestigators didn’t change the relative values of the individual components of the standard FRS. Instead, they calculated the standard baseline FRS values for 905 women with SLE who were prospectively followed at the Toronto Lupus Clinic. None had a prior history of CAD or diabetes when they enrolled in the cohort. Then the investigators tried multiplying those scores by 1.5, 2, 3, and 4 to learn which multiplier yielded the best sensitivity and specificity for prediction of coronary events, which occurred in 95 women during follow-up. Doubling the classic FRS score turned out to be the clear winner, according to Dr. Gladman, professor of medicine and codirector of the University of Toronto Lupus Clinic.
The classic FRS categorized 2.4% of the patients as being at moderate or high cardiovascular risk. With the 2FRS, this figure climbed to 17.3%. In a time-dependent covariant analysis for prediction of the first coronary event, a moderate/high score on the classic FRS was associated with a 3.2-fold increased risk, while a moderate/high score on the 2FRS conferred a 4.4-fold increased risk (J Rheumatol. 2016 Feb 15. doi: 10.3899/jrheum.150983).
The reason the classic FRS underestimates the true risk of atherosclerotic vascular events in lupus patients was illustrated in an earlier study by the Toronto group. Among 1,087 patients with no history of cardiovascular disease or diabetes when they enrolled at the clinic, 10.9% went on to experience an atherosclerotic vascular event during follow-up. In a multivariate analysis, disease-specific risk factors – namely, the presence of vasculitis or neuropsychiatric findings – proved to be more potent predictors of subsequent atherosclerotic vascular events than did the number of FRS risk factors or smoking status (J Rheumatol. 2007 Jan;34[1]:70-5).
It has long been known that mortality in SLE patients follows a bimodal pattern. Premature cardiovascular deaths explain a large portion of the second peak.
“In our total Toronto SLE cohort, we find there’s a fivefold increased risk for the development of acute MI compared to controls. The average age at which MI occurs is 69 for women in the general population, but it’s 49 for women in our lupus cohort,” according to Dr. Gladman.
In yet another study focusing on atherosclerotic comorbidity in lupus patients, the Toronto group showed that carotid total plaque area as measured by high-resolution ultrasound correlated significantly better – by nearly fivefold – with clinical ischemic cardiovascular events than did carotid intima-media thickness, suggesting total plaque area may be the better tool for investigating atherosclerotic vascular disease as expressed in SLE patients. Carotid plaque also displayed a much stronger correlation with conventional cardiovascular risk factors, including hypertension and elevated LDL cholesterol, in the lupus patients (Lupus. 2014 Oct;23[11]:1142-8).
Dr. Gladman reported receiving research grants from GlaxoSmithKline and Janssen and serving as a consultant to those pharmaceutical companies as well as to Bristol-Myers Squibb and UCB.
EXPERT ANALYSIS FROM RWCS 2016
Expert advises how to use shingles vaccine in rheumatology patients
MAUI, HAWAII – The herpes zoster vaccine is particularly important in patients with rheumatic diseases because their risks of shingles and postherpetic neuralgia are substantially higher than in the general population, Dr. John J. Cush observed at the 2016 Rheumatology Winter Clinical Symposium.
This is a live attenuated virus vaccine, and the rules regarding its use in patients with rheumatic diseases are fairly complicated. Here’s what physicians need to know: the Centers for Disease Control and Prevention and the Advisory Committee on Immunization Practices say the shingles vaccine can safely be given to patients on prednisone at less than 20 mg/day, azathioprine at up to 3 mg/kg/day, or methotrexate at up to 0.4 mg/kg/week, which works out to about 25 mg/week in anyone weighing more than 136 pounds.
However, the shingles vaccine is contraindicated in patients on recombinant biologic agents, including tumor necrosis factor (TNF) inhibitors, abatacept (Orencia), rituximab (Rituxan), or Janus kinase inhibitors, explained Dr. Cush, professor of medicine and rheumatology at Baylor University, Dallas, and director of clinical rheumatology at the Baylor Research Institute.
The lifetime risk of shingles in the general population is roughly one in three. The risk in patients with rheumatoid arthritis is roughly twice that of the age-matched general population, and the risks are substantially greater than that in individuals with other rheumatic diseases, including lupus and granulomatosis with polyangiitis.
Payers cover the vaccine in patients age 60 or older. The vaccine is approved for and has been shown to be effective in 50- to 59-year-olds as well, but that typically entails an out-of-pocket expense of around $200.
Given that close to 60% of all rheumatoid arthritis patients will eventually be placed on biologic therapy, Dr. Cush believes in seizing any opportunity to give the shingles vaccine in age-appropriate patients beforehand. However, he advises against temporarily stopping a biologic for the express purpose of administering the live virus vaccine.
“Find the opportunity: between changes in medication, after they have surgery, during a lapse in therapy,” he suggested.
It’s recommended that Zostavax be deferred until after a patient has been off biologic therapy or high-dose steroids for at least 4 weeks, and that a biologic agent shouldn’t be started for 2-4 weeks after vaccination.
The shingles vaccine can safely be given with multiple inactivated virus vaccines such as an influenza vaccine or pneumococcal vaccine on a single day.
Controversy surrounds the issue of whether TNF inhibitors increase the risk of shingles. Several retrospective studies have reported they do. But the largest retrospective study, involving more than 33,000 new users of anti-TNF agents, found that patients with rheumatoid arthritis, psoriasis, psoriatic arthritis, ankylosing spondylitis, and other inflammatory diseases who initiated anti-TNF therapy weren’t at any higher risk of herpes zoster than those who started on methotrexate or other nonbiologic disease-modifying antirheumatic drugs (JAMA. 2013 Mar 6;309[9]:887-95). The lead investigator in this study, Dr. Kevin L. Winthrop of Oregon Health and Science University, Portland, is currently conducting a prospective study in an effort to confirm these findings.
The recommendation against giving the zoster vaccine to patients while on biologics is based upon the theoretical risk that exposure to the live attenuated virus will trigger an acute shingles attack. However, when Dr. Cush conducted a survey of his fellow rheumatologists, they reported that among more than a collective 200 patients inadvertently given the vaccine while on biologic therapy, not one case of shingles subsequently occurred over the short term.
More persuasively, Dr. Jeffrey R. Curtis of the University of Alabama at Birmingham and coinvestigators conducted a formal retrospective study of close to a half-million Medicare patients and found there were no cases of herpes zoster or varicella within 42 days following inadvertent vaccination of 633 patients while on biologics. During a median 2-year follow-up of Medicare patients with rheumatoid arthritis and other immune-mediated diseases, herpes zoster vaccination was associated with a 39% reduction in the risk of shingles (JAMA. 2012 Jul 4;308[1]:43-9).
“You shouldn’t be vaccinating for herpes zoster while patients are on a biologic, but you know what? If it happens, don’t wig out. Move on and try to avoid it,” Dr. Cush advised.
In any event, this is an issue that is eventually likely to go away. An inactivated virus vaccine for the prevention of shingles is now in clinical trials. It appears to be more effective than the current vaccine, according to the rheumatologist.
He reported having no financial interests relevant to his presentation.
MAUI, HAWAII – The herpes zoster vaccine is particularly important in patients with rheumatic diseases because their risks of shingles and postherpetic neuralgia are substantially higher than in the general population, Dr. John J. Cush observed at the 2016 Rheumatology Winter Clinical Symposium.
This is a live attenuated virus vaccine, and the rules regarding its use in patients with rheumatic diseases are fairly complicated. Here’s what physicians need to know: the Centers for Disease Control and Prevention and the Advisory Committee on Immunization Practices say the shingles vaccine can safely be given to patients on prednisone at less than 20 mg/day, azathioprine at up to 3 mg/kg/day, or methotrexate at up to 0.4 mg/kg/week, which works out to about 25 mg/week in anyone weighing more than 136 pounds.
However, the shingles vaccine is contraindicated in patients on recombinant biologic agents, including tumor necrosis factor (TNF) inhibitors, abatacept (Orencia), rituximab (Rituxan), or Janus kinase inhibitors, explained Dr. Cush, professor of medicine and rheumatology at Baylor University, Dallas, and director of clinical rheumatology at the Baylor Research Institute.
The lifetime risk of shingles in the general population is roughly one in three. The risk in patients with rheumatoid arthritis is roughly twice that of the age-matched general population, and the risks are substantially greater than that in individuals with other rheumatic diseases, including lupus and granulomatosis with polyangiitis.
Payers cover the vaccine in patients age 60 or older. The vaccine is approved for and has been shown to be effective in 50- to 59-year-olds as well, but that typically entails an out-of-pocket expense of around $200.
Given that close to 60% of all rheumatoid arthritis patients will eventually be placed on biologic therapy, Dr. Cush believes in seizing any opportunity to give the shingles vaccine in age-appropriate patients beforehand. However, he advises against temporarily stopping a biologic for the express purpose of administering the live virus vaccine.
“Find the opportunity: between changes in medication, after they have surgery, during a lapse in therapy,” he suggested.
It’s recommended that Zostavax be deferred until after a patient has been off biologic therapy or high-dose steroids for at least 4 weeks, and that a biologic agent shouldn’t be started for 2-4 weeks after vaccination.
The shingles vaccine can safely be given with multiple inactivated virus vaccines such as an influenza vaccine or pneumococcal vaccine on a single day.
Controversy surrounds the issue of whether TNF inhibitors increase the risk of shingles. Several retrospective studies have reported they do. But the largest retrospective study, involving more than 33,000 new users of anti-TNF agents, found that patients with rheumatoid arthritis, psoriasis, psoriatic arthritis, ankylosing spondylitis, and other inflammatory diseases who initiated anti-TNF therapy weren’t at any higher risk of herpes zoster than those who started on methotrexate or other nonbiologic disease-modifying antirheumatic drugs (JAMA. 2013 Mar 6;309[9]:887-95). The lead investigator in this study, Dr. Kevin L. Winthrop of Oregon Health and Science University, Portland, is currently conducting a prospective study in an effort to confirm these findings.
The recommendation against giving the zoster vaccine to patients while on biologics is based upon the theoretical risk that exposure to the live attenuated virus will trigger an acute shingles attack. However, when Dr. Cush conducted a survey of his fellow rheumatologists, they reported that among more than a collective 200 patients inadvertently given the vaccine while on biologic therapy, not one case of shingles subsequently occurred over the short term.
More persuasively, Dr. Jeffrey R. Curtis of the University of Alabama at Birmingham and coinvestigators conducted a formal retrospective study of close to a half-million Medicare patients and found there were no cases of herpes zoster or varicella within 42 days following inadvertent vaccination of 633 patients while on biologics. During a median 2-year follow-up of Medicare patients with rheumatoid arthritis and other immune-mediated diseases, herpes zoster vaccination was associated with a 39% reduction in the risk of shingles (JAMA. 2012 Jul 4;308[1]:43-9).
“You shouldn’t be vaccinating for herpes zoster while patients are on a biologic, but you know what? If it happens, don’t wig out. Move on and try to avoid it,” Dr. Cush advised.
In any event, this is an issue that is eventually likely to go away. An inactivated virus vaccine for the prevention of shingles is now in clinical trials. It appears to be more effective than the current vaccine, according to the rheumatologist.
He reported having no financial interests relevant to his presentation.
MAUI, HAWAII – The herpes zoster vaccine is particularly important in patients with rheumatic diseases because their risks of shingles and postherpetic neuralgia are substantially higher than in the general population, Dr. John J. Cush observed at the 2016 Rheumatology Winter Clinical Symposium.
This is a live attenuated virus vaccine, and the rules regarding its use in patients with rheumatic diseases are fairly complicated. Here’s what physicians need to know: the Centers for Disease Control and Prevention and the Advisory Committee on Immunization Practices say the shingles vaccine can safely be given to patients on prednisone at less than 20 mg/day, azathioprine at up to 3 mg/kg/day, or methotrexate at up to 0.4 mg/kg/week, which works out to about 25 mg/week in anyone weighing more than 136 pounds.
However, the shingles vaccine is contraindicated in patients on recombinant biologic agents, including tumor necrosis factor (TNF) inhibitors, abatacept (Orencia), rituximab (Rituxan), or Janus kinase inhibitors, explained Dr. Cush, professor of medicine and rheumatology at Baylor University, Dallas, and director of clinical rheumatology at the Baylor Research Institute.
The lifetime risk of shingles in the general population is roughly one in three. The risk in patients with rheumatoid arthritis is roughly twice that of the age-matched general population, and the risks are substantially greater than that in individuals with other rheumatic diseases, including lupus and granulomatosis with polyangiitis.
Payers cover the vaccine in patients age 60 or older. The vaccine is approved for and has been shown to be effective in 50- to 59-year-olds as well, but that typically entails an out-of-pocket expense of around $200.
Given that close to 60% of all rheumatoid arthritis patients will eventually be placed on biologic therapy, Dr. Cush believes in seizing any opportunity to give the shingles vaccine in age-appropriate patients beforehand. However, he advises against temporarily stopping a biologic for the express purpose of administering the live virus vaccine.
“Find the opportunity: between changes in medication, after they have surgery, during a lapse in therapy,” he suggested.
It’s recommended that Zostavax be deferred until after a patient has been off biologic therapy or high-dose steroids for at least 4 weeks, and that a biologic agent shouldn’t be started for 2-4 weeks after vaccination.
The shingles vaccine can safely be given with multiple inactivated virus vaccines such as an influenza vaccine or pneumococcal vaccine on a single day.
Controversy surrounds the issue of whether TNF inhibitors increase the risk of shingles. Several retrospective studies have reported they do. But the largest retrospective study, involving more than 33,000 new users of anti-TNF agents, found that patients with rheumatoid arthritis, psoriasis, psoriatic arthritis, ankylosing spondylitis, and other inflammatory diseases who initiated anti-TNF therapy weren’t at any higher risk of herpes zoster than those who started on methotrexate or other nonbiologic disease-modifying antirheumatic drugs (JAMA. 2013 Mar 6;309[9]:887-95). The lead investigator in this study, Dr. Kevin L. Winthrop of Oregon Health and Science University, Portland, is currently conducting a prospective study in an effort to confirm these findings.
The recommendation against giving the zoster vaccine to patients while on biologics is based upon the theoretical risk that exposure to the live attenuated virus will trigger an acute shingles attack. However, when Dr. Cush conducted a survey of his fellow rheumatologists, they reported that among more than a collective 200 patients inadvertently given the vaccine while on biologic therapy, not one case of shingles subsequently occurred over the short term.
More persuasively, Dr. Jeffrey R. Curtis of the University of Alabama at Birmingham and coinvestigators conducted a formal retrospective study of close to a half-million Medicare patients and found there were no cases of herpes zoster or varicella within 42 days following inadvertent vaccination of 633 patients while on biologics. During a median 2-year follow-up of Medicare patients with rheumatoid arthritis and other immune-mediated diseases, herpes zoster vaccination was associated with a 39% reduction in the risk of shingles (JAMA. 2012 Jul 4;308[1]:43-9).
“You shouldn’t be vaccinating for herpes zoster while patients are on a biologic, but you know what? If it happens, don’t wig out. Move on and try to avoid it,” Dr. Cush advised.
In any event, this is an issue that is eventually likely to go away. An inactivated virus vaccine for the prevention of shingles is now in clinical trials. It appears to be more effective than the current vaccine, according to the rheumatologist.
He reported having no financial interests relevant to his presentation.
EXPERT ANALYSIS FROM RWCS 2016


























