User login
Gyn. oncologists are in demand for robotic hysterectomy
SAN DIEGO – The demand for gynecologic oncologists to perform robotic hysterectomies – even for benign indications – has increased to the point that additional fellowship training spots will be necessary to meet the need, Dr. Kayla M. Wishall said at the annual meeting of the Society of Gynecologic Oncology.
More and more patients want their hysterectomies performed robotically. They find the high-quality optics and minimally invasive nature of the robotic procedure appealing – smaller incisions, less blood loss, shorter hospital stay, and faster recovery. And gynecologic oncologists are getting an increasing number of referrals because of their special expertise in robotic surgery and extensive experience with higher-risk patients, explained Dr. Wishall, a gynecologic oncologist at Hahnemann University Hospital/Drexel University in Philadelphia.
“This trend will likely tax the limited resources of gynecologic oncologists,” she added.
Another possible reason for the growing demand for gynecologic oncologist–performed robotic hysterectomies is that these subspecialists achieve better outcomes than gynecologists who do robotic hysterectomies, at least according to the findings of a retrospective study performed by Dr. Wishall, which included all of the 468 robotic hysterectomies performed at a large academic medical center in a recent 5-year period.
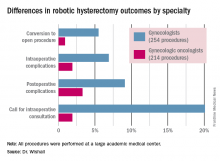
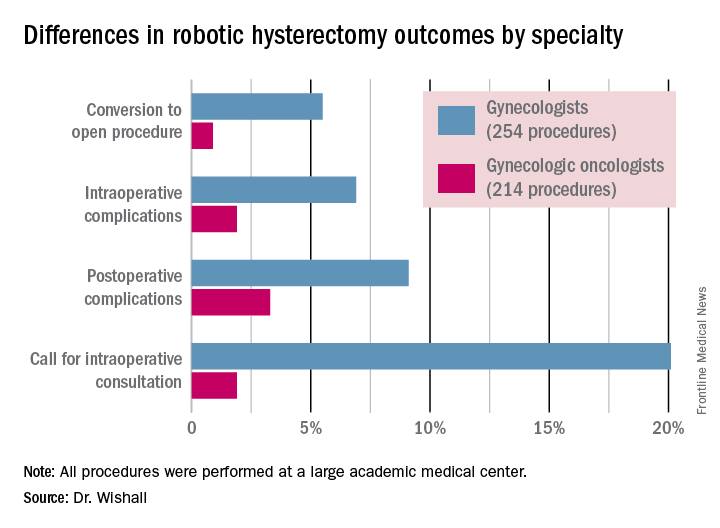
Gynecologic oncologists performed 64 (16.5%) of the 387 robotic hysterectomies done for benign indications. All told, gynecologists did 254 of the robotic hysterectomies; gynecologic oncologists performed 214.
Even though patients referred to gynecologic oncologists for these procedures were older, heavier, more likely to have had previous abdominal surgery, more often members of racial minorities, and had a higher prevalence of cardiac comorbidities, they experienced significantly fewer intra- and postoperative complications than patients whose robotic hysterectomies were performed by gynecologists, Dr. Wishall reported.
The combined intraoperative and postoperative complication rate for robotic hysterectomies performed by gynecologic oncologists was 5.2%, compared with 16% for gynecologists. But the rate of cardiac comorbidities, for instance, was 36.4% among patients seeing gynecologic oncologists, compared with 23.6% among those seeing gynecologists.
Moreover, gynecologists were about 10-fold more likely than gynecologic oncologists to call for an intraoperative consultation and sixfold more likely to convert their robotic hysterectomy to an open procedure. Their average operating room time was about 40% longer (244 minutes versus 171 minutes), too, in this single-center experience.
Dr. Wishall reported having no financial conflicts related to her study, which was conducted free of commercial support.
I read this article initially with amusement and then with outrage and disdain. This article summarizes the single-site, retrospective study by Dr. Kayla Wishall at the annual meeting of the Society of Gynecologic Oncology. Not only is this nonscience, but nonsensical science. As a single center retrospective study, conclusions must be suspect.

|
Dr. Charles E. Miller |
The comparison numbers of the two groups are small. While confounders would appear to be greater in the oncology group, we know nothing about the difficulty of the surgeries themselves – size of uterus, adnexal disease, endometriosis, pelvic adhesions, etc. Oftentimes, gynecologic oncologists dealing with endometrial carcinoma are going to face a less difficult challenge than a generalist dealing with an 18-weeks–size uterus in a woman who has undergone three prior C-sections, an open myomectomy, or stage IV endometriosis.
We are also not privy to the experience of the surgeons involved; that is, the number of procedures performed by each surgeon in the compared groups. It is certainly well known that complications decrease with surgeon experience. In a multicenter analysis by Peter Lim et al., looking at robotic assisted hysterectomies performed by high-volume surgeons (60 or more prior procedures), the intraoperative complication rate was only 0.7% and the postoperative complication rate 6.3% (Int J Gynaecol Obstet. 2016 Jun;133[3]:359-64).
As a benign gynecologist who has been performing minimally invasive gynecologic surgery for 30 years and more recently, robotic surgery, I am shocked with the tenor of this study, as it would imply that unless someone is boarded in gynecologic oncology, he or she should not be performing robotic hysterectomies.
I would advise Dr. Wishall to reevaluate her surgeon population and look at the impact of experience as well as procedure difficultly. I am absolutely sure that she will find that many of the surgeons with excellent outcomes will be generalists, who are well experienced in robotic hysterectomy.
Dr. Charles E. Miller is a clinical associate professor at the University of Illinois at Chicago, and a reproductive endocrinologist and minimally invasive gynecologic surgeon in private practice in Naperville and Schaumburg, Ill. He reported having no financial disclosures relevant to this article.
I read this article initially with amusement and then with outrage and disdain. This article summarizes the single-site, retrospective study by Dr. Kayla Wishall at the annual meeting of the Society of Gynecologic Oncology. Not only is this nonscience, but nonsensical science. As a single center retrospective study, conclusions must be suspect.

|
Dr. Charles E. Miller |
The comparison numbers of the two groups are small. While confounders would appear to be greater in the oncology group, we know nothing about the difficulty of the surgeries themselves – size of uterus, adnexal disease, endometriosis, pelvic adhesions, etc. Oftentimes, gynecologic oncologists dealing with endometrial carcinoma are going to face a less difficult challenge than a generalist dealing with an 18-weeks–size uterus in a woman who has undergone three prior C-sections, an open myomectomy, or stage IV endometriosis.
We are also not privy to the experience of the surgeons involved; that is, the number of procedures performed by each surgeon in the compared groups. It is certainly well known that complications decrease with surgeon experience. In a multicenter analysis by Peter Lim et al., looking at robotic assisted hysterectomies performed by high-volume surgeons (60 or more prior procedures), the intraoperative complication rate was only 0.7% and the postoperative complication rate 6.3% (Int J Gynaecol Obstet. 2016 Jun;133[3]:359-64).
As a benign gynecologist who has been performing minimally invasive gynecologic surgery for 30 years and more recently, robotic surgery, I am shocked with the tenor of this study, as it would imply that unless someone is boarded in gynecologic oncology, he or she should not be performing robotic hysterectomies.
I would advise Dr. Wishall to reevaluate her surgeon population and look at the impact of experience as well as procedure difficultly. I am absolutely sure that she will find that many of the surgeons with excellent outcomes will be generalists, who are well experienced in robotic hysterectomy.
Dr. Charles E. Miller is a clinical associate professor at the University of Illinois at Chicago, and a reproductive endocrinologist and minimally invasive gynecologic surgeon in private practice in Naperville and Schaumburg, Ill. He reported having no financial disclosures relevant to this article.
I read this article initially with amusement and then with outrage and disdain. This article summarizes the single-site, retrospective study by Dr. Kayla Wishall at the annual meeting of the Society of Gynecologic Oncology. Not only is this nonscience, but nonsensical science. As a single center retrospective study, conclusions must be suspect.

|
Dr. Charles E. Miller |
The comparison numbers of the two groups are small. While confounders would appear to be greater in the oncology group, we know nothing about the difficulty of the surgeries themselves – size of uterus, adnexal disease, endometriosis, pelvic adhesions, etc. Oftentimes, gynecologic oncologists dealing with endometrial carcinoma are going to face a less difficult challenge than a generalist dealing with an 18-weeks–size uterus in a woman who has undergone three prior C-sections, an open myomectomy, or stage IV endometriosis.
We are also not privy to the experience of the surgeons involved; that is, the number of procedures performed by each surgeon in the compared groups. It is certainly well known that complications decrease with surgeon experience. In a multicenter analysis by Peter Lim et al., looking at robotic assisted hysterectomies performed by high-volume surgeons (60 or more prior procedures), the intraoperative complication rate was only 0.7% and the postoperative complication rate 6.3% (Int J Gynaecol Obstet. 2016 Jun;133[3]:359-64).
As a benign gynecologist who has been performing minimally invasive gynecologic surgery for 30 years and more recently, robotic surgery, I am shocked with the tenor of this study, as it would imply that unless someone is boarded in gynecologic oncology, he or she should not be performing robotic hysterectomies.
I would advise Dr. Wishall to reevaluate her surgeon population and look at the impact of experience as well as procedure difficultly. I am absolutely sure that she will find that many of the surgeons with excellent outcomes will be generalists, who are well experienced in robotic hysterectomy.
Dr. Charles E. Miller is a clinical associate professor at the University of Illinois at Chicago, and a reproductive endocrinologist and minimally invasive gynecologic surgeon in private practice in Naperville and Schaumburg, Ill. He reported having no financial disclosures relevant to this article.
SAN DIEGO – The demand for gynecologic oncologists to perform robotic hysterectomies – even for benign indications – has increased to the point that additional fellowship training spots will be necessary to meet the need, Dr. Kayla M. Wishall said at the annual meeting of the Society of Gynecologic Oncology.
More and more patients want their hysterectomies performed robotically. They find the high-quality optics and minimally invasive nature of the robotic procedure appealing – smaller incisions, less blood loss, shorter hospital stay, and faster recovery. And gynecologic oncologists are getting an increasing number of referrals because of their special expertise in robotic surgery and extensive experience with higher-risk patients, explained Dr. Wishall, a gynecologic oncologist at Hahnemann University Hospital/Drexel University in Philadelphia.
“This trend will likely tax the limited resources of gynecologic oncologists,” she added.
Another possible reason for the growing demand for gynecologic oncologist–performed robotic hysterectomies is that these subspecialists achieve better outcomes than gynecologists who do robotic hysterectomies, at least according to the findings of a retrospective study performed by Dr. Wishall, which included all of the 468 robotic hysterectomies performed at a large academic medical center in a recent 5-year period.
Gynecologic oncologists performed 64 (16.5%) of the 387 robotic hysterectomies done for benign indications. All told, gynecologists did 254 of the robotic hysterectomies; gynecologic oncologists performed 214.
Even though patients referred to gynecologic oncologists for these procedures were older, heavier, more likely to have had previous abdominal surgery, more often members of racial minorities, and had a higher prevalence of cardiac comorbidities, they experienced significantly fewer intra- and postoperative complications than patients whose robotic hysterectomies were performed by gynecologists, Dr. Wishall reported.
The combined intraoperative and postoperative complication rate for robotic hysterectomies performed by gynecologic oncologists was 5.2%, compared with 16% for gynecologists. But the rate of cardiac comorbidities, for instance, was 36.4% among patients seeing gynecologic oncologists, compared with 23.6% among those seeing gynecologists.
Moreover, gynecologists were about 10-fold more likely than gynecologic oncologists to call for an intraoperative consultation and sixfold more likely to convert their robotic hysterectomy to an open procedure. Their average operating room time was about 40% longer (244 minutes versus 171 minutes), too, in this single-center experience.
Dr. Wishall reported having no financial conflicts related to her study, which was conducted free of commercial support.
SAN DIEGO – The demand for gynecologic oncologists to perform robotic hysterectomies – even for benign indications – has increased to the point that additional fellowship training spots will be necessary to meet the need, Dr. Kayla M. Wishall said at the annual meeting of the Society of Gynecologic Oncology.
More and more patients want their hysterectomies performed robotically. They find the high-quality optics and minimally invasive nature of the robotic procedure appealing – smaller incisions, less blood loss, shorter hospital stay, and faster recovery. And gynecologic oncologists are getting an increasing number of referrals because of their special expertise in robotic surgery and extensive experience with higher-risk patients, explained Dr. Wishall, a gynecologic oncologist at Hahnemann University Hospital/Drexel University in Philadelphia.
“This trend will likely tax the limited resources of gynecologic oncologists,” she added.
Another possible reason for the growing demand for gynecologic oncologist–performed robotic hysterectomies is that these subspecialists achieve better outcomes than gynecologists who do robotic hysterectomies, at least according to the findings of a retrospective study performed by Dr. Wishall, which included all of the 468 robotic hysterectomies performed at a large academic medical center in a recent 5-year period.
Gynecologic oncologists performed 64 (16.5%) of the 387 robotic hysterectomies done for benign indications. All told, gynecologists did 254 of the robotic hysterectomies; gynecologic oncologists performed 214.
Even though patients referred to gynecologic oncologists for these procedures were older, heavier, more likely to have had previous abdominal surgery, more often members of racial minorities, and had a higher prevalence of cardiac comorbidities, they experienced significantly fewer intra- and postoperative complications than patients whose robotic hysterectomies were performed by gynecologists, Dr. Wishall reported.
The combined intraoperative and postoperative complication rate for robotic hysterectomies performed by gynecologic oncologists was 5.2%, compared with 16% for gynecologists. But the rate of cardiac comorbidities, for instance, was 36.4% among patients seeing gynecologic oncologists, compared with 23.6% among those seeing gynecologists.
Moreover, gynecologists were about 10-fold more likely than gynecologic oncologists to call for an intraoperative consultation and sixfold more likely to convert their robotic hysterectomy to an open procedure. Their average operating room time was about 40% longer (244 minutes versus 171 minutes), too, in this single-center experience.
Dr. Wishall reported having no financial conflicts related to her study, which was conducted free of commercial support.
AT THE ANNUAL MEETING ON WOMEN’S CANCER
Key clinical point: Gynecologic oncologists achieved better robotic hysterectomy outcomes than gynecologists despite challenging referrals.
Major finding: The combined intraoperative and postoperative complication rate for robotic hysterectomies performed by gynecologic oncologists was 5.2%, compared with 16% for gynecologists.
Data source: A retrospective observational study conducted at a single center included 254 women whose robotic hysterectomies were performed by gynecologists and 214 done by gynecologic oncologists.
Disclosures: Dr. Wishall reported having no financial conflicts related to the study, which was conducted free of commercial support.
Optical coherence tomography for PCI gets boost in OPINION trial
PARIS – The first-ever head-to-head randomized trial comparing clinical outcomes of optical coherence tomography and intravascular ultrasound (IVUS) for guidance of percutaneous coronary intervention with a second-generation drug-eluting stent has ended in a draw.
“The clinical outcomes in both OCT-guided PCI and IVUS-guided PCI were excellent in the OPINION study,” Dr. Takashi Kubo reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
The form of OCT used in this randomized trial is called optimal frequency domain imaging (OFDI). On the strength of the OPINION results, OFDI deserves to get an upgrade in the PCI treatment guidelines, said Dr. Kubo of Wakayama (Japan) University.
He noted that the 2014 European Society of Cardiology guidelines give IVUS a Class IIa recommendation in selected patients to optimize stent implantation, with a Level of Evidence of B (Eur Heart J. 2014 Oct 1;35:2541-619). The guidelines give OCT (optimal coherence tomography), the more recent and less-studied technology, a Class IIb, Level of Evidence C.
“Our results might influence the next ESC guidelines,” according to Dr. Kubo. “OCT use during PCI should have a Class IIa recommendation.”
The OPINION trial was a prospective, 42-site Japanese study in which 800 patients scheduled for PCI with the Terumo Nobori biolimus-eluting resorbable polymer stent were randomized to an OFDI- or IVUS-guided procedure. All participants underwent follow-up coronary angiography at 8 months and clinical assessment at 12 months.
The primary study endpoint was target vessel failure at 12 months post-PCI, a composite comprising cardiac death, target vessel–related MI, or clinically driven target vessel revascularization. The rate was 5.2% in the OFDI group and statistically similar at 4.9% in the IVUS arm. No cases of contrast-induced nephropathy occurred in either study arm, and stroke rates in both groups were similarly low.
Also noteworthy was the finding that the two intracoronary imaging technologies resulted in similar rates of procedural change: 38% of patients in the OFDI group had a procedural change as result of the imaging findings, as did 36% of the IVUS group. Examples of these procedural changes included upsizing the pre- or postdilatation balloon size or pressure, addition of an another stent, or the use of a distal protection device.
In Japan, where both OCT and IVUS during PCI are routinely reimbursed, roughly 80% of PCI patients undergo one of the two intracoronary imaging procedures. In the United States and Europe, the situation is reversed, Dr. Kubo observed.
Discussant Dr. Ron Waksman agreed with Dr. Kubo that the OPINION results warrant reconsideration of OCT’s Class IIb recommendation in the ESC PCI guidelines. But he thinks the study has a major limitation.
“In my view, this was a missed opportunity to include an angiographically guided PCI arm to establish the superiority of invasive imaging over angiographically guided PCI,” said Dr. Waksman of the MedStar Heart Institute in Washington. While he noted that a recent meta-analysis of 20 studies in more than 29,000 patients concluded that IVUS-guided implantation of drug-eluting stents was associated with a 38% reduction in the risk of mortality, a 23% decrease in major adverse cardiovascular events, and a 41% reduction in stent thrombosis, compared with angiographically guided PCI (BMC Cardiovasc Disord. 2015 Nov 17;15:153), given the inherent limitations of meta-analyses he’s not convinced that cardiologists really need imaging guidance.
“ILUMIEN III, to my view, is the right study design because it randomizes patients to OCT guidance, IVUS guidance, or angiographic guidance to see if there are important differences. We will have to wait for the ILUMIEN III study results to prove the superiority of invasive imaging over angiographically guided PCI,” according to Dr. Waksman.
It’s anticipated that the ILUMIEN III trial will be ready for presentation at EuroPCR 2017.
The OPINION trial was sponsored by Terumo. Dr. Kubo is a consultant to and recipient of an institutional research grant from the company.
PARIS – The first-ever head-to-head randomized trial comparing clinical outcomes of optical coherence tomography and intravascular ultrasound (IVUS) for guidance of percutaneous coronary intervention with a second-generation drug-eluting stent has ended in a draw.
“The clinical outcomes in both OCT-guided PCI and IVUS-guided PCI were excellent in the OPINION study,” Dr. Takashi Kubo reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
The form of OCT used in this randomized trial is called optimal frequency domain imaging (OFDI). On the strength of the OPINION results, OFDI deserves to get an upgrade in the PCI treatment guidelines, said Dr. Kubo of Wakayama (Japan) University.
He noted that the 2014 European Society of Cardiology guidelines give IVUS a Class IIa recommendation in selected patients to optimize stent implantation, with a Level of Evidence of B (Eur Heart J. 2014 Oct 1;35:2541-619). The guidelines give OCT (optimal coherence tomography), the more recent and less-studied technology, a Class IIb, Level of Evidence C.
“Our results might influence the next ESC guidelines,” according to Dr. Kubo. “OCT use during PCI should have a Class IIa recommendation.”
The OPINION trial was a prospective, 42-site Japanese study in which 800 patients scheduled for PCI with the Terumo Nobori biolimus-eluting resorbable polymer stent were randomized to an OFDI- or IVUS-guided procedure. All participants underwent follow-up coronary angiography at 8 months and clinical assessment at 12 months.
The primary study endpoint was target vessel failure at 12 months post-PCI, a composite comprising cardiac death, target vessel–related MI, or clinically driven target vessel revascularization. The rate was 5.2% in the OFDI group and statistically similar at 4.9% in the IVUS arm. No cases of contrast-induced nephropathy occurred in either study arm, and stroke rates in both groups were similarly low.
Also noteworthy was the finding that the two intracoronary imaging technologies resulted in similar rates of procedural change: 38% of patients in the OFDI group had a procedural change as result of the imaging findings, as did 36% of the IVUS group. Examples of these procedural changes included upsizing the pre- or postdilatation balloon size or pressure, addition of an another stent, or the use of a distal protection device.
In Japan, where both OCT and IVUS during PCI are routinely reimbursed, roughly 80% of PCI patients undergo one of the two intracoronary imaging procedures. In the United States and Europe, the situation is reversed, Dr. Kubo observed.
Discussant Dr. Ron Waksman agreed with Dr. Kubo that the OPINION results warrant reconsideration of OCT’s Class IIb recommendation in the ESC PCI guidelines. But he thinks the study has a major limitation.
“In my view, this was a missed opportunity to include an angiographically guided PCI arm to establish the superiority of invasive imaging over angiographically guided PCI,” said Dr. Waksman of the MedStar Heart Institute in Washington. While he noted that a recent meta-analysis of 20 studies in more than 29,000 patients concluded that IVUS-guided implantation of drug-eluting stents was associated with a 38% reduction in the risk of mortality, a 23% decrease in major adverse cardiovascular events, and a 41% reduction in stent thrombosis, compared with angiographically guided PCI (BMC Cardiovasc Disord. 2015 Nov 17;15:153), given the inherent limitations of meta-analyses he’s not convinced that cardiologists really need imaging guidance.
“ILUMIEN III, to my view, is the right study design because it randomizes patients to OCT guidance, IVUS guidance, or angiographic guidance to see if there are important differences. We will have to wait for the ILUMIEN III study results to prove the superiority of invasive imaging over angiographically guided PCI,” according to Dr. Waksman.
It’s anticipated that the ILUMIEN III trial will be ready for presentation at EuroPCR 2017.
The OPINION trial was sponsored by Terumo. Dr. Kubo is a consultant to and recipient of an institutional research grant from the company.
PARIS – The first-ever head-to-head randomized trial comparing clinical outcomes of optical coherence tomography and intravascular ultrasound (IVUS) for guidance of percutaneous coronary intervention with a second-generation drug-eluting stent has ended in a draw.
“The clinical outcomes in both OCT-guided PCI and IVUS-guided PCI were excellent in the OPINION study,” Dr. Takashi Kubo reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
The form of OCT used in this randomized trial is called optimal frequency domain imaging (OFDI). On the strength of the OPINION results, OFDI deserves to get an upgrade in the PCI treatment guidelines, said Dr. Kubo of Wakayama (Japan) University.
He noted that the 2014 European Society of Cardiology guidelines give IVUS a Class IIa recommendation in selected patients to optimize stent implantation, with a Level of Evidence of B (Eur Heart J. 2014 Oct 1;35:2541-619). The guidelines give OCT (optimal coherence tomography), the more recent and less-studied technology, a Class IIb, Level of Evidence C.
“Our results might influence the next ESC guidelines,” according to Dr. Kubo. “OCT use during PCI should have a Class IIa recommendation.”
The OPINION trial was a prospective, 42-site Japanese study in which 800 patients scheduled for PCI with the Terumo Nobori biolimus-eluting resorbable polymer stent were randomized to an OFDI- or IVUS-guided procedure. All participants underwent follow-up coronary angiography at 8 months and clinical assessment at 12 months.
The primary study endpoint was target vessel failure at 12 months post-PCI, a composite comprising cardiac death, target vessel–related MI, or clinically driven target vessel revascularization. The rate was 5.2% in the OFDI group and statistically similar at 4.9% in the IVUS arm. No cases of contrast-induced nephropathy occurred in either study arm, and stroke rates in both groups were similarly low.
Also noteworthy was the finding that the two intracoronary imaging technologies resulted in similar rates of procedural change: 38% of patients in the OFDI group had a procedural change as result of the imaging findings, as did 36% of the IVUS group. Examples of these procedural changes included upsizing the pre- or postdilatation balloon size or pressure, addition of an another stent, or the use of a distal protection device.
In Japan, where both OCT and IVUS during PCI are routinely reimbursed, roughly 80% of PCI patients undergo one of the two intracoronary imaging procedures. In the United States and Europe, the situation is reversed, Dr. Kubo observed.
Discussant Dr. Ron Waksman agreed with Dr. Kubo that the OPINION results warrant reconsideration of OCT’s Class IIb recommendation in the ESC PCI guidelines. But he thinks the study has a major limitation.
“In my view, this was a missed opportunity to include an angiographically guided PCI arm to establish the superiority of invasive imaging over angiographically guided PCI,” said Dr. Waksman of the MedStar Heart Institute in Washington. While he noted that a recent meta-analysis of 20 studies in more than 29,000 patients concluded that IVUS-guided implantation of drug-eluting stents was associated with a 38% reduction in the risk of mortality, a 23% decrease in major adverse cardiovascular events, and a 41% reduction in stent thrombosis, compared with angiographically guided PCI (BMC Cardiovasc Disord. 2015 Nov 17;15:153), given the inherent limitations of meta-analyses he’s not convinced that cardiologists really need imaging guidance.
“ILUMIEN III, to my view, is the right study design because it randomizes patients to OCT guidance, IVUS guidance, or angiographic guidance to see if there are important differences. We will have to wait for the ILUMIEN III study results to prove the superiority of invasive imaging over angiographically guided PCI,” according to Dr. Waksman.
It’s anticipated that the ILUMIEN III trial will be ready for presentation at EuroPCR 2017.
The OPINION trial was sponsored by Terumo. Dr. Kubo is a consultant to and recipient of an institutional research grant from the company.
AT EUROPCR 2016
Key clinical point: A large, randomized trial shows PCI clinical outcomes are equivalent with optical coherence tomography and intravascular ultrasound guidance.
Major finding: The composite rate of cardiac death, target vessel–related MI, or clinically driven target vessel revascularization within 12 months of PCI was 5.2% in the group whose procedure was guided by optical coherence tomography and statistically similar at 4.9% in patients whose PCI was guided by intravascular ultrasound.
Data source: This was a randomized, prospective, multicenter, 12-month follow-up trial of 800 Japanese patients scheduled for PCI under intracoronary imaging guidance provided by either IVUS or OCT.
Disclosures: The OPINION trial was sponsored by Terumo. The study presenter is a consultant to and recipient of an institutional research grant from the company.
TAVR cerebral protection device appears safe, effective
PARIS – The TriGuard neuroprotection device for use during transcatheter aortic valve replacement effectively prevented strokes while raising no safety concerns in a pooled analysis of three controlled trials, according to Dr. Alexandra J. Lansky.
The TriGuard, which is investigational in the United States but approved in Europe, also significantly reduced the risk of central nervous system infarction, as assessed by diffusion-weighted MRI. Moreover, when imaging did show CNS infarcts in patients with the TriGuard in place during their TAVR (transcatheter aortic valve replacement), the total brain lesion volume was about 40% less than in controls without the neuroprotection device, according to Dr. Lansky, professor of medicine and director of the cardiovascular clinical research program at Yale University in New Haven, Conn.
“Essentially what’s happening is that we’re reducing with this device the frequency of CNS infarctions, and also reducing the size of the lesions when they are present,” she said at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
The TriGuard is designed to fill an unmet need for stroke protection in TAVR patients. The incidence of clinical stroke within 30 days after TAVR in recent randomized controlled trials is 1.5%-6%. But there is clear evidence of underreporting of stroke in these trials. When neurologists examine TAVR patients or the patients are evaluated by serial testing using the NIH Stroke Scale plus brain imaging, the 30-day stroke rates are 15%-28%, according to the cardiologist.
“We know that about 50% of these strokes happen in the periprocedural period, and stroke is one of the strongest predictors of mortality, conferring a three- to ninefold increased risk,” Dr. Lansky emphasized.
She presented a pooled analysis including 59 TriGuard recipients and 83 controls who underwent TAVR in the DEFLECT I and III trials and the NeuroTAVR registry. They were evaluated using the NIH Stroke Scale before TAVR and again at 4 and 30 days post procedure. In addition, they underwent brain imaging via diffusion-weighted MRI 4 days post TAVR.
Stroke as defined by the Valve Academic Research Consortium–2 (VARC2) criteria occurred in none of the TriGuard group but in 6% of controls. And stroke as defined by the American Stroke Association, which requires a worsening score on the serial NIH Stroke Scale measurements plus imaging evidence of CNS infarction, occurred in 0 TriGuard-protected patients and in 19% of controls.
The incidence of CNS infarction on MRI was 92% in controls and 72% in the TriGuard group. Thus, 28% of patients with the TriGuard in place developed no brain infarct lesions at all; that’s a first for any TAVR neuroprotection device, according to Dr. Lansky.
In patients with CNS lesions, the total lesion volume was 101 mm3 in the TriGuard group, compared with 174 mm3 in the controls. The average lesion volume was 25 mm3 in the TriGuard group versus 43 mm3 in the controls.
TriGuard is a relatively simple device consisting of a single-wire nitinol frame and mesh filter with a pore size of 130 mcm. It’s designed to deflect emboli during TAVR while allowing maximal cerebral blood flow. After being delivered by a 9 French sheath from the contralateral femoral artery, the device sits at the roof of the aortic arch. Importantly, it covers all three cerebral arteries, Dr. Lansky said. The device is held in position by a stabilizer in the innominate artery.
Although introducing an additional element into TAVR raises the theoretic possibility of safety concerns, no safety signal was seen in the pooled analysis. In-hospital major adverse event rates were similar in the two groups.
Asked why 72% of patients with the TriGuard in place nonetheless developed CNS infarcts, Dr. Lansky said she believes the device has gaps on the sides that allow smaller emboli to pass through. Future iterations of the TriGuard will address this.
The clinical significance of the CNS infarcts seen on MRI in TAVR patients is a controversial issue among interventional cardiologists. Some cardiologists consider these to be silent lesions of dubious clinical relevance. That’s not Dr. Lansky’s view.
“When you track these MRI lesions out to 30 days, many times they disappear. They don’t disappear because there’s no damage; they disappear because the cells die. When you talk to neurologists about the MRI lesions, they will tell you that they actually represent cell death and correlate with brain infarction,” she said.
Dr. Nicolo Piazza commented that he considered the pooled analysis findings hypothesis generating but not definitive because of baseline imbalances between the two study arms. The control group had numerically higher – albeit not statistically significantly so – rates of atrial fibrillation at hospital admission as well as higher Society of Thoracic Surgeons risk scores, both of which increase stroke risk, noted Dr. Piazza of McGill University in Montreal.
Dr. Lansky replied that the much larger ongoing pivotal randomized, phase III REFLECT trial should provide definitive answers.
She reported receiving institutional research grant support from Keystone Heart, which produces the TriGuard device.
PARIS – The TriGuard neuroprotection device for use during transcatheter aortic valve replacement effectively prevented strokes while raising no safety concerns in a pooled analysis of three controlled trials, according to Dr. Alexandra J. Lansky.
The TriGuard, which is investigational in the United States but approved in Europe, also significantly reduced the risk of central nervous system infarction, as assessed by diffusion-weighted MRI. Moreover, when imaging did show CNS infarcts in patients with the TriGuard in place during their TAVR (transcatheter aortic valve replacement), the total brain lesion volume was about 40% less than in controls without the neuroprotection device, according to Dr. Lansky, professor of medicine and director of the cardiovascular clinical research program at Yale University in New Haven, Conn.
“Essentially what’s happening is that we’re reducing with this device the frequency of CNS infarctions, and also reducing the size of the lesions when they are present,” she said at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
The TriGuard is designed to fill an unmet need for stroke protection in TAVR patients. The incidence of clinical stroke within 30 days after TAVR in recent randomized controlled trials is 1.5%-6%. But there is clear evidence of underreporting of stroke in these trials. When neurologists examine TAVR patients or the patients are evaluated by serial testing using the NIH Stroke Scale plus brain imaging, the 30-day stroke rates are 15%-28%, according to the cardiologist.
“We know that about 50% of these strokes happen in the periprocedural period, and stroke is one of the strongest predictors of mortality, conferring a three- to ninefold increased risk,” Dr. Lansky emphasized.
She presented a pooled analysis including 59 TriGuard recipients and 83 controls who underwent TAVR in the DEFLECT I and III trials and the NeuroTAVR registry. They were evaluated using the NIH Stroke Scale before TAVR and again at 4 and 30 days post procedure. In addition, they underwent brain imaging via diffusion-weighted MRI 4 days post TAVR.
Stroke as defined by the Valve Academic Research Consortium–2 (VARC2) criteria occurred in none of the TriGuard group but in 6% of controls. And stroke as defined by the American Stroke Association, which requires a worsening score on the serial NIH Stroke Scale measurements plus imaging evidence of CNS infarction, occurred in 0 TriGuard-protected patients and in 19% of controls.
The incidence of CNS infarction on MRI was 92% in controls and 72% in the TriGuard group. Thus, 28% of patients with the TriGuard in place developed no brain infarct lesions at all; that’s a first for any TAVR neuroprotection device, according to Dr. Lansky.
In patients with CNS lesions, the total lesion volume was 101 mm3 in the TriGuard group, compared with 174 mm3 in the controls. The average lesion volume was 25 mm3 in the TriGuard group versus 43 mm3 in the controls.
TriGuard is a relatively simple device consisting of a single-wire nitinol frame and mesh filter with a pore size of 130 mcm. It’s designed to deflect emboli during TAVR while allowing maximal cerebral blood flow. After being delivered by a 9 French sheath from the contralateral femoral artery, the device sits at the roof of the aortic arch. Importantly, it covers all three cerebral arteries, Dr. Lansky said. The device is held in position by a stabilizer in the innominate artery.
Although introducing an additional element into TAVR raises the theoretic possibility of safety concerns, no safety signal was seen in the pooled analysis. In-hospital major adverse event rates were similar in the two groups.
Asked why 72% of patients with the TriGuard in place nonetheless developed CNS infarcts, Dr. Lansky said she believes the device has gaps on the sides that allow smaller emboli to pass through. Future iterations of the TriGuard will address this.
The clinical significance of the CNS infarcts seen on MRI in TAVR patients is a controversial issue among interventional cardiologists. Some cardiologists consider these to be silent lesions of dubious clinical relevance. That’s not Dr. Lansky’s view.
“When you track these MRI lesions out to 30 days, many times they disappear. They don’t disappear because there’s no damage; they disappear because the cells die. When you talk to neurologists about the MRI lesions, they will tell you that they actually represent cell death and correlate with brain infarction,” she said.
Dr. Nicolo Piazza commented that he considered the pooled analysis findings hypothesis generating but not definitive because of baseline imbalances between the two study arms. The control group had numerically higher – albeit not statistically significantly so – rates of atrial fibrillation at hospital admission as well as higher Society of Thoracic Surgeons risk scores, both of which increase stroke risk, noted Dr. Piazza of McGill University in Montreal.
Dr. Lansky replied that the much larger ongoing pivotal randomized, phase III REFLECT trial should provide definitive answers.
She reported receiving institutional research grant support from Keystone Heart, which produces the TriGuard device.
PARIS – The TriGuard neuroprotection device for use during transcatheter aortic valve replacement effectively prevented strokes while raising no safety concerns in a pooled analysis of three controlled trials, according to Dr. Alexandra J. Lansky.
The TriGuard, which is investigational in the United States but approved in Europe, also significantly reduced the risk of central nervous system infarction, as assessed by diffusion-weighted MRI. Moreover, when imaging did show CNS infarcts in patients with the TriGuard in place during their TAVR (transcatheter aortic valve replacement), the total brain lesion volume was about 40% less than in controls without the neuroprotection device, according to Dr. Lansky, professor of medicine and director of the cardiovascular clinical research program at Yale University in New Haven, Conn.
“Essentially what’s happening is that we’re reducing with this device the frequency of CNS infarctions, and also reducing the size of the lesions when they are present,” she said at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
The TriGuard is designed to fill an unmet need for stroke protection in TAVR patients. The incidence of clinical stroke within 30 days after TAVR in recent randomized controlled trials is 1.5%-6%. But there is clear evidence of underreporting of stroke in these trials. When neurologists examine TAVR patients or the patients are evaluated by serial testing using the NIH Stroke Scale plus brain imaging, the 30-day stroke rates are 15%-28%, according to the cardiologist.
“We know that about 50% of these strokes happen in the periprocedural period, and stroke is one of the strongest predictors of mortality, conferring a three- to ninefold increased risk,” Dr. Lansky emphasized.
She presented a pooled analysis including 59 TriGuard recipients and 83 controls who underwent TAVR in the DEFLECT I and III trials and the NeuroTAVR registry. They were evaluated using the NIH Stroke Scale before TAVR and again at 4 and 30 days post procedure. In addition, they underwent brain imaging via diffusion-weighted MRI 4 days post TAVR.
Stroke as defined by the Valve Academic Research Consortium–2 (VARC2) criteria occurred in none of the TriGuard group but in 6% of controls. And stroke as defined by the American Stroke Association, which requires a worsening score on the serial NIH Stroke Scale measurements plus imaging evidence of CNS infarction, occurred in 0 TriGuard-protected patients and in 19% of controls.
The incidence of CNS infarction on MRI was 92% in controls and 72% in the TriGuard group. Thus, 28% of patients with the TriGuard in place developed no brain infarct lesions at all; that’s a first for any TAVR neuroprotection device, according to Dr. Lansky.
In patients with CNS lesions, the total lesion volume was 101 mm3 in the TriGuard group, compared with 174 mm3 in the controls. The average lesion volume was 25 mm3 in the TriGuard group versus 43 mm3 in the controls.
TriGuard is a relatively simple device consisting of a single-wire nitinol frame and mesh filter with a pore size of 130 mcm. It’s designed to deflect emboli during TAVR while allowing maximal cerebral blood flow. After being delivered by a 9 French sheath from the contralateral femoral artery, the device sits at the roof of the aortic arch. Importantly, it covers all three cerebral arteries, Dr. Lansky said. The device is held in position by a stabilizer in the innominate artery.
Although introducing an additional element into TAVR raises the theoretic possibility of safety concerns, no safety signal was seen in the pooled analysis. In-hospital major adverse event rates were similar in the two groups.
Asked why 72% of patients with the TriGuard in place nonetheless developed CNS infarcts, Dr. Lansky said she believes the device has gaps on the sides that allow smaller emboli to pass through. Future iterations of the TriGuard will address this.
The clinical significance of the CNS infarcts seen on MRI in TAVR patients is a controversial issue among interventional cardiologists. Some cardiologists consider these to be silent lesions of dubious clinical relevance. That’s not Dr. Lansky’s view.
“When you track these MRI lesions out to 30 days, many times they disappear. They don’t disappear because there’s no damage; they disappear because the cells die. When you talk to neurologists about the MRI lesions, they will tell you that they actually represent cell death and correlate with brain infarction,” she said.
Dr. Nicolo Piazza commented that he considered the pooled analysis findings hypothesis generating but not definitive because of baseline imbalances between the two study arms. The control group had numerically higher – albeit not statistically significantly so – rates of atrial fibrillation at hospital admission as well as higher Society of Thoracic Surgeons risk scores, both of which increase stroke risk, noted Dr. Piazza of McGill University in Montreal.
Dr. Lansky replied that the much larger ongoing pivotal randomized, phase III REFLECT trial should provide definitive answers.
She reported receiving institutional research grant support from Keystone Heart, which produces the TriGuard device.
AT EUROPCR 2016
Key clinical point: The TriGuard neuroprotection device for use in TAVR effectively prevented strokes.
Major finding: The 30-day incidence of stroke in TAVR patients with the TriGard embolic protection device in place was 0, compared with 6% or 19% in controls, depending upon the stroke definition used.
Data source: A post hoc analysis of pooled data on 59 TriGuard recipients and 83 controls in three trials.
Disclosures: The presenter reported receiving institutional research grant support from Keystone Heart, which produces the TriGuard device.
Low hematocrit in elderly portends increased bleeding post PCI
PARIS – A low hematocrit in an elderly patient who’s going to undergo percutaneous coronary intervention signals a markedly increased risk of major bleeding within 30 days of the procedure, according to Dr. David Marti.
“Analysis of hematocrit in elderly patients can guide important procedural characteristics, such as access site and antithrombotic regimen,” he said at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
For example, studies have established that transradial artery access percutaneous coronary intervention (PCI) results in significantly less bleeding than the transfemoral route, said Dr. Marti, an interventional cardiologist at the University of Alcalá in Madrid.
He presented a prospective study of 212 consecutive patients aged 75 or older who underwent PCI at a single university hospital. Their mean age was 81.4 years, and slightly over half of them presented with an acute coronary syndrome.
All patients received dual-antiplatelet therapy in accord with current guidelines. Stent type and procedural anticoagulant regimen were left to the discretion of the cardiologist; 80% of the subjects received bivalirudin-based anticoagulation.
The primary study outcome was the 30-day incidence of major bleeding, as defined by a Bleeding Academic Research Consortium (BARC) type 3-5 event. The overall rate in this elderly PCI population was 5.5%. However, the rate varied markedly by baseline hematocrit tertile, in accord with the investigators’ study hypothesis.
Major bleeding occurred in 2.9% of patients with an Hct greater than 42% and 3.1% in those with an Hct of 38%-52%, and jumped to 10.6% in the one-third of subjects whose baseline Hct was below 38%, Dr. Marti reported.
Thus, a preprocedural Hct below 38% was associated with a 4.1-fold increased risk of major bleeding within 30 days following PCI. An Hct in this range was a stronger predictor of BARC type 3-5 bleeding risk than were other factors better known as being important, including advanced age, greater body weight, female sex, or an elevated serum creatinine indicative of chronic kidney disease. Indeed, an Hct below 38% was the only statistically significant predictor of major bleeding in this elderly population.
The likely explanation for the observed results is that a low Hct level in elderly patients usually reflects subclinical blood loss that can be worsened by antithrombotic therapies, the cardiologist explained.
The presenter reported having no financial conflicts regarding this study, conducted without commercial support.
PARIS – A low hematocrit in an elderly patient who’s going to undergo percutaneous coronary intervention signals a markedly increased risk of major bleeding within 30 days of the procedure, according to Dr. David Marti.
“Analysis of hematocrit in elderly patients can guide important procedural characteristics, such as access site and antithrombotic regimen,” he said at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
For example, studies have established that transradial artery access percutaneous coronary intervention (PCI) results in significantly less bleeding than the transfemoral route, said Dr. Marti, an interventional cardiologist at the University of Alcalá in Madrid.
He presented a prospective study of 212 consecutive patients aged 75 or older who underwent PCI at a single university hospital. Their mean age was 81.4 years, and slightly over half of them presented with an acute coronary syndrome.
All patients received dual-antiplatelet therapy in accord with current guidelines. Stent type and procedural anticoagulant regimen were left to the discretion of the cardiologist; 80% of the subjects received bivalirudin-based anticoagulation.
The primary study outcome was the 30-day incidence of major bleeding, as defined by a Bleeding Academic Research Consortium (BARC) type 3-5 event. The overall rate in this elderly PCI population was 5.5%. However, the rate varied markedly by baseline hematocrit tertile, in accord with the investigators’ study hypothesis.
Major bleeding occurred in 2.9% of patients with an Hct greater than 42% and 3.1% in those with an Hct of 38%-52%, and jumped to 10.6% in the one-third of subjects whose baseline Hct was below 38%, Dr. Marti reported.
Thus, a preprocedural Hct below 38% was associated with a 4.1-fold increased risk of major bleeding within 30 days following PCI. An Hct in this range was a stronger predictor of BARC type 3-5 bleeding risk than were other factors better known as being important, including advanced age, greater body weight, female sex, or an elevated serum creatinine indicative of chronic kidney disease. Indeed, an Hct below 38% was the only statistically significant predictor of major bleeding in this elderly population.
The likely explanation for the observed results is that a low Hct level in elderly patients usually reflects subclinical blood loss that can be worsened by antithrombotic therapies, the cardiologist explained.
The presenter reported having no financial conflicts regarding this study, conducted without commercial support.
PARIS – A low hematocrit in an elderly patient who’s going to undergo percutaneous coronary intervention signals a markedly increased risk of major bleeding within 30 days of the procedure, according to Dr. David Marti.
“Analysis of hematocrit in elderly patients can guide important procedural characteristics, such as access site and antithrombotic regimen,” he said at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
For example, studies have established that transradial artery access percutaneous coronary intervention (PCI) results in significantly less bleeding than the transfemoral route, said Dr. Marti, an interventional cardiologist at the University of Alcalá in Madrid.
He presented a prospective study of 212 consecutive patients aged 75 or older who underwent PCI at a single university hospital. Their mean age was 81.4 years, and slightly over half of them presented with an acute coronary syndrome.
All patients received dual-antiplatelet therapy in accord with current guidelines. Stent type and procedural anticoagulant regimen were left to the discretion of the cardiologist; 80% of the subjects received bivalirudin-based anticoagulation.
The primary study outcome was the 30-day incidence of major bleeding, as defined by a Bleeding Academic Research Consortium (BARC) type 3-5 event. The overall rate in this elderly PCI population was 5.5%. However, the rate varied markedly by baseline hematocrit tertile, in accord with the investigators’ study hypothesis.
Major bleeding occurred in 2.9% of patients with an Hct greater than 42% and 3.1% in those with an Hct of 38%-52%, and jumped to 10.6% in the one-third of subjects whose baseline Hct was below 38%, Dr. Marti reported.
Thus, a preprocedural Hct below 38% was associated with a 4.1-fold increased risk of major bleeding within 30 days following PCI. An Hct in this range was a stronger predictor of BARC type 3-5 bleeding risk than were other factors better known as being important, including advanced age, greater body weight, female sex, or an elevated serum creatinine indicative of chronic kidney disease. Indeed, an Hct below 38% was the only statistically significant predictor of major bleeding in this elderly population.
The likely explanation for the observed results is that a low Hct level in elderly patients usually reflects subclinical blood loss that can be worsened by antithrombotic therapies, the cardiologist explained.
The presenter reported having no financial conflicts regarding this study, conducted without commercial support.
AT EUROPCR 2016
Key clinical point: Elderly patients scheduled for PCI have a fourfold greater risk of major bleeding within 30 days if their Hct is less than 38%.
Major finding: The 30-day incidence of BARC types 3-5 major bleeding was 10.9% in elderly patients with a pre-PCI Hct below 38%, compared with 2.9% in those in the top Hct tertile.
Data source: A prospective study of 212 consecutive patients aged 75 or older who underwent PCI at a single university hospital.
Disclosures: The presenter reported having no financial conflicts regarding this study, conducted without commercial support.
Obesity paradox extends to TAVR but not SAVR
PARIS – The obesity paradox appears to apply to patients undergoing transcatheter aortic valve replacement (TAVR) but doesn’t extend to those with surgical aortic valve replacement (SAVR), Dr. Marco Spaziano reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
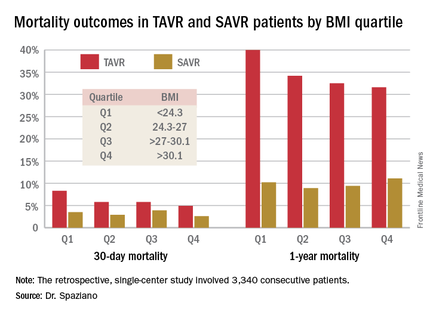
He presented a retrospective single-center study of 3,340 consecutive patients who underwent either TAVR or SAVR, with all valve replacement procedures being performed by the same surgeons. Investigators divided the patients – 1,301 with TAVR, 2,039 SAVR – into quartiles on the basis of body mass index.
Rates of 30-day and 1-year mortality, major vascular complications, and major bleeding events were consistently lowest in TAVR patients in the top body mass index (BMI) quartile, defined as greater than 30.1 kg/m2, and highest in those in the bottom quartile, reserved for patients with a BMI below 24.3 kg/m2.
It’s worth noting that with a mean BMI of 21.8 kg/m2 in the bottom BMI quartile, most patients in that group were actually normal weight, not underweight, observed Dr. Spaziano of the Paris South Cardiovascular Institute in Massy, France.
Among the TAVR group, there were no significant differences between the BMI quartiles in TAVR devices, size, or procedural approach.
The TAVR patients’ BMI quartile had no impact on other outcomes, including rates of stroke, MI, permanent pacemaker implantation, acute kidney injury, or aortic regurgitation.
While being overweight or obese was protective in TAVR patients, BMI quartiles had no relationship with outcomes in the SAVR cohort. The SAVR patients were on average about 10 years younger than the TAVR group, and their logistic EuroSCORE – a tool for estimating mortality risk after cardiac surgery – was much lower as well.
The obesity paradox remains an ongoing puzzle and source of intrigue for physicians in many different specialties. The paradox is this: Obesity is well established as a major risk factor for the development of cardiovascular diseases and diabetes, yet a higher BMI seems to be associated with lower mortality and better procedural outcomes for patients once they actually have a number of chronic diseases, including coronary artery disease, heart failure, peripheral arterial disease, hypertension, stroke, chronic obstructive pulmonary disease, renal disease, and acute venous thromboembolism.
The TAVR obesity paradox findings really got under the skin of at least one audience member.
“What’s the message?” he asked. “We’re telling people to lose weight, eat healthy, run thousands of miles and so forth, but then we’re supposed to tell patients undergoing a high-risk procedure that if you’re overweight you somehow survive better, live longer, feel better?”
Dr. Spaziano said the independent predictors of mortality in the lowest-BMI quartile of TAVR patients were a high serum creatinine, chronic obstructive pulmonary disease, and low BMI. Therein lies a likely explanation for the obesity paradox, at least in this particular study: The TAVR patients were generally sicker than the SAVR patients, which is why they weren’t undergoing surgery, and the lowest-BMI TAVR group was frailer than the others. Also, perhaps the heaviest patients benefited from having more energy reserves to draw upon.
He reported having no financial conflicts regarding this study, conducted free of commercial support.
PARIS – The obesity paradox appears to apply to patients undergoing transcatheter aortic valve replacement (TAVR) but doesn’t extend to those with surgical aortic valve replacement (SAVR), Dr. Marco Spaziano reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
He presented a retrospective single-center study of 3,340 consecutive patients who underwent either TAVR or SAVR, with all valve replacement procedures being performed by the same surgeons. Investigators divided the patients – 1,301 with TAVR, 2,039 SAVR – into quartiles on the basis of body mass index.
Rates of 30-day and 1-year mortality, major vascular complications, and major bleeding events were consistently lowest in TAVR patients in the top body mass index (BMI) quartile, defined as greater than 30.1 kg/m2, and highest in those in the bottom quartile, reserved for patients with a BMI below 24.3 kg/m2.
It’s worth noting that with a mean BMI of 21.8 kg/m2 in the bottom BMI quartile, most patients in that group were actually normal weight, not underweight, observed Dr. Spaziano of the Paris South Cardiovascular Institute in Massy, France.
Among the TAVR group, there were no significant differences between the BMI quartiles in TAVR devices, size, or procedural approach.
The TAVR patients’ BMI quartile had no impact on other outcomes, including rates of stroke, MI, permanent pacemaker implantation, acute kidney injury, or aortic regurgitation.
While being overweight or obese was protective in TAVR patients, BMI quartiles had no relationship with outcomes in the SAVR cohort. The SAVR patients were on average about 10 years younger than the TAVR group, and their logistic EuroSCORE – a tool for estimating mortality risk after cardiac surgery – was much lower as well.

The obesity paradox remains an ongoing puzzle and source of intrigue for physicians in many different specialties. The paradox is this: Obesity is well established as a major risk factor for the development of cardiovascular diseases and diabetes, yet a higher BMI seems to be associated with lower mortality and better procedural outcomes for patients once they actually have a number of chronic diseases, including coronary artery disease, heart failure, peripheral arterial disease, hypertension, stroke, chronic obstructive pulmonary disease, renal disease, and acute venous thromboembolism.
The TAVR obesity paradox findings really got under the skin of at least one audience member.
“What’s the message?” he asked. “We’re telling people to lose weight, eat healthy, run thousands of miles and so forth, but then we’re supposed to tell patients undergoing a high-risk procedure that if you’re overweight you somehow survive better, live longer, feel better?”
Dr. Spaziano said the independent predictors of mortality in the lowest-BMI quartile of TAVR patients were a high serum creatinine, chronic obstructive pulmonary disease, and low BMI. Therein lies a likely explanation for the obesity paradox, at least in this particular study: The TAVR patients were generally sicker than the SAVR patients, which is why they weren’t undergoing surgery, and the lowest-BMI TAVR group was frailer than the others. Also, perhaps the heaviest patients benefited from having more energy reserves to draw upon.
He reported having no financial conflicts regarding this study, conducted free of commercial support.
PARIS – The obesity paradox appears to apply to patients undergoing transcatheter aortic valve replacement (TAVR) but doesn’t extend to those with surgical aortic valve replacement (SAVR), Dr. Marco Spaziano reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
He presented a retrospective single-center study of 3,340 consecutive patients who underwent either TAVR or SAVR, with all valve replacement procedures being performed by the same surgeons. Investigators divided the patients – 1,301 with TAVR, 2,039 SAVR – into quartiles on the basis of body mass index.
Rates of 30-day and 1-year mortality, major vascular complications, and major bleeding events were consistently lowest in TAVR patients in the top body mass index (BMI) quartile, defined as greater than 30.1 kg/m2, and highest in those in the bottom quartile, reserved for patients with a BMI below 24.3 kg/m2.
It’s worth noting that with a mean BMI of 21.8 kg/m2 in the bottom BMI quartile, most patients in that group were actually normal weight, not underweight, observed Dr. Spaziano of the Paris South Cardiovascular Institute in Massy, France.
Among the TAVR group, there were no significant differences between the BMI quartiles in TAVR devices, size, or procedural approach.
The TAVR patients’ BMI quartile had no impact on other outcomes, including rates of stroke, MI, permanent pacemaker implantation, acute kidney injury, or aortic regurgitation.
While being overweight or obese was protective in TAVR patients, BMI quartiles had no relationship with outcomes in the SAVR cohort. The SAVR patients were on average about 10 years younger than the TAVR group, and their logistic EuroSCORE – a tool for estimating mortality risk after cardiac surgery – was much lower as well.

The obesity paradox remains an ongoing puzzle and source of intrigue for physicians in many different specialties. The paradox is this: Obesity is well established as a major risk factor for the development of cardiovascular diseases and diabetes, yet a higher BMI seems to be associated with lower mortality and better procedural outcomes for patients once they actually have a number of chronic diseases, including coronary artery disease, heart failure, peripheral arterial disease, hypertension, stroke, chronic obstructive pulmonary disease, renal disease, and acute venous thromboembolism.
The TAVR obesity paradox findings really got under the skin of at least one audience member.
“What’s the message?” he asked. “We’re telling people to lose weight, eat healthy, run thousands of miles and so forth, but then we’re supposed to tell patients undergoing a high-risk procedure that if you’re overweight you somehow survive better, live longer, feel better?”
Dr. Spaziano said the independent predictors of mortality in the lowest-BMI quartile of TAVR patients were a high serum creatinine, chronic obstructive pulmonary disease, and low BMI. Therein lies a likely explanation for the obesity paradox, at least in this particular study: The TAVR patients were generally sicker than the SAVR patients, which is why they weren’t undergoing surgery, and the lowest-BMI TAVR group was frailer than the others. Also, perhaps the heaviest patients benefited from having more energy reserves to draw upon.
He reported having no financial conflicts regarding this study, conducted free of commercial support.
AT EUROPCR 2016
Key clinical point: Being overweight or obese may have a protective effect in patients undergoing transcatheter aortic valve replacement, but not in those who have surgical replacement.
Major finding: Thirty-day mortality was 4.9% in TAVR patients in the top BMI quartile and climbed stepwise with lowering BMIs.
Data source: A single-center retrospective study of 3,340 consecutive patients who underwent TAVR or SAVR.
Disclosures: The presenter reported having no financial conflicts regarding this study, conducted free of commercial support.
TAVR degeneration estimated at 50% after 8 years
PARIS – The first-ever study of the long-term durability of transcatheter bioprosthetic aortic valves has documented a disturbing rise in the valve degeneration rate occurring 5-7 years post implant.
Prior consistently reassuring follow-up studies have been intermediate in length, with a maximum of 5 years. The PARTNER 2A trial, which generated enormous enthusiasm for moving TAVR to intermediate-risk patients on the basis of positive results presented at the 2016 meeting of the American College of Cardiology, reported 2-year results.
“We found, as have others, that there’s very little degeneration in the first 4 years: 94% freedom from degeneration. But at 6 years, it’s 82%, and we estimate that by 8 years, it’s about 50%,” Dr. Danny Dvir reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
“We need to be cautious: This is our first look at the data. But we have a signal for a problem,” said Dr. Dvir of St. Paul’s Hospital in Vancouver.
He presented a retrospective study of 378 patients who underwent TAVR 5-14 years ago at two pioneering centers for the procedure: St. Paul’s and Charles Nicolle Hospital in Rouen, France. Patients’ average age at the time of TAVR was 82.3 years, with an STS score of 8.3%. The study featured serial echocardiography conducted during house calls in this frail elderly population.
Thirty-five patients developed prosthetic valve degeneration, defined by at least moderate regurgitation and/or a mean gradient of at least 20 mm Hg in 23 cases and stenosis in 12. The risk of degeneration was unrelated to the use of warfarin, a finding that suggests the valve deterioration issue is unrelated to clotting. The strongest risk factor for transcatheter valve degeneration in this study was baseline renal failure at the time of TAVR.
Dr. Dvir’s presentation was the talk of the meeting, and it cast a pall over the proceedings. The red flag raised by the study regarding valve durability has major implications regarding the current enthusiasm among many interventional cardiologists to routinely extend TAVR to intermediate and even lower-risk patients. As one audience member later confessed, “I have felt sick since hearing that presentation.”
Discussant Dr. A. Pieter Kappetein observed that transcatheter heart valve durability was a hot topic of discussion about 4 years ago but subsequently faded below the radar as a concern – until Dr. Dvir’s study.
“This is a very important study that puts transcatheter heart valve implantation in a little bit different light,” said Dr. Kappetein, professor of cardiothoracic surgery at Erasmus University in Rotterdam, the Netherlands.
He noted that the surgical aortic valve replacement (SAVR) literature shows that the rate of structural valve deterioration is age related. It’s higher in 75-year-olds than in 85-year-olds, and higher still in 65-year-olds.
“Valve degeneration didn’t play a major role when we were doing TAVR in 80- and 85-year-olds because of their limited life expectancy, but it will play a role in younger patients. So I think we have to be careful before we move toward lower-risk patients,” the surgeon continued.
Dr. Jean-Francois Obadia, who performs both SAVR and TAVR, noted that the median duration of freedom from valve degeneration for Edwards Lifescience’s Carpentier surgical aortic valve is a hefty 17.9 years.
“This should be the gold standard,” declared Dr. Obadia, head of the department of adult cardiovascular surgery and heart transplantation at the Louis Pradel Cardiothoracic Hospital of Claude Bernard University in Lyon, France.
“Dr. Dvir’s study is one of the key messages we all should take back home: a 50% rate of valve deterioration at 8 years. Valve deterioration is the Achilles’ heel of bioprostheses. There is a lot of improvement left to do for the TAVR,” he said.
Dr. Dvir and others were quick to note that his long-term study was of necessity restricted to early-generation, balloon-expandable devices: the Cribier Edwards, Edwards Sapien, and Sapien XT valves. Contemporary valves, patient selection methods, and procedural techniques are far advanced in comparison.
“The Sapien 3 has much less paravalvular leakage than earlier-generation valves. Maybe with less paravalvular leakage and better hemodynamics there will be a decreased rate of degeneration. It could be. We need to see. We have to wait a few more years to see if later-generation transcatheter heart valves are more durable,” Dr. Dvir said.
To gain a better understanding of the full dimensions of the valve degeneration issue, he and his coinvestigators have formed the VALID (VAlve Long-term Durability International Data) registry. Operators interested in contributing patients to what is hoped will be a very large and informative data base are encouraged to contact Dr. Dvir (ddvir@providencehealth.bc.ca).
In the meantime, he has reservations about extending TAVR to intermediate-risk patients outside of the rigorous clinical trial setting. He added that he’d feel far more comfortable in performing TAVR in intermediate-risk 70- to 75-year-olds if there was a tried and true valve-in-valve replacement method, something that doesn’t yet exist. The major limitation of current attempts at valve-in-valve replacement is underexpansion because the former valve doesn’t allow sufficient room for the new one to expand fully, resulting in residual stenosis.
“If you tell me that you can implant a platform that will enable a safe valve-in-valve procedure in 5, 7, 10 years – a less invasive bailout for a failed prosthetic valve – if you can do that safely and effectively I would be more keen to do TAVR even in a young patient,” the interventional cardiologist said.
He and others are working on this unmet need. Dr. Dvir’s novel valve, being developed with Edwards Lifesciences, has performed well in valve-in-valve procedures in cadavers and animals. The first clinical trials are being planned.
“We need to think always that a bioprosthetic valve is not a cure, it’s a palliation. We treat the patients, they feel better, but we leave them with some kind of a chronic disease that’s prone to thrombosis, prone to degeneration and failure, prone to many different things,” he reflected.
The study was conducted without commercial support. Dr. Dvir reported serving as a consultant to Edwards Lifesciences, Medtronic, and St. Jude Medical.
PARIS – The first-ever study of the long-term durability of transcatheter bioprosthetic aortic valves has documented a disturbing rise in the valve degeneration rate occurring 5-7 years post implant.
Prior consistently reassuring follow-up studies have been intermediate in length, with a maximum of 5 years. The PARTNER 2A trial, which generated enormous enthusiasm for moving TAVR to intermediate-risk patients on the basis of positive results presented at the 2016 meeting of the American College of Cardiology, reported 2-year results.
“We found, as have others, that there’s very little degeneration in the first 4 years: 94% freedom from degeneration. But at 6 years, it’s 82%, and we estimate that by 8 years, it’s about 50%,” Dr. Danny Dvir reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
“We need to be cautious: This is our first look at the data. But we have a signal for a problem,” said Dr. Dvir of St. Paul’s Hospital in Vancouver.
He presented a retrospective study of 378 patients who underwent TAVR 5-14 years ago at two pioneering centers for the procedure: St. Paul’s and Charles Nicolle Hospital in Rouen, France. Patients’ average age at the time of TAVR was 82.3 years, with an STS score of 8.3%. The study featured serial echocardiography conducted during house calls in this frail elderly population.
Thirty-five patients developed prosthetic valve degeneration, defined by at least moderate regurgitation and/or a mean gradient of at least 20 mm Hg in 23 cases and stenosis in 12. The risk of degeneration was unrelated to the use of warfarin, a finding that suggests the valve deterioration issue is unrelated to clotting. The strongest risk factor for transcatheter valve degeneration in this study was baseline renal failure at the time of TAVR.
Dr. Dvir’s presentation was the talk of the meeting, and it cast a pall over the proceedings. The red flag raised by the study regarding valve durability has major implications regarding the current enthusiasm among many interventional cardiologists to routinely extend TAVR to intermediate and even lower-risk patients. As one audience member later confessed, “I have felt sick since hearing that presentation.”
Discussant Dr. A. Pieter Kappetein observed that transcatheter heart valve durability was a hot topic of discussion about 4 years ago but subsequently faded below the radar as a concern – until Dr. Dvir’s study.
“This is a very important study that puts transcatheter heart valve implantation in a little bit different light,” said Dr. Kappetein, professor of cardiothoracic surgery at Erasmus University in Rotterdam, the Netherlands.
He noted that the surgical aortic valve replacement (SAVR) literature shows that the rate of structural valve deterioration is age related. It’s higher in 75-year-olds than in 85-year-olds, and higher still in 65-year-olds.
“Valve degeneration didn’t play a major role when we were doing TAVR in 80- and 85-year-olds because of their limited life expectancy, but it will play a role in younger patients. So I think we have to be careful before we move toward lower-risk patients,” the surgeon continued.
Dr. Jean-Francois Obadia, who performs both SAVR and TAVR, noted that the median duration of freedom from valve degeneration for Edwards Lifescience’s Carpentier surgical aortic valve is a hefty 17.9 years.
“This should be the gold standard,” declared Dr. Obadia, head of the department of adult cardiovascular surgery and heart transplantation at the Louis Pradel Cardiothoracic Hospital of Claude Bernard University in Lyon, France.
“Dr. Dvir’s study is one of the key messages we all should take back home: a 50% rate of valve deterioration at 8 years. Valve deterioration is the Achilles’ heel of bioprostheses. There is a lot of improvement left to do for the TAVR,” he said.
Dr. Dvir and others were quick to note that his long-term study was of necessity restricted to early-generation, balloon-expandable devices: the Cribier Edwards, Edwards Sapien, and Sapien XT valves. Contemporary valves, patient selection methods, and procedural techniques are far advanced in comparison.
“The Sapien 3 has much less paravalvular leakage than earlier-generation valves. Maybe with less paravalvular leakage and better hemodynamics there will be a decreased rate of degeneration. It could be. We need to see. We have to wait a few more years to see if later-generation transcatheter heart valves are more durable,” Dr. Dvir said.
To gain a better understanding of the full dimensions of the valve degeneration issue, he and his coinvestigators have formed the VALID (VAlve Long-term Durability International Data) registry. Operators interested in contributing patients to what is hoped will be a very large and informative data base are encouraged to contact Dr. Dvir (ddvir@providencehealth.bc.ca).
In the meantime, he has reservations about extending TAVR to intermediate-risk patients outside of the rigorous clinical trial setting. He added that he’d feel far more comfortable in performing TAVR in intermediate-risk 70- to 75-year-olds if there was a tried and true valve-in-valve replacement method, something that doesn’t yet exist. The major limitation of current attempts at valve-in-valve replacement is underexpansion because the former valve doesn’t allow sufficient room for the new one to expand fully, resulting in residual stenosis.
“If you tell me that you can implant a platform that will enable a safe valve-in-valve procedure in 5, 7, 10 years – a less invasive bailout for a failed prosthetic valve – if you can do that safely and effectively I would be more keen to do TAVR even in a young patient,” the interventional cardiologist said.
He and others are working on this unmet need. Dr. Dvir’s novel valve, being developed with Edwards Lifesciences, has performed well in valve-in-valve procedures in cadavers and animals. The first clinical trials are being planned.
“We need to think always that a bioprosthetic valve is not a cure, it’s a palliation. We treat the patients, they feel better, but we leave them with some kind of a chronic disease that’s prone to thrombosis, prone to degeneration and failure, prone to many different things,” he reflected.
The study was conducted without commercial support. Dr. Dvir reported serving as a consultant to Edwards Lifesciences, Medtronic, and St. Jude Medical.
PARIS – The first-ever study of the long-term durability of transcatheter bioprosthetic aortic valves has documented a disturbing rise in the valve degeneration rate occurring 5-7 years post implant.
Prior consistently reassuring follow-up studies have been intermediate in length, with a maximum of 5 years. The PARTNER 2A trial, which generated enormous enthusiasm for moving TAVR to intermediate-risk patients on the basis of positive results presented at the 2016 meeting of the American College of Cardiology, reported 2-year results.
“We found, as have others, that there’s very little degeneration in the first 4 years: 94% freedom from degeneration. But at 6 years, it’s 82%, and we estimate that by 8 years, it’s about 50%,” Dr. Danny Dvir reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
“We need to be cautious: This is our first look at the data. But we have a signal for a problem,” said Dr. Dvir of St. Paul’s Hospital in Vancouver.
He presented a retrospective study of 378 patients who underwent TAVR 5-14 years ago at two pioneering centers for the procedure: St. Paul’s and Charles Nicolle Hospital in Rouen, France. Patients’ average age at the time of TAVR was 82.3 years, with an STS score of 8.3%. The study featured serial echocardiography conducted during house calls in this frail elderly population.
Thirty-five patients developed prosthetic valve degeneration, defined by at least moderate regurgitation and/or a mean gradient of at least 20 mm Hg in 23 cases and stenosis in 12. The risk of degeneration was unrelated to the use of warfarin, a finding that suggests the valve deterioration issue is unrelated to clotting. The strongest risk factor for transcatheter valve degeneration in this study was baseline renal failure at the time of TAVR.
Dr. Dvir’s presentation was the talk of the meeting, and it cast a pall over the proceedings. The red flag raised by the study regarding valve durability has major implications regarding the current enthusiasm among many interventional cardiologists to routinely extend TAVR to intermediate and even lower-risk patients. As one audience member later confessed, “I have felt sick since hearing that presentation.”
Discussant Dr. A. Pieter Kappetein observed that transcatheter heart valve durability was a hot topic of discussion about 4 years ago but subsequently faded below the radar as a concern – until Dr. Dvir’s study.
“This is a very important study that puts transcatheter heart valve implantation in a little bit different light,” said Dr. Kappetein, professor of cardiothoracic surgery at Erasmus University in Rotterdam, the Netherlands.
He noted that the surgical aortic valve replacement (SAVR) literature shows that the rate of structural valve deterioration is age related. It’s higher in 75-year-olds than in 85-year-olds, and higher still in 65-year-olds.
“Valve degeneration didn’t play a major role when we were doing TAVR in 80- and 85-year-olds because of their limited life expectancy, but it will play a role in younger patients. So I think we have to be careful before we move toward lower-risk patients,” the surgeon continued.
Dr. Jean-Francois Obadia, who performs both SAVR and TAVR, noted that the median duration of freedom from valve degeneration for Edwards Lifescience’s Carpentier surgical aortic valve is a hefty 17.9 years.
“This should be the gold standard,” declared Dr. Obadia, head of the department of adult cardiovascular surgery and heart transplantation at the Louis Pradel Cardiothoracic Hospital of Claude Bernard University in Lyon, France.
“Dr. Dvir’s study is one of the key messages we all should take back home: a 50% rate of valve deterioration at 8 years. Valve deterioration is the Achilles’ heel of bioprostheses. There is a lot of improvement left to do for the TAVR,” he said.
Dr. Dvir and others were quick to note that his long-term study was of necessity restricted to early-generation, balloon-expandable devices: the Cribier Edwards, Edwards Sapien, and Sapien XT valves. Contemporary valves, patient selection methods, and procedural techniques are far advanced in comparison.
“The Sapien 3 has much less paravalvular leakage than earlier-generation valves. Maybe with less paravalvular leakage and better hemodynamics there will be a decreased rate of degeneration. It could be. We need to see. We have to wait a few more years to see if later-generation transcatheter heart valves are more durable,” Dr. Dvir said.
To gain a better understanding of the full dimensions of the valve degeneration issue, he and his coinvestigators have formed the VALID (VAlve Long-term Durability International Data) registry. Operators interested in contributing patients to what is hoped will be a very large and informative data base are encouraged to contact Dr. Dvir (ddvir@providencehealth.bc.ca).
In the meantime, he has reservations about extending TAVR to intermediate-risk patients outside of the rigorous clinical trial setting. He added that he’d feel far more comfortable in performing TAVR in intermediate-risk 70- to 75-year-olds if there was a tried and true valve-in-valve replacement method, something that doesn’t yet exist. The major limitation of current attempts at valve-in-valve replacement is underexpansion because the former valve doesn’t allow sufficient room for the new one to expand fully, resulting in residual stenosis.
“If you tell me that you can implant a platform that will enable a safe valve-in-valve procedure in 5, 7, 10 years – a less invasive bailout for a failed prosthetic valve – if you can do that safely and effectively I would be more keen to do TAVR even in a young patient,” the interventional cardiologist said.
He and others are working on this unmet need. Dr. Dvir’s novel valve, being developed with Edwards Lifesciences, has performed well in valve-in-valve procedures in cadavers and animals. The first clinical trials are being planned.
“We need to think always that a bioprosthetic valve is not a cure, it’s a palliation. We treat the patients, they feel better, but we leave them with some kind of a chronic disease that’s prone to thrombosis, prone to degeneration and failure, prone to many different things,” he reflected.
The study was conducted without commercial support. Dr. Dvir reported serving as a consultant to Edwards Lifesciences, Medtronic, and St. Jude Medical.
AT EUROPCR 2016
Key clinical point: The first study to examine transcatheter aortic bioprosthetic valve performance beyond 5 years has found a 50% rate of valve degeneration 8 years post TAVR.
Major finding: A sharp increase in the incidence of degeneration of these early-generation valves occurred 5-7 years post TAVR.
Data source: This retrospective study featured serial home echocardiography in 378 patients who underwent TAVR 5-14 years ago at two pioneering centers for the procedure.
Disclosures: The presenter of this study, conducted without commercial support, serves as a consultant to Edwards Lifesciences, Medtronic, and St. Jude Medical.
Few locally advanced cervical cancer patients get guideline-directed therapies
SAN DIEGO – Failure to adhere to category 1 National Comprehensive Cancer Network guidelines for the management of locally advanced stage IIB-IVA cervical cancer is rampant – and it’s associated with significantly reduced survival, Dr. Jennifer L. Brown reported at the annual meeting of the Society of Gynecologic Oncology.
“Our study demonstrates opportunities to improve quality of care for women with locally advanced cervical cancer,” said Dr. Brown, a gynecologic oncologist at Fox Chase Cancer Center in Philadelphia.
She and her coinvestigators analyzed the American College of Surgeons/American Cancer Society National Cancer Data Base registry for 2002-2012 and identified 24,952 women treated for stage IIB-IVA cervical cancer. The investigators’ interest was in examining patterns of adherence to category 1 National Comprehensive Cancer Network (NCCN) guidelines in this population, and how adherence rates changed during the decade-long study period.
The key category 1 guidelines recommend concurrent radiation and cisplatin-based chemotherapy followed by brachytherapy for the primary treatment of locally advanced cervical cancer. Moreover, prior to initiating this therapy, pelvic and para-aortic lymph node involvement should be assessed with imaging or surgical staging in order to guide the appropriate radiotherapy dose.
Overall, according to Dr. Brown, only 5,935 women (23.8%) were treated in accordance with the NCCN guidelines recommending concurrent radiation and cisplatin-based chemotherapy followed by brachytherapy. Their median survival was 99.8 months, compared with 52.5 months for patients who didn’t receive category 1 treatment.
Similarly, a mere 23.5% of patients with locally advanced cervical cancer underwent pretreatment lymph node assessment. Those who did had a huge advantage in subsequent median survival: 123.2 months, compared with 48.6 months in those who didn’t undergo pretreatment lymph node staging, she continued.
Also, survival was significantly better among patients whose primary therapy was completed in 8 weeks or less: a median survival of 115.4 versus 82.3 months.
Nonadherence to the category 1 guidelines was disturbingly more common for African Americans, Hispanics, women over age 70, and those who were low income or had a low education level.
Compliance with the NCCN treatment guidelines improved over the study period, from roughly 19% in 2002 to 26% by 2012. However, adherence to the pretreatment lymph node assessment recommendations actually declined over time.
Dr. Brown found marked regional variation in adherence to the NCCN guidelines. It was greatest in the Rocky Mountain region and lowest in Kentucky, Tennessee, Mississippi, and Alabama.
She reported having no relevant financial conflicts.
SAN DIEGO – Failure to adhere to category 1 National Comprehensive Cancer Network guidelines for the management of locally advanced stage IIB-IVA cervical cancer is rampant – and it’s associated with significantly reduced survival, Dr. Jennifer L. Brown reported at the annual meeting of the Society of Gynecologic Oncology.
“Our study demonstrates opportunities to improve quality of care for women with locally advanced cervical cancer,” said Dr. Brown, a gynecologic oncologist at Fox Chase Cancer Center in Philadelphia.
She and her coinvestigators analyzed the American College of Surgeons/American Cancer Society National Cancer Data Base registry for 2002-2012 and identified 24,952 women treated for stage IIB-IVA cervical cancer. The investigators’ interest was in examining patterns of adherence to category 1 National Comprehensive Cancer Network (NCCN) guidelines in this population, and how adherence rates changed during the decade-long study period.
The key category 1 guidelines recommend concurrent radiation and cisplatin-based chemotherapy followed by brachytherapy for the primary treatment of locally advanced cervical cancer. Moreover, prior to initiating this therapy, pelvic and para-aortic lymph node involvement should be assessed with imaging or surgical staging in order to guide the appropriate radiotherapy dose.
Overall, according to Dr. Brown, only 5,935 women (23.8%) were treated in accordance with the NCCN guidelines recommending concurrent radiation and cisplatin-based chemotherapy followed by brachytherapy. Their median survival was 99.8 months, compared with 52.5 months for patients who didn’t receive category 1 treatment.
Similarly, a mere 23.5% of patients with locally advanced cervical cancer underwent pretreatment lymph node assessment. Those who did had a huge advantage in subsequent median survival: 123.2 months, compared with 48.6 months in those who didn’t undergo pretreatment lymph node staging, she continued.
Also, survival was significantly better among patients whose primary therapy was completed in 8 weeks or less: a median survival of 115.4 versus 82.3 months.
Nonadherence to the category 1 guidelines was disturbingly more common for African Americans, Hispanics, women over age 70, and those who were low income or had a low education level.
Compliance with the NCCN treatment guidelines improved over the study period, from roughly 19% in 2002 to 26% by 2012. However, adherence to the pretreatment lymph node assessment recommendations actually declined over time.
Dr. Brown found marked regional variation in adherence to the NCCN guidelines. It was greatest in the Rocky Mountain region and lowest in Kentucky, Tennessee, Mississippi, and Alabama.
She reported having no relevant financial conflicts.
SAN DIEGO – Failure to adhere to category 1 National Comprehensive Cancer Network guidelines for the management of locally advanced stage IIB-IVA cervical cancer is rampant – and it’s associated with significantly reduced survival, Dr. Jennifer L. Brown reported at the annual meeting of the Society of Gynecologic Oncology.
“Our study demonstrates opportunities to improve quality of care for women with locally advanced cervical cancer,” said Dr. Brown, a gynecologic oncologist at Fox Chase Cancer Center in Philadelphia.
She and her coinvestigators analyzed the American College of Surgeons/American Cancer Society National Cancer Data Base registry for 2002-2012 and identified 24,952 women treated for stage IIB-IVA cervical cancer. The investigators’ interest was in examining patterns of adherence to category 1 National Comprehensive Cancer Network (NCCN) guidelines in this population, and how adherence rates changed during the decade-long study period.
The key category 1 guidelines recommend concurrent radiation and cisplatin-based chemotherapy followed by brachytherapy for the primary treatment of locally advanced cervical cancer. Moreover, prior to initiating this therapy, pelvic and para-aortic lymph node involvement should be assessed with imaging or surgical staging in order to guide the appropriate radiotherapy dose.
Overall, according to Dr. Brown, only 5,935 women (23.8%) were treated in accordance with the NCCN guidelines recommending concurrent radiation and cisplatin-based chemotherapy followed by brachytherapy. Their median survival was 99.8 months, compared with 52.5 months for patients who didn’t receive category 1 treatment.
Similarly, a mere 23.5% of patients with locally advanced cervical cancer underwent pretreatment lymph node assessment. Those who did had a huge advantage in subsequent median survival: 123.2 months, compared with 48.6 months in those who didn’t undergo pretreatment lymph node staging, she continued.
Also, survival was significantly better among patients whose primary therapy was completed in 8 weeks or less: a median survival of 115.4 versus 82.3 months.
Nonadherence to the category 1 guidelines was disturbingly more common for African Americans, Hispanics, women over age 70, and those who were low income or had a low education level.
Compliance with the NCCN treatment guidelines improved over the study period, from roughly 19% in 2002 to 26% by 2012. However, adherence to the pretreatment lymph node assessment recommendations actually declined over time.
Dr. Brown found marked regional variation in adherence to the NCCN guidelines. It was greatest in the Rocky Mountain region and lowest in Kentucky, Tennessee, Mississippi, and Alabama.
She reported having no relevant financial conflicts.
AT THE ANNUAL MEETING ON WOMEN’S CANCER
Key clinical point: Only about one in four women with locally advanced cervical cancer receive guideline-recommended treatment.
Major finding: Median survival was 99.8 months among the 23.8% of women whose locally advanced cervical cancer was treated in accord with category 1 NCCN guidelines, compared with 52.5 months when it was not.
Data source: A retrospective study of the National Cancer Data Base including nearly 25,000 women treated for stage IIB-IVA cervical cancer.
Disclosures: The presenter reported having no relevant financial conflicts.
Cangrelor offers advantages for antiplatelet management in PCI
PARIS – Parenteral cangrelor is a potent antiplatelet agent that provides similar anti-ischemic effects but a significantly lower risk of major bleeding than the popular combination of clopidogrel plus a glycoprotein IIb/IIIa inhibitor in patients undergoing elective or urgent percutaneous coronary intervention.
Moreover, cangrelor (Kengreal), a rapid-acting and reversible intravenous platelet P2Y12 inhibitor, cuts the blood transfusion requirement by more than half, Dr. Muthiah Vaduganathan reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
These findings, based upon a propensity score–matched exploratory pooled analysis of individual patient-level data from the three phase III CHAMPION trials, suggest that cangrelor alone is an attractive alternative to clopidogrel plus a glycoprotein IIb/IIIa inhibitor (GPI) for antiplatelet management in percutaneous coronary intervention (PCI), according to Dr. Vaduganathan of Brigham and Women’s Hospital, Boston.
CHAMPION PLATFORM (N Engl J Med 2009 Dec 10;361[24]:2330-41), CHAMPION PCI (N Engl J Med 2009 Dec 10;361[24]:2318-29), and CHAMPION PHOENIX (N Engl J Med 2013 Apr 11;368[14]:1303-13) collectively included nearly 25,000 patients randomized to cangrelor versus standard therapy for platelet inhibition in elective or urgent PCI. The trials, which established that cangrelor led to fewer ischemic complications within 30 days post PCI than did clopidogrel plus a GPI, led to Food and Drug Administration approval of the drug in 2015. Cangrelor is also approved in Europe.
The cangrelor and clopidogrel groups differed in terms of baseline bleeding risk. For example, the clopidogrel-treated patients were younger, more likely to be male, had fewer comorbid conditions, and more often presented with an acute coronary syndrome. In order to accurately assess the bleeding risk associated with the two antiplatelet strategies, Dr. Vaduganathan and coinvestigators engaged in propensity score matching on the basis of 16 baseline clinical variables. They identified two closely matched cohorts consisting of 1,021 patient pairs.
The 1,021 patients in the cangrelor-alone group had a 2.6% rate of the primary composite efficacy endpoint consisting of all-cause mortality, MI, ischemia-driven revascularization, or stent thrombosis at 48 hours. Although this was 21% lower than the 3.3% rate in the clopidogrel plus GPI group, the difference didn’t achieve statistical significance because of the relatively small sample size.
In contrast, the rate of major bleeding within 48 hours by ACUITY (Acute Catheterization and Urgent Intervention Triage) criteria did differ significantly between the two groups: 3.6% with cangrelor and 5.8% with clopidogrel plus a GPI, for a 39% relative risk reduction in favor of the newer agent. Major bleeding rates were also lower in the cangrelor-treated patients on the GUSTO (Global Utilization of Streptokinase and TPA for Occluded Coronary Arteries) and TIMI (Thrombolysis in Myocardial Infarction) scales, although those trends didn’t achieve significance. The blood transfusion requirement was significantly lower in the cangrelor group: 1% versus 2.1%, for a 55% relative risk reduction.
Dr. Vaduganathan said the CHAMPION investigators’ future plans include examining the possibility of a favorable pharmacologic interaction between cangrelor and bivalirudin, a combination seeing increasing use in the United States.
“Finally, I think it’ll be important moving forward to look at comparative effectiveness data – either observational or randomized – as well as cost-effectiveness data between potent parenteral antiplatelet strategies,” he concluded.
Session chair Dr. Anthony Gershlick of Glenfield General Hospital in Leicester, England, commented that while he found this analysis of the CHAMPION data quite interesting, they are of little relevance in Europe, where cardiologists seldom use GPIs as part of their up-front PCI management strategy, instead resorting to those agents mainly as bailout therapy.
The U.S. situation is very different, according to Dr. Vaduganathan. American cardiologists use GPI therapy in about one-third of PCIs.
The CHAMPION trials were funded by the Medicines Co. Dr. Vaduganathan reported having no relevant financial conflicts.
PARIS – Parenteral cangrelor is a potent antiplatelet agent that provides similar anti-ischemic effects but a significantly lower risk of major bleeding than the popular combination of clopidogrel plus a glycoprotein IIb/IIIa inhibitor in patients undergoing elective or urgent percutaneous coronary intervention.
Moreover, cangrelor (Kengreal), a rapid-acting and reversible intravenous platelet P2Y12 inhibitor, cuts the blood transfusion requirement by more than half, Dr. Muthiah Vaduganathan reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
These findings, based upon a propensity score–matched exploratory pooled analysis of individual patient-level data from the three phase III CHAMPION trials, suggest that cangrelor alone is an attractive alternative to clopidogrel plus a glycoprotein IIb/IIIa inhibitor (GPI) for antiplatelet management in percutaneous coronary intervention (PCI), according to Dr. Vaduganathan of Brigham and Women’s Hospital, Boston.
CHAMPION PLATFORM (N Engl J Med 2009 Dec 10;361[24]:2330-41), CHAMPION PCI (N Engl J Med 2009 Dec 10;361[24]:2318-29), and CHAMPION PHOENIX (N Engl J Med 2013 Apr 11;368[14]:1303-13) collectively included nearly 25,000 patients randomized to cangrelor versus standard therapy for platelet inhibition in elective or urgent PCI. The trials, which established that cangrelor led to fewer ischemic complications within 30 days post PCI than did clopidogrel plus a GPI, led to Food and Drug Administration approval of the drug in 2015. Cangrelor is also approved in Europe.
The cangrelor and clopidogrel groups differed in terms of baseline bleeding risk. For example, the clopidogrel-treated patients were younger, more likely to be male, had fewer comorbid conditions, and more often presented with an acute coronary syndrome. In order to accurately assess the bleeding risk associated with the two antiplatelet strategies, Dr. Vaduganathan and coinvestigators engaged in propensity score matching on the basis of 16 baseline clinical variables. They identified two closely matched cohorts consisting of 1,021 patient pairs.
The 1,021 patients in the cangrelor-alone group had a 2.6% rate of the primary composite efficacy endpoint consisting of all-cause mortality, MI, ischemia-driven revascularization, or stent thrombosis at 48 hours. Although this was 21% lower than the 3.3% rate in the clopidogrel plus GPI group, the difference didn’t achieve statistical significance because of the relatively small sample size.
In contrast, the rate of major bleeding within 48 hours by ACUITY (Acute Catheterization and Urgent Intervention Triage) criteria did differ significantly between the two groups: 3.6% with cangrelor and 5.8% with clopidogrel plus a GPI, for a 39% relative risk reduction in favor of the newer agent. Major bleeding rates were also lower in the cangrelor-treated patients on the GUSTO (Global Utilization of Streptokinase and TPA for Occluded Coronary Arteries) and TIMI (Thrombolysis in Myocardial Infarction) scales, although those trends didn’t achieve significance. The blood transfusion requirement was significantly lower in the cangrelor group: 1% versus 2.1%, for a 55% relative risk reduction.
Dr. Vaduganathan said the CHAMPION investigators’ future plans include examining the possibility of a favorable pharmacologic interaction between cangrelor and bivalirudin, a combination seeing increasing use in the United States.
“Finally, I think it’ll be important moving forward to look at comparative effectiveness data – either observational or randomized – as well as cost-effectiveness data between potent parenteral antiplatelet strategies,” he concluded.
Session chair Dr. Anthony Gershlick of Glenfield General Hospital in Leicester, England, commented that while he found this analysis of the CHAMPION data quite interesting, they are of little relevance in Europe, where cardiologists seldom use GPIs as part of their up-front PCI management strategy, instead resorting to those agents mainly as bailout therapy.
The U.S. situation is very different, according to Dr. Vaduganathan. American cardiologists use GPI therapy in about one-third of PCIs.
The CHAMPION trials were funded by the Medicines Co. Dr. Vaduganathan reported having no relevant financial conflicts.
PARIS – Parenteral cangrelor is a potent antiplatelet agent that provides similar anti-ischemic effects but a significantly lower risk of major bleeding than the popular combination of clopidogrel plus a glycoprotein IIb/IIIa inhibitor in patients undergoing elective or urgent percutaneous coronary intervention.
Moreover, cangrelor (Kengreal), a rapid-acting and reversible intravenous platelet P2Y12 inhibitor, cuts the blood transfusion requirement by more than half, Dr. Muthiah Vaduganathan reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
These findings, based upon a propensity score–matched exploratory pooled analysis of individual patient-level data from the three phase III CHAMPION trials, suggest that cangrelor alone is an attractive alternative to clopidogrel plus a glycoprotein IIb/IIIa inhibitor (GPI) for antiplatelet management in percutaneous coronary intervention (PCI), according to Dr. Vaduganathan of Brigham and Women’s Hospital, Boston.
CHAMPION PLATFORM (N Engl J Med 2009 Dec 10;361[24]:2330-41), CHAMPION PCI (N Engl J Med 2009 Dec 10;361[24]:2318-29), and CHAMPION PHOENIX (N Engl J Med 2013 Apr 11;368[14]:1303-13) collectively included nearly 25,000 patients randomized to cangrelor versus standard therapy for platelet inhibition in elective or urgent PCI. The trials, which established that cangrelor led to fewer ischemic complications within 30 days post PCI than did clopidogrel plus a GPI, led to Food and Drug Administration approval of the drug in 2015. Cangrelor is also approved in Europe.
The cangrelor and clopidogrel groups differed in terms of baseline bleeding risk. For example, the clopidogrel-treated patients were younger, more likely to be male, had fewer comorbid conditions, and more often presented with an acute coronary syndrome. In order to accurately assess the bleeding risk associated with the two antiplatelet strategies, Dr. Vaduganathan and coinvestigators engaged in propensity score matching on the basis of 16 baseline clinical variables. They identified two closely matched cohorts consisting of 1,021 patient pairs.
The 1,021 patients in the cangrelor-alone group had a 2.6% rate of the primary composite efficacy endpoint consisting of all-cause mortality, MI, ischemia-driven revascularization, or stent thrombosis at 48 hours. Although this was 21% lower than the 3.3% rate in the clopidogrel plus GPI group, the difference didn’t achieve statistical significance because of the relatively small sample size.
In contrast, the rate of major bleeding within 48 hours by ACUITY (Acute Catheterization and Urgent Intervention Triage) criteria did differ significantly between the two groups: 3.6% with cangrelor and 5.8% with clopidogrel plus a GPI, for a 39% relative risk reduction in favor of the newer agent. Major bleeding rates were also lower in the cangrelor-treated patients on the GUSTO (Global Utilization of Streptokinase and TPA for Occluded Coronary Arteries) and TIMI (Thrombolysis in Myocardial Infarction) scales, although those trends didn’t achieve significance. The blood transfusion requirement was significantly lower in the cangrelor group: 1% versus 2.1%, for a 55% relative risk reduction.
Dr. Vaduganathan said the CHAMPION investigators’ future plans include examining the possibility of a favorable pharmacologic interaction between cangrelor and bivalirudin, a combination seeing increasing use in the United States.
“Finally, I think it’ll be important moving forward to look at comparative effectiveness data – either observational or randomized – as well as cost-effectiveness data between potent parenteral antiplatelet strategies,” he concluded.
Session chair Dr. Anthony Gershlick of Glenfield General Hospital in Leicester, England, commented that while he found this analysis of the CHAMPION data quite interesting, they are of little relevance in Europe, where cardiologists seldom use GPIs as part of their up-front PCI management strategy, instead resorting to those agents mainly as bailout therapy.
The U.S. situation is very different, according to Dr. Vaduganathan. American cardiologists use GPI therapy in about one-third of PCIs.
The CHAMPION trials were funded by the Medicines Co. Dr. Vaduganathan reported having no relevant financial conflicts.
AT EUROPCR 2016
Key clinical point: Cangrelor in patients undergoing PCI offers similar protection against ischemic events, compared with clopidogrel plus a glycoprotein IIb/IIIa inhibitor, but with significantly less major bleeding risk.
Major finding: The 48-hour rate of major bleeding as defined by the ACUITY scale was 3.6% with cangrelor alone, compared with 5.8% with clopidogrel plus a glycoprotein IIb/IIIa inhibitor.
Data source: This propensity score–matched analysis included 1,021 pairs of patients drawn from the three phase III CHAMPION trials.
Disclosures: The CHAMPION trials were funded by the Medicines Co. The presenter reported having no relevant financial conflicts.
Prior pregnancy protects against TAVR complications
PARIS – Results of the first-ever all-female patient TAVR registry indicate that a history of pregnancy – albeit typically half a century or more ago – is strongly protective against stroke, major bleeding, and other serious adverse events within 30 days of undergoing transcatheter aortic valve replacement.
The explanation for this novel observation remains unclear and warrants further study. One plausible hypothesis: “We can postulate that there is a hormonal effect in pregnancy that is still present after 50 years,” Dr. Alaide Chieffo said in presenting the Women’s International Transcatheter Aortic Valve Implantation (WIN TAVI) registry findings at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
WIN TAVI is a prospective observational registry of women undergoing TAVR for symptomatic severe aortic stenosis. Dr. Chieffo reported on acute and 30-day outcomes in 1,019 women enrolled in the registry at 20 highly experienced TAVR centers in Europe and North America. The average age was 82.5 years and mean STS score was 8.3%, putting the women at intermediate to high risk. Nearly three-quarters of subjects were more older than 80 years. Most patients had at least three major procedural risk factors. Two-thirds of the women were deemed frail upon surgical assessment.
The rationale for creating the WIN TAVI registry lies in the fact that even though half of patients undergoing TAVR are women, their optimal treatment strategy remains undefined. This is an important issue, particularly in light of evidence to suggest that the superiority of transfemoral TAVR over surgical aortic valve replacement in the PARTNER 2A trial may have been driven by better outcomes in women than in men.
WIN TAVI is designed to evaluate the safety and performance of TAVR in women in real-world clinical practice. It’s also a unique opportunity to prospectively gather data on female sex-specific baseline characteristics that haven’t been collected in previous TAVR studies and to examine their influence on outcomes, explained Dr. Chieffo of San Raffaele Scientific Institute in Milan. The primary outcome was the 30-day standard Second Valve Academic Research Consortium (VARC 2) composite of all-cause mortality, stroke, life-threatening bleeding, major vascular complications, stage 2 or 3 acute kidney injury, coronary artery obstruction, or a repeat intervention for valve-related dysfunction. The incidence was 14.4%, including a 3.9% all-cause mortality rate, a 1.3% incidence of stroke, major vascular complications in 7.9% of women, and a 4.4% incidence of life-threatening bleeding.
“The findings of the current registry underscore the importance and safety of moving to a lower-risk population of women with TAVR,” Dr. Chieffo declared.
The VARC 2 rates seen in WIN TAVI are low in comparison to other reports in TAVR patients. Dr. Chieffo attributed this to several factors. Patients underwent current state-of-the-art TAVR procedures, with a percutaneous transfemoral approach used in 91% of cases. Only one-third of patients had general anesthesia; the rest underwent TAVR under conscious sedation or a local anesthetic. One-third of the procedures were carried out with sheath sizes of 16 French or less.
Moreover, 42% of patients received new-generation TAVR devices that are compatible with smaller sheath sizes, are completely or partially retrievable, and have a lower paravalvular leak rate than that of earlier devices. The predominantly female interventional cardiologists were all highly experienced, and more than three-quarters of patients were discharged on dual-antiplatelet therapy or an oral anticoagulant.
Seventy-two percent of women had a remote history of pregnancy. Their rate of the primary VARC 2 safety endpoint was 12.7%, compared to 18.9% in patients without a prior pregnancy. In a multivariate logistic regression analysis, prior pregnancy, regardless of whether it resulted in a live birth, was independently associated with a 43% reduction in the risk of a VARC 2 safety endpoint. The greater the number of a woman’s pregnancies, the larger the reduction in the rate of VARC 2 complications.
The other independent predictor of reduced risk was the use of a new-generation TAVR device, defined as any device commercially available in Europe other than the Edwards Sapien XT or Medtronic CoreValve. The newer devices were associated with a 41% relative risk reduction in the VARC 2 composite endpoint.
Risk factors for the primary safety endpoint were prior stroke, associated with an adjusted twofold increased risk; a left ventricular ejection fraction less than 30%, with a 2.6-fold increased risk; and advanced age.
Of note, while 17.5% of women had a history of osteoporosis, only one-fifth of them were on treatment for it. This may impose important limitations on their postprocedural functional recovery, according to Dr. Chieffo.
Further follow-up of WIN TAVI registry participants is planned at 12, 18, and 24 months post procedure.
The WIN TAVI registry is carried out with no external funding. All participating investigators worked gratis. Dr. Chieffo reported having no financial conflicts of interest.
PARIS – Results of the first-ever all-female patient TAVR registry indicate that a history of pregnancy – albeit typically half a century or more ago – is strongly protective against stroke, major bleeding, and other serious adverse events within 30 days of undergoing transcatheter aortic valve replacement.
The explanation for this novel observation remains unclear and warrants further study. One plausible hypothesis: “We can postulate that there is a hormonal effect in pregnancy that is still present after 50 years,” Dr. Alaide Chieffo said in presenting the Women’s International Transcatheter Aortic Valve Implantation (WIN TAVI) registry findings at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
WIN TAVI is a prospective observational registry of women undergoing TAVR for symptomatic severe aortic stenosis. Dr. Chieffo reported on acute and 30-day outcomes in 1,019 women enrolled in the registry at 20 highly experienced TAVR centers in Europe and North America. The average age was 82.5 years and mean STS score was 8.3%, putting the women at intermediate to high risk. Nearly three-quarters of subjects were more older than 80 years. Most patients had at least three major procedural risk factors. Two-thirds of the women were deemed frail upon surgical assessment.
The rationale for creating the WIN TAVI registry lies in the fact that even though half of patients undergoing TAVR are women, their optimal treatment strategy remains undefined. This is an important issue, particularly in light of evidence to suggest that the superiority of transfemoral TAVR over surgical aortic valve replacement in the PARTNER 2A trial may have been driven by better outcomes in women than in men.
WIN TAVI is designed to evaluate the safety and performance of TAVR in women in real-world clinical practice. It’s also a unique opportunity to prospectively gather data on female sex-specific baseline characteristics that haven’t been collected in previous TAVR studies and to examine their influence on outcomes, explained Dr. Chieffo of San Raffaele Scientific Institute in Milan. The primary outcome was the 30-day standard Second Valve Academic Research Consortium (VARC 2) composite of all-cause mortality, stroke, life-threatening bleeding, major vascular complications, stage 2 or 3 acute kidney injury, coronary artery obstruction, or a repeat intervention for valve-related dysfunction. The incidence was 14.4%, including a 3.9% all-cause mortality rate, a 1.3% incidence of stroke, major vascular complications in 7.9% of women, and a 4.4% incidence of life-threatening bleeding.
“The findings of the current registry underscore the importance and safety of moving to a lower-risk population of women with TAVR,” Dr. Chieffo declared.
The VARC 2 rates seen in WIN TAVI are low in comparison to other reports in TAVR patients. Dr. Chieffo attributed this to several factors. Patients underwent current state-of-the-art TAVR procedures, with a percutaneous transfemoral approach used in 91% of cases. Only one-third of patients had general anesthesia; the rest underwent TAVR under conscious sedation or a local anesthetic. One-third of the procedures were carried out with sheath sizes of 16 French or less.
Moreover, 42% of patients received new-generation TAVR devices that are compatible with smaller sheath sizes, are completely or partially retrievable, and have a lower paravalvular leak rate than that of earlier devices. The predominantly female interventional cardiologists were all highly experienced, and more than three-quarters of patients were discharged on dual-antiplatelet therapy or an oral anticoagulant.
Seventy-two percent of women had a remote history of pregnancy. Their rate of the primary VARC 2 safety endpoint was 12.7%, compared to 18.9% in patients without a prior pregnancy. In a multivariate logistic regression analysis, prior pregnancy, regardless of whether it resulted in a live birth, was independently associated with a 43% reduction in the risk of a VARC 2 safety endpoint. The greater the number of a woman’s pregnancies, the larger the reduction in the rate of VARC 2 complications.
The other independent predictor of reduced risk was the use of a new-generation TAVR device, defined as any device commercially available in Europe other than the Edwards Sapien XT or Medtronic CoreValve. The newer devices were associated with a 41% relative risk reduction in the VARC 2 composite endpoint.
Risk factors for the primary safety endpoint were prior stroke, associated with an adjusted twofold increased risk; a left ventricular ejection fraction less than 30%, with a 2.6-fold increased risk; and advanced age.
Of note, while 17.5% of women had a history of osteoporosis, only one-fifth of them were on treatment for it. This may impose important limitations on their postprocedural functional recovery, according to Dr. Chieffo.
Further follow-up of WIN TAVI registry participants is planned at 12, 18, and 24 months post procedure.
The WIN TAVI registry is carried out with no external funding. All participating investigators worked gratis. Dr. Chieffo reported having no financial conflicts of interest.
PARIS – Results of the first-ever all-female patient TAVR registry indicate that a history of pregnancy – albeit typically half a century or more ago – is strongly protective against stroke, major bleeding, and other serious adverse events within 30 days of undergoing transcatheter aortic valve replacement.
The explanation for this novel observation remains unclear and warrants further study. One plausible hypothesis: “We can postulate that there is a hormonal effect in pregnancy that is still present after 50 years,” Dr. Alaide Chieffo said in presenting the Women’s International Transcatheter Aortic Valve Implantation (WIN TAVI) registry findings at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
WIN TAVI is a prospective observational registry of women undergoing TAVR for symptomatic severe aortic stenosis. Dr. Chieffo reported on acute and 30-day outcomes in 1,019 women enrolled in the registry at 20 highly experienced TAVR centers in Europe and North America. The average age was 82.5 years and mean STS score was 8.3%, putting the women at intermediate to high risk. Nearly three-quarters of subjects were more older than 80 years. Most patients had at least three major procedural risk factors. Two-thirds of the women were deemed frail upon surgical assessment.
The rationale for creating the WIN TAVI registry lies in the fact that even though half of patients undergoing TAVR are women, their optimal treatment strategy remains undefined. This is an important issue, particularly in light of evidence to suggest that the superiority of transfemoral TAVR over surgical aortic valve replacement in the PARTNER 2A trial may have been driven by better outcomes in women than in men.
WIN TAVI is designed to evaluate the safety and performance of TAVR in women in real-world clinical practice. It’s also a unique opportunity to prospectively gather data on female sex-specific baseline characteristics that haven’t been collected in previous TAVR studies and to examine their influence on outcomes, explained Dr. Chieffo of San Raffaele Scientific Institute in Milan. The primary outcome was the 30-day standard Second Valve Academic Research Consortium (VARC 2) composite of all-cause mortality, stroke, life-threatening bleeding, major vascular complications, stage 2 or 3 acute kidney injury, coronary artery obstruction, or a repeat intervention for valve-related dysfunction. The incidence was 14.4%, including a 3.9% all-cause mortality rate, a 1.3% incidence of stroke, major vascular complications in 7.9% of women, and a 4.4% incidence of life-threatening bleeding.
“The findings of the current registry underscore the importance and safety of moving to a lower-risk population of women with TAVR,” Dr. Chieffo declared.
The VARC 2 rates seen in WIN TAVI are low in comparison to other reports in TAVR patients. Dr. Chieffo attributed this to several factors. Patients underwent current state-of-the-art TAVR procedures, with a percutaneous transfemoral approach used in 91% of cases. Only one-third of patients had general anesthesia; the rest underwent TAVR under conscious sedation or a local anesthetic. One-third of the procedures were carried out with sheath sizes of 16 French or less.
Moreover, 42% of patients received new-generation TAVR devices that are compatible with smaller sheath sizes, are completely or partially retrievable, and have a lower paravalvular leak rate than that of earlier devices. The predominantly female interventional cardiologists were all highly experienced, and more than three-quarters of patients were discharged on dual-antiplatelet therapy or an oral anticoagulant.
Seventy-two percent of women had a remote history of pregnancy. Their rate of the primary VARC 2 safety endpoint was 12.7%, compared to 18.9% in patients without a prior pregnancy. In a multivariate logistic regression analysis, prior pregnancy, regardless of whether it resulted in a live birth, was independently associated with a 43% reduction in the risk of a VARC 2 safety endpoint. The greater the number of a woman’s pregnancies, the larger the reduction in the rate of VARC 2 complications.
The other independent predictor of reduced risk was the use of a new-generation TAVR device, defined as any device commercially available in Europe other than the Edwards Sapien XT or Medtronic CoreValve. The newer devices were associated with a 41% relative risk reduction in the VARC 2 composite endpoint.
Risk factors for the primary safety endpoint were prior stroke, associated with an adjusted twofold increased risk; a left ventricular ejection fraction less than 30%, with a 2.6-fold increased risk; and advanced age.
Of note, while 17.5% of women had a history of osteoporosis, only one-fifth of them were on treatment for it. This may impose important limitations on their postprocedural functional recovery, according to Dr. Chieffo.
Further follow-up of WIN TAVI registry participants is planned at 12, 18, and 24 months post procedure.
The WIN TAVI registry is carried out with no external funding. All participating investigators worked gratis. Dr. Chieffo reported having no financial conflicts of interest.
AT EUROPCR 2016
Key clinical point: Initial results of the first all-female TAVR registry provide surprises.
Major finding: Women who underwent transcatheter aortic valve replacement were 43% less likely to experience a major adverse safety event within 30 days if they had a history of pregnancy.
Data source: WIN TAVI, a prospective observational registry including 1,019 women who underwent TAVR for symptomatic severe aortic stenosis at 20 European and North American centers.
Disclosures: The study is being conducted without external funding. The presenter reported having no financial conflicts of interest.
CT renders transesophageal echo largely avoidable in TAVR
PARIS – Advances in CT scanner technology make cardiac CT a viable alternative to transesophageal echocardiography for preprocedural detection of left atrial appendage thrombus in candidates for transcatheter aortic valve replacement, Dr. Paul D. Williams said at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
This strategy helps simplify transcatheter aortic valve replacement (TAVR). That has become a major goal for the field now that TAVR’s safety and effectiveness have been established, said Dr. Williams of James Cook University Hospital in Middlesbrough, England.
“With the use of CT as the preferred method for annular sizing prior to the procedure and the increasing use of conscious sedation, transesophageal echo can be avoided altogether in many patients. And identification of left atrial appendage thrombus [LAAT] on CT may lead to changes in management, including optimization of oral anticoagulation, the use of an embolic protection device, and possibly obtaining consent from patients for a higher risk of procedural stroke,” he added.
Transesophageal echocardiography (TEE) has long been considered the gold standard method for detecting LAAT. But it has several disadvantages: It’s invasive, requires heavy sedation, and poses a small risk of serious complications.
Dr. Williams presented a single-center prospective study involving 198 consecutive patients who underwent dual source CT scanning with retrospective gated acquisition and flash angiographic acquisition during their pre-TAVR workup. The study showed that atrial fibrillation (AF) is very common in TAVR candidates, that LAAT is far more common in the TAVR population with AF than in the general AF population, and that while TAVR can still be performed in patients with LAAT, the periprocedural stroke risk appears to be higher.
Of the 198 TAVR candidates, 32% had AF. Two independent cardiologists with CT expertise rated 11.1% of TAVR candidates as having definite LAAT on the basis of a filling defect in both phases of imaging. Another 83.8% were deemed to definitely not have LAAT, while in 5.1% of cases the CT image quality wasn’t sufficient to render a judgment.
“The literature would suggest only about 5% of patients in the general AF population have LAAT. The rate is much higher in a TAVR population,” the cardiologist observed.
As expected, AF was a strong predictor of LAAT being found on CT, with a 32% prevalence in the AF subgroup, compared with just 1.6% in patients without AF.
Ninety-eight patients with a diagnostic CT also had a TEE. Six of the eight with LAAT on CT also showed LAAT on TEE. Two patients had LAAT on CT but not TEE. Thus, CT had 100% sensitivity, 97.8% specificity, a 75% positive predictive value, and a 100% negative predictive value, Dr. Williams continued.
Of the 198 patients evaluated by CT, 124 actually underwent TAVR. AF was present in 34% of these patients, whose mean CHA2DS2-VASc score was 3.7. CT showed that 8.1% of the patients who had TAVR had definite LAAT, and 84.7% definitely did not.
Six of the 124 patients (4.8%) had a stroke during their hospital stay for TAVR. Two of the six had LAAT on their preprocedural CT; both were being anticoagulated with warfarin at the time. The other four patients with periprocedural stroke didn’t have AF, were negative for LAAT on preprocedural CT, and weren’t being anticoagulated.
“Importantly, in the overall TAVR cohort, 8 of the 10 patients with LAAT on CT did not have a clinically evident periprocedural stroke,” Dr. Williams noted.
Session chair Dr. Rajendra Makkar, director of interventional cardiology at the Cedars-Sinai Heart Institute in Los Angeles, commented, “Your study has very, very important implications for how we actually change the management of some of our patients. You’ve shown LAAT is much more important in TAVR patients than I’d thought. A stroke risk of 20% with LAAT versus 3.8% in patients without LAAT is an impressive difference.”
His take away lesson from the study, Dr. Makkar added, is that if a patient has a preprocedural CT scan that’s negative for LAAT, there’s no need to do a TEE. If CT is positive or nondefinitive, it makes sense to review the patient’s anticoagulation regimen, then bring the patient back a few weeks later for a TEE to see if the LAAT has resolved.
Dr. Williams replied that’s exactly the practice now being followed at his hospital. If CT shows LAAT in a patient with AF who’s already on warfarin, physicians will consider aiming for a higher target INR [international normalized ratio], or they’ll switch to one of the novel oral anticoagulants if there’s a warfarin compliance issue. Then they’ll bring the patient back for a TEE.
“If the TEE is positive for LAAT we will still offer them TAVR, with the use of an embolic protection device if the anatomy is suitable, but we will tell them that potentially they may be at higher risk of stroke,” Dr. Williams said.
He reported having no financial conflicts of interest regarding this study.
PARIS – Advances in CT scanner technology make cardiac CT a viable alternative to transesophageal echocardiography for preprocedural detection of left atrial appendage thrombus in candidates for transcatheter aortic valve replacement, Dr. Paul D. Williams said at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
This strategy helps simplify transcatheter aortic valve replacement (TAVR). That has become a major goal for the field now that TAVR’s safety and effectiveness have been established, said Dr. Williams of James Cook University Hospital in Middlesbrough, England.
“With the use of CT as the preferred method for annular sizing prior to the procedure and the increasing use of conscious sedation, transesophageal echo can be avoided altogether in many patients. And identification of left atrial appendage thrombus [LAAT] on CT may lead to changes in management, including optimization of oral anticoagulation, the use of an embolic protection device, and possibly obtaining consent from patients for a higher risk of procedural stroke,” he added.
Transesophageal echocardiography (TEE) has long been considered the gold standard method for detecting LAAT. But it has several disadvantages: It’s invasive, requires heavy sedation, and poses a small risk of serious complications.
Dr. Williams presented a single-center prospective study involving 198 consecutive patients who underwent dual source CT scanning with retrospective gated acquisition and flash angiographic acquisition during their pre-TAVR workup. The study showed that atrial fibrillation (AF) is very common in TAVR candidates, that LAAT is far more common in the TAVR population with AF than in the general AF population, and that while TAVR can still be performed in patients with LAAT, the periprocedural stroke risk appears to be higher.
Of the 198 TAVR candidates, 32% had AF. Two independent cardiologists with CT expertise rated 11.1% of TAVR candidates as having definite LAAT on the basis of a filling defect in both phases of imaging. Another 83.8% were deemed to definitely not have LAAT, while in 5.1% of cases the CT image quality wasn’t sufficient to render a judgment.
“The literature would suggest only about 5% of patients in the general AF population have LAAT. The rate is much higher in a TAVR population,” the cardiologist observed.
As expected, AF was a strong predictor of LAAT being found on CT, with a 32% prevalence in the AF subgroup, compared with just 1.6% in patients without AF.
Ninety-eight patients with a diagnostic CT also had a TEE. Six of the eight with LAAT on CT also showed LAAT on TEE. Two patients had LAAT on CT but not TEE. Thus, CT had 100% sensitivity, 97.8% specificity, a 75% positive predictive value, and a 100% negative predictive value, Dr. Williams continued.
Of the 198 patients evaluated by CT, 124 actually underwent TAVR. AF was present in 34% of these patients, whose mean CHA2DS2-VASc score was 3.7. CT showed that 8.1% of the patients who had TAVR had definite LAAT, and 84.7% definitely did not.
Six of the 124 patients (4.8%) had a stroke during their hospital stay for TAVR. Two of the six had LAAT on their preprocedural CT; both were being anticoagulated with warfarin at the time. The other four patients with periprocedural stroke didn’t have AF, were negative for LAAT on preprocedural CT, and weren’t being anticoagulated.
“Importantly, in the overall TAVR cohort, 8 of the 10 patients with LAAT on CT did not have a clinically evident periprocedural stroke,” Dr. Williams noted.
Session chair Dr. Rajendra Makkar, director of interventional cardiology at the Cedars-Sinai Heart Institute in Los Angeles, commented, “Your study has very, very important implications for how we actually change the management of some of our patients. You’ve shown LAAT is much more important in TAVR patients than I’d thought. A stroke risk of 20% with LAAT versus 3.8% in patients without LAAT is an impressive difference.”
His take away lesson from the study, Dr. Makkar added, is that if a patient has a preprocedural CT scan that’s negative for LAAT, there’s no need to do a TEE. If CT is positive or nondefinitive, it makes sense to review the patient’s anticoagulation regimen, then bring the patient back a few weeks later for a TEE to see if the LAAT has resolved.
Dr. Williams replied that’s exactly the practice now being followed at his hospital. If CT shows LAAT in a patient with AF who’s already on warfarin, physicians will consider aiming for a higher target INR [international normalized ratio], or they’ll switch to one of the novel oral anticoagulants if there’s a warfarin compliance issue. Then they’ll bring the patient back for a TEE.
“If the TEE is positive for LAAT we will still offer them TAVR, with the use of an embolic protection device if the anatomy is suitable, but we will tell them that potentially they may be at higher risk of stroke,” Dr. Williams said.
He reported having no financial conflicts of interest regarding this study.
PARIS – Advances in CT scanner technology make cardiac CT a viable alternative to transesophageal echocardiography for preprocedural detection of left atrial appendage thrombus in candidates for transcatheter aortic valve replacement, Dr. Paul D. Williams said at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
This strategy helps simplify transcatheter aortic valve replacement (TAVR). That has become a major goal for the field now that TAVR’s safety and effectiveness have been established, said Dr. Williams of James Cook University Hospital in Middlesbrough, England.
“With the use of CT as the preferred method for annular sizing prior to the procedure and the increasing use of conscious sedation, transesophageal echo can be avoided altogether in many patients. And identification of left atrial appendage thrombus [LAAT] on CT may lead to changes in management, including optimization of oral anticoagulation, the use of an embolic protection device, and possibly obtaining consent from patients for a higher risk of procedural stroke,” he added.
Transesophageal echocardiography (TEE) has long been considered the gold standard method for detecting LAAT. But it has several disadvantages: It’s invasive, requires heavy sedation, and poses a small risk of serious complications.
Dr. Williams presented a single-center prospective study involving 198 consecutive patients who underwent dual source CT scanning with retrospective gated acquisition and flash angiographic acquisition during their pre-TAVR workup. The study showed that atrial fibrillation (AF) is very common in TAVR candidates, that LAAT is far more common in the TAVR population with AF than in the general AF population, and that while TAVR can still be performed in patients with LAAT, the periprocedural stroke risk appears to be higher.
Of the 198 TAVR candidates, 32% had AF. Two independent cardiologists with CT expertise rated 11.1% of TAVR candidates as having definite LAAT on the basis of a filling defect in both phases of imaging. Another 83.8% were deemed to definitely not have LAAT, while in 5.1% of cases the CT image quality wasn’t sufficient to render a judgment.
“The literature would suggest only about 5% of patients in the general AF population have LAAT. The rate is much higher in a TAVR population,” the cardiologist observed.
As expected, AF was a strong predictor of LAAT being found on CT, with a 32% prevalence in the AF subgroup, compared with just 1.6% in patients without AF.
Ninety-eight patients with a diagnostic CT also had a TEE. Six of the eight with LAAT on CT also showed LAAT on TEE. Two patients had LAAT on CT but not TEE. Thus, CT had 100% sensitivity, 97.8% specificity, a 75% positive predictive value, and a 100% negative predictive value, Dr. Williams continued.
Of the 198 patients evaluated by CT, 124 actually underwent TAVR. AF was present in 34% of these patients, whose mean CHA2DS2-VASc score was 3.7. CT showed that 8.1% of the patients who had TAVR had definite LAAT, and 84.7% definitely did not.
Six of the 124 patients (4.8%) had a stroke during their hospital stay for TAVR. Two of the six had LAAT on their preprocedural CT; both were being anticoagulated with warfarin at the time. The other four patients with periprocedural stroke didn’t have AF, were negative for LAAT on preprocedural CT, and weren’t being anticoagulated.
“Importantly, in the overall TAVR cohort, 8 of the 10 patients with LAAT on CT did not have a clinically evident periprocedural stroke,” Dr. Williams noted.
Session chair Dr. Rajendra Makkar, director of interventional cardiology at the Cedars-Sinai Heart Institute in Los Angeles, commented, “Your study has very, very important implications for how we actually change the management of some of our patients. You’ve shown LAAT is much more important in TAVR patients than I’d thought. A stroke risk of 20% with LAAT versus 3.8% in patients without LAAT is an impressive difference.”
His take away lesson from the study, Dr. Makkar added, is that if a patient has a preprocedural CT scan that’s negative for LAAT, there’s no need to do a TEE. If CT is positive or nondefinitive, it makes sense to review the patient’s anticoagulation regimen, then bring the patient back a few weeks later for a TEE to see if the LAAT has resolved.
Dr. Williams replied that’s exactly the practice now being followed at his hospital. If CT shows LAAT in a patient with AF who’s already on warfarin, physicians will consider aiming for a higher target INR [international normalized ratio], or they’ll switch to one of the novel oral anticoagulants if there’s a warfarin compliance issue. Then they’ll bring the patient back for a TEE.
“If the TEE is positive for LAAT we will still offer them TAVR, with the use of an embolic protection device if the anatomy is suitable, but we will tell them that potentially they may be at higher risk of stroke,” Dr. Williams said.
He reported having no financial conflicts of interest regarding this study.
AT EUROPCR 2016
Key clinical point: It’s essential to look for left atrial appendage thrombus in all TAVR candidates, and second-generation CT scanning offers advantages for this purpose.
Major finding: Left atrial appendage thrombus was identified by CT in 11.1% of TAVR candidates.
Data source: A single-center study of 198 consecutive TAVR candidates who underwent CT scanning for detection of left atrial appendage thrombus as well as for annular sizing.
Disclosures: The presenter reported having no financial conflicts of interest regarding his study.