User login
Malignancy Risk Among Psoriasis Patients Treated With Interleukin Inhibitors: A Retrospective Matched-Cohort Study
Malignancy Risk Among Psoriasis Patients Treated With Interleukin Inhibitors: A Retrospective Matched-Cohort Study
To the Editor:
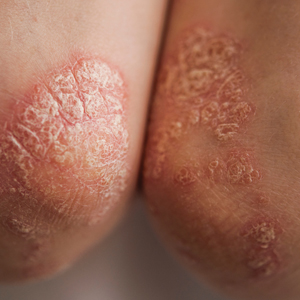
Psoriasis is a chronic immune-mediated inflammatory skin disease that affects approximately 2% to 3% of the global population and an estimated 7.5 million adults in the United States.1 The condition is characterized by recurrent episodes of erythematous scaly plaques driven by dysregulated immune responses, particularly involving the interleukin (IL) 23/T-helper (Th) 17 axis.2 Although cutaneous symptoms are the most visible manifestation, psoriasis is a systemic disorder with broad multisystem involvement. Comorbidities include psoriatic arthritis, metabolic syndrome, cardiovascular disease, inflammatory bowel disease, depression, and anxiety.1 These conditions contribute to a heightened risk for premature mortality, increased health care utilization, and an estimated direct cost burden exceeding $11 billion annually in the United States alone.3 Patients with moderate to severe disease frequently require systemic therapy, and long-term disease control is essential to prevent cumulative inflammatory damage and reduce associated morbidity.4
Globally, psoriasis prevalence and disease severity vary by geography, ethnicity, and environmental factors, with higher rates in Northern Europe and North America and lower reported prevalence in East Asia and sub-Saharan Africa.5 In lower-resource settings, access to advanced therapies is limited, and patients often are treated with less effective or more toxic systemic agents, such as methotrexate or cyclosporine.5 These disparities not only affect quality of life but also may influence comorbidity and malignancy patterns, underscoring the importance of studying biologic safety in diverse real-world populations.
Over the past decade, the therapeutic landscape for psoriasis has been transformed by biologic agents targeting specific immune pathways.6 Interleukin 17 inhibitors (eg, secukinumab, ixekizumab, brodalumab, bimekizumab) act by neutralizing IL-17A, IL-17F, or the IL-17 receptor, thereby reducing keratinocyte activation, neutrophil recruitment, and downstream cytokine production.6 Interleukin 23 inhibitors (eg, guselkumab, risankizumab, tildrakizumab) block the p19 subunit of IL-23, halting the expansion and maintenance of pathogenic Th17 cells.6 Ustekinumab, an IL-12/23 inhibitor, targets the shared p40 subunit of IL-12 and IL-23, attenuating both Th1 and Th17 signaling.6 These agents achieve rapid, durable skin clearance in a large proportion of patients, improve psoriatic arthritis symptoms, and generally are well tolerated, even with long-term use.6
Although efficacy is well established, the immunomodulatory nature of IL inhibitors raises theoretical concerns about malignancy risk. Immune surveillance plays a critical role in detecting and eliminating emerging tumor cells.7 Data from other systemic immunosuppressants, such as cyclosporine, show increased risks for certain cancers8; however, the IL-17 and IL-23 pathways have dual roles in cancer biology.7 In some tumor contexts, these cytokines promote carcinogenesis through angiogenesis, epithelial proliferation, and suppression of antitumor immunity; therefore, inhibiting these pathways could theoretically reduce cancer risk.7 The uncertainty around this risk-benefit balance has made malignancy a central consideration for dermatologists, particularly when initiating therapy in patients with a history of cancer or other risk factors.
The perception of malignancy risk can influence patient willingness to start biologics as well as physician prescribing patterns.9 Some clinicians opt for alternative therapies in individuals with a personal or family history of cancer despite limited direct evidence of harm from IL inhibitors. Conversely, a reassuring malignancy safety profile may support broader adoption of these therapies, especially in patients requiring lifelong disease control.9 Shared decision-making in this context requires robust, real-world evidence that accounts for both common and rare malignancy outcomes.
Randomized controlled trials of IL inhibitors have not demonstrated a consistent malignancy signal, but these studies often are underpowered for rare outcomes and limited by short follow-up durations, typically less than 1 year. They also frequently exclude high-risk populations, limiting generalizability.10 Observational studies using real-world data can address these gaps by including more diverse patient populations, longer observation windows, and larger sample sizes capable of detecting differences in uncommon outcomes.
The TriNetX Analytics Network (http://www.trinetx.com) offers a unique platform for large-scale, real-world pharmacoepidemiologic research. This federated database aggregates deidentified electronic health record data from more than 100 million patients across the United States and internationally, including at academic medical centers, integrated delivery networks, and community hospitals.4 Data contributors refresh their datasets regularly, ensuring near-contemporary representation of prescribing trends and clinical outcomes. Standardized terminology mapping, consistent International Classification of Diseases, Tenth Revision, Clinical Modification (ICD-10-CM) coding, and centralized data-quality checks enhance the reliability of analyses. Governance protocols and compliance with the Health Insurance Portability and Accountability Act deidentification standards further ensure ethical use of the data. The breadth and depth of the TriNetX network make it possible to evaluate not only common malignancies but also rare cancer types that smaller studies cannot assess with sufficient statistical power.
We performed a retrospective matched-cohort study, querying data from January 1, 2014, through December 31, 2024, using TriNetX to examine whether IL inhibitor exposure is associated with differences in incident malignancy risk among adults with psoriasis. Patients aged 18 years or older with a psoriasis diagnosis (ICD-10-CM code L40.x) and documented exposure to an IL-17, IL-23, or IL-12/23 inhibitor were eligible. Patients with a prior malignancy diagnosis were excluded to reduce prevalence bias. To ensure that malignancies were incident, we included only those diagnosed at least 1 day after initiation of an IL inhibitor.
The comparison cohort consisted of psoriasis patients without IL inhibitor exposure during their observation period. We used 1:1 propensity score matching based on age, sex, race, and ethnicity, applying a caliper of 0.1 to balance baseline characteristics and minimize demographic confounding. The index date for unexposed patients was randomly assigned within their observation period to align follow-up timing with exposed patients. Outcomes were identified by ICD-10-CM codes grouped by skin, hematologic, and solid-organ malignancies. Odds ratios (ORs) with 95% confidence intervals (CIs) were calculated, with statistical significance set at P<.05. Odds ratios were selected over hazard ratios due to variability in precise follow-up time capture and the primary analytic goal of comparing proportional odds within matched follow-up windows.
Propensity score matching was employed because it is a well-established technique in pharmacoepidemiology to mimic some of the balance achieved in randomized trials. By equating treatment and control groups on measured confounders, matching helps isolate the treatment effect, particularly important in nonrandomized datasets in which prescribing decisions may be influenced by baseline characteristics. Grouping cancers into clinically relevant categories allowed us to assess patterns of association, as some cancer types (eg, melanoma, lymphomas) may have pathophysiologic links to inflammatory pathways targeted by IL inhibitors.
The final cohort included 133,352 patients, with 66,676 in each group. The mean (SD) age was 49.3 (16.0) years, and demographic variables were well balanced after matching. The mean follow-up was approximately 3.8 years. Interleukin 17 inhibitors were the most frequently prescribed, followed by IL-23 inhibitors and ustekinumab. Baseline comorbidities such as cardiovascular disease, diabetes, and obesity were comparable between groups, reducing the likelihood of confounding from these factors.
Interleukin inhibitor exposure was associated with significantly reduced odds of several malignancies (eTable). Among skin cancers, melanoma risk was reduced by 36% (OR, 0.641; 95% CI, 0.534-0.77; P<.0001), basal cell carcinoma by 43% (OR, 0.565; 95% CI, 0.48-0.665; P<.0001), and squamous cell carcinoma by 18% (OR, 0.821; 95% CI, 0.676-0.996; P=.0452). Hematologic malignancies showed similar reductions, with non-Hodgkin lymphoma odds reduced by 35% (OR, 0.646; 95% CI, 0.512-0.815; P=.0002) and Hodgkin lymphoma by 50% (OR, 0.5; 95% CI, 0.292-0.855; P=.0098).
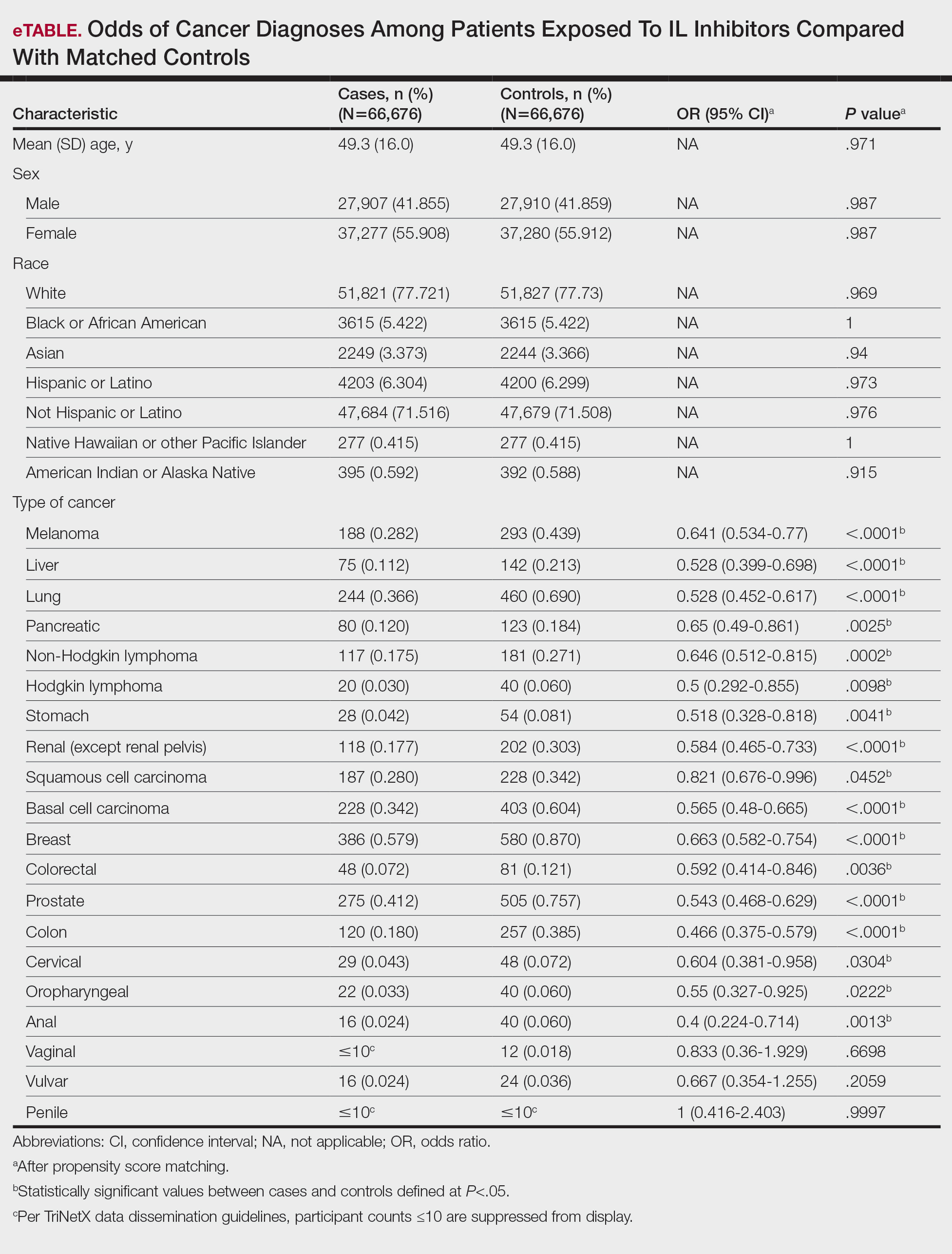
Protective associations also were observed for several solid tumors: lung (OR, 0.528; 95% CI, 0.452-0.617; P<.0001), liver (OR, 0.528; 95% CI, 0.399-0.698; P<.0001), pancreatic (OR, 0.65; 95% CI, 0.49-0.861; P=.0025), breast (OR, 0.663; 95% CI, 0.582-0.754; P<.0001), prostate (OR, 0.543; 95% CI, 0.468-0.629; P<.0001), colorectal (OR, 0.592; 95% CI, 0.414-0.846; P=.0036), colon (OR, 0.466; 95% CI, 0.375-0.579; P<.0001), and oropharyngeal (OR, 0.55; 95% CI, 0.327-0.925; P=.0222) cancers. Cervical cancer (OR, 0.604; 95% CI, 0.381-0.958; P=.0304) and anal cancer (OR, 0.4; 95% CI, 0.224-0.714; P=.0013) also showed significant reductions. Vaginal, vulvar, and penile cancers demonstrated no significant differences, likely due to their low incidence and limited statistical power.
The biological plausibility of these findings is supported by preclinical studies implicating IL-17 and IL-23 in tumor-promoting inflammation.11 These cytokines can recruit myeloid-derived suppressor cells, promote angiogenesis, and facilitate tumor-immune evasion. Inhibition may shift the immune microenvironment toward enhanced tumor surveillance, reduce protumorigenic cytokine signaling, and normalize regulatory T-cell function.11 These mechanisms could explain observed reductions in melanoma, lymphomas, and certain solid tumors.
Our results are consistent with several large registry studies showing no increased cancer incidence in IL inhibitor users and extend prior findings by demonstrating significant reductions in multiple cancer types.12 The melanoma reduction contrasts with the findings in earlier biologic safety studies, possibly due to our larger sample size, broader geographic representation, and inclusion of multiple IL inhibitor classes.13 Similar reductions have not been consistently observed with tumor necrosis factor α inhibitors, which have different immunologic targets and a more complex malignancy safety history.14
Limitations of our study include the retrospective design, potential misclassification of cancer diagnoses, and lack of data on unmeasured confounders such as sun exposure, smoking, alcohol use, and family cancer history. Surveillance bias is possible, though it would likely bias toward higher, not lower, cancer detection in biologic users. Our mean follow-up period of 3.8 years may not be sufficient for cancers with long latency periods.
If replicated, our findings could have meaningful public health implications. Reassurance regarding malignancy safety may increase patient acceptance and physician confidence in prescribing IL inhibitors, particularly for patients requiring long-term therapy. From a payer perspective, the potential for reduced cancer incidence could translate into substantial cost savings over time, offsetting the high up-front cost of biologics. Additionally, these results may be relevant to other IL inhibitor indications, including psoriatic arthritis, ankylosing spondylitis, and inflammatory bowel disease, in which similar pathophysiologic mechanisms may be at play.
In conclusion, this large matched-cohort study found that IL inhibitor therapy in psoriasis was associated with significantly reduced odds of multiple malignancies, including melanoma, lymphomas, and several solid tumors. These findings contribute to the growing body of real-world evidence supporting the long-term safety of IL inhibitors and underscore the need for continued pharmacovigilance and mechanistic research.
- Armstrong AW, Mehta MD, Schupp CW, et al. Psoriasis prevalence in adults in the United States. JAMA Dermatol. 2021;157:940-946. doi:10.1001/jamadermatol.2021.2007
- Deng Z, Wang S, Wu C, et al. IL-17 inhibitor-associated inflammatory bowel disease: a study based on literature and database analysis. Front Pharmacol. 2023;14:1124628. doi:10.3389/fphar.2023.1124628
- Al Sawah S, Foster SA, Goldblum OM, et al. Healthcare costs in psoriasis and psoriasis sub-groups over time following psoriasis diagnosis. J Med Econ. 2017;20:982-990. doi:10.1080/13696998.2017.1345749
- Korman NJ. Management of psoriasis as a systemic disease: what is the evidence? Br J Dermatol. 2020;182:840-848. doi:10.1111/bjd.18245
- Damiani G, Bragazzi NL, Karimkhani Aksut C, et al. The global, regional, and national burden of psoriasis: results and insights from the Global Burden of Disease 2019 Study. Front Med (Lausanne). 2021;8:743180. doi:10.3389/fmed.2021.743180
- Metko D, Torres T, Vender R. Viewpoint about biologic agents for psoriasis: are they immunosuppressants or immunomodulators? J Int Med Res. 2023;51:3000605231175547. doi:10.1177/03000605231175547
- Tsai YC, Tsai TF. Anti-interleukin and interleukin therapies for psoriasis: current evidence and clinical usefulness. Ther Adv Musculoskelet Dis. 2017;9:277-294. doi:10.1177/1759720X17735756
- Durnian JM, Stewart RM, Tatham R, et al. Cyclosporin-A associated malignancy. Clin Ophthalmol. 2007;1:421-430.
- DeWitt EM, Lin L, Glick HA, et al. Pattern and predictors of the initiation of biologic agents for the treatment of rheumatoid arthritis in the United States: an analysis using a large observational data bank. Clin Ther. 2009;31:1871-1858. doi:10.1016/j.clinthera.2009.08.020
- Vangilbergen M, Stockman A, Van De Velde A, et al. The role of interleukin-17 and interleukin-23 inhibitors in the development, progression, and recurrence of cancer: a systematic review. JAAD Int. 2024;17:71-79. doi:10.1016/j.jdin.2024.06.006
- Navarro-Compán V, Puig L, Vidal S, et al. The paradigm of IL-23-independent production of IL-17F and IL-17A and their role in chronic inflammatory diseases. Front Immunol. 2023;14:1191782. doi:10.3389/fimmu.2023.1191782
- Bencardino S, Bernardi F, Allocca M, et al. Advanced therapies for inflammatory bowel disease and risk of skin cancer: what’s new? Cancers (Basel). 2025;17:1710. doi:10.3390/cancers17101710
- Esse S, Mason KJ, Green AC, et al. Melanoma risk in patients treated with biologic therapy for common inflammatory diseases: a systematic review and meta-analysis. JAMA Dermatol. 2020;156:787-794. doi:10.1001/jamadermatol.2020.1300
- Solomon DH, Mercer E, Kavanaugh A. Observational studies on the risk of cancer associated with tumor necrosis factor inhibitors in rheumatoid arthritis: a review of their methodologies and results. Arthritis Rheum. 2012;64:21-32. doi:10.1002/art.30653
To the Editor:
Psoriasis is a chronic immune-mediated inflammatory skin disease that affects approximately 2% to 3% of the global population and an estimated 7.5 million adults in the United States.1 The condition is characterized by recurrent episodes of erythematous scaly plaques driven by dysregulated immune responses, particularly involving the interleukin (IL) 23/T-helper (Th) 17 axis.2 Although cutaneous symptoms are the most visible manifestation, psoriasis is a systemic disorder with broad multisystem involvement. Comorbidities include psoriatic arthritis, metabolic syndrome, cardiovascular disease, inflammatory bowel disease, depression, and anxiety.1 These conditions contribute to a heightened risk for premature mortality, increased health care utilization, and an estimated direct cost burden exceeding $11 billion annually in the United States alone.3 Patients with moderate to severe disease frequently require systemic therapy, and long-term disease control is essential to prevent cumulative inflammatory damage and reduce associated morbidity.4
Globally, psoriasis prevalence and disease severity vary by geography, ethnicity, and environmental factors, with higher rates in Northern Europe and North America and lower reported prevalence in East Asia and sub-Saharan Africa.5 In lower-resource settings, access to advanced therapies is limited, and patients often are treated with less effective or more toxic systemic agents, such as methotrexate or cyclosporine.5 These disparities not only affect quality of life but also may influence comorbidity and malignancy patterns, underscoring the importance of studying biologic safety in diverse real-world populations.
Over the past decade, the therapeutic landscape for psoriasis has been transformed by biologic agents targeting specific immune pathways.6 Interleukin 17 inhibitors (eg, secukinumab, ixekizumab, brodalumab, bimekizumab) act by neutralizing IL-17A, IL-17F, or the IL-17 receptor, thereby reducing keratinocyte activation, neutrophil recruitment, and downstream cytokine production.6 Interleukin 23 inhibitors (eg, guselkumab, risankizumab, tildrakizumab) block the p19 subunit of IL-23, halting the expansion and maintenance of pathogenic Th17 cells.6 Ustekinumab, an IL-12/23 inhibitor, targets the shared p40 subunit of IL-12 and IL-23, attenuating both Th1 and Th17 signaling.6 These agents achieve rapid, durable skin clearance in a large proportion of patients, improve psoriatic arthritis symptoms, and generally are well tolerated, even with long-term use.6
Although efficacy is well established, the immunomodulatory nature of IL inhibitors raises theoretical concerns about malignancy risk. Immune surveillance plays a critical role in detecting and eliminating emerging tumor cells.7 Data from other systemic immunosuppressants, such as cyclosporine, show increased risks for certain cancers8; however, the IL-17 and IL-23 pathways have dual roles in cancer biology.7 In some tumor contexts, these cytokines promote carcinogenesis through angiogenesis, epithelial proliferation, and suppression of antitumor immunity; therefore, inhibiting these pathways could theoretically reduce cancer risk.7 The uncertainty around this risk-benefit balance has made malignancy a central consideration for dermatologists, particularly when initiating therapy in patients with a history of cancer or other risk factors.
The perception of malignancy risk can influence patient willingness to start biologics as well as physician prescribing patterns.9 Some clinicians opt for alternative therapies in individuals with a personal or family history of cancer despite limited direct evidence of harm from IL inhibitors. Conversely, a reassuring malignancy safety profile may support broader adoption of these therapies, especially in patients requiring lifelong disease control.9 Shared decision-making in this context requires robust, real-world evidence that accounts for both common and rare malignancy outcomes.
Randomized controlled trials of IL inhibitors have not demonstrated a consistent malignancy signal, but these studies often are underpowered for rare outcomes and limited by short follow-up durations, typically less than 1 year. They also frequently exclude high-risk populations, limiting generalizability.10 Observational studies using real-world data can address these gaps by including more diverse patient populations, longer observation windows, and larger sample sizes capable of detecting differences in uncommon outcomes.
The TriNetX Analytics Network (http://www.trinetx.com) offers a unique platform for large-scale, real-world pharmacoepidemiologic research. This federated database aggregates deidentified electronic health record data from more than 100 million patients across the United States and internationally, including at academic medical centers, integrated delivery networks, and community hospitals.4 Data contributors refresh their datasets regularly, ensuring near-contemporary representation of prescribing trends and clinical outcomes. Standardized terminology mapping, consistent International Classification of Diseases, Tenth Revision, Clinical Modification (ICD-10-CM) coding, and centralized data-quality checks enhance the reliability of analyses. Governance protocols and compliance with the Health Insurance Portability and Accountability Act deidentification standards further ensure ethical use of the data. The breadth and depth of the TriNetX network make it possible to evaluate not only common malignancies but also rare cancer types that smaller studies cannot assess with sufficient statistical power.
We performed a retrospective matched-cohort study, querying data from January 1, 2014, through December 31, 2024, using TriNetX to examine whether IL inhibitor exposure is associated with differences in incident malignancy risk among adults with psoriasis. Patients aged 18 years or older with a psoriasis diagnosis (ICD-10-CM code L40.x) and documented exposure to an IL-17, IL-23, or IL-12/23 inhibitor were eligible. Patients with a prior malignancy diagnosis were excluded to reduce prevalence bias. To ensure that malignancies were incident, we included only those diagnosed at least 1 day after initiation of an IL inhibitor.
The comparison cohort consisted of psoriasis patients without IL inhibitor exposure during their observation period. We used 1:1 propensity score matching based on age, sex, race, and ethnicity, applying a caliper of 0.1 to balance baseline characteristics and minimize demographic confounding. The index date for unexposed patients was randomly assigned within their observation period to align follow-up timing with exposed patients. Outcomes were identified by ICD-10-CM codes grouped by skin, hematologic, and solid-organ malignancies. Odds ratios (ORs) with 95% confidence intervals (CIs) were calculated, with statistical significance set at P<.05. Odds ratios were selected over hazard ratios due to variability in precise follow-up time capture and the primary analytic goal of comparing proportional odds within matched follow-up windows.
Propensity score matching was employed because it is a well-established technique in pharmacoepidemiology to mimic some of the balance achieved in randomized trials. By equating treatment and control groups on measured confounders, matching helps isolate the treatment effect, particularly important in nonrandomized datasets in which prescribing decisions may be influenced by baseline characteristics. Grouping cancers into clinically relevant categories allowed us to assess patterns of association, as some cancer types (eg, melanoma, lymphomas) may have pathophysiologic links to inflammatory pathways targeted by IL inhibitors.
The final cohort included 133,352 patients, with 66,676 in each group. The mean (SD) age was 49.3 (16.0) years, and demographic variables were well balanced after matching. The mean follow-up was approximately 3.8 years. Interleukin 17 inhibitors were the most frequently prescribed, followed by IL-23 inhibitors and ustekinumab. Baseline comorbidities such as cardiovascular disease, diabetes, and obesity were comparable between groups, reducing the likelihood of confounding from these factors.
Interleukin inhibitor exposure was associated with significantly reduced odds of several malignancies (eTable). Among skin cancers, melanoma risk was reduced by 36% (OR, 0.641; 95% CI, 0.534-0.77; P<.0001), basal cell carcinoma by 43% (OR, 0.565; 95% CI, 0.48-0.665; P<.0001), and squamous cell carcinoma by 18% (OR, 0.821; 95% CI, 0.676-0.996; P=.0452). Hematologic malignancies showed similar reductions, with non-Hodgkin lymphoma odds reduced by 35% (OR, 0.646; 95% CI, 0.512-0.815; P=.0002) and Hodgkin lymphoma by 50% (OR, 0.5; 95% CI, 0.292-0.855; P=.0098).

Protective associations also were observed for several solid tumors: lung (OR, 0.528; 95% CI, 0.452-0.617; P<.0001), liver (OR, 0.528; 95% CI, 0.399-0.698; P<.0001), pancreatic (OR, 0.65; 95% CI, 0.49-0.861; P=.0025), breast (OR, 0.663; 95% CI, 0.582-0.754; P<.0001), prostate (OR, 0.543; 95% CI, 0.468-0.629; P<.0001), colorectal (OR, 0.592; 95% CI, 0.414-0.846; P=.0036), colon (OR, 0.466; 95% CI, 0.375-0.579; P<.0001), and oropharyngeal (OR, 0.55; 95% CI, 0.327-0.925; P=.0222) cancers. Cervical cancer (OR, 0.604; 95% CI, 0.381-0.958; P=.0304) and anal cancer (OR, 0.4; 95% CI, 0.224-0.714; P=.0013) also showed significant reductions. Vaginal, vulvar, and penile cancers demonstrated no significant differences, likely due to their low incidence and limited statistical power.
The biological plausibility of these findings is supported by preclinical studies implicating IL-17 and IL-23 in tumor-promoting inflammation.11 These cytokines can recruit myeloid-derived suppressor cells, promote angiogenesis, and facilitate tumor-immune evasion. Inhibition may shift the immune microenvironment toward enhanced tumor surveillance, reduce protumorigenic cytokine signaling, and normalize regulatory T-cell function.11 These mechanisms could explain observed reductions in melanoma, lymphomas, and certain solid tumors.
Our results are consistent with several large registry studies showing no increased cancer incidence in IL inhibitor users and extend prior findings by demonstrating significant reductions in multiple cancer types.12 The melanoma reduction contrasts with the findings in earlier biologic safety studies, possibly due to our larger sample size, broader geographic representation, and inclusion of multiple IL inhibitor classes.13 Similar reductions have not been consistently observed with tumor necrosis factor α inhibitors, which have different immunologic targets and a more complex malignancy safety history.14
Limitations of our study include the retrospective design, potential misclassification of cancer diagnoses, and lack of data on unmeasured confounders such as sun exposure, smoking, alcohol use, and family cancer history. Surveillance bias is possible, though it would likely bias toward higher, not lower, cancer detection in biologic users. Our mean follow-up period of 3.8 years may not be sufficient for cancers with long latency periods.
If replicated, our findings could have meaningful public health implications. Reassurance regarding malignancy safety may increase patient acceptance and physician confidence in prescribing IL inhibitors, particularly for patients requiring long-term therapy. From a payer perspective, the potential for reduced cancer incidence could translate into substantial cost savings over time, offsetting the high up-front cost of biologics. Additionally, these results may be relevant to other IL inhibitor indications, including psoriatic arthritis, ankylosing spondylitis, and inflammatory bowel disease, in which similar pathophysiologic mechanisms may be at play.
In conclusion, this large matched-cohort study found that IL inhibitor therapy in psoriasis was associated with significantly reduced odds of multiple malignancies, including melanoma, lymphomas, and several solid tumors. These findings contribute to the growing body of real-world evidence supporting the long-term safety of IL inhibitors and underscore the need for continued pharmacovigilance and mechanistic research.
To the Editor:
Psoriasis is a chronic immune-mediated inflammatory skin disease that affects approximately 2% to 3% of the global population and an estimated 7.5 million adults in the United States.1 The condition is characterized by recurrent episodes of erythematous scaly plaques driven by dysregulated immune responses, particularly involving the interleukin (IL) 23/T-helper (Th) 17 axis.2 Although cutaneous symptoms are the most visible manifestation, psoriasis is a systemic disorder with broad multisystem involvement. Comorbidities include psoriatic arthritis, metabolic syndrome, cardiovascular disease, inflammatory bowel disease, depression, and anxiety.1 These conditions contribute to a heightened risk for premature mortality, increased health care utilization, and an estimated direct cost burden exceeding $11 billion annually in the United States alone.3 Patients with moderate to severe disease frequently require systemic therapy, and long-term disease control is essential to prevent cumulative inflammatory damage and reduce associated morbidity.4
Globally, psoriasis prevalence and disease severity vary by geography, ethnicity, and environmental factors, with higher rates in Northern Europe and North America and lower reported prevalence in East Asia and sub-Saharan Africa.5 In lower-resource settings, access to advanced therapies is limited, and patients often are treated with less effective or more toxic systemic agents, such as methotrexate or cyclosporine.5 These disparities not only affect quality of life but also may influence comorbidity and malignancy patterns, underscoring the importance of studying biologic safety in diverse real-world populations.
Over the past decade, the therapeutic landscape for psoriasis has been transformed by biologic agents targeting specific immune pathways.6 Interleukin 17 inhibitors (eg, secukinumab, ixekizumab, brodalumab, bimekizumab) act by neutralizing IL-17A, IL-17F, or the IL-17 receptor, thereby reducing keratinocyte activation, neutrophil recruitment, and downstream cytokine production.6 Interleukin 23 inhibitors (eg, guselkumab, risankizumab, tildrakizumab) block the p19 subunit of IL-23, halting the expansion and maintenance of pathogenic Th17 cells.6 Ustekinumab, an IL-12/23 inhibitor, targets the shared p40 subunit of IL-12 and IL-23, attenuating both Th1 and Th17 signaling.6 These agents achieve rapid, durable skin clearance in a large proportion of patients, improve psoriatic arthritis symptoms, and generally are well tolerated, even with long-term use.6
Although efficacy is well established, the immunomodulatory nature of IL inhibitors raises theoretical concerns about malignancy risk. Immune surveillance plays a critical role in detecting and eliminating emerging tumor cells.7 Data from other systemic immunosuppressants, such as cyclosporine, show increased risks for certain cancers8; however, the IL-17 and IL-23 pathways have dual roles in cancer biology.7 In some tumor contexts, these cytokines promote carcinogenesis through angiogenesis, epithelial proliferation, and suppression of antitumor immunity; therefore, inhibiting these pathways could theoretically reduce cancer risk.7 The uncertainty around this risk-benefit balance has made malignancy a central consideration for dermatologists, particularly when initiating therapy in patients with a history of cancer or other risk factors.
The perception of malignancy risk can influence patient willingness to start biologics as well as physician prescribing patterns.9 Some clinicians opt for alternative therapies in individuals with a personal or family history of cancer despite limited direct evidence of harm from IL inhibitors. Conversely, a reassuring malignancy safety profile may support broader adoption of these therapies, especially in patients requiring lifelong disease control.9 Shared decision-making in this context requires robust, real-world evidence that accounts for both common and rare malignancy outcomes.
Randomized controlled trials of IL inhibitors have not demonstrated a consistent malignancy signal, but these studies often are underpowered for rare outcomes and limited by short follow-up durations, typically less than 1 year. They also frequently exclude high-risk populations, limiting generalizability.10 Observational studies using real-world data can address these gaps by including more diverse patient populations, longer observation windows, and larger sample sizes capable of detecting differences in uncommon outcomes.
The TriNetX Analytics Network (http://www.trinetx.com) offers a unique platform for large-scale, real-world pharmacoepidemiologic research. This federated database aggregates deidentified electronic health record data from more than 100 million patients across the United States and internationally, including at academic medical centers, integrated delivery networks, and community hospitals.4 Data contributors refresh their datasets regularly, ensuring near-contemporary representation of prescribing trends and clinical outcomes. Standardized terminology mapping, consistent International Classification of Diseases, Tenth Revision, Clinical Modification (ICD-10-CM) coding, and centralized data-quality checks enhance the reliability of analyses. Governance protocols and compliance with the Health Insurance Portability and Accountability Act deidentification standards further ensure ethical use of the data. The breadth and depth of the TriNetX network make it possible to evaluate not only common malignancies but also rare cancer types that smaller studies cannot assess with sufficient statistical power.
We performed a retrospective matched-cohort study, querying data from January 1, 2014, through December 31, 2024, using TriNetX to examine whether IL inhibitor exposure is associated with differences in incident malignancy risk among adults with psoriasis. Patients aged 18 years or older with a psoriasis diagnosis (ICD-10-CM code L40.x) and documented exposure to an IL-17, IL-23, or IL-12/23 inhibitor were eligible. Patients with a prior malignancy diagnosis were excluded to reduce prevalence bias. To ensure that malignancies were incident, we included only those diagnosed at least 1 day after initiation of an IL inhibitor.
The comparison cohort consisted of psoriasis patients without IL inhibitor exposure during their observation period. We used 1:1 propensity score matching based on age, sex, race, and ethnicity, applying a caliper of 0.1 to balance baseline characteristics and minimize demographic confounding. The index date for unexposed patients was randomly assigned within their observation period to align follow-up timing with exposed patients. Outcomes were identified by ICD-10-CM codes grouped by skin, hematologic, and solid-organ malignancies. Odds ratios (ORs) with 95% confidence intervals (CIs) were calculated, with statistical significance set at P<.05. Odds ratios were selected over hazard ratios due to variability in precise follow-up time capture and the primary analytic goal of comparing proportional odds within matched follow-up windows.
Propensity score matching was employed because it is a well-established technique in pharmacoepidemiology to mimic some of the balance achieved in randomized trials. By equating treatment and control groups on measured confounders, matching helps isolate the treatment effect, particularly important in nonrandomized datasets in which prescribing decisions may be influenced by baseline characteristics. Grouping cancers into clinically relevant categories allowed us to assess patterns of association, as some cancer types (eg, melanoma, lymphomas) may have pathophysiologic links to inflammatory pathways targeted by IL inhibitors.
The final cohort included 133,352 patients, with 66,676 in each group. The mean (SD) age was 49.3 (16.0) years, and demographic variables were well balanced after matching. The mean follow-up was approximately 3.8 years. Interleukin 17 inhibitors were the most frequently prescribed, followed by IL-23 inhibitors and ustekinumab. Baseline comorbidities such as cardiovascular disease, diabetes, and obesity were comparable between groups, reducing the likelihood of confounding from these factors.
Interleukin inhibitor exposure was associated with significantly reduced odds of several malignancies (eTable). Among skin cancers, melanoma risk was reduced by 36% (OR, 0.641; 95% CI, 0.534-0.77; P<.0001), basal cell carcinoma by 43% (OR, 0.565; 95% CI, 0.48-0.665; P<.0001), and squamous cell carcinoma by 18% (OR, 0.821; 95% CI, 0.676-0.996; P=.0452). Hematologic malignancies showed similar reductions, with non-Hodgkin lymphoma odds reduced by 35% (OR, 0.646; 95% CI, 0.512-0.815; P=.0002) and Hodgkin lymphoma by 50% (OR, 0.5; 95% CI, 0.292-0.855; P=.0098).

Protective associations also were observed for several solid tumors: lung (OR, 0.528; 95% CI, 0.452-0.617; P<.0001), liver (OR, 0.528; 95% CI, 0.399-0.698; P<.0001), pancreatic (OR, 0.65; 95% CI, 0.49-0.861; P=.0025), breast (OR, 0.663; 95% CI, 0.582-0.754; P<.0001), prostate (OR, 0.543; 95% CI, 0.468-0.629; P<.0001), colorectal (OR, 0.592; 95% CI, 0.414-0.846; P=.0036), colon (OR, 0.466; 95% CI, 0.375-0.579; P<.0001), and oropharyngeal (OR, 0.55; 95% CI, 0.327-0.925; P=.0222) cancers. Cervical cancer (OR, 0.604; 95% CI, 0.381-0.958; P=.0304) and anal cancer (OR, 0.4; 95% CI, 0.224-0.714; P=.0013) also showed significant reductions. Vaginal, vulvar, and penile cancers demonstrated no significant differences, likely due to their low incidence and limited statistical power.
The biological plausibility of these findings is supported by preclinical studies implicating IL-17 and IL-23 in tumor-promoting inflammation.11 These cytokines can recruit myeloid-derived suppressor cells, promote angiogenesis, and facilitate tumor-immune evasion. Inhibition may shift the immune microenvironment toward enhanced tumor surveillance, reduce protumorigenic cytokine signaling, and normalize regulatory T-cell function.11 These mechanisms could explain observed reductions in melanoma, lymphomas, and certain solid tumors.
Our results are consistent with several large registry studies showing no increased cancer incidence in IL inhibitor users and extend prior findings by demonstrating significant reductions in multiple cancer types.12 The melanoma reduction contrasts with the findings in earlier biologic safety studies, possibly due to our larger sample size, broader geographic representation, and inclusion of multiple IL inhibitor classes.13 Similar reductions have not been consistently observed with tumor necrosis factor α inhibitors, which have different immunologic targets and a more complex malignancy safety history.14
Limitations of our study include the retrospective design, potential misclassification of cancer diagnoses, and lack of data on unmeasured confounders such as sun exposure, smoking, alcohol use, and family cancer history. Surveillance bias is possible, though it would likely bias toward higher, not lower, cancer detection in biologic users. Our mean follow-up period of 3.8 years may not be sufficient for cancers with long latency periods.
If replicated, our findings could have meaningful public health implications. Reassurance regarding malignancy safety may increase patient acceptance and physician confidence in prescribing IL inhibitors, particularly for patients requiring long-term therapy. From a payer perspective, the potential for reduced cancer incidence could translate into substantial cost savings over time, offsetting the high up-front cost of biologics. Additionally, these results may be relevant to other IL inhibitor indications, including psoriatic arthritis, ankylosing spondylitis, and inflammatory bowel disease, in which similar pathophysiologic mechanisms may be at play.
In conclusion, this large matched-cohort study found that IL inhibitor therapy in psoriasis was associated with significantly reduced odds of multiple malignancies, including melanoma, lymphomas, and several solid tumors. These findings contribute to the growing body of real-world evidence supporting the long-term safety of IL inhibitors and underscore the need for continued pharmacovigilance and mechanistic research.
- Armstrong AW, Mehta MD, Schupp CW, et al. Psoriasis prevalence in adults in the United States. JAMA Dermatol. 2021;157:940-946. doi:10.1001/jamadermatol.2021.2007
- Deng Z, Wang S, Wu C, et al. IL-17 inhibitor-associated inflammatory bowel disease: a study based on literature and database analysis. Front Pharmacol. 2023;14:1124628. doi:10.3389/fphar.2023.1124628
- Al Sawah S, Foster SA, Goldblum OM, et al. Healthcare costs in psoriasis and psoriasis sub-groups over time following psoriasis diagnosis. J Med Econ. 2017;20:982-990. doi:10.1080/13696998.2017.1345749
- Korman NJ. Management of psoriasis as a systemic disease: what is the evidence? Br J Dermatol. 2020;182:840-848. doi:10.1111/bjd.18245
- Damiani G, Bragazzi NL, Karimkhani Aksut C, et al. The global, regional, and national burden of psoriasis: results and insights from the Global Burden of Disease 2019 Study. Front Med (Lausanne). 2021;8:743180. doi:10.3389/fmed.2021.743180
- Metko D, Torres T, Vender R. Viewpoint about biologic agents for psoriasis: are they immunosuppressants or immunomodulators? J Int Med Res. 2023;51:3000605231175547. doi:10.1177/03000605231175547
- Tsai YC, Tsai TF. Anti-interleukin and interleukin therapies for psoriasis: current evidence and clinical usefulness. Ther Adv Musculoskelet Dis. 2017;9:277-294. doi:10.1177/1759720X17735756
- Durnian JM, Stewart RM, Tatham R, et al. Cyclosporin-A associated malignancy. Clin Ophthalmol. 2007;1:421-430.
- DeWitt EM, Lin L, Glick HA, et al. Pattern and predictors of the initiation of biologic agents for the treatment of rheumatoid arthritis in the United States: an analysis using a large observational data bank. Clin Ther. 2009;31:1871-1858. doi:10.1016/j.clinthera.2009.08.020
- Vangilbergen M, Stockman A, Van De Velde A, et al. The role of interleukin-17 and interleukin-23 inhibitors in the development, progression, and recurrence of cancer: a systematic review. JAAD Int. 2024;17:71-79. doi:10.1016/j.jdin.2024.06.006
- Navarro-Compán V, Puig L, Vidal S, et al. The paradigm of IL-23-independent production of IL-17F and IL-17A and their role in chronic inflammatory diseases. Front Immunol. 2023;14:1191782. doi:10.3389/fimmu.2023.1191782
- Bencardino S, Bernardi F, Allocca M, et al. Advanced therapies for inflammatory bowel disease and risk of skin cancer: what’s new? Cancers (Basel). 2025;17:1710. doi:10.3390/cancers17101710
- Esse S, Mason KJ, Green AC, et al. Melanoma risk in patients treated with biologic therapy for common inflammatory diseases: a systematic review and meta-analysis. JAMA Dermatol. 2020;156:787-794. doi:10.1001/jamadermatol.2020.1300
- Solomon DH, Mercer E, Kavanaugh A. Observational studies on the risk of cancer associated with tumor necrosis factor inhibitors in rheumatoid arthritis: a review of their methodologies and results. Arthritis Rheum. 2012;64:21-32. doi:10.1002/art.30653
- Armstrong AW, Mehta MD, Schupp CW, et al. Psoriasis prevalence in adults in the United States. JAMA Dermatol. 2021;157:940-946. doi:10.1001/jamadermatol.2021.2007
- Deng Z, Wang S, Wu C, et al. IL-17 inhibitor-associated inflammatory bowel disease: a study based on literature and database analysis. Front Pharmacol. 2023;14:1124628. doi:10.3389/fphar.2023.1124628
- Al Sawah S, Foster SA, Goldblum OM, et al. Healthcare costs in psoriasis and psoriasis sub-groups over time following psoriasis diagnosis. J Med Econ. 2017;20:982-990. doi:10.1080/13696998.2017.1345749
- Korman NJ. Management of psoriasis as a systemic disease: what is the evidence? Br J Dermatol. 2020;182:840-848. doi:10.1111/bjd.18245
- Damiani G, Bragazzi NL, Karimkhani Aksut C, et al. The global, regional, and national burden of psoriasis: results and insights from the Global Burden of Disease 2019 Study. Front Med (Lausanne). 2021;8:743180. doi:10.3389/fmed.2021.743180
- Metko D, Torres T, Vender R. Viewpoint about biologic agents for psoriasis: are they immunosuppressants or immunomodulators? J Int Med Res. 2023;51:3000605231175547. doi:10.1177/03000605231175547
- Tsai YC, Tsai TF. Anti-interleukin and interleukin therapies for psoriasis: current evidence and clinical usefulness. Ther Adv Musculoskelet Dis. 2017;9:277-294. doi:10.1177/1759720X17735756
- Durnian JM, Stewart RM, Tatham R, et al. Cyclosporin-A associated malignancy. Clin Ophthalmol. 2007;1:421-430.
- DeWitt EM, Lin L, Glick HA, et al. Pattern and predictors of the initiation of biologic agents for the treatment of rheumatoid arthritis in the United States: an analysis using a large observational data bank. Clin Ther. 2009;31:1871-1858. doi:10.1016/j.clinthera.2009.08.020
- Vangilbergen M, Stockman A, Van De Velde A, et al. The role of interleukin-17 and interleukin-23 inhibitors in the development, progression, and recurrence of cancer: a systematic review. JAAD Int. 2024;17:71-79. doi:10.1016/j.jdin.2024.06.006
- Navarro-Compán V, Puig L, Vidal S, et al. The paradigm of IL-23-independent production of IL-17F and IL-17A and their role in chronic inflammatory diseases. Front Immunol. 2023;14:1191782. doi:10.3389/fimmu.2023.1191782
- Bencardino S, Bernardi F, Allocca M, et al. Advanced therapies for inflammatory bowel disease and risk of skin cancer: what’s new? Cancers (Basel). 2025;17:1710. doi:10.3390/cancers17101710
- Esse S, Mason KJ, Green AC, et al. Melanoma risk in patients treated with biologic therapy for common inflammatory diseases: a systematic review and meta-analysis. JAMA Dermatol. 2020;156:787-794. doi:10.1001/jamadermatol.2020.1300
- Solomon DH, Mercer E, Kavanaugh A. Observational studies on the risk of cancer associated with tumor necrosis factor inhibitors in rheumatoid arthritis: a review of their methodologies and results. Arthritis Rheum. 2012;64:21-32. doi:10.1002/art.30653
Malignancy Risk Among Psoriasis Patients Treated With Interleukin Inhibitors: A Retrospective Matched-Cohort Study
Malignancy Risk Among Psoriasis Patients Treated With Interleukin Inhibitors: A Retrospective Matched-Cohort Study
Practice Points
- Interleukin (IL) inhibitor therapy for psoriasis was associated with reduced odds of multiple malignancies in a large matched-cohort analysis.
- Potential mechanisms for reduced cancer risk include inhibition of tumor-promoting inflammation and restoration of antitumor immune surveillance, although further mechanistic and longitudinal studies are needed.
- These findings provide real-world evidence supporting the long-term malignancy safety of IL inhibitors, which may reassure clinicians and patients considering these agents for chronic disease management.
A Simple Alternative for Intralesional Cryosurgery of Keloids and Hypertrophic Scars Using a Disposable Infusion Set
A Simple Alternative for Intralesional Cryosurgery of Keloids and Hypertrophic Scars Using a Disposable Infusion Set
Practice Gap
Intralesional cryosurgery is a highly effective treatment for dermatologic conditions, notably keloids and hypertrophic scars.1 Conventional methods typically use specialized double-lumen intralesional probes or Luer lock adapters connected to hypodermic or lumbar puncture needles, allowing cryogen to flow internally to cool the probe or needle and treat the lesion via conduction.2 However, specialized intralesional probes are expensive and often are difficult to obtain. Furthermore, Luer lock adapters with needles directly attached to the handle unit can be ergonomically challenging, as the procedure requires simultaneous maintenance of a perpendicular handheld position, precise needle passage through the exact center of the lesion, and protection of the surrounding perilesional healthy skin from cold injury. Consequently, these limitations restrict widespread adoption, necessitating simpler, more accessible, and cost-effective alternatives. Herein, we present a novel, practical, and economical cryogen delivery method that adapts a disposable infusion set to a standard cryospray nozzle.
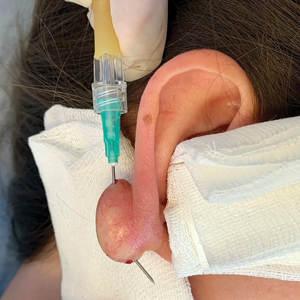
The Technique
This technique involves detaching the infusion set tubing and securely connecting it to the cryospray nozzle (Figure 1). Brief activation of the cryospray to constrict the nozzle or a small incision in the tubing may be required to ensure a tight fit, which can be secured with medical tape to maintain consistent cryogen flow. Local anesthesia is administered directly into and around the lesion, particularly translesionally for keloids, to avoid unnecessary trauma to the surrounding healthy skin, which could trigger further keloid formation. A needle is inserted through the lesion with the tip extending beyond its distal boundary, ensuring the tip remains outside the lesion during cryogen application to prevent cryoinsufflation. If necessary, gentle bending of the needle helps ensure optimal cryogen distribution within the lesion (Figures 2A and 2B). However, this may slightly reduce flow and extend freezing duration; therefore, bending the needle should be performed cautiously and is specifically recommended for effectively treating lesions on curved anatomic sites (eg, the auricle of the ear) to optimize freezing and protect surrounding tissues.

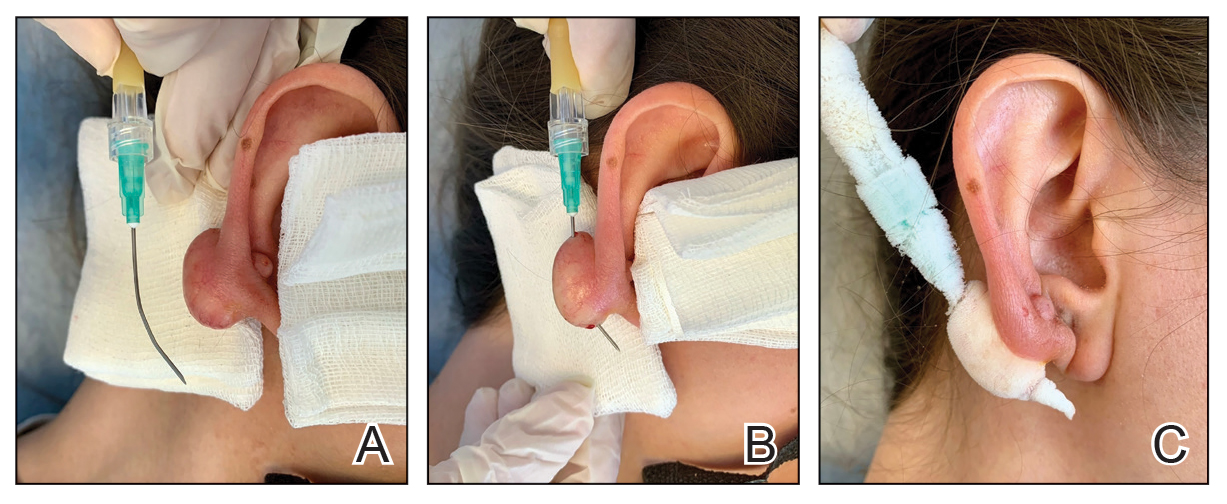
During initial cryogen release, covering the needle tip with gauze prevents aerosolization of biological debris, while placing a wooden tongue depressor between the needle tip and the patient’s skin prevents inadvertent cold injury. After cryogen flow is initiated, the lesion begins to freeze at both the needle entry and exit points, forming what is referred to as ice balls. Over time, typically within several seconds to a few minutes depending on lesion size and tissue characteristics, these ice balls merge centrally, forming a single ice ball encompassing the entire lesion (Figure 2C). Cryogen flow should be maintained during a single application until the unified ice ball appearance is achieved, confirming effective cooling.
Practice Implications
Studies have consistently shown that intralesional cryosurgery is associated with a reduction in the size and symptoms of hypertrophic scars and keloids.1,2 Multimodal treatment approaches, including intralesional methods, are especially valued for their targeted efficacy and minimal adverse effects. Our simplified method offers practical economic advantages, making it highly suitable for broad adoption across diverse clinical settings, particularly those that are resource limited. Clinicians can safely and effectively utilize this technique without specialized or costly equipment, considerably enhancing clinical efficiency and accessibility. The straightforwardness of this method also facilitates the training of medical personnel, enabling rapid integration into clinical practice and the flexibility to treat various lesion types and sizes effectively.
- McGoldrick RB, Theodorakopoulou E, Azzopardi E, et al. Lasers and ancillary treatments for scar management part 2: keloid, hypertrophic, pigmented and acne scars. Scars Burn Heal. 2017;3:1-16. doi:10.1177/2059513116689805
- Gupta S, Kumar B. Intralesional cryosurgery using lumbar puncture and/or hypodermic needles for large, bulky, recalcitrant keloids. Int J Dermatol. 2001;40:349-353. doi:10.1046/j.1365-4362.2001.01117.x
Practice Gap
Intralesional cryosurgery is a highly effective treatment for dermatologic conditions, notably keloids and hypertrophic scars.1 Conventional methods typically use specialized double-lumen intralesional probes or Luer lock adapters connected to hypodermic or lumbar puncture needles, allowing cryogen to flow internally to cool the probe or needle and treat the lesion via conduction.2 However, specialized intralesional probes are expensive and often are difficult to obtain. Furthermore, Luer lock adapters with needles directly attached to the handle unit can be ergonomically challenging, as the procedure requires simultaneous maintenance of a perpendicular handheld position, precise needle passage through the exact center of the lesion, and protection of the surrounding perilesional healthy skin from cold injury. Consequently, these limitations restrict widespread adoption, necessitating simpler, more accessible, and cost-effective alternatives. Herein, we present a novel, practical, and economical cryogen delivery method that adapts a disposable infusion set to a standard cryospray nozzle.
The Technique
This technique involves detaching the infusion set tubing and securely connecting it to the cryospray nozzle (Figure 1). Brief activation of the cryospray to constrict the nozzle or a small incision in the tubing may be required to ensure a tight fit, which can be secured with medical tape to maintain consistent cryogen flow. Local anesthesia is administered directly into and around the lesion, particularly translesionally for keloids, to avoid unnecessary trauma to the surrounding healthy skin, which could trigger further keloid formation. A needle is inserted through the lesion with the tip extending beyond its distal boundary, ensuring the tip remains outside the lesion during cryogen application to prevent cryoinsufflation. If necessary, gentle bending of the needle helps ensure optimal cryogen distribution within the lesion (Figures 2A and 2B). However, this may slightly reduce flow and extend freezing duration; therefore, bending the needle should be performed cautiously and is specifically recommended for effectively treating lesions on curved anatomic sites (eg, the auricle of the ear) to optimize freezing and protect surrounding tissues.


During initial cryogen release, covering the needle tip with gauze prevents aerosolization of biological debris, while placing a wooden tongue depressor between the needle tip and the patient’s skin prevents inadvertent cold injury. After cryogen flow is initiated, the lesion begins to freeze at both the needle entry and exit points, forming what is referred to as ice balls. Over time, typically within several seconds to a few minutes depending on lesion size and tissue characteristics, these ice balls merge centrally, forming a single ice ball encompassing the entire lesion (Figure 2C). Cryogen flow should be maintained during a single application until the unified ice ball appearance is achieved, confirming effective cooling.
Practice Implications
Studies have consistently shown that intralesional cryosurgery is associated with a reduction in the size and symptoms of hypertrophic scars and keloids.1,2 Multimodal treatment approaches, including intralesional methods, are especially valued for their targeted efficacy and minimal adverse effects. Our simplified method offers practical economic advantages, making it highly suitable for broad adoption across diverse clinical settings, particularly those that are resource limited. Clinicians can safely and effectively utilize this technique without specialized or costly equipment, considerably enhancing clinical efficiency and accessibility. The straightforwardness of this method also facilitates the training of medical personnel, enabling rapid integration into clinical practice and the flexibility to treat various lesion types and sizes effectively.
Practice Gap
Intralesional cryosurgery is a highly effective treatment for dermatologic conditions, notably keloids and hypertrophic scars.1 Conventional methods typically use specialized double-lumen intralesional probes or Luer lock adapters connected to hypodermic or lumbar puncture needles, allowing cryogen to flow internally to cool the probe or needle and treat the lesion via conduction.2 However, specialized intralesional probes are expensive and often are difficult to obtain. Furthermore, Luer lock adapters with needles directly attached to the handle unit can be ergonomically challenging, as the procedure requires simultaneous maintenance of a perpendicular handheld position, precise needle passage through the exact center of the lesion, and protection of the surrounding perilesional healthy skin from cold injury. Consequently, these limitations restrict widespread adoption, necessitating simpler, more accessible, and cost-effective alternatives. Herein, we present a novel, practical, and economical cryogen delivery method that adapts a disposable infusion set to a standard cryospray nozzle.
The Technique
This technique involves detaching the infusion set tubing and securely connecting it to the cryospray nozzle (Figure 1). Brief activation of the cryospray to constrict the nozzle or a small incision in the tubing may be required to ensure a tight fit, which can be secured with medical tape to maintain consistent cryogen flow. Local anesthesia is administered directly into and around the lesion, particularly translesionally for keloids, to avoid unnecessary trauma to the surrounding healthy skin, which could trigger further keloid formation. A needle is inserted through the lesion with the tip extending beyond its distal boundary, ensuring the tip remains outside the lesion during cryogen application to prevent cryoinsufflation. If necessary, gentle bending of the needle helps ensure optimal cryogen distribution within the lesion (Figures 2A and 2B). However, this may slightly reduce flow and extend freezing duration; therefore, bending the needle should be performed cautiously and is specifically recommended for effectively treating lesions on curved anatomic sites (eg, the auricle of the ear) to optimize freezing and protect surrounding tissues.


During initial cryogen release, covering the needle tip with gauze prevents aerosolization of biological debris, while placing a wooden tongue depressor between the needle tip and the patient’s skin prevents inadvertent cold injury. After cryogen flow is initiated, the lesion begins to freeze at both the needle entry and exit points, forming what is referred to as ice balls. Over time, typically within several seconds to a few minutes depending on lesion size and tissue characteristics, these ice balls merge centrally, forming a single ice ball encompassing the entire lesion (Figure 2C). Cryogen flow should be maintained during a single application until the unified ice ball appearance is achieved, confirming effective cooling.
Practice Implications
Studies have consistently shown that intralesional cryosurgery is associated with a reduction in the size and symptoms of hypertrophic scars and keloids.1,2 Multimodal treatment approaches, including intralesional methods, are especially valued for their targeted efficacy and minimal adverse effects. Our simplified method offers practical economic advantages, making it highly suitable for broad adoption across diverse clinical settings, particularly those that are resource limited. Clinicians can safely and effectively utilize this technique without specialized or costly equipment, considerably enhancing clinical efficiency and accessibility. The straightforwardness of this method also facilitates the training of medical personnel, enabling rapid integration into clinical practice and the flexibility to treat various lesion types and sizes effectively.
- McGoldrick RB, Theodorakopoulou E, Azzopardi E, et al. Lasers and ancillary treatments for scar management part 2: keloid, hypertrophic, pigmented and acne scars. Scars Burn Heal. 2017;3:1-16. doi:10.1177/2059513116689805
- Gupta S, Kumar B. Intralesional cryosurgery using lumbar puncture and/or hypodermic needles for large, bulky, recalcitrant keloids. Int J Dermatol. 2001;40:349-353. doi:10.1046/j.1365-4362.2001.01117.x
- McGoldrick RB, Theodorakopoulou E, Azzopardi E, et al. Lasers and ancillary treatments for scar management part 2: keloid, hypertrophic, pigmented and acne scars. Scars Burn Heal. 2017;3:1-16. doi:10.1177/2059513116689805
- Gupta S, Kumar B. Intralesional cryosurgery using lumbar puncture and/or hypodermic needles for large, bulky, recalcitrant keloids. Int J Dermatol. 2001;40:349-353. doi:10.1046/j.1365-4362.2001.01117.x
A Simple Alternative for Intralesional Cryosurgery of Keloids and Hypertrophic Scars Using a Disposable Infusion Set
A Simple Alternative for Intralesional Cryosurgery of Keloids and Hypertrophic Scars Using a Disposable Infusion Set
Thick Yellow Plaques on the Eyelids
Thick Yellow Plaques on the Eyelids
THE DIAGNOSIS: Adult-Onset Asthma With Periocular Xanthogranuloma
In the context of pre-existing sinonasal disease and features consistent with adult-onset asthma, the constellation of clinical findings including linear periorbital yellow-orange plaques, imaging demonstrating extension of xanthogranulomatous lesions into the orbital fat, histopathologic features, and serologic abnormalities including elevated IgG4 levels supported a diagnosis of adult-onset asthma with periocular xanthogranuloma (AAPOX).
Adult-onset xanthogranuloma is a non–Langerhans cell histiocytosis (historically classified as type II) within the group of adult orbital xanthogranulomatous diseases resulting from infiltration and proliferation of histiocytes in the orbital and ocular adnexal structures and eyelids. Adult orbital xanthogranulomatous diseases are classified as 4 distinct conditions: adult-onset xanthogranuloma, AAPOX, Erdheim-Chester disease, and necrobiotic xanthogranuloma (NXG). Erdheim-Chester disease is the most severe among this group and often is fatal due to infiltration of the xanthogranulomas into multiple organ systems and tissues, including the long bones, heart, lungs, and retroperitoneum. Neurologic symptoms such as incoordination can occur. Adult-onset xanthogranuloma manifests as an isolated cutaneous lesion without systemic involvement. This entity often is self-limited and does not require aggressive treatment. Adult-onset asthma with periocular xanthogranuloma affects more males than females.1 Clinically, it manifests as bilateral yellow-orange, thickened, indurated eyelid plaques that can extend to the extraocular muscles or lacrimal glands. As the name suggests, this entity is associated with adult-onset asthma or rhinosinusitis as well as lymphadenopathy and extension into the orbital fat.1
When patients present with periorbital lesions and optic symptoms such as visual disturbances, tearing, and/ or a foreign body sensation in the eyes, a work-up should be performed to rule out infiltration of orbital adnexal structures and other organ systems, as AAPOX can be associated with IgG4-related disease. Histologically, adult xanthogranulomatous diseases are characterized by sheets of foamy histiocytes accompanied by variable numbers of lymphoid aggregates, plasma cells, and Touton giant cells. These infiltrating xanthoma cells appear as a garland or wreathlike nuclei surrounded by foamy cytoplasm. Oil-red O staining of frozen sections confirms the lipid content of the xanthoma cells.2 Immunohistochemically, the foamy histiocytes are strongly positive for CD68, CD163, and factor XIIIa but usually are negative for S100, CD1a and Birbeck granules.3 More distinctively, AAPOX is associated with prominent lymphoid aggregates containing reactive germinal centers.1
Given that AAPOX is a systemic multiorgan disease, local therapies such as surgical debulking or intralesional corticosteroids generally are insufficient to address the underlying pathology and therefore necessitate systemic, often multimodal, treatment within a multidisciplinary framework.1,4 Systemic corticosteroids remain first-line therapy, with steroid-sparing agents (eg, methotrexate, azathioprine, cyclophosphamide) used in refractory cases or to reduce steroid dependence.4,5 Rituximab has demonstrated efficacy in AAPOX, further highlighting the association between AAPOX and IgG4-related disease.5 Inebilizumab, a B-cell–depleting monoclonal antibody targeting CD19 and approved for the treatment of IgG4- related disease, represents a theoretically promising therapeutic option; however, additional studies are needed to establish its efficacy and safety in AAPOX.5
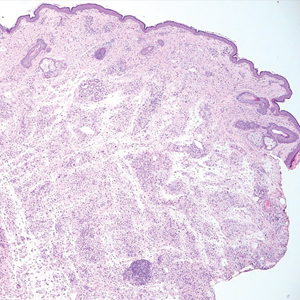
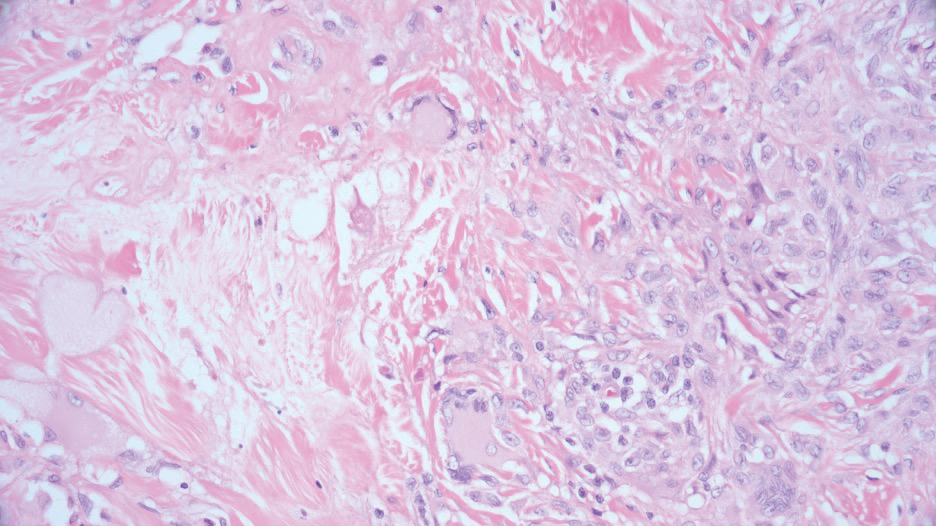
Necrobiotic xanthogranuloma is another xanthogranulomatous disorder that manifests as yellow-orange papules or nodules that gradually form infiltrative plaques. Scarring and ulceration can occur in 40% to 50% of patients.6 The most common site of involvement is the periorbital area, affecting 80% of cases, often resulting in ophthalmologic complications.6 Necrobiotic xanthogranuloma lesions also can involve the trunk, arms, and legs. Extracutaneous sites include the lungs, myocardium, larynx, pharynx, skeletal muscles, kidneys, ovaries, and intestines. The prognosis of NXG is poor due to associated hematologic malignancies such as multiple myeloma and lymphoma. About 80% of patients have a serum monoclonal gammopathy.6 Histologically, NXG shows features overlapping with other xanthogranulomatous disorders, such as foamy histiocytes, multinucleated giant cells, Touton giant cells, and nodular lymphocytic aggregates6,7; however, broad zones of necrobiosis are a distinct histologic finding in NXG that helps differentiate it from other xanthogranulomatous disorders (Figure 1).6
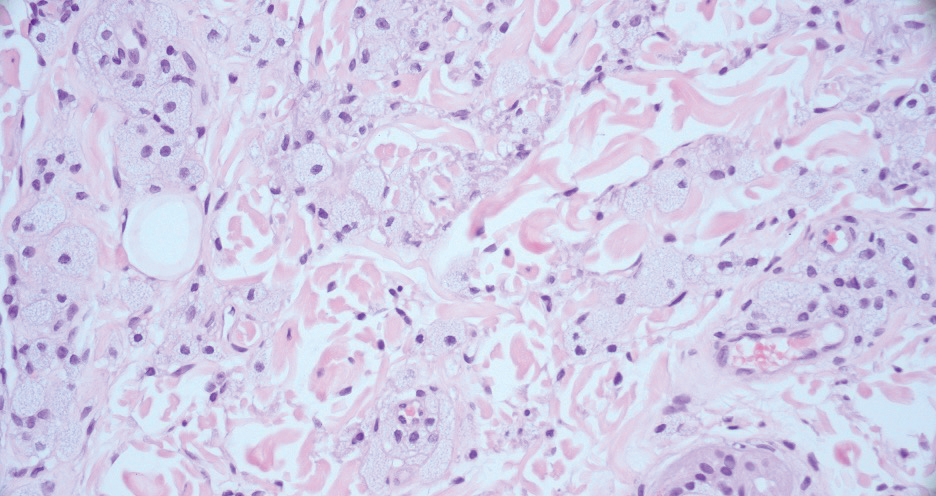
Xanthelasma manifests as yellow plaques on the medial upper and lower eyelids and lateral canthi resulting from accumulation of cholesterol-rich material in the skin, soft tissue, and sometimes the tendons. Fifty percent of patients have a primary or secondary lipid disorder such as familial dyslipidemia, thyroid disease, diabetes mellitus, or primary biliary cholangitis.8 Histologically, xanthelasmas demonstrate lipid-laden foamy histiocytes in the superficial dermis (Figure 2).8 Despite some clinical overlap in our case, the depth of orbital involvement and supportive systemic and histopathologic findings (including Touton giant cells) supported the diagnosis of AAPOX rather than xanthelasma.
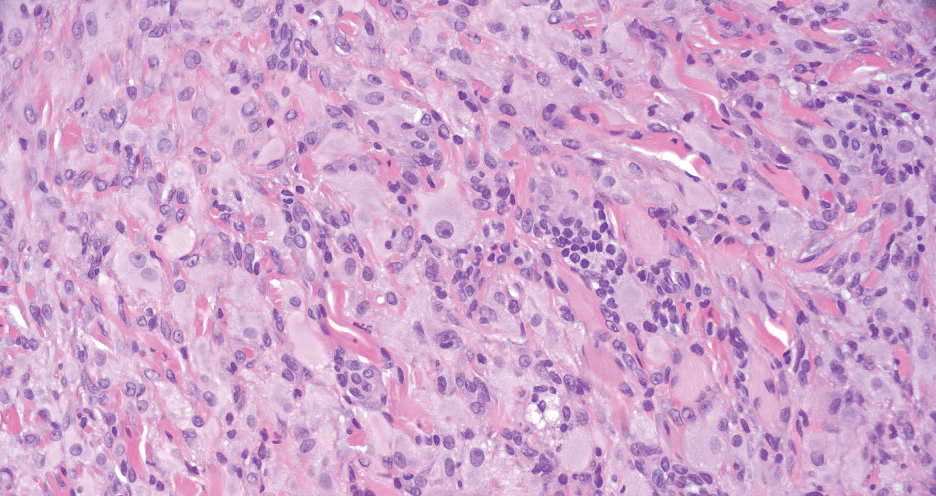
Juvenile xanthogranuloma manifests as solitary to multiple firm, yellow-orange papules or nodules on the face, neck, and upper torso. The lesions develop in early childhood, with 75% of lesions appearing in the first year of life, but rarely it may develop in adulthood.9 The most common extracutaneous manifestation involves ocular structures, most frequently the iris, followed by the lungs. Cutaneous lesions usually are asymptomatic and involute over the span of a few years. Ocular lesions can result in blindness, and juvenile xanthogranuloma also has been associated with neurofibromatosis type 1 and juvenile chronic myelogenous leukemia.9 The histopathology of juvenile xanthogranuloma often will show a dense histiocytic infiltrate in the dermis with blunting of the overlying rete ridges admixed with lymphocytes, plasma cells, and eosinophils. In the more mature phase, foam cells, foreign body giant cells, and Touton giant cells predominate. Touton giant cells have a garlandlike appearance (Figure 3).9
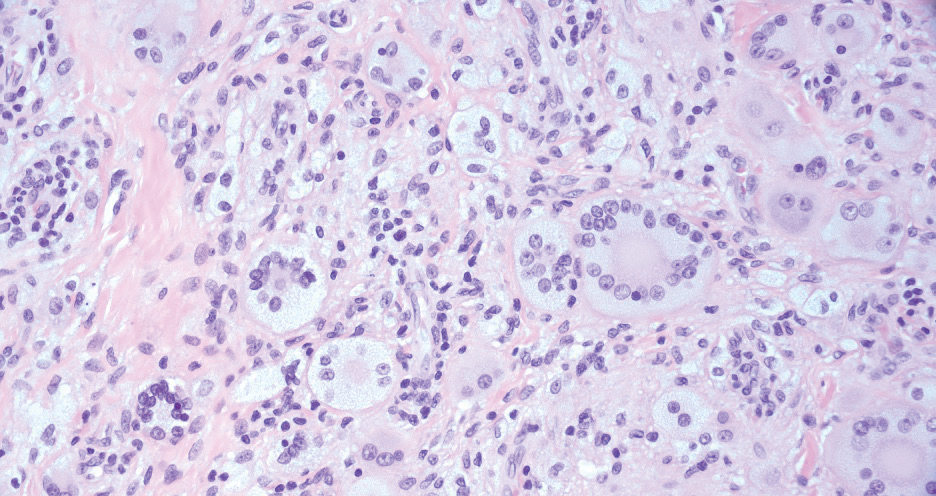
Reticulohistiocytoma (or solitary epithelioid histiocytoma) and multicentric reticulohistiocytosis are rare histiocytic proliferations. Multicentric reticulohistiocytosis refers to a systemic disease with arthropathy and multiple cutaneous histiocytic lesions located on acral sites and the face. Solitary reticulohistiocytoma manifests as papules or nodules found in many body locations, such as the trunk, arms, and legs. The lesions are uncommon on the face, which almost always is involved in multicentric reticulohistiocytosis. Solitary reticulohistiocytomas tend not to recur once excised and do not demonstrate systemic involvement. Histologically, the lesions demonstrate large eosinophilic epithelioid histiocytes with abundant glassy cytoplasm (Figure 4). Some of the epithelioid histiocytes are multinucleated, and immunophenotyping will show positivity for lysozyme, CD68, and CD163.10
- Kerstetter J, Wang J. Adult orbital xanthogranulomatous disease: a review with emphasis on etiology, systemic associations, diagnostic tools, and treatment. Dermatol Clin. 2015;33:457-463. doi:10.1016 /j.det.2015.03.010
- Mandic` JJ, Bakula M, Šklebar LK, et al. Histiocytosis and adult-onset orbital xanthogranuloma in 2023: a review of the literature and mini case series. Int Ophthalmol. 2024;44:301. doi:10.1007/s10792-024-03181-y
- Campochiaro C, Tomelleri A, Cavalli G, et al. Erdheim-Chester disease. Eur J Intern Med. 2015;26:223-229. doi: 10.1016/j.ejim.2015.03.004
- Detiger SE, Hötte GJ, Verdijk RM, et al. Adult orbital xanthogranuloma: long-term follow-up of treated cases. Eye (Lond). 2023;37:2475-2481. doi: 10.1038/s41433-022-02357-z
- Stone JH, Khosroshahi A, Zhang W, et al. Inebilizumab for treatment of IgG4-related disease. N Engl J Med. 2025;392:1168-1177. doi:10.1056 /NEJMoa2409712
- Wood AJ, Wagner MVU, Abbott JJ, et al. Necrobiotic xanthogranuloma: a review of 17 cases with emphasis on clinical and pathologic correlation. Arch Dermatol. 2009;145:279–284. doi:10.1001 /archdermatol.2008.583
- Schadt C, Jacobsen E. Necrobiotic xanthogranuloma. In: Connor RF, ed. UpToDate. Wolters Kluwer.
- Al Aboud AM, Shah SS, Blair K, et al. Xanthelasma palpebrarum. StatPearls [Internet]. StatPearls Publishing; 2024. Updated March 1, 2024. Accessed February 10, 2026. https://www.ncbi.nlm .nih.gov/books/NBK531501/
- Collie JS, Harper CD, Fillman EP. Juvenile xanthogranuloma. StatPearls [Internet]. StatPearls Publishing; 2024. Updated August 8, 2023. Accessed February 10, 2026. https://www.ncbi.nlm.nih .gov/books/NBK526103/
- Miettinen M, Fetsch JF. Reticulohistiocytoma (solitary epithelioid histiocytoma): a clinicopathologic and immunohistochemical study of 44 cases. Am J Surg Pathol. 2006;30:521-528. doi:10.1097/00000478 -200604000-00014
THE DIAGNOSIS: Adult-Onset Asthma With Periocular Xanthogranuloma
In the context of pre-existing sinonasal disease and features consistent with adult-onset asthma, the constellation of clinical findings including linear periorbital yellow-orange plaques, imaging demonstrating extension of xanthogranulomatous lesions into the orbital fat, histopathologic features, and serologic abnormalities including elevated IgG4 levels supported a diagnosis of adult-onset asthma with periocular xanthogranuloma (AAPOX).
Adult-onset xanthogranuloma is a non–Langerhans cell histiocytosis (historically classified as type II) within the group of adult orbital xanthogranulomatous diseases resulting from infiltration and proliferation of histiocytes in the orbital and ocular adnexal structures and eyelids. Adult orbital xanthogranulomatous diseases are classified as 4 distinct conditions: adult-onset xanthogranuloma, AAPOX, Erdheim-Chester disease, and necrobiotic xanthogranuloma (NXG). Erdheim-Chester disease is the most severe among this group and often is fatal due to infiltration of the xanthogranulomas into multiple organ systems and tissues, including the long bones, heart, lungs, and retroperitoneum. Neurologic symptoms such as incoordination can occur. Adult-onset xanthogranuloma manifests as an isolated cutaneous lesion without systemic involvement. This entity often is self-limited and does not require aggressive treatment. Adult-onset asthma with periocular xanthogranuloma affects more males than females.1 Clinically, it manifests as bilateral yellow-orange, thickened, indurated eyelid plaques that can extend to the extraocular muscles or lacrimal glands. As the name suggests, this entity is associated with adult-onset asthma or rhinosinusitis as well as lymphadenopathy and extension into the orbital fat.1
When patients present with periorbital lesions and optic symptoms such as visual disturbances, tearing, and/ or a foreign body sensation in the eyes, a work-up should be performed to rule out infiltration of orbital adnexal structures and other organ systems, as AAPOX can be associated with IgG4-related disease. Histologically, adult xanthogranulomatous diseases are characterized by sheets of foamy histiocytes accompanied by variable numbers of lymphoid aggregates, plasma cells, and Touton giant cells. These infiltrating xanthoma cells appear as a garland or wreathlike nuclei surrounded by foamy cytoplasm. Oil-red O staining of frozen sections confirms the lipid content of the xanthoma cells.2 Immunohistochemically, the foamy histiocytes are strongly positive for CD68, CD163, and factor XIIIa but usually are negative for S100, CD1a and Birbeck granules.3 More distinctively, AAPOX is associated with prominent lymphoid aggregates containing reactive germinal centers.1
Given that AAPOX is a systemic multiorgan disease, local therapies such as surgical debulking or intralesional corticosteroids generally are insufficient to address the underlying pathology and therefore necessitate systemic, often multimodal, treatment within a multidisciplinary framework.1,4 Systemic corticosteroids remain first-line therapy, with steroid-sparing agents (eg, methotrexate, azathioprine, cyclophosphamide) used in refractory cases or to reduce steroid dependence.4,5 Rituximab has demonstrated efficacy in AAPOX, further highlighting the association between AAPOX and IgG4-related disease.5 Inebilizumab, a B-cell–depleting monoclonal antibody targeting CD19 and approved for the treatment of IgG4- related disease, represents a theoretically promising therapeutic option; however, additional studies are needed to establish its efficacy and safety in AAPOX.5
Necrobiotic xanthogranuloma is another xanthogranulomatous disorder that manifests as yellow-orange papules or nodules that gradually form infiltrative plaques. Scarring and ulceration can occur in 40% to 50% of patients.6 The most common site of involvement is the periorbital area, affecting 80% of cases, often resulting in ophthalmologic complications.6 Necrobiotic xanthogranuloma lesions also can involve the trunk, arms, and legs. Extracutaneous sites include the lungs, myocardium, larynx, pharynx, skeletal muscles, kidneys, ovaries, and intestines. The prognosis of NXG is poor due to associated hematologic malignancies such as multiple myeloma and lymphoma. About 80% of patients have a serum monoclonal gammopathy.6 Histologically, NXG shows features overlapping with other xanthogranulomatous disorders, such as foamy histiocytes, multinucleated giant cells, Touton giant cells, and nodular lymphocytic aggregates6,7; however, broad zones of necrobiosis are a distinct histologic finding in NXG that helps differentiate it from other xanthogranulomatous disorders (Figure 1).6

Xanthelasma manifests as yellow plaques on the medial upper and lower eyelids and lateral canthi resulting from accumulation of cholesterol-rich material in the skin, soft tissue, and sometimes the tendons. Fifty percent of patients have a primary or secondary lipid disorder such as familial dyslipidemia, thyroid disease, diabetes mellitus, or primary biliary cholangitis.8 Histologically, xanthelasmas demonstrate lipid-laden foamy histiocytes in the superficial dermis (Figure 2).8 Despite some clinical overlap in our case, the depth of orbital involvement and supportive systemic and histopathologic findings (including Touton giant cells) supported the diagnosis of AAPOX rather than xanthelasma.

Juvenile xanthogranuloma manifests as solitary to multiple firm, yellow-orange papules or nodules on the face, neck, and upper torso. The lesions develop in early childhood, with 75% of lesions appearing in the first year of life, but rarely it may develop in adulthood.9 The most common extracutaneous manifestation involves ocular structures, most frequently the iris, followed by the lungs. Cutaneous lesions usually are asymptomatic and involute over the span of a few years. Ocular lesions can result in blindness, and juvenile xanthogranuloma also has been associated with neurofibromatosis type 1 and juvenile chronic myelogenous leukemia.9 The histopathology of juvenile xanthogranuloma often will show a dense histiocytic infiltrate in the dermis with blunting of the overlying rete ridges admixed with lymphocytes, plasma cells, and eosinophils. In the more mature phase, foam cells, foreign body giant cells, and Touton giant cells predominate. Touton giant cells have a garlandlike appearance (Figure 3).9

Reticulohistiocytoma (or solitary epithelioid histiocytoma) and multicentric reticulohistiocytosis are rare histiocytic proliferations. Multicentric reticulohistiocytosis refers to a systemic disease with arthropathy and multiple cutaneous histiocytic lesions located on acral sites and the face. Solitary reticulohistiocytoma manifests as papules or nodules found in many body locations, such as the trunk, arms, and legs. The lesions are uncommon on the face, which almost always is involved in multicentric reticulohistiocytosis. Solitary reticulohistiocytomas tend not to recur once excised and do not demonstrate systemic involvement. Histologically, the lesions demonstrate large eosinophilic epithelioid histiocytes with abundant glassy cytoplasm (Figure 4). Some of the epithelioid histiocytes are multinucleated, and immunophenotyping will show positivity for lysozyme, CD68, and CD163.10

THE DIAGNOSIS: Adult-Onset Asthma With Periocular Xanthogranuloma
In the context of pre-existing sinonasal disease and features consistent with adult-onset asthma, the constellation of clinical findings including linear periorbital yellow-orange plaques, imaging demonstrating extension of xanthogranulomatous lesions into the orbital fat, histopathologic features, and serologic abnormalities including elevated IgG4 levels supported a diagnosis of adult-onset asthma with periocular xanthogranuloma (AAPOX).
Adult-onset xanthogranuloma is a non–Langerhans cell histiocytosis (historically classified as type II) within the group of adult orbital xanthogranulomatous diseases resulting from infiltration and proliferation of histiocytes in the orbital and ocular adnexal structures and eyelids. Adult orbital xanthogranulomatous diseases are classified as 4 distinct conditions: adult-onset xanthogranuloma, AAPOX, Erdheim-Chester disease, and necrobiotic xanthogranuloma (NXG). Erdheim-Chester disease is the most severe among this group and often is fatal due to infiltration of the xanthogranulomas into multiple organ systems and tissues, including the long bones, heart, lungs, and retroperitoneum. Neurologic symptoms such as incoordination can occur. Adult-onset xanthogranuloma manifests as an isolated cutaneous lesion without systemic involvement. This entity often is self-limited and does not require aggressive treatment. Adult-onset asthma with periocular xanthogranuloma affects more males than females.1 Clinically, it manifests as bilateral yellow-orange, thickened, indurated eyelid plaques that can extend to the extraocular muscles or lacrimal glands. As the name suggests, this entity is associated with adult-onset asthma or rhinosinusitis as well as lymphadenopathy and extension into the orbital fat.1
When patients present with periorbital lesions and optic symptoms such as visual disturbances, tearing, and/ or a foreign body sensation in the eyes, a work-up should be performed to rule out infiltration of orbital adnexal structures and other organ systems, as AAPOX can be associated with IgG4-related disease. Histologically, adult xanthogranulomatous diseases are characterized by sheets of foamy histiocytes accompanied by variable numbers of lymphoid aggregates, plasma cells, and Touton giant cells. These infiltrating xanthoma cells appear as a garland or wreathlike nuclei surrounded by foamy cytoplasm. Oil-red O staining of frozen sections confirms the lipid content of the xanthoma cells.2 Immunohistochemically, the foamy histiocytes are strongly positive for CD68, CD163, and factor XIIIa but usually are negative for S100, CD1a and Birbeck granules.3 More distinctively, AAPOX is associated with prominent lymphoid aggregates containing reactive germinal centers.1
Given that AAPOX is a systemic multiorgan disease, local therapies such as surgical debulking or intralesional corticosteroids generally are insufficient to address the underlying pathology and therefore necessitate systemic, often multimodal, treatment within a multidisciplinary framework.1,4 Systemic corticosteroids remain first-line therapy, with steroid-sparing agents (eg, methotrexate, azathioprine, cyclophosphamide) used in refractory cases or to reduce steroid dependence.4,5 Rituximab has demonstrated efficacy in AAPOX, further highlighting the association between AAPOX and IgG4-related disease.5 Inebilizumab, a B-cell–depleting monoclonal antibody targeting CD19 and approved for the treatment of IgG4- related disease, represents a theoretically promising therapeutic option; however, additional studies are needed to establish its efficacy and safety in AAPOX.5
Necrobiotic xanthogranuloma is another xanthogranulomatous disorder that manifests as yellow-orange papules or nodules that gradually form infiltrative plaques. Scarring and ulceration can occur in 40% to 50% of patients.6 The most common site of involvement is the periorbital area, affecting 80% of cases, often resulting in ophthalmologic complications.6 Necrobiotic xanthogranuloma lesions also can involve the trunk, arms, and legs. Extracutaneous sites include the lungs, myocardium, larynx, pharynx, skeletal muscles, kidneys, ovaries, and intestines. The prognosis of NXG is poor due to associated hematologic malignancies such as multiple myeloma and lymphoma. About 80% of patients have a serum monoclonal gammopathy.6 Histologically, NXG shows features overlapping with other xanthogranulomatous disorders, such as foamy histiocytes, multinucleated giant cells, Touton giant cells, and nodular lymphocytic aggregates6,7; however, broad zones of necrobiosis are a distinct histologic finding in NXG that helps differentiate it from other xanthogranulomatous disorders (Figure 1).6

Xanthelasma manifests as yellow plaques on the medial upper and lower eyelids and lateral canthi resulting from accumulation of cholesterol-rich material in the skin, soft tissue, and sometimes the tendons. Fifty percent of patients have a primary or secondary lipid disorder such as familial dyslipidemia, thyroid disease, diabetes mellitus, or primary biliary cholangitis.8 Histologically, xanthelasmas demonstrate lipid-laden foamy histiocytes in the superficial dermis (Figure 2).8 Despite some clinical overlap in our case, the depth of orbital involvement and supportive systemic and histopathologic findings (including Touton giant cells) supported the diagnosis of AAPOX rather than xanthelasma.

Juvenile xanthogranuloma manifests as solitary to multiple firm, yellow-orange papules or nodules on the face, neck, and upper torso. The lesions develop in early childhood, with 75% of lesions appearing in the first year of life, but rarely it may develop in adulthood.9 The most common extracutaneous manifestation involves ocular structures, most frequently the iris, followed by the lungs. Cutaneous lesions usually are asymptomatic and involute over the span of a few years. Ocular lesions can result in blindness, and juvenile xanthogranuloma also has been associated with neurofibromatosis type 1 and juvenile chronic myelogenous leukemia.9 The histopathology of juvenile xanthogranuloma often will show a dense histiocytic infiltrate in the dermis with blunting of the overlying rete ridges admixed with lymphocytes, plasma cells, and eosinophils. In the more mature phase, foam cells, foreign body giant cells, and Touton giant cells predominate. Touton giant cells have a garlandlike appearance (Figure 3).9

Reticulohistiocytoma (or solitary epithelioid histiocytoma) and multicentric reticulohistiocytosis are rare histiocytic proliferations. Multicentric reticulohistiocytosis refers to a systemic disease with arthropathy and multiple cutaneous histiocytic lesions located on acral sites and the face. Solitary reticulohistiocytoma manifests as papules or nodules found in many body locations, such as the trunk, arms, and legs. The lesions are uncommon on the face, which almost always is involved in multicentric reticulohistiocytosis. Solitary reticulohistiocytomas tend not to recur once excised and do not demonstrate systemic involvement. Histologically, the lesions demonstrate large eosinophilic epithelioid histiocytes with abundant glassy cytoplasm (Figure 4). Some of the epithelioid histiocytes are multinucleated, and immunophenotyping will show positivity for lysozyme, CD68, and CD163.10

- Kerstetter J, Wang J. Adult orbital xanthogranulomatous disease: a review with emphasis on etiology, systemic associations, diagnostic tools, and treatment. Dermatol Clin. 2015;33:457-463. doi:10.1016 /j.det.2015.03.010
- Mandic` JJ, Bakula M, Šklebar LK, et al. Histiocytosis and adult-onset orbital xanthogranuloma in 2023: a review of the literature and mini case series. Int Ophthalmol. 2024;44:301. doi:10.1007/s10792-024-03181-y
- Campochiaro C, Tomelleri A, Cavalli G, et al. Erdheim-Chester disease. Eur J Intern Med. 2015;26:223-229. doi: 10.1016/j.ejim.2015.03.004
- Detiger SE, Hötte GJ, Verdijk RM, et al. Adult orbital xanthogranuloma: long-term follow-up of treated cases. Eye (Lond). 2023;37:2475-2481. doi: 10.1038/s41433-022-02357-z
- Stone JH, Khosroshahi A, Zhang W, et al. Inebilizumab for treatment of IgG4-related disease. N Engl J Med. 2025;392:1168-1177. doi:10.1056 /NEJMoa2409712
- Wood AJ, Wagner MVU, Abbott JJ, et al. Necrobiotic xanthogranuloma: a review of 17 cases with emphasis on clinical and pathologic correlation. Arch Dermatol. 2009;145:279–284. doi:10.1001 /archdermatol.2008.583
- Schadt C, Jacobsen E. Necrobiotic xanthogranuloma. In: Connor RF, ed. UpToDate. Wolters Kluwer.
- Al Aboud AM, Shah SS, Blair K, et al. Xanthelasma palpebrarum. StatPearls [Internet]. StatPearls Publishing; 2024. Updated March 1, 2024. Accessed February 10, 2026. https://www.ncbi.nlm .nih.gov/books/NBK531501/
- Collie JS, Harper CD, Fillman EP. Juvenile xanthogranuloma. StatPearls [Internet]. StatPearls Publishing; 2024. Updated August 8, 2023. Accessed February 10, 2026. https://www.ncbi.nlm.nih .gov/books/NBK526103/
- Miettinen M, Fetsch JF. Reticulohistiocytoma (solitary epithelioid histiocytoma): a clinicopathologic and immunohistochemical study of 44 cases. Am J Surg Pathol. 2006;30:521-528. doi:10.1097/00000478 -200604000-00014
- Kerstetter J, Wang J. Adult orbital xanthogranulomatous disease: a review with emphasis on etiology, systemic associations, diagnostic tools, and treatment. Dermatol Clin. 2015;33:457-463. doi:10.1016 /j.det.2015.03.010
- Mandic` JJ, Bakula M, Šklebar LK, et al. Histiocytosis and adult-onset orbital xanthogranuloma in 2023: a review of the literature and mini case series. Int Ophthalmol. 2024;44:301. doi:10.1007/s10792-024-03181-y
- Campochiaro C, Tomelleri A, Cavalli G, et al. Erdheim-Chester disease. Eur J Intern Med. 2015;26:223-229. doi: 10.1016/j.ejim.2015.03.004
- Detiger SE, Hötte GJ, Verdijk RM, et al. Adult orbital xanthogranuloma: long-term follow-up of treated cases. Eye (Lond). 2023;37:2475-2481. doi: 10.1038/s41433-022-02357-z
- Stone JH, Khosroshahi A, Zhang W, et al. Inebilizumab for treatment of IgG4-related disease. N Engl J Med. 2025;392:1168-1177. doi:10.1056 /NEJMoa2409712
- Wood AJ, Wagner MVU, Abbott JJ, et al. Necrobiotic xanthogranuloma: a review of 17 cases with emphasis on clinical and pathologic correlation. Arch Dermatol. 2009;145:279–284. doi:10.1001 /archdermatol.2008.583
- Schadt C, Jacobsen E. Necrobiotic xanthogranuloma. In: Connor RF, ed. UpToDate. Wolters Kluwer.
- Al Aboud AM, Shah SS, Blair K, et al. Xanthelasma palpebrarum. StatPearls [Internet]. StatPearls Publishing; 2024. Updated March 1, 2024. Accessed February 10, 2026. https://www.ncbi.nlm .nih.gov/books/NBK531501/
- Collie JS, Harper CD, Fillman EP. Juvenile xanthogranuloma. StatPearls [Internet]. StatPearls Publishing; 2024. Updated August 8, 2023. Accessed February 10, 2026. https://www.ncbi.nlm.nih .gov/books/NBK526103/
- Miettinen M, Fetsch JF. Reticulohistiocytoma (solitary epithelioid histiocytoma): a clinicopathologic and immunohistochemical study of 44 cases. Am J Surg Pathol. 2006;30:521-528. doi:10.1097/00000478 -200604000-00014
Thick Yellow Plaques on the Eyelids
Thick Yellow Plaques on the Eyelids
A 54-year-old man presented to the dermatology department for evaluation of enlarging lesions on the eyelids of 18 months’ duration causing tearing, dryness, and heaviness. The patient’s medical history was positive for hyperlipidemia, chronic rhinosinusitis, and mild asthma diagnosed in adulthood. A review of systems was negative for bone pain, polyuria, polydipsia, dysuria, hematuria, decreased coordination, chest pain, palpitations, abdominal pain, easy bruising, and jaundice. Laboratory testing revealed elevated IgG4 levels and a slight increase in gamma globulins on serum protein electrophoresis, with no evidence of paraproteinemia. Liver and kidney function test results were within normal limits. Magnetic resonance imaging of the orbits revealed bilateral superolateral intraorbital masses within the extraconal fat that were displacing the superior and lateral rectus muscles. No intraconal masses were identified. Due to bilateral eyelid ptosis, the patient underwent debulking by oculoplastic surgery, and the tissue was submitted for histologic examination.
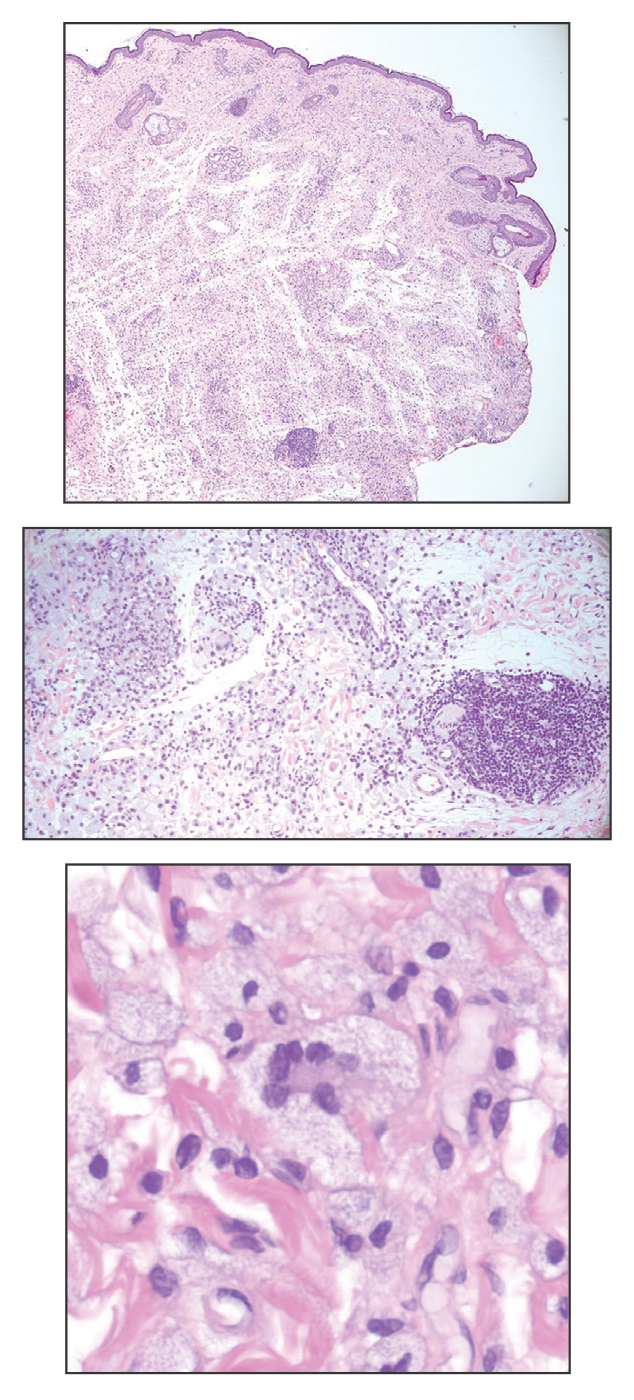
Scarring Alopecia in Skin of Color: Practical Guidance From Dr. Amy J. McMichael
Scarring Alopecia in Skin of Color: Practical Guidance From Dr. Amy J. McMichael
How do you distinguish active cicatricial alopecia from chronic nonprogressive hair loss in patients with skin of color, and which clinical or trichoscopic features suggest ongoing inflammation requiring treatment?
DR. McMICHAEL: Typically, I use trichoscopy to help determine if there are fine or vellus hairs in the affected area. Often, if there are vellus hairs, that means there is the potential to lengthen growth time and have thicker longer hairs in the area, even in a chronic nonprogressive hair loss state. If there is clinical or trichoscopic erythema (suggesting inflammation), that is a sure sign of active scarring. Patient-reported symptoms of itch, stinging, or pain are a sign of active scarring as well. Finally, even if none of the above are true, a biopsy can help determine if there is active inflammation, as can increasing size of the affected area on the scalp upon return visit to the clinic.
What is your initial diagnostic workup for suspected scarring alopecia in patients with skin of color, including key history, trichoscopy, and biopsy approach?
DR. McMICHAEL: In terms of historical info, I want to know how long the patient has had hair loss, what symptoms they have/had, and if there is a family history of hair loss. Also, if the patient has been treated in the past, it is helpful to know if any of the previous treatments worked. For me, the clinical exam is going to tell the most. Just the clinical location will give the first cues, followed by trichoscopy. I don’t biopsy every patient, but when there is any doubt about the form of hair loss the patient has, biopsy is important. I use the dermatoscope to guide the biopsy and perform two 4-mm punch biopsies in the appropriate area.
How do you manage active inflammatory scarring alopecias in skin of color, including first-line therapies and criteria for escalation?
DR. McMICHAEL: I like to discuss and give guidance on hair care first. Sometimes this will include increased frequency of hair washing and/or use of an antidandruff shampoo. Next, I usually start patients on a potent or ultrapotent steroid several times per week. If there is very symptomatic or clinically raging inflammation, I will begin either an oral antibiotic for 3 to 6 months or a series of intralesional steroids. Depending on the condition and patient preference, a stronger anti-inflammatory agent such as hydroxychloroquine, methotrexate, or dapsone may be used. Monitoring depends on the medication that is started. Many of the anti-inflammatory medications require laboratory monitoring every 4 or 6 months, and the drug dictates this timing. For monitoring of disease progression, questions for the patient on continued symptoms and serial clinical and trichoscopic photographs make the difference.
When and how do you use adjunctive or emerging therapies for scarring alopecias in skin of color, and what evidence and outcomes guide their use?
DR. McMICHAEL: Our research group has shown via small clinical trials and case series that both platelet-rich plasma and low-level laser light can be helpful for central centrifugal cicatricial alopecia, so I tend to begin these treatments once the inflammation is at a mild or minimal level. I often begin low-dose oral minoxidil right at the start or at any time during the course of treatment. I do not use topical minoxidil when the patient is symptomatic but often use this when patients are asymptomatic and do not wish to take oral minoxidil. There are now good case series to show minoxidil can be helpful for scarring forms of alopecia. Patients can show improvement on these treatments, but I only promise stabilization. It is difficult to predict who will have increased growth prior to treatment, and that is what I tell patients.
When are surgical options appropriate for scarring alopecia in patients with skin of color, and what precautions or criteria are required?
DR. McMICHAEL: Once everything has been done medically for scarring alopecia, surgical options can be considered. Unfortunately, these options typically are not covered by insurance, so patients have to be willing and able to pay out of pocket. Other criteria include that patients must have quiescent inflammation and enough donor hair for hair restoration via transplant. I do not personally perform hair transplantation, but I do like to refer patients with tightly coiled hair and scarring alopecia to physicians who specialize in this kind of treatment. Not all transplant surgeons have experience with skin of color or follicular unit extraction, which is sometimes needed.
What culturally tailored strategies help prevent scarring alopecia in patients with skin of color, and where are research or practice gaps?
DR. McMICHAEL: For all patients, it is important to ask about daily hair care practices, hair care at the salon, and night-time hair care before making recommendations. Any hair style that puts tension on the hair shaft needs to be discontinued as do those that are tight around the frontal or posterior hair line. Hair washing should be once per week or every 2 weeks at the least, and conditioning of the hair is important with hair trims every 2 to 3 months.
How do you distinguish active cicatricial alopecia from chronic nonprogressive hair loss in patients with skin of color, and which clinical or trichoscopic features suggest ongoing inflammation requiring treatment?
DR. McMICHAEL: Typically, I use trichoscopy to help determine if there are fine or vellus hairs in the affected area. Often, if there are vellus hairs, that means there is the potential to lengthen growth time and have thicker longer hairs in the area, even in a chronic nonprogressive hair loss state. If there is clinical or trichoscopic erythema (suggesting inflammation), that is a sure sign of active scarring. Patient-reported symptoms of itch, stinging, or pain are a sign of active scarring as well. Finally, even if none of the above are true, a biopsy can help determine if there is active inflammation, as can increasing size of the affected area on the scalp upon return visit to the clinic.
What is your initial diagnostic workup for suspected scarring alopecia in patients with skin of color, including key history, trichoscopy, and biopsy approach?
DR. McMICHAEL: In terms of historical info, I want to know how long the patient has had hair loss, what symptoms they have/had, and if there is a family history of hair loss. Also, if the patient has been treated in the past, it is helpful to know if any of the previous treatments worked. For me, the clinical exam is going to tell the most. Just the clinical location will give the first cues, followed by trichoscopy. I don’t biopsy every patient, but when there is any doubt about the form of hair loss the patient has, biopsy is important. I use the dermatoscope to guide the biopsy and perform two 4-mm punch biopsies in the appropriate area.
How do you manage active inflammatory scarring alopecias in skin of color, including first-line therapies and criteria for escalation?
DR. McMICHAEL: I like to discuss and give guidance on hair care first. Sometimes this will include increased frequency of hair washing and/or use of an antidandruff shampoo. Next, I usually start patients on a potent or ultrapotent steroid several times per week. If there is very symptomatic or clinically raging inflammation, I will begin either an oral antibiotic for 3 to 6 months or a series of intralesional steroids. Depending on the condition and patient preference, a stronger anti-inflammatory agent such as hydroxychloroquine, methotrexate, or dapsone may be used. Monitoring depends on the medication that is started. Many of the anti-inflammatory medications require laboratory monitoring every 4 or 6 months, and the drug dictates this timing. For monitoring of disease progression, questions for the patient on continued symptoms and serial clinical and trichoscopic photographs make the difference.
When and how do you use adjunctive or emerging therapies for scarring alopecias in skin of color, and what evidence and outcomes guide their use?
DR. McMICHAEL: Our research group has shown via small clinical trials and case series that both platelet-rich plasma and low-level laser light can be helpful for central centrifugal cicatricial alopecia, so I tend to begin these treatments once the inflammation is at a mild or minimal level. I often begin low-dose oral minoxidil right at the start or at any time during the course of treatment. I do not use topical minoxidil when the patient is symptomatic but often use this when patients are asymptomatic and do not wish to take oral minoxidil. There are now good case series to show minoxidil can be helpful for scarring forms of alopecia. Patients can show improvement on these treatments, but I only promise stabilization. It is difficult to predict who will have increased growth prior to treatment, and that is what I tell patients.
When are surgical options appropriate for scarring alopecia in patients with skin of color, and what precautions or criteria are required?
DR. McMICHAEL: Once everything has been done medically for scarring alopecia, surgical options can be considered. Unfortunately, these options typically are not covered by insurance, so patients have to be willing and able to pay out of pocket. Other criteria include that patients must have quiescent inflammation and enough donor hair for hair restoration via transplant. I do not personally perform hair transplantation, but I do like to refer patients with tightly coiled hair and scarring alopecia to physicians who specialize in this kind of treatment. Not all transplant surgeons have experience with skin of color or follicular unit extraction, which is sometimes needed.
What culturally tailored strategies help prevent scarring alopecia in patients with skin of color, and where are research or practice gaps?
DR. McMICHAEL: For all patients, it is important to ask about daily hair care practices, hair care at the salon, and night-time hair care before making recommendations. Any hair style that puts tension on the hair shaft needs to be discontinued as do those that are tight around the frontal or posterior hair line. Hair washing should be once per week or every 2 weeks at the least, and conditioning of the hair is important with hair trims every 2 to 3 months.
How do you distinguish active cicatricial alopecia from chronic nonprogressive hair loss in patients with skin of color, and which clinical or trichoscopic features suggest ongoing inflammation requiring treatment?
DR. McMICHAEL: Typically, I use trichoscopy to help determine if there are fine or vellus hairs in the affected area. Often, if there are vellus hairs, that means there is the potential to lengthen growth time and have thicker longer hairs in the area, even in a chronic nonprogressive hair loss state. If there is clinical or trichoscopic erythema (suggesting inflammation), that is a sure sign of active scarring. Patient-reported symptoms of itch, stinging, or pain are a sign of active scarring as well. Finally, even if none of the above are true, a biopsy can help determine if there is active inflammation, as can increasing size of the affected area on the scalp upon return visit to the clinic.
What is your initial diagnostic workup for suspected scarring alopecia in patients with skin of color, including key history, trichoscopy, and biopsy approach?
DR. McMICHAEL: In terms of historical info, I want to know how long the patient has had hair loss, what symptoms they have/had, and if there is a family history of hair loss. Also, if the patient has been treated in the past, it is helpful to know if any of the previous treatments worked. For me, the clinical exam is going to tell the most. Just the clinical location will give the first cues, followed by trichoscopy. I don’t biopsy every patient, but when there is any doubt about the form of hair loss the patient has, biopsy is important. I use the dermatoscope to guide the biopsy and perform two 4-mm punch biopsies in the appropriate area.
How do you manage active inflammatory scarring alopecias in skin of color, including first-line therapies and criteria for escalation?
DR. McMICHAEL: I like to discuss and give guidance on hair care first. Sometimes this will include increased frequency of hair washing and/or use of an antidandruff shampoo. Next, I usually start patients on a potent or ultrapotent steroid several times per week. If there is very symptomatic or clinically raging inflammation, I will begin either an oral antibiotic for 3 to 6 months or a series of intralesional steroids. Depending on the condition and patient preference, a stronger anti-inflammatory agent such as hydroxychloroquine, methotrexate, or dapsone may be used. Monitoring depends on the medication that is started. Many of the anti-inflammatory medications require laboratory monitoring every 4 or 6 months, and the drug dictates this timing. For monitoring of disease progression, questions for the patient on continued symptoms and serial clinical and trichoscopic photographs make the difference.
When and how do you use adjunctive or emerging therapies for scarring alopecias in skin of color, and what evidence and outcomes guide their use?
DR. McMICHAEL: Our research group has shown via small clinical trials and case series that both platelet-rich plasma and low-level laser light can be helpful for central centrifugal cicatricial alopecia, so I tend to begin these treatments once the inflammation is at a mild or minimal level. I often begin low-dose oral minoxidil right at the start or at any time during the course of treatment. I do not use topical minoxidil when the patient is symptomatic but often use this when patients are asymptomatic and do not wish to take oral minoxidil. There are now good case series to show minoxidil can be helpful for scarring forms of alopecia. Patients can show improvement on these treatments, but I only promise stabilization. It is difficult to predict who will have increased growth prior to treatment, and that is what I tell patients.
When are surgical options appropriate for scarring alopecia in patients with skin of color, and what precautions or criteria are required?
DR. McMICHAEL: Once everything has been done medically for scarring alopecia, surgical options can be considered. Unfortunately, these options typically are not covered by insurance, so patients have to be willing and able to pay out of pocket. Other criteria include that patients must have quiescent inflammation and enough donor hair for hair restoration via transplant. I do not personally perform hair transplantation, but I do like to refer patients with tightly coiled hair and scarring alopecia to physicians who specialize in this kind of treatment. Not all transplant surgeons have experience with skin of color or follicular unit extraction, which is sometimes needed.
What culturally tailored strategies help prevent scarring alopecia in patients with skin of color, and where are research or practice gaps?
DR. McMICHAEL: For all patients, it is important to ask about daily hair care practices, hair care at the salon, and night-time hair care before making recommendations. Any hair style that puts tension on the hair shaft needs to be discontinued as do those that are tight around the frontal or posterior hair line. Hair washing should be once per week or every 2 weeks at the least, and conditioning of the hair is important with hair trims every 2 to 3 months.
Scarring Alopecia in Skin of Color: Practical Guidance From Dr. Amy J. McMichael
Scarring Alopecia in Skin of Color: Practical Guidance From Dr. Amy J. McMichael
Evaluating GPT-4o for Automated Classification of Skin Lesions Using the HAM10000 Dataset
Evaluating GPT-4o for Automated Classification of Skin Lesions Using the HAM10000 Dataset
To the Editor:
The widespread availability and popularity of ChatGPT (OpenAI) have sparked interest in its potential applications within various fields, including medical diagnostics.1 In dermatology, large language models (LLMs) already are being cited as a possible way to reliably respond to common patient queries and produce concise patient education materials.2,3 That being said, there is skepticism regarding the technology’s efficacy and reliability in producing accurate treatment plans, with variability among popular LLMs; for example, a recent study by Chau et al4 demonstrated that ChatGPT was best at providing specific and accurate information regarding patient-facing responses to questions about 5 dermatologic diagnoses compared to Google Bard (now rebranded as Google Gemini) and Bing AI (now rebranded as Microsoft Copilot), which more often produced inaccurate or nonspecific responses. Google Bard also declined to answer one prompt.4 Large language models also have been evaluated in diagnosing skin lesions. In 2024, SkinGPT-4 (a pretrained multimodel LLM developed by Zhou et al5) achieved just over 80% accuracy in interpreting images of skin lesions and was considered informative by 82.5% of board-certified dermatologists, demonstrating that LLMs may have the potential to become integrated into clinical practice.5
Our study aimed to evaluate the performance of GPT-4o (OpenAI)—a widely accessible, low-cost LLM—in diagnosing dermatologic conditions using the HAM10000 dataset, a well-curated collection of dermatoscopic images developed for training and benchmarking artificial intelligence (AI) algorithms.6 HAM10000 comprises images representing 7 distinct skin conditions: actinic keratoses (ak), basal cell carcinoma (bcc), benign keratosis (bk), dermatofibroma (df), melanoma (mel), melanocytic nevi (nv), and vascular skin lesions (vsl), providing a robust platform for multiclass classification assessment. We evaluated GPT-4o using 100 dermatoscopic images per condition to assess diagnostic accuracy, potential biases, and limitations in skin lesion identification. The HAM10000 dataset was selected because it offers a large standardized reference set of dermatoscopic (rather than conventional clinical) images commonly used in dermatologic AI research. GPT-4o was chosen due to its patient-friendly interface, widespread use, and prior reports suggesting greater reliability in skin lesion assessment compared with other LLMs.
One hundred images from each of the 7 dermatologic categories were randomly selected for use in our analysis in 2024. The images were selected by our data scientist (J.C.) through random sampling from the dataset. Each image was separately presented to GPT-4o without any preprocessing or modification alongside 2 prompts designed to evaluate the diagnostic capabilities of GPT-4o. Both prompts included the same list of 7 dermatologic conditions for answer choices but differed in contextual information, where prompt 1 provided patient demographic information and localization of the dermatological condition but prompt 2 did not provide these details (Table). No follow-up questions were presented.

For prompt 1, the confusion matrix showed a strong bias toward detecting mel and bcc, with high true positives (mel, 83%; bcc, 37%)(eFigure 1). This pattern possibly suggests a tendency to favor malignant labels (eg, mel, BCC) when uncertainty is present. Interestingly, df and vsl also had notable true positives (46% and 37%, respectively), which is unexpected for less critical conditions because the model’s correct classifications were uneven across benign lesions. Actinic keratoses and nv showed higher misclassification rates, suggesting the model struggled to distinguish them from other lesions.
As shown in eTable 1, prompt 1 exhibited the highest recall for mel at 0.83 but performed worse in precision (0.242) and specificity (0.567) compared to ak, which had an extremely low recall (0.03) but very high specificity (0.992) and moderate precision score (0.375). The highest precision score was seen with vsl (0.738), which also achieved high scores in specificity (0.982) and accuracy (0.88) and performed moderately well in recall (0.31). All performance metrics are reported as proportions (0-1.0), wherein 1.0 indicates 100.
For prompt 2, the second confusion matrix followed similar trends as prompt 1 but still differed in key areas (eFigure 2). Melanoma detection remained strong (true positives, 95%), while bcc shows slightly fewer true positives (24%). Vascular skin lesions improve in true positives (40%), and df dropped slightly (33%). The model continues to struggle with ak and nv, with notable misclassifications observed across other categories
Similar to prompt 1, prompt 2 achieved its highest recall for mel (0.95%), but demonstrated lower precision (0.223%) and specificity (0.488%) for this class. Prompt 2 also produced the highest accuracy for vascular skin lesions (0.90%). The highest specificity was observed for both bk and ak (0.992% each); however, ak again demonstrated the lowest recall, with a value of 0.01%.
A previous study utilizing a model of binary classification to distinguish between mel and benign dermatologic conditions demonstrated poor performance.1 Additionally, prior studies have employed a less-strict, open-ended style question approach to examine ChatGPT’s ability to diagnose mel with limited efficacy.7 The HAM10000 dataset was specifically selected despite its limitations (including the absence of clinical images and limited diversity in skin tones) due to its comprehensive nature, robust annotation standards, and widespread acceptance in dermatologic AI research. Compared to the Diverse Dermatology Images dataset, which notably lacks skin tone diversity, HAM10000 provides a balanced representation of several dermatologic conditions crucial for multiclass classification tasks, making it suitable for benchmarking AI performance. This study aimed to eliminate these limitations by employing a multiclass classification approach; however, despite this switch, our results indicate continued and major limitations of the diagnostic capabilities of GPT-4o.
In its current form, GPT-4o appeared to demonstrate a clear accuracy bias toward correctly identifying specific and severe dermatologic conditions (eg, mel, bcc) but showed low and variable class-level performance for other categories (eg, ak, nv, df, vsl), with frequent misclassification into melanoma or basal cell carcinoma and low recall for some classes (eTables 1 and 2). This finding emphasized that GPT-4o currently lacks the reliability needed for real-life clinical applications in dermatology, as both binary and multiclass models fail to achieve consistent accurate performance across all skin conditions. Notably, GPT-4o may generate false-positive malignant classifications among patients due to its skew in predicted labels toward labeling benign lesions as malignant.
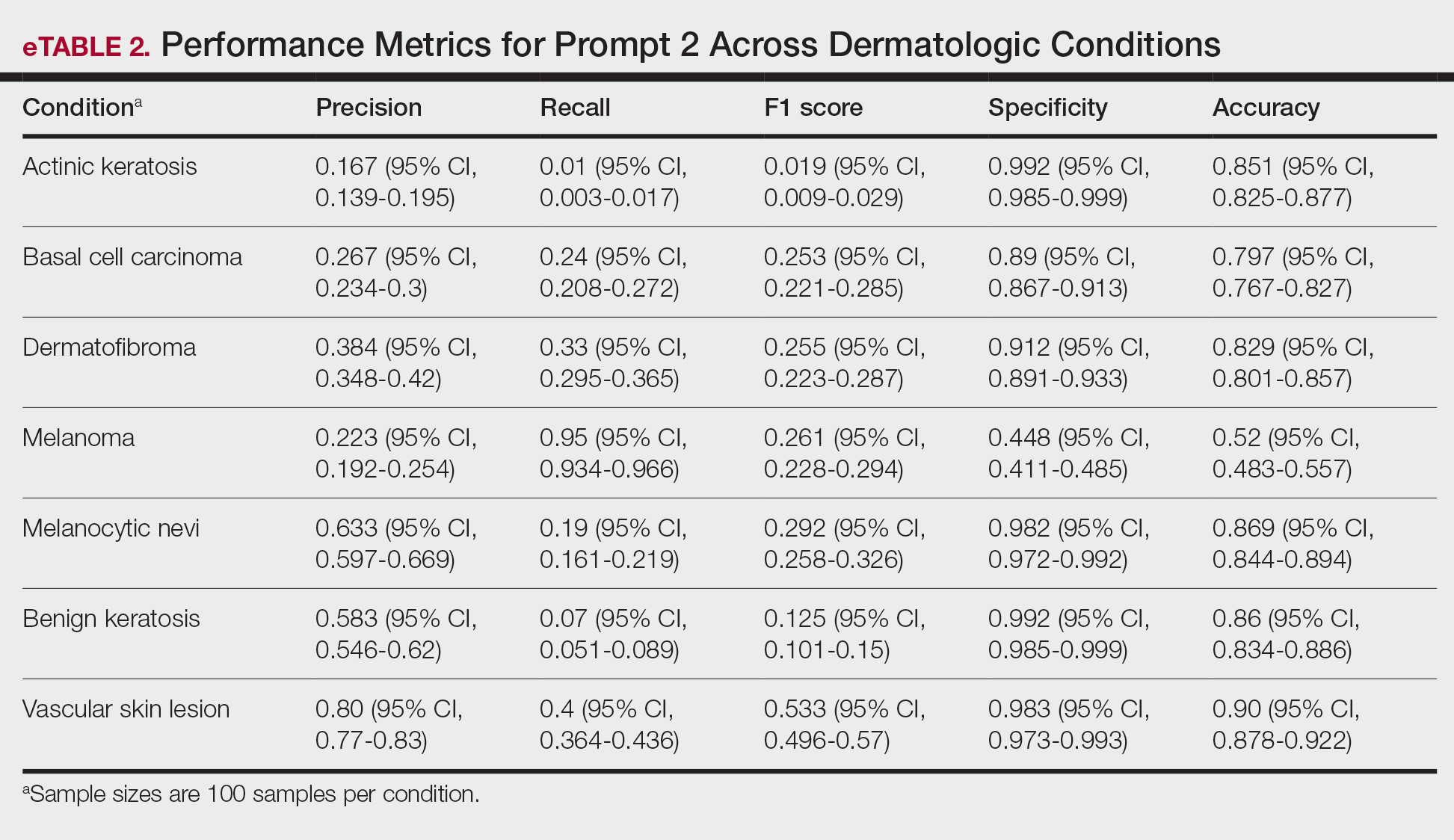
From the patient perspective, younger individuals may upload images of benign nevi only to unnecessarily fear a mel diagnosis after receiving GPT-4o results. Statistically, younger patients are less likely than older patients to have malignant lesions and more likely to instead present with common vsl or df—lesions that GPT-4o appears likely to identify correctly.8 For older users, however, the situation may differ. Beyond ak being misclassified as bcc, older patients also may encounter GPT-4o outputs that mislabel lesions as mel, raising concerns and heightening anxiety. Given the technology’s tendency to overestimate the risk of serious dermatologic conditions, this behavior poses a considerable challenge in its current state and may inadvertently intensify public anxiety around mel.
A notable limitation of our study was that, compared to publicly available datasets, the HAM10000 dataset includes only dermatoscopic images rather than a combination of clinical and dermatoscopic images. Furthermore, the HAM10000 dataset comprises images primarily from White patients, whereas other diverse databases (eg, the Diverse Dermatology Images dataset) may be more suitable for training AI algorithms to accurately diagnose skin lesions in individuals with a variety of skin tones.9
Ultimately, our results signal that major advancements in the design and training of LLMs such as GPT-4o are necessary before these systems can be integrated into dermatologic diagnostic decision-making to offer benefit rather than cause harm. Consulting a health care professional rather than relying solely on AI, which might otherwise lead to avoidable stress, unnecessary alarm, and potentially increased health care costs due to unwarranted follow-up and testing, should remain the recommended standard of care for patients suspecting a skin lesion.
- Caruccio L, Cirillo S, Polese G, et al. Can ChatGPT provide intelligent diagnoses? A comparative study between predictive models and ChatGPT to define a new medical diagnostic bot. Expert Syst Appl. 2024;235:121186. doi:10.1016/j.eswa.2023.121186
- Ferreira AL, Chu B, Grant-Kels JM, et al. Evaluation of ChatGPT dermatology responses to common patient queries. JMIR Dermatol. 2023;6:E49280. doi:10.2196/49280
- Chen R, Zhang Y, Choi S, et al. The chatbots are coming: risks and benefits of consumer-facing artificial intelligence in clinical dermatology. J Am Acad Dermatol. 2023;89:872-874. doi:10.1016/j.jaad.2023.05.088
- Chau C, Feng H, Cobos G, et al. The comparative sufficiency of ChatGPT, Google Bard, and Bing AI in answering diagnosis, treatment, and prognosis questions about common dermatological diagnoses. JMIR Dermatol. 2025;8:E60827. doi:10.2196/60827
- Zhou J, He X, Sun L, et al. Pre-trained multimodal large language model enhances dermatological diagnosis using SkinGPT-4. Nat Commun. 2024;15:5649. doi:10.1038/s41467-024-50043-3
- Tschandl P, Rosendahl C, Kittler H. The HAM10000 dataset, a large collection of multi-source dermatoscopic images of common pigmented skin lesions. Sci Data. 2018;5:180161. doi:10.1038/sdata.2018.161
- Shifai N, van Doorn R, Malvehy J, et al. Can ChatGPT vision diagnose melanoma? An exploratory diagnostic accuracy study. J Am Acad Dermatol. 2024;90:1057-1059. doi:10.1016/j.jaad.2023.12.062
- Cortez JL, Vasquez J, Wei ML. The impact of demographics, socioeconomics, and health care access on melanoma outcomes. J Am Acad Dermatol. 2021;84:1677-1683. doi:10.1016/j.jaad.2020.07.125
- Daneshjou R, Vodrahalli K, Novoa RA, et al. Disparities in dermatology AI performance on a diverse, curated clinical image set. Sci Adv. 2022;8:Eabq6147. doi:10.1126/sciadv.abq6147
To the Editor:
The widespread availability and popularity of ChatGPT (OpenAI) have sparked interest in its potential applications within various fields, including medical diagnostics.1 In dermatology, large language models (LLMs) already are being cited as a possible way to reliably respond to common patient queries and produce concise patient education materials.2,3 That being said, there is skepticism regarding the technology’s efficacy and reliability in producing accurate treatment plans, with variability among popular LLMs; for example, a recent study by Chau et al4 demonstrated that ChatGPT was best at providing specific and accurate information regarding patient-facing responses to questions about 5 dermatologic diagnoses compared to Google Bard (now rebranded as Google Gemini) and Bing AI (now rebranded as Microsoft Copilot), which more often produced inaccurate or nonspecific responses. Google Bard also declined to answer one prompt.4 Large language models also have been evaluated in diagnosing skin lesions. In 2024, SkinGPT-4 (a pretrained multimodel LLM developed by Zhou et al5) achieved just over 80% accuracy in interpreting images of skin lesions and was considered informative by 82.5% of board-certified dermatologists, demonstrating that LLMs may have the potential to become integrated into clinical practice.5
Our study aimed to evaluate the performance of GPT-4o (OpenAI)—a widely accessible, low-cost LLM—in diagnosing dermatologic conditions using the HAM10000 dataset, a well-curated collection of dermatoscopic images developed for training and benchmarking artificial intelligence (AI) algorithms.6 HAM10000 comprises images representing 7 distinct skin conditions: actinic keratoses (ak), basal cell carcinoma (bcc), benign keratosis (bk), dermatofibroma (df), melanoma (mel), melanocytic nevi (nv), and vascular skin lesions (vsl), providing a robust platform for multiclass classification assessment. We evaluated GPT-4o using 100 dermatoscopic images per condition to assess diagnostic accuracy, potential biases, and limitations in skin lesion identification. The HAM10000 dataset was selected because it offers a large standardized reference set of dermatoscopic (rather than conventional clinical) images commonly used in dermatologic AI research. GPT-4o was chosen due to its patient-friendly interface, widespread use, and prior reports suggesting greater reliability in skin lesion assessment compared with other LLMs.
One hundred images from each of the 7 dermatologic categories were randomly selected for use in our analysis in 2024. The images were selected by our data scientist (J.C.) through random sampling from the dataset. Each image was separately presented to GPT-4o without any preprocessing or modification alongside 2 prompts designed to evaluate the diagnostic capabilities of GPT-4o. Both prompts included the same list of 7 dermatologic conditions for answer choices but differed in contextual information, where prompt 1 provided patient demographic information and localization of the dermatological condition but prompt 2 did not provide these details (Table). No follow-up questions were presented.

For prompt 1, the confusion matrix showed a strong bias toward detecting mel and bcc, with high true positives (mel, 83%; bcc, 37%)(eFigure 1). This pattern possibly suggests a tendency to favor malignant labels (eg, mel, BCC) when uncertainty is present. Interestingly, df and vsl also had notable true positives (46% and 37%, respectively), which is unexpected for less critical conditions because the model’s correct classifications were uneven across benign lesions. Actinic keratoses and nv showed higher misclassification rates, suggesting the model struggled to distinguish them from other lesions.

As shown in eTable 1, prompt 1 exhibited the highest recall for mel at 0.83 but performed worse in precision (0.242) and specificity (0.567) compared to ak, which had an extremely low recall (0.03) but very high specificity (0.992) and moderate precision score (0.375). The highest precision score was seen with vsl (0.738), which also achieved high scores in specificity (0.982) and accuracy (0.88) and performed moderately well in recall (0.31). All performance metrics are reported as proportions (0-1.0), wherein 1.0 indicates 100.

For prompt 2, the second confusion matrix followed similar trends as prompt 1 but still differed in key areas (eFigure 2). Melanoma detection remained strong (true positives, 95%), while bcc shows slightly fewer true positives (24%). Vascular skin lesions improve in true positives (40%), and df dropped slightly (33%). The model continues to struggle with ak and nv, with notable misclassifications observed across other categories

Similar to prompt 1, prompt 2 achieved its highest recall for mel (0.95%), but demonstrated lower precision (0.223%) and specificity (0.488%) for this class. Prompt 2 also produced the highest accuracy for vascular skin lesions (0.90%). The highest specificity was observed for both bk and ak (0.992% each); however, ak again demonstrated the lowest recall, with a value of 0.01%.
A previous study utilizing a model of binary classification to distinguish between mel and benign dermatologic conditions demonstrated poor performance.1 Additionally, prior studies have employed a less-strict, open-ended style question approach to examine ChatGPT’s ability to diagnose mel with limited efficacy.7 The HAM10000 dataset was specifically selected despite its limitations (including the absence of clinical images and limited diversity in skin tones) due to its comprehensive nature, robust annotation standards, and widespread acceptance in dermatologic AI research. Compared to the Diverse Dermatology Images dataset, which notably lacks skin tone diversity, HAM10000 provides a balanced representation of several dermatologic conditions crucial for multiclass classification tasks, making it suitable for benchmarking AI performance. This study aimed to eliminate these limitations by employing a multiclass classification approach; however, despite this switch, our results indicate continued and major limitations of the diagnostic capabilities of GPT-4o.
In its current form, GPT-4o appeared to demonstrate a clear accuracy bias toward correctly identifying specific and severe dermatologic conditions (eg, mel, bcc) but showed low and variable class-level performance for other categories (eg, ak, nv, df, vsl), with frequent misclassification into melanoma or basal cell carcinoma and low recall for some classes (eTables 1 and 2). This finding emphasized that GPT-4o currently lacks the reliability needed for real-life clinical applications in dermatology, as both binary and multiclass models fail to achieve consistent accurate performance across all skin conditions. Notably, GPT-4o may generate false-positive malignant classifications among patients due to its skew in predicted labels toward labeling benign lesions as malignant.

From the patient perspective, younger individuals may upload images of benign nevi only to unnecessarily fear a mel diagnosis after receiving GPT-4o results. Statistically, younger patients are less likely than older patients to have malignant lesions and more likely to instead present with common vsl or df—lesions that GPT-4o appears likely to identify correctly.8 For older users, however, the situation may differ. Beyond ak being misclassified as bcc, older patients also may encounter GPT-4o outputs that mislabel lesions as mel, raising concerns and heightening anxiety. Given the technology’s tendency to overestimate the risk of serious dermatologic conditions, this behavior poses a considerable challenge in its current state and may inadvertently intensify public anxiety around mel.
A notable limitation of our study was that, compared to publicly available datasets, the HAM10000 dataset includes only dermatoscopic images rather than a combination of clinical and dermatoscopic images. Furthermore, the HAM10000 dataset comprises images primarily from White patients, whereas other diverse databases (eg, the Diverse Dermatology Images dataset) may be more suitable for training AI algorithms to accurately diagnose skin lesions in individuals with a variety of skin tones.9
Ultimately, our results signal that major advancements in the design and training of LLMs such as GPT-4o are necessary before these systems can be integrated into dermatologic diagnostic decision-making to offer benefit rather than cause harm. Consulting a health care professional rather than relying solely on AI, which might otherwise lead to avoidable stress, unnecessary alarm, and potentially increased health care costs due to unwarranted follow-up and testing, should remain the recommended standard of care for patients suspecting a skin lesion.
To the Editor:
The widespread availability and popularity of ChatGPT (OpenAI) have sparked interest in its potential applications within various fields, including medical diagnostics.1 In dermatology, large language models (LLMs) already are being cited as a possible way to reliably respond to common patient queries and produce concise patient education materials.2,3 That being said, there is skepticism regarding the technology’s efficacy and reliability in producing accurate treatment plans, with variability among popular LLMs; for example, a recent study by Chau et al4 demonstrated that ChatGPT was best at providing specific and accurate information regarding patient-facing responses to questions about 5 dermatologic diagnoses compared to Google Bard (now rebranded as Google Gemini) and Bing AI (now rebranded as Microsoft Copilot), which more often produced inaccurate or nonspecific responses. Google Bard also declined to answer one prompt.4 Large language models also have been evaluated in diagnosing skin lesions. In 2024, SkinGPT-4 (a pretrained multimodel LLM developed by Zhou et al5) achieved just over 80% accuracy in interpreting images of skin lesions and was considered informative by 82.5% of board-certified dermatologists, demonstrating that LLMs may have the potential to become integrated into clinical practice.5
Our study aimed to evaluate the performance of GPT-4o (OpenAI)—a widely accessible, low-cost LLM—in diagnosing dermatologic conditions using the HAM10000 dataset, a well-curated collection of dermatoscopic images developed for training and benchmarking artificial intelligence (AI) algorithms.6 HAM10000 comprises images representing 7 distinct skin conditions: actinic keratoses (ak), basal cell carcinoma (bcc), benign keratosis (bk), dermatofibroma (df), melanoma (mel), melanocytic nevi (nv), and vascular skin lesions (vsl), providing a robust platform for multiclass classification assessment. We evaluated GPT-4o using 100 dermatoscopic images per condition to assess diagnostic accuracy, potential biases, and limitations in skin lesion identification. The HAM10000 dataset was selected because it offers a large standardized reference set of dermatoscopic (rather than conventional clinical) images commonly used in dermatologic AI research. GPT-4o was chosen due to its patient-friendly interface, widespread use, and prior reports suggesting greater reliability in skin lesion assessment compared with other LLMs.
One hundred images from each of the 7 dermatologic categories were randomly selected for use in our analysis in 2024. The images were selected by our data scientist (J.C.) through random sampling from the dataset. Each image was separately presented to GPT-4o without any preprocessing or modification alongside 2 prompts designed to evaluate the diagnostic capabilities of GPT-4o. Both prompts included the same list of 7 dermatologic conditions for answer choices but differed in contextual information, where prompt 1 provided patient demographic information and localization of the dermatological condition but prompt 2 did not provide these details (Table). No follow-up questions were presented.

For prompt 1, the confusion matrix showed a strong bias toward detecting mel and bcc, with high true positives (mel, 83%; bcc, 37%)(eFigure 1). This pattern possibly suggests a tendency to favor malignant labels (eg, mel, BCC) when uncertainty is present. Interestingly, df and vsl also had notable true positives (46% and 37%, respectively), which is unexpected for less critical conditions because the model’s correct classifications were uneven across benign lesions. Actinic keratoses and nv showed higher misclassification rates, suggesting the model struggled to distinguish them from other lesions.

As shown in eTable 1, prompt 1 exhibited the highest recall for mel at 0.83 but performed worse in precision (0.242) and specificity (0.567) compared to ak, which had an extremely low recall (0.03) but very high specificity (0.992) and moderate precision score (0.375). The highest precision score was seen with vsl (0.738), which also achieved high scores in specificity (0.982) and accuracy (0.88) and performed moderately well in recall (0.31). All performance metrics are reported as proportions (0-1.0), wherein 1.0 indicates 100.

For prompt 2, the second confusion matrix followed similar trends as prompt 1 but still differed in key areas (eFigure 2). Melanoma detection remained strong (true positives, 95%), while bcc shows slightly fewer true positives (24%). Vascular skin lesions improve in true positives (40%), and df dropped slightly (33%). The model continues to struggle with ak and nv, with notable misclassifications observed across other categories

Similar to prompt 1, prompt 2 achieved its highest recall for mel (0.95%), but demonstrated lower precision (0.223%) and specificity (0.488%) for this class. Prompt 2 also produced the highest accuracy for vascular skin lesions (0.90%). The highest specificity was observed for both bk and ak (0.992% each); however, ak again demonstrated the lowest recall, with a value of 0.01%.
A previous study utilizing a model of binary classification to distinguish between mel and benign dermatologic conditions demonstrated poor performance.1 Additionally, prior studies have employed a less-strict, open-ended style question approach to examine ChatGPT’s ability to diagnose mel with limited efficacy.7 The HAM10000 dataset was specifically selected despite its limitations (including the absence of clinical images and limited diversity in skin tones) due to its comprehensive nature, robust annotation standards, and widespread acceptance in dermatologic AI research. Compared to the Diverse Dermatology Images dataset, which notably lacks skin tone diversity, HAM10000 provides a balanced representation of several dermatologic conditions crucial for multiclass classification tasks, making it suitable for benchmarking AI performance. This study aimed to eliminate these limitations by employing a multiclass classification approach; however, despite this switch, our results indicate continued and major limitations of the diagnostic capabilities of GPT-4o.
In its current form, GPT-4o appeared to demonstrate a clear accuracy bias toward correctly identifying specific and severe dermatologic conditions (eg, mel, bcc) but showed low and variable class-level performance for other categories (eg, ak, nv, df, vsl), with frequent misclassification into melanoma or basal cell carcinoma and low recall for some classes (eTables 1 and 2). This finding emphasized that GPT-4o currently lacks the reliability needed for real-life clinical applications in dermatology, as both binary and multiclass models fail to achieve consistent accurate performance across all skin conditions. Notably, GPT-4o may generate false-positive malignant classifications among patients due to its skew in predicted labels toward labeling benign lesions as malignant.

From the patient perspective, younger individuals may upload images of benign nevi only to unnecessarily fear a mel diagnosis after receiving GPT-4o results. Statistically, younger patients are less likely than older patients to have malignant lesions and more likely to instead present with common vsl or df—lesions that GPT-4o appears likely to identify correctly.8 For older users, however, the situation may differ. Beyond ak being misclassified as bcc, older patients also may encounter GPT-4o outputs that mislabel lesions as mel, raising concerns and heightening anxiety. Given the technology’s tendency to overestimate the risk of serious dermatologic conditions, this behavior poses a considerable challenge in its current state and may inadvertently intensify public anxiety around mel.
A notable limitation of our study was that, compared to publicly available datasets, the HAM10000 dataset includes only dermatoscopic images rather than a combination of clinical and dermatoscopic images. Furthermore, the HAM10000 dataset comprises images primarily from White patients, whereas other diverse databases (eg, the Diverse Dermatology Images dataset) may be more suitable for training AI algorithms to accurately diagnose skin lesions in individuals with a variety of skin tones.9
Ultimately, our results signal that major advancements in the design and training of LLMs such as GPT-4o are necessary before these systems can be integrated into dermatologic diagnostic decision-making to offer benefit rather than cause harm. Consulting a health care professional rather than relying solely on AI, which might otherwise lead to avoidable stress, unnecessary alarm, and potentially increased health care costs due to unwarranted follow-up and testing, should remain the recommended standard of care for patients suspecting a skin lesion.
- Caruccio L, Cirillo S, Polese G, et al. Can ChatGPT provide intelligent diagnoses? A comparative study between predictive models and ChatGPT to define a new medical diagnostic bot. Expert Syst Appl. 2024;235:121186. doi:10.1016/j.eswa.2023.121186
- Ferreira AL, Chu B, Grant-Kels JM, et al. Evaluation of ChatGPT dermatology responses to common patient queries. JMIR Dermatol. 2023;6:E49280. doi:10.2196/49280
- Chen R, Zhang Y, Choi S, et al. The chatbots are coming: risks and benefits of consumer-facing artificial intelligence in clinical dermatology. J Am Acad Dermatol. 2023;89:872-874. doi:10.1016/j.jaad.2023.05.088
- Chau C, Feng H, Cobos G, et al. The comparative sufficiency of ChatGPT, Google Bard, and Bing AI in answering diagnosis, treatment, and prognosis questions about common dermatological diagnoses. JMIR Dermatol. 2025;8:E60827. doi:10.2196/60827
- Zhou J, He X, Sun L, et al. Pre-trained multimodal large language model enhances dermatological diagnosis using SkinGPT-4. Nat Commun. 2024;15:5649. doi:10.1038/s41467-024-50043-3
- Tschandl P, Rosendahl C, Kittler H. The HAM10000 dataset, a large collection of multi-source dermatoscopic images of common pigmented skin lesions. Sci Data. 2018;5:180161. doi:10.1038/sdata.2018.161
- Shifai N, van Doorn R, Malvehy J, et al. Can ChatGPT vision diagnose melanoma? An exploratory diagnostic accuracy study. J Am Acad Dermatol. 2024;90:1057-1059. doi:10.1016/j.jaad.2023.12.062
- Cortez JL, Vasquez J, Wei ML. The impact of demographics, socioeconomics, and health care access on melanoma outcomes. J Am Acad Dermatol. 2021;84:1677-1683. doi:10.1016/j.jaad.2020.07.125
- Daneshjou R, Vodrahalli K, Novoa RA, et al. Disparities in dermatology AI performance on a diverse, curated clinical image set. Sci Adv. 2022;8:Eabq6147. doi:10.1126/sciadv.abq6147
- Caruccio L, Cirillo S, Polese G, et al. Can ChatGPT provide intelligent diagnoses? A comparative study between predictive models and ChatGPT to define a new medical diagnostic bot. Expert Syst Appl. 2024;235:121186. doi:10.1016/j.eswa.2023.121186
- Ferreira AL, Chu B, Grant-Kels JM, et al. Evaluation of ChatGPT dermatology responses to common patient queries. JMIR Dermatol. 2023;6:E49280. doi:10.2196/49280
- Chen R, Zhang Y, Choi S, et al. The chatbots are coming: risks and benefits of consumer-facing artificial intelligence in clinical dermatology. J Am Acad Dermatol. 2023;89:872-874. doi:10.1016/j.jaad.2023.05.088
- Chau C, Feng H, Cobos G, et al. The comparative sufficiency of ChatGPT, Google Bard, and Bing AI in answering diagnosis, treatment, and prognosis questions about common dermatological diagnoses. JMIR Dermatol. 2025;8:E60827. doi:10.2196/60827
- Zhou J, He X, Sun L, et al. Pre-trained multimodal large language model enhances dermatological diagnosis using SkinGPT-4. Nat Commun. 2024;15:5649. doi:10.1038/s41467-024-50043-3
- Tschandl P, Rosendahl C, Kittler H. The HAM10000 dataset, a large collection of multi-source dermatoscopic images of common pigmented skin lesions. Sci Data. 2018;5:180161. doi:10.1038/sdata.2018.161
- Shifai N, van Doorn R, Malvehy J, et al. Can ChatGPT vision diagnose melanoma? An exploratory diagnostic accuracy study. J Am Acad Dermatol. 2024;90:1057-1059. doi:10.1016/j.jaad.2023.12.062
- Cortez JL, Vasquez J, Wei ML. The impact of demographics, socioeconomics, and health care access on melanoma outcomes. J Am Acad Dermatol. 2021;84:1677-1683. doi:10.1016/j.jaad.2020.07.125
- Daneshjou R, Vodrahalli K, Novoa RA, et al. Disparities in dermatology AI performance on a diverse, curated clinical image set. Sci Adv. 2022;8:Eabq6147. doi:10.1126/sciadv.abq6147
Evaluating GPT-4o for Automated Classification of Skin Lesions Using the HAM10000 Dataset
Evaluating GPT-4o for Automated Classification of Skin Lesions Using the HAM10000 Dataset
Practice Points
- Even with a multiclass classification framework designed to assist GPT-4o, the model encountered notable challenges in accurately diagnosing skin lesions.
- In its current form, GPT-4o may provide inaccurate and misleading information to patients who use its interface to evaluate suspected skin lesions. Patients should continue to seek clinical consultation from health care professionals.
The Future of Psoriasis Care
The Future of Psoriasis Care
Psoriasis research and treatment have come a long way in the past 2 to 3 decades. With the advent of biologic therapy, increasingly more targeted therapies, and a better pathophysiological understanding, our treatment paradigms and ability to treat psoriatic disease have shown great improvement; however, despite these advances, there remain several areas in need of further development to continue to improve our care of patients with psoriasis, including comorbidities, access to care, technology, and clinical care.
Treatment Implications of Comorbidities
It has become increasingly clear that psoriasis carries with it numerous medical and psychiatric comorbidities; however, our ability to utilize these factors in treatment decision-making is still nascent. Clinically, multiple studies have demonstrated a connection between cardiovascular disease (CVD) and psoriasis, often with a direct relationship between CVD and psoriasis severity.1-3 The cytokines involved (interleukin [IL]–17) and cell types (primarily neutrophils) are the same in psoriatic disease and evolving atherosclerotic plaques.4,5 In contrast, other analyses do not support a relationship between CVD and psoriasis, and there has been no direct and definitive demonstration that giving patients a specific psoriasis treatment could help reduce cardiovascular risk. Perhaps this is due to the sample sizes and time needed to demonstrate such a connection, as we are dealing with fairly rare events overall. Strategies to identify patients at risk for cardiovascular events, such as starting from a cohort with existing CVD and investigating treatment effects in that population, may yield worthwhile dividends. Perhaps one day we will be able to offer treatments that not only help clear psoriasis but also modulate cardiovascular health.
Our understanding of the psychiatric effects of psoriasis is even less developed. The strongest links have been demonstrated between psoriasis and depression, anxiety, and suicidal ideation.6 Some of these connections have been recognized for more than 3 decades: one study from 1993 showed that almost 10% of patients with psoriasis wished to be dead and 5.6% reported active suicidal ideation at the time of the study.7 Why is it, then, that we still do not have a good understanding of the interrelationship between psoriasis, mental health, and therapeutics? There likely is a connection between these components, as it is now well accepted that cytokines (eg, interferons) can have a considerable impact on depression and that treatment with biologics for psoriasis tends to improve depressive symptoms.8 This is an area in which we need better awareness and understanding as well as some guidance on how to approach this topic with our patients—particularly how mental health may play into therapeutic decisions for psoriasis, such as earlier escalation to rapid-acting systemic therapy in patients with psychiatric comorbidities.
Access to Psoriasis Care
With so many effective treatments for psoriasis, one of the most frustrating challenges we face is that many patients with psoriasis still experience notable barriers to care. While access in urban areas generally is reasonable, in rural areas, 75% of patients have no psoriasis-treating providers in their ZIP code and have to seek psoriasis-related care outside the 3-digit ZIP code prefix.9 Unfortunately, in most cases, even after traveling and waiting for an appointment patients will not be offered the full spectrum of available psoriasis treatments. Dermatologists already are much harder to find in rural areas, but the proportion of rural counties without a dermatologist who prescribes biologics approaches 90%.10 Functionally, this places a huge burden on our patients, who frustratingly see commercials for highly effective psoriasis treatments on television but are not able to access them. What good is having medicines that can help more than two-thirds of patients achieve 100% clearance11 when patients cannot access them?
Technology and Treatment Optimization
As our society becomes ever more technologically advanced, medicine seems to be caught in a bit of a quagmire, with our practices often using outdated technology in the name of HIPAA compliance and communicating via fax on important matters such as medication coverage. Nevertheless, dermatologists are beginning to increase integration of artificial intelligence (AI) and advanced technologies to make patient care more efficient and effective via education/awareness, image analysis, remote management, and telemedicine.12 Recently, the National Psoriasis Foundation published guidance for the use of telemedicine, suggesting that it could be used for expanded access and expedited care in appropriate settings.13 However, some caution should be used when interpreting data in this sphere. While AI technology has been purported to outpace dermatologists’ diagnosis of psoriasis in some cases, the conditions tested (ie, the training set and evaluation image bank) and special tools used (such as dermoscopy, which is not routinely used in clinical practice for psoriasis diagnosis) may make the results inapplicable to general care.14
Perhaps more promising is the use of digital aids to help with long-term care, treatment reminders, and comorbidity evaluation/screening. Similarly, telemedicine can be utilized to provide skilled psoriasis care to patients in rural areas who otherwise might not have access. One such program demonstrated that asynchronous e-consults were able to achieve Psoriasis Area and Severity Index (PASI) and body surface area outcomes similar to in-person dermatologist care.15 Using AI and technology also could assist with drug development and guide treatment. For example, a psoriatic arthritis (PsA) risk model developed in a Danish cohort suggested that early treatment with an IL-17 inhibitor in high-risk patients could reduce PsA incidence by 64%.16
Personalized Clinical Care
Even as we become accustomed to higher PASI 90, PASI 100, and mean PASI improvement numbers with our newer biologics, drug development in psoriasis has not stopped. Pipeline medications include an oral peptide-based IL-23 inhibitor17 and targeted tyrosine kinase 2 inhibitors.18,19 What is perhaps most interesting is to envision a future in which we could select treatments based on either patient phenotype (eg, involvement of hands and feet could suggest a certain single or class of medicine) or genotype.20 This has clear impacts on patient care, as dedicated trials of psoriasis medications tend to result in lower achievement of outcome measure thresholds than subanalyses of clinical trials; for example, in a dedicated trial of risankizumab for nonpustular palmoplantar psoriasis, achievement of a palmoplantar Investigator Global Assessment score of clear or almost clear was demonstrated in 33.3% of treated patients vs 16.1% of those receiving placebo at week 16 (P=.006).21 A subanalysis from the pivotal UltiMMA trials showed that more than 70% of risankizumab-treated patients achieved complete clearance (palmoplantar PASI score of 0) by week 16.22 Indeed, there is some evidence to suggest that the pathophysiology of plaque psoriasis, nonpustular palmoplantar psoriasis, and palmoplantar pustular psoriasis are different, with more interferon-γ signaling involved in nonpustular palmoplantar psoriasis23—which may explain why some limited case reports have suggested the use of Janus kinase inhibitors for recalcitrant cases of palmoplantar plaque psoriasis.24
Even with such high rates of skin clearance, the treatment landscape in PsA lags behind. There is a need for higher-efficacy treatments in PsA. On a positive note, it may be reflective of how advanced our treatment conversations about psoriasis have become that rather than analyzing gross PASI improvements between one drug and another, we now are able to address nuanced differences between various presentations of psoriasis to help us select the right tool from our treatment toolbox.
Final Thoughts
We are lucky to practice dermatology in a time when there has been so much development, with many good treatment options for patients with psoriasis. What we had thought of as the ultimate goal in the past—to get the skin relatively clear—is now a realistic outcome for most patients. This allows us to focus on other important considerations, such as assessing and addressing comorbidities, improving access to care, implementing technology to improve psoriasis care, and refining our understanding of how different manifestations of psoriasis should alter our approach to treating patients. And though we have come a long way in recent years, there still is much to be done to lift up the psoriasis community as a whole. It’s reassuring to know that many are still working toward this goal.
- Cui P, Li D, Shi L, et al. Cardiovascular comorbidities among patients with psoriasis: a national register-based study in China. Sci Rep. 2024;14:19683.
- Tinggaard AB, Hjuler KF, Andersen IT, et al. Prevalence and severity of coronary artery disease linked to prognosis in psoriasis and psoriatic arthritis patients: a multi-centre cohort study. J Intern Med. 2021;290:693-703.
- Yang YW, Keller JJ, Lin HC. Medical comorbidity associated with psoriasis in adults: a population-based study. Br J Dermatol. 2011;165:1037-1043.
- Silvestre-Roig C, Braster Q, Ortega-Gomez A, et al. Neutrophils as regulators of cardiovascular inflammation. Nat Rev Cardiol. 2020;17:327-340.
- von Stebut E, Boehncke WH, Ghoreschi K, et al. IL-17A in psoriasis and beyond: cardiovascular and metabolic implications. Front Immunol. 2019;10:3096.
- Kurd SK, Troxel AB, Crits-Christoph P, et al. The risk of depression, anxiety, and suicidality in patients with psoriasis: a population-based cohort study. Arch Dermatol. 2010;146:891-895.
- Gupta MA, Schork NJ, Gupta AK, et al. Suicidal ideation in psoriasis. Int J Dermatol. 1993;32:188-190.
- Mrowietz U, Sumbul M, Gerdes S. Depression, a major comorbidity of psoriatic disease, is caused by metabolic inflammation. J Eur Acad Dermatol Venereol. 2023;37:1731-1738.
- Seigel L, Shoaib S, Maughn K, et al. Health disparities in psoriasis: geographic barriers to access in the United States. J Dermatolog Treat. 2024;35:2365820.
- Sun QW, Feng H, Cohen JM. Geographic trends in psoriasis injectable biologic prescriptions by dermatologists among Medicare beneficiaries. J Am Acad Dermatol. 2024;91:1214-1216.
- Reich K, Warren RB, Lebwohl M, et al. Bimekizumab versus secukinumab in plaque psoriasis. N Engl J Med. 2021;385:142-152.
- Wongvibulsin S, Lee I. Artificial intelligence and dermatology. JAMA Dermatol. 2025;161:344.
- Pelet Del Toro N, Yahia R, Feldman SR, et al. National Psoriasis Foundation Telemedicine Task Force guidance for management of psoriatic disease via telemedicine. JAAD Int. 2023;12:32-36.
- Yang Y, Wang J, Xie F, et al. A convolutional neural network trained with dermoscopic images of psoriasis performed on par with 230 dermatologists. Comput Biol Med. 2021;139:104924.
- Armstrong AW, Chambers CJ, Maverakis E, et al. Effectiveness of online vs in-person care for adults with psoriasis: a randomized clinical trial. JAMA Netw Open. 2018;1:E183062.
- Gong Z, Cheng Y, Wei X, et al. Digital technologies in psoriasis management: from precision diagnosis to therapeutic innovation and holistic care. Front Digit Health. 2025;8:1656585.
- Bissonnette R, Soung J, Hebert AA, et al. Oral icotrokinra for plaque psoriasis in adults and adolescents. N Engl J Med. 2025;393:1784-1795.
- Armstrong AW, Gooderham M, Lynde C, et al. Tyrosine kinase 2 inhibition with zasocitinib (TAK-279) in psoriasis: a randomized clinical trial. JAMA Dermatol. 2024;160:1066-1074.
- Blauvelt A, Arenberger P, Sauder MB, et al. Highly selective, allosteric inhibition of TYK2 with oral ESK-001 in patients with moderate-to-severe plaque psoriasis: results from STRIDE, a 12-week, randomized, double-blinded, placebo-controlled, dose-ranging phase 2 study. J Am Acad Dermatol. 2026;94:57-65.
- Tchack M KN, Sandeep R, Muller E, et al. Gene expression or mutational profile for psoriasis in guiding treatment: a systematic review. JAAD Reviews. 2025;5:100-102.
- Lebwohl M, Bukhalo M, Stein Gold L, et al. A randomized phase 3b study evaluating the safety and efficacy of risankizumab in adult patients with moderate-to-severe plaque psoriasis with non-pustular palmoplantar involvement. J Am Acad Dermatol. 2024;91:1150-1157.
- Strober B, Bachelez H, Crowley J, et al. Efficacy of long-term risankizumab treatment for moderate-to-severe plaque psoriasis: subgroup analyses by baseline characteristics and psoriatic disease manifestations through 256 weeks (LIMMitless trial). J Eur Acad Dermatol Venereol. 2024;38:864-872.
- Wang CQ, Haxhinasto S, Garcet S, et al. Comparison of the inflammatory circuits in psoriasis vulgaris, non‒pustular palmoplantar psoriasis, and palmoplantar pustular psoriasis. J Invest Dermatol. 2023;143:87-97.
- Choi B, Li HO, Glassman SJ. Palmoplantar plaque psoriasis responsive to upadacitinib: a report of two cases. SAGE Open Med Case Rep. 2025;13:2050313X251317763.
Psoriasis research and treatment have come a long way in the past 2 to 3 decades. With the advent of biologic therapy, increasingly more targeted therapies, and a better pathophysiological understanding, our treatment paradigms and ability to treat psoriatic disease have shown great improvement; however, despite these advances, there remain several areas in need of further development to continue to improve our care of patients with psoriasis, including comorbidities, access to care, technology, and clinical care.
Treatment Implications of Comorbidities
It has become increasingly clear that psoriasis carries with it numerous medical and psychiatric comorbidities; however, our ability to utilize these factors in treatment decision-making is still nascent. Clinically, multiple studies have demonstrated a connection between cardiovascular disease (CVD) and psoriasis, often with a direct relationship between CVD and psoriasis severity.1-3 The cytokines involved (interleukin [IL]–17) and cell types (primarily neutrophils) are the same in psoriatic disease and evolving atherosclerotic plaques.4,5 In contrast, other analyses do not support a relationship between CVD and psoriasis, and there has been no direct and definitive demonstration that giving patients a specific psoriasis treatment could help reduce cardiovascular risk. Perhaps this is due to the sample sizes and time needed to demonstrate such a connection, as we are dealing with fairly rare events overall. Strategies to identify patients at risk for cardiovascular events, such as starting from a cohort with existing CVD and investigating treatment effects in that population, may yield worthwhile dividends. Perhaps one day we will be able to offer treatments that not only help clear psoriasis but also modulate cardiovascular health.
Our understanding of the psychiatric effects of psoriasis is even less developed. The strongest links have been demonstrated between psoriasis and depression, anxiety, and suicidal ideation.6 Some of these connections have been recognized for more than 3 decades: one study from 1993 showed that almost 10% of patients with psoriasis wished to be dead and 5.6% reported active suicidal ideation at the time of the study.7 Why is it, then, that we still do not have a good understanding of the interrelationship between psoriasis, mental health, and therapeutics? There likely is a connection between these components, as it is now well accepted that cytokines (eg, interferons) can have a considerable impact on depression and that treatment with biologics for psoriasis tends to improve depressive symptoms.8 This is an area in which we need better awareness and understanding as well as some guidance on how to approach this topic with our patients—particularly how mental health may play into therapeutic decisions for psoriasis, such as earlier escalation to rapid-acting systemic therapy in patients with psychiatric comorbidities.
Access to Psoriasis Care
With so many effective treatments for psoriasis, one of the most frustrating challenges we face is that many patients with psoriasis still experience notable barriers to care. While access in urban areas generally is reasonable, in rural areas, 75% of patients have no psoriasis-treating providers in their ZIP code and have to seek psoriasis-related care outside the 3-digit ZIP code prefix.9 Unfortunately, in most cases, even after traveling and waiting for an appointment patients will not be offered the full spectrum of available psoriasis treatments. Dermatologists already are much harder to find in rural areas, but the proportion of rural counties without a dermatologist who prescribes biologics approaches 90%.10 Functionally, this places a huge burden on our patients, who frustratingly see commercials for highly effective psoriasis treatments on television but are not able to access them. What good is having medicines that can help more than two-thirds of patients achieve 100% clearance11 when patients cannot access them?
Technology and Treatment Optimization
As our society becomes ever more technologically advanced, medicine seems to be caught in a bit of a quagmire, with our practices often using outdated technology in the name of HIPAA compliance and communicating via fax on important matters such as medication coverage. Nevertheless, dermatologists are beginning to increase integration of artificial intelligence (AI) and advanced technologies to make patient care more efficient and effective via education/awareness, image analysis, remote management, and telemedicine.12 Recently, the National Psoriasis Foundation published guidance for the use of telemedicine, suggesting that it could be used for expanded access and expedited care in appropriate settings.13 However, some caution should be used when interpreting data in this sphere. While AI technology has been purported to outpace dermatologists’ diagnosis of psoriasis in some cases, the conditions tested (ie, the training set and evaluation image bank) and special tools used (such as dermoscopy, which is not routinely used in clinical practice for psoriasis diagnosis) may make the results inapplicable to general care.14
Perhaps more promising is the use of digital aids to help with long-term care, treatment reminders, and comorbidity evaluation/screening. Similarly, telemedicine can be utilized to provide skilled psoriasis care to patients in rural areas who otherwise might not have access. One such program demonstrated that asynchronous e-consults were able to achieve Psoriasis Area and Severity Index (PASI) and body surface area outcomes similar to in-person dermatologist care.15 Using AI and technology also could assist with drug development and guide treatment. For example, a psoriatic arthritis (PsA) risk model developed in a Danish cohort suggested that early treatment with an IL-17 inhibitor in high-risk patients could reduce PsA incidence by 64%.16
Personalized Clinical Care
Even as we become accustomed to higher PASI 90, PASI 100, and mean PASI improvement numbers with our newer biologics, drug development in psoriasis has not stopped. Pipeline medications include an oral peptide-based IL-23 inhibitor17 and targeted tyrosine kinase 2 inhibitors.18,19 What is perhaps most interesting is to envision a future in which we could select treatments based on either patient phenotype (eg, involvement of hands and feet could suggest a certain single or class of medicine) or genotype.20 This has clear impacts on patient care, as dedicated trials of psoriasis medications tend to result in lower achievement of outcome measure thresholds than subanalyses of clinical trials; for example, in a dedicated trial of risankizumab for nonpustular palmoplantar psoriasis, achievement of a palmoplantar Investigator Global Assessment score of clear or almost clear was demonstrated in 33.3% of treated patients vs 16.1% of those receiving placebo at week 16 (P=.006).21 A subanalysis from the pivotal UltiMMA trials showed that more than 70% of risankizumab-treated patients achieved complete clearance (palmoplantar PASI score of 0) by week 16.22 Indeed, there is some evidence to suggest that the pathophysiology of plaque psoriasis, nonpustular palmoplantar psoriasis, and palmoplantar pustular psoriasis are different, with more interferon-γ signaling involved in nonpustular palmoplantar psoriasis23—which may explain why some limited case reports have suggested the use of Janus kinase inhibitors for recalcitrant cases of palmoplantar plaque psoriasis.24
Even with such high rates of skin clearance, the treatment landscape in PsA lags behind. There is a need for higher-efficacy treatments in PsA. On a positive note, it may be reflective of how advanced our treatment conversations about psoriasis have become that rather than analyzing gross PASI improvements between one drug and another, we now are able to address nuanced differences between various presentations of psoriasis to help us select the right tool from our treatment toolbox.
Final Thoughts
We are lucky to practice dermatology in a time when there has been so much development, with many good treatment options for patients with psoriasis. What we had thought of as the ultimate goal in the past—to get the skin relatively clear—is now a realistic outcome for most patients. This allows us to focus on other important considerations, such as assessing and addressing comorbidities, improving access to care, implementing technology to improve psoriasis care, and refining our understanding of how different manifestations of psoriasis should alter our approach to treating patients. And though we have come a long way in recent years, there still is much to be done to lift up the psoriasis community as a whole. It’s reassuring to know that many are still working toward this goal.
Psoriasis research and treatment have come a long way in the past 2 to 3 decades. With the advent of biologic therapy, increasingly more targeted therapies, and a better pathophysiological understanding, our treatment paradigms and ability to treat psoriatic disease have shown great improvement; however, despite these advances, there remain several areas in need of further development to continue to improve our care of patients with psoriasis, including comorbidities, access to care, technology, and clinical care.
Treatment Implications of Comorbidities
It has become increasingly clear that psoriasis carries with it numerous medical and psychiatric comorbidities; however, our ability to utilize these factors in treatment decision-making is still nascent. Clinically, multiple studies have demonstrated a connection between cardiovascular disease (CVD) and psoriasis, often with a direct relationship between CVD and psoriasis severity.1-3 The cytokines involved (interleukin [IL]–17) and cell types (primarily neutrophils) are the same in psoriatic disease and evolving atherosclerotic plaques.4,5 In contrast, other analyses do not support a relationship between CVD and psoriasis, and there has been no direct and definitive demonstration that giving patients a specific psoriasis treatment could help reduce cardiovascular risk. Perhaps this is due to the sample sizes and time needed to demonstrate such a connection, as we are dealing with fairly rare events overall. Strategies to identify patients at risk for cardiovascular events, such as starting from a cohort with existing CVD and investigating treatment effects in that population, may yield worthwhile dividends. Perhaps one day we will be able to offer treatments that not only help clear psoriasis but also modulate cardiovascular health.
Our understanding of the psychiatric effects of psoriasis is even less developed. The strongest links have been demonstrated between psoriasis and depression, anxiety, and suicidal ideation.6 Some of these connections have been recognized for more than 3 decades: one study from 1993 showed that almost 10% of patients with psoriasis wished to be dead and 5.6% reported active suicidal ideation at the time of the study.7 Why is it, then, that we still do not have a good understanding of the interrelationship between psoriasis, mental health, and therapeutics? There likely is a connection between these components, as it is now well accepted that cytokines (eg, interferons) can have a considerable impact on depression and that treatment with biologics for psoriasis tends to improve depressive symptoms.8 This is an area in which we need better awareness and understanding as well as some guidance on how to approach this topic with our patients—particularly how mental health may play into therapeutic decisions for psoriasis, such as earlier escalation to rapid-acting systemic therapy in patients with psychiatric comorbidities.
Access to Psoriasis Care
With so many effective treatments for psoriasis, one of the most frustrating challenges we face is that many patients with psoriasis still experience notable barriers to care. While access in urban areas generally is reasonable, in rural areas, 75% of patients have no psoriasis-treating providers in their ZIP code and have to seek psoriasis-related care outside the 3-digit ZIP code prefix.9 Unfortunately, in most cases, even after traveling and waiting for an appointment patients will not be offered the full spectrum of available psoriasis treatments. Dermatologists already are much harder to find in rural areas, but the proportion of rural counties without a dermatologist who prescribes biologics approaches 90%.10 Functionally, this places a huge burden on our patients, who frustratingly see commercials for highly effective psoriasis treatments on television but are not able to access them. What good is having medicines that can help more than two-thirds of patients achieve 100% clearance11 when patients cannot access them?
Technology and Treatment Optimization
As our society becomes ever more technologically advanced, medicine seems to be caught in a bit of a quagmire, with our practices often using outdated technology in the name of HIPAA compliance and communicating via fax on important matters such as medication coverage. Nevertheless, dermatologists are beginning to increase integration of artificial intelligence (AI) and advanced technologies to make patient care more efficient and effective via education/awareness, image analysis, remote management, and telemedicine.12 Recently, the National Psoriasis Foundation published guidance for the use of telemedicine, suggesting that it could be used for expanded access and expedited care in appropriate settings.13 However, some caution should be used when interpreting data in this sphere. While AI technology has been purported to outpace dermatologists’ diagnosis of psoriasis in some cases, the conditions tested (ie, the training set and evaluation image bank) and special tools used (such as dermoscopy, which is not routinely used in clinical practice for psoriasis diagnosis) may make the results inapplicable to general care.14
Perhaps more promising is the use of digital aids to help with long-term care, treatment reminders, and comorbidity evaluation/screening. Similarly, telemedicine can be utilized to provide skilled psoriasis care to patients in rural areas who otherwise might not have access. One such program demonstrated that asynchronous e-consults were able to achieve Psoriasis Area and Severity Index (PASI) and body surface area outcomes similar to in-person dermatologist care.15 Using AI and technology also could assist with drug development and guide treatment. For example, a psoriatic arthritis (PsA) risk model developed in a Danish cohort suggested that early treatment with an IL-17 inhibitor in high-risk patients could reduce PsA incidence by 64%.16
Personalized Clinical Care
Even as we become accustomed to higher PASI 90, PASI 100, and mean PASI improvement numbers with our newer biologics, drug development in psoriasis has not stopped. Pipeline medications include an oral peptide-based IL-23 inhibitor17 and targeted tyrosine kinase 2 inhibitors.18,19 What is perhaps most interesting is to envision a future in which we could select treatments based on either patient phenotype (eg, involvement of hands and feet could suggest a certain single or class of medicine) or genotype.20 This has clear impacts on patient care, as dedicated trials of psoriasis medications tend to result in lower achievement of outcome measure thresholds than subanalyses of clinical trials; for example, in a dedicated trial of risankizumab for nonpustular palmoplantar psoriasis, achievement of a palmoplantar Investigator Global Assessment score of clear or almost clear was demonstrated in 33.3% of treated patients vs 16.1% of those receiving placebo at week 16 (P=.006).21 A subanalysis from the pivotal UltiMMA trials showed that more than 70% of risankizumab-treated patients achieved complete clearance (palmoplantar PASI score of 0) by week 16.22 Indeed, there is some evidence to suggest that the pathophysiology of plaque psoriasis, nonpustular palmoplantar psoriasis, and palmoplantar pustular psoriasis are different, with more interferon-γ signaling involved in nonpustular palmoplantar psoriasis23—which may explain why some limited case reports have suggested the use of Janus kinase inhibitors for recalcitrant cases of palmoplantar plaque psoriasis.24
Even with such high rates of skin clearance, the treatment landscape in PsA lags behind. There is a need for higher-efficacy treatments in PsA. On a positive note, it may be reflective of how advanced our treatment conversations about psoriasis have become that rather than analyzing gross PASI improvements between one drug and another, we now are able to address nuanced differences between various presentations of psoriasis to help us select the right tool from our treatment toolbox.
Final Thoughts
We are lucky to practice dermatology in a time when there has been so much development, with many good treatment options for patients with psoriasis. What we had thought of as the ultimate goal in the past—to get the skin relatively clear—is now a realistic outcome for most patients. This allows us to focus on other important considerations, such as assessing and addressing comorbidities, improving access to care, implementing technology to improve psoriasis care, and refining our understanding of how different manifestations of psoriasis should alter our approach to treating patients. And though we have come a long way in recent years, there still is much to be done to lift up the psoriasis community as a whole. It’s reassuring to know that many are still working toward this goal.
- Cui P, Li D, Shi L, et al. Cardiovascular comorbidities among patients with psoriasis: a national register-based study in China. Sci Rep. 2024;14:19683.
- Tinggaard AB, Hjuler KF, Andersen IT, et al. Prevalence and severity of coronary artery disease linked to prognosis in psoriasis and psoriatic arthritis patients: a multi-centre cohort study. J Intern Med. 2021;290:693-703.
- Yang YW, Keller JJ, Lin HC. Medical comorbidity associated with psoriasis in adults: a population-based study. Br J Dermatol. 2011;165:1037-1043.
- Silvestre-Roig C, Braster Q, Ortega-Gomez A, et al. Neutrophils as regulators of cardiovascular inflammation. Nat Rev Cardiol. 2020;17:327-340.
- von Stebut E, Boehncke WH, Ghoreschi K, et al. IL-17A in psoriasis and beyond: cardiovascular and metabolic implications. Front Immunol. 2019;10:3096.
- Kurd SK, Troxel AB, Crits-Christoph P, et al. The risk of depression, anxiety, and suicidality in patients with psoriasis: a population-based cohort study. Arch Dermatol. 2010;146:891-895.
- Gupta MA, Schork NJ, Gupta AK, et al. Suicidal ideation in psoriasis. Int J Dermatol. 1993;32:188-190.
- Mrowietz U, Sumbul M, Gerdes S. Depression, a major comorbidity of psoriatic disease, is caused by metabolic inflammation. J Eur Acad Dermatol Venereol. 2023;37:1731-1738.
- Seigel L, Shoaib S, Maughn K, et al. Health disparities in psoriasis: geographic barriers to access in the United States. J Dermatolog Treat. 2024;35:2365820.
- Sun QW, Feng H, Cohen JM. Geographic trends in psoriasis injectable biologic prescriptions by dermatologists among Medicare beneficiaries. J Am Acad Dermatol. 2024;91:1214-1216.
- Reich K, Warren RB, Lebwohl M, et al. Bimekizumab versus secukinumab in plaque psoriasis. N Engl J Med. 2021;385:142-152.
- Wongvibulsin S, Lee I. Artificial intelligence and dermatology. JAMA Dermatol. 2025;161:344.
- Pelet Del Toro N, Yahia R, Feldman SR, et al. National Psoriasis Foundation Telemedicine Task Force guidance for management of psoriatic disease via telemedicine. JAAD Int. 2023;12:32-36.
- Yang Y, Wang J, Xie F, et al. A convolutional neural network trained with dermoscopic images of psoriasis performed on par with 230 dermatologists. Comput Biol Med. 2021;139:104924.
- Armstrong AW, Chambers CJ, Maverakis E, et al. Effectiveness of online vs in-person care for adults with psoriasis: a randomized clinical trial. JAMA Netw Open. 2018;1:E183062.
- Gong Z, Cheng Y, Wei X, et al. Digital technologies in psoriasis management: from precision diagnosis to therapeutic innovation and holistic care. Front Digit Health. 2025;8:1656585.
- Bissonnette R, Soung J, Hebert AA, et al. Oral icotrokinra for plaque psoriasis in adults and adolescents. N Engl J Med. 2025;393:1784-1795.
- Armstrong AW, Gooderham M, Lynde C, et al. Tyrosine kinase 2 inhibition with zasocitinib (TAK-279) in psoriasis: a randomized clinical trial. JAMA Dermatol. 2024;160:1066-1074.
- Blauvelt A, Arenberger P, Sauder MB, et al. Highly selective, allosteric inhibition of TYK2 with oral ESK-001 in patients with moderate-to-severe plaque psoriasis: results from STRIDE, a 12-week, randomized, double-blinded, placebo-controlled, dose-ranging phase 2 study. J Am Acad Dermatol. 2026;94:57-65.
- Tchack M KN, Sandeep R, Muller E, et al. Gene expression or mutational profile for psoriasis in guiding treatment: a systematic review. JAAD Reviews. 2025;5:100-102.
- Lebwohl M, Bukhalo M, Stein Gold L, et al. A randomized phase 3b study evaluating the safety and efficacy of risankizumab in adult patients with moderate-to-severe plaque psoriasis with non-pustular palmoplantar involvement. J Am Acad Dermatol. 2024;91:1150-1157.
- Strober B, Bachelez H, Crowley J, et al. Efficacy of long-term risankizumab treatment for moderate-to-severe plaque psoriasis: subgroup analyses by baseline characteristics and psoriatic disease manifestations through 256 weeks (LIMMitless trial). J Eur Acad Dermatol Venereol. 2024;38:864-872.
- Wang CQ, Haxhinasto S, Garcet S, et al. Comparison of the inflammatory circuits in psoriasis vulgaris, non‒pustular palmoplantar psoriasis, and palmoplantar pustular psoriasis. J Invest Dermatol. 2023;143:87-97.
- Choi B, Li HO, Glassman SJ. Palmoplantar plaque psoriasis responsive to upadacitinib: a report of two cases. SAGE Open Med Case Rep. 2025;13:2050313X251317763.
- Cui P, Li D, Shi L, et al. Cardiovascular comorbidities among patients with psoriasis: a national register-based study in China. Sci Rep. 2024;14:19683.
- Tinggaard AB, Hjuler KF, Andersen IT, et al. Prevalence and severity of coronary artery disease linked to prognosis in psoriasis and psoriatic arthritis patients: a multi-centre cohort study. J Intern Med. 2021;290:693-703.
- Yang YW, Keller JJ, Lin HC. Medical comorbidity associated with psoriasis in adults: a population-based study. Br J Dermatol. 2011;165:1037-1043.
- Silvestre-Roig C, Braster Q, Ortega-Gomez A, et al. Neutrophils as regulators of cardiovascular inflammation. Nat Rev Cardiol. 2020;17:327-340.
- von Stebut E, Boehncke WH, Ghoreschi K, et al. IL-17A in psoriasis and beyond: cardiovascular and metabolic implications. Front Immunol. 2019;10:3096.
- Kurd SK, Troxel AB, Crits-Christoph P, et al. The risk of depression, anxiety, and suicidality in patients with psoriasis: a population-based cohort study. Arch Dermatol. 2010;146:891-895.
- Gupta MA, Schork NJ, Gupta AK, et al. Suicidal ideation in psoriasis. Int J Dermatol. 1993;32:188-190.
- Mrowietz U, Sumbul M, Gerdes S. Depression, a major comorbidity of psoriatic disease, is caused by metabolic inflammation. J Eur Acad Dermatol Venereol. 2023;37:1731-1738.
- Seigel L, Shoaib S, Maughn K, et al. Health disparities in psoriasis: geographic barriers to access in the United States. J Dermatolog Treat. 2024;35:2365820.
- Sun QW, Feng H, Cohen JM. Geographic trends in psoriasis injectable biologic prescriptions by dermatologists among Medicare beneficiaries. J Am Acad Dermatol. 2024;91:1214-1216.
- Reich K, Warren RB, Lebwohl M, et al. Bimekizumab versus secukinumab in plaque psoriasis. N Engl J Med. 2021;385:142-152.
- Wongvibulsin S, Lee I. Artificial intelligence and dermatology. JAMA Dermatol. 2025;161:344.
- Pelet Del Toro N, Yahia R, Feldman SR, et al. National Psoriasis Foundation Telemedicine Task Force guidance for management of psoriatic disease via telemedicine. JAAD Int. 2023;12:32-36.
- Yang Y, Wang J, Xie F, et al. A convolutional neural network trained with dermoscopic images of psoriasis performed on par with 230 dermatologists. Comput Biol Med. 2021;139:104924.
- Armstrong AW, Chambers CJ, Maverakis E, et al. Effectiveness of online vs in-person care for adults with psoriasis: a randomized clinical trial. JAMA Netw Open. 2018;1:E183062.
- Gong Z, Cheng Y, Wei X, et al. Digital technologies in psoriasis management: from precision diagnosis to therapeutic innovation and holistic care. Front Digit Health. 2025;8:1656585.
- Bissonnette R, Soung J, Hebert AA, et al. Oral icotrokinra for plaque psoriasis in adults and adolescents. N Engl J Med. 2025;393:1784-1795.
- Armstrong AW, Gooderham M, Lynde C, et al. Tyrosine kinase 2 inhibition with zasocitinib (TAK-279) in psoriasis: a randomized clinical trial. JAMA Dermatol. 2024;160:1066-1074.
- Blauvelt A, Arenberger P, Sauder MB, et al. Highly selective, allosteric inhibition of TYK2 with oral ESK-001 in patients with moderate-to-severe plaque psoriasis: results from STRIDE, a 12-week, randomized, double-blinded, placebo-controlled, dose-ranging phase 2 study. J Am Acad Dermatol. 2026;94:57-65.
- Tchack M KN, Sandeep R, Muller E, et al. Gene expression or mutational profile for psoriasis in guiding treatment: a systematic review. JAAD Reviews. 2025;5:100-102.
- Lebwohl M, Bukhalo M, Stein Gold L, et al. A randomized phase 3b study evaluating the safety and efficacy of risankizumab in adult patients with moderate-to-severe plaque psoriasis with non-pustular palmoplantar involvement. J Am Acad Dermatol. 2024;91:1150-1157.
- Strober B, Bachelez H, Crowley J, et al. Efficacy of long-term risankizumab treatment for moderate-to-severe plaque psoriasis: subgroup analyses by baseline characteristics and psoriatic disease manifestations through 256 weeks (LIMMitless trial). J Eur Acad Dermatol Venereol. 2024;38:864-872.
- Wang CQ, Haxhinasto S, Garcet S, et al. Comparison of the inflammatory circuits in psoriasis vulgaris, non‒pustular palmoplantar psoriasis, and palmoplantar pustular psoriasis. J Invest Dermatol. 2023;143:87-97.
- Choi B, Li HO, Glassman SJ. Palmoplantar plaque psoriasis responsive to upadacitinib: a report of two cases. SAGE Open Med Case Rep. 2025;13:2050313X251317763.
The Future of Psoriasis Care
The Future of Psoriasis Care
Combination Guselkumab and Secukinumab for Plaque Psoriasis
Combination Guselkumab and Secukinumab for Plaque Psoriasis
To the Editor:
A 23-year-old man with plaque psoriasis experienced a flare after a 3-month interruption in his secukinumab therapy. The patient’s psoriasis previously was well controlled with secukinumab 300 mg every 4 weeks. Due to social impairment from the current flare, he received a one-time 100-mg dose of sample guselkumab. Secukinumab therapy was resumed 3 weeks later at 300 mg every 4 weeks. Improvement was gradual; 3 weeks after receiving guselkumab, plaques remained on the bilateral dorsal forearms and the lower abdomen. By 2 months posttreatment, there was decreased erythema and reduced scaling with the continued secukinumab therapy (eFigure 1). The patient reported complete clearance of the plaques 4 months after receiving guselkumab but experienced a brief recurrence at month 5. By month 6, his psoriasis returned to its prior baseline of mild disease. In this 6-month period after resuming treatment, the patient reported subjective improvement in itch and quality of life. He reported no infection or other adverse effects from the additional therapy.
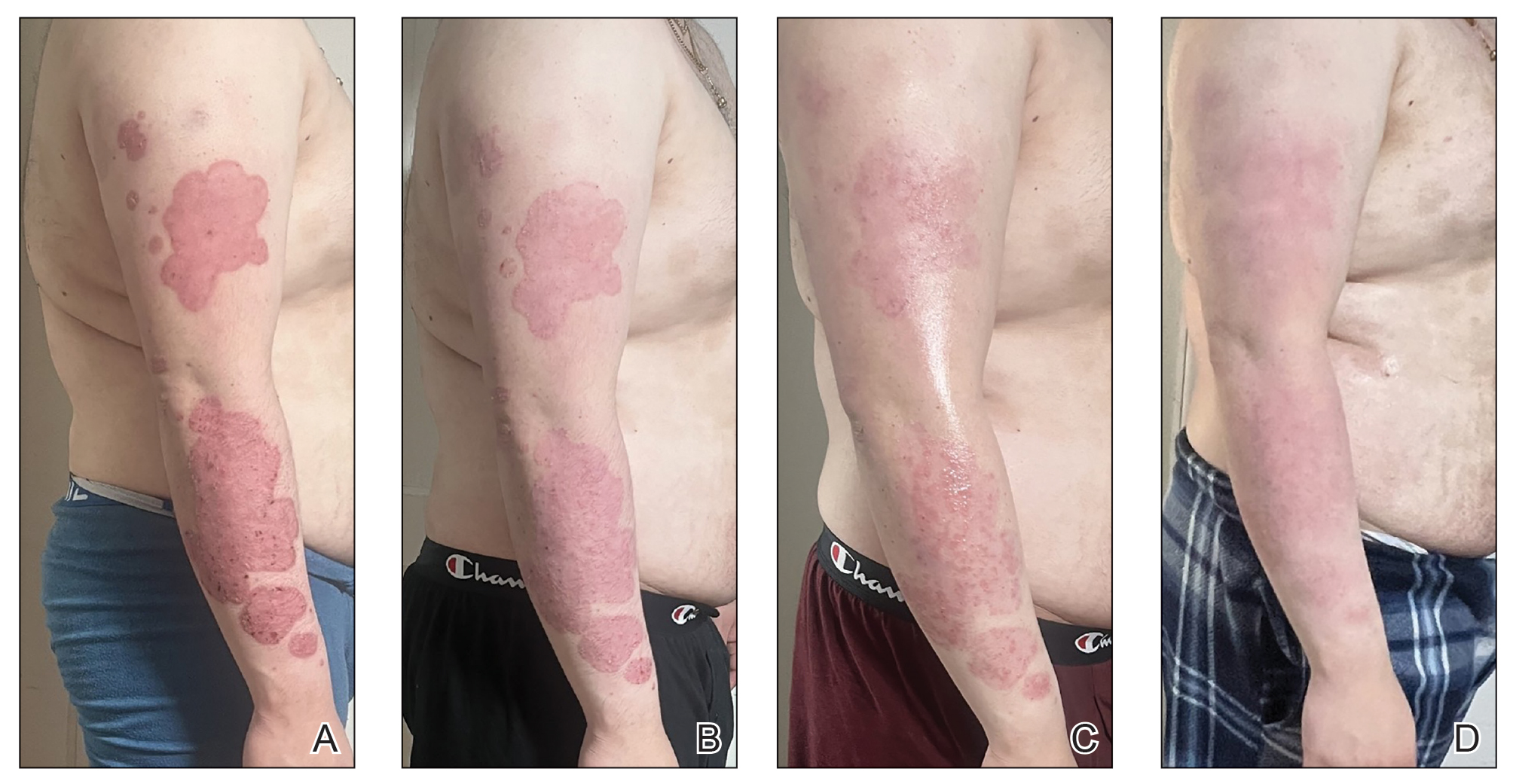
Psoriasis is driven by the interleukin (IL)-23–tyrosine kinase (TYK) 2–IL-17 axis.1 Guselkumab, an IL-23 inhibitor, and secukinumab, an IL-17A inhibitor, target distinct yet interconnected pathways in the pathogenesis of psoriasis.1 A key upstream regulator of the IL-17 pathway, IL-23 drives the expansion and survival of T helper 17 cells, which in turn produce IL-17A and IL-17F—cytokines involved in psoriatic inflammation.1 While monotherapy with either IL-23 or IL-17A inhibition generally is safe and highly effective, it does not always provide adequate disease control. By simultaneously inhibiting IL-23 and IL-17A, a combined approach may offer enhanced disease control compared to monotherapy.
Combined therapy with a single 100-mg dose of guselkumab and secukinumab 300 mg every 4 weeks may be effective for short-term control in patients who experience psoriasis flares. Although guselkumab has a half-life of approximately 15 to 18 days, its effects may extend beyond 4 to 5 half-lives, as improvement was seen in our case for 57 to 180 days. Although we did not test for the development of antidrug antibodies to secukinumab, there is evidence that the incidence of such development is exceedingly low and not correlated with clinical efficacy.2 We cannot exclude the possibility that the single dose of guselkumab may have been responsible for the entirety of our patient’s improvement, as up to 46% of patients treated with 12 months of guselkumab continued to experience remission at 6 months after discontinuation.3 Population data suggest a longer duration of activity or disease-modifying potential of guselkumab; however, affected patients had a longer duration of initial treatment before cessation.3
While data on the safety of dual cytokine inhibition in psoriasis are limited, patients in clinical trials for psoriatic arthritis often are treated with a biologic and methotrexate and/or oral corticosteroids; the safety of biologics even with such a combination is excellent.4 Combination biologic therapy with ustekinumab and etanercept in refractory psoriasis caused few adverse events.4,5 Combination tumor necrosis factor α and IL-12/23 inhibition is comparatively more immunosuppressive than combination IL-23 and IL-17 inhibition. Additionally, there may be redundancies in the immune system to the IL-23–TYK-2–IL-17 axis.1 While inhibition of both IL-17A and IL-17F increases cutaneous candidiasis risk, inhibiting IL-23 or IL-17A alone is associated with less risk for candidal infection.6,7 This suggests that redundancy via IL-17F derived from non-IL-23–dependent pathways may offer protection during dual inhibition (eFigure 2). Our patient experienced no adverse effects with a single 100-mg dose of guselkumab, suggesting that at least short-term combination therapy may be well tolerated.
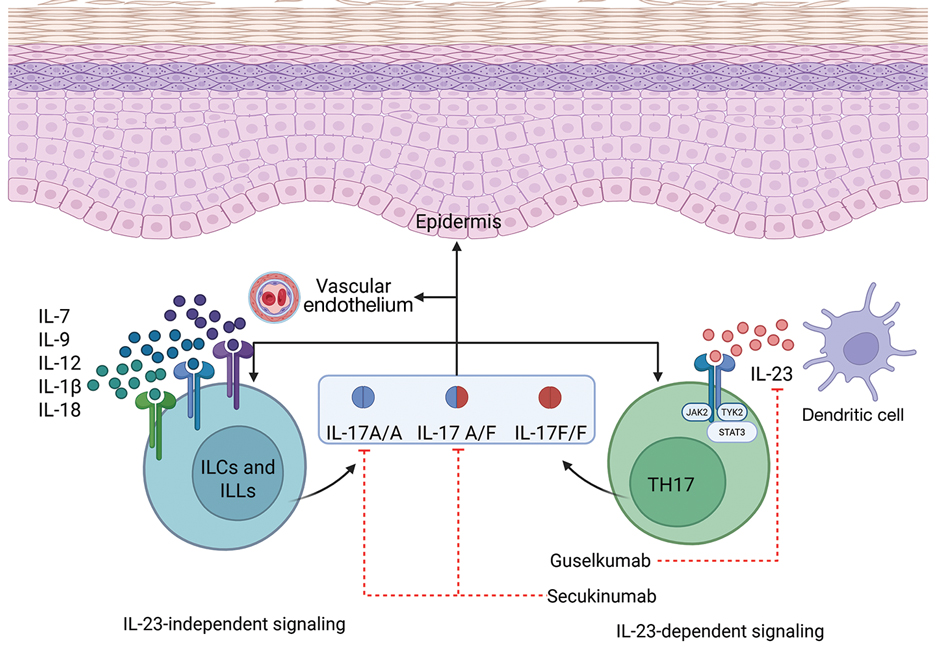
The safety profile seen in our case supports forgoing a washout period when switching between biologics, which can delay access to much-needed therapy and induce flares. Our patient had no adverse events, which provides anecdotal evidence supporting initiation of new biologics without delay. It is possible that the risks of overlapping two low-risk biologics may be lower than combining one biologic with a more immunosuppressive oral immunomodulator.
Combining standard-therapy secukinumab with a one-time dose of guselkumab may provide a synergistic effect by targeting different cytokines involved in the pathogenesis of psoriasis. Using 2 monoclonal antibodies with complementary targets may offer a strategy for rapid control of severe psoriasis that is refractory to single biologics.
- Griffiths CEM, Armstrong AW, Gudjonsson JE, et al. Psoriasis. Lancet. 2021;397:1301-1315. doi:10.1016/S0140-6736(20)32549-6
- Tsakok T, Rispens T, Spuls P, et al. Immunogenicity of biologic therapies in psoriasis: myths, facts and a suggested approach. J Eur Acad Dermatol Venereol. 2021;35:329-337. doi:10.1111/jdv.16980
- Zhdanava M, Fitzgerald T, Pilon D, et al. Comparative analysis of persistence and remission with guselkumab versus secukinumab and ixekizumab in the United States. J Dermatolog Treat. 2024;35:1. doi:10.1080/09546634.2024.2349658
- Kumthekar A, Ashrafi M, Deodhar A. Difficult to treat psoriatic arthritis—how should we manage? Clin Rheumatol. 2023;42:2251-2265. doi:10.1007/S10067-023-06605-9
- Thibodeaux Q, Ly K, Reddy V, et al. Dual biologic therapy for recalcitrant psoriasis and psoriatic arthritis. JAAD Case Rep. 2019;5:928-930. doi:10.1016/j.jdcr.2019.08.015
- Lebwohl MG, Merola JF, Rowland K, et al. Safety of guselkumab treatment for up to 5 years in patients with moderate-to-severe psoriasis: pooled analyses across seven clinical trials with more than 8600 patient-years of exposure. Br J Dermatol. 2023;189:42-52. doi:10.1093/BJD/LJAD115
- Reich K, Warren RB, Lebwohl M, et al. Bimekizumab versus secukinumab in plaque psoriasis. N Engl J Med. 2021;385:142-152. doi:10.1056/NEJMOA2102383
To the Editor:
A 23-year-old man with plaque psoriasis experienced a flare after a 3-month interruption in his secukinumab therapy. The patient’s psoriasis previously was well controlled with secukinumab 300 mg every 4 weeks. Due to social impairment from the current flare, he received a one-time 100-mg dose of sample guselkumab. Secukinumab therapy was resumed 3 weeks later at 300 mg every 4 weeks. Improvement was gradual; 3 weeks after receiving guselkumab, plaques remained on the bilateral dorsal forearms and the lower abdomen. By 2 months posttreatment, there was decreased erythema and reduced scaling with the continued secukinumab therapy (eFigure 1). The patient reported complete clearance of the plaques 4 months after receiving guselkumab but experienced a brief recurrence at month 5. By month 6, his psoriasis returned to its prior baseline of mild disease. In this 6-month period after resuming treatment, the patient reported subjective improvement in itch and quality of life. He reported no infection or other adverse effects from the additional therapy.

Psoriasis is driven by the interleukin (IL)-23–tyrosine kinase (TYK) 2–IL-17 axis.1 Guselkumab, an IL-23 inhibitor, and secukinumab, an IL-17A inhibitor, target distinct yet interconnected pathways in the pathogenesis of psoriasis.1 A key upstream regulator of the IL-17 pathway, IL-23 drives the expansion and survival of T helper 17 cells, which in turn produce IL-17A and IL-17F—cytokines involved in psoriatic inflammation.1 While monotherapy with either IL-23 or IL-17A inhibition generally is safe and highly effective, it does not always provide adequate disease control. By simultaneously inhibiting IL-23 and IL-17A, a combined approach may offer enhanced disease control compared to monotherapy.
Combined therapy with a single 100-mg dose of guselkumab and secukinumab 300 mg every 4 weeks may be effective for short-term control in patients who experience psoriasis flares. Although guselkumab has a half-life of approximately 15 to 18 days, its effects may extend beyond 4 to 5 half-lives, as improvement was seen in our case for 57 to 180 days. Although we did not test for the development of antidrug antibodies to secukinumab, there is evidence that the incidence of such development is exceedingly low and not correlated with clinical efficacy.2 We cannot exclude the possibility that the single dose of guselkumab may have been responsible for the entirety of our patient’s improvement, as up to 46% of patients treated with 12 months of guselkumab continued to experience remission at 6 months after discontinuation.3 Population data suggest a longer duration of activity or disease-modifying potential of guselkumab; however, affected patients had a longer duration of initial treatment before cessation.3
While data on the safety of dual cytokine inhibition in psoriasis are limited, patients in clinical trials for psoriatic arthritis often are treated with a biologic and methotrexate and/or oral corticosteroids; the safety of biologics even with such a combination is excellent.4 Combination biologic therapy with ustekinumab and etanercept in refractory psoriasis caused few adverse events.4,5 Combination tumor necrosis factor α and IL-12/23 inhibition is comparatively more immunosuppressive than combination IL-23 and IL-17 inhibition. Additionally, there may be redundancies in the immune system to the IL-23–TYK-2–IL-17 axis.1 While inhibition of both IL-17A and IL-17F increases cutaneous candidiasis risk, inhibiting IL-23 or IL-17A alone is associated with less risk for candidal infection.6,7 This suggests that redundancy via IL-17F derived from non-IL-23–dependent pathways may offer protection during dual inhibition (eFigure 2). Our patient experienced no adverse effects with a single 100-mg dose of guselkumab, suggesting that at least short-term combination therapy may be well tolerated.

The safety profile seen in our case supports forgoing a washout period when switching between biologics, which can delay access to much-needed therapy and induce flares. Our patient had no adverse events, which provides anecdotal evidence supporting initiation of new biologics without delay. It is possible that the risks of overlapping two low-risk biologics may be lower than combining one biologic with a more immunosuppressive oral immunomodulator.
Combining standard-therapy secukinumab with a one-time dose of guselkumab may provide a synergistic effect by targeting different cytokines involved in the pathogenesis of psoriasis. Using 2 monoclonal antibodies with complementary targets may offer a strategy for rapid control of severe psoriasis that is refractory to single biologics.
To the Editor:
A 23-year-old man with plaque psoriasis experienced a flare after a 3-month interruption in his secukinumab therapy. The patient’s psoriasis previously was well controlled with secukinumab 300 mg every 4 weeks. Due to social impairment from the current flare, he received a one-time 100-mg dose of sample guselkumab. Secukinumab therapy was resumed 3 weeks later at 300 mg every 4 weeks. Improvement was gradual; 3 weeks after receiving guselkumab, plaques remained on the bilateral dorsal forearms and the lower abdomen. By 2 months posttreatment, there was decreased erythema and reduced scaling with the continued secukinumab therapy (eFigure 1). The patient reported complete clearance of the plaques 4 months after receiving guselkumab but experienced a brief recurrence at month 5. By month 6, his psoriasis returned to its prior baseline of mild disease. In this 6-month period after resuming treatment, the patient reported subjective improvement in itch and quality of life. He reported no infection or other adverse effects from the additional therapy.

Psoriasis is driven by the interleukin (IL)-23–tyrosine kinase (TYK) 2–IL-17 axis.1 Guselkumab, an IL-23 inhibitor, and secukinumab, an IL-17A inhibitor, target distinct yet interconnected pathways in the pathogenesis of psoriasis.1 A key upstream regulator of the IL-17 pathway, IL-23 drives the expansion and survival of T helper 17 cells, which in turn produce IL-17A and IL-17F—cytokines involved in psoriatic inflammation.1 While monotherapy with either IL-23 or IL-17A inhibition generally is safe and highly effective, it does not always provide adequate disease control. By simultaneously inhibiting IL-23 and IL-17A, a combined approach may offer enhanced disease control compared to monotherapy.
Combined therapy with a single 100-mg dose of guselkumab and secukinumab 300 mg every 4 weeks may be effective for short-term control in patients who experience psoriasis flares. Although guselkumab has a half-life of approximately 15 to 18 days, its effects may extend beyond 4 to 5 half-lives, as improvement was seen in our case for 57 to 180 days. Although we did not test for the development of antidrug antibodies to secukinumab, there is evidence that the incidence of such development is exceedingly low and not correlated with clinical efficacy.2 We cannot exclude the possibility that the single dose of guselkumab may have been responsible for the entirety of our patient’s improvement, as up to 46% of patients treated with 12 months of guselkumab continued to experience remission at 6 months after discontinuation.3 Population data suggest a longer duration of activity or disease-modifying potential of guselkumab; however, affected patients had a longer duration of initial treatment before cessation.3
While data on the safety of dual cytokine inhibition in psoriasis are limited, patients in clinical trials for psoriatic arthritis often are treated with a biologic and methotrexate and/or oral corticosteroids; the safety of biologics even with such a combination is excellent.4 Combination biologic therapy with ustekinumab and etanercept in refractory psoriasis caused few adverse events.4,5 Combination tumor necrosis factor α and IL-12/23 inhibition is comparatively more immunosuppressive than combination IL-23 and IL-17 inhibition. Additionally, there may be redundancies in the immune system to the IL-23–TYK-2–IL-17 axis.1 While inhibition of both IL-17A and IL-17F increases cutaneous candidiasis risk, inhibiting IL-23 or IL-17A alone is associated with less risk for candidal infection.6,7 This suggests that redundancy via IL-17F derived from non-IL-23–dependent pathways may offer protection during dual inhibition (eFigure 2). Our patient experienced no adverse effects with a single 100-mg dose of guselkumab, suggesting that at least short-term combination therapy may be well tolerated.

The safety profile seen in our case supports forgoing a washout period when switching between biologics, which can delay access to much-needed therapy and induce flares. Our patient had no adverse events, which provides anecdotal evidence supporting initiation of new biologics without delay. It is possible that the risks of overlapping two low-risk biologics may be lower than combining one biologic with a more immunosuppressive oral immunomodulator.
Combining standard-therapy secukinumab with a one-time dose of guselkumab may provide a synergistic effect by targeting different cytokines involved in the pathogenesis of psoriasis. Using 2 monoclonal antibodies with complementary targets may offer a strategy for rapid control of severe psoriasis that is refractory to single biologics.
- Griffiths CEM, Armstrong AW, Gudjonsson JE, et al. Psoriasis. Lancet. 2021;397:1301-1315. doi:10.1016/S0140-6736(20)32549-6
- Tsakok T, Rispens T, Spuls P, et al. Immunogenicity of biologic therapies in psoriasis: myths, facts and a suggested approach. J Eur Acad Dermatol Venereol. 2021;35:329-337. doi:10.1111/jdv.16980
- Zhdanava M, Fitzgerald T, Pilon D, et al. Comparative analysis of persistence and remission with guselkumab versus secukinumab and ixekizumab in the United States. J Dermatolog Treat. 2024;35:1. doi:10.1080/09546634.2024.2349658
- Kumthekar A, Ashrafi M, Deodhar A. Difficult to treat psoriatic arthritis—how should we manage? Clin Rheumatol. 2023;42:2251-2265. doi:10.1007/S10067-023-06605-9
- Thibodeaux Q, Ly K, Reddy V, et al. Dual biologic therapy for recalcitrant psoriasis and psoriatic arthritis. JAAD Case Rep. 2019;5:928-930. doi:10.1016/j.jdcr.2019.08.015
- Lebwohl MG, Merola JF, Rowland K, et al. Safety of guselkumab treatment for up to 5 years in patients with moderate-to-severe psoriasis: pooled analyses across seven clinical trials with more than 8600 patient-years of exposure. Br J Dermatol. 2023;189:42-52. doi:10.1093/BJD/LJAD115
- Reich K, Warren RB, Lebwohl M, et al. Bimekizumab versus secukinumab in plaque psoriasis. N Engl J Med. 2021;385:142-152. doi:10.1056/NEJMOA2102383
- Griffiths CEM, Armstrong AW, Gudjonsson JE, et al. Psoriasis. Lancet. 2021;397:1301-1315. doi:10.1016/S0140-6736(20)32549-6
- Tsakok T, Rispens T, Spuls P, et al. Immunogenicity of biologic therapies in psoriasis: myths, facts and a suggested approach. J Eur Acad Dermatol Venereol. 2021;35:329-337. doi:10.1111/jdv.16980
- Zhdanava M, Fitzgerald T, Pilon D, et al. Comparative analysis of persistence and remission with guselkumab versus secukinumab and ixekizumab in the United States. J Dermatolog Treat. 2024;35:1. doi:10.1080/09546634.2024.2349658
- Kumthekar A, Ashrafi M, Deodhar A. Difficult to treat psoriatic arthritis—how should we manage? Clin Rheumatol. 2023;42:2251-2265. doi:10.1007/S10067-023-06605-9
- Thibodeaux Q, Ly K, Reddy V, et al. Dual biologic therapy for recalcitrant psoriasis and psoriatic arthritis. JAAD Case Rep. 2019;5:928-930. doi:10.1016/j.jdcr.2019.08.015
- Lebwohl MG, Merola JF, Rowland K, et al. Safety of guselkumab treatment for up to 5 years in patients with moderate-to-severe psoriasis: pooled analyses across seven clinical trials with more than 8600 patient-years of exposure. Br J Dermatol. 2023;189:42-52. doi:10.1093/BJD/LJAD115
- Reich K, Warren RB, Lebwohl M, et al. Bimekizumab versus secukinumab in plaque psoriasis. N Engl J Med. 2021;385:142-152. doi:10.1056/NEJMOA2102383
Combination Guselkumab and Secukinumab for Plaque Psoriasis
Combination Guselkumab and Secukinumab for Plaque Psoriasis
Practice Points
- Combination therapy with 2 complementary biologics for psoriasis may have synergistic effects.
- Washout periods likely are unnecessary when switching between guselkumab and secukinumab.
Psoriasis and Obesity: A Clinical Review of the Bidirectional Link and Management Implications
Psoriasis and Obesity: A Clinical Review of the Bidirectional Link and Management Implications
Psoriasis, a chronic immune-mediated skin disease, is increasingly recognized for its systemic inflammation and associated cardiometabolic risk. The global rise of obesity has revealed a critical link between these 2 conditions. Current evidence confirms that obesity is an independent risk factor that can trigger psoriasis onset, drive more severe disease, and substantially impair treatment efficacy.1,2 This review synthesizes the latest evidence on the shared pathophysiology, clinical consequences, and integrated management strategies for patients with both psoriasis and obesity.
Pathophysiologic Links Between Psoriasis and Obesity
Emerging evidence highlights a bidirectional relationship between psoriasis and obesity rooted in overlapping inflammatory pathways. Both conditions are characterized by chronic inflammation mediated by cytokines that sustain systemic immune activation and metabolic dysfunction. This interplay creates a reciprocal process in which psoriatic inflammation promotes metabolic disturbances while obesity amplifies systemic inflammation and disease severity.3
Psoriasis may contribute to obesity through cytokine-driven metabolic alterations in insulin signaling and adipocyte function. The psoriatic immune response is dominated by T helper (Th) 1, Th17, and Th22 cell activation, leading to elevated levels of interferon-γ, tumor necrosis factor (TNF) α, interleukin (IL) 6, IL-17, and IL-22 from keratinocytes.4 These cytokines contribute not only to cutaneous inflammation but also to insulin resistance and adipocyte dysfunction.5 Tumor necrosis factor α and IL-6 interfere with insulin signaling via activation of stress kinases (eg, IκB kinase and c-Jun N-terminal kinase), implicating these molecules in insulin resistance and weight gain.6 Moreover, IL-17, a central cytokine in psoriasis, has been implicated in vascular inflammation, insulin resistance, and type 2 diabetes, suggesting a mechanistic link between psoriatic inflammation and metabolic disease.5 Additionally, chronic systemic inflammation in psoriasis suppresses adiponectin, a protective adipokine that enhances insulin sensitivity and exerts anti-inflammatory effects by inhibiting TNF-α and IL-6 production while promoting IL-10 synthesis. Reduced adiponectin levels have consistently been observed in patients with psoriasis and concomitant obesity or metabolic syndrome.3 The resultant imbalance between proinflammatory and anti-inflammatory mediators creates a metabolic environment conducive to obesity.3
Conversely, obesity itself may intensify both the incidence and severity of psoriasis through shared inflammatory pathways. Leptin, whose expression rises proportionally with adipocyte mass, acts as a proinflammatory mediator linking obesity to psoriasis exacerbation. By promoting Th1 and Th17 cell differentiation and suppressing regulatory T-cell activity, leptin increases IL-17A, IL-6, and TNF-α production.7 These cytokines stimulate keratinocyte proliferation and perpetuate cutaneous inflammation, thereby intensifying disease activity. Similarly, resistin, another adipokine that is elevated in obesity, stimulates monocytes and macrophages to secrete TNF-α and IL-6, creating a proinflammatory state in the body that drives the relationship between excessive fat storage (adiposity) and the development and severity of psoriasis.8 In contrast, reduced adiponectin levels in obesity remove a key anti-inflammatory regulator that normally inhibits TNF-α and IL-6 synthesis and promotes IL-10 production. This deficiency provokes unrestrained cytokine activation within both adipose and cutaneous tissue, exacerbating psoriatic immune dysregulation.9
Free fatty acids derived from abundant adipocytes in obesity further activate nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB)–signaling and induce oxidative stress, augmenting systemic inflammation. Adipose tissue macrophages additionally secrete IL-1Β, IL-6, and TNF-α, which promote keratinocyte proliferation and amplify the psoriatic inflammatory cascade. Importantly, anti–TNF-α therapy has been shown to improve metabolic parameters, reducing low-density lipoprotein and total cholesterol levels and enhancing insulin sensitivity, highlighting the reciprocal benefits of dampening the inflammatory signaling from TNF-α.5
Together, these findings highlight that psoriasis and obesity are interconnected inflammatory disorders driven by overlapping cytokine networks, most notably TNF-α, IL-6, IL-17, and IL-23, and by imbalances in adipokines such as leptin, resistin, and adiponectin (Table).
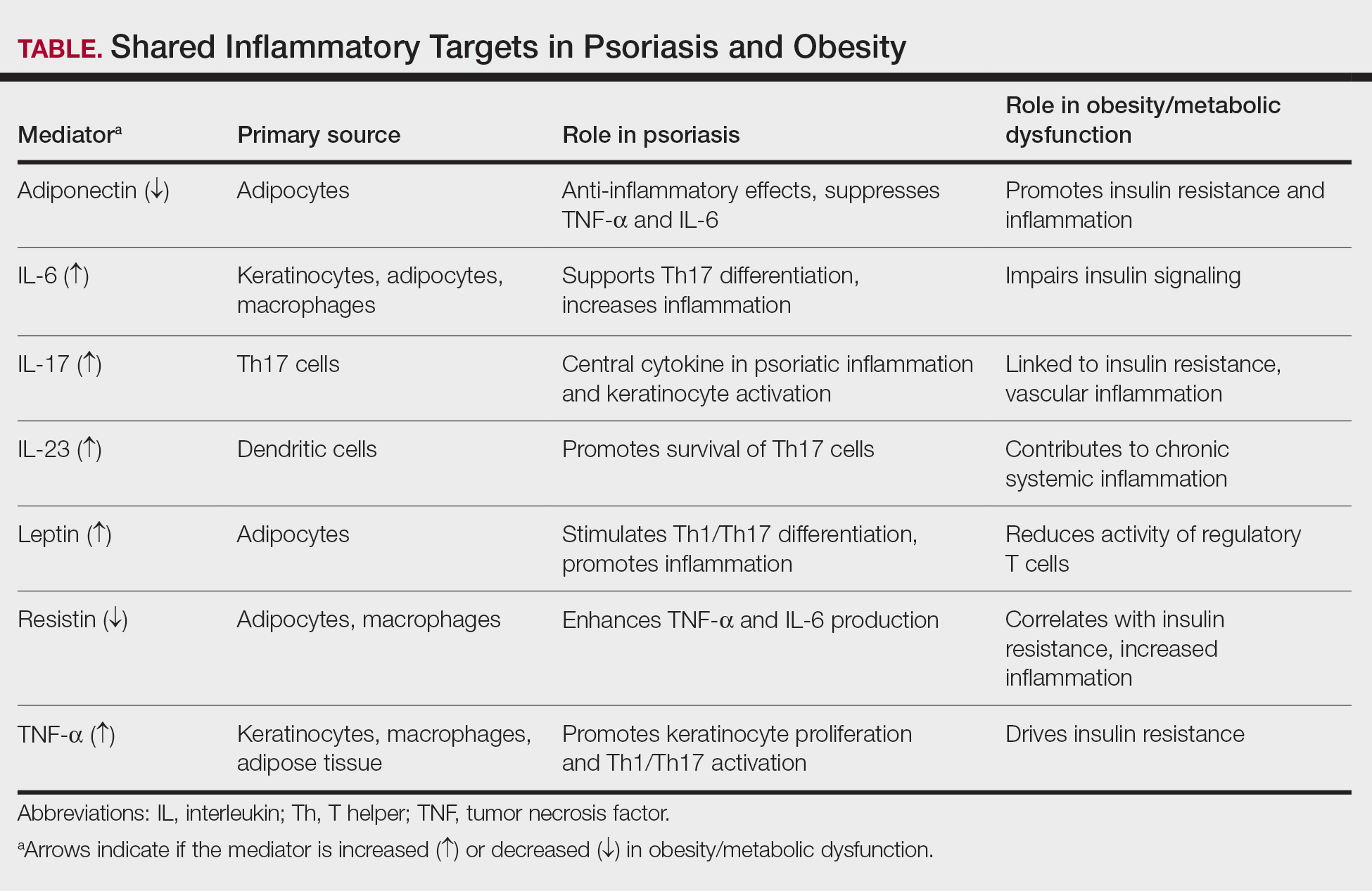
Clinical Implications of Obesity-Related Comorbidities
Higher body mass index (BMI) has been associated with an increased and earlier incidence of psoriasis. When treated as continuous variables, both Psoriasis Area and Severity Index (PASI) and Dermatology Life Quality Index scores were positively correlated with increases in BMI.10,11 This close association suggests BMI may function as a practical indicator of disease severity and prognosis.7,8,11
Psoriatic arthritis (PsA) is a major comorbidity of psoriasis that impacts disease severity and quality of life. Obesity is associated with an increased risk for PsA after accounting for traditional risk factors. Psoriatic arthritis can cause chronic mobility issues and complicate a patient’s ability to stay active. Early rheumatologist involvement may be warranted to prevent PsA development in patients with obesity and psoriasis.11,12
Patients with psoriasis have an increased baseline risk for metabolic syndrome, including obesity, type 2 diabetes, hypertension, and dyslipidemia11,13,14; therefore, the presence of obesity warrants regular metabolic evaluation. Unmanaged metabolic syndrome contributes to the increased prevalence of myocardial infarction, stroke, and heart failure seen with psoriasis.13,14 Obesity also may cause obstructive sleep apnea, which can exacerbate hypertension and heart disease due to chronic hypoxia.11 The interplay of these metabolic factors puts patients with psoriasis and obesity at heightened cardiovascular risk.13,14
Both psoriasis and obesity present psychosocial challenges such as elevated rates of depression, anxiety, and body-image concerns, all of which become more pronounced when these conditions coexist. These psychological stressors may hinder a patient’s motivation for lifestyle changes or treatment adherence.13
Dermatology visits serve as an important opportunity to monitor obesity-related comorbidities.13,15 Dermatologists are uniquely positioned to initiate metabolic screening while collaborating with primary care physicians for ongoing cardiometabolic management. Metabolic and cardiovascular baselines should be measured when care is established and should be followed longitudinally—these include BMI, waist circumference measurements, blood pressure, lipid panels, fasting glucose or A1C levels, and liver enzymes.10,14,15 Regular screening for depression, suicidality, and disordered eating also is encouraged.13 Clinicians should follow established guidelines to identify and manage metabolic, cardiovascular, and psychological comorbidities.13
Impact of Obesity on Psoriasis Treatment
Obesity is a critical factor in clinical decision-making, as it consistently is associated with diminished response to numerous systemic psoriasis therapies. This reduced efficacy has been observed with conventional oral agents such as methotrexate and cyclosporine and is particularly well documented in the context of biologic therapies.15,16 Several meta-analyses and large real-world studies have shown that higher BMI is associated with a suboptimal treatment response, with patients in higher BMI categories achieving lower rates of PASI 75 and PASI 90 than their nonobese counterparts receiving the same regimen.15,17
This efficacy gap often is rooted in pharmacokinetic challenges. For many biologics administered via subcutaneous injection, increased BMI can lead to altered drug distribution, such as sequestration in adipose tissue. Altered distribution combined with potentially increased drug clearance can result in lower overall serum drug concentrations and subsequent underdosing for a patient’s inflammatory burden.15 This reality highlights the important distinction between fixed and weight-based dosing strategies. Therapies dosed by weight, such as infliximab, have demonstrated more consistent efficacy in populations with obesity, as the dose is escalated to match patient size.18 Despite the weight-dependent effect, recent real-world studies have suggested that fixed dosing of some IL-17A inhibitors (eg, ixekizumab) remains highly effective across BMI categories, while others (eg, secukinumab) show diminished efficacy in obese patients.16,19 Furthermore, some real-world studies have reported an inverse relationship between elevated BMI and efficacy of IL-23 inhibitors, particularly guselkumab, while other studies reported no association.20,21 These mixed observations support a nuanced interpretation of BMI’s role in treatment modification; consideration should be given to the specific medication and the dosing strategy over biologic class alone.
Whether obesity independently drives psoriasis severity or mainly diminishes treatment efficacy through pharmacokinetic effects remains debated. Observational studies show a dose-dependent relationship between BMI and disease severity even in untreated patients, supporting a proinflammatory role for obesity.10 Concurrently, higher BMI may predict lower responses to some fixed-dose biologics, likely due to altered distribution and sequestration in adipose tissue.15,20 The precise contributions are challenging to delineate; both likely converge to heighten disease severity and reduce treatment response.
Beyond efficacy, obesity compounds the safety considerations of systemic treatments. Psoriasis and obesity are both strong independent risk factors for metabolic dysfunction–associated steatotic liver disease. This prevalent comorbidity creates a substantial clinical dilemma, as a first-line, cost-effective agent such as methotrexate carries a known risk for hepatotoxicity, which is amplified in patients with pre-existing liver steatosis.13 Consequently, the presence of obesity and metabolic dysfunction–associated steatotic liver disease often limits the use of methotrexate, forcing a change in therapy. Thorough baseline comorbidity screening in all patients with psoriasis and obesity is necessary to select a therapy that balances efficacy with safety.13,14
Management Strategies
Weight management plays a pivotal role in improving psoriasis outcomes. Even modest weight loss of 5% to 10% has been shown in randomized and observational studies to substantially reduce disease severity, reflected by lower PASI and Dermatology Life Quality Index scores and enhance treatment responsiveness.22,23 Dietary approaches emphasizing hypocaloric, Mediterranean, or anti-inflammatory patterns have demonstrated additional improvements in disease activity, likely through attenuation of systemic inflammation and metabolic dysregulation.21 Bariatric surgery provides more sustained benefits, with multiple studies reporting long-term remission or reduced psoriasis severity following substantial postoperative weight loss.23,24
Pharmacologic weight-loss therapies, particularly glucagonlike peptide-1 receptor agonists, have emerged as potential adjuncts in psoriasis management. Although data remain limited, these agents may reduce systemic inflammation, improve insulin sensitivity, and indirectly enhance biologic response.25
Conclusion
Psoriasis and obesity are interconnected chronic inflammatory conditions that share overlapping cytokine pathways and mutually exacerbate the clinical course. Systemic inflammation driven by cytokines such as TNF-α, IL-6, and IL-17 not only promotes psoriatic skin disease but also contributes to metabolic dysfunction and cardiovascular risk. In turn, excess adiposity amplifies inflammatory signaling and diminishes therapeutic response, creating a self-perpetuating cycle of disease.
Dermatologists should identify obesity-related risks early; counsel patients on lifestyle changes; initiate metabolic screening; and coordinate care across primary care, nutrition, and rheumatology. Regular screening for metabolic syndrome, cardiovascular comorbidities, and psychosocial distress should be integrated into psoriasis management. Future research should focus on personalized treatment strategies that integrate management of inflammatory skin disease with underlying metabolic health, such as optimizing biologic dosing and identifying novel targets that disrupt the pathophysiologic loop. By recognizing and addressing the shared inflammatory mechanisms of psoriasis and obesity, clinicians can improve both dermatologic and systemic outcomes for affected patients.
- Barrea L, Muscogiuri G, Annunziata G, et al. Update on obesity in psoriasis patients: narrative review and practical insights. Clin Cosmet Investig Dermatol. 2023;16:3089-3104.
- Owczarczyk-Saczonek A, Gornowicz-Porowska J, Zegarska B. Psoriasis comorbidities: obesity, diet, and metabolic syndrome. Int J Mol Sci. 2024;25:1832.
- Vata D, Tarcau BM, Popescu IA, et al. Update on obesity in psoriasis patients. Life (Basel). 2023;13:1947.
- Piaserico S, Orlando G, Messina F. Psoriasis and cardiometabolic diseases: shared genetic and molecular pathways. Int J Mol Sci. 2022;23:9063.
- Hao Y, Zhu YJ, Zou S, et al. Metabolic syndrome and psoriasis: mechanisms and future directions. Front Immunol. 2021;12:711060.
- Kern L, Mittenbühler MJ, Vesting AJ, et al. Obesity-induced TNF-α and IL-6 signaling: the missing link between obesity and inflammation-driven liver and colorectal cancers. Cancers (Basel). 2019;11:24.
- Hwang J, Yoo JA, Yoon H, et al. Role of leptin in the association between obesity and psoriasis. Biomol Ther (Seoul). 2021;29:11-21.
- Smith B, Devjani S, Collier MR, et al. Association between psoriasis and obesity among US adults in the 2009-2014 National Health and Nutrition Examination Survey. Cutis. 2023;112:49-51. doi:10.12788/cutis.0807
- Ellulu MS, Patimah I, Khaza’ai H. Obesity and inflammation: the linking mechanism and the complications. Arch Med Sci. 2017;13:851-863.
- Wang H, Hou S, Kang X, et al. BMI matters: understanding the link between weight and severe psoriasis. Sci Rep. 2025;15:11158.
- Norden A, Rekhtman S, Strunk A, et al. Risk of psoriasis according to body mass index: a retrospective cohort analysis. J Am Acad Dermatol. 2022;86:1020-1026.
- Di Caprio R, Nigro E, Di Brizzi EV, et al. Exploring the link between psoriasis and adipose tissue: one amplifies the other. Int J Mol Sci. 2024;25:13435.
- Elmets CA, Leonardi CL, Davis DMR, et al. Joint AAD-NPF guidelines of care for the management and treatment of psoriasis with awareness and attention to comorbidities. J Am Acad Dermatol. 2019;80:1073-1113.
- Secchiero P, Rimondi E, Marcuzzi A, et al. Metabolic syndrome and psoriasis: pivotal roles of chronic inflammation and gut microbiota. Int J Mol Sci. 2024;25:8098.
- Burshtein J, Armstrong A, Chow M, et al. Association between obesity and efficacy of psoriasis therapies: an expert consensus panel. J Am Acad Dermatol. 2025;92:807-815. doi:10.1016/j.jaad.2024.12.016
- Pirro F, Caldarola G, Chiricozzi A, et al. Impact of body mass index on the efficacy of biological therapies in patients with psoriasis: a real-world study. Clin Drug Investig. 2021;41:917-925.
- Hjort G, Schwarz CW, Skov L, et al. Clinical characteristics associated with response to biologics in the treatment of psoriasis: a meta-analysis. JAMA Dermatol. 2024;160:830-837.
- Naldi L, Chimenti S, Girolomoni G, et al. Efficacy and safety of infliximab in obese and non-obese patients with plaque-type psoriasis: subanalysis of the EXPRESS II trial. Br J Dermatol. 2008;159:761-766.
- Puig L, Thom H, Mollon P, et al. Effect of body weight on the efficacy of biologics in moderate-to-severe plaque psoriasis: a systematic review and meta-analysis. J Eur Acad Dermatol Venereol. 2020;34:237-245.
- Dai M, Jiang Y, Wang Y, et al. Differential clinical factors influencing the effectiveness of distinct biologic agents in psoriasis: insights from a prospective cohort study in China. Inflamm Res. 2026;75:25. doi:10.1007/s00011-025-02179-1
- Ricceri F, Chiricozzi A, Peris K, et al. Successful use of anti–IL-23 molecules in overweight-to-obese psoriatic patients: a multicentric retrospective study. Dermatol Ther. 2022;35:E15793. doi:10.1111/dth.15793
- Jensen P, Zachariae C, Christensen R, et al. Effect of weight loss on the severity of psoriasis: a randomized clinical study. Br J Dermatol. 2013;168:319-327.
- Hossler EW, Wood GC, Still CD, et al. Psoriasis improvement following bariatric surgery is durable: 5-year follow-up in the Geisinger bariatric surgery cohort. Obes Surg. 2020;30:3350-3356.
- Romero-Talamás H, Daigle CR, Aminian A, et al. Psoriasis improvement after bariatric surgery. Surg Obes Relat Dis. 2014;10:1155-1159.
- Buonanno S, Gaggiano C, Terribili R, et al. Potential role of GLP-1 receptor agonists in the management of psoriatic disease: a scoping review. Inflamm Res. 2025;74:167. doi:10.1007/s00011-025-02140-2
Psoriasis, a chronic immune-mediated skin disease, is increasingly recognized for its systemic inflammation and associated cardiometabolic risk. The global rise of obesity has revealed a critical link between these 2 conditions. Current evidence confirms that obesity is an independent risk factor that can trigger psoriasis onset, drive more severe disease, and substantially impair treatment efficacy.1,2 This review synthesizes the latest evidence on the shared pathophysiology, clinical consequences, and integrated management strategies for patients with both psoriasis and obesity.
Pathophysiologic Links Between Psoriasis and Obesity
Emerging evidence highlights a bidirectional relationship between psoriasis and obesity rooted in overlapping inflammatory pathways. Both conditions are characterized by chronic inflammation mediated by cytokines that sustain systemic immune activation and metabolic dysfunction. This interplay creates a reciprocal process in which psoriatic inflammation promotes metabolic disturbances while obesity amplifies systemic inflammation and disease severity.3
Psoriasis may contribute to obesity through cytokine-driven metabolic alterations in insulin signaling and adipocyte function. The psoriatic immune response is dominated by T helper (Th) 1, Th17, and Th22 cell activation, leading to elevated levels of interferon-γ, tumor necrosis factor (TNF) α, interleukin (IL) 6, IL-17, and IL-22 from keratinocytes.4 These cytokines contribute not only to cutaneous inflammation but also to insulin resistance and adipocyte dysfunction.5 Tumor necrosis factor α and IL-6 interfere with insulin signaling via activation of stress kinases (eg, IκB kinase and c-Jun N-terminal kinase), implicating these molecules in insulin resistance and weight gain.6 Moreover, IL-17, a central cytokine in psoriasis, has been implicated in vascular inflammation, insulin resistance, and type 2 diabetes, suggesting a mechanistic link between psoriatic inflammation and metabolic disease.5 Additionally, chronic systemic inflammation in psoriasis suppresses adiponectin, a protective adipokine that enhances insulin sensitivity and exerts anti-inflammatory effects by inhibiting TNF-α and IL-6 production while promoting IL-10 synthesis. Reduced adiponectin levels have consistently been observed in patients with psoriasis and concomitant obesity or metabolic syndrome.3 The resultant imbalance between proinflammatory and anti-inflammatory mediators creates a metabolic environment conducive to obesity.3
Conversely, obesity itself may intensify both the incidence and severity of psoriasis through shared inflammatory pathways. Leptin, whose expression rises proportionally with adipocyte mass, acts as a proinflammatory mediator linking obesity to psoriasis exacerbation. By promoting Th1 and Th17 cell differentiation and suppressing regulatory T-cell activity, leptin increases IL-17A, IL-6, and TNF-α production.7 These cytokines stimulate keratinocyte proliferation and perpetuate cutaneous inflammation, thereby intensifying disease activity. Similarly, resistin, another adipokine that is elevated in obesity, stimulates monocytes and macrophages to secrete TNF-α and IL-6, creating a proinflammatory state in the body that drives the relationship between excessive fat storage (adiposity) and the development and severity of psoriasis.8 In contrast, reduced adiponectin levels in obesity remove a key anti-inflammatory regulator that normally inhibits TNF-α and IL-6 synthesis and promotes IL-10 production. This deficiency provokes unrestrained cytokine activation within both adipose and cutaneous tissue, exacerbating psoriatic immune dysregulation.9
Free fatty acids derived from abundant adipocytes in obesity further activate nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB)–signaling and induce oxidative stress, augmenting systemic inflammation. Adipose tissue macrophages additionally secrete IL-1Β, IL-6, and TNF-α, which promote keratinocyte proliferation and amplify the psoriatic inflammatory cascade. Importantly, anti–TNF-α therapy has been shown to improve metabolic parameters, reducing low-density lipoprotein and total cholesterol levels and enhancing insulin sensitivity, highlighting the reciprocal benefits of dampening the inflammatory signaling from TNF-α.5
Together, these findings highlight that psoriasis and obesity are interconnected inflammatory disorders driven by overlapping cytokine networks, most notably TNF-α, IL-6, IL-17, and IL-23, and by imbalances in adipokines such as leptin, resistin, and adiponectin (Table).

Clinical Implications of Obesity-Related Comorbidities
Higher body mass index (BMI) has been associated with an increased and earlier incidence of psoriasis. When treated as continuous variables, both Psoriasis Area and Severity Index (PASI) and Dermatology Life Quality Index scores were positively correlated with increases in BMI.10,11 This close association suggests BMI may function as a practical indicator of disease severity and prognosis.7,8,11
Psoriatic arthritis (PsA) is a major comorbidity of psoriasis that impacts disease severity and quality of life. Obesity is associated with an increased risk for PsA after accounting for traditional risk factors. Psoriatic arthritis can cause chronic mobility issues and complicate a patient’s ability to stay active. Early rheumatologist involvement may be warranted to prevent PsA development in patients with obesity and psoriasis.11,12
Patients with psoriasis have an increased baseline risk for metabolic syndrome, including obesity, type 2 diabetes, hypertension, and dyslipidemia11,13,14; therefore, the presence of obesity warrants regular metabolic evaluation. Unmanaged metabolic syndrome contributes to the increased prevalence of myocardial infarction, stroke, and heart failure seen with psoriasis.13,14 Obesity also may cause obstructive sleep apnea, which can exacerbate hypertension and heart disease due to chronic hypoxia.11 The interplay of these metabolic factors puts patients with psoriasis and obesity at heightened cardiovascular risk.13,14
Both psoriasis and obesity present psychosocial challenges such as elevated rates of depression, anxiety, and body-image concerns, all of which become more pronounced when these conditions coexist. These psychological stressors may hinder a patient’s motivation for lifestyle changes or treatment adherence.13
Dermatology visits serve as an important opportunity to monitor obesity-related comorbidities.13,15 Dermatologists are uniquely positioned to initiate metabolic screening while collaborating with primary care physicians for ongoing cardiometabolic management. Metabolic and cardiovascular baselines should be measured when care is established and should be followed longitudinally—these include BMI, waist circumference measurements, blood pressure, lipid panels, fasting glucose or A1C levels, and liver enzymes.10,14,15 Regular screening for depression, suicidality, and disordered eating also is encouraged.13 Clinicians should follow established guidelines to identify and manage metabolic, cardiovascular, and psychological comorbidities.13
Impact of Obesity on Psoriasis Treatment
Obesity is a critical factor in clinical decision-making, as it consistently is associated with diminished response to numerous systemic psoriasis therapies. This reduced efficacy has been observed with conventional oral agents such as methotrexate and cyclosporine and is particularly well documented in the context of biologic therapies.15,16 Several meta-analyses and large real-world studies have shown that higher BMI is associated with a suboptimal treatment response, with patients in higher BMI categories achieving lower rates of PASI 75 and PASI 90 than their nonobese counterparts receiving the same regimen.15,17
This efficacy gap often is rooted in pharmacokinetic challenges. For many biologics administered via subcutaneous injection, increased BMI can lead to altered drug distribution, such as sequestration in adipose tissue. Altered distribution combined with potentially increased drug clearance can result in lower overall serum drug concentrations and subsequent underdosing for a patient’s inflammatory burden.15 This reality highlights the important distinction between fixed and weight-based dosing strategies. Therapies dosed by weight, such as infliximab, have demonstrated more consistent efficacy in populations with obesity, as the dose is escalated to match patient size.18 Despite the weight-dependent effect, recent real-world studies have suggested that fixed dosing of some IL-17A inhibitors (eg, ixekizumab) remains highly effective across BMI categories, while others (eg, secukinumab) show diminished efficacy in obese patients.16,19 Furthermore, some real-world studies have reported an inverse relationship between elevated BMI and efficacy of IL-23 inhibitors, particularly guselkumab, while other studies reported no association.20,21 These mixed observations support a nuanced interpretation of BMI’s role in treatment modification; consideration should be given to the specific medication and the dosing strategy over biologic class alone.
Whether obesity independently drives psoriasis severity or mainly diminishes treatment efficacy through pharmacokinetic effects remains debated. Observational studies show a dose-dependent relationship between BMI and disease severity even in untreated patients, supporting a proinflammatory role for obesity.10 Concurrently, higher BMI may predict lower responses to some fixed-dose biologics, likely due to altered distribution and sequestration in adipose tissue.15,20 The precise contributions are challenging to delineate; both likely converge to heighten disease severity and reduce treatment response.
Beyond efficacy, obesity compounds the safety considerations of systemic treatments. Psoriasis and obesity are both strong independent risk factors for metabolic dysfunction–associated steatotic liver disease. This prevalent comorbidity creates a substantial clinical dilemma, as a first-line, cost-effective agent such as methotrexate carries a known risk for hepatotoxicity, which is amplified in patients with pre-existing liver steatosis.13 Consequently, the presence of obesity and metabolic dysfunction–associated steatotic liver disease often limits the use of methotrexate, forcing a change in therapy. Thorough baseline comorbidity screening in all patients with psoriasis and obesity is necessary to select a therapy that balances efficacy with safety.13,14
Management Strategies
Weight management plays a pivotal role in improving psoriasis outcomes. Even modest weight loss of 5% to 10% has been shown in randomized and observational studies to substantially reduce disease severity, reflected by lower PASI and Dermatology Life Quality Index scores and enhance treatment responsiveness.22,23 Dietary approaches emphasizing hypocaloric, Mediterranean, or anti-inflammatory patterns have demonstrated additional improvements in disease activity, likely through attenuation of systemic inflammation and metabolic dysregulation.21 Bariatric surgery provides more sustained benefits, with multiple studies reporting long-term remission or reduced psoriasis severity following substantial postoperative weight loss.23,24
Pharmacologic weight-loss therapies, particularly glucagonlike peptide-1 receptor agonists, have emerged as potential adjuncts in psoriasis management. Although data remain limited, these agents may reduce systemic inflammation, improve insulin sensitivity, and indirectly enhance biologic response.25
Conclusion
Psoriasis and obesity are interconnected chronic inflammatory conditions that share overlapping cytokine pathways and mutually exacerbate the clinical course. Systemic inflammation driven by cytokines such as TNF-α, IL-6, and IL-17 not only promotes psoriatic skin disease but also contributes to metabolic dysfunction and cardiovascular risk. In turn, excess adiposity amplifies inflammatory signaling and diminishes therapeutic response, creating a self-perpetuating cycle of disease.
Dermatologists should identify obesity-related risks early; counsel patients on lifestyle changes; initiate metabolic screening; and coordinate care across primary care, nutrition, and rheumatology. Regular screening for metabolic syndrome, cardiovascular comorbidities, and psychosocial distress should be integrated into psoriasis management. Future research should focus on personalized treatment strategies that integrate management of inflammatory skin disease with underlying metabolic health, such as optimizing biologic dosing and identifying novel targets that disrupt the pathophysiologic loop. By recognizing and addressing the shared inflammatory mechanisms of psoriasis and obesity, clinicians can improve both dermatologic and systemic outcomes for affected patients.
Psoriasis, a chronic immune-mediated skin disease, is increasingly recognized for its systemic inflammation and associated cardiometabolic risk. The global rise of obesity has revealed a critical link between these 2 conditions. Current evidence confirms that obesity is an independent risk factor that can trigger psoriasis onset, drive more severe disease, and substantially impair treatment efficacy.1,2 This review synthesizes the latest evidence on the shared pathophysiology, clinical consequences, and integrated management strategies for patients with both psoriasis and obesity.
Pathophysiologic Links Between Psoriasis and Obesity
Emerging evidence highlights a bidirectional relationship between psoriasis and obesity rooted in overlapping inflammatory pathways. Both conditions are characterized by chronic inflammation mediated by cytokines that sustain systemic immune activation and metabolic dysfunction. This interplay creates a reciprocal process in which psoriatic inflammation promotes metabolic disturbances while obesity amplifies systemic inflammation and disease severity.3
Psoriasis may contribute to obesity through cytokine-driven metabolic alterations in insulin signaling and adipocyte function. The psoriatic immune response is dominated by T helper (Th) 1, Th17, and Th22 cell activation, leading to elevated levels of interferon-γ, tumor necrosis factor (TNF) α, interleukin (IL) 6, IL-17, and IL-22 from keratinocytes.4 These cytokines contribute not only to cutaneous inflammation but also to insulin resistance and adipocyte dysfunction.5 Tumor necrosis factor α and IL-6 interfere with insulin signaling via activation of stress kinases (eg, IκB kinase and c-Jun N-terminal kinase), implicating these molecules in insulin resistance and weight gain.6 Moreover, IL-17, a central cytokine in psoriasis, has been implicated in vascular inflammation, insulin resistance, and type 2 diabetes, suggesting a mechanistic link between psoriatic inflammation and metabolic disease.5 Additionally, chronic systemic inflammation in psoriasis suppresses adiponectin, a protective adipokine that enhances insulin sensitivity and exerts anti-inflammatory effects by inhibiting TNF-α and IL-6 production while promoting IL-10 synthesis. Reduced adiponectin levels have consistently been observed in patients with psoriasis and concomitant obesity or metabolic syndrome.3 The resultant imbalance between proinflammatory and anti-inflammatory mediators creates a metabolic environment conducive to obesity.3
Conversely, obesity itself may intensify both the incidence and severity of psoriasis through shared inflammatory pathways. Leptin, whose expression rises proportionally with adipocyte mass, acts as a proinflammatory mediator linking obesity to psoriasis exacerbation. By promoting Th1 and Th17 cell differentiation and suppressing regulatory T-cell activity, leptin increases IL-17A, IL-6, and TNF-α production.7 These cytokines stimulate keratinocyte proliferation and perpetuate cutaneous inflammation, thereby intensifying disease activity. Similarly, resistin, another adipokine that is elevated in obesity, stimulates monocytes and macrophages to secrete TNF-α and IL-6, creating a proinflammatory state in the body that drives the relationship between excessive fat storage (adiposity) and the development and severity of psoriasis.8 In contrast, reduced adiponectin levels in obesity remove a key anti-inflammatory regulator that normally inhibits TNF-α and IL-6 synthesis and promotes IL-10 production. This deficiency provokes unrestrained cytokine activation within both adipose and cutaneous tissue, exacerbating psoriatic immune dysregulation.9
Free fatty acids derived from abundant adipocytes in obesity further activate nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB)–signaling and induce oxidative stress, augmenting systemic inflammation. Adipose tissue macrophages additionally secrete IL-1Β, IL-6, and TNF-α, which promote keratinocyte proliferation and amplify the psoriatic inflammatory cascade. Importantly, anti–TNF-α therapy has been shown to improve metabolic parameters, reducing low-density lipoprotein and total cholesterol levels and enhancing insulin sensitivity, highlighting the reciprocal benefits of dampening the inflammatory signaling from TNF-α.5
Together, these findings highlight that psoriasis and obesity are interconnected inflammatory disorders driven by overlapping cytokine networks, most notably TNF-α, IL-6, IL-17, and IL-23, and by imbalances in adipokines such as leptin, resistin, and adiponectin (Table).

Clinical Implications of Obesity-Related Comorbidities
Higher body mass index (BMI) has been associated with an increased and earlier incidence of psoriasis. When treated as continuous variables, both Psoriasis Area and Severity Index (PASI) and Dermatology Life Quality Index scores were positively correlated with increases in BMI.10,11 This close association suggests BMI may function as a practical indicator of disease severity and prognosis.7,8,11
Psoriatic arthritis (PsA) is a major comorbidity of psoriasis that impacts disease severity and quality of life. Obesity is associated with an increased risk for PsA after accounting for traditional risk factors. Psoriatic arthritis can cause chronic mobility issues and complicate a patient’s ability to stay active. Early rheumatologist involvement may be warranted to prevent PsA development in patients with obesity and psoriasis.11,12
Patients with psoriasis have an increased baseline risk for metabolic syndrome, including obesity, type 2 diabetes, hypertension, and dyslipidemia11,13,14; therefore, the presence of obesity warrants regular metabolic evaluation. Unmanaged metabolic syndrome contributes to the increased prevalence of myocardial infarction, stroke, and heart failure seen with psoriasis.13,14 Obesity also may cause obstructive sleep apnea, which can exacerbate hypertension and heart disease due to chronic hypoxia.11 The interplay of these metabolic factors puts patients with psoriasis and obesity at heightened cardiovascular risk.13,14
Both psoriasis and obesity present psychosocial challenges such as elevated rates of depression, anxiety, and body-image concerns, all of which become more pronounced when these conditions coexist. These psychological stressors may hinder a patient’s motivation for lifestyle changes or treatment adherence.13
Dermatology visits serve as an important opportunity to monitor obesity-related comorbidities.13,15 Dermatologists are uniquely positioned to initiate metabolic screening while collaborating with primary care physicians for ongoing cardiometabolic management. Metabolic and cardiovascular baselines should be measured when care is established and should be followed longitudinally—these include BMI, waist circumference measurements, blood pressure, lipid panels, fasting glucose or A1C levels, and liver enzymes.10,14,15 Regular screening for depression, suicidality, and disordered eating also is encouraged.13 Clinicians should follow established guidelines to identify and manage metabolic, cardiovascular, and psychological comorbidities.13
Impact of Obesity on Psoriasis Treatment
Obesity is a critical factor in clinical decision-making, as it consistently is associated with diminished response to numerous systemic psoriasis therapies. This reduced efficacy has been observed with conventional oral agents such as methotrexate and cyclosporine and is particularly well documented in the context of biologic therapies.15,16 Several meta-analyses and large real-world studies have shown that higher BMI is associated with a suboptimal treatment response, with patients in higher BMI categories achieving lower rates of PASI 75 and PASI 90 than their nonobese counterparts receiving the same regimen.15,17
This efficacy gap often is rooted in pharmacokinetic challenges. For many biologics administered via subcutaneous injection, increased BMI can lead to altered drug distribution, such as sequestration in adipose tissue. Altered distribution combined with potentially increased drug clearance can result in lower overall serum drug concentrations and subsequent underdosing for a patient’s inflammatory burden.15 This reality highlights the important distinction between fixed and weight-based dosing strategies. Therapies dosed by weight, such as infliximab, have demonstrated more consistent efficacy in populations with obesity, as the dose is escalated to match patient size.18 Despite the weight-dependent effect, recent real-world studies have suggested that fixed dosing of some IL-17A inhibitors (eg, ixekizumab) remains highly effective across BMI categories, while others (eg, secukinumab) show diminished efficacy in obese patients.16,19 Furthermore, some real-world studies have reported an inverse relationship between elevated BMI and efficacy of IL-23 inhibitors, particularly guselkumab, while other studies reported no association.20,21 These mixed observations support a nuanced interpretation of BMI’s role in treatment modification; consideration should be given to the specific medication and the dosing strategy over biologic class alone.
Whether obesity independently drives psoriasis severity or mainly diminishes treatment efficacy through pharmacokinetic effects remains debated. Observational studies show a dose-dependent relationship between BMI and disease severity even in untreated patients, supporting a proinflammatory role for obesity.10 Concurrently, higher BMI may predict lower responses to some fixed-dose biologics, likely due to altered distribution and sequestration in adipose tissue.15,20 The precise contributions are challenging to delineate; both likely converge to heighten disease severity and reduce treatment response.
Beyond efficacy, obesity compounds the safety considerations of systemic treatments. Psoriasis and obesity are both strong independent risk factors for metabolic dysfunction–associated steatotic liver disease. This prevalent comorbidity creates a substantial clinical dilemma, as a first-line, cost-effective agent such as methotrexate carries a known risk for hepatotoxicity, which is amplified in patients with pre-existing liver steatosis.13 Consequently, the presence of obesity and metabolic dysfunction–associated steatotic liver disease often limits the use of methotrexate, forcing a change in therapy. Thorough baseline comorbidity screening in all patients with psoriasis and obesity is necessary to select a therapy that balances efficacy with safety.13,14
Management Strategies
Weight management plays a pivotal role in improving psoriasis outcomes. Even modest weight loss of 5% to 10% has been shown in randomized and observational studies to substantially reduce disease severity, reflected by lower PASI and Dermatology Life Quality Index scores and enhance treatment responsiveness.22,23 Dietary approaches emphasizing hypocaloric, Mediterranean, or anti-inflammatory patterns have demonstrated additional improvements in disease activity, likely through attenuation of systemic inflammation and metabolic dysregulation.21 Bariatric surgery provides more sustained benefits, with multiple studies reporting long-term remission or reduced psoriasis severity following substantial postoperative weight loss.23,24
Pharmacologic weight-loss therapies, particularly glucagonlike peptide-1 receptor agonists, have emerged as potential adjuncts in psoriasis management. Although data remain limited, these agents may reduce systemic inflammation, improve insulin sensitivity, and indirectly enhance biologic response.25
Conclusion
Psoriasis and obesity are interconnected chronic inflammatory conditions that share overlapping cytokine pathways and mutually exacerbate the clinical course. Systemic inflammation driven by cytokines such as TNF-α, IL-6, and IL-17 not only promotes psoriatic skin disease but also contributes to metabolic dysfunction and cardiovascular risk. In turn, excess adiposity amplifies inflammatory signaling and diminishes therapeutic response, creating a self-perpetuating cycle of disease.
Dermatologists should identify obesity-related risks early; counsel patients on lifestyle changes; initiate metabolic screening; and coordinate care across primary care, nutrition, and rheumatology. Regular screening for metabolic syndrome, cardiovascular comorbidities, and psychosocial distress should be integrated into psoriasis management. Future research should focus on personalized treatment strategies that integrate management of inflammatory skin disease with underlying metabolic health, such as optimizing biologic dosing and identifying novel targets that disrupt the pathophysiologic loop. By recognizing and addressing the shared inflammatory mechanisms of psoriasis and obesity, clinicians can improve both dermatologic and systemic outcomes for affected patients.
- Barrea L, Muscogiuri G, Annunziata G, et al. Update on obesity in psoriasis patients: narrative review and practical insights. Clin Cosmet Investig Dermatol. 2023;16:3089-3104.
- Owczarczyk-Saczonek A, Gornowicz-Porowska J, Zegarska B. Psoriasis comorbidities: obesity, diet, and metabolic syndrome. Int J Mol Sci. 2024;25:1832.
- Vata D, Tarcau BM, Popescu IA, et al. Update on obesity in psoriasis patients. Life (Basel). 2023;13:1947.
- Piaserico S, Orlando G, Messina F. Psoriasis and cardiometabolic diseases: shared genetic and molecular pathways. Int J Mol Sci. 2022;23:9063.
- Hao Y, Zhu YJ, Zou S, et al. Metabolic syndrome and psoriasis: mechanisms and future directions. Front Immunol. 2021;12:711060.
- Kern L, Mittenbühler MJ, Vesting AJ, et al. Obesity-induced TNF-α and IL-6 signaling: the missing link between obesity and inflammation-driven liver and colorectal cancers. Cancers (Basel). 2019;11:24.
- Hwang J, Yoo JA, Yoon H, et al. Role of leptin in the association between obesity and psoriasis. Biomol Ther (Seoul). 2021;29:11-21.
- Smith B, Devjani S, Collier MR, et al. Association between psoriasis and obesity among US adults in the 2009-2014 National Health and Nutrition Examination Survey. Cutis. 2023;112:49-51. doi:10.12788/cutis.0807
- Ellulu MS, Patimah I, Khaza’ai H. Obesity and inflammation: the linking mechanism and the complications. Arch Med Sci. 2017;13:851-863.
- Wang H, Hou S, Kang X, et al. BMI matters: understanding the link between weight and severe psoriasis. Sci Rep. 2025;15:11158.
- Norden A, Rekhtman S, Strunk A, et al. Risk of psoriasis according to body mass index: a retrospective cohort analysis. J Am Acad Dermatol. 2022;86:1020-1026.
- Di Caprio R, Nigro E, Di Brizzi EV, et al. Exploring the link between psoriasis and adipose tissue: one amplifies the other. Int J Mol Sci. 2024;25:13435.
- Elmets CA, Leonardi CL, Davis DMR, et al. Joint AAD-NPF guidelines of care for the management and treatment of psoriasis with awareness and attention to comorbidities. J Am Acad Dermatol. 2019;80:1073-1113.
- Secchiero P, Rimondi E, Marcuzzi A, et al. Metabolic syndrome and psoriasis: pivotal roles of chronic inflammation and gut microbiota. Int J Mol Sci. 2024;25:8098.
- Burshtein J, Armstrong A, Chow M, et al. Association between obesity and efficacy of psoriasis therapies: an expert consensus panel. J Am Acad Dermatol. 2025;92:807-815. doi:10.1016/j.jaad.2024.12.016
- Pirro F, Caldarola G, Chiricozzi A, et al. Impact of body mass index on the efficacy of biological therapies in patients with psoriasis: a real-world study. Clin Drug Investig. 2021;41:917-925.
- Hjort G, Schwarz CW, Skov L, et al. Clinical characteristics associated with response to biologics in the treatment of psoriasis: a meta-analysis. JAMA Dermatol. 2024;160:830-837.
- Naldi L, Chimenti S, Girolomoni G, et al. Efficacy and safety of infliximab in obese and non-obese patients with plaque-type psoriasis: subanalysis of the EXPRESS II trial. Br J Dermatol. 2008;159:761-766.
- Puig L, Thom H, Mollon P, et al. Effect of body weight on the efficacy of biologics in moderate-to-severe plaque psoriasis: a systematic review and meta-analysis. J Eur Acad Dermatol Venereol. 2020;34:237-245.
- Dai M, Jiang Y, Wang Y, et al. Differential clinical factors influencing the effectiveness of distinct biologic agents in psoriasis: insights from a prospective cohort study in China. Inflamm Res. 2026;75:25. doi:10.1007/s00011-025-02179-1
- Ricceri F, Chiricozzi A, Peris K, et al. Successful use of anti–IL-23 molecules in overweight-to-obese psoriatic patients: a multicentric retrospective study. Dermatol Ther. 2022;35:E15793. doi:10.1111/dth.15793
- Jensen P, Zachariae C, Christensen R, et al. Effect of weight loss on the severity of psoriasis: a randomized clinical study. Br J Dermatol. 2013;168:319-327.
- Hossler EW, Wood GC, Still CD, et al. Psoriasis improvement following bariatric surgery is durable: 5-year follow-up in the Geisinger bariatric surgery cohort. Obes Surg. 2020;30:3350-3356.
- Romero-Talamás H, Daigle CR, Aminian A, et al. Psoriasis improvement after bariatric surgery. Surg Obes Relat Dis. 2014;10:1155-1159.
- Buonanno S, Gaggiano C, Terribili R, et al. Potential role of GLP-1 receptor agonists in the management of psoriatic disease: a scoping review. Inflamm Res. 2025;74:167. doi:10.1007/s00011-025-02140-2
- Barrea L, Muscogiuri G, Annunziata G, et al. Update on obesity in psoriasis patients: narrative review and practical insights. Clin Cosmet Investig Dermatol. 2023;16:3089-3104.
- Owczarczyk-Saczonek A, Gornowicz-Porowska J, Zegarska B. Psoriasis comorbidities: obesity, diet, and metabolic syndrome. Int J Mol Sci. 2024;25:1832.
- Vata D, Tarcau BM, Popescu IA, et al. Update on obesity in psoriasis patients. Life (Basel). 2023;13:1947.
- Piaserico S, Orlando G, Messina F. Psoriasis and cardiometabolic diseases: shared genetic and molecular pathways. Int J Mol Sci. 2022;23:9063.
- Hao Y, Zhu YJ, Zou S, et al. Metabolic syndrome and psoriasis: mechanisms and future directions. Front Immunol. 2021;12:711060.
- Kern L, Mittenbühler MJ, Vesting AJ, et al. Obesity-induced TNF-α and IL-6 signaling: the missing link between obesity and inflammation-driven liver and colorectal cancers. Cancers (Basel). 2019;11:24.
- Hwang J, Yoo JA, Yoon H, et al. Role of leptin in the association between obesity and psoriasis. Biomol Ther (Seoul). 2021;29:11-21.
- Smith B, Devjani S, Collier MR, et al. Association between psoriasis and obesity among US adults in the 2009-2014 National Health and Nutrition Examination Survey. Cutis. 2023;112:49-51. doi:10.12788/cutis.0807
- Ellulu MS, Patimah I, Khaza’ai H. Obesity and inflammation: the linking mechanism and the complications. Arch Med Sci. 2017;13:851-863.
- Wang H, Hou S, Kang X, et al. BMI matters: understanding the link between weight and severe psoriasis. Sci Rep. 2025;15:11158.
- Norden A, Rekhtman S, Strunk A, et al. Risk of psoriasis according to body mass index: a retrospective cohort analysis. J Am Acad Dermatol. 2022;86:1020-1026.
- Di Caprio R, Nigro E, Di Brizzi EV, et al. Exploring the link between psoriasis and adipose tissue: one amplifies the other. Int J Mol Sci. 2024;25:13435.
- Elmets CA, Leonardi CL, Davis DMR, et al. Joint AAD-NPF guidelines of care for the management and treatment of psoriasis with awareness and attention to comorbidities. J Am Acad Dermatol. 2019;80:1073-1113.
- Secchiero P, Rimondi E, Marcuzzi A, et al. Metabolic syndrome and psoriasis: pivotal roles of chronic inflammation and gut microbiota. Int J Mol Sci. 2024;25:8098.
- Burshtein J, Armstrong A, Chow M, et al. Association between obesity and efficacy of psoriasis therapies: an expert consensus panel. J Am Acad Dermatol. 2025;92:807-815. doi:10.1016/j.jaad.2024.12.016
- Pirro F, Caldarola G, Chiricozzi A, et al. Impact of body mass index on the efficacy of biological therapies in patients with psoriasis: a real-world study. Clin Drug Investig. 2021;41:917-925.
- Hjort G, Schwarz CW, Skov L, et al. Clinical characteristics associated with response to biologics in the treatment of psoriasis: a meta-analysis. JAMA Dermatol. 2024;160:830-837.
- Naldi L, Chimenti S, Girolomoni G, et al. Efficacy and safety of infliximab in obese and non-obese patients with plaque-type psoriasis: subanalysis of the EXPRESS II trial. Br J Dermatol. 2008;159:761-766.
- Puig L, Thom H, Mollon P, et al. Effect of body weight on the efficacy of biologics in moderate-to-severe plaque psoriasis: a systematic review and meta-analysis. J Eur Acad Dermatol Venereol. 2020;34:237-245.
- Dai M, Jiang Y, Wang Y, et al. Differential clinical factors influencing the effectiveness of distinct biologic agents in psoriasis: insights from a prospective cohort study in China. Inflamm Res. 2026;75:25. doi:10.1007/s00011-025-02179-1
- Ricceri F, Chiricozzi A, Peris K, et al. Successful use of anti–IL-23 molecules in overweight-to-obese psoriatic patients: a multicentric retrospective study. Dermatol Ther. 2022;35:E15793. doi:10.1111/dth.15793
- Jensen P, Zachariae C, Christensen R, et al. Effect of weight loss on the severity of psoriasis: a randomized clinical study. Br J Dermatol. 2013;168:319-327.
- Hossler EW, Wood GC, Still CD, et al. Psoriasis improvement following bariatric surgery is durable: 5-year follow-up in the Geisinger bariatric surgery cohort. Obes Surg. 2020;30:3350-3356.
- Romero-Talamás H, Daigle CR, Aminian A, et al. Psoriasis improvement after bariatric surgery. Surg Obes Relat Dis. 2014;10:1155-1159.
- Buonanno S, Gaggiano C, Terribili R, et al. Potential role of GLP-1 receptor agonists in the management of psoriatic disease: a scoping review. Inflamm Res. 2025;74:167. doi:10.1007/s00011-025-02140-2
Psoriasis and Obesity: A Clinical Review of the Bidirectional Link and Management Implications
Psoriasis and Obesity: A Clinical Review of the Bidirectional Link and Management Implications
Practice Points
- Obesity is an independent risk factor for psoriasis onset and severity. Both conditions share overlapping inflammatory pathways that create a self-perpetuating cycle of metabolic and cutaneous dysfunction.
- Dermatologists play a key role in early detection of comorbidities, and patients with psoriasis and obesity should undergo regular screening for metabolic syndrome, liver disease, and psoriatic arthritis.
- Weight loss is a critical therapeutic intervention that may improve Psoriasis Area and Severity Index scores and restore therapeutic responsiveness.
Black Dots on the Scalp of a Child
Black Dots on the Scalp of a Child
THE DIAGNOSIS: Terra Firma-Forme Dermatosis
During clinical examination, a 70% alcohol swab was utilized to gently rub several of the lesions, which were successfully removed. This confirmed a diagnosis of terra firma-forme dermatosis (TFFD)(also known as Duncan’s dirty dermatosis). The patient’s mother was counseled about the diagnosis and was instructed on how to use alcohol pads to remove the remaining lesions. Three days later, after several treatment sessions at home, the mother reported complete resolution of the lesions with no residual pigmentary changes, ulceration, or scarring (Figures 1 and 2).
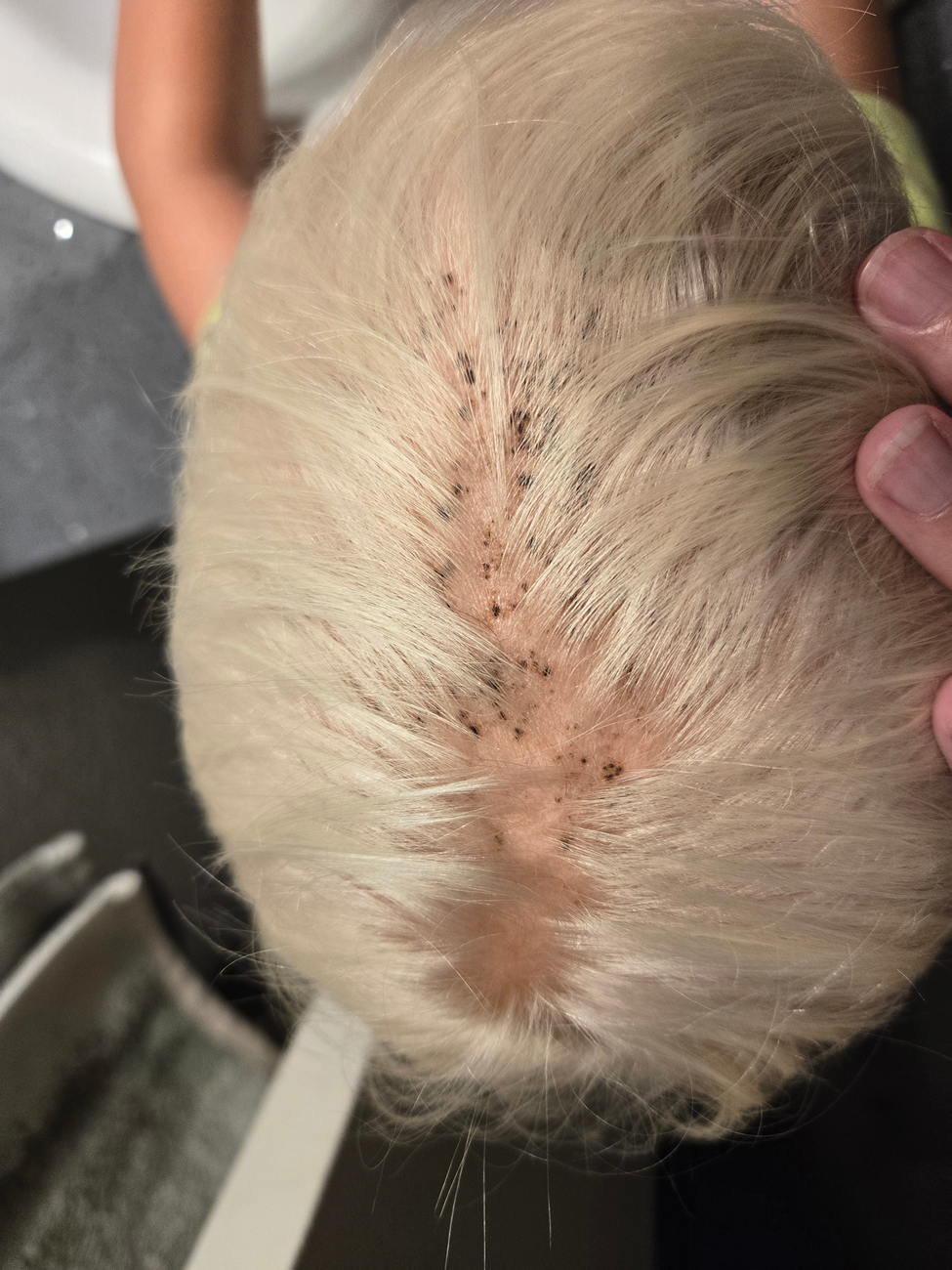
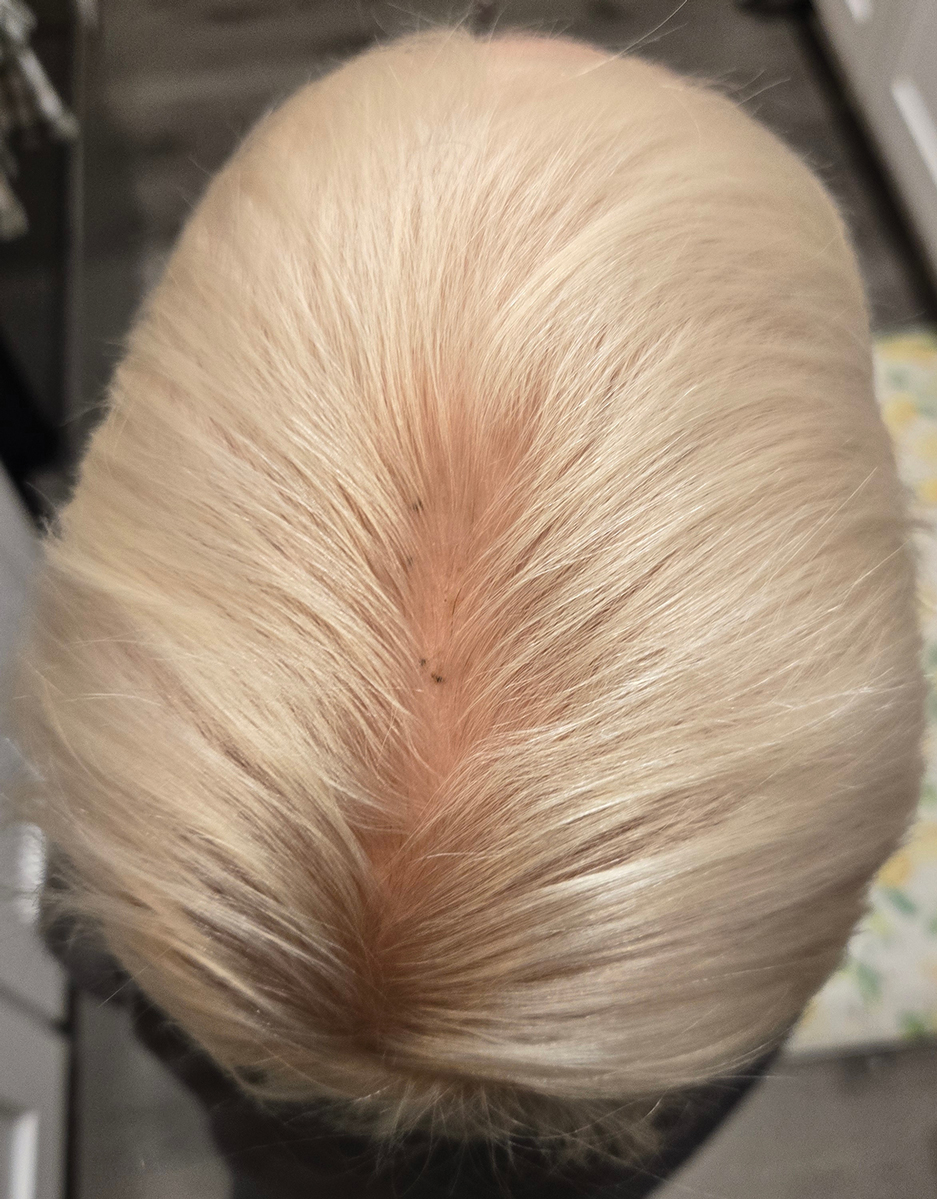
Terra firma-forme dermatosis was first described in 1987 in a 12-year-old girl with hyperpigmented plaques on the neck that cleared when rubbing alcohol was applied before biopsy.1,2 The term terra firma is Latin for “firm land” (or essentially “dirt”) in reference to what often is described as a characteristically “dirty” clinical appearance.2 Terra firmaforme dermatosis can manifest anywhere on the body but shows a predilection for the neck, arms and legs, axillae, inguinal region, and umbilicus.3 Lesions typically are described as asymptomatic, smooth, well-circumscribed, reticular papules or patches that are brown or black. Terra firma-forme dermatosis also may demonstrate secondary features such as hyperkeratotic, scaly, velvety, or verrucous plaques and nodules.3
The etiology of this condition is theorized to be a result of abnormal or delayed keratinization and prolonged keratinocyte adhesion.3,4 There are limited epidemiologic data, but TFFD has shown a predominance in children younger than 18 years (average age of onset, 10 years) with no known predilection for sex or race and no recognized pattern of inheritance.3-5
Histopathology typically demonstrates epidermal atrophy, hyperkeratosis, and often a component of trapping and compaction of melanin, sebum, microorganisms, and environmental debris.5
Management of TFFD is straightforward and generally consists of rubbing with 70% isopropyl alcohol to remove the lesions. For more adherent lesions or for extensive involvement, other keratolytics such as salicylic acid or alpha-hydroxy acids may be used.5 For TFFD manifesting in infants and young children, widespread involvement, or lesions involving the face or genitals, a urea-based keratolytic with or without a topical anti-inflammatory is suggested.5 Other treatment options include other alpha-hydroxy acids, topical retinoids, and nonpolar solvents such as acetone or CO2 laser for recalcitrant cases.4,5 Fortunately, most TFFD lesions respond well to conservative therapies, with recurrence reported only in 6.3% (5/79) of patients in one study.3
Dermatosis neglecta is clinically similar to TFFD and often is considered on the same spectrum of disease6; however, this entity is associated with decreased bathing or limited hygiene, which could be related to child or elder abuse/neglect or comorbid psychiatric disorders. These conditions can be distinguished by attempting to remove the lesions using soap and water; lesions of dermatosis neglecta will clear, whereas those of TFFD will not.
Metastatic melanoma in pediatric patients has a polymorphous appearance and may or may not be pigmented. Lesions often may be associated with lymphadenopathy of the draining lymph node basins, and nodules and lesions may be firm on palpation.7 Linear configurations of metastatic melanoma may represent a satellite or in-transit metastasis. Fortunately, melanoma is extraordinarily rare in children, with an estimated incidence of 2.1 per million for individuals younger than 20 years.8
Acanthosis nigricans is characterized by velvety plaques most commonly affecting the posterior neck, axillae, and flexor extremities. These lesions commonly are associated with obesity and insulin resistance but occasionally can be associated with underlying malignancy. In the latter association, acanthosis nigricans lesions tend to manifest more abruptly, often are pruritic, and can involve the mucous membranes. Fortunately, acanthosis nigricans related to malignancy in the pediatric population is rare.9
Epidermal nevi may exhibit clinical similarities to TFFD, particularly in lesions with brown/black pigment or with a reticulated or verrucous appearance; however, epidermal nevi often are congenital or manifest within the first few years of life. They commonly are distributed over the lines of Blaschko and have a linear appearance; they also enlarge and thicken as the patient ages.10
Black-dot tinea capitis, a classic manifestation of endothrix infection, manifests as alopecia with broken hairs and is most commonly caused by Tinea tonsurans.11 The black dots refer to the appearance of the infected hair shafts, which have been weakened and broken off at the follicular ostia. As such, lesions typically are monomorphic and may be interspersed with uninvolved hair shafts. There often is associated scale and a lack of inflammation.11,12
Additional differential diagnoses to consider include seborrheic keratoses and confluent and reticulated papillomatosis. Further workup (eg, potassium hydroxide preparation of skin scrapings or skin biopsy) may help elucidate the diagnosis.5 A simple and cost-effective initial diagnostic tool involves wiping suspicious lesions with a 70% isopropyl alcohol pad to confirm this diagnosis.
- Duncan WC. Terra firma-forme dermatosis. Arch Dermatol. 1987;123:567. doi:10.1001/archderm.1987.01660290031009
- Greywal T, Cohen PR. Terra firma-forme dermatosis: a report of ten individuals with Duncan’s dirty dermatosis and literature review. Dermatol Pract Concept. 2015:29-33. doi:10.5826/dpc.0503a08
- Aslan NÇ, Güler S, Demirci K, et al. Features of terra firma-forme dermatosis. Ann Fam Med. 2018;16:52-54. doi:10.1370/afm.2175
- Sechi A, Patrizi A, Savoia F, et al. Terra firma-forme dermatosis. Clin Dermatol. 2021;39:202-205. doi:10.1016/j.clindermatol.2020.10.019
- Mohta A, Sarkar R, Narayan RV, et al. Terra firma-forme dermatosis—more than just dirty. Indian Dermatol Online J. 2024;15:99-104. doi:10.4103/idoj.idoj_424_23
- Erkek E, Çetin E, Sahin S, et al. Terra firma-forme dermatosis. Indian J Dermatol Venereol Leprol. 2012;78:358. doi:10.4103 /0378-6323.95455
- McMullan P, Grant-Kels JM. Childhood and adolescent melanoma: an update. Clin Dermatol. 2025;43:16-23. doi:10.1016 /j.clindermatol.2025.01.010
- NCCR*Explorer: An interactive website for NCCR cancer statistics. National Cancer Institute website. Accessed January 10, 2025. https://nccrexplorer.ccdi.cancer.gov/data-products.html
- Sinha S, Schwartz RA. Juvenile acanthosis nigricans. J Am Acad Dermatol. 2007;57:502-508. doi:10.1016/j.jaad.2006.08.016
- Waldman AR, Garzon MC, Morel KD. Epidermal nevi: what is new. Dermatol Clin. 2022;40:61-71. doi:10.1016/j.det.2021.09.006
- Wang X. Black dot tinea capitis. N Engl J Med. 2024; 391:E7. doi:10.1056/NEJMicm2401964
- Gupta AK, Summerbell RC. Tinea capitis. Med Mycol. 2000; 38:255-287. doi:10.1080/mmy.38.4.255.287
THE DIAGNOSIS: Terra Firma-Forme Dermatosis
During clinical examination, a 70% alcohol swab was utilized to gently rub several of the lesions, which were successfully removed. This confirmed a diagnosis of terra firma-forme dermatosis (TFFD)(also known as Duncan’s dirty dermatosis). The patient’s mother was counseled about the diagnosis and was instructed on how to use alcohol pads to remove the remaining lesions. Three days later, after several treatment sessions at home, the mother reported complete resolution of the lesions with no residual pigmentary changes, ulceration, or scarring (Figures 1 and 2).


Terra firma-forme dermatosis was first described in 1987 in a 12-year-old girl with hyperpigmented plaques on the neck that cleared when rubbing alcohol was applied before biopsy.1,2 The term terra firma is Latin for “firm land” (or essentially “dirt”) in reference to what often is described as a characteristically “dirty” clinical appearance.2 Terra firmaforme dermatosis can manifest anywhere on the body but shows a predilection for the neck, arms and legs, axillae, inguinal region, and umbilicus.3 Lesions typically are described as asymptomatic, smooth, well-circumscribed, reticular papules or patches that are brown or black. Terra firma-forme dermatosis also may demonstrate secondary features such as hyperkeratotic, scaly, velvety, or verrucous plaques and nodules.3
The etiology of this condition is theorized to be a result of abnormal or delayed keratinization and prolonged keratinocyte adhesion.3,4 There are limited epidemiologic data, but TFFD has shown a predominance in children younger than 18 years (average age of onset, 10 years) with no known predilection for sex or race and no recognized pattern of inheritance.3-5
Histopathology typically demonstrates epidermal atrophy, hyperkeratosis, and often a component of trapping and compaction of melanin, sebum, microorganisms, and environmental debris.5
Management of TFFD is straightforward and generally consists of rubbing with 70% isopropyl alcohol to remove the lesions. For more adherent lesions or for extensive involvement, other keratolytics such as salicylic acid or alpha-hydroxy acids may be used.5 For TFFD manifesting in infants and young children, widespread involvement, or lesions involving the face or genitals, a urea-based keratolytic with or without a topical anti-inflammatory is suggested.5 Other treatment options include other alpha-hydroxy acids, topical retinoids, and nonpolar solvents such as acetone or CO2 laser for recalcitrant cases.4,5 Fortunately, most TFFD lesions respond well to conservative therapies, with recurrence reported only in 6.3% (5/79) of patients in one study.3
Dermatosis neglecta is clinically similar to TFFD and often is considered on the same spectrum of disease6; however, this entity is associated with decreased bathing or limited hygiene, which could be related to child or elder abuse/neglect or comorbid psychiatric disorders. These conditions can be distinguished by attempting to remove the lesions using soap and water; lesions of dermatosis neglecta will clear, whereas those of TFFD will not.
Metastatic melanoma in pediatric patients has a polymorphous appearance and may or may not be pigmented. Lesions often may be associated with lymphadenopathy of the draining lymph node basins, and nodules and lesions may be firm on palpation.7 Linear configurations of metastatic melanoma may represent a satellite or in-transit metastasis. Fortunately, melanoma is extraordinarily rare in children, with an estimated incidence of 2.1 per million for individuals younger than 20 years.8
Acanthosis nigricans is characterized by velvety plaques most commonly affecting the posterior neck, axillae, and flexor extremities. These lesions commonly are associated with obesity and insulin resistance but occasionally can be associated with underlying malignancy. In the latter association, acanthosis nigricans lesions tend to manifest more abruptly, often are pruritic, and can involve the mucous membranes. Fortunately, acanthosis nigricans related to malignancy in the pediatric population is rare.9
Epidermal nevi may exhibit clinical similarities to TFFD, particularly in lesions with brown/black pigment or with a reticulated or verrucous appearance; however, epidermal nevi often are congenital or manifest within the first few years of life. They commonly are distributed over the lines of Blaschko and have a linear appearance; they also enlarge and thicken as the patient ages.10
Black-dot tinea capitis, a classic manifestation of endothrix infection, manifests as alopecia with broken hairs and is most commonly caused by Tinea tonsurans.11 The black dots refer to the appearance of the infected hair shafts, which have been weakened and broken off at the follicular ostia. As such, lesions typically are monomorphic and may be interspersed with uninvolved hair shafts. There often is associated scale and a lack of inflammation.11,12
Additional differential diagnoses to consider include seborrheic keratoses and confluent and reticulated papillomatosis. Further workup (eg, potassium hydroxide preparation of skin scrapings or skin biopsy) may help elucidate the diagnosis.5 A simple and cost-effective initial diagnostic tool involves wiping suspicious lesions with a 70% isopropyl alcohol pad to confirm this diagnosis.
THE DIAGNOSIS: Terra Firma-Forme Dermatosis
During clinical examination, a 70% alcohol swab was utilized to gently rub several of the lesions, which were successfully removed. This confirmed a diagnosis of terra firma-forme dermatosis (TFFD)(also known as Duncan’s dirty dermatosis). The patient’s mother was counseled about the diagnosis and was instructed on how to use alcohol pads to remove the remaining lesions. Three days later, after several treatment sessions at home, the mother reported complete resolution of the lesions with no residual pigmentary changes, ulceration, or scarring (Figures 1 and 2).


Terra firma-forme dermatosis was first described in 1987 in a 12-year-old girl with hyperpigmented plaques on the neck that cleared when rubbing alcohol was applied before biopsy.1,2 The term terra firma is Latin for “firm land” (or essentially “dirt”) in reference to what often is described as a characteristically “dirty” clinical appearance.2 Terra firmaforme dermatosis can manifest anywhere on the body but shows a predilection for the neck, arms and legs, axillae, inguinal region, and umbilicus.3 Lesions typically are described as asymptomatic, smooth, well-circumscribed, reticular papules or patches that are brown or black. Terra firma-forme dermatosis also may demonstrate secondary features such as hyperkeratotic, scaly, velvety, or verrucous plaques and nodules.3
The etiology of this condition is theorized to be a result of abnormal or delayed keratinization and prolonged keratinocyte adhesion.3,4 There are limited epidemiologic data, but TFFD has shown a predominance in children younger than 18 years (average age of onset, 10 years) with no known predilection for sex or race and no recognized pattern of inheritance.3-5
Histopathology typically demonstrates epidermal atrophy, hyperkeratosis, and often a component of trapping and compaction of melanin, sebum, microorganisms, and environmental debris.5
Management of TFFD is straightforward and generally consists of rubbing with 70% isopropyl alcohol to remove the lesions. For more adherent lesions or for extensive involvement, other keratolytics such as salicylic acid or alpha-hydroxy acids may be used.5 For TFFD manifesting in infants and young children, widespread involvement, or lesions involving the face or genitals, a urea-based keratolytic with or without a topical anti-inflammatory is suggested.5 Other treatment options include other alpha-hydroxy acids, topical retinoids, and nonpolar solvents such as acetone or CO2 laser for recalcitrant cases.4,5 Fortunately, most TFFD lesions respond well to conservative therapies, with recurrence reported only in 6.3% (5/79) of patients in one study.3
Dermatosis neglecta is clinically similar to TFFD and often is considered on the same spectrum of disease6; however, this entity is associated with decreased bathing or limited hygiene, which could be related to child or elder abuse/neglect or comorbid psychiatric disorders. These conditions can be distinguished by attempting to remove the lesions using soap and water; lesions of dermatosis neglecta will clear, whereas those of TFFD will not.
Metastatic melanoma in pediatric patients has a polymorphous appearance and may or may not be pigmented. Lesions often may be associated with lymphadenopathy of the draining lymph node basins, and nodules and lesions may be firm on palpation.7 Linear configurations of metastatic melanoma may represent a satellite or in-transit metastasis. Fortunately, melanoma is extraordinarily rare in children, with an estimated incidence of 2.1 per million for individuals younger than 20 years.8
Acanthosis nigricans is characterized by velvety plaques most commonly affecting the posterior neck, axillae, and flexor extremities. These lesions commonly are associated with obesity and insulin resistance but occasionally can be associated with underlying malignancy. In the latter association, acanthosis nigricans lesions tend to manifest more abruptly, often are pruritic, and can involve the mucous membranes. Fortunately, acanthosis nigricans related to malignancy in the pediatric population is rare.9
Epidermal nevi may exhibit clinical similarities to TFFD, particularly in lesions with brown/black pigment or with a reticulated or verrucous appearance; however, epidermal nevi often are congenital or manifest within the first few years of life. They commonly are distributed over the lines of Blaschko and have a linear appearance; they also enlarge and thicken as the patient ages.10
Black-dot tinea capitis, a classic manifestation of endothrix infection, manifests as alopecia with broken hairs and is most commonly caused by Tinea tonsurans.11 The black dots refer to the appearance of the infected hair shafts, which have been weakened and broken off at the follicular ostia. As such, lesions typically are monomorphic and may be interspersed with uninvolved hair shafts. There often is associated scale and a lack of inflammation.11,12
Additional differential diagnoses to consider include seborrheic keratoses and confluent and reticulated papillomatosis. Further workup (eg, potassium hydroxide preparation of skin scrapings or skin biopsy) may help elucidate the diagnosis.5 A simple and cost-effective initial diagnostic tool involves wiping suspicious lesions with a 70% isopropyl alcohol pad to confirm this diagnosis.
- Duncan WC. Terra firma-forme dermatosis. Arch Dermatol. 1987;123:567. doi:10.1001/archderm.1987.01660290031009
- Greywal T, Cohen PR. Terra firma-forme dermatosis: a report of ten individuals with Duncan’s dirty dermatosis and literature review. Dermatol Pract Concept. 2015:29-33. doi:10.5826/dpc.0503a08
- Aslan NÇ, Güler S, Demirci K, et al. Features of terra firma-forme dermatosis. Ann Fam Med. 2018;16:52-54. doi:10.1370/afm.2175
- Sechi A, Patrizi A, Savoia F, et al. Terra firma-forme dermatosis. Clin Dermatol. 2021;39:202-205. doi:10.1016/j.clindermatol.2020.10.019
- Mohta A, Sarkar R, Narayan RV, et al. Terra firma-forme dermatosis—more than just dirty. Indian Dermatol Online J. 2024;15:99-104. doi:10.4103/idoj.idoj_424_23
- Erkek E, Çetin E, Sahin S, et al. Terra firma-forme dermatosis. Indian J Dermatol Venereol Leprol. 2012;78:358. doi:10.4103 /0378-6323.95455
- McMullan P, Grant-Kels JM. Childhood and adolescent melanoma: an update. Clin Dermatol. 2025;43:16-23. doi:10.1016 /j.clindermatol.2025.01.010
- NCCR*Explorer: An interactive website for NCCR cancer statistics. National Cancer Institute website. Accessed January 10, 2025. https://nccrexplorer.ccdi.cancer.gov/data-products.html
- Sinha S, Schwartz RA. Juvenile acanthosis nigricans. J Am Acad Dermatol. 2007;57:502-508. doi:10.1016/j.jaad.2006.08.016
- Waldman AR, Garzon MC, Morel KD. Epidermal nevi: what is new. Dermatol Clin. 2022;40:61-71. doi:10.1016/j.det.2021.09.006
- Wang X. Black dot tinea capitis. N Engl J Med. 2024; 391:E7. doi:10.1056/NEJMicm2401964
- Gupta AK, Summerbell RC. Tinea capitis. Med Mycol. 2000; 38:255-287. doi:10.1080/mmy.38.4.255.287
- Duncan WC. Terra firma-forme dermatosis. Arch Dermatol. 1987;123:567. doi:10.1001/archderm.1987.01660290031009
- Greywal T, Cohen PR. Terra firma-forme dermatosis: a report of ten individuals with Duncan’s dirty dermatosis and literature review. Dermatol Pract Concept. 2015:29-33. doi:10.5826/dpc.0503a08
- Aslan NÇ, Güler S, Demirci K, et al. Features of terra firma-forme dermatosis. Ann Fam Med. 2018;16:52-54. doi:10.1370/afm.2175
- Sechi A, Patrizi A, Savoia F, et al. Terra firma-forme dermatosis. Clin Dermatol. 2021;39:202-205. doi:10.1016/j.clindermatol.2020.10.019
- Mohta A, Sarkar R, Narayan RV, et al. Terra firma-forme dermatosis—more than just dirty. Indian Dermatol Online J. 2024;15:99-104. doi:10.4103/idoj.idoj_424_23
- Erkek E, Çetin E, Sahin S, et al. Terra firma-forme dermatosis. Indian J Dermatol Venereol Leprol. 2012;78:358. doi:10.4103 /0378-6323.95455
- McMullan P, Grant-Kels JM. Childhood and adolescent melanoma: an update. Clin Dermatol. 2025;43:16-23. doi:10.1016 /j.clindermatol.2025.01.010
- NCCR*Explorer: An interactive website for NCCR cancer statistics. National Cancer Institute website. Accessed January 10, 2025. https://nccrexplorer.ccdi.cancer.gov/data-products.html
- Sinha S, Schwartz RA. Juvenile acanthosis nigricans. J Am Acad Dermatol. 2007;57:502-508. doi:10.1016/j.jaad.2006.08.016
- Waldman AR, Garzon MC, Morel KD. Epidermal nevi: what is new. Dermatol Clin. 2022;40:61-71. doi:10.1016/j.det.2021.09.006
- Wang X. Black dot tinea capitis. N Engl J Med. 2024; 391:E7. doi:10.1056/NEJMicm2401964
- Gupta AK, Summerbell RC. Tinea capitis. Med Mycol. 2000; 38:255-287. doi:10.1080/mmy.38.4.255.287
Black Dots on the Scalp of a Child
Black Dots on the Scalp of a Child
A 4-year-old boy was referred to the dermatology clinic by his pediatrician for evaluation of persistent black spots on the scalp of 1 month’s duration. The patient was otherwise healthy, and his mother stated that the lesions had appeared gradually, were not tender or pruritic, and did not wash off with shampoo and scrubbing. The patient had no history of any systemic illness, recent travel, genetic disorders, or genodermatoses. Physical examination revealed multiple well-circumscribed, 1- to 2-mm black papules and macules with confluence scattered over the vertex scalp. No erythema, scale, or induration was noted.
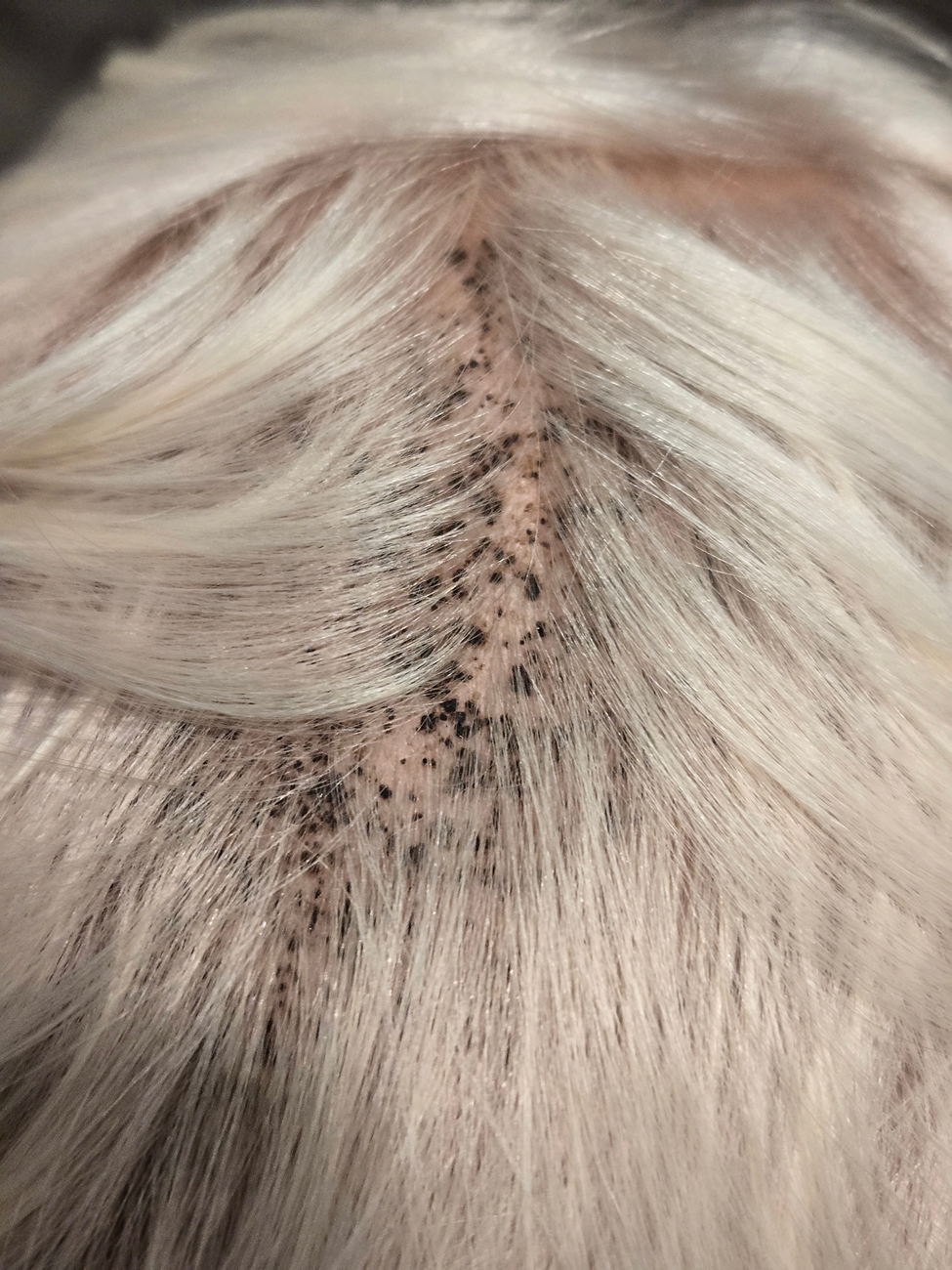
Assessing Inpatient Dermatology Availability in Virginia
Assessing Inpatient Dermatology Availability in Virginia
To the Editor:
It is known that dermatologist evaluation of skin conditions in hospitalized patients confers enhanced diagnostic accuracy, timely and appropriate treatment, and an overall reduction in readmissions compared to assessments by nondermatology hospitalists.1 Dermatology consultations have been shown to alter diagnoses in up to 50% of cases and lead to changes in management in nearly 75% of cases, even for prevalent dermatologic conditions such as drug rashes, cellulitis, and stasis dermatitis.1,2 Previous studies have observed a multiday reduction in length of hospital stay, a 10-fold reduction in readmission rate, and lower 30-day mortality, all leading to a reduction in patient morbidity and costs to both the patient and the health care system.3,4 Despite these benefits, there has been a decrease in the number of dermatologists providing inpatient services and a reduction in medical centers offering dermatology consultations over the past several years.5 To better appreciate current trends of declining dermatology inpatient and consultative services within our region, we evaluated the availability of dermatology care at hospitals across Virginia.
A simple telephone survey was conducted across community hospitals in Virginia wherein medical staff administrators were asked to provide details regarding their dermatology staffing. The following figures were collected: number of dermatologists on staff, number of dermatologists with consulting privileges, number of affiliated dermatologists, and number of advanced-practice dermatology providers. Follow-up calls were carried out to elaborate on how dermatologists (when available) were integrated into inpatient care workflow and made accessible to hospitalists and emergency medicine departments. Academic centers, military hospitals, and specialty hospitals were excluded from the survey.
To better appreciate the relationships between hospital and population characteristics and the availability of dermatology care, publicly available data were collected on hospital bed counts and regional population density for each facility.6-9 Spearman rank correlation analyses were conducted in Microsoft Excel to evaluate the association between the number of dermatologists on staff, number of consulting dermatologists, staffed inpatient beds, and population size.
Sixty-four hospitals—more than 70% of the 90 eligible community hospitals—responded to the survey between May and August 2024 and were included in the study. On-staff dermatologists were present at 8 (12.5%) of the hospitals surveyed; of these, 4 (50.0%) hospitals had between 1 and 5 dermatologists, 3 (37.5%) had between 6 and 10 dermatologists, and 1 (12.5%) had between 11 and 15 dermatologists. An additional 4 (6.3%) hospitals provided consultative dermatology services from outside dermatology clinics. Urban hospitals accounted for 9 of 12 (75%) hospitals offering in-house dermatology services, either through on-staff physicians or consultations with clinic-based providers.
Based on Spearman rank correlation analysis, there was a positive correlation between the number of dermatologists on staff and the number of staffed hospital beds (r=0.61; P <.001). Similarly, there was a positive correlation between the number of dermatologists on staff and the population density of the affiliated region (r=0.58; P <.001). Finally, there was a positive correlation between the number of dermatologists on staff and the number of available consulting dermatologists (r=0.89; P <.001).
At facilities with only consultative dermatology services accessible, there often was no formal dermatology team or department present. Rather, the hospitals relied on a loosely affiliated network of dermatology providers or navigated inpatient dermatology needs almost exclusively via internal medicine hospitalists or emergency medicine physicians. When available, dermatology support from dermatology physicians often was provided through teledermatology platforms. Although teledermatology has a large role in increasing access to care within underserved areas, its reliance on images and second-hand case descriptions can limit the provider’s ability to perform a comprehensive examination and assessment. Moreover, it was noted that few hospital representatives could offer clarity on how dermatologists were integrated into the inpatient setting. It remained unclear whether dermatologists were practically accessible to the inpatient care teams in a structured manner.
The uneven distribution and limited availability of dermatology inpatient care in Virginia reflect national trends and underscore ongoing access issues for patients. Without intentional intervention, these trends are expected to continue, contributing to a glaring gap in hospital services as well as to patient morbidity and mortality. The correlation data obtained in our study further qualify these disparities. The positive correlations between dermatologist availability and hospital size and population density suggest that larger, more urban facilities are more likely to offer inpatient dermatology care, whether through staffing or consultation. This relationship is not unexpected, given the greater financial resources and specialist networks available to facilities with large patient volumes. This suggests that dermatology care is shaped by institutional capacity and geographic leverage rather than clinical need, reinforcing existing disparities.
Importantly, it should be noted that the data may overestimate the true availability of dermatologists to these patients. As revealed via follow-up survey calls, respondent facilities that provided dermatology via consultative services often did not have a defined structure for integrating this care into the inpatient workflow. In some instances, dermatologists were technically affiliated with the hospital but had varying levels of practical interaction with the hospital providers and their patients. Administrative staff's differing awareness regarding dermatology interaction with the hospital facility may reveal systemic underutilization and opportunities to improve coordination to achieve the greatest benefit from dermatology services. These observations are further informed by the scope of our study, which focused specifically on community hospitals. The exclusion of academic and military institutions—and the tendency of these to exist in more densely populated areas—may have limited how broadly our findings reflect nationwide dermatology access by omitting more established dermatology departments and specialty care. As a result, regional variations in predominant facility type should be considered when interpreting the implications of these results beyond Virginia’s community hospital system.
In response to access limitations and differences in availability, facilities are turning to integrated teledermatology as a valuable tool to expand the reach of specialist care, particularly in rural or resource-limited settings. This modality acts as an important step toward improving equity in care by beginning to bridge geographic gaps; however, along with these logistical advantages, teledermatology also confers diagnostic limitations and clinical trade-offs that should be thoughtfully considered. Our findings highlight the need to expand access in a way that integrates technological advances with in-person care to build a sustainable and effective path forward without compromising the quality of care patients receive. We present these outcomes to emphasize the importance of increasing dermatology involvement in the care of hospitalized patients, which is a promising strategy to improve patient outcomes and reduce existing disparities in Virginia and nationwide.
- Hu L, Haynes H, Ferrazza D, et al. Impact of specialist consultations on inpatient admissions for dermatology-specific and related DRGs. J Gen Intern Med. 2013;28:1477-1482. doi:10.1007s11606-013-2440-2
- Madigan LM, Fox LP. Where are we now with inpatient consultative dermatology?: Assessing the value and evolution of this subspecialty over the past decade. J Am Acad Dermatol. 2019;80:1804-1808. doi:10.1016/j.jaad.2019.01.031
- Milani-Nejad N, Zhang M, Kaffenberger BH. Association of dermatology consultations with patient care outcomes in hospitalized patients with inflammatory skin diseases. JAMA Dermatol. 2017;153:523-528. doi:10.1001/jamadermatol.2016.6130
- Puri P, Pollock BD, Yousif M, et al. Association of society of dermatology hospitalist institutions with improved outcomes in Medicare beneficiaries hospitalized for skin disease. J Am Acad Dermatol. 2023;88:1372-1375. doi:10.1016/j.jaad.2023.01.021
- Hydol-Smith JA, Gallardo MA, Korman A, et al. The United States dermatology inpatient workforce between 2013 and 2019: a Medicare analysis reveals contraction of the workforce and vast access deserts—a cross-sectional analysis. Arch Dermatol Res. 2024;316:103. doi:10.1007/s00403-024-02845-0
- QuickFacts: Virginia. 2024. Census Bureau QuickFacts. https://www.census.gov/quickfacts/fact/table/VA/PST045224
- American Hospital Directory. Individual hospital statistics for Virginia. Updated May 7, 2023. Accessed November 12, 2025. https://www.ahd.com/states/hospital_VA.html
- Virginia Office of Data Governance and Analytics. Definitive healthcare: USA hospital beds (CSV). Virginia Open Data Portal. Accessed November 12, 2025. https://data.virginia.gov/dataset/definitive-healthcare-usa-hospital-beds/resource/c39226d7-1b28-4ce0-8f35-3a0ff974eba5
- Virginia Health Information. Virginia hospitals. Updated February 26, 2021. Accessed November 12, 2025. https://www.vhi.org/Hospitals/vahospitals.asp
To the Editor:
It is known that dermatologist evaluation of skin conditions in hospitalized patients confers enhanced diagnostic accuracy, timely and appropriate treatment, and an overall reduction in readmissions compared to assessments by nondermatology hospitalists.1 Dermatology consultations have been shown to alter diagnoses in up to 50% of cases and lead to changes in management in nearly 75% of cases, even for prevalent dermatologic conditions such as drug rashes, cellulitis, and stasis dermatitis.1,2 Previous studies have observed a multiday reduction in length of hospital stay, a 10-fold reduction in readmission rate, and lower 30-day mortality, all leading to a reduction in patient morbidity and costs to both the patient and the health care system.3,4 Despite these benefits, there has been a decrease in the number of dermatologists providing inpatient services and a reduction in medical centers offering dermatology consultations over the past several years.5 To better appreciate current trends of declining dermatology inpatient and consultative services within our region, we evaluated the availability of dermatology care at hospitals across Virginia.
A simple telephone survey was conducted across community hospitals in Virginia wherein medical staff administrators were asked to provide details regarding their dermatology staffing. The following figures were collected: number of dermatologists on staff, number of dermatologists with consulting privileges, number of affiliated dermatologists, and number of advanced-practice dermatology providers. Follow-up calls were carried out to elaborate on how dermatologists (when available) were integrated into inpatient care workflow and made accessible to hospitalists and emergency medicine departments. Academic centers, military hospitals, and specialty hospitals were excluded from the survey.
To better appreciate the relationships between hospital and population characteristics and the availability of dermatology care, publicly available data were collected on hospital bed counts and regional population density for each facility.6-9 Spearman rank correlation analyses were conducted in Microsoft Excel to evaluate the association between the number of dermatologists on staff, number of consulting dermatologists, staffed inpatient beds, and population size.
Sixty-four hospitals—more than 70% of the 90 eligible community hospitals—responded to the survey between May and August 2024 and were included in the study. On-staff dermatologists were present at 8 (12.5%) of the hospitals surveyed; of these, 4 (50.0%) hospitals had between 1 and 5 dermatologists, 3 (37.5%) had between 6 and 10 dermatologists, and 1 (12.5%) had between 11 and 15 dermatologists. An additional 4 (6.3%) hospitals provided consultative dermatology services from outside dermatology clinics. Urban hospitals accounted for 9 of 12 (75%) hospitals offering in-house dermatology services, either through on-staff physicians or consultations with clinic-based providers.
Based on Spearman rank correlation analysis, there was a positive correlation between the number of dermatologists on staff and the number of staffed hospital beds (r=0.61; P <.001). Similarly, there was a positive correlation between the number of dermatologists on staff and the population density of the affiliated region (r=0.58; P <.001). Finally, there was a positive correlation between the number of dermatologists on staff and the number of available consulting dermatologists (r=0.89; P <.001).
At facilities with only consultative dermatology services accessible, there often was no formal dermatology team or department present. Rather, the hospitals relied on a loosely affiliated network of dermatology providers or navigated inpatient dermatology needs almost exclusively via internal medicine hospitalists or emergency medicine physicians. When available, dermatology support from dermatology physicians often was provided through teledermatology platforms. Although teledermatology has a large role in increasing access to care within underserved areas, its reliance on images and second-hand case descriptions can limit the provider’s ability to perform a comprehensive examination and assessment. Moreover, it was noted that few hospital representatives could offer clarity on how dermatologists were integrated into the inpatient setting. It remained unclear whether dermatologists were practically accessible to the inpatient care teams in a structured manner.
The uneven distribution and limited availability of dermatology inpatient care in Virginia reflect national trends and underscore ongoing access issues for patients. Without intentional intervention, these trends are expected to continue, contributing to a glaring gap in hospital services as well as to patient morbidity and mortality. The correlation data obtained in our study further qualify these disparities. The positive correlations between dermatologist availability and hospital size and population density suggest that larger, more urban facilities are more likely to offer inpatient dermatology care, whether through staffing or consultation. This relationship is not unexpected, given the greater financial resources and specialist networks available to facilities with large patient volumes. This suggests that dermatology care is shaped by institutional capacity and geographic leverage rather than clinical need, reinforcing existing disparities.
Importantly, it should be noted that the data may overestimate the true availability of dermatologists to these patients. As revealed via follow-up survey calls, respondent facilities that provided dermatology via consultative services often did not have a defined structure for integrating this care into the inpatient workflow. In some instances, dermatologists were technically affiliated with the hospital but had varying levels of practical interaction with the hospital providers and their patients. Administrative staff's differing awareness regarding dermatology interaction with the hospital facility may reveal systemic underutilization and opportunities to improve coordination to achieve the greatest benefit from dermatology services. These observations are further informed by the scope of our study, which focused specifically on community hospitals. The exclusion of academic and military institutions—and the tendency of these to exist in more densely populated areas—may have limited how broadly our findings reflect nationwide dermatology access by omitting more established dermatology departments and specialty care. As a result, regional variations in predominant facility type should be considered when interpreting the implications of these results beyond Virginia’s community hospital system.
In response to access limitations and differences in availability, facilities are turning to integrated teledermatology as a valuable tool to expand the reach of specialist care, particularly in rural or resource-limited settings. This modality acts as an important step toward improving equity in care by beginning to bridge geographic gaps; however, along with these logistical advantages, teledermatology also confers diagnostic limitations and clinical trade-offs that should be thoughtfully considered. Our findings highlight the need to expand access in a way that integrates technological advances with in-person care to build a sustainable and effective path forward without compromising the quality of care patients receive. We present these outcomes to emphasize the importance of increasing dermatology involvement in the care of hospitalized patients, which is a promising strategy to improve patient outcomes and reduce existing disparities in Virginia and nationwide.
To the Editor:
It is known that dermatologist evaluation of skin conditions in hospitalized patients confers enhanced diagnostic accuracy, timely and appropriate treatment, and an overall reduction in readmissions compared to assessments by nondermatology hospitalists.1 Dermatology consultations have been shown to alter diagnoses in up to 50% of cases and lead to changes in management in nearly 75% of cases, even for prevalent dermatologic conditions such as drug rashes, cellulitis, and stasis dermatitis.1,2 Previous studies have observed a multiday reduction in length of hospital stay, a 10-fold reduction in readmission rate, and lower 30-day mortality, all leading to a reduction in patient morbidity and costs to both the patient and the health care system.3,4 Despite these benefits, there has been a decrease in the number of dermatologists providing inpatient services and a reduction in medical centers offering dermatology consultations over the past several years.5 To better appreciate current trends of declining dermatology inpatient and consultative services within our region, we evaluated the availability of dermatology care at hospitals across Virginia.
A simple telephone survey was conducted across community hospitals in Virginia wherein medical staff administrators were asked to provide details regarding their dermatology staffing. The following figures were collected: number of dermatologists on staff, number of dermatologists with consulting privileges, number of affiliated dermatologists, and number of advanced-practice dermatology providers. Follow-up calls were carried out to elaborate on how dermatologists (when available) were integrated into inpatient care workflow and made accessible to hospitalists and emergency medicine departments. Academic centers, military hospitals, and specialty hospitals were excluded from the survey.
To better appreciate the relationships between hospital and population characteristics and the availability of dermatology care, publicly available data were collected on hospital bed counts and regional population density for each facility.6-9 Spearman rank correlation analyses were conducted in Microsoft Excel to evaluate the association between the number of dermatologists on staff, number of consulting dermatologists, staffed inpatient beds, and population size.
Sixty-four hospitals—more than 70% of the 90 eligible community hospitals—responded to the survey between May and August 2024 and were included in the study. On-staff dermatologists were present at 8 (12.5%) of the hospitals surveyed; of these, 4 (50.0%) hospitals had between 1 and 5 dermatologists, 3 (37.5%) had between 6 and 10 dermatologists, and 1 (12.5%) had between 11 and 15 dermatologists. An additional 4 (6.3%) hospitals provided consultative dermatology services from outside dermatology clinics. Urban hospitals accounted for 9 of 12 (75%) hospitals offering in-house dermatology services, either through on-staff physicians or consultations with clinic-based providers.
Based on Spearman rank correlation analysis, there was a positive correlation between the number of dermatologists on staff and the number of staffed hospital beds (r=0.61; P <.001). Similarly, there was a positive correlation between the number of dermatologists on staff and the population density of the affiliated region (r=0.58; P <.001). Finally, there was a positive correlation between the number of dermatologists on staff and the number of available consulting dermatologists (r=0.89; P <.001).
At facilities with only consultative dermatology services accessible, there often was no formal dermatology team or department present. Rather, the hospitals relied on a loosely affiliated network of dermatology providers or navigated inpatient dermatology needs almost exclusively via internal medicine hospitalists or emergency medicine physicians. When available, dermatology support from dermatology physicians often was provided through teledermatology platforms. Although teledermatology has a large role in increasing access to care within underserved areas, its reliance on images and second-hand case descriptions can limit the provider’s ability to perform a comprehensive examination and assessment. Moreover, it was noted that few hospital representatives could offer clarity on how dermatologists were integrated into the inpatient setting. It remained unclear whether dermatologists were practically accessible to the inpatient care teams in a structured manner.
The uneven distribution and limited availability of dermatology inpatient care in Virginia reflect national trends and underscore ongoing access issues for patients. Without intentional intervention, these trends are expected to continue, contributing to a glaring gap in hospital services as well as to patient morbidity and mortality. The correlation data obtained in our study further qualify these disparities. The positive correlations between dermatologist availability and hospital size and population density suggest that larger, more urban facilities are more likely to offer inpatient dermatology care, whether through staffing or consultation. This relationship is not unexpected, given the greater financial resources and specialist networks available to facilities with large patient volumes. This suggests that dermatology care is shaped by institutional capacity and geographic leverage rather than clinical need, reinforcing existing disparities.
Importantly, it should be noted that the data may overestimate the true availability of dermatologists to these patients. As revealed via follow-up survey calls, respondent facilities that provided dermatology via consultative services often did not have a defined structure for integrating this care into the inpatient workflow. In some instances, dermatologists were technically affiliated with the hospital but had varying levels of practical interaction with the hospital providers and their patients. Administrative staff's differing awareness regarding dermatology interaction with the hospital facility may reveal systemic underutilization and opportunities to improve coordination to achieve the greatest benefit from dermatology services. These observations are further informed by the scope of our study, which focused specifically on community hospitals. The exclusion of academic and military institutions—and the tendency of these to exist in more densely populated areas—may have limited how broadly our findings reflect nationwide dermatology access by omitting more established dermatology departments and specialty care. As a result, regional variations in predominant facility type should be considered when interpreting the implications of these results beyond Virginia’s community hospital system.
In response to access limitations and differences in availability, facilities are turning to integrated teledermatology as a valuable tool to expand the reach of specialist care, particularly in rural or resource-limited settings. This modality acts as an important step toward improving equity in care by beginning to bridge geographic gaps; however, along with these logistical advantages, teledermatology also confers diagnostic limitations and clinical trade-offs that should be thoughtfully considered. Our findings highlight the need to expand access in a way that integrates technological advances with in-person care to build a sustainable and effective path forward without compromising the quality of care patients receive. We present these outcomes to emphasize the importance of increasing dermatology involvement in the care of hospitalized patients, which is a promising strategy to improve patient outcomes and reduce existing disparities in Virginia and nationwide.
- Hu L, Haynes H, Ferrazza D, et al. Impact of specialist consultations on inpatient admissions for dermatology-specific and related DRGs. J Gen Intern Med. 2013;28:1477-1482. doi:10.1007s11606-013-2440-2
- Madigan LM, Fox LP. Where are we now with inpatient consultative dermatology?: Assessing the value and evolution of this subspecialty over the past decade. J Am Acad Dermatol. 2019;80:1804-1808. doi:10.1016/j.jaad.2019.01.031
- Milani-Nejad N, Zhang M, Kaffenberger BH. Association of dermatology consultations with patient care outcomes in hospitalized patients with inflammatory skin diseases. JAMA Dermatol. 2017;153:523-528. doi:10.1001/jamadermatol.2016.6130
- Puri P, Pollock BD, Yousif M, et al. Association of society of dermatology hospitalist institutions with improved outcomes in Medicare beneficiaries hospitalized for skin disease. J Am Acad Dermatol. 2023;88:1372-1375. doi:10.1016/j.jaad.2023.01.021
- Hydol-Smith JA, Gallardo MA, Korman A, et al. The United States dermatology inpatient workforce between 2013 and 2019: a Medicare analysis reveals contraction of the workforce and vast access deserts—a cross-sectional analysis. Arch Dermatol Res. 2024;316:103. doi:10.1007/s00403-024-02845-0
- QuickFacts: Virginia. 2024. Census Bureau QuickFacts. https://www.census.gov/quickfacts/fact/table/VA/PST045224
- American Hospital Directory. Individual hospital statistics for Virginia. Updated May 7, 2023. Accessed November 12, 2025. https://www.ahd.com/states/hospital_VA.html
- Virginia Office of Data Governance and Analytics. Definitive healthcare: USA hospital beds (CSV). Virginia Open Data Portal. Accessed November 12, 2025. https://data.virginia.gov/dataset/definitive-healthcare-usa-hospital-beds/resource/c39226d7-1b28-4ce0-8f35-3a0ff974eba5
- Virginia Health Information. Virginia hospitals. Updated February 26, 2021. Accessed November 12, 2025. https://www.vhi.org/Hospitals/vahospitals.asp
- Hu L, Haynes H, Ferrazza D, et al. Impact of specialist consultations on inpatient admissions for dermatology-specific and related DRGs. J Gen Intern Med. 2013;28:1477-1482. doi:10.1007s11606-013-2440-2
- Madigan LM, Fox LP. Where are we now with inpatient consultative dermatology?: Assessing the value and evolution of this subspecialty over the past decade. J Am Acad Dermatol. 2019;80:1804-1808. doi:10.1016/j.jaad.2019.01.031
- Milani-Nejad N, Zhang M, Kaffenberger BH. Association of dermatology consultations with patient care outcomes in hospitalized patients with inflammatory skin diseases. JAMA Dermatol. 2017;153:523-528. doi:10.1001/jamadermatol.2016.6130
- Puri P, Pollock BD, Yousif M, et al. Association of society of dermatology hospitalist institutions with improved outcomes in Medicare beneficiaries hospitalized for skin disease. J Am Acad Dermatol. 2023;88:1372-1375. doi:10.1016/j.jaad.2023.01.021
- Hydol-Smith JA, Gallardo MA, Korman A, et al. The United States dermatology inpatient workforce between 2013 and 2019: a Medicare analysis reveals contraction of the workforce and vast access deserts—a cross-sectional analysis. Arch Dermatol Res. 2024;316:103. doi:10.1007/s00403-024-02845-0
- QuickFacts: Virginia. 2024. Census Bureau QuickFacts. https://www.census.gov/quickfacts/fact/table/VA/PST045224
- American Hospital Directory. Individual hospital statistics for Virginia. Updated May 7, 2023. Accessed November 12, 2025. https://www.ahd.com/states/hospital_VA.html
- Virginia Office of Data Governance and Analytics. Definitive healthcare: USA hospital beds (CSV). Virginia Open Data Portal. Accessed November 12, 2025. https://data.virginia.gov/dataset/definitive-healthcare-usa-hospital-beds/resource/c39226d7-1b28-4ce0-8f35-3a0ff974eba5
- Virginia Health Information. Virginia hospitals. Updated February 26, 2021. Accessed November 12, 2025. https://www.vhi.org/Hospitals/vahospitals.asp
Assessing Inpatient Dermatology Availability in Virginia
Assessing Inpatient Dermatology Availability in Virginia