User login
Telecare intervention lessens chronic joint pain
A telecare intervention within the primary care practice setting elicited clinically meaningful and statistically significant improvements in long-standing chronic joint and other musculoskeletal pain, according to a report published online July 15 in JAMA.
A total of 250 adults with refractory, moderately severe regional (joints, limbs, back, or neck) or generalized (fibromyalgia or widespread) musculoskeletal pain of long duration participated in a randomized, blinded, 1-year clinical trial comparing the telecare intervention (124 patients) against usual chronic pain treatment (126 patients) in the primary care setting, reported Dr. Kurt Kroenke of the Veterans Affairs Center for Health Information and Communication, Roudebush VA Medical Center, Indianapolis, and his associates.
The intervention had three key components: frequent automated monitoring of patient symptoms using telephone or Internet voice recordings; nurse calls to patients who reported inadequate treatment response, adverse effects, or nonadherence; and analgesic management using a stepped-care algorithm, which was implemented by a nurse-manager who worked with a physician pain specialist, in collaboration with the primary care physician. The estimated total time spent per patient during the study year was 3-4 hours for the study nurse and 1 hour for the study physician.
Patients frequently checked in to the automated system to report their pain, anxiety, and depression symptoms; how difficult their pain made it for them to carry out usual activities; their adherence to medications; the degree of relief they obtained from pain medications; any change in pain, including the degree of improvement; adverse effects from their analgesics; and whether they wanted to change their medication regimen or speak to a nurse.
The medication algorithm called for the stepwise use of simple analgesics (acetaminophen and nonsteroidal anti-inflammatory drugs); tricyclic antidepressants (amitriptyline and nortriptyline) and cyclobenzaprine; tramadol; gabapentoids (gabapentin and pregabalin); topical analgesics; and opioids.
After 1 year, patients in the intervention group showed significantly greater improvement than the control group in measures of total pain, pain severity, and pain interference with daily activities, along with significantly greater satisfaction with their medical care. They were nearly twice as likely to report a 30% improvement from baseline (51.7%) than were patients in the usual-care group (27.1%).
They also were much less likely to report worsening pain (19.2% vs. 36.0%), which indicates a reduced risk of deterioration of their condition because their pain therapy was optimized, and much more likely to rate their medication as "good to excellent" (73.9% vs. 50.9%). And they reported greater improvements in secondary outcomes such as depression, anxiety, somatization, sleep, and social functioning, the investigators reported (JAMA 2014;312:240-248).
Patients in the intervention group received a greater number of analgesics for a greater length of time and at a higher mean dose than did those in the control group. However, only six patients in the entire study population initiated opioid use, and the median daily dose for all patients taking opioids was identical at the end of the study to what it was at the beginning. The two study groups showed no differences in their use of health care services such as outpatient visits, emergency department visits, and hospitalizations.
This study was supported by the U.S. Department of Veterans Affairs’ Health Services Research and Development Service. Dr. Kroenke reported receiving honoraria from Eli Lilly for work unrelated to this study.
This promising intervention doesn’t require significant additional time or effort from primary care physicians and is simple enough that it could be implemented in most primary care settings, said Dr. Michael E. Ohl and Dr. Gary E. Rosenthal.
"The authors estimated that the intervention required 3-4 hours of nurse-manager time and 1 hour of physician time per patient during a 12-month period. Thus, a single nurse could potentially manage the care of 500-600 patients with the support of a 0.25 full-time equivalent physician pain specialist," they noted.
Dr. Ohl and Dr. Rosenthal are at the Iowa City VA Medical Center and in the department of internal medicine at the University of Iowa. Dr. Rosenthal is also at the Institute for Clinical and Translational Sciences at the university. They reported no financial conflicts of interest. These remarks were taken from their editorial accompanying Dr. Kroenke’s report (JAMA 2014;312:235-6).
This promising intervention doesn’t require significant additional time or effort from primary care physicians and is simple enough that it could be implemented in most primary care settings, said Dr. Michael E. Ohl and Dr. Gary E. Rosenthal.
"The authors estimated that the intervention required 3-4 hours of nurse-manager time and 1 hour of physician time per patient during a 12-month period. Thus, a single nurse could potentially manage the care of 500-600 patients with the support of a 0.25 full-time equivalent physician pain specialist," they noted.
Dr. Ohl and Dr. Rosenthal are at the Iowa City VA Medical Center and in the department of internal medicine at the University of Iowa. Dr. Rosenthal is also at the Institute for Clinical and Translational Sciences at the university. They reported no financial conflicts of interest. These remarks were taken from their editorial accompanying Dr. Kroenke’s report (JAMA 2014;312:235-6).
This promising intervention doesn’t require significant additional time or effort from primary care physicians and is simple enough that it could be implemented in most primary care settings, said Dr. Michael E. Ohl and Dr. Gary E. Rosenthal.
"The authors estimated that the intervention required 3-4 hours of nurse-manager time and 1 hour of physician time per patient during a 12-month period. Thus, a single nurse could potentially manage the care of 500-600 patients with the support of a 0.25 full-time equivalent physician pain specialist," they noted.
Dr. Ohl and Dr. Rosenthal are at the Iowa City VA Medical Center and in the department of internal medicine at the University of Iowa. Dr. Rosenthal is also at the Institute for Clinical and Translational Sciences at the university. They reported no financial conflicts of interest. These remarks were taken from their editorial accompanying Dr. Kroenke’s report (JAMA 2014;312:235-6).
A telecare intervention within the primary care practice setting elicited clinically meaningful and statistically significant improvements in long-standing chronic joint and other musculoskeletal pain, according to a report published online July 15 in JAMA.
A total of 250 adults with refractory, moderately severe regional (joints, limbs, back, or neck) or generalized (fibromyalgia or widespread) musculoskeletal pain of long duration participated in a randomized, blinded, 1-year clinical trial comparing the telecare intervention (124 patients) against usual chronic pain treatment (126 patients) in the primary care setting, reported Dr. Kurt Kroenke of the Veterans Affairs Center for Health Information and Communication, Roudebush VA Medical Center, Indianapolis, and his associates.
The intervention had three key components: frequent automated monitoring of patient symptoms using telephone or Internet voice recordings; nurse calls to patients who reported inadequate treatment response, adverse effects, or nonadherence; and analgesic management using a stepped-care algorithm, which was implemented by a nurse-manager who worked with a physician pain specialist, in collaboration with the primary care physician. The estimated total time spent per patient during the study year was 3-4 hours for the study nurse and 1 hour for the study physician.
Patients frequently checked in to the automated system to report their pain, anxiety, and depression symptoms; how difficult their pain made it for them to carry out usual activities; their adherence to medications; the degree of relief they obtained from pain medications; any change in pain, including the degree of improvement; adverse effects from their analgesics; and whether they wanted to change their medication regimen or speak to a nurse.
The medication algorithm called for the stepwise use of simple analgesics (acetaminophen and nonsteroidal anti-inflammatory drugs); tricyclic antidepressants (amitriptyline and nortriptyline) and cyclobenzaprine; tramadol; gabapentoids (gabapentin and pregabalin); topical analgesics; and opioids.
After 1 year, patients in the intervention group showed significantly greater improvement than the control group in measures of total pain, pain severity, and pain interference with daily activities, along with significantly greater satisfaction with their medical care. They were nearly twice as likely to report a 30% improvement from baseline (51.7%) than were patients in the usual-care group (27.1%).
They also were much less likely to report worsening pain (19.2% vs. 36.0%), which indicates a reduced risk of deterioration of their condition because their pain therapy was optimized, and much more likely to rate their medication as "good to excellent" (73.9% vs. 50.9%). And they reported greater improvements in secondary outcomes such as depression, anxiety, somatization, sleep, and social functioning, the investigators reported (JAMA 2014;312:240-248).
Patients in the intervention group received a greater number of analgesics for a greater length of time and at a higher mean dose than did those in the control group. However, only six patients in the entire study population initiated opioid use, and the median daily dose for all patients taking opioids was identical at the end of the study to what it was at the beginning. The two study groups showed no differences in their use of health care services such as outpatient visits, emergency department visits, and hospitalizations.
This study was supported by the U.S. Department of Veterans Affairs’ Health Services Research and Development Service. Dr. Kroenke reported receiving honoraria from Eli Lilly for work unrelated to this study.
A telecare intervention within the primary care practice setting elicited clinically meaningful and statistically significant improvements in long-standing chronic joint and other musculoskeletal pain, according to a report published online July 15 in JAMA.
A total of 250 adults with refractory, moderately severe regional (joints, limbs, back, or neck) or generalized (fibromyalgia or widespread) musculoskeletal pain of long duration participated in a randomized, blinded, 1-year clinical trial comparing the telecare intervention (124 patients) against usual chronic pain treatment (126 patients) in the primary care setting, reported Dr. Kurt Kroenke of the Veterans Affairs Center for Health Information and Communication, Roudebush VA Medical Center, Indianapolis, and his associates.
The intervention had three key components: frequent automated monitoring of patient symptoms using telephone or Internet voice recordings; nurse calls to patients who reported inadequate treatment response, adverse effects, or nonadherence; and analgesic management using a stepped-care algorithm, which was implemented by a nurse-manager who worked with a physician pain specialist, in collaboration with the primary care physician. The estimated total time spent per patient during the study year was 3-4 hours for the study nurse and 1 hour for the study physician.
Patients frequently checked in to the automated system to report their pain, anxiety, and depression symptoms; how difficult their pain made it for them to carry out usual activities; their adherence to medications; the degree of relief they obtained from pain medications; any change in pain, including the degree of improvement; adverse effects from their analgesics; and whether they wanted to change their medication regimen or speak to a nurse.
The medication algorithm called for the stepwise use of simple analgesics (acetaminophen and nonsteroidal anti-inflammatory drugs); tricyclic antidepressants (amitriptyline and nortriptyline) and cyclobenzaprine; tramadol; gabapentoids (gabapentin and pregabalin); topical analgesics; and opioids.
After 1 year, patients in the intervention group showed significantly greater improvement than the control group in measures of total pain, pain severity, and pain interference with daily activities, along with significantly greater satisfaction with their medical care. They were nearly twice as likely to report a 30% improvement from baseline (51.7%) than were patients in the usual-care group (27.1%).
They also were much less likely to report worsening pain (19.2% vs. 36.0%), which indicates a reduced risk of deterioration of their condition because their pain therapy was optimized, and much more likely to rate their medication as "good to excellent" (73.9% vs. 50.9%). And they reported greater improvements in secondary outcomes such as depression, anxiety, somatization, sleep, and social functioning, the investigators reported (JAMA 2014;312:240-248).
Patients in the intervention group received a greater number of analgesics for a greater length of time and at a higher mean dose than did those in the control group. However, only six patients in the entire study population initiated opioid use, and the median daily dose for all patients taking opioids was identical at the end of the study to what it was at the beginning. The two study groups showed no differences in their use of health care services such as outpatient visits, emergency department visits, and hospitalizations.
This study was supported by the U.S. Department of Veterans Affairs’ Health Services Research and Development Service. Dr. Kroenke reported receiving honoraria from Eli Lilly for work unrelated to this study.
FROM JAMA
Key clinical finding: A hands-off telecare approach helps patients with chronic joint and other musculoskeletal pain manage it.
Major finding: Patients in the intervention group showed significantly greater improvement than the control group in measures of total pain, pain severity, and pain interference with daily activities; they were nearly twice as likely to report a 30% improvement from baseline (51.7%) than were patients in the usual-care group (27.1%), much less likely to report worsening pain (19.2% vs. 36.0%), and much more likely to rate their medication as "good to excellent" (73.9% vs. 50.9%).
Data source: A 1-year randomized, controlled trial involving 250 adults with refractory, moderately severe, chronic musculoskeletal pain.
Disclosures: This study was supported by the U.S. Department of Veterans Affairs’ Health Services Research and Development Service.
Patients become full partners in inflammatory arthritis and psoriasis research
NEW YORK – Improvements in clinical trial design for psoriasis, psoriatic arthritis, and other inflammatory diseases are being credited to a decision to enlist patients as full partners, not just advisors or consultants, in research initiatives.
"Rheumatologists, I think, have been leaders is recognizing that patients can bring an expertise to clinical research that is unique and ensure that study endpoints are relevant to outcomes important to them," reported Dr. William Tillett, a research fellow in the department of rheumatology at the Royal National Hospital for Rheumatic Diseases in Bath, England.
The value of patient research partners (PRP) was a recurring theme at the joint meetings of the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis (GRAPPA) and the Spondyloarthritis Research & Treatment Network. GRAPPA, in particular, has been fostering research collaborations with patients since 2006.
"It has been an evolution. Patients were initially enlisted to sit in when trial designs were being discussed. Now, we are talking about full partnership so that they are involved at the inception with equal partnership that includes veto power and author credit," Dr. Tillett explained.
At the GRAPPA meeting, an afternoon symposium was devoted to optimal strategies for fostering collaboration with PRPs, which builds on work already initiated with a group called Patient Involvement for Outcome Measures in PsA (PIOMPsA). In turn, PIOMPsA, formed 2 years ago, was largely modeled on PRP initiatives led by OMERACT (Outcomes Measurement in Rheumatology).
"OMERACT has been involving patients in clinical research design for about 10 years," reported Dr. Philip J. Mease, director of rheumatology research at Swedish Medical Center, Seattle. "The idea of making patients full-blown partners is more recent, but I think there is increasing appreciation for what the right patients can contribute to improve study design."
The initiatives are spreading through rheumatology and other inflammatory diseases. At the GRAPPA meeting, updates on patient initiatives to influence clinical research were presented not only from the work of PIOMPsA and OMERACT but also from the International Dermatology Outcomes Measures (IDEOM) consortium. Created in collaboration with the National Psoriasis Foundation (NPF), IDEOM is bringing clinicians and patients together to define standard outcome measures.
"There are no really good measures to evaluate relative severity of psoriasis from the patient’s perspective," reported Dr. Alice Gottlieb, professor of dermatology at Tufts Medical Center, Boston. Tools traditionally used in clinical trials, such as the Psoriasis Area Severity Index (PASI) "are not practical in the clinic" and do not necessarily reflect the impact of psoriasis on quality of life when used as a study endpoint, she said.
PIOMPsA has now conducted several meetings, including one held in conjunction with OMERACT in Budapest, Hungary, in early May 2014. Like IDEOM, PIOMPsA has been focused on developing consensus on core symptoms of its target inflammatory disease. This is critical because the ability of clinical trials to generate relevant data is dependent on first defining meaningful endpoints, according to Dr. Tillett, who presented the PIOMPsA deliberations at the GRAPPA meeting.
In Budapest, for example, a vote was taken on whether to add fatigue to a list of core symptoms for PsA that includes impaired physical function, skin lesions, and joint pain. Fatigue was added to the list by a vote in which 70% supported it as a core PsA symptom, said Dr. Tillett, who recently published on the goals and underlying concepts of PIOMPsA (Curr. Rheumatol. Rep. 2014;16:418).
According to Dr. Tillett and Dr. Mease, PRPs are an answer to the repeatedly reported disconnect between physicians and patients in rating disease severity. By involving patients with interest in clinical research and collaborative skills, treatment trials have the potential to generate data more useful to practical patient management.
"It will be very difficult to show objectively that patient-aided trial design leads to better studies, but this is a reasonable expectation. I think that the contributions we have already seen from these collaborations bear this out," said Dr. Tillett, who expects the concept to spread to other fields of medicine.
Dr. Tillett reported financial relationships with AbbVie and Amgen. Dr. Mease reported financial relationships with AbbVie, Amgen, Biogen Idec, Bristol-Myers Squibb, Celgene, Crescendo Bioscience, Genentech, Janssen Pharmaceuticals, Lilly, Merck & Co, Novartis, Pfizer, UCB Pharma, and Vertex. Dr. Gottlieb reported financial relationships with Abbott, Actelion, Amgen, Bristol-Myers Squibb, Celgene, Centocor, Novo Nordisk, Teva, and UCB.
NEW YORK – Improvements in clinical trial design for psoriasis, psoriatic arthritis, and other inflammatory diseases are being credited to a decision to enlist patients as full partners, not just advisors or consultants, in research initiatives.
"Rheumatologists, I think, have been leaders is recognizing that patients can bring an expertise to clinical research that is unique and ensure that study endpoints are relevant to outcomes important to them," reported Dr. William Tillett, a research fellow in the department of rheumatology at the Royal National Hospital for Rheumatic Diseases in Bath, England.
The value of patient research partners (PRP) was a recurring theme at the joint meetings of the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis (GRAPPA) and the Spondyloarthritis Research & Treatment Network. GRAPPA, in particular, has been fostering research collaborations with patients since 2006.
"It has been an evolution. Patients were initially enlisted to sit in when trial designs were being discussed. Now, we are talking about full partnership so that they are involved at the inception with equal partnership that includes veto power and author credit," Dr. Tillett explained.
At the GRAPPA meeting, an afternoon symposium was devoted to optimal strategies for fostering collaboration with PRPs, which builds on work already initiated with a group called Patient Involvement for Outcome Measures in PsA (PIOMPsA). In turn, PIOMPsA, formed 2 years ago, was largely modeled on PRP initiatives led by OMERACT (Outcomes Measurement in Rheumatology).
"OMERACT has been involving patients in clinical research design for about 10 years," reported Dr. Philip J. Mease, director of rheumatology research at Swedish Medical Center, Seattle. "The idea of making patients full-blown partners is more recent, but I think there is increasing appreciation for what the right patients can contribute to improve study design."
The initiatives are spreading through rheumatology and other inflammatory diseases. At the GRAPPA meeting, updates on patient initiatives to influence clinical research were presented not only from the work of PIOMPsA and OMERACT but also from the International Dermatology Outcomes Measures (IDEOM) consortium. Created in collaboration with the National Psoriasis Foundation (NPF), IDEOM is bringing clinicians and patients together to define standard outcome measures.
"There are no really good measures to evaluate relative severity of psoriasis from the patient’s perspective," reported Dr. Alice Gottlieb, professor of dermatology at Tufts Medical Center, Boston. Tools traditionally used in clinical trials, such as the Psoriasis Area Severity Index (PASI) "are not practical in the clinic" and do not necessarily reflect the impact of psoriasis on quality of life when used as a study endpoint, she said.
PIOMPsA has now conducted several meetings, including one held in conjunction with OMERACT in Budapest, Hungary, in early May 2014. Like IDEOM, PIOMPsA has been focused on developing consensus on core symptoms of its target inflammatory disease. This is critical because the ability of clinical trials to generate relevant data is dependent on first defining meaningful endpoints, according to Dr. Tillett, who presented the PIOMPsA deliberations at the GRAPPA meeting.
In Budapest, for example, a vote was taken on whether to add fatigue to a list of core symptoms for PsA that includes impaired physical function, skin lesions, and joint pain. Fatigue was added to the list by a vote in which 70% supported it as a core PsA symptom, said Dr. Tillett, who recently published on the goals and underlying concepts of PIOMPsA (Curr. Rheumatol. Rep. 2014;16:418).
According to Dr. Tillett and Dr. Mease, PRPs are an answer to the repeatedly reported disconnect between physicians and patients in rating disease severity. By involving patients with interest in clinical research and collaborative skills, treatment trials have the potential to generate data more useful to practical patient management.
"It will be very difficult to show objectively that patient-aided trial design leads to better studies, but this is a reasonable expectation. I think that the contributions we have already seen from these collaborations bear this out," said Dr. Tillett, who expects the concept to spread to other fields of medicine.
Dr. Tillett reported financial relationships with AbbVie and Amgen. Dr. Mease reported financial relationships with AbbVie, Amgen, Biogen Idec, Bristol-Myers Squibb, Celgene, Crescendo Bioscience, Genentech, Janssen Pharmaceuticals, Lilly, Merck & Co, Novartis, Pfizer, UCB Pharma, and Vertex. Dr. Gottlieb reported financial relationships with Abbott, Actelion, Amgen, Bristol-Myers Squibb, Celgene, Centocor, Novo Nordisk, Teva, and UCB.
NEW YORK – Improvements in clinical trial design for psoriasis, psoriatic arthritis, and other inflammatory diseases are being credited to a decision to enlist patients as full partners, not just advisors or consultants, in research initiatives.
"Rheumatologists, I think, have been leaders is recognizing that patients can bring an expertise to clinical research that is unique and ensure that study endpoints are relevant to outcomes important to them," reported Dr. William Tillett, a research fellow in the department of rheumatology at the Royal National Hospital for Rheumatic Diseases in Bath, England.
The value of patient research partners (PRP) was a recurring theme at the joint meetings of the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis (GRAPPA) and the Spondyloarthritis Research & Treatment Network. GRAPPA, in particular, has been fostering research collaborations with patients since 2006.
"It has been an evolution. Patients were initially enlisted to sit in when trial designs were being discussed. Now, we are talking about full partnership so that they are involved at the inception with equal partnership that includes veto power and author credit," Dr. Tillett explained.
At the GRAPPA meeting, an afternoon symposium was devoted to optimal strategies for fostering collaboration with PRPs, which builds on work already initiated with a group called Patient Involvement for Outcome Measures in PsA (PIOMPsA). In turn, PIOMPsA, formed 2 years ago, was largely modeled on PRP initiatives led by OMERACT (Outcomes Measurement in Rheumatology).
"OMERACT has been involving patients in clinical research design for about 10 years," reported Dr. Philip J. Mease, director of rheumatology research at Swedish Medical Center, Seattle. "The idea of making patients full-blown partners is more recent, but I think there is increasing appreciation for what the right patients can contribute to improve study design."
The initiatives are spreading through rheumatology and other inflammatory diseases. At the GRAPPA meeting, updates on patient initiatives to influence clinical research were presented not only from the work of PIOMPsA and OMERACT but also from the International Dermatology Outcomes Measures (IDEOM) consortium. Created in collaboration with the National Psoriasis Foundation (NPF), IDEOM is bringing clinicians and patients together to define standard outcome measures.
"There are no really good measures to evaluate relative severity of psoriasis from the patient’s perspective," reported Dr. Alice Gottlieb, professor of dermatology at Tufts Medical Center, Boston. Tools traditionally used in clinical trials, such as the Psoriasis Area Severity Index (PASI) "are not practical in the clinic" and do not necessarily reflect the impact of psoriasis on quality of life when used as a study endpoint, she said.
PIOMPsA has now conducted several meetings, including one held in conjunction with OMERACT in Budapest, Hungary, in early May 2014. Like IDEOM, PIOMPsA has been focused on developing consensus on core symptoms of its target inflammatory disease. This is critical because the ability of clinical trials to generate relevant data is dependent on first defining meaningful endpoints, according to Dr. Tillett, who presented the PIOMPsA deliberations at the GRAPPA meeting.
In Budapest, for example, a vote was taken on whether to add fatigue to a list of core symptoms for PsA that includes impaired physical function, skin lesions, and joint pain. Fatigue was added to the list by a vote in which 70% supported it as a core PsA symptom, said Dr. Tillett, who recently published on the goals and underlying concepts of PIOMPsA (Curr. Rheumatol. Rep. 2014;16:418).
According to Dr. Tillett and Dr. Mease, PRPs are an answer to the repeatedly reported disconnect between physicians and patients in rating disease severity. By involving patients with interest in clinical research and collaborative skills, treatment trials have the potential to generate data more useful to practical patient management.
"It will be very difficult to show objectively that patient-aided trial design leads to better studies, but this is a reasonable expectation. I think that the contributions we have already seen from these collaborations bear this out," said Dr. Tillett, who expects the concept to spread to other fields of medicine.
Dr. Tillett reported financial relationships with AbbVie and Amgen. Dr. Mease reported financial relationships with AbbVie, Amgen, Biogen Idec, Bristol-Myers Squibb, Celgene, Crescendo Bioscience, Genentech, Janssen Pharmaceuticals, Lilly, Merck & Co, Novartis, Pfizer, UCB Pharma, and Vertex. Dr. Gottlieb reported financial relationships with Abbott, Actelion, Amgen, Bristol-Myers Squibb, Celgene, Centocor, Novo Nordisk, Teva, and UCB.
EXPERT ANALYSIS FROM THE 2014 GRAPPA AND SPARTAN ANNUAL MEETINGS
Long-acting–opioid prescribing rate highest in Maine
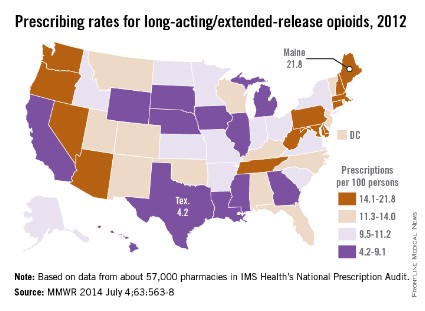
The prescribing rate for long-acting/extended-release opioid pain relievers in Maine, which is the highest in the country, is more than five times higher than that of Texas, which has the lowest rate, the Centers for Disease Control and Prevention reported.
Maine’s prescribing rate for long-acting/extended-release opioid pain relievers (LA/ER OPRs) was 21.8 per 100 persons in 2012, compared with 4.2 per 100 in Texas, making Maine’s rate 5.2 times higher. Delaware had the second-highest LA/ER OPR rate – just behind Maine at 21.7 per 100. After Texas, the next-lowest state was Illinois, which had a prescribing rate of 5.2 per 100, according to CDC investigators (MMWR 2014;63:563-7).
The national rate for LA/ER OPRs was 10.3 per 100 persons, or about 12.5% of the total 258.9 million opioid prescriptions written in 2012. By region, the Northeast was the highest at 12.6 per 100, followed by the South (10.2), the West (9.6), and the Midwest (9.3), they said.
The CDC researchers defined LA/ER OPRs "as those that should be taken only two or three times a day, such as methadone, OxyContin [oxycodone], and Opana [oxymorphone] ER." The analysis was based on a sample of approximately 57,000 pharmacies from IMS Health’s National Prescription Audit that "dispense nearly 80% of the retail prescriptions in the United States," they wrote.
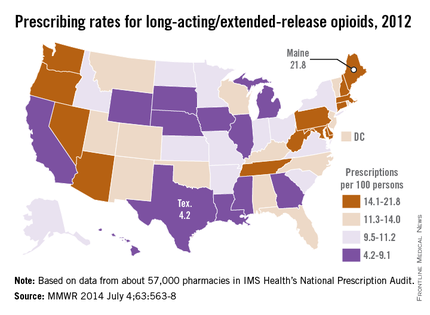
The prescribing rate for long-acting/extended-release opioid pain relievers in Maine, which is the highest in the country, is more than five times higher than that of Texas, which has the lowest rate, the Centers for Disease Control and Prevention reported.
Maine’s prescribing rate for long-acting/extended-release opioid pain relievers (LA/ER OPRs) was 21.8 per 100 persons in 2012, compared with 4.2 per 100 in Texas, making Maine’s rate 5.2 times higher. Delaware had the second-highest LA/ER OPR rate – just behind Maine at 21.7 per 100. After Texas, the next-lowest state was Illinois, which had a prescribing rate of 5.2 per 100, according to CDC investigators (MMWR 2014;63:563-7).
The national rate for LA/ER OPRs was 10.3 per 100 persons, or about 12.5% of the total 258.9 million opioid prescriptions written in 2012. By region, the Northeast was the highest at 12.6 per 100, followed by the South (10.2), the West (9.6), and the Midwest (9.3), they said.
The CDC researchers defined LA/ER OPRs "as those that should be taken only two or three times a day, such as methadone, OxyContin [oxycodone], and Opana [oxymorphone] ER." The analysis was based on a sample of approximately 57,000 pharmacies from IMS Health’s National Prescription Audit that "dispense nearly 80% of the retail prescriptions in the United States," they wrote.

The prescribing rate for long-acting/extended-release opioid pain relievers in Maine, which is the highest in the country, is more than five times higher than that of Texas, which has the lowest rate, the Centers for Disease Control and Prevention reported.
Maine’s prescribing rate for long-acting/extended-release opioid pain relievers (LA/ER OPRs) was 21.8 per 100 persons in 2012, compared with 4.2 per 100 in Texas, making Maine’s rate 5.2 times higher. Delaware had the second-highest LA/ER OPR rate – just behind Maine at 21.7 per 100. After Texas, the next-lowest state was Illinois, which had a prescribing rate of 5.2 per 100, according to CDC investigators (MMWR 2014;63:563-7).
The national rate for LA/ER OPRs was 10.3 per 100 persons, or about 12.5% of the total 258.9 million opioid prescriptions written in 2012. By region, the Northeast was the highest at 12.6 per 100, followed by the South (10.2), the West (9.6), and the Midwest (9.3), they said.
The CDC researchers defined LA/ER OPRs "as those that should be taken only two or three times a day, such as methadone, OxyContin [oxycodone], and Opana [oxymorphone] ER." The analysis was based on a sample of approximately 57,000 pharmacies from IMS Health’s National Prescription Audit that "dispense nearly 80% of the retail prescriptions in the United States," they wrote.

FROM MMWR
VIDEO: Does obesity’s effect on RA support different treatment goals?
PARIS – Treat-to-target goals for obese patients with rheumatoid arthritis should take into account new research indicating that they already have a higher level of systemic inflammation and higher Disease Activity Scores than do normal-weight patients, according to Dr. Christopher Sparks of the University of Liverpool, England.
In an international sample of 3,534 patients with rheumatoid arthritis (RA), Dr. Sparks and his colleagues found that those with a body mass index of 30-34.9 kg/m2 (obese) or 35 kg/m2 or greater (obese II) had higher 28-joint Disease Activity Scores (DAS28) than did normal-weight patients, a difference that was largely driven by higher erythrocyte sedimentation rates and higher Visual Analog Scale scores, rather than higher tender and swollen joint counts. The data were reported at the annual European Congress of Rheumatology.
It cannot be known whether obese patients with higher DAS28 scores have clinically more severe disease, so it leads one to wonder, "Is it just an inflated DAS28 score that’s really driving this? And so potentially it brings up the question, Is obesity a confounding factor when looking at DAS28 scores in the RA population?" Dr. Sparks said in a video interview with Dr. Christian Beyer of the University of Erlangen-Nuremberg, Germany.
"If there is an artificial rise in the DAS28 in all of our obese patients out there, should the treatment goals in terms of remission or response be potentially slightly different for obese patients, compared to normal-weight patients, because they have a baseline higher DAS28?"
PARIS – Treat-to-target goals for obese patients with rheumatoid arthritis should take into account new research indicating that they already have a higher level of systemic inflammation and higher Disease Activity Scores than do normal-weight patients, according to Dr. Christopher Sparks of the University of Liverpool, England.
In an international sample of 3,534 patients with rheumatoid arthritis (RA), Dr. Sparks and his colleagues found that those with a body mass index of 30-34.9 kg/m2 (obese) or 35 kg/m2 or greater (obese II) had higher 28-joint Disease Activity Scores (DAS28) than did normal-weight patients, a difference that was largely driven by higher erythrocyte sedimentation rates and higher Visual Analog Scale scores, rather than higher tender and swollen joint counts. The data were reported at the annual European Congress of Rheumatology.
It cannot be known whether obese patients with higher DAS28 scores have clinically more severe disease, so it leads one to wonder, "Is it just an inflated DAS28 score that’s really driving this? And so potentially it brings up the question, Is obesity a confounding factor when looking at DAS28 scores in the RA population?" Dr. Sparks said in a video interview with Dr. Christian Beyer of the University of Erlangen-Nuremberg, Germany.
"If there is an artificial rise in the DAS28 in all of our obese patients out there, should the treatment goals in terms of remission or response be potentially slightly different for obese patients, compared to normal-weight patients, because they have a baseline higher DAS28?"
PARIS – Treat-to-target goals for obese patients with rheumatoid arthritis should take into account new research indicating that they already have a higher level of systemic inflammation and higher Disease Activity Scores than do normal-weight patients, according to Dr. Christopher Sparks of the University of Liverpool, England.
In an international sample of 3,534 patients with rheumatoid arthritis (RA), Dr. Sparks and his colleagues found that those with a body mass index of 30-34.9 kg/m2 (obese) or 35 kg/m2 or greater (obese II) had higher 28-joint Disease Activity Scores (DAS28) than did normal-weight patients, a difference that was largely driven by higher erythrocyte sedimentation rates and higher Visual Analog Scale scores, rather than higher tender and swollen joint counts. The data were reported at the annual European Congress of Rheumatology.
It cannot be known whether obese patients with higher DAS28 scores have clinically more severe disease, so it leads one to wonder, "Is it just an inflated DAS28 score that’s really driving this? And so potentially it brings up the question, Is obesity a confounding factor when looking at DAS28 scores in the RA population?" Dr. Sparks said in a video interview with Dr. Christian Beyer of the University of Erlangen-Nuremberg, Germany.
"If there is an artificial rise in the DAS28 in all of our obese patients out there, should the treatment goals in terms of remission or response be potentially slightly different for obese patients, compared to normal-weight patients, because they have a baseline higher DAS28?"
AT THE EULAR CONGRESS 2014
Arthritis drug restores hair in man with alopecia universalis and psoriasis
A 25-year-old man with plaque psoriasis and virtually no hair of any sort now sports a full head of hair plus body hair after treatment with the arthritis drug tofacitinib, according to Dr. Brittany G. Craiglow and Dr. Brett A. King of Yale University, New Haven, Conn.
The treatment has been so successful that Dr. King has submitted a proposal for a clinical trial involving a cream form of tofacitinib as a treatment for alopecia areata, according to a statement from the university.
Tofacitinib is approved only for the treatment of adult patients with moderately to severely active rheumatoid arthritis who have had an inadequate response or intolerance to methotrexate, but it is in clinical development for the treatment of psoriasis.
Dr. Craiglow and Dr. King began treating the patient with 10 mg oral tofacitinib (Xeljanz) daily. At baseline, the patient had been diagnosed with plaque psoriasis and alopecia universalis. His only body hair was a small amount of hair within the psoriasis plaques on his head, the researchers said (J. Invest. Dermatol. 2014 June 18 [doi:10.1038/jid.2014.260]).
After 2 months of tofacitinib dosed at 5 mg twice daily, the psoriasis on the patient’s scalp, torso, and elbows showed some improvement, and there was some hair growth on his face and scalp. The researchers increased the dose to 10 mg in the morning and 5 mg at night. After 3 more months, the patient had complete regrowth of scalp hair, as well as some growth of eyebrows, eyelashes, armpit hair, and pubic hair. After 8 months, the patient had full regrowth of all body hair, with the exception of hair on the arms and legs (which had been sparse prior to his alopecia diagnosis, the researchers said).
Although the hair growth has been dramatic, improvements in the patient’s psoriasis have been slower, likely because of the dosage.
“While we considered increasing the dose of tofacitinib, the patient is so pleased with the regrowth of his hair (and is not particularly bothered by the remaining psoriasis) that he has chosen to continue at the present dose,” the researchers noted.
The researchers considered using tofacitinib to treat the patient’s alopecia universalis based on the research of Angela M. Christiano, Ph.D., of Columbia University, New York, in which the drug reversed hair loss in a mouse model of alopecia areata.
The patient has reported no side effects, and lab testing has shown no abnormalities in complete blood count, serum creatinine, electrolytes, liver function, glucose, or lipids, the researchers noted.
The researchers had no financial conflicts to disclose.
hsplete@frontlinemedcom.com
A 25-year-old man with plaque psoriasis and virtually no hair of any sort now sports a full head of hair plus body hair after treatment with the arthritis drug tofacitinib, according to Dr. Brittany G. Craiglow and Dr. Brett A. King of Yale University, New Haven, Conn.
The treatment has been so successful that Dr. King has submitted a proposal for a clinical trial involving a cream form of tofacitinib as a treatment for alopecia areata, according to a statement from the university.
Tofacitinib is approved only for the treatment of adult patients with moderately to severely active rheumatoid arthritis who have had an inadequate response or intolerance to methotrexate, but it is in clinical development for the treatment of psoriasis.
Dr. Craiglow and Dr. King began treating the patient with 10 mg oral tofacitinib (Xeljanz) daily. At baseline, the patient had been diagnosed with plaque psoriasis and alopecia universalis. His only body hair was a small amount of hair within the psoriasis plaques on his head, the researchers said (J. Invest. Dermatol. 2014 June 18 [doi:10.1038/jid.2014.260]).
After 2 months of tofacitinib dosed at 5 mg twice daily, the psoriasis on the patient’s scalp, torso, and elbows showed some improvement, and there was some hair growth on his face and scalp. The researchers increased the dose to 10 mg in the morning and 5 mg at night. After 3 more months, the patient had complete regrowth of scalp hair, as well as some growth of eyebrows, eyelashes, armpit hair, and pubic hair. After 8 months, the patient had full regrowth of all body hair, with the exception of hair on the arms and legs (which had been sparse prior to his alopecia diagnosis, the researchers said).
Although the hair growth has been dramatic, improvements in the patient’s psoriasis have been slower, likely because of the dosage.
“While we considered increasing the dose of tofacitinib, the patient is so pleased with the regrowth of his hair (and is not particularly bothered by the remaining psoriasis) that he has chosen to continue at the present dose,” the researchers noted.
The researchers considered using tofacitinib to treat the patient’s alopecia universalis based on the research of Angela M. Christiano, Ph.D., of Columbia University, New York, in which the drug reversed hair loss in a mouse model of alopecia areata.
The patient has reported no side effects, and lab testing has shown no abnormalities in complete blood count, serum creatinine, electrolytes, liver function, glucose, or lipids, the researchers noted.
The researchers had no financial conflicts to disclose.
hsplete@frontlinemedcom.com
A 25-year-old man with plaque psoriasis and virtually no hair of any sort now sports a full head of hair plus body hair after treatment with the arthritis drug tofacitinib, according to Dr. Brittany G. Craiglow and Dr. Brett A. King of Yale University, New Haven, Conn.
The treatment has been so successful that Dr. King has submitted a proposal for a clinical trial involving a cream form of tofacitinib as a treatment for alopecia areata, according to a statement from the university.
Tofacitinib is approved only for the treatment of adult patients with moderately to severely active rheumatoid arthritis who have had an inadequate response or intolerance to methotrexate, but it is in clinical development for the treatment of psoriasis.
Dr. Craiglow and Dr. King began treating the patient with 10 mg oral tofacitinib (Xeljanz) daily. At baseline, the patient had been diagnosed with plaque psoriasis and alopecia universalis. His only body hair was a small amount of hair within the psoriasis plaques on his head, the researchers said (J. Invest. Dermatol. 2014 June 18 [doi:10.1038/jid.2014.260]).
After 2 months of tofacitinib dosed at 5 mg twice daily, the psoriasis on the patient’s scalp, torso, and elbows showed some improvement, and there was some hair growth on his face and scalp. The researchers increased the dose to 10 mg in the morning and 5 mg at night. After 3 more months, the patient had complete regrowth of scalp hair, as well as some growth of eyebrows, eyelashes, armpit hair, and pubic hair. After 8 months, the patient had full regrowth of all body hair, with the exception of hair on the arms and legs (which had been sparse prior to his alopecia diagnosis, the researchers said).
Although the hair growth has been dramatic, improvements in the patient’s psoriasis have been slower, likely because of the dosage.
“While we considered increasing the dose of tofacitinib, the patient is so pleased with the regrowth of his hair (and is not particularly bothered by the remaining psoriasis) that he has chosen to continue at the present dose,” the researchers noted.
The researchers considered using tofacitinib to treat the patient’s alopecia universalis based on the research of Angela M. Christiano, Ph.D., of Columbia University, New York, in which the drug reversed hair loss in a mouse model of alopecia areata.
The patient has reported no side effects, and lab testing has shown no abnormalities in complete blood count, serum creatinine, electrolytes, liver function, glucose, or lipids, the researchers noted.
The researchers had no financial conflicts to disclose.
hsplete@frontlinemedcom.com
Arthritis drug restores hair in man with alopecia universalis and psoriasis
A 25-year-old man with plaque psoriasis and virtually no hair of any sort now sports a full head of hair plus body hair after treatment with the arthritis drug tofacitinib, according to Dr. Brittany G. Craiglow and Dr. Brett A. King of Yale University, New Haven, Conn.
The treatment has been so successful that Dr. King has submitted a proposal for a clinical trial involving a cream form of tofacitinib as a treatment for alopecia areata, according to a statement from the university.
Tofacitinib is approved only for the treatment of adult patients with moderately to severely active rheumatoid arthritis who have had an inadequate response or intolerance to methotrexate, but it is in clinical development for the treatment of psoriasis.
Dr. Craiglow and Dr. King began treating the patient with 10 mg oral tofacitinib (Xeljanz) daily. At baseline, the patient had been diagnosed with plaque psoriasis and alopecia universalis. His only body hair was a small amount of hair within the psoriasis plaques on his head, the researchers said (J. Invest. Dermatol. 2014 June 18 [doi:10.1038/jid.2014.260]).
After 2 months of tofacitinib dosed at 5 mg twice daily, the psoriasis on the patient’s scalp, torso, and elbows showed some improvement, and there was some hair growth on his face and scalp. The researchers increased the dose to 10 mg in the morning and 5 mg at night. After 3 more months, the patient had complete regrowth of scalp hair, as well as some growth of eyebrows, eyelashes, armpit hair, and pubic hair. After 8 months, the patient had full regrowth of all body hair, with the exception of hair on the arms and legs (which had been sparse prior to his alopecia diagnosis, the researchers said).
Although the hair growth has been dramatic, improvements in the patient’s psoriasis have been slower, likely because of the dosage.
“While we considered increasing the dose of tofacitinib, the patient is so pleased with the regrowth of his hair (and is not particularly bothered by the remaining psoriasis) that he has chosen to continue at the present dose,” the researchers noted.
The researchers considered using tofacitinib to treat the patient’s alopecia universalis based on the research of Angela M. Christiano, Ph.D., of Columbia University, New York, in which the drug reversed hair loss in a mouse model of alopecia areata.
The patient has reported no side effects, and lab testing has shown no abnormalities in complete blood count, serum creatinine, electrolytes, liver function, glucose, or lipids, the researchers noted.
The researchers had no financial conflicts to disclose.
hsplete@frontlinemedcom.com
A 25-year-old man with plaque psoriasis and virtually no hair of any sort now sports a full head of hair plus body hair after treatment with the arthritis drug tofacitinib, according to Dr. Brittany G. Craiglow and Dr. Brett A. King of Yale University, New Haven, Conn.
The treatment has been so successful that Dr. King has submitted a proposal for a clinical trial involving a cream form of tofacitinib as a treatment for alopecia areata, according to a statement from the university.
Tofacitinib is approved only for the treatment of adult patients with moderately to severely active rheumatoid arthritis who have had an inadequate response or intolerance to methotrexate, but it is in clinical development for the treatment of psoriasis.
Dr. Craiglow and Dr. King began treating the patient with 10 mg oral tofacitinib (Xeljanz) daily. At baseline, the patient had been diagnosed with plaque psoriasis and alopecia universalis. His only body hair was a small amount of hair within the psoriasis plaques on his head, the researchers said (J. Invest. Dermatol. 2014 June 18 [doi:10.1038/jid.2014.260]).
After 2 months of tofacitinib dosed at 5 mg twice daily, the psoriasis on the patient’s scalp, torso, and elbows showed some improvement, and there was some hair growth on his face and scalp. The researchers increased the dose to 10 mg in the morning and 5 mg at night. After 3 more months, the patient had complete regrowth of scalp hair, as well as some growth of eyebrows, eyelashes, armpit hair, and pubic hair. After 8 months, the patient had full regrowth of all body hair, with the exception of hair on the arms and legs (which had been sparse prior to his alopecia diagnosis, the researchers said).
Although the hair growth has been dramatic, improvements in the patient’s psoriasis have been slower, likely because of the dosage.
“While we considered increasing the dose of tofacitinib, the patient is so pleased with the regrowth of his hair (and is not particularly bothered by the remaining psoriasis) that he has chosen to continue at the present dose,” the researchers noted.
The researchers considered using tofacitinib to treat the patient’s alopecia universalis based on the research of Angela M. Christiano, Ph.D., of Columbia University, New York, in which the drug reversed hair loss in a mouse model of alopecia areata.
The patient has reported no side effects, and lab testing has shown no abnormalities in complete blood count, serum creatinine, electrolytes, liver function, glucose, or lipids, the researchers noted.
The researchers had no financial conflicts to disclose.
hsplete@frontlinemedcom.com
A 25-year-old man with plaque psoriasis and virtually no hair of any sort now sports a full head of hair plus body hair after treatment with the arthritis drug tofacitinib, according to Dr. Brittany G. Craiglow and Dr. Brett A. King of Yale University, New Haven, Conn.
The treatment has been so successful that Dr. King has submitted a proposal for a clinical trial involving a cream form of tofacitinib as a treatment for alopecia areata, according to a statement from the university.
Tofacitinib is approved only for the treatment of adult patients with moderately to severely active rheumatoid arthritis who have had an inadequate response or intolerance to methotrexate, but it is in clinical development for the treatment of psoriasis.
Dr. Craiglow and Dr. King began treating the patient with 10 mg oral tofacitinib (Xeljanz) daily. At baseline, the patient had been diagnosed with plaque psoriasis and alopecia universalis. His only body hair was a small amount of hair within the psoriasis plaques on his head, the researchers said (J. Invest. Dermatol. 2014 June 18 [doi:10.1038/jid.2014.260]).
After 2 months of tofacitinib dosed at 5 mg twice daily, the psoriasis on the patient’s scalp, torso, and elbows showed some improvement, and there was some hair growth on his face and scalp. The researchers increased the dose to 10 mg in the morning and 5 mg at night. After 3 more months, the patient had complete regrowth of scalp hair, as well as some growth of eyebrows, eyelashes, armpit hair, and pubic hair. After 8 months, the patient had full regrowth of all body hair, with the exception of hair on the arms and legs (which had been sparse prior to his alopecia diagnosis, the researchers said).
Although the hair growth has been dramatic, improvements in the patient’s psoriasis have been slower, likely because of the dosage.
“While we considered increasing the dose of tofacitinib, the patient is so pleased with the regrowth of his hair (and is not particularly bothered by the remaining psoriasis) that he has chosen to continue at the present dose,” the researchers noted.
The researchers considered using tofacitinib to treat the patient’s alopecia universalis based on the research of Angela M. Christiano, Ph.D., of Columbia University, New York, in which the drug reversed hair loss in a mouse model of alopecia areata.
The patient has reported no side effects, and lab testing has shown no abnormalities in complete blood count, serum creatinine, electrolytes, liver function, glucose, or lipids, the researchers noted.
The researchers had no financial conflicts to disclose.
hsplete@frontlinemedcom.com
Obesity may drive symptom severity in rheumatoid arthritis
PARIS – The higher disease activity scores seen in obese patients with rheumatoid arthritis may be driven by the proinflammatory state that is associated with obesity, Dr. Christopher Sparks reported in a press conference at the annual European Congress of Rheumatology.
Although obese RA patients tend to have less radiographic joint damage than do normal-weight RA patients, they have comparable DAS (disease activity score) 28. With the clinical focus on treat-to-target, an approach guided by DAS scores, obese RA patients may be getting more aggressive treatment. The finding may explain why obese patients with RA have better outcomes than normal-weight and thin RA patients, said Dr. Sparks, a clinical research fellow at the University of Liverpool, England.
Dr. Sparks and his colleagues used an international RA database to examine two patient subgroups: those diagnosed with RA in the previous year and those with longstanding RA. The 3,534 patients were stratified by their body mass index into five groups: underweight (less than 18.5 kg/m2), normal weight (18.5-24.9 kg/m2), overweight (25-29.9 kg/m2), obese (30-34.9 kg/m2), and obese II (35 kg/m2 or more). In the 1,981 patients with longstanding disease, median disease duration was about 7 years; the other 1,553 patients had disease duration of 1 year or less.
About 73% of the each cohort was female, and the distribution of BMI measures was similar for the two cohorts.
The groups were compared by BMI and RA disease measures that included DAS28, erythrocyte sedimentation rate, tender joint count; swollen joint count; and visual analog scale disease activity.
After adjusting for RA risk factors, obesity (BMI of 28 kg/m2 or greater) was significantly associated with a DAS28 exceeding 5.1, an elevated erythrocyte sedimentation rate, high tender joint count, and high visual analog scale score. For instance, compared with normal weight and overweight patients, underweight and both groups of obese patients were 1.5-2.2 times as likely to have a DAS28 exceeding 5.1, Dr. Sparks reported.
He had no financial disclosures.
PARIS – The higher disease activity scores seen in obese patients with rheumatoid arthritis may be driven by the proinflammatory state that is associated with obesity, Dr. Christopher Sparks reported in a press conference at the annual European Congress of Rheumatology.
Although obese RA patients tend to have less radiographic joint damage than do normal-weight RA patients, they have comparable DAS (disease activity score) 28. With the clinical focus on treat-to-target, an approach guided by DAS scores, obese RA patients may be getting more aggressive treatment. The finding may explain why obese patients with RA have better outcomes than normal-weight and thin RA patients, said Dr. Sparks, a clinical research fellow at the University of Liverpool, England.
Dr. Sparks and his colleagues used an international RA database to examine two patient subgroups: those diagnosed with RA in the previous year and those with longstanding RA. The 3,534 patients were stratified by their body mass index into five groups: underweight (less than 18.5 kg/m2), normal weight (18.5-24.9 kg/m2), overweight (25-29.9 kg/m2), obese (30-34.9 kg/m2), and obese II (35 kg/m2 or more). In the 1,981 patients with longstanding disease, median disease duration was about 7 years; the other 1,553 patients had disease duration of 1 year or less.
About 73% of the each cohort was female, and the distribution of BMI measures was similar for the two cohorts.
The groups were compared by BMI and RA disease measures that included DAS28, erythrocyte sedimentation rate, tender joint count; swollen joint count; and visual analog scale disease activity.
After adjusting for RA risk factors, obesity (BMI of 28 kg/m2 or greater) was significantly associated with a DAS28 exceeding 5.1, an elevated erythrocyte sedimentation rate, high tender joint count, and high visual analog scale score. For instance, compared with normal weight and overweight patients, underweight and both groups of obese patients were 1.5-2.2 times as likely to have a DAS28 exceeding 5.1, Dr. Sparks reported.
He had no financial disclosures.
PARIS – The higher disease activity scores seen in obese patients with rheumatoid arthritis may be driven by the proinflammatory state that is associated with obesity, Dr. Christopher Sparks reported in a press conference at the annual European Congress of Rheumatology.
Although obese RA patients tend to have less radiographic joint damage than do normal-weight RA patients, they have comparable DAS (disease activity score) 28. With the clinical focus on treat-to-target, an approach guided by DAS scores, obese RA patients may be getting more aggressive treatment. The finding may explain why obese patients with RA have better outcomes than normal-weight and thin RA patients, said Dr. Sparks, a clinical research fellow at the University of Liverpool, England.
Dr. Sparks and his colleagues used an international RA database to examine two patient subgroups: those diagnosed with RA in the previous year and those with longstanding RA. The 3,534 patients were stratified by their body mass index into five groups: underweight (less than 18.5 kg/m2), normal weight (18.5-24.9 kg/m2), overweight (25-29.9 kg/m2), obese (30-34.9 kg/m2), and obese II (35 kg/m2 or more). In the 1,981 patients with longstanding disease, median disease duration was about 7 years; the other 1,553 patients had disease duration of 1 year or less.
About 73% of the each cohort was female, and the distribution of BMI measures was similar for the two cohorts.
The groups were compared by BMI and RA disease measures that included DAS28, erythrocyte sedimentation rate, tender joint count; swollen joint count; and visual analog scale disease activity.
After adjusting for RA risk factors, obesity (BMI of 28 kg/m2 or greater) was significantly associated with a DAS28 exceeding 5.1, an elevated erythrocyte sedimentation rate, high tender joint count, and high visual analog scale score. For instance, compared with normal weight and overweight patients, underweight and both groups of obese patients were 1.5-2.2 times as likely to have a DAS28 exceeding 5.1, Dr. Sparks reported.
He had no financial disclosures.
AT THE EULAR CONGRESS 2014
Key clinical point: Obesity and underweight status are associated with worse symptoms of RA.
Major finding: Compared with normal weight and overweight patients, underweight and both groups of obese patients were 1.5-2.2 times as likely to have a DAS28 exceeding 5.1.
Data source: An international RA database sample of 3,534 patients.
Disclosures: Dr. Sparks had no financial disclosures.
Obesity may drive symptom severity in rheumatoid arthritis
PARIS – The higher disease activity scores seen in obese patients with rheumatoid arthritis may be driven by the proinflammatory state that is associated with obesity, Dr. Christopher Sparks reported in a press conference at the annual European Congress of Rheumatology.
Although obese RA patients tend to have less radiographic joint damage than do normal-weight RA patients, they have comparable DAS (disease activity score) 28. With the clinical focus on treat-to-target, an approach guided by DAS scores, obese RA patients may be getting more aggressive treatment. The finding may explain why obese patients with RA have better outcomes than normal-weight and thin RA patients, said Dr. Sparks, a clinical research fellow at the University of Liverpool, England.
Dr. Sparks and his colleagues used an international RA database to examine two patient subgroups: those diagnosed with RA in the previous year and those with longstanding RA. The 3,534 patients were stratified by their body mass index into five groups: underweight (less than 18.5 kg/m2), normal weight (18.5-24.9 kg/m2), overweight (25-29.9 kg/m2), obese (30-34.9 kg/m2), and obese II (35 kg/m2 or more). In the 1,981 patients with longstanding disease, median disease duration was about 7 years; the other 1,553 patients had disease duration of 1 year or less.
About 73% of the each cohort was female, and the distribution of BMI measures was similar for the two cohorts.
The groups were compared by BMI and RA disease measures that included DAS28, erythrocyte sedimentation rate, tender joint count; swollen joint count; and visual analog scale disease activity.
After adjusting for RA risk factors, obesity (BMI of 28 kg/m2 or greater) was significantly associated with a DAS28 exceeding 5.1, an elevated erythrocyte sedimentation rate, high tender joint count, and high visual analog scale score. For instance, compared with normal weight and overweight patients, underweight and both groups of obese patients were 1.5-2.2 times as likely to have a DAS28 exceeding 5.1, Dr. Sparks reported.
He had no financial disclosures.
PARIS – The higher disease activity scores seen in obese patients with rheumatoid arthritis may be driven by the proinflammatory state that is associated with obesity, Dr. Christopher Sparks reported in a press conference at the annual European Congress of Rheumatology.
Although obese RA patients tend to have less radiographic joint damage than do normal-weight RA patients, they have comparable DAS (disease activity score) 28. With the clinical focus on treat-to-target, an approach guided by DAS scores, obese RA patients may be getting more aggressive treatment. The finding may explain why obese patients with RA have better outcomes than normal-weight and thin RA patients, said Dr. Sparks, a clinical research fellow at the University of Liverpool, England.
Dr. Sparks and his colleagues used an international RA database to examine two patient subgroups: those diagnosed with RA in the previous year and those with longstanding RA. The 3,534 patients were stratified by their body mass index into five groups: underweight (less than 18.5 kg/m2), normal weight (18.5-24.9 kg/m2), overweight (25-29.9 kg/m2), obese (30-34.9 kg/m2), and obese II (35 kg/m2 or more). In the 1,981 patients with longstanding disease, median disease duration was about 7 years; the other 1,553 patients had disease duration of 1 year or less.
About 73% of the each cohort was female, and the distribution of BMI measures was similar for the two cohorts.
The groups were compared by BMI and RA disease measures that included DAS28, erythrocyte sedimentation rate, tender joint count; swollen joint count; and visual analog scale disease activity.
After adjusting for RA risk factors, obesity (BMI of 28 kg/m2 or greater) was significantly associated with a DAS28 exceeding 5.1, an elevated erythrocyte sedimentation rate, high tender joint count, and high visual analog scale score. For instance, compared with normal weight and overweight patients, underweight and both groups of obese patients were 1.5-2.2 times as likely to have a DAS28 exceeding 5.1, Dr. Sparks reported.
He had no financial disclosures.
PARIS – The higher disease activity scores seen in obese patients with rheumatoid arthritis may be driven by the proinflammatory state that is associated with obesity, Dr. Christopher Sparks reported in a press conference at the annual European Congress of Rheumatology.
Although obese RA patients tend to have less radiographic joint damage than do normal-weight RA patients, they have comparable DAS (disease activity score) 28. With the clinical focus on treat-to-target, an approach guided by DAS scores, obese RA patients may be getting more aggressive treatment. The finding may explain why obese patients with RA have better outcomes than normal-weight and thin RA patients, said Dr. Sparks, a clinical research fellow at the University of Liverpool, England.
Dr. Sparks and his colleagues used an international RA database to examine two patient subgroups: those diagnosed with RA in the previous year and those with longstanding RA. The 3,534 patients were stratified by their body mass index into five groups: underweight (less than 18.5 kg/m2), normal weight (18.5-24.9 kg/m2), overweight (25-29.9 kg/m2), obese (30-34.9 kg/m2), and obese II (35 kg/m2 or more). In the 1,981 patients with longstanding disease, median disease duration was about 7 years; the other 1,553 patients had disease duration of 1 year or less.
About 73% of the each cohort was female, and the distribution of BMI measures was similar for the two cohorts.
The groups were compared by BMI and RA disease measures that included DAS28, erythrocyte sedimentation rate, tender joint count; swollen joint count; and visual analog scale disease activity.
After adjusting for RA risk factors, obesity (BMI of 28 kg/m2 or greater) was significantly associated with a DAS28 exceeding 5.1, an elevated erythrocyte sedimentation rate, high tender joint count, and high visual analog scale score. For instance, compared with normal weight and overweight patients, underweight and both groups of obese patients were 1.5-2.2 times as likely to have a DAS28 exceeding 5.1, Dr. Sparks reported.
He had no financial disclosures.
AT THE EULAR CONGRESS 2014
Key clinical point: Obesity and underweight status are associated with worse symptoms of RA.
Major finding: Compared with normal weight and overweight patients, underweight and both groups of obese patients were 1.5-2.2 times as likely to have a DAS28 exceeding 5.1.
Data source: An international RA database sample of 3,534 patients.
Disclosures: Dr. Sparks had no financial disclosures.
VIDEO: Sarilumab shows RA safety, efficacy in phase III trial
PARIS – Treatment with the interleukin-6–blocking drug sarilumab led to statistically significant and clinically meaningful improvements in patients with rheumatoid arthritis in a multicenter, placebo controlled, phase III trial with 1,197 patients.
The safety and efficacy results from the SARIL-RA-MOBILITY trial are the first outcomes from a panel of phase III studies of sarilumab in patients with rheumatoid arthritis, said Dr. Mark Genovese at the annual European Congress of Rheumatology. The results showed that treatment with sarilumab plus methotrexate led to less joint damage over time, compared with methotrexate plus placebo, a benefit that should result in patients feeling better, said Dr. Genovese, professor of medicine and co-chief of the division of immunology and rheumatology at Stanford (Calif.) University.
Sarilumab is the first agent to progress this far in testing from a novel class of immunosuppressive drugs that work by blocking interleukin-6. Having safe drugs from a new class available to treat patients with rheumatoid arthritis holds promise for better controlling this disease in patients who inadequately respond to existing drug options, Dr. Genovese said in a video interview.
The SARIL-RA-MOBILITY trial was sponsored by Sanofi and Regeneron, the companies developing the drug. Dr. Genovese said that he has been a consultant to and received research support from Sanofi. Several of the coauthors are employees of Sanofi or Regeneron.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @mitchelzoler
PARIS – Treatment with the interleukin-6–blocking drug sarilumab led to statistically significant and clinically meaningful improvements in patients with rheumatoid arthritis in a multicenter, placebo controlled, phase III trial with 1,197 patients.
The safety and efficacy results from the SARIL-RA-MOBILITY trial are the first outcomes from a panel of phase III studies of sarilumab in patients with rheumatoid arthritis, said Dr. Mark Genovese at the annual European Congress of Rheumatology. The results showed that treatment with sarilumab plus methotrexate led to less joint damage over time, compared with methotrexate plus placebo, a benefit that should result in patients feeling better, said Dr. Genovese, professor of medicine and co-chief of the division of immunology and rheumatology at Stanford (Calif.) University.
Sarilumab is the first agent to progress this far in testing from a novel class of immunosuppressive drugs that work by blocking interleukin-6. Having safe drugs from a new class available to treat patients with rheumatoid arthritis holds promise for better controlling this disease in patients who inadequately respond to existing drug options, Dr. Genovese said in a video interview.
The SARIL-RA-MOBILITY trial was sponsored by Sanofi and Regeneron, the companies developing the drug. Dr. Genovese said that he has been a consultant to and received research support from Sanofi. Several of the coauthors are employees of Sanofi or Regeneron.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @mitchelzoler
PARIS – Treatment with the interleukin-6–blocking drug sarilumab led to statistically significant and clinically meaningful improvements in patients with rheumatoid arthritis in a multicenter, placebo controlled, phase III trial with 1,197 patients.
The safety and efficacy results from the SARIL-RA-MOBILITY trial are the first outcomes from a panel of phase III studies of sarilumab in patients with rheumatoid arthritis, said Dr. Mark Genovese at the annual European Congress of Rheumatology. The results showed that treatment with sarilumab plus methotrexate led to less joint damage over time, compared with methotrexate plus placebo, a benefit that should result in patients feeling better, said Dr. Genovese, professor of medicine and co-chief of the division of immunology and rheumatology at Stanford (Calif.) University.
Sarilumab is the first agent to progress this far in testing from a novel class of immunosuppressive drugs that work by blocking interleukin-6. Having safe drugs from a new class available to treat patients with rheumatoid arthritis holds promise for better controlling this disease in patients who inadequately respond to existing drug options, Dr. Genovese said in a video interview.
The SARIL-RA-MOBILITY trial was sponsored by Sanofi and Regeneron, the companies developing the drug. Dr. Genovese said that he has been a consultant to and received research support from Sanofi. Several of the coauthors are employees of Sanofi or Regeneron.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @mitchelzoler
AT THE EULAR CONGRESS 2014
Higher risk of death seen with oral steroids in RA interstitial lung disease
The use of prednisone for 3 or more months at a time was associated with a significantly elevated risk of death in patients with rheumatoid arthritis and interstitial lung disease in a retrospective cohort study.
Interstitial lung disease is present in about 5% of patients with rheumatoid arthritis. For years, oral steroids were commonly used in patients with the disease, but today’s rheumatologists "no longer view oral steroids as optimal treatment in RA-ILD [rheumatoid arthritis–associated interstitial lung disease], and our data now confirm that," said Dr. Clive Kelly of Queen Elizabeth Hospital in Gateshead, England, senior investigator of the study. He added that clinicians should avoid long-term treatment with steroids in this patient group whenever possible.
Dr. Kelly led the British Rheumatoid Interstitial Lung (BRILL) Network’s cohort study of 260 patients with RA-ILD diagnosed over a 25-year period. The BRILL study compared patients with RA-ILD and an equal number of RA controls without lung involvement who were matched for age, sex, and time of diagnosis.
At the annual European Congress of Rheumatology, Dr. Kelly reported that steroid-treated RA-ILD patients, who represented nearly 60% of the cohort, had an elevated relative risk of all-cause death, compared with those who had never been treated with steroids (RR, 1.65; 95% confidence interval, 1.2-2.3; P = .002). Although the relative risk of respiratory death was significantly increased for RA-ILD patients, regardless of treatment, when compared with RA patients without ILD, the risk was higher in those who had been on steroids (RR, 2.75; 95% CI, 1.6-4.7; P = .0002) than in those who had not received steroids (RR, 2.06; 95% CI, 1.1-3.8; P = .02), the investigators found.
The comparison also revealed other important findings related to RA-ILD. Patients with RA-ILD had significantly higher mortality than did those with RA alone. Over the course of the 25-year study period, however, mortality progressively improved among the RA-ILD patients, with median age at death rising from 63 to 76 years. This steady improvement, Dr. Kelly said, is partly the result of better and earlier diagnosis of lung involvement.
"It’s one of my many missions in life to get rheumatologists to listen to the lungs when they examine the joints in patients with rheumatoid arthritis," he said. "I think we are getting better. We’ve persuaded the British Society for Rheumatology to incorporate lung function testing and clinical examination of the chest into their basic assessment of a rheumatoid patient."
Also likely affecting the improved mortality seen over the cohort’s study period is a change in therapeutic approach. While RA-ILD patients diagnosed in the first half of the study period were likely to have been treated with only prednisone and azathioprine, in the latter half they were more likely to have received cyclophosphamide and methylprednisolone or mycophenolate. Over the last 12 years, more were treated with biologics, and in the final 6 years of the cohort, patients requiring biologics tended to be treated with rituximab, a B-cell inhibitor, rather than anti–tumor necrosis factor (anti-TNF) agents, the BRILL investigators found.
Mortality was lower among RA-ILD patients treated with mycophenolate than in those treated with other immunosuppressive agents. Among biologic agents used in the cohort, rituximab treatment was associated with improved mortality, but anti-TNF inhibitors were seen to be associated with elevated risk of death.
About 95% of RA-ILD patients are anticyclic citrullinated peptide (anti-CCP) antibody positive, compared with 55%-60% of the RA population as a whole, Dr. Kelly said, "so there’s a strong statistical association of seropositivity, and in those who are seropositive, rituximab works well."
The finding that rituximab was associated with improved survival in the cohort not only has implications for RA-ILD, he said, but also, potentially, for people with idiopathic pulmonary fibrosis (IPF) who are anti-CCP antibody positive. "What [rheumatologists] have, and chest physicians traditionally don’t, is access to rituximab and mycophenolate. But these might be worth trying in IPF as well," he said.
Dr. Kelly noted that prospective trials in RA-ILD are beginning to enroll patients with progressive disease to compare azathioprine and mycophenolate, allowing for the use of oral steroids, as well as patients with active RA and ILD to compare anti-TNF inhibitors against rituximab, also allowing oral steroids.
Dr. Kelly reported that he had no conflicts of interest related to his findings and that none of his fellow BRILL investigators had conflicts.
The use of prednisone for 3 or more months at a time was associated with a significantly elevated risk of death in patients with rheumatoid arthritis and interstitial lung disease in a retrospective cohort study.
Interstitial lung disease is present in about 5% of patients with rheumatoid arthritis. For years, oral steroids were commonly used in patients with the disease, but today’s rheumatologists "no longer view oral steroids as optimal treatment in RA-ILD [rheumatoid arthritis–associated interstitial lung disease], and our data now confirm that," said Dr. Clive Kelly of Queen Elizabeth Hospital in Gateshead, England, senior investigator of the study. He added that clinicians should avoid long-term treatment with steroids in this patient group whenever possible.
Dr. Kelly led the British Rheumatoid Interstitial Lung (BRILL) Network’s cohort study of 260 patients with RA-ILD diagnosed over a 25-year period. The BRILL study compared patients with RA-ILD and an equal number of RA controls without lung involvement who were matched for age, sex, and time of diagnosis.
At the annual European Congress of Rheumatology, Dr. Kelly reported that steroid-treated RA-ILD patients, who represented nearly 60% of the cohort, had an elevated relative risk of all-cause death, compared with those who had never been treated with steroids (RR, 1.65; 95% confidence interval, 1.2-2.3; P = .002). Although the relative risk of respiratory death was significantly increased for RA-ILD patients, regardless of treatment, when compared with RA patients without ILD, the risk was higher in those who had been on steroids (RR, 2.75; 95% CI, 1.6-4.7; P = .0002) than in those who had not received steroids (RR, 2.06; 95% CI, 1.1-3.8; P = .02), the investigators found.
The comparison also revealed other important findings related to RA-ILD. Patients with RA-ILD had significantly higher mortality than did those with RA alone. Over the course of the 25-year study period, however, mortality progressively improved among the RA-ILD patients, with median age at death rising from 63 to 76 years. This steady improvement, Dr. Kelly said, is partly the result of better and earlier diagnosis of lung involvement.
"It’s one of my many missions in life to get rheumatologists to listen to the lungs when they examine the joints in patients with rheumatoid arthritis," he said. "I think we are getting better. We’ve persuaded the British Society for Rheumatology to incorporate lung function testing and clinical examination of the chest into their basic assessment of a rheumatoid patient."
Also likely affecting the improved mortality seen over the cohort’s study period is a change in therapeutic approach. While RA-ILD patients diagnosed in the first half of the study period were likely to have been treated with only prednisone and azathioprine, in the latter half they were more likely to have received cyclophosphamide and methylprednisolone or mycophenolate. Over the last 12 years, more were treated with biologics, and in the final 6 years of the cohort, patients requiring biologics tended to be treated with rituximab, a B-cell inhibitor, rather than anti–tumor necrosis factor (anti-TNF) agents, the BRILL investigators found.
Mortality was lower among RA-ILD patients treated with mycophenolate than in those treated with other immunosuppressive agents. Among biologic agents used in the cohort, rituximab treatment was associated with improved mortality, but anti-TNF inhibitors were seen to be associated with elevated risk of death.
About 95% of RA-ILD patients are anticyclic citrullinated peptide (anti-CCP) antibody positive, compared with 55%-60% of the RA population as a whole, Dr. Kelly said, "so there’s a strong statistical association of seropositivity, and in those who are seropositive, rituximab works well."
The finding that rituximab was associated with improved survival in the cohort not only has implications for RA-ILD, he said, but also, potentially, for people with idiopathic pulmonary fibrosis (IPF) who are anti-CCP antibody positive. "What [rheumatologists] have, and chest physicians traditionally don’t, is access to rituximab and mycophenolate. But these might be worth trying in IPF as well," he said.
Dr. Kelly noted that prospective trials in RA-ILD are beginning to enroll patients with progressive disease to compare azathioprine and mycophenolate, allowing for the use of oral steroids, as well as patients with active RA and ILD to compare anti-TNF inhibitors against rituximab, also allowing oral steroids.
Dr. Kelly reported that he had no conflicts of interest related to his findings and that none of his fellow BRILL investigators had conflicts.
The use of prednisone for 3 or more months at a time was associated with a significantly elevated risk of death in patients with rheumatoid arthritis and interstitial lung disease in a retrospective cohort study.
Interstitial lung disease is present in about 5% of patients with rheumatoid arthritis. For years, oral steroids were commonly used in patients with the disease, but today’s rheumatologists "no longer view oral steroids as optimal treatment in RA-ILD [rheumatoid arthritis–associated interstitial lung disease], and our data now confirm that," said Dr. Clive Kelly of Queen Elizabeth Hospital in Gateshead, England, senior investigator of the study. He added that clinicians should avoid long-term treatment with steroids in this patient group whenever possible.
Dr. Kelly led the British Rheumatoid Interstitial Lung (BRILL) Network’s cohort study of 260 patients with RA-ILD diagnosed over a 25-year period. The BRILL study compared patients with RA-ILD and an equal number of RA controls without lung involvement who were matched for age, sex, and time of diagnosis.
At the annual European Congress of Rheumatology, Dr. Kelly reported that steroid-treated RA-ILD patients, who represented nearly 60% of the cohort, had an elevated relative risk of all-cause death, compared with those who had never been treated with steroids (RR, 1.65; 95% confidence interval, 1.2-2.3; P = .002). Although the relative risk of respiratory death was significantly increased for RA-ILD patients, regardless of treatment, when compared with RA patients without ILD, the risk was higher in those who had been on steroids (RR, 2.75; 95% CI, 1.6-4.7; P = .0002) than in those who had not received steroids (RR, 2.06; 95% CI, 1.1-3.8; P = .02), the investigators found.
The comparison also revealed other important findings related to RA-ILD. Patients with RA-ILD had significantly higher mortality than did those with RA alone. Over the course of the 25-year study period, however, mortality progressively improved among the RA-ILD patients, with median age at death rising from 63 to 76 years. This steady improvement, Dr. Kelly said, is partly the result of better and earlier diagnosis of lung involvement.
"It’s one of my many missions in life to get rheumatologists to listen to the lungs when they examine the joints in patients with rheumatoid arthritis," he said. "I think we are getting better. We’ve persuaded the British Society for Rheumatology to incorporate lung function testing and clinical examination of the chest into their basic assessment of a rheumatoid patient."
Also likely affecting the improved mortality seen over the cohort’s study period is a change in therapeutic approach. While RA-ILD patients diagnosed in the first half of the study period were likely to have been treated with only prednisone and azathioprine, in the latter half they were more likely to have received cyclophosphamide and methylprednisolone or mycophenolate. Over the last 12 years, more were treated with biologics, and in the final 6 years of the cohort, patients requiring biologics tended to be treated with rituximab, a B-cell inhibitor, rather than anti–tumor necrosis factor (anti-TNF) agents, the BRILL investigators found.
Mortality was lower among RA-ILD patients treated with mycophenolate than in those treated with other immunosuppressive agents. Among biologic agents used in the cohort, rituximab treatment was associated with improved mortality, but anti-TNF inhibitors were seen to be associated with elevated risk of death.
About 95% of RA-ILD patients are anticyclic citrullinated peptide (anti-CCP) antibody positive, compared with 55%-60% of the RA population as a whole, Dr. Kelly said, "so there’s a strong statistical association of seropositivity, and in those who are seropositive, rituximab works well."
The finding that rituximab was associated with improved survival in the cohort not only has implications for RA-ILD, he said, but also, potentially, for people with idiopathic pulmonary fibrosis (IPF) who are anti-CCP antibody positive. "What [rheumatologists] have, and chest physicians traditionally don’t, is access to rituximab and mycophenolate. But these might be worth trying in IPF as well," he said.
Dr. Kelly noted that prospective trials in RA-ILD are beginning to enroll patients with progressive disease to compare azathioprine and mycophenolate, allowing for the use of oral steroids, as well as patients with active RA and ILD to compare anti-TNF inhibitors against rituximab, also allowing oral steroids.
Dr. Kelly reported that he had no conflicts of interest related to his findings and that none of his fellow BRILL investigators had conflicts.
FROM THE EULAR CONGRESS 2014
Key clinical point: Rather than corticosteroids, consider using mycophenolate or rituximab in patients with RA-ILD.
Major finding: Steroid-treated RA-ILD patients had an elevated relative risk of all-cause death, compared with those who had never been treated with steroids (RR, 1.65; 95% confidence interval, 1.2-2.3; P = .002).
Data source: A retrospective study of 260 patients with RA-ILD in the British Rheumatoid Interstitial Lung Network.
Disclosures: Dr. Kelly reported that he had no conflicts of interest related to his findings and that none of his fellow BRILL investigators had conflicts.