User login
Pediatric dermatology update: New research offers insight into psoriasis, alopecia
LAS VEGAS – Recent research is offering new insights into psoriasis and alopecia in the pediatric population, a dermatologist told colleagues, and it’s time to be on the lookout for psoriasis linked to treatment with tumor necrosis factor (TNF) inhibitors.
Lawrence F. Eichenfield, MD, professor of dermatology and pediatrics, at the University of California, San Diego, offered these tips and comments in a presentation at Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar:
Psoriasis
It’s a brand new day for adult psoriasis sufferers, but it seems to be only a brand new morning for their pediatric counterparts. “Kids and teenagers were left behind in the biologic revolution,” Dr. Eichenfield said. “Only two systemic biologics have been approved for psoriasis in children.” They are ustekinumab (Stelara), approved by the Food and Drug Administration for treating psoriasis in children aged 12 years and older, and etanercept, approved for aged 4 years and older.
The good news, he said, is that “our new biologic agents are now being studied in children.”
Research is also providing new insight into pediatric psoriasis, said Dr. Eichenfield, who is also chief of pediatric and adolescent dermatology at Rady Children’s Hospital in San Diego. It’s now clear that “there’s a lot more facial involvement, and a high involvement of scalp and nail,” he noted.
It’s also clear, he said, that inflammation begins early in pediatric psoriasis. That raises the question of whether it’s a good idea to launch aggressive treatment to stop the “psoriatic march” toward cardiovascular and other medical problems down the line, he commented.
“Keep an open mind to getting aggressive in therapy,” he advised, although he acknowledged that “it’s hard to get beyond the two biologics, and only one is approved for children under 12.”
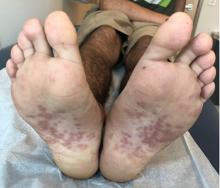
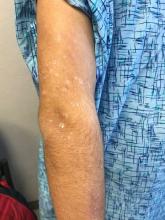
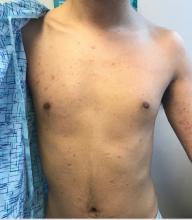
Dr. Eichenfield advised colleagues to keep an eye out for TNF inhibitor–induced psoriasis. “We’re seeing it pretty regularly,” he said, commonly in children who are treated with TNF inhibitors for rheumatoid arthritis or Crohn’s disease.
The lesions “look like dermatitis but are very psoriasiform,” he said, and research suggests this can appear after a single dose or after as many as 63 months of treatment. Topical and light therapy can be helpful. But if those treatments do not help, he said, it’s time to consider changing the biologic that the patient is taking. “Is the biologic adequately controlling their underlying disease? If not, you can help find one that would be great for their underlying disease and clear up their psoriasis.”
Alopecia
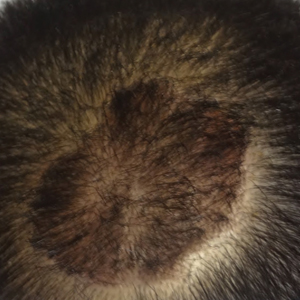
Pediatric alopecia “is a problem I see pretty regularly in practice,” Dr. Eichenfield said. When he sees patients with alopecia, he says that, “‘if your child doesn’t have 50% hair loss, you’re in the good group. It will generally heal up and never come back again.’ ”
He referred to a recent study, where investigators at the Children’s Hospital of Philadelphia retrospectively studied 125 children under age 4 years who were diagnosed with alopecia areata and followed for 2 years. Over time, those children with over 50% of hair loss initially were more likely to have worsening Severity of Alopecia Tool (SALT) scores over the follow-up period. But a high proportion of those with mild alopecia initially continued to have mild alopecia at follow-up (Pediatr Dermatol. 2019 Aug 29. doi: 10.1111/pde.13990).
Dr. Eichenfield noted that the study found that 41% of the patients also had atopic dermatitis.
He also highlighted two other recent studies on pediatric alopecia: One found that while vitamin D levels were low in a majority of children with alopecia in the study, the proportion who had a deficiency was similar to the proportion in a larger pediatric population, at about 22% in both groups (J Am Acad Dermatol. 2018 Sep;79(3):e43-e44). Supplementation doesn’t seem to help. “It’s not important to test levels,” he said.
Another study examined whether it’s a good idea to test patients for celiac disease in children with alopecia (Pediatr Dermatol. 2018 Jul;35[4]:535-8). Some parents may ask this question, but the answer, he said, is generally no.
What’s next? “We were hoping oral and topical JAK inhibitors would work well” in this population, Dr. Eichenfield said, but study findings haven’t been promising.
Still, oral tofacitinib (Xeljanz) showed some “pretty impressive” success in a recent study in four children, he noted. Based on the results, the authors wrote that “we suggest that, after proper counseling regarding the risks, including severe infection and malignancy, the use of tofacitinib may be considered for preadolescent children with AA [alopecia areata] who are experiencing psychosocial impairment” (J Am Acad Dermatol. 2019 Feb;80[2]:568-70).
In general, Dr. Eichenfield said, research on pediatric alopecia “will be secondary, especially with JAK inhibitors because of the risk of side effects. But [children will] probably tolerate them better than adults do because they have fewer medical problems.”
Meanwhile, he added, controversy continues to swirl around how to treat children over age 10 years who have lost 50% or more of their hair. “I’ve seen hundreds of kids with alopecia areata,” he said, “and I can’t predict what the course may be.”
Dr. Eichenfield reports multiple relationships (consultant or investigator) with various pharmaceutical companies, including Abbvie, Allergan, Lilly, Novartis, and others. SDEF and this news organization are owned by the same parent company.
LAS VEGAS – Recent research is offering new insights into psoriasis and alopecia in the pediatric population, a dermatologist told colleagues, and it’s time to be on the lookout for psoriasis linked to treatment with tumor necrosis factor (TNF) inhibitors.
Lawrence F. Eichenfield, MD, professor of dermatology and pediatrics, at the University of California, San Diego, offered these tips and comments in a presentation at Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar:
Psoriasis
It’s a brand new day for adult psoriasis sufferers, but it seems to be only a brand new morning for their pediatric counterparts. “Kids and teenagers were left behind in the biologic revolution,” Dr. Eichenfield said. “Only two systemic biologics have been approved for psoriasis in children.” They are ustekinumab (Stelara), approved by the Food and Drug Administration for treating psoriasis in children aged 12 years and older, and etanercept, approved for aged 4 years and older.
The good news, he said, is that “our new biologic agents are now being studied in children.”
Research is also providing new insight into pediatric psoriasis, said Dr. Eichenfield, who is also chief of pediatric and adolescent dermatology at Rady Children’s Hospital in San Diego. It’s now clear that “there’s a lot more facial involvement, and a high involvement of scalp and nail,” he noted.
It’s also clear, he said, that inflammation begins early in pediatric psoriasis. That raises the question of whether it’s a good idea to launch aggressive treatment to stop the “psoriatic march” toward cardiovascular and other medical problems down the line, he commented.
“Keep an open mind to getting aggressive in therapy,” he advised, although he acknowledged that “it’s hard to get beyond the two biologics, and only one is approved for children under 12.”
Dr. Eichenfield advised colleagues to keep an eye out for TNF inhibitor–induced psoriasis. “We’re seeing it pretty regularly,” he said, commonly in children who are treated with TNF inhibitors for rheumatoid arthritis or Crohn’s disease.
The lesions “look like dermatitis but are very psoriasiform,” he said, and research suggests this can appear after a single dose or after as many as 63 months of treatment. Topical and light therapy can be helpful. But if those treatments do not help, he said, it’s time to consider changing the biologic that the patient is taking. “Is the biologic adequately controlling their underlying disease? If not, you can help find one that would be great for their underlying disease and clear up their psoriasis.”
Alopecia
Pediatric alopecia “is a problem I see pretty regularly in practice,” Dr. Eichenfield said. When he sees patients with alopecia, he says that, “‘if your child doesn’t have 50% hair loss, you’re in the good group. It will generally heal up and never come back again.’ ”
He referred to a recent study, where investigators at the Children’s Hospital of Philadelphia retrospectively studied 125 children under age 4 years who were diagnosed with alopecia areata and followed for 2 years. Over time, those children with over 50% of hair loss initially were more likely to have worsening Severity of Alopecia Tool (SALT) scores over the follow-up period. But a high proportion of those with mild alopecia initially continued to have mild alopecia at follow-up (Pediatr Dermatol. 2019 Aug 29. doi: 10.1111/pde.13990).
Dr. Eichenfield noted that the study found that 41% of the patients also had atopic dermatitis.
He also highlighted two other recent studies on pediatric alopecia: One found that while vitamin D levels were low in a majority of children with alopecia in the study, the proportion who had a deficiency was similar to the proportion in a larger pediatric population, at about 22% in both groups (J Am Acad Dermatol. 2018 Sep;79(3):e43-e44). Supplementation doesn’t seem to help. “It’s not important to test levels,” he said.
Another study examined whether it’s a good idea to test patients for celiac disease in children with alopecia (Pediatr Dermatol. 2018 Jul;35[4]:535-8). Some parents may ask this question, but the answer, he said, is generally no.
What’s next? “We were hoping oral and topical JAK inhibitors would work well” in this population, Dr. Eichenfield said, but study findings haven’t been promising.
Still, oral tofacitinib (Xeljanz) showed some “pretty impressive” success in a recent study in four children, he noted. Based on the results, the authors wrote that “we suggest that, after proper counseling regarding the risks, including severe infection and malignancy, the use of tofacitinib may be considered for preadolescent children with AA [alopecia areata] who are experiencing psychosocial impairment” (J Am Acad Dermatol. 2019 Feb;80[2]:568-70).
In general, Dr. Eichenfield said, research on pediatric alopecia “will be secondary, especially with JAK inhibitors because of the risk of side effects. But [children will] probably tolerate them better than adults do because they have fewer medical problems.”
Meanwhile, he added, controversy continues to swirl around how to treat children over age 10 years who have lost 50% or more of their hair. “I’ve seen hundreds of kids with alopecia areata,” he said, “and I can’t predict what the course may be.”
Dr. Eichenfield reports multiple relationships (consultant or investigator) with various pharmaceutical companies, including Abbvie, Allergan, Lilly, Novartis, and others. SDEF and this news organization are owned by the same parent company.
LAS VEGAS – Recent research is offering new insights into psoriasis and alopecia in the pediatric population, a dermatologist told colleagues, and it’s time to be on the lookout for psoriasis linked to treatment with tumor necrosis factor (TNF) inhibitors.
Lawrence F. Eichenfield, MD, professor of dermatology and pediatrics, at the University of California, San Diego, offered these tips and comments in a presentation at Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar:
Psoriasis
It’s a brand new day for adult psoriasis sufferers, but it seems to be only a brand new morning for their pediatric counterparts. “Kids and teenagers were left behind in the biologic revolution,” Dr. Eichenfield said. “Only two systemic biologics have been approved for psoriasis in children.” They are ustekinumab (Stelara), approved by the Food and Drug Administration for treating psoriasis in children aged 12 years and older, and etanercept, approved for aged 4 years and older.
The good news, he said, is that “our new biologic agents are now being studied in children.”
Research is also providing new insight into pediatric psoriasis, said Dr. Eichenfield, who is also chief of pediatric and adolescent dermatology at Rady Children’s Hospital in San Diego. It’s now clear that “there’s a lot more facial involvement, and a high involvement of scalp and nail,” he noted.
It’s also clear, he said, that inflammation begins early in pediatric psoriasis. That raises the question of whether it’s a good idea to launch aggressive treatment to stop the “psoriatic march” toward cardiovascular and other medical problems down the line, he commented.
“Keep an open mind to getting aggressive in therapy,” he advised, although he acknowledged that “it’s hard to get beyond the two biologics, and only one is approved for children under 12.”
Dr. Eichenfield advised colleagues to keep an eye out for TNF inhibitor–induced psoriasis. “We’re seeing it pretty regularly,” he said, commonly in children who are treated with TNF inhibitors for rheumatoid arthritis or Crohn’s disease.
The lesions “look like dermatitis but are very psoriasiform,” he said, and research suggests this can appear after a single dose or after as many as 63 months of treatment. Topical and light therapy can be helpful. But if those treatments do not help, he said, it’s time to consider changing the biologic that the patient is taking. “Is the biologic adequately controlling their underlying disease? If not, you can help find one that would be great for their underlying disease and clear up their psoriasis.”
Alopecia
Pediatric alopecia “is a problem I see pretty regularly in practice,” Dr. Eichenfield said. When he sees patients with alopecia, he says that, “‘if your child doesn’t have 50% hair loss, you’re in the good group. It will generally heal up and never come back again.’ ”
He referred to a recent study, where investigators at the Children’s Hospital of Philadelphia retrospectively studied 125 children under age 4 years who were diagnosed with alopecia areata and followed for 2 years. Over time, those children with over 50% of hair loss initially were more likely to have worsening Severity of Alopecia Tool (SALT) scores over the follow-up period. But a high proportion of those with mild alopecia initially continued to have mild alopecia at follow-up (Pediatr Dermatol. 2019 Aug 29. doi: 10.1111/pde.13990).
Dr. Eichenfield noted that the study found that 41% of the patients also had atopic dermatitis.
He also highlighted two other recent studies on pediatric alopecia: One found that while vitamin D levels were low in a majority of children with alopecia in the study, the proportion who had a deficiency was similar to the proportion in a larger pediatric population, at about 22% in both groups (J Am Acad Dermatol. 2018 Sep;79(3):e43-e44). Supplementation doesn’t seem to help. “It’s not important to test levels,” he said.
Another study examined whether it’s a good idea to test patients for celiac disease in children with alopecia (Pediatr Dermatol. 2018 Jul;35[4]:535-8). Some parents may ask this question, but the answer, he said, is generally no.
What’s next? “We were hoping oral and topical JAK inhibitors would work well” in this population, Dr. Eichenfield said, but study findings haven’t been promising.
Still, oral tofacitinib (Xeljanz) showed some “pretty impressive” success in a recent study in four children, he noted. Based on the results, the authors wrote that “we suggest that, after proper counseling regarding the risks, including severe infection and malignancy, the use of tofacitinib may be considered for preadolescent children with AA [alopecia areata] who are experiencing psychosocial impairment” (J Am Acad Dermatol. 2019 Feb;80[2]:568-70).
In general, Dr. Eichenfield said, research on pediatric alopecia “will be secondary, especially with JAK inhibitors because of the risk of side effects. But [children will] probably tolerate them better than adults do because they have fewer medical problems.”
Meanwhile, he added, controversy continues to swirl around how to treat children over age 10 years who have lost 50% or more of their hair. “I’ve seen hundreds of kids with alopecia areata,” he said, “and I can’t predict what the course may be.”
Dr. Eichenfield reports multiple relationships (consultant or investigator) with various pharmaceutical companies, including Abbvie, Allergan, Lilly, Novartis, and others. SDEF and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF LAS VEGAS DERMATOLOGY SEMINAR
In psoriasis, methotrexate and other older drugs can still be useful
LAS VEGAS – While biologics have dramatically changed the picture, drugs like , a dermatologist told colleagues.
However, caution is necessary, especially when the drugs are used in combination with biologics, Bruce E. Strober, MD, PhD, of Yale University, New Haven, Conn., and Central Connecticut Dermatology, Cromwell, Conn., said at Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar.
Dr. Strober offered these tips about the proper use of these four drugs in psoriasis patients:
Acitretin (Soriatane). “This was used as monotherapy initially, but at this point in history, fewer and fewer patients are getting it as monotherapy,” he said. A dose of 25 mg/day appears to provide the best mix of efficacy and side-effect control, “although it’s not a high-efficacy drug, especially at 25 mg a day. It’s a slow-acting drug, and you may need 4 if not 6 months to see the maximum effect before you give up on it.”
What about using acitretin in combination with other therapies? Studies examining its use with phototherapy haven’t been promising, Dr. Strober said. The drug can be used with methotrexate, he said, even though the combination will worry pharmacists. “Follow the liver, and you’ll be fine” he noted. “That combination can be successful. Laboratory monitoring is not onerous: Discontinue after a few months if you’ve not seen any movement.” The drug can also be used with biologics, he said.
Apremilast (Otezla). This drug will bring about a third of patients to a Psoriasis Area and Severity Index (PASI) 75. “That’s not the most impressive efficacy. Rarely do we clear patients with this drug, and it has tolerability issues in some patients,” Dr. Strober said. Side effects can include diarrhea, nausea, headache, and depression. “Warn patients of these possibilities,” he added.
Methotrexate. “It’s very helpful and not a drug to be feared if it’s monitored correctly,” Dr. Strober said. “It’s certainly not a biologic, but it’s not a bad drug from an efficacy standpoint, and it does have efficacy in psoriatic arthritis.”
The drug’s low cost can make it a good alternative to biologics in patients with limited insurance options – such as those on Medicare – or those who don’t have insurance, he said.
“Psoriasis is often controlled at a mean dose of 15 mg/week [orally], with no test dose; start at 15-mg weekly,” he said. “It’s an interesting drug that allows you to dose weekly and still get efficacy,” especially when dosed subcutaneously.
Beware the many contraindications such as pregnancy, possible pregnancy, and high alcohol intake, he added. Dr. Strober doesn’t recommend liver biopsies to monitor hepatic effects. “It’s a poor test with risk and sampling error,” he said.
Cyclosporine. This drug is best “in severe patients in need of a quick response,” said Dr. Strober, who added that biologics are often a better option even in patients who are sensitive to price since samples and free-drug programs are available. “It’s in and out of the body quickly, and most people skip doses and get recurrence of their disease quickly,” he said.
Blood tests are a hassle for patients, he said, and “people often don’t feel great on the drug,” said Dr. Strober, who added, however, that he still does occasionally use it.
Dr. Strober reported multiple disclosures including consultant/advisory board (AbbVie, Amgen, Lilly, Pfizer, among others) and investigator relationships. SDEF and this news organization are owned by the same parent company.
LAS VEGAS – While biologics have dramatically changed the picture, drugs like , a dermatologist told colleagues.
However, caution is necessary, especially when the drugs are used in combination with biologics, Bruce E. Strober, MD, PhD, of Yale University, New Haven, Conn., and Central Connecticut Dermatology, Cromwell, Conn., said at Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar.
Dr. Strober offered these tips about the proper use of these four drugs in psoriasis patients:
Acitretin (Soriatane). “This was used as monotherapy initially, but at this point in history, fewer and fewer patients are getting it as monotherapy,” he said. A dose of 25 mg/day appears to provide the best mix of efficacy and side-effect control, “although it’s not a high-efficacy drug, especially at 25 mg a day. It’s a slow-acting drug, and you may need 4 if not 6 months to see the maximum effect before you give up on it.”
What about using acitretin in combination with other therapies? Studies examining its use with phototherapy haven’t been promising, Dr. Strober said. The drug can be used with methotrexate, he said, even though the combination will worry pharmacists. “Follow the liver, and you’ll be fine” he noted. “That combination can be successful. Laboratory monitoring is not onerous: Discontinue after a few months if you’ve not seen any movement.” The drug can also be used with biologics, he said.
Apremilast (Otezla). This drug will bring about a third of patients to a Psoriasis Area and Severity Index (PASI) 75. “That’s not the most impressive efficacy. Rarely do we clear patients with this drug, and it has tolerability issues in some patients,” Dr. Strober said. Side effects can include diarrhea, nausea, headache, and depression. “Warn patients of these possibilities,” he added.
Methotrexate. “It’s very helpful and not a drug to be feared if it’s monitored correctly,” Dr. Strober said. “It’s certainly not a biologic, but it’s not a bad drug from an efficacy standpoint, and it does have efficacy in psoriatic arthritis.”
The drug’s low cost can make it a good alternative to biologics in patients with limited insurance options – such as those on Medicare – or those who don’t have insurance, he said.
“Psoriasis is often controlled at a mean dose of 15 mg/week [orally], with no test dose; start at 15-mg weekly,” he said. “It’s an interesting drug that allows you to dose weekly and still get efficacy,” especially when dosed subcutaneously.
Beware the many contraindications such as pregnancy, possible pregnancy, and high alcohol intake, he added. Dr. Strober doesn’t recommend liver biopsies to monitor hepatic effects. “It’s a poor test with risk and sampling error,” he said.
Cyclosporine. This drug is best “in severe patients in need of a quick response,” said Dr. Strober, who added that biologics are often a better option even in patients who are sensitive to price since samples and free-drug programs are available. “It’s in and out of the body quickly, and most people skip doses and get recurrence of their disease quickly,” he said.
Blood tests are a hassle for patients, he said, and “people often don’t feel great on the drug,” said Dr. Strober, who added, however, that he still does occasionally use it.
Dr. Strober reported multiple disclosures including consultant/advisory board (AbbVie, Amgen, Lilly, Pfizer, among others) and investigator relationships. SDEF and this news organization are owned by the same parent company.
LAS VEGAS – While biologics have dramatically changed the picture, drugs like , a dermatologist told colleagues.
However, caution is necessary, especially when the drugs are used in combination with biologics, Bruce E. Strober, MD, PhD, of Yale University, New Haven, Conn., and Central Connecticut Dermatology, Cromwell, Conn., said at Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar.
Dr. Strober offered these tips about the proper use of these four drugs in psoriasis patients:
Acitretin (Soriatane). “This was used as monotherapy initially, but at this point in history, fewer and fewer patients are getting it as monotherapy,” he said. A dose of 25 mg/day appears to provide the best mix of efficacy and side-effect control, “although it’s not a high-efficacy drug, especially at 25 mg a day. It’s a slow-acting drug, and you may need 4 if not 6 months to see the maximum effect before you give up on it.”
What about using acitretin in combination with other therapies? Studies examining its use with phototherapy haven’t been promising, Dr. Strober said. The drug can be used with methotrexate, he said, even though the combination will worry pharmacists. “Follow the liver, and you’ll be fine” he noted. “That combination can be successful. Laboratory monitoring is not onerous: Discontinue after a few months if you’ve not seen any movement.” The drug can also be used with biologics, he said.
Apremilast (Otezla). This drug will bring about a third of patients to a Psoriasis Area and Severity Index (PASI) 75. “That’s not the most impressive efficacy. Rarely do we clear patients with this drug, and it has tolerability issues in some patients,” Dr. Strober said. Side effects can include diarrhea, nausea, headache, and depression. “Warn patients of these possibilities,” he added.
Methotrexate. “It’s very helpful and not a drug to be feared if it’s monitored correctly,” Dr. Strober said. “It’s certainly not a biologic, but it’s not a bad drug from an efficacy standpoint, and it does have efficacy in psoriatic arthritis.”
The drug’s low cost can make it a good alternative to biologics in patients with limited insurance options – such as those on Medicare – or those who don’t have insurance, he said.
“Psoriasis is often controlled at a mean dose of 15 mg/week [orally], with no test dose; start at 15-mg weekly,” he said. “It’s an interesting drug that allows you to dose weekly and still get efficacy,” especially when dosed subcutaneously.
Beware the many contraindications such as pregnancy, possible pregnancy, and high alcohol intake, he added. Dr. Strober doesn’t recommend liver biopsies to monitor hepatic effects. “It’s a poor test with risk and sampling error,” he said.
Cyclosporine. This drug is best “in severe patients in need of a quick response,” said Dr. Strober, who added that biologics are often a better option even in patients who are sensitive to price since samples and free-drug programs are available. “It’s in and out of the body quickly, and most people skip doses and get recurrence of their disease quickly,” he said.
Blood tests are a hassle for patients, he said, and “people often don’t feel great on the drug,” said Dr. Strober, who added, however, that he still does occasionally use it.
Dr. Strober reported multiple disclosures including consultant/advisory board (AbbVie, Amgen, Lilly, Pfizer, among others) and investigator relationships. SDEF and this news organization are owned by the same parent company.
REPORTING FROM SDEF LAS VEGAS DERMATOLOGY SEMINAR
SPIRIT-H2H results confirm superiority of ixekizumab over adalimumab for PsA
ATLANTA – Ixekizumab (Taltz) provided significantly greater improvement in joint and skin symptoms, compared with adalimumab (Humira), in biologic-naive patients with active psoriatic arthritis (PsA), according to final 52-week safety and efficacy results from the randomized SPIRIT-H2H study.
The high-affinity monoclonal antibody against interleukin-17A also performed at least as well as the tumor necrosis factor (TNF)–inhibitor adalimumab across multiple PsA domains and regardless of methotrexate use, Josef Smolen, MD, reported during a late-breaking abstract session at the annual meeting of the American College of Rheumatology.
Multiple biologic disease-modifying antirheumatic drugs (bDMARDs) are available for the treatment of PsA, but few studies have directly compared their efficacy and safety, said Dr. Smolen of the Medical University of Vienna. He noted that the SPIRIT-H2H study aimed to compare ixekizumab and adalimumab and also to address “one of the most clinically relevant questions for clinicians,” which relates to the efficacy of bDMARDs with and without concomitant methotrexate.
Ixekizumab is approved for adults with active PsA and moderate to severe plaque psoriasis, but TNF inhibitors like adalimumab have long been considered the gold standard for PsA treatment, he explained.
Of 283 patients with PsA randomized to receive ixekizumab and 283 randomized to receive adalimumab, 87% and 84%, respectively, completed week 52 of the head-to-head, open-label study comparing the bDMARDs. Treatment with ixekizumab achieved the primary endpoint of simultaneous improvement of 50% on ACR response criteria (ACR50) and 100% on the Psoriasis Area and Severity Index (PASI100) in 39% of patients, which was significantly higher than the rate of 26% with adalimumab, Dr. Smolen said.
Ixekizumab also performed at least as well as adalimumab for the secondary outcome measures of ACR50 response (50% in both groups) and PASI100 response (64% vs. 41%), as well as for all other outcomes measures, including multiple musculoskeletal PsA domains, he said.
“Remarkably ... at 1 year, more than one-third of the patients achieved an ACR70 in both groups, and half of the patients achieved an ACR50,” he added, noting that the ACR100 responses were in line with previous investigations.
Stratification by methotrexate use showed that the simultaneous ACR50 and PASI100 response rates were improved with ixekizumab versus adalimumab both in users and nonusers of methotrexate (39% vs. 30% and 40% vs. 20%, respectively). This finding highlights the ongoing debate about whether TNF inhibitors should or should not be used with methotrexate for PsA.
“This study was not adequately powered to say that, but there is some indication, and I think that this is food for thought for future further analysis because the data in the literature are discrepant in this respect,” Dr. Smolen said.
In non-methotrexate users in SPIRIT-H2H, the ACR20 responses were 53% with ixekinumab vs. 40% with adalimumab, ACR50 responses were 72% vs. 60%, and ACR70 responses were 41% vs. 27%, respectively, he said noting that the difference for ACR70 was statistically significant, and that the ACR70 response with ixekinumab was about the same as the ACR50 for adalimumab.
As for ACR20, ACR50, and ACR70 responses in methotrexate users, “the lines criss-crossed” early on, he said, but all were “slightly superior” with adalimumab than with ixekizumab at 52 weeks (75% vs. 68%, 56% vs. 48%, and 39% vs. 32%, respectively).
Study participants had a mean age of 48 years and had active PsA with at least 3/66 tender joints, at least 3/68 swollen joints, at least 3% psoriasis body surface area involvement, no prior treatment with bDMARDs, and prior inadequate response to one or more conventional synthetic DMARDs. Treatment was dosed according to drug labeling through 52 weeks.
The safety profiles of both agents were consistent with previous reports; treatment-emergent adverse events occurred in 73.9% of ixekizumab and 68.6% of adalimumab patients, and serious adverse events occurred in 4.2% and 12.4%, respectively.
“On the other hand, ixekizumab had more injection site reactions: 11% vs. close to 4%,” he said, noting that 4.2% of the ixekizumab patients and 7.4% of the adalimumab patients discontinued treatment because of adverse events. No deaths occurred in either group.
As reported previously in Annals of the Rheumatic Diseases, ixekizumab was superior to adalimumab for simultaneous achievement of ACR50 and PASI100 at 24 weeks, and these final 52-week results confirm those results, he said.
The study was funded by Eli Lilly, which markets ixekizumab. Dr. Smolen reported research grants and/or honoraria from Eli Lilly and AbbVie, which markets adalimumab, as well as many other pharmaceutical companies.
SOURCE: Smolen J et al. Arthritis Rheumatol. 2019;71(suppl 10), Abstract L20.
ATLANTA – Ixekizumab (Taltz) provided significantly greater improvement in joint and skin symptoms, compared with adalimumab (Humira), in biologic-naive patients with active psoriatic arthritis (PsA), according to final 52-week safety and efficacy results from the randomized SPIRIT-H2H study.
The high-affinity monoclonal antibody against interleukin-17A also performed at least as well as the tumor necrosis factor (TNF)–inhibitor adalimumab across multiple PsA domains and regardless of methotrexate use, Josef Smolen, MD, reported during a late-breaking abstract session at the annual meeting of the American College of Rheumatology.
Multiple biologic disease-modifying antirheumatic drugs (bDMARDs) are available for the treatment of PsA, but few studies have directly compared their efficacy and safety, said Dr. Smolen of the Medical University of Vienna. He noted that the SPIRIT-H2H study aimed to compare ixekizumab and adalimumab and also to address “one of the most clinically relevant questions for clinicians,” which relates to the efficacy of bDMARDs with and without concomitant methotrexate.
Ixekizumab is approved for adults with active PsA and moderate to severe plaque psoriasis, but TNF inhibitors like adalimumab have long been considered the gold standard for PsA treatment, he explained.
Of 283 patients with PsA randomized to receive ixekizumab and 283 randomized to receive adalimumab, 87% and 84%, respectively, completed week 52 of the head-to-head, open-label study comparing the bDMARDs. Treatment with ixekizumab achieved the primary endpoint of simultaneous improvement of 50% on ACR response criteria (ACR50) and 100% on the Psoriasis Area and Severity Index (PASI100) in 39% of patients, which was significantly higher than the rate of 26% with adalimumab, Dr. Smolen said.
Ixekizumab also performed at least as well as adalimumab for the secondary outcome measures of ACR50 response (50% in both groups) and PASI100 response (64% vs. 41%), as well as for all other outcomes measures, including multiple musculoskeletal PsA domains, he said.
“Remarkably ... at 1 year, more than one-third of the patients achieved an ACR70 in both groups, and half of the patients achieved an ACR50,” he added, noting that the ACR100 responses were in line with previous investigations.
Stratification by methotrexate use showed that the simultaneous ACR50 and PASI100 response rates were improved with ixekizumab versus adalimumab both in users and nonusers of methotrexate (39% vs. 30% and 40% vs. 20%, respectively). This finding highlights the ongoing debate about whether TNF inhibitors should or should not be used with methotrexate for PsA.
“This study was not adequately powered to say that, but there is some indication, and I think that this is food for thought for future further analysis because the data in the literature are discrepant in this respect,” Dr. Smolen said.
In non-methotrexate users in SPIRIT-H2H, the ACR20 responses were 53% with ixekinumab vs. 40% with adalimumab, ACR50 responses were 72% vs. 60%, and ACR70 responses were 41% vs. 27%, respectively, he said noting that the difference for ACR70 was statistically significant, and that the ACR70 response with ixekinumab was about the same as the ACR50 for adalimumab.
As for ACR20, ACR50, and ACR70 responses in methotrexate users, “the lines criss-crossed” early on, he said, but all were “slightly superior” with adalimumab than with ixekizumab at 52 weeks (75% vs. 68%, 56% vs. 48%, and 39% vs. 32%, respectively).
Study participants had a mean age of 48 years and had active PsA with at least 3/66 tender joints, at least 3/68 swollen joints, at least 3% psoriasis body surface area involvement, no prior treatment with bDMARDs, and prior inadequate response to one or more conventional synthetic DMARDs. Treatment was dosed according to drug labeling through 52 weeks.
The safety profiles of both agents were consistent with previous reports; treatment-emergent adverse events occurred in 73.9% of ixekizumab and 68.6% of adalimumab patients, and serious adverse events occurred in 4.2% and 12.4%, respectively.
“On the other hand, ixekizumab had more injection site reactions: 11% vs. close to 4%,” he said, noting that 4.2% of the ixekizumab patients and 7.4% of the adalimumab patients discontinued treatment because of adverse events. No deaths occurred in either group.
As reported previously in Annals of the Rheumatic Diseases, ixekizumab was superior to adalimumab for simultaneous achievement of ACR50 and PASI100 at 24 weeks, and these final 52-week results confirm those results, he said.
The study was funded by Eli Lilly, which markets ixekizumab. Dr. Smolen reported research grants and/or honoraria from Eli Lilly and AbbVie, which markets adalimumab, as well as many other pharmaceutical companies.
SOURCE: Smolen J et al. Arthritis Rheumatol. 2019;71(suppl 10), Abstract L20.
ATLANTA – Ixekizumab (Taltz) provided significantly greater improvement in joint and skin symptoms, compared with adalimumab (Humira), in biologic-naive patients with active psoriatic arthritis (PsA), according to final 52-week safety and efficacy results from the randomized SPIRIT-H2H study.
The high-affinity monoclonal antibody against interleukin-17A also performed at least as well as the tumor necrosis factor (TNF)–inhibitor adalimumab across multiple PsA domains and regardless of methotrexate use, Josef Smolen, MD, reported during a late-breaking abstract session at the annual meeting of the American College of Rheumatology.
Multiple biologic disease-modifying antirheumatic drugs (bDMARDs) are available for the treatment of PsA, but few studies have directly compared their efficacy and safety, said Dr. Smolen of the Medical University of Vienna. He noted that the SPIRIT-H2H study aimed to compare ixekizumab and adalimumab and also to address “one of the most clinically relevant questions for clinicians,” which relates to the efficacy of bDMARDs with and without concomitant methotrexate.
Ixekizumab is approved for adults with active PsA and moderate to severe plaque psoriasis, but TNF inhibitors like adalimumab have long been considered the gold standard for PsA treatment, he explained.
Of 283 patients with PsA randomized to receive ixekizumab and 283 randomized to receive adalimumab, 87% and 84%, respectively, completed week 52 of the head-to-head, open-label study comparing the bDMARDs. Treatment with ixekizumab achieved the primary endpoint of simultaneous improvement of 50% on ACR response criteria (ACR50) and 100% on the Psoriasis Area and Severity Index (PASI100) in 39% of patients, which was significantly higher than the rate of 26% with adalimumab, Dr. Smolen said.
Ixekizumab also performed at least as well as adalimumab for the secondary outcome measures of ACR50 response (50% in both groups) and PASI100 response (64% vs. 41%), as well as for all other outcomes measures, including multiple musculoskeletal PsA domains, he said.
“Remarkably ... at 1 year, more than one-third of the patients achieved an ACR70 in both groups, and half of the patients achieved an ACR50,” he added, noting that the ACR100 responses were in line with previous investigations.
Stratification by methotrexate use showed that the simultaneous ACR50 and PASI100 response rates were improved with ixekizumab versus adalimumab both in users and nonusers of methotrexate (39% vs. 30% and 40% vs. 20%, respectively). This finding highlights the ongoing debate about whether TNF inhibitors should or should not be used with methotrexate for PsA.
“This study was not adequately powered to say that, but there is some indication, and I think that this is food for thought for future further analysis because the data in the literature are discrepant in this respect,” Dr. Smolen said.
In non-methotrexate users in SPIRIT-H2H, the ACR20 responses were 53% with ixekinumab vs. 40% with adalimumab, ACR50 responses were 72% vs. 60%, and ACR70 responses were 41% vs. 27%, respectively, he said noting that the difference for ACR70 was statistically significant, and that the ACR70 response with ixekinumab was about the same as the ACR50 for adalimumab.
As for ACR20, ACR50, and ACR70 responses in methotrexate users, “the lines criss-crossed” early on, he said, but all were “slightly superior” with adalimumab than with ixekizumab at 52 weeks (75% vs. 68%, 56% vs. 48%, and 39% vs. 32%, respectively).
Study participants had a mean age of 48 years and had active PsA with at least 3/66 tender joints, at least 3/68 swollen joints, at least 3% psoriasis body surface area involvement, no prior treatment with bDMARDs, and prior inadequate response to one or more conventional synthetic DMARDs. Treatment was dosed according to drug labeling through 52 weeks.
The safety profiles of both agents were consistent with previous reports; treatment-emergent adverse events occurred in 73.9% of ixekizumab and 68.6% of adalimumab patients, and serious adverse events occurred in 4.2% and 12.4%, respectively.
“On the other hand, ixekizumab had more injection site reactions: 11% vs. close to 4%,” he said, noting that 4.2% of the ixekizumab patients and 7.4% of the adalimumab patients discontinued treatment because of adverse events. No deaths occurred in either group.
As reported previously in Annals of the Rheumatic Diseases, ixekizumab was superior to adalimumab for simultaneous achievement of ACR50 and PASI100 at 24 weeks, and these final 52-week results confirm those results, he said.
The study was funded by Eli Lilly, which markets ixekizumab. Dr. Smolen reported research grants and/or honoraria from Eli Lilly and AbbVie, which markets adalimumab, as well as many other pharmaceutical companies.
SOURCE: Smolen J et al. Arthritis Rheumatol. 2019;71(suppl 10), Abstract L20.
REPORTING FROM ACR 2019
Scalp Psoriasis Considerations
1. Blakely K, Gooderham M. Management of scalp psoriasis: current perspectives. Psoriasis (Auckl). 2016;6:33-40.
2. Krueger G, Koo J, Lebwohl M, et al. The impact of psoriasis on quality of life: results of a 1998 National Psoriasis Foundation patient-membership survey. Arch Dermatol. 2001;137:280-284.
3. Merola JF, Li T, Li WQ, et al. Prevalence of psoriasis phenotypes among men and women in the USA. Clin Exp Dermatol. 2016;41:486-489.
4. Frez ML, Asawanonda P, Gunasekara C, et al. Recommendations for a patient-centered approach to the assessment and treatment of scalp psoriasis: a consensus statement from the Asia Scalp Psoriasis Study Group. J Dermatol Treat. 2014;25:38-45.
5. van de Kerkhof PC, Franssen ME. Psoriasis of the scalp. diagnosis and management. Am J Clin Dermatol. 2001;2:159-165.
6. Chan CS, Van Voorhees AS, Lebwohl MG, et al. Treatment of severe scalp psoriasis: from the Medical Board of the National Psoriasis Foundation. J Am Acad Dermatol. 2009;60:962-971.
7. Aldredge LM, Higham RC. Manifestations and management of difficult-to-treat psoriasis. J Dermatol Nurses Assoc. 2018;10:189-197.
8. Dopytalska K, Sobolewski P, Blaszczak A, et al. Psoriasis in special localizations. Reumatologia. 2018;56:392-398.
9. Papp K, Berth-Jones J, Kragballe K, et al. Scalp psoriasis: a review of current topical treatment options. J Eur Acad Dermatol Venereol. 2007;21:1151-1160.
10. Kircik LH, Kumar S. Scalp psoriasis. J Drugs Dermatol. 2010;9(8 suppl):S101-S105.
11. Wozel G. Psoriasis treatment in difficult locations: scalp, nails, and intertriginous areas. Clin Dermatol. 2008;26:448-459.
12. Sampogna F, Linder D, Piaserico S, et al. Quality of life assessment of patients with scalp dermatitis using the Italian version of the Scalpdex. Acta Dermato-Venereologica. 2014;94:411-414.
13. Crowley J. Scalp psoriasis: an overview of the disease and available therapies. J Drugs Dermatol. 2010;9:912-918.
14. Shah VV, Lee EB, Reddy SP, et al. Scalp psoriasis with increased hair density. Cutis. 2018;102:63-64.
15. George SM, Taylor MR, Farrant PB. Psoriatic alopecia. Clin Exp Dermatol. 2015;40:717-721.
16. Shuster S. Psoriatic alopecia. Br J Dermatol. 1972;87:73-77.
17. Wyatt E, Bottoms E, Comaish S. Abnormal hair shafts in psoriasis on scanning electron microscopy. Br J Dermatol. 1972;87:368-373.
18. Schoorl WJ, van Baar HJ, van de Kerkhof PC. The hair root pattern in psoriasis of the scalp. Acta Derm Venereol. 1992;72:141-142.
1. Blakely K, Gooderham M. Management of scalp psoriasis: current perspectives. Psoriasis (Auckl). 2016;6:33-40.
2. Krueger G, Koo J, Lebwohl M, et al. The impact of psoriasis on quality of life: results of a 1998 National Psoriasis Foundation patient-membership survey. Arch Dermatol. 2001;137:280-284.
3. Merola JF, Li T, Li WQ, et al. Prevalence of psoriasis phenotypes among men and women in the USA. Clin Exp Dermatol. 2016;41:486-489.
4. Frez ML, Asawanonda P, Gunasekara C, et al. Recommendations for a patient-centered approach to the assessment and treatment of scalp psoriasis: a consensus statement from the Asia Scalp Psoriasis Study Group. J Dermatol Treat. 2014;25:38-45.
5. van de Kerkhof PC, Franssen ME. Psoriasis of the scalp. diagnosis and management. Am J Clin Dermatol. 2001;2:159-165.
6. Chan CS, Van Voorhees AS, Lebwohl MG, et al. Treatment of severe scalp psoriasis: from the Medical Board of the National Psoriasis Foundation. J Am Acad Dermatol. 2009;60:962-971.
7. Aldredge LM, Higham RC. Manifestations and management of difficult-to-treat psoriasis. J Dermatol Nurses Assoc. 2018;10:189-197.
8. Dopytalska K, Sobolewski P, Blaszczak A, et al. Psoriasis in special localizations. Reumatologia. 2018;56:392-398.
9. Papp K, Berth-Jones J, Kragballe K, et al. Scalp psoriasis: a review of current topical treatment options. J Eur Acad Dermatol Venereol. 2007;21:1151-1160.
10. Kircik LH, Kumar S. Scalp psoriasis. J Drugs Dermatol. 2010;9(8 suppl):S101-S105.
11. Wozel G. Psoriasis treatment in difficult locations: scalp, nails, and intertriginous areas. Clin Dermatol. 2008;26:448-459.
12. Sampogna F, Linder D, Piaserico S, et al. Quality of life assessment of patients with scalp dermatitis using the Italian version of the Scalpdex. Acta Dermato-Venereologica. 2014;94:411-414.
13. Crowley J. Scalp psoriasis: an overview of the disease and available therapies. J Drugs Dermatol. 2010;9:912-918.
14. Shah VV, Lee EB, Reddy SP, et al. Scalp psoriasis with increased hair density. Cutis. 2018;102:63-64.
15. George SM, Taylor MR, Farrant PB. Psoriatic alopecia. Clin Exp Dermatol. 2015;40:717-721.
16. Shuster S. Psoriatic alopecia. Br J Dermatol. 1972;87:73-77.
17. Wyatt E, Bottoms E, Comaish S. Abnormal hair shafts in psoriasis on scanning electron microscopy. Br J Dermatol. 1972;87:368-373.
18. Schoorl WJ, van Baar HJ, van de Kerkhof PC. The hair root pattern in psoriasis of the scalp. Acta Derm Venereol. 1992;72:141-142.
1. Blakely K, Gooderham M. Management of scalp psoriasis: current perspectives. Psoriasis (Auckl). 2016;6:33-40.
2. Krueger G, Koo J, Lebwohl M, et al. The impact of psoriasis on quality of life: results of a 1998 National Psoriasis Foundation patient-membership survey. Arch Dermatol. 2001;137:280-284.
3. Merola JF, Li T, Li WQ, et al. Prevalence of psoriasis phenotypes among men and women in the USA. Clin Exp Dermatol. 2016;41:486-489.
4. Frez ML, Asawanonda P, Gunasekara C, et al. Recommendations for a patient-centered approach to the assessment and treatment of scalp psoriasis: a consensus statement from the Asia Scalp Psoriasis Study Group. J Dermatol Treat. 2014;25:38-45.
5. van de Kerkhof PC, Franssen ME. Psoriasis of the scalp. diagnosis and management. Am J Clin Dermatol. 2001;2:159-165.
6. Chan CS, Van Voorhees AS, Lebwohl MG, et al. Treatment of severe scalp psoriasis: from the Medical Board of the National Psoriasis Foundation. J Am Acad Dermatol. 2009;60:962-971.
7. Aldredge LM, Higham RC. Manifestations and management of difficult-to-treat psoriasis. J Dermatol Nurses Assoc. 2018;10:189-197.
8. Dopytalska K, Sobolewski P, Blaszczak A, et al. Psoriasis in special localizations. Reumatologia. 2018;56:392-398.
9. Papp K, Berth-Jones J, Kragballe K, et al. Scalp psoriasis: a review of current topical treatment options. J Eur Acad Dermatol Venereol. 2007;21:1151-1160.
10. Kircik LH, Kumar S. Scalp psoriasis. J Drugs Dermatol. 2010;9(8 suppl):S101-S105.
11. Wozel G. Psoriasis treatment in difficult locations: scalp, nails, and intertriginous areas. Clin Dermatol. 2008;26:448-459.
12. Sampogna F, Linder D, Piaserico S, et al. Quality of life assessment of patients with scalp dermatitis using the Italian version of the Scalpdex. Acta Dermato-Venereologica. 2014;94:411-414.
13. Crowley J. Scalp psoriasis: an overview of the disease and available therapies. J Drugs Dermatol. 2010;9:912-918.
14. Shah VV, Lee EB, Reddy SP, et al. Scalp psoriasis with increased hair density. Cutis. 2018;102:63-64.
15. George SM, Taylor MR, Farrant PB. Psoriatic alopecia. Clin Exp Dermatol. 2015;40:717-721.
16. Shuster S. Psoriatic alopecia. Br J Dermatol. 1972;87:73-77.
17. Wyatt E, Bottoms E, Comaish S. Abnormal hair shafts in psoriasis on scanning electron microscopy. Br J Dermatol. 1972;87:368-373.
18. Schoorl WJ, van Baar HJ, van de Kerkhof PC. The hair root pattern in psoriasis of the scalp. Acta Derm Venereol. 1992;72:141-142.
Satisfaction high among psoriasis patients on apremilast
MADRID – within the next 12 months than were patients on their first tumor necrosis factor (TNF) inhibitor, in a large retrospective national propensity score-matched study.
“This was surprising to us,” David L. Kaplan, MD, admitted in presenting the study findings at the annual congress of the European Academy of Dermatology and Venereology.
The surprise came because apremilast, a phosphodiesterase 4 (PDE4)-inhibitor, is less potent than the injectable biologics at driving down Psoriasis Area and Severity Index (PASI) scores.
“This is real-world data. And this is what patients are saying at 1 year: that they’re actually happier [with apremilast] and they’re not interested in changing,” said Dr. Kaplan, a dermatologist at the University of Kansas and in private practice in Overland Park, Kan.
He and his coinvestigators tapped the IBM Watson MarketScan health insurance claims database for 2015-2016 and identified 1,645 biologic-naive adults with psoriasis who started on apremilast therapy and an equal number of biologic-naive psoriasis patients who initiated treatment with a biologic, of whom 1,207 started on an TNF inhibitor and 438 began on an interleukin inhibitor, which was ustekinumab in 81% of cases. The TNF inhibitor cohort was split 80/20 between adalimumab and etanercept. The three groups – new users of apremilast, a TNF inhibitor, or an interleukin inhibitor – were propensity-matched based upon age, prior usage of systemic psoriasis therapies, Charlson Comorbidity Index scores, and other potential confounders.
The primary endpoint was the switch rate to a different psoriasis treatment within 12 months. The switch rate was significantly lower in patients who had started on apremilast than in those on a TNF inhibitor by a margin of 14% to 25%, while the 11% switch rate among patients on an interleukin inhibitor was not significantly different from the rate in the apremilast group.
“I think this data kind of gives us pause,” the dermatologist said. “As a clinician myself, when patients come back in the first question I always ask is, ‘How’re you doing? Are you happy?’ And at the end of the day, the data in terms of switch rates shows where patients are at. And that doesn’t really follow what we see with PASI scores.”
A secondary endpoint was the switch rate through 24 months. The same pattern held true: 24.9% in the apremilast starters, which was similar to the 22.9% in patients initiated on an interleukin inhibitor, and significantly less than the 39.1% rate in the TNF inhibitor group.
Among patients who switched medications within the first 12 months, the mean number of days to the switch was similar across all three groups.
The study had several limitations. Propensity score–matching is not a cure-all that can eradicate all potential biases. And the claims database didn’t include information on why patients switched, nor what their PASI scores were. “This is real-world data, and clinicians don’t do PASI scores in the real world,” he noted.
Audience member Andrew Blauvelt, MD, a dermatologist and president of the Oregon Medical Research Center, Portland, rose to challenge Dr. Kaplan’s conclusion that patients on apremilast were happier with their care.
“How can you rule out that it’s just practices that don’t use biologics, and they’re keeping patients on apremilast regardless of whether they’re better or happy because they’re not using biologics?” inquired Dr. Blauvelt.
Dr. Kaplan conceded that might well be a partial explanation for the results.
“Reluctance to use biologics is out there,” he agreed.
Dr. Kaplan reported serving as a consultant and paid speaker for Celgene, the study sponsor, as well as several other pharmaceutical companies.
SOURCE: Kaplan DL. EADV Abstract FC04.04.
MADRID – within the next 12 months than were patients on their first tumor necrosis factor (TNF) inhibitor, in a large retrospective national propensity score-matched study.
“This was surprising to us,” David L. Kaplan, MD, admitted in presenting the study findings at the annual congress of the European Academy of Dermatology and Venereology.
The surprise came because apremilast, a phosphodiesterase 4 (PDE4)-inhibitor, is less potent than the injectable biologics at driving down Psoriasis Area and Severity Index (PASI) scores.
“This is real-world data. And this is what patients are saying at 1 year: that they’re actually happier [with apremilast] and they’re not interested in changing,” said Dr. Kaplan, a dermatologist at the University of Kansas and in private practice in Overland Park, Kan.
He and his coinvestigators tapped the IBM Watson MarketScan health insurance claims database for 2015-2016 and identified 1,645 biologic-naive adults with psoriasis who started on apremilast therapy and an equal number of biologic-naive psoriasis patients who initiated treatment with a biologic, of whom 1,207 started on an TNF inhibitor and 438 began on an interleukin inhibitor, which was ustekinumab in 81% of cases. The TNF inhibitor cohort was split 80/20 between adalimumab and etanercept. The three groups – new users of apremilast, a TNF inhibitor, or an interleukin inhibitor – were propensity-matched based upon age, prior usage of systemic psoriasis therapies, Charlson Comorbidity Index scores, and other potential confounders.
The primary endpoint was the switch rate to a different psoriasis treatment within 12 months. The switch rate was significantly lower in patients who had started on apremilast than in those on a TNF inhibitor by a margin of 14% to 25%, while the 11% switch rate among patients on an interleukin inhibitor was not significantly different from the rate in the apremilast group.
“I think this data kind of gives us pause,” the dermatologist said. “As a clinician myself, when patients come back in the first question I always ask is, ‘How’re you doing? Are you happy?’ And at the end of the day, the data in terms of switch rates shows where patients are at. And that doesn’t really follow what we see with PASI scores.”
A secondary endpoint was the switch rate through 24 months. The same pattern held true: 24.9% in the apremilast starters, which was similar to the 22.9% in patients initiated on an interleukin inhibitor, and significantly less than the 39.1% rate in the TNF inhibitor group.
Among patients who switched medications within the first 12 months, the mean number of days to the switch was similar across all three groups.
The study had several limitations. Propensity score–matching is not a cure-all that can eradicate all potential biases. And the claims database didn’t include information on why patients switched, nor what their PASI scores were. “This is real-world data, and clinicians don’t do PASI scores in the real world,” he noted.
Audience member Andrew Blauvelt, MD, a dermatologist and president of the Oregon Medical Research Center, Portland, rose to challenge Dr. Kaplan’s conclusion that patients on apremilast were happier with their care.
“How can you rule out that it’s just practices that don’t use biologics, and they’re keeping patients on apremilast regardless of whether they’re better or happy because they’re not using biologics?” inquired Dr. Blauvelt.
Dr. Kaplan conceded that might well be a partial explanation for the results.
“Reluctance to use biologics is out there,” he agreed.
Dr. Kaplan reported serving as a consultant and paid speaker for Celgene, the study sponsor, as well as several other pharmaceutical companies.
SOURCE: Kaplan DL. EADV Abstract FC04.04.
MADRID – within the next 12 months than were patients on their first tumor necrosis factor (TNF) inhibitor, in a large retrospective national propensity score-matched study.
“This was surprising to us,” David L. Kaplan, MD, admitted in presenting the study findings at the annual congress of the European Academy of Dermatology and Venereology.
The surprise came because apremilast, a phosphodiesterase 4 (PDE4)-inhibitor, is less potent than the injectable biologics at driving down Psoriasis Area and Severity Index (PASI) scores.
“This is real-world data. And this is what patients are saying at 1 year: that they’re actually happier [with apremilast] and they’re not interested in changing,” said Dr. Kaplan, a dermatologist at the University of Kansas and in private practice in Overland Park, Kan.
He and his coinvestigators tapped the IBM Watson MarketScan health insurance claims database for 2015-2016 and identified 1,645 biologic-naive adults with psoriasis who started on apremilast therapy and an equal number of biologic-naive psoriasis patients who initiated treatment with a biologic, of whom 1,207 started on an TNF inhibitor and 438 began on an interleukin inhibitor, which was ustekinumab in 81% of cases. The TNF inhibitor cohort was split 80/20 between adalimumab and etanercept. The three groups – new users of apremilast, a TNF inhibitor, or an interleukin inhibitor – were propensity-matched based upon age, prior usage of systemic psoriasis therapies, Charlson Comorbidity Index scores, and other potential confounders.
The primary endpoint was the switch rate to a different psoriasis treatment within 12 months. The switch rate was significantly lower in patients who had started on apremilast than in those on a TNF inhibitor by a margin of 14% to 25%, while the 11% switch rate among patients on an interleukin inhibitor was not significantly different from the rate in the apremilast group.
“I think this data kind of gives us pause,” the dermatologist said. “As a clinician myself, when patients come back in the first question I always ask is, ‘How’re you doing? Are you happy?’ And at the end of the day, the data in terms of switch rates shows where patients are at. And that doesn’t really follow what we see with PASI scores.”
A secondary endpoint was the switch rate through 24 months. The same pattern held true: 24.9% in the apremilast starters, which was similar to the 22.9% in patients initiated on an interleukin inhibitor, and significantly less than the 39.1% rate in the TNF inhibitor group.
Among patients who switched medications within the first 12 months, the mean number of days to the switch was similar across all three groups.
The study had several limitations. Propensity score–matching is not a cure-all that can eradicate all potential biases. And the claims database didn’t include information on why patients switched, nor what their PASI scores were. “This is real-world data, and clinicians don’t do PASI scores in the real world,” he noted.
Audience member Andrew Blauvelt, MD, a dermatologist and president of the Oregon Medical Research Center, Portland, rose to challenge Dr. Kaplan’s conclusion that patients on apremilast were happier with their care.
“How can you rule out that it’s just practices that don’t use biologics, and they’re keeping patients on apremilast regardless of whether they’re better or happy because they’re not using biologics?” inquired Dr. Blauvelt.
Dr. Kaplan conceded that might well be a partial explanation for the results.
“Reluctance to use biologics is out there,” he agreed.
Dr. Kaplan reported serving as a consultant and paid speaker for Celgene, the study sponsor, as well as several other pharmaceutical companies.
SOURCE: Kaplan DL. EADV Abstract FC04.04.
REPORTING FROM EADV 2019
Ixekizumab effective over long term for psoriasis
The according to a study published in the Journal of the European Academy of Dermatology and Venereology.
The UNCOVER-3 trial was a double-blind, multicenter, phase 3 study in 1,346 individuals with moderate to severe psoriasis who were randomized to placebo, 80 mg of ixekizumab every 2 or 4 weeks, or 50 mg of etanercept twice weekly. At week 12, all patients were transferred to 80 mg of ixekizumab every 4 weeks for the long-term extension period, and after 60 weeks, some were dose adjusted to 80 mg every 2 weeks.
At week 204, 48.3% of the 385 patients who were receiving treatment either every 2 or 4 weeks achieved Psoriasis Area and Severity Index (PASI) 100 according to the modified nonresponder imputation method to summarize efficacy – in which patients who drop out of the study are counted as nonresponders – 66.4% achieved PASI 90 and 82.8% achieved PASI 75.
Using the as-observed method for assessing efficacy, 67.1% of patients achieved PASI 100, 87.8% achieved PASI 90, and 98.2% achieved PASI 75. Using the multiple-imputation method, those same figures were 52.7%, 73.3%, and 94.8% respectively.
They also saw consistently high response rates according to the static Physician’s Global Assessment (sPGA) score, which goes from 0 (clear) to 5 (severe disease). Using the as-observed, multiple imputation and modified nonresponder imputation methods, 68.9%, 54.6%, and 49.7% of patients respectively achieved a score of 0.
The study also saw complete resolution in 95.8% of patients with baseline palmoplantar involvement, 75.9% of those with baseline nail involvement, and 87.1% of those with scalp involvement, using the as-observed method.
“These results corroborate the results that were reported previously in patients with moderate to severe psoriasis,” wrote Mark G. Lebwohl, MD, professor and chair of the department of dermatology at the Icahn School of Medicine at Mount Sinai, New York, and coauthors. “Sustained high response was observed with all the efficacy parameters such as PASI 75, 90, 100, and sPGA (0) or (0, 1), regardless of the statistical analyses ... performed.”
The majority of adverse events were mild or moderate, but serious adverse events were seen in 18.1% of patients in the long-term extension and 9.4% of patients stopped taking the study drug because of adverse events.
The most frequently reported adverse events of special interest were infections, such as nasopharyngitis (28.5% of patients), upper respiratory tract infections (10.8%), and Candida infection (5.2%). However, clinically significant neutropenia only occurred in 0.7% of patients, and malignancies occurred in 2.2% of patients.
“These safety findings support the consistency in the safety profile of [ixekizumab] treatment with no new signals occurring even after the longer exposure,” the authors wrote.
The study was funded by Eli Lilly, which manufactures ixekizumab. Two authors were employees of Eli Lilly and own company stocks. The remaining three authors reported receiving research funding and consultancies from different pharmaceutical companies, including Eli Lilly.
SOURCE: Lebwohl MG et al. J Eur Acad Dermatol Venereol. 2019 Sep 3. doi: 10.1111/jdv.15921.
The according to a study published in the Journal of the European Academy of Dermatology and Venereology.
The UNCOVER-3 trial was a double-blind, multicenter, phase 3 study in 1,346 individuals with moderate to severe psoriasis who were randomized to placebo, 80 mg of ixekizumab every 2 or 4 weeks, or 50 mg of etanercept twice weekly. At week 12, all patients were transferred to 80 mg of ixekizumab every 4 weeks for the long-term extension period, and after 60 weeks, some were dose adjusted to 80 mg every 2 weeks.
At week 204, 48.3% of the 385 patients who were receiving treatment either every 2 or 4 weeks achieved Psoriasis Area and Severity Index (PASI) 100 according to the modified nonresponder imputation method to summarize efficacy – in which patients who drop out of the study are counted as nonresponders – 66.4% achieved PASI 90 and 82.8% achieved PASI 75.
Using the as-observed method for assessing efficacy, 67.1% of patients achieved PASI 100, 87.8% achieved PASI 90, and 98.2% achieved PASI 75. Using the multiple-imputation method, those same figures were 52.7%, 73.3%, and 94.8% respectively.
They also saw consistently high response rates according to the static Physician’s Global Assessment (sPGA) score, which goes from 0 (clear) to 5 (severe disease). Using the as-observed, multiple imputation and modified nonresponder imputation methods, 68.9%, 54.6%, and 49.7% of patients respectively achieved a score of 0.
The study also saw complete resolution in 95.8% of patients with baseline palmoplantar involvement, 75.9% of those with baseline nail involvement, and 87.1% of those with scalp involvement, using the as-observed method.
“These results corroborate the results that were reported previously in patients with moderate to severe psoriasis,” wrote Mark G. Lebwohl, MD, professor and chair of the department of dermatology at the Icahn School of Medicine at Mount Sinai, New York, and coauthors. “Sustained high response was observed with all the efficacy parameters such as PASI 75, 90, 100, and sPGA (0) or (0, 1), regardless of the statistical analyses ... performed.”
The majority of adverse events were mild or moderate, but serious adverse events were seen in 18.1% of patients in the long-term extension and 9.4% of patients stopped taking the study drug because of adverse events.
The most frequently reported adverse events of special interest were infections, such as nasopharyngitis (28.5% of patients), upper respiratory tract infections (10.8%), and Candida infection (5.2%). However, clinically significant neutropenia only occurred in 0.7% of patients, and malignancies occurred in 2.2% of patients.
“These safety findings support the consistency in the safety profile of [ixekizumab] treatment with no new signals occurring even after the longer exposure,” the authors wrote.
The study was funded by Eli Lilly, which manufactures ixekizumab. Two authors were employees of Eli Lilly and own company stocks. The remaining three authors reported receiving research funding and consultancies from different pharmaceutical companies, including Eli Lilly.
SOURCE: Lebwohl MG et al. J Eur Acad Dermatol Venereol. 2019 Sep 3. doi: 10.1111/jdv.15921.
The according to a study published in the Journal of the European Academy of Dermatology and Venereology.
The UNCOVER-3 trial was a double-blind, multicenter, phase 3 study in 1,346 individuals with moderate to severe psoriasis who were randomized to placebo, 80 mg of ixekizumab every 2 or 4 weeks, or 50 mg of etanercept twice weekly. At week 12, all patients were transferred to 80 mg of ixekizumab every 4 weeks for the long-term extension period, and after 60 weeks, some were dose adjusted to 80 mg every 2 weeks.
At week 204, 48.3% of the 385 patients who were receiving treatment either every 2 or 4 weeks achieved Psoriasis Area and Severity Index (PASI) 100 according to the modified nonresponder imputation method to summarize efficacy – in which patients who drop out of the study are counted as nonresponders – 66.4% achieved PASI 90 and 82.8% achieved PASI 75.
Using the as-observed method for assessing efficacy, 67.1% of patients achieved PASI 100, 87.8% achieved PASI 90, and 98.2% achieved PASI 75. Using the multiple-imputation method, those same figures were 52.7%, 73.3%, and 94.8% respectively.
They also saw consistently high response rates according to the static Physician’s Global Assessment (sPGA) score, which goes from 0 (clear) to 5 (severe disease). Using the as-observed, multiple imputation and modified nonresponder imputation methods, 68.9%, 54.6%, and 49.7% of patients respectively achieved a score of 0.
The study also saw complete resolution in 95.8% of patients with baseline palmoplantar involvement, 75.9% of those with baseline nail involvement, and 87.1% of those with scalp involvement, using the as-observed method.
“These results corroborate the results that were reported previously in patients with moderate to severe psoriasis,” wrote Mark G. Lebwohl, MD, professor and chair of the department of dermatology at the Icahn School of Medicine at Mount Sinai, New York, and coauthors. “Sustained high response was observed with all the efficacy parameters such as PASI 75, 90, 100, and sPGA (0) or (0, 1), regardless of the statistical analyses ... performed.”
The majority of adverse events were mild or moderate, but serious adverse events were seen in 18.1% of patients in the long-term extension and 9.4% of patients stopped taking the study drug because of adverse events.
The most frequently reported adverse events of special interest were infections, such as nasopharyngitis (28.5% of patients), upper respiratory tract infections (10.8%), and Candida infection (5.2%). However, clinically significant neutropenia only occurred in 0.7% of patients, and malignancies occurred in 2.2% of patients.
“These safety findings support the consistency in the safety profile of [ixekizumab] treatment with no new signals occurring even after the longer exposure,” the authors wrote.
The study was funded by Eli Lilly, which manufactures ixekizumab. Two authors were employees of Eli Lilly and own company stocks. The remaining three authors reported receiving research funding and consultancies from different pharmaceutical companies, including Eli Lilly.
SOURCE: Lebwohl MG et al. J Eur Acad Dermatol Venereol. 2019 Sep 3. doi: 10.1111/jdv.15921.
FROM THE JOURNAL OF THE EUROPEAN ACADEMY OF DERMATOLOGY AND VENEREOLOGY
Key clinical point: Long-term follow-up shows ixekizumab efficacy in moderate to severe psoriasis.
Major finding: At 4 years, 66.5% of patients on ixekizumab for psoriasis achieved Psoriasis Area and Severity Index 100.
Study details: A long-term extension of the UNCOVER-3 double-blind, multicenter, phase 3 study in 1,346 individuals with psoriasis.
Disclosures: The study was funded by Eli Lilly, which manufactures ixekizumab. Two authors were employees of Eli Lilly and own company stocks. The remaining three authors reported receiving research funding and consultancies from different pharmaceutical companies, including Eli Lilly.
Source: Lebwohl MG et al. J Eur Acad Dermatol Venereol. 2019 Sep 3. doi: 10.1111/jdv.15921.
FDA announces approval of fifth adalimumab biosimilar, Abrilada
The Food and Drug Administration has cleared adalimumab-afzb (Abrilada) as the fifth approved Humira biosimilar and the 25th approved biosimilar drug overall, the agency said in a Nov. 15 announcement.
According to a press release from Pfizer, approval for Abrilada was based on review of a comprehensive data package demonstrating biosimilarity of the drug to the reference product. This included data from a clinical comparative study, which found no clinically meaningful difference between Abrilada and the reference in terms of efficacy, safety, and immunogenicity in patients with moderate to severe rheumatoid arthritis (RA). In addition to RA, Abrilada is indicated for juvenile idiopathic arthritis, psoriatic arthritis, ankylosing spondylitis, adult Crohn’s disease, ulcerative colitis, and plaque psoriasis.
Common adverse events in adalimumab clinical trials included infection, injection-site reactions, headache, and rash.
Pfizer said that it “is working to make Abrilada available to U.S. patients as soon as feasible based on the terms of our agreement with AbbVie [the manufacturer of Humira]. Our current plans are to launch in 2023.”
The Food and Drug Administration has cleared adalimumab-afzb (Abrilada) as the fifth approved Humira biosimilar and the 25th approved biosimilar drug overall, the agency said in a Nov. 15 announcement.
According to a press release from Pfizer, approval for Abrilada was based on review of a comprehensive data package demonstrating biosimilarity of the drug to the reference product. This included data from a clinical comparative study, which found no clinically meaningful difference between Abrilada and the reference in terms of efficacy, safety, and immunogenicity in patients with moderate to severe rheumatoid arthritis (RA). In addition to RA, Abrilada is indicated for juvenile idiopathic arthritis, psoriatic arthritis, ankylosing spondylitis, adult Crohn’s disease, ulcerative colitis, and plaque psoriasis.
Common adverse events in adalimumab clinical trials included infection, injection-site reactions, headache, and rash.
Pfizer said that it “is working to make Abrilada available to U.S. patients as soon as feasible based on the terms of our agreement with AbbVie [the manufacturer of Humira]. Our current plans are to launch in 2023.”
The Food and Drug Administration has cleared adalimumab-afzb (Abrilada) as the fifth approved Humira biosimilar and the 25th approved biosimilar drug overall, the agency said in a Nov. 15 announcement.
According to a press release from Pfizer, approval for Abrilada was based on review of a comprehensive data package demonstrating biosimilarity of the drug to the reference product. This included data from a clinical comparative study, which found no clinically meaningful difference between Abrilada and the reference in terms of efficacy, safety, and immunogenicity in patients with moderate to severe rheumatoid arthritis (RA). In addition to RA, Abrilada is indicated for juvenile idiopathic arthritis, psoriatic arthritis, ankylosing spondylitis, adult Crohn’s disease, ulcerative colitis, and plaque psoriasis.
Common adverse events in adalimumab clinical trials included infection, injection-site reactions, headache, and rash.
Pfizer said that it “is working to make Abrilada available to U.S. patients as soon as feasible based on the terms of our agreement with AbbVie [the manufacturer of Humira]. Our current plans are to launch in 2023.”
Early use of ustekinumab levels could predict psoriasis outcomes
Early ustekinumab levels are significantly associated with a 75% reduction in Psoriasis Area and Severity Index scores from baseline, according to data from 491 adults with psoriasis.
“Evidence suggests that ustekinumab dosing is suboptimal in some patients,” because of factors including weight-based dosing and dosing intervals; therefore “individualized dose optimization and therapeutic drug monitoring (TDM) of ustekinumab may have clinical utility,” Teresa Tsakok, MRCP, of King’s College London, and colleagues wrote in JAMA Dermatology.
The researchers identified 491 adults with psoriasis who were part of the BSTOP (Biomarkers of Systemic Treatment Outcomes in Psoriasis) cohort study in the United Kingdom. Blood samples were collected during clinical reviews to assess ustekinumab levels, and primary treatment response was 75% reduction in Psoriasis Area and Severity Index (PASI 75) scores.
Ustekinumab levels measured from 1-12 weeks after the start of treatment were significantly associated with PASI 75 after 6 months (odds ratio, 1.38) after controlling for factors including baseline PASI scores, age, and ustekinumab dose. The association, however, did not hold for other PASI outcomes, including PASI 90 and PASI scores of 1.5 or less.
The participants had at least one serum sample collected at 0-56 weeks from the start of treatment and at least one PSAI score measured within the first year of treatment. The average baseline PASI score was 13.3, the average body mass index was 32 kg/m2, and 65% of the patients were male.
Antidrug antibodies were detected in 17 patients (3.5% of the study population), compared with a rate of 37.5% in patients from the same study cohort who were taking adalimumab, the researchers noted.
The study findings were limited by several factors, including a dropoff in patient numbers over the study period and the difficulty in accounting for the association between drug level and treatment response in a logistic regression model, the researchers said.
The results, however, suggest “that adequate drug exposure early in the treatment cycle may be particularly important in determining clinical outcome with ustekinumab,” and that “future work should focus on pharmacokinetic-pharmacodynamic modeling of the whole time course of response to ustekinumab” as a key step toward personalized treatment regimens, they concluded.
The study was supported by several entities, including the Medical Research Council (MRC), the National Institute for Health Research Biomedical Research Center and the Psoriasis Association. Dr. Tsakok had no financial conflicts to disclose and was supported by an MRC Clinical Research Training Fellowship.
SOURCE: Tsakok T et al. JAMA Dermatol. 2019 Sep 18. doi: 10.1001/jamadermatol.2019.1783.
Early ustekinumab levels are significantly associated with a 75% reduction in Psoriasis Area and Severity Index scores from baseline, according to data from 491 adults with psoriasis.
“Evidence suggests that ustekinumab dosing is suboptimal in some patients,” because of factors including weight-based dosing and dosing intervals; therefore “individualized dose optimization and therapeutic drug monitoring (TDM) of ustekinumab may have clinical utility,” Teresa Tsakok, MRCP, of King’s College London, and colleagues wrote in JAMA Dermatology.
The researchers identified 491 adults with psoriasis who were part of the BSTOP (Biomarkers of Systemic Treatment Outcomes in Psoriasis) cohort study in the United Kingdom. Blood samples were collected during clinical reviews to assess ustekinumab levels, and primary treatment response was 75% reduction in Psoriasis Area and Severity Index (PASI 75) scores.
Ustekinumab levels measured from 1-12 weeks after the start of treatment were significantly associated with PASI 75 after 6 months (odds ratio, 1.38) after controlling for factors including baseline PASI scores, age, and ustekinumab dose. The association, however, did not hold for other PASI outcomes, including PASI 90 and PASI scores of 1.5 or less.
The participants had at least one serum sample collected at 0-56 weeks from the start of treatment and at least one PSAI score measured within the first year of treatment. The average baseline PASI score was 13.3, the average body mass index was 32 kg/m2, and 65% of the patients were male.
Antidrug antibodies were detected in 17 patients (3.5% of the study population), compared with a rate of 37.5% in patients from the same study cohort who were taking adalimumab, the researchers noted.
The study findings were limited by several factors, including a dropoff in patient numbers over the study period and the difficulty in accounting for the association between drug level and treatment response in a logistic regression model, the researchers said.
The results, however, suggest “that adequate drug exposure early in the treatment cycle may be particularly important in determining clinical outcome with ustekinumab,” and that “future work should focus on pharmacokinetic-pharmacodynamic modeling of the whole time course of response to ustekinumab” as a key step toward personalized treatment regimens, they concluded.
The study was supported by several entities, including the Medical Research Council (MRC), the National Institute for Health Research Biomedical Research Center and the Psoriasis Association. Dr. Tsakok had no financial conflicts to disclose and was supported by an MRC Clinical Research Training Fellowship.
SOURCE: Tsakok T et al. JAMA Dermatol. 2019 Sep 18. doi: 10.1001/jamadermatol.2019.1783.
Early ustekinumab levels are significantly associated with a 75% reduction in Psoriasis Area and Severity Index scores from baseline, according to data from 491 adults with psoriasis.
“Evidence suggests that ustekinumab dosing is suboptimal in some patients,” because of factors including weight-based dosing and dosing intervals; therefore “individualized dose optimization and therapeutic drug monitoring (TDM) of ustekinumab may have clinical utility,” Teresa Tsakok, MRCP, of King’s College London, and colleagues wrote in JAMA Dermatology.
The researchers identified 491 adults with psoriasis who were part of the BSTOP (Biomarkers of Systemic Treatment Outcomes in Psoriasis) cohort study in the United Kingdom. Blood samples were collected during clinical reviews to assess ustekinumab levels, and primary treatment response was 75% reduction in Psoriasis Area and Severity Index (PASI 75) scores.
Ustekinumab levels measured from 1-12 weeks after the start of treatment were significantly associated with PASI 75 after 6 months (odds ratio, 1.38) after controlling for factors including baseline PASI scores, age, and ustekinumab dose. The association, however, did not hold for other PASI outcomes, including PASI 90 and PASI scores of 1.5 or less.
The participants had at least one serum sample collected at 0-56 weeks from the start of treatment and at least one PSAI score measured within the first year of treatment. The average baseline PASI score was 13.3, the average body mass index was 32 kg/m2, and 65% of the patients were male.
Antidrug antibodies were detected in 17 patients (3.5% of the study population), compared with a rate of 37.5% in patients from the same study cohort who were taking adalimumab, the researchers noted.
The study findings were limited by several factors, including a dropoff in patient numbers over the study period and the difficulty in accounting for the association between drug level and treatment response in a logistic regression model, the researchers said.
The results, however, suggest “that adequate drug exposure early in the treatment cycle may be particularly important in determining clinical outcome with ustekinumab,” and that “future work should focus on pharmacokinetic-pharmacodynamic modeling of the whole time course of response to ustekinumab” as a key step toward personalized treatment regimens, they concluded.
The study was supported by several entities, including the Medical Research Council (MRC), the National Institute for Health Research Biomedical Research Center and the Psoriasis Association. Dr. Tsakok had no financial conflicts to disclose and was supported by an MRC Clinical Research Training Fellowship.
SOURCE: Tsakok T et al. JAMA Dermatol. 2019 Sep 18. doi: 10.1001/jamadermatol.2019.1783.
FROM JAMA DERMATOLOGY
Key clinical point: Measurement of ustekinumab levels early in treatment could help direct strategies and ensure correct dosage in psoriasis patients.
Major finding: Early serum ustekinumab levels 1-12 weeks after starting treatment were associated with a 75% reduction in PASI scores from baseline at 6 months.
Study details: The prospective, observational study involved 491 adults with psoriasis.
Disclosures: The study was supported by several entities, including the Medical Research Council (MRC), the National Institute for Health Research Biomedical Research Center, and the Psoriasis Association. Dr. Tsakok disclosed financial conflicts and was supported by an MRC Clinical Research Training Fellowship.
Source: Tsakok T et al. JAMA Dermatol. 2019 Sep 18. doi: 10.1001/jamadermatol.2019.1783.
Pink scaly rash on torso and extremities
Tumor necrosis factor–alpha (TNF-alpha) inhibitors are therapeutic agents used to treat a variety of inflammatory conditions such as rheumatoid arthritis and inflammatory bowel disease, as well as psoriasis of the skin (PSO) and psoriatic arthritis. In a 2017 systematic review, there were 216 reported cases of new-onset TNF-alpha inhibitor–induced psoriasis, with an estimated rate of 1 per 1,000. The cases thus far have had a wide range of presentations, the most common being plaque psoriasis, scalp psoriasis, as well as palmoplantar pustular psoriasis.1
A retrospective chart review study at Mayo clinic published in 2017 evaluated children younger than 19 years seen in 2003-2015 who developed new-onset or recurrent PSO with a history of inflammatory bowel disease being treated with anti-TNF-alpha therapy. The review showed variable latency in the development of PSO in these patients, although it typically occurred during inflammatory bowel disease remission.2 It is unclear whether there is an association between a personal or family history of psoriasis and development of these lesions.
TNF-alpha, interleukin (IL)–17) and interferon-alpha (IFN-alpha) are main cytokines that contribute to the development of psoriasis. The mechanism of action for paradoxical PSO/psoriasis in patients treated with anti-TNF is not clearly understood; however, many hypotheses are based on an imbalance between TNF-alpha and interferon-alpha – more specifically, an increased production of interferon-alpha. TNF-alpha inhibits the activity of plasmacytoid dendritic cells which are key producers of IFN-alpha. Because of this blockade, there is unopposed IFN-alpha production. Interferon-alpha allows for the expression of chemokines such as CXCR3, which favor T cells homing to the skin. IFN-alpha also stimulates and activates T cells to produce TNF-alpha and IL-17, which in turn sustains inflammatory mechanisms and allows for the development of psoriatic lesions.3
There are no universal management guidelines. Most of these patients’ treatment plans mirror standard psoriasis therapies while the main question remains the decision to continue the same anti-TNF therapy, change anti-TNF agents, or entirely switch classes of biologic or other systemic therapy. This decision in management requires several considerations: treatability of TNF-alpha inhibitor-induced psoriasis, the severity of background disease (i.e., rheumatoid arthritis, inflammatory bowel disease, other systemic condition), and whether the underlying disease is well controlled on current therapy, as well as the consideration of possible loss in efficacy if a drug is discontinued and then restarted at a later date.4
A reasonable initial approach in patients with well-controlled underlying disease and mild skin eruption is to continue anti-TNF therapy and manage skin topically with topical corticosteroids and/or phototherapy. In patients that either do not have well-controlled underlying disease or moderate skin involvement, changing to an alternative anti-TNF or other agent may be reasonable, and requires coordinated care with involved specialists. In the 2017 pediatric review mentioned previously, nearly half of the patients required a change in their initial anti-TNF-alpha agent despite conventional skin-directed therapies, and one-third of patients discontinued all anti-TNF-alpha therapy because of PSO.2
The psoriasiform papulosquamous features of this case along with the history suggests the diagnosis. Pityriasis rosea would be highly atypical on the feet and with the duration of findings. Lichen planus and atopic dermatitis morphology are inconsistent with this eruption, and coxsackie viral infection would have a shorter course.
Dr. Tracy is a research fellow in pediatric dermatology at Rady Children’s Hospital–San Diego and the University of California, San Diego. Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children’s Hospital–San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. Email them at pdnews@mdedge.com.
References
1. J Am Acad Dermatol. 2017 Feb;76(2):334-41.
2. Pediatr Dermatol. 2017 May;34(3):253-60.
3. RMD Open. 2016 Jul 15;2(2):e000239.
4. J Psoriasis Psoriatic Arthritis. 2019 Apr;4(2):70-80.
Tumor necrosis factor–alpha (TNF-alpha) inhibitors are therapeutic agents used to treat a variety of inflammatory conditions such as rheumatoid arthritis and inflammatory bowel disease, as well as psoriasis of the skin (PSO) and psoriatic arthritis. In a 2017 systematic review, there were 216 reported cases of new-onset TNF-alpha inhibitor–induced psoriasis, with an estimated rate of 1 per 1,000. The cases thus far have had a wide range of presentations, the most common being plaque psoriasis, scalp psoriasis, as well as palmoplantar pustular psoriasis.1
A retrospective chart review study at Mayo clinic published in 2017 evaluated children younger than 19 years seen in 2003-2015 who developed new-onset or recurrent PSO with a history of inflammatory bowel disease being treated with anti-TNF-alpha therapy. The review showed variable latency in the development of PSO in these patients, although it typically occurred during inflammatory bowel disease remission.2 It is unclear whether there is an association between a personal or family history of psoriasis and development of these lesions.
TNF-alpha, interleukin (IL)–17) and interferon-alpha (IFN-alpha) are main cytokines that contribute to the development of psoriasis. The mechanism of action for paradoxical PSO/psoriasis in patients treated with anti-TNF is not clearly understood; however, many hypotheses are based on an imbalance between TNF-alpha and interferon-alpha – more specifically, an increased production of interferon-alpha. TNF-alpha inhibits the activity of plasmacytoid dendritic cells which are key producers of IFN-alpha. Because of this blockade, there is unopposed IFN-alpha production. Interferon-alpha allows for the expression of chemokines such as CXCR3, which favor T cells homing to the skin. IFN-alpha also stimulates and activates T cells to produce TNF-alpha and IL-17, which in turn sustains inflammatory mechanisms and allows for the development of psoriatic lesions.3
There are no universal management guidelines. Most of these patients’ treatment plans mirror standard psoriasis therapies while the main question remains the decision to continue the same anti-TNF therapy, change anti-TNF agents, or entirely switch classes of biologic or other systemic therapy. This decision in management requires several considerations: treatability of TNF-alpha inhibitor-induced psoriasis, the severity of background disease (i.e., rheumatoid arthritis, inflammatory bowel disease, other systemic condition), and whether the underlying disease is well controlled on current therapy, as well as the consideration of possible loss in efficacy if a drug is discontinued and then restarted at a later date.4
A reasonable initial approach in patients with well-controlled underlying disease and mild skin eruption is to continue anti-TNF therapy and manage skin topically with topical corticosteroids and/or phototherapy. In patients that either do not have well-controlled underlying disease or moderate skin involvement, changing to an alternative anti-TNF or other agent may be reasonable, and requires coordinated care with involved specialists. In the 2017 pediatric review mentioned previously, nearly half of the patients required a change in their initial anti-TNF-alpha agent despite conventional skin-directed therapies, and one-third of patients discontinued all anti-TNF-alpha therapy because of PSO.2
The psoriasiform papulosquamous features of this case along with the history suggests the diagnosis. Pityriasis rosea would be highly atypical on the feet and with the duration of findings. Lichen planus and atopic dermatitis morphology are inconsistent with this eruption, and coxsackie viral infection would have a shorter course.
Dr. Tracy is a research fellow in pediatric dermatology at Rady Children’s Hospital–San Diego and the University of California, San Diego. Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children’s Hospital–San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. Email them at pdnews@mdedge.com.
References
1. J Am Acad Dermatol. 2017 Feb;76(2):334-41.
2. Pediatr Dermatol. 2017 May;34(3):253-60.
3. RMD Open. 2016 Jul 15;2(2):e000239.
4. J Psoriasis Psoriatic Arthritis. 2019 Apr;4(2):70-80.
Tumor necrosis factor–alpha (TNF-alpha) inhibitors are therapeutic agents used to treat a variety of inflammatory conditions such as rheumatoid arthritis and inflammatory bowel disease, as well as psoriasis of the skin (PSO) and psoriatic arthritis. In a 2017 systematic review, there were 216 reported cases of new-onset TNF-alpha inhibitor–induced psoriasis, with an estimated rate of 1 per 1,000. The cases thus far have had a wide range of presentations, the most common being plaque psoriasis, scalp psoriasis, as well as palmoplantar pustular psoriasis.1
A retrospective chart review study at Mayo clinic published in 2017 evaluated children younger than 19 years seen in 2003-2015 who developed new-onset or recurrent PSO with a history of inflammatory bowel disease being treated with anti-TNF-alpha therapy. The review showed variable latency in the development of PSO in these patients, although it typically occurred during inflammatory bowel disease remission.2 It is unclear whether there is an association between a personal or family history of psoriasis and development of these lesions.
TNF-alpha, interleukin (IL)–17) and interferon-alpha (IFN-alpha) are main cytokines that contribute to the development of psoriasis. The mechanism of action for paradoxical PSO/psoriasis in patients treated with anti-TNF is not clearly understood; however, many hypotheses are based on an imbalance between TNF-alpha and interferon-alpha – more specifically, an increased production of interferon-alpha. TNF-alpha inhibits the activity of plasmacytoid dendritic cells which are key producers of IFN-alpha. Because of this blockade, there is unopposed IFN-alpha production. Interferon-alpha allows for the expression of chemokines such as CXCR3, which favor T cells homing to the skin. IFN-alpha also stimulates and activates T cells to produce TNF-alpha and IL-17, which in turn sustains inflammatory mechanisms and allows for the development of psoriatic lesions.3
There are no universal management guidelines. Most of these patients’ treatment plans mirror standard psoriasis therapies while the main question remains the decision to continue the same anti-TNF therapy, change anti-TNF agents, or entirely switch classes of biologic or other systemic therapy. This decision in management requires several considerations: treatability of TNF-alpha inhibitor-induced psoriasis, the severity of background disease (i.e., rheumatoid arthritis, inflammatory bowel disease, other systemic condition), and whether the underlying disease is well controlled on current therapy, as well as the consideration of possible loss in efficacy if a drug is discontinued and then restarted at a later date.4
A reasonable initial approach in patients with well-controlled underlying disease and mild skin eruption is to continue anti-TNF therapy and manage skin topically with topical corticosteroids and/or phototherapy. In patients that either do not have well-controlled underlying disease or moderate skin involvement, changing to an alternative anti-TNF or other agent may be reasonable, and requires coordinated care with involved specialists. In the 2017 pediatric review mentioned previously, nearly half of the patients required a change in their initial anti-TNF-alpha agent despite conventional skin-directed therapies, and one-third of patients discontinued all anti-TNF-alpha therapy because of PSO.2
The psoriasiform papulosquamous features of this case along with the history suggests the diagnosis. Pityriasis rosea would be highly atypical on the feet and with the duration of findings. Lichen planus and atopic dermatitis morphology are inconsistent with this eruption, and coxsackie viral infection would have a shorter course.
Dr. Tracy is a research fellow in pediatric dermatology at Rady Children’s Hospital–San Diego and the University of California, San Diego. Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children’s Hospital–San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. Email them at pdnews@mdedge.com.
References
1. J Am Acad Dermatol. 2017 Feb;76(2):334-41.
2. Pediatr Dermatol. 2017 May;34(3):253-60.
3. RMD Open. 2016 Jul 15;2(2):e000239.
4. J Psoriasis Psoriatic Arthritis. 2019 Apr;4(2):70-80.
A 17-year-old male with a history of Crohn's disease, well controlled on infliximab, is seen for evaluation of a pink scaly rash on the torso, upper extremities, and lower extremities. The rash began 5 months previously and has been mostly asymptomatic. The patient denies pruritus or pain at the affected areas. There is no fever or drainage from any of the sites. The patient has not undergone any treatments. He does not have a personal or family history of chronic skin conditions.
On physical exam, he is noted to have numerous pink papules and plaques with overlying scale on his trunk, as well as the dorsal aspects of bilateral hands and the plantar surfaces of bilateral feet. His skin is otherwise unremarkable.
Expert shares tips for TNF-alpha inhibitor use in special populations
LAS VEGAS – Francisco A. Kerdel, BSc, MBBS, said at the Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar.
Dr. Kerdel, professor and vice chair of the department of dermatology at Florida International University, Miami, noted that, while tumor necrosis factor (TNF)–alpha inhibitors are category B drugs, inadequate data exist regarding lactation and exposure throughout pregnancy. “Rates of malformations and spontaneous abortions with therapy are similar to those in the general population, higher concentrations of infliximab and adalimumab have been found in infant and cord blood, compared with certolizumab pegol,” an anti-TNF biologic, he said.
In a prospective, postmarketing, multicenter pharmacokinetic study, researchers found a lack of placental transfer of certolizumab pegol during pregnancy (Ann Rheum Dis. 2018;77:228-33). Specifically, certolizumab levels were below the lower limit of quantification (less than 0.032 mcg/mL) in 13 of 14 infant samples at birth and in all infant samples at weeks 4 and 8. Only one infant had a minimal certolizumab level at birth (infant/mother ratio of 0.0009). No antibodies were detected at any time point during the study. Safety data in mothers were in line with the known safety profile of certolizumab and pregnancy profile of these underlying diseases. Adverse events experienced by the infants did not show any patterns or clusters of events suggesting a specific safety signal in children.
In a separate postmarketing pharmacokinetic study, investigators evaluated the transfer of certolizumab into breast milk (Ann Rheum Dis. 2017;76:1890-6). They found that the average daily infant dose of certolizumab was minimal. Specifically, the highest concentration of certolizumab in breast milk (0.0758 mcg/mL) was less than 1% of the expected mean plasma trough concentration of a therapeutic dose.
How do TNF-alpha inhibitors fare in the pediatric population? In a retrospective study of 390 children with psoriasis treated at 20 centers in the United States, Canada, and Europe, researchers evaluated the safety of systemic agents (JAMA Dermatol. 2017;153[11]:1147-57). Most (69%) were prescribed methotrexate, followed by biologics, acitretin, cyclosporine, and fumaric acid. Drug discontinuation (because of adverse events), which is sometimes used as an efficacy parameter, occurred in 12% of those who were on methotrexate, compared with 3% of those on biologics, 67% of those on acitretin, and 68% of those on fumaric acid.
At the other end of the age spectrum, biologic therapy is generally effective and well tolerated in elderly patients. “Sometimes, they may be more effective than other traditional drugs,” Dr. Kerdel said. “We’re a little bit concerned about immunosenescence, which can increase the risk for severe infections and malignancies. And, 90% of elderly patients with psoriasis may have comorbidities that need to be taken into account when treating psoriasis.”
Other factors come into play when choosing the right anti-TNF agent, including weight. While clinical trials show efficacy across weight groups, infliximab has weight-based dosing, “which may make it a better choice,” Dr. Kerdel said. “Patients taking etanercept may need a biweekly dose.”
Treatment flexibility also comes into play. For example, stopping therapy because of an infection or surgery may be problematic in drugs with a long half-life. Then there’s the issue of patient preference. “Some people don’t want to be injected frequently,” he said. “Some people don’t want to be injected at all and may require a simpler dosing regimen.”
Optimizing anti-TNF-alpha treatment starts with recognizing that there is a loss of response over time, Dr. Kerdel said, “or there may not be a response at all.” Contributing factors may include immunogenicity, suboptimal dosing, and poor patient adherence. In order to optimize treatment, clinicians can try switching agents or combination therapy, and explore continuous versus intermittent dosing.
“We really don’t have good data on the best protocol for switching treatment after failure of an anti-TNF-alpha agent,” he added. In cases of primary and secondary treatment failure, there is no consensus or guidelines on which second-line agent to use, nor good data on which measures to use.
No evidence-based guidelines are available for screening and monitoring patients receiving biologic therapy for psoriasis, either. “Evidence is strongest [grade B] for tuberculosis screening in patients treated with biologic agents,” Dr. Kerdel said. “Among known hepatitis B virus carriers, consider monitoring liver function tests and viral load [grade C]. High-grade evidence is lacking to support other routine testing. Physicians should use clinical judgment when screening and monitoring patients.”
He concluded his presentation by noting that there are a number of biosimilar agents available or in the pipeline for infliximab, adalimumab, and etanercept. This raises a number of questions for current and future consideration. For one, “will biosimilars show the same long-term efficacy and safety as the innovator products?” he asked. “Real-world, postmarketing, and registry data are needed. Will biosimilar agents offer significant cost benefits? Will biosimilar labeling be adequately transparent? Will we find biomarkers to help us target biologic agents to specific patients and subtypes of psoriasis?”
Dr. Kerdel reported that he is a member of the speaker’s bureau for AbbVie, Amgen, Celgene, Janssen, Novartis, Lilly, Leo, Ortho, and Novartis. He has also received grant/research support from AbbVie, Amgen, AstraZeneca, Celgene, Janssen, Leo, Lilly, Menlo Therapeutics, Novartis, Pfizer, and XBiotech.
SDEF and this news organization are owned by the same parent company.
LAS VEGAS – Francisco A. Kerdel, BSc, MBBS, said at the Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar.
Dr. Kerdel, professor and vice chair of the department of dermatology at Florida International University, Miami, noted that, while tumor necrosis factor (TNF)–alpha inhibitors are category B drugs, inadequate data exist regarding lactation and exposure throughout pregnancy. “Rates of malformations and spontaneous abortions with therapy are similar to those in the general population, higher concentrations of infliximab and adalimumab have been found in infant and cord blood, compared with certolizumab pegol,” an anti-TNF biologic, he said.
In a prospective, postmarketing, multicenter pharmacokinetic study, researchers found a lack of placental transfer of certolizumab pegol during pregnancy (Ann Rheum Dis. 2018;77:228-33). Specifically, certolizumab levels were below the lower limit of quantification (less than 0.032 mcg/mL) in 13 of 14 infant samples at birth and in all infant samples at weeks 4 and 8. Only one infant had a minimal certolizumab level at birth (infant/mother ratio of 0.0009). No antibodies were detected at any time point during the study. Safety data in mothers were in line with the known safety profile of certolizumab and pregnancy profile of these underlying diseases. Adverse events experienced by the infants did not show any patterns or clusters of events suggesting a specific safety signal in children.
In a separate postmarketing pharmacokinetic study, investigators evaluated the transfer of certolizumab into breast milk (Ann Rheum Dis. 2017;76:1890-6). They found that the average daily infant dose of certolizumab was minimal. Specifically, the highest concentration of certolizumab in breast milk (0.0758 mcg/mL) was less than 1% of the expected mean plasma trough concentration of a therapeutic dose.
How do TNF-alpha inhibitors fare in the pediatric population? In a retrospective study of 390 children with psoriasis treated at 20 centers in the United States, Canada, and Europe, researchers evaluated the safety of systemic agents (JAMA Dermatol. 2017;153[11]:1147-57). Most (69%) were prescribed methotrexate, followed by biologics, acitretin, cyclosporine, and fumaric acid. Drug discontinuation (because of adverse events), which is sometimes used as an efficacy parameter, occurred in 12% of those who were on methotrexate, compared with 3% of those on biologics, 67% of those on acitretin, and 68% of those on fumaric acid.
At the other end of the age spectrum, biologic therapy is generally effective and well tolerated in elderly patients. “Sometimes, they may be more effective than other traditional drugs,” Dr. Kerdel said. “We’re a little bit concerned about immunosenescence, which can increase the risk for severe infections and malignancies. And, 90% of elderly patients with psoriasis may have comorbidities that need to be taken into account when treating psoriasis.”
Other factors come into play when choosing the right anti-TNF agent, including weight. While clinical trials show efficacy across weight groups, infliximab has weight-based dosing, “which may make it a better choice,” Dr. Kerdel said. “Patients taking etanercept may need a biweekly dose.”
Treatment flexibility also comes into play. For example, stopping therapy because of an infection or surgery may be problematic in drugs with a long half-life. Then there’s the issue of patient preference. “Some people don’t want to be injected frequently,” he said. “Some people don’t want to be injected at all and may require a simpler dosing regimen.”
Optimizing anti-TNF-alpha treatment starts with recognizing that there is a loss of response over time, Dr. Kerdel said, “or there may not be a response at all.” Contributing factors may include immunogenicity, suboptimal dosing, and poor patient adherence. In order to optimize treatment, clinicians can try switching agents or combination therapy, and explore continuous versus intermittent dosing.
“We really don’t have good data on the best protocol for switching treatment after failure of an anti-TNF-alpha agent,” he added. In cases of primary and secondary treatment failure, there is no consensus or guidelines on which second-line agent to use, nor good data on which measures to use.
No evidence-based guidelines are available for screening and monitoring patients receiving biologic therapy for psoriasis, either. “Evidence is strongest [grade B] for tuberculosis screening in patients treated with biologic agents,” Dr. Kerdel said. “Among known hepatitis B virus carriers, consider monitoring liver function tests and viral load [grade C]. High-grade evidence is lacking to support other routine testing. Physicians should use clinical judgment when screening and monitoring patients.”
He concluded his presentation by noting that there are a number of biosimilar agents available or in the pipeline for infliximab, adalimumab, and etanercept. This raises a number of questions for current and future consideration. For one, “will biosimilars show the same long-term efficacy and safety as the innovator products?” he asked. “Real-world, postmarketing, and registry data are needed. Will biosimilar agents offer significant cost benefits? Will biosimilar labeling be adequately transparent? Will we find biomarkers to help us target biologic agents to specific patients and subtypes of psoriasis?”
Dr. Kerdel reported that he is a member of the speaker’s bureau for AbbVie, Amgen, Celgene, Janssen, Novartis, Lilly, Leo, Ortho, and Novartis. He has also received grant/research support from AbbVie, Amgen, AstraZeneca, Celgene, Janssen, Leo, Lilly, Menlo Therapeutics, Novartis, Pfizer, and XBiotech.
SDEF and this news organization are owned by the same parent company.
LAS VEGAS – Francisco A. Kerdel, BSc, MBBS, said at the Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar.
Dr. Kerdel, professor and vice chair of the department of dermatology at Florida International University, Miami, noted that, while tumor necrosis factor (TNF)–alpha inhibitors are category B drugs, inadequate data exist regarding lactation and exposure throughout pregnancy. “Rates of malformations and spontaneous abortions with therapy are similar to those in the general population, higher concentrations of infliximab and adalimumab have been found in infant and cord blood, compared with certolizumab pegol,” an anti-TNF biologic, he said.
In a prospective, postmarketing, multicenter pharmacokinetic study, researchers found a lack of placental transfer of certolizumab pegol during pregnancy (Ann Rheum Dis. 2018;77:228-33). Specifically, certolizumab levels were below the lower limit of quantification (less than 0.032 mcg/mL) in 13 of 14 infant samples at birth and in all infant samples at weeks 4 and 8. Only one infant had a minimal certolizumab level at birth (infant/mother ratio of 0.0009). No antibodies were detected at any time point during the study. Safety data in mothers were in line with the known safety profile of certolizumab and pregnancy profile of these underlying diseases. Adverse events experienced by the infants did not show any patterns or clusters of events suggesting a specific safety signal in children.
In a separate postmarketing pharmacokinetic study, investigators evaluated the transfer of certolizumab into breast milk (Ann Rheum Dis. 2017;76:1890-6). They found that the average daily infant dose of certolizumab was minimal. Specifically, the highest concentration of certolizumab in breast milk (0.0758 mcg/mL) was less than 1% of the expected mean plasma trough concentration of a therapeutic dose.
How do TNF-alpha inhibitors fare in the pediatric population? In a retrospective study of 390 children with psoriasis treated at 20 centers in the United States, Canada, and Europe, researchers evaluated the safety of systemic agents (JAMA Dermatol. 2017;153[11]:1147-57). Most (69%) were prescribed methotrexate, followed by biologics, acitretin, cyclosporine, and fumaric acid. Drug discontinuation (because of adverse events), which is sometimes used as an efficacy parameter, occurred in 12% of those who were on methotrexate, compared with 3% of those on biologics, 67% of those on acitretin, and 68% of those on fumaric acid.
At the other end of the age spectrum, biologic therapy is generally effective and well tolerated in elderly patients. “Sometimes, they may be more effective than other traditional drugs,” Dr. Kerdel said. “We’re a little bit concerned about immunosenescence, which can increase the risk for severe infections and malignancies. And, 90% of elderly patients with psoriasis may have comorbidities that need to be taken into account when treating psoriasis.”
Other factors come into play when choosing the right anti-TNF agent, including weight. While clinical trials show efficacy across weight groups, infliximab has weight-based dosing, “which may make it a better choice,” Dr. Kerdel said. “Patients taking etanercept may need a biweekly dose.”
Treatment flexibility also comes into play. For example, stopping therapy because of an infection or surgery may be problematic in drugs with a long half-life. Then there’s the issue of patient preference. “Some people don’t want to be injected frequently,” he said. “Some people don’t want to be injected at all and may require a simpler dosing regimen.”
Optimizing anti-TNF-alpha treatment starts with recognizing that there is a loss of response over time, Dr. Kerdel said, “or there may not be a response at all.” Contributing factors may include immunogenicity, suboptimal dosing, and poor patient adherence. In order to optimize treatment, clinicians can try switching agents or combination therapy, and explore continuous versus intermittent dosing.
“We really don’t have good data on the best protocol for switching treatment after failure of an anti-TNF-alpha agent,” he added. In cases of primary and secondary treatment failure, there is no consensus or guidelines on which second-line agent to use, nor good data on which measures to use.
No evidence-based guidelines are available for screening and monitoring patients receiving biologic therapy for psoriasis, either. “Evidence is strongest [grade B] for tuberculosis screening in patients treated with biologic agents,” Dr. Kerdel said. “Among known hepatitis B virus carriers, consider monitoring liver function tests and viral load [grade C]. High-grade evidence is lacking to support other routine testing. Physicians should use clinical judgment when screening and monitoring patients.”
He concluded his presentation by noting that there are a number of biosimilar agents available or in the pipeline for infliximab, adalimumab, and etanercept. This raises a number of questions for current and future consideration. For one, “will biosimilars show the same long-term efficacy and safety as the innovator products?” he asked. “Real-world, postmarketing, and registry data are needed. Will biosimilar agents offer significant cost benefits? Will biosimilar labeling be adequately transparent? Will we find biomarkers to help us target biologic agents to specific patients and subtypes of psoriasis?”
Dr. Kerdel reported that he is a member of the speaker’s bureau for AbbVie, Amgen, Celgene, Janssen, Novartis, Lilly, Leo, Ortho, and Novartis. He has also received grant/research support from AbbVie, Amgen, AstraZeneca, Celgene, Janssen, Leo, Lilly, Menlo Therapeutics, Novartis, Pfizer, and XBiotech.
SDEF and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM THE SDEF LAS VEGAS DERMATOLOGY SEMINAR