User login
Rare Lymphoma Reports Continue in Young Patients on TNF Blockers
Cases of a rare, aggressive, and usually fatal lymphoma continue to be reported in people being treated with tumor necrosis factor blockers, azathioprine, and/or mercaptopurine, the Food and Drug Administration announced in an April 14 statement.
The reports of the lymphoma, hepatosplenic T-cell lymphoma (HSTCL), have primarily involved adolescents and young adults being treated with these agents for Crohn’s disease or ulcerative colitis. One patient, however, was being treated for psoriasis, and two others for rheumatoid arthritis.
Most patients were on a combination of treatments that are known to suppress the immune system, but there have been cases in patients taking azathioprine or mercaptopurine alone, the statement said.
"The risks and benefits of using TNF blockers, azathioprine, and/or mercaptopurine should be carefully weighed when prescribing these drugs to children and young adults, especially for the treatment of Crohn’s disease and ulcerative colitis," according to the FDA.
The statement recommends that health care professionals monitor patients on these treatments for malignancies and educate patients and their caregivers about the signs and symptoms of HSTCL, which can include splenomegaly, hepatomegaly, abdominal pain, persistent fever, night sweats, and weight loss.
The statement also notes that people with rheumatoid arthritis, Crohn’s, ankylosing spondylitis, psoriatic arthritis, and plaque psoriasis "may be more likely to develop lymphoma," compared with the general U.S. population, making it difficult to estimate the increased risk of malignancies associated with TNF blockers, azathioprine and/or mercaptopurine.
As of Dec. 31, 2010, the FDA’s Adverse Event Reporting System (AERS), the medical literature, and the Cancer Survivors Network had received the following unduplicated reports of HSTCL:
20 cases in patients taking infliximab (Remicade), including 18 patients also taking mercaptopurine or azathioprine.
• 1 case in a patient taking etanercept (Enbrel).
• 2 cases in patients taking adalimumab (Humira).
• 5 cases in patients taking a combination of infliximab and adalimumab (including 4 patients also taking mercaptopurine or azathioprine).
• 12 cases in patients taking azathioprine.
• 3 cases in patients taking mercaptopurine.
No cases have been reported in the TNF blockers certolizumab pegol (Cimzia) and golimumab (Simponi).
Reports of serious adverse events associated with these and other drugs should be reported online to the FDA’s MedWatch program or by phone to 800-332-1088.
Cases of a rare, aggressive, and usually fatal lymphoma continue to be reported in people being treated with tumor necrosis factor blockers, azathioprine, and/or mercaptopurine, the Food and Drug Administration announced in an April 14 statement.
The reports of the lymphoma, hepatosplenic T-cell lymphoma (HSTCL), have primarily involved adolescents and young adults being treated with these agents for Crohn’s disease or ulcerative colitis. One patient, however, was being treated for psoriasis, and two others for rheumatoid arthritis.
Most patients were on a combination of treatments that are known to suppress the immune system, but there have been cases in patients taking azathioprine or mercaptopurine alone, the statement said.
"The risks and benefits of using TNF blockers, azathioprine, and/or mercaptopurine should be carefully weighed when prescribing these drugs to children and young adults, especially for the treatment of Crohn’s disease and ulcerative colitis," according to the FDA.
The statement recommends that health care professionals monitor patients on these treatments for malignancies and educate patients and their caregivers about the signs and symptoms of HSTCL, which can include splenomegaly, hepatomegaly, abdominal pain, persistent fever, night sweats, and weight loss.
The statement also notes that people with rheumatoid arthritis, Crohn’s, ankylosing spondylitis, psoriatic arthritis, and plaque psoriasis "may be more likely to develop lymphoma," compared with the general U.S. population, making it difficult to estimate the increased risk of malignancies associated with TNF blockers, azathioprine and/or mercaptopurine.
As of Dec. 31, 2010, the FDA’s Adverse Event Reporting System (AERS), the medical literature, and the Cancer Survivors Network had received the following unduplicated reports of HSTCL:
20 cases in patients taking infliximab (Remicade), including 18 patients also taking mercaptopurine or azathioprine.
• 1 case in a patient taking etanercept (Enbrel).
• 2 cases in patients taking adalimumab (Humira).
• 5 cases in patients taking a combination of infliximab and adalimumab (including 4 patients also taking mercaptopurine or azathioprine).
• 12 cases in patients taking azathioprine.
• 3 cases in patients taking mercaptopurine.
No cases have been reported in the TNF blockers certolizumab pegol (Cimzia) and golimumab (Simponi).
Reports of serious adverse events associated with these and other drugs should be reported online to the FDA’s MedWatch program or by phone to 800-332-1088.
Cases of a rare, aggressive, and usually fatal lymphoma continue to be reported in people being treated with tumor necrosis factor blockers, azathioprine, and/or mercaptopurine, the Food and Drug Administration announced in an April 14 statement.
The reports of the lymphoma, hepatosplenic T-cell lymphoma (HSTCL), have primarily involved adolescents and young adults being treated with these agents for Crohn’s disease or ulcerative colitis. One patient, however, was being treated for psoriasis, and two others for rheumatoid arthritis.
Most patients were on a combination of treatments that are known to suppress the immune system, but there have been cases in patients taking azathioprine or mercaptopurine alone, the statement said.
"The risks and benefits of using TNF blockers, azathioprine, and/or mercaptopurine should be carefully weighed when prescribing these drugs to children and young adults, especially for the treatment of Crohn’s disease and ulcerative colitis," according to the FDA.
The statement recommends that health care professionals monitor patients on these treatments for malignancies and educate patients and their caregivers about the signs and symptoms of HSTCL, which can include splenomegaly, hepatomegaly, abdominal pain, persistent fever, night sweats, and weight loss.
The statement also notes that people with rheumatoid arthritis, Crohn’s, ankylosing spondylitis, psoriatic arthritis, and plaque psoriasis "may be more likely to develop lymphoma," compared with the general U.S. population, making it difficult to estimate the increased risk of malignancies associated with TNF blockers, azathioprine and/or mercaptopurine.
As of Dec. 31, 2010, the FDA’s Adverse Event Reporting System (AERS), the medical literature, and the Cancer Survivors Network had received the following unduplicated reports of HSTCL:
20 cases in patients taking infliximab (Remicade), including 18 patients also taking mercaptopurine or azathioprine.
• 1 case in a patient taking etanercept (Enbrel).
• 2 cases in patients taking adalimumab (Humira).
• 5 cases in patients taking a combination of infliximab and adalimumab (including 4 patients also taking mercaptopurine or azathioprine).
• 12 cases in patients taking azathioprine.
• 3 cases in patients taking mercaptopurine.
No cases have been reported in the TNF blockers certolizumab pegol (Cimzia) and golimumab (Simponi).
Reports of serious adverse events associated with these and other drugs should be reported online to the FDA’s MedWatch program or by phone to 800-332-1088.
Treating Psoriasis During Pregnancy Presents Unique Challenges
WAILEA, HAWAII – Moderate to severe psoriasis is an independent risk factor for a variety of adverse pregnancy outcomes, according to Dr. Jennifer C. Cather.
Yet much remains unknown about the impact of psoriasis and its treatment in pregnancy. For this reason, every psoriasis patient who becomes pregnant while on a biologic agent should be strongly encouraged to enter one of the pregnancy registries, Dr. Cather said at the Hawaii Dermatology Seminar sponsored by Skin Disease Education Foundation (SDEF).
She recommended the Organization of Teratology Information Specialists (OTIS), because the group helps inform concerned patients. OTIS operates pregnancy registries for women taking etanercept (Enbrel) or adalimumab (Humira). In addition, pharmaceutical companies that market biologics maintain pregnancy registries for their agents.
The Food and Drug Administration rates methotrexate and acitretin as pregnancy category X drugs and cyclosporine as a category C drug. The anti–tumor necrosis factor agents are category B – meaning animal studies have shown no fetal risk – as are alefacept (Amevive) and ustekinumab (Stelara).
"That doesn’t mean I think you should give a category B agent to people who are pregnant or are planning pregnancy, because I think that the best drug in pregnancy is probably no drug, or light treatment if you can get away with that," said Dr. Cather, who is in private practice in Dallas.
Still, there is sometimes no satisfactory alternative to using a biologic agent in pregnancy. Two of the toughest challenges Dr. Cather said she encounters in her clinical practice arise in such situations. One involves the psoriasis patient who calls and says that her ob.gyn. told her to come off the biologic therapy immediately.
The other challenge is the patient whose psoriasis worsens during pregnancy to the extent that she needs to start on a biologic agent or switch to another one. This is not an uncommon situation, Dr. Cather noted, citing a University of California, Irvine, study which found that while psoriasis improved during pregnancy in 55% of pregnant patients, it worsened in 23%. Postpartum, psoriasis worsened in 65% (Arch. Dermatol. 2005;141:601-6).
A recent retrospective matched cohort study of pregnancy outcomes in psoriasis patients involved 68 deliveries in 35 women with moderate to severe psoriasis and 237 deliveries in 236 controls in Israel.
The incidence of pregnancy-induced hypertensive diseases was 7.4% in the psoriasis patients, significantly greater than the 2.1% rate in controls. Premature rupture of membranes occurred in 16% of psoriasis patients, compared with 5.5% of controls. The 24% incidence of large-for-gestational-age newborns among the psoriasis patients’ babies was twice that of controls. Macrosomia occurred in 13% of the babies of women with psoriasis, compared with 4.2% of matched controls.
In this multivariate analysis, moderate to severe psoriasis was also an independent risk factor for previous spontaneous and induced abortions (J. Eur. Acad. Dermatol. Venereol. 2010 Nov. 25 doi: 10.1111/j.1468-3083.2010.03917.x]).
Dr. Cather’s biologics of choice for psoriasis and psoriatic arthritis, whether during pregnancy or not, are TNF-antagonists. She has been monitoring her own psoriasis patients who have been on biologic agents during pregnancy and has not noted any increase in adverse outcomes. While she finds this somewhat reassuring, the definitive answers will come from the large pregnancy registries.
Nursing mothers on methotrexate, acitretin, or cyclosporine should not breast-feed. The risk of using anti–tumor necrosis factor agents during breast-feeding is "probably negligible," she said.
Psoriasis patients have a below-average rate of childbearing. Theories abound as to why. Possibilities include an increased infertility rate, voluntary childlessness due to concern about genetic transmission of psoriasis to the next generation, and the interference with sexual activity that has been documented in patients with active disease.
"I do have people who say they never want to have children because of their disease. That’s a very sad conversation," Dr. Cather said.
Intriguingly, there is some recent evidence to suggest anti-TNF therapy may improve the results of in vitro fertilization in women with infertility and recurrent spontaneous abortion. The therapeutic rationale is that TNF-alpha has been shown to have antireproductive effects.
In a nonrandomized study involving 75 women without psoriasis under age 38 with Th1/Th2 cytokine elevation undergoing IVF for infertility, investigators found that implantation, clinical pregnancy, and live birth rates were significantly higher in those on adalimumab and IVIG than in patients on neither (Am. J. Repro. Immunol. 2009;61:113-20). This is a controversial study among assisted reproduction specialists, Dr. Cather noted.
She disclosed that she serves as a consultant to Amgen (manufacturer of Enbrel), Abbott (manufacturer of Humira), and Centocor (manufacturer of Stelara) and has received research grants from Amgen, Celgene, and Pfizer.
SDEF and this news organization are owned by Elsevier.
WAILEA, HAWAII – Moderate to severe psoriasis is an independent risk factor for a variety of adverse pregnancy outcomes, according to Dr. Jennifer C. Cather.
Yet much remains unknown about the impact of psoriasis and its treatment in pregnancy. For this reason, every psoriasis patient who becomes pregnant while on a biologic agent should be strongly encouraged to enter one of the pregnancy registries, Dr. Cather said at the Hawaii Dermatology Seminar sponsored by Skin Disease Education Foundation (SDEF).
She recommended the Organization of Teratology Information Specialists (OTIS), because the group helps inform concerned patients. OTIS operates pregnancy registries for women taking etanercept (Enbrel) or adalimumab (Humira). In addition, pharmaceutical companies that market biologics maintain pregnancy registries for their agents.
The Food and Drug Administration rates methotrexate and acitretin as pregnancy category X drugs and cyclosporine as a category C drug. The anti–tumor necrosis factor agents are category B – meaning animal studies have shown no fetal risk – as are alefacept (Amevive) and ustekinumab (Stelara).
"That doesn’t mean I think you should give a category B agent to people who are pregnant or are planning pregnancy, because I think that the best drug in pregnancy is probably no drug, or light treatment if you can get away with that," said Dr. Cather, who is in private practice in Dallas.
Still, there is sometimes no satisfactory alternative to using a biologic agent in pregnancy. Two of the toughest challenges Dr. Cather said she encounters in her clinical practice arise in such situations. One involves the psoriasis patient who calls and says that her ob.gyn. told her to come off the biologic therapy immediately.
The other challenge is the patient whose psoriasis worsens during pregnancy to the extent that she needs to start on a biologic agent or switch to another one. This is not an uncommon situation, Dr. Cather noted, citing a University of California, Irvine, study which found that while psoriasis improved during pregnancy in 55% of pregnant patients, it worsened in 23%. Postpartum, psoriasis worsened in 65% (Arch. Dermatol. 2005;141:601-6).
A recent retrospective matched cohort study of pregnancy outcomes in psoriasis patients involved 68 deliveries in 35 women with moderate to severe psoriasis and 237 deliveries in 236 controls in Israel.
The incidence of pregnancy-induced hypertensive diseases was 7.4% in the psoriasis patients, significantly greater than the 2.1% rate in controls. Premature rupture of membranes occurred in 16% of psoriasis patients, compared with 5.5% of controls. The 24% incidence of large-for-gestational-age newborns among the psoriasis patients’ babies was twice that of controls. Macrosomia occurred in 13% of the babies of women with psoriasis, compared with 4.2% of matched controls.
In this multivariate analysis, moderate to severe psoriasis was also an independent risk factor for previous spontaneous and induced abortions (J. Eur. Acad. Dermatol. Venereol. 2010 Nov. 25 doi: 10.1111/j.1468-3083.2010.03917.x]).
Dr. Cather’s biologics of choice for psoriasis and psoriatic arthritis, whether during pregnancy or not, are TNF-antagonists. She has been monitoring her own psoriasis patients who have been on biologic agents during pregnancy and has not noted any increase in adverse outcomes. While she finds this somewhat reassuring, the definitive answers will come from the large pregnancy registries.
Nursing mothers on methotrexate, acitretin, or cyclosporine should not breast-feed. The risk of using anti–tumor necrosis factor agents during breast-feeding is "probably negligible," she said.
Psoriasis patients have a below-average rate of childbearing. Theories abound as to why. Possibilities include an increased infertility rate, voluntary childlessness due to concern about genetic transmission of psoriasis to the next generation, and the interference with sexual activity that has been documented in patients with active disease.
"I do have people who say they never want to have children because of their disease. That’s a very sad conversation," Dr. Cather said.
Intriguingly, there is some recent evidence to suggest anti-TNF therapy may improve the results of in vitro fertilization in women with infertility and recurrent spontaneous abortion. The therapeutic rationale is that TNF-alpha has been shown to have antireproductive effects.
In a nonrandomized study involving 75 women without psoriasis under age 38 with Th1/Th2 cytokine elevation undergoing IVF for infertility, investigators found that implantation, clinical pregnancy, and live birth rates were significantly higher in those on adalimumab and IVIG than in patients on neither (Am. J. Repro. Immunol. 2009;61:113-20). This is a controversial study among assisted reproduction specialists, Dr. Cather noted.
She disclosed that she serves as a consultant to Amgen (manufacturer of Enbrel), Abbott (manufacturer of Humira), and Centocor (manufacturer of Stelara) and has received research grants from Amgen, Celgene, and Pfizer.
SDEF and this news organization are owned by Elsevier.
WAILEA, HAWAII – Moderate to severe psoriasis is an independent risk factor for a variety of adverse pregnancy outcomes, according to Dr. Jennifer C. Cather.
Yet much remains unknown about the impact of psoriasis and its treatment in pregnancy. For this reason, every psoriasis patient who becomes pregnant while on a biologic agent should be strongly encouraged to enter one of the pregnancy registries, Dr. Cather said at the Hawaii Dermatology Seminar sponsored by Skin Disease Education Foundation (SDEF).
She recommended the Organization of Teratology Information Specialists (OTIS), because the group helps inform concerned patients. OTIS operates pregnancy registries for women taking etanercept (Enbrel) or adalimumab (Humira). In addition, pharmaceutical companies that market biologics maintain pregnancy registries for their agents.
The Food and Drug Administration rates methotrexate and acitretin as pregnancy category X drugs and cyclosporine as a category C drug. The anti–tumor necrosis factor agents are category B – meaning animal studies have shown no fetal risk – as are alefacept (Amevive) and ustekinumab (Stelara).
"That doesn’t mean I think you should give a category B agent to people who are pregnant or are planning pregnancy, because I think that the best drug in pregnancy is probably no drug, or light treatment if you can get away with that," said Dr. Cather, who is in private practice in Dallas.
Still, there is sometimes no satisfactory alternative to using a biologic agent in pregnancy. Two of the toughest challenges Dr. Cather said she encounters in her clinical practice arise in such situations. One involves the psoriasis patient who calls and says that her ob.gyn. told her to come off the biologic therapy immediately.
The other challenge is the patient whose psoriasis worsens during pregnancy to the extent that she needs to start on a biologic agent or switch to another one. This is not an uncommon situation, Dr. Cather noted, citing a University of California, Irvine, study which found that while psoriasis improved during pregnancy in 55% of pregnant patients, it worsened in 23%. Postpartum, psoriasis worsened in 65% (Arch. Dermatol. 2005;141:601-6).
A recent retrospective matched cohort study of pregnancy outcomes in psoriasis patients involved 68 deliveries in 35 women with moderate to severe psoriasis and 237 deliveries in 236 controls in Israel.
The incidence of pregnancy-induced hypertensive diseases was 7.4% in the psoriasis patients, significantly greater than the 2.1% rate in controls. Premature rupture of membranes occurred in 16% of psoriasis patients, compared with 5.5% of controls. The 24% incidence of large-for-gestational-age newborns among the psoriasis patients’ babies was twice that of controls. Macrosomia occurred in 13% of the babies of women with psoriasis, compared with 4.2% of matched controls.
In this multivariate analysis, moderate to severe psoriasis was also an independent risk factor for previous spontaneous and induced abortions (J. Eur. Acad. Dermatol. Venereol. 2010 Nov. 25 doi: 10.1111/j.1468-3083.2010.03917.x]).
Dr. Cather’s biologics of choice for psoriasis and psoriatic arthritis, whether during pregnancy or not, are TNF-antagonists. She has been monitoring her own psoriasis patients who have been on biologic agents during pregnancy and has not noted any increase in adverse outcomes. While she finds this somewhat reassuring, the definitive answers will come from the large pregnancy registries.
Nursing mothers on methotrexate, acitretin, or cyclosporine should not breast-feed. The risk of using anti–tumor necrosis factor agents during breast-feeding is "probably negligible," she said.
Psoriasis patients have a below-average rate of childbearing. Theories abound as to why. Possibilities include an increased infertility rate, voluntary childlessness due to concern about genetic transmission of psoriasis to the next generation, and the interference with sexual activity that has been documented in patients with active disease.
"I do have people who say they never want to have children because of their disease. That’s a very sad conversation," Dr. Cather said.
Intriguingly, there is some recent evidence to suggest anti-TNF therapy may improve the results of in vitro fertilization in women with infertility and recurrent spontaneous abortion. The therapeutic rationale is that TNF-alpha has been shown to have antireproductive effects.
In a nonrandomized study involving 75 women without psoriasis under age 38 with Th1/Th2 cytokine elevation undergoing IVF for infertility, investigators found that implantation, clinical pregnancy, and live birth rates were significantly higher in those on adalimumab and IVIG than in patients on neither (Am. J. Repro. Immunol. 2009;61:113-20). This is a controversial study among assisted reproduction specialists, Dr. Cather noted.
She disclosed that she serves as a consultant to Amgen (manufacturer of Enbrel), Abbott (manufacturer of Humira), and Centocor (manufacturer of Stelara) and has received research grants from Amgen, Celgene, and Pfizer.
SDEF and this news organization are owned by Elsevier.
EXPERT ANALYSIS FROM THE SDEF HAWAII DERMATOLOGY SEMINAR
Platelet C4d Predicts Cardiovascular Events, Mortality in Lupus
NEW YORK – The 18%-20% of systemic lupus erythematosus patients who test positive for platelet C4d face a much greater likelihood of having a future cardiovascular event, stroke, or death than those who are not platelet C4d positive, according to Dr. Susan M. Manzi, who presented the results at a rheumatology meeting sponsored by New York University.
"The only factor that predicted mortality greater than being positive for P-C4d [platelet C4d] was having cancer," said Dr. Manzi, chair of the department of medicine and codirector of the Lupus Center of Excellence at West Penn Allegheny Health System, Pittsburgh.
In the study of 356 patients with systemic lupus erythematosus (SLE), which was carried out between July 2001 and August 2009, 70 tested positive for P-C4d (20%) and 286 tested negative. This confirms a previous study that found that P-C4d was present in 18% of SLE patients and that P-C4d was highly specific for SLE (Arthritis Rheum. 2006;54:670-4). "P-C4d carries a 95% specificity for SLE," said Dr. Manzi, who is also vice chair and professor of medicine at Temple University, Philadelphia.
Patients who were P-C4d positive had twice as many cardiovascular events (36 vs. 18, P = .001). The odds ratio of having a cardiovascular event was 2.09 for positive P-C4d (P = .02), which was much higher than for smoking (OR 1.36), SLE disease duration (0.99), or the presence of renal disease (1.42). The odds ratio for aPL antibodies was 2.30 (P = 0.004).
The results for stroke were even more striking. Thirteen patients with P-C4d had stroke, compared with three in those who tested negative for P-C4d (P = .002). There was nearly a fivefold increased risk of stroke in this group (OR 4.96, CI 1.75-14.06, P = .003). Almost three times as many patients had a pulmonary embolism in the P-C4d group, compared with controls (11 vs. 4, P = .007).
Fourteen SLE patients (4%) died during the follow-up. Of those, more P-C4d-positive patients died compared with those who tested negative for P-C4d (11% vs. 2%, P = .002). Causes of death included infection (4), cardiac arrest (2), congestive heart failure (1), cancer (2), hemorrhage (1), and unspecified (4). The risk of all-cause mortality was increased almost eightfold in those who were P-C4d positive (OR 7.92, CI 2.13-29.48, P = .002). Only cancer (OR 19.74) surpassed being P-C4d as a risk for all-cause mortality in multivariate regression analysis.
"Usually when we measure complement in the circulation, we measure the parent protein C4 in the plasma. When complement is activated, it breaks down into fragments and we see that one fragment, C4d, can covalently bind to cell surfaces. So, as we study how these fragments of complement activation bind to cells, we are exploring their potential use as disease biomarkers and prognostic indicators," Dr. Manzi said.
Dr. Manzi serves on the advisory boards of Bristol-Myers Squibb, Human Genome Sciences, and Cephalon. She receives funding for research from Amgen USA, Aspreva Pharmaceuticals, Bristol-Myers Squibb, Cephalon, Genentech, Human Genome Sciences, and Immunomedics.
NEW YORK – The 18%-20% of systemic lupus erythematosus patients who test positive for platelet C4d face a much greater likelihood of having a future cardiovascular event, stroke, or death than those who are not platelet C4d positive, according to Dr. Susan M. Manzi, who presented the results at a rheumatology meeting sponsored by New York University.
"The only factor that predicted mortality greater than being positive for P-C4d [platelet C4d] was having cancer," said Dr. Manzi, chair of the department of medicine and codirector of the Lupus Center of Excellence at West Penn Allegheny Health System, Pittsburgh.
In the study of 356 patients with systemic lupus erythematosus (SLE), which was carried out between July 2001 and August 2009, 70 tested positive for P-C4d (20%) and 286 tested negative. This confirms a previous study that found that P-C4d was present in 18% of SLE patients and that P-C4d was highly specific for SLE (Arthritis Rheum. 2006;54:670-4). "P-C4d carries a 95% specificity for SLE," said Dr. Manzi, who is also vice chair and professor of medicine at Temple University, Philadelphia.
Patients who were P-C4d positive had twice as many cardiovascular events (36 vs. 18, P = .001). The odds ratio of having a cardiovascular event was 2.09 for positive P-C4d (P = .02), which was much higher than for smoking (OR 1.36), SLE disease duration (0.99), or the presence of renal disease (1.42). The odds ratio for aPL antibodies was 2.30 (P = 0.004).
The results for stroke were even more striking. Thirteen patients with P-C4d had stroke, compared with three in those who tested negative for P-C4d (P = .002). There was nearly a fivefold increased risk of stroke in this group (OR 4.96, CI 1.75-14.06, P = .003). Almost three times as many patients had a pulmonary embolism in the P-C4d group, compared with controls (11 vs. 4, P = .007).
Fourteen SLE patients (4%) died during the follow-up. Of those, more P-C4d-positive patients died compared with those who tested negative for P-C4d (11% vs. 2%, P = .002). Causes of death included infection (4), cardiac arrest (2), congestive heart failure (1), cancer (2), hemorrhage (1), and unspecified (4). The risk of all-cause mortality was increased almost eightfold in those who were P-C4d positive (OR 7.92, CI 2.13-29.48, P = .002). Only cancer (OR 19.74) surpassed being P-C4d as a risk for all-cause mortality in multivariate regression analysis.
"Usually when we measure complement in the circulation, we measure the parent protein C4 in the plasma. When complement is activated, it breaks down into fragments and we see that one fragment, C4d, can covalently bind to cell surfaces. So, as we study how these fragments of complement activation bind to cells, we are exploring their potential use as disease biomarkers and prognostic indicators," Dr. Manzi said.
Dr. Manzi serves on the advisory boards of Bristol-Myers Squibb, Human Genome Sciences, and Cephalon. She receives funding for research from Amgen USA, Aspreva Pharmaceuticals, Bristol-Myers Squibb, Cephalon, Genentech, Human Genome Sciences, and Immunomedics.
NEW YORK – The 18%-20% of systemic lupus erythematosus patients who test positive for platelet C4d face a much greater likelihood of having a future cardiovascular event, stroke, or death than those who are not platelet C4d positive, according to Dr. Susan M. Manzi, who presented the results at a rheumatology meeting sponsored by New York University.
"The only factor that predicted mortality greater than being positive for P-C4d [platelet C4d] was having cancer," said Dr. Manzi, chair of the department of medicine and codirector of the Lupus Center of Excellence at West Penn Allegheny Health System, Pittsburgh.
In the study of 356 patients with systemic lupus erythematosus (SLE), which was carried out between July 2001 and August 2009, 70 tested positive for P-C4d (20%) and 286 tested negative. This confirms a previous study that found that P-C4d was present in 18% of SLE patients and that P-C4d was highly specific for SLE (Arthritis Rheum. 2006;54:670-4). "P-C4d carries a 95% specificity for SLE," said Dr. Manzi, who is also vice chair and professor of medicine at Temple University, Philadelphia.
Patients who were P-C4d positive had twice as many cardiovascular events (36 vs. 18, P = .001). The odds ratio of having a cardiovascular event was 2.09 for positive P-C4d (P = .02), which was much higher than for smoking (OR 1.36), SLE disease duration (0.99), or the presence of renal disease (1.42). The odds ratio for aPL antibodies was 2.30 (P = 0.004).
The results for stroke were even more striking. Thirteen patients with P-C4d had stroke, compared with three in those who tested negative for P-C4d (P = .002). There was nearly a fivefold increased risk of stroke in this group (OR 4.96, CI 1.75-14.06, P = .003). Almost three times as many patients had a pulmonary embolism in the P-C4d group, compared with controls (11 vs. 4, P = .007).
Fourteen SLE patients (4%) died during the follow-up. Of those, more P-C4d-positive patients died compared with those who tested negative for P-C4d (11% vs. 2%, P = .002). Causes of death included infection (4), cardiac arrest (2), congestive heart failure (1), cancer (2), hemorrhage (1), and unspecified (4). The risk of all-cause mortality was increased almost eightfold in those who were P-C4d positive (OR 7.92, CI 2.13-29.48, P = .002). Only cancer (OR 19.74) surpassed being P-C4d as a risk for all-cause mortality in multivariate regression analysis.
"Usually when we measure complement in the circulation, we measure the parent protein C4 in the plasma. When complement is activated, it breaks down into fragments and we see that one fragment, C4d, can covalently bind to cell surfaces. So, as we study how these fragments of complement activation bind to cells, we are exploring their potential use as disease biomarkers and prognostic indicators," Dr. Manzi said.
Dr. Manzi serves on the advisory boards of Bristol-Myers Squibb, Human Genome Sciences, and Cephalon. She receives funding for research from Amgen USA, Aspreva Pharmaceuticals, Bristol-Myers Squibb, Cephalon, Genentech, Human Genome Sciences, and Immunomedics.
EXPERT ANALYSIS FROM A RHEUMATOLOGY MEETING SPONSORED BY NEW YORK UNIVERSITY
Juvenile Dermatomyositis Is Easily Missed Diagnosis
NEW YORK – Because juvenile dermatomyositis is a rare disease and its symptoms often differ from those seen in adult dermatomyositis, physicians may not recognize it and diagnosis can be delayed, Dr. Brian Feldman said.
He described a recent practice survey that found variability in juvenile dermatomyositis (JDM) treatment and, in the absence of randomized controlled trials, urged clinicians to consult newly created consensus protocols developed by rheumatologists and to gather data that may help to optimize treatment and minimize side effects in the future for those with JDM.
Dr. Feldman described a 15-year-old patient who had progressively deteriorated over a 3-year period despite being seen by pediatricians and dermatologists. His initial symptoms included fatigue and elevated muscle enzymes, including aspartate transaminase (AST), alanine transaminase (ALT), lactate dehydrogenase (LDH), and creatine phosphokinase (CPK).
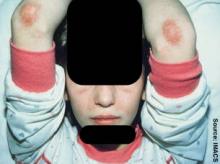
Within a year, the patient developed a purple skin rash on his hands, elbows, and knees. He later developed Raynaud's syndrome and muscle weakness with decreased range of motion that interfered with participation in sports and other activities of daily living.
Upon examination, he was quite thin with a scaly rash over his knuckles (Gottron's papules), healing skin ulcerations on his hands (from severe Raynaud's), periungual erythema, and lipodystrophy and erythema of his forearms. Magnetic resonance imaging showed acute inflammation of his shoulder girdle, said Dr. Feldman, chief of pediatric rheumatology at the Hospital for Sick Children in Toronto.
Dr. Feldman said that one of the best diagnostic clues for JDM comes from microscopic examination of nail folds. In this case, the patient had tortuous, bushy nail folds, dilated or missing capillaries (capillary density of 3 per mm of nailfold length while normal ranges from 7-11 per mm), and cuticular overgrowth indicative of JDM.
The different presentation seen with children compared with adults may hinder a correct diagnosis. For instance, calcinosis is seen much more frequently in children than adults. Children are less likely to have some of the systemic symptoms, such as fever, poor weight gain, and pulmonary effects, and they almost never have cardiac problems. Unlike adults with dermatomyositis or polymyositis who appear to have a fourfold increased risk of malignancy, very few cases of malignancy have been associated with JDM. Children are more likely to experience dysphonia/dysphagia but are as likely to have arthritic symptoms (around 58% of each group). Myositis specific antibodies do not seem to play as important a diagnostic role for children as adults.
Children who develop JDM appear to have a better prognosis than adults. Although adults with myositis have appreciable mortality (about 10%) and progressive disability, which may be related to treatment of a long-term condition such as avascular necrosis or osteoporosis with compression fracture, findings from Dr. Feldman's group show that outcomes were often excellent in children. "We have not had a single death from JDM in 30 years," he said.
About two-thirds of children with JDM have a polycyclic, chronic, unremitting disease course, but about one-third follow a monocyclic course and have disease symptoms that last for 2-3 years and then go away, with, or potentially even without, treatment.
In a very small proportion of these patients, symptoms may return many years later. "The message here is that when the disease goes away, it almost certainly is gone forever," Dr. Feldman noted.
It is possible to predict which patients will follow a chronic course and which patients will have early remission. In his experience, the risk of persistent disease falls by half if Gottron's rash disappears within 3 months of diagnosis (Arthritis Rheum. 2008;58:3585-92).
The results of a recent practice survey by the Childhood Arthritis and Rheumatology Research Alliance (CARRA) of almost 200 pediatric rheumatologists in North America found both similarities and differences in treatment approaches to JDM (J. Rheumatol. 2010;37:1953-61).
Corticosteroids and methotrexate (MTX) are mainstays of care, but the route and pattern of corticosteroid administration varied: 82% of respondents initially pulsed with high-dose intravenous methylprednisolone (IVMP) while 18% prescribed oral prednisone. MTX was used concurrently with steroids 84% of the time. Sixteen percent reported also using IV immunoglobulin, with or without MTX, for more severe disease, refractory disease, and prominent cutaneous disease. Hydroxychloroquine was given for milder cases, especially for rash, while cyclophosphamide was prescribed for ulcerative disease or for patients with pulmonary symptoms.
While these findings reflect the current prescribing practices, there are almost no randomized controlled trials of medications for JDM, Dr. Feldman pointed out.
This reflects in part the rare nature of the disease, its complexity, the difficulty of conducting studies in children, and the high cost of randomized controlled trials. There have been studies using advanced analytic techniques to provide strong comparative data in lieu of an randomized controlled trial. One such study by Dr. Feldman and associates (Arthritis Rheum. 2008;59:989-95) showed that more aggressive corticosteroid therapy does not give better 3-year outcomes.
A recent randomized controlled trial presented at the annual meeting of the American College of Rheumatology looked at dermatomyositis in 76 adults and 48 children and found that 80% had a response to rituximab within a year of treatment. Interpretation of the study may have been limited by the design, which allowed patients to receive add-on medications during the trial.
In light of the CARRA study's findings of heterogeneity in the treatment of JDM and the absence of randomized controlled trials, a group of 12 pediatric rheumatologists met to study treatments in JDM using a new approach: by developing consensus treatment protocols (Arthritis Care Res. 2010;62:219-25).
"This is similar to what has been done in pediatric oncology," said Dr. Feldman, who was one of the participants. "We are hoping physicians throughout the world will take these protocols off the shelf, and by using standardized doses, follow-up, and measurements, we will be able to accumulate enough evidence over time to know which is the best therapy."
In brief, the group recommended three protocols for the treatment of patients with moderately severe JDM: pulse IVMP plus MTX; IVMP, MTX, plus IVIG; or oral prednisone plus MTX. The third protocol is the one followed most often at the Hospital for Sick Children. The treatment protocols are not intended as treatment recommendations, although it is hoped that a physician will choose to follow the standardized protocol that most closely reflects his or her preferred practice. It is presented as a "first step to allow comparison of different approaches to the treatment of JDM," he said.
Dr. Feldman has done contracted research with Bayer Healthcare Pharmaceuticals. He referenced unlabeled/unapproved uses of drugs or products in his presentation.
NEW YORK – Because juvenile dermatomyositis is a rare disease and its symptoms often differ from those seen in adult dermatomyositis, physicians may not recognize it and diagnosis can be delayed, Dr. Brian Feldman said.
He described a recent practice survey that found variability in juvenile dermatomyositis (JDM) treatment and, in the absence of randomized controlled trials, urged clinicians to consult newly created consensus protocols developed by rheumatologists and to gather data that may help to optimize treatment and minimize side effects in the future for those with JDM.
Dr. Feldman described a 15-year-old patient who had progressively deteriorated over a 3-year period despite being seen by pediatricians and dermatologists. His initial symptoms included fatigue and elevated muscle enzymes, including aspartate transaminase (AST), alanine transaminase (ALT), lactate dehydrogenase (LDH), and creatine phosphokinase (CPK).
Within a year, the patient developed a purple skin rash on his hands, elbows, and knees. He later developed Raynaud's syndrome and muscle weakness with decreased range of motion that interfered with participation in sports and other activities of daily living.
Upon examination, he was quite thin with a scaly rash over his knuckles (Gottron's papules), healing skin ulcerations on his hands (from severe Raynaud's), periungual erythema, and lipodystrophy and erythema of his forearms. Magnetic resonance imaging showed acute inflammation of his shoulder girdle, said Dr. Feldman, chief of pediatric rheumatology at the Hospital for Sick Children in Toronto.
Dr. Feldman said that one of the best diagnostic clues for JDM comes from microscopic examination of nail folds. In this case, the patient had tortuous, bushy nail folds, dilated or missing capillaries (capillary density of 3 per mm of nailfold length while normal ranges from 7-11 per mm), and cuticular overgrowth indicative of JDM.
The different presentation seen with children compared with adults may hinder a correct diagnosis. For instance, calcinosis is seen much more frequently in children than adults. Children are less likely to have some of the systemic symptoms, such as fever, poor weight gain, and pulmonary effects, and they almost never have cardiac problems. Unlike adults with dermatomyositis or polymyositis who appear to have a fourfold increased risk of malignancy, very few cases of malignancy have been associated with JDM. Children are more likely to experience dysphonia/dysphagia but are as likely to have arthritic symptoms (around 58% of each group). Myositis specific antibodies do not seem to play as important a diagnostic role for children as adults.
Children who develop JDM appear to have a better prognosis than adults. Although adults with myositis have appreciable mortality (about 10%) and progressive disability, which may be related to treatment of a long-term condition such as avascular necrosis or osteoporosis with compression fracture, findings from Dr. Feldman's group show that outcomes were often excellent in children. "We have not had a single death from JDM in 30 years," he said.
About two-thirds of children with JDM have a polycyclic, chronic, unremitting disease course, but about one-third follow a monocyclic course and have disease symptoms that last for 2-3 years and then go away, with, or potentially even without, treatment.
In a very small proportion of these patients, symptoms may return many years later. "The message here is that when the disease goes away, it almost certainly is gone forever," Dr. Feldman noted.
It is possible to predict which patients will follow a chronic course and which patients will have early remission. In his experience, the risk of persistent disease falls by half if Gottron's rash disappears within 3 months of diagnosis (Arthritis Rheum. 2008;58:3585-92).
The results of a recent practice survey by the Childhood Arthritis and Rheumatology Research Alliance (CARRA) of almost 200 pediatric rheumatologists in North America found both similarities and differences in treatment approaches to JDM (J. Rheumatol. 2010;37:1953-61).
Corticosteroids and methotrexate (MTX) are mainstays of care, but the route and pattern of corticosteroid administration varied: 82% of respondents initially pulsed with high-dose intravenous methylprednisolone (IVMP) while 18% prescribed oral prednisone. MTX was used concurrently with steroids 84% of the time. Sixteen percent reported also using IV immunoglobulin, with or without MTX, for more severe disease, refractory disease, and prominent cutaneous disease. Hydroxychloroquine was given for milder cases, especially for rash, while cyclophosphamide was prescribed for ulcerative disease or for patients with pulmonary symptoms.
While these findings reflect the current prescribing practices, there are almost no randomized controlled trials of medications for JDM, Dr. Feldman pointed out.
This reflects in part the rare nature of the disease, its complexity, the difficulty of conducting studies in children, and the high cost of randomized controlled trials. There have been studies using advanced analytic techniques to provide strong comparative data in lieu of an randomized controlled trial. One such study by Dr. Feldman and associates (Arthritis Rheum. 2008;59:989-95) showed that more aggressive corticosteroid therapy does not give better 3-year outcomes.
A recent randomized controlled trial presented at the annual meeting of the American College of Rheumatology looked at dermatomyositis in 76 adults and 48 children and found that 80% had a response to rituximab within a year of treatment. Interpretation of the study may have been limited by the design, which allowed patients to receive add-on medications during the trial.
In light of the CARRA study's findings of heterogeneity in the treatment of JDM and the absence of randomized controlled trials, a group of 12 pediatric rheumatologists met to study treatments in JDM using a new approach: by developing consensus treatment protocols (Arthritis Care Res. 2010;62:219-25).
"This is similar to what has been done in pediatric oncology," said Dr. Feldman, who was one of the participants. "We are hoping physicians throughout the world will take these protocols off the shelf, and by using standardized doses, follow-up, and measurements, we will be able to accumulate enough evidence over time to know which is the best therapy."
In brief, the group recommended three protocols for the treatment of patients with moderately severe JDM: pulse IVMP plus MTX; IVMP, MTX, plus IVIG; or oral prednisone plus MTX. The third protocol is the one followed most often at the Hospital for Sick Children. The treatment protocols are not intended as treatment recommendations, although it is hoped that a physician will choose to follow the standardized protocol that most closely reflects his or her preferred practice. It is presented as a "first step to allow comparison of different approaches to the treatment of JDM," he said.
Dr. Feldman has done contracted research with Bayer Healthcare Pharmaceuticals. He referenced unlabeled/unapproved uses of drugs or products in his presentation.
NEW YORK – Because juvenile dermatomyositis is a rare disease and its symptoms often differ from those seen in adult dermatomyositis, physicians may not recognize it and diagnosis can be delayed, Dr. Brian Feldman said.
He described a recent practice survey that found variability in juvenile dermatomyositis (JDM) treatment and, in the absence of randomized controlled trials, urged clinicians to consult newly created consensus protocols developed by rheumatologists and to gather data that may help to optimize treatment and minimize side effects in the future for those with JDM.
Dr. Feldman described a 15-year-old patient who had progressively deteriorated over a 3-year period despite being seen by pediatricians and dermatologists. His initial symptoms included fatigue and elevated muscle enzymes, including aspartate transaminase (AST), alanine transaminase (ALT), lactate dehydrogenase (LDH), and creatine phosphokinase (CPK).
Within a year, the patient developed a purple skin rash on his hands, elbows, and knees. He later developed Raynaud's syndrome and muscle weakness with decreased range of motion that interfered with participation in sports and other activities of daily living.
Upon examination, he was quite thin with a scaly rash over his knuckles (Gottron's papules), healing skin ulcerations on his hands (from severe Raynaud's), periungual erythema, and lipodystrophy and erythema of his forearms. Magnetic resonance imaging showed acute inflammation of his shoulder girdle, said Dr. Feldman, chief of pediatric rheumatology at the Hospital for Sick Children in Toronto.
Dr. Feldman said that one of the best diagnostic clues for JDM comes from microscopic examination of nail folds. In this case, the patient had tortuous, bushy nail folds, dilated or missing capillaries (capillary density of 3 per mm of nailfold length while normal ranges from 7-11 per mm), and cuticular overgrowth indicative of JDM.
The different presentation seen with children compared with adults may hinder a correct diagnosis. For instance, calcinosis is seen much more frequently in children than adults. Children are less likely to have some of the systemic symptoms, such as fever, poor weight gain, and pulmonary effects, and they almost never have cardiac problems. Unlike adults with dermatomyositis or polymyositis who appear to have a fourfold increased risk of malignancy, very few cases of malignancy have been associated with JDM. Children are more likely to experience dysphonia/dysphagia but are as likely to have arthritic symptoms (around 58% of each group). Myositis specific antibodies do not seem to play as important a diagnostic role for children as adults.
Children who develop JDM appear to have a better prognosis than adults. Although adults with myositis have appreciable mortality (about 10%) and progressive disability, which may be related to treatment of a long-term condition such as avascular necrosis or osteoporosis with compression fracture, findings from Dr. Feldman's group show that outcomes were often excellent in children. "We have not had a single death from JDM in 30 years," he said.
About two-thirds of children with JDM have a polycyclic, chronic, unremitting disease course, but about one-third follow a monocyclic course and have disease symptoms that last for 2-3 years and then go away, with, or potentially even without, treatment.
In a very small proportion of these patients, symptoms may return many years later. "The message here is that when the disease goes away, it almost certainly is gone forever," Dr. Feldman noted.
It is possible to predict which patients will follow a chronic course and which patients will have early remission. In his experience, the risk of persistent disease falls by half if Gottron's rash disappears within 3 months of diagnosis (Arthritis Rheum. 2008;58:3585-92).
The results of a recent practice survey by the Childhood Arthritis and Rheumatology Research Alliance (CARRA) of almost 200 pediatric rheumatologists in North America found both similarities and differences in treatment approaches to JDM (J. Rheumatol. 2010;37:1953-61).
Corticosteroids and methotrexate (MTX) are mainstays of care, but the route and pattern of corticosteroid administration varied: 82% of respondents initially pulsed with high-dose intravenous methylprednisolone (IVMP) while 18% prescribed oral prednisone. MTX was used concurrently with steroids 84% of the time. Sixteen percent reported also using IV immunoglobulin, with or without MTX, for more severe disease, refractory disease, and prominent cutaneous disease. Hydroxychloroquine was given for milder cases, especially for rash, while cyclophosphamide was prescribed for ulcerative disease or for patients with pulmonary symptoms.
While these findings reflect the current prescribing practices, there are almost no randomized controlled trials of medications for JDM, Dr. Feldman pointed out.
This reflects in part the rare nature of the disease, its complexity, the difficulty of conducting studies in children, and the high cost of randomized controlled trials. There have been studies using advanced analytic techniques to provide strong comparative data in lieu of an randomized controlled trial. One such study by Dr. Feldman and associates (Arthritis Rheum. 2008;59:989-95) showed that more aggressive corticosteroid therapy does not give better 3-year outcomes.
A recent randomized controlled trial presented at the annual meeting of the American College of Rheumatology looked at dermatomyositis in 76 adults and 48 children and found that 80% had a response to rituximab within a year of treatment. Interpretation of the study may have been limited by the design, which allowed patients to receive add-on medications during the trial.
In light of the CARRA study's findings of heterogeneity in the treatment of JDM and the absence of randomized controlled trials, a group of 12 pediatric rheumatologists met to study treatments in JDM using a new approach: by developing consensus treatment protocols (Arthritis Care Res. 2010;62:219-25).
"This is similar to what has been done in pediatric oncology," said Dr. Feldman, who was one of the participants. "We are hoping physicians throughout the world will take these protocols off the shelf, and by using standardized doses, follow-up, and measurements, we will be able to accumulate enough evidence over time to know which is the best therapy."
In brief, the group recommended three protocols for the treatment of patients with moderately severe JDM: pulse IVMP plus MTX; IVMP, MTX, plus IVIG; or oral prednisone plus MTX. The third protocol is the one followed most often at the Hospital for Sick Children. The treatment protocols are not intended as treatment recommendations, although it is hoped that a physician will choose to follow the standardized protocol that most closely reflects his or her preferred practice. It is presented as a "first step to allow comparison of different approaches to the treatment of JDM," he said.
Dr. Feldman has done contracted research with Bayer Healthcare Pharmaceuticals. He referenced unlabeled/unapproved uses of drugs or products in his presentation.
EXPERT ANALYSIS FROM A MEETING SPONSORED BY NEW YORK UNIVERSITY
Interaction Between Adalimumab With Concurrent Pregabalin and Duloxetine Administration in a Psoriasis Patient With Diabetic Peripheral Neuropathy [letter]
Ask the Expert: Scleroderma Mimics
Systemic sclerosis and scleroderma are characterized by inflammation and fibrosis of the skin, as well as by vascular abnormalities, visceral damage, and the production of autoantibodies.
Diagnosing the chronic connective tissue disease, which can present in both limited and diffuse forms, can be complicated by the fact that it shares clinical and/or histologic features with a number of "scleroderma mimics," according to Dr. Janet E. Pope.
As with systemic sclerosis, most of these sclerodermalike conditions present with hard skin and tissue fibrosis and should be included in the differential diagnosis, noted Dr. Pope. She offered insight into ways to distinguish sclerodermalike fibrosing skin disorders from systemic sclerosis.
Skin & Allergy News: What are some of the more common "mimics" of systemic sclerosis.
Dr. Pope: Systemic sclerosis can be confused with such sclerodermalike skin disorders as eosinophilic fasciitis (EF), morphea, scleromyxedema, and nephrogenic systemic fibrosis (NSF).
SAN: How can physicians differentiate these from systemic sclerosis?
Dr. Pope: Most of these conditions lack Raynaud's phenomenon, sclerodactyly, and nailfold capillary changes.
Eosinophilic fasciitis (also called Shulman's syndrome) is a rare, localized, fibrosing disorder of subcutaneous tissues. Unlike systemic sclerosis, the overlying skin in EF is flexible enough to be pinched because the pathological disturbance is subcutaneous. This condition is often associated with venous grooving.
Generalized morphea is characterized by cutaneous sclerosis plaques caused by excessive collagen deposition, which leads to thickening of the dermis and/or subcutaneous tissues. Three or more major anatomical regions are affected by plaques in generalized morphea. Studies have shown that morphea often resolves within 2-5 years, but deeper lesions may take longer to resolve and may be accompanied by more disability and damage, depending on where they are located (J. Rheumatol. 1997;24:73-80). Additionally, many patients with morphea have antinuclear antibody and rheumatoid factor autoantibodies.
Scleromyxedema (also called papular mucinosis) is a chronic connective tissue disorder in which mucin deposits cause the skin to become red and raised, and make the movement of affected areas difficult. It tends to involve the head and neck rather than the extremities, and in most cases the sclerodermiform plaques and lichenoid papules are associated with a monoclonal gammopathy.
Finally, nephrogenic systemic fibrosis is a systemic fibrosing condition that occurs in people with kidney disease. NSF affects the skin, muscles, heart, and kidneys. It is strongly associated with exposure to gadolinium-based contrast agents.
Unlike systemic sclerosis, NSF tends to spare the skin of the face, and there is no association with autoantibodies or inflammatory cells.
The predominant histopathologic features of NSF include fibrocytelike cells, histiocytes, dermal dendritic cells, scarlike fibrosis, mucin, edema, calcification, and ossification. Diagnosing NSF requires both histopathologic and clinical findings.
SAN: Have any risk factors for the various conditions been identified?
Dr. Pope: As noted, NSF develops in patients with kidney disease and exposure to gadolinium. Renal failure is a major risk factor. With morphea, many patients who develop the condition are more likely than those without it to have an additional autoimmune disease.
SAN: How are the various conditions treated?
Dr. Pope: Corticosteroids and often methotrexate are used to treat EF, and early, aggressive treatment can minimize damage associated with the disease. Morphea is usually self-limited, and there is no proven treatment, although anecdotal evidence suggests that morphea patients who are being treated with antimalarials often develop new lesions when the treatment is stopped. IV immunoglobulin has been used in scleromyxedema treatment with mixed results.
Finally, there is no single therapeutic agent that has been proved effective for NSF. Anecdotal evidence suggests that extracorporeal photopheresis can lead to some lessening of skin thickening as well as increased mobility. Agents such as pentoxifylline, sodium, thiosulfate, and imatinib mesylate might improve skin fibrosis, but prevention through avoidance of gadolinium exposure in at-risk patients is advocated.
Dr. Pope is an associate professor of medicine in the division of rheumatology at the University of Western Ontario and a rheumatologist at St. Joseph's Health Care, both in London, Ont. She reported having no financial conflicts of interest.
Systemic sclerosis and scleroderma are characterized by inflammation and fibrosis of the skin, as well as by vascular abnormalities, visceral damage, and the production of autoantibodies.
Diagnosing the chronic connective tissue disease, which can present in both limited and diffuse forms, can be complicated by the fact that it shares clinical and/or histologic features with a number of "scleroderma mimics," according to Dr. Janet E. Pope.
As with systemic sclerosis, most of these sclerodermalike conditions present with hard skin and tissue fibrosis and should be included in the differential diagnosis, noted Dr. Pope. She offered insight into ways to distinguish sclerodermalike fibrosing skin disorders from systemic sclerosis.
Skin & Allergy News: What are some of the more common "mimics" of systemic sclerosis.
Dr. Pope: Systemic sclerosis can be confused with such sclerodermalike skin disorders as eosinophilic fasciitis (EF), morphea, scleromyxedema, and nephrogenic systemic fibrosis (NSF).
SAN: How can physicians differentiate these from systemic sclerosis?
Dr. Pope: Most of these conditions lack Raynaud's phenomenon, sclerodactyly, and nailfold capillary changes.
Eosinophilic fasciitis (also called Shulman's syndrome) is a rare, localized, fibrosing disorder of subcutaneous tissues. Unlike systemic sclerosis, the overlying skin in EF is flexible enough to be pinched because the pathological disturbance is subcutaneous. This condition is often associated with venous grooving.
Generalized morphea is characterized by cutaneous sclerosis plaques caused by excessive collagen deposition, which leads to thickening of the dermis and/or subcutaneous tissues. Three or more major anatomical regions are affected by plaques in generalized morphea. Studies have shown that morphea often resolves within 2-5 years, but deeper lesions may take longer to resolve and may be accompanied by more disability and damage, depending on where they are located (J. Rheumatol. 1997;24:73-80). Additionally, many patients with morphea have antinuclear antibody and rheumatoid factor autoantibodies.
Scleromyxedema (also called papular mucinosis) is a chronic connective tissue disorder in which mucin deposits cause the skin to become red and raised, and make the movement of affected areas difficult. It tends to involve the head and neck rather than the extremities, and in most cases the sclerodermiform plaques and lichenoid papules are associated with a monoclonal gammopathy.
Finally, nephrogenic systemic fibrosis is a systemic fibrosing condition that occurs in people with kidney disease. NSF affects the skin, muscles, heart, and kidneys. It is strongly associated with exposure to gadolinium-based contrast agents.
Unlike systemic sclerosis, NSF tends to spare the skin of the face, and there is no association with autoantibodies or inflammatory cells.
The predominant histopathologic features of NSF include fibrocytelike cells, histiocytes, dermal dendritic cells, scarlike fibrosis, mucin, edema, calcification, and ossification. Diagnosing NSF requires both histopathologic and clinical findings.
SAN: Have any risk factors for the various conditions been identified?
Dr. Pope: As noted, NSF develops in patients with kidney disease and exposure to gadolinium. Renal failure is a major risk factor. With morphea, many patients who develop the condition are more likely than those without it to have an additional autoimmune disease.
SAN: How are the various conditions treated?
Dr. Pope: Corticosteroids and often methotrexate are used to treat EF, and early, aggressive treatment can minimize damage associated with the disease. Morphea is usually self-limited, and there is no proven treatment, although anecdotal evidence suggests that morphea patients who are being treated with antimalarials often develop new lesions when the treatment is stopped. IV immunoglobulin has been used in scleromyxedema treatment with mixed results.
Finally, there is no single therapeutic agent that has been proved effective for NSF. Anecdotal evidence suggests that extracorporeal photopheresis can lead to some lessening of skin thickening as well as increased mobility. Agents such as pentoxifylline, sodium, thiosulfate, and imatinib mesylate might improve skin fibrosis, but prevention through avoidance of gadolinium exposure in at-risk patients is advocated.
Dr. Pope is an associate professor of medicine in the division of rheumatology at the University of Western Ontario and a rheumatologist at St. Joseph's Health Care, both in London, Ont. She reported having no financial conflicts of interest.
Systemic sclerosis and scleroderma are characterized by inflammation and fibrosis of the skin, as well as by vascular abnormalities, visceral damage, and the production of autoantibodies.
Diagnosing the chronic connective tissue disease, which can present in both limited and diffuse forms, can be complicated by the fact that it shares clinical and/or histologic features with a number of "scleroderma mimics," according to Dr. Janet E. Pope.
As with systemic sclerosis, most of these sclerodermalike conditions present with hard skin and tissue fibrosis and should be included in the differential diagnosis, noted Dr. Pope. She offered insight into ways to distinguish sclerodermalike fibrosing skin disorders from systemic sclerosis.
Skin & Allergy News: What are some of the more common "mimics" of systemic sclerosis.
Dr. Pope: Systemic sclerosis can be confused with such sclerodermalike skin disorders as eosinophilic fasciitis (EF), morphea, scleromyxedema, and nephrogenic systemic fibrosis (NSF).
SAN: How can physicians differentiate these from systemic sclerosis?
Dr. Pope: Most of these conditions lack Raynaud's phenomenon, sclerodactyly, and nailfold capillary changes.
Eosinophilic fasciitis (also called Shulman's syndrome) is a rare, localized, fibrosing disorder of subcutaneous tissues. Unlike systemic sclerosis, the overlying skin in EF is flexible enough to be pinched because the pathological disturbance is subcutaneous. This condition is often associated with venous grooving.
Generalized morphea is characterized by cutaneous sclerosis plaques caused by excessive collagen deposition, which leads to thickening of the dermis and/or subcutaneous tissues. Three or more major anatomical regions are affected by plaques in generalized morphea. Studies have shown that morphea often resolves within 2-5 years, but deeper lesions may take longer to resolve and may be accompanied by more disability and damage, depending on where they are located (J. Rheumatol. 1997;24:73-80). Additionally, many patients with morphea have antinuclear antibody and rheumatoid factor autoantibodies.
Scleromyxedema (also called papular mucinosis) is a chronic connective tissue disorder in which mucin deposits cause the skin to become red and raised, and make the movement of affected areas difficult. It tends to involve the head and neck rather than the extremities, and in most cases the sclerodermiform plaques and lichenoid papules are associated with a monoclonal gammopathy.
Finally, nephrogenic systemic fibrosis is a systemic fibrosing condition that occurs in people with kidney disease. NSF affects the skin, muscles, heart, and kidneys. It is strongly associated with exposure to gadolinium-based contrast agents.
Unlike systemic sclerosis, NSF tends to spare the skin of the face, and there is no association with autoantibodies or inflammatory cells.
The predominant histopathologic features of NSF include fibrocytelike cells, histiocytes, dermal dendritic cells, scarlike fibrosis, mucin, edema, calcification, and ossification. Diagnosing NSF requires both histopathologic and clinical findings.
SAN: Have any risk factors for the various conditions been identified?
Dr. Pope: As noted, NSF develops in patients with kidney disease and exposure to gadolinium. Renal failure is a major risk factor. With morphea, many patients who develop the condition are more likely than those without it to have an additional autoimmune disease.
SAN: How are the various conditions treated?
Dr. Pope: Corticosteroids and often methotrexate are used to treat EF, and early, aggressive treatment can minimize damage associated with the disease. Morphea is usually self-limited, and there is no proven treatment, although anecdotal evidence suggests that morphea patients who are being treated with antimalarials often develop new lesions when the treatment is stopped. IV immunoglobulin has been used in scleromyxedema treatment with mixed results.
Finally, there is no single therapeutic agent that has been proved effective for NSF. Anecdotal evidence suggests that extracorporeal photopheresis can lead to some lessening of skin thickening as well as increased mobility. Agents such as pentoxifylline, sodium, thiosulfate, and imatinib mesylate might improve skin fibrosis, but prevention through avoidance of gadolinium exposure in at-risk patients is advocated.
Dr. Pope is an associate professor of medicine in the division of rheumatology at the University of Western Ontario and a rheumatologist at St. Joseph's Health Care, both in London, Ont. She reported having no financial conflicts of interest.
Cancer Risk Greater in Treated RA Patients Than in General Population
BRIGHTON, ENGLAND – The risk of cancer is almost 50% higher in people with rheumatoid arthritis who are treated with nonbiologic agents when compared to the general population, according to data just released from the British Society for Rheumatology Biologics Register.
Although the link between rheumatoid arthritis (RA) and malignancy has been previously identified – particularly with respect to a higher risk of non-Hodgkin’s lymphoma (NHL) – these new data from the British Society for Rheumatology Biologics Register (BSRBR) are from a more-contemporary population of patients who have been treated in the era of widespread use of methotrexate (MTX) and other nonbiologic disease-modifying antirheumatic drugs (DMARDs).
Dr. Eric L. Matteson, who is a professor of medicine at the division of rheumatology at the Mayo Clinic, Rochester, Minn., noted in an interview that the registry findings seem to indicate that overall cancer risk is increased in patients with RA and that this risk is similar in patients exposed to biologics and nonbiologics, with the exception that nonmelanoma skin cancer appears to be somewhat more frequent in the biologics treated patients.
"I think the conclusions arrived at are consistent with previous studies and so not surprising regarding nonbiologics, in particular, methotrexate. A study from Australia by Dr. Rachelle Buchbinder, for example, already reported in 2008 an almost identical finding of 50% increased risk of cancer in patients with RA treated with methotrexate."
There was an estimated 50% excess risk of malignancy among RA patients exposed to MTX relative to the general population (standardized incidence ratio, 1.5; 95% confidence interval, 1.2-1.9). In that study, the risk of non-Hodgkin’s lymphoma was more than five times higher in RA patients than in the general population (SIR, 5.1; 95% CI 2.2-10.0). Other cancers for which there was evidence of increased risk were lung cancer (SIR, 2.9; 95% CI 1.6-4.8) and melanoma (SIR, 3.0; 95% CI 1.2-6.2). (Arthritis Care Res. 2008;59:794-9).
Dr. Matteson qualified his remarks by noting that he did not hear the presentation and assumes methotrexate was the comparator drug.
In reporting the findings, Dr. Louise Mercer, a clinical research fellow at the Arthritis Research UK Epidemiology Unit, University of Manchester, England, where the BRSBR is coordinated, on April 13 at the British Society for Rheumatology Annual Conference (Rheumatology 2011;50:iii37-8, abstract OP18) noted that they set a baseline for analyzing the cancer risk of biologic therapy, compared with conventional RA therapy within the BSRBR patient population – the primary reason the register was set up 10 years ago.
The analysis included 3,727 RA patients treated with nonbiologic DMARDS who were enrolled in the register between 2002 and 2008. Data from the national cancer registry for England and Wales provided information about whether or not they developed cancer. SIRs were used to compare the observed vs. the expected number of cancers and were adjusted for age and gender.
Of 148 malignancies occurring in 10,447 patient-years, 128 were solid cancers – including melanoma, and lung, breast, and colorectal cancers – and 20 were hematologic – including NHL and Hodgkin’s lymphoma. The median time from enrollment in the register to cancer diagnosis was 1.3 years, with 52% of patients dying as a result of incident cancer.
The standardized incidence ratio for all cancers was 1.48. Looking at specific types of malignancy, the highest SIR was recorded for NHL, at 3.73, although the risk of lung cancer was also substantial, with a SIR of 2.71. The SIR for colorectal cancer was 0.96.
Considering these findings, "vigilance for cancer, especially lung cancer and lymphoma should be maintained," Dr. Mercer advised.
"Our experience is that all cancers were increased by about 48%, with quite a marked increase in NHL and lung cancer, said Dr. Deborah Symmons, during a separate presentation at the meeting. An interesting finding, she noted, was that any benefit of NSAID drugs used to treat RA in lowering the risk of colorectal cancer appeared to be "wearing off."
Dr. Symmons, also of the Arthritis Research UK Epidemiology Unit in Manchester and a principle investigator for the BSRBR, explained that this adds to the evidence that, "the cancer risk in our general rheumatoid population is increased for some cancers."
These data are of interest, as they will eventually be used to compare the cancer risk of biologic vs. nonbiologic DMARDs. Such analysis is unlikely to be undertaken any time soon, however, as it is planned to occur when 20,000 years of patient follow-up have been accumulated. To date, there has been no signal that an earlier analysis is warranted.
"In the [anti-TNF] clinical trials, most of which were less than a year [in duration] there was not an increase in cancer except for [nonmelanoma skin cancer]," Dr. Symmons observed. "The cancer risk in observational studies is not increased in the short term," she added, "but we still don’t know what the long-term risk might be."
Echoing her comments were the separate findings of a Wyeth-sponsored study presented as a poster at the meeting. Dr. Peter Taylor of the Kennedy Institute of Rheumatology in London and his associates looked at the risk of malignancy associated with TNF inhibitors in registries and prospective observational studies. They reported a pooled-risk estimate of 1.11 for lymphoma, 1.45 for nonmelanoma skin cancer, and 1.79 for melanoma comparing TNF-treated patients with nonexposed RA patients.
Dr. Taylor and his team said of their findings: "This systematic review and meta-analysis provide reassurance to physicians and patients that treatment of RA patients with TNF inhibitors does not increase the risk of malignancy in general, or of lymphoma in particular, but does appear to increase the risk of skin cancer, including melanoma."
The systemic review and meta-analysis was sponsored by Wyeth. Dr. Taylor has received research grants and honoraria from Abbott, Bristol-Myers Squibb, Pfizer, Roche, Schering-Plough, and UCB. The BSRBR is funded by a grant from the British Society for Rheumatology. The BSR receives funding from Abbott Laboratories, Biovitrum/SOBI, Merck Sharp & Dohme Ltd., Pfizer, Roche, and UCB. This income finances a separate contract between the BSR and the University of Manchester that provides and run the BSRBR. All decisions concerning data analysis, interpretation and publications are made autonomously of any industrial contribution.
Dr. Mercer, Dr. Symmons, and Dr. Matteson declared that they had no personal conflicts of interest.
BRIGHTON, ENGLAND – The risk of cancer is almost 50% higher in people with rheumatoid arthritis who are treated with nonbiologic agents when compared to the general population, according to data just released from the British Society for Rheumatology Biologics Register.
Although the link between rheumatoid arthritis (RA) and malignancy has been previously identified – particularly with respect to a higher risk of non-Hodgkin’s lymphoma (NHL) – these new data from the British Society for Rheumatology Biologics Register (BSRBR) are from a more-contemporary population of patients who have been treated in the era of widespread use of methotrexate (MTX) and other nonbiologic disease-modifying antirheumatic drugs (DMARDs).
Dr. Eric L. Matteson, who is a professor of medicine at the division of rheumatology at the Mayo Clinic, Rochester, Minn., noted in an interview that the registry findings seem to indicate that overall cancer risk is increased in patients with RA and that this risk is similar in patients exposed to biologics and nonbiologics, with the exception that nonmelanoma skin cancer appears to be somewhat more frequent in the biologics treated patients.
"I think the conclusions arrived at are consistent with previous studies and so not surprising regarding nonbiologics, in particular, methotrexate. A study from Australia by Dr. Rachelle Buchbinder, for example, already reported in 2008 an almost identical finding of 50% increased risk of cancer in patients with RA treated with methotrexate."
There was an estimated 50% excess risk of malignancy among RA patients exposed to MTX relative to the general population (standardized incidence ratio, 1.5; 95% confidence interval, 1.2-1.9). In that study, the risk of non-Hodgkin’s lymphoma was more than five times higher in RA patients than in the general population (SIR, 5.1; 95% CI 2.2-10.0). Other cancers for which there was evidence of increased risk were lung cancer (SIR, 2.9; 95% CI 1.6-4.8) and melanoma (SIR, 3.0; 95% CI 1.2-6.2). (Arthritis Care Res. 2008;59:794-9).
Dr. Matteson qualified his remarks by noting that he did not hear the presentation and assumes methotrexate was the comparator drug.
In reporting the findings, Dr. Louise Mercer, a clinical research fellow at the Arthritis Research UK Epidemiology Unit, University of Manchester, England, where the BRSBR is coordinated, on April 13 at the British Society for Rheumatology Annual Conference (Rheumatology 2011;50:iii37-8, abstract OP18) noted that they set a baseline for analyzing the cancer risk of biologic therapy, compared with conventional RA therapy within the BSRBR patient population – the primary reason the register was set up 10 years ago.
The analysis included 3,727 RA patients treated with nonbiologic DMARDS who were enrolled in the register between 2002 and 2008. Data from the national cancer registry for England and Wales provided information about whether or not they developed cancer. SIRs were used to compare the observed vs. the expected number of cancers and were adjusted for age and gender.
Of 148 malignancies occurring in 10,447 patient-years, 128 were solid cancers – including melanoma, and lung, breast, and colorectal cancers – and 20 were hematologic – including NHL and Hodgkin’s lymphoma. The median time from enrollment in the register to cancer diagnosis was 1.3 years, with 52% of patients dying as a result of incident cancer.
The standardized incidence ratio for all cancers was 1.48. Looking at specific types of malignancy, the highest SIR was recorded for NHL, at 3.73, although the risk of lung cancer was also substantial, with a SIR of 2.71. The SIR for colorectal cancer was 0.96.
Considering these findings, "vigilance for cancer, especially lung cancer and lymphoma should be maintained," Dr. Mercer advised.
"Our experience is that all cancers were increased by about 48%, with quite a marked increase in NHL and lung cancer, said Dr. Deborah Symmons, during a separate presentation at the meeting. An interesting finding, she noted, was that any benefit of NSAID drugs used to treat RA in lowering the risk of colorectal cancer appeared to be "wearing off."
Dr. Symmons, also of the Arthritis Research UK Epidemiology Unit in Manchester and a principle investigator for the BSRBR, explained that this adds to the evidence that, "the cancer risk in our general rheumatoid population is increased for some cancers."
These data are of interest, as they will eventually be used to compare the cancer risk of biologic vs. nonbiologic DMARDs. Such analysis is unlikely to be undertaken any time soon, however, as it is planned to occur when 20,000 years of patient follow-up have been accumulated. To date, there has been no signal that an earlier analysis is warranted.
"In the [anti-TNF] clinical trials, most of which were less than a year [in duration] there was not an increase in cancer except for [nonmelanoma skin cancer]," Dr. Symmons observed. "The cancer risk in observational studies is not increased in the short term," she added, "but we still don’t know what the long-term risk might be."
Echoing her comments were the separate findings of a Wyeth-sponsored study presented as a poster at the meeting. Dr. Peter Taylor of the Kennedy Institute of Rheumatology in London and his associates looked at the risk of malignancy associated with TNF inhibitors in registries and prospective observational studies. They reported a pooled-risk estimate of 1.11 for lymphoma, 1.45 for nonmelanoma skin cancer, and 1.79 for melanoma comparing TNF-treated patients with nonexposed RA patients.
Dr. Taylor and his team said of their findings: "This systematic review and meta-analysis provide reassurance to physicians and patients that treatment of RA patients with TNF inhibitors does not increase the risk of malignancy in general, or of lymphoma in particular, but does appear to increase the risk of skin cancer, including melanoma."
The systemic review and meta-analysis was sponsored by Wyeth. Dr. Taylor has received research grants and honoraria from Abbott, Bristol-Myers Squibb, Pfizer, Roche, Schering-Plough, and UCB. The BSRBR is funded by a grant from the British Society for Rheumatology. The BSR receives funding from Abbott Laboratories, Biovitrum/SOBI, Merck Sharp & Dohme Ltd., Pfizer, Roche, and UCB. This income finances a separate contract between the BSR and the University of Manchester that provides and run the BSRBR. All decisions concerning data analysis, interpretation and publications are made autonomously of any industrial contribution.
Dr. Mercer, Dr. Symmons, and Dr. Matteson declared that they had no personal conflicts of interest.
BRIGHTON, ENGLAND – The risk of cancer is almost 50% higher in people with rheumatoid arthritis who are treated with nonbiologic agents when compared to the general population, according to data just released from the British Society for Rheumatology Biologics Register.
Although the link between rheumatoid arthritis (RA) and malignancy has been previously identified – particularly with respect to a higher risk of non-Hodgkin’s lymphoma (NHL) – these new data from the British Society for Rheumatology Biologics Register (BSRBR) are from a more-contemporary population of patients who have been treated in the era of widespread use of methotrexate (MTX) and other nonbiologic disease-modifying antirheumatic drugs (DMARDs).
Dr. Eric L. Matteson, who is a professor of medicine at the division of rheumatology at the Mayo Clinic, Rochester, Minn., noted in an interview that the registry findings seem to indicate that overall cancer risk is increased in patients with RA and that this risk is similar in patients exposed to biologics and nonbiologics, with the exception that nonmelanoma skin cancer appears to be somewhat more frequent in the biologics treated patients.
"I think the conclusions arrived at are consistent with previous studies and so not surprising regarding nonbiologics, in particular, methotrexate. A study from Australia by Dr. Rachelle Buchbinder, for example, already reported in 2008 an almost identical finding of 50% increased risk of cancer in patients with RA treated with methotrexate."
There was an estimated 50% excess risk of malignancy among RA patients exposed to MTX relative to the general population (standardized incidence ratio, 1.5; 95% confidence interval, 1.2-1.9). In that study, the risk of non-Hodgkin’s lymphoma was more than five times higher in RA patients than in the general population (SIR, 5.1; 95% CI 2.2-10.0). Other cancers for which there was evidence of increased risk were lung cancer (SIR, 2.9; 95% CI 1.6-4.8) and melanoma (SIR, 3.0; 95% CI 1.2-6.2). (Arthritis Care Res. 2008;59:794-9).
Dr. Matteson qualified his remarks by noting that he did not hear the presentation and assumes methotrexate was the comparator drug.
In reporting the findings, Dr. Louise Mercer, a clinical research fellow at the Arthritis Research UK Epidemiology Unit, University of Manchester, England, where the BRSBR is coordinated, on April 13 at the British Society for Rheumatology Annual Conference (Rheumatology 2011;50:iii37-8, abstract OP18) noted that they set a baseline for analyzing the cancer risk of biologic therapy, compared with conventional RA therapy within the BSRBR patient population – the primary reason the register was set up 10 years ago.
The analysis included 3,727 RA patients treated with nonbiologic DMARDS who were enrolled in the register between 2002 and 2008. Data from the national cancer registry for England and Wales provided information about whether or not they developed cancer. SIRs were used to compare the observed vs. the expected number of cancers and were adjusted for age and gender.
Of 148 malignancies occurring in 10,447 patient-years, 128 were solid cancers – including melanoma, and lung, breast, and colorectal cancers – and 20 were hematologic – including NHL and Hodgkin’s lymphoma. The median time from enrollment in the register to cancer diagnosis was 1.3 years, with 52% of patients dying as a result of incident cancer.
The standardized incidence ratio for all cancers was 1.48. Looking at specific types of malignancy, the highest SIR was recorded for NHL, at 3.73, although the risk of lung cancer was also substantial, with a SIR of 2.71. The SIR for colorectal cancer was 0.96.
Considering these findings, "vigilance for cancer, especially lung cancer and lymphoma should be maintained," Dr. Mercer advised.
"Our experience is that all cancers were increased by about 48%, with quite a marked increase in NHL and lung cancer, said Dr. Deborah Symmons, during a separate presentation at the meeting. An interesting finding, she noted, was that any benefit of NSAID drugs used to treat RA in lowering the risk of colorectal cancer appeared to be "wearing off."
Dr. Symmons, also of the Arthritis Research UK Epidemiology Unit in Manchester and a principle investigator for the BSRBR, explained that this adds to the evidence that, "the cancer risk in our general rheumatoid population is increased for some cancers."
These data are of interest, as they will eventually be used to compare the cancer risk of biologic vs. nonbiologic DMARDs. Such analysis is unlikely to be undertaken any time soon, however, as it is planned to occur when 20,000 years of patient follow-up have been accumulated. To date, there has been no signal that an earlier analysis is warranted.
"In the [anti-TNF] clinical trials, most of which were less than a year [in duration] there was not an increase in cancer except for [nonmelanoma skin cancer]," Dr. Symmons observed. "The cancer risk in observational studies is not increased in the short term," she added, "but we still don’t know what the long-term risk might be."
Echoing her comments were the separate findings of a Wyeth-sponsored study presented as a poster at the meeting. Dr. Peter Taylor of the Kennedy Institute of Rheumatology in London and his associates looked at the risk of malignancy associated with TNF inhibitors in registries and prospective observational studies. They reported a pooled-risk estimate of 1.11 for lymphoma, 1.45 for nonmelanoma skin cancer, and 1.79 for melanoma comparing TNF-treated patients with nonexposed RA patients.
Dr. Taylor and his team said of their findings: "This systematic review and meta-analysis provide reassurance to physicians and patients that treatment of RA patients with TNF inhibitors does not increase the risk of malignancy in general, or of lymphoma in particular, but does appear to increase the risk of skin cancer, including melanoma."
The systemic review and meta-analysis was sponsored by Wyeth. Dr. Taylor has received research grants and honoraria from Abbott, Bristol-Myers Squibb, Pfizer, Roche, Schering-Plough, and UCB. The BSRBR is funded by a grant from the British Society for Rheumatology. The BSR receives funding from Abbott Laboratories, Biovitrum/SOBI, Merck Sharp & Dohme Ltd., Pfizer, Roche, and UCB. This income finances a separate contract between the BSR and the University of Manchester that provides and run the BSRBR. All decisions concerning data analysis, interpretation and publications are made autonomously of any industrial contribution.
Dr. Mercer, Dr. Symmons, and Dr. Matteson declared that they had no personal conflicts of interest.
FROM THE BRITISH SOCIETY FOR RHEUMATOLOGY ANNUAL CONFERENCE
Major Finding: Standardized incident ratios for NHL, lung cancer, and colorectal cancer were 3.73, 2.71, and 0.96, respectively, comparing RA patients treated with nonbiologic DMARDs with the general population.
Data Source: Six-year, prospective follow-up of 3727 patients with nonbiologically treated RA enrolled in the British Society for Rheumatology Biologics Register (BSRBR).
Disclosures: The BSRBR is funded by a grant from the British Society for Rheumatology (BSR). The BSR receives funding from Abbott Laboratories, Biovitrum/SOBI, Merck Sharp & Dohme Limited, Pfizer, Roche and UCB. This income finances a separate contract between the BSR and the University of Manchester that provides and run the BSRBR. All decisions concerning data analysis, interpretation and publications are made autonomously of any industrial contribution. Dr. Mercer and Dr. Symmons declared that they had no personal conflicts of interest.
Study: Scleroderma Survival Improving
BRIGHTON, ENGLAND – Survival from scleroderma is improving, but major organ-based complications are still a significant reason for premature death, according to the results of a long-term outcomes study that were presented at the annual meeting of the British Society for Rheumatology.
The 10-year survival rates were 81% for patients with limited cutaneous systemic sclerosis (SSc) and 70% for patients with diffuse cutaneous SSc (P = .006).
Survival was significantly reduced (39%) in those who developed major heart, lung, or kidney complications, compared with 71% for those who developed no complications at 15 years' follow-up (P less than .001).
"The 10-year survival is impressive when you consider that not so long ago, we had a 5-year morality approaching 50% in the worst cases. So clearly there has been overall progress in managing the disease," commented study investigator Dr. Christopher Denton.
"Although outcomes are improving, we're still dealing with a disease that has very high case-specific mortality," added Dr. Denton, who is a professor of experimental rheumatology at University College London and a consultant rheumatologist at the Royal Free Hospital, London.
"Around 60% of patients actually die as a direct consequence of their scleroderma, and this is mostly from cardiorespiratory complications of the disease," he added.
Almost 400 patients with scleroderma – of whom 252 (63.3%) had limited cutaneous SSc and 146 (36.7%) had diffuse cutaneous SSc – were studied from their diagnosis in 1995 to 1999 (Rheumatology [Oxford] 2011;50 [suppl. 3]:iii129-iii37, abstract 223). The percentage of men with diffuse cutaneous SSc vs. limited cutaneous SSc disease was higher (17.9% vs. 9.9%; P = .037) and the mean age of onset was 50 years for limited cutaneous SSc patients and 46 years for diffuse cutaneous SSc patients (P = .003).
An important point of the study was that the frequency and timing of the major complications of scleroderma have now been defined over a 10- to 15-year period. Complications of scleroderma can be life threatening, and include pulmonary arterial hypertension (PAH), pulmonary fibrosis, renal disease, and cardiac problems.
At the 10-year follow-up, the incidences of PAH in the limited cutaneous SSc and diffuse cutaneous SSc groups were similar (15% vs. 13%), with the earliest cases seen 2 years after diagnosis. Patients who developed this complication later in the course of their disease tended to have higher 5-year survival rates than did those who had PAH earlier (49% vs. 24%), but this did not reach statistical significance.
Clinically significant pulmonary fibrosis was identified in a higher percentage of patients with diffuse than with limited disease at 10 years (45% vs. 21%; P less than .001), with almost half of patients developing lung disease within the first 3 years, and more than three-quarters within the first 5 years.
Patients with diffuse cutaneous SSc were more likely than those with limited cutaneous SSc to experience renal crisis (12% vs. 4%) at 10 years, with the higher rates occurring in the first 3 years (10% vs. 3%). Similarly, cardiac disease was more common in diffuse cutaneous SSc than in limited cutaneous SSc patients early on (7% vs. 1% at 5 years; P less than .001), but remained unchanged at 10 years.
"Whereas the limited or diffuse distinction is very relevant in the first 5 years, after the 5 years it doesn’t really matter which type of scleroderma you've got; you have an equally high frequency of complications," Dr. Denton observed in an interview.
He added that this "is a treated cohort that received standard management, so I think it's very important to start to understand the timing and frequency of the major heart, lung, and kidney complications, so that you can really identify when in the course of the disease you should be particularly vigilant."
Dr. Denton disclosed financial relationships with Actelion, Aspreva, BioVitrum, Digna, Dyax, Encysive, Genzyme, GlaxoSmithKline, Medimmune, Pfizer, and Sanofi-Aventis.
BRIGHTON, ENGLAND – Survival from scleroderma is improving, but major organ-based complications are still a significant reason for premature death, according to the results of a long-term outcomes study that were presented at the annual meeting of the British Society for Rheumatology.
The 10-year survival rates were 81% for patients with limited cutaneous systemic sclerosis (SSc) and 70% for patients with diffuse cutaneous SSc (P = .006).
Survival was significantly reduced (39%) in those who developed major heart, lung, or kidney complications, compared with 71% for those who developed no complications at 15 years' follow-up (P less than .001).
"The 10-year survival is impressive when you consider that not so long ago, we had a 5-year morality approaching 50% in the worst cases. So clearly there has been overall progress in managing the disease," commented study investigator Dr. Christopher Denton.
"Although outcomes are improving, we're still dealing with a disease that has very high case-specific mortality," added Dr. Denton, who is a professor of experimental rheumatology at University College London and a consultant rheumatologist at the Royal Free Hospital, London.
"Around 60% of patients actually die as a direct consequence of their scleroderma, and this is mostly from cardiorespiratory complications of the disease," he added.
Almost 400 patients with scleroderma – of whom 252 (63.3%) had limited cutaneous SSc and 146 (36.7%) had diffuse cutaneous SSc – were studied from their diagnosis in 1995 to 1999 (Rheumatology [Oxford] 2011;50 [suppl. 3]:iii129-iii37, abstract 223). The percentage of men with diffuse cutaneous SSc vs. limited cutaneous SSc disease was higher (17.9% vs. 9.9%; P = .037) and the mean age of onset was 50 years for limited cutaneous SSc patients and 46 years for diffuse cutaneous SSc patients (P = .003).
An important point of the study was that the frequency and timing of the major complications of scleroderma have now been defined over a 10- to 15-year period. Complications of scleroderma can be life threatening, and include pulmonary arterial hypertension (PAH), pulmonary fibrosis, renal disease, and cardiac problems.
At the 10-year follow-up, the incidences of PAH in the limited cutaneous SSc and diffuse cutaneous SSc groups were similar (15% vs. 13%), with the earliest cases seen 2 years after diagnosis. Patients who developed this complication later in the course of their disease tended to have higher 5-year survival rates than did those who had PAH earlier (49% vs. 24%), but this did not reach statistical significance.
Clinically significant pulmonary fibrosis was identified in a higher percentage of patients with diffuse than with limited disease at 10 years (45% vs. 21%; P less than .001), with almost half of patients developing lung disease within the first 3 years, and more than three-quarters within the first 5 years.
Patients with diffuse cutaneous SSc were more likely than those with limited cutaneous SSc to experience renal crisis (12% vs. 4%) at 10 years, with the higher rates occurring in the first 3 years (10% vs. 3%). Similarly, cardiac disease was more common in diffuse cutaneous SSc than in limited cutaneous SSc patients early on (7% vs. 1% at 5 years; P less than .001), but remained unchanged at 10 years.
"Whereas the limited or diffuse distinction is very relevant in the first 5 years, after the 5 years it doesn’t really matter which type of scleroderma you've got; you have an equally high frequency of complications," Dr. Denton observed in an interview.
He added that this "is a treated cohort that received standard management, so I think it's very important to start to understand the timing and frequency of the major heart, lung, and kidney complications, so that you can really identify when in the course of the disease you should be particularly vigilant."
Dr. Denton disclosed financial relationships with Actelion, Aspreva, BioVitrum, Digna, Dyax, Encysive, Genzyme, GlaxoSmithKline, Medimmune, Pfizer, and Sanofi-Aventis.
BRIGHTON, ENGLAND – Survival from scleroderma is improving, but major organ-based complications are still a significant reason for premature death, according to the results of a long-term outcomes study that were presented at the annual meeting of the British Society for Rheumatology.
The 10-year survival rates were 81% for patients with limited cutaneous systemic sclerosis (SSc) and 70% for patients with diffuse cutaneous SSc (P = .006).
Survival was significantly reduced (39%) in those who developed major heart, lung, or kidney complications, compared with 71% for those who developed no complications at 15 years' follow-up (P less than .001).
"The 10-year survival is impressive when you consider that not so long ago, we had a 5-year morality approaching 50% in the worst cases. So clearly there has been overall progress in managing the disease," commented study investigator Dr. Christopher Denton.
"Although outcomes are improving, we're still dealing with a disease that has very high case-specific mortality," added Dr. Denton, who is a professor of experimental rheumatology at University College London and a consultant rheumatologist at the Royal Free Hospital, London.
"Around 60% of patients actually die as a direct consequence of their scleroderma, and this is mostly from cardiorespiratory complications of the disease," he added.
Almost 400 patients with scleroderma – of whom 252 (63.3%) had limited cutaneous SSc and 146 (36.7%) had diffuse cutaneous SSc – were studied from their diagnosis in 1995 to 1999 (Rheumatology [Oxford] 2011;50 [suppl. 3]:iii129-iii37, abstract 223). The percentage of men with diffuse cutaneous SSc vs. limited cutaneous SSc disease was higher (17.9% vs. 9.9%; P = .037) and the mean age of onset was 50 years for limited cutaneous SSc patients and 46 years for diffuse cutaneous SSc patients (P = .003).
An important point of the study was that the frequency and timing of the major complications of scleroderma have now been defined over a 10- to 15-year period. Complications of scleroderma can be life threatening, and include pulmonary arterial hypertension (PAH), pulmonary fibrosis, renal disease, and cardiac problems.
At the 10-year follow-up, the incidences of PAH in the limited cutaneous SSc and diffuse cutaneous SSc groups were similar (15% vs. 13%), with the earliest cases seen 2 years after diagnosis. Patients who developed this complication later in the course of their disease tended to have higher 5-year survival rates than did those who had PAH earlier (49% vs. 24%), but this did not reach statistical significance.
Clinically significant pulmonary fibrosis was identified in a higher percentage of patients with diffuse than with limited disease at 10 years (45% vs. 21%; P less than .001), with almost half of patients developing lung disease within the first 3 years, and more than three-quarters within the first 5 years.
Patients with diffuse cutaneous SSc were more likely than those with limited cutaneous SSc to experience renal crisis (12% vs. 4%) at 10 years, with the higher rates occurring in the first 3 years (10% vs. 3%). Similarly, cardiac disease was more common in diffuse cutaneous SSc than in limited cutaneous SSc patients early on (7% vs. 1% at 5 years; P less than .001), but remained unchanged at 10 years.
"Whereas the limited or diffuse distinction is very relevant in the first 5 years, after the 5 years it doesn’t really matter which type of scleroderma you've got; you have an equally high frequency of complications," Dr. Denton observed in an interview.
He added that this "is a treated cohort that received standard management, so I think it's very important to start to understand the timing and frequency of the major heart, lung, and kidney complications, so that you can really identify when in the course of the disease you should be particularly vigilant."
Dr. Denton disclosed financial relationships with Actelion, Aspreva, BioVitrum, Digna, Dyax, Encysive, Genzyme, GlaxoSmithKline, Medimmune, Pfizer, and Sanofi-Aventis.
FROM THE ANNUAL MEETING OF THE BRITISH SOCIETY FOR RHEUMATOLOGY
Major Finding: At 10 years after diagnosis, survival was 81% for patients with limited cutaneous SSc and 70% for those with diffuse cutaneous SSc.
Data Source: Long-term follow-up of 398 scleroderma patients with disease onset from 1995 to 1999.
Disclosures: Dr. Denton disclosed financial relationships with Actelion, Aspreva, BioVitrum, Digna, Dyax, Encysive, Genzyme, GSK, Medimmune, Pfizer, and Sanofi-Aventis.
Heart Involvement in Systemic Sclerosis Underappreciated
SNOWMASS, COLO. – Cardiac abnormalities were detected by magnetic resonance imaging in three-quarters of an unselected consecutive series of systemic sclerosis patients, underscoring the impressive frequency of heart involvement in this collagen vascular disease.
"The heart is something we often forget in scleroderma. The heart disease is underestimated," Dr. Fredrick M. Wigley said at the symposium sponsored by the American College of Rheumatology.
The hallmark of cardiac involvement in systemic sclerosis (SSc) is fibrosis and inflammation. Cardiac MRI is unequalled at visualizing these features, he said.
"You can see fibrosis of the myocardium, pericardium, coronary circulation, and conduction system. Arrhythmias are common. Coronary vasospasm is thought to occur, particularly with cold conduction – the so-called Raynaud's of the heart – leading to ischemic reperfusion injury and fibrosis of the heart," said Dr. Wigley, professor of medicine and director of the scleroderma center at Johns Hopkins University, Baltimore.
A resting tachycardia in patients with systemic sclerosis is a common clinical manifestation of cardiac involvement. Clinically evident heart disease carries an unfavorable prognosis, as do cardiac abnormalities detected via right heart catheterization or other invasive methods. The prognostic significance of asymptomatic abnormalities that are detected only on cardiac MRI and that are not evident at the bedside remains to be established. The noninvasive imaging technique has only recently been applied in systemic sclerosis.
Scleroderma patients at greatest risk for clinically severe cardiac involvement are those with myopathy and rapidly progressing skin disease.
Dr. Wigley highlighted a recent study by investigators at Lille 2 (France) University that effectively demonstrated the power of cardiac MRI in detecting heart involvement in SSc. The French investigators examined 52 consecutive unselected scleroderma patients with both Doppler echocardiography and cardiac MRI. One or more cardiac abnormalities were found on cardiac MRI in 75% of the patients, while Doppler detected the abnormalities in only 48% of the patients.
Moreover, only cardiac MRI permitted precise analysis of the patterns of cardiac involvement in SSc, as it was able to distinguish between the fibrotic, inflammatory, and microvascular components. Interestingly, patients with limited cutaneous SSc had cardiac MRI abnormalities that were similar to those with diffuse cutaneous disease.
Seven of the 40 patients without pulmonary arterial hypertension were found to have right ventricular dilation on cardiac MRI, underscoring the point that right ventricular dilation is not specific for this common respiratory manifestation of SSc.
Study participants had a mean 11.2-year disease history since developing Raynaud’s phenomenon. The longer a patient’s disease duration, the greater the number of cardiac segments with kinetic abnormalities and delayed contrast enhancement on MRI (Ann. Rheum. Dis. 2009;68:1878-84).
Dr. Wigley said that while to date no therapy has been shown to alter the natural course of cardiac disease in patients with scleroderma, French investigators strongly believe calcium channel blockers are cardioprotective, and they have documented increased myocardial perfusion in nifedipine-treated SSc patients.
He said that he receives consulting fees and/or research grants from Actelion, Amira, Kinemed, Medimmune, Novartis, Orion, Pfizer, and United Therapeutics.
SNOWMASS, COLO. – Cardiac abnormalities were detected by magnetic resonance imaging in three-quarters of an unselected consecutive series of systemic sclerosis patients, underscoring the impressive frequency of heart involvement in this collagen vascular disease.
"The heart is something we often forget in scleroderma. The heart disease is underestimated," Dr. Fredrick M. Wigley said at the symposium sponsored by the American College of Rheumatology.
The hallmark of cardiac involvement in systemic sclerosis (SSc) is fibrosis and inflammation. Cardiac MRI is unequalled at visualizing these features, he said.
"You can see fibrosis of the myocardium, pericardium, coronary circulation, and conduction system. Arrhythmias are common. Coronary vasospasm is thought to occur, particularly with cold conduction – the so-called Raynaud's of the heart – leading to ischemic reperfusion injury and fibrosis of the heart," said Dr. Wigley, professor of medicine and director of the scleroderma center at Johns Hopkins University, Baltimore.
A resting tachycardia in patients with systemic sclerosis is a common clinical manifestation of cardiac involvement. Clinically evident heart disease carries an unfavorable prognosis, as do cardiac abnormalities detected via right heart catheterization or other invasive methods. The prognostic significance of asymptomatic abnormalities that are detected only on cardiac MRI and that are not evident at the bedside remains to be established. The noninvasive imaging technique has only recently been applied in systemic sclerosis.
Scleroderma patients at greatest risk for clinically severe cardiac involvement are those with myopathy and rapidly progressing skin disease.
Dr. Wigley highlighted a recent study by investigators at Lille 2 (France) University that effectively demonstrated the power of cardiac MRI in detecting heart involvement in SSc. The French investigators examined 52 consecutive unselected scleroderma patients with both Doppler echocardiography and cardiac MRI. One or more cardiac abnormalities were found on cardiac MRI in 75% of the patients, while Doppler detected the abnormalities in only 48% of the patients.
Moreover, only cardiac MRI permitted precise analysis of the patterns of cardiac involvement in SSc, as it was able to distinguish between the fibrotic, inflammatory, and microvascular components. Interestingly, patients with limited cutaneous SSc had cardiac MRI abnormalities that were similar to those with diffuse cutaneous disease.
Seven of the 40 patients without pulmonary arterial hypertension were found to have right ventricular dilation on cardiac MRI, underscoring the point that right ventricular dilation is not specific for this common respiratory manifestation of SSc.
Study participants had a mean 11.2-year disease history since developing Raynaud’s phenomenon. The longer a patient’s disease duration, the greater the number of cardiac segments with kinetic abnormalities and delayed contrast enhancement on MRI (Ann. Rheum. Dis. 2009;68:1878-84).
Dr. Wigley said that while to date no therapy has been shown to alter the natural course of cardiac disease in patients with scleroderma, French investigators strongly believe calcium channel blockers are cardioprotective, and they have documented increased myocardial perfusion in nifedipine-treated SSc patients.
He said that he receives consulting fees and/or research grants from Actelion, Amira, Kinemed, Medimmune, Novartis, Orion, Pfizer, and United Therapeutics.
SNOWMASS, COLO. – Cardiac abnormalities were detected by magnetic resonance imaging in three-quarters of an unselected consecutive series of systemic sclerosis patients, underscoring the impressive frequency of heart involvement in this collagen vascular disease.
"The heart is something we often forget in scleroderma. The heart disease is underestimated," Dr. Fredrick M. Wigley said at the symposium sponsored by the American College of Rheumatology.
The hallmark of cardiac involvement in systemic sclerosis (SSc) is fibrosis and inflammation. Cardiac MRI is unequalled at visualizing these features, he said.
"You can see fibrosis of the myocardium, pericardium, coronary circulation, and conduction system. Arrhythmias are common. Coronary vasospasm is thought to occur, particularly with cold conduction – the so-called Raynaud's of the heart – leading to ischemic reperfusion injury and fibrosis of the heart," said Dr. Wigley, professor of medicine and director of the scleroderma center at Johns Hopkins University, Baltimore.
A resting tachycardia in patients with systemic sclerosis is a common clinical manifestation of cardiac involvement. Clinically evident heart disease carries an unfavorable prognosis, as do cardiac abnormalities detected via right heart catheterization or other invasive methods. The prognostic significance of asymptomatic abnormalities that are detected only on cardiac MRI and that are not evident at the bedside remains to be established. The noninvasive imaging technique has only recently been applied in systemic sclerosis.
Scleroderma patients at greatest risk for clinically severe cardiac involvement are those with myopathy and rapidly progressing skin disease.
Dr. Wigley highlighted a recent study by investigators at Lille 2 (France) University that effectively demonstrated the power of cardiac MRI in detecting heart involvement in SSc. The French investigators examined 52 consecutive unselected scleroderma patients with both Doppler echocardiography and cardiac MRI. One or more cardiac abnormalities were found on cardiac MRI in 75% of the patients, while Doppler detected the abnormalities in only 48% of the patients.
Moreover, only cardiac MRI permitted precise analysis of the patterns of cardiac involvement in SSc, as it was able to distinguish between the fibrotic, inflammatory, and microvascular components. Interestingly, patients with limited cutaneous SSc had cardiac MRI abnormalities that were similar to those with diffuse cutaneous disease.
Seven of the 40 patients without pulmonary arterial hypertension were found to have right ventricular dilation on cardiac MRI, underscoring the point that right ventricular dilation is not specific for this common respiratory manifestation of SSc.
Study participants had a mean 11.2-year disease history since developing Raynaud’s phenomenon. The longer a patient’s disease duration, the greater the number of cardiac segments with kinetic abnormalities and delayed contrast enhancement on MRI (Ann. Rheum. Dis. 2009;68:1878-84).
Dr. Wigley said that while to date no therapy has been shown to alter the natural course of cardiac disease in patients with scleroderma, French investigators strongly believe calcium channel blockers are cardioprotective, and they have documented increased myocardial perfusion in nifedipine-treated SSc patients.
He said that he receives consulting fees and/or research grants from Actelion, Amira, Kinemed, Medimmune, Novartis, Orion, Pfizer, and United Therapeutics.
EXPERT ANALYSIS FROM A SYMPOSIUM SPONSORED BY THE AMERICAN COLLEGE OF RHEUMATOLOGY
Psoriasis Linked With 6% Higher Cardiovascular Disease Risk
NEW ORLEANS – Patients with severe psoriasis face a 6% higher 10-year risk for a cardiovascular event than do comparable people without psoriasis, according to the findings of a prospective cohort study of nearly 18,000 people.
This added cardiovascular risk can have substantial implications, as it can move psoriasis patients into a higher Framingham Risk Score category and shift the way physicians need to think about cardiovascular risk management of these patients, Dr. Nehal N. Mehta said at the annual meeting of the American College of Cardiology.
In his practice that focuses on adults with psoriasis, the average background Framingham Risk Score based on low density lipoprotein cholesterol is a 7% 10-year risk for having a cardiovascular event, a level defined as low risk. However, adding the 6% additional risk linked with their psoriasis results in many patients having a 10-year risk of 13% or higher, placing them in the intermediate risk category (generally defined as a 10%-20% risk of having a cardiovascular event).
Patients at that intermediate risk category usually have more stringent targets for lipid levels, blood pressure, and weight although, in this context, it's unclear whether patients who leap into a higher cardiovascular risk level because of their psoriasis require more aggressive medical management; cardiovascular risk management guidelines have yet to elucidate optimal management for this patient subgroup.
Dr. Mehta deals with this dilemma by implementing aggressive lifestyle interventions for these patients, and also by suggesting naturally occurring risk-reduction interventions, such as the consumption of fiber, red yeast rice, soy, phytoestrogens, fish oil, and niacin.
If, after all these interventions, a patient's low density lipoprotein cholesterol or blood pressure remains at a questionably high level, he discusses the option of starting treatment with a statin or an antihypertensive medication, making clear that these steps have not yet been endorsed by most society management guidelines.
"Ultimately, about 5% of my psoriasis patients end up on a statin," said Dr. Mehta, a cardiologist and director of the inflammatory risk clinic in preventive cardiology at the University of Pennsylvania, Philadelphia.
Dr. Mehta and his associates derived an estimate of cardiovascular disease risk attributable to psoriasis by reviewing follow-up data maintained on 3,603 patients with severe psoriasis and 14,330 control participants without psoriasis enrolled in the General Practice Research Database, a collection of records from more than 5 million people seen by U.K. general practice physicians.
The researchers identified cases of severe psoriasis based on their receiving systemic therapy or phototherapy and excluded people with a prior history of cardiovascular events. The average age of all the people in the analysis was about 50 years and, on average, people were followed for about 3 years.
In a multivariable analysis that controlled for diabetes, hypertension, hyperlipidemia, age, gender, body mass index, and smoking status, the risk for a myocardial infarction, stroke, or death from a cardiovascular cause was 53% higher among the psoriasis patients, compared with the controls, a statistically significant difference. This higher cardiovascular risk among patients with psoriasis matched the 50% increased risk proposed last year for patients with rheumatoid arthritis and other forms of inflammatory arthritis including psoriatic arthritis, according to a panel convened by the European League Against Rheumatism (Ann. Rheum. Dis. 2010;69:325-31).
To translate the 1.53 relative risk into an attributable risk, Dr. Mehta and his associates multiplied that factor against the background cardiovascular risk for someone in the general population of the study to derive an adjusted risk. They then subtracted the background risk from the adjusted risk. Over a 10-year period, this translated into an excess risk for a cardiovascular event of 6.2%.
To illustrate the potential impact of this estimate, the researchers then applied this to a consecutive sample of 103 psoriasis patients seen in Dr. Mehta's psoriasis clinic at the Penn Heart and Vascular Center, including nine patients with psoriatic arthritis.
The baseline risk for these men and women averaged 7.3%, a low-risk level, but with the additional 6.2% risk added their functional risk jumped to an average of 13.5%, or to the intermediate-risk level. For individual patients, this signaled a substantial shift in their Framingham Risk Score risk level. Dr. Mehta conceded that the risk adjustment he applied derived from patients with severe psoriasis, while only 10% of patients in his practice have severe disease. About 60% have mild psoriasis, and about 30% have moderately severe disease, he said.
"This is the best we can do" for the time being, he said. "We applied the severe psoriasis metric to everyone to get a hazard estimate. We believe this is better than just multiplying" to recalculate a person's risk, the approach suggested by the EULAR committee.
He hopes that a larger, prospective study he has begun in collaboration with Dr. Joel M. Gelfand, a dermatologist at the University of Pennsylvania and the senior investigator for this work, will eventually provide a more nuanced risk adjustment for all levels of psoriasis severity. But he said that the current estimate of the increased risk will help persuade psoriasis patients to adopt healthier lifestyles. Patients with psoriasis, at all severity levels, tend to have relatively high rates of obesity, smoking, diabetes, hypertension, and inactivity.
Dr. Mehta added that better medical control of psoriasis also might help blunt the increased cardiovascular risk.
"Psoriasis and atherosclerosis are both T-cell mediated diseases," he observed. Most likely what goes on in the skin -to form the psoriasis plaques - also is going on inside patients' blood vessels, he said.
Dr. Mehta said that he had no disclosures.
NEW ORLEANS – Patients with severe psoriasis face a 6% higher 10-year risk for a cardiovascular event than do comparable people without psoriasis, according to the findings of a prospective cohort study of nearly 18,000 people.
This added cardiovascular risk can have substantial implications, as it can move psoriasis patients into a higher Framingham Risk Score category and shift the way physicians need to think about cardiovascular risk management of these patients, Dr. Nehal N. Mehta said at the annual meeting of the American College of Cardiology.
In his practice that focuses on adults with psoriasis, the average background Framingham Risk Score based on low density lipoprotein cholesterol is a 7% 10-year risk for having a cardiovascular event, a level defined as low risk. However, adding the 6% additional risk linked with their psoriasis results in many patients having a 10-year risk of 13% or higher, placing them in the intermediate risk category (generally defined as a 10%-20% risk of having a cardiovascular event).
Patients at that intermediate risk category usually have more stringent targets for lipid levels, blood pressure, and weight although, in this context, it's unclear whether patients who leap into a higher cardiovascular risk level because of their psoriasis require more aggressive medical management; cardiovascular risk management guidelines have yet to elucidate optimal management for this patient subgroup.
Dr. Mehta deals with this dilemma by implementing aggressive lifestyle interventions for these patients, and also by suggesting naturally occurring risk-reduction interventions, such as the consumption of fiber, red yeast rice, soy, phytoestrogens, fish oil, and niacin.
If, after all these interventions, a patient's low density lipoprotein cholesterol or blood pressure remains at a questionably high level, he discusses the option of starting treatment with a statin or an antihypertensive medication, making clear that these steps have not yet been endorsed by most society management guidelines.
"Ultimately, about 5% of my psoriasis patients end up on a statin," said Dr. Mehta, a cardiologist and director of the inflammatory risk clinic in preventive cardiology at the University of Pennsylvania, Philadelphia.
Dr. Mehta and his associates derived an estimate of cardiovascular disease risk attributable to psoriasis by reviewing follow-up data maintained on 3,603 patients with severe psoriasis and 14,330 control participants without psoriasis enrolled in the General Practice Research Database, a collection of records from more than 5 million people seen by U.K. general practice physicians.
The researchers identified cases of severe psoriasis based on their receiving systemic therapy or phototherapy and excluded people with a prior history of cardiovascular events. The average age of all the people in the analysis was about 50 years and, on average, people were followed for about 3 years.
In a multivariable analysis that controlled for diabetes, hypertension, hyperlipidemia, age, gender, body mass index, and smoking status, the risk for a myocardial infarction, stroke, or death from a cardiovascular cause was 53% higher among the psoriasis patients, compared with the controls, a statistically significant difference. This higher cardiovascular risk among patients with psoriasis matched the 50% increased risk proposed last year for patients with rheumatoid arthritis and other forms of inflammatory arthritis including psoriatic arthritis, according to a panel convened by the European League Against Rheumatism (Ann. Rheum. Dis. 2010;69:325-31).
To translate the 1.53 relative risk into an attributable risk, Dr. Mehta and his associates multiplied that factor against the background cardiovascular risk for someone in the general population of the study to derive an adjusted risk. They then subtracted the background risk from the adjusted risk. Over a 10-year period, this translated into an excess risk for a cardiovascular event of 6.2%.
To illustrate the potential impact of this estimate, the researchers then applied this to a consecutive sample of 103 psoriasis patients seen in Dr. Mehta's psoriasis clinic at the Penn Heart and Vascular Center, including nine patients with psoriatic arthritis.
The baseline risk for these men and women averaged 7.3%, a low-risk level, but with the additional 6.2% risk added their functional risk jumped to an average of 13.5%, or to the intermediate-risk level. For individual patients, this signaled a substantial shift in their Framingham Risk Score risk level. Dr. Mehta conceded that the risk adjustment he applied derived from patients with severe psoriasis, while only 10% of patients in his practice have severe disease. About 60% have mild psoriasis, and about 30% have moderately severe disease, he said.
"This is the best we can do" for the time being, he said. "We applied the severe psoriasis metric to everyone to get a hazard estimate. We believe this is better than just multiplying" to recalculate a person's risk, the approach suggested by the EULAR committee.
He hopes that a larger, prospective study he has begun in collaboration with Dr. Joel M. Gelfand, a dermatologist at the University of Pennsylvania and the senior investigator for this work, will eventually provide a more nuanced risk adjustment for all levels of psoriasis severity. But he said that the current estimate of the increased risk will help persuade psoriasis patients to adopt healthier lifestyles. Patients with psoriasis, at all severity levels, tend to have relatively high rates of obesity, smoking, diabetes, hypertension, and inactivity.
Dr. Mehta added that better medical control of psoriasis also might help blunt the increased cardiovascular risk.
"Psoriasis and atherosclerosis are both T-cell mediated diseases," he observed. Most likely what goes on in the skin -to form the psoriasis plaques - also is going on inside patients' blood vessels, he said.
Dr. Mehta said that he had no disclosures.
NEW ORLEANS – Patients with severe psoriasis face a 6% higher 10-year risk for a cardiovascular event than do comparable people without psoriasis, according to the findings of a prospective cohort study of nearly 18,000 people.
This added cardiovascular risk can have substantial implications, as it can move psoriasis patients into a higher Framingham Risk Score category and shift the way physicians need to think about cardiovascular risk management of these patients, Dr. Nehal N. Mehta said at the annual meeting of the American College of Cardiology.
In his practice that focuses on adults with psoriasis, the average background Framingham Risk Score based on low density lipoprotein cholesterol is a 7% 10-year risk for having a cardiovascular event, a level defined as low risk. However, adding the 6% additional risk linked with their psoriasis results in many patients having a 10-year risk of 13% or higher, placing them in the intermediate risk category (generally defined as a 10%-20% risk of having a cardiovascular event).
Patients at that intermediate risk category usually have more stringent targets for lipid levels, blood pressure, and weight although, in this context, it's unclear whether patients who leap into a higher cardiovascular risk level because of their psoriasis require more aggressive medical management; cardiovascular risk management guidelines have yet to elucidate optimal management for this patient subgroup.
Dr. Mehta deals with this dilemma by implementing aggressive lifestyle interventions for these patients, and also by suggesting naturally occurring risk-reduction interventions, such as the consumption of fiber, red yeast rice, soy, phytoestrogens, fish oil, and niacin.
If, after all these interventions, a patient's low density lipoprotein cholesterol or blood pressure remains at a questionably high level, he discusses the option of starting treatment with a statin or an antihypertensive medication, making clear that these steps have not yet been endorsed by most society management guidelines.
"Ultimately, about 5% of my psoriasis patients end up on a statin," said Dr. Mehta, a cardiologist and director of the inflammatory risk clinic in preventive cardiology at the University of Pennsylvania, Philadelphia.
Dr. Mehta and his associates derived an estimate of cardiovascular disease risk attributable to psoriasis by reviewing follow-up data maintained on 3,603 patients with severe psoriasis and 14,330 control participants without psoriasis enrolled in the General Practice Research Database, a collection of records from more than 5 million people seen by U.K. general practice physicians.
The researchers identified cases of severe psoriasis based on their receiving systemic therapy or phototherapy and excluded people with a prior history of cardiovascular events. The average age of all the people in the analysis was about 50 years and, on average, people were followed for about 3 years.
In a multivariable analysis that controlled for diabetes, hypertension, hyperlipidemia, age, gender, body mass index, and smoking status, the risk for a myocardial infarction, stroke, or death from a cardiovascular cause was 53% higher among the psoriasis patients, compared with the controls, a statistically significant difference. This higher cardiovascular risk among patients with psoriasis matched the 50% increased risk proposed last year for patients with rheumatoid arthritis and other forms of inflammatory arthritis including psoriatic arthritis, according to a panel convened by the European League Against Rheumatism (Ann. Rheum. Dis. 2010;69:325-31).
To translate the 1.53 relative risk into an attributable risk, Dr. Mehta and his associates multiplied that factor against the background cardiovascular risk for someone in the general population of the study to derive an adjusted risk. They then subtracted the background risk from the adjusted risk. Over a 10-year period, this translated into an excess risk for a cardiovascular event of 6.2%.
To illustrate the potential impact of this estimate, the researchers then applied this to a consecutive sample of 103 psoriasis patients seen in Dr. Mehta's psoriasis clinic at the Penn Heart and Vascular Center, including nine patients with psoriatic arthritis.
The baseline risk for these men and women averaged 7.3%, a low-risk level, but with the additional 6.2% risk added their functional risk jumped to an average of 13.5%, or to the intermediate-risk level. For individual patients, this signaled a substantial shift in their Framingham Risk Score risk level. Dr. Mehta conceded that the risk adjustment he applied derived from patients with severe psoriasis, while only 10% of patients in his practice have severe disease. About 60% have mild psoriasis, and about 30% have moderately severe disease, he said.
"This is the best we can do" for the time being, he said. "We applied the severe psoriasis metric to everyone to get a hazard estimate. We believe this is better than just multiplying" to recalculate a person's risk, the approach suggested by the EULAR committee.
He hopes that a larger, prospective study he has begun in collaboration with Dr. Joel M. Gelfand, a dermatologist at the University of Pennsylvania and the senior investigator for this work, will eventually provide a more nuanced risk adjustment for all levels of psoriasis severity. But he said that the current estimate of the increased risk will help persuade psoriasis patients to adopt healthier lifestyles. Patients with psoriasis, at all severity levels, tend to have relatively high rates of obesity, smoking, diabetes, hypertension, and inactivity.
Dr. Mehta added that better medical control of psoriasis also might help blunt the increased cardiovascular risk.
"Psoriasis and atherosclerosis are both T-cell mediated diseases," he observed. Most likely what goes on in the skin -to form the psoriasis plaques - also is going on inside patients' blood vessels, he said.
Dr. Mehta said that he had no disclosures.
FROM THE ANNUAL MEETING OF THE AMERICAN COLLEGE OF CARDIOLOGY
Major Finding: Patients with severe psoriasis have a 6% increased 10-year risk for cardiovascular disease events, compared with similar people without psoriasis.
Data Source: A prospective cohort study of 3,603 patients with severe psoriasis and 14,330 matched people without psoriasis followed for an average of 3 years in the General Practice Research Database.
Disclosures: Dr. Mehta said that he had no disclosures.