User login
New Psoriatic Arthritis Guidelines Highlight Paucity of Data
Use of disease-modifying antirheumatic drugs like methotrexate, sulfasalazine, and leflunomide may improve the outcome of severe psoriatic arthritis if they are given earlier in the course of the disease.
That is one of the recommendations from a new set of guidelines on the treatment of psoriatic arthritis (PsA), issued by a European League Against Rheumatism (EULAR) task force and published online Sept. 27 in Annals of the Rheumatic Diseases.
According to Dr. Laure Gossec, lead author of the report, the recommendations from 28 rheumatologists, 2 patients, an infectious disease specialist, a dermatologist, a physiotherapist, and 2 rheumatology fellows are meant to offer some guidance for what is often a "clinically heterogeneous" disease with few available high-quality studies.
"The treatment of psoriatic arthritis has changed dramatically over recent years, despite the lack of sufficient knowledge on etiology and detailed pathogenesis," wrote Dr. Gossec of Paris Descartes University and colleagues (Ann. Rheum. Dis. 2011 [doi:10.1136/annrheumdis-2011-200350]).
And although the new guidelines "reflect the current state of evidence and thought in the area of PsA management, ... several of the recommendations are strongly based on expert opinion, which in turn is based on clinical practice that has emerged in certain institutions, rather than available evidence," wrote the authors. "This is due to the paucity of data in the field of PsA."
The first recommendation, agreed upon unanimously by the task force, is that "in the vast majority of PsA patients, NSAIDs [nonsteroidal anti-inflammatory drugs] should be used as first-line treatment, although the data regarding the usefulness of NSAIDs in PsA are limited."
The task force conceded that in the setting of cardiovascular or gastrointestinal disease, these drugs, including cyclooxygenase-2 inhibitors – which data suggest are as effective as nonselective NSAIDs for PsA – may not be appropriate.
A second recommendation concerned the "early" use of disease-modifying antirheumatic drugs (DMARDs), specifically within 1 year of diagnosis for patients with active disease (i.e., one or more inflamed/tender joints) whose symptoms are unrelieved by NSAIDs.
However, "regarding the choice of a DMARD, here again there are few data and almost no head-to-head comparisons," wrote the authors.
Nevertheless, "the experts recommended methotrexate as the first-choice DMARD ... based in particular on broad therapeutic dose ranges, different application forms [oral or subcutaneous preparations], and available data in PsA and in other inflammatory diseases."
"In the vast majority of PsA patients, NSAIDs should be used as first-line treatment."
"Transaminase enzymes should be carefully monitored in patients with PsA who receive treatment with methotrexate or leflunomide," especially in the setting of comorbid alcohol abuse, diabetes, or statin therapy, they added.
The authors also weighed in on the use of steroids, especially in localized disease. "Intra-articular steroids are efficacious for mono/oligoarthritis or single joint flares in otherwise well-controlled polyarthritis," they wrote. Systemic steroid use, however, should be undertaken with caution, in light of case reports of skin flares that occur at the time of tapering. And when doses are being tapered, "attention should be paid to the potential worsening of skin disease (rebound)," they added.
Finally, the group addressed the use of tumor necrosis factor inhibitors, which can be considered in the event of "treatment failure," defined as an inability to reach target low disease activity after 3-6 months of treatment. In a very few patients, TNF inhibitors may be considered as a first-line treatment in DMARD-naive patients with very active disease. This recommendation is based entirely on expert opinion; there are no data to support it.
"TNF inhibitors (adalimumab, etanercept, golimumab, and infliximab) have demonstrated efficacy in PsA, both for skin and joint involvement, as well as in preventing radiographic damage," wrote the authors.
They added: "This information may be taken into consideration for the choice of TNF inhibitors in patients with clinically significant skin involvement."
The development of the recommendations was sponsored by EULAR. Several of the authors of this article disclosed financial relationships to numerous pharmaceutical companies and the makers of drugs used in PsA treatment.
Use of disease-modifying antirheumatic drugs like methotrexate, sulfasalazine, and leflunomide may improve the outcome of severe psoriatic arthritis if they are given earlier in the course of the disease.
That is one of the recommendations from a new set of guidelines on the treatment of psoriatic arthritis (PsA), issued by a European League Against Rheumatism (EULAR) task force and published online Sept. 27 in Annals of the Rheumatic Diseases.
According to Dr. Laure Gossec, lead author of the report, the recommendations from 28 rheumatologists, 2 patients, an infectious disease specialist, a dermatologist, a physiotherapist, and 2 rheumatology fellows are meant to offer some guidance for what is often a "clinically heterogeneous" disease with few available high-quality studies.
"The treatment of psoriatic arthritis has changed dramatically over recent years, despite the lack of sufficient knowledge on etiology and detailed pathogenesis," wrote Dr. Gossec of Paris Descartes University and colleagues (Ann. Rheum. Dis. 2011 [doi:10.1136/annrheumdis-2011-200350]).
And although the new guidelines "reflect the current state of evidence and thought in the area of PsA management, ... several of the recommendations are strongly based on expert opinion, which in turn is based on clinical practice that has emerged in certain institutions, rather than available evidence," wrote the authors. "This is due to the paucity of data in the field of PsA."
The first recommendation, agreed upon unanimously by the task force, is that "in the vast majority of PsA patients, NSAIDs [nonsteroidal anti-inflammatory drugs] should be used as first-line treatment, although the data regarding the usefulness of NSAIDs in PsA are limited."
The task force conceded that in the setting of cardiovascular or gastrointestinal disease, these drugs, including cyclooxygenase-2 inhibitors – which data suggest are as effective as nonselective NSAIDs for PsA – may not be appropriate.
A second recommendation concerned the "early" use of disease-modifying antirheumatic drugs (DMARDs), specifically within 1 year of diagnosis for patients with active disease (i.e., one or more inflamed/tender joints) whose symptoms are unrelieved by NSAIDs.
However, "regarding the choice of a DMARD, here again there are few data and almost no head-to-head comparisons," wrote the authors.
Nevertheless, "the experts recommended methotrexate as the first-choice DMARD ... based in particular on broad therapeutic dose ranges, different application forms [oral or subcutaneous preparations], and available data in PsA and in other inflammatory diseases."
"In the vast majority of PsA patients, NSAIDs should be used as first-line treatment."
"Transaminase enzymes should be carefully monitored in patients with PsA who receive treatment with methotrexate or leflunomide," especially in the setting of comorbid alcohol abuse, diabetes, or statin therapy, they added.
The authors also weighed in on the use of steroids, especially in localized disease. "Intra-articular steroids are efficacious for mono/oligoarthritis or single joint flares in otherwise well-controlled polyarthritis," they wrote. Systemic steroid use, however, should be undertaken with caution, in light of case reports of skin flares that occur at the time of tapering. And when doses are being tapered, "attention should be paid to the potential worsening of skin disease (rebound)," they added.
Finally, the group addressed the use of tumor necrosis factor inhibitors, which can be considered in the event of "treatment failure," defined as an inability to reach target low disease activity after 3-6 months of treatment. In a very few patients, TNF inhibitors may be considered as a first-line treatment in DMARD-naive patients with very active disease. This recommendation is based entirely on expert opinion; there are no data to support it.
"TNF inhibitors (adalimumab, etanercept, golimumab, and infliximab) have demonstrated efficacy in PsA, both for skin and joint involvement, as well as in preventing radiographic damage," wrote the authors.
They added: "This information may be taken into consideration for the choice of TNF inhibitors in patients with clinically significant skin involvement."
The development of the recommendations was sponsored by EULAR. Several of the authors of this article disclosed financial relationships to numerous pharmaceutical companies and the makers of drugs used in PsA treatment.
Use of disease-modifying antirheumatic drugs like methotrexate, sulfasalazine, and leflunomide may improve the outcome of severe psoriatic arthritis if they are given earlier in the course of the disease.
That is one of the recommendations from a new set of guidelines on the treatment of psoriatic arthritis (PsA), issued by a European League Against Rheumatism (EULAR) task force and published online Sept. 27 in Annals of the Rheumatic Diseases.
According to Dr. Laure Gossec, lead author of the report, the recommendations from 28 rheumatologists, 2 patients, an infectious disease specialist, a dermatologist, a physiotherapist, and 2 rheumatology fellows are meant to offer some guidance for what is often a "clinically heterogeneous" disease with few available high-quality studies.
"The treatment of psoriatic arthritis has changed dramatically over recent years, despite the lack of sufficient knowledge on etiology and detailed pathogenesis," wrote Dr. Gossec of Paris Descartes University and colleagues (Ann. Rheum. Dis. 2011 [doi:10.1136/annrheumdis-2011-200350]).
And although the new guidelines "reflect the current state of evidence and thought in the area of PsA management, ... several of the recommendations are strongly based on expert opinion, which in turn is based on clinical practice that has emerged in certain institutions, rather than available evidence," wrote the authors. "This is due to the paucity of data in the field of PsA."
The first recommendation, agreed upon unanimously by the task force, is that "in the vast majority of PsA patients, NSAIDs [nonsteroidal anti-inflammatory drugs] should be used as first-line treatment, although the data regarding the usefulness of NSAIDs in PsA are limited."
The task force conceded that in the setting of cardiovascular or gastrointestinal disease, these drugs, including cyclooxygenase-2 inhibitors – which data suggest are as effective as nonselective NSAIDs for PsA – may not be appropriate.
A second recommendation concerned the "early" use of disease-modifying antirheumatic drugs (DMARDs), specifically within 1 year of diagnosis for patients with active disease (i.e., one or more inflamed/tender joints) whose symptoms are unrelieved by NSAIDs.
However, "regarding the choice of a DMARD, here again there are few data and almost no head-to-head comparisons," wrote the authors.
Nevertheless, "the experts recommended methotrexate as the first-choice DMARD ... based in particular on broad therapeutic dose ranges, different application forms [oral or subcutaneous preparations], and available data in PsA and in other inflammatory diseases."
"In the vast majority of PsA patients, NSAIDs should be used as first-line treatment."
"Transaminase enzymes should be carefully monitored in patients with PsA who receive treatment with methotrexate or leflunomide," especially in the setting of comorbid alcohol abuse, diabetes, or statin therapy, they added.
The authors also weighed in on the use of steroids, especially in localized disease. "Intra-articular steroids are efficacious for mono/oligoarthritis or single joint flares in otherwise well-controlled polyarthritis," they wrote. Systemic steroid use, however, should be undertaken with caution, in light of case reports of skin flares that occur at the time of tapering. And when doses are being tapered, "attention should be paid to the potential worsening of skin disease (rebound)," they added.
Finally, the group addressed the use of tumor necrosis factor inhibitors, which can be considered in the event of "treatment failure," defined as an inability to reach target low disease activity after 3-6 months of treatment. In a very few patients, TNF inhibitors may be considered as a first-line treatment in DMARD-naive patients with very active disease. This recommendation is based entirely on expert opinion; there are no data to support it.
"TNF inhibitors (adalimumab, etanercept, golimumab, and infliximab) have demonstrated efficacy in PsA, both for skin and joint involvement, as well as in preventing radiographic damage," wrote the authors.
They added: "This information may be taken into consideration for the choice of TNF inhibitors in patients with clinically significant skin involvement."
The development of the recommendations was sponsored by EULAR. Several of the authors of this article disclosed financial relationships to numerous pharmaceutical companies and the makers of drugs used in PsA treatment.
FROM ANNALS OF THE RHEUMATIC DISEASES
Major Finding: New recommendations on the treatment of PsA emphasize early use of disease-modifying antirheumatic drugs, as well as anti–tumor necrosis factor inhibitors for skin involvement.
Data Source: A panel of experts convened by the European League Against Rheumatism.
Disclosures: The development of the recommendations was sponsored by EULAR. Several of the authors of this article disclosed financial relationships to numerous pharmaceutical companies and the makers of drugs used in PsA treatment.
Metal Orthopedic Implants Unlikely to Trigger Allergy
NAPA, CALIF. – There is little risk that patients with orthopedic metal implants will have a skin reaction or joint pain from the device, said Dr. Joseph F. Fowler.
"I do patch test patients ... who have concerns about a metal allergy or are going to have an implant. I tell them if they have a positive patch test, they may have a skin reaction," said Dr. Fowler. However, there is a small chance they will have a cutaneous reaction, and a smaller chance they will have problems with the joint.
Metals used in orthopedic implants include stainless steel, which is composed of an iron-chromium alloy plus nickel and molybdenum, and sometimes a little chromium and titanium; nitinol (55% nickel and 45% titanium); vitallium (iron cobalt plus chromium 30% and molybdenum 5%); and titanium. Two of the four metals contain nickel, a common contact allergen, noted Dr. Fowler, a dermatologist at the University of Louisville (Ky.).
When it comes to joint pain from implants, findings from a small German study of 15 patients showed that levels of the inflammatory marker interleukin-17 (IL-17) were increased in only those patients with nickel containing replacement joints who both had a positive patch test to nickel and complaints of joint pain. IL-17 levels were not increased in patients with a positive patch test to nickel who did not complain of joint pain (Contact Dermatitis 2010;63:15-22).
In an unpublished study, Dr. James Taylor of the Cleveland Clinic found that 60% of a small group of patients (10) with a metal implant who had a positive patch to metal experienced resolution of joint pain after the metal was replaced. "There is getting to be more and more of a suggestion that metal allergies and joint problems do go hand in hand," said Dr. Fowler.
If you just had to guess what would be the best material to put in a patient who with a chance of a metal allergy, titanium is the best bet, he said. "It comes down to a medical legal question. Will doctors and patients accept a small amount of risk if there is an implant that is better than titanium?"
He said that he will patch test patients if requested by the physician who will be performing the implant surgery, but it is not normally done because there is less than a 1% chance that even if they do have a positive patch test to metal, dermatitis will occur. There have been reports, although rare, that a metal allergy may cause joint failure, but prospective studies are needed, he said.
Dr. Fowler reported financial conflicts of interest with multiple pharmaceutical companies.
NAPA, CALIF. – There is little risk that patients with orthopedic metal implants will have a skin reaction or joint pain from the device, said Dr. Joseph F. Fowler.
"I do patch test patients ... who have concerns about a metal allergy or are going to have an implant. I tell them if they have a positive patch test, they may have a skin reaction," said Dr. Fowler. However, there is a small chance they will have a cutaneous reaction, and a smaller chance they will have problems with the joint.
Metals used in orthopedic implants include stainless steel, which is composed of an iron-chromium alloy plus nickel and molybdenum, and sometimes a little chromium and titanium; nitinol (55% nickel and 45% titanium); vitallium (iron cobalt plus chromium 30% and molybdenum 5%); and titanium. Two of the four metals contain nickel, a common contact allergen, noted Dr. Fowler, a dermatologist at the University of Louisville (Ky.).
When it comes to joint pain from implants, findings from a small German study of 15 patients showed that levels of the inflammatory marker interleukin-17 (IL-17) were increased in only those patients with nickel containing replacement joints who both had a positive patch test to nickel and complaints of joint pain. IL-17 levels were not increased in patients with a positive patch test to nickel who did not complain of joint pain (Contact Dermatitis 2010;63:15-22).
In an unpublished study, Dr. James Taylor of the Cleveland Clinic found that 60% of a small group of patients (10) with a metal implant who had a positive patch to metal experienced resolution of joint pain after the metal was replaced. "There is getting to be more and more of a suggestion that metal allergies and joint problems do go hand in hand," said Dr. Fowler.
If you just had to guess what would be the best material to put in a patient who with a chance of a metal allergy, titanium is the best bet, he said. "It comes down to a medical legal question. Will doctors and patients accept a small amount of risk if there is an implant that is better than titanium?"
He said that he will patch test patients if requested by the physician who will be performing the implant surgery, but it is not normally done because there is less than a 1% chance that even if they do have a positive patch test to metal, dermatitis will occur. There have been reports, although rare, that a metal allergy may cause joint failure, but prospective studies are needed, he said.
Dr. Fowler reported financial conflicts of interest with multiple pharmaceutical companies.
NAPA, CALIF. – There is little risk that patients with orthopedic metal implants will have a skin reaction or joint pain from the device, said Dr. Joseph F. Fowler.
"I do patch test patients ... who have concerns about a metal allergy or are going to have an implant. I tell them if they have a positive patch test, they may have a skin reaction," said Dr. Fowler. However, there is a small chance they will have a cutaneous reaction, and a smaller chance they will have problems with the joint.
Metals used in orthopedic implants include stainless steel, which is composed of an iron-chromium alloy plus nickel and molybdenum, and sometimes a little chromium and titanium; nitinol (55% nickel and 45% titanium); vitallium (iron cobalt plus chromium 30% and molybdenum 5%); and titanium. Two of the four metals contain nickel, a common contact allergen, noted Dr. Fowler, a dermatologist at the University of Louisville (Ky.).
When it comes to joint pain from implants, findings from a small German study of 15 patients showed that levels of the inflammatory marker interleukin-17 (IL-17) were increased in only those patients with nickel containing replacement joints who both had a positive patch test to nickel and complaints of joint pain. IL-17 levels were not increased in patients with a positive patch test to nickel who did not complain of joint pain (Contact Dermatitis 2010;63:15-22).
In an unpublished study, Dr. James Taylor of the Cleveland Clinic found that 60% of a small group of patients (10) with a metal implant who had a positive patch to metal experienced resolution of joint pain after the metal was replaced. "There is getting to be more and more of a suggestion that metal allergies and joint problems do go hand in hand," said Dr. Fowler.
If you just had to guess what would be the best material to put in a patient who with a chance of a metal allergy, titanium is the best bet, he said. "It comes down to a medical legal question. Will doctors and patients accept a small amount of risk if there is an implant that is better than titanium?"
He said that he will patch test patients if requested by the physician who will be performing the implant surgery, but it is not normally done because there is less than a 1% chance that even if they do have a positive patch test to metal, dermatitis will occur. There have been reports, although rare, that a metal allergy may cause joint failure, but prospective studies are needed, he said.
Dr. Fowler reported financial conflicts of interest with multiple pharmaceutical companies.
EXPERT ANALYSIS FROM THE COASTAL DERMATOLOGY SYMPOSIUM
Pharma-Sponsored Comparison Studies Hold Clinical Value
NAPA, CALIF. – "You are almost never going to see a study where the sponsoring company’s product wasn’t at least as good if not better," than the comparator drug, said Dr. Joseph F. Fowler. But that reality does not necessarily mean the study results don’t provide good clinical information.
"Once a drug goes generic, you’re almost never going to see any further study on it."
Most comparison studies are done by pharmaceutical companies and are limited to comparing competing drugs to the company’s branded drugs, explained Dr. Fowler, a dermatologist at the University of Louisville (Ky.). Also, "once a drug goes generic, you’re almost never going to see any further study on it."
Dr. Fowler discussed several comparison studies that run across the dermatology gamut at the meeting, and then asked the audience which drug they prefer.
Ivermectin Vs. Malathion for Lice
In a recent comparison of oral ivermectin to malathion lotion for difficult-to-treat head lice, ivermectin came out slightly on top in terms of clearance at day 15 (95% vs. 85%, respectively), he reported.
In the multicenter trial, 812 patients from 376 households were randomized to receive 400 mcg/kg of body weight of ivermectin or 0.5% malathion lotion on days 1 and 8. The study patients had lice that were not cleared with a topical insecticide 2-6 weeks before study enrollment (N. Engl. J. Med. 2010;362:896-905).
"I tend to use more ivermectin when I see these patients than I ever used to, and I tend to use less topicals," said Dr. Fowler. However, systemic drugs probably have more of a chance of causing toxicity than topicals.
Ivermectin Vs. Permethrin for Scabies
In another comparison of oral ivermectin, this time for scabies, permethrin came out slightly on top. Patients with scabies (n = 85) were randomized to treatment with 200 mcg/kg body weight of ivermectin or a single topical application of 5% permethrin cream. Clearance was seen in 95% of permethrin-treated patients, compared with 70% of ivermectin-treated patients at 8 weeks (J. Am. Acad. Dermatol. 2000;42:236-40).
Topical permethrin was also found to be more effective than topical crotamiton, topical lindane, and oral ivermectin for scabies in a Cochrane database system review (Arch. Dermatol. 2008;144:1638-40).
A show of hands in the audience found that the room was divided on their preferred treatment for head lice and scabies.
Griseofulvin Vs. Terbinafine for Tinea Capitis
A meta-analysis of seven randomized, controlled trials comparing griseofulvin and terbinafine in the treatment of tinea capitis resulted in a draw. However, griseofulvin was found to be slightly better for the Microsporum species, while terbinafine was found to be slightly more effective for the Trichophyton species (J. Am. Acad. Dermatol. 2011;64:663-70).
If cost is an issue, terbinafine is the cheaper drug, noted Dr. Fowler, who said the cost is about $4 in Kentucky. Further, treatment time is shorter with terbinafine at 4 weeks as compared with griseofulvin at 8 weeks. Also, terbinafine appears to be associated with fewer adverse effects than griseofulvin.
The majority of the audience members agreed, via a show of hands, noting their preference for terbinafine to treat children and adults with tinea capitis.
Tazarotene Vs. Adapalene for Acne
In a 16-week, multicenter, randomized, investigator-blinded study, patients treated once a day with tazarotene 0.1% cream had better acne lesion clearance than patients treated with adapalene 0.3% gel. More patients treated with tazarotene had at least a 50% reduction in lesions and less disease severity as compared with adapalene-treated patients (J. Drugs Dermatol. 2010;9:549-58).
However, tazarotene was somewhat less tolerated than adapalene, reported Dr. Fowler. Erythema occurred in 6% of tazarotene patients, dryness in 7%, and peeling in 11%. In the adapalene group, peeling was the only adverse event and was reported by 1% of patients.
By a show of hands, the majority of the audience members prefer adapalene.
Calcitriol Vs. Calcipotriol for Psoriasis
Calcitriol ointment was found to be better tolerated than calcipotriol for treating mild to moderate psoriasis in sensitive areas, such as on the face, around the hairline, and behind the ears (Br. J. Dermatol. 2003;148:326-33).
In the left-to-right, randomized, multicenter, investigator-blinded study, calcitriol 3 mcg ointment was compared with calcipotriol 50 mcg ointment in 75 patients. After 6 weeks, study patients reported preferring calcitriol ointment, which demonstrated "superior" tolerability and equal efficacy, reported Dr. Fowler. Adverse events (contact or irritant dermatitis) occurred in eight patients on the calcipotriol-treated side of their face (one patient reported dermatitis on both sides).
When asked to take cost and possible systemic adverse events into consideration, most audience members noted a preference for calcitriol ointment.
Methotrexate Vs. Azathioprine for Atopy
In the treatment of severe atopic dermatitis, methotrexate and azathioprine were found to be almost equal in efficacy (42% vs. 39%, respectively). Patients (52 completed the study) were randomized to receive 10-22.5 mg of methotrexate per week or 1.5-2.2 mg/kg per day for 12 weeks (J. Allergy Clin. Immunol. 2011;128:353-9). Outcome was assessed by blinded reviewers.
"I think that the adverse event profile of methotrexate in most people is much better, so I tend to disagree with the outcome of this study," said Dr. Fowler.
Dr. Fowler reported financial conflicts of interest with multiple pharmaceutical companies, including many of the products mentioned during his presentation.
NAPA, CALIF. – "You are almost never going to see a study where the sponsoring company’s product wasn’t at least as good if not better," than the comparator drug, said Dr. Joseph F. Fowler. But that reality does not necessarily mean the study results don’t provide good clinical information.
"Once a drug goes generic, you’re almost never going to see any further study on it."
Most comparison studies are done by pharmaceutical companies and are limited to comparing competing drugs to the company’s branded drugs, explained Dr. Fowler, a dermatologist at the University of Louisville (Ky.). Also, "once a drug goes generic, you’re almost never going to see any further study on it."
Dr. Fowler discussed several comparison studies that run across the dermatology gamut at the meeting, and then asked the audience which drug they prefer.
Ivermectin Vs. Malathion for Lice
In a recent comparison of oral ivermectin to malathion lotion for difficult-to-treat head lice, ivermectin came out slightly on top in terms of clearance at day 15 (95% vs. 85%, respectively), he reported.
In the multicenter trial, 812 patients from 376 households were randomized to receive 400 mcg/kg of body weight of ivermectin or 0.5% malathion lotion on days 1 and 8. The study patients had lice that were not cleared with a topical insecticide 2-6 weeks before study enrollment (N. Engl. J. Med. 2010;362:896-905).
"I tend to use more ivermectin when I see these patients than I ever used to, and I tend to use less topicals," said Dr. Fowler. However, systemic drugs probably have more of a chance of causing toxicity than topicals.
Ivermectin Vs. Permethrin for Scabies
In another comparison of oral ivermectin, this time for scabies, permethrin came out slightly on top. Patients with scabies (n = 85) were randomized to treatment with 200 mcg/kg body weight of ivermectin or a single topical application of 5% permethrin cream. Clearance was seen in 95% of permethrin-treated patients, compared with 70% of ivermectin-treated patients at 8 weeks (J. Am. Acad. Dermatol. 2000;42:236-40).
Topical permethrin was also found to be more effective than topical crotamiton, topical lindane, and oral ivermectin for scabies in a Cochrane database system review (Arch. Dermatol. 2008;144:1638-40).
A show of hands in the audience found that the room was divided on their preferred treatment for head lice and scabies.
Griseofulvin Vs. Terbinafine for Tinea Capitis
A meta-analysis of seven randomized, controlled trials comparing griseofulvin and terbinafine in the treatment of tinea capitis resulted in a draw. However, griseofulvin was found to be slightly better for the Microsporum species, while terbinafine was found to be slightly more effective for the Trichophyton species (J. Am. Acad. Dermatol. 2011;64:663-70).
If cost is an issue, terbinafine is the cheaper drug, noted Dr. Fowler, who said the cost is about $4 in Kentucky. Further, treatment time is shorter with terbinafine at 4 weeks as compared with griseofulvin at 8 weeks. Also, terbinafine appears to be associated with fewer adverse effects than griseofulvin.
The majority of the audience members agreed, via a show of hands, noting their preference for terbinafine to treat children and adults with tinea capitis.
Tazarotene Vs. Adapalene for Acne
In a 16-week, multicenter, randomized, investigator-blinded study, patients treated once a day with tazarotene 0.1% cream had better acne lesion clearance than patients treated with adapalene 0.3% gel. More patients treated with tazarotene had at least a 50% reduction in lesions and less disease severity as compared with adapalene-treated patients (J. Drugs Dermatol. 2010;9:549-58).
However, tazarotene was somewhat less tolerated than adapalene, reported Dr. Fowler. Erythema occurred in 6% of tazarotene patients, dryness in 7%, and peeling in 11%. In the adapalene group, peeling was the only adverse event and was reported by 1% of patients.
By a show of hands, the majority of the audience members prefer adapalene.
Calcitriol Vs. Calcipotriol for Psoriasis
Calcitriol ointment was found to be better tolerated than calcipotriol for treating mild to moderate psoriasis in sensitive areas, such as on the face, around the hairline, and behind the ears (Br. J. Dermatol. 2003;148:326-33).
In the left-to-right, randomized, multicenter, investigator-blinded study, calcitriol 3 mcg ointment was compared with calcipotriol 50 mcg ointment in 75 patients. After 6 weeks, study patients reported preferring calcitriol ointment, which demonstrated "superior" tolerability and equal efficacy, reported Dr. Fowler. Adverse events (contact or irritant dermatitis) occurred in eight patients on the calcipotriol-treated side of their face (one patient reported dermatitis on both sides).
When asked to take cost and possible systemic adverse events into consideration, most audience members noted a preference for calcitriol ointment.
Methotrexate Vs. Azathioprine for Atopy
In the treatment of severe atopic dermatitis, methotrexate and azathioprine were found to be almost equal in efficacy (42% vs. 39%, respectively). Patients (52 completed the study) were randomized to receive 10-22.5 mg of methotrexate per week or 1.5-2.2 mg/kg per day for 12 weeks (J. Allergy Clin. Immunol. 2011;128:353-9). Outcome was assessed by blinded reviewers.
"I think that the adverse event profile of methotrexate in most people is much better, so I tend to disagree with the outcome of this study," said Dr. Fowler.
Dr. Fowler reported financial conflicts of interest with multiple pharmaceutical companies, including many of the products mentioned during his presentation.
NAPA, CALIF. – "You are almost never going to see a study where the sponsoring company’s product wasn’t at least as good if not better," than the comparator drug, said Dr. Joseph F. Fowler. But that reality does not necessarily mean the study results don’t provide good clinical information.
"Once a drug goes generic, you’re almost never going to see any further study on it."
Most comparison studies are done by pharmaceutical companies and are limited to comparing competing drugs to the company’s branded drugs, explained Dr. Fowler, a dermatologist at the University of Louisville (Ky.). Also, "once a drug goes generic, you’re almost never going to see any further study on it."
Dr. Fowler discussed several comparison studies that run across the dermatology gamut at the meeting, and then asked the audience which drug they prefer.
Ivermectin Vs. Malathion for Lice
In a recent comparison of oral ivermectin to malathion lotion for difficult-to-treat head lice, ivermectin came out slightly on top in terms of clearance at day 15 (95% vs. 85%, respectively), he reported.
In the multicenter trial, 812 patients from 376 households were randomized to receive 400 mcg/kg of body weight of ivermectin or 0.5% malathion lotion on days 1 and 8. The study patients had lice that were not cleared with a topical insecticide 2-6 weeks before study enrollment (N. Engl. J. Med. 2010;362:896-905).
"I tend to use more ivermectin when I see these patients than I ever used to, and I tend to use less topicals," said Dr. Fowler. However, systemic drugs probably have more of a chance of causing toxicity than topicals.
Ivermectin Vs. Permethrin for Scabies
In another comparison of oral ivermectin, this time for scabies, permethrin came out slightly on top. Patients with scabies (n = 85) were randomized to treatment with 200 mcg/kg body weight of ivermectin or a single topical application of 5% permethrin cream. Clearance was seen in 95% of permethrin-treated patients, compared with 70% of ivermectin-treated patients at 8 weeks (J. Am. Acad. Dermatol. 2000;42:236-40).
Topical permethrin was also found to be more effective than topical crotamiton, topical lindane, and oral ivermectin for scabies in a Cochrane database system review (Arch. Dermatol. 2008;144:1638-40).
A show of hands in the audience found that the room was divided on their preferred treatment for head lice and scabies.
Griseofulvin Vs. Terbinafine for Tinea Capitis
A meta-analysis of seven randomized, controlled trials comparing griseofulvin and terbinafine in the treatment of tinea capitis resulted in a draw. However, griseofulvin was found to be slightly better for the Microsporum species, while terbinafine was found to be slightly more effective for the Trichophyton species (J. Am. Acad. Dermatol. 2011;64:663-70).
If cost is an issue, terbinafine is the cheaper drug, noted Dr. Fowler, who said the cost is about $4 in Kentucky. Further, treatment time is shorter with terbinafine at 4 weeks as compared with griseofulvin at 8 weeks. Also, terbinafine appears to be associated with fewer adverse effects than griseofulvin.
The majority of the audience members agreed, via a show of hands, noting their preference for terbinafine to treat children and adults with tinea capitis.
Tazarotene Vs. Adapalene for Acne
In a 16-week, multicenter, randomized, investigator-blinded study, patients treated once a day with tazarotene 0.1% cream had better acne lesion clearance than patients treated with adapalene 0.3% gel. More patients treated with tazarotene had at least a 50% reduction in lesions and less disease severity as compared with adapalene-treated patients (J. Drugs Dermatol. 2010;9:549-58).
However, tazarotene was somewhat less tolerated than adapalene, reported Dr. Fowler. Erythema occurred in 6% of tazarotene patients, dryness in 7%, and peeling in 11%. In the adapalene group, peeling was the only adverse event and was reported by 1% of patients.
By a show of hands, the majority of the audience members prefer adapalene.
Calcitriol Vs. Calcipotriol for Psoriasis
Calcitriol ointment was found to be better tolerated than calcipotriol for treating mild to moderate psoriasis in sensitive areas, such as on the face, around the hairline, and behind the ears (Br. J. Dermatol. 2003;148:326-33).
In the left-to-right, randomized, multicenter, investigator-blinded study, calcitriol 3 mcg ointment was compared with calcipotriol 50 mcg ointment in 75 patients. After 6 weeks, study patients reported preferring calcitriol ointment, which demonstrated "superior" tolerability and equal efficacy, reported Dr. Fowler. Adverse events (contact or irritant dermatitis) occurred in eight patients on the calcipotriol-treated side of their face (one patient reported dermatitis on both sides).
When asked to take cost and possible systemic adverse events into consideration, most audience members noted a preference for calcitriol ointment.
Methotrexate Vs. Azathioprine for Atopy
In the treatment of severe atopic dermatitis, methotrexate and azathioprine were found to be almost equal in efficacy (42% vs. 39%, respectively). Patients (52 completed the study) were randomized to receive 10-22.5 mg of methotrexate per week or 1.5-2.2 mg/kg per day for 12 weeks (J. Allergy Clin. Immunol. 2011;128:353-9). Outcome was assessed by blinded reviewers.
"I think that the adverse event profile of methotrexate in most people is much better, so I tend to disagree with the outcome of this study," said Dr. Fowler.
Dr. Fowler reported financial conflicts of interest with multiple pharmaceutical companies, including many of the products mentioned during his presentation.
EXPERT ANALYSIS FROM THE COASTAL DERMATOLOGY SYMPOSIUM
An Open-label, Multicenter Study of the Efficacy and Safety of a Weekday/Weekend Treatment Regimen With Calcitriol Ointment 3 μg/g and Clobetasol Propionate Spray 0.05% in the Management of Plaque Psoriasis
Researchers Confirm Psoriatic Arthritis Risk Allele
European researchers have confirmed a new independent susceptibility allele for psoriatic arthritis that could possibly represent a new candidate pharmacogenetic marker for the disease.
"In this collaborative work, we replicated the association of TNF*-857T as a susceptibility allele for [psoriatic arthritis (PsA)] independent of the main PSORS1 risk allele," wrote Emiliano Giardina, Ph.D., of the University of Rome, and his colleagues in an article published online in Arthritis & Rheumatism (doi:10.1002/art.30591).
It’s long been known that the strongest and most replicated susceptibility region for psoriatic vulgaris – and by extension PsA – is within the major histocompatibility complex (MHC) region (PSORS1-psoriasis susceptibility region). "This locus maps to chromosome 6p21.3, comprising many different class I antigens associated with disease expression. Human leukocyte antigen (HLA)-C was repetitively reported as the PSORS1 gene and HLA-Cw*06:02 as the susceptibility allele," they noted.
In this study, the researchers attempted to confirm this finding by assessing allele and genotype frequencies of TNF*-857 in three independent cohorts. They included a German cohort (374 cases and 561 controls), an Italian cohort (400 cases and 400 controls), and a British cohort (135 cases and 354 controls.
The overall cohort of 2,224 individuals of European ancestry was assessed for distribution of HLA-Cw*06:02/PSORS1 risk allele and also was typed for TNF*-857T. The overall allele frequency of TNF*-857T was significantly higher in individuals with PsA (27%) than in control individuals (20%).
The researchers calculated the genotype frequencies of TNF*-857T in samples that were positive and negative for the PSORS1 risk allele, in order to verify the existence of an association independent of the PSORS1 risk allele.
"Overall, the frequency of heterozygous or homozygous carriers of TNF*-857T (TT/CT) in individuals negative for the PSORS1/HLA-C risk allele was significantly higher in PsA patients (30%) than in control subjects (21%)," they reported. This yielded an odds ratio of 1.35
"As expected, the haplotype analysis confirmed TNF*-857T as a susceptibility factor independent of the PSORS1/HLA-C risk allele," they wrote. Association of PsA to TNF*-857T was significant in individuals not carrying the PSORS1/HLA-C risk allele, with an odds ratio of 1.27.
"Although the functional role of TNF*-857T remains to be determined, previous data could show that the allele T increases the transcription of TNF-alpha." In addition, tumor necrosis factor–alpha is known to play a pivotal role in both the activation and extravasation of T-cells in the highly vascularized synovium, as well as in subchondral osteoclastogenesis promoting bone erosions." Genes encoding for TNF-alpha, along with other cytokines, could be candidate pharmacogenetic markers.
The study was supported by a research grant from the Italian Association for the Defense of Psoriatic Patients; the Interdisciplinary Centre for Clinical Research at the University of Erlangen-Nuremberg, Germany; and the Bath Institute for Rheumatic Diseases. Two researchers were received grants from Wyeth-Pharm GmbH (Germany).
A most intriguing aspect of psoriasis epidemiology is that about a quarter of these patients will develop psoriatic arthritis, and this disease clusters in families with remarkably high heritability. Despite evidence for genetic factors in this disease, the search for genes associated with arthritis and not psoriasis has been challenging because almost all PsA patients also have psoriasis. PSORS-1, the MHC region that contains Cw6, the allele with the strongest association with psoriasis, is characterized by long linkage disequilibrium. One report noted an association of PsA with the TNF*-857T allele that was independent of PSORS1. In this multinational study of 909 patients and 1,315 healthy controls, the TNF*-857T allele was found at significantly higher frequency in PsA patients negative for the PSORS1 allele (30%), compared with controls (21%). This study replicates findings in the previous study and is also of note because the TNF*-867T allele increases the transcription of TNF, a pivotal cytokine in the pathogenesis of PsA. Also of possible relevance is that this allele is associated with response to etanercept in RA. The replication of this important association should catalyze additional genetic and pharmacogenetic studies to better understand the diagnostic and functional significance of this allele in psoriatic disease.
Christopher T. Ritchlin, M.D., is professor of medicine, allergy/immunology, and rheumatology at the University of Rochester (N.Y.) He reported having no conflicts of interest that are relevant to this piece.
A most intriguing aspect of psoriasis epidemiology is that about a quarter of these patients will develop psoriatic arthritis, and this disease clusters in families with remarkably high heritability. Despite evidence for genetic factors in this disease, the search for genes associated with arthritis and not psoriasis has been challenging because almost all PsA patients also have psoriasis. PSORS-1, the MHC region that contains Cw6, the allele with the strongest association with psoriasis, is characterized by long linkage disequilibrium. One report noted an association of PsA with the TNF*-857T allele that was independent of PSORS1. In this multinational study of 909 patients and 1,315 healthy controls, the TNF*-857T allele was found at significantly higher frequency in PsA patients negative for the PSORS1 allele (30%), compared with controls (21%). This study replicates findings in the previous study and is also of note because the TNF*-867T allele increases the transcription of TNF, a pivotal cytokine in the pathogenesis of PsA. Also of possible relevance is that this allele is associated with response to etanercept in RA. The replication of this important association should catalyze additional genetic and pharmacogenetic studies to better understand the diagnostic and functional significance of this allele in psoriatic disease.
Christopher T. Ritchlin, M.D., is professor of medicine, allergy/immunology, and rheumatology at the University of Rochester (N.Y.) He reported having no conflicts of interest that are relevant to this piece.
A most intriguing aspect of psoriasis epidemiology is that about a quarter of these patients will develop psoriatic arthritis, and this disease clusters in families with remarkably high heritability. Despite evidence for genetic factors in this disease, the search for genes associated with arthritis and not psoriasis has been challenging because almost all PsA patients also have psoriasis. PSORS-1, the MHC region that contains Cw6, the allele with the strongest association with psoriasis, is characterized by long linkage disequilibrium. One report noted an association of PsA with the TNF*-857T allele that was independent of PSORS1. In this multinational study of 909 patients and 1,315 healthy controls, the TNF*-857T allele was found at significantly higher frequency in PsA patients negative for the PSORS1 allele (30%), compared with controls (21%). This study replicates findings in the previous study and is also of note because the TNF*-867T allele increases the transcription of TNF, a pivotal cytokine in the pathogenesis of PsA. Also of possible relevance is that this allele is associated with response to etanercept in RA. The replication of this important association should catalyze additional genetic and pharmacogenetic studies to better understand the diagnostic and functional significance of this allele in psoriatic disease.
Christopher T. Ritchlin, M.D., is professor of medicine, allergy/immunology, and rheumatology at the University of Rochester (N.Y.) He reported having no conflicts of interest that are relevant to this piece.
European researchers have confirmed a new independent susceptibility allele for psoriatic arthritis that could possibly represent a new candidate pharmacogenetic marker for the disease.
"In this collaborative work, we replicated the association of TNF*-857T as a susceptibility allele for [psoriatic arthritis (PsA)] independent of the main PSORS1 risk allele," wrote Emiliano Giardina, Ph.D., of the University of Rome, and his colleagues in an article published online in Arthritis & Rheumatism (doi:10.1002/art.30591).
It’s long been known that the strongest and most replicated susceptibility region for psoriatic vulgaris – and by extension PsA – is within the major histocompatibility complex (MHC) region (PSORS1-psoriasis susceptibility region). "This locus maps to chromosome 6p21.3, comprising many different class I antigens associated with disease expression. Human leukocyte antigen (HLA)-C was repetitively reported as the PSORS1 gene and HLA-Cw*06:02 as the susceptibility allele," they noted.
In this study, the researchers attempted to confirm this finding by assessing allele and genotype frequencies of TNF*-857 in three independent cohorts. They included a German cohort (374 cases and 561 controls), an Italian cohort (400 cases and 400 controls), and a British cohort (135 cases and 354 controls.
The overall cohort of 2,224 individuals of European ancestry was assessed for distribution of HLA-Cw*06:02/PSORS1 risk allele and also was typed for TNF*-857T. The overall allele frequency of TNF*-857T was significantly higher in individuals with PsA (27%) than in control individuals (20%).
The researchers calculated the genotype frequencies of TNF*-857T in samples that were positive and negative for the PSORS1 risk allele, in order to verify the existence of an association independent of the PSORS1 risk allele.
"Overall, the frequency of heterozygous or homozygous carriers of TNF*-857T (TT/CT) in individuals negative for the PSORS1/HLA-C risk allele was significantly higher in PsA patients (30%) than in control subjects (21%)," they reported. This yielded an odds ratio of 1.35
"As expected, the haplotype analysis confirmed TNF*-857T as a susceptibility factor independent of the PSORS1/HLA-C risk allele," they wrote. Association of PsA to TNF*-857T was significant in individuals not carrying the PSORS1/HLA-C risk allele, with an odds ratio of 1.27.
"Although the functional role of TNF*-857T remains to be determined, previous data could show that the allele T increases the transcription of TNF-alpha." In addition, tumor necrosis factor–alpha is known to play a pivotal role in both the activation and extravasation of T-cells in the highly vascularized synovium, as well as in subchondral osteoclastogenesis promoting bone erosions." Genes encoding for TNF-alpha, along with other cytokines, could be candidate pharmacogenetic markers.
The study was supported by a research grant from the Italian Association for the Defense of Psoriatic Patients; the Interdisciplinary Centre for Clinical Research at the University of Erlangen-Nuremberg, Germany; and the Bath Institute for Rheumatic Diseases. Two researchers were received grants from Wyeth-Pharm GmbH (Germany).
European researchers have confirmed a new independent susceptibility allele for psoriatic arthritis that could possibly represent a new candidate pharmacogenetic marker for the disease.
"In this collaborative work, we replicated the association of TNF*-857T as a susceptibility allele for [psoriatic arthritis (PsA)] independent of the main PSORS1 risk allele," wrote Emiliano Giardina, Ph.D., of the University of Rome, and his colleagues in an article published online in Arthritis & Rheumatism (doi:10.1002/art.30591).
It’s long been known that the strongest and most replicated susceptibility region for psoriatic vulgaris – and by extension PsA – is within the major histocompatibility complex (MHC) region (PSORS1-psoriasis susceptibility region). "This locus maps to chromosome 6p21.3, comprising many different class I antigens associated with disease expression. Human leukocyte antigen (HLA)-C was repetitively reported as the PSORS1 gene and HLA-Cw*06:02 as the susceptibility allele," they noted.
In this study, the researchers attempted to confirm this finding by assessing allele and genotype frequencies of TNF*-857 in three independent cohorts. They included a German cohort (374 cases and 561 controls), an Italian cohort (400 cases and 400 controls), and a British cohort (135 cases and 354 controls.
The overall cohort of 2,224 individuals of European ancestry was assessed for distribution of HLA-Cw*06:02/PSORS1 risk allele and also was typed for TNF*-857T. The overall allele frequency of TNF*-857T was significantly higher in individuals with PsA (27%) than in control individuals (20%).
The researchers calculated the genotype frequencies of TNF*-857T in samples that were positive and negative for the PSORS1 risk allele, in order to verify the existence of an association independent of the PSORS1 risk allele.
"Overall, the frequency of heterozygous or homozygous carriers of TNF*-857T (TT/CT) in individuals negative for the PSORS1/HLA-C risk allele was significantly higher in PsA patients (30%) than in control subjects (21%)," they reported. This yielded an odds ratio of 1.35
"As expected, the haplotype analysis confirmed TNF*-857T as a susceptibility factor independent of the PSORS1/HLA-C risk allele," they wrote. Association of PsA to TNF*-857T was significant in individuals not carrying the PSORS1/HLA-C risk allele, with an odds ratio of 1.27.
"Although the functional role of TNF*-857T remains to be determined, previous data could show that the allele T increases the transcription of TNF-alpha." In addition, tumor necrosis factor–alpha is known to play a pivotal role in both the activation and extravasation of T-cells in the highly vascularized synovium, as well as in subchondral osteoclastogenesis promoting bone erosions." Genes encoding for TNF-alpha, along with other cytokines, could be candidate pharmacogenetic markers.
The study was supported by a research grant from the Italian Association for the Defense of Psoriatic Patients; the Interdisciplinary Centre for Clinical Research at the University of Erlangen-Nuremberg, Germany; and the Bath Institute for Rheumatic Diseases. Two researchers were received grants from Wyeth-Pharm GmbH (Germany).
FROM ARTHRITIS & RHEUMATISM
Major Finding: TNF*-857T is a susceptibility allele for PsA independent of the main PSORS1 risk allele.
Data Source: A study of 2,224 individuals.
Disclosures: The study was supported by a research grant from the Italian Association for the Defense of Psoriatic Patients, the Interdisciplinary Centre for Clinical Research at the University of Erlangen-Nuremberg, and the Bath Institute for Rheumatic Diseases. Two researchers were received grants from Wyeth-Pharm GmbH (Germany).
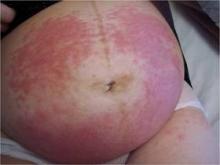
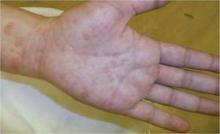
Itchy and Pregnant? Consider Range of Skin Conditions
SAN FRANCISCO – When a pregnant woman presents with a complaint of itching, consider a range of causes, not just those triggered by pregnancy, Dr. Bethanee J. Schlosser advised.
Although some dermatoses of pregnancy are common, a pregnant woman’s itching may have nothing to do with her pregnancy and could be the result of contact dermatitis, drug eruption, scabies, folliculitis, or another cause, Dr. Schlosser said at SDEF Women’s & Pediatric Dermatology Seminar sponsored by Skin Disease Education Foundation (SDEF).
"Just because they’re pregnant doesn’t mean they only have to fit in the pregnancy dermatoses box," Dr. Schlosser of the department of dermatology, and director of the women’s skin health program, at Northwestern University in Chicago said in an interview.
With that said, the two most prominent dermatoses of pregnancy are pruritic urticarial papules and plaques of pregnancy, a condition now known under the umbrella term polymorphic eruption of pregnancy, and pemphigoid gestationis, previously called herpes gestationis.
Polymorphic eruption of pregnancy occurs in about 1 in 300 pregnancies and is generally associated with multiple gestations and increased maternal weight gain. It is also more common in women having their first child. The mean onset is at about 35 weeks, but in about 15% of cases, the onset can be post partum, according to Dr. Schlosser.
Pemphigoid gestationis is a rare acquired autoimmune blistering disease unique to pregnancy. It occurs in 1 in 50,000 pregnancies and is probably the least common dermatosis of pregnancy. The onset is usually in the second or third trimester, but in about 14% of cases, the onset can occur post partum. With pemphigoid gestationis, there is no change in maternal outcome, but there are risks to the fetus including being small for gestational age, preterm delivery, and neonatal pemphigoid disease.
Typically, patients with the polymorphic eruption present with "hivelike" or urticarial papules and plaques, but no blisters, while women with pemphigoid gestationis often have more blistering. However, the clinical presentations and the routine histopathology can be identical, Dr. Schlosser explained.
"I’ve seen patients with both entities, with both kinds of clinical features," she noted. "If it’s in your differential diagnosis and you can’t distinguish 100% clinically, then that’s where the utility of biopsy comes in."
Cutaneous biopsy is a common procedure and is low risk, she reported, even in the context of pregnancy. Routine histopathology and direct immunofluorescence are essential in terms of differentiating between pemphigoid gestationis and polymorphic eruption.
The first-line treatment for both conditions is topical corticosteroids and oral antihistamines when the condition is mild or localized and systemic corticosteroids in severe cases. Although the treatments are generally the same, the difference between the two conditions is not academic, Dr. Schlosser said, because the potential sequelae and considerations for mother and child are different.
Dr. Schlosser also recommended that dermatologists make it a priority to communicate with the referring physician, specifically to review the risks to both the mother and child that may be associated with a particular skin condition or its treatments.
For example, polymorphic eruption of pregnancy is generally nonthreatening to the mother and child. But Dr. Schlosser said she has seen patients with widespread, severe polymorphic eruptions who have needed treatment with systemic corticosteroids. That’s essential information for the ob.gyn.; if the patient has a cesarean delivery, the patient will likely require stress-dose corticosteroids. Similarly, the newborn would need to be monitored for hypoglycemia during the immediate after-birth period.
"That doesn’t mean that dermatologists shouldn’t treat pregnant women aggressively, when appropriate," Dr. Schlosser said. "But the entire multidisciplinary care team needs to be kept informed so that the risks can be managed."
Dr. Schlosser said she had no relevant financial disclosures. SDEF and this news organization are owned by Elsevier.
SAN FRANCISCO – When a pregnant woman presents with a complaint of itching, consider a range of causes, not just those triggered by pregnancy, Dr. Bethanee J. Schlosser advised.
Although some dermatoses of pregnancy are common, a pregnant woman’s itching may have nothing to do with her pregnancy and could be the result of contact dermatitis, drug eruption, scabies, folliculitis, or another cause, Dr. Schlosser said at SDEF Women’s & Pediatric Dermatology Seminar sponsored by Skin Disease Education Foundation (SDEF).
"Just because they’re pregnant doesn’t mean they only have to fit in the pregnancy dermatoses box," Dr. Schlosser of the department of dermatology, and director of the women’s skin health program, at Northwestern University in Chicago said in an interview.
With that said, the two most prominent dermatoses of pregnancy are pruritic urticarial papules and plaques of pregnancy, a condition now known under the umbrella term polymorphic eruption of pregnancy, and pemphigoid gestationis, previously called herpes gestationis.
Polymorphic eruption of pregnancy occurs in about 1 in 300 pregnancies and is generally associated with multiple gestations and increased maternal weight gain. It is also more common in women having their first child. The mean onset is at about 35 weeks, but in about 15% of cases, the onset can be post partum, according to Dr. Schlosser.
Pemphigoid gestationis is a rare acquired autoimmune blistering disease unique to pregnancy. It occurs in 1 in 50,000 pregnancies and is probably the least common dermatosis of pregnancy. The onset is usually in the second or third trimester, but in about 14% of cases, the onset can occur post partum. With pemphigoid gestationis, there is no change in maternal outcome, but there are risks to the fetus including being small for gestational age, preterm delivery, and neonatal pemphigoid disease.
Typically, patients with the polymorphic eruption present with "hivelike" or urticarial papules and plaques, but no blisters, while women with pemphigoid gestationis often have more blistering. However, the clinical presentations and the routine histopathology can be identical, Dr. Schlosser explained.
"I’ve seen patients with both entities, with both kinds of clinical features," she noted. "If it’s in your differential diagnosis and you can’t distinguish 100% clinically, then that’s where the utility of biopsy comes in."
Cutaneous biopsy is a common procedure and is low risk, she reported, even in the context of pregnancy. Routine histopathology and direct immunofluorescence are essential in terms of differentiating between pemphigoid gestationis and polymorphic eruption.
The first-line treatment for both conditions is topical corticosteroids and oral antihistamines when the condition is mild or localized and systemic corticosteroids in severe cases. Although the treatments are generally the same, the difference between the two conditions is not academic, Dr. Schlosser said, because the potential sequelae and considerations for mother and child are different.
Dr. Schlosser also recommended that dermatologists make it a priority to communicate with the referring physician, specifically to review the risks to both the mother and child that may be associated with a particular skin condition or its treatments.
For example, polymorphic eruption of pregnancy is generally nonthreatening to the mother and child. But Dr. Schlosser said she has seen patients with widespread, severe polymorphic eruptions who have needed treatment with systemic corticosteroids. That’s essential information for the ob.gyn.; if the patient has a cesarean delivery, the patient will likely require stress-dose corticosteroids. Similarly, the newborn would need to be monitored for hypoglycemia during the immediate after-birth period.
"That doesn’t mean that dermatologists shouldn’t treat pregnant women aggressively, when appropriate," Dr. Schlosser said. "But the entire multidisciplinary care team needs to be kept informed so that the risks can be managed."
Dr. Schlosser said she had no relevant financial disclosures. SDEF and this news organization are owned by Elsevier.
SAN FRANCISCO – When a pregnant woman presents with a complaint of itching, consider a range of causes, not just those triggered by pregnancy, Dr. Bethanee J. Schlosser advised.
Although some dermatoses of pregnancy are common, a pregnant woman’s itching may have nothing to do with her pregnancy and could be the result of contact dermatitis, drug eruption, scabies, folliculitis, or another cause, Dr. Schlosser said at SDEF Women’s & Pediatric Dermatology Seminar sponsored by Skin Disease Education Foundation (SDEF).
"Just because they’re pregnant doesn’t mean they only have to fit in the pregnancy dermatoses box," Dr. Schlosser of the department of dermatology, and director of the women’s skin health program, at Northwestern University in Chicago said in an interview.
With that said, the two most prominent dermatoses of pregnancy are pruritic urticarial papules and plaques of pregnancy, a condition now known under the umbrella term polymorphic eruption of pregnancy, and pemphigoid gestationis, previously called herpes gestationis.
Polymorphic eruption of pregnancy occurs in about 1 in 300 pregnancies and is generally associated with multiple gestations and increased maternal weight gain. It is also more common in women having their first child. The mean onset is at about 35 weeks, but in about 15% of cases, the onset can be post partum, according to Dr. Schlosser.
Pemphigoid gestationis is a rare acquired autoimmune blistering disease unique to pregnancy. It occurs in 1 in 50,000 pregnancies and is probably the least common dermatosis of pregnancy. The onset is usually in the second or third trimester, but in about 14% of cases, the onset can occur post partum. With pemphigoid gestationis, there is no change in maternal outcome, but there are risks to the fetus including being small for gestational age, preterm delivery, and neonatal pemphigoid disease.
Typically, patients with the polymorphic eruption present with "hivelike" or urticarial papules and plaques, but no blisters, while women with pemphigoid gestationis often have more blistering. However, the clinical presentations and the routine histopathology can be identical, Dr. Schlosser explained.
"I’ve seen patients with both entities, with both kinds of clinical features," she noted. "If it’s in your differential diagnosis and you can’t distinguish 100% clinically, then that’s where the utility of biopsy comes in."
Cutaneous biopsy is a common procedure and is low risk, she reported, even in the context of pregnancy. Routine histopathology and direct immunofluorescence are essential in terms of differentiating between pemphigoid gestationis and polymorphic eruption.
The first-line treatment for both conditions is topical corticosteroids and oral antihistamines when the condition is mild or localized and systemic corticosteroids in severe cases. Although the treatments are generally the same, the difference between the two conditions is not academic, Dr. Schlosser said, because the potential sequelae and considerations for mother and child are different.
Dr. Schlosser also recommended that dermatologists make it a priority to communicate with the referring physician, specifically to review the risks to both the mother and child that may be associated with a particular skin condition or its treatments.
For example, polymorphic eruption of pregnancy is generally nonthreatening to the mother and child. But Dr. Schlosser said she has seen patients with widespread, severe polymorphic eruptions who have needed treatment with systemic corticosteroids. That’s essential information for the ob.gyn.; if the patient has a cesarean delivery, the patient will likely require stress-dose corticosteroids. Similarly, the newborn would need to be monitored for hypoglycemia during the immediate after-birth period.
"That doesn’t mean that dermatologists shouldn’t treat pregnant women aggressively, when appropriate," Dr. Schlosser said. "But the entire multidisciplinary care team needs to be kept informed so that the risks can be managed."
Dr. Schlosser said she had no relevant financial disclosures. SDEF and this news organization are owned by Elsevier.
EXPERT ANALYSIS FROM THE SDEF WOMEN'S & PEDIATRIC DERMATOLOGY SEMINAR
FDA: TNF Blockers Up Risk of Legionella, Listeria Infection
The Food and Drug Administration has updated the boxed warning label of all tumor necrosis factor–alpha blockers to include information on the risk of infection from Legionella and Listeria.
The warnings and precautions sections of the drug labels also have been revised to include information on the risk for serious infections that could lead to death from multiple disease-causing pathogens.
The TNF blockers that will receive the warnings include Remicade (infliximab), Enbrel (etanercept), Humira (adalimumab), Cimzia (certolizumab pegol), and Simponi (golimumab), according to the FDA announcement.
The FDA advised that risks and benefits should be weighed before initiating TNF blocker therapy in patients with chronic or reoccurring infections, or in patients with conditions that may put them at higher risk for infection.
Reports of serious adverse events associated with these and other drugs should be reported to MedWatch at 800-332-1088.
The Food and Drug Administration has updated the boxed warning label of all tumor necrosis factor–alpha blockers to include information on the risk of infection from Legionella and Listeria.
The warnings and precautions sections of the drug labels also have been revised to include information on the risk for serious infections that could lead to death from multiple disease-causing pathogens.
The TNF blockers that will receive the warnings include Remicade (infliximab), Enbrel (etanercept), Humira (adalimumab), Cimzia (certolizumab pegol), and Simponi (golimumab), according to the FDA announcement.
The FDA advised that risks and benefits should be weighed before initiating TNF blocker therapy in patients with chronic or reoccurring infections, or in patients with conditions that may put them at higher risk for infection.
Reports of serious adverse events associated with these and other drugs should be reported to MedWatch at 800-332-1088.
The Food and Drug Administration has updated the boxed warning label of all tumor necrosis factor–alpha blockers to include information on the risk of infection from Legionella and Listeria.
The warnings and precautions sections of the drug labels also have been revised to include information on the risk for serious infections that could lead to death from multiple disease-causing pathogens.
The TNF blockers that will receive the warnings include Remicade (infliximab), Enbrel (etanercept), Humira (adalimumab), Cimzia (certolizumab pegol), and Simponi (golimumab), according to the FDA announcement.
The FDA advised that risks and benefits should be weighed before initiating TNF blocker therapy in patients with chronic or reoccurring infections, or in patients with conditions that may put them at higher risk for infection.
Reports of serious adverse events associated with these and other drugs should be reported to MedWatch at 800-332-1088.