User login
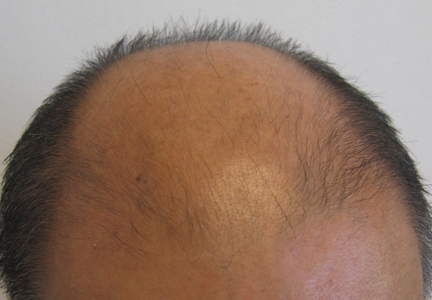
Partial Reversal of Androgenetic Alopecia With Methotrexate Therapy for Psoriasis
Five pearls guide pediatric psoriasis systemic therapy
SAN FRANCISCO – Five practice tips can help physicians consider systemic therapy for the subset of children with psoriasis who develop severe disease at some point, Dr. Kelly M. Cordoro said.
The key is to weigh the risks and benefits to decide which is worse, the treatment or the disease, in terms of how it affects the patient medically, psychosocially, and physically, she said at the annual meeting of the Pacific Dermatologic Association.
Approximately a quarter of the physicians in the audience had treated patients younger than 18 years with systemic therapy for psoriasis, an informal poll showed.*"
The exact approach varies per patient, and not all cases of severe pediatric psoriasis need systemic treatment. "It’s an art as much as it is a science," said Dr. Cordoro of the University of California, San Francisco.
It’s often a good idea to start with nonimmunosuppressive systemic agents for psoriasis in children, such as phototherapy or acitretin, before trying immunosuppressive treatments such as cyclosporine, methotrexate, or biologics, she suggested. Systemic therapy should be added to a multimodal care plan that includes identification and treatment of triggers and comorbidities, topical therapy, lifestyle modifications, education, and support. Keep in mind that no systemic medications are approved to treat psoriasis in children, and management is based on expert opinion, Dr. Cordoro noted.
Dr. Cordoro offered the following five pearls and principles to guide physicians managing severe pediatric psoriasis:
• Don’t just do something, stand there! Treat these children conservatively at first and observe the response, especially in patients with new-onset psoriasis and in particular if they have a family history of psoriasis, said Dr. Cordoro. It’s also a good approach in patients who’ve had a recent infection – group A streptococcal infection is a very common trigger of pediatric psoriasis, for example – or a recent viral illness or skin trauma.
"If you can find the trigger" and treat it, "you may be able to prevent the use of systemic therapy" for the psoriasis, she said. Conservative, yet aggressive, treatment might include a combination of topical therapy, wet wraps, itch control, and managing the triggers. Let the disease evolve for a few days to weeks before moving to systemic therapy, she suggested.
• Don’t just stand there, do something! The psychological impact of chronic disease can be as disabling as the physical impact, and uncontrolled inflammation can cause long-term health consequences, Dr. Cordoro said. No systemic therapies are approved for pediatric psoriasis because of a lack of studies, "but I encourage you to treat these kids" with severe disease who fail topical and other treatment strategies, she said.
Off-label use of methotrexate can help children with any form of psoriasis, in a dose range of 0.2-0.7 mg/kg per week, with a maximum of 25 mg/week, said Dr. Cordoro. Consider this a rescue phase, and then try to taper to the lowest effective dose or off of systemic therapy.
"Methotrexate doesn’t have to be forever," Dr. Cordoro emphasized. In children, methotrexate’s side effects commonly include GI symptoms and anorexia, and the drug very rarely causes pulmonary and hepatotoxicity. Dr. Cordoro recommended a supplemental folate dose of 1-5 mg every day, except methotrexate dose days, to ameliorate some of the GI side effects and the potential bone marrow toxicity. Monitoring is less intense than in adults, and involves frequent lab evaluations but no liver biopsies. Drug interactions with NSAIDs or trimethoprim/sulfamethoxazole can cause bone marrow toxicity, so be sure to send a letter to the child’s pediatrician and parents to alert them, she said.
For severe, diffuse plaque psoriasis in children, any of the conventional systemic or biologic treatments can be effective, and the choice depends on patient and clinical factors, Dr. Cordoro said. The goal is to gain control, taper the systemic treatment, and hold it at the lowest effective dose or transition to topical therapy or phototherapy.
• Don’t forget oral retinoids. Often a forgotten choice for therapy, short-term oral retinoids can be the best first systemic treatment for severe guttate psoriasis if phototherapy is not indicated or unavailable, or for palmoplantar psoriasis or pustular psoriasis, Dr. Cordoro said. In some cases, there can be synergistic effects from combining a low-dose retinoid with low-dose narrow-band UVB phototherapy.
Acitretin is not immunosuppressive but is teratogenic, so it should be avoided in girls older than 8 years, Dr. Cordoro noted. Reversible, dose-dependent side effects include mucocutaneous effects, dyslipidemia, or transaminitis. "Kids cannot tolerate high doses of retinoids, so 1 mg/kg per day or less is where you need to be," she said. Much-feared potential skeletal effects are very rare at doses that low when the medication is used for no more than a year or two, she added.
• When you need speed, try cyclosporine. Cyclosporine acts rapidly. An initial high dose of 5 mg/kg per day can take control of refractory plaque psoriasis, rapidly moving pustular psoriasis, or severe, rapidly progressive psoriasis. No more than a year of continuous use is the guideline, so you don’t want to start low and go slow. "Kids and typically tolerate and need higher doses" compared with adults, Dr. Cordoro said. Use the modified form (Neoral) for better oral bioavailability and better sustained drug levels, she suggested.
This, too, is a rescue treatment intended to control and stabilize the disease, followed by tapering and transitioning to other therapies. The most serious potential side effects include nephrotoxicity, hypertension, and immunosuppression. The most common side effects include nausea, vomiting and diarrhea, anorexia, and headache. Less often, cyclosporine can cause myalgia, arthralgia, paresthesia, gingival hyperplasia, or hypertrichosis. "My colleagues in oncology assure me that at the doses we’re using for psoriasis, we’re not at risk for giving these kids carcinomas down the road," she said.
• Biologics play an important role. Biologics are potent, but wouldn’t be the first choice for systemic therapy because they don’t have prolonged efficacy, said Dr. Cordoro. Their precise place in the therapeutic armamentarium is yet to be defined. "I like to reserve a biologic for when I don’t necessarily have a better choice, because I know I have a finite period of time for biologics," she said.
Nonetheless, anti–tumor necrosis factor agents can play a role in treating refractory plaque psoriasis, severe or refractory generalized pustular psoriasis, or psoriatic arthritis, Dr. Cordoro said. Anecdotal reports suggest that IL-12 and IL-23 inhibitors may help with pediatric psoriasis, but there are no study data yet.
The potential advantages of using biologics include less frequent dosing, less laboratory monitoring, and targeted treatment. On the other hand, biologics are expensive, require injection or infusion, have as-yet-unknown long-term risks, and are not approved for treating pediatric psoriasis (so insurance coverage is a battle), said Dr. Cordoro. Questions remain about standardized dosing and monitoring protocols, and about the endpoints of biologic therapy in this setting, although experience with biologics and evidence of their efficacy and safety in children are accumulating, she said.
Dr. Cordoro recommended two articles that she coauthored that provide expert consensus guidelines, tables, and charts for systemic treatment and monitoring of children with psoriasis (Dermatol. Clin. 2013;31:267-88 and Skin Therapy Lett. 2013;18:1-4).
Dr. Cordoro reported having no financial disclosures.
On Twitter @sherryboschert
*CORRECTION, 11/4/2013: An earlier version of this article imprecisely stated the results of the informal study.
SAN FRANCISCO – Five practice tips can help physicians consider systemic therapy for the subset of children with psoriasis who develop severe disease at some point, Dr. Kelly M. Cordoro said.
The key is to weigh the risks and benefits to decide which is worse, the treatment or the disease, in terms of how it affects the patient medically, psychosocially, and physically, she said at the annual meeting of the Pacific Dermatologic Association.
Approximately a quarter of the physicians in the audience had treated patients younger than 18 years with systemic therapy for psoriasis, an informal poll showed.*"
The exact approach varies per patient, and not all cases of severe pediatric psoriasis need systemic treatment. "It’s an art as much as it is a science," said Dr. Cordoro of the University of California, San Francisco.
It’s often a good idea to start with nonimmunosuppressive systemic agents for psoriasis in children, such as phototherapy or acitretin, before trying immunosuppressive treatments such as cyclosporine, methotrexate, or biologics, she suggested. Systemic therapy should be added to a multimodal care plan that includes identification and treatment of triggers and comorbidities, topical therapy, lifestyle modifications, education, and support. Keep in mind that no systemic medications are approved to treat psoriasis in children, and management is based on expert opinion, Dr. Cordoro noted.
Dr. Cordoro offered the following five pearls and principles to guide physicians managing severe pediatric psoriasis:
• Don’t just do something, stand there! Treat these children conservatively at first and observe the response, especially in patients with new-onset psoriasis and in particular if they have a family history of psoriasis, said Dr. Cordoro. It’s also a good approach in patients who’ve had a recent infection – group A streptococcal infection is a very common trigger of pediatric psoriasis, for example – or a recent viral illness or skin trauma.
"If you can find the trigger" and treat it, "you may be able to prevent the use of systemic therapy" for the psoriasis, she said. Conservative, yet aggressive, treatment might include a combination of topical therapy, wet wraps, itch control, and managing the triggers. Let the disease evolve for a few days to weeks before moving to systemic therapy, she suggested.
• Don’t just stand there, do something! The psychological impact of chronic disease can be as disabling as the physical impact, and uncontrolled inflammation can cause long-term health consequences, Dr. Cordoro said. No systemic therapies are approved for pediatric psoriasis because of a lack of studies, "but I encourage you to treat these kids" with severe disease who fail topical and other treatment strategies, she said.
Off-label use of methotrexate can help children with any form of psoriasis, in a dose range of 0.2-0.7 mg/kg per week, with a maximum of 25 mg/week, said Dr. Cordoro. Consider this a rescue phase, and then try to taper to the lowest effective dose or off of systemic therapy.
"Methotrexate doesn’t have to be forever," Dr. Cordoro emphasized. In children, methotrexate’s side effects commonly include GI symptoms and anorexia, and the drug very rarely causes pulmonary and hepatotoxicity. Dr. Cordoro recommended a supplemental folate dose of 1-5 mg every day, except methotrexate dose days, to ameliorate some of the GI side effects and the potential bone marrow toxicity. Monitoring is less intense than in adults, and involves frequent lab evaluations but no liver biopsies. Drug interactions with NSAIDs or trimethoprim/sulfamethoxazole can cause bone marrow toxicity, so be sure to send a letter to the child’s pediatrician and parents to alert them, she said.
For severe, diffuse plaque psoriasis in children, any of the conventional systemic or biologic treatments can be effective, and the choice depends on patient and clinical factors, Dr. Cordoro said. The goal is to gain control, taper the systemic treatment, and hold it at the lowest effective dose or transition to topical therapy or phototherapy.
• Don’t forget oral retinoids. Often a forgotten choice for therapy, short-term oral retinoids can be the best first systemic treatment for severe guttate psoriasis if phototherapy is not indicated or unavailable, or for palmoplantar psoriasis or pustular psoriasis, Dr. Cordoro said. In some cases, there can be synergistic effects from combining a low-dose retinoid with low-dose narrow-band UVB phototherapy.
Acitretin is not immunosuppressive but is teratogenic, so it should be avoided in girls older than 8 years, Dr. Cordoro noted. Reversible, dose-dependent side effects include mucocutaneous effects, dyslipidemia, or transaminitis. "Kids cannot tolerate high doses of retinoids, so 1 mg/kg per day or less is where you need to be," she said. Much-feared potential skeletal effects are very rare at doses that low when the medication is used for no more than a year or two, she added.
• When you need speed, try cyclosporine. Cyclosporine acts rapidly. An initial high dose of 5 mg/kg per day can take control of refractory plaque psoriasis, rapidly moving pustular psoriasis, or severe, rapidly progressive psoriasis. No more than a year of continuous use is the guideline, so you don’t want to start low and go slow. "Kids and typically tolerate and need higher doses" compared with adults, Dr. Cordoro said. Use the modified form (Neoral) for better oral bioavailability and better sustained drug levels, she suggested.
This, too, is a rescue treatment intended to control and stabilize the disease, followed by tapering and transitioning to other therapies. The most serious potential side effects include nephrotoxicity, hypertension, and immunosuppression. The most common side effects include nausea, vomiting and diarrhea, anorexia, and headache. Less often, cyclosporine can cause myalgia, arthralgia, paresthesia, gingival hyperplasia, or hypertrichosis. "My colleagues in oncology assure me that at the doses we’re using for psoriasis, we’re not at risk for giving these kids carcinomas down the road," she said.
• Biologics play an important role. Biologics are potent, but wouldn’t be the first choice for systemic therapy because they don’t have prolonged efficacy, said Dr. Cordoro. Their precise place in the therapeutic armamentarium is yet to be defined. "I like to reserve a biologic for when I don’t necessarily have a better choice, because I know I have a finite period of time for biologics," she said.
Nonetheless, anti–tumor necrosis factor agents can play a role in treating refractory plaque psoriasis, severe or refractory generalized pustular psoriasis, or psoriatic arthritis, Dr. Cordoro said. Anecdotal reports suggest that IL-12 and IL-23 inhibitors may help with pediatric psoriasis, but there are no study data yet.
The potential advantages of using biologics include less frequent dosing, less laboratory monitoring, and targeted treatment. On the other hand, biologics are expensive, require injection or infusion, have as-yet-unknown long-term risks, and are not approved for treating pediatric psoriasis (so insurance coverage is a battle), said Dr. Cordoro. Questions remain about standardized dosing and monitoring protocols, and about the endpoints of biologic therapy in this setting, although experience with biologics and evidence of their efficacy and safety in children are accumulating, she said.
Dr. Cordoro recommended two articles that she coauthored that provide expert consensus guidelines, tables, and charts for systemic treatment and monitoring of children with psoriasis (Dermatol. Clin. 2013;31:267-88 and Skin Therapy Lett. 2013;18:1-4).
Dr. Cordoro reported having no financial disclosures.
On Twitter @sherryboschert
*CORRECTION, 11/4/2013: An earlier version of this article imprecisely stated the results of the informal study.
SAN FRANCISCO – Five practice tips can help physicians consider systemic therapy for the subset of children with psoriasis who develop severe disease at some point, Dr. Kelly M. Cordoro said.
The key is to weigh the risks and benefits to decide which is worse, the treatment or the disease, in terms of how it affects the patient medically, psychosocially, and physically, she said at the annual meeting of the Pacific Dermatologic Association.
Approximately a quarter of the physicians in the audience had treated patients younger than 18 years with systemic therapy for psoriasis, an informal poll showed.*"
The exact approach varies per patient, and not all cases of severe pediatric psoriasis need systemic treatment. "It’s an art as much as it is a science," said Dr. Cordoro of the University of California, San Francisco.
It’s often a good idea to start with nonimmunosuppressive systemic agents for psoriasis in children, such as phototherapy or acitretin, before trying immunosuppressive treatments such as cyclosporine, methotrexate, or biologics, she suggested. Systemic therapy should be added to a multimodal care plan that includes identification and treatment of triggers and comorbidities, topical therapy, lifestyle modifications, education, and support. Keep in mind that no systemic medications are approved to treat psoriasis in children, and management is based on expert opinion, Dr. Cordoro noted.
Dr. Cordoro offered the following five pearls and principles to guide physicians managing severe pediatric psoriasis:
• Don’t just do something, stand there! Treat these children conservatively at first and observe the response, especially in patients with new-onset psoriasis and in particular if they have a family history of psoriasis, said Dr. Cordoro. It’s also a good approach in patients who’ve had a recent infection – group A streptococcal infection is a very common trigger of pediatric psoriasis, for example – or a recent viral illness or skin trauma.
"If you can find the trigger" and treat it, "you may be able to prevent the use of systemic therapy" for the psoriasis, she said. Conservative, yet aggressive, treatment might include a combination of topical therapy, wet wraps, itch control, and managing the triggers. Let the disease evolve for a few days to weeks before moving to systemic therapy, she suggested.
• Don’t just stand there, do something! The psychological impact of chronic disease can be as disabling as the physical impact, and uncontrolled inflammation can cause long-term health consequences, Dr. Cordoro said. No systemic therapies are approved for pediatric psoriasis because of a lack of studies, "but I encourage you to treat these kids" with severe disease who fail topical and other treatment strategies, she said.
Off-label use of methotrexate can help children with any form of psoriasis, in a dose range of 0.2-0.7 mg/kg per week, with a maximum of 25 mg/week, said Dr. Cordoro. Consider this a rescue phase, and then try to taper to the lowest effective dose or off of systemic therapy.
"Methotrexate doesn’t have to be forever," Dr. Cordoro emphasized. In children, methotrexate’s side effects commonly include GI symptoms and anorexia, and the drug very rarely causes pulmonary and hepatotoxicity. Dr. Cordoro recommended a supplemental folate dose of 1-5 mg every day, except methotrexate dose days, to ameliorate some of the GI side effects and the potential bone marrow toxicity. Monitoring is less intense than in adults, and involves frequent lab evaluations but no liver biopsies. Drug interactions with NSAIDs or trimethoprim/sulfamethoxazole can cause bone marrow toxicity, so be sure to send a letter to the child’s pediatrician and parents to alert them, she said.
For severe, diffuse plaque psoriasis in children, any of the conventional systemic or biologic treatments can be effective, and the choice depends on patient and clinical factors, Dr. Cordoro said. The goal is to gain control, taper the systemic treatment, and hold it at the lowest effective dose or transition to topical therapy or phototherapy.
• Don’t forget oral retinoids. Often a forgotten choice for therapy, short-term oral retinoids can be the best first systemic treatment for severe guttate psoriasis if phototherapy is not indicated or unavailable, or for palmoplantar psoriasis or pustular psoriasis, Dr. Cordoro said. In some cases, there can be synergistic effects from combining a low-dose retinoid with low-dose narrow-band UVB phototherapy.
Acitretin is not immunosuppressive but is teratogenic, so it should be avoided in girls older than 8 years, Dr. Cordoro noted. Reversible, dose-dependent side effects include mucocutaneous effects, dyslipidemia, or transaminitis. "Kids cannot tolerate high doses of retinoids, so 1 mg/kg per day or less is where you need to be," she said. Much-feared potential skeletal effects are very rare at doses that low when the medication is used for no more than a year or two, she added.
• When you need speed, try cyclosporine. Cyclosporine acts rapidly. An initial high dose of 5 mg/kg per day can take control of refractory plaque psoriasis, rapidly moving pustular psoriasis, or severe, rapidly progressive psoriasis. No more than a year of continuous use is the guideline, so you don’t want to start low and go slow. "Kids and typically tolerate and need higher doses" compared with adults, Dr. Cordoro said. Use the modified form (Neoral) for better oral bioavailability and better sustained drug levels, she suggested.
This, too, is a rescue treatment intended to control and stabilize the disease, followed by tapering and transitioning to other therapies. The most serious potential side effects include nephrotoxicity, hypertension, and immunosuppression. The most common side effects include nausea, vomiting and diarrhea, anorexia, and headache. Less often, cyclosporine can cause myalgia, arthralgia, paresthesia, gingival hyperplasia, or hypertrichosis. "My colleagues in oncology assure me that at the doses we’re using for psoriasis, we’re not at risk for giving these kids carcinomas down the road," she said.
• Biologics play an important role. Biologics are potent, but wouldn’t be the first choice for systemic therapy because they don’t have prolonged efficacy, said Dr. Cordoro. Their precise place in the therapeutic armamentarium is yet to be defined. "I like to reserve a biologic for when I don’t necessarily have a better choice, because I know I have a finite period of time for biologics," she said.
Nonetheless, anti–tumor necrosis factor agents can play a role in treating refractory plaque psoriasis, severe or refractory generalized pustular psoriasis, or psoriatic arthritis, Dr. Cordoro said. Anecdotal reports suggest that IL-12 and IL-23 inhibitors may help with pediatric psoriasis, but there are no study data yet.
The potential advantages of using biologics include less frequent dosing, less laboratory monitoring, and targeted treatment. On the other hand, biologics are expensive, require injection or infusion, have as-yet-unknown long-term risks, and are not approved for treating pediatric psoriasis (so insurance coverage is a battle), said Dr. Cordoro. Questions remain about standardized dosing and monitoring protocols, and about the endpoints of biologic therapy in this setting, although experience with biologics and evidence of their efficacy and safety in children are accumulating, she said.
Dr. Cordoro recommended two articles that she coauthored that provide expert consensus guidelines, tables, and charts for systemic treatment and monitoring of children with psoriasis (Dermatol. Clin. 2013;31:267-88 and Skin Therapy Lett. 2013;18:1-4).
Dr. Cordoro reported having no financial disclosures.
On Twitter @sherryboschert
*CORRECTION, 11/4/2013: An earlier version of this article imprecisely stated the results of the informal study.
EXPERT ANALYSIS FROM THE PDA ANNUAL MEETING
The Relationship Between Obesity and Psoriasis in the Pediatric Population: Implications and Future Directions
Suboptimal, dissatisfying treatment affects many psoriasis patients
Despite advancements in therapies for psoriasis, a large proportion of patients are not treated or are receiving suboptimal treatment, according to analysis of a series of comprehensive patient surveys.
The analysis showed that nearly half of the patients with mild psoriasis were receiving no treatments at all in 2011, while 42% of them were treated only with topical agents. In other words, they were undertreated.
On a positive note, the proportion of patients with severe psoriasis who reported receiving no treatment dropped from 30% in the early 2000s to 9% in 2011. Still, 22% of them were undertreated and were prescribed topical medications alone (JAMA Dermatol. 2013 Aug. 14 [doi: 10.1001/jamadermatol.2013.5264]).
The study also revealed that many of the patients were dissatisfied with their treatment, highlighting "a call to action for [dermatologists] to actively seek feedback from psoriasis patients, and find out how their condition is being treated from their perspective," said Dr. April W. Armstrong, lead author of the study and associate professor of dermatology at the University of California, Davis.
"And since many new medications are coming up as well, we should be up to date on the data and literature, and be comfortable with using all sorts of different treatment modalities so that we can offer patients a wide range of treatment options and be able to individualize the treatments," Dr. Armstrong said in an interview.
Meanwhile, the type of health insurance can limit treatment options. In two of the surveys, patients were asked why they discontinued using biological agents. Lack of health insurance was among the top reasons.
"Hopefully, payers will pay attention and find a way to work with providers to make therapies more accessible," Dr. Armstrong said. "Many of our patients need systemic treatments, and I hope payers will pay attention and understand consequences."
Some estimates show that the cost of psoriasis is nearly $11 billion in the United States. But there’s a dearth of studies on patients’ perspectives and the extent to which patients are treated, the authors noted.
One source that has captured such data is the National Psoriasis Foundation’s (NPF’s) biannual surveys of its members. A 2007 study of the survey data was the first to show that as many as 40% of patients with moderate to severe psoriasis didn’t receive treatment (J. Am. Acad. Dermatol. 2007;57:957-62).
For their analysis, Dr. Armstrong and her colleagues examined 13 NPF surveys conducted during 2003-2011. More than 5,600 patients with psoriasis and/or psoriatic arthritis completed the surveys. They had a mean age of 50 years, and most were white.
The analysis showed that the proportion of patients with mild psoriasis who didn’t receive any treatment rose from 42% in the 2003-2005 period to 49% in 2011. The percentage of untreated patients with moderate psoriasis dropped from 36% to 24% during that period, and from 30% to 9% for patients with severe psoriasis.
Dr. Armstrong said there are several explanations for why patients go untreated. Psoriasis is chronic, and after going to one, two, or three doctors and not getting satisfactory results, the patients may give up, she said. "Some of the untreated patients might have sought help before and decided that nothing could be done, and resolved [themselves] to their situation."
Meanwhile, she expressed her concern with the proportion of patients who were undertreated. Close to 30% of patients with moderate psoriasis and 22% of those with severe psoriasis were treated only with topical agents, and the proportions were higher in 2011 than in the 2003-2005 period.
Patients said the top three reasons they used topical agents alone were because they had fewer adverse effects than other treatments, their disease wasn’t serious enough for other kinds of treatments, and their physician wouldn’t prescribe any other treatments.
"There is still much to be learned about exactly why psoriasis patients are undertreated," Dr. Junko Takeshita of the University of Pennsylvania said via e-mail.
But, "it is essential that patients be properly educated about the risks and benefits of various therapies so that they can make informed treatment choices. It is also important for physicians to be aware of and inform their psoriasis patients about the overall health implications of psoriasis itself (i.e., associations with cardiovascular and metabolic diseases as well as emerging associations with other comorbid diseases)," said Dr. Takeshita, who was not involved in the study.
The study also showed the most common forms of various treatment modalities, with ultraviolet B as the most common form of phototherapy, methotrexate as the top oral agent, and etanercept and adalimumab as the most common biological agents.
The authors said that the study had some limitations. The results may have underestimated the data in the general population, because NPF members are more involved in their health care. Also, severity of the disease at the time of the survey may not have been representative of the patients’ disease course, they noted.
"Going forward, it will be important for us to better understand why psoriasis patients are being undertreated and their reasons for treatment dissatisfaction and discontinuation so that, as physicians, we can provide better care to our psoriasis patients," Dr. Takeshita said.
Dr. Armstrong has received research grants or consultant honoraria from Abbott, Amgen, and Janssen. Dr. Takeshita is a former recipient of the National Psoriasis Foundation’s research fellowship in 2011-2012 and 2012-2013.
On Twitter @naseemsmiller
Despite advancements in therapies for psoriasis, a large proportion of patients are not treated or are receiving suboptimal treatment, according to analysis of a series of comprehensive patient surveys.
The analysis showed that nearly half of the patients with mild psoriasis were receiving no treatments at all in 2011, while 42% of them were treated only with topical agents. In other words, they were undertreated.
On a positive note, the proportion of patients with severe psoriasis who reported receiving no treatment dropped from 30% in the early 2000s to 9% in 2011. Still, 22% of them were undertreated and were prescribed topical medications alone (JAMA Dermatol. 2013 Aug. 14 [doi: 10.1001/jamadermatol.2013.5264]).
The study also revealed that many of the patients were dissatisfied with their treatment, highlighting "a call to action for [dermatologists] to actively seek feedback from psoriasis patients, and find out how their condition is being treated from their perspective," said Dr. April W. Armstrong, lead author of the study and associate professor of dermatology at the University of California, Davis.
"And since many new medications are coming up as well, we should be up to date on the data and literature, and be comfortable with using all sorts of different treatment modalities so that we can offer patients a wide range of treatment options and be able to individualize the treatments," Dr. Armstrong said in an interview.
Meanwhile, the type of health insurance can limit treatment options. In two of the surveys, patients were asked why they discontinued using biological agents. Lack of health insurance was among the top reasons.
"Hopefully, payers will pay attention and find a way to work with providers to make therapies more accessible," Dr. Armstrong said. "Many of our patients need systemic treatments, and I hope payers will pay attention and understand consequences."
Some estimates show that the cost of psoriasis is nearly $11 billion in the United States. But there’s a dearth of studies on patients’ perspectives and the extent to which patients are treated, the authors noted.
One source that has captured such data is the National Psoriasis Foundation’s (NPF’s) biannual surveys of its members. A 2007 study of the survey data was the first to show that as many as 40% of patients with moderate to severe psoriasis didn’t receive treatment (J. Am. Acad. Dermatol. 2007;57:957-62).
For their analysis, Dr. Armstrong and her colleagues examined 13 NPF surveys conducted during 2003-2011. More than 5,600 patients with psoriasis and/or psoriatic arthritis completed the surveys. They had a mean age of 50 years, and most were white.
The analysis showed that the proportion of patients with mild psoriasis who didn’t receive any treatment rose from 42% in the 2003-2005 period to 49% in 2011. The percentage of untreated patients with moderate psoriasis dropped from 36% to 24% during that period, and from 30% to 9% for patients with severe psoriasis.
Dr. Armstrong said there are several explanations for why patients go untreated. Psoriasis is chronic, and after going to one, two, or three doctors and not getting satisfactory results, the patients may give up, she said. "Some of the untreated patients might have sought help before and decided that nothing could be done, and resolved [themselves] to their situation."
Meanwhile, she expressed her concern with the proportion of patients who were undertreated. Close to 30% of patients with moderate psoriasis and 22% of those with severe psoriasis were treated only with topical agents, and the proportions were higher in 2011 than in the 2003-2005 period.
Patients said the top three reasons they used topical agents alone were because they had fewer adverse effects than other treatments, their disease wasn’t serious enough for other kinds of treatments, and their physician wouldn’t prescribe any other treatments.
"There is still much to be learned about exactly why psoriasis patients are undertreated," Dr. Junko Takeshita of the University of Pennsylvania said via e-mail.
But, "it is essential that patients be properly educated about the risks and benefits of various therapies so that they can make informed treatment choices. It is also important for physicians to be aware of and inform their psoriasis patients about the overall health implications of psoriasis itself (i.e., associations with cardiovascular and metabolic diseases as well as emerging associations with other comorbid diseases)," said Dr. Takeshita, who was not involved in the study.
The study also showed the most common forms of various treatment modalities, with ultraviolet B as the most common form of phototherapy, methotrexate as the top oral agent, and etanercept and adalimumab as the most common biological agents.
The authors said that the study had some limitations. The results may have underestimated the data in the general population, because NPF members are more involved in their health care. Also, severity of the disease at the time of the survey may not have been representative of the patients’ disease course, they noted.
"Going forward, it will be important for us to better understand why psoriasis patients are being undertreated and their reasons for treatment dissatisfaction and discontinuation so that, as physicians, we can provide better care to our psoriasis patients," Dr. Takeshita said.
Dr. Armstrong has received research grants or consultant honoraria from Abbott, Amgen, and Janssen. Dr. Takeshita is a former recipient of the National Psoriasis Foundation’s research fellowship in 2011-2012 and 2012-2013.
On Twitter @naseemsmiller
Despite advancements in therapies for psoriasis, a large proportion of patients are not treated or are receiving suboptimal treatment, according to analysis of a series of comprehensive patient surveys.
The analysis showed that nearly half of the patients with mild psoriasis were receiving no treatments at all in 2011, while 42% of them were treated only with topical agents. In other words, they were undertreated.
On a positive note, the proportion of patients with severe psoriasis who reported receiving no treatment dropped from 30% in the early 2000s to 9% in 2011. Still, 22% of them were undertreated and were prescribed topical medications alone (JAMA Dermatol. 2013 Aug. 14 [doi: 10.1001/jamadermatol.2013.5264]).
The study also revealed that many of the patients were dissatisfied with their treatment, highlighting "a call to action for [dermatologists] to actively seek feedback from psoriasis patients, and find out how their condition is being treated from their perspective," said Dr. April W. Armstrong, lead author of the study and associate professor of dermatology at the University of California, Davis.
"And since many new medications are coming up as well, we should be up to date on the data and literature, and be comfortable with using all sorts of different treatment modalities so that we can offer patients a wide range of treatment options and be able to individualize the treatments," Dr. Armstrong said in an interview.
Meanwhile, the type of health insurance can limit treatment options. In two of the surveys, patients were asked why they discontinued using biological agents. Lack of health insurance was among the top reasons.
"Hopefully, payers will pay attention and find a way to work with providers to make therapies more accessible," Dr. Armstrong said. "Many of our patients need systemic treatments, and I hope payers will pay attention and understand consequences."
Some estimates show that the cost of psoriasis is nearly $11 billion in the United States. But there’s a dearth of studies on patients’ perspectives and the extent to which patients are treated, the authors noted.
One source that has captured such data is the National Psoriasis Foundation’s (NPF’s) biannual surveys of its members. A 2007 study of the survey data was the first to show that as many as 40% of patients with moderate to severe psoriasis didn’t receive treatment (J. Am. Acad. Dermatol. 2007;57:957-62).
For their analysis, Dr. Armstrong and her colleagues examined 13 NPF surveys conducted during 2003-2011. More than 5,600 patients with psoriasis and/or psoriatic arthritis completed the surveys. They had a mean age of 50 years, and most were white.
The analysis showed that the proportion of patients with mild psoriasis who didn’t receive any treatment rose from 42% in the 2003-2005 period to 49% in 2011. The percentage of untreated patients with moderate psoriasis dropped from 36% to 24% during that period, and from 30% to 9% for patients with severe psoriasis.
Dr. Armstrong said there are several explanations for why patients go untreated. Psoriasis is chronic, and after going to one, two, or three doctors and not getting satisfactory results, the patients may give up, she said. "Some of the untreated patients might have sought help before and decided that nothing could be done, and resolved [themselves] to their situation."
Meanwhile, she expressed her concern with the proportion of patients who were undertreated. Close to 30% of patients with moderate psoriasis and 22% of those with severe psoriasis were treated only with topical agents, and the proportions were higher in 2011 than in the 2003-2005 period.
Patients said the top three reasons they used topical agents alone were because they had fewer adverse effects than other treatments, their disease wasn’t serious enough for other kinds of treatments, and their physician wouldn’t prescribe any other treatments.
"There is still much to be learned about exactly why psoriasis patients are undertreated," Dr. Junko Takeshita of the University of Pennsylvania said via e-mail.
But, "it is essential that patients be properly educated about the risks and benefits of various therapies so that they can make informed treatment choices. It is also important for physicians to be aware of and inform their psoriasis patients about the overall health implications of psoriasis itself (i.e., associations with cardiovascular and metabolic diseases as well as emerging associations with other comorbid diseases)," said Dr. Takeshita, who was not involved in the study.
The study also showed the most common forms of various treatment modalities, with ultraviolet B as the most common form of phototherapy, methotrexate as the top oral agent, and etanercept and adalimumab as the most common biological agents.
The authors said that the study had some limitations. The results may have underestimated the data in the general population, because NPF members are more involved in their health care. Also, severity of the disease at the time of the survey may not have been representative of the patients’ disease course, they noted.
"Going forward, it will be important for us to better understand why psoriasis patients are being undertreated and their reasons for treatment dissatisfaction and discontinuation so that, as physicians, we can provide better care to our psoriasis patients," Dr. Takeshita said.
Dr. Armstrong has received research grants or consultant honoraria from Abbott, Amgen, and Janssen. Dr. Takeshita is a former recipient of the National Psoriasis Foundation’s research fellowship in 2011-2012 and 2012-2013.
On Twitter @naseemsmiller
FROM JAMA DERMATOLOGY
Major finding: Nearly half of the patients with mild psoriasis were receiving no treatments at all in 2011, while 42% of them were treated with topical agents only.
Data source: 13 National Psoriasis Foundation surveys conducted between 2003 and 2011.
Disclosures: Dr. Armstrong has received research grants or consultant honoraria from Abbott, Amgen, and Janssen. Dr. Takeshita is a former recipient of the National Psoriasis Foundation’s research fellowship in 2011-2012 and 2012-2013.
Puffy fingers improves predictiveness of very early systemic sclerosis diagnosis
The presence of puffy fingers in patients with Raynaud’s phenomenon who are antinuclear antibody positive was further validated as an important sign of possible early systemic sclerosis in a recent study.
The findings support the value of using the European League Against Rheumatism Scleroderma Trial and Research Group’s (EUSTAR’s) new criteria of ANA positivity, Raynaud’s phenomenon, and puffy fingers as three red flags that raise suspicion for very early systemic sclerosis, reported Dr. Tünde Minier of the University of Pécs in Hungary and her colleagues (Ann. Rheum. Dis. 2013 Aug. 12 [doi: 10.1136/annrheumdis-2013-203716]). They calculated that the positive predictive value of ANA positivity for developing systemic sclerosis in patients with Raynaud’s phenomenon increased from 33.9% to 88.5% when combined with the presence of puffy fingers.
The researchers examined 469 patients with Raynaud’s phenomenon who were enrolled in the multicenter, prospective, observational Very Early Diagnosis of Systemic Sclerosis (VEDOSS) study at 33 EUSTAR centers throughout and outside Europe. About a third of the patients were ANA negative (32.2%), and 67.8% were ANA positive. Among the ANA-positive participants, 53.6% had a systemic sclerosis pattern on nailfold capillaroscopy, compared with only 13.4% of the ANA-negative patients (P less than .001).
Just over half of the ANA-negative patients lacked a systemic sclerosis pattern on nailfold capillaroscopy, systemic sclerosis–related specific clinical symptoms, or an erythrocyte sedimentation rate of 25 mm/hr or greater, and so were diagnosed with primary Raynaud’s.
The examinations of the patients also included assessments for digital ulcers, digital pitting scars, telangiectases, calcinosis, tendon friction rubs, esophageal symptoms, and symptoms consistent with median nerve compression syndrome.
The most common clinical features in the ANA-positive patients were previous or current puffy fingers, found in 38.5% of the ANA-positive patients versus 23.3% of the ANA-negative patients, and esophageal symptoms, identified in 35.2% of the ANA-positive patients and 18.4% of the ANA-negative patients.
"Almost 90% of ANA-positive Raynaud’s phenomenon patients with previous or current finger edematous skin changes (puffy fingers) already had a nailfold capillaroscopy systemic sclerosis pattern and/or systemic sclerosis–specific autoantibodies," Dr. Minier’s team wrote. Specifically, 73.3% of ANA-positive patients with Raynaud’s and puffy fingers had a nailfold capillaroscopy systemic sclerosis pattern, compared with only 41.2% of ANA-positive patients without puffy fingers. Overall, 88.5% of ANA-positive patients with Raynaud’s and puffy fingers met the criteria for very early systemic sclerosis.
The ANA-positive patients with puffy fingers were also more likely to have other symptoms than were the ANA-positive patients without puffy fingers. Sclerodactyly was identified in 17.8% of the ANA-positive patients with puffy fingers, compared with 6.2% of the ANA-positive patients without current or previous puffy fingers (P = .002). Telangiectases also appeared on 17.3% of the ANA-positive patients with puffy fingers, compared with 9.2% of those without puffy fingers (P = .033). Similarly, 42.1% of ANA-positive patients with puffy fingers had esophageal symptoms, compared with 30.9% of those without puffy fingers (P = .043).
The researchers also noted that even puffy fingers in ANA-negative patients may require more careful follow-up, because 17% of the ANA-negative patients with current or previous puffy fingers had a nailfold capillaroscopy systemic sclerosis pattern and 20% of the ANA-negative patients with puffy fingers had sclerodactyly with other systemic sclerosis symptoms.
The authors noted that one limitation of their study is the inability to generalize their findings to the broader population of Raynaud’s phenomenon patients seen by general physicians, since a higher percentage of the patients enrolled in this cohort meet very early systemic sclerosis classification.
"Patients identified with the very early diagnostic criteria may also have scleroderma with limited cutaneous involvement or even undifferentiated connective tissue disease," a limitation that longer-term follow-up data may clarify with a comparison of the ACR 1980 classification criteria and the new ACR-EULAR criteria.
The study did not use external funding. The authors had no disclosures.
* This story was updated 8/20/2013.
It is appreciated among rheumatologists that although
Raynaud’s phenomenon is common in the general population, it occurs more
commonly in patients with connective tissue disorders and in particular in those
with scleroderma. Recently, preliminary criteria have been proposed by the European
League Against Rheumatism (EULAR) Scleroderma Trials and Research Group to
identify very early systemic sclerosis. Three red-flag features were identified
as those which would raise suspicion for early scleroderma: Raynaud’s phenomenon,
puffy fingers, and a positive ANA. In Dr. Minier’s study, puffy fingers emerged
as an important feature suggesting a higher likelihood of systemic sclerosis.

|
| Dr. Robert Spiera |
It has already been appreciated that patients with Raynaud’s phenomenon who have a positive ANA have a higher likelihood of developing a
connective tissue disorder. In this cohort study, the presence of puffy fingers
in the past or at the time of evaluation markedly increased a positive
predictive value that scleroderma would be present or develop. Perhaps even
more important was their observation that among patients with Raynaud’s phenomenon
without detectable ANA (in whom the likelihood of a connective tissue disorder
evolving was traditionally felt to be low), if puffy fingers were present,
as many as 20% ultimately developed features suggestive of systemic sclerosis.
Rheumatologists have typically recognized the presence
of puffy fingers as potentially an important clinical finding, indicating a
likelihood of the patient having an inflammatory arthritis, connective tissue
disorder, or scleroderma. Of course there is a differential diagnosis of
puffy hands, including thyroid disease, edema, or other metabolic
abnormalities. Nevertheless, in the context of Raynaud’s phenomenon and
detectable ANA, vigilance for evolution toward a scleroderma spectrum disorder
is warranted.
Ultimately, the balance of disease criteria can be
difficult. More liberal diagnostic criteria allow the recognition and diagnosis
of the disorder in patients earlier in the disease course. That must be
balanced against the downside of establishing a diagnosis of a potentially dangerous
and debilitating rheumatic disease in a patient in whom it might not more fully
evolve. Ultimately, the very diagnosis itself could be a major cause of anxiety
and, indeed, morbidity to those patients.
The finding, however, of the importance of puffy
fingers as a predictive factor in establishing a diagnosis of systemic
sclerosis is important, and would be particularly of value if recognized in the
general medical community in terms of identifying patients worthy of further
investigation or rheumatologic referral.
Dr. Robert Spiera is a professor of clinical medicine at
Weill Cornell Medical College and director of the Scleroderma, Vasculitis and Myositis Center at the Hospital for Special Surgery in New York.
It is appreciated among rheumatologists that although
Raynaud’s phenomenon is common in the general population, it occurs more
commonly in patients with connective tissue disorders and in particular in those
with scleroderma. Recently, preliminary criteria have been proposed by the European
League Against Rheumatism (EULAR) Scleroderma Trials and Research Group to
identify very early systemic sclerosis. Three red-flag features were identified
as those which would raise suspicion for early scleroderma: Raynaud’s phenomenon,
puffy fingers, and a positive ANA. In Dr. Minier’s study, puffy fingers emerged
as an important feature suggesting a higher likelihood of systemic sclerosis.

|
| Dr. Robert Spiera |
It has already been appreciated that patients with Raynaud’s phenomenon who have a positive ANA have a higher likelihood of developing a
connective tissue disorder. In this cohort study, the presence of puffy fingers
in the past or at the time of evaluation markedly increased a positive
predictive value that scleroderma would be present or develop. Perhaps even
more important was their observation that among patients with Raynaud’s phenomenon
without detectable ANA (in whom the likelihood of a connective tissue disorder
evolving was traditionally felt to be low), if puffy fingers were present,
as many as 20% ultimately developed features suggestive of systemic sclerosis.
Rheumatologists have typically recognized the presence
of puffy fingers as potentially an important clinical finding, indicating a
likelihood of the patient having an inflammatory arthritis, connective tissue
disorder, or scleroderma. Of course there is a differential diagnosis of
puffy hands, including thyroid disease, edema, or other metabolic
abnormalities. Nevertheless, in the context of Raynaud’s phenomenon and
detectable ANA, vigilance for evolution toward a scleroderma spectrum disorder
is warranted.
Ultimately, the balance of disease criteria can be
difficult. More liberal diagnostic criteria allow the recognition and diagnosis
of the disorder in patients earlier in the disease course. That must be
balanced against the downside of establishing a diagnosis of a potentially dangerous
and debilitating rheumatic disease in a patient in whom it might not more fully
evolve. Ultimately, the very diagnosis itself could be a major cause of anxiety
and, indeed, morbidity to those patients.
The finding, however, of the importance of puffy
fingers as a predictive factor in establishing a diagnosis of systemic
sclerosis is important, and would be particularly of value if recognized in the
general medical community in terms of identifying patients worthy of further
investigation or rheumatologic referral.
Dr. Robert Spiera is a professor of clinical medicine at
Weill Cornell Medical College and director of the Scleroderma, Vasculitis and Myositis Center at the Hospital for Special Surgery in New York.
It is appreciated among rheumatologists that although
Raynaud’s phenomenon is common in the general population, it occurs more
commonly in patients with connective tissue disorders and in particular in those
with scleroderma. Recently, preliminary criteria have been proposed by the European
League Against Rheumatism (EULAR) Scleroderma Trials and Research Group to
identify very early systemic sclerosis. Three red-flag features were identified
as those which would raise suspicion for early scleroderma: Raynaud’s phenomenon,
puffy fingers, and a positive ANA. In Dr. Minier’s study, puffy fingers emerged
as an important feature suggesting a higher likelihood of systemic sclerosis.

|
| Dr. Robert Spiera |
It has already been appreciated that patients with Raynaud’s phenomenon who have a positive ANA have a higher likelihood of developing a
connective tissue disorder. In this cohort study, the presence of puffy fingers
in the past or at the time of evaluation markedly increased a positive
predictive value that scleroderma would be present or develop. Perhaps even
more important was their observation that among patients with Raynaud’s phenomenon
without detectable ANA (in whom the likelihood of a connective tissue disorder
evolving was traditionally felt to be low), if puffy fingers were present,
as many as 20% ultimately developed features suggestive of systemic sclerosis.
Rheumatologists have typically recognized the presence
of puffy fingers as potentially an important clinical finding, indicating a
likelihood of the patient having an inflammatory arthritis, connective tissue
disorder, or scleroderma. Of course there is a differential diagnosis of
puffy hands, including thyroid disease, edema, or other metabolic
abnormalities. Nevertheless, in the context of Raynaud’s phenomenon and
detectable ANA, vigilance for evolution toward a scleroderma spectrum disorder
is warranted.
Ultimately, the balance of disease criteria can be
difficult. More liberal diagnostic criteria allow the recognition and diagnosis
of the disorder in patients earlier in the disease course. That must be
balanced against the downside of establishing a diagnosis of a potentially dangerous
and debilitating rheumatic disease in a patient in whom it might not more fully
evolve. Ultimately, the very diagnosis itself could be a major cause of anxiety
and, indeed, morbidity to those patients.
The finding, however, of the importance of puffy
fingers as a predictive factor in establishing a diagnosis of systemic
sclerosis is important, and would be particularly of value if recognized in the
general medical community in terms of identifying patients worthy of further
investigation or rheumatologic referral.
Dr. Robert Spiera is a professor of clinical medicine at
Weill Cornell Medical College and director of the Scleroderma, Vasculitis and Myositis Center at the Hospital for Special Surgery in New York.
The presence of puffy fingers in patients with Raynaud’s phenomenon who are antinuclear antibody positive was further validated as an important sign of possible early systemic sclerosis in a recent study.
The findings support the value of using the European League Against Rheumatism Scleroderma Trial and Research Group’s (EUSTAR’s) new criteria of ANA positivity, Raynaud’s phenomenon, and puffy fingers as three red flags that raise suspicion for very early systemic sclerosis, reported Dr. Tünde Minier of the University of Pécs in Hungary and her colleagues (Ann. Rheum. Dis. 2013 Aug. 12 [doi: 10.1136/annrheumdis-2013-203716]). They calculated that the positive predictive value of ANA positivity for developing systemic sclerosis in patients with Raynaud’s phenomenon increased from 33.9% to 88.5% when combined with the presence of puffy fingers.
The researchers examined 469 patients with Raynaud’s phenomenon who were enrolled in the multicenter, prospective, observational Very Early Diagnosis of Systemic Sclerosis (VEDOSS) study at 33 EUSTAR centers throughout and outside Europe. About a third of the patients were ANA negative (32.2%), and 67.8% were ANA positive. Among the ANA-positive participants, 53.6% had a systemic sclerosis pattern on nailfold capillaroscopy, compared with only 13.4% of the ANA-negative patients (P less than .001).
Just over half of the ANA-negative patients lacked a systemic sclerosis pattern on nailfold capillaroscopy, systemic sclerosis–related specific clinical symptoms, or an erythrocyte sedimentation rate of 25 mm/hr or greater, and so were diagnosed with primary Raynaud’s.
The examinations of the patients also included assessments for digital ulcers, digital pitting scars, telangiectases, calcinosis, tendon friction rubs, esophageal symptoms, and symptoms consistent with median nerve compression syndrome.
The most common clinical features in the ANA-positive patients were previous or current puffy fingers, found in 38.5% of the ANA-positive patients versus 23.3% of the ANA-negative patients, and esophageal symptoms, identified in 35.2% of the ANA-positive patients and 18.4% of the ANA-negative patients.
"Almost 90% of ANA-positive Raynaud’s phenomenon patients with previous or current finger edematous skin changes (puffy fingers) already had a nailfold capillaroscopy systemic sclerosis pattern and/or systemic sclerosis–specific autoantibodies," Dr. Minier’s team wrote. Specifically, 73.3% of ANA-positive patients with Raynaud’s and puffy fingers had a nailfold capillaroscopy systemic sclerosis pattern, compared with only 41.2% of ANA-positive patients without puffy fingers. Overall, 88.5% of ANA-positive patients with Raynaud’s and puffy fingers met the criteria for very early systemic sclerosis.
The ANA-positive patients with puffy fingers were also more likely to have other symptoms than were the ANA-positive patients without puffy fingers. Sclerodactyly was identified in 17.8% of the ANA-positive patients with puffy fingers, compared with 6.2% of the ANA-positive patients without current or previous puffy fingers (P = .002). Telangiectases also appeared on 17.3% of the ANA-positive patients with puffy fingers, compared with 9.2% of those without puffy fingers (P = .033). Similarly, 42.1% of ANA-positive patients with puffy fingers had esophageal symptoms, compared with 30.9% of those without puffy fingers (P = .043).
The researchers also noted that even puffy fingers in ANA-negative patients may require more careful follow-up, because 17% of the ANA-negative patients with current or previous puffy fingers had a nailfold capillaroscopy systemic sclerosis pattern and 20% of the ANA-negative patients with puffy fingers had sclerodactyly with other systemic sclerosis symptoms.
The authors noted that one limitation of their study is the inability to generalize their findings to the broader population of Raynaud’s phenomenon patients seen by general physicians, since a higher percentage of the patients enrolled in this cohort meet very early systemic sclerosis classification.
"Patients identified with the very early diagnostic criteria may also have scleroderma with limited cutaneous involvement or even undifferentiated connective tissue disease," a limitation that longer-term follow-up data may clarify with a comparison of the ACR 1980 classification criteria and the new ACR-EULAR criteria.
The study did not use external funding. The authors had no disclosures.
* This story was updated 8/20/2013.
The presence of puffy fingers in patients with Raynaud’s phenomenon who are antinuclear antibody positive was further validated as an important sign of possible early systemic sclerosis in a recent study.
The findings support the value of using the European League Against Rheumatism Scleroderma Trial and Research Group’s (EUSTAR’s) new criteria of ANA positivity, Raynaud’s phenomenon, and puffy fingers as three red flags that raise suspicion for very early systemic sclerosis, reported Dr. Tünde Minier of the University of Pécs in Hungary and her colleagues (Ann. Rheum. Dis. 2013 Aug. 12 [doi: 10.1136/annrheumdis-2013-203716]). They calculated that the positive predictive value of ANA positivity for developing systemic sclerosis in patients with Raynaud’s phenomenon increased from 33.9% to 88.5% when combined with the presence of puffy fingers.
The researchers examined 469 patients with Raynaud’s phenomenon who were enrolled in the multicenter, prospective, observational Very Early Diagnosis of Systemic Sclerosis (VEDOSS) study at 33 EUSTAR centers throughout and outside Europe. About a third of the patients were ANA negative (32.2%), and 67.8% were ANA positive. Among the ANA-positive participants, 53.6% had a systemic sclerosis pattern on nailfold capillaroscopy, compared with only 13.4% of the ANA-negative patients (P less than .001).
Just over half of the ANA-negative patients lacked a systemic sclerosis pattern on nailfold capillaroscopy, systemic sclerosis–related specific clinical symptoms, or an erythrocyte sedimentation rate of 25 mm/hr or greater, and so were diagnosed with primary Raynaud’s.
The examinations of the patients also included assessments for digital ulcers, digital pitting scars, telangiectases, calcinosis, tendon friction rubs, esophageal symptoms, and symptoms consistent with median nerve compression syndrome.
The most common clinical features in the ANA-positive patients were previous or current puffy fingers, found in 38.5% of the ANA-positive patients versus 23.3% of the ANA-negative patients, and esophageal symptoms, identified in 35.2% of the ANA-positive patients and 18.4% of the ANA-negative patients.
"Almost 90% of ANA-positive Raynaud’s phenomenon patients with previous or current finger edematous skin changes (puffy fingers) already had a nailfold capillaroscopy systemic sclerosis pattern and/or systemic sclerosis–specific autoantibodies," Dr. Minier’s team wrote. Specifically, 73.3% of ANA-positive patients with Raynaud’s and puffy fingers had a nailfold capillaroscopy systemic sclerosis pattern, compared with only 41.2% of ANA-positive patients without puffy fingers. Overall, 88.5% of ANA-positive patients with Raynaud’s and puffy fingers met the criteria for very early systemic sclerosis.
The ANA-positive patients with puffy fingers were also more likely to have other symptoms than were the ANA-positive patients without puffy fingers. Sclerodactyly was identified in 17.8% of the ANA-positive patients with puffy fingers, compared with 6.2% of the ANA-positive patients without current or previous puffy fingers (P = .002). Telangiectases also appeared on 17.3% of the ANA-positive patients with puffy fingers, compared with 9.2% of those without puffy fingers (P = .033). Similarly, 42.1% of ANA-positive patients with puffy fingers had esophageal symptoms, compared with 30.9% of those without puffy fingers (P = .043).
The researchers also noted that even puffy fingers in ANA-negative patients may require more careful follow-up, because 17% of the ANA-negative patients with current or previous puffy fingers had a nailfold capillaroscopy systemic sclerosis pattern and 20% of the ANA-negative patients with puffy fingers had sclerodactyly with other systemic sclerosis symptoms.
The authors noted that one limitation of their study is the inability to generalize their findings to the broader population of Raynaud’s phenomenon patients seen by general physicians, since a higher percentage of the patients enrolled in this cohort meet very early systemic sclerosis classification.
"Patients identified with the very early diagnostic criteria may also have scleroderma with limited cutaneous involvement or even undifferentiated connective tissue disease," a limitation that longer-term follow-up data may clarify with a comparison of the ACR 1980 classification criteria and the new ACR-EULAR criteria.
The study did not use external funding. The authors had no disclosures.
* This story was updated 8/20/2013.
FROM ANNALS OF THE RHEUMATIC DISEASES
Major finding: Puffy fingers were identified in more Raynaud's phenomenon patients who were antinuclear antibody positive (38.5%) than ANA negative (23.3%, P less than .01).
Data source: The findings are based on an analysis of signs and symptoms in 469 Reynaud’s phenomenon patients enrolled in the Very Early Diagnosis of Systemic Sclerosis cohort from 33 EUSTAR centers throughout and outside Europe.
Disclosures: Information on the study’s funding was unavailable. The authors had no disclosures.
Venous eczema and lipodermatosclerosis common in venous insufficiency
Venous eczema and lipodermatosclerosis should be considered in patients with painful dermatitis or pruritic erythematous eruptions on their lower extremities, especially if there are other signs of venous insufficiency, according to Dr. Laurel M. Morton and Dr. Tania J. Phillips.
"Venous eczema and lipodermatosclerosis are relatively common conditions caused by chronic venous insufficiency, yet at times they can be a challenge to diagnose and treat," said Dr. Morton in an interview.
Writing in the September issue of Seminars in Cutaneous Medicine and Surgery, Dr. Morton and Dr. Phillips of the department of dermatology, Boston University, reviewed the challenges associated with the diagnosis and treatment of these conditions (Semin. Cutan. Med. Surg. 2013;32:169-76).
Venous eczema presents as erythematous, scaly, pruritic skin on the lower legs and ankles, often in association with other signs of venous disease such as varicose veins, edema, hemosiderin pigmentation, atrophie blanche, and lipodermatosclerosis.
Lipodermatosclerosis is a progressive fibrotic process affecting the dermis and subcutaneous fat of the lower leg, resulting in hyperpigmentation and induration.
There are few data on the prevalence of venous eczema and lipodermatosclerosis, or even for chronic venous disease in general, the researchers noted. However, they suggested that as many as 17% of men and 40% of women suffer from chronic venous insufficiency, and of the 23% of Americans with varicose veins, 2 million will develop skin changes.
"Perhaps the most challenging condition to rule out is allergic contact dermatitis, since this may be seen in conjunction with venous eczema, which is characterized by a decreased skin barrier that may increase the rate of sensitization."
Venous eczema can be confused with other papulosquamous conditions such as nummular eczema and psoriasis, as well as xerosis, eczema craquele, and cellulitis.
"Perhaps the most challenging condition to rule out is allergic contact dermatitis, since this may be seen in conjunction with venous eczema, which is characterized by a decreased skin barrier that may increase the rate of sensitization," the researchers reported.
Consider patch testing in cases where allergy is suspected and, if irritant contact dermatitis is a possibility, patients should be asked about topical applications, they added.
Acute lipodermatosclerosis presents as painful, erythematous, purple indurated plaques – well demarcated from normal skin – confined to the lower extremity, possibly with white scale, while chronic lipodermatosclerosis is generally associated with a classic "inverted champagne bottle" appearance of the distal third of the lower leg.
Acute lipodermatosclerosis is also often misdiagnosed as cellulitis, although a key distinguishing factor is lack of improvement with antibiotics. Acute lipodermatosclerosis also can be confused with conditions such as erythema nodosum, thrombophlebitis, and fibrosing conditions such as inflammatory morphea.
As venous eczema and lipodermatosclerosis are caused by venous insufficiency, the authors argue that compression should be the first line of treatment.
"The most important treatment for both conditions is graduated compression, but oral, topical, and surgical interventions should be considered as adjunctive approaches," Dr. Morton said.
Venous eczema also can be managed topically with emollients and immunomodulators – including corticosteroids and calcineurin inhibitors – although scant data support this approach.
However, the researchers highlighted one study in which patients treated with a combination of oral doxycycline and topical tacrolimus showed statistically significant improvement in pain, edema, erythema, pigmentation, pruritus, and exudate (Indian J. Pharmacol. 2012;44:111-13).
Patients with lipodermatosclerosis may not tolerate compression due to extreme skin tenderness; however, there is reasonable evidence in favor of stanozolol, an anabolic steroid with fibrinolytic properties, the researchers noted. Other agents that may help these patients include danazol, oxandrolone, pentoxifylline, and intralesional triamcinolone.
There is little evidence to support the use of ultrasound, the researchers said. However, it is safe and may offer an alternative to other therapeutic options in recalcitrant disease, they added.
The researchers had no financial conflicts to disclose.
Venous eczema and lipodermatosclerosis should be considered in patients with painful dermatitis or pruritic erythematous eruptions on their lower extremities, especially if there are other signs of venous insufficiency, according to Dr. Laurel M. Morton and Dr. Tania J. Phillips.
"Venous eczema and lipodermatosclerosis are relatively common conditions caused by chronic venous insufficiency, yet at times they can be a challenge to diagnose and treat," said Dr. Morton in an interview.
Writing in the September issue of Seminars in Cutaneous Medicine and Surgery, Dr. Morton and Dr. Phillips of the department of dermatology, Boston University, reviewed the challenges associated with the diagnosis and treatment of these conditions (Semin. Cutan. Med. Surg. 2013;32:169-76).
Venous eczema presents as erythematous, scaly, pruritic skin on the lower legs and ankles, often in association with other signs of venous disease such as varicose veins, edema, hemosiderin pigmentation, atrophie blanche, and lipodermatosclerosis.
Lipodermatosclerosis is a progressive fibrotic process affecting the dermis and subcutaneous fat of the lower leg, resulting in hyperpigmentation and induration.
There are few data on the prevalence of venous eczema and lipodermatosclerosis, or even for chronic venous disease in general, the researchers noted. However, they suggested that as many as 17% of men and 40% of women suffer from chronic venous insufficiency, and of the 23% of Americans with varicose veins, 2 million will develop skin changes.
"Perhaps the most challenging condition to rule out is allergic contact dermatitis, since this may be seen in conjunction with venous eczema, which is characterized by a decreased skin barrier that may increase the rate of sensitization."
Venous eczema can be confused with other papulosquamous conditions such as nummular eczema and psoriasis, as well as xerosis, eczema craquele, and cellulitis.
"Perhaps the most challenging condition to rule out is allergic contact dermatitis, since this may be seen in conjunction with venous eczema, which is characterized by a decreased skin barrier that may increase the rate of sensitization," the researchers reported.
Consider patch testing in cases where allergy is suspected and, if irritant contact dermatitis is a possibility, patients should be asked about topical applications, they added.
Acute lipodermatosclerosis presents as painful, erythematous, purple indurated plaques – well demarcated from normal skin – confined to the lower extremity, possibly with white scale, while chronic lipodermatosclerosis is generally associated with a classic "inverted champagne bottle" appearance of the distal third of the lower leg.
Acute lipodermatosclerosis is also often misdiagnosed as cellulitis, although a key distinguishing factor is lack of improvement with antibiotics. Acute lipodermatosclerosis also can be confused with conditions such as erythema nodosum, thrombophlebitis, and fibrosing conditions such as inflammatory morphea.
As venous eczema and lipodermatosclerosis are caused by venous insufficiency, the authors argue that compression should be the first line of treatment.
"The most important treatment for both conditions is graduated compression, but oral, topical, and surgical interventions should be considered as adjunctive approaches," Dr. Morton said.
Venous eczema also can be managed topically with emollients and immunomodulators – including corticosteroids and calcineurin inhibitors – although scant data support this approach.
However, the researchers highlighted one study in which patients treated with a combination of oral doxycycline and topical tacrolimus showed statistically significant improvement in pain, edema, erythema, pigmentation, pruritus, and exudate (Indian J. Pharmacol. 2012;44:111-13).
Patients with lipodermatosclerosis may not tolerate compression due to extreme skin tenderness; however, there is reasonable evidence in favor of stanozolol, an anabolic steroid with fibrinolytic properties, the researchers noted. Other agents that may help these patients include danazol, oxandrolone, pentoxifylline, and intralesional triamcinolone.
There is little evidence to support the use of ultrasound, the researchers said. However, it is safe and may offer an alternative to other therapeutic options in recalcitrant disease, they added.
The researchers had no financial conflicts to disclose.
Venous eczema and lipodermatosclerosis should be considered in patients with painful dermatitis or pruritic erythematous eruptions on their lower extremities, especially if there are other signs of venous insufficiency, according to Dr. Laurel M. Morton and Dr. Tania J. Phillips.
"Venous eczema and lipodermatosclerosis are relatively common conditions caused by chronic venous insufficiency, yet at times they can be a challenge to diagnose and treat," said Dr. Morton in an interview.
Writing in the September issue of Seminars in Cutaneous Medicine and Surgery, Dr. Morton and Dr. Phillips of the department of dermatology, Boston University, reviewed the challenges associated with the diagnosis and treatment of these conditions (Semin. Cutan. Med. Surg. 2013;32:169-76).
Venous eczema presents as erythematous, scaly, pruritic skin on the lower legs and ankles, often in association with other signs of venous disease such as varicose veins, edema, hemosiderin pigmentation, atrophie blanche, and lipodermatosclerosis.
Lipodermatosclerosis is a progressive fibrotic process affecting the dermis and subcutaneous fat of the lower leg, resulting in hyperpigmentation and induration.
There are few data on the prevalence of venous eczema and lipodermatosclerosis, or even for chronic venous disease in general, the researchers noted. However, they suggested that as many as 17% of men and 40% of women suffer from chronic venous insufficiency, and of the 23% of Americans with varicose veins, 2 million will develop skin changes.
"Perhaps the most challenging condition to rule out is allergic contact dermatitis, since this may be seen in conjunction with venous eczema, which is characterized by a decreased skin barrier that may increase the rate of sensitization."
Venous eczema can be confused with other papulosquamous conditions such as nummular eczema and psoriasis, as well as xerosis, eczema craquele, and cellulitis.
"Perhaps the most challenging condition to rule out is allergic contact dermatitis, since this may be seen in conjunction with venous eczema, which is characterized by a decreased skin barrier that may increase the rate of sensitization," the researchers reported.
Consider patch testing in cases where allergy is suspected and, if irritant contact dermatitis is a possibility, patients should be asked about topical applications, they added.
Acute lipodermatosclerosis presents as painful, erythematous, purple indurated plaques – well demarcated from normal skin – confined to the lower extremity, possibly with white scale, while chronic lipodermatosclerosis is generally associated with a classic "inverted champagne bottle" appearance of the distal third of the lower leg.
Acute lipodermatosclerosis is also often misdiagnosed as cellulitis, although a key distinguishing factor is lack of improvement with antibiotics. Acute lipodermatosclerosis also can be confused with conditions such as erythema nodosum, thrombophlebitis, and fibrosing conditions such as inflammatory morphea.
As venous eczema and lipodermatosclerosis are caused by venous insufficiency, the authors argue that compression should be the first line of treatment.
"The most important treatment for both conditions is graduated compression, but oral, topical, and surgical interventions should be considered as adjunctive approaches," Dr. Morton said.
Venous eczema also can be managed topically with emollients and immunomodulators – including corticosteroids and calcineurin inhibitors – although scant data support this approach.
However, the researchers highlighted one study in which patients treated with a combination of oral doxycycline and topical tacrolimus showed statistically significant improvement in pain, edema, erythema, pigmentation, pruritus, and exudate (Indian J. Pharmacol. 2012;44:111-13).
Patients with lipodermatosclerosis may not tolerate compression due to extreme skin tenderness; however, there is reasonable evidence in favor of stanozolol, an anabolic steroid with fibrinolytic properties, the researchers noted. Other agents that may help these patients include danazol, oxandrolone, pentoxifylline, and intralesional triamcinolone.
There is little evidence to support the use of ultrasound, the researchers said. However, it is safe and may offer an alternative to other therapeutic options in recalcitrant disease, they added.
The researchers had no financial conflicts to disclose.
FROM SEMINARS IN CUTANEOUS MEDICINE AND SURGERY
The chicken or the egg: Obesity or psoriasis?
MILWAUKEE – Excess adiposity occurred prior to psoriasis in 93% of 29 overweight and obese psoriatic children, in a chart review examining the relationship between psoriasis and obesity.
Further, 78% of patients were obese before developing psoriasis, Dr. Lauren Becker reported at the annual meeting of the Society for Pediatric Dermatology.
Although the authors anticipated that most of the children would show increased adiposity before psoriasis based on clinical observations, "none of us predicted that it would be 27 of 29," she said in an interview. "It really was incredible, and even the ones who were normal weight became overweight or obese within a year of their psoriasis."
The review was sparked by a recent international study, led by senior author Dr. Amy Paller of Northwestern University in Chicago, in which 38% of psoriatic children had excess central adiposity (high waist-to-height ratio) compared with 21% of controls. The odds of obesity were also several times higher than those reported for adults with psoriasis, jumping 4.29-fold overall in psoriatic children versus controls, 4.92-fold in those with severe versus mild psoriasis, and 7.6-fold in the United States. (JAMA Dermatol. 2013;149:166-76).
Although other studies in children support an association between obesity and psoriasis, the international study was the first to measure central adiposity, a more sensitive indicator of cardiovascular risk in children than BMI, Dr. Kelly Cordoro remarked in a separate lecture on the comorbidities of pediatric psoriasis at the meeting.
"Adiposity, hypertension, hyperlipidemia, and diabetes are increased in prevalence among pediatric psoriasis patients, but the obesity association is the strongest," she said. "It’s global, and the effect is most pronounced in the United States, where in particular, central obesity is highest and has the accompanying higher cardiovascular risk."
Previous research has shown that obesity is strongly correlated with psoriasis in adults. A recent meta-analysis showed that psoriatic adults were more likely to be obese than those without psoriasis (pooled odds ratio, 1.66), and that the odds were even higher in patients with severe versus mild psoriasis (OR, 2.23, vs. OR, 1.46) (Nutr. Diabetes 2012 ;2:e54).
Both clinicians observed that psoriasis and obesity are chronic inflammatory states, marked by overexpression of circulating proinflammatory cytokines derived from Th1 and Th17 lymphocyte subsets, as well as tumor necrosis factor–alpha and adipokines. In addition, a fat cell is a microenvironment of inflammation, with adipose tissue releasing proinflammatory cytokines such as interleukin-6 and TNF-alpha.
The two diseases drive one another, but the exact relationship remains unclear, said Dr. Becker, a pediatrician and dermatology fellow at Northwestern at the time of the study. In addition, the impact on quality of life and social interactions of being both obese or overweight and having psoriasis may predispose patients to mental and physical health problems.
"Children with excess adiposity or with the highly visible lesions of psoriasis are more often teased or bullied, which may contribute to the tendency to become socially isolated, decrease physical activity, and increase eating," she said. "It really is a vicious circle."
Although the data are clear that psoriasis and obesity are associated, the current chart review suggesting that obesity precedes psoriasis is too small to definitively answer the question, Dr. Cordoro, a pediatric dermatologist at the University of California, San Francisco, said in an interview.
Dr. Becker said obesity clearly came before psoriasis in their study cohort, but agreed that further study is needed to confirm the results. She also stressed the need for biomarker analyses to identify overweight/obese children who are at risk for psoriasis, and studies to address whether weight loss can reverse the severity of pediatric psoriasis.
For example, data from a study of overweight psoriatic adults showed that eating a low-calorie diet improved psoriasis severity and quality of life after 4 months (JAMA Dermatol. 2013;149:795-801).
However, in the absence of adequate data, clinicians should consider metabolic testing to determine whether their overweight pediatric patients are on the path to the metabolic syndrome, Dr. Becker and Dr. Cordoro suggested. Metabolic testing was not performed on any of the 29 children in the chart review, although 48% had a family history of hyperlipidemia and 45% had a family history of obesity.
The average age of the children was 12.6 years; the average age of onset of obesity was 4 years (range, 2-12 years), and the average age of onset of psoriasis was 9 years (range, 2-14 years).
Two obese patients were able to reduce their BMIs from obese to overweight status 1 year after being diagnosed with psoriasis; however, both have remained in the 85th-95th BMI percentile for more than 2 years, Dr. Becker said. Both of the children who had a normal BMI prior to their psoriasis had a BMI in the overweight or obese range within 1 year after diagnosis, Dr. Becker noted.
Dr. Cordoro also made an impassioned plea for clinicians to address the significant psychosocial comorbidity present in psoriatic children.
"You can think about obesity, psoriasis, and depression as being reciprocal exacerbating factors, such that each triggers the other and represents an insult to self-esteem and the overall well-being of these patients," she said. "They end up having high stress levels and really dismal quality of life. These issues are as important in the management of the child as the medical considerations."
Dr. Becker, her coauthors, and Dr. Cordoro reported having no relevant financial disclosures.
MILWAUKEE – Excess adiposity occurred prior to psoriasis in 93% of 29 overweight and obese psoriatic children, in a chart review examining the relationship between psoriasis and obesity.
Further, 78% of patients were obese before developing psoriasis, Dr. Lauren Becker reported at the annual meeting of the Society for Pediatric Dermatology.
Although the authors anticipated that most of the children would show increased adiposity before psoriasis based on clinical observations, "none of us predicted that it would be 27 of 29," she said in an interview. "It really was incredible, and even the ones who were normal weight became overweight or obese within a year of their psoriasis."
The review was sparked by a recent international study, led by senior author Dr. Amy Paller of Northwestern University in Chicago, in which 38% of psoriatic children had excess central adiposity (high waist-to-height ratio) compared with 21% of controls. The odds of obesity were also several times higher than those reported for adults with psoriasis, jumping 4.29-fold overall in psoriatic children versus controls, 4.92-fold in those with severe versus mild psoriasis, and 7.6-fold in the United States. (JAMA Dermatol. 2013;149:166-76).
Although other studies in children support an association between obesity and psoriasis, the international study was the first to measure central adiposity, a more sensitive indicator of cardiovascular risk in children than BMI, Dr. Kelly Cordoro remarked in a separate lecture on the comorbidities of pediatric psoriasis at the meeting.
"Adiposity, hypertension, hyperlipidemia, and diabetes are increased in prevalence among pediatric psoriasis patients, but the obesity association is the strongest," she said. "It’s global, and the effect is most pronounced in the United States, where in particular, central obesity is highest and has the accompanying higher cardiovascular risk."
Previous research has shown that obesity is strongly correlated with psoriasis in adults. A recent meta-analysis showed that psoriatic adults were more likely to be obese than those without psoriasis (pooled odds ratio, 1.66), and that the odds were even higher in patients with severe versus mild psoriasis (OR, 2.23, vs. OR, 1.46) (Nutr. Diabetes 2012 ;2:e54).
Both clinicians observed that psoriasis and obesity are chronic inflammatory states, marked by overexpression of circulating proinflammatory cytokines derived from Th1 and Th17 lymphocyte subsets, as well as tumor necrosis factor–alpha and adipokines. In addition, a fat cell is a microenvironment of inflammation, with adipose tissue releasing proinflammatory cytokines such as interleukin-6 and TNF-alpha.
The two diseases drive one another, but the exact relationship remains unclear, said Dr. Becker, a pediatrician and dermatology fellow at Northwestern at the time of the study. In addition, the impact on quality of life and social interactions of being both obese or overweight and having psoriasis may predispose patients to mental and physical health problems.
"Children with excess adiposity or with the highly visible lesions of psoriasis are more often teased or bullied, which may contribute to the tendency to become socially isolated, decrease physical activity, and increase eating," she said. "It really is a vicious circle."
Although the data are clear that psoriasis and obesity are associated, the current chart review suggesting that obesity precedes psoriasis is too small to definitively answer the question, Dr. Cordoro, a pediatric dermatologist at the University of California, San Francisco, said in an interview.
Dr. Becker said obesity clearly came before psoriasis in their study cohort, but agreed that further study is needed to confirm the results. She also stressed the need for biomarker analyses to identify overweight/obese children who are at risk for psoriasis, and studies to address whether weight loss can reverse the severity of pediatric psoriasis.
For example, data from a study of overweight psoriatic adults showed that eating a low-calorie diet improved psoriasis severity and quality of life after 4 months (JAMA Dermatol. 2013;149:795-801).
However, in the absence of adequate data, clinicians should consider metabolic testing to determine whether their overweight pediatric patients are on the path to the metabolic syndrome, Dr. Becker and Dr. Cordoro suggested. Metabolic testing was not performed on any of the 29 children in the chart review, although 48% had a family history of hyperlipidemia and 45% had a family history of obesity.
The average age of the children was 12.6 years; the average age of onset of obesity was 4 years (range, 2-12 years), and the average age of onset of psoriasis was 9 years (range, 2-14 years).
Two obese patients were able to reduce their BMIs from obese to overweight status 1 year after being diagnosed with psoriasis; however, both have remained in the 85th-95th BMI percentile for more than 2 years, Dr. Becker said. Both of the children who had a normal BMI prior to their psoriasis had a BMI in the overweight or obese range within 1 year after diagnosis, Dr. Becker noted.
Dr. Cordoro also made an impassioned plea for clinicians to address the significant psychosocial comorbidity present in psoriatic children.
"You can think about obesity, psoriasis, and depression as being reciprocal exacerbating factors, such that each triggers the other and represents an insult to self-esteem and the overall well-being of these patients," she said. "They end up having high stress levels and really dismal quality of life. These issues are as important in the management of the child as the medical considerations."
Dr. Becker, her coauthors, and Dr. Cordoro reported having no relevant financial disclosures.
MILWAUKEE – Excess adiposity occurred prior to psoriasis in 93% of 29 overweight and obese psoriatic children, in a chart review examining the relationship between psoriasis and obesity.
Further, 78% of patients were obese before developing psoriasis, Dr. Lauren Becker reported at the annual meeting of the Society for Pediatric Dermatology.
Although the authors anticipated that most of the children would show increased adiposity before psoriasis based on clinical observations, "none of us predicted that it would be 27 of 29," she said in an interview. "It really was incredible, and even the ones who were normal weight became overweight or obese within a year of their psoriasis."
The review was sparked by a recent international study, led by senior author Dr. Amy Paller of Northwestern University in Chicago, in which 38% of psoriatic children had excess central adiposity (high waist-to-height ratio) compared with 21% of controls. The odds of obesity were also several times higher than those reported for adults with psoriasis, jumping 4.29-fold overall in psoriatic children versus controls, 4.92-fold in those with severe versus mild psoriasis, and 7.6-fold in the United States. (JAMA Dermatol. 2013;149:166-76).
Although other studies in children support an association between obesity and psoriasis, the international study was the first to measure central adiposity, a more sensitive indicator of cardiovascular risk in children than BMI, Dr. Kelly Cordoro remarked in a separate lecture on the comorbidities of pediatric psoriasis at the meeting.
"Adiposity, hypertension, hyperlipidemia, and diabetes are increased in prevalence among pediatric psoriasis patients, but the obesity association is the strongest," she said. "It’s global, and the effect is most pronounced in the United States, where in particular, central obesity is highest and has the accompanying higher cardiovascular risk."
Previous research has shown that obesity is strongly correlated with psoriasis in adults. A recent meta-analysis showed that psoriatic adults were more likely to be obese than those without psoriasis (pooled odds ratio, 1.66), and that the odds were even higher in patients with severe versus mild psoriasis (OR, 2.23, vs. OR, 1.46) (Nutr. Diabetes 2012 ;2:e54).
Both clinicians observed that psoriasis and obesity are chronic inflammatory states, marked by overexpression of circulating proinflammatory cytokines derived from Th1 and Th17 lymphocyte subsets, as well as tumor necrosis factor–alpha and adipokines. In addition, a fat cell is a microenvironment of inflammation, with adipose tissue releasing proinflammatory cytokines such as interleukin-6 and TNF-alpha.
The two diseases drive one another, but the exact relationship remains unclear, said Dr. Becker, a pediatrician and dermatology fellow at Northwestern at the time of the study. In addition, the impact on quality of life and social interactions of being both obese or overweight and having psoriasis may predispose patients to mental and physical health problems.
"Children with excess adiposity or with the highly visible lesions of psoriasis are more often teased or bullied, which may contribute to the tendency to become socially isolated, decrease physical activity, and increase eating," she said. "It really is a vicious circle."
Although the data are clear that psoriasis and obesity are associated, the current chart review suggesting that obesity precedes psoriasis is too small to definitively answer the question, Dr. Cordoro, a pediatric dermatologist at the University of California, San Francisco, said in an interview.
Dr. Becker said obesity clearly came before psoriasis in their study cohort, but agreed that further study is needed to confirm the results. She also stressed the need for biomarker analyses to identify overweight/obese children who are at risk for psoriasis, and studies to address whether weight loss can reverse the severity of pediatric psoriasis.
For example, data from a study of overweight psoriatic adults showed that eating a low-calorie diet improved psoriasis severity and quality of life after 4 months (JAMA Dermatol. 2013;149:795-801).
However, in the absence of adequate data, clinicians should consider metabolic testing to determine whether their overweight pediatric patients are on the path to the metabolic syndrome, Dr. Becker and Dr. Cordoro suggested. Metabolic testing was not performed on any of the 29 children in the chart review, although 48% had a family history of hyperlipidemia and 45% had a family history of obesity.
The average age of the children was 12.6 years; the average age of onset of obesity was 4 years (range, 2-12 years), and the average age of onset of psoriasis was 9 years (range, 2-14 years).
Two obese patients were able to reduce their BMIs from obese to overweight status 1 year after being diagnosed with psoriasis; however, both have remained in the 85th-95th BMI percentile for more than 2 years, Dr. Becker said. Both of the children who had a normal BMI prior to their psoriasis had a BMI in the overweight or obese range within 1 year after diagnosis, Dr. Becker noted.
Dr. Cordoro also made an impassioned plea for clinicians to address the significant psychosocial comorbidity present in psoriatic children.
"You can think about obesity, psoriasis, and depression as being reciprocal exacerbating factors, such that each triggers the other and represents an insult to self-esteem and the overall well-being of these patients," she said. "They end up having high stress levels and really dismal quality of life. These issues are as important in the management of the child as the medical considerations."
Dr. Becker, her coauthors, and Dr. Cordoro reported having no relevant financial disclosures.
AT THE SPD ANNUAL MEETING
Major finding: Among 29 children, 93% met criteria for excess adiposity prior to developing psoriasis, and 78% of these were obese before their psoriasis.
Data source: A growth chart review of 29 obese/overweight children with psoriasis.
Disclosures: Dr. Becker, her coauthors, and Dr. Cordoro reported having no relevant financial disclosures.
Methotrexate assay useful in severe pediatric dermatitis, psoriasis
MILWAUKEE – In children with severe atopic dermatitis or psoriasis, the methotrexate polyglutamate assay appears useful in guiding dose modification in those failing to respond to methotrexate at 12 weeks, a retrospective study has shown.
The methotrexate polyglutamate 3 assay (MTX PG3) measures concentrations of methotrexate polyglutamates, the active metabolites of methotrexate, Syed Rahman said at the annual meeting of the Society for Pediatric Dermatology.
Methotrexate, a prodrug, requires enzymatic conversion via sequential addition of glutamic acid residues. Those residues form a glutamate "tail" that enhances intracellular methotrexate retention. The 3-tail metabolite, MTX PG3, is the predominant species in most patients, explained Mr. Rahman of St. Louis University.
"This study supports the usefulness of a commercially available tool for customizing optimal methotrexate dosing, coauthor Dr. Elaine Siegfried, professor of pediatrics and dermatology at the university, said in an interview.
The MTX PG3 assay has been validated in adults with rheumatoid arthritis where patients with a therapeutic methotrexate polyglutamate level of more than 60 nmol/L were fivefold more likely to have a good response to methotrexate than those with levels below this threshold, according to poster results presented at the 2008 American College of Rheumatology annual meeting (cited on the company website). However, the test was not predictive of response in 36% of patients whose arthritis improved with treatment despite subtherapeutic levels.
For the current analysis, the investigators reviewed the charts of 46 children with severe atopic dermatitis, psoriasis, or atopic dermatitis–psoriasis overlap, who were treated with a starting dose of oral methotrexate 0.5 mg/kg per week (maximum, 15 mg) and assessed by MTX PG3 assay at week 12. In a subset of children who failed to respond, the dose was modified and the level rechecked at 8 weeks. Disease severity was rated using a 5-point physician global assessment scale, with a good to excellent response defined by a 2-point improvement.
In all, 83% of children achieved a good to excellent response, with 27 patients doing so within 12 weeks and 11 responding after dose adjustment involving either a higher oral dose or switching to subcutaneous administration, Mr. Rahman reported.
Response rates to methotrexate were slightly higher in children with psoriasis or overlap (the two groups were combined) than those with atopic dermatitis (94% vs. 77%; no P value given). Overall, 8 children failed to respond.
The mean MTX PG3 level significantly correlated with efficacy in patients with atopic dermatitis (responders, 34.8 nmol/L, vs. 17.8 nmol/L, nonresponders; P = .018), but not in those with psoriasis–atopic dermatitis overlap (responders, 26.5 nmol/L, vs. 20.7, nonresponders; P = .724), Mr. Rahman reported.
The data suggest that compared with the therapeutic level of 60 nmol/L identified in adults, "pediatric patients have a lower therapeutic value," he said.
A subset analysis of MTX PG3 levels based on response identified a significantly higher mean maximum MTX PG3 level for responders than nonresponders (31.5 vs. 18.1 nmol/L; P = .035).
More importantly, late responders had the highest methotrexate levels. All late responders achieved levels greater than 30 nmol/L, and mean levels were significantly higher than for early responders (41.9 vs. 27.3 nmol/L; P = .024), suggesting that the test is most valuable for the subset of children requiring dose adjustment, Mr. Rahman said.
"Patients who respond won’t even need the test, but for those at the 12-week juncture who do not respond and their levels are less than 30 nmol/L, this [study] suggests that if we increase the PO [dose] or change to subcutaneous [administration], we will eventually have a response," he said.
During a discussion of the results, an audience member questioned whether the test, with its added expense, really enhances clinical judgment because most clinicians would already adjust the dose in their patients failing to respond at 12 weeks.
Mr. Rahman replied that not all patients will respond to continued treatment with methotrexate, and that the test could differentiate between responders and nonresponders, thereby directing the patient to alternative therapy earlier.
Session moderator Dr. Amy Paller, chair of dermatology and professor of pediatrics and dermatology at Northwestern University in Chicago, agreed that the test is most valuable for the subset of children who are nonresponders at 12 weeks.
"In these children, this test can guide dose modification," she said in an interview. "Pharmacogenetic variation can require relatively higher oral doses or subcutaneous administration."
In contrast to a therapeutic level of more than 60 nmol/L established for adults, a level of 30 nmol/L may be sufficient in children, Dr. Paller said.
"A MTX PG3 less than 30 nmol/L in a child who has failed to respond within 12 weeks suggests that an increase in oral dose, or even a change to subcutaneous [administration], will boost efficacy," she commented.
Mr. Rahman, Dr. Siegfried, and Dr. Paller reported having no relevant financial disclosures.
MILWAUKEE – In children with severe atopic dermatitis or psoriasis, the methotrexate polyglutamate assay appears useful in guiding dose modification in those failing to respond to methotrexate at 12 weeks, a retrospective study has shown.
The methotrexate polyglutamate 3 assay (MTX PG3) measures concentrations of methotrexate polyglutamates, the active metabolites of methotrexate, Syed Rahman said at the annual meeting of the Society for Pediatric Dermatology.
Methotrexate, a prodrug, requires enzymatic conversion via sequential addition of glutamic acid residues. Those residues form a glutamate "tail" that enhances intracellular methotrexate retention. The 3-tail metabolite, MTX PG3, is the predominant species in most patients, explained Mr. Rahman of St. Louis University.
"This study supports the usefulness of a commercially available tool for customizing optimal methotrexate dosing, coauthor Dr. Elaine Siegfried, professor of pediatrics and dermatology at the university, said in an interview.
The MTX PG3 assay has been validated in adults with rheumatoid arthritis where patients with a therapeutic methotrexate polyglutamate level of more than 60 nmol/L were fivefold more likely to have a good response to methotrexate than those with levels below this threshold, according to poster results presented at the 2008 American College of Rheumatology annual meeting (cited on the company website). However, the test was not predictive of response in 36% of patients whose arthritis improved with treatment despite subtherapeutic levels.
For the current analysis, the investigators reviewed the charts of 46 children with severe atopic dermatitis, psoriasis, or atopic dermatitis–psoriasis overlap, who were treated with a starting dose of oral methotrexate 0.5 mg/kg per week (maximum, 15 mg) and assessed by MTX PG3 assay at week 12. In a subset of children who failed to respond, the dose was modified and the level rechecked at 8 weeks. Disease severity was rated using a 5-point physician global assessment scale, with a good to excellent response defined by a 2-point improvement.
In all, 83% of children achieved a good to excellent response, with 27 patients doing so within 12 weeks and 11 responding after dose adjustment involving either a higher oral dose or switching to subcutaneous administration, Mr. Rahman reported.
Response rates to methotrexate were slightly higher in children with psoriasis or overlap (the two groups were combined) than those with atopic dermatitis (94% vs. 77%; no P value given). Overall, 8 children failed to respond.
The mean MTX PG3 level significantly correlated with efficacy in patients with atopic dermatitis (responders, 34.8 nmol/L, vs. 17.8 nmol/L, nonresponders; P = .018), but not in those with psoriasis–atopic dermatitis overlap (responders, 26.5 nmol/L, vs. 20.7, nonresponders; P = .724), Mr. Rahman reported.
The data suggest that compared with the therapeutic level of 60 nmol/L identified in adults, "pediatric patients have a lower therapeutic value," he said.
A subset analysis of MTX PG3 levels based on response identified a significantly higher mean maximum MTX PG3 level for responders than nonresponders (31.5 vs. 18.1 nmol/L; P = .035).
More importantly, late responders had the highest methotrexate levels. All late responders achieved levels greater than 30 nmol/L, and mean levels were significantly higher than for early responders (41.9 vs. 27.3 nmol/L; P = .024), suggesting that the test is most valuable for the subset of children requiring dose adjustment, Mr. Rahman said.
"Patients who respond won’t even need the test, but for those at the 12-week juncture who do not respond and their levels are less than 30 nmol/L, this [study] suggests that if we increase the PO [dose] or change to subcutaneous [administration], we will eventually have a response," he said.
During a discussion of the results, an audience member questioned whether the test, with its added expense, really enhances clinical judgment because most clinicians would already adjust the dose in their patients failing to respond at 12 weeks.
Mr. Rahman replied that not all patients will respond to continued treatment with methotrexate, and that the test could differentiate between responders and nonresponders, thereby directing the patient to alternative therapy earlier.
Session moderator Dr. Amy Paller, chair of dermatology and professor of pediatrics and dermatology at Northwestern University in Chicago, agreed that the test is most valuable for the subset of children who are nonresponders at 12 weeks.
"In these children, this test can guide dose modification," she said in an interview. "Pharmacogenetic variation can require relatively higher oral doses or subcutaneous administration."
In contrast to a therapeutic level of more than 60 nmol/L established for adults, a level of 30 nmol/L may be sufficient in children, Dr. Paller said.
"A MTX PG3 less than 30 nmol/L in a child who has failed to respond within 12 weeks suggests that an increase in oral dose, or even a change to subcutaneous [administration], will boost efficacy," she commented.
Mr. Rahman, Dr. Siegfried, and Dr. Paller reported having no relevant financial disclosures.
MILWAUKEE – In children with severe atopic dermatitis or psoriasis, the methotrexate polyglutamate assay appears useful in guiding dose modification in those failing to respond to methotrexate at 12 weeks, a retrospective study has shown.
The methotrexate polyglutamate 3 assay (MTX PG3) measures concentrations of methotrexate polyglutamates, the active metabolites of methotrexate, Syed Rahman said at the annual meeting of the Society for Pediatric Dermatology.
Methotrexate, a prodrug, requires enzymatic conversion via sequential addition of glutamic acid residues. Those residues form a glutamate "tail" that enhances intracellular methotrexate retention. The 3-tail metabolite, MTX PG3, is the predominant species in most patients, explained Mr. Rahman of St. Louis University.
"This study supports the usefulness of a commercially available tool for customizing optimal methotrexate dosing, coauthor Dr. Elaine Siegfried, professor of pediatrics and dermatology at the university, said in an interview.
The MTX PG3 assay has been validated in adults with rheumatoid arthritis where patients with a therapeutic methotrexate polyglutamate level of more than 60 nmol/L were fivefold more likely to have a good response to methotrexate than those with levels below this threshold, according to poster results presented at the 2008 American College of Rheumatology annual meeting (cited on the company website). However, the test was not predictive of response in 36% of patients whose arthritis improved with treatment despite subtherapeutic levels.
For the current analysis, the investigators reviewed the charts of 46 children with severe atopic dermatitis, psoriasis, or atopic dermatitis–psoriasis overlap, who were treated with a starting dose of oral methotrexate 0.5 mg/kg per week (maximum, 15 mg) and assessed by MTX PG3 assay at week 12. In a subset of children who failed to respond, the dose was modified and the level rechecked at 8 weeks. Disease severity was rated using a 5-point physician global assessment scale, with a good to excellent response defined by a 2-point improvement.
In all, 83% of children achieved a good to excellent response, with 27 patients doing so within 12 weeks and 11 responding after dose adjustment involving either a higher oral dose or switching to subcutaneous administration, Mr. Rahman reported.
Response rates to methotrexate were slightly higher in children with psoriasis or overlap (the two groups were combined) than those with atopic dermatitis (94% vs. 77%; no P value given). Overall, 8 children failed to respond.
The mean MTX PG3 level significantly correlated with efficacy in patients with atopic dermatitis (responders, 34.8 nmol/L, vs. 17.8 nmol/L, nonresponders; P = .018), but not in those with psoriasis–atopic dermatitis overlap (responders, 26.5 nmol/L, vs. 20.7, nonresponders; P = .724), Mr. Rahman reported.
The data suggest that compared with the therapeutic level of 60 nmol/L identified in adults, "pediatric patients have a lower therapeutic value," he said.
A subset analysis of MTX PG3 levels based on response identified a significantly higher mean maximum MTX PG3 level for responders than nonresponders (31.5 vs. 18.1 nmol/L; P = .035).
More importantly, late responders had the highest methotrexate levels. All late responders achieved levels greater than 30 nmol/L, and mean levels were significantly higher than for early responders (41.9 vs. 27.3 nmol/L; P = .024), suggesting that the test is most valuable for the subset of children requiring dose adjustment, Mr. Rahman said.
"Patients who respond won’t even need the test, but for those at the 12-week juncture who do not respond and their levels are less than 30 nmol/L, this [study] suggests that if we increase the PO [dose] or change to subcutaneous [administration], we will eventually have a response," he said.
During a discussion of the results, an audience member questioned whether the test, with its added expense, really enhances clinical judgment because most clinicians would already adjust the dose in their patients failing to respond at 12 weeks.
Mr. Rahman replied that not all patients will respond to continued treatment with methotrexate, and that the test could differentiate between responders and nonresponders, thereby directing the patient to alternative therapy earlier.
Session moderator Dr. Amy Paller, chair of dermatology and professor of pediatrics and dermatology at Northwestern University in Chicago, agreed that the test is most valuable for the subset of children who are nonresponders at 12 weeks.
"In these children, this test can guide dose modification," she said in an interview. "Pharmacogenetic variation can require relatively higher oral doses or subcutaneous administration."
In contrast to a therapeutic level of more than 60 nmol/L established for adults, a level of 30 nmol/L may be sufficient in children, Dr. Paller said.
"A MTX PG3 less than 30 nmol/L in a child who has failed to respond within 12 weeks suggests that an increase in oral dose, or even a change to subcutaneous [administration], will boost efficacy," she commented.
Mr. Rahman, Dr. Siegfried, and Dr. Paller reported having no relevant financial disclosures.
AT THE ANNUAL MEETING OF THE SOCIETY FOR PEDIATRIC DERMATOLOGY
Major finding: Late responders had the highest methotrexate levels (41.9 nmol/L vs. 27.3 nmol/L in early responders; P = .024) on the MTX PG3 assay.
Data source: A retrospective study of 46 children on methotrexate for atopic dermatitis, psoriasis, or atopic dermatitis–psoriasis overlap who were assessed by MTX PG3 assay.
Disclosures: Mr. Rahman, Dr. Siegfried, and Dr. Paller reported having no relevant financial disclosures.
Expert panel sets broad SpA treat-to-target goals
MADRID – A panel of clinicians with expertise in the management of patients with spondyloarthritis took another step toward better defining this disease category and the goals of its treatment by producing the first "treat-to-target" recommendations for spondyloarthritis.
The new recommendations will "guide physicians, patients, and other stakeholders on how to optimize reduction of signs and symptoms and, ideally, outcomes, and will drive" the current research agenda, Dr. Josef S. Smolen said while presenting the panel’s recommendations at the annual European Congress of Rheumatology.
"Treat to target proposes a stepwise approach to achieving optimal outcomes based on available evidence and expert opinion. Treat to target has been successfully applied to management of diabetes, hypertension, hyperlipidemia, and rheumatoid arthritis," and the new task force set out to address whether it could be used when treating patients with spondyloarthritis (SpA), a question that required the expert task force to review the evidence for setting specific treatment goals for patients with SpA, said Dr. Smolen, professor of medicine at the Medical University of Vienna.
The task force eventually decided that the treat-to-target concept was applicable to SpA, but that the data available so far prevented the task force from setting specific treatment goals. While the recommendations underscore the importance of treating SpA patients with a goal of remission or inactive disease, and failing that, at least low disease activity, they fall short of giving clinicians guidance on how to best measure and define remission or low disease activity or how to achieve these goals.
For example, for axial SpA – the subtype that received the most specific recommendations – the panel advised clinicians to guide treatment decisions by using a "validated composite measure of disease activity" such as the Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) plus acute phase reactants, or the Ankylosing Spondylitis Disease Activity Score (ASDAS) with or without functional measures such as the Bath Ankylosing Spondylitis Functional Index (BASFI). But the recommendations say nothing about what scores by these measures would define remission or low disease activity. The recommendations also talk about using other factors to further gauge axial SpA disease activity such as MRI analysis of inflammation and radiographic progression, but again Dr. Smolen provided no specific targets when using these measures.
For other, less well studied types of SpA, the new recommendations were even more nebulous. For psoriatic arthritis, the panel indicated that "validated measures of musculoskeletal disease activity" should be "performed and documented regularly" as should other factors such as spinal and extraarticular manifestations, imaging findings, and comorbidities, but the recommendations gave no specifics on what any of these measures might be or what level might equal remission or low disease. The same held true for peripheral SpA.
Other factors touched on by the new recommendations include the need for individualized and shared decision making between physicians and patients, the need for coordination of care among rheumatologists and other medical specialists who treat SpA patients (dermatologists, gastroenterologists, and ophthalmologists), and the need to take into account extraarticular manifestations of SpA diseases, comorbidities, and treatment risks along with the goal of low disease activity.
"I think it was important to document that there is a paucity of trials that have looked at strategies to treat" SpA. "For rheumatoid arthritis we have strategies, but not in spondyloarthritis, and I think it is important to say that," said Dr. Jürgen Braun, medical director of Rheumazentrum Ruhrgebiet in Herne, Germany.
SpA also lags behind rheumatoid arthritis by not having any treatments proven to slow radiographic progression. "Although we say that it is presumably important to treat inflammation [in patients with SpA], we are not sure," said Dr. Braun, who served as a member of the treat-to-target task force.
Even though the task force’s report highlights the absence of evidence for most facets of SpA management, "in the absence of evidence you need eminence, and that’s what was produced." Dr. Braun predicted that rheumatologists and other clinicians who care for patients with SpA would welcome these new recommendations despite their shortcomings. "What we say is what clinicians feel: If a patient’s CRP is elevated we must do something about it, but the evidence supporting this is limited. Presumably, it is important to treat inflammation, but we are not yet sure," he said in an interview.
"We don’t have trial results yet where you set up a quantifiable endpoint as your target, but that is coming," said Dr. Philip J. Mease, director of the rheumatology clinical research division at Swedish Medical Center in Seattle and a member of the task force. For example, the results from the Tight Control of Psoriatic Arthritis (TICOPA) trial "will tell us whether more aggressive treatment to a quantifiable target is appropriate and makes a difference. We anticipate that it will, but evidence is currently lacking." The primary endpoint of the TICOPA trial is change in the ACR20 response, but the trial includes several other clinical measures as secondary endpoints, including the Assessment in Ankylosing Spondylitis (ASAS), the BASDAI, and the psoriasis area severity index (PASI).
"The rheumatoid model is prompting us to develop quantifiable measures like the ASDAS and the new Psoriatic Arthritis Disease Activity Score (PASDAS) that we’ll start to see used. I just spoke with a colleague about the need to also develop a similar measure for peripheral SpA," Dr. Mease said in an interview.
The task force that developed the treat-to-target recommendations included 16 physicians and patients on its steering committee and 16 on an advisory committee. The majority came from various European locations, but about a quarter of the task force members were from the United States. To arrive at its recommendations, the task force used a comprehensive literature review that identified 22 published reports that addressed treatment targets for SpA (Ann. Rheum. Dis. 2013 June 10 [doi:10.1136/annrheumdis-2013-203860]). The treat-to-target recommendations were published online a few days before Dr. Smolen’s presentation at the meeting (Ann. Rheum. Dis. 2013 June 8 [doi:10.1136/annrheumdis-2013-203419]).
Dr. Smolen, Dr. Braun, and Dr. Mease said that they had no disclosures relevant to the topic.
On Twitter @mitchelzoler
MADRID – A panel of clinicians with expertise in the management of patients with spondyloarthritis took another step toward better defining this disease category and the goals of its treatment by producing the first "treat-to-target" recommendations for spondyloarthritis.
The new recommendations will "guide physicians, patients, and other stakeholders on how to optimize reduction of signs and symptoms and, ideally, outcomes, and will drive" the current research agenda, Dr. Josef S. Smolen said while presenting the panel’s recommendations at the annual European Congress of Rheumatology.
"Treat to target proposes a stepwise approach to achieving optimal outcomes based on available evidence and expert opinion. Treat to target has been successfully applied to management of diabetes, hypertension, hyperlipidemia, and rheumatoid arthritis," and the new task force set out to address whether it could be used when treating patients with spondyloarthritis (SpA), a question that required the expert task force to review the evidence for setting specific treatment goals for patients with SpA, said Dr. Smolen, professor of medicine at the Medical University of Vienna.
The task force eventually decided that the treat-to-target concept was applicable to SpA, but that the data available so far prevented the task force from setting specific treatment goals. While the recommendations underscore the importance of treating SpA patients with a goal of remission or inactive disease, and failing that, at least low disease activity, they fall short of giving clinicians guidance on how to best measure and define remission or low disease activity or how to achieve these goals.
For example, for axial SpA – the subtype that received the most specific recommendations – the panel advised clinicians to guide treatment decisions by using a "validated composite measure of disease activity" such as the Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) plus acute phase reactants, or the Ankylosing Spondylitis Disease Activity Score (ASDAS) with or without functional measures such as the Bath Ankylosing Spondylitis Functional Index (BASFI). But the recommendations say nothing about what scores by these measures would define remission or low disease activity. The recommendations also talk about using other factors to further gauge axial SpA disease activity such as MRI analysis of inflammation and radiographic progression, but again Dr. Smolen provided no specific targets when using these measures.
For other, less well studied types of SpA, the new recommendations were even more nebulous. For psoriatic arthritis, the panel indicated that "validated measures of musculoskeletal disease activity" should be "performed and documented regularly" as should other factors such as spinal and extraarticular manifestations, imaging findings, and comorbidities, but the recommendations gave no specifics on what any of these measures might be or what level might equal remission or low disease. The same held true for peripheral SpA.
Other factors touched on by the new recommendations include the need for individualized and shared decision making between physicians and patients, the need for coordination of care among rheumatologists and other medical specialists who treat SpA patients (dermatologists, gastroenterologists, and ophthalmologists), and the need to take into account extraarticular manifestations of SpA diseases, comorbidities, and treatment risks along with the goal of low disease activity.
"I think it was important to document that there is a paucity of trials that have looked at strategies to treat" SpA. "For rheumatoid arthritis we have strategies, but not in spondyloarthritis, and I think it is important to say that," said Dr. Jürgen Braun, medical director of Rheumazentrum Ruhrgebiet in Herne, Germany.
SpA also lags behind rheumatoid arthritis by not having any treatments proven to slow radiographic progression. "Although we say that it is presumably important to treat inflammation [in patients with SpA], we are not sure," said Dr. Braun, who served as a member of the treat-to-target task force.
Even though the task force’s report highlights the absence of evidence for most facets of SpA management, "in the absence of evidence you need eminence, and that’s what was produced." Dr. Braun predicted that rheumatologists and other clinicians who care for patients with SpA would welcome these new recommendations despite their shortcomings. "What we say is what clinicians feel: If a patient’s CRP is elevated we must do something about it, but the evidence supporting this is limited. Presumably, it is important to treat inflammation, but we are not yet sure," he said in an interview.
"We don’t have trial results yet where you set up a quantifiable endpoint as your target, but that is coming," said Dr. Philip J. Mease, director of the rheumatology clinical research division at Swedish Medical Center in Seattle and a member of the task force. For example, the results from the Tight Control of Psoriatic Arthritis (TICOPA) trial "will tell us whether more aggressive treatment to a quantifiable target is appropriate and makes a difference. We anticipate that it will, but evidence is currently lacking." The primary endpoint of the TICOPA trial is change in the ACR20 response, but the trial includes several other clinical measures as secondary endpoints, including the Assessment in Ankylosing Spondylitis (ASAS), the BASDAI, and the psoriasis area severity index (PASI).
"The rheumatoid model is prompting us to develop quantifiable measures like the ASDAS and the new Psoriatic Arthritis Disease Activity Score (PASDAS) that we’ll start to see used. I just spoke with a colleague about the need to also develop a similar measure for peripheral SpA," Dr. Mease said in an interview.
The task force that developed the treat-to-target recommendations included 16 physicians and patients on its steering committee and 16 on an advisory committee. The majority came from various European locations, but about a quarter of the task force members were from the United States. To arrive at its recommendations, the task force used a comprehensive literature review that identified 22 published reports that addressed treatment targets for SpA (Ann. Rheum. Dis. 2013 June 10 [doi:10.1136/annrheumdis-2013-203860]). The treat-to-target recommendations were published online a few days before Dr. Smolen’s presentation at the meeting (Ann. Rheum. Dis. 2013 June 8 [doi:10.1136/annrheumdis-2013-203419]).
Dr. Smolen, Dr. Braun, and Dr. Mease said that they had no disclosures relevant to the topic.
On Twitter @mitchelzoler
MADRID – A panel of clinicians with expertise in the management of patients with spondyloarthritis took another step toward better defining this disease category and the goals of its treatment by producing the first "treat-to-target" recommendations for spondyloarthritis.
The new recommendations will "guide physicians, patients, and other stakeholders on how to optimize reduction of signs and symptoms and, ideally, outcomes, and will drive" the current research agenda, Dr. Josef S. Smolen said while presenting the panel’s recommendations at the annual European Congress of Rheumatology.
"Treat to target proposes a stepwise approach to achieving optimal outcomes based on available evidence and expert opinion. Treat to target has been successfully applied to management of diabetes, hypertension, hyperlipidemia, and rheumatoid arthritis," and the new task force set out to address whether it could be used when treating patients with spondyloarthritis (SpA), a question that required the expert task force to review the evidence for setting specific treatment goals for patients with SpA, said Dr. Smolen, professor of medicine at the Medical University of Vienna.
The task force eventually decided that the treat-to-target concept was applicable to SpA, but that the data available so far prevented the task force from setting specific treatment goals. While the recommendations underscore the importance of treating SpA patients with a goal of remission or inactive disease, and failing that, at least low disease activity, they fall short of giving clinicians guidance on how to best measure and define remission or low disease activity or how to achieve these goals.
For example, for axial SpA – the subtype that received the most specific recommendations – the panel advised clinicians to guide treatment decisions by using a "validated composite measure of disease activity" such as the Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) plus acute phase reactants, or the Ankylosing Spondylitis Disease Activity Score (ASDAS) with or without functional measures such as the Bath Ankylosing Spondylitis Functional Index (BASFI). But the recommendations say nothing about what scores by these measures would define remission or low disease activity. The recommendations also talk about using other factors to further gauge axial SpA disease activity such as MRI analysis of inflammation and radiographic progression, but again Dr. Smolen provided no specific targets when using these measures.
For other, less well studied types of SpA, the new recommendations were even more nebulous. For psoriatic arthritis, the panel indicated that "validated measures of musculoskeletal disease activity" should be "performed and documented regularly" as should other factors such as spinal and extraarticular manifestations, imaging findings, and comorbidities, but the recommendations gave no specifics on what any of these measures might be or what level might equal remission or low disease. The same held true for peripheral SpA.
Other factors touched on by the new recommendations include the need for individualized and shared decision making between physicians and patients, the need for coordination of care among rheumatologists and other medical specialists who treat SpA patients (dermatologists, gastroenterologists, and ophthalmologists), and the need to take into account extraarticular manifestations of SpA diseases, comorbidities, and treatment risks along with the goal of low disease activity.
"I think it was important to document that there is a paucity of trials that have looked at strategies to treat" SpA. "For rheumatoid arthritis we have strategies, but not in spondyloarthritis, and I think it is important to say that," said Dr. Jürgen Braun, medical director of Rheumazentrum Ruhrgebiet in Herne, Germany.
SpA also lags behind rheumatoid arthritis by not having any treatments proven to slow radiographic progression. "Although we say that it is presumably important to treat inflammation [in patients with SpA], we are not sure," said Dr. Braun, who served as a member of the treat-to-target task force.
Even though the task force’s report highlights the absence of evidence for most facets of SpA management, "in the absence of evidence you need eminence, and that’s what was produced." Dr. Braun predicted that rheumatologists and other clinicians who care for patients with SpA would welcome these new recommendations despite their shortcomings. "What we say is what clinicians feel: If a patient’s CRP is elevated we must do something about it, but the evidence supporting this is limited. Presumably, it is important to treat inflammation, but we are not yet sure," he said in an interview.
"We don’t have trial results yet where you set up a quantifiable endpoint as your target, but that is coming," said Dr. Philip J. Mease, director of the rheumatology clinical research division at Swedish Medical Center in Seattle and a member of the task force. For example, the results from the Tight Control of Psoriatic Arthritis (TICOPA) trial "will tell us whether more aggressive treatment to a quantifiable target is appropriate and makes a difference. We anticipate that it will, but evidence is currently lacking." The primary endpoint of the TICOPA trial is change in the ACR20 response, but the trial includes several other clinical measures as secondary endpoints, including the Assessment in Ankylosing Spondylitis (ASAS), the BASDAI, and the psoriasis area severity index (PASI).
"The rheumatoid model is prompting us to develop quantifiable measures like the ASDAS and the new Psoriatic Arthritis Disease Activity Score (PASDAS) that we’ll start to see used. I just spoke with a colleague about the need to also develop a similar measure for peripheral SpA," Dr. Mease said in an interview.
The task force that developed the treat-to-target recommendations included 16 physicians and patients on its steering committee and 16 on an advisory committee. The majority came from various European locations, but about a quarter of the task force members were from the United States. To arrive at its recommendations, the task force used a comprehensive literature review that identified 22 published reports that addressed treatment targets for SpA (Ann. Rheum. Dis. 2013 June 10 [doi:10.1136/annrheumdis-2013-203860]). The treat-to-target recommendations were published online a few days before Dr. Smolen’s presentation at the meeting (Ann. Rheum. Dis. 2013 June 8 [doi:10.1136/annrheumdis-2013-203419]).
Dr. Smolen, Dr. Braun, and Dr. Mease said that they had no disclosures relevant to the topic.
On Twitter @mitchelzoler
AT THE EULAR CONGRESS 2013