User login
Diabetes tied to Parkinson’s risk, more rapid disease progression
Diabetes mellitus (DM) is associated with Parkinson’s disease (PD) development, as well as more severe symptoms and more rapid disease progression, new research suggests.
In a systematic review, patients with type 2 diabetes were 34% more likely to develop PD than those without comorbid DM. In addition, patients with both conditions had significantly worse scores on the Unified Parkinson’s Disease Rating Scale (UPDRS) and worse cognitive performance.
Together, the results suggest that “DM may be a facilitating factor of neurodegeneration,” wrote the investigators, led by Gennaro Pagano, MD, PhD, expert medical director at Roche Pharma Research and Early Development, in Basel, Switzerland.
The findings were published in a recent issue of the Journal of Parkinson’s Disease.
Unanswered questions
Researchers have long proposed a potential relationship between diabetes and PD. However, case-control studies have yielded conflicting results about this relationship – and previous systematic reviews have failed to clarify the question.
In the current systematic review and meta-analysis, investigators identified relevant studies in databases such as MEDLINE/PubMed, Cochrane CENTRAL, and Scopus.
Eligible studies reported prevalence of DM in patients with PD, reported incidence of PD in those with and those without DM, and analyzed Parkinson’s phenotype and progression in those with and those without DM.
The researchers identified 3,829 articles in their initial search, evaluated 90 articles in detail, and included 43 studies in their analysis. Study quality was judged to be moderate or good, and the investigators did not find significant publication bias.
Twenty-one studies that encompassed 11,396 patients were examined to determine prevalence of DM in PD. This prevalence was calculated to be 10.02%, which is similar to the global prevalence of 9.3% reported in 2019.
The researchers also analyzed 12 cohort studies that included 17,797,221 patients to calculate risk for PD in patients with comorbid diabetes. The pooled summary odds ratio for incident PD among patients with type 2 diabetes was 1.34.
The evaluation of the effect of diabetes on PD severity was based on 10 studies that included 603 patients with both diseases. Because data on motor symptoms were not available for all studies, the researchers considered Hoehn and Yahr stage, UPDRS score, and cognitive impairment.
Patients with both conditions had a worse Hoehn and Yahr stage (standardized mean difference, 0.36; P < .001), and higher UPDRS score (SMD, 0.60; P < .001). In 7 of the 10 studies, diabetes was associated with worse cognitive performance in patients with PD.
Mechanisms uncertain
The mechanisms of the effect of diabetes on risk for and severity of PD are uncertain, but the researchers have developed hypotheses.
“Overlapping mechanisms between insulin resistance, mitochondrial dysfunction, oxidative stress, and alpha-synuclein expression could influence the development of the neurodegeneration process,” they wrote.
Because the current analysis demonstrated a trend toward more pronounced cognitive decline in patients with the comorbidities, clinicians should pay particular attention to the progression of motor and cognitive symptoms in patients with these diseases, the investigators noted.
“Additional studies are needed in order to better define the clinical phenotype of PD-DM patients and explore the role of antidiabetic drugs on PD progression,” they wrote.
They add that future studies also are needed to evaluate whether antidiabetic drugs might reduce risk for PD in these patients.
The investigators noted several limitations of their research. In many of the studies they examined, for example, diagnostic criteria of type 2 diabetes and PD were based only on medical records or self-reported health questionnaires. The diagnoses were rarely confirmed.
In addition, not all studies clearly stated that their populations presented with type 2 diabetes. Finally, patients with diabetes may be at increased risk for cardiovascular death, which could affect follow-up related to the development of PD, the investigators noted.
A version of this article first appeared on Medscape.com.
Diabetes mellitus (DM) is associated with Parkinson’s disease (PD) development, as well as more severe symptoms and more rapid disease progression, new research suggests.
In a systematic review, patients with type 2 diabetes were 34% more likely to develop PD than those without comorbid DM. In addition, patients with both conditions had significantly worse scores on the Unified Parkinson’s Disease Rating Scale (UPDRS) and worse cognitive performance.
Together, the results suggest that “DM may be a facilitating factor of neurodegeneration,” wrote the investigators, led by Gennaro Pagano, MD, PhD, expert medical director at Roche Pharma Research and Early Development, in Basel, Switzerland.
The findings were published in a recent issue of the Journal of Parkinson’s Disease.
Unanswered questions
Researchers have long proposed a potential relationship between diabetes and PD. However, case-control studies have yielded conflicting results about this relationship – and previous systematic reviews have failed to clarify the question.
In the current systematic review and meta-analysis, investigators identified relevant studies in databases such as MEDLINE/PubMed, Cochrane CENTRAL, and Scopus.
Eligible studies reported prevalence of DM in patients with PD, reported incidence of PD in those with and those without DM, and analyzed Parkinson’s phenotype and progression in those with and those without DM.
The researchers identified 3,829 articles in their initial search, evaluated 90 articles in detail, and included 43 studies in their analysis. Study quality was judged to be moderate or good, and the investigators did not find significant publication bias.
Twenty-one studies that encompassed 11,396 patients were examined to determine prevalence of DM in PD. This prevalence was calculated to be 10.02%, which is similar to the global prevalence of 9.3% reported in 2019.
The researchers also analyzed 12 cohort studies that included 17,797,221 patients to calculate risk for PD in patients with comorbid diabetes. The pooled summary odds ratio for incident PD among patients with type 2 diabetes was 1.34.
The evaluation of the effect of diabetes on PD severity was based on 10 studies that included 603 patients with both diseases. Because data on motor symptoms were not available for all studies, the researchers considered Hoehn and Yahr stage, UPDRS score, and cognitive impairment.
Patients with both conditions had a worse Hoehn and Yahr stage (standardized mean difference, 0.36; P < .001), and higher UPDRS score (SMD, 0.60; P < .001). In 7 of the 10 studies, diabetes was associated with worse cognitive performance in patients with PD.
Mechanisms uncertain
The mechanisms of the effect of diabetes on risk for and severity of PD are uncertain, but the researchers have developed hypotheses.
“Overlapping mechanisms between insulin resistance, mitochondrial dysfunction, oxidative stress, and alpha-synuclein expression could influence the development of the neurodegeneration process,” they wrote.
Because the current analysis demonstrated a trend toward more pronounced cognitive decline in patients with the comorbidities, clinicians should pay particular attention to the progression of motor and cognitive symptoms in patients with these diseases, the investigators noted.
“Additional studies are needed in order to better define the clinical phenotype of PD-DM patients and explore the role of antidiabetic drugs on PD progression,” they wrote.
They add that future studies also are needed to evaluate whether antidiabetic drugs might reduce risk for PD in these patients.
The investigators noted several limitations of their research. In many of the studies they examined, for example, diagnostic criteria of type 2 diabetes and PD were based only on medical records or self-reported health questionnaires. The diagnoses were rarely confirmed.
In addition, not all studies clearly stated that their populations presented with type 2 diabetes. Finally, patients with diabetes may be at increased risk for cardiovascular death, which could affect follow-up related to the development of PD, the investigators noted.
A version of this article first appeared on Medscape.com.
Diabetes mellitus (DM) is associated with Parkinson’s disease (PD) development, as well as more severe symptoms and more rapid disease progression, new research suggests.
In a systematic review, patients with type 2 diabetes were 34% more likely to develop PD than those without comorbid DM. In addition, patients with both conditions had significantly worse scores on the Unified Parkinson’s Disease Rating Scale (UPDRS) and worse cognitive performance.
Together, the results suggest that “DM may be a facilitating factor of neurodegeneration,” wrote the investigators, led by Gennaro Pagano, MD, PhD, expert medical director at Roche Pharma Research and Early Development, in Basel, Switzerland.
The findings were published in a recent issue of the Journal of Parkinson’s Disease.
Unanswered questions
Researchers have long proposed a potential relationship between diabetes and PD. However, case-control studies have yielded conflicting results about this relationship – and previous systematic reviews have failed to clarify the question.
In the current systematic review and meta-analysis, investigators identified relevant studies in databases such as MEDLINE/PubMed, Cochrane CENTRAL, and Scopus.
Eligible studies reported prevalence of DM in patients with PD, reported incidence of PD in those with and those without DM, and analyzed Parkinson’s phenotype and progression in those with and those without DM.
The researchers identified 3,829 articles in their initial search, evaluated 90 articles in detail, and included 43 studies in their analysis. Study quality was judged to be moderate or good, and the investigators did not find significant publication bias.
Twenty-one studies that encompassed 11,396 patients were examined to determine prevalence of DM in PD. This prevalence was calculated to be 10.02%, which is similar to the global prevalence of 9.3% reported in 2019.
The researchers also analyzed 12 cohort studies that included 17,797,221 patients to calculate risk for PD in patients with comorbid diabetes. The pooled summary odds ratio for incident PD among patients with type 2 diabetes was 1.34.
The evaluation of the effect of diabetes on PD severity was based on 10 studies that included 603 patients with both diseases. Because data on motor symptoms were not available for all studies, the researchers considered Hoehn and Yahr stage, UPDRS score, and cognitive impairment.
Patients with both conditions had a worse Hoehn and Yahr stage (standardized mean difference, 0.36; P < .001), and higher UPDRS score (SMD, 0.60; P < .001). In 7 of the 10 studies, diabetes was associated with worse cognitive performance in patients with PD.
Mechanisms uncertain
The mechanisms of the effect of diabetes on risk for and severity of PD are uncertain, but the researchers have developed hypotheses.
“Overlapping mechanisms between insulin resistance, mitochondrial dysfunction, oxidative stress, and alpha-synuclein expression could influence the development of the neurodegeneration process,” they wrote.
Because the current analysis demonstrated a trend toward more pronounced cognitive decline in patients with the comorbidities, clinicians should pay particular attention to the progression of motor and cognitive symptoms in patients with these diseases, the investigators noted.
“Additional studies are needed in order to better define the clinical phenotype of PD-DM patients and explore the role of antidiabetic drugs on PD progression,” they wrote.
They add that future studies also are needed to evaluate whether antidiabetic drugs might reduce risk for PD in these patients.
The investigators noted several limitations of their research. In many of the studies they examined, for example, diagnostic criteria of type 2 diabetes and PD were based only on medical records or self-reported health questionnaires. The diagnoses were rarely confirmed.
In addition, not all studies clearly stated that their populations presented with type 2 diabetes. Finally, patients with diabetes may be at increased risk for cardiovascular death, which could affect follow-up related to the development of PD, the investigators noted.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF PARKINSON’S DISEASE
Parkinson’s death rate rising, reasons unclear
, according to what investigators say is the most comprehensive study in the nation of temporal trends in Parkinson’s disease mortality.
“The reason behind the rising death rates from Parkinson’s disease is not clear at present and warrants further investigation,” Wei Bao, MD, PhD, associate professor in the department of epidemiology at the University of Iowa College of Public Health, in Iowa City, said in an interview. “We know that people are living longer and the general population is getting older, but that doesn’t fully explain the increase we saw in the death rate in people with Parkinson’s disease,” Dr. Bao added in a statement.
“Understanding why more people are dying from this disease is critical if we are going to reverse the trend,” Dr. Bao said.
The study was published online Oct. 27 in Neurology.
Long-term data
The researchers used data from the National Vital Statistics System to determine national trends in Parkinson’s disease mortality overall and in several key subgroups. The analyses included 479,059 people who died of Parkinson’s disease between 1999 and 2019.
Over the 21-year period, the age-adjusted mortality from Parkinson’s disease rose from 5.4 per 100,000 in 1999 to 8.8 per 100,000 in 2019. The average annual percent change (APC) was 2.4% for the entire period.
During the study period, the number of deaths from Parkinson’s disease more than doubled, from 14,593 to 35,311.
The death rate from Parkinson’s disease increased significantly across all age groups. The average APC was 5.0% among adults younger than 65 years, 1.9% among those aged 65-74 years, 2.2% among those 75-84 years, and 2.7% among those 85 and older.
The death rate increased in both men and women, but age-adjusted Parkinson’s disease mortality was twice as high in men as in women. The researchers say one possible explanation for the sex difference is estrogen, which leads to higher dopamine levels in areas of the brain that control motor responses and may protect women from Parkinson’s disease.
The study also showed that White people are more likely to die from Parkinson’s disease than persons of other racial and ethnic groups. In 2019, the death rate per 100,000 was 9.7 for Whites, 6.5 for Hispanics, and 4.7 for non-Hispanic Blacks.
Previous studies have shown that compared with White people, Black and Hispanic people are less likely to see a neurologist, owing to socioeconomic barriers. This suggests that White people may be more likely to receive a Parkinson’s disease diagnosis, the researchers noted.
“It’s important to continue to evaluate long-term trends in Parkinson’s death rates,” Dr. Bao said.
“This can inform future research that may help pinpoint why more people are dying of the disease. Also, updating vital statistics about Parkinson’s death rates may be used for priority setting and financing of health care and policy,” Dr. Bao added.
1.2 million patients by 2030
Reached for comment, James Beck, PhD, chief scientific officer for the Parkinson’s Foundation, said these findings are not surprising. “They are aligned with the work the Parkinson’s Foundation has done to show that the number of people with Parkinson’s disease has increased over time. We are working on an improved estimate of Parkinson’s disease incidence and predict that Parkinson’s disease will continue to rise as the population ages, so an increase in mortality rates would be expected,” Dr. Beck said.
He noted that much of the public health statistics regarding Parkinson’s disease are outdated and that the Parkinson’s Foundation has been partnering with others to update them.
“For instance, to calculate an accurate estimate of the prevalence of Parkinson’s disease, the Parkinson’s Foundation Prevalence Project was formed. The findings from this group demonstrated that the number of people living with Parkinson’s disease will rise to nearly 1.2 million by 2030, a substantial increase from the estimate of 930,000 for 2020,” Dr. Beck said.
“The overarching message is that more people are being diagnosed with Parkinson’s disease, not that more people are dying from the disease,” he added.
“Over the last 20 years, our understanding of Parkinson’s disease has changed and developed, so clinicians are more aware and better able to properly diagnose Parkinson’s disease. This could mean that the cause is likely due to an increase in diagnosis rates and better recognition of Parkinson’s disease, which would lead to higher rates of identifying Parkinson’s disease as a cause of death,” said Dr. Beck.
The study had no targeted funding. Dr. Bao and Dr. Beck have indicated no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, according to what investigators say is the most comprehensive study in the nation of temporal trends in Parkinson’s disease mortality.
“The reason behind the rising death rates from Parkinson’s disease is not clear at present and warrants further investigation,” Wei Bao, MD, PhD, associate professor in the department of epidemiology at the University of Iowa College of Public Health, in Iowa City, said in an interview. “We know that people are living longer and the general population is getting older, but that doesn’t fully explain the increase we saw in the death rate in people with Parkinson’s disease,” Dr. Bao added in a statement.
“Understanding why more people are dying from this disease is critical if we are going to reverse the trend,” Dr. Bao said.
The study was published online Oct. 27 in Neurology.
Long-term data
The researchers used data from the National Vital Statistics System to determine national trends in Parkinson’s disease mortality overall and in several key subgroups. The analyses included 479,059 people who died of Parkinson’s disease between 1999 and 2019.
Over the 21-year period, the age-adjusted mortality from Parkinson’s disease rose from 5.4 per 100,000 in 1999 to 8.8 per 100,000 in 2019. The average annual percent change (APC) was 2.4% for the entire period.
During the study period, the number of deaths from Parkinson’s disease more than doubled, from 14,593 to 35,311.
The death rate from Parkinson’s disease increased significantly across all age groups. The average APC was 5.0% among adults younger than 65 years, 1.9% among those aged 65-74 years, 2.2% among those 75-84 years, and 2.7% among those 85 and older.
The death rate increased in both men and women, but age-adjusted Parkinson’s disease mortality was twice as high in men as in women. The researchers say one possible explanation for the sex difference is estrogen, which leads to higher dopamine levels in areas of the brain that control motor responses and may protect women from Parkinson’s disease.
The study also showed that White people are more likely to die from Parkinson’s disease than persons of other racial and ethnic groups. In 2019, the death rate per 100,000 was 9.7 for Whites, 6.5 for Hispanics, and 4.7 for non-Hispanic Blacks.
Previous studies have shown that compared with White people, Black and Hispanic people are less likely to see a neurologist, owing to socioeconomic barriers. This suggests that White people may be more likely to receive a Parkinson’s disease diagnosis, the researchers noted.
“It’s important to continue to evaluate long-term trends in Parkinson’s death rates,” Dr. Bao said.
“This can inform future research that may help pinpoint why more people are dying of the disease. Also, updating vital statistics about Parkinson’s death rates may be used for priority setting and financing of health care and policy,” Dr. Bao added.
1.2 million patients by 2030
Reached for comment, James Beck, PhD, chief scientific officer for the Parkinson’s Foundation, said these findings are not surprising. “They are aligned with the work the Parkinson’s Foundation has done to show that the number of people with Parkinson’s disease has increased over time. We are working on an improved estimate of Parkinson’s disease incidence and predict that Parkinson’s disease will continue to rise as the population ages, so an increase in mortality rates would be expected,” Dr. Beck said.
He noted that much of the public health statistics regarding Parkinson’s disease are outdated and that the Parkinson’s Foundation has been partnering with others to update them.
“For instance, to calculate an accurate estimate of the prevalence of Parkinson’s disease, the Parkinson’s Foundation Prevalence Project was formed. The findings from this group demonstrated that the number of people living with Parkinson’s disease will rise to nearly 1.2 million by 2030, a substantial increase from the estimate of 930,000 for 2020,” Dr. Beck said.
“The overarching message is that more people are being diagnosed with Parkinson’s disease, not that more people are dying from the disease,” he added.
“Over the last 20 years, our understanding of Parkinson’s disease has changed and developed, so clinicians are more aware and better able to properly diagnose Parkinson’s disease. This could mean that the cause is likely due to an increase in diagnosis rates and better recognition of Parkinson’s disease, which would lead to higher rates of identifying Parkinson’s disease as a cause of death,” said Dr. Beck.
The study had no targeted funding. Dr. Bao and Dr. Beck have indicated no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, according to what investigators say is the most comprehensive study in the nation of temporal trends in Parkinson’s disease mortality.
“The reason behind the rising death rates from Parkinson’s disease is not clear at present and warrants further investigation,” Wei Bao, MD, PhD, associate professor in the department of epidemiology at the University of Iowa College of Public Health, in Iowa City, said in an interview. “We know that people are living longer and the general population is getting older, but that doesn’t fully explain the increase we saw in the death rate in people with Parkinson’s disease,” Dr. Bao added in a statement.
“Understanding why more people are dying from this disease is critical if we are going to reverse the trend,” Dr. Bao said.
The study was published online Oct. 27 in Neurology.
Long-term data
The researchers used data from the National Vital Statistics System to determine national trends in Parkinson’s disease mortality overall and in several key subgroups. The analyses included 479,059 people who died of Parkinson’s disease between 1999 and 2019.
Over the 21-year period, the age-adjusted mortality from Parkinson’s disease rose from 5.4 per 100,000 in 1999 to 8.8 per 100,000 in 2019. The average annual percent change (APC) was 2.4% for the entire period.
During the study period, the number of deaths from Parkinson’s disease more than doubled, from 14,593 to 35,311.
The death rate from Parkinson’s disease increased significantly across all age groups. The average APC was 5.0% among adults younger than 65 years, 1.9% among those aged 65-74 years, 2.2% among those 75-84 years, and 2.7% among those 85 and older.
The death rate increased in both men and women, but age-adjusted Parkinson’s disease mortality was twice as high in men as in women. The researchers say one possible explanation for the sex difference is estrogen, which leads to higher dopamine levels in areas of the brain that control motor responses and may protect women from Parkinson’s disease.
The study also showed that White people are more likely to die from Parkinson’s disease than persons of other racial and ethnic groups. In 2019, the death rate per 100,000 was 9.7 for Whites, 6.5 for Hispanics, and 4.7 for non-Hispanic Blacks.
Previous studies have shown that compared with White people, Black and Hispanic people are less likely to see a neurologist, owing to socioeconomic barriers. This suggests that White people may be more likely to receive a Parkinson’s disease diagnosis, the researchers noted.
“It’s important to continue to evaluate long-term trends in Parkinson’s death rates,” Dr. Bao said.
“This can inform future research that may help pinpoint why more people are dying of the disease. Also, updating vital statistics about Parkinson’s death rates may be used for priority setting and financing of health care and policy,” Dr. Bao added.
1.2 million patients by 2030
Reached for comment, James Beck, PhD, chief scientific officer for the Parkinson’s Foundation, said these findings are not surprising. “They are aligned with the work the Parkinson’s Foundation has done to show that the number of people with Parkinson’s disease has increased over time. We are working on an improved estimate of Parkinson’s disease incidence and predict that Parkinson’s disease will continue to rise as the population ages, so an increase in mortality rates would be expected,” Dr. Beck said.
He noted that much of the public health statistics regarding Parkinson’s disease are outdated and that the Parkinson’s Foundation has been partnering with others to update them.
“For instance, to calculate an accurate estimate of the prevalence of Parkinson’s disease, the Parkinson’s Foundation Prevalence Project was formed. The findings from this group demonstrated that the number of people living with Parkinson’s disease will rise to nearly 1.2 million by 2030, a substantial increase from the estimate of 930,000 for 2020,” Dr. Beck said.
“The overarching message is that more people are being diagnosed with Parkinson’s disease, not that more people are dying from the disease,” he added.
“Over the last 20 years, our understanding of Parkinson’s disease has changed and developed, so clinicians are more aware and better able to properly diagnose Parkinson’s disease. This could mean that the cause is likely due to an increase in diagnosis rates and better recognition of Parkinson’s disease, which would lead to higher rates of identifying Parkinson’s disease as a cause of death,” said Dr. Beck.
The study had no targeted funding. Dr. Bao and Dr. Beck have indicated no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM NEUROLOGY
FDA not recognizing efficacy of psychopharmacologic therapies
Many years ago, drug development in psychiatry turned to control of specific symptoms across disorders rather than within disorders, but regulatory agencies are still not yet on board, according to an expert psychopharmacologist outlining the ongoing evolution at the virtual Psychopharmacology Update presented by Current Psychiatry and the American Academy of Clinical Psychiatrists, sponsored by Medscape Live.
If this reorientation is going to lead to the broad indications the newer drugs likely deserve, which is control of specific types of symptoms regardless of the diagnosis, “we have to move the [Food and Drug Administration] along,” said Stephen M. Stahl, MD, PhD, chairman of the Neuroscience Institute and an adjunct professor of psychiatry at the University of California, San Diego.
On the side of drug development and clinical practice, the reorientation has already taken place. Dr. Stahl described numerous brain circuits known to produce symptoms when function is altered that are now treatment targets. This includes the ventral medial prefrontal cortex where deficient information processing leads to depression and the orbital frontal cortex where altered function leads to impulsivity.
“It is not like each part of the brain does a little bit of everything. Rather, each part of the brain has an assignment and duty and function,” Dr. Stahl explained. By addressing the disturbed signaling in brain circuits that lead to depression, impulsivity, agitation, or other symptoms, there is an opportunity for control, regardless of the psychiatric diagnosis with which the symptom is associated.
For example, Dr. Stahl predicted that pimavanserin, a highly selective 5-HT2A inverse agonist that is already approved for psychosis in Parkinson’s disease, is now likely to be approved for psychosis associated with other conditions on the basis of recent positive clinical studies in these other disorders.
Brexpiprazole, a serotonin-dopamine activity modulator already known to be useful for control of the agitation characteristic of schizophrenia, is now showing the same type of activity against agitation when it is associated with Alzheimer’s disease. Again, Dr. Stahl thinks this drug is on course for an indication across diseases once studies are conducted in each disease individually.
Another drug being evaluated for agitation, the N-methyl-D-aspartate receptor antagonist dextromethorphan bupropion, is also being tested for treatment of symptoms across multiple disorders, he reported.
However, the FDA has so far taken the position that each drug must be tested separately for a given symptom in each disorder for which it is being considered despite the underlying premise that it is the symptom, not the disease, that is important.
Unlike physiological diseases where symptoms, like a fever or abdominal cramps, are the product of a disease, psychiatric symptoms are the disease and a fundamental target – regardless of the DSM-based diagnosis.
To some degree, the symptoms of psychiatric disorders have always been the focus of treatment, but a pivot toward developing therapies that will control a symptom regardless of the underlying diagnosis is an important conceptual change. It is being made possible by advances in the detail with which the neuropathology of these symptoms is understood .
“By my count, 79 symptoms are described in DSM-5, but they are spread across hundreds of syndromes because they are grouped together in different ways,” Dr. Stahl observed.
He noted that clinicians make a diagnosis on the basis symptom groupings, but their interventions are selected to address the manifestations of the disease, not the disease itself.
“If you are a real psychopharmacologist treating real patients, you are treating the specific symptoms of the specific patient,” according to Dr. Stahl.
So far, the FDA has not made this leap, insisting on trials in these categorical disorders rather than permitting trial designs that allow benefit to be demonstrated against a symptom regardless of the syndrome with which it is associated.
Of egregious examples, Dr. Stahl recounted a recent trial of a 5-HT2 antagonist that looked so promising against psychosis in Alzheimer’s disease that the trialists enrolled patients with psychosis regardless of type of dementia, such as vascular dementia and Lewy body disease. The efficacy was impressive.
“It worked so well that they stopped the trial, but the FDA declined to approve it,” Dr. Stahl recounted. Despite clear evidence of benefit, the regulators insisted that the investigators needed to show a significant benefit in each condition individually.
While the trial investigators acknowledged that there was not enough power in the trial to show a statistically significant benefit in each category, they argued that the overall benefit and the consistent response across categories required them to stop the trial for ethical reasons.
“That’s your problem, the FDA said to the investigators,” according to Dr. Stahl.
The failure of the FDA to recognize the efficacy of psychopharmacologic therapies across symptoms regardless of the associated disease is a failure to stay current with an important evolution in medicine, Dr. Stahl indicated.
“What we have come to understand is the neurobiology of any given symptom is likely to be the same across disorders,” he said.
Agency’s arbitrary decisions cited
“I completely agree with Dr. Stahl,” said Henry A. Nasrallah, MD, professor of psychiatry, neurology, and neuroscience, University of Cincinnati.
In addition to the fact that symptoms are present across multiple categories, many patients manifest multiple symptoms at one time, Dr. Nasrallah pointed out. For neurodegenerative disorders associated with psychosis, depression, anxiety, aggression, and other symptoms, it is already well known that the heterogeneous symptoms “cannot be treated with a single drug,” he said. Rather different drugs targeting each symptom individually is essential for effective management.
Dr. Nasrallah, who chaired the Psychopharmacology Update meeting, has made this point many times in the past, including in his role as the editor of Current Psychiatry. In one editorial 10 years ago, he wrote that “it makes little sense for the FDA to mandate that a drug must work for a DSM diagnosis instead of specific symptoms.”
“The FDA must update its old policy, which has led to the widespread off-label use of psychiatric drugs, an artificial concept, simply because the FDA arbitrarily decided a long time ago that new drugs must be approved for a specific DSM diagnosis,” Dr. Nasrallah said.
Dr. Stahl reported financial relationships with more than 20 pharmaceutical companies, including those that are involved in the development of drugs included in his talk. Medscape Live and this news organization are owned by the same parent company.
Many years ago, drug development in psychiatry turned to control of specific symptoms across disorders rather than within disorders, but regulatory agencies are still not yet on board, according to an expert psychopharmacologist outlining the ongoing evolution at the virtual Psychopharmacology Update presented by Current Psychiatry and the American Academy of Clinical Psychiatrists, sponsored by Medscape Live.
If this reorientation is going to lead to the broad indications the newer drugs likely deserve, which is control of specific types of symptoms regardless of the diagnosis, “we have to move the [Food and Drug Administration] along,” said Stephen M. Stahl, MD, PhD, chairman of the Neuroscience Institute and an adjunct professor of psychiatry at the University of California, San Diego.
On the side of drug development and clinical practice, the reorientation has already taken place. Dr. Stahl described numerous brain circuits known to produce symptoms when function is altered that are now treatment targets. This includes the ventral medial prefrontal cortex where deficient information processing leads to depression and the orbital frontal cortex where altered function leads to impulsivity.
“It is not like each part of the brain does a little bit of everything. Rather, each part of the brain has an assignment and duty and function,” Dr. Stahl explained. By addressing the disturbed signaling in brain circuits that lead to depression, impulsivity, agitation, or other symptoms, there is an opportunity for control, regardless of the psychiatric diagnosis with which the symptom is associated.
For example, Dr. Stahl predicted that pimavanserin, a highly selective 5-HT2A inverse agonist that is already approved for psychosis in Parkinson’s disease, is now likely to be approved for psychosis associated with other conditions on the basis of recent positive clinical studies in these other disorders.
Brexpiprazole, a serotonin-dopamine activity modulator already known to be useful for control of the agitation characteristic of schizophrenia, is now showing the same type of activity against agitation when it is associated with Alzheimer’s disease. Again, Dr. Stahl thinks this drug is on course for an indication across diseases once studies are conducted in each disease individually.
Another drug being evaluated for agitation, the N-methyl-D-aspartate receptor antagonist dextromethorphan bupropion, is also being tested for treatment of symptoms across multiple disorders, he reported.
However, the FDA has so far taken the position that each drug must be tested separately for a given symptom in each disorder for which it is being considered despite the underlying premise that it is the symptom, not the disease, that is important.
Unlike physiological diseases where symptoms, like a fever or abdominal cramps, are the product of a disease, psychiatric symptoms are the disease and a fundamental target – regardless of the DSM-based diagnosis.
To some degree, the symptoms of psychiatric disorders have always been the focus of treatment, but a pivot toward developing therapies that will control a symptom regardless of the underlying diagnosis is an important conceptual change. It is being made possible by advances in the detail with which the neuropathology of these symptoms is understood .
“By my count, 79 symptoms are described in DSM-5, but they are spread across hundreds of syndromes because they are grouped together in different ways,” Dr. Stahl observed.
He noted that clinicians make a diagnosis on the basis symptom groupings, but their interventions are selected to address the manifestations of the disease, not the disease itself.
“If you are a real psychopharmacologist treating real patients, you are treating the specific symptoms of the specific patient,” according to Dr. Stahl.
So far, the FDA has not made this leap, insisting on trials in these categorical disorders rather than permitting trial designs that allow benefit to be demonstrated against a symptom regardless of the syndrome with which it is associated.
Of egregious examples, Dr. Stahl recounted a recent trial of a 5-HT2 antagonist that looked so promising against psychosis in Alzheimer’s disease that the trialists enrolled patients with psychosis regardless of type of dementia, such as vascular dementia and Lewy body disease. The efficacy was impressive.
“It worked so well that they stopped the trial, but the FDA declined to approve it,” Dr. Stahl recounted. Despite clear evidence of benefit, the regulators insisted that the investigators needed to show a significant benefit in each condition individually.
While the trial investigators acknowledged that there was not enough power in the trial to show a statistically significant benefit in each category, they argued that the overall benefit and the consistent response across categories required them to stop the trial for ethical reasons.
“That’s your problem, the FDA said to the investigators,” according to Dr. Stahl.
The failure of the FDA to recognize the efficacy of psychopharmacologic therapies across symptoms regardless of the associated disease is a failure to stay current with an important evolution in medicine, Dr. Stahl indicated.
“What we have come to understand is the neurobiology of any given symptom is likely to be the same across disorders,” he said.
Agency’s arbitrary decisions cited
“I completely agree with Dr. Stahl,” said Henry A. Nasrallah, MD, professor of psychiatry, neurology, and neuroscience, University of Cincinnati.
In addition to the fact that symptoms are present across multiple categories, many patients manifest multiple symptoms at one time, Dr. Nasrallah pointed out. For neurodegenerative disorders associated with psychosis, depression, anxiety, aggression, and other symptoms, it is already well known that the heterogeneous symptoms “cannot be treated with a single drug,” he said. Rather different drugs targeting each symptom individually is essential for effective management.
Dr. Nasrallah, who chaired the Psychopharmacology Update meeting, has made this point many times in the past, including in his role as the editor of Current Psychiatry. In one editorial 10 years ago, he wrote that “it makes little sense for the FDA to mandate that a drug must work for a DSM diagnosis instead of specific symptoms.”
“The FDA must update its old policy, which has led to the widespread off-label use of psychiatric drugs, an artificial concept, simply because the FDA arbitrarily decided a long time ago that new drugs must be approved for a specific DSM diagnosis,” Dr. Nasrallah said.
Dr. Stahl reported financial relationships with more than 20 pharmaceutical companies, including those that are involved in the development of drugs included in his talk. Medscape Live and this news organization are owned by the same parent company.
Many years ago, drug development in psychiatry turned to control of specific symptoms across disorders rather than within disorders, but regulatory agencies are still not yet on board, according to an expert psychopharmacologist outlining the ongoing evolution at the virtual Psychopharmacology Update presented by Current Psychiatry and the American Academy of Clinical Psychiatrists, sponsored by Medscape Live.
If this reorientation is going to lead to the broad indications the newer drugs likely deserve, which is control of specific types of symptoms regardless of the diagnosis, “we have to move the [Food and Drug Administration] along,” said Stephen M. Stahl, MD, PhD, chairman of the Neuroscience Institute and an adjunct professor of psychiatry at the University of California, San Diego.
On the side of drug development and clinical practice, the reorientation has already taken place. Dr. Stahl described numerous brain circuits known to produce symptoms when function is altered that are now treatment targets. This includes the ventral medial prefrontal cortex where deficient information processing leads to depression and the orbital frontal cortex where altered function leads to impulsivity.
“It is not like each part of the brain does a little bit of everything. Rather, each part of the brain has an assignment and duty and function,” Dr. Stahl explained. By addressing the disturbed signaling in brain circuits that lead to depression, impulsivity, agitation, or other symptoms, there is an opportunity for control, regardless of the psychiatric diagnosis with which the symptom is associated.
For example, Dr. Stahl predicted that pimavanserin, a highly selective 5-HT2A inverse agonist that is already approved for psychosis in Parkinson’s disease, is now likely to be approved for psychosis associated with other conditions on the basis of recent positive clinical studies in these other disorders.
Brexpiprazole, a serotonin-dopamine activity modulator already known to be useful for control of the agitation characteristic of schizophrenia, is now showing the same type of activity against agitation when it is associated with Alzheimer’s disease. Again, Dr. Stahl thinks this drug is on course for an indication across diseases once studies are conducted in each disease individually.
Another drug being evaluated for agitation, the N-methyl-D-aspartate receptor antagonist dextromethorphan bupropion, is also being tested for treatment of symptoms across multiple disorders, he reported.
However, the FDA has so far taken the position that each drug must be tested separately for a given symptom in each disorder for which it is being considered despite the underlying premise that it is the symptom, not the disease, that is important.
Unlike physiological diseases where symptoms, like a fever or abdominal cramps, are the product of a disease, psychiatric symptoms are the disease and a fundamental target – regardless of the DSM-based diagnosis.
To some degree, the symptoms of psychiatric disorders have always been the focus of treatment, but a pivot toward developing therapies that will control a symptom regardless of the underlying diagnosis is an important conceptual change. It is being made possible by advances in the detail with which the neuropathology of these symptoms is understood .
“By my count, 79 symptoms are described in DSM-5, but they are spread across hundreds of syndromes because they are grouped together in different ways,” Dr. Stahl observed.
He noted that clinicians make a diagnosis on the basis symptom groupings, but their interventions are selected to address the manifestations of the disease, not the disease itself.
“If you are a real psychopharmacologist treating real patients, you are treating the specific symptoms of the specific patient,” according to Dr. Stahl.
So far, the FDA has not made this leap, insisting on trials in these categorical disorders rather than permitting trial designs that allow benefit to be demonstrated against a symptom regardless of the syndrome with which it is associated.
Of egregious examples, Dr. Stahl recounted a recent trial of a 5-HT2 antagonist that looked so promising against psychosis in Alzheimer’s disease that the trialists enrolled patients with psychosis regardless of type of dementia, such as vascular dementia and Lewy body disease. The efficacy was impressive.
“It worked so well that they stopped the trial, but the FDA declined to approve it,” Dr. Stahl recounted. Despite clear evidence of benefit, the regulators insisted that the investigators needed to show a significant benefit in each condition individually.
While the trial investigators acknowledged that there was not enough power in the trial to show a statistically significant benefit in each category, they argued that the overall benefit and the consistent response across categories required them to stop the trial for ethical reasons.
“That’s your problem, the FDA said to the investigators,” according to Dr. Stahl.
The failure of the FDA to recognize the efficacy of psychopharmacologic therapies across symptoms regardless of the associated disease is a failure to stay current with an important evolution in medicine, Dr. Stahl indicated.
“What we have come to understand is the neurobiology of any given symptom is likely to be the same across disorders,” he said.
Agency’s arbitrary decisions cited
“I completely agree with Dr. Stahl,” said Henry A. Nasrallah, MD, professor of psychiatry, neurology, and neuroscience, University of Cincinnati.
In addition to the fact that symptoms are present across multiple categories, many patients manifest multiple symptoms at one time, Dr. Nasrallah pointed out. For neurodegenerative disorders associated with psychosis, depression, anxiety, aggression, and other symptoms, it is already well known that the heterogeneous symptoms “cannot be treated with a single drug,” he said. Rather different drugs targeting each symptom individually is essential for effective management.
Dr. Nasrallah, who chaired the Psychopharmacology Update meeting, has made this point many times in the past, including in his role as the editor of Current Psychiatry. In one editorial 10 years ago, he wrote that “it makes little sense for the FDA to mandate that a drug must work for a DSM diagnosis instead of specific symptoms.”
“The FDA must update its old policy, which has led to the widespread off-label use of psychiatric drugs, an artificial concept, simply because the FDA arbitrarily decided a long time ago that new drugs must be approved for a specific DSM diagnosis,” Dr. Nasrallah said.
Dr. Stahl reported financial relationships with more than 20 pharmaceutical companies, including those that are involved in the development of drugs included in his talk. Medscape Live and this news organization are owned by the same parent company.
FROM PSYCHOPHARMACOLOGY UPDATE
Influenza tied to long-term increased risk for Parkinson’s disease
Influenza infection is linked to a subsequent diagnosis of Parkinson’s disease (PD) more than 10 years later, resurfacing a long-held debate about whether infection increases the risk for movement disorders over the long term.
In a large case-control study, investigators found and by more than 70% for PD occurring more than 10 years after the flu.
“This study is not definitive by any means, but it certainly suggests there are potential long-term consequences from influenza,” study investigator Noelle M. Cocoros, DSc, research scientist at Harvard Pilgrim Health Care Institute and Harvard Medical School, Boston, said in an interview.
The study was published online Oct. 25 in JAMA Neurology.
Ongoing debate
The debate about whether influenza is associated with PD has been going on as far back as the 1918 influenza pandemic, when experts documented parkinsonism in affected individuals.
Using data from the Danish patient registry, researchers identified 10,271 subjects diagnosed with PD during a 17-year period (2000-2016). Of these, 38.7% were female, and the mean age was 71.4 years.
They matched these subjects for age and sex to 51,355 controls without PD. Compared with controls, slightly fewer individuals with PD had chronic obstructive pulmonary disease (COPD) or emphysema, but there was a similar distribution of cardiovascular disease and various other conditions.
Researchers collected data on influenza diagnoses from inpatient and outpatient hospital clinics from 1977 to 2016. They plotted these by month and year on a graph, calculated the median number of diagnoses per month, and identified peaks as those with more than threefold the median.
They categorized cases in groups related to the time between the infection and PD: More than 10 years, 10-15 years, and more than 15 years.
The time lapse accounts for a rather long “run-up” to PD, said Dr. Cocoros. There’s a sometimes decades-long preclinical phase before patients develop typical motor signs and a prodromal phase where they may present with nonmotor symptoms such as sleep disorders and constipation.
“We expected there would be at least 10 years between any infection and PD if there was an association present,” said Dr. Cocoros.
Investigators found an association between influenza exposure and PD diagnosis “that held up over time,” she said.
For more than 10 years before PD, the likelihood of a diagnosis for the infected compared with the unexposed was increased 73% (odds ratio [OR] 1.73; 95% confidence interval, 1.11-2.71; P = .02) after adjustment for cardiovascular disease, diabetes, chronic obstructive pulmonary disease, emphysema, lung cancer, Crohn’s disease, and ulcerative colitis.
The odds increased with more time from infection. For more than 15 years, the adjusted OR was 1.91 (95% CI, 1.14 - 3.19; P =.01).
However, for the 10- to 15-year time frame, the point estimate was reduced and the CI nonsignificant (OR, 1.33; 95% CI, 0.54-3.27; P = .53). This “is a little hard to interpret,” but could be a result of the small numbers, exposure misclassification, or because “the longer time interval is what’s meaningful,” said Dr. Cocoros.
Potential COVID-19–related PD surge?
In a sensitivity analysis, researchers looked at peak infection activity. “We wanted to increase the likelihood of these diagnoses representing actual infection,” Dr. Cocoros noted.
Here, the OR was still elevated at more than 10 years, but the CI was quite wide and included 1 (OR, 1.52; 95% CI, 0.80-2.89; P = .21). “So the association holds up, but the estimates are quite unstable,” said Dr. Cocoros.
Researchers examined associations with numerous other infection types, but did not see the same trend over time. Some infections – for example, gastrointestinal infections and septicemia – were associated with PD within 5 years, but most associations appeared to be null after more than 10 years.
“There seemed to be associations earlier between the infection and PD, which we interpret to suggest there’s actually not a meaningful association,” said Dr. Cocoros.
An exception might be urinary tract infections (UTIs), where after 10 years, the adjusted OR was 1.19 (95% CI, 1.01-1.40). Research suggests patients with PD often have UTIs and neurogenic bladder.
“It’s possible that UTIs could be an early symptom of PD rather than a causative factor,” said Dr. Cocoros.
It’s unclear how influenza might lead to PD but it could be that the virus gets into the central nervous system, resulting in neuroinflammation. Cytokines generated in response to the influenza infection might damage the brain.
“The infection could be a ‘primer’ or an initial ‘hit’ to the system, maybe setting people up for PD,” said Dr. Cocoros.
As for the current COVID-19 pandemic, some experts are concerned about a potential surge in PD cases in decades to come, and are calling for prospective monitoring of patients with this infection, said Dr. Cocoros.
However, she noted that infections don’t account for all PD cases and that genetic and environmental factors also influence risk.
Many individuals who contract influenza don’t seek medical care or get tested, so it’s possible the study counted those who had the infection as unexposed. Another potential study limitation was that small numbers for some infections, for example, Helicobacter pylori and hepatitis C, limited the ability to interpret results.
‘Exciting and important’ findings
Commenting on the research for this news organization, Aparna Wagle Shukla, MD, professor, Norman Fixel Institute for Neurological Diseases, University of Florida, Gainesville, said the results amid the current pandemic are “exciting and important” and “have reinvigorated interest” in the role of infection in PD.
However, the study had some limitations, an important one being lack of accounting for confounding factors, including environmental factors, she said. Exposure to pesticides, living in a rural area, drinking well water, and having had a head injury may increase PD risk, whereas high intake of caffeine, nicotine, alcohol, and nonsteroidal anti-inflammatory drugs might lower the risk.
The researchers did not take into account exposure to multiple microbes or “infection burden,” said Dr. Wagle Shukla, who was not involved in the current study. In addition, as the data are from a single country with exposure to specific influenza strains, application of the findings elsewhere may be limited.
Dr. Wagle Shukla noted that a case-control design “isn’t ideal” from an epidemiological perspective. “Future studies should involve large cohorts followed longitudinally.”
The study was supported by grants from the Lundbeck Foundation and the Augustinus Foundation. Dr. Cocoros has disclosed no relevant financial relationships. Several coauthors have disclosed relationships with industry. The full list can be found with the original article.
A version of this article first appeared on Medscape.com.
Influenza infection is linked to a subsequent diagnosis of Parkinson’s disease (PD) more than 10 years later, resurfacing a long-held debate about whether infection increases the risk for movement disorders over the long term.
In a large case-control study, investigators found and by more than 70% for PD occurring more than 10 years after the flu.
“This study is not definitive by any means, but it certainly suggests there are potential long-term consequences from influenza,” study investigator Noelle M. Cocoros, DSc, research scientist at Harvard Pilgrim Health Care Institute and Harvard Medical School, Boston, said in an interview.
The study was published online Oct. 25 in JAMA Neurology.
Ongoing debate
The debate about whether influenza is associated with PD has been going on as far back as the 1918 influenza pandemic, when experts documented parkinsonism in affected individuals.
Using data from the Danish patient registry, researchers identified 10,271 subjects diagnosed with PD during a 17-year period (2000-2016). Of these, 38.7% were female, and the mean age was 71.4 years.
They matched these subjects for age and sex to 51,355 controls without PD. Compared with controls, slightly fewer individuals with PD had chronic obstructive pulmonary disease (COPD) or emphysema, but there was a similar distribution of cardiovascular disease and various other conditions.
Researchers collected data on influenza diagnoses from inpatient and outpatient hospital clinics from 1977 to 2016. They plotted these by month and year on a graph, calculated the median number of diagnoses per month, and identified peaks as those with more than threefold the median.
They categorized cases in groups related to the time between the infection and PD: More than 10 years, 10-15 years, and more than 15 years.
The time lapse accounts for a rather long “run-up” to PD, said Dr. Cocoros. There’s a sometimes decades-long preclinical phase before patients develop typical motor signs and a prodromal phase where they may present with nonmotor symptoms such as sleep disorders and constipation.
“We expected there would be at least 10 years between any infection and PD if there was an association present,” said Dr. Cocoros.
Investigators found an association between influenza exposure and PD diagnosis “that held up over time,” she said.
For more than 10 years before PD, the likelihood of a diagnosis for the infected compared with the unexposed was increased 73% (odds ratio [OR] 1.73; 95% confidence interval, 1.11-2.71; P = .02) after adjustment for cardiovascular disease, diabetes, chronic obstructive pulmonary disease, emphysema, lung cancer, Crohn’s disease, and ulcerative colitis.
The odds increased with more time from infection. For more than 15 years, the adjusted OR was 1.91 (95% CI, 1.14 - 3.19; P =.01).
However, for the 10- to 15-year time frame, the point estimate was reduced and the CI nonsignificant (OR, 1.33; 95% CI, 0.54-3.27; P = .53). This “is a little hard to interpret,” but could be a result of the small numbers, exposure misclassification, or because “the longer time interval is what’s meaningful,” said Dr. Cocoros.
Potential COVID-19–related PD surge?
In a sensitivity analysis, researchers looked at peak infection activity. “We wanted to increase the likelihood of these diagnoses representing actual infection,” Dr. Cocoros noted.
Here, the OR was still elevated at more than 10 years, but the CI was quite wide and included 1 (OR, 1.52; 95% CI, 0.80-2.89; P = .21). “So the association holds up, but the estimates are quite unstable,” said Dr. Cocoros.
Researchers examined associations with numerous other infection types, but did not see the same trend over time. Some infections – for example, gastrointestinal infections and septicemia – were associated with PD within 5 years, but most associations appeared to be null after more than 10 years.
“There seemed to be associations earlier between the infection and PD, which we interpret to suggest there’s actually not a meaningful association,” said Dr. Cocoros.
An exception might be urinary tract infections (UTIs), where after 10 years, the adjusted OR was 1.19 (95% CI, 1.01-1.40). Research suggests patients with PD often have UTIs and neurogenic bladder.
“It’s possible that UTIs could be an early symptom of PD rather than a causative factor,” said Dr. Cocoros.
It’s unclear how influenza might lead to PD but it could be that the virus gets into the central nervous system, resulting in neuroinflammation. Cytokines generated in response to the influenza infection might damage the brain.
“The infection could be a ‘primer’ or an initial ‘hit’ to the system, maybe setting people up for PD,” said Dr. Cocoros.
As for the current COVID-19 pandemic, some experts are concerned about a potential surge in PD cases in decades to come, and are calling for prospective monitoring of patients with this infection, said Dr. Cocoros.
However, she noted that infections don’t account for all PD cases and that genetic and environmental factors also influence risk.
Many individuals who contract influenza don’t seek medical care or get tested, so it’s possible the study counted those who had the infection as unexposed. Another potential study limitation was that small numbers for some infections, for example, Helicobacter pylori and hepatitis C, limited the ability to interpret results.
‘Exciting and important’ findings
Commenting on the research for this news organization, Aparna Wagle Shukla, MD, professor, Norman Fixel Institute for Neurological Diseases, University of Florida, Gainesville, said the results amid the current pandemic are “exciting and important” and “have reinvigorated interest” in the role of infection in PD.
However, the study had some limitations, an important one being lack of accounting for confounding factors, including environmental factors, she said. Exposure to pesticides, living in a rural area, drinking well water, and having had a head injury may increase PD risk, whereas high intake of caffeine, nicotine, alcohol, and nonsteroidal anti-inflammatory drugs might lower the risk.
The researchers did not take into account exposure to multiple microbes or “infection burden,” said Dr. Wagle Shukla, who was not involved in the current study. In addition, as the data are from a single country with exposure to specific influenza strains, application of the findings elsewhere may be limited.
Dr. Wagle Shukla noted that a case-control design “isn’t ideal” from an epidemiological perspective. “Future studies should involve large cohorts followed longitudinally.”
The study was supported by grants from the Lundbeck Foundation and the Augustinus Foundation. Dr. Cocoros has disclosed no relevant financial relationships. Several coauthors have disclosed relationships with industry. The full list can be found with the original article.
A version of this article first appeared on Medscape.com.
Influenza infection is linked to a subsequent diagnosis of Parkinson’s disease (PD) more than 10 years later, resurfacing a long-held debate about whether infection increases the risk for movement disorders over the long term.
In a large case-control study, investigators found and by more than 70% for PD occurring more than 10 years after the flu.
“This study is not definitive by any means, but it certainly suggests there are potential long-term consequences from influenza,” study investigator Noelle M. Cocoros, DSc, research scientist at Harvard Pilgrim Health Care Institute and Harvard Medical School, Boston, said in an interview.
The study was published online Oct. 25 in JAMA Neurology.
Ongoing debate
The debate about whether influenza is associated with PD has been going on as far back as the 1918 influenza pandemic, when experts documented parkinsonism in affected individuals.
Using data from the Danish patient registry, researchers identified 10,271 subjects diagnosed with PD during a 17-year period (2000-2016). Of these, 38.7% were female, and the mean age was 71.4 years.
They matched these subjects for age and sex to 51,355 controls without PD. Compared with controls, slightly fewer individuals with PD had chronic obstructive pulmonary disease (COPD) or emphysema, but there was a similar distribution of cardiovascular disease and various other conditions.
Researchers collected data on influenza diagnoses from inpatient and outpatient hospital clinics from 1977 to 2016. They plotted these by month and year on a graph, calculated the median number of diagnoses per month, and identified peaks as those with more than threefold the median.
They categorized cases in groups related to the time between the infection and PD: More than 10 years, 10-15 years, and more than 15 years.
The time lapse accounts for a rather long “run-up” to PD, said Dr. Cocoros. There’s a sometimes decades-long preclinical phase before patients develop typical motor signs and a prodromal phase where they may present with nonmotor symptoms such as sleep disorders and constipation.
“We expected there would be at least 10 years between any infection and PD if there was an association present,” said Dr. Cocoros.
Investigators found an association between influenza exposure and PD diagnosis “that held up over time,” she said.
For more than 10 years before PD, the likelihood of a diagnosis for the infected compared with the unexposed was increased 73% (odds ratio [OR] 1.73; 95% confidence interval, 1.11-2.71; P = .02) after adjustment for cardiovascular disease, diabetes, chronic obstructive pulmonary disease, emphysema, lung cancer, Crohn’s disease, and ulcerative colitis.
The odds increased with more time from infection. For more than 15 years, the adjusted OR was 1.91 (95% CI, 1.14 - 3.19; P =.01).
However, for the 10- to 15-year time frame, the point estimate was reduced and the CI nonsignificant (OR, 1.33; 95% CI, 0.54-3.27; P = .53). This “is a little hard to interpret,” but could be a result of the small numbers, exposure misclassification, or because “the longer time interval is what’s meaningful,” said Dr. Cocoros.
Potential COVID-19–related PD surge?
In a sensitivity analysis, researchers looked at peak infection activity. “We wanted to increase the likelihood of these diagnoses representing actual infection,” Dr. Cocoros noted.
Here, the OR was still elevated at more than 10 years, but the CI was quite wide and included 1 (OR, 1.52; 95% CI, 0.80-2.89; P = .21). “So the association holds up, but the estimates are quite unstable,” said Dr. Cocoros.
Researchers examined associations with numerous other infection types, but did not see the same trend over time. Some infections – for example, gastrointestinal infections and septicemia – were associated with PD within 5 years, but most associations appeared to be null after more than 10 years.
“There seemed to be associations earlier between the infection and PD, which we interpret to suggest there’s actually not a meaningful association,” said Dr. Cocoros.
An exception might be urinary tract infections (UTIs), where after 10 years, the adjusted OR was 1.19 (95% CI, 1.01-1.40). Research suggests patients with PD often have UTIs and neurogenic bladder.
“It’s possible that UTIs could be an early symptom of PD rather than a causative factor,” said Dr. Cocoros.
It’s unclear how influenza might lead to PD but it could be that the virus gets into the central nervous system, resulting in neuroinflammation. Cytokines generated in response to the influenza infection might damage the brain.
“The infection could be a ‘primer’ or an initial ‘hit’ to the system, maybe setting people up for PD,” said Dr. Cocoros.
As for the current COVID-19 pandemic, some experts are concerned about a potential surge in PD cases in decades to come, and are calling for prospective monitoring of patients with this infection, said Dr. Cocoros.
However, she noted that infections don’t account for all PD cases and that genetic and environmental factors also influence risk.
Many individuals who contract influenza don’t seek medical care or get tested, so it’s possible the study counted those who had the infection as unexposed. Another potential study limitation was that small numbers for some infections, for example, Helicobacter pylori and hepatitis C, limited the ability to interpret results.
‘Exciting and important’ findings
Commenting on the research for this news organization, Aparna Wagle Shukla, MD, professor, Norman Fixel Institute for Neurological Diseases, University of Florida, Gainesville, said the results amid the current pandemic are “exciting and important” and “have reinvigorated interest” in the role of infection in PD.
However, the study had some limitations, an important one being lack of accounting for confounding factors, including environmental factors, she said. Exposure to pesticides, living in a rural area, drinking well water, and having had a head injury may increase PD risk, whereas high intake of caffeine, nicotine, alcohol, and nonsteroidal anti-inflammatory drugs might lower the risk.
The researchers did not take into account exposure to multiple microbes or “infection burden,” said Dr. Wagle Shukla, who was not involved in the current study. In addition, as the data are from a single country with exposure to specific influenza strains, application of the findings elsewhere may be limited.
Dr. Wagle Shukla noted that a case-control design “isn’t ideal” from an epidemiological perspective. “Future studies should involve large cohorts followed longitudinally.”
The study was supported by grants from the Lundbeck Foundation and the Augustinus Foundation. Dr. Cocoros has disclosed no relevant financial relationships. Several coauthors have disclosed relationships with industry. The full list can be found with the original article.
A version of this article first appeared on Medscape.com.
Art therapy linked to slowed Parkinson’s progression
Adding art therapy to standard drug treatment in Parkinson’s disease (PD) not only improves severity of both motor and nonmotor symptoms, but also slows rates of disease progression, new research suggests.
Fifty PD patients were randomly assigned to receive either art therapy, including sculpting and drawing, plus drug therapy or drug therapy alone, and followed up over 12 months.
Patients receiving combined therapy experienced improvements in symptoms, depression, and cognitive scores, and had reduced tremor and daytime sleepiness. They were also substantially less likely to experience disease progression.
“The use of art therapy can reduce the severity of motor and nonmotor manifestations of Parkinson’s disease,” said study investigator Iryna Khubetova, MD, PhD, head of the neurology department, Odessa (Ukraine) Regional Clinical Hospital.
she added.
The findings were presented at the virtual congress of the European College of Neuropsychopharmacology.
A promising approach
Dr. Khubetova told this news organization that offering art therapy to PD patients was “very affordable,” especially as professional artists “provided materials for painting and other art supplies free of charge.”
“We hope this approach is very promising and would be widely adopted.”
She suggested the positive effect of art therapy could be related to “activating the brain’s reward neural network.”
This may be via improved visual attention acting on visuospatial mechanisms and emotional drive, with “activation of the medial orbitofrontal cortex, ventral striatum, and other structures.”
The researchers note PD, a “multisystem progressive neurodegenerative disease,” is among the three most common neurological disorders, with an incidence of 100-150 cases per 100,000 people.
They also note that nonpharmacologic approaches are “widely used” as an adjunct to drug therapy and as part of an “integrated approach” to disease management.
To examine the clinical efficacy of art therapy, the team recruited patients with PD who had preserved facility for independent movement, defined as stages 1-2.5 on the Hoehn and Yahr scale.
Patients were randomly assigned to art therapy sessions alongside standard drug therapy or to standard drug therapy alone. The art therapy included sculpting, free drawing, and coloring patterns.
Multiple benefits
Participants were assessed at baseline and at 6 and 12 months with the Unified Parkinson Disease Rating Scale (UPDRS), the Beck Depression Inventory, the Montreal Cognitive Assessment, and the Pegboard Test of finger dexterity.
Fifty patients were included in the study, with 30 assigned to standard drug therapy alone and 20 to the combined intervention. Participants had a mean age of 57.8 years, and 46% were women.
Over the study period, investigators found patients assigned to art therapy plus drug treatment had improved mood, as well as decreased daytime sleeping, reduced tremor, and a decrease in anxiety and fear intensity.
Between baseline and the 6- and 12-month assessments, patients in the combined therapy group showed improvements in scores on all of the questionnaires, and on the Pegboard Test. In contrast, scores were either stable or worsened in the standard drug therapy–alone group.
The team notes that there was also a marked difference in rates of disease progression, defined as a change on the Hoehn and Yahr scale of at least 0.5 points, between the two groups.
Only two (10%) patients in the combined drug and art therapy progressed over the study period, compared with 10 (33%) in the control group (P = .05).
The findings complement those of a recent study conducted by Alberto Cucca, MD, of the Fresco Institute for Parkinson’s and Movement Disorders, New York University, and colleagues.
Eighteen patients took part in the prospective, open-label trial. They were assessed before and after 20 sessions of art therapy on a range of measures.
Results revealed that following the art therapy, patients had improvements in the Navon Test (which assesses visual neglect, eye tracking, and UPDRS scores), as well as significantly increased functional connectivity levels in the visual cortex on resting-state functional MRI.
Many benefits, no side effects
Rebecca Gilbert, MD, PhD, vice president and chief scientific officer of the American Parkinson Disease Association, who was not involved in either study, told this news organization that the idea of art therapy for patients with Parkinson’s is “very reasonable.”
She highlighted that “people with Parkinson’s have many issues with their visuospatial abilities,” as well as their depth and distance perception, and so “enhancing that aspect could potentially be very beneficial.”
“So I’m hopeful that it’s a really good avenue to explore, and the preliminary data are very exciting.”
Dr. Gilbert also highlighted that the “wonderful” aspect of art therapy is that there are “so many benefits and not really any side effects.” Patients can “take the meds … and then enhance that with various therapies, and this would be an additional option.”
Another notable aspect of art therapy is the “social element” and the sense of “camaraderie,” although that has “to be teased out from the benefits you would get from the actual art therapy.”
Finally, Dr. Gilbert pointed out that the difference between the current trial and Dr. Cucca’s trial is the presence of a control group.
“Of course, it’s not blinded, because you know whether you got therapy or not … but that extra element of being able to compare with a group that didn’t get the treatment gives it a little more weight in terms of the field.”
No funding was declared. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Adding art therapy to standard drug treatment in Parkinson’s disease (PD) not only improves severity of both motor and nonmotor symptoms, but also slows rates of disease progression, new research suggests.
Fifty PD patients were randomly assigned to receive either art therapy, including sculpting and drawing, plus drug therapy or drug therapy alone, and followed up over 12 months.
Patients receiving combined therapy experienced improvements in symptoms, depression, and cognitive scores, and had reduced tremor and daytime sleepiness. They were also substantially less likely to experience disease progression.
“The use of art therapy can reduce the severity of motor and nonmotor manifestations of Parkinson’s disease,” said study investigator Iryna Khubetova, MD, PhD, head of the neurology department, Odessa (Ukraine) Regional Clinical Hospital.
she added.
The findings were presented at the virtual congress of the European College of Neuropsychopharmacology.
A promising approach
Dr. Khubetova told this news organization that offering art therapy to PD patients was “very affordable,” especially as professional artists “provided materials for painting and other art supplies free of charge.”
“We hope this approach is very promising and would be widely adopted.”
She suggested the positive effect of art therapy could be related to “activating the brain’s reward neural network.”
This may be via improved visual attention acting on visuospatial mechanisms and emotional drive, with “activation of the medial orbitofrontal cortex, ventral striatum, and other structures.”
The researchers note PD, a “multisystem progressive neurodegenerative disease,” is among the three most common neurological disorders, with an incidence of 100-150 cases per 100,000 people.
They also note that nonpharmacologic approaches are “widely used” as an adjunct to drug therapy and as part of an “integrated approach” to disease management.
To examine the clinical efficacy of art therapy, the team recruited patients with PD who had preserved facility for independent movement, defined as stages 1-2.5 on the Hoehn and Yahr scale.
Patients were randomly assigned to art therapy sessions alongside standard drug therapy or to standard drug therapy alone. The art therapy included sculpting, free drawing, and coloring patterns.
Multiple benefits
Participants were assessed at baseline and at 6 and 12 months with the Unified Parkinson Disease Rating Scale (UPDRS), the Beck Depression Inventory, the Montreal Cognitive Assessment, and the Pegboard Test of finger dexterity.
Fifty patients were included in the study, with 30 assigned to standard drug therapy alone and 20 to the combined intervention. Participants had a mean age of 57.8 years, and 46% were women.
Over the study period, investigators found patients assigned to art therapy plus drug treatment had improved mood, as well as decreased daytime sleeping, reduced tremor, and a decrease in anxiety and fear intensity.
Between baseline and the 6- and 12-month assessments, patients in the combined therapy group showed improvements in scores on all of the questionnaires, and on the Pegboard Test. In contrast, scores were either stable or worsened in the standard drug therapy–alone group.
The team notes that there was also a marked difference in rates of disease progression, defined as a change on the Hoehn and Yahr scale of at least 0.5 points, between the two groups.
Only two (10%) patients in the combined drug and art therapy progressed over the study period, compared with 10 (33%) in the control group (P = .05).
The findings complement those of a recent study conducted by Alberto Cucca, MD, of the Fresco Institute for Parkinson’s and Movement Disorders, New York University, and colleagues.
Eighteen patients took part in the prospective, open-label trial. They were assessed before and after 20 sessions of art therapy on a range of measures.
Results revealed that following the art therapy, patients had improvements in the Navon Test (which assesses visual neglect, eye tracking, and UPDRS scores), as well as significantly increased functional connectivity levels in the visual cortex on resting-state functional MRI.
Many benefits, no side effects
Rebecca Gilbert, MD, PhD, vice president and chief scientific officer of the American Parkinson Disease Association, who was not involved in either study, told this news organization that the idea of art therapy for patients with Parkinson’s is “very reasonable.”
She highlighted that “people with Parkinson’s have many issues with their visuospatial abilities,” as well as their depth and distance perception, and so “enhancing that aspect could potentially be very beneficial.”
“So I’m hopeful that it’s a really good avenue to explore, and the preliminary data are very exciting.”
Dr. Gilbert also highlighted that the “wonderful” aspect of art therapy is that there are “so many benefits and not really any side effects.” Patients can “take the meds … and then enhance that with various therapies, and this would be an additional option.”
Another notable aspect of art therapy is the “social element” and the sense of “camaraderie,” although that has “to be teased out from the benefits you would get from the actual art therapy.”
Finally, Dr. Gilbert pointed out that the difference between the current trial and Dr. Cucca’s trial is the presence of a control group.
“Of course, it’s not blinded, because you know whether you got therapy or not … but that extra element of being able to compare with a group that didn’t get the treatment gives it a little more weight in terms of the field.”
No funding was declared. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Adding art therapy to standard drug treatment in Parkinson’s disease (PD) not only improves severity of both motor and nonmotor symptoms, but also slows rates of disease progression, new research suggests.
Fifty PD patients were randomly assigned to receive either art therapy, including sculpting and drawing, plus drug therapy or drug therapy alone, and followed up over 12 months.
Patients receiving combined therapy experienced improvements in symptoms, depression, and cognitive scores, and had reduced tremor and daytime sleepiness. They were also substantially less likely to experience disease progression.
“The use of art therapy can reduce the severity of motor and nonmotor manifestations of Parkinson’s disease,” said study investigator Iryna Khubetova, MD, PhD, head of the neurology department, Odessa (Ukraine) Regional Clinical Hospital.
she added.
The findings were presented at the virtual congress of the European College of Neuropsychopharmacology.
A promising approach
Dr. Khubetova told this news organization that offering art therapy to PD patients was “very affordable,” especially as professional artists “provided materials for painting and other art supplies free of charge.”
“We hope this approach is very promising and would be widely adopted.”
She suggested the positive effect of art therapy could be related to “activating the brain’s reward neural network.”
This may be via improved visual attention acting on visuospatial mechanisms and emotional drive, with “activation of the medial orbitofrontal cortex, ventral striatum, and other structures.”
The researchers note PD, a “multisystem progressive neurodegenerative disease,” is among the three most common neurological disorders, with an incidence of 100-150 cases per 100,000 people.
They also note that nonpharmacologic approaches are “widely used” as an adjunct to drug therapy and as part of an “integrated approach” to disease management.
To examine the clinical efficacy of art therapy, the team recruited patients with PD who had preserved facility for independent movement, defined as stages 1-2.5 on the Hoehn and Yahr scale.
Patients were randomly assigned to art therapy sessions alongside standard drug therapy or to standard drug therapy alone. The art therapy included sculpting, free drawing, and coloring patterns.
Multiple benefits
Participants were assessed at baseline and at 6 and 12 months with the Unified Parkinson Disease Rating Scale (UPDRS), the Beck Depression Inventory, the Montreal Cognitive Assessment, and the Pegboard Test of finger dexterity.
Fifty patients were included in the study, with 30 assigned to standard drug therapy alone and 20 to the combined intervention. Participants had a mean age of 57.8 years, and 46% were women.
Over the study period, investigators found patients assigned to art therapy plus drug treatment had improved mood, as well as decreased daytime sleeping, reduced tremor, and a decrease in anxiety and fear intensity.
Between baseline and the 6- and 12-month assessments, patients in the combined therapy group showed improvements in scores on all of the questionnaires, and on the Pegboard Test. In contrast, scores were either stable or worsened in the standard drug therapy–alone group.
The team notes that there was also a marked difference in rates of disease progression, defined as a change on the Hoehn and Yahr scale of at least 0.5 points, between the two groups.
Only two (10%) patients in the combined drug and art therapy progressed over the study period, compared with 10 (33%) in the control group (P = .05).
The findings complement those of a recent study conducted by Alberto Cucca, MD, of the Fresco Institute for Parkinson’s and Movement Disorders, New York University, and colleagues.
Eighteen patients took part in the prospective, open-label trial. They were assessed before and after 20 sessions of art therapy on a range of measures.
Results revealed that following the art therapy, patients had improvements in the Navon Test (which assesses visual neglect, eye tracking, and UPDRS scores), as well as significantly increased functional connectivity levels in the visual cortex on resting-state functional MRI.
Many benefits, no side effects
Rebecca Gilbert, MD, PhD, vice president and chief scientific officer of the American Parkinson Disease Association, who was not involved in either study, told this news organization that the idea of art therapy for patients with Parkinson’s is “very reasonable.”
She highlighted that “people with Parkinson’s have many issues with their visuospatial abilities,” as well as their depth and distance perception, and so “enhancing that aspect could potentially be very beneficial.”
“So I’m hopeful that it’s a really good avenue to explore, and the preliminary data are very exciting.”
Dr. Gilbert also highlighted that the “wonderful” aspect of art therapy is that there are “so many benefits and not really any side effects.” Patients can “take the meds … and then enhance that with various therapies, and this would be an additional option.”
Another notable aspect of art therapy is the “social element” and the sense of “camaraderie,” although that has “to be teased out from the benefits you would get from the actual art therapy.”
Finally, Dr. Gilbert pointed out that the difference between the current trial and Dr. Cucca’s trial is the presence of a control group.
“Of course, it’s not blinded, because you know whether you got therapy or not … but that extra element of being able to compare with a group that didn’t get the treatment gives it a little more weight in terms of the field.”
No funding was declared. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ECNP 2021
Customized brain stimulation: New hope for severe depression
Personalized deep brain stimulation (DBS) appears to rapidly and effectively improve symptoms of treatment-resistant depression, new research suggests.
In a proof-of-concept study, investigators identified specific brain activity patterns responsible for a single patient’s severe depression and customized a DBS protocol to modulate the patterns. Results showed rapid and sustained improvement in depression scores.
“This study points the way to a new paradigm that is desperately needed in psychiatry,” Andrew Krystal, PhD, Weill Institute for Neurosciences, University of California, San Francisco, said in a news release.
“ by identifying and modulating the circuit in her brain that’s uniquely associated with her symptoms,” Dr. Krystal added.
The findings were published online Oct. 4 in Nature Medicine.
Closed-loop, on-demand stimulation
The patient was a 36-year-old woman with longstanding, severe, and treatment-resistant major depressive disorder. She was unresponsive to multiple antidepressant combinations and electroconvulsive therapy.
The researchers used intracranial electrophysiology and focal electrical stimulation to identify the specific pattern of electrical brain activity that correlated with her depressed mood.
They identified the right ventral striatum – which is involved in emotion, motivation, and reward – as the stimulation site that led to consistent, sustained, and dose-dependent improvement of symptoms and served as the neural biomarker.
In addition, the investigators identified a neural activity pattern in the amygdala that predicted both the mood symptoms, symptom severity, and stimulation efficacy.
The patient was implanted with the Food and Drug Administration–approved NeuroPace RNS System. The device was placed in the right hemisphere. A single sensing lead was positioned in the amygdala and the second stimulation lead was placed in the ventral striatum.
When the sensing lead detected the activity pattern associated with depression, the other lead delivered a tiny dose (1 milliampere/1 mA) of electricity for 6 seconds, which altered the neural activity and relieved mood symptoms.
Remission achieved
Once this personalized, closed-loop therapy was fully operational, the patient’s depression score on the Montgomery-Åsberg Depression Rating Scale (MADRS) dropped from 33 before turning treatment ON to 14 at the first ON-treatment assessment carried out after 12 days of stimulation. The score dropped below 10, representing remission, several months later.
The treatment also rapidly improved symptom severity, as measured daily with Hamilton Depression Rating Scale (HAMD-6) and visual analog scales.
“Success was predicated on a clinical mapping stage before chronic device placement, a strategy that has been utilized in epilepsy to map seizure foci in a personalized manner but has not previously been performed in other neuropsychiatric conditions,” the investigators wrote.
This patient represents “one of the first examples of precision psychiatry – a treatment tailored to an individual,” the study’s lead author, Katherine Scangos, MD, also with UCSF Weill Institute, said in an interview.
She added that the treatment “was personally tailored both spatially,” meaning at the brain location, and temporally – the time it was delivered.
“This is the first time a neural biomarker has been used to automatically trigger therapeutic stimulation in depression as a successful long-term treatment,” said Dr. Scangos. However, “we have a lot of work left to do,” she added.
“This study provides proof-of-principle that we can utilize a multimodal brain mapping approach to identify a personalized depression circuit and target that circuit with successful treatment. We will need to test the approach in more patients before we can determine its efficacy,” Dr. Scangos said.
First reliable biomarker in psychiatry
In a statement from the UK nonprofit Science Media Centre, Vladimir Litvak, PhD, with the Wellcome Centre for Human Neuroimaging, University College London, said that the study is interesting, noting that it is from “one of the leading groups in the field.”
The fact that depression symptoms can be treated in some patients by electrical stimulation of the ventral striatum is not new, Dr. Litvak said. However, what is “exciting” is that the authors identified a particular neural activity pattern in the amygdala as a reliable predictor of both symptom severity and stimulation effectiveness, he noted.
“Patterns of brain activity correlated with disease symptoms when testing over a large group of patients are commonly discovered. But there are just a handful of examples of patterns that are reliable enough to be predictive on a short time scale in a single patient,” said Dr. Litvak, who was not associated with the research.
“Furthermore, to my knowledge, this is the first example of such a reliable biomarker for psychiatric symptoms. The other examples were all for neurological disorders such as Parkinson’s disease, dystonia, and epilepsy,” he added.
He cautioned that this is a single case, but “if reproduced in additional patients, it will bring at least some psychiatric conditions into the domain of brain diseases that can be characterized and diagnosed objectively rather than based on symptoms alone.”
Dr. Litvak pointed out two other critical aspects of the study: the use of exploratory recordings and stimulation to determine the most effective treatment strategy, and the use of a closed-loop device that stimulates only when detecting the amygdala biomarker.
“It is hard to say based on this single case how important these will be in the future. There is no comparison to constant stimulation that might have worked as well because the implanted device used in the study is not suitable for that,” Dr. Litvak said.
It should also be noted that implanting multiple depth electrodes at different brain sites is a “traumatic invasive procedure only reserved to date for severe cases of drug-resistant epilepsy,” he said. “Furthermore, it only allows [researchers] to test a small number of candidate sites, so it relies heavily on prior knowledge.
“Once clinicians know better what to look for, it might be possible to avoid this procedure altogether by using noninvasive methods,” such as functional MRI or EEG, to match the right treatment option to a patient, Dr. Litvak concluded.
The research was funded by the National Institutes of Health, the Brain & Behavior Research Foundation, and the Ray and Dagmar Dolby Family Fund through the department of psychiatry at UCSF. Dr. Scangos has reported no relevant financial relationships. A complete list of author disclosures is available in the original article. Dr. Litvak is participating in a research funding application to search for electrophysiological biomarkers of depression symptoms using invasive recordings.
A version of this article first appeared on Medscape.com.
Personalized deep brain stimulation (DBS) appears to rapidly and effectively improve symptoms of treatment-resistant depression, new research suggests.
In a proof-of-concept study, investigators identified specific brain activity patterns responsible for a single patient’s severe depression and customized a DBS protocol to modulate the patterns. Results showed rapid and sustained improvement in depression scores.
“This study points the way to a new paradigm that is desperately needed in psychiatry,” Andrew Krystal, PhD, Weill Institute for Neurosciences, University of California, San Francisco, said in a news release.
“ by identifying and modulating the circuit in her brain that’s uniquely associated with her symptoms,” Dr. Krystal added.
The findings were published online Oct. 4 in Nature Medicine.
Closed-loop, on-demand stimulation
The patient was a 36-year-old woman with longstanding, severe, and treatment-resistant major depressive disorder. She was unresponsive to multiple antidepressant combinations and electroconvulsive therapy.
The researchers used intracranial electrophysiology and focal electrical stimulation to identify the specific pattern of electrical brain activity that correlated with her depressed mood.
They identified the right ventral striatum – which is involved in emotion, motivation, and reward – as the stimulation site that led to consistent, sustained, and dose-dependent improvement of symptoms and served as the neural biomarker.
In addition, the investigators identified a neural activity pattern in the amygdala that predicted both the mood symptoms, symptom severity, and stimulation efficacy.
The patient was implanted with the Food and Drug Administration–approved NeuroPace RNS System. The device was placed in the right hemisphere. A single sensing lead was positioned in the amygdala and the second stimulation lead was placed in the ventral striatum.
When the sensing lead detected the activity pattern associated with depression, the other lead delivered a tiny dose (1 milliampere/1 mA) of electricity for 6 seconds, which altered the neural activity and relieved mood symptoms.
Remission achieved
Once this personalized, closed-loop therapy was fully operational, the patient’s depression score on the Montgomery-Åsberg Depression Rating Scale (MADRS) dropped from 33 before turning treatment ON to 14 at the first ON-treatment assessment carried out after 12 days of stimulation. The score dropped below 10, representing remission, several months later.
The treatment also rapidly improved symptom severity, as measured daily with Hamilton Depression Rating Scale (HAMD-6) and visual analog scales.
“Success was predicated on a clinical mapping stage before chronic device placement, a strategy that has been utilized in epilepsy to map seizure foci in a personalized manner but has not previously been performed in other neuropsychiatric conditions,” the investigators wrote.
This patient represents “one of the first examples of precision psychiatry – a treatment tailored to an individual,” the study’s lead author, Katherine Scangos, MD, also with UCSF Weill Institute, said in an interview.
She added that the treatment “was personally tailored both spatially,” meaning at the brain location, and temporally – the time it was delivered.
“This is the first time a neural biomarker has been used to automatically trigger therapeutic stimulation in depression as a successful long-term treatment,” said Dr. Scangos. However, “we have a lot of work left to do,” she added.
“This study provides proof-of-principle that we can utilize a multimodal brain mapping approach to identify a personalized depression circuit and target that circuit with successful treatment. We will need to test the approach in more patients before we can determine its efficacy,” Dr. Scangos said.
First reliable biomarker in psychiatry
In a statement from the UK nonprofit Science Media Centre, Vladimir Litvak, PhD, with the Wellcome Centre for Human Neuroimaging, University College London, said that the study is interesting, noting that it is from “one of the leading groups in the field.”
The fact that depression symptoms can be treated in some patients by electrical stimulation of the ventral striatum is not new, Dr. Litvak said. However, what is “exciting” is that the authors identified a particular neural activity pattern in the amygdala as a reliable predictor of both symptom severity and stimulation effectiveness, he noted.
“Patterns of brain activity correlated with disease symptoms when testing over a large group of patients are commonly discovered. But there are just a handful of examples of patterns that are reliable enough to be predictive on a short time scale in a single patient,” said Dr. Litvak, who was not associated with the research.
“Furthermore, to my knowledge, this is the first example of such a reliable biomarker for psychiatric symptoms. The other examples were all for neurological disorders such as Parkinson’s disease, dystonia, and epilepsy,” he added.
He cautioned that this is a single case, but “if reproduced in additional patients, it will bring at least some psychiatric conditions into the domain of brain diseases that can be characterized and diagnosed objectively rather than based on symptoms alone.”
Dr. Litvak pointed out two other critical aspects of the study: the use of exploratory recordings and stimulation to determine the most effective treatment strategy, and the use of a closed-loop device that stimulates only when detecting the amygdala biomarker.
“It is hard to say based on this single case how important these will be in the future. There is no comparison to constant stimulation that might have worked as well because the implanted device used in the study is not suitable for that,” Dr. Litvak said.
It should also be noted that implanting multiple depth electrodes at different brain sites is a “traumatic invasive procedure only reserved to date for severe cases of drug-resistant epilepsy,” he said. “Furthermore, it only allows [researchers] to test a small number of candidate sites, so it relies heavily on prior knowledge.
“Once clinicians know better what to look for, it might be possible to avoid this procedure altogether by using noninvasive methods,” such as functional MRI or EEG, to match the right treatment option to a patient, Dr. Litvak concluded.
The research was funded by the National Institutes of Health, the Brain & Behavior Research Foundation, and the Ray and Dagmar Dolby Family Fund through the department of psychiatry at UCSF. Dr. Scangos has reported no relevant financial relationships. A complete list of author disclosures is available in the original article. Dr. Litvak is participating in a research funding application to search for electrophysiological biomarkers of depression symptoms using invasive recordings.
A version of this article first appeared on Medscape.com.
Personalized deep brain stimulation (DBS) appears to rapidly and effectively improve symptoms of treatment-resistant depression, new research suggests.
In a proof-of-concept study, investigators identified specific brain activity patterns responsible for a single patient’s severe depression and customized a DBS protocol to modulate the patterns. Results showed rapid and sustained improvement in depression scores.
“This study points the way to a new paradigm that is desperately needed in psychiatry,” Andrew Krystal, PhD, Weill Institute for Neurosciences, University of California, San Francisco, said in a news release.
“ by identifying and modulating the circuit in her brain that’s uniquely associated with her symptoms,” Dr. Krystal added.
The findings were published online Oct. 4 in Nature Medicine.
Closed-loop, on-demand stimulation
The patient was a 36-year-old woman with longstanding, severe, and treatment-resistant major depressive disorder. She was unresponsive to multiple antidepressant combinations and electroconvulsive therapy.
The researchers used intracranial electrophysiology and focal electrical stimulation to identify the specific pattern of electrical brain activity that correlated with her depressed mood.
They identified the right ventral striatum – which is involved in emotion, motivation, and reward – as the stimulation site that led to consistent, sustained, and dose-dependent improvement of symptoms and served as the neural biomarker.
In addition, the investigators identified a neural activity pattern in the amygdala that predicted both the mood symptoms, symptom severity, and stimulation efficacy.
The patient was implanted with the Food and Drug Administration–approved NeuroPace RNS System. The device was placed in the right hemisphere. A single sensing lead was positioned in the amygdala and the second stimulation lead was placed in the ventral striatum.
When the sensing lead detected the activity pattern associated with depression, the other lead delivered a tiny dose (1 milliampere/1 mA) of electricity for 6 seconds, which altered the neural activity and relieved mood symptoms.
Remission achieved
Once this personalized, closed-loop therapy was fully operational, the patient’s depression score on the Montgomery-Åsberg Depression Rating Scale (MADRS) dropped from 33 before turning treatment ON to 14 at the first ON-treatment assessment carried out after 12 days of stimulation. The score dropped below 10, representing remission, several months later.
The treatment also rapidly improved symptom severity, as measured daily with Hamilton Depression Rating Scale (HAMD-6) and visual analog scales.
“Success was predicated on a clinical mapping stage before chronic device placement, a strategy that has been utilized in epilepsy to map seizure foci in a personalized manner but has not previously been performed in other neuropsychiatric conditions,” the investigators wrote.
This patient represents “one of the first examples of precision psychiatry – a treatment tailored to an individual,” the study’s lead author, Katherine Scangos, MD, also with UCSF Weill Institute, said in an interview.
She added that the treatment “was personally tailored both spatially,” meaning at the brain location, and temporally – the time it was delivered.
“This is the first time a neural biomarker has been used to automatically trigger therapeutic stimulation in depression as a successful long-term treatment,” said Dr. Scangos. However, “we have a lot of work left to do,” she added.
“This study provides proof-of-principle that we can utilize a multimodal brain mapping approach to identify a personalized depression circuit and target that circuit with successful treatment. We will need to test the approach in more patients before we can determine its efficacy,” Dr. Scangos said.
First reliable biomarker in psychiatry
In a statement from the UK nonprofit Science Media Centre, Vladimir Litvak, PhD, with the Wellcome Centre for Human Neuroimaging, University College London, said that the study is interesting, noting that it is from “one of the leading groups in the field.”
The fact that depression symptoms can be treated in some patients by electrical stimulation of the ventral striatum is not new, Dr. Litvak said. However, what is “exciting” is that the authors identified a particular neural activity pattern in the amygdala as a reliable predictor of both symptom severity and stimulation effectiveness, he noted.
“Patterns of brain activity correlated with disease symptoms when testing over a large group of patients are commonly discovered. But there are just a handful of examples of patterns that are reliable enough to be predictive on a short time scale in a single patient,” said Dr. Litvak, who was not associated with the research.
“Furthermore, to my knowledge, this is the first example of such a reliable biomarker for psychiatric symptoms. The other examples were all for neurological disorders such as Parkinson’s disease, dystonia, and epilepsy,” he added.
He cautioned that this is a single case, but “if reproduced in additional patients, it will bring at least some psychiatric conditions into the domain of brain diseases that can be characterized and diagnosed objectively rather than based on symptoms alone.”
Dr. Litvak pointed out two other critical aspects of the study: the use of exploratory recordings and stimulation to determine the most effective treatment strategy, and the use of a closed-loop device that stimulates only when detecting the amygdala biomarker.
“It is hard to say based on this single case how important these will be in the future. There is no comparison to constant stimulation that might have worked as well because the implanted device used in the study is not suitable for that,” Dr. Litvak said.
It should also be noted that implanting multiple depth electrodes at different brain sites is a “traumatic invasive procedure only reserved to date for severe cases of drug-resistant epilepsy,” he said. “Furthermore, it only allows [researchers] to test a small number of candidate sites, so it relies heavily on prior knowledge.
“Once clinicians know better what to look for, it might be possible to avoid this procedure altogether by using noninvasive methods,” such as functional MRI or EEG, to match the right treatment option to a patient, Dr. Litvak concluded.
The research was funded by the National Institutes of Health, the Brain & Behavior Research Foundation, and the Ray and Dagmar Dolby Family Fund through the department of psychiatry at UCSF. Dr. Scangos has reported no relevant financial relationships. A complete list of author disclosures is available in the original article. Dr. Litvak is participating in a research funding application to search for electrophysiological biomarkers of depression symptoms using invasive recordings.
A version of this article first appeared on Medscape.com.
Worsening motor function tied to post COVID syndrome in Parkinson’s disease
, new research suggests.
Results from a small, international retrospective case study show that about half of participants with Parkinson’s disease who developed post–COVID-19 syndrome experienced a worsening of motor symptoms and that their need for anti-Parkinson’s medication increased.
“In our series of 27 patients with Parkinson’s disease, 85% developed post–COVID-19 symptoms,” said lead investigator Valentina Leta, MD, Parkinson’s Foundation Center of Excellence, Kings College Hospital, London.
The most common long-term effects were worsening of motor function and an increase in the need for daily levodopa. Other adverse effects included fatigue; cognitive disturbances, including brain fog, loss of concentration, and memory deficits; and sleep disturbances, such as insomnia, Dr. Leta said.
The findings were presented at the International Congress of Parkinson’s Disease and Movement Disorders.
Long-term sequelae
Previous studies have documented worsening of motor and nonmotor symptoms among patients with Parkinson’s disease in the acute phase of COVID-19. Results of these studies suggest that mortality may be higher among patients with more advanced Parkinson’s disease, comorbidities, and frailty.
Dr. Leta noted that long-term sequelae with so-called long COVID have not been adequately explored, prompting the current study.
The case series included 27 patients with Parkinson’s disease in the United Kingdom, Italy, Romania, and Mexico who were also affected by COVID-19. The investigators defined post–COVID-19 syndrome as “signs and symptoms that develop during or after an infection consistent with COVID-19, continue for more than 12 weeks, and are not explained by an alternative diagnosis.”
Because some of the symptoms are also associated with Parkinson’s disease, symptoms were attributed to post–COVID-19 only if they occurred after a confirmed severe acute respiratory infection with SARS-CoV-2 or if patients experienced an acute or subacute worsening of a pre-existing symptom that had previously been stable.
Among the participants, 59.3% were men. The mean age at the time of Parkinson’s disease diagnosis was 59.0 ± 12.7 years, and the mean Parkinson’s disease duration was 9.2 ± 7.8 years. The patients were in Hoehn and Yahr stage 2.0 ± 1.0 at the time of their COVID-19 diagnosis.
Charlson Comorbidity Index score at COVID-19 diagnosis was 2.0 ± 1.5, and the levodopa equivalent daily dose (LEDD) was 1053.5 ± 842.4 mg.
Symptom worsening
“Cognitive disturbances” were defined as brain fog, concentration difficulty, or memory problems. “Peripheral neuropathy symptoms” were defined as having feelings of pins and needles or numbness.
By far, the most prevalent sequelae were worsening motor symptoms and increased need for anti-Parkinson’s medications. Each affected about half of the study cohort, the investigators noted.
Dr. Leta added the non-Parkinson’s disease-specific findings are in line with the existing literature on long COVID in the general population. The severity of COVID-19, as indicated by a history of hospitalization, did not seem to correlate with development of post–COVID-19 syndrome in patients with Parkinson’s disease.
In this series, few patients had respiratory, cardiovascular, gastrointestinal, musculoskeletal, or dermatologic symptoms. Interestingly, only four patients reported a loss of taste or smell.
The investigators noted that in addition to viral illness, the stress of prolonged lockdown during the pandemic and reduced access to health care and rehabilitation programs may contribute to the burden of post–COVID-19 syndrome in patients with Parkinson’s disease.
Study limitations cited include the relatively small sample size and the lack of a control group. The researchers noted the need for larger studies to elucidate the natural history of COVID-19 among patients with Parkinson’s disease in order to raise awareness of their needs and to help develop personalized management strategies.
Meaningful addition
Commenting on the findings, Kyle Mitchell, MD, movement disorders neurologist, Duke University, Durham, N.C., said he found the study to be a meaningful addition in light of the fact that data on the challenges that patients with Parkinson’s disease may face after having COVID-19 are limited.
“What I liked about this study was there’s data from multiple countries, what looks like a diverse population of study participants, and really just addressing a question that we get asked a lot in clinic and we see a fair amount, but we don’t really know a lot about: how people with Parkinson’s disease will do during and post COVID-19 infection,” said Dr. Mitchell, who was not involved with the research.
He said the worsening of motor symptoms and the need for increased dopaminergic medication brought some questions to mind.
“Is this increase in medications permanent, or is it temporary until post-COVID resolves? Or is it truly something where they stay on a higher dose?” he asked.
Dr. Mitchell said he does not believe the worsening of symptoms is specific to COVID-19 and that he sees individuals with Parkinson’s disease who experience setbacks “from any number of infections.” These include urinary tract infections and influenza, which are associated with worsening mobility, rigidity, tremor, fatigue, and cognition.
“People with Parkinson’s disease seem to get hit harder by infections in general,” he said.
The study had no outside funding. Dr. Leta and Dr. Mitchell have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, new research suggests.
Results from a small, international retrospective case study show that about half of participants with Parkinson’s disease who developed post–COVID-19 syndrome experienced a worsening of motor symptoms and that their need for anti-Parkinson’s medication increased.
“In our series of 27 patients with Parkinson’s disease, 85% developed post–COVID-19 symptoms,” said lead investigator Valentina Leta, MD, Parkinson’s Foundation Center of Excellence, Kings College Hospital, London.
The most common long-term effects were worsening of motor function and an increase in the need for daily levodopa. Other adverse effects included fatigue; cognitive disturbances, including brain fog, loss of concentration, and memory deficits; and sleep disturbances, such as insomnia, Dr. Leta said.
The findings were presented at the International Congress of Parkinson’s Disease and Movement Disorders.
Long-term sequelae
Previous studies have documented worsening of motor and nonmotor symptoms among patients with Parkinson’s disease in the acute phase of COVID-19. Results of these studies suggest that mortality may be higher among patients with more advanced Parkinson’s disease, comorbidities, and frailty.
Dr. Leta noted that long-term sequelae with so-called long COVID have not been adequately explored, prompting the current study.
The case series included 27 patients with Parkinson’s disease in the United Kingdom, Italy, Romania, and Mexico who were also affected by COVID-19. The investigators defined post–COVID-19 syndrome as “signs and symptoms that develop during or after an infection consistent with COVID-19, continue for more than 12 weeks, and are not explained by an alternative diagnosis.”
Because some of the symptoms are also associated with Parkinson’s disease, symptoms were attributed to post–COVID-19 only if they occurred after a confirmed severe acute respiratory infection with SARS-CoV-2 or if patients experienced an acute or subacute worsening of a pre-existing symptom that had previously been stable.
Among the participants, 59.3% were men. The mean age at the time of Parkinson’s disease diagnosis was 59.0 ± 12.7 years, and the mean Parkinson’s disease duration was 9.2 ± 7.8 years. The patients were in Hoehn and Yahr stage 2.0 ± 1.0 at the time of their COVID-19 diagnosis.
Charlson Comorbidity Index score at COVID-19 diagnosis was 2.0 ± 1.5, and the levodopa equivalent daily dose (LEDD) was 1053.5 ± 842.4 mg.
Symptom worsening
“Cognitive disturbances” were defined as brain fog, concentration difficulty, or memory problems. “Peripheral neuropathy symptoms” were defined as having feelings of pins and needles or numbness.
By far, the most prevalent sequelae were worsening motor symptoms and increased need for anti-Parkinson’s medications. Each affected about half of the study cohort, the investigators noted.
Dr. Leta added the non-Parkinson’s disease-specific findings are in line with the existing literature on long COVID in the general population. The severity of COVID-19, as indicated by a history of hospitalization, did not seem to correlate with development of post–COVID-19 syndrome in patients with Parkinson’s disease.
In this series, few patients had respiratory, cardiovascular, gastrointestinal, musculoskeletal, or dermatologic symptoms. Interestingly, only four patients reported a loss of taste or smell.
The investigators noted that in addition to viral illness, the stress of prolonged lockdown during the pandemic and reduced access to health care and rehabilitation programs may contribute to the burden of post–COVID-19 syndrome in patients with Parkinson’s disease.
Study limitations cited include the relatively small sample size and the lack of a control group. The researchers noted the need for larger studies to elucidate the natural history of COVID-19 among patients with Parkinson’s disease in order to raise awareness of their needs and to help develop personalized management strategies.
Meaningful addition
Commenting on the findings, Kyle Mitchell, MD, movement disorders neurologist, Duke University, Durham, N.C., said he found the study to be a meaningful addition in light of the fact that data on the challenges that patients with Parkinson’s disease may face after having COVID-19 are limited.
“What I liked about this study was there’s data from multiple countries, what looks like a diverse population of study participants, and really just addressing a question that we get asked a lot in clinic and we see a fair amount, but we don’t really know a lot about: how people with Parkinson’s disease will do during and post COVID-19 infection,” said Dr. Mitchell, who was not involved with the research.
He said the worsening of motor symptoms and the need for increased dopaminergic medication brought some questions to mind.
“Is this increase in medications permanent, or is it temporary until post-COVID resolves? Or is it truly something where they stay on a higher dose?” he asked.
Dr. Mitchell said he does not believe the worsening of symptoms is specific to COVID-19 and that he sees individuals with Parkinson’s disease who experience setbacks “from any number of infections.” These include urinary tract infections and influenza, which are associated with worsening mobility, rigidity, tremor, fatigue, and cognition.
“People with Parkinson’s disease seem to get hit harder by infections in general,” he said.
The study had no outside funding. Dr. Leta and Dr. Mitchell have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, new research suggests.
Results from a small, international retrospective case study show that about half of participants with Parkinson’s disease who developed post–COVID-19 syndrome experienced a worsening of motor symptoms and that their need for anti-Parkinson’s medication increased.
“In our series of 27 patients with Parkinson’s disease, 85% developed post–COVID-19 symptoms,” said lead investigator Valentina Leta, MD, Parkinson’s Foundation Center of Excellence, Kings College Hospital, London.
The most common long-term effects were worsening of motor function and an increase in the need for daily levodopa. Other adverse effects included fatigue; cognitive disturbances, including brain fog, loss of concentration, and memory deficits; and sleep disturbances, such as insomnia, Dr. Leta said.
The findings were presented at the International Congress of Parkinson’s Disease and Movement Disorders.
Long-term sequelae
Previous studies have documented worsening of motor and nonmotor symptoms among patients with Parkinson’s disease in the acute phase of COVID-19. Results of these studies suggest that mortality may be higher among patients with more advanced Parkinson’s disease, comorbidities, and frailty.
Dr. Leta noted that long-term sequelae with so-called long COVID have not been adequately explored, prompting the current study.
The case series included 27 patients with Parkinson’s disease in the United Kingdom, Italy, Romania, and Mexico who were also affected by COVID-19. The investigators defined post–COVID-19 syndrome as “signs and symptoms that develop during or after an infection consistent with COVID-19, continue for more than 12 weeks, and are not explained by an alternative diagnosis.”
Because some of the symptoms are also associated with Parkinson’s disease, symptoms were attributed to post–COVID-19 only if they occurred after a confirmed severe acute respiratory infection with SARS-CoV-2 or if patients experienced an acute or subacute worsening of a pre-existing symptom that had previously been stable.
Among the participants, 59.3% were men. The mean age at the time of Parkinson’s disease diagnosis was 59.0 ± 12.7 years, and the mean Parkinson’s disease duration was 9.2 ± 7.8 years. The patients were in Hoehn and Yahr stage 2.0 ± 1.0 at the time of their COVID-19 diagnosis.
Charlson Comorbidity Index score at COVID-19 diagnosis was 2.0 ± 1.5, and the levodopa equivalent daily dose (LEDD) was 1053.5 ± 842.4 mg.
Symptom worsening
“Cognitive disturbances” were defined as brain fog, concentration difficulty, or memory problems. “Peripheral neuropathy symptoms” were defined as having feelings of pins and needles or numbness.
By far, the most prevalent sequelae were worsening motor symptoms and increased need for anti-Parkinson’s medications. Each affected about half of the study cohort, the investigators noted.
Dr. Leta added the non-Parkinson’s disease-specific findings are in line with the existing literature on long COVID in the general population. The severity of COVID-19, as indicated by a history of hospitalization, did not seem to correlate with development of post–COVID-19 syndrome in patients with Parkinson’s disease.
In this series, few patients had respiratory, cardiovascular, gastrointestinal, musculoskeletal, or dermatologic symptoms. Interestingly, only four patients reported a loss of taste or smell.
The investigators noted that in addition to viral illness, the stress of prolonged lockdown during the pandemic and reduced access to health care and rehabilitation programs may contribute to the burden of post–COVID-19 syndrome in patients with Parkinson’s disease.
Study limitations cited include the relatively small sample size and the lack of a control group. The researchers noted the need for larger studies to elucidate the natural history of COVID-19 among patients with Parkinson’s disease in order to raise awareness of their needs and to help develop personalized management strategies.
Meaningful addition
Commenting on the findings, Kyle Mitchell, MD, movement disorders neurologist, Duke University, Durham, N.C., said he found the study to be a meaningful addition in light of the fact that data on the challenges that patients with Parkinson’s disease may face after having COVID-19 are limited.
“What I liked about this study was there’s data from multiple countries, what looks like a diverse population of study participants, and really just addressing a question that we get asked a lot in clinic and we see a fair amount, but we don’t really know a lot about: how people with Parkinson’s disease will do during and post COVID-19 infection,” said Dr. Mitchell, who was not involved with the research.
He said the worsening of motor symptoms and the need for increased dopaminergic medication brought some questions to mind.
“Is this increase in medications permanent, or is it temporary until post-COVID resolves? Or is it truly something where they stay on a higher dose?” he asked.
Dr. Mitchell said he does not believe the worsening of symptoms is specific to COVID-19 and that he sees individuals with Parkinson’s disease who experience setbacks “from any number of infections.” These include urinary tract infections and influenza, which are associated with worsening mobility, rigidity, tremor, fatigue, and cognition.
“People with Parkinson’s disease seem to get hit harder by infections in general,” he said.
The study had no outside funding. Dr. Leta and Dr. Mitchell have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM MDS VIRTUAL CONGRESS 2021
Apple devices identify early Parkinson’s disease
, new research shows. Results from the WATCH-PD study show clear differences in a finger-tapping task in the Parkinson’s disease versus control group. The finger-tapping task also correlated with “traditional measures,” such as the Movement Disorder Society–Unified Parkinson’s Disease Rating Scale (MDS-UPDRS), investigators reported.
“And then the smartphone and smartwatch also showed differences in gait between groups,” said lead investigator Jamie Adams, MD, University of Rochester, New York.
The findings were presented at the International Congress of Parkinson’s Disease and Movement Disorders.
WATCH-PD
The 12-month WATCH-PD study included 132 individuals at 17 Parkinson’s Study Group sites, 82 with Parkinson’s disease and 50 controls.
Participants with Parkinson’s disease were untreated, were no more than 2 years out from diagnosis (mean disease duration, 10.0 ±7.3 months), and were in Hoehn and Yahr stage 1 or 2.
Apple Watches and iPhones were provided to participants, all of whom underwent in-clinic assessments at baseline and at months 1, 3, 6, 9, and 12. The assessments included motor and cognitive tasks using the devices, which contained motion sensors.
The phone also contained an app that could assess verbal, cognitive, and other abilities. Participants wore a set of inertial sensors (APDM Mobility Lab) while performing the MDS-UPDRS Part III motor examination.
In addition, there were biweekly at-home tasks. Questions and tests on the watch assessed symptoms of mood, fatigue, cognition, and falls as well as cognitive performance involving perceptual, verbal, visual spatial, and fine motor abilities. Both the watch and iPhone were used to gauge gait, balance, and tremor.
Ages of the participants were approximately the same in the Parkinson’s disease and control groups (63.3 years vs. 60.2 years, respectively), but male to female ratios differed between the groups. There were more men in the Parkinson’s disease cohort (56% men vs. 44% women) and more women in the control cohort (36% vs. 64%; P =.03).
Between-group differences
Results showed that MDS-UPDRS total scores and on all individual parts of the rating scale were significantly better for the control group (lower scores are better), as shown in the following table.
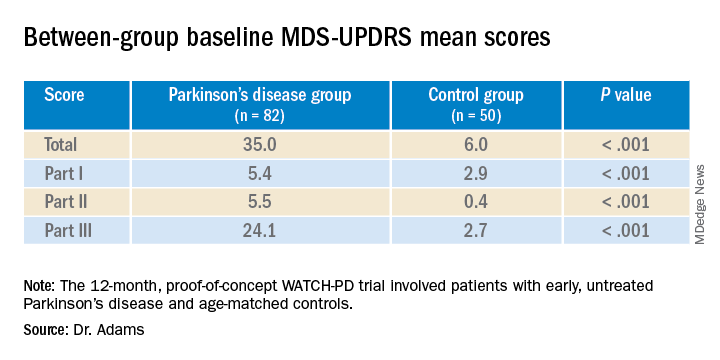
Similarly, the control group performed better than the Parkinson’s disease group on the Montreal Cognitive Assessment (MoCA), with higher scores showing better performance on the 0 to 30 scale (28.1 vs. 27.6, respectively).
Touchscreen assessments on the phone also showed group differences in a finger-tapping task, with more taps by the control group than by the Parkinson’s disease group. The difference was more pronounced when the dominant hand was used.
The median numbers of taps in 20 seconds for the dominant hand were 103.7 for the Parkinson’s disease cohort versus 131.9 for control cohort (P < .005); and for the nondominant hand the numbers of taps were 106.6 versus 122.1 (P < .05), respectively. The control group also scored better on tests of hand fine-motor control (P < .01) and on the mobile digit symbols modalities test (P < .05)
Measures of gait in a 1-minute walk test also showed group differences.
“The five gait measures that differed most were cadence, which is steps per minute, double support, arm swing amplitude, arm swing variation, and turn duration,” Dr. Adams said.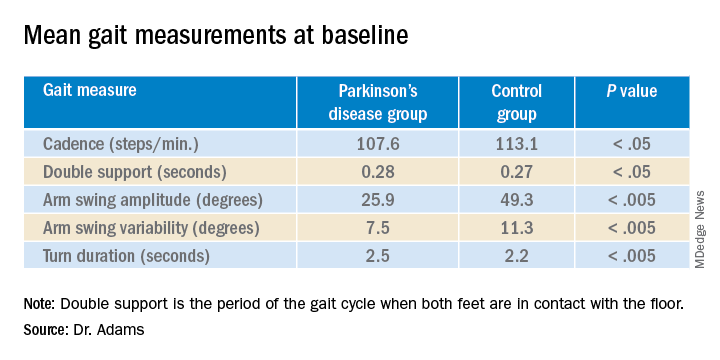
‘Tremendous interest’
Commenting on the findings, Ludy Shih, MD, MMSc, of Boston University, noted that in the future, devices such as the ones used in this study may help clinicians remotely monitor their patients’ Parkinson’s disease conditions and response to therapy.
That would “eliminate some of the transportation barrier for people with Parkinson’s disease,” said Dr. Shih, who was not involved with the research.
The devices can give objective measurements, reducing inter-rater variability in assessment of movements, she noted.
“I think there’s tremendous interest in using digital measures to pick up on subtle disease phenotypes earlier than a clinical diagnosis can be made,” Dr. Shih said.
She also referred to literature “going back a few decades” showing that finger tapping can be used as a pharmacodynamic measure of how well a patient’s dopaminergic medications are working, so the devices may be a way to remotely assess treatment efficacy and decide when it is time to make adjustments.
Dr. Shih said she thinks regulatory agencies are now open “to consider these as part of the totality of evidence that a therapeutic [device] might be working.”
Whether these would need to be professional grade and approved as medical devices or if patients could just buy smartwatches and smartphones to generate useful data is still a question, she said. Already, there are several Parkinson’s apps that the public can download to track symptoms, improve voice, provide exercises, find support groups or research studies, and more.
Dr. Shih predicted that the biweekly at-home tasks, as in the current protocol, could be a burden to some people. If only a segment of the population were willing to comply, it could call into question how generalizable the results were, she added.
“There’s even a prior publication showing that compliance rate really dropped like a rock,” she noted. However, for those people willing to perform the tasks on a regular schedule, the results could be valuable, Dr. Shih said.
Dr. Adams concurred, saying that she had received feedback from some of her study participants that the biweekly tasks were a bit much.
The study was supported by Biogen and Takeda Pharmaceuticals. Dr. Adams receives research support from Biogen. Dr. Shih has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, new research shows. Results from the WATCH-PD study show clear differences in a finger-tapping task in the Parkinson’s disease versus control group. The finger-tapping task also correlated with “traditional measures,” such as the Movement Disorder Society–Unified Parkinson’s Disease Rating Scale (MDS-UPDRS), investigators reported.
“And then the smartphone and smartwatch also showed differences in gait between groups,” said lead investigator Jamie Adams, MD, University of Rochester, New York.
The findings were presented at the International Congress of Parkinson’s Disease and Movement Disorders.
WATCH-PD
The 12-month WATCH-PD study included 132 individuals at 17 Parkinson’s Study Group sites, 82 with Parkinson’s disease and 50 controls.
Participants with Parkinson’s disease were untreated, were no more than 2 years out from diagnosis (mean disease duration, 10.0 ±7.3 months), and were in Hoehn and Yahr stage 1 or 2.
Apple Watches and iPhones were provided to participants, all of whom underwent in-clinic assessments at baseline and at months 1, 3, 6, 9, and 12. The assessments included motor and cognitive tasks using the devices, which contained motion sensors.
The phone also contained an app that could assess verbal, cognitive, and other abilities. Participants wore a set of inertial sensors (APDM Mobility Lab) while performing the MDS-UPDRS Part III motor examination.
In addition, there were biweekly at-home tasks. Questions and tests on the watch assessed symptoms of mood, fatigue, cognition, and falls as well as cognitive performance involving perceptual, verbal, visual spatial, and fine motor abilities. Both the watch and iPhone were used to gauge gait, balance, and tremor.
Ages of the participants were approximately the same in the Parkinson’s disease and control groups (63.3 years vs. 60.2 years, respectively), but male to female ratios differed between the groups. There were more men in the Parkinson’s disease cohort (56% men vs. 44% women) and more women in the control cohort (36% vs. 64%; P =.03).
Between-group differences
Results showed that MDS-UPDRS total scores and on all individual parts of the rating scale were significantly better for the control group (lower scores are better), as shown in the following table.

Similarly, the control group performed better than the Parkinson’s disease group on the Montreal Cognitive Assessment (MoCA), with higher scores showing better performance on the 0 to 30 scale (28.1 vs. 27.6, respectively).
Touchscreen assessments on the phone also showed group differences in a finger-tapping task, with more taps by the control group than by the Parkinson’s disease group. The difference was more pronounced when the dominant hand was used.
The median numbers of taps in 20 seconds for the dominant hand were 103.7 for the Parkinson’s disease cohort versus 131.9 for control cohort (P < .005); and for the nondominant hand the numbers of taps were 106.6 versus 122.1 (P < .05), respectively. The control group also scored better on tests of hand fine-motor control (P < .01) and on the mobile digit symbols modalities test (P < .05)
Measures of gait in a 1-minute walk test also showed group differences.
“The five gait measures that differed most were cadence, which is steps per minute, double support, arm swing amplitude, arm swing variation, and turn duration,” Dr. Adams said.
‘Tremendous interest’
Commenting on the findings, Ludy Shih, MD, MMSc, of Boston University, noted that in the future, devices such as the ones used in this study may help clinicians remotely monitor their patients’ Parkinson’s disease conditions and response to therapy.
That would “eliminate some of the transportation barrier for people with Parkinson’s disease,” said Dr. Shih, who was not involved with the research.
The devices can give objective measurements, reducing inter-rater variability in assessment of movements, she noted.
“I think there’s tremendous interest in using digital measures to pick up on subtle disease phenotypes earlier than a clinical diagnosis can be made,” Dr. Shih said.
She also referred to literature “going back a few decades” showing that finger tapping can be used as a pharmacodynamic measure of how well a patient’s dopaminergic medications are working, so the devices may be a way to remotely assess treatment efficacy and decide when it is time to make adjustments.
Dr. Shih said she thinks regulatory agencies are now open “to consider these as part of the totality of evidence that a therapeutic [device] might be working.”
Whether these would need to be professional grade and approved as medical devices or if patients could just buy smartwatches and smartphones to generate useful data is still a question, she said. Already, there are several Parkinson’s apps that the public can download to track symptoms, improve voice, provide exercises, find support groups or research studies, and more.
Dr. Shih predicted that the biweekly at-home tasks, as in the current protocol, could be a burden to some people. If only a segment of the population were willing to comply, it could call into question how generalizable the results were, she added.
“There’s even a prior publication showing that compliance rate really dropped like a rock,” she noted. However, for those people willing to perform the tasks on a regular schedule, the results could be valuable, Dr. Shih said.
Dr. Adams concurred, saying that she had received feedback from some of her study participants that the biweekly tasks were a bit much.
The study was supported by Biogen and Takeda Pharmaceuticals. Dr. Adams receives research support from Biogen. Dr. Shih has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, new research shows. Results from the WATCH-PD study show clear differences in a finger-tapping task in the Parkinson’s disease versus control group. The finger-tapping task also correlated with “traditional measures,” such as the Movement Disorder Society–Unified Parkinson’s Disease Rating Scale (MDS-UPDRS), investigators reported.
“And then the smartphone and smartwatch also showed differences in gait between groups,” said lead investigator Jamie Adams, MD, University of Rochester, New York.
The findings were presented at the International Congress of Parkinson’s Disease and Movement Disorders.
WATCH-PD
The 12-month WATCH-PD study included 132 individuals at 17 Parkinson’s Study Group sites, 82 with Parkinson’s disease and 50 controls.
Participants with Parkinson’s disease were untreated, were no more than 2 years out from diagnosis (mean disease duration, 10.0 ±7.3 months), and were in Hoehn and Yahr stage 1 or 2.
Apple Watches and iPhones were provided to participants, all of whom underwent in-clinic assessments at baseline and at months 1, 3, 6, 9, and 12. The assessments included motor and cognitive tasks using the devices, which contained motion sensors.
The phone also contained an app that could assess verbal, cognitive, and other abilities. Participants wore a set of inertial sensors (APDM Mobility Lab) while performing the MDS-UPDRS Part III motor examination.
In addition, there were biweekly at-home tasks. Questions and tests on the watch assessed symptoms of mood, fatigue, cognition, and falls as well as cognitive performance involving perceptual, verbal, visual spatial, and fine motor abilities. Both the watch and iPhone were used to gauge gait, balance, and tremor.
Ages of the participants were approximately the same in the Parkinson’s disease and control groups (63.3 years vs. 60.2 years, respectively), but male to female ratios differed between the groups. There were more men in the Parkinson’s disease cohort (56% men vs. 44% women) and more women in the control cohort (36% vs. 64%; P =.03).
Between-group differences
Results showed that MDS-UPDRS total scores and on all individual parts of the rating scale were significantly better for the control group (lower scores are better), as shown in the following table.

Similarly, the control group performed better than the Parkinson’s disease group on the Montreal Cognitive Assessment (MoCA), with higher scores showing better performance on the 0 to 30 scale (28.1 vs. 27.6, respectively).
Touchscreen assessments on the phone also showed group differences in a finger-tapping task, with more taps by the control group than by the Parkinson’s disease group. The difference was more pronounced when the dominant hand was used.
The median numbers of taps in 20 seconds for the dominant hand were 103.7 for the Parkinson’s disease cohort versus 131.9 for control cohort (P < .005); and for the nondominant hand the numbers of taps were 106.6 versus 122.1 (P < .05), respectively. The control group also scored better on tests of hand fine-motor control (P < .01) and on the mobile digit symbols modalities test (P < .05)
Measures of gait in a 1-minute walk test also showed group differences.
“The five gait measures that differed most were cadence, which is steps per minute, double support, arm swing amplitude, arm swing variation, and turn duration,” Dr. Adams said.
‘Tremendous interest’
Commenting on the findings, Ludy Shih, MD, MMSc, of Boston University, noted that in the future, devices such as the ones used in this study may help clinicians remotely monitor their patients’ Parkinson’s disease conditions and response to therapy.
That would “eliminate some of the transportation barrier for people with Parkinson’s disease,” said Dr. Shih, who was not involved with the research.
The devices can give objective measurements, reducing inter-rater variability in assessment of movements, she noted.
“I think there’s tremendous interest in using digital measures to pick up on subtle disease phenotypes earlier than a clinical diagnosis can be made,” Dr. Shih said.
She also referred to literature “going back a few decades” showing that finger tapping can be used as a pharmacodynamic measure of how well a patient’s dopaminergic medications are working, so the devices may be a way to remotely assess treatment efficacy and decide when it is time to make adjustments.
Dr. Shih said she thinks regulatory agencies are now open “to consider these as part of the totality of evidence that a therapeutic [device] might be working.”
Whether these would need to be professional grade and approved as medical devices or if patients could just buy smartwatches and smartphones to generate useful data is still a question, she said. Already, there are several Parkinson’s apps that the public can download to track symptoms, improve voice, provide exercises, find support groups or research studies, and more.
Dr. Shih predicted that the biweekly at-home tasks, as in the current protocol, could be a burden to some people. If only a segment of the population were willing to comply, it could call into question how generalizable the results were, she added.
“There’s even a prior publication showing that compliance rate really dropped like a rock,” she noted. However, for those people willing to perform the tasks on a regular schedule, the results could be valuable, Dr. Shih said.
Dr. Adams concurred, saying that she had received feedback from some of her study participants that the biweekly tasks were a bit much.
The study was supported by Biogen and Takeda Pharmaceuticals. Dr. Adams receives research support from Biogen. Dr. Shih has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM MDS VIRTUAL CONGRESS 2021
Study identifies pandemic-related stressor in Parkinson’s disease
a team of researchers in the Netherlands reported, but they also identified meaningful targets for intervention.
Lisanne Dommershuijsen, MSc, a PhD candidate and researcher in epidemiology at the Erasmus University Medical Center in Rotterdam, the Netherlands, reported on a cross-sectional study of 833 participants with Parkinson’s disease in the PRIME-NL study at the International Congress of Parkinson’s Disease and Movement Disorders. The average age of participants was 70.2 and 38% were women.
“We studied targeted hypothetical interventions on COVID-19 stressors in people with Parkinson’s disease,” Ms. Dommershuijsen said. “This disruption in normal life caused considerable psychological stress in community-dwelling individuals. People with Parkinson’s disease might be especially vulnerable to this stress.
“For instance, because reduced levels of physical activity have worsened symptoms or because people with Parkinson’s often have difficulty with flexible [adaptations] to drastic and rapid changes in daily routines, such as those introduced by the COVID-19 pandemic, previous studies found that COVID-19 worsened depression and anxiety symptoms and reduced quality of life (QOL) in people with Parkinson’s disease,” Ms. Dommershuijsen said.
Hence, the goal of the study was to identify the most vulnerable subgroups in the Parkinson’s population and to suggest potential interventions to ameliorate these impacts, she said.
The study focused on eight different stressors that emerged in the pandemic: access to care, medicine and nursing services; loss of social contact; canceled social events; tension or conflict in the home; inability to perform physical activity or relax; and COVID-19 symptoms. The outcomes of interest were depression, as measured with the Beck Depression Inventory (BDI); anxiety, as measured with the Spielberger State-Trait Anxiety Inventory (STAI); and QOL, with the Parkinson’s Disease Quality of Life Questionnaire. The aggregate resulted in a scale of 0-40, with the mean stressor score in the study being 9.6, Ms. Dommershuijsen said.
The BDI and STAI scores for social stressors – loss of social contacts, social events canceled and tension or conflict at home – exceeded those for the so-called care stressors – problems accessing care, medication or nursing – she said, although all eight stressors yielded higher BDI and STAI scores across the board.
Vulnerable subgroups
“When we looked at vulnerable subgroups of people with Parkinson’s disease, we found more pronounced associations between the COVID-19 stress and mental health in women, in highly educated participants, and in participants with advanced Parkinson’s disease,” Ms. Dommershuijsen said. The impact on women and people with advanced disease is explainable, Ms. Dommershuijsen added in an interview; the former because depressive symptoms are more common in women, and the latter because loss of access to care impacts mental wellness.
“The finding that social stressors were more related to anxiety in highly educated people was surprising to us, given that depression in general is more common in people with a lower education,” she said in an interview. “One previous study of the general population suggested this might be related to expectations about available resources, but this findings and the possible explanation warrants further investigation.”
When the study stratified for coping strategies, the COVID-19 stressors had a smaller effect on depressive and anxiety symptoms in Parkinson’s disease patients prone to confrontive coping and planful problem solving, she said. “Whereas, we observed a larger effect of these stressors in people who are prone to using distancing or seeking social support as coping mechanisms,” Ms. Dommershuijsen said.
The researchers also created a model of a hypothetical 50% reduction in COVID-19 stressors among all study participants, but the effect wasn’t clinically relevant, Ms. Dommershuijsen said. However, in people with advanced Parkinson’s disease – that is, with an Movement Disorder Society–Unified Parkinson Disease Rating Scale score above median – the effect was clinically relevant in all outcomes.
The potential interventions the study identified were telemedicine via virtual consultations to alleviate care stressors, and virtual support groups and online classes to address social stressors. “However, a more personalized approach is needed to target tension or conflict at home, which was the most important social stressor influencing depression and anxiety symptoms in our study,” she said. “Social work can play an important role here.”
Asked to comment on the study, Roy Alcalay, MD, professor of neurology at Columbia University Irving Medical Center in New York, said in an interview that the findings align with his research on the impact of COVID-19 and related restrictions on people with Parkinson’s disease.
“The pandemic has affected people in different ways,” he said. “Initially very acutely, people just didn’t have access to doctors. There was also the acute question in movement disorders, but also in other diseases where the people with Parkinson’s disease are going to have the worse outcome when they have COVID-19.” Dr. Alcalay authored two recent papers on the impact of COVID-19 in people with Parkinson’s disease.
“Then we see that, in addition to that question, there’s the question of even if they don’t have COVID-19, just the social distancing and the lack of access to health care, and specifically to physical and occupational therapy and other services, can be quite damaging,” he said.
What’s commendable about the study, he said, was that it just doesn’t highlight the problem. “They’re also highlighting potential solutions, that planful problem solving and coping strategies can be helpful to people.”
Neither Ms. Dommershuijsen nor Dr. Alcalay have any relevant relationships to disclose.
a team of researchers in the Netherlands reported, but they also identified meaningful targets for intervention.
Lisanne Dommershuijsen, MSc, a PhD candidate and researcher in epidemiology at the Erasmus University Medical Center in Rotterdam, the Netherlands, reported on a cross-sectional study of 833 participants with Parkinson’s disease in the PRIME-NL study at the International Congress of Parkinson’s Disease and Movement Disorders. The average age of participants was 70.2 and 38% were women.
“We studied targeted hypothetical interventions on COVID-19 stressors in people with Parkinson’s disease,” Ms. Dommershuijsen said. “This disruption in normal life caused considerable psychological stress in community-dwelling individuals. People with Parkinson’s disease might be especially vulnerable to this stress.
“For instance, because reduced levels of physical activity have worsened symptoms or because people with Parkinson’s often have difficulty with flexible [adaptations] to drastic and rapid changes in daily routines, such as those introduced by the COVID-19 pandemic, previous studies found that COVID-19 worsened depression and anxiety symptoms and reduced quality of life (QOL) in people with Parkinson’s disease,” Ms. Dommershuijsen said.
Hence, the goal of the study was to identify the most vulnerable subgroups in the Parkinson’s population and to suggest potential interventions to ameliorate these impacts, she said.
The study focused on eight different stressors that emerged in the pandemic: access to care, medicine and nursing services; loss of social contact; canceled social events; tension or conflict in the home; inability to perform physical activity or relax; and COVID-19 symptoms. The outcomes of interest were depression, as measured with the Beck Depression Inventory (BDI); anxiety, as measured with the Spielberger State-Trait Anxiety Inventory (STAI); and QOL, with the Parkinson’s Disease Quality of Life Questionnaire. The aggregate resulted in a scale of 0-40, with the mean stressor score in the study being 9.6, Ms. Dommershuijsen said.
The BDI and STAI scores for social stressors – loss of social contacts, social events canceled and tension or conflict at home – exceeded those for the so-called care stressors – problems accessing care, medication or nursing – she said, although all eight stressors yielded higher BDI and STAI scores across the board.
Vulnerable subgroups
“When we looked at vulnerable subgroups of people with Parkinson’s disease, we found more pronounced associations between the COVID-19 stress and mental health in women, in highly educated participants, and in participants with advanced Parkinson’s disease,” Ms. Dommershuijsen said. The impact on women and people with advanced disease is explainable, Ms. Dommershuijsen added in an interview; the former because depressive symptoms are more common in women, and the latter because loss of access to care impacts mental wellness.
“The finding that social stressors were more related to anxiety in highly educated people was surprising to us, given that depression in general is more common in people with a lower education,” she said in an interview. “One previous study of the general population suggested this might be related to expectations about available resources, but this findings and the possible explanation warrants further investigation.”
When the study stratified for coping strategies, the COVID-19 stressors had a smaller effect on depressive and anxiety symptoms in Parkinson’s disease patients prone to confrontive coping and planful problem solving, she said. “Whereas, we observed a larger effect of these stressors in people who are prone to using distancing or seeking social support as coping mechanisms,” Ms. Dommershuijsen said.
The researchers also created a model of a hypothetical 50% reduction in COVID-19 stressors among all study participants, but the effect wasn’t clinically relevant, Ms. Dommershuijsen said. However, in people with advanced Parkinson’s disease – that is, with an Movement Disorder Society–Unified Parkinson Disease Rating Scale score above median – the effect was clinically relevant in all outcomes.
The potential interventions the study identified were telemedicine via virtual consultations to alleviate care stressors, and virtual support groups and online classes to address social stressors. “However, a more personalized approach is needed to target tension or conflict at home, which was the most important social stressor influencing depression and anxiety symptoms in our study,” she said. “Social work can play an important role here.”
Asked to comment on the study, Roy Alcalay, MD, professor of neurology at Columbia University Irving Medical Center in New York, said in an interview that the findings align with his research on the impact of COVID-19 and related restrictions on people with Parkinson’s disease.
“The pandemic has affected people in different ways,” he said. “Initially very acutely, people just didn’t have access to doctors. There was also the acute question in movement disorders, but also in other diseases where the people with Parkinson’s disease are going to have the worse outcome when they have COVID-19.” Dr. Alcalay authored two recent papers on the impact of COVID-19 in people with Parkinson’s disease.
“Then we see that, in addition to that question, there’s the question of even if they don’t have COVID-19, just the social distancing and the lack of access to health care, and specifically to physical and occupational therapy and other services, can be quite damaging,” he said.
What’s commendable about the study, he said, was that it just doesn’t highlight the problem. “They’re also highlighting potential solutions, that planful problem solving and coping strategies can be helpful to people.”
Neither Ms. Dommershuijsen nor Dr. Alcalay have any relevant relationships to disclose.
a team of researchers in the Netherlands reported, but they also identified meaningful targets for intervention.
Lisanne Dommershuijsen, MSc, a PhD candidate and researcher in epidemiology at the Erasmus University Medical Center in Rotterdam, the Netherlands, reported on a cross-sectional study of 833 participants with Parkinson’s disease in the PRIME-NL study at the International Congress of Parkinson’s Disease and Movement Disorders. The average age of participants was 70.2 and 38% were women.
“We studied targeted hypothetical interventions on COVID-19 stressors in people with Parkinson’s disease,” Ms. Dommershuijsen said. “This disruption in normal life caused considerable psychological stress in community-dwelling individuals. People with Parkinson’s disease might be especially vulnerable to this stress.
“For instance, because reduced levels of physical activity have worsened symptoms or because people with Parkinson’s often have difficulty with flexible [adaptations] to drastic and rapid changes in daily routines, such as those introduced by the COVID-19 pandemic, previous studies found that COVID-19 worsened depression and anxiety symptoms and reduced quality of life (QOL) in people with Parkinson’s disease,” Ms. Dommershuijsen said.
Hence, the goal of the study was to identify the most vulnerable subgroups in the Parkinson’s population and to suggest potential interventions to ameliorate these impacts, she said.
The study focused on eight different stressors that emerged in the pandemic: access to care, medicine and nursing services; loss of social contact; canceled social events; tension or conflict in the home; inability to perform physical activity or relax; and COVID-19 symptoms. The outcomes of interest were depression, as measured with the Beck Depression Inventory (BDI); anxiety, as measured with the Spielberger State-Trait Anxiety Inventory (STAI); and QOL, with the Parkinson’s Disease Quality of Life Questionnaire. The aggregate resulted in a scale of 0-40, with the mean stressor score in the study being 9.6, Ms. Dommershuijsen said.
The BDI and STAI scores for social stressors – loss of social contacts, social events canceled and tension or conflict at home – exceeded those for the so-called care stressors – problems accessing care, medication or nursing – she said, although all eight stressors yielded higher BDI and STAI scores across the board.
Vulnerable subgroups
“When we looked at vulnerable subgroups of people with Parkinson’s disease, we found more pronounced associations between the COVID-19 stress and mental health in women, in highly educated participants, and in participants with advanced Parkinson’s disease,” Ms. Dommershuijsen said. The impact on women and people with advanced disease is explainable, Ms. Dommershuijsen added in an interview; the former because depressive symptoms are more common in women, and the latter because loss of access to care impacts mental wellness.
“The finding that social stressors were more related to anxiety in highly educated people was surprising to us, given that depression in general is more common in people with a lower education,” she said in an interview. “One previous study of the general population suggested this might be related to expectations about available resources, but this findings and the possible explanation warrants further investigation.”
When the study stratified for coping strategies, the COVID-19 stressors had a smaller effect on depressive and anxiety symptoms in Parkinson’s disease patients prone to confrontive coping and planful problem solving, she said. “Whereas, we observed a larger effect of these stressors in people who are prone to using distancing or seeking social support as coping mechanisms,” Ms. Dommershuijsen said.
The researchers also created a model of a hypothetical 50% reduction in COVID-19 stressors among all study participants, but the effect wasn’t clinically relevant, Ms. Dommershuijsen said. However, in people with advanced Parkinson’s disease – that is, with an Movement Disorder Society–Unified Parkinson Disease Rating Scale score above median – the effect was clinically relevant in all outcomes.
The potential interventions the study identified were telemedicine via virtual consultations to alleviate care stressors, and virtual support groups and online classes to address social stressors. “However, a more personalized approach is needed to target tension or conflict at home, which was the most important social stressor influencing depression and anxiety symptoms in our study,” she said. “Social work can play an important role here.”
Asked to comment on the study, Roy Alcalay, MD, professor of neurology at Columbia University Irving Medical Center in New York, said in an interview that the findings align with his research on the impact of COVID-19 and related restrictions on people with Parkinson’s disease.
“The pandemic has affected people in different ways,” he said. “Initially very acutely, people just didn’t have access to doctors. There was also the acute question in movement disorders, but also in other diseases where the people with Parkinson’s disease are going to have the worse outcome when they have COVID-19.” Dr. Alcalay authored two recent papers on the impact of COVID-19 in people with Parkinson’s disease.
“Then we see that, in addition to that question, there’s the question of even if they don’t have COVID-19, just the social distancing and the lack of access to health care, and specifically to physical and occupational therapy and other services, can be quite damaging,” he said.
What’s commendable about the study, he said, was that it just doesn’t highlight the problem. “They’re also highlighting potential solutions, that planful problem solving and coping strategies can be helpful to people.”
Neither Ms. Dommershuijsen nor Dr. Alcalay have any relevant relationships to disclose.
FROM MDS VIRTUAL CONGRESS 2021
Efforts target underrepresented populations in Parkinson’s disease genetic studies
, attendees at the International Congress of Parkinson’s Disease and Movement Disorders were told.
“Through the years, as we’ve increased the number of individuals that we’ve included in our genetic studies, the number of risk factors that we’ve been able to identify has increased exponentially,” said Ignacio F. Mata, PhD, a neurogeneticist and principal investigator with the Genomic Medicine Institute of the Cleveland Clinic Lerner Research Institute. “This is all due to collaborations.”
Dr. Mata reviewed no fewer than seven initiatives that are gathering genetic data from people with Parkinson’s disease in Central and South America, India, China, Africa, Oceania, the Middle East, and Central Asia, along with efforts to target diverse populations in London and African Americans in the United States.
“One of the problems that we’ve had in the past is that most of the studies have been done just with individuals that are of European ancestry, so there’s a big gap of other populations that we haven’t been able to study,” Dr. Mata said. “And this is true for all of the current studies that are ongoing here in the United States.” That includes the Parkinson’s Progression Markers Initiative, he said, in which fewer than 6% of participants are non-European. Dr. Mata is also the lead in the Global Parkinson’s Genetics Program (GP2) for underrepresented populations.
Lack of diversity in genetic studies isn’t an issue in Parkinson’s studies alone, Dr. Mata said. “This is a generalized problem across all genetic studies,” he said, citing a 2016 analysis that found the proportion of participants in genome-wide studies was 96% European descent in 2009, shifting to 80% by 2016. “There’s still a big gap because most of the non-European populations came mostly from Asia,” Dr. Mata said, with Latinos and people of African descent representing less than 1% of the study populations.
In an interview, Dr. Mata noted there are a multitude of reasons for enrolling more diverse populations. “We’re going to be able to use genetics to create new treatments and do risk prediction – the so-called precision or personalized medicine,” he said. “We’re leaving a big chunk of the population behind if we don’t include those individuals.”
Scientific basis for diversity
There are a multitude of scientific reasons for doing so, too, said Dr. Mata. “In the whole genome we try to find gene variants that modify the risk for certain disease,” he said. “These regions can be quite large, so increasing the number of individuals that come from different genetic backgrounds can actually help us reduce the number of regions that need to be studied to find the causal variants.”
Andrew Singleton, PhD, director of the Center for Alzheimer’s and Related Dementias at the National Institute of Aging in Bethesda, Md., concurred that enrolling more diverse populations can speed up research for targeting genetic variants.
“We can use the differences in genetics to narrow down our search for variants, reduce the places where we’re looking for risk variants, and reduce the number of genes we’re looking at,” he said in an interview.
Roy Alcalay, MD, professor of neurology at Columbia University Irving Medical Center in New York, offered two more scientific reasons for more diverse study populations “in addition to being more ethically appropriate,” he said. “One is, you may identify new genes that you wouldn’t have identified otherwise; and also in the genes that already exist, you may recognize that some of the pathogenic variants may be more prevalent in populations that were unknown.”
One of the challenges in casting a wider net is that much of the research funding has been concentrated in the United States and Europe, Dr. Mata said. And even in the United States, with large, diverse populations, minority groups are underrepresented in these studies, he said, but a potential solution is emerging. “This is something that we’re learning now with COVID,” he said. “We can do a lot remotely. This should help bring some of those barriers down.”
Cultural barriers are also foreboding. “Individuals may not feel comfortable participating in research,” he said in the interview. “I see this especially in the Hispanic community; many don’t understand what they can do with genetic material, or they’re afraid it will be shared with police, and if they’re here in nonofficial immigration status, they’re afraid they could be deported. There are a lot of misconceptions about genetic research.”
An initiative of the Parkinson’s Foundation PD GENEration Study is to provide free genetic tests and give the patient a report on genetic counseling “to empower patients,” Dr. Mata said.
Solutions for targeting underrepresented groups are emerging, Dr. Singleton said. “Actually there’s a really elegant solution, which is that in the populations that we go into and work with, we make sure the ownership of those cohorts, the ownership of the science and the analysis belongs to those populations,” he said.
“Part of that is creating infrastructure on site,” Dr. Singleton added. “Another part is providing training and outreach so we can help to train a whole new generation of scientists and researchers who can work in those populations embedded within those populations. They’re really the champion of moving that research forward.”
Dr. Alcalay credited Dr. Mata for his work with cohorts in Central and South America and in reaching out to other countries to recruit more diverse populations for genetic Parkinson’s studies. “And it’s not just because it’s the politically correct thing to do, about inclusivity and diversity,” Dr. Alcalay said. “It’s because it’s really meaningful. In addition to being ethically more appropriate, it will advance the entire field.
“I also really think it’s a no-brainer,” Dr. Alcalay said. “It’s something that needs to happen.”
Dr. Mata receives grant funding from the National Institutes of Health. Dr. Singleton and Dr. Alcalay have no relevant disclosures.
, attendees at the International Congress of Parkinson’s Disease and Movement Disorders were told.
“Through the years, as we’ve increased the number of individuals that we’ve included in our genetic studies, the number of risk factors that we’ve been able to identify has increased exponentially,” said Ignacio F. Mata, PhD, a neurogeneticist and principal investigator with the Genomic Medicine Institute of the Cleveland Clinic Lerner Research Institute. “This is all due to collaborations.”
Dr. Mata reviewed no fewer than seven initiatives that are gathering genetic data from people with Parkinson’s disease in Central and South America, India, China, Africa, Oceania, the Middle East, and Central Asia, along with efforts to target diverse populations in London and African Americans in the United States.
“One of the problems that we’ve had in the past is that most of the studies have been done just with individuals that are of European ancestry, so there’s a big gap of other populations that we haven’t been able to study,” Dr. Mata said. “And this is true for all of the current studies that are ongoing here in the United States.” That includes the Parkinson’s Progression Markers Initiative, he said, in which fewer than 6% of participants are non-European. Dr. Mata is also the lead in the Global Parkinson’s Genetics Program (GP2) for underrepresented populations.
Lack of diversity in genetic studies isn’t an issue in Parkinson’s studies alone, Dr. Mata said. “This is a generalized problem across all genetic studies,” he said, citing a 2016 analysis that found the proportion of participants in genome-wide studies was 96% European descent in 2009, shifting to 80% by 2016. “There’s still a big gap because most of the non-European populations came mostly from Asia,” Dr. Mata said, with Latinos and people of African descent representing less than 1% of the study populations.
In an interview, Dr. Mata noted there are a multitude of reasons for enrolling more diverse populations. “We’re going to be able to use genetics to create new treatments and do risk prediction – the so-called precision or personalized medicine,” he said. “We’re leaving a big chunk of the population behind if we don’t include those individuals.”
Scientific basis for diversity
There are a multitude of scientific reasons for doing so, too, said Dr. Mata. “In the whole genome we try to find gene variants that modify the risk for certain disease,” he said. “These regions can be quite large, so increasing the number of individuals that come from different genetic backgrounds can actually help us reduce the number of regions that need to be studied to find the causal variants.”
Andrew Singleton, PhD, director of the Center for Alzheimer’s and Related Dementias at the National Institute of Aging in Bethesda, Md., concurred that enrolling more diverse populations can speed up research for targeting genetic variants.
“We can use the differences in genetics to narrow down our search for variants, reduce the places where we’re looking for risk variants, and reduce the number of genes we’re looking at,” he said in an interview.
Roy Alcalay, MD, professor of neurology at Columbia University Irving Medical Center in New York, offered two more scientific reasons for more diverse study populations “in addition to being more ethically appropriate,” he said. “One is, you may identify new genes that you wouldn’t have identified otherwise; and also in the genes that already exist, you may recognize that some of the pathogenic variants may be more prevalent in populations that were unknown.”
One of the challenges in casting a wider net is that much of the research funding has been concentrated in the United States and Europe, Dr. Mata said. And even in the United States, with large, diverse populations, minority groups are underrepresented in these studies, he said, but a potential solution is emerging. “This is something that we’re learning now with COVID,” he said. “We can do a lot remotely. This should help bring some of those barriers down.”
Cultural barriers are also foreboding. “Individuals may not feel comfortable participating in research,” he said in the interview. “I see this especially in the Hispanic community; many don’t understand what they can do with genetic material, or they’re afraid it will be shared with police, and if they’re here in nonofficial immigration status, they’re afraid they could be deported. There are a lot of misconceptions about genetic research.”
An initiative of the Parkinson’s Foundation PD GENEration Study is to provide free genetic tests and give the patient a report on genetic counseling “to empower patients,” Dr. Mata said.
Solutions for targeting underrepresented groups are emerging, Dr. Singleton said. “Actually there’s a really elegant solution, which is that in the populations that we go into and work with, we make sure the ownership of those cohorts, the ownership of the science and the analysis belongs to those populations,” he said.
“Part of that is creating infrastructure on site,” Dr. Singleton added. “Another part is providing training and outreach so we can help to train a whole new generation of scientists and researchers who can work in those populations embedded within those populations. They’re really the champion of moving that research forward.”
Dr. Alcalay credited Dr. Mata for his work with cohorts in Central and South America and in reaching out to other countries to recruit more diverse populations for genetic Parkinson’s studies. “And it’s not just because it’s the politically correct thing to do, about inclusivity and diversity,” Dr. Alcalay said. “It’s because it’s really meaningful. In addition to being ethically more appropriate, it will advance the entire field.
“I also really think it’s a no-brainer,” Dr. Alcalay said. “It’s something that needs to happen.”
Dr. Mata receives grant funding from the National Institutes of Health. Dr. Singleton and Dr. Alcalay have no relevant disclosures.
, attendees at the International Congress of Parkinson’s Disease and Movement Disorders were told.
“Through the years, as we’ve increased the number of individuals that we’ve included in our genetic studies, the number of risk factors that we’ve been able to identify has increased exponentially,” said Ignacio F. Mata, PhD, a neurogeneticist and principal investigator with the Genomic Medicine Institute of the Cleveland Clinic Lerner Research Institute. “This is all due to collaborations.”
Dr. Mata reviewed no fewer than seven initiatives that are gathering genetic data from people with Parkinson’s disease in Central and South America, India, China, Africa, Oceania, the Middle East, and Central Asia, along with efforts to target diverse populations in London and African Americans in the United States.
“One of the problems that we’ve had in the past is that most of the studies have been done just with individuals that are of European ancestry, so there’s a big gap of other populations that we haven’t been able to study,” Dr. Mata said. “And this is true for all of the current studies that are ongoing here in the United States.” That includes the Parkinson’s Progression Markers Initiative, he said, in which fewer than 6% of participants are non-European. Dr. Mata is also the lead in the Global Parkinson’s Genetics Program (GP2) for underrepresented populations.
Lack of diversity in genetic studies isn’t an issue in Parkinson’s studies alone, Dr. Mata said. “This is a generalized problem across all genetic studies,” he said, citing a 2016 analysis that found the proportion of participants in genome-wide studies was 96% European descent in 2009, shifting to 80% by 2016. “There’s still a big gap because most of the non-European populations came mostly from Asia,” Dr. Mata said, with Latinos and people of African descent representing less than 1% of the study populations.
In an interview, Dr. Mata noted there are a multitude of reasons for enrolling more diverse populations. “We’re going to be able to use genetics to create new treatments and do risk prediction – the so-called precision or personalized medicine,” he said. “We’re leaving a big chunk of the population behind if we don’t include those individuals.”
Scientific basis for diversity
There are a multitude of scientific reasons for doing so, too, said Dr. Mata. “In the whole genome we try to find gene variants that modify the risk for certain disease,” he said. “These regions can be quite large, so increasing the number of individuals that come from different genetic backgrounds can actually help us reduce the number of regions that need to be studied to find the causal variants.”
Andrew Singleton, PhD, director of the Center for Alzheimer’s and Related Dementias at the National Institute of Aging in Bethesda, Md., concurred that enrolling more diverse populations can speed up research for targeting genetic variants.
“We can use the differences in genetics to narrow down our search for variants, reduce the places where we’re looking for risk variants, and reduce the number of genes we’re looking at,” he said in an interview.
Roy Alcalay, MD, professor of neurology at Columbia University Irving Medical Center in New York, offered two more scientific reasons for more diverse study populations “in addition to being more ethically appropriate,” he said. “One is, you may identify new genes that you wouldn’t have identified otherwise; and also in the genes that already exist, you may recognize that some of the pathogenic variants may be more prevalent in populations that were unknown.”
One of the challenges in casting a wider net is that much of the research funding has been concentrated in the United States and Europe, Dr. Mata said. And even in the United States, with large, diverse populations, minority groups are underrepresented in these studies, he said, but a potential solution is emerging. “This is something that we’re learning now with COVID,” he said. “We can do a lot remotely. This should help bring some of those barriers down.”
Cultural barriers are also foreboding. “Individuals may not feel comfortable participating in research,” he said in the interview. “I see this especially in the Hispanic community; many don’t understand what they can do with genetic material, or they’re afraid it will be shared with police, and if they’re here in nonofficial immigration status, they’re afraid they could be deported. There are a lot of misconceptions about genetic research.”
An initiative of the Parkinson’s Foundation PD GENEration Study is to provide free genetic tests and give the patient a report on genetic counseling “to empower patients,” Dr. Mata said.
Solutions for targeting underrepresented groups are emerging, Dr. Singleton said. “Actually there’s a really elegant solution, which is that in the populations that we go into and work with, we make sure the ownership of those cohorts, the ownership of the science and the analysis belongs to those populations,” he said.
“Part of that is creating infrastructure on site,” Dr. Singleton added. “Another part is providing training and outreach so we can help to train a whole new generation of scientists and researchers who can work in those populations embedded within those populations. They’re really the champion of moving that research forward.”
Dr. Alcalay credited Dr. Mata for his work with cohorts in Central and South America and in reaching out to other countries to recruit more diverse populations for genetic Parkinson’s studies. “And it’s not just because it’s the politically correct thing to do, about inclusivity and diversity,” Dr. Alcalay said. “It’s because it’s really meaningful. In addition to being ethically more appropriate, it will advance the entire field.
“I also really think it’s a no-brainer,” Dr. Alcalay said. “It’s something that needs to happen.”
Dr. Mata receives grant funding from the National Institutes of Health. Dr. Singleton and Dr. Alcalay have no relevant disclosures.
FROM MDS VIRTUAL CONGRESS 2021













