User login
Americans’ sun protection practices fall short of intentions
commissioned by the American Academy of Dermatology.
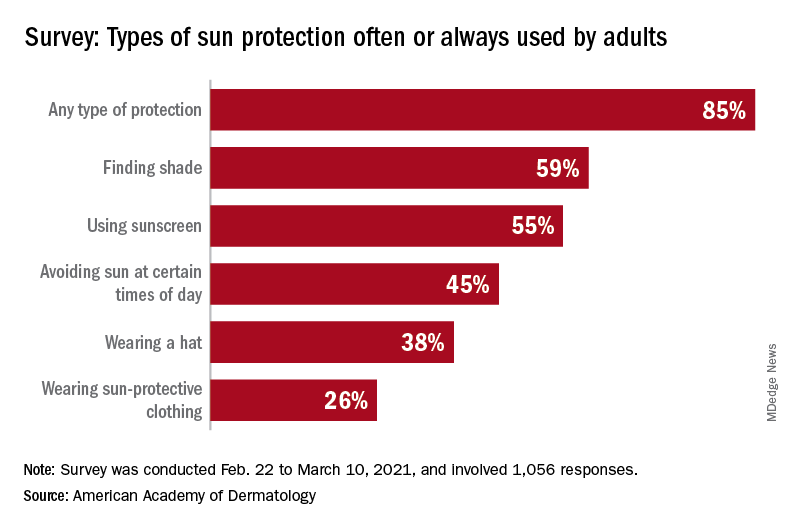
With the pandemic seemingly behind it, the United States enters the summer months facing the paradox of sun protection. Four out of five adults know that sunscreen should be reapplied every 2 hours when they’re outdoors, but only one in three make the actual effort, and 77% are likely to use sunscreen at the beach or a pool, compared with 41% when they’re gardening or working outside on their homes, the AAD reported.
“These findings are surprising and seem to suggest that many people do not take skin cancer seriously or perhaps believe skin cancer won’t happen to them,” Robert T. Brodell, MD, professor of dermatology at the University of Mississippi Medical Center, Jackson, said in a written statement from the AAD, adding that “unprotected exposure to ultraviolet rays is the most preventable risk factor for skin cancer, including melanoma.”
A quarter of all survey respondents reported getting sunburned in 2020, with the youngest adults most likely to feel the wrath of the sun. Sunburn was reported by 43% of those aged 18-23 years, 37% of those aged 24-39, 25% of the 40- to 55-year-olds, 12% of the 56- to 74-year-olds, and 7% of those aged 75 and older. More than a quarter of those who got sunburned said that it was bad enough to make their clothes feel uncomfortable, the academy said.
“Americans see the damaging effects of the sun on their skin as they get older, and two out of three look back and wish they had been more careful. But when it comes to cancer, specifically, most feel unconcerned in spite of their own risk,” according to a statement from Versta Research, which conducted the poll on behalf of the AAD. The survey was conducted from Feb. 22 to March 10, 2021, and involved 1,056 respondents, with a ±3% margin of error.
The lack of concern for skin cancer looks like this: More than two-thirds of the respondents (69%) have at least one possible risk factor – lighter skin tone, blue or green eyes, more than 50 moles, family history – but only 36% expressed concern about developing it. “Indeed, half of survey respondents (49%) say they are more worried about avoiding sunburn than they are about preventing skin cancer, and a third (32%) are more worried about avoiding premature wrinkles than they are about preventing cancer,” the AAD said.
The AAD is considering the creation of a social media quiz or interactive tool, and if the results of this survey were recast as a potential “Knowledge and Awareness Quiz” and graded with a traditional scheme (A = 90%-100%, B = 80%-89%, etc.), then 34% of the respondents would have failed, 15% would have gotten a D, and only 5% would have earned an A, the academy noted.
commissioned by the American Academy of Dermatology.

With the pandemic seemingly behind it, the United States enters the summer months facing the paradox of sun protection. Four out of five adults know that sunscreen should be reapplied every 2 hours when they’re outdoors, but only one in three make the actual effort, and 77% are likely to use sunscreen at the beach or a pool, compared with 41% when they’re gardening or working outside on their homes, the AAD reported.
“These findings are surprising and seem to suggest that many people do not take skin cancer seriously or perhaps believe skin cancer won’t happen to them,” Robert T. Brodell, MD, professor of dermatology at the University of Mississippi Medical Center, Jackson, said in a written statement from the AAD, adding that “unprotected exposure to ultraviolet rays is the most preventable risk factor for skin cancer, including melanoma.”
A quarter of all survey respondents reported getting sunburned in 2020, with the youngest adults most likely to feel the wrath of the sun. Sunburn was reported by 43% of those aged 18-23 years, 37% of those aged 24-39, 25% of the 40- to 55-year-olds, 12% of the 56- to 74-year-olds, and 7% of those aged 75 and older. More than a quarter of those who got sunburned said that it was bad enough to make their clothes feel uncomfortable, the academy said.
“Americans see the damaging effects of the sun on their skin as they get older, and two out of three look back and wish they had been more careful. But when it comes to cancer, specifically, most feel unconcerned in spite of their own risk,” according to a statement from Versta Research, which conducted the poll on behalf of the AAD. The survey was conducted from Feb. 22 to March 10, 2021, and involved 1,056 respondents, with a ±3% margin of error.
The lack of concern for skin cancer looks like this: More than two-thirds of the respondents (69%) have at least one possible risk factor – lighter skin tone, blue or green eyes, more than 50 moles, family history – but only 36% expressed concern about developing it. “Indeed, half of survey respondents (49%) say they are more worried about avoiding sunburn than they are about preventing skin cancer, and a third (32%) are more worried about avoiding premature wrinkles than they are about preventing cancer,” the AAD said.
The AAD is considering the creation of a social media quiz or interactive tool, and if the results of this survey were recast as a potential “Knowledge and Awareness Quiz” and graded with a traditional scheme (A = 90%-100%, B = 80%-89%, etc.), then 34% of the respondents would have failed, 15% would have gotten a D, and only 5% would have earned an A, the academy noted.
commissioned by the American Academy of Dermatology.

With the pandemic seemingly behind it, the United States enters the summer months facing the paradox of sun protection. Four out of five adults know that sunscreen should be reapplied every 2 hours when they’re outdoors, but only one in three make the actual effort, and 77% are likely to use sunscreen at the beach or a pool, compared with 41% when they’re gardening or working outside on their homes, the AAD reported.
“These findings are surprising and seem to suggest that many people do not take skin cancer seriously or perhaps believe skin cancer won’t happen to them,” Robert T. Brodell, MD, professor of dermatology at the University of Mississippi Medical Center, Jackson, said in a written statement from the AAD, adding that “unprotected exposure to ultraviolet rays is the most preventable risk factor for skin cancer, including melanoma.”
A quarter of all survey respondents reported getting sunburned in 2020, with the youngest adults most likely to feel the wrath of the sun. Sunburn was reported by 43% of those aged 18-23 years, 37% of those aged 24-39, 25% of the 40- to 55-year-olds, 12% of the 56- to 74-year-olds, and 7% of those aged 75 and older. More than a quarter of those who got sunburned said that it was bad enough to make their clothes feel uncomfortable, the academy said.
“Americans see the damaging effects of the sun on their skin as they get older, and two out of three look back and wish they had been more careful. But when it comes to cancer, specifically, most feel unconcerned in spite of their own risk,” according to a statement from Versta Research, which conducted the poll on behalf of the AAD. The survey was conducted from Feb. 22 to March 10, 2021, and involved 1,056 respondents, with a ±3% margin of error.
The lack of concern for skin cancer looks like this: More than two-thirds of the respondents (69%) have at least one possible risk factor – lighter skin tone, blue or green eyes, more than 50 moles, family history – but only 36% expressed concern about developing it. “Indeed, half of survey respondents (49%) say they are more worried about avoiding sunburn than they are about preventing skin cancer, and a third (32%) are more worried about avoiding premature wrinkles than they are about preventing cancer,” the AAD said.
The AAD is considering the creation of a social media quiz or interactive tool, and if the results of this survey were recast as a potential “Knowledge and Awareness Quiz” and graded with a traditional scheme (A = 90%-100%, B = 80%-89%, etc.), then 34% of the respondents would have failed, 15% would have gotten a D, and only 5% would have earned an A, the academy noted.
Nivolumab-Induced Granuloma Annulare
Granuloma annulare (GA) is a benign, cutaneous, granulomatous disease of unclear etiology. Typically, GA presents in young adults as asymptomatic, annular, flesh-colored to pink papules and plaques, commonly on the upper and lower extremities. Histologically, GA is characterized by mucin deposition, palisading or an interstitial granulomatous pattern, and collagen and elastic fiber degeneration.1
Granuloma annulare has been associated with various medications and medical conditions, including diabetes mellitus, hyperlipidemia, thyroid disease, and HIV.1 More recently, immune-checkpoint inhibitors (ICIs) have been reported to trigger GA.2 We report a case of nivolumab-induced GA in a 54-year-old woman.
Case Report
A 54-year-old woman presented with an itchy rash on the upper extremities, face, and chest of 4 months’ duration. The patient noted that the rash started on the hands and progressed to include the arms, face, and chest. She also reported associated mild tenderness. She had a history of stage IV non–small-cell lung carcinoma with metastases to the ribs and adrenal glands. She had been started on biweekly intravenous infusions of the ICI nivolumab by her oncologist approximately 1 year prior to the current presentation after failing a course of conventional chemotherapy. The most recent positron emission tomography–computed tomography scan 1 month prior to presentation showed a stable lung mass with radiologic disappearance of metastases, indicating a favorable response to nivolumab. The patient also had a history of hypothyroidism and depression, which were treated with oral levothyroxine 75 μg once daily and oral sertraline 50 mg once daily, respectively, both for longer than 5 years.
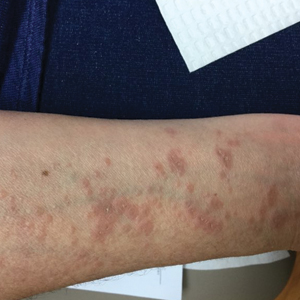
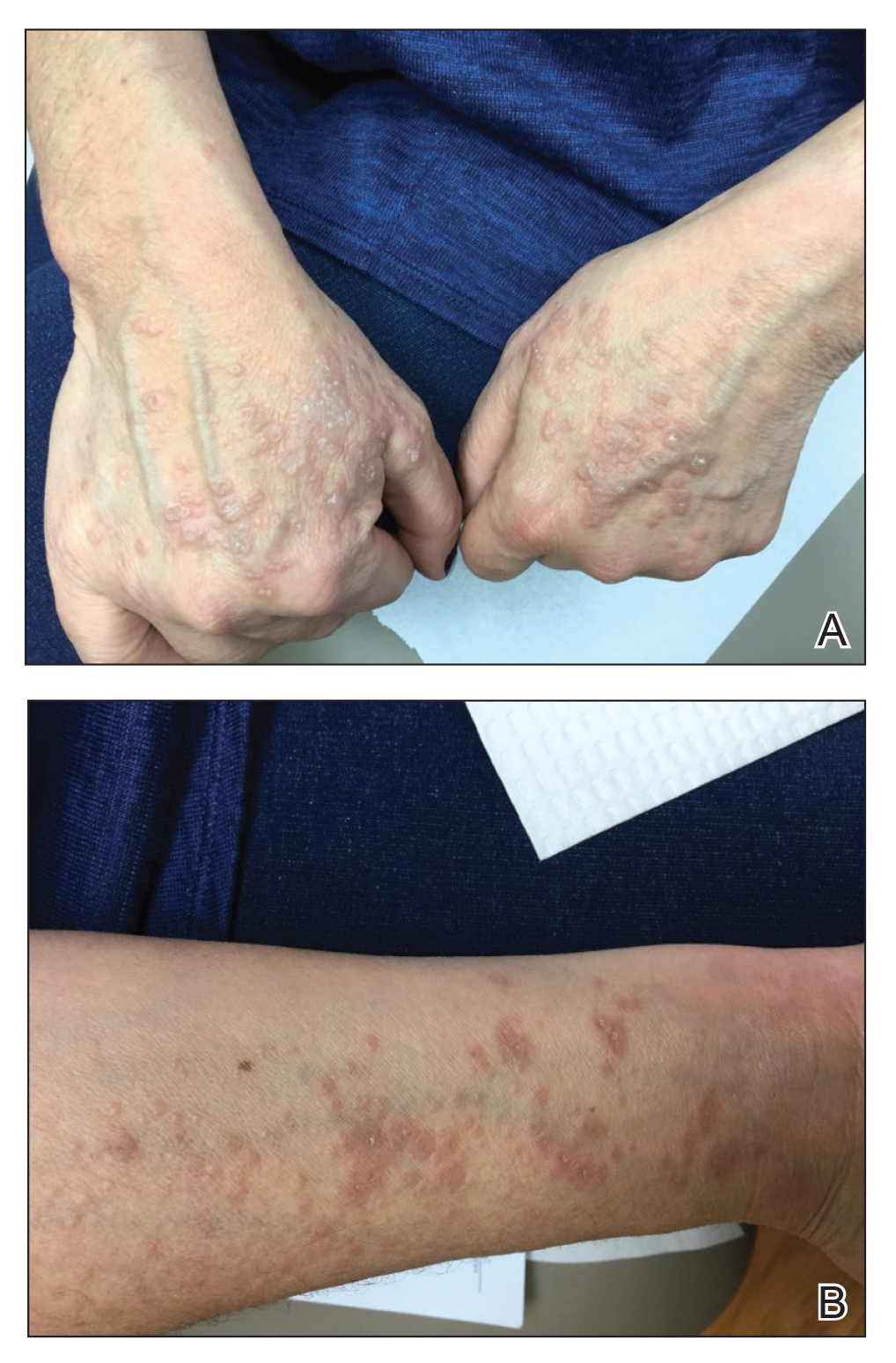
Physical examination revealed annular, erythematous, flat-topped papules, some with surmounting fine scale, coalescing into larger plaques along the dorsal surface of the hands and arms (Figure 1) as well as the forehead and chest. A biopsy of a papule on the dorsal aspect of the left hand revealed nodules of histiocytes admixed with Langerhans giant cells within the dermis; mucin was noted centrally within some nodules (Figure 2). Periodic acid–Schiff staining was negative for fungal elements compared to control. Polarization of the specimen was negative for foreign bodies. The biopsy findings therefore were consistent with a diagnosis of GA.
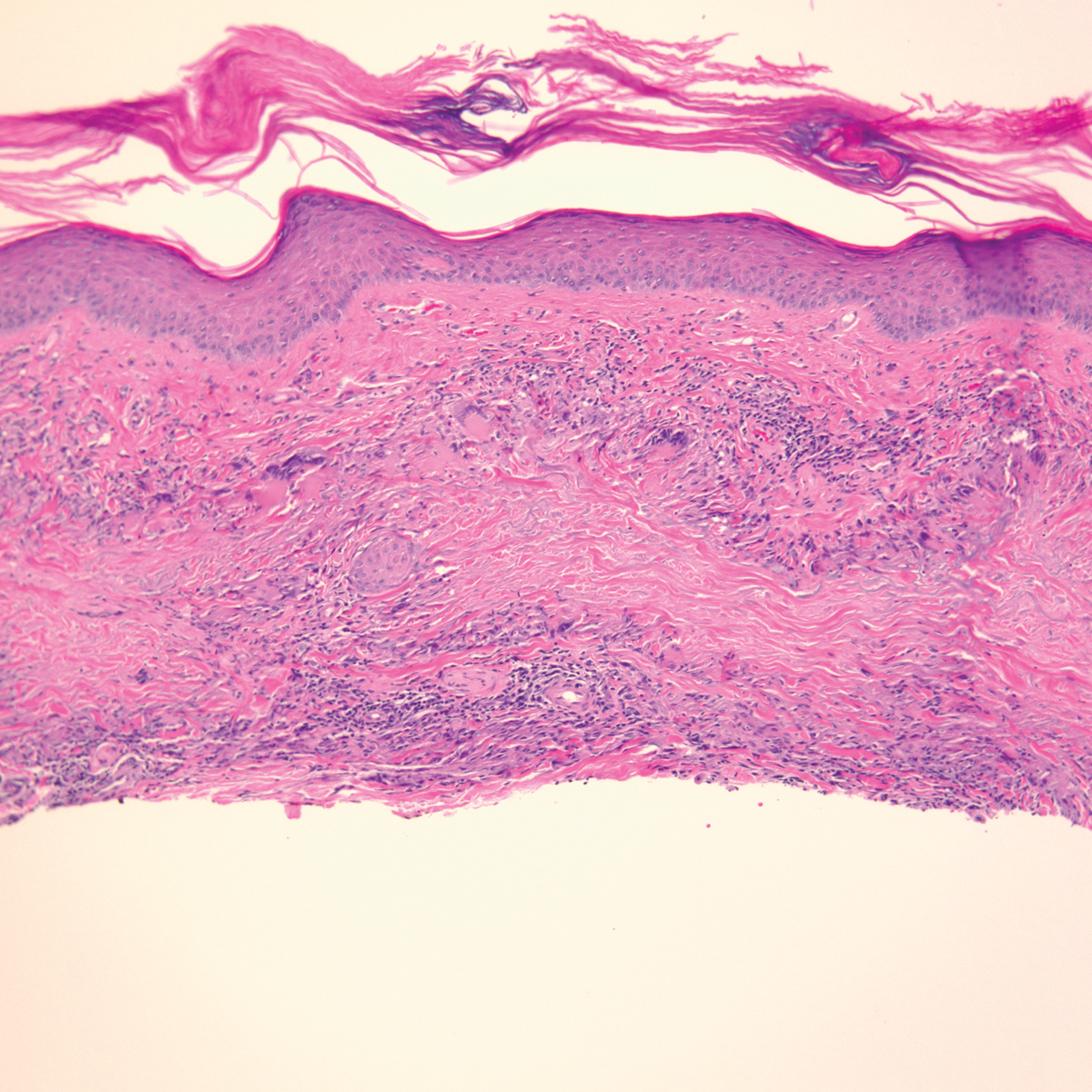
A 3-month treatment course of betamethasone dipropionate 0.05% cream twice daily failed. Narrowband UVB phototherapy was then initiated at 3 sessions weekly. The eruption of GA improved after 3 months of phototherapy. Subsequently, the patient was lost to follow-up.
Comment
Discovery of specific immune checkpoints in tumor-induced immunosuppression revolutionized oncologic therapy. An example is the programmed cell-death protein 1 (PD-1) receptor that is expressed on activated immune cells, including T cells and macrophages.3,4 Upon binding to the PD-1 ligand (PD-L1), T-cell proliferation is inhibited, resulting in downregulation of the immune response. As a result, tumor cells have evolved to overexpress PD-L1 to evade immunologic detection.3 Nivolumab, a fully human IgG4 antibody to PD-1, has emerged along with other ICIs as effective treatments for numerous cancers, including melanoma and non–small-cell lung cancer. By disrupting downregulation of T cells, ICIs improve immune-mediated antitumor activity.3
However, the resulting immunologic disturbance by ICIs has been reported to induce various cutaneous and systemic immune-mediated adverse reactions, including granulomatous reactions such as sarcoidosis, GA, and a cutaneous sarcoidlike granulomatous reaction.1,2,5,6 Our patient represents a rare case of nivolumab-induced GA.
Recent evidence suggests that GA might be caused in part by a cell-mediated hypersensitivity reaction that is regulated by a helper T cell subset 1 inflammatory reaction. Through release of cytokines by activated CD4+ T cells, macrophages are recruited, forming the granulomatous pattern and secreting enzymes that can degrade connective tissue. Nivolumab and other ICIs can thus trigger this reaction because their blockade of PD-1 enhances T cell–mediated immune reactions.2 In addition, because macrophages themselves also express PD-1, ICIs can directly enhance macrophage recruitment and proliferation, further increasing the risk of a granulomatous reaction.4
Interestingly, cutaneous adverse reactions to nivolumab have been associated with improved survival in melanoma patients.7 The nature of this association with granulomatous reactions in general and with GA specifically remains to be determined.
Conclusion
Since the approval of the first PD-1 inhibitors, pembrolizumab and nivolumab, in 2014, other ICIs targeting the immune checkpoint pathway have been developed. Newer agents targeting PD-L1 (avelumab, atezolizumab, and durvalumab) were recently approved. Additionally, cemiplimab, another PD-1 inhibitor, was approved by the US Food and Drug Administration in 2018 for the treatment of advanced cutaneous squamous cell carcinoma.8 Indications for all ICIs also have expanded considerably.3 Therefore, the incidence of immune-mediated adverse reactions, including GA, is bound to increase. Physicians should be cognizant of this association to accurately diagnose and effectively treat adverse reactions in patients who are taking ICIs.
- Piette EW, Rosenbach M. Granuloma annulare: pathogenesis, disease associations and triggers, and therapeutic options. J Am Acad Dermatol. 2016;75:467-479. doi:10.1016/j.jaad.2015.03.055
- Wu J, Kwong BY, Martires KJ, et al. Granuloma annulare associated with immune checkpoint inhibitors. J Eur Acad Dermatol. 2018;32:E124-E126. doi:10.1111/jdv.14617
- Gong J, Chehrazi-Raffle A, Reddi S, et al. Development of PD-1 and PD-L1 inhibitors as a form of cancer immunotherapy: a comprehensive review of registration trials and future considerations. J Immunother Cancer. 2018;6:8. doi:10.1186/s40425-018-0316-z
- Gordon SR, Maute RL, Dulken BW, et al. PD-1 expression by tumour-associated macrophages inhibits phagocytosis and tumour immunity. Nature. 2017;545:495-499. doi:10.1038/nature22396
- Birnbaum MR, Ma MW, Fleisig S, et al. Nivolumab-related cutaneous sarcoidosis in a patient with lung adenocarcinoma. JAAD Case Rep. 2017;3:208-211. doi:10.1016/j.jdcr.2017.02.015
- Danlos F-X, Pagès C, Baroudjian B, et al. Nivolumab-induced sarcoid-like granulomatous reaction in a patient with advanced melanoma. Chest. 2016;149:E133-E136. doi:10.1016/j.chest.2015.10.082
- Freeman-Keller M, Kim Y, Cronin H, et al. Nivolumab in resected and unresectable metastatic melanoma: characteristics of immune-related adverse events and association with outcomes. Clin Cancer Res. 2016;22:886-894. doi:10.1158/1078-0432.CCR-15-1136
- Migden MR, Rischin D, Schmults CD, et al. PD-1 blockade with cemiplimab in advanced cutaneous squamous-cell carcinoma. N Engl J Med. 2018;379:341-351. doi:10.1056/NEJMoa1805131
Granuloma annulare (GA) is a benign, cutaneous, granulomatous disease of unclear etiology. Typically, GA presents in young adults as asymptomatic, annular, flesh-colored to pink papules and plaques, commonly on the upper and lower extremities. Histologically, GA is characterized by mucin deposition, palisading or an interstitial granulomatous pattern, and collagen and elastic fiber degeneration.1
Granuloma annulare has been associated with various medications and medical conditions, including diabetes mellitus, hyperlipidemia, thyroid disease, and HIV.1 More recently, immune-checkpoint inhibitors (ICIs) have been reported to trigger GA.2 We report a case of nivolumab-induced GA in a 54-year-old woman.
Case Report
A 54-year-old woman presented with an itchy rash on the upper extremities, face, and chest of 4 months’ duration. The patient noted that the rash started on the hands and progressed to include the arms, face, and chest. She also reported associated mild tenderness. She had a history of stage IV non–small-cell lung carcinoma with metastases to the ribs and adrenal glands. She had been started on biweekly intravenous infusions of the ICI nivolumab by her oncologist approximately 1 year prior to the current presentation after failing a course of conventional chemotherapy. The most recent positron emission tomography–computed tomography scan 1 month prior to presentation showed a stable lung mass with radiologic disappearance of metastases, indicating a favorable response to nivolumab. The patient also had a history of hypothyroidism and depression, which were treated with oral levothyroxine 75 μg once daily and oral sertraline 50 mg once daily, respectively, both for longer than 5 years.
Physical examination revealed annular, erythematous, flat-topped papules, some with surmounting fine scale, coalescing into larger plaques along the dorsal surface of the hands and arms (Figure 1) as well as the forehead and chest. A biopsy of a papule on the dorsal aspect of the left hand revealed nodules of histiocytes admixed with Langerhans giant cells within the dermis; mucin was noted centrally within some nodules (Figure 2). Periodic acid–Schiff staining was negative for fungal elements compared to control. Polarization of the specimen was negative for foreign bodies. The biopsy findings therefore were consistent with a diagnosis of GA.


A 3-month treatment course of betamethasone dipropionate 0.05% cream twice daily failed. Narrowband UVB phototherapy was then initiated at 3 sessions weekly. The eruption of GA improved after 3 months of phototherapy. Subsequently, the patient was lost to follow-up.
Comment
Discovery of specific immune checkpoints in tumor-induced immunosuppression revolutionized oncologic therapy. An example is the programmed cell-death protein 1 (PD-1) receptor that is expressed on activated immune cells, including T cells and macrophages.3,4 Upon binding to the PD-1 ligand (PD-L1), T-cell proliferation is inhibited, resulting in downregulation of the immune response. As a result, tumor cells have evolved to overexpress PD-L1 to evade immunologic detection.3 Nivolumab, a fully human IgG4 antibody to PD-1, has emerged along with other ICIs as effective treatments for numerous cancers, including melanoma and non–small-cell lung cancer. By disrupting downregulation of T cells, ICIs improve immune-mediated antitumor activity.3
However, the resulting immunologic disturbance by ICIs has been reported to induce various cutaneous and systemic immune-mediated adverse reactions, including granulomatous reactions such as sarcoidosis, GA, and a cutaneous sarcoidlike granulomatous reaction.1,2,5,6 Our patient represents a rare case of nivolumab-induced GA.
Recent evidence suggests that GA might be caused in part by a cell-mediated hypersensitivity reaction that is regulated by a helper T cell subset 1 inflammatory reaction. Through release of cytokines by activated CD4+ T cells, macrophages are recruited, forming the granulomatous pattern and secreting enzymes that can degrade connective tissue. Nivolumab and other ICIs can thus trigger this reaction because their blockade of PD-1 enhances T cell–mediated immune reactions.2 In addition, because macrophages themselves also express PD-1, ICIs can directly enhance macrophage recruitment and proliferation, further increasing the risk of a granulomatous reaction.4
Interestingly, cutaneous adverse reactions to nivolumab have been associated with improved survival in melanoma patients.7 The nature of this association with granulomatous reactions in general and with GA specifically remains to be determined.
Conclusion
Since the approval of the first PD-1 inhibitors, pembrolizumab and nivolumab, in 2014, other ICIs targeting the immune checkpoint pathway have been developed. Newer agents targeting PD-L1 (avelumab, atezolizumab, and durvalumab) were recently approved. Additionally, cemiplimab, another PD-1 inhibitor, was approved by the US Food and Drug Administration in 2018 for the treatment of advanced cutaneous squamous cell carcinoma.8 Indications for all ICIs also have expanded considerably.3 Therefore, the incidence of immune-mediated adverse reactions, including GA, is bound to increase. Physicians should be cognizant of this association to accurately diagnose and effectively treat adverse reactions in patients who are taking ICIs.
Granuloma annulare (GA) is a benign, cutaneous, granulomatous disease of unclear etiology. Typically, GA presents in young adults as asymptomatic, annular, flesh-colored to pink papules and plaques, commonly on the upper and lower extremities. Histologically, GA is characterized by mucin deposition, palisading or an interstitial granulomatous pattern, and collagen and elastic fiber degeneration.1
Granuloma annulare has been associated with various medications and medical conditions, including diabetes mellitus, hyperlipidemia, thyroid disease, and HIV.1 More recently, immune-checkpoint inhibitors (ICIs) have been reported to trigger GA.2 We report a case of nivolumab-induced GA in a 54-year-old woman.
Case Report
A 54-year-old woman presented with an itchy rash on the upper extremities, face, and chest of 4 months’ duration. The patient noted that the rash started on the hands and progressed to include the arms, face, and chest. She also reported associated mild tenderness. She had a history of stage IV non–small-cell lung carcinoma with metastases to the ribs and adrenal glands. She had been started on biweekly intravenous infusions of the ICI nivolumab by her oncologist approximately 1 year prior to the current presentation after failing a course of conventional chemotherapy. The most recent positron emission tomography–computed tomography scan 1 month prior to presentation showed a stable lung mass with radiologic disappearance of metastases, indicating a favorable response to nivolumab. The patient also had a history of hypothyroidism and depression, which were treated with oral levothyroxine 75 μg once daily and oral sertraline 50 mg once daily, respectively, both for longer than 5 years.
Physical examination revealed annular, erythematous, flat-topped papules, some with surmounting fine scale, coalescing into larger plaques along the dorsal surface of the hands and arms (Figure 1) as well as the forehead and chest. A biopsy of a papule on the dorsal aspect of the left hand revealed nodules of histiocytes admixed with Langerhans giant cells within the dermis; mucin was noted centrally within some nodules (Figure 2). Periodic acid–Schiff staining was negative for fungal elements compared to control. Polarization of the specimen was negative for foreign bodies. The biopsy findings therefore were consistent with a diagnosis of GA.


A 3-month treatment course of betamethasone dipropionate 0.05% cream twice daily failed. Narrowband UVB phototherapy was then initiated at 3 sessions weekly. The eruption of GA improved after 3 months of phototherapy. Subsequently, the patient was lost to follow-up.
Comment
Discovery of specific immune checkpoints in tumor-induced immunosuppression revolutionized oncologic therapy. An example is the programmed cell-death protein 1 (PD-1) receptor that is expressed on activated immune cells, including T cells and macrophages.3,4 Upon binding to the PD-1 ligand (PD-L1), T-cell proliferation is inhibited, resulting in downregulation of the immune response. As a result, tumor cells have evolved to overexpress PD-L1 to evade immunologic detection.3 Nivolumab, a fully human IgG4 antibody to PD-1, has emerged along with other ICIs as effective treatments for numerous cancers, including melanoma and non–small-cell lung cancer. By disrupting downregulation of T cells, ICIs improve immune-mediated antitumor activity.3
However, the resulting immunologic disturbance by ICIs has been reported to induce various cutaneous and systemic immune-mediated adverse reactions, including granulomatous reactions such as sarcoidosis, GA, and a cutaneous sarcoidlike granulomatous reaction.1,2,5,6 Our patient represents a rare case of nivolumab-induced GA.
Recent evidence suggests that GA might be caused in part by a cell-mediated hypersensitivity reaction that is regulated by a helper T cell subset 1 inflammatory reaction. Through release of cytokines by activated CD4+ T cells, macrophages are recruited, forming the granulomatous pattern and secreting enzymes that can degrade connective tissue. Nivolumab and other ICIs can thus trigger this reaction because their blockade of PD-1 enhances T cell–mediated immune reactions.2 In addition, because macrophages themselves also express PD-1, ICIs can directly enhance macrophage recruitment and proliferation, further increasing the risk of a granulomatous reaction.4
Interestingly, cutaneous adverse reactions to nivolumab have been associated with improved survival in melanoma patients.7 The nature of this association with granulomatous reactions in general and with GA specifically remains to be determined.
Conclusion
Since the approval of the first PD-1 inhibitors, pembrolizumab and nivolumab, in 2014, other ICIs targeting the immune checkpoint pathway have been developed. Newer agents targeting PD-L1 (avelumab, atezolizumab, and durvalumab) were recently approved. Additionally, cemiplimab, another PD-1 inhibitor, was approved by the US Food and Drug Administration in 2018 for the treatment of advanced cutaneous squamous cell carcinoma.8 Indications for all ICIs also have expanded considerably.3 Therefore, the incidence of immune-mediated adverse reactions, including GA, is bound to increase. Physicians should be cognizant of this association to accurately diagnose and effectively treat adverse reactions in patients who are taking ICIs.
- Piette EW, Rosenbach M. Granuloma annulare: pathogenesis, disease associations and triggers, and therapeutic options. J Am Acad Dermatol. 2016;75:467-479. doi:10.1016/j.jaad.2015.03.055
- Wu J, Kwong BY, Martires KJ, et al. Granuloma annulare associated with immune checkpoint inhibitors. J Eur Acad Dermatol. 2018;32:E124-E126. doi:10.1111/jdv.14617
- Gong J, Chehrazi-Raffle A, Reddi S, et al. Development of PD-1 and PD-L1 inhibitors as a form of cancer immunotherapy: a comprehensive review of registration trials and future considerations. J Immunother Cancer. 2018;6:8. doi:10.1186/s40425-018-0316-z
- Gordon SR, Maute RL, Dulken BW, et al. PD-1 expression by tumour-associated macrophages inhibits phagocytosis and tumour immunity. Nature. 2017;545:495-499. doi:10.1038/nature22396
- Birnbaum MR, Ma MW, Fleisig S, et al. Nivolumab-related cutaneous sarcoidosis in a patient with lung adenocarcinoma. JAAD Case Rep. 2017;3:208-211. doi:10.1016/j.jdcr.2017.02.015
- Danlos F-X, Pagès C, Baroudjian B, et al. Nivolumab-induced sarcoid-like granulomatous reaction in a patient with advanced melanoma. Chest. 2016;149:E133-E136. doi:10.1016/j.chest.2015.10.082
- Freeman-Keller M, Kim Y, Cronin H, et al. Nivolumab in resected and unresectable metastatic melanoma: characteristics of immune-related adverse events and association with outcomes. Clin Cancer Res. 2016;22:886-894. doi:10.1158/1078-0432.CCR-15-1136
- Migden MR, Rischin D, Schmults CD, et al. PD-1 blockade with cemiplimab in advanced cutaneous squamous-cell carcinoma. N Engl J Med. 2018;379:341-351. doi:10.1056/NEJMoa1805131
- Piette EW, Rosenbach M. Granuloma annulare: pathogenesis, disease associations and triggers, and therapeutic options. J Am Acad Dermatol. 2016;75:467-479. doi:10.1016/j.jaad.2015.03.055
- Wu J, Kwong BY, Martires KJ, et al. Granuloma annulare associated with immune checkpoint inhibitors. J Eur Acad Dermatol. 2018;32:E124-E126. doi:10.1111/jdv.14617
- Gong J, Chehrazi-Raffle A, Reddi S, et al. Development of PD-1 and PD-L1 inhibitors as a form of cancer immunotherapy: a comprehensive review of registration trials and future considerations. J Immunother Cancer. 2018;6:8. doi:10.1186/s40425-018-0316-z
- Gordon SR, Maute RL, Dulken BW, et al. PD-1 expression by tumour-associated macrophages inhibits phagocytosis and tumour immunity. Nature. 2017;545:495-499. doi:10.1038/nature22396
- Birnbaum MR, Ma MW, Fleisig S, et al. Nivolumab-related cutaneous sarcoidosis in a patient with lung adenocarcinoma. JAAD Case Rep. 2017;3:208-211. doi:10.1016/j.jdcr.2017.02.015
- Danlos F-X, Pagès C, Baroudjian B, et al. Nivolumab-induced sarcoid-like granulomatous reaction in a patient with advanced melanoma. Chest. 2016;149:E133-E136. doi:10.1016/j.chest.2015.10.082
- Freeman-Keller M, Kim Y, Cronin H, et al. Nivolumab in resected and unresectable metastatic melanoma: characteristics of immune-related adverse events and association with outcomes. Clin Cancer Res. 2016;22:886-894. doi:10.1158/1078-0432.CCR-15-1136
- Migden MR, Rischin D, Schmults CD, et al. PD-1 blockade with cemiplimab in advanced cutaneous squamous-cell carcinoma. N Engl J Med. 2018;379:341-351. doi:10.1056/NEJMoa1805131
Practice Points
- Immune-related adverse events (irAEs) frequently occur in patients on immunotherapy, with the skin representing the most common site of involvement.
- Although rare, granulomatous reactions such as granuloma annulare increasingly are recognized as potential irAEs.
- Clinicians should be aware of this novel association to accurately diagnose and effectively treat adverse reactions in patients receiving immunotherapy.
Benzene was found in some sunscreens. Now what?
Just before Memorial Day, which include batches from Neutrogena, Banana Boat, CVS Health, and other brands. More than three-quarters of the products are sprays.
“We’re asking our patients to put sunscreen on from 6 months of age, telling them to do it their entire life, their whole body, multiple times a day,” Christopher G. Bunick, MD, PhD, associate professor of dermatology at Yale University, New Haven, Conn., said in an interview. If benzene-contaminated sunscreen proves to be a widespread problem, he said, “the benzene amounts can add up to a significant chronic exposure over a lifetime.”
In the Valisure statement announcing the findings, Dr. Bunick, who is also quoted in the petition, said that “it is critical that regulatory agencies address benzene contamination in sunscreens, and all topical medications at the manufacturing and final product level, so that all individuals feel safe using sunscreen products.”
The list of products that tested positive is included in the citizen petition, and a full list of products that did not show any contamination is available in an attachment.
Benzene is not an ingredient in sunscreen, and Valisure’s petition suggests that the findings are a result of contamination somewhere in the manufacturing process, not of product degradation.
“This isn’t a sunscreen issue, it’s a manufacturing issue,” said Adam Friedman, MD, professor and chief of dermatology at George Washington University, Washington. “We don’t want those things to be blurred.”
When asked to comment on Valisure’s findings, an FDA spokesperson said, “The FDA takes seriously any safety concerns raised about products we regulate, including sunscreen. While the agency evaluates the submitted citizen petition, we will continue to monitor the sunscreen marketplace and manufacturing efforts to help ensure the availability of safe sunscreens for U.S. consumers.”
Both Johnson & Johnson, Neutrogena’s parent company, and Banana Boat issued statements reiterating that benzene is not an ingredient in their products.
Assessing the risks
There is a risk of patients taking away the wrong message from these findings.
“People already have ambivalence about sunscreen, and this is just going to make that worse,” Dr. Friedman said in an interview. He pointed out that benzene is present in car exhaust, second-hand smoke, and elsewhere. Inhalation exposure has been the primary focus of toxicology investigations, as has exposure from ingesting things such as contaminated drinking water – not via topical application. “We don’t know how effectively [benzene] gets through the skin, if it gets absorbed systemically, and how that then behaves downstream,” he noted.
On the other hand, ultraviolet radiation is a well-established carcinogen. Avoiding an effective preventive measure such as sunscreen could prove more harmful than exposure to trace amounts of benzene, ultimately to be determined by the FDA.
“Just because those particular products do pose a risk, that doesn’t erase the message that sunscreens are safe and should be used,” Dr. Bunick said. “It’s not mutually exclusive.”
And then there’s the fact that the benzene contamination appears to be fairly limited. “The majority of products we tested, over 200 of them, had no detectable amounts of benzene, and uncontaminated sunscreen should certainly continue to be used,” David Light, CEO of Valisure, told this news organization.
Advising patients
With headlines blaring the news about a carcinogen in sunscreen, patients will be reaching out for advice.
“The number one question patients will have is, ‘What sunscreen do you recommend?’” said Dr. Bunick. “The answer should be to pick a sunscreen that we know wasn’t contaminated. Reassure your patient the ingredients themselves are effective and safe, and that’s not what’s leading to the contamination.”
Dr. Friedman agrees. “We need to be mindful. Dermatologists need to be armed with the facts in order to counsel patients: Sunscreen is still a very important, effective, and safe, scientifically based way to prevent the harmful effects of the sun, in addition to things like sun protective clothing and seeking shade between 10 a.m. and 4 p.m.”
As alarming as Valisure’s findings may seem initially, Dr. Bunick noted a silver lining. “The consumer, the public should feel reassured this report is out there. It shows that someone’s watching out. That’s an important safety message: These things aren’t going undetected.”
Just before Memorial Day, which include batches from Neutrogena, Banana Boat, CVS Health, and other brands. More than three-quarters of the products are sprays.
“We’re asking our patients to put sunscreen on from 6 months of age, telling them to do it their entire life, their whole body, multiple times a day,” Christopher G. Bunick, MD, PhD, associate professor of dermatology at Yale University, New Haven, Conn., said in an interview. If benzene-contaminated sunscreen proves to be a widespread problem, he said, “the benzene amounts can add up to a significant chronic exposure over a lifetime.”
In the Valisure statement announcing the findings, Dr. Bunick, who is also quoted in the petition, said that “it is critical that regulatory agencies address benzene contamination in sunscreens, and all topical medications at the manufacturing and final product level, so that all individuals feel safe using sunscreen products.”
The list of products that tested positive is included in the citizen petition, and a full list of products that did not show any contamination is available in an attachment.
Benzene is not an ingredient in sunscreen, and Valisure’s petition suggests that the findings are a result of contamination somewhere in the manufacturing process, not of product degradation.
“This isn’t a sunscreen issue, it’s a manufacturing issue,” said Adam Friedman, MD, professor and chief of dermatology at George Washington University, Washington. “We don’t want those things to be blurred.”
When asked to comment on Valisure’s findings, an FDA spokesperson said, “The FDA takes seriously any safety concerns raised about products we regulate, including sunscreen. While the agency evaluates the submitted citizen petition, we will continue to monitor the sunscreen marketplace and manufacturing efforts to help ensure the availability of safe sunscreens for U.S. consumers.”
Both Johnson & Johnson, Neutrogena’s parent company, and Banana Boat issued statements reiterating that benzene is not an ingredient in their products.
Assessing the risks
There is a risk of patients taking away the wrong message from these findings.
“People already have ambivalence about sunscreen, and this is just going to make that worse,” Dr. Friedman said in an interview. He pointed out that benzene is present in car exhaust, second-hand smoke, and elsewhere. Inhalation exposure has been the primary focus of toxicology investigations, as has exposure from ingesting things such as contaminated drinking water – not via topical application. “We don’t know how effectively [benzene] gets through the skin, if it gets absorbed systemically, and how that then behaves downstream,” he noted.
On the other hand, ultraviolet radiation is a well-established carcinogen. Avoiding an effective preventive measure such as sunscreen could prove more harmful than exposure to trace amounts of benzene, ultimately to be determined by the FDA.
“Just because those particular products do pose a risk, that doesn’t erase the message that sunscreens are safe and should be used,” Dr. Bunick said. “It’s not mutually exclusive.”
And then there’s the fact that the benzene contamination appears to be fairly limited. “The majority of products we tested, over 200 of them, had no detectable amounts of benzene, and uncontaminated sunscreen should certainly continue to be used,” David Light, CEO of Valisure, told this news organization.
Advising patients
With headlines blaring the news about a carcinogen in sunscreen, patients will be reaching out for advice.
“The number one question patients will have is, ‘What sunscreen do you recommend?’” said Dr. Bunick. “The answer should be to pick a sunscreen that we know wasn’t contaminated. Reassure your patient the ingredients themselves are effective and safe, and that’s not what’s leading to the contamination.”
Dr. Friedman agrees. “We need to be mindful. Dermatologists need to be armed with the facts in order to counsel patients: Sunscreen is still a very important, effective, and safe, scientifically based way to prevent the harmful effects of the sun, in addition to things like sun protective clothing and seeking shade between 10 a.m. and 4 p.m.”
As alarming as Valisure’s findings may seem initially, Dr. Bunick noted a silver lining. “The consumer, the public should feel reassured this report is out there. It shows that someone’s watching out. That’s an important safety message: These things aren’t going undetected.”
Just before Memorial Day, which include batches from Neutrogena, Banana Boat, CVS Health, and other brands. More than three-quarters of the products are sprays.
“We’re asking our patients to put sunscreen on from 6 months of age, telling them to do it their entire life, their whole body, multiple times a day,” Christopher G. Bunick, MD, PhD, associate professor of dermatology at Yale University, New Haven, Conn., said in an interview. If benzene-contaminated sunscreen proves to be a widespread problem, he said, “the benzene amounts can add up to a significant chronic exposure over a lifetime.”
In the Valisure statement announcing the findings, Dr. Bunick, who is also quoted in the petition, said that “it is critical that regulatory agencies address benzene contamination in sunscreens, and all topical medications at the manufacturing and final product level, so that all individuals feel safe using sunscreen products.”
The list of products that tested positive is included in the citizen petition, and a full list of products that did not show any contamination is available in an attachment.
Benzene is not an ingredient in sunscreen, and Valisure’s petition suggests that the findings are a result of contamination somewhere in the manufacturing process, not of product degradation.
“This isn’t a sunscreen issue, it’s a manufacturing issue,” said Adam Friedman, MD, professor and chief of dermatology at George Washington University, Washington. “We don’t want those things to be blurred.”
When asked to comment on Valisure’s findings, an FDA spokesperson said, “The FDA takes seriously any safety concerns raised about products we regulate, including sunscreen. While the agency evaluates the submitted citizen petition, we will continue to monitor the sunscreen marketplace and manufacturing efforts to help ensure the availability of safe sunscreens for U.S. consumers.”
Both Johnson & Johnson, Neutrogena’s parent company, and Banana Boat issued statements reiterating that benzene is not an ingredient in their products.
Assessing the risks
There is a risk of patients taking away the wrong message from these findings.
“People already have ambivalence about sunscreen, and this is just going to make that worse,” Dr. Friedman said in an interview. He pointed out that benzene is present in car exhaust, second-hand smoke, and elsewhere. Inhalation exposure has been the primary focus of toxicology investigations, as has exposure from ingesting things such as contaminated drinking water – not via topical application. “We don’t know how effectively [benzene] gets through the skin, if it gets absorbed systemically, and how that then behaves downstream,” he noted.
On the other hand, ultraviolet radiation is a well-established carcinogen. Avoiding an effective preventive measure such as sunscreen could prove more harmful than exposure to trace amounts of benzene, ultimately to be determined by the FDA.
“Just because those particular products do pose a risk, that doesn’t erase the message that sunscreens are safe and should be used,” Dr. Bunick said. “It’s not mutually exclusive.”
And then there’s the fact that the benzene contamination appears to be fairly limited. “The majority of products we tested, over 200 of them, had no detectable amounts of benzene, and uncontaminated sunscreen should certainly continue to be used,” David Light, CEO of Valisure, told this news organization.
Advising patients
With headlines blaring the news about a carcinogen in sunscreen, patients will be reaching out for advice.
“The number one question patients will have is, ‘What sunscreen do you recommend?’” said Dr. Bunick. “The answer should be to pick a sunscreen that we know wasn’t contaminated. Reassure your patient the ingredients themselves are effective and safe, and that’s not what’s leading to the contamination.”
Dr. Friedman agrees. “We need to be mindful. Dermatologists need to be armed with the facts in order to counsel patients: Sunscreen is still a very important, effective, and safe, scientifically based way to prevent the harmful effects of the sun, in addition to things like sun protective clothing and seeking shade between 10 a.m. and 4 p.m.”
As alarming as Valisure’s findings may seem initially, Dr. Bunick noted a silver lining. “The consumer, the public should feel reassured this report is out there. It shows that someone’s watching out. That’s an important safety message: These things aren’t going undetected.”
Study findings support consideration of second biopsy for transected melanomas
in a review of cases at the university.
Had their true Breslow depths been known before definitive surgery, sentinel lymph node (SLN) biopsies and wider surgical margins would likely have been recommended.
The findings led the investigators to conclude that a second biopsy should be considered when the first one is transected to ensure surgical and other management decisions are based on an accurate Breslow depth.
A second biopsy is especially warranted for broadly transected biopsies and transected T1a tumors with gross residual tumor or pigment on preoperative exam; both scenarios significantly increased the risk of up-staging in the study, according to lead investigator James Duncan, MD, a Mohs surgery and dermatologic oncology fellow at the University of Alabama at Birmingham, who presented the findings at the annual meeting of the American College of Mohs Surgery.
“Accurate staging of malignancies, especially melanoma, is critical to determine prognosis and the best treatment approach,” said Vishal Patel, MD, director of cutaneous oncology at George Washington University, Washington, when asked for comment.
“This study identifies how transected biopsies can underestimate a melanoma’s true depth and thus impact treatment and outcomes. The authors highlight that when a biopsy is transected, or there is notable pigment at the base, attempts should be taken to sample the remaining tumor prior to surgery so the accurate tumor depth can be determined and treatment options be fully discussed with the patient,” Dr. Patel said.
The Birmingham team reviewed invasive melanoma cases at their university from 2017 to 2019.
Almost half (49.6%) of the 726 melanomas they identified were transected on biopsy, which is in line with prior reports. About 60% of the patients were men and 98% were White; the average age was 63 years.
Of the 360 transected tumors, 49 (13.6%) had up-staging at final excision that “would have prompted discussion of alternate surgical treatment such as SLN biopsy or wider surgical margins,” the team said.
Of the 89 transected pT1a melanomas identified, 47.1% with gross residual tumor or pigment on preoperative physical examination were up-staged following excision versus 6.9% with no remaining pigment or tumor prior to surgery (P < .01).
Broadly transected tumors were up-staged in 21.7% of cases versus 4.9% of focally transected tumors (P = .038). The average increase in Breslow depth for broadly transected tumors was 1.03 mm versus 0.03 mm for focally transected lesions (P = .04).
Shave biopsies, ulceration, and lack of concern for melanoma at the initial biopsy were among the factors associated with a higher risk of transection.
Superficial spreading melanoma was the most common subtype. Tumors were evenly distributed between the head, neck, and extremities. The average Breslow depth was 1.51 mm, and the majority of tumors were pT1a or pT2a.
The review excluded melanoma in situ, recurrences, metastases, noncutaneous melanomas, and biopsies where deep margin status was unknown.
There was no funding for the study, and Dr. Duncan and Dr. Patel had no relevant disclosures.
in a review of cases at the university.
Had their true Breslow depths been known before definitive surgery, sentinel lymph node (SLN) biopsies and wider surgical margins would likely have been recommended.
The findings led the investigators to conclude that a second biopsy should be considered when the first one is transected to ensure surgical and other management decisions are based on an accurate Breslow depth.
A second biopsy is especially warranted for broadly transected biopsies and transected T1a tumors with gross residual tumor or pigment on preoperative exam; both scenarios significantly increased the risk of up-staging in the study, according to lead investigator James Duncan, MD, a Mohs surgery and dermatologic oncology fellow at the University of Alabama at Birmingham, who presented the findings at the annual meeting of the American College of Mohs Surgery.
“Accurate staging of malignancies, especially melanoma, is critical to determine prognosis and the best treatment approach,” said Vishal Patel, MD, director of cutaneous oncology at George Washington University, Washington, when asked for comment.
“This study identifies how transected biopsies can underestimate a melanoma’s true depth and thus impact treatment and outcomes. The authors highlight that when a biopsy is transected, or there is notable pigment at the base, attempts should be taken to sample the remaining tumor prior to surgery so the accurate tumor depth can be determined and treatment options be fully discussed with the patient,” Dr. Patel said.
The Birmingham team reviewed invasive melanoma cases at their university from 2017 to 2019.
Almost half (49.6%) of the 726 melanomas they identified were transected on biopsy, which is in line with prior reports. About 60% of the patients were men and 98% were White; the average age was 63 years.
Of the 360 transected tumors, 49 (13.6%) had up-staging at final excision that “would have prompted discussion of alternate surgical treatment such as SLN biopsy or wider surgical margins,” the team said.
Of the 89 transected pT1a melanomas identified, 47.1% with gross residual tumor or pigment on preoperative physical examination were up-staged following excision versus 6.9% with no remaining pigment or tumor prior to surgery (P < .01).
Broadly transected tumors were up-staged in 21.7% of cases versus 4.9% of focally transected tumors (P = .038). The average increase in Breslow depth for broadly transected tumors was 1.03 mm versus 0.03 mm for focally transected lesions (P = .04).
Shave biopsies, ulceration, and lack of concern for melanoma at the initial biopsy were among the factors associated with a higher risk of transection.
Superficial spreading melanoma was the most common subtype. Tumors were evenly distributed between the head, neck, and extremities. The average Breslow depth was 1.51 mm, and the majority of tumors were pT1a or pT2a.
The review excluded melanoma in situ, recurrences, metastases, noncutaneous melanomas, and biopsies where deep margin status was unknown.
There was no funding for the study, and Dr. Duncan and Dr. Patel had no relevant disclosures.
in a review of cases at the university.
Had their true Breslow depths been known before definitive surgery, sentinel lymph node (SLN) biopsies and wider surgical margins would likely have been recommended.
The findings led the investigators to conclude that a second biopsy should be considered when the first one is transected to ensure surgical and other management decisions are based on an accurate Breslow depth.
A second biopsy is especially warranted for broadly transected biopsies and transected T1a tumors with gross residual tumor or pigment on preoperative exam; both scenarios significantly increased the risk of up-staging in the study, according to lead investigator James Duncan, MD, a Mohs surgery and dermatologic oncology fellow at the University of Alabama at Birmingham, who presented the findings at the annual meeting of the American College of Mohs Surgery.
“Accurate staging of malignancies, especially melanoma, is critical to determine prognosis and the best treatment approach,” said Vishal Patel, MD, director of cutaneous oncology at George Washington University, Washington, when asked for comment.
“This study identifies how transected biopsies can underestimate a melanoma’s true depth and thus impact treatment and outcomes. The authors highlight that when a biopsy is transected, or there is notable pigment at the base, attempts should be taken to sample the remaining tumor prior to surgery so the accurate tumor depth can be determined and treatment options be fully discussed with the patient,” Dr. Patel said.
The Birmingham team reviewed invasive melanoma cases at their university from 2017 to 2019.
Almost half (49.6%) of the 726 melanomas they identified were transected on biopsy, which is in line with prior reports. About 60% of the patients were men and 98% were White; the average age was 63 years.
Of the 360 transected tumors, 49 (13.6%) had up-staging at final excision that “would have prompted discussion of alternate surgical treatment such as SLN biopsy or wider surgical margins,” the team said.
Of the 89 transected pT1a melanomas identified, 47.1% with gross residual tumor or pigment on preoperative physical examination were up-staged following excision versus 6.9% with no remaining pigment or tumor prior to surgery (P < .01).
Broadly transected tumors were up-staged in 21.7% of cases versus 4.9% of focally transected tumors (P = .038). The average increase in Breslow depth for broadly transected tumors was 1.03 mm versus 0.03 mm for focally transected lesions (P = .04).
Shave biopsies, ulceration, and lack of concern for melanoma at the initial biopsy were among the factors associated with a higher risk of transection.
Superficial spreading melanoma was the most common subtype. Tumors were evenly distributed between the head, neck, and extremities. The average Breslow depth was 1.51 mm, and the majority of tumors were pT1a or pT2a.
The review excluded melanoma in situ, recurrences, metastases, noncutaneous melanomas, and biopsies where deep margin status was unknown.
There was no funding for the study, and Dr. Duncan and Dr. Patel had no relevant disclosures.
FROM ACMS 2021
The Power of a Multidisciplinary Tumor Board: Managing Unresectable and/or High-Risk Skin Cancers
Multidisciplinary tumor boards are composed of providers from many fields who deliver coordinated care for patients with unresectable and high-risk skin cancers. Providers who comprise the tumor board often are radiation oncologists, hematologists/oncologists, general surgeons, dermatologists, dermatologic surgeons, and pathologists. The benefit of having a tumor board is that each patient is evaluated simultaneously by a group of physicians from various specialties who bring diverse perspectives that will contribute to the overall treatment plan. The cases often encompass high-risk tumors including unresectable basal cell carcinomas or invasive melanomas. By combining knowledge from each specialty in a team approach, the tumor board can effectively and holistically develop a care plan for each patient.
For the tumor board at the Warren Alpert Medical School of Brown University (Providence, Rhode Island), we often prepare a presentation with comprehensive details about the patient and tumor. During the presentation, we also propose a treatment plan prior to describing each patient at the weekly conference and amend the plans during the discussion. Tumor boards also provide a consulting role to the community and hospital providers in which patients are being referred by their primary provider and are seeking a second opinion or guidance.
In many ways, the tumor board is a multidisciplinary approach for patient advocacy in the form of treatment. These physicians meet on a regular basis to check on the patient’s progress and continually reevaluate how to have discussions about the patient’s care. There are many reasons why it is important to refer patients to a multidisciplinary tumor board.
Improved Workup and Diagnosis
One of the values of a tumor board is that it allows for patient data to be collected and assembled in a way that tells a story. The specialist from each field can then discuss and weigh the benefits and risks for each diagnostic test that should be performed for the workup in each patient. Physicians who refer their patients to the tumor board use their recommendations to both confirm the diagnosis and shift their treatment plans, depending on the information presented during the meeting.1 There may be a change in the tumor type, decision to refer for surgery, cancer staging, and list of viable options, especially after reviewing pathology and imaging.2 The discussion of the treatment plan may consider not only surgical considerations but also the patient’s quality of life. At times, noninvasive interventions are more appropriate and align with the patient’s goals of care. In addition, during the tumor board clinic there may be new tumors that are identified and biopsied, providing increased diagnosis and surveillance for patients who may have a higher risk for developing skin cancer.
Education for Residents and Providers
The multidisciplinary tumor board not only helps patients but also educates both residents and providers on the evidence-based therapeutic management of high-risk tumors.2 Research literature on cutaneous oncology is dynamic, and the weekly tumor board meetings help providers stay informed about the best and most effective treatments for their patients.3 In addition to the attending specialists, participants of the tumor board also may include residents, medical students, medical assistance staff, nurses, physician assistants, and fellows. Furthermore, the recommendations given by the tumor board serve to educate both the patient and the provider who referred them to the tumor board. Although we have access to excellent dermatology textbooks as residents, the most impactful educational experience is seeing the patients in tumor board clinic and participating in the immensely educational discussions at the weekly conferences. Through this experience, I have learned that treatment plans should be personalized to the patient. There are many factors to take into consideration when deciphering what the best course of treatment will be for a patient. Sometimes the best option is Mohs micrographic surgery, while other times it may be scheduling several sessions of palliative radiation oncology. Treatment depends on the individual patient and their condition.
Coordination of Care
During a week that I was on call, I was consulted to biopsy a patient with a giant hemorrhagic basal cell carcinoma that caused substantial cheek and nose distortion as well as anemia secondary to acute blood loss. The patient not only did not have a dermatologist but also did not have a primary care physician given he had not had contact with the health care system in more than 30 years. The reason for him not seeking care was multifactorial, but the approach to his care became multidisciplinary. We sought to connect him with the right providers to help him in any way that we could. We presented him at our multidisciplinary tumor board and started him on sonedigib, a medication that binds to and inhibits the smoothened protein.4 Through the tumor board, we were able to establish sustained contact with the patient. The tumor board created effective communication between providers to get him the referrals that he needed for dermatology, pathology, radiation oncology, hematology/oncology, and otolaryngology. The discussions centered around being cognizant of the patient’s apprehension with the health care system as well as providing medical and surgical treatment that would help his quality of life. We built a consensus on what the best plan was for the patient and his family. This consensus would have been more difficult had it not been for the combined specialties of the tumor board. In general, studies have shown that weekly tumor boards have resulted in decreased mortality rates for patients with advanced cancers.5
Final Thoughts
The multidisciplinary tumor board is a powerful resource for hospitals and the greater medical community. At these weekly conferences you realize there may still be hope that begins at the line where your expertise ends. It represents a team of providers who compassionately refuse to give up on patients when they are the last refuge.
- Foster TJ, Bouchard-Fortier A, Olivotto IA, et al. Effect of multidisciplinary case conferences on physician decision making: breast diagnostic rounds. Cureus. 2016;8:E895.
- El Saghir NS, Charara RN, Kreidieh FY, et al. Global practice and efficiency of multidisciplinary tumor boards: results of an American Society of Clinical Oncology international survey. J Glob Oncol. 2015;1:57-64.
- Mori S, Navarrete-Dechent C, Petukhova TA, et al. Tumor board conferences for multidisciplinary skin cancer management: a survey of US cancer centers. J Natl Compr Canc Netw. 2018;16:1209-1215.
- Dummer R, Ascierto PA, Basset-Seguin N, et al. Sonidegib and vismodegib in the treatment of patients with locally advanced basal cell carcinoma: a joint expert opinion. J Eur Acad Dermatol Venereol. 2020;34:1944-1956.
- Kehl KL, Landrum MB, Kahn KL, et al. Tumor board participation among physicians caring for patients with lung or colorectal cancer. J Oncol Pract. 2015;11:E267-E278.
Multidisciplinary tumor boards are composed of providers from many fields who deliver coordinated care for patients with unresectable and high-risk skin cancers. Providers who comprise the tumor board often are radiation oncologists, hematologists/oncologists, general surgeons, dermatologists, dermatologic surgeons, and pathologists. The benefit of having a tumor board is that each patient is evaluated simultaneously by a group of physicians from various specialties who bring diverse perspectives that will contribute to the overall treatment plan. The cases often encompass high-risk tumors including unresectable basal cell carcinomas or invasive melanomas. By combining knowledge from each specialty in a team approach, the tumor board can effectively and holistically develop a care plan for each patient.
For the tumor board at the Warren Alpert Medical School of Brown University (Providence, Rhode Island), we often prepare a presentation with comprehensive details about the patient and tumor. During the presentation, we also propose a treatment plan prior to describing each patient at the weekly conference and amend the plans during the discussion. Tumor boards also provide a consulting role to the community and hospital providers in which patients are being referred by their primary provider and are seeking a second opinion or guidance.
In many ways, the tumor board is a multidisciplinary approach for patient advocacy in the form of treatment. These physicians meet on a regular basis to check on the patient’s progress and continually reevaluate how to have discussions about the patient’s care. There are many reasons why it is important to refer patients to a multidisciplinary tumor board.
Improved Workup and Diagnosis
One of the values of a tumor board is that it allows for patient data to be collected and assembled in a way that tells a story. The specialist from each field can then discuss and weigh the benefits and risks for each diagnostic test that should be performed for the workup in each patient. Physicians who refer their patients to the tumor board use their recommendations to both confirm the diagnosis and shift their treatment plans, depending on the information presented during the meeting.1 There may be a change in the tumor type, decision to refer for surgery, cancer staging, and list of viable options, especially after reviewing pathology and imaging.2 The discussion of the treatment plan may consider not only surgical considerations but also the patient’s quality of life. At times, noninvasive interventions are more appropriate and align with the patient’s goals of care. In addition, during the tumor board clinic there may be new tumors that are identified and biopsied, providing increased diagnosis and surveillance for patients who may have a higher risk for developing skin cancer.
Education for Residents and Providers
The multidisciplinary tumor board not only helps patients but also educates both residents and providers on the evidence-based therapeutic management of high-risk tumors.2 Research literature on cutaneous oncology is dynamic, and the weekly tumor board meetings help providers stay informed about the best and most effective treatments for their patients.3 In addition to the attending specialists, participants of the tumor board also may include residents, medical students, medical assistance staff, nurses, physician assistants, and fellows. Furthermore, the recommendations given by the tumor board serve to educate both the patient and the provider who referred them to the tumor board. Although we have access to excellent dermatology textbooks as residents, the most impactful educational experience is seeing the patients in tumor board clinic and participating in the immensely educational discussions at the weekly conferences. Through this experience, I have learned that treatment plans should be personalized to the patient. There are many factors to take into consideration when deciphering what the best course of treatment will be for a patient. Sometimes the best option is Mohs micrographic surgery, while other times it may be scheduling several sessions of palliative radiation oncology. Treatment depends on the individual patient and their condition.
Coordination of Care
During a week that I was on call, I was consulted to biopsy a patient with a giant hemorrhagic basal cell carcinoma that caused substantial cheek and nose distortion as well as anemia secondary to acute blood loss. The patient not only did not have a dermatologist but also did not have a primary care physician given he had not had contact with the health care system in more than 30 years. The reason for him not seeking care was multifactorial, but the approach to his care became multidisciplinary. We sought to connect him with the right providers to help him in any way that we could. We presented him at our multidisciplinary tumor board and started him on sonedigib, a medication that binds to and inhibits the smoothened protein.4 Through the tumor board, we were able to establish sustained contact with the patient. The tumor board created effective communication between providers to get him the referrals that he needed for dermatology, pathology, radiation oncology, hematology/oncology, and otolaryngology. The discussions centered around being cognizant of the patient’s apprehension with the health care system as well as providing medical and surgical treatment that would help his quality of life. We built a consensus on what the best plan was for the patient and his family. This consensus would have been more difficult had it not been for the combined specialties of the tumor board. In general, studies have shown that weekly tumor boards have resulted in decreased mortality rates for patients with advanced cancers.5
Final Thoughts
The multidisciplinary tumor board is a powerful resource for hospitals and the greater medical community. At these weekly conferences you realize there may still be hope that begins at the line where your expertise ends. It represents a team of providers who compassionately refuse to give up on patients when they are the last refuge.
Multidisciplinary tumor boards are composed of providers from many fields who deliver coordinated care for patients with unresectable and high-risk skin cancers. Providers who comprise the tumor board often are radiation oncologists, hematologists/oncologists, general surgeons, dermatologists, dermatologic surgeons, and pathologists. The benefit of having a tumor board is that each patient is evaluated simultaneously by a group of physicians from various specialties who bring diverse perspectives that will contribute to the overall treatment plan. The cases often encompass high-risk tumors including unresectable basal cell carcinomas or invasive melanomas. By combining knowledge from each specialty in a team approach, the tumor board can effectively and holistically develop a care plan for each patient.
For the tumor board at the Warren Alpert Medical School of Brown University (Providence, Rhode Island), we often prepare a presentation with comprehensive details about the patient and tumor. During the presentation, we also propose a treatment plan prior to describing each patient at the weekly conference and amend the plans during the discussion. Tumor boards also provide a consulting role to the community and hospital providers in which patients are being referred by their primary provider and are seeking a second opinion or guidance.
In many ways, the tumor board is a multidisciplinary approach for patient advocacy in the form of treatment. These physicians meet on a regular basis to check on the patient’s progress and continually reevaluate how to have discussions about the patient’s care. There are many reasons why it is important to refer patients to a multidisciplinary tumor board.
Improved Workup and Diagnosis
One of the values of a tumor board is that it allows for patient data to be collected and assembled in a way that tells a story. The specialist from each field can then discuss and weigh the benefits and risks for each diagnostic test that should be performed for the workup in each patient. Physicians who refer their patients to the tumor board use their recommendations to both confirm the diagnosis and shift their treatment plans, depending on the information presented during the meeting.1 There may be a change in the tumor type, decision to refer for surgery, cancer staging, and list of viable options, especially after reviewing pathology and imaging.2 The discussion of the treatment plan may consider not only surgical considerations but also the patient’s quality of life. At times, noninvasive interventions are more appropriate and align with the patient’s goals of care. In addition, during the tumor board clinic there may be new tumors that are identified and biopsied, providing increased diagnosis and surveillance for patients who may have a higher risk for developing skin cancer.
Education for Residents and Providers
The multidisciplinary tumor board not only helps patients but also educates both residents and providers on the evidence-based therapeutic management of high-risk tumors.2 Research literature on cutaneous oncology is dynamic, and the weekly tumor board meetings help providers stay informed about the best and most effective treatments for their patients.3 In addition to the attending specialists, participants of the tumor board also may include residents, medical students, medical assistance staff, nurses, physician assistants, and fellows. Furthermore, the recommendations given by the tumor board serve to educate both the patient and the provider who referred them to the tumor board. Although we have access to excellent dermatology textbooks as residents, the most impactful educational experience is seeing the patients in tumor board clinic and participating in the immensely educational discussions at the weekly conferences. Through this experience, I have learned that treatment plans should be personalized to the patient. There are many factors to take into consideration when deciphering what the best course of treatment will be for a patient. Sometimes the best option is Mohs micrographic surgery, while other times it may be scheduling several sessions of palliative radiation oncology. Treatment depends on the individual patient and their condition.
Coordination of Care
During a week that I was on call, I was consulted to biopsy a patient with a giant hemorrhagic basal cell carcinoma that caused substantial cheek and nose distortion as well as anemia secondary to acute blood loss. The patient not only did not have a dermatologist but also did not have a primary care physician given he had not had contact with the health care system in more than 30 years. The reason for him not seeking care was multifactorial, but the approach to his care became multidisciplinary. We sought to connect him with the right providers to help him in any way that we could. We presented him at our multidisciplinary tumor board and started him on sonedigib, a medication that binds to and inhibits the smoothened protein.4 Through the tumor board, we were able to establish sustained contact with the patient. The tumor board created effective communication between providers to get him the referrals that he needed for dermatology, pathology, radiation oncology, hematology/oncology, and otolaryngology. The discussions centered around being cognizant of the patient’s apprehension with the health care system as well as providing medical and surgical treatment that would help his quality of life. We built a consensus on what the best plan was for the patient and his family. This consensus would have been more difficult had it not been for the combined specialties of the tumor board. In general, studies have shown that weekly tumor boards have resulted in decreased mortality rates for patients with advanced cancers.5
Final Thoughts
The multidisciplinary tumor board is a powerful resource for hospitals and the greater medical community. At these weekly conferences you realize there may still be hope that begins at the line where your expertise ends. It represents a team of providers who compassionately refuse to give up on patients when they are the last refuge.
- Foster TJ, Bouchard-Fortier A, Olivotto IA, et al. Effect of multidisciplinary case conferences on physician decision making: breast diagnostic rounds. Cureus. 2016;8:E895.
- El Saghir NS, Charara RN, Kreidieh FY, et al. Global practice and efficiency of multidisciplinary tumor boards: results of an American Society of Clinical Oncology international survey. J Glob Oncol. 2015;1:57-64.
- Mori S, Navarrete-Dechent C, Petukhova TA, et al. Tumor board conferences for multidisciplinary skin cancer management: a survey of US cancer centers. J Natl Compr Canc Netw. 2018;16:1209-1215.
- Dummer R, Ascierto PA, Basset-Seguin N, et al. Sonidegib and vismodegib in the treatment of patients with locally advanced basal cell carcinoma: a joint expert opinion. J Eur Acad Dermatol Venereol. 2020;34:1944-1956.
- Kehl KL, Landrum MB, Kahn KL, et al. Tumor board participation among physicians caring for patients with lung or colorectal cancer. J Oncol Pract. 2015;11:E267-E278.
- Foster TJ, Bouchard-Fortier A, Olivotto IA, et al. Effect of multidisciplinary case conferences on physician decision making: breast diagnostic rounds. Cureus. 2016;8:E895.
- El Saghir NS, Charara RN, Kreidieh FY, et al. Global practice and efficiency of multidisciplinary tumor boards: results of an American Society of Clinical Oncology international survey. J Glob Oncol. 2015;1:57-64.
- Mori S, Navarrete-Dechent C, Petukhova TA, et al. Tumor board conferences for multidisciplinary skin cancer management: a survey of US cancer centers. J Natl Compr Canc Netw. 2018;16:1209-1215.
- Dummer R, Ascierto PA, Basset-Seguin N, et al. Sonidegib and vismodegib in the treatment of patients with locally advanced basal cell carcinoma: a joint expert opinion. J Eur Acad Dermatol Venereol. 2020;34:1944-1956.
- Kehl KL, Landrum MB, Kahn KL, et al. Tumor board participation among physicians caring for patients with lung or colorectal cancer. J Oncol Pract. 2015;11:E267-E278.
Resident Pearl
- Participating in a multidisciplinary tumor board allows residents to learn more about how to manage and treat high-risk skin cancers. The multidisciplinary team approach provides high-quality care for challenging patients.
Benzene found in some sunscreen products, online pharmacy says
Valisure, an online pharmacy known for testing every batch of medication it sells, announced that it has
The company tested 294 batches from 69 companies and found benzene in 27% – many in major national brands like Neutrogena and Banana Boat. Some batches contained as much as three times the emergency FDA limit of 2 parts per million.
Long-term exposure to benzene is known to cause cancer in humans.
“This is especially concerning with sunscreen because multiple FDA studies have shown that sunscreen ingredients absorb through the skin and end up in the blood at high levels,” said David Light, CEO of Valisure.
The FDA is seeking more information about the potential risks from common sunscreen ingredients.
“There is not a safe level of benzene that can exist in sunscreen products,” Christopher Bunick, MD, PhD, associate professor of dermatology at Yale University, New Haven, Conn., said in Valisure’s FDA petition. “The total mass of sunscreen required to cover and protect the human body, in single daily application or repeated applications daily, means that even benzene at 0.1 ppm in a sunscreen could expose people to excessively high nanogram amounts of benzene.”
Valisure’s testing previously led to FDA recalls of heartburn medications and hand sanitizers.
Examining sunscreen’s environmental impact
Chemicals in sunscreen may be harmful to other forms of life, too. For years, scientists have been examining whether certain chemicals in sunscreen could be causing damage to marine life, in particular the world’s coral reefs. Specific ingredients, including oxybenzone, benzophenone-1, benzophenone-8, OD-PABA, 4-methylbenzylidene camphor, 3-benzylidene camphor, nano-titanium dioxide, nano-zinc oxide, octinoxate, and octocrylene, have been identified as potential risks.
Earlier this year, the National Academies of Sciences, Engineering, and Medicine created a committee to review the existing science about the potential environmental hazards. Over the next 2 years, they’ll also consider the public health implications if people stopped using sunscreen.
Valisure’s announcement included this message: “It is important to note that not all sunscreen products contain benzene and that uncontaminated products are available, should continue to be used, and are important for protecting against potentially harmful solar radiation.”
Using sunscreen with SPF 15 every day can lower risk of squamous cell carcinoma by around 40% and melanoma by 50%. The American Academy of Dermatology recommends a broad-spectrum, water-resistant sunscreen with an SPF of 30 or higher.
A version of this article first appeared on WebMD.com.
Valisure, an online pharmacy known for testing every batch of medication it sells, announced that it has
The company tested 294 batches from 69 companies and found benzene in 27% – many in major national brands like Neutrogena and Banana Boat. Some batches contained as much as three times the emergency FDA limit of 2 parts per million.
Long-term exposure to benzene is known to cause cancer in humans.
“This is especially concerning with sunscreen because multiple FDA studies have shown that sunscreen ingredients absorb through the skin and end up in the blood at high levels,” said David Light, CEO of Valisure.
The FDA is seeking more information about the potential risks from common sunscreen ingredients.
“There is not a safe level of benzene that can exist in sunscreen products,” Christopher Bunick, MD, PhD, associate professor of dermatology at Yale University, New Haven, Conn., said in Valisure’s FDA petition. “The total mass of sunscreen required to cover and protect the human body, in single daily application or repeated applications daily, means that even benzene at 0.1 ppm in a sunscreen could expose people to excessively high nanogram amounts of benzene.”
Valisure’s testing previously led to FDA recalls of heartburn medications and hand sanitizers.
Examining sunscreen’s environmental impact
Chemicals in sunscreen may be harmful to other forms of life, too. For years, scientists have been examining whether certain chemicals in sunscreen could be causing damage to marine life, in particular the world’s coral reefs. Specific ingredients, including oxybenzone, benzophenone-1, benzophenone-8, OD-PABA, 4-methylbenzylidene camphor, 3-benzylidene camphor, nano-titanium dioxide, nano-zinc oxide, octinoxate, and octocrylene, have been identified as potential risks.
Earlier this year, the National Academies of Sciences, Engineering, and Medicine created a committee to review the existing science about the potential environmental hazards. Over the next 2 years, they’ll also consider the public health implications if people stopped using sunscreen.
Valisure’s announcement included this message: “It is important to note that not all sunscreen products contain benzene and that uncontaminated products are available, should continue to be used, and are important for protecting against potentially harmful solar radiation.”
Using sunscreen with SPF 15 every day can lower risk of squamous cell carcinoma by around 40% and melanoma by 50%. The American Academy of Dermatology recommends a broad-spectrum, water-resistant sunscreen with an SPF of 30 or higher.
A version of this article first appeared on WebMD.com.
Valisure, an online pharmacy known for testing every batch of medication it sells, announced that it has
The company tested 294 batches from 69 companies and found benzene in 27% – many in major national brands like Neutrogena and Banana Boat. Some batches contained as much as three times the emergency FDA limit of 2 parts per million.
Long-term exposure to benzene is known to cause cancer in humans.
“This is especially concerning with sunscreen because multiple FDA studies have shown that sunscreen ingredients absorb through the skin and end up in the blood at high levels,” said David Light, CEO of Valisure.
The FDA is seeking more information about the potential risks from common sunscreen ingredients.
“There is not a safe level of benzene that can exist in sunscreen products,” Christopher Bunick, MD, PhD, associate professor of dermatology at Yale University, New Haven, Conn., said in Valisure’s FDA petition. “The total mass of sunscreen required to cover and protect the human body, in single daily application or repeated applications daily, means that even benzene at 0.1 ppm in a sunscreen could expose people to excessively high nanogram amounts of benzene.”
Valisure’s testing previously led to FDA recalls of heartburn medications and hand sanitizers.
Examining sunscreen’s environmental impact
Chemicals in sunscreen may be harmful to other forms of life, too. For years, scientists have been examining whether certain chemicals in sunscreen could be causing damage to marine life, in particular the world’s coral reefs. Specific ingredients, including oxybenzone, benzophenone-1, benzophenone-8, OD-PABA, 4-methylbenzylidene camphor, 3-benzylidene camphor, nano-titanium dioxide, nano-zinc oxide, octinoxate, and octocrylene, have been identified as potential risks.
Earlier this year, the National Academies of Sciences, Engineering, and Medicine created a committee to review the existing science about the potential environmental hazards. Over the next 2 years, they’ll also consider the public health implications if people stopped using sunscreen.
Valisure’s announcement included this message: “It is important to note that not all sunscreen products contain benzene and that uncontaminated products are available, should continue to be used, and are important for protecting against potentially harmful solar radiation.”
Using sunscreen with SPF 15 every day can lower risk of squamous cell carcinoma by around 40% and melanoma by 50%. The American Academy of Dermatology recommends a broad-spectrum, water-resistant sunscreen with an SPF of 30 or higher.
A version of this article first appeared on WebMD.com.
Survey: Many Mohs surgeons are struggling on the job
.
In a measurement of well-being, 40% of members of the American College of Mohs
Surgery (ACMS) who responded to the survey – and 52% of women – scored at a level considered “at-risk” for adverse outcomes, such as poor quality of life.
“I didn’t think the numbers were going to be that high,” said study author Kemi O. Awe, MD, PhD, a dermatology resident at the University of Alabama at Birmingham, especially in light of Mohs surgery’s reputation as being an especially desirable field in dermatology. She presented the findings at the annual meeting of the ACMS.
Dr. Awe, who hopes to become a Mohs surgeon herself, said in an interview that she launched the study in part to understand how colleagues are faring. “Dermatology is known as a specialty that has a good lifestyle and less stress, but the rate of burnout is actually going up.”
For the study, Dr. Awe and colleagues sent a survey to ACMS members between October and December 2020. The 91 respondents had an average age of 46, and 58% were male. Most practiced in academic facilities (56%), while the rest worked in private practice (39%) or multispecialty (4%) practices. Almost all (89%) were married or in partnerships.
The survey calculated scores on the expanded Physician Well Being Index, a validated tool for measuring physician distress. Forty percent of 68 respondents to this part of the survey got a score of 3 or higher, which the study describes as “a threshold for respondents who are ‘at-risk’ of adverse outcomes such as poor quality of life, depression, and a high level of fatigue.”
Women were more likely to be considered at risk (52%) than men (28%). “This isn’t different than what’s already out there: Female physicians are more likely to be burned out compared to men,” Dr. Awe said.
Compared with their male counterparts, female Mohs surgeons were more likely to say that time at work, malpractice concerns, insurance reimbursement, and compensation structure negatively affected their well-being (P ≤ .05).
It’s unclear whether there’s a well-being gender gap among dermatologists overall, however. Dr. Awe highlighted a 2019 survey of 108 dermatologists that found no significant difference in overall burnout between men and women – about 42% of both genders reported symptoms. But the survey did find that “dermatologists with children living at home had significantly higher levels of burnout,” with a P value of .03.
Dr. Awe said the findings offer insight into what to look out for when pursuing a career as a Mohs surgeon. “There’s potentially excess stress about being a Mohs surgeon,” she said, although the field also has a reputation as being fulfilling and rewarding.
In an interview, Stanford (Calif.) University dermatologist Zakia Rahman, MD, praised the study and said it “certainly provides a framework to address professional fulfillment amongst Mohs surgeons.”
It was especially surprising, she said, that female surgeons didn’t rate their compensation structure as positively as did their male colleagues. “It is possible that there is still a significant amount of gender-based difference in compensation between male and female Mohs surgeons. This is an area that can be further explored.”
Moving forward, she said, “our professional dermatology societies must examine the increase in burnout within our specialty. Further funding and research in this area is needed.”
For now, dermatologists can focus on strategies that can reduce burnout in the field, Sailesh Konda, MD, a Mohs surgeon at the Univeristy of Florida, Gainesville, said in an interview. Dr. Konda highlighted a report published in 2020 that, he said, "recommended focusing on incremental changes that help restore autonomy and control over work, connecting with colleagues within dermatology and the broader medical community, developing self-awareness and recognition of a perfectionist mindset, and restoring meaning and joy to patient care.”*
No funding is reported for the study. Dr. Awe, Dr. Rahman, and Dr. Konda have no relevant disclosures.
*This story was updated on June 2 for clarity.
.
In a measurement of well-being, 40% of members of the American College of Mohs
Surgery (ACMS) who responded to the survey – and 52% of women – scored at a level considered “at-risk” for adverse outcomes, such as poor quality of life.
“I didn’t think the numbers were going to be that high,” said study author Kemi O. Awe, MD, PhD, a dermatology resident at the University of Alabama at Birmingham, especially in light of Mohs surgery’s reputation as being an especially desirable field in dermatology. She presented the findings at the annual meeting of the ACMS.
Dr. Awe, who hopes to become a Mohs surgeon herself, said in an interview that she launched the study in part to understand how colleagues are faring. “Dermatology is known as a specialty that has a good lifestyle and less stress, but the rate of burnout is actually going up.”
For the study, Dr. Awe and colleagues sent a survey to ACMS members between October and December 2020. The 91 respondents had an average age of 46, and 58% were male. Most practiced in academic facilities (56%), while the rest worked in private practice (39%) or multispecialty (4%) practices. Almost all (89%) were married or in partnerships.
The survey calculated scores on the expanded Physician Well Being Index, a validated tool for measuring physician distress. Forty percent of 68 respondents to this part of the survey got a score of 3 or higher, which the study describes as “a threshold for respondents who are ‘at-risk’ of adverse outcomes such as poor quality of life, depression, and a high level of fatigue.”
Women were more likely to be considered at risk (52%) than men (28%). “This isn’t different than what’s already out there: Female physicians are more likely to be burned out compared to men,” Dr. Awe said.
Compared with their male counterparts, female Mohs surgeons were more likely to say that time at work, malpractice concerns, insurance reimbursement, and compensation structure negatively affected their well-being (P ≤ .05).
It’s unclear whether there’s a well-being gender gap among dermatologists overall, however. Dr. Awe highlighted a 2019 survey of 108 dermatologists that found no significant difference in overall burnout between men and women – about 42% of both genders reported symptoms. But the survey did find that “dermatologists with children living at home had significantly higher levels of burnout,” with a P value of .03.
Dr. Awe said the findings offer insight into what to look out for when pursuing a career as a Mohs surgeon. “There’s potentially excess stress about being a Mohs surgeon,” she said, although the field also has a reputation as being fulfilling and rewarding.
In an interview, Stanford (Calif.) University dermatologist Zakia Rahman, MD, praised the study and said it “certainly provides a framework to address professional fulfillment amongst Mohs surgeons.”
It was especially surprising, she said, that female surgeons didn’t rate their compensation structure as positively as did their male colleagues. “It is possible that there is still a significant amount of gender-based difference in compensation between male and female Mohs surgeons. This is an area that can be further explored.”
Moving forward, she said, “our professional dermatology societies must examine the increase in burnout within our specialty. Further funding and research in this area is needed.”
For now, dermatologists can focus on strategies that can reduce burnout in the field, Sailesh Konda, MD, a Mohs surgeon at the Univeristy of Florida, Gainesville, said in an interview. Dr. Konda highlighted a report published in 2020 that, he said, "recommended focusing on incremental changes that help restore autonomy and control over work, connecting with colleagues within dermatology and the broader medical community, developing self-awareness and recognition of a perfectionist mindset, and restoring meaning and joy to patient care.”*
No funding is reported for the study. Dr. Awe, Dr. Rahman, and Dr. Konda have no relevant disclosures.
*This story was updated on June 2 for clarity.
.
In a measurement of well-being, 40% of members of the American College of Mohs
Surgery (ACMS) who responded to the survey – and 52% of women – scored at a level considered “at-risk” for adverse outcomes, such as poor quality of life.
“I didn’t think the numbers were going to be that high,” said study author Kemi O. Awe, MD, PhD, a dermatology resident at the University of Alabama at Birmingham, especially in light of Mohs surgery’s reputation as being an especially desirable field in dermatology. She presented the findings at the annual meeting of the ACMS.
Dr. Awe, who hopes to become a Mohs surgeon herself, said in an interview that she launched the study in part to understand how colleagues are faring. “Dermatology is known as a specialty that has a good lifestyle and less stress, but the rate of burnout is actually going up.”
For the study, Dr. Awe and colleagues sent a survey to ACMS members between October and December 2020. The 91 respondents had an average age of 46, and 58% were male. Most practiced in academic facilities (56%), while the rest worked in private practice (39%) or multispecialty (4%) practices. Almost all (89%) were married or in partnerships.
The survey calculated scores on the expanded Physician Well Being Index, a validated tool for measuring physician distress. Forty percent of 68 respondents to this part of the survey got a score of 3 or higher, which the study describes as “a threshold for respondents who are ‘at-risk’ of adverse outcomes such as poor quality of life, depression, and a high level of fatigue.”
Women were more likely to be considered at risk (52%) than men (28%). “This isn’t different than what’s already out there: Female physicians are more likely to be burned out compared to men,” Dr. Awe said.
Compared with their male counterparts, female Mohs surgeons were more likely to say that time at work, malpractice concerns, insurance reimbursement, and compensation structure negatively affected their well-being (P ≤ .05).
It’s unclear whether there’s a well-being gender gap among dermatologists overall, however. Dr. Awe highlighted a 2019 survey of 108 dermatologists that found no significant difference in overall burnout between men and women – about 42% of both genders reported symptoms. But the survey did find that “dermatologists with children living at home had significantly higher levels of burnout,” with a P value of .03.
Dr. Awe said the findings offer insight into what to look out for when pursuing a career as a Mohs surgeon. “There’s potentially excess stress about being a Mohs surgeon,” she said, although the field also has a reputation as being fulfilling and rewarding.
In an interview, Stanford (Calif.) University dermatologist Zakia Rahman, MD, praised the study and said it “certainly provides a framework to address professional fulfillment amongst Mohs surgeons.”
It was especially surprising, she said, that female surgeons didn’t rate their compensation structure as positively as did their male colleagues. “It is possible that there is still a significant amount of gender-based difference in compensation between male and female Mohs surgeons. This is an area that can be further explored.”
Moving forward, she said, “our professional dermatology societies must examine the increase in burnout within our specialty. Further funding and research in this area is needed.”
For now, dermatologists can focus on strategies that can reduce burnout in the field, Sailesh Konda, MD, a Mohs surgeon at the Univeristy of Florida, Gainesville, said in an interview. Dr. Konda highlighted a report published in 2020 that, he said, "recommended focusing on incremental changes that help restore autonomy and control over work, connecting with colleagues within dermatology and the broader medical community, developing self-awareness and recognition of a perfectionist mindset, and restoring meaning and joy to patient care.”*
No funding is reported for the study. Dr. Awe, Dr. Rahman, and Dr. Konda have no relevant disclosures.
*This story was updated on June 2 for clarity.
FROM THE ACMS ANNUAL MEETING
Novel immunotherapy relatlimab in advanced melanoma
Adding the novel immune checkpoint inhibitor relatlimab to the more established nivolumab (Opdivo) significantly extended the progression-free survival (PFS) of patients with previously untreated advanced melanoma in comparison with nivolumab alone in the phase 3 RELATIVITY-047 trial.
Both drugs are from Bristol-Myers Squibb, which funded the study.
“Our findings demonstrate that relatlimab plus nivolumab is a potential novel treatment option for this patient population,” said lead researcher Evan J. Lipson, MD, Sidney Kimmel Comprehensive Cancer Center, Johns Hopkins University, Baltimore.
Relatlimab has a different mechanism of action from currently available immune checkpoint inhibitors, such as nivolumab and similar agents, which act as inhibitors of the programmed cell death protein–1 (PD-1) or programmed cell death–ligand-1 (PD-L1). In contrast, relatlimab acts as an antibody that targets lymphocyte-activation gene 3 (LAG-3), which inhibits T cells and thus helps cancer cells evade immune attack.
“This is the first phase 3 study to validate inhibition of the LAG-3 immune checkpoint as a therapeutic strategy for patients with cancer, and it establishes the LAG-3 pathway as the third immune checkpoint pathway in history, after CLTA-4 and PD-1, for which blockade appears to have clinical benefit,” Dr. Lipson said at a press briefing ahead of the annual meeting of the American Society of Clinical Oncology (ASCO), where this study will be presented (abstract 9503).
Commenting for ASCO, Julie R. Gralow, MD, chief medical officer and executive vice president, agreed that “these results provide validation of the LAG-3 immune checkpoint as a therapeutic target ... and they also support combination treatment with immunotherapies that act on different parts of the immune system.”
When Dr. Lipson was asked whether he would recommend the combination of relatlimab plus nivolumab as a first-line treatment for this patient population, he said that “for many patients,” the first-line treatment choice is made on a “case-by-case” basis.
“We are fortunate in melanoma that we have an ever-expanding list of seemingly effective options, and I think we’ll find at some point this will be added to that list,” he said. “Whether this is the first-line choice for any given patient really depends on a lot of factors,” he added.
Dr. Gralow added a note of caution. “The combination was clearly more toxic, and so I think there will be a lot of discussion” as to when it would be used and for which patients, she said.
In the absence of head-to-head comparisons, “I’m not sure that we have one answer” as to which treatment to choose, she added. With the ever-increasing number of options available in melanoma, the individual treatment choice is “getting more complicated,” she said.
Study details
The global RELATIVITY-047 study was conducted in 714 patients with previously untreated unresectable or metastatic melanoma. The participants were randomly assigned to receive either relatlimab plus nivolumab or nivolumab alone.
Dr. Lipson explained that the treatments were given as a fixed-dosed combination, meaning the preparation of relatlimab and nivolumab was given in the “same medication phial and administered as a single intravenous infusion in order to reduce preparation and infusion times and minimize the risk of administration errors.”
PFS, as determined on blinded independent central review, was significantly longer with the combination therapy than with nivolumab alone, at a median of 10.12 months vs. 4.63 months (hazard ratio, 0.75; P = .0055).
At 12 months, the PFS rate among patients given relatlimab plus nivolumab was 47.7%, versus 36.0% among those given nivolumab alone.
“This significant improvement meant that the study met its primary endpoint,” Dr. Lipson said, adding that the PFS benefit “appeared relatively early in the course of therapy.” The curves separated at 12 weeks, and benefit was “sustained” over the course of follow-up.
He added that the performance of nivolumab alone was “in the range” of that seen in previous studies, although he underlined that cross-trial comparison is difficult, given the differences in study design.
“In general, treatment-related adverse events” associated with the combination therapy were “manageable and reflected the safety profile that we typically see with immune checkpoint inhibitors,” he noted.
The results showed that 40.3% of patients who received the combination therapy experienced a grade 3-4 adverse event, compared with 33.4% of those given nivolumab alone. Grade 3-4 treatment-related adverse events leading to discontinuation occurred in 8.5% and 3.1% of patients, respectively.
Three treatment-related deaths occurred in the relatlimab and nivolumab arm. Two such deaths occurred in the nivolumab-alone group.
The study was funded by Bristol Myers Squibb. Dr. Lipson has relationships with Array BioPharma, Bristol Myers Squibb, EMD Serono, Genentech, Macrogenics, Merck, Millennium, Novartis, Sanofi/Regeneron, and Sysmex (inst). Dr. Gralow has relationships with AstraZeneca, Genentech, Sandoz, and Immunomedics.
A version of this article first appeared on Medscape.com.
Adding the novel immune checkpoint inhibitor relatlimab to the more established nivolumab (Opdivo) significantly extended the progression-free survival (PFS) of patients with previously untreated advanced melanoma in comparison with nivolumab alone in the phase 3 RELATIVITY-047 trial.
Both drugs are from Bristol-Myers Squibb, which funded the study.
“Our findings demonstrate that relatlimab plus nivolumab is a potential novel treatment option for this patient population,” said lead researcher Evan J. Lipson, MD, Sidney Kimmel Comprehensive Cancer Center, Johns Hopkins University, Baltimore.
Relatlimab has a different mechanism of action from currently available immune checkpoint inhibitors, such as nivolumab and similar agents, which act as inhibitors of the programmed cell death protein–1 (PD-1) or programmed cell death–ligand-1 (PD-L1). In contrast, relatlimab acts as an antibody that targets lymphocyte-activation gene 3 (LAG-3), which inhibits T cells and thus helps cancer cells evade immune attack.
“This is the first phase 3 study to validate inhibition of the LAG-3 immune checkpoint as a therapeutic strategy for patients with cancer, and it establishes the LAG-3 pathway as the third immune checkpoint pathway in history, after CLTA-4 and PD-1, for which blockade appears to have clinical benefit,” Dr. Lipson said at a press briefing ahead of the annual meeting of the American Society of Clinical Oncology (ASCO), where this study will be presented (abstract 9503).
Commenting for ASCO, Julie R. Gralow, MD, chief medical officer and executive vice president, agreed that “these results provide validation of the LAG-3 immune checkpoint as a therapeutic target ... and they also support combination treatment with immunotherapies that act on different parts of the immune system.”
When Dr. Lipson was asked whether he would recommend the combination of relatlimab plus nivolumab as a first-line treatment for this patient population, he said that “for many patients,” the first-line treatment choice is made on a “case-by-case” basis.
“We are fortunate in melanoma that we have an ever-expanding list of seemingly effective options, and I think we’ll find at some point this will be added to that list,” he said. “Whether this is the first-line choice for any given patient really depends on a lot of factors,” he added.
Dr. Gralow added a note of caution. “The combination was clearly more toxic, and so I think there will be a lot of discussion” as to when it would be used and for which patients, she said.
In the absence of head-to-head comparisons, “I’m not sure that we have one answer” as to which treatment to choose, she added. With the ever-increasing number of options available in melanoma, the individual treatment choice is “getting more complicated,” she said.
Study details
The global RELATIVITY-047 study was conducted in 714 patients with previously untreated unresectable or metastatic melanoma. The participants were randomly assigned to receive either relatlimab plus nivolumab or nivolumab alone.
Dr. Lipson explained that the treatments were given as a fixed-dosed combination, meaning the preparation of relatlimab and nivolumab was given in the “same medication phial and administered as a single intravenous infusion in order to reduce preparation and infusion times and minimize the risk of administration errors.”
PFS, as determined on blinded independent central review, was significantly longer with the combination therapy than with nivolumab alone, at a median of 10.12 months vs. 4.63 months (hazard ratio, 0.75; P = .0055).
At 12 months, the PFS rate among patients given relatlimab plus nivolumab was 47.7%, versus 36.0% among those given nivolumab alone.
“This significant improvement meant that the study met its primary endpoint,” Dr. Lipson said, adding that the PFS benefit “appeared relatively early in the course of therapy.” The curves separated at 12 weeks, and benefit was “sustained” over the course of follow-up.
He added that the performance of nivolumab alone was “in the range” of that seen in previous studies, although he underlined that cross-trial comparison is difficult, given the differences in study design.
“In general, treatment-related adverse events” associated with the combination therapy were “manageable and reflected the safety profile that we typically see with immune checkpoint inhibitors,” he noted.
The results showed that 40.3% of patients who received the combination therapy experienced a grade 3-4 adverse event, compared with 33.4% of those given nivolumab alone. Grade 3-4 treatment-related adverse events leading to discontinuation occurred in 8.5% and 3.1% of patients, respectively.
Three treatment-related deaths occurred in the relatlimab and nivolumab arm. Two such deaths occurred in the nivolumab-alone group.
The study was funded by Bristol Myers Squibb. Dr. Lipson has relationships with Array BioPharma, Bristol Myers Squibb, EMD Serono, Genentech, Macrogenics, Merck, Millennium, Novartis, Sanofi/Regeneron, and Sysmex (inst). Dr. Gralow has relationships with AstraZeneca, Genentech, Sandoz, and Immunomedics.
A version of this article first appeared on Medscape.com.
Adding the novel immune checkpoint inhibitor relatlimab to the more established nivolumab (Opdivo) significantly extended the progression-free survival (PFS) of patients with previously untreated advanced melanoma in comparison with nivolumab alone in the phase 3 RELATIVITY-047 trial.
Both drugs are from Bristol-Myers Squibb, which funded the study.
“Our findings demonstrate that relatlimab plus nivolumab is a potential novel treatment option for this patient population,” said lead researcher Evan J. Lipson, MD, Sidney Kimmel Comprehensive Cancer Center, Johns Hopkins University, Baltimore.
Relatlimab has a different mechanism of action from currently available immune checkpoint inhibitors, such as nivolumab and similar agents, which act as inhibitors of the programmed cell death protein–1 (PD-1) or programmed cell death–ligand-1 (PD-L1). In contrast, relatlimab acts as an antibody that targets lymphocyte-activation gene 3 (LAG-3), which inhibits T cells and thus helps cancer cells evade immune attack.
“This is the first phase 3 study to validate inhibition of the LAG-3 immune checkpoint as a therapeutic strategy for patients with cancer, and it establishes the LAG-3 pathway as the third immune checkpoint pathway in history, after CLTA-4 and PD-1, for which blockade appears to have clinical benefit,” Dr. Lipson said at a press briefing ahead of the annual meeting of the American Society of Clinical Oncology (ASCO), where this study will be presented (abstract 9503).
Commenting for ASCO, Julie R. Gralow, MD, chief medical officer and executive vice president, agreed that “these results provide validation of the LAG-3 immune checkpoint as a therapeutic target ... and they also support combination treatment with immunotherapies that act on different parts of the immune system.”
When Dr. Lipson was asked whether he would recommend the combination of relatlimab plus nivolumab as a first-line treatment for this patient population, he said that “for many patients,” the first-line treatment choice is made on a “case-by-case” basis.
“We are fortunate in melanoma that we have an ever-expanding list of seemingly effective options, and I think we’ll find at some point this will be added to that list,” he said. “Whether this is the first-line choice for any given patient really depends on a lot of factors,” he added.
Dr. Gralow added a note of caution. “The combination was clearly more toxic, and so I think there will be a lot of discussion” as to when it would be used and for which patients, she said.
In the absence of head-to-head comparisons, “I’m not sure that we have one answer” as to which treatment to choose, she added. With the ever-increasing number of options available in melanoma, the individual treatment choice is “getting more complicated,” she said.
Study details
The global RELATIVITY-047 study was conducted in 714 patients with previously untreated unresectable or metastatic melanoma. The participants were randomly assigned to receive either relatlimab plus nivolumab or nivolumab alone.
Dr. Lipson explained that the treatments were given as a fixed-dosed combination, meaning the preparation of relatlimab and nivolumab was given in the “same medication phial and administered as a single intravenous infusion in order to reduce preparation and infusion times and minimize the risk of administration errors.”
PFS, as determined on blinded independent central review, was significantly longer with the combination therapy than with nivolumab alone, at a median of 10.12 months vs. 4.63 months (hazard ratio, 0.75; P = .0055).
At 12 months, the PFS rate among patients given relatlimab plus nivolumab was 47.7%, versus 36.0% among those given nivolumab alone.
“This significant improvement meant that the study met its primary endpoint,” Dr. Lipson said, adding that the PFS benefit “appeared relatively early in the course of therapy.” The curves separated at 12 weeks, and benefit was “sustained” over the course of follow-up.
He added that the performance of nivolumab alone was “in the range” of that seen in previous studies, although he underlined that cross-trial comparison is difficult, given the differences in study design.
“In general, treatment-related adverse events” associated with the combination therapy were “manageable and reflected the safety profile that we typically see with immune checkpoint inhibitors,” he noted.
The results showed that 40.3% of patients who received the combination therapy experienced a grade 3-4 adverse event, compared with 33.4% of those given nivolumab alone. Grade 3-4 treatment-related adverse events leading to discontinuation occurred in 8.5% and 3.1% of patients, respectively.
Three treatment-related deaths occurred in the relatlimab and nivolumab arm. Two such deaths occurred in the nivolumab-alone group.
The study was funded by Bristol Myers Squibb. Dr. Lipson has relationships with Array BioPharma, Bristol Myers Squibb, EMD Serono, Genentech, Macrogenics, Merck, Millennium, Novartis, Sanofi/Regeneron, and Sysmex (inst). Dr. Gralow has relationships with AstraZeneca, Genentech, Sandoz, and Immunomedics.
A version of this article first appeared on Medscape.com.
Telemedicine is popular among Mohs surgeons – for now
A majority of
A variety of factors combine to make it “very difficult for surgeons to make long-term plans for implementing telemedicine in their practices,” said Mario Maruthur, MD, who presented the findings at the annual meeting of the American College of Mohs Surgery. “Telemedicine likely has a role in Mohs practices, particularly with postop follow-up visits. However, postpandemic reimbursement and regulatory issues need to be formally laid out before Mohs surgeons are able to incorporate it into their permanent work flow.”
Dr. Maruthur, a Mohs surgery and dermatologic oncology fellow at Memorial Sloan Kettering Cancer Center, New York, and colleagues sent a survey to ACMS members in September and October 2020. “We saw first-hand in our surgical practice that telemedicine quickly became an important tool when the pandemic surged in the spring of 2020,” he said. Considering that surgical practices are highly dependent on in-person visits, the impetus for this study was to assess to what degree Mohs practices from across the spectrum, including academic and private practices, embraced telemedicine during the pandemic, and “what these surgical practices used telemedicine for, how it was received by their patients, which telemedicine platforms were most often utilized, and lastly, what are their plans if any for incorporating telemedicine into their surgical practices after the pandemic subsides.”
The researchers received responses from 115 surgeons representing all regions of the country (40% Northeast, 21% South, 21% Midwest, and 18% West). Half practiced in urban areas (37%) and large cities (13%), and 40% were in an academic setting versus 36% in a single-specialty private practice.
More than 70% of the respondents said their case load fell by at least 75% during the initial surge of the pandemic; 80% turned to telemedicine, compared with just 23% who relied on the technology prior to the pandemic. The most commonly used telemedicine technologies were FaceTime, Zoom, Doximity, and Epic.
Mohs surgeons reported most commonly using telemedicine for postsurgery management (77% of the total 115 responses). “Telemedicine is a great fit for this category of visits as they allow the surgeon to view the surgical site and answer any questions they patient may have,” Dr. Maruthur said. “If the surgeon does suspect a postop infection or other concern based on a patient’s signs or symptoms, they can easily schedule the patient for an in-person assessment. We suspect that postop follow-up visits may be the best candidate for long-term use of telemedicine in Mohs surgery practices.”
Surgeons also reported using telemedicine for “spot checks” (61%) and surgical consultations (59%).
However, Dr. Maruther noted that preoperative assessments and spot checks can be difficult to perform using telemedicine. “The quality of the video image is not always great, patients can have a difficult time pointing the camera at the right spot and at the right distance. Even appreciating the actual size of the lesion are all difficult over a video encounter. And there is a lot of information gleaned from in-person physical examination, such as whether the lesion is fixed to a deeper structure and whether there are any nearby scars or other suspicious lesions.”
Nearly three-quarters of the surgeons using the technology said most or all patients were receptive to telemedicine.
However, the surgeons reported multiple barriers to the use of telemedicine: Limitations when compared with physical exams (88%), fitting it into the work flow (58%), patient response and training (57%), reimbursement concerns (50%), implementation of the technology (37%), regulations such as HIPAA (24%), training of staff (17%), and licensing (8%).
In an interview, Sumaira Z. Aasi, MD, director of Mohs and dermatologic surgery, Stanford University, agreed that there are many obstacles to routine use of telemedicine by Mohs surgeons. “As surgeons, we rely on the physical and tactile exam to get a sense of the size and extent of the cancer and characteristics such as the laxity of the surrounding tissue whether the tumor is fixed,” she said. “It is very difficult to access this on a telemedicine visit.”
In addition, she said, “many of our patients are in the elderly population, and some may not be comfortable using this technology. Also, it’s not a work flow that we are comfortable or familiar with. And I think that the technology has to improve to allow for better resolution of images as we ‘examine’ patients through a telemedicine visit.”
She added that “another con is there is a reliance on having the patient point out lesions of concern. Many cancers are picked by a careful in-person examination by a qualified physician/dermatologist/Mohs surgeon when the lesion is quite small or subtle and not even noticed by the patient themselves. This approach invariably leads to earlier biopsies and earlier treatments that can prevent morbidity and save health care money.”
On the other hand, she said, telemedicine “may save patients some time and money in terms of the effort and cost of transportation to come in for simpler postoperative medical visits that are often short in their very nature, such as postop check-ups.”
Most of the surgeons surveyed (69%) said telemedicine probably or definitely deserves a place in the practice Mohs surgery, but only 50% said they’d like to or would definitely pursue giving telemedicine a role in their practices once the pandemic is over.
“At the start of the pandemic, many regulations in areas such as HIPAA were eased, and reimbursements were increased, which allowed telemedicine to be quickly adopted,” Dr. Maruther said. “The government and payers have yet to decide which regulations and reimbursements will be in place after the pandemic. That makes it very difficult for surgeons to make long-term plans for implementing telemedicine in their practices.”
Dr. Aasi predicted that telemedicine will become more appealing to patients and physicians as it its technology and usability improves. More familiarity with its use will also be helpful, she said, and surgeons will be more receptive as it’s incorporated into efficient daily work flow.
The study was funded in part by the National Institutes of Health.
A majority of
A variety of factors combine to make it “very difficult for surgeons to make long-term plans for implementing telemedicine in their practices,” said Mario Maruthur, MD, who presented the findings at the annual meeting of the American College of Mohs Surgery. “Telemedicine likely has a role in Mohs practices, particularly with postop follow-up visits. However, postpandemic reimbursement and regulatory issues need to be formally laid out before Mohs surgeons are able to incorporate it into their permanent work flow.”
Dr. Maruthur, a Mohs surgery and dermatologic oncology fellow at Memorial Sloan Kettering Cancer Center, New York, and colleagues sent a survey to ACMS members in September and October 2020. “We saw first-hand in our surgical practice that telemedicine quickly became an important tool when the pandemic surged in the spring of 2020,” he said. Considering that surgical practices are highly dependent on in-person visits, the impetus for this study was to assess to what degree Mohs practices from across the spectrum, including academic and private practices, embraced telemedicine during the pandemic, and “what these surgical practices used telemedicine for, how it was received by their patients, which telemedicine platforms were most often utilized, and lastly, what are their plans if any for incorporating telemedicine into their surgical practices after the pandemic subsides.”
The researchers received responses from 115 surgeons representing all regions of the country (40% Northeast, 21% South, 21% Midwest, and 18% West). Half practiced in urban areas (37%) and large cities (13%), and 40% were in an academic setting versus 36% in a single-specialty private practice.
More than 70% of the respondents said their case load fell by at least 75% during the initial surge of the pandemic; 80% turned to telemedicine, compared with just 23% who relied on the technology prior to the pandemic. The most commonly used telemedicine technologies were FaceTime, Zoom, Doximity, and Epic.
Mohs surgeons reported most commonly using telemedicine for postsurgery management (77% of the total 115 responses). “Telemedicine is a great fit for this category of visits as they allow the surgeon to view the surgical site and answer any questions they patient may have,” Dr. Maruthur said. “If the surgeon does suspect a postop infection or other concern based on a patient’s signs or symptoms, they can easily schedule the patient for an in-person assessment. We suspect that postop follow-up visits may be the best candidate for long-term use of telemedicine in Mohs surgery practices.”
Surgeons also reported using telemedicine for “spot checks” (61%) and surgical consultations (59%).
However, Dr. Maruther noted that preoperative assessments and spot checks can be difficult to perform using telemedicine. “The quality of the video image is not always great, patients can have a difficult time pointing the camera at the right spot and at the right distance. Even appreciating the actual size of the lesion are all difficult over a video encounter. And there is a lot of information gleaned from in-person physical examination, such as whether the lesion is fixed to a deeper structure and whether there are any nearby scars or other suspicious lesions.”
Nearly three-quarters of the surgeons using the technology said most or all patients were receptive to telemedicine.
However, the surgeons reported multiple barriers to the use of telemedicine: Limitations when compared with physical exams (88%), fitting it into the work flow (58%), patient response and training (57%), reimbursement concerns (50%), implementation of the technology (37%), regulations such as HIPAA (24%), training of staff (17%), and licensing (8%).
In an interview, Sumaira Z. Aasi, MD, director of Mohs and dermatologic surgery, Stanford University, agreed that there are many obstacles to routine use of telemedicine by Mohs surgeons. “As surgeons, we rely on the physical and tactile exam to get a sense of the size and extent of the cancer and characteristics such as the laxity of the surrounding tissue whether the tumor is fixed,” she said. “It is very difficult to access this on a telemedicine visit.”
In addition, she said, “many of our patients are in the elderly population, and some may not be comfortable using this technology. Also, it’s not a work flow that we are comfortable or familiar with. And I think that the technology has to improve to allow for better resolution of images as we ‘examine’ patients through a telemedicine visit.”
She added that “another con is there is a reliance on having the patient point out lesions of concern. Many cancers are picked by a careful in-person examination by a qualified physician/dermatologist/Mohs surgeon when the lesion is quite small or subtle and not even noticed by the patient themselves. This approach invariably leads to earlier biopsies and earlier treatments that can prevent morbidity and save health care money.”
On the other hand, she said, telemedicine “may save patients some time and money in terms of the effort and cost of transportation to come in for simpler postoperative medical visits that are often short in their very nature, such as postop check-ups.”
Most of the surgeons surveyed (69%) said telemedicine probably or definitely deserves a place in the practice Mohs surgery, but only 50% said they’d like to or would definitely pursue giving telemedicine a role in their practices once the pandemic is over.
“At the start of the pandemic, many regulations in areas such as HIPAA were eased, and reimbursements were increased, which allowed telemedicine to be quickly adopted,” Dr. Maruther said. “The government and payers have yet to decide which regulations and reimbursements will be in place after the pandemic. That makes it very difficult for surgeons to make long-term plans for implementing telemedicine in their practices.”
Dr. Aasi predicted that telemedicine will become more appealing to patients and physicians as it its technology and usability improves. More familiarity with its use will also be helpful, she said, and surgeons will be more receptive as it’s incorporated into efficient daily work flow.
The study was funded in part by the National Institutes of Health.
A majority of
A variety of factors combine to make it “very difficult for surgeons to make long-term plans for implementing telemedicine in their practices,” said Mario Maruthur, MD, who presented the findings at the annual meeting of the American College of Mohs Surgery. “Telemedicine likely has a role in Mohs practices, particularly with postop follow-up visits. However, postpandemic reimbursement and regulatory issues need to be formally laid out before Mohs surgeons are able to incorporate it into their permanent work flow.”
Dr. Maruthur, a Mohs surgery and dermatologic oncology fellow at Memorial Sloan Kettering Cancer Center, New York, and colleagues sent a survey to ACMS members in September and October 2020. “We saw first-hand in our surgical practice that telemedicine quickly became an important tool when the pandemic surged in the spring of 2020,” he said. Considering that surgical practices are highly dependent on in-person visits, the impetus for this study was to assess to what degree Mohs practices from across the spectrum, including academic and private practices, embraced telemedicine during the pandemic, and “what these surgical practices used telemedicine for, how it was received by their patients, which telemedicine platforms were most often utilized, and lastly, what are their plans if any for incorporating telemedicine into their surgical practices after the pandemic subsides.”
The researchers received responses from 115 surgeons representing all regions of the country (40% Northeast, 21% South, 21% Midwest, and 18% West). Half practiced in urban areas (37%) and large cities (13%), and 40% were in an academic setting versus 36% in a single-specialty private practice.
More than 70% of the respondents said their case load fell by at least 75% during the initial surge of the pandemic; 80% turned to telemedicine, compared with just 23% who relied on the technology prior to the pandemic. The most commonly used telemedicine technologies were FaceTime, Zoom, Doximity, and Epic.
Mohs surgeons reported most commonly using telemedicine for postsurgery management (77% of the total 115 responses). “Telemedicine is a great fit for this category of visits as they allow the surgeon to view the surgical site and answer any questions they patient may have,” Dr. Maruthur said. “If the surgeon does suspect a postop infection or other concern based on a patient’s signs or symptoms, they can easily schedule the patient for an in-person assessment. We suspect that postop follow-up visits may be the best candidate for long-term use of telemedicine in Mohs surgery practices.”
Surgeons also reported using telemedicine for “spot checks” (61%) and surgical consultations (59%).
However, Dr. Maruther noted that preoperative assessments and spot checks can be difficult to perform using telemedicine. “The quality of the video image is not always great, patients can have a difficult time pointing the camera at the right spot and at the right distance. Even appreciating the actual size of the lesion are all difficult over a video encounter. And there is a lot of information gleaned from in-person physical examination, such as whether the lesion is fixed to a deeper structure and whether there are any nearby scars or other suspicious lesions.”
Nearly three-quarters of the surgeons using the technology said most or all patients were receptive to telemedicine.
However, the surgeons reported multiple barriers to the use of telemedicine: Limitations when compared with physical exams (88%), fitting it into the work flow (58%), patient response and training (57%), reimbursement concerns (50%), implementation of the technology (37%), regulations such as HIPAA (24%), training of staff (17%), and licensing (8%).
In an interview, Sumaira Z. Aasi, MD, director of Mohs and dermatologic surgery, Stanford University, agreed that there are many obstacles to routine use of telemedicine by Mohs surgeons. “As surgeons, we rely on the physical and tactile exam to get a sense of the size and extent of the cancer and characteristics such as the laxity of the surrounding tissue whether the tumor is fixed,” she said. “It is very difficult to access this on a telemedicine visit.”
In addition, she said, “many of our patients are in the elderly population, and some may not be comfortable using this technology. Also, it’s not a work flow that we are comfortable or familiar with. And I think that the technology has to improve to allow for better resolution of images as we ‘examine’ patients through a telemedicine visit.”
She added that “another con is there is a reliance on having the patient point out lesions of concern. Many cancers are picked by a careful in-person examination by a qualified physician/dermatologist/Mohs surgeon when the lesion is quite small or subtle and not even noticed by the patient themselves. This approach invariably leads to earlier biopsies and earlier treatments that can prevent morbidity and save health care money.”
On the other hand, she said, telemedicine “may save patients some time and money in terms of the effort and cost of transportation to come in for simpler postoperative medical visits that are often short in their very nature, such as postop check-ups.”
Most of the surgeons surveyed (69%) said telemedicine probably or definitely deserves a place in the practice Mohs surgery, but only 50% said they’d like to or would definitely pursue giving telemedicine a role in their practices once the pandemic is over.
“At the start of the pandemic, many regulations in areas such as HIPAA were eased, and reimbursements were increased, which allowed telemedicine to be quickly adopted,” Dr. Maruther said. “The government and payers have yet to decide which regulations and reimbursements will be in place after the pandemic. That makes it very difficult for surgeons to make long-term plans for implementing telemedicine in their practices.”
Dr. Aasi predicted that telemedicine will become more appealing to patients and physicians as it its technology and usability improves. More familiarity with its use will also be helpful, she said, and surgeons will be more receptive as it’s incorporated into efficient daily work flow.
The study was funded in part by the National Institutes of Health.
FROM THE ACMS ANNUAL MEETING
New guideline provides recommendations on reconstruction after skin cancer resection
You’ve successfully resected a skin cancer lesion, leaving clear margins. Now what?
That’s
The guideline – a joint effort of the American Society of Plastic Surgeons, American Society for Dermatologic Surgery, American Academy of Dermatology, American Academy of Facial Plastic and Reconstructive Surgery, American Academy of Otolaryngology – Head and Neck Surgery Foundation, American College of Mohs Surgery, American Society for Mohs Surgery, and American Society of Ophthalmic Plastic and Reconstructive Surgery – was published online in the Journal of the American Academy of Dermatology.
From the outset, the panel members realized that to keep the guideline manageable they had to limit recommendations to the practice of reconstruction defined as “cutaneous closure that requires a flap, graft, or tissue rearrangement.”
Other wound closure methods, such as secondary intention healing; simple closures; and complex closures that do not involve flaps, grafts, muscle, or bone, were not covered in the recommendations.
As with similar guidelines, the developers selected seven clinical questions to be addressed, and attempted to find consensus through literature searches, appraisal of the evidence, grading of recommendations, peer review, and public comment.
“We had a very heterogeneous set of things that we were trying to comment on, so we had to keep things somewhat generic,” lead author Andrew Chen, MD, chief of the division of plastic surgery, at the University of Connecticut Health Center, Farmington, said in an interview.
“Skin cancer and reconstruction affect different body areas and areas of different sizes. When we were creating the guidelines, we had to tailor the questions we could ask based on things that would make sense to answer, because obviously we couldn’t ask a question such as: ‘What’s better, a skin graft or a flap?’ Well, there are some things you can’t put a skin graft on – it won’t last, so we couldn’t ask that kind of question,” Dr. Chen said.
Curtis Cetrulo, MD, a plastic and reconstructive surgeon at Massachusetts General Hospital, Boston, who was not involved in the guideline process, said in an interview that the broad recommendations are in keeping with his practice and experience. He also acknowledged, however, the difficulty in creating a guideline that covers the complexity and heterogeneity of reconstructive surgery.
“These are generally good recommendations, but they’re recommendations only, with generally weak levels of evidence. What we really need are clinical trials that can give us definitive answers to some of these questions,” he said.
Recommendations
The seven key recommendations, based on the clinical questions raised, are summarized below:
- Delayed (asynchronous) reconstruction is acceptable. Although the quality of the evidence is low and the recommendations are listed as an option, the guideline authors said that depending on the situation, reconstruction can be performed either immediately after resection or delayed by days, weeks, “or even months.”
- Systemic antibiotics should not be routinely prescribed in the interim between resection and reconstruction in adults. Here too, the evidence is low and the recommendation strength is weak, but in “the absence of data showing convincing benefits, systemic antibiotic therapy does not appear necessary or desirable in most cases when there is an interval between cancer resection and reconstruction,” the work group wrote.
- Clinicians may administer perioperative systemic antibiotics in a facility-based setting for adults undergoing reconstruction (3a), but antibiotics should not be routinely prescribed in an office-based setting (3b). The rationale for these recommendations, supported by a moderate level of evidence, is that the risk of surgical-site infection is generally higher in facilities, compared with an office-based setting. Patients who undergo reconstruction in hospitals or surgical centers are more likely to have complex reconstructions or have risks that may make them suitable candidates for antibiotics, but patients in office-based setting may often be spared from the additional costs, side effects, and possible drug interactions from antibiotic use. “There is no evidence in either setting that long-term antibiotic prophylaxis provides infection risk reduction, compared with short-term prophylaxis,” the guideline working group wrote.
- Continue anticoagulant, antithrombotic, and antiplatelet medications for adult patients undergoing reconstruction after skin cancer resection in the office-based setting (4a), and in the facility-based setting should coordinate with the physician managing anticoagulation before modifying the medication prior to surgery (4b). Evidence quality and recommendation strength are both moderate.
- The guideline authors recommend against routine prescription of narcotics as first-line treatment for pain in adults undergoing skin reconstruction (5a), favoring instead acetaminophen and NSAIDs as first-line therapy (5b). Evidence quality and recommendation strength are both moderate.
- In the absence of standardized protocols for the management of pain medications, oral antibiotics, and/or anticoagulants in the perioperative period, clinicians should discuss possible approaches with adult patients. “Educating patients about their perioperative treatment through discussion of treatment strategies may help alleviate anxiety, improve communication, increase patient satisfaction, and maximize patient compliance with the postoperative orders,” the guideline authors wrote.
- The authors suggest that adult patients may be offered follow-up assessments to discuss functional and cosmetic outcomes. “The return of the patient for follow-up visits is an excellent opportunity to better understand and measure these outcomes, improve patient-physician communication, and foster quality improvement. Postoperative follow-up can lead to increased communication between the patient and physician, thereby empowering patients to comment on satisfaction and other important outcomes measures,” they wrote.
What’s next
The guideline developers acknowledged that data are limited regarding reconstructive surgery following skin cancer resection, and that higher-quality studies would help to improve future guidelines. Dr. Chen said that greater use of prospective surgical databases and more systematic collection of patient-reported outcomes could inform further efforts.
The guideline development process was supported by the various groups represented. Dr. Chen and Dr. Cetrulo reported no relevant disclosures.
You’ve successfully resected a skin cancer lesion, leaving clear margins. Now what?
That’s
The guideline – a joint effort of the American Society of Plastic Surgeons, American Society for Dermatologic Surgery, American Academy of Dermatology, American Academy of Facial Plastic and Reconstructive Surgery, American Academy of Otolaryngology – Head and Neck Surgery Foundation, American College of Mohs Surgery, American Society for Mohs Surgery, and American Society of Ophthalmic Plastic and Reconstructive Surgery – was published online in the Journal of the American Academy of Dermatology.
From the outset, the panel members realized that to keep the guideline manageable they had to limit recommendations to the practice of reconstruction defined as “cutaneous closure that requires a flap, graft, or tissue rearrangement.”
Other wound closure methods, such as secondary intention healing; simple closures; and complex closures that do not involve flaps, grafts, muscle, or bone, were not covered in the recommendations.
As with similar guidelines, the developers selected seven clinical questions to be addressed, and attempted to find consensus through literature searches, appraisal of the evidence, grading of recommendations, peer review, and public comment.
“We had a very heterogeneous set of things that we were trying to comment on, so we had to keep things somewhat generic,” lead author Andrew Chen, MD, chief of the division of plastic surgery, at the University of Connecticut Health Center, Farmington, said in an interview.
“Skin cancer and reconstruction affect different body areas and areas of different sizes. When we were creating the guidelines, we had to tailor the questions we could ask based on things that would make sense to answer, because obviously we couldn’t ask a question such as: ‘What’s better, a skin graft or a flap?’ Well, there are some things you can’t put a skin graft on – it won’t last, so we couldn’t ask that kind of question,” Dr. Chen said.
Curtis Cetrulo, MD, a plastic and reconstructive surgeon at Massachusetts General Hospital, Boston, who was not involved in the guideline process, said in an interview that the broad recommendations are in keeping with his practice and experience. He also acknowledged, however, the difficulty in creating a guideline that covers the complexity and heterogeneity of reconstructive surgery.
“These are generally good recommendations, but they’re recommendations only, with generally weak levels of evidence. What we really need are clinical trials that can give us definitive answers to some of these questions,” he said.
Recommendations
The seven key recommendations, based on the clinical questions raised, are summarized below:
- Delayed (asynchronous) reconstruction is acceptable. Although the quality of the evidence is low and the recommendations are listed as an option, the guideline authors said that depending on the situation, reconstruction can be performed either immediately after resection or delayed by days, weeks, “or even months.”
- Systemic antibiotics should not be routinely prescribed in the interim between resection and reconstruction in adults. Here too, the evidence is low and the recommendation strength is weak, but in “the absence of data showing convincing benefits, systemic antibiotic therapy does not appear necessary or desirable in most cases when there is an interval between cancer resection and reconstruction,” the work group wrote.
- Clinicians may administer perioperative systemic antibiotics in a facility-based setting for adults undergoing reconstruction (3a), but antibiotics should not be routinely prescribed in an office-based setting (3b). The rationale for these recommendations, supported by a moderate level of evidence, is that the risk of surgical-site infection is generally higher in facilities, compared with an office-based setting. Patients who undergo reconstruction in hospitals or surgical centers are more likely to have complex reconstructions or have risks that may make them suitable candidates for antibiotics, but patients in office-based setting may often be spared from the additional costs, side effects, and possible drug interactions from antibiotic use. “There is no evidence in either setting that long-term antibiotic prophylaxis provides infection risk reduction, compared with short-term prophylaxis,” the guideline working group wrote.
- Continue anticoagulant, antithrombotic, and antiplatelet medications for adult patients undergoing reconstruction after skin cancer resection in the office-based setting (4a), and in the facility-based setting should coordinate with the physician managing anticoagulation before modifying the medication prior to surgery (4b). Evidence quality and recommendation strength are both moderate.
- The guideline authors recommend against routine prescription of narcotics as first-line treatment for pain in adults undergoing skin reconstruction (5a), favoring instead acetaminophen and NSAIDs as first-line therapy (5b). Evidence quality and recommendation strength are both moderate.
- In the absence of standardized protocols for the management of pain medications, oral antibiotics, and/or anticoagulants in the perioperative period, clinicians should discuss possible approaches with adult patients. “Educating patients about their perioperative treatment through discussion of treatment strategies may help alleviate anxiety, improve communication, increase patient satisfaction, and maximize patient compliance with the postoperative orders,” the guideline authors wrote.
- The authors suggest that adult patients may be offered follow-up assessments to discuss functional and cosmetic outcomes. “The return of the patient for follow-up visits is an excellent opportunity to better understand and measure these outcomes, improve patient-physician communication, and foster quality improvement. Postoperative follow-up can lead to increased communication between the patient and physician, thereby empowering patients to comment on satisfaction and other important outcomes measures,” they wrote.
What’s next
The guideline developers acknowledged that data are limited regarding reconstructive surgery following skin cancer resection, and that higher-quality studies would help to improve future guidelines. Dr. Chen said that greater use of prospective surgical databases and more systematic collection of patient-reported outcomes could inform further efforts.
The guideline development process was supported by the various groups represented. Dr. Chen and Dr. Cetrulo reported no relevant disclosures.
You’ve successfully resected a skin cancer lesion, leaving clear margins. Now what?
That’s
The guideline – a joint effort of the American Society of Plastic Surgeons, American Society for Dermatologic Surgery, American Academy of Dermatology, American Academy of Facial Plastic and Reconstructive Surgery, American Academy of Otolaryngology – Head and Neck Surgery Foundation, American College of Mohs Surgery, American Society for Mohs Surgery, and American Society of Ophthalmic Plastic and Reconstructive Surgery – was published online in the Journal of the American Academy of Dermatology.
From the outset, the panel members realized that to keep the guideline manageable they had to limit recommendations to the practice of reconstruction defined as “cutaneous closure that requires a flap, graft, or tissue rearrangement.”
Other wound closure methods, such as secondary intention healing; simple closures; and complex closures that do not involve flaps, grafts, muscle, or bone, were not covered in the recommendations.
As with similar guidelines, the developers selected seven clinical questions to be addressed, and attempted to find consensus through literature searches, appraisal of the evidence, grading of recommendations, peer review, and public comment.
“We had a very heterogeneous set of things that we were trying to comment on, so we had to keep things somewhat generic,” lead author Andrew Chen, MD, chief of the division of plastic surgery, at the University of Connecticut Health Center, Farmington, said in an interview.
“Skin cancer and reconstruction affect different body areas and areas of different sizes. When we were creating the guidelines, we had to tailor the questions we could ask based on things that would make sense to answer, because obviously we couldn’t ask a question such as: ‘What’s better, a skin graft or a flap?’ Well, there are some things you can’t put a skin graft on – it won’t last, so we couldn’t ask that kind of question,” Dr. Chen said.
Curtis Cetrulo, MD, a plastic and reconstructive surgeon at Massachusetts General Hospital, Boston, who was not involved in the guideline process, said in an interview that the broad recommendations are in keeping with his practice and experience. He also acknowledged, however, the difficulty in creating a guideline that covers the complexity and heterogeneity of reconstructive surgery.
“These are generally good recommendations, but they’re recommendations only, with generally weak levels of evidence. What we really need are clinical trials that can give us definitive answers to some of these questions,” he said.
Recommendations
The seven key recommendations, based on the clinical questions raised, are summarized below:
- Delayed (asynchronous) reconstruction is acceptable. Although the quality of the evidence is low and the recommendations are listed as an option, the guideline authors said that depending on the situation, reconstruction can be performed either immediately after resection or delayed by days, weeks, “or even months.”
- Systemic antibiotics should not be routinely prescribed in the interim between resection and reconstruction in adults. Here too, the evidence is low and the recommendation strength is weak, but in “the absence of data showing convincing benefits, systemic antibiotic therapy does not appear necessary or desirable in most cases when there is an interval between cancer resection and reconstruction,” the work group wrote.
- Clinicians may administer perioperative systemic antibiotics in a facility-based setting for adults undergoing reconstruction (3a), but antibiotics should not be routinely prescribed in an office-based setting (3b). The rationale for these recommendations, supported by a moderate level of evidence, is that the risk of surgical-site infection is generally higher in facilities, compared with an office-based setting. Patients who undergo reconstruction in hospitals or surgical centers are more likely to have complex reconstructions or have risks that may make them suitable candidates for antibiotics, but patients in office-based setting may often be spared from the additional costs, side effects, and possible drug interactions from antibiotic use. “There is no evidence in either setting that long-term antibiotic prophylaxis provides infection risk reduction, compared with short-term prophylaxis,” the guideline working group wrote.
- Continue anticoagulant, antithrombotic, and antiplatelet medications for adult patients undergoing reconstruction after skin cancer resection in the office-based setting (4a), and in the facility-based setting should coordinate with the physician managing anticoagulation before modifying the medication prior to surgery (4b). Evidence quality and recommendation strength are both moderate.
- The guideline authors recommend against routine prescription of narcotics as first-line treatment for pain in adults undergoing skin reconstruction (5a), favoring instead acetaminophen and NSAIDs as first-line therapy (5b). Evidence quality and recommendation strength are both moderate.
- In the absence of standardized protocols for the management of pain medications, oral antibiotics, and/or anticoagulants in the perioperative period, clinicians should discuss possible approaches with adult patients. “Educating patients about their perioperative treatment through discussion of treatment strategies may help alleviate anxiety, improve communication, increase patient satisfaction, and maximize patient compliance with the postoperative orders,” the guideline authors wrote.
- The authors suggest that adult patients may be offered follow-up assessments to discuss functional and cosmetic outcomes. “The return of the patient for follow-up visits is an excellent opportunity to better understand and measure these outcomes, improve patient-physician communication, and foster quality improvement. Postoperative follow-up can lead to increased communication between the patient and physician, thereby empowering patients to comment on satisfaction and other important outcomes measures,” they wrote.
What’s next
The guideline developers acknowledged that data are limited regarding reconstructive surgery following skin cancer resection, and that higher-quality studies would help to improve future guidelines. Dr. Chen said that greater use of prospective surgical databases and more systematic collection of patient-reported outcomes could inform further efforts.
The guideline development process was supported by the various groups represented. Dr. Chen and Dr. Cetrulo reported no relevant disclosures.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY