User login
Cyclops Lambs Played Important Role in Vismodegib's Approval
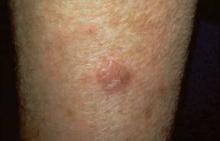
MIAMI BEACH – A new therapy to combat advanced basal cell carcinoma is generating excitement in the few months since its Food and Drug Administration approval.
"Vismodegib provides substantial clinical benefit for patients with advanced basal cell carcinoma [BCC]," Dr. Scott M. Dinehart said at the South Beach Symposium. "For dermatology, this is a very important pathway." Through its novel ability to block a signaling pathway implicated in the development BCC, it can help treat "the kinds of terrible skin cancers where maybe you can operate on them or maybe not," said Dr. Dinehart, a dermatologist in private practice in Little Rock, Ark.
The FDA approved the oral, once-daily medication in January 2012 to treat adults with metastatic BCC or locally advanced, recurrent BCC after surgery. It is also indicated for patients with locally advanced BCC who are not candidates for surgery or radiation treatment.
Most side effects are mild to moderate, Dr. Dinehart said. "Muscle spasms are the one I am most worried about," he added, saying that such events might cause patients to discontinue use of the agent. Patients might also experience hair loss and taste changes because the hedgehog pathway is active in differentiation and proliferation of hair follicles and taste buds. "The side effects are the kind we can work around," Dr. Dinehart said.
Weight loss, fatigue, nausea, diarrhea, decreased appetite, constipation, arthralgias, and vomiting were other adverse events reported by 10% or more of participants in preclinical trials, according to the medication guide. Because of its teratogenicity, vismodegib carries a black box warning about embryo-fetal death and severe birth defects.
The hedgehog pathway inhibitor, vismodegib (Erivedge, Genentech), not only offers promise for patients with advanced BCC, but its approval also culminates a fascinating story of scientific discovery.
Five and half decades ago, in 1957, a herd of sheep in Idaho gave birth to one-eyed lambs. U.S. Department of Agriculture investigators determined that the "cyclops" lambs were born after dry weather drove the sheep to higher ground, where they ate corn lilies that contained a teratogenic toxin. They dubbed the toxin "cyclopamine."
The toxin blocks the segmentation of the brain and the two halves of the brain don’t separate during embryonic development, Dr. Brian Berman said in a separate presentation at the meeting. "Humans are also susceptible," added Dr. Berman, a professor of dermatology and cutaneous surgery at the University of Miami.
The toxin discovery probably would have remained a footnote in history, Dr. Dinehart said, except for scientists who looked at oncologic properties of this teratogenic compound. For example, Philip A. Beachy, Ph.D., while at Johns Hopkins in the early 1990s, cloned a hedgehog pathway gene in fruit flies that regulates embryonic cell differentiation. He observed that fruit fly embryos born with a faulty copy of the gene had a spiky or pointy appearance.
Dr. Berman said the most important of the hedgehog pathways in humans is called sonic hedgehog, which was named "after the Nintendo character with the spiky hair."
Dr. Dinehart said he is a consultant for Genentech. Dr. Berman is a consultant and a member of the speakers bureau for Genentech.
MIAMI BEACH – A new therapy to combat advanced basal cell carcinoma is generating excitement in the few months since its Food and Drug Administration approval.
"Vismodegib provides substantial clinical benefit for patients with advanced basal cell carcinoma [BCC]," Dr. Scott M. Dinehart said at the South Beach Symposium. "For dermatology, this is a very important pathway." Through its novel ability to block a signaling pathway implicated in the development BCC, it can help treat "the kinds of terrible skin cancers where maybe you can operate on them or maybe not," said Dr. Dinehart, a dermatologist in private practice in Little Rock, Ark.
The FDA approved the oral, once-daily medication in January 2012 to treat adults with metastatic BCC or locally advanced, recurrent BCC after surgery. It is also indicated for patients with locally advanced BCC who are not candidates for surgery or radiation treatment.
Most side effects are mild to moderate, Dr. Dinehart said. "Muscle spasms are the one I am most worried about," he added, saying that such events might cause patients to discontinue use of the agent. Patients might also experience hair loss and taste changes because the hedgehog pathway is active in differentiation and proliferation of hair follicles and taste buds. "The side effects are the kind we can work around," Dr. Dinehart said.
Weight loss, fatigue, nausea, diarrhea, decreased appetite, constipation, arthralgias, and vomiting were other adverse events reported by 10% or more of participants in preclinical trials, according to the medication guide. Because of its teratogenicity, vismodegib carries a black box warning about embryo-fetal death and severe birth defects.
The hedgehog pathway inhibitor, vismodegib (Erivedge, Genentech), not only offers promise for patients with advanced BCC, but its approval also culminates a fascinating story of scientific discovery.
Five and half decades ago, in 1957, a herd of sheep in Idaho gave birth to one-eyed lambs. U.S. Department of Agriculture investigators determined that the "cyclops" lambs were born after dry weather drove the sheep to higher ground, where they ate corn lilies that contained a teratogenic toxin. They dubbed the toxin "cyclopamine."
The toxin blocks the segmentation of the brain and the two halves of the brain don’t separate during embryonic development, Dr. Brian Berman said in a separate presentation at the meeting. "Humans are also susceptible," added Dr. Berman, a professor of dermatology and cutaneous surgery at the University of Miami.
The toxin discovery probably would have remained a footnote in history, Dr. Dinehart said, except for scientists who looked at oncologic properties of this teratogenic compound. For example, Philip A. Beachy, Ph.D., while at Johns Hopkins in the early 1990s, cloned a hedgehog pathway gene in fruit flies that regulates embryonic cell differentiation. He observed that fruit fly embryos born with a faulty copy of the gene had a spiky or pointy appearance.
Dr. Berman said the most important of the hedgehog pathways in humans is called sonic hedgehog, which was named "after the Nintendo character with the spiky hair."
Dr. Dinehart said he is a consultant for Genentech. Dr. Berman is a consultant and a member of the speakers bureau for Genentech.
MIAMI BEACH – A new therapy to combat advanced basal cell carcinoma is generating excitement in the few months since its Food and Drug Administration approval.
"Vismodegib provides substantial clinical benefit for patients with advanced basal cell carcinoma [BCC]," Dr. Scott M. Dinehart said at the South Beach Symposium. "For dermatology, this is a very important pathway." Through its novel ability to block a signaling pathway implicated in the development BCC, it can help treat "the kinds of terrible skin cancers where maybe you can operate on them or maybe not," said Dr. Dinehart, a dermatologist in private practice in Little Rock, Ark.
The FDA approved the oral, once-daily medication in January 2012 to treat adults with metastatic BCC or locally advanced, recurrent BCC after surgery. It is also indicated for patients with locally advanced BCC who are not candidates for surgery or radiation treatment.
Most side effects are mild to moderate, Dr. Dinehart said. "Muscle spasms are the one I am most worried about," he added, saying that such events might cause patients to discontinue use of the agent. Patients might also experience hair loss and taste changes because the hedgehog pathway is active in differentiation and proliferation of hair follicles and taste buds. "The side effects are the kind we can work around," Dr. Dinehart said.
Weight loss, fatigue, nausea, diarrhea, decreased appetite, constipation, arthralgias, and vomiting were other adverse events reported by 10% or more of participants in preclinical trials, according to the medication guide. Because of its teratogenicity, vismodegib carries a black box warning about embryo-fetal death and severe birth defects.
The hedgehog pathway inhibitor, vismodegib (Erivedge, Genentech), not only offers promise for patients with advanced BCC, but its approval also culminates a fascinating story of scientific discovery.
Five and half decades ago, in 1957, a herd of sheep in Idaho gave birth to one-eyed lambs. U.S. Department of Agriculture investigators determined that the "cyclops" lambs were born after dry weather drove the sheep to higher ground, where they ate corn lilies that contained a teratogenic toxin. They dubbed the toxin "cyclopamine."
The toxin blocks the segmentation of the brain and the two halves of the brain don’t separate during embryonic development, Dr. Brian Berman said in a separate presentation at the meeting. "Humans are also susceptible," added Dr. Berman, a professor of dermatology and cutaneous surgery at the University of Miami.
The toxin discovery probably would have remained a footnote in history, Dr. Dinehart said, except for scientists who looked at oncologic properties of this teratogenic compound. For example, Philip A. Beachy, Ph.D., while at Johns Hopkins in the early 1990s, cloned a hedgehog pathway gene in fruit flies that regulates embryonic cell differentiation. He observed that fruit fly embryos born with a faulty copy of the gene had a spiky or pointy appearance.
Dr. Berman said the most important of the hedgehog pathways in humans is called sonic hedgehog, which was named "after the Nintendo character with the spiky hair."
Dr. Dinehart said he is a consultant for Genentech. Dr. Berman is a consultant and a member of the speakers bureau for Genentech.
EXPERT ANALYSIS FROM THE SOUTH BEACH SYMPOSIUM
Cancer Diagnosis Appears to Raise Suicide, Cardiovascular Risks
People who receive a cancer diagnosis are at markedly increased risk for suicide and for fatal cardiovascular events in the following weeks, according to a large Swedish cohort study reported in the April 5 issue of the New England Journal of Medicine.
"This spike in risk was particularly prominent among patients in whom cancers with a poor prognosis were diagnosed, and was not explained by preexisting psychiatric or cardiovascular conditions," said Dr. Fang Fang of the department of medical epidemiology and biostatistics, Karolinska Institutet, Stockholm, and her associates.
The dramatic increase in mortality risk from suicide or cardiovascular causes also cannot be attributed to cancer treatment or cancer progression, since it peaks during the first week after diagnosis and declined over time rather than increasing as treatment intensified or disease worsened, they noted.
Previous studies have demonstrated that cancer patients are at increased risk for suicide and CV events, but most have attributed it to "the burden of living with a progressing cancer." Very few have explored the period immediately following a cancer diagnosis, Dr. Fang and her colleagues said.
They examined this time period using data from a Swedish national registry of cancers and causes of death, both of which must be reported by law in Sweden. The study period, 1991 through 2006, covered more than 6 million Swedes aged 30 and older at baseline.
A total of 534,154 people received a first cancer diagnosis during this interval, including 95,786 with prostate cancer, 74,977 with breast cancer (among women), 62,719 with colorectal cancer, 47,169 with skin cancer, 36,648 with lymphatic or hematopoietic cancer, 34,743 with lung cancer, and 13,447 with CNS cancer. Another 26,335 patients were pooled in a single category of "highly fatal cancers of the esophagus, liver, and pancreas," and the remaining 142,330 patients had other types of cancer.
During follow-up, there were 786 completed suicides among patients with any type of cancer, for a rate of 0.36 per 1,000 person-years. This was double the rate among Swedish adults who did not have cancer: 13,284 completed suicides for a rate of 0.18 per 1,000 person-years (N. Engl. J. Med. 2012;366:1310-8).
The rate of completed suicides peaked during the first week after cancer diagnosis, at 2.50 per 1,000 person-years.
"We found a relative risk of 4.8 during the first 12 weeks after diagnosis (110 patients; incidence rate, 0.95 per 1,000 person-years), with the highest relative risk observed for highly fatal cancers of the esophagus, liver, or pancreas, followed by lung cancer," they added.
The magnitude of elevated risk dropped rapidly after that, but remained higher than average beyond the first year for all cancers.
"Focusing on the first 52 weeks after a diagnosis of any cancer, we found a relative risk of 3.1 for suicide (260 patients; incidence rate, 0.60 per 1,000 person-years). The expected number of suicides, adjusted for all demographic factors, during these 52 weeks was 87, leaving 173 cases associated with cancer diagnoses," the researchers said.
The relative risks of suicide were stronger among patients who had no concomitant psychiatric disorders than among those with psychiatric disorders.
Turning to fatal cardiovascular events, there were 48,991 CV deaths among people who received a diagnosis of any cancer, for an incidence of 23.10 per 1,000 person-years. This rate is approximately three times higher than that for people who did not have cancer (543,144 deaths; incidence rate, 7.53 per 1,000 person-years.
As with suicide, the highest relative risk of CV death (5.6) peaked during the first week after diagnosis (1,318 patients; incidence rate, 116.8 per 1,000 person-years). The risk elevation was highest for central nervous system cancers, followed by highly fatal cancers of the esophagus, liver, or pancreas, and then by lung cancer.
Also as with suicide, the magnitude of the increased risk of CV death dropped rapidly after the first several weeks. It did not persist beyond the first year after diagnosis.
"Focusing on the first 4 weeks after a diagnosis of any cancer but CNS tumors, we found a relative risk of 3.3 for cardiovascular death (2,641 patients; incidence rate, 65.81 per 1,000 person-years). The adjusted expected number of CV deaths was 766 during these 4 weeks, leaving 1,875 deaths associated with a cancer diagnosis," the investigators said.
Of note was the finding that the increased hazards were seen in both men and women.
To adjust for unmeasured confounders, the researchers performed an additional case-crossover analysis among all cancer patients in the cohort who died from suicide or CV causes. The results "further allayed the concern" that there may be some alternative explanation for the observed associations, such as an unknown factor that causes cancer, suicide, and CV death.
Dr. Fang and her associates emphasized that their study "focused on hard outcomes alone, and thus probably did not capture the full extent of the psychological burden among patients with newly diagnosed cancer. Other potentially relevant outcomes, such as attempted suicide and severe but nonfatal CV events, remain to be explored," they noted.
In addition, this study involved only adults aged 30 years and older. Further studies are warranted to examine the immediate after-effects of a cancer diagnosis in children, adolescents, and young adults.
Most importantly, now that these elevated risks have been identified, future research must address prevention strategies, they said.
This study was supported by the Swedish Council for Working Life and Social Research, the Swedish Research Council, Hjärnfonden, and Svenska Sällskapet för Medicinsk Forskning. No financial conflicts of interest were reported.
People who receive a cancer diagnosis are at markedly increased risk for suicide and for fatal cardiovascular events in the following weeks, according to a large Swedish cohort study reported in the April 5 issue of the New England Journal of Medicine.
"This spike in risk was particularly prominent among patients in whom cancers with a poor prognosis were diagnosed, and was not explained by preexisting psychiatric or cardiovascular conditions," said Dr. Fang Fang of the department of medical epidemiology and biostatistics, Karolinska Institutet, Stockholm, and her associates.
The dramatic increase in mortality risk from suicide or cardiovascular causes also cannot be attributed to cancer treatment or cancer progression, since it peaks during the first week after diagnosis and declined over time rather than increasing as treatment intensified or disease worsened, they noted.
Previous studies have demonstrated that cancer patients are at increased risk for suicide and CV events, but most have attributed it to "the burden of living with a progressing cancer." Very few have explored the period immediately following a cancer diagnosis, Dr. Fang and her colleagues said.
They examined this time period using data from a Swedish national registry of cancers and causes of death, both of which must be reported by law in Sweden. The study period, 1991 through 2006, covered more than 6 million Swedes aged 30 and older at baseline.
A total of 534,154 people received a first cancer diagnosis during this interval, including 95,786 with prostate cancer, 74,977 with breast cancer (among women), 62,719 with colorectal cancer, 47,169 with skin cancer, 36,648 with lymphatic or hematopoietic cancer, 34,743 with lung cancer, and 13,447 with CNS cancer. Another 26,335 patients were pooled in a single category of "highly fatal cancers of the esophagus, liver, and pancreas," and the remaining 142,330 patients had other types of cancer.
During follow-up, there were 786 completed suicides among patients with any type of cancer, for a rate of 0.36 per 1,000 person-years. This was double the rate among Swedish adults who did not have cancer: 13,284 completed suicides for a rate of 0.18 per 1,000 person-years (N. Engl. J. Med. 2012;366:1310-8).
The rate of completed suicides peaked during the first week after cancer diagnosis, at 2.50 per 1,000 person-years.
"We found a relative risk of 4.8 during the first 12 weeks after diagnosis (110 patients; incidence rate, 0.95 per 1,000 person-years), with the highest relative risk observed for highly fatal cancers of the esophagus, liver, or pancreas, followed by lung cancer," they added.
The magnitude of elevated risk dropped rapidly after that, but remained higher than average beyond the first year for all cancers.
"Focusing on the first 52 weeks after a diagnosis of any cancer, we found a relative risk of 3.1 for suicide (260 patients; incidence rate, 0.60 per 1,000 person-years). The expected number of suicides, adjusted for all demographic factors, during these 52 weeks was 87, leaving 173 cases associated with cancer diagnoses," the researchers said.
The relative risks of suicide were stronger among patients who had no concomitant psychiatric disorders than among those with psychiatric disorders.
Turning to fatal cardiovascular events, there were 48,991 CV deaths among people who received a diagnosis of any cancer, for an incidence of 23.10 per 1,000 person-years. This rate is approximately three times higher than that for people who did not have cancer (543,144 deaths; incidence rate, 7.53 per 1,000 person-years.
As with suicide, the highest relative risk of CV death (5.6) peaked during the first week after diagnosis (1,318 patients; incidence rate, 116.8 per 1,000 person-years). The risk elevation was highest for central nervous system cancers, followed by highly fatal cancers of the esophagus, liver, or pancreas, and then by lung cancer.
Also as with suicide, the magnitude of the increased risk of CV death dropped rapidly after the first several weeks. It did not persist beyond the first year after diagnosis.
"Focusing on the first 4 weeks after a diagnosis of any cancer but CNS tumors, we found a relative risk of 3.3 for cardiovascular death (2,641 patients; incidence rate, 65.81 per 1,000 person-years). The adjusted expected number of CV deaths was 766 during these 4 weeks, leaving 1,875 deaths associated with a cancer diagnosis," the investigators said.
Of note was the finding that the increased hazards were seen in both men and women.
To adjust for unmeasured confounders, the researchers performed an additional case-crossover analysis among all cancer patients in the cohort who died from suicide or CV causes. The results "further allayed the concern" that there may be some alternative explanation for the observed associations, such as an unknown factor that causes cancer, suicide, and CV death.
Dr. Fang and her associates emphasized that their study "focused on hard outcomes alone, and thus probably did not capture the full extent of the psychological burden among patients with newly diagnosed cancer. Other potentially relevant outcomes, such as attempted suicide and severe but nonfatal CV events, remain to be explored," they noted.
In addition, this study involved only adults aged 30 years and older. Further studies are warranted to examine the immediate after-effects of a cancer diagnosis in children, adolescents, and young adults.
Most importantly, now that these elevated risks have been identified, future research must address prevention strategies, they said.
This study was supported by the Swedish Council for Working Life and Social Research, the Swedish Research Council, Hjärnfonden, and Svenska Sällskapet för Medicinsk Forskning. No financial conflicts of interest were reported.
People who receive a cancer diagnosis are at markedly increased risk for suicide and for fatal cardiovascular events in the following weeks, according to a large Swedish cohort study reported in the April 5 issue of the New England Journal of Medicine.
"This spike in risk was particularly prominent among patients in whom cancers with a poor prognosis were diagnosed, and was not explained by preexisting psychiatric or cardiovascular conditions," said Dr. Fang Fang of the department of medical epidemiology and biostatistics, Karolinska Institutet, Stockholm, and her associates.
The dramatic increase in mortality risk from suicide or cardiovascular causes also cannot be attributed to cancer treatment or cancer progression, since it peaks during the first week after diagnosis and declined over time rather than increasing as treatment intensified or disease worsened, they noted.
Previous studies have demonstrated that cancer patients are at increased risk for suicide and CV events, but most have attributed it to "the burden of living with a progressing cancer." Very few have explored the period immediately following a cancer diagnosis, Dr. Fang and her colleagues said.
They examined this time period using data from a Swedish national registry of cancers and causes of death, both of which must be reported by law in Sweden. The study period, 1991 through 2006, covered more than 6 million Swedes aged 30 and older at baseline.
A total of 534,154 people received a first cancer diagnosis during this interval, including 95,786 with prostate cancer, 74,977 with breast cancer (among women), 62,719 with colorectal cancer, 47,169 with skin cancer, 36,648 with lymphatic or hematopoietic cancer, 34,743 with lung cancer, and 13,447 with CNS cancer. Another 26,335 patients were pooled in a single category of "highly fatal cancers of the esophagus, liver, and pancreas," and the remaining 142,330 patients had other types of cancer.
During follow-up, there were 786 completed suicides among patients with any type of cancer, for a rate of 0.36 per 1,000 person-years. This was double the rate among Swedish adults who did not have cancer: 13,284 completed suicides for a rate of 0.18 per 1,000 person-years (N. Engl. J. Med. 2012;366:1310-8).
The rate of completed suicides peaked during the first week after cancer diagnosis, at 2.50 per 1,000 person-years.
"We found a relative risk of 4.8 during the first 12 weeks after diagnosis (110 patients; incidence rate, 0.95 per 1,000 person-years), with the highest relative risk observed for highly fatal cancers of the esophagus, liver, or pancreas, followed by lung cancer," they added.
The magnitude of elevated risk dropped rapidly after that, but remained higher than average beyond the first year for all cancers.
"Focusing on the first 52 weeks after a diagnosis of any cancer, we found a relative risk of 3.1 for suicide (260 patients; incidence rate, 0.60 per 1,000 person-years). The expected number of suicides, adjusted for all demographic factors, during these 52 weeks was 87, leaving 173 cases associated with cancer diagnoses," the researchers said.
The relative risks of suicide were stronger among patients who had no concomitant psychiatric disorders than among those with psychiatric disorders.
Turning to fatal cardiovascular events, there were 48,991 CV deaths among people who received a diagnosis of any cancer, for an incidence of 23.10 per 1,000 person-years. This rate is approximately three times higher than that for people who did not have cancer (543,144 deaths; incidence rate, 7.53 per 1,000 person-years.
As with suicide, the highest relative risk of CV death (5.6) peaked during the first week after diagnosis (1,318 patients; incidence rate, 116.8 per 1,000 person-years). The risk elevation was highest for central nervous system cancers, followed by highly fatal cancers of the esophagus, liver, or pancreas, and then by lung cancer.
Also as with suicide, the magnitude of the increased risk of CV death dropped rapidly after the first several weeks. It did not persist beyond the first year after diagnosis.
"Focusing on the first 4 weeks after a diagnosis of any cancer but CNS tumors, we found a relative risk of 3.3 for cardiovascular death (2,641 patients; incidence rate, 65.81 per 1,000 person-years). The adjusted expected number of CV deaths was 766 during these 4 weeks, leaving 1,875 deaths associated with a cancer diagnosis," the investigators said.
Of note was the finding that the increased hazards were seen in both men and women.
To adjust for unmeasured confounders, the researchers performed an additional case-crossover analysis among all cancer patients in the cohort who died from suicide or CV causes. The results "further allayed the concern" that there may be some alternative explanation for the observed associations, such as an unknown factor that causes cancer, suicide, and CV death.
Dr. Fang and her associates emphasized that their study "focused on hard outcomes alone, and thus probably did not capture the full extent of the psychological burden among patients with newly diagnosed cancer. Other potentially relevant outcomes, such as attempted suicide and severe but nonfatal CV events, remain to be explored," they noted.
In addition, this study involved only adults aged 30 years and older. Further studies are warranted to examine the immediate after-effects of a cancer diagnosis in children, adolescents, and young adults.
Most importantly, now that these elevated risks have been identified, future research must address prevention strategies, they said.
This study was supported by the Swedish Council for Working Life and Social Research, the Swedish Research Council, Hjärnfonden, and Svenska Sällskapet för Medicinsk Forskning. No financial conflicts of interest were reported.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major Finding: The rate of completed suicide was twice as high among adults with newly diagnosed cancer of any kind (0.36 per 1,000 person-years) than among adults without cancer (0.18 per 1,000 person-years), and the rate of fatal CV events was three times higher (23.20 per 1,000 person-years vs. 7.53 per 1,000 person-years).
Data Source: This was a nationwide historical cohort study of 6,073,240 Swedish adults followed from 1991 through 2006 for cancer diagnoses and for death by suicide or CV events.
Disclosures: This study was supported by the Swedish Council for Working Life and Social Research, the Swedish Research Council, Hjärnfonden, and Svenska Sällskapet för Medicinsk Forskning. No financial conflicts of interest were reported.
AAD Unveils SPOT Program: The Skinny Podcast
In this month's program, Dr. Daniel E. Furst delivers tips for differentiating arthritis types in psoriasis patients with joint pain.
Dr. Thomas E. Rohrer shares the unveiling of the AAD's new prevention program: SPOT Skin Cancer.
Then, Dr. Lawrence F. Eichenfield discusses the first-ever acne treatment guidelines for children.
Cosmetic counter host Dr. Lily Talakoub explains how to talk to patients about the potentially toxic chemicals in their makeup bag.
And finally, Dr. Alan Rockoff explains why his young grandson doesn't want to be a "lotion" doctor.
In this month's program, Dr. Daniel E. Furst delivers tips for differentiating arthritis types in psoriasis patients with joint pain.
Dr. Thomas E. Rohrer shares the unveiling of the AAD's new prevention program: SPOT Skin Cancer.
Then, Dr. Lawrence F. Eichenfield discusses the first-ever acne treatment guidelines for children.
Cosmetic counter host Dr. Lily Talakoub explains how to talk to patients about the potentially toxic chemicals in their makeup bag.
And finally, Dr. Alan Rockoff explains why his young grandson doesn't want to be a "lotion" doctor.
In this month's program, Dr. Daniel E. Furst delivers tips for differentiating arthritis types in psoriasis patients with joint pain.
Dr. Thomas E. Rohrer shares the unveiling of the AAD's new prevention program: SPOT Skin Cancer.
Then, Dr. Lawrence F. Eichenfield discusses the first-ever acne treatment guidelines for children.
Cosmetic counter host Dr. Lily Talakoub explains how to talk to patients about the potentially toxic chemicals in their makeup bag.
And finally, Dr. Alan Rockoff explains why his young grandson doesn't want to be a "lotion" doctor.
Limb Perfusion for In-Transit Melanoma Reduces Distant Recurrences
ORLANDO – The type of regional chemotherapy given to patients with in-transit or intralymphatic melanoma of the extremities appears to make a difference in out-of-field recurrences and time to distant recurrence, reported investigators at a symposium of the Society of Surgical Oncology.
A study of 214 patients who underwent either first-time hyperthermic isolated limb perfusion (HILP) or isolated limb infusion (ILI) for in-transit melanomas showed that HILP was associated with significantly more in-field complete responses (P = .01), and a longer median time to out-of-field recurrences, compared with ILI, said Dr. Ketan Sharma of Duke University Medical Center in Durham, NC.
"We found that perfusion complete response and infusion complete response exhibit similar degrees of in-field disease control," Dr. Sharma said.
However, "recurrent disease after a regional therapy complete response is complex, and requires a multidisciplinary approach to treatment," he added.
The National Cancer Institute defines an in-transit metastasis as a "type of metastasis in which skin cancer spreads through a lymph vessel and begins to grow more than 2 centimeters away from the primary tumor but before it reaches the nearest lymph node."
The investigators used data from a prospective database of patients with in-transit melanomas to take a retrospective look at complete responders to either of the two isolated limb therapies. They compared patterns of recurrence and effects on outcomes between the two modalities. In all, 81 patients had first-time HILP and 133 had first-time ILI.
Among 36 patients with a complete response to HILP, 24 had recurrences. Of these patients, 11 experienced in-field-only recurrences, 12 had out-of-field-only recurrences, and 1 patient had a mixed recurrence pattern.
In comparison, 28 of 37 patients with complete responses to ILI had recurrences (9 had in-field-only, 16 had out-of-field, and 3 had mixed recurrence patterns).
There were no significant differences between the perfusion or infusion therapies in time to in-field recurrence, but time to out-of-field recurrence to regional nodes was significantly longer with HILP (42 months vs. 14 months; P = .02). When the authors looked at distant out-of-field recurrences, however, the difference between the treatment types was not significant.
Overall survival after all procedures (including partial responses, stable disease, and nonresponses) was also similar among the treatment types. The overall survival rate after a complete response was higher with HILP (77% vs. 54%); the investigators described this as clinically significant, although it was not statistically significant (P = .1).
At last follow-up (median, 4.0 years for HILP and 2.5 years for ILI), 12 patients who had a complete response to HILP were alive without recurrence for a median duration of 6.5 years, and 24 had recurrences at a median of 3.3 years after perfusion therapy and received additional therapy. Among the latter group, 2 had no evidence of disease, 9 were alive with disease, and 13 died, at a median of 3.1 years.
Among the complete responders to ILI, 9 had no recurrence at a median of 2.6 years; 28 had recurrences at a median of 2.3 years, and received additional treatment. At last follow-up, 8 of the 28 had no evidence of disease (median, 3.9 years), 7 were alive with disease (median, 1.2 years), and 13 had died (median, 2.1 years).
The investigators concluded that the higher proportion of recurrences among patients with an initial complete response to ILI may be due to more frequent lymph node recurrences (nine vs. one in patients who had a complete response to HILP).
The study was supported by Roche/Schering-Plough. Dr. Sharma had no disclosures.
ORLANDO – The type of regional chemotherapy given to patients with in-transit or intralymphatic melanoma of the extremities appears to make a difference in out-of-field recurrences and time to distant recurrence, reported investigators at a symposium of the Society of Surgical Oncology.
A study of 214 patients who underwent either first-time hyperthermic isolated limb perfusion (HILP) or isolated limb infusion (ILI) for in-transit melanomas showed that HILP was associated with significantly more in-field complete responses (P = .01), and a longer median time to out-of-field recurrences, compared with ILI, said Dr. Ketan Sharma of Duke University Medical Center in Durham, NC.
"We found that perfusion complete response and infusion complete response exhibit similar degrees of in-field disease control," Dr. Sharma said.
However, "recurrent disease after a regional therapy complete response is complex, and requires a multidisciplinary approach to treatment," he added.
The National Cancer Institute defines an in-transit metastasis as a "type of metastasis in which skin cancer spreads through a lymph vessel and begins to grow more than 2 centimeters away from the primary tumor but before it reaches the nearest lymph node."
The investigators used data from a prospective database of patients with in-transit melanomas to take a retrospective look at complete responders to either of the two isolated limb therapies. They compared patterns of recurrence and effects on outcomes between the two modalities. In all, 81 patients had first-time HILP and 133 had first-time ILI.
Among 36 patients with a complete response to HILP, 24 had recurrences. Of these patients, 11 experienced in-field-only recurrences, 12 had out-of-field-only recurrences, and 1 patient had a mixed recurrence pattern.
In comparison, 28 of 37 patients with complete responses to ILI had recurrences (9 had in-field-only, 16 had out-of-field, and 3 had mixed recurrence patterns).
There were no significant differences between the perfusion or infusion therapies in time to in-field recurrence, but time to out-of-field recurrence to regional nodes was significantly longer with HILP (42 months vs. 14 months; P = .02). When the authors looked at distant out-of-field recurrences, however, the difference between the treatment types was not significant.
Overall survival after all procedures (including partial responses, stable disease, and nonresponses) was also similar among the treatment types. The overall survival rate after a complete response was higher with HILP (77% vs. 54%); the investigators described this as clinically significant, although it was not statistically significant (P = .1).
At last follow-up (median, 4.0 years for HILP and 2.5 years for ILI), 12 patients who had a complete response to HILP were alive without recurrence for a median duration of 6.5 years, and 24 had recurrences at a median of 3.3 years after perfusion therapy and received additional therapy. Among the latter group, 2 had no evidence of disease, 9 were alive with disease, and 13 died, at a median of 3.1 years.
Among the complete responders to ILI, 9 had no recurrence at a median of 2.6 years; 28 had recurrences at a median of 2.3 years, and received additional treatment. At last follow-up, 8 of the 28 had no evidence of disease (median, 3.9 years), 7 were alive with disease (median, 1.2 years), and 13 had died (median, 2.1 years).
The investigators concluded that the higher proportion of recurrences among patients with an initial complete response to ILI may be due to more frequent lymph node recurrences (nine vs. one in patients who had a complete response to HILP).
The study was supported by Roche/Schering-Plough. Dr. Sharma had no disclosures.
ORLANDO – The type of regional chemotherapy given to patients with in-transit or intralymphatic melanoma of the extremities appears to make a difference in out-of-field recurrences and time to distant recurrence, reported investigators at a symposium of the Society of Surgical Oncology.
A study of 214 patients who underwent either first-time hyperthermic isolated limb perfusion (HILP) or isolated limb infusion (ILI) for in-transit melanomas showed that HILP was associated with significantly more in-field complete responses (P = .01), and a longer median time to out-of-field recurrences, compared with ILI, said Dr. Ketan Sharma of Duke University Medical Center in Durham, NC.
"We found that perfusion complete response and infusion complete response exhibit similar degrees of in-field disease control," Dr. Sharma said.
However, "recurrent disease after a regional therapy complete response is complex, and requires a multidisciplinary approach to treatment," he added.
The National Cancer Institute defines an in-transit metastasis as a "type of metastasis in which skin cancer spreads through a lymph vessel and begins to grow more than 2 centimeters away from the primary tumor but before it reaches the nearest lymph node."
The investigators used data from a prospective database of patients with in-transit melanomas to take a retrospective look at complete responders to either of the two isolated limb therapies. They compared patterns of recurrence and effects on outcomes between the two modalities. In all, 81 patients had first-time HILP and 133 had first-time ILI.
Among 36 patients with a complete response to HILP, 24 had recurrences. Of these patients, 11 experienced in-field-only recurrences, 12 had out-of-field-only recurrences, and 1 patient had a mixed recurrence pattern.
In comparison, 28 of 37 patients with complete responses to ILI had recurrences (9 had in-field-only, 16 had out-of-field, and 3 had mixed recurrence patterns).
There were no significant differences between the perfusion or infusion therapies in time to in-field recurrence, but time to out-of-field recurrence to regional nodes was significantly longer with HILP (42 months vs. 14 months; P = .02). When the authors looked at distant out-of-field recurrences, however, the difference between the treatment types was not significant.
Overall survival after all procedures (including partial responses, stable disease, and nonresponses) was also similar among the treatment types. The overall survival rate after a complete response was higher with HILP (77% vs. 54%); the investigators described this as clinically significant, although it was not statistically significant (P = .1).
At last follow-up (median, 4.0 years for HILP and 2.5 years for ILI), 12 patients who had a complete response to HILP were alive without recurrence for a median duration of 6.5 years, and 24 had recurrences at a median of 3.3 years after perfusion therapy and received additional therapy. Among the latter group, 2 had no evidence of disease, 9 were alive with disease, and 13 died, at a median of 3.1 years.
Among the complete responders to ILI, 9 had no recurrence at a median of 2.6 years; 28 had recurrences at a median of 2.3 years, and received additional treatment. At last follow-up, 8 of the 28 had no evidence of disease (median, 3.9 years), 7 were alive with disease (median, 1.2 years), and 13 had died (median, 2.1 years).
The investigators concluded that the higher proportion of recurrences among patients with an initial complete response to ILI may be due to more frequent lymph node recurrences (nine vs. one in patients who had a complete response to HILP).
The study was supported by Roche/Schering-Plough. Dr. Sharma had no disclosures.
FROM A SYMPOSIUM SPONSORED BY THE SOCIETY OF SURGICAL ONCOLOGY
Major Finding: Hyperthermic isolated limb perfusion was significantly better than isolated limb infusion at preventing out-of-field, in-transit melanoma recurrences (P = .02)
Data Source: Investigators conducted a retrospective review of prospective data on 214 patients with in-transit melanomas.
Disclosures: The study was supported by Roche/Schering-Plough. Dr. Sharma had no disclosures.
Melanoma on Scalp Signals Worse Prognosis Than Other Sites
ORLANDO – Malignant melanomas of the scalp behave differently from melanomas arising at other body sites, and are associated with poor disease-free and overall survival compared with other head and neck melanomas, investigators reported here.
A retrospective study of more than 11,000 patients with malignant melanomas showed that 5-year melanoma-specific survival was 65% for patients with lesions on the scalp, compared with 78% for patients with tumors on the trunk or elsewhere on the head, face, neck, or ear (P = .0003), said Dr. Junko Ozao-Choy, a fellow at the John Wayne Cancer Institute in Santa Monica, Calif.
Five-year overall survival for patients with melanomas of the scalp was 58%, compared with 72% for those with head, face, neck, or ear lesions, 74% for those with trunk lesions, and 77% for those with tumors on an extremity (P less than .0001), Dr. Ozao-Choy reported at a symposium sponsored by the Society of Surgical Oncology.
Melanomas of the scalp may account for the poor prognosis of head and neck melanoma relative to tumors originating at other body sites, Dr. Ozao-Choy and her colleagues suggested.
"Scalp melanomas may warrant further studies to ascertain whether biology or anatomy contributes to their worse clinical course," she said, adding that the results indicate "scalp melanomas may need closer clinical follow-up."
Compared with melanomas originating at other body sites, scalp melanomas tend to occur in older patients, predominantly men, according to the investigators. The lesions tend to have higher Breslow thickness, advanced nodal stage and overall stage, and more ulceration.
Dr. Ozao-Choy and her colleagues based their findings on a database review of 11,396 patients presenting for treatment within 4 months of diagnosis from 1971 through 2010. In univariate analysis controlling for sex, they found that 80% of the 799 patients with melanoma originating on the scalp were men (P = .0001).
The mean age at presentation was 54 years for those with scalp lesions and 55 for those with head, neck, or ear tumors. Taken together, the mean age at diagnosis for patients with scalp and head melanomas was higher than for patients with lesions on the trunk (age 47 years) or extremities (age 51 years, P less than .0001).
Scalp tumors had greater Breslow thickness, at a mean of 2.5 mm compared with 1.7 mm for other head and neck melanomas, 1.8 mm for trunk tumors, and 1.9 mm for lesions on an extremity (P less than .0001).
Looking at 5-year overall survival by stage, the authors found that patients with stage I/II scalp lesions had worse survival than those with stage I/II lesions at other sites (P less than .0001). Similarly, stage III scalp primary tumors were associated with worse survival than other stage III tumors (P = .009).
Multivariate analysis controlling for age, male sex, Breslow thickness, lymph node status, and ulceration revealed that patients with scalp tumors had worse 5-year disease-free survival, at 47%, compared with 61% for other head and neck tumors, 66% for trunk tumors, and 69% for extremity melanomas (hazard ratio, 1.8; P less than .0001).
In the question and answer session, an audience member commented that the worse prognosis for head and neck melanomas may be related to the greater frequency of aggressive NRAS and BRAF mutations in tumors originating at those sites.
The study was internally funded. The authors had no disclosures.
ORLANDO – Malignant melanomas of the scalp behave differently from melanomas arising at other body sites, and are associated with poor disease-free and overall survival compared with other head and neck melanomas, investigators reported here.
A retrospective study of more than 11,000 patients with malignant melanomas showed that 5-year melanoma-specific survival was 65% for patients with lesions on the scalp, compared with 78% for patients with tumors on the trunk or elsewhere on the head, face, neck, or ear (P = .0003), said Dr. Junko Ozao-Choy, a fellow at the John Wayne Cancer Institute in Santa Monica, Calif.
Five-year overall survival for patients with melanomas of the scalp was 58%, compared with 72% for those with head, face, neck, or ear lesions, 74% for those with trunk lesions, and 77% for those with tumors on an extremity (P less than .0001), Dr. Ozao-Choy reported at a symposium sponsored by the Society of Surgical Oncology.
Melanomas of the scalp may account for the poor prognosis of head and neck melanoma relative to tumors originating at other body sites, Dr. Ozao-Choy and her colleagues suggested.
"Scalp melanomas may warrant further studies to ascertain whether biology or anatomy contributes to their worse clinical course," she said, adding that the results indicate "scalp melanomas may need closer clinical follow-up."
Compared with melanomas originating at other body sites, scalp melanomas tend to occur in older patients, predominantly men, according to the investigators. The lesions tend to have higher Breslow thickness, advanced nodal stage and overall stage, and more ulceration.
Dr. Ozao-Choy and her colleagues based their findings on a database review of 11,396 patients presenting for treatment within 4 months of diagnosis from 1971 through 2010. In univariate analysis controlling for sex, they found that 80% of the 799 patients with melanoma originating on the scalp were men (P = .0001).
The mean age at presentation was 54 years for those with scalp lesions and 55 for those with head, neck, or ear tumors. Taken together, the mean age at diagnosis for patients with scalp and head melanomas was higher than for patients with lesions on the trunk (age 47 years) or extremities (age 51 years, P less than .0001).
Scalp tumors had greater Breslow thickness, at a mean of 2.5 mm compared with 1.7 mm for other head and neck melanomas, 1.8 mm for trunk tumors, and 1.9 mm for lesions on an extremity (P less than .0001).
Looking at 5-year overall survival by stage, the authors found that patients with stage I/II scalp lesions had worse survival than those with stage I/II lesions at other sites (P less than .0001). Similarly, stage III scalp primary tumors were associated with worse survival than other stage III tumors (P = .009).
Multivariate analysis controlling for age, male sex, Breslow thickness, lymph node status, and ulceration revealed that patients with scalp tumors had worse 5-year disease-free survival, at 47%, compared with 61% for other head and neck tumors, 66% for trunk tumors, and 69% for extremity melanomas (hazard ratio, 1.8; P less than .0001).
In the question and answer session, an audience member commented that the worse prognosis for head and neck melanomas may be related to the greater frequency of aggressive NRAS and BRAF mutations in tumors originating at those sites.
The study was internally funded. The authors had no disclosures.
ORLANDO – Malignant melanomas of the scalp behave differently from melanomas arising at other body sites, and are associated with poor disease-free and overall survival compared with other head and neck melanomas, investigators reported here.
A retrospective study of more than 11,000 patients with malignant melanomas showed that 5-year melanoma-specific survival was 65% for patients with lesions on the scalp, compared with 78% for patients with tumors on the trunk or elsewhere on the head, face, neck, or ear (P = .0003), said Dr. Junko Ozao-Choy, a fellow at the John Wayne Cancer Institute in Santa Monica, Calif.
Five-year overall survival for patients with melanomas of the scalp was 58%, compared with 72% for those with head, face, neck, or ear lesions, 74% for those with trunk lesions, and 77% for those with tumors on an extremity (P less than .0001), Dr. Ozao-Choy reported at a symposium sponsored by the Society of Surgical Oncology.
Melanomas of the scalp may account for the poor prognosis of head and neck melanoma relative to tumors originating at other body sites, Dr. Ozao-Choy and her colleagues suggested.
"Scalp melanomas may warrant further studies to ascertain whether biology or anatomy contributes to their worse clinical course," she said, adding that the results indicate "scalp melanomas may need closer clinical follow-up."
Compared with melanomas originating at other body sites, scalp melanomas tend to occur in older patients, predominantly men, according to the investigators. The lesions tend to have higher Breslow thickness, advanced nodal stage and overall stage, and more ulceration.
Dr. Ozao-Choy and her colleagues based their findings on a database review of 11,396 patients presenting for treatment within 4 months of diagnosis from 1971 through 2010. In univariate analysis controlling for sex, they found that 80% of the 799 patients with melanoma originating on the scalp were men (P = .0001).
The mean age at presentation was 54 years for those with scalp lesions and 55 for those with head, neck, or ear tumors. Taken together, the mean age at diagnosis for patients with scalp and head melanomas was higher than for patients with lesions on the trunk (age 47 years) or extremities (age 51 years, P less than .0001).
Scalp tumors had greater Breslow thickness, at a mean of 2.5 mm compared with 1.7 mm for other head and neck melanomas, 1.8 mm for trunk tumors, and 1.9 mm for lesions on an extremity (P less than .0001).
Looking at 5-year overall survival by stage, the authors found that patients with stage I/II scalp lesions had worse survival than those with stage I/II lesions at other sites (P less than .0001). Similarly, stage III scalp primary tumors were associated with worse survival than other stage III tumors (P = .009).
Multivariate analysis controlling for age, male sex, Breslow thickness, lymph node status, and ulceration revealed that patients with scalp tumors had worse 5-year disease-free survival, at 47%, compared with 61% for other head and neck tumors, 66% for trunk tumors, and 69% for extremity melanomas (hazard ratio, 1.8; P less than .0001).
In the question and answer session, an audience member commented that the worse prognosis for head and neck melanomas may be related to the greater frequency of aggressive NRAS and BRAF mutations in tumors originating at those sites.
The study was internally funded. The authors had no disclosures.
FROM A SYMPOSIUM SPONSORED BY THE SOCIETY OF SURGICAL ONCOLOGY
Major Finding: Compared with melanomas originating at other body sites, scalp melanomas are associated with worse 5-year melanoma-specific survival (P = .0003), and overall survival (P less than .0001)
Data Source: Investigators conducted a data review on 11,396 patients with malignant melanoma.
Disclosures: The study was internally funded. The authors had no disclosures.
Extramammary Paget's Needs More Than Mohs
WAIKOLOA, HAWAII – Extramammary Paget’s disease poses a particular challenge because of its multifocal/multicentric nature.
"What you see with extramammary Paget’s is not necessarily what you get," Dr. Theodore Rosen cautioned at the Hawaii Dermatology Seminar sponsored by Skin Disease Education Foundation (SDEF).
This is an uncommon neoplasia that’s typically described as an erythematous, erosive, itchy patch or plaque having a strawberries-and-cream appearance. Yet there may be other areas of subclinical involvement at a distance from the obvious lesion.
"That’s why Mohs surgery for this condition may be difficult without something being done in advance," said Dr. Rosen, professor of dermatology at Baylor College of Medicine, Houston.
He suggested applying topical 5% imiquimod or 5-fluorouracil to identify all of the active foci by lighting up the affected areas to allow more precise surgery or ablative therapy.
Dr. Rosen does not, however, recommend using imiquimod as primary therapy. He noted that a review of 27 published cases of 5% imiquimod for the treatment of extramammary Paget’s disease reported a 22% failure rate (Arch. Dermatol. 2011;147:704-8).
Mohs surgery shows promise for treatment of extramammary Paget’s disease, with lower recurrence rates than reported for wide surgical excision. However, experience to date with Mohs surgery for extensive disease is limited. For this reason, most authorities still consider wide surgical excision the gold standard therapy for extramammary Paget’s disease, despite published recurrence rates of 42%-54% even with clear surgical margins. Use of preoperative topical 5-fluorouracil or 5% imiquimod should substantially reduce those high recurrence rates, the dermatologist said.
Extramammary Paget’s disease is typically slow-growing for a decade or more before invading the dermis, at which point it quickly becomes widely metastatic.
"Once extramammary Paget’s disease has broken through the dermal/epidermal junction, it becomes a very nasty, bad-acting disease," Dr. Rosen said.
It’s well recognized that extramammary Paget’s is associated with an increased risk of underlying internal malignancy of the lower gastrointestinal or genitourinary tract. This increased risk is typically described as being in the 10%-20% range. But that figure may be too low. A recent report from investigators at Houston’s M.D. Anderson Cancer Center involving 20 consecutive patients with extramammary Paget’s on the penis or scrotum indicated that 8 of them – fully 40% – had an associated underlying internal adenocarcinoma (J. Urol. 2011;186:97-102).
The risk of underlying internal malignancy is known to be considerably greater in white patients than in Asians. It’s very uncommon for black patients with extramammary Paget’s to have an associated internal malignancy.
Because the full workup for an associated occult internal adenocarcinoma is elaborate and costly, a means of determining which patients are at greater or lesser risk would be welcome in order to guide the extent of testing. Cytokeratin staining may be the solution. Spanish dermatologists have reported that cutaneous extramammary Paget’s disease is characteristically positive for cytokeratin 7, negative for cytokeratin 20, and positive for cystic disease fluid protein 15.
In contrast, endodermal extramammary Paget’s, which is more strongly associated with internal malignancy, is positive for cytokeratin 7 and 20 and negative for cystic disease fluid protein 15, according to the investigators (Clin. Exp. Dermatol. 2008;33:595-8). A cautionary note: Dr. Rosen said that to his knowledge these findings haven’t yet been confirmed by other groups.
He reported having no financial conflicts.
SDEF and this news organization are owned by Elsevier.
WAIKOLOA, HAWAII – Extramammary Paget’s disease poses a particular challenge because of its multifocal/multicentric nature.
"What you see with extramammary Paget’s is not necessarily what you get," Dr. Theodore Rosen cautioned at the Hawaii Dermatology Seminar sponsored by Skin Disease Education Foundation (SDEF).
This is an uncommon neoplasia that’s typically described as an erythematous, erosive, itchy patch or plaque having a strawberries-and-cream appearance. Yet there may be other areas of subclinical involvement at a distance from the obvious lesion.
"That’s why Mohs surgery for this condition may be difficult without something being done in advance," said Dr. Rosen, professor of dermatology at Baylor College of Medicine, Houston.
He suggested applying topical 5% imiquimod or 5-fluorouracil to identify all of the active foci by lighting up the affected areas to allow more precise surgery or ablative therapy.
Dr. Rosen does not, however, recommend using imiquimod as primary therapy. He noted that a review of 27 published cases of 5% imiquimod for the treatment of extramammary Paget’s disease reported a 22% failure rate (Arch. Dermatol. 2011;147:704-8).
Mohs surgery shows promise for treatment of extramammary Paget’s disease, with lower recurrence rates than reported for wide surgical excision. However, experience to date with Mohs surgery for extensive disease is limited. For this reason, most authorities still consider wide surgical excision the gold standard therapy for extramammary Paget’s disease, despite published recurrence rates of 42%-54% even with clear surgical margins. Use of preoperative topical 5-fluorouracil or 5% imiquimod should substantially reduce those high recurrence rates, the dermatologist said.
Extramammary Paget’s disease is typically slow-growing for a decade or more before invading the dermis, at which point it quickly becomes widely metastatic.
"Once extramammary Paget’s disease has broken through the dermal/epidermal junction, it becomes a very nasty, bad-acting disease," Dr. Rosen said.
It’s well recognized that extramammary Paget’s is associated with an increased risk of underlying internal malignancy of the lower gastrointestinal or genitourinary tract. This increased risk is typically described as being in the 10%-20% range. But that figure may be too low. A recent report from investigators at Houston’s M.D. Anderson Cancer Center involving 20 consecutive patients with extramammary Paget’s on the penis or scrotum indicated that 8 of them – fully 40% – had an associated underlying internal adenocarcinoma (J. Urol. 2011;186:97-102).
The risk of underlying internal malignancy is known to be considerably greater in white patients than in Asians. It’s very uncommon for black patients with extramammary Paget’s to have an associated internal malignancy.
Because the full workup for an associated occult internal adenocarcinoma is elaborate and costly, a means of determining which patients are at greater or lesser risk would be welcome in order to guide the extent of testing. Cytokeratin staining may be the solution. Spanish dermatologists have reported that cutaneous extramammary Paget’s disease is characteristically positive for cytokeratin 7, negative for cytokeratin 20, and positive for cystic disease fluid protein 15.
In contrast, endodermal extramammary Paget’s, which is more strongly associated with internal malignancy, is positive for cytokeratin 7 and 20 and negative for cystic disease fluid protein 15, according to the investigators (Clin. Exp. Dermatol. 2008;33:595-8). A cautionary note: Dr. Rosen said that to his knowledge these findings haven’t yet been confirmed by other groups.
He reported having no financial conflicts.
SDEF and this news organization are owned by Elsevier.
WAIKOLOA, HAWAII – Extramammary Paget’s disease poses a particular challenge because of its multifocal/multicentric nature.
"What you see with extramammary Paget’s is not necessarily what you get," Dr. Theodore Rosen cautioned at the Hawaii Dermatology Seminar sponsored by Skin Disease Education Foundation (SDEF).
This is an uncommon neoplasia that’s typically described as an erythematous, erosive, itchy patch or plaque having a strawberries-and-cream appearance. Yet there may be other areas of subclinical involvement at a distance from the obvious lesion.
"That’s why Mohs surgery for this condition may be difficult without something being done in advance," said Dr. Rosen, professor of dermatology at Baylor College of Medicine, Houston.
He suggested applying topical 5% imiquimod or 5-fluorouracil to identify all of the active foci by lighting up the affected areas to allow more precise surgery or ablative therapy.
Dr. Rosen does not, however, recommend using imiquimod as primary therapy. He noted that a review of 27 published cases of 5% imiquimod for the treatment of extramammary Paget’s disease reported a 22% failure rate (Arch. Dermatol. 2011;147:704-8).
Mohs surgery shows promise for treatment of extramammary Paget’s disease, with lower recurrence rates than reported for wide surgical excision. However, experience to date with Mohs surgery for extensive disease is limited. For this reason, most authorities still consider wide surgical excision the gold standard therapy for extramammary Paget’s disease, despite published recurrence rates of 42%-54% even with clear surgical margins. Use of preoperative topical 5-fluorouracil or 5% imiquimod should substantially reduce those high recurrence rates, the dermatologist said.
Extramammary Paget’s disease is typically slow-growing for a decade or more before invading the dermis, at which point it quickly becomes widely metastatic.
"Once extramammary Paget’s disease has broken through the dermal/epidermal junction, it becomes a very nasty, bad-acting disease," Dr. Rosen said.
It’s well recognized that extramammary Paget’s is associated with an increased risk of underlying internal malignancy of the lower gastrointestinal or genitourinary tract. This increased risk is typically described as being in the 10%-20% range. But that figure may be too low. A recent report from investigators at Houston’s M.D. Anderson Cancer Center involving 20 consecutive patients with extramammary Paget’s on the penis or scrotum indicated that 8 of them – fully 40% – had an associated underlying internal adenocarcinoma (J. Urol. 2011;186:97-102).
The risk of underlying internal malignancy is known to be considerably greater in white patients than in Asians. It’s very uncommon for black patients with extramammary Paget’s to have an associated internal malignancy.
Because the full workup for an associated occult internal adenocarcinoma is elaborate and costly, a means of determining which patients are at greater or lesser risk would be welcome in order to guide the extent of testing. Cytokeratin staining may be the solution. Spanish dermatologists have reported that cutaneous extramammary Paget’s disease is characteristically positive for cytokeratin 7, negative for cytokeratin 20, and positive for cystic disease fluid protein 15.
In contrast, endodermal extramammary Paget’s, which is more strongly associated with internal malignancy, is positive for cytokeratin 7 and 20 and negative for cystic disease fluid protein 15, according to the investigators (Clin. Exp. Dermatol. 2008;33:595-8). A cautionary note: Dr. Rosen said that to his knowledge these findings haven’t yet been confirmed by other groups.
He reported having no financial conflicts.
SDEF and this news organization are owned by Elsevier.
EXPERT ANALYSIS FROM THE SDEF HAWAII DERMATOLOGY SEMINAR
Bexarotene Confers No Survival Benefit for Mycosis Fungoides
SAN DIEGO – The median survival for patients with tumor stage mycosis fungoides who received oral bexarotene therapy was 3.3 years, compared with a median of 7.7 years for patients who did not receive the drug, results from a long-term, single-center study demonstrated.
The finding "was not intuitive, because approximately 54% of patients who took bexarotene responded to the drug, but it had a negative impact on survival," Dr. John A. Zic said in an interview following a poster session at the annual meeting of the American Academy of Dermatology, where the study was presented.
Mycosis fungoides accounts for the majority of cutaneous T-cell lymphoma cases, yet no randomized controlled trials exist to compare existing therapies head to head. "Therefore, it is important to gather long-term clinical data for retrospective analysis when these patient cohorts exist to analyze how different therapies contribute to patient outcome," Dr. Zic and his associates wrote in their poster abstract.
For the current study, the researchers reviewed data from 39 patients with tumor stage mycosis fungoides who were followed at the Vanderbilt University Cutaneous Lymphoma Clinic in Nashville, Tenn., between July of 1995 and July of 2010. They set out to determine if patients who received therapy with oral bexarotene for the treatment of tumor stage mycosis fungoides had improved outcome, compared with those who did not take the drug. Bexarotene is a synthetic retinoid approved by the Food and Drug Administration in 1999 for the treatment of refractory, advanced-stage cutaneous T-cell lymphoma, including mycosis fungoides.
Of the 39 patients 27 (69%) were male. More than half of patients (67%) received oral bexarotene while 33% did not. Patients in the bexarotene group were older than those who did not receive the drug (a mean of 61 vs. 56 years, respectively), and a higher proportion had late clinical stage disease at diagnosis (19 patients vs. 10 patinets). They were also more likely to have large cell transformation, "which is a negative prognostic indicator," said Dr. Zic, associate professor of dermatology at Vanderbilt University.
He went on to report that 54% of patients in the bexarotene group achieved durable response, which was defined as a greater than 50% clearing for at least 1 month. However, the median overall survival for patients in the bexarotene group was 3.3 years, compared with 7.7 years for patients who did not receive the drug.
The researchers also found that patients who were diagnosed with mycosis fungoides before the year 2000 "seemed to have a longer survival than patients who were diagnosed after 2000," Dr. Zic said. "You would think that with the introduction of newer therapies we might be able to impact survival in the past decade versus survival two decades ago. We didn’t find that, and we’re not sure why. However, the more recently enrolled patients appear to be sicker; they have higher stages of disease and more [large cell] transformation. That might explain the difference."
Dr. Zic acknowledged that a chief limitation of the study was its retrospective design. "There could be certain biases introduced such as selection bias and referral bias that might help to explain some of the differences that were seen," he said.
Further analyses of the patients are planned.
Dr. Zic said that he had no relevant financial disclosures.
SAN DIEGO – The median survival for patients with tumor stage mycosis fungoides who received oral bexarotene therapy was 3.3 years, compared with a median of 7.7 years for patients who did not receive the drug, results from a long-term, single-center study demonstrated.
The finding "was not intuitive, because approximately 54% of patients who took bexarotene responded to the drug, but it had a negative impact on survival," Dr. John A. Zic said in an interview following a poster session at the annual meeting of the American Academy of Dermatology, where the study was presented.
Mycosis fungoides accounts for the majority of cutaneous T-cell lymphoma cases, yet no randomized controlled trials exist to compare existing therapies head to head. "Therefore, it is important to gather long-term clinical data for retrospective analysis when these patient cohorts exist to analyze how different therapies contribute to patient outcome," Dr. Zic and his associates wrote in their poster abstract.
For the current study, the researchers reviewed data from 39 patients with tumor stage mycosis fungoides who were followed at the Vanderbilt University Cutaneous Lymphoma Clinic in Nashville, Tenn., between July of 1995 and July of 2010. They set out to determine if patients who received therapy with oral bexarotene for the treatment of tumor stage mycosis fungoides had improved outcome, compared with those who did not take the drug. Bexarotene is a synthetic retinoid approved by the Food and Drug Administration in 1999 for the treatment of refractory, advanced-stage cutaneous T-cell lymphoma, including mycosis fungoides.
Of the 39 patients 27 (69%) were male. More than half of patients (67%) received oral bexarotene while 33% did not. Patients in the bexarotene group were older than those who did not receive the drug (a mean of 61 vs. 56 years, respectively), and a higher proportion had late clinical stage disease at diagnosis (19 patients vs. 10 patinets). They were also more likely to have large cell transformation, "which is a negative prognostic indicator," said Dr. Zic, associate professor of dermatology at Vanderbilt University.
He went on to report that 54% of patients in the bexarotene group achieved durable response, which was defined as a greater than 50% clearing for at least 1 month. However, the median overall survival for patients in the bexarotene group was 3.3 years, compared with 7.7 years for patients who did not receive the drug.
The researchers also found that patients who were diagnosed with mycosis fungoides before the year 2000 "seemed to have a longer survival than patients who were diagnosed after 2000," Dr. Zic said. "You would think that with the introduction of newer therapies we might be able to impact survival in the past decade versus survival two decades ago. We didn’t find that, and we’re not sure why. However, the more recently enrolled patients appear to be sicker; they have higher stages of disease and more [large cell] transformation. That might explain the difference."
Dr. Zic acknowledged that a chief limitation of the study was its retrospective design. "There could be certain biases introduced such as selection bias and referral bias that might help to explain some of the differences that were seen," he said.
Further analyses of the patients are planned.
Dr. Zic said that he had no relevant financial disclosures.
SAN DIEGO – The median survival for patients with tumor stage mycosis fungoides who received oral bexarotene therapy was 3.3 years, compared with a median of 7.7 years for patients who did not receive the drug, results from a long-term, single-center study demonstrated.
The finding "was not intuitive, because approximately 54% of patients who took bexarotene responded to the drug, but it had a negative impact on survival," Dr. John A. Zic said in an interview following a poster session at the annual meeting of the American Academy of Dermatology, where the study was presented.
Mycosis fungoides accounts for the majority of cutaneous T-cell lymphoma cases, yet no randomized controlled trials exist to compare existing therapies head to head. "Therefore, it is important to gather long-term clinical data for retrospective analysis when these patient cohorts exist to analyze how different therapies contribute to patient outcome," Dr. Zic and his associates wrote in their poster abstract.
For the current study, the researchers reviewed data from 39 patients with tumor stage mycosis fungoides who were followed at the Vanderbilt University Cutaneous Lymphoma Clinic in Nashville, Tenn., between July of 1995 and July of 2010. They set out to determine if patients who received therapy with oral bexarotene for the treatment of tumor stage mycosis fungoides had improved outcome, compared with those who did not take the drug. Bexarotene is a synthetic retinoid approved by the Food and Drug Administration in 1999 for the treatment of refractory, advanced-stage cutaneous T-cell lymphoma, including mycosis fungoides.
Of the 39 patients 27 (69%) were male. More than half of patients (67%) received oral bexarotene while 33% did not. Patients in the bexarotene group were older than those who did not receive the drug (a mean of 61 vs. 56 years, respectively), and a higher proportion had late clinical stage disease at diagnosis (19 patients vs. 10 patinets). They were also more likely to have large cell transformation, "which is a negative prognostic indicator," said Dr. Zic, associate professor of dermatology at Vanderbilt University.
He went on to report that 54% of patients in the bexarotene group achieved durable response, which was defined as a greater than 50% clearing for at least 1 month. However, the median overall survival for patients in the bexarotene group was 3.3 years, compared with 7.7 years for patients who did not receive the drug.
The researchers also found that patients who were diagnosed with mycosis fungoides before the year 2000 "seemed to have a longer survival than patients who were diagnosed after 2000," Dr. Zic said. "You would think that with the introduction of newer therapies we might be able to impact survival in the past decade versus survival two decades ago. We didn’t find that, and we’re not sure why. However, the more recently enrolled patients appear to be sicker; they have higher stages of disease and more [large cell] transformation. That might explain the difference."
Dr. Zic acknowledged that a chief limitation of the study was its retrospective design. "There could be certain biases introduced such as selection bias and referral bias that might help to explain some of the differences that were seen," he said.
Further analyses of the patients are planned.
Dr. Zic said that he had no relevant financial disclosures.
FROM THE ANNUAL MEETING OF THE AMERICAN ACADEMY OF DERMATOLOGY
Major Finding: The median survival time for patients with tumor stage mycosis fungoides who received oral bexarotene therapy was 3.3 years, compared with a median of 7.7 years for patients who did not receive the drug.
Data Source: A retrospective analysis of 39 patients with tumor stage mycosis fungoides who were followed at the Vanderbilt University Cutaneous Lymphoma Clinic between July of 1995 and July of 2010.
Disclosures: Dr. Zic said that he had no relevant financial conflicts to disclose.
Study Results Support Ingenol Mebutate's AK Effectiveness
Topical ingenol mebutate gel effectively treated actinic keratoses when applied to the face, scalp, trunk, or extremities for 2-3 days, according to a report in the March 15 issue of the New England Journal of Medicine.
Compared with existing therapies for actinic keratoses, the chief advantage of ingenol mebutate gel is the short exposure time, reported Dr. Mark Lebwohl, professor and chairman of the department of dermatology at Mount Sinai School of Medicine, New York, and his associates. This allows for relatively rapid resolution of local reactions, and it will likely improve adherence to treatment, which in turn should improve the therapy’s effectiveness, they noted.
"Many patients find it difficult to adhere to the currently available regimens of topical treatment that last for periods of 1-4 months, which may result in ‘real-world’ effectiveness lower than that achieved in supervised and patient-compensated clinical trials," Dr. Lebwohl and his colleagues wrote.
The researchers evaluated the safety and effectiveness of ingenol mebutate gel compared with a placebo in a manufacturer-sponsored, randomized, double-blind trial of 1,005 patients treated at four medical centers. The majority of the patients had Fitzpatrick type I or II skin, and their mean age was 65 years.
Approximately half of the study patients had a history of skin cancer, and 75% had already had their keratosis treated with cryotherapy, imiquimod, or topical fluorouracil.
In each patient, a 25-cm contiguous field containing at least four to eight clinically typical and discrete actinic keratoses was selected for treatment. The study patients were divided into two groups according to the location of the treated area: on the head (face or scalp), and on the body (trunk or extremities).
Those with facial or scalp lesions were randomly assigned to self-apply either 0.015% active gel (277 patients) or placebo gel (270 patients) to the area once daily for 3 consecutive days. Those with trunk or extremity lesions were randomly assigned to self-apply 0.05% active gel (226 patients) or placebo gel (232 patients) to the area once daily for 2 consecutive days.
The primary end point was complete clearance of all clinically visible actinic keratoses in the treatment area on day 57.
Among patients with facial or scalp lesions, 42.2% of those who used active gel reached this end point, compared with only 3.7% of those who used placebo gel. Among patients with trunk or extremity lesions, 34.1% who used active gel reached this end point, compared with only 4.7% of those who used placebo gel.
In addition, patients with face or scalp lesions that were treated with ingenol mebutate showed a median reduction of 83% in the number of actinic keratoses, compared with a 0% reduction with placebo gel. "We calculated that the number of patients who needed to be treated with ingenol mebutate to obtain complete clearance in one patient was 2.6," the investigators reported (N. Engl. J. Med. 2012;366:1010-19).
Patients with trunk or extremity lesions treated with ingenol mebutate showed a median reduction of 75% in the number of actinic keratoses, compared with a 0% reduction with placebo gel. "We calculated that the number of patients who would need to be treated with ingenol mebutate to obtain complete clearance in one patient was 3.4," the researchers noted.
A secondary end point was partial (75% or more) clearance in the number of clinically visible actinic keratoses in the treatment area on day 57.
Among patients with facial or scalp lesions, 63.9% of those who used active gel reached this end point, compared with only 7.4% of those who used placebo gel. Among patients with trunk or extremity lesions, 49.1% of those who used active gel reached this end point, compared with only 6.9% of those who used placebo gel.
Patients who used ingenol mebutate showed minimal scarring or change in pigmentation. Local reactions such as erythema, crusting, swelling, vesiculation, or pustulation were common, but were mild to moderate in intensity; they peaked within a few days of treatment and resolved rapidly afterward, with no sequelae. One patient developed eye pain, burning, and periorbital edema related to ingenol mebutate and dropped out of the study, Dr. Lebwohl and his associates noted.
"Future studies are needed to assess the risks and benefits of treating larger areas of skin, using multiple treatments in the same area, and using combination therapies," they added.
This study was funded by LEO Pharma, maker of ingenol mebutate. Dr. Lebwohl reported ties to LEO Pharma, Graceway, PharmaDerm, and Peplin LTD.
Topical ingenol mebutate gel effectively treated actinic keratoses when applied to the face, scalp, trunk, or extremities for 2-3 days, according to a report in the March 15 issue of the New England Journal of Medicine.
Compared with existing therapies for actinic keratoses, the chief advantage of ingenol mebutate gel is the short exposure time, reported Dr. Mark Lebwohl, professor and chairman of the department of dermatology at Mount Sinai School of Medicine, New York, and his associates. This allows for relatively rapid resolution of local reactions, and it will likely improve adherence to treatment, which in turn should improve the therapy’s effectiveness, they noted.
"Many patients find it difficult to adhere to the currently available regimens of topical treatment that last for periods of 1-4 months, which may result in ‘real-world’ effectiveness lower than that achieved in supervised and patient-compensated clinical trials," Dr. Lebwohl and his colleagues wrote.
The researchers evaluated the safety and effectiveness of ingenol mebutate gel compared with a placebo in a manufacturer-sponsored, randomized, double-blind trial of 1,005 patients treated at four medical centers. The majority of the patients had Fitzpatrick type I or II skin, and their mean age was 65 years.
Approximately half of the study patients had a history of skin cancer, and 75% had already had their keratosis treated with cryotherapy, imiquimod, or topical fluorouracil.
In each patient, a 25-cm contiguous field containing at least four to eight clinically typical and discrete actinic keratoses was selected for treatment. The study patients were divided into two groups according to the location of the treated area: on the head (face or scalp), and on the body (trunk or extremities).
Those with facial or scalp lesions were randomly assigned to self-apply either 0.015% active gel (277 patients) or placebo gel (270 patients) to the area once daily for 3 consecutive days. Those with trunk or extremity lesions were randomly assigned to self-apply 0.05% active gel (226 patients) or placebo gel (232 patients) to the area once daily for 2 consecutive days.
The primary end point was complete clearance of all clinically visible actinic keratoses in the treatment area on day 57.
Among patients with facial or scalp lesions, 42.2% of those who used active gel reached this end point, compared with only 3.7% of those who used placebo gel. Among patients with trunk or extremity lesions, 34.1% who used active gel reached this end point, compared with only 4.7% of those who used placebo gel.
In addition, patients with face or scalp lesions that were treated with ingenol mebutate showed a median reduction of 83% in the number of actinic keratoses, compared with a 0% reduction with placebo gel. "We calculated that the number of patients who needed to be treated with ingenol mebutate to obtain complete clearance in one patient was 2.6," the investigators reported (N. Engl. J. Med. 2012;366:1010-19).
Patients with trunk or extremity lesions treated with ingenol mebutate showed a median reduction of 75% in the number of actinic keratoses, compared with a 0% reduction with placebo gel. "We calculated that the number of patients who would need to be treated with ingenol mebutate to obtain complete clearance in one patient was 3.4," the researchers noted.
A secondary end point was partial (75% or more) clearance in the number of clinically visible actinic keratoses in the treatment area on day 57.
Among patients with facial or scalp lesions, 63.9% of those who used active gel reached this end point, compared with only 7.4% of those who used placebo gel. Among patients with trunk or extremity lesions, 49.1% of those who used active gel reached this end point, compared with only 6.9% of those who used placebo gel.
Patients who used ingenol mebutate showed minimal scarring or change in pigmentation. Local reactions such as erythema, crusting, swelling, vesiculation, or pustulation were common, but were mild to moderate in intensity; they peaked within a few days of treatment and resolved rapidly afterward, with no sequelae. One patient developed eye pain, burning, and periorbital edema related to ingenol mebutate and dropped out of the study, Dr. Lebwohl and his associates noted.
"Future studies are needed to assess the risks and benefits of treating larger areas of skin, using multiple treatments in the same area, and using combination therapies," they added.
This study was funded by LEO Pharma, maker of ingenol mebutate. Dr. Lebwohl reported ties to LEO Pharma, Graceway, PharmaDerm, and Peplin LTD.
Topical ingenol mebutate gel effectively treated actinic keratoses when applied to the face, scalp, trunk, or extremities for 2-3 days, according to a report in the March 15 issue of the New England Journal of Medicine.
Compared with existing therapies for actinic keratoses, the chief advantage of ingenol mebutate gel is the short exposure time, reported Dr. Mark Lebwohl, professor and chairman of the department of dermatology at Mount Sinai School of Medicine, New York, and his associates. This allows for relatively rapid resolution of local reactions, and it will likely improve adherence to treatment, which in turn should improve the therapy’s effectiveness, they noted.
"Many patients find it difficult to adhere to the currently available regimens of topical treatment that last for periods of 1-4 months, which may result in ‘real-world’ effectiveness lower than that achieved in supervised and patient-compensated clinical trials," Dr. Lebwohl and his colleagues wrote.
The researchers evaluated the safety and effectiveness of ingenol mebutate gel compared with a placebo in a manufacturer-sponsored, randomized, double-blind trial of 1,005 patients treated at four medical centers. The majority of the patients had Fitzpatrick type I or II skin, and their mean age was 65 years.
Approximately half of the study patients had a history of skin cancer, and 75% had already had their keratosis treated with cryotherapy, imiquimod, or topical fluorouracil.
In each patient, a 25-cm contiguous field containing at least four to eight clinically typical and discrete actinic keratoses was selected for treatment. The study patients were divided into two groups according to the location of the treated area: on the head (face or scalp), and on the body (trunk or extremities).
Those with facial or scalp lesions were randomly assigned to self-apply either 0.015% active gel (277 patients) or placebo gel (270 patients) to the area once daily for 3 consecutive days. Those with trunk or extremity lesions were randomly assigned to self-apply 0.05% active gel (226 patients) or placebo gel (232 patients) to the area once daily for 2 consecutive days.
The primary end point was complete clearance of all clinically visible actinic keratoses in the treatment area on day 57.
Among patients with facial or scalp lesions, 42.2% of those who used active gel reached this end point, compared with only 3.7% of those who used placebo gel. Among patients with trunk or extremity lesions, 34.1% who used active gel reached this end point, compared with only 4.7% of those who used placebo gel.
In addition, patients with face or scalp lesions that were treated with ingenol mebutate showed a median reduction of 83% in the number of actinic keratoses, compared with a 0% reduction with placebo gel. "We calculated that the number of patients who needed to be treated with ingenol mebutate to obtain complete clearance in one patient was 2.6," the investigators reported (N. Engl. J. Med. 2012;366:1010-19).
Patients with trunk or extremity lesions treated with ingenol mebutate showed a median reduction of 75% in the number of actinic keratoses, compared with a 0% reduction with placebo gel. "We calculated that the number of patients who would need to be treated with ingenol mebutate to obtain complete clearance in one patient was 3.4," the researchers noted.
A secondary end point was partial (75% or more) clearance in the number of clinically visible actinic keratoses in the treatment area on day 57.
Among patients with facial or scalp lesions, 63.9% of those who used active gel reached this end point, compared with only 7.4% of those who used placebo gel. Among patients with trunk or extremity lesions, 49.1% of those who used active gel reached this end point, compared with only 6.9% of those who used placebo gel.
Patients who used ingenol mebutate showed minimal scarring or change in pigmentation. Local reactions such as erythema, crusting, swelling, vesiculation, or pustulation were common, but were mild to moderate in intensity; they peaked within a few days of treatment and resolved rapidly afterward, with no sequelae. One patient developed eye pain, burning, and periorbital edema related to ingenol mebutate and dropped out of the study, Dr. Lebwohl and his associates noted.
"Future studies are needed to assess the risks and benefits of treating larger areas of skin, using multiple treatments in the same area, and using combination therapies," they added.
This study was funded by LEO Pharma, maker of ingenol mebutate. Dr. Lebwohl reported ties to LEO Pharma, Graceway, PharmaDerm, and Peplin LTD.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major Finding: In patients with facial or scalp lesions, 42.2% who used ingenol mebutate gel achieved complete clearance at 2 months, compared with only 3.7% of those who used placebo gel; in patients with trunk or extremity lesions, 34.1% who used active gel achieved complete clearance, compared with only 4.7% of those who used placebo gel.
Data Source: Data are from a manufacturer-sponsored, randomized, double-blind trial of 1,005 patients treated at four medical centers.
Disclosures: This study was funded by LEO Pharma, maker of ingenol mebutate. Dr. Lebwohl reported ties to LEO Pharma, Graceway, PharmaDerm, and Peplin LTD.
Sunburns Still Common Despite Protective Efforts
WASHINGTON – While the use of sun protection measures became more common between 2000 and 2010, there was not a corresponding decrease in sunburns, according to an analysis of national data.
Overall among women, staying in the shade, using sunscreen, and wearing clothing to the ankles increased significantly over time by 5%, 6%, and 5%, respectively, between 2000 and 2010. Similarly, among men, staying in the shade, sunscreen use, and wearing clothing to the ankles increased by 7%, 2%, and 5%, respectively. However, the overall prevalence of sunburn did not change significantly over those years. In 2010, 51% of women and 49% of men reported having at least one sunburn in the past year. The results come from a poster presented at the annual meeting of the American Society of Preventive Oncology.
The rates of melanoma and nonmelanoma skin cancers have increased in recent years. Monitoring and reporting sun-protective behaviors and sunburns over time are important ways to measure the impact of skin cancer prevention activity and to track progress toward Healthy People 2020 objectives, noted lead author Dawn M. Holman and her coinvestigators at the Centers for Disease Control and Prevention.
To estimate how commonly people engage in these behaviors that protect against sun exposure, the researchers used data from the National Health Interview Survey – Cancer Control Supplement, for years 2000, 2003, 2005, 2008, and 2010. The survey assessed the following behaviors as related to sun protection and sunburn: use of sunscreen, staying in the shade, wearing a wide-brimmed hat, wearing a long-sleeved shirt, and wearing long clothing to the ankles. Respondents reported their use as being: always, most of the time, sometimes, rarely, or never for each item. Respondents were also asked about the number of sunburns they’ve had in the last year.
Data were weighted to produce nationally representative estimates. Analyses were limited to those aged 18-29 years – age-adjusted to the 2000 U.S. population. The researchers estimated the percentage who reported engaging in each behavior always or most of the time and the percentage reporting one or more sunburns in the past year overall, by gender and by race/ethnicity.
Among women, using sunscreen (37%) and staying in the shade (35%) were the most common reported protective behaviors in 2010. Wearing a long-sleeved shirt (5%) and wearing a wide-brimmed hat (4%) were the least common. Black women were significantly less likely to report sunscreen use than were other racial/ethnic groups.
Among men, wearing long clothing to the ankles (33%) and staying in the shade (26%) were the most commonly reported behaviors in 2010. Fewer men reported using sunscreen (16%), wearing a long-sleeved shirt (8%), and wearing a wide-brimmed hat (7%). Of note, sunburn was significantly more common among non-Hispanic whites, compared with other racial/ethnic groups.
The results point to the "need for continued public health efforts to facilitate sun protection by: creating environments that support protective behaviors and by changing social norms regarding tanning and tanned skin. Facilitating sun protection may prevent sunburns and future increases in the burden of skin cancer," the researchers wrote.
The authors did not report whether they had any relevant financial interests.
WASHINGTON – While the use of sun protection measures became more common between 2000 and 2010, there was not a corresponding decrease in sunburns, according to an analysis of national data.
Overall among women, staying in the shade, using sunscreen, and wearing clothing to the ankles increased significantly over time by 5%, 6%, and 5%, respectively, between 2000 and 2010. Similarly, among men, staying in the shade, sunscreen use, and wearing clothing to the ankles increased by 7%, 2%, and 5%, respectively. However, the overall prevalence of sunburn did not change significantly over those years. In 2010, 51% of women and 49% of men reported having at least one sunburn in the past year. The results come from a poster presented at the annual meeting of the American Society of Preventive Oncology.
The rates of melanoma and nonmelanoma skin cancers have increased in recent years. Monitoring and reporting sun-protective behaviors and sunburns over time are important ways to measure the impact of skin cancer prevention activity and to track progress toward Healthy People 2020 objectives, noted lead author Dawn M. Holman and her coinvestigators at the Centers for Disease Control and Prevention.
To estimate how commonly people engage in these behaviors that protect against sun exposure, the researchers used data from the National Health Interview Survey – Cancer Control Supplement, for years 2000, 2003, 2005, 2008, and 2010. The survey assessed the following behaviors as related to sun protection and sunburn: use of sunscreen, staying in the shade, wearing a wide-brimmed hat, wearing a long-sleeved shirt, and wearing long clothing to the ankles. Respondents reported their use as being: always, most of the time, sometimes, rarely, or never for each item. Respondents were also asked about the number of sunburns they’ve had in the last year.
Data were weighted to produce nationally representative estimates. Analyses were limited to those aged 18-29 years – age-adjusted to the 2000 U.S. population. The researchers estimated the percentage who reported engaging in each behavior always or most of the time and the percentage reporting one or more sunburns in the past year overall, by gender and by race/ethnicity.
Among women, using sunscreen (37%) and staying in the shade (35%) were the most common reported protective behaviors in 2010. Wearing a long-sleeved shirt (5%) and wearing a wide-brimmed hat (4%) were the least common. Black women were significantly less likely to report sunscreen use than were other racial/ethnic groups.
Among men, wearing long clothing to the ankles (33%) and staying in the shade (26%) were the most commonly reported behaviors in 2010. Fewer men reported using sunscreen (16%), wearing a long-sleeved shirt (8%), and wearing a wide-brimmed hat (7%). Of note, sunburn was significantly more common among non-Hispanic whites, compared with other racial/ethnic groups.
The results point to the "need for continued public health efforts to facilitate sun protection by: creating environments that support protective behaviors and by changing social norms regarding tanning and tanned skin. Facilitating sun protection may prevent sunburns and future increases in the burden of skin cancer," the researchers wrote.
The authors did not report whether they had any relevant financial interests.
WASHINGTON – While the use of sun protection measures became more common between 2000 and 2010, there was not a corresponding decrease in sunburns, according to an analysis of national data.
Overall among women, staying in the shade, using sunscreen, and wearing clothing to the ankles increased significantly over time by 5%, 6%, and 5%, respectively, between 2000 and 2010. Similarly, among men, staying in the shade, sunscreen use, and wearing clothing to the ankles increased by 7%, 2%, and 5%, respectively. However, the overall prevalence of sunburn did not change significantly over those years. In 2010, 51% of women and 49% of men reported having at least one sunburn in the past year. The results come from a poster presented at the annual meeting of the American Society of Preventive Oncology.
The rates of melanoma and nonmelanoma skin cancers have increased in recent years. Monitoring and reporting sun-protective behaviors and sunburns over time are important ways to measure the impact of skin cancer prevention activity and to track progress toward Healthy People 2020 objectives, noted lead author Dawn M. Holman and her coinvestigators at the Centers for Disease Control and Prevention.
To estimate how commonly people engage in these behaviors that protect against sun exposure, the researchers used data from the National Health Interview Survey – Cancer Control Supplement, for years 2000, 2003, 2005, 2008, and 2010. The survey assessed the following behaviors as related to sun protection and sunburn: use of sunscreen, staying in the shade, wearing a wide-brimmed hat, wearing a long-sleeved shirt, and wearing long clothing to the ankles. Respondents reported their use as being: always, most of the time, sometimes, rarely, or never for each item. Respondents were also asked about the number of sunburns they’ve had in the last year.
Data were weighted to produce nationally representative estimates. Analyses were limited to those aged 18-29 years – age-adjusted to the 2000 U.S. population. The researchers estimated the percentage who reported engaging in each behavior always or most of the time and the percentage reporting one or more sunburns in the past year overall, by gender and by race/ethnicity.
Among women, using sunscreen (37%) and staying in the shade (35%) were the most common reported protective behaviors in 2010. Wearing a long-sleeved shirt (5%) and wearing a wide-brimmed hat (4%) were the least common. Black women were significantly less likely to report sunscreen use than were other racial/ethnic groups.
Among men, wearing long clothing to the ankles (33%) and staying in the shade (26%) were the most commonly reported behaviors in 2010. Fewer men reported using sunscreen (16%), wearing a long-sleeved shirt (8%), and wearing a wide-brimmed hat (7%). Of note, sunburn was significantly more common among non-Hispanic whites, compared with other racial/ethnic groups.
The results point to the "need for continued public health efforts to facilitate sun protection by: creating environments that support protective behaviors and by changing social norms regarding tanning and tanned skin. Facilitating sun protection may prevent sunburns and future increases in the burden of skin cancer," the researchers wrote.
The authors did not report whether they had any relevant financial interests.
FROM THE ANNUAL MEETING OF THE AMERICAN SOCIETY OF PREVENTIVE ONCOLOGY
Major Findings: In 2010, 51% of women and 49% of men reported having at least one sunburn in the past year. Sunburn was significantly more common among non-Hispanic whites, compared with other racial/ethnic groups.
Data Source: Researchers used data from the National Health Interview Survey – Cancer Control Supplement for years 2000, 2003, 2005, 2008, and 2010.
Disclosures: The authors did not report whether they had any relevant financial interests.
Tanning Regulations Linger at FDA
Rep. Rosa DeLauro (D-Conn.) sent a letter on March 9 to FDA Commissioner Margaret Hamburg urging her to act on recommendations - made by the General and Plastic Surgery Devices Panel on March 25, 2010 - to enforce stricter tanning bed regulations.
Rep. DeLauro noted that it will soon be exactly 2 years since the panel met and asked whether there will be any action before the end of May.
She also included a letter from two melanoma experts: Dr. David Fisher, director of the Melanoma Program at Massachusetts General Hospital and Mr. Alan Geller of the Harvard School of Public Health.
In the letter, the two experts contend that during the 2 years since the FDA panel meeting, tanning bed use has led to more than 5,000 cases of new melanomas, and an estimated 750 unnecessary deaths. They also asked, "How can the established skin cancer risk from tanning beds be continuously permitted, in the face of so much scientific and clinical evidence?"
Rep. DeLauro, who is a cancer survivor, said, "I simply cannot accept this inaction." She added," When will we have honest and accurate regulations and labels in place to protect Americans and end these unnecessary deaths caused by an inappropriately-regulated device?"
Her letter comes about a month after Democrats on the House Energy and Commerce Committee released results of an investigation that showed tanning salons were misleading users about the potential health risks.
The American Academy of Dermatology supports tougher regulations of indoor tanning and has urged Congress to pass H.R. 1676, the Tanning Bed Cancer Control Act. The bill had only 12 cosponsors at press time and had not been the subject of any hearings.
Rep. Rosa DeLauro (D-Conn.) sent a letter on March 9 to FDA Commissioner Margaret Hamburg urging her to act on recommendations - made by the General and Plastic Surgery Devices Panel on March 25, 2010 - to enforce stricter tanning bed regulations.
Rep. DeLauro noted that it will soon be exactly 2 years since the panel met and asked whether there will be any action before the end of May.
She also included a letter from two melanoma experts: Dr. David Fisher, director of the Melanoma Program at Massachusetts General Hospital and Mr. Alan Geller of the Harvard School of Public Health.
In the letter, the two experts contend that during the 2 years since the FDA panel meeting, tanning bed use has led to more than 5,000 cases of new melanomas, and an estimated 750 unnecessary deaths. They also asked, "How can the established skin cancer risk from tanning beds be continuously permitted, in the face of so much scientific and clinical evidence?"
Rep. DeLauro, who is a cancer survivor, said, "I simply cannot accept this inaction." She added," When will we have honest and accurate regulations and labels in place to protect Americans and end these unnecessary deaths caused by an inappropriately-regulated device?"
Her letter comes about a month after Democrats on the House Energy and Commerce Committee released results of an investigation that showed tanning salons were misleading users about the potential health risks.
The American Academy of Dermatology supports tougher regulations of indoor tanning and has urged Congress to pass H.R. 1676, the Tanning Bed Cancer Control Act. The bill had only 12 cosponsors at press time and had not been the subject of any hearings.
Rep. Rosa DeLauro (D-Conn.) sent a letter on March 9 to FDA Commissioner Margaret Hamburg urging her to act on recommendations - made by the General and Plastic Surgery Devices Panel on March 25, 2010 - to enforce stricter tanning bed regulations.
Rep. DeLauro noted that it will soon be exactly 2 years since the panel met and asked whether there will be any action before the end of May.
She also included a letter from two melanoma experts: Dr. David Fisher, director of the Melanoma Program at Massachusetts General Hospital and Mr. Alan Geller of the Harvard School of Public Health.
In the letter, the two experts contend that during the 2 years since the FDA panel meeting, tanning bed use has led to more than 5,000 cases of new melanomas, and an estimated 750 unnecessary deaths. They also asked, "How can the established skin cancer risk from tanning beds be continuously permitted, in the face of so much scientific and clinical evidence?"
Rep. DeLauro, who is a cancer survivor, said, "I simply cannot accept this inaction." She added," When will we have honest and accurate regulations and labels in place to protect Americans and end these unnecessary deaths caused by an inappropriately-regulated device?"
Her letter comes about a month after Democrats on the House Energy and Commerce Committee released results of an investigation that showed tanning salons were misleading users about the potential health risks.
The American Academy of Dermatology supports tougher regulations of indoor tanning and has urged Congress to pass H.R. 1676, the Tanning Bed Cancer Control Act. The bill had only 12 cosponsors at press time and had not been the subject of any hearings.