User login
Standard-dose RT with concurrent chemo found superior for unresectable NSCLC
More is not better when it comes to radiation therapy administered with concurrent chemotherapy for unresectable stage III non–small cell lung cancer (NSCLC), suggests a long-term update of the RTOG 0617 trial.
Initial results of the phase 3 randomized controlled trial, at a median follow-up of 1.9 years, showed that median overall survival was about 8 months longer with the 60-Gy standard dose of radiation compared with a 74-Gy high dose, each given along with paclitaxel and carboplatin (Lancet Oncol. 2015;16:187-99).
The update, now at a median follow-up of 5.1 years and reported in the Journal of Clinical Oncology, recapitulates that finding, again showing a roughly 8-month longer overall survival with the standard dose of radiation. Results continue to show no benefit of adding the anti-EGFR antibody cetuximab (Erbitux) to treatment.
“The 5-year overall survival estimate for the standard-dose radiation arm of RTOG 0617, regardless of cetuximab delivery, was 32.1%. This is among the highest overall survival results of any phase III trial for patients with stage III NSCLC,” noted the investigators, led by Jeffrey D. Bradley, MD, department of radiation oncology, Emory University, Atlanta.
“These results argue strongly that the current standard-of-care radiation dose should be 60 Gy given in 2-Gy daily fractions to a target volume directed at tumor plus margin on the basis of CT and PET/CT, excluding elective nodal irradiation.”
The RTOG 0617 trial was conducted among 496 patients with unresectable stage III NSCLC in the United States and Canada. They were randomized to receive standard-dose or high-dose radiation in addition to concurrent chemotherapy, and randomized again to receive cetuximab or not.
Median overall survival as of the update was 28.7 months with standard-dose radiation therapy, compared with 20.3 months with high-dose radiation therapy (P = .0072), Dr. Bradley and coinvestigators reported. This survival benefit stood up in multivariate analysis (hazard ratio, 1.30; P = .0315).
The standard dose of radiation also yielded better 5-year overall survival (32.1% vs. 23%; P = .007) and 5-year progression-free survival (18.3% vs. 13%; P = .055). Further analyses suggested that these differences were not due to differential radiation therapy compliance.
The high-dose radiation group had more grade 5 adverse events (nine vs. three events), as well as higher rates of treatment-related grade 3 or worse dysphagia (12.1% vs. 3.2%; P = .0005) and esophagitis (17.4% vs. 5.0%; P less than .0001). Pulmonary toxicity was statistically indistinguishable between groups.
Since the RTOG 0617 results were first reported, the standard of care for unresectable stage III NSCLC has changed, as the PACIFIC trial showed an overall survival advantage of adding the immune checkpoint inhibitor durvalumab (Imfinzi) as maintenance therapy after concurrent chemoradiotherapy (N Engl J Med. 2018;379:2342-50).
However, the 2-year overall survival rate with chemoradiation using the standard radiation dose in the former trial (59.6%) is fairly close to that seen with chemoradiation plus maintenance durvalumab in the latter trial (66.3%), the investigators noted.
Dr. Bradley disclosed having a consulting or advisory role with AstraZeneca. The trial was supported by the National Cancer Institute and Eli Lilly.
SOURCE: Bradley JD et al. J Clin Oncol. 2019 Dec 16. doi: 10.1200/JCO.19.01162.
More is not better when it comes to radiation therapy administered with concurrent chemotherapy for unresectable stage III non–small cell lung cancer (NSCLC), suggests a long-term update of the RTOG 0617 trial.
Initial results of the phase 3 randomized controlled trial, at a median follow-up of 1.9 years, showed that median overall survival was about 8 months longer with the 60-Gy standard dose of radiation compared with a 74-Gy high dose, each given along with paclitaxel and carboplatin (Lancet Oncol. 2015;16:187-99).
The update, now at a median follow-up of 5.1 years and reported in the Journal of Clinical Oncology, recapitulates that finding, again showing a roughly 8-month longer overall survival with the standard dose of radiation. Results continue to show no benefit of adding the anti-EGFR antibody cetuximab (Erbitux) to treatment.
“The 5-year overall survival estimate for the standard-dose radiation arm of RTOG 0617, regardless of cetuximab delivery, was 32.1%. This is among the highest overall survival results of any phase III trial for patients with stage III NSCLC,” noted the investigators, led by Jeffrey D. Bradley, MD, department of radiation oncology, Emory University, Atlanta.
“These results argue strongly that the current standard-of-care radiation dose should be 60 Gy given in 2-Gy daily fractions to a target volume directed at tumor plus margin on the basis of CT and PET/CT, excluding elective nodal irradiation.”
The RTOG 0617 trial was conducted among 496 patients with unresectable stage III NSCLC in the United States and Canada. They were randomized to receive standard-dose or high-dose radiation in addition to concurrent chemotherapy, and randomized again to receive cetuximab or not.
Median overall survival as of the update was 28.7 months with standard-dose radiation therapy, compared with 20.3 months with high-dose radiation therapy (P = .0072), Dr. Bradley and coinvestigators reported. This survival benefit stood up in multivariate analysis (hazard ratio, 1.30; P = .0315).
The standard dose of radiation also yielded better 5-year overall survival (32.1% vs. 23%; P = .007) and 5-year progression-free survival (18.3% vs. 13%; P = .055). Further analyses suggested that these differences were not due to differential radiation therapy compliance.
The high-dose radiation group had more grade 5 adverse events (nine vs. three events), as well as higher rates of treatment-related grade 3 or worse dysphagia (12.1% vs. 3.2%; P = .0005) and esophagitis (17.4% vs. 5.0%; P less than .0001). Pulmonary toxicity was statistically indistinguishable between groups.
Since the RTOG 0617 results were first reported, the standard of care for unresectable stage III NSCLC has changed, as the PACIFIC trial showed an overall survival advantage of adding the immune checkpoint inhibitor durvalumab (Imfinzi) as maintenance therapy after concurrent chemoradiotherapy (N Engl J Med. 2018;379:2342-50).
However, the 2-year overall survival rate with chemoradiation using the standard radiation dose in the former trial (59.6%) is fairly close to that seen with chemoradiation plus maintenance durvalumab in the latter trial (66.3%), the investigators noted.
Dr. Bradley disclosed having a consulting or advisory role with AstraZeneca. The trial was supported by the National Cancer Institute and Eli Lilly.
SOURCE: Bradley JD et al. J Clin Oncol. 2019 Dec 16. doi: 10.1200/JCO.19.01162.
More is not better when it comes to radiation therapy administered with concurrent chemotherapy for unresectable stage III non–small cell lung cancer (NSCLC), suggests a long-term update of the RTOG 0617 trial.
Initial results of the phase 3 randomized controlled trial, at a median follow-up of 1.9 years, showed that median overall survival was about 8 months longer with the 60-Gy standard dose of radiation compared with a 74-Gy high dose, each given along with paclitaxel and carboplatin (Lancet Oncol. 2015;16:187-99).
The update, now at a median follow-up of 5.1 years and reported in the Journal of Clinical Oncology, recapitulates that finding, again showing a roughly 8-month longer overall survival with the standard dose of radiation. Results continue to show no benefit of adding the anti-EGFR antibody cetuximab (Erbitux) to treatment.
“The 5-year overall survival estimate for the standard-dose radiation arm of RTOG 0617, regardless of cetuximab delivery, was 32.1%. This is among the highest overall survival results of any phase III trial for patients with stage III NSCLC,” noted the investigators, led by Jeffrey D. Bradley, MD, department of radiation oncology, Emory University, Atlanta.
“These results argue strongly that the current standard-of-care radiation dose should be 60 Gy given in 2-Gy daily fractions to a target volume directed at tumor plus margin on the basis of CT and PET/CT, excluding elective nodal irradiation.”
The RTOG 0617 trial was conducted among 496 patients with unresectable stage III NSCLC in the United States and Canada. They were randomized to receive standard-dose or high-dose radiation in addition to concurrent chemotherapy, and randomized again to receive cetuximab or not.
Median overall survival as of the update was 28.7 months with standard-dose radiation therapy, compared with 20.3 months with high-dose radiation therapy (P = .0072), Dr. Bradley and coinvestigators reported. This survival benefit stood up in multivariate analysis (hazard ratio, 1.30; P = .0315).
The standard dose of radiation also yielded better 5-year overall survival (32.1% vs. 23%; P = .007) and 5-year progression-free survival (18.3% vs. 13%; P = .055). Further analyses suggested that these differences were not due to differential radiation therapy compliance.
The high-dose radiation group had more grade 5 adverse events (nine vs. three events), as well as higher rates of treatment-related grade 3 or worse dysphagia (12.1% vs. 3.2%; P = .0005) and esophagitis (17.4% vs. 5.0%; P less than .0001). Pulmonary toxicity was statistically indistinguishable between groups.
Since the RTOG 0617 results were first reported, the standard of care for unresectable stage III NSCLC has changed, as the PACIFIC trial showed an overall survival advantage of adding the immune checkpoint inhibitor durvalumab (Imfinzi) as maintenance therapy after concurrent chemoradiotherapy (N Engl J Med. 2018;379:2342-50).
However, the 2-year overall survival rate with chemoradiation using the standard radiation dose in the former trial (59.6%) is fairly close to that seen with chemoradiation plus maintenance durvalumab in the latter trial (66.3%), the investigators noted.
Dr. Bradley disclosed having a consulting or advisory role with AstraZeneca. The trial was supported by the National Cancer Institute and Eli Lilly.
SOURCE: Bradley JD et al. J Clin Oncol. 2019 Dec 16. doi: 10.1200/JCO.19.01162.
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Benefits and drawbacks found for risk-based lung cancer screening tools
Risk-based lung cancer screening tools can prevent significantly more lung cancer deaths than the current United States Preventive Services Task Force recommendations, but life-year gains were negligible or reduced and patients would experience greater overdiagnosis, according to new research.
“Current guidelines propose screening eligibility using age and smoking-related criteria, through combinations of accumulated pack-years and years since smoking cessation,” Kevin ten Haaf, PhD, of the department of public health at Erasmus Medical Center in Rotterdam, the Netherlands, and associates wrote in the Journal of the National Cancer Institute. “The USPSTF recommends annual screening between the ages of 55 and 80 years for current and former smokers (quit less than 15 years) who smoked 30 or more pack-years.”
Individual risk assessment utilizing established lung cancer risk–prediction models may have some superiority over pack-years in identifying those most likely to benefit from screening, they wrote, because the models incorporate smoking history in greater detail and consider risk factors such as chronic obstructive pulmonary disease.
Three risk-assessment models were used for the study, in addition to the USPSTF guidelines: the Bach, PLCOm2012, and Lung Cancer Death Risk Assessment Tool (LCDRAT). The study population was a simulated 1950 U.S. cohort from the Smoking History Generator aged between 55 years and 80 years; each simulated smoking history consists of whether and when the person initiates and ceases smoking, average number of cigarettes smoked per day by age, and the age of death from non–lung cancer causes.
The number of lung cancer deaths averted was significantly higher in the risk-based models, compared with the USPSTF recommendations (Bach, 693 per 100,000 population; PLCOm2012, 698 per 100,000 population; LCDRAT, 696 per 100,000 population; USPSTF, 613 per 100,000 population).
However, life-years gained was only modestly higher in the models, compared with the guideline (Bach, 8,660 per 100,000 life-years; PLCOm2012, 8,862 per 100,000 life-years; LCDRAT, 8,631 per 100,000 life-years; USPSTF, 8,590 per 100,000 life-years). In addition, life-years gained for every lung cancer death prevented was greater in the guideline (14.0 years) than in the risk-based models (12.1-12.4 years).
Overdiagnosis was also more common using risk-based tools (Bach, 149 per 100,000; PLCOm2012, 147 per 100,000; LCDRAT, 150 per 100,000; USPSTF, 115 per 100,000). This was mainly because of eligibility for risk-based screening tools increasing with age, the investigators noted.
According to a sensitivity analysis, risk-based models would retain the life-years gained by the USPSTF model if individuals with limited life expectancies (less than 5 years) were excluded. This would also reduce overdiagnosis by 65.3%.
“Future studies should investigate the cost-effectiveness of risk-based screening and the potential for reducing overdiagnosis in high-risk individuals,” the investigators concluded.
One coauthor developed the PLCOm2012 model, but the model is available free to noncommercial users, and the investigator has received no money from its usage. No other conflicts of interest were reported.
SOURCE: ten Haaf K et al. J Natl Cancer Inst. 2019 Nov 29. doi: 10.1093/jnci/djz164.
Risk-based lung cancer screening tools can prevent significantly more lung cancer deaths than the current United States Preventive Services Task Force recommendations, but life-year gains were negligible or reduced and patients would experience greater overdiagnosis, according to new research.
“Current guidelines propose screening eligibility using age and smoking-related criteria, through combinations of accumulated pack-years and years since smoking cessation,” Kevin ten Haaf, PhD, of the department of public health at Erasmus Medical Center in Rotterdam, the Netherlands, and associates wrote in the Journal of the National Cancer Institute. “The USPSTF recommends annual screening between the ages of 55 and 80 years for current and former smokers (quit less than 15 years) who smoked 30 or more pack-years.”
Individual risk assessment utilizing established lung cancer risk–prediction models may have some superiority over pack-years in identifying those most likely to benefit from screening, they wrote, because the models incorporate smoking history in greater detail and consider risk factors such as chronic obstructive pulmonary disease.
Three risk-assessment models were used for the study, in addition to the USPSTF guidelines: the Bach, PLCOm2012, and Lung Cancer Death Risk Assessment Tool (LCDRAT). The study population was a simulated 1950 U.S. cohort from the Smoking History Generator aged between 55 years and 80 years; each simulated smoking history consists of whether and when the person initiates and ceases smoking, average number of cigarettes smoked per day by age, and the age of death from non–lung cancer causes.
The number of lung cancer deaths averted was significantly higher in the risk-based models, compared with the USPSTF recommendations (Bach, 693 per 100,000 population; PLCOm2012, 698 per 100,000 population; LCDRAT, 696 per 100,000 population; USPSTF, 613 per 100,000 population).
However, life-years gained was only modestly higher in the models, compared with the guideline (Bach, 8,660 per 100,000 life-years; PLCOm2012, 8,862 per 100,000 life-years; LCDRAT, 8,631 per 100,000 life-years; USPSTF, 8,590 per 100,000 life-years). In addition, life-years gained for every lung cancer death prevented was greater in the guideline (14.0 years) than in the risk-based models (12.1-12.4 years).
Overdiagnosis was also more common using risk-based tools (Bach, 149 per 100,000; PLCOm2012, 147 per 100,000; LCDRAT, 150 per 100,000; USPSTF, 115 per 100,000). This was mainly because of eligibility for risk-based screening tools increasing with age, the investigators noted.
According to a sensitivity analysis, risk-based models would retain the life-years gained by the USPSTF model if individuals with limited life expectancies (less than 5 years) were excluded. This would also reduce overdiagnosis by 65.3%.
“Future studies should investigate the cost-effectiveness of risk-based screening and the potential for reducing overdiagnosis in high-risk individuals,” the investigators concluded.
One coauthor developed the PLCOm2012 model, but the model is available free to noncommercial users, and the investigator has received no money from its usage. No other conflicts of interest were reported.
SOURCE: ten Haaf K et al. J Natl Cancer Inst. 2019 Nov 29. doi: 10.1093/jnci/djz164.
Risk-based lung cancer screening tools can prevent significantly more lung cancer deaths than the current United States Preventive Services Task Force recommendations, but life-year gains were negligible or reduced and patients would experience greater overdiagnosis, according to new research.
“Current guidelines propose screening eligibility using age and smoking-related criteria, through combinations of accumulated pack-years and years since smoking cessation,” Kevin ten Haaf, PhD, of the department of public health at Erasmus Medical Center in Rotterdam, the Netherlands, and associates wrote in the Journal of the National Cancer Institute. “The USPSTF recommends annual screening between the ages of 55 and 80 years for current and former smokers (quit less than 15 years) who smoked 30 or more pack-years.”
Individual risk assessment utilizing established lung cancer risk–prediction models may have some superiority over pack-years in identifying those most likely to benefit from screening, they wrote, because the models incorporate smoking history in greater detail and consider risk factors such as chronic obstructive pulmonary disease.
Three risk-assessment models were used for the study, in addition to the USPSTF guidelines: the Bach, PLCOm2012, and Lung Cancer Death Risk Assessment Tool (LCDRAT). The study population was a simulated 1950 U.S. cohort from the Smoking History Generator aged between 55 years and 80 years; each simulated smoking history consists of whether and when the person initiates and ceases smoking, average number of cigarettes smoked per day by age, and the age of death from non–lung cancer causes.
The number of lung cancer deaths averted was significantly higher in the risk-based models, compared with the USPSTF recommendations (Bach, 693 per 100,000 population; PLCOm2012, 698 per 100,000 population; LCDRAT, 696 per 100,000 population; USPSTF, 613 per 100,000 population).
However, life-years gained was only modestly higher in the models, compared with the guideline (Bach, 8,660 per 100,000 life-years; PLCOm2012, 8,862 per 100,000 life-years; LCDRAT, 8,631 per 100,000 life-years; USPSTF, 8,590 per 100,000 life-years). In addition, life-years gained for every lung cancer death prevented was greater in the guideline (14.0 years) than in the risk-based models (12.1-12.4 years).
Overdiagnosis was also more common using risk-based tools (Bach, 149 per 100,000; PLCOm2012, 147 per 100,000; LCDRAT, 150 per 100,000; USPSTF, 115 per 100,000). This was mainly because of eligibility for risk-based screening tools increasing with age, the investigators noted.
According to a sensitivity analysis, risk-based models would retain the life-years gained by the USPSTF model if individuals with limited life expectancies (less than 5 years) were excluded. This would also reduce overdiagnosis by 65.3%.
“Future studies should investigate the cost-effectiveness of risk-based screening and the potential for reducing overdiagnosis in high-risk individuals,” the investigators concluded.
One coauthor developed the PLCOm2012 model, but the model is available free to noncommercial users, and the investigator has received no money from its usage. No other conflicts of interest were reported.
SOURCE: ten Haaf K et al. J Natl Cancer Inst. 2019 Nov 29. doi: 10.1093/jnci/djz164.
FROM THE JOURNAL OF THE NATIONAL CANCER INSTITUTE
Patient-reported outcomes support first-line pembrolizumab for NSCLC
Patient-reported outcomes support pembrolizumab plus chemotherapy for first-line treatment of metastatic non–small cell lung cancer (NSCLC), based on results from the KEYNOTE-407 trial.
At week 18, patients given pembrolizumab more often reported clinically meaningful health-related quality of life improvements than those in the placebo group, according to lead author Julien Mazieres, MD, PhD, of Paul Sabatier University in Toulouse, France, and colleagues.
Writing in the Journal of Clinical Oncology, the investigators explained that these findings build upon previously published results from KEYNOTE-407, which showed that adding pembrolizumab to chemotherapy in the first line could extend both progression-free and overall survival among patients with NSCLC. The benefits to quality of life associated with pembrolizumab align with similar findings from the KEYNOTE-024 and KEYNOTE-189 trials, they added.
The present analysis involved 559 patients with treatment-naive metastatic NSCLC. Patients were randomized to receive 4 cycles of placebo or pembrolizumab once every 3 weeks with carboplatin-based chemotherapy, followed by pembrolizumab or placebo for an additional 31 cycles. Health-related quality of life was assessed by two measures: the European Organisation for the Research and Treatment of Cancer Quality of Life Questionnaire–Core 30 (QLQ-C30) and QLQ–Lung Cancer Module 13 (QLQ-LC13).
Of the 559 patients enrolled, 554 completed at least one QLQ-C30 assessment and 553 completed at least one QLQ-LC13 assessment. These quality of life scores were compared temporally within treatment groups, from baseline to week 9 and week 18, and between groups. The investigators also analyzed median time to deterioration in chest pain, cough, and dyspnea.
Results showed that patients in the pembrolizumab group had statistically significant improvements in patient-reported outcomes over time and more frequently reported improvements than patients in the placebo group. Specifically, in the pembrolizumab group, least-squares mean score improved from baseline to week 9 (1.8) and week 18 (4.3); in comparison, least-squares mean score deteriorated in the placebo group from baseline to week 9 (–1.8) and week 18 (–0.57). Compared with placebo, treatment with pembrolizumab was associated with a least-squares mean change of 3.6 at week 9 (nominal P = .0337) and 4.9 at week 18 (nominal P = .0060). Stated differently, at week 18, compared with placebo, more patients in the pembrolizumab group reported clinically meaningful improvements in health-related quality of life (36.2% vs. 27.7%), and relatively fewer reported deterioration (22.8% vs. 31.3%). Median time to deterioration in symptoms was not reached in either treatment arm.
“These health-related quality of life findings, along with the improved efficacy (including overall survival benefit) of pembrolizumab plus carboplatin and paclitaxel/nab-paclitaxel, support its use as a first-line treatment of metastatic squamous NSCLC, regardless of programmed death–ligand 1 expression,” the investigators concluded.
The study was funded by Merck. The investigators reported additional relationships with Novartis, Genentech, Pfizer, and others.
SOURCE: Mazieres J et al. J Clin Oncol. 2019 Nov 21. doi: 10.1200/JCO.19.01348.
Patient-reported outcomes support pembrolizumab plus chemotherapy for first-line treatment of metastatic non–small cell lung cancer (NSCLC), based on results from the KEYNOTE-407 trial.
At week 18, patients given pembrolizumab more often reported clinically meaningful health-related quality of life improvements than those in the placebo group, according to lead author Julien Mazieres, MD, PhD, of Paul Sabatier University in Toulouse, France, and colleagues.
Writing in the Journal of Clinical Oncology, the investigators explained that these findings build upon previously published results from KEYNOTE-407, which showed that adding pembrolizumab to chemotherapy in the first line could extend both progression-free and overall survival among patients with NSCLC. The benefits to quality of life associated with pembrolizumab align with similar findings from the KEYNOTE-024 and KEYNOTE-189 trials, they added.
The present analysis involved 559 patients with treatment-naive metastatic NSCLC. Patients were randomized to receive 4 cycles of placebo or pembrolizumab once every 3 weeks with carboplatin-based chemotherapy, followed by pembrolizumab or placebo for an additional 31 cycles. Health-related quality of life was assessed by two measures: the European Organisation for the Research and Treatment of Cancer Quality of Life Questionnaire–Core 30 (QLQ-C30) and QLQ–Lung Cancer Module 13 (QLQ-LC13).
Of the 559 patients enrolled, 554 completed at least one QLQ-C30 assessment and 553 completed at least one QLQ-LC13 assessment. These quality of life scores were compared temporally within treatment groups, from baseline to week 9 and week 18, and between groups. The investigators also analyzed median time to deterioration in chest pain, cough, and dyspnea.
Results showed that patients in the pembrolizumab group had statistically significant improvements in patient-reported outcomes over time and more frequently reported improvements than patients in the placebo group. Specifically, in the pembrolizumab group, least-squares mean score improved from baseline to week 9 (1.8) and week 18 (4.3); in comparison, least-squares mean score deteriorated in the placebo group from baseline to week 9 (–1.8) and week 18 (–0.57). Compared with placebo, treatment with pembrolizumab was associated with a least-squares mean change of 3.6 at week 9 (nominal P = .0337) and 4.9 at week 18 (nominal P = .0060). Stated differently, at week 18, compared with placebo, more patients in the pembrolizumab group reported clinically meaningful improvements in health-related quality of life (36.2% vs. 27.7%), and relatively fewer reported deterioration (22.8% vs. 31.3%). Median time to deterioration in symptoms was not reached in either treatment arm.
“These health-related quality of life findings, along with the improved efficacy (including overall survival benefit) of pembrolizumab plus carboplatin and paclitaxel/nab-paclitaxel, support its use as a first-line treatment of metastatic squamous NSCLC, regardless of programmed death–ligand 1 expression,” the investigators concluded.
The study was funded by Merck. The investigators reported additional relationships with Novartis, Genentech, Pfizer, and others.
SOURCE: Mazieres J et al. J Clin Oncol. 2019 Nov 21. doi: 10.1200/JCO.19.01348.
Patient-reported outcomes support pembrolizumab plus chemotherapy for first-line treatment of metastatic non–small cell lung cancer (NSCLC), based on results from the KEYNOTE-407 trial.
At week 18, patients given pembrolizumab more often reported clinically meaningful health-related quality of life improvements than those in the placebo group, according to lead author Julien Mazieres, MD, PhD, of Paul Sabatier University in Toulouse, France, and colleagues.
Writing in the Journal of Clinical Oncology, the investigators explained that these findings build upon previously published results from KEYNOTE-407, which showed that adding pembrolizumab to chemotherapy in the first line could extend both progression-free and overall survival among patients with NSCLC. The benefits to quality of life associated with pembrolizumab align with similar findings from the KEYNOTE-024 and KEYNOTE-189 trials, they added.
The present analysis involved 559 patients with treatment-naive metastatic NSCLC. Patients were randomized to receive 4 cycles of placebo or pembrolizumab once every 3 weeks with carboplatin-based chemotherapy, followed by pembrolizumab or placebo for an additional 31 cycles. Health-related quality of life was assessed by two measures: the European Organisation for the Research and Treatment of Cancer Quality of Life Questionnaire–Core 30 (QLQ-C30) and QLQ–Lung Cancer Module 13 (QLQ-LC13).
Of the 559 patients enrolled, 554 completed at least one QLQ-C30 assessment and 553 completed at least one QLQ-LC13 assessment. These quality of life scores were compared temporally within treatment groups, from baseline to week 9 and week 18, and between groups. The investigators also analyzed median time to deterioration in chest pain, cough, and dyspnea.
Results showed that patients in the pembrolizumab group had statistically significant improvements in patient-reported outcomes over time and more frequently reported improvements than patients in the placebo group. Specifically, in the pembrolizumab group, least-squares mean score improved from baseline to week 9 (1.8) and week 18 (4.3); in comparison, least-squares mean score deteriorated in the placebo group from baseline to week 9 (–1.8) and week 18 (–0.57). Compared with placebo, treatment with pembrolizumab was associated with a least-squares mean change of 3.6 at week 9 (nominal P = .0337) and 4.9 at week 18 (nominal P = .0060). Stated differently, at week 18, compared with placebo, more patients in the pembrolizumab group reported clinically meaningful improvements in health-related quality of life (36.2% vs. 27.7%), and relatively fewer reported deterioration (22.8% vs. 31.3%). Median time to deterioration in symptoms was not reached in either treatment arm.
“These health-related quality of life findings, along with the improved efficacy (including overall survival benefit) of pembrolizumab plus carboplatin and paclitaxel/nab-paclitaxel, support its use as a first-line treatment of metastatic squamous NSCLC, regardless of programmed death–ligand 1 expression,” the investigators concluded.
The study was funded by Merck. The investigators reported additional relationships with Novartis, Genentech, Pfizer, and others.
SOURCE: Mazieres J et al. J Clin Oncol. 2019 Nov 21. doi: 10.1200/JCO.19.01348.
FROM THE JOURNAL OF CLINICAL ONCOLOGY
FDA approves atezolizumab combo as first line for advanced NSCLC
chemotherapy for first-line treatment of adults with metastatic, nonsquamous non–small cell lung cancer (NSCLC) with no EGFR or ALK genomic tumor aberrations.
Atezolizumab has been previously approved in combination with bevacizumab, paclitaxel, and carboplatin for the first-line treatment of adults with metastatic NSCLC with no EGFR or ALK genomic tumor aberrations. The monoclonal antibody is also approved to treat adults with metastatic NSCLC who have disease progression during or following chemotherapy, and for those with extensive-stage SCLC.
The current approval was based on a demonstrated improvement in overall survival in the phase 3 IMpower130 trial (NCT02367781). Median overall survival for advanced NSCLC patients who received atezolizumab in combination with chemotherapy was 18.6 months, compared with 13.9 months for patients who received chemotherapy alone (hazard ratio, 0.80; 95% confidence interval, 0.64-0.99; P = .0384) in the intention-to-treat wild-type population of 681 patients.
Grade 3-4 treatment-related adverse events were reported in 73.2% of people receiving atezolizumab plus chemotherapy, compared with 60.3% of people receiving chemotherapy alone, according to the company press release.
chemotherapy for first-line treatment of adults with metastatic, nonsquamous non–small cell lung cancer (NSCLC) with no EGFR or ALK genomic tumor aberrations.
Atezolizumab has been previously approved in combination with bevacizumab, paclitaxel, and carboplatin for the first-line treatment of adults with metastatic NSCLC with no EGFR or ALK genomic tumor aberrations. The monoclonal antibody is also approved to treat adults with metastatic NSCLC who have disease progression during or following chemotherapy, and for those with extensive-stage SCLC.
The current approval was based on a demonstrated improvement in overall survival in the phase 3 IMpower130 trial (NCT02367781). Median overall survival for advanced NSCLC patients who received atezolizumab in combination with chemotherapy was 18.6 months, compared with 13.9 months for patients who received chemotherapy alone (hazard ratio, 0.80; 95% confidence interval, 0.64-0.99; P = .0384) in the intention-to-treat wild-type population of 681 patients.
Grade 3-4 treatment-related adverse events were reported in 73.2% of people receiving atezolizumab plus chemotherapy, compared with 60.3% of people receiving chemotherapy alone, according to the company press release.
chemotherapy for first-line treatment of adults with metastatic, nonsquamous non–small cell lung cancer (NSCLC) with no EGFR or ALK genomic tumor aberrations.
Atezolizumab has been previously approved in combination with bevacizumab, paclitaxel, and carboplatin for the first-line treatment of adults with metastatic NSCLC with no EGFR or ALK genomic tumor aberrations. The monoclonal antibody is also approved to treat adults with metastatic NSCLC who have disease progression during or following chemotherapy, and for those with extensive-stage SCLC.
The current approval was based on a demonstrated improvement in overall survival in the phase 3 IMpower130 trial (NCT02367781). Median overall survival for advanced NSCLC patients who received atezolizumab in combination with chemotherapy was 18.6 months, compared with 13.9 months for patients who received chemotherapy alone (hazard ratio, 0.80; 95% confidence interval, 0.64-0.99; P = .0384) in the intention-to-treat wild-type population of 681 patients.
Grade 3-4 treatment-related adverse events were reported in 73.2% of people receiving atezolizumab plus chemotherapy, compared with 60.3% of people receiving chemotherapy alone, according to the company press release.
Single-fraction radiation just misses mark for spinal compression relief
Single-fraction radiation could not be shown to be noninferior to multi-fraction radiation at improving walking function in patients with spinal compression from metastatic cancer, but the small differences seen in a noninferiority trial may not matter to patients, investigators suggest.
Among 686 patients with spinal compression from metastatic cancer randomly assigned in a clinical trial to receive either 8 Gy of radiation in a single fraction or 20 Gy delivered in 5 fractions over 5 consecutive days, 69.3% of patients in the single-fraction arm had good ambulatory status at 8 weeks, compared with 72.7% of patients in the multi-fraction arm (P for noninferiority = .06), reported Peter J Hoskin, BSc, MBBS, MD, of Mount Vernon Cancer Centre in Northwood, England, and colleagues.
The trial did not meet the endpoint of noninferiority of single-fraction radiation for improving ambulation at 8 weeks because the lower limit of the 95% confidence interval (CI) was –11.5%, overlapping the noninferiority margin of –11%.
“However, for all other time points, the CI limits were within the noninferiority margin, and the observed risk differences between single-fraction and multi-fraction radiotherapy groups in ambulatory status were small and unlikely to be of clinical importance,” the investigators wrote in JAMA.
The authors note that although radiotherapy is widely used as a palliative measure for patients with spinal canal compression caused my metastatic disease, there is no agreement on the optimum schedule, with some guidelines recommending higher doses in multiple fractions, and others recommending a single 8 Gy does for patients with painful spinal sites.
To see whether single-fraction radiation could be noninferior to multi-fraction, the investigators enrolled patients in 42 sites in the United Kingdom and 5 in Australia into the SCORAD trial, and randomly assigned them to either single-fraction (345 patients) or multi-fraction (341 patients) radiation. The median age of those enrolled was 70 years, and 44% had prostate cancer, 19% had lung cancer, and 12% had breast cancer.
As noted, the primary endpoint of noninferiority of single-fraction radiation at improving ambulatory status at week 8 was not met. Ambulatory status was based on a 4-point scale and was classified as either grade 1: ambulatory without the use of aids and grade 5 of 5 of muscle power, or grade 2: ambulatory with aids or grade 4 of 5 of muscle power.
An analysis of secondary endpoints showed that the difference in ambulatory status grade 1 or 2 in the single- vs. multi-fraction group at week 1 was −0.4% (P value for noninferiority = .004), at week 4 it was −0.7% (P value for noninferiority = .01), and at week 12 it was 4.1% (P value for noninferiority = .002).
Overall survival rates at 12 weeks were 50% in the single-fraction group vs. 55% in the multi-fraction group; this difference was not statistically significant.
Of 11 other secondary endpoints analyzed, including ambulatory and safety endpoints, the between-group differences were not statistically significant or did not meet noninferiority criteria, the authors noted.
They concluded that although the trial did not meet the primary endpoint, ”the extent to which the lower bound of the CI overlapped with the noninferiority margin should be taken into account when interpreting the clinical importance of these findings.”
Cancer Research UK and Cancer Council Queensland funded the trial. Dr. Hoskin reported being supported by the National Institute for Health Research Manchester Biomedical Research Centre.
SOURCE: Hoskin PJ et al. JAMA 2019 Dec 3. doi: 10.1001/jama.2019.17913.
Single-fraction radiation could not be shown to be noninferior to multi-fraction radiation at improving walking function in patients with spinal compression from metastatic cancer, but the small differences seen in a noninferiority trial may not matter to patients, investigators suggest.
Among 686 patients with spinal compression from metastatic cancer randomly assigned in a clinical trial to receive either 8 Gy of radiation in a single fraction or 20 Gy delivered in 5 fractions over 5 consecutive days, 69.3% of patients in the single-fraction arm had good ambulatory status at 8 weeks, compared with 72.7% of patients in the multi-fraction arm (P for noninferiority = .06), reported Peter J Hoskin, BSc, MBBS, MD, of Mount Vernon Cancer Centre in Northwood, England, and colleagues.
The trial did not meet the endpoint of noninferiority of single-fraction radiation for improving ambulation at 8 weeks because the lower limit of the 95% confidence interval (CI) was –11.5%, overlapping the noninferiority margin of –11%.
“However, for all other time points, the CI limits were within the noninferiority margin, and the observed risk differences between single-fraction and multi-fraction radiotherapy groups in ambulatory status were small and unlikely to be of clinical importance,” the investigators wrote in JAMA.
The authors note that although radiotherapy is widely used as a palliative measure for patients with spinal canal compression caused my metastatic disease, there is no agreement on the optimum schedule, with some guidelines recommending higher doses in multiple fractions, and others recommending a single 8 Gy does for patients with painful spinal sites.
To see whether single-fraction radiation could be noninferior to multi-fraction, the investigators enrolled patients in 42 sites in the United Kingdom and 5 in Australia into the SCORAD trial, and randomly assigned them to either single-fraction (345 patients) or multi-fraction (341 patients) radiation. The median age of those enrolled was 70 years, and 44% had prostate cancer, 19% had lung cancer, and 12% had breast cancer.
As noted, the primary endpoint of noninferiority of single-fraction radiation at improving ambulatory status at week 8 was not met. Ambulatory status was based on a 4-point scale and was classified as either grade 1: ambulatory without the use of aids and grade 5 of 5 of muscle power, or grade 2: ambulatory with aids or grade 4 of 5 of muscle power.
An analysis of secondary endpoints showed that the difference in ambulatory status grade 1 or 2 in the single- vs. multi-fraction group at week 1 was −0.4% (P value for noninferiority = .004), at week 4 it was −0.7% (P value for noninferiority = .01), and at week 12 it was 4.1% (P value for noninferiority = .002).
Overall survival rates at 12 weeks were 50% in the single-fraction group vs. 55% in the multi-fraction group; this difference was not statistically significant.
Of 11 other secondary endpoints analyzed, including ambulatory and safety endpoints, the between-group differences were not statistically significant or did not meet noninferiority criteria, the authors noted.
They concluded that although the trial did not meet the primary endpoint, ”the extent to which the lower bound of the CI overlapped with the noninferiority margin should be taken into account when interpreting the clinical importance of these findings.”
Cancer Research UK and Cancer Council Queensland funded the trial. Dr. Hoskin reported being supported by the National Institute for Health Research Manchester Biomedical Research Centre.
SOURCE: Hoskin PJ et al. JAMA 2019 Dec 3. doi: 10.1001/jama.2019.17913.
Single-fraction radiation could not be shown to be noninferior to multi-fraction radiation at improving walking function in patients with spinal compression from metastatic cancer, but the small differences seen in a noninferiority trial may not matter to patients, investigators suggest.
Among 686 patients with spinal compression from metastatic cancer randomly assigned in a clinical trial to receive either 8 Gy of radiation in a single fraction or 20 Gy delivered in 5 fractions over 5 consecutive days, 69.3% of patients in the single-fraction arm had good ambulatory status at 8 weeks, compared with 72.7% of patients in the multi-fraction arm (P for noninferiority = .06), reported Peter J Hoskin, BSc, MBBS, MD, of Mount Vernon Cancer Centre in Northwood, England, and colleagues.
The trial did not meet the endpoint of noninferiority of single-fraction radiation for improving ambulation at 8 weeks because the lower limit of the 95% confidence interval (CI) was –11.5%, overlapping the noninferiority margin of –11%.
“However, for all other time points, the CI limits were within the noninferiority margin, and the observed risk differences between single-fraction and multi-fraction radiotherapy groups in ambulatory status were small and unlikely to be of clinical importance,” the investigators wrote in JAMA.
The authors note that although radiotherapy is widely used as a palliative measure for patients with spinal canal compression caused my metastatic disease, there is no agreement on the optimum schedule, with some guidelines recommending higher doses in multiple fractions, and others recommending a single 8 Gy does for patients with painful spinal sites.
To see whether single-fraction radiation could be noninferior to multi-fraction, the investigators enrolled patients in 42 sites in the United Kingdom and 5 in Australia into the SCORAD trial, and randomly assigned them to either single-fraction (345 patients) or multi-fraction (341 patients) radiation. The median age of those enrolled was 70 years, and 44% had prostate cancer, 19% had lung cancer, and 12% had breast cancer.
As noted, the primary endpoint of noninferiority of single-fraction radiation at improving ambulatory status at week 8 was not met. Ambulatory status was based on a 4-point scale and was classified as either grade 1: ambulatory without the use of aids and grade 5 of 5 of muscle power, or grade 2: ambulatory with aids or grade 4 of 5 of muscle power.
An analysis of secondary endpoints showed that the difference in ambulatory status grade 1 or 2 in the single- vs. multi-fraction group at week 1 was −0.4% (P value for noninferiority = .004), at week 4 it was −0.7% (P value for noninferiority = .01), and at week 12 it was 4.1% (P value for noninferiority = .002).
Overall survival rates at 12 weeks were 50% in the single-fraction group vs. 55% in the multi-fraction group; this difference was not statistically significant.
Of 11 other secondary endpoints analyzed, including ambulatory and safety endpoints, the between-group differences were not statistically significant or did not meet noninferiority criteria, the authors noted.
They concluded that although the trial did not meet the primary endpoint, ”the extent to which the lower bound of the CI overlapped with the noninferiority margin should be taken into account when interpreting the clinical importance of these findings.”
Cancer Research UK and Cancer Council Queensland funded the trial. Dr. Hoskin reported being supported by the National Institute for Health Research Manchester Biomedical Research Centre.
SOURCE: Hoskin PJ et al. JAMA 2019 Dec 3. doi: 10.1001/jama.2019.17913.
FROM JAMA
Large state disparities seen for lung cancer screening
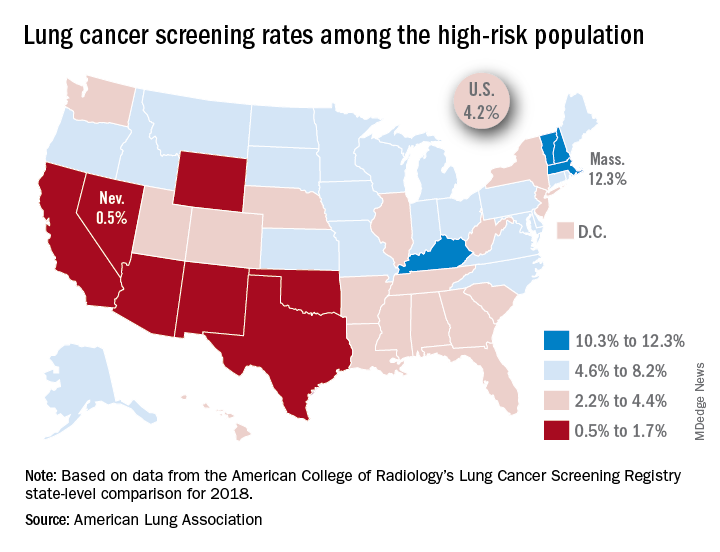
That disparity might suggest that Massachusetts has an exceptionally high rate, but it’s only 12.3%. And that means Nevada’s rate is very low, which it is: Only 0.5% of those at high risk are getting screened with annual low-dose CT scans, the ALA said in its 2019 State of Lung Cancer report.
“[The low rate of screening] may be because of a lack of access or low awareness and knowledge among patients and providers. As rates vary tremendously between states, it is clear that more can be done to increase screening rates,” the ALA stated.
Nationally, the screening rate is 4.2% among those at high risk for lung cancer, but “if everyone currently eligible were screened, close to 48,000 lives could be saved,” the ALA noted in its report.
Six states other than Nevada are below the 2% mark: Arizona, California, New Mexico, Oklahoma, Texas, and Wyoming. Besides Massachusetts, the three other states above 10% are Kentucky, New Hampshire, and Vermont, according to data from the American College of Radiology’s Lung Cancer Screening Registry state-level comparison for 2018.
For individuals at high risk for lung cancer – those aged 55-80 years who have at least a 30 pack-year history and either still smoke or have quit within 15 years – “screening with annual low-dose CT scans can reduce the lung cancer death rate by up to 20% by detecting tumors at early stages when the cancer is more likely to be curable,” the ALA wrote.

That disparity might suggest that Massachusetts has an exceptionally high rate, but it’s only 12.3%. And that means Nevada’s rate is very low, which it is: Only 0.5% of those at high risk are getting screened with annual low-dose CT scans, the ALA said in its 2019 State of Lung Cancer report.
“[The low rate of screening] may be because of a lack of access or low awareness and knowledge among patients and providers. As rates vary tremendously between states, it is clear that more can be done to increase screening rates,” the ALA stated.
Nationally, the screening rate is 4.2% among those at high risk for lung cancer, but “if everyone currently eligible were screened, close to 48,000 lives could be saved,” the ALA noted in its report.
Six states other than Nevada are below the 2% mark: Arizona, California, New Mexico, Oklahoma, Texas, and Wyoming. Besides Massachusetts, the three other states above 10% are Kentucky, New Hampshire, and Vermont, according to data from the American College of Radiology’s Lung Cancer Screening Registry state-level comparison for 2018.
For individuals at high risk for lung cancer – those aged 55-80 years who have at least a 30 pack-year history and either still smoke or have quit within 15 years – “screening with annual low-dose CT scans can reduce the lung cancer death rate by up to 20% by detecting tumors at early stages when the cancer is more likely to be curable,” the ALA wrote.

That disparity might suggest that Massachusetts has an exceptionally high rate, but it’s only 12.3%. And that means Nevada’s rate is very low, which it is: Only 0.5% of those at high risk are getting screened with annual low-dose CT scans, the ALA said in its 2019 State of Lung Cancer report.
“[The low rate of screening] may be because of a lack of access or low awareness and knowledge among patients and providers. As rates vary tremendously between states, it is clear that more can be done to increase screening rates,” the ALA stated.
Nationally, the screening rate is 4.2% among those at high risk for lung cancer, but “if everyone currently eligible were screened, close to 48,000 lives could be saved,” the ALA noted in its report.
Six states other than Nevada are below the 2% mark: Arizona, California, New Mexico, Oklahoma, Texas, and Wyoming. Besides Massachusetts, the three other states above 10% are Kentucky, New Hampshire, and Vermont, according to data from the American College of Radiology’s Lung Cancer Screening Registry state-level comparison for 2018.
For individuals at high risk for lung cancer – those aged 55-80 years who have at least a 30 pack-year history and either still smoke or have quit within 15 years – “screening with annual low-dose CT scans can reduce the lung cancer death rate by up to 20% by detecting tumors at early stages when the cancer is more likely to be curable,” the ALA wrote.
Expanding the reach of available cancer therapies
In this edition of “How I Will Treat My Next Patient,” I highlight two articles that demonstrate the safety of established treatments – nephrectomy and stereotactic ablative body radiotherapy (SABR) – in patient populations that previously may have been excluded from those treatments at many centers.
Nephrectomy in advanced RCC
Nirmish Singla, MD, and colleagues reported a single-center retrospective cohort study, assessing outcomes of 11 nephrectomies (10 radical, 1 partial) in 10 patients with advanced renal cell carcinoma (RCC) who had received front- or later-line immune checkpoint inhibitor therapy (ICIs). Half had received nivolumab alone; the others received nivolumab plus ipilimumab. Surgery was performed laparoscopically in five cases (Urol Oncol. 2019 Dec;37[12]:924-31).
No patient experienced a major intraoperative complication. Four experienced postoperative complications, the majority of which were addressed with interventional radiology procedures. The median hospital stay was 4 days. One patient died of progressive disease more than 3 months after surgery, and another died of pulmonary embolism and sepsis. Six of the 10 patients did not have any complications or readmissions. There were no immune-related toxicities and no wound-healing issues. ICI therapy was resumed postoperatively in six patients.
At nephrectomy (plus or minus metastatectomy), one patient achieved a response to immunotherapy in the primary tumor, and three of four patients who underwent resection of hepatic, pulmonary, or adrenal metastases had no detectable cancer. All surgical margins were negative.
During a median postoperative follow-up of 180 days, nephrectomy following ICI was safe. Pathologic response in both the primary tumor and metastatic sites was encouraging.
What this means in clinical practice
In medical school, all of us are admonished not to be afraid to unlearn something and to learn something new. Historically, nephrectomy was felt to be helpful in improving overall survival in patients with advanced RCC. Effective targeted therapies and ICIs have caused us to question the role of nephrectomy and its timing, since 20%-40% of patients who have apparently localized RCC at the time of nephrectomy develop recurrences within 3 years. Preoperative therapy could mitigate potentially aggressive tumor biology, treat micrometastatic disease, and help select patients who should not be treated surgically.
In the CARMENA trial of the treatment of advanced RCC patients with the tyrosine kinase inhibitor sunitinib versus nephrectomy followed by sunitinib, most patients could avoid nephrectomy without compromising survival (N Engl J Med. 2018; 379:417-27). Results were updated at the 2019 annual meeting of the American Society of Clinical Oncology. Overall, nephrectomy was not beneficial. However, delayed nephrectomy (after sunitinib) appeared be beneficial for good responders with only one IMDC (International Metastatic RCC Database Consortium) risk factor and only one metastatic site.
The small study by Dr. Singla and colleagues illustrates that nephrectomy is feasible after ICI, plus or minus anti-CTLA4-targeted treatment, and that favorable histologic results can be achieved. With ICI plus or minus anti-CTLA4-targeted treatment, no patient had progressive disease prior to surgery. This experience is germane in view of recently updated results of the CheckMate 214 trial, showing superior overall survival, response rates, and response duration for nivolumab plus ipilimumab, in comparison with sunitinib.
There are still unresolved questions, including whether these favorable outcomes can be achieved in community practice and whether there are genomic or immunohistochemistry expression profiles to select patients who can benefit from this approach. It’s unclear whether there are practical issues that influence outcome, such as type of ICI, number of preoperative treatment cycles, and additional systemic therapies including postoperative treatment. However, the current series rings the starting bell for the study of those questions and a promising era for patients with this deadly disease.
SABR in moderately central NSCLC
SABR to peripheral, small non–small cell lung cancers (NSCLCs) produces high local control rates, with low grade 3-4 toxicity, and is an alternative to resection in patients who are unfit for surgery. In a pragmatic, community-based, prospective cohort experience in Scotland, Robert Rulach, MBChB, and colleagues, treated 50 T1-2N0M0 NSCLC patients with SABR 50-Gy in five fractions (Clin Oncol. 2019 Oct 10. doi: 10.1016/j.clon.2019.09.055). The dose and fractionation schedule was safe and effective in the phase 1/2 RTOG 0813 trial and is concordant with guidelines from the National Comprehensive Cancer Network (NCCN).
All of the tumors were moderately central, as in the RTOG trial. One patient had an additional tumor that was ultracentral. Notably, 84% of patients were deemed medically unfit for surgery.
All patients completed radiotherapy without treatment delays. Two patients died within 90 days of treatment. There were no grade 4 or grade 5 toxicities and the overall rate of grade 3 toxicity was 4%. With a median follow-up of 25.2 months, 34 patients died: 18 from causes unrelated to cancer and 16 from cancer recurrence. The median overall survival was 27 months. The 2-year overall survival rate was 67.6%, commensurate with the rate seen in RTOG 0813.
The researchers concluded that, for frail patients with centrally-located NSCLC treated uniformly in a community practice, SABR with the RTOG 0813 treatment protocol produced acceptable toxicity and overall survival comparable with the published literature.
What this means in clinical practice
The results and conclusions of the study by Dr. Rulach and colleagues are straightforward: SABR can be used for centrally-located NSCLC without producing massive hemoptysis, bronchial stricture, and fistula formation. Since the majority of patients had no histologic diagnosis, T1N0 lesions, and no routine follow-up CT scans beyond 3 months post treatment, conclusions beyond that are unjustified.
In a community-based practice, NCCN guideline–concordant SABR treatment in moderately centrally-located NSCLC was safely delivered. For the burgeoning population of medically inoperable and/or elderly NSCLC patients, this alone is reassuring for clinicians and is helpful information for patients who require and/or desire nonsurgical treatment.
Dr. Lyss has been a community-based medical oncologist and clinical researcher for more than 35 years, practicing in St. Louis. His clinical and research interests are in the prevention, diagnosis, and treatment of breast and lung cancers and in expanding access to clinical trials to medically underserved populations.
In this edition of “How I Will Treat My Next Patient,” I highlight two articles that demonstrate the safety of established treatments – nephrectomy and stereotactic ablative body radiotherapy (SABR) – in patient populations that previously may have been excluded from those treatments at many centers.
Nephrectomy in advanced RCC
Nirmish Singla, MD, and colleagues reported a single-center retrospective cohort study, assessing outcomes of 11 nephrectomies (10 radical, 1 partial) in 10 patients with advanced renal cell carcinoma (RCC) who had received front- or later-line immune checkpoint inhibitor therapy (ICIs). Half had received nivolumab alone; the others received nivolumab plus ipilimumab. Surgery was performed laparoscopically in five cases (Urol Oncol. 2019 Dec;37[12]:924-31).
No patient experienced a major intraoperative complication. Four experienced postoperative complications, the majority of which were addressed with interventional radiology procedures. The median hospital stay was 4 days. One patient died of progressive disease more than 3 months after surgery, and another died of pulmonary embolism and sepsis. Six of the 10 patients did not have any complications or readmissions. There were no immune-related toxicities and no wound-healing issues. ICI therapy was resumed postoperatively in six patients.
At nephrectomy (plus or minus metastatectomy), one patient achieved a response to immunotherapy in the primary tumor, and three of four patients who underwent resection of hepatic, pulmonary, or adrenal metastases had no detectable cancer. All surgical margins were negative.
During a median postoperative follow-up of 180 days, nephrectomy following ICI was safe. Pathologic response in both the primary tumor and metastatic sites was encouraging.
What this means in clinical practice
In medical school, all of us are admonished not to be afraid to unlearn something and to learn something new. Historically, nephrectomy was felt to be helpful in improving overall survival in patients with advanced RCC. Effective targeted therapies and ICIs have caused us to question the role of nephrectomy and its timing, since 20%-40% of patients who have apparently localized RCC at the time of nephrectomy develop recurrences within 3 years. Preoperative therapy could mitigate potentially aggressive tumor biology, treat micrometastatic disease, and help select patients who should not be treated surgically.
In the CARMENA trial of the treatment of advanced RCC patients with the tyrosine kinase inhibitor sunitinib versus nephrectomy followed by sunitinib, most patients could avoid nephrectomy without compromising survival (N Engl J Med. 2018; 379:417-27). Results were updated at the 2019 annual meeting of the American Society of Clinical Oncology. Overall, nephrectomy was not beneficial. However, delayed nephrectomy (after sunitinib) appeared be beneficial for good responders with only one IMDC (International Metastatic RCC Database Consortium) risk factor and only one metastatic site.
The small study by Dr. Singla and colleagues illustrates that nephrectomy is feasible after ICI, plus or minus anti-CTLA4-targeted treatment, and that favorable histologic results can be achieved. With ICI plus or minus anti-CTLA4-targeted treatment, no patient had progressive disease prior to surgery. This experience is germane in view of recently updated results of the CheckMate 214 trial, showing superior overall survival, response rates, and response duration for nivolumab plus ipilimumab, in comparison with sunitinib.
There are still unresolved questions, including whether these favorable outcomes can be achieved in community practice and whether there are genomic or immunohistochemistry expression profiles to select patients who can benefit from this approach. It’s unclear whether there are practical issues that influence outcome, such as type of ICI, number of preoperative treatment cycles, and additional systemic therapies including postoperative treatment. However, the current series rings the starting bell for the study of those questions and a promising era for patients with this deadly disease.
SABR in moderately central NSCLC
SABR to peripheral, small non–small cell lung cancers (NSCLCs) produces high local control rates, with low grade 3-4 toxicity, and is an alternative to resection in patients who are unfit for surgery. In a pragmatic, community-based, prospective cohort experience in Scotland, Robert Rulach, MBChB, and colleagues, treated 50 T1-2N0M0 NSCLC patients with SABR 50-Gy in five fractions (Clin Oncol. 2019 Oct 10. doi: 10.1016/j.clon.2019.09.055). The dose and fractionation schedule was safe and effective in the phase 1/2 RTOG 0813 trial and is concordant with guidelines from the National Comprehensive Cancer Network (NCCN).
All of the tumors were moderately central, as in the RTOG trial. One patient had an additional tumor that was ultracentral. Notably, 84% of patients were deemed medically unfit for surgery.
All patients completed radiotherapy without treatment delays. Two patients died within 90 days of treatment. There were no grade 4 or grade 5 toxicities and the overall rate of grade 3 toxicity was 4%. With a median follow-up of 25.2 months, 34 patients died: 18 from causes unrelated to cancer and 16 from cancer recurrence. The median overall survival was 27 months. The 2-year overall survival rate was 67.6%, commensurate with the rate seen in RTOG 0813.
The researchers concluded that, for frail patients with centrally-located NSCLC treated uniformly in a community practice, SABR with the RTOG 0813 treatment protocol produced acceptable toxicity and overall survival comparable with the published literature.
What this means in clinical practice
The results and conclusions of the study by Dr. Rulach and colleagues are straightforward: SABR can be used for centrally-located NSCLC without producing massive hemoptysis, bronchial stricture, and fistula formation. Since the majority of patients had no histologic diagnosis, T1N0 lesions, and no routine follow-up CT scans beyond 3 months post treatment, conclusions beyond that are unjustified.
In a community-based practice, NCCN guideline–concordant SABR treatment in moderately centrally-located NSCLC was safely delivered. For the burgeoning population of medically inoperable and/or elderly NSCLC patients, this alone is reassuring for clinicians and is helpful information for patients who require and/or desire nonsurgical treatment.
Dr. Lyss has been a community-based medical oncologist and clinical researcher for more than 35 years, practicing in St. Louis. His clinical and research interests are in the prevention, diagnosis, and treatment of breast and lung cancers and in expanding access to clinical trials to medically underserved populations.
In this edition of “How I Will Treat My Next Patient,” I highlight two articles that demonstrate the safety of established treatments – nephrectomy and stereotactic ablative body radiotherapy (SABR) – in patient populations that previously may have been excluded from those treatments at many centers.
Nephrectomy in advanced RCC
Nirmish Singla, MD, and colleagues reported a single-center retrospective cohort study, assessing outcomes of 11 nephrectomies (10 radical, 1 partial) in 10 patients with advanced renal cell carcinoma (RCC) who had received front- or later-line immune checkpoint inhibitor therapy (ICIs). Half had received nivolumab alone; the others received nivolumab plus ipilimumab. Surgery was performed laparoscopically in five cases (Urol Oncol. 2019 Dec;37[12]:924-31).
No patient experienced a major intraoperative complication. Four experienced postoperative complications, the majority of which were addressed with interventional radiology procedures. The median hospital stay was 4 days. One patient died of progressive disease more than 3 months after surgery, and another died of pulmonary embolism and sepsis. Six of the 10 patients did not have any complications or readmissions. There were no immune-related toxicities and no wound-healing issues. ICI therapy was resumed postoperatively in six patients.
At nephrectomy (plus or minus metastatectomy), one patient achieved a response to immunotherapy in the primary tumor, and three of four patients who underwent resection of hepatic, pulmonary, or adrenal metastases had no detectable cancer. All surgical margins were negative.
During a median postoperative follow-up of 180 days, nephrectomy following ICI was safe. Pathologic response in both the primary tumor and metastatic sites was encouraging.
What this means in clinical practice
In medical school, all of us are admonished not to be afraid to unlearn something and to learn something new. Historically, nephrectomy was felt to be helpful in improving overall survival in patients with advanced RCC. Effective targeted therapies and ICIs have caused us to question the role of nephrectomy and its timing, since 20%-40% of patients who have apparently localized RCC at the time of nephrectomy develop recurrences within 3 years. Preoperative therapy could mitigate potentially aggressive tumor biology, treat micrometastatic disease, and help select patients who should not be treated surgically.
In the CARMENA trial of the treatment of advanced RCC patients with the tyrosine kinase inhibitor sunitinib versus nephrectomy followed by sunitinib, most patients could avoid nephrectomy without compromising survival (N Engl J Med. 2018; 379:417-27). Results were updated at the 2019 annual meeting of the American Society of Clinical Oncology. Overall, nephrectomy was not beneficial. However, delayed nephrectomy (after sunitinib) appeared be beneficial for good responders with only one IMDC (International Metastatic RCC Database Consortium) risk factor and only one metastatic site.
The small study by Dr. Singla and colleagues illustrates that nephrectomy is feasible after ICI, plus or minus anti-CTLA4-targeted treatment, and that favorable histologic results can be achieved. With ICI plus or minus anti-CTLA4-targeted treatment, no patient had progressive disease prior to surgery. This experience is germane in view of recently updated results of the CheckMate 214 trial, showing superior overall survival, response rates, and response duration for nivolumab plus ipilimumab, in comparison with sunitinib.
There are still unresolved questions, including whether these favorable outcomes can be achieved in community practice and whether there are genomic or immunohistochemistry expression profiles to select patients who can benefit from this approach. It’s unclear whether there are practical issues that influence outcome, such as type of ICI, number of preoperative treatment cycles, and additional systemic therapies including postoperative treatment. However, the current series rings the starting bell for the study of those questions and a promising era for patients with this deadly disease.
SABR in moderately central NSCLC
SABR to peripheral, small non–small cell lung cancers (NSCLCs) produces high local control rates, with low grade 3-4 toxicity, and is an alternative to resection in patients who are unfit for surgery. In a pragmatic, community-based, prospective cohort experience in Scotland, Robert Rulach, MBChB, and colleagues, treated 50 T1-2N0M0 NSCLC patients with SABR 50-Gy in five fractions (Clin Oncol. 2019 Oct 10. doi: 10.1016/j.clon.2019.09.055). The dose and fractionation schedule was safe and effective in the phase 1/2 RTOG 0813 trial and is concordant with guidelines from the National Comprehensive Cancer Network (NCCN).
All of the tumors were moderately central, as in the RTOG trial. One patient had an additional tumor that was ultracentral. Notably, 84% of patients were deemed medically unfit for surgery.
All patients completed radiotherapy without treatment delays. Two patients died within 90 days of treatment. There were no grade 4 or grade 5 toxicities and the overall rate of grade 3 toxicity was 4%. With a median follow-up of 25.2 months, 34 patients died: 18 from causes unrelated to cancer and 16 from cancer recurrence. The median overall survival was 27 months. The 2-year overall survival rate was 67.6%, commensurate with the rate seen in RTOG 0813.
The researchers concluded that, for frail patients with centrally-located NSCLC treated uniformly in a community practice, SABR with the RTOG 0813 treatment protocol produced acceptable toxicity and overall survival comparable with the published literature.
What this means in clinical practice
The results and conclusions of the study by Dr. Rulach and colleagues are straightforward: SABR can be used for centrally-located NSCLC without producing massive hemoptysis, bronchial stricture, and fistula formation. Since the majority of patients had no histologic diagnosis, T1N0 lesions, and no routine follow-up CT scans beyond 3 months post treatment, conclusions beyond that are unjustified.
In a community-based practice, NCCN guideline–concordant SABR treatment in moderately centrally-located NSCLC was safely delivered. For the burgeoning population of medically inoperable and/or elderly NSCLC patients, this alone is reassuring for clinicians and is helpful information for patients who require and/or desire nonsurgical treatment.
Dr. Lyss has been a community-based medical oncologist and clinical researcher for more than 35 years, practicing in St. Louis. His clinical and research interests are in the prevention, diagnosis, and treatment of breast and lung cancers and in expanding access to clinical trials to medically underserved populations.
Not all lung cancer patients receive treatment
In the United States, just over 15% of patients with lung cancer receive no treatment, according to the American Lung Association.
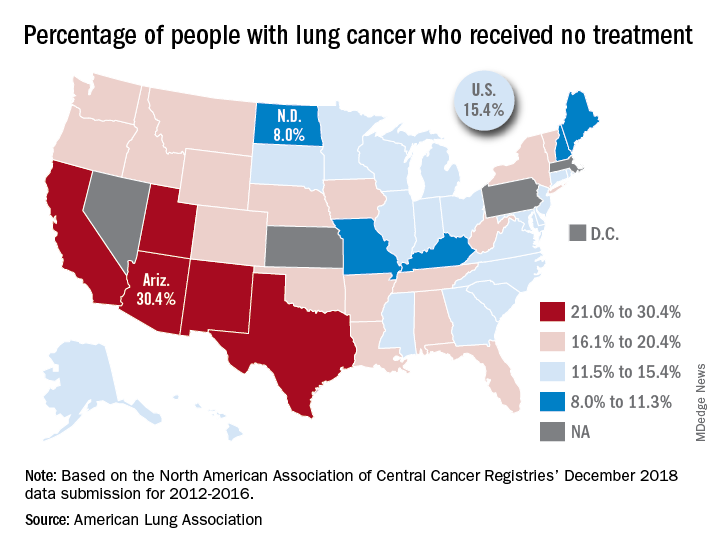
“This can happen for multiple reasons, such as the tumor having spread too far, poor health, or refusal of treatment,” the ALA said in its 2019 State of Lung Cancer report.
On the state level, the disparities were considerable. Arizona had the highest rate of nontreatment at 30.4%, followed by the neighboring states of New Mexico (24.2%) and California (24.0%). The lowest rate in the country, 8.0%, came from North Dakota, with Missouri next at 9.4% and Maine third at 9.6%, based on data from the North American Association of Central Cancer Registries’ December 2018 data submission, which covered the years from 2012 to 2016.
Although some cases of lung cancer may be unavoidable, “no one should go untreated because of lack of provider or patient knowledge, stigma associated with lung cancer, fatalism after diagnosis, or cost of treatment. Dismantling these and other barriers is important to reducing the percent of untreated patients,” the ALA said.
In the United States, just over 15% of patients with lung cancer receive no treatment, according to the American Lung Association.

“This can happen for multiple reasons, such as the tumor having spread too far, poor health, or refusal of treatment,” the ALA said in its 2019 State of Lung Cancer report.
On the state level, the disparities were considerable. Arizona had the highest rate of nontreatment at 30.4%, followed by the neighboring states of New Mexico (24.2%) and California (24.0%). The lowest rate in the country, 8.0%, came from North Dakota, with Missouri next at 9.4% and Maine third at 9.6%, based on data from the North American Association of Central Cancer Registries’ December 2018 data submission, which covered the years from 2012 to 2016.
Although some cases of lung cancer may be unavoidable, “no one should go untreated because of lack of provider or patient knowledge, stigma associated with lung cancer, fatalism after diagnosis, or cost of treatment. Dismantling these and other barriers is important to reducing the percent of untreated patients,” the ALA said.
In the United States, just over 15% of patients with lung cancer receive no treatment, according to the American Lung Association.

“This can happen for multiple reasons, such as the tumor having spread too far, poor health, or refusal of treatment,” the ALA said in its 2019 State of Lung Cancer report.
On the state level, the disparities were considerable. Arizona had the highest rate of nontreatment at 30.4%, followed by the neighboring states of New Mexico (24.2%) and California (24.0%). The lowest rate in the country, 8.0%, came from North Dakota, with Missouri next at 9.4% and Maine third at 9.6%, based on data from the North American Association of Central Cancer Registries’ December 2018 data submission, which covered the years from 2012 to 2016.
Although some cases of lung cancer may be unavoidable, “no one should go untreated because of lack of provider or patient knowledge, stigma associated with lung cancer, fatalism after diagnosis, or cost of treatment. Dismantling these and other barriers is important to reducing the percent of untreated patients,” the ALA said.
Neoantigen vaccine appears safe and active in NSCLC
NATIONAL HARBOR, MD. – Trial results suggest a personalized vaccination approach is feasible and safe, and the vaccine can produce clinical responses in patients with non–small cell lung cancer (NSCLC).
The neoantigen vaccine produced only grade 1 adverse events, yielded responses in patients with epidermal growth factor receptor (EGFR) mutations, and proved particularly effective in patients who were also receiving an EGFR inhibitor.
“EGFR inhibitors seemed to reduce tumor immunosuppression barriers and may enhance antitumor immune responses before and during immunization, suggesting there may be a potential synergy of EGFR with immunotherapies,” Gregory A. Lizee, PhD, of University of Texas MD Anderson Cancer Center, Houston, said at the annual meeting of the Society for Immunotherapy of Cancer.
The research began with an elderly patient who had heavily pretreated NSCLC (Oncoimmunology. 2016;5[12]:e1238539). Dr. Lizee and colleagues used tumor mutational profiling and human leukocyte antigen (HLA) typing to develop a personalized peptide vaccine for the patient. He received the vaccine along with topical imiquimod and had multiple lung tumor nodules regress. However, the patient also had liver metastasis that remained refractory to treatment, and he ultimately died.
To investigate this treatment approach in a larger group, Dr. Lizee and colleagues began a phase 1b trial of patients with advanced NSCLC (ChiCTR-IIR-16009867). As with the prior patient, the researchers designed personalized peptide vaccines for the trial subjects based on mutational profiling of 508 cancer-associated genes and high-resolution HLA typing. The peptides were selected based on nonsynonymous somatic tumor–associated mutations with variant allele frequency greater than 0.04 and the highest predicted neoantigen peptide binding to each patient’s HLA class I and II molecules. The vaccines targeted up to eight independent somatic mutations (mean, 3.75 mutations).
In all, 31 patients provided lung tumor biopsies and peripheral blood for mutational and HLA analyses. The researchers designed 27 personalized neoantigen vaccines, and 24 patients were ultimately vaccinated. This translates to a vaccination rate of 77%, which suggests this treatment approach is feasible, Dr. Lizee said.
Of the 24 vaccinated patients, 18 had adenocarcinoma, and 6 had squamous cell carcinoma. All patients had received multiple prior therapies, including surgery, chemotherapy, radiation, and EGFR inhibitors.
Each patient was vaccinated with a personalized mixture of short and long neoantigen peptides (mean, 9.4 peptides) dissolved in isotonic saline. Patients received at least 12 weekly immunizations and had topical imiquimod applied over the injection site for costimulation through toll-like receptor 7. The 16 patients with EGFR mutations were given the option of continuing on an EGFR inhibitor, and 9 patients elected to do so.
Results
Dr. Lizee said this treatment approach was “very safe,” with only grade 1 treatment-related adverse events. The events were fatigue (n = 2), rash (n = 1), and fever (n = 1).
Seven patients achieved a response after vaccination, and one patient achieved a complete response. All seven responders had EGFR mutations, and four of them were receiving an EGFR inhibitor.
The patients on an EGFR inhibitor had significantly better overall survival than that of EGFR-mutated patients who had stopped taking an EGFR inhibitor – 13.8 months and 7.6 months, respectively (P = .038).
Immune profiling revealed that neoantigen-specific T-cell reactivity was associated with clinical responses. The researchers observed EGFR neoantigen-specific T-cell responses in five responders. In three responders, the strongest response was against a peptide encompassing the L858R driver mutation.
The researchers also found evidence of synergy between EGFR inhibitor therapy and the peptide vaccine. EGFR inhibition caused immunomodulatory pathways in EGFR-mutated cancer cells to favor immune-cell infiltration and HLA-mediated antigen presentation.
“Our mechanistic working model is that, in the circulation, the personalized vaccine increased the T-cell frequency,” Dr. Lizee said. “The EGFR inhibitor increased chemokines and antigen presentation at the tumor site, which then attracted those T cells to migrate to the tumor. Then, recognition of the antigen caused interferon gamma [to increase], which caused, potentially, a feed-forward loop by increasing chemokines and antigen presentation further.”
This research is sponsored by Tianjin Beichen Hospital and funded by Tianjin HengJia Biotechnology Development Co. Ltd. Dr. Lizee disclosed a consulting relationship with Tianjin HengJia Biotechnology Development Co. Ltd.
SOURCE: Lizee G et al. SITC 2019. Abstract O18.
NATIONAL HARBOR, MD. – Trial results suggest a personalized vaccination approach is feasible and safe, and the vaccine can produce clinical responses in patients with non–small cell lung cancer (NSCLC).
The neoantigen vaccine produced only grade 1 adverse events, yielded responses in patients with epidermal growth factor receptor (EGFR) mutations, and proved particularly effective in patients who were also receiving an EGFR inhibitor.
“EGFR inhibitors seemed to reduce tumor immunosuppression barriers and may enhance antitumor immune responses before and during immunization, suggesting there may be a potential synergy of EGFR with immunotherapies,” Gregory A. Lizee, PhD, of University of Texas MD Anderson Cancer Center, Houston, said at the annual meeting of the Society for Immunotherapy of Cancer.
The research began with an elderly patient who had heavily pretreated NSCLC (Oncoimmunology. 2016;5[12]:e1238539). Dr. Lizee and colleagues used tumor mutational profiling and human leukocyte antigen (HLA) typing to develop a personalized peptide vaccine for the patient. He received the vaccine along with topical imiquimod and had multiple lung tumor nodules regress. However, the patient also had liver metastasis that remained refractory to treatment, and he ultimately died.
To investigate this treatment approach in a larger group, Dr. Lizee and colleagues began a phase 1b trial of patients with advanced NSCLC (ChiCTR-IIR-16009867). As with the prior patient, the researchers designed personalized peptide vaccines for the trial subjects based on mutational profiling of 508 cancer-associated genes and high-resolution HLA typing. The peptides were selected based on nonsynonymous somatic tumor–associated mutations with variant allele frequency greater than 0.04 and the highest predicted neoantigen peptide binding to each patient’s HLA class I and II molecules. The vaccines targeted up to eight independent somatic mutations (mean, 3.75 mutations).
In all, 31 patients provided lung tumor biopsies and peripheral blood for mutational and HLA analyses. The researchers designed 27 personalized neoantigen vaccines, and 24 patients were ultimately vaccinated. This translates to a vaccination rate of 77%, which suggests this treatment approach is feasible, Dr. Lizee said.
Of the 24 vaccinated patients, 18 had adenocarcinoma, and 6 had squamous cell carcinoma. All patients had received multiple prior therapies, including surgery, chemotherapy, radiation, and EGFR inhibitors.
Each patient was vaccinated with a personalized mixture of short and long neoantigen peptides (mean, 9.4 peptides) dissolved in isotonic saline. Patients received at least 12 weekly immunizations and had topical imiquimod applied over the injection site for costimulation through toll-like receptor 7. The 16 patients with EGFR mutations were given the option of continuing on an EGFR inhibitor, and 9 patients elected to do so.
Results
Dr. Lizee said this treatment approach was “very safe,” with only grade 1 treatment-related adverse events. The events were fatigue (n = 2), rash (n = 1), and fever (n = 1).
Seven patients achieved a response after vaccination, and one patient achieved a complete response. All seven responders had EGFR mutations, and four of them were receiving an EGFR inhibitor.
The patients on an EGFR inhibitor had significantly better overall survival than that of EGFR-mutated patients who had stopped taking an EGFR inhibitor – 13.8 months and 7.6 months, respectively (P = .038).
Immune profiling revealed that neoantigen-specific T-cell reactivity was associated with clinical responses. The researchers observed EGFR neoantigen-specific T-cell responses in five responders. In three responders, the strongest response was against a peptide encompassing the L858R driver mutation.
The researchers also found evidence of synergy between EGFR inhibitor therapy and the peptide vaccine. EGFR inhibition caused immunomodulatory pathways in EGFR-mutated cancer cells to favor immune-cell infiltration and HLA-mediated antigen presentation.
“Our mechanistic working model is that, in the circulation, the personalized vaccine increased the T-cell frequency,” Dr. Lizee said. “The EGFR inhibitor increased chemokines and antigen presentation at the tumor site, which then attracted those T cells to migrate to the tumor. Then, recognition of the antigen caused interferon gamma [to increase], which caused, potentially, a feed-forward loop by increasing chemokines and antigen presentation further.”
This research is sponsored by Tianjin Beichen Hospital and funded by Tianjin HengJia Biotechnology Development Co. Ltd. Dr. Lizee disclosed a consulting relationship with Tianjin HengJia Biotechnology Development Co. Ltd.
SOURCE: Lizee G et al. SITC 2019. Abstract O18.
NATIONAL HARBOR, MD. – Trial results suggest a personalized vaccination approach is feasible and safe, and the vaccine can produce clinical responses in patients with non–small cell lung cancer (NSCLC).
The neoantigen vaccine produced only grade 1 adverse events, yielded responses in patients with epidermal growth factor receptor (EGFR) mutations, and proved particularly effective in patients who were also receiving an EGFR inhibitor.
“EGFR inhibitors seemed to reduce tumor immunosuppression barriers and may enhance antitumor immune responses before and during immunization, suggesting there may be a potential synergy of EGFR with immunotherapies,” Gregory A. Lizee, PhD, of University of Texas MD Anderson Cancer Center, Houston, said at the annual meeting of the Society for Immunotherapy of Cancer.
The research began with an elderly patient who had heavily pretreated NSCLC (Oncoimmunology. 2016;5[12]:e1238539). Dr. Lizee and colleagues used tumor mutational profiling and human leukocyte antigen (HLA) typing to develop a personalized peptide vaccine for the patient. He received the vaccine along with topical imiquimod and had multiple lung tumor nodules regress. However, the patient also had liver metastasis that remained refractory to treatment, and he ultimately died.
To investigate this treatment approach in a larger group, Dr. Lizee and colleagues began a phase 1b trial of patients with advanced NSCLC (ChiCTR-IIR-16009867). As with the prior patient, the researchers designed personalized peptide vaccines for the trial subjects based on mutational profiling of 508 cancer-associated genes and high-resolution HLA typing. The peptides were selected based on nonsynonymous somatic tumor–associated mutations with variant allele frequency greater than 0.04 and the highest predicted neoantigen peptide binding to each patient’s HLA class I and II molecules. The vaccines targeted up to eight independent somatic mutations (mean, 3.75 mutations).
In all, 31 patients provided lung tumor biopsies and peripheral blood for mutational and HLA analyses. The researchers designed 27 personalized neoantigen vaccines, and 24 patients were ultimately vaccinated. This translates to a vaccination rate of 77%, which suggests this treatment approach is feasible, Dr. Lizee said.
Of the 24 vaccinated patients, 18 had adenocarcinoma, and 6 had squamous cell carcinoma. All patients had received multiple prior therapies, including surgery, chemotherapy, radiation, and EGFR inhibitors.
Each patient was vaccinated with a personalized mixture of short and long neoantigen peptides (mean, 9.4 peptides) dissolved in isotonic saline. Patients received at least 12 weekly immunizations and had topical imiquimod applied over the injection site for costimulation through toll-like receptor 7. The 16 patients with EGFR mutations were given the option of continuing on an EGFR inhibitor, and 9 patients elected to do so.
Results
Dr. Lizee said this treatment approach was “very safe,” with only grade 1 treatment-related adverse events. The events were fatigue (n = 2), rash (n = 1), and fever (n = 1).
Seven patients achieved a response after vaccination, and one patient achieved a complete response. All seven responders had EGFR mutations, and four of them were receiving an EGFR inhibitor.
The patients on an EGFR inhibitor had significantly better overall survival than that of EGFR-mutated patients who had stopped taking an EGFR inhibitor – 13.8 months and 7.6 months, respectively (P = .038).
Immune profiling revealed that neoantigen-specific T-cell reactivity was associated with clinical responses. The researchers observed EGFR neoantigen-specific T-cell responses in five responders. In three responders, the strongest response was against a peptide encompassing the L858R driver mutation.
The researchers also found evidence of synergy between EGFR inhibitor therapy and the peptide vaccine. EGFR inhibition caused immunomodulatory pathways in EGFR-mutated cancer cells to favor immune-cell infiltration and HLA-mediated antigen presentation.
“Our mechanistic working model is that, in the circulation, the personalized vaccine increased the T-cell frequency,” Dr. Lizee said. “The EGFR inhibitor increased chemokines and antigen presentation at the tumor site, which then attracted those T cells to migrate to the tumor. Then, recognition of the antigen caused interferon gamma [to increase], which caused, potentially, a feed-forward loop by increasing chemokines and antigen presentation further.”
This research is sponsored by Tianjin Beichen Hospital and funded by Tianjin HengJia Biotechnology Development Co. Ltd. Dr. Lizee disclosed a consulting relationship with Tianjin HengJia Biotechnology Development Co. Ltd.
SOURCE: Lizee G et al. SITC 2019. Abstract O18.
REPORTING FROM SITC 2019
Lorlatinib induces deep responses in ROS1-positive NSCLC
The tyrosine kinase inhibitor (TKI) lorlatinib showed deep responses and intracranial activity in both TKI-pretreated and TKI-naive patients with advanced ROS1-positive non–small cell lung cancer (NSCLC), according to results from a phase 1-2 trial.
“We investigated the antitumour activity and safety of lorlatinib in advanced, ROS1-positive NSCLC,” wrote Alice T. Shaw, MD, PhD, of Massachusetts General Hospital Cancer Center, Boston, and colleagues. Their report is in The Lancet Oncology.
The single-arm, open-label study included 69 patients with advanced ROS1-positive disease with or without CNS involvement. The effects of lorlatinib were evaluated across 28 institutions in 12 different countries around the globe.
At baseline, the median age of study participants was 54 years (range, 44-61 years), and 57% were positive for brain metastases.
Study participants received 100 mg of oral lorlatinib once daily in repeated 21-day cycles. Drug therapy was continued until death, disease progression, unacceptable toxicity, or withdrawal of consent.
The primary outcome measured was intracranial and overall response. Activity outcomes were evaluated in subjects given a minimum of one dose of lorlatinib.
A total of 58% of patients were previously treated with crizotinib, while 30% of patients were TKI-naive. Among 40 crizotinib-pretreated patients, 14 patients (35%) had an objective response, with a median duration of response and PFS of 13.8 and 8.5 months, respectively.
Among 21 TKI-naive patients, 13 patients (62%) had an objective response, with a median duration of response and PFS of 25.3 and 21 months, respectively.
“Intracranial responses were achieved in seven (64%) of 11 TKI-naive patients and 12 (50%) of 24 previous crizotinib-only patients,” they reported.
With respect to safety, serious lorlatinib-related adverse events were observed in 7% of patients, with no therapy-related deaths reported. The most frequently seen grade 3-4 TEAEs were hypertriglyceridemia (19%) and hypercholesterolemia (14%).
The researchers noted a key limitation of the study was the small sample size; however, due to the rare nature of ROS1 rearrangements in patients with NSCLC, increasing enrollment for future studies could be challenging.
“Because crizotinib-refractory patients have few treatment options, lorlatinib could represent an important next-line targeted agent,” they concluded.
Pfizer funded the study. The authors reported financial affiliations with Ariad, Blueprint Medicines, Chugai Pharmaceutical, Daiichi Sankyo, EMD Serono, Pfizer, KSQ Therapeutics, Servier, TP Therapeutics, and other companies.
SOURCE: Shaw AT et al. Lancet Oncol. 2019 Oct 25. doi: 10.1016/S1470-2045(19)30655-2.
The tyrosine kinase inhibitor (TKI) crizotinib was recently established as an optimal first-line treatment option for patients with ROS1-positive non–small cell lung cancer (NSCLC). Despite strong efficacy seen in clinical trials, disease progression can still occur in patients on crizotinib, often through the development of resistance, which is largely the result of on-target mutations, such as Gly2032Arg.
Early results suggest the novel oral TKI candidate, lorlatinib, a potent inhibitor of the Gly2032Arg mutation, may be a treatment of choice in patients with crizotinib-resistance. Recent phase 1 data showed lorlatinib had antitumor activity in ROS1-positive patients.
Correspondingly, the deep and durable responses reported by Dr. Shaw and colleagues represents a significant milestone for lorlatinib, particularly in the setting of crizotinib resistance, where a paucity of later-line treatment options exist. In comparison to platinum-pemetrexed chemotherapy, lorlatinib is better tolerated and has demonstrated potent intracranial activity, which may prevent or delay CNS progression in the disease.
One question that remains from the current study is whether other ROS1 TKI drug candidates, such as repotrectinib and entrectinib, will show similar results to lorlatinib. Several trials are presently ongoing in an attempt to help answer this, and other remaining questions.
Michaël Duruisseaux, MD, PhD, is affiliated with the Hospices Civils de Lyon (France), Universit é Claude Bernard Lyon. Dr. Duruisseaux reported financial affiliations with Boehringer Ingelheim, Bristol-Myers Squibb, Roche, and Takeda. These comments are adapted from his editorial (Lancet Oncol. 2019 Oct 25. doi: 10.1016/S1470-2045[19]30716-8 ).
The tyrosine kinase inhibitor (TKI) crizotinib was recently established as an optimal first-line treatment option for patients with ROS1-positive non–small cell lung cancer (NSCLC). Despite strong efficacy seen in clinical trials, disease progression can still occur in patients on crizotinib, often through the development of resistance, which is largely the result of on-target mutations, such as Gly2032Arg.
Early results suggest the novel oral TKI candidate, lorlatinib, a potent inhibitor of the Gly2032Arg mutation, may be a treatment of choice in patients with crizotinib-resistance. Recent phase 1 data showed lorlatinib had antitumor activity in ROS1-positive patients.
Correspondingly, the deep and durable responses reported by Dr. Shaw and colleagues represents a significant milestone for lorlatinib, particularly in the setting of crizotinib resistance, where a paucity of later-line treatment options exist. In comparison to platinum-pemetrexed chemotherapy, lorlatinib is better tolerated and has demonstrated potent intracranial activity, which may prevent or delay CNS progression in the disease.
One question that remains from the current study is whether other ROS1 TKI drug candidates, such as repotrectinib and entrectinib, will show similar results to lorlatinib. Several trials are presently ongoing in an attempt to help answer this, and other remaining questions.
Michaël Duruisseaux, MD, PhD, is affiliated with the Hospices Civils de Lyon (France), Universit é Claude Bernard Lyon. Dr. Duruisseaux reported financial affiliations with Boehringer Ingelheim, Bristol-Myers Squibb, Roche, and Takeda. These comments are adapted from his editorial (Lancet Oncol. 2019 Oct 25. doi: 10.1016/S1470-2045[19]30716-8 ).
The tyrosine kinase inhibitor (TKI) crizotinib was recently established as an optimal first-line treatment option for patients with ROS1-positive non–small cell lung cancer (NSCLC). Despite strong efficacy seen in clinical trials, disease progression can still occur in patients on crizotinib, often through the development of resistance, which is largely the result of on-target mutations, such as Gly2032Arg.
Early results suggest the novel oral TKI candidate, lorlatinib, a potent inhibitor of the Gly2032Arg mutation, may be a treatment of choice in patients with crizotinib-resistance. Recent phase 1 data showed lorlatinib had antitumor activity in ROS1-positive patients.
Correspondingly, the deep and durable responses reported by Dr. Shaw and colleagues represents a significant milestone for lorlatinib, particularly in the setting of crizotinib resistance, where a paucity of later-line treatment options exist. In comparison to platinum-pemetrexed chemotherapy, lorlatinib is better tolerated and has demonstrated potent intracranial activity, which may prevent or delay CNS progression in the disease.
One question that remains from the current study is whether other ROS1 TKI drug candidates, such as repotrectinib and entrectinib, will show similar results to lorlatinib. Several trials are presently ongoing in an attempt to help answer this, and other remaining questions.
Michaël Duruisseaux, MD, PhD, is affiliated with the Hospices Civils de Lyon (France), Universit é Claude Bernard Lyon. Dr. Duruisseaux reported financial affiliations with Boehringer Ingelheim, Bristol-Myers Squibb, Roche, and Takeda. These comments are adapted from his editorial (Lancet Oncol. 2019 Oct 25. doi: 10.1016/S1470-2045[19]30716-8 ).
The tyrosine kinase inhibitor (TKI) lorlatinib showed deep responses and intracranial activity in both TKI-pretreated and TKI-naive patients with advanced ROS1-positive non–small cell lung cancer (NSCLC), according to results from a phase 1-2 trial.
“We investigated the antitumour activity and safety of lorlatinib in advanced, ROS1-positive NSCLC,” wrote Alice T. Shaw, MD, PhD, of Massachusetts General Hospital Cancer Center, Boston, and colleagues. Their report is in The Lancet Oncology.
The single-arm, open-label study included 69 patients with advanced ROS1-positive disease with or without CNS involvement. The effects of lorlatinib were evaluated across 28 institutions in 12 different countries around the globe.
At baseline, the median age of study participants was 54 years (range, 44-61 years), and 57% were positive for brain metastases.
Study participants received 100 mg of oral lorlatinib once daily in repeated 21-day cycles. Drug therapy was continued until death, disease progression, unacceptable toxicity, or withdrawal of consent.
The primary outcome measured was intracranial and overall response. Activity outcomes were evaluated in subjects given a minimum of one dose of lorlatinib.
A total of 58% of patients were previously treated with crizotinib, while 30% of patients were TKI-naive. Among 40 crizotinib-pretreated patients, 14 patients (35%) had an objective response, with a median duration of response and PFS of 13.8 and 8.5 months, respectively.
Among 21 TKI-naive patients, 13 patients (62%) had an objective response, with a median duration of response and PFS of 25.3 and 21 months, respectively.
“Intracranial responses were achieved in seven (64%) of 11 TKI-naive patients and 12 (50%) of 24 previous crizotinib-only patients,” they reported.
With respect to safety, serious lorlatinib-related adverse events were observed in 7% of patients, with no therapy-related deaths reported. The most frequently seen grade 3-4 TEAEs were hypertriglyceridemia (19%) and hypercholesterolemia (14%).
The researchers noted a key limitation of the study was the small sample size; however, due to the rare nature of ROS1 rearrangements in patients with NSCLC, increasing enrollment for future studies could be challenging.
“Because crizotinib-refractory patients have few treatment options, lorlatinib could represent an important next-line targeted agent,” they concluded.
Pfizer funded the study. The authors reported financial affiliations with Ariad, Blueprint Medicines, Chugai Pharmaceutical, Daiichi Sankyo, EMD Serono, Pfizer, KSQ Therapeutics, Servier, TP Therapeutics, and other companies.
SOURCE: Shaw AT et al. Lancet Oncol. 2019 Oct 25. doi: 10.1016/S1470-2045(19)30655-2.
The tyrosine kinase inhibitor (TKI) lorlatinib showed deep responses and intracranial activity in both TKI-pretreated and TKI-naive patients with advanced ROS1-positive non–small cell lung cancer (NSCLC), according to results from a phase 1-2 trial.
“We investigated the antitumour activity and safety of lorlatinib in advanced, ROS1-positive NSCLC,” wrote Alice T. Shaw, MD, PhD, of Massachusetts General Hospital Cancer Center, Boston, and colleagues. Their report is in The Lancet Oncology.
The single-arm, open-label study included 69 patients with advanced ROS1-positive disease with or without CNS involvement. The effects of lorlatinib were evaluated across 28 institutions in 12 different countries around the globe.
At baseline, the median age of study participants was 54 years (range, 44-61 years), and 57% were positive for brain metastases.
Study participants received 100 mg of oral lorlatinib once daily in repeated 21-day cycles. Drug therapy was continued until death, disease progression, unacceptable toxicity, or withdrawal of consent.
The primary outcome measured was intracranial and overall response. Activity outcomes were evaluated in subjects given a minimum of one dose of lorlatinib.
A total of 58% of patients were previously treated with crizotinib, while 30% of patients were TKI-naive. Among 40 crizotinib-pretreated patients, 14 patients (35%) had an objective response, with a median duration of response and PFS of 13.8 and 8.5 months, respectively.
Among 21 TKI-naive patients, 13 patients (62%) had an objective response, with a median duration of response and PFS of 25.3 and 21 months, respectively.
“Intracranial responses were achieved in seven (64%) of 11 TKI-naive patients and 12 (50%) of 24 previous crizotinib-only patients,” they reported.
With respect to safety, serious lorlatinib-related adverse events were observed in 7% of patients, with no therapy-related deaths reported. The most frequently seen grade 3-4 TEAEs were hypertriglyceridemia (19%) and hypercholesterolemia (14%).
The researchers noted a key limitation of the study was the small sample size; however, due to the rare nature of ROS1 rearrangements in patients with NSCLC, increasing enrollment for future studies could be challenging.
“Because crizotinib-refractory patients have few treatment options, lorlatinib could represent an important next-line targeted agent,” they concluded.
Pfizer funded the study. The authors reported financial affiliations with Ariad, Blueprint Medicines, Chugai Pharmaceutical, Daiichi Sankyo, EMD Serono, Pfizer, KSQ Therapeutics, Servier, TP Therapeutics, and other companies.
SOURCE: Shaw AT et al. Lancet Oncol. 2019 Oct 25. doi: 10.1016/S1470-2045(19)30655-2.
FROM THE LANCET ONCOLOGY



